User login
The Starting Line of a New Era
AMSUS, the Society of Federal Health Professionals, is holding its 2015 Annual Continuing Education Meeting from December 1-4 in San Antonio, Texas, and has issued a call for lecture and poster abstracts for consideration. The conference is the largest health care meeting focused on federal providers and is expected to draw more than 1,000 attendees from across the VA, DoD, PHS, NIH, CDC, and other federal agencies.
The theme for this year’s meeting is “the New Normal,” reflecting the reality that federal medicine has just come through an extraordinary time of change and is settling into its future. Concurrent and dramatic events in global politics, the evolution of new technologies, and advances in the organization of federal medicine have drawn the starting line of a new era. Not necessarily a stable one, but a new one.
Related: 2014 AMSUS Meeting Wrap-Up
Plenaries, discussions, and presentations of original work will enable attendees to develop their own sight picture of the challenges and opportunities facing today’s health professionals in all federal spaces. Physicians, nurses, dentists, rehabilitation specialists, and health care administrators all have opportunities for professional growth and learning at the AMSUS 2015 conference—not to mention the unparalleled opportunities for interagency and international networking.
Visit http://amsusce.org/2015-call-for-abstracts for more information before the submission portal closes on May 4.
AMSUS, the Society of Federal Health Professionals, is holding its 2015 Annual Continuing Education Meeting from December 1-4 in San Antonio, Texas, and has issued a call for lecture and poster abstracts for consideration. The conference is the largest health care meeting focused on federal providers and is expected to draw more than 1,000 attendees from across the VA, DoD, PHS, NIH, CDC, and other federal agencies.
The theme for this year’s meeting is “the New Normal,” reflecting the reality that federal medicine has just come through an extraordinary time of change and is settling into its future. Concurrent and dramatic events in global politics, the evolution of new technologies, and advances in the organization of federal medicine have drawn the starting line of a new era. Not necessarily a stable one, but a new one.
Related: 2014 AMSUS Meeting Wrap-Up
Plenaries, discussions, and presentations of original work will enable attendees to develop their own sight picture of the challenges and opportunities facing today’s health professionals in all federal spaces. Physicians, nurses, dentists, rehabilitation specialists, and health care administrators all have opportunities for professional growth and learning at the AMSUS 2015 conference—not to mention the unparalleled opportunities for interagency and international networking.
Visit http://amsusce.org/2015-call-for-abstracts for more information before the submission portal closes on May 4.
AMSUS, the Society of Federal Health Professionals, is holding its 2015 Annual Continuing Education Meeting from December 1-4 in San Antonio, Texas, and has issued a call for lecture and poster abstracts for consideration. The conference is the largest health care meeting focused on federal providers and is expected to draw more than 1,000 attendees from across the VA, DoD, PHS, NIH, CDC, and other federal agencies.
The theme for this year’s meeting is “the New Normal,” reflecting the reality that federal medicine has just come through an extraordinary time of change and is settling into its future. Concurrent and dramatic events in global politics, the evolution of new technologies, and advances in the organization of federal medicine have drawn the starting line of a new era. Not necessarily a stable one, but a new one.
Related: 2014 AMSUS Meeting Wrap-Up
Plenaries, discussions, and presentations of original work will enable attendees to develop their own sight picture of the challenges and opportunities facing today’s health professionals in all federal spaces. Physicians, nurses, dentists, rehabilitation specialists, and health care administrators all have opportunities for professional growth and learning at the AMSUS 2015 conference—not to mention the unparalleled opportunities for interagency and international networking.
Visit http://amsusce.org/2015-call-for-abstracts for more information before the submission portal closes on May 4.
New Guidance on Compounded Drugs
As of May 1, 2015, pharmacy contractor for TRICARE, Express Scripts, will screen all ingredients in compound drug claims to make sure they are safe and effective.
The screening process, already in effect for other prescription drugs, will apply to drugs for which a pharmacist mixes ≥ 2 ingredients, even when altering the drug by adding a flavor to make it more palatable.
Related: Preparing the Military Health System for the 21st Century
“The changes really are not so much a policy change as really just us enforcing the regulatory guidance that we already have on our books,” said U.S. Army Maj. Gen. Richard W. Thomas, chief medical officer and director of health care operations for the Defense Health Agency (DHA), in an interview with DoD News.
Maj. Gen. Thomas said that in 2012, the pharmacy contractor completed a system upgrade, allowing the DoD to see specific ingredients in compounded drugs. They found that not all the ingredients were approved by the FDA and that safety and effectiveness were not always supported by scientific evidence. In 2014, moreover, a Government Accountability Office report indicated DHA was not in compliance with its own regulation to provide only FDA-approved medicine to beneficiaries, Maj. Gen. Thomas noted.
Related: TRICARE Walk-in Service Eliminated
The changes may also save money or at least help ensure that money is being spent wisely. According to Maj. Gen. Thomas, compounded drugs make up only 0.5% of the total number of prescriptions provided through TRICARE but account for > 20% of total pharmacy expenditures. Screening, he added, is part of fulfilling the DoD’s obligation to be responsible stewards of taxpayer dollars.
Related: VA and DoD Set to Share More Patients
TRICARE will continue to cover most compound drugs, and most beneficiaries will experience no delay in getting their prescriptions. If a compound doesn’t pass the initial screen, the pharmacist can switch a nonapproved ingredient with an approved one or request that the doctor write a new prescription.
As of May 1, 2015, pharmacy contractor for TRICARE, Express Scripts, will screen all ingredients in compound drug claims to make sure they are safe and effective.
The screening process, already in effect for other prescription drugs, will apply to drugs for which a pharmacist mixes ≥ 2 ingredients, even when altering the drug by adding a flavor to make it more palatable.
Related: Preparing the Military Health System for the 21st Century
“The changes really are not so much a policy change as really just us enforcing the regulatory guidance that we already have on our books,” said U.S. Army Maj. Gen. Richard W. Thomas, chief medical officer and director of health care operations for the Defense Health Agency (DHA), in an interview with DoD News.
Maj. Gen. Thomas said that in 2012, the pharmacy contractor completed a system upgrade, allowing the DoD to see specific ingredients in compounded drugs. They found that not all the ingredients were approved by the FDA and that safety and effectiveness were not always supported by scientific evidence. In 2014, moreover, a Government Accountability Office report indicated DHA was not in compliance with its own regulation to provide only FDA-approved medicine to beneficiaries, Maj. Gen. Thomas noted.
Related: TRICARE Walk-in Service Eliminated
The changes may also save money or at least help ensure that money is being spent wisely. According to Maj. Gen. Thomas, compounded drugs make up only 0.5% of the total number of prescriptions provided through TRICARE but account for > 20% of total pharmacy expenditures. Screening, he added, is part of fulfilling the DoD’s obligation to be responsible stewards of taxpayer dollars.
Related: VA and DoD Set to Share More Patients
TRICARE will continue to cover most compound drugs, and most beneficiaries will experience no delay in getting their prescriptions. If a compound doesn’t pass the initial screen, the pharmacist can switch a nonapproved ingredient with an approved one or request that the doctor write a new prescription.
As of May 1, 2015, pharmacy contractor for TRICARE, Express Scripts, will screen all ingredients in compound drug claims to make sure they are safe and effective.
The screening process, already in effect for other prescription drugs, will apply to drugs for which a pharmacist mixes ≥ 2 ingredients, even when altering the drug by adding a flavor to make it more palatable.
Related: Preparing the Military Health System for the 21st Century
“The changes really are not so much a policy change as really just us enforcing the regulatory guidance that we already have on our books,” said U.S. Army Maj. Gen. Richard W. Thomas, chief medical officer and director of health care operations for the Defense Health Agency (DHA), in an interview with DoD News.
Maj. Gen. Thomas said that in 2012, the pharmacy contractor completed a system upgrade, allowing the DoD to see specific ingredients in compounded drugs. They found that not all the ingredients were approved by the FDA and that safety and effectiveness were not always supported by scientific evidence. In 2014, moreover, a Government Accountability Office report indicated DHA was not in compliance with its own regulation to provide only FDA-approved medicine to beneficiaries, Maj. Gen. Thomas noted.
Related: TRICARE Walk-in Service Eliminated
The changes may also save money or at least help ensure that money is being spent wisely. According to Maj. Gen. Thomas, compounded drugs make up only 0.5% of the total number of prescriptions provided through TRICARE but account for > 20% of total pharmacy expenditures. Screening, he added, is part of fulfilling the DoD’s obligation to be responsible stewards of taxpayer dollars.
Related: VA and DoD Set to Share More Patients
TRICARE will continue to cover most compound drugs, and most beneficiaries will experience no delay in getting their prescriptions. If a compound doesn’t pass the initial screen, the pharmacist can switch a nonapproved ingredient with an approved one or request that the doctor write a new prescription.
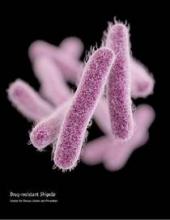
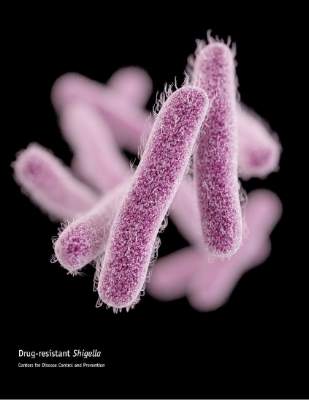
United States experiencing drug-resistant shigellosis outbreak
The United States is currently experiencing an outbreak of shigellosis caused by a strain of the Shigella sonnei bacteria that is resistant to ciprofloxacin, the most commonly prescribed antimicrobial treatment for shigellosis.
In its Morbidity and Mortality Weekly Report, the CDC revealed that 243 individuals in 32 states and the territory of Puerto Rico have come down with shigellosis between May 2014 and February 2015. Of those 243 cases, 126 isolates were tested and 109 (87%) of those were found to be nonsusceptible to ciprofloxacin. The largest clusters of the disease were found in Massachusetts (45 cases), California (25 cases), and Pennsylvania (18 cases).
Ninety-five of the cases associated with the current outbreak were traced back to the homeless population of San Francisco; about half of the remaining cases were attributed to international travelers – specifically, those visiting the Dominican Republic and India – who contracted the bacteria while abroad and unknowingly brought it to the United States. The disease is known to spread quickly in populations of children who attend child care facilities, homeless individuals, and men who have sex with men.
“These outbreaks show a troubling trend in Shigella infections in the United States,” Dr. Thomas Frieden, CDC director, said in a statement. “Drug-resistant infections are harder to treat and because Shigella spreads so easily between people, the potential for more – and larger – outbreaks is a real concern. We’re moving quickly to implement a national strategy to curb antibiotic resistance because we can’t take for granted that we’ll always have the drugs we need to fight common infections.”
Shigellosis causes an estimated 500,000 cases of diarrhea in the United States each year. To help curb the growing number of shigellosis cases, the CDC recommends that international travelers wash their hands meticulously while abroad, and follow strict dietary precautions, such as eating hot foods and drinking beverages only from sealed containers, especially when consuming water.
The United States is currently experiencing an outbreak of shigellosis caused by a strain of the Shigella sonnei bacteria that is resistant to ciprofloxacin, the most commonly prescribed antimicrobial treatment for shigellosis.
In its Morbidity and Mortality Weekly Report, the CDC revealed that 243 individuals in 32 states and the territory of Puerto Rico have come down with shigellosis between May 2014 and February 2015. Of those 243 cases, 126 isolates were tested and 109 (87%) of those were found to be nonsusceptible to ciprofloxacin. The largest clusters of the disease were found in Massachusetts (45 cases), California (25 cases), and Pennsylvania (18 cases).
Ninety-five of the cases associated with the current outbreak were traced back to the homeless population of San Francisco; about half of the remaining cases were attributed to international travelers – specifically, those visiting the Dominican Republic and India – who contracted the bacteria while abroad and unknowingly brought it to the United States. The disease is known to spread quickly in populations of children who attend child care facilities, homeless individuals, and men who have sex with men.
“These outbreaks show a troubling trend in Shigella infections in the United States,” Dr. Thomas Frieden, CDC director, said in a statement. “Drug-resistant infections are harder to treat and because Shigella spreads so easily between people, the potential for more – and larger – outbreaks is a real concern. We’re moving quickly to implement a national strategy to curb antibiotic resistance because we can’t take for granted that we’ll always have the drugs we need to fight common infections.”
Shigellosis causes an estimated 500,000 cases of diarrhea in the United States each year. To help curb the growing number of shigellosis cases, the CDC recommends that international travelers wash their hands meticulously while abroad, and follow strict dietary precautions, such as eating hot foods and drinking beverages only from sealed containers, especially when consuming water.
The United States is currently experiencing an outbreak of shigellosis caused by a strain of the Shigella sonnei bacteria that is resistant to ciprofloxacin, the most commonly prescribed antimicrobial treatment for shigellosis.
In its Morbidity and Mortality Weekly Report, the CDC revealed that 243 individuals in 32 states and the territory of Puerto Rico have come down with shigellosis between May 2014 and February 2015. Of those 243 cases, 126 isolates were tested and 109 (87%) of those were found to be nonsusceptible to ciprofloxacin. The largest clusters of the disease were found in Massachusetts (45 cases), California (25 cases), and Pennsylvania (18 cases).
Ninety-five of the cases associated with the current outbreak were traced back to the homeless population of San Francisco; about half of the remaining cases were attributed to international travelers – specifically, those visiting the Dominican Republic and India – who contracted the bacteria while abroad and unknowingly brought it to the United States. The disease is known to spread quickly in populations of children who attend child care facilities, homeless individuals, and men who have sex with men.
“These outbreaks show a troubling trend in Shigella infections in the United States,” Dr. Thomas Frieden, CDC director, said in a statement. “Drug-resistant infections are harder to treat and because Shigella spreads so easily between people, the potential for more – and larger – outbreaks is a real concern. We’re moving quickly to implement a national strategy to curb antibiotic resistance because we can’t take for granted that we’ll always have the drugs we need to fight common infections.”
Shigellosis causes an estimated 500,000 cases of diarrhea in the United States each year. To help curb the growing number of shigellosis cases, the CDC recommends that international travelers wash their hands meticulously while abroad, and follow strict dietary precautions, such as eating hot foods and drinking beverages only from sealed containers, especially when consuming water.
FROM MMWR
SGR cuts pay by 21%; CMS to hold checks for 2 weeks
Medicare physician pay was cut by approximately 21% effective April 1, due to the expiration of the last temporary fix to the Sustainable Growth Rate formula.
Because legislation to repeal the SGR has passed the House and awaits action in the Senate, the Centers for Medicare & Medicaid Services announced it will hold Medicare payments for 2 weeks, allowing Congress to complete action on the issue.
Without a legislative fix, beginning April 15, “CMS will begin processing claims received on or after April 1 with a 21% reduction in the physician’s rate to limit the impact on Medicare providers and beneficiaries,” the agency said in a statement.
The House on March 26 overwhelming passed H.R.2, the Medicare Access and CHIP Reauthorization Act, which would repeal the SGR, reauthorize the Children’s Health Insurance Program for 2 years, and reform Medicare. The Senate did not address the bill before taking a recess until April 13.
Should SGR repeal legislation pass Congress and be signed by the President – something he has indicated he will do – CMS will reprocess any claims processed at the lower rate.
Medicare physician pay was cut by approximately 21% effective April 1, due to the expiration of the last temporary fix to the Sustainable Growth Rate formula.
Because legislation to repeal the SGR has passed the House and awaits action in the Senate, the Centers for Medicare & Medicaid Services announced it will hold Medicare payments for 2 weeks, allowing Congress to complete action on the issue.
Without a legislative fix, beginning April 15, “CMS will begin processing claims received on or after April 1 with a 21% reduction in the physician’s rate to limit the impact on Medicare providers and beneficiaries,” the agency said in a statement.
The House on March 26 overwhelming passed H.R.2, the Medicare Access and CHIP Reauthorization Act, which would repeal the SGR, reauthorize the Children’s Health Insurance Program for 2 years, and reform Medicare. The Senate did not address the bill before taking a recess until April 13.
Should SGR repeal legislation pass Congress and be signed by the President – something he has indicated he will do – CMS will reprocess any claims processed at the lower rate.
Medicare physician pay was cut by approximately 21% effective April 1, due to the expiration of the last temporary fix to the Sustainable Growth Rate formula.
Because legislation to repeal the SGR has passed the House and awaits action in the Senate, the Centers for Medicare & Medicaid Services announced it will hold Medicare payments for 2 weeks, allowing Congress to complete action on the issue.
Without a legislative fix, beginning April 15, “CMS will begin processing claims received on or after April 1 with a 21% reduction in the physician’s rate to limit the impact on Medicare providers and beneficiaries,” the agency said in a statement.
The House on March 26 overwhelming passed H.R.2, the Medicare Access and CHIP Reauthorization Act, which would repeal the SGR, reauthorize the Children’s Health Insurance Program for 2 years, and reform Medicare. The Senate did not address the bill before taking a recess until April 13.
Should SGR repeal legislation pass Congress and be signed by the President – something he has indicated he will do – CMS will reprocess any claims processed at the lower rate.
Alcohol Awareness Month Is Here
Alcohol is the number one drug of choice for young Americans and is more likely to kill them than are all illegal drugs combined, according to the National Council on Alcoholism and Drug Dependence, Inc. (NCADD). This year’s awareness theme—Help for Today. Hope for Tomorrow.—is designed to draw attention to the “pervasive impact” alcohol has on young people, their friends and families, and the community.
Related: Civilian Stress Compounds Service-Related Stress
The April 2015 Toolkit from the HHS Office of Disease Prevention and Health Promotion and the NCADD offers strategies for spreading the word about alcohol abuse prevention (http://www.healthfinder.gov/NHO/APRILtoolkit.aspx). In addition to handouts, suggested tweets, e-cards, and other ways to educate patients, personal health tools, such as the Cocktail Content Calculator and Drink Size Calculator, may prove to be helpful.
Alcohol is the number one drug of choice for young Americans and is more likely to kill them than are all illegal drugs combined, according to the National Council on Alcoholism and Drug Dependence, Inc. (NCADD). This year’s awareness theme—Help for Today. Hope for Tomorrow.—is designed to draw attention to the “pervasive impact” alcohol has on young people, their friends and families, and the community.
Related: Civilian Stress Compounds Service-Related Stress
The April 2015 Toolkit from the HHS Office of Disease Prevention and Health Promotion and the NCADD offers strategies for spreading the word about alcohol abuse prevention (http://www.healthfinder.gov/NHO/APRILtoolkit.aspx). In addition to handouts, suggested tweets, e-cards, and other ways to educate patients, personal health tools, such as the Cocktail Content Calculator and Drink Size Calculator, may prove to be helpful.
Alcohol is the number one drug of choice for young Americans and is more likely to kill them than are all illegal drugs combined, according to the National Council on Alcoholism and Drug Dependence, Inc. (NCADD). This year’s awareness theme—Help for Today. Hope for Tomorrow.—is designed to draw attention to the “pervasive impact” alcohol has on young people, their friends and families, and the community.
Related: Civilian Stress Compounds Service-Related Stress
The April 2015 Toolkit from the HHS Office of Disease Prevention and Health Promotion and the NCADD offers strategies for spreading the word about alcohol abuse prevention (http://www.healthfinder.gov/NHO/APRILtoolkit.aspx). In addition to handouts, suggested tweets, e-cards, and other ways to educate patients, personal health tools, such as the Cocktail Content Calculator and Drink Size Calculator, may prove to be helpful.
National Priority: Combating Antibiotic Resistance
Antibiotic resistance causes 2 million illnesses and about 23,000 deaths each year in the U.S. and limits the ability to perform a range of medical procedures, including chemotherapy, surgery, and organ transplants, according to the HHS.
In response to this very real threat, on March 27, 2015, HHS released the National Action Plan for Combating Antibiotic Resistant Bacteria (NAP). HHS Secretary Sylvia M. Burwell, U.S. Department of Agriculture Secretary Tom Vilsack, and U.S. Defense Secretary Ash Carter describe NAP as a “whole-of-government approach,” requiring a long-term commitment over the next 5 years to:
- Slow the emergence of resistant bacteria and prevent the spread of resistant infections
- Strengthen national “One-Health” surveillance efforts
- Advance development and use of rapid and innovative diagnostic tests
- Accelerate basic and applied research and development
- Improve international collaboration and capacities
Related: Distinguishing Influenza From Bacterial Infection
The NAP outlines each of these 5 goals in detail, and 1-, 3-, and 5-year milestones benchmark many of the goals’ multiple objectives. Much of the 1-year milestones describe research, data collection, and the enhancement or establishment of interdepartmental and international cooperative data sharing; whereas much of the 3- and 5-year milestones describe ongoing research, data reporting, revised regulations, and measurement of the impact that any and all revised strategies have had or will have on the ultimate goal of combating antibiotic resistant bacteria worldwide.
Related: Antibiotic Failure: Not Only a Hospital Phenomenon
President Obama first outlined NAP in his September 2014 Executive Order, Combating Antibiotic-Resistant Bacteria. The NAP is part of the more than $1.2 billion proposed in the President’s fiscal year 2016 budget for combating and preventing antibiotic resistance.
Related: Global Health Cooperation
The coordinated approach to accomplishing NAP’s goals will require efforts of individuals and groups around the world, including public and private sector partners, health care providers, health care leaders, veterinarians, agriculture industry leaders, manufacturers, policymakers, and patients.
Antibiotic resistance causes 2 million illnesses and about 23,000 deaths each year in the U.S. and limits the ability to perform a range of medical procedures, including chemotherapy, surgery, and organ transplants, according to the HHS.
In response to this very real threat, on March 27, 2015, HHS released the National Action Plan for Combating Antibiotic Resistant Bacteria (NAP). HHS Secretary Sylvia M. Burwell, U.S. Department of Agriculture Secretary Tom Vilsack, and U.S. Defense Secretary Ash Carter describe NAP as a “whole-of-government approach,” requiring a long-term commitment over the next 5 years to:
- Slow the emergence of resistant bacteria and prevent the spread of resistant infections
- Strengthen national “One-Health” surveillance efforts
- Advance development and use of rapid and innovative diagnostic tests
- Accelerate basic and applied research and development
- Improve international collaboration and capacities
Related: Distinguishing Influenza From Bacterial Infection
The NAP outlines each of these 5 goals in detail, and 1-, 3-, and 5-year milestones benchmark many of the goals’ multiple objectives. Much of the 1-year milestones describe research, data collection, and the enhancement or establishment of interdepartmental and international cooperative data sharing; whereas much of the 3- and 5-year milestones describe ongoing research, data reporting, revised regulations, and measurement of the impact that any and all revised strategies have had or will have on the ultimate goal of combating antibiotic resistant bacteria worldwide.
Related: Antibiotic Failure: Not Only a Hospital Phenomenon
President Obama first outlined NAP in his September 2014 Executive Order, Combating Antibiotic-Resistant Bacteria. The NAP is part of the more than $1.2 billion proposed in the President’s fiscal year 2016 budget for combating and preventing antibiotic resistance.
Related: Global Health Cooperation
The coordinated approach to accomplishing NAP’s goals will require efforts of individuals and groups around the world, including public and private sector partners, health care providers, health care leaders, veterinarians, agriculture industry leaders, manufacturers, policymakers, and patients.
Antibiotic resistance causes 2 million illnesses and about 23,000 deaths each year in the U.S. and limits the ability to perform a range of medical procedures, including chemotherapy, surgery, and organ transplants, according to the HHS.
In response to this very real threat, on March 27, 2015, HHS released the National Action Plan for Combating Antibiotic Resistant Bacteria (NAP). HHS Secretary Sylvia M. Burwell, U.S. Department of Agriculture Secretary Tom Vilsack, and U.S. Defense Secretary Ash Carter describe NAP as a “whole-of-government approach,” requiring a long-term commitment over the next 5 years to:
- Slow the emergence of resistant bacteria and prevent the spread of resistant infections
- Strengthen national “One-Health” surveillance efforts
- Advance development and use of rapid and innovative diagnostic tests
- Accelerate basic and applied research and development
- Improve international collaboration and capacities
Related: Distinguishing Influenza From Bacterial Infection
The NAP outlines each of these 5 goals in detail, and 1-, 3-, and 5-year milestones benchmark many of the goals’ multiple objectives. Much of the 1-year milestones describe research, data collection, and the enhancement or establishment of interdepartmental and international cooperative data sharing; whereas much of the 3- and 5-year milestones describe ongoing research, data reporting, revised regulations, and measurement of the impact that any and all revised strategies have had or will have on the ultimate goal of combating antibiotic resistant bacteria worldwide.
Related: Antibiotic Failure: Not Only a Hospital Phenomenon
President Obama first outlined NAP in his September 2014 Executive Order, Combating Antibiotic-Resistant Bacteria. The NAP is part of the more than $1.2 billion proposed in the President’s fiscal year 2016 budget for combating and preventing antibiotic resistance.
Related: Global Health Cooperation
The coordinated approach to accomplishing NAP’s goals will require efforts of individuals and groups around the world, including public and private sector partners, health care providers, health care leaders, veterinarians, agriculture industry leaders, manufacturers, policymakers, and patients.
FDA strengthens allergy warning for IV anemia therapy ferumoxytol
The warning about potentially life-threatening allergic reactions with the intravenous anemia drug ferumoxytol is being strengthened to reflect cases of allergic reactions, including fatalities, associated with this drug since approval, the Food and Drug Administration announced on March 30.
Information about the risk of potentially life-threatening allergic reactions was included in the warnings and precautions section of the prescribing information for ferumoxytol (Feraheme) when it was approved in 2009, and this risk is associated with all IV iron therapies. Since approval of ferumoxytol, however, “serious reactions, including deaths, have occurred despite the proper use of therapies to treat these reactions and emergency resuscitation measures,” according to the FDA’s statement, a drug safety communication.
Ways to reduce this risk have been identified and “health care professionals should follow the new recommendations in the drug label,” which includes the new contraindication, a “strong recommendation against use of Feraheme in patients who have had an allergic reaction to any intravenous (IV) iron replacement product,” the statement said.
Other recommendations included in the revised prescribing information include instructions to administer diluted ferumoxytol in an IV infusion administered over at least 15 minutes and to never administer it as an undiluted solution; to closely monitor patients for signs and symptoms of serious allergic reactions, which includes monitoring pulse and blood pressure during administration and for at least 30 minutes after each infusion; and to “carefully consider the potential risks and benefits” of the drug in elderly patients and in patients who have multiple drug allergies, two groups of patients who may be at increased risk for serious reactions.
Between June 2009 and June 30, 2014, 79 reports of anaphylactic reactions associated with ferumoxytol were identified in people from aged 19-96 years in the FDA Adverse Event Reporting System database, of which 18 were fatal, “despite immediate medical intervention and emergency resuscitation attempts. Almost half were reported with the first dose and 60 cases – about 75% – were reported to have started during the infusion or within 5 minutes after it had been fully infused.
Of the 79 cases, 34 (43%) were reported in people with a history of drug allergy; and 24% had a history of multiple drug allergies. Cardiac arrest, hypotension, dyspnea, nausea, vomiting, and flushing were among the common symptoms described.
Serious adverse events associated with ferumoxytol or other IV iron therapies should be reported to the FDA’s MedWatch program at 800-332-1099 or on-line at https://www.accessdata.fda.gov/scripts/medwatch/.
The warning about potentially life-threatening allergic reactions with the intravenous anemia drug ferumoxytol is being strengthened to reflect cases of allergic reactions, including fatalities, associated with this drug since approval, the Food and Drug Administration announced on March 30.
Information about the risk of potentially life-threatening allergic reactions was included in the warnings and precautions section of the prescribing information for ferumoxytol (Feraheme) when it was approved in 2009, and this risk is associated with all IV iron therapies. Since approval of ferumoxytol, however, “serious reactions, including deaths, have occurred despite the proper use of therapies to treat these reactions and emergency resuscitation measures,” according to the FDA’s statement, a drug safety communication.
Ways to reduce this risk have been identified and “health care professionals should follow the new recommendations in the drug label,” which includes the new contraindication, a “strong recommendation against use of Feraheme in patients who have had an allergic reaction to any intravenous (IV) iron replacement product,” the statement said.
Other recommendations included in the revised prescribing information include instructions to administer diluted ferumoxytol in an IV infusion administered over at least 15 minutes and to never administer it as an undiluted solution; to closely monitor patients for signs and symptoms of serious allergic reactions, which includes monitoring pulse and blood pressure during administration and for at least 30 minutes after each infusion; and to “carefully consider the potential risks and benefits” of the drug in elderly patients and in patients who have multiple drug allergies, two groups of patients who may be at increased risk for serious reactions.
Between June 2009 and June 30, 2014, 79 reports of anaphylactic reactions associated with ferumoxytol were identified in people from aged 19-96 years in the FDA Adverse Event Reporting System database, of which 18 were fatal, “despite immediate medical intervention and emergency resuscitation attempts. Almost half were reported with the first dose and 60 cases – about 75% – were reported to have started during the infusion or within 5 minutes after it had been fully infused.
Of the 79 cases, 34 (43%) were reported in people with a history of drug allergy; and 24% had a history of multiple drug allergies. Cardiac arrest, hypotension, dyspnea, nausea, vomiting, and flushing were among the common symptoms described.
Serious adverse events associated with ferumoxytol or other IV iron therapies should be reported to the FDA’s MedWatch program at 800-332-1099 or on-line at https://www.accessdata.fda.gov/scripts/medwatch/.
The warning about potentially life-threatening allergic reactions with the intravenous anemia drug ferumoxytol is being strengthened to reflect cases of allergic reactions, including fatalities, associated with this drug since approval, the Food and Drug Administration announced on March 30.
Information about the risk of potentially life-threatening allergic reactions was included in the warnings and precautions section of the prescribing information for ferumoxytol (Feraheme) when it was approved in 2009, and this risk is associated with all IV iron therapies. Since approval of ferumoxytol, however, “serious reactions, including deaths, have occurred despite the proper use of therapies to treat these reactions and emergency resuscitation measures,” according to the FDA’s statement, a drug safety communication.
Ways to reduce this risk have been identified and “health care professionals should follow the new recommendations in the drug label,” which includes the new contraindication, a “strong recommendation against use of Feraheme in patients who have had an allergic reaction to any intravenous (IV) iron replacement product,” the statement said.
Other recommendations included in the revised prescribing information include instructions to administer diluted ferumoxytol in an IV infusion administered over at least 15 minutes and to never administer it as an undiluted solution; to closely monitor patients for signs and symptoms of serious allergic reactions, which includes monitoring pulse and blood pressure during administration and for at least 30 minutes after each infusion; and to “carefully consider the potential risks and benefits” of the drug in elderly patients and in patients who have multiple drug allergies, two groups of patients who may be at increased risk for serious reactions.
Between June 2009 and June 30, 2014, 79 reports of anaphylactic reactions associated with ferumoxytol were identified in people from aged 19-96 years in the FDA Adverse Event Reporting System database, of which 18 were fatal, “despite immediate medical intervention and emergency resuscitation attempts. Almost half were reported with the first dose and 60 cases – about 75% – were reported to have started during the infusion or within 5 minutes after it had been fully infused.
Of the 79 cases, 34 (43%) were reported in people with a history of drug allergy; and 24% had a history of multiple drug allergies. Cardiac arrest, hypotension, dyspnea, nausea, vomiting, and flushing were among the common symptoms described.
Serious adverse events associated with ferumoxytol or other IV iron therapies should be reported to the FDA’s MedWatch program at 800-332-1099 or on-line at https://www.accessdata.fda.gov/scripts/medwatch/.
VA and DoD Set to Share More Patients
Brooke Army Medical Center, the U.S. Air Force 59th Medical Wing, and the South Texas Veterans Health Care System now have a combined 15 sharing agreements, including equipment sterilization; medical services; and surgical services for general, gynecologic, orthopedic, and ear, nose, and throat surgery.
“Military medicine is focused on providing world-class, high-quality, safe health care to our number one customer—the patient,” said Maj. Gen. Bart Iddins, 59th Medical Wing commander, in a Brooke Army Medical Center press release. “We remain patient-centered in all we do, and work-share agreements bolster this commitment."
The Brooke Army Medical Center and South Texas Veterans Health Care System facilities join the existing work-share agreements between the facilities of VA Northern California Health Care System and the U.S. Air Force 60th Medical Group’s David Grant Medical Center. More details about VA and DoD work-share collaborations are available in “Mutual Alignment Trumps Merger for Joint VA/DoD Health Care Programs,” written by Scott A. Hundahl, MD.
“These programs deliver sustained, cost-effective care with improved access for veterans and military beneficiaries alike and continue to do so through planned, mutually aligned effort, not merger,” writes Dr. Hundahl.
Brooke Army Medical Center, the U.S. Air Force 59th Medical Wing, and the South Texas Veterans Health Care System now have a combined 15 sharing agreements, including equipment sterilization; medical services; and surgical services for general, gynecologic, orthopedic, and ear, nose, and throat surgery.
“Military medicine is focused on providing world-class, high-quality, safe health care to our number one customer—the patient,” said Maj. Gen. Bart Iddins, 59th Medical Wing commander, in a Brooke Army Medical Center press release. “We remain patient-centered in all we do, and work-share agreements bolster this commitment."
The Brooke Army Medical Center and South Texas Veterans Health Care System facilities join the existing work-share agreements between the facilities of VA Northern California Health Care System and the U.S. Air Force 60th Medical Group’s David Grant Medical Center. More details about VA and DoD work-share collaborations are available in “Mutual Alignment Trumps Merger for Joint VA/DoD Health Care Programs,” written by Scott A. Hundahl, MD.
“These programs deliver sustained, cost-effective care with improved access for veterans and military beneficiaries alike and continue to do so through planned, mutually aligned effort, not merger,” writes Dr. Hundahl.
Brooke Army Medical Center, the U.S. Air Force 59th Medical Wing, and the South Texas Veterans Health Care System now have a combined 15 sharing agreements, including equipment sterilization; medical services; and surgical services for general, gynecologic, orthopedic, and ear, nose, and throat surgery.
“Military medicine is focused on providing world-class, high-quality, safe health care to our number one customer—the patient,” said Maj. Gen. Bart Iddins, 59th Medical Wing commander, in a Brooke Army Medical Center press release. “We remain patient-centered in all we do, and work-share agreements bolster this commitment."
The Brooke Army Medical Center and South Texas Veterans Health Care System facilities join the existing work-share agreements between the facilities of VA Northern California Health Care System and the U.S. Air Force 60th Medical Group’s David Grant Medical Center. More details about VA and DoD work-share collaborations are available in “Mutual Alignment Trumps Merger for Joint VA/DoD Health Care Programs,” written by Scott A. Hundahl, MD.
“These programs deliver sustained, cost-effective care with improved access for veterans and military beneficiaries alike and continue to do so through planned, mutually aligned effort, not merger,” writes Dr. Hundahl.
Senate recesses without addressing SGR repeal bill
The Senate adjourned for a 2-week recess early on March 27, leaving without taking action on House-passed legislation to repeal the Medicare Sustainable Growth Rate formula and reauthorize the Children’s Health Insurance Program.
The Senate will not convene again until April 13, nearly 2 weeks after a temporary SGR patch expires. Doctors will see their Medicare pay cut by 21% on April 1.
As has happened in the SGR’s troubled past and for other reasons, doctors and legislators expect the Centers for Medicare & Medicaid Services to hold Medicare payments for a while until the Senate can act.
“We’ve seen CMS hold checks for 2 weeks. We know that can happen,” Rep. Michael Burgess (R-Texas), lead sponsor of H.R. 2, the Medicare Access and CHIP Reauthorization Act, told reporters. “If it doesn’t pass this week, the world wouldn’t come to an end.”
A 2-week hold generally does not impact physicians’ practices too harshly, Dr. Robert Wergin, president of the American Academy of Family Physicians, said regarding an administrative hold CMS announced in January 2015. “It does affect your cash flow, but it has happened in the past, particularly around the time with SGR patches.”
The American Medical Association called for the Senate to address H.R. 2 as soon as it returns.
“Physicians are always working to provide the highest quality of care for their patients and the bipartisan bill passed by the House provides a clear pathway for them to do that,” Dr. Robert Wah, president of the AMA, said in a statement. “We urge the Senate to immediately address this issue upon their return and once-and-for-all lay this destructive issue to rest by building the stable and sustainable Medicare program that our nation’s patients and physicians need and deserve.”
On Twitter @denisefulton
The Senate adjourned for a 2-week recess early on March 27, leaving without taking action on House-passed legislation to repeal the Medicare Sustainable Growth Rate formula and reauthorize the Children’s Health Insurance Program.
The Senate will not convene again until April 13, nearly 2 weeks after a temporary SGR patch expires. Doctors will see their Medicare pay cut by 21% on April 1.
As has happened in the SGR’s troubled past and for other reasons, doctors and legislators expect the Centers for Medicare & Medicaid Services to hold Medicare payments for a while until the Senate can act.
“We’ve seen CMS hold checks for 2 weeks. We know that can happen,” Rep. Michael Burgess (R-Texas), lead sponsor of H.R. 2, the Medicare Access and CHIP Reauthorization Act, told reporters. “If it doesn’t pass this week, the world wouldn’t come to an end.”
A 2-week hold generally does not impact physicians’ practices too harshly, Dr. Robert Wergin, president of the American Academy of Family Physicians, said regarding an administrative hold CMS announced in January 2015. “It does affect your cash flow, but it has happened in the past, particularly around the time with SGR patches.”
The American Medical Association called for the Senate to address H.R. 2 as soon as it returns.
“Physicians are always working to provide the highest quality of care for their patients and the bipartisan bill passed by the House provides a clear pathway for them to do that,” Dr. Robert Wah, president of the AMA, said in a statement. “We urge the Senate to immediately address this issue upon their return and once-and-for-all lay this destructive issue to rest by building the stable and sustainable Medicare program that our nation’s patients and physicians need and deserve.”
On Twitter @denisefulton
The Senate adjourned for a 2-week recess early on March 27, leaving without taking action on House-passed legislation to repeal the Medicare Sustainable Growth Rate formula and reauthorize the Children’s Health Insurance Program.
The Senate will not convene again until April 13, nearly 2 weeks after a temporary SGR patch expires. Doctors will see their Medicare pay cut by 21% on April 1.
As has happened in the SGR’s troubled past and for other reasons, doctors and legislators expect the Centers for Medicare & Medicaid Services to hold Medicare payments for a while until the Senate can act.
“We’ve seen CMS hold checks for 2 weeks. We know that can happen,” Rep. Michael Burgess (R-Texas), lead sponsor of H.R. 2, the Medicare Access and CHIP Reauthorization Act, told reporters. “If it doesn’t pass this week, the world wouldn’t come to an end.”
A 2-week hold generally does not impact physicians’ practices too harshly, Dr. Robert Wergin, president of the American Academy of Family Physicians, said regarding an administrative hold CMS announced in January 2015. “It does affect your cash flow, but it has happened in the past, particularly around the time with SGR patches.”
The American Medical Association called for the Senate to address H.R. 2 as soon as it returns.
“Physicians are always working to provide the highest quality of care for their patients and the bipartisan bill passed by the House provides a clear pathway for them to do that,” Dr. Robert Wah, president of the AMA, said in a statement. “We urge the Senate to immediately address this issue upon their return and once-and-for-all lay this destructive issue to rest by building the stable and sustainable Medicare program that our nation’s patients and physicians need and deserve.”
On Twitter @denisefulton
Doctors hail House vote to repeal, replace SGR
Physicians are lauding passage of legislation by the House of Representatives to repeal the Medicare Sustainable Growth Rate formula (SGR) and replace it with an alternative system that would raise physician payments and focus on value-based performance. The bill also reauthorizes the Children’s Health Insurance Program (CHIP) for 2 years.
By a vote of 329-37, the House on March 26 passed H.R. 2, the Medicare Access and CHIP Reauthorization Act. The legislation builds on H.R. 1470, the SGR Repeal and Medicare Provider Payment Modernization Act – reintroduced March 19 – which includes a 0.5% pay increase per year for providers over the next 5 years.
Physician leaders praised the vote and thanked lawmakers for finally passing what they called a critical SGR fix.
“Never before has there been such broad and bipartisan support within and outside of Congress for policies to repeal the Medicare SGR formula and to create a better payment system for physician services provided to patients enrolled in Medicare,” ACP President David A. Fleming said in a statement. “It is imperative that the Senate pass H.R. 2 before it recesses on Friday, March 27, and before a devastating 21% SGR cut is applied cut is applied to all physician services provided to Medicare enrollees.”
If enacted, H.R. 2 also would consolidate existing quality programs – including those regarding the meaningful use of electronic health records – into a single value-based performance program; incentivize physicians to use alternate payment models that focus on care coordination and preventive care; and push for more transparency of Medicare data for physicians, providers, and patients.
The bill also would reauthorize for 2 years CHIP, the Community Health Centers program, the National Health Service Corps, and the Teaching Health Centers program, all are scheduled to expire later this year. Additionally, the legislation continues a partial delay of the Medicare two-midnights ruleuntil Sept. 30.
The Congressional Budget Office estimates that enactment of H.R. 2 will increase the deficit by $141 billion over 10 years. The CBO’s score also found that the legislation would save money compared to the price of continued patches.
A total of $73 billion of the $214 billion cost of package is offset through spending reductions and revenue increases included in the bill, the CBO found. These include income-related premium adjustments for Medicare Parts B and D, Medigap reforms, an increase of levy authority on payments to Medicare providers with delinquent tax debt, adjustments to inpatient hospital payment rates, a delay of Medicaid Disproportionate Share Hospital (DSH) changes until 2018, and a 1% market basket update for postacute care providers
During House debate, Rep. Michael C. Burgess (R-Texas), the bill’s sponsor and a member of the Energy and Commerce Committee’s Health Subcommittee, said the legislation resolves an issue that many lawmakers have worked to repair their entire congressional careers.
“Everyone agrees, the Medicare Sustainable Growth Rate formula has got to go,” said Rep. Burgess, an ob.gyn. “The bill before us provides 5 years of payment transition and allows medicine to concentrate on moving to broad adoption of quality reporting and most importantly, allows congress to move past the distraction of the SGR formula and to begin to identify Medicare reforms that can further benefit our citizens.”
Physician associations commended passage of the bill.
“The American Medical Association applauds the U.S. House of Representatives for overwhelmingly passing a long overdue bill to permanently eliminate the flawed SGR formula and put in place important Medicare payment and delivery reforms that will improve the health of the nation,” AMA President Robert M. Wah said in a statement.
Long-term health security for elderly and disabled patients is closer to reality thanks to the passage of H.R. 2, according to Dr. Robert L. Wergin, president of the American Academy of Family Physicians.
“For the first time in 12 years, true Medicare reform is feasible,” Dr. Wergin said in a statement. “H.R. 2 repeals the disruptive SGR and restructures physician payment to help expand access, encourage quality improvements and spur innovation. The SGR has plagued Medicare patients for far too long. It’s destabilized their access to care and threatened the financial viability of their physicians’ practices for more than a decade.”
By reauthorizing CHIP, the bill will also help ensure millions of children, adolescents, and pregnant women have access to the care that they need, according to a joint statement by the American Academy of Pediatrics, the AMA, and the American Congress of Obstetricians and Gynecologists.
“CHIP is a bipartisan program that works,” the organizations said. “Since its beginning 17 years ago, the program has worked together with Medicaid to cut the child uninsurance rate in half, offering timely access to high-quality, affordable health insurance.”
Enactment of H.R. 2 will allow physicians to focus more fully on patient care rather than face the threat of dramatic cuts that could make caring for Medicare patients unsustainable, according to Dr. Kim Allan Williams Sr., president of the American College of Cardiology.
“Instead of kicking the can down the road one more time, Speaker [John] Boehner and Leader [Nancy] Pelosi demonstrated strong bipartisan leadership to address this problem at last. We urge the Senate to follow suit and quickly take up and pass the provisions of the House measure,” Dr. Williams said in a statement.
President Obama has indicated that he will sign the bill, calling the replacement model a system that offers predictability and accelerates participation in alternative payment models that encourage quality and efficiency.
“The proposal would advance the administration’s goal of moving the nation’s health care delivery system toward one that achieves better care, smarter spending, and healthier people through the expansion of new health care payment models, which could contribute to slowing long-term health care cost growth,” Mr. Obama said in a March 25 statement.
The bill’s passage comes as the latest SGR patch is set to expire on March 31.
The Senate could take up the bill as early as March 27; however, it also could wait until mid-April after a 2-week recess. Without a permanent or temporary SGR fix, doctors can expect a 21% Medicare pay cut come April 1.
Democrats on the Senate Finance Committee have criticized H.R. 2, saying that it unfairly penalizes patients and does not reauthorize CHIP for long enough.
“Any legislation of this magnitude sent to the Senate must be balanced,” according to a joint statement signed by all 12 Democrats who serve on the Finance Committee. “Our current understanding of what the House is negotiating does not sufficiently pass that test....While our concerns vary, we are united by the necessity of extending CHIP funding for 4 years.”
But Finance Committee Chair Sen. Orrin Hatch (R-Utah) has called for swift passage of the bill.
“The time to act is now,” Sen. Hatch said in a March 25 speech on the Senate floor. “I can’t imagine another bipartisan opportunity like this coming around again anytime soon.”
On Twitter @legal_med
Physicians are lauding passage of legislation by the House of Representatives to repeal the Medicare Sustainable Growth Rate formula (SGR) and replace it with an alternative system that would raise physician payments and focus on value-based performance. The bill also reauthorizes the Children’s Health Insurance Program (CHIP) for 2 years.
By a vote of 329-37, the House on March 26 passed H.R. 2, the Medicare Access and CHIP Reauthorization Act. The legislation builds on H.R. 1470, the SGR Repeal and Medicare Provider Payment Modernization Act – reintroduced March 19 – which includes a 0.5% pay increase per year for providers over the next 5 years.
Physician leaders praised the vote and thanked lawmakers for finally passing what they called a critical SGR fix.
“Never before has there been such broad and bipartisan support within and outside of Congress for policies to repeal the Medicare SGR formula and to create a better payment system for physician services provided to patients enrolled in Medicare,” ACP President David A. Fleming said in a statement. “It is imperative that the Senate pass H.R. 2 before it recesses on Friday, March 27, and before a devastating 21% SGR cut is applied cut is applied to all physician services provided to Medicare enrollees.”
If enacted, H.R. 2 also would consolidate existing quality programs – including those regarding the meaningful use of electronic health records – into a single value-based performance program; incentivize physicians to use alternate payment models that focus on care coordination and preventive care; and push for more transparency of Medicare data for physicians, providers, and patients.
The bill also would reauthorize for 2 years CHIP, the Community Health Centers program, the National Health Service Corps, and the Teaching Health Centers program, all are scheduled to expire later this year. Additionally, the legislation continues a partial delay of the Medicare two-midnights ruleuntil Sept. 30.
The Congressional Budget Office estimates that enactment of H.R. 2 will increase the deficit by $141 billion over 10 years. The CBO’s score also found that the legislation would save money compared to the price of continued patches.
A total of $73 billion of the $214 billion cost of package is offset through spending reductions and revenue increases included in the bill, the CBO found. These include income-related premium adjustments for Medicare Parts B and D, Medigap reforms, an increase of levy authority on payments to Medicare providers with delinquent tax debt, adjustments to inpatient hospital payment rates, a delay of Medicaid Disproportionate Share Hospital (DSH) changes until 2018, and a 1% market basket update for postacute care providers
During House debate, Rep. Michael C. Burgess (R-Texas), the bill’s sponsor and a member of the Energy and Commerce Committee’s Health Subcommittee, said the legislation resolves an issue that many lawmakers have worked to repair their entire congressional careers.
“Everyone agrees, the Medicare Sustainable Growth Rate formula has got to go,” said Rep. Burgess, an ob.gyn. “The bill before us provides 5 years of payment transition and allows medicine to concentrate on moving to broad adoption of quality reporting and most importantly, allows congress to move past the distraction of the SGR formula and to begin to identify Medicare reforms that can further benefit our citizens.”
Physician associations commended passage of the bill.
“The American Medical Association applauds the U.S. House of Representatives for overwhelmingly passing a long overdue bill to permanently eliminate the flawed SGR formula and put in place important Medicare payment and delivery reforms that will improve the health of the nation,” AMA President Robert M. Wah said in a statement.
Long-term health security for elderly and disabled patients is closer to reality thanks to the passage of H.R. 2, according to Dr. Robert L. Wergin, president of the American Academy of Family Physicians.
“For the first time in 12 years, true Medicare reform is feasible,” Dr. Wergin said in a statement. “H.R. 2 repeals the disruptive SGR and restructures physician payment to help expand access, encourage quality improvements and spur innovation. The SGR has plagued Medicare patients for far too long. It’s destabilized their access to care and threatened the financial viability of their physicians’ practices for more than a decade.”
By reauthorizing CHIP, the bill will also help ensure millions of children, adolescents, and pregnant women have access to the care that they need, according to a joint statement by the American Academy of Pediatrics, the AMA, and the American Congress of Obstetricians and Gynecologists.
“CHIP is a bipartisan program that works,” the organizations said. “Since its beginning 17 years ago, the program has worked together with Medicaid to cut the child uninsurance rate in half, offering timely access to high-quality, affordable health insurance.”
Enactment of H.R. 2 will allow physicians to focus more fully on patient care rather than face the threat of dramatic cuts that could make caring for Medicare patients unsustainable, according to Dr. Kim Allan Williams Sr., president of the American College of Cardiology.
“Instead of kicking the can down the road one more time, Speaker [John] Boehner and Leader [Nancy] Pelosi demonstrated strong bipartisan leadership to address this problem at last. We urge the Senate to follow suit and quickly take up and pass the provisions of the House measure,” Dr. Williams said in a statement.
President Obama has indicated that he will sign the bill, calling the replacement model a system that offers predictability and accelerates participation in alternative payment models that encourage quality and efficiency.
“The proposal would advance the administration’s goal of moving the nation’s health care delivery system toward one that achieves better care, smarter spending, and healthier people through the expansion of new health care payment models, which could contribute to slowing long-term health care cost growth,” Mr. Obama said in a March 25 statement.
The bill’s passage comes as the latest SGR patch is set to expire on March 31.
The Senate could take up the bill as early as March 27; however, it also could wait until mid-April after a 2-week recess. Without a permanent or temporary SGR fix, doctors can expect a 21% Medicare pay cut come April 1.
Democrats on the Senate Finance Committee have criticized H.R. 2, saying that it unfairly penalizes patients and does not reauthorize CHIP for long enough.
“Any legislation of this magnitude sent to the Senate must be balanced,” according to a joint statement signed by all 12 Democrats who serve on the Finance Committee. “Our current understanding of what the House is negotiating does not sufficiently pass that test....While our concerns vary, we are united by the necessity of extending CHIP funding for 4 years.”
But Finance Committee Chair Sen. Orrin Hatch (R-Utah) has called for swift passage of the bill.
“The time to act is now,” Sen. Hatch said in a March 25 speech on the Senate floor. “I can’t imagine another bipartisan opportunity like this coming around again anytime soon.”
On Twitter @legal_med
Physicians are lauding passage of legislation by the House of Representatives to repeal the Medicare Sustainable Growth Rate formula (SGR) and replace it with an alternative system that would raise physician payments and focus on value-based performance. The bill also reauthorizes the Children’s Health Insurance Program (CHIP) for 2 years.
By a vote of 329-37, the House on March 26 passed H.R. 2, the Medicare Access and CHIP Reauthorization Act. The legislation builds on H.R. 1470, the SGR Repeal and Medicare Provider Payment Modernization Act – reintroduced March 19 – which includes a 0.5% pay increase per year for providers over the next 5 years.
Physician leaders praised the vote and thanked lawmakers for finally passing what they called a critical SGR fix.
“Never before has there been such broad and bipartisan support within and outside of Congress for policies to repeal the Medicare SGR formula and to create a better payment system for physician services provided to patients enrolled in Medicare,” ACP President David A. Fleming said in a statement. “It is imperative that the Senate pass H.R. 2 before it recesses on Friday, March 27, and before a devastating 21% SGR cut is applied cut is applied to all physician services provided to Medicare enrollees.”
If enacted, H.R. 2 also would consolidate existing quality programs – including those regarding the meaningful use of electronic health records – into a single value-based performance program; incentivize physicians to use alternate payment models that focus on care coordination and preventive care; and push for more transparency of Medicare data for physicians, providers, and patients.
The bill also would reauthorize for 2 years CHIP, the Community Health Centers program, the National Health Service Corps, and the Teaching Health Centers program, all are scheduled to expire later this year. Additionally, the legislation continues a partial delay of the Medicare two-midnights ruleuntil Sept. 30.
The Congressional Budget Office estimates that enactment of H.R. 2 will increase the deficit by $141 billion over 10 years. The CBO’s score also found that the legislation would save money compared to the price of continued patches.
A total of $73 billion of the $214 billion cost of package is offset through spending reductions and revenue increases included in the bill, the CBO found. These include income-related premium adjustments for Medicare Parts B and D, Medigap reforms, an increase of levy authority on payments to Medicare providers with delinquent tax debt, adjustments to inpatient hospital payment rates, a delay of Medicaid Disproportionate Share Hospital (DSH) changes until 2018, and a 1% market basket update for postacute care providers
During House debate, Rep. Michael C. Burgess (R-Texas), the bill’s sponsor and a member of the Energy and Commerce Committee’s Health Subcommittee, said the legislation resolves an issue that many lawmakers have worked to repair their entire congressional careers.
“Everyone agrees, the Medicare Sustainable Growth Rate formula has got to go,” said Rep. Burgess, an ob.gyn. “The bill before us provides 5 years of payment transition and allows medicine to concentrate on moving to broad adoption of quality reporting and most importantly, allows congress to move past the distraction of the SGR formula and to begin to identify Medicare reforms that can further benefit our citizens.”
Physician associations commended passage of the bill.
“The American Medical Association applauds the U.S. House of Representatives for overwhelmingly passing a long overdue bill to permanently eliminate the flawed SGR formula and put in place important Medicare payment and delivery reforms that will improve the health of the nation,” AMA President Robert M. Wah said in a statement.
Long-term health security for elderly and disabled patients is closer to reality thanks to the passage of H.R. 2, according to Dr. Robert L. Wergin, president of the American Academy of Family Physicians.
“For the first time in 12 years, true Medicare reform is feasible,” Dr. Wergin said in a statement. “H.R. 2 repeals the disruptive SGR and restructures physician payment to help expand access, encourage quality improvements and spur innovation. The SGR has plagued Medicare patients for far too long. It’s destabilized their access to care and threatened the financial viability of their physicians’ practices for more than a decade.”
By reauthorizing CHIP, the bill will also help ensure millions of children, adolescents, and pregnant women have access to the care that they need, according to a joint statement by the American Academy of Pediatrics, the AMA, and the American Congress of Obstetricians and Gynecologists.
“CHIP is a bipartisan program that works,” the organizations said. “Since its beginning 17 years ago, the program has worked together with Medicaid to cut the child uninsurance rate in half, offering timely access to high-quality, affordable health insurance.”
Enactment of H.R. 2 will allow physicians to focus more fully on patient care rather than face the threat of dramatic cuts that could make caring for Medicare patients unsustainable, according to Dr. Kim Allan Williams Sr., president of the American College of Cardiology.
“Instead of kicking the can down the road one more time, Speaker [John] Boehner and Leader [Nancy] Pelosi demonstrated strong bipartisan leadership to address this problem at last. We urge the Senate to follow suit and quickly take up and pass the provisions of the House measure,” Dr. Williams said in a statement.
President Obama has indicated that he will sign the bill, calling the replacement model a system that offers predictability and accelerates participation in alternative payment models that encourage quality and efficiency.
“The proposal would advance the administration’s goal of moving the nation’s health care delivery system toward one that achieves better care, smarter spending, and healthier people through the expansion of new health care payment models, which could contribute to slowing long-term health care cost growth,” Mr. Obama said in a March 25 statement.
The bill’s passage comes as the latest SGR patch is set to expire on March 31.
The Senate could take up the bill as early as March 27; however, it also could wait until mid-April after a 2-week recess. Without a permanent or temporary SGR fix, doctors can expect a 21% Medicare pay cut come April 1.
Democrats on the Senate Finance Committee have criticized H.R. 2, saying that it unfairly penalizes patients and does not reauthorize CHIP for long enough.
“Any legislation of this magnitude sent to the Senate must be balanced,” according to a joint statement signed by all 12 Democrats who serve on the Finance Committee. “Our current understanding of what the House is negotiating does not sufficiently pass that test....While our concerns vary, we are united by the necessity of extending CHIP funding for 4 years.”
But Finance Committee Chair Sen. Orrin Hatch (R-Utah) has called for swift passage of the bill.
“The time to act is now,” Sen. Hatch said in a March 25 speech on the Senate floor. “I can’t imagine another bipartisan opportunity like this coming around again anytime soon.”
On Twitter @legal_med