User login
Problematic Medications: Antibiotics in Renal Patients
Q) At a lecture I recently attended, the speaker said sulfamethoxazole/trimethoprim is a potentially dangerous medication. I use it all the time. Is there any data to support her comments? Where did she get her information?
Sulfamethoxazole/trimethoprim (SMX/TMP) is a combination of two antibiotics, each of which has the potential to interact with other substances.
It is well documented that sulfamethoxazole can inhibit the metabolism of cytochrome P450 2C9 substrates. Frequently prescribed medications that also use the cytochrome substrate include warfarin and oral antihypoglycemic agents.
Trimethoprim’s distinct properties also lead to drug interactions. Trimethoprim inhibits sodium uptake by the appropriate channels in the distal tubule of the kidney, preventing reabsorption and altering the electrical balance of the tubular cells. As a result, the amount of potassium excreted into the urine is reduced, yielding an accumulation of serum potassium.1
High serum potassium retention can manifest as hyperkalemia in patients with chronic kidney disease (CKD). Use of potassium-sparing drugs by patients with comorbidities, including CKD, can increase risk for hyperkalemia; concurrent use of these drugs with ACE inhibitors or angiotensin II receptor blockers (ARBs) compounds the risk.2 The first reports of hyperkalemia with trimethoprim use occurred in HIV patients treated with large doses for Pneumocystis carinii infection.3
In a population-based case-control study, the results of which were published in the British Medical Journal, Fralick and colleagues analyzed data on older patients (age 66 or older) who were taking either ACE inhibitors or ARBs in combination with an antibiotic.4 They found a significantly increased risk for sudden death within seven days of prescription of SMX/TMP, compared to amoxicillin; a secondary analysis also revealed an increased risk for sudden death within 14 days with SMX/TMP. The researchers speculated that this excess risk, which translated to 3 sudden deaths in 1,000 patients taking SMX/TMP versus 1 sudden death in 1,000 patients taking amoxicillin, “reflects unrecognized arrhythmic death due to hyperkalemia.”
Since more than 250 million prescriptions for ACE inhibitors/ARBs and 20 million prescriptions for SMX/TMP are written each year, there will be instances of overlap. The prudent clinician would prescribe a different antibiotic or, if avoidance is not possible, use the lowest effective dose and duration of SMX/TMP. Close monitoring of serum potassium levels is warranted in patients with comorbidities, especially CKD, who are taking ACE inhibitors or ARBs—and of course, in our geriatric population. —DLC
Debra L. Coplon, DNP, DCC
City of Memphis Wellness Clinic, Tennessee
REFERENCES
1. Velazquez H, Perazella MA, Wright FS, Ellison DH. Renal mechanism of trimethoprim-induced hyperkalemia. Ann Intern Med. 1993;119:296-301.
2. Horn JR, Hansten PD. Trimethoprim and potassium-sparing drugs: a risk for hyperkalemia. www.pharmacytimes.com/publications/issue/2011/February2011/DrugInteractions-0211. Accessed August 24, 2015.
3. Medina I, Mills J, Leoung G, et al. Oral therapy for Pneumocystis carinii pneumonia in the acquired immunodeficiency syndrome: a controlled trial of trimethoprim-sulfamethoxazole versus trimethoprim-dapsone. N Engl J Med. 1990;323:776-782.
4. Fralick M, Macdonald EM, Gomes T, et al. Co-trimoxazole and sudden death in patients receiving inhibitors of renin-angiotensin system: population based study. BMJ. 2014;349:g6196.
5. Gilbert SJ, Weiner DE, Gipson DS, et al. National Kidney Foundation’s Primer on Kidney Diseases. Philadelphia, PA: Elsevier; 2014.
6. Muriithi AK, Leung N, Valeri AM, et al. Biopsy-proven acute interstitial nephritis, 1993-2011: a case series. Am J Kidney Dis. 2014;64(4):558-566.
7. Blank ML, Parkin L, Paul C, Herbison P. A nationwide nested case-control study indicates an increased risk of acute interstitial nephritis with proton pump inhibitor use. Kidney Int. 2014;86(4):837-844.
8. Klepser DG, Collier DS, Cochran GL. Proton pump inhibitors and acute kidney injury: a nested case-control study. BMC Nephrology. 2013;14:150.
Q) At a lecture I recently attended, the speaker said sulfamethoxazole/trimethoprim is a potentially dangerous medication. I use it all the time. Is there any data to support her comments? Where did she get her information?
Sulfamethoxazole/trimethoprim (SMX/TMP) is a combination of two antibiotics, each of which has the potential to interact with other substances.
It is well documented that sulfamethoxazole can inhibit the metabolism of cytochrome P450 2C9 substrates. Frequently prescribed medications that also use the cytochrome substrate include warfarin and oral antihypoglycemic agents.
Trimethoprim’s distinct properties also lead to drug interactions. Trimethoprim inhibits sodium uptake by the appropriate channels in the distal tubule of the kidney, preventing reabsorption and altering the electrical balance of the tubular cells. As a result, the amount of potassium excreted into the urine is reduced, yielding an accumulation of serum potassium.1
High serum potassium retention can manifest as hyperkalemia in patients with chronic kidney disease (CKD). Use of potassium-sparing drugs by patients with comorbidities, including CKD, can increase risk for hyperkalemia; concurrent use of these drugs with ACE inhibitors or angiotensin II receptor blockers (ARBs) compounds the risk.2 The first reports of hyperkalemia with trimethoprim use occurred in HIV patients treated with large doses for Pneumocystis carinii infection.3
In a population-based case-control study, the results of which were published in the British Medical Journal, Fralick and colleagues analyzed data on older patients (age 66 or older) who were taking either ACE inhibitors or ARBs in combination with an antibiotic.4 They found a significantly increased risk for sudden death within seven days of prescription of SMX/TMP, compared to amoxicillin; a secondary analysis also revealed an increased risk for sudden death within 14 days with SMX/TMP. The researchers speculated that this excess risk, which translated to 3 sudden deaths in 1,000 patients taking SMX/TMP versus 1 sudden death in 1,000 patients taking amoxicillin, “reflects unrecognized arrhythmic death due to hyperkalemia.”
Since more than 250 million prescriptions for ACE inhibitors/ARBs and 20 million prescriptions for SMX/TMP are written each year, there will be instances of overlap. The prudent clinician would prescribe a different antibiotic or, if avoidance is not possible, use the lowest effective dose and duration of SMX/TMP. Close monitoring of serum potassium levels is warranted in patients with comorbidities, especially CKD, who are taking ACE inhibitors or ARBs—and of course, in our geriatric population. —DLC
Debra L. Coplon, DNP, DCC
City of Memphis Wellness Clinic, Tennessee
REFERENCES
1. Velazquez H, Perazella MA, Wright FS, Ellison DH. Renal mechanism of trimethoprim-induced hyperkalemia. Ann Intern Med. 1993;119:296-301.
2. Horn JR, Hansten PD. Trimethoprim and potassium-sparing drugs: a risk for hyperkalemia. www.pharmacytimes.com/publications/issue/2011/February2011/DrugInteractions-0211. Accessed August 24, 2015.
3. Medina I, Mills J, Leoung G, et al. Oral therapy for Pneumocystis carinii pneumonia in the acquired immunodeficiency syndrome: a controlled trial of trimethoprim-sulfamethoxazole versus trimethoprim-dapsone. N Engl J Med. 1990;323:776-782.
4. Fralick M, Macdonald EM, Gomes T, et al. Co-trimoxazole and sudden death in patients receiving inhibitors of renin-angiotensin system: population based study. BMJ. 2014;349:g6196.
5. Gilbert SJ, Weiner DE, Gipson DS, et al. National Kidney Foundation’s Primer on Kidney Diseases. Philadelphia, PA: Elsevier; 2014.
6. Muriithi AK, Leung N, Valeri AM, et al. Biopsy-proven acute interstitial nephritis, 1993-2011: a case series. Am J Kidney Dis. 2014;64(4):558-566.
7. Blank ML, Parkin L, Paul C, Herbison P. A nationwide nested case-control study indicates an increased risk of acute interstitial nephritis with proton pump inhibitor use. Kidney Int. 2014;86(4):837-844.
8. Klepser DG, Collier DS, Cochran GL. Proton pump inhibitors and acute kidney injury: a nested case-control study. BMC Nephrology. 2013;14:150.
Q) At a lecture I recently attended, the speaker said sulfamethoxazole/trimethoprim is a potentially dangerous medication. I use it all the time. Is there any data to support her comments? Where did she get her information?
Sulfamethoxazole/trimethoprim (SMX/TMP) is a combination of two antibiotics, each of which has the potential to interact with other substances.
It is well documented that sulfamethoxazole can inhibit the metabolism of cytochrome P450 2C9 substrates. Frequently prescribed medications that also use the cytochrome substrate include warfarin and oral antihypoglycemic agents.
Trimethoprim’s distinct properties also lead to drug interactions. Trimethoprim inhibits sodium uptake by the appropriate channels in the distal tubule of the kidney, preventing reabsorption and altering the electrical balance of the tubular cells. As a result, the amount of potassium excreted into the urine is reduced, yielding an accumulation of serum potassium.1
High serum potassium retention can manifest as hyperkalemia in patients with chronic kidney disease (CKD). Use of potassium-sparing drugs by patients with comorbidities, including CKD, can increase risk for hyperkalemia; concurrent use of these drugs with ACE inhibitors or angiotensin II receptor blockers (ARBs) compounds the risk.2 The first reports of hyperkalemia with trimethoprim use occurred in HIV patients treated with large doses for Pneumocystis carinii infection.3
In a population-based case-control study, the results of which were published in the British Medical Journal, Fralick and colleagues analyzed data on older patients (age 66 or older) who were taking either ACE inhibitors or ARBs in combination with an antibiotic.4 They found a significantly increased risk for sudden death within seven days of prescription of SMX/TMP, compared to amoxicillin; a secondary analysis also revealed an increased risk for sudden death within 14 days with SMX/TMP. The researchers speculated that this excess risk, which translated to 3 sudden deaths in 1,000 patients taking SMX/TMP versus 1 sudden death in 1,000 patients taking amoxicillin, “reflects unrecognized arrhythmic death due to hyperkalemia.”
Since more than 250 million prescriptions for ACE inhibitors/ARBs and 20 million prescriptions for SMX/TMP are written each year, there will be instances of overlap. The prudent clinician would prescribe a different antibiotic or, if avoidance is not possible, use the lowest effective dose and duration of SMX/TMP. Close monitoring of serum potassium levels is warranted in patients with comorbidities, especially CKD, who are taking ACE inhibitors or ARBs—and of course, in our geriatric population. —DLC
Debra L. Coplon, DNP, DCC
City of Memphis Wellness Clinic, Tennessee
REFERENCES
1. Velazquez H, Perazella MA, Wright FS, Ellison DH. Renal mechanism of trimethoprim-induced hyperkalemia. Ann Intern Med. 1993;119:296-301.
2. Horn JR, Hansten PD. Trimethoprim and potassium-sparing drugs: a risk for hyperkalemia. www.pharmacytimes.com/publications/issue/2011/February2011/DrugInteractions-0211. Accessed August 24, 2015.
3. Medina I, Mills J, Leoung G, et al. Oral therapy for Pneumocystis carinii pneumonia in the acquired immunodeficiency syndrome: a controlled trial of trimethoprim-sulfamethoxazole versus trimethoprim-dapsone. N Engl J Med. 1990;323:776-782.
4. Fralick M, Macdonald EM, Gomes T, et al. Co-trimoxazole and sudden death in patients receiving inhibitors of renin-angiotensin system: population based study. BMJ. 2014;349:g6196.
5. Gilbert SJ, Weiner DE, Gipson DS, et al. National Kidney Foundation’s Primer on Kidney Diseases. Philadelphia, PA: Elsevier; 2014.
6. Muriithi AK, Leung N, Valeri AM, et al. Biopsy-proven acute interstitial nephritis, 1993-2011: a case series. Am J Kidney Dis. 2014;64(4):558-566.
7. Blank ML, Parkin L, Paul C, Herbison P. A nationwide nested case-control study indicates an increased risk of acute interstitial nephritis with proton pump inhibitor use. Kidney Int. 2014;86(4):837-844.
8. Klepser DG, Collier DS, Cochran GL. Proton pump inhibitors and acute kidney injury: a nested case-control study. BMC Nephrology. 2013;14:150.
Be Sure to Look for Secondary Diabetes
A 63-year-old Hispanic woman was referred to endocrinology by her primary care provider for uncontrolled type 2 diabetes mellitus (T2DM), which was diagnosed 16 years ago. Her antidiabetic medications included insulin glargine (55 U bid), metformin (1,000 mg bid), and glipizide (10 mg bid). She also had known dyslipidemia, hypertension, and depression. There was a history of poorly controlled glucose (A1C between 9% and 13% in the past three years).
This was a relatively common new patient consult in our endocrine clinic. Upon entering the room, I was greeted by the patient and two family members. I quickly noticed the patient’s facial plethora and central obesity with comparatively thin extremities. Further inquiry revealed that the greatest challenge for the patient and her family was her bouts of severe depression, during which she would stop caring and cease to take her medications.
During the physical exam, mild but not significant supraclavicular and dorsocervical fat pads were appreciated. The exam was otherwise unremarkable, with no purple striae on the torso, abdomen, breasts, and extremities.
In addition to routine diabetes lab tests (ie, A1C, chemistry panel, lipid panel, urine microalbumin-to-creatinine ratio), an overnight 1-mg oral dexamethasone suppression test was ordered. Results of the latter were abnormal, and further workup confirmed Cushing disease (see Table 1 for results). The patient was referred for neurosurgery.
Continue for Discussion >>
DISCUSSION: SECONDARY DIABETES
It is well known that the prevalence of diabetes is skyrocketing, and medical offices are filled with affected patients. According to a 2011 report from the CDC, 90% to 95% of all diabetes cases are type 2, 5% are type 1 (autoimmune), and the rest (about 1% to 5%) are “other types” of diabetes.3 Due to these disproportionate statistics, clinicians often overlook the possibility of uncommon etiologies and assume all patients with diabetes have type 2—especially when the patient is overweight or obese.
Table 2 lists conditions and medications that may contribute to significant hyperglycemia.4 Some contributors are rather obvious (eg, status post pancreatectomy) or have no impact on treatment strategy (eg, chromosomal defects such as Down or Turner syndrome). However, certain conditions, such as Cushing syndrome, acromegaly, and hemochromatosis, can be relatively hard to recognize due to the variable rate of clinical manifestation, especially in the earlier stages of the disease. Experts have raised concerns that the prevalence of secondary diabetes (1% to 5%) may actually be underestimated due to “misdiagnosis” as T2DM.
Early detection of the underlying disorder, followed by initiation of appropriate treatment, is critical. It will not only improve but also may resolve the patient’s hyperglycemia, and it may also reverse or stop the damage to other vital organs.
The case patient had an unfortunate situation in which her Cushing syndrome was masked by commonly encountered diagnoses of hypertension, T2DM, obesity, and depression. Cushing is an easy diagnosis to miss, since it has an insidious onset and it can take more than five years for some of the physical findings to become evident.
Pancreatic cancer is another uncommon but critical disease worth mentioning. Pancreatic cancer should be in the differential diagnosis for previously euglycemic patients who experience abrupt elevation of glucose or previously well-managed patients whose glucose values quickly get out of control without obvious cause (eg, medication cessation, addition of glucocorticoid therapy, uncontrolled diet).
In our practice, we have encountered three patients with pancreatic cancer in this setting. The only sign was a sudden rise in glucose (300 to 500 mg/dL throughout the day) in patients whose A1C had been low (in the 6% range) with one or two oral medications. Thorough history taking did not reveal any potential causes for sudden hyperglycemia. Only one patient had a palpable mass on abdominal exam and elevated liver enzymes and bilirubin. Unfortunately, that patient died eight months later. The other two had favorable outcomes from surgery and chemotherapy. Early detection was the key for those two patients.
Next page: Conclusion >>
CONCLUSION
Since the majority of patients with diabetes have T2DM, it is easy to “default” and start treating all patients as such, especially if they are overweight or obese. However, up to 5% of patients actually have underlying disease that may cause or worsen their diabetic status. Overlooking these rare conditions can be detrimental to the patient, as it will adversely affect not only glycemic control but more importantly, overall health. Identifying the underlying disease will allow the patient to receive appropriate treatment, which may offload a significant burden on glycemic control and in some cases, cure the hyperglycemia.
REFERENCES
1. Nieman LK, Biller BM, Findling JW, et al. The diagnosis of Cushing’s syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2008;93(5):1526-1540.
2. Nieman LK. Establishing the cause of Cushing’s syndrome. Up-to-Date. www.uptodate.com/contents/establishing-the-cause-of-cushings-syndrome. Accessed June 24, 2015.
3. CDC. National Diabetes Fact Sheet, 2011. www.cdc.gov/diabetes/pubs/pdf/ndfs_2011.pdf. Accessed June 24, 2015.
4. Ganda OP. Prevalence and incidence of secondary and other types of diabetes. In: Diabetes in America. 2nd ed. Bethesda, MD: National Institutes of Health; 1995:69-84.
A 63-year-old Hispanic woman was referred to endocrinology by her primary care provider for uncontrolled type 2 diabetes mellitus (T2DM), which was diagnosed 16 years ago. Her antidiabetic medications included insulin glargine (55 U bid), metformin (1,000 mg bid), and glipizide (10 mg bid). She also had known dyslipidemia, hypertension, and depression. There was a history of poorly controlled glucose (A1C between 9% and 13% in the past three years).
This was a relatively common new patient consult in our endocrine clinic. Upon entering the room, I was greeted by the patient and two family members. I quickly noticed the patient’s facial plethora and central obesity with comparatively thin extremities. Further inquiry revealed that the greatest challenge for the patient and her family was her bouts of severe depression, during which she would stop caring and cease to take her medications.
During the physical exam, mild but not significant supraclavicular and dorsocervical fat pads were appreciated. The exam was otherwise unremarkable, with no purple striae on the torso, abdomen, breasts, and extremities.
In addition to routine diabetes lab tests (ie, A1C, chemistry panel, lipid panel, urine microalbumin-to-creatinine ratio), an overnight 1-mg oral dexamethasone suppression test was ordered. Results of the latter were abnormal, and further workup confirmed Cushing disease (see Table 1 for results). The patient was referred for neurosurgery.
Continue for Discussion >>
DISCUSSION: SECONDARY DIABETES
It is well known that the prevalence of diabetes is skyrocketing, and medical offices are filled with affected patients. According to a 2011 report from the CDC, 90% to 95% of all diabetes cases are type 2, 5% are type 1 (autoimmune), and the rest (about 1% to 5%) are “other types” of diabetes.3 Due to these disproportionate statistics, clinicians often overlook the possibility of uncommon etiologies and assume all patients with diabetes have type 2—especially when the patient is overweight or obese.
Table 2 lists conditions and medications that may contribute to significant hyperglycemia.4 Some contributors are rather obvious (eg, status post pancreatectomy) or have no impact on treatment strategy (eg, chromosomal defects such as Down or Turner syndrome). However, certain conditions, such as Cushing syndrome, acromegaly, and hemochromatosis, can be relatively hard to recognize due to the variable rate of clinical manifestation, especially in the earlier stages of the disease. Experts have raised concerns that the prevalence of secondary diabetes (1% to 5%) may actually be underestimated due to “misdiagnosis” as T2DM.
Early detection of the underlying disorder, followed by initiation of appropriate treatment, is critical. It will not only improve but also may resolve the patient’s hyperglycemia, and it may also reverse or stop the damage to other vital organs.
The case patient had an unfortunate situation in which her Cushing syndrome was masked by commonly encountered diagnoses of hypertension, T2DM, obesity, and depression. Cushing is an easy diagnosis to miss, since it has an insidious onset and it can take more than five years for some of the physical findings to become evident.
Pancreatic cancer is another uncommon but critical disease worth mentioning. Pancreatic cancer should be in the differential diagnosis for previously euglycemic patients who experience abrupt elevation of glucose or previously well-managed patients whose glucose values quickly get out of control without obvious cause (eg, medication cessation, addition of glucocorticoid therapy, uncontrolled diet).
In our practice, we have encountered three patients with pancreatic cancer in this setting. The only sign was a sudden rise in glucose (300 to 500 mg/dL throughout the day) in patients whose A1C had been low (in the 6% range) with one or two oral medications. Thorough history taking did not reveal any potential causes for sudden hyperglycemia. Only one patient had a palpable mass on abdominal exam and elevated liver enzymes and bilirubin. Unfortunately, that patient died eight months later. The other two had favorable outcomes from surgery and chemotherapy. Early detection was the key for those two patients.
Next page: Conclusion >>
CONCLUSION
Since the majority of patients with diabetes have T2DM, it is easy to “default” and start treating all patients as such, especially if they are overweight or obese. However, up to 5% of patients actually have underlying disease that may cause or worsen their diabetic status. Overlooking these rare conditions can be detrimental to the patient, as it will adversely affect not only glycemic control but more importantly, overall health. Identifying the underlying disease will allow the patient to receive appropriate treatment, which may offload a significant burden on glycemic control and in some cases, cure the hyperglycemia.
REFERENCES
1. Nieman LK, Biller BM, Findling JW, et al. The diagnosis of Cushing’s syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2008;93(5):1526-1540.
2. Nieman LK. Establishing the cause of Cushing’s syndrome. Up-to-Date. www.uptodate.com/contents/establishing-the-cause-of-cushings-syndrome. Accessed June 24, 2015.
3. CDC. National Diabetes Fact Sheet, 2011. www.cdc.gov/diabetes/pubs/pdf/ndfs_2011.pdf. Accessed June 24, 2015.
4. Ganda OP. Prevalence and incidence of secondary and other types of diabetes. In: Diabetes in America. 2nd ed. Bethesda, MD: National Institutes of Health; 1995:69-84.
A 63-year-old Hispanic woman was referred to endocrinology by her primary care provider for uncontrolled type 2 diabetes mellitus (T2DM), which was diagnosed 16 years ago. Her antidiabetic medications included insulin glargine (55 U bid), metformin (1,000 mg bid), and glipizide (10 mg bid). She also had known dyslipidemia, hypertension, and depression. There was a history of poorly controlled glucose (A1C between 9% and 13% in the past three years).
This was a relatively common new patient consult in our endocrine clinic. Upon entering the room, I was greeted by the patient and two family members. I quickly noticed the patient’s facial plethora and central obesity with comparatively thin extremities. Further inquiry revealed that the greatest challenge for the patient and her family was her bouts of severe depression, during which she would stop caring and cease to take her medications.
During the physical exam, mild but not significant supraclavicular and dorsocervical fat pads were appreciated. The exam was otherwise unremarkable, with no purple striae on the torso, abdomen, breasts, and extremities.
In addition to routine diabetes lab tests (ie, A1C, chemistry panel, lipid panel, urine microalbumin-to-creatinine ratio), an overnight 1-mg oral dexamethasone suppression test was ordered. Results of the latter were abnormal, and further workup confirmed Cushing disease (see Table 1 for results). The patient was referred for neurosurgery.
Continue for Discussion >>
DISCUSSION: SECONDARY DIABETES
It is well known that the prevalence of diabetes is skyrocketing, and medical offices are filled with affected patients. According to a 2011 report from the CDC, 90% to 95% of all diabetes cases are type 2, 5% are type 1 (autoimmune), and the rest (about 1% to 5%) are “other types” of diabetes.3 Due to these disproportionate statistics, clinicians often overlook the possibility of uncommon etiologies and assume all patients with diabetes have type 2—especially when the patient is overweight or obese.
Table 2 lists conditions and medications that may contribute to significant hyperglycemia.4 Some contributors are rather obvious (eg, status post pancreatectomy) or have no impact on treatment strategy (eg, chromosomal defects such as Down or Turner syndrome). However, certain conditions, such as Cushing syndrome, acromegaly, and hemochromatosis, can be relatively hard to recognize due to the variable rate of clinical manifestation, especially in the earlier stages of the disease. Experts have raised concerns that the prevalence of secondary diabetes (1% to 5%) may actually be underestimated due to “misdiagnosis” as T2DM.
Early detection of the underlying disorder, followed by initiation of appropriate treatment, is critical. It will not only improve but also may resolve the patient’s hyperglycemia, and it may also reverse or stop the damage to other vital organs.
The case patient had an unfortunate situation in which her Cushing syndrome was masked by commonly encountered diagnoses of hypertension, T2DM, obesity, and depression. Cushing is an easy diagnosis to miss, since it has an insidious onset and it can take more than five years for some of the physical findings to become evident.
Pancreatic cancer is another uncommon but critical disease worth mentioning. Pancreatic cancer should be in the differential diagnosis for previously euglycemic patients who experience abrupt elevation of glucose or previously well-managed patients whose glucose values quickly get out of control without obvious cause (eg, medication cessation, addition of glucocorticoid therapy, uncontrolled diet).
In our practice, we have encountered three patients with pancreatic cancer in this setting. The only sign was a sudden rise in glucose (300 to 500 mg/dL throughout the day) in patients whose A1C had been low (in the 6% range) with one or two oral medications. Thorough history taking did not reveal any potential causes for sudden hyperglycemia. Only one patient had a palpable mass on abdominal exam and elevated liver enzymes and bilirubin. Unfortunately, that patient died eight months later. The other two had favorable outcomes from surgery and chemotherapy. Early detection was the key for those two patients.
Next page: Conclusion >>
CONCLUSION
Since the majority of patients with diabetes have T2DM, it is easy to “default” and start treating all patients as such, especially if they are overweight or obese. However, up to 5% of patients actually have underlying disease that may cause or worsen their diabetic status. Overlooking these rare conditions can be detrimental to the patient, as it will adversely affect not only glycemic control but more importantly, overall health. Identifying the underlying disease will allow the patient to receive appropriate treatment, which may offload a significant burden on glycemic control and in some cases, cure the hyperglycemia.
REFERENCES
1. Nieman LK, Biller BM, Findling JW, et al. The diagnosis of Cushing’s syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2008;93(5):1526-1540.
2. Nieman LK. Establishing the cause of Cushing’s syndrome. Up-to-Date. www.uptodate.com/contents/establishing-the-cause-of-cushings-syndrome. Accessed June 24, 2015.
3. CDC. National Diabetes Fact Sheet, 2011. www.cdc.gov/diabetes/pubs/pdf/ndfs_2011.pdf. Accessed June 24, 2015.
4. Ganda OP. Prevalence and incidence of secondary and other types of diabetes. In: Diabetes in America. 2nd ed. Bethesda, MD: National Institutes of Health; 1995:69-84.
The Right Care at the Right Time and in the Right Place: The Role of Technology in the VHA
Embracing technology is nothing new for the VHA, whether it is telehealth, e-consults, or electronic health records. “The VHA is in a unique position to create the first truly national telemedicine network in the U.S.,” Adam W. Darkins, MD, then acting chief consultant of telemedicine at the VA wrote in a 2001 newsletter. “It is our collective task to make sure that if this happens, we have a system that can ‘plug and play.’”1
To better understand the progress in delivering health care, Federal Practitioner decided to devote this entire issue to the topic and to discuss the VHA and technology with Madhulika Agarwal, MD, MPH. As deputy under secretary for health for policy and services, Dr. Agarwal has been at the heart of the VHA’s embrace of many of these technologies for health care delivery and has been in a position to oversee their execution. More than anyone else at the VHA, she is familiar with the potential and limitations of telehealth.
Below is an edited and condensed version of the interview. To hear the complete interview, visit http://www.fedprac.com/multimedia/multimedia-library.html.
Importance of Telehealth to the VHA
Madhulika Agarwal, MD, MPH. Our goal is to ensure that veterans have optimal health and that we deliver the best health care with a focus on timely access and with an exceptional experience. And over the years, we have been building technologic tools so that we can provide the right care at the right time and in the right place. Telehealth affords veterans the convenience of accessing primary or specialized care services either from their local VA community clinic or from the privacy of their own home.
Now we have many virtual access solutions. The home telehealth, clinical video teleconferencing, store-and-forward technologies, e-consults, My HealtheVet, plus SCAN-ECHO [Specialty Care Access Network-Extension for Community Healthcare Outcomes]; and these all have demonstrated that they are mission-critical tools, which improve and expand the access for veterans who may have difficulty accessing care for multiple reasons.
It could be some clinical issues where there are transportation difficulties, such as for veterans with spinal cord injury, or mild traumatic brain injury, or geographic barriers. Many of our veterans, I would say roughly 40% to 45% of them, live in rural and highly rural areas where they may not have access to care nearby. Or it could be further exacerbated with geographic challenges by inclement weather or the drive times. And lastly, I would say it’s the lack of specialists in these rural communities where many of our veterans live.
VHA is successfully integrating into the existing technical administrative clinical infrastructures, and this infrastructure provides a reliable and robust IT network. We have an electronic health record. We provide national policy guidance regarding health information security, credentialing, privileging, etc. And our strategic goal has been to have personalized, proactive, patient-driven care; and telehealth supports that goal.
Improving Veteran Access
Dr. Agarwal. It’s interesting that both the Choice program, which is part of the Veterans Access, Choice, and Accountability Act of 2014, known as VACAA, and telehealth aim at improving veteran access to care. Under the Choice program, many veterans now have the option to access community partner health care rather than waiting for a VA appointment, or traveling to a VA facility when the geographic distance is more than 40 miles, or if the appointment in the VA is not available for 30 days.
The Choice program and telehealth are 2 very concrete examples of VHA’s transformation from a facility or provider-centric health care delivery model to a model that puts the veteran’s needs at the center and improving the veteran’s access to resources to meet their health care needs.
Related: Committed to Showing Results at the VA
More than 717,000 veterans have accessed VA care through telehealth in fiscal year [FY] 14, and 45% of these veterans live in rural and highly rural areas. In FY14, the total for veterans using telehealth represented about an 18% growth from the prior year; and the telehealth services provide access to help in more than 45 different specialty areas, including those areas where VHA has a particular expertise, especially, for example, in mental health that may not be available from the local community partner.
Telehealth Uses
Dr. Agarwal. A veteran who is living in a rural area, let’s just say in some rural part of Maryland, and has to commute to the Baltimore VA, which you know is an inner-city VA medical center, to keep his appointment for a mental health condition with his VA provider. Now, using telemental health, this veteran can access this provider from his or her own home through encrypted video conferencing and complete the telemental health visit in the comfort of his or her own home so that they are not subject to the traffic and other challenges that they would otherwise face and get even more stressed than what they started out with. The ability, the convenience of having the service of counseling or cognitive behavioral therapy into their own homes, is just remarkable.
Another example that I could cite for you would be an appointment in the hearing aid clinic. So a veteran who lives in the Florida Keys normally would have to travel 5 hours from the Florida Keys, go to Miami, stay in a hotel overnight so that they can go to their appointment at 8 am. Instead, the veteran now can visit the Key West clinic and have his hearing aid adjusted by a VA audiologist who’s located in Miami; and it saves the entire trip.
The third one I will cite you has to do with the C&P [Compensation & Pension] exams. Now, a veteran living even out of the country can access a VA provider in Connecticut or some [other] state, using the encrypted video conferencing; and they can have the whole clinical evaluation for C&P completed using the video conferencing. These are some of the examples of how telehealth has been used very successfully.
Technologic and Educational Challenges
Dr. Agarwal. We have been a pioneer of telehealth. And with that, of course, all those challenges come into play. And we certainly have implementation challenges that include provider and patient education and their buy-in into the use of technology and providing services as well as the technology itself and some administrative issues. They can all be very closely linked.
You know, one illustrative example that I just cited earlier about video conferencing is one such example into the veteran’s home. It is very convenient.… We started to implement this home telemental health program a couple of years ago. But since then, about 108,000 veterans have accessed using the video conferencing technology; but fewer than 2,000 or so have done it from their own home. And that’s largely because the current video visit from home is quite cumbersome. It requires passwords for each visit. It requires that the veteran download VA-licensed software on their own device. And in addition, there are restrictions because of the availability of the broadband Internet connectivity, which is required for the video visit—more so in the rural areas.
Related: Preparing the Military Health System for the 21st Century
Our general counsel is reviewing and attempting to resolve state licensure requirements that have been raised by some states, because the veterans here receive care at home and outside of our VA brick-and-mortar facility, as well as the legality of VA providers potentially prescribing a controlled substance for a veteran at home without a prior in-person office visit.
But to overcome the provider challenges, the national telehealth training and resource center has been working on training the providers in the use of telehealth. Roughly 11,400 VA staff have been trained in the use of telehealth in FY14. We have currently 144 facility telehealth coordinators and more than 1,100 telehealth clinical technicians who assist with training and outreach for both VA staff and veteran patients.
Legal/Security Challenges
Dr. Agarwal. High-speed connectivity happens to be one of the key ones.… Using 4G services, I think, is going to be essential for every veteran regardless of rurality. And when these 4G services are not available, that certainly hinders the ability to provide telehealth to all veterans. Having the right security with full data encryption is essential so that we can protect the private health information of the veterans.
But unfortunately, at this time, there is not an easy way to do that. I think a lot of innovation is required so that we can make it much easier for the veterans with 1-button access, both for the veterans as well as for the providers. And that’s going to require significant effort in the grid technology as well as overcoming certain legal requirements.
What Is Driving Telehealth?
Dr. Agarwal. The real driver here has to be the veterans’ needs, not the needs of telehealth nor the clinical services or operations. I think the whole goal here is that we must use technology to the extent possible. We have to move toward virtual access as the norm.
As much as possible, we should provide the virtual access in the veterans’ homes or wherever the veterans would like to receive their services. Make the connectivity as simple as possible for the veterans and move beyond the concept of the episodic visit so that the health information and self-care management tools are available to the veterans at all times. And that essentially needs to be the overarching strategy, and that should drive how we develop the technologies to provide the services.
Data Analysis
Dr. Agarwal. We have the general enrollee data. We look at access gaps in clinical services and the telehealth activity data for our program management and oversight as well as in developing an overarching strategy for the clinical services and telehealth services. It’s done somewhat in conjunction. And our outcome analysis shows that there has been significant reduction in admissions and bed days of care with the use of telehealth.
For example, in FY14, an analysis of 10,621 veterans who were newly enrolled in home telehealth with noninstitutional care needs and chronic care management categories had a decrease of about 54% of bed days of care. This was about a 32% decrease in the hospital admissions compared [with] the same patient data prior to the enrollment and home telehealth. The analysis of telemental health outcomes shows that there was a 35% reduction in acute psychiatric bed days of care for veterans receiving CBT [cognitive behavioral therapy] or the clinical video conferencing telemental health in FY14 when it was compared [with] the utilization in the prior year.
Telehealth Pilot Programs
Dr. Agarwal. I must admit that there are many more programs that begin in the facilities, but at the national level. The first one is the tele-ICU implementation, where VISN 23 is supporting VISN 15, 5 of the medical centers with clinical video teleconferencing capability for live interactive consults with ICU specialists; and it covers about 78 beds. VISN 10 is supporting VISN 7 in 7 of their medical centers, which covers about 72 beds.
Another program, which is in the pilot phase right now, is the telewound care pilot, which is being implemented in 6 VISNs and combines the use of home telehealth, clinical video teleconferencing, and store-and-forward telehealth technologies to create access to a continuum of wound care options across multiple patients and provider settings and locations, all with the goal of enhancing and improving wound care treatment and healing.… The initial phase has been that all the participating facilities have been identified, and some of the operations manuals have been developed.
Related: Acting Surgeon General Confident in Battle Against Tobacco, Ebola, and Preventable Diseases
The third quarter of this year, we will have a completion of the operations manual Provider Training and Treatment Template. The local sites are also working on the infrastructure and knowledge base so that this project can be completed by FY15.
And the last highlight that I’ll mention, which is in its very early stages, is a low-acuity/low-intensity pilot with the focus on health promotion and health prevention behaviors, such as tobacco cessation, weight management, and newly diagnosed but stable veterans with diabetes, high blood pressure, and heart failure, using a web-based browser technology.
VA Telehealth Leadership
Dr. Agarwal. Overall, when we start to look at the monumental impact of technology on other industries, such as banking, shopping, travel, and even personal communications, the emerging technologies continue to change the overall landscape of all these environments. This is an exciting time to be in the health care industry, because I think we have lagged somewhat behind in using technology. But as we look forward, the consumer-driven health care is going to become the norm.
As you know, VA has long been a pioneer with electronic medical records and with virtual modalities, such as telehealth both in the home and in the community, the use of patient web portals, such as My HealtheVet, secure messaging for various apps, kiosks; and we remain on the forefront of developing and utilizing these approaches to enhance health care delivery.
We all know that health care in the U.S. is complex and fragmented. VA is looking to become the benchmark in U.S. health care delivery, aiding in the transformation of the delivery of services for veterans and families, focusing on unified, integrated, and personalized virtual services that seamlessly connect them with the state-of-the-art health care system.
1. Darkins A. A message from the acting chief consultant: telemedicine grows throughout VHA. Telemedicine News. 2001;1(1):1. U.S. Department of Veterans Affairs Website. http://www.telehealth.va.gov/newsletter/2001/040201-newsletter_spring_01.pdf. Published April 2, 2001. Accessed June 23, 2015.
Embracing technology is nothing new for the VHA, whether it is telehealth, e-consults, or electronic health records. “The VHA is in a unique position to create the first truly national telemedicine network in the U.S.,” Adam W. Darkins, MD, then acting chief consultant of telemedicine at the VA wrote in a 2001 newsletter. “It is our collective task to make sure that if this happens, we have a system that can ‘plug and play.’”1
To better understand the progress in delivering health care, Federal Practitioner decided to devote this entire issue to the topic and to discuss the VHA and technology with Madhulika Agarwal, MD, MPH. As deputy under secretary for health for policy and services, Dr. Agarwal has been at the heart of the VHA’s embrace of many of these technologies for health care delivery and has been in a position to oversee their execution. More than anyone else at the VHA, she is familiar with the potential and limitations of telehealth.
Below is an edited and condensed version of the interview. To hear the complete interview, visit http://www.fedprac.com/multimedia/multimedia-library.html.
Importance of Telehealth to the VHA
Madhulika Agarwal, MD, MPH. Our goal is to ensure that veterans have optimal health and that we deliver the best health care with a focus on timely access and with an exceptional experience. And over the years, we have been building technologic tools so that we can provide the right care at the right time and in the right place. Telehealth affords veterans the convenience of accessing primary or specialized care services either from their local VA community clinic or from the privacy of their own home.
Now we have many virtual access solutions. The home telehealth, clinical video teleconferencing, store-and-forward technologies, e-consults, My HealtheVet, plus SCAN-ECHO [Specialty Care Access Network-Extension for Community Healthcare Outcomes]; and these all have demonstrated that they are mission-critical tools, which improve and expand the access for veterans who may have difficulty accessing care for multiple reasons.
It could be some clinical issues where there are transportation difficulties, such as for veterans with spinal cord injury, or mild traumatic brain injury, or geographic barriers. Many of our veterans, I would say roughly 40% to 45% of them, live in rural and highly rural areas where they may not have access to care nearby. Or it could be further exacerbated with geographic challenges by inclement weather or the drive times. And lastly, I would say it’s the lack of specialists in these rural communities where many of our veterans live.
VHA is successfully integrating into the existing technical administrative clinical infrastructures, and this infrastructure provides a reliable and robust IT network. We have an electronic health record. We provide national policy guidance regarding health information security, credentialing, privileging, etc. And our strategic goal has been to have personalized, proactive, patient-driven care; and telehealth supports that goal.
Improving Veteran Access
Dr. Agarwal. It’s interesting that both the Choice program, which is part of the Veterans Access, Choice, and Accountability Act of 2014, known as VACAA, and telehealth aim at improving veteran access to care. Under the Choice program, many veterans now have the option to access community partner health care rather than waiting for a VA appointment, or traveling to a VA facility when the geographic distance is more than 40 miles, or if the appointment in the VA is not available for 30 days.
The Choice program and telehealth are 2 very concrete examples of VHA’s transformation from a facility or provider-centric health care delivery model to a model that puts the veteran’s needs at the center and improving the veteran’s access to resources to meet their health care needs.
Related: Committed to Showing Results at the VA
More than 717,000 veterans have accessed VA care through telehealth in fiscal year [FY] 14, and 45% of these veterans live in rural and highly rural areas. In FY14, the total for veterans using telehealth represented about an 18% growth from the prior year; and the telehealth services provide access to help in more than 45 different specialty areas, including those areas where VHA has a particular expertise, especially, for example, in mental health that may not be available from the local community partner.
Telehealth Uses
Dr. Agarwal. A veteran who is living in a rural area, let’s just say in some rural part of Maryland, and has to commute to the Baltimore VA, which you know is an inner-city VA medical center, to keep his appointment for a mental health condition with his VA provider. Now, using telemental health, this veteran can access this provider from his or her own home through encrypted video conferencing and complete the telemental health visit in the comfort of his or her own home so that they are not subject to the traffic and other challenges that they would otherwise face and get even more stressed than what they started out with. The ability, the convenience of having the service of counseling or cognitive behavioral therapy into their own homes, is just remarkable.
Another example that I could cite for you would be an appointment in the hearing aid clinic. So a veteran who lives in the Florida Keys normally would have to travel 5 hours from the Florida Keys, go to Miami, stay in a hotel overnight so that they can go to their appointment at 8 am. Instead, the veteran now can visit the Key West clinic and have his hearing aid adjusted by a VA audiologist who’s located in Miami; and it saves the entire trip.
The third one I will cite you has to do with the C&P [Compensation & Pension] exams. Now, a veteran living even out of the country can access a VA provider in Connecticut or some [other] state, using the encrypted video conferencing; and they can have the whole clinical evaluation for C&P completed using the video conferencing. These are some of the examples of how telehealth has been used very successfully.
Technologic and Educational Challenges
Dr. Agarwal. We have been a pioneer of telehealth. And with that, of course, all those challenges come into play. And we certainly have implementation challenges that include provider and patient education and their buy-in into the use of technology and providing services as well as the technology itself and some administrative issues. They can all be very closely linked.
You know, one illustrative example that I just cited earlier about video conferencing is one such example into the veteran’s home. It is very convenient.… We started to implement this home telemental health program a couple of years ago. But since then, about 108,000 veterans have accessed using the video conferencing technology; but fewer than 2,000 or so have done it from their own home. And that’s largely because the current video visit from home is quite cumbersome. It requires passwords for each visit. It requires that the veteran download VA-licensed software on their own device. And in addition, there are restrictions because of the availability of the broadband Internet connectivity, which is required for the video visit—more so in the rural areas.
Related: Preparing the Military Health System for the 21st Century
Our general counsel is reviewing and attempting to resolve state licensure requirements that have been raised by some states, because the veterans here receive care at home and outside of our VA brick-and-mortar facility, as well as the legality of VA providers potentially prescribing a controlled substance for a veteran at home without a prior in-person office visit.
But to overcome the provider challenges, the national telehealth training and resource center has been working on training the providers in the use of telehealth. Roughly 11,400 VA staff have been trained in the use of telehealth in FY14. We have currently 144 facility telehealth coordinators and more than 1,100 telehealth clinical technicians who assist with training and outreach for both VA staff and veteran patients.
Legal/Security Challenges
Dr. Agarwal. High-speed connectivity happens to be one of the key ones.… Using 4G services, I think, is going to be essential for every veteran regardless of rurality. And when these 4G services are not available, that certainly hinders the ability to provide telehealth to all veterans. Having the right security with full data encryption is essential so that we can protect the private health information of the veterans.
But unfortunately, at this time, there is not an easy way to do that. I think a lot of innovation is required so that we can make it much easier for the veterans with 1-button access, both for the veterans as well as for the providers. And that’s going to require significant effort in the grid technology as well as overcoming certain legal requirements.
What Is Driving Telehealth?
Dr. Agarwal. The real driver here has to be the veterans’ needs, not the needs of telehealth nor the clinical services or operations. I think the whole goal here is that we must use technology to the extent possible. We have to move toward virtual access as the norm.
As much as possible, we should provide the virtual access in the veterans’ homes or wherever the veterans would like to receive their services. Make the connectivity as simple as possible for the veterans and move beyond the concept of the episodic visit so that the health information and self-care management tools are available to the veterans at all times. And that essentially needs to be the overarching strategy, and that should drive how we develop the technologies to provide the services.
Data Analysis
Dr. Agarwal. We have the general enrollee data. We look at access gaps in clinical services and the telehealth activity data for our program management and oversight as well as in developing an overarching strategy for the clinical services and telehealth services. It’s done somewhat in conjunction. And our outcome analysis shows that there has been significant reduction in admissions and bed days of care with the use of telehealth.
For example, in FY14, an analysis of 10,621 veterans who were newly enrolled in home telehealth with noninstitutional care needs and chronic care management categories had a decrease of about 54% of bed days of care. This was about a 32% decrease in the hospital admissions compared [with] the same patient data prior to the enrollment and home telehealth. The analysis of telemental health outcomes shows that there was a 35% reduction in acute psychiatric bed days of care for veterans receiving CBT [cognitive behavioral therapy] or the clinical video conferencing telemental health in FY14 when it was compared [with] the utilization in the prior year.
Telehealth Pilot Programs
Dr. Agarwal. I must admit that there are many more programs that begin in the facilities, but at the national level. The first one is the tele-ICU implementation, where VISN 23 is supporting VISN 15, 5 of the medical centers with clinical video teleconferencing capability for live interactive consults with ICU specialists; and it covers about 78 beds. VISN 10 is supporting VISN 7 in 7 of their medical centers, which covers about 72 beds.
Another program, which is in the pilot phase right now, is the telewound care pilot, which is being implemented in 6 VISNs and combines the use of home telehealth, clinical video teleconferencing, and store-and-forward telehealth technologies to create access to a continuum of wound care options across multiple patients and provider settings and locations, all with the goal of enhancing and improving wound care treatment and healing.… The initial phase has been that all the participating facilities have been identified, and some of the operations manuals have been developed.
Related: Acting Surgeon General Confident in Battle Against Tobacco, Ebola, and Preventable Diseases
The third quarter of this year, we will have a completion of the operations manual Provider Training and Treatment Template. The local sites are also working on the infrastructure and knowledge base so that this project can be completed by FY15.
And the last highlight that I’ll mention, which is in its very early stages, is a low-acuity/low-intensity pilot with the focus on health promotion and health prevention behaviors, such as tobacco cessation, weight management, and newly diagnosed but stable veterans with diabetes, high blood pressure, and heart failure, using a web-based browser technology.
VA Telehealth Leadership
Dr. Agarwal. Overall, when we start to look at the monumental impact of technology on other industries, such as banking, shopping, travel, and even personal communications, the emerging technologies continue to change the overall landscape of all these environments. This is an exciting time to be in the health care industry, because I think we have lagged somewhat behind in using technology. But as we look forward, the consumer-driven health care is going to become the norm.
As you know, VA has long been a pioneer with electronic medical records and with virtual modalities, such as telehealth both in the home and in the community, the use of patient web portals, such as My HealtheVet, secure messaging for various apps, kiosks; and we remain on the forefront of developing and utilizing these approaches to enhance health care delivery.
We all know that health care in the U.S. is complex and fragmented. VA is looking to become the benchmark in U.S. health care delivery, aiding in the transformation of the delivery of services for veterans and families, focusing on unified, integrated, and personalized virtual services that seamlessly connect them with the state-of-the-art health care system.
Embracing technology is nothing new for the VHA, whether it is telehealth, e-consults, or electronic health records. “The VHA is in a unique position to create the first truly national telemedicine network in the U.S.,” Adam W. Darkins, MD, then acting chief consultant of telemedicine at the VA wrote in a 2001 newsletter. “It is our collective task to make sure that if this happens, we have a system that can ‘plug and play.’”1
To better understand the progress in delivering health care, Federal Practitioner decided to devote this entire issue to the topic and to discuss the VHA and technology with Madhulika Agarwal, MD, MPH. As deputy under secretary for health for policy and services, Dr. Agarwal has been at the heart of the VHA’s embrace of many of these technologies for health care delivery and has been in a position to oversee their execution. More than anyone else at the VHA, she is familiar with the potential and limitations of telehealth.
Below is an edited and condensed version of the interview. To hear the complete interview, visit http://www.fedprac.com/multimedia/multimedia-library.html.
Importance of Telehealth to the VHA
Madhulika Agarwal, MD, MPH. Our goal is to ensure that veterans have optimal health and that we deliver the best health care with a focus on timely access and with an exceptional experience. And over the years, we have been building technologic tools so that we can provide the right care at the right time and in the right place. Telehealth affords veterans the convenience of accessing primary or specialized care services either from their local VA community clinic or from the privacy of their own home.
Now we have many virtual access solutions. The home telehealth, clinical video teleconferencing, store-and-forward technologies, e-consults, My HealtheVet, plus SCAN-ECHO [Specialty Care Access Network-Extension for Community Healthcare Outcomes]; and these all have demonstrated that they are mission-critical tools, which improve and expand the access for veterans who may have difficulty accessing care for multiple reasons.
It could be some clinical issues where there are transportation difficulties, such as for veterans with spinal cord injury, or mild traumatic brain injury, or geographic barriers. Many of our veterans, I would say roughly 40% to 45% of them, live in rural and highly rural areas where they may not have access to care nearby. Or it could be further exacerbated with geographic challenges by inclement weather or the drive times. And lastly, I would say it’s the lack of specialists in these rural communities where many of our veterans live.
VHA is successfully integrating into the existing technical administrative clinical infrastructures, and this infrastructure provides a reliable and robust IT network. We have an electronic health record. We provide national policy guidance regarding health information security, credentialing, privileging, etc. And our strategic goal has been to have personalized, proactive, patient-driven care; and telehealth supports that goal.
Improving Veteran Access
Dr. Agarwal. It’s interesting that both the Choice program, which is part of the Veterans Access, Choice, and Accountability Act of 2014, known as VACAA, and telehealth aim at improving veteran access to care. Under the Choice program, many veterans now have the option to access community partner health care rather than waiting for a VA appointment, or traveling to a VA facility when the geographic distance is more than 40 miles, or if the appointment in the VA is not available for 30 days.
The Choice program and telehealth are 2 very concrete examples of VHA’s transformation from a facility or provider-centric health care delivery model to a model that puts the veteran’s needs at the center and improving the veteran’s access to resources to meet their health care needs.
Related: Committed to Showing Results at the VA
More than 717,000 veterans have accessed VA care through telehealth in fiscal year [FY] 14, and 45% of these veterans live in rural and highly rural areas. In FY14, the total for veterans using telehealth represented about an 18% growth from the prior year; and the telehealth services provide access to help in more than 45 different specialty areas, including those areas where VHA has a particular expertise, especially, for example, in mental health that may not be available from the local community partner.
Telehealth Uses
Dr. Agarwal. A veteran who is living in a rural area, let’s just say in some rural part of Maryland, and has to commute to the Baltimore VA, which you know is an inner-city VA medical center, to keep his appointment for a mental health condition with his VA provider. Now, using telemental health, this veteran can access this provider from his or her own home through encrypted video conferencing and complete the telemental health visit in the comfort of his or her own home so that they are not subject to the traffic and other challenges that they would otherwise face and get even more stressed than what they started out with. The ability, the convenience of having the service of counseling or cognitive behavioral therapy into their own homes, is just remarkable.
Another example that I could cite for you would be an appointment in the hearing aid clinic. So a veteran who lives in the Florida Keys normally would have to travel 5 hours from the Florida Keys, go to Miami, stay in a hotel overnight so that they can go to their appointment at 8 am. Instead, the veteran now can visit the Key West clinic and have his hearing aid adjusted by a VA audiologist who’s located in Miami; and it saves the entire trip.
The third one I will cite you has to do with the C&P [Compensation & Pension] exams. Now, a veteran living even out of the country can access a VA provider in Connecticut or some [other] state, using the encrypted video conferencing; and they can have the whole clinical evaluation for C&P completed using the video conferencing. These are some of the examples of how telehealth has been used very successfully.
Technologic and Educational Challenges
Dr. Agarwal. We have been a pioneer of telehealth. And with that, of course, all those challenges come into play. And we certainly have implementation challenges that include provider and patient education and their buy-in into the use of technology and providing services as well as the technology itself and some administrative issues. They can all be very closely linked.
You know, one illustrative example that I just cited earlier about video conferencing is one such example into the veteran’s home. It is very convenient.… We started to implement this home telemental health program a couple of years ago. But since then, about 108,000 veterans have accessed using the video conferencing technology; but fewer than 2,000 or so have done it from their own home. And that’s largely because the current video visit from home is quite cumbersome. It requires passwords for each visit. It requires that the veteran download VA-licensed software on their own device. And in addition, there are restrictions because of the availability of the broadband Internet connectivity, which is required for the video visit—more so in the rural areas.
Related: Preparing the Military Health System for the 21st Century
Our general counsel is reviewing and attempting to resolve state licensure requirements that have been raised by some states, because the veterans here receive care at home and outside of our VA brick-and-mortar facility, as well as the legality of VA providers potentially prescribing a controlled substance for a veteran at home without a prior in-person office visit.
But to overcome the provider challenges, the national telehealth training and resource center has been working on training the providers in the use of telehealth. Roughly 11,400 VA staff have been trained in the use of telehealth in FY14. We have currently 144 facility telehealth coordinators and more than 1,100 telehealth clinical technicians who assist with training and outreach for both VA staff and veteran patients.
Legal/Security Challenges
Dr. Agarwal. High-speed connectivity happens to be one of the key ones.… Using 4G services, I think, is going to be essential for every veteran regardless of rurality. And when these 4G services are not available, that certainly hinders the ability to provide telehealth to all veterans. Having the right security with full data encryption is essential so that we can protect the private health information of the veterans.
But unfortunately, at this time, there is not an easy way to do that. I think a lot of innovation is required so that we can make it much easier for the veterans with 1-button access, both for the veterans as well as for the providers. And that’s going to require significant effort in the grid technology as well as overcoming certain legal requirements.
What Is Driving Telehealth?
Dr. Agarwal. The real driver here has to be the veterans’ needs, not the needs of telehealth nor the clinical services or operations. I think the whole goal here is that we must use technology to the extent possible. We have to move toward virtual access as the norm.
As much as possible, we should provide the virtual access in the veterans’ homes or wherever the veterans would like to receive their services. Make the connectivity as simple as possible for the veterans and move beyond the concept of the episodic visit so that the health information and self-care management tools are available to the veterans at all times. And that essentially needs to be the overarching strategy, and that should drive how we develop the technologies to provide the services.
Data Analysis
Dr. Agarwal. We have the general enrollee data. We look at access gaps in clinical services and the telehealth activity data for our program management and oversight as well as in developing an overarching strategy for the clinical services and telehealth services. It’s done somewhat in conjunction. And our outcome analysis shows that there has been significant reduction in admissions and bed days of care with the use of telehealth.
For example, in FY14, an analysis of 10,621 veterans who were newly enrolled in home telehealth with noninstitutional care needs and chronic care management categories had a decrease of about 54% of bed days of care. This was about a 32% decrease in the hospital admissions compared [with] the same patient data prior to the enrollment and home telehealth. The analysis of telemental health outcomes shows that there was a 35% reduction in acute psychiatric bed days of care for veterans receiving CBT [cognitive behavioral therapy] or the clinical video conferencing telemental health in FY14 when it was compared [with] the utilization in the prior year.
Telehealth Pilot Programs
Dr. Agarwal. I must admit that there are many more programs that begin in the facilities, but at the national level. The first one is the tele-ICU implementation, where VISN 23 is supporting VISN 15, 5 of the medical centers with clinical video teleconferencing capability for live interactive consults with ICU specialists; and it covers about 78 beds. VISN 10 is supporting VISN 7 in 7 of their medical centers, which covers about 72 beds.
Another program, which is in the pilot phase right now, is the telewound care pilot, which is being implemented in 6 VISNs and combines the use of home telehealth, clinical video teleconferencing, and store-and-forward telehealth technologies to create access to a continuum of wound care options across multiple patients and provider settings and locations, all with the goal of enhancing and improving wound care treatment and healing.… The initial phase has been that all the participating facilities have been identified, and some of the operations manuals have been developed.
Related: Acting Surgeon General Confident in Battle Against Tobacco, Ebola, and Preventable Diseases
The third quarter of this year, we will have a completion of the operations manual Provider Training and Treatment Template. The local sites are also working on the infrastructure and knowledge base so that this project can be completed by FY15.
And the last highlight that I’ll mention, which is in its very early stages, is a low-acuity/low-intensity pilot with the focus on health promotion and health prevention behaviors, such as tobacco cessation, weight management, and newly diagnosed but stable veterans with diabetes, high blood pressure, and heart failure, using a web-based browser technology.
VA Telehealth Leadership
Dr. Agarwal. Overall, when we start to look at the monumental impact of technology on other industries, such as banking, shopping, travel, and even personal communications, the emerging technologies continue to change the overall landscape of all these environments. This is an exciting time to be in the health care industry, because I think we have lagged somewhat behind in using technology. But as we look forward, the consumer-driven health care is going to become the norm.
As you know, VA has long been a pioneer with electronic medical records and with virtual modalities, such as telehealth both in the home and in the community, the use of patient web portals, such as My HealtheVet, secure messaging for various apps, kiosks; and we remain on the forefront of developing and utilizing these approaches to enhance health care delivery.
We all know that health care in the U.S. is complex and fragmented. VA is looking to become the benchmark in U.S. health care delivery, aiding in the transformation of the delivery of services for veterans and families, focusing on unified, integrated, and personalized virtual services that seamlessly connect them with the state-of-the-art health care system.
1. Darkins A. A message from the acting chief consultant: telemedicine grows throughout VHA. Telemedicine News. 2001;1(1):1. U.S. Department of Veterans Affairs Website. http://www.telehealth.va.gov/newsletter/2001/040201-newsletter_spring_01.pdf. Published April 2, 2001. Accessed June 23, 2015.
1. Darkins A. A message from the acting chief consultant: telemedicine grows throughout VHA. Telemedicine News. 2001;1(1):1. U.S. Department of Veterans Affairs Website. http://www.telehealth.va.gov/newsletter/2001/040201-newsletter_spring_01.pdf. Published April 2, 2001. Accessed June 23, 2015.
Understanding Hematuria: Causes
Q) I have been treating a 60-year-old man with a long history of microscopic hematuria and waxing/waning proteinuria. What could be the cause of his hematuria?
Hematuria is a consequence of erythrocytes, or red blood cells (RBCs), in the urine. This can cause a visible change in color, considered gross or macroscopic hematuria; or the blood may only be visible under microscopy or by urine dipstick (referred to as microscopic hematuria).
Both findings are followed up with urinalysis to quantify erythrocytes, protein, and presence of casts and to review RBC morphology. This information will assist in determining if the hematuria is glomerular or nonglomerular in origin.1
The examination and treatment plan for nonglomerular hematuria will focus on urinary tract diseases. If the patient is found to have glomerular hematuria, the focus will be on diseases of the kidney. A thorough history and physical should be performed in addition to urinalysis.
Glomerular disease is suggested in those with micro- or macroscopic proteinuria, proteinuria > 1 g/24h, or an absence of casts. Our index patient has microscopic hematuria and “waxing/waning” (unquantified) proteinuria, suggesting glomerular origin.
There are a number of renal causes for glomerular bleeding, including primary glomerulonephritis, multisystem autoimmune disease, and hereditary or infective glomerulonephritis.2 Renal biopsy is recommended for patients who have hypertension, proteinuria, and hematuria, to determine the cause and thus determine the appropriate treatment.
Amy L. Hazel, RN, MSN, CNP
Kidney & Hypertension Consultants, Canton, Ohio
REFERENCES
1. Greenberg A. Primer on Kidney Diseases. 5th ed. Philadelphia, PA: Elsevier Saunders; 2005.
2. Barratt J, Feehally J. IgA nephropathy [disease of the month]. J Am Soc Nephrol. 2005;16(7): 2088-2097.
Q) I have been treating a 60-year-old man with a long history of microscopic hematuria and waxing/waning proteinuria. What could be the cause of his hematuria?
Hematuria is a consequence of erythrocytes, or red blood cells (RBCs), in the urine. This can cause a visible change in color, considered gross or macroscopic hematuria; or the blood may only be visible under microscopy or by urine dipstick (referred to as microscopic hematuria).
Both findings are followed up with urinalysis to quantify erythrocytes, protein, and presence of casts and to review RBC morphology. This information will assist in determining if the hematuria is glomerular or nonglomerular in origin.1
The examination and treatment plan for nonglomerular hematuria will focus on urinary tract diseases. If the patient is found to have glomerular hematuria, the focus will be on diseases of the kidney. A thorough history and physical should be performed in addition to urinalysis.
Glomerular disease is suggested in those with micro- or macroscopic proteinuria, proteinuria > 1 g/24h, or an absence of casts. Our index patient has microscopic hematuria and “waxing/waning” (unquantified) proteinuria, suggesting glomerular origin.
There are a number of renal causes for glomerular bleeding, including primary glomerulonephritis, multisystem autoimmune disease, and hereditary or infective glomerulonephritis.2 Renal biopsy is recommended for patients who have hypertension, proteinuria, and hematuria, to determine the cause and thus determine the appropriate treatment.
Amy L. Hazel, RN, MSN, CNP
Kidney & Hypertension Consultants, Canton, Ohio
REFERENCES
1. Greenberg A. Primer on Kidney Diseases. 5th ed. Philadelphia, PA: Elsevier Saunders; 2005.
2. Barratt J, Feehally J. IgA nephropathy [disease of the month]. J Am Soc Nephrol. 2005;16(7): 2088-2097.
Q) I have been treating a 60-year-old man with a long history of microscopic hematuria and waxing/waning proteinuria. What could be the cause of his hematuria?
Hematuria is a consequence of erythrocytes, or red blood cells (RBCs), in the urine. This can cause a visible change in color, considered gross or macroscopic hematuria; or the blood may only be visible under microscopy or by urine dipstick (referred to as microscopic hematuria).
Both findings are followed up with urinalysis to quantify erythrocytes, protein, and presence of casts and to review RBC morphology. This information will assist in determining if the hematuria is glomerular or nonglomerular in origin.1
The examination and treatment plan for nonglomerular hematuria will focus on urinary tract diseases. If the patient is found to have glomerular hematuria, the focus will be on diseases of the kidney. A thorough history and physical should be performed in addition to urinalysis.
Glomerular disease is suggested in those with micro- or macroscopic proteinuria, proteinuria > 1 g/24h, or an absence of casts. Our index patient has microscopic hematuria and “waxing/waning” (unquantified) proteinuria, suggesting glomerular origin.
There are a number of renal causes for glomerular bleeding, including primary glomerulonephritis, multisystem autoimmune disease, and hereditary or infective glomerulonephritis.2 Renal biopsy is recommended for patients who have hypertension, proteinuria, and hematuria, to determine the cause and thus determine the appropriate treatment.
Amy L. Hazel, RN, MSN, CNP
Kidney & Hypertension Consultants, Canton, Ohio
REFERENCES
1. Greenberg A. Primer on Kidney Diseases. 5th ed. Philadelphia, PA: Elsevier Saunders; 2005.
2. Barratt J, Feehally J. IgA nephropathy [disease of the month]. J Am Soc Nephrol. 2005;16(7): 2088-2097.
Understanding Hematuria: IgA Nephropathy
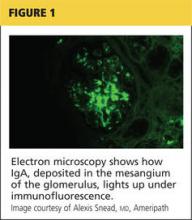
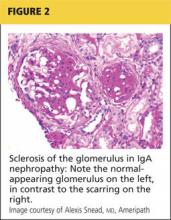
Q) My hematuria patient had more significant proteinuria recently, so the nephrologist sent him for kidney biopsy. It was read as IgA nephropathy: “classic mesangial staining on IF with moderate-advanced chronic injury (15/32 gloms globally sclerosed, 40% IFTA, mild arteriosclerosis).” What exactly does this mean, and what is IgA nephropathy?
Immunoglobulin A (IgA) nephropathy is the most common type of glomerulonephritis; up to 40% of patients with IgA nephropathy develop end-stage renal disease within 20 years of diagnosis. More common in men, IgA nephropathy is usually diagnosed in people in their second or third decades of life.2,3
Considered an autoimmune disease, IgA nephropathy typically presents with macroscopic or gross hematuria that occurs within 24 hours of the onset of an upper respiratory infection (URI). The hematuria typically resolves quickly, in one to three days. An individual bacterial or viral element has not yet been identified.
IgA nephropathy is an immune response to the URI. IgA is secreted from mucosal surfaces at the back of the mouth and then deposited in the glomerular mesangium, a “stalk of cells” associated with the capillaries of the renal glomerulus.1 It is speculated that genetics, environment, and/or hypersensitivity to food antigens may play a part in IgA nephropathy. Results from biopsies of blood relatives of patients with confirmed IgA nephropathy suggest a familial role.1
IgA nephropathy is prevalent in persons who live in the Pacific Rim and Southern Europe. However, this association may be the result of a sampling error due to investigation of all microscopic hematuria in these areas. In all, 90% of IgA is sporadic.4 It is often asymptomatic, aside from occasional back and flank pain secondary to inflammation of the renal capsule. Unfortunately, many patients develop renal impairment and hypertension by the time they are diagnosed.
Renal biopsy is used to confirm/diagnose IgA nephropathy. IgA, deposited in the mesangium of the glomerulus, lights up under immunofluorescence (IF; see Figure 1). In some patients, this mesangial deposition results in sclerosis, scarring, and/or inflammation of the glomerulus (see Figure 2).
An international panel of experts created guidelines (the Oxford classification system) for reporting IgA kidney biopsies. Six adverse pathologic features have been identified:
• Mesangial cellularity score
• Percentage of segmental sclerosis
• Endocapillary hypercellularity
• Cellular and/or fibrocellular crescents
• Percentage of interstitial fibrosis/tubular atrophy (IFTA)
• Arteriosclerosis score5,6
Interstitial fibrosis, crescents, and as little as 25% glomerular sclerosis found on biopsy increases the likelihood of disease progression.5 Clinically, hypertension, a reduced glomerular filtration rate, increasing age, and proteinuria of > 1g/24h have been identified as risk factors for progression of IgA nephropathy. Up to 30% of patients diagnosed will require renal replacement therapy within 20 years.1
The case patient’s findings include the typical IF staining of IgA in the glomerulus. The biopsy report also indicates that 40% of the glomeruli (less than half) have interstitial fibrosis and that the structural integrity of the tubules has been affected secondary to IgA accumulation in the mesangium. These findings are suggestive of progressive disease.
There is no known way to stop IgA deposition in the mesangium. Tonsillectomy to reduce mucosal IgA release has been suggested but is controversial.
Treatment of IgA nephropathy focuses on preserving renal function by reducing proteinuria through the use of ACE inhibitors and/or angiotensin receptor blockers. Aggressive blood pressure management is achieved by blocking the renin-angiotensin-aldosterone system (RAAS).
Other methods for decreasing progression of renal disease are directed at reducing the immune and inflammatory response via immunosuppressant medications.3 The use of immunosuppressive agents, though controversial, is recommended for those who have progressive disease and/or proteinuria despite achieving target blood pressure with full RAAS blockade.1
Amy L. Hazel, RN, MSN, CNP
Kidney & Hypertension Consultants, Canton, Ohio
REFERENCES
1. Greenberg A. Primer on Kidney Diseases. 5th ed. Philadelphia, PA: Elsevier Saunders; 2005.
2. Barratt J, Feehally J. IgA nephropathy [disease of the month]. J Am Soc Nephrol. 2005;16(7): 2088-2097.
3. Barratt J, Feehally J. Treatment of IgA nephropathy. Kidney Int. 2006;69(11):1934-1938.
4. Johnson R, Feehally J. Comprehensive Clinical Nephrology. 2nd ed. London: Mosby; 2000.
5. Walsh M, Sar A, Lee D, et al. Histopathologic features aid in predicting risk for progression of IgA nephropathy. Clin J Am Soc Nephrol. 2010; 5(3):425-430.
6. Roberts I. The Oxford classification of IgA nephropathy: pathology definitions, correlations, and reproducibility. Kidney Int. 2009; 76(5):546-556.
Q) My hematuria patient had more significant proteinuria recently, so the nephrologist sent him for kidney biopsy. It was read as IgA nephropathy: “classic mesangial staining on IF with moderate-advanced chronic injury (15/32 gloms globally sclerosed, 40% IFTA, mild arteriosclerosis).” What exactly does this mean, and what is IgA nephropathy?
Immunoglobulin A (IgA) nephropathy is the most common type of glomerulonephritis; up to 40% of patients with IgA nephropathy develop end-stage renal disease within 20 years of diagnosis. More common in men, IgA nephropathy is usually diagnosed in people in their second or third decades of life.2,3
Considered an autoimmune disease, IgA nephropathy typically presents with macroscopic or gross hematuria that occurs within 24 hours of the onset of an upper respiratory infection (URI). The hematuria typically resolves quickly, in one to three days. An individual bacterial or viral element has not yet been identified.
IgA nephropathy is an immune response to the URI. IgA is secreted from mucosal surfaces at the back of the mouth and then deposited in the glomerular mesangium, a “stalk of cells” associated with the capillaries of the renal glomerulus.1 It is speculated that genetics, environment, and/or hypersensitivity to food antigens may play a part in IgA nephropathy. Results from biopsies of blood relatives of patients with confirmed IgA nephropathy suggest a familial role.1
IgA nephropathy is prevalent in persons who live in the Pacific Rim and Southern Europe. However, this association may be the result of a sampling error due to investigation of all microscopic hematuria in these areas. In all, 90% of IgA is sporadic.4 It is often asymptomatic, aside from occasional back and flank pain secondary to inflammation of the renal capsule. Unfortunately, many patients develop renal impairment and hypertension by the time they are diagnosed.
Renal biopsy is used to confirm/diagnose IgA nephropathy. IgA, deposited in the mesangium of the glomerulus, lights up under immunofluorescence (IF; see Figure 1). In some patients, this mesangial deposition results in sclerosis, scarring, and/or inflammation of the glomerulus (see Figure 2).
An international panel of experts created guidelines (the Oxford classification system) for reporting IgA kidney biopsies. Six adverse pathologic features have been identified:
• Mesangial cellularity score
• Percentage of segmental sclerosis
• Endocapillary hypercellularity
• Cellular and/or fibrocellular crescents
• Percentage of interstitial fibrosis/tubular atrophy (IFTA)
• Arteriosclerosis score5,6
Interstitial fibrosis, crescents, and as little as 25% glomerular sclerosis found on biopsy increases the likelihood of disease progression.5 Clinically, hypertension, a reduced glomerular filtration rate, increasing age, and proteinuria of > 1g/24h have been identified as risk factors for progression of IgA nephropathy. Up to 30% of patients diagnosed will require renal replacement therapy within 20 years.1
The case patient’s findings include the typical IF staining of IgA in the glomerulus. The biopsy report also indicates that 40% of the glomeruli (less than half) have interstitial fibrosis and that the structural integrity of the tubules has been affected secondary to IgA accumulation in the mesangium. These findings are suggestive of progressive disease.
There is no known way to stop IgA deposition in the mesangium. Tonsillectomy to reduce mucosal IgA release has been suggested but is controversial.
Treatment of IgA nephropathy focuses on preserving renal function by reducing proteinuria through the use of ACE inhibitors and/or angiotensin receptor blockers. Aggressive blood pressure management is achieved by blocking the renin-angiotensin-aldosterone system (RAAS).
Other methods for decreasing progression of renal disease are directed at reducing the immune and inflammatory response via immunosuppressant medications.3 The use of immunosuppressive agents, though controversial, is recommended for those who have progressive disease and/or proteinuria despite achieving target blood pressure with full RAAS blockade.1
Amy L. Hazel, RN, MSN, CNP
Kidney & Hypertension Consultants, Canton, Ohio
REFERENCES
1. Greenberg A. Primer on Kidney Diseases. 5th ed. Philadelphia, PA: Elsevier Saunders; 2005.
2. Barratt J, Feehally J. IgA nephropathy [disease of the month]. J Am Soc Nephrol. 2005;16(7): 2088-2097.
3. Barratt J, Feehally J. Treatment of IgA nephropathy. Kidney Int. 2006;69(11):1934-1938.
4. Johnson R, Feehally J. Comprehensive Clinical Nephrology. 2nd ed. London: Mosby; 2000.
5. Walsh M, Sar A, Lee D, et al. Histopathologic features aid in predicting risk for progression of IgA nephropathy. Clin J Am Soc Nephrol. 2010; 5(3):425-430.
6. Roberts I. The Oxford classification of IgA nephropathy: pathology definitions, correlations, and reproducibility. Kidney Int. 2009; 76(5):546-556.
Q) My hematuria patient had more significant proteinuria recently, so the nephrologist sent him for kidney biopsy. It was read as IgA nephropathy: “classic mesangial staining on IF with moderate-advanced chronic injury (15/32 gloms globally sclerosed, 40% IFTA, mild arteriosclerosis).” What exactly does this mean, and what is IgA nephropathy?
Immunoglobulin A (IgA) nephropathy is the most common type of glomerulonephritis; up to 40% of patients with IgA nephropathy develop end-stage renal disease within 20 years of diagnosis. More common in men, IgA nephropathy is usually diagnosed in people in their second or third decades of life.2,3
Considered an autoimmune disease, IgA nephropathy typically presents with macroscopic or gross hematuria that occurs within 24 hours of the onset of an upper respiratory infection (URI). The hematuria typically resolves quickly, in one to three days. An individual bacterial or viral element has not yet been identified.
IgA nephropathy is an immune response to the URI. IgA is secreted from mucosal surfaces at the back of the mouth and then deposited in the glomerular mesangium, a “stalk of cells” associated with the capillaries of the renal glomerulus.1 It is speculated that genetics, environment, and/or hypersensitivity to food antigens may play a part in IgA nephropathy. Results from biopsies of blood relatives of patients with confirmed IgA nephropathy suggest a familial role.1
IgA nephropathy is prevalent in persons who live in the Pacific Rim and Southern Europe. However, this association may be the result of a sampling error due to investigation of all microscopic hematuria in these areas. In all, 90% of IgA is sporadic.4 It is often asymptomatic, aside from occasional back and flank pain secondary to inflammation of the renal capsule. Unfortunately, many patients develop renal impairment and hypertension by the time they are diagnosed.
Renal biopsy is used to confirm/diagnose IgA nephropathy. IgA, deposited in the mesangium of the glomerulus, lights up under immunofluorescence (IF; see Figure 1). In some patients, this mesangial deposition results in sclerosis, scarring, and/or inflammation of the glomerulus (see Figure 2).
An international panel of experts created guidelines (the Oxford classification system) for reporting IgA kidney biopsies. Six adverse pathologic features have been identified:
• Mesangial cellularity score
• Percentage of segmental sclerosis
• Endocapillary hypercellularity
• Cellular and/or fibrocellular crescents
• Percentage of interstitial fibrosis/tubular atrophy (IFTA)
• Arteriosclerosis score5,6
Interstitial fibrosis, crescents, and as little as 25% glomerular sclerosis found on biopsy increases the likelihood of disease progression.5 Clinically, hypertension, a reduced glomerular filtration rate, increasing age, and proteinuria of > 1g/24h have been identified as risk factors for progression of IgA nephropathy. Up to 30% of patients diagnosed will require renal replacement therapy within 20 years.1
The case patient’s findings include the typical IF staining of IgA in the glomerulus. The biopsy report also indicates that 40% of the glomeruli (less than half) have interstitial fibrosis and that the structural integrity of the tubules has been affected secondary to IgA accumulation in the mesangium. These findings are suggestive of progressive disease.
There is no known way to stop IgA deposition in the mesangium. Tonsillectomy to reduce mucosal IgA release has been suggested but is controversial.
Treatment of IgA nephropathy focuses on preserving renal function by reducing proteinuria through the use of ACE inhibitors and/or angiotensin receptor blockers. Aggressive blood pressure management is achieved by blocking the renin-angiotensin-aldosterone system (RAAS).
Other methods for decreasing progression of renal disease are directed at reducing the immune and inflammatory response via immunosuppressant medications.3 The use of immunosuppressive agents, though controversial, is recommended for those who have progressive disease and/or proteinuria despite achieving target blood pressure with full RAAS blockade.1
Amy L. Hazel, RN, MSN, CNP
Kidney & Hypertension Consultants, Canton, Ohio
REFERENCES
1. Greenberg A. Primer on Kidney Diseases. 5th ed. Philadelphia, PA: Elsevier Saunders; 2005.
2. Barratt J, Feehally J. IgA nephropathy [disease of the month]. J Am Soc Nephrol. 2005;16(7): 2088-2097.
3. Barratt J, Feehally J. Treatment of IgA nephropathy. Kidney Int. 2006;69(11):1934-1938.
4. Johnson R, Feehally J. Comprehensive Clinical Nephrology. 2nd ed. London: Mosby; 2000.
5. Walsh M, Sar A, Lee D, et al. Histopathologic features aid in predicting risk for progression of IgA nephropathy. Clin J Am Soc Nephrol. 2010; 5(3):425-430.
6. Roberts I. The Oxford classification of IgA nephropathy: pathology definitions, correlations, and reproducibility. Kidney Int. 2009; 76(5):546-556.
Weighing the Options for Obesity Meds
In June 2013, the American Medical Association classified obesity as a disease. Since then, several medical societies have published guidelines to help clinicians improve care of affected patients. One avenue is, of course, pharmacologic treatment.
Until recently, there was only one FDA-approved medication for chronic weight loss on the market: orlistat, which was approved in 1999. (Phentermine and diethylpropion are only indicated for short-term use). After a long hiatus, the FDA approved two additional agents (phentermine/topiramate and lorcaserin)in 2012 and another two (liraglutide and bupropion/naltrexone) in 2014.
While clinicians appreciate having options for managing their patients’ conditions, in this case, many are overwhelmed by the choices. Most health care providers have not received formal training in obesity management. This column will attempt to fill the information gap in terms of what agents are available and what factors should be assessed before prescribing any of them.
Proviso: Experts claim obesity is a chronic disease, similar to hypertension, and should be managed as such. Although not discussed here, the most important aspect of weight loss and maintenance is lifestyle intervention (diet, exercise, and behavioral modification). It should be emphasized that no medication works by itself; all should be used as an adjunct tool to reinforce adherence to lifestyle changes.1 Furthermore, patients may be disappointed to learn that without these changes, the weight may return when they cease medication use.
CASE Deb, age 61, presents to your office for routine follow-up. She has a history of type 2 diabetes, dyslipidemia, hypertension, atrial fibrillation, depression, and chronic back pain due to a herniated disc. Her medications include insulin glargine, glyburide, pioglitazone, atorvastatin, metoprolol, paroxetine, and acetaminophen/hydrocodone.
Her vital signs include a blood pressure of 143/91 mm Hg and a pulse of 93 beats/min. She has a BMI of 37 and a waist circumference of 35 in.
Deb, concerned about her weight, would like to discuss weight-loss options. She has tried three different commercial programs; each time, she was able to lose 30 to 50 lb in three to six months but regained the weight once she stopped the program. She reports excessive appetite as the main reason for her rebound weight gain. Her exercise is limited due to her back pain.
She recently tried OTC orlistat but could not tolerate it due to flatulence and fecal urgency. She reports an incident in which she couldn’t reach the bathroom in time.
Continue for Discussion >>
DISCUSSION
The Endocrine Society’s recommended approaches to obesity management include diet, exercise, and behavioral modification for patients with a BMI ≥ 25. The addition of pharmacotherapy can be considered for those with a BMI ≥ 30 or with a BMI ≥ 27 and one or more weight-related comorbidities (eg, diabetes, dyslipidemia, hypertension). This matches the FDA-approved product labeling for chronic weight-loss medications. Bariatric surgery should be considered for patients with a BMI ≥ 40 or with a BMI ≥ 35 and at least one weight-related comorbidity.
Orlistat
Orlistat is available OTC in a 60-mg thrice-daily form. A higher dosage (120 mg tid) is available via prescription. Orlistat decreases fat absorption in the gastrointestinal (GI) tract by inhibiting GI lipase. Average weight loss with orlistat is 3% at first and second year, and, when compared with placebo, 2.4% greater at four years.2
Orlistat should be prescribed with a multivitamin due to decreased absorption of fat-soluble vitamins. It is contraindicated in patients with malabsorption syndrome and gallbladder disease (> 2% incidence3). It can increase cyclosporine exposure, and rare cases of liver failure have been reported. The most common adverse effect is related to steatorrhea. Of the available options, orlistat is the only medication that has no effect on neurohormonal regulation in appetite control and metabolic rate, which may be a limiting factor.
CASE POINT Due to Deb’s intolerance of and embarrassment with GI adverse effects, she requests an alternative medication.
Lorcaserin
Lorcaserin is a selective serotonin 2C receptor agonist that reduces appetite by affecting anorexigenic pro-opiomelanocortin (POMC) neurons in the hypothalamus. Of note, lorcaserin “selects” the 2C receptor instead of 2A and 2B; 2B receptors are found in both aortic and mitral valves, which may explain the association between fenfluramine/phentermine (commonly known as “fen/phen” and withdrawn from the market in 1997) and possible cardiac valvulopathy. (Fenfluramine is an amphetamine derivative that nonselectively stimulates serotonin release and inhibits reuptake.)
Lorcaserin comes in a 10-mg twice-daily dosage. In studies, patients taking lorcaserin had an average weight loss of 3.3% more than those taking placebo at one year; weight loss was maintained through the second year for those who continued on medication. However, those who stopped the medication at one year had regained their weight by the two-year mark.4
It is recommended that the medication be discontinued if patients don’t achieve a loss of more than 5% of body weight by 12 weeks.
In a study that enrolled diabetic patients, lorcaserin also demonstrated a 0.9% reduction in A1C, which is similar to or even better than some oral antidiabetic medications.4 However, since the manufacturer was not planning for an antidiabetic indication, A1C was only a secondary endpoint. The reduction is most likely due to decreased caloric intake and weight loss.
The most common adverse effects of lorcaserin include headache, dizziness, and fatigue. The discontinuation rate due to intolerance was 8.6%, compared to 6.7% with placebo.5
Although this was not observed in clinical studies, co-administration of lorcaserin (a serotonin receptor agonist) with other serotonergic or antidopaminergic agents can theoretically cause serotonin syndrome or neuroleptic malignant syndrome–like reactions. Caution is therefore advisable when prescribing these agents. The package insert carries a warning for cardiac valvulopathy due to fen/phen’s history and a lack of long-term cardiovascular safety data.
CASE POINT Deb is taking paroxetine (an SSRI) for her depression. Since you are concerned about serotonin syndrome, you decide to keep exploring options. Checking the package insert for phentermine/topiramate, you learn that it does not have a potential adverse reaction related to co-administration with SSRIs.
Phentermine/Topiramate
Phentermine, a sympathomimetic medication, was approved for short-term (12-week) use for weight loss in the 1960s. Topiramate, an antiseizure and migraine prophylactic medication, enhances appetite suppression—although the exact mechanism of action is unknown.1
Four once-daily doses are available: 3.75/23 mg, 7.5/46 mg, 11.25/69 mg, and 15/92 mg. Dosing starts with 3.75/23 mg for two weeks, then increases to 7.5/46 mg. If a loss of 5% or more of body weight is achieved, the patient can continue the dosage; if not, it can be increased to 11.25/69 mg for two weeks and then to 15/92 mg. The average weight loss for mid and maximum dose was 6.6% and 8.6% greater than placebo at one year.5
Commonly reported adverse effects include paraesthesia, dysgeusia (distortion of sense of taste), dizziness, insomnia, constipation, and dry mouth. Due to phentermine’s sympathomimetic action, mild increases in heart rate and blood pressure were reported. The Endocrine Society recommends against the use of phentermine in patients with uncontrolled hypertension and a history of heart disease.1
Weight loss is generally not recommended during pregnancy, and all weight loss medications are classified as category X for pregnancy. Strict caution is advised with this particular agent, as topiramate has known teratogenicity and therefore comes with a Risk Evaluation Mitigation Strategy. Patients must be advised to use appropriate contraception while taking topiramate, and a pregnancy test should be performed before medication commencement and monthly thereafter.
Abrupt cessation of topiramate can cause seizure. When taking the 15/92-mg dosage, the patient should reduce to one tablet every other day for at least one week before discontinuation.
CASE POINT Deb’s blood pressure is still not at goal. This, along with her history of atrial fibrillation and high pulse, prompts you to consider another option.
Bupropion/Naltrexone
Bupropion, a widely used antidepressant, inhibits the uptake of norepinephrine and dopamine and thereby blocks the reward pathway that various foods can induce. Naltrexone, an opioid antagonist, blocks the opioid pathway and can be helpful in enhancing weight loss.
This combination comes in an 8/90-mg tablet. The suggested titration regimen is to start with one tablet per day and increase by one tablet every week, up to the maximum dosage of two tablets twice a day. Average weight loss was 3.1% greater than placebo at one year with the maximum dosage. An A1C reduction of 0.6% was seen in diabetic patients.6 It is recommended to stop the medication and seek an alternative treatment option if > 5% loss of body weight is not achieved by 12 weeks.
GI adverse effects (eg, nausea and vomiting) are common; these can be reduced with a slower titration regimen or by prescribing a maximum of one tablet twice daily (instead of two). Every antidepressant carries suicidal risk, and caution is advised with their use. Bupropion can also lower the seizure threshold, and it is contraindicated for patients with seizure disorder. It is also contraindicated in patients who are undergoing abrupt cessation of alcohol, benzodiazepines, or barbiturates. It can increase pulse and blood pressure during early titration; regular blood pressure monitoring is warranted.
CASE POINT Due to Deb’s opioid usage and uncontrolled hypertension, you discuss a final option that was recently approved for weight loss.
Liraglutide
This glucagon-like peptide-1 (GLP1) receptor agonist affects the brain to suppress/control appetite, slows down gastric emptying, and induces early satiety. A 3-mg dosage was approved in December 2014, but 0.6-, 1.2-, and 1.8-mg dosages have been available since 2010 for patients with type 2 diabetes.
Average weight loss was 4.5% greater than placebo at one year.7 If < 4% weight loss is achieved by 16 weeks, consider using an alternative agent.
The most common adverse effect is GI upset, which could be related to the mechanism of action (slower gastric emptying). Although self-reported GI upset was high (39%), the actual discontinuation rate was low (2.9% for nausea, 1.7% for vomiting, and 1.4% for diarrhea).3
This adverse effect could, in certain contexts, be considered “wanted,” since it discourages overeating or eating too quickly. My clinical pearl is to tell patients taking liraglutide that they are “trapped” and have to eat smaller portions and eat more slowly or they will be more prone to GI effects. With this strategy, we can encourage portion control and responsibility for behavior. (Please note that this is my experience with the diabetic dosage of liraglutide; I do not have any clinical experience with the obesity dosage, which was not clinically available at the time of writing.)
Both branded versions of liraglutide carry a black-box warning for thyroid C-cell tumors, which were observed in rodents but unproven in humans. The medication is contraindicated in patients with medullary thyroid cancer or with multiple endocrine neoplasia 2 syndrome. Increased rates of acute pancreatitis, cholecystitis, and cholelithiasis were seen in studies, and caution is advised.
Continue for A Word About Meds That Cause Weight Gain >>
A WORD ABOUT MEDS THAT CAUSE WEIGHT GAIN
The Endocrine Society has published a list of medications that can influence weight gain, along with suggestions for alternative agents that are either weight neutral or promote weight loss.
Note that our case patient, Deb, is taking insulin, a sulfonylurea (glyburide), and thiazolidinedione (pioglitazone) for diabetes—all of which can promote weight gain. Guidelines suggest choosing metformin, DPP4 inhibitors, GLP1 agonists, amylin analog, and SGLT2 inhibitors instead when weight gain is a major concern.1
Guidelines also suggest using ACE inhibitors, angiotensin receptor blockers, and calcium channel blockers instead of β-blockers as firstline antihypertensive therapy for diabetic patients.1 Adequate blood pressure and lipid control are imperative in diabetes management.
CASE POINT Deb would need better hypertension control before she considers weight-loss medication. Since she is also taking paroxetine, which among SSRIs is associated with greatest weight gain, a changed to fluoxetine or sertraline should be considered.2
Next page: Conclusion >>
CONCLUSION
There are now five medications approved by the FDA for chronic weight loss, with more to come. Agents with different mechanisms of action give us options to help obese patients and hopefully reduce and prevent obesity-related complications. It is important for clinicians to be competent in managing obesity, especially since we live in an era in which the disease is considered pandemic.
REFERENCES
1. Apovian CM, Aronne LJ, Bessesen DH, et al; Endocrine Society. Pharmacological management of obesity: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2015;100(2):342-362.
2. Xenical [package insert]. South San Francisco, CA: Genentech USA, Inc; 2012.
3. Fujioka K. Safety and tolerability of medications approved for chronic weight management. Obesity (Silver Spring). 2015;22 (suppl 1):S7-S11.
4. Belviq [package insert]. Woodcliff Lake, NJ: Eisai Inc; 2015.
5. Qsymia [package insert]. Mountain View, CA: Vivus, Inc; 2013.
6. Contrave [package insert]. Deerfield, IL: Takeda USA, Inc; 2014.
7. Saxenda [package insert]. Plainsboro, NJ: Novo Nordisk; 2014.
In June 2013, the American Medical Association classified obesity as a disease. Since then, several medical societies have published guidelines to help clinicians improve care of affected patients. One avenue is, of course, pharmacologic treatment.
Until recently, there was only one FDA-approved medication for chronic weight loss on the market: orlistat, which was approved in 1999. (Phentermine and diethylpropion are only indicated for short-term use). After a long hiatus, the FDA approved two additional agents (phentermine/topiramate and lorcaserin)in 2012 and another two (liraglutide and bupropion/naltrexone) in 2014.
While clinicians appreciate having options for managing their patients’ conditions, in this case, many are overwhelmed by the choices. Most health care providers have not received formal training in obesity management. This column will attempt to fill the information gap in terms of what agents are available and what factors should be assessed before prescribing any of them.
Proviso: Experts claim obesity is a chronic disease, similar to hypertension, and should be managed as such. Although not discussed here, the most important aspect of weight loss and maintenance is lifestyle intervention (diet, exercise, and behavioral modification). It should be emphasized that no medication works by itself; all should be used as an adjunct tool to reinforce adherence to lifestyle changes.1 Furthermore, patients may be disappointed to learn that without these changes, the weight may return when they cease medication use.
CASE Deb, age 61, presents to your office for routine follow-up. She has a history of type 2 diabetes, dyslipidemia, hypertension, atrial fibrillation, depression, and chronic back pain due to a herniated disc. Her medications include insulin glargine, glyburide, pioglitazone, atorvastatin, metoprolol, paroxetine, and acetaminophen/hydrocodone.
Her vital signs include a blood pressure of 143/91 mm Hg and a pulse of 93 beats/min. She has a BMI of 37 and a waist circumference of 35 in.
Deb, concerned about her weight, would like to discuss weight-loss options. She has tried three different commercial programs; each time, she was able to lose 30 to 50 lb in three to six months but regained the weight once she stopped the program. She reports excessive appetite as the main reason for her rebound weight gain. Her exercise is limited due to her back pain.
She recently tried OTC orlistat but could not tolerate it due to flatulence and fecal urgency. She reports an incident in which she couldn’t reach the bathroom in time.
Continue for Discussion >>
DISCUSSION
The Endocrine Society’s recommended approaches to obesity management include diet, exercise, and behavioral modification for patients with a BMI ≥ 25. The addition of pharmacotherapy can be considered for those with a BMI ≥ 30 or with a BMI ≥ 27 and one or more weight-related comorbidities (eg, diabetes, dyslipidemia, hypertension). This matches the FDA-approved product labeling for chronic weight-loss medications. Bariatric surgery should be considered for patients with a BMI ≥ 40 or with a BMI ≥ 35 and at least one weight-related comorbidity.
Orlistat
Orlistat is available OTC in a 60-mg thrice-daily form. A higher dosage (120 mg tid) is available via prescription. Orlistat decreases fat absorption in the gastrointestinal (GI) tract by inhibiting GI lipase. Average weight loss with orlistat is 3% at first and second year, and, when compared with placebo, 2.4% greater at four years.2
Orlistat should be prescribed with a multivitamin due to decreased absorption of fat-soluble vitamins. It is contraindicated in patients with malabsorption syndrome and gallbladder disease (> 2% incidence3). It can increase cyclosporine exposure, and rare cases of liver failure have been reported. The most common adverse effect is related to steatorrhea. Of the available options, orlistat is the only medication that has no effect on neurohormonal regulation in appetite control and metabolic rate, which may be a limiting factor.
CASE POINT Due to Deb’s intolerance of and embarrassment with GI adverse effects, she requests an alternative medication.
Lorcaserin
Lorcaserin is a selective serotonin 2C receptor agonist that reduces appetite by affecting anorexigenic pro-opiomelanocortin (POMC) neurons in the hypothalamus. Of note, lorcaserin “selects” the 2C receptor instead of 2A and 2B; 2B receptors are found in both aortic and mitral valves, which may explain the association between fenfluramine/phentermine (commonly known as “fen/phen” and withdrawn from the market in 1997) and possible cardiac valvulopathy. (Fenfluramine is an amphetamine derivative that nonselectively stimulates serotonin release and inhibits reuptake.)
Lorcaserin comes in a 10-mg twice-daily dosage. In studies, patients taking lorcaserin had an average weight loss of 3.3% more than those taking placebo at one year; weight loss was maintained through the second year for those who continued on medication. However, those who stopped the medication at one year had regained their weight by the two-year mark.4
It is recommended that the medication be discontinued if patients don’t achieve a loss of more than 5% of body weight by 12 weeks.
In a study that enrolled diabetic patients, lorcaserin also demonstrated a 0.9% reduction in A1C, which is similar to or even better than some oral antidiabetic medications.4 However, since the manufacturer was not planning for an antidiabetic indication, A1C was only a secondary endpoint. The reduction is most likely due to decreased caloric intake and weight loss.
The most common adverse effects of lorcaserin include headache, dizziness, and fatigue. The discontinuation rate due to intolerance was 8.6%, compared to 6.7% with placebo.5
Although this was not observed in clinical studies, co-administration of lorcaserin (a serotonin receptor agonist) with other serotonergic or antidopaminergic agents can theoretically cause serotonin syndrome or neuroleptic malignant syndrome–like reactions. Caution is therefore advisable when prescribing these agents. The package insert carries a warning for cardiac valvulopathy due to fen/phen’s history and a lack of long-term cardiovascular safety data.
CASE POINT Deb is taking paroxetine (an SSRI) for her depression. Since you are concerned about serotonin syndrome, you decide to keep exploring options. Checking the package insert for phentermine/topiramate, you learn that it does not have a potential adverse reaction related to co-administration with SSRIs.
Phentermine/Topiramate
Phentermine, a sympathomimetic medication, was approved for short-term (12-week) use for weight loss in the 1960s. Topiramate, an antiseizure and migraine prophylactic medication, enhances appetite suppression—although the exact mechanism of action is unknown.1
Four once-daily doses are available: 3.75/23 mg, 7.5/46 mg, 11.25/69 mg, and 15/92 mg. Dosing starts with 3.75/23 mg for two weeks, then increases to 7.5/46 mg. If a loss of 5% or more of body weight is achieved, the patient can continue the dosage; if not, it can be increased to 11.25/69 mg for two weeks and then to 15/92 mg. The average weight loss for mid and maximum dose was 6.6% and 8.6% greater than placebo at one year.5
Commonly reported adverse effects include paraesthesia, dysgeusia (distortion of sense of taste), dizziness, insomnia, constipation, and dry mouth. Due to phentermine’s sympathomimetic action, mild increases in heart rate and blood pressure were reported. The Endocrine Society recommends against the use of phentermine in patients with uncontrolled hypertension and a history of heart disease.1
Weight loss is generally not recommended during pregnancy, and all weight loss medications are classified as category X for pregnancy. Strict caution is advised with this particular agent, as topiramate has known teratogenicity and therefore comes with a Risk Evaluation Mitigation Strategy. Patients must be advised to use appropriate contraception while taking topiramate, and a pregnancy test should be performed before medication commencement and monthly thereafter.
Abrupt cessation of topiramate can cause seizure. When taking the 15/92-mg dosage, the patient should reduce to one tablet every other day for at least one week before discontinuation.
CASE POINT Deb’s blood pressure is still not at goal. This, along with her history of atrial fibrillation and high pulse, prompts you to consider another option.
Bupropion/Naltrexone
Bupropion, a widely used antidepressant, inhibits the uptake of norepinephrine and dopamine and thereby blocks the reward pathway that various foods can induce. Naltrexone, an opioid antagonist, blocks the opioid pathway and can be helpful in enhancing weight loss.
This combination comes in an 8/90-mg tablet. The suggested titration regimen is to start with one tablet per day and increase by one tablet every week, up to the maximum dosage of two tablets twice a day. Average weight loss was 3.1% greater than placebo at one year with the maximum dosage. An A1C reduction of 0.6% was seen in diabetic patients.6 It is recommended to stop the medication and seek an alternative treatment option if > 5% loss of body weight is not achieved by 12 weeks.
GI adverse effects (eg, nausea and vomiting) are common; these can be reduced with a slower titration regimen or by prescribing a maximum of one tablet twice daily (instead of two). Every antidepressant carries suicidal risk, and caution is advised with their use. Bupropion can also lower the seizure threshold, and it is contraindicated for patients with seizure disorder. It is also contraindicated in patients who are undergoing abrupt cessation of alcohol, benzodiazepines, or barbiturates. It can increase pulse and blood pressure during early titration; regular blood pressure monitoring is warranted.
CASE POINT Due to Deb’s opioid usage and uncontrolled hypertension, you discuss a final option that was recently approved for weight loss.
Liraglutide
This glucagon-like peptide-1 (GLP1) receptor agonist affects the brain to suppress/control appetite, slows down gastric emptying, and induces early satiety. A 3-mg dosage was approved in December 2014, but 0.6-, 1.2-, and 1.8-mg dosages have been available since 2010 for patients with type 2 diabetes.
Average weight loss was 4.5% greater than placebo at one year.7 If < 4% weight loss is achieved by 16 weeks, consider using an alternative agent.
The most common adverse effect is GI upset, which could be related to the mechanism of action (slower gastric emptying). Although self-reported GI upset was high (39%), the actual discontinuation rate was low (2.9% for nausea, 1.7% for vomiting, and 1.4% for diarrhea).3
This adverse effect could, in certain contexts, be considered “wanted,” since it discourages overeating or eating too quickly. My clinical pearl is to tell patients taking liraglutide that they are “trapped” and have to eat smaller portions and eat more slowly or they will be more prone to GI effects. With this strategy, we can encourage portion control and responsibility for behavior. (Please note that this is my experience with the diabetic dosage of liraglutide; I do not have any clinical experience with the obesity dosage, which was not clinically available at the time of writing.)
Both branded versions of liraglutide carry a black-box warning for thyroid C-cell tumors, which were observed in rodents but unproven in humans. The medication is contraindicated in patients with medullary thyroid cancer or with multiple endocrine neoplasia 2 syndrome. Increased rates of acute pancreatitis, cholecystitis, and cholelithiasis were seen in studies, and caution is advised.
Continue for A Word About Meds That Cause Weight Gain >>
A WORD ABOUT MEDS THAT CAUSE WEIGHT GAIN
The Endocrine Society has published a list of medications that can influence weight gain, along with suggestions for alternative agents that are either weight neutral or promote weight loss.
Note that our case patient, Deb, is taking insulin, a sulfonylurea (glyburide), and thiazolidinedione (pioglitazone) for diabetes—all of which can promote weight gain. Guidelines suggest choosing metformin, DPP4 inhibitors, GLP1 agonists, amylin analog, and SGLT2 inhibitors instead when weight gain is a major concern.1
Guidelines also suggest using ACE inhibitors, angiotensin receptor blockers, and calcium channel blockers instead of β-blockers as firstline antihypertensive therapy for diabetic patients.1 Adequate blood pressure and lipid control are imperative in diabetes management.
CASE POINT Deb would need better hypertension control before she considers weight-loss medication. Since she is also taking paroxetine, which among SSRIs is associated with greatest weight gain, a changed to fluoxetine or sertraline should be considered.2
Next page: Conclusion >>
CONCLUSION
There are now five medications approved by the FDA for chronic weight loss, with more to come. Agents with different mechanisms of action give us options to help obese patients and hopefully reduce and prevent obesity-related complications. It is important for clinicians to be competent in managing obesity, especially since we live in an era in which the disease is considered pandemic.
REFERENCES
1. Apovian CM, Aronne LJ, Bessesen DH, et al; Endocrine Society. Pharmacological management of obesity: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2015;100(2):342-362.
2. Xenical [package insert]. South San Francisco, CA: Genentech USA, Inc; 2012.
3. Fujioka K. Safety and tolerability of medications approved for chronic weight management. Obesity (Silver Spring). 2015;22 (suppl 1):S7-S11.
4. Belviq [package insert]. Woodcliff Lake, NJ: Eisai Inc; 2015.
5. Qsymia [package insert]. Mountain View, CA: Vivus, Inc; 2013.
6. Contrave [package insert]. Deerfield, IL: Takeda USA, Inc; 2014.
7. Saxenda [package insert]. Plainsboro, NJ: Novo Nordisk; 2014.
In June 2013, the American Medical Association classified obesity as a disease. Since then, several medical societies have published guidelines to help clinicians improve care of affected patients. One avenue is, of course, pharmacologic treatment.
Until recently, there was only one FDA-approved medication for chronic weight loss on the market: orlistat, which was approved in 1999. (Phentermine and diethylpropion are only indicated for short-term use). After a long hiatus, the FDA approved two additional agents (phentermine/topiramate and lorcaserin)in 2012 and another two (liraglutide and bupropion/naltrexone) in 2014.
While clinicians appreciate having options for managing their patients’ conditions, in this case, many are overwhelmed by the choices. Most health care providers have not received formal training in obesity management. This column will attempt to fill the information gap in terms of what agents are available and what factors should be assessed before prescribing any of them.
Proviso: Experts claim obesity is a chronic disease, similar to hypertension, and should be managed as such. Although not discussed here, the most important aspect of weight loss and maintenance is lifestyle intervention (diet, exercise, and behavioral modification). It should be emphasized that no medication works by itself; all should be used as an adjunct tool to reinforce adherence to lifestyle changes.1 Furthermore, patients may be disappointed to learn that without these changes, the weight may return when they cease medication use.
CASE Deb, age 61, presents to your office for routine follow-up. She has a history of type 2 diabetes, dyslipidemia, hypertension, atrial fibrillation, depression, and chronic back pain due to a herniated disc. Her medications include insulin glargine, glyburide, pioglitazone, atorvastatin, metoprolol, paroxetine, and acetaminophen/hydrocodone.
Her vital signs include a blood pressure of 143/91 mm Hg and a pulse of 93 beats/min. She has a BMI of 37 and a waist circumference of 35 in.
Deb, concerned about her weight, would like to discuss weight-loss options. She has tried three different commercial programs; each time, she was able to lose 30 to 50 lb in three to six months but regained the weight once she stopped the program. She reports excessive appetite as the main reason for her rebound weight gain. Her exercise is limited due to her back pain.
She recently tried OTC orlistat but could not tolerate it due to flatulence and fecal urgency. She reports an incident in which she couldn’t reach the bathroom in time.
Continue for Discussion >>
DISCUSSION
The Endocrine Society’s recommended approaches to obesity management include diet, exercise, and behavioral modification for patients with a BMI ≥ 25. The addition of pharmacotherapy can be considered for those with a BMI ≥ 30 or with a BMI ≥ 27 and one or more weight-related comorbidities (eg, diabetes, dyslipidemia, hypertension). This matches the FDA-approved product labeling for chronic weight-loss medications. Bariatric surgery should be considered for patients with a BMI ≥ 40 or with a BMI ≥ 35 and at least one weight-related comorbidity.
Orlistat
Orlistat is available OTC in a 60-mg thrice-daily form. A higher dosage (120 mg tid) is available via prescription. Orlistat decreases fat absorption in the gastrointestinal (GI) tract by inhibiting GI lipase. Average weight loss with orlistat is 3% at first and second year, and, when compared with placebo, 2.4% greater at four years.2
Orlistat should be prescribed with a multivitamin due to decreased absorption of fat-soluble vitamins. It is contraindicated in patients with malabsorption syndrome and gallbladder disease (> 2% incidence3). It can increase cyclosporine exposure, and rare cases of liver failure have been reported. The most common adverse effect is related to steatorrhea. Of the available options, orlistat is the only medication that has no effect on neurohormonal regulation in appetite control and metabolic rate, which may be a limiting factor.
CASE POINT Due to Deb’s intolerance of and embarrassment with GI adverse effects, she requests an alternative medication.
Lorcaserin
Lorcaserin is a selective serotonin 2C receptor agonist that reduces appetite by affecting anorexigenic pro-opiomelanocortin (POMC) neurons in the hypothalamus. Of note, lorcaserin “selects” the 2C receptor instead of 2A and 2B; 2B receptors are found in both aortic and mitral valves, which may explain the association between fenfluramine/phentermine (commonly known as “fen/phen” and withdrawn from the market in 1997) and possible cardiac valvulopathy. (Fenfluramine is an amphetamine derivative that nonselectively stimulates serotonin release and inhibits reuptake.)
Lorcaserin comes in a 10-mg twice-daily dosage. In studies, patients taking lorcaserin had an average weight loss of 3.3% more than those taking placebo at one year; weight loss was maintained through the second year for those who continued on medication. However, those who stopped the medication at one year had regained their weight by the two-year mark.4
It is recommended that the medication be discontinued if patients don’t achieve a loss of more than 5% of body weight by 12 weeks.
In a study that enrolled diabetic patients, lorcaserin also demonstrated a 0.9% reduction in A1C, which is similar to or even better than some oral antidiabetic medications.4 However, since the manufacturer was not planning for an antidiabetic indication, A1C was only a secondary endpoint. The reduction is most likely due to decreased caloric intake and weight loss.
The most common adverse effects of lorcaserin include headache, dizziness, and fatigue. The discontinuation rate due to intolerance was 8.6%, compared to 6.7% with placebo.5
Although this was not observed in clinical studies, co-administration of lorcaserin (a serotonin receptor agonist) with other serotonergic or antidopaminergic agents can theoretically cause serotonin syndrome or neuroleptic malignant syndrome–like reactions. Caution is therefore advisable when prescribing these agents. The package insert carries a warning for cardiac valvulopathy due to fen/phen’s history and a lack of long-term cardiovascular safety data.
CASE POINT Deb is taking paroxetine (an SSRI) for her depression. Since you are concerned about serotonin syndrome, you decide to keep exploring options. Checking the package insert for phentermine/topiramate, you learn that it does not have a potential adverse reaction related to co-administration with SSRIs.
Phentermine/Topiramate
Phentermine, a sympathomimetic medication, was approved for short-term (12-week) use for weight loss in the 1960s. Topiramate, an antiseizure and migraine prophylactic medication, enhances appetite suppression—although the exact mechanism of action is unknown.1
Four once-daily doses are available: 3.75/23 mg, 7.5/46 mg, 11.25/69 mg, and 15/92 mg. Dosing starts with 3.75/23 mg for two weeks, then increases to 7.5/46 mg. If a loss of 5% or more of body weight is achieved, the patient can continue the dosage; if not, it can be increased to 11.25/69 mg for two weeks and then to 15/92 mg. The average weight loss for mid and maximum dose was 6.6% and 8.6% greater than placebo at one year.5
Commonly reported adverse effects include paraesthesia, dysgeusia (distortion of sense of taste), dizziness, insomnia, constipation, and dry mouth. Due to phentermine’s sympathomimetic action, mild increases in heart rate and blood pressure were reported. The Endocrine Society recommends against the use of phentermine in patients with uncontrolled hypertension and a history of heart disease.1
Weight loss is generally not recommended during pregnancy, and all weight loss medications are classified as category X for pregnancy. Strict caution is advised with this particular agent, as topiramate has known teratogenicity and therefore comes with a Risk Evaluation Mitigation Strategy. Patients must be advised to use appropriate contraception while taking topiramate, and a pregnancy test should be performed before medication commencement and monthly thereafter.
Abrupt cessation of topiramate can cause seizure. When taking the 15/92-mg dosage, the patient should reduce to one tablet every other day for at least one week before discontinuation.
CASE POINT Deb’s blood pressure is still not at goal. This, along with her history of atrial fibrillation and high pulse, prompts you to consider another option.
Bupropion/Naltrexone
Bupropion, a widely used antidepressant, inhibits the uptake of norepinephrine and dopamine and thereby blocks the reward pathway that various foods can induce. Naltrexone, an opioid antagonist, blocks the opioid pathway and can be helpful in enhancing weight loss.
This combination comes in an 8/90-mg tablet. The suggested titration regimen is to start with one tablet per day and increase by one tablet every week, up to the maximum dosage of two tablets twice a day. Average weight loss was 3.1% greater than placebo at one year with the maximum dosage. An A1C reduction of 0.6% was seen in diabetic patients.6 It is recommended to stop the medication and seek an alternative treatment option if > 5% loss of body weight is not achieved by 12 weeks.
GI adverse effects (eg, nausea and vomiting) are common; these can be reduced with a slower titration regimen or by prescribing a maximum of one tablet twice daily (instead of two). Every antidepressant carries suicidal risk, and caution is advised with their use. Bupropion can also lower the seizure threshold, and it is contraindicated for patients with seizure disorder. It is also contraindicated in patients who are undergoing abrupt cessation of alcohol, benzodiazepines, or barbiturates. It can increase pulse and blood pressure during early titration; regular blood pressure monitoring is warranted.
CASE POINT Due to Deb’s opioid usage and uncontrolled hypertension, you discuss a final option that was recently approved for weight loss.
Liraglutide
This glucagon-like peptide-1 (GLP1) receptor agonist affects the brain to suppress/control appetite, slows down gastric emptying, and induces early satiety. A 3-mg dosage was approved in December 2014, but 0.6-, 1.2-, and 1.8-mg dosages have been available since 2010 for patients with type 2 diabetes.
Average weight loss was 4.5% greater than placebo at one year.7 If < 4% weight loss is achieved by 16 weeks, consider using an alternative agent.
The most common adverse effect is GI upset, which could be related to the mechanism of action (slower gastric emptying). Although self-reported GI upset was high (39%), the actual discontinuation rate was low (2.9% for nausea, 1.7% for vomiting, and 1.4% for diarrhea).3
This adverse effect could, in certain contexts, be considered “wanted,” since it discourages overeating or eating too quickly. My clinical pearl is to tell patients taking liraglutide that they are “trapped” and have to eat smaller portions and eat more slowly or they will be more prone to GI effects. With this strategy, we can encourage portion control and responsibility for behavior. (Please note that this is my experience with the diabetic dosage of liraglutide; I do not have any clinical experience with the obesity dosage, which was not clinically available at the time of writing.)
Both branded versions of liraglutide carry a black-box warning for thyroid C-cell tumors, which were observed in rodents but unproven in humans. The medication is contraindicated in patients with medullary thyroid cancer or with multiple endocrine neoplasia 2 syndrome. Increased rates of acute pancreatitis, cholecystitis, and cholelithiasis were seen in studies, and caution is advised.
Continue for A Word About Meds That Cause Weight Gain >>
A WORD ABOUT MEDS THAT CAUSE WEIGHT GAIN
The Endocrine Society has published a list of medications that can influence weight gain, along with suggestions for alternative agents that are either weight neutral or promote weight loss.
Note that our case patient, Deb, is taking insulin, a sulfonylurea (glyburide), and thiazolidinedione (pioglitazone) for diabetes—all of which can promote weight gain. Guidelines suggest choosing metformin, DPP4 inhibitors, GLP1 agonists, amylin analog, and SGLT2 inhibitors instead when weight gain is a major concern.1
Guidelines also suggest using ACE inhibitors, angiotensin receptor blockers, and calcium channel blockers instead of β-blockers as firstline antihypertensive therapy for diabetic patients.1 Adequate blood pressure and lipid control are imperative in diabetes management.
CASE POINT Deb would need better hypertension control before she considers weight-loss medication. Since she is also taking paroxetine, which among SSRIs is associated with greatest weight gain, a changed to fluoxetine or sertraline should be considered.2
Next page: Conclusion >>
CONCLUSION
There are now five medications approved by the FDA for chronic weight loss, with more to come. Agents with different mechanisms of action give us options to help obese patients and hopefully reduce and prevent obesity-related complications. It is important for clinicians to be competent in managing obesity, especially since we live in an era in which the disease is considered pandemic.
REFERENCES
1. Apovian CM, Aronne LJ, Bessesen DH, et al; Endocrine Society. Pharmacological management of obesity: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2015;100(2):342-362.
2. Xenical [package insert]. South San Francisco, CA: Genentech USA, Inc; 2012.
3. Fujioka K. Safety and tolerability of medications approved for chronic weight management. Obesity (Silver Spring). 2015;22 (suppl 1):S7-S11.
4. Belviq [package insert]. Woodcliff Lake, NJ: Eisai Inc; 2015.
5. Qsymia [package insert]. Mountain View, CA: Vivus, Inc; 2013.
6. Contrave [package insert]. Deerfield, IL: Takeda USA, Inc; 2014.
7. Saxenda [package insert]. Plainsboro, NJ: Novo Nordisk; 2014.
No Man Is an Island in the Public Health Service
Below is an edited and condensed version of the Federal Practitioner interview with Jeffrey W. Sherman, DO, chief medical officer of the Department of Homeland Security’s Transportation Security Administration. Dr. Sherman recently received the Outstanding Service Medal from PHS. To hear the complete interview, visit: http://www.fedprac.com/multimedia/multimedia-library.html.
The Transportation Security Administration
Jeffrey W. Sherman, DO. I’m primarily responsible for providing expert opinion to the senior leadership of the Transportation Security Administration (TSA) as it relates to occupational health, preventive medicine, and other medical topics. The position of chief medical officer at the TSA has been on the books for quite a while. However, it hasn’t been filled on a permanent basis until the time that I came over from the Office of Health Affairs…. For the most part, the marching orders that I was given from the senior leadership was to take a fairly neutral look at the agency’s ability to manage the health and wellness of its workforce and find both positive areas and areas for improvement where the TSA could impact the welfare of the TSA population.
Related: Committed to Showing Results at the VA
Programs that we were able to identify that were already in existence and working really well for the TSA were in abundance. However, there were a number of programs that I found would probably enhance the TSA’ s ability to manage its workforce better and to provide a more comfortable, safe workplace. The main one was an ability to measure the transportation security officers’ medical capability and aptitude over a period of time, so more or less to manage them periodically and allow us to review the requirements and assessments for that particular workforce from an occupational health standpoint.
Having a Career in the PHS
Dr. Sherman. My previous work with the PHS included being a director for a number of different medical programs in other government agencies, including Immigration and Customs Enforcement (ICE), where I was acting director of the Division of Immigration Health Services for a period of time. I moved on to be the senior medical advisor to the principal legal advisor at ICE; and again, working with the attorneys on various medical issues allowed me to build greater confidence in my ability to manage the interface between what’s appropriate legally from the medical standpoint and what’s appropriate from the clinical practice of medicine in occupational health. So that was very vital and important.
Just prior to coming to the TSA, I was the medical director for a number of programs for the U.S. Marshals Services. Working alongside law enforcement in their unique roles and managing programs that are variously clinical and nonclinical gives good insight as to how to come into a large organization such as the TSA, with more than 48,000 transportation security officers, and put in place programs in a preexisting organization. To retrofit programs into an organization of that size requires some tact and ability. So all that time previously spent has allowed me to gain those skills.
Joining the PHS
Dr. Sherman. We are a group of dedicated professionals; we have a very close connection and a close network of collegial interactions. I was the Professional Advisory Committee (PAC) chair for the Physicians PAC for a year and vice chair before that. Meeting all the individuals and working with them on various cross-agency public health and professional projects has absolutely brought a lot of their wealth into what I do here at the TSA specifically. No man is an island; and certainly, in the PHS, you never feel that way…
Related: Pharmacist Management of Adult Asthma at an Indian Health Service Facility
I was in private practice in rural upstate New York in 2005 when Hurricane Katrina and Hurricane Rita came ashore, and I was part of the National Health Service Corps at that time. Part of my response as a National Health Service Corps Scholar was to join colleagues—some of whom were PHS officers—down in the recovery area in Louisiana; that is where I met my first uniformed Commissioned Corps officer, 2 of them, in fact. I worked with them for several weeks and was so incredibly impressed with the work of the PHS that while I was there, I applied for my commission. So I was hit pretty hard and pretty heavy, and I haven’t looked back…
It’s, of course, very satisfying to hear your uniformed service spoken highly of in a public forum and especially by elected officials such as the president. I mean it’s difficult not to smile when you hear that. I will say, as one of the 7 uniformed services of the U.S., we do take a lot of knuckling under from our sister services that are more well known. But frankly, at the end of the day, we work beside them regardless of the notoriety or note that we get from them or from anyone else.
I’ve served alongside the Navy on the USNS Mercy, and I’ve been out with Air Force and Coast Guard on their vessels as well. So I’m very comfortable, and I think most of the PHS officers are comfortable working with our sister uniformed services. It is nice to hear the recognition.
The PHS Outstanding Service Medal
Dr. Sherman. It’s a real honor and a privilege to have received that medal…. There are things you do in your professional career that you do not do for notoriety; you do it because it’s the right thing to do, and you know it’s in the greater service to your profession.
In this case, our profession is also a uniformed service. And so for the uniformed service itself to take note and to give recognition for what I would do anyway and the manner in which I do it, it’s again, very satisfying; and it’s very humbling.
Mentoring Junior Officers
Dr. Sherman. The PHS has a very robust program of mentoring junior officers; and I think one of the things that has been most satisfying in my time in uniform, outside of the obvious professional things, has really been mentoring junior officers on their way up through the ranks and as they find their pathway forward in a career as a Commissioned Corps officer. And if I can say one thing to any new officer coming into the organization, it’s make sure that you reach out to your senior officers, and make sure you learn from both their successes and their errors so that by the time you finish your career, you can look back and say you’ve done everything you’ve wanted to.
I still maintain relationships with [my] mentors. I don’t think you’re ever too old or ever too experienced to have a mentor. There’s always something you can learn from another individual. So you know, you never stop learning, and you never stop appreciating the people who you’re working beside who come before you and who are coming behind you. And please don’t ask me to state names, because there are too many.
Below is an edited and condensed version of the Federal Practitioner interview with Jeffrey W. Sherman, DO, chief medical officer of the Department of Homeland Security’s Transportation Security Administration. Dr. Sherman recently received the Outstanding Service Medal from PHS. To hear the complete interview, visit: http://www.fedprac.com/multimedia/multimedia-library.html.
The Transportation Security Administration
Jeffrey W. Sherman, DO. I’m primarily responsible for providing expert opinion to the senior leadership of the Transportation Security Administration (TSA) as it relates to occupational health, preventive medicine, and other medical topics. The position of chief medical officer at the TSA has been on the books for quite a while. However, it hasn’t been filled on a permanent basis until the time that I came over from the Office of Health Affairs…. For the most part, the marching orders that I was given from the senior leadership was to take a fairly neutral look at the agency’s ability to manage the health and wellness of its workforce and find both positive areas and areas for improvement where the TSA could impact the welfare of the TSA population.
Related: Committed to Showing Results at the VA
Programs that we were able to identify that were already in existence and working really well for the TSA were in abundance. However, there were a number of programs that I found would probably enhance the TSA’ s ability to manage its workforce better and to provide a more comfortable, safe workplace. The main one was an ability to measure the transportation security officers’ medical capability and aptitude over a period of time, so more or less to manage them periodically and allow us to review the requirements and assessments for that particular workforce from an occupational health standpoint.
Having a Career in the PHS
Dr. Sherman. My previous work with the PHS included being a director for a number of different medical programs in other government agencies, including Immigration and Customs Enforcement (ICE), where I was acting director of the Division of Immigration Health Services for a period of time. I moved on to be the senior medical advisor to the principal legal advisor at ICE; and again, working with the attorneys on various medical issues allowed me to build greater confidence in my ability to manage the interface between what’s appropriate legally from the medical standpoint and what’s appropriate from the clinical practice of medicine in occupational health. So that was very vital and important.
Just prior to coming to the TSA, I was the medical director for a number of programs for the U.S. Marshals Services. Working alongside law enforcement in their unique roles and managing programs that are variously clinical and nonclinical gives good insight as to how to come into a large organization such as the TSA, with more than 48,000 transportation security officers, and put in place programs in a preexisting organization. To retrofit programs into an organization of that size requires some tact and ability. So all that time previously spent has allowed me to gain those skills.
Joining the PHS
Dr. Sherman. We are a group of dedicated professionals; we have a very close connection and a close network of collegial interactions. I was the Professional Advisory Committee (PAC) chair for the Physicians PAC for a year and vice chair before that. Meeting all the individuals and working with them on various cross-agency public health and professional projects has absolutely brought a lot of their wealth into what I do here at the TSA specifically. No man is an island; and certainly, in the PHS, you never feel that way…
Related: Pharmacist Management of Adult Asthma at an Indian Health Service Facility
I was in private practice in rural upstate New York in 2005 when Hurricane Katrina and Hurricane Rita came ashore, and I was part of the National Health Service Corps at that time. Part of my response as a National Health Service Corps Scholar was to join colleagues—some of whom were PHS officers—down in the recovery area in Louisiana; that is where I met my first uniformed Commissioned Corps officer, 2 of them, in fact. I worked with them for several weeks and was so incredibly impressed with the work of the PHS that while I was there, I applied for my commission. So I was hit pretty hard and pretty heavy, and I haven’t looked back…
It’s, of course, very satisfying to hear your uniformed service spoken highly of in a public forum and especially by elected officials such as the president. I mean it’s difficult not to smile when you hear that. I will say, as one of the 7 uniformed services of the U.S., we do take a lot of knuckling under from our sister services that are more well known. But frankly, at the end of the day, we work beside them regardless of the notoriety or note that we get from them or from anyone else.
I’ve served alongside the Navy on the USNS Mercy, and I’ve been out with Air Force and Coast Guard on their vessels as well. So I’m very comfortable, and I think most of the PHS officers are comfortable working with our sister uniformed services. It is nice to hear the recognition.
The PHS Outstanding Service Medal
Dr. Sherman. It’s a real honor and a privilege to have received that medal…. There are things you do in your professional career that you do not do for notoriety; you do it because it’s the right thing to do, and you know it’s in the greater service to your profession.
In this case, our profession is also a uniformed service. And so for the uniformed service itself to take note and to give recognition for what I would do anyway and the manner in which I do it, it’s again, very satisfying; and it’s very humbling.
Mentoring Junior Officers
Dr. Sherman. The PHS has a very robust program of mentoring junior officers; and I think one of the things that has been most satisfying in my time in uniform, outside of the obvious professional things, has really been mentoring junior officers on their way up through the ranks and as they find their pathway forward in a career as a Commissioned Corps officer. And if I can say one thing to any new officer coming into the organization, it’s make sure that you reach out to your senior officers, and make sure you learn from both their successes and their errors so that by the time you finish your career, you can look back and say you’ve done everything you’ve wanted to.
I still maintain relationships with [my] mentors. I don’t think you’re ever too old or ever too experienced to have a mentor. There’s always something you can learn from another individual. So you know, you never stop learning, and you never stop appreciating the people who you’re working beside who come before you and who are coming behind you. And please don’t ask me to state names, because there are too many.
Below is an edited and condensed version of the Federal Practitioner interview with Jeffrey W. Sherman, DO, chief medical officer of the Department of Homeland Security’s Transportation Security Administration. Dr. Sherman recently received the Outstanding Service Medal from PHS. To hear the complete interview, visit: http://www.fedprac.com/multimedia/multimedia-library.html.
The Transportation Security Administration
Jeffrey W. Sherman, DO. I’m primarily responsible for providing expert opinion to the senior leadership of the Transportation Security Administration (TSA) as it relates to occupational health, preventive medicine, and other medical topics. The position of chief medical officer at the TSA has been on the books for quite a while. However, it hasn’t been filled on a permanent basis until the time that I came over from the Office of Health Affairs…. For the most part, the marching orders that I was given from the senior leadership was to take a fairly neutral look at the agency’s ability to manage the health and wellness of its workforce and find both positive areas and areas for improvement where the TSA could impact the welfare of the TSA population.
Related: Committed to Showing Results at the VA
Programs that we were able to identify that were already in existence and working really well for the TSA were in abundance. However, there were a number of programs that I found would probably enhance the TSA’ s ability to manage its workforce better and to provide a more comfortable, safe workplace. The main one was an ability to measure the transportation security officers’ medical capability and aptitude over a period of time, so more or less to manage them periodically and allow us to review the requirements and assessments for that particular workforce from an occupational health standpoint.
Having a Career in the PHS
Dr. Sherman. My previous work with the PHS included being a director for a number of different medical programs in other government agencies, including Immigration and Customs Enforcement (ICE), where I was acting director of the Division of Immigration Health Services for a period of time. I moved on to be the senior medical advisor to the principal legal advisor at ICE; and again, working with the attorneys on various medical issues allowed me to build greater confidence in my ability to manage the interface between what’s appropriate legally from the medical standpoint and what’s appropriate from the clinical practice of medicine in occupational health. So that was very vital and important.
Just prior to coming to the TSA, I was the medical director for a number of programs for the U.S. Marshals Services. Working alongside law enforcement in their unique roles and managing programs that are variously clinical and nonclinical gives good insight as to how to come into a large organization such as the TSA, with more than 48,000 transportation security officers, and put in place programs in a preexisting organization. To retrofit programs into an organization of that size requires some tact and ability. So all that time previously spent has allowed me to gain those skills.
Joining the PHS
Dr. Sherman. We are a group of dedicated professionals; we have a very close connection and a close network of collegial interactions. I was the Professional Advisory Committee (PAC) chair for the Physicians PAC for a year and vice chair before that. Meeting all the individuals and working with them on various cross-agency public health and professional projects has absolutely brought a lot of their wealth into what I do here at the TSA specifically. No man is an island; and certainly, in the PHS, you never feel that way…
Related: Pharmacist Management of Adult Asthma at an Indian Health Service Facility
I was in private practice in rural upstate New York in 2005 when Hurricane Katrina and Hurricane Rita came ashore, and I was part of the National Health Service Corps at that time. Part of my response as a National Health Service Corps Scholar was to join colleagues—some of whom were PHS officers—down in the recovery area in Louisiana; that is where I met my first uniformed Commissioned Corps officer, 2 of them, in fact. I worked with them for several weeks and was so incredibly impressed with the work of the PHS that while I was there, I applied for my commission. So I was hit pretty hard and pretty heavy, and I haven’t looked back…
It’s, of course, very satisfying to hear your uniformed service spoken highly of in a public forum and especially by elected officials such as the president. I mean it’s difficult not to smile when you hear that. I will say, as one of the 7 uniformed services of the U.S., we do take a lot of knuckling under from our sister services that are more well known. But frankly, at the end of the day, we work beside them regardless of the notoriety or note that we get from them or from anyone else.
I’ve served alongside the Navy on the USNS Mercy, and I’ve been out with Air Force and Coast Guard on their vessels as well. So I’m very comfortable, and I think most of the PHS officers are comfortable working with our sister uniformed services. It is nice to hear the recognition.
The PHS Outstanding Service Medal
Dr. Sherman. It’s a real honor and a privilege to have received that medal…. There are things you do in your professional career that you do not do for notoriety; you do it because it’s the right thing to do, and you know it’s in the greater service to your profession.
In this case, our profession is also a uniformed service. And so for the uniformed service itself to take note and to give recognition for what I would do anyway and the manner in which I do it, it’s again, very satisfying; and it’s very humbling.
Mentoring Junior Officers
Dr. Sherman. The PHS has a very robust program of mentoring junior officers; and I think one of the things that has been most satisfying in my time in uniform, outside of the obvious professional things, has really been mentoring junior officers on their way up through the ranks and as they find their pathway forward in a career as a Commissioned Corps officer. And if I can say one thing to any new officer coming into the organization, it’s make sure that you reach out to your senior officers, and make sure you learn from both their successes and their errors so that by the time you finish your career, you can look back and say you’ve done everything you’ve wanted to.
I still maintain relationships with [my] mentors. I don’t think you’re ever too old or ever too experienced to have a mentor. There’s always something you can learn from another individual. So you know, you never stop learning, and you never stop appreciating the people who you’re working beside who come before you and who are coming behind you. And please don’t ask me to state names, because there are too many.
Acute Kidney Injury: Prevalent in Sugarcane Harvesters
Q) I’ve heard a lot of talk about all the kidney problems that the sugarcane workers in Central America have. Does anyone know why this is happening?
The unusually high rates of chronic kidney disease (CKD) among sugarcane workers in Central America have been a subject of great interest since National Public Radio (NPR) aired a special on this topic.3 There has been a rising epidemic of CKD in otherwise healthy male farm workers (ages 20 to 50), particularly those who harvest sugarcane.4,5 It has been hypothesized that recurrent episodes of acute kidney injury (AKI)—related to dehydration, volume depletion, pollutants, and rhabdomyolysis with inflammatory stress—are the underlying cause.5
Sugarcane harvesters typically work nine-hour days, six days per week, in extremely high temperatures and while wearing heavy, hot clothing. Each worker cuts approximately 10 tons of sugarcane daily, since they are paid based on cutting volume. Workers drink between five and 10 L of water during their shifts.
Santos et al designed a study to prospectively examine the effects of burnt sugarcane harvesting on renal function in healthy male farm workers. Twenty-eight men (ages 19 to 39) with no CKD risk factors (diabetes, smoking, obesity, hypertension, illicit drug or alcohol use) were followed for eight months from preharvest to postharvest. Blood samples were collected at the beginning and at the end of the workday and preharvest and postharvest season.5
Preseason lab values were normal in all 28 men. But postseason, all workers had elevated creatinine levels, with five meeting the criteria for AKI (see Table at left).5,6
Santos and colleagues identified potential causes for AKI in this population. These included
• Dehydration and volume depletion (episodes of tachycardia, increased urine density, lower urinary/serum sodium, higher hematocrit)
• Rhabdomyolysis (increased creatine kinase at the end of each workday)
• Systemic inflammation (increased white blood count, neutrophils, lymphocytes, and monocytes during the workday—possibly indicative of an inflammatory burst)
• Other factors (burning of the sugarcane releasing unknown nephrotoxic substances; unreported NSAIDs use)5
Compared to workers who showed early signs of CKD, those who developed frank AKI were more likely to have hyponatremia. Recommendations to reduce the problem include consumption of water/salt hydrating drinks, use of appropriate clothing, work-hour limitations, and changes to payment structures (ie, from a volume system to an hourly or daily system). Furthermore, education on the need to avoid alcohol, illicit drugs, and NSAIDs during the harvest season should help to decrease incidence of AKI among these workers.
Elizabeth C. Evans, RN, MSN, CNP, DNP
Renal Medicine Associates, Albuquerque, New Mexico
REFERENCES
1. Ayuk J, Gittoes N. Contemporary view of the clinical relevance of magnesium homeostasis. Ann Clin Biochem. 2014;51(Pt 2):179-188.
2. Firouzi A, Maadani M, Kiani R, et al. Intravenous magnesium sulfate: new method in prevention of contrast-induced nephropathy in primary percutaneous coronary intervention. Int Urol Nephrol. 2015;47(3):521-525.
3. Beaubien J. Mysterious kidney disease slays farm workers in central America. National Public Radio; 2014. www.npr.org/blogs/health/2014/04/30/306907097/mysterious-kidney-disease-slays-farmworkers-in-central-america. Accessed April 1, 2015.
4. Almaguer M, Herrera R, Orantes CM. Chronic kidney disease of unknown etiology in agricultural communities. MEDICC Rev. 2014;16(2):9-15.
5. Santos UP, Zanetta DMT, Burdmann EA. Burnt sugarcane harvesting is associated with acute renal dysfunction. Kidney Int. 2015;87(4):792-799.
6. Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Int Suppl. 2012;2:1-138.
Q) I’ve heard a lot of talk about all the kidney problems that the sugarcane workers in Central America have. Does anyone know why this is happening?
The unusually high rates of chronic kidney disease (CKD) among sugarcane workers in Central America have been a subject of great interest since National Public Radio (NPR) aired a special on this topic.3 There has been a rising epidemic of CKD in otherwise healthy male farm workers (ages 20 to 50), particularly those who harvest sugarcane.4,5 It has been hypothesized that recurrent episodes of acute kidney injury (AKI)—related to dehydration, volume depletion, pollutants, and rhabdomyolysis with inflammatory stress—are the underlying cause.5
Sugarcane harvesters typically work nine-hour days, six days per week, in extremely high temperatures and while wearing heavy, hot clothing. Each worker cuts approximately 10 tons of sugarcane daily, since they are paid based on cutting volume. Workers drink between five and 10 L of water during their shifts.
Santos et al designed a study to prospectively examine the effects of burnt sugarcane harvesting on renal function in healthy male farm workers. Twenty-eight men (ages 19 to 39) with no CKD risk factors (diabetes, smoking, obesity, hypertension, illicit drug or alcohol use) were followed for eight months from preharvest to postharvest. Blood samples were collected at the beginning and at the end of the workday and preharvest and postharvest season.5
Preseason lab values were normal in all 28 men. But postseason, all workers had elevated creatinine levels, with five meeting the criteria for AKI (see Table at left).5,6
Santos and colleagues identified potential causes for AKI in this population. These included
• Dehydration and volume depletion (episodes of tachycardia, increased urine density, lower urinary/serum sodium, higher hematocrit)
• Rhabdomyolysis (increased creatine kinase at the end of each workday)
• Systemic inflammation (increased white blood count, neutrophils, lymphocytes, and monocytes during the workday—possibly indicative of an inflammatory burst)
• Other factors (burning of the sugarcane releasing unknown nephrotoxic substances; unreported NSAIDs use)5
Compared to workers who showed early signs of CKD, those who developed frank AKI were more likely to have hyponatremia. Recommendations to reduce the problem include consumption of water/salt hydrating drinks, use of appropriate clothing, work-hour limitations, and changes to payment structures (ie, from a volume system to an hourly or daily system). Furthermore, education on the need to avoid alcohol, illicit drugs, and NSAIDs during the harvest season should help to decrease incidence of AKI among these workers.
Elizabeth C. Evans, RN, MSN, CNP, DNP
Renal Medicine Associates, Albuquerque, New Mexico
REFERENCES
1. Ayuk J, Gittoes N. Contemporary view of the clinical relevance of magnesium homeostasis. Ann Clin Biochem. 2014;51(Pt 2):179-188.
2. Firouzi A, Maadani M, Kiani R, et al. Intravenous magnesium sulfate: new method in prevention of contrast-induced nephropathy in primary percutaneous coronary intervention. Int Urol Nephrol. 2015;47(3):521-525.
3. Beaubien J. Mysterious kidney disease slays farm workers in central America. National Public Radio; 2014. www.npr.org/blogs/health/2014/04/30/306907097/mysterious-kidney-disease-slays-farmworkers-in-central-america. Accessed April 1, 2015.
4. Almaguer M, Herrera R, Orantes CM. Chronic kidney disease of unknown etiology in agricultural communities. MEDICC Rev. 2014;16(2):9-15.
5. Santos UP, Zanetta DMT, Burdmann EA. Burnt sugarcane harvesting is associated with acute renal dysfunction. Kidney Int. 2015;87(4):792-799.
6. Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Int Suppl. 2012;2:1-138.
Q) I’ve heard a lot of talk about all the kidney problems that the sugarcane workers in Central America have. Does anyone know why this is happening?
The unusually high rates of chronic kidney disease (CKD) among sugarcane workers in Central America have been a subject of great interest since National Public Radio (NPR) aired a special on this topic.3 There has been a rising epidemic of CKD in otherwise healthy male farm workers (ages 20 to 50), particularly those who harvest sugarcane.4,5 It has been hypothesized that recurrent episodes of acute kidney injury (AKI)—related to dehydration, volume depletion, pollutants, and rhabdomyolysis with inflammatory stress—are the underlying cause.5
Sugarcane harvesters typically work nine-hour days, six days per week, in extremely high temperatures and while wearing heavy, hot clothing. Each worker cuts approximately 10 tons of sugarcane daily, since they are paid based on cutting volume. Workers drink between five and 10 L of water during their shifts.
Santos et al designed a study to prospectively examine the effects of burnt sugarcane harvesting on renal function in healthy male farm workers. Twenty-eight men (ages 19 to 39) with no CKD risk factors (diabetes, smoking, obesity, hypertension, illicit drug or alcohol use) were followed for eight months from preharvest to postharvest. Blood samples were collected at the beginning and at the end of the workday and preharvest and postharvest season.5
Preseason lab values were normal in all 28 men. But postseason, all workers had elevated creatinine levels, with five meeting the criteria for AKI (see Table at left).5,6
Santos and colleagues identified potential causes for AKI in this population. These included
• Dehydration and volume depletion (episodes of tachycardia, increased urine density, lower urinary/serum sodium, higher hematocrit)
• Rhabdomyolysis (increased creatine kinase at the end of each workday)
• Systemic inflammation (increased white blood count, neutrophils, lymphocytes, and monocytes during the workday—possibly indicative of an inflammatory burst)
• Other factors (burning of the sugarcane releasing unknown nephrotoxic substances; unreported NSAIDs use)5
Compared to workers who showed early signs of CKD, those who developed frank AKI were more likely to have hyponatremia. Recommendations to reduce the problem include consumption of water/salt hydrating drinks, use of appropriate clothing, work-hour limitations, and changes to payment structures (ie, from a volume system to an hourly or daily system). Furthermore, education on the need to avoid alcohol, illicit drugs, and NSAIDs during the harvest season should help to decrease incidence of AKI among these workers.
Elizabeth C. Evans, RN, MSN, CNP, DNP
Renal Medicine Associates, Albuquerque, New Mexico
REFERENCES
1. Ayuk J, Gittoes N. Contemporary view of the clinical relevance of magnesium homeostasis. Ann Clin Biochem. 2014;51(Pt 2):179-188.
2. Firouzi A, Maadani M, Kiani R, et al. Intravenous magnesium sulfate: new method in prevention of contrast-induced nephropathy in primary percutaneous coronary intervention. Int Urol Nephrol. 2015;47(3):521-525.
3. Beaubien J. Mysterious kidney disease slays farm workers in central America. National Public Radio; 2014. www.npr.org/blogs/health/2014/04/30/306907097/mysterious-kidney-disease-slays-farmworkers-in-central-america. Accessed April 1, 2015.
4. Almaguer M, Herrera R, Orantes CM. Chronic kidney disease of unknown etiology in agricultural communities. MEDICC Rev. 2014;16(2):9-15.
5. Santos UP, Zanetta DMT, Burdmann EA. Burnt sugarcane harvesting is associated with acute renal dysfunction. Kidney Int. 2015;87(4):792-799.
6. Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Int Suppl. 2012;2:1-138.
Acute Kidney Injury: Magnesium for Protection
Q) Our radiology department is discussing use of IV magnesium for diabetic patients to “protect them from kidney injury.” Is this a standard of care now?
Magnesium, the fourth most abundant cation in the body, plays an important physiologic role. Balance is maintained by renal regulation of magnesium reabsorption, and deficiency occurs when there is increased renal excretion initiated by osmotic diuresis. Clinical manifestations of deficiency include cardiac arrhythmias, neuromuscular hyperexcitability, and biochemical abnormalities of hypocalcaemia and hypokalemia.
Diabetes is one of the leading causes of magnesium deficiency, with incidence ranging from 25% to 39%.1 Fluctuations in serum magnesium concentrations are directly correlated with fasting blood glucose, A1C levels, albumin excretion, and the duration of diabetes. It has been postulated that magnesium depletion, via its effect on inositol transport, is pathogenic in the progression of diabetic complications.
Contrast-induced acute kidney injury (CI-AKI) is a potentially adverse consequence of percutaneous coronary interventions (PCI), particularly in diabetic patients. It results in significant morbidity and mortality and adds to the costs of diagnostic and interventional cardiology procedures. Intravenous (IV) agents used during radiologic imaging are notorious for causing acute kidney injury in diabetic patients. Preprocedural hydration and discontinuation of all nephrotoxic medications have proven beneficial in protecting these patients from CI-AKI.
A recent prospective, randomized, open-label clinical trial looked at the effect of administering IV magnesium prior to PCI.2 The control group underwent standard preprocedural hydration and discontinuation of nephrotoxic medications. The study group added IV magnesium to the standard protocol.
In this single-center study, 26.6% of patients in the control group and 14.5% in the study group sustained CI-AKI, a statistically significant result (P = .01). Neither group experienced mortality or required dialysis.
Although not considered standard of care at this time, prophylactic use of IV magnesium (pending pre-op labs), along with the recognized benefit of preprocedural hydration and discontinuation of nephrotoxic medications, can be supported in primary PCI patients. Your radiology department is on the cutting edge of protecting these very high-risk patients.
Debra L. Coplon, DNP, DCC
City of Memphis Wellness Clinic, Tennessee
REFERENCES
1. Ayuk J, Gittoes N. Contemporary view of the clinical relevance of magnesium homeostasis. Ann Clin Biochem. 2014;51(Pt 2):179-188.
2. Firouzi A, Maadani M, Kiani R, et al. Intravenous magnesium sulfate: new method in prevention of contrast-induced nephropathy in primary percutaneous coronary intervention. Int Urol Nephrol. 2015;47(3):521-525.
Q) Our radiology department is discussing use of IV magnesium for diabetic patients to “protect them from kidney injury.” Is this a standard of care now?
Magnesium, the fourth most abundant cation in the body, plays an important physiologic role. Balance is maintained by renal regulation of magnesium reabsorption, and deficiency occurs when there is increased renal excretion initiated by osmotic diuresis. Clinical manifestations of deficiency include cardiac arrhythmias, neuromuscular hyperexcitability, and biochemical abnormalities of hypocalcaemia and hypokalemia.
Diabetes is one of the leading causes of magnesium deficiency, with incidence ranging from 25% to 39%.1 Fluctuations in serum magnesium concentrations are directly correlated with fasting blood glucose, A1C levels, albumin excretion, and the duration of diabetes. It has been postulated that magnesium depletion, via its effect on inositol transport, is pathogenic in the progression of diabetic complications.
Contrast-induced acute kidney injury (CI-AKI) is a potentially adverse consequence of percutaneous coronary interventions (PCI), particularly in diabetic patients. It results in significant morbidity and mortality and adds to the costs of diagnostic and interventional cardiology procedures. Intravenous (IV) agents used during radiologic imaging are notorious for causing acute kidney injury in diabetic patients. Preprocedural hydration and discontinuation of all nephrotoxic medications have proven beneficial in protecting these patients from CI-AKI.
A recent prospective, randomized, open-label clinical trial looked at the effect of administering IV magnesium prior to PCI.2 The control group underwent standard preprocedural hydration and discontinuation of nephrotoxic medications. The study group added IV magnesium to the standard protocol.
In this single-center study, 26.6% of patients in the control group and 14.5% in the study group sustained CI-AKI, a statistically significant result (P = .01). Neither group experienced mortality or required dialysis.
Although not considered standard of care at this time, prophylactic use of IV magnesium (pending pre-op labs), along with the recognized benefit of preprocedural hydration and discontinuation of nephrotoxic medications, can be supported in primary PCI patients. Your radiology department is on the cutting edge of protecting these very high-risk patients.
Debra L. Coplon, DNP, DCC
City of Memphis Wellness Clinic, Tennessee
REFERENCES
1. Ayuk J, Gittoes N. Contemporary view of the clinical relevance of magnesium homeostasis. Ann Clin Biochem. 2014;51(Pt 2):179-188.
2. Firouzi A, Maadani M, Kiani R, et al. Intravenous magnesium sulfate: new method in prevention of contrast-induced nephropathy in primary percutaneous coronary intervention. Int Urol Nephrol. 2015;47(3):521-525.
Q) Our radiology department is discussing use of IV magnesium for diabetic patients to “protect them from kidney injury.” Is this a standard of care now?
Magnesium, the fourth most abundant cation in the body, plays an important physiologic role. Balance is maintained by renal regulation of magnesium reabsorption, and deficiency occurs when there is increased renal excretion initiated by osmotic diuresis. Clinical manifestations of deficiency include cardiac arrhythmias, neuromuscular hyperexcitability, and biochemical abnormalities of hypocalcaemia and hypokalemia.
Diabetes is one of the leading causes of magnesium deficiency, with incidence ranging from 25% to 39%.1 Fluctuations in serum magnesium concentrations are directly correlated with fasting blood glucose, A1C levels, albumin excretion, and the duration of diabetes. It has been postulated that magnesium depletion, via its effect on inositol transport, is pathogenic in the progression of diabetic complications.
Contrast-induced acute kidney injury (CI-AKI) is a potentially adverse consequence of percutaneous coronary interventions (PCI), particularly in diabetic patients. It results in significant morbidity and mortality and adds to the costs of diagnostic and interventional cardiology procedures. Intravenous (IV) agents used during radiologic imaging are notorious for causing acute kidney injury in diabetic patients. Preprocedural hydration and discontinuation of all nephrotoxic medications have proven beneficial in protecting these patients from CI-AKI.
A recent prospective, randomized, open-label clinical trial looked at the effect of administering IV magnesium prior to PCI.2 The control group underwent standard preprocedural hydration and discontinuation of nephrotoxic medications. The study group added IV magnesium to the standard protocol.
In this single-center study, 26.6% of patients in the control group and 14.5% in the study group sustained CI-AKI, a statistically significant result (P = .01). Neither group experienced mortality or required dialysis.
Although not considered standard of care at this time, prophylactic use of IV magnesium (pending pre-op labs), along with the recognized benefit of preprocedural hydration and discontinuation of nephrotoxic medications, can be supported in primary PCI patients. Your radiology department is on the cutting edge of protecting these very high-risk patients.
Debra L. Coplon, DNP, DCC
City of Memphis Wellness Clinic, Tennessee
REFERENCES
1. Ayuk J, Gittoes N. Contemporary view of the clinical relevance of magnesium homeostasis. Ann Clin Biochem. 2014;51(Pt 2):179-188.
2. Firouzi A, Maadani M, Kiani R, et al. Intravenous magnesium sulfate: new method in prevention of contrast-induced nephropathy in primary percutaneous coronary intervention. Int Urol Nephrol. 2015;47(3):521-525.
Managing Thyroid Disease in Pregnancy
Management of thyroid disease during pregnancy presents unique challenges due to physiologic changes that occur. These include
• Serum levels of thyroxine-binding globulin (TBG) increase along with estrogen; in turn, total thyroxine (T4) and triiodothyronine (T3) levels increase.
• Human chorionic gonadotropin (hCG) stimulates the thyroid stimulating hormone (TSH) receptors.1
Since hCG and TSH share similar glycoprotein subunits, a transient suppression of TSH—especially around weeks 10 to 12, when hCG concentrations peak—is considered a physiologic finding. Interpretation of thyroid function testing should be made in relation to the hCG-mediated decrease in serum TSH levels.2
The following four cases will help guide your clinical management of thyroid disease in both preconception and pregnancy. Inadequately controlled thyroid dysfunction can lead to poor pregnancy outcomes for both mother and child, which will be further discussed.
Continue for Case 1: Stable Hypothyroidism >>
CASE 1: STABLE HYPOTHYROIDISM
A 29-year-old woman with stable primary hypothyroidism calls your office to report that she is pregnant. She has taken levothyroxine (100 mg) for the past three years, and her TSH level was 1.21 mIU/L at last measurement. She denies any symptoms of hyperthyroidism or hypothyroidism. What is your next step in her management?
Recommendation
The American Thyroid Association recommends monitoring serum TSH every four weeks during the first half of pregnancy and at least once per trimester thereafter, with frequency depending on symptoms and TSH levels.3 Most women will require higher doses of levothyroxine supplementation to maintain therapeutic TSH levels.
Prior to 18 weeks’ gestation, the fetus is dependent on maternal thyroid hormone. When pregnancy is confirmed, there is support in the literature for having the patient take two additional doses of levothyroxine per week until TSH can be tested.4 However, many endocrinology practices opt to check TSH and total T4 as soon as pregnancy is confirmed.
Since free T4 results may be unreliable during pregnancy (due to the effect of TBG), free thyroxine index (FTI) or total T4 should be monitored instead. FTI mathematically corrects free T4 for TBG levels, making it a useful marker. If total T4 is measured, it is important to remember that results will be approximately 1.5x the nonpregnancy value; thus, the reference range must be multiplied by 1.5 to calculate appropriate high and low parameters for pregnant patients.
Ideally, all women of childbearing age should be encouraged to plan pregnancy, to ensure TSH is at target prior to conception. Maintaining a euthyroid state throughout pregnancy (starting at conception) is important to decrease risk for such adverse outcomes as spontaneous abortion, placental abruption, and gestational hypertension.2 Low birth weight and respiratory distress are potential complications for newborns whose mothers have inadequately controlled hypothyroidism.
Patients should be counseled against simultaneous dosing of prenatal vitamins and levothyroxine. Prenatal vitamins contain iron, which reduces absorption of levothyroxine; therefore, it is recommended that the levothyroxine be taken four hours or more apart from prenatal vitamins.
The Endocrine Society recommends a TSH no higher than 2.5 mIU/L for hypothyroidism diagnosed prior to pregnancy.2,3 After delivery, levothyroxine doses should be reduced to prepregnancy levels, with close monitoring of TSH.2
Next page: Case 2 >>
CASE 2: HISTORY OF SPONTANEOUS ABORTION
A 36-year-old G3P0 woman visits your office for a work-up after her third spontaneous abortion at 16 weeks. The patient denies history of thyroid disease but notes her maternal grandmother has Hashimoto disease. She denies symptoms of hyperthyroidism or hypothyroidism.
Recommendation
Both hyperthyroidism and hypothyroidism are associated with an increase in spontaneous abortion, premature labor, and low birth weight. Negro et al observed an increased risk for fetal loss, small-for-gestational-age fetus, premature delivery, and premature mortality in women who were TPO-antibody-positive, even if they were euthyroid. Improved fetal outcomes occurred when TPO-antibody-positive mothers received supplemental levothyroxine.5
However, the American Thyroid Association and the Endocrine Society state there is currently insufficient evidence to recommend universal screening of thyroid antibodies during pregnancy.2,3 Obtaining thyroid function studies and TPO-antibody tests could be considered as part of a work-up for women who experience multiple spontaneous abortions or have a personal or family history of autoimmune diseases.
Continue for Case 3: Cardiovascular Symptoms >>
CASE 3: CARDIOVASCULAR SYMPTOMS
A 24-year-old primigravida woman presents with complaints of palpitations and increased anxiety. She is currently 28 weeks pregnant. Her TSH level is undetectable (< 0.01 mIU/L), and her free T4 is 2.1 mg/dL (reference range, 0.5-1.6 mg/dL). An ECG performed at your office shows sinus tachycardia with a rate of 127 beats/min.
Recommendation
Maternal hyperthyroidism increases risk for maternal congestive heart failure, uncontrolled hypertension, atrial fibrillation, and thyroid storm. Additionally, fetal hyperthyroidism can occur, especially if the mother has Graves disease. Since thyroid-stimulating immunoglobulins (TSI) can permeate the placental barrier, poor fetal growth, cardiac failure, and fetal thyrotoxicosis are severe adverse effects of in-utero TSI exposure.
To prevent further complications, antithyroid medications should be started in this case. Methimazole (MMI) carries a risk for a rare birth defect, aplasia cutis, in the first trimester and is best avoided during this time. Propylthiouracil (PTU) should be given in the first trimester and then switched to MMI in the second trimester to decrease the risk for hepatotoxicity associated with PTU. Breastfeeding mothers should be assured that low-dose MMI is generally considered safe for breastfed infants but should be taken after feeding in divided doses if possible.1
For symptom relief, b-blockers can be used, although they do come with some risks. As pregnancy Category C drugs, b-blockers are associated with neonatal growth retardation, hypoglycemia, hypoxia, lower Apgar scores, and neonatal respiratory distress.6 Consider giving the lowest dose possible for the duration of the patient’s symptoms.
Radioactive iodine (I-131) should not be given to patients who plan to become pregnant or who are pregnant.2,3 The Endocrine Society recommends that if a woman inadvertently becomes pregnant, she should be counseled on the risks of radiation to the fetus, which include thyroid destruction if treatment occurs/continues after the 12th week of pregnancy.2 Furthermore, pregnancy should be avoided for the first six months after thyroid ablation to allow sufficient time to obtain the target maternal serum TSH level of 0.3 to 2.5 mIU/L.
Next page: Case 4 >>
CASE 4: PRECONCEPTION SCREENING
A 39-year-old G0P0 woman presents for preconception counseling. She denies family or personal history of thyroid disease and symptoms of thyroid disease. Should she be screened?
Recommendation
There is no consensus or guideline regarding preconception laboratory screening for thyroid disease. Current guidelines by the American Thyroid Association, the American College of Obstetricians and Gynecologists, and The Endocrine Society recommend targeted, not universal, screening.2,3,7
The American Thyroid Association and the Endocrine Society recommend screening TSH in women who are pregnant or intend to become pregnant and
• Have a personal or family history of thyroid disease
• Are older than 30
• Demonstrate symptoms of thyroid dysfunction
• Have goiter
• Are TPO-antibody positive
• Have type 1 diabetes or other autoimmune disorders
• Have a history of miscarriage or preterm delivery
• Have a history of head or neck radiation or thyroid surgery
• Are morbidly obese (BMI > 40)
• Use amiodarone or lithium or were exposed to iodinated radiologic contrast
• Are infertile
• Live in an area with moderate to severe iodine insufficiency.2,3
Rationale for targeted screening of asymptomatic women: Large-scale research has not demonstrated significantly better outcomes in those with subclinical hypothyroidism who receive treatment.7 Small studies2 have demonstrated improved fetal outcomes when subclinical hypothyroidism is treated, but for large bodies (eg, the US Preventive Services Task Force) to recommend screening, a clear improvement in health outcomes must be established via controlled studies. Future research should evaluate the effect of treating subclinical hypothyroidism during pregnancy.
REFERENCES
1. Ballabio, M, Poshychinda M, Ekins RP. Pregnancy-induced changes in thyroid function: role of human chorionic gonadotropin as putative regulator of maternal thyroid. J Clin Endocrinol Metab. 1991;73(4):824.
2. The Endocrine Society. Management of thyroid disease in pregnancy and postpartum. J Clin Endocrinol Metab. 2012;97:2543–2565.
3. Stagnaro-Green A, Abalovich M, Alexander E, et al. Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and postpartum. Thyroid. 2011;21(10):1081-1125.
4. Alexander EK, Marqusee E, Lawrence J, et al. Timing and magnitude of increases in levothyroxine requirements during pregnancy in women with hypothyroidism. N Engl J Med. 2004;351:241-249.
5. Negro R, Formoso G, Mangieri T, et al. Levothyroxine treatment in euthyroid pregnant women with autoimmune thyroid disease: effects on obstetrical complications. Clin Endocrinol Metab. 2006;91(7):2587-2591.
6. Cooper DH. Antithyroid drugs. N Engl J Med. 2005;352:905-917.
7. American College of Obstetricians and Gynecologists. Routine thyroid screening not recommended for pregnant women. J Obstet Gynecol. 2007;110:959-960.
Management of thyroid disease during pregnancy presents unique challenges due to physiologic changes that occur. These include
• Serum levels of thyroxine-binding globulin (TBG) increase along with estrogen; in turn, total thyroxine (T4) and triiodothyronine (T3) levels increase.
• Human chorionic gonadotropin (hCG) stimulates the thyroid stimulating hormone (TSH) receptors.1
Since hCG and TSH share similar glycoprotein subunits, a transient suppression of TSH—especially around weeks 10 to 12, when hCG concentrations peak—is considered a physiologic finding. Interpretation of thyroid function testing should be made in relation to the hCG-mediated decrease in serum TSH levels.2
The following four cases will help guide your clinical management of thyroid disease in both preconception and pregnancy. Inadequately controlled thyroid dysfunction can lead to poor pregnancy outcomes for both mother and child, which will be further discussed.
Continue for Case 1: Stable Hypothyroidism >>
CASE 1: STABLE HYPOTHYROIDISM
A 29-year-old woman with stable primary hypothyroidism calls your office to report that she is pregnant. She has taken levothyroxine (100 mg) for the past three years, and her TSH level was 1.21 mIU/L at last measurement. She denies any symptoms of hyperthyroidism or hypothyroidism. What is your next step in her management?
Recommendation
The American Thyroid Association recommends monitoring serum TSH every four weeks during the first half of pregnancy and at least once per trimester thereafter, with frequency depending on symptoms and TSH levels.3 Most women will require higher doses of levothyroxine supplementation to maintain therapeutic TSH levels.
Prior to 18 weeks’ gestation, the fetus is dependent on maternal thyroid hormone. When pregnancy is confirmed, there is support in the literature for having the patient take two additional doses of levothyroxine per week until TSH can be tested.4 However, many endocrinology practices opt to check TSH and total T4 as soon as pregnancy is confirmed.
Since free T4 results may be unreliable during pregnancy (due to the effect of TBG), free thyroxine index (FTI) or total T4 should be monitored instead. FTI mathematically corrects free T4 for TBG levels, making it a useful marker. If total T4 is measured, it is important to remember that results will be approximately 1.5x the nonpregnancy value; thus, the reference range must be multiplied by 1.5 to calculate appropriate high and low parameters for pregnant patients.
Ideally, all women of childbearing age should be encouraged to plan pregnancy, to ensure TSH is at target prior to conception. Maintaining a euthyroid state throughout pregnancy (starting at conception) is important to decrease risk for such adverse outcomes as spontaneous abortion, placental abruption, and gestational hypertension.2 Low birth weight and respiratory distress are potential complications for newborns whose mothers have inadequately controlled hypothyroidism.
Patients should be counseled against simultaneous dosing of prenatal vitamins and levothyroxine. Prenatal vitamins contain iron, which reduces absorption of levothyroxine; therefore, it is recommended that the levothyroxine be taken four hours or more apart from prenatal vitamins.
The Endocrine Society recommends a TSH no higher than 2.5 mIU/L for hypothyroidism diagnosed prior to pregnancy.2,3 After delivery, levothyroxine doses should be reduced to prepregnancy levels, with close monitoring of TSH.2
Next page: Case 2 >>
CASE 2: HISTORY OF SPONTANEOUS ABORTION
A 36-year-old G3P0 woman visits your office for a work-up after her third spontaneous abortion at 16 weeks. The patient denies history of thyroid disease but notes her maternal grandmother has Hashimoto disease. She denies symptoms of hyperthyroidism or hypothyroidism.
Recommendation
Both hyperthyroidism and hypothyroidism are associated with an increase in spontaneous abortion, premature labor, and low birth weight. Negro et al observed an increased risk for fetal loss, small-for-gestational-age fetus, premature delivery, and premature mortality in women who were TPO-antibody-positive, even if they were euthyroid. Improved fetal outcomes occurred when TPO-antibody-positive mothers received supplemental levothyroxine.5
However, the American Thyroid Association and the Endocrine Society state there is currently insufficient evidence to recommend universal screening of thyroid antibodies during pregnancy.2,3 Obtaining thyroid function studies and TPO-antibody tests could be considered as part of a work-up for women who experience multiple spontaneous abortions or have a personal or family history of autoimmune diseases.
Continue for Case 3: Cardiovascular Symptoms >>
CASE 3: CARDIOVASCULAR SYMPTOMS
A 24-year-old primigravida woman presents with complaints of palpitations and increased anxiety. She is currently 28 weeks pregnant. Her TSH level is undetectable (< 0.01 mIU/L), and her free T4 is 2.1 mg/dL (reference range, 0.5-1.6 mg/dL). An ECG performed at your office shows sinus tachycardia with a rate of 127 beats/min.
Recommendation
Maternal hyperthyroidism increases risk for maternal congestive heart failure, uncontrolled hypertension, atrial fibrillation, and thyroid storm. Additionally, fetal hyperthyroidism can occur, especially if the mother has Graves disease. Since thyroid-stimulating immunoglobulins (TSI) can permeate the placental barrier, poor fetal growth, cardiac failure, and fetal thyrotoxicosis are severe adverse effects of in-utero TSI exposure.
To prevent further complications, antithyroid medications should be started in this case. Methimazole (MMI) carries a risk for a rare birth defect, aplasia cutis, in the first trimester and is best avoided during this time. Propylthiouracil (PTU) should be given in the first trimester and then switched to MMI in the second trimester to decrease the risk for hepatotoxicity associated with PTU. Breastfeeding mothers should be assured that low-dose MMI is generally considered safe for breastfed infants but should be taken after feeding in divided doses if possible.1
For symptom relief, b-blockers can be used, although they do come with some risks. As pregnancy Category C drugs, b-blockers are associated with neonatal growth retardation, hypoglycemia, hypoxia, lower Apgar scores, and neonatal respiratory distress.6 Consider giving the lowest dose possible for the duration of the patient’s symptoms.
Radioactive iodine (I-131) should not be given to patients who plan to become pregnant or who are pregnant.2,3 The Endocrine Society recommends that if a woman inadvertently becomes pregnant, she should be counseled on the risks of radiation to the fetus, which include thyroid destruction if treatment occurs/continues after the 12th week of pregnancy.2 Furthermore, pregnancy should be avoided for the first six months after thyroid ablation to allow sufficient time to obtain the target maternal serum TSH level of 0.3 to 2.5 mIU/L.
Next page: Case 4 >>
CASE 4: PRECONCEPTION SCREENING
A 39-year-old G0P0 woman presents for preconception counseling. She denies family or personal history of thyroid disease and symptoms of thyroid disease. Should she be screened?
Recommendation
There is no consensus or guideline regarding preconception laboratory screening for thyroid disease. Current guidelines by the American Thyroid Association, the American College of Obstetricians and Gynecologists, and The Endocrine Society recommend targeted, not universal, screening.2,3,7
The American Thyroid Association and the Endocrine Society recommend screening TSH in women who are pregnant or intend to become pregnant and
• Have a personal or family history of thyroid disease
• Are older than 30
• Demonstrate symptoms of thyroid dysfunction
• Have goiter
• Are TPO-antibody positive
• Have type 1 diabetes or other autoimmune disorders
• Have a history of miscarriage or preterm delivery
• Have a history of head or neck radiation or thyroid surgery
• Are morbidly obese (BMI > 40)
• Use amiodarone or lithium or were exposed to iodinated radiologic contrast
• Are infertile
• Live in an area with moderate to severe iodine insufficiency.2,3
Rationale for targeted screening of asymptomatic women: Large-scale research has not demonstrated significantly better outcomes in those with subclinical hypothyroidism who receive treatment.7 Small studies2 have demonstrated improved fetal outcomes when subclinical hypothyroidism is treated, but for large bodies (eg, the US Preventive Services Task Force) to recommend screening, a clear improvement in health outcomes must be established via controlled studies. Future research should evaluate the effect of treating subclinical hypothyroidism during pregnancy.
REFERENCES
1. Ballabio, M, Poshychinda M, Ekins RP. Pregnancy-induced changes in thyroid function: role of human chorionic gonadotropin as putative regulator of maternal thyroid. J Clin Endocrinol Metab. 1991;73(4):824.
2. The Endocrine Society. Management of thyroid disease in pregnancy and postpartum. J Clin Endocrinol Metab. 2012;97:2543–2565.
3. Stagnaro-Green A, Abalovich M, Alexander E, et al. Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and postpartum. Thyroid. 2011;21(10):1081-1125.
4. Alexander EK, Marqusee E, Lawrence J, et al. Timing and magnitude of increases in levothyroxine requirements during pregnancy in women with hypothyroidism. N Engl J Med. 2004;351:241-249.
5. Negro R, Formoso G, Mangieri T, et al. Levothyroxine treatment in euthyroid pregnant women with autoimmune thyroid disease: effects on obstetrical complications. Clin Endocrinol Metab. 2006;91(7):2587-2591.
6. Cooper DH. Antithyroid drugs. N Engl J Med. 2005;352:905-917.
7. American College of Obstetricians and Gynecologists. Routine thyroid screening not recommended for pregnant women. J Obstet Gynecol. 2007;110:959-960.
Management of thyroid disease during pregnancy presents unique challenges due to physiologic changes that occur. These include
• Serum levels of thyroxine-binding globulin (TBG) increase along with estrogen; in turn, total thyroxine (T4) and triiodothyronine (T3) levels increase.
• Human chorionic gonadotropin (hCG) stimulates the thyroid stimulating hormone (TSH) receptors.1
Since hCG and TSH share similar glycoprotein subunits, a transient suppression of TSH—especially around weeks 10 to 12, when hCG concentrations peak—is considered a physiologic finding. Interpretation of thyroid function testing should be made in relation to the hCG-mediated decrease in serum TSH levels.2
The following four cases will help guide your clinical management of thyroid disease in both preconception and pregnancy. Inadequately controlled thyroid dysfunction can lead to poor pregnancy outcomes for both mother and child, which will be further discussed.
Continue for Case 1: Stable Hypothyroidism >>
CASE 1: STABLE HYPOTHYROIDISM
A 29-year-old woman with stable primary hypothyroidism calls your office to report that she is pregnant. She has taken levothyroxine (100 mg) for the past three years, and her TSH level was 1.21 mIU/L at last measurement. She denies any symptoms of hyperthyroidism or hypothyroidism. What is your next step in her management?
Recommendation
The American Thyroid Association recommends monitoring serum TSH every four weeks during the first half of pregnancy and at least once per trimester thereafter, with frequency depending on symptoms and TSH levels.3 Most women will require higher doses of levothyroxine supplementation to maintain therapeutic TSH levels.
Prior to 18 weeks’ gestation, the fetus is dependent on maternal thyroid hormone. When pregnancy is confirmed, there is support in the literature for having the patient take two additional doses of levothyroxine per week until TSH can be tested.4 However, many endocrinology practices opt to check TSH and total T4 as soon as pregnancy is confirmed.
Since free T4 results may be unreliable during pregnancy (due to the effect of TBG), free thyroxine index (FTI) or total T4 should be monitored instead. FTI mathematically corrects free T4 for TBG levels, making it a useful marker. If total T4 is measured, it is important to remember that results will be approximately 1.5x the nonpregnancy value; thus, the reference range must be multiplied by 1.5 to calculate appropriate high and low parameters for pregnant patients.
Ideally, all women of childbearing age should be encouraged to plan pregnancy, to ensure TSH is at target prior to conception. Maintaining a euthyroid state throughout pregnancy (starting at conception) is important to decrease risk for such adverse outcomes as spontaneous abortion, placental abruption, and gestational hypertension.2 Low birth weight and respiratory distress are potential complications for newborns whose mothers have inadequately controlled hypothyroidism.
Patients should be counseled against simultaneous dosing of prenatal vitamins and levothyroxine. Prenatal vitamins contain iron, which reduces absorption of levothyroxine; therefore, it is recommended that the levothyroxine be taken four hours or more apart from prenatal vitamins.
The Endocrine Society recommends a TSH no higher than 2.5 mIU/L for hypothyroidism diagnosed prior to pregnancy.2,3 After delivery, levothyroxine doses should be reduced to prepregnancy levels, with close monitoring of TSH.2
Next page: Case 2 >>
CASE 2: HISTORY OF SPONTANEOUS ABORTION
A 36-year-old G3P0 woman visits your office for a work-up after her third spontaneous abortion at 16 weeks. The patient denies history of thyroid disease but notes her maternal grandmother has Hashimoto disease. She denies symptoms of hyperthyroidism or hypothyroidism.
Recommendation
Both hyperthyroidism and hypothyroidism are associated with an increase in spontaneous abortion, premature labor, and low birth weight. Negro et al observed an increased risk for fetal loss, small-for-gestational-age fetus, premature delivery, and premature mortality in women who were TPO-antibody-positive, even if they were euthyroid. Improved fetal outcomes occurred when TPO-antibody-positive mothers received supplemental levothyroxine.5
However, the American Thyroid Association and the Endocrine Society state there is currently insufficient evidence to recommend universal screening of thyroid antibodies during pregnancy.2,3 Obtaining thyroid function studies and TPO-antibody tests could be considered as part of a work-up for women who experience multiple spontaneous abortions or have a personal or family history of autoimmune diseases.
Continue for Case 3: Cardiovascular Symptoms >>
CASE 3: CARDIOVASCULAR SYMPTOMS
A 24-year-old primigravida woman presents with complaints of palpitations and increased anxiety. She is currently 28 weeks pregnant. Her TSH level is undetectable (< 0.01 mIU/L), and her free T4 is 2.1 mg/dL (reference range, 0.5-1.6 mg/dL). An ECG performed at your office shows sinus tachycardia with a rate of 127 beats/min.
Recommendation
Maternal hyperthyroidism increases risk for maternal congestive heart failure, uncontrolled hypertension, atrial fibrillation, and thyroid storm. Additionally, fetal hyperthyroidism can occur, especially if the mother has Graves disease. Since thyroid-stimulating immunoglobulins (TSI) can permeate the placental barrier, poor fetal growth, cardiac failure, and fetal thyrotoxicosis are severe adverse effects of in-utero TSI exposure.
To prevent further complications, antithyroid medications should be started in this case. Methimazole (MMI) carries a risk for a rare birth defect, aplasia cutis, in the first trimester and is best avoided during this time. Propylthiouracil (PTU) should be given in the first trimester and then switched to MMI in the second trimester to decrease the risk for hepatotoxicity associated with PTU. Breastfeeding mothers should be assured that low-dose MMI is generally considered safe for breastfed infants but should be taken after feeding in divided doses if possible.1
For symptom relief, b-blockers can be used, although they do come with some risks. As pregnancy Category C drugs, b-blockers are associated with neonatal growth retardation, hypoglycemia, hypoxia, lower Apgar scores, and neonatal respiratory distress.6 Consider giving the lowest dose possible for the duration of the patient’s symptoms.
Radioactive iodine (I-131) should not be given to patients who plan to become pregnant or who are pregnant.2,3 The Endocrine Society recommends that if a woman inadvertently becomes pregnant, she should be counseled on the risks of radiation to the fetus, which include thyroid destruction if treatment occurs/continues after the 12th week of pregnancy.2 Furthermore, pregnancy should be avoided for the first six months after thyroid ablation to allow sufficient time to obtain the target maternal serum TSH level of 0.3 to 2.5 mIU/L.
Next page: Case 4 >>
CASE 4: PRECONCEPTION SCREENING
A 39-year-old G0P0 woman presents for preconception counseling. She denies family or personal history of thyroid disease and symptoms of thyroid disease. Should she be screened?
Recommendation
There is no consensus or guideline regarding preconception laboratory screening for thyroid disease. Current guidelines by the American Thyroid Association, the American College of Obstetricians and Gynecologists, and The Endocrine Society recommend targeted, not universal, screening.2,3,7
The American Thyroid Association and the Endocrine Society recommend screening TSH in women who are pregnant or intend to become pregnant and
• Have a personal or family history of thyroid disease
• Are older than 30
• Demonstrate symptoms of thyroid dysfunction
• Have goiter
• Are TPO-antibody positive
• Have type 1 diabetes or other autoimmune disorders
• Have a history of miscarriage or preterm delivery
• Have a history of head or neck radiation or thyroid surgery
• Are morbidly obese (BMI > 40)
• Use amiodarone or lithium or were exposed to iodinated radiologic contrast
• Are infertile
• Live in an area with moderate to severe iodine insufficiency.2,3
Rationale for targeted screening of asymptomatic women: Large-scale research has not demonstrated significantly better outcomes in those with subclinical hypothyroidism who receive treatment.7 Small studies2 have demonstrated improved fetal outcomes when subclinical hypothyroidism is treated, but for large bodies (eg, the US Preventive Services Task Force) to recommend screening, a clear improvement in health outcomes must be established via controlled studies. Future research should evaluate the effect of treating subclinical hypothyroidism during pregnancy.
REFERENCES
1. Ballabio, M, Poshychinda M, Ekins RP. Pregnancy-induced changes in thyroid function: role of human chorionic gonadotropin as putative regulator of maternal thyroid. J Clin Endocrinol Metab. 1991;73(4):824.
2. The Endocrine Society. Management of thyroid disease in pregnancy and postpartum. J Clin Endocrinol Metab. 2012;97:2543–2565.
3. Stagnaro-Green A, Abalovich M, Alexander E, et al. Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and postpartum. Thyroid. 2011;21(10):1081-1125.
4. Alexander EK, Marqusee E, Lawrence J, et al. Timing and magnitude of increases in levothyroxine requirements during pregnancy in women with hypothyroidism. N Engl J Med. 2004;351:241-249.
5. Negro R, Formoso G, Mangieri T, et al. Levothyroxine treatment in euthyroid pregnant women with autoimmune thyroid disease: effects on obstetrical complications. Clin Endocrinol Metab. 2006;91(7):2587-2591.
6. Cooper DH. Antithyroid drugs. N Engl J Med. 2005;352:905-917.
7. American College of Obstetricians and Gynecologists. Routine thyroid screening not recommended for pregnant women. J Obstet Gynecol. 2007;110:959-960.