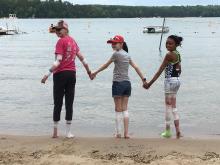User login
When treating AD in children, experts consider adherence, other aspects of treatment
ASHEVILLE, N.C. – according to a three-member expert panel mulling over strategies at the annual meeting of the Society for Pediatric Dermatology.
In introductory remarks, the three panelists briefly addressed different aspects for controlling AD, including drugs in the pipeline, the potential value of alternative therapies, and whom to blame when compliance is poor.
But panel discussion following these presentations provided an opportunity for audience engagement on practical strategies for improving AD control.
In her formal remarks prior to the panel discussion, Amy S. Paller, MD, professor of dermatology and pediatrics and chair of dermatology, Northwestern University, Chicago, and a pediatric dermatologist at the Lurie Children’s Hospital of Chicago, described emerging AD treatments. This included an update on the status of the interleukin-13 (IL-13) inhibitors tralokinumab (Adbry), which was approved by the FDA for treating AD in adults in December 2021, and lebrikizumab, which is thought likely to be soon approved in the United States on the basis of two recently published phase 3 trials.
Along with dupilumab (Dupixent) for moderate-to-severe AD in children who do not respond to optimized use of topical therapies, these new biologics appear likely to further expand choices for AD control for adults (and for kids with AD too, if eventually licensed in children), according to the data from the phase 3 studies.
During a panel discussion that followed, Stephen Gellis, MD, pediatric dermatologist and former chief of pediatric dermatology at Boston Children’s Hospital and Harvard Medical School, raised the point of optimizing tried and true topical therapies before using systemic agents. He noted that parents sometimes pressure clinicians to use a biologic – and that moving too quickly to the latest and most expensive drugs may not be necessary.
Dr. Paller acknowledged that she, like many pediatric dermatologists, employed immunosuppressants as her drugs of choice for many years – commonly starting with a few months of cyclosporine before transitioning to methotrexate, which has a delayed onset of action. In fact, she still uses this regimen in some children.
However, she now prefers dupilumab, which is the first biologic available for children in the United States with an AD indication in children as young as 6 months. She said dupilumab has fewer potential risks than cyclosporine, and it offers clinically meaningful improvement in most children. She noted that current guidelines discourage the use of systemic corticosteroids for AD in children, given their potential toxicity.
She strongly agreed with Dr. Gellis that clinicians should resist pressure to use any systemic agent if children are responding well to topical medications. In her own practice, Dr. Paller moves to systemic medications only after ensuring that there has been adherence to appropriate therapy and that there is not another diagnosis that might explain the recalcitrance to topical agents.
When a systemic medication is considered the next step, Dr. Paller reminded the audience of the importance of presenting the benefits and risks of all the options for AD control, which could include dupilumab and immunosuppressants as initial systemic therapy.
“Many parents choose biologic treatment first, given its lack of requirement for blood monitoring and faster action than methotrexate,” Dr. Paller noted.
Nevertheless, “biologics are much more costly than immunosuppressants, require an injection – which is stressful for the child and the parents – and may not be accessible for our patients,” Dr. Paller said. Cyclosporine and methotrexate are effective and are often the best options for moderate to severe disease in areas of the world where dupilumab is not available, but Dr. Paller most commonly uses these therapies only when reimbursement for dupilumab cannot be secured, injection is not an option, or when dupilumab is not sufficiently effective and tolerated.
Providing different perspectives, the two other panelists discussing the treatment of pediatric AD also saw a role for ensuring that topical agents are not offering adequate AD control before turning to the latest and most sophisticated therapies for AD.
For meeting parent expectations when children are improving slowly on topical therapies, Peter A. Lio, MD, director of the Chicago Integrative Eczema Center and clinical assistant professor of dermatology and pediatrics at Northwestern University, suggested that integrative medicine might be helpful.
For parents not fully comfortable with standard pharmacologic agents, Dr. Lio said there is evidence to support some of the complementary approaches, and these can be reassuring to parents with an interest in alternative medicines.
In Western medicine, it is common to hear terms like “attack,” “kill,” and “suppress,” disease, but alternative therapies are generally coupled with terms like “restore,” “strengthen,” and “tonify,” he said. “Who doesn’t want to be tonified?” he asked, noting that there are many sources of data suggesting that the number of patients seeking alternative medicine is “huge.” The alternative medicines are not generally taught in medical school and remain widely ignored in typical practice, but “our patients are interested even if we are not.”
Yet, there are data to support benefit from some of these alternative therapies, providing a win-win situation for patients who derive satisfaction from nontraditional therapies alone or combined with established pharmaceutical treatments.
Of these, Dr. Lio said there is support for the use of hempseed oil as a moisturizing agent and a strategy for improving barrier function in the skin of patients with AD. In a controlled crossover study, 2 teaspoons per day of dietary hempseed oil, a product that can be purchased in some grocery stores, was associated with significant reductions in skin dryness, itchiness, and use of topical medications relative to the same amount of olive oil, he noted.
Other examples include a compress made with black tea that was associated with an anti-inflammatory effect when followed by a moisturizer, a published study asserts. Although this was a trial in adults with facial dermatitis, Dr. Lio suggested that the same anti-inflammatory effect would be anticipated for other skin conditions, including AD in children.
As a third example, Dr. Lio said topical indigo, a traditional Chinese medicine used for a variety of dermatologic conditions, including psoriasis, has also demonstrated efficacy in a randomized trial, compared with vehicle for mild to severe AD.
Complementary medicines are not for everyone, but they may have a role when managing the expectations of parents who are not fully satisfied or express concern about regimens limited to mainstream therapies alone, according to Dr. Lio. In diseases that are not curable, such as AD, he thinks this is a strategy with potential for benefit and is reassuring to patients.
Another way to avoid moving to riskier or more expensive drugs quickly is to assure patients use the drugs that were prescribed first, according to Steven R. Feldman, MD, PhD, professor of dermatology, Wake Forest University, Winston-Salem, N.C.
Dr. Feldman believes that failure to adhere to therapy is basically the fault of the medical care system, not the patient. He made an analogy to a successful piano teacher, who provides a child with sheet music and then sees the child once a week to track progress. He juxtaposed this piano teacher to one who gives the child sheet music and tells the child to come back in 10 weeks for the recital. It is not hard to guess which approach would be more effective.
“Typically, doctors are worse than that second teacher,” he said. “Doctors are like a piano teacher that does not give you the sheet music but says, ‘Here is a prescription for some sheet music. Take this prescription to the sheet music store. I have no idea how much it will cost or whether your insurance will pay for it. But once you fill this prescription for sheet music, I want you to practice this every day,’ ” he said, adding, “Practicing this sheet music may cause rashes, diarrhea, or serious infection. When the patient next comes in 10-12 weeks later and is not better, the doctor says, ‘I will give you a harder piece of sheet music and maybe two or three other instruments to practice at the same time,’ ” said Dr. Feldman, expressing why the way clinicians practice might explain much of the poor adherence problem.
This largely explains why patients with AD do not immediately respond to the therapies doctors prescribe, Dr. Feldman implied, reiterating the theme that emerged from the AD panel: Better and more options are needed for AD of the most severe types, but better management, not better drugs, is typically what is needed for most patients.
Dr. Feldman, Dr. Lio, and Dr. Paller have financial relationships with more than 30 pharmaceutical and cosmetic companies, some of which manufacture therapies for atopic dermatitis.
This article was updated July 28, 2023, to clarify the comments and viewpoints of Dr. Amy Paller.
A version of this article first appeared on Medscape.com.
ASHEVILLE, N.C. – according to a three-member expert panel mulling over strategies at the annual meeting of the Society for Pediatric Dermatology.
In introductory remarks, the three panelists briefly addressed different aspects for controlling AD, including drugs in the pipeline, the potential value of alternative therapies, and whom to blame when compliance is poor.
But panel discussion following these presentations provided an opportunity for audience engagement on practical strategies for improving AD control.
In her formal remarks prior to the panel discussion, Amy S. Paller, MD, professor of dermatology and pediatrics and chair of dermatology, Northwestern University, Chicago, and a pediatric dermatologist at the Lurie Children’s Hospital of Chicago, described emerging AD treatments. This included an update on the status of the interleukin-13 (IL-13) inhibitors tralokinumab (Adbry), which was approved by the FDA for treating AD in adults in December 2021, and lebrikizumab, which is thought likely to be soon approved in the United States on the basis of two recently published phase 3 trials.
Along with dupilumab (Dupixent) for moderate-to-severe AD in children who do not respond to optimized use of topical therapies, these new biologics appear likely to further expand choices for AD control for adults (and for kids with AD too, if eventually licensed in children), according to the data from the phase 3 studies.
During a panel discussion that followed, Stephen Gellis, MD, pediatric dermatologist and former chief of pediatric dermatology at Boston Children’s Hospital and Harvard Medical School, raised the point of optimizing tried and true topical therapies before using systemic agents. He noted that parents sometimes pressure clinicians to use a biologic – and that moving too quickly to the latest and most expensive drugs may not be necessary.
Dr. Paller acknowledged that she, like many pediatric dermatologists, employed immunosuppressants as her drugs of choice for many years – commonly starting with a few months of cyclosporine before transitioning to methotrexate, which has a delayed onset of action. In fact, she still uses this regimen in some children.
However, she now prefers dupilumab, which is the first biologic available for children in the United States with an AD indication in children as young as 6 months. She said dupilumab has fewer potential risks than cyclosporine, and it offers clinically meaningful improvement in most children. She noted that current guidelines discourage the use of systemic corticosteroids for AD in children, given their potential toxicity.
She strongly agreed with Dr. Gellis that clinicians should resist pressure to use any systemic agent if children are responding well to topical medications. In her own practice, Dr. Paller moves to systemic medications only after ensuring that there has been adherence to appropriate therapy and that there is not another diagnosis that might explain the recalcitrance to topical agents.
When a systemic medication is considered the next step, Dr. Paller reminded the audience of the importance of presenting the benefits and risks of all the options for AD control, which could include dupilumab and immunosuppressants as initial systemic therapy.
“Many parents choose biologic treatment first, given its lack of requirement for blood monitoring and faster action than methotrexate,” Dr. Paller noted.
Nevertheless, “biologics are much more costly than immunosuppressants, require an injection – which is stressful for the child and the parents – and may not be accessible for our patients,” Dr. Paller said. Cyclosporine and methotrexate are effective and are often the best options for moderate to severe disease in areas of the world where dupilumab is not available, but Dr. Paller most commonly uses these therapies only when reimbursement for dupilumab cannot be secured, injection is not an option, or when dupilumab is not sufficiently effective and tolerated.
Providing different perspectives, the two other panelists discussing the treatment of pediatric AD also saw a role for ensuring that topical agents are not offering adequate AD control before turning to the latest and most sophisticated therapies for AD.
For meeting parent expectations when children are improving slowly on topical therapies, Peter A. Lio, MD, director of the Chicago Integrative Eczema Center and clinical assistant professor of dermatology and pediatrics at Northwestern University, suggested that integrative medicine might be helpful.
For parents not fully comfortable with standard pharmacologic agents, Dr. Lio said there is evidence to support some of the complementary approaches, and these can be reassuring to parents with an interest in alternative medicines.
In Western medicine, it is common to hear terms like “attack,” “kill,” and “suppress,” disease, but alternative therapies are generally coupled with terms like “restore,” “strengthen,” and “tonify,” he said. “Who doesn’t want to be tonified?” he asked, noting that there are many sources of data suggesting that the number of patients seeking alternative medicine is “huge.” The alternative medicines are not generally taught in medical school and remain widely ignored in typical practice, but “our patients are interested even if we are not.”
Yet, there are data to support benefit from some of these alternative therapies, providing a win-win situation for patients who derive satisfaction from nontraditional therapies alone or combined with established pharmaceutical treatments.
Of these, Dr. Lio said there is support for the use of hempseed oil as a moisturizing agent and a strategy for improving barrier function in the skin of patients with AD. In a controlled crossover study, 2 teaspoons per day of dietary hempseed oil, a product that can be purchased in some grocery stores, was associated with significant reductions in skin dryness, itchiness, and use of topical medications relative to the same amount of olive oil, he noted.
Other examples include a compress made with black tea that was associated with an anti-inflammatory effect when followed by a moisturizer, a published study asserts. Although this was a trial in adults with facial dermatitis, Dr. Lio suggested that the same anti-inflammatory effect would be anticipated for other skin conditions, including AD in children.
As a third example, Dr. Lio said topical indigo, a traditional Chinese medicine used for a variety of dermatologic conditions, including psoriasis, has also demonstrated efficacy in a randomized trial, compared with vehicle for mild to severe AD.
Complementary medicines are not for everyone, but they may have a role when managing the expectations of parents who are not fully satisfied or express concern about regimens limited to mainstream therapies alone, according to Dr. Lio. In diseases that are not curable, such as AD, he thinks this is a strategy with potential for benefit and is reassuring to patients.
Another way to avoid moving to riskier or more expensive drugs quickly is to assure patients use the drugs that were prescribed first, according to Steven R. Feldman, MD, PhD, professor of dermatology, Wake Forest University, Winston-Salem, N.C.
Dr. Feldman believes that failure to adhere to therapy is basically the fault of the medical care system, not the patient. He made an analogy to a successful piano teacher, who provides a child with sheet music and then sees the child once a week to track progress. He juxtaposed this piano teacher to one who gives the child sheet music and tells the child to come back in 10 weeks for the recital. It is not hard to guess which approach would be more effective.
“Typically, doctors are worse than that second teacher,” he said. “Doctors are like a piano teacher that does not give you the sheet music but says, ‘Here is a prescription for some sheet music. Take this prescription to the sheet music store. I have no idea how much it will cost or whether your insurance will pay for it. But once you fill this prescription for sheet music, I want you to practice this every day,’ ” he said, adding, “Practicing this sheet music may cause rashes, diarrhea, or serious infection. When the patient next comes in 10-12 weeks later and is not better, the doctor says, ‘I will give you a harder piece of sheet music and maybe two or three other instruments to practice at the same time,’ ” said Dr. Feldman, expressing why the way clinicians practice might explain much of the poor adherence problem.
This largely explains why patients with AD do not immediately respond to the therapies doctors prescribe, Dr. Feldman implied, reiterating the theme that emerged from the AD panel: Better and more options are needed for AD of the most severe types, but better management, not better drugs, is typically what is needed for most patients.
Dr. Feldman, Dr. Lio, and Dr. Paller have financial relationships with more than 30 pharmaceutical and cosmetic companies, some of which manufacture therapies for atopic dermatitis.
This article was updated July 28, 2023, to clarify the comments and viewpoints of Dr. Amy Paller.
A version of this article first appeared on Medscape.com.
ASHEVILLE, N.C. – according to a three-member expert panel mulling over strategies at the annual meeting of the Society for Pediatric Dermatology.
In introductory remarks, the three panelists briefly addressed different aspects for controlling AD, including drugs in the pipeline, the potential value of alternative therapies, and whom to blame when compliance is poor.
But panel discussion following these presentations provided an opportunity for audience engagement on practical strategies for improving AD control.
In her formal remarks prior to the panel discussion, Amy S. Paller, MD, professor of dermatology and pediatrics and chair of dermatology, Northwestern University, Chicago, and a pediatric dermatologist at the Lurie Children’s Hospital of Chicago, described emerging AD treatments. This included an update on the status of the interleukin-13 (IL-13) inhibitors tralokinumab (Adbry), which was approved by the FDA for treating AD in adults in December 2021, and lebrikizumab, which is thought likely to be soon approved in the United States on the basis of two recently published phase 3 trials.
Along with dupilumab (Dupixent) for moderate-to-severe AD in children who do not respond to optimized use of topical therapies, these new biologics appear likely to further expand choices for AD control for adults (and for kids with AD too, if eventually licensed in children), according to the data from the phase 3 studies.
During a panel discussion that followed, Stephen Gellis, MD, pediatric dermatologist and former chief of pediatric dermatology at Boston Children’s Hospital and Harvard Medical School, raised the point of optimizing tried and true topical therapies before using systemic agents. He noted that parents sometimes pressure clinicians to use a biologic – and that moving too quickly to the latest and most expensive drugs may not be necessary.
Dr. Paller acknowledged that she, like many pediatric dermatologists, employed immunosuppressants as her drugs of choice for many years – commonly starting with a few months of cyclosporine before transitioning to methotrexate, which has a delayed onset of action. In fact, she still uses this regimen in some children.
However, she now prefers dupilumab, which is the first biologic available for children in the United States with an AD indication in children as young as 6 months. She said dupilumab has fewer potential risks than cyclosporine, and it offers clinically meaningful improvement in most children. She noted that current guidelines discourage the use of systemic corticosteroids for AD in children, given their potential toxicity.
She strongly agreed with Dr. Gellis that clinicians should resist pressure to use any systemic agent if children are responding well to topical medications. In her own practice, Dr. Paller moves to systemic medications only after ensuring that there has been adherence to appropriate therapy and that there is not another diagnosis that might explain the recalcitrance to topical agents.
When a systemic medication is considered the next step, Dr. Paller reminded the audience of the importance of presenting the benefits and risks of all the options for AD control, which could include dupilumab and immunosuppressants as initial systemic therapy.
“Many parents choose biologic treatment first, given its lack of requirement for blood monitoring and faster action than methotrexate,” Dr. Paller noted.
Nevertheless, “biologics are much more costly than immunosuppressants, require an injection – which is stressful for the child and the parents – and may not be accessible for our patients,” Dr. Paller said. Cyclosporine and methotrexate are effective and are often the best options for moderate to severe disease in areas of the world where dupilumab is not available, but Dr. Paller most commonly uses these therapies only when reimbursement for dupilumab cannot be secured, injection is not an option, or when dupilumab is not sufficiently effective and tolerated.
Providing different perspectives, the two other panelists discussing the treatment of pediatric AD also saw a role for ensuring that topical agents are not offering adequate AD control before turning to the latest and most sophisticated therapies for AD.
For meeting parent expectations when children are improving slowly on topical therapies, Peter A. Lio, MD, director of the Chicago Integrative Eczema Center and clinical assistant professor of dermatology and pediatrics at Northwestern University, suggested that integrative medicine might be helpful.
For parents not fully comfortable with standard pharmacologic agents, Dr. Lio said there is evidence to support some of the complementary approaches, and these can be reassuring to parents with an interest in alternative medicines.
In Western medicine, it is common to hear terms like “attack,” “kill,” and “suppress,” disease, but alternative therapies are generally coupled with terms like “restore,” “strengthen,” and “tonify,” he said. “Who doesn’t want to be tonified?” he asked, noting that there are many sources of data suggesting that the number of patients seeking alternative medicine is “huge.” The alternative medicines are not generally taught in medical school and remain widely ignored in typical practice, but “our patients are interested even if we are not.”
Yet, there are data to support benefit from some of these alternative therapies, providing a win-win situation for patients who derive satisfaction from nontraditional therapies alone or combined with established pharmaceutical treatments.
Of these, Dr. Lio said there is support for the use of hempseed oil as a moisturizing agent and a strategy for improving barrier function in the skin of patients with AD. In a controlled crossover study, 2 teaspoons per day of dietary hempseed oil, a product that can be purchased in some grocery stores, was associated with significant reductions in skin dryness, itchiness, and use of topical medications relative to the same amount of olive oil, he noted.
Other examples include a compress made with black tea that was associated with an anti-inflammatory effect when followed by a moisturizer, a published study asserts. Although this was a trial in adults with facial dermatitis, Dr. Lio suggested that the same anti-inflammatory effect would be anticipated for other skin conditions, including AD in children.
As a third example, Dr. Lio said topical indigo, a traditional Chinese medicine used for a variety of dermatologic conditions, including psoriasis, has also demonstrated efficacy in a randomized trial, compared with vehicle for mild to severe AD.
Complementary medicines are not for everyone, but they may have a role when managing the expectations of parents who are not fully satisfied or express concern about regimens limited to mainstream therapies alone, according to Dr. Lio. In diseases that are not curable, such as AD, he thinks this is a strategy with potential for benefit and is reassuring to patients.
Another way to avoid moving to riskier or more expensive drugs quickly is to assure patients use the drugs that were prescribed first, according to Steven R. Feldman, MD, PhD, professor of dermatology, Wake Forest University, Winston-Salem, N.C.
Dr. Feldman believes that failure to adhere to therapy is basically the fault of the medical care system, not the patient. He made an analogy to a successful piano teacher, who provides a child with sheet music and then sees the child once a week to track progress. He juxtaposed this piano teacher to one who gives the child sheet music and tells the child to come back in 10 weeks for the recital. It is not hard to guess which approach would be more effective.
“Typically, doctors are worse than that second teacher,” he said. “Doctors are like a piano teacher that does not give you the sheet music but says, ‘Here is a prescription for some sheet music. Take this prescription to the sheet music store. I have no idea how much it will cost or whether your insurance will pay for it. But once you fill this prescription for sheet music, I want you to practice this every day,’ ” he said, adding, “Practicing this sheet music may cause rashes, diarrhea, or serious infection. When the patient next comes in 10-12 weeks later and is not better, the doctor says, ‘I will give you a harder piece of sheet music and maybe two or three other instruments to practice at the same time,’ ” said Dr. Feldman, expressing why the way clinicians practice might explain much of the poor adherence problem.
This largely explains why patients with AD do not immediately respond to the therapies doctors prescribe, Dr. Feldman implied, reiterating the theme that emerged from the AD panel: Better and more options are needed for AD of the most severe types, but better management, not better drugs, is typically what is needed for most patients.
Dr. Feldman, Dr. Lio, and Dr. Paller have financial relationships with more than 30 pharmaceutical and cosmetic companies, some of which manufacture therapies for atopic dermatitis.
This article was updated July 28, 2023, to clarify the comments and viewpoints of Dr. Amy Paller.
A version of this article first appeared on Medscape.com.
AT SPD 2023
Ocular complications of dermatologic treatments: Advice from a pediatric ophthalmologist
ASHEVILLE, N.C. – The, according to one of several clinical messages from a pediatric ophthalmologist who spoke at the annual meeting of the Society for Pediatric Dermatology.
“There is a lot of steroid fear out there, which you can argue is actually harmful in itself, because not treating periorbital eczema is related to a lot of eye problems, including chronic discomfort and the eye rubbing that can cause corneal abrasions and keratoconus,” said Sara Grace, MD, a pediatric ophthalmologist who is on the clinical staff at Duke University, Durham, N.C. She maintains a practice at North Carolina Eye, Ear, Nose, and Throat in Durham.
Although the risks of periorbital steroid absorption are real, a limited course of low potency topical steroids is generally adequate for common periorbital indications, and these appear to be safe.
“There is insufficient evidence to link weak periocular topical corticosteroids such as desonide or hydrocortisone with ocular complications,” said Dr. Grace, suggesting that pediatric dermatologists can be reassured when using these medications at low concentrations.
“Potent periocular steroids have been associated with ocular complications, but this has typically involved exposures over months to years,” Dr. Grace specified.
When topical corticosteroids are applied at high concentrations on the face away from the periorbital area, glaucoma and other feared ophthalmic complications cannot be entirely ruled out, but, again, the risk is low in the absence of “very large quantities” of potent topical agents applied for lengthy periods of time, according to Dr. Grace, basing this observation on case studies.
In children, as in adults, the potential exception is a child with existing ocular disease. In such cases, or in children with risk factors for ocular disease, Dr. Grace recommends referral to an ophthalmologist for a baseline examination prior to a course of topical corticosteroids with the potential of periocular absorption. With a baseline assessment, adverse effects are more easily documented if exposure is prolonged.
The message, although not identical, is similar for use of dupilumab (Dupixent) or other biologics that target the interleukin-13 (IL-13) pathway. The potential for complications cannot be ignored but these are often time-limited and the benefit is likely to exceed the risk in children who have severe atopic dermatitis or other skin conditions for which these treatments are effective.
There are several potential mechanisms by which biologics targeting IL-13 might increase risk of ocular complications, one of which is the role that IL-13 plays in ocular mucus production, regulation of conjunctival goblet cells, and tear production, according to several published reports.
“Up to 30% of children will get some type of eye complication but, fortunately, most of them will not have to stop therapy,” Dr. Grace said. These side effects include conjunctivitis, blepharitis, keratitis, dry eye, and itching, but they are typically manageable. Topical steroids or calcineurin inhibitors can be offered if needed, but many of these conditions will self-resolve. Dr. Grace estimated that less than 1% of patients need to stop treatment because of ophthalmic side effects.
Lesions that obstruct vision
Dr. Grace urged pediatric dermatologists to be aware of the risk for amblyopia in young children with lesions that obstruct vision in one eye. In early development, prolonged obstruction of vision in one eye can alter neural communication with the brain, producing permanent vision impairment.
She explained that clearing the obstructed vision, whether from a capillary hemangioma or any periorbital growth, should be considered urgent to avoid irreversible damage.
Similarly, periorbital port-wine stains associated with Sturge-Weber syndrome, which is primarily a vascular disorder that predisposes children to glaucoma, represents a condition that requires prompt attention. Sturge-Weber syndrome is often but not always identified at birth, but it is a condition for which evaluation and treatment should involve the participation of an ophthalmologist.
Meibomian gland disease is another disorder that is often seen first by a pediatric dermatologist but also requires collaborative management. The challenge is sorting out the underlying cause or causes and initiating a therapy that unclogs the gland without having to resort to incision and drainage.
“Drainage is hard to do and is not necessarily effective,” explained Dr. Grace. While scrubs, warmth, and massage frequently are adequate to unclog the gland – which secretes meibum, a complex of lipids that perform several functions in protecting the eye – therapies specific to the cause, such as Demodex-related blepharitis, chalazions, and styes, might be needed.
Dr. Grace indicated that patience is often needed. The process of unclogging these glands often takes time, but she emphasized that a first-line conservative approach is always appropriate to avoid the difficulty and potential problems of incisions.
In general, these messages are not novel, but they provide a refresher for pediatric dermatologists who do not regularly confront complications that involve the eyes. According to session moderator, Elizabeth Neiman, MD, assistant professor of pediatric dermatology, University of North Carolina at Chapel Hill, the messages regarding topical steroids on the face and the eyes are “important” and worth emphasizing.
“It’s useful to reinforce the point that corticosteroids should be used when needed in the periorbital area [to control skin diseases] if they are used in low concentrations,” Dr. Neiman told this news organization.
Similarly, conjunctivitis and other ocular complications of dupilumab are a source of concern for parents as well as dermatologists. Dr. Neiman indicated that a review of the benefit-to-risk ratio is important when considering these treatments in patients with indications for severe skin disorders.
Dr. Grace and Dr. Nieman have no potential financial conflicts related to this topic.
A version of this article first appeared on Medscape.com.
ASHEVILLE, N.C. – The, according to one of several clinical messages from a pediatric ophthalmologist who spoke at the annual meeting of the Society for Pediatric Dermatology.
“There is a lot of steroid fear out there, which you can argue is actually harmful in itself, because not treating periorbital eczema is related to a lot of eye problems, including chronic discomfort and the eye rubbing that can cause corneal abrasions and keratoconus,” said Sara Grace, MD, a pediatric ophthalmologist who is on the clinical staff at Duke University, Durham, N.C. She maintains a practice at North Carolina Eye, Ear, Nose, and Throat in Durham.
Although the risks of periorbital steroid absorption are real, a limited course of low potency topical steroids is generally adequate for common periorbital indications, and these appear to be safe.
“There is insufficient evidence to link weak periocular topical corticosteroids such as desonide or hydrocortisone with ocular complications,” said Dr. Grace, suggesting that pediatric dermatologists can be reassured when using these medications at low concentrations.
“Potent periocular steroids have been associated with ocular complications, but this has typically involved exposures over months to years,” Dr. Grace specified.
When topical corticosteroids are applied at high concentrations on the face away from the periorbital area, glaucoma and other feared ophthalmic complications cannot be entirely ruled out, but, again, the risk is low in the absence of “very large quantities” of potent topical agents applied for lengthy periods of time, according to Dr. Grace, basing this observation on case studies.
In children, as in adults, the potential exception is a child with existing ocular disease. In such cases, or in children with risk factors for ocular disease, Dr. Grace recommends referral to an ophthalmologist for a baseline examination prior to a course of topical corticosteroids with the potential of periocular absorption. With a baseline assessment, adverse effects are more easily documented if exposure is prolonged.
The message, although not identical, is similar for use of dupilumab (Dupixent) or other biologics that target the interleukin-13 (IL-13) pathway. The potential for complications cannot be ignored but these are often time-limited and the benefit is likely to exceed the risk in children who have severe atopic dermatitis or other skin conditions for which these treatments are effective.
There are several potential mechanisms by which biologics targeting IL-13 might increase risk of ocular complications, one of which is the role that IL-13 plays in ocular mucus production, regulation of conjunctival goblet cells, and tear production, according to several published reports.
“Up to 30% of children will get some type of eye complication but, fortunately, most of them will not have to stop therapy,” Dr. Grace said. These side effects include conjunctivitis, blepharitis, keratitis, dry eye, and itching, but they are typically manageable. Topical steroids or calcineurin inhibitors can be offered if needed, but many of these conditions will self-resolve. Dr. Grace estimated that less than 1% of patients need to stop treatment because of ophthalmic side effects.
Lesions that obstruct vision
Dr. Grace urged pediatric dermatologists to be aware of the risk for amblyopia in young children with lesions that obstruct vision in one eye. In early development, prolonged obstruction of vision in one eye can alter neural communication with the brain, producing permanent vision impairment.
She explained that clearing the obstructed vision, whether from a capillary hemangioma or any periorbital growth, should be considered urgent to avoid irreversible damage.
Similarly, periorbital port-wine stains associated with Sturge-Weber syndrome, which is primarily a vascular disorder that predisposes children to glaucoma, represents a condition that requires prompt attention. Sturge-Weber syndrome is often but not always identified at birth, but it is a condition for which evaluation and treatment should involve the participation of an ophthalmologist.
Meibomian gland disease is another disorder that is often seen first by a pediatric dermatologist but also requires collaborative management. The challenge is sorting out the underlying cause or causes and initiating a therapy that unclogs the gland without having to resort to incision and drainage.
“Drainage is hard to do and is not necessarily effective,” explained Dr. Grace. While scrubs, warmth, and massage frequently are adequate to unclog the gland – which secretes meibum, a complex of lipids that perform several functions in protecting the eye – therapies specific to the cause, such as Demodex-related blepharitis, chalazions, and styes, might be needed.
Dr. Grace indicated that patience is often needed. The process of unclogging these glands often takes time, but she emphasized that a first-line conservative approach is always appropriate to avoid the difficulty and potential problems of incisions.
In general, these messages are not novel, but they provide a refresher for pediatric dermatologists who do not regularly confront complications that involve the eyes. According to session moderator, Elizabeth Neiman, MD, assistant professor of pediatric dermatology, University of North Carolina at Chapel Hill, the messages regarding topical steroids on the face and the eyes are “important” and worth emphasizing.
“It’s useful to reinforce the point that corticosteroids should be used when needed in the periorbital area [to control skin diseases] if they are used in low concentrations,” Dr. Neiman told this news organization.
Similarly, conjunctivitis and other ocular complications of dupilumab are a source of concern for parents as well as dermatologists. Dr. Neiman indicated that a review of the benefit-to-risk ratio is important when considering these treatments in patients with indications for severe skin disorders.
Dr. Grace and Dr. Nieman have no potential financial conflicts related to this topic.
A version of this article first appeared on Medscape.com.
ASHEVILLE, N.C. – The, according to one of several clinical messages from a pediatric ophthalmologist who spoke at the annual meeting of the Society for Pediatric Dermatology.
“There is a lot of steroid fear out there, which you can argue is actually harmful in itself, because not treating periorbital eczema is related to a lot of eye problems, including chronic discomfort and the eye rubbing that can cause corneal abrasions and keratoconus,” said Sara Grace, MD, a pediatric ophthalmologist who is on the clinical staff at Duke University, Durham, N.C. She maintains a practice at North Carolina Eye, Ear, Nose, and Throat in Durham.
Although the risks of periorbital steroid absorption are real, a limited course of low potency topical steroids is generally adequate for common periorbital indications, and these appear to be safe.
“There is insufficient evidence to link weak periocular topical corticosteroids such as desonide or hydrocortisone with ocular complications,” said Dr. Grace, suggesting that pediatric dermatologists can be reassured when using these medications at low concentrations.
“Potent periocular steroids have been associated with ocular complications, but this has typically involved exposures over months to years,” Dr. Grace specified.
When topical corticosteroids are applied at high concentrations on the face away from the periorbital area, glaucoma and other feared ophthalmic complications cannot be entirely ruled out, but, again, the risk is low in the absence of “very large quantities” of potent topical agents applied for lengthy periods of time, according to Dr. Grace, basing this observation on case studies.
In children, as in adults, the potential exception is a child with existing ocular disease. In such cases, or in children with risk factors for ocular disease, Dr. Grace recommends referral to an ophthalmologist for a baseline examination prior to a course of topical corticosteroids with the potential of periocular absorption. With a baseline assessment, adverse effects are more easily documented if exposure is prolonged.
The message, although not identical, is similar for use of dupilumab (Dupixent) or other biologics that target the interleukin-13 (IL-13) pathway. The potential for complications cannot be ignored but these are often time-limited and the benefit is likely to exceed the risk in children who have severe atopic dermatitis or other skin conditions for which these treatments are effective.
There are several potential mechanisms by which biologics targeting IL-13 might increase risk of ocular complications, one of which is the role that IL-13 plays in ocular mucus production, regulation of conjunctival goblet cells, and tear production, according to several published reports.
“Up to 30% of children will get some type of eye complication but, fortunately, most of them will not have to stop therapy,” Dr. Grace said. These side effects include conjunctivitis, blepharitis, keratitis, dry eye, and itching, but they are typically manageable. Topical steroids or calcineurin inhibitors can be offered if needed, but many of these conditions will self-resolve. Dr. Grace estimated that less than 1% of patients need to stop treatment because of ophthalmic side effects.
Lesions that obstruct vision
Dr. Grace urged pediatric dermatologists to be aware of the risk for amblyopia in young children with lesions that obstruct vision in one eye. In early development, prolonged obstruction of vision in one eye can alter neural communication with the brain, producing permanent vision impairment.
She explained that clearing the obstructed vision, whether from a capillary hemangioma or any periorbital growth, should be considered urgent to avoid irreversible damage.
Similarly, periorbital port-wine stains associated with Sturge-Weber syndrome, which is primarily a vascular disorder that predisposes children to glaucoma, represents a condition that requires prompt attention. Sturge-Weber syndrome is often but not always identified at birth, but it is a condition for which evaluation and treatment should involve the participation of an ophthalmologist.
Meibomian gland disease is another disorder that is often seen first by a pediatric dermatologist but also requires collaborative management. The challenge is sorting out the underlying cause or causes and initiating a therapy that unclogs the gland without having to resort to incision and drainage.
“Drainage is hard to do and is not necessarily effective,” explained Dr. Grace. While scrubs, warmth, and massage frequently are adequate to unclog the gland – which secretes meibum, a complex of lipids that perform several functions in protecting the eye – therapies specific to the cause, such as Demodex-related blepharitis, chalazions, and styes, might be needed.
Dr. Grace indicated that patience is often needed. The process of unclogging these glands often takes time, but she emphasized that a first-line conservative approach is always appropriate to avoid the difficulty and potential problems of incisions.
In general, these messages are not novel, but they provide a refresher for pediatric dermatologists who do not regularly confront complications that involve the eyes. According to session moderator, Elizabeth Neiman, MD, assistant professor of pediatric dermatology, University of North Carolina at Chapel Hill, the messages regarding topical steroids on the face and the eyes are “important” and worth emphasizing.
“It’s useful to reinforce the point that corticosteroids should be used when needed in the periorbital area [to control skin diseases] if they are used in low concentrations,” Dr. Neiman told this news organization.
Similarly, conjunctivitis and other ocular complications of dupilumab are a source of concern for parents as well as dermatologists. Dr. Neiman indicated that a review of the benefit-to-risk ratio is important when considering these treatments in patients with indications for severe skin disorders.
Dr. Grace and Dr. Nieman have no potential financial conflicts related to this topic.
A version of this article first appeared on Medscape.com.
AT SPD 2023
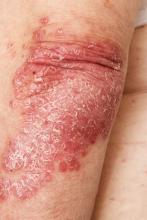
Intensely pruritic rash
The history and findings in this case are consistent with atopic dermatitis (AD).
AD is a chronic inflammatory skin condition that affects more than 200 million people worldwide, including as many as 30% of children and 10% of adults. Although it is more common in children (and may persist into adulthood), approximately 1 in 4 adults with AD have adult-onset disease.
The etiology of AD is complex and includes both genetic and environmental factors, including a weakened skin barrier, immune dysregulation, and abnormalities of the skin microbiome. AD is a member of the atopic triad (ie, AD, allergic rhinoconjunctivitis, and asthma), which may commence concurrently or in succession in what is referred to as the "atopic march."
The presentation of adult-onset AD may differ from that seen in children. For example, the most commonly reported body regions affected in adult-onset AD are the hands, eyelids, neck, and flexural surfaces of the upper limbs. In contrast, childhood-onset AD is less specific to body regions other than flexural areas. Xerosis is a prominent feature, and lichenification may be present. Some patients may have a rippled, brown macular ring around the neck, simulating the pigmentations seen in macular amyloid but due instead to postinflammatory melanin deposition. Pruritus is the most common and bothersome symptom associated with AD; patients may also experience anxiety, depression, and sleep disturbances.
Diminished quality of life, reduced productivity at work and school, and increased healthcare costs (hospitalizations, emergency visits, outpatient visits, and medications) have all been reported in patients with AD. Triggers for flare-ups vary among individuals; commonly reported triggers include physical or emotional stress, changes in temperature or humidity, sweating, allergens, and irritants.
AD is typically diagnosed clinically given the characteristic distribution of lesions in various age groups (infancy, childhood, and adult). Associated findings such as keratosis pilaris may help to facilitate the diagnosis. No biomarker for the diagnosis of AD has been found and laboratory testing is rarely necessary. However, a swab of infected skin may help to isolate a specific involved organism (eg, Staphylococcus or Streptococcus) and antibiotic sensitivity. Allergy and radioallergosorbent testing are not necessary to make the diagnosis. A swab for viral polymerase chain reaction may be beneficial to help identify superinfection with herpes simplex virus and identify a diagnosis of eczema herpeticum. Testing for serum IgE level can also be helpful for supporting the diagnosis of AD.
The management of AD includes trigger avoidance, daily skin care with application of emollients, anti-inflammatory therapy, and other complementary modalities. For mild or moderate AD, first-line treatment consists of topical anti-inflammatory ointments and creams, including topical corticosteroids, which are available in a broad range of potencies. Other topical medications include topical calcineurin inhibitors (tacrolimus and pimecrolimus for patients aged ≥ 2 years), which may be particularly appropriate when there is concern for adverse events secondary to corticosteroid use; topical phosphodiesterase 4 inhibitor (crisaborole ointment for patients aged ≥ 3 months); and topical Janus kinase inhibitor (ruxolitinib cream for patients aged ≥ 12 years).
For patients with moderate to severe AD, or for those who are refractory to topical medications, treatment may include biologic therapy (dupilumab and tralokinumab for patients aged ≥ 6 months and ≥ 18 years, respectively), oral Janus kinase inhibitors (upadacitinib and abrocitinib for patients ages ≥ 12 and ≥ 18 years, respectively), phototherapy (commonly narrow-band ultraviolet light type B treatment), and oral immunomodulators (including methotrexate, mycophenolate, and azathioprine). Combination therapy may be required for the long-term management of more severe AD.
William D. James, MD, Professor, Department of Dermatology, University of Pennsylvania, Philadelphia.
Disclosure: William D. James, MD, has disclosed the following relevant financial relationships:
Received income in an amount equal to or greater than $250 from: Elsevier.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
The history and findings in this case are consistent with atopic dermatitis (AD).
AD is a chronic inflammatory skin condition that affects more than 200 million people worldwide, including as many as 30% of children and 10% of adults. Although it is more common in children (and may persist into adulthood), approximately 1 in 4 adults with AD have adult-onset disease.
The etiology of AD is complex and includes both genetic and environmental factors, including a weakened skin barrier, immune dysregulation, and abnormalities of the skin microbiome. AD is a member of the atopic triad (ie, AD, allergic rhinoconjunctivitis, and asthma), which may commence concurrently or in succession in what is referred to as the "atopic march."
The presentation of adult-onset AD may differ from that seen in children. For example, the most commonly reported body regions affected in adult-onset AD are the hands, eyelids, neck, and flexural surfaces of the upper limbs. In contrast, childhood-onset AD is less specific to body regions other than flexural areas. Xerosis is a prominent feature, and lichenification may be present. Some patients may have a rippled, brown macular ring around the neck, simulating the pigmentations seen in macular amyloid but due instead to postinflammatory melanin deposition. Pruritus is the most common and bothersome symptom associated with AD; patients may also experience anxiety, depression, and sleep disturbances.
Diminished quality of life, reduced productivity at work and school, and increased healthcare costs (hospitalizations, emergency visits, outpatient visits, and medications) have all been reported in patients with AD. Triggers for flare-ups vary among individuals; commonly reported triggers include physical or emotional stress, changes in temperature or humidity, sweating, allergens, and irritants.
AD is typically diagnosed clinically given the characteristic distribution of lesions in various age groups (infancy, childhood, and adult). Associated findings such as keratosis pilaris may help to facilitate the diagnosis. No biomarker for the diagnosis of AD has been found and laboratory testing is rarely necessary. However, a swab of infected skin may help to isolate a specific involved organism (eg, Staphylococcus or Streptococcus) and antibiotic sensitivity. Allergy and radioallergosorbent testing are not necessary to make the diagnosis. A swab for viral polymerase chain reaction may be beneficial to help identify superinfection with herpes simplex virus and identify a diagnosis of eczema herpeticum. Testing for serum IgE level can also be helpful for supporting the diagnosis of AD.
The management of AD includes trigger avoidance, daily skin care with application of emollients, anti-inflammatory therapy, and other complementary modalities. For mild or moderate AD, first-line treatment consists of topical anti-inflammatory ointments and creams, including topical corticosteroids, which are available in a broad range of potencies. Other topical medications include topical calcineurin inhibitors (tacrolimus and pimecrolimus for patients aged ≥ 2 years), which may be particularly appropriate when there is concern for adverse events secondary to corticosteroid use; topical phosphodiesterase 4 inhibitor (crisaborole ointment for patients aged ≥ 3 months); and topical Janus kinase inhibitor (ruxolitinib cream for patients aged ≥ 12 years).
For patients with moderate to severe AD, or for those who are refractory to topical medications, treatment may include biologic therapy (dupilumab and tralokinumab for patients aged ≥ 6 months and ≥ 18 years, respectively), oral Janus kinase inhibitors (upadacitinib and abrocitinib for patients ages ≥ 12 and ≥ 18 years, respectively), phototherapy (commonly narrow-band ultraviolet light type B treatment), and oral immunomodulators (including methotrexate, mycophenolate, and azathioprine). Combination therapy may be required for the long-term management of more severe AD.
William D. James, MD, Professor, Department of Dermatology, University of Pennsylvania, Philadelphia.
Disclosure: William D. James, MD, has disclosed the following relevant financial relationships:
Received income in an amount equal to or greater than $250 from: Elsevier.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
The history and findings in this case are consistent with atopic dermatitis (AD).
AD is a chronic inflammatory skin condition that affects more than 200 million people worldwide, including as many as 30% of children and 10% of adults. Although it is more common in children (and may persist into adulthood), approximately 1 in 4 adults with AD have adult-onset disease.
The etiology of AD is complex and includes both genetic and environmental factors, including a weakened skin barrier, immune dysregulation, and abnormalities of the skin microbiome. AD is a member of the atopic triad (ie, AD, allergic rhinoconjunctivitis, and asthma), which may commence concurrently or in succession in what is referred to as the "atopic march."
The presentation of adult-onset AD may differ from that seen in children. For example, the most commonly reported body regions affected in adult-onset AD are the hands, eyelids, neck, and flexural surfaces of the upper limbs. In contrast, childhood-onset AD is less specific to body regions other than flexural areas. Xerosis is a prominent feature, and lichenification may be present. Some patients may have a rippled, brown macular ring around the neck, simulating the pigmentations seen in macular amyloid but due instead to postinflammatory melanin deposition. Pruritus is the most common and bothersome symptom associated with AD; patients may also experience anxiety, depression, and sleep disturbances.
Diminished quality of life, reduced productivity at work and school, and increased healthcare costs (hospitalizations, emergency visits, outpatient visits, and medications) have all been reported in patients with AD. Triggers for flare-ups vary among individuals; commonly reported triggers include physical or emotional stress, changes in temperature or humidity, sweating, allergens, and irritants.
AD is typically diagnosed clinically given the characteristic distribution of lesions in various age groups (infancy, childhood, and adult). Associated findings such as keratosis pilaris may help to facilitate the diagnosis. No biomarker for the diagnosis of AD has been found and laboratory testing is rarely necessary. However, a swab of infected skin may help to isolate a specific involved organism (eg, Staphylococcus or Streptococcus) and antibiotic sensitivity. Allergy and radioallergosorbent testing are not necessary to make the diagnosis. A swab for viral polymerase chain reaction may be beneficial to help identify superinfection with herpes simplex virus and identify a diagnosis of eczema herpeticum. Testing for serum IgE level can also be helpful for supporting the diagnosis of AD.
The management of AD includes trigger avoidance, daily skin care with application of emollients, anti-inflammatory therapy, and other complementary modalities. For mild or moderate AD, first-line treatment consists of topical anti-inflammatory ointments and creams, including topical corticosteroids, which are available in a broad range of potencies. Other topical medications include topical calcineurin inhibitors (tacrolimus and pimecrolimus for patients aged ≥ 2 years), which may be particularly appropriate when there is concern for adverse events secondary to corticosteroid use; topical phosphodiesterase 4 inhibitor (crisaborole ointment for patients aged ≥ 3 months); and topical Janus kinase inhibitor (ruxolitinib cream for patients aged ≥ 12 years).
For patients with moderate to severe AD, or for those who are refractory to topical medications, treatment may include biologic therapy (dupilumab and tralokinumab for patients aged ≥ 6 months and ≥ 18 years, respectively), oral Janus kinase inhibitors (upadacitinib and abrocitinib for patients ages ≥ 12 and ≥ 18 years, respectively), phototherapy (commonly narrow-band ultraviolet light type B treatment), and oral immunomodulators (including methotrexate, mycophenolate, and azathioprine). Combination therapy may be required for the long-term management of more severe AD.
William D. James, MD, Professor, Department of Dermatology, University of Pennsylvania, Philadelphia.
Disclosure: William D. James, MD, has disclosed the following relevant financial relationships:
Received income in an amount equal to or greater than $250 from: Elsevier.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
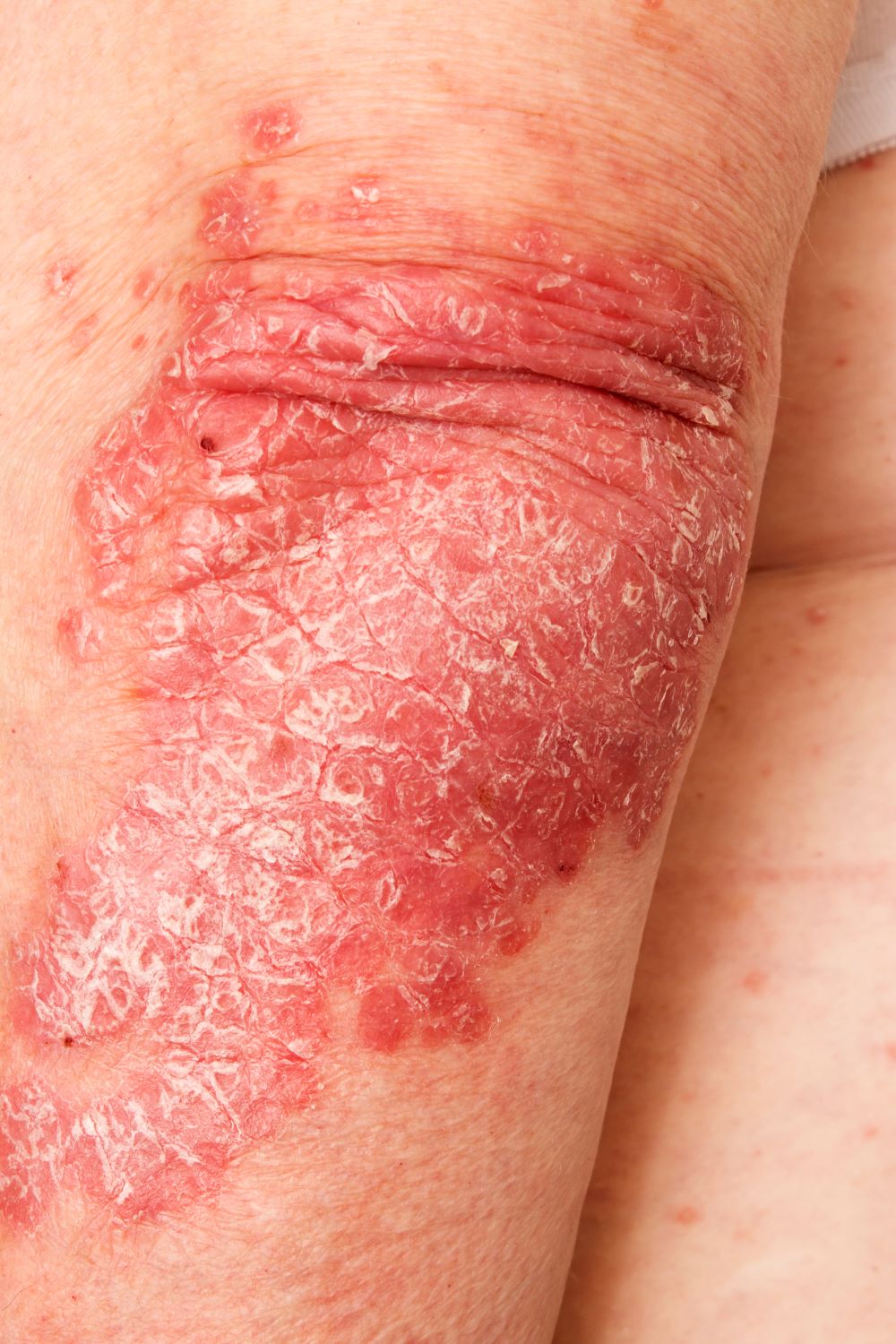
A 52-year-old woman presents with complaints of an itchy rash on her arms, legs, neck, and eyelids. She reports having flares with a similar eruption on her arms and legs over the past 2 years, but on previous occasions she was able to manage it with topical emollients. Over the past 6 months, however, it has worsened both in intensity and spread. She describes the rash as intensely pruritic, and now that it has become more visible, she reports feeling embarrassed by it at work and during social outings. The itch is also disrupting her sleep. The patient states that she is undergoing an extremely stressful period in her life because of her parents' declining health and a recent separation from her husband.
Approximately 3 months ago, she visited her primary care provider, who diagnosed her with an allergic rash and prescribed a course of an oral glucocorticoid. Initially, she thought the treatment worked, but the rash soon recurred after she finished her treatment.
Physical examination reveals scaly, crusted hyperpigmented lesions involving the arms, flexural areas of the elbows and knees, neck, and eyelids. Lichenification and xerosis are observed. There is no evidence of conjunctivitis or scalp involvement. The turbinates are not inflamed. Complete blood count findings are within normal range. The patient is 5 ft 3 in and weighs 125 lb (BMI 22.1) and is a nonsmoker.
Camp Discovery: A place for children to be comfortable in their own skin
The talent show, the grand finale of the 1-week camp, was nearly 7 years ago, but Emily Haygood of Houston, now 17 and about to start her senior year, remembers it in detail. She sang “Death of a Bachelor,” an R&B pop song and Billboard No. 1 hit at the time about a former bachelor who had happily married. These days, she said, if she watched the video of her 10-year-old singing self, “I would probably throw up.” But she still treasures the audience response, “having all those people I’d gotten close to cheer for me.”
Emily was at , but share one feature: they are the kind of dermatologic issues that can make doing everyday kid or teen activities like swimming difficult and can elicit mean comments from classmates and other would-be friends.

Emily was first diagnosed with atopic dermatitis at age 4, her mother, Amber Haygood, says. By age 9, it had become severe. Emily remembers being teased some in elementary school. “I did feel bad a lot of the time, when asked insensitive questions.” Her mother still bristles that adults often could be cruel, too.
But at Camp Discovery, those issues were nonexistent. “Camp was so cool,” Emily said. Besides the usual camp activities, it had things that “normal” camp didn’t, like other kids who didn’t stare at your skin condition or make fun of it.
30th anniversary season begins
This year is the 30th anniversary of Camp Discovery. Sessions began July 23 and continue through Aug. 18, with locations in Crosslake, Minn.; Hebron, Conn.; and Millville, Pa., in addition to Burton, Tex. About 300 campers will attend this year, according to the AAD, and 6,151 campers have attended from 1993 to 2022.
The 1-week camp accepts youth with conditions ranging from eczema and psoriasis to vitiligo, alopecia, epidermolysis bullosa, and ichthyosis, according to the academy. A dermatologist first refers a child, downloading and completing the referral form and sending it to the academy.
The 1-week session, including travel, is free for the campers, thanks to donors. As a nonprofit and membership-based organization, the AAD does not release the detailed financial information about the operating budget for the camp. Dermatologists, nurses, and counselors volunteer their time.
In his presidential address at the AAD’s annual meeting in March, outgoing president Mark D. Kaufmann, MD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai in New York, referred to camp volunteering as an antidote to professional burnout. Remembering why as a dermatologist one entered the profession can be one solution, he said, and described his own recent 3-day volunteer stint at the camp.
“Those 3 magical days, being with kids as they discovered they weren’t alone in the world, sharing their experiences and ideas, reminded me why I became a physician in the first place,” he told the audience of meeting attendees. He vowed to expand the program, with a goal of having every dermatology resident attend Camp Discovery.
Mental health effects of skin conditions
Much research has focused on the mental health fallout from living with chronic skin conditions, and even young children can be adversely affected. In one review of the literature, researchers concluded that pediatric skin disease, including acne, atopic dermatitis, and psoriasis, can affect quality of life, carry stigma, and lead to bullying and eventually even suicidal behavior. Another study, published earlier this year, found that atopic dermatitis affected children’s quality of life, impacting sleep and leading to feelings of being ashamed.
“It’s not necessarily about what their skin condition is and more about the psychosocial impact,’’ said Samantha Hill, MD, a pediatric and general dermatologist in Lynchburg, Va., who is the medical director of Camp Discovery in Minnesota this year.
Camp activities, reactions
The overriding theme of camp is allowing all the youth to be “just one of the kids at camp,” Dr. Hill said in an interview. “They come to do all kinds of things they don’t do in normal life because people don’t give them the credit to [be able to] do it.”
Every year, she said, “I tell my staff we are in the business of making things happen, so if there is a kid bandaged head to toe [because of a skin condition] and they want to go tubing and get in the lake, we figure out how to make it happen. We have done that multiple times.”
Newcomers are initially nervous, Dr. Hill acknowledged, but in time let their guard down. Returnees are a different story. “When kids who have been at camp before arrive, you can see them start breathing again, looking for their friends. You can see them relax right before your eyes.”
“The single most empowering thing is the realization you are not alone,” said Meena Julapalli, MD, a Houston dermatologist who is a medical team member and long-time volunteer at Camp Discovery. That, she said, and “You get to be a kid, and you don’t have to have people staring at you.”
Dr. Julapalli remembers one of her patients with keratitis-ichthyosis-deafness (KID) syndrome. “She needed more than what I could offer,” she said. “She needed camp.” At camp, the organizers found a counselor who knew sign language to accompany her. At first, she was quiet and didn’t smile much. By the end of the week, as she was about to observe her birthday, things changed. After breakfast, she was led to the stage, where fellow campers began singing – and signing the song they had just learned.
Camp staff gets it
Allyson Garin, who was diagnosed with vitiligo at age 6 months, is a camp program director at Camp Discovery in Crosslake, Minn. She first went to camp in 1990 at age 11, returning until she “aged out” at 16, then worked as a counselor. She gets it when campers tell her they hear rude comments about their skin conditions.
“I remember being in swimming pools, in lines at fairgrounds or amusement parks,” she said in an interview, “and hearing people say, ‘Don’t touch her,’ ’’ fearing contagion, perhaps. “People would make jokes about cows, since they are spotted,” she said, or people would simply step back.
All those years ago, her mother found out about the camp and decided to figure out how to get her there. She got there, and she met a fellow camper with vitiligo, and they became pen pals. “We still talk,” she said.
Meeting someone with the same skin condition, she said, isn’t just about commiserating. “There is a lot of information sharing,” on topics such as best treatments, strategies, and other conversations.
Other lessons
While campers can feel comfortable around others who also have skin conditions, and understand, the lesson extends beyond that, Ms. Garin said. “It gave me a perspective,” she said of her camp experience. “I always felt, ‘Woe is me.’ ” But when she met others with, as she said, conditions “way worse than vitiligo, it really grounds you.”
Dr. Hill agreed. Campers get the benefit of others accepting and including them, but also practicing that same attitude toward fellow campers, she said. “It insures that we are providing this environment of inclusion, but that they are practicing it as well. They need to practice it like everyone else.”
Getting parents on board
The idea of camp, especially for those at the younger end of the 8- to 16-years age range accepted for Camp Discovery, can take some getting used to for some parents. Ms. Haygood, Emily’s mother, relates to that. Her daughter’s dermatologist at the time, who is now retired, had first suggested the camp. Her first reaction? “I am not sending my chronically ill child to camp with strangers.” She also acknowledged that she, like other parents of children with a chronic illness, can be a helicopter parent.
Then, she noticed that Emily seemed interested, so she got more information, finding out that it was staffed by doctors. It all sounded good, she said, and the social interaction, she knew, would be beneficial. “Then my husband was a no,” she said, concerned about their daughter being with strangers. “Eventually he came around,” Ms. Haygood said. All along, Emily said, “it seemed fun. I was probably trying to talk them into it.” She admits she was very nervous at first, but calmed down when she realized her own dermatologist was going to be there.
Vanessa Hadley of Spring, Tex., was on board the moment she heard about Camp Discovery. “I just thought it was amazing,” she said. Her daughter Isabelle, 13, has been to the camp. “She has alopecia areata and severe eczema,” Ms. Hadley said. Now, Isabelle is returning to camp and coaching her sister Penelope, 8, who has eczema and mild alopecia and is a first-timer this summer.
One tip the 8-year-old has learned so far: Turn to your counselor for support if you’re nervous. That worked, Isabelle said, the first year when she was wary of the zipline – then surprised herself and conquered it.
Dr. Hill and Dr. Julapalli have no disclosures.
The talent show, the grand finale of the 1-week camp, was nearly 7 years ago, but Emily Haygood of Houston, now 17 and about to start her senior year, remembers it in detail. She sang “Death of a Bachelor,” an R&B pop song and Billboard No. 1 hit at the time about a former bachelor who had happily married. These days, she said, if she watched the video of her 10-year-old singing self, “I would probably throw up.” But she still treasures the audience response, “having all those people I’d gotten close to cheer for me.”
Emily was at , but share one feature: they are the kind of dermatologic issues that can make doing everyday kid or teen activities like swimming difficult and can elicit mean comments from classmates and other would-be friends.

Emily was first diagnosed with atopic dermatitis at age 4, her mother, Amber Haygood, says. By age 9, it had become severe. Emily remembers being teased some in elementary school. “I did feel bad a lot of the time, when asked insensitive questions.” Her mother still bristles that adults often could be cruel, too.
But at Camp Discovery, those issues were nonexistent. “Camp was so cool,” Emily said. Besides the usual camp activities, it had things that “normal” camp didn’t, like other kids who didn’t stare at your skin condition or make fun of it.
30th anniversary season begins
This year is the 30th anniversary of Camp Discovery. Sessions began July 23 and continue through Aug. 18, with locations in Crosslake, Minn.; Hebron, Conn.; and Millville, Pa., in addition to Burton, Tex. About 300 campers will attend this year, according to the AAD, and 6,151 campers have attended from 1993 to 2022.
The 1-week camp accepts youth with conditions ranging from eczema and psoriasis to vitiligo, alopecia, epidermolysis bullosa, and ichthyosis, according to the academy. A dermatologist first refers a child, downloading and completing the referral form and sending it to the academy.
The 1-week session, including travel, is free for the campers, thanks to donors. As a nonprofit and membership-based organization, the AAD does not release the detailed financial information about the operating budget for the camp. Dermatologists, nurses, and counselors volunteer their time.
In his presidential address at the AAD’s annual meeting in March, outgoing president Mark D. Kaufmann, MD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai in New York, referred to camp volunteering as an antidote to professional burnout. Remembering why as a dermatologist one entered the profession can be one solution, he said, and described his own recent 3-day volunteer stint at the camp.
“Those 3 magical days, being with kids as they discovered they weren’t alone in the world, sharing their experiences and ideas, reminded me why I became a physician in the first place,” he told the audience of meeting attendees. He vowed to expand the program, with a goal of having every dermatology resident attend Camp Discovery.
Mental health effects of skin conditions
Much research has focused on the mental health fallout from living with chronic skin conditions, and even young children can be adversely affected. In one review of the literature, researchers concluded that pediatric skin disease, including acne, atopic dermatitis, and psoriasis, can affect quality of life, carry stigma, and lead to bullying and eventually even suicidal behavior. Another study, published earlier this year, found that atopic dermatitis affected children’s quality of life, impacting sleep and leading to feelings of being ashamed.
“It’s not necessarily about what their skin condition is and more about the psychosocial impact,’’ said Samantha Hill, MD, a pediatric and general dermatologist in Lynchburg, Va., who is the medical director of Camp Discovery in Minnesota this year.
Camp activities, reactions
The overriding theme of camp is allowing all the youth to be “just one of the kids at camp,” Dr. Hill said in an interview. “They come to do all kinds of things they don’t do in normal life because people don’t give them the credit to [be able to] do it.”
Every year, she said, “I tell my staff we are in the business of making things happen, so if there is a kid bandaged head to toe [because of a skin condition] and they want to go tubing and get in the lake, we figure out how to make it happen. We have done that multiple times.”
Newcomers are initially nervous, Dr. Hill acknowledged, but in time let their guard down. Returnees are a different story. “When kids who have been at camp before arrive, you can see them start breathing again, looking for their friends. You can see them relax right before your eyes.”
“The single most empowering thing is the realization you are not alone,” said Meena Julapalli, MD, a Houston dermatologist who is a medical team member and long-time volunteer at Camp Discovery. That, she said, and “You get to be a kid, and you don’t have to have people staring at you.”
Dr. Julapalli remembers one of her patients with keratitis-ichthyosis-deafness (KID) syndrome. “She needed more than what I could offer,” she said. “She needed camp.” At camp, the organizers found a counselor who knew sign language to accompany her. At first, she was quiet and didn’t smile much. By the end of the week, as she was about to observe her birthday, things changed. After breakfast, she was led to the stage, where fellow campers began singing – and signing the song they had just learned.
Camp staff gets it
Allyson Garin, who was diagnosed with vitiligo at age 6 months, is a camp program director at Camp Discovery in Crosslake, Minn. She first went to camp in 1990 at age 11, returning until she “aged out” at 16, then worked as a counselor. She gets it when campers tell her they hear rude comments about their skin conditions.
“I remember being in swimming pools, in lines at fairgrounds or amusement parks,” she said in an interview, “and hearing people say, ‘Don’t touch her,’ ’’ fearing contagion, perhaps. “People would make jokes about cows, since they are spotted,” she said, or people would simply step back.
All those years ago, her mother found out about the camp and decided to figure out how to get her there. She got there, and she met a fellow camper with vitiligo, and they became pen pals. “We still talk,” she said.
Meeting someone with the same skin condition, she said, isn’t just about commiserating. “There is a lot of information sharing,” on topics such as best treatments, strategies, and other conversations.
Other lessons
While campers can feel comfortable around others who also have skin conditions, and understand, the lesson extends beyond that, Ms. Garin said. “It gave me a perspective,” she said of her camp experience. “I always felt, ‘Woe is me.’ ” But when she met others with, as she said, conditions “way worse than vitiligo, it really grounds you.”
Dr. Hill agreed. Campers get the benefit of others accepting and including them, but also practicing that same attitude toward fellow campers, she said. “It insures that we are providing this environment of inclusion, but that they are practicing it as well. They need to practice it like everyone else.”
Getting parents on board
The idea of camp, especially for those at the younger end of the 8- to 16-years age range accepted for Camp Discovery, can take some getting used to for some parents. Ms. Haygood, Emily’s mother, relates to that. Her daughter’s dermatologist at the time, who is now retired, had first suggested the camp. Her first reaction? “I am not sending my chronically ill child to camp with strangers.” She also acknowledged that she, like other parents of children with a chronic illness, can be a helicopter parent.
Then, she noticed that Emily seemed interested, so she got more information, finding out that it was staffed by doctors. It all sounded good, she said, and the social interaction, she knew, would be beneficial. “Then my husband was a no,” she said, concerned about their daughter being with strangers. “Eventually he came around,” Ms. Haygood said. All along, Emily said, “it seemed fun. I was probably trying to talk them into it.” She admits she was very nervous at first, but calmed down when she realized her own dermatologist was going to be there.
Vanessa Hadley of Spring, Tex., was on board the moment she heard about Camp Discovery. “I just thought it was amazing,” she said. Her daughter Isabelle, 13, has been to the camp. “She has alopecia areata and severe eczema,” Ms. Hadley said. Now, Isabelle is returning to camp and coaching her sister Penelope, 8, who has eczema and mild alopecia and is a first-timer this summer.
One tip the 8-year-old has learned so far: Turn to your counselor for support if you’re nervous. That worked, Isabelle said, the first year when she was wary of the zipline – then surprised herself and conquered it.
Dr. Hill and Dr. Julapalli have no disclosures.
The talent show, the grand finale of the 1-week camp, was nearly 7 years ago, but Emily Haygood of Houston, now 17 and about to start her senior year, remembers it in detail. She sang “Death of a Bachelor,” an R&B pop song and Billboard No. 1 hit at the time about a former bachelor who had happily married. These days, she said, if she watched the video of her 10-year-old singing self, “I would probably throw up.” But she still treasures the audience response, “having all those people I’d gotten close to cheer for me.”
Emily was at , but share one feature: they are the kind of dermatologic issues that can make doing everyday kid or teen activities like swimming difficult and can elicit mean comments from classmates and other would-be friends.

Emily was first diagnosed with atopic dermatitis at age 4, her mother, Amber Haygood, says. By age 9, it had become severe. Emily remembers being teased some in elementary school. “I did feel bad a lot of the time, when asked insensitive questions.” Her mother still bristles that adults often could be cruel, too.
But at Camp Discovery, those issues were nonexistent. “Camp was so cool,” Emily said. Besides the usual camp activities, it had things that “normal” camp didn’t, like other kids who didn’t stare at your skin condition or make fun of it.
30th anniversary season begins
This year is the 30th anniversary of Camp Discovery. Sessions began July 23 and continue through Aug. 18, with locations in Crosslake, Minn.; Hebron, Conn.; and Millville, Pa., in addition to Burton, Tex. About 300 campers will attend this year, according to the AAD, and 6,151 campers have attended from 1993 to 2022.
The 1-week camp accepts youth with conditions ranging from eczema and psoriasis to vitiligo, alopecia, epidermolysis bullosa, and ichthyosis, according to the academy. A dermatologist first refers a child, downloading and completing the referral form and sending it to the academy.
The 1-week session, including travel, is free for the campers, thanks to donors. As a nonprofit and membership-based organization, the AAD does not release the detailed financial information about the operating budget for the camp. Dermatologists, nurses, and counselors volunteer their time.
In his presidential address at the AAD’s annual meeting in March, outgoing president Mark D. Kaufmann, MD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai in New York, referred to camp volunteering as an antidote to professional burnout. Remembering why as a dermatologist one entered the profession can be one solution, he said, and described his own recent 3-day volunteer stint at the camp.
“Those 3 magical days, being with kids as they discovered they weren’t alone in the world, sharing their experiences and ideas, reminded me why I became a physician in the first place,” he told the audience of meeting attendees. He vowed to expand the program, with a goal of having every dermatology resident attend Camp Discovery.
Mental health effects of skin conditions
Much research has focused on the mental health fallout from living with chronic skin conditions, and even young children can be adversely affected. In one review of the literature, researchers concluded that pediatric skin disease, including acne, atopic dermatitis, and psoriasis, can affect quality of life, carry stigma, and lead to bullying and eventually even suicidal behavior. Another study, published earlier this year, found that atopic dermatitis affected children’s quality of life, impacting sleep and leading to feelings of being ashamed.
“It’s not necessarily about what their skin condition is and more about the psychosocial impact,’’ said Samantha Hill, MD, a pediatric and general dermatologist in Lynchburg, Va., who is the medical director of Camp Discovery in Minnesota this year.
Camp activities, reactions
The overriding theme of camp is allowing all the youth to be “just one of the kids at camp,” Dr. Hill said in an interview. “They come to do all kinds of things they don’t do in normal life because people don’t give them the credit to [be able to] do it.”
Every year, she said, “I tell my staff we are in the business of making things happen, so if there is a kid bandaged head to toe [because of a skin condition] and they want to go tubing and get in the lake, we figure out how to make it happen. We have done that multiple times.”
Newcomers are initially nervous, Dr. Hill acknowledged, but in time let their guard down. Returnees are a different story. “When kids who have been at camp before arrive, you can see them start breathing again, looking for their friends. You can see them relax right before your eyes.”
“The single most empowering thing is the realization you are not alone,” said Meena Julapalli, MD, a Houston dermatologist who is a medical team member and long-time volunteer at Camp Discovery. That, she said, and “You get to be a kid, and you don’t have to have people staring at you.”
Dr. Julapalli remembers one of her patients with keratitis-ichthyosis-deafness (KID) syndrome. “She needed more than what I could offer,” she said. “She needed camp.” At camp, the organizers found a counselor who knew sign language to accompany her. At first, she was quiet and didn’t smile much. By the end of the week, as she was about to observe her birthday, things changed. After breakfast, she was led to the stage, where fellow campers began singing – and signing the song they had just learned.
Camp staff gets it
Allyson Garin, who was diagnosed with vitiligo at age 6 months, is a camp program director at Camp Discovery in Crosslake, Minn. She first went to camp in 1990 at age 11, returning until she “aged out” at 16, then worked as a counselor. She gets it when campers tell her they hear rude comments about their skin conditions.
“I remember being in swimming pools, in lines at fairgrounds or amusement parks,” she said in an interview, “and hearing people say, ‘Don’t touch her,’ ’’ fearing contagion, perhaps. “People would make jokes about cows, since they are spotted,” she said, or people would simply step back.
All those years ago, her mother found out about the camp and decided to figure out how to get her there. She got there, and she met a fellow camper with vitiligo, and they became pen pals. “We still talk,” she said.
Meeting someone with the same skin condition, she said, isn’t just about commiserating. “There is a lot of information sharing,” on topics such as best treatments, strategies, and other conversations.
Other lessons
While campers can feel comfortable around others who also have skin conditions, and understand, the lesson extends beyond that, Ms. Garin said. “It gave me a perspective,” she said of her camp experience. “I always felt, ‘Woe is me.’ ” But when she met others with, as she said, conditions “way worse than vitiligo, it really grounds you.”
Dr. Hill agreed. Campers get the benefit of others accepting and including them, but also practicing that same attitude toward fellow campers, she said. “It insures that we are providing this environment of inclusion, but that they are practicing it as well. They need to practice it like everyone else.”
Getting parents on board
The idea of camp, especially for those at the younger end of the 8- to 16-years age range accepted for Camp Discovery, can take some getting used to for some parents. Ms. Haygood, Emily’s mother, relates to that. Her daughter’s dermatologist at the time, who is now retired, had first suggested the camp. Her first reaction? “I am not sending my chronically ill child to camp with strangers.” She also acknowledged that she, like other parents of children with a chronic illness, can be a helicopter parent.
Then, she noticed that Emily seemed interested, so she got more information, finding out that it was staffed by doctors. It all sounded good, she said, and the social interaction, she knew, would be beneficial. “Then my husband was a no,” she said, concerned about their daughter being with strangers. “Eventually he came around,” Ms. Haygood said. All along, Emily said, “it seemed fun. I was probably trying to talk them into it.” She admits she was very nervous at first, but calmed down when she realized her own dermatologist was going to be there.
Vanessa Hadley of Spring, Tex., was on board the moment she heard about Camp Discovery. “I just thought it was amazing,” she said. Her daughter Isabelle, 13, has been to the camp. “She has alopecia areata and severe eczema,” Ms. Hadley said. Now, Isabelle is returning to camp and coaching her sister Penelope, 8, who has eczema and mild alopecia and is a first-timer this summer.
One tip the 8-year-old has learned so far: Turn to your counselor for support if you’re nervous. That worked, Isabelle said, the first year when she was wary of the zipline – then surprised herself and conquered it.
Dr. Hill and Dr. Julapalli have no disclosures.
JAK inhibitors efficacious for atopic dermatitis in Asian patients, study finds
SINGAPORE – conducted in Singapore has found.
“Abrocitinib and upadacitinib surprisingly appeared to have better treatment efficacy compared to baricitinib,” said study lead Yik Weng Yew, MD, PhD, MPH, deputy head of research at Singapore’s National Skin Centre (NSC), who presented the results at the 25th World Congress of Dermatology. “But overall, as a group, I think they show a very good treatment response, as well as a good effect on itch response.”
JAK inhibitors are used to treat a variety of inflammatory diseases including alopecia areata, rheumatoid arthritis, and inflammatory bowel disease. Although treatment for severe eczema was previously limited to topical steroids and oral immunosuppressants, there are now two oral JAK inhibitors – abrocitinib and upadacitinib – approved in 2022 by the Food and Drug Administration for treating AD, which affects up to 2.4% of the global population. (A topical formulation of ruxolitinib, a JAK inhibitor, was approved for AD in 2021.)
The Singapore study is one of the few that have examined the safety and efficacy of JAK inhibitors for treatment of AD in a non-White population.
Chinese population
For the 12-week trial, conducted in 2022, Dr. Yew and associates recruited 35 patients from the NSC. More than half of participants (64%) were men and most (96%) were of Chinese ethnicity. Four of every five patients had previously received systemic agents: 17% had been treated with one systemic agent, 18.9% with two, 15.1% with three, 22.6% with four, and 3.8% with five. The most commonly used agents were cyclosporine (62.3%), methotrexate (47.2%), azathioprine (39.6%), and dupilumab (35.8%).
“The switch in therapy could have been a result of inadequate efficacy or cost reasons because in Singapore patients pay out of pocket for AD treatments,” said Dr. Yew.
Additionally, he offered a caveat on the profile of participants: “Perhaps they were more difficult atopic eczema patients, and therefore, the efficacy [of JAK inhibitors] might be a bit different.”
Clearer skin, less itch
Patients received one of the three study drugs: baricitinib (66%), abrocitinib (21%), and upadacitinib (13%). The distribution was “affected by reimbursement patterns and availability of the drug,” explained Dr. Yew.
They were assessed at weeks 4 and 12. By study end, the proportion of patients who self-reported an improvement in their condition was 100% for upadacitinib, 90% for abrocitinib, and 69% for baricitinib.
Scores on the Investigator Global Assessment (IGA) also improved with treatment. Patients in the baricitinib group saw their mean score fall from 4.0 to 3.0 by week 4, then to 2.0 by week 12. With upadacitinib and abrocitinib, “you can see that there is a nice decrease in IGA responses,” said Dr. Yew, referring to the larger improvement in scores experienced by patients on those two treatments. For patients on upadacitinib, IGA decreased from 3.5 to 2 at 4 weeks, then to 0.5 at 12 weeks, while those taking abrocitinib had their scores drop from 4.0 to 2.0 at 4 weeks, then to 1.0 at 12 weeks.
When it came to itch reduction, the abrocitinib group experienced the biggest reduction, with a median reduction of 5.5 points in itch score. Median reduction in itch score was 4 points for the other two groups. “Oral JAK inhibitors appear to have a good effect on itch response,” said Dr. Yew.
However, the researchers observed no significant reduction in percentage of body surface area affected, the last outcome assessed.
The most commonly reported adverse events were increased creatine kinase levels (11.3% of patients), increased LDL cholesterol levels (9.4%), and herpes zoster (9.4%). Those in the abrocitinib reported a higher number of these adverse events, compared with the other two treatment groups. (There were no herpes zoster cases among those taking baricitinib.)
For herpes zoster, Dr. Yew said “the common recommendation” is to give the inactivated shingles vaccine. “But the problem is that, number one, these patients would have probably failed multiple agents so they probably can’t wait for you to vaccinate before you initiate treatment.”
In addition, people in Singapore have to pay out-of-pocket for the two vaccine doses, “which is probably a month’s worth of medication,” he noted. “So we have a lot of resistance from patients.”
Additionally, Dr. Yew noted that contrary to what has previously been reported in the literature, there were few complaints of acne as a side effect in the Singaporean study population.
Toward greater representation
Dr. Yew pointed out that the study was limited by a few factors: neither the Eczema Area and Severity Index or Scoring of Atopic Dermatitis index data was used, and the study population was small and not representative of the real world.
Still, the new findings contribute to the overall safety and efficacy profile of JAK inhibitors in AD, which has so far been scarce in non-White populations.
“In Western studies, unfortunately, the representation of the population of skin of color or different ethnicities is underrepresented,” said Yousef Binamer, MD, chair of the dermatology department at King Faisal Specialist Hospital, Riyadh, Saudi Arabia, when approached for an independent comment on the results.
“This is now why researchers are looking into specific groups to study them,” which he pointed out, is crucial because “the immunophenotyping of AD is different for each background.”
The incidence and severity of AD tend to be higher in Asian and Middle Eastern populations, for instance, he noted. “It’s very common in Asia, and not so common in very white skin. I did my training in Canada so I see the difference,” said Dr. Binamer. “Asian people tend to be more itchy and have a tendency to scar on pigmentation.” Whereas White people “usually do not have this issue.”
“So I think real-world evidence of JAK inhibitors in the other populations is important,” he said. Studies such as the one conducted in Singapore, as well as the recently reported QUARTZ3 study, which examined the use of the JAK inhibitor ivarmacitinib in 256 Chinese patients with AD, are helping to pave the way.
The study was independently supported. Dr. Yew and Dr. Binamer have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SINGAPORE – conducted in Singapore has found.
“Abrocitinib and upadacitinib surprisingly appeared to have better treatment efficacy compared to baricitinib,” said study lead Yik Weng Yew, MD, PhD, MPH, deputy head of research at Singapore’s National Skin Centre (NSC), who presented the results at the 25th World Congress of Dermatology. “But overall, as a group, I think they show a very good treatment response, as well as a good effect on itch response.”
JAK inhibitors are used to treat a variety of inflammatory diseases including alopecia areata, rheumatoid arthritis, and inflammatory bowel disease. Although treatment for severe eczema was previously limited to topical steroids and oral immunosuppressants, there are now two oral JAK inhibitors – abrocitinib and upadacitinib – approved in 2022 by the Food and Drug Administration for treating AD, which affects up to 2.4% of the global population. (A topical formulation of ruxolitinib, a JAK inhibitor, was approved for AD in 2021.)
The Singapore study is one of the few that have examined the safety and efficacy of JAK inhibitors for treatment of AD in a non-White population.
Chinese population
For the 12-week trial, conducted in 2022, Dr. Yew and associates recruited 35 patients from the NSC. More than half of participants (64%) were men and most (96%) were of Chinese ethnicity. Four of every five patients had previously received systemic agents: 17% had been treated with one systemic agent, 18.9% with two, 15.1% with three, 22.6% with four, and 3.8% with five. The most commonly used agents were cyclosporine (62.3%), methotrexate (47.2%), azathioprine (39.6%), and dupilumab (35.8%).
“The switch in therapy could have been a result of inadequate efficacy or cost reasons because in Singapore patients pay out of pocket for AD treatments,” said Dr. Yew.
Additionally, he offered a caveat on the profile of participants: “Perhaps they were more difficult atopic eczema patients, and therefore, the efficacy [of JAK inhibitors] might be a bit different.”
Clearer skin, less itch
Patients received one of the three study drugs: baricitinib (66%), abrocitinib (21%), and upadacitinib (13%). The distribution was “affected by reimbursement patterns and availability of the drug,” explained Dr. Yew.
They were assessed at weeks 4 and 12. By study end, the proportion of patients who self-reported an improvement in their condition was 100% for upadacitinib, 90% for abrocitinib, and 69% for baricitinib.
Scores on the Investigator Global Assessment (IGA) also improved with treatment. Patients in the baricitinib group saw their mean score fall from 4.0 to 3.0 by week 4, then to 2.0 by week 12. With upadacitinib and abrocitinib, “you can see that there is a nice decrease in IGA responses,” said Dr. Yew, referring to the larger improvement in scores experienced by patients on those two treatments. For patients on upadacitinib, IGA decreased from 3.5 to 2 at 4 weeks, then to 0.5 at 12 weeks, while those taking abrocitinib had their scores drop from 4.0 to 2.0 at 4 weeks, then to 1.0 at 12 weeks.
When it came to itch reduction, the abrocitinib group experienced the biggest reduction, with a median reduction of 5.5 points in itch score. Median reduction in itch score was 4 points for the other two groups. “Oral JAK inhibitors appear to have a good effect on itch response,” said Dr. Yew.
However, the researchers observed no significant reduction in percentage of body surface area affected, the last outcome assessed.
The most commonly reported adverse events were increased creatine kinase levels (11.3% of patients), increased LDL cholesterol levels (9.4%), and herpes zoster (9.4%). Those in the abrocitinib reported a higher number of these adverse events, compared with the other two treatment groups. (There were no herpes zoster cases among those taking baricitinib.)
For herpes zoster, Dr. Yew said “the common recommendation” is to give the inactivated shingles vaccine. “But the problem is that, number one, these patients would have probably failed multiple agents so they probably can’t wait for you to vaccinate before you initiate treatment.”
In addition, people in Singapore have to pay out-of-pocket for the two vaccine doses, “which is probably a month’s worth of medication,” he noted. “So we have a lot of resistance from patients.”
Additionally, Dr. Yew noted that contrary to what has previously been reported in the literature, there were few complaints of acne as a side effect in the Singaporean study population.
Toward greater representation
Dr. Yew pointed out that the study was limited by a few factors: neither the Eczema Area and Severity Index or Scoring of Atopic Dermatitis index data was used, and the study population was small and not representative of the real world.
Still, the new findings contribute to the overall safety and efficacy profile of JAK inhibitors in AD, which has so far been scarce in non-White populations.
“In Western studies, unfortunately, the representation of the population of skin of color or different ethnicities is underrepresented,” said Yousef Binamer, MD, chair of the dermatology department at King Faisal Specialist Hospital, Riyadh, Saudi Arabia, when approached for an independent comment on the results.
“This is now why researchers are looking into specific groups to study them,” which he pointed out, is crucial because “the immunophenotyping of AD is different for each background.”
The incidence and severity of AD tend to be higher in Asian and Middle Eastern populations, for instance, he noted. “It’s very common in Asia, and not so common in very white skin. I did my training in Canada so I see the difference,” said Dr. Binamer. “Asian people tend to be more itchy and have a tendency to scar on pigmentation.” Whereas White people “usually do not have this issue.”
“So I think real-world evidence of JAK inhibitors in the other populations is important,” he said. Studies such as the one conducted in Singapore, as well as the recently reported QUARTZ3 study, which examined the use of the JAK inhibitor ivarmacitinib in 256 Chinese patients with AD, are helping to pave the way.
The study was independently supported. Dr. Yew and Dr. Binamer have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SINGAPORE – conducted in Singapore has found.
“Abrocitinib and upadacitinib surprisingly appeared to have better treatment efficacy compared to baricitinib,” said study lead Yik Weng Yew, MD, PhD, MPH, deputy head of research at Singapore’s National Skin Centre (NSC), who presented the results at the 25th World Congress of Dermatology. “But overall, as a group, I think they show a very good treatment response, as well as a good effect on itch response.”
JAK inhibitors are used to treat a variety of inflammatory diseases including alopecia areata, rheumatoid arthritis, and inflammatory bowel disease. Although treatment for severe eczema was previously limited to topical steroids and oral immunosuppressants, there are now two oral JAK inhibitors – abrocitinib and upadacitinib – approved in 2022 by the Food and Drug Administration for treating AD, which affects up to 2.4% of the global population. (A topical formulation of ruxolitinib, a JAK inhibitor, was approved for AD in 2021.)
The Singapore study is one of the few that have examined the safety and efficacy of JAK inhibitors for treatment of AD in a non-White population.
Chinese population
For the 12-week trial, conducted in 2022, Dr. Yew and associates recruited 35 patients from the NSC. More than half of participants (64%) were men and most (96%) were of Chinese ethnicity. Four of every five patients had previously received systemic agents: 17% had been treated with one systemic agent, 18.9% with two, 15.1% with three, 22.6% with four, and 3.8% with five. The most commonly used agents were cyclosporine (62.3%), methotrexate (47.2%), azathioprine (39.6%), and dupilumab (35.8%).
“The switch in therapy could have been a result of inadequate efficacy or cost reasons because in Singapore patients pay out of pocket for AD treatments,” said Dr. Yew.
Additionally, he offered a caveat on the profile of participants: “Perhaps they were more difficult atopic eczema patients, and therefore, the efficacy [of JAK inhibitors] might be a bit different.”
Clearer skin, less itch
Patients received one of the three study drugs: baricitinib (66%), abrocitinib (21%), and upadacitinib (13%). The distribution was “affected by reimbursement patterns and availability of the drug,” explained Dr. Yew.
They were assessed at weeks 4 and 12. By study end, the proportion of patients who self-reported an improvement in their condition was 100% for upadacitinib, 90% for abrocitinib, and 69% for baricitinib.
Scores on the Investigator Global Assessment (IGA) also improved with treatment. Patients in the baricitinib group saw their mean score fall from 4.0 to 3.0 by week 4, then to 2.0 by week 12. With upadacitinib and abrocitinib, “you can see that there is a nice decrease in IGA responses,” said Dr. Yew, referring to the larger improvement in scores experienced by patients on those two treatments. For patients on upadacitinib, IGA decreased from 3.5 to 2 at 4 weeks, then to 0.5 at 12 weeks, while those taking abrocitinib had their scores drop from 4.0 to 2.0 at 4 weeks, then to 1.0 at 12 weeks.
When it came to itch reduction, the abrocitinib group experienced the biggest reduction, with a median reduction of 5.5 points in itch score. Median reduction in itch score was 4 points for the other two groups. “Oral JAK inhibitors appear to have a good effect on itch response,” said Dr. Yew.
However, the researchers observed no significant reduction in percentage of body surface area affected, the last outcome assessed.
The most commonly reported adverse events were increased creatine kinase levels (11.3% of patients), increased LDL cholesterol levels (9.4%), and herpes zoster (9.4%). Those in the abrocitinib reported a higher number of these adverse events, compared with the other two treatment groups. (There were no herpes zoster cases among those taking baricitinib.)
For herpes zoster, Dr. Yew said “the common recommendation” is to give the inactivated shingles vaccine. “But the problem is that, number one, these patients would have probably failed multiple agents so they probably can’t wait for you to vaccinate before you initiate treatment.”
In addition, people in Singapore have to pay out-of-pocket for the two vaccine doses, “which is probably a month’s worth of medication,” he noted. “So we have a lot of resistance from patients.”
Additionally, Dr. Yew noted that contrary to what has previously been reported in the literature, there were few complaints of acne as a side effect in the Singaporean study population.
Toward greater representation
Dr. Yew pointed out that the study was limited by a few factors: neither the Eczema Area and Severity Index or Scoring of Atopic Dermatitis index data was used, and the study population was small and not representative of the real world.
Still, the new findings contribute to the overall safety and efficacy profile of JAK inhibitors in AD, which has so far been scarce in non-White populations.
“In Western studies, unfortunately, the representation of the population of skin of color or different ethnicities is underrepresented,” said Yousef Binamer, MD, chair of the dermatology department at King Faisal Specialist Hospital, Riyadh, Saudi Arabia, when approached for an independent comment on the results.
“This is now why researchers are looking into specific groups to study them,” which he pointed out, is crucial because “the immunophenotyping of AD is different for each background.”
The incidence and severity of AD tend to be higher in Asian and Middle Eastern populations, for instance, he noted. “It’s very common in Asia, and not so common in very white skin. I did my training in Canada so I see the difference,” said Dr. Binamer. “Asian people tend to be more itchy and have a tendency to scar on pigmentation.” Whereas White people “usually do not have this issue.”
“So I think real-world evidence of JAK inhibitors in the other populations is important,” he said. Studies such as the one conducted in Singapore, as well as the recently reported QUARTZ3 study, which examined the use of the JAK inhibitor ivarmacitinib in 256 Chinese patients with AD, are helping to pave the way.
The study was independently supported. Dr. Yew and Dr. Binamer have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT WCD 2023
New guidelines for MTX use in pediatric inflammatory skin disease unveiled
While the typical dose of methotrexate (MTX) for inflammatory disease in pediatric patients varies in published studies, the maximum dose is considered to be 1 mg/kg and not to exceed 25 mg/week. In addition, test doses are not necessary for pediatric patients starting low dose (1 mg/kg or less) MTX for inflammatory skin disease, and the onset of efficacy with MTX may take 8-16 weeks.
and published online in Pediatric Dermatology.
“Methotrexate is a cost-effective, readily accessible, well-tolerated, useful, and time-honored option for children with a spectrum of inflammatory skin diseases,” project cochair Elaine C. Siegfried, MD, professor of pediatrics and dermatology at Saint Louis University, told this news organization. “Although considered an ‘immune suppressant’ by some, it is more accurately classified as an immune modulator and has been widely used for more than 50 years, and remains the standard of care when administered at very high doses and intrathecally in children with acute lymphoblastic leukemia – a practice that supports safety. But many details that support optimized treatment are not widely appreciated.”
In their guidelines document, Dr. Siegfried and her 22 coauthors noted that Food and Drug Administration labeling does not include approved indications for the use of MTX for many inflammatory skin diseases in pediatric patients, including morphea, psoriasis, atopic dermatitis, and alopecia areata. “Furthermore, some clinicians may be unfamiliar or uncomfortable prescribing medications off label for pediatric patients, causing delayed initiation, premature drug discontinuation, or use of less advantageous alternatives,” they wrote.
To address this unmet need, Dr. Siegfried and the other committee members used a modified Delphi process to reach agreement on recommendations related to five key topic areas: indications and contraindications, dosing, interactions with immunizations and medications, potential for and management of adverse effects, and monitoring needs. Consensus was predefined as at least 70% of participants rating a statement as 7-9 on the Likert scale. The effort to develop 46 recommendations has been a work in progress for almost 5 years, “somewhat delayed by the pandemic,” Dr. Siegfried, past president and director of the American Board of Dermatology, said in an interview. “But it remains relevant, despite the emergence of biologics and JAK inhibitors for treating inflammatory skin conditions in children. Although the mechanism-of-action of low-dose MTX is not clear, it may overlap with the newer small molecules.”
The guidelines contain several pearls to guide optimal dosing, including the following key points:
- MTX can be discontinued abruptly without adverse effects, other than the risk of disease worsening.
- Folic acid supplementation (starting at 1 mg/day, regardless of weight) is an effective approach to minimizing associated gastrointestinal adverse effects.
- Concomitant use of MTX and antibiotics (including trimethoprim-sulfamethoxazole) and NSAIDS are not contraindicated for most pediatric patients treated for inflammatory skin disease.
- Live virus vaccine boosters such as varicella-zoster virus (VZV) and measles, mumps, and rubella (MMR) are not contraindicated in patients taking MTX; there are insufficient data to make recommendations for or against primary immunization with MMR vaccine in patients taking MTX; inactivated vaccines should be given to patients taking MTX.
- Routine surveillance laboratory monitoring (i.e., CBC with differential, alanine transaminase, aspartate aminotransferase, creatinine) is recommended at baseline, after 1 month of treatment, and every 3-4 months thereafter.
- Transient transaminase elevation (≤ 3 upper limit normal for < 3 months) is not uncommon with low-dose MTX and does not usually require interruption of MTX. The most likely causes are concomitant viral infection, MTX dosing within 24 hours prior to phlebotomy, recent administration of other medications (such as acetaminophen), and/or recent alcohol consumption.
- Liver biopsy is not indicated for routine monitoring of pediatric patients taking low-dose MTX.
According to Dr. Siegfried, consensus of the committee members was lowest on the need for a test dose of MTX.
Overall, she said in the interview, helping to craft the guidelines caused her to reflect on how her approach to using MTX has evolved over the past 35 years, after treating “many hundreds” of patients. “I was gratified to confirm similar practice patterns among my colleagues,” she added.
The project’s other cochair was Heather Brandling-Bennett, MD, a dermatologist at Seattle Children’s Hospital. This work was supported by a grant from the Pediatric Dermatology Research Alliance (PeDRA), with additional funding from the National Eczema Association and the National Psoriasis Foundation. Dr. Siegfried disclosed ties with AbbVie, Boehringer Ingelheim, Incyte, LEO Pharma, Novan, Novartis, Pierre Fabre, Pfizer, Regeneron, Sanofi Genzyme, UCB, and Verrica. She has participated in contracted research for AI Therapeutics, and has served as principal investigator for Janssen. Many of the guideline coauthors disclosed having received grant support and other funding from pharmaceutical companies.
While the typical dose of methotrexate (MTX) for inflammatory disease in pediatric patients varies in published studies, the maximum dose is considered to be 1 mg/kg and not to exceed 25 mg/week. In addition, test doses are not necessary for pediatric patients starting low dose (1 mg/kg or less) MTX for inflammatory skin disease, and the onset of efficacy with MTX may take 8-16 weeks.
and published online in Pediatric Dermatology.
“Methotrexate is a cost-effective, readily accessible, well-tolerated, useful, and time-honored option for children with a spectrum of inflammatory skin diseases,” project cochair Elaine C. Siegfried, MD, professor of pediatrics and dermatology at Saint Louis University, told this news organization. “Although considered an ‘immune suppressant’ by some, it is more accurately classified as an immune modulator and has been widely used for more than 50 years, and remains the standard of care when administered at very high doses and intrathecally in children with acute lymphoblastic leukemia – a practice that supports safety. But many details that support optimized treatment are not widely appreciated.”
In their guidelines document, Dr. Siegfried and her 22 coauthors noted that Food and Drug Administration labeling does not include approved indications for the use of MTX for many inflammatory skin diseases in pediatric patients, including morphea, psoriasis, atopic dermatitis, and alopecia areata. “Furthermore, some clinicians may be unfamiliar or uncomfortable prescribing medications off label for pediatric patients, causing delayed initiation, premature drug discontinuation, or use of less advantageous alternatives,” they wrote.
To address this unmet need, Dr. Siegfried and the other committee members used a modified Delphi process to reach agreement on recommendations related to five key topic areas: indications and contraindications, dosing, interactions with immunizations and medications, potential for and management of adverse effects, and monitoring needs. Consensus was predefined as at least 70% of participants rating a statement as 7-9 on the Likert scale. The effort to develop 46 recommendations has been a work in progress for almost 5 years, “somewhat delayed by the pandemic,” Dr. Siegfried, past president and director of the American Board of Dermatology, said in an interview. “But it remains relevant, despite the emergence of biologics and JAK inhibitors for treating inflammatory skin conditions in children. Although the mechanism-of-action of low-dose MTX is not clear, it may overlap with the newer small molecules.”
The guidelines contain several pearls to guide optimal dosing, including the following key points:
- MTX can be discontinued abruptly without adverse effects, other than the risk of disease worsening.
- Folic acid supplementation (starting at 1 mg/day, regardless of weight) is an effective approach to minimizing associated gastrointestinal adverse effects.
- Concomitant use of MTX and antibiotics (including trimethoprim-sulfamethoxazole) and NSAIDS are not contraindicated for most pediatric patients treated for inflammatory skin disease.
- Live virus vaccine boosters such as varicella-zoster virus (VZV) and measles, mumps, and rubella (MMR) are not contraindicated in patients taking MTX; there are insufficient data to make recommendations for or against primary immunization with MMR vaccine in patients taking MTX; inactivated vaccines should be given to patients taking MTX.
- Routine surveillance laboratory monitoring (i.e., CBC with differential, alanine transaminase, aspartate aminotransferase, creatinine) is recommended at baseline, after 1 month of treatment, and every 3-4 months thereafter.
- Transient transaminase elevation (≤ 3 upper limit normal for < 3 months) is not uncommon with low-dose MTX and does not usually require interruption of MTX. The most likely causes are concomitant viral infection, MTX dosing within 24 hours prior to phlebotomy, recent administration of other medications (such as acetaminophen), and/or recent alcohol consumption.
- Liver biopsy is not indicated for routine monitoring of pediatric patients taking low-dose MTX.
According to Dr. Siegfried, consensus of the committee members was lowest on the need for a test dose of MTX.
Overall, she said in the interview, helping to craft the guidelines caused her to reflect on how her approach to using MTX has evolved over the past 35 years, after treating “many hundreds” of patients. “I was gratified to confirm similar practice patterns among my colleagues,” she added.
The project’s other cochair was Heather Brandling-Bennett, MD, a dermatologist at Seattle Children’s Hospital. This work was supported by a grant from the Pediatric Dermatology Research Alliance (PeDRA), with additional funding from the National Eczema Association and the National Psoriasis Foundation. Dr. Siegfried disclosed ties with AbbVie, Boehringer Ingelheim, Incyte, LEO Pharma, Novan, Novartis, Pierre Fabre, Pfizer, Regeneron, Sanofi Genzyme, UCB, and Verrica. She has participated in contracted research for AI Therapeutics, and has served as principal investigator for Janssen. Many of the guideline coauthors disclosed having received grant support and other funding from pharmaceutical companies.
While the typical dose of methotrexate (MTX) for inflammatory disease in pediatric patients varies in published studies, the maximum dose is considered to be 1 mg/kg and not to exceed 25 mg/week. In addition, test doses are not necessary for pediatric patients starting low dose (1 mg/kg or less) MTX for inflammatory skin disease, and the onset of efficacy with MTX may take 8-16 weeks.
and published online in Pediatric Dermatology.
“Methotrexate is a cost-effective, readily accessible, well-tolerated, useful, and time-honored option for children with a spectrum of inflammatory skin diseases,” project cochair Elaine C. Siegfried, MD, professor of pediatrics and dermatology at Saint Louis University, told this news organization. “Although considered an ‘immune suppressant’ by some, it is more accurately classified as an immune modulator and has been widely used for more than 50 years, and remains the standard of care when administered at very high doses and intrathecally in children with acute lymphoblastic leukemia – a practice that supports safety. But many details that support optimized treatment are not widely appreciated.”
In their guidelines document, Dr. Siegfried and her 22 coauthors noted that Food and Drug Administration labeling does not include approved indications for the use of MTX for many inflammatory skin diseases in pediatric patients, including morphea, psoriasis, atopic dermatitis, and alopecia areata. “Furthermore, some clinicians may be unfamiliar or uncomfortable prescribing medications off label for pediatric patients, causing delayed initiation, premature drug discontinuation, or use of less advantageous alternatives,” they wrote.
To address this unmet need, Dr. Siegfried and the other committee members used a modified Delphi process to reach agreement on recommendations related to five key topic areas: indications and contraindications, dosing, interactions with immunizations and medications, potential for and management of adverse effects, and monitoring needs. Consensus was predefined as at least 70% of participants rating a statement as 7-9 on the Likert scale. The effort to develop 46 recommendations has been a work in progress for almost 5 years, “somewhat delayed by the pandemic,” Dr. Siegfried, past president and director of the American Board of Dermatology, said in an interview. “But it remains relevant, despite the emergence of biologics and JAK inhibitors for treating inflammatory skin conditions in children. Although the mechanism-of-action of low-dose MTX is not clear, it may overlap with the newer small molecules.”
The guidelines contain several pearls to guide optimal dosing, including the following key points:
- MTX can be discontinued abruptly without adverse effects, other than the risk of disease worsening.
- Folic acid supplementation (starting at 1 mg/day, regardless of weight) is an effective approach to minimizing associated gastrointestinal adverse effects.
- Concomitant use of MTX and antibiotics (including trimethoprim-sulfamethoxazole) and NSAIDS are not contraindicated for most pediatric patients treated for inflammatory skin disease.
- Live virus vaccine boosters such as varicella-zoster virus (VZV) and measles, mumps, and rubella (MMR) are not contraindicated in patients taking MTX; there are insufficient data to make recommendations for or against primary immunization with MMR vaccine in patients taking MTX; inactivated vaccines should be given to patients taking MTX.
- Routine surveillance laboratory monitoring (i.e., CBC with differential, alanine transaminase, aspartate aminotransferase, creatinine) is recommended at baseline, after 1 month of treatment, and every 3-4 months thereafter.
- Transient transaminase elevation (≤ 3 upper limit normal for < 3 months) is not uncommon with low-dose MTX and does not usually require interruption of MTX. The most likely causes are concomitant viral infection, MTX dosing within 24 hours prior to phlebotomy, recent administration of other medications (such as acetaminophen), and/or recent alcohol consumption.
- Liver biopsy is not indicated for routine monitoring of pediatric patients taking low-dose MTX.
According to Dr. Siegfried, consensus of the committee members was lowest on the need for a test dose of MTX.
Overall, she said in the interview, helping to craft the guidelines caused her to reflect on how her approach to using MTX has evolved over the past 35 years, after treating “many hundreds” of patients. “I was gratified to confirm similar practice patterns among my colleagues,” she added.
The project’s other cochair was Heather Brandling-Bennett, MD, a dermatologist at Seattle Children’s Hospital. This work was supported by a grant from the Pediatric Dermatology Research Alliance (PeDRA), with additional funding from the National Eczema Association and the National Psoriasis Foundation. Dr. Siegfried disclosed ties with AbbVie, Boehringer Ingelheim, Incyte, LEO Pharma, Novan, Novartis, Pierre Fabre, Pfizer, Regeneron, Sanofi Genzyme, UCB, and Verrica. She has participated in contracted research for AI Therapeutics, and has served as principal investigator for Janssen. Many of the guideline coauthors disclosed having received grant support and other funding from pharmaceutical companies.
FROM PEDIATRIC DERMATOLOGY
Understanding Medical Standards for Entrance Into Military Service and Disqualifying Dermatologic Conditions
Purpose of Medical Standards in the US Military
Young adults in the United States traditionally have viewed military service as a viable career given its stable salary, career training, opportunities for progression, comprehensive health care coverage, tuition assistance, and other benefits; however, not all who desire to serve in the US Military are eligible to join. The Department of Defense (DoD) maintains fitness and health requirements (ie, accession standards), which are codified in DoD Instruction 6130.03, Volume 1,1 that help ensure potential recruits can safely and fully perform their military duties. These accession standards change over time with the evolving understanding of diseases, medical advances, and accrued experience conducting operations in various environments. Accession standards serve to both preserve the health of the applicant and to ensure military mission success.
Dermatologic diseases have been prevalent in conflicts throughout US military history, representing a considerable source of morbidity to service members, inability of service members to remain on active duty, and costly use of resources. Hospitalizations of US Army soldiers for skin conditions led to the loss of more than 2 million days of service in World War I.2 In World War II, skin diseases made up 25% and 75% of all temperate and tropical climate visits, respectively. Cutaneous diseases were the most frequently addressed category for US service members in Vietnam, representing more than 1.5 million visits and nearly 10% of disease-related evacuations.2 Skin disease remains vital in 21st-century conflict. At a military hospital in Afghanistan, a review of 2421 outpatient medical records from June through July 2007 identified that dermatologic conditions resulted in 20% of military patient evaluations, 7% of nontraumatic hospital admissions, and 2% of total patient evacuations, at an estimated cost of $80,000 per evacuee.3 Between 2003 and 2006, 918 service members were evacuated for dermatologic reasons from combat zones in Afghanistan and Iraq.4
Unpredictable military environments may result in flares of a previously controlled condition, new skin diseases, or infection with endemic diseases. Mild cases of common conditions such as psoriasis or atopic dermatitis can present an unacceptable risk for severe flare in the setting of deployed military operations.5 Personnel may face extremes in temperature and humidity and work long hours under stress with limited or nonexistent opportunities for hygiene or self-care. Shared equipment and close living quarters permit the spread of infectious diseases and complicate the treatment of infestations. Military equipment and supplies such as gas masks and insect repellents can contain compounds that act as irritants or sensitizing agents, leading to contact dermatitis or urticaria. When dermatologic conditions develop or flare, further challenges are associated with evaluation and management. Health care resources vary considerably by location, with potential limitations in the availability of medications; supplies; refrigeration capabilities; and laboratory, microbiology, and histology services. Furthermore, dermatology referrals and services typically are not feasible in most deployed settings,3 though teledermatology has been available in the armed forces since 2002.
Deployed environments compound the consequences of dermatologic conditions and can impact the military mission. Military units deploy with the number of personnel needed to complete a mission and cannot replace members who become ill or injured or are medically evacuated. Something seemingly trivial, such as poor sleep due to pruritic dermatitis, may impair daytime alertness with potentially grave consequences in critical tasks such as guard or flying duties. The evacuation of a service member can compromise those left behind, and losing a service member with a unique required skill set may jeopardize a unit’s chance of success. Additionally, the impact of an evacuation itself extends beyond its direct cost and effects on the service member’s unit. The military does not maintain dedicated medical evacuation aircraft, instead repurposing aircraft in the deployed setting as needed.6 Evacuations can delay flights initially scheduled to move troops, ammunition, food, or other supplies and equipment elsewhere.
Disqualifying Skin and Soft Tissue Conditions
Current accession standards, which are listed in a publicly released document (DoD Instruction 6130.03, Volume 1), are updated based on medical, societal, and technical advances.1 These standards differ from retention standards, which apply to members actively serving in the military. Although the DoD creates a minimum standard for the entire military, the US Army, Navy, and Air Force adopt these standards and adjust as required for each branch’s needs. An updated copy can be found on the DoD Directives Division website (https://www.esd.whs.mil/dd/) or Med Standards, a third-party mobile application (app) available as a free download for Apple iOS and Android devices (https://www.doc-apps.com/). The app also includes each military branch’s interpretation of the requirements.
The accession standards outline medical conditions that, if present or verified in an applicant’s medical history, preclude joining the military (eTable). These standards are organized into general systems, with a section dedicated to dermatologic (skin and soft tissue) conditions.1 When a candidate has a potentially disqualifying medical condition identified by a screening questionnaire, medical record review, or military entrance physical examination, a referral for a determination of fitness for duty may be required. Medical accession standards are not solely driven by the diagnosis but also by the extent, nature, and timing of medical management. Procedures or prescriptions requiring frequent clinical monitoring, special handling, or severe dietary restrictions may deem the applicant’s condition potentially unsuitable. The need for immunosuppressive, anticoagulant, or refrigerated medications can impact a patient’s eligibility due to future deployment requirements and suitability for prolonged service, especially if treated for any substantial length of time. Chronic dermatologic conditions that are unresponsive to treatment, are susceptible to exacerbation despite treatment, require regular follow-up care, or interfere with the wear of military gear may be inconsistent with future deployment standards. Although the dermatologist should primarily focus on the skin and soft tissue conditions section of the accession standards, some dermatologic conditions can overlap with other medical systems and be located in a different section; for example, the section on lower extremity conditions includes a disqualifying condition of “[c]urrent ingrown toenails, if infected or symptomatic.”1
Waiver Process
Medical conditions listed in the accession standards are deemed ineligible for military service; however, applicants can apply for a waiver.1 The goal is for service members to be well controlled without treatment or with treatment widely available at military clinics and hospitals. Waivers ensure that service members are “[m]edically capable of performing duties without aggravating physical defects or medical conditions,” are “[m]edically adaptable to the military environment without geographical area limitations,” and are “free of medical conditions or physical defects that may reasonably be expected to require excessive time lost from duty for necessary treatment or hospitalization, or may result in separation from the Military Service for unfitness.”1 The waiver process requires an evaluation from specialists with verification and documentation but does not guarantee approval. Although each military branch follows the same guidelines for disqualifying medical conditions, the evaluation and waiver process varies.
Considerations for Civilian Dermatologists
For several reasons, accurate and detailed medical documentation is essential for patients who pursue military service. Applicants must complete detailed health questionnaires and may need to provide copies of health records. The military electronic health record connects to large civilian health information exchanges and pulls primary documentation from records at many hospitals and clinics. Although applicants may request supportive clarification from their dermatologists, the military relies on primary medical documentation throughout the recruitment process. Accurate diagnostic codes reduce ambiguity, as accession standards are organized by diagnosis; for example, an unspecified history of psoriasis disqualifies applicants unless documentation supports nonrecurrent childhood guttate psoriasis.1 Clear documentation of symptom severity, response to treatment, or resolution of a condition may elucidate suitability for service when matching a potentially disqualifying condition to a standard is not straightforward. Correct documentation will ensure that potential service members achieve a waiver when it is appropriate. If they are found to be unfit, it may save a patient from a bad outcome or a military unit from mission failure.
Dermatologists in the United States can reference current military medical accession standards to guide patients when needed. For example, a prospective recruit may be hesitant to start isotretinoin for severe nodulocystic acne, concerned that this medication may preclude them from joining the military. The current standards state that “[a]pplicants under treatment with systemic retinoids . . . do not meet the standard until 4 weeks after completing therapy,” while active severe nodulocystic acne is a disqualifying condition.1 Therefore, the patient could proceed with isotretinoin therapy and, pending clinical response, meet accession standards as soon as 4 weeks after treatment. A clear understanding of the purpose of these standards, including protecting the applicant’s health and maximizing the chance of combat mission accomplishment, helps to reinforce responsibilities when caring for patients who wish to serve.


- US Department of Defense. DoD Instruction 6130.03, Volume 1. Medical Standards for Military Service: Appointment, Enlistment, or Induction. Updated November 16, 2022. Accessed May 22, 2023. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003_vol1.PDF?ver=7fhqacc0jGX_R9_1iexudA%3D%3D
- Becker LE, James WD. Historical overview and principles of diagnosis. In: Becker LE, James WD. Military Dermatology. Office of the Surgeon General, US Department of the Army; 1994: 1-20.
- Arnold JG, Michener MD. Evaluation of dermatologic conditions by primary care providers in deployed military settings. Mil Med. 2008;173:882-888. doi:10.7205/MILMED.173.9.882
- McGraw TA, Norton SA. Military aeromedical evacuations from central and southwest Asia for ill-defined dermatologic diseases. Arch Dermatol. 2009;145:165-170.
- Gelman AB, Norton SA, Valdes-Rodriguez R, et al. A review of skin conditions in modern warfare and peacekeeping operations. Mil Med. 2015;180:32-37.
- Fang R, Dorlac GR, Allan PF, et al. Intercontinental aeromedical evacuation of patients with traumatic brain injuries during Operations Iraqi Freedom and Enduring Freedom. Neurosurg Focus. 2010;28:E11.
Purpose of Medical Standards in the US Military
Young adults in the United States traditionally have viewed military service as a viable career given its stable salary, career training, opportunities for progression, comprehensive health care coverage, tuition assistance, and other benefits; however, not all who desire to serve in the US Military are eligible to join. The Department of Defense (DoD) maintains fitness and health requirements (ie, accession standards), which are codified in DoD Instruction 6130.03, Volume 1,1 that help ensure potential recruits can safely and fully perform their military duties. These accession standards change over time with the evolving understanding of diseases, medical advances, and accrued experience conducting operations in various environments. Accession standards serve to both preserve the health of the applicant and to ensure military mission success.
Dermatologic diseases have been prevalent in conflicts throughout US military history, representing a considerable source of morbidity to service members, inability of service members to remain on active duty, and costly use of resources. Hospitalizations of US Army soldiers for skin conditions led to the loss of more than 2 million days of service in World War I.2 In World War II, skin diseases made up 25% and 75% of all temperate and tropical climate visits, respectively. Cutaneous diseases were the most frequently addressed category for US service members in Vietnam, representing more than 1.5 million visits and nearly 10% of disease-related evacuations.2 Skin disease remains vital in 21st-century conflict. At a military hospital in Afghanistan, a review of 2421 outpatient medical records from June through July 2007 identified that dermatologic conditions resulted in 20% of military patient evaluations, 7% of nontraumatic hospital admissions, and 2% of total patient evacuations, at an estimated cost of $80,000 per evacuee.3 Between 2003 and 2006, 918 service members were evacuated for dermatologic reasons from combat zones in Afghanistan and Iraq.4
Unpredictable military environments may result in flares of a previously controlled condition, new skin diseases, or infection with endemic diseases. Mild cases of common conditions such as psoriasis or atopic dermatitis can present an unacceptable risk for severe flare in the setting of deployed military operations.5 Personnel may face extremes in temperature and humidity and work long hours under stress with limited or nonexistent opportunities for hygiene or self-care. Shared equipment and close living quarters permit the spread of infectious diseases and complicate the treatment of infestations. Military equipment and supplies such as gas masks and insect repellents can contain compounds that act as irritants or sensitizing agents, leading to contact dermatitis or urticaria. When dermatologic conditions develop or flare, further challenges are associated with evaluation and management. Health care resources vary considerably by location, with potential limitations in the availability of medications; supplies; refrigeration capabilities; and laboratory, microbiology, and histology services. Furthermore, dermatology referrals and services typically are not feasible in most deployed settings,3 though teledermatology has been available in the armed forces since 2002.
Deployed environments compound the consequences of dermatologic conditions and can impact the military mission. Military units deploy with the number of personnel needed to complete a mission and cannot replace members who become ill or injured or are medically evacuated. Something seemingly trivial, such as poor sleep due to pruritic dermatitis, may impair daytime alertness with potentially grave consequences in critical tasks such as guard or flying duties. The evacuation of a service member can compromise those left behind, and losing a service member with a unique required skill set may jeopardize a unit’s chance of success. Additionally, the impact of an evacuation itself extends beyond its direct cost and effects on the service member’s unit. The military does not maintain dedicated medical evacuation aircraft, instead repurposing aircraft in the deployed setting as needed.6 Evacuations can delay flights initially scheduled to move troops, ammunition, food, or other supplies and equipment elsewhere.
Disqualifying Skin and Soft Tissue Conditions
Current accession standards, which are listed in a publicly released document (DoD Instruction 6130.03, Volume 1), are updated based on medical, societal, and technical advances.1 These standards differ from retention standards, which apply to members actively serving in the military. Although the DoD creates a minimum standard for the entire military, the US Army, Navy, and Air Force adopt these standards and adjust as required for each branch’s needs. An updated copy can be found on the DoD Directives Division website (https://www.esd.whs.mil/dd/) or Med Standards, a third-party mobile application (app) available as a free download for Apple iOS and Android devices (https://www.doc-apps.com/). The app also includes each military branch’s interpretation of the requirements.
The accession standards outline medical conditions that, if present or verified in an applicant’s medical history, preclude joining the military (eTable). These standards are organized into general systems, with a section dedicated to dermatologic (skin and soft tissue) conditions.1 When a candidate has a potentially disqualifying medical condition identified by a screening questionnaire, medical record review, or military entrance physical examination, a referral for a determination of fitness for duty may be required. Medical accession standards are not solely driven by the diagnosis but also by the extent, nature, and timing of medical management. Procedures or prescriptions requiring frequent clinical monitoring, special handling, or severe dietary restrictions may deem the applicant’s condition potentially unsuitable. The need for immunosuppressive, anticoagulant, or refrigerated medications can impact a patient’s eligibility due to future deployment requirements and suitability for prolonged service, especially if treated for any substantial length of time. Chronic dermatologic conditions that are unresponsive to treatment, are susceptible to exacerbation despite treatment, require regular follow-up care, or interfere with the wear of military gear may be inconsistent with future deployment standards. Although the dermatologist should primarily focus on the skin and soft tissue conditions section of the accession standards, some dermatologic conditions can overlap with other medical systems and be located in a different section; for example, the section on lower extremity conditions includes a disqualifying condition of “[c]urrent ingrown toenails, if infected or symptomatic.”1
Waiver Process
Medical conditions listed in the accession standards are deemed ineligible for military service; however, applicants can apply for a waiver.1 The goal is for service members to be well controlled without treatment or with treatment widely available at military clinics and hospitals. Waivers ensure that service members are “[m]edically capable of performing duties without aggravating physical defects or medical conditions,” are “[m]edically adaptable to the military environment without geographical area limitations,” and are “free of medical conditions or physical defects that may reasonably be expected to require excessive time lost from duty for necessary treatment or hospitalization, or may result in separation from the Military Service for unfitness.”1 The waiver process requires an evaluation from specialists with verification and documentation but does not guarantee approval. Although each military branch follows the same guidelines for disqualifying medical conditions, the evaluation and waiver process varies.
Considerations for Civilian Dermatologists
For several reasons, accurate and detailed medical documentation is essential for patients who pursue military service. Applicants must complete detailed health questionnaires and may need to provide copies of health records. The military electronic health record connects to large civilian health information exchanges and pulls primary documentation from records at many hospitals and clinics. Although applicants may request supportive clarification from their dermatologists, the military relies on primary medical documentation throughout the recruitment process. Accurate diagnostic codes reduce ambiguity, as accession standards are organized by diagnosis; for example, an unspecified history of psoriasis disqualifies applicants unless documentation supports nonrecurrent childhood guttate psoriasis.1 Clear documentation of symptom severity, response to treatment, or resolution of a condition may elucidate suitability for service when matching a potentially disqualifying condition to a standard is not straightforward. Correct documentation will ensure that potential service members achieve a waiver when it is appropriate. If they are found to be unfit, it may save a patient from a bad outcome or a military unit from mission failure.
Dermatologists in the United States can reference current military medical accession standards to guide patients when needed. For example, a prospective recruit may be hesitant to start isotretinoin for severe nodulocystic acne, concerned that this medication may preclude them from joining the military. The current standards state that “[a]pplicants under treatment with systemic retinoids . . . do not meet the standard until 4 weeks after completing therapy,” while active severe nodulocystic acne is a disqualifying condition.1 Therefore, the patient could proceed with isotretinoin therapy and, pending clinical response, meet accession standards as soon as 4 weeks after treatment. A clear understanding of the purpose of these standards, including protecting the applicant’s health and maximizing the chance of combat mission accomplishment, helps to reinforce responsibilities when caring for patients who wish to serve.


Purpose of Medical Standards in the US Military
Young adults in the United States traditionally have viewed military service as a viable career given its stable salary, career training, opportunities for progression, comprehensive health care coverage, tuition assistance, and other benefits; however, not all who desire to serve in the US Military are eligible to join. The Department of Defense (DoD) maintains fitness and health requirements (ie, accession standards), which are codified in DoD Instruction 6130.03, Volume 1,1 that help ensure potential recruits can safely and fully perform their military duties. These accession standards change over time with the evolving understanding of diseases, medical advances, and accrued experience conducting operations in various environments. Accession standards serve to both preserve the health of the applicant and to ensure military mission success.
Dermatologic diseases have been prevalent in conflicts throughout US military history, representing a considerable source of morbidity to service members, inability of service members to remain on active duty, and costly use of resources. Hospitalizations of US Army soldiers for skin conditions led to the loss of more than 2 million days of service in World War I.2 In World War II, skin diseases made up 25% and 75% of all temperate and tropical climate visits, respectively. Cutaneous diseases were the most frequently addressed category for US service members in Vietnam, representing more than 1.5 million visits and nearly 10% of disease-related evacuations.2 Skin disease remains vital in 21st-century conflict. At a military hospital in Afghanistan, a review of 2421 outpatient medical records from June through July 2007 identified that dermatologic conditions resulted in 20% of military patient evaluations, 7% of nontraumatic hospital admissions, and 2% of total patient evacuations, at an estimated cost of $80,000 per evacuee.3 Between 2003 and 2006, 918 service members were evacuated for dermatologic reasons from combat zones in Afghanistan and Iraq.4
Unpredictable military environments may result in flares of a previously controlled condition, new skin diseases, or infection with endemic diseases. Mild cases of common conditions such as psoriasis or atopic dermatitis can present an unacceptable risk for severe flare in the setting of deployed military operations.5 Personnel may face extremes in temperature and humidity and work long hours under stress with limited or nonexistent opportunities for hygiene or self-care. Shared equipment and close living quarters permit the spread of infectious diseases and complicate the treatment of infestations. Military equipment and supplies such as gas masks and insect repellents can contain compounds that act as irritants or sensitizing agents, leading to contact dermatitis or urticaria. When dermatologic conditions develop or flare, further challenges are associated with evaluation and management. Health care resources vary considerably by location, with potential limitations in the availability of medications; supplies; refrigeration capabilities; and laboratory, microbiology, and histology services. Furthermore, dermatology referrals and services typically are not feasible in most deployed settings,3 though teledermatology has been available in the armed forces since 2002.
Deployed environments compound the consequences of dermatologic conditions and can impact the military mission. Military units deploy with the number of personnel needed to complete a mission and cannot replace members who become ill or injured or are medically evacuated. Something seemingly trivial, such as poor sleep due to pruritic dermatitis, may impair daytime alertness with potentially grave consequences in critical tasks such as guard or flying duties. The evacuation of a service member can compromise those left behind, and losing a service member with a unique required skill set may jeopardize a unit’s chance of success. Additionally, the impact of an evacuation itself extends beyond its direct cost and effects on the service member’s unit. The military does not maintain dedicated medical evacuation aircraft, instead repurposing aircraft in the deployed setting as needed.6 Evacuations can delay flights initially scheduled to move troops, ammunition, food, or other supplies and equipment elsewhere.
Disqualifying Skin and Soft Tissue Conditions
Current accession standards, which are listed in a publicly released document (DoD Instruction 6130.03, Volume 1), are updated based on medical, societal, and technical advances.1 These standards differ from retention standards, which apply to members actively serving in the military. Although the DoD creates a minimum standard for the entire military, the US Army, Navy, and Air Force adopt these standards and adjust as required for each branch’s needs. An updated copy can be found on the DoD Directives Division website (https://www.esd.whs.mil/dd/) or Med Standards, a third-party mobile application (app) available as a free download for Apple iOS and Android devices (https://www.doc-apps.com/). The app also includes each military branch’s interpretation of the requirements.
The accession standards outline medical conditions that, if present or verified in an applicant’s medical history, preclude joining the military (eTable). These standards are organized into general systems, with a section dedicated to dermatologic (skin and soft tissue) conditions.1 When a candidate has a potentially disqualifying medical condition identified by a screening questionnaire, medical record review, or military entrance physical examination, a referral for a determination of fitness for duty may be required. Medical accession standards are not solely driven by the diagnosis but also by the extent, nature, and timing of medical management. Procedures or prescriptions requiring frequent clinical monitoring, special handling, or severe dietary restrictions may deem the applicant’s condition potentially unsuitable. The need for immunosuppressive, anticoagulant, or refrigerated medications can impact a patient’s eligibility due to future deployment requirements and suitability for prolonged service, especially if treated for any substantial length of time. Chronic dermatologic conditions that are unresponsive to treatment, are susceptible to exacerbation despite treatment, require regular follow-up care, or interfere with the wear of military gear may be inconsistent with future deployment standards. Although the dermatologist should primarily focus on the skin and soft tissue conditions section of the accession standards, some dermatologic conditions can overlap with other medical systems and be located in a different section; for example, the section on lower extremity conditions includes a disqualifying condition of “[c]urrent ingrown toenails, if infected or symptomatic.”1
Waiver Process
Medical conditions listed in the accession standards are deemed ineligible for military service; however, applicants can apply for a waiver.1 The goal is for service members to be well controlled without treatment or with treatment widely available at military clinics and hospitals. Waivers ensure that service members are “[m]edically capable of performing duties without aggravating physical defects or medical conditions,” are “[m]edically adaptable to the military environment without geographical area limitations,” and are “free of medical conditions or physical defects that may reasonably be expected to require excessive time lost from duty for necessary treatment or hospitalization, or may result in separation from the Military Service for unfitness.”1 The waiver process requires an evaluation from specialists with verification and documentation but does not guarantee approval. Although each military branch follows the same guidelines for disqualifying medical conditions, the evaluation and waiver process varies.
Considerations for Civilian Dermatologists
For several reasons, accurate and detailed medical documentation is essential for patients who pursue military service. Applicants must complete detailed health questionnaires and may need to provide copies of health records. The military electronic health record connects to large civilian health information exchanges and pulls primary documentation from records at many hospitals and clinics. Although applicants may request supportive clarification from their dermatologists, the military relies on primary medical documentation throughout the recruitment process. Accurate diagnostic codes reduce ambiguity, as accession standards are organized by diagnosis; for example, an unspecified history of psoriasis disqualifies applicants unless documentation supports nonrecurrent childhood guttate psoriasis.1 Clear documentation of symptom severity, response to treatment, or resolution of a condition may elucidate suitability for service when matching a potentially disqualifying condition to a standard is not straightforward. Correct documentation will ensure that potential service members achieve a waiver when it is appropriate. If they are found to be unfit, it may save a patient from a bad outcome or a military unit from mission failure.
Dermatologists in the United States can reference current military medical accession standards to guide patients when needed. For example, a prospective recruit may be hesitant to start isotretinoin for severe nodulocystic acne, concerned that this medication may preclude them from joining the military. The current standards state that “[a]pplicants under treatment with systemic retinoids . . . do not meet the standard until 4 weeks after completing therapy,” while active severe nodulocystic acne is a disqualifying condition.1 Therefore, the patient could proceed with isotretinoin therapy and, pending clinical response, meet accession standards as soon as 4 weeks after treatment. A clear understanding of the purpose of these standards, including protecting the applicant’s health and maximizing the chance of combat mission accomplishment, helps to reinforce responsibilities when caring for patients who wish to serve.


- US Department of Defense. DoD Instruction 6130.03, Volume 1. Medical Standards for Military Service: Appointment, Enlistment, or Induction. Updated November 16, 2022. Accessed May 22, 2023. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003_vol1.PDF?ver=7fhqacc0jGX_R9_1iexudA%3D%3D
- Becker LE, James WD. Historical overview and principles of diagnosis. In: Becker LE, James WD. Military Dermatology. Office of the Surgeon General, US Department of the Army; 1994: 1-20.
- Arnold JG, Michener MD. Evaluation of dermatologic conditions by primary care providers in deployed military settings. Mil Med. 2008;173:882-888. doi:10.7205/MILMED.173.9.882
- McGraw TA, Norton SA. Military aeromedical evacuations from central and southwest Asia for ill-defined dermatologic diseases. Arch Dermatol. 2009;145:165-170.
- Gelman AB, Norton SA, Valdes-Rodriguez R, et al. A review of skin conditions in modern warfare and peacekeeping operations. Mil Med. 2015;180:32-37.
- Fang R, Dorlac GR, Allan PF, et al. Intercontinental aeromedical evacuation of patients with traumatic brain injuries during Operations Iraqi Freedom and Enduring Freedom. Neurosurg Focus. 2010;28:E11.
- US Department of Defense. DoD Instruction 6130.03, Volume 1. Medical Standards for Military Service: Appointment, Enlistment, or Induction. Updated November 16, 2022. Accessed May 22, 2023. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003_vol1.PDF?ver=7fhqacc0jGX_R9_1iexudA%3D%3D
- Becker LE, James WD. Historical overview and principles of diagnosis. In: Becker LE, James WD. Military Dermatology. Office of the Surgeon General, US Department of the Army; 1994: 1-20.
- Arnold JG, Michener MD. Evaluation of dermatologic conditions by primary care providers in deployed military settings. Mil Med. 2008;173:882-888. doi:10.7205/MILMED.173.9.882
- McGraw TA, Norton SA. Military aeromedical evacuations from central and southwest Asia for ill-defined dermatologic diseases. Arch Dermatol. 2009;145:165-170.
- Gelman AB, Norton SA, Valdes-Rodriguez R, et al. A review of skin conditions in modern warfare and peacekeeping operations. Mil Med. 2015;180:32-37.
- Fang R, Dorlac GR, Allan PF, et al. Intercontinental aeromedical evacuation of patients with traumatic brain injuries during Operations Iraqi Freedom and Enduring Freedom. Neurosurg Focus. 2010;28:E11.
Practice Points
- Dermatologic diseases have played a substantial role in conflicts throughout US military history, representing a considerable source of morbidity to service members, loss of active-duty service members trained with necessary skills, and costly use of resources.
- The strict standards are designed to protect the health of the individual and maximize mission success.
- The Department of Defense has a publicly available document (DoD Instruction 6130.03, Volume 1) that details conditions that are disqualifying for entrance into the military. Dermatologists can reference this to provide guidance to adolescents and young adults interested in joining the military.
Dupilumab-Associated Sweet Syndrome
Sweet syndrome (SS), also known as acute febrile neutrophilic dermatosis, was first described in 1964. 1 Since then, several subtypes of SS have been recognized, including classic or idiopathic, which typically follows an acute viral illness; cancer related, typically in the form of a paraneoplastic syndrome; and drug induced. 2 Drug-induced SS is defined by the following: (1) an abrupt onset of painful erythematous plaques or nodules; (2) histopathologic evidence of a dense neutrophilic infiltrate without evidence of leukocytoclastic vasculitis; (3) pyrexia above 38 ° C; (4) temporal relationship between drug and clinical presentation or temporally related recurrence after rechallenge; and (5) temporally related resolution of lesions after drug withdrawal or treatment with systemic corticosteroids. 3 All 5 criteria must be met to make a diagnosis of drug-induced SS. Since these criteria were established by Walker and Cohen, 3 various drugs have been identified as causative agents, including antibiotics, antiepileptics, antiretrovirals, antineoplastic agents, antipsychotics, oral contraceptives, nonsteroidal anti-inflammatory agents, and retinoids. 4 W e present a rare case of SS caused by dupilumab, a monoclonal antibody therapy, used in the treatment of severe eosinophilic asthma and atopic dermatitis.
Case Report
A 53-year-old woman presented with painful skin lesions, arthralgia, fever, and leukocytosis following initiation of dupilumab. She had a history of adult-onset, severe, persistent eosinophilic asthma, as well as chronic rhinosinusitis with nasal polyps, plaque psoriasis, and hypertrophic cardiomyopathy. She started mepolizumab 3 years prior to the current presentation for persistently uncontrolled asthma with a baseline peripheral eosinophil count of 860 cells/µL. After 3 years of minimal response to mepolizumab, she was started on dupilumab. Within 2 weeks of the first dose of dupilumab, she started experiencing bilateral knee pain. She subsequently developed daily fevers (temperature, 38.3 °C to 39.4 °C), fatigue, and pain in the back of the neck and head. After the second dose of dupilumab, she started experiencing painful skin lesions on the bilateral knuckles, elbows, and abdomen (Figure 1). She had difficulty using her hands and walking secondary to intense arthralgia involving the bilateral finger joints, elbows, and knees. Her primary care physician obtained a laboratory evaluation, which revealed an elevated total white blood cell count of 20×103/mm3 (reference range, 4–11×103/mm3) with 27.5% neutrophils and severely elevated eosinophils above her baseline to 57.3% with an absolute eosinophil count of 11,700 cells/µL (reference range, <400 cells/µL). Further assessment revealed an elevated erythrocyte sedimentation rate of 64 mm/h (reference range, 0–30 mm/h) and C-reactive protein level of 34 mg/dL (reference range, ≤0.80 mg/dL), with negative antinuclear antibody, rheumatoid factor, antineutrophilic cytoplasmic antibody, and Lyme antibody. IgG, IgA, and IgM levels were within reference range, and the IgE level was not elevated above her baseline. She had normal serum tryptase, and a peripheral D816V c-KIT mutation was not detected. She was subsequently hospitalized for further evaluation, at which time there was no fever or localizing infectious signs or symptoms. An infectious evaluation including urinalysis; respiratory swab for adenovirus, coronaviruses, human metapneumovirus, rhinovirus/enterovirus, influenza A and B, parainfluenza viruses, respiratory syncytial virus, Chlamydophila pneumoniae, and Mycoplasma pneumoniae; Lyme serology; and a computed tomography (CT) scan of the chest, abdomen, and pelvis revealed no evidence of infection. A parasite evaluation was ordered but was not performed. There was no evidence of malignancy on CT of the chest, abdomen, and pelvis or CT of the head without contrast. A lumbar puncture was considered but was ultimately deferred.

At the current presentation, the patient was following up in the dermatology clinic shortly after discharge. The lesions on the fingers and arms were described by the dermatologist as deep, erythematous, 0.5-cm bullous papules. The differential diagnosis at this time included a cutaneous or systemic infection, vasculitis, drug eruption, or cutaneous manifestation of an autoimmune condition. A shave biopsy of a skin lesion on the right hand demonstrated epidermal necrosis with a dense dermal neutrophilic infiltrate consistent with a neutrophilic dermatosis (Figure 2). There was no evidence of leukocytoclastic vasculitis. The histologic differential diagnosis included cutaneous infection, neutrophilic dermatosis of the hand, and SS. Special stains for infectious organisms including Gram, Grocott methenamine-silver, and auramine-rhodamine stains were negative for bacterial, fungal, and mycobacterial organisms, ruling out cutaneous infection. A diagnosis of drug-induced SS was made based on the histologic findings, diffuse distribution of the lesions, negative infectious evaluation, lack of underlying malignancy or autoimmune conditions, and onset following initiation of dupilumab.
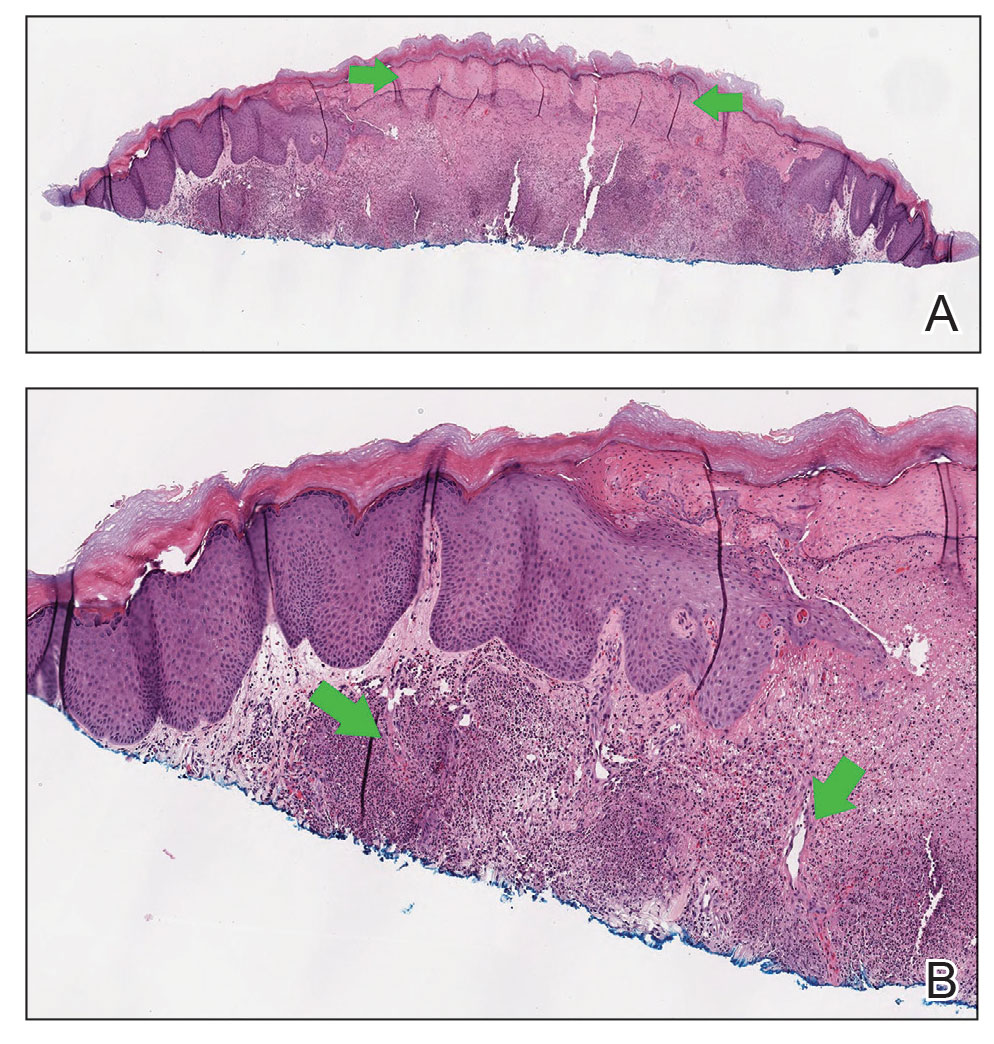
Dupilumab was discontinued, and the patient was started on prednisone with rapid improvement in the symptoms. She underwent a slow taper of the prednisone over approximately 2 months with a slow downtrend of eosinophils. She was transitioned to a different biologic agent, benralizumab, with no further recurrence of the rash or arthralgia.
Comment
Dupilumab is a human monoclonal IgG4 antibody that inhibits IL-4 and IL-13 signaling by binding to the IL-4Rα subunit. By blocking IL-4Rα, dupilumab inhibits IL-4 and IL-13 cytokine-induced inflammatory responses, including the release of proinflammatory cytokines, chemokines, nitric oxide, and IgE. Currently, dupilumab is approved to treat refractory forms of moderate to severe asthma characterized by an eosinophilic phenotype or with corticosteroid-dependent asthma, moderate to severe atopic dermatitis, chronic rhinosinusitis with nasal polyposis, and eosinophilic esophagitis. The most common adverse events (incidence ≥1%) are injection-site reactions, oropharyngeal pain, and eosinophilia.5 Interestingly, our patient did exhibit a high degree of eosinophilia; however, she met all criteria for drug-induced SS, and the skin biopsy was not consistent with an eosinophilic process. Notably, the peripheral neutrophils were not elevated. Neutrophilia often is seen in classic SS but is not required for a diagnosis of drug-induced SS. Rare cases of dupilumab-associated arthritis and serum sickness–like reaction have been described,6-8 but our patient’s presentation was distinct, given other described signs, symptoms, and skin biopsy results. Histopathology results were not consistent with leukocytoclastic vasculitis, a potential mimicker of SS. Although the infectious and paraneoplastic evaluation was not exhaustive, the negative imaging from head to pelvis, the lack of recurrence of skin lesions, and the laboratory abnormalities after dupilumab discontinuation supported the conclusion that the culprit was not an infection or underlying malignancy. She had not started any other medications during this time frame, leaving dupilumab as the most likely offending agent. The mechanism for this reaction is not clear. It is possible that inhibition of IL-4 and IL-13 in the T helper 2 (TH2) cell pathway may have led to upregulated IL-17–mediated inflammation9 as well as a neutrophilic process in the skin, but this would not explain the concurrent peripheral eosinophilia that was noted. Further studies are needed to investigate the pathophysiology of SS.
Conclusion
We report a rare case of dupilumab-induced SS. Corticosteroids accompanied by cessation of the medication proved to be an effective treatment.
- Sweet RB. An acute febrile neutrophilic dermatosis. Br J Dermatol. 1964;76:349-356.
- Cohen PR, Kurzrock R. Sweet’s syndrome revisited: a review of disease concepts. Int J Dermatol. 2003;42:761-778.
- Walker DC, Cohen PR. Trimethoprim-sulfamethoxazole-associated acute febrile neutrophilic dermatosis: case report and review of drug-induced Sweet’s syndrome. J Am Acad Dermatol. 1996;34:918-923.
- Cohen PR. Sweet’s syndrome—a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. 2007;2:34.
- Jackson K, Bahna SL. Hypersensitivity and adverse reactions to biologics for asthma and allergic diseases. Expert Rev Clin Immunol. 2020;16:311-319.
- Willsmore ZN, Woolf RT, Hughes C, et al. Development of inflammatory arthritis and enthesitis in patients on dupilumab: a case series. Br J Dermatol. 2019;181:1068-1070.
- de Wijs LEM, van der Waa JD, de Jong PHP, et al. Acute arthritis and arthralgia as an adverse drug reaction to dupilumab. Clin Exp Dermatol. 2020;45:262-263.
- Treudler R, Delaroque N, Puder M, et al. Dupilumab-induced serum sickness-like reaction: an unusual adverse effect in a patient with atopic eczema. J Eur Acad Dermatol Venereol. 2021;35:E30-E32.
- Guenova E, Skabytska Y, Hoetzenecker W, et al. IL-4 abrogates TH17 cell-mediated inflammation by selective silencing of IL-23 in antigen-presenting cells. Proc Natl Acad Sci U S A. 2015;112:2163-2168.
Sweet syndrome (SS), also known as acute febrile neutrophilic dermatosis, was first described in 1964. 1 Since then, several subtypes of SS have been recognized, including classic or idiopathic, which typically follows an acute viral illness; cancer related, typically in the form of a paraneoplastic syndrome; and drug induced. 2 Drug-induced SS is defined by the following: (1) an abrupt onset of painful erythematous plaques or nodules; (2) histopathologic evidence of a dense neutrophilic infiltrate without evidence of leukocytoclastic vasculitis; (3) pyrexia above 38 ° C; (4) temporal relationship between drug and clinical presentation or temporally related recurrence after rechallenge; and (5) temporally related resolution of lesions after drug withdrawal or treatment with systemic corticosteroids. 3 All 5 criteria must be met to make a diagnosis of drug-induced SS. Since these criteria were established by Walker and Cohen, 3 various drugs have been identified as causative agents, including antibiotics, antiepileptics, antiretrovirals, antineoplastic agents, antipsychotics, oral contraceptives, nonsteroidal anti-inflammatory agents, and retinoids. 4 W e present a rare case of SS caused by dupilumab, a monoclonal antibody therapy, used in the treatment of severe eosinophilic asthma and atopic dermatitis.
Case Report
A 53-year-old woman presented with painful skin lesions, arthralgia, fever, and leukocytosis following initiation of dupilumab. She had a history of adult-onset, severe, persistent eosinophilic asthma, as well as chronic rhinosinusitis with nasal polyps, plaque psoriasis, and hypertrophic cardiomyopathy. She started mepolizumab 3 years prior to the current presentation for persistently uncontrolled asthma with a baseline peripheral eosinophil count of 860 cells/µL. After 3 years of minimal response to mepolizumab, she was started on dupilumab. Within 2 weeks of the first dose of dupilumab, she started experiencing bilateral knee pain. She subsequently developed daily fevers (temperature, 38.3 °C to 39.4 °C), fatigue, and pain in the back of the neck and head. After the second dose of dupilumab, she started experiencing painful skin lesions on the bilateral knuckles, elbows, and abdomen (Figure 1). She had difficulty using her hands and walking secondary to intense arthralgia involving the bilateral finger joints, elbows, and knees. Her primary care physician obtained a laboratory evaluation, which revealed an elevated total white blood cell count of 20×103/mm3 (reference range, 4–11×103/mm3) with 27.5% neutrophils and severely elevated eosinophils above her baseline to 57.3% with an absolute eosinophil count of 11,700 cells/µL (reference range, <400 cells/µL). Further assessment revealed an elevated erythrocyte sedimentation rate of 64 mm/h (reference range, 0–30 mm/h) and C-reactive protein level of 34 mg/dL (reference range, ≤0.80 mg/dL), with negative antinuclear antibody, rheumatoid factor, antineutrophilic cytoplasmic antibody, and Lyme antibody. IgG, IgA, and IgM levels were within reference range, and the IgE level was not elevated above her baseline. She had normal serum tryptase, and a peripheral D816V c-KIT mutation was not detected. She was subsequently hospitalized for further evaluation, at which time there was no fever or localizing infectious signs or symptoms. An infectious evaluation including urinalysis; respiratory swab for adenovirus, coronaviruses, human metapneumovirus, rhinovirus/enterovirus, influenza A and B, parainfluenza viruses, respiratory syncytial virus, Chlamydophila pneumoniae, and Mycoplasma pneumoniae; Lyme serology; and a computed tomography (CT) scan of the chest, abdomen, and pelvis revealed no evidence of infection. A parasite evaluation was ordered but was not performed. There was no evidence of malignancy on CT of the chest, abdomen, and pelvis or CT of the head without contrast. A lumbar puncture was considered but was ultimately deferred.

At the current presentation, the patient was following up in the dermatology clinic shortly after discharge. The lesions on the fingers and arms were described by the dermatologist as deep, erythematous, 0.5-cm bullous papules. The differential diagnosis at this time included a cutaneous or systemic infection, vasculitis, drug eruption, or cutaneous manifestation of an autoimmune condition. A shave biopsy of a skin lesion on the right hand demonstrated epidermal necrosis with a dense dermal neutrophilic infiltrate consistent with a neutrophilic dermatosis (Figure 2). There was no evidence of leukocytoclastic vasculitis. The histologic differential diagnosis included cutaneous infection, neutrophilic dermatosis of the hand, and SS. Special stains for infectious organisms including Gram, Grocott methenamine-silver, and auramine-rhodamine stains were negative for bacterial, fungal, and mycobacterial organisms, ruling out cutaneous infection. A diagnosis of drug-induced SS was made based on the histologic findings, diffuse distribution of the lesions, negative infectious evaluation, lack of underlying malignancy or autoimmune conditions, and onset following initiation of dupilumab.

Dupilumab was discontinued, and the patient was started on prednisone with rapid improvement in the symptoms. She underwent a slow taper of the prednisone over approximately 2 months with a slow downtrend of eosinophils. She was transitioned to a different biologic agent, benralizumab, with no further recurrence of the rash or arthralgia.
Comment
Dupilumab is a human monoclonal IgG4 antibody that inhibits IL-4 and IL-13 signaling by binding to the IL-4Rα subunit. By blocking IL-4Rα, dupilumab inhibits IL-4 and IL-13 cytokine-induced inflammatory responses, including the release of proinflammatory cytokines, chemokines, nitric oxide, and IgE. Currently, dupilumab is approved to treat refractory forms of moderate to severe asthma characterized by an eosinophilic phenotype or with corticosteroid-dependent asthma, moderate to severe atopic dermatitis, chronic rhinosinusitis with nasal polyposis, and eosinophilic esophagitis. The most common adverse events (incidence ≥1%) are injection-site reactions, oropharyngeal pain, and eosinophilia.5 Interestingly, our patient did exhibit a high degree of eosinophilia; however, she met all criteria for drug-induced SS, and the skin biopsy was not consistent with an eosinophilic process. Notably, the peripheral neutrophils were not elevated. Neutrophilia often is seen in classic SS but is not required for a diagnosis of drug-induced SS. Rare cases of dupilumab-associated arthritis and serum sickness–like reaction have been described,6-8 but our patient’s presentation was distinct, given other described signs, symptoms, and skin biopsy results. Histopathology results were not consistent with leukocytoclastic vasculitis, a potential mimicker of SS. Although the infectious and paraneoplastic evaluation was not exhaustive, the negative imaging from head to pelvis, the lack of recurrence of skin lesions, and the laboratory abnormalities after dupilumab discontinuation supported the conclusion that the culprit was not an infection or underlying malignancy. She had not started any other medications during this time frame, leaving dupilumab as the most likely offending agent. The mechanism for this reaction is not clear. It is possible that inhibition of IL-4 and IL-13 in the T helper 2 (TH2) cell pathway may have led to upregulated IL-17–mediated inflammation9 as well as a neutrophilic process in the skin, but this would not explain the concurrent peripheral eosinophilia that was noted. Further studies are needed to investigate the pathophysiology of SS.
Conclusion
We report a rare case of dupilumab-induced SS. Corticosteroids accompanied by cessation of the medication proved to be an effective treatment.
Sweet syndrome (SS), also known as acute febrile neutrophilic dermatosis, was first described in 1964. 1 Since then, several subtypes of SS have been recognized, including classic or idiopathic, which typically follows an acute viral illness; cancer related, typically in the form of a paraneoplastic syndrome; and drug induced. 2 Drug-induced SS is defined by the following: (1) an abrupt onset of painful erythematous plaques or nodules; (2) histopathologic evidence of a dense neutrophilic infiltrate without evidence of leukocytoclastic vasculitis; (3) pyrexia above 38 ° C; (4) temporal relationship between drug and clinical presentation or temporally related recurrence after rechallenge; and (5) temporally related resolution of lesions after drug withdrawal or treatment with systemic corticosteroids. 3 All 5 criteria must be met to make a diagnosis of drug-induced SS. Since these criteria were established by Walker and Cohen, 3 various drugs have been identified as causative agents, including antibiotics, antiepileptics, antiretrovirals, antineoplastic agents, antipsychotics, oral contraceptives, nonsteroidal anti-inflammatory agents, and retinoids. 4 W e present a rare case of SS caused by dupilumab, a monoclonal antibody therapy, used in the treatment of severe eosinophilic asthma and atopic dermatitis.
Case Report
A 53-year-old woman presented with painful skin lesions, arthralgia, fever, and leukocytosis following initiation of dupilumab. She had a history of adult-onset, severe, persistent eosinophilic asthma, as well as chronic rhinosinusitis with nasal polyps, plaque psoriasis, and hypertrophic cardiomyopathy. She started mepolizumab 3 years prior to the current presentation for persistently uncontrolled asthma with a baseline peripheral eosinophil count of 860 cells/µL. After 3 years of minimal response to mepolizumab, she was started on dupilumab. Within 2 weeks of the first dose of dupilumab, she started experiencing bilateral knee pain. She subsequently developed daily fevers (temperature, 38.3 °C to 39.4 °C), fatigue, and pain in the back of the neck and head. After the second dose of dupilumab, she started experiencing painful skin lesions on the bilateral knuckles, elbows, and abdomen (Figure 1). She had difficulty using her hands and walking secondary to intense arthralgia involving the bilateral finger joints, elbows, and knees. Her primary care physician obtained a laboratory evaluation, which revealed an elevated total white blood cell count of 20×103/mm3 (reference range, 4–11×103/mm3) with 27.5% neutrophils and severely elevated eosinophils above her baseline to 57.3% with an absolute eosinophil count of 11,700 cells/µL (reference range, <400 cells/µL). Further assessment revealed an elevated erythrocyte sedimentation rate of 64 mm/h (reference range, 0–30 mm/h) and C-reactive protein level of 34 mg/dL (reference range, ≤0.80 mg/dL), with negative antinuclear antibody, rheumatoid factor, antineutrophilic cytoplasmic antibody, and Lyme antibody. IgG, IgA, and IgM levels were within reference range, and the IgE level was not elevated above her baseline. She had normal serum tryptase, and a peripheral D816V c-KIT mutation was not detected. She was subsequently hospitalized for further evaluation, at which time there was no fever or localizing infectious signs or symptoms. An infectious evaluation including urinalysis; respiratory swab for adenovirus, coronaviruses, human metapneumovirus, rhinovirus/enterovirus, influenza A and B, parainfluenza viruses, respiratory syncytial virus, Chlamydophila pneumoniae, and Mycoplasma pneumoniae; Lyme serology; and a computed tomography (CT) scan of the chest, abdomen, and pelvis revealed no evidence of infection. A parasite evaluation was ordered but was not performed. There was no evidence of malignancy on CT of the chest, abdomen, and pelvis or CT of the head without contrast. A lumbar puncture was considered but was ultimately deferred.

At the current presentation, the patient was following up in the dermatology clinic shortly after discharge. The lesions on the fingers and arms were described by the dermatologist as deep, erythematous, 0.5-cm bullous papules. The differential diagnosis at this time included a cutaneous or systemic infection, vasculitis, drug eruption, or cutaneous manifestation of an autoimmune condition. A shave biopsy of a skin lesion on the right hand demonstrated epidermal necrosis with a dense dermal neutrophilic infiltrate consistent with a neutrophilic dermatosis (Figure 2). There was no evidence of leukocytoclastic vasculitis. The histologic differential diagnosis included cutaneous infection, neutrophilic dermatosis of the hand, and SS. Special stains for infectious organisms including Gram, Grocott methenamine-silver, and auramine-rhodamine stains were negative for bacterial, fungal, and mycobacterial organisms, ruling out cutaneous infection. A diagnosis of drug-induced SS was made based on the histologic findings, diffuse distribution of the lesions, negative infectious evaluation, lack of underlying malignancy or autoimmune conditions, and onset following initiation of dupilumab.

Dupilumab was discontinued, and the patient was started on prednisone with rapid improvement in the symptoms. She underwent a slow taper of the prednisone over approximately 2 months with a slow downtrend of eosinophils. She was transitioned to a different biologic agent, benralizumab, with no further recurrence of the rash or arthralgia.
Comment
Dupilumab is a human monoclonal IgG4 antibody that inhibits IL-4 and IL-13 signaling by binding to the IL-4Rα subunit. By blocking IL-4Rα, dupilumab inhibits IL-4 and IL-13 cytokine-induced inflammatory responses, including the release of proinflammatory cytokines, chemokines, nitric oxide, and IgE. Currently, dupilumab is approved to treat refractory forms of moderate to severe asthma characterized by an eosinophilic phenotype or with corticosteroid-dependent asthma, moderate to severe atopic dermatitis, chronic rhinosinusitis with nasal polyposis, and eosinophilic esophagitis. The most common adverse events (incidence ≥1%) are injection-site reactions, oropharyngeal pain, and eosinophilia.5 Interestingly, our patient did exhibit a high degree of eosinophilia; however, she met all criteria for drug-induced SS, and the skin biopsy was not consistent with an eosinophilic process. Notably, the peripheral neutrophils were not elevated. Neutrophilia often is seen in classic SS but is not required for a diagnosis of drug-induced SS. Rare cases of dupilumab-associated arthritis and serum sickness–like reaction have been described,6-8 but our patient’s presentation was distinct, given other described signs, symptoms, and skin biopsy results. Histopathology results were not consistent with leukocytoclastic vasculitis, a potential mimicker of SS. Although the infectious and paraneoplastic evaluation was not exhaustive, the negative imaging from head to pelvis, the lack of recurrence of skin lesions, and the laboratory abnormalities after dupilumab discontinuation supported the conclusion that the culprit was not an infection or underlying malignancy. She had not started any other medications during this time frame, leaving dupilumab as the most likely offending agent. The mechanism for this reaction is not clear. It is possible that inhibition of IL-4 and IL-13 in the T helper 2 (TH2) cell pathway may have led to upregulated IL-17–mediated inflammation9 as well as a neutrophilic process in the skin, but this would not explain the concurrent peripheral eosinophilia that was noted. Further studies are needed to investigate the pathophysiology of SS.
Conclusion
We report a rare case of dupilumab-induced SS. Corticosteroids accompanied by cessation of the medication proved to be an effective treatment.
- Sweet RB. An acute febrile neutrophilic dermatosis. Br J Dermatol. 1964;76:349-356.
- Cohen PR, Kurzrock R. Sweet’s syndrome revisited: a review of disease concepts. Int J Dermatol. 2003;42:761-778.
- Walker DC, Cohen PR. Trimethoprim-sulfamethoxazole-associated acute febrile neutrophilic dermatosis: case report and review of drug-induced Sweet’s syndrome. J Am Acad Dermatol. 1996;34:918-923.
- Cohen PR. Sweet’s syndrome—a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. 2007;2:34.
- Jackson K, Bahna SL. Hypersensitivity and adverse reactions to biologics for asthma and allergic diseases. Expert Rev Clin Immunol. 2020;16:311-319.
- Willsmore ZN, Woolf RT, Hughes C, et al. Development of inflammatory arthritis and enthesitis in patients on dupilumab: a case series. Br J Dermatol. 2019;181:1068-1070.
- de Wijs LEM, van der Waa JD, de Jong PHP, et al. Acute arthritis and arthralgia as an adverse drug reaction to dupilumab. Clin Exp Dermatol. 2020;45:262-263.
- Treudler R, Delaroque N, Puder M, et al. Dupilumab-induced serum sickness-like reaction: an unusual adverse effect in a patient with atopic eczema. J Eur Acad Dermatol Venereol. 2021;35:E30-E32.
- Guenova E, Skabytska Y, Hoetzenecker W, et al. IL-4 abrogates TH17 cell-mediated inflammation by selective silencing of IL-23 in antigen-presenting cells. Proc Natl Acad Sci U S A. 2015;112:2163-2168.
- Sweet RB. An acute febrile neutrophilic dermatosis. Br J Dermatol. 1964;76:349-356.
- Cohen PR, Kurzrock R. Sweet’s syndrome revisited: a review of disease concepts. Int J Dermatol. 2003;42:761-778.
- Walker DC, Cohen PR. Trimethoprim-sulfamethoxazole-associated acute febrile neutrophilic dermatosis: case report and review of drug-induced Sweet’s syndrome. J Am Acad Dermatol. 1996;34:918-923.
- Cohen PR. Sweet’s syndrome—a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. 2007;2:34.
- Jackson K, Bahna SL. Hypersensitivity and adverse reactions to biologics for asthma and allergic diseases. Expert Rev Clin Immunol. 2020;16:311-319.
- Willsmore ZN, Woolf RT, Hughes C, et al. Development of inflammatory arthritis and enthesitis in patients on dupilumab: a case series. Br J Dermatol. 2019;181:1068-1070.
- de Wijs LEM, van der Waa JD, de Jong PHP, et al. Acute arthritis and arthralgia as an adverse drug reaction to dupilumab. Clin Exp Dermatol. 2020;45:262-263.
- Treudler R, Delaroque N, Puder M, et al. Dupilumab-induced serum sickness-like reaction: an unusual adverse effect in a patient with atopic eczema. J Eur Acad Dermatol Venereol. 2021;35:E30-E32.
- Guenova E, Skabytska Y, Hoetzenecker W, et al. IL-4 abrogates TH17 cell-mediated inflammation by selective silencing of IL-23 in antigen-presenting cells. Proc Natl Acad Sci U S A. 2015;112:2163-2168.
Practice Points
- Prescribers of dupilumab should be aware that Sweet syndrome is a potential adverse reaction.
- Sweet syndrome should be suspected if there is abrupt onset of painful erythematous plaques or nodules accompanied by pyrexia following injection of dupilumab. Biopsy of the nodules should be obtained to confirm the diagnosis.
- Systemic corticosteroids with cessation of dupilumab are effective treatments.
- Following treatment, dupilumab should not be reinitiated, and alternative therapies should be used.
Commentary: Topical treatments, dupilumab, and long-term treatment of AD, July 2023
Silverberg and colleagues described a very well-designed, vehicle-controlled, randomized 8-week study of a topical formulation of a purified strain of Nitrosomonas eutropha, an ammonia-oxidizing bacterium. In theory, this bacterium may reduce Staphylococcus aureus. The study compared two concentrations of the bacterium vs vehicle delivered as a spray twice per day. Study participants were adults with AD affecting 10%-40% of body surface area.
The study found "meaningful" improvements in itch and objective signs of disease, with clear separation between both doses of the bacterial spray compared with vehicle. At week 4, about 23% of participants treated with the bacterium were clear or almost clear (with a 2-point improvement) compared with 12% in the vehicle group (for comparison, in a phase 2 study comparing topical ruxolitinib with 0.1% triamcinolone cream, there was a 25% clear or almost clear rate [with 2-point improvement] in the triamcinolone-treated individuals).
Though an "all-natural" bacterial approach to managing AD may be appealing to some, it sounded like magic to me. But this well-done study makes it seem like the bacterial approach could be more promising than I had thought. This study also reported about twice as many adverse events (including gastrointestinal issues) with the bacterium-treated participants compared with those who received vehicle, adding to my belief that the bacterial product has efficacy. Whether any other topical will be more effective and safer than is topical triamcinolone remains to be seen. I'm still pessimistic about topicals because of patients' poor adherence to topical treatment, but perhaps an easy-to-use spray that isn't associated with patients' fear of "steroids" will be helpful.
Chen and colleagues. They analyzed data on hundreds of thousands of patients with and without AD. Adults with AD had a "significantly increased risk" of developing venous thromboembolism compared with adults without AD. The huge sample size of their study seems compelling. That huge sample size allows detection of effects so small that they may be clinically insignificant.
They report that patients with AD had a venous thromboembolism at a rate of 1.05/1000 patients-years; the rate was 0.82 for patients without AD. From that, we can calculate that there would be an additional 23 patients with venous thromboembolism for every 100,000 patient-years or about one more venous thromboembolism in the AD group in every 4000 patient-years. Though the finding was statistically significant, I don't think it is clinically meaningful.
The authors correctly conclude that "vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients with AD who present with relevant symptoms (eg, unexplained dyspnea, chest tightness, and limb swelling)." But it is probably also true that vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients without AD who present with those symptoms. I think the authors might have been on solid ground if they had concluded that there was a statistically significant but clinically insignificant increased risk for venous thromboembolism in patients with AD.
Eichenfeld and colleagues examined the use of topical crisaborole once per day as a maintenance treatment for patients with mild to moderate AD. The study compared patients given topical crisaborole with those randomly assigned to vehicle. The active treatment was effective because topical crisaborole treated patients had longer times to the first flare following treatment and fewer flares over the 1 year of treatment. The differences were not huge, but I think they were clinically meaningful. I'm guessing that the topical crisaborole maintenance treatment would have been even more effective had it been used regularly. The study did not, as far as I could tell, assess how well the treatment was used.
An interesting aspect of this study is that it began with nearly 500 participants who started on twice daily topical crisaborole. The 270 patients who responded to the treatment (achieving clear or almost clear with at least a 2-point improvement) were enrolled in the 1-year maintenance phase. Thus, the participants in the maintenance phase were preselected for patients who respond to topical crisaborole. We don't know why they were responders (I, of course, expect it is because they selected for patients who are better than others are at using a topical treatment), but it may be best not to try to generalize these results and assume this form of maintenance treatment would work equally well in a population who achieve initial success with an oral therapy regimen (for example, a quick course of oral prednisone).
Dupilumab was a revolutionary treatment for AD. I didn't think that I'd ever see a more effective treatment. It's so safe too! It has been a first-line treatment for AD since its introduction. Now, we also have oral Janus kinase inhibitor options. Blauvelt and colleagues examined what happens when patients who have been on dupilumab are switched to a high dose (30 mg/d) of upadacitinib (the standard starting dose of upadacitinib is 15 mg/d). Though dupilumab is very effective, upadacitinib is more so. After 4 weeks of switching to upadacitinib, nearly half the patients were completely clear of AD compared with only 16.0% after 24 weeks of dupilumab! The authors point out, optimistically, that "No new safety risks were observed." Though there were no cancers, gastrointestinal perforations, major adverse cardiovascular events, or venous thromboembolic events, there were cases of eczema herpeticum and zoster in patients treated with upadacitinib. Having upadacitinib available for patients who fail dupilumab is a clear benefit; the role of upadacitinib before dupilumab seems less clear.
Patients doing great on dupilumab for AD may be wondering: Do I still need to take it every 2 weeks? Spekhorst and colleagues may have the answer. They describe the response to tapering dupilumab in patients who had been on the drug for at least 1 year with well-controlled disease for at least 6 months. Patients in the study then continued dupilumab with the longest possible dosing interval while maintaining control of their AD.
Generally, patients maintained good control of their AD, with only a small increase in mean disease severity and in concomitant use of topical steroids. For the patients who attempted prolongation, 83% successfully continued dupilumab treatment with a prolonged interval. Not at all surprisingly, the authors calculated that prolonging the interval between dosing led to large savings in cost.
One of the nice features of dupilumab treatment is that loss of response over time seems unusual. Perhaps there is a low propensity for forming antidrug antibodies when dupilumab is used in the standard every 2-week dosing regimen. I don't know whether antidrug antibodies would be more likely with the intermittent dosing regimen. But now that we have other good systemic treatment options for AD, losing dupilumab efficacy would not be as critical a problem as it used to be. I also want to point out that patients' adherence to injection treatment, though better than adherence to topicals, is far from perfect. It's likely that many patients have already been prolonging the interval between taking their treatments. If you want to know, just ask them. The way I like to phrase the question is: "Are you keeping the extra injectors you've accumulated refrigerated like you are supposed to?"
Silverberg and colleagues described a very well-designed, vehicle-controlled, randomized 8-week study of a topical formulation of a purified strain of Nitrosomonas eutropha, an ammonia-oxidizing bacterium. In theory, this bacterium may reduce Staphylococcus aureus. The study compared two concentrations of the bacterium vs vehicle delivered as a spray twice per day. Study participants were adults with AD affecting 10%-40% of body surface area.
The study found "meaningful" improvements in itch and objective signs of disease, with clear separation between both doses of the bacterial spray compared with vehicle. At week 4, about 23% of participants treated with the bacterium were clear or almost clear (with a 2-point improvement) compared with 12% in the vehicle group (for comparison, in a phase 2 study comparing topical ruxolitinib with 0.1% triamcinolone cream, there was a 25% clear or almost clear rate [with 2-point improvement] in the triamcinolone-treated individuals).
Though an "all-natural" bacterial approach to managing AD may be appealing to some, it sounded like magic to me. But this well-done study makes it seem like the bacterial approach could be more promising than I had thought. This study also reported about twice as many adverse events (including gastrointestinal issues) with the bacterium-treated participants compared with those who received vehicle, adding to my belief that the bacterial product has efficacy. Whether any other topical will be more effective and safer than is topical triamcinolone remains to be seen. I'm still pessimistic about topicals because of patients' poor adherence to topical treatment, but perhaps an easy-to-use spray that isn't associated with patients' fear of "steroids" will be helpful.
Chen and colleagues. They analyzed data on hundreds of thousands of patients with and without AD. Adults with AD had a "significantly increased risk" of developing venous thromboembolism compared with adults without AD. The huge sample size of their study seems compelling. That huge sample size allows detection of effects so small that they may be clinically insignificant.
They report that patients with AD had a venous thromboembolism at a rate of 1.05/1000 patients-years; the rate was 0.82 for patients without AD. From that, we can calculate that there would be an additional 23 patients with venous thromboembolism for every 100,000 patient-years or about one more venous thromboembolism in the AD group in every 4000 patient-years. Though the finding was statistically significant, I don't think it is clinically meaningful.
The authors correctly conclude that "vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients with AD who present with relevant symptoms (eg, unexplained dyspnea, chest tightness, and limb swelling)." But it is probably also true that vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients without AD who present with those symptoms. I think the authors might have been on solid ground if they had concluded that there was a statistically significant but clinically insignificant increased risk for venous thromboembolism in patients with AD.
Eichenfeld and colleagues examined the use of topical crisaborole once per day as a maintenance treatment for patients with mild to moderate AD. The study compared patients given topical crisaborole with those randomly assigned to vehicle. The active treatment was effective because topical crisaborole treated patients had longer times to the first flare following treatment and fewer flares over the 1 year of treatment. The differences were not huge, but I think they were clinically meaningful. I'm guessing that the topical crisaborole maintenance treatment would have been even more effective had it been used regularly. The study did not, as far as I could tell, assess how well the treatment was used.
An interesting aspect of this study is that it began with nearly 500 participants who started on twice daily topical crisaborole. The 270 patients who responded to the treatment (achieving clear or almost clear with at least a 2-point improvement) were enrolled in the 1-year maintenance phase. Thus, the participants in the maintenance phase were preselected for patients who respond to topical crisaborole. We don't know why they were responders (I, of course, expect it is because they selected for patients who are better than others are at using a topical treatment), but it may be best not to try to generalize these results and assume this form of maintenance treatment would work equally well in a population who achieve initial success with an oral therapy regimen (for example, a quick course of oral prednisone).
Dupilumab was a revolutionary treatment for AD. I didn't think that I'd ever see a more effective treatment. It's so safe too! It has been a first-line treatment for AD since its introduction. Now, we also have oral Janus kinase inhibitor options. Blauvelt and colleagues examined what happens when patients who have been on dupilumab are switched to a high dose (30 mg/d) of upadacitinib (the standard starting dose of upadacitinib is 15 mg/d). Though dupilumab is very effective, upadacitinib is more so. After 4 weeks of switching to upadacitinib, nearly half the patients were completely clear of AD compared with only 16.0% after 24 weeks of dupilumab! The authors point out, optimistically, that "No new safety risks were observed." Though there were no cancers, gastrointestinal perforations, major adverse cardiovascular events, or venous thromboembolic events, there were cases of eczema herpeticum and zoster in patients treated with upadacitinib. Having upadacitinib available for patients who fail dupilumab is a clear benefit; the role of upadacitinib before dupilumab seems less clear.
Patients doing great on dupilumab for AD may be wondering: Do I still need to take it every 2 weeks? Spekhorst and colleagues may have the answer. They describe the response to tapering dupilumab in patients who had been on the drug for at least 1 year with well-controlled disease for at least 6 months. Patients in the study then continued dupilumab with the longest possible dosing interval while maintaining control of their AD.
Generally, patients maintained good control of their AD, with only a small increase in mean disease severity and in concomitant use of topical steroids. For the patients who attempted prolongation, 83% successfully continued dupilumab treatment with a prolonged interval. Not at all surprisingly, the authors calculated that prolonging the interval between dosing led to large savings in cost.
One of the nice features of dupilumab treatment is that loss of response over time seems unusual. Perhaps there is a low propensity for forming antidrug antibodies when dupilumab is used in the standard every 2-week dosing regimen. I don't know whether antidrug antibodies would be more likely with the intermittent dosing regimen. But now that we have other good systemic treatment options for AD, losing dupilumab efficacy would not be as critical a problem as it used to be. I also want to point out that patients' adherence to injection treatment, though better than adherence to topicals, is far from perfect. It's likely that many patients have already been prolonging the interval between taking their treatments. If you want to know, just ask them. The way I like to phrase the question is: "Are you keeping the extra injectors you've accumulated refrigerated like you are supposed to?"
Silverberg and colleagues described a very well-designed, vehicle-controlled, randomized 8-week study of a topical formulation of a purified strain of Nitrosomonas eutropha, an ammonia-oxidizing bacterium. In theory, this bacterium may reduce Staphylococcus aureus. The study compared two concentrations of the bacterium vs vehicle delivered as a spray twice per day. Study participants were adults with AD affecting 10%-40% of body surface area.
The study found "meaningful" improvements in itch and objective signs of disease, with clear separation between both doses of the bacterial spray compared with vehicle. At week 4, about 23% of participants treated with the bacterium were clear or almost clear (with a 2-point improvement) compared with 12% in the vehicle group (for comparison, in a phase 2 study comparing topical ruxolitinib with 0.1% triamcinolone cream, there was a 25% clear or almost clear rate [with 2-point improvement] in the triamcinolone-treated individuals).
Though an "all-natural" bacterial approach to managing AD may be appealing to some, it sounded like magic to me. But this well-done study makes it seem like the bacterial approach could be more promising than I had thought. This study also reported about twice as many adverse events (including gastrointestinal issues) with the bacterium-treated participants compared with those who received vehicle, adding to my belief that the bacterial product has efficacy. Whether any other topical will be more effective and safer than is topical triamcinolone remains to be seen. I'm still pessimistic about topicals because of patients' poor adherence to topical treatment, but perhaps an easy-to-use spray that isn't associated with patients' fear of "steroids" will be helpful.
Chen and colleagues. They analyzed data on hundreds of thousands of patients with and without AD. Adults with AD had a "significantly increased risk" of developing venous thromboembolism compared with adults without AD. The huge sample size of their study seems compelling. That huge sample size allows detection of effects so small that they may be clinically insignificant.
They report that patients with AD had a venous thromboembolism at a rate of 1.05/1000 patients-years; the rate was 0.82 for patients without AD. From that, we can calculate that there would be an additional 23 patients with venous thromboembolism for every 100,000 patient-years or about one more venous thromboembolism in the AD group in every 4000 patient-years. Though the finding was statistically significant, I don't think it is clinically meaningful.
The authors correctly conclude that "vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients with AD who present with relevant symptoms (eg, unexplained dyspnea, chest tightness, and limb swelling)." But it is probably also true that vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients without AD who present with those symptoms. I think the authors might have been on solid ground if they had concluded that there was a statistically significant but clinically insignificant increased risk for venous thromboembolism in patients with AD.
Eichenfeld and colleagues examined the use of topical crisaborole once per day as a maintenance treatment for patients with mild to moderate AD. The study compared patients given topical crisaborole with those randomly assigned to vehicle. The active treatment was effective because topical crisaborole treated patients had longer times to the first flare following treatment and fewer flares over the 1 year of treatment. The differences were not huge, but I think they were clinically meaningful. I'm guessing that the topical crisaborole maintenance treatment would have been even more effective had it been used regularly. The study did not, as far as I could tell, assess how well the treatment was used.
An interesting aspect of this study is that it began with nearly 500 participants who started on twice daily topical crisaborole. The 270 patients who responded to the treatment (achieving clear or almost clear with at least a 2-point improvement) were enrolled in the 1-year maintenance phase. Thus, the participants in the maintenance phase were preselected for patients who respond to topical crisaborole. We don't know why they were responders (I, of course, expect it is because they selected for patients who are better than others are at using a topical treatment), but it may be best not to try to generalize these results and assume this form of maintenance treatment would work equally well in a population who achieve initial success with an oral therapy regimen (for example, a quick course of oral prednisone).
Dupilumab was a revolutionary treatment for AD. I didn't think that I'd ever see a more effective treatment. It's so safe too! It has been a first-line treatment for AD since its introduction. Now, we also have oral Janus kinase inhibitor options. Blauvelt and colleagues examined what happens when patients who have been on dupilumab are switched to a high dose (30 mg/d) of upadacitinib (the standard starting dose of upadacitinib is 15 mg/d). Though dupilumab is very effective, upadacitinib is more so. After 4 weeks of switching to upadacitinib, nearly half the patients were completely clear of AD compared with only 16.0% after 24 weeks of dupilumab! The authors point out, optimistically, that "No new safety risks were observed." Though there were no cancers, gastrointestinal perforations, major adverse cardiovascular events, or venous thromboembolic events, there were cases of eczema herpeticum and zoster in patients treated with upadacitinib. Having upadacitinib available for patients who fail dupilumab is a clear benefit; the role of upadacitinib before dupilumab seems less clear.
Patients doing great on dupilumab for AD may be wondering: Do I still need to take it every 2 weeks? Spekhorst and colleagues may have the answer. They describe the response to tapering dupilumab in patients who had been on the drug for at least 1 year with well-controlled disease for at least 6 months. Patients in the study then continued dupilumab with the longest possible dosing interval while maintaining control of their AD.
Generally, patients maintained good control of their AD, with only a small increase in mean disease severity and in concomitant use of topical steroids. For the patients who attempted prolongation, 83% successfully continued dupilumab treatment with a prolonged interval. Not at all surprisingly, the authors calculated that prolonging the interval between dosing led to large savings in cost.
One of the nice features of dupilumab treatment is that loss of response over time seems unusual. Perhaps there is a low propensity for forming antidrug antibodies when dupilumab is used in the standard every 2-week dosing regimen. I don't know whether antidrug antibodies would be more likely with the intermittent dosing regimen. But now that we have other good systemic treatment options for AD, losing dupilumab efficacy would not be as critical a problem as it used to be. I also want to point out that patients' adherence to injection treatment, though better than adherence to topicals, is far from perfect. It's likely that many patients have already been prolonging the interval between taking their treatments. If you want to know, just ask them. The way I like to phrase the question is: "Are you keeping the extra injectors you've accumulated refrigerated like you are supposed to?"
Commentary: Topical treatments, dupilumab, and long-term treatment of AD, July 2023
There is a tremendous amount of atopic dermatitis (AD) research underway. This month, we have several interesting articles to present.
Silverberg and colleagues described a very well-designed, vehicle-controlled, randomized 8-week study of a topical formulation of a purified strain of Nitrosomonas eutropha, an ammonia-oxidizing bacterium. In theory, this bacterium may reduce Staphylococcus aureus. The study compared two concentrations of the bacterium vs vehicle delivered as a spray twice per day. Study participants were adults with AD affecting 10%-40% of body surface area.
The study found "meaningful" improvements in itch and objective signs of disease, with clear separation between both doses of the bacterial spray compared with vehicle. At week 4, about 23% of participants treated with the bacterium were clear or almost clear (with a 2-point improvement) compared with 12% in the vehicle group (for comparison, in a phase 2 study comparing topical ruxolitinib with 0.1% triamcinolone cream, there was a 25% clear or almost clear rate [with 2-point improvement] in the triamcinolone-treated individuals).
Though an "all-natural" bacterial approach to managing AD may be appealing to some, it sounded like magic to me. But this well-done study makes it seem like the bacterial approach could be more promising than I had thought. This study also reported about twice as many adverse events (including gastrointestinal issues) with the bacterium-treated participants compared with those who received vehicle, adding to my belief that the bacterial product has efficacy. Whether any other topical will be more effective and safer than is topical triamcinolone remains to be seen. I'm still pessimistic about topicals because of patients' poor adherence to topical treatment, but perhaps an easy-to-use spray that isn't associated with patients' fear of "steroids" will be helpful.
I love articles like this one from Chen and colleagues. They analyzed data on hundreds of thousands of patients with and without AD. Adults with AD had a "significantly increased risk" of developing venous thromboembolism compared with adults without AD. The huge sample size of their study seems compelling. That huge sample size allows detection of effects so small that they may be clinically insignificant.
They report that patients with AD had a venous thromboembolism at a rate of 1.05/1000 patients-years; the rate was 0.82 for patients without AD. From that, we can calculate that there would be an additional 23 patients with venous thromboembolism for every 100,000 patient-years or about one more venous thromboembolism in the AD group in every 4000 patient-years. Though the finding was statistically significant, I don't think it is clinically meaningful.
The authors correctly conclude that "vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients with AD who present with relevant symptoms (eg, unexplained dyspnea, chest tightness, and limb swelling)." But it is probably also true that vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients without AD who present with those symptoms. I think the authors might have been on solid ground if they had concluded that there was a statistically significant but clinically insignificant increased risk for venous thromboembolism in patients with AD.
Eichenfeld and colleagues examined the use of topical crisaborole once per day as a maintenance treatment for patients with mild to moderate AD. The study compared patients given topical crisaborole with those randomly assigned to vehicle. The active treatment was effective because topical crisaborole treated patients had longer times to the first flare following treatment and fewer flares over the 1 year of treatment. The differences were not huge, but I think they were clinically meaningful. I'm guessing that the topical crisaborole maintenance treatment would have been even more effective had it been used regularly. The study did not, as far as I could tell, assess how well the treatment was used.
An interesting aspect of this study is that it began with nearly 500 participants who started on twice daily topical crisaborole. The 270 patients who responded to the treatment (achieving clear or almost clear with at least a 2-point improvement) were enrolled in the 1-year maintenance phase. Thus, the participants in the maintenance phase were preselected for patients who respond to topical crisaborole. We don't know why they were responders (I, of course, expect it is because they selected for patients who are better than others are at using a topical treatment), but it may be best not to try to generalize these results and assume this form of maintenance treatment would work equally well in a population who achieve initial success with an oral therapy regimen (for example, a quick course of oral prednisone).
Dupilumab was a revolutionary treatment for AD. I didn't think that I'd ever see a more effective treatment. It's so safe too! It has been a first-line treatment for AD since its introduction. Now, we also have oral Janus kinase inhibitor options. Blauvelt and colleagues examined what happens when patients who have been on dupilumab are switched to a high dose (30 mg/d) of upadacitinib (the standard starting dose of upadacitinib is 15 mg/d). Though dupilumab is very effective, upadacitinib is more so. After 4 weeks of switching to upadacitinib, nearly half the patients were completely clear of AD compared with only 16.0% after 24 weeks of dupilumab! The authors point out, optimistically, that "No new safety risks were observed." Though there were no cancers, gastrointestinal perforations, major adverse cardiovascular events, or venous thromboembolic events, there were cases of eczema herpeticum and zoster in patients treated with upadacitinib. Having upadacitinib available for patients who fail dupilumab is a clear benefit; the role of upadacitinib before dupilumab seems less clear.
Patients doing great on dupilumab for AD may be wondering: Do I still need to take it every 2 weeks? Spekhorst and colleagues may have the answer. They describe the response to tapering dupilumab in patients who had been on the drug for at least 1 year with well-controlled disease for at least 6 months. Patients in the study then continued dupilumab with the longest possible dosing interval while maintaining control of their AD.
Generally, patients maintained good control of their AD, with only a small increase in mean disease severity and in concomitant use of topical steroids. For the patients who attempted prolongation, 83% successfully continued dupilumab treatment with a prolonged interval. Not at all surprisingly, the authors calculated that prolonging the interval between dosing led to large savings in cost.
One of the nice features of dupilumab treatment is that loss of response over time seems unusual. Perhaps there is a low propensity for forming antidrug antibodies when dupilumab is used in the standard every 2-week dosing regimen. I don't know whether antidrug antibodies would be more likely with the intermittent dosing regimen. But now that we have other good systemic treatment options for AD, losing dupilumab efficacy would not be as critical a problem as it used to be. I also want to point out that patients' adherence to injection treatment, though better than adherence to topicals, is far from perfect. It's likely that many patients have already been prolonging the interval between taking their treatments. If you want to know, just ask them. The way I like to phrase the question is: "Are you keeping the extra injectors you've accumulated refrigerated like you are supposed to?"
There is a tremendous amount of atopic dermatitis (AD) research underway. This month, we have several interesting articles to present.
Silverberg and colleagues described a very well-designed, vehicle-controlled, randomized 8-week study of a topical formulation of a purified strain of Nitrosomonas eutropha, an ammonia-oxidizing bacterium. In theory, this bacterium may reduce Staphylococcus aureus. The study compared two concentrations of the bacterium vs vehicle delivered as a spray twice per day. Study participants were adults with AD affecting 10%-40% of body surface area.
The study found "meaningful" improvements in itch and objective signs of disease, with clear separation between both doses of the bacterial spray compared with vehicle. At week 4, about 23% of participants treated with the bacterium were clear or almost clear (with a 2-point improvement) compared with 12% in the vehicle group (for comparison, in a phase 2 study comparing topical ruxolitinib with 0.1% triamcinolone cream, there was a 25% clear or almost clear rate [with 2-point improvement] in the triamcinolone-treated individuals).
Though an "all-natural" bacterial approach to managing AD may be appealing to some, it sounded like magic to me. But this well-done study makes it seem like the bacterial approach could be more promising than I had thought. This study also reported about twice as many adverse events (including gastrointestinal issues) with the bacterium-treated participants compared with those who received vehicle, adding to my belief that the bacterial product has efficacy. Whether any other topical will be more effective and safer than is topical triamcinolone remains to be seen. I'm still pessimistic about topicals because of patients' poor adherence to topical treatment, but perhaps an easy-to-use spray that isn't associated with patients' fear of "steroids" will be helpful.
I love articles like this one from Chen and colleagues. They analyzed data on hundreds of thousands of patients with and without AD. Adults with AD had a "significantly increased risk" of developing venous thromboembolism compared with adults without AD. The huge sample size of their study seems compelling. That huge sample size allows detection of effects so small that they may be clinically insignificant.
They report that patients with AD had a venous thromboembolism at a rate of 1.05/1000 patients-years; the rate was 0.82 for patients without AD. From that, we can calculate that there would be an additional 23 patients with venous thromboembolism for every 100,000 patient-years or about one more venous thromboembolism in the AD group in every 4000 patient-years. Though the finding was statistically significant, I don't think it is clinically meaningful.
The authors correctly conclude that "vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients with AD who present with relevant symptoms (eg, unexplained dyspnea, chest tightness, and limb swelling)." But it is probably also true that vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients without AD who present with those symptoms. I think the authors might have been on solid ground if they had concluded that there was a statistically significant but clinically insignificant increased risk for venous thromboembolism in patients with AD.
Eichenfeld and colleagues examined the use of topical crisaborole once per day as a maintenance treatment for patients with mild to moderate AD. The study compared patients given topical crisaborole with those randomly assigned to vehicle. The active treatment was effective because topical crisaborole treated patients had longer times to the first flare following treatment and fewer flares over the 1 year of treatment. The differences were not huge, but I think they were clinically meaningful. I'm guessing that the topical crisaborole maintenance treatment would have been even more effective had it been used regularly. The study did not, as far as I could tell, assess how well the treatment was used.
An interesting aspect of this study is that it began with nearly 500 participants who started on twice daily topical crisaborole. The 270 patients who responded to the treatment (achieving clear or almost clear with at least a 2-point improvement) were enrolled in the 1-year maintenance phase. Thus, the participants in the maintenance phase were preselected for patients who respond to topical crisaborole. We don't know why they were responders (I, of course, expect it is because they selected for patients who are better than others are at using a topical treatment), but it may be best not to try to generalize these results and assume this form of maintenance treatment would work equally well in a population who achieve initial success with an oral therapy regimen (for example, a quick course of oral prednisone).
Dupilumab was a revolutionary treatment for AD. I didn't think that I'd ever see a more effective treatment. It's so safe too! It has been a first-line treatment for AD since its introduction. Now, we also have oral Janus kinase inhibitor options. Blauvelt and colleagues examined what happens when patients who have been on dupilumab are switched to a high dose (30 mg/d) of upadacitinib (the standard starting dose of upadacitinib is 15 mg/d). Though dupilumab is very effective, upadacitinib is more so. After 4 weeks of switching to upadacitinib, nearly half the patients were completely clear of AD compared with only 16.0% after 24 weeks of dupilumab! The authors point out, optimistically, that "No new safety risks were observed." Though there were no cancers, gastrointestinal perforations, major adverse cardiovascular events, or venous thromboembolic events, there were cases of eczema herpeticum and zoster in patients treated with upadacitinib. Having upadacitinib available for patients who fail dupilumab is a clear benefit; the role of upadacitinib before dupilumab seems less clear.
Patients doing great on dupilumab for AD may be wondering: Do I still need to take it every 2 weeks? Spekhorst and colleagues may have the answer. They describe the response to tapering dupilumab in patients who had been on the drug for at least 1 year with well-controlled disease for at least 6 months. Patients in the study then continued dupilumab with the longest possible dosing interval while maintaining control of their AD.
Generally, patients maintained good control of their AD, with only a small increase in mean disease severity and in concomitant use of topical steroids. For the patients who attempted prolongation, 83% successfully continued dupilumab treatment with a prolonged interval. Not at all surprisingly, the authors calculated that prolonging the interval between dosing led to large savings in cost.
One of the nice features of dupilumab treatment is that loss of response over time seems unusual. Perhaps there is a low propensity for forming antidrug antibodies when dupilumab is used in the standard every 2-week dosing regimen. I don't know whether antidrug antibodies would be more likely with the intermittent dosing regimen. But now that we have other good systemic treatment options for AD, losing dupilumab efficacy would not be as critical a problem as it used to be. I also want to point out that patients' adherence to injection treatment, though better than adherence to topicals, is far from perfect. It's likely that many patients have already been prolonging the interval between taking their treatments. If you want to know, just ask them. The way I like to phrase the question is: "Are you keeping the extra injectors you've accumulated refrigerated like you are supposed to?"
There is a tremendous amount of atopic dermatitis (AD) research underway. This month, we have several interesting articles to present.
Silverberg and colleagues described a very well-designed, vehicle-controlled, randomized 8-week study of a topical formulation of a purified strain of Nitrosomonas eutropha, an ammonia-oxidizing bacterium. In theory, this bacterium may reduce Staphylococcus aureus. The study compared two concentrations of the bacterium vs vehicle delivered as a spray twice per day. Study participants were adults with AD affecting 10%-40% of body surface area.
The study found "meaningful" improvements in itch and objective signs of disease, with clear separation between both doses of the bacterial spray compared with vehicle. At week 4, about 23% of participants treated with the bacterium were clear or almost clear (with a 2-point improvement) compared with 12% in the vehicle group (for comparison, in a phase 2 study comparing topical ruxolitinib with 0.1% triamcinolone cream, there was a 25% clear or almost clear rate [with 2-point improvement] in the triamcinolone-treated individuals).
Though an "all-natural" bacterial approach to managing AD may be appealing to some, it sounded like magic to me. But this well-done study makes it seem like the bacterial approach could be more promising than I had thought. This study also reported about twice as many adverse events (including gastrointestinal issues) with the bacterium-treated participants compared with those who received vehicle, adding to my belief that the bacterial product has efficacy. Whether any other topical will be more effective and safer than is topical triamcinolone remains to be seen. I'm still pessimistic about topicals because of patients' poor adherence to topical treatment, but perhaps an easy-to-use spray that isn't associated with patients' fear of "steroids" will be helpful.
I love articles like this one from Chen and colleagues. They analyzed data on hundreds of thousands of patients with and without AD. Adults with AD had a "significantly increased risk" of developing venous thromboembolism compared with adults without AD. The huge sample size of their study seems compelling. That huge sample size allows detection of effects so small that they may be clinically insignificant.
They report that patients with AD had a venous thromboembolism at a rate of 1.05/1000 patients-years; the rate was 0.82 for patients without AD. From that, we can calculate that there would be an additional 23 patients with venous thromboembolism for every 100,000 patient-years or about one more venous thromboembolism in the AD group in every 4000 patient-years. Though the finding was statistically significant, I don't think it is clinically meaningful.
The authors correctly conclude that "vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients with AD who present with relevant symptoms (eg, unexplained dyspnea, chest tightness, and limb swelling)." But it is probably also true that vascular examination and consultation with the emergency department, cardiologists, or pulmonologists are indicated for patients without AD who present with those symptoms. I think the authors might have been on solid ground if they had concluded that there was a statistically significant but clinically insignificant increased risk for venous thromboembolism in patients with AD.
Eichenfeld and colleagues examined the use of topical crisaborole once per day as a maintenance treatment for patients with mild to moderate AD. The study compared patients given topical crisaborole with those randomly assigned to vehicle. The active treatment was effective because topical crisaborole treated patients had longer times to the first flare following treatment and fewer flares over the 1 year of treatment. The differences were not huge, but I think they were clinically meaningful. I'm guessing that the topical crisaborole maintenance treatment would have been even more effective had it been used regularly. The study did not, as far as I could tell, assess how well the treatment was used.
An interesting aspect of this study is that it began with nearly 500 participants who started on twice daily topical crisaborole. The 270 patients who responded to the treatment (achieving clear or almost clear with at least a 2-point improvement) were enrolled in the 1-year maintenance phase. Thus, the participants in the maintenance phase were preselected for patients who respond to topical crisaborole. We don't know why they were responders (I, of course, expect it is because they selected for patients who are better than others are at using a topical treatment), but it may be best not to try to generalize these results and assume this form of maintenance treatment would work equally well in a population who achieve initial success with an oral therapy regimen (for example, a quick course of oral prednisone).
Dupilumab was a revolutionary treatment for AD. I didn't think that I'd ever see a more effective treatment. It's so safe too! It has been a first-line treatment for AD since its introduction. Now, we also have oral Janus kinase inhibitor options. Blauvelt and colleagues examined what happens when patients who have been on dupilumab are switched to a high dose (30 mg/d) of upadacitinib (the standard starting dose of upadacitinib is 15 mg/d). Though dupilumab is very effective, upadacitinib is more so. After 4 weeks of switching to upadacitinib, nearly half the patients were completely clear of AD compared with only 16.0% after 24 weeks of dupilumab! The authors point out, optimistically, that "No new safety risks were observed." Though there were no cancers, gastrointestinal perforations, major adverse cardiovascular events, or venous thromboembolic events, there were cases of eczema herpeticum and zoster in patients treated with upadacitinib. Having upadacitinib available for patients who fail dupilumab is a clear benefit; the role of upadacitinib before dupilumab seems less clear.
Patients doing great on dupilumab for AD may be wondering: Do I still need to take it every 2 weeks? Spekhorst and colleagues may have the answer. They describe the response to tapering dupilumab in patients who had been on the drug for at least 1 year with well-controlled disease for at least 6 months. Patients in the study then continued dupilumab with the longest possible dosing interval while maintaining control of their AD.
Generally, patients maintained good control of their AD, with only a small increase in mean disease severity and in concomitant use of topical steroids. For the patients who attempted prolongation, 83% successfully continued dupilumab treatment with a prolonged interval. Not at all surprisingly, the authors calculated that prolonging the interval between dosing led to large savings in cost.
One of the nice features of dupilumab treatment is that loss of response over time seems unusual. Perhaps there is a low propensity for forming antidrug antibodies when dupilumab is used in the standard every 2-week dosing regimen. I don't know whether antidrug antibodies would be more likely with the intermittent dosing regimen. But now that we have other good systemic treatment options for AD, losing dupilumab efficacy would not be as critical a problem as it used to be. I also want to point out that patients' adherence to injection treatment, though better than adherence to topicals, is far from perfect. It's likely that many patients have already been prolonging the interval between taking their treatments. If you want to know, just ask them. The way I like to phrase the question is: "Are you keeping the extra injectors you've accumulated refrigerated like you are supposed to?"