User login
HOME-PE trial clarifies which pulmonary embolism patients to treat at home
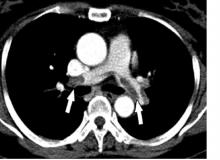
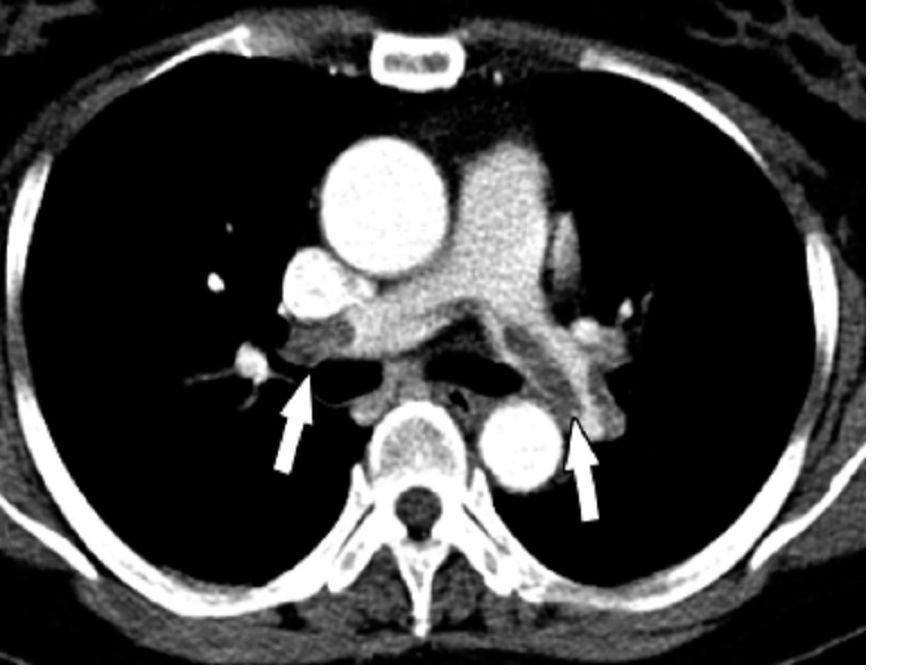
The pragmatic Hestia criteria proved as safe as the more structured, points-based simplified Pulmonary Embolism Severity Index (sPESI) score for selection of patients with acute pulmonary embolism for outpatient care in the large, randomized HOME-PE trial presented at the virtual annual congress of the European Society of Cardiology.
“These results support outpatient management of acute pulmonary embolism patients using either the Hestia method or the sPESI score with the option for the physician-in-charge to override the decision. In hospitals organized for outpatient management, both triaging strategies enable more than a third of pulmonary embolism patients to be managed at home with a low rate of complications,” Pierre-Marie Roy, MD, said in presenting the HOME-PE findings.
The study clarifies a transatlantic controversy regarding how best to triage patients with acute pulmonary embolism (PE) for outpatient care. The answer? It’s basically a tie between the points-based sPESI score recommended in the current ESC guidelines (Eur Respir J. 2019 Oct 9;54[3]:1901647) and the Hestia method endorsed in the American College of Chest Physician guidelines (Chest. 2016 Feb;149[2]:315-52).
The sPESI is a validated tool that grants 1 point each for age over 80 years, background cardiopulmonary disease, a systolic blood pressure below 100 mm Hg, cancer, a heart rate of 110 bpm or more, and an oxygen saturation level below 90%. A patient needs a score of zero to be eligible for outpatient management. In contrast, the Hestia method relies upon 11 simple bedside criteria rather than a points system, explained Dr. Roy of University Hospital of Angers, France (J Thromb Haemost. 2011 Aug;9[8]:1500-7).
HOME-PE was a randomized, open-label, noninferiority trial conducted at 26 hospitals in France, Belgium, Switzerland, and the Netherlands. The study included 1,974 patients presenting to the emergency department with non–high-risk acute PE as defined by hemodynamic stability. About 39% of patients in the Hestia group were eligible for outpatient care on the basis of ‘no’ answers regarding all 11 criteria, while 48% of patients had an sPESI score of 0 and were thus initially considered appropriate for outpatient management.
However, the investigators recognized that no scoring system for acute PE is perfect, and that the judgment of a physician with extensive experience in managing this life-threatening condition counts for a lot. So they stipulated that a patient’s physician-in-charge could overrule a decision for early discharge. This happened 29% of the time in patients with a sPESI score of 0, as compared with a 3% overrule rate with the Hestia rule. The physician-in-charge also moved small numbers of patients who were Hestia or sPESI positive into the outpatient care group. As a result, a similar proportion of patients in both groups were discharged home within 24 hours for outpatient treatment: 38% of the total Hestia group and 37% in the sPESI arm.
Major adverse event rates were reassuringly low in both groups managed on an outpatient basis. The composite of recurrent venous thromboembolism, bleeding, or death within 30 days occurred in 1.3% of Hestia outpatients and 1.1% of sPESI outpatients. Among patients managed in the hospital, these rates were 5.6% in the Hestia group and 4.7% in the sPESI group.
Discussant Stavros V. Konstantinides, MD, who chaired the ESC guideline committee, asked rhetorically, “who’s happy with the HOME-PE trial? I think everybody.”
“The Hestia criteria integrate the feasibility of family support of the individual patient. This is a good thing. And eligibility based on the Hestia criteria, unlike sPESI, does not require age younger than 80 years or no cancer, and it appears from the HOME-PE study that this is okay,” observed Dr. Konstantinides of the Center for Thrombosis and Hemostasis at the University of Mainz (Germany).
In an interview, Hadley Wilson, MD, called the HOME-PE trial “transformative” and predicted it will change clinical practice. He was particularly impressed with the high quality of the trial, noting that 87% of participants managed as outpatients received a direct oral anticoagulant.
The Hestia rule is simpler and more user-friendly. And greater use of this triaging strategy might have advantages in terms of economics and health care utilization by potentially encouraging movement of decision-making regarding outpatient management of acute PE out of the hospital wards and into emergency departments, said Dr. Wilson, executive vice chair of the Sanger Heart and Vascular Institute and a cardiologist at the University of North Carolina at Chapel Hill.
Dr. Roy reported receiving research grants to conduct HOME-PE from the French Ministry of Health, the study sponsor. In addition, he is on scientific advisory boards and/or speakers’ panels for Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Aspen, Daiichi Sankyo, and Sanofi Aventis.
The pragmatic Hestia criteria proved as safe as the more structured, points-based simplified Pulmonary Embolism Severity Index (sPESI) score for selection of patients with acute pulmonary embolism for outpatient care in the large, randomized HOME-PE trial presented at the virtual annual congress of the European Society of Cardiology.
“These results support outpatient management of acute pulmonary embolism patients using either the Hestia method or the sPESI score with the option for the physician-in-charge to override the decision. In hospitals organized for outpatient management, both triaging strategies enable more than a third of pulmonary embolism patients to be managed at home with a low rate of complications,” Pierre-Marie Roy, MD, said in presenting the HOME-PE findings.
The study clarifies a transatlantic controversy regarding how best to triage patients with acute pulmonary embolism (PE) for outpatient care. The answer? It’s basically a tie between the points-based sPESI score recommended in the current ESC guidelines (Eur Respir J. 2019 Oct 9;54[3]:1901647) and the Hestia method endorsed in the American College of Chest Physician guidelines (Chest. 2016 Feb;149[2]:315-52).
The sPESI is a validated tool that grants 1 point each for age over 80 years, background cardiopulmonary disease, a systolic blood pressure below 100 mm Hg, cancer, a heart rate of 110 bpm or more, and an oxygen saturation level below 90%. A patient needs a score of zero to be eligible for outpatient management. In contrast, the Hestia method relies upon 11 simple bedside criteria rather than a points system, explained Dr. Roy of University Hospital of Angers, France (J Thromb Haemost. 2011 Aug;9[8]:1500-7).
HOME-PE was a randomized, open-label, noninferiority trial conducted at 26 hospitals in France, Belgium, Switzerland, and the Netherlands. The study included 1,974 patients presenting to the emergency department with non–high-risk acute PE as defined by hemodynamic stability. About 39% of patients in the Hestia group were eligible for outpatient care on the basis of ‘no’ answers regarding all 11 criteria, while 48% of patients had an sPESI score of 0 and were thus initially considered appropriate for outpatient management.
However, the investigators recognized that no scoring system for acute PE is perfect, and that the judgment of a physician with extensive experience in managing this life-threatening condition counts for a lot. So they stipulated that a patient’s physician-in-charge could overrule a decision for early discharge. This happened 29% of the time in patients with a sPESI score of 0, as compared with a 3% overrule rate with the Hestia rule. The physician-in-charge also moved small numbers of patients who were Hestia or sPESI positive into the outpatient care group. As a result, a similar proportion of patients in both groups were discharged home within 24 hours for outpatient treatment: 38% of the total Hestia group and 37% in the sPESI arm.
Major adverse event rates were reassuringly low in both groups managed on an outpatient basis. The composite of recurrent venous thromboembolism, bleeding, or death within 30 days occurred in 1.3% of Hestia outpatients and 1.1% of sPESI outpatients. Among patients managed in the hospital, these rates were 5.6% in the Hestia group and 4.7% in the sPESI group.
Discussant Stavros V. Konstantinides, MD, who chaired the ESC guideline committee, asked rhetorically, “who’s happy with the HOME-PE trial? I think everybody.”
“The Hestia criteria integrate the feasibility of family support of the individual patient. This is a good thing. And eligibility based on the Hestia criteria, unlike sPESI, does not require age younger than 80 years or no cancer, and it appears from the HOME-PE study that this is okay,” observed Dr. Konstantinides of the Center for Thrombosis and Hemostasis at the University of Mainz (Germany).
In an interview, Hadley Wilson, MD, called the HOME-PE trial “transformative” and predicted it will change clinical practice. He was particularly impressed with the high quality of the trial, noting that 87% of participants managed as outpatients received a direct oral anticoagulant.
The Hestia rule is simpler and more user-friendly. And greater use of this triaging strategy might have advantages in terms of economics and health care utilization by potentially encouraging movement of decision-making regarding outpatient management of acute PE out of the hospital wards and into emergency departments, said Dr. Wilson, executive vice chair of the Sanger Heart and Vascular Institute and a cardiologist at the University of North Carolina at Chapel Hill.
Dr. Roy reported receiving research grants to conduct HOME-PE from the French Ministry of Health, the study sponsor. In addition, he is on scientific advisory boards and/or speakers’ panels for Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Aspen, Daiichi Sankyo, and Sanofi Aventis.
The pragmatic Hestia criteria proved as safe as the more structured, points-based simplified Pulmonary Embolism Severity Index (sPESI) score for selection of patients with acute pulmonary embolism for outpatient care in the large, randomized HOME-PE trial presented at the virtual annual congress of the European Society of Cardiology.
“These results support outpatient management of acute pulmonary embolism patients using either the Hestia method or the sPESI score with the option for the physician-in-charge to override the decision. In hospitals organized for outpatient management, both triaging strategies enable more than a third of pulmonary embolism patients to be managed at home with a low rate of complications,” Pierre-Marie Roy, MD, said in presenting the HOME-PE findings.
The study clarifies a transatlantic controversy regarding how best to triage patients with acute pulmonary embolism (PE) for outpatient care. The answer? It’s basically a tie between the points-based sPESI score recommended in the current ESC guidelines (Eur Respir J. 2019 Oct 9;54[3]:1901647) and the Hestia method endorsed in the American College of Chest Physician guidelines (Chest. 2016 Feb;149[2]:315-52).
The sPESI is a validated tool that grants 1 point each for age over 80 years, background cardiopulmonary disease, a systolic blood pressure below 100 mm Hg, cancer, a heart rate of 110 bpm or more, and an oxygen saturation level below 90%. A patient needs a score of zero to be eligible for outpatient management. In contrast, the Hestia method relies upon 11 simple bedside criteria rather than a points system, explained Dr. Roy of University Hospital of Angers, France (J Thromb Haemost. 2011 Aug;9[8]:1500-7).
HOME-PE was a randomized, open-label, noninferiority trial conducted at 26 hospitals in France, Belgium, Switzerland, and the Netherlands. The study included 1,974 patients presenting to the emergency department with non–high-risk acute PE as defined by hemodynamic stability. About 39% of patients in the Hestia group were eligible for outpatient care on the basis of ‘no’ answers regarding all 11 criteria, while 48% of patients had an sPESI score of 0 and were thus initially considered appropriate for outpatient management.
However, the investigators recognized that no scoring system for acute PE is perfect, and that the judgment of a physician with extensive experience in managing this life-threatening condition counts for a lot. So they stipulated that a patient’s physician-in-charge could overrule a decision for early discharge. This happened 29% of the time in patients with a sPESI score of 0, as compared with a 3% overrule rate with the Hestia rule. The physician-in-charge also moved small numbers of patients who were Hestia or sPESI positive into the outpatient care group. As a result, a similar proportion of patients in both groups were discharged home within 24 hours for outpatient treatment: 38% of the total Hestia group and 37% in the sPESI arm.
Major adverse event rates were reassuringly low in both groups managed on an outpatient basis. The composite of recurrent venous thromboembolism, bleeding, or death within 30 days occurred in 1.3% of Hestia outpatients and 1.1% of sPESI outpatients. Among patients managed in the hospital, these rates were 5.6% in the Hestia group and 4.7% in the sPESI group.
Discussant Stavros V. Konstantinides, MD, who chaired the ESC guideline committee, asked rhetorically, “who’s happy with the HOME-PE trial? I think everybody.”
“The Hestia criteria integrate the feasibility of family support of the individual patient. This is a good thing. And eligibility based on the Hestia criteria, unlike sPESI, does not require age younger than 80 years or no cancer, and it appears from the HOME-PE study that this is okay,” observed Dr. Konstantinides of the Center for Thrombosis and Hemostasis at the University of Mainz (Germany).
In an interview, Hadley Wilson, MD, called the HOME-PE trial “transformative” and predicted it will change clinical practice. He was particularly impressed with the high quality of the trial, noting that 87% of participants managed as outpatients received a direct oral anticoagulant.
The Hestia rule is simpler and more user-friendly. And greater use of this triaging strategy might have advantages in terms of economics and health care utilization by potentially encouraging movement of decision-making regarding outpatient management of acute PE out of the hospital wards and into emergency departments, said Dr. Wilson, executive vice chair of the Sanger Heart and Vascular Institute and a cardiologist at the University of North Carolina at Chapel Hill.
Dr. Roy reported receiving research grants to conduct HOME-PE from the French Ministry of Health, the study sponsor. In addition, he is on scientific advisory boards and/or speakers’ panels for Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Aspen, Daiichi Sankyo, and Sanofi Aventis.
REPORTING FROM ESC CONGRESS 2020
ESC’s revised NSTE-ACS guidelines embrace hsT, personalized anti-ischemia treatments
The first revision since 2015 to the European Society of Cardiology’s guidelines for diagnosing and managing non ST-elevation acute coronary syndrome placed increased reliance on high-sensitivity cardiac troponin testing for diagnosis, and embraced coronary CT to rule out lower-risk patients.
It also highlighted the need for personalized antiplatelet regimens, systems of care, and quality improvement.
The society released the new guidelines on August 29 (Eur Heart J. 2020 Aug 29;doi: 10.1093/eurheartj/ehaa575), and then devoted a session to them the next day at the virtual annual congress of the European Society of Cardiology to highlight some of the key updates, starting with the further emphasis placed on high-sensitivity cardiac troponin (hs-cTn), a reliance that contrasts with what remains inconsistent use of this metric in U.S. practice.
An hs-cTn test is the “preferred” diagnostic test and a “key” testing element, said Marco Roffi, MD, professor and director of interventional cardiology at University Hospital, Geneva, and a member of the guideline committee. He also stressed an update to the time frame of initial hs-cTn testing, which now involves a baseline reading and then a second measure after 2 hours to discern how the marker level is evolving with time. The guidelines advise against measuring any other biomarker of myocardial injury.
U.S. lags in measuring high-sensitivity cardiac troponin
U.S. medical systems and centers “are not uniform in adopting hs-cTn,” noted Richard J. Kovacs, MD, professor of cardiology at the Indiana University School of Medicine in Indianapolis. “The new European guidelines should spur U.S. institutions to at least take a close look at the advantages of hs-cTn. There is a strong case that it leads to more efficient emergency care and allows for quicker decisions and triage,” added Dr. Kovacs in an interview.
The new guideline’s emphasis on hs-cTn should hasten broader uptake in U.S. practice, agreed Deepak L. Bhatt, MD, professor of medicine at Harvard Medical School in Boston and a member of the guideline-writing panel
Another plus of the guidelines is its endorsement of an “organized approach to risk assessment” early on in these patients, said Dr. Kovacs, who is also the immediate past-president of the American College of Cardiology (ACC). An ACC committee is developing a new set of recommendations for managing patients with cardiac chest pain and is on track for release in 2021. It would represent the first update to U.S. guidelines for non ST-elevation ACS patients since 2014.
The new ESC guidelines give coronary CT angiography a class Ia rating as an alternative to invasive coronary angiography for assessing patients with a low or intermediate risk of having coronary disease, a “tremendous upgrade,” commented Ashish Pershad, MD, an interventional cardiologist at Banner-University Medical Heart Institute in Phoenix. While he welcomes this support for using coronary CT angiography in this setting, he acknowledged that the method remains limited in availability as it requires highly trained technicians to obtain good images and experienced clinicians to interpret the results.
Personalizing antiplatelet and antithrombotic treatments
Notable revisions to medical treatments to minimize ischemia included an admonition not to use routine pretreatment with a P2Y12 receptor inhibitor (such as clopidogrel) before testing determines coronary anatomy.
Not using one of these antiplatelet drugs upfront on all patients “is a tremendous change,” Dr. Pershad said in an interview. Many patients currently get these drugs while awaiting an angiogram, but a more selected and deferred antiplatelet approach would be better when angiography shows that some patients need coronary bypass surgery, he noted. Recent study results have shown no added benefit from pretreatment, and its use can be especially problematic for patients who are slated for a planned invasive strategy, said Dr. Bhatt.
Dr. Pershad, Dr. Bhatt, and Dr. Kovacs all praised the overall emphasis on a personalized approach to treating patients with antiplatelet and antithrombotic drugs, with endorsement of a flexible approach to treatment intensity and duration. The guideline acknowledges the need to take into account a patient’s bleeding risk and comorbidities, and specifically endorsed the Academic Research Consortium’s formula for identifying and stratifying high bleeding risk (Eur Heart J. 2019 Aug 14;40[31]:2632-53).
The new guidelines also provide guidance on how to apply recent study results that addressed balancing efficacy and safety when pairing an antiplatelet drug with a direct-acting oral anticoagulant (DOAC) for patients who potentially need both drug classes, such as patients with atrial fibrillation and a recent ACS event. “It’s tremendous to get clarity on this issue; there’s been a lot of uncertainty,” said Dr. Pershad. The guidelines call for a week of triple therapy with a DOAC, aspirin, and a second antiplatelet drug, followed by 12 months on a DOAC plus a single antiplatelet drug, and then the DOAC alone as the “default” strategy for most patients, but also presents alternative options for patients with high risk for either bleeding or ischemia.
The new guidelines also give much-needed direction on how to apply an invasive strategy, with an emphasis on immediate intervention for within 2 hours for very-high-risk patients, and early intervention within 24 hours for high-risk patients. Adhering to this timetable can mean increasing catheter laboratory availability on an urgent basis over weekends, Dr. Bhatt noted.
Improving quality of care
A novel section in the new guidelines was devoted to nine quality measures that can help health systems and medical centers monitor their adherence to the guideline recommendations, track their performance relative to peer institutions, and follow changes in performance that result from quality improvement steps. It’s something of a “futuristic” step for a guideline to take, with a goal of persuading administrators to implement tracking of these measures and improve patient outcomes, noted Dr. Bhatt.
“It’s very important to see that this is not just a set of guidelines but also a tool to improve quality of care,” commented Dr. Kovacs. The key to success in this effort will be to follow registered patients, set benchmarks that systems can aspire to achieve, and use this to improve the quality of care.
Until now, optimizing care for patients with NSTE-ACS has been “challenging,” he said. “The focus must be on moving toward systems of care” that can provide consistent patient evaluation and care, and do it quickly, said Dr. Kovacs.
Dr. Roffi has received research funding from Biotronik, Boston Scientific, GE Healthcare, and Medtronic. Dr. Kovacs was formerly an employee of Eli Lilly. Dr. Bhatt has been a consultant to and has received research funding from several companies. Dr. Pershad had no disclosures.
The first revision since 2015 to the European Society of Cardiology’s guidelines for diagnosing and managing non ST-elevation acute coronary syndrome placed increased reliance on high-sensitivity cardiac troponin testing for diagnosis, and embraced coronary CT to rule out lower-risk patients.
It also highlighted the need for personalized antiplatelet regimens, systems of care, and quality improvement.
The society released the new guidelines on August 29 (Eur Heart J. 2020 Aug 29;doi: 10.1093/eurheartj/ehaa575), and then devoted a session to them the next day at the virtual annual congress of the European Society of Cardiology to highlight some of the key updates, starting with the further emphasis placed on high-sensitivity cardiac troponin (hs-cTn), a reliance that contrasts with what remains inconsistent use of this metric in U.S. practice.
An hs-cTn test is the “preferred” diagnostic test and a “key” testing element, said Marco Roffi, MD, professor and director of interventional cardiology at University Hospital, Geneva, and a member of the guideline committee. He also stressed an update to the time frame of initial hs-cTn testing, which now involves a baseline reading and then a second measure after 2 hours to discern how the marker level is evolving with time. The guidelines advise against measuring any other biomarker of myocardial injury.
U.S. lags in measuring high-sensitivity cardiac troponin
U.S. medical systems and centers “are not uniform in adopting hs-cTn,” noted Richard J. Kovacs, MD, professor of cardiology at the Indiana University School of Medicine in Indianapolis. “The new European guidelines should spur U.S. institutions to at least take a close look at the advantages of hs-cTn. There is a strong case that it leads to more efficient emergency care and allows for quicker decisions and triage,” added Dr. Kovacs in an interview.
The new guideline’s emphasis on hs-cTn should hasten broader uptake in U.S. practice, agreed Deepak L. Bhatt, MD, professor of medicine at Harvard Medical School in Boston and a member of the guideline-writing panel
Another plus of the guidelines is its endorsement of an “organized approach to risk assessment” early on in these patients, said Dr. Kovacs, who is also the immediate past-president of the American College of Cardiology (ACC). An ACC committee is developing a new set of recommendations for managing patients with cardiac chest pain and is on track for release in 2021. It would represent the first update to U.S. guidelines for non ST-elevation ACS patients since 2014.
The new ESC guidelines give coronary CT angiography a class Ia rating as an alternative to invasive coronary angiography for assessing patients with a low or intermediate risk of having coronary disease, a “tremendous upgrade,” commented Ashish Pershad, MD, an interventional cardiologist at Banner-University Medical Heart Institute in Phoenix. While he welcomes this support for using coronary CT angiography in this setting, he acknowledged that the method remains limited in availability as it requires highly trained technicians to obtain good images and experienced clinicians to interpret the results.
Personalizing antiplatelet and antithrombotic treatments
Notable revisions to medical treatments to minimize ischemia included an admonition not to use routine pretreatment with a P2Y12 receptor inhibitor (such as clopidogrel) before testing determines coronary anatomy.
Not using one of these antiplatelet drugs upfront on all patients “is a tremendous change,” Dr. Pershad said in an interview. Many patients currently get these drugs while awaiting an angiogram, but a more selected and deferred antiplatelet approach would be better when angiography shows that some patients need coronary bypass surgery, he noted. Recent study results have shown no added benefit from pretreatment, and its use can be especially problematic for patients who are slated for a planned invasive strategy, said Dr. Bhatt.
Dr. Pershad, Dr. Bhatt, and Dr. Kovacs all praised the overall emphasis on a personalized approach to treating patients with antiplatelet and antithrombotic drugs, with endorsement of a flexible approach to treatment intensity and duration. The guideline acknowledges the need to take into account a patient’s bleeding risk and comorbidities, and specifically endorsed the Academic Research Consortium’s formula for identifying and stratifying high bleeding risk (Eur Heart J. 2019 Aug 14;40[31]:2632-53).
The new guidelines also provide guidance on how to apply recent study results that addressed balancing efficacy and safety when pairing an antiplatelet drug with a direct-acting oral anticoagulant (DOAC) for patients who potentially need both drug classes, such as patients with atrial fibrillation and a recent ACS event. “It’s tremendous to get clarity on this issue; there’s been a lot of uncertainty,” said Dr. Pershad. The guidelines call for a week of triple therapy with a DOAC, aspirin, and a second antiplatelet drug, followed by 12 months on a DOAC plus a single antiplatelet drug, and then the DOAC alone as the “default” strategy for most patients, but also presents alternative options for patients with high risk for either bleeding or ischemia.
The new guidelines also give much-needed direction on how to apply an invasive strategy, with an emphasis on immediate intervention for within 2 hours for very-high-risk patients, and early intervention within 24 hours for high-risk patients. Adhering to this timetable can mean increasing catheter laboratory availability on an urgent basis over weekends, Dr. Bhatt noted.
Improving quality of care
A novel section in the new guidelines was devoted to nine quality measures that can help health systems and medical centers monitor their adherence to the guideline recommendations, track their performance relative to peer institutions, and follow changes in performance that result from quality improvement steps. It’s something of a “futuristic” step for a guideline to take, with a goal of persuading administrators to implement tracking of these measures and improve patient outcomes, noted Dr. Bhatt.
“It’s very important to see that this is not just a set of guidelines but also a tool to improve quality of care,” commented Dr. Kovacs. The key to success in this effort will be to follow registered patients, set benchmarks that systems can aspire to achieve, and use this to improve the quality of care.
Until now, optimizing care for patients with NSTE-ACS has been “challenging,” he said. “The focus must be on moving toward systems of care” that can provide consistent patient evaluation and care, and do it quickly, said Dr. Kovacs.
Dr. Roffi has received research funding from Biotronik, Boston Scientific, GE Healthcare, and Medtronic. Dr. Kovacs was formerly an employee of Eli Lilly. Dr. Bhatt has been a consultant to and has received research funding from several companies. Dr. Pershad had no disclosures.
The first revision since 2015 to the European Society of Cardiology’s guidelines for diagnosing and managing non ST-elevation acute coronary syndrome placed increased reliance on high-sensitivity cardiac troponin testing for diagnosis, and embraced coronary CT to rule out lower-risk patients.
It also highlighted the need for personalized antiplatelet regimens, systems of care, and quality improvement.
The society released the new guidelines on August 29 (Eur Heart J. 2020 Aug 29;doi: 10.1093/eurheartj/ehaa575), and then devoted a session to them the next day at the virtual annual congress of the European Society of Cardiology to highlight some of the key updates, starting with the further emphasis placed on high-sensitivity cardiac troponin (hs-cTn), a reliance that contrasts with what remains inconsistent use of this metric in U.S. practice.
An hs-cTn test is the “preferred” diagnostic test and a “key” testing element, said Marco Roffi, MD, professor and director of interventional cardiology at University Hospital, Geneva, and a member of the guideline committee. He also stressed an update to the time frame of initial hs-cTn testing, which now involves a baseline reading and then a second measure after 2 hours to discern how the marker level is evolving with time. The guidelines advise against measuring any other biomarker of myocardial injury.
U.S. lags in measuring high-sensitivity cardiac troponin
U.S. medical systems and centers “are not uniform in adopting hs-cTn,” noted Richard J. Kovacs, MD, professor of cardiology at the Indiana University School of Medicine in Indianapolis. “The new European guidelines should spur U.S. institutions to at least take a close look at the advantages of hs-cTn. There is a strong case that it leads to more efficient emergency care and allows for quicker decisions and triage,” added Dr. Kovacs in an interview.
The new guideline’s emphasis on hs-cTn should hasten broader uptake in U.S. practice, agreed Deepak L. Bhatt, MD, professor of medicine at Harvard Medical School in Boston and a member of the guideline-writing panel
Another plus of the guidelines is its endorsement of an “organized approach to risk assessment” early on in these patients, said Dr. Kovacs, who is also the immediate past-president of the American College of Cardiology (ACC). An ACC committee is developing a new set of recommendations for managing patients with cardiac chest pain and is on track for release in 2021. It would represent the first update to U.S. guidelines for non ST-elevation ACS patients since 2014.
The new ESC guidelines give coronary CT angiography a class Ia rating as an alternative to invasive coronary angiography for assessing patients with a low or intermediate risk of having coronary disease, a “tremendous upgrade,” commented Ashish Pershad, MD, an interventional cardiologist at Banner-University Medical Heart Institute in Phoenix. While he welcomes this support for using coronary CT angiography in this setting, he acknowledged that the method remains limited in availability as it requires highly trained technicians to obtain good images and experienced clinicians to interpret the results.
Personalizing antiplatelet and antithrombotic treatments
Notable revisions to medical treatments to minimize ischemia included an admonition not to use routine pretreatment with a P2Y12 receptor inhibitor (such as clopidogrel) before testing determines coronary anatomy.
Not using one of these antiplatelet drugs upfront on all patients “is a tremendous change,” Dr. Pershad said in an interview. Many patients currently get these drugs while awaiting an angiogram, but a more selected and deferred antiplatelet approach would be better when angiography shows that some patients need coronary bypass surgery, he noted. Recent study results have shown no added benefit from pretreatment, and its use can be especially problematic for patients who are slated for a planned invasive strategy, said Dr. Bhatt.
Dr. Pershad, Dr. Bhatt, and Dr. Kovacs all praised the overall emphasis on a personalized approach to treating patients with antiplatelet and antithrombotic drugs, with endorsement of a flexible approach to treatment intensity and duration. The guideline acknowledges the need to take into account a patient’s bleeding risk and comorbidities, and specifically endorsed the Academic Research Consortium’s formula for identifying and stratifying high bleeding risk (Eur Heart J. 2019 Aug 14;40[31]:2632-53).
The new guidelines also provide guidance on how to apply recent study results that addressed balancing efficacy and safety when pairing an antiplatelet drug with a direct-acting oral anticoagulant (DOAC) for patients who potentially need both drug classes, such as patients with atrial fibrillation and a recent ACS event. “It’s tremendous to get clarity on this issue; there’s been a lot of uncertainty,” said Dr. Pershad. The guidelines call for a week of triple therapy with a DOAC, aspirin, and a second antiplatelet drug, followed by 12 months on a DOAC plus a single antiplatelet drug, and then the DOAC alone as the “default” strategy for most patients, but also presents alternative options for patients with high risk for either bleeding or ischemia.
The new guidelines also give much-needed direction on how to apply an invasive strategy, with an emphasis on immediate intervention for within 2 hours for very-high-risk patients, and early intervention within 24 hours for high-risk patients. Adhering to this timetable can mean increasing catheter laboratory availability on an urgent basis over weekends, Dr. Bhatt noted.
Improving quality of care
A novel section in the new guidelines was devoted to nine quality measures that can help health systems and medical centers monitor their adherence to the guideline recommendations, track their performance relative to peer institutions, and follow changes in performance that result from quality improvement steps. It’s something of a “futuristic” step for a guideline to take, with a goal of persuading administrators to implement tracking of these measures and improve patient outcomes, noted Dr. Bhatt.
“It’s very important to see that this is not just a set of guidelines but also a tool to improve quality of care,” commented Dr. Kovacs. The key to success in this effort will be to follow registered patients, set benchmarks that systems can aspire to achieve, and use this to improve the quality of care.
Until now, optimizing care for patients with NSTE-ACS has been “challenging,” he said. “The focus must be on moving toward systems of care” that can provide consistent patient evaluation and care, and do it quickly, said Dr. Kovacs.
Dr. Roffi has received research funding from Biotronik, Boston Scientific, GE Healthcare, and Medtronic. Dr. Kovacs was formerly an employee of Eli Lilly. Dr. Bhatt has been a consultant to and has received research funding from several companies. Dr. Pershad had no disclosures.
FROM ESC CONGRESS 2020
Evolocumab safe and effective in pediatric FH
The PCSK9 monoclonal antibody evolocumab (Repatha) was well tolerated and effectively lowered LDL cholesterol by 38% compared with placebo in a randomized controlled trial in pediatric patients with heterozygous familial hypercholesterolemia (FH) already taking statins with or without ezetimibe.
“HAUSER-RCT is the largest study and the first placebo-controlled randomized trial of a PCSK9 inhibitor in pediatric FH,” senior author Daniel Gaudet, MD, PhD, Universite de Montreal, said in an interview.
“The study showed good safety and efficacy of the drug in this population, with an excellent 44% reduction in LDL cholesterol compared with 6% in the placebo group.”
The trial also found evolocumab to be well tolerated in this group, with adverse effects similar in the active and placebo groups.
“Some people have wondered about using a drug with a monthly injection in a pediatric population, but this was not an issue in our study,” Dr. Gaudet said. “The idea of a monthly injection was well received, and no patient withdrew because of this.”
The HAUSER-RCT trial was presented on Aug. 29 at the virtual annual congress of the European Society of Cardiology (ESC Congress 2020) and simultaneously published online in the New England Journal of Medicine.
“With patients recruited from 23 countries in five continents, the study provides an accurate picture of the safety and efficacy of evolocumab in pediatric FH patients worldwide,” Dr. Gaudet said.
The 24-week, randomized, double-blind, placebo-controlled trial involved 157 patients aged 10-17 years with heterozygous FH already taking statins with or without ezetimibe and who had an LDL cholesterol level of 130 mg/dL or more and a triglyceride level of 400 mg/dL or less.
They were randomly assigned in a 2:1 ratio to receive monthly subcutaneous injections of evolocumab (420 mg) or placebo.
Results showed that at week 24, the mean percentage change from baseline in LDL cholesterol level was −44.5% in the evolocumab group and −6.2% in the placebo group, giving a difference of −38.3 percentage points (P < .001).
The absolute change in the LDL cholesterol level was −77.5 mg/dL in the evolocumab group and −9.0 mg/dL in the placebo group, giving a difference of −68.6 mg/dL (P < .001).
Results for all secondary lipid variables were significantly better with evolocumab than with placebo. The incidence of adverse events that occurred during the treatment period was similar in the evolocumab and placebo groups. Laboratory abnormalities did not differ between groups.
Dr. Gaudet noted that FH is the most common genetic disease worldwide, affecting 1 in 250 people. “It is very treatable, so it is important to identify these patients, but it is massively underdiagnosed, with only around 15%-20% of patients with the condition having been identified,” he said.
“The vast majority of pediatric FH patients can reach target LDL levels with statins and ezetimibe, but there are 5%-10% of patients who may need additional therapy. We have now shown that evolocumab is safe and effective for these patients and can be used to fill this gap,” Dr. Gaudet said. “We can now say that we can cover all situations in treating FH whatever the severity of the disease.”
However, the challenge remains to improve the diagnosis of FH. “If there is one person with FH in a family, then it is essential that the whole extended family is tested. Our toolbox for treating this condition is now sufficiently effective, so there is no reason not to diagnose this disease,” Dr. Gaudet stressed.
The HAUSER-RCT study was supported by Amgen. Gaudet reports grants and personal fees from Amgen during the conduct of the study.
A version of this article originally appeared on Medscape.com.
The PCSK9 monoclonal antibody evolocumab (Repatha) was well tolerated and effectively lowered LDL cholesterol by 38% compared with placebo in a randomized controlled trial in pediatric patients with heterozygous familial hypercholesterolemia (FH) already taking statins with or without ezetimibe.
“HAUSER-RCT is the largest study and the first placebo-controlled randomized trial of a PCSK9 inhibitor in pediatric FH,” senior author Daniel Gaudet, MD, PhD, Universite de Montreal, said in an interview.
“The study showed good safety and efficacy of the drug in this population, with an excellent 44% reduction in LDL cholesterol compared with 6% in the placebo group.”
The trial also found evolocumab to be well tolerated in this group, with adverse effects similar in the active and placebo groups.
“Some people have wondered about using a drug with a monthly injection in a pediatric population, but this was not an issue in our study,” Dr. Gaudet said. “The idea of a monthly injection was well received, and no patient withdrew because of this.”
The HAUSER-RCT trial was presented on Aug. 29 at the virtual annual congress of the European Society of Cardiology (ESC Congress 2020) and simultaneously published online in the New England Journal of Medicine.
“With patients recruited from 23 countries in five continents, the study provides an accurate picture of the safety and efficacy of evolocumab in pediatric FH patients worldwide,” Dr. Gaudet said.
The 24-week, randomized, double-blind, placebo-controlled trial involved 157 patients aged 10-17 years with heterozygous FH already taking statins with or without ezetimibe and who had an LDL cholesterol level of 130 mg/dL or more and a triglyceride level of 400 mg/dL or less.
They were randomly assigned in a 2:1 ratio to receive monthly subcutaneous injections of evolocumab (420 mg) or placebo.
Results showed that at week 24, the mean percentage change from baseline in LDL cholesterol level was −44.5% in the evolocumab group and −6.2% in the placebo group, giving a difference of −38.3 percentage points (P < .001).
The absolute change in the LDL cholesterol level was −77.5 mg/dL in the evolocumab group and −9.0 mg/dL in the placebo group, giving a difference of −68.6 mg/dL (P < .001).
Results for all secondary lipid variables were significantly better with evolocumab than with placebo. The incidence of adverse events that occurred during the treatment period was similar in the evolocumab and placebo groups. Laboratory abnormalities did not differ between groups.
Dr. Gaudet noted that FH is the most common genetic disease worldwide, affecting 1 in 250 people. “It is very treatable, so it is important to identify these patients, but it is massively underdiagnosed, with only around 15%-20% of patients with the condition having been identified,” he said.
“The vast majority of pediatric FH patients can reach target LDL levels with statins and ezetimibe, but there are 5%-10% of patients who may need additional therapy. We have now shown that evolocumab is safe and effective for these patients and can be used to fill this gap,” Dr. Gaudet said. “We can now say that we can cover all situations in treating FH whatever the severity of the disease.”
However, the challenge remains to improve the diagnosis of FH. “If there is one person with FH in a family, then it is essential that the whole extended family is tested. Our toolbox for treating this condition is now sufficiently effective, so there is no reason not to diagnose this disease,” Dr. Gaudet stressed.
The HAUSER-RCT study was supported by Amgen. Gaudet reports grants and personal fees from Amgen during the conduct of the study.
A version of this article originally appeared on Medscape.com.
The PCSK9 monoclonal antibody evolocumab (Repatha) was well tolerated and effectively lowered LDL cholesterol by 38% compared with placebo in a randomized controlled trial in pediatric patients with heterozygous familial hypercholesterolemia (FH) already taking statins with or without ezetimibe.
“HAUSER-RCT is the largest study and the first placebo-controlled randomized trial of a PCSK9 inhibitor in pediatric FH,” senior author Daniel Gaudet, MD, PhD, Universite de Montreal, said in an interview.
“The study showed good safety and efficacy of the drug in this population, with an excellent 44% reduction in LDL cholesterol compared with 6% in the placebo group.”
The trial also found evolocumab to be well tolerated in this group, with adverse effects similar in the active and placebo groups.
“Some people have wondered about using a drug with a monthly injection in a pediatric population, but this was not an issue in our study,” Dr. Gaudet said. “The idea of a monthly injection was well received, and no patient withdrew because of this.”
The HAUSER-RCT trial was presented on Aug. 29 at the virtual annual congress of the European Society of Cardiology (ESC Congress 2020) and simultaneously published online in the New England Journal of Medicine.
“With patients recruited from 23 countries in five continents, the study provides an accurate picture of the safety and efficacy of evolocumab in pediatric FH patients worldwide,” Dr. Gaudet said.
The 24-week, randomized, double-blind, placebo-controlled trial involved 157 patients aged 10-17 years with heterozygous FH already taking statins with or without ezetimibe and who had an LDL cholesterol level of 130 mg/dL or more and a triglyceride level of 400 mg/dL or less.
They were randomly assigned in a 2:1 ratio to receive monthly subcutaneous injections of evolocumab (420 mg) or placebo.
Results showed that at week 24, the mean percentage change from baseline in LDL cholesterol level was −44.5% in the evolocumab group and −6.2% in the placebo group, giving a difference of −38.3 percentage points (P < .001).
The absolute change in the LDL cholesterol level was −77.5 mg/dL in the evolocumab group and −9.0 mg/dL in the placebo group, giving a difference of −68.6 mg/dL (P < .001).
Results for all secondary lipid variables were significantly better with evolocumab than with placebo. The incidence of adverse events that occurred during the treatment period was similar in the evolocumab and placebo groups. Laboratory abnormalities did not differ between groups.
Dr. Gaudet noted that FH is the most common genetic disease worldwide, affecting 1 in 250 people. “It is very treatable, so it is important to identify these patients, but it is massively underdiagnosed, with only around 15%-20% of patients with the condition having been identified,” he said.
“The vast majority of pediatric FH patients can reach target LDL levels with statins and ezetimibe, but there are 5%-10% of patients who may need additional therapy. We have now shown that evolocumab is safe and effective for these patients and can be used to fill this gap,” Dr. Gaudet said. “We can now say that we can cover all situations in treating FH whatever the severity of the disease.”
However, the challenge remains to improve the diagnosis of FH. “If there is one person with FH in a family, then it is essential that the whole extended family is tested. Our toolbox for treating this condition is now sufficiently effective, so there is no reason not to diagnose this disease,” Dr. Gaudet stressed.
The HAUSER-RCT study was supported by Amgen. Gaudet reports grants and personal fees from Amgen during the conduct of the study.
A version of this article originally appeared on Medscape.com.
Gut bacteria linked to cardiovascular, other health conditions
Microorganisms in the human digestive tract are linked to 29 specific health conditions, including chronic obstructive pulmonary disease, high blood pressure, and type 2 diabetes, according to a genome analysis in more than 400,000 individuals.
Although previous studies have suggested a link between gut microbiota and diseases in humans, “the extent to which the human gut microbiome can be considered a determinant of disease and healthy aging remains unknown,” Hilde E. Groot, MD, of the University of Groningen (The Netherlands), said in a presentation at the virtual annual congress of the European Society of Cardiology.
To identify the spectrum of diseases linked to the gut microbiome, the researchers identified 422,417 unrelated adults of White British ancestry with genotype and matching genetic data. The average age of the participants was 57 years and 46% were male.
The researchers conducted a phenomewide association study including 35 distinct single-nucleotide polymorphisms (SNPs) that are known to influence the microbiome of the human gut.
Overall, seven SNPs were significantly associated with 29 disease outcomes including hypertension, type 2 diabetes, hypercholesterolemia, heart failure, renal failure, and osteoarthritis.
In addition, after a further sensitivity analysis using a Mendelian randomization (MR) approach, associations between Ruminococcus flavefaciens and hypertension and between Clostridium and platelet count might point to a causal link, the researchers said.
“Over the past few years, the amount of research concerning the human gut microbiome and the associations with health and disease has tremendously increased. However, most studies investigated one or a few traits. The strength of our study is the possibility to cover a wide range of traits simultaneously within one population,” Dr. Groot said in an interview.
“Our data support the hypothesis that the human gut microbiome is a complex system, involved in many pathophysiological mechanisms in the human body. So, our results are additional to earlier research and strengthen this hypothesis,” Dr. Groot added.
“Microbiota and their metabolites might be of importance in the interplay between overlapping pathophysiological processes, and could serve as potential therapeutic targets for the maintenance of health and prevention and treatment of cardiovascular diseases. However, before it is possible to give advice for the public and medical practice, further research is needed to study causality,” she emphasized.
“Currently, it is too soon to advise patients concerning their microbiome,” Dr. Groot noted. “However, genetic studies like ours might help other researchers to study causality between the gut microbiome and particular traits, which might potentially lead to new therapeutic targets. Next to genetic variants as a proxy, we’re currently studying the gut microbiome composition in myocardial infarction patients and healthy controls in a longitudinal setting.”
“Previous studies have suggested a potential link between the gut microbiome and the development of cardiovascular disease, type 2 diabetes mellitus, and other chronic disorders,” Carol Ann Remme, MD, of the Amsterdam University Medical Center, said in an interview. “However, it is challenging to study the effect of gut microbiome composition in large patient cohorts. As an alternative approach, the study authors showed in a very large population that genetic variants previously shown to influence gut microbiome composition were significantly associated with conditions such as hypertension, type 2 diabetes, hypercholesterolemia, and heart failure.”
The study is unique in that it employed a very large cohort of more than 400,000 individuals, which is typically required to be able to draw clear conclusions, Dr. Remme continued. “The authors were able to further refine their findings by linking genetic variants known to influence specific gut bacteria to some particular disorders,” she noted.
“It is becoming increasingly clear that an individual’s gut microbiome composition, which is defined by both genetic and environmental factors such as diet, may affect his/her susceptibility to certain diseases – including cardiovascular – in addition to disease progression and outcome,” said Dr. Remme. “This may ultimately lead to development of novel, personalized strategies for risk stratification in addition to potential preventive measures targeting the gut microbiome. I expect this area of research will become increasingly important in the coming years.”
The study received no outside funding. Dr. Groot and colleagues had no financial conflicts to disclose. Dr. Remme had no financial conflicts to disclose.
Microorganisms in the human digestive tract are linked to 29 specific health conditions, including chronic obstructive pulmonary disease, high blood pressure, and type 2 diabetes, according to a genome analysis in more than 400,000 individuals.
Although previous studies have suggested a link between gut microbiota and diseases in humans, “the extent to which the human gut microbiome can be considered a determinant of disease and healthy aging remains unknown,” Hilde E. Groot, MD, of the University of Groningen (The Netherlands), said in a presentation at the virtual annual congress of the European Society of Cardiology.
To identify the spectrum of diseases linked to the gut microbiome, the researchers identified 422,417 unrelated adults of White British ancestry with genotype and matching genetic data. The average age of the participants was 57 years and 46% were male.
The researchers conducted a phenomewide association study including 35 distinct single-nucleotide polymorphisms (SNPs) that are known to influence the microbiome of the human gut.
Overall, seven SNPs were significantly associated with 29 disease outcomes including hypertension, type 2 diabetes, hypercholesterolemia, heart failure, renal failure, and osteoarthritis.
In addition, after a further sensitivity analysis using a Mendelian randomization (MR) approach, associations between Ruminococcus flavefaciens and hypertension and between Clostridium and platelet count might point to a causal link, the researchers said.
“Over the past few years, the amount of research concerning the human gut microbiome and the associations with health and disease has tremendously increased. However, most studies investigated one or a few traits. The strength of our study is the possibility to cover a wide range of traits simultaneously within one population,” Dr. Groot said in an interview.
“Our data support the hypothesis that the human gut microbiome is a complex system, involved in many pathophysiological mechanisms in the human body. So, our results are additional to earlier research and strengthen this hypothesis,” Dr. Groot added.
“Microbiota and their metabolites might be of importance in the interplay between overlapping pathophysiological processes, and could serve as potential therapeutic targets for the maintenance of health and prevention and treatment of cardiovascular diseases. However, before it is possible to give advice for the public and medical practice, further research is needed to study causality,” she emphasized.
“Currently, it is too soon to advise patients concerning their microbiome,” Dr. Groot noted. “However, genetic studies like ours might help other researchers to study causality between the gut microbiome and particular traits, which might potentially lead to new therapeutic targets. Next to genetic variants as a proxy, we’re currently studying the gut microbiome composition in myocardial infarction patients and healthy controls in a longitudinal setting.”
“Previous studies have suggested a potential link between the gut microbiome and the development of cardiovascular disease, type 2 diabetes mellitus, and other chronic disorders,” Carol Ann Remme, MD, of the Amsterdam University Medical Center, said in an interview. “However, it is challenging to study the effect of gut microbiome composition in large patient cohorts. As an alternative approach, the study authors showed in a very large population that genetic variants previously shown to influence gut microbiome composition were significantly associated with conditions such as hypertension, type 2 diabetes, hypercholesterolemia, and heart failure.”
The study is unique in that it employed a very large cohort of more than 400,000 individuals, which is typically required to be able to draw clear conclusions, Dr. Remme continued. “The authors were able to further refine their findings by linking genetic variants known to influence specific gut bacteria to some particular disorders,” she noted.
“It is becoming increasingly clear that an individual’s gut microbiome composition, which is defined by both genetic and environmental factors such as diet, may affect his/her susceptibility to certain diseases – including cardiovascular – in addition to disease progression and outcome,” said Dr. Remme. “This may ultimately lead to development of novel, personalized strategies for risk stratification in addition to potential preventive measures targeting the gut microbiome. I expect this area of research will become increasingly important in the coming years.”
The study received no outside funding. Dr. Groot and colleagues had no financial conflicts to disclose. Dr. Remme had no financial conflicts to disclose.
Microorganisms in the human digestive tract are linked to 29 specific health conditions, including chronic obstructive pulmonary disease, high blood pressure, and type 2 diabetes, according to a genome analysis in more than 400,000 individuals.
Although previous studies have suggested a link between gut microbiota and diseases in humans, “the extent to which the human gut microbiome can be considered a determinant of disease and healthy aging remains unknown,” Hilde E. Groot, MD, of the University of Groningen (The Netherlands), said in a presentation at the virtual annual congress of the European Society of Cardiology.
To identify the spectrum of diseases linked to the gut microbiome, the researchers identified 422,417 unrelated adults of White British ancestry with genotype and matching genetic data. The average age of the participants was 57 years and 46% were male.
The researchers conducted a phenomewide association study including 35 distinct single-nucleotide polymorphisms (SNPs) that are known to influence the microbiome of the human gut.
Overall, seven SNPs were significantly associated with 29 disease outcomes including hypertension, type 2 diabetes, hypercholesterolemia, heart failure, renal failure, and osteoarthritis.
In addition, after a further sensitivity analysis using a Mendelian randomization (MR) approach, associations between Ruminococcus flavefaciens and hypertension and between Clostridium and platelet count might point to a causal link, the researchers said.
“Over the past few years, the amount of research concerning the human gut microbiome and the associations with health and disease has tremendously increased. However, most studies investigated one or a few traits. The strength of our study is the possibility to cover a wide range of traits simultaneously within one population,” Dr. Groot said in an interview.
“Our data support the hypothesis that the human gut microbiome is a complex system, involved in many pathophysiological mechanisms in the human body. So, our results are additional to earlier research and strengthen this hypothesis,” Dr. Groot added.
“Microbiota and their metabolites might be of importance in the interplay between overlapping pathophysiological processes, and could serve as potential therapeutic targets for the maintenance of health and prevention and treatment of cardiovascular diseases. However, before it is possible to give advice for the public and medical practice, further research is needed to study causality,” she emphasized.
“Currently, it is too soon to advise patients concerning their microbiome,” Dr. Groot noted. “However, genetic studies like ours might help other researchers to study causality between the gut microbiome and particular traits, which might potentially lead to new therapeutic targets. Next to genetic variants as a proxy, we’re currently studying the gut microbiome composition in myocardial infarction patients and healthy controls in a longitudinal setting.”
“Previous studies have suggested a potential link between the gut microbiome and the development of cardiovascular disease, type 2 diabetes mellitus, and other chronic disorders,” Carol Ann Remme, MD, of the Amsterdam University Medical Center, said in an interview. “However, it is challenging to study the effect of gut microbiome composition in large patient cohorts. As an alternative approach, the study authors showed in a very large population that genetic variants previously shown to influence gut microbiome composition were significantly associated with conditions such as hypertension, type 2 diabetes, hypercholesterolemia, and heart failure.”
The study is unique in that it employed a very large cohort of more than 400,000 individuals, which is typically required to be able to draw clear conclusions, Dr. Remme continued. “The authors were able to further refine their findings by linking genetic variants known to influence specific gut bacteria to some particular disorders,” she noted.
“It is becoming increasingly clear that an individual’s gut microbiome composition, which is defined by both genetic and environmental factors such as diet, may affect his/her susceptibility to certain diseases – including cardiovascular – in addition to disease progression and outcome,” said Dr. Remme. “This may ultimately lead to development of novel, personalized strategies for risk stratification in addition to potential preventive measures targeting the gut microbiome. I expect this area of research will become increasingly important in the coming years.”
The study received no outside funding. Dr. Groot and colleagues had no financial conflicts to disclose. Dr. Remme had no financial conflicts to disclose.
FROM ESC CONGRESS 2020
ATPCI: Trimetazidine fizzles for post-PCI angina
Adding trimetazidine to optimal medical therapy does not improve outcomes following successful percutaneous coronary intervention (PCI) for stable angina or a non–ST-elevated myocardial infarction, results of the ATPCI trial show.
There was no benefit for the composite primary endpoint of cardiac death, hospitalization for cardiac events, or recurrent/persistent angina requiring an addition, switch, or increased dose of antianginal therapies, or requiring coronary angiography (hazard ratio, 0.98; 95% confidence interval, 0.88-1.09).
Further, there were no between-group differences in any of the individual components of the endpoint or any prespecified subgroups, Roberto Ferrari, MD, professor of cardiology at the University of Ferrara (Italy), reported in a Hot Line session at the digital European Society of Cardiology Congress 2020.
“I think one of the reasons why we couldn’t see any results was really due to this population was extremely well treated,” he said. “Almost all of them were receiving either a beta-blocker or calcium blocker and, on top of this, they had a successful angioplasty and that is what we should do, at least according to ESC guidelines.”
Research has shown that about 85% to 90% of patients have a change in New York Heart Association angina class within 30 days of PCI, leaving very little angina leftover to treat, observed Magnus Ohman, MD, director of the advanced coronary disease program at Duke University, Durham, N.C., who was not involved in the study.
“The fundamental question is whether this was the right study. Is this agent ineffective, or is it just that it was studied in the wrong population? That to me is really the crux of the matter,” he said in an interview.
There is potential benefit in chronic angina, which reflects the level II recommendation by the ESC, said Dr. Ohman. “Those patients typically require more therapy and, in the ideal world of treating angina, you need both physiological and metabolic agents to treat angina and trimetazidine is one metabolic agent.”
Trimetazidine is not available in the United States, but the anti-ischemic metabolic agent is recommended as second-line therapy for angina after beta-blockers and calcium-channel blockers in the 2019 ESC guidelines on chronic coronary syndrome.
Unlike other commonly used first- and second-line antianginal drugs, trimetazidine is devoid of hemodynamic effects, Dr. Ferrari said. It improves myocardial utilization by favoring glucose to fatty acids, thus allowing anaerobic adenosine triphosphate formation and preventing acidosis.
In the absence of contemporary data on the prognostic benefits of antianginal drugs in post-PCI patients, ATPCI investigators at 365 centers in 27 countries randomly assigned 6007 patients with stable angina or non–ST-segment MI after successful elective or urgent PCI to optimal medical therapy alone or with trimetazidine, 35 mg modified-release twice daily.
Patients with severe heart failure, valvular disease, arrhythmia, renal failure or acute ST-elevation MI were excluded.
Most patients (77% male) had Canadian Class Cardiovascular Society class III/IV angina (58%) and were receiving aspirin plus a P2Y12 inhibitor (97%), lipid-lowering agent (96.6%), renin-angiotensin inhibitors (82.2%), and beta-blockers (83.9%). A quarter were receiving calcium-channel blockers (27.6%). In all, 2517 patients had an urgent PCI and 3490 had an elective PCI.
After a median follow-up of 47.5 months, the composite primary endpoint occurred in 23.3% of the trimetazidine group and 23.7% of the control group, according to the study, which was published simultaneously in The Lancet.
The incidence of the individual components was similar:
- Cardiac death: 2.1% vs. 2.6% (HR, 0.81)
- Hospital admission for cardiac events: 13.4% vs. 13.4% (HR, 1.01)
- Angina leading to coronary angiography: 16.9% vs. 16.6% (HR, 1.02)
- Angina leading to increase/switch in antianginal drugs (HR, 1.01)
There was no between-group difference in the composite major secondary endpoint, which included the primary endpoint components plus ischemia leading to coronary angiography and an increase or switch in antianginal therapies. This outcome occurred in 23.5% and 24.0% of patients in the trimetazidine and control groups, respectively (HR, 0.98; 95% CI, 0.88-1.08).
Results were also similar when the primary endpoint was analyzed based on whether patients underwent elective PCI (HR, 0.94; 95% CI, 0.82-1.08) or urgent PCI (HR, 1.04; 95% CI, 0.88-1.22), Dr. Ferrari reported.
Given the lack of observed efficacy, trimetazidine has no use or place in the population studied, said Stephan Windecker, MD, the formal discussant for the study and chair of cardiology at Bern (Switzerland) University Hospital. “Notwithstanding, I think we have to recognize that the optimal medical therapy is so potent and has been well implemented in this trial that any additional medication beyond this is just unable to exploit additional benefit.”
The study was supported by Servier. Dr. Ferrari received fees, honoraria, and travel expenses from Servier. Dr. Ohman reports no relevant financial conflicts of interest. Dr. Windecker is an unpaid member of the steering/executive group for trials funded by Abbott, Abiomed, Amgen, BMS, Boston Scientific, Biotronik, Cardiovalve, Edwards Lifesciences, MedAlliancé, Medtronic, Polares, Sinomed, V-Wave, and Xeltis but has not received personal payments from any pharmaceutical or device company.
This article first appeared on Medscape.com.
Adding trimetazidine to optimal medical therapy does not improve outcomes following successful percutaneous coronary intervention (PCI) for stable angina or a non–ST-elevated myocardial infarction, results of the ATPCI trial show.
There was no benefit for the composite primary endpoint of cardiac death, hospitalization for cardiac events, or recurrent/persistent angina requiring an addition, switch, or increased dose of antianginal therapies, or requiring coronary angiography (hazard ratio, 0.98; 95% confidence interval, 0.88-1.09).
Further, there were no between-group differences in any of the individual components of the endpoint or any prespecified subgroups, Roberto Ferrari, MD, professor of cardiology at the University of Ferrara (Italy), reported in a Hot Line session at the digital European Society of Cardiology Congress 2020.
“I think one of the reasons why we couldn’t see any results was really due to this population was extremely well treated,” he said. “Almost all of them were receiving either a beta-blocker or calcium blocker and, on top of this, they had a successful angioplasty and that is what we should do, at least according to ESC guidelines.”
Research has shown that about 85% to 90% of patients have a change in New York Heart Association angina class within 30 days of PCI, leaving very little angina leftover to treat, observed Magnus Ohman, MD, director of the advanced coronary disease program at Duke University, Durham, N.C., who was not involved in the study.
“The fundamental question is whether this was the right study. Is this agent ineffective, or is it just that it was studied in the wrong population? That to me is really the crux of the matter,” he said in an interview.
There is potential benefit in chronic angina, which reflects the level II recommendation by the ESC, said Dr. Ohman. “Those patients typically require more therapy and, in the ideal world of treating angina, you need both physiological and metabolic agents to treat angina and trimetazidine is one metabolic agent.”
Trimetazidine is not available in the United States, but the anti-ischemic metabolic agent is recommended as second-line therapy for angina after beta-blockers and calcium-channel blockers in the 2019 ESC guidelines on chronic coronary syndrome.
Unlike other commonly used first- and second-line antianginal drugs, trimetazidine is devoid of hemodynamic effects, Dr. Ferrari said. It improves myocardial utilization by favoring glucose to fatty acids, thus allowing anaerobic adenosine triphosphate formation and preventing acidosis.
In the absence of contemporary data on the prognostic benefits of antianginal drugs in post-PCI patients, ATPCI investigators at 365 centers in 27 countries randomly assigned 6007 patients with stable angina or non–ST-segment MI after successful elective or urgent PCI to optimal medical therapy alone or with trimetazidine, 35 mg modified-release twice daily.
Patients with severe heart failure, valvular disease, arrhythmia, renal failure or acute ST-elevation MI were excluded.
Most patients (77% male) had Canadian Class Cardiovascular Society class III/IV angina (58%) and were receiving aspirin plus a P2Y12 inhibitor (97%), lipid-lowering agent (96.6%), renin-angiotensin inhibitors (82.2%), and beta-blockers (83.9%). A quarter were receiving calcium-channel blockers (27.6%). In all, 2517 patients had an urgent PCI and 3490 had an elective PCI.
After a median follow-up of 47.5 months, the composite primary endpoint occurred in 23.3% of the trimetazidine group and 23.7% of the control group, according to the study, which was published simultaneously in The Lancet.
The incidence of the individual components was similar:
- Cardiac death: 2.1% vs. 2.6% (HR, 0.81)
- Hospital admission for cardiac events: 13.4% vs. 13.4% (HR, 1.01)
- Angina leading to coronary angiography: 16.9% vs. 16.6% (HR, 1.02)
- Angina leading to increase/switch in antianginal drugs (HR, 1.01)
There was no between-group difference in the composite major secondary endpoint, which included the primary endpoint components plus ischemia leading to coronary angiography and an increase or switch in antianginal therapies. This outcome occurred in 23.5% and 24.0% of patients in the trimetazidine and control groups, respectively (HR, 0.98; 95% CI, 0.88-1.08).
Results were also similar when the primary endpoint was analyzed based on whether patients underwent elective PCI (HR, 0.94; 95% CI, 0.82-1.08) or urgent PCI (HR, 1.04; 95% CI, 0.88-1.22), Dr. Ferrari reported.
Given the lack of observed efficacy, trimetazidine has no use or place in the population studied, said Stephan Windecker, MD, the formal discussant for the study and chair of cardiology at Bern (Switzerland) University Hospital. “Notwithstanding, I think we have to recognize that the optimal medical therapy is so potent and has been well implemented in this trial that any additional medication beyond this is just unable to exploit additional benefit.”
The study was supported by Servier. Dr. Ferrari received fees, honoraria, and travel expenses from Servier. Dr. Ohman reports no relevant financial conflicts of interest. Dr. Windecker is an unpaid member of the steering/executive group for trials funded by Abbott, Abiomed, Amgen, BMS, Boston Scientific, Biotronik, Cardiovalve, Edwards Lifesciences, MedAlliancé, Medtronic, Polares, Sinomed, V-Wave, and Xeltis but has not received personal payments from any pharmaceutical or device company.
This article first appeared on Medscape.com.
Adding trimetazidine to optimal medical therapy does not improve outcomes following successful percutaneous coronary intervention (PCI) for stable angina or a non–ST-elevated myocardial infarction, results of the ATPCI trial show.
There was no benefit for the composite primary endpoint of cardiac death, hospitalization for cardiac events, or recurrent/persistent angina requiring an addition, switch, or increased dose of antianginal therapies, or requiring coronary angiography (hazard ratio, 0.98; 95% confidence interval, 0.88-1.09).
Further, there were no between-group differences in any of the individual components of the endpoint or any prespecified subgroups, Roberto Ferrari, MD, professor of cardiology at the University of Ferrara (Italy), reported in a Hot Line session at the digital European Society of Cardiology Congress 2020.
“I think one of the reasons why we couldn’t see any results was really due to this population was extremely well treated,” he said. “Almost all of them were receiving either a beta-blocker or calcium blocker and, on top of this, they had a successful angioplasty and that is what we should do, at least according to ESC guidelines.”
Research has shown that about 85% to 90% of patients have a change in New York Heart Association angina class within 30 days of PCI, leaving very little angina leftover to treat, observed Magnus Ohman, MD, director of the advanced coronary disease program at Duke University, Durham, N.C., who was not involved in the study.
“The fundamental question is whether this was the right study. Is this agent ineffective, or is it just that it was studied in the wrong population? That to me is really the crux of the matter,” he said in an interview.
There is potential benefit in chronic angina, which reflects the level II recommendation by the ESC, said Dr. Ohman. “Those patients typically require more therapy and, in the ideal world of treating angina, you need both physiological and metabolic agents to treat angina and trimetazidine is one metabolic agent.”
Trimetazidine is not available in the United States, but the anti-ischemic metabolic agent is recommended as second-line therapy for angina after beta-blockers and calcium-channel blockers in the 2019 ESC guidelines on chronic coronary syndrome.
Unlike other commonly used first- and second-line antianginal drugs, trimetazidine is devoid of hemodynamic effects, Dr. Ferrari said. It improves myocardial utilization by favoring glucose to fatty acids, thus allowing anaerobic adenosine triphosphate formation and preventing acidosis.
In the absence of contemporary data on the prognostic benefits of antianginal drugs in post-PCI patients, ATPCI investigators at 365 centers in 27 countries randomly assigned 6007 patients with stable angina or non–ST-segment MI after successful elective or urgent PCI to optimal medical therapy alone or with trimetazidine, 35 mg modified-release twice daily.
Patients with severe heart failure, valvular disease, arrhythmia, renal failure or acute ST-elevation MI were excluded.
Most patients (77% male) had Canadian Class Cardiovascular Society class III/IV angina (58%) and were receiving aspirin plus a P2Y12 inhibitor (97%), lipid-lowering agent (96.6%), renin-angiotensin inhibitors (82.2%), and beta-blockers (83.9%). A quarter were receiving calcium-channel blockers (27.6%). In all, 2517 patients had an urgent PCI and 3490 had an elective PCI.
After a median follow-up of 47.5 months, the composite primary endpoint occurred in 23.3% of the trimetazidine group and 23.7% of the control group, according to the study, which was published simultaneously in The Lancet.
The incidence of the individual components was similar:
- Cardiac death: 2.1% vs. 2.6% (HR, 0.81)
- Hospital admission for cardiac events: 13.4% vs. 13.4% (HR, 1.01)
- Angina leading to coronary angiography: 16.9% vs. 16.6% (HR, 1.02)
- Angina leading to increase/switch in antianginal drugs (HR, 1.01)
There was no between-group difference in the composite major secondary endpoint, which included the primary endpoint components plus ischemia leading to coronary angiography and an increase or switch in antianginal therapies. This outcome occurred in 23.5% and 24.0% of patients in the trimetazidine and control groups, respectively (HR, 0.98; 95% CI, 0.88-1.08).
Results were also similar when the primary endpoint was analyzed based on whether patients underwent elective PCI (HR, 0.94; 95% CI, 0.82-1.08) or urgent PCI (HR, 1.04; 95% CI, 0.88-1.22), Dr. Ferrari reported.
Given the lack of observed efficacy, trimetazidine has no use or place in the population studied, said Stephan Windecker, MD, the formal discussant for the study and chair of cardiology at Bern (Switzerland) University Hospital. “Notwithstanding, I think we have to recognize that the optimal medical therapy is so potent and has been well implemented in this trial that any additional medication beyond this is just unable to exploit additional benefit.”
The study was supported by Servier. Dr. Ferrari received fees, honoraria, and travel expenses from Servier. Dr. Ohman reports no relevant financial conflicts of interest. Dr. Windecker is an unpaid member of the steering/executive group for trials funded by Abbott, Abiomed, Amgen, BMS, Boston Scientific, Biotronik, Cardiovalve, Edwards Lifesciences, MedAlliancé, Medtronic, Polares, Sinomed, V-Wave, and Xeltis but has not received personal payments from any pharmaceutical or device company.
This article first appeared on Medscape.com.
Two PR employees at FDA fired after plasma therapy controversy
The US Food and Drug Administration has removed two senior public relations employees, one of whom advised the agency against unbridled promotion of convalescent blood plasma as a treatment for people with COVID-19, multiple media outlets reported Aug. 28.
Officials claim the dismissals are coincidental and are not related to a controversy about whether claims regarding convalescent plasma therapy that were put forth by President Donald Trump and FDA Commissioner Stephen M. Hahn, MD, were exaggerated, according to reports from The New York Times , CNN, and elsewhere.
One of the PR employees, Emily Miller, was on the job less than 2 weeks. The White House named her FDA chief spokeswoman 11 days ago, but Hahn removed her from that post Aug. 28.
On Aug. 27, the US Department of Health and Human Services terminated the contract for Wayne L. Pines, a PR consultant to the FDA. Pines reportedly advised Hahn to apologize for making misleading claims about the therapeutic benefits of convalescent plasma therapy for COVID-19.
The FDA did not respond to multiple requests for comment.
The controversy stems from comments Hahn made about the announcement of the emergency use authorization for convalescent plasma for patients with COVID-19. He said that plasma had been found to save the lives of 35 out of every 100 people who were treated. That statement was later found to be erroneous because he presented a relative risk reduction as an absolute decrease in risk. He later apologized via Twitter.
Researchers running clinical trials to evaluate the efficacy of convalescent plasma for COVID-19 are concerned that the emergency use authorization could thwart efforts to recruit participants for their studies.
This article first appeared on Medscape.com.
The US Food and Drug Administration has removed two senior public relations employees, one of whom advised the agency against unbridled promotion of convalescent blood plasma as a treatment for people with COVID-19, multiple media outlets reported Aug. 28.
Officials claim the dismissals are coincidental and are not related to a controversy about whether claims regarding convalescent plasma therapy that were put forth by President Donald Trump and FDA Commissioner Stephen M. Hahn, MD, were exaggerated, according to reports from The New York Times , CNN, and elsewhere.
One of the PR employees, Emily Miller, was on the job less than 2 weeks. The White House named her FDA chief spokeswoman 11 days ago, but Hahn removed her from that post Aug. 28.
On Aug. 27, the US Department of Health and Human Services terminated the contract for Wayne L. Pines, a PR consultant to the FDA. Pines reportedly advised Hahn to apologize for making misleading claims about the therapeutic benefits of convalescent plasma therapy for COVID-19.
The FDA did not respond to multiple requests for comment.
The controversy stems from comments Hahn made about the announcement of the emergency use authorization for convalescent plasma for patients with COVID-19. He said that plasma had been found to save the lives of 35 out of every 100 people who were treated. That statement was later found to be erroneous because he presented a relative risk reduction as an absolute decrease in risk. He later apologized via Twitter.
Researchers running clinical trials to evaluate the efficacy of convalescent plasma for COVID-19 are concerned that the emergency use authorization could thwart efforts to recruit participants for their studies.
This article first appeared on Medscape.com.
The US Food and Drug Administration has removed two senior public relations employees, one of whom advised the agency against unbridled promotion of convalescent blood plasma as a treatment for people with COVID-19, multiple media outlets reported Aug. 28.
Officials claim the dismissals are coincidental and are not related to a controversy about whether claims regarding convalescent plasma therapy that were put forth by President Donald Trump and FDA Commissioner Stephen M. Hahn, MD, were exaggerated, according to reports from The New York Times , CNN, and elsewhere.
One of the PR employees, Emily Miller, was on the job less than 2 weeks. The White House named her FDA chief spokeswoman 11 days ago, but Hahn removed her from that post Aug. 28.
On Aug. 27, the US Department of Health and Human Services terminated the contract for Wayne L. Pines, a PR consultant to the FDA. Pines reportedly advised Hahn to apologize for making misleading claims about the therapeutic benefits of convalescent plasma therapy for COVID-19.
The FDA did not respond to multiple requests for comment.
The controversy stems from comments Hahn made about the announcement of the emergency use authorization for convalescent plasma for patients with COVID-19. He said that plasma had been found to save the lives of 35 out of every 100 people who were treated. That statement was later found to be erroneous because he presented a relative risk reduction as an absolute decrease in risk. He later apologized via Twitter.
Researchers running clinical trials to evaluate the efficacy of convalescent plasma for COVID-19 are concerned that the emergency use authorization could thwart efforts to recruit participants for their studies.
This article first appeared on Medscape.com.
DAPA-CKD: SGLT2 inhibitor benefit extends to chronic kidney disease without diabetes
In the DAPA-CKD trial, treatment with the SGLT2 inhibitor dapagliflozin (Farxiga) cut the incidence of substantially worsened chronic kidney disease by an average of 39% compared with placebo when added to standard treatment, with a number needed to treat of 19 to prevent one primary outcome event after a median of 2.4 years.
The level of benefit was similar in both the one-third of enrolled patients without diabetes and in the two-thirds with diabetes, showing a statistically significant 50% cut in the primary endpoint among patients without diabetes, Hiddo J.L. Heerspink, MD, reported at the virtual annual congress of the European Society of Cardiology.
“We found that dapagliflozin delayed the initiation of dialysis, and reduced the number of deaths,” regardless of diabetes status, Dr. Heerspink, of University Medical Centre Groningen, the Netherlands, said during a press conference. “DAPA-CKD trial has shown dapagliflozin’s potential as a long-awaited new treatment for patients with chronic kidney disease.”
This finding ushers in a “completely new era in chronic kidney disease management,” said Janani Rangaswami, MD, a nephrologist and cardiorenal syndrome specialist at Einstein Medical Center in Philadelphia. “It’s good news” for these patients.
The results showed that dapagliflozin is the first “game changing” drug for chronic kidney disease in 2 decades, following the introduction of angiotensin converting enzyme inhibitors and angiotensin receptor blockers, she said in an interview. And given the consistency of the findings with the results from several other studies that documented meaningful renal protection by several different SGLT2 inhibitors, the results from this single trial also convincingly establish dapagliflozin as a standard-of-care agent to use on the types of patients the study enrolled, she said in an interview.
Representing many real-world patients
The DAPA-CKD trial enrolled 4,304 patients with albuminuria based on having a urinary albumin-to-creatinine ratio of at least 200 mg/g, and an estimated glomerular filtration rate (eGFR) of 25-75 mL/min per 1.73 m2 (with 90% of patients having an eGFR of less than 60 mL/min per 1.73 m2), and 97% were on treatment with a renin-angiotensin system–blocking drug. The primary endpoint was the combined rate of a drop in eGFR of at least 50% from baseline, progression to end stage renal disease, or renal or cardiovascular death; the between-group difference in this composite was driven primarily by both preserved eGFR and by prevention of end stage renal disease.
This represents both an appropriate target population, and meaningful endpoints, Dr. Rangaswami said. The study was “very representative of who we see in real-world practice,” a group that likely includes “hundreds of thousands” of U.S. patients with nondiabetic chronic kidney disease, she estimated.
Another notable finding was that 14% of the enrolled patients had eGFR values at baseline of 25-29 mL/min per 1.73 m2, pegging them as having stage 4 chronic kidney disease, and the median baseline eGFR was 43 mL/min per 1.73 m2, but dapagliflozin treatment was as safe and effective in these patients as it was in enrolled patients with a higher level of retained renal activity. This experience should give clinicians greater confidence about using dapagliflozin and other drugs in the sodium-glucose cotransporter (SGLT) 2 inhibitor class in patients with substantially depressed renal function, Dr. Rangaswami said.
“We now need to be more proactive about treating patients with more advanced kidney disease who can still benefit” from dapagliflozin treatment. “The sooner you intervene the better,” to slow further progression, but the new findings show “benefit even when treating patients with lower eGFRs. There is still hope to prevent or delay dialysis.”
A heart-kidney connection
Dapagliflozin treatment also cut all-cause mortality by a statistically significant, relative 31%, and another secondary-endpoint analysis showed a statistically significant 29% relative reduction in the rate of cardiovascular death or heart failure hospitalization, a benefit seen consistently in several prior studies of SGLT2 inhibitors, but possibly unexpected here because enrolled patients underwent no selection for a history of heart failure or any other cardiovascular disease. But the finding shouldn’t surprise, because “chronic kidney disease is an independent risk factor for cardiovascular disease across the board, and especially for heart failure,” noted Dr. Rangaswami.
“Heart and kidney disease is one big spectrum,” and the collected experience of several trials that have now proven the efficacy of SGLT2 inhibitors among patients with heart failure with reduced ejection fraction or with chronic kidney disease, regardless of their glycemic control, shows how broadly this drug class can benefit patients across the breadth of this spectrum, she said.
DAPA-CKD was funded by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Heerspink has been a consultant to and received research funding from AstraZeneca and from several other companies. Dr. Rangaswami had no disclosures.
In the DAPA-CKD trial, treatment with the SGLT2 inhibitor dapagliflozin (Farxiga) cut the incidence of substantially worsened chronic kidney disease by an average of 39% compared with placebo when added to standard treatment, with a number needed to treat of 19 to prevent one primary outcome event after a median of 2.4 years.
The level of benefit was similar in both the one-third of enrolled patients without diabetes and in the two-thirds with diabetes, showing a statistically significant 50% cut in the primary endpoint among patients without diabetes, Hiddo J.L. Heerspink, MD, reported at the virtual annual congress of the European Society of Cardiology.
“We found that dapagliflozin delayed the initiation of dialysis, and reduced the number of deaths,” regardless of diabetes status, Dr. Heerspink, of University Medical Centre Groningen, the Netherlands, said during a press conference. “DAPA-CKD trial has shown dapagliflozin’s potential as a long-awaited new treatment for patients with chronic kidney disease.”
This finding ushers in a “completely new era in chronic kidney disease management,” said Janani Rangaswami, MD, a nephrologist and cardiorenal syndrome specialist at Einstein Medical Center in Philadelphia. “It’s good news” for these patients.
The results showed that dapagliflozin is the first “game changing” drug for chronic kidney disease in 2 decades, following the introduction of angiotensin converting enzyme inhibitors and angiotensin receptor blockers, she said in an interview. And given the consistency of the findings with the results from several other studies that documented meaningful renal protection by several different SGLT2 inhibitors, the results from this single trial also convincingly establish dapagliflozin as a standard-of-care agent to use on the types of patients the study enrolled, she said in an interview.
Representing many real-world patients
The DAPA-CKD trial enrolled 4,304 patients with albuminuria based on having a urinary albumin-to-creatinine ratio of at least 200 mg/g, and an estimated glomerular filtration rate (eGFR) of 25-75 mL/min per 1.73 m2 (with 90% of patients having an eGFR of less than 60 mL/min per 1.73 m2), and 97% were on treatment with a renin-angiotensin system–blocking drug. The primary endpoint was the combined rate of a drop in eGFR of at least 50% from baseline, progression to end stage renal disease, or renal or cardiovascular death; the between-group difference in this composite was driven primarily by both preserved eGFR and by prevention of end stage renal disease.
This represents both an appropriate target population, and meaningful endpoints, Dr. Rangaswami said. The study was “very representative of who we see in real-world practice,” a group that likely includes “hundreds of thousands” of U.S. patients with nondiabetic chronic kidney disease, she estimated.
Another notable finding was that 14% of the enrolled patients had eGFR values at baseline of 25-29 mL/min per 1.73 m2, pegging them as having stage 4 chronic kidney disease, and the median baseline eGFR was 43 mL/min per 1.73 m2, but dapagliflozin treatment was as safe and effective in these patients as it was in enrolled patients with a higher level of retained renal activity. This experience should give clinicians greater confidence about using dapagliflozin and other drugs in the sodium-glucose cotransporter (SGLT) 2 inhibitor class in patients with substantially depressed renal function, Dr. Rangaswami said.
“We now need to be more proactive about treating patients with more advanced kidney disease who can still benefit” from dapagliflozin treatment. “The sooner you intervene the better,” to slow further progression, but the new findings show “benefit even when treating patients with lower eGFRs. There is still hope to prevent or delay dialysis.”
A heart-kidney connection
Dapagliflozin treatment also cut all-cause mortality by a statistically significant, relative 31%, and another secondary-endpoint analysis showed a statistically significant 29% relative reduction in the rate of cardiovascular death or heart failure hospitalization, a benefit seen consistently in several prior studies of SGLT2 inhibitors, but possibly unexpected here because enrolled patients underwent no selection for a history of heart failure or any other cardiovascular disease. But the finding shouldn’t surprise, because “chronic kidney disease is an independent risk factor for cardiovascular disease across the board, and especially for heart failure,” noted Dr. Rangaswami.
“Heart and kidney disease is one big spectrum,” and the collected experience of several trials that have now proven the efficacy of SGLT2 inhibitors among patients with heart failure with reduced ejection fraction or with chronic kidney disease, regardless of their glycemic control, shows how broadly this drug class can benefit patients across the breadth of this spectrum, she said.
DAPA-CKD was funded by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Heerspink has been a consultant to and received research funding from AstraZeneca and from several other companies. Dr. Rangaswami had no disclosures.
In the DAPA-CKD trial, treatment with the SGLT2 inhibitor dapagliflozin (Farxiga) cut the incidence of substantially worsened chronic kidney disease by an average of 39% compared with placebo when added to standard treatment, with a number needed to treat of 19 to prevent one primary outcome event after a median of 2.4 years.
The level of benefit was similar in both the one-third of enrolled patients without diabetes and in the two-thirds with diabetes, showing a statistically significant 50% cut in the primary endpoint among patients without diabetes, Hiddo J.L. Heerspink, MD, reported at the virtual annual congress of the European Society of Cardiology.
“We found that dapagliflozin delayed the initiation of dialysis, and reduced the number of deaths,” regardless of diabetes status, Dr. Heerspink, of University Medical Centre Groningen, the Netherlands, said during a press conference. “DAPA-CKD trial has shown dapagliflozin’s potential as a long-awaited new treatment for patients with chronic kidney disease.”
This finding ushers in a “completely new era in chronic kidney disease management,” said Janani Rangaswami, MD, a nephrologist and cardiorenal syndrome specialist at Einstein Medical Center in Philadelphia. “It’s good news” for these patients.
The results showed that dapagliflozin is the first “game changing” drug for chronic kidney disease in 2 decades, following the introduction of angiotensin converting enzyme inhibitors and angiotensin receptor blockers, she said in an interview. And given the consistency of the findings with the results from several other studies that documented meaningful renal protection by several different SGLT2 inhibitors, the results from this single trial also convincingly establish dapagliflozin as a standard-of-care agent to use on the types of patients the study enrolled, she said in an interview.
Representing many real-world patients
The DAPA-CKD trial enrolled 4,304 patients with albuminuria based on having a urinary albumin-to-creatinine ratio of at least 200 mg/g, and an estimated glomerular filtration rate (eGFR) of 25-75 mL/min per 1.73 m2 (with 90% of patients having an eGFR of less than 60 mL/min per 1.73 m2), and 97% were on treatment with a renin-angiotensin system–blocking drug. The primary endpoint was the combined rate of a drop in eGFR of at least 50% from baseline, progression to end stage renal disease, or renal or cardiovascular death; the between-group difference in this composite was driven primarily by both preserved eGFR and by prevention of end stage renal disease.
This represents both an appropriate target population, and meaningful endpoints, Dr. Rangaswami said. The study was “very representative of who we see in real-world practice,” a group that likely includes “hundreds of thousands” of U.S. patients with nondiabetic chronic kidney disease, she estimated.
Another notable finding was that 14% of the enrolled patients had eGFR values at baseline of 25-29 mL/min per 1.73 m2, pegging them as having stage 4 chronic kidney disease, and the median baseline eGFR was 43 mL/min per 1.73 m2, but dapagliflozin treatment was as safe and effective in these patients as it was in enrolled patients with a higher level of retained renal activity. This experience should give clinicians greater confidence about using dapagliflozin and other drugs in the sodium-glucose cotransporter (SGLT) 2 inhibitor class in patients with substantially depressed renal function, Dr. Rangaswami said.
“We now need to be more proactive about treating patients with more advanced kidney disease who can still benefit” from dapagliflozin treatment. “The sooner you intervene the better,” to slow further progression, but the new findings show “benefit even when treating patients with lower eGFRs. There is still hope to prevent or delay dialysis.”
A heart-kidney connection
Dapagliflozin treatment also cut all-cause mortality by a statistically significant, relative 31%, and another secondary-endpoint analysis showed a statistically significant 29% relative reduction in the rate of cardiovascular death or heart failure hospitalization, a benefit seen consistently in several prior studies of SGLT2 inhibitors, but possibly unexpected here because enrolled patients underwent no selection for a history of heart failure or any other cardiovascular disease. But the finding shouldn’t surprise, because “chronic kidney disease is an independent risk factor for cardiovascular disease across the board, and especially for heart failure,” noted Dr. Rangaswami.
“Heart and kidney disease is one big spectrum,” and the collected experience of several trials that have now proven the efficacy of SGLT2 inhibitors among patients with heart failure with reduced ejection fraction or with chronic kidney disease, regardless of their glycemic control, shows how broadly this drug class can benefit patients across the breadth of this spectrum, she said.
DAPA-CKD was funded by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Heerspink has been a consultant to and received research funding from AstraZeneca and from several other companies. Dr. Rangaswami had no disclosures.
FROM ESC CONGRESS 2020
RATE-AF trial boosts digoxin for rate control in permanent AFib
Digoxin now deserves to be considered first-line therapy for long-term heart rate control in older patients with permanent atrial fibrillation and symptoms of heart failure, Dipak Kotecha, MBChB, PhD, MSc, declared at the virtual annual congress of the European Society of Cardiology.
He presented the 12-month results of RATE-AF (Rate Control Therapy Evaluation in Permanent Atrial Fibrillation), in which 160 seniors (mean age, 76 years) with moderate or severe symptoms caused by permanent atrial fibrillation (AFib) as well as heart failure symptoms were randomized to low-dose digoxin or the beta-blocker bisoprolol for rate control.
The open-label trial was designed to address a centuries-old unmet need: “Although digoxin has been in use since 1785, we have no longer-term clinical trials of digoxin in patients with AFib or AFib with heart failure,” noted Dr. Kotecha, professor of cardiology at the University of Birmingham (England).
Not only is digoxin greatly understudied in AFib, but permanent AFib – the most common form of the arrhythmia – has received only a tiny fraction of the research attention that’s been devoted to paroxysmal or persistent AFib, he added.
In RATE-AF, digoxin and bisoprolol proved similarly effective at reducing heart rate, from about 100 bpm at baseline to the mid-70s at 6 and 12 months. Notably, only a handful of study participants required an additional rate control drug during the 12-month study. Nor did the two drugs differ in terms of their impact on patient-reported quality of life at 6 months as reflected by their Short Form–36 physical component score, the primary study endpoint. And both drugs were well tolerated, with 96% of patients in the digoxin group still on the drug at a mean of 161 mcg/day at 6 months, and 89% still on their beta-blocker.
But that’s pretty much where the similarities in outcomes ended.
For example, at 12 months, the digoxin group scored significantly higher than the beta-blocker group on several domains of the Short Form–36 physical component score, including vitality, physical function, and global health. More than half of the digoxin group had a two-class improvement in modified European Heart Rhythm Association AFib-related symptoms at 6 months, compared with 10% of the beta-blocker group. At 12 months, nearly 70% of the digoxin group had a two-class improvement, as did 30% on bisoprolol.
Heart failure symptoms in the digoxin group improved from a mean baseline New York Heart Association class of 2.4 to 1.5 at both 6 and 12 months; the improvement was more modest in the beta-blocker group, going from NYHA 2.4 at baseline to 2.0 at both 6 and 12 months. And while N-terminal of the prohormone brain natriuretic peptide levels improved in the digoxin group from a baseline of 1,095 pg/mL to 1,058 at 6 months and 960 at 12 months, NT-proBNP actually went up in the beta-blocker group, from 1,041 to 1,209-1,250 pg/mL at 12 months.
Moreover, Dr. Kotecha continued, while RATE-AF was underpowered to assess clinical events, it’s nevertheless noteworthy that a total of 29 adverse events occurred in 12 months in the digoxin group, compared with 142 with beta-blocker therapy. There were 12 unplanned hospital admissions in the digoxin group and 28 in the beta-blocker group, and 22 primary care visits for either AFib or cardiovascular symptoms in patients on digoxin versus 64 in the beta-blocker group.
“Our results suggest a wider use of digoxin for stable patients with permanent AFib,” Dr. Kotecha concluded. However, in an interview, Jonathan Piccini, MD, had a different take on the study results.
“I don’t think this study should widely impact clinical practice in the U.S.,” according to Dr. Piccini, director of cardiac electrophysiology at Duke University, Durham, N.C.
His reservations included RATE-AF’s modest sample size as well as uncertainty as to the trial’s generalizability, given that bisoprolol isn’t much used in the United States. Also, these were elderly patients with shortness of breath, and it’s unclear how effective digoxin would be for rate control in patients with permanent AFib who are more active.
“The classic teaching is that digoxin is great for rate control at rest, but when people are active it’s not nearly as good as beta-blockers or calcium-channel blockers,” the cardiologist said.
“A beta-blocker is still going to be my first-line rate control agent. But the results of RATE-AF do open my mind that for an older sedentary patient I may very well think twice now about using digoxin, because in that situation it looks like it achieves similar goals as a beta-blocker,” Dr. Piccini added.
On the plus side for RATE-AF: “I am very pleased to see that we have a randomized controlled trial focused on rate control in permanent AFib. It also tickles me pink to see a randomized, controlled study of digoxin. And I’m really excited to see a clinical trial that focuses on quality of life. It should give some confidence to know that from a quality of life perspective clinicians can consider using either digoxin or a beta-blocker for rate control,” he said.
American College of Cardiology vice president Dipti Itchhaporia, MD, said she’d need to see a much larger randomized trial including a calcium-channel blocker as a third-rate control arm before she’d consider digoxin as first-line rate control therapy in patients with AFib with or without heart failure. Also, she has reservations about drawing definitive conclusions from an open, unblinded study in which patient-reported outcomes are the primary endpoint.
“I think these were surprising findings given what we all think about digoxin in this country. In general, digoxin fell out of favor for rate control, mainly because of observational studies showing increased mortality. So most of us choose a beta-blocker,” she observed in an interview.
But of course, a randomized trial, even a 160-patient randomized trial, constitutes a higher level of evidence.
“I don’t think I’m going to convert tomorrow and make digoxin my first-line rate control therapy without more data. But RATE-AF does makes me stop and think about using it more than I did before in some of my permanent AFib patients,” said Dr. Itchhaporia, director of disease management at Hoag Memorial Hospital in Newport Beach, Calif.
Dr. Kotecha reported having no financial conflicts regarding the study, which was funded by the U.K. National Institute for Health Research, the British Heart Foundation, and the European Union.
Digoxin now deserves to be considered first-line therapy for long-term heart rate control in older patients with permanent atrial fibrillation and symptoms of heart failure, Dipak Kotecha, MBChB, PhD, MSc, declared at the virtual annual congress of the European Society of Cardiology.
He presented the 12-month results of RATE-AF (Rate Control Therapy Evaluation in Permanent Atrial Fibrillation), in which 160 seniors (mean age, 76 years) with moderate or severe symptoms caused by permanent atrial fibrillation (AFib) as well as heart failure symptoms were randomized to low-dose digoxin or the beta-blocker bisoprolol for rate control.
The open-label trial was designed to address a centuries-old unmet need: “Although digoxin has been in use since 1785, we have no longer-term clinical trials of digoxin in patients with AFib or AFib with heart failure,” noted Dr. Kotecha, professor of cardiology at the University of Birmingham (England).
Not only is digoxin greatly understudied in AFib, but permanent AFib – the most common form of the arrhythmia – has received only a tiny fraction of the research attention that’s been devoted to paroxysmal or persistent AFib, he added.
In RATE-AF, digoxin and bisoprolol proved similarly effective at reducing heart rate, from about 100 bpm at baseline to the mid-70s at 6 and 12 months. Notably, only a handful of study participants required an additional rate control drug during the 12-month study. Nor did the two drugs differ in terms of their impact on patient-reported quality of life at 6 months as reflected by their Short Form–36 physical component score, the primary study endpoint. And both drugs were well tolerated, with 96% of patients in the digoxin group still on the drug at a mean of 161 mcg/day at 6 months, and 89% still on their beta-blocker.
But that’s pretty much where the similarities in outcomes ended.
For example, at 12 months, the digoxin group scored significantly higher than the beta-blocker group on several domains of the Short Form–36 physical component score, including vitality, physical function, and global health. More than half of the digoxin group had a two-class improvement in modified European Heart Rhythm Association AFib-related symptoms at 6 months, compared with 10% of the beta-blocker group. At 12 months, nearly 70% of the digoxin group had a two-class improvement, as did 30% on bisoprolol.
Heart failure symptoms in the digoxin group improved from a mean baseline New York Heart Association class of 2.4 to 1.5 at both 6 and 12 months; the improvement was more modest in the beta-blocker group, going from NYHA 2.4 at baseline to 2.0 at both 6 and 12 months. And while N-terminal of the prohormone brain natriuretic peptide levels improved in the digoxin group from a baseline of 1,095 pg/mL to 1,058 at 6 months and 960 at 12 months, NT-proBNP actually went up in the beta-blocker group, from 1,041 to 1,209-1,250 pg/mL at 12 months.
Moreover, Dr. Kotecha continued, while RATE-AF was underpowered to assess clinical events, it’s nevertheless noteworthy that a total of 29 adverse events occurred in 12 months in the digoxin group, compared with 142 with beta-blocker therapy. There were 12 unplanned hospital admissions in the digoxin group and 28 in the beta-blocker group, and 22 primary care visits for either AFib or cardiovascular symptoms in patients on digoxin versus 64 in the beta-blocker group.
“Our results suggest a wider use of digoxin for stable patients with permanent AFib,” Dr. Kotecha concluded. However, in an interview, Jonathan Piccini, MD, had a different take on the study results.
“I don’t think this study should widely impact clinical practice in the U.S.,” according to Dr. Piccini, director of cardiac electrophysiology at Duke University, Durham, N.C.
His reservations included RATE-AF’s modest sample size as well as uncertainty as to the trial’s generalizability, given that bisoprolol isn’t much used in the United States. Also, these were elderly patients with shortness of breath, and it’s unclear how effective digoxin would be for rate control in patients with permanent AFib who are more active.
“The classic teaching is that digoxin is great for rate control at rest, but when people are active it’s not nearly as good as beta-blockers or calcium-channel blockers,” the cardiologist said.
“A beta-blocker is still going to be my first-line rate control agent. But the results of RATE-AF do open my mind that for an older sedentary patient I may very well think twice now about using digoxin, because in that situation it looks like it achieves similar goals as a beta-blocker,” Dr. Piccini added.
On the plus side for RATE-AF: “I am very pleased to see that we have a randomized controlled trial focused on rate control in permanent AFib. It also tickles me pink to see a randomized, controlled study of digoxin. And I’m really excited to see a clinical trial that focuses on quality of life. It should give some confidence to know that from a quality of life perspective clinicians can consider using either digoxin or a beta-blocker for rate control,” he said.
American College of Cardiology vice president Dipti Itchhaporia, MD, said she’d need to see a much larger randomized trial including a calcium-channel blocker as a third-rate control arm before she’d consider digoxin as first-line rate control therapy in patients with AFib with or without heart failure. Also, she has reservations about drawing definitive conclusions from an open, unblinded study in which patient-reported outcomes are the primary endpoint.
“I think these were surprising findings given what we all think about digoxin in this country. In general, digoxin fell out of favor for rate control, mainly because of observational studies showing increased mortality. So most of us choose a beta-blocker,” she observed in an interview.
But of course, a randomized trial, even a 160-patient randomized trial, constitutes a higher level of evidence.
“I don’t think I’m going to convert tomorrow and make digoxin my first-line rate control therapy without more data. But RATE-AF does makes me stop and think about using it more than I did before in some of my permanent AFib patients,” said Dr. Itchhaporia, director of disease management at Hoag Memorial Hospital in Newport Beach, Calif.
Dr. Kotecha reported having no financial conflicts regarding the study, which was funded by the U.K. National Institute for Health Research, the British Heart Foundation, and the European Union.
Digoxin now deserves to be considered first-line therapy for long-term heart rate control in older patients with permanent atrial fibrillation and symptoms of heart failure, Dipak Kotecha, MBChB, PhD, MSc, declared at the virtual annual congress of the European Society of Cardiology.
He presented the 12-month results of RATE-AF (Rate Control Therapy Evaluation in Permanent Atrial Fibrillation), in which 160 seniors (mean age, 76 years) with moderate or severe symptoms caused by permanent atrial fibrillation (AFib) as well as heart failure symptoms were randomized to low-dose digoxin or the beta-blocker bisoprolol for rate control.
The open-label trial was designed to address a centuries-old unmet need: “Although digoxin has been in use since 1785, we have no longer-term clinical trials of digoxin in patients with AFib or AFib with heart failure,” noted Dr. Kotecha, professor of cardiology at the University of Birmingham (England).
Not only is digoxin greatly understudied in AFib, but permanent AFib – the most common form of the arrhythmia – has received only a tiny fraction of the research attention that’s been devoted to paroxysmal or persistent AFib, he added.
In RATE-AF, digoxin and bisoprolol proved similarly effective at reducing heart rate, from about 100 bpm at baseline to the mid-70s at 6 and 12 months. Notably, only a handful of study participants required an additional rate control drug during the 12-month study. Nor did the two drugs differ in terms of their impact on patient-reported quality of life at 6 months as reflected by their Short Form–36 physical component score, the primary study endpoint. And both drugs were well tolerated, with 96% of patients in the digoxin group still on the drug at a mean of 161 mcg/day at 6 months, and 89% still on their beta-blocker.
But that’s pretty much where the similarities in outcomes ended.
For example, at 12 months, the digoxin group scored significantly higher than the beta-blocker group on several domains of the Short Form–36 physical component score, including vitality, physical function, and global health. More than half of the digoxin group had a two-class improvement in modified European Heart Rhythm Association AFib-related symptoms at 6 months, compared with 10% of the beta-blocker group. At 12 months, nearly 70% of the digoxin group had a two-class improvement, as did 30% on bisoprolol.
Heart failure symptoms in the digoxin group improved from a mean baseline New York Heart Association class of 2.4 to 1.5 at both 6 and 12 months; the improvement was more modest in the beta-blocker group, going from NYHA 2.4 at baseline to 2.0 at both 6 and 12 months. And while N-terminal of the prohormone brain natriuretic peptide levels improved in the digoxin group from a baseline of 1,095 pg/mL to 1,058 at 6 months and 960 at 12 months, NT-proBNP actually went up in the beta-blocker group, from 1,041 to 1,209-1,250 pg/mL at 12 months.
Moreover, Dr. Kotecha continued, while RATE-AF was underpowered to assess clinical events, it’s nevertheless noteworthy that a total of 29 adverse events occurred in 12 months in the digoxin group, compared with 142 with beta-blocker therapy. There were 12 unplanned hospital admissions in the digoxin group and 28 in the beta-blocker group, and 22 primary care visits for either AFib or cardiovascular symptoms in patients on digoxin versus 64 in the beta-blocker group.
“Our results suggest a wider use of digoxin for stable patients with permanent AFib,” Dr. Kotecha concluded. However, in an interview, Jonathan Piccini, MD, had a different take on the study results.
“I don’t think this study should widely impact clinical practice in the U.S.,” according to Dr. Piccini, director of cardiac electrophysiology at Duke University, Durham, N.C.
His reservations included RATE-AF’s modest sample size as well as uncertainty as to the trial’s generalizability, given that bisoprolol isn’t much used in the United States. Also, these were elderly patients with shortness of breath, and it’s unclear how effective digoxin would be for rate control in patients with permanent AFib who are more active.
“The classic teaching is that digoxin is great for rate control at rest, but when people are active it’s not nearly as good as beta-blockers or calcium-channel blockers,” the cardiologist said.
“A beta-blocker is still going to be my first-line rate control agent. But the results of RATE-AF do open my mind that for an older sedentary patient I may very well think twice now about using digoxin, because in that situation it looks like it achieves similar goals as a beta-blocker,” Dr. Piccini added.
On the plus side for RATE-AF: “I am very pleased to see that we have a randomized controlled trial focused on rate control in permanent AFib. It also tickles me pink to see a randomized, controlled study of digoxin. And I’m really excited to see a clinical trial that focuses on quality of life. It should give some confidence to know that from a quality of life perspective clinicians can consider using either digoxin or a beta-blocker for rate control,” he said.
American College of Cardiology vice president Dipti Itchhaporia, MD, said she’d need to see a much larger randomized trial including a calcium-channel blocker as a third-rate control arm before she’d consider digoxin as first-line rate control therapy in patients with AFib with or without heart failure. Also, she has reservations about drawing definitive conclusions from an open, unblinded study in which patient-reported outcomes are the primary endpoint.
“I think these were surprising findings given what we all think about digoxin in this country. In general, digoxin fell out of favor for rate control, mainly because of observational studies showing increased mortality. So most of us choose a beta-blocker,” she observed in an interview.
But of course, a randomized trial, even a 160-patient randomized trial, constitutes a higher level of evidence.
“I don’t think I’m going to convert tomorrow and make digoxin my first-line rate control therapy without more data. But RATE-AF does makes me stop and think about using it more than I did before in some of my permanent AFib patients,” said Dr. Itchhaporia, director of disease management at Hoag Memorial Hospital in Newport Beach, Calif.
Dr. Kotecha reported having no financial conflicts regarding the study, which was funded by the U.K. National Institute for Health Research, the British Heart Foundation, and the European Union.
REPORTING FROM ESC CONGRESS 2020
COVID-19 at home: What does optimal care look like?
Marilyn Stebbins, PharmD, fell ill at the end of February 2020. Initially diagnosed with multifocal pneumonia and treated with antibiotics, she later developed severe gastrointestinal symptoms, fatigue, and shortness of breath. She was hospitalized in early March and was diagnosed with COVID-19.
It was still early in the pandemic, and testing was not available for her husband. After she was discharged, her husband isolated himself as much as possible. But that limited the amount of care he could offer.
“When I came home after 8 days in the ICU, I felt completely alone and terrified of not being able to care for myself and not knowing how much care my husband could provide,” said Dr. Stebbins, professor of clinical pharmacy at the University of California, San Francisco.
“I can’t even imagine what it would have been like if I had been home alone without my husband in the house,” she said. “I think about the people who died at home and understand how that might happen.”
Dr. Stebbins is one of tens of thousands of people who, whether hospitalized and discharged or never admitted for inpatient care, needed to find ways to convalesce at home. Data from the Centers for Medicare & Medicaid Services show that, of 326,674 beneficiaries who tested positive for COVID-19 between May 16 and June 11, 2020, 109,607 were hospitalized, suggesting that two-thirds were outpatients.
Most attention has focused on the sickest patients, leaving less severe cases to fall through the cracks. Despite fever, cough, difficulty breathing, and a surfeit of other symptoms, there are few available resources and all too little support to help patients navigate the physical and emotional struggles of contending with COVID-19 at home.
No ‘cookie-cutter’ approach
The speed with which the pandemic progressed caught public health systems off guard, but now, “it is essential to put into place the infrastructure to care for the physical and mental health needs of patients at home because most are in the community and many, if not most, still aren’t receiving sufficient support at home,” said Dr. Stebbins.
said Gary LeRoy, MD, a family physician in Dayton, Ohio. He emphasized that there is “no cookie-cutter formula” for home care, because every patient’s situation is different.
“I begin by having a detailed conversation with each patient to ascertain whether their home environment is safe and to paint a picture of their circumstances,” Dr. LeRoy, who is the president of the American Academy of Family Physicians, said in an interview.
Dr. LeRoy suggested questions that constitute “not just a ‘medical’ checklist but a ‘whole life’ checklist.”
- Do you have access to food, water, medications, sanitation/cleaning supplies, a thermometer, and other necessities? If not, who might assist in providing those?
- Do you need help with activities of daily living and self-care?
- Who else lives in your household? Do they have signs and symptoms of the virus? Have they been tested?
- Do you have enough physical space between you and other household members?
- Do you have children? How are they being cared for?
- What type of work do you do? What are the implications for your employment if you are unable to work for an extended period?
- Do you have an emotional, social, and spiritual support system (e.g., family, friends, community, church)?
- Do you have concerns I haven’t mentioned?
Patients’ responses will inform the management plan and determine what medical and social resources are needed, he said.
Daily check-in
Dr. Stebbins said the nurse case manager from her insurance company called her daily after she came home from the hospital. She was told that a public health nurse would also call, but no one from the health department called for days – a situation she hopes has improved.
One way or another, she said, “health care providers [or their staff] should check in with patients daily, either telephonically or via video.” She noted that video is superior, because “someone who isn’t a family member needs to put eyes on a patient and might be able to detect warning signs that a family member without healthcare training might not notice.”
Dr. LeRoy, who is also an associate professor of medicine at Wright State University, Dayton, Ohio, said that, given his time constraints, a nurse or medical assistant in his practice conducts the daily check-ins and notifies him if the patient has fever or other symptoms.
“Under ordinary circumstances, when a patient comes to see me for some type of medical condition, I get to meet the patient, consider what might be going on, then order a test, wait for the results, and suggest a treatment plan. But these are anything but ordinary circumstances,” said Matthew Exline, MD, a pulmonary and critical care specialist at the Ohio State University Wexner Medical Center, Columbus.
“That traditional structure broke down with COVID-19, when we may have test results without even seeing the patient. And without this interaction, it is harder to know as a physician what course of action to take,” he said in an interview.
Once a diagnosis has been made, the physician has at least some data to help guide next steps, even if there has been no prior meeting with the patient.
For example, a positive test raises a host of issues, not the least of which is the risk of spreading the infection to other household members and questions about whether to go the hospital. Moreover, for patients, positive tests can have serious ramifications.
“Severe shortness of breath at rest is not typical of the flu, nor is loss of taste or smell,” said Dr. Exline. Practitioners must educate patients and families about specific symptoms of COVID-19, including shortness of breath, loss of taste or smell, and gastrointestinal or neurologic symptoms, and when to seek emergency care.
Dr. LeRoy suggests buying a pulse oximeter to gauge blood oxygen levels and pulse rate. Together with a thermometer, a portable blood pressure monitor, and, if indicated, a blood glucose monitor, these devices provide a comprehensive and accurate assessment of vital signs.
Dr. LeRoy also educates patients and their families about when to seek medical attention.
Dr. Stebbins takes a similar approach. “Family members are part of, not apart from, the care of patients with COVID-19, and it’s our responsibility as healthcare providers to consider them in the patient’s care plan.”
Keeping family safe
Beyond care, family members need a plan to keep themselves healthy, too.
“A patient with COVID-19 at home should self-quarantine as much as possible to keep other family members safe, if they continue to live in the same house,” Dr. Exline said.
Ideally, uninfected family members should stay with relatives or friends. When that’s not possible, everyone in the household should wear a mask, be vigilant about hand washing, and wipe down all surfaces – including doorknobs, light switches, faucet handles, cellphones, and utensils – regularly with bleach or an alcohol solution.
Caregivers should also minimize the amount of time they are exposed to the patient.
“Set food, water, and medication on the night table and leave the room rather than spending hours at the bedside, since limiting exposure to viral load reduces the chances of contagion,” said Dr. Exline.
The Centers for Disease Control and Prevention offers guidance for household members caring for COVID-19 patients at home. It provides tips on how to help patients follow the doctor’s instructions and ways to ensure adequate hydration and rest, among others.
Patients with COVID-19 who live alone face more formidable challenges.
Dr. LeRoy says physicians can help patients by educating themselves about available social services in their community so they can provide appropriate referrals and connections. Such initiatives can include meal programs, friendly visit and financial assistance programs, as well as childcare and home health agencies.
He noted that Aunt Bertha, a social care network, provides a guide to social services throughout the United States. Additional resources are available on USA.gov.
Comfort and support
Patients with COVID-19 need to be as comfortable and as supported as possible, both physically and emotionally.
“While I was sick, my dogs curled up next to me and didn’t leave my side, and they were my saving grace. There’s not enough to be said about emotional support,” Dr. Stebbins said.
Although important, emotional support is not enough. For patients with respiratory disorders, such as chronic obstructive pulmonary disease, asthma, heart failure, or pneumonia, their subjective symptoms of shortness of breath, air hunger, or cough may improve with supplemental oxygen at home. Other measures include repositioning of the patient to lessen the body weight over the lungs or the use of lung percussion, Leroy said.
He added that improvement may also come from drainage of sputum from the airway passages, the use of agents to liquefy thick sputum (mucolytics), or aerosolized bronchodilator medications.
However, Dr. LeRoy cautioned, “one remedy does not work for everyone – an individual can improve gradually by using these home support interventions, or their respiratory status can deteriorate rapidly despite all these interventions.”
For this reason, he says patients should consult their personal physician to determine which, if any, of these home treatments would be best for their particular situation.
Patients who need emotional support, psychotherapy, or psychotropic medications may find teletherapy helpful. Guidance for psychiatrists, psychologists, and social workers regarding the treatment of COVID-19 patients via teletherapy can be found on the American Psychiatric Association, the American Psychological Association, and the National Association of Social Workers websites.
Pharmacists can also help ensure patient safety, Dr. Stebbins said.
If a patient has not picked up their usual medications, Dr. Stebbins said, “they may need a check-in call. Some may be ill and alone and may need encouragement to seek medical attention, and some may have no means of getting to the pharmacy and may need medications delivered.”
A home healthcare agency may also be helpful for homebound patients. David Bersson, director of operations at Synergy Home Care of Bergen County, N.J., has arranged in-home caregivers for patients with COVID-19.
The amount of care that professional caregivers provide can range from several hours per week to full-time, depending on the patient’s needs and budget, and can include companionship, Mr. Bersson said in an interview.
Because patient and caregiver safety are paramount, caregivers are thoroughly trained in protection and decontamination procedures and are regularly tested for COVID-19 prior to being sent into a client’s home.
Health insurance companies do not cover this service, Mr. Bersson noted, but the VetAssist program covers home care for veterans and their spouses who meet income requirements.
Caregiving and companionship are both vital pieces of the at-home care puzzle. “It was the virtual emotional support I got from friends, family, coworkers, and healthcare professionals that meant so much to me, and I know they played an important part in my recovery,” Dr. Stebbins said.
Dr. LeRoy agreed, noting that he calls patients, even if they only have mild symptoms and his nurse has already spoken to them. “The call doesn’t take much time – maybe just a 5-minute conversation – but it makes patients aware that I care.”
Dr. Stebbins, Dr. Exline, and Dr. LeRoy report no relevant financial relationships. Mr. Bersson is the director of operations at Synergy Home Care of Bergen County, New Jersey.
This story first appeared on Medscape.com.
Marilyn Stebbins, PharmD, fell ill at the end of February 2020. Initially diagnosed with multifocal pneumonia and treated with antibiotics, she later developed severe gastrointestinal symptoms, fatigue, and shortness of breath. She was hospitalized in early March and was diagnosed with COVID-19.
It was still early in the pandemic, and testing was not available for her husband. After she was discharged, her husband isolated himself as much as possible. But that limited the amount of care he could offer.
“When I came home after 8 days in the ICU, I felt completely alone and terrified of not being able to care for myself and not knowing how much care my husband could provide,” said Dr. Stebbins, professor of clinical pharmacy at the University of California, San Francisco.
“I can’t even imagine what it would have been like if I had been home alone without my husband in the house,” she said. “I think about the people who died at home and understand how that might happen.”
Dr. Stebbins is one of tens of thousands of people who, whether hospitalized and discharged or never admitted for inpatient care, needed to find ways to convalesce at home. Data from the Centers for Medicare & Medicaid Services show that, of 326,674 beneficiaries who tested positive for COVID-19 between May 16 and June 11, 2020, 109,607 were hospitalized, suggesting that two-thirds were outpatients.
Most attention has focused on the sickest patients, leaving less severe cases to fall through the cracks. Despite fever, cough, difficulty breathing, and a surfeit of other symptoms, there are few available resources and all too little support to help patients navigate the physical and emotional struggles of contending with COVID-19 at home.
No ‘cookie-cutter’ approach
The speed with which the pandemic progressed caught public health systems off guard, but now, “it is essential to put into place the infrastructure to care for the physical and mental health needs of patients at home because most are in the community and many, if not most, still aren’t receiving sufficient support at home,” said Dr. Stebbins.
said Gary LeRoy, MD, a family physician in Dayton, Ohio. He emphasized that there is “no cookie-cutter formula” for home care, because every patient’s situation is different.
“I begin by having a detailed conversation with each patient to ascertain whether their home environment is safe and to paint a picture of their circumstances,” Dr. LeRoy, who is the president of the American Academy of Family Physicians, said in an interview.
Dr. LeRoy suggested questions that constitute “not just a ‘medical’ checklist but a ‘whole life’ checklist.”
- Do you have access to food, water, medications, sanitation/cleaning supplies, a thermometer, and other necessities? If not, who might assist in providing those?
- Do you need help with activities of daily living and self-care?
- Who else lives in your household? Do they have signs and symptoms of the virus? Have they been tested?
- Do you have enough physical space between you and other household members?
- Do you have children? How are they being cared for?
- What type of work do you do? What are the implications for your employment if you are unable to work for an extended period?
- Do you have an emotional, social, and spiritual support system (e.g., family, friends, community, church)?
- Do you have concerns I haven’t mentioned?
Patients’ responses will inform the management plan and determine what medical and social resources are needed, he said.
Daily check-in
Dr. Stebbins said the nurse case manager from her insurance company called her daily after she came home from the hospital. She was told that a public health nurse would also call, but no one from the health department called for days – a situation she hopes has improved.
One way or another, she said, “health care providers [or their staff] should check in with patients daily, either telephonically or via video.” She noted that video is superior, because “someone who isn’t a family member needs to put eyes on a patient and might be able to detect warning signs that a family member without healthcare training might not notice.”
Dr. LeRoy, who is also an associate professor of medicine at Wright State University, Dayton, Ohio, said that, given his time constraints, a nurse or medical assistant in his practice conducts the daily check-ins and notifies him if the patient has fever or other symptoms.
“Under ordinary circumstances, when a patient comes to see me for some type of medical condition, I get to meet the patient, consider what might be going on, then order a test, wait for the results, and suggest a treatment plan. But these are anything but ordinary circumstances,” said Matthew Exline, MD, a pulmonary and critical care specialist at the Ohio State University Wexner Medical Center, Columbus.
“That traditional structure broke down with COVID-19, when we may have test results without even seeing the patient. And without this interaction, it is harder to know as a physician what course of action to take,” he said in an interview.
Once a diagnosis has been made, the physician has at least some data to help guide next steps, even if there has been no prior meeting with the patient.
For example, a positive test raises a host of issues, not the least of which is the risk of spreading the infection to other household members and questions about whether to go the hospital. Moreover, for patients, positive tests can have serious ramifications.
“Severe shortness of breath at rest is not typical of the flu, nor is loss of taste or smell,” said Dr. Exline. Practitioners must educate patients and families about specific symptoms of COVID-19, including shortness of breath, loss of taste or smell, and gastrointestinal or neurologic symptoms, and when to seek emergency care.
Dr. LeRoy suggests buying a pulse oximeter to gauge blood oxygen levels and pulse rate. Together with a thermometer, a portable blood pressure monitor, and, if indicated, a blood glucose monitor, these devices provide a comprehensive and accurate assessment of vital signs.
Dr. LeRoy also educates patients and their families about when to seek medical attention.
Dr. Stebbins takes a similar approach. “Family members are part of, not apart from, the care of patients with COVID-19, and it’s our responsibility as healthcare providers to consider them in the patient’s care plan.”
Keeping family safe
Beyond care, family members need a plan to keep themselves healthy, too.
“A patient with COVID-19 at home should self-quarantine as much as possible to keep other family members safe, if they continue to live in the same house,” Dr. Exline said.
Ideally, uninfected family members should stay with relatives or friends. When that’s not possible, everyone in the household should wear a mask, be vigilant about hand washing, and wipe down all surfaces – including doorknobs, light switches, faucet handles, cellphones, and utensils – regularly with bleach or an alcohol solution.
Caregivers should also minimize the amount of time they are exposed to the patient.
“Set food, water, and medication on the night table and leave the room rather than spending hours at the bedside, since limiting exposure to viral load reduces the chances of contagion,” said Dr. Exline.
The Centers for Disease Control and Prevention offers guidance for household members caring for COVID-19 patients at home. It provides tips on how to help patients follow the doctor’s instructions and ways to ensure adequate hydration and rest, among others.
Patients with COVID-19 who live alone face more formidable challenges.
Dr. LeRoy says physicians can help patients by educating themselves about available social services in their community so they can provide appropriate referrals and connections. Such initiatives can include meal programs, friendly visit and financial assistance programs, as well as childcare and home health agencies.
He noted that Aunt Bertha, a social care network, provides a guide to social services throughout the United States. Additional resources are available on USA.gov.
Comfort and support
Patients with COVID-19 need to be as comfortable and as supported as possible, both physically and emotionally.
“While I was sick, my dogs curled up next to me and didn’t leave my side, and they were my saving grace. There’s not enough to be said about emotional support,” Dr. Stebbins said.
Although important, emotional support is not enough. For patients with respiratory disorders, such as chronic obstructive pulmonary disease, asthma, heart failure, or pneumonia, their subjective symptoms of shortness of breath, air hunger, or cough may improve with supplemental oxygen at home. Other measures include repositioning of the patient to lessen the body weight over the lungs or the use of lung percussion, Leroy said.
He added that improvement may also come from drainage of sputum from the airway passages, the use of agents to liquefy thick sputum (mucolytics), or aerosolized bronchodilator medications.
However, Dr. LeRoy cautioned, “one remedy does not work for everyone – an individual can improve gradually by using these home support interventions, or their respiratory status can deteriorate rapidly despite all these interventions.”
For this reason, he says patients should consult their personal physician to determine which, if any, of these home treatments would be best for their particular situation.
Patients who need emotional support, psychotherapy, or psychotropic medications may find teletherapy helpful. Guidance for psychiatrists, psychologists, and social workers regarding the treatment of COVID-19 patients via teletherapy can be found on the American Psychiatric Association, the American Psychological Association, and the National Association of Social Workers websites.
Pharmacists can also help ensure patient safety, Dr. Stebbins said.
If a patient has not picked up their usual medications, Dr. Stebbins said, “they may need a check-in call. Some may be ill and alone and may need encouragement to seek medical attention, and some may have no means of getting to the pharmacy and may need medications delivered.”
A home healthcare agency may also be helpful for homebound patients. David Bersson, director of operations at Synergy Home Care of Bergen County, N.J., has arranged in-home caregivers for patients with COVID-19.
The amount of care that professional caregivers provide can range from several hours per week to full-time, depending on the patient’s needs and budget, and can include companionship, Mr. Bersson said in an interview.
Because patient and caregiver safety are paramount, caregivers are thoroughly trained in protection and decontamination procedures and are regularly tested for COVID-19 prior to being sent into a client’s home.
Health insurance companies do not cover this service, Mr. Bersson noted, but the VetAssist program covers home care for veterans and their spouses who meet income requirements.
Caregiving and companionship are both vital pieces of the at-home care puzzle. “It was the virtual emotional support I got from friends, family, coworkers, and healthcare professionals that meant so much to me, and I know they played an important part in my recovery,” Dr. Stebbins said.
Dr. LeRoy agreed, noting that he calls patients, even if they only have mild symptoms and his nurse has already spoken to them. “The call doesn’t take much time – maybe just a 5-minute conversation – but it makes patients aware that I care.”
Dr. Stebbins, Dr. Exline, and Dr. LeRoy report no relevant financial relationships. Mr. Bersson is the director of operations at Synergy Home Care of Bergen County, New Jersey.
This story first appeared on Medscape.com.
Marilyn Stebbins, PharmD, fell ill at the end of February 2020. Initially diagnosed with multifocal pneumonia and treated with antibiotics, she later developed severe gastrointestinal symptoms, fatigue, and shortness of breath. She was hospitalized in early March and was diagnosed with COVID-19.
It was still early in the pandemic, and testing was not available for her husband. After she was discharged, her husband isolated himself as much as possible. But that limited the amount of care he could offer.
“When I came home after 8 days in the ICU, I felt completely alone and terrified of not being able to care for myself and not knowing how much care my husband could provide,” said Dr. Stebbins, professor of clinical pharmacy at the University of California, San Francisco.
“I can’t even imagine what it would have been like if I had been home alone without my husband in the house,” she said. “I think about the people who died at home and understand how that might happen.”
Dr. Stebbins is one of tens of thousands of people who, whether hospitalized and discharged or never admitted for inpatient care, needed to find ways to convalesce at home. Data from the Centers for Medicare & Medicaid Services show that, of 326,674 beneficiaries who tested positive for COVID-19 between May 16 and June 11, 2020, 109,607 were hospitalized, suggesting that two-thirds were outpatients.
Most attention has focused on the sickest patients, leaving less severe cases to fall through the cracks. Despite fever, cough, difficulty breathing, and a surfeit of other symptoms, there are few available resources and all too little support to help patients navigate the physical and emotional struggles of contending with COVID-19 at home.
No ‘cookie-cutter’ approach
The speed with which the pandemic progressed caught public health systems off guard, but now, “it is essential to put into place the infrastructure to care for the physical and mental health needs of patients at home because most are in the community and many, if not most, still aren’t receiving sufficient support at home,” said Dr. Stebbins.
said Gary LeRoy, MD, a family physician in Dayton, Ohio. He emphasized that there is “no cookie-cutter formula” for home care, because every patient’s situation is different.
“I begin by having a detailed conversation with each patient to ascertain whether their home environment is safe and to paint a picture of their circumstances,” Dr. LeRoy, who is the president of the American Academy of Family Physicians, said in an interview.
Dr. LeRoy suggested questions that constitute “not just a ‘medical’ checklist but a ‘whole life’ checklist.”
- Do you have access to food, water, medications, sanitation/cleaning supplies, a thermometer, and other necessities? If not, who might assist in providing those?
- Do you need help with activities of daily living and self-care?
- Who else lives in your household? Do they have signs and symptoms of the virus? Have they been tested?
- Do you have enough physical space between you and other household members?
- Do you have children? How are they being cared for?
- What type of work do you do? What are the implications for your employment if you are unable to work for an extended period?
- Do you have an emotional, social, and spiritual support system (e.g., family, friends, community, church)?
- Do you have concerns I haven’t mentioned?
Patients’ responses will inform the management plan and determine what medical and social resources are needed, he said.
Daily check-in
Dr. Stebbins said the nurse case manager from her insurance company called her daily after she came home from the hospital. She was told that a public health nurse would also call, but no one from the health department called for days – a situation she hopes has improved.
One way or another, she said, “health care providers [or their staff] should check in with patients daily, either telephonically or via video.” She noted that video is superior, because “someone who isn’t a family member needs to put eyes on a patient and might be able to detect warning signs that a family member without healthcare training might not notice.”
Dr. LeRoy, who is also an associate professor of medicine at Wright State University, Dayton, Ohio, said that, given his time constraints, a nurse or medical assistant in his practice conducts the daily check-ins and notifies him if the patient has fever or other symptoms.
“Under ordinary circumstances, when a patient comes to see me for some type of medical condition, I get to meet the patient, consider what might be going on, then order a test, wait for the results, and suggest a treatment plan. But these are anything but ordinary circumstances,” said Matthew Exline, MD, a pulmonary and critical care specialist at the Ohio State University Wexner Medical Center, Columbus.
“That traditional structure broke down with COVID-19, when we may have test results without even seeing the patient. And without this interaction, it is harder to know as a physician what course of action to take,” he said in an interview.
Once a diagnosis has been made, the physician has at least some data to help guide next steps, even if there has been no prior meeting with the patient.
For example, a positive test raises a host of issues, not the least of which is the risk of spreading the infection to other household members and questions about whether to go the hospital. Moreover, for patients, positive tests can have serious ramifications.
“Severe shortness of breath at rest is not typical of the flu, nor is loss of taste or smell,” said Dr. Exline. Practitioners must educate patients and families about specific symptoms of COVID-19, including shortness of breath, loss of taste or smell, and gastrointestinal or neurologic symptoms, and when to seek emergency care.
Dr. LeRoy suggests buying a pulse oximeter to gauge blood oxygen levels and pulse rate. Together with a thermometer, a portable blood pressure monitor, and, if indicated, a blood glucose monitor, these devices provide a comprehensive and accurate assessment of vital signs.
Dr. LeRoy also educates patients and their families about when to seek medical attention.
Dr. Stebbins takes a similar approach. “Family members are part of, not apart from, the care of patients with COVID-19, and it’s our responsibility as healthcare providers to consider them in the patient’s care plan.”
Keeping family safe
Beyond care, family members need a plan to keep themselves healthy, too.
“A patient with COVID-19 at home should self-quarantine as much as possible to keep other family members safe, if they continue to live in the same house,” Dr. Exline said.
Ideally, uninfected family members should stay with relatives or friends. When that’s not possible, everyone in the household should wear a mask, be vigilant about hand washing, and wipe down all surfaces – including doorknobs, light switches, faucet handles, cellphones, and utensils – regularly with bleach or an alcohol solution.
Caregivers should also minimize the amount of time they are exposed to the patient.
“Set food, water, and medication on the night table and leave the room rather than spending hours at the bedside, since limiting exposure to viral load reduces the chances of contagion,” said Dr. Exline.
The Centers for Disease Control and Prevention offers guidance for household members caring for COVID-19 patients at home. It provides tips on how to help patients follow the doctor’s instructions and ways to ensure adequate hydration and rest, among others.
Patients with COVID-19 who live alone face more formidable challenges.
Dr. LeRoy says physicians can help patients by educating themselves about available social services in their community so they can provide appropriate referrals and connections. Such initiatives can include meal programs, friendly visit and financial assistance programs, as well as childcare and home health agencies.
He noted that Aunt Bertha, a social care network, provides a guide to social services throughout the United States. Additional resources are available on USA.gov.
Comfort and support
Patients with COVID-19 need to be as comfortable and as supported as possible, both physically and emotionally.
“While I was sick, my dogs curled up next to me and didn’t leave my side, and they were my saving grace. There’s not enough to be said about emotional support,” Dr. Stebbins said.
Although important, emotional support is not enough. For patients with respiratory disorders, such as chronic obstructive pulmonary disease, asthma, heart failure, or pneumonia, their subjective symptoms of shortness of breath, air hunger, or cough may improve with supplemental oxygen at home. Other measures include repositioning of the patient to lessen the body weight over the lungs or the use of lung percussion, Leroy said.
He added that improvement may also come from drainage of sputum from the airway passages, the use of agents to liquefy thick sputum (mucolytics), or aerosolized bronchodilator medications.
However, Dr. LeRoy cautioned, “one remedy does not work for everyone – an individual can improve gradually by using these home support interventions, or their respiratory status can deteriorate rapidly despite all these interventions.”
For this reason, he says patients should consult their personal physician to determine which, if any, of these home treatments would be best for their particular situation.
Patients who need emotional support, psychotherapy, or psychotropic medications may find teletherapy helpful. Guidance for psychiatrists, psychologists, and social workers regarding the treatment of COVID-19 patients via teletherapy can be found on the American Psychiatric Association, the American Psychological Association, and the National Association of Social Workers websites.
Pharmacists can also help ensure patient safety, Dr. Stebbins said.
If a patient has not picked up their usual medications, Dr. Stebbins said, “they may need a check-in call. Some may be ill and alone and may need encouragement to seek medical attention, and some may have no means of getting to the pharmacy and may need medications delivered.”
A home healthcare agency may also be helpful for homebound patients. David Bersson, director of operations at Synergy Home Care of Bergen County, N.J., has arranged in-home caregivers for patients with COVID-19.
The amount of care that professional caregivers provide can range from several hours per week to full-time, depending on the patient’s needs and budget, and can include companionship, Mr. Bersson said in an interview.
Because patient and caregiver safety are paramount, caregivers are thoroughly trained in protection and decontamination procedures and are regularly tested for COVID-19 prior to being sent into a client’s home.
Health insurance companies do not cover this service, Mr. Bersson noted, but the VetAssist program covers home care for veterans and their spouses who meet income requirements.
Caregiving and companionship are both vital pieces of the at-home care puzzle. “It was the virtual emotional support I got from friends, family, coworkers, and healthcare professionals that meant so much to me, and I know they played an important part in my recovery,” Dr. Stebbins said.
Dr. LeRoy agreed, noting that he calls patients, even if they only have mild symptoms and his nurse has already spoken to them. “The call doesn’t take much time – maybe just a 5-minute conversation – but it makes patients aware that I care.”
Dr. Stebbins, Dr. Exline, and Dr. LeRoy report no relevant financial relationships. Mr. Bersson is the director of operations at Synergy Home Care of Bergen County, New Jersey.
This story first appeared on Medscape.com.
New ESC/EACTS guideline on atrial fibrillation
New atrial fibrillation (AFib) management guidelines from the European Society of Cardiology (ESC) call for diagnostic confirmation and structured characterization of AFib and the need to streamline integrated care with the Atrial fibrillation Better Care (ABC) pathway.
“It’s as simple as CC to ABC,” quipped one task force member during the virtual unveiling of the guidelines at the ESC Congress 2020.
The guidelines were developed in collaboration with the European Association of Cardio-Thoracic Surgery (EACTS) and published simultaneously August 29 in the European Heart Journal.
Acknowledging the slew of novel screening tools now available and their reported sensitivity and specificity rates, the document supports opportunistic screening for AFib by pulse taking or electrocardiogram (ECG) rhythm strip in patients at least 65 years of age, with a class 1 recommendation, evidence level B.
Systematic ECG screening should also be considered to detect AFib in individuals at least 75 years of age or in those at high risk for stroke (class IIa, level B).
Other new class I screening recommendations are to inform individuals undergoing screening about the significance and treatment implications of detecting AFib and to have a structured referral platform in place for further physician-led evaluation.
A definite diagnosis of clinical AFib is established only after confirmation by a conventional 12-lead ECG or single-lead ECG strip with at least 30 seconds of AFib.
In line with ESC’s 2016 AFib guidelines, the new iteration classifies AFib as first diagnosed, paroxysmal, persistent, long-standing persistent, and permanent. But it’s also important to classify the clinical profile of AFib, task force member Giuseppe Boriani, MD, PhD, University of Modena, Italy, said in the first of five presentations.
“So the novelty of the 2020 guidelines is related to the proposal of the 4S-AF scheme for a structured characterization of atrial fibrillation that takes into account Stroke risk, severity of Symptoms, Severity of atrial fibrillation burden, and Substrate severity,” he said.
This represents a paradigm shift from a single-domain conventional classification of AFib toward a structured characterization that streamlines assessment, informs treatment decision-making, and facilitates communication among physicians of various specialties, said Tatjana Potpara, MD, PhD, guideline co-chair and head of the Department for Intensive Arrhythmia Care, Clinical Centre of Serbia, Belgrade.
“The beauty of this approach is that, at present, the assessment of the ‘S’ components are performed using available tools, but in the future, the 4S-AF has a great potential to incorporate whatever becomes available for a more precision assessment of substrate or symptoms or arrhythmia burden and so forth,” she said.
ABC pathway
The guidelines advocate the previously described ABC pathway for integrated care management, which includes ‘A’ for Anticoagulation/Avoid stroke, ‘B’ for Better symptom control, and ‘C’ for Comorbidity/Cardiovascular risk factor optimization.
The document strengthens support for formal risk score–based assessment of bleeding risk in all patients, including use of the HAS-BLED score to help address modifiable bleeding risk factors and to identify patients at high bleeding risk (HAS-BLED score ≥3) for early and more frequent follow-up.
These assessments should be done regularly, given that both stroke and bleeding risk are dynamic and change over time with aging and comorbidities, Dr. Potpara stressed. In patients with AFib initially at low risk for stroke, the next assessment should be optimally performed at 4-6 months.
The guideline also targets weight loss in patients who are obese and have AFib, particularly those being evaluated for ablation, and good blood pressure control in patients with AFib and hypertension to reduce AFib recurrences and risk for stroke and bleeding (both class I, up from IIa).
It’s particularly important that these risk factors are addressed, and that modifiable risk factors that go along with increased AFib occurrence and persistence are addressed and communicated to patients, said Gerhard Hindricks, MD, PhD, guideline cochair and medical director of the Rhythmology Department, Heart Centre Leipzig (Germany).
“I have to confess, as an interventional electrophysiologist, there has been a time where I have not appreciated these risk factors intensely enough,” he said. “But we have learned, also in the field of catheter ablation, that weight loss is an essential basis for a good procedure. If we can motivate patients to lose weight and then come to the intervention with better outcome, it’s a true benefit for the patient and addresses patient values. So I’m particularly happy we have introduced that with such intensity in the guidelines.”
Rate and rhythm control
The guidelines make no recommendation of one novel oral anticoagulant (NOAC) over another. However, in patients already receiving vitamin K antagonists with low time in the therapeutic range, they recommend switching to a different NOAC but ensuring good adherence and persistence with therapy (class I recommendation) or efforts to improve time in therapeutic range (class IIa).
Catheter ablation takes on a more prominent role for rhythm control and is now recommended after one antiarrhythmic drug therapy fails to improve symptoms of AF recurrence in patients with paroxysmal AFib, or persistent AFib with or without major risk factors for recurrence. The class I recommendation is based on results from the CAPTAF and CABANA trials, said task force member Carina Blomström-Lundqvist, MD, PhD, Uppsala University, Sweden.
Catheter ablation is also now a first-line therapy for patients with AFib who have a high likelihood of tachycardia-induced cardiomyopathy, independent of symptom status. “In this subset of patients, catheter ablation may offer a lot with respect to restoration of left ventricular function,” observed Dr. Hindricks.
Complete electrical isolation of the pulmonary veins is recommended during all AFib catheter ablation procedures (class I).
“Even as a medical conservative, I think it is totally reasonable to move to catheter ablation after a failed drug trial,” commented John Mandrola, MD, Baptist Health, Louisville, Ky., who was not a part of the guideline development.
Although the chance of a second drug working after one failure is low, he noted that operators in the United States have dofetilide, which is not used much in Europe, and sometimes works surprisingly well.
“That said, the caveat is that moving to catheter ablation after drug failure is only appropriate if we have addressed all the pertinent risk factors: sleep apnea, weight loss, lack of fitness, blood pressure control, and alcohol excess,” he said.
As for tachycardia-mediated cardiomyopathy, this too can be reasonable, Dr. Mandrola said. “I often get people ‘out of a hole’ with amiodarone plus cardioversion for a few months and then proceed to ablation.”
Notably, the 2020 iteration sharpens its recommendation that amiodarone not be used first-line for long-term rhythm control in all patients with AFib, including those with heart failure with reduced ejection fraction, given its extracardiac toxicity (class I, up from IIa).
Quality counts
In response to growing evidence that guideline-adherence is associated with significantly better outcomes in AFib, the 2020 ESC/EACTS guidelines explicitly included a recommendation on the need to measure quality of care to identify opportunities for improvement.
With this framework in mind, a task force with 23 people – including members from ESC and heart rhythm societies in the United States, Asia Pacific, and Latin America, along with patient representatives – was created to develop a list of quality indicators (QIs), ultimately settling on 17 main QIs and 17 secondary ones, said Elena Arbelo, MD, PhD, MSc, University of Barcelona.
The QIs are classified into six domains: patient assessment, anticoagulation, rate control, rhythm control, risk factor modification, and, importantly, outcome measures. A full list is accessible in a paper, simultaneously published in EP EuroPace.
Five patient-reported outcomes fall under the outcomes domain but only one – health-related quality of life – is a main quality indicator. The remaining outcomes are still important but are listed as secondary because of the lack of evidence to sustain or defend their systematic implementation, particularly evidence on how to measure them appropriately, Dr. Arbelo said.
“Hopefully, following the [class I] recommendation by the 2020 ESC guidelines to routinely collect patient-reported outcomes will allow us to collect further evidence and in the future have sufficient evidence to include these as a main outcome,” she said.
The QI work was driven in parallel with the guidelines and had a huge impact on its development, including inclusion of clear recommendations on how to measure quality, Dr. Hindricks said. “I believe that the whole issue of quality management in the treatment of patients with a focus on patient values cannot be overestimated.”
Disclosure information for all writing committee members is in the report. Dr. Mandrola is a writer and podcaster for Medscape.
A version of this article originally appeared on Medscape.com.
New atrial fibrillation (AFib) management guidelines from the European Society of Cardiology (ESC) call for diagnostic confirmation and structured characterization of AFib and the need to streamline integrated care with the Atrial fibrillation Better Care (ABC) pathway.
“It’s as simple as CC to ABC,” quipped one task force member during the virtual unveiling of the guidelines at the ESC Congress 2020.
The guidelines were developed in collaboration with the European Association of Cardio-Thoracic Surgery (EACTS) and published simultaneously August 29 in the European Heart Journal.
Acknowledging the slew of novel screening tools now available and their reported sensitivity and specificity rates, the document supports opportunistic screening for AFib by pulse taking or electrocardiogram (ECG) rhythm strip in patients at least 65 years of age, with a class 1 recommendation, evidence level B.
Systematic ECG screening should also be considered to detect AFib in individuals at least 75 years of age or in those at high risk for stroke (class IIa, level B).
Other new class I screening recommendations are to inform individuals undergoing screening about the significance and treatment implications of detecting AFib and to have a structured referral platform in place for further physician-led evaluation.
A definite diagnosis of clinical AFib is established only after confirmation by a conventional 12-lead ECG or single-lead ECG strip with at least 30 seconds of AFib.
In line with ESC’s 2016 AFib guidelines, the new iteration classifies AFib as first diagnosed, paroxysmal, persistent, long-standing persistent, and permanent. But it’s also important to classify the clinical profile of AFib, task force member Giuseppe Boriani, MD, PhD, University of Modena, Italy, said in the first of five presentations.
“So the novelty of the 2020 guidelines is related to the proposal of the 4S-AF scheme for a structured characterization of atrial fibrillation that takes into account Stroke risk, severity of Symptoms, Severity of atrial fibrillation burden, and Substrate severity,” he said.
This represents a paradigm shift from a single-domain conventional classification of AFib toward a structured characterization that streamlines assessment, informs treatment decision-making, and facilitates communication among physicians of various specialties, said Tatjana Potpara, MD, PhD, guideline co-chair and head of the Department for Intensive Arrhythmia Care, Clinical Centre of Serbia, Belgrade.
“The beauty of this approach is that, at present, the assessment of the ‘S’ components are performed using available tools, but in the future, the 4S-AF has a great potential to incorporate whatever becomes available for a more precision assessment of substrate or symptoms or arrhythmia burden and so forth,” she said.
ABC pathway
The guidelines advocate the previously described ABC pathway for integrated care management, which includes ‘A’ for Anticoagulation/Avoid stroke, ‘B’ for Better symptom control, and ‘C’ for Comorbidity/Cardiovascular risk factor optimization.
The document strengthens support for formal risk score–based assessment of bleeding risk in all patients, including use of the HAS-BLED score to help address modifiable bleeding risk factors and to identify patients at high bleeding risk (HAS-BLED score ≥3) for early and more frequent follow-up.
These assessments should be done regularly, given that both stroke and bleeding risk are dynamic and change over time with aging and comorbidities, Dr. Potpara stressed. In patients with AFib initially at low risk for stroke, the next assessment should be optimally performed at 4-6 months.
The guideline also targets weight loss in patients who are obese and have AFib, particularly those being evaluated for ablation, and good blood pressure control in patients with AFib and hypertension to reduce AFib recurrences and risk for stroke and bleeding (both class I, up from IIa).
It’s particularly important that these risk factors are addressed, and that modifiable risk factors that go along with increased AFib occurrence and persistence are addressed and communicated to patients, said Gerhard Hindricks, MD, PhD, guideline cochair and medical director of the Rhythmology Department, Heart Centre Leipzig (Germany).
“I have to confess, as an interventional electrophysiologist, there has been a time where I have not appreciated these risk factors intensely enough,” he said. “But we have learned, also in the field of catheter ablation, that weight loss is an essential basis for a good procedure. If we can motivate patients to lose weight and then come to the intervention with better outcome, it’s a true benefit for the patient and addresses patient values. So I’m particularly happy we have introduced that with such intensity in the guidelines.”
Rate and rhythm control
The guidelines make no recommendation of one novel oral anticoagulant (NOAC) over another. However, in patients already receiving vitamin K antagonists with low time in the therapeutic range, they recommend switching to a different NOAC but ensuring good adherence and persistence with therapy (class I recommendation) or efforts to improve time in therapeutic range (class IIa).
Catheter ablation takes on a more prominent role for rhythm control and is now recommended after one antiarrhythmic drug therapy fails to improve symptoms of AF recurrence in patients with paroxysmal AFib, or persistent AFib with or without major risk factors for recurrence. The class I recommendation is based on results from the CAPTAF and CABANA trials, said task force member Carina Blomström-Lundqvist, MD, PhD, Uppsala University, Sweden.
Catheter ablation is also now a first-line therapy for patients with AFib who have a high likelihood of tachycardia-induced cardiomyopathy, independent of symptom status. “In this subset of patients, catheter ablation may offer a lot with respect to restoration of left ventricular function,” observed Dr. Hindricks.
Complete electrical isolation of the pulmonary veins is recommended during all AFib catheter ablation procedures (class I).
“Even as a medical conservative, I think it is totally reasonable to move to catheter ablation after a failed drug trial,” commented John Mandrola, MD, Baptist Health, Louisville, Ky., who was not a part of the guideline development.
Although the chance of a second drug working after one failure is low, he noted that operators in the United States have dofetilide, which is not used much in Europe, and sometimes works surprisingly well.
“That said, the caveat is that moving to catheter ablation after drug failure is only appropriate if we have addressed all the pertinent risk factors: sleep apnea, weight loss, lack of fitness, blood pressure control, and alcohol excess,” he said.
As for tachycardia-mediated cardiomyopathy, this too can be reasonable, Dr. Mandrola said. “I often get people ‘out of a hole’ with amiodarone plus cardioversion for a few months and then proceed to ablation.”
Notably, the 2020 iteration sharpens its recommendation that amiodarone not be used first-line for long-term rhythm control in all patients with AFib, including those with heart failure with reduced ejection fraction, given its extracardiac toxicity (class I, up from IIa).
Quality counts
In response to growing evidence that guideline-adherence is associated with significantly better outcomes in AFib, the 2020 ESC/EACTS guidelines explicitly included a recommendation on the need to measure quality of care to identify opportunities for improvement.
With this framework in mind, a task force with 23 people – including members from ESC and heart rhythm societies in the United States, Asia Pacific, and Latin America, along with patient representatives – was created to develop a list of quality indicators (QIs), ultimately settling on 17 main QIs and 17 secondary ones, said Elena Arbelo, MD, PhD, MSc, University of Barcelona.
The QIs are classified into six domains: patient assessment, anticoagulation, rate control, rhythm control, risk factor modification, and, importantly, outcome measures. A full list is accessible in a paper, simultaneously published in EP EuroPace.
Five patient-reported outcomes fall under the outcomes domain but only one – health-related quality of life – is a main quality indicator. The remaining outcomes are still important but are listed as secondary because of the lack of evidence to sustain or defend their systematic implementation, particularly evidence on how to measure them appropriately, Dr. Arbelo said.
“Hopefully, following the [class I] recommendation by the 2020 ESC guidelines to routinely collect patient-reported outcomes will allow us to collect further evidence and in the future have sufficient evidence to include these as a main outcome,” she said.
The QI work was driven in parallel with the guidelines and had a huge impact on its development, including inclusion of clear recommendations on how to measure quality, Dr. Hindricks said. “I believe that the whole issue of quality management in the treatment of patients with a focus on patient values cannot be overestimated.”
Disclosure information for all writing committee members is in the report. Dr. Mandrola is a writer and podcaster for Medscape.
A version of this article originally appeared on Medscape.com.
New atrial fibrillation (AFib) management guidelines from the European Society of Cardiology (ESC) call for diagnostic confirmation and structured characterization of AFib and the need to streamline integrated care with the Atrial fibrillation Better Care (ABC) pathway.
“It’s as simple as CC to ABC,” quipped one task force member during the virtual unveiling of the guidelines at the ESC Congress 2020.
The guidelines were developed in collaboration with the European Association of Cardio-Thoracic Surgery (EACTS) and published simultaneously August 29 in the European Heart Journal.
Acknowledging the slew of novel screening tools now available and their reported sensitivity and specificity rates, the document supports opportunistic screening for AFib by pulse taking or electrocardiogram (ECG) rhythm strip in patients at least 65 years of age, with a class 1 recommendation, evidence level B.
Systematic ECG screening should also be considered to detect AFib in individuals at least 75 years of age or in those at high risk for stroke (class IIa, level B).
Other new class I screening recommendations are to inform individuals undergoing screening about the significance and treatment implications of detecting AFib and to have a structured referral platform in place for further physician-led evaluation.
A definite diagnosis of clinical AFib is established only after confirmation by a conventional 12-lead ECG or single-lead ECG strip with at least 30 seconds of AFib.
In line with ESC’s 2016 AFib guidelines, the new iteration classifies AFib as first diagnosed, paroxysmal, persistent, long-standing persistent, and permanent. But it’s also important to classify the clinical profile of AFib, task force member Giuseppe Boriani, MD, PhD, University of Modena, Italy, said in the first of five presentations.
“So the novelty of the 2020 guidelines is related to the proposal of the 4S-AF scheme for a structured characterization of atrial fibrillation that takes into account Stroke risk, severity of Symptoms, Severity of atrial fibrillation burden, and Substrate severity,” he said.
This represents a paradigm shift from a single-domain conventional classification of AFib toward a structured characterization that streamlines assessment, informs treatment decision-making, and facilitates communication among physicians of various specialties, said Tatjana Potpara, MD, PhD, guideline co-chair and head of the Department for Intensive Arrhythmia Care, Clinical Centre of Serbia, Belgrade.
“The beauty of this approach is that, at present, the assessment of the ‘S’ components are performed using available tools, but in the future, the 4S-AF has a great potential to incorporate whatever becomes available for a more precision assessment of substrate or symptoms or arrhythmia burden and so forth,” she said.
ABC pathway
The guidelines advocate the previously described ABC pathway for integrated care management, which includes ‘A’ for Anticoagulation/Avoid stroke, ‘B’ for Better symptom control, and ‘C’ for Comorbidity/Cardiovascular risk factor optimization.
The document strengthens support for formal risk score–based assessment of bleeding risk in all patients, including use of the HAS-BLED score to help address modifiable bleeding risk factors and to identify patients at high bleeding risk (HAS-BLED score ≥3) for early and more frequent follow-up.
These assessments should be done regularly, given that both stroke and bleeding risk are dynamic and change over time with aging and comorbidities, Dr. Potpara stressed. In patients with AFib initially at low risk for stroke, the next assessment should be optimally performed at 4-6 months.
The guideline also targets weight loss in patients who are obese and have AFib, particularly those being evaluated for ablation, and good blood pressure control in patients with AFib and hypertension to reduce AFib recurrences and risk for stroke and bleeding (both class I, up from IIa).
It’s particularly important that these risk factors are addressed, and that modifiable risk factors that go along with increased AFib occurrence and persistence are addressed and communicated to patients, said Gerhard Hindricks, MD, PhD, guideline cochair and medical director of the Rhythmology Department, Heart Centre Leipzig (Germany).
“I have to confess, as an interventional electrophysiologist, there has been a time where I have not appreciated these risk factors intensely enough,” he said. “But we have learned, also in the field of catheter ablation, that weight loss is an essential basis for a good procedure. If we can motivate patients to lose weight and then come to the intervention with better outcome, it’s a true benefit for the patient and addresses patient values. So I’m particularly happy we have introduced that with such intensity in the guidelines.”
Rate and rhythm control
The guidelines make no recommendation of one novel oral anticoagulant (NOAC) over another. However, in patients already receiving vitamin K antagonists with low time in the therapeutic range, they recommend switching to a different NOAC but ensuring good adherence and persistence with therapy (class I recommendation) or efforts to improve time in therapeutic range (class IIa).
Catheter ablation takes on a more prominent role for rhythm control and is now recommended after one antiarrhythmic drug therapy fails to improve symptoms of AF recurrence in patients with paroxysmal AFib, or persistent AFib with or without major risk factors for recurrence. The class I recommendation is based on results from the CAPTAF and CABANA trials, said task force member Carina Blomström-Lundqvist, MD, PhD, Uppsala University, Sweden.
Catheter ablation is also now a first-line therapy for patients with AFib who have a high likelihood of tachycardia-induced cardiomyopathy, independent of symptom status. “In this subset of patients, catheter ablation may offer a lot with respect to restoration of left ventricular function,” observed Dr. Hindricks.
Complete electrical isolation of the pulmonary veins is recommended during all AFib catheter ablation procedures (class I).
“Even as a medical conservative, I think it is totally reasonable to move to catheter ablation after a failed drug trial,” commented John Mandrola, MD, Baptist Health, Louisville, Ky., who was not a part of the guideline development.
Although the chance of a second drug working after one failure is low, he noted that operators in the United States have dofetilide, which is not used much in Europe, and sometimes works surprisingly well.
“That said, the caveat is that moving to catheter ablation after drug failure is only appropriate if we have addressed all the pertinent risk factors: sleep apnea, weight loss, lack of fitness, blood pressure control, and alcohol excess,” he said.
As for tachycardia-mediated cardiomyopathy, this too can be reasonable, Dr. Mandrola said. “I often get people ‘out of a hole’ with amiodarone plus cardioversion for a few months and then proceed to ablation.”
Notably, the 2020 iteration sharpens its recommendation that amiodarone not be used first-line for long-term rhythm control in all patients with AFib, including those with heart failure with reduced ejection fraction, given its extracardiac toxicity (class I, up from IIa).
Quality counts
In response to growing evidence that guideline-adherence is associated with significantly better outcomes in AFib, the 2020 ESC/EACTS guidelines explicitly included a recommendation on the need to measure quality of care to identify opportunities for improvement.
With this framework in mind, a task force with 23 people – including members from ESC and heart rhythm societies in the United States, Asia Pacific, and Latin America, along with patient representatives – was created to develop a list of quality indicators (QIs), ultimately settling on 17 main QIs and 17 secondary ones, said Elena Arbelo, MD, PhD, MSc, University of Barcelona.
The QIs are classified into six domains: patient assessment, anticoagulation, rate control, rhythm control, risk factor modification, and, importantly, outcome measures. A full list is accessible in a paper, simultaneously published in EP EuroPace.
Five patient-reported outcomes fall under the outcomes domain but only one – health-related quality of life – is a main quality indicator. The remaining outcomes are still important but are listed as secondary because of the lack of evidence to sustain or defend their systematic implementation, particularly evidence on how to measure them appropriately, Dr. Arbelo said.
“Hopefully, following the [class I] recommendation by the 2020 ESC guidelines to routinely collect patient-reported outcomes will allow us to collect further evidence and in the future have sufficient evidence to include these as a main outcome,” she said.
The QI work was driven in parallel with the guidelines and had a huge impact on its development, including inclusion of clear recommendations on how to measure quality, Dr. Hindricks said. “I believe that the whole issue of quality management in the treatment of patients with a focus on patient values cannot be overestimated.”
Disclosure information for all writing committee members is in the report. Dr. Mandrola is a writer and podcaster for Medscape.
A version of this article originally appeared on Medscape.com.