User login
Cell-Free DNA Blood Test Shows Strong Performance in Detecting Early-Stage CRC
Cell-Free DNA Blood Test Shows Strong Performance in Detecting Early-Stage CRC
TOPLINE:
A novel, blood-based test developed using fragmentomic features of cell-free DNA (cfDNA) detects colorectal cancer (CRC) with a 90.4% sensitivity and shows consistent performance across stages and tumor locations.
METHODOLOGY:
- Researchers conducted a prospective case-control study to develop and validate a noninvasive cfDNA-based screening test for CRC.
- Adults aged 40-89 years with CRC or advanced adenomas were enrolled at a tertiary center in South Korea between 2021 and 2024.
- Blood samples were drawn after colonoscopy, but prior to treatment, in patients with CRC, advanced adenomas, and asymptomatic controls with normal colonoscopy results.
- A model was trained on fragmentonic features derived from whole genome sequencing of cfDNA from 1250 participants and validated for its diagnostic performance in the remaining 427 participants, including all with advanced adenomas.
- The primary endpoint was the sensitivity of the cfDNA test for detecting CRC. The area under the receiver operating characteristic curve (AUROC) was also calculated.
TAKEAWAY:
- The cfDNA test detected CRC with 90.4% sensitivity and an AUROC of 0.978.
- Sensitivity by CRC stage was 84.2% for stage I, 85.0% for stage II, 94.4% for stage III, 100% for stage IV.
- Advanced adenomas were detected with 58.3% sensitivity and an AUROC of 0.862.
- Among individuals with normal colonoscopy findings, the test was correctly negative 94.7% of the time.
- Diagnostic sensitivities were consistent between left- and right-sided CRC tumors, among participants aged < 60 years and ≥ 60 years, and across left- and right-sided advanced adenomas.
IN PRACTICE:
"This highlights the potential clinical utility of the test in identifying candidates for minimally invasive therapeutic approaches tool for CRC," the authors wrote. "Notably, the high sensitivity observed for early-stage CRC and the favorable sensitivity for [advanced adenoma] suggest that this cfDNA test may offer benefits not only in diagnosis but also in prognosis and ultimately in CRC prevention."
SOURCE:
This study was led by Seung Wook Hong, MD, Asan Medical Center in Seoul, South Korea. It was published online on November 19, 2025, in the American Journal of Gastroenterology.
LIMITATIONS:
The case-control design introduced spectrum bias by comparing clearly defined CRC and advanced adenomas cases with individuals who had normal colonoscopy results. The CRC prevalence of 17% to 18% was higher than that observed in true screening populations, limiting generalizability. The exclusively Korean cohort limited extrapolation to non-Asian populations.
DISCLOSURES:
The study received support from GC Genome, Yongin, South Korea. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
A novel, blood-based test developed using fragmentomic features of cell-free DNA (cfDNA) detects colorectal cancer (CRC) with a 90.4% sensitivity and shows consistent performance across stages and tumor locations.
METHODOLOGY:
- Researchers conducted a prospective case-control study to develop and validate a noninvasive cfDNA-based screening test for CRC.
- Adults aged 40-89 years with CRC or advanced adenomas were enrolled at a tertiary center in South Korea between 2021 and 2024.
- Blood samples were drawn after colonoscopy, but prior to treatment, in patients with CRC, advanced adenomas, and asymptomatic controls with normal colonoscopy results.
- A model was trained on fragmentonic features derived from whole genome sequencing of cfDNA from 1250 participants and validated for its diagnostic performance in the remaining 427 participants, including all with advanced adenomas.
- The primary endpoint was the sensitivity of the cfDNA test for detecting CRC. The area under the receiver operating characteristic curve (AUROC) was also calculated.
TAKEAWAY:
- The cfDNA test detected CRC with 90.4% sensitivity and an AUROC of 0.978.
- Sensitivity by CRC stage was 84.2% for stage I, 85.0% for stage II, 94.4% for stage III, 100% for stage IV.
- Advanced adenomas were detected with 58.3% sensitivity and an AUROC of 0.862.
- Among individuals with normal colonoscopy findings, the test was correctly negative 94.7% of the time.
- Diagnostic sensitivities were consistent between left- and right-sided CRC tumors, among participants aged < 60 years and ≥ 60 years, and across left- and right-sided advanced adenomas.
IN PRACTICE:
"This highlights the potential clinical utility of the test in identifying candidates for minimally invasive therapeutic approaches tool for CRC," the authors wrote. "Notably, the high sensitivity observed for early-stage CRC and the favorable sensitivity for [advanced adenoma] suggest that this cfDNA test may offer benefits not only in diagnosis but also in prognosis and ultimately in CRC prevention."
SOURCE:
This study was led by Seung Wook Hong, MD, Asan Medical Center in Seoul, South Korea. It was published online on November 19, 2025, in the American Journal of Gastroenterology.
LIMITATIONS:
The case-control design introduced spectrum bias by comparing clearly defined CRC and advanced adenomas cases with individuals who had normal colonoscopy results. The CRC prevalence of 17% to 18% was higher than that observed in true screening populations, limiting generalizability. The exclusively Korean cohort limited extrapolation to non-Asian populations.
DISCLOSURES:
The study received support from GC Genome, Yongin, South Korea. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
A novel, blood-based test developed using fragmentomic features of cell-free DNA (cfDNA) detects colorectal cancer (CRC) with a 90.4% sensitivity and shows consistent performance across stages and tumor locations.
METHODOLOGY:
- Researchers conducted a prospective case-control study to develop and validate a noninvasive cfDNA-based screening test for CRC.
- Adults aged 40-89 years with CRC or advanced adenomas were enrolled at a tertiary center in South Korea between 2021 and 2024.
- Blood samples were drawn after colonoscopy, but prior to treatment, in patients with CRC, advanced adenomas, and asymptomatic controls with normal colonoscopy results.
- A model was trained on fragmentonic features derived from whole genome sequencing of cfDNA from 1250 participants and validated for its diagnostic performance in the remaining 427 participants, including all with advanced adenomas.
- The primary endpoint was the sensitivity of the cfDNA test for detecting CRC. The area under the receiver operating characteristic curve (AUROC) was also calculated.
TAKEAWAY:
- The cfDNA test detected CRC with 90.4% sensitivity and an AUROC of 0.978.
- Sensitivity by CRC stage was 84.2% for stage I, 85.0% for stage II, 94.4% for stage III, 100% for stage IV.
- Advanced adenomas were detected with 58.3% sensitivity and an AUROC of 0.862.
- Among individuals with normal colonoscopy findings, the test was correctly negative 94.7% of the time.
- Diagnostic sensitivities were consistent between left- and right-sided CRC tumors, among participants aged < 60 years and ≥ 60 years, and across left- and right-sided advanced adenomas.
IN PRACTICE:
"This highlights the potential clinical utility of the test in identifying candidates for minimally invasive therapeutic approaches tool for CRC," the authors wrote. "Notably, the high sensitivity observed for early-stage CRC and the favorable sensitivity for [advanced adenoma] suggest that this cfDNA test may offer benefits not only in diagnosis but also in prognosis and ultimately in CRC prevention."
SOURCE:
This study was led by Seung Wook Hong, MD, Asan Medical Center in Seoul, South Korea. It was published online on November 19, 2025, in the American Journal of Gastroenterology.
LIMITATIONS:
The case-control design introduced spectrum bias by comparing clearly defined CRC and advanced adenomas cases with individuals who had normal colonoscopy results. The CRC prevalence of 17% to 18% was higher than that observed in true screening populations, limiting generalizability. The exclusively Korean cohort limited extrapolation to non-Asian populations.
DISCLOSURES:
The study received support from GC Genome, Yongin, South Korea. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Cell-Free DNA Blood Test Shows Strong Performance in Detecting Early-Stage CRC
Cell-Free DNA Blood Test Shows Strong Performance in Detecting Early-Stage CRC
Geographic Clusters Show Uneven Cancer Screening in the US
Geographic Clusters Show Uneven Cancer Screening in the US
TOPLINE:
An analysis of 3142 US counties revealed that county-level screening for breast, cervical, and colorectal cancer increased overall between 1997 and 2019; however, despite the reduced geographic variation, persistently high-screening clusters remained in the Northeast, whereas persistently low-screening clusters remained in the Southwest.
METHODOLOGY:
- Cancer screening reduces mortality. Despite guideline recommendation, the uptake of breast, cervical, and colorectal cancer screening in the US falls short of national goals and varies across sociodemographic groups. To date, only a few studies have examined geographic and temporal patterns of screening.
- To address this gap, researchers conducted a cross-sectional study using an ecological panel design to analyze county-level screening prevalence across 3142 US mainland counties from 1997 to 2019, deriving prevalence estimates from Behavioral Risk Factor Surveillance System (BRFSS) and National Health Interview Survey (NHIS) data over 3- to 5-year periods.
- Spatial autocorrelation analyses, including Global Moran I and the bivariate local indicator of spatial autocorrelation, were performed to assess geographic clusters of cancer screening within each period. Four types of local geographic clusters of county-level cancer screening were identified: counties with persistently high screening rates, counties with persistently low screening rates, counties in which screening rates decreased from high to low, and counties in which screening rates increased from low to high.
- Screening prevalence was compared across multiple time windows for different modalities (mammography, a Papanicolaou test, colonoscopy, colorectal cancer test, endoscopy, and a fecal occult blood test [FOBT]). Overall, 3101 counties were analyzed for mammography and the Papanicolaou test, 3107 counties for colonoscopy, 3100 counties for colorectal cancer test, 3089 counties for endoscopy, and 3090 counties for the FOBT.
TAKEAWAY:
- Overall screening prevalence increased from 1997 to 2019, and global spatial autocorrelation declined over time. For instance, the distribution of mammography screening became 83% more uniform in more recent years (Moran I, 0.57 in 1997-1999 vs 0.10 in 2017-2019). Similarly, Papanicolaou test screening became more uniform in more recent years (Moran I, 0.44 vs. 0.07). These changes indicate reduced geographic heterogeneity.
- Colonoscopy and endoscopy use increased, surpassing a 50% prevalence in many counties for 2010; however, FOBT use declined. Spatial clustering also attenuated, with a 23.4% declined in Moran I for colonoscopy from 2011-2016 to 2017-2019, a 12.3% decline in the colorectal cancer test from 2004-2007 to 2008-2010, and a 14.0% decline for endoscopy from 2004-2007 to 2008-2010.
- Persistently high-/high-screening clusters were concentrated in the Northeast for mammography and colorectal cancer screening and in the East for Papanicolaou test screening, whereas persistently low-/low-screening clusters were concentrated in the Southwest for the same modalities.
- Clusters of low- and high-screening counties were more disadvantaged -- with lower socioeconomic status and a higher proportion of non-White residents -- than other cluster types, suggesting some improvement in screening uptake in more disadvantaged areas. Counties with persistently low screening exhibited greater socioeconomic disadvantages -- lower media household income, higher poverty, lower home values, and lower educational attainment -- than those with persistently high screening.
IN PRACTICE:
"This cross-sectional study found that despite secular increases that reduced geographic variation in screening, local clusters of high and low screening persisted in the Northeast and Southwest US, respectively. Future studies could incorporate health care access characteristics to explain why areas of low screening did not catch up to optimize cancer screening practice," the authors wrote.
SOURCE:
The study, led by Pranoti Pradhan, PhD, Harvard T.H. Chan School of Public Health, Boston, was published online in JAMA Network Open.
LIMITATIONS:
The county-level estimates were modeled using BRFSS, NHIS, and US Census data, which might be susceptible to sampling biases despite corrections for nonresponse and noncoverage. Researchers lacked data on specific health systems characteristics that may have directly driven changes in prevalence and were restricted to using screening time intervals available from the Small Area Estimates for Cancer-Relates Measures from the National Cancer Institute, rather than those according to US Preventive Services Task Force guidelines. Additionally, the spatial cluster method was sensitive to county size and arrangement, which may have influenced local cluster detection.
DISCLOSURES:
This research was supported by the T32 Cancer Prevention and Control Funding Fellowship and T32 Cancer Epidemiology Fellowship at the Harvard T.H. Chan School of Public Health. The authors declared having no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
An analysis of 3142 US counties revealed that county-level screening for breast, cervical, and colorectal cancer increased overall between 1997 and 2019; however, despite the reduced geographic variation, persistently high-screening clusters remained in the Northeast, whereas persistently low-screening clusters remained in the Southwest.
METHODOLOGY:
- Cancer screening reduces mortality. Despite guideline recommendation, the uptake of breast, cervical, and colorectal cancer screening in the US falls short of national goals and varies across sociodemographic groups. To date, only a few studies have examined geographic and temporal patterns of screening.
- To address this gap, researchers conducted a cross-sectional study using an ecological panel design to analyze county-level screening prevalence across 3142 US mainland counties from 1997 to 2019, deriving prevalence estimates from Behavioral Risk Factor Surveillance System (BRFSS) and National Health Interview Survey (NHIS) data over 3- to 5-year periods.
- Spatial autocorrelation analyses, including Global Moran I and the bivariate local indicator of spatial autocorrelation, were performed to assess geographic clusters of cancer screening within each period. Four types of local geographic clusters of county-level cancer screening were identified: counties with persistently high screening rates, counties with persistently low screening rates, counties in which screening rates decreased from high to low, and counties in which screening rates increased from low to high.
- Screening prevalence was compared across multiple time windows for different modalities (mammography, a Papanicolaou test, colonoscopy, colorectal cancer test, endoscopy, and a fecal occult blood test [FOBT]). Overall, 3101 counties were analyzed for mammography and the Papanicolaou test, 3107 counties for colonoscopy, 3100 counties for colorectal cancer test, 3089 counties for endoscopy, and 3090 counties for the FOBT.
TAKEAWAY:
- Overall screening prevalence increased from 1997 to 2019, and global spatial autocorrelation declined over time. For instance, the distribution of mammography screening became 83% more uniform in more recent years (Moran I, 0.57 in 1997-1999 vs 0.10 in 2017-2019). Similarly, Papanicolaou test screening became more uniform in more recent years (Moran I, 0.44 vs. 0.07). These changes indicate reduced geographic heterogeneity.
- Colonoscopy and endoscopy use increased, surpassing a 50% prevalence in many counties for 2010; however, FOBT use declined. Spatial clustering also attenuated, with a 23.4% declined in Moran I for colonoscopy from 2011-2016 to 2017-2019, a 12.3% decline in the colorectal cancer test from 2004-2007 to 2008-2010, and a 14.0% decline for endoscopy from 2004-2007 to 2008-2010.
- Persistently high-/high-screening clusters were concentrated in the Northeast for mammography and colorectal cancer screening and in the East for Papanicolaou test screening, whereas persistently low-/low-screening clusters were concentrated in the Southwest for the same modalities.
- Clusters of low- and high-screening counties were more disadvantaged -- with lower socioeconomic status and a higher proportion of non-White residents -- than other cluster types, suggesting some improvement in screening uptake in more disadvantaged areas. Counties with persistently low screening exhibited greater socioeconomic disadvantages -- lower media household income, higher poverty, lower home values, and lower educational attainment -- than those with persistently high screening.
IN PRACTICE:
"This cross-sectional study found that despite secular increases that reduced geographic variation in screening, local clusters of high and low screening persisted in the Northeast and Southwest US, respectively. Future studies could incorporate health care access characteristics to explain why areas of low screening did not catch up to optimize cancer screening practice," the authors wrote.
SOURCE:
The study, led by Pranoti Pradhan, PhD, Harvard T.H. Chan School of Public Health, Boston, was published online in JAMA Network Open.
LIMITATIONS:
The county-level estimates were modeled using BRFSS, NHIS, and US Census data, which might be susceptible to sampling biases despite corrections for nonresponse and noncoverage. Researchers lacked data on specific health systems characteristics that may have directly driven changes in prevalence and were restricted to using screening time intervals available from the Small Area Estimates for Cancer-Relates Measures from the National Cancer Institute, rather than those according to US Preventive Services Task Force guidelines. Additionally, the spatial cluster method was sensitive to county size and arrangement, which may have influenced local cluster detection.
DISCLOSURES:
This research was supported by the T32 Cancer Prevention and Control Funding Fellowship and T32 Cancer Epidemiology Fellowship at the Harvard T.H. Chan School of Public Health. The authors declared having no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
An analysis of 3142 US counties revealed that county-level screening for breast, cervical, and colorectal cancer increased overall between 1997 and 2019; however, despite the reduced geographic variation, persistently high-screening clusters remained in the Northeast, whereas persistently low-screening clusters remained in the Southwest.
METHODOLOGY:
- Cancer screening reduces mortality. Despite guideline recommendation, the uptake of breast, cervical, and colorectal cancer screening in the US falls short of national goals and varies across sociodemographic groups. To date, only a few studies have examined geographic and temporal patterns of screening.
- To address this gap, researchers conducted a cross-sectional study using an ecological panel design to analyze county-level screening prevalence across 3142 US mainland counties from 1997 to 2019, deriving prevalence estimates from Behavioral Risk Factor Surveillance System (BRFSS) and National Health Interview Survey (NHIS) data over 3- to 5-year periods.
- Spatial autocorrelation analyses, including Global Moran I and the bivariate local indicator of spatial autocorrelation, were performed to assess geographic clusters of cancer screening within each period. Four types of local geographic clusters of county-level cancer screening were identified: counties with persistently high screening rates, counties with persistently low screening rates, counties in which screening rates decreased from high to low, and counties in which screening rates increased from low to high.
- Screening prevalence was compared across multiple time windows for different modalities (mammography, a Papanicolaou test, colonoscopy, colorectal cancer test, endoscopy, and a fecal occult blood test [FOBT]). Overall, 3101 counties were analyzed for mammography and the Papanicolaou test, 3107 counties for colonoscopy, 3100 counties for colorectal cancer test, 3089 counties for endoscopy, and 3090 counties for the FOBT.
TAKEAWAY:
- Overall screening prevalence increased from 1997 to 2019, and global spatial autocorrelation declined over time. For instance, the distribution of mammography screening became 83% more uniform in more recent years (Moran I, 0.57 in 1997-1999 vs 0.10 in 2017-2019). Similarly, Papanicolaou test screening became more uniform in more recent years (Moran I, 0.44 vs. 0.07). These changes indicate reduced geographic heterogeneity.
- Colonoscopy and endoscopy use increased, surpassing a 50% prevalence in many counties for 2010; however, FOBT use declined. Spatial clustering also attenuated, with a 23.4% declined in Moran I for colonoscopy from 2011-2016 to 2017-2019, a 12.3% decline in the colorectal cancer test from 2004-2007 to 2008-2010, and a 14.0% decline for endoscopy from 2004-2007 to 2008-2010.
- Persistently high-/high-screening clusters were concentrated in the Northeast for mammography and colorectal cancer screening and in the East for Papanicolaou test screening, whereas persistently low-/low-screening clusters were concentrated in the Southwest for the same modalities.
- Clusters of low- and high-screening counties were more disadvantaged -- with lower socioeconomic status and a higher proportion of non-White residents -- than other cluster types, suggesting some improvement in screening uptake in more disadvantaged areas. Counties with persistently low screening exhibited greater socioeconomic disadvantages -- lower media household income, higher poverty, lower home values, and lower educational attainment -- than those with persistently high screening.
IN PRACTICE:
"This cross-sectional study found that despite secular increases that reduced geographic variation in screening, local clusters of high and low screening persisted in the Northeast and Southwest US, respectively. Future studies could incorporate health care access characteristics to explain why areas of low screening did not catch up to optimize cancer screening practice," the authors wrote.
SOURCE:
The study, led by Pranoti Pradhan, PhD, Harvard T.H. Chan School of Public Health, Boston, was published online in JAMA Network Open.
LIMITATIONS:
The county-level estimates were modeled using BRFSS, NHIS, and US Census data, which might be susceptible to sampling biases despite corrections for nonresponse and noncoverage. Researchers lacked data on specific health systems characteristics that may have directly driven changes in prevalence and were restricted to using screening time intervals available from the Small Area Estimates for Cancer-Relates Measures from the National Cancer Institute, rather than those according to US Preventive Services Task Force guidelines. Additionally, the spatial cluster method was sensitive to county size and arrangement, which may have influenced local cluster detection.
DISCLOSURES:
This research was supported by the T32 Cancer Prevention and Control Funding Fellowship and T32 Cancer Epidemiology Fellowship at the Harvard T.H. Chan School of Public Health. The authors declared having no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Geographic Clusters Show Uneven Cancer Screening in the US
Geographic Clusters Show Uneven Cancer Screening in the US
Improving Colorectal Cancer Screening via Mailed Fecal Immunochemical Testing in a Veterans Affairs Health System
Colorectal cancer (CRC) is among the most common cancers and causes of cancer-related deaths in the United States.1 Reflective of a nationwide trend, CRC screening rates at the Veterans Affairs Connecticut Healthcare System (VACHS) decreased during the COVID-19 pandemic.2-5 Contributing factors to this decrease included cancellations of elective colonoscopies during the initial phase of the pandemic and concurrent turnover of endoscopists. In 2021, the US Preventive Services Task Force lowered the recommended initial CRC screening age from 50 years to 45 years, further increasing the backlog of unscreened patients.6
Fecal immunochemical testing (FIT) is a noninvasive screening method in which antibodies are used to detect hemoglobin in the stool. The sensitivity and specificity of 1-time FIT are 79% to 80% and 94%, respectively, for the detection of CRC, with sensitivity improving with successive testing.7,8 Annual FIT is recognized as a tier 1 preferred screening method by the US Multi-Society Task Force on Colorectal Cancer.7,9 Programs that mail FIT kits to eligible patients outside of physician visits have been successfully implemented in health care systems.10,11
The VACHS designed and implemented a mailed FIT program using existing infrastructure and staffing.
Program Description
A team of local stakeholders comprised of VACHS leadership, primary care, nursing, and gastroenterology staff, as well as representatives from laboratory, informatics, mail services, and group practice management, was established to execute the project. The team met monthly to plan the project.
The team developed a dataset consisting of patients aged 45 to 75 years who were at average risk for CRC and due for CRC screening. Patients were defined as due for CRC screening if they had not had a colonoscopy in the previous 9 years or a FIT or fecal occult blood test in the previous 11 months. Average risk for CRC was defined by excluding patients with associated diagnosis codes for CRC, colectomy, inflammatory bowel disease, and anemia. The program also excluded patients with diagnosis codes associated with dementia, deferring discussions about cancer screening to their primary care practitioners (PCPs). Patients with invalid mailing addresses were also excluded, as well as those whose PCPs had indicated in the electronic health record that the patient received CRC screening outside the US Department of Veterans Affairs (VA) system.
Letter Templates
Two patient letter electronic health record templates were developed. The first was a primer letter, which was mailed to patients 2 to 3 weeks before the mailed FIT kit as an introduction to the program.12 The purpose of the primer letter was to give advance notice to patients that they could expect a FIT kit to arrive in the mail. The goal was to prepare patients to complete FIT when the kit arrived and prompt them to call the VA to opt out of the mailed FIT program if they were up to date with CRC screening or if they had a condition which made them at high risk for CRC.
The second FIT letter arrived with the FIT kit, introduced FIT and described the importance of CRC screening. The letter detailed instructions for completing FIT and automatically created a FIT order. It also included a list of common conditions that may exclude patients, with a recommendation for patients to contact their medical team if they felt they were not candidates for FIT.
Staff Education
A previous VACHS pilot project demonstrated the success of a mailed FIT program to increase FIT use. Implemented as part of the pilot program, staff education consisted of a session for clinicians about the role of FIT in CRC screening and an all-staff education session. An additional education session about CRC and FIT for all staff was repeated with the program launch.
Program Launch
The mailed FIT program was introduced during a VACHS primary care all-staff meeting. After the meeting, each patient aligned care team (PACT) received an encrypted email that included a list of the patients on their team who were candidates for the program, a patient-facing FIT instruction sheet, detailed instructions on how to send the FIT primer letter, and a FIT package consisting of the labeled FIT kit, FIT letter, and patient instruction sheet. A reminder letter was sent to each patient 3 weeks after the FIT package was mailed. The patient lists were populated into a shared, encrypted Microsoft Teams folder that was edited in real time by PACT teams and viewed by VACHS leadership to track progress.
Program Metrics
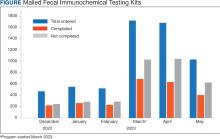
At program launch, the VACHS had 4642 patients due for CRC screening who were eligible for the mailed FIT program. On March 7, 2023, the data consisting of FIT tests ordered between December 2022 and May 2023—3 months before and after the launch of the program—were reviewed and categorized. In the 3 months before program launch, 1528 FIT were ordered and 714 were returned (46.7%). In the 3 months after the launch of the program, 4383 FIT were ordered and 1712 were returned (39.1%) (Figure). Test orders increased 287% from the preintervention to the postintervention period. The mean (SD) number of monthly FIT tests prelaunch was 509 (32.7), which increased to 1461 (331.6) postlaunch.
At the VACHS, 61.4% of patients aged 45 to 75 years were up to date with CRC screening before the program launch. In the 3 months after program launch, the rate increased to 63.8% among patients aged 45 to 75 years, the highest rate in our Veterans Integrated Services Network and exceeding the VA national average CRC screening rate, according to unpublished VA Monthly Management Report data.
In the 3 months following the program launch, 139 FIT kits tested positive for potential CRC. Of these, 79 (56.8%) patients had completed a diagnostic colonoscopy. PACT PCPs and nurses received reports on patients with positive FIT tests and those with no colonoscopy scheduled or completed and were asked to follow up.
Discussion
Through a proactive, population-based CRC screening program centered on mailed FIT kits outside of the traditional patient visit, the VACHS increased the use of FIT and rates of CRC screening. The numbers of FIT kits ordered and completed substantially increased in the 3 months after program launch.
Compared to mailed FIT programs described in the literature that rely on centralized processes in that a separate team operates the mailed FIT program for the entire organization, this program used existing PACT infrastructure and staff.10,11 This strategy allowed VACHS to design and implement the program in several months. Not needing to hire new staff or create a central team for the sole purpose of implementing the program allowed us to save on any organizational funding and efforts that would have accompanied the additional staff. The program described in this article may be more attainable for primary care practices or smaller health systems that do not have the capacity for the creation of a centralized process.
Limitations
Although the total number of FIT completions substantially increased during the program, the rate of FIT completion during the mailed FIT program was lower than the rate of completion prior to program launch. This decreased rate of FIT kit completion may be related to separation from a patient visit and potential loss of real-time education with a clinician. The program’s decentralized design increased the existing workload for primary care staff, and as a result, consideration must be given to local staffing levels. Additionally, the report of eligible patients depended on diagnosis codes and may have captured patients with higher-than-average risk of CRC, such as patients with prior history of adenomatous polyps, family history of CRC, or other medical or genetic conditions. We attempted to mitigate this by including a list of conditions that would exclude patients from FIT eligibility in the FIT letter and giving them the option to opt out.
Conclusions
CRC screening rates improved following implementation of a primary care team-centered quality improvement process to proactively identify patients appropriate for FIT and mail them FIT kits. This project highlights that population-health interventions around CRC screening via use of FIT can be successful within a primary care patient-centered medical home model, considering the increases in both CRC screening rates and increase in FIT tests ordered.
1. American Cancer Society. Key statistics for colorectal cancer. Revised January 29, 2024. Accessed June 11, 2024. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
2. Chen RC, Haynes K, Du S, Barron J, Katz AJ. Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncol. 2021;7(6):878-884. doi:10.1001/jamaoncol.2021.0884
3. Mazidimoradi A, Tiznobaik A, Salehiniya H. Impact of the COVID-19 pandemic on colorectal cancer screening: a systematic review. J Gastrointest Cancer. 2022;53(3):730-744. doi:10.1007/s12029-021-00679-x
4. Adams MA, Kurlander JE, Gao Y, Yankey N, Saini SD. Impact of coronavirus disease 2019 on screening colonoscopy utilization in a large integrated health system. Gastroenterology. 2022;162(7):2098-2100.e2. doi:10.1053/j.gastro.2022.02.034
5. Sundaram S, Olson S, Sharma P, Rajendra S. A review of the impact of the COVID-19 pandemic on colorectal cancer screening: implications and solutions. Pathogens. 2021;10(11):558. doi:10.3390/pathogens10111508
6. US Preventive Services Task Force. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(19):1965-1977. doi:10.1001/jama.2021.6238
7. Robertson DJ, Lee JK, Boland CR, et al. Recommendations on fecal immunochemical testing to screen for colorectal neoplasia: a consensus statement by the US Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc. 2017;85(1):2-21.e3. doi:10.1016/j.gie.2016.09.025
8. Lee JK, Liles EG, Bent S, Levin TR, Corley DA. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160(3):171. doi:10.7326/M13-1484
9. Rex DK, Boland CR, Dominitz JA, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2017;153(1):307-323. doi:10.1053/j.gastro.2017.05.013
10. Deeds SA, Moore CB, Gunnink EJ, et al. Implementation of a mailed faecal immunochemical test programme for colorectal cancer screening among veterans. BMJ Open Qual. 2022;11(4):e001927. doi:10.1136/bmjoq-2022-001927
11. Selby K, Jensen CD, Levin TR, et al. Program components and results from an organized colorectal cancer screening program using annual fecal immunochemical testing. Clin Gastroenterol Hepatol. 2022;20(1):145-152. doi:10.1016/j.cgh.2020.09.042
12. Deeds S, Liu T, Schuttner L, et al. A postcard primer prior to mailed fecal immunochemical test among veterans: a randomized controlled trial. J Gen Intern Med. 2023:38(14):3235-3241. doi:10.1007/s11606-023-08248-7
Colorectal cancer (CRC) is among the most common cancers and causes of cancer-related deaths in the United States.1 Reflective of a nationwide trend, CRC screening rates at the Veterans Affairs Connecticut Healthcare System (VACHS) decreased during the COVID-19 pandemic.2-5 Contributing factors to this decrease included cancellations of elective colonoscopies during the initial phase of the pandemic and concurrent turnover of endoscopists. In 2021, the US Preventive Services Task Force lowered the recommended initial CRC screening age from 50 years to 45 years, further increasing the backlog of unscreened patients.6
Fecal immunochemical testing (FIT) is a noninvasive screening method in which antibodies are used to detect hemoglobin in the stool. The sensitivity and specificity of 1-time FIT are 79% to 80% and 94%, respectively, for the detection of CRC, with sensitivity improving with successive testing.7,8 Annual FIT is recognized as a tier 1 preferred screening method by the US Multi-Society Task Force on Colorectal Cancer.7,9 Programs that mail FIT kits to eligible patients outside of physician visits have been successfully implemented in health care systems.10,11
The VACHS designed and implemented a mailed FIT program using existing infrastructure and staffing.
Program Description
A team of local stakeholders comprised of VACHS leadership, primary care, nursing, and gastroenterology staff, as well as representatives from laboratory, informatics, mail services, and group practice management, was established to execute the project. The team met monthly to plan the project.
The team developed a dataset consisting of patients aged 45 to 75 years who were at average risk for CRC and due for CRC screening. Patients were defined as due for CRC screening if they had not had a colonoscopy in the previous 9 years or a FIT or fecal occult blood test in the previous 11 months. Average risk for CRC was defined by excluding patients with associated diagnosis codes for CRC, colectomy, inflammatory bowel disease, and anemia. The program also excluded patients with diagnosis codes associated with dementia, deferring discussions about cancer screening to their primary care practitioners (PCPs). Patients with invalid mailing addresses were also excluded, as well as those whose PCPs had indicated in the electronic health record that the patient received CRC screening outside the US Department of Veterans Affairs (VA) system.
Letter Templates
Two patient letter electronic health record templates were developed. The first was a primer letter, which was mailed to patients 2 to 3 weeks before the mailed FIT kit as an introduction to the program.12 The purpose of the primer letter was to give advance notice to patients that they could expect a FIT kit to arrive in the mail. The goal was to prepare patients to complete FIT when the kit arrived and prompt them to call the VA to opt out of the mailed FIT program if they were up to date with CRC screening or if they had a condition which made them at high risk for CRC.
The second FIT letter arrived with the FIT kit, introduced FIT and described the importance of CRC screening. The letter detailed instructions for completing FIT and automatically created a FIT order. It also included a list of common conditions that may exclude patients, with a recommendation for patients to contact their medical team if they felt they were not candidates for FIT.
Staff Education
A previous VACHS pilot project demonstrated the success of a mailed FIT program to increase FIT use. Implemented as part of the pilot program, staff education consisted of a session for clinicians about the role of FIT in CRC screening and an all-staff education session. An additional education session about CRC and FIT for all staff was repeated with the program launch.
Program Launch
The mailed FIT program was introduced during a VACHS primary care all-staff meeting. After the meeting, each patient aligned care team (PACT) received an encrypted email that included a list of the patients on their team who were candidates for the program, a patient-facing FIT instruction sheet, detailed instructions on how to send the FIT primer letter, and a FIT package consisting of the labeled FIT kit, FIT letter, and patient instruction sheet. A reminder letter was sent to each patient 3 weeks after the FIT package was mailed. The patient lists were populated into a shared, encrypted Microsoft Teams folder that was edited in real time by PACT teams and viewed by VACHS leadership to track progress.
Program Metrics
At program launch, the VACHS had 4642 patients due for CRC screening who were eligible for the mailed FIT program. On March 7, 2023, the data consisting of FIT tests ordered between December 2022 and May 2023—3 months before and after the launch of the program—were reviewed and categorized. In the 3 months before program launch, 1528 FIT were ordered and 714 were returned (46.7%). In the 3 months after the launch of the program, 4383 FIT were ordered and 1712 were returned (39.1%) (Figure). Test orders increased 287% from the preintervention to the postintervention period. The mean (SD) number of monthly FIT tests prelaunch was 509 (32.7), which increased to 1461 (331.6) postlaunch.
At the VACHS, 61.4% of patients aged 45 to 75 years were up to date with CRC screening before the program launch. In the 3 months after program launch, the rate increased to 63.8% among patients aged 45 to 75 years, the highest rate in our Veterans Integrated Services Network and exceeding the VA national average CRC screening rate, according to unpublished VA Monthly Management Report data.
In the 3 months following the program launch, 139 FIT kits tested positive for potential CRC. Of these, 79 (56.8%) patients had completed a diagnostic colonoscopy. PACT PCPs and nurses received reports on patients with positive FIT tests and those with no colonoscopy scheduled or completed and were asked to follow up.
Discussion
Through a proactive, population-based CRC screening program centered on mailed FIT kits outside of the traditional patient visit, the VACHS increased the use of FIT and rates of CRC screening. The numbers of FIT kits ordered and completed substantially increased in the 3 months after program launch.
Compared to mailed FIT programs described in the literature that rely on centralized processes in that a separate team operates the mailed FIT program for the entire organization, this program used existing PACT infrastructure and staff.10,11 This strategy allowed VACHS to design and implement the program in several months. Not needing to hire new staff or create a central team for the sole purpose of implementing the program allowed us to save on any organizational funding and efforts that would have accompanied the additional staff. The program described in this article may be more attainable for primary care practices or smaller health systems that do not have the capacity for the creation of a centralized process.
Limitations
Although the total number of FIT completions substantially increased during the program, the rate of FIT completion during the mailed FIT program was lower than the rate of completion prior to program launch. This decreased rate of FIT kit completion may be related to separation from a patient visit and potential loss of real-time education with a clinician. The program’s decentralized design increased the existing workload for primary care staff, and as a result, consideration must be given to local staffing levels. Additionally, the report of eligible patients depended on diagnosis codes and may have captured patients with higher-than-average risk of CRC, such as patients with prior history of adenomatous polyps, family history of CRC, or other medical or genetic conditions. We attempted to mitigate this by including a list of conditions that would exclude patients from FIT eligibility in the FIT letter and giving them the option to opt out.
Conclusions
CRC screening rates improved following implementation of a primary care team-centered quality improvement process to proactively identify patients appropriate for FIT and mail them FIT kits. This project highlights that population-health interventions around CRC screening via use of FIT can be successful within a primary care patient-centered medical home model, considering the increases in both CRC screening rates and increase in FIT tests ordered.
Colorectal cancer (CRC) is among the most common cancers and causes of cancer-related deaths in the United States.1 Reflective of a nationwide trend, CRC screening rates at the Veterans Affairs Connecticut Healthcare System (VACHS) decreased during the COVID-19 pandemic.2-5 Contributing factors to this decrease included cancellations of elective colonoscopies during the initial phase of the pandemic and concurrent turnover of endoscopists. In 2021, the US Preventive Services Task Force lowered the recommended initial CRC screening age from 50 years to 45 years, further increasing the backlog of unscreened patients.6
Fecal immunochemical testing (FIT) is a noninvasive screening method in which antibodies are used to detect hemoglobin in the stool. The sensitivity and specificity of 1-time FIT are 79% to 80% and 94%, respectively, for the detection of CRC, with sensitivity improving with successive testing.7,8 Annual FIT is recognized as a tier 1 preferred screening method by the US Multi-Society Task Force on Colorectal Cancer.7,9 Programs that mail FIT kits to eligible patients outside of physician visits have been successfully implemented in health care systems.10,11
The VACHS designed and implemented a mailed FIT program using existing infrastructure and staffing.
Program Description
A team of local stakeholders comprised of VACHS leadership, primary care, nursing, and gastroenterology staff, as well as representatives from laboratory, informatics, mail services, and group practice management, was established to execute the project. The team met monthly to plan the project.
The team developed a dataset consisting of patients aged 45 to 75 years who were at average risk for CRC and due for CRC screening. Patients were defined as due for CRC screening if they had not had a colonoscopy in the previous 9 years or a FIT or fecal occult blood test in the previous 11 months. Average risk for CRC was defined by excluding patients with associated diagnosis codes for CRC, colectomy, inflammatory bowel disease, and anemia. The program also excluded patients with diagnosis codes associated with dementia, deferring discussions about cancer screening to their primary care practitioners (PCPs). Patients with invalid mailing addresses were also excluded, as well as those whose PCPs had indicated in the electronic health record that the patient received CRC screening outside the US Department of Veterans Affairs (VA) system.
Letter Templates
Two patient letter electronic health record templates were developed. The first was a primer letter, which was mailed to patients 2 to 3 weeks before the mailed FIT kit as an introduction to the program.12 The purpose of the primer letter was to give advance notice to patients that they could expect a FIT kit to arrive in the mail. The goal was to prepare patients to complete FIT when the kit arrived and prompt them to call the VA to opt out of the mailed FIT program if they were up to date with CRC screening or if they had a condition which made them at high risk for CRC.
The second FIT letter arrived with the FIT kit, introduced FIT and described the importance of CRC screening. The letter detailed instructions for completing FIT and automatically created a FIT order. It also included a list of common conditions that may exclude patients, with a recommendation for patients to contact their medical team if they felt they were not candidates for FIT.
Staff Education
A previous VACHS pilot project demonstrated the success of a mailed FIT program to increase FIT use. Implemented as part of the pilot program, staff education consisted of a session for clinicians about the role of FIT in CRC screening and an all-staff education session. An additional education session about CRC and FIT for all staff was repeated with the program launch.
Program Launch
The mailed FIT program was introduced during a VACHS primary care all-staff meeting. After the meeting, each patient aligned care team (PACT) received an encrypted email that included a list of the patients on their team who were candidates for the program, a patient-facing FIT instruction sheet, detailed instructions on how to send the FIT primer letter, and a FIT package consisting of the labeled FIT kit, FIT letter, and patient instruction sheet. A reminder letter was sent to each patient 3 weeks after the FIT package was mailed. The patient lists were populated into a shared, encrypted Microsoft Teams folder that was edited in real time by PACT teams and viewed by VACHS leadership to track progress.
Program Metrics
At program launch, the VACHS had 4642 patients due for CRC screening who were eligible for the mailed FIT program. On March 7, 2023, the data consisting of FIT tests ordered between December 2022 and May 2023—3 months before and after the launch of the program—were reviewed and categorized. In the 3 months before program launch, 1528 FIT were ordered and 714 were returned (46.7%). In the 3 months after the launch of the program, 4383 FIT were ordered and 1712 were returned (39.1%) (Figure). Test orders increased 287% from the preintervention to the postintervention period. The mean (SD) number of monthly FIT tests prelaunch was 509 (32.7), which increased to 1461 (331.6) postlaunch.
At the VACHS, 61.4% of patients aged 45 to 75 years were up to date with CRC screening before the program launch. In the 3 months after program launch, the rate increased to 63.8% among patients aged 45 to 75 years, the highest rate in our Veterans Integrated Services Network and exceeding the VA national average CRC screening rate, according to unpublished VA Monthly Management Report data.
In the 3 months following the program launch, 139 FIT kits tested positive for potential CRC. Of these, 79 (56.8%) patients had completed a diagnostic colonoscopy. PACT PCPs and nurses received reports on patients with positive FIT tests and those with no colonoscopy scheduled or completed and were asked to follow up.
Discussion
Through a proactive, population-based CRC screening program centered on mailed FIT kits outside of the traditional patient visit, the VACHS increased the use of FIT and rates of CRC screening. The numbers of FIT kits ordered and completed substantially increased in the 3 months after program launch.
Compared to mailed FIT programs described in the literature that rely on centralized processes in that a separate team operates the mailed FIT program for the entire organization, this program used existing PACT infrastructure and staff.10,11 This strategy allowed VACHS to design and implement the program in several months. Not needing to hire new staff or create a central team for the sole purpose of implementing the program allowed us to save on any organizational funding and efforts that would have accompanied the additional staff. The program described in this article may be more attainable for primary care practices or smaller health systems that do not have the capacity for the creation of a centralized process.
Limitations
Although the total number of FIT completions substantially increased during the program, the rate of FIT completion during the mailed FIT program was lower than the rate of completion prior to program launch. This decreased rate of FIT kit completion may be related to separation from a patient visit and potential loss of real-time education with a clinician. The program’s decentralized design increased the existing workload for primary care staff, and as a result, consideration must be given to local staffing levels. Additionally, the report of eligible patients depended on diagnosis codes and may have captured patients with higher-than-average risk of CRC, such as patients with prior history of adenomatous polyps, family history of CRC, or other medical or genetic conditions. We attempted to mitigate this by including a list of conditions that would exclude patients from FIT eligibility in the FIT letter and giving them the option to opt out.
Conclusions
CRC screening rates improved following implementation of a primary care team-centered quality improvement process to proactively identify patients appropriate for FIT and mail them FIT kits. This project highlights that population-health interventions around CRC screening via use of FIT can be successful within a primary care patient-centered medical home model, considering the increases in both CRC screening rates and increase in FIT tests ordered.
1. American Cancer Society. Key statistics for colorectal cancer. Revised January 29, 2024. Accessed June 11, 2024. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
2. Chen RC, Haynes K, Du S, Barron J, Katz AJ. Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncol. 2021;7(6):878-884. doi:10.1001/jamaoncol.2021.0884
3. Mazidimoradi A, Tiznobaik A, Salehiniya H. Impact of the COVID-19 pandemic on colorectal cancer screening: a systematic review. J Gastrointest Cancer. 2022;53(3):730-744. doi:10.1007/s12029-021-00679-x
4. Adams MA, Kurlander JE, Gao Y, Yankey N, Saini SD. Impact of coronavirus disease 2019 on screening colonoscopy utilization in a large integrated health system. Gastroenterology. 2022;162(7):2098-2100.e2. doi:10.1053/j.gastro.2022.02.034
5. Sundaram S, Olson S, Sharma P, Rajendra S. A review of the impact of the COVID-19 pandemic on colorectal cancer screening: implications and solutions. Pathogens. 2021;10(11):558. doi:10.3390/pathogens10111508
6. US Preventive Services Task Force. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(19):1965-1977. doi:10.1001/jama.2021.6238
7. Robertson DJ, Lee JK, Boland CR, et al. Recommendations on fecal immunochemical testing to screen for colorectal neoplasia: a consensus statement by the US Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc. 2017;85(1):2-21.e3. doi:10.1016/j.gie.2016.09.025
8. Lee JK, Liles EG, Bent S, Levin TR, Corley DA. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160(3):171. doi:10.7326/M13-1484
9. Rex DK, Boland CR, Dominitz JA, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2017;153(1):307-323. doi:10.1053/j.gastro.2017.05.013
10. Deeds SA, Moore CB, Gunnink EJ, et al. Implementation of a mailed faecal immunochemical test programme for colorectal cancer screening among veterans. BMJ Open Qual. 2022;11(4):e001927. doi:10.1136/bmjoq-2022-001927
11. Selby K, Jensen CD, Levin TR, et al. Program components and results from an organized colorectal cancer screening program using annual fecal immunochemical testing. Clin Gastroenterol Hepatol. 2022;20(1):145-152. doi:10.1016/j.cgh.2020.09.042
12. Deeds S, Liu T, Schuttner L, et al. A postcard primer prior to mailed fecal immunochemical test among veterans: a randomized controlled trial. J Gen Intern Med. 2023:38(14):3235-3241. doi:10.1007/s11606-023-08248-7
1. American Cancer Society. Key statistics for colorectal cancer. Revised January 29, 2024. Accessed June 11, 2024. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
2. Chen RC, Haynes K, Du S, Barron J, Katz AJ. Association of cancer screening deficit in the United States with the COVID-19 pandemic. JAMA Oncol. 2021;7(6):878-884. doi:10.1001/jamaoncol.2021.0884
3. Mazidimoradi A, Tiznobaik A, Salehiniya H. Impact of the COVID-19 pandemic on colorectal cancer screening: a systematic review. J Gastrointest Cancer. 2022;53(3):730-744. doi:10.1007/s12029-021-00679-x
4. Adams MA, Kurlander JE, Gao Y, Yankey N, Saini SD. Impact of coronavirus disease 2019 on screening colonoscopy utilization in a large integrated health system. Gastroenterology. 2022;162(7):2098-2100.e2. doi:10.1053/j.gastro.2022.02.034
5. Sundaram S, Olson S, Sharma P, Rajendra S. A review of the impact of the COVID-19 pandemic on colorectal cancer screening: implications and solutions. Pathogens. 2021;10(11):558. doi:10.3390/pathogens10111508
6. US Preventive Services Task Force. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(19):1965-1977. doi:10.1001/jama.2021.6238
7. Robertson DJ, Lee JK, Boland CR, et al. Recommendations on fecal immunochemical testing to screen for colorectal neoplasia: a consensus statement by the US Multi-Society Task Force on Colorectal Cancer. Gastrointest Endosc. 2017;85(1):2-21.e3. doi:10.1016/j.gie.2016.09.025
8. Lee JK, Liles EG, Bent S, Levin TR, Corley DA. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160(3):171. doi:10.7326/M13-1484
9. Rex DK, Boland CR, Dominitz JA, et al. Colorectal cancer screening: recommendations for physicians and patients from the U.S. Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2017;153(1):307-323. doi:10.1053/j.gastro.2017.05.013
10. Deeds SA, Moore CB, Gunnink EJ, et al. Implementation of a mailed faecal immunochemical test programme for colorectal cancer screening among veterans. BMJ Open Qual. 2022;11(4):e001927. doi:10.1136/bmjoq-2022-001927
11. Selby K, Jensen CD, Levin TR, et al. Program components and results from an organized colorectal cancer screening program using annual fecal immunochemical testing. Clin Gastroenterol Hepatol. 2022;20(1):145-152. doi:10.1016/j.cgh.2020.09.042
12. Deeds S, Liu T, Schuttner L, et al. A postcard primer prior to mailed fecal immunochemical test among veterans: a randomized controlled trial. J Gen Intern Med. 2023:38(14):3235-3241. doi:10.1007/s11606-023-08248-7
VHA CRC Screening Has Blind Spots, Disparities
TOPLINE:
More than 1 in 8 colorectal cancer (CRC) cases among veterans occur outside the standard screening age of 50-75 years or those with high-risk personal or family history. High-risk patients face > 6 times the risk for CRC compared with average-risk patients aged 50-75 years who are up to date with screening, while Black patients have > 50% higher risk compared with White patients.
METHODOLOGY:
Researchers conducted a case-control analysis using Veterans Health Administration (VHA) Corporate Data Warehouse data from 2012-2018 at 2 sites: Veterans Affairs (VA) New York Harbor Health Care System and VA Puget Sound Health Care System.
Participants included 3714 cases among veterans with CRC matched to 14,856 controls (4:1), with matching on age (± 3 years), sex, and facility site; each control was used once.
Screening categories included 5 groups by age (50-75 years vs < 50 years or > 75 years), screening up-to-date status, and high-risk status (inflammatory bowel disease, hereditary cancer syndromes, or family history).
CRC screening was considered up to date if US Preventive Services Task Force-recommended tests were completed on time (colonoscopy ≤ 10 years; guaiac-based fecal occult blood test or fecal immunochemical test ≤ 1 year).
TAKEAWAY:
Compared with category 1 (age 50-75 years and up-to-date with screening), CRC was associated with category 4 (age < 50 years or > 75 years and not up to date) (odds ratio [OR], 1.40; 95% CI, 1.11-1.78), and category 5 (high risk) (OR, 6.23; 95% CI, 5.06-7.66).
Race and comorbidity associations included higher CRC risk for Black vs White patients (OR, 1.54; 95% CI, 1.37-1.73), and higher CRC risk with diabetes (OR, 1.65; 95% CI, 1.51-1.81) and alcohol use disorder (OR, 1.53; 95% CI, 1.35-1.73).
Among 3714 CRC cases, 71.1% occurred in individuals aged 50-75 years not up to date with screening.
A total of 12.5% of CRC cases occurred in people outside age 50-75 or with high-risk personal or family history, suggesting that conventional screening-adherence metrics may miss a clinically relevant minority.
IN PRACTICE:
“The conventional measure of CRC screening, focused on average-risk individuals aged 50 to 75, does not reflect screening status in an important minority of CRC patients," the authors wrote.
SOURCE:
The study was led by researchers at NYU Grossman School of Medicine and Veterans Affairs New York Harbor Health Care System, and published online July 9, 2026 in Medicine.
LIMITATIONS:
The study population consisted predominantly of male veterans (97.1%), who tend to be older and have more comorbidities compared with the US population, which may limit the generalizability of findings to other populations. Researchers defined screening status cross-sectionally relative to a single point in time rather than assessing longitudinal screening adherence, which may not fully capture the consistency of screening over time that is likely important for defining CRC risk. Veterans may receive screening at non-VA medical facilities, potentially leading to incomplete documentation of screening status and important covariates such as race, ethnicity, and comorbidities. The possibility of residual confounding cannot be excluded despite adjustment for multiple risk factors in the analysis.
DISCLOSURES:
This study received support from NIH grant K08 CA230162 and the AGA Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer, both awarded to Peter S. Liang. Liang disclosed receiving research support from Freenome and serving on the advisory boards for Guardant Health and Natera. The remaining authors reported no funding or conflicts of interest to disclose.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE:
More than 1 in 8 colorectal cancer (CRC) cases among veterans occur outside the standard screening age of 50-75 years or those with high-risk personal or family history. High-risk patients face > 6 times the risk for CRC compared with average-risk patients aged 50-75 years who are up to date with screening, while Black patients have > 50% higher risk compared with White patients.
METHODOLOGY:
Researchers conducted a case-control analysis using Veterans Health Administration (VHA) Corporate Data Warehouse data from 2012-2018 at 2 sites: Veterans Affairs (VA) New York Harbor Health Care System and VA Puget Sound Health Care System.
Participants included 3714 cases among veterans with CRC matched to 14,856 controls (4:1), with matching on age (± 3 years), sex, and facility site; each control was used once.
Screening categories included 5 groups by age (50-75 years vs < 50 years or > 75 years), screening up-to-date status, and high-risk status (inflammatory bowel disease, hereditary cancer syndromes, or family history).
CRC screening was considered up to date if US Preventive Services Task Force-recommended tests were completed on time (colonoscopy ≤ 10 years; guaiac-based fecal occult blood test or fecal immunochemical test ≤ 1 year).
TAKEAWAY:
Compared with category 1 (age 50-75 years and up-to-date with screening), CRC was associated with category 4 (age < 50 years or > 75 years and not up to date) (odds ratio [OR], 1.40; 95% CI, 1.11-1.78), and category 5 (high risk) (OR, 6.23; 95% CI, 5.06-7.66).
Race and comorbidity associations included higher CRC risk for Black vs White patients (OR, 1.54; 95% CI, 1.37-1.73), and higher CRC risk with diabetes (OR, 1.65; 95% CI, 1.51-1.81) and alcohol use disorder (OR, 1.53; 95% CI, 1.35-1.73).
Among 3714 CRC cases, 71.1% occurred in individuals aged 50-75 years not up to date with screening.
A total of 12.5% of CRC cases occurred in people outside age 50-75 or with high-risk personal or family history, suggesting that conventional screening-adherence metrics may miss a clinically relevant minority.
IN PRACTICE:
“The conventional measure of CRC screening, focused on average-risk individuals aged 50 to 75, does not reflect screening status in an important minority of CRC patients," the authors wrote.
SOURCE:
The study was led by researchers at NYU Grossman School of Medicine and Veterans Affairs New York Harbor Health Care System, and published online July 9, 2026 in Medicine.
LIMITATIONS:
The study population consisted predominantly of male veterans (97.1%), who tend to be older and have more comorbidities compared with the US population, which may limit the generalizability of findings to other populations. Researchers defined screening status cross-sectionally relative to a single point in time rather than assessing longitudinal screening adherence, which may not fully capture the consistency of screening over time that is likely important for defining CRC risk. Veterans may receive screening at non-VA medical facilities, potentially leading to incomplete documentation of screening status and important covariates such as race, ethnicity, and comorbidities. The possibility of residual confounding cannot be excluded despite adjustment for multiple risk factors in the analysis.
DISCLOSURES:
This study received support from NIH grant K08 CA230162 and the AGA Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer, both awarded to Peter S. Liang. Liang disclosed receiving research support from Freenome and serving on the advisory boards for Guardant Health and Natera. The remaining authors reported no funding or conflicts of interest to disclose.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE:
More than 1 in 8 colorectal cancer (CRC) cases among veterans occur outside the standard screening age of 50-75 years or those with high-risk personal or family history. High-risk patients face > 6 times the risk for CRC compared with average-risk patients aged 50-75 years who are up to date with screening, while Black patients have > 50% higher risk compared with White patients.
METHODOLOGY:
Researchers conducted a case-control analysis using Veterans Health Administration (VHA) Corporate Data Warehouse data from 2012-2018 at 2 sites: Veterans Affairs (VA) New York Harbor Health Care System and VA Puget Sound Health Care System.
Participants included 3714 cases among veterans with CRC matched to 14,856 controls (4:1), with matching on age (± 3 years), sex, and facility site; each control was used once.
Screening categories included 5 groups by age (50-75 years vs < 50 years or > 75 years), screening up-to-date status, and high-risk status (inflammatory bowel disease, hereditary cancer syndromes, or family history).
CRC screening was considered up to date if US Preventive Services Task Force-recommended tests were completed on time (colonoscopy ≤ 10 years; guaiac-based fecal occult blood test or fecal immunochemical test ≤ 1 year).
TAKEAWAY:
Compared with category 1 (age 50-75 years and up-to-date with screening), CRC was associated with category 4 (age < 50 years or > 75 years and not up to date) (odds ratio [OR], 1.40; 95% CI, 1.11-1.78), and category 5 (high risk) (OR, 6.23; 95% CI, 5.06-7.66).
Race and comorbidity associations included higher CRC risk for Black vs White patients (OR, 1.54; 95% CI, 1.37-1.73), and higher CRC risk with diabetes (OR, 1.65; 95% CI, 1.51-1.81) and alcohol use disorder (OR, 1.53; 95% CI, 1.35-1.73).
Among 3714 CRC cases, 71.1% occurred in individuals aged 50-75 years not up to date with screening.
A total of 12.5% of CRC cases occurred in people outside age 50-75 or with high-risk personal or family history, suggesting that conventional screening-adherence metrics may miss a clinically relevant minority.
IN PRACTICE:
“The conventional measure of CRC screening, focused on average-risk individuals aged 50 to 75, does not reflect screening status in an important minority of CRC patients," the authors wrote.
SOURCE:
The study was led by researchers at NYU Grossman School of Medicine and Veterans Affairs New York Harbor Health Care System, and published online July 9, 2026 in Medicine.
LIMITATIONS:
The study population consisted predominantly of male veterans (97.1%), who tend to be older and have more comorbidities compared with the US population, which may limit the generalizability of findings to other populations. Researchers defined screening status cross-sectionally relative to a single point in time rather than assessing longitudinal screening adherence, which may not fully capture the consistency of screening over time that is likely important for defining CRC risk. Veterans may receive screening at non-VA medical facilities, potentially leading to incomplete documentation of screening status and important covariates such as race, ethnicity, and comorbidities. The possibility of residual confounding cannot be excluded despite adjustment for multiple risk factors in the analysis.
DISCLOSURES:
This study received support from NIH grant K08 CA230162 and the AGA Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer, both awarded to Peter S. Liang. Liang disclosed receiving research support from Freenome and serving on the advisory boards for Guardant Health and Natera. The remaining authors reported no funding or conflicts of interest to disclose.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Tumor Debulking Fails to Boost Survival in Metastatic CRC
Tumor Debulking Fails to Boost Survival in Metastatic CRC
TOPLINE:
In a randomized phase 3 trial, adding tumor debulking to first-line chemotherapy did not significantly improve overall survival or progression-free survival (PFS) and was associated with an increased risk for serious adverse events in patients with multiorgan metastatic colorectal cancer (mCRC). The study found that patients receiving tumor debulking plus chemotherapy and those receiving chemotherapy alone had similar overall survival (median, 30.0 and 27.5 months, respectively) and PFS (median, 10.5 and 10.4 months, respectively).
METHODOLOGY:
- CRC frequently metastasizes, and when the spread is limited, local curative treatments (such as surgery and ablation) yield 5‑year survival rates of 35%-65%. With median overall survival from systemic therapy now exceeding 30 months, local ablative therapies are increasingly combined with systemic treatment for more extensive mCRC; however, randomized trial based-evidence of survival benefits of this approach is lacking.
- Researchers conducted an open-label, multicenter randomized clinical trial, involving 454 patients with multiorgan mCRC, to determine whether reducing the total amount of tumor (referred to as tumor debulking) could improve survival. Only those deemed amenable to at least 80% debulking prior to starting first-line palliative chemotherapy were included.
- A total of 382 patients were randomly assigned 1:1 to receive either chemotherapy alone (n = 192) or tumor debulking followed by chemotherapy (n = 190) after achieving an objective partial or complete response or stable disease following 3 cycles of capecitabine and oxaliplatin or 4 cycles of 5-fluorouracil or leucovorin and oxaliplatin with or without bevacizumab. The chemotherapy alone group continued standard oxaliplatin‑based chemotherapy; in the debulking group, patients with a response received one additional cycle without bevacizumab before local therapy.
- The primary outcome was overall survival, and secondary outcomes included PFS and serious adverse events. The median follow-up duration was 32.3 months.
TAKEAWAY:
- The median overall survival in the chemotherapy alone group vs chemotherapy plus tumor debulking group was 27.5 vs 30.0 months (adjusted hazard ratio [AHR], 0.88; 95% CI, 0.70-1.10; P = .26), indicating no overall survival benefit from adding tumor debulking to first-line palliative chemotherapy.
- The median PFS was also similar between the chemotherapy alone and chemotherapy plus tumor debulking groups (10.4 and 10.5 months, respectively; AHR, 0.83; 95% CI, 0.67-1.02; P = .08). More patients in the combination therapy group vs chemotherapy alone group experienced any serious adverse events of grade 1 or higher (53% vs 39%; P = .006).
- Among patients who achieved a state of stable disease at randomization, a significant overall survival benefit was observed in the intervention group (P for interaction = .04), although no differences in PFS were noted between subgroups (P for interaction = .13).
- Regarding exploratory outcomes, incomplete debulking was associated with much worse survival (median, 16.8 months), whereas maximal (80% or more) and radical debulking were associated with longer median survival (36.6 vs 35.3 months).
- Additionally, fewer patients in the debulking arm completed at least 6 months of chemotherapy (64% vs 77%), and prespecified analyses by BRAF V600E and RAS mutation status did not show a clear overall survival benefit from adding debulking for either mutant or wild‑type tumors.
IN PRACTICE:
“The results of this trial reveal no significant improvement in overall survival or PFS from additional tumor debulking compared with palliative systemic treatment alone in patients with multiorgan mCRC,” the authors of the study wrote, reiterating that “the addition of tumor debulking to palliative chemotherapy should therefore not be considered standard of care” and “use of local therapies for patients with more limited, oligometastatic CRC needs further consideration.”
SOURCE:
The study, led by Elske C. Gootjes, MD, PhD, and Lotte Bakkerus, MD, from the Radboud University Medical Center, Nijmegen, Netherlands, and Anviti A. Adhin, from Erasmus MC Cancer Institute, University Medical Center Rotterdam, Rotterdam, Netherlands, was published online in JAMA.
LIMITATIONS:
Prolonged enrollment could have led to outdated survival estimates and systemic therapy regimens. Additionally, modern systemic chemotherapy regimens such as triplet chemotherapy or chemotherapy plus anti-epidermal growth factor receptor antibodies for left-sided/RAS wild-type tumors were uniformly used.
DISCLOSURES:
The study received funding from the Dutch Cancer Society, the Blokker-Verwer Foundation, and Roche Nederland BV. Some authors reported receiving grants or personal fees or having other ties with various sources. Full disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In a randomized phase 3 trial, adding tumor debulking to first-line chemotherapy did not significantly improve overall survival or progression-free survival (PFS) and was associated with an increased risk for serious adverse events in patients with multiorgan metastatic colorectal cancer (mCRC). The study found that patients receiving tumor debulking plus chemotherapy and those receiving chemotherapy alone had similar overall survival (median, 30.0 and 27.5 months, respectively) and PFS (median, 10.5 and 10.4 months, respectively).
METHODOLOGY:
- CRC frequently metastasizes, and when the spread is limited, local curative treatments (such as surgery and ablation) yield 5‑year survival rates of 35%-65%. With median overall survival from systemic therapy now exceeding 30 months, local ablative therapies are increasingly combined with systemic treatment for more extensive mCRC; however, randomized trial based-evidence of survival benefits of this approach is lacking.
- Researchers conducted an open-label, multicenter randomized clinical trial, involving 454 patients with multiorgan mCRC, to determine whether reducing the total amount of tumor (referred to as tumor debulking) could improve survival. Only those deemed amenable to at least 80% debulking prior to starting first-line palliative chemotherapy were included.
- A total of 382 patients were randomly assigned 1:1 to receive either chemotherapy alone (n = 192) or tumor debulking followed by chemotherapy (n = 190) after achieving an objective partial or complete response or stable disease following 3 cycles of capecitabine and oxaliplatin or 4 cycles of 5-fluorouracil or leucovorin and oxaliplatin with or without bevacizumab. The chemotherapy alone group continued standard oxaliplatin‑based chemotherapy; in the debulking group, patients with a response received one additional cycle without bevacizumab before local therapy.
- The primary outcome was overall survival, and secondary outcomes included PFS and serious adverse events. The median follow-up duration was 32.3 months.
TAKEAWAY:
- The median overall survival in the chemotherapy alone group vs chemotherapy plus tumor debulking group was 27.5 vs 30.0 months (adjusted hazard ratio [AHR], 0.88; 95% CI, 0.70-1.10; P = .26), indicating no overall survival benefit from adding tumor debulking to first-line palliative chemotherapy.
- The median PFS was also similar between the chemotherapy alone and chemotherapy plus tumor debulking groups (10.4 and 10.5 months, respectively; AHR, 0.83; 95% CI, 0.67-1.02; P = .08). More patients in the combination therapy group vs chemotherapy alone group experienced any serious adverse events of grade 1 or higher (53% vs 39%; P = .006).
- Among patients who achieved a state of stable disease at randomization, a significant overall survival benefit was observed in the intervention group (P for interaction = .04), although no differences in PFS were noted between subgroups (P for interaction = .13).
- Regarding exploratory outcomes, incomplete debulking was associated with much worse survival (median, 16.8 months), whereas maximal (80% or more) and radical debulking were associated with longer median survival (36.6 vs 35.3 months).
- Additionally, fewer patients in the debulking arm completed at least 6 months of chemotherapy (64% vs 77%), and prespecified analyses by BRAF V600E and RAS mutation status did not show a clear overall survival benefit from adding debulking for either mutant or wild‑type tumors.
IN PRACTICE:
“The results of this trial reveal no significant improvement in overall survival or PFS from additional tumor debulking compared with palliative systemic treatment alone in patients with multiorgan mCRC,” the authors of the study wrote, reiterating that “the addition of tumor debulking to palliative chemotherapy should therefore not be considered standard of care” and “use of local therapies for patients with more limited, oligometastatic CRC needs further consideration.”
SOURCE:
The study, led by Elske C. Gootjes, MD, PhD, and Lotte Bakkerus, MD, from the Radboud University Medical Center, Nijmegen, Netherlands, and Anviti A. Adhin, from Erasmus MC Cancer Institute, University Medical Center Rotterdam, Rotterdam, Netherlands, was published online in JAMA.
LIMITATIONS:
Prolonged enrollment could have led to outdated survival estimates and systemic therapy regimens. Additionally, modern systemic chemotherapy regimens such as triplet chemotherapy or chemotherapy plus anti-epidermal growth factor receptor antibodies for left-sided/RAS wild-type tumors were uniformly used.
DISCLOSURES:
The study received funding from the Dutch Cancer Society, the Blokker-Verwer Foundation, and Roche Nederland BV. Some authors reported receiving grants or personal fees or having other ties with various sources. Full disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In a randomized phase 3 trial, adding tumor debulking to first-line chemotherapy did not significantly improve overall survival or progression-free survival (PFS) and was associated with an increased risk for serious adverse events in patients with multiorgan metastatic colorectal cancer (mCRC). The study found that patients receiving tumor debulking plus chemotherapy and those receiving chemotherapy alone had similar overall survival (median, 30.0 and 27.5 months, respectively) and PFS (median, 10.5 and 10.4 months, respectively).
METHODOLOGY:
- CRC frequently metastasizes, and when the spread is limited, local curative treatments (such as surgery and ablation) yield 5‑year survival rates of 35%-65%. With median overall survival from systemic therapy now exceeding 30 months, local ablative therapies are increasingly combined with systemic treatment for more extensive mCRC; however, randomized trial based-evidence of survival benefits of this approach is lacking.
- Researchers conducted an open-label, multicenter randomized clinical trial, involving 454 patients with multiorgan mCRC, to determine whether reducing the total amount of tumor (referred to as tumor debulking) could improve survival. Only those deemed amenable to at least 80% debulking prior to starting first-line palliative chemotherapy were included.
- A total of 382 patients were randomly assigned 1:1 to receive either chemotherapy alone (n = 192) or tumor debulking followed by chemotherapy (n = 190) after achieving an objective partial or complete response or stable disease following 3 cycles of capecitabine and oxaliplatin or 4 cycles of 5-fluorouracil or leucovorin and oxaliplatin with or without bevacizumab. The chemotherapy alone group continued standard oxaliplatin‑based chemotherapy; in the debulking group, patients with a response received one additional cycle without bevacizumab before local therapy.
- The primary outcome was overall survival, and secondary outcomes included PFS and serious adverse events. The median follow-up duration was 32.3 months.
TAKEAWAY:
- The median overall survival in the chemotherapy alone group vs chemotherapy plus tumor debulking group was 27.5 vs 30.0 months (adjusted hazard ratio [AHR], 0.88; 95% CI, 0.70-1.10; P = .26), indicating no overall survival benefit from adding tumor debulking to first-line palliative chemotherapy.
- The median PFS was also similar between the chemotherapy alone and chemotherapy plus tumor debulking groups (10.4 and 10.5 months, respectively; AHR, 0.83; 95% CI, 0.67-1.02; P = .08). More patients in the combination therapy group vs chemotherapy alone group experienced any serious adverse events of grade 1 or higher (53% vs 39%; P = .006).
- Among patients who achieved a state of stable disease at randomization, a significant overall survival benefit was observed in the intervention group (P for interaction = .04), although no differences in PFS were noted between subgroups (P for interaction = .13).
- Regarding exploratory outcomes, incomplete debulking was associated with much worse survival (median, 16.8 months), whereas maximal (80% or more) and radical debulking were associated with longer median survival (36.6 vs 35.3 months).
- Additionally, fewer patients in the debulking arm completed at least 6 months of chemotherapy (64% vs 77%), and prespecified analyses by BRAF V600E and RAS mutation status did not show a clear overall survival benefit from adding debulking for either mutant or wild‑type tumors.
IN PRACTICE:
“The results of this trial reveal no significant improvement in overall survival or PFS from additional tumor debulking compared with palliative systemic treatment alone in patients with multiorgan mCRC,” the authors of the study wrote, reiterating that “the addition of tumor debulking to palliative chemotherapy should therefore not be considered standard of care” and “use of local therapies for patients with more limited, oligometastatic CRC needs further consideration.”
SOURCE:
The study, led by Elske C. Gootjes, MD, PhD, and Lotte Bakkerus, MD, from the Radboud University Medical Center, Nijmegen, Netherlands, and Anviti A. Adhin, from Erasmus MC Cancer Institute, University Medical Center Rotterdam, Rotterdam, Netherlands, was published online in JAMA.
LIMITATIONS:
Prolonged enrollment could have led to outdated survival estimates and systemic therapy regimens. Additionally, modern systemic chemotherapy regimens such as triplet chemotherapy or chemotherapy plus anti-epidermal growth factor receptor antibodies for left-sided/RAS wild-type tumors were uniformly used.
DISCLOSURES:
The study received funding from the Dutch Cancer Society, the Blokker-Verwer Foundation, and Roche Nederland BV. Some authors reported receiving grants or personal fees or having other ties with various sources. Full disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Tumor Debulking Fails to Boost Survival in Metastatic CRC
Tumor Debulking Fails to Boost Survival in Metastatic CRC
Treatment Delays in Colorectal Cancer More Common in Urban Men, Racial Minorities
Treatment Delays in Colorectal Cancer More Common in Urban Men, Racial Minorities
TOPLINE:
Among patients with early-onset colorectal cancer (CRC), treatment delays exceeding 90 days were more common in all-urban populations and seemed to disproportionately affect men and Asian or Pacific Islander, Black, and Hispanic patients. Although several differences were statistically significant, the absolute differences in treatment timing were modest — for instance, the mean time to treatment was 20.7 days in all-urban areas vs 17.8 days in mostly rural areas.
METHODOLOGY:
- Adults with early-onset CRC frequently face diagnostic delays and present at an advanced stage, and this is particularly common among men and racially or ethnically minoritized groups in disadvantaged areas. However, studies evaluating how sex, race and ethnicity, and geography affect timely treatment are scarce.
- Researchers conducted a retrospective cross-sectional analysis using data from the Surveillance, Epidemiology, and End Results (SEER) Program, involving 79,090 patients with early-onset CRC between 2006 and 2020.
- Overall, 53.22% were men; 73.9% were aged 40-49 years; and 54.7% were White, 21.0% Hispanic, 13.8% Black, 9.0% Asian or Pacific Islander, and 0.6% American Indian or Alaska Native. More than half (66.5%) resided in all-urban areas, 20.6% in mostly urban areas, 7.0% in mostly rural areas, and 5.9% in all-rural areas.
- Researchers evaluated the time to treatment (defined as treatment initiation within 30, 60, or 90 days after diagnosis) and assessed its associations with sex, race, and rurality. False discovery rate (FDR) adjustment was applied to multivariable analyses to account for multiple comparisons, and FDR-adjusted two-sided P values were reported.
TAKEAWAY:
- The mean time to treatment in the overall cohort was 20.0 days; it was shortest in mostly rural areas (17.8 days) and longest in all-urban areas (20.7 days).
- Among patients in all-urban areas, men had 5% lower likelihood of initiating treatment within 90 days than women (hazard ratio [HR], 0.95; 95% CI, 0.93-0.97).
- Similarly, Asian or Pacific Islander (HR, 0.96; 95% CI, 0.93-0.99; P = .01), Black (HR, 0.95; 95% CI, 0.92-0.98; P = .001), and Hispanic (HR, 0.93; 95% CI, 0.91-0.95; P < .001) patients in all-urban areas were less likely than White patients to start treatment within 90 days. Comparable patterns were seen at the 30- and 60-day thresholds.
- In mostly rural areas, Black patients were more likely than White patients to start treatment earlier (30-day HR, 1.19; 95% CI, 1.06-1.34 and 90-day HR, 1.15; 95% CI, 1.02-1.28), whereas men were less likely than women to initiate treatment within 90 days (HR, 0.90; 95% CI, 0.85-0.96).
- Researchers found that several HRs were statistically significant but were numerically close to 1.00, indicating modest absolute differences in treatment timing.
IN PRACTICE:
“The consistency of these delays across sociodemographic groups challenges assumptions of uniformly timely access in urban settings. Overcrowded urban health care systems or inefficient public transportation may limit access to care,” the authors wrote, noting that “young adults face distinct challenges across life stages, including lack of health insurance among patients aged 18 to 29 years and financial strain among patients aged 30 to 39 years that hinder timely access to treatment.”
SOURCE:
The study, led by Meng-Han Tsai, PhD, Georgia Prevention Institute, Augusta University, Augusta, Georgia, was published online as a research letter in JAMA Network Open.
LIMITATIONS:
The study characterized time-to-treatment patterns rather than clinical outcomes and relied on SEER data without day-level treatment timing. Additionally, the observed HRs were small, but even modest delays may have led to population-level disparities.
DISCLOSURES:
This research was supported by the Augusta ROAR SCORE Career Enhancement Core through a grant awarded to Tsai. The authors declared having no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Among patients with early-onset colorectal cancer (CRC), treatment delays exceeding 90 days were more common in all-urban populations and seemed to disproportionately affect men and Asian or Pacific Islander, Black, and Hispanic patients. Although several differences were statistically significant, the absolute differences in treatment timing were modest — for instance, the mean time to treatment was 20.7 days in all-urban areas vs 17.8 days in mostly rural areas.
METHODOLOGY:
- Adults with early-onset CRC frequently face diagnostic delays and present at an advanced stage, and this is particularly common among men and racially or ethnically minoritized groups in disadvantaged areas. However, studies evaluating how sex, race and ethnicity, and geography affect timely treatment are scarce.
- Researchers conducted a retrospective cross-sectional analysis using data from the Surveillance, Epidemiology, and End Results (SEER) Program, involving 79,090 patients with early-onset CRC between 2006 and 2020.
- Overall, 53.22% were men; 73.9% were aged 40-49 years; and 54.7% were White, 21.0% Hispanic, 13.8% Black, 9.0% Asian or Pacific Islander, and 0.6% American Indian or Alaska Native. More than half (66.5%) resided in all-urban areas, 20.6% in mostly urban areas, 7.0% in mostly rural areas, and 5.9% in all-rural areas.
- Researchers evaluated the time to treatment (defined as treatment initiation within 30, 60, or 90 days after diagnosis) and assessed its associations with sex, race, and rurality. False discovery rate (FDR) adjustment was applied to multivariable analyses to account for multiple comparisons, and FDR-adjusted two-sided P values were reported.
TAKEAWAY:
- The mean time to treatment in the overall cohort was 20.0 days; it was shortest in mostly rural areas (17.8 days) and longest in all-urban areas (20.7 days).
- Among patients in all-urban areas, men had 5% lower likelihood of initiating treatment within 90 days than women (hazard ratio [HR], 0.95; 95% CI, 0.93-0.97).
- Similarly, Asian or Pacific Islander (HR, 0.96; 95% CI, 0.93-0.99; P = .01), Black (HR, 0.95; 95% CI, 0.92-0.98; P = .001), and Hispanic (HR, 0.93; 95% CI, 0.91-0.95; P < .001) patients in all-urban areas were less likely than White patients to start treatment within 90 days. Comparable patterns were seen at the 30- and 60-day thresholds.
- In mostly rural areas, Black patients were more likely than White patients to start treatment earlier (30-day HR, 1.19; 95% CI, 1.06-1.34 and 90-day HR, 1.15; 95% CI, 1.02-1.28), whereas men were less likely than women to initiate treatment within 90 days (HR, 0.90; 95% CI, 0.85-0.96).
- Researchers found that several HRs were statistically significant but were numerically close to 1.00, indicating modest absolute differences in treatment timing.
IN PRACTICE:
“The consistency of these delays across sociodemographic groups challenges assumptions of uniformly timely access in urban settings. Overcrowded urban health care systems or inefficient public transportation may limit access to care,” the authors wrote, noting that “young adults face distinct challenges across life stages, including lack of health insurance among patients aged 18 to 29 years and financial strain among patients aged 30 to 39 years that hinder timely access to treatment.”
SOURCE:
The study, led by Meng-Han Tsai, PhD, Georgia Prevention Institute, Augusta University, Augusta, Georgia, was published online as a research letter in JAMA Network Open.
LIMITATIONS:
The study characterized time-to-treatment patterns rather than clinical outcomes and relied on SEER data without day-level treatment timing. Additionally, the observed HRs were small, but even modest delays may have led to population-level disparities.
DISCLOSURES:
This research was supported by the Augusta ROAR SCORE Career Enhancement Core through a grant awarded to Tsai. The authors declared having no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Among patients with early-onset colorectal cancer (CRC), treatment delays exceeding 90 days were more common in all-urban populations and seemed to disproportionately affect men and Asian or Pacific Islander, Black, and Hispanic patients. Although several differences were statistically significant, the absolute differences in treatment timing were modest — for instance, the mean time to treatment was 20.7 days in all-urban areas vs 17.8 days in mostly rural areas.
METHODOLOGY:
- Adults with early-onset CRC frequently face diagnostic delays and present at an advanced stage, and this is particularly common among men and racially or ethnically minoritized groups in disadvantaged areas. However, studies evaluating how sex, race and ethnicity, and geography affect timely treatment are scarce.
- Researchers conducted a retrospective cross-sectional analysis using data from the Surveillance, Epidemiology, and End Results (SEER) Program, involving 79,090 patients with early-onset CRC between 2006 and 2020.
- Overall, 53.22% were men; 73.9% were aged 40-49 years; and 54.7% were White, 21.0% Hispanic, 13.8% Black, 9.0% Asian or Pacific Islander, and 0.6% American Indian or Alaska Native. More than half (66.5%) resided in all-urban areas, 20.6% in mostly urban areas, 7.0% in mostly rural areas, and 5.9% in all-rural areas.
- Researchers evaluated the time to treatment (defined as treatment initiation within 30, 60, or 90 days after diagnosis) and assessed its associations with sex, race, and rurality. False discovery rate (FDR) adjustment was applied to multivariable analyses to account for multiple comparisons, and FDR-adjusted two-sided P values were reported.
TAKEAWAY:
- The mean time to treatment in the overall cohort was 20.0 days; it was shortest in mostly rural areas (17.8 days) and longest in all-urban areas (20.7 days).
- Among patients in all-urban areas, men had 5% lower likelihood of initiating treatment within 90 days than women (hazard ratio [HR], 0.95; 95% CI, 0.93-0.97).
- Similarly, Asian or Pacific Islander (HR, 0.96; 95% CI, 0.93-0.99; P = .01), Black (HR, 0.95; 95% CI, 0.92-0.98; P = .001), and Hispanic (HR, 0.93; 95% CI, 0.91-0.95; P < .001) patients in all-urban areas were less likely than White patients to start treatment within 90 days. Comparable patterns were seen at the 30- and 60-day thresholds.
- In mostly rural areas, Black patients were more likely than White patients to start treatment earlier (30-day HR, 1.19; 95% CI, 1.06-1.34 and 90-day HR, 1.15; 95% CI, 1.02-1.28), whereas men were less likely than women to initiate treatment within 90 days (HR, 0.90; 95% CI, 0.85-0.96).
- Researchers found that several HRs were statistically significant but were numerically close to 1.00, indicating modest absolute differences in treatment timing.
IN PRACTICE:
“The consistency of these delays across sociodemographic groups challenges assumptions of uniformly timely access in urban settings. Overcrowded urban health care systems or inefficient public transportation may limit access to care,” the authors wrote, noting that “young adults face distinct challenges across life stages, including lack of health insurance among patients aged 18 to 29 years and financial strain among patients aged 30 to 39 years that hinder timely access to treatment.”
SOURCE:
The study, led by Meng-Han Tsai, PhD, Georgia Prevention Institute, Augusta University, Augusta, Georgia, was published online as a research letter in JAMA Network Open.
LIMITATIONS:
The study characterized time-to-treatment patterns rather than clinical outcomes and relied on SEER data without day-level treatment timing. Additionally, the observed HRs were small, but even modest delays may have led to population-level disparities.
DISCLOSURES:
This research was supported by the Augusta ROAR SCORE Career Enhancement Core through a grant awarded to Tsai. The authors declared having no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Treatment Delays in Colorectal Cancer More Common in Urban Men, Racial Minorities
Treatment Delays in Colorectal Cancer More Common in Urban Men, Racial Minorities
The GLP-1 Paradox in Colorectal Cancer
The GLP-1 Paradox in Colorectal Cancer
This transcript has been edited for clarity.
Hi. I'm David Kerr, professor of cancer medicine at University of Oxford. One of the harder lessons I've learned as a cancer doctor, not surprisingly, is that prevention's better than cure. This is something I've become increasingly interested in as I've become more senior in the profession. I say that rather than "becoming older."
I'd like to draw your attention to some interesting work that's been done looking at the risk of developing colorectal cancer. We talk about lifestyle factors, exercise, vitamin D, and sometimes aspirin. There is some plausible evidence, not from randomized trials, suggesting that these interventions can reduce the chance of developing colorectal cancer. With my friend Ian Tomlinson, colleague in Oxford, we have a huge interest in the genetics of predicting who will develop colorectal cancer.
Today I'd like to talk about these new agents, the so-called glucagon-like peptide 1 receptor agonists, or GLP-1 receptor agonists, which are being used widely now to treat type 2 diabetes and obesity. These are remarkably successful drugs with huge worldwide global uptake, but there is debate in the literature and in real-world evidence as to what they do about cancer risk.
You would think that if we reduce body weight and if we reduce adiposity, that truly would reduce the chance of developing cancer. We know that a number of cancers are related to body fat content and so on.
I'd like to focus particularly on my own field of interest, which is colorectal cancer, and an article I picked up recently by Professor Zhong and colleagues, where they did a meta-analysis. This is a statistical method for clumping together large datasets from different studies.
They did a meta-analysis using very conventional, widely accepted methods to look at a very large dataset of just over 5 million individuals from seven retrospective cohort studies, so a big database to study.
There was a pooled analysis, which revealed that there was a significant but slight increase in the risk for colorectal cancer in patients receiving the GLP-1 agonists. Overall, they felt that, given the small but significant increase in the risk of developing colorectal cancer, we need further evidence.
This was a retrospective review of a large dataset, but given debate in the literature, more forward-looking studies are required. It’s the sort of thing that, in real-world use, one might take into account when recommending these treatments, such as Mounjaro.
In patients who have a higher-than-expected risk of developing colorectal cancer, one might hesitate a little. Clearly, if they get diabetes or cardiac disease, those beneficial risks would, of course, weigh one in favor of using these effective new drugs.
For somebody who had borderline BMI, where there were some questions as to whether you would use the drugs or not, and if they had some other colorectal cancer risk factors, such as relatives affected, then one might pause for thought before using them.
This was a well-conducted study that adds to the rather confused literature on the effects of these widely used drugs on the risk for cancer. Again, just that thought that, although it would seem plausible to think the opposite, these drugs would reduce colorectal cancer risk, on review of a very large dataset, actually the opposite seems to be the case. Always go for evidence. The larger, the more convincing the dataset, the better.
I’d be interested in what you thought about this and whether information like this might tip your balance as to whether you would accept using these drugs to reduce your own body weight.
Thanks for listening. For the time being, Medscapers, over and out. Thank you.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I'm David Kerr, professor of cancer medicine at University of Oxford. One of the harder lessons I've learned as a cancer doctor, not surprisingly, is that prevention's better than cure. This is something I've become increasingly interested in as I've become more senior in the profession. I say that rather than "becoming older."
I'd like to draw your attention to some interesting work that's been done looking at the risk of developing colorectal cancer. We talk about lifestyle factors, exercise, vitamin D, and sometimes aspirin. There is some plausible evidence, not from randomized trials, suggesting that these interventions can reduce the chance of developing colorectal cancer. With my friend Ian Tomlinson, colleague in Oxford, we have a huge interest in the genetics of predicting who will develop colorectal cancer.
Today I'd like to talk about these new agents, the so-called glucagon-like peptide 1 receptor agonists, or GLP-1 receptor agonists, which are being used widely now to treat type 2 diabetes and obesity. These are remarkably successful drugs with huge worldwide global uptake, but there is debate in the literature and in real-world evidence as to what they do about cancer risk.
You would think that if we reduce body weight and if we reduce adiposity, that truly would reduce the chance of developing cancer. We know that a number of cancers are related to body fat content and so on.
I'd like to focus particularly on my own field of interest, which is colorectal cancer, and an article I picked up recently by Professor Zhong and colleagues, where they did a meta-analysis. This is a statistical method for clumping together large datasets from different studies.
They did a meta-analysis using very conventional, widely accepted methods to look at a very large dataset of just over 5 million individuals from seven retrospective cohort studies, so a big database to study.
There was a pooled analysis, which revealed that there was a significant but slight increase in the risk for colorectal cancer in patients receiving the GLP-1 agonists. Overall, they felt that, given the small but significant increase in the risk of developing colorectal cancer, we need further evidence.
This was a retrospective review of a large dataset, but given debate in the literature, more forward-looking studies are required. It’s the sort of thing that, in real-world use, one might take into account when recommending these treatments, such as Mounjaro.
In patients who have a higher-than-expected risk of developing colorectal cancer, one might hesitate a little. Clearly, if they get diabetes or cardiac disease, those beneficial risks would, of course, weigh one in favor of using these effective new drugs.
For somebody who had borderline BMI, where there were some questions as to whether you would use the drugs or not, and if they had some other colorectal cancer risk factors, such as relatives affected, then one might pause for thought before using them.
This was a well-conducted study that adds to the rather confused literature on the effects of these widely used drugs on the risk for cancer. Again, just that thought that, although it would seem plausible to think the opposite, these drugs would reduce colorectal cancer risk, on review of a very large dataset, actually the opposite seems to be the case. Always go for evidence. The larger, the more convincing the dataset, the better.
I’d be interested in what you thought about this and whether information like this might tip your balance as to whether you would accept using these drugs to reduce your own body weight.
Thanks for listening. For the time being, Medscapers, over and out. Thank you.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I'm David Kerr, professor of cancer medicine at University of Oxford. One of the harder lessons I've learned as a cancer doctor, not surprisingly, is that prevention's better than cure. This is something I've become increasingly interested in as I've become more senior in the profession. I say that rather than "becoming older."
I'd like to draw your attention to some interesting work that's been done looking at the risk of developing colorectal cancer. We talk about lifestyle factors, exercise, vitamin D, and sometimes aspirin. There is some plausible evidence, not from randomized trials, suggesting that these interventions can reduce the chance of developing colorectal cancer. With my friend Ian Tomlinson, colleague in Oxford, we have a huge interest in the genetics of predicting who will develop colorectal cancer.
Today I'd like to talk about these new agents, the so-called glucagon-like peptide 1 receptor agonists, or GLP-1 receptor agonists, which are being used widely now to treat type 2 diabetes and obesity. These are remarkably successful drugs with huge worldwide global uptake, but there is debate in the literature and in real-world evidence as to what they do about cancer risk.
You would think that if we reduce body weight and if we reduce adiposity, that truly would reduce the chance of developing cancer. We know that a number of cancers are related to body fat content and so on.
I'd like to focus particularly on my own field of interest, which is colorectal cancer, and an article I picked up recently by Professor Zhong and colleagues, where they did a meta-analysis. This is a statistical method for clumping together large datasets from different studies.
They did a meta-analysis using very conventional, widely accepted methods to look at a very large dataset of just over 5 million individuals from seven retrospective cohort studies, so a big database to study.
There was a pooled analysis, which revealed that there was a significant but slight increase in the risk for colorectal cancer in patients receiving the GLP-1 agonists. Overall, they felt that, given the small but significant increase in the risk of developing colorectal cancer, we need further evidence.
This was a retrospective review of a large dataset, but given debate in the literature, more forward-looking studies are required. It’s the sort of thing that, in real-world use, one might take into account when recommending these treatments, such as Mounjaro.
In patients who have a higher-than-expected risk of developing colorectal cancer, one might hesitate a little. Clearly, if they get diabetes or cardiac disease, those beneficial risks would, of course, weigh one in favor of using these effective new drugs.
For somebody who had borderline BMI, where there were some questions as to whether you would use the drugs or not, and if they had some other colorectal cancer risk factors, such as relatives affected, then one might pause for thought before using them.
This was a well-conducted study that adds to the rather confused literature on the effects of these widely used drugs on the risk for cancer. Again, just that thought that, although it would seem plausible to think the opposite, these drugs would reduce colorectal cancer risk, on review of a very large dataset, actually the opposite seems to be the case. Always go for evidence. The larger, the more convincing the dataset, the better.
I’d be interested in what you thought about this and whether information like this might tip your balance as to whether you would accept using these drugs to reduce your own body weight.
Thanks for listening. For the time being, Medscapers, over and out. Thank you.
A version of this article first appeared on Medscape.com.
The GLP-1 Paradox in Colorectal Cancer
The GLP-1 Paradox in Colorectal Cancer
“Colon Age” Tool Evaluates Early CRC Risk in Male Vets
TOPLINE: Interviews with 23 male veterans (aged 35-49 years) at average-risk for colorectal cancer (CRC) and 8 primary care practitioners (PCPs) found broad acceptability of the Colon Age concept, with 96% of patients agreeing to calculation. PCPs describe its potential use to support screening discussions (fecal immunochemical test [FIT] vs colonoscopy) but emphasize workflow barriers, requesting electronic medical record integration and “time neutral” implementation.
METHODOLOGY:
Researchers conducted semistructured qualitative interviews with 31 participants (23 male veteran patients aged 35-49 years and 8 PCPs) at the Richard L. Roudebush Veterans Affairs Medical Center between June and September 2022.
Patients were eligible if they were at average risk for CRC, had no prior screening (colonoscopy or fecal immunochemical test [FIT]), no inflammatory bowel disease, and no significant family history of CRC.
Interviews explored participants' experiences with CRC screening, understanding of the Colon Age tool, and perceived clinical use.
Audio-recorded interviews were transcribed, deidentified, and analyzed using the constant comparison method with open and focused coding phases until saturation was reached.
TAKEAWAY:
Among 23 male veteran patients (mean age 47 years), 96% agreed to have their Colon Age calculated; 68% had a Colon Age below their biological age, 14% higher than their biological age, and 18% equal to their biological age.
Patients accepted the Colon Age concept, finding it easy to understand and helpful for being informed about their health, though most were unaware of screening options beyond colonoscopy prior to the interview.
The 8 PCPs (mean age 53 years, 50% female, mean 29 years in practice) interviewed found the tool acceptable and useful for screening conversations, improving uptake, and facilitating shared decision-making, particularly in gray zone cases where screening decisions are unclear.
PCPs emphasized the need for the tool to be integrated into the electronic medical record system and expressed concerns about time commitment, consistency with practice guidelines, and the validation process, stating they would only use the tool if it were time neutral and evidence-based.
IN PRACTICE: “Although the age at which to begin colorectal cancer screening in the US was lowered to 45 years in 2018, uptake of screening in persons aged 45 to 49 has been slow,” wrote the authors of the study.
SOURCE:The study was led by researchers at the VA Center for Health Information and Communication. It was published online on July 15 in BMC Primary Care.
LIMITATIONS: The study was conducted at a single VA medical center in the Midwest and all patient participants were male, which may limit generalizability to nonveteran patients, female patients, and non-VA clinicians. The Colon Age tool has limitations, as it was based on a risk prediction model with modest discrimination, and the linkage to screening recommendations was based on arbitrary Surveillance, Epidemiology and End Results thresholds chosen by the tool developers. Additionally, the qualitative nature of the study with a small sample size may not capture the full range of perspectives across diverse health care settings and patient populations.
DISCLOSURES: The primary author received support from Health Services Research and Development, Veterans Administration. Funding for this project was provided by Richard L. Roudebush VA Medical Center Indianapolis, Indiana Center for Health Information, and Communication COIN funds. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Interviews with 23 male veterans (aged 35-49 years) at average-risk for colorectal cancer (CRC) and 8 primary care practitioners (PCPs) found broad acceptability of the Colon Age concept, with 96% of patients agreeing to calculation. PCPs describe its potential use to support screening discussions (fecal immunochemical test [FIT] vs colonoscopy) but emphasize workflow barriers, requesting electronic medical record integration and “time neutral” implementation.
METHODOLOGY:
Researchers conducted semistructured qualitative interviews with 31 participants (23 male veteran patients aged 35-49 years and 8 PCPs) at the Richard L. Roudebush Veterans Affairs Medical Center between June and September 2022.
Patients were eligible if they were at average risk for CRC, had no prior screening (colonoscopy or fecal immunochemical test [FIT]), no inflammatory bowel disease, and no significant family history of CRC.
Interviews explored participants' experiences with CRC screening, understanding of the Colon Age tool, and perceived clinical use.
Audio-recorded interviews were transcribed, deidentified, and analyzed using the constant comparison method with open and focused coding phases until saturation was reached.
TAKEAWAY:
Among 23 male veteran patients (mean age 47 years), 96% agreed to have their Colon Age calculated; 68% had a Colon Age below their biological age, 14% higher than their biological age, and 18% equal to their biological age.
Patients accepted the Colon Age concept, finding it easy to understand and helpful for being informed about their health, though most were unaware of screening options beyond colonoscopy prior to the interview.
The 8 PCPs (mean age 53 years, 50% female, mean 29 years in practice) interviewed found the tool acceptable and useful for screening conversations, improving uptake, and facilitating shared decision-making, particularly in gray zone cases where screening decisions are unclear.
PCPs emphasized the need for the tool to be integrated into the electronic medical record system and expressed concerns about time commitment, consistency with practice guidelines, and the validation process, stating they would only use the tool if it were time neutral and evidence-based.
IN PRACTICE: “Although the age at which to begin colorectal cancer screening in the US was lowered to 45 years in 2018, uptake of screening in persons aged 45 to 49 has been slow,” wrote the authors of the study.
SOURCE:The study was led by researchers at the VA Center for Health Information and Communication. It was published online on July 15 in BMC Primary Care.
LIMITATIONS: The study was conducted at a single VA medical center in the Midwest and all patient participants were male, which may limit generalizability to nonveteran patients, female patients, and non-VA clinicians. The Colon Age tool has limitations, as it was based on a risk prediction model with modest discrimination, and the linkage to screening recommendations was based on arbitrary Surveillance, Epidemiology and End Results thresholds chosen by the tool developers. Additionally, the qualitative nature of the study with a small sample size may not capture the full range of perspectives across diverse health care settings and patient populations.
DISCLOSURES: The primary author received support from Health Services Research and Development, Veterans Administration. Funding for this project was provided by Richard L. Roudebush VA Medical Center Indianapolis, Indiana Center for Health Information, and Communication COIN funds. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Interviews with 23 male veterans (aged 35-49 years) at average-risk for colorectal cancer (CRC) and 8 primary care practitioners (PCPs) found broad acceptability of the Colon Age concept, with 96% of patients agreeing to calculation. PCPs describe its potential use to support screening discussions (fecal immunochemical test [FIT] vs colonoscopy) but emphasize workflow barriers, requesting electronic medical record integration and “time neutral” implementation.
METHODOLOGY:
Researchers conducted semistructured qualitative interviews with 31 participants (23 male veteran patients aged 35-49 years and 8 PCPs) at the Richard L. Roudebush Veterans Affairs Medical Center between June and September 2022.
Patients were eligible if they were at average risk for CRC, had no prior screening (colonoscopy or fecal immunochemical test [FIT]), no inflammatory bowel disease, and no significant family history of CRC.
Interviews explored participants' experiences with CRC screening, understanding of the Colon Age tool, and perceived clinical use.
Audio-recorded interviews were transcribed, deidentified, and analyzed using the constant comparison method with open and focused coding phases until saturation was reached.
TAKEAWAY:
Among 23 male veteran patients (mean age 47 years), 96% agreed to have their Colon Age calculated; 68% had a Colon Age below their biological age, 14% higher than their biological age, and 18% equal to their biological age.
Patients accepted the Colon Age concept, finding it easy to understand and helpful for being informed about their health, though most were unaware of screening options beyond colonoscopy prior to the interview.
The 8 PCPs (mean age 53 years, 50% female, mean 29 years in practice) interviewed found the tool acceptable and useful for screening conversations, improving uptake, and facilitating shared decision-making, particularly in gray zone cases where screening decisions are unclear.
PCPs emphasized the need for the tool to be integrated into the electronic medical record system and expressed concerns about time commitment, consistency with practice guidelines, and the validation process, stating they would only use the tool if it were time neutral and evidence-based.
IN PRACTICE: “Although the age at which to begin colorectal cancer screening in the US was lowered to 45 years in 2018, uptake of screening in persons aged 45 to 49 has been slow,” wrote the authors of the study.
SOURCE:The study was led by researchers at the VA Center for Health Information and Communication. It was published online on July 15 in BMC Primary Care.
LIMITATIONS: The study was conducted at a single VA medical center in the Midwest and all patient participants were male, which may limit generalizability to nonveteran patients, female patients, and non-VA clinicians. The Colon Age tool has limitations, as it was based on a risk prediction model with modest discrimination, and the linkage to screening recommendations was based on arbitrary Surveillance, Epidemiology and End Results thresholds chosen by the tool developers. Additionally, the qualitative nature of the study with a small sample size may not capture the full range of perspectives across diverse health care settings and patient populations.
DISCLOSURES: The primary author received support from Health Services Research and Development, Veterans Administration. Funding for this project was provided by Richard L. Roudebush VA Medical Center Indianapolis, Indiana Center for Health Information, and Communication COIN funds. The authors reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
'Concerning': CRC Continues to Shift Toward Younger Adults
'Concerning': CRC Continues to Shift Toward Younger Adults
Colorectal cancer (CRC) in the United States continues to move in two different directions — decreasing in older adults and rising in younger adults, especially in those aged 20-49 years, according to the latest statistics from the American Cancer Society (ACS).
The ACS report, published online earlier this month, revealed that CRC incidence rates declined by 2.5% annually in adults aged ≥ 65 years but increased by 3% annually in adults aged 20-49 between 2013 and 2022 — higher than earlier estimates of 1% to 2% annual increases.
The trends are “concerning” and a “stark reminder that we’re seeing a shifting epidemiology,” said Folasade (Fola) May, MD, PhD, MPhil, director of the gastroenterology quality improvement program at UCLA Health in Los Angeles, who wasn’t involved in the analysis.
The report highlights the need for better education and symptom awareness — including bleeding, iron deficiency symptoms, and changes in bowel habits — among patients and doctors, who may not routinely consider cancer in younger adults, May explained.
“Because so many of the young people diagnosed present with advanced stage disease, early workup is critical to saving lives,” she said.
Rapidly Changing Landscape
In the United States, CRC is the third-most commonly diagnosed cancer in both men and women. CRC is also the second-leading cause of cancer-related deaths and the leading cause in adults aged < 50 years.
“After decades of progress, the risk of dying from colorectal cancer is climbing in younger generations of men and women, confirming a real uptick in disease because of something we’re doing or some other exposure,” Rebecca Siegel, MPH, senior scientific director of surveillance research at ACS and lead author of the report, said in a statement.
For the latest CRC statistics report, ACS scientists analyzed population-based registries, including the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program, and mortality data from the CDC’s National Center for Health Statistics.
The report estimated that 158,850 new CRC cases will be diagnosed in the US in 2026, including 108,860 colon cancers and 49,990 rectal cancers; an estimated 55,230 people will die from the disease.
Overall, CRC incidence declined by 0.9% annually from 2013 to 2022, driven by decreases of 2.5% per year in adults aged ≥ 65 years. During the same period, however, incidence rates rose by about 3% per year in adults aged 20-49 years and by 0.4% per year in those aged 50-64 years. CRC mortality also continued to trend downward in adults aged ≥ 65 years by > 2% per year, but mortality increased by 1% per year in adults aged < 50 years since 2004 and in adults aged 50-64 years since 2019.
Nearly half of new CRC cases (45%) now occur in adults aged < 65 years, up from 27% in 1995, illustrating a major shift toward younger age groups, the authors said. Half of early-onset cases occur in people aged 45-49 years who are now eligible for screening, and 3 of 4 early-onset CRC cases are diagnosed at an advanced stage, including about 27% with distant metastases.
“This is partly because of less screening, but it also reflects diagnostic delays,” according to Siegel and coauthors, who noted data show screening uptake remains low in individuals aged 45-49 (37%) and 50-54 (55%) years. The incidence of early-onset CRC increased across all racial and ethnic groups in the US, from 2% annually in Black individuals to 4% annually in Hispanic individuals between 2013 and 2022.
Aside from early-onset trends, the analysis found that tumor location trends shifted as well. Rectal cancer incidence increased in all ages combined (by 1% per year from 2018 to 2022), reversing decades of decline and now accounting for nearly one third of all CRC, compared with 27% in the mid-2000s.
The report also indicated that racial and ethnic disparities persist. Alaska Native individuals had the highest CRC incidence (80.9 per 100,000) and mortality (31.5 per 100,000) in the US, more than twofold that of White patients (35.2 and 12.9 per 100,000, respectively). Asian American, Native Hawaiian, and other Pacific Islanders had the lowest incidence (28.5 per 100,000) and mortality rates (9.2 per 100,000).
Although cancer registries like SEER are not perfect, they are “the best data we have” and overall the SEER data “very reliably represent what is going on in the US population,” May said.
The latest findings also further underscore that CRC is “worsening among younger generations and highlight the immediate need for eligible adults to begin screening at the recommended age of 45,” William Dahut, MD, ACS chief scientific officer, said in the statement.
The study had no commercial funding. The authors and May reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Colorectal cancer (CRC) in the United States continues to move in two different directions — decreasing in older adults and rising in younger adults, especially in those aged 20-49 years, according to the latest statistics from the American Cancer Society (ACS).
The ACS report, published online earlier this month, revealed that CRC incidence rates declined by 2.5% annually in adults aged ≥ 65 years but increased by 3% annually in adults aged 20-49 between 2013 and 2022 — higher than earlier estimates of 1% to 2% annual increases.
The trends are “concerning” and a “stark reminder that we’re seeing a shifting epidemiology,” said Folasade (Fola) May, MD, PhD, MPhil, director of the gastroenterology quality improvement program at UCLA Health in Los Angeles, who wasn’t involved in the analysis.
The report highlights the need for better education and symptom awareness — including bleeding, iron deficiency symptoms, and changes in bowel habits — among patients and doctors, who may not routinely consider cancer in younger adults, May explained.
“Because so many of the young people diagnosed present with advanced stage disease, early workup is critical to saving lives,” she said.
Rapidly Changing Landscape
In the United States, CRC is the third-most commonly diagnosed cancer in both men and women. CRC is also the second-leading cause of cancer-related deaths and the leading cause in adults aged < 50 years.
“After decades of progress, the risk of dying from colorectal cancer is climbing in younger generations of men and women, confirming a real uptick in disease because of something we’re doing or some other exposure,” Rebecca Siegel, MPH, senior scientific director of surveillance research at ACS and lead author of the report, said in a statement.
For the latest CRC statistics report, ACS scientists analyzed population-based registries, including the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program, and mortality data from the CDC’s National Center for Health Statistics.
The report estimated that 158,850 new CRC cases will be diagnosed in the US in 2026, including 108,860 colon cancers and 49,990 rectal cancers; an estimated 55,230 people will die from the disease.
Overall, CRC incidence declined by 0.9% annually from 2013 to 2022, driven by decreases of 2.5% per year in adults aged ≥ 65 years. During the same period, however, incidence rates rose by about 3% per year in adults aged 20-49 years and by 0.4% per year in those aged 50-64 years. CRC mortality also continued to trend downward in adults aged ≥ 65 years by > 2% per year, but mortality increased by 1% per year in adults aged < 50 years since 2004 and in adults aged 50-64 years since 2019.
Nearly half of new CRC cases (45%) now occur in adults aged < 65 years, up from 27% in 1995, illustrating a major shift toward younger age groups, the authors said. Half of early-onset cases occur in people aged 45-49 years who are now eligible for screening, and 3 of 4 early-onset CRC cases are diagnosed at an advanced stage, including about 27% with distant metastases.
“This is partly because of less screening, but it also reflects diagnostic delays,” according to Siegel and coauthors, who noted data show screening uptake remains low in individuals aged 45-49 (37%) and 50-54 (55%) years. The incidence of early-onset CRC increased across all racial and ethnic groups in the US, from 2% annually in Black individuals to 4% annually in Hispanic individuals between 2013 and 2022.
Aside from early-onset trends, the analysis found that tumor location trends shifted as well. Rectal cancer incidence increased in all ages combined (by 1% per year from 2018 to 2022), reversing decades of decline and now accounting for nearly one third of all CRC, compared with 27% in the mid-2000s.
The report also indicated that racial and ethnic disparities persist. Alaska Native individuals had the highest CRC incidence (80.9 per 100,000) and mortality (31.5 per 100,000) in the US, more than twofold that of White patients (35.2 and 12.9 per 100,000, respectively). Asian American, Native Hawaiian, and other Pacific Islanders had the lowest incidence (28.5 per 100,000) and mortality rates (9.2 per 100,000).
Although cancer registries like SEER are not perfect, they are “the best data we have” and overall the SEER data “very reliably represent what is going on in the US population,” May said.
The latest findings also further underscore that CRC is “worsening among younger generations and highlight the immediate need for eligible adults to begin screening at the recommended age of 45,” William Dahut, MD, ACS chief scientific officer, said in the statement.
The study had no commercial funding. The authors and May reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Colorectal cancer (CRC) in the United States continues to move in two different directions — decreasing in older adults and rising in younger adults, especially in those aged 20-49 years, according to the latest statistics from the American Cancer Society (ACS).
The ACS report, published online earlier this month, revealed that CRC incidence rates declined by 2.5% annually in adults aged ≥ 65 years but increased by 3% annually in adults aged 20-49 between 2013 and 2022 — higher than earlier estimates of 1% to 2% annual increases.
The trends are “concerning” and a “stark reminder that we’re seeing a shifting epidemiology,” said Folasade (Fola) May, MD, PhD, MPhil, director of the gastroenterology quality improvement program at UCLA Health in Los Angeles, who wasn’t involved in the analysis.
The report highlights the need for better education and symptom awareness — including bleeding, iron deficiency symptoms, and changes in bowel habits — among patients and doctors, who may not routinely consider cancer in younger adults, May explained.
“Because so many of the young people diagnosed present with advanced stage disease, early workup is critical to saving lives,” she said.
Rapidly Changing Landscape
In the United States, CRC is the third-most commonly diagnosed cancer in both men and women. CRC is also the second-leading cause of cancer-related deaths and the leading cause in adults aged < 50 years.
“After decades of progress, the risk of dying from colorectal cancer is climbing in younger generations of men and women, confirming a real uptick in disease because of something we’re doing or some other exposure,” Rebecca Siegel, MPH, senior scientific director of surveillance research at ACS and lead author of the report, said in a statement.
For the latest CRC statistics report, ACS scientists analyzed population-based registries, including the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program, and mortality data from the CDC’s National Center for Health Statistics.
The report estimated that 158,850 new CRC cases will be diagnosed in the US in 2026, including 108,860 colon cancers and 49,990 rectal cancers; an estimated 55,230 people will die from the disease.
Overall, CRC incidence declined by 0.9% annually from 2013 to 2022, driven by decreases of 2.5% per year in adults aged ≥ 65 years. During the same period, however, incidence rates rose by about 3% per year in adults aged 20-49 years and by 0.4% per year in those aged 50-64 years. CRC mortality also continued to trend downward in adults aged ≥ 65 years by > 2% per year, but mortality increased by 1% per year in adults aged < 50 years since 2004 and in adults aged 50-64 years since 2019.
Nearly half of new CRC cases (45%) now occur in adults aged < 65 years, up from 27% in 1995, illustrating a major shift toward younger age groups, the authors said. Half of early-onset cases occur in people aged 45-49 years who are now eligible for screening, and 3 of 4 early-onset CRC cases are diagnosed at an advanced stage, including about 27% with distant metastases.
“This is partly because of less screening, but it also reflects diagnostic delays,” according to Siegel and coauthors, who noted data show screening uptake remains low in individuals aged 45-49 (37%) and 50-54 (55%) years. The incidence of early-onset CRC increased across all racial and ethnic groups in the US, from 2% annually in Black individuals to 4% annually in Hispanic individuals between 2013 and 2022.
Aside from early-onset trends, the analysis found that tumor location trends shifted as well. Rectal cancer incidence increased in all ages combined (by 1% per year from 2018 to 2022), reversing decades of decline and now accounting for nearly one third of all CRC, compared with 27% in the mid-2000s.
The report also indicated that racial and ethnic disparities persist. Alaska Native individuals had the highest CRC incidence (80.9 per 100,000) and mortality (31.5 per 100,000) in the US, more than twofold that of White patients (35.2 and 12.9 per 100,000, respectively). Asian American, Native Hawaiian, and other Pacific Islanders had the lowest incidence (28.5 per 100,000) and mortality rates (9.2 per 100,000).
Although cancer registries like SEER are not perfect, they are “the best data we have” and overall the SEER data “very reliably represent what is going on in the US population,” May said.
The latest findings also further underscore that CRC is “worsening among younger generations and highlight the immediate need for eligible adults to begin screening at the recommended age of 45,” William Dahut, MD, ACS chief scientific officer, said in the statement.
The study had no commercial funding. The authors and May reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
'Concerning': CRC Continues to Shift Toward Younger Adults
'Concerning': CRC Continues to Shift Toward Younger Adults
FDA Grants Full Approval to Encorafenib in Metastatic CRC
FDA Grants Full Approval to Encorafenib in Metastatic CRC
The FDA has granted traditional approval to encorafenib (Braftovi, Pfizer) in combination with cetuximab (Erbitux, Eli Lilly) and fluorouracil-based chemotherapy for treatment of adults with metastatic colorectal cancer with a BRAF V600E mutation, as detected by an FDA-authorized test.
Encorafenib received accelerated approval for use with cetuximab plus mFOLFOX6 in this patient population in 2024, based on results from the BREAKWATER trial showing improved objective response rates. The conversion to full approval is supported by progression-free and overall survival outcomes.
As reported previously by Medscape Medical News, the combination of encorafenib/cetuximab/mFOLFOX6 doubled median overall survival compared with standard chemotherapy with or without bevacizumab. At a median follow-up of 22 months, overall survival was 30 months with the encorafenib regimen vs 15 months with standard chemotherapy (hazard ratio [HR], 0.49; P < .0001).
At median follow up of 16.8 months, median progression-free survival was 12.8 in the encorafenib group vs 7.1 months in the standard chemotherapy group (HR, 0.53; P < .0001).
The survival results are “unprecedented” and “practice changing” for these patients, who historically have a poor prognosis, lead investigator Elena Élez, MD, PhD, of Vall d’Hebron University Hospital in Barcelona, Spain, said in presenting the findings at the American Society of Clinical Oncology (ASCO) 2025 annual meeting.
The results were simultaneously published in The New England Journal of Medicine.
Speaking at the ASCO meeting, study discussant Andrea Sartore-Bianchi, MD, of the University of Milan, Italy, called the results “striking” and said the encorafenib combination should be considered the first-line standard of care.
As for safety, the rate of treatment-related grade 3/4 adverse events in the trial was 76% with encorafenib vs 59% with standard chemotherapy. Patients receiving encorafenib also had higher rates of anemia, arthralgia, rash, and pyrexia, but there was no substantial increase in treatment discontinuation.
The recommended encorafenib dose is 300 mg (four 75 mg capsules) once daily, in combination with cetuximab and mFOLFOX6 or in combination with cetuximab and FOLFIRI until disease progression or unacceptable toxicity, the FDA said in its approval announcement.
Prescribing information includes warnings and precautions for new primary malignancies (cutaneous and noncutaneous), tumor promotion in BRAF-wild-type tumors, cardiomyopathy, hepatotoxicity, hemorrhage, uveitis, QT prolongation, and embryo-fetal toxicity.
A version of this article first appeared on Medscape.com.
The FDA has granted traditional approval to encorafenib (Braftovi, Pfizer) in combination with cetuximab (Erbitux, Eli Lilly) and fluorouracil-based chemotherapy for treatment of adults with metastatic colorectal cancer with a BRAF V600E mutation, as detected by an FDA-authorized test.
Encorafenib received accelerated approval for use with cetuximab plus mFOLFOX6 in this patient population in 2024, based on results from the BREAKWATER trial showing improved objective response rates. The conversion to full approval is supported by progression-free and overall survival outcomes.
As reported previously by Medscape Medical News, the combination of encorafenib/cetuximab/mFOLFOX6 doubled median overall survival compared with standard chemotherapy with or without bevacizumab. At a median follow-up of 22 months, overall survival was 30 months with the encorafenib regimen vs 15 months with standard chemotherapy (hazard ratio [HR], 0.49; P < .0001).
At median follow up of 16.8 months, median progression-free survival was 12.8 in the encorafenib group vs 7.1 months in the standard chemotherapy group (HR, 0.53; P < .0001).
The survival results are “unprecedented” and “practice changing” for these patients, who historically have a poor prognosis, lead investigator Elena Élez, MD, PhD, of Vall d’Hebron University Hospital in Barcelona, Spain, said in presenting the findings at the American Society of Clinical Oncology (ASCO) 2025 annual meeting.
The results were simultaneously published in The New England Journal of Medicine.
Speaking at the ASCO meeting, study discussant Andrea Sartore-Bianchi, MD, of the University of Milan, Italy, called the results “striking” and said the encorafenib combination should be considered the first-line standard of care.
As for safety, the rate of treatment-related grade 3/4 adverse events in the trial was 76% with encorafenib vs 59% with standard chemotherapy. Patients receiving encorafenib also had higher rates of anemia, arthralgia, rash, and pyrexia, but there was no substantial increase in treatment discontinuation.
The recommended encorafenib dose is 300 mg (four 75 mg capsules) once daily, in combination with cetuximab and mFOLFOX6 or in combination with cetuximab and FOLFIRI until disease progression or unacceptable toxicity, the FDA said in its approval announcement.
Prescribing information includes warnings and precautions for new primary malignancies (cutaneous and noncutaneous), tumor promotion in BRAF-wild-type tumors, cardiomyopathy, hepatotoxicity, hemorrhage, uveitis, QT prolongation, and embryo-fetal toxicity.
A version of this article first appeared on Medscape.com.
The FDA has granted traditional approval to encorafenib (Braftovi, Pfizer) in combination with cetuximab (Erbitux, Eli Lilly) and fluorouracil-based chemotherapy for treatment of adults with metastatic colorectal cancer with a BRAF V600E mutation, as detected by an FDA-authorized test.
Encorafenib received accelerated approval for use with cetuximab plus mFOLFOX6 in this patient population in 2024, based on results from the BREAKWATER trial showing improved objective response rates. The conversion to full approval is supported by progression-free and overall survival outcomes.
As reported previously by Medscape Medical News, the combination of encorafenib/cetuximab/mFOLFOX6 doubled median overall survival compared with standard chemotherapy with or without bevacizumab. At a median follow-up of 22 months, overall survival was 30 months with the encorafenib regimen vs 15 months with standard chemotherapy (hazard ratio [HR], 0.49; P < .0001).
At median follow up of 16.8 months, median progression-free survival was 12.8 in the encorafenib group vs 7.1 months in the standard chemotherapy group (HR, 0.53; P < .0001).
The survival results are “unprecedented” and “practice changing” for these patients, who historically have a poor prognosis, lead investigator Elena Élez, MD, PhD, of Vall d’Hebron University Hospital in Barcelona, Spain, said in presenting the findings at the American Society of Clinical Oncology (ASCO) 2025 annual meeting.
The results were simultaneously published in The New England Journal of Medicine.
Speaking at the ASCO meeting, study discussant Andrea Sartore-Bianchi, MD, of the University of Milan, Italy, called the results “striking” and said the encorafenib combination should be considered the first-line standard of care.
As for safety, the rate of treatment-related grade 3/4 adverse events in the trial was 76% with encorafenib vs 59% with standard chemotherapy. Patients receiving encorafenib also had higher rates of anemia, arthralgia, rash, and pyrexia, but there was no substantial increase in treatment discontinuation.
The recommended encorafenib dose is 300 mg (four 75 mg capsules) once daily, in combination with cetuximab and mFOLFOX6 or in combination with cetuximab and FOLFIRI until disease progression or unacceptable toxicity, the FDA said in its approval announcement.
Prescribing information includes warnings and precautions for new primary malignancies (cutaneous and noncutaneous), tumor promotion in BRAF-wild-type tumors, cardiomyopathy, hepatotoxicity, hemorrhage, uveitis, QT prolongation, and embryo-fetal toxicity.
A version of this article first appeared on Medscape.com.
FDA Grants Full Approval to Encorafenib in Metastatic CRC
FDA Grants Full Approval to Encorafenib in Metastatic CRC