User login
Study finds psoriasis link to melanoma and hematologic cancers
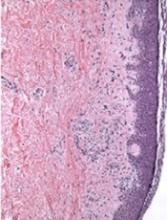
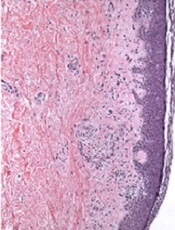
Patients with psoriasis may be at a greater risk of melanoma and hematologic cancers, compared with the general population, but treatments do not appear to increase risk, according to Shivani P. Reddy of the University of Illinois at Chicago, and associates.
In a retrospective cohort study, they identified 815,765 patients at Kaiser Permanente Southern California who had at least one medical encounter from January 2004 through December 2013. Of these patients, 8,161 (1%) met the diagnostic and inclusion criteria for psoriasis, and there were 7,167 (0.89%) cases of melanoma and 5,399 (0.66%) cases of lymphoma or leukemia.
Among the patients with psoriasis, there were 62 (0.87%) melanoma cases and 47 (0.87%) cases of lymphoma or leukemia.
Of the 109 patients with psoriasis who went on to develop melanoma or lymphoma, the time to diagnosis of melanoma or hematologic cancers was significantly less for patients with psoriasis than for patients without psoriasis (P = .01). The patients with psoriasis had a 1.53 times higher risk of developing a malignancy compared with patients without psoriasis (P less than .01) in the multivariable Cox proportional hazards model.
“There were no differences between patients with melanoma and hematologic cancer by treatment type,” which were phototherapy, tumor necrosis factor inhibitor therapy, and topical medications, they wrote.
“Our study demonstrates that the risk of hematologic cancer and melanoma is increased in patients with psoriasis over time, although this risk is not impacted by psoriasis therapies,” the researchers concluded. “Defining the risk of malignancy in these patients is important for proper workup and management.”
Read the study in the Journal of the American Academy of Dermatology (doi: 10.1016/j.jaad.2016.09.047).
Patients with psoriasis may be at a greater risk of melanoma and hematologic cancers, compared with the general population, but treatments do not appear to increase risk, according to Shivani P. Reddy of the University of Illinois at Chicago, and associates.
In a retrospective cohort study, they identified 815,765 patients at Kaiser Permanente Southern California who had at least one medical encounter from January 2004 through December 2013. Of these patients, 8,161 (1%) met the diagnostic and inclusion criteria for psoriasis, and there were 7,167 (0.89%) cases of melanoma and 5,399 (0.66%) cases of lymphoma or leukemia.
Among the patients with psoriasis, there were 62 (0.87%) melanoma cases and 47 (0.87%) cases of lymphoma or leukemia.
Of the 109 patients with psoriasis who went on to develop melanoma or lymphoma, the time to diagnosis of melanoma or hematologic cancers was significantly less for patients with psoriasis than for patients without psoriasis (P = .01). The patients with psoriasis had a 1.53 times higher risk of developing a malignancy compared with patients without psoriasis (P less than .01) in the multivariable Cox proportional hazards model.
“There were no differences between patients with melanoma and hematologic cancer by treatment type,” which were phototherapy, tumor necrosis factor inhibitor therapy, and topical medications, they wrote.
“Our study demonstrates that the risk of hematologic cancer and melanoma is increased in patients with psoriasis over time, although this risk is not impacted by psoriasis therapies,” the researchers concluded. “Defining the risk of malignancy in these patients is important for proper workup and management.”
Read the study in the Journal of the American Academy of Dermatology (doi: 10.1016/j.jaad.2016.09.047).
Patients with psoriasis may be at a greater risk of melanoma and hematologic cancers, compared with the general population, but treatments do not appear to increase risk, according to Shivani P. Reddy of the University of Illinois at Chicago, and associates.
In a retrospective cohort study, they identified 815,765 patients at Kaiser Permanente Southern California who had at least one medical encounter from January 2004 through December 2013. Of these patients, 8,161 (1%) met the diagnostic and inclusion criteria for psoriasis, and there were 7,167 (0.89%) cases of melanoma and 5,399 (0.66%) cases of lymphoma or leukemia.
Among the patients with psoriasis, there were 62 (0.87%) melanoma cases and 47 (0.87%) cases of lymphoma or leukemia.
Of the 109 patients with psoriasis who went on to develop melanoma or lymphoma, the time to diagnosis of melanoma or hematologic cancers was significantly less for patients with psoriasis than for patients without psoriasis (P = .01). The patients with psoriasis had a 1.53 times higher risk of developing a malignancy compared with patients without psoriasis (P less than .01) in the multivariable Cox proportional hazards model.
“There were no differences between patients with melanoma and hematologic cancer by treatment type,” which were phototherapy, tumor necrosis factor inhibitor therapy, and topical medications, they wrote.
“Our study demonstrates that the risk of hematologic cancer and melanoma is increased in patients with psoriasis over time, although this risk is not impacted by psoriasis therapies,” the researchers concluded. “Defining the risk of malignancy in these patients is important for proper workup and management.”
Read the study in the Journal of the American Academy of Dermatology (doi: 10.1016/j.jaad.2016.09.047).
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Waldenström macroglobulinemia panel advises on IgM paraproteinemic neuropathies
With optimal approaches still evolving for the diagnosis and management of peripheral neuropathies associated with Waldenström macroglobulinemia and other IgM paraproteinemias, new consensus recommendations from a multidisciplinary panel were recently published in the British Journal of Haematology.
Up to half of patients with IgM monoclonal gammopathies develop peripheral neuropathy, according to the 11-member panel (Br J Haematol. 2017 Mar;176[5]:728-42). The panel began deliberations at the eighth International Workshop on Waldenström Macroglobulinemia in London and was led by Shirley D’Sa, MD, of the Waldenström Clinic, Cancer Division, University College London Hospitals NHS Foundation Trust.
• Diagnostic evaluation: “The indications for invasive investigations such as cerebrospinal fluid analysis, nerve conduction tests, and sensory nerve biopsies are unclear,” according to the panelists.
When clinical examination identifies a neuropathy, neurophysiologic testing can ascertain its nature and inform additional work-up. Cerebrospinal fluid examination is not mandatory in cases of demyelinating neuropathy, but it is indicated when clinical evaluation is inconclusive and malignancy or CNS invasion is suspected.
Nerve biopsy carries substantial risk and is rarely indicated. It may be warranted when a comprehensive systemic work-up has not identified a cause and clinicians still suspect amyloid, vasculitis, or direct cellular invasion; in atypical cases not responding to treatment; or when the neuropathy is progressive and debilitating.
When it comes to imaging, “MRI of the neuraxis should be performed prior to lumbar puncture to avoid false positive meningeal enhancement,” they advised. “Prior discussion of likely sites of involvement with an experienced neuroradiologist will ensure that the correct sequences of the correct anatomical area are performed with appropriate gadolinium enhancement.”
• Clinical phenotypes and their treatment: IgM-associated neuropathies vary with respect to specific antibodies present and the likelihood that they are causally associated with the neuropathy, Dr. D’Sa and her colleagues noted. They provided a decision tree to help guide the work-up to determine the specific etiology.
“The presence of a neuropathy alone is not a justification for treatment, but steady progression with accumulating disability should prompt action,” they maintained.
Patients with antibody-negative peripheral neuropathy associated with IgM monoclonal gammopathies of undetermined significance who have mild disease and no hematologic reason for treatment can be managed with surveillance, according to the panelists.
However, immunosuppressive or immunomodulatory treatment should be considered when there is substantial or progressive disability associated with demyelination.
Patients with anti-MAG (myelin-associated glycoprotein) demyelinating neuropathy may benefit from rituximab (Rituxan). In those with more advanced disease, clinicians should consider immunosuppressive or immunomodulatory treatment instead.
Surveillance is also an option for Waldenström macroglobulinemia–associated peripheral neuropathy that is progressing slowly. When used, treatment should be tailored to severity of both systemic and neurologic disease.
• Treatment response assessment: “The optimum way to measure clinical response to treatment unknown,” Dr. D’Sa and her fellow panelists noted. A variety of measures of muscle strength, sensory function, and disability are used.
“The I-RODS [Inflammatory Rasch-Built Overall Disability Scale] more often captures clinically meaningful changes over time, with a greater magnitude of change, compared with the INCAT-ONLS [Inflammatory Neuropathy Cause and Treatment–Overall Neuropathy Limitation Scale] disability scale and its use is therefore suggested in future trials involving patients with inflammatory neuropathies,” they wrote.
• Model of care: Management of patients with IgM-associated neuropathies requires multidisciplinary care with good collaboration to optimize patient outcomes, the consensus panel said.
“A suggested model of care is a combined neurological and hematological clinic, in which patients are seen jointly by a specialist neurologist and hematologist and a decision can be made about the sequence of investigations, interventions, and the formulation of a treatment plan,” they proposed. “Appropriate and timely referral to physical, occupational, and orthotic professionals is recommended in order to maximize safety and function.”
• Future perspectives: “There is much to be done to improve outcomes for patients with IgM and [Waldenström macroglobulinemia]-associated peripheral neuropathies,” the panelists concluded.
Key areas of focus are “early recognition of the problem, appropriate causal attribution achieved through sensitive diagnostics that are not overly invasive, timely therapeutic intervention with effective and nonneurotoxic therapies, achievement of an appropriate degree of clonal reduction for optimum clinical outcomes, and the use of reproducible and readily applicable tools to measure outcomes.”
Dr. D’Sa disclosed that she receives honoraria from Janssen.
With optimal approaches still evolving for the diagnosis and management of peripheral neuropathies associated with Waldenström macroglobulinemia and other IgM paraproteinemias, new consensus recommendations from a multidisciplinary panel were recently published in the British Journal of Haematology.
Up to half of patients with IgM monoclonal gammopathies develop peripheral neuropathy, according to the 11-member panel (Br J Haematol. 2017 Mar;176[5]:728-42). The panel began deliberations at the eighth International Workshop on Waldenström Macroglobulinemia in London and was led by Shirley D’Sa, MD, of the Waldenström Clinic, Cancer Division, University College London Hospitals NHS Foundation Trust.
• Diagnostic evaluation: “The indications for invasive investigations such as cerebrospinal fluid analysis, nerve conduction tests, and sensory nerve biopsies are unclear,” according to the panelists.
When clinical examination identifies a neuropathy, neurophysiologic testing can ascertain its nature and inform additional work-up. Cerebrospinal fluid examination is not mandatory in cases of demyelinating neuropathy, but it is indicated when clinical evaluation is inconclusive and malignancy or CNS invasion is suspected.
Nerve biopsy carries substantial risk and is rarely indicated. It may be warranted when a comprehensive systemic work-up has not identified a cause and clinicians still suspect amyloid, vasculitis, or direct cellular invasion; in atypical cases not responding to treatment; or when the neuropathy is progressive and debilitating.
When it comes to imaging, “MRI of the neuraxis should be performed prior to lumbar puncture to avoid false positive meningeal enhancement,” they advised. “Prior discussion of likely sites of involvement with an experienced neuroradiologist will ensure that the correct sequences of the correct anatomical area are performed with appropriate gadolinium enhancement.”
• Clinical phenotypes and their treatment: IgM-associated neuropathies vary with respect to specific antibodies present and the likelihood that they are causally associated with the neuropathy, Dr. D’Sa and her colleagues noted. They provided a decision tree to help guide the work-up to determine the specific etiology.
“The presence of a neuropathy alone is not a justification for treatment, but steady progression with accumulating disability should prompt action,” they maintained.
Patients with antibody-negative peripheral neuropathy associated with IgM monoclonal gammopathies of undetermined significance who have mild disease and no hematologic reason for treatment can be managed with surveillance, according to the panelists.
However, immunosuppressive or immunomodulatory treatment should be considered when there is substantial or progressive disability associated with demyelination.
Patients with anti-MAG (myelin-associated glycoprotein) demyelinating neuropathy may benefit from rituximab (Rituxan). In those with more advanced disease, clinicians should consider immunosuppressive or immunomodulatory treatment instead.
Surveillance is also an option for Waldenström macroglobulinemia–associated peripheral neuropathy that is progressing slowly. When used, treatment should be tailored to severity of both systemic and neurologic disease.
• Treatment response assessment: “The optimum way to measure clinical response to treatment unknown,” Dr. D’Sa and her fellow panelists noted. A variety of measures of muscle strength, sensory function, and disability are used.
“The I-RODS [Inflammatory Rasch-Built Overall Disability Scale] more often captures clinically meaningful changes over time, with a greater magnitude of change, compared with the INCAT-ONLS [Inflammatory Neuropathy Cause and Treatment–Overall Neuropathy Limitation Scale] disability scale and its use is therefore suggested in future trials involving patients with inflammatory neuropathies,” they wrote.
• Model of care: Management of patients with IgM-associated neuropathies requires multidisciplinary care with good collaboration to optimize patient outcomes, the consensus panel said.
“A suggested model of care is a combined neurological and hematological clinic, in which patients are seen jointly by a specialist neurologist and hematologist and a decision can be made about the sequence of investigations, interventions, and the formulation of a treatment plan,” they proposed. “Appropriate and timely referral to physical, occupational, and orthotic professionals is recommended in order to maximize safety and function.”
• Future perspectives: “There is much to be done to improve outcomes for patients with IgM and [Waldenström macroglobulinemia]-associated peripheral neuropathies,” the panelists concluded.
Key areas of focus are “early recognition of the problem, appropriate causal attribution achieved through sensitive diagnostics that are not overly invasive, timely therapeutic intervention with effective and nonneurotoxic therapies, achievement of an appropriate degree of clonal reduction for optimum clinical outcomes, and the use of reproducible and readily applicable tools to measure outcomes.”
Dr. D’Sa disclosed that she receives honoraria from Janssen.
With optimal approaches still evolving for the diagnosis and management of peripheral neuropathies associated with Waldenström macroglobulinemia and other IgM paraproteinemias, new consensus recommendations from a multidisciplinary panel were recently published in the British Journal of Haematology.
Up to half of patients with IgM monoclonal gammopathies develop peripheral neuropathy, according to the 11-member panel (Br J Haematol. 2017 Mar;176[5]:728-42). The panel began deliberations at the eighth International Workshop on Waldenström Macroglobulinemia in London and was led by Shirley D’Sa, MD, of the Waldenström Clinic, Cancer Division, University College London Hospitals NHS Foundation Trust.
• Diagnostic evaluation: “The indications for invasive investigations such as cerebrospinal fluid analysis, nerve conduction tests, and sensory nerve biopsies are unclear,” according to the panelists.
When clinical examination identifies a neuropathy, neurophysiologic testing can ascertain its nature and inform additional work-up. Cerebrospinal fluid examination is not mandatory in cases of demyelinating neuropathy, but it is indicated when clinical evaluation is inconclusive and malignancy or CNS invasion is suspected.
Nerve biopsy carries substantial risk and is rarely indicated. It may be warranted when a comprehensive systemic work-up has not identified a cause and clinicians still suspect amyloid, vasculitis, or direct cellular invasion; in atypical cases not responding to treatment; or when the neuropathy is progressive and debilitating.
When it comes to imaging, “MRI of the neuraxis should be performed prior to lumbar puncture to avoid false positive meningeal enhancement,” they advised. “Prior discussion of likely sites of involvement with an experienced neuroradiologist will ensure that the correct sequences of the correct anatomical area are performed with appropriate gadolinium enhancement.”
• Clinical phenotypes and their treatment: IgM-associated neuropathies vary with respect to specific antibodies present and the likelihood that they are causally associated with the neuropathy, Dr. D’Sa and her colleagues noted. They provided a decision tree to help guide the work-up to determine the specific etiology.
“The presence of a neuropathy alone is not a justification for treatment, but steady progression with accumulating disability should prompt action,” they maintained.
Patients with antibody-negative peripheral neuropathy associated with IgM monoclonal gammopathies of undetermined significance who have mild disease and no hematologic reason for treatment can be managed with surveillance, according to the panelists.
However, immunosuppressive or immunomodulatory treatment should be considered when there is substantial or progressive disability associated with demyelination.
Patients with anti-MAG (myelin-associated glycoprotein) demyelinating neuropathy may benefit from rituximab (Rituxan). In those with more advanced disease, clinicians should consider immunosuppressive or immunomodulatory treatment instead.
Surveillance is also an option for Waldenström macroglobulinemia–associated peripheral neuropathy that is progressing slowly. When used, treatment should be tailored to severity of both systemic and neurologic disease.
• Treatment response assessment: “The optimum way to measure clinical response to treatment unknown,” Dr. D’Sa and her fellow panelists noted. A variety of measures of muscle strength, sensory function, and disability are used.
“The I-RODS [Inflammatory Rasch-Built Overall Disability Scale] more often captures clinically meaningful changes over time, with a greater magnitude of change, compared with the INCAT-ONLS [Inflammatory Neuropathy Cause and Treatment–Overall Neuropathy Limitation Scale] disability scale and its use is therefore suggested in future trials involving patients with inflammatory neuropathies,” they wrote.
• Model of care: Management of patients with IgM-associated neuropathies requires multidisciplinary care with good collaboration to optimize patient outcomes, the consensus panel said.
“A suggested model of care is a combined neurological and hematological clinic, in which patients are seen jointly by a specialist neurologist and hematologist and a decision can be made about the sequence of investigations, interventions, and the formulation of a treatment plan,” they proposed. “Appropriate and timely referral to physical, occupational, and orthotic professionals is recommended in order to maximize safety and function.”
• Future perspectives: “There is much to be done to improve outcomes for patients with IgM and [Waldenström macroglobulinemia]-associated peripheral neuropathies,” the panelists concluded.
Key areas of focus are “early recognition of the problem, appropriate causal attribution achieved through sensitive diagnostics that are not overly invasive, timely therapeutic intervention with effective and nonneurotoxic therapies, achievement of an appropriate degree of clonal reduction for optimum clinical outcomes, and the use of reproducible and readily applicable tools to measure outcomes.”
Dr. D’Sa disclosed that she receives honoraria from Janssen.
FROM THE BRITISH JOURNAL OF HAEMATOLOGY
Key clinical point:
Major finding: The indications for invasive testing and definitive answers about when and how to treat peripheral neuropathies due to Waldenström macroglobulinemia and other IgM paraproteinemias are unclear.
Data source: Recommendations from the eighth International Workshop on Waldenström Macroglobulinemia (IWWM-8) consensus panel.
Disclosures: Dr. D’Sa disclosed that she receives honoraria from Janssen.
Method could prevent GVHD while preserving GVL effect
Researchers believe they have found a way to prevent graft-versus-host disease (GVHD) after hematopoietic stem cell transplant (HSCT) while preserving a strong graft-versus-leukemia (GVL) effect.
In experiments with mice, the team found that temporary in vivo depletion of CD4+ T cells soon after HSCT prevented GVHD without inhibiting GVL effects.
The depletion of CD4+ cells essentially caused CD8+ cells to become exhausted in their quest to destroy normal tissue but strengthened in their fight against leukemia, which meant the CD8+ cells could eliminate leukemic cells without causing GVHD.
Defu Zeng, MD, of City of Hope in Duarte, California, and his colleagues recounted these findings in the Journal of Clinical Investigation.
The researchers were able to achieve temporary in vivo depletion of CD4+ T cells by injecting mice with an anti-CD4 monoclonal antibody (mAb).
The team found that a single injection of the mAb given immediately after HSCT prevented acute but not chronic GVHD. Three injections of the mAb—given on days 0, 14, and 28—prevented both types of GVHD.
The researchers said their results suggest GVHD is more effectively prevented by temporary in vivo depletion of donor CD4+ T cells early after HSCT than by ex vivo depletion of donor CD4+ T cells.
This is because treatment with an anti-CD4 mAb temporarily depletes both the injected mature CD4+ T cells and the CD4+ T cells generated de novo from the marrow progenitors early after HSCT.
In explaining the mechanism behind their observations, the researchers noted that the interaction between PD-L1 and CD80 has been shown to exacerbate GVHD, but costimulation of CD80 and PD-1 ameliorates GVHD.
In their experiments, the team found that depleting CD4+ T cells increased serum IFN-γ and reduced IL-2 concentrations. And this led to upregulation of PD-L1 expression by recipient tissues and donor CD8+ T cells.
The researchers said that, in GVHD target tissues, the interactions of PD-L1 and PD-1 on donor CD8+ T cells caused anergy, exhaustion, and apoptosis. These effects prevented the development of GVHD.
On the other hand, in lymphoid tissues, the interactions of PD-L1 and CD80 augmented CD8+ T-cell expansion without increasing anergy, exhaustion, or apoptosis. This allowed for the preservation of GVL effects.
“If successfully translated into clinical application, this [CD4+ T-cell-depleting] regimen may represent one of the novel approaches that allow strong GVL effects without causing GVHD,” Dr Zeng said.
“This kind of regimen has the potential to promote widespread application of allogenic [HSCT] as a curative therapy for hematological malignancies.” ![]()
Researchers believe they have found a way to prevent graft-versus-host disease (GVHD) after hematopoietic stem cell transplant (HSCT) while preserving a strong graft-versus-leukemia (GVL) effect.
In experiments with mice, the team found that temporary in vivo depletion of CD4+ T cells soon after HSCT prevented GVHD without inhibiting GVL effects.
The depletion of CD4+ cells essentially caused CD8+ cells to become exhausted in their quest to destroy normal tissue but strengthened in their fight against leukemia, which meant the CD8+ cells could eliminate leukemic cells without causing GVHD.
Defu Zeng, MD, of City of Hope in Duarte, California, and his colleagues recounted these findings in the Journal of Clinical Investigation.
The researchers were able to achieve temporary in vivo depletion of CD4+ T cells by injecting mice with an anti-CD4 monoclonal antibody (mAb).
The team found that a single injection of the mAb given immediately after HSCT prevented acute but not chronic GVHD. Three injections of the mAb—given on days 0, 14, and 28—prevented both types of GVHD.
The researchers said their results suggest GVHD is more effectively prevented by temporary in vivo depletion of donor CD4+ T cells early after HSCT than by ex vivo depletion of donor CD4+ T cells.
This is because treatment with an anti-CD4 mAb temporarily depletes both the injected mature CD4+ T cells and the CD4+ T cells generated de novo from the marrow progenitors early after HSCT.
In explaining the mechanism behind their observations, the researchers noted that the interaction between PD-L1 and CD80 has been shown to exacerbate GVHD, but costimulation of CD80 and PD-1 ameliorates GVHD.
In their experiments, the team found that depleting CD4+ T cells increased serum IFN-γ and reduced IL-2 concentrations. And this led to upregulation of PD-L1 expression by recipient tissues and donor CD8+ T cells.
The researchers said that, in GVHD target tissues, the interactions of PD-L1 and PD-1 on donor CD8+ T cells caused anergy, exhaustion, and apoptosis. These effects prevented the development of GVHD.
On the other hand, in lymphoid tissues, the interactions of PD-L1 and CD80 augmented CD8+ T-cell expansion without increasing anergy, exhaustion, or apoptosis. This allowed for the preservation of GVL effects.
“If successfully translated into clinical application, this [CD4+ T-cell-depleting] regimen may represent one of the novel approaches that allow strong GVL effects without causing GVHD,” Dr Zeng said.
“This kind of regimen has the potential to promote widespread application of allogenic [HSCT] as a curative therapy for hematological malignancies.” ![]()
Researchers believe they have found a way to prevent graft-versus-host disease (GVHD) after hematopoietic stem cell transplant (HSCT) while preserving a strong graft-versus-leukemia (GVL) effect.
In experiments with mice, the team found that temporary in vivo depletion of CD4+ T cells soon after HSCT prevented GVHD without inhibiting GVL effects.
The depletion of CD4+ cells essentially caused CD8+ cells to become exhausted in their quest to destroy normal tissue but strengthened in their fight against leukemia, which meant the CD8+ cells could eliminate leukemic cells without causing GVHD.
Defu Zeng, MD, of City of Hope in Duarte, California, and his colleagues recounted these findings in the Journal of Clinical Investigation.
The researchers were able to achieve temporary in vivo depletion of CD4+ T cells by injecting mice with an anti-CD4 monoclonal antibody (mAb).
The team found that a single injection of the mAb given immediately after HSCT prevented acute but not chronic GVHD. Three injections of the mAb—given on days 0, 14, and 28—prevented both types of GVHD.
The researchers said their results suggest GVHD is more effectively prevented by temporary in vivo depletion of donor CD4+ T cells early after HSCT than by ex vivo depletion of donor CD4+ T cells.
This is because treatment with an anti-CD4 mAb temporarily depletes both the injected mature CD4+ T cells and the CD4+ T cells generated de novo from the marrow progenitors early after HSCT.
In explaining the mechanism behind their observations, the researchers noted that the interaction between PD-L1 and CD80 has been shown to exacerbate GVHD, but costimulation of CD80 and PD-1 ameliorates GVHD.
In their experiments, the team found that depleting CD4+ T cells increased serum IFN-γ and reduced IL-2 concentrations. And this led to upregulation of PD-L1 expression by recipient tissues and donor CD8+ T cells.
The researchers said that, in GVHD target tissues, the interactions of PD-L1 and PD-1 on donor CD8+ T cells caused anergy, exhaustion, and apoptosis. These effects prevented the development of GVHD.
On the other hand, in lymphoid tissues, the interactions of PD-L1 and CD80 augmented CD8+ T-cell expansion without increasing anergy, exhaustion, or apoptosis. This allowed for the preservation of GVL effects.
“If successfully translated into clinical application, this [CD4+ T-cell-depleting] regimen may represent one of the novel approaches that allow strong GVL effects without causing GVHD,” Dr Zeng said.
“This kind of regimen has the potential to promote widespread application of allogenic [HSCT] as a curative therapy for hematological malignancies.” ![]()
Study correlates genetic mutations with AML subgroups
A large study of adults with acute myeloid leukemia (AML) correlates cancer-related genetic mutations with subtypes of AML defined by the presence of specific cytogenetic abnormalities.
Researchers combined the cytogenetic abnormalities that define each of 34 AML subgroups with the mutation status of 80 cancer-related genes to produce an “oncoprint,” a tabular summary of the mutations associated with each cytogenetic group.
This allowed the researchers to identify genetic differences among the 34 subgroups and unexpected associations between the AML subsets and specific genetic mutations as well as gene functional groups.
The researchers believe their findings, published in Leukemia, might help guide mutation testing and treatment decisions in the future.
The study involved 1603 newly diagnosed adult AML patients who were treated on Cancer and Leukemia Group B/Alliance for Clinical Trials in Oncology trials in multiple centers across the US. Most patients (n=1080) were under age 60.
The researchers classified the patients into 34 karyotype subgroups within 5 major cytogenetic groups—cytogenetically normal (CN) AML, complex karyotype (CK) AML, core-binding factor (CBF) AML, AML with balanced translocations or inversions other than those associated with CBF-AML, and AML with unbalanced chromosomal abnormalities detected in non-complex karyotypes.
The team sequenced pretreatment bone marrow or peripheral blood samples from each patient to learn the mutational status of 80 cancer- and leukemia-related genes.
The mutations were assigned to 1 of 9 categories based on the gene’s biological function—for example, methylation-related, cohesin complex, chromatin remodeling, and tumor-suppressor genes.
The researchers identified 4390 mutations, with a median of 3 mutations per patient.
Overall, the most frequently mutated genes that contributed to AML in this cohort belonged to the methylation group.
The researchers observed a high incidence of mutations in methylation-related genes in patients with CN-AML, CK-AML, or unbalanced chromosomal abnormalities.
In contrast, mutations in methylation-related genes were almost absent in CBF-AML patients and were rather rare in patients with non-CBF-AML-related balanced translocations or inversions.
Mutations in spliceosome genes were frequent in patients with unbalanced chromosomal abnormalities, especially those with sole trisomy of chromosomes 4, 8, 11, 13, or 21.
“Our study summarizes cytogenetic and mutational information of 1603 AML patients in a single image,” said Ann-Kathrin Eisfeld, MD, of The Ohio State University Comprehensive Cancer Center in Columbus.
“The identification of key mutational features of each subgroup may help us to better understand the pathogenesis of the different AML types and provides a wealth of information for ongoing and future research.” ![]()
A large study of adults with acute myeloid leukemia (AML) correlates cancer-related genetic mutations with subtypes of AML defined by the presence of specific cytogenetic abnormalities.
Researchers combined the cytogenetic abnormalities that define each of 34 AML subgroups with the mutation status of 80 cancer-related genes to produce an “oncoprint,” a tabular summary of the mutations associated with each cytogenetic group.
This allowed the researchers to identify genetic differences among the 34 subgroups and unexpected associations between the AML subsets and specific genetic mutations as well as gene functional groups.
The researchers believe their findings, published in Leukemia, might help guide mutation testing and treatment decisions in the future.
The study involved 1603 newly diagnosed adult AML patients who were treated on Cancer and Leukemia Group B/Alliance for Clinical Trials in Oncology trials in multiple centers across the US. Most patients (n=1080) were under age 60.
The researchers classified the patients into 34 karyotype subgroups within 5 major cytogenetic groups—cytogenetically normal (CN) AML, complex karyotype (CK) AML, core-binding factor (CBF) AML, AML with balanced translocations or inversions other than those associated with CBF-AML, and AML with unbalanced chromosomal abnormalities detected in non-complex karyotypes.
The team sequenced pretreatment bone marrow or peripheral blood samples from each patient to learn the mutational status of 80 cancer- and leukemia-related genes.
The mutations were assigned to 1 of 9 categories based on the gene’s biological function—for example, methylation-related, cohesin complex, chromatin remodeling, and tumor-suppressor genes.
The researchers identified 4390 mutations, with a median of 3 mutations per patient.
Overall, the most frequently mutated genes that contributed to AML in this cohort belonged to the methylation group.
The researchers observed a high incidence of mutations in methylation-related genes in patients with CN-AML, CK-AML, or unbalanced chromosomal abnormalities.
In contrast, mutations in methylation-related genes were almost absent in CBF-AML patients and were rather rare in patients with non-CBF-AML-related balanced translocations or inversions.
Mutations in spliceosome genes were frequent in patients with unbalanced chromosomal abnormalities, especially those with sole trisomy of chromosomes 4, 8, 11, 13, or 21.
“Our study summarizes cytogenetic and mutational information of 1603 AML patients in a single image,” said Ann-Kathrin Eisfeld, MD, of The Ohio State University Comprehensive Cancer Center in Columbus.
“The identification of key mutational features of each subgroup may help us to better understand the pathogenesis of the different AML types and provides a wealth of information for ongoing and future research.” ![]()
A large study of adults with acute myeloid leukemia (AML) correlates cancer-related genetic mutations with subtypes of AML defined by the presence of specific cytogenetic abnormalities.
Researchers combined the cytogenetic abnormalities that define each of 34 AML subgroups with the mutation status of 80 cancer-related genes to produce an “oncoprint,” a tabular summary of the mutations associated with each cytogenetic group.
This allowed the researchers to identify genetic differences among the 34 subgroups and unexpected associations between the AML subsets and specific genetic mutations as well as gene functional groups.
The researchers believe their findings, published in Leukemia, might help guide mutation testing and treatment decisions in the future.
The study involved 1603 newly diagnosed adult AML patients who were treated on Cancer and Leukemia Group B/Alliance for Clinical Trials in Oncology trials in multiple centers across the US. Most patients (n=1080) were under age 60.
The researchers classified the patients into 34 karyotype subgroups within 5 major cytogenetic groups—cytogenetically normal (CN) AML, complex karyotype (CK) AML, core-binding factor (CBF) AML, AML with balanced translocations or inversions other than those associated with CBF-AML, and AML with unbalanced chromosomal abnormalities detected in non-complex karyotypes.
The team sequenced pretreatment bone marrow or peripheral blood samples from each patient to learn the mutational status of 80 cancer- and leukemia-related genes.
The mutations were assigned to 1 of 9 categories based on the gene’s biological function—for example, methylation-related, cohesin complex, chromatin remodeling, and tumor-suppressor genes.
The researchers identified 4390 mutations, with a median of 3 mutations per patient.
Overall, the most frequently mutated genes that contributed to AML in this cohort belonged to the methylation group.
The researchers observed a high incidence of mutations in methylation-related genes in patients with CN-AML, CK-AML, or unbalanced chromosomal abnormalities.
In contrast, mutations in methylation-related genes were almost absent in CBF-AML patients and were rather rare in patients with non-CBF-AML-related balanced translocations or inversions.
Mutations in spliceosome genes were frequent in patients with unbalanced chromosomal abnormalities, especially those with sole trisomy of chromosomes 4, 8, 11, 13, or 21.
“Our study summarizes cytogenetic and mutational information of 1603 AML patients in a single image,” said Ann-Kathrin Eisfeld, MD, of The Ohio State University Comprehensive Cancer Center in Columbus.
“The identification of key mutational features of each subgroup may help us to better understand the pathogenesis of the different AML types and provides a wealth of information for ongoing and future research.” ![]()
Nanoparticles allow for creation of CAR T cells in vivo
Researchers say they have developed biodegradable nanoparticles that can be used to genetically reprogram T cells while they are still in the body.
The nanoparticles were able to program T cells with genes encoding leukemia-specific chimeric antigen receptors (CARs).
The resulting CAR T cells were able to eliminate leukemia or slow the progression of disease in a mouse model of B-cell acute lymphoblastic leukemia (B-ALL).
Researchers reported these results in Nature Nanotechnology.
“Our technology is the first that we know of to quickly program tumor-recognizing capabilities into T cells without extracting them for laboratory manipulation,” said Matthias Stephan, MD, PhD, of Fred Hutchinson Cancer Research Center in Seattle, Washington.
“The reprogrammed cells begin to work within 24 to 48 hours and continue to produce these receptors for weeks. This suggests that our technology has the potential to allow the immune system to quickly mount a strong enough response to destroy cancerous cells before the disease becomes fatal.”
Dr Stephan and his colleagues designed their nanoparticles to carry genes that encode for CARs intended to target and eliminate B-ALL. The nanoparticles are coated with ligands that make them seek out and bind to T cells.
When a nanoparticle binds to a T cell, the cell engulfs the particle. The nanoparticle then travels to the cell’s nucleus and dissolves.
The CAR genes integrate into chromosomes housed in the nucleus, making it possible for the T cells to begin decoding the new genes and producing CARs within 1 or 2 days.
Once they determined their CAR-carrying nanoparticles reprogrammed a noticeable percentage of T cells, the researchers tested the T cells’ efficacy in a mouse model of B-ALL.
The team infused the nanoparticles into 10 mice and found the treatment eradicated tumors in 7 of the animals. The other 3 mice “showed substantial regression” of leukemia, the researchers said.
On average, mice that received CAR-carrying nanoparticles had a 58-day improvement in survival compared to control mice.
Mice that received the nanoparticles also had “dramatically reduced” B-cell numbers in their spleens. The researchers noted that this is consistent with the reversible B-cell aplasia observed in patients who receive conventional CD19 CAR T-cell therapy.
Dr Stephan and his colleagues also tested conventional CAR T-cell therapy in the B-ALL mouse model. The mice received cyclophosphamide followed by CAR T cells created ex vivo.
These mice had significantly better survival than controls, but their survival was comparable to that of the mice that received the CAR-carrying nanoparticles.
Although these nanoparticles are several steps away from the clinic, Dr Stephan said he imagines a future in which nanoparticles transform cell-based immunotherapies into easily administered, off-the-shelf treatments that are available anywhere.
“I’ve never had cancer, but if I did get a cancer diagnosis, I would want to start treatment right away,” Dr Stephan said. “I want to make cellular immunotherapy a treatment option the day of diagnosis and have it able to be done in an outpatient setting near where people live.” ![]()
Researchers say they have developed biodegradable nanoparticles that can be used to genetically reprogram T cells while they are still in the body.
The nanoparticles were able to program T cells with genes encoding leukemia-specific chimeric antigen receptors (CARs).
The resulting CAR T cells were able to eliminate leukemia or slow the progression of disease in a mouse model of B-cell acute lymphoblastic leukemia (B-ALL).
Researchers reported these results in Nature Nanotechnology.
“Our technology is the first that we know of to quickly program tumor-recognizing capabilities into T cells without extracting them for laboratory manipulation,” said Matthias Stephan, MD, PhD, of Fred Hutchinson Cancer Research Center in Seattle, Washington.
“The reprogrammed cells begin to work within 24 to 48 hours and continue to produce these receptors for weeks. This suggests that our technology has the potential to allow the immune system to quickly mount a strong enough response to destroy cancerous cells before the disease becomes fatal.”
Dr Stephan and his colleagues designed their nanoparticles to carry genes that encode for CARs intended to target and eliminate B-ALL. The nanoparticles are coated with ligands that make them seek out and bind to T cells.
When a nanoparticle binds to a T cell, the cell engulfs the particle. The nanoparticle then travels to the cell’s nucleus and dissolves.
The CAR genes integrate into chromosomes housed in the nucleus, making it possible for the T cells to begin decoding the new genes and producing CARs within 1 or 2 days.
Once they determined their CAR-carrying nanoparticles reprogrammed a noticeable percentage of T cells, the researchers tested the T cells’ efficacy in a mouse model of B-ALL.
The team infused the nanoparticles into 10 mice and found the treatment eradicated tumors in 7 of the animals. The other 3 mice “showed substantial regression” of leukemia, the researchers said.
On average, mice that received CAR-carrying nanoparticles had a 58-day improvement in survival compared to control mice.
Mice that received the nanoparticles also had “dramatically reduced” B-cell numbers in their spleens. The researchers noted that this is consistent with the reversible B-cell aplasia observed in patients who receive conventional CD19 CAR T-cell therapy.
Dr Stephan and his colleagues also tested conventional CAR T-cell therapy in the B-ALL mouse model. The mice received cyclophosphamide followed by CAR T cells created ex vivo.
These mice had significantly better survival than controls, but their survival was comparable to that of the mice that received the CAR-carrying nanoparticles.
Although these nanoparticles are several steps away from the clinic, Dr Stephan said he imagines a future in which nanoparticles transform cell-based immunotherapies into easily administered, off-the-shelf treatments that are available anywhere.
“I’ve never had cancer, but if I did get a cancer diagnosis, I would want to start treatment right away,” Dr Stephan said. “I want to make cellular immunotherapy a treatment option the day of diagnosis and have it able to be done in an outpatient setting near where people live.” ![]()
Researchers say they have developed biodegradable nanoparticles that can be used to genetically reprogram T cells while they are still in the body.
The nanoparticles were able to program T cells with genes encoding leukemia-specific chimeric antigen receptors (CARs).
The resulting CAR T cells were able to eliminate leukemia or slow the progression of disease in a mouse model of B-cell acute lymphoblastic leukemia (B-ALL).
Researchers reported these results in Nature Nanotechnology.
“Our technology is the first that we know of to quickly program tumor-recognizing capabilities into T cells without extracting them for laboratory manipulation,” said Matthias Stephan, MD, PhD, of Fred Hutchinson Cancer Research Center in Seattle, Washington.
“The reprogrammed cells begin to work within 24 to 48 hours and continue to produce these receptors for weeks. This suggests that our technology has the potential to allow the immune system to quickly mount a strong enough response to destroy cancerous cells before the disease becomes fatal.”
Dr Stephan and his colleagues designed their nanoparticles to carry genes that encode for CARs intended to target and eliminate B-ALL. The nanoparticles are coated with ligands that make them seek out and bind to T cells.
When a nanoparticle binds to a T cell, the cell engulfs the particle. The nanoparticle then travels to the cell’s nucleus and dissolves.
The CAR genes integrate into chromosomes housed in the nucleus, making it possible for the T cells to begin decoding the new genes and producing CARs within 1 or 2 days.
Once they determined their CAR-carrying nanoparticles reprogrammed a noticeable percentage of T cells, the researchers tested the T cells’ efficacy in a mouse model of B-ALL.
The team infused the nanoparticles into 10 mice and found the treatment eradicated tumors in 7 of the animals. The other 3 mice “showed substantial regression” of leukemia, the researchers said.
On average, mice that received CAR-carrying nanoparticles had a 58-day improvement in survival compared to control mice.
Mice that received the nanoparticles also had “dramatically reduced” B-cell numbers in their spleens. The researchers noted that this is consistent with the reversible B-cell aplasia observed in patients who receive conventional CD19 CAR T-cell therapy.
Dr Stephan and his colleagues also tested conventional CAR T-cell therapy in the B-ALL mouse model. The mice received cyclophosphamide followed by CAR T cells created ex vivo.
These mice had significantly better survival than controls, but their survival was comparable to that of the mice that received the CAR-carrying nanoparticles.
Although these nanoparticles are several steps away from the clinic, Dr Stephan said he imagines a future in which nanoparticles transform cell-based immunotherapies into easily administered, off-the-shelf treatments that are available anywhere.
“I’ve never had cancer, but if I did get a cancer diagnosis, I would want to start treatment right away,” Dr Stephan said. “I want to make cellular immunotherapy a treatment option the day of diagnosis and have it able to be done in an outpatient setting near where people live.” ![]()
Inhibitor exhibits activity against hematologic malignancies
A dual kinase inhibitor is active against a range of hematologic malignancies, according to preclinical research.
Investigators found that ASN002, a SYK/JAK inhibitor, exhibited “potent” antiproliferative activity in leukemia, lymphoma, and myeloma cell lines.
ASN002 also inhibited tumor growth in mouse models of these malignancies and proved active against ibrutinib-resistant diffuse large B-cell lymphoma (DLBCL).
The investigators presented these results at the AACR Annual Meeting 2017 (abstract 4204).
The work was conducted by employees of Asana BioSciences, the company developing ASN002.
The investigators tested ASN002 in 178 cell lines and found the drug exhibited “strong antiproliferative activity” in a range of hematologic cancer cell lines, including:
- Leukemia—HEL31, HL60, Jurkat, MOLM-13, and MOLM-16
- Lymphoma—KARPAS-299, OCI-LY10, OCI-LY-19, Pfeiffer, Raji, Ramos, SU-DHL-1, SU-DHL-6, and SU-DHL-10
- Myeloma—H929, JJN3, OPM2, and U266.
In addition, ASN002 was active against ibrutinib-resistant clones derived from the DLBCL cell line SU-DHL-6.
In a SU-DHL-6 xenograft model, the combination of ASN002 and ibrutinib was more effective than either compound alone.
The investigators also found that ASN002 alone demonstrated “strong tumor growth inhibition” in mouse models of DLBCL (Pfeiffer and SU-DHL-6), myeloma (H929), and erythroleukemia (HEL).
The team pointed out that ASN002 is currently under investigation in a phase 1/2 study of patients with B-cell lymphomas (DLBCL, mantle cell lymphoma, and follicular lymphoma) as well as solid tumors.
The investigators said that, to date, ASN002 has been well tolerated and has shown encouraging early evidence of clinical activity and symptomatic benefit in the lymphoma patients. ![]()
A dual kinase inhibitor is active against a range of hematologic malignancies, according to preclinical research.
Investigators found that ASN002, a SYK/JAK inhibitor, exhibited “potent” antiproliferative activity in leukemia, lymphoma, and myeloma cell lines.
ASN002 also inhibited tumor growth in mouse models of these malignancies and proved active against ibrutinib-resistant diffuse large B-cell lymphoma (DLBCL).
The investigators presented these results at the AACR Annual Meeting 2017 (abstract 4204).
The work was conducted by employees of Asana BioSciences, the company developing ASN002.
The investigators tested ASN002 in 178 cell lines and found the drug exhibited “strong antiproliferative activity” in a range of hematologic cancer cell lines, including:
- Leukemia—HEL31, HL60, Jurkat, MOLM-13, and MOLM-16
- Lymphoma—KARPAS-299, OCI-LY10, OCI-LY-19, Pfeiffer, Raji, Ramos, SU-DHL-1, SU-DHL-6, and SU-DHL-10
- Myeloma—H929, JJN3, OPM2, and U266.
In addition, ASN002 was active against ibrutinib-resistant clones derived from the DLBCL cell line SU-DHL-6.
In a SU-DHL-6 xenograft model, the combination of ASN002 and ibrutinib was more effective than either compound alone.
The investigators also found that ASN002 alone demonstrated “strong tumor growth inhibition” in mouse models of DLBCL (Pfeiffer and SU-DHL-6), myeloma (H929), and erythroleukemia (HEL).
The team pointed out that ASN002 is currently under investigation in a phase 1/2 study of patients with B-cell lymphomas (DLBCL, mantle cell lymphoma, and follicular lymphoma) as well as solid tumors.
The investigators said that, to date, ASN002 has been well tolerated and has shown encouraging early evidence of clinical activity and symptomatic benefit in the lymphoma patients. ![]()
A dual kinase inhibitor is active against a range of hematologic malignancies, according to preclinical research.
Investigators found that ASN002, a SYK/JAK inhibitor, exhibited “potent” antiproliferative activity in leukemia, lymphoma, and myeloma cell lines.
ASN002 also inhibited tumor growth in mouse models of these malignancies and proved active against ibrutinib-resistant diffuse large B-cell lymphoma (DLBCL).
The investigators presented these results at the AACR Annual Meeting 2017 (abstract 4204).
The work was conducted by employees of Asana BioSciences, the company developing ASN002.
The investigators tested ASN002 in 178 cell lines and found the drug exhibited “strong antiproliferative activity” in a range of hematologic cancer cell lines, including:
- Leukemia—HEL31, HL60, Jurkat, MOLM-13, and MOLM-16
- Lymphoma—KARPAS-299, OCI-LY10, OCI-LY-19, Pfeiffer, Raji, Ramos, SU-DHL-1, SU-DHL-6, and SU-DHL-10
- Myeloma—H929, JJN3, OPM2, and U266.
In addition, ASN002 was active against ibrutinib-resistant clones derived from the DLBCL cell line SU-DHL-6.
In a SU-DHL-6 xenograft model, the combination of ASN002 and ibrutinib was more effective than either compound alone.
The investigators also found that ASN002 alone demonstrated “strong tumor growth inhibition” in mouse models of DLBCL (Pfeiffer and SU-DHL-6), myeloma (H929), and erythroleukemia (HEL).
The team pointed out that ASN002 is currently under investigation in a phase 1/2 study of patients with B-cell lymphomas (DLBCL, mantle cell lymphoma, and follicular lymphoma) as well as solid tumors.
The investigators said that, to date, ASN002 has been well tolerated and has shown encouraging early evidence of clinical activity and symptomatic benefit in the lymphoma patients. ![]()
Imbalance drives development of B-ALL, team says
Researchers say they have discovered an imbalance that drives the development of B-cell acute lymphoblastic leukemia (B-ALL).
The group’s study suggests that activation of STAT5 causes competition among other transcription factors that leads to B-ALL.
Therefore, the researchers believe that inhibiting the activation of STAT5 and restoring the natural balance of proteins could mean more effective treatment for B-ALL.
Seth Frietze, PhD, of the University of Vermont in Burlington, Vermont, and his colleagues conducted this research and reported the results in Nature Immunology.
The researchers first studied the role of STAT5 in B-ALL using mouse models.
The experiments revealed that STAT5 activation and defects in a signaling pathway worked together to promote B-ALL. The defects were in signaling components of the B-cell antigen receptor precursor—IKAROS, NF-κB, BLNK, BTK, and PKCβ.
With further investigation, the researchers found that STAT5 “antagonized NF-κB and IKAROS by opposing the regulation of shared target genes.”
The team also studied samples from patients with B-ALL and found that patients with a high ratio of active STAT5 to NF-κB or IKAROS had more aggressive disease.
Specifically, the ratio of active STAT5 to IKAROS was negatively correlated with patient survival and the duration of remission. The ratio of active STAT5 to the NF-κB subunit RELA correlated with remission duration but not survival.
“The major outcome of this story is that a signature emerged from looking at the level of activated proteins compared to other proteins that’s very predictive of how a patient will respond to therapy,” Dr Frietze said.
“That’s a novel finding. If we could find drugs to target that activation, that could be an incredibly effective way to treat leukemia.” ![]()
Researchers say they have discovered an imbalance that drives the development of B-cell acute lymphoblastic leukemia (B-ALL).
The group’s study suggests that activation of STAT5 causes competition among other transcription factors that leads to B-ALL.
Therefore, the researchers believe that inhibiting the activation of STAT5 and restoring the natural balance of proteins could mean more effective treatment for B-ALL.
Seth Frietze, PhD, of the University of Vermont in Burlington, Vermont, and his colleagues conducted this research and reported the results in Nature Immunology.
The researchers first studied the role of STAT5 in B-ALL using mouse models.
The experiments revealed that STAT5 activation and defects in a signaling pathway worked together to promote B-ALL. The defects were in signaling components of the B-cell antigen receptor precursor—IKAROS, NF-κB, BLNK, BTK, and PKCβ.
With further investigation, the researchers found that STAT5 “antagonized NF-κB and IKAROS by opposing the regulation of shared target genes.”
The team also studied samples from patients with B-ALL and found that patients with a high ratio of active STAT5 to NF-κB or IKAROS had more aggressive disease.
Specifically, the ratio of active STAT5 to IKAROS was negatively correlated with patient survival and the duration of remission. The ratio of active STAT5 to the NF-κB subunit RELA correlated with remission duration but not survival.
“The major outcome of this story is that a signature emerged from looking at the level of activated proteins compared to other proteins that’s very predictive of how a patient will respond to therapy,” Dr Frietze said.
“That’s a novel finding. If we could find drugs to target that activation, that could be an incredibly effective way to treat leukemia.” ![]()
Researchers say they have discovered an imbalance that drives the development of B-cell acute lymphoblastic leukemia (B-ALL).
The group’s study suggests that activation of STAT5 causes competition among other transcription factors that leads to B-ALL.
Therefore, the researchers believe that inhibiting the activation of STAT5 and restoring the natural balance of proteins could mean more effective treatment for B-ALL.
Seth Frietze, PhD, of the University of Vermont in Burlington, Vermont, and his colleagues conducted this research and reported the results in Nature Immunology.
The researchers first studied the role of STAT5 in B-ALL using mouse models.
The experiments revealed that STAT5 activation and defects in a signaling pathway worked together to promote B-ALL. The defects were in signaling components of the B-cell antigen receptor precursor—IKAROS, NF-κB, BLNK, BTK, and PKCβ.
With further investigation, the researchers found that STAT5 “antagonized NF-κB and IKAROS by opposing the regulation of shared target genes.”
The team also studied samples from patients with B-ALL and found that patients with a high ratio of active STAT5 to NF-κB or IKAROS had more aggressive disease.
Specifically, the ratio of active STAT5 to IKAROS was negatively correlated with patient survival and the duration of remission. The ratio of active STAT5 to the NF-κB subunit RELA correlated with remission duration but not survival.
“The major outcome of this story is that a signature emerged from looking at the level of activated proteins compared to other proteins that’s very predictive of how a patient will respond to therapy,” Dr Frietze said.
“That’s a novel finding. If we could find drugs to target that activation, that could be an incredibly effective way to treat leukemia.” ![]()
TKI shows promise in preclinical study of AML
WASHINGTON, DC—Preclinical data suggest a novel tyrosine kinase inhibitor (TKI) may be an effective treatment for patients with NTRK-rearranged acute myeloid leukemia (AML).
Entrectinib is a TKI targeting tumors that harbor TRK, ROS1, or ALK fusions.
Researchers found that entrectinib inhibited cell proliferation in NTRK-rearranged AML cell lines.
In mouse models of NTRK-rearranged AML, entrectinib induced tumor regression and eliminated residual AML cells from the bone marrow.
These results were presented at the AACR Annual Meeting 2017 (abstract 5158). The research was conducted by employees of Ignyta, Inc., the company developing entrectinib.
The researchers first tested entrectinib in AML cell lines. They observed “potent” anti-proliferative activity in a pair of NTRK-fusion-positive AML cell lines, IMS-M2 and M0-91.
Entrectinib inhibited TRK signaling and induced cell-cycle arrest in these cell lines. The TKI also induced both caspase 3-dependent apoptosis and PARP cleavage in a dose- and time-dependent manner.
However, entrectinib showed minimal activity against an NTRK-fusion-negative AML cell line, Kasumi-1.
The researchers also tested entrectinib in mouse models of NTRK-fusion-driven AML.
The TKI induced tumor regression in both IMS-M2 and M0-91 models, and the drug eliminated leukemic cells in the bone marrow.
The researchers said these results provide rationale for the clinical development of entrectinib in molecularly defined hematologic malignancies.
Entrectinib is currently being studied in a phase 2 trial of solid tumor malignancies. ![]()
WASHINGTON, DC—Preclinical data suggest a novel tyrosine kinase inhibitor (TKI) may be an effective treatment for patients with NTRK-rearranged acute myeloid leukemia (AML).
Entrectinib is a TKI targeting tumors that harbor TRK, ROS1, or ALK fusions.
Researchers found that entrectinib inhibited cell proliferation in NTRK-rearranged AML cell lines.
In mouse models of NTRK-rearranged AML, entrectinib induced tumor regression and eliminated residual AML cells from the bone marrow.
These results were presented at the AACR Annual Meeting 2017 (abstract 5158). The research was conducted by employees of Ignyta, Inc., the company developing entrectinib.
The researchers first tested entrectinib in AML cell lines. They observed “potent” anti-proliferative activity in a pair of NTRK-fusion-positive AML cell lines, IMS-M2 and M0-91.
Entrectinib inhibited TRK signaling and induced cell-cycle arrest in these cell lines. The TKI also induced both caspase 3-dependent apoptosis and PARP cleavage in a dose- and time-dependent manner.
However, entrectinib showed minimal activity against an NTRK-fusion-negative AML cell line, Kasumi-1.
The researchers also tested entrectinib in mouse models of NTRK-fusion-driven AML.
The TKI induced tumor regression in both IMS-M2 and M0-91 models, and the drug eliminated leukemic cells in the bone marrow.
The researchers said these results provide rationale for the clinical development of entrectinib in molecularly defined hematologic malignancies.
Entrectinib is currently being studied in a phase 2 trial of solid tumor malignancies. ![]()
WASHINGTON, DC—Preclinical data suggest a novel tyrosine kinase inhibitor (TKI) may be an effective treatment for patients with NTRK-rearranged acute myeloid leukemia (AML).
Entrectinib is a TKI targeting tumors that harbor TRK, ROS1, or ALK fusions.
Researchers found that entrectinib inhibited cell proliferation in NTRK-rearranged AML cell lines.
In mouse models of NTRK-rearranged AML, entrectinib induced tumor regression and eliminated residual AML cells from the bone marrow.
These results were presented at the AACR Annual Meeting 2017 (abstract 5158). The research was conducted by employees of Ignyta, Inc., the company developing entrectinib.
The researchers first tested entrectinib in AML cell lines. They observed “potent” anti-proliferative activity in a pair of NTRK-fusion-positive AML cell lines, IMS-M2 and M0-91.
Entrectinib inhibited TRK signaling and induced cell-cycle arrest in these cell lines. The TKI also induced both caspase 3-dependent apoptosis and PARP cleavage in a dose- and time-dependent manner.
However, entrectinib showed minimal activity against an NTRK-fusion-negative AML cell line, Kasumi-1.
The researchers also tested entrectinib in mouse models of NTRK-fusion-driven AML.
The TKI induced tumor regression in both IMS-M2 and M0-91 models, and the drug eliminated leukemic cells in the bone marrow.
The researchers said these results provide rationale for the clinical development of entrectinib in molecularly defined hematologic malignancies.
Entrectinib is currently being studied in a phase 2 trial of solid tumor malignancies. ![]()
Expanded drug combinations produce best myeloma induction
NEW YORK – Optimal induction therapy for patients with multiple myeloma requires all the therapeutic tools currently available for combination therapy, which means using four agents followed by autologous stem cell transplantation, Paul G. Richardson, MD, said at a conference held by Imedex.
He suggested that a rational combination regimen for induction therapy of multiple myeloma would include a second- or third-generation immunomodulator such as lenalidomide (Revlimid) or pomalidomide (Pomalyst); a proteasome inhibitor such as bortezomib (Velcade), carfilzomib (Kyprolis), or ixazomib (Ninlaro); plus standard dexamethasone to form a contemporary backbone regimen for inducing remission in patients with multiple myeloma.
“Therapeutic parsimony is not recommended. You need a combination” of all available drug classes, suggested Dr. Richardson, professor of medicine at Harvard Medical School and clinical program leader of the Multiple Myeloma Center at the Dana-Farber Cancer Institute in Boston.
“It isn’t rational to limit the treatment choices. We need to bring them all together,” he said. The “most rational” combination melds an immunomodulator, proteasome inhibitor, plus a monoclonal antibody that he called a “true game changer. Add the antibody on top of the three-drug platform” of an immunomodulator, proteasome inhibitor, and dexamethasone. Dr. Richardson conceded, however, that a concern with such combinations is how to manage the toxicity that would likely result.
He cited several recent examples of demonstrated superior efficacy in the form of more complete responses from combined regimens, followed by autologous stem cell transplantation.
For example, a recent report from a French-led group compared the efficacy of a combination of an immunomodulator, proteasome inhibitor, and dexamethasone against the same combination, followed by autologous stem cell transplantation (N Engl J Med. 2017 Apr 6;376[14]:1311-20). The combined regimen plus transplant resulted in a 59% complete response rate, “the best response rate we’ve ever seen” in multiple myeloma, Dr. Richardson noted, compared with a 48% complete response rate in patients who did not undergo a stem cell transplant.
Another speaker at the meeting, Ruben Niesvizky, MD, suggested a more cautious approach to using the monoclonal antibody daratumumab for induction. He cited the published experience in adding the antibody to pared-down backbone therapy in the setting of relapsed or relapsed and refractory disease, such as a proteasome inhibitor plus dexamethasone (N Engl J Med. 2016 Aug 25;375[8]:754-66) or an immunomodulator plus dexamethasone (N Engl J Med. 2016 Oct 6;375[14]:1319-31).
Adding a monoclonal antibody such as daratumumab to combination therapy is the “wave of the future,” said Dr. Niesvizky, professor of medicine and director of the Multiple Myeloma Center at New York Presbyterian Hospital/Weill Cornell Medical Center. It provides treatment that reduces disease-related complications and achieves effective and extended disease control with improved overall survival, while being well tolerated and facilitating stem cell collection.
Dr. Richardson has been a consultant to Celgene, Genmab, Janssen, Novartis, Oncopeptides AB, and Takeda and has received research funding from Celgene and Takeda. Dr. Niesvizky has been a consultant to Amgen, Bristol-Myers Squibb, Celgene, Janssen, and Takeda and has received research support from Amgen, Bristol-Myers Squibb, Celgene, and Takeda.
[email protected]
On Twitter @mitchelzoler
NEW YORK – Optimal induction therapy for patients with multiple myeloma requires all the therapeutic tools currently available for combination therapy, which means using four agents followed by autologous stem cell transplantation, Paul G. Richardson, MD, said at a conference held by Imedex.
He suggested that a rational combination regimen for induction therapy of multiple myeloma would include a second- or third-generation immunomodulator such as lenalidomide (Revlimid) or pomalidomide (Pomalyst); a proteasome inhibitor such as bortezomib (Velcade), carfilzomib (Kyprolis), or ixazomib (Ninlaro); plus standard dexamethasone to form a contemporary backbone regimen for inducing remission in patients with multiple myeloma.
“Therapeutic parsimony is not recommended. You need a combination” of all available drug classes, suggested Dr. Richardson, professor of medicine at Harvard Medical School and clinical program leader of the Multiple Myeloma Center at the Dana-Farber Cancer Institute in Boston.
“It isn’t rational to limit the treatment choices. We need to bring them all together,” he said. The “most rational” combination melds an immunomodulator, proteasome inhibitor, plus a monoclonal antibody that he called a “true game changer. Add the antibody on top of the three-drug platform” of an immunomodulator, proteasome inhibitor, and dexamethasone. Dr. Richardson conceded, however, that a concern with such combinations is how to manage the toxicity that would likely result.
He cited several recent examples of demonstrated superior efficacy in the form of more complete responses from combined regimens, followed by autologous stem cell transplantation.
For example, a recent report from a French-led group compared the efficacy of a combination of an immunomodulator, proteasome inhibitor, and dexamethasone against the same combination, followed by autologous stem cell transplantation (N Engl J Med. 2017 Apr 6;376[14]:1311-20). The combined regimen plus transplant resulted in a 59% complete response rate, “the best response rate we’ve ever seen” in multiple myeloma, Dr. Richardson noted, compared with a 48% complete response rate in patients who did not undergo a stem cell transplant.
Another speaker at the meeting, Ruben Niesvizky, MD, suggested a more cautious approach to using the monoclonal antibody daratumumab for induction. He cited the published experience in adding the antibody to pared-down backbone therapy in the setting of relapsed or relapsed and refractory disease, such as a proteasome inhibitor plus dexamethasone (N Engl J Med. 2016 Aug 25;375[8]:754-66) or an immunomodulator plus dexamethasone (N Engl J Med. 2016 Oct 6;375[14]:1319-31).
Adding a monoclonal antibody such as daratumumab to combination therapy is the “wave of the future,” said Dr. Niesvizky, professor of medicine and director of the Multiple Myeloma Center at New York Presbyterian Hospital/Weill Cornell Medical Center. It provides treatment that reduces disease-related complications and achieves effective and extended disease control with improved overall survival, while being well tolerated and facilitating stem cell collection.
Dr. Richardson has been a consultant to Celgene, Genmab, Janssen, Novartis, Oncopeptides AB, and Takeda and has received research funding from Celgene and Takeda. Dr. Niesvizky has been a consultant to Amgen, Bristol-Myers Squibb, Celgene, Janssen, and Takeda and has received research support from Amgen, Bristol-Myers Squibb, Celgene, and Takeda.
[email protected]
On Twitter @mitchelzoler
NEW YORK – Optimal induction therapy for patients with multiple myeloma requires all the therapeutic tools currently available for combination therapy, which means using four agents followed by autologous stem cell transplantation, Paul G. Richardson, MD, said at a conference held by Imedex.
He suggested that a rational combination regimen for induction therapy of multiple myeloma would include a second- or third-generation immunomodulator such as lenalidomide (Revlimid) or pomalidomide (Pomalyst); a proteasome inhibitor such as bortezomib (Velcade), carfilzomib (Kyprolis), or ixazomib (Ninlaro); plus standard dexamethasone to form a contemporary backbone regimen for inducing remission in patients with multiple myeloma.
“Therapeutic parsimony is not recommended. You need a combination” of all available drug classes, suggested Dr. Richardson, professor of medicine at Harvard Medical School and clinical program leader of the Multiple Myeloma Center at the Dana-Farber Cancer Institute in Boston.
“It isn’t rational to limit the treatment choices. We need to bring them all together,” he said. The “most rational” combination melds an immunomodulator, proteasome inhibitor, plus a monoclonal antibody that he called a “true game changer. Add the antibody on top of the three-drug platform” of an immunomodulator, proteasome inhibitor, and dexamethasone. Dr. Richardson conceded, however, that a concern with such combinations is how to manage the toxicity that would likely result.
He cited several recent examples of demonstrated superior efficacy in the form of more complete responses from combined regimens, followed by autologous stem cell transplantation.
For example, a recent report from a French-led group compared the efficacy of a combination of an immunomodulator, proteasome inhibitor, and dexamethasone against the same combination, followed by autologous stem cell transplantation (N Engl J Med. 2017 Apr 6;376[14]:1311-20). The combined regimen plus transplant resulted in a 59% complete response rate, “the best response rate we’ve ever seen” in multiple myeloma, Dr. Richardson noted, compared with a 48% complete response rate in patients who did not undergo a stem cell transplant.
Another speaker at the meeting, Ruben Niesvizky, MD, suggested a more cautious approach to using the monoclonal antibody daratumumab for induction. He cited the published experience in adding the antibody to pared-down backbone therapy in the setting of relapsed or relapsed and refractory disease, such as a proteasome inhibitor plus dexamethasone (N Engl J Med. 2016 Aug 25;375[8]:754-66) or an immunomodulator plus dexamethasone (N Engl J Med. 2016 Oct 6;375[14]:1319-31).
Adding a monoclonal antibody such as daratumumab to combination therapy is the “wave of the future,” said Dr. Niesvizky, professor of medicine and director of the Multiple Myeloma Center at New York Presbyterian Hospital/Weill Cornell Medical Center. It provides treatment that reduces disease-related complications and achieves effective and extended disease control with improved overall survival, while being well tolerated and facilitating stem cell collection.
Dr. Richardson has been a consultant to Celgene, Genmab, Janssen, Novartis, Oncopeptides AB, and Takeda and has received research funding from Celgene and Takeda. Dr. Niesvizky has been a consultant to Amgen, Bristol-Myers Squibb, Celgene, Janssen, and Takeda and has received research support from Amgen, Bristol-Myers Squibb, Celgene, and Takeda.
[email protected]
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM A MEETING ON HEMATOLOGIC MALIGNANCIES
Leukemia, NHL more common in African-born blacks than US-born blacks
The cancer profile of black US immigrants born in sub-Saharan Africa (ABs) differs from that of US-born black individuals (USBs) and varies by region of birth, according to a new study.
For instance, the data showed that ABs who immigrated to the US had a higher incidence of leukemia and non-Hodgkin lymphoma (NHL) than USBs.
And there was a higher incidence of NHL among ABs born in Eastern Africa than among those born in Western Africa.
“Typically, cancer occurrence among blacks in the United States is presented as one homogeneous group, with no breakdown by country or region of birth,” said Ahemdin Jemal, DVM, PhD, of the American Cancer Society in Atlanta, Georgia.
“Our study shows that approach masks important potential differences that may be key to guiding cancer prevention programs for African-born black immigrants.”
Dr Jemal and his colleagues described this study in the journal Cancer.
The team reviewed cancer incidence data from the Surveillance, Epidemiology, and End Results (SEER 17) program covering the period from 2000 through 2012.
They calculated age-standardized proportional incidence ratios (PIRs), comparing the frequency of the top 15 cancers in ABs with that of USBs, by sex and region of birth.
ABs had a significantly higher proportion of hematologic malignancies (leukemia and NHL), infection-related cancers (liver, stomach, and Kaposi sarcoma), prostate cancer, and thyroid cancers (in females only), when compared to USBs.
In females, the incidence of NHL was 3.4% in ABs and 2.5% in USBs, with a PIR of 1.19. The incidence of leukemia was 3.0% in ABs and 1.9% in USBs, with a PIR of 1.62.
In males, the incidence of NHL was 5.9% in ABs and 3.0% in USBs, with a PIR of 1.34. The incidence of leukemia was 3.0% in ABs and 2.2% in USBs, with a PIR of 1.40.
The investigators also calculated the PIRs of the 5 most frequent cancers in ABs compared to USBs by region of origin (Western Africa vs Eastern Africa).
For both sexes, the incidence of NHL was 4.0% in ABs born in Western Africa (PIR=1.17) and 5.5% in ABs born in Eastern Africa (PIR=1.44).
Leukemia was not included in this analysis because it was not among the top 5 cancers. ![]()
The cancer profile of black US immigrants born in sub-Saharan Africa (ABs) differs from that of US-born black individuals (USBs) and varies by region of birth, according to a new study.
For instance, the data showed that ABs who immigrated to the US had a higher incidence of leukemia and non-Hodgkin lymphoma (NHL) than USBs.
And there was a higher incidence of NHL among ABs born in Eastern Africa than among those born in Western Africa.
“Typically, cancer occurrence among blacks in the United States is presented as one homogeneous group, with no breakdown by country or region of birth,” said Ahemdin Jemal, DVM, PhD, of the American Cancer Society in Atlanta, Georgia.
“Our study shows that approach masks important potential differences that may be key to guiding cancer prevention programs for African-born black immigrants.”
Dr Jemal and his colleagues described this study in the journal Cancer.
The team reviewed cancer incidence data from the Surveillance, Epidemiology, and End Results (SEER 17) program covering the period from 2000 through 2012.
They calculated age-standardized proportional incidence ratios (PIRs), comparing the frequency of the top 15 cancers in ABs with that of USBs, by sex and region of birth.
ABs had a significantly higher proportion of hematologic malignancies (leukemia and NHL), infection-related cancers (liver, stomach, and Kaposi sarcoma), prostate cancer, and thyroid cancers (in females only), when compared to USBs.
In females, the incidence of NHL was 3.4% in ABs and 2.5% in USBs, with a PIR of 1.19. The incidence of leukemia was 3.0% in ABs and 1.9% in USBs, with a PIR of 1.62.
In males, the incidence of NHL was 5.9% in ABs and 3.0% in USBs, with a PIR of 1.34. The incidence of leukemia was 3.0% in ABs and 2.2% in USBs, with a PIR of 1.40.
The investigators also calculated the PIRs of the 5 most frequent cancers in ABs compared to USBs by region of origin (Western Africa vs Eastern Africa).
For both sexes, the incidence of NHL was 4.0% in ABs born in Western Africa (PIR=1.17) and 5.5% in ABs born in Eastern Africa (PIR=1.44).
Leukemia was not included in this analysis because it was not among the top 5 cancers. ![]()
The cancer profile of black US immigrants born in sub-Saharan Africa (ABs) differs from that of US-born black individuals (USBs) and varies by region of birth, according to a new study.
For instance, the data showed that ABs who immigrated to the US had a higher incidence of leukemia and non-Hodgkin lymphoma (NHL) than USBs.
And there was a higher incidence of NHL among ABs born in Eastern Africa than among those born in Western Africa.
“Typically, cancer occurrence among blacks in the United States is presented as one homogeneous group, with no breakdown by country or region of birth,” said Ahemdin Jemal, DVM, PhD, of the American Cancer Society in Atlanta, Georgia.
“Our study shows that approach masks important potential differences that may be key to guiding cancer prevention programs for African-born black immigrants.”
Dr Jemal and his colleagues described this study in the journal Cancer.
The team reviewed cancer incidence data from the Surveillance, Epidemiology, and End Results (SEER 17) program covering the period from 2000 through 2012.
They calculated age-standardized proportional incidence ratios (PIRs), comparing the frequency of the top 15 cancers in ABs with that of USBs, by sex and region of birth.
ABs had a significantly higher proportion of hematologic malignancies (leukemia and NHL), infection-related cancers (liver, stomach, and Kaposi sarcoma), prostate cancer, and thyroid cancers (in females only), when compared to USBs.
In females, the incidence of NHL was 3.4% in ABs and 2.5% in USBs, with a PIR of 1.19. The incidence of leukemia was 3.0% in ABs and 1.9% in USBs, with a PIR of 1.62.
In males, the incidence of NHL was 5.9% in ABs and 3.0% in USBs, with a PIR of 1.34. The incidence of leukemia was 3.0% in ABs and 2.2% in USBs, with a PIR of 1.40.
The investigators also calculated the PIRs of the 5 most frequent cancers in ABs compared to USBs by region of origin (Western Africa vs Eastern Africa).
For both sexes, the incidence of NHL was 4.0% in ABs born in Western Africa (PIR=1.17) and 5.5% in ABs born in Eastern Africa (PIR=1.44).
Leukemia was not included in this analysis because it was not among the top 5 cancers.