User login
Consider treatment, testing when CLL symptoms emerge
CHICAGO – said Paul M. Barr, MD.
He described a patient who had been observed for 7 years when he began to complain of increasing fatigue and lost work time. A complete blood count (CBC) showed thrombocytopenia.
The recently updated International Workshop on Chronic Lymphocytic Leukemia (iwCLL) guidelines state that assessments before treatment in this type of patient should include history and physical, evaluation of infectious disease status, and routine laboratory testing – including CBC and differential, chemistry, serum immunoglobulin, and direct antiglobulin test.
“Bone marrow biopsies and [computed tomography] scans are listed as ‘not necessarily required,’ ” Dr. Barr, medical director of the Clinical Trials Office for Wilmot Cancer Institute at the University of Rochester (N.Y.) said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
He added that he opts for CT scans prior to therapy “to understand the patient’s disease burden and potentially to compare to later” and that bone marrow biopsy is “still very reasonable” for understanding the source of a patient’s cytopenias.
“Is it marrow failure or [immune thrombocytopenia]? Could the patient have [myelodysplastic syndrome]? All important considerations,” he said.
Positron emission tomography (PET) scans, however, are only considered when there is concern about transformation, he noted.
Predictive tests that should be conducted before initiating therapy, and that could help in guiding therapy decisions, include TP53 mutation testing and immunoglobulin heavy chain variable region gene (IGHV) mutational status testing (although this doesn’t need to be repeated if it was done at diagnosis because mutational status doesn’t change). Another helpful test is molecular cytogenetics using fluorescence in situ hybridization (FISH) for del(13q), del(17p), and trisomy 12 in peripheral blood lymphocytes. This should be repeated even it was done at diagnosis because patients can acquire additional molecular aberrations over time, Dr. Barr said.
Among the data that justify this advice for predictive testing are studies showing the curative potential of fludarabine/cyclophosphamide/rituximab (FCR) in mutated IGHV CLL, the progression-free survival (PFS) benefits of ibrutinib for patients with del(17p), and the activity of idelalisib in relapsed/refractory CLL patients, including those with TP53 dysfunction.
“The IGHV mutation status is useful to know what to expect from chemoimmunotherapy over time,” Dr. Barr said, explaining that several analyses demonstrate that patients with mutated IGHV genes (patients with low-risk disease) respond exceptionally well to chemoimmunotherapy, especially FCR.
In fact, studies, including a 2016 study by Philip A. Thompson and his colleagues and another by Kirsten Fischer and her colleagues, show that nearly 60% of patients with IGHV mutation remain in remission 10 years after FCR treatment, he said. However, the same is not necessarily true for bendamustine/rituximab (BR); the CLL10 study showed a significantly greater PFS with FCR, compared with that seen with BR.
Unmutated patients in that study had lower PFS, but the outcomes were still better with FCR than with BR, he said.
Studies of novel agents, including ibrutinib and idelalisib, suggest they may have particular benefit in higher-risk patients.
Ibrutinib was shown in a phase 2 study to be of benefit regardless of IGHV status, and this was replicated in the first-line RESONATE 2 study, which compared ibrutinib with chlorambucil and showed it had better PFS than that seen in unmutated patients treated with FCR in other studies, said Dr. Barr, the first author on that study.
“So you can see how the treatment paradigms are starting to evolve. It does look like ... comparing across trials, that ibrutinib leads to better remission durations, compared to chemoimmunotherapy, so far,” he said.
Ibrutinib has also been shown to be of benefit for patients with del(17p). A single-arm phase 2 study showed 79% PFS in relapsed, high-risk patients, which is much better than has been seen with chemoimmunotherapy, he noted.
“Venetoclax is also a very good option for this patient population in the relapse setting,” he said, adding that the PFS with venetoclax has been shown to be very similar to that with ibrutinib.
Similarly, idelalisib has been shown to provide comparable benefit in relapsed/refractory CLL patients, with and without del(17p)/TP53 mutation, he said.
Dr. Barr is a consultant for Pharmacyclics, AbbVie, Celgene, Gilead, Infinity, Novartis, and Seattle Genetics and has received research funding from Pharmacyclics and AbbVie.
CHICAGO – said Paul M. Barr, MD.
He described a patient who had been observed for 7 years when he began to complain of increasing fatigue and lost work time. A complete blood count (CBC) showed thrombocytopenia.
The recently updated International Workshop on Chronic Lymphocytic Leukemia (iwCLL) guidelines state that assessments before treatment in this type of patient should include history and physical, evaluation of infectious disease status, and routine laboratory testing – including CBC and differential, chemistry, serum immunoglobulin, and direct antiglobulin test.
“Bone marrow biopsies and [computed tomography] scans are listed as ‘not necessarily required,’ ” Dr. Barr, medical director of the Clinical Trials Office for Wilmot Cancer Institute at the University of Rochester (N.Y.) said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
He added that he opts for CT scans prior to therapy “to understand the patient’s disease burden and potentially to compare to later” and that bone marrow biopsy is “still very reasonable” for understanding the source of a patient’s cytopenias.
“Is it marrow failure or [immune thrombocytopenia]? Could the patient have [myelodysplastic syndrome]? All important considerations,” he said.
Positron emission tomography (PET) scans, however, are only considered when there is concern about transformation, he noted.
Predictive tests that should be conducted before initiating therapy, and that could help in guiding therapy decisions, include TP53 mutation testing and immunoglobulin heavy chain variable region gene (IGHV) mutational status testing (although this doesn’t need to be repeated if it was done at diagnosis because mutational status doesn’t change). Another helpful test is molecular cytogenetics using fluorescence in situ hybridization (FISH) for del(13q), del(17p), and trisomy 12 in peripheral blood lymphocytes. This should be repeated even it was done at diagnosis because patients can acquire additional molecular aberrations over time, Dr. Barr said.
Among the data that justify this advice for predictive testing are studies showing the curative potential of fludarabine/cyclophosphamide/rituximab (FCR) in mutated IGHV CLL, the progression-free survival (PFS) benefits of ibrutinib for patients with del(17p), and the activity of idelalisib in relapsed/refractory CLL patients, including those with TP53 dysfunction.
“The IGHV mutation status is useful to know what to expect from chemoimmunotherapy over time,” Dr. Barr said, explaining that several analyses demonstrate that patients with mutated IGHV genes (patients with low-risk disease) respond exceptionally well to chemoimmunotherapy, especially FCR.
In fact, studies, including a 2016 study by Philip A. Thompson and his colleagues and another by Kirsten Fischer and her colleagues, show that nearly 60% of patients with IGHV mutation remain in remission 10 years after FCR treatment, he said. However, the same is not necessarily true for bendamustine/rituximab (BR); the CLL10 study showed a significantly greater PFS with FCR, compared with that seen with BR.
Unmutated patients in that study had lower PFS, but the outcomes were still better with FCR than with BR, he said.
Studies of novel agents, including ibrutinib and idelalisib, suggest they may have particular benefit in higher-risk patients.
Ibrutinib was shown in a phase 2 study to be of benefit regardless of IGHV status, and this was replicated in the first-line RESONATE 2 study, which compared ibrutinib with chlorambucil and showed it had better PFS than that seen in unmutated patients treated with FCR in other studies, said Dr. Barr, the first author on that study.
“So you can see how the treatment paradigms are starting to evolve. It does look like ... comparing across trials, that ibrutinib leads to better remission durations, compared to chemoimmunotherapy, so far,” he said.
Ibrutinib has also been shown to be of benefit for patients with del(17p). A single-arm phase 2 study showed 79% PFS in relapsed, high-risk patients, which is much better than has been seen with chemoimmunotherapy, he noted.
“Venetoclax is also a very good option for this patient population in the relapse setting,” he said, adding that the PFS with venetoclax has been shown to be very similar to that with ibrutinib.
Similarly, idelalisib has been shown to provide comparable benefit in relapsed/refractory CLL patients, with and without del(17p)/TP53 mutation, he said.
Dr. Barr is a consultant for Pharmacyclics, AbbVie, Celgene, Gilead, Infinity, Novartis, and Seattle Genetics and has received research funding from Pharmacyclics and AbbVie.
CHICAGO – said Paul M. Barr, MD.
He described a patient who had been observed for 7 years when he began to complain of increasing fatigue and lost work time. A complete blood count (CBC) showed thrombocytopenia.
The recently updated International Workshop on Chronic Lymphocytic Leukemia (iwCLL) guidelines state that assessments before treatment in this type of patient should include history and physical, evaluation of infectious disease status, and routine laboratory testing – including CBC and differential, chemistry, serum immunoglobulin, and direct antiglobulin test.
“Bone marrow biopsies and [computed tomography] scans are listed as ‘not necessarily required,’ ” Dr. Barr, medical director of the Clinical Trials Office for Wilmot Cancer Institute at the University of Rochester (N.Y.) said during a presentation at the American Society of Hematology Meeting on Hematologic Malignancies.
He added that he opts for CT scans prior to therapy “to understand the patient’s disease burden and potentially to compare to later” and that bone marrow biopsy is “still very reasonable” for understanding the source of a patient’s cytopenias.
“Is it marrow failure or [immune thrombocytopenia]? Could the patient have [myelodysplastic syndrome]? All important considerations,” he said.
Positron emission tomography (PET) scans, however, are only considered when there is concern about transformation, he noted.
Predictive tests that should be conducted before initiating therapy, and that could help in guiding therapy decisions, include TP53 mutation testing and immunoglobulin heavy chain variable region gene (IGHV) mutational status testing (although this doesn’t need to be repeated if it was done at diagnosis because mutational status doesn’t change). Another helpful test is molecular cytogenetics using fluorescence in situ hybridization (FISH) for del(13q), del(17p), and trisomy 12 in peripheral blood lymphocytes. This should be repeated even it was done at diagnosis because patients can acquire additional molecular aberrations over time, Dr. Barr said.
Among the data that justify this advice for predictive testing are studies showing the curative potential of fludarabine/cyclophosphamide/rituximab (FCR) in mutated IGHV CLL, the progression-free survival (PFS) benefits of ibrutinib for patients with del(17p), and the activity of idelalisib in relapsed/refractory CLL patients, including those with TP53 dysfunction.
“The IGHV mutation status is useful to know what to expect from chemoimmunotherapy over time,” Dr. Barr said, explaining that several analyses demonstrate that patients with mutated IGHV genes (patients with low-risk disease) respond exceptionally well to chemoimmunotherapy, especially FCR.
In fact, studies, including a 2016 study by Philip A. Thompson and his colleagues and another by Kirsten Fischer and her colleagues, show that nearly 60% of patients with IGHV mutation remain in remission 10 years after FCR treatment, he said. However, the same is not necessarily true for bendamustine/rituximab (BR); the CLL10 study showed a significantly greater PFS with FCR, compared with that seen with BR.
Unmutated patients in that study had lower PFS, but the outcomes were still better with FCR than with BR, he said.
Studies of novel agents, including ibrutinib and idelalisib, suggest they may have particular benefit in higher-risk patients.
Ibrutinib was shown in a phase 2 study to be of benefit regardless of IGHV status, and this was replicated in the first-line RESONATE 2 study, which compared ibrutinib with chlorambucil and showed it had better PFS than that seen in unmutated patients treated with FCR in other studies, said Dr. Barr, the first author on that study.
“So you can see how the treatment paradigms are starting to evolve. It does look like ... comparing across trials, that ibrutinib leads to better remission durations, compared to chemoimmunotherapy, so far,” he said.
Ibrutinib has also been shown to be of benefit for patients with del(17p). A single-arm phase 2 study showed 79% PFS in relapsed, high-risk patients, which is much better than has been seen with chemoimmunotherapy, he noted.
“Venetoclax is also a very good option for this patient population in the relapse setting,” he said, adding that the PFS with venetoclax has been shown to be very similar to that with ibrutinib.
Similarly, idelalisib has been shown to provide comparable benefit in relapsed/refractory CLL patients, with and without del(17p)/TP53 mutation, he said.
Dr. Barr is a consultant for Pharmacyclics, AbbVie, Celgene, Gilead, Infinity, Novartis, and Seattle Genetics and has received research funding from Pharmacyclics and AbbVie.
EXPERT ANALYSIS FROM MHM 2018
Prognostic features could improve ALL outcomes
CHICAGO – New recognition of the prognostic value of cytogenetic factors, minimal residual disease activity, and Philadelphia chromosome–like signature could improve the treatment and outcomes of acute lymphoblastic leukemia (ALL), according to Anjali Advani, MD.
CD20
About 80% of ALL is B-cell ALL and the majority of patients have pre–B-cell ALL, Dr. Advani, a hematologist and director of the inpatient leukemia program at the Cleveland Clinic, said at the American Society of Hematology Meeting on Hematologic Malignancies.
“And on the B lymphoblast, many antigens are expressed, including CD19, CD20, and CD52,” she said.
Rituximab, a drug often used for the treatment of lymphoma, is a chimeric monoclonal antibody against the protein CD20, which is expressed in 41% of ALL patients.
“Interestingly, CD20 expression in ALL has been associated with an adverse prognostic impact, which suggests that targeting this may potentially improve outcomes in these patients,” Dr. Advani said.
In fact, a recent randomized study by Sébastien Maury, MD, of the University of Paris-Est, and his colleagues, demonstrated that adding 16-18 doses of rituximab to a Berlin-Frankfurt-Münster (BFM)–based chemotherapy in Philadelphia chromosome (Ph)–negative patients aged 18-59 years with CD20-positive pre–B-cell ALL improved 2-year event-free survival from 52% to 65%. The data are consistent with those from prior studies, including a German study that showed a higher degree of minimal residual disease (MRD) negativity in patients treated with rituximab, she noted (N Engl J Med 2016;375:1044-53).
While the study by Dr. Maury and his colleagues didn’t look at MRD, that may offer an explanation for the improved event-free survival in their study, Dr. Advani suggested.
MRD
Minimal residual disease has become a standard part of practice in ALL, but pediatric ALL led the way in using early MRD measurement for risk-stratifying therapy, and it has taken a bit longer for it to be incorporated in the adult disease realm, Dr. Advani said.
Either flow cytometry or polymerase chain reaction (PCR) amplification can be used to measure MRD, she noted.
In one of the larger studies done in adults, researchers used PCR to look at MRD at two time points and stratified patients into three risk groups, including low, intermediate, and high risk (Blood. 2006 Feb 1;107:1116-23).
Measuring MRD at those two time points “clearly separated the prognosis of patients not only in terms of disease-free survival but [in] overall survival,” she said. “So that’s why, for ALL, this has really become very important.”
In the United States, where flow-based cytometry is used more, it is necessary to find a properly equipped laboratory that can provide reliable results, she added. “For example, at our center we actually send our MRD to Fred Hutchinson [Cancer Center in Seattle].”
Johns Hopkins [Baltimore] also has such a lab, and both can arrange to accept send-outs, she said.
The other “really exciting thing” in regard to MRD in ALL is the recent approval of blinatumomab for MRD-positive ALL, she said.
In a study of 113 evaluable patients who were treated with the monoclonal antibody, 78% achieved complete molecular response (Blood. 2018 Apr 5;131:1522-31).
“And probably most importantly, when they looked at those patients who responded to blinatumomab in terms of MRD, these patients had, again, not only improved relapse-free survival but also increased overall survival,” she said. “I think this really explains why in ALL, we are measuring MRD and how it can really impact these patients.”
One of the remaining questions that will be important to address going forward is whether patients with MRD-positive ALL should continue to be considered for transplant; some of these studies have shown “very, very good outcomes” in patients who have not been transplanted, she noted.
Ph-like signature
Another important new prognostic feature in ALL is the presence of the Ph-like signature, a gene expression signature that was initially described in children with poor-risk ALL, and which looks a lot like Ph-positive disease.
“When they delved in further, they identified that this signature actually correlated with multiple different [kinase] fusions ... and it turns out that 20%-25% of young adults have this signature,” she said.
Since event-free survival in young adults with ALL is usually in the 65%-70% range, most of the remaining 30%-35% likely have this signature, she explained.
The kinase fusions associated with the Ph-like signature retain intact tyrosine kinase domains, and the spectrum of the fusions changes across age groups.
Treatments targeting some of these – for example, dasatinib for patients with a Ph-like dasatinib-sensitive kinase mutation – are being investigated.
Additionally, the Children’s Oncology Group has developed a clinically adaptable screening assay to identify the signature, she noted.
“So I would say that, and there is probably some difference in opinion, it is now becoming fairly standard that at diagnosis in an adult with ALL we’re sending the Ph-like signature,” she said. “And again, usually you’re going to have to send this out, and at our center we send it out to [Nationwide Children’s Hospital] in Columbus [Ohio].”
As important as it is to identify the Ph-like signature, given its association with poor prognosis, a number of questions remain, including whether transplant improves outcomes.
“The hope is it probably does, and that’s something that’s being evaluated in studies,” she said, noting that clinical studies are also specifically targeting these patients.
“So these patients should probably be enrolled on a clinical trial, because their outcome is clearly inferior,” she said.
Dr. Advani reported consultancy for Pfizer; research funding from Genzyme, Novartis, Pfizer, and Sigma Tau; and honoraria from Genzyme, Pfizer, and Sigma Tau. She is also on the speakers bureau for Sigma Tau.
CHICAGO – New recognition of the prognostic value of cytogenetic factors, minimal residual disease activity, and Philadelphia chromosome–like signature could improve the treatment and outcomes of acute lymphoblastic leukemia (ALL), according to Anjali Advani, MD.
CD20
About 80% of ALL is B-cell ALL and the majority of patients have pre–B-cell ALL, Dr. Advani, a hematologist and director of the inpatient leukemia program at the Cleveland Clinic, said at the American Society of Hematology Meeting on Hematologic Malignancies.
“And on the B lymphoblast, many antigens are expressed, including CD19, CD20, and CD52,” she said.
Rituximab, a drug often used for the treatment of lymphoma, is a chimeric monoclonal antibody against the protein CD20, which is expressed in 41% of ALL patients.
“Interestingly, CD20 expression in ALL has been associated with an adverse prognostic impact, which suggests that targeting this may potentially improve outcomes in these patients,” Dr. Advani said.
In fact, a recent randomized study by Sébastien Maury, MD, of the University of Paris-Est, and his colleagues, demonstrated that adding 16-18 doses of rituximab to a Berlin-Frankfurt-Münster (BFM)–based chemotherapy in Philadelphia chromosome (Ph)–negative patients aged 18-59 years with CD20-positive pre–B-cell ALL improved 2-year event-free survival from 52% to 65%. The data are consistent with those from prior studies, including a German study that showed a higher degree of minimal residual disease (MRD) negativity in patients treated with rituximab, she noted (N Engl J Med 2016;375:1044-53).
While the study by Dr. Maury and his colleagues didn’t look at MRD, that may offer an explanation for the improved event-free survival in their study, Dr. Advani suggested.
MRD
Minimal residual disease has become a standard part of practice in ALL, but pediatric ALL led the way in using early MRD measurement for risk-stratifying therapy, and it has taken a bit longer for it to be incorporated in the adult disease realm, Dr. Advani said.
Either flow cytometry or polymerase chain reaction (PCR) amplification can be used to measure MRD, she noted.
In one of the larger studies done in adults, researchers used PCR to look at MRD at two time points and stratified patients into three risk groups, including low, intermediate, and high risk (Blood. 2006 Feb 1;107:1116-23).
Measuring MRD at those two time points “clearly separated the prognosis of patients not only in terms of disease-free survival but [in] overall survival,” she said. “So that’s why, for ALL, this has really become very important.”
In the United States, where flow-based cytometry is used more, it is necessary to find a properly equipped laboratory that can provide reliable results, she added. “For example, at our center we actually send our MRD to Fred Hutchinson [Cancer Center in Seattle].”
Johns Hopkins [Baltimore] also has such a lab, and both can arrange to accept send-outs, she said.
The other “really exciting thing” in regard to MRD in ALL is the recent approval of blinatumomab for MRD-positive ALL, she said.
In a study of 113 evaluable patients who were treated with the monoclonal antibody, 78% achieved complete molecular response (Blood. 2018 Apr 5;131:1522-31).
“And probably most importantly, when they looked at those patients who responded to blinatumomab in terms of MRD, these patients had, again, not only improved relapse-free survival but also increased overall survival,” she said. “I think this really explains why in ALL, we are measuring MRD and how it can really impact these patients.”
One of the remaining questions that will be important to address going forward is whether patients with MRD-positive ALL should continue to be considered for transplant; some of these studies have shown “very, very good outcomes” in patients who have not been transplanted, she noted.
Ph-like signature
Another important new prognostic feature in ALL is the presence of the Ph-like signature, a gene expression signature that was initially described in children with poor-risk ALL, and which looks a lot like Ph-positive disease.
“When they delved in further, they identified that this signature actually correlated with multiple different [kinase] fusions ... and it turns out that 20%-25% of young adults have this signature,” she said.
Since event-free survival in young adults with ALL is usually in the 65%-70% range, most of the remaining 30%-35% likely have this signature, she explained.
The kinase fusions associated with the Ph-like signature retain intact tyrosine kinase domains, and the spectrum of the fusions changes across age groups.
Treatments targeting some of these – for example, dasatinib for patients with a Ph-like dasatinib-sensitive kinase mutation – are being investigated.
Additionally, the Children’s Oncology Group has developed a clinically adaptable screening assay to identify the signature, she noted.
“So I would say that, and there is probably some difference in opinion, it is now becoming fairly standard that at diagnosis in an adult with ALL we’re sending the Ph-like signature,” she said. “And again, usually you’re going to have to send this out, and at our center we send it out to [Nationwide Children’s Hospital] in Columbus [Ohio].”
As important as it is to identify the Ph-like signature, given its association with poor prognosis, a number of questions remain, including whether transplant improves outcomes.
“The hope is it probably does, and that’s something that’s being evaluated in studies,” she said, noting that clinical studies are also specifically targeting these patients.
“So these patients should probably be enrolled on a clinical trial, because their outcome is clearly inferior,” she said.
Dr. Advani reported consultancy for Pfizer; research funding from Genzyme, Novartis, Pfizer, and Sigma Tau; and honoraria from Genzyme, Pfizer, and Sigma Tau. She is also on the speakers bureau for Sigma Tau.
CHICAGO – New recognition of the prognostic value of cytogenetic factors, minimal residual disease activity, and Philadelphia chromosome–like signature could improve the treatment and outcomes of acute lymphoblastic leukemia (ALL), according to Anjali Advani, MD.
CD20
About 80% of ALL is B-cell ALL and the majority of patients have pre–B-cell ALL, Dr. Advani, a hematologist and director of the inpatient leukemia program at the Cleveland Clinic, said at the American Society of Hematology Meeting on Hematologic Malignancies.
“And on the B lymphoblast, many antigens are expressed, including CD19, CD20, and CD52,” she said.
Rituximab, a drug often used for the treatment of lymphoma, is a chimeric monoclonal antibody against the protein CD20, which is expressed in 41% of ALL patients.
“Interestingly, CD20 expression in ALL has been associated with an adverse prognostic impact, which suggests that targeting this may potentially improve outcomes in these patients,” Dr. Advani said.
In fact, a recent randomized study by Sébastien Maury, MD, of the University of Paris-Est, and his colleagues, demonstrated that adding 16-18 doses of rituximab to a Berlin-Frankfurt-Münster (BFM)–based chemotherapy in Philadelphia chromosome (Ph)–negative patients aged 18-59 years with CD20-positive pre–B-cell ALL improved 2-year event-free survival from 52% to 65%. The data are consistent with those from prior studies, including a German study that showed a higher degree of minimal residual disease (MRD) negativity in patients treated with rituximab, she noted (N Engl J Med 2016;375:1044-53).
While the study by Dr. Maury and his colleagues didn’t look at MRD, that may offer an explanation for the improved event-free survival in their study, Dr. Advani suggested.
MRD
Minimal residual disease has become a standard part of practice in ALL, but pediatric ALL led the way in using early MRD measurement for risk-stratifying therapy, and it has taken a bit longer for it to be incorporated in the adult disease realm, Dr. Advani said.
Either flow cytometry or polymerase chain reaction (PCR) amplification can be used to measure MRD, she noted.
In one of the larger studies done in adults, researchers used PCR to look at MRD at two time points and stratified patients into three risk groups, including low, intermediate, and high risk (Blood. 2006 Feb 1;107:1116-23).
Measuring MRD at those two time points “clearly separated the prognosis of patients not only in terms of disease-free survival but [in] overall survival,” she said. “So that’s why, for ALL, this has really become very important.”
In the United States, where flow-based cytometry is used more, it is necessary to find a properly equipped laboratory that can provide reliable results, she added. “For example, at our center we actually send our MRD to Fred Hutchinson [Cancer Center in Seattle].”
Johns Hopkins [Baltimore] also has such a lab, and both can arrange to accept send-outs, she said.
The other “really exciting thing” in regard to MRD in ALL is the recent approval of blinatumomab for MRD-positive ALL, she said.
In a study of 113 evaluable patients who were treated with the monoclonal antibody, 78% achieved complete molecular response (Blood. 2018 Apr 5;131:1522-31).
“And probably most importantly, when they looked at those patients who responded to blinatumomab in terms of MRD, these patients had, again, not only improved relapse-free survival but also increased overall survival,” she said. “I think this really explains why in ALL, we are measuring MRD and how it can really impact these patients.”
One of the remaining questions that will be important to address going forward is whether patients with MRD-positive ALL should continue to be considered for transplant; some of these studies have shown “very, very good outcomes” in patients who have not been transplanted, she noted.
Ph-like signature
Another important new prognostic feature in ALL is the presence of the Ph-like signature, a gene expression signature that was initially described in children with poor-risk ALL, and which looks a lot like Ph-positive disease.
“When they delved in further, they identified that this signature actually correlated with multiple different [kinase] fusions ... and it turns out that 20%-25% of young adults have this signature,” she said.
Since event-free survival in young adults with ALL is usually in the 65%-70% range, most of the remaining 30%-35% likely have this signature, she explained.
The kinase fusions associated with the Ph-like signature retain intact tyrosine kinase domains, and the spectrum of the fusions changes across age groups.
Treatments targeting some of these – for example, dasatinib for patients with a Ph-like dasatinib-sensitive kinase mutation – are being investigated.
Additionally, the Children’s Oncology Group has developed a clinically adaptable screening assay to identify the signature, she noted.
“So I would say that, and there is probably some difference in opinion, it is now becoming fairly standard that at diagnosis in an adult with ALL we’re sending the Ph-like signature,” she said. “And again, usually you’re going to have to send this out, and at our center we send it out to [Nationwide Children’s Hospital] in Columbus [Ohio].”
As important as it is to identify the Ph-like signature, given its association with poor prognosis, a number of questions remain, including whether transplant improves outcomes.
“The hope is it probably does, and that’s something that’s being evaluated in studies,” she said, noting that clinical studies are also specifically targeting these patients.
“So these patients should probably be enrolled on a clinical trial, because their outcome is clearly inferior,” she said.
Dr. Advani reported consultancy for Pfizer; research funding from Genzyme, Novartis, Pfizer, and Sigma Tau; and honoraria from Genzyme, Pfizer, and Sigma Tau. She is also on the speakers bureau for Sigma Tau.
EXPERT ANALYSIS FROM MHM 2018
AP-1 plays key role in various AML subtypes, team says
The AP-1 transcription factor family is of “major importance” in acute myeloid leukemia (AML), according to researchers.
The team said they identified transcription factor networks specific to AML subtypes, which showed that leukemic growth is dependent upon certain transcription factors, and “the global activation of signaling pathways parallels a growth dependence on AP-1 activity in multiple types of AML.”
Constanze Bonifer, PhD, of the University of Birmingham in the U.K., and her colleagues conducted this research and detailed their findings in Nature Genetics.
The researchers noted that previous work revealed the existence of gene regulatory networks in different types of AML classified by gene expression and DNA methylation patterns.
“Our work now defines these networks in detail and shows that leukemic drivers determine the regulatory phenotype by establishing and maintaining specific gene regulatory and signaling networks that are distinct from those in normal cells,” Dr. Bonifer and her colleagues wrote.
The researchers combined data obtained via several analytic techniques to construct transcription factor networks in normal CD34+ cells and cells from AML patients with defined mutations, including RUNX1 mutations, t(8;21) translocations, mutations of both alleles of the CEBPA gene, and FLT3-ITD with or without NPM1 mutation.
The AP-1 family network was of “high regulatory relevance” for all AML subtypes evaluated, the team reported.
Follow-up in vitro and in vivo studies confirmed the importance of AP-1 for different AML subtypes.
In the in vitro study, the researchers transduced AML cells with a doxycycline-inducible version of a dominant-negative (dn) FOS protein.
“AP-1 is a heterodimer formed by members of the FOS, JUN, ATF, CREB, and JDP families of transcription factors,” the researchers wrote. “[T]hus, it is challenging to target by defined RNA interference approaches.”
Results of the in vitro study showed that induction of dnFOS, mediated by doxycycline, inhibited proliferation of t(8;21)+ Kasumi-1 cells and FLT3-ITD-expressing MV4-11 cells.
Induction of dnFOS also inhibited the colony-forming ability of primary CD34+ FLT3-ITD cells but not CD34+ hematopoietic stem and progenitor cells.
To evaluate the relevance of AP-1 for leukemia propagation in vivo, the researchers transplanted either of two cell lines—Kasumi-1 or MV4-11—expressing inducible dnFOS in immunodeficient mice.
With Kasumi-1, granulosarcomas developed in six of seven untreated control mice and two mice treated with doxycycline, neither of which expressed the inducible protein.
With MV4-11, doxycycline inhibited leukemia development, and untreated mice rapidly developed tumors.
The researchers declared no competing interests related to this work, which was funded by Bloodwise, Cancer Research UK, a Kay Kendall Clinical Training Fellowship, and an MRC/Leuka Clinical Training Fellowship.
The AP-1 transcription factor family is of “major importance” in acute myeloid leukemia (AML), according to researchers.
The team said they identified transcription factor networks specific to AML subtypes, which showed that leukemic growth is dependent upon certain transcription factors, and “the global activation of signaling pathways parallels a growth dependence on AP-1 activity in multiple types of AML.”
Constanze Bonifer, PhD, of the University of Birmingham in the U.K., and her colleagues conducted this research and detailed their findings in Nature Genetics.
The researchers noted that previous work revealed the existence of gene regulatory networks in different types of AML classified by gene expression and DNA methylation patterns.
“Our work now defines these networks in detail and shows that leukemic drivers determine the regulatory phenotype by establishing and maintaining specific gene regulatory and signaling networks that are distinct from those in normal cells,” Dr. Bonifer and her colleagues wrote.
The researchers combined data obtained via several analytic techniques to construct transcription factor networks in normal CD34+ cells and cells from AML patients with defined mutations, including RUNX1 mutations, t(8;21) translocations, mutations of both alleles of the CEBPA gene, and FLT3-ITD with or without NPM1 mutation.
The AP-1 family network was of “high regulatory relevance” for all AML subtypes evaluated, the team reported.
Follow-up in vitro and in vivo studies confirmed the importance of AP-1 for different AML subtypes.
In the in vitro study, the researchers transduced AML cells with a doxycycline-inducible version of a dominant-negative (dn) FOS protein.
“AP-1 is a heterodimer formed by members of the FOS, JUN, ATF, CREB, and JDP families of transcription factors,” the researchers wrote. “[T]hus, it is challenging to target by defined RNA interference approaches.”
Results of the in vitro study showed that induction of dnFOS, mediated by doxycycline, inhibited proliferation of t(8;21)+ Kasumi-1 cells and FLT3-ITD-expressing MV4-11 cells.
Induction of dnFOS also inhibited the colony-forming ability of primary CD34+ FLT3-ITD cells but not CD34+ hematopoietic stem and progenitor cells.
To evaluate the relevance of AP-1 for leukemia propagation in vivo, the researchers transplanted either of two cell lines—Kasumi-1 or MV4-11—expressing inducible dnFOS in immunodeficient mice.
With Kasumi-1, granulosarcomas developed in six of seven untreated control mice and two mice treated with doxycycline, neither of which expressed the inducible protein.
With MV4-11, doxycycline inhibited leukemia development, and untreated mice rapidly developed tumors.
The researchers declared no competing interests related to this work, which was funded by Bloodwise, Cancer Research UK, a Kay Kendall Clinical Training Fellowship, and an MRC/Leuka Clinical Training Fellowship.
The AP-1 transcription factor family is of “major importance” in acute myeloid leukemia (AML), according to researchers.
The team said they identified transcription factor networks specific to AML subtypes, which showed that leukemic growth is dependent upon certain transcription factors, and “the global activation of signaling pathways parallels a growth dependence on AP-1 activity in multiple types of AML.”
Constanze Bonifer, PhD, of the University of Birmingham in the U.K., and her colleagues conducted this research and detailed their findings in Nature Genetics.
The researchers noted that previous work revealed the existence of gene regulatory networks in different types of AML classified by gene expression and DNA methylation patterns.
“Our work now defines these networks in detail and shows that leukemic drivers determine the regulatory phenotype by establishing and maintaining specific gene regulatory and signaling networks that are distinct from those in normal cells,” Dr. Bonifer and her colleagues wrote.
The researchers combined data obtained via several analytic techniques to construct transcription factor networks in normal CD34+ cells and cells from AML patients with defined mutations, including RUNX1 mutations, t(8;21) translocations, mutations of both alleles of the CEBPA gene, and FLT3-ITD with or without NPM1 mutation.
The AP-1 family network was of “high regulatory relevance” for all AML subtypes evaluated, the team reported.
Follow-up in vitro and in vivo studies confirmed the importance of AP-1 for different AML subtypes.
In the in vitro study, the researchers transduced AML cells with a doxycycline-inducible version of a dominant-negative (dn) FOS protein.
“AP-1 is a heterodimer formed by members of the FOS, JUN, ATF, CREB, and JDP families of transcription factors,” the researchers wrote. “[T]hus, it is challenging to target by defined RNA interference approaches.”
Results of the in vitro study showed that induction of dnFOS, mediated by doxycycline, inhibited proliferation of t(8;21)+ Kasumi-1 cells and FLT3-ITD-expressing MV4-11 cells.
Induction of dnFOS also inhibited the colony-forming ability of primary CD34+ FLT3-ITD cells but not CD34+ hematopoietic stem and progenitor cells.
To evaluate the relevance of AP-1 for leukemia propagation in vivo, the researchers transplanted either of two cell lines—Kasumi-1 or MV4-11—expressing inducible dnFOS in immunodeficient mice.
With Kasumi-1, granulosarcomas developed in six of seven untreated control mice and two mice treated with doxycycline, neither of which expressed the inducible protein.
With MV4-11, doxycycline inhibited leukemia development, and untreated mice rapidly developed tumors.
The researchers declared no competing interests related to this work, which was funded by Bloodwise, Cancer Research UK, a Kay Kendall Clinical Training Fellowship, and an MRC/Leuka Clinical Training Fellowship.
Transcription factor plays key role in AML gene regulatory networks
The AP-1 transcription factor family, important in many tumor types, plays a major role in acute myeloid leukemia, according to researchers who conducted a comprehensive global analysis of gene regulatory networks involved in this disease.
This observation suggests new opportunities for targeted treatment of AML, according to the researchers, led by Peter N. Cockerill, PhD, and Constanze Bonifer, PhD, with the Institute of Cancer and Genomic Sciences, University of Birmingham, England.
“Induced and aberrantly expressed transcription factors are not bystanders, but are important for network maintenance and leukemic growth,” the investigators wrote in Nature Genetics.
Investigators combined data obtained via several different analytic techniques to construct transcription factor networks in normal CD34+ cells and cells from specific subgroups of subjects with defined mutations, including RUNX1 mutations, t(8;21) translocations, mutations of both alleles of the CEBPA gene, and FLT3-ITD with or without NPM1 mutation.
The AP-1 family network was of “high regulatory relevance” for all AML subtypes evaluated, the investigators reported.
Previous work revealed the existence of gene regulatory networks in different types of AML classified by gene expression and DNA-methylation patterns.
“Our work now defines these networks in detail, and shows that leukemic drivers determine the regulatory phenotype by establishing and maintaining specific gene regulatory and signaling networks that are distinct from those in normal cells,” the authors said in their report.
Follow-up in vitro and in vivo studies confirmed the importance of AP-1 for different AML subtypes.
In the in vitro study, investigators transduced AML cells with a doxycycline-inducible version of a dominant negative FOS protein.
“AP-1 is a heterodimer formed by members of the FOS, JUN, ATF, CREB and JDP families of transcription factors, thus it is challenging to target by defined RNA interference approaches,” the investigators explained.
Results of the in vitro study showed that induction of that protein, mediated by doxycycline, inhibited proliferation and colony-forming ability in AML cell lines.
To evaluate the relevance of AP-1 for leukemia propagation in vivo, they transplanted two different types of cells expressing inducible dominant negative FOS protein in immunodeficient mice.
For the first cell type, granulosarcomas developed in six out of seven mice in a control group, but in only two mice treated with doxycycline, neither of which expressed the inducible protein, suggesting that the transgene was silenced, according to the investigators. For the second cell type, doxycycline inhibited leukemia development, while untreated mice rapidly developed tumors.
“Taken together, these findings demonstrate the importance of AP-1 for several AML subtypes and emphasize the potential of transcriptional network analyses to predict transcription factors crucial for malignant propagation,” the investigators wrote.
They declared no competing interests related to their research, which was funded by Bloodwise, Cancer Research UK, a Kay Kendall Clinical Training Fellowship and a MRC/Leuka Clinical Training Fellowship.
SOURCE: Assi SA et al. Nat Genet. 2018 Nov 12. doi: 10.1038/s41588-018-0270-1.
The AP-1 transcription factor family, important in many tumor types, plays a major role in acute myeloid leukemia, according to researchers who conducted a comprehensive global analysis of gene regulatory networks involved in this disease.
This observation suggests new opportunities for targeted treatment of AML, according to the researchers, led by Peter N. Cockerill, PhD, and Constanze Bonifer, PhD, with the Institute of Cancer and Genomic Sciences, University of Birmingham, England.
“Induced and aberrantly expressed transcription factors are not bystanders, but are important for network maintenance and leukemic growth,” the investigators wrote in Nature Genetics.
Investigators combined data obtained via several different analytic techniques to construct transcription factor networks in normal CD34+ cells and cells from specific subgroups of subjects with defined mutations, including RUNX1 mutations, t(8;21) translocations, mutations of both alleles of the CEBPA gene, and FLT3-ITD with or without NPM1 mutation.
The AP-1 family network was of “high regulatory relevance” for all AML subtypes evaluated, the investigators reported.
Previous work revealed the existence of gene regulatory networks in different types of AML classified by gene expression and DNA-methylation patterns.
“Our work now defines these networks in detail, and shows that leukemic drivers determine the regulatory phenotype by establishing and maintaining specific gene regulatory and signaling networks that are distinct from those in normal cells,” the authors said in their report.
Follow-up in vitro and in vivo studies confirmed the importance of AP-1 for different AML subtypes.
In the in vitro study, investigators transduced AML cells with a doxycycline-inducible version of a dominant negative FOS protein.
“AP-1 is a heterodimer formed by members of the FOS, JUN, ATF, CREB and JDP families of transcription factors, thus it is challenging to target by defined RNA interference approaches,” the investigators explained.
Results of the in vitro study showed that induction of that protein, mediated by doxycycline, inhibited proliferation and colony-forming ability in AML cell lines.
To evaluate the relevance of AP-1 for leukemia propagation in vivo, they transplanted two different types of cells expressing inducible dominant negative FOS protein in immunodeficient mice.
For the first cell type, granulosarcomas developed in six out of seven mice in a control group, but in only two mice treated with doxycycline, neither of which expressed the inducible protein, suggesting that the transgene was silenced, according to the investigators. For the second cell type, doxycycline inhibited leukemia development, while untreated mice rapidly developed tumors.
“Taken together, these findings demonstrate the importance of AP-1 for several AML subtypes and emphasize the potential of transcriptional network analyses to predict transcription factors crucial for malignant propagation,” the investigators wrote.
They declared no competing interests related to their research, which was funded by Bloodwise, Cancer Research UK, a Kay Kendall Clinical Training Fellowship and a MRC/Leuka Clinical Training Fellowship.
SOURCE: Assi SA et al. Nat Genet. 2018 Nov 12. doi: 10.1038/s41588-018-0270-1.
The AP-1 transcription factor family, important in many tumor types, plays a major role in acute myeloid leukemia, according to researchers who conducted a comprehensive global analysis of gene regulatory networks involved in this disease.
This observation suggests new opportunities for targeted treatment of AML, according to the researchers, led by Peter N. Cockerill, PhD, and Constanze Bonifer, PhD, with the Institute of Cancer and Genomic Sciences, University of Birmingham, England.
“Induced and aberrantly expressed transcription factors are not bystanders, but are important for network maintenance and leukemic growth,” the investigators wrote in Nature Genetics.
Investigators combined data obtained via several different analytic techniques to construct transcription factor networks in normal CD34+ cells and cells from specific subgroups of subjects with defined mutations, including RUNX1 mutations, t(8;21) translocations, mutations of both alleles of the CEBPA gene, and FLT3-ITD with or without NPM1 mutation.
The AP-1 family network was of “high regulatory relevance” for all AML subtypes evaluated, the investigators reported.
Previous work revealed the existence of gene regulatory networks in different types of AML classified by gene expression and DNA-methylation patterns.
“Our work now defines these networks in detail, and shows that leukemic drivers determine the regulatory phenotype by establishing and maintaining specific gene regulatory and signaling networks that are distinct from those in normal cells,” the authors said in their report.
Follow-up in vitro and in vivo studies confirmed the importance of AP-1 for different AML subtypes.
In the in vitro study, investigators transduced AML cells with a doxycycline-inducible version of a dominant negative FOS protein.
“AP-1 is a heterodimer formed by members of the FOS, JUN, ATF, CREB and JDP families of transcription factors, thus it is challenging to target by defined RNA interference approaches,” the investigators explained.
Results of the in vitro study showed that induction of that protein, mediated by doxycycline, inhibited proliferation and colony-forming ability in AML cell lines.
To evaluate the relevance of AP-1 for leukemia propagation in vivo, they transplanted two different types of cells expressing inducible dominant negative FOS protein in immunodeficient mice.
For the first cell type, granulosarcomas developed in six out of seven mice in a control group, but in only two mice treated with doxycycline, neither of which expressed the inducible protein, suggesting that the transgene was silenced, according to the investigators. For the second cell type, doxycycline inhibited leukemia development, while untreated mice rapidly developed tumors.
“Taken together, these findings demonstrate the importance of AP-1 for several AML subtypes and emphasize the potential of transcriptional network analyses to predict transcription factors crucial for malignant propagation,” the investigators wrote.
They declared no competing interests related to their research, which was funded by Bloodwise, Cancer Research UK, a Kay Kendall Clinical Training Fellowship and a MRC/Leuka Clinical Training Fellowship.
SOURCE: Assi SA et al. Nat Genet. 2018 Nov 12. doi: 10.1038/s41588-018-0270-1.
FROM NATURE GENETICS
Key clinical point:
Major finding: The AP-1 factor family gene regulatory network was of high regulatory relevance in multiple subtypes of AML with defined mutations.
Study details: Analysis of normal CD34+ cells and cells from AML subjects.
Disclosures: Funding came from Bloodwise and Cancer Research UK, among other sources. The researchers reported having no competing financial interests.
Source: Assi SA et al. Nat Genet. 2018 Nov 12. doi: 10.1038/s41588-018-0270-1.
Financial burden of blood cancers in the U.S.
An analysis of more than 2,000 U.S. patients with blood cancers revealed an average healthcare cost of almost $157,000 in the first year after diagnosis.
Costs were highest for acute leukemia patients—almost triple the average for all blood cancers.
Out-of-pocket (OOP) costs were initially highest for acute leukemia patients. However, over time, OOP costs became highest for patients with multiple myeloma.
These results are included in a report commissioned by the Leukemia & Lymphoma Society and prepared by the actuarial firm Milliman.
The report is based on data from the Truven Health MarketScan commercial claims databases.
The cost figures are drawn from data for 2,332 patients, ages 18 to 64, who were diagnosed with blood cancer in 2014 and followed through 2016. This includes the following:
- 1,468 patients with lymphoma
- 286 with chronic leukemia
- 282 with multiple myeloma
- 148 with acute leukemia
- 148 with bone marrow disorders (myelodysplastic syndromes).
The average allowed spending—the amount paid by the payer and patient combined—in the first 12 months after diagnosis was:
- $156,845 overall
- $463,414 for acute leukemia
- $213,879 for multiple myeloma
- $133,744 for bone marrow disorders
- $130,545 for lymphoma
- $88,913 for chronic leukemia.
Differences in OOP costs were smaller, although OOP spending was 32% higher for acute leukemia patients than the overall average.
Average OOP costs—which include coinsurance, copay, and deductible—in the first 12 months after diagnosis were:
- $3,877 overall
- $5,147 for acute leukemia
- $4,849 for multiple myeloma
- $3,695 for lymphoma
- $3,480 for chronic leukemia
- $3,336 for bone marrow disorders.
Although OOP costs were initially highest for acute leukemia patients, over time, costs for multiple myeloma patients became the highest.
The average OOP costs in the month of diagnosis were $1,637 for acute leukemia patients and $1,210 for multiple myeloma patients.
The total accumulated OOP costs 3 years after diagnosis were $8,797 for acute leukemia and $9,127 for multiple myeloma. For the other blood cancers, the average 3-year accumulated OOP costs were under $7,800.
The Leukemia & Lymphoma Society received support from Pfizer, Genentech, and Amgen for this work.
An analysis of more than 2,000 U.S. patients with blood cancers revealed an average healthcare cost of almost $157,000 in the first year after diagnosis.
Costs were highest for acute leukemia patients—almost triple the average for all blood cancers.
Out-of-pocket (OOP) costs were initially highest for acute leukemia patients. However, over time, OOP costs became highest for patients with multiple myeloma.
These results are included in a report commissioned by the Leukemia & Lymphoma Society and prepared by the actuarial firm Milliman.
The report is based on data from the Truven Health MarketScan commercial claims databases.
The cost figures are drawn from data for 2,332 patients, ages 18 to 64, who were diagnosed with blood cancer in 2014 and followed through 2016. This includes the following:
- 1,468 patients with lymphoma
- 286 with chronic leukemia
- 282 with multiple myeloma
- 148 with acute leukemia
- 148 with bone marrow disorders (myelodysplastic syndromes).
The average allowed spending—the amount paid by the payer and patient combined—in the first 12 months after diagnosis was:
- $156,845 overall
- $463,414 for acute leukemia
- $213,879 for multiple myeloma
- $133,744 for bone marrow disorders
- $130,545 for lymphoma
- $88,913 for chronic leukemia.
Differences in OOP costs were smaller, although OOP spending was 32% higher for acute leukemia patients than the overall average.
Average OOP costs—which include coinsurance, copay, and deductible—in the first 12 months after diagnosis were:
- $3,877 overall
- $5,147 for acute leukemia
- $4,849 for multiple myeloma
- $3,695 for lymphoma
- $3,480 for chronic leukemia
- $3,336 for bone marrow disorders.
Although OOP costs were initially highest for acute leukemia patients, over time, costs for multiple myeloma patients became the highest.
The average OOP costs in the month of diagnosis were $1,637 for acute leukemia patients and $1,210 for multiple myeloma patients.
The total accumulated OOP costs 3 years after diagnosis were $8,797 for acute leukemia and $9,127 for multiple myeloma. For the other blood cancers, the average 3-year accumulated OOP costs were under $7,800.
The Leukemia & Lymphoma Society received support from Pfizer, Genentech, and Amgen for this work.
An analysis of more than 2,000 U.S. patients with blood cancers revealed an average healthcare cost of almost $157,000 in the first year after diagnosis.
Costs were highest for acute leukemia patients—almost triple the average for all blood cancers.
Out-of-pocket (OOP) costs were initially highest for acute leukemia patients. However, over time, OOP costs became highest for patients with multiple myeloma.
These results are included in a report commissioned by the Leukemia & Lymphoma Society and prepared by the actuarial firm Milliman.
The report is based on data from the Truven Health MarketScan commercial claims databases.
The cost figures are drawn from data for 2,332 patients, ages 18 to 64, who were diagnosed with blood cancer in 2014 and followed through 2016. This includes the following:
- 1,468 patients with lymphoma
- 286 with chronic leukemia
- 282 with multiple myeloma
- 148 with acute leukemia
- 148 with bone marrow disorders (myelodysplastic syndromes).
The average allowed spending—the amount paid by the payer and patient combined—in the first 12 months after diagnosis was:
- $156,845 overall
- $463,414 for acute leukemia
- $213,879 for multiple myeloma
- $133,744 for bone marrow disorders
- $130,545 for lymphoma
- $88,913 for chronic leukemia.
Differences in OOP costs were smaller, although OOP spending was 32% higher for acute leukemia patients than the overall average.
Average OOP costs—which include coinsurance, copay, and deductible—in the first 12 months after diagnosis were:
- $3,877 overall
- $5,147 for acute leukemia
- $4,849 for multiple myeloma
- $3,695 for lymphoma
- $3,480 for chronic leukemia
- $3,336 for bone marrow disorders.
Although OOP costs were initially highest for acute leukemia patients, over time, costs for multiple myeloma patients became the highest.
The average OOP costs in the month of diagnosis were $1,637 for acute leukemia patients and $1,210 for multiple myeloma patients.
The total accumulated OOP costs 3 years after diagnosis were $8,797 for acute leukemia and $9,127 for multiple myeloma. For the other blood cancers, the average 3-year accumulated OOP costs were under $7,800.
The Leukemia & Lymphoma Society received support from Pfizer, Genentech, and Amgen for this work.
‘Compelling’ new target found for monocytic AML
Efforts to determine why immune checkpoint blockade is not successful in treating leukemia have resulted in a “compelling” new target to treat monocytic acute myeloid leukemia (AML), according to researchers.
They discovered that leukocyte immunoglobulin-like receptor B4 (LILRB4), a marker of monocytic leukemia, creates an immunosuppressive microenvironment by mediating T-cell suppression.
Using a mouse model and human cells, the research team showed that LILRB4 supports tumor infiltration into tissues and suppresses T-cell activity through a signaling pathway involving APOE, LILRB4, SHP-2, uPAR, and ARG1.
Senior author Chengcheng “Alec” Zhang, PhD, of the University of Texas Southwestern Medical Center in Dallas, and his colleagues reported their findings in Nature.
The team first compared surface expression of LILRB4 on normal monocytes and neoplastic monocytes from 105 AML patient samples. They observed that LILRB4 levels were higher on monocytic AML cells than on normal monocytes.
The researchers then tested whether LILRB4 expressed on AML cells suppressed T cells. They cultured LILRB4-positive leukemia cells, LILRB4-negative leukemia cells, and normal hematopoietic cells with either autologous T cells or T cells from healthy donors. The team determined that only LILRB4-positive monocytic AML cells substantially suppressed T-cell proliferation.
When the researchers knocked out LILRB4, the ability of AML cells to suppress T cells was reduced and could be restored with forced expression of wild-type LILRB4. Additionally, LILRB4-mediated T-cell inhibition could be reversed by LILRB4-blocking antibodies.
The team then used a humanized mouse xenograft model and an immunocompetent mouse model to investigate LILRB4 function in immune checkpoint blockade. Blocking LILRB4 lowered tumor burden and prolonged survival in the mice.
The researchers performed numerous in vitro and in vivo experiments and observed that antibody blockade of LILRB4 shrank tumors and decreased leukemic infiltration into internal organs, including the bone marrow, liver, and brain.
And so the team hypothesized that LILRB4 promotes leukemia infiltration in addition to inhibiting T cells.
To test the hypothesis, they performed transendothelial migration and homing assays and monitored leukemia infiltration relative to LILRB4 expression on leukemia cells. They observed that LILRB4-mediated migration enhanced extramedullary infiltration of monocytic AML cells, thus contributing to immune evasion.
The researchers also found that APOE protein activated the immune inhibitory receptor LILRB4.
To ascertain whether suppression of T cells by LILRB4 depends on APOE, the team co-cultured T cells with control or human AML cells with APOE knocked out.
Through a series of experiments, they determined that APOE is an extracellular binding protein of LILRB4 and that APOE activates LILRB4 to support T-cell proliferation and AML cell migration.
The researchers believe that targeting LILRB4 may have minimal toxicity. This is because LILRB4 expression on normal monocytic cells is limited, LILRB4 signaling may differ in leukemia cells, and LILRB4 blockade did not significantly interfere with normal hematopoietic function.
Dr. Zhang anticipates that if the preclinical studies go well, clinical trials could begin as early as next year.
The University of Texas System has exclusively licensed LILRB4-related patent applications to California-based Immune-Onc Therapeutics Inc., which contributed to the research and is conducting preclinical studies.
Dr. Zhang and another author are scientific advisory board members with Immune-Onc Therapeutics. Two other authors are employees of and hold equities in Immune-Onc Therapeutics.
The researchers received additional funding for this work from the National Cancer Institute, Leukemia & Lymphoma Society, the March of Dimes, the Cancer Prevention and Research Institute of Texas, the Robert A. Welch Foundation, the National Natural Science Foundation of China, the National Basic Research Program of China, and the China Scholarship Council.
Efforts to determine why immune checkpoint blockade is not successful in treating leukemia have resulted in a “compelling” new target to treat monocytic acute myeloid leukemia (AML), according to researchers.
They discovered that leukocyte immunoglobulin-like receptor B4 (LILRB4), a marker of monocytic leukemia, creates an immunosuppressive microenvironment by mediating T-cell suppression.
Using a mouse model and human cells, the research team showed that LILRB4 supports tumor infiltration into tissues and suppresses T-cell activity through a signaling pathway involving APOE, LILRB4, SHP-2, uPAR, and ARG1.
Senior author Chengcheng “Alec” Zhang, PhD, of the University of Texas Southwestern Medical Center in Dallas, and his colleagues reported their findings in Nature.
The team first compared surface expression of LILRB4 on normal monocytes and neoplastic monocytes from 105 AML patient samples. They observed that LILRB4 levels were higher on monocytic AML cells than on normal monocytes.
The researchers then tested whether LILRB4 expressed on AML cells suppressed T cells. They cultured LILRB4-positive leukemia cells, LILRB4-negative leukemia cells, and normal hematopoietic cells with either autologous T cells or T cells from healthy donors. The team determined that only LILRB4-positive monocytic AML cells substantially suppressed T-cell proliferation.
When the researchers knocked out LILRB4, the ability of AML cells to suppress T cells was reduced and could be restored with forced expression of wild-type LILRB4. Additionally, LILRB4-mediated T-cell inhibition could be reversed by LILRB4-blocking antibodies.
The team then used a humanized mouse xenograft model and an immunocompetent mouse model to investigate LILRB4 function in immune checkpoint blockade. Blocking LILRB4 lowered tumor burden and prolonged survival in the mice.
The researchers performed numerous in vitro and in vivo experiments and observed that antibody blockade of LILRB4 shrank tumors and decreased leukemic infiltration into internal organs, including the bone marrow, liver, and brain.
And so the team hypothesized that LILRB4 promotes leukemia infiltration in addition to inhibiting T cells.
To test the hypothesis, they performed transendothelial migration and homing assays and monitored leukemia infiltration relative to LILRB4 expression on leukemia cells. They observed that LILRB4-mediated migration enhanced extramedullary infiltration of monocytic AML cells, thus contributing to immune evasion.
The researchers also found that APOE protein activated the immune inhibitory receptor LILRB4.
To ascertain whether suppression of T cells by LILRB4 depends on APOE, the team co-cultured T cells with control or human AML cells with APOE knocked out.
Through a series of experiments, they determined that APOE is an extracellular binding protein of LILRB4 and that APOE activates LILRB4 to support T-cell proliferation and AML cell migration.
The researchers believe that targeting LILRB4 may have minimal toxicity. This is because LILRB4 expression on normal monocytic cells is limited, LILRB4 signaling may differ in leukemia cells, and LILRB4 blockade did not significantly interfere with normal hematopoietic function.
Dr. Zhang anticipates that if the preclinical studies go well, clinical trials could begin as early as next year.
The University of Texas System has exclusively licensed LILRB4-related patent applications to California-based Immune-Onc Therapeutics Inc., which contributed to the research and is conducting preclinical studies.
Dr. Zhang and another author are scientific advisory board members with Immune-Onc Therapeutics. Two other authors are employees of and hold equities in Immune-Onc Therapeutics.
The researchers received additional funding for this work from the National Cancer Institute, Leukemia & Lymphoma Society, the March of Dimes, the Cancer Prevention and Research Institute of Texas, the Robert A. Welch Foundation, the National Natural Science Foundation of China, the National Basic Research Program of China, and the China Scholarship Council.
Efforts to determine why immune checkpoint blockade is not successful in treating leukemia have resulted in a “compelling” new target to treat monocytic acute myeloid leukemia (AML), according to researchers.
They discovered that leukocyte immunoglobulin-like receptor B4 (LILRB4), a marker of monocytic leukemia, creates an immunosuppressive microenvironment by mediating T-cell suppression.
Using a mouse model and human cells, the research team showed that LILRB4 supports tumor infiltration into tissues and suppresses T-cell activity through a signaling pathway involving APOE, LILRB4, SHP-2, uPAR, and ARG1.
Senior author Chengcheng “Alec” Zhang, PhD, of the University of Texas Southwestern Medical Center in Dallas, and his colleagues reported their findings in Nature.
The team first compared surface expression of LILRB4 on normal monocytes and neoplastic monocytes from 105 AML patient samples. They observed that LILRB4 levels were higher on monocytic AML cells than on normal monocytes.
The researchers then tested whether LILRB4 expressed on AML cells suppressed T cells. They cultured LILRB4-positive leukemia cells, LILRB4-negative leukemia cells, and normal hematopoietic cells with either autologous T cells or T cells from healthy donors. The team determined that only LILRB4-positive monocytic AML cells substantially suppressed T-cell proliferation.
When the researchers knocked out LILRB4, the ability of AML cells to suppress T cells was reduced and could be restored with forced expression of wild-type LILRB4. Additionally, LILRB4-mediated T-cell inhibition could be reversed by LILRB4-blocking antibodies.
The team then used a humanized mouse xenograft model and an immunocompetent mouse model to investigate LILRB4 function in immune checkpoint blockade. Blocking LILRB4 lowered tumor burden and prolonged survival in the mice.
The researchers performed numerous in vitro and in vivo experiments and observed that antibody blockade of LILRB4 shrank tumors and decreased leukemic infiltration into internal organs, including the bone marrow, liver, and brain.
And so the team hypothesized that LILRB4 promotes leukemia infiltration in addition to inhibiting T cells.
To test the hypothesis, they performed transendothelial migration and homing assays and monitored leukemia infiltration relative to LILRB4 expression on leukemia cells. They observed that LILRB4-mediated migration enhanced extramedullary infiltration of monocytic AML cells, thus contributing to immune evasion.
The researchers also found that APOE protein activated the immune inhibitory receptor LILRB4.
To ascertain whether suppression of T cells by LILRB4 depends on APOE, the team co-cultured T cells with control or human AML cells with APOE knocked out.
Through a series of experiments, they determined that APOE is an extracellular binding protein of LILRB4 and that APOE activates LILRB4 to support T-cell proliferation and AML cell migration.
The researchers believe that targeting LILRB4 may have minimal toxicity. This is because LILRB4 expression on normal monocytic cells is limited, LILRB4 signaling may differ in leukemia cells, and LILRB4 blockade did not significantly interfere with normal hematopoietic function.
Dr. Zhang anticipates that if the preclinical studies go well, clinical trials could begin as early as next year.
The University of Texas System has exclusively licensed LILRB4-related patent applications to California-based Immune-Onc Therapeutics Inc., which contributed to the research and is conducting preclinical studies.
Dr. Zhang and another author are scientific advisory board members with Immune-Onc Therapeutics. Two other authors are employees of and hold equities in Immune-Onc Therapeutics.
The researchers received additional funding for this work from the National Cancer Institute, Leukemia & Lymphoma Society, the March of Dimes, the Cancer Prevention and Research Institute of Texas, the Robert A. Welch Foundation, the National Natural Science Foundation of China, the National Basic Research Program of China, and the China Scholarship Council.
‘Encouraging’ phase 2 results in rel/ref AML
The combination of azacitidine and nivolumab produced “encouraging” results in a phase 2 trial of patients with relapsed or refractory acute myeloid leukemia (AML), according to researchers.
The overall response rate was 33%, and the median overall survival (OS) was 6.3 months.
However, the researchers identified factors associated with improved response and survival that they believe could be used to select patients for this treatment.
A quarter of patients on this trial had immune-related adverse events (AEs) that were considered related to treatment, and two patients died of AEs that may have been treatment-related.
Naval Daver, MD, of The University of Texas MD Anderson Cancer Center in Houston, and his colleagues reported these results in Cancer Discovery.
The trial included 70 patients with a median age of 70 (range, 22-90). Fifty-six percent had de novo AML, and 44% had secondary AML.
The median number of prior therapies was 2 (range, 1 to 7). Sixty-four percent of patients had received hypomethylating agents, 47% had received targeted therapies, and 19% had received allogeneic stem cell transplant (SCT).
For this trial, patients received azacitidine at 75 mg/m2 on days 1 to 7 and nivolumab at 3 mg/kg on days 1 and 14 of each cycle. The median number of cycles was 3 (range, 1 to 25).
Patients had a median time on study of 3.5 months (range, 0.3 to 26.3 months). Reasons for discontinuation included primary refractory disease (n=27), relapse after initial response (n=19), death (n=16), proceeding to SCT (n=3), and patient preference (n=3).
Safety
The most common treatment-related, non-hematologic AEs were constipation (26%), diarrhea (20%), pneumonitis (13%), nausea (11%), and lung infection (11%).
The rate of immune-related AEs was 25% (n=18), with grade 2-4 immune-related AEs occurring in 16 patients (8 with grade 3-4). Fourteen of these patients responded to steroids and were safely re-challenged with nivolumab, according to the researchers.
Nine patients (13%) discontinued nivolumab (but continued with azacitidine) due to AEs—pneumonitis (n=7), cytokine release syndrome (n=1), and immune nephritis (n=1).
Two patients died of AEs that were considered possibly related to treatment. One death was due to progressive pneumonia/pneumonitis, and one was due to hemophagocytosis lymphohistiocytosis.
Response
The overall response rate was 33% (n=23). Four patients had a complete response (CR), and 11 had a CR with incomplete count recovery (CRi).
One patient had a partial response, and seven had hematologic improvement in one or more parameter maintained for more than 6 months. Six patients had stable disease lasting more than 6 months.
The researchers noted that the response rate was higher among patients who had not received prior treatment with hypomethylating agents. In addition, a higher frequency of pre-therapy CD3 and CD8 cells in the bone marrow or peripheral blood appeared to predict response.
“In particular, CD3 appeared to have a high sensitivity and specificity rate for predicting response, indicating it might serve as a reliable biomarker for selecting patients for this combination therapy,” Dr. Daver said.
Survival
At a median follow-up of 21.4 months, 81% of patients (n=57) had died. Sixteen patients died on study treatment, and 41 died after discontinuation.
The median OS was 6.3 months, and the median event-free survival was 4.5 months.
The median OS was 16.1 months in patients with CR/CRi, partial response, hematologic improvement, or stable disease and 4.1 months in non-responders (P<0.0001). This difference was still significant after the researchers censored the three patients who had gone on to SCT in CR/CRi (P<0.001).
The researchers also found that being in first salvage was associated with improved OS in a univariate analysis and in a comparison with historical controls.
Dr. Daver and his colleagues concluded that azacitidine and nivolumab “produced an encouraging response rate and overall survival” in patients with relapsed/refractory AML.
“We believe that implementation of clinical and immune biomarkers to select patients are likely to yield further improved outcomes with these types of therapies in AML,” Dr. Daver noted.
This research was supported by Bristol-Myers Squibb, MD Anderson, and the Dick Clark Immunotherapy Research Fund. In addition, individual researchers reported financial relationships with Bristol-Myers Squibb.
The combination of azacitidine and nivolumab produced “encouraging” results in a phase 2 trial of patients with relapsed or refractory acute myeloid leukemia (AML), according to researchers.
The overall response rate was 33%, and the median overall survival (OS) was 6.3 months.
However, the researchers identified factors associated with improved response and survival that they believe could be used to select patients for this treatment.
A quarter of patients on this trial had immune-related adverse events (AEs) that were considered related to treatment, and two patients died of AEs that may have been treatment-related.
Naval Daver, MD, of The University of Texas MD Anderson Cancer Center in Houston, and his colleagues reported these results in Cancer Discovery.
The trial included 70 patients with a median age of 70 (range, 22-90). Fifty-six percent had de novo AML, and 44% had secondary AML.
The median number of prior therapies was 2 (range, 1 to 7). Sixty-four percent of patients had received hypomethylating agents, 47% had received targeted therapies, and 19% had received allogeneic stem cell transplant (SCT).
For this trial, patients received azacitidine at 75 mg/m2 on days 1 to 7 and nivolumab at 3 mg/kg on days 1 and 14 of each cycle. The median number of cycles was 3 (range, 1 to 25).
Patients had a median time on study of 3.5 months (range, 0.3 to 26.3 months). Reasons for discontinuation included primary refractory disease (n=27), relapse after initial response (n=19), death (n=16), proceeding to SCT (n=3), and patient preference (n=3).
Safety
The most common treatment-related, non-hematologic AEs were constipation (26%), diarrhea (20%), pneumonitis (13%), nausea (11%), and lung infection (11%).
The rate of immune-related AEs was 25% (n=18), with grade 2-4 immune-related AEs occurring in 16 patients (8 with grade 3-4). Fourteen of these patients responded to steroids and were safely re-challenged with nivolumab, according to the researchers.
Nine patients (13%) discontinued nivolumab (but continued with azacitidine) due to AEs—pneumonitis (n=7), cytokine release syndrome (n=1), and immune nephritis (n=1).
Two patients died of AEs that were considered possibly related to treatment. One death was due to progressive pneumonia/pneumonitis, and one was due to hemophagocytosis lymphohistiocytosis.
Response
The overall response rate was 33% (n=23). Four patients had a complete response (CR), and 11 had a CR with incomplete count recovery (CRi).
One patient had a partial response, and seven had hematologic improvement in one or more parameter maintained for more than 6 months. Six patients had stable disease lasting more than 6 months.
The researchers noted that the response rate was higher among patients who had not received prior treatment with hypomethylating agents. In addition, a higher frequency of pre-therapy CD3 and CD8 cells in the bone marrow or peripheral blood appeared to predict response.
“In particular, CD3 appeared to have a high sensitivity and specificity rate for predicting response, indicating it might serve as a reliable biomarker for selecting patients for this combination therapy,” Dr. Daver said.
Survival
At a median follow-up of 21.4 months, 81% of patients (n=57) had died. Sixteen patients died on study treatment, and 41 died after discontinuation.
The median OS was 6.3 months, and the median event-free survival was 4.5 months.
The median OS was 16.1 months in patients with CR/CRi, partial response, hematologic improvement, or stable disease and 4.1 months in non-responders (P<0.0001). This difference was still significant after the researchers censored the three patients who had gone on to SCT in CR/CRi (P<0.001).
The researchers also found that being in first salvage was associated with improved OS in a univariate analysis and in a comparison with historical controls.
Dr. Daver and his colleagues concluded that azacitidine and nivolumab “produced an encouraging response rate and overall survival” in patients with relapsed/refractory AML.
“We believe that implementation of clinical and immune biomarkers to select patients are likely to yield further improved outcomes with these types of therapies in AML,” Dr. Daver noted.
This research was supported by Bristol-Myers Squibb, MD Anderson, and the Dick Clark Immunotherapy Research Fund. In addition, individual researchers reported financial relationships with Bristol-Myers Squibb.
The combination of azacitidine and nivolumab produced “encouraging” results in a phase 2 trial of patients with relapsed or refractory acute myeloid leukemia (AML), according to researchers.
The overall response rate was 33%, and the median overall survival (OS) was 6.3 months.
However, the researchers identified factors associated with improved response and survival that they believe could be used to select patients for this treatment.
A quarter of patients on this trial had immune-related adverse events (AEs) that were considered related to treatment, and two patients died of AEs that may have been treatment-related.
Naval Daver, MD, of The University of Texas MD Anderson Cancer Center in Houston, and his colleagues reported these results in Cancer Discovery.
The trial included 70 patients with a median age of 70 (range, 22-90). Fifty-six percent had de novo AML, and 44% had secondary AML.
The median number of prior therapies was 2 (range, 1 to 7). Sixty-four percent of patients had received hypomethylating agents, 47% had received targeted therapies, and 19% had received allogeneic stem cell transplant (SCT).
For this trial, patients received azacitidine at 75 mg/m2 on days 1 to 7 and nivolumab at 3 mg/kg on days 1 and 14 of each cycle. The median number of cycles was 3 (range, 1 to 25).
Patients had a median time on study of 3.5 months (range, 0.3 to 26.3 months). Reasons for discontinuation included primary refractory disease (n=27), relapse after initial response (n=19), death (n=16), proceeding to SCT (n=3), and patient preference (n=3).
Safety
The most common treatment-related, non-hematologic AEs were constipation (26%), diarrhea (20%), pneumonitis (13%), nausea (11%), and lung infection (11%).
The rate of immune-related AEs was 25% (n=18), with grade 2-4 immune-related AEs occurring in 16 patients (8 with grade 3-4). Fourteen of these patients responded to steroids and were safely re-challenged with nivolumab, according to the researchers.
Nine patients (13%) discontinued nivolumab (but continued with azacitidine) due to AEs—pneumonitis (n=7), cytokine release syndrome (n=1), and immune nephritis (n=1).
Two patients died of AEs that were considered possibly related to treatment. One death was due to progressive pneumonia/pneumonitis, and one was due to hemophagocytosis lymphohistiocytosis.
Response
The overall response rate was 33% (n=23). Four patients had a complete response (CR), and 11 had a CR with incomplete count recovery (CRi).
One patient had a partial response, and seven had hematologic improvement in one or more parameter maintained for more than 6 months. Six patients had stable disease lasting more than 6 months.
The researchers noted that the response rate was higher among patients who had not received prior treatment with hypomethylating agents. In addition, a higher frequency of pre-therapy CD3 and CD8 cells in the bone marrow or peripheral blood appeared to predict response.
“In particular, CD3 appeared to have a high sensitivity and specificity rate for predicting response, indicating it might serve as a reliable biomarker for selecting patients for this combination therapy,” Dr. Daver said.
Survival
At a median follow-up of 21.4 months, 81% of patients (n=57) had died. Sixteen patients died on study treatment, and 41 died after discontinuation.
The median OS was 6.3 months, and the median event-free survival was 4.5 months.
The median OS was 16.1 months in patients with CR/CRi, partial response, hematologic improvement, or stable disease and 4.1 months in non-responders (P<0.0001). This difference was still significant after the researchers censored the three patients who had gone on to SCT in CR/CRi (P<0.001).
The researchers also found that being in first salvage was associated with improved OS in a univariate analysis and in a comparison with historical controls.
Dr. Daver and his colleagues concluded that azacitidine and nivolumab “produced an encouraging response rate and overall survival” in patients with relapsed/refractory AML.
“We believe that implementation of clinical and immune biomarkers to select patients are likely to yield further improved outcomes with these types of therapies in AML,” Dr. Daver noted.
This research was supported by Bristol-Myers Squibb, MD Anderson, and the Dick Clark Immunotherapy Research Fund. In addition, individual researchers reported financial relationships with Bristol-Myers Squibb.
Report details financial burden of blood cancers
with costs for acute leukemia almost tripling that amount, according to a new report from the Leukemia & Lymphoma Society (LLS).
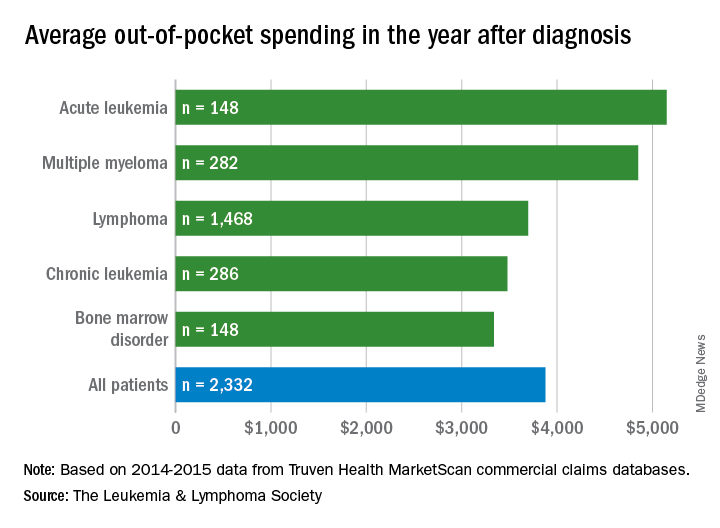
Total allowed cost – the average amount paid by the insurer and patient combined – for acute leukemia was more than $463,000 for the 12 months after initial diagnosis. Averages for the other four cancers included in the analysis came in at $214,000 for multiple myeloma, $134,000 for bone marrow disorders, $131,000 for lymphoma, and $89,000 for chronic leukemia, the LLS said.
The cost figures are drawn from claims data for 2,332 patients diagnosed in 2014.
Differences in out-of-pocket (OOP) costs were smaller, with the average for all patients at almost $3,900 in the year after diagnosis and acute leukemia coming in the highest at $5,100. Over time, however, OOP costs for multiple myeloma patients became the highest, totaling $9,100 for the 3 years after diagnosis, compared with $8,800 for acute leukemia and an average of less than $7,800 for the other blood cancers, the LLS said in the report, which was prepared by the actuarial firm Milliman.
OOP costs also varied by the type of plan. Patients in high-deductible plans averaged nearly $5,400 for the first year after diagnosis, compared with $3,300 for those with traditional insurance, the LLS noted. For acute leukemia, the OOP costs of high-deductible plans were more than twice as high as those of traditional plans.
The study was based on data for adults aged 18-64 years from the Truven Health MarketScan commercial claims databases for the years from 2013 to 2016. The LLS received support for the study from Pfizer, Genentech, and Amgen.
with costs for acute leukemia almost tripling that amount, according to a new report from the Leukemia & Lymphoma Society (LLS).

Total allowed cost – the average amount paid by the insurer and patient combined – for acute leukemia was more than $463,000 for the 12 months after initial diagnosis. Averages for the other four cancers included in the analysis came in at $214,000 for multiple myeloma, $134,000 for bone marrow disorders, $131,000 for lymphoma, and $89,000 for chronic leukemia, the LLS said.
The cost figures are drawn from claims data for 2,332 patients diagnosed in 2014.
Differences in out-of-pocket (OOP) costs were smaller, with the average for all patients at almost $3,900 in the year after diagnosis and acute leukemia coming in the highest at $5,100. Over time, however, OOP costs for multiple myeloma patients became the highest, totaling $9,100 for the 3 years after diagnosis, compared with $8,800 for acute leukemia and an average of less than $7,800 for the other blood cancers, the LLS said in the report, which was prepared by the actuarial firm Milliman.
OOP costs also varied by the type of plan. Patients in high-deductible plans averaged nearly $5,400 for the first year after diagnosis, compared with $3,300 for those with traditional insurance, the LLS noted. For acute leukemia, the OOP costs of high-deductible plans were more than twice as high as those of traditional plans.
The study was based on data for adults aged 18-64 years from the Truven Health MarketScan commercial claims databases for the years from 2013 to 2016. The LLS received support for the study from Pfizer, Genentech, and Amgen.
with costs for acute leukemia almost tripling that amount, according to a new report from the Leukemia & Lymphoma Society (LLS).

Total allowed cost – the average amount paid by the insurer and patient combined – for acute leukemia was more than $463,000 for the 12 months after initial diagnosis. Averages for the other four cancers included in the analysis came in at $214,000 for multiple myeloma, $134,000 for bone marrow disorders, $131,000 for lymphoma, and $89,000 for chronic leukemia, the LLS said.
The cost figures are drawn from claims data for 2,332 patients diagnosed in 2014.
Differences in out-of-pocket (OOP) costs were smaller, with the average for all patients at almost $3,900 in the year after diagnosis and acute leukemia coming in the highest at $5,100. Over time, however, OOP costs for multiple myeloma patients became the highest, totaling $9,100 for the 3 years after diagnosis, compared with $8,800 for acute leukemia and an average of less than $7,800 for the other blood cancers, the LLS said in the report, which was prepared by the actuarial firm Milliman.
OOP costs also varied by the type of plan. Patients in high-deductible plans averaged nearly $5,400 for the first year after diagnosis, compared with $3,300 for those with traditional insurance, the LLS noted. For acute leukemia, the OOP costs of high-deductible plans were more than twice as high as those of traditional plans.
The study was based on data for adults aged 18-64 years from the Truven Health MarketScan commercial claims databases for the years from 2013 to 2016. The LLS received support for the study from Pfizer, Genentech, and Amgen.
FDA clears portable hematology analyzer
The U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for PixCell Medical’s HemoScreen™.
This portable hematology analyzer is used to perform a complete blood count at the point of care.
HemoScreen requires a single drop of blood and uses disposable cartridges that provide automatic sample preparation.
HemoScreen can analyze 20 standard complete blood count parameters and produces results within 5 minutes.
Study results suggested that HemoScreen provides results comparable to those of another hematology analyzer, Sysmex XE-2100. This study was published in the Journal of Clinical Pathology in 2016.
“The HemoScreen delivers lab-accurate results,” said Avishay Bransky, PhD, chief executive officer of PixCell Medical.
He added that HemoScreen “would be especially useful” in physicians’ offices, emergency rooms, intensive care units, oncology clinics, and remote locations.
HemoScreen makes use of a technology called viscoelastic focusing, which employs microfluidics and machine vision algorithms to analyze cells.
The U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for PixCell Medical’s HemoScreen™.
This portable hematology analyzer is used to perform a complete blood count at the point of care.
HemoScreen requires a single drop of blood and uses disposable cartridges that provide automatic sample preparation.
HemoScreen can analyze 20 standard complete blood count parameters and produces results within 5 minutes.
Study results suggested that HemoScreen provides results comparable to those of another hematology analyzer, Sysmex XE-2100. This study was published in the Journal of Clinical Pathology in 2016.
“The HemoScreen delivers lab-accurate results,” said Avishay Bransky, PhD, chief executive officer of PixCell Medical.
He added that HemoScreen “would be especially useful” in physicians’ offices, emergency rooms, intensive care units, oncology clinics, and remote locations.
HemoScreen makes use of a technology called viscoelastic focusing, which employs microfluidics and machine vision algorithms to analyze cells.
The U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for PixCell Medical’s HemoScreen™.
This portable hematology analyzer is used to perform a complete blood count at the point of care.
HemoScreen requires a single drop of blood and uses disposable cartridges that provide automatic sample preparation.
HemoScreen can analyze 20 standard complete blood count parameters and produces results within 5 minutes.
Study results suggested that HemoScreen provides results comparable to those of another hematology analyzer, Sysmex XE-2100. This study was published in the Journal of Clinical Pathology in 2016.
“The HemoScreen delivers lab-accurate results,” said Avishay Bransky, PhD, chief executive officer of PixCell Medical.
He added that HemoScreen “would be especially useful” in physicians’ offices, emergency rooms, intensive care units, oncology clinics, and remote locations.
HemoScreen makes use of a technology called viscoelastic focusing, which employs microfluidics and machine vision algorithms to analyze cells.
Variant not linked to CLL in Southeast Europe
DUBROVNIK, CROATIA – New research suggests there is no association between the PTPN22 R620W polymorphism and chronic lymphocytic leukemia (CLL) or autoimmune hematologic disorders in patients from the Republic of Macedonia.
Past studies have shown an association between the PTPN22 R620W variant and both CLL and autoimmune diseases in patients from Northwest Europe. However, a new study of Macedonian patients suggests there is no association between the variant and CLL, autoimmune hemolytic anemia (AIHA), or idiopathic thrombocytopenic purpura (ITP) for patients from Southeast Europe.
Irina Panovska-Stavridis, PhD, of Ss. Cyril and Methodius University in Skopje, Republic of Macedonia, and her colleagues presented this finding at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
“A lot of data from the literature suggests [the PTPN22 R620W variant ] has a role in developing multiple immune diseases, but it is validated just in patients from Northwest Europe,” Dr. Panovska-Stavridis noted.
She and her colleagues decided to assess the frequency of the PTPN22 R620W variant (C1858T, rs2476601) in individuals from Southeast Europe, particularly the Republic of Macedonia.
The researchers evaluated 320 patients – 168 with CLL, 66 with AIHA, and 86 with ITP – and 182 age- and sex-matched control subjects with no history of malignant or autoimmune disease.
The team found a similar frequency of the minor T allele and genotype distribution in control subjects and patients. For example, minor T allele was 0.107 in CLL, 0.067 in AIHA, 0.036 in ITP, and 0.05 in controls. Similarly, the frequency of the CC genotype was 0.809 in CLL, 0.166 in AIHA, 0.023 in ITP, and 0.901 in controls.
Dr. Panovska-Stavridis said these results suggest the PTPN22 R620W variant is not a risk factor for the development of CLL, AIHA, or ITP in patients from Southeast Europe.
She also said the results suggest the influence of the variant on lymphocytic homeostasis is affected by certain genetic and environmental factors, and the development of CLL and autoimmune diseases is influenced by race/ethnicity-based variations in the germline composition of the IGHV locus in correlation with environmental factors.
Dr. Panovska-Stavridis did not declare any conflicts of interest.
The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Associates, which is owned by the parent company of this news organization.
DUBROVNIK, CROATIA – New research suggests there is no association between the PTPN22 R620W polymorphism and chronic lymphocytic leukemia (CLL) or autoimmune hematologic disorders in patients from the Republic of Macedonia.
Past studies have shown an association between the PTPN22 R620W variant and both CLL and autoimmune diseases in patients from Northwest Europe. However, a new study of Macedonian patients suggests there is no association between the variant and CLL, autoimmune hemolytic anemia (AIHA), or idiopathic thrombocytopenic purpura (ITP) for patients from Southeast Europe.
Irina Panovska-Stavridis, PhD, of Ss. Cyril and Methodius University in Skopje, Republic of Macedonia, and her colleagues presented this finding at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
“A lot of data from the literature suggests [the PTPN22 R620W variant ] has a role in developing multiple immune diseases, but it is validated just in patients from Northwest Europe,” Dr. Panovska-Stavridis noted.
She and her colleagues decided to assess the frequency of the PTPN22 R620W variant (C1858T, rs2476601) in individuals from Southeast Europe, particularly the Republic of Macedonia.
The researchers evaluated 320 patients – 168 with CLL, 66 with AIHA, and 86 with ITP – and 182 age- and sex-matched control subjects with no history of malignant or autoimmune disease.
The team found a similar frequency of the minor T allele and genotype distribution in control subjects and patients. For example, minor T allele was 0.107 in CLL, 0.067 in AIHA, 0.036 in ITP, and 0.05 in controls. Similarly, the frequency of the CC genotype was 0.809 in CLL, 0.166 in AIHA, 0.023 in ITP, and 0.901 in controls.
Dr. Panovska-Stavridis said these results suggest the PTPN22 R620W variant is not a risk factor for the development of CLL, AIHA, or ITP in patients from Southeast Europe.
She also said the results suggest the influence of the variant on lymphocytic homeostasis is affected by certain genetic and environmental factors, and the development of CLL and autoimmune diseases is influenced by race/ethnicity-based variations in the germline composition of the IGHV locus in correlation with environmental factors.
Dr. Panovska-Stavridis did not declare any conflicts of interest.
The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Associates, which is owned by the parent company of this news organization.
DUBROVNIK, CROATIA – New research suggests there is no association between the PTPN22 R620W polymorphism and chronic lymphocytic leukemia (CLL) or autoimmune hematologic disorders in patients from the Republic of Macedonia.
Past studies have shown an association between the PTPN22 R620W variant and both CLL and autoimmune diseases in patients from Northwest Europe. However, a new study of Macedonian patients suggests there is no association between the variant and CLL, autoimmune hemolytic anemia (AIHA), or idiopathic thrombocytopenic purpura (ITP) for patients from Southeast Europe.
Irina Panovska-Stavridis, PhD, of Ss. Cyril and Methodius University in Skopje, Republic of Macedonia, and her colleagues presented this finding at Leukemia and Lymphoma, a meeting jointly sponsored by the University of Texas MD Anderson Cancer Center and the School of Medicine at the University of Zagreb, Croatia.
“A lot of data from the literature suggests [the PTPN22 R620W variant ] has a role in developing multiple immune diseases, but it is validated just in patients from Northwest Europe,” Dr. Panovska-Stavridis noted.
She and her colleagues decided to assess the frequency of the PTPN22 R620W variant (C1858T, rs2476601) in individuals from Southeast Europe, particularly the Republic of Macedonia.
The researchers evaluated 320 patients – 168 with CLL, 66 with AIHA, and 86 with ITP – and 182 age- and sex-matched control subjects with no history of malignant or autoimmune disease.
The team found a similar frequency of the minor T allele and genotype distribution in control subjects and patients. For example, minor T allele was 0.107 in CLL, 0.067 in AIHA, 0.036 in ITP, and 0.05 in controls. Similarly, the frequency of the CC genotype was 0.809 in CLL, 0.166 in AIHA, 0.023 in ITP, and 0.901 in controls.
Dr. Panovska-Stavridis said these results suggest the PTPN22 R620W variant is not a risk factor for the development of CLL, AIHA, or ITP in patients from Southeast Europe.
She also said the results suggest the influence of the variant on lymphocytic homeostasis is affected by certain genetic and environmental factors, and the development of CLL and autoimmune diseases is influenced by race/ethnicity-based variations in the germline composition of the IGHV locus in correlation with environmental factors.
Dr. Panovska-Stavridis did not declare any conflicts of interest.
The Leukemia and Lymphoma meeting is organized by Jonathan Wood & Associates, which is owned by the parent company of this news organization.
REPORTING FROM LEUKEMIA AND LYMPHOMA 2018
Key clinical point:
Major finding: The frequency of minor T allele was 0.107 in patients with CLL, 0.067 in patients with autoimmune hemolytic anemia, 0.036 in patients with idiopathic thrombocytopenic purpura, and 0.05 in controls.
Study details: An analysis of the frequency of the PTPN22 R620W variant in 320 individuals from the Republic of Macedonia.
Disclosures: Dr. Panovska-Stavridis did not declare any conflicts of interest.