User login
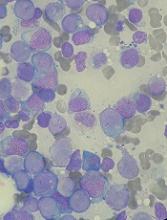
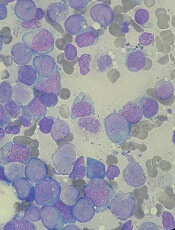
Quizartinib receives accelerated assessment for AML
The European Medicines Agency has granted accelerated assessment to the marketing authorization application (MAA) for the FLT3 inhibitor quizartinib.
With this MAA, Daiichi Sankyo Company, Ltd., is seeking authorization for quizartinib to treat adults with FLT3-ITD-positive, relapsed or refractory acute myeloid leukemia (AML).
Accelerated assessment is given to products expected to be of major interest for public health and therapeutic innovation, and it can reduce the review timeline from 210 days to 150 days.
Quizartinib also has orphan drug designation from the European Commission.
The MAA for quizartinib is based on the phase 3 QuANTUM-R study. Results from this trial were presented at the 23rd Congress of the European Hematology Association in June.
QuANTUM-R enrolled adults with FLT3-ITD AML (at least 3% FLT3-ITD allelic ratio) who had refractory disease or had relapsed within 6 months of their first complete response (CR).
Patients were randomized to receive once-daily treatment with quizartinib (n=245) or a salvage chemotherapy regimen (n=122)—low-dose cytarabine (LoDAC, n=29); combination mitoxantrone, etoposide, and cytarabine (MEC, n=40); or combination fludarabine, cytarabine, and idarubicin (FLAG-IDA, n=53).
Patients who responded to treatment could proceed to hematopoietic stem cell transplant (HSCT), and those in the quizartinib arm could resume quizartinib after HSCT.
In all, 241 patients received quizartinib, and 94 received salvage chemotherapy—LoDAC (n=22), MEC (n=25), and FLAG-IDA (n=47). Of the 28 patients in the chemotherapy group who were not treated, most withdrew consent.
Thirty-two percent of quizartinib-treated patients and 12% of the chemotherapy group went on to HSCT.
Efficacy
The median follow-up was 23.5 months. The efficacy results include all randomized patients.
The overall response rate was 69% in the quizartinib arm and 30% in the chemotherapy arm.
The composite CR rate was 48% in the quizartinib arm and 27% in the chemotherapy arm. This includes:
- The CR rate (4% and 1%, respectively)
- The rate of CR with incomplete platelet recovery (4% and 0%, respectively)
- The rate of CR with incomplete hematologic recovery (40% and 26%, respectively).
The median event-free survival was 6.0 weeks in the quizartinib arm and 3.7 weeks in the chemotherapy arm (hazard ratio=0.90, P=0.1071).
The median overall survival was 6.2 months in the quizartinib arm and 4.7 months in the chemotherapy arm (hazard ratio=0.76, P=0.0177). The 1-year overall survival rate was 27% and 20%, respectively.
Safety
The safety results include only patients who received their assigned treatment.
Grade 3 or higher hematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included:
- Thrombocytopenia (35% and 34%)
- Anemia (30% and 29%)
- Neutropenia (32% and 25%)
- Febrile neutropenia (31% and 21%)
- Leukopenia (17% and 16%).
Grade 3 or higher non-hematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included:
- Sepsis/septic shock (16% and 18%)
- Hypokalemia (12% and 9%)
- Pneumonia (12% and 9%)
- Fatigue (8% and 1%)
- Dyspnea (5% for both)
- Hypophosphatemia (5% for both).
The European Medicines Agency has granted accelerated assessment to the marketing authorization application (MAA) for the FLT3 inhibitor quizartinib.
With this MAA, Daiichi Sankyo Company, Ltd., is seeking authorization for quizartinib to treat adults with FLT3-ITD-positive, relapsed or refractory acute myeloid leukemia (AML).
Accelerated assessment is given to products expected to be of major interest for public health and therapeutic innovation, and it can reduce the review timeline from 210 days to 150 days.
Quizartinib also has orphan drug designation from the European Commission.
The MAA for quizartinib is based on the phase 3 QuANTUM-R study. Results from this trial were presented at the 23rd Congress of the European Hematology Association in June.
QuANTUM-R enrolled adults with FLT3-ITD AML (at least 3% FLT3-ITD allelic ratio) who had refractory disease or had relapsed within 6 months of their first complete response (CR).
Patients were randomized to receive once-daily treatment with quizartinib (n=245) or a salvage chemotherapy regimen (n=122)—low-dose cytarabine (LoDAC, n=29); combination mitoxantrone, etoposide, and cytarabine (MEC, n=40); or combination fludarabine, cytarabine, and idarubicin (FLAG-IDA, n=53).
Patients who responded to treatment could proceed to hematopoietic stem cell transplant (HSCT), and those in the quizartinib arm could resume quizartinib after HSCT.
In all, 241 patients received quizartinib, and 94 received salvage chemotherapy—LoDAC (n=22), MEC (n=25), and FLAG-IDA (n=47). Of the 28 patients in the chemotherapy group who were not treated, most withdrew consent.
Thirty-two percent of quizartinib-treated patients and 12% of the chemotherapy group went on to HSCT.
Efficacy
The median follow-up was 23.5 months. The efficacy results include all randomized patients.
The overall response rate was 69% in the quizartinib arm and 30% in the chemotherapy arm.
The composite CR rate was 48% in the quizartinib arm and 27% in the chemotherapy arm. This includes:
- The CR rate (4% and 1%, respectively)
- The rate of CR with incomplete platelet recovery (4% and 0%, respectively)
- The rate of CR with incomplete hematologic recovery (40% and 26%, respectively).
The median event-free survival was 6.0 weeks in the quizartinib arm and 3.7 weeks in the chemotherapy arm (hazard ratio=0.90, P=0.1071).
The median overall survival was 6.2 months in the quizartinib arm and 4.7 months in the chemotherapy arm (hazard ratio=0.76, P=0.0177). The 1-year overall survival rate was 27% and 20%, respectively.
Safety
The safety results include only patients who received their assigned treatment.
Grade 3 or higher hematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included:
- Thrombocytopenia (35% and 34%)
- Anemia (30% and 29%)
- Neutropenia (32% and 25%)
- Febrile neutropenia (31% and 21%)
- Leukopenia (17% and 16%).
Grade 3 or higher non-hematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included:
- Sepsis/septic shock (16% and 18%)
- Hypokalemia (12% and 9%)
- Pneumonia (12% and 9%)
- Fatigue (8% and 1%)
- Dyspnea (5% for both)
- Hypophosphatemia (5% for both).
The European Medicines Agency has granted accelerated assessment to the marketing authorization application (MAA) for the FLT3 inhibitor quizartinib.
With this MAA, Daiichi Sankyo Company, Ltd., is seeking authorization for quizartinib to treat adults with FLT3-ITD-positive, relapsed or refractory acute myeloid leukemia (AML).
Accelerated assessment is given to products expected to be of major interest for public health and therapeutic innovation, and it can reduce the review timeline from 210 days to 150 days.
Quizartinib also has orphan drug designation from the European Commission.
The MAA for quizartinib is based on the phase 3 QuANTUM-R study. Results from this trial were presented at the 23rd Congress of the European Hematology Association in June.
QuANTUM-R enrolled adults with FLT3-ITD AML (at least 3% FLT3-ITD allelic ratio) who had refractory disease or had relapsed within 6 months of their first complete response (CR).
Patients were randomized to receive once-daily treatment with quizartinib (n=245) or a salvage chemotherapy regimen (n=122)—low-dose cytarabine (LoDAC, n=29); combination mitoxantrone, etoposide, and cytarabine (MEC, n=40); or combination fludarabine, cytarabine, and idarubicin (FLAG-IDA, n=53).
Patients who responded to treatment could proceed to hematopoietic stem cell transplant (HSCT), and those in the quizartinib arm could resume quizartinib after HSCT.
In all, 241 patients received quizartinib, and 94 received salvage chemotherapy—LoDAC (n=22), MEC (n=25), and FLAG-IDA (n=47). Of the 28 patients in the chemotherapy group who were not treated, most withdrew consent.
Thirty-two percent of quizartinib-treated patients and 12% of the chemotherapy group went on to HSCT.
Efficacy
The median follow-up was 23.5 months. The efficacy results include all randomized patients.
The overall response rate was 69% in the quizartinib arm and 30% in the chemotherapy arm.
The composite CR rate was 48% in the quizartinib arm and 27% in the chemotherapy arm. This includes:
- The CR rate (4% and 1%, respectively)
- The rate of CR with incomplete platelet recovery (4% and 0%, respectively)
- The rate of CR with incomplete hematologic recovery (40% and 26%, respectively).
The median event-free survival was 6.0 weeks in the quizartinib arm and 3.7 weeks in the chemotherapy arm (hazard ratio=0.90, P=0.1071).
The median overall survival was 6.2 months in the quizartinib arm and 4.7 months in the chemotherapy arm (hazard ratio=0.76, P=0.0177). The 1-year overall survival rate was 27% and 20%, respectively.
Safety
The safety results include only patients who received their assigned treatment.
Grade 3 or higher hematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included:
- Thrombocytopenia (35% and 34%)
- Anemia (30% and 29%)
- Neutropenia (32% and 25%)
- Febrile neutropenia (31% and 21%)
- Leukopenia (17% and 16%).
Grade 3 or higher non-hematologic treatment-emergent adverse events occurring in at least 5% of patients (in the quizartinib and chemotherapy groups, respectively) included:
- Sepsis/septic shock (16% and 18%)
- Hypokalemia (12% and 9%)
- Pneumonia (12% and 9%)
- Fatigue (8% and 1%)
- Dyspnea (5% for both)
- Hypophosphatemia (5% for both).
Changes related to AML relapse may be reversible
New research suggests relapse of acute myeloid leukemia (AML) after allogeneic hematopoietic stem cell transplant (HSCT) is related to changes in immune-related gene expression that may be reversible.
Researchers observed downregulation of major histocompatibility complex (MHC) class II genes in samples from patients who relapsed after HSCT.
However, interferon-gamma “rapidly reversed this phenotype” in vitro, according to the researchers.
Matthew J. Christopher, MD, PhD, of Washington University School of Medicine in St. Louis, Missouri, and his colleagues reported these findings in The New England Journal of Medicine.
The researchers set out to determine how genetic and epigenetic changes after HSCT may allow leukemic cells to avoid the graft-vs-leukemia effect and to see whether immune-related genes are affected by HSCT.
The team analyzed paired samples obtained at diagnosis and relapse from 15 AML patients who relapsed after HSCT and 20 AML patients who relapsed after chemotherapy. The team also analyzed additional samples from patients who relapsed after HSCT to validate initial findings.
Methods of analysis included enhanced exome sequencing, RNA sequencing, flow cytometry, and immunohistochemical analysis.
Findings
The researchers first looked for relapse-specific mutations but found no driver mutations associated with relapse after HSCT.
The mutations seen post-HSCT relapse were generally similar to those seen both before treatment and after relapse in patients who had undergone chemotherapy, and the researchers could not identify any patterns of mutations related to relapse.
They then looked for, but did not find, relapse-specific mutations in genes involved in modulation of immune checkpoints, antigen presentation, or cytokine signaling.
The researchers did, however, find evidence of epigenetic changes that were more common in the samples from patients with post-transplant relapses.
RNA sequencing showed that MHC class II genes (HLA-DPA1, HLA-DPB1, HLA-DQB1, and HLA-DRB1) were downregulated three- to 12-fold after transplant.
Flow cytometry and immunohistochemical analysis confirmed that MHC class II expression was decreased at relapse after HSCT in 17 of 34 samples evaluated.
The researchers said there was no association between the downregulation of MHC class II and donor type or use of immunosuppression.
To see whether the downregulation of MHC class II genes was reversible, the researchers treated three post-HSCT relapse samples with interferon-gamma, which is known to upregulate MHC class II on certain cells.
Culturing patient cells with interferon-gamma “rapidly induced MHC class II protein expression on leukemic blasts,” the researchers said. They observed “essentially full restoration of MHC class II protein expression in nearly all AML blasts after 72 hours.”
This study was supported by the National Institutes of Health, Leukemia and Lymphoma Society, and the Barnes-Jewish Hospital Foundation.
Several study authors reported personal fees and/or research support from industry outside the submitted work.
New research suggests relapse of acute myeloid leukemia (AML) after allogeneic hematopoietic stem cell transplant (HSCT) is related to changes in immune-related gene expression that may be reversible.
Researchers observed downregulation of major histocompatibility complex (MHC) class II genes in samples from patients who relapsed after HSCT.
However, interferon-gamma “rapidly reversed this phenotype” in vitro, according to the researchers.
Matthew J. Christopher, MD, PhD, of Washington University School of Medicine in St. Louis, Missouri, and his colleagues reported these findings in The New England Journal of Medicine.
The researchers set out to determine how genetic and epigenetic changes after HSCT may allow leukemic cells to avoid the graft-vs-leukemia effect and to see whether immune-related genes are affected by HSCT.
The team analyzed paired samples obtained at diagnosis and relapse from 15 AML patients who relapsed after HSCT and 20 AML patients who relapsed after chemotherapy. The team also analyzed additional samples from patients who relapsed after HSCT to validate initial findings.
Methods of analysis included enhanced exome sequencing, RNA sequencing, flow cytometry, and immunohistochemical analysis.
Findings
The researchers first looked for relapse-specific mutations but found no driver mutations associated with relapse after HSCT.
The mutations seen post-HSCT relapse were generally similar to those seen both before treatment and after relapse in patients who had undergone chemotherapy, and the researchers could not identify any patterns of mutations related to relapse.
They then looked for, but did not find, relapse-specific mutations in genes involved in modulation of immune checkpoints, antigen presentation, or cytokine signaling.
The researchers did, however, find evidence of epigenetic changes that were more common in the samples from patients with post-transplant relapses.
RNA sequencing showed that MHC class II genes (HLA-DPA1, HLA-DPB1, HLA-DQB1, and HLA-DRB1) were downregulated three- to 12-fold after transplant.
Flow cytometry and immunohistochemical analysis confirmed that MHC class II expression was decreased at relapse after HSCT in 17 of 34 samples evaluated.
The researchers said there was no association between the downregulation of MHC class II and donor type or use of immunosuppression.
To see whether the downregulation of MHC class II genes was reversible, the researchers treated three post-HSCT relapse samples with interferon-gamma, which is known to upregulate MHC class II on certain cells.
Culturing patient cells with interferon-gamma “rapidly induced MHC class II protein expression on leukemic blasts,” the researchers said. They observed “essentially full restoration of MHC class II protein expression in nearly all AML blasts after 72 hours.”
This study was supported by the National Institutes of Health, Leukemia and Lymphoma Society, and the Barnes-Jewish Hospital Foundation.
Several study authors reported personal fees and/or research support from industry outside the submitted work.
New research suggests relapse of acute myeloid leukemia (AML) after allogeneic hematopoietic stem cell transplant (HSCT) is related to changes in immune-related gene expression that may be reversible.
Researchers observed downregulation of major histocompatibility complex (MHC) class II genes in samples from patients who relapsed after HSCT.
However, interferon-gamma “rapidly reversed this phenotype” in vitro, according to the researchers.
Matthew J. Christopher, MD, PhD, of Washington University School of Medicine in St. Louis, Missouri, and his colleagues reported these findings in The New England Journal of Medicine.
The researchers set out to determine how genetic and epigenetic changes after HSCT may allow leukemic cells to avoid the graft-vs-leukemia effect and to see whether immune-related genes are affected by HSCT.
The team analyzed paired samples obtained at diagnosis and relapse from 15 AML patients who relapsed after HSCT and 20 AML patients who relapsed after chemotherapy. The team also analyzed additional samples from patients who relapsed after HSCT to validate initial findings.
Methods of analysis included enhanced exome sequencing, RNA sequencing, flow cytometry, and immunohistochemical analysis.
Findings
The researchers first looked for relapse-specific mutations but found no driver mutations associated with relapse after HSCT.
The mutations seen post-HSCT relapse were generally similar to those seen both before treatment and after relapse in patients who had undergone chemotherapy, and the researchers could not identify any patterns of mutations related to relapse.
They then looked for, but did not find, relapse-specific mutations in genes involved in modulation of immune checkpoints, antigen presentation, or cytokine signaling.
The researchers did, however, find evidence of epigenetic changes that were more common in the samples from patients with post-transplant relapses.
RNA sequencing showed that MHC class II genes (HLA-DPA1, HLA-DPB1, HLA-DQB1, and HLA-DRB1) were downregulated three- to 12-fold after transplant.
Flow cytometry and immunohistochemical analysis confirmed that MHC class II expression was decreased at relapse after HSCT in 17 of 34 samples evaluated.
The researchers said there was no association between the downregulation of MHC class II and donor type or use of immunosuppression.
To see whether the downregulation of MHC class II genes was reversible, the researchers treated three post-HSCT relapse samples with interferon-gamma, which is known to upregulate MHC class II on certain cells.
Culturing patient cells with interferon-gamma “rapidly induced MHC class II protein expression on leukemic blasts,” the researchers said. They observed “essentially full restoration of MHC class II protein expression in nearly all AML blasts after 72 hours.”
This study was supported by the National Institutes of Health, Leukemia and Lymphoma Society, and the Barnes-Jewish Hospital Foundation.
Several study authors reported personal fees and/or research support from industry outside the submitted work.
Venetoclax/HMA combo still safe, effective for elderly with AML
In patients aged 65 years and older with acute myeloid leukemia (AML), the combination of venetoclax (Venclexta) and a hypomethylating agent had good efficacy and was well tolerated, according to updated results from a phase 1b dose-escalation and expansion trial.
At a median of 8.9 months of study, the overall response rate (ORR) among all treated patients was 68%, with a median duration of complete remission (CR) plus CR with incomplete count recovery (CRi) of 11.3 months, reported Courtney DiNardo, MD, from the University of Texas MD Anderson Center in Houston and her colleagues.
“Venetoclax in combination with azacitidine or decitabine was well tolerated, with similar safety profiles within all arms of the dose escalation and expansion phases in elderly patients with previously untreated AML ineligible for standard induction therapy,” they wrote in a paper published in Blood.
At the 2017 European Hematology Association Congress, the investigators reported that the combined rate of complete remission CR and CRi was 60% among patients with poor-risk cytogenetics and 78% among patients with intermediate-risk disease. In addition, the drug combination was effective among patients with both primary de novo AML (68%) and secondary AML (related to myelodysplasia or myeloproliferative neoplasms or previous therapy; 73%).
In this, the most recent analysis, Dr. DiNardo and her colleagues reported on follow-up of 145 patients aged 65 and older with treatment-naive AML who were not eligible for intensive chemotherapy regimens used for younger adults. The median age was 74 years. Approximately half of all patients (49%) had poor-risk cytogenetics.
The patients were treated with either decitabine or azacitidine plus venetoclax at a dose of either 400 mg or 800 mg. Decitabine was dosed at 20 mg/m2 intravenously on days 1-5 of a 28-day cycle. Azacitidine was dosed at 75 mg/m2 subcutaneously on days 1-7 of every cycle.
The median time on study was 8.9 months. Among all patients treated at all doses, 67% had either a CR or CRi. The combined CR/CRi rate in patients treated at the 400 mg dose of venetoclax was 73%.
The CR/CRi rate for patients with poor-risk cytogenetics was 60%, and the rate for patients aged 75 years and older was 65%.
Among all patients, the median duration of CR/CRi was 11.3 months, and median overall survival was 17.5 months. In the 400 mg venetoclax cohort, the median duration of CR/CRi was 12.5 months, with the median OS not reached at the time of data cutoff.
Adverse events occurring in 30% or more of patients included constipation, diarrhea, vomiting, nausea, fatigue, febrile neutropenia, hypokalemia, decreased appetite, and decreased white blood cell count. There were no reported cases of the tumor lysis syndrome, a known complication of venetoclax therapy.
Venetoclax plus decitabine or azacitidine was effective in high-risk subgroups, including patients aged 75 years and older, those with poor cytogenetic risk, and those with secondary AML, the investigators noted.
“Though these observations are drawn from a relatively small subset of patients, the remission rates achieved by our low-intensity regimen are encouraging in light of the traditionally lower remission rates in the elderly AML population (40%-50%) compared with young patients receiving chemotherapy (60%-70%) and the relatively short duration of these remissions,” Dr. DiNardo and her colleagues wrote.
A phase 3 trial is currently underway comparing venetoclax at the 400 mg dose plus azacitidine with azacitidine alone in treatment-naive patients with AML who are ineligible for standard induction therapy.
The trial was supported by AbbVie and Genentech. Dr. DiNardo and multiple coauthors disclosed relationships with AbbVie, Genentech, and other companies.
SOURCE: DiNardo C et al. Blood. 2018 Oct 25. doi: 10.1182/blood-2018-08-868752.
In patients aged 65 years and older with acute myeloid leukemia (AML), the combination of venetoclax (Venclexta) and a hypomethylating agent had good efficacy and was well tolerated, according to updated results from a phase 1b dose-escalation and expansion trial.
At a median of 8.9 months of study, the overall response rate (ORR) among all treated patients was 68%, with a median duration of complete remission (CR) plus CR with incomplete count recovery (CRi) of 11.3 months, reported Courtney DiNardo, MD, from the University of Texas MD Anderson Center in Houston and her colleagues.
“Venetoclax in combination with azacitidine or decitabine was well tolerated, with similar safety profiles within all arms of the dose escalation and expansion phases in elderly patients with previously untreated AML ineligible for standard induction therapy,” they wrote in a paper published in Blood.
At the 2017 European Hematology Association Congress, the investigators reported that the combined rate of complete remission CR and CRi was 60% among patients with poor-risk cytogenetics and 78% among patients with intermediate-risk disease. In addition, the drug combination was effective among patients with both primary de novo AML (68%) and secondary AML (related to myelodysplasia or myeloproliferative neoplasms or previous therapy; 73%).
In this, the most recent analysis, Dr. DiNardo and her colleagues reported on follow-up of 145 patients aged 65 and older with treatment-naive AML who were not eligible for intensive chemotherapy regimens used for younger adults. The median age was 74 years. Approximately half of all patients (49%) had poor-risk cytogenetics.
The patients were treated with either decitabine or azacitidine plus venetoclax at a dose of either 400 mg or 800 mg. Decitabine was dosed at 20 mg/m2 intravenously on days 1-5 of a 28-day cycle. Azacitidine was dosed at 75 mg/m2 subcutaneously on days 1-7 of every cycle.
The median time on study was 8.9 months. Among all patients treated at all doses, 67% had either a CR or CRi. The combined CR/CRi rate in patients treated at the 400 mg dose of venetoclax was 73%.
The CR/CRi rate for patients with poor-risk cytogenetics was 60%, and the rate for patients aged 75 years and older was 65%.
Among all patients, the median duration of CR/CRi was 11.3 months, and median overall survival was 17.5 months. In the 400 mg venetoclax cohort, the median duration of CR/CRi was 12.5 months, with the median OS not reached at the time of data cutoff.
Adverse events occurring in 30% or more of patients included constipation, diarrhea, vomiting, nausea, fatigue, febrile neutropenia, hypokalemia, decreased appetite, and decreased white blood cell count. There were no reported cases of the tumor lysis syndrome, a known complication of venetoclax therapy.
Venetoclax plus decitabine or azacitidine was effective in high-risk subgroups, including patients aged 75 years and older, those with poor cytogenetic risk, and those with secondary AML, the investigators noted.
“Though these observations are drawn from a relatively small subset of patients, the remission rates achieved by our low-intensity regimen are encouraging in light of the traditionally lower remission rates in the elderly AML population (40%-50%) compared with young patients receiving chemotherapy (60%-70%) and the relatively short duration of these remissions,” Dr. DiNardo and her colleagues wrote.
A phase 3 trial is currently underway comparing venetoclax at the 400 mg dose plus azacitidine with azacitidine alone in treatment-naive patients with AML who are ineligible for standard induction therapy.
The trial was supported by AbbVie and Genentech. Dr. DiNardo and multiple coauthors disclosed relationships with AbbVie, Genentech, and other companies.
SOURCE: DiNardo C et al. Blood. 2018 Oct 25. doi: 10.1182/blood-2018-08-868752.
In patients aged 65 years and older with acute myeloid leukemia (AML), the combination of venetoclax (Venclexta) and a hypomethylating agent had good efficacy and was well tolerated, according to updated results from a phase 1b dose-escalation and expansion trial.
At a median of 8.9 months of study, the overall response rate (ORR) among all treated patients was 68%, with a median duration of complete remission (CR) plus CR with incomplete count recovery (CRi) of 11.3 months, reported Courtney DiNardo, MD, from the University of Texas MD Anderson Center in Houston and her colleagues.
“Venetoclax in combination with azacitidine or decitabine was well tolerated, with similar safety profiles within all arms of the dose escalation and expansion phases in elderly patients with previously untreated AML ineligible for standard induction therapy,” they wrote in a paper published in Blood.
At the 2017 European Hematology Association Congress, the investigators reported that the combined rate of complete remission CR and CRi was 60% among patients with poor-risk cytogenetics and 78% among patients with intermediate-risk disease. In addition, the drug combination was effective among patients with both primary de novo AML (68%) and secondary AML (related to myelodysplasia or myeloproliferative neoplasms or previous therapy; 73%).
In this, the most recent analysis, Dr. DiNardo and her colleagues reported on follow-up of 145 patients aged 65 and older with treatment-naive AML who were not eligible for intensive chemotherapy regimens used for younger adults. The median age was 74 years. Approximately half of all patients (49%) had poor-risk cytogenetics.
The patients were treated with either decitabine or azacitidine plus venetoclax at a dose of either 400 mg or 800 mg. Decitabine was dosed at 20 mg/m2 intravenously on days 1-5 of a 28-day cycle. Azacitidine was dosed at 75 mg/m2 subcutaneously on days 1-7 of every cycle.
The median time on study was 8.9 months. Among all patients treated at all doses, 67% had either a CR or CRi. The combined CR/CRi rate in patients treated at the 400 mg dose of venetoclax was 73%.
The CR/CRi rate for patients with poor-risk cytogenetics was 60%, and the rate for patients aged 75 years and older was 65%.
Among all patients, the median duration of CR/CRi was 11.3 months, and median overall survival was 17.5 months. In the 400 mg venetoclax cohort, the median duration of CR/CRi was 12.5 months, with the median OS not reached at the time of data cutoff.
Adverse events occurring in 30% or more of patients included constipation, diarrhea, vomiting, nausea, fatigue, febrile neutropenia, hypokalemia, decreased appetite, and decreased white blood cell count. There were no reported cases of the tumor lysis syndrome, a known complication of venetoclax therapy.
Venetoclax plus decitabine or azacitidine was effective in high-risk subgroups, including patients aged 75 years and older, those with poor cytogenetic risk, and those with secondary AML, the investigators noted.
“Though these observations are drawn from a relatively small subset of patients, the remission rates achieved by our low-intensity regimen are encouraging in light of the traditionally lower remission rates in the elderly AML population (40%-50%) compared with young patients receiving chemotherapy (60%-70%) and the relatively short duration of these remissions,” Dr. DiNardo and her colleagues wrote.
A phase 3 trial is currently underway comparing venetoclax at the 400 mg dose plus azacitidine with azacitidine alone in treatment-naive patients with AML who are ineligible for standard induction therapy.
The trial was supported by AbbVie and Genentech. Dr. DiNardo and multiple coauthors disclosed relationships with AbbVie, Genentech, and other companies.
SOURCE: DiNardo C et al. Blood. 2018 Oct 25. doi: 10.1182/blood-2018-08-868752.
FROM BLOOD
Key clinical point:
Major finding: At a median time of study of 8.9 months, the overall response rate among all treated patients was 68%.
Study details: Follow-up of a phase 1b dose-escalation and expansion cohort of 145 patients aged 65 years and older with treatment-naive AML.
Disclosures: The trial was supported by AbbVie and Genentech. Dr. DiNardo and multiple coauthors disclosed relationships with AbbVie, Genentech, and other companies.
Source: DiNardo C et al. Blood. 2018 Oct 25. doi: 10.1182/blood-2018-08-868752.
FDA and EC approve pegfilgrastim biosimilar
The U.S. Food and Drug Administration (FDA) and European Commission (EC) have approved Coherus BioSciences, Inc.’s pegfilgrastim-cbqv (Udenyca™), a biosimilar of Amgen’s pegfilgrastim product (Neulasta).
Both agencies approved pegfilgrastim-cbqv (formerly CHS-1701) for cancer patients receiving myelosuppressive chemotherapy.
Pegfilgrastim-cbqv is FDA-approved “to decrease the incidence of infection, as manifested by febrile neutropenia, in patients with non-myeloid malignancies receiving myelosuppressive anticancer drugs associated with a clinically significant incidence of febrile neutropenia.”
The product is EC-approved to reduce “the duration of neutropenia and the incidence of febrile neutropenia in adult patients treated with cytotoxic chemotherapy for malignancy (with the exception of chronic myeloid leukemia and myelodysplastic syndromes).”
The U.S. prescribing information for pegfilgrastim-cbqv is available at www.UDENYCA.com, and the European summary of product characteristics is available on the European Medicines Agency’s website.
The FDA and EC approvals of pegfilgrastim-cbqv were supported by analyses establishing biosimilarity as well as pharmacokinetic, pharmacodynamic, and immunogenicity studies of healthy subjects (NCT02650973, NCT02385851, and NCT02418104).
Results from one of these studies (NCT02650973) were presented at the 2017 ASCO Annual Meeting.
“Udenyca’s robust clinical package includes a dedicated immunogenicity similarity study in over 300 healthy subjects,” said Barbara Finck, MD, chief medical officer of Coherus BioSciences.
“In support of that study, and as part of our commitment to ensuring patient safety, we deployed a battery of sensitive immunogenicity assays. This effort not only supported the biosimilarity of Udenyca but also advanced the understanding of the immunogenic response of pegfilgrastim products.”
The U.S. Food and Drug Administration (FDA) and European Commission (EC) have approved Coherus BioSciences, Inc.’s pegfilgrastim-cbqv (Udenyca™), a biosimilar of Amgen’s pegfilgrastim product (Neulasta).
Both agencies approved pegfilgrastim-cbqv (formerly CHS-1701) for cancer patients receiving myelosuppressive chemotherapy.
Pegfilgrastim-cbqv is FDA-approved “to decrease the incidence of infection, as manifested by febrile neutropenia, in patients with non-myeloid malignancies receiving myelosuppressive anticancer drugs associated with a clinically significant incidence of febrile neutropenia.”
The product is EC-approved to reduce “the duration of neutropenia and the incidence of febrile neutropenia in adult patients treated with cytotoxic chemotherapy for malignancy (with the exception of chronic myeloid leukemia and myelodysplastic syndromes).”
The U.S. prescribing information for pegfilgrastim-cbqv is available at www.UDENYCA.com, and the European summary of product characteristics is available on the European Medicines Agency’s website.
The FDA and EC approvals of pegfilgrastim-cbqv were supported by analyses establishing biosimilarity as well as pharmacokinetic, pharmacodynamic, and immunogenicity studies of healthy subjects (NCT02650973, NCT02385851, and NCT02418104).
Results from one of these studies (NCT02650973) were presented at the 2017 ASCO Annual Meeting.
“Udenyca’s robust clinical package includes a dedicated immunogenicity similarity study in over 300 healthy subjects,” said Barbara Finck, MD, chief medical officer of Coherus BioSciences.
“In support of that study, and as part of our commitment to ensuring patient safety, we deployed a battery of sensitive immunogenicity assays. This effort not only supported the biosimilarity of Udenyca but also advanced the understanding of the immunogenic response of pegfilgrastim products.”
The U.S. Food and Drug Administration (FDA) and European Commission (EC) have approved Coherus BioSciences, Inc.’s pegfilgrastim-cbqv (Udenyca™), a biosimilar of Amgen’s pegfilgrastim product (Neulasta).
Both agencies approved pegfilgrastim-cbqv (formerly CHS-1701) for cancer patients receiving myelosuppressive chemotherapy.
Pegfilgrastim-cbqv is FDA-approved “to decrease the incidence of infection, as manifested by febrile neutropenia, in patients with non-myeloid malignancies receiving myelosuppressive anticancer drugs associated with a clinically significant incidence of febrile neutropenia.”
The product is EC-approved to reduce “the duration of neutropenia and the incidence of febrile neutropenia in adult patients treated with cytotoxic chemotherapy for malignancy (with the exception of chronic myeloid leukemia and myelodysplastic syndromes).”
The U.S. prescribing information for pegfilgrastim-cbqv is available at www.UDENYCA.com, and the European summary of product characteristics is available on the European Medicines Agency’s website.
The FDA and EC approvals of pegfilgrastim-cbqv were supported by analyses establishing biosimilarity as well as pharmacokinetic, pharmacodynamic, and immunogenicity studies of healthy subjects (NCT02650973, NCT02385851, and NCT02418104).
Results from one of these studies (NCT02650973) were presented at the 2017 ASCO Annual Meeting.
“Udenyca’s robust clinical package includes a dedicated immunogenicity similarity study in over 300 healthy subjects,” said Barbara Finck, MD, chief medical officer of Coherus BioSciences.
“In support of that study, and as part of our commitment to ensuring patient safety, we deployed a battery of sensitive immunogenicity assays. This effort not only supported the biosimilarity of Udenyca but also advanced the understanding of the immunogenic response of pegfilgrastim products.”
AML relapse after HSCT linked to potentially reversible immune changes
Relapse of acute myeloid leukemia after hematopoietic stem cell transplantation appears to be related to posttransplant changes in immune function that may be reversible with interferon-gamma therapy, investigators said.
Researchers performed a comparison of acute myeloid leukemia (AML) samples taken from patients before hematopoietic stem cell transplantation (HSCT) and at the time of relapse. They found that, while the general genomic changes seen at relapse resembled changes seen when patients experience relapse after chemotherapy, HSCT was associated with changes in genes believed to control both adaptive and innate immunity.
The findings suggest that transplantation results in a dampening of immune surveillance that could potentially be reversed with interferon gamma, an immunostimulatory cytokine, reported Matthew J. Christopher, MD, PhD, from Washington University, St. Louis, and his colleagues.
“These changes appeared to be epigenetic in nature in at least some cases, which suggests that therapeutic strategies to resensitize AML cells to the graft-versus-leukemia effect may be feasible,” they wrote in the New England Journal of Medicine.
The researchers noted that, while the presence of certain AML mutations may predict risk for relapse following HSCT, “the mechanisms by which these mutations promote relapse remain unclear.”
To get a better sense of how genetic and epigenetic changes after transplantation may allow leukemic cells to avoid the graft-versus-leukemia effect – and to see whether immune-related genes are affected by HSCT – they performed enhanced exome sequencing, flow cytometry, and immunohistochemical analyses on samples from 15 patients with AML who had a relapse after receiving transplants from HLA-matched siblings, matched unrelated donors, or HLA-mismatched unrelated donors, and on paired samples from 20 patients who experienced relapses after chemotherapy.
To validate their findings, they also evaluated samples from 28 other patients with AML who had a relapse after transplantation.
They first looked for relapse-specific mutations, but found no driver mutations associated with relapse after transplantation. The mutations seen during relapse after transplantation were generally similar to those seen both before treatment and after relapse in patients who had undergone chemotherapy. The researchers could not identify any patterns of mutations related to relapse.
They then looked for, but did not find, relapse-specific mutations in genes involved in either modulation of immune checkpoints, antigen presentation, or cytokine signaling.
The researchers did, however, find evidence of epigenetic changes that were more common in the samples of patients with posttransplant relapses, compared with postchemotherapy relapses. Specifically, they found that major histocompatibility (MHC) class II genes were down-regulated 200%-1100% after transplant, compared with the pretransplant samples.
In samples from 17 of 34 patients who experienced a relapse after transplantation, both flow cytometry and immunohistochemical analyses confirmed that expression of MHC class II molecules were decreased at relapse.
To see whether this down-regulation was reversible, the researchers treated samples from three patients with posttransplant relapse with interferon gamma, which is known to up-regulate MHC class II protein on myeloid cells and other cell types.
“Culture of these cells with interferon-gamma rapidly induced MHC class II protein expression on leukemic blasts, with essentially full restoration of MHC class II protein expression in nearly all AML blasts after 72 hours,” they wrote, adding that the reversibility of down-regulation of MHC class II in these blasts “strongly suggests that this phenomenon is mediated by an epigenetic mechanism.”
The study was supported by grants to investigators from the National Institutes of Health, Leukemia & Lymphoma Society, and the Barnes-Jewish Hospital Foundation. Dr. Christopher and several coauthors reported receiving grants from the study funders but no other relevant conflicts of interest. Several coauthors reported receiving personal fees and/or research support from industry outside the submitted work.
SOURCE: Christopher MJ et al. N Engl J Med. 2018 Oct 31. doi: 10.1056/NEJMoa1808777.
Relapse of acute myeloid leukemia after hematopoietic stem cell transplantation appears to be related to posttransplant changes in immune function that may be reversible with interferon-gamma therapy, investigators said.
Researchers performed a comparison of acute myeloid leukemia (AML) samples taken from patients before hematopoietic stem cell transplantation (HSCT) and at the time of relapse. They found that, while the general genomic changes seen at relapse resembled changes seen when patients experience relapse after chemotherapy, HSCT was associated with changes in genes believed to control both adaptive and innate immunity.
The findings suggest that transplantation results in a dampening of immune surveillance that could potentially be reversed with interferon gamma, an immunostimulatory cytokine, reported Matthew J. Christopher, MD, PhD, from Washington University, St. Louis, and his colleagues.
“These changes appeared to be epigenetic in nature in at least some cases, which suggests that therapeutic strategies to resensitize AML cells to the graft-versus-leukemia effect may be feasible,” they wrote in the New England Journal of Medicine.
The researchers noted that, while the presence of certain AML mutations may predict risk for relapse following HSCT, “the mechanisms by which these mutations promote relapse remain unclear.”
To get a better sense of how genetic and epigenetic changes after transplantation may allow leukemic cells to avoid the graft-versus-leukemia effect – and to see whether immune-related genes are affected by HSCT – they performed enhanced exome sequencing, flow cytometry, and immunohistochemical analyses on samples from 15 patients with AML who had a relapse after receiving transplants from HLA-matched siblings, matched unrelated donors, or HLA-mismatched unrelated donors, and on paired samples from 20 patients who experienced relapses after chemotherapy.
To validate their findings, they also evaluated samples from 28 other patients with AML who had a relapse after transplantation.
They first looked for relapse-specific mutations, but found no driver mutations associated with relapse after transplantation. The mutations seen during relapse after transplantation were generally similar to those seen both before treatment and after relapse in patients who had undergone chemotherapy. The researchers could not identify any patterns of mutations related to relapse.
They then looked for, but did not find, relapse-specific mutations in genes involved in either modulation of immune checkpoints, antigen presentation, or cytokine signaling.
The researchers did, however, find evidence of epigenetic changes that were more common in the samples of patients with posttransplant relapses, compared with postchemotherapy relapses. Specifically, they found that major histocompatibility (MHC) class II genes were down-regulated 200%-1100% after transplant, compared with the pretransplant samples.
In samples from 17 of 34 patients who experienced a relapse after transplantation, both flow cytometry and immunohistochemical analyses confirmed that expression of MHC class II molecules were decreased at relapse.
To see whether this down-regulation was reversible, the researchers treated samples from three patients with posttransplant relapse with interferon gamma, which is known to up-regulate MHC class II protein on myeloid cells and other cell types.
“Culture of these cells with interferon-gamma rapidly induced MHC class II protein expression on leukemic blasts, with essentially full restoration of MHC class II protein expression in nearly all AML blasts after 72 hours,” they wrote, adding that the reversibility of down-regulation of MHC class II in these blasts “strongly suggests that this phenomenon is mediated by an epigenetic mechanism.”
The study was supported by grants to investigators from the National Institutes of Health, Leukemia & Lymphoma Society, and the Barnes-Jewish Hospital Foundation. Dr. Christopher and several coauthors reported receiving grants from the study funders but no other relevant conflicts of interest. Several coauthors reported receiving personal fees and/or research support from industry outside the submitted work.
SOURCE: Christopher MJ et al. N Engl J Med. 2018 Oct 31. doi: 10.1056/NEJMoa1808777.
Relapse of acute myeloid leukemia after hematopoietic stem cell transplantation appears to be related to posttransplant changes in immune function that may be reversible with interferon-gamma therapy, investigators said.
Researchers performed a comparison of acute myeloid leukemia (AML) samples taken from patients before hematopoietic stem cell transplantation (HSCT) and at the time of relapse. They found that, while the general genomic changes seen at relapse resembled changes seen when patients experience relapse after chemotherapy, HSCT was associated with changes in genes believed to control both adaptive and innate immunity.
The findings suggest that transplantation results in a dampening of immune surveillance that could potentially be reversed with interferon gamma, an immunostimulatory cytokine, reported Matthew J. Christopher, MD, PhD, from Washington University, St. Louis, and his colleagues.
“These changes appeared to be epigenetic in nature in at least some cases, which suggests that therapeutic strategies to resensitize AML cells to the graft-versus-leukemia effect may be feasible,” they wrote in the New England Journal of Medicine.
The researchers noted that, while the presence of certain AML mutations may predict risk for relapse following HSCT, “the mechanisms by which these mutations promote relapse remain unclear.”
To get a better sense of how genetic and epigenetic changes after transplantation may allow leukemic cells to avoid the graft-versus-leukemia effect – and to see whether immune-related genes are affected by HSCT – they performed enhanced exome sequencing, flow cytometry, and immunohistochemical analyses on samples from 15 patients with AML who had a relapse after receiving transplants from HLA-matched siblings, matched unrelated donors, or HLA-mismatched unrelated donors, and on paired samples from 20 patients who experienced relapses after chemotherapy.
To validate their findings, they also evaluated samples from 28 other patients with AML who had a relapse after transplantation.
They first looked for relapse-specific mutations, but found no driver mutations associated with relapse after transplantation. The mutations seen during relapse after transplantation were generally similar to those seen both before treatment and after relapse in patients who had undergone chemotherapy. The researchers could not identify any patterns of mutations related to relapse.
They then looked for, but did not find, relapse-specific mutations in genes involved in either modulation of immune checkpoints, antigen presentation, or cytokine signaling.
The researchers did, however, find evidence of epigenetic changes that were more common in the samples of patients with posttransplant relapses, compared with postchemotherapy relapses. Specifically, they found that major histocompatibility (MHC) class II genes were down-regulated 200%-1100% after transplant, compared with the pretransplant samples.
In samples from 17 of 34 patients who experienced a relapse after transplantation, both flow cytometry and immunohistochemical analyses confirmed that expression of MHC class II molecules were decreased at relapse.
To see whether this down-regulation was reversible, the researchers treated samples from three patients with posttransplant relapse with interferon gamma, which is known to up-regulate MHC class II protein on myeloid cells and other cell types.
“Culture of these cells with interferon-gamma rapidly induced MHC class II protein expression on leukemic blasts, with essentially full restoration of MHC class II protein expression in nearly all AML blasts after 72 hours,” they wrote, adding that the reversibility of down-regulation of MHC class II in these blasts “strongly suggests that this phenomenon is mediated by an epigenetic mechanism.”
The study was supported by grants to investigators from the National Institutes of Health, Leukemia & Lymphoma Society, and the Barnes-Jewish Hospital Foundation. Dr. Christopher and several coauthors reported receiving grants from the study funders but no other relevant conflicts of interest. Several coauthors reported receiving personal fees and/or research support from industry outside the submitted work.
SOURCE: Christopher MJ et al. N Engl J Med. 2018 Oct 31. doi: 10.1056/NEJMoa1808777.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point:
Major finding: MHC class II genes were down-regulated 200%-1100% after transplant, compared with pretransplant samples.
Study details: Analysis of genetic changes pre- and posttransplant in 15 patients with AML relapse after transplant, 20 patients with relapse after chemotherapy, and 28 patients in a validation sample.
Disclosures: The study was supported by grants to investigators from the National Institutes of Health, Leukemia & Lymphoma Society, and the Barnes-Jewish Hospital Foundation. Dr. Christopher and several coauthors reported receiving grants from the study funders but no other relevant conflicts of interest. Several coauthors reported receiving personal fees and/or research support from industry outside the submitted work.
Source: Christopher MJ et al. N Engl J Med. 2018 Oct 31. doi: 10.1056/NEJMoa1808777.
EC approves venetoclax in combo with rituximab
The European Commission (EC) has approved a new indication for venetoclax (Venclyxto®).
The drug is now approved for use in combination with rituximab to treat patients with relapsed/refractory chronic lymphocytic leukemia (CLL) who have received at least one prior therapy.
The approval is valid in all member states of the European Union as well as Iceland, Liechtenstein, and Norway.
The EC’s approval is based on results from the phase 3 MURANO trial, which were published in The New England Journal of Medicine in March.
The trial included 389 CLL patients who were randomized to receive venetoclax plus rituximab (VEN+R) or bendamustine plus rituximab (B+R). The median follow-up was 23.8 months.
According to an independent review committee, the overall response rate was 92.3% in the VEN+R arm and 72.3% in the B+R arm. The investigator-assessed overall response rates were 93.3% and 67.7%, respectively.
According to investigators, the median progression-free survival (PFS) was not reached in the VEN+R arm and was 17.0 months in the B+R arm (hazard ratio [HR]=0.17; P<0.0001).
According to the independent review committee, the median PFS was not reached in the VEN+R arm and was 18.1 months in the B+R arm (HR=0.20; P<0.0001).
Investigators said the 2-year PFS rate was 84.9% in the VEN+R arm and 36.3% in the B+R arm.
They said the 2-year overall survival rates were 91.9% and 86.6%, respectively (HR=0.48; P<0.0001). The median overall survival was not reached in either arm.
Grade 3/4 adverse events (AEs) with at least a 2% difference in incidence between the treatment arms (in the VEN+R and B+R arms, respectively) included:
- Neutropenia (57.7% and 38.8%)
- Infections and infestations (17.5% and 21.8%)
- Anemia (10.8% and 13.8%)
- Thrombocytopenia (5.7% and 10.1%)
- Febrile neutropenia (3.6% and 9.6%)
- Pneumonia (5.2% and 8.0%)
- Infusion-related reactions (1.5% and 5.3%)
- Tumor lysis syndrome (3.1% and 1.1%)
- Hypotension (0% and 2.7%)
- Hyperglycemia (2.1% and 0%)
- Hypogammaglobulinemia (2.1% and 0%).
Serious AEs with at least a 2% difference in incidence between the arms (in the VEN+R and B+R arms, respectively) were:
- Pneumonia (8.2% and 8.0%)
- Febrile neutropenia (3.6% and 8.5%)
- Pyrexia (2.6% and 6.9%)
- Anemia (1.5% and 2.7%)
- Infusion-related reactions (0.5% and 3.2%)
- Sepsis (0.5% and 2.1%)
- Tumor lysis syndrome (2.1% and 0.5%)
- Hypotension (0% and 2.7%).
Fatal AEs occurred in 5.2% of patients in the VEN+R arm and 5.9% in the B+R arm.
Fatal AEs in the VEN+R arm included pneumonia (n=3), sepsis (n=1), thrombocytopenia (n=1), cardiac failure (n=1), myocardial infarction (n=1), sudden cardiac death (n=1), colorectal cancer (n=1), status epilepticus (n=1), and acute respiratory failure (n=1). Two cases of pneumonia occurred in the setting of progression/Richter’s transformation.
The European Commission (EC) has approved a new indication for venetoclax (Venclyxto®).
The drug is now approved for use in combination with rituximab to treat patients with relapsed/refractory chronic lymphocytic leukemia (CLL) who have received at least one prior therapy.
The approval is valid in all member states of the European Union as well as Iceland, Liechtenstein, and Norway.
The EC’s approval is based on results from the phase 3 MURANO trial, which were published in The New England Journal of Medicine in March.
The trial included 389 CLL patients who were randomized to receive venetoclax plus rituximab (VEN+R) or bendamustine plus rituximab (B+R). The median follow-up was 23.8 months.
According to an independent review committee, the overall response rate was 92.3% in the VEN+R arm and 72.3% in the B+R arm. The investigator-assessed overall response rates were 93.3% and 67.7%, respectively.
According to investigators, the median progression-free survival (PFS) was not reached in the VEN+R arm and was 17.0 months in the B+R arm (hazard ratio [HR]=0.17; P<0.0001).
According to the independent review committee, the median PFS was not reached in the VEN+R arm and was 18.1 months in the B+R arm (HR=0.20; P<0.0001).
Investigators said the 2-year PFS rate was 84.9% in the VEN+R arm and 36.3% in the B+R arm.
They said the 2-year overall survival rates were 91.9% and 86.6%, respectively (HR=0.48; P<0.0001). The median overall survival was not reached in either arm.
Grade 3/4 adverse events (AEs) with at least a 2% difference in incidence between the treatment arms (in the VEN+R and B+R arms, respectively) included:
- Neutropenia (57.7% and 38.8%)
- Infections and infestations (17.5% and 21.8%)
- Anemia (10.8% and 13.8%)
- Thrombocytopenia (5.7% and 10.1%)
- Febrile neutropenia (3.6% and 9.6%)
- Pneumonia (5.2% and 8.0%)
- Infusion-related reactions (1.5% and 5.3%)
- Tumor lysis syndrome (3.1% and 1.1%)
- Hypotension (0% and 2.7%)
- Hyperglycemia (2.1% and 0%)
- Hypogammaglobulinemia (2.1% and 0%).
Serious AEs with at least a 2% difference in incidence between the arms (in the VEN+R and B+R arms, respectively) were:
- Pneumonia (8.2% and 8.0%)
- Febrile neutropenia (3.6% and 8.5%)
- Pyrexia (2.6% and 6.9%)
- Anemia (1.5% and 2.7%)
- Infusion-related reactions (0.5% and 3.2%)
- Sepsis (0.5% and 2.1%)
- Tumor lysis syndrome (2.1% and 0.5%)
- Hypotension (0% and 2.7%).
Fatal AEs occurred in 5.2% of patients in the VEN+R arm and 5.9% in the B+R arm.
Fatal AEs in the VEN+R arm included pneumonia (n=3), sepsis (n=1), thrombocytopenia (n=1), cardiac failure (n=1), myocardial infarction (n=1), sudden cardiac death (n=1), colorectal cancer (n=1), status epilepticus (n=1), and acute respiratory failure (n=1). Two cases of pneumonia occurred in the setting of progression/Richter’s transformation.
The European Commission (EC) has approved a new indication for venetoclax (Venclyxto®).
The drug is now approved for use in combination with rituximab to treat patients with relapsed/refractory chronic lymphocytic leukemia (CLL) who have received at least one prior therapy.
The approval is valid in all member states of the European Union as well as Iceland, Liechtenstein, and Norway.
The EC’s approval is based on results from the phase 3 MURANO trial, which were published in The New England Journal of Medicine in March.
The trial included 389 CLL patients who were randomized to receive venetoclax plus rituximab (VEN+R) or bendamustine plus rituximab (B+R). The median follow-up was 23.8 months.
According to an independent review committee, the overall response rate was 92.3% in the VEN+R arm and 72.3% in the B+R arm. The investigator-assessed overall response rates were 93.3% and 67.7%, respectively.
According to investigators, the median progression-free survival (PFS) was not reached in the VEN+R arm and was 17.0 months in the B+R arm (hazard ratio [HR]=0.17; P<0.0001).
According to the independent review committee, the median PFS was not reached in the VEN+R arm and was 18.1 months in the B+R arm (HR=0.20; P<0.0001).
Investigators said the 2-year PFS rate was 84.9% in the VEN+R arm and 36.3% in the B+R arm.
They said the 2-year overall survival rates were 91.9% and 86.6%, respectively (HR=0.48; P<0.0001). The median overall survival was not reached in either arm.
Grade 3/4 adverse events (AEs) with at least a 2% difference in incidence between the treatment arms (in the VEN+R and B+R arms, respectively) included:
- Neutropenia (57.7% and 38.8%)
- Infections and infestations (17.5% and 21.8%)
- Anemia (10.8% and 13.8%)
- Thrombocytopenia (5.7% and 10.1%)
- Febrile neutropenia (3.6% and 9.6%)
- Pneumonia (5.2% and 8.0%)
- Infusion-related reactions (1.5% and 5.3%)
- Tumor lysis syndrome (3.1% and 1.1%)
- Hypotension (0% and 2.7%)
- Hyperglycemia (2.1% and 0%)
- Hypogammaglobulinemia (2.1% and 0%).
Serious AEs with at least a 2% difference in incidence between the arms (in the VEN+R and B+R arms, respectively) were:
- Pneumonia (8.2% and 8.0%)
- Febrile neutropenia (3.6% and 8.5%)
- Pyrexia (2.6% and 6.9%)
- Anemia (1.5% and 2.7%)
- Infusion-related reactions (0.5% and 3.2%)
- Sepsis (0.5% and 2.1%)
- Tumor lysis syndrome (2.1% and 0.5%)
- Hypotension (0% and 2.7%).
Fatal AEs occurred in 5.2% of patients in the VEN+R arm and 5.9% in the B+R arm.
Fatal AEs in the VEN+R arm included pneumonia (n=3), sepsis (n=1), thrombocytopenia (n=1), cardiac failure (n=1), myocardial infarction (n=1), sudden cardiac death (n=1), colorectal cancer (n=1), status epilepticus (n=1), and acute respiratory failure (n=1). Two cases of pneumonia occurred in the setting of progression/Richter’s transformation.
EVI1 overexpression promotes leukemogenesis, study suggests
Preclinical research suggests the oncoprotein EVI1 can promote leukemogenesis by suppressing erythropoiesis and lymphopoiesis while shifting differentiation toward the expansion of myeloid cells.
Researchers developed a new mouse model that mimics chromosomal rearrangements at 3q26, which are associated with poor-prognosis acute myeloid leukemia (AML), myelodysplastic syndromes, and myeloproliferative neoplasms.
Using the mouse model, the team demonstrated that EVI1 overexpression distorts hematopoiesis and markedly expands premalignant myelopoiesis that eventually results in leukemic transformation.
Archibald Perkins, MD, PhD, of the University of Rochester Medical Center in New York, and his colleagues published these findings in Nature Communications.
The team demonstrated that the “myeloid-skewed phenotype” is dependent upon EVI1-binding DNA. This upregulates Spi1 and encodes the master myeloid regulator PU.1.
When the researchers knocked down Spi1, the myeloid skewing diminished.
“It’s not so pie-in-the-sky anymore,” Dr. Perkins said, “to think we can interrupt the process within the genome that leads to leukemia.”
The researchers first created a mouse model of 3q26 AML with a tetracycline-inducible allele of EVI1 by inserting tetracycline operons within the first exon. This allowed the induction of all three isoforms of EVI1.
These mice were viable and fertile but had no phenotype, which indicated that the allele functioned normally unless induced.
To assess the effect of EVI1 overexpression, the researchers transplanted oncogene-expressing bone marrow mixed 1:1 with wild-type bone marrow into recipient mice.
After confirming successful engraftment, the researchers fed the mice doxycycline-treated food to induce EVI1. The team analyzed cells in the peripheral blood and bone marrow at 10 weeks post-induction.
The researchers observed a more than two-fold expansion of the EVI1-overexpressing compartment in the mouse model.
Suppression of erythropoiesis
The researchers analyzed erythroid lineage in the transplanted mice at 2, 6, and 10 weeks post-induction and found the EVI1-overexpressing cells did not contribute effectively to erythropoiesis.
Using flow cytometry, the researchers quantitated apoptosis and proliferation in erythroid progenitors. They observed a six-fold increase in apoptosis within the erythroblasts compared to wild-type cells.
They also observed a drop in the proliferation of proerythroblasts and erythroblasts compared to wild-type.
Suppression of lymphopoiesis
The researchers observed significantly lower numbers of EVI1-overexpressing B-lineage cells within the bone marrow at 6 and 10 weeks.
And at 10 weeks post-induction, the team observed a decrease in peripheral T cells from approximately 1,800 cells/µL to approximately 750 cells/µL.
EVI1 nearly eliminated the peripheral B cells completely, they noted.
Expansion of myelopoiesis
The team reported that, at 2 weeks post-induction, the EVI1-overexpressing bone marrow and control bone marrow showed the same number of myeloid cells.
But at 6 and 10 weeks post-induction, the EVI1-overexpressing myeloid compartment expanded markedly.
The researchers aged a cohort of five mice transplanted with the 1:1 mix of wild-type and EVI1 bone marrow cells to determine if chronic overexpression of EVI1 results in leukemia.
All five mice died at 90 to 119 days of doxycycline treatment. Analysis revealed AML in all mice. Bone marrows were replete with blasts, and the peripheral blood revealed severe anemia.
The researchers then proceeded to establish the relationship between EVI1 and Spi1/PU.1 transcriptional regulation.
They documented binding of EVI1 to the regulatory element -14kbURE, which, together with EVI1., induced upregulation of PU.1.
When the team knocked down PU.1, myeloid skewing diminished. This, they say, indicates PU.1 is necessary for EVI1-induced myeloid expansion.
Funding for this research was provided by the National Institutes of Health, New York State Stem Cell Science, the Wilmot Cancer Institute, and the Clinical and Translational Science Institute at the University of Rochester.
The authors had no competing interests to disclose.
Preclinical research suggests the oncoprotein EVI1 can promote leukemogenesis by suppressing erythropoiesis and lymphopoiesis while shifting differentiation toward the expansion of myeloid cells.
Researchers developed a new mouse model that mimics chromosomal rearrangements at 3q26, which are associated with poor-prognosis acute myeloid leukemia (AML), myelodysplastic syndromes, and myeloproliferative neoplasms.
Using the mouse model, the team demonstrated that EVI1 overexpression distorts hematopoiesis and markedly expands premalignant myelopoiesis that eventually results in leukemic transformation.
Archibald Perkins, MD, PhD, of the University of Rochester Medical Center in New York, and his colleagues published these findings in Nature Communications.
The team demonstrated that the “myeloid-skewed phenotype” is dependent upon EVI1-binding DNA. This upregulates Spi1 and encodes the master myeloid regulator PU.1.
When the researchers knocked down Spi1, the myeloid skewing diminished.
“It’s not so pie-in-the-sky anymore,” Dr. Perkins said, “to think we can interrupt the process within the genome that leads to leukemia.”
The researchers first created a mouse model of 3q26 AML with a tetracycline-inducible allele of EVI1 by inserting tetracycline operons within the first exon. This allowed the induction of all three isoforms of EVI1.
These mice were viable and fertile but had no phenotype, which indicated that the allele functioned normally unless induced.
To assess the effect of EVI1 overexpression, the researchers transplanted oncogene-expressing bone marrow mixed 1:1 with wild-type bone marrow into recipient mice.
After confirming successful engraftment, the researchers fed the mice doxycycline-treated food to induce EVI1. The team analyzed cells in the peripheral blood and bone marrow at 10 weeks post-induction.
The researchers observed a more than two-fold expansion of the EVI1-overexpressing compartment in the mouse model.
Suppression of erythropoiesis
The researchers analyzed erythroid lineage in the transplanted mice at 2, 6, and 10 weeks post-induction and found the EVI1-overexpressing cells did not contribute effectively to erythropoiesis.
Using flow cytometry, the researchers quantitated apoptosis and proliferation in erythroid progenitors. They observed a six-fold increase in apoptosis within the erythroblasts compared to wild-type cells.
They also observed a drop in the proliferation of proerythroblasts and erythroblasts compared to wild-type.
Suppression of lymphopoiesis
The researchers observed significantly lower numbers of EVI1-overexpressing B-lineage cells within the bone marrow at 6 and 10 weeks.
And at 10 weeks post-induction, the team observed a decrease in peripheral T cells from approximately 1,800 cells/µL to approximately 750 cells/µL.
EVI1 nearly eliminated the peripheral B cells completely, they noted.
Expansion of myelopoiesis
The team reported that, at 2 weeks post-induction, the EVI1-overexpressing bone marrow and control bone marrow showed the same number of myeloid cells.
But at 6 and 10 weeks post-induction, the EVI1-overexpressing myeloid compartment expanded markedly.
The researchers aged a cohort of five mice transplanted with the 1:1 mix of wild-type and EVI1 bone marrow cells to determine if chronic overexpression of EVI1 results in leukemia.
All five mice died at 90 to 119 days of doxycycline treatment. Analysis revealed AML in all mice. Bone marrows were replete with blasts, and the peripheral blood revealed severe anemia.
The researchers then proceeded to establish the relationship between EVI1 and Spi1/PU.1 transcriptional regulation.
They documented binding of EVI1 to the regulatory element -14kbURE, which, together with EVI1., induced upregulation of PU.1.
When the team knocked down PU.1, myeloid skewing diminished. This, they say, indicates PU.1 is necessary for EVI1-induced myeloid expansion.
Funding for this research was provided by the National Institutes of Health, New York State Stem Cell Science, the Wilmot Cancer Institute, and the Clinical and Translational Science Institute at the University of Rochester.
The authors had no competing interests to disclose.
Preclinical research suggests the oncoprotein EVI1 can promote leukemogenesis by suppressing erythropoiesis and lymphopoiesis while shifting differentiation toward the expansion of myeloid cells.
Researchers developed a new mouse model that mimics chromosomal rearrangements at 3q26, which are associated with poor-prognosis acute myeloid leukemia (AML), myelodysplastic syndromes, and myeloproliferative neoplasms.
Using the mouse model, the team demonstrated that EVI1 overexpression distorts hematopoiesis and markedly expands premalignant myelopoiesis that eventually results in leukemic transformation.
Archibald Perkins, MD, PhD, of the University of Rochester Medical Center in New York, and his colleagues published these findings in Nature Communications.
The team demonstrated that the “myeloid-skewed phenotype” is dependent upon EVI1-binding DNA. This upregulates Spi1 and encodes the master myeloid regulator PU.1.
When the researchers knocked down Spi1, the myeloid skewing diminished.
“It’s not so pie-in-the-sky anymore,” Dr. Perkins said, “to think we can interrupt the process within the genome that leads to leukemia.”
The researchers first created a mouse model of 3q26 AML with a tetracycline-inducible allele of EVI1 by inserting tetracycline operons within the first exon. This allowed the induction of all three isoforms of EVI1.
These mice were viable and fertile but had no phenotype, which indicated that the allele functioned normally unless induced.
To assess the effect of EVI1 overexpression, the researchers transplanted oncogene-expressing bone marrow mixed 1:1 with wild-type bone marrow into recipient mice.
After confirming successful engraftment, the researchers fed the mice doxycycline-treated food to induce EVI1. The team analyzed cells in the peripheral blood and bone marrow at 10 weeks post-induction.
The researchers observed a more than two-fold expansion of the EVI1-overexpressing compartment in the mouse model.
Suppression of erythropoiesis
The researchers analyzed erythroid lineage in the transplanted mice at 2, 6, and 10 weeks post-induction and found the EVI1-overexpressing cells did not contribute effectively to erythropoiesis.
Using flow cytometry, the researchers quantitated apoptosis and proliferation in erythroid progenitors. They observed a six-fold increase in apoptosis within the erythroblasts compared to wild-type cells.
They also observed a drop in the proliferation of proerythroblasts and erythroblasts compared to wild-type.
Suppression of lymphopoiesis
The researchers observed significantly lower numbers of EVI1-overexpressing B-lineage cells within the bone marrow at 6 and 10 weeks.
And at 10 weeks post-induction, the team observed a decrease in peripheral T cells from approximately 1,800 cells/µL to approximately 750 cells/µL.
EVI1 nearly eliminated the peripheral B cells completely, they noted.
Expansion of myelopoiesis
The team reported that, at 2 weeks post-induction, the EVI1-overexpressing bone marrow and control bone marrow showed the same number of myeloid cells.
But at 6 and 10 weeks post-induction, the EVI1-overexpressing myeloid compartment expanded markedly.
The researchers aged a cohort of five mice transplanted with the 1:1 mix of wild-type and EVI1 bone marrow cells to determine if chronic overexpression of EVI1 results in leukemia.
All five mice died at 90 to 119 days of doxycycline treatment. Analysis revealed AML in all mice. Bone marrows were replete with blasts, and the peripheral blood revealed severe anemia.
The researchers then proceeded to establish the relationship between EVI1 and Spi1/PU.1 transcriptional regulation.
They documented binding of EVI1 to the regulatory element -14kbURE, which, together with EVI1., induced upregulation of PU.1.
When the team knocked down PU.1, myeloid skewing diminished. This, they say, indicates PU.1 is necessary for EVI1-induced myeloid expansion.
Funding for this research was provided by the National Institutes of Health, New York State Stem Cell Science, the Wilmot Cancer Institute, and the Clinical and Translational Science Institute at the University of Rochester.
The authors had no competing interests to disclose.
Palliative care guidelines relevant for hematologists, doc says
The latest edition of the national palliative care guidelines provides new clinical strategies relevant to hematology practice in the United States, according to a physician-researcher specializing in hematology.
The Clinical Practice Guidelines for Quality Palliative Care, 4th edition, represents a “blueprint for what it looks like to provide high-quality, comprehensive palliative care to people with serious illness,” said Thomas W. LeBlanc, MD, a physician-researcher at Duke University School of Medicine in Durham, North Carolina.
However, unlike previous editions, this update to the guidelines emphasizes the importance of palliative care provided by both primary care and specialty care clinicians.
“Part of this report is about trying to raise the game of everybody in medicine and provide a higher basic level of primary palliative care to all people with serious illness, but then also to figure out who has higher levels of needs where the specialists should be applied, since they are a scarce resource,” Dr. LeBlanc said.
The latest edition helps establish a foundation for gold standard palliative care for people living with serious illness, regardless of diagnosis, prognosis, setting, or age, according to The National Coalition for Hospice and Palliative Care, which published the clinical practice guidelines.
The update was developed by the National Consensus Project for Quality Palliative Care (NCP), which includes 16 national organizations with palliative care and hospice expertise, and is endorsed by more than 80 national organizations, including the American Society of Hematology.
One key reason for the update, according to NCP, was to acknowledge that today’s healthcare system may not be meeting patients’ palliative care needs.
Specifically, the guidelines call on clinicians who don’t practice palliative care to integrate palliative care principles into their routine assessment of seriously ill patients with conditions such as heart failure, lung disease, and cancer.
That differs from the way palliative care is traditionally practiced, in which specially trained doctors, nurses, and other specialists provide that support.
An issue with that traditional model is a shortage of specialized clinicians to meet palliative care needs, said Dr. LeBlanc, whose clinical practice and research focuses on palliative care needs of patients with hematologic malignancies.
“Palliative care has matured as a field such that we are now actually facing workforce shortage issues and really fundamental questions about who really needs us the most and how we increase our reach to improve the lives of more patients and families facing serious illness,” he said.
That’s a major driver behind the emphasis in the latest guidelines on providing palliative care in the community, coordinating care, and dealing with care transitions, Dr. LeBlanc added.
“I hope that this document will help to demonstrate the value and the need for palliative care specialists and for improvements in primary care in the care of patients with hematologic diseases in general,” he said. “To me, this adds increasing legitimacy to this whole field.”
The latest edition of the national palliative care guidelines provides new clinical strategies relevant to hematology practice in the United States, according to a physician-researcher specializing in hematology.
The Clinical Practice Guidelines for Quality Palliative Care, 4th edition, represents a “blueprint for what it looks like to provide high-quality, comprehensive palliative care to people with serious illness,” said Thomas W. LeBlanc, MD, a physician-researcher at Duke University School of Medicine in Durham, North Carolina.
However, unlike previous editions, this update to the guidelines emphasizes the importance of palliative care provided by both primary care and specialty care clinicians.
“Part of this report is about trying to raise the game of everybody in medicine and provide a higher basic level of primary palliative care to all people with serious illness, but then also to figure out who has higher levels of needs where the specialists should be applied, since they are a scarce resource,” Dr. LeBlanc said.
The latest edition helps establish a foundation for gold standard palliative care for people living with serious illness, regardless of diagnosis, prognosis, setting, or age, according to The National Coalition for Hospice and Palliative Care, which published the clinical practice guidelines.
The update was developed by the National Consensus Project for Quality Palliative Care (NCP), which includes 16 national organizations with palliative care and hospice expertise, and is endorsed by more than 80 national organizations, including the American Society of Hematology.
One key reason for the update, according to NCP, was to acknowledge that today’s healthcare system may not be meeting patients’ palliative care needs.
Specifically, the guidelines call on clinicians who don’t practice palliative care to integrate palliative care principles into their routine assessment of seriously ill patients with conditions such as heart failure, lung disease, and cancer.
That differs from the way palliative care is traditionally practiced, in which specially trained doctors, nurses, and other specialists provide that support.
An issue with that traditional model is a shortage of specialized clinicians to meet palliative care needs, said Dr. LeBlanc, whose clinical practice and research focuses on palliative care needs of patients with hematologic malignancies.
“Palliative care has matured as a field such that we are now actually facing workforce shortage issues and really fundamental questions about who really needs us the most and how we increase our reach to improve the lives of more patients and families facing serious illness,” he said.
That’s a major driver behind the emphasis in the latest guidelines on providing palliative care in the community, coordinating care, and dealing with care transitions, Dr. LeBlanc added.
“I hope that this document will help to demonstrate the value and the need for palliative care specialists and for improvements in primary care in the care of patients with hematologic diseases in general,” he said. “To me, this adds increasing legitimacy to this whole field.”
The latest edition of the national palliative care guidelines provides new clinical strategies relevant to hematology practice in the United States, according to a physician-researcher specializing in hematology.
The Clinical Practice Guidelines for Quality Palliative Care, 4th edition, represents a “blueprint for what it looks like to provide high-quality, comprehensive palliative care to people with serious illness,” said Thomas W. LeBlanc, MD, a physician-researcher at Duke University School of Medicine in Durham, North Carolina.
However, unlike previous editions, this update to the guidelines emphasizes the importance of palliative care provided by both primary care and specialty care clinicians.
“Part of this report is about trying to raise the game of everybody in medicine and provide a higher basic level of primary palliative care to all people with serious illness, but then also to figure out who has higher levels of needs where the specialists should be applied, since they are a scarce resource,” Dr. LeBlanc said.
The latest edition helps establish a foundation for gold standard palliative care for people living with serious illness, regardless of diagnosis, prognosis, setting, or age, according to The National Coalition for Hospice and Palliative Care, which published the clinical practice guidelines.
The update was developed by the National Consensus Project for Quality Palliative Care (NCP), which includes 16 national organizations with palliative care and hospice expertise, and is endorsed by more than 80 national organizations, including the American Society of Hematology.
One key reason for the update, according to NCP, was to acknowledge that today’s healthcare system may not be meeting patients’ palliative care needs.
Specifically, the guidelines call on clinicians who don’t practice palliative care to integrate palliative care principles into their routine assessment of seriously ill patients with conditions such as heart failure, lung disease, and cancer.
That differs from the way palliative care is traditionally practiced, in which specially trained doctors, nurses, and other specialists provide that support.
An issue with that traditional model is a shortage of specialized clinicians to meet palliative care needs, said Dr. LeBlanc, whose clinical practice and research focuses on palliative care needs of patients with hematologic malignancies.
“Palliative care has matured as a field such that we are now actually facing workforce shortage issues and really fundamental questions about who really needs us the most and how we increase our reach to improve the lives of more patients and families facing serious illness,” he said.
That’s a major driver behind the emphasis in the latest guidelines on providing palliative care in the community, coordinating care, and dealing with care transitions, Dr. LeBlanc added.
“I hope that this document will help to demonstrate the value and the need for palliative care specialists and for improvements in primary care in the care of patients with hematologic diseases in general,” he said. “To me, this adds increasing legitimacy to this whole field.”
Team finds potential therapeutic target for AML
Researchers have found the cancer-associated pseudokinase Tribbles 2 (TRIB2) to be a potential therapeutic target in solid tumors and blood cancers, including acute myeloid leukemia (AML).
Previous research had described TRIB2 as a target of small-molecule protein kinase inhibitors originally designed to interfere with kinase domains of the epidermal growth factor receptor (EGFR) tyrosine kinase family.
Using a thermal shift assay, the team discovered TRIB2-binding compounds within the Published Kinase Inhibitor Set (PKIS). They then employed a biochemical drug repurposing approach to classify compounds that either stabilized or destabilized TRIB2 in vitro.
The researchers found that afatinib, which is already approved by the U.S. Food and Drug Administration to treat non-small cell lung cancer, led to rapid TRIB2 degradation in human AML cells.
Patrick A. Eyers, PhD, of the University of Liverpool in the U.K., and his colleagues published their findings in Science Signaling.
The team found afatinib to be relatively specific for EGFR and human epidermal growth factor receptor 2 (HER2) at nanomolar concentrations in cells.
The researchers confirmed that at least two TRIB2 Cys residues interact with afatinib in vitro.
The team also discovered TRIB2 could be destabilized by neratinib and osimertinib in vitro.
“Our data prove that the cellular mechanism by which TRIB2 stability is regulated by compounds is proteasome-based,” the researchers wrote, “and we speculate that an afatinib-induced conformational change might induce TRIB2 ubiquitination.”
The researchers plan to study further TRIB2 small-molecule interactions with dynamic changes in ubiquitination status.
Furthermore, they report their work demonstrates that covalent inhibitors such as afatinib have TRIB2-degrading activity in human cells at micromolar concentrations.
The researchers determined that afatinib has similar efficacy to the TRIB2-destabilizing quinazoline neratinib at similar ranges.
The team believes their data “raise the intriguing possibility that clinical inhibitors might be used as TRIB2-degrading agents in research, and possibly clinical, contexts.”
“A long-standing goal in cancer research is drug-induced degradation of oncogenic proteins,” Dr. Eyers commented. “Our study highlights how information obtained with ‘off-target’ effects of known drugs is potentially useful because it might be exploited in the future to help eliminate a protein that is involved in a completely different type of cancer.”
The TRIB proteins play many diverse roles in cell signaling, development, and cancer. According to a paper in Developmental Dynamics, they were named after the small, round, fictional organisms from the original Star Trek television series. Their major role was to eat and reproduce.
This work was funded by two U.K. Biotechnology and Biological Sciences Research Council Doctoral Training Partnership studentships, a Tools and Resources Development Fund award, Royal Society Research Grants, North West Cancer Research grants, and funding from the National Institutes of Health.
The authors disclosed no perceived conflicts of interest, although several authors are affiliated with the Structural Genomics Consortium at the University of North Carolina at Chapel Hill, which receives direct funds from various pharmaceutical companies but remains entirely independent.
Researchers have found the cancer-associated pseudokinase Tribbles 2 (TRIB2) to be a potential therapeutic target in solid tumors and blood cancers, including acute myeloid leukemia (AML).
Previous research had described TRIB2 as a target of small-molecule protein kinase inhibitors originally designed to interfere with kinase domains of the epidermal growth factor receptor (EGFR) tyrosine kinase family.
Using a thermal shift assay, the team discovered TRIB2-binding compounds within the Published Kinase Inhibitor Set (PKIS). They then employed a biochemical drug repurposing approach to classify compounds that either stabilized or destabilized TRIB2 in vitro.
The researchers found that afatinib, which is already approved by the U.S. Food and Drug Administration to treat non-small cell lung cancer, led to rapid TRIB2 degradation in human AML cells.
Patrick A. Eyers, PhD, of the University of Liverpool in the U.K., and his colleagues published their findings in Science Signaling.
The team found afatinib to be relatively specific for EGFR and human epidermal growth factor receptor 2 (HER2) at nanomolar concentrations in cells.
The researchers confirmed that at least two TRIB2 Cys residues interact with afatinib in vitro.
The team also discovered TRIB2 could be destabilized by neratinib and osimertinib in vitro.
“Our data prove that the cellular mechanism by which TRIB2 stability is regulated by compounds is proteasome-based,” the researchers wrote, “and we speculate that an afatinib-induced conformational change might induce TRIB2 ubiquitination.”
The researchers plan to study further TRIB2 small-molecule interactions with dynamic changes in ubiquitination status.
Furthermore, they report their work demonstrates that covalent inhibitors such as afatinib have TRIB2-degrading activity in human cells at micromolar concentrations.
The researchers determined that afatinib has similar efficacy to the TRIB2-destabilizing quinazoline neratinib at similar ranges.
The team believes their data “raise the intriguing possibility that clinical inhibitors might be used as TRIB2-degrading agents in research, and possibly clinical, contexts.”
“A long-standing goal in cancer research is drug-induced degradation of oncogenic proteins,” Dr. Eyers commented. “Our study highlights how information obtained with ‘off-target’ effects of known drugs is potentially useful because it might be exploited in the future to help eliminate a protein that is involved in a completely different type of cancer.”
The TRIB proteins play many diverse roles in cell signaling, development, and cancer. According to a paper in Developmental Dynamics, they were named after the small, round, fictional organisms from the original Star Trek television series. Their major role was to eat and reproduce.
This work was funded by two U.K. Biotechnology and Biological Sciences Research Council Doctoral Training Partnership studentships, a Tools and Resources Development Fund award, Royal Society Research Grants, North West Cancer Research grants, and funding from the National Institutes of Health.
The authors disclosed no perceived conflicts of interest, although several authors are affiliated with the Structural Genomics Consortium at the University of North Carolina at Chapel Hill, which receives direct funds from various pharmaceutical companies but remains entirely independent.
Researchers have found the cancer-associated pseudokinase Tribbles 2 (TRIB2) to be a potential therapeutic target in solid tumors and blood cancers, including acute myeloid leukemia (AML).
Previous research had described TRIB2 as a target of small-molecule protein kinase inhibitors originally designed to interfere with kinase domains of the epidermal growth factor receptor (EGFR) tyrosine kinase family.
Using a thermal shift assay, the team discovered TRIB2-binding compounds within the Published Kinase Inhibitor Set (PKIS). They then employed a biochemical drug repurposing approach to classify compounds that either stabilized or destabilized TRIB2 in vitro.
The researchers found that afatinib, which is already approved by the U.S. Food and Drug Administration to treat non-small cell lung cancer, led to rapid TRIB2 degradation in human AML cells.
Patrick A. Eyers, PhD, of the University of Liverpool in the U.K., and his colleagues published their findings in Science Signaling.
The team found afatinib to be relatively specific for EGFR and human epidermal growth factor receptor 2 (HER2) at nanomolar concentrations in cells.
The researchers confirmed that at least two TRIB2 Cys residues interact with afatinib in vitro.
The team also discovered TRIB2 could be destabilized by neratinib and osimertinib in vitro.
“Our data prove that the cellular mechanism by which TRIB2 stability is regulated by compounds is proteasome-based,” the researchers wrote, “and we speculate that an afatinib-induced conformational change might induce TRIB2 ubiquitination.”
The researchers plan to study further TRIB2 small-molecule interactions with dynamic changes in ubiquitination status.
Furthermore, they report their work demonstrates that covalent inhibitors such as afatinib have TRIB2-degrading activity in human cells at micromolar concentrations.
The researchers determined that afatinib has similar efficacy to the TRIB2-destabilizing quinazoline neratinib at similar ranges.
The team believes their data “raise the intriguing possibility that clinical inhibitors might be used as TRIB2-degrading agents in research, and possibly clinical, contexts.”
“A long-standing goal in cancer research is drug-induced degradation of oncogenic proteins,” Dr. Eyers commented. “Our study highlights how information obtained with ‘off-target’ effects of known drugs is potentially useful because it might be exploited in the future to help eliminate a protein that is involved in a completely different type of cancer.”
The TRIB proteins play many diverse roles in cell signaling, development, and cancer. According to a paper in Developmental Dynamics, they were named after the small, round, fictional organisms from the original Star Trek television series. Their major role was to eat and reproduce.
This work was funded by two U.K. Biotechnology and Biological Sciences Research Council Doctoral Training Partnership studentships, a Tools and Resources Development Fund award, Royal Society Research Grants, North West Cancer Research grants, and funding from the National Institutes of Health.
The authors disclosed no perceived conflicts of interest, although several authors are affiliated with the Structural Genomics Consortium at the University of North Carolina at Chapel Hill, which receives direct funds from various pharmaceutical companies but remains entirely independent.
Team tracks changes in height, weight in pediatric ALL
New research suggests several factors may be associated with the risk of short stature and excess weight gain in children with acute lymphoblastic leukemia (ALL).
Researchers found that patients who were younger at ALL diagnosis had an increased risk of becoming overweight or obese both during and after therapy.
Patients had an increased risk of short stature after therapy if they were older at diagnosis or had standard or high-risk disease, higher white blood cell counts at diagnosis, and central nervous system disease.
The researchers reported these findings in Cancer.
The team looked at 372 children with ALL, reviewing changes in their body mass index (BMI), weight, and height from diagnosis to 5 years after treatment ended.
The patients were treated with the Total XV protocol between 2000 and 2007 (NCT00137111). They received 6 weeks of induction therapy, 8 weeks of consolidation, and continuation for 120 weeks in females and 146 weeks in males.
BMI changes
Roughly a quarter of patients were overweight or obese at diagnosis, but that increased to roughly half of patients by the time they had been off therapy for 5 years.
“Over the whole population that was studied, we found statistically significant weight gain even during remission-induction therapy,” said study author Hiroto Inaba, MD, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
Patients’ median BMI z scores increased significantly during induction (P<0.001) and reinduction (P=0.001) with glucocorticoid therapy as well as in the first year after therapy ended (P=0.006).
At various points during treatment, there were significant differences in BMI z scores according to sex, race, and disease risk group. However, these differences were not present after therapy.
On the other hand, there were significant differences in BMI z scores according to age both during and after therapy.
Between week 21 of treatment and 3 years after therapy ended, patients who were ages 2 to 9 at diagnosis had median BMI z scores that were significantly higher than scores of patients who were age 10 or older at diagnosis (P≤0.033 for all time points).
The researchers also found that patients who were of a healthy weight or underweight at the time of diagnosis had a significantly higher risk of becoming overweight or obese during or after therapy if they were ages 2 to 9 at diagnosis, compared to the older patients (P=0.001).
Height changes
The researchers found that height z scores declined during treatment and improved after it ended, although z scores “never improved to the levels noted at the time of diagnosis.”
Median height z scores at the end of induction and in continuation weeks 1 to 21 were significantly higher in patients age 10 or older at diagnosis than in patients ages to 2 to 9 at diagnosis (P≤0.038 for all time points).
However, the median height z scores at 5 years off therapy were significantly higher for the younger patients than for the older patients (P=0.011).
The median height z scores were higher for patients with low-risk disease than for standard- or high-risk patients in weeks 17, 21, 48, and 146 of treatment and at 1 to 3 years after therapy ended (P≤0.024 for all time points).
At 3 years to 5 years after treatment ended, the median height z scores were significantly higher among patients with white blood cell counts below 50 × 109/L at diagnosis (P≤0.018 for all time points).
Patients without central nervous system disease had significantly higher median height z scores at 3 years after treatment ended (P=0.029).
Males had significantly higher median height z scores than females in weeks 96 and 120 of therapy (P≤0.009 for both time points).
And white patients had higher median height z scores than black patients at 2 to 4 years after treatment ended (P≤0.027 for all time points).
Implications
To address the issue of excess weight gain in ALL patients, the researchers suggested early interventions, such as education about proper diet and exercise.
“When you look at the literature of childhood obesity prevention for the general population, there are interventions that could also help ALL patients,” said study author Emily Browne, of St. Jude.
“But we need to adapt those recommendations to take the cancer therapy into account.”
For the issue of height, the researchers recommended evaluating certain patients for growth hormone deficiency.
The team also noted that further study is needed to determine whether emerging therapeutic approaches can reduce toxicities without compromising antileukemic effects.
“We are hoping new therapeutic options can decrease intensity of chemotherapy and keep normal tissues intact,” Dr. Inaba said. “But until then, we’re collaborating with multiple clinical departments to help ensure a good, quality cure and a good quality of life in survivorship.”
This research was supported by grants from the National Institutes of Health and ALSAC, the fundraising and awareness organization of St. Jude.
New research suggests several factors may be associated with the risk of short stature and excess weight gain in children with acute lymphoblastic leukemia (ALL).
Researchers found that patients who were younger at ALL diagnosis had an increased risk of becoming overweight or obese both during and after therapy.
Patients had an increased risk of short stature after therapy if they were older at diagnosis or had standard or high-risk disease, higher white blood cell counts at diagnosis, and central nervous system disease.
The researchers reported these findings in Cancer.
The team looked at 372 children with ALL, reviewing changes in their body mass index (BMI), weight, and height from diagnosis to 5 years after treatment ended.
The patients were treated with the Total XV protocol between 2000 and 2007 (NCT00137111). They received 6 weeks of induction therapy, 8 weeks of consolidation, and continuation for 120 weeks in females and 146 weeks in males.
BMI changes
Roughly a quarter of patients were overweight or obese at diagnosis, but that increased to roughly half of patients by the time they had been off therapy for 5 years.
“Over the whole population that was studied, we found statistically significant weight gain even during remission-induction therapy,” said study author Hiroto Inaba, MD, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
Patients’ median BMI z scores increased significantly during induction (P<0.001) and reinduction (P=0.001) with glucocorticoid therapy as well as in the first year after therapy ended (P=0.006).
At various points during treatment, there were significant differences in BMI z scores according to sex, race, and disease risk group. However, these differences were not present after therapy.
On the other hand, there were significant differences in BMI z scores according to age both during and after therapy.
Between week 21 of treatment and 3 years after therapy ended, patients who were ages 2 to 9 at diagnosis had median BMI z scores that were significantly higher than scores of patients who were age 10 or older at diagnosis (P≤0.033 for all time points).
The researchers also found that patients who were of a healthy weight or underweight at the time of diagnosis had a significantly higher risk of becoming overweight or obese during or after therapy if they were ages 2 to 9 at diagnosis, compared to the older patients (P=0.001).
Height changes
The researchers found that height z scores declined during treatment and improved after it ended, although z scores “never improved to the levels noted at the time of diagnosis.”
Median height z scores at the end of induction and in continuation weeks 1 to 21 were significantly higher in patients age 10 or older at diagnosis than in patients ages to 2 to 9 at diagnosis (P≤0.038 for all time points).
However, the median height z scores at 5 years off therapy were significantly higher for the younger patients than for the older patients (P=0.011).
The median height z scores were higher for patients with low-risk disease than for standard- or high-risk patients in weeks 17, 21, 48, and 146 of treatment and at 1 to 3 years after therapy ended (P≤0.024 for all time points).
At 3 years to 5 years after treatment ended, the median height z scores were significantly higher among patients with white blood cell counts below 50 × 109/L at diagnosis (P≤0.018 for all time points).
Patients without central nervous system disease had significantly higher median height z scores at 3 years after treatment ended (P=0.029).
Males had significantly higher median height z scores than females in weeks 96 and 120 of therapy (P≤0.009 for both time points).
And white patients had higher median height z scores than black patients at 2 to 4 years after treatment ended (P≤0.027 for all time points).
Implications
To address the issue of excess weight gain in ALL patients, the researchers suggested early interventions, such as education about proper diet and exercise.
“When you look at the literature of childhood obesity prevention for the general population, there are interventions that could also help ALL patients,” said study author Emily Browne, of St. Jude.
“But we need to adapt those recommendations to take the cancer therapy into account.”
For the issue of height, the researchers recommended evaluating certain patients for growth hormone deficiency.
The team also noted that further study is needed to determine whether emerging therapeutic approaches can reduce toxicities without compromising antileukemic effects.
“We are hoping new therapeutic options can decrease intensity of chemotherapy and keep normal tissues intact,” Dr. Inaba said. “But until then, we’re collaborating with multiple clinical departments to help ensure a good, quality cure and a good quality of life in survivorship.”
This research was supported by grants from the National Institutes of Health and ALSAC, the fundraising and awareness organization of St. Jude.
New research suggests several factors may be associated with the risk of short stature and excess weight gain in children with acute lymphoblastic leukemia (ALL).
Researchers found that patients who were younger at ALL diagnosis had an increased risk of becoming overweight or obese both during and after therapy.
Patients had an increased risk of short stature after therapy if they were older at diagnosis or had standard or high-risk disease, higher white blood cell counts at diagnosis, and central nervous system disease.
The researchers reported these findings in Cancer.
The team looked at 372 children with ALL, reviewing changes in their body mass index (BMI), weight, and height from diagnosis to 5 years after treatment ended.
The patients were treated with the Total XV protocol between 2000 and 2007 (NCT00137111). They received 6 weeks of induction therapy, 8 weeks of consolidation, and continuation for 120 weeks in females and 146 weeks in males.
BMI changes
Roughly a quarter of patients were overweight or obese at diagnosis, but that increased to roughly half of patients by the time they had been off therapy for 5 years.
“Over the whole population that was studied, we found statistically significant weight gain even during remission-induction therapy,” said study author Hiroto Inaba, MD, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
Patients’ median BMI z scores increased significantly during induction (P<0.001) and reinduction (P=0.001) with glucocorticoid therapy as well as in the first year after therapy ended (P=0.006).
At various points during treatment, there were significant differences in BMI z scores according to sex, race, and disease risk group. However, these differences were not present after therapy.
On the other hand, there were significant differences in BMI z scores according to age both during and after therapy.
Between week 21 of treatment and 3 years after therapy ended, patients who were ages 2 to 9 at diagnosis had median BMI z scores that were significantly higher than scores of patients who were age 10 or older at diagnosis (P≤0.033 for all time points).
The researchers also found that patients who were of a healthy weight or underweight at the time of diagnosis had a significantly higher risk of becoming overweight or obese during or after therapy if they were ages 2 to 9 at diagnosis, compared to the older patients (P=0.001).
Height changes
The researchers found that height z scores declined during treatment and improved after it ended, although z scores “never improved to the levels noted at the time of diagnosis.”
Median height z scores at the end of induction and in continuation weeks 1 to 21 were significantly higher in patients age 10 or older at diagnosis than in patients ages to 2 to 9 at diagnosis (P≤0.038 for all time points).
However, the median height z scores at 5 years off therapy were significantly higher for the younger patients than for the older patients (P=0.011).
The median height z scores were higher for patients with low-risk disease than for standard- or high-risk patients in weeks 17, 21, 48, and 146 of treatment and at 1 to 3 years after therapy ended (P≤0.024 for all time points).
At 3 years to 5 years after treatment ended, the median height z scores were significantly higher among patients with white blood cell counts below 50 × 109/L at diagnosis (P≤0.018 for all time points).
Patients without central nervous system disease had significantly higher median height z scores at 3 years after treatment ended (P=0.029).
Males had significantly higher median height z scores than females in weeks 96 and 120 of therapy (P≤0.009 for both time points).
And white patients had higher median height z scores than black patients at 2 to 4 years after treatment ended (P≤0.027 for all time points).
Implications
To address the issue of excess weight gain in ALL patients, the researchers suggested early interventions, such as education about proper diet and exercise.
“When you look at the literature of childhood obesity prevention for the general population, there are interventions that could also help ALL patients,” said study author Emily Browne, of St. Jude.
“But we need to adapt those recommendations to take the cancer therapy into account.”
For the issue of height, the researchers recommended evaluating certain patients for growth hormone deficiency.
The team also noted that further study is needed to determine whether emerging therapeutic approaches can reduce toxicities without compromising antileukemic effects.
“We are hoping new therapeutic options can decrease intensity of chemotherapy and keep normal tissues intact,” Dr. Inaba said. “But until then, we’re collaborating with multiple clinical departments to help ensure a good, quality cure and a good quality of life in survivorship.”
This research was supported by grants from the National Institutes of Health and ALSAC, the fundraising and awareness organization of St. Jude.