User login
UCART19 can bridge to transplant in adults
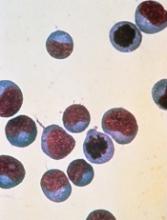
LISBON—Preliminary data from a phase 1 trial suggest UCART19 can serve as a bridge to transplant in adults with relapsed/refractory (R/R) B-cell acute lymphoblastic leukemia (B-ALL).
This “universal,” donor-derived, chimeric antigen receptor (CAR) T-cell therapy produced complete responses (CRs) in 6 of 9 patients treated.
Five of the patients who were negative for minimal residual disease (MRD) proceeded to allogeneic hematopoietic stem cell transplant (allo-HSCT).
Two of the 5 patients were still alive and in MRD-negative remission at last follow-up, but 1 had relapsed and 2 died.
A total of 4 patients died in this trial—1 from a dose-limiting toxicity (DLT), 1 from progressive disease, 1 from infection, and 1 from pulmonary hemorrhage.
“These early results for UCART19 are very encouraging, both in terms of manageable safety and the impressive complete molecular remission rate in these hard-to-treat adult patients with R/R B-ALL,” said principal investigator Reuben Benjamin, MBBS, PhD, a consultant hematologist at King’s College London in the UK.
Dr Benjamin presented these results, from the CALM trial, at the 44th Annual Meeting of the EBMT (abstract OS10-4*).
The CALM trial is sponsored by Servier. In 2015, Servier acquired exclusive rights from Cellectis for UCART19, which is being co-developed by Servier and Pfizer.
Patients and treatment
Dr Benjamin presented results in 9 patients with R/R B-ALL who had a median age of 23 (range, 18-49).
All patients had morphological disease or an MRD level of at least 1 x 10-3 (via flow cytometry and/or qPCR) at baseline. The median disease burden was 8% (range, 0-95).
Four patients had received 1 to 3 prior therapies, and 5 had received 4 or more prior treatments. Six patients had prior inotuzumab ozogamicin, 2 had prior blinatumomab, and 7 had prior allo-HSCT. The median time to relapse after allo-HSCT was 5.9 months (range, 4.1-11).
Patients underwent lymphodepletion with fludarabine, cyclophosphamide, and alemtuzumab (n=8) or fludarabine and cyclophosphamide (n=1).
They received UCART19 at 2 dose levels—6 x 106 total cells (n=6) or 6 to 8 x 107 total cells (n=3).
Toxicity
There were 2 DLTs, 1 at each dose level. At the lower dose, the DLT was grade 4 cytokine release syndrome (CRS) associated with grade 5 neutropenic sepsis. The patient with this DLT died at day 15 post-infusion.
At the higher dose level, the DLT was grade 4 prolonged cytopenia associated with infection and pulmonary hemorrhage. This patient died at day 82 post-infusion, 19 days after undergoing allo-HSCT.
In all, 8 patients had CRS. One patient had grade 1, 6 had grade 2, and 1 had grade 4 CRS. Seven patients had complete resolution of CRS.
Two patients had neurotoxic events, both grade 1.
Three patients had prolonged cytopenia, all grade 4. This was defined as persistent neutropenia and/or thrombocytopenia beyond day 42 after UCART19 infusion, except if the patient had >5% bone marrow blasts.
Two patients had neutropenic sepsis, grade 4 and grade 5.
Three patients had cytomegalovirus infection, all grade 2. Two patients had adenovirus infection, grade 1 and grade 3.
One patient had grade 1 acute cutaneous graft-versus-host disease.
Efficacy
Six of 9 patients achieved a CR, and 5 were MRD-negative by flow cytometry or qPCR. All 5 patients proceeded to allo-HSCT.
After transplant, 2 patients were still MRD-negative at 8 months, 1 patient relapsed after 6 months, and 2 patients died while MRD-negative. The deaths were due to infection and pulmonary hemorrhage.
Two other deaths were due to a DLT (neutropenic sepsis associated with CRS) and progressive disease.
Of the 5 patients who are alive, 2 are still MRD-negative after allo-HSCT. One of these patients relapsed after the first UCART19 infusion, received a second infusion, and achieved an MRD-negative CR that enabled transplant.
The remaining 3 patients who are still alive did not undergo transplant. One of these patients relapsed after achieving MRD-positive remission, and 1 relapsed after MRD-negative remission. The third patient relapsed and received a second UCART19 infusion. This patient has yet to respond to the second infusion but has minimal follow-up.
Recruitment in this study is ongoing at the higher dose level—6 to 8 x 107 total cells.
*Data in the abstract were updated in the presentation.
LISBON—Preliminary data from a phase 1 trial suggest UCART19 can serve as a bridge to transplant in adults with relapsed/refractory (R/R) B-cell acute lymphoblastic leukemia (B-ALL).
This “universal,” donor-derived, chimeric antigen receptor (CAR) T-cell therapy produced complete responses (CRs) in 6 of 9 patients treated.
Five of the patients who were negative for minimal residual disease (MRD) proceeded to allogeneic hematopoietic stem cell transplant (allo-HSCT).
Two of the 5 patients were still alive and in MRD-negative remission at last follow-up, but 1 had relapsed and 2 died.
A total of 4 patients died in this trial—1 from a dose-limiting toxicity (DLT), 1 from progressive disease, 1 from infection, and 1 from pulmonary hemorrhage.
“These early results for UCART19 are very encouraging, both in terms of manageable safety and the impressive complete molecular remission rate in these hard-to-treat adult patients with R/R B-ALL,” said principal investigator Reuben Benjamin, MBBS, PhD, a consultant hematologist at King’s College London in the UK.
Dr Benjamin presented these results, from the CALM trial, at the 44th Annual Meeting of the EBMT (abstract OS10-4*).
The CALM trial is sponsored by Servier. In 2015, Servier acquired exclusive rights from Cellectis for UCART19, which is being co-developed by Servier and Pfizer.
Patients and treatment
Dr Benjamin presented results in 9 patients with R/R B-ALL who had a median age of 23 (range, 18-49).
All patients had morphological disease or an MRD level of at least 1 x 10-3 (via flow cytometry and/or qPCR) at baseline. The median disease burden was 8% (range, 0-95).
Four patients had received 1 to 3 prior therapies, and 5 had received 4 or more prior treatments. Six patients had prior inotuzumab ozogamicin, 2 had prior blinatumomab, and 7 had prior allo-HSCT. The median time to relapse after allo-HSCT was 5.9 months (range, 4.1-11).
Patients underwent lymphodepletion with fludarabine, cyclophosphamide, and alemtuzumab (n=8) or fludarabine and cyclophosphamide (n=1).
They received UCART19 at 2 dose levels—6 x 106 total cells (n=6) or 6 to 8 x 107 total cells (n=3).
Toxicity
There were 2 DLTs, 1 at each dose level. At the lower dose, the DLT was grade 4 cytokine release syndrome (CRS) associated with grade 5 neutropenic sepsis. The patient with this DLT died at day 15 post-infusion.
At the higher dose level, the DLT was grade 4 prolonged cytopenia associated with infection and pulmonary hemorrhage. This patient died at day 82 post-infusion, 19 days after undergoing allo-HSCT.
In all, 8 patients had CRS. One patient had grade 1, 6 had grade 2, and 1 had grade 4 CRS. Seven patients had complete resolution of CRS.
Two patients had neurotoxic events, both grade 1.
Three patients had prolonged cytopenia, all grade 4. This was defined as persistent neutropenia and/or thrombocytopenia beyond day 42 after UCART19 infusion, except if the patient had >5% bone marrow blasts.
Two patients had neutropenic sepsis, grade 4 and grade 5.
Three patients had cytomegalovirus infection, all grade 2. Two patients had adenovirus infection, grade 1 and grade 3.
One patient had grade 1 acute cutaneous graft-versus-host disease.
Efficacy
Six of 9 patients achieved a CR, and 5 were MRD-negative by flow cytometry or qPCR. All 5 patients proceeded to allo-HSCT.
After transplant, 2 patients were still MRD-negative at 8 months, 1 patient relapsed after 6 months, and 2 patients died while MRD-negative. The deaths were due to infection and pulmonary hemorrhage.
Two other deaths were due to a DLT (neutropenic sepsis associated with CRS) and progressive disease.
Of the 5 patients who are alive, 2 are still MRD-negative after allo-HSCT. One of these patients relapsed after the first UCART19 infusion, received a second infusion, and achieved an MRD-negative CR that enabled transplant.
The remaining 3 patients who are still alive did not undergo transplant. One of these patients relapsed after achieving MRD-positive remission, and 1 relapsed after MRD-negative remission. The third patient relapsed and received a second UCART19 infusion. This patient has yet to respond to the second infusion but has minimal follow-up.
Recruitment in this study is ongoing at the higher dose level—6 to 8 x 107 total cells.
*Data in the abstract were updated in the presentation.
LISBON—Preliminary data from a phase 1 trial suggest UCART19 can serve as a bridge to transplant in adults with relapsed/refractory (R/R) B-cell acute lymphoblastic leukemia (B-ALL).
This “universal,” donor-derived, chimeric antigen receptor (CAR) T-cell therapy produced complete responses (CRs) in 6 of 9 patients treated.
Five of the patients who were negative for minimal residual disease (MRD) proceeded to allogeneic hematopoietic stem cell transplant (allo-HSCT).
Two of the 5 patients were still alive and in MRD-negative remission at last follow-up, but 1 had relapsed and 2 died.
A total of 4 patients died in this trial—1 from a dose-limiting toxicity (DLT), 1 from progressive disease, 1 from infection, and 1 from pulmonary hemorrhage.
“These early results for UCART19 are very encouraging, both in terms of manageable safety and the impressive complete molecular remission rate in these hard-to-treat adult patients with R/R B-ALL,” said principal investigator Reuben Benjamin, MBBS, PhD, a consultant hematologist at King’s College London in the UK.
Dr Benjamin presented these results, from the CALM trial, at the 44th Annual Meeting of the EBMT (abstract OS10-4*).
The CALM trial is sponsored by Servier. In 2015, Servier acquired exclusive rights from Cellectis for UCART19, which is being co-developed by Servier and Pfizer.
Patients and treatment
Dr Benjamin presented results in 9 patients with R/R B-ALL who had a median age of 23 (range, 18-49).
All patients had morphological disease or an MRD level of at least 1 x 10-3 (via flow cytometry and/or qPCR) at baseline. The median disease burden was 8% (range, 0-95).
Four patients had received 1 to 3 prior therapies, and 5 had received 4 or more prior treatments. Six patients had prior inotuzumab ozogamicin, 2 had prior blinatumomab, and 7 had prior allo-HSCT. The median time to relapse after allo-HSCT was 5.9 months (range, 4.1-11).
Patients underwent lymphodepletion with fludarabine, cyclophosphamide, and alemtuzumab (n=8) or fludarabine and cyclophosphamide (n=1).
They received UCART19 at 2 dose levels—6 x 106 total cells (n=6) or 6 to 8 x 107 total cells (n=3).
Toxicity
There were 2 DLTs, 1 at each dose level. At the lower dose, the DLT was grade 4 cytokine release syndrome (CRS) associated with grade 5 neutropenic sepsis. The patient with this DLT died at day 15 post-infusion.
At the higher dose level, the DLT was grade 4 prolonged cytopenia associated with infection and pulmonary hemorrhage. This patient died at day 82 post-infusion, 19 days after undergoing allo-HSCT.
In all, 8 patients had CRS. One patient had grade 1, 6 had grade 2, and 1 had grade 4 CRS. Seven patients had complete resolution of CRS.
Two patients had neurotoxic events, both grade 1.
Three patients had prolonged cytopenia, all grade 4. This was defined as persistent neutropenia and/or thrombocytopenia beyond day 42 after UCART19 infusion, except if the patient had >5% bone marrow blasts.
Two patients had neutropenic sepsis, grade 4 and grade 5.
Three patients had cytomegalovirus infection, all grade 2. Two patients had adenovirus infection, grade 1 and grade 3.
One patient had grade 1 acute cutaneous graft-versus-host disease.
Efficacy
Six of 9 patients achieved a CR, and 5 were MRD-negative by flow cytometry or qPCR. All 5 patients proceeded to allo-HSCT.
After transplant, 2 patients were still MRD-negative at 8 months, 1 patient relapsed after 6 months, and 2 patients died while MRD-negative. The deaths were due to infection and pulmonary hemorrhage.
Two other deaths were due to a DLT (neutropenic sepsis associated with CRS) and progressive disease.
Of the 5 patients who are alive, 2 are still MRD-negative after allo-HSCT. One of these patients relapsed after the first UCART19 infusion, received a second infusion, and achieved an MRD-negative CR that enabled transplant.
The remaining 3 patients who are still alive did not undergo transplant. One of these patients relapsed after achieving MRD-positive remission, and 1 relapsed after MRD-negative remission. The third patient relapsed and received a second UCART19 infusion. This patient has yet to respond to the second infusion but has minimal follow-up.
Recruitment in this study is ongoing at the higher dose level—6 to 8 x 107 total cells.
*Data in the abstract were updated in the presentation.
CHMP supports expanded approval for fosaprepitant
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended changing the terms of marketing authorization for fosaprepitant (Ivemend).
The product is already approved in the European Union (EU) for the prevention of acute and delayed nausea and vomiting associated with moderately or highly emetogenic cancer chemotherapy in adults.
Now, the CHMP is recommending that fosaprepitant be authorized for the same indication in pediatric patients age 6 months and older.
As it is in adults, fosaprepitant would be given to children as part of combination therapy.
The CHMP’s opinion on fosaprepitant will be reviewed by the European Commission (EC).
If the EC agrees with the CHMP, the commission will grant a centralized marketing authorization that will be valid in the EU. Norway, Iceland, and Liechtenstein will make corresponding decisions on the basis of the EC’s decision.
The EC typically makes a decision within 67 days of the CHMP’s recommendation.
Merck Sharp & Dohme Corp., the company developing fosaprepitant, has conducted a phase 2 trial assessing the pharmacokinetics, pharmacodynamics, safety, and tolerability of fosaprepitant for the prevention of chemotherapy-induced nausea and vomiting in children.
Patients ages 2 to 17 were randomized to receive 1 of 4 doses of fosaprepitant (0.4 mg/kg, 1.2 mg/kg, 3 mg/kg, and 5 mg/kg) or placebo in cycle 1. All patients also received ondansetron, with or without dexamethasone. Patients ages 0 to 11 were invited to participate in optional cycles 2 to 6, during which they received fosaprepitant at 3 mg/kg or 5 mg/kg.
Results from this trial have been posted on its clinicaltrials.gov page (NCT01697579).
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended changing the terms of marketing authorization for fosaprepitant (Ivemend).
The product is already approved in the European Union (EU) for the prevention of acute and delayed nausea and vomiting associated with moderately or highly emetogenic cancer chemotherapy in adults.
Now, the CHMP is recommending that fosaprepitant be authorized for the same indication in pediatric patients age 6 months and older.
As it is in adults, fosaprepitant would be given to children as part of combination therapy.
The CHMP’s opinion on fosaprepitant will be reviewed by the European Commission (EC).
If the EC agrees with the CHMP, the commission will grant a centralized marketing authorization that will be valid in the EU. Norway, Iceland, and Liechtenstein will make corresponding decisions on the basis of the EC’s decision.
The EC typically makes a decision within 67 days of the CHMP’s recommendation.
Merck Sharp & Dohme Corp., the company developing fosaprepitant, has conducted a phase 2 trial assessing the pharmacokinetics, pharmacodynamics, safety, and tolerability of fosaprepitant for the prevention of chemotherapy-induced nausea and vomiting in children.
Patients ages 2 to 17 were randomized to receive 1 of 4 doses of fosaprepitant (0.4 mg/kg, 1.2 mg/kg, 3 mg/kg, and 5 mg/kg) or placebo in cycle 1. All patients also received ondansetron, with or without dexamethasone. Patients ages 0 to 11 were invited to participate in optional cycles 2 to 6, during which they received fosaprepitant at 3 mg/kg or 5 mg/kg.
Results from this trial have been posted on its clinicaltrials.gov page (NCT01697579).
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended changing the terms of marketing authorization for fosaprepitant (Ivemend).
The product is already approved in the European Union (EU) for the prevention of acute and delayed nausea and vomiting associated with moderately or highly emetogenic cancer chemotherapy in adults.
Now, the CHMP is recommending that fosaprepitant be authorized for the same indication in pediatric patients age 6 months and older.
As it is in adults, fosaprepitant would be given to children as part of combination therapy.
The CHMP’s opinion on fosaprepitant will be reviewed by the European Commission (EC).
If the EC agrees with the CHMP, the commission will grant a centralized marketing authorization that will be valid in the EU. Norway, Iceland, and Liechtenstein will make corresponding decisions on the basis of the EC’s decision.
The EC typically makes a decision within 67 days of the CHMP’s recommendation.
Merck Sharp & Dohme Corp., the company developing fosaprepitant, has conducted a phase 2 trial assessing the pharmacokinetics, pharmacodynamics, safety, and tolerability of fosaprepitant for the prevention of chemotherapy-induced nausea and vomiting in children.
Patients ages 2 to 17 were randomized to receive 1 of 4 doses of fosaprepitant (0.4 mg/kg, 1.2 mg/kg, 3 mg/kg, and 5 mg/kg) or placebo in cycle 1. All patients also received ondansetron, with or without dexamethasone. Patients ages 0 to 11 were invited to participate in optional cycles 2 to 6, during which they received fosaprepitant at 3 mg/kg or 5 mg/kg.
Results from this trial have been posted on its clinicaltrials.gov page (NCT01697579).
Phase 1 results with UCART19 in kids
LISBON—Early results from a phase 1, pediatric trial of UCART19 expand upon results observed in children who received the therapy via a compassionate use program.
Two patients with relapsed/refractory B-cell acute lymphoblastic leukemia (B-ALL) received UCART19, a “universal,” donor-derived chimeric antigen receptor (CAR) T-cell therapy, via the program.
Both achieved remission and were still alive at last follow-up, more than 2 years after proceeding to transplant.
In the phase 1 trial, 5 of 6 B-ALL patients have achieved remission and gone on to transplant.
Three of the patients are still alive, and 2 are still negative for minimal residual disease (MRD) at 10 months and 11 months after UCART19 infusion.
However, 2 patients died of progression, and 1 died of transplant-related complications.
Paul Veys, MBBS, of Great Ormond Street Hospital (GOSH) in London, UK, presented these results, from the PALL trial, at the 44th Annual Meeting of the EBMT (abstract OS18-5*).
The trial is sponsored by Servier. In 2015, Servier acquired exclusive rights from Cellectis for UCART19, which is being co-developed by Servier and Pfizer.
Prior experience
Researchers previously reported results with UCART19 in 2 infants with relapsed/refractory B-ALL who had exhausted all other treatment options and received UCART19 via a compassionate use program.
Both patients achieved remission after UCART19 and proceeded to allogeneic hematopoietic stem cell transplant (allo-HSCT).
When these results were published, in January 2017, both patients were still alive and leukemia-free at last follow-up—12 months and 18 months after UCART19 infusion.
Dr Veys provided an update, noting that these patients were still alive and in remission at 24 months and 30 months after allo-HSCT.
Phase 1 patients and treatment
Thus far, the phase 1 trial has enrolled and treated 6 patients with relapsed B-ALL. They had a median age of 3.75 (range, 0.8-16.4).
All patients had morphological disease or an MRD level of at least 1 x 10-3 (via flow cytometry and/or qPCR) at baseline.
One patient had received 1 prior therapy, 2 had 3 prior therapies, and 3 had 4 or more prior therapies. Two patients had prior inotuzumab ozogamicin, and 2 had prior allo-HSCT. Both had relapsed within 6 months of allo-HSCT.
Five patients had less than 10% bone marrow blasts prior to lymphodepletion, and 1 had greater than 50% blasts.
Patients underwent lymphodepletion with fludarabine, cyclophosphamide, and alemtuzumab (n=5) or fludarabine and cyclophosphamide (n=1).
The patients received UCART19 at doses of 2 x 107 total cells or 1.1 to 2.3 x 106 cells/kg.
Toxicity
All 6 patients developed cytokine release syndrome (CRS), including grade 1 (n=1), grade 2 (n=4), and grade 3 (n-1) CRS. However, all 6 cases resolved completely.
Three patients had neurotoxic events, 2 grade 1 and 1 grade 2. One patient had grade 3 febrile neutropenia.
Three patients had grade 4 prolonged cytopenia. This was defined as persistent neutropenia and/or thrombocytopenia beyond day 42 after UCART19 infusion, except if the patient had >5% bone marrow blasts.
One patient had grade 1 adenovirus infection, 1 had grade 3 cytomegalovirus infection, 2 had grade 3 BK virus hemorrhagic cystitis, and 1 had grade 4 metapneumovirus infection.
One patient had grade 1 acute cutaneous graft-versus-host disease.
Efficacy
All 6 patients achieved a complete response at day 28 to 42 after UCART19 infusion. Five patients achieved MRD negativity according to flow cytometry, and 3 were MRD-negative according to PCR.
The 5 flow-MRD-negative patients went on to receive an allo-HSCT between 49 days and 62 days after UCART19 infusion. Conditioning consisted of total body irradiation and fludarabine, with or without cyclophosphamide and antithymocyte globulin. All of these patients received a dose of rituximab as well, which was intended to target any remaining UCART19 cells.
Two patients relapsed 3 months after transplant and died at 7 months and 8 months after UCART19 infusion. One of these patients was CD19-, and 1 was CD19+, but both were MRD-positive by PCR prior to receiving their transplant.
A third patient died 2.5 months after allo-HSCT from transplant-related complications, including thrombotic microangiopathy, BK hemorrhagic cystitis, and nephritis.
The remaining 3 patients are still alive at 1.5 months, 10 months, and 11 months after UCART19 infusion. Two are still MRD-negative, and 1 is MRD-positive. The MRD-positive patient has not undergone allo-HSCT.
*Data in the abstract were updated in the presentation.
LISBON—Early results from a phase 1, pediatric trial of UCART19 expand upon results observed in children who received the therapy via a compassionate use program.
Two patients with relapsed/refractory B-cell acute lymphoblastic leukemia (B-ALL) received UCART19, a “universal,” donor-derived chimeric antigen receptor (CAR) T-cell therapy, via the program.
Both achieved remission and were still alive at last follow-up, more than 2 years after proceeding to transplant.
In the phase 1 trial, 5 of 6 B-ALL patients have achieved remission and gone on to transplant.
Three of the patients are still alive, and 2 are still negative for minimal residual disease (MRD) at 10 months and 11 months after UCART19 infusion.
However, 2 patients died of progression, and 1 died of transplant-related complications.
Paul Veys, MBBS, of Great Ormond Street Hospital (GOSH) in London, UK, presented these results, from the PALL trial, at the 44th Annual Meeting of the EBMT (abstract OS18-5*).
The trial is sponsored by Servier. In 2015, Servier acquired exclusive rights from Cellectis for UCART19, which is being co-developed by Servier and Pfizer.
Prior experience
Researchers previously reported results with UCART19 in 2 infants with relapsed/refractory B-ALL who had exhausted all other treatment options and received UCART19 via a compassionate use program.
Both patients achieved remission after UCART19 and proceeded to allogeneic hematopoietic stem cell transplant (allo-HSCT).
When these results were published, in January 2017, both patients were still alive and leukemia-free at last follow-up—12 months and 18 months after UCART19 infusion.
Dr Veys provided an update, noting that these patients were still alive and in remission at 24 months and 30 months after allo-HSCT.
Phase 1 patients and treatment
Thus far, the phase 1 trial has enrolled and treated 6 patients with relapsed B-ALL. They had a median age of 3.75 (range, 0.8-16.4).
All patients had morphological disease or an MRD level of at least 1 x 10-3 (via flow cytometry and/or qPCR) at baseline.
One patient had received 1 prior therapy, 2 had 3 prior therapies, and 3 had 4 or more prior therapies. Two patients had prior inotuzumab ozogamicin, and 2 had prior allo-HSCT. Both had relapsed within 6 months of allo-HSCT.
Five patients had less than 10% bone marrow blasts prior to lymphodepletion, and 1 had greater than 50% blasts.
Patients underwent lymphodepletion with fludarabine, cyclophosphamide, and alemtuzumab (n=5) or fludarabine and cyclophosphamide (n=1).
The patients received UCART19 at doses of 2 x 107 total cells or 1.1 to 2.3 x 106 cells/kg.
Toxicity
All 6 patients developed cytokine release syndrome (CRS), including grade 1 (n=1), grade 2 (n=4), and grade 3 (n-1) CRS. However, all 6 cases resolved completely.
Three patients had neurotoxic events, 2 grade 1 and 1 grade 2. One patient had grade 3 febrile neutropenia.
Three patients had grade 4 prolonged cytopenia. This was defined as persistent neutropenia and/or thrombocytopenia beyond day 42 after UCART19 infusion, except if the patient had >5% bone marrow blasts.
One patient had grade 1 adenovirus infection, 1 had grade 3 cytomegalovirus infection, 2 had grade 3 BK virus hemorrhagic cystitis, and 1 had grade 4 metapneumovirus infection.
One patient had grade 1 acute cutaneous graft-versus-host disease.
Efficacy
All 6 patients achieved a complete response at day 28 to 42 after UCART19 infusion. Five patients achieved MRD negativity according to flow cytometry, and 3 were MRD-negative according to PCR.
The 5 flow-MRD-negative patients went on to receive an allo-HSCT between 49 days and 62 days after UCART19 infusion. Conditioning consisted of total body irradiation and fludarabine, with or without cyclophosphamide and antithymocyte globulin. All of these patients received a dose of rituximab as well, which was intended to target any remaining UCART19 cells.
Two patients relapsed 3 months after transplant and died at 7 months and 8 months after UCART19 infusion. One of these patients was CD19-, and 1 was CD19+, but both were MRD-positive by PCR prior to receiving their transplant.
A third patient died 2.5 months after allo-HSCT from transplant-related complications, including thrombotic microangiopathy, BK hemorrhagic cystitis, and nephritis.
The remaining 3 patients are still alive at 1.5 months, 10 months, and 11 months after UCART19 infusion. Two are still MRD-negative, and 1 is MRD-positive. The MRD-positive patient has not undergone allo-HSCT.
*Data in the abstract were updated in the presentation.
LISBON—Early results from a phase 1, pediatric trial of UCART19 expand upon results observed in children who received the therapy via a compassionate use program.
Two patients with relapsed/refractory B-cell acute lymphoblastic leukemia (B-ALL) received UCART19, a “universal,” donor-derived chimeric antigen receptor (CAR) T-cell therapy, via the program.
Both achieved remission and were still alive at last follow-up, more than 2 years after proceeding to transplant.
In the phase 1 trial, 5 of 6 B-ALL patients have achieved remission and gone on to transplant.
Three of the patients are still alive, and 2 are still negative for minimal residual disease (MRD) at 10 months and 11 months after UCART19 infusion.
However, 2 patients died of progression, and 1 died of transplant-related complications.
Paul Veys, MBBS, of Great Ormond Street Hospital (GOSH) in London, UK, presented these results, from the PALL trial, at the 44th Annual Meeting of the EBMT (abstract OS18-5*).
The trial is sponsored by Servier. In 2015, Servier acquired exclusive rights from Cellectis for UCART19, which is being co-developed by Servier and Pfizer.
Prior experience
Researchers previously reported results with UCART19 in 2 infants with relapsed/refractory B-ALL who had exhausted all other treatment options and received UCART19 via a compassionate use program.
Both patients achieved remission after UCART19 and proceeded to allogeneic hematopoietic stem cell transplant (allo-HSCT).
When these results were published, in January 2017, both patients were still alive and leukemia-free at last follow-up—12 months and 18 months after UCART19 infusion.
Dr Veys provided an update, noting that these patients were still alive and in remission at 24 months and 30 months after allo-HSCT.
Phase 1 patients and treatment
Thus far, the phase 1 trial has enrolled and treated 6 patients with relapsed B-ALL. They had a median age of 3.75 (range, 0.8-16.4).
All patients had morphological disease or an MRD level of at least 1 x 10-3 (via flow cytometry and/or qPCR) at baseline.
One patient had received 1 prior therapy, 2 had 3 prior therapies, and 3 had 4 or more prior therapies. Two patients had prior inotuzumab ozogamicin, and 2 had prior allo-HSCT. Both had relapsed within 6 months of allo-HSCT.
Five patients had less than 10% bone marrow blasts prior to lymphodepletion, and 1 had greater than 50% blasts.
Patients underwent lymphodepletion with fludarabine, cyclophosphamide, and alemtuzumab (n=5) or fludarabine and cyclophosphamide (n=1).
The patients received UCART19 at doses of 2 x 107 total cells or 1.1 to 2.3 x 106 cells/kg.
Toxicity
All 6 patients developed cytokine release syndrome (CRS), including grade 1 (n=1), grade 2 (n=4), and grade 3 (n-1) CRS. However, all 6 cases resolved completely.
Three patients had neurotoxic events, 2 grade 1 and 1 grade 2. One patient had grade 3 febrile neutropenia.
Three patients had grade 4 prolonged cytopenia. This was defined as persistent neutropenia and/or thrombocytopenia beyond day 42 after UCART19 infusion, except if the patient had >5% bone marrow blasts.
One patient had grade 1 adenovirus infection, 1 had grade 3 cytomegalovirus infection, 2 had grade 3 BK virus hemorrhagic cystitis, and 1 had grade 4 metapneumovirus infection.
One patient had grade 1 acute cutaneous graft-versus-host disease.
Efficacy
All 6 patients achieved a complete response at day 28 to 42 after UCART19 infusion. Five patients achieved MRD negativity according to flow cytometry, and 3 were MRD-negative according to PCR.
The 5 flow-MRD-negative patients went on to receive an allo-HSCT between 49 days and 62 days after UCART19 infusion. Conditioning consisted of total body irradiation and fludarabine, with or without cyclophosphamide and antithymocyte globulin. All of these patients received a dose of rituximab as well, which was intended to target any remaining UCART19 cells.
Two patients relapsed 3 months after transplant and died at 7 months and 8 months after UCART19 infusion. One of these patients was CD19-, and 1 was CD19+, but both were MRD-positive by PCR prior to receiving their transplant.
A third patient died 2.5 months after allo-HSCT from transplant-related complications, including thrombotic microangiopathy, BK hemorrhagic cystitis, and nephritis.
The remaining 3 patients are still alive at 1.5 months, 10 months, and 11 months after UCART19 infusion. Two are still MRD-negative, and 1 is MRD-positive. The MRD-positive patient has not undergone allo-HSCT.
*Data in the abstract were updated in the presentation.
ICER assesses value of CAR T-cell therapies
The Institute for Clinical and Economic Review (ICER) has made policy recommendations intended to ensure affordability and access to chimeric antigen receptor (CAR) T-cell therapies.
ICER released a Final Evidence Report on tisagenlecleucel (Kymriah, Novartis) and axicabtagene ciloleucel (Yescarta, Kite Pharma/Gilead), 2 CAR T-cell therapies approved in the US to treat B-cell acute lymphoblastic leukemia (B-ALL) and non-Hodgkin lymphoma (NHL), respectively.
The report says the pricing of these therapies aligns with patient benefit, but changes will be needed in future pricing, payment, and delivery mechanisms to ensure patient access without threatening health system affordability.
“Given the currently available evidence, these therapies appear to be effective options for those with B-ALL or NHL, though uncertainty in the evidence raised questions around the long-term value for money,” said Dan Ollendorf, PhD, ICER’s chief scientific officer.
Net health benefit
ICER’s report says tisagenlecleucel provides a net health benefit for children with B-ALL, and both tisagenlecleucel and axicabtagene ciloleucel provide a net health benefit for adults with certain types of NHL. (Novartis is seeking approval for tisagenlecleucel in NHL).
The evidence suggests there is at least a small net health benefit of the CAR T-cell therapies compared to other therapies. The benefit may be substantial, but uncertainties remain.
The data show complete remission (CR), disease-free survival (DFS), and overall survival (OS) rates are superior for NHL patients who receive axicabtagene ciloleucel, compared to patients who receive standard chemoimmunotherapy regimens.
Similarly, B-ALL patients treated with tisagenlecleucel have superior CR, DFS, and OS rates to patients treated with standard therapies. CR and OS rates are also superior in NHL patients treated with tisagenlecleucel, but DFS has not been reported in this population.
The report says there is insufficient evidence to distinguish between the 2 CAR T-cell therapies for the treatment of NHL.
Toxicity and uncertainty
The report highlights the fact that cytokine release syndrome, neurological symptoms, and B-cell aplasia have been observed in patients who receive CAR T-cell therapies. However, these sometimes severe adverse events are generally “manageable.”
In addition to toxicity, the report highlights sources of uncertainty. These include the fact that studies of tisagenlecleucel and axicabtagene ciloleucel are small, single-arm trials with short follow-up; comparisons with historical controls may be misleading; and improvements in the CAR T-cell manufacturing process may change outcomes.
Cost-effectiveness
The report states that the cost-effectiveness of each therapy fell below or within commonly cited thresholds of $50,000 to $150,000 per quality-adjusted life-year (QALY) over a lifetime.
For its analyses, ICER used the wholesale acquisition cost (WAC) plus an assumed hospital mark-up. The analyses were also based on the assumption that survival benefits observed in clinical trials would continue after the trials ended.
For tisagenlecleucel in pediatric B-ALL, the WAC is $475,000. The long-term cost-effectiveness compared to clofarabine is $45,871 per QALY gained.
For axicabtagene ciloleucel in adults with NHL, the WAC is $373,000. The long-term cost-effectiveness compared to salvage chemotherapy is $136,078 per QALY gained. The effectiveness assumptions for chemotherapy were based on an average of salvage chemotherapy regimens from the SCHOLAR-1 trial, and the cost assumptions were based on the cost of the R-DHAP (rituximab, dexamethasone, cytarabine, and cisplatin) regimen.
The report says tisagenlecleucel’s price would remain in alignment with value even if price premiums of 102% to 194% were applied.
Meanwhile, axicabtagene ciloleucel’s price could be increased by up to 11% and remain in alignment with the upper threshold ($150,000 per QALY gained) but would need to be discounted by 28% to align with the lower threshold ($100,000 per QALY gained).
Tisagenlecleucel, as a treatment for B-ALL, is not expected to cross the $915 million threshold for annual budget impact.
However, the short-term costs of axicabtagene ciloleucel for relapsed/refractory NHL could exceed the threshold. Only 38% of the estimated 5900 eligible patients could receive axicabtagene ciloleucel in a year before crossing the threshold.
Because of these findings, ICER issued an “Affordability and Access Alert” for axicabtagene ciloleucel.
This alert is intended to signal when the added costs associated with a new treatment may be difficult for the healthcare system to absorb over the short-term without displacing other needed services or contributing to unsustainable growth in healthcare insurance costs.
“Based on current evidence, both therapies appear to be priced in alignment with their clinical value, but there are potential short-term affordability concerns—for axicabtagene ciloleucel under its current indication and for both treatments should they receive future approvals for broader patient populations,” Dr Ollendorf said.
Panel voting results
ICER’s report was reviewed at a public meeting of the California Technology Assessment Forum on March 2.
Most of the panel said tisagenlecleucel provides intermediate long-term value for money when treating B-ALL. However, the significant uncertainty surrounding the long-term risks and benefits of the therapy precluded a high-value vote.
After deliberating on the value of axicabtagene ciloleucel to treat NHL, the panel’s votes were split between low-value and intermediate-value, driven by similar concerns about long-term uncertainty.
Policy recommendations
Following the voting session, ICER convened a policy roundtable of experts, including physicians, patient advocates, manufacturer representatives, and payer representatives.
Based on the roundtable discussion, ICER developed recommendations for enhanced stakeholder communication, innovative payment models, generation of additional evidence, settings of care, and patient education.
“With many other potentially transformative therapies in the pipeline, stakeholders must collaborate now to develop payment and delivery systems that can ensure timely patient access, manage short-term affordability for expensive one-time treatments, and continue to reward the innovation that brings these new treatments to market,” Dr Ollendorf said.
Some of ICER’s recommendations include:
- When launching novel therapies approved with limited clinical evidence, such as CAR T-cell therapies, manufacturers and payers should consider using a lower launch price that could be increased if substantial clinical benefits are confirmed or using a higher initial price tied to a requirement for refunds or rebates if real-world evidence fails to confirm high expectations.
- Outcomes-based pricing arrangements must be linked to “meaningful clinical outcomes assessed with sufficient follow up.”
- Hospital mark-up for CAR T-cell therapies “should reflect the expected additional cost for care delivered in the hospital, rather than a percentage of the drug cost to avoid perverse incentives in choosing the treatment location.”
- Initially, CAR T-cell therapies should be delivered in “manufacturer-accredited centers to ensure the quality and appropriateness of care.” Later, “centers of excellence accredited by specialty societies” can administer these therapies, as long as providers have “sufficient expertise” to manage serious side effects.
- Centers should ensure that patients understand what to expect from CAR T-cell therapy, including long-term consequences.
- Because additional evidence on CAR T-cell therapies is needed, all patients who receive these therapies should enter into a registry with planned long-term follow-up.
- Studies should determine the optimal timing of CAR T-cell therapy in the sequence of treatments for B-ALL and NHL.
Additional recommendations and more details are available in ICER’s report.
About ICER
ICER is an independent, non-profit research institute that produces reports analyzing evidence on the effectiveness and value of drugs and other medical services.
ICER’s reports include evidence-based calculations of prices for new drugs that reflect the degree of improvement expected in long-term patient outcomes, while also highlighting price levels that might contribute to unaffordable short-term cost growth for the overall healthcare system.
ICER’s reports incorporate input from stakeholders and are the subject of public hearings through 3 core programs: the California Technology Assessment Forum, the Midwest Comparative Effectiveness Public Advisory Council, and the New England Comparative Effectiveness Public Advisory Council.
These independent panels review ICER’s reports at public meetings to deliberate on the evidence and develop recommendations for how patients, clinicians, insurers, and policymakers can improve the quality and value of healthcare.
The Institute for Clinical and Economic Review (ICER) has made policy recommendations intended to ensure affordability and access to chimeric antigen receptor (CAR) T-cell therapies.
ICER released a Final Evidence Report on tisagenlecleucel (Kymriah, Novartis) and axicabtagene ciloleucel (Yescarta, Kite Pharma/Gilead), 2 CAR T-cell therapies approved in the US to treat B-cell acute lymphoblastic leukemia (B-ALL) and non-Hodgkin lymphoma (NHL), respectively.
The report says the pricing of these therapies aligns with patient benefit, but changes will be needed in future pricing, payment, and delivery mechanisms to ensure patient access without threatening health system affordability.
“Given the currently available evidence, these therapies appear to be effective options for those with B-ALL or NHL, though uncertainty in the evidence raised questions around the long-term value for money,” said Dan Ollendorf, PhD, ICER’s chief scientific officer.
Net health benefit
ICER’s report says tisagenlecleucel provides a net health benefit for children with B-ALL, and both tisagenlecleucel and axicabtagene ciloleucel provide a net health benefit for adults with certain types of NHL. (Novartis is seeking approval for tisagenlecleucel in NHL).
The evidence suggests there is at least a small net health benefit of the CAR T-cell therapies compared to other therapies. The benefit may be substantial, but uncertainties remain.
The data show complete remission (CR), disease-free survival (DFS), and overall survival (OS) rates are superior for NHL patients who receive axicabtagene ciloleucel, compared to patients who receive standard chemoimmunotherapy regimens.
Similarly, B-ALL patients treated with tisagenlecleucel have superior CR, DFS, and OS rates to patients treated with standard therapies. CR and OS rates are also superior in NHL patients treated with tisagenlecleucel, but DFS has not been reported in this population.
The report says there is insufficient evidence to distinguish between the 2 CAR T-cell therapies for the treatment of NHL.
Toxicity and uncertainty
The report highlights the fact that cytokine release syndrome, neurological symptoms, and B-cell aplasia have been observed in patients who receive CAR T-cell therapies. However, these sometimes severe adverse events are generally “manageable.”
In addition to toxicity, the report highlights sources of uncertainty. These include the fact that studies of tisagenlecleucel and axicabtagene ciloleucel are small, single-arm trials with short follow-up; comparisons with historical controls may be misleading; and improvements in the CAR T-cell manufacturing process may change outcomes.
Cost-effectiveness
The report states that the cost-effectiveness of each therapy fell below or within commonly cited thresholds of $50,000 to $150,000 per quality-adjusted life-year (QALY) over a lifetime.
For its analyses, ICER used the wholesale acquisition cost (WAC) plus an assumed hospital mark-up. The analyses were also based on the assumption that survival benefits observed in clinical trials would continue after the trials ended.
For tisagenlecleucel in pediatric B-ALL, the WAC is $475,000. The long-term cost-effectiveness compared to clofarabine is $45,871 per QALY gained.
For axicabtagene ciloleucel in adults with NHL, the WAC is $373,000. The long-term cost-effectiveness compared to salvage chemotherapy is $136,078 per QALY gained. The effectiveness assumptions for chemotherapy were based on an average of salvage chemotherapy regimens from the SCHOLAR-1 trial, and the cost assumptions were based on the cost of the R-DHAP (rituximab, dexamethasone, cytarabine, and cisplatin) regimen.
The report says tisagenlecleucel’s price would remain in alignment with value even if price premiums of 102% to 194% were applied.
Meanwhile, axicabtagene ciloleucel’s price could be increased by up to 11% and remain in alignment with the upper threshold ($150,000 per QALY gained) but would need to be discounted by 28% to align with the lower threshold ($100,000 per QALY gained).
Tisagenlecleucel, as a treatment for B-ALL, is not expected to cross the $915 million threshold for annual budget impact.
However, the short-term costs of axicabtagene ciloleucel for relapsed/refractory NHL could exceed the threshold. Only 38% of the estimated 5900 eligible patients could receive axicabtagene ciloleucel in a year before crossing the threshold.
Because of these findings, ICER issued an “Affordability and Access Alert” for axicabtagene ciloleucel.
This alert is intended to signal when the added costs associated with a new treatment may be difficult for the healthcare system to absorb over the short-term without displacing other needed services or contributing to unsustainable growth in healthcare insurance costs.
“Based on current evidence, both therapies appear to be priced in alignment with their clinical value, but there are potential short-term affordability concerns—for axicabtagene ciloleucel under its current indication and for both treatments should they receive future approvals for broader patient populations,” Dr Ollendorf said.
Panel voting results
ICER’s report was reviewed at a public meeting of the California Technology Assessment Forum on March 2.
Most of the panel said tisagenlecleucel provides intermediate long-term value for money when treating B-ALL. However, the significant uncertainty surrounding the long-term risks and benefits of the therapy precluded a high-value vote.
After deliberating on the value of axicabtagene ciloleucel to treat NHL, the panel’s votes were split between low-value and intermediate-value, driven by similar concerns about long-term uncertainty.
Policy recommendations
Following the voting session, ICER convened a policy roundtable of experts, including physicians, patient advocates, manufacturer representatives, and payer representatives.
Based on the roundtable discussion, ICER developed recommendations for enhanced stakeholder communication, innovative payment models, generation of additional evidence, settings of care, and patient education.
“With many other potentially transformative therapies in the pipeline, stakeholders must collaborate now to develop payment and delivery systems that can ensure timely patient access, manage short-term affordability for expensive one-time treatments, and continue to reward the innovation that brings these new treatments to market,” Dr Ollendorf said.
Some of ICER’s recommendations include:
- When launching novel therapies approved with limited clinical evidence, such as CAR T-cell therapies, manufacturers and payers should consider using a lower launch price that could be increased if substantial clinical benefits are confirmed or using a higher initial price tied to a requirement for refunds or rebates if real-world evidence fails to confirm high expectations.
- Outcomes-based pricing arrangements must be linked to “meaningful clinical outcomes assessed with sufficient follow up.”
- Hospital mark-up for CAR T-cell therapies “should reflect the expected additional cost for care delivered in the hospital, rather than a percentage of the drug cost to avoid perverse incentives in choosing the treatment location.”
- Initially, CAR T-cell therapies should be delivered in “manufacturer-accredited centers to ensure the quality and appropriateness of care.” Later, “centers of excellence accredited by specialty societies” can administer these therapies, as long as providers have “sufficient expertise” to manage serious side effects.
- Centers should ensure that patients understand what to expect from CAR T-cell therapy, including long-term consequences.
- Because additional evidence on CAR T-cell therapies is needed, all patients who receive these therapies should enter into a registry with planned long-term follow-up.
- Studies should determine the optimal timing of CAR T-cell therapy in the sequence of treatments for B-ALL and NHL.
Additional recommendations and more details are available in ICER’s report.
About ICER
ICER is an independent, non-profit research institute that produces reports analyzing evidence on the effectiveness and value of drugs and other medical services.
ICER’s reports include evidence-based calculations of prices for new drugs that reflect the degree of improvement expected in long-term patient outcomes, while also highlighting price levels that might contribute to unaffordable short-term cost growth for the overall healthcare system.
ICER’s reports incorporate input from stakeholders and are the subject of public hearings through 3 core programs: the California Technology Assessment Forum, the Midwest Comparative Effectiveness Public Advisory Council, and the New England Comparative Effectiveness Public Advisory Council.
These independent panels review ICER’s reports at public meetings to deliberate on the evidence and develop recommendations for how patients, clinicians, insurers, and policymakers can improve the quality and value of healthcare.
The Institute for Clinical and Economic Review (ICER) has made policy recommendations intended to ensure affordability and access to chimeric antigen receptor (CAR) T-cell therapies.
ICER released a Final Evidence Report on tisagenlecleucel (Kymriah, Novartis) and axicabtagene ciloleucel (Yescarta, Kite Pharma/Gilead), 2 CAR T-cell therapies approved in the US to treat B-cell acute lymphoblastic leukemia (B-ALL) and non-Hodgkin lymphoma (NHL), respectively.
The report says the pricing of these therapies aligns with patient benefit, but changes will be needed in future pricing, payment, and delivery mechanisms to ensure patient access without threatening health system affordability.
“Given the currently available evidence, these therapies appear to be effective options for those with B-ALL or NHL, though uncertainty in the evidence raised questions around the long-term value for money,” said Dan Ollendorf, PhD, ICER’s chief scientific officer.
Net health benefit
ICER’s report says tisagenlecleucel provides a net health benefit for children with B-ALL, and both tisagenlecleucel and axicabtagene ciloleucel provide a net health benefit for adults with certain types of NHL. (Novartis is seeking approval for tisagenlecleucel in NHL).
The evidence suggests there is at least a small net health benefit of the CAR T-cell therapies compared to other therapies. The benefit may be substantial, but uncertainties remain.
The data show complete remission (CR), disease-free survival (DFS), and overall survival (OS) rates are superior for NHL patients who receive axicabtagene ciloleucel, compared to patients who receive standard chemoimmunotherapy regimens.
Similarly, B-ALL patients treated with tisagenlecleucel have superior CR, DFS, and OS rates to patients treated with standard therapies. CR and OS rates are also superior in NHL patients treated with tisagenlecleucel, but DFS has not been reported in this population.
The report says there is insufficient evidence to distinguish between the 2 CAR T-cell therapies for the treatment of NHL.
Toxicity and uncertainty
The report highlights the fact that cytokine release syndrome, neurological symptoms, and B-cell aplasia have been observed in patients who receive CAR T-cell therapies. However, these sometimes severe adverse events are generally “manageable.”
In addition to toxicity, the report highlights sources of uncertainty. These include the fact that studies of tisagenlecleucel and axicabtagene ciloleucel are small, single-arm trials with short follow-up; comparisons with historical controls may be misleading; and improvements in the CAR T-cell manufacturing process may change outcomes.
Cost-effectiveness
The report states that the cost-effectiveness of each therapy fell below or within commonly cited thresholds of $50,000 to $150,000 per quality-adjusted life-year (QALY) over a lifetime.
For its analyses, ICER used the wholesale acquisition cost (WAC) plus an assumed hospital mark-up. The analyses were also based on the assumption that survival benefits observed in clinical trials would continue after the trials ended.
For tisagenlecleucel in pediatric B-ALL, the WAC is $475,000. The long-term cost-effectiveness compared to clofarabine is $45,871 per QALY gained.
For axicabtagene ciloleucel in adults with NHL, the WAC is $373,000. The long-term cost-effectiveness compared to salvage chemotherapy is $136,078 per QALY gained. The effectiveness assumptions for chemotherapy were based on an average of salvage chemotherapy regimens from the SCHOLAR-1 trial, and the cost assumptions were based on the cost of the R-DHAP (rituximab, dexamethasone, cytarabine, and cisplatin) regimen.
The report says tisagenlecleucel’s price would remain in alignment with value even if price premiums of 102% to 194% were applied.
Meanwhile, axicabtagene ciloleucel’s price could be increased by up to 11% and remain in alignment with the upper threshold ($150,000 per QALY gained) but would need to be discounted by 28% to align with the lower threshold ($100,000 per QALY gained).
Tisagenlecleucel, as a treatment for B-ALL, is not expected to cross the $915 million threshold for annual budget impact.
However, the short-term costs of axicabtagene ciloleucel for relapsed/refractory NHL could exceed the threshold. Only 38% of the estimated 5900 eligible patients could receive axicabtagene ciloleucel in a year before crossing the threshold.
Because of these findings, ICER issued an “Affordability and Access Alert” for axicabtagene ciloleucel.
This alert is intended to signal when the added costs associated with a new treatment may be difficult for the healthcare system to absorb over the short-term without displacing other needed services or contributing to unsustainable growth in healthcare insurance costs.
“Based on current evidence, both therapies appear to be priced in alignment with their clinical value, but there are potential short-term affordability concerns—for axicabtagene ciloleucel under its current indication and for both treatments should they receive future approvals for broader patient populations,” Dr Ollendorf said.
Panel voting results
ICER’s report was reviewed at a public meeting of the California Technology Assessment Forum on March 2.
Most of the panel said tisagenlecleucel provides intermediate long-term value for money when treating B-ALL. However, the significant uncertainty surrounding the long-term risks and benefits of the therapy precluded a high-value vote.
After deliberating on the value of axicabtagene ciloleucel to treat NHL, the panel’s votes were split between low-value and intermediate-value, driven by similar concerns about long-term uncertainty.
Policy recommendations
Following the voting session, ICER convened a policy roundtable of experts, including physicians, patient advocates, manufacturer representatives, and payer representatives.
Based on the roundtable discussion, ICER developed recommendations for enhanced stakeholder communication, innovative payment models, generation of additional evidence, settings of care, and patient education.
“With many other potentially transformative therapies in the pipeline, stakeholders must collaborate now to develop payment and delivery systems that can ensure timely patient access, manage short-term affordability for expensive one-time treatments, and continue to reward the innovation that brings these new treatments to market,” Dr Ollendorf said.
Some of ICER’s recommendations include:
- When launching novel therapies approved with limited clinical evidence, such as CAR T-cell therapies, manufacturers and payers should consider using a lower launch price that could be increased if substantial clinical benefits are confirmed or using a higher initial price tied to a requirement for refunds or rebates if real-world evidence fails to confirm high expectations.
- Outcomes-based pricing arrangements must be linked to “meaningful clinical outcomes assessed with sufficient follow up.”
- Hospital mark-up for CAR T-cell therapies “should reflect the expected additional cost for care delivered in the hospital, rather than a percentage of the drug cost to avoid perverse incentives in choosing the treatment location.”
- Initially, CAR T-cell therapies should be delivered in “manufacturer-accredited centers to ensure the quality and appropriateness of care.” Later, “centers of excellence accredited by specialty societies” can administer these therapies, as long as providers have “sufficient expertise” to manage serious side effects.
- Centers should ensure that patients understand what to expect from CAR T-cell therapy, including long-term consequences.
- Because additional evidence on CAR T-cell therapies is needed, all patients who receive these therapies should enter into a registry with planned long-term follow-up.
- Studies should determine the optimal timing of CAR T-cell therapy in the sequence of treatments for B-ALL and NHL.
Additional recommendations and more details are available in ICER’s report.
About ICER
ICER is an independent, non-profit research institute that produces reports analyzing evidence on the effectiveness and value of drugs and other medical services.
ICER’s reports include evidence-based calculations of prices for new drugs that reflect the degree of improvement expected in long-term patient outcomes, while also highlighting price levels that might contribute to unaffordable short-term cost growth for the overall healthcare system.
ICER’s reports incorporate input from stakeholders and are the subject of public hearings through 3 core programs: the California Technology Assessment Forum, the Midwest Comparative Effectiveness Public Advisory Council, and the New England Comparative Effectiveness Public Advisory Council.
These independent panels review ICER’s reports at public meetings to deliberate on the evidence and develop recommendations for how patients, clinicians, insurers, and policymakers can improve the quality and value of healthcare.
Leukemia research pioneer dies at 92
James F. Holland, MD, passed away last week, at the age of 92, due to complications of cardiovascular disease.
Dr Holland has been called a pioneer in the field of leukemia research.
He and his colleagues are credited with using combination chemotherapy to transform pediatric acute lymphoblastic leukemia from an incurable illness to one with a high survival rate.
Dr Holland and his colleagues also developed the 7+3 regimen—3 daily injections of daunorubicin and 7 days of intravenous cytarabine—for patients with acute myeloid leukemia.
Dr Holland was born on May 16, 1925, in Morristown, New Jersey. He graduated from Princeton University in 1944 and earned his medical degree from Columbia University College of Physicians and Surgeons in 1947.
Dr Holland was a captain in the US Army Medical Corps from 1949 to 1951. After that, he worked at Francis Delafield Hospital (which closed in 1975) in New York, New York. He joined the National Cancer Institute (NCI) in 1953. Two years later, he began working at Roswell Park Cancer Institute in Buffalo, New York.
Dr Holland became Roswell Park’s chief of medicine and director of the Cancer Clinical Research Center. But he continued to work with the NCI, conducting research as part of Acute Leukemia Group B, which later became Cancer and Leukemia Group B.
After spending a year on an oncology exchange program in the Soviet Union, Dr Holland started at Mount Sinai in New York, New York, in 1973. While there, he established the Department of Neoplastic Diseases at The Tisch Cancer Institute, Icahn School of Medicine.
Most recently, Dr Holland was a distinguished professor of neoplastic diseases at Mount Sinai. He saw patients but also conducted research on the human mammary tumor virus.
Dr Holland collaborated with Emil Frei III to publish the textbook Cancer Medicine, which is now in its ninth edition. Dr Holland served as president of the American Association for Cancer Research and the American Society of Clinical Oncology. He was a co-founder of the African Organization for Research and Training in Cancer as well.
Dr Holland was married to Jimmie C. Holland, MD, who is credited with founding the field of psycho-oncology. Jimmie passed away in December 2017. The couple is survived by 6 children and 9 grandchildren.
James F. Holland, MD, passed away last week, at the age of 92, due to complications of cardiovascular disease.
Dr Holland has been called a pioneer in the field of leukemia research.
He and his colleagues are credited with using combination chemotherapy to transform pediatric acute lymphoblastic leukemia from an incurable illness to one with a high survival rate.
Dr Holland and his colleagues also developed the 7+3 regimen—3 daily injections of daunorubicin and 7 days of intravenous cytarabine—for patients with acute myeloid leukemia.
Dr Holland was born on May 16, 1925, in Morristown, New Jersey. He graduated from Princeton University in 1944 and earned his medical degree from Columbia University College of Physicians and Surgeons in 1947.
Dr Holland was a captain in the US Army Medical Corps from 1949 to 1951. After that, he worked at Francis Delafield Hospital (which closed in 1975) in New York, New York. He joined the National Cancer Institute (NCI) in 1953. Two years later, he began working at Roswell Park Cancer Institute in Buffalo, New York.
Dr Holland became Roswell Park’s chief of medicine and director of the Cancer Clinical Research Center. But he continued to work with the NCI, conducting research as part of Acute Leukemia Group B, which later became Cancer and Leukemia Group B.
After spending a year on an oncology exchange program in the Soviet Union, Dr Holland started at Mount Sinai in New York, New York, in 1973. While there, he established the Department of Neoplastic Diseases at The Tisch Cancer Institute, Icahn School of Medicine.
Most recently, Dr Holland was a distinguished professor of neoplastic diseases at Mount Sinai. He saw patients but also conducted research on the human mammary tumor virus.
Dr Holland collaborated with Emil Frei III to publish the textbook Cancer Medicine, which is now in its ninth edition. Dr Holland served as president of the American Association for Cancer Research and the American Society of Clinical Oncology. He was a co-founder of the African Organization for Research and Training in Cancer as well.
Dr Holland was married to Jimmie C. Holland, MD, who is credited with founding the field of psycho-oncology. Jimmie passed away in December 2017. The couple is survived by 6 children and 9 grandchildren.
James F. Holland, MD, passed away last week, at the age of 92, due to complications of cardiovascular disease.
Dr Holland has been called a pioneer in the field of leukemia research.
He and his colleagues are credited with using combination chemotherapy to transform pediatric acute lymphoblastic leukemia from an incurable illness to one with a high survival rate.
Dr Holland and his colleagues also developed the 7+3 regimen—3 daily injections of daunorubicin and 7 days of intravenous cytarabine—for patients with acute myeloid leukemia.
Dr Holland was born on May 16, 1925, in Morristown, New Jersey. He graduated from Princeton University in 1944 and earned his medical degree from Columbia University College of Physicians and Surgeons in 1947.
Dr Holland was a captain in the US Army Medical Corps from 1949 to 1951. After that, he worked at Francis Delafield Hospital (which closed in 1975) in New York, New York. He joined the National Cancer Institute (NCI) in 1953. Two years later, he began working at Roswell Park Cancer Institute in Buffalo, New York.
Dr Holland became Roswell Park’s chief of medicine and director of the Cancer Clinical Research Center. But he continued to work with the NCI, conducting research as part of Acute Leukemia Group B, which later became Cancer and Leukemia Group B.
After spending a year on an oncology exchange program in the Soviet Union, Dr Holland started at Mount Sinai in New York, New York, in 1973. While there, he established the Department of Neoplastic Diseases at The Tisch Cancer Institute, Icahn School of Medicine.
Most recently, Dr Holland was a distinguished professor of neoplastic diseases at Mount Sinai. He saw patients but also conducted research on the human mammary tumor virus.
Dr Holland collaborated with Emil Frei III to publish the textbook Cancer Medicine, which is now in its ninth edition. Dr Holland served as president of the American Association for Cancer Research and the American Society of Clinical Oncology. He was a co-founder of the African Organization for Research and Training in Cancer as well.
Dr Holland was married to Jimmie C. Holland, MD, who is credited with founding the field of psycho-oncology. Jimmie passed away in December 2017. The couple is survived by 6 children and 9 grandchildren.
FDA approves nilotinib for children with CML
Nilotinib is now approved for use by children aged 1 year and older with Philadelphia chromosome–positive chronic myeloid leukemia (Ph+ CML) in the chronic phase.
The Food and Drug Administration expanded the drug’s indication to include use as first- and second-line treatment in children.
Adverse events in the pediatric studies were similar to those observed in adults. However, children experienced hyperbilirubinemia (grade 3/4: 13%) and transaminase elevation (AST grade 3/4: 1%; ALT grade 3/4: 9%). Additionally, one previously treated pediatric patient progressed with advance phase/blast crisis after about 10 months of treatment.
Nilotinib (Tasigna) was already approved in adults with newly diagnosed Ph+ CML in the chronic phase and adults with chronic phase and accelerated phase Ph+ CML resistant or intolerant to prior therapy.
Nilotinib is now approved for use by children aged 1 year and older with Philadelphia chromosome–positive chronic myeloid leukemia (Ph+ CML) in the chronic phase.
The Food and Drug Administration expanded the drug’s indication to include use as first- and second-line treatment in children.
Adverse events in the pediatric studies were similar to those observed in adults. However, children experienced hyperbilirubinemia (grade 3/4: 13%) and transaminase elevation (AST grade 3/4: 1%; ALT grade 3/4: 9%). Additionally, one previously treated pediatric patient progressed with advance phase/blast crisis after about 10 months of treatment.
Nilotinib (Tasigna) was already approved in adults with newly diagnosed Ph+ CML in the chronic phase and adults with chronic phase and accelerated phase Ph+ CML resistant or intolerant to prior therapy.
Nilotinib is now approved for use by children aged 1 year and older with Philadelphia chromosome–positive chronic myeloid leukemia (Ph+ CML) in the chronic phase.
The Food and Drug Administration expanded the drug’s indication to include use as first- and second-line treatment in children.
Adverse events in the pediatric studies were similar to those observed in adults. However, children experienced hyperbilirubinemia (grade 3/4: 13%) and transaminase elevation (AST grade 3/4: 1%; ALT grade 3/4: 9%). Additionally, one previously treated pediatric patient progressed with advance phase/blast crisis after about 10 months of treatment.
Nilotinib (Tasigna) was already approved in adults with newly diagnosed Ph+ CML in the chronic phase and adults with chronic phase and accelerated phase Ph+ CML resistant or intolerant to prior therapy.
FDA approves nilotinib for kids with CML
The US Food and Drug Administration (FDA) has expanded the approved indication for nilotinib (Tasigna®) to include the treatment of children.
The drug is now approved to treat patients age 1 and older who have newly diagnosed Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) in the chronic phase.
Nilotinib is also approved to treat pediatric patients age 1 and older who have chronic phase, Ph+ CML that is resistant or intolerant to prior tyrosine kinase inhibitor (TKI) therapy, as well as adults with Ph+ CML in chronic phase and accelerated phase that is resistant or intolerant to prior therapy including imatinib.
The new pediatric indications for nilotinib, granted under the FDA’s priority review designation, are based on results from 2 studies of the drug—a phase 1 and phase 2.
According to Novartis, the studies included 69 CML patients who ranged in age from 2 to 17. They had either newly diagnosed, chronic phase, Ph+ CML or chronic phase, Ph+ CML with resistance or intolerance to prior TKI therapy.
In the newly diagnosed patients, the major molecular response (MMR) rate was 60.0% at 12 cycles, with 15 patients achieving MMR.
In patients with resistance or intolerance to prior therapy, the MMR rate was 40.9% at 12 cycles, with 18 patients being in MMR.
In newly diagnosed patients, the cumulative MMR rate was 64.0% by cycle 12. In patients with resistance or intolerance to prior therapy, the cumulative MMR rate was 47.7% by cycle 12.
Adverse events were generally consistent with those observed in adults, with the exception of hyperbilirubinemia and transaminase elevation, which were reported at a higher frequency than in adults.
The rate of grade 3/4 hyperbilirubinemia was 13.0%, the rate of grade 3/4 AST elevation was 1.4%, and the rate of grade 3/4 ALT elevation was 8.7%.
There were no deaths on treatment or after treatment discontinuation.
There was 1 patient with resistant/intolerant CML who progressed to advance phase/blast crisis after about 10 months on nilotinib.
The US Food and Drug Administration (FDA) has expanded the approved indication for nilotinib (Tasigna®) to include the treatment of children.
The drug is now approved to treat patients age 1 and older who have newly diagnosed Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) in the chronic phase.
Nilotinib is also approved to treat pediatric patients age 1 and older who have chronic phase, Ph+ CML that is resistant or intolerant to prior tyrosine kinase inhibitor (TKI) therapy, as well as adults with Ph+ CML in chronic phase and accelerated phase that is resistant or intolerant to prior therapy including imatinib.
The new pediatric indications for nilotinib, granted under the FDA’s priority review designation, are based on results from 2 studies of the drug—a phase 1 and phase 2.
According to Novartis, the studies included 69 CML patients who ranged in age from 2 to 17. They had either newly diagnosed, chronic phase, Ph+ CML or chronic phase, Ph+ CML with resistance or intolerance to prior TKI therapy.
In the newly diagnosed patients, the major molecular response (MMR) rate was 60.0% at 12 cycles, with 15 patients achieving MMR.
In patients with resistance or intolerance to prior therapy, the MMR rate was 40.9% at 12 cycles, with 18 patients being in MMR.
In newly diagnosed patients, the cumulative MMR rate was 64.0% by cycle 12. In patients with resistance or intolerance to prior therapy, the cumulative MMR rate was 47.7% by cycle 12.
Adverse events were generally consistent with those observed in adults, with the exception of hyperbilirubinemia and transaminase elevation, which were reported at a higher frequency than in adults.
The rate of grade 3/4 hyperbilirubinemia was 13.0%, the rate of grade 3/4 AST elevation was 1.4%, and the rate of grade 3/4 ALT elevation was 8.7%.
There were no deaths on treatment or after treatment discontinuation.
There was 1 patient with resistant/intolerant CML who progressed to advance phase/blast crisis after about 10 months on nilotinib.
The US Food and Drug Administration (FDA) has expanded the approved indication for nilotinib (Tasigna®) to include the treatment of children.
The drug is now approved to treat patients age 1 and older who have newly diagnosed Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) in the chronic phase.
Nilotinib is also approved to treat pediatric patients age 1 and older who have chronic phase, Ph+ CML that is resistant or intolerant to prior tyrosine kinase inhibitor (TKI) therapy, as well as adults with Ph+ CML in chronic phase and accelerated phase that is resistant or intolerant to prior therapy including imatinib.
The new pediatric indications for nilotinib, granted under the FDA’s priority review designation, are based on results from 2 studies of the drug—a phase 1 and phase 2.
According to Novartis, the studies included 69 CML patients who ranged in age from 2 to 17. They had either newly diagnosed, chronic phase, Ph+ CML or chronic phase, Ph+ CML with resistance or intolerance to prior TKI therapy.
In the newly diagnosed patients, the major molecular response (MMR) rate was 60.0% at 12 cycles, with 15 patients achieving MMR.
In patients with resistance or intolerance to prior therapy, the MMR rate was 40.9% at 12 cycles, with 18 patients being in MMR.
In newly diagnosed patients, the cumulative MMR rate was 64.0% by cycle 12. In patients with resistance or intolerance to prior therapy, the cumulative MMR rate was 47.7% by cycle 12.
Adverse events were generally consistent with those observed in adults, with the exception of hyperbilirubinemia and transaminase elevation, which were reported at a higher frequency than in adults.
The rate of grade 3/4 hyperbilirubinemia was 13.0%, the rate of grade 3/4 AST elevation was 1.4%, and the rate of grade 3/4 ALT elevation was 8.7%.
There were no deaths on treatment or after treatment discontinuation.
There was 1 patient with resistant/intolerant CML who progressed to advance phase/blast crisis after about 10 months on nilotinib.
Health Canada approves SC rituximab for CLL
Health Canada has approved a subcutaneous (SC) formulation of rituximab (Rituxan®) to treat patients with chronic lymphocytic leukemia (CLL).
The product is now approved for use in combination with fludarabine and cyclophosphamide to treat patients with previously treated or untreated CLL, Binet Stage B or C.
The SC formulation is intended to provide a more convenient delivery method than intravenous (IV) rituximab. The SC formulation enables administration of the drug in large volumes under the skin.
“The approval of Rituxan SC is exciting news and a meaningful advancement for those living with CLL, as intravenous treatments can take around 4 hours to receive, requiring patients to spend up to a half day sitting in the chemo suite,” said Robin Markowitz, chief executive officer of Lymphoma Canada.
“The administration of Rituxan SC only takes 7 minutes, giving patients valuable time back in their day.”
SAWYER study
Health Canada’s approval of SC rituximab is based on data from the phase 1b SAWYER study. This randomized study was conducted in patients with previously untreated CLL to investigate the pharmacokinetic profile, safety, and efficacy of SC rituximab in combination with chemotherapy.
Results from this study were published in The Lancet Haematology in March 2016.
The trial included 176 patients who were randomized to receive SC rituximab (1600 mg, n=88) or IV rituximab (500 mg/m2, n=88) on day 1 from cycles 2 to 6. In cycle 1, both groups received IV rituximab (375 mg/m2) on day 0.
All patients also received fludarabine (IV 25 mg/m2 on days 1-3 of all cycles or orally at either 25 mg/m2 on days 1-5 of all cycles or 30-40 mg/m2 on days 1-3 of all cycles) and cyclophosphamide (IV at 250 mg/m2 on days 1-3 of all cycles or orally at either 150 mg/m2 on days 1-5 of all cycles or 200-250 mg/m2 on days 1-3 of all cycles) every 4 weeks for up to 6 cycles.
The primary endpoint was pharmacokinetic noninferiority of SC to IV rituximab assessed at cycle 5. At that time, the geometric mean trough serum concentration in the SC rituximab arm was noninferior to that in the IV arm—97.5 µg/mL and 61.5 µg/mL, respectively—with an adjusted geometric mean ratio of 1.53 (90% CI, 1.27 to 1.85).
Safety was a secondary endpoint, and efficacy was an exploratory endpoint.
The overall response rate was 85% in the SC arm and 81% in the IV arm. Complete response rates were 26% and 33%, respectively.
Rates of adverse events (AEs) were 96% in the SC arm and 91% in the IV arm. Rates of grade 3 or higher AEs were 69% and 71%, respectively, and rates of serious AEs were 29% and 33%, respectively.
The most common serious AE overall was febrile neutropenia, which occurred in 11% of patients in the SC arm and 4% in the IV arm.
The most common AEs of any grade (occurring in >20% of patients in either arm) were:
- Neutropenia—65% with SC and 58% with IV
- Thrombocytopenia—24% with SC and 26% with IV
- Anemia—13% with SC and 24% with IV
- Nausea—38% with SC and 35% with IV
- Vomiting—21% with SC and 22% with IV
- Pyrexia—32% with SC and 25% with IV
- Injection-site erythema—26% with SC and 0% with IV.
Local cutaneous reactions were reported in 42% of patients in the SC arm. The most common of these were injection-site erythema (26%) and injection-site pain (16%).
Health Canada has approved a subcutaneous (SC) formulation of rituximab (Rituxan®) to treat patients with chronic lymphocytic leukemia (CLL).
The product is now approved for use in combination with fludarabine and cyclophosphamide to treat patients with previously treated or untreated CLL, Binet Stage B or C.
The SC formulation is intended to provide a more convenient delivery method than intravenous (IV) rituximab. The SC formulation enables administration of the drug in large volumes under the skin.
“The approval of Rituxan SC is exciting news and a meaningful advancement for those living with CLL, as intravenous treatments can take around 4 hours to receive, requiring patients to spend up to a half day sitting in the chemo suite,” said Robin Markowitz, chief executive officer of Lymphoma Canada.
“The administration of Rituxan SC only takes 7 minutes, giving patients valuable time back in their day.”
SAWYER study
Health Canada’s approval of SC rituximab is based on data from the phase 1b SAWYER study. This randomized study was conducted in patients with previously untreated CLL to investigate the pharmacokinetic profile, safety, and efficacy of SC rituximab in combination with chemotherapy.
Results from this study were published in The Lancet Haematology in March 2016.
The trial included 176 patients who were randomized to receive SC rituximab (1600 mg, n=88) or IV rituximab (500 mg/m2, n=88) on day 1 from cycles 2 to 6. In cycle 1, both groups received IV rituximab (375 mg/m2) on day 0.
All patients also received fludarabine (IV 25 mg/m2 on days 1-3 of all cycles or orally at either 25 mg/m2 on days 1-5 of all cycles or 30-40 mg/m2 on days 1-3 of all cycles) and cyclophosphamide (IV at 250 mg/m2 on days 1-3 of all cycles or orally at either 150 mg/m2 on days 1-5 of all cycles or 200-250 mg/m2 on days 1-3 of all cycles) every 4 weeks for up to 6 cycles.
The primary endpoint was pharmacokinetic noninferiority of SC to IV rituximab assessed at cycle 5. At that time, the geometric mean trough serum concentration in the SC rituximab arm was noninferior to that in the IV arm—97.5 µg/mL and 61.5 µg/mL, respectively—with an adjusted geometric mean ratio of 1.53 (90% CI, 1.27 to 1.85).
Safety was a secondary endpoint, and efficacy was an exploratory endpoint.
The overall response rate was 85% in the SC arm and 81% in the IV arm. Complete response rates were 26% and 33%, respectively.
Rates of adverse events (AEs) were 96% in the SC arm and 91% in the IV arm. Rates of grade 3 or higher AEs were 69% and 71%, respectively, and rates of serious AEs were 29% and 33%, respectively.
The most common serious AE overall was febrile neutropenia, which occurred in 11% of patients in the SC arm and 4% in the IV arm.
The most common AEs of any grade (occurring in >20% of patients in either arm) were:
- Neutropenia—65% with SC and 58% with IV
- Thrombocytopenia—24% with SC and 26% with IV
- Anemia—13% with SC and 24% with IV
- Nausea—38% with SC and 35% with IV
- Vomiting—21% with SC and 22% with IV
- Pyrexia—32% with SC and 25% with IV
- Injection-site erythema—26% with SC and 0% with IV.
Local cutaneous reactions were reported in 42% of patients in the SC arm. The most common of these were injection-site erythema (26%) and injection-site pain (16%).
Health Canada has approved a subcutaneous (SC) formulation of rituximab (Rituxan®) to treat patients with chronic lymphocytic leukemia (CLL).
The product is now approved for use in combination with fludarabine and cyclophosphamide to treat patients with previously treated or untreated CLL, Binet Stage B or C.
The SC formulation is intended to provide a more convenient delivery method than intravenous (IV) rituximab. The SC formulation enables administration of the drug in large volumes under the skin.
“The approval of Rituxan SC is exciting news and a meaningful advancement for those living with CLL, as intravenous treatments can take around 4 hours to receive, requiring patients to spend up to a half day sitting in the chemo suite,” said Robin Markowitz, chief executive officer of Lymphoma Canada.
“The administration of Rituxan SC only takes 7 minutes, giving patients valuable time back in their day.”
SAWYER study
Health Canada’s approval of SC rituximab is based on data from the phase 1b SAWYER study. This randomized study was conducted in patients with previously untreated CLL to investigate the pharmacokinetic profile, safety, and efficacy of SC rituximab in combination with chemotherapy.
Results from this study were published in The Lancet Haematology in March 2016.
The trial included 176 patients who were randomized to receive SC rituximab (1600 mg, n=88) or IV rituximab (500 mg/m2, n=88) on day 1 from cycles 2 to 6. In cycle 1, both groups received IV rituximab (375 mg/m2) on day 0.
All patients also received fludarabine (IV 25 mg/m2 on days 1-3 of all cycles or orally at either 25 mg/m2 on days 1-5 of all cycles or 30-40 mg/m2 on days 1-3 of all cycles) and cyclophosphamide (IV at 250 mg/m2 on days 1-3 of all cycles or orally at either 150 mg/m2 on days 1-5 of all cycles or 200-250 mg/m2 on days 1-3 of all cycles) every 4 weeks for up to 6 cycles.
The primary endpoint was pharmacokinetic noninferiority of SC to IV rituximab assessed at cycle 5. At that time, the geometric mean trough serum concentration in the SC rituximab arm was noninferior to that in the IV arm—97.5 µg/mL and 61.5 µg/mL, respectively—with an adjusted geometric mean ratio of 1.53 (90% CI, 1.27 to 1.85).
Safety was a secondary endpoint, and efficacy was an exploratory endpoint.
The overall response rate was 85% in the SC arm and 81% in the IV arm. Complete response rates were 26% and 33%, respectively.
Rates of adverse events (AEs) were 96% in the SC arm and 91% in the IV arm. Rates of grade 3 or higher AEs were 69% and 71%, respectively, and rates of serious AEs were 29% and 33%, respectively.
The most common serious AE overall was febrile neutropenia, which occurred in 11% of patients in the SC arm and 4% in the IV arm.
The most common AEs of any grade (occurring in >20% of patients in either arm) were:
- Neutropenia—65% with SC and 58% with IV
- Thrombocytopenia—24% with SC and 26% with IV
- Anemia—13% with SC and 24% with IV
- Nausea—38% with SC and 35% with IV
- Vomiting—21% with SC and 22% with IV
- Pyrexia—32% with SC and 25% with IV
- Injection-site erythema—26% with SC and 0% with IV.
Local cutaneous reactions were reported in 42% of patients in the SC arm. The most common of these were injection-site erythema (26%) and injection-site pain (16%).
Ponatinib efficacy maintained despite dose reductions
Final results from the phase 2 PACE trial suggest dose reductions did not have an effect on the efficacy of ponatinib in patients with chronic phase (CP) chronic myeloid leukemia (CML).
Trial investigators began reducing ponatinib doses after the drug was linked to arterial occlusive events (AOEs).
Most patients who achieved a major molecular response (MMR) or major cytogenetic response (MCyR) on higher doses of ponatinib maintained those responses after dose reductions.
These results were published in Blood. The trial was sponsored by Ariad Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited.
Earlier results from the PACE trial indicated that ponatinib increased a patient’s risk of vascular occlusive events (VOEs), including AOEs.
Therefore, in October 2013, a phase 3 trial of ponatinib was discontinued, and all other ponatinib trials were put on partial clinical hold. Trial enrollment was halted temporarily, and investigators began reducing ponatinib doses.
Then, the US Food and Drug Administration suspended sales and marketing of ponatinib, pending results of a safety evaluation.
However, in December 2013, the agency decided ponatinib could return to the market if new safety measures were implemented. In January 2014, the drug was back on the market.
Patients
The PACE trial included 449 patients with CML or acute lymphoblastic leukemia, but the results published in Blood focused only on patients with CP-CML. All of these patients were resistant or intolerant to dasatinib or nilotinib, or they had the BCR-ABLT315I mutation.
There were 270 patients with CP-CML. They had a median age of 60 (range, 18-94), and 47% were female.
Ninety-three percent had received at least 2 prior approved tyrosine kinase inhibitors (TKIs), and 57% had received at least 3. The median duration of prior TKI treatment was 5.4 years (range, 0.4-13.3).
Eighty percent of patients were resistant to dasatinib or nilotinib, 14% were intolerant to either drug, and 19% were both resistant and intolerant. Twenty-four percent of patients had the T315I mutation.
Treatment
The starting dose of ponatinib was 45 mg once daily. Doses were reduced to 30 mg or 15 mg once daily to manage adverse events (AEs), or reductions were implemented proactively (starting on October 10, 2013) due to concerns about VOEs.
Unless a benefit-risk analysis justified use of a higher dose, the recommendation for CP-CML patients was a 15 mg daily dose for those with an MCyR and a 30 mg daily dose for those without an MCyR.
Patients received ponatinib until disease progression, intolerance, or the patient or investigator decided to stop treatment.
The median duration of treatment was 32.1 months (range, 0.1-73.0), and the median follow-up was 56.8 months (range, 0.1-73.1).
Efficacy
Of the 267 evaluable patients, 60% achieved an MCyR at any time, with 54% achieving a complete cytogenetic response. Forty-eight percent of patients were still in MCyR at the last response assessment, and 82% of patients who achieved an MCyR were estimated to remain in MCyR at 5 years.
There were 69 patients in MCyR as of October 10, 2013, when pre-emptive dose reduction began, who had their dose reduced. Ninety-six percent of these patients (n=66) maintained MCyR after dose reduction. Of the 34 patients in MCyR who did not have pre-emptive dose reductions, 94% (n=32) maintained MCyR.
Forty percent of patients attained a major molecular response (MMR) at any time during the study, with 30% achieving MR4 and 24% achieving MR4.5. Thirty percent of patients were in MMR at the last response assessment, and 59% of patients who achieved MMR were estimated to remain in MMR at 5 years.
There were 52 patients in MMR as of October 10, 2013, who had their dose reduced. Ninety percent (n=47) of these patients maintained MMR following dose reduction. Of the 19 patients in MMR who did not have pre-emptive dose reductions, 95% (n=18) maintained MMR.
“The PACE trial is among the longest and largest studies of patients with CP-CML who have received 2 or 3 prior TKIs, and the findings provide treating physicians with important updated information . . . ,” said study author Jorge Eduardo Cortes, MD, of MD Anderson Cancer Center in Houston, Texas.
“These final PACE results demonstrate that [ponatinib] provides lasting clinically meaningful responses, irrespective of dose reductions, in this population.”
At 5 years, the progression-free survival rate was 53%, and the overall survival rate was 73%.
AOEs
The incidence of AOEs was 31%, and the incidence of serious AOEs was 26%. This included cardiovascular AOEs (16%, 12% serious), cerebrovascular (13%, 10% serious), and peripheral vascular AOEs (14%, 11% serious). The exposure-adjusted incidence of AOEs was 14.1 per 100 patient-years.
Among patients without AOEs prior to October 2013 when pre-emptive dose reductions began, the incidence of AOEs was 19% in patients who had dose reductions and 18% in patients who did not.
The cumulative incidence of AOEs increased over time, but the exposure-adjusted incidence of new AOEs did not. It was 15.8 per 100 patient-years in year 1 and 4.9 per 100 patient-years in year 5.
The investigators said the lack of increase in exposure-adjusted AOE incidence could be due to the natural history or etiology of AOEs, dose reductions, or a change in the patient population (ie, an enrichment of patients who may have a lower risk of vascular events).
Therefore, it is unclear whether lower doses of ponatinib reduce the risk of AOEs in patients with risk factors. However, AOEs appear to be dose-related and modified by pre-existing cardiovascular disease and other risk factors.
Venous thromboembolism (VTE), on the other hand, does not seem to be dose-related. The investigators said the rate of VTE in this study was consistent with rates typically observed in cancer patients.
The incidence of VTE was 6%, and 5% of patients had serious VTEs. The exposure-adjusted incidence of VTEs was 2.1 per 100 patient-years.
Other AEs
The most common any-grade treatment-emergent AEs (≥40%) were rash (47%), abdominal pain (46%), thrombocytopenia (46%), headache (43%), dry skin (42%), and constipation (41%).
The most common grade 3/4 treatment-emergent AEs (≥10%) were thrombocytopenia (35%), neutropenia (17%), hypertension (14%), increased lipase (13%), abdominal pain (10%), and anemia (10%).
Serious AEs (≥5%) included pancreatitis (7%), atrial fibrillation (6%), pneumonia (6%), and angina pectoris (5%).
There were 12 deaths (4%) that occurred on study or within 30 days of the end of study treatment. Two deaths were considered possibly or probably related to ponatinib, 1 due to pneumonia and 1 due to acute myocardial infarction.
Final results from the phase 2 PACE trial suggest dose reductions did not have an effect on the efficacy of ponatinib in patients with chronic phase (CP) chronic myeloid leukemia (CML).
Trial investigators began reducing ponatinib doses after the drug was linked to arterial occlusive events (AOEs).
Most patients who achieved a major molecular response (MMR) or major cytogenetic response (MCyR) on higher doses of ponatinib maintained those responses after dose reductions.
These results were published in Blood. The trial was sponsored by Ariad Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited.
Earlier results from the PACE trial indicated that ponatinib increased a patient’s risk of vascular occlusive events (VOEs), including AOEs.
Therefore, in October 2013, a phase 3 trial of ponatinib was discontinued, and all other ponatinib trials were put on partial clinical hold. Trial enrollment was halted temporarily, and investigators began reducing ponatinib doses.
Then, the US Food and Drug Administration suspended sales and marketing of ponatinib, pending results of a safety evaluation.
However, in December 2013, the agency decided ponatinib could return to the market if new safety measures were implemented. In January 2014, the drug was back on the market.
Patients
The PACE trial included 449 patients with CML or acute lymphoblastic leukemia, but the results published in Blood focused only on patients with CP-CML. All of these patients were resistant or intolerant to dasatinib or nilotinib, or they had the BCR-ABLT315I mutation.
There were 270 patients with CP-CML. They had a median age of 60 (range, 18-94), and 47% were female.
Ninety-three percent had received at least 2 prior approved tyrosine kinase inhibitors (TKIs), and 57% had received at least 3. The median duration of prior TKI treatment was 5.4 years (range, 0.4-13.3).
Eighty percent of patients were resistant to dasatinib or nilotinib, 14% were intolerant to either drug, and 19% were both resistant and intolerant. Twenty-four percent of patients had the T315I mutation.
Treatment
The starting dose of ponatinib was 45 mg once daily. Doses were reduced to 30 mg or 15 mg once daily to manage adverse events (AEs), or reductions were implemented proactively (starting on October 10, 2013) due to concerns about VOEs.
Unless a benefit-risk analysis justified use of a higher dose, the recommendation for CP-CML patients was a 15 mg daily dose for those with an MCyR and a 30 mg daily dose for those without an MCyR.
Patients received ponatinib until disease progression, intolerance, or the patient or investigator decided to stop treatment.
The median duration of treatment was 32.1 months (range, 0.1-73.0), and the median follow-up was 56.8 months (range, 0.1-73.1).
Efficacy
Of the 267 evaluable patients, 60% achieved an MCyR at any time, with 54% achieving a complete cytogenetic response. Forty-eight percent of patients were still in MCyR at the last response assessment, and 82% of patients who achieved an MCyR were estimated to remain in MCyR at 5 years.
There were 69 patients in MCyR as of October 10, 2013, when pre-emptive dose reduction began, who had their dose reduced. Ninety-six percent of these patients (n=66) maintained MCyR after dose reduction. Of the 34 patients in MCyR who did not have pre-emptive dose reductions, 94% (n=32) maintained MCyR.
Forty percent of patients attained a major molecular response (MMR) at any time during the study, with 30% achieving MR4 and 24% achieving MR4.5. Thirty percent of patients were in MMR at the last response assessment, and 59% of patients who achieved MMR were estimated to remain in MMR at 5 years.
There were 52 patients in MMR as of October 10, 2013, who had their dose reduced. Ninety percent (n=47) of these patients maintained MMR following dose reduction. Of the 19 patients in MMR who did not have pre-emptive dose reductions, 95% (n=18) maintained MMR.
“The PACE trial is among the longest and largest studies of patients with CP-CML who have received 2 or 3 prior TKIs, and the findings provide treating physicians with important updated information . . . ,” said study author Jorge Eduardo Cortes, MD, of MD Anderson Cancer Center in Houston, Texas.
“These final PACE results demonstrate that [ponatinib] provides lasting clinically meaningful responses, irrespective of dose reductions, in this population.”
At 5 years, the progression-free survival rate was 53%, and the overall survival rate was 73%.
AOEs
The incidence of AOEs was 31%, and the incidence of serious AOEs was 26%. This included cardiovascular AOEs (16%, 12% serious), cerebrovascular (13%, 10% serious), and peripheral vascular AOEs (14%, 11% serious). The exposure-adjusted incidence of AOEs was 14.1 per 100 patient-years.
Among patients without AOEs prior to October 2013 when pre-emptive dose reductions began, the incidence of AOEs was 19% in patients who had dose reductions and 18% in patients who did not.
The cumulative incidence of AOEs increased over time, but the exposure-adjusted incidence of new AOEs did not. It was 15.8 per 100 patient-years in year 1 and 4.9 per 100 patient-years in year 5.
The investigators said the lack of increase in exposure-adjusted AOE incidence could be due to the natural history or etiology of AOEs, dose reductions, or a change in the patient population (ie, an enrichment of patients who may have a lower risk of vascular events).
Therefore, it is unclear whether lower doses of ponatinib reduce the risk of AOEs in patients with risk factors. However, AOEs appear to be dose-related and modified by pre-existing cardiovascular disease and other risk factors.
Venous thromboembolism (VTE), on the other hand, does not seem to be dose-related. The investigators said the rate of VTE in this study was consistent with rates typically observed in cancer patients.
The incidence of VTE was 6%, and 5% of patients had serious VTEs. The exposure-adjusted incidence of VTEs was 2.1 per 100 patient-years.
Other AEs
The most common any-grade treatment-emergent AEs (≥40%) were rash (47%), abdominal pain (46%), thrombocytopenia (46%), headache (43%), dry skin (42%), and constipation (41%).
The most common grade 3/4 treatment-emergent AEs (≥10%) were thrombocytopenia (35%), neutropenia (17%), hypertension (14%), increased lipase (13%), abdominal pain (10%), and anemia (10%).
Serious AEs (≥5%) included pancreatitis (7%), atrial fibrillation (6%), pneumonia (6%), and angina pectoris (5%).
There were 12 deaths (4%) that occurred on study or within 30 days of the end of study treatment. Two deaths were considered possibly or probably related to ponatinib, 1 due to pneumonia and 1 due to acute myocardial infarction.
Final results from the phase 2 PACE trial suggest dose reductions did not have an effect on the efficacy of ponatinib in patients with chronic phase (CP) chronic myeloid leukemia (CML).
Trial investigators began reducing ponatinib doses after the drug was linked to arterial occlusive events (AOEs).
Most patients who achieved a major molecular response (MMR) or major cytogenetic response (MCyR) on higher doses of ponatinib maintained those responses after dose reductions.
These results were published in Blood. The trial was sponsored by Ariad Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited.
Earlier results from the PACE trial indicated that ponatinib increased a patient’s risk of vascular occlusive events (VOEs), including AOEs.
Therefore, in October 2013, a phase 3 trial of ponatinib was discontinued, and all other ponatinib trials were put on partial clinical hold. Trial enrollment was halted temporarily, and investigators began reducing ponatinib doses.
Then, the US Food and Drug Administration suspended sales and marketing of ponatinib, pending results of a safety evaluation.
However, in December 2013, the agency decided ponatinib could return to the market if new safety measures were implemented. In January 2014, the drug was back on the market.
Patients
The PACE trial included 449 patients with CML or acute lymphoblastic leukemia, but the results published in Blood focused only on patients with CP-CML. All of these patients were resistant or intolerant to dasatinib or nilotinib, or they had the BCR-ABLT315I mutation.
There were 270 patients with CP-CML. They had a median age of 60 (range, 18-94), and 47% were female.
Ninety-three percent had received at least 2 prior approved tyrosine kinase inhibitors (TKIs), and 57% had received at least 3. The median duration of prior TKI treatment was 5.4 years (range, 0.4-13.3).
Eighty percent of patients were resistant to dasatinib or nilotinib, 14% were intolerant to either drug, and 19% were both resistant and intolerant. Twenty-four percent of patients had the T315I mutation.
Treatment
The starting dose of ponatinib was 45 mg once daily. Doses were reduced to 30 mg or 15 mg once daily to manage adverse events (AEs), or reductions were implemented proactively (starting on October 10, 2013) due to concerns about VOEs.
Unless a benefit-risk analysis justified use of a higher dose, the recommendation for CP-CML patients was a 15 mg daily dose for those with an MCyR and a 30 mg daily dose for those without an MCyR.
Patients received ponatinib until disease progression, intolerance, or the patient or investigator decided to stop treatment.
The median duration of treatment was 32.1 months (range, 0.1-73.0), and the median follow-up was 56.8 months (range, 0.1-73.1).
Efficacy
Of the 267 evaluable patients, 60% achieved an MCyR at any time, with 54% achieving a complete cytogenetic response. Forty-eight percent of patients were still in MCyR at the last response assessment, and 82% of patients who achieved an MCyR were estimated to remain in MCyR at 5 years.
There were 69 patients in MCyR as of October 10, 2013, when pre-emptive dose reduction began, who had their dose reduced. Ninety-six percent of these patients (n=66) maintained MCyR after dose reduction. Of the 34 patients in MCyR who did not have pre-emptive dose reductions, 94% (n=32) maintained MCyR.
Forty percent of patients attained a major molecular response (MMR) at any time during the study, with 30% achieving MR4 and 24% achieving MR4.5. Thirty percent of patients were in MMR at the last response assessment, and 59% of patients who achieved MMR were estimated to remain in MMR at 5 years.
There were 52 patients in MMR as of October 10, 2013, who had their dose reduced. Ninety percent (n=47) of these patients maintained MMR following dose reduction. Of the 19 patients in MMR who did not have pre-emptive dose reductions, 95% (n=18) maintained MMR.
“The PACE trial is among the longest and largest studies of patients with CP-CML who have received 2 or 3 prior TKIs, and the findings provide treating physicians with important updated information . . . ,” said study author Jorge Eduardo Cortes, MD, of MD Anderson Cancer Center in Houston, Texas.
“These final PACE results demonstrate that [ponatinib] provides lasting clinically meaningful responses, irrespective of dose reductions, in this population.”
At 5 years, the progression-free survival rate was 53%, and the overall survival rate was 73%.
AOEs
The incidence of AOEs was 31%, and the incidence of serious AOEs was 26%. This included cardiovascular AOEs (16%, 12% serious), cerebrovascular (13%, 10% serious), and peripheral vascular AOEs (14%, 11% serious). The exposure-adjusted incidence of AOEs was 14.1 per 100 patient-years.
Among patients without AOEs prior to October 2013 when pre-emptive dose reductions began, the incidence of AOEs was 19% in patients who had dose reductions and 18% in patients who did not.
The cumulative incidence of AOEs increased over time, but the exposure-adjusted incidence of new AOEs did not. It was 15.8 per 100 patient-years in year 1 and 4.9 per 100 patient-years in year 5.
The investigators said the lack of increase in exposure-adjusted AOE incidence could be due to the natural history or etiology of AOEs, dose reductions, or a change in the patient population (ie, an enrichment of patients who may have a lower risk of vascular events).
Therefore, it is unclear whether lower doses of ponatinib reduce the risk of AOEs in patients with risk factors. However, AOEs appear to be dose-related and modified by pre-existing cardiovascular disease and other risk factors.
Venous thromboembolism (VTE), on the other hand, does not seem to be dose-related. The investigators said the rate of VTE in this study was consistent with rates typically observed in cancer patients.
The incidence of VTE was 6%, and 5% of patients had serious VTEs. The exposure-adjusted incidence of VTEs was 2.1 per 100 patient-years.
Other AEs
The most common any-grade treatment-emergent AEs (≥40%) were rash (47%), abdominal pain (46%), thrombocytopenia (46%), headache (43%), dry skin (42%), and constipation (41%).
The most common grade 3/4 treatment-emergent AEs (≥10%) were thrombocytopenia (35%), neutropenia (17%), hypertension (14%), increased lipase (13%), abdominal pain (10%), and anemia (10%).
Serious AEs (≥5%) included pancreatitis (7%), atrial fibrillation (6%), pneumonia (6%), and angina pectoris (5%).
There were 12 deaths (4%) that occurred on study or within 30 days of the end of study treatment. Two deaths were considered possibly or probably related to ponatinib, 1 due to pneumonia and 1 due to acute myocardial infarction.
Chemotherapy, metabolic pathway may affect CAR T-cell potential
Two critical factors – prior exposure to chemotherapy and a glycolytic metabolism – appear to degrade the potential of T cells to become chimeric antigen receptor–T cells.
Chemotherapy, especially with cyclophosphamide and doxorubicin, seems particularly toxic to T cells, damaging the mitochondria and decreasing the cells’ spare respiratory capacity – a measure of mitochondrial health, David Barrett, MD, said during a press briefing held in advance of the annual meeting of the American Association for Cancer Research.
These new findings may help explain why children with acute lymphoblastic leukemia (ALL) tend to respond so vigorously to CAR T treatment, and why T cells from patients with solid tumors simply don’t grow, or die soon after patient infusion, he said in an interview. They also suggest a benefit of harvesting T cells before any chemotherapy, a procedure Dr. Barrett and his colleagues have advocated.
“Based on these data we have altered our practice for T-cell therapy in high-risk leukemia patients. If we have a patient who may have a poor prognosis, we try to collect the cells early and store them before proceeding, because we know chemotherapy will progressively degrade them.”
There still is no successful CAR T-cell protocol for solid tumors, but Dr. Barrett said these findings eventually may help such patients, particularly if more advanced experiments in manipulating the cells’ metabolism prove successful.
He and his colleagues investigated why T cells from some patients result in a poor clinical product that either fails manufacture or does not proliferate in the patient. They examined T cells from 157 pediatric patients with a variety of cancers, including ALL, non-Hodgkin lymphoma, neuroblastoma, osteosarcoma, rhabdomyosarcoma, Wilms tumor, Hodgkin disease, chronic myelogenous leukemia, and Ewing sarcoma. The team obtained cells at diagnosis and after each cycle of chemotherapy.
They examined how well the cells grew in the transformation and expansion process. A “pass” was considered a fivefold expansion in response to CD3/CD28 exposure for 7 days. Normal donor cells typically expand 20- to 30-fold in this time.
Only T cells taken from ALL and Wilms tumor patients before chemotherapy achieved a pass, Dr. Barrett said. Most of the ALL expansions (80%) and half of the Wilms tumor expansions passed. “We noted very poor CAR T-cell potential in all the other tumor types – less than a 30% pass. We noted a decline in potential with cumulative chemotherapy in all cases, though this was particularly significant in children less than 3 years old.”
The team also used RNA profiling to look at the cells’ metabolic pathways. Dr. Barrett noted that T cells are highly metabolically adaptable, capable of using several different fuel types and switching from one to another. Glucose and fatty acids are frequent fuels. Most of the cells from patients with solid tumors exhibited a glycolytic metabolism, while cells from patients with ALL and Wilms tumor appeared to rely more on fatty acids.
“One is not inherently worse than the other,” he said. “But glycolysis appears to be a bad thing when we’re trying to turn them into CAR T cells. Those T cells were too exhausted to do anything.”
However, Dr. Barrett encouraged the cells to switch fuels by treating them in vitro with palmitic acid, the most common fatty acid in plants and animals.
“We were growing the cells in a media containing sugar, fatty acids, and amino acids,” he explained. “We just started overloading them with palmitic acid, which has a natural transporter on the T-cell surface, so it already had a good pathway to get into the cell. It helped restore some of the performance of these T cells in some assays, although it wasn’t a complete reversal. But it was encouraging that something as simple as providing an alternate fuel was enough to get some positive effect. Whether or not we would also have to block glucose use to get it to really work is something we continue to study.”
T cells that had been exposed to chemotherapy also did poorly. Cyclophosphamide and doxorubicin seemed particularly toxic. Cells with exposure to these two agents had severely depleted CAR T cell potential with very poor spare respiratory capacity. This is a marker of mitochondrial injury, Dr. Barrett said. “That wasn’t a huge surprise. We already knew that cyclophosphamide is very toxic to T cells.”
But the finding did suggest the simple intervention of harvesting T cells before chemotherapy, which is what Dr. Barrett and his colleagues now do in their high-risk ALL patients. Whether or not this would improve response in patients with solid tumors is still unknown.
He had no financial disclosures. This study was supported by the AACR, the Doris Duke Charitable Foundation Clinical Science Development Award, the Jeffrey Pride Foundation Research Award, and the St. Baldrick’s Foundation Scholar Award.
SOURCE: Barrett DM et al. AACR 2018, Abstract 1631.
Two critical factors – prior exposure to chemotherapy and a glycolytic metabolism – appear to degrade the potential of T cells to become chimeric antigen receptor–T cells.
Chemotherapy, especially with cyclophosphamide and doxorubicin, seems particularly toxic to T cells, damaging the mitochondria and decreasing the cells’ spare respiratory capacity – a measure of mitochondrial health, David Barrett, MD, said during a press briefing held in advance of the annual meeting of the American Association for Cancer Research.
These new findings may help explain why children with acute lymphoblastic leukemia (ALL) tend to respond so vigorously to CAR T treatment, and why T cells from patients with solid tumors simply don’t grow, or die soon after patient infusion, he said in an interview. They also suggest a benefit of harvesting T cells before any chemotherapy, a procedure Dr. Barrett and his colleagues have advocated.
“Based on these data we have altered our practice for T-cell therapy in high-risk leukemia patients. If we have a patient who may have a poor prognosis, we try to collect the cells early and store them before proceeding, because we know chemotherapy will progressively degrade them.”
There still is no successful CAR T-cell protocol for solid tumors, but Dr. Barrett said these findings eventually may help such patients, particularly if more advanced experiments in manipulating the cells’ metabolism prove successful.
He and his colleagues investigated why T cells from some patients result in a poor clinical product that either fails manufacture or does not proliferate in the patient. They examined T cells from 157 pediatric patients with a variety of cancers, including ALL, non-Hodgkin lymphoma, neuroblastoma, osteosarcoma, rhabdomyosarcoma, Wilms tumor, Hodgkin disease, chronic myelogenous leukemia, and Ewing sarcoma. The team obtained cells at diagnosis and after each cycle of chemotherapy.
They examined how well the cells grew in the transformation and expansion process. A “pass” was considered a fivefold expansion in response to CD3/CD28 exposure for 7 days. Normal donor cells typically expand 20- to 30-fold in this time.
Only T cells taken from ALL and Wilms tumor patients before chemotherapy achieved a pass, Dr. Barrett said. Most of the ALL expansions (80%) and half of the Wilms tumor expansions passed. “We noted very poor CAR T-cell potential in all the other tumor types – less than a 30% pass. We noted a decline in potential with cumulative chemotherapy in all cases, though this was particularly significant in children less than 3 years old.”
The team also used RNA profiling to look at the cells’ metabolic pathways. Dr. Barrett noted that T cells are highly metabolically adaptable, capable of using several different fuel types and switching from one to another. Glucose and fatty acids are frequent fuels. Most of the cells from patients with solid tumors exhibited a glycolytic metabolism, while cells from patients with ALL and Wilms tumor appeared to rely more on fatty acids.
“One is not inherently worse than the other,” he said. “But glycolysis appears to be a bad thing when we’re trying to turn them into CAR T cells. Those T cells were too exhausted to do anything.”
However, Dr. Barrett encouraged the cells to switch fuels by treating them in vitro with palmitic acid, the most common fatty acid in plants and animals.
“We were growing the cells in a media containing sugar, fatty acids, and amino acids,” he explained. “We just started overloading them with palmitic acid, which has a natural transporter on the T-cell surface, so it already had a good pathway to get into the cell. It helped restore some of the performance of these T cells in some assays, although it wasn’t a complete reversal. But it was encouraging that something as simple as providing an alternate fuel was enough to get some positive effect. Whether or not we would also have to block glucose use to get it to really work is something we continue to study.”
T cells that had been exposed to chemotherapy also did poorly. Cyclophosphamide and doxorubicin seemed particularly toxic. Cells with exposure to these two agents had severely depleted CAR T cell potential with very poor spare respiratory capacity. This is a marker of mitochondrial injury, Dr. Barrett said. “That wasn’t a huge surprise. We already knew that cyclophosphamide is very toxic to T cells.”
But the finding did suggest the simple intervention of harvesting T cells before chemotherapy, which is what Dr. Barrett and his colleagues now do in their high-risk ALL patients. Whether or not this would improve response in patients with solid tumors is still unknown.
He had no financial disclosures. This study was supported by the AACR, the Doris Duke Charitable Foundation Clinical Science Development Award, the Jeffrey Pride Foundation Research Award, and the St. Baldrick’s Foundation Scholar Award.
SOURCE: Barrett DM et al. AACR 2018, Abstract 1631.
Two critical factors – prior exposure to chemotherapy and a glycolytic metabolism – appear to degrade the potential of T cells to become chimeric antigen receptor–T cells.
Chemotherapy, especially with cyclophosphamide and doxorubicin, seems particularly toxic to T cells, damaging the mitochondria and decreasing the cells’ spare respiratory capacity – a measure of mitochondrial health, David Barrett, MD, said during a press briefing held in advance of the annual meeting of the American Association for Cancer Research.
These new findings may help explain why children with acute lymphoblastic leukemia (ALL) tend to respond so vigorously to CAR T treatment, and why T cells from patients with solid tumors simply don’t grow, or die soon after patient infusion, he said in an interview. They also suggest a benefit of harvesting T cells before any chemotherapy, a procedure Dr. Barrett and his colleagues have advocated.
“Based on these data we have altered our practice for T-cell therapy in high-risk leukemia patients. If we have a patient who may have a poor prognosis, we try to collect the cells early and store them before proceeding, because we know chemotherapy will progressively degrade them.”
There still is no successful CAR T-cell protocol for solid tumors, but Dr. Barrett said these findings eventually may help such patients, particularly if more advanced experiments in manipulating the cells’ metabolism prove successful.
He and his colleagues investigated why T cells from some patients result in a poor clinical product that either fails manufacture or does not proliferate in the patient. They examined T cells from 157 pediatric patients with a variety of cancers, including ALL, non-Hodgkin lymphoma, neuroblastoma, osteosarcoma, rhabdomyosarcoma, Wilms tumor, Hodgkin disease, chronic myelogenous leukemia, and Ewing sarcoma. The team obtained cells at diagnosis and after each cycle of chemotherapy.
They examined how well the cells grew in the transformation and expansion process. A “pass” was considered a fivefold expansion in response to CD3/CD28 exposure for 7 days. Normal donor cells typically expand 20- to 30-fold in this time.
Only T cells taken from ALL and Wilms tumor patients before chemotherapy achieved a pass, Dr. Barrett said. Most of the ALL expansions (80%) and half of the Wilms tumor expansions passed. “We noted very poor CAR T-cell potential in all the other tumor types – less than a 30% pass. We noted a decline in potential with cumulative chemotherapy in all cases, though this was particularly significant in children less than 3 years old.”
The team also used RNA profiling to look at the cells’ metabolic pathways. Dr. Barrett noted that T cells are highly metabolically adaptable, capable of using several different fuel types and switching from one to another. Glucose and fatty acids are frequent fuels. Most of the cells from patients with solid tumors exhibited a glycolytic metabolism, while cells from patients with ALL and Wilms tumor appeared to rely more on fatty acids.
“One is not inherently worse than the other,” he said. “But glycolysis appears to be a bad thing when we’re trying to turn them into CAR T cells. Those T cells were too exhausted to do anything.”
However, Dr. Barrett encouraged the cells to switch fuels by treating them in vitro with palmitic acid, the most common fatty acid in plants and animals.
“We were growing the cells in a media containing sugar, fatty acids, and amino acids,” he explained. “We just started overloading them with palmitic acid, which has a natural transporter on the T-cell surface, so it already had a good pathway to get into the cell. It helped restore some of the performance of these T cells in some assays, although it wasn’t a complete reversal. But it was encouraging that something as simple as providing an alternate fuel was enough to get some positive effect. Whether or not we would also have to block glucose use to get it to really work is something we continue to study.”
T cells that had been exposed to chemotherapy also did poorly. Cyclophosphamide and doxorubicin seemed particularly toxic. Cells with exposure to these two agents had severely depleted CAR T cell potential with very poor spare respiratory capacity. This is a marker of mitochondrial injury, Dr. Barrett said. “That wasn’t a huge surprise. We already knew that cyclophosphamide is very toxic to T cells.”
But the finding did suggest the simple intervention of harvesting T cells before chemotherapy, which is what Dr. Barrett and his colleagues now do in their high-risk ALL patients. Whether or not this would improve response in patients with solid tumors is still unknown.
He had no financial disclosures. This study was supported by the AACR, the Doris Duke Charitable Foundation Clinical Science Development Award, the Jeffrey Pride Foundation Research Award, and the St. Baldrick’s Foundation Scholar Award.
SOURCE: Barrett DM et al. AACR 2018, Abstract 1631.
FROM AACR 2018
Key clinical point: Prior exposure to chemotherapy may degrade the potential of T cells to become CAR T cells, suggesting a benefit of harvesting T cells before any chemotherapy.
Major finding: Only T cells taken from ALL and Wilm’s tumor patients before chemotherapy achieved a fivefold expansion in response to CD3/CD28 exposure for 7 days.
Study details: An examination of T cells from 157 pediatric patients with a variety of cancers at diagnosis and after each cycle of chemotherapy.
Disclosures: The study was supported by the American Association of Cancer Research, the Doris Duke Charitable Foundation Clinical Science Development Award, the Jeffrey Pride Foundation Research Award, and the St. Baldrick’s Foundation Scholar Award. Dr. Barrett and his coauthors had no financial disclosures.
Source: Barrett DM et al. AACR 2018, Abstract 1631.