User login
Study reveals misperceptions among AML patients
SAN DIEGO—A study of acute myeloid leukemia (AML) patients has revealed misperceptions about treatment risks and the likelihood of cure.
Investigators surveyed 100 AML patients receiving intensive and non-intensive chemotherapy, as well as the patients’ oncologists.
The results showed that patients tended to overestimate both the risk of dying due to treatment and the likelihood of cure.
These findings were presented at the 2017 Palliative and Supportive Care in Oncology Symposium (abstract 43).
“Patients with AML face very challenging treatment decisions that are often placed upon them within days after being diagnosed,” said study investigator Areej El-Jawahri, MD, of Massachusetts General Hospital in Boston.
“Because they face a grave decision, they need to understand what the risks of treatment are versus the possibility of a cure.”
For this study, Dr El-Jawahri and her colleagues enrolled 50 patients who were receiving intensive care for AML (which usually meant hospitalization for 4 to 6 weeks) and 50 patients who were receiving non-intensive care (often given as outpatient treatment).
The patients’ median age was 71 (range, 60-100), and 92% were white. Six percent of patients had low-risk disease, 48% had intermediate-risk, and 46% had high-risk disease.
Within 3 days of starting treatment, both the patients and their physicians were given a questionnaire to assess how they perceived the likelihood of the patient dying from treatment.
One month later, patients and physicians completed a follow-up questionnaire to assess perceptions of patient prognosis. Within that time frame, most patients received laboratory results that more definitively established the type and stage of cancer.
At 24 weeks, the investigators asked patients if they had discussed their end-of-life wishes with their oncologists.
Results
Initially, most of the patient population (91.3%) thought it was “somewhat” or “extremely” likely they would die from their treatment. However, only 22% of treating oncologists said the same.
One month later, a majority of patients in both treatment groups thought it was “somewhat” or “extremely” likely they would be cured of their AML.
Specifically, 82.1% of patients receiving non-intensive chemotherapy said it was “somewhat” or “extremely” likely they would be cured, while 10% of their oncologists said the same.
Meanwhile, 97.6% of patients receiving intensive chemotherapy said it was “somewhat” or “extremely” likely they would be cured, and 42% of their oncologists said the same.
Overall, 77.8% of patients said they had not discussed their end-of-life wishes with their oncologists at 24 weeks.
“There were several very important factors we were not able to capture in our study, including what was actually discussed between patients and their oncologists and whether patients simply misunderstood or misheard the information conveyed to them,” Dr El-Jawahri said.
“Perhaps most importantly, we did not audio-record the discussions between the patients and their physicians, which could provide additional details regarding barriers to accurate prognostic understanding in these conversations.”
Related research and next steps
Prior to this study, Dr El-Jawahri and her colleagues had looked at similar perceptions in patients with solid tumor malignancies as well as in patients with hematologic malignancies who were receiving hematopoietic stem cell transplants.
The gaps in perception of treatment risk and cure for patients compared to their physicians were not as large in those cases as in the AML patients in this study. The investigators attribute this to higher levels of distress seen in AML patients due to the urgency of their treatment decisions.
Dr El-Jawahri and her colleagues have found that early consideration of palliative care in a treatment plan for patients with solid tumors improves patients’ understanding of the prognosis. The team hopes to implement a similar study in patients with AML.
“Clearly, there are important communication gaps between oncologists and their patients,” Dr El-Jawahri said. “We need to find ways to help physicians do a better job of communicating with their patients, especially in diseases like AML where stress levels are remarkably high.” ![]()
SAN DIEGO—A study of acute myeloid leukemia (AML) patients has revealed misperceptions about treatment risks and the likelihood of cure.
Investigators surveyed 100 AML patients receiving intensive and non-intensive chemotherapy, as well as the patients’ oncologists.
The results showed that patients tended to overestimate both the risk of dying due to treatment and the likelihood of cure.
These findings were presented at the 2017 Palliative and Supportive Care in Oncology Symposium (abstract 43).
“Patients with AML face very challenging treatment decisions that are often placed upon them within days after being diagnosed,” said study investigator Areej El-Jawahri, MD, of Massachusetts General Hospital in Boston.
“Because they face a grave decision, they need to understand what the risks of treatment are versus the possibility of a cure.”
For this study, Dr El-Jawahri and her colleagues enrolled 50 patients who were receiving intensive care for AML (which usually meant hospitalization for 4 to 6 weeks) and 50 patients who were receiving non-intensive care (often given as outpatient treatment).
The patients’ median age was 71 (range, 60-100), and 92% were white. Six percent of patients had low-risk disease, 48% had intermediate-risk, and 46% had high-risk disease.
Within 3 days of starting treatment, both the patients and their physicians were given a questionnaire to assess how they perceived the likelihood of the patient dying from treatment.
One month later, patients and physicians completed a follow-up questionnaire to assess perceptions of patient prognosis. Within that time frame, most patients received laboratory results that more definitively established the type and stage of cancer.
At 24 weeks, the investigators asked patients if they had discussed their end-of-life wishes with their oncologists.
Results
Initially, most of the patient population (91.3%) thought it was “somewhat” or “extremely” likely they would die from their treatment. However, only 22% of treating oncologists said the same.
One month later, a majority of patients in both treatment groups thought it was “somewhat” or “extremely” likely they would be cured of their AML.
Specifically, 82.1% of patients receiving non-intensive chemotherapy said it was “somewhat” or “extremely” likely they would be cured, while 10% of their oncologists said the same.
Meanwhile, 97.6% of patients receiving intensive chemotherapy said it was “somewhat” or “extremely” likely they would be cured, and 42% of their oncologists said the same.
Overall, 77.8% of patients said they had not discussed their end-of-life wishes with their oncologists at 24 weeks.
“There were several very important factors we were not able to capture in our study, including what was actually discussed between patients and their oncologists and whether patients simply misunderstood or misheard the information conveyed to them,” Dr El-Jawahri said.
“Perhaps most importantly, we did not audio-record the discussions between the patients and their physicians, which could provide additional details regarding barriers to accurate prognostic understanding in these conversations.”
Related research and next steps
Prior to this study, Dr El-Jawahri and her colleagues had looked at similar perceptions in patients with solid tumor malignancies as well as in patients with hematologic malignancies who were receiving hematopoietic stem cell transplants.
The gaps in perception of treatment risk and cure for patients compared to their physicians were not as large in those cases as in the AML patients in this study. The investigators attribute this to higher levels of distress seen in AML patients due to the urgency of their treatment decisions.
Dr El-Jawahri and her colleagues have found that early consideration of palliative care in a treatment plan for patients with solid tumors improves patients’ understanding of the prognosis. The team hopes to implement a similar study in patients with AML.
“Clearly, there are important communication gaps between oncologists and their patients,” Dr El-Jawahri said. “We need to find ways to help physicians do a better job of communicating with their patients, especially in diseases like AML where stress levels are remarkably high.” ![]()
SAN DIEGO—A study of acute myeloid leukemia (AML) patients has revealed misperceptions about treatment risks and the likelihood of cure.
Investigators surveyed 100 AML patients receiving intensive and non-intensive chemotherapy, as well as the patients’ oncologists.
The results showed that patients tended to overestimate both the risk of dying due to treatment and the likelihood of cure.
These findings were presented at the 2017 Palliative and Supportive Care in Oncology Symposium (abstract 43).
“Patients with AML face very challenging treatment decisions that are often placed upon them within days after being diagnosed,” said study investigator Areej El-Jawahri, MD, of Massachusetts General Hospital in Boston.
“Because they face a grave decision, they need to understand what the risks of treatment are versus the possibility of a cure.”
For this study, Dr El-Jawahri and her colleagues enrolled 50 patients who were receiving intensive care for AML (which usually meant hospitalization for 4 to 6 weeks) and 50 patients who were receiving non-intensive care (often given as outpatient treatment).
The patients’ median age was 71 (range, 60-100), and 92% were white. Six percent of patients had low-risk disease, 48% had intermediate-risk, and 46% had high-risk disease.
Within 3 days of starting treatment, both the patients and their physicians were given a questionnaire to assess how they perceived the likelihood of the patient dying from treatment.
One month later, patients and physicians completed a follow-up questionnaire to assess perceptions of patient prognosis. Within that time frame, most patients received laboratory results that more definitively established the type and stage of cancer.
At 24 weeks, the investigators asked patients if they had discussed their end-of-life wishes with their oncologists.
Results
Initially, most of the patient population (91.3%) thought it was “somewhat” or “extremely” likely they would die from their treatment. However, only 22% of treating oncologists said the same.
One month later, a majority of patients in both treatment groups thought it was “somewhat” or “extremely” likely they would be cured of their AML.
Specifically, 82.1% of patients receiving non-intensive chemotherapy said it was “somewhat” or “extremely” likely they would be cured, while 10% of their oncologists said the same.
Meanwhile, 97.6% of patients receiving intensive chemotherapy said it was “somewhat” or “extremely” likely they would be cured, and 42% of their oncologists said the same.
Overall, 77.8% of patients said they had not discussed their end-of-life wishes with their oncologists at 24 weeks.
“There were several very important factors we were not able to capture in our study, including what was actually discussed between patients and their oncologists and whether patients simply misunderstood or misheard the information conveyed to them,” Dr El-Jawahri said.
“Perhaps most importantly, we did not audio-record the discussions between the patients and their physicians, which could provide additional details regarding barriers to accurate prognostic understanding in these conversations.”
Related research and next steps
Prior to this study, Dr El-Jawahri and her colleagues had looked at similar perceptions in patients with solid tumor malignancies as well as in patients with hematologic malignancies who were receiving hematopoietic stem cell transplants.
The gaps in perception of treatment risk and cure for patients compared to their physicians were not as large in those cases as in the AML patients in this study. The investigators attribute this to higher levels of distress seen in AML patients due to the urgency of their treatment decisions.
Dr El-Jawahri and her colleagues have found that early consideration of palliative care in a treatment plan for patients with solid tumors improves patients’ understanding of the prognosis. The team hopes to implement a similar study in patients with AML.
“Clearly, there are important communication gaps between oncologists and their patients,” Dr El-Jawahri said. “We need to find ways to help physicians do a better job of communicating with their patients, especially in diseases like AML where stress levels are remarkably high.” ![]()
Idelalisib efficacy against CLL tarnished by toxicity
NEW YORK – PI3K inhibitors are highly active against B-cell malignancies, but this class of drugs, led by
Idelalisib is a potent inhibitor of the delta isoform of phosphatidylinositol 3-kinase (PI3K) that in a phase 1 trial was associated at higher dose levels with a median progression-free survival (PFS) of 32 months in patients with CLL who had received a median of five prior lines of therapy, noted Jennifer R. Brown, MD, PhD, director of the CLL center at the Dana-Farber Cancer Institute in Boston.
“This is really a very effective drug. So what’s happened? Why aren’t we using it more?” she asked rhetorically at an international congress on hematologic malignancies.
“This relates to a pattern of toxicities that has becoming increasingly familiar to us,” she added.
There is increasing evidence to suggest that the toxicities associated with idelalisib are immune mediated, indicating both the need for caution among clinicians who think about prescribing the drug, and a potential future use for this and other PI3K inhibitors as immunomodulatory agents, Dr. Brown said.
Registration trial toxicities
Among 760 patients enrolled in trials for the idelalisib registration programs, grade 3 or greater diarrhea and/or colitis and transaminitis each occurred in 14% of patients, rash occurred in 6%, and pneumonitis of any grade was seen in 3%.
Among patients with relapsed disease, transaminitis was often self-limiting and usually resolved when the drug was withheld, and about 75% of patients were successfully restarted on idelalisib at the same or lower dose, Dr. Brown noted.
Rashes, which can occur any time with therapy, were also successfully managed by withholding drug and then rechallenging, with the addition of corticosteroids as necessary.
Patients who developed drug-related pneumonitis were less likely than those with other toxicities to be rechallenged, and most required steroids until the infections resolved.
“The steroid responsiveness of many of these side effects suggested that they were autoimmune,” Dr. Brown said.
Drugs only work when you take them
The toxicities seen with idelalisib have had a marked effect on the use of the drug. In registration trials for idelalisib in combination with rituximab or ofatumumab (Arzerra), each of which had at least 2 years of follow-up, only 22.5% of 369 patients remained on idelalisib, primarily because of toxicities rather than disease progression. The combined 2-year progression in these trials was 13.3% In contrast, 40.7% of patients discontinued idelalisib because of adverse events.
Out to about 7 months, survival rates for patients who discontinued idelalisib because of disease progression or adverse events were roughly similar, but survival for the patients who stopped because of side effects began to plateau out to 2 years, Dr. Brown noted.
As of March 2016, 23.2% of patients who received idelalisib in clinical trials in combination with other agents as second- or third-line therapy had died, compared with 31% of controls, indicating a clear survival benefit with the drug.
“This is probably because the benefit of disease control in that setting overwhelmed the adverse event or infections problem,” she said.
Many of the deaths in registration trials were related to opportunistic infections, including Pneumocystis jiroveci pneumonia, fungal infection, and cytomegalovirus.
“Idelalisib, I think, is a prototypical delta inhibitor with a pattern of immune-mediated toxicity that remains unpredictable and can be severe. We now have pretty good data, based on the Gilead [sponsor] trials, that younger age and less prior therapy predispose to this toxicity,” Dr. Brown said.
Evidence is less robust, but growing, that mutated IGHV and a decrease in regulatory T cells may be also be risk factors for immune-mediated toxicities with idelalisib. Immune modulation with the drug may also account for associated neutropenia, sepsis, and opportunistic infections seen with idelalisib therapy, she added.
So how to use it?
Currently, the best uses for idelalisib and other PI3K inhibitors in CLL appear to be in single-agent therapy in patients with relapsed disease who cannot tolerate a Bruton’s tyrosine kinase (BTK) inhibitor such as ibrutinib (Imbruvica) or in patients whose disease has progressed on a BTK inhibitor.
“Where I think about this drug is in older, more heavily pretreated patients, who are generally at less risk for toxicities, and if they have significant comorbidities that may impact BTK-inhibitor tolerability, usually cardiac,” Dr. Brown said.
Future expansion of PI3K inhibitors in B-cell malignancies may require identifying a biomarker for tolerance, alternative dosing schedules, or identification of an idelalisib/drug X combination that might mitigate the toxicity, she said.
The immune-activation properties of PI3K-delta inhibitors suggests that they might also play a role as antitumor immunomodulatory agents in treatment of both hematologic malignancies and solid tumors, Dr. Brown concluded.
Idelalisib trials were sponsored by Gilead Sciences. Dr. Brown disclosed serving as a consultant for Gilead and other companies.
NEW YORK – PI3K inhibitors are highly active against B-cell malignancies, but this class of drugs, led by
Idelalisib is a potent inhibitor of the delta isoform of phosphatidylinositol 3-kinase (PI3K) that in a phase 1 trial was associated at higher dose levels with a median progression-free survival (PFS) of 32 months in patients with CLL who had received a median of five prior lines of therapy, noted Jennifer R. Brown, MD, PhD, director of the CLL center at the Dana-Farber Cancer Institute in Boston.
“This is really a very effective drug. So what’s happened? Why aren’t we using it more?” she asked rhetorically at an international congress on hematologic malignancies.
“This relates to a pattern of toxicities that has becoming increasingly familiar to us,” she added.
There is increasing evidence to suggest that the toxicities associated with idelalisib are immune mediated, indicating both the need for caution among clinicians who think about prescribing the drug, and a potential future use for this and other PI3K inhibitors as immunomodulatory agents, Dr. Brown said.
Registration trial toxicities
Among 760 patients enrolled in trials for the idelalisib registration programs, grade 3 or greater diarrhea and/or colitis and transaminitis each occurred in 14% of patients, rash occurred in 6%, and pneumonitis of any grade was seen in 3%.
Among patients with relapsed disease, transaminitis was often self-limiting and usually resolved when the drug was withheld, and about 75% of patients were successfully restarted on idelalisib at the same or lower dose, Dr. Brown noted.
Rashes, which can occur any time with therapy, were also successfully managed by withholding drug and then rechallenging, with the addition of corticosteroids as necessary.
Patients who developed drug-related pneumonitis were less likely than those with other toxicities to be rechallenged, and most required steroids until the infections resolved.
“The steroid responsiveness of many of these side effects suggested that they were autoimmune,” Dr. Brown said.
Drugs only work when you take them
The toxicities seen with idelalisib have had a marked effect on the use of the drug. In registration trials for idelalisib in combination with rituximab or ofatumumab (Arzerra), each of which had at least 2 years of follow-up, only 22.5% of 369 patients remained on idelalisib, primarily because of toxicities rather than disease progression. The combined 2-year progression in these trials was 13.3% In contrast, 40.7% of patients discontinued idelalisib because of adverse events.
Out to about 7 months, survival rates for patients who discontinued idelalisib because of disease progression or adverse events were roughly similar, but survival for the patients who stopped because of side effects began to plateau out to 2 years, Dr. Brown noted.
As of March 2016, 23.2% of patients who received idelalisib in clinical trials in combination with other agents as second- or third-line therapy had died, compared with 31% of controls, indicating a clear survival benefit with the drug.
“This is probably because the benefit of disease control in that setting overwhelmed the adverse event or infections problem,” she said.
Many of the deaths in registration trials were related to opportunistic infections, including Pneumocystis jiroveci pneumonia, fungal infection, and cytomegalovirus.
“Idelalisib, I think, is a prototypical delta inhibitor with a pattern of immune-mediated toxicity that remains unpredictable and can be severe. We now have pretty good data, based on the Gilead [sponsor] trials, that younger age and less prior therapy predispose to this toxicity,” Dr. Brown said.
Evidence is less robust, but growing, that mutated IGHV and a decrease in regulatory T cells may be also be risk factors for immune-mediated toxicities with idelalisib. Immune modulation with the drug may also account for associated neutropenia, sepsis, and opportunistic infections seen with idelalisib therapy, she added.
So how to use it?
Currently, the best uses for idelalisib and other PI3K inhibitors in CLL appear to be in single-agent therapy in patients with relapsed disease who cannot tolerate a Bruton’s tyrosine kinase (BTK) inhibitor such as ibrutinib (Imbruvica) or in patients whose disease has progressed on a BTK inhibitor.
“Where I think about this drug is in older, more heavily pretreated patients, who are generally at less risk for toxicities, and if they have significant comorbidities that may impact BTK-inhibitor tolerability, usually cardiac,” Dr. Brown said.
Future expansion of PI3K inhibitors in B-cell malignancies may require identifying a biomarker for tolerance, alternative dosing schedules, or identification of an idelalisib/drug X combination that might mitigate the toxicity, she said.
The immune-activation properties of PI3K-delta inhibitors suggests that they might also play a role as antitumor immunomodulatory agents in treatment of both hematologic malignancies and solid tumors, Dr. Brown concluded.
Idelalisib trials were sponsored by Gilead Sciences. Dr. Brown disclosed serving as a consultant for Gilead and other companies.
NEW YORK – PI3K inhibitors are highly active against B-cell malignancies, but this class of drugs, led by
Idelalisib is a potent inhibitor of the delta isoform of phosphatidylinositol 3-kinase (PI3K) that in a phase 1 trial was associated at higher dose levels with a median progression-free survival (PFS) of 32 months in patients with CLL who had received a median of five prior lines of therapy, noted Jennifer R. Brown, MD, PhD, director of the CLL center at the Dana-Farber Cancer Institute in Boston.
“This is really a very effective drug. So what’s happened? Why aren’t we using it more?” she asked rhetorically at an international congress on hematologic malignancies.
“This relates to a pattern of toxicities that has becoming increasingly familiar to us,” she added.
There is increasing evidence to suggest that the toxicities associated with idelalisib are immune mediated, indicating both the need for caution among clinicians who think about prescribing the drug, and a potential future use for this and other PI3K inhibitors as immunomodulatory agents, Dr. Brown said.
Registration trial toxicities
Among 760 patients enrolled in trials for the idelalisib registration programs, grade 3 or greater diarrhea and/or colitis and transaminitis each occurred in 14% of patients, rash occurred in 6%, and pneumonitis of any grade was seen in 3%.
Among patients with relapsed disease, transaminitis was often self-limiting and usually resolved when the drug was withheld, and about 75% of patients were successfully restarted on idelalisib at the same or lower dose, Dr. Brown noted.
Rashes, which can occur any time with therapy, were also successfully managed by withholding drug and then rechallenging, with the addition of corticosteroids as necessary.
Patients who developed drug-related pneumonitis were less likely than those with other toxicities to be rechallenged, and most required steroids until the infections resolved.
“The steroid responsiveness of many of these side effects suggested that they were autoimmune,” Dr. Brown said.
Drugs only work when you take them
The toxicities seen with idelalisib have had a marked effect on the use of the drug. In registration trials for idelalisib in combination with rituximab or ofatumumab (Arzerra), each of which had at least 2 years of follow-up, only 22.5% of 369 patients remained on idelalisib, primarily because of toxicities rather than disease progression. The combined 2-year progression in these trials was 13.3% In contrast, 40.7% of patients discontinued idelalisib because of adverse events.
Out to about 7 months, survival rates for patients who discontinued idelalisib because of disease progression or adverse events were roughly similar, but survival for the patients who stopped because of side effects began to plateau out to 2 years, Dr. Brown noted.
As of March 2016, 23.2% of patients who received idelalisib in clinical trials in combination with other agents as second- or third-line therapy had died, compared with 31% of controls, indicating a clear survival benefit with the drug.
“This is probably because the benefit of disease control in that setting overwhelmed the adverse event or infections problem,” she said.
Many of the deaths in registration trials were related to opportunistic infections, including Pneumocystis jiroveci pneumonia, fungal infection, and cytomegalovirus.
“Idelalisib, I think, is a prototypical delta inhibitor with a pattern of immune-mediated toxicity that remains unpredictable and can be severe. We now have pretty good data, based on the Gilead [sponsor] trials, that younger age and less prior therapy predispose to this toxicity,” Dr. Brown said.
Evidence is less robust, but growing, that mutated IGHV and a decrease in regulatory T cells may be also be risk factors for immune-mediated toxicities with idelalisib. Immune modulation with the drug may also account for associated neutropenia, sepsis, and opportunistic infections seen with idelalisib therapy, she added.
So how to use it?
Currently, the best uses for idelalisib and other PI3K inhibitors in CLL appear to be in single-agent therapy in patients with relapsed disease who cannot tolerate a Bruton’s tyrosine kinase (BTK) inhibitor such as ibrutinib (Imbruvica) or in patients whose disease has progressed on a BTK inhibitor.
“Where I think about this drug is in older, more heavily pretreated patients, who are generally at less risk for toxicities, and if they have significant comorbidities that may impact BTK-inhibitor tolerability, usually cardiac,” Dr. Brown said.
Future expansion of PI3K inhibitors in B-cell malignancies may require identifying a biomarker for tolerance, alternative dosing schedules, or identification of an idelalisib/drug X combination that might mitigate the toxicity, she said.
The immune-activation properties of PI3K-delta inhibitors suggests that they might also play a role as antitumor immunomodulatory agents in treatment of both hematologic malignancies and solid tumors, Dr. Brown concluded.
Idelalisib trials were sponsored by Gilead Sciences. Dr. Brown disclosed serving as a consultant for Gilead and other companies.
EXPERT ANALYSIS FROM LYMPHOMA & MYELOMA
Deep remission or long-term control? Choice is key in early CLL
SAN FRANCISCO – Pursue a deep remission that allows a patient to stay treatment free for some period of time, or go for long-term disease control that might not allow for a drug holiday?
It’s a key decision facing physicians in the frontline setting of chronic lymphocytic leukemia, William Wierda, MD, PhD, said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
For those with del(17p) or TP53 mutations, it’s probably best to aim for durable disease control with ibrutinib or high-dose methylprednisolone, plus an anti-CD20 monoclonal antibody, explained Dr. Wierda, medical director of the department of leukemia at MD Anderson Cancer Center, Houston.
The decision is more uncertain for those who are older or frail, he added.
“This is really where we need to select the option based on what our preference is, what our patient’s preference is, and have an understanding of the durability and toxicities with remission with the oral agent versus the toxicities and responses with regard to the chemoimmunotherapy regimens,” Dr. Wierda explained.
For those who are younger and fit, a chemoimmunotherapy regimen likely makes the most sense for those with a mutated IgHV gene, he said, because those patients have been shown to have a better prognosis on the fludarabine-cyclophosphamide-rituximab (FCR) combination.
Those with an unmutated IgHV gene probably should be approached differently, he added. “I know if they get FCR treatment, they will eventually relapse and progress. So, saving chemoimmunotherapy for later is an important endpoint.”
For relapsed patients who’ve had prior chemoimmunotherapy or who have del(17p) or TP53 mutations, options include ibrutinib, venetoclax with or without rituximab, idelalisib with or without rituximab, high-dose methylprednisolone plus an anti-CD20 monoclonal antibody, or lenalidomide plus an anti-CD20 monoclonal antibody.
For patients who’ve already had prior experience with a BTK inhibitor such as ibrutinib, Dr. Wierda suggested venetoclax, idelalisib with or without rituximab, chemoimmunotherapy if they’ve had no prior treatment, or high-dose methylprednisolone with an anti-CD20 monoclonal antibody.
It’s important to keep in mind ibrutinib’s effectiveness in that setting, Dr. Wierda noted. “You can effectively salvage patients with ibrutinib nearly as effectively as you can in the frontline setting.”
A recent study found that, for patients refractory to a kinase inhibitor, switching to a different kinase inhibitor was better than chemoimmunotherapy combinations. Researchers also found that using venetoclax after ibrutinib failure could be better than idelalisib (Ann Oncol. 2017 May 1;28[5]:1050-6)
Trials underway are testing first-line chemoimmunotherapy regimens to reach minimal residual disease-negativity, Dr. Wierda said, and examining combinations in the sequencing of small-molecule inhibitors for patients who have the unmutated IgHV gene.
“We’re also looking at consolidation strategies and have a definite interest in making progress for Richter’s transformation,” he added, an uncommon phenomenon that, in most cases, involves slow-growing CLL becoming aggressive diffuse large B-cell lymphoma. “We don’t know as much as we should know about it, and we have very few effective therapies for it.”
Dr. Wierda reported financial relationships with AbbVie, Celgene, Genentech, Merck, Novartis, Roche, and other companies.
SAN FRANCISCO – Pursue a deep remission that allows a patient to stay treatment free for some period of time, or go for long-term disease control that might not allow for a drug holiday?
It’s a key decision facing physicians in the frontline setting of chronic lymphocytic leukemia, William Wierda, MD, PhD, said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
For those with del(17p) or TP53 mutations, it’s probably best to aim for durable disease control with ibrutinib or high-dose methylprednisolone, plus an anti-CD20 monoclonal antibody, explained Dr. Wierda, medical director of the department of leukemia at MD Anderson Cancer Center, Houston.
The decision is more uncertain for those who are older or frail, he added.
“This is really where we need to select the option based on what our preference is, what our patient’s preference is, and have an understanding of the durability and toxicities with remission with the oral agent versus the toxicities and responses with regard to the chemoimmunotherapy regimens,” Dr. Wierda explained.
For those who are younger and fit, a chemoimmunotherapy regimen likely makes the most sense for those with a mutated IgHV gene, he said, because those patients have been shown to have a better prognosis on the fludarabine-cyclophosphamide-rituximab (FCR) combination.
Those with an unmutated IgHV gene probably should be approached differently, he added. “I know if they get FCR treatment, they will eventually relapse and progress. So, saving chemoimmunotherapy for later is an important endpoint.”
For relapsed patients who’ve had prior chemoimmunotherapy or who have del(17p) or TP53 mutations, options include ibrutinib, venetoclax with or without rituximab, idelalisib with or without rituximab, high-dose methylprednisolone plus an anti-CD20 monoclonal antibody, or lenalidomide plus an anti-CD20 monoclonal antibody.
For patients who’ve already had prior experience with a BTK inhibitor such as ibrutinib, Dr. Wierda suggested venetoclax, idelalisib with or without rituximab, chemoimmunotherapy if they’ve had no prior treatment, or high-dose methylprednisolone with an anti-CD20 monoclonal antibody.
It’s important to keep in mind ibrutinib’s effectiveness in that setting, Dr. Wierda noted. “You can effectively salvage patients with ibrutinib nearly as effectively as you can in the frontline setting.”
A recent study found that, for patients refractory to a kinase inhibitor, switching to a different kinase inhibitor was better than chemoimmunotherapy combinations. Researchers also found that using venetoclax after ibrutinib failure could be better than idelalisib (Ann Oncol. 2017 May 1;28[5]:1050-6)
Trials underway are testing first-line chemoimmunotherapy regimens to reach minimal residual disease-negativity, Dr. Wierda said, and examining combinations in the sequencing of small-molecule inhibitors for patients who have the unmutated IgHV gene.
“We’re also looking at consolidation strategies and have a definite interest in making progress for Richter’s transformation,” he added, an uncommon phenomenon that, in most cases, involves slow-growing CLL becoming aggressive diffuse large B-cell lymphoma. “We don’t know as much as we should know about it, and we have very few effective therapies for it.”
Dr. Wierda reported financial relationships with AbbVie, Celgene, Genentech, Merck, Novartis, Roche, and other companies.
SAN FRANCISCO – Pursue a deep remission that allows a patient to stay treatment free for some period of time, or go for long-term disease control that might not allow for a drug holiday?
It’s a key decision facing physicians in the frontline setting of chronic lymphocytic leukemia, William Wierda, MD, PhD, said at the annual congress on Hematologic Malignancies held by the National Comprehensive Cancer Network.
For those with del(17p) or TP53 mutations, it’s probably best to aim for durable disease control with ibrutinib or high-dose methylprednisolone, plus an anti-CD20 monoclonal antibody, explained Dr. Wierda, medical director of the department of leukemia at MD Anderson Cancer Center, Houston.
The decision is more uncertain for those who are older or frail, he added.
“This is really where we need to select the option based on what our preference is, what our patient’s preference is, and have an understanding of the durability and toxicities with remission with the oral agent versus the toxicities and responses with regard to the chemoimmunotherapy regimens,” Dr. Wierda explained.
For those who are younger and fit, a chemoimmunotherapy regimen likely makes the most sense for those with a mutated IgHV gene, he said, because those patients have been shown to have a better prognosis on the fludarabine-cyclophosphamide-rituximab (FCR) combination.
Those with an unmutated IgHV gene probably should be approached differently, he added. “I know if they get FCR treatment, they will eventually relapse and progress. So, saving chemoimmunotherapy for later is an important endpoint.”
For relapsed patients who’ve had prior chemoimmunotherapy or who have del(17p) or TP53 mutations, options include ibrutinib, venetoclax with or without rituximab, idelalisib with or without rituximab, high-dose methylprednisolone plus an anti-CD20 monoclonal antibody, or lenalidomide plus an anti-CD20 monoclonal antibody.
For patients who’ve already had prior experience with a BTK inhibitor such as ibrutinib, Dr. Wierda suggested venetoclax, idelalisib with or without rituximab, chemoimmunotherapy if they’ve had no prior treatment, or high-dose methylprednisolone with an anti-CD20 monoclonal antibody.
It’s important to keep in mind ibrutinib’s effectiveness in that setting, Dr. Wierda noted. “You can effectively salvage patients with ibrutinib nearly as effectively as you can in the frontline setting.”
A recent study found that, for patients refractory to a kinase inhibitor, switching to a different kinase inhibitor was better than chemoimmunotherapy combinations. Researchers also found that using venetoclax after ibrutinib failure could be better than idelalisib (Ann Oncol. 2017 May 1;28[5]:1050-6)
Trials underway are testing first-line chemoimmunotherapy regimens to reach minimal residual disease-negativity, Dr. Wierda said, and examining combinations in the sequencing of small-molecule inhibitors for patients who have the unmutated IgHV gene.
“We’re also looking at consolidation strategies and have a definite interest in making progress for Richter’s transformation,” he added, an uncommon phenomenon that, in most cases, involves slow-growing CLL becoming aggressive diffuse large B-cell lymphoma. “We don’t know as much as we should know about it, and we have very few effective therapies for it.”
Dr. Wierda reported financial relationships with AbbVie, Celgene, Genentech, Merck, Novartis, Roche, and other companies.
AT NCCN HEMATOLOGIC MALIGNANCIES CONGRESS
Targeting key pathways to eradicate AML
Targeting two pathways simultaneously—one critical for oncogenesis and one essential for cell survival—may be an effective strategy for treating acute myeloid leukemia (AML), according to researchers.
The team studied mouse models of FLT3-ITD AML and found an inhibitor targeting the FLT3 pathway was largely effective against the disease.
However, targeting the BCL-2 pathway as well proved even more effective, completely eliminating AML in most cases.
Fumihiko Ishikawa, MD, PhD, of RIKEN Center for Integrative Medical Sciences in Yokohama, Kanagawa, Japan, and his colleagues described this work in Science Translational Medicine.
The researchers noted that mutations observed in AML patients have also been observed in elderly people without AML. So the team set out to determine which mutations actually contribute to the disease.
The researchers obtained bone marrow or blood samples from patients with FLT3-ITD AML and transplanted different cellular populations from each individual into mice. The team then examined how the cells behaved.
They were surprised to find that cells with similar surface marker profiles behaved differently. Therefore, the team used single-cell genomic sequencing to correlate mutational profiles with malignant potential.
The researchers said their results suggest that FLT3-ITD is “a critical trigger for leukemia initiation,” and it cooperates with accumulated mutations in DNMT3A, TET2, NPM1, and/or WT1.
The team went on to treat the FLT3-ITD AML mice with RK-20449, a FLT3/HCK inhibitor, and they observed “significant responses.”
In fact, RK-20449 eradicated leukemia originating from 5 different patients. The recipient mice experienced complete elimination of AML cells, in spite of the fact that they also carried mutations not directly targeted by RK-20449.
However, the researchers also noted the presence of RK-20449-resistant AML cells in some mice. The team therefore theorized that co-inhibition of an antiapoptotic signal—BCL-2—might remedy this.
So they treated resistant mice with the BCL-2 inhibitor venetoclax as well as RK-20449. The combination produced responses in all mice treated and completely eliminated AML cells in 9 of 12 cases.
“This shows that determining which of the mutations in a diverse landscape are critical in leukemia onset and which of the pathways are critical for therapeutic resistance in leukemia, and simultaneously targeting those pathways, is an encouraging way to treat difficult cancers such as AML,” Dr Ishikawa said. ![]()
Targeting two pathways simultaneously—one critical for oncogenesis and one essential for cell survival—may be an effective strategy for treating acute myeloid leukemia (AML), according to researchers.
The team studied mouse models of FLT3-ITD AML and found an inhibitor targeting the FLT3 pathway was largely effective against the disease.
However, targeting the BCL-2 pathway as well proved even more effective, completely eliminating AML in most cases.
Fumihiko Ishikawa, MD, PhD, of RIKEN Center for Integrative Medical Sciences in Yokohama, Kanagawa, Japan, and his colleagues described this work in Science Translational Medicine.
The researchers noted that mutations observed in AML patients have also been observed in elderly people without AML. So the team set out to determine which mutations actually contribute to the disease.
The researchers obtained bone marrow or blood samples from patients with FLT3-ITD AML and transplanted different cellular populations from each individual into mice. The team then examined how the cells behaved.
They were surprised to find that cells with similar surface marker profiles behaved differently. Therefore, the team used single-cell genomic sequencing to correlate mutational profiles with malignant potential.
The researchers said their results suggest that FLT3-ITD is “a critical trigger for leukemia initiation,” and it cooperates with accumulated mutations in DNMT3A, TET2, NPM1, and/or WT1.
The team went on to treat the FLT3-ITD AML mice with RK-20449, a FLT3/HCK inhibitor, and they observed “significant responses.”
In fact, RK-20449 eradicated leukemia originating from 5 different patients. The recipient mice experienced complete elimination of AML cells, in spite of the fact that they also carried mutations not directly targeted by RK-20449.
However, the researchers also noted the presence of RK-20449-resistant AML cells in some mice. The team therefore theorized that co-inhibition of an antiapoptotic signal—BCL-2—might remedy this.
So they treated resistant mice with the BCL-2 inhibitor venetoclax as well as RK-20449. The combination produced responses in all mice treated and completely eliminated AML cells in 9 of 12 cases.
“This shows that determining which of the mutations in a diverse landscape are critical in leukemia onset and which of the pathways are critical for therapeutic resistance in leukemia, and simultaneously targeting those pathways, is an encouraging way to treat difficult cancers such as AML,” Dr Ishikawa said. ![]()
Targeting two pathways simultaneously—one critical for oncogenesis and one essential for cell survival—may be an effective strategy for treating acute myeloid leukemia (AML), according to researchers.
The team studied mouse models of FLT3-ITD AML and found an inhibitor targeting the FLT3 pathway was largely effective against the disease.
However, targeting the BCL-2 pathway as well proved even more effective, completely eliminating AML in most cases.
Fumihiko Ishikawa, MD, PhD, of RIKEN Center for Integrative Medical Sciences in Yokohama, Kanagawa, Japan, and his colleagues described this work in Science Translational Medicine.
The researchers noted that mutations observed in AML patients have also been observed in elderly people without AML. So the team set out to determine which mutations actually contribute to the disease.
The researchers obtained bone marrow or blood samples from patients with FLT3-ITD AML and transplanted different cellular populations from each individual into mice. The team then examined how the cells behaved.
They were surprised to find that cells with similar surface marker profiles behaved differently. Therefore, the team used single-cell genomic sequencing to correlate mutational profiles with malignant potential.
The researchers said their results suggest that FLT3-ITD is “a critical trigger for leukemia initiation,” and it cooperates with accumulated mutations in DNMT3A, TET2, NPM1, and/or WT1.
The team went on to treat the FLT3-ITD AML mice with RK-20449, a FLT3/HCK inhibitor, and they observed “significant responses.”
In fact, RK-20449 eradicated leukemia originating from 5 different patients. The recipient mice experienced complete elimination of AML cells, in spite of the fact that they also carried mutations not directly targeted by RK-20449.
However, the researchers also noted the presence of RK-20449-resistant AML cells in some mice. The team therefore theorized that co-inhibition of an antiapoptotic signal—BCL-2—might remedy this.
So they treated resistant mice with the BCL-2 inhibitor venetoclax as well as RK-20449. The combination produced responses in all mice treated and completely eliminated AML cells in 9 of 12 cases.
“This shows that determining which of the mutations in a diverse landscape are critical in leukemia onset and which of the pathways are critical for therapeutic resistance in leukemia, and simultaneously targeting those pathways, is an encouraging way to treat difficult cancers such as AML,” Dr Ishikawa said. ![]()
FDA approves IV formulation of drug for CINV
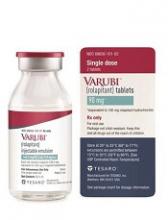
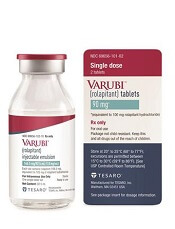
The US Food and Drug Administration (FDA) has approved an intravenous (IV) formulation of rolapitant (VARUBI®) for the same indication as the oral formulation.
This means IV rolapitant is now FDA-approved for use in combination with other antiemetic agents to prevent delayed nausea and vomiting associated with initial and repeat courses of emetogenic chemotherapy in adults with cancer.
TESARO Inc., makers of rolapitant, said the US commercial launch of IV rolapitant is planned for November.
Rolapitant is a selective and competitive antagonist of human substance P/neurokinin 1 receptors, which play an important role in the delayed phase of chemotherapy-induced nausea and vomiting (CINV).
IV rolapitant features a ready-to-use, single-dose vial for administration. It does not require refrigerated storage or mixing.
The recommended dose of IV rolapitant is 166.5 mg, administered over 30 minutes. The drug is to be administered up to 2 hours before chemotherapy administration in combination with a 5-HT3 receptor antagonist and dexamethasone.
The full prescribing information for IV rolapitant is available at www.varubirx.com.
Bioequivalence trial
Results from a bioequivalence trial suggest the IV and oral formulations of rolapitant are comparable.
The study was conducted in healthy volunteers. Subjects were randomized to receive a single dose of IV rolapitant at 166.5 mg administered over 30 minutes (n=61) or oral rolapitant at 180 mg (n=62).
The primary endpoint was bioequivalence, and the 166.5 mg IV infusion of rolapitant met bioequivalence criteria.
Researchers said the safety profile of IV rolapitant was largely consistent with that of oral rolapitant, although infusion-site reactions were observed with the IV formulation. These included the sensation of warmth, abdominal pain, dizziness, and paresthesia.
These results were recently published in The Journal of Clinical Pharmacology.
Oral rolapitant trials
Two phase 3 trials showed that oral rolapitant, in combination with a 5-HT3 receptor antagonist and dexamethasone, was well tolerated and more effective than active control in preventing CINV after highly emetogenic chemotherapy.
Results from these trials (NCT01499849 and NCT01500213) were published in a single article in The Lancet Oncology.
A third phase 3 trial showed that oral rolapitant, in combination with a 5-HT3 receptor antagonist and dexamethasone, was well tolerated and more effective than active control in preventing CINV after moderately emetogenic chemotherapy.
Results from this trial (NCT01500226) were also published in The Lancet Oncology. ![]()
The US Food and Drug Administration (FDA) has approved an intravenous (IV) formulation of rolapitant (VARUBI®) for the same indication as the oral formulation.
This means IV rolapitant is now FDA-approved for use in combination with other antiemetic agents to prevent delayed nausea and vomiting associated with initial and repeat courses of emetogenic chemotherapy in adults with cancer.
TESARO Inc., makers of rolapitant, said the US commercial launch of IV rolapitant is planned for November.
Rolapitant is a selective and competitive antagonist of human substance P/neurokinin 1 receptors, which play an important role in the delayed phase of chemotherapy-induced nausea and vomiting (CINV).
IV rolapitant features a ready-to-use, single-dose vial for administration. It does not require refrigerated storage or mixing.
The recommended dose of IV rolapitant is 166.5 mg, administered over 30 minutes. The drug is to be administered up to 2 hours before chemotherapy administration in combination with a 5-HT3 receptor antagonist and dexamethasone.
The full prescribing information for IV rolapitant is available at www.varubirx.com.
Bioequivalence trial
Results from a bioequivalence trial suggest the IV and oral formulations of rolapitant are comparable.
The study was conducted in healthy volunteers. Subjects were randomized to receive a single dose of IV rolapitant at 166.5 mg administered over 30 minutes (n=61) or oral rolapitant at 180 mg (n=62).
The primary endpoint was bioequivalence, and the 166.5 mg IV infusion of rolapitant met bioequivalence criteria.
Researchers said the safety profile of IV rolapitant was largely consistent with that of oral rolapitant, although infusion-site reactions were observed with the IV formulation. These included the sensation of warmth, abdominal pain, dizziness, and paresthesia.
These results were recently published in The Journal of Clinical Pharmacology.
Oral rolapitant trials
Two phase 3 trials showed that oral rolapitant, in combination with a 5-HT3 receptor antagonist and dexamethasone, was well tolerated and more effective than active control in preventing CINV after highly emetogenic chemotherapy.
Results from these trials (NCT01499849 and NCT01500213) were published in a single article in The Lancet Oncology.
A third phase 3 trial showed that oral rolapitant, in combination with a 5-HT3 receptor antagonist and dexamethasone, was well tolerated and more effective than active control in preventing CINV after moderately emetogenic chemotherapy.
Results from this trial (NCT01500226) were also published in The Lancet Oncology. ![]()
The US Food and Drug Administration (FDA) has approved an intravenous (IV) formulation of rolapitant (VARUBI®) for the same indication as the oral formulation.
This means IV rolapitant is now FDA-approved for use in combination with other antiemetic agents to prevent delayed nausea and vomiting associated with initial and repeat courses of emetogenic chemotherapy in adults with cancer.
TESARO Inc., makers of rolapitant, said the US commercial launch of IV rolapitant is planned for November.
Rolapitant is a selective and competitive antagonist of human substance P/neurokinin 1 receptors, which play an important role in the delayed phase of chemotherapy-induced nausea and vomiting (CINV).
IV rolapitant features a ready-to-use, single-dose vial for administration. It does not require refrigerated storage or mixing.
The recommended dose of IV rolapitant is 166.5 mg, administered over 30 minutes. The drug is to be administered up to 2 hours before chemotherapy administration in combination with a 5-HT3 receptor antagonist and dexamethasone.
The full prescribing information for IV rolapitant is available at www.varubirx.com.
Bioequivalence trial
Results from a bioequivalence trial suggest the IV and oral formulations of rolapitant are comparable.
The study was conducted in healthy volunteers. Subjects were randomized to receive a single dose of IV rolapitant at 166.5 mg administered over 30 minutes (n=61) or oral rolapitant at 180 mg (n=62).
The primary endpoint was bioequivalence, and the 166.5 mg IV infusion of rolapitant met bioequivalence criteria.
Researchers said the safety profile of IV rolapitant was largely consistent with that of oral rolapitant, although infusion-site reactions were observed with the IV formulation. These included the sensation of warmth, abdominal pain, dizziness, and paresthesia.
These results were recently published in The Journal of Clinical Pharmacology.
Oral rolapitant trials
Two phase 3 trials showed that oral rolapitant, in combination with a 5-HT3 receptor antagonist and dexamethasone, was well tolerated and more effective than active control in preventing CINV after highly emetogenic chemotherapy.
Results from these trials (NCT01499849 and NCT01500213) were published in a single article in The Lancet Oncology.
A third phase 3 trial showed that oral rolapitant, in combination with a 5-HT3 receptor antagonist and dexamethasone, was well tolerated and more effective than active control in preventing CINV after moderately emetogenic chemotherapy.
Results from this trial (NCT01500226) were also published in The Lancet Oncology. ![]()
Overcoming resistance to proteasome inhibitors
Preclinical research has revealed a potential method of overcoming resistance to proteasome inhibitors.
By studying a rare genetic disease known as NGLY1 deficiency, researchers have gained new understanding of resistance to proteasome inhibitors.
The team found that treatment with a NGLY1 inhibitor can enhance the activity of the proteasome inhibitor carfilzomib against multiple myeloma (MM) and T-cell acute lymphoblastic leukemia (T-ALL).
Carolyn Bertozzi, PhD, of Stanford University in California, and her colleagues reported these findings in ACS Central Science.
Previous studies have suggested that proteasome inhibitor resistance could be linked to a protein called Nrf1. When proteasome inhibitors swing into action, Nrf1 is spurred into overdrive to restore cancer cells’ normal activities and keep them alive.
Researchers theorized that, if they could block Nrf1, they might be able to overcome proteasome inhibitor resistance.
Through studying NGLY1 deficiency, Dr Bertozzi and her colleagues may have hit upon an approach to do just that.
The researchers were investigating how lacking NGLY1 causes a host of debilitating symptoms, and they found that NGLY1 is responsible for activating Nrf1.
Further testing revealed that inhibiting NGLY1 eliminated interference from Nrf1 and enhanced the cytotoxicity of carfilzomib in MM and T-ALL cell lines.
The researchers treated the MM cell lines U266 and H929 with the NGLY1 inhibitor, known as WRR139, and carfilzomib and observed a significant decrease in cell survival when compared to treatment with carfilzomib alone. The team observed the same results when testing the T-ALL Jurkat cell line.
The addition of WRR139 resulted in a 2.6-fold reduction in carfilzomib’s LD50 for U266, a 2.0-fold reduction for H929, and a 1.5-fold reduction for Jurkat cells.
The researchers said these findings hold promise for the development of combination therapies for hematologic malignancies. ![]()
Preclinical research has revealed a potential method of overcoming resistance to proteasome inhibitors.
By studying a rare genetic disease known as NGLY1 deficiency, researchers have gained new understanding of resistance to proteasome inhibitors.
The team found that treatment with a NGLY1 inhibitor can enhance the activity of the proteasome inhibitor carfilzomib against multiple myeloma (MM) and T-cell acute lymphoblastic leukemia (T-ALL).
Carolyn Bertozzi, PhD, of Stanford University in California, and her colleagues reported these findings in ACS Central Science.
Previous studies have suggested that proteasome inhibitor resistance could be linked to a protein called Nrf1. When proteasome inhibitors swing into action, Nrf1 is spurred into overdrive to restore cancer cells’ normal activities and keep them alive.
Researchers theorized that, if they could block Nrf1, they might be able to overcome proteasome inhibitor resistance.
Through studying NGLY1 deficiency, Dr Bertozzi and her colleagues may have hit upon an approach to do just that.
The researchers were investigating how lacking NGLY1 causes a host of debilitating symptoms, and they found that NGLY1 is responsible for activating Nrf1.
Further testing revealed that inhibiting NGLY1 eliminated interference from Nrf1 and enhanced the cytotoxicity of carfilzomib in MM and T-ALL cell lines.
The researchers treated the MM cell lines U266 and H929 with the NGLY1 inhibitor, known as WRR139, and carfilzomib and observed a significant decrease in cell survival when compared to treatment with carfilzomib alone. The team observed the same results when testing the T-ALL Jurkat cell line.
The addition of WRR139 resulted in a 2.6-fold reduction in carfilzomib’s LD50 for U266, a 2.0-fold reduction for H929, and a 1.5-fold reduction for Jurkat cells.
The researchers said these findings hold promise for the development of combination therapies for hematologic malignancies. ![]()
Preclinical research has revealed a potential method of overcoming resistance to proteasome inhibitors.
By studying a rare genetic disease known as NGLY1 deficiency, researchers have gained new understanding of resistance to proteasome inhibitors.
The team found that treatment with a NGLY1 inhibitor can enhance the activity of the proteasome inhibitor carfilzomib against multiple myeloma (MM) and T-cell acute lymphoblastic leukemia (T-ALL).
Carolyn Bertozzi, PhD, of Stanford University in California, and her colleagues reported these findings in ACS Central Science.
Previous studies have suggested that proteasome inhibitor resistance could be linked to a protein called Nrf1. When proteasome inhibitors swing into action, Nrf1 is spurred into overdrive to restore cancer cells’ normal activities and keep them alive.
Researchers theorized that, if they could block Nrf1, they might be able to overcome proteasome inhibitor resistance.
Through studying NGLY1 deficiency, Dr Bertozzi and her colleagues may have hit upon an approach to do just that.
The researchers were investigating how lacking NGLY1 causes a host of debilitating symptoms, and they found that NGLY1 is responsible for activating Nrf1.
Further testing revealed that inhibiting NGLY1 eliminated interference from Nrf1 and enhanced the cytotoxicity of carfilzomib in MM and T-ALL cell lines.
The researchers treated the MM cell lines U266 and H929 with the NGLY1 inhibitor, known as WRR139, and carfilzomib and observed a significant decrease in cell survival when compared to treatment with carfilzomib alone. The team observed the same results when testing the T-ALL Jurkat cell line.
The addition of WRR139 resulted in a 2.6-fold reduction in carfilzomib’s LD50 for U266, a 2.0-fold reduction for H929, and a 1.5-fold reduction for Jurkat cells.
The researchers said these findings hold promise for the development of combination therapies for hematologic malignancies. ![]()
‘Year of AML’ just the beginning, expert says
SAN FRANCISCO – After years of stagnation in the field of acute myeloid leukemia – with most standard therapies developed in the 1970s – times are changing, Bruno Medeiros, MD, said at the annual congress on hematologic malignancies held by the National Comprehensive Cancer Network.
“2017 is the year of AML,” he said. Four new therapies have been approved by the FDA since April. They include midostaurin for newly diagnosed, FLT-3–mutated patients; enasidenib, for relapsed/refractory IDH2-mutated patients; CPX-351, for high-risk AML patients; and gemtuzumab ozogamicin for newly diagnosed, CD-33–positive patients.
“Development of novel therapies in order to improve the outcomes of these patients is crucial,” said Dr. Medeiros, director of the inpatient hematology service at Stanford (Calif.) Cancer Institute. “I think all of us in the community hope that this is just the tip of the iceberg – this is just the beginning.”
The field is still struggling to negotiate the newly broadened landscape of AML treatment, he said. For instance, it’s not known exactly which patients are likely to respond to isocitrate dehydrogenase (IDH) inhibitors, he said.
He did offer some guidance on the use of CPX-351, a new formulation of the chemotherapeutic agents cytarabine and daunorubicin that is active in chemotherapy-resistant patients and could be a useful tool leading up to transplant.
“It appears that this drug is able to actually get patients into remission more effectively, leads to fewer toxicities and then allows patients to get to transplant in better shape with better disease response, translating into better overall outcomes,” Dr. Medeiros said.
Many more drugs are in development, with results likely to be revealed soon. Approval for a novel IDH1 inhibitor – only the IDH2 inhibitor is currently approved – is expected early next year. Also under investigation are the hypomethylating agents guadecitabine, a formulation that protects decitabine from degradation, and oral azacitidine, which might be beneficial particularly to patients not eligible for allogeneic stem cell transplant; the B-cell lymphoma 2–inhibitor venetoclax; and an E-selectin antagonist that targets an adhesion molecule in AML cells.
Chimeric antigen receptor T-cell therapy – so promising in other areas of hematologic treatment – is complicated in AML, he said, because of the lack of a target that doesn’t bring on unwanted effects.
“The expression of any antigen in leukemic stem cells is also shared by the expression in hematopoietic stem cells and therefore the use of agents that will target these particular antigens consequently leads to an ‘on-target, off-leukemia’ side effect associated with myeloid cell aplasia.”
Dr. Medeiros reports financial relationships with Celgene, Jazz, Novartis, Pfizer, and other companies.
SAN FRANCISCO – After years of stagnation in the field of acute myeloid leukemia – with most standard therapies developed in the 1970s – times are changing, Bruno Medeiros, MD, said at the annual congress on hematologic malignancies held by the National Comprehensive Cancer Network.
“2017 is the year of AML,” he said. Four new therapies have been approved by the FDA since April. They include midostaurin for newly diagnosed, FLT-3–mutated patients; enasidenib, for relapsed/refractory IDH2-mutated patients; CPX-351, for high-risk AML patients; and gemtuzumab ozogamicin for newly diagnosed, CD-33–positive patients.
“Development of novel therapies in order to improve the outcomes of these patients is crucial,” said Dr. Medeiros, director of the inpatient hematology service at Stanford (Calif.) Cancer Institute. “I think all of us in the community hope that this is just the tip of the iceberg – this is just the beginning.”
The field is still struggling to negotiate the newly broadened landscape of AML treatment, he said. For instance, it’s not known exactly which patients are likely to respond to isocitrate dehydrogenase (IDH) inhibitors, he said.
He did offer some guidance on the use of CPX-351, a new formulation of the chemotherapeutic agents cytarabine and daunorubicin that is active in chemotherapy-resistant patients and could be a useful tool leading up to transplant.
“It appears that this drug is able to actually get patients into remission more effectively, leads to fewer toxicities and then allows patients to get to transplant in better shape with better disease response, translating into better overall outcomes,” Dr. Medeiros said.
Many more drugs are in development, with results likely to be revealed soon. Approval for a novel IDH1 inhibitor – only the IDH2 inhibitor is currently approved – is expected early next year. Also under investigation are the hypomethylating agents guadecitabine, a formulation that protects decitabine from degradation, and oral azacitidine, which might be beneficial particularly to patients not eligible for allogeneic stem cell transplant; the B-cell lymphoma 2–inhibitor venetoclax; and an E-selectin antagonist that targets an adhesion molecule in AML cells.
Chimeric antigen receptor T-cell therapy – so promising in other areas of hematologic treatment – is complicated in AML, he said, because of the lack of a target that doesn’t bring on unwanted effects.
“The expression of any antigen in leukemic stem cells is also shared by the expression in hematopoietic stem cells and therefore the use of agents that will target these particular antigens consequently leads to an ‘on-target, off-leukemia’ side effect associated with myeloid cell aplasia.”
Dr. Medeiros reports financial relationships with Celgene, Jazz, Novartis, Pfizer, and other companies.
SAN FRANCISCO – After years of stagnation in the field of acute myeloid leukemia – with most standard therapies developed in the 1970s – times are changing, Bruno Medeiros, MD, said at the annual congress on hematologic malignancies held by the National Comprehensive Cancer Network.
“2017 is the year of AML,” he said. Four new therapies have been approved by the FDA since April. They include midostaurin for newly diagnosed, FLT-3–mutated patients; enasidenib, for relapsed/refractory IDH2-mutated patients; CPX-351, for high-risk AML patients; and gemtuzumab ozogamicin for newly diagnosed, CD-33–positive patients.
“Development of novel therapies in order to improve the outcomes of these patients is crucial,” said Dr. Medeiros, director of the inpatient hematology service at Stanford (Calif.) Cancer Institute. “I think all of us in the community hope that this is just the tip of the iceberg – this is just the beginning.”
The field is still struggling to negotiate the newly broadened landscape of AML treatment, he said. For instance, it’s not known exactly which patients are likely to respond to isocitrate dehydrogenase (IDH) inhibitors, he said.
He did offer some guidance on the use of CPX-351, a new formulation of the chemotherapeutic agents cytarabine and daunorubicin that is active in chemotherapy-resistant patients and could be a useful tool leading up to transplant.
“It appears that this drug is able to actually get patients into remission more effectively, leads to fewer toxicities and then allows patients to get to transplant in better shape with better disease response, translating into better overall outcomes,” Dr. Medeiros said.
Many more drugs are in development, with results likely to be revealed soon. Approval for a novel IDH1 inhibitor – only the IDH2 inhibitor is currently approved – is expected early next year. Also under investigation are the hypomethylating agents guadecitabine, a formulation that protects decitabine from degradation, and oral azacitidine, which might be beneficial particularly to patients not eligible for allogeneic stem cell transplant; the B-cell lymphoma 2–inhibitor venetoclax; and an E-selectin antagonist that targets an adhesion molecule in AML cells.
Chimeric antigen receptor T-cell therapy – so promising in other areas of hematologic treatment – is complicated in AML, he said, because of the lack of a target that doesn’t bring on unwanted effects.
“The expression of any antigen in leukemic stem cells is also shared by the expression in hematopoietic stem cells and therefore the use of agents that will target these particular antigens consequently leads to an ‘on-target, off-leukemia’ side effect associated with myeloid cell aplasia.”
Dr. Medeiros reports financial relationships with Celgene, Jazz, Novartis, Pfizer, and other companies.
EXPERT ANALYSIS FROM THE NCCN HEMATOLOGIC MALIGNANCIES CONGRESS
ATLG fights GVHD but reduces PFS, OS
Results of a phase 3 trial suggest rabbit anti-T lymphocyte globulin (ATLG) can reduce graft-versus-host disease (GVHD) but also decrease survival in patients who have received a hematopoietic stem cell transplant (HSCT) from a matched, unrelated donor.
In this randomized trial, ATLG significantly decreased the incidence of moderate-to-severe chronic GVHD and acute grade 2-4 GVHD, when compared to placebo.
However, patients who received ATLG also had significantly lower progression-free survival (PFS) and overall survival (OS) than placebo-treated patients.
On the other hand, the data also suggest that patients who receive conditioning regimens that do not lower absolute lymphocyte counts (ALCs) substantially may not experience a significant decrease in survival with ATLG.
These results were published in the Journal of Clinical Oncology. The study was sponsored by Neovii Pharmaceuticals AG, which is developing ATLG as Grafalon®.
The study was a prospective, randomized, double-blind trial conducted in North America and Australia (NCT01295710). It enrolled 254 patients, ages 18 to 65, who had acute lymphoblastic leukemia, acute myeloid leukemia, or myelodysplastic syndromes. All patients were undergoing myeloablative, HLA-matched, unrelated HSCT.
Patients were randomized in a 1:1 fashion to receive ATLG (given at 20 mg/kg/day, n=126) or placebo (250 ml of normal saline, n=128) on days -3, -2, and -1 prior to HSCT.
In addition, all patients received antihistamine and methylprednisolone (at 2 mg/kg on day -3 and 1 mg/kg on days -2 and -1).
Patients also received GVHD prophylaxis in the form of tacrolimus (with a target serum trough level of 5 to 15 ng/mL) and methotrexate (15 mg/m2 on day 1, then 10 mg/m2 on days 3, 6, and 11). If patients did not develop clinical GVHD, tacrolimus was tapered starting on day 50 or later over a minimum of 26 weeks and ultimately discontinued.
Patients received 1 of 3 conditioning regimens, which were declared prior to randomization and included:
- Cyclophosphamide at 120 mg/kg intravenously (IV) and fractionated total body irradiation (TBI, ≥12 Gy)
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and cyclophosphamide at 120 mg/kg IV
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and fludarabine at 120 mg/m2 IV.
Overall results
Compared to placebo-treated patients, those who received ATLG had a significant reduction in grade 2-4 acute GVHD—23% and 40%, respectively (P=0.004)—and moderate-to-severe chronic GVHD—12% and 33%, respectively (P<0.001).
However, there was no significant difference between the ATLG and placebo arms with regard to moderate-severe chronic GVHD-free survival. The 2-year estimate was 48% and 44%, respectively (P=0.47).
In addition, PFS and OS were significantly lower in patients who received ATLG. The estimated 2-year PFS was 47% in the ATLG arm and 65% in the placebo arm (P=0.04). The estimated 2-year OS was 59% and 74%, respectively (P=0.034).
In a multivariable analysis, ATLG remained significantly associated with inferior PFS (hazard ratio [HR]=1.55, P=0.026) and OS (hazard ratio=1.74, P=0.01).
Role of conditioning, ALC
The researchers found evidence to suggest that conditioning regimen and ALC played a role in patient outcomes.
For patients who received cyclophosphamide and TBI, 2-year moderate-severe chronic GVHD-free survival was 61% in the placebo arm and 38% in the ATLG arm (P=0.080). Two-year OS was 88% and 48%, respectively (P=0.006). And 2-year PFS was 75% and 29%, respectively (P=0.007).
For patients who received busulfan and cyclophosphamide, 2-year moderate-severe chronic GVHD-free survival was 47% in the placebo arm and 53% in the ATLG arm (P=0.650). Two-year OS was 77% and 71%, respectively (P=0.350). And 2-year PFS was 73% and 60%, respectively (P=0.460).
For patients who received busulfan and fludarabine, 2-year moderate-severe chronic GVHD-free survival was 33% in the placebo arm and 49% in the ATLG arm (P=0.047). Two-year OS was 66% and 53%, respectively (P=0.520). And 2-year PFS was 58% and 48%, respectively (P=0.540).
The researchers noted that the choice of conditioning regimen had a “profound effect” on ALC at day -3 (the time of ATLG/placebo initiation). More than 70% of patients who received TBI had an ALC <0.1 x 109/L, compared to less than 35% of patients who received busulfan-based conditioning.
ALC, in turn, had an impact on PFS and OS. In patients with an ALC ≥ 0.1 x 109/L on day -3, ATLG did not compromise PFS or OS, but PFS and OS were negatively affected in patients with an ALC < 0.1.
ATLG recipients with an ALC < 0.1 had significantly worse OS (HR=4.13, P<0.001) and PFS (HR=3.19, P<0.001) than patients with an ALC ≥ 0.1. ![]()
Results of a phase 3 trial suggest rabbit anti-T lymphocyte globulin (ATLG) can reduce graft-versus-host disease (GVHD) but also decrease survival in patients who have received a hematopoietic stem cell transplant (HSCT) from a matched, unrelated donor.
In this randomized trial, ATLG significantly decreased the incidence of moderate-to-severe chronic GVHD and acute grade 2-4 GVHD, when compared to placebo.
However, patients who received ATLG also had significantly lower progression-free survival (PFS) and overall survival (OS) than placebo-treated patients.
On the other hand, the data also suggest that patients who receive conditioning regimens that do not lower absolute lymphocyte counts (ALCs) substantially may not experience a significant decrease in survival with ATLG.
These results were published in the Journal of Clinical Oncology. The study was sponsored by Neovii Pharmaceuticals AG, which is developing ATLG as Grafalon®.
The study was a prospective, randomized, double-blind trial conducted in North America and Australia (NCT01295710). It enrolled 254 patients, ages 18 to 65, who had acute lymphoblastic leukemia, acute myeloid leukemia, or myelodysplastic syndromes. All patients were undergoing myeloablative, HLA-matched, unrelated HSCT.
Patients were randomized in a 1:1 fashion to receive ATLG (given at 20 mg/kg/day, n=126) or placebo (250 ml of normal saline, n=128) on days -3, -2, and -1 prior to HSCT.
In addition, all patients received antihistamine and methylprednisolone (at 2 mg/kg on day -3 and 1 mg/kg on days -2 and -1).
Patients also received GVHD prophylaxis in the form of tacrolimus (with a target serum trough level of 5 to 15 ng/mL) and methotrexate (15 mg/m2 on day 1, then 10 mg/m2 on days 3, 6, and 11). If patients did not develop clinical GVHD, tacrolimus was tapered starting on day 50 or later over a minimum of 26 weeks and ultimately discontinued.
Patients received 1 of 3 conditioning regimens, which were declared prior to randomization and included:
- Cyclophosphamide at 120 mg/kg intravenously (IV) and fractionated total body irradiation (TBI, ≥12 Gy)
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and cyclophosphamide at 120 mg/kg IV
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and fludarabine at 120 mg/m2 IV.
Overall results
Compared to placebo-treated patients, those who received ATLG had a significant reduction in grade 2-4 acute GVHD—23% and 40%, respectively (P=0.004)—and moderate-to-severe chronic GVHD—12% and 33%, respectively (P<0.001).
However, there was no significant difference between the ATLG and placebo arms with regard to moderate-severe chronic GVHD-free survival. The 2-year estimate was 48% and 44%, respectively (P=0.47).
In addition, PFS and OS were significantly lower in patients who received ATLG. The estimated 2-year PFS was 47% in the ATLG arm and 65% in the placebo arm (P=0.04). The estimated 2-year OS was 59% and 74%, respectively (P=0.034).
In a multivariable analysis, ATLG remained significantly associated with inferior PFS (hazard ratio [HR]=1.55, P=0.026) and OS (hazard ratio=1.74, P=0.01).
Role of conditioning, ALC
The researchers found evidence to suggest that conditioning regimen and ALC played a role in patient outcomes.
For patients who received cyclophosphamide and TBI, 2-year moderate-severe chronic GVHD-free survival was 61% in the placebo arm and 38% in the ATLG arm (P=0.080). Two-year OS was 88% and 48%, respectively (P=0.006). And 2-year PFS was 75% and 29%, respectively (P=0.007).
For patients who received busulfan and cyclophosphamide, 2-year moderate-severe chronic GVHD-free survival was 47% in the placebo arm and 53% in the ATLG arm (P=0.650). Two-year OS was 77% and 71%, respectively (P=0.350). And 2-year PFS was 73% and 60%, respectively (P=0.460).
For patients who received busulfan and fludarabine, 2-year moderate-severe chronic GVHD-free survival was 33% in the placebo arm and 49% in the ATLG arm (P=0.047). Two-year OS was 66% and 53%, respectively (P=0.520). And 2-year PFS was 58% and 48%, respectively (P=0.540).
The researchers noted that the choice of conditioning regimen had a “profound effect” on ALC at day -3 (the time of ATLG/placebo initiation). More than 70% of patients who received TBI had an ALC <0.1 x 109/L, compared to less than 35% of patients who received busulfan-based conditioning.
ALC, in turn, had an impact on PFS and OS. In patients with an ALC ≥ 0.1 x 109/L on day -3, ATLG did not compromise PFS or OS, but PFS and OS were negatively affected in patients with an ALC < 0.1.
ATLG recipients with an ALC < 0.1 had significantly worse OS (HR=4.13, P<0.001) and PFS (HR=3.19, P<0.001) than patients with an ALC ≥ 0.1. ![]()
Results of a phase 3 trial suggest rabbit anti-T lymphocyte globulin (ATLG) can reduce graft-versus-host disease (GVHD) but also decrease survival in patients who have received a hematopoietic stem cell transplant (HSCT) from a matched, unrelated donor.
In this randomized trial, ATLG significantly decreased the incidence of moderate-to-severe chronic GVHD and acute grade 2-4 GVHD, when compared to placebo.
However, patients who received ATLG also had significantly lower progression-free survival (PFS) and overall survival (OS) than placebo-treated patients.
On the other hand, the data also suggest that patients who receive conditioning regimens that do not lower absolute lymphocyte counts (ALCs) substantially may not experience a significant decrease in survival with ATLG.
These results were published in the Journal of Clinical Oncology. The study was sponsored by Neovii Pharmaceuticals AG, which is developing ATLG as Grafalon®.
The study was a prospective, randomized, double-blind trial conducted in North America and Australia (NCT01295710). It enrolled 254 patients, ages 18 to 65, who had acute lymphoblastic leukemia, acute myeloid leukemia, or myelodysplastic syndromes. All patients were undergoing myeloablative, HLA-matched, unrelated HSCT.
Patients were randomized in a 1:1 fashion to receive ATLG (given at 20 mg/kg/day, n=126) or placebo (250 ml of normal saline, n=128) on days -3, -2, and -1 prior to HSCT.
In addition, all patients received antihistamine and methylprednisolone (at 2 mg/kg on day -3 and 1 mg/kg on days -2 and -1).
Patients also received GVHD prophylaxis in the form of tacrolimus (with a target serum trough level of 5 to 15 ng/mL) and methotrexate (15 mg/m2 on day 1, then 10 mg/m2 on days 3, 6, and 11). If patients did not develop clinical GVHD, tacrolimus was tapered starting on day 50 or later over a minimum of 26 weeks and ultimately discontinued.
Patients received 1 of 3 conditioning regimens, which were declared prior to randomization and included:
- Cyclophosphamide at 120 mg/kg intravenously (IV) and fractionated total body irradiation (TBI, ≥12 Gy)
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and cyclophosphamide at 120 mg/kg IV
- Busulfan at 16 mg/kg orally or 12.8 mg/kg IV and fludarabine at 120 mg/m2 IV.
Overall results
Compared to placebo-treated patients, those who received ATLG had a significant reduction in grade 2-4 acute GVHD—23% and 40%, respectively (P=0.004)—and moderate-to-severe chronic GVHD—12% and 33%, respectively (P<0.001).
However, there was no significant difference between the ATLG and placebo arms with regard to moderate-severe chronic GVHD-free survival. The 2-year estimate was 48% and 44%, respectively (P=0.47).
In addition, PFS and OS were significantly lower in patients who received ATLG. The estimated 2-year PFS was 47% in the ATLG arm and 65% in the placebo arm (P=0.04). The estimated 2-year OS was 59% and 74%, respectively (P=0.034).
In a multivariable analysis, ATLG remained significantly associated with inferior PFS (hazard ratio [HR]=1.55, P=0.026) and OS (hazard ratio=1.74, P=0.01).
Role of conditioning, ALC
The researchers found evidence to suggest that conditioning regimen and ALC played a role in patient outcomes.
For patients who received cyclophosphamide and TBI, 2-year moderate-severe chronic GVHD-free survival was 61% in the placebo arm and 38% in the ATLG arm (P=0.080). Two-year OS was 88% and 48%, respectively (P=0.006). And 2-year PFS was 75% and 29%, respectively (P=0.007).
For patients who received busulfan and cyclophosphamide, 2-year moderate-severe chronic GVHD-free survival was 47% in the placebo arm and 53% in the ATLG arm (P=0.650). Two-year OS was 77% and 71%, respectively (P=0.350). And 2-year PFS was 73% and 60%, respectively (P=0.460).
For patients who received busulfan and fludarabine, 2-year moderate-severe chronic GVHD-free survival was 33% in the placebo arm and 49% in the ATLG arm (P=0.047). Two-year OS was 66% and 53%, respectively (P=0.520). And 2-year PFS was 58% and 48%, respectively (P=0.540).
The researchers noted that the choice of conditioning regimen had a “profound effect” on ALC at day -3 (the time of ATLG/placebo initiation). More than 70% of patients who received TBI had an ALC <0.1 x 109/L, compared to less than 35% of patients who received busulfan-based conditioning.
ALC, in turn, had an impact on PFS and OS. In patients with an ALC ≥ 0.1 x 109/L on day -3, ATLG did not compromise PFS or OS, but PFS and OS were negatively affected in patients with an ALC < 0.1.
ATLG recipients with an ALC < 0.1 had significantly worse OS (HR=4.13, P<0.001) and PFS (HR=3.19, P<0.001) than patients with an ALC ≥ 0.1. ![]()
AML patients overestimate treatment risk and chance of cure
Older patients with acute myeloid leukemia (AML) tend to overestimate not only the risks of treatment, but also their likelihood of cure, according to the results of a 100-patient longitudinal study presented at the Palliative and Supportive Care in Oncology Symposium.
Compared with what their oncologists thought, patients were significantly more likely to believe they would die from treatment, said senior study author Areej El-Jawahri, MD, of Massachusetts General Hospital in Boston.
Yet when surveyed again 1 month later, they were significantly more likely to overestimate the chances of being cured, versus the chances of cure given by their oncologists.
“We really need interventions to facilitate communication and ensure accurate prognostic understanding in this patient population, where understanding the treatment risk and prognosis can have a significant effect on their treatment choices and treatment decisions,” Dr. El-Jawahri said in a press conference from the symposium, which was cosponsored by AAHPM, ASCO, ASTRO, and MASCC.
Patients were first surveyed within 72 hours of starting chemotherapy.
At that point, 91% of patients said it was somewhat likely (63%) or extremely likely (28%) they would die as a result of their treatment, but among their oncologists, only 22% said death due to treatment was somewhat likely, and none thought it was very likely, according to the presented data.
A much different picture emerged 1 month later, when 90% of patients said they were somewhat or very likely to be cured of their leukemia, while only 26% of physicians said it was somewhat likely for the patient to be cured.
For both the question on treatment risk and the question on cure potential, the differences between patient responses and physician responses was significant (P less than .001), according to Dr. El-Jawahri.
Of note, half of the patients (n = 50) received intensive chemotherapy, while the other half received nonintensive (that is, palliative) chemotherapy. Patients receiving palliative therapy were even more likely to overestimate their chances of cure, Dr. El-Jawahi said.
This study highlights how stress and anxiety might shape a patient’s perception of treatment and prognosis, and provides new evidence that “accurate knowledge” can lead to “efficiencies on both sides of this [doctor-patient] interface,” said Andrew S. Epstein, MD, of Memorial Sloan-Kettering Cancer Center, New York.
A shared understanding of prognosis and treatment risk between clinician and patient is “crucial in informed consent” and can help patients make better-informed decisions with their doctor, said Dr. Epstein, who was not involved with the study.
This study was funded by a grant from the National Cancer Institute. Dr. El-Jawahri, the senior author, reported no relevant financial disclosures.
Older patients with acute myeloid leukemia (AML) tend to overestimate not only the risks of treatment, but also their likelihood of cure, according to the results of a 100-patient longitudinal study presented at the Palliative and Supportive Care in Oncology Symposium.
Compared with what their oncologists thought, patients were significantly more likely to believe they would die from treatment, said senior study author Areej El-Jawahri, MD, of Massachusetts General Hospital in Boston.
Yet when surveyed again 1 month later, they were significantly more likely to overestimate the chances of being cured, versus the chances of cure given by their oncologists.
“We really need interventions to facilitate communication and ensure accurate prognostic understanding in this patient population, where understanding the treatment risk and prognosis can have a significant effect on their treatment choices and treatment decisions,” Dr. El-Jawahri said in a press conference from the symposium, which was cosponsored by AAHPM, ASCO, ASTRO, and MASCC.
Patients were first surveyed within 72 hours of starting chemotherapy.
At that point, 91% of patients said it was somewhat likely (63%) or extremely likely (28%) they would die as a result of their treatment, but among their oncologists, only 22% said death due to treatment was somewhat likely, and none thought it was very likely, according to the presented data.
A much different picture emerged 1 month later, when 90% of patients said they were somewhat or very likely to be cured of their leukemia, while only 26% of physicians said it was somewhat likely for the patient to be cured.
For both the question on treatment risk and the question on cure potential, the differences between patient responses and physician responses was significant (P less than .001), according to Dr. El-Jawahri.
Of note, half of the patients (n = 50) received intensive chemotherapy, while the other half received nonintensive (that is, palliative) chemotherapy. Patients receiving palliative therapy were even more likely to overestimate their chances of cure, Dr. El-Jawahi said.
This study highlights how stress and anxiety might shape a patient’s perception of treatment and prognosis, and provides new evidence that “accurate knowledge” can lead to “efficiencies on both sides of this [doctor-patient] interface,” said Andrew S. Epstein, MD, of Memorial Sloan-Kettering Cancer Center, New York.
A shared understanding of prognosis and treatment risk between clinician and patient is “crucial in informed consent” and can help patients make better-informed decisions with their doctor, said Dr. Epstein, who was not involved with the study.
This study was funded by a grant from the National Cancer Institute. Dr. El-Jawahri, the senior author, reported no relevant financial disclosures.
Older patients with acute myeloid leukemia (AML) tend to overestimate not only the risks of treatment, but also their likelihood of cure, according to the results of a 100-patient longitudinal study presented at the Palliative and Supportive Care in Oncology Symposium.
Compared with what their oncologists thought, patients were significantly more likely to believe they would die from treatment, said senior study author Areej El-Jawahri, MD, of Massachusetts General Hospital in Boston.
Yet when surveyed again 1 month later, they were significantly more likely to overestimate the chances of being cured, versus the chances of cure given by their oncologists.
“We really need interventions to facilitate communication and ensure accurate prognostic understanding in this patient population, where understanding the treatment risk and prognosis can have a significant effect on their treatment choices and treatment decisions,” Dr. El-Jawahri said in a press conference from the symposium, which was cosponsored by AAHPM, ASCO, ASTRO, and MASCC.
Patients were first surveyed within 72 hours of starting chemotherapy.
At that point, 91% of patients said it was somewhat likely (63%) or extremely likely (28%) they would die as a result of their treatment, but among their oncologists, only 22% said death due to treatment was somewhat likely, and none thought it was very likely, according to the presented data.
A much different picture emerged 1 month later, when 90% of patients said they were somewhat or very likely to be cured of their leukemia, while only 26% of physicians said it was somewhat likely for the patient to be cured.
For both the question on treatment risk and the question on cure potential, the differences between patient responses and physician responses was significant (P less than .001), according to Dr. El-Jawahri.
Of note, half of the patients (n = 50) received intensive chemotherapy, while the other half received nonintensive (that is, palliative) chemotherapy. Patients receiving palliative therapy were even more likely to overestimate their chances of cure, Dr. El-Jawahi said.
This study highlights how stress and anxiety might shape a patient’s perception of treatment and prognosis, and provides new evidence that “accurate knowledge” can lead to “efficiencies on both sides of this [doctor-patient] interface,” said Andrew S. Epstein, MD, of Memorial Sloan-Kettering Cancer Center, New York.
A shared understanding of prognosis and treatment risk between clinician and patient is “crucial in informed consent” and can help patients make better-informed decisions with their doctor, said Dr. Epstein, who was not involved with the study.
This study was funded by a grant from the National Cancer Institute. Dr. El-Jawahri, the senior author, reported no relevant financial disclosures.
FROM PALLONC 2017
Key clinical point: Acute myeloid leukemia (AML) patients overestimated both the risks of treatment and the chances of cure, compared with estimates from their own oncologists.
Major finding: Ninety-one percent of patients thought it was somewhat or extremely likely they would die from the treatment, while only 22% of oncologists said it was somewhat likely. A month later, 90% of patients thought it was somewhat or very likely they would be cured of their AML, but only 26% of oncologists said cure was somewhat likely.
Data source: A longitudinal study including 100 patients with newly diagnosed AML treated at one of two tertiary hospitals.
Disclosures: This study was funded by a grant from the National Cancer Institute. Dr. El-Jawahri, the senior author, reported no relevant financial disclosures.
CCSs more likely to stay at jobs to keep health insurance
Survey results suggest childhood cancer survivors (CCSs) in the US are more likely than individuals without a history of cancer to experience “job lock,” or staying at a job to keep work-related health insurance.
CCSs are also more likely than individuals without a history of cancer to report problems paying medical bills and being denied health insurance.
Anne Kirchhoff, PhD, of Huntsman Cancer Institute at the University of Utah in Salt Lake City, and her colleagues reported these findings in JAMA Oncology.
The researchers analyzed 394 CCSs from pediatric oncology institutions across the US, along with 128 of their siblings who had no history of cancer. All study participants worked 35 hours or more per week.
The most common cancer diagnosis among CCSs was leukemia (35.4%), followed by Hodgkin lymphoma (14.9%). Most patients had undergone chemotherapy (77.2%), radiotherapy (63.9%), and surgery (81.1%).
Overall, sociodemographic and clinical characteristics were similar between CCSs and siblings. However, CCSs were more likely than siblings to have severe, disabling, or life-threatening chronic conditions—33.9% and 17.7%, respectively (P<0.001).
Most CCSs (88.0%) and siblings (88.5%) had employer-sponsored health insurance. Three percent of siblings and 5.3% of CCSs had individual insurance; 1.9% and 2.3%, respectively, had public insurance; and 6.7% and 4.4%, respectively, were uninsured.
Results
CCSs were more likely than siblings to report:
- Job lock—23.2% and 16.9%, respectively (P=0.16)
- Problems paying medical bills—20.1% and 12.9%, respectively (P=0.09)
- Denial of health insurance—13.4% and 1.8%, respectively (P<0.001).
In a multivariable analysis, insurance denial remained significantly more common among CCSs than siblings (relative risk [RR]=7.38).
In another multivariable analysis, 38% of CCSs with a previous insurance denial reported job lock, compared with 20% of those who never experienced insurance denial (RR=1.60). And 44% of CCSs who reported problems paying their medical bills also reported job lock, compared to 16% of those who had no problems paying medical bills (RR=2.43).
The researchers also found that female CCSs (RR=1.70) and CCSs with severe, disabling, or life-threatening chronic conditions (RR=1.72) were more likely to report job lock.
“This information gives us a feel for high-risk groups of survivors who may need more information about insurance,” Dr Kirchhoff said. “Many people experience a gap in education and literacy around insurance, and it’s important for people to understand their options—even those who are employed and consistently had access to insurance through work. We want to know what their concerns are so we can help patients and survivors. Getting healthcare should not be a worry for cancer survivors.”
“Survivors have been through a lot when they were younger and understand the importance of making sure they can get healthcare when they need it. I think a lot of them also saw what their parents and families went through in terms of the financial stress and burden of dealing with a health crisis. So they’re just primed to understand the importance of health insurance.”
Dr Kirchhoff noted that this study was conducted as the Affordable Care Act was rolling out. Therefore, she would like to do a follow-up study to see if the insurance exchanges and Medicaid expansion lessened job-related insurance worries. ![]()
Survey results suggest childhood cancer survivors (CCSs) in the US are more likely than individuals without a history of cancer to experience “job lock,” or staying at a job to keep work-related health insurance.
CCSs are also more likely than individuals without a history of cancer to report problems paying medical bills and being denied health insurance.
Anne Kirchhoff, PhD, of Huntsman Cancer Institute at the University of Utah in Salt Lake City, and her colleagues reported these findings in JAMA Oncology.
The researchers analyzed 394 CCSs from pediatric oncology institutions across the US, along with 128 of their siblings who had no history of cancer. All study participants worked 35 hours or more per week.
The most common cancer diagnosis among CCSs was leukemia (35.4%), followed by Hodgkin lymphoma (14.9%). Most patients had undergone chemotherapy (77.2%), radiotherapy (63.9%), and surgery (81.1%).
Overall, sociodemographic and clinical characteristics were similar between CCSs and siblings. However, CCSs were more likely than siblings to have severe, disabling, or life-threatening chronic conditions—33.9% and 17.7%, respectively (P<0.001).
Most CCSs (88.0%) and siblings (88.5%) had employer-sponsored health insurance. Three percent of siblings and 5.3% of CCSs had individual insurance; 1.9% and 2.3%, respectively, had public insurance; and 6.7% and 4.4%, respectively, were uninsured.
Results
CCSs were more likely than siblings to report:
- Job lock—23.2% and 16.9%, respectively (P=0.16)
- Problems paying medical bills—20.1% and 12.9%, respectively (P=0.09)
- Denial of health insurance—13.4% and 1.8%, respectively (P<0.001).
In a multivariable analysis, insurance denial remained significantly more common among CCSs than siblings (relative risk [RR]=7.38).
In another multivariable analysis, 38% of CCSs with a previous insurance denial reported job lock, compared with 20% of those who never experienced insurance denial (RR=1.60). And 44% of CCSs who reported problems paying their medical bills also reported job lock, compared to 16% of those who had no problems paying medical bills (RR=2.43).
The researchers also found that female CCSs (RR=1.70) and CCSs with severe, disabling, or life-threatening chronic conditions (RR=1.72) were more likely to report job lock.
“This information gives us a feel for high-risk groups of survivors who may need more information about insurance,” Dr Kirchhoff said. “Many people experience a gap in education and literacy around insurance, and it’s important for people to understand their options—even those who are employed and consistently had access to insurance through work. We want to know what their concerns are so we can help patients and survivors. Getting healthcare should not be a worry for cancer survivors.”
“Survivors have been through a lot when they were younger and understand the importance of making sure they can get healthcare when they need it. I think a lot of them also saw what their parents and families went through in terms of the financial stress and burden of dealing with a health crisis. So they’re just primed to understand the importance of health insurance.”
Dr Kirchhoff noted that this study was conducted as the Affordable Care Act was rolling out. Therefore, she would like to do a follow-up study to see if the insurance exchanges and Medicaid expansion lessened job-related insurance worries. ![]()
Survey results suggest childhood cancer survivors (CCSs) in the US are more likely than individuals without a history of cancer to experience “job lock,” or staying at a job to keep work-related health insurance.
CCSs are also more likely than individuals without a history of cancer to report problems paying medical bills and being denied health insurance.
Anne Kirchhoff, PhD, of Huntsman Cancer Institute at the University of Utah in Salt Lake City, and her colleagues reported these findings in JAMA Oncology.
The researchers analyzed 394 CCSs from pediatric oncology institutions across the US, along with 128 of their siblings who had no history of cancer. All study participants worked 35 hours or more per week.
The most common cancer diagnosis among CCSs was leukemia (35.4%), followed by Hodgkin lymphoma (14.9%). Most patients had undergone chemotherapy (77.2%), radiotherapy (63.9%), and surgery (81.1%).
Overall, sociodemographic and clinical characteristics were similar between CCSs and siblings. However, CCSs were more likely than siblings to have severe, disabling, or life-threatening chronic conditions—33.9% and 17.7%, respectively (P<0.001).
Most CCSs (88.0%) and siblings (88.5%) had employer-sponsored health insurance. Three percent of siblings and 5.3% of CCSs had individual insurance; 1.9% and 2.3%, respectively, had public insurance; and 6.7% and 4.4%, respectively, were uninsured.
Results
CCSs were more likely than siblings to report:
- Job lock—23.2% and 16.9%, respectively (P=0.16)
- Problems paying medical bills—20.1% and 12.9%, respectively (P=0.09)
- Denial of health insurance—13.4% and 1.8%, respectively (P<0.001).
In a multivariable analysis, insurance denial remained significantly more common among CCSs than siblings (relative risk [RR]=7.38).
In another multivariable analysis, 38% of CCSs with a previous insurance denial reported job lock, compared with 20% of those who never experienced insurance denial (RR=1.60). And 44% of CCSs who reported problems paying their medical bills also reported job lock, compared to 16% of those who had no problems paying medical bills (RR=2.43).
The researchers also found that female CCSs (RR=1.70) and CCSs with severe, disabling, or life-threatening chronic conditions (RR=1.72) were more likely to report job lock.
“This information gives us a feel for high-risk groups of survivors who may need more information about insurance,” Dr Kirchhoff said. “Many people experience a gap in education and literacy around insurance, and it’s important for people to understand their options—even those who are employed and consistently had access to insurance through work. We want to know what their concerns are so we can help patients and survivors. Getting healthcare should not be a worry for cancer survivors.”
“Survivors have been through a lot when they were younger and understand the importance of making sure they can get healthcare when they need it. I think a lot of them also saw what their parents and families went through in terms of the financial stress and burden of dealing with a health crisis. So they’re just primed to understand the importance of health insurance.”
Dr Kirchhoff noted that this study was conducted as the Affordable Care Act was rolling out. Therefore, she would like to do a follow-up study to see if the insurance exchanges and Medicaid expansion lessened job-related insurance worries. ![]()