User login
Bleomycin can be safely omitted after negative PET2 in HL
Photo by Jens Maus
LUGANO—Results of the RATHL trial indicate that bleomycin can be omitted from ABVD therapy following a negative interim FDG-PET scan in patients with Hodgkin lymphoma.
Progression-free survival (PFS) and overall survival (OS) were the same at 3 years for patients who were PET-negative after 2 cycles of ABVD and then continued therapy with or without bleomycin.
These results were presented at the 13th International Conference on Malignant Lymphoma (13-ICML).
Investigators based the RATHL study on the principles that it’s desirable to de-escalate treatment in the best responders to avoid late toxicity and that PET scans after 2 cycles of ABVD are highly predictive.
The team enrolled 1214 patients from 6 countries, 861 of whom were in the UK. Patients received a PET scan at staging, 2 cycles of ABVD, and then a second PET scan (PET2).
If patients were negative after PET2, they were randomized to receive 4 more cycles of ABVD or AVD and no radiotherapy.
If they were positive after PET2, patients received 4 cycles of BEACOPP-14 or 3 cycles of escalated BEACOPP. These patients then received a third PET scan, and the positive patients went on to receive radiotherapy or a salvage regimen.
The PET3-negative patients received 2 more cycles of BEACOPP-14 or one of escalated BEACOPP without radiotherapy.
Peter W. Johnson, MD, of the University of Southampton in the UK, presented the results of these treatment regimens during the plenary session of 13-ICML as abstract 008.
Patient characteristics
Patients were a median age of 33 (range, 18-79), and 55% were male. They had disease stages of II (41%), III (31%), or IV (28%).
Seventy-four percent of patients had a performance status of 0. Almost half (49%) had an IPS score of 2 to 3, and 18% had an IPS score of 4 or more. Thirty-two percent had bulky disease.
Investigators followed the patients for a median of 34.7 months (range, 1 day to 68.2 months).
Results after PET2
Seventy-seven patients were missing a second PET scan, mostly due to PET protocol violations of having to use the same scanner for the baseline and second scan and the same acquisition time.
“We were very strict on our quality control,” Dr Johnson said, “because we wished to make sure this was reproducible data.”
So the results after 2 cycles of ABVD treatment were based on 1137 patients.
PET-negative patients
More than 80% of patients were PET-negative after 2 cycles. Four hundred and sixty-nine patients were randomized to receive ABVD and 466 to AVD.
The groups were well-balanced in terms of median age, performance status, stage, B symptoms, bulky disease, and IPS score.
There was a significant excess of neutropenic fever (P=0.032) and infection (P=0.040) in those patients continuing on ABVD compared to AVD. And any hematologic toxicity was highly significantly different between the 2 arms (P<0.001).
“So we have demonstrated that continuing with bleomycin beyond cycle 2 is accompanied by significantly more toxicity,” Dr Johnson said.
Ninety-eight percent of patients in both cohorts received at least 6 cycles of therapy post-randomization.
At a median follow-up of 36.3 months, 65% of patients in the ABVD arm and 69% in the AVD arm achieved a complete remission (CR) or unconfirmed CR (CRu).
Fourteen patients died in each of the arms. Seven patients died of their disease in the AVD arm, compared with 1 in the ABVD arm. Slightly more patients died from toxicity in the ABVD arm.
The primary endpoint of PFS showed very little difference between the 2 arms. The 3-year PFS in the intent-to-treat analysis was 85.4% for patients in the ABVD arm and 84.4% for those in the AVD arm.
The investigators observed that the PFS of 85% was somewhat lower than the 95% PFS observed in the literature. So they looked at the association between baseline factors and PFS after negative PET2.
“And what stands out from this is that if you have high-stage disease at presentation, there is a slightly higher chance of treatment failure following a negative PET scan,” Dr Johnson said. “And you can see the trend here, from early stage disease up to advanced-stage disease, the PET scan becomes a less reliable indicator of result.”
The investigators also conducted a subgroup analysis of the PET2-negative patients and found there was no difference in outcome between treatment arms in patients with more advanced disease, with bulky disease, with a high IPS score, or according to the PET score.
“So we have not succeeded in finding any subgroup where it appears to be beneficial to continue bleomycin,” Dr Johnson said.
The OS rate was also the same between the 2 arms, at 97%.
PET2-positive patients
One hundred and seventy-four patients who were positive after the second PET scan received either BEACOPP for 14 days or escalated BEACOPP.
The percentage of patients who experienced grade 3-4 toxicities was largely similar between the 2 regimens, although the patients receiving escalated BEACOPP had more neutropenia (P=0.057), thrombocytopenia (P=0.001), and neutropenic fever (P=0.08).
In terms of efficacy, two-thirds of patients became PET-negative by the third PET scan, and 48% of patients achieved a CR or CRu.
Twenty-one patients died, 8 due to Hodgkin lymphoma.
The PFS was 66.0% in the BEACOPP-14 group and 71.1% in the escalated-BEACOPP group. The 3-year OS was 89.6% in the BEACOPP-14 group and 82.8% in the escalated-BEACOPP group.
For the entire group of 1214 patients, the 3-year PFS was 82.5%, and the OS was 95.4%.
Based on these results, the investigators concluded that it is safe to omit bleomycin and consolidation radiotherapy from subsequent ABVD therapy after a negative interim PET scan. And doing so reduces toxicity, especially dyspnea, thromboembolism, and neutropenic fever.
“[B]y using more selective chemotherapy and much less radiotherapy than we have previously used in our studies, where we’re giving less than 3% of patients consolidation radiotherapy, the results appear to be favorable and an improvement over what we have seen previously,” Dr Johnson said.
Details on lung toxicity in this study were presented separately at 13-ICML as abstract 041. ![]()

Photo by Jens Maus
LUGANO—Results of the RATHL trial indicate that bleomycin can be omitted from ABVD therapy following a negative interim FDG-PET scan in patients with Hodgkin lymphoma.
Progression-free survival (PFS) and overall survival (OS) were the same at 3 years for patients who were PET-negative after 2 cycles of ABVD and then continued therapy with or without bleomycin.
These results were presented at the 13th International Conference on Malignant Lymphoma (13-ICML).
Investigators based the RATHL study on the principles that it’s desirable to de-escalate treatment in the best responders to avoid late toxicity and that PET scans after 2 cycles of ABVD are highly predictive.
The team enrolled 1214 patients from 6 countries, 861 of whom were in the UK. Patients received a PET scan at staging, 2 cycles of ABVD, and then a second PET scan (PET2).
If patients were negative after PET2, they were randomized to receive 4 more cycles of ABVD or AVD and no radiotherapy.
If they were positive after PET2, patients received 4 cycles of BEACOPP-14 or 3 cycles of escalated BEACOPP. These patients then received a third PET scan, and the positive patients went on to receive radiotherapy or a salvage regimen.
The PET3-negative patients received 2 more cycles of BEACOPP-14 or one of escalated BEACOPP without radiotherapy.
Peter W. Johnson, MD, of the University of Southampton in the UK, presented the results of these treatment regimens during the plenary session of 13-ICML as abstract 008.
Patient characteristics
Patients were a median age of 33 (range, 18-79), and 55% were male. They had disease stages of II (41%), III (31%), or IV (28%).
Seventy-four percent of patients had a performance status of 0. Almost half (49%) had an IPS score of 2 to 3, and 18% had an IPS score of 4 or more. Thirty-two percent had bulky disease.
Investigators followed the patients for a median of 34.7 months (range, 1 day to 68.2 months).
Results after PET2
Seventy-seven patients were missing a second PET scan, mostly due to PET protocol violations of having to use the same scanner for the baseline and second scan and the same acquisition time.
“We were very strict on our quality control,” Dr Johnson said, “because we wished to make sure this was reproducible data.”
So the results after 2 cycles of ABVD treatment were based on 1137 patients.
PET-negative patients
More than 80% of patients were PET-negative after 2 cycles. Four hundred and sixty-nine patients were randomized to receive ABVD and 466 to AVD.
The groups were well-balanced in terms of median age, performance status, stage, B symptoms, bulky disease, and IPS score.
There was a significant excess of neutropenic fever (P=0.032) and infection (P=0.040) in those patients continuing on ABVD compared to AVD. And any hematologic toxicity was highly significantly different between the 2 arms (P<0.001).
“So we have demonstrated that continuing with bleomycin beyond cycle 2 is accompanied by significantly more toxicity,” Dr Johnson said.
Ninety-eight percent of patients in both cohorts received at least 6 cycles of therapy post-randomization.
At a median follow-up of 36.3 months, 65% of patients in the ABVD arm and 69% in the AVD arm achieved a complete remission (CR) or unconfirmed CR (CRu).
Fourteen patients died in each of the arms. Seven patients died of their disease in the AVD arm, compared with 1 in the ABVD arm. Slightly more patients died from toxicity in the ABVD arm.
The primary endpoint of PFS showed very little difference between the 2 arms. The 3-year PFS in the intent-to-treat analysis was 85.4% for patients in the ABVD arm and 84.4% for those in the AVD arm.
The investigators observed that the PFS of 85% was somewhat lower than the 95% PFS observed in the literature. So they looked at the association between baseline factors and PFS after negative PET2.
“And what stands out from this is that if you have high-stage disease at presentation, there is a slightly higher chance of treatment failure following a negative PET scan,” Dr Johnson said. “And you can see the trend here, from early stage disease up to advanced-stage disease, the PET scan becomes a less reliable indicator of result.”
The investigators also conducted a subgroup analysis of the PET2-negative patients and found there was no difference in outcome between treatment arms in patients with more advanced disease, with bulky disease, with a high IPS score, or according to the PET score.
“So we have not succeeded in finding any subgroup where it appears to be beneficial to continue bleomycin,” Dr Johnson said.
The OS rate was also the same between the 2 arms, at 97%.
PET2-positive patients
One hundred and seventy-four patients who were positive after the second PET scan received either BEACOPP for 14 days or escalated BEACOPP.
The percentage of patients who experienced grade 3-4 toxicities was largely similar between the 2 regimens, although the patients receiving escalated BEACOPP had more neutropenia (P=0.057), thrombocytopenia (P=0.001), and neutropenic fever (P=0.08).
In terms of efficacy, two-thirds of patients became PET-negative by the third PET scan, and 48% of patients achieved a CR or CRu.
Twenty-one patients died, 8 due to Hodgkin lymphoma.
The PFS was 66.0% in the BEACOPP-14 group and 71.1% in the escalated-BEACOPP group. The 3-year OS was 89.6% in the BEACOPP-14 group and 82.8% in the escalated-BEACOPP group.
For the entire group of 1214 patients, the 3-year PFS was 82.5%, and the OS was 95.4%.
Based on these results, the investigators concluded that it is safe to omit bleomycin and consolidation radiotherapy from subsequent ABVD therapy after a negative interim PET scan. And doing so reduces toxicity, especially dyspnea, thromboembolism, and neutropenic fever.
“[B]y using more selective chemotherapy and much less radiotherapy than we have previously used in our studies, where we’re giving less than 3% of patients consolidation radiotherapy, the results appear to be favorable and an improvement over what we have seen previously,” Dr Johnson said.
Details on lung toxicity in this study were presented separately at 13-ICML as abstract 041. ![]()

Photo by Jens Maus
LUGANO—Results of the RATHL trial indicate that bleomycin can be omitted from ABVD therapy following a negative interim FDG-PET scan in patients with Hodgkin lymphoma.
Progression-free survival (PFS) and overall survival (OS) were the same at 3 years for patients who were PET-negative after 2 cycles of ABVD and then continued therapy with or without bleomycin.
These results were presented at the 13th International Conference on Malignant Lymphoma (13-ICML).
Investigators based the RATHL study on the principles that it’s desirable to de-escalate treatment in the best responders to avoid late toxicity and that PET scans after 2 cycles of ABVD are highly predictive.
The team enrolled 1214 patients from 6 countries, 861 of whom were in the UK. Patients received a PET scan at staging, 2 cycles of ABVD, and then a second PET scan (PET2).
If patients were negative after PET2, they were randomized to receive 4 more cycles of ABVD or AVD and no radiotherapy.
If they were positive after PET2, patients received 4 cycles of BEACOPP-14 or 3 cycles of escalated BEACOPP. These patients then received a third PET scan, and the positive patients went on to receive radiotherapy or a salvage regimen.
The PET3-negative patients received 2 more cycles of BEACOPP-14 or one of escalated BEACOPP without radiotherapy.
Peter W. Johnson, MD, of the University of Southampton in the UK, presented the results of these treatment regimens during the plenary session of 13-ICML as abstract 008.
Patient characteristics
Patients were a median age of 33 (range, 18-79), and 55% were male. They had disease stages of II (41%), III (31%), or IV (28%).
Seventy-four percent of patients had a performance status of 0. Almost half (49%) had an IPS score of 2 to 3, and 18% had an IPS score of 4 or more. Thirty-two percent had bulky disease.
Investigators followed the patients for a median of 34.7 months (range, 1 day to 68.2 months).
Results after PET2
Seventy-seven patients were missing a second PET scan, mostly due to PET protocol violations of having to use the same scanner for the baseline and second scan and the same acquisition time.
“We were very strict on our quality control,” Dr Johnson said, “because we wished to make sure this was reproducible data.”
So the results after 2 cycles of ABVD treatment were based on 1137 patients.
PET-negative patients
More than 80% of patients were PET-negative after 2 cycles. Four hundred and sixty-nine patients were randomized to receive ABVD and 466 to AVD.
The groups were well-balanced in terms of median age, performance status, stage, B symptoms, bulky disease, and IPS score.
There was a significant excess of neutropenic fever (P=0.032) and infection (P=0.040) in those patients continuing on ABVD compared to AVD. And any hematologic toxicity was highly significantly different between the 2 arms (P<0.001).
“So we have demonstrated that continuing with bleomycin beyond cycle 2 is accompanied by significantly more toxicity,” Dr Johnson said.
Ninety-eight percent of patients in both cohorts received at least 6 cycles of therapy post-randomization.
At a median follow-up of 36.3 months, 65% of patients in the ABVD arm and 69% in the AVD arm achieved a complete remission (CR) or unconfirmed CR (CRu).
Fourteen patients died in each of the arms. Seven patients died of their disease in the AVD arm, compared with 1 in the ABVD arm. Slightly more patients died from toxicity in the ABVD arm.
The primary endpoint of PFS showed very little difference between the 2 arms. The 3-year PFS in the intent-to-treat analysis was 85.4% for patients in the ABVD arm and 84.4% for those in the AVD arm.
The investigators observed that the PFS of 85% was somewhat lower than the 95% PFS observed in the literature. So they looked at the association between baseline factors and PFS after negative PET2.
“And what stands out from this is that if you have high-stage disease at presentation, there is a slightly higher chance of treatment failure following a negative PET scan,” Dr Johnson said. “And you can see the trend here, from early stage disease up to advanced-stage disease, the PET scan becomes a less reliable indicator of result.”
The investigators also conducted a subgroup analysis of the PET2-negative patients and found there was no difference in outcome between treatment arms in patients with more advanced disease, with bulky disease, with a high IPS score, or according to the PET score.
“So we have not succeeded in finding any subgroup where it appears to be beneficial to continue bleomycin,” Dr Johnson said.
The OS rate was also the same between the 2 arms, at 97%.
PET2-positive patients
One hundred and seventy-four patients who were positive after the second PET scan received either BEACOPP for 14 days or escalated BEACOPP.
The percentage of patients who experienced grade 3-4 toxicities was largely similar between the 2 regimens, although the patients receiving escalated BEACOPP had more neutropenia (P=0.057), thrombocytopenia (P=0.001), and neutropenic fever (P=0.08).
In terms of efficacy, two-thirds of patients became PET-negative by the third PET scan, and 48% of patients achieved a CR or CRu.
Twenty-one patients died, 8 due to Hodgkin lymphoma.
The PFS was 66.0% in the BEACOPP-14 group and 71.1% in the escalated-BEACOPP group. The 3-year OS was 89.6% in the BEACOPP-14 group and 82.8% in the escalated-BEACOPP group.
For the entire group of 1214 patients, the 3-year PFS was 82.5%, and the OS was 95.4%.
Based on these results, the investigators concluded that it is safe to omit bleomycin and consolidation radiotherapy from subsequent ABVD therapy after a negative interim PET scan. And doing so reduces toxicity, especially dyspnea, thromboembolism, and neutropenic fever.
“[B]y using more selective chemotherapy and much less radiotherapy than we have previously used in our studies, where we’re giving less than 3% of patients consolidation radiotherapy, the results appear to be favorable and an improvement over what we have seen previously,” Dr Johnson said.
Details on lung toxicity in this study were presented separately at 13-ICML as abstract 041. ![]()
Alisertib shows activity in non-Hodgkin lymphoma
The novel Aurora A kinase inhibitor alisertib has shown significant antitumor activity in patients with relapsed or refractory peripheral T-cell non-Hodgkin lymphoma but not transformed mycosis fungoides, according to data from a phase II trial.
The study in 37 patients showed the overall treatment response rate among peripheral T-cell non-Hodgkin lymphoma (PTCL) was 30%, with two (7%) complete responses, seven (23%) partial responses, and five patients (17%) achieving stable disease, although there was no response from patients with transformed mycosis fungoides.
Grade 3 and 4 adverse events were observed in less than 5% of patients, the most common being neutropenia (32%), followed by anemia, thrombocytopenia, and febrile neutropenia; around half of all patients experienced fatigue, according to a study published online in the Journal of Clinical Oncology (J. Clin. Oncol. 2015 June 15 [doi:10.1200/JCO.2014.60.6327]).
“Frequent drug resistance and overall poor outcomes complicate the management of relapsed and refractory PTCL; the need for effective new agents with favorable toxicity profiles is clear,” wrote Dr. Paul M. Barr of the University of Rochester (N.Y.) and his coauthors.
The study was supported by the National Institutes of Health and the National Cancer Institute. Dr. Barr reported consulting or advisory roles with Millennium, Pharmacyclics, Gilead Sciences, and TG Therapeutics and research funding from Pharmacyclics.
The novel Aurora A kinase inhibitor alisertib has shown significant antitumor activity in patients with relapsed or refractory peripheral T-cell non-Hodgkin lymphoma but not transformed mycosis fungoides, according to data from a phase II trial.
The study in 37 patients showed the overall treatment response rate among peripheral T-cell non-Hodgkin lymphoma (PTCL) was 30%, with two (7%) complete responses, seven (23%) partial responses, and five patients (17%) achieving stable disease, although there was no response from patients with transformed mycosis fungoides.
Grade 3 and 4 adverse events were observed in less than 5% of patients, the most common being neutropenia (32%), followed by anemia, thrombocytopenia, and febrile neutropenia; around half of all patients experienced fatigue, according to a study published online in the Journal of Clinical Oncology (J. Clin. Oncol. 2015 June 15 [doi:10.1200/JCO.2014.60.6327]).
“Frequent drug resistance and overall poor outcomes complicate the management of relapsed and refractory PTCL; the need for effective new agents with favorable toxicity profiles is clear,” wrote Dr. Paul M. Barr of the University of Rochester (N.Y.) and his coauthors.
The study was supported by the National Institutes of Health and the National Cancer Institute. Dr. Barr reported consulting or advisory roles with Millennium, Pharmacyclics, Gilead Sciences, and TG Therapeutics and research funding from Pharmacyclics.
The novel Aurora A kinase inhibitor alisertib has shown significant antitumor activity in patients with relapsed or refractory peripheral T-cell non-Hodgkin lymphoma but not transformed mycosis fungoides, according to data from a phase II trial.
The study in 37 patients showed the overall treatment response rate among peripheral T-cell non-Hodgkin lymphoma (PTCL) was 30%, with two (7%) complete responses, seven (23%) partial responses, and five patients (17%) achieving stable disease, although there was no response from patients with transformed mycosis fungoides.
Grade 3 and 4 adverse events were observed in less than 5% of patients, the most common being neutropenia (32%), followed by anemia, thrombocytopenia, and febrile neutropenia; around half of all patients experienced fatigue, according to a study published online in the Journal of Clinical Oncology (J. Clin. Oncol. 2015 June 15 [doi:10.1200/JCO.2014.60.6327]).
“Frequent drug resistance and overall poor outcomes complicate the management of relapsed and refractory PTCL; the need for effective new agents with favorable toxicity profiles is clear,” wrote Dr. Paul M. Barr of the University of Rochester (N.Y.) and his coauthors.
The study was supported by the National Institutes of Health and the National Cancer Institute. Dr. Barr reported consulting or advisory roles with Millennium, Pharmacyclics, Gilead Sciences, and TG Therapeutics and research funding from Pharmacyclics.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Alisertib has shown significant antitumor activity in patients with relapsed or refractory peripheral T-cell non-Hodgkin lymphoma.
Major finding: Alisertib showed an overall response rate of 30% among patients with peripheral T-cell non-Hodgkin lymphoma.
Data source: A prospective phase II clinical trial in 37 patients with peripheral T-cell non-Hodgkin lymphoma or transformed mycosis fungoides.
Disclosures: The study was supported by the National Institutes of Health and the National Cancer Institute. Dr. Barr reported consulting or advisory roles with Millennium, Pharmacyclics, Gilead Sciences, and TG Therapeutics and research funding from Pharmacyclics.
Combo delays progression in relapsed CLL

VIENNA—Results of the COMPLEMENT 2 trial indicate that adding ofatumumab to treatment with fludarabine and cyclophosphamide (OFC) can improve some outcome measures in patients with relapsed chronic lymphocytic leukemia (CLL), when compared to fludarabine and cyclophosphamide alone (FC).
Patients who received OFC had a significantly higher overall response rate and longer median progression-free survival than patients who received FC.
On the other hand, there was no significant difference between the treatment arms with regard to response duration or overall survival. And there were more grade 3 or higher adverse events (AEs) in the 3-drug arm than the 2-drug arm.
“There are limited treatment options for patients who have stopped responding to current CLL treatments, which happens in many patients with this disease over time,” said study investigator Tadeusz Robak, MD, PhD, of the Medical University of Lodz and Copernicus Memorial Hospital in Lodz, Poland.
“These data showed that the addition of ofatumumab to fludarabine and cyclophosphamide extended the amount of time before a patient’s CLL progressed, and further add to the body of evidence supporting the potential use of ofatumumab for these patients.”
The data were presented at the 20th Congress of the European Hematology Association (abstract LB219). The study was sponsored by GlaxoSmithKline and Genmab, which were previously co-developing ofatumumab. The drug is now an asset of Novartis AG.
Efficacy data
COMPLEMENT 2 is a phase 3 trial of 365 patients with relapsed CLL. Patients were randomized 1:1 to receive treatment with up to 6 cycles of OFC or FC. Baseline characteristics were well-balanced between the treatment arms.
The overall response rate was higher in the OFC arm than the FC arm—84% and 68%, respectively (P=0.0003)—as was the complete response rate—27% and 7%, respectively.
However, there was no significant difference in time to response or response duration. The median duration of response was 29.6 months in the OFC arm and 24.9 months in the FC arm (P=0.0878). And the median time to response was 0.99 months in both arms (P=0.449).
Still, patients in the OFC arm experienced a 54% improvement in progression-free survival. The median progression-free survival was 28.9 months in the OFC arm and 18.8 months in the FC arm (P=0.0032). And time to progression was 42.1 months and 26.8 months, respectively (P=0.0036).
But there was no significant difference in overall survival or time to next cancer treatment between the arms. The median overall survival was 56.4 months in the OFC arm and 45.8 months in the FC arm (P=0.1410). The median time to next therapy was 48.13 months and 40.08 months, respectively (P=0.0735).
Safety data
The rate of treatment-related AEs was 93% in the OFC arm and 85% in the FC arm. The rate of grade 3 or higher AEs was 74% and 69%, respectively.
The most common treatment-related AEs occurring in the OFC and FC arms, respectively, were neutropenia (58% vs 41%), thrombocytopenia (26% vs 32%), anemia (15% vs 26%), nausea (19% in both), leukopenia (14% vs 6%), vomiting (7% vs 10%), pyrexia (10% vs 3%), rash (10% vs 2%), fatigue (6% vs 4%), and pneumonia (6% vs 4%).
Treatment-related infections occurred in 20% of patients in the OFC arm and 15% in the FC arm. Infusion reactions occurred in 60% and 28% of patients, respectively. ![]()

VIENNA—Results of the COMPLEMENT 2 trial indicate that adding ofatumumab to treatment with fludarabine and cyclophosphamide (OFC) can improve some outcome measures in patients with relapsed chronic lymphocytic leukemia (CLL), when compared to fludarabine and cyclophosphamide alone (FC).
Patients who received OFC had a significantly higher overall response rate and longer median progression-free survival than patients who received FC.
On the other hand, there was no significant difference between the treatment arms with regard to response duration or overall survival. And there were more grade 3 or higher adverse events (AEs) in the 3-drug arm than the 2-drug arm.
“There are limited treatment options for patients who have stopped responding to current CLL treatments, which happens in many patients with this disease over time,” said study investigator Tadeusz Robak, MD, PhD, of the Medical University of Lodz and Copernicus Memorial Hospital in Lodz, Poland.
“These data showed that the addition of ofatumumab to fludarabine and cyclophosphamide extended the amount of time before a patient’s CLL progressed, and further add to the body of evidence supporting the potential use of ofatumumab for these patients.”
The data were presented at the 20th Congress of the European Hematology Association (abstract LB219). The study was sponsored by GlaxoSmithKline and Genmab, which were previously co-developing ofatumumab. The drug is now an asset of Novartis AG.
Efficacy data
COMPLEMENT 2 is a phase 3 trial of 365 patients with relapsed CLL. Patients were randomized 1:1 to receive treatment with up to 6 cycles of OFC or FC. Baseline characteristics were well-balanced between the treatment arms.
The overall response rate was higher in the OFC arm than the FC arm—84% and 68%, respectively (P=0.0003)—as was the complete response rate—27% and 7%, respectively.
However, there was no significant difference in time to response or response duration. The median duration of response was 29.6 months in the OFC arm and 24.9 months in the FC arm (P=0.0878). And the median time to response was 0.99 months in both arms (P=0.449).
Still, patients in the OFC arm experienced a 54% improvement in progression-free survival. The median progression-free survival was 28.9 months in the OFC arm and 18.8 months in the FC arm (P=0.0032). And time to progression was 42.1 months and 26.8 months, respectively (P=0.0036).
But there was no significant difference in overall survival or time to next cancer treatment between the arms. The median overall survival was 56.4 months in the OFC arm and 45.8 months in the FC arm (P=0.1410). The median time to next therapy was 48.13 months and 40.08 months, respectively (P=0.0735).
Safety data
The rate of treatment-related AEs was 93% in the OFC arm and 85% in the FC arm. The rate of grade 3 or higher AEs was 74% and 69%, respectively.
The most common treatment-related AEs occurring in the OFC and FC arms, respectively, were neutropenia (58% vs 41%), thrombocytopenia (26% vs 32%), anemia (15% vs 26%), nausea (19% in both), leukopenia (14% vs 6%), vomiting (7% vs 10%), pyrexia (10% vs 3%), rash (10% vs 2%), fatigue (6% vs 4%), and pneumonia (6% vs 4%).
Treatment-related infections occurred in 20% of patients in the OFC arm and 15% in the FC arm. Infusion reactions occurred in 60% and 28% of patients, respectively. ![]()

VIENNA—Results of the COMPLEMENT 2 trial indicate that adding ofatumumab to treatment with fludarabine and cyclophosphamide (OFC) can improve some outcome measures in patients with relapsed chronic lymphocytic leukemia (CLL), when compared to fludarabine and cyclophosphamide alone (FC).
Patients who received OFC had a significantly higher overall response rate and longer median progression-free survival than patients who received FC.
On the other hand, there was no significant difference between the treatment arms with regard to response duration or overall survival. And there were more grade 3 or higher adverse events (AEs) in the 3-drug arm than the 2-drug arm.
“There are limited treatment options for patients who have stopped responding to current CLL treatments, which happens in many patients with this disease over time,” said study investigator Tadeusz Robak, MD, PhD, of the Medical University of Lodz and Copernicus Memorial Hospital in Lodz, Poland.
“These data showed that the addition of ofatumumab to fludarabine and cyclophosphamide extended the amount of time before a patient’s CLL progressed, and further add to the body of evidence supporting the potential use of ofatumumab for these patients.”
The data were presented at the 20th Congress of the European Hematology Association (abstract LB219). The study was sponsored by GlaxoSmithKline and Genmab, which were previously co-developing ofatumumab. The drug is now an asset of Novartis AG.
Efficacy data
COMPLEMENT 2 is a phase 3 trial of 365 patients with relapsed CLL. Patients were randomized 1:1 to receive treatment with up to 6 cycles of OFC or FC. Baseline characteristics were well-balanced between the treatment arms.
The overall response rate was higher in the OFC arm than the FC arm—84% and 68%, respectively (P=0.0003)—as was the complete response rate—27% and 7%, respectively.
However, there was no significant difference in time to response or response duration. The median duration of response was 29.6 months in the OFC arm and 24.9 months in the FC arm (P=0.0878). And the median time to response was 0.99 months in both arms (P=0.449).
Still, patients in the OFC arm experienced a 54% improvement in progression-free survival. The median progression-free survival was 28.9 months in the OFC arm and 18.8 months in the FC arm (P=0.0032). And time to progression was 42.1 months and 26.8 months, respectively (P=0.0036).
But there was no significant difference in overall survival or time to next cancer treatment between the arms. The median overall survival was 56.4 months in the OFC arm and 45.8 months in the FC arm (P=0.1410). The median time to next therapy was 48.13 months and 40.08 months, respectively (P=0.0735).
Safety data
The rate of treatment-related AEs was 93% in the OFC arm and 85% in the FC arm. The rate of grade 3 or higher AEs was 74% and 69%, respectively.
The most common treatment-related AEs occurring in the OFC and FC arms, respectively, were neutropenia (58% vs 41%), thrombocytopenia (26% vs 32%), anemia (15% vs 26%), nausea (19% in both), leukopenia (14% vs 6%), vomiting (7% vs 10%), pyrexia (10% vs 3%), rash (10% vs 2%), fatigue (6% vs 4%), and pneumonia (6% vs 4%).
Treatment-related infections occurred in 20% of patients in the OFC arm and 15% in the FC arm. Infusion reactions occurred in 60% and 28% of patients, respectively. ![]()
EHA: Inotuzumab rallies against refractory/relapsed ALL
VIENNA – The investigational agent inotuzumab ozagamicin more than doubled complete remission rates compared with standard therapy in relapsed or refractory acute lymphoblastic leukemia, preliminary results from the INO-VATE study show.
The co-primary endpoint of complete remission or CR with incomplete hematologic recovery (CRi) by independent review was achieved by 80.7% of patients treated with inotuzumab and 33.3% treated with standard of care (SOC) (P < .0001).
Significantly more CR/CRi responders treated with inotuzumab were minimal residual disease (MRD)-negative by multicolor flow cytometry (78.4% vs. 28.1%; P < .0001), Dr. Daniel DeAngelo reported in a late-breaking abstract (LBA2073) at the annual congress of the European Hematology Association.
“The fact that the response rate was astronomically high with a high MRD-negative status really allows this or this should be an opportunity for patients with relapsed/refractory disease,” he said in an interview.
Inotuzumab ozagamicin is an investigational anti-CD22 antibody conjugated to calicheamicin, an antitumor antibiotic. CD22 is expressed on the surface of about 90% of B-cell ALL cells.
Previous phase II studies reported strong initial antitumor activity and safety with inotuzumab in relapsed or refractory ALL, Dr. DeAngelo, of the Dana Farber Cancer Institute in Boston, said.
The ongoing phase III trial randomized 326 patients with relapsed/refractory CD22-positive ALL due for salvage 1 or 2 therapy to inotuzumab or SOC: either the FLAG regimen (fludarabine (Fludara)/cytarabine (Ara-C)/granulocyte colony-stimulating factor), Ara-C plus mitoxantrone (Novantrone), or high-dose Ara-C. The starting dose for inotuzumab was 1.8 mg/m2/cycle and was reduced to 1.5 mg/m2/cycle once CR/CRi was achieved. Patients were stratified by duration of first remission, salvage 1 or 2, and age.
The first 218 randomized patients were included in the intention-to-treat CR/CRi analysis, which was modified after excluding 13 patients from the SOC arm who refused to start treatment.
The patients’ median age was 47 years (ranging up to 79 years), two-thirds were salvage 1, and more than half had a remission duration of less than 12 months, an adverse prognostic feature.
Data for the co-primary endpoint of overall survival in all 326 patients are still blinded and not expected to mature until 2016, Dr. DeAngelo said.
CR/CRi analyses significantly favored inotuzumab in all stratification factors and baseline factors including peripheral blasts and CD22 expression. Cytogenetics are still being evaluated, but 11 of 14 (79%) patients with Philadelphia-positive karyotype achieved a CR or CRi, he said.
Median duration of remission among responders was 4.6 months in the inotuzumab arm and 3.1 months in the SOC arm (hazard ratio, 0.55; P = .016).
Safety assessed in 259 patients who received at least one dose of study drug showed similar incidence of grade 3 or higher adverse events in the inotuzumab and SOC arms (91% vs. 95%). There were 2 fatal events in the SOC arm and 4 in the inotuzumab arm: 2 veno-occlusive disease (VOD)/sinusoidal obstruction syndrome (SOS), both after poststudy transplant, 1 intestinal ischemia/septic shock, and 1 acute respiratory distress syndrome as a terminal event of pneumonia. In multivariate analysis, dual alkylator conditioning was the only significant covariate of VOD/SOS (P = .039), Dr. DeAngelo said.
An audience member chided the author for the short duration of remission, but session co-moderator Dr. Anthony Moorman, of Newcastle University, Newcastle upon Tyne, England, said it is not that concerning because of the aggressive nature of ALL.
“For all patients that have relapsed or refractory adult ALL, their responses are incredibly low. So any kind of complete remission is a major achievement in this patient population, especially if they are refractory or relapse after tyrosine kinase inhibitors or Philadelphia-positive,” he said in an interview.
“When you have an active agent that works with relapsed refractory disease, in this case leukemia, the goal is to move it up front,” Dr. DeAngelo told this publication.
Indeed, updated results presented at the meeting from M.D. Anderson Cancer Center of frontline inotuzumab added to low-intensity chemotherapy (Mini-hyper CVD) in elderly ALL patients were “provocative,” he added. CR rates reached 97% in the study, according to the abstract (S114).
VIENNA – The investigational agent inotuzumab ozagamicin more than doubled complete remission rates compared with standard therapy in relapsed or refractory acute lymphoblastic leukemia, preliminary results from the INO-VATE study show.
The co-primary endpoint of complete remission or CR with incomplete hematologic recovery (CRi) by independent review was achieved by 80.7% of patients treated with inotuzumab and 33.3% treated with standard of care (SOC) (P < .0001).
Significantly more CR/CRi responders treated with inotuzumab were minimal residual disease (MRD)-negative by multicolor flow cytometry (78.4% vs. 28.1%; P < .0001), Dr. Daniel DeAngelo reported in a late-breaking abstract (LBA2073) at the annual congress of the European Hematology Association.
“The fact that the response rate was astronomically high with a high MRD-negative status really allows this or this should be an opportunity for patients with relapsed/refractory disease,” he said in an interview.
Inotuzumab ozagamicin is an investigational anti-CD22 antibody conjugated to calicheamicin, an antitumor antibiotic. CD22 is expressed on the surface of about 90% of B-cell ALL cells.
Previous phase II studies reported strong initial antitumor activity and safety with inotuzumab in relapsed or refractory ALL, Dr. DeAngelo, of the Dana Farber Cancer Institute in Boston, said.
The ongoing phase III trial randomized 326 patients with relapsed/refractory CD22-positive ALL due for salvage 1 or 2 therapy to inotuzumab or SOC: either the FLAG regimen (fludarabine (Fludara)/cytarabine (Ara-C)/granulocyte colony-stimulating factor), Ara-C plus mitoxantrone (Novantrone), or high-dose Ara-C. The starting dose for inotuzumab was 1.8 mg/m2/cycle and was reduced to 1.5 mg/m2/cycle once CR/CRi was achieved. Patients were stratified by duration of first remission, salvage 1 or 2, and age.
The first 218 randomized patients were included in the intention-to-treat CR/CRi analysis, which was modified after excluding 13 patients from the SOC arm who refused to start treatment.
The patients’ median age was 47 years (ranging up to 79 years), two-thirds were salvage 1, and more than half had a remission duration of less than 12 months, an adverse prognostic feature.
Data for the co-primary endpoint of overall survival in all 326 patients are still blinded and not expected to mature until 2016, Dr. DeAngelo said.
CR/CRi analyses significantly favored inotuzumab in all stratification factors and baseline factors including peripheral blasts and CD22 expression. Cytogenetics are still being evaluated, but 11 of 14 (79%) patients with Philadelphia-positive karyotype achieved a CR or CRi, he said.
Median duration of remission among responders was 4.6 months in the inotuzumab arm and 3.1 months in the SOC arm (hazard ratio, 0.55; P = .016).
Safety assessed in 259 patients who received at least one dose of study drug showed similar incidence of grade 3 or higher adverse events in the inotuzumab and SOC arms (91% vs. 95%). There were 2 fatal events in the SOC arm and 4 in the inotuzumab arm: 2 veno-occlusive disease (VOD)/sinusoidal obstruction syndrome (SOS), both after poststudy transplant, 1 intestinal ischemia/septic shock, and 1 acute respiratory distress syndrome as a terminal event of pneumonia. In multivariate analysis, dual alkylator conditioning was the only significant covariate of VOD/SOS (P = .039), Dr. DeAngelo said.
An audience member chided the author for the short duration of remission, but session co-moderator Dr. Anthony Moorman, of Newcastle University, Newcastle upon Tyne, England, said it is not that concerning because of the aggressive nature of ALL.
“For all patients that have relapsed or refractory adult ALL, their responses are incredibly low. So any kind of complete remission is a major achievement in this patient population, especially if they are refractory or relapse after tyrosine kinase inhibitors or Philadelphia-positive,” he said in an interview.
“When you have an active agent that works with relapsed refractory disease, in this case leukemia, the goal is to move it up front,” Dr. DeAngelo told this publication.
Indeed, updated results presented at the meeting from M.D. Anderson Cancer Center of frontline inotuzumab added to low-intensity chemotherapy (Mini-hyper CVD) in elderly ALL patients were “provocative,” he added. CR rates reached 97% in the study, according to the abstract (S114).
VIENNA – The investigational agent inotuzumab ozagamicin more than doubled complete remission rates compared with standard therapy in relapsed or refractory acute lymphoblastic leukemia, preliminary results from the INO-VATE study show.
The co-primary endpoint of complete remission or CR with incomplete hematologic recovery (CRi) by independent review was achieved by 80.7% of patients treated with inotuzumab and 33.3% treated with standard of care (SOC) (P < .0001).
Significantly more CR/CRi responders treated with inotuzumab were minimal residual disease (MRD)-negative by multicolor flow cytometry (78.4% vs. 28.1%; P < .0001), Dr. Daniel DeAngelo reported in a late-breaking abstract (LBA2073) at the annual congress of the European Hematology Association.
“The fact that the response rate was astronomically high with a high MRD-negative status really allows this or this should be an opportunity for patients with relapsed/refractory disease,” he said in an interview.
Inotuzumab ozagamicin is an investigational anti-CD22 antibody conjugated to calicheamicin, an antitumor antibiotic. CD22 is expressed on the surface of about 90% of B-cell ALL cells.
Previous phase II studies reported strong initial antitumor activity and safety with inotuzumab in relapsed or refractory ALL, Dr. DeAngelo, of the Dana Farber Cancer Institute in Boston, said.
The ongoing phase III trial randomized 326 patients with relapsed/refractory CD22-positive ALL due for salvage 1 or 2 therapy to inotuzumab or SOC: either the FLAG regimen (fludarabine (Fludara)/cytarabine (Ara-C)/granulocyte colony-stimulating factor), Ara-C plus mitoxantrone (Novantrone), or high-dose Ara-C. The starting dose for inotuzumab was 1.8 mg/m2/cycle and was reduced to 1.5 mg/m2/cycle once CR/CRi was achieved. Patients were stratified by duration of first remission, salvage 1 or 2, and age.
The first 218 randomized patients were included in the intention-to-treat CR/CRi analysis, which was modified after excluding 13 patients from the SOC arm who refused to start treatment.
The patients’ median age was 47 years (ranging up to 79 years), two-thirds were salvage 1, and more than half had a remission duration of less than 12 months, an adverse prognostic feature.
Data for the co-primary endpoint of overall survival in all 326 patients are still blinded and not expected to mature until 2016, Dr. DeAngelo said.
CR/CRi analyses significantly favored inotuzumab in all stratification factors and baseline factors including peripheral blasts and CD22 expression. Cytogenetics are still being evaluated, but 11 of 14 (79%) patients with Philadelphia-positive karyotype achieved a CR or CRi, he said.
Median duration of remission among responders was 4.6 months in the inotuzumab arm and 3.1 months in the SOC arm (hazard ratio, 0.55; P = .016).
Safety assessed in 259 patients who received at least one dose of study drug showed similar incidence of grade 3 or higher adverse events in the inotuzumab and SOC arms (91% vs. 95%). There were 2 fatal events in the SOC arm and 4 in the inotuzumab arm: 2 veno-occlusive disease (VOD)/sinusoidal obstruction syndrome (SOS), both after poststudy transplant, 1 intestinal ischemia/septic shock, and 1 acute respiratory distress syndrome as a terminal event of pneumonia. In multivariate analysis, dual alkylator conditioning was the only significant covariate of VOD/SOS (P = .039), Dr. DeAngelo said.
An audience member chided the author for the short duration of remission, but session co-moderator Dr. Anthony Moorman, of Newcastle University, Newcastle upon Tyne, England, said it is not that concerning because of the aggressive nature of ALL.
“For all patients that have relapsed or refractory adult ALL, their responses are incredibly low. So any kind of complete remission is a major achievement in this patient population, especially if they are refractory or relapse after tyrosine kinase inhibitors or Philadelphia-positive,” he said in an interview.
“When you have an active agent that works with relapsed refractory disease, in this case leukemia, the goal is to move it up front,” Dr. DeAngelo told this publication.
Indeed, updated results presented at the meeting from M.D. Anderson Cancer Center of frontline inotuzumab added to low-intensity chemotherapy (Mini-hyper CVD) in elderly ALL patients were “provocative,” he added. CR rates reached 97% in the study, according to the abstract (S114).
AT THE EHA CONGRESS
Key clinical point: Inotuzumab ozagamicin shows promise as a new treatment option for relapsed or refractory acute lymphoblastic leukemia.
Major finding: The rate of complete remission or CR with incomplete hematologic recovery was 80.7% with inotuzumab vs. 28.1% with standard of care (P < .0001).
Data source: Randomized, phase III study in the first 218 of 326 patients.
Disclosures: Pfizer sponsored the study and funded editorial assistance supplied by Complete Heathcare Communications. Dr. De Angelo reported research support from Sigma Tau and consulting for Novartis, Sigma Tau, Bristol-Myers Squibb, Amgen, and Pfizer.
EHA: Dasatinib gets early edge over imatinib in CML
VIENNA – Patients with chronic-phase chronic myeloid leukemia treated with first-line dasatinib achieved significantly more molecular responses at 2 years than those treated with imatinib in the SPIRIT 2 trial.
So far there is no difference, however, in disease progression or overall survival in the ongoing phase III trial, Dr. Stephen O’Brien reported at the annual congress of the European Hematology Association.
With 814 patients, SPIRIT 2 is the largest randomized trial of dasatinib (Sprycel) vs. imatinib (Gleevec).
Its design is similar to the ongoing 519-patient DASISION trial, which reported higher response rates with dasatinib than imatinib in the same setting, but similar progression-free and overall survival rates at 3-year follow-up.
The primary endpoint of SPIRIT 2 is event-free survival at 5 years and will be available in March 2018, he said. Patients at 172 hospitals in the United Kingdom were evenly randomized to imatinib 400 mg daily or dasatinib 100 mg daily. One patient in each group was excluded due to protocol violation or withdrawal of consent. Median follow-up is 42.4 months.
At 24 months, 60.6% of imatinib patients (246/406) and 71.4% of dasatinib patients (290/406) remained on treatment.
Significantly more patients treated with dasatinib than imatinib achieved a complete cytogenetic response at 12 months (53.3% vs. 42%; P = .003), but the difference was diminished at 24 months (33.7% vs. 27.5%; P = .189). These results should be interpreted with caution, however, because the data were incomplete, Dr. O’Brien, of Newcastle University Medical School, Newcastle upon Tyne, England, said.
He noted that the molecular data are more reliable and were calculated based on samples drawn within a 6-week window on either side of the 24-month time point. Values had to be imputed for 22 patients who had no 24-month sample taken, although this imputation should not impact survival outcomes, he said. Major molecular response was defined as a 3-log reduction in the BCR-ABL/ABL ratio, relative to baseline, with data also captured for patients achieving a 4-log reduction.
Significantly more patients on dasatinib than imatinib achieved an MR3 response (57.5% vs. 46%; P < .001) and MR4.5 response (20.2% vs. 14.3%; P = .026).
More patients stopped imatinib than dasatinib due to investigator and/or patient concerns about inadequate response (10.8% vs. 1.3%), whereas nonhematologic toxicities drove more patients to abandon dasatinib (22% vs. 12%), according to Dr. O’Brien.
Pleural effusion, a known toxicity with dasatinib, occurred in 24.1% of patients given the drug vs. 1.2% given imatinib, requiring drainage in 22 cases vs. 1 case, respectively. There was also a “difficult-to-explain” signal for breathlessness with no obvious cause (15.5% vs. 8%). Hypertension was confirmed in only one of these cases and symptoms resolved in others when the drug was withdrawn, he said.
Serious cardiac adverse events were reported in 2.2% of patients in the imatinib arm and 4.2% in the dasatinib arm. Again, the results should be interpreted with caution because trials set up at the time of SPIRIT2 in 2008 were not designed to look carefully at this outcome, Dr. O’Brien observed.
In all, 38 patients have died; 19 in each group.
*Correction 6/18/2015: The headline for an earlier version of this article misstated the type of cancer treated in this study.
VIENNA – Patients with chronic-phase chronic myeloid leukemia treated with first-line dasatinib achieved significantly more molecular responses at 2 years than those treated with imatinib in the SPIRIT 2 trial.
So far there is no difference, however, in disease progression or overall survival in the ongoing phase III trial, Dr. Stephen O’Brien reported at the annual congress of the European Hematology Association.
With 814 patients, SPIRIT 2 is the largest randomized trial of dasatinib (Sprycel) vs. imatinib (Gleevec).
Its design is similar to the ongoing 519-patient DASISION trial, which reported higher response rates with dasatinib than imatinib in the same setting, but similar progression-free and overall survival rates at 3-year follow-up.
The primary endpoint of SPIRIT 2 is event-free survival at 5 years and will be available in March 2018, he said. Patients at 172 hospitals in the United Kingdom were evenly randomized to imatinib 400 mg daily or dasatinib 100 mg daily. One patient in each group was excluded due to protocol violation or withdrawal of consent. Median follow-up is 42.4 months.
At 24 months, 60.6% of imatinib patients (246/406) and 71.4% of dasatinib patients (290/406) remained on treatment.
Significantly more patients treated with dasatinib than imatinib achieved a complete cytogenetic response at 12 months (53.3% vs. 42%; P = .003), but the difference was diminished at 24 months (33.7% vs. 27.5%; P = .189). These results should be interpreted with caution, however, because the data were incomplete, Dr. O’Brien, of Newcastle University Medical School, Newcastle upon Tyne, England, said.
He noted that the molecular data are more reliable and were calculated based on samples drawn within a 6-week window on either side of the 24-month time point. Values had to be imputed for 22 patients who had no 24-month sample taken, although this imputation should not impact survival outcomes, he said. Major molecular response was defined as a 3-log reduction in the BCR-ABL/ABL ratio, relative to baseline, with data also captured for patients achieving a 4-log reduction.
Significantly more patients on dasatinib than imatinib achieved an MR3 response (57.5% vs. 46%; P < .001) and MR4.5 response (20.2% vs. 14.3%; P = .026).
More patients stopped imatinib than dasatinib due to investigator and/or patient concerns about inadequate response (10.8% vs. 1.3%), whereas nonhematologic toxicities drove more patients to abandon dasatinib (22% vs. 12%), according to Dr. O’Brien.
Pleural effusion, a known toxicity with dasatinib, occurred in 24.1% of patients given the drug vs. 1.2% given imatinib, requiring drainage in 22 cases vs. 1 case, respectively. There was also a “difficult-to-explain” signal for breathlessness with no obvious cause (15.5% vs. 8%). Hypertension was confirmed in only one of these cases and symptoms resolved in others when the drug was withdrawn, he said.
Serious cardiac adverse events were reported in 2.2% of patients in the imatinib arm and 4.2% in the dasatinib arm. Again, the results should be interpreted with caution because trials set up at the time of SPIRIT2 in 2008 were not designed to look carefully at this outcome, Dr. O’Brien observed.
In all, 38 patients have died; 19 in each group.
*Correction 6/18/2015: The headline for an earlier version of this article misstated the type of cancer treated in this study.
VIENNA – Patients with chronic-phase chronic myeloid leukemia treated with first-line dasatinib achieved significantly more molecular responses at 2 years than those treated with imatinib in the SPIRIT 2 trial.
So far there is no difference, however, in disease progression or overall survival in the ongoing phase III trial, Dr. Stephen O’Brien reported at the annual congress of the European Hematology Association.
With 814 patients, SPIRIT 2 is the largest randomized trial of dasatinib (Sprycel) vs. imatinib (Gleevec).
Its design is similar to the ongoing 519-patient DASISION trial, which reported higher response rates with dasatinib than imatinib in the same setting, but similar progression-free and overall survival rates at 3-year follow-up.
The primary endpoint of SPIRIT 2 is event-free survival at 5 years and will be available in March 2018, he said. Patients at 172 hospitals in the United Kingdom were evenly randomized to imatinib 400 mg daily or dasatinib 100 mg daily. One patient in each group was excluded due to protocol violation or withdrawal of consent. Median follow-up is 42.4 months.
At 24 months, 60.6% of imatinib patients (246/406) and 71.4% of dasatinib patients (290/406) remained on treatment.
Significantly more patients treated with dasatinib than imatinib achieved a complete cytogenetic response at 12 months (53.3% vs. 42%; P = .003), but the difference was diminished at 24 months (33.7% vs. 27.5%; P = .189). These results should be interpreted with caution, however, because the data were incomplete, Dr. O’Brien, of Newcastle University Medical School, Newcastle upon Tyne, England, said.
He noted that the molecular data are more reliable and were calculated based on samples drawn within a 6-week window on either side of the 24-month time point. Values had to be imputed for 22 patients who had no 24-month sample taken, although this imputation should not impact survival outcomes, he said. Major molecular response was defined as a 3-log reduction in the BCR-ABL/ABL ratio, relative to baseline, with data also captured for patients achieving a 4-log reduction.
Significantly more patients on dasatinib than imatinib achieved an MR3 response (57.5% vs. 46%; P < .001) and MR4.5 response (20.2% vs. 14.3%; P = .026).
More patients stopped imatinib than dasatinib due to investigator and/or patient concerns about inadequate response (10.8% vs. 1.3%), whereas nonhematologic toxicities drove more patients to abandon dasatinib (22% vs. 12%), according to Dr. O’Brien.
Pleural effusion, a known toxicity with dasatinib, occurred in 24.1% of patients given the drug vs. 1.2% given imatinib, requiring drainage in 22 cases vs. 1 case, respectively. There was also a “difficult-to-explain” signal for breathlessness with no obvious cause (15.5% vs. 8%). Hypertension was confirmed in only one of these cases and symptoms resolved in others when the drug was withdrawn, he said.
Serious cardiac adverse events were reported in 2.2% of patients in the imatinib arm and 4.2% in the dasatinib arm. Again, the results should be interpreted with caution because trials set up at the time of SPIRIT2 in 2008 were not designed to look carefully at this outcome, Dr. O’Brien observed.
In all, 38 patients have died; 19 in each group.
*Correction 6/18/2015: The headline for an earlier version of this article misstated the type of cancer treated in this study.
AT THE EHA CONGRESS
Key clinical point: Dasatinib provides more molecular responses than imatinib, but no survival advantage at 2 years in the first-line treatment of chronic-phase chronic myeloid leukemia.
Major finding: More patients receiving dasatinib than imatinib achieved an MR3 response (57.5% vs. 46%; P < .001) and MR4.5 response (20.2% vs. 6%; P = .02).
Data source: Randomized, phase III trial in 814 patients with newly diagnosed chronic myeloid leukemia in chronic phase.
Disclosures: Bristol-Myers Squibb sponsored the study. Dr. O’Brien reported honoraria and research funding from Ariad Pharmaceuticals, Bristol-Myers Squibb, Novartis, and Pfizer.
Viral protein protects EBV-infected B cells
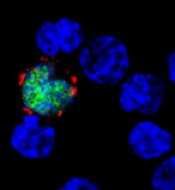
expresses the ligand (red)
that activates NKG2D, while
uninfected cells (blue) do not
Benjamin Chaigne-Delalande
A study published in PLOS Pathogens sheds new light on why the immune system cannot eliminate Epstein-Barr virus (EBV) or the risk of cancer associated with the virus.
Researchers investigated the immune system’s response against EBV, focusing on the role of LMP2A.
This viral protein is present in latently infected B cells and in many EBV-associated cancers, which have somehow escaped detection and elimination by the immune system.
Andreas Moosmann, PhD, of the Helmholtz-Zentrum in Munich, Germany, and his colleagues studied an engineered EBV virus that cannot make LMP2A and compared this mutant virus with the normal one.
The researchers infected human B cells with normal and LMP2A-deficient EBV. Because EBV transforms these cells, the team was able to examine lymphoblastic cell lines that contained either virus.
They found that LMP2A counteracts the recognition of EBV-infected B cells by EBV-specific, CD8+ killer T cells. In contrast, EBV-transformed cells without LMP2A are more efficiently identified, and the T cells’ ability to recognize and kill the EBV-infected B cells is enhanced.
The researchers examined the mechanism underlying the LMP2A-mediated evasion and found several ways in which it interferes with the recognition of EBV-infected cells.
First, LMP2A reduced the levels of several EBV proteins whose fragments are recognized by CD8+ T cells on the surface of the cell targeted for killing.
Second, LMP2A disturbs the expression of cellular molecules on infected B cells that interact with NKG2D, a host molecule on the surface of CD8+ T cells that aids their activation, thereby weakening the immune response against EBV-infected cells.
The researchers said these results suggest a functional immunomodulatory effect for the EBV protein LMP2A and show that LMP2A mediates the partial escape of infected B cells from recognition by CD8+ T cells.
The team also said similar immune evasion mechanisms may operate in different types of LMP2A-expressing cancers caused by EBV. ![]()

expresses the ligand (red)
that activates NKG2D, while
uninfected cells (blue) do not
Benjamin Chaigne-Delalande
A study published in PLOS Pathogens sheds new light on why the immune system cannot eliminate Epstein-Barr virus (EBV) or the risk of cancer associated with the virus.
Researchers investigated the immune system’s response against EBV, focusing on the role of LMP2A.
This viral protein is present in latently infected B cells and in many EBV-associated cancers, which have somehow escaped detection and elimination by the immune system.
Andreas Moosmann, PhD, of the Helmholtz-Zentrum in Munich, Germany, and his colleagues studied an engineered EBV virus that cannot make LMP2A and compared this mutant virus with the normal one.
The researchers infected human B cells with normal and LMP2A-deficient EBV. Because EBV transforms these cells, the team was able to examine lymphoblastic cell lines that contained either virus.
They found that LMP2A counteracts the recognition of EBV-infected B cells by EBV-specific, CD8+ killer T cells. In contrast, EBV-transformed cells without LMP2A are more efficiently identified, and the T cells’ ability to recognize and kill the EBV-infected B cells is enhanced.
The researchers examined the mechanism underlying the LMP2A-mediated evasion and found several ways in which it interferes with the recognition of EBV-infected cells.
First, LMP2A reduced the levels of several EBV proteins whose fragments are recognized by CD8+ T cells on the surface of the cell targeted for killing.
Second, LMP2A disturbs the expression of cellular molecules on infected B cells that interact with NKG2D, a host molecule on the surface of CD8+ T cells that aids their activation, thereby weakening the immune response against EBV-infected cells.
The researchers said these results suggest a functional immunomodulatory effect for the EBV protein LMP2A and show that LMP2A mediates the partial escape of infected B cells from recognition by CD8+ T cells.
The team also said similar immune evasion mechanisms may operate in different types of LMP2A-expressing cancers caused by EBV. ![]()

expresses the ligand (red)
that activates NKG2D, while
uninfected cells (blue) do not
Benjamin Chaigne-Delalande
A study published in PLOS Pathogens sheds new light on why the immune system cannot eliminate Epstein-Barr virus (EBV) or the risk of cancer associated with the virus.
Researchers investigated the immune system’s response against EBV, focusing on the role of LMP2A.
This viral protein is present in latently infected B cells and in many EBV-associated cancers, which have somehow escaped detection and elimination by the immune system.
Andreas Moosmann, PhD, of the Helmholtz-Zentrum in Munich, Germany, and his colleagues studied an engineered EBV virus that cannot make LMP2A and compared this mutant virus with the normal one.
The researchers infected human B cells with normal and LMP2A-deficient EBV. Because EBV transforms these cells, the team was able to examine lymphoblastic cell lines that contained either virus.
They found that LMP2A counteracts the recognition of EBV-infected B cells by EBV-specific, CD8+ killer T cells. In contrast, EBV-transformed cells without LMP2A are more efficiently identified, and the T cells’ ability to recognize and kill the EBV-infected B cells is enhanced.
The researchers examined the mechanism underlying the LMP2A-mediated evasion and found several ways in which it interferes with the recognition of EBV-infected cells.
First, LMP2A reduced the levels of several EBV proteins whose fragments are recognized by CD8+ T cells on the surface of the cell targeted for killing.
Second, LMP2A disturbs the expression of cellular molecules on infected B cells that interact with NKG2D, a host molecule on the surface of CD8+ T cells that aids their activation, thereby weakening the immune response against EBV-infected cells.
The researchers said these results suggest a functional immunomodulatory effect for the EBV protein LMP2A and show that LMP2A mediates the partial escape of infected B cells from recognition by CD8+ T cells.
The team also said similar immune evasion mechanisms may operate in different types of LMP2A-expressing cancers caused by EBV. ![]()
VIDEO: Pacritinib safe with thrombocytopenia in myelofibrosis
CHICAGO – The investigational oral Janus kinase 2 (JAK2) inhibitor pacritinib may offer a new myelofibrosis treatment option for patients with baseline thrombocytopenia.
Analyses from an ongoing phase III trial of pacritinib, reported at the annual meeting of the American Society of Clinical Oncology, indicate that the JAK2 inhibitor is superior to best available therapy for alleviating splenomegaly and other symptoms of myelofibrosis.
“In the very short term, it very clearly identifies a therapy that’s very impactful for those individuals with significant thrombocytopenia and potentially those with anemia,” noted lead study author Dr. Ruben A. Mesa, deputy director of the Mayo Clinic Cancer Center in Scottsdale, Ariz.
In a video interview, Dr. Mesa discussed the study findings so far and the potential implications for patients with myelofibrosis.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – The investigational oral Janus kinase 2 (JAK2) inhibitor pacritinib may offer a new myelofibrosis treatment option for patients with baseline thrombocytopenia.
Analyses from an ongoing phase III trial of pacritinib, reported at the annual meeting of the American Society of Clinical Oncology, indicate that the JAK2 inhibitor is superior to best available therapy for alleviating splenomegaly and other symptoms of myelofibrosis.
“In the very short term, it very clearly identifies a therapy that’s very impactful for those individuals with significant thrombocytopenia and potentially those with anemia,” noted lead study author Dr. Ruben A. Mesa, deputy director of the Mayo Clinic Cancer Center in Scottsdale, Ariz.
In a video interview, Dr. Mesa discussed the study findings so far and the potential implications for patients with myelofibrosis.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – The investigational oral Janus kinase 2 (JAK2) inhibitor pacritinib may offer a new myelofibrosis treatment option for patients with baseline thrombocytopenia.
Analyses from an ongoing phase III trial of pacritinib, reported at the annual meeting of the American Society of Clinical Oncology, indicate that the JAK2 inhibitor is superior to best available therapy for alleviating splenomegaly and other symptoms of myelofibrosis.
“In the very short term, it very clearly identifies a therapy that’s very impactful for those individuals with significant thrombocytopenia and potentially those with anemia,” noted lead study author Dr. Ruben A. Mesa, deputy director of the Mayo Clinic Cancer Center in Scottsdale, Ariz.
In a video interview, Dr. Mesa discussed the study findings so far and the potential implications for patients with myelofibrosis.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE 2015 ASCO ANNUAL MEETING
CAR T-cell therapy seems feasible for NHL, MM

©ASCO/Rodney White
CHICAGO—The CD19-directed chimeric antigen receptor (CAR) T-cell therapy CTL019 has shown promise for treating non-Hodgkin lymphoma (NHL) and may be a feasible treatment option for multiple myeloma (MM) as well, according to researchers.
In an ongoing phase 2 trial, CTL019 has produced durable responses in patients with relapsed or refractory NHL.
And early results of a phase 1 trial suggest CTL019 can provide clinical benefit in heavily pretreated patients with MM.
Both studies were presented at the 2015 ASCO Annual Meeting. The University of Pennsylvania and Novartis have an exclusive global collaboration to research, develop, and commercialize CTL019.
CTL019 in NHL
Stephen Schuster, MD, of the Abramson Cancer Center of the University of Pennsylvania in Philadelphia, presented results of the phase 2 NHL trial (abstract 8516*).
The trial included 20 evaluable patients, 13 with diffuse large B-cell lymphoma (DLBCL) and 7 with follicular lymphoma (FL). At the time of presentation, the median follow-up was 274 days for the patients with DLBCL and 290 days for those with FL.
The overall response rate was 100% in patients with FL and 50% in those with DLBCL. Thirteen patients responded to the therapy, including 11 who achieved a complete response and 2 who experienced a partial response.
Six patients with a partial response to treatment at 3 months achieved a complete response by 6 months. Two patients with a partial response experienced disease progression at 6 and 12 months after treatment.
The researchers said toxicity appeared to be acceptable, with primarily grade 2 cytokine release syndrome (CRS). Two patients developed CRS of grade 3 or higher at peak T-cell expansion. There were no deaths from CRS.
“The results from this ongoing study of CTL019 are encouraging, as we now have data through 6 months showing that patients may have achieved durable overall response rates,” Dr Schuster said. “These data support our ongoing efforts to determine the potential role of CTL019 in improving outcomes for patients with certain types of B-cell lymphomas.”
CTL019 in MM
Alfred Garfall, MD, of the Abramson Cancer Center, presented preliminary results of an ongoing phase 1 study investigating CTL019 in patients with MM (abstract 8517*).
Dr Garfall and his colleagues hypothesized that CTL019 would exhibit efficacy in MM due to low-level CD19 expression on MM plasma cells or CD19 expression in drug-resistant, disease-propagating subsets of the MM clone.
The study included 5 patients who experienced disease progression within a year of a prior autologous stem cell transplant and were medically fit to undergo a second autologous transplant. The patients had received a median of 7.5 prior lines of therapy.
“We found potential evidence of clinical benefit in 3 of 4 patients with more than 100 days of follow-up,” Dr Garfall said.
Two patients experienced longer, deeper responses, and 1 patient experienced CRS.
The data suggest “it is safe and feasible to manufacture and administered CTL019 to refractory multiple myeloma patients,” Dr Garfall said.
*Information in the abstract differs from that presented at the meeting.

©ASCO/Rodney White
CHICAGO—The CD19-directed chimeric antigen receptor (CAR) T-cell therapy CTL019 has shown promise for treating non-Hodgkin lymphoma (NHL) and may be a feasible treatment option for multiple myeloma (MM) as well, according to researchers.
In an ongoing phase 2 trial, CTL019 has produced durable responses in patients with relapsed or refractory NHL.
And early results of a phase 1 trial suggest CTL019 can provide clinical benefit in heavily pretreated patients with MM.
Both studies were presented at the 2015 ASCO Annual Meeting. The University of Pennsylvania and Novartis have an exclusive global collaboration to research, develop, and commercialize CTL019.
CTL019 in NHL
Stephen Schuster, MD, of the Abramson Cancer Center of the University of Pennsylvania in Philadelphia, presented results of the phase 2 NHL trial (abstract 8516*).
The trial included 20 evaluable patients, 13 with diffuse large B-cell lymphoma (DLBCL) and 7 with follicular lymphoma (FL). At the time of presentation, the median follow-up was 274 days for the patients with DLBCL and 290 days for those with FL.
The overall response rate was 100% in patients with FL and 50% in those with DLBCL. Thirteen patients responded to the therapy, including 11 who achieved a complete response and 2 who experienced a partial response.
Six patients with a partial response to treatment at 3 months achieved a complete response by 6 months. Two patients with a partial response experienced disease progression at 6 and 12 months after treatment.
The researchers said toxicity appeared to be acceptable, with primarily grade 2 cytokine release syndrome (CRS). Two patients developed CRS of grade 3 or higher at peak T-cell expansion. There were no deaths from CRS.
“The results from this ongoing study of CTL019 are encouraging, as we now have data through 6 months showing that patients may have achieved durable overall response rates,” Dr Schuster said. “These data support our ongoing efforts to determine the potential role of CTL019 in improving outcomes for patients with certain types of B-cell lymphomas.”
CTL019 in MM
Alfred Garfall, MD, of the Abramson Cancer Center, presented preliminary results of an ongoing phase 1 study investigating CTL019 in patients with MM (abstract 8517*).
Dr Garfall and his colleagues hypothesized that CTL019 would exhibit efficacy in MM due to low-level CD19 expression on MM plasma cells or CD19 expression in drug-resistant, disease-propagating subsets of the MM clone.
The study included 5 patients who experienced disease progression within a year of a prior autologous stem cell transplant and were medically fit to undergo a second autologous transplant. The patients had received a median of 7.5 prior lines of therapy.
“We found potential evidence of clinical benefit in 3 of 4 patients with more than 100 days of follow-up,” Dr Garfall said.
Two patients experienced longer, deeper responses, and 1 patient experienced CRS.
The data suggest “it is safe and feasible to manufacture and administered CTL019 to refractory multiple myeloma patients,” Dr Garfall said.
*Information in the abstract differs from that presented at the meeting.

©ASCO/Rodney White
CHICAGO—The CD19-directed chimeric antigen receptor (CAR) T-cell therapy CTL019 has shown promise for treating non-Hodgkin lymphoma (NHL) and may be a feasible treatment option for multiple myeloma (MM) as well, according to researchers.
In an ongoing phase 2 trial, CTL019 has produced durable responses in patients with relapsed or refractory NHL.
And early results of a phase 1 trial suggest CTL019 can provide clinical benefit in heavily pretreated patients with MM.
Both studies were presented at the 2015 ASCO Annual Meeting. The University of Pennsylvania and Novartis have an exclusive global collaboration to research, develop, and commercialize CTL019.
CTL019 in NHL
Stephen Schuster, MD, of the Abramson Cancer Center of the University of Pennsylvania in Philadelphia, presented results of the phase 2 NHL trial (abstract 8516*).
The trial included 20 evaluable patients, 13 with diffuse large B-cell lymphoma (DLBCL) and 7 with follicular lymphoma (FL). At the time of presentation, the median follow-up was 274 days for the patients with DLBCL and 290 days for those with FL.
The overall response rate was 100% in patients with FL and 50% in those with DLBCL. Thirteen patients responded to the therapy, including 11 who achieved a complete response and 2 who experienced a partial response.
Six patients with a partial response to treatment at 3 months achieved a complete response by 6 months. Two patients with a partial response experienced disease progression at 6 and 12 months after treatment.
The researchers said toxicity appeared to be acceptable, with primarily grade 2 cytokine release syndrome (CRS). Two patients developed CRS of grade 3 or higher at peak T-cell expansion. There were no deaths from CRS.
“The results from this ongoing study of CTL019 are encouraging, as we now have data through 6 months showing that patients may have achieved durable overall response rates,” Dr Schuster said. “These data support our ongoing efforts to determine the potential role of CTL019 in improving outcomes for patients with certain types of B-cell lymphomas.”
CTL019 in MM
Alfred Garfall, MD, of the Abramson Cancer Center, presented preliminary results of an ongoing phase 1 study investigating CTL019 in patients with MM (abstract 8517*).
Dr Garfall and his colleagues hypothesized that CTL019 would exhibit efficacy in MM due to low-level CD19 expression on MM plasma cells or CD19 expression in drug-resistant, disease-propagating subsets of the MM clone.
The study included 5 patients who experienced disease progression within a year of a prior autologous stem cell transplant and were medically fit to undergo a second autologous transplant. The patients had received a median of 7.5 prior lines of therapy.
“We found potential evidence of clinical benefit in 3 of 4 patients with more than 100 days of follow-up,” Dr Garfall said.
Two patients experienced longer, deeper responses, and 1 patient experienced CRS.
The data suggest “it is safe and feasible to manufacture and administered CTL019 to refractory multiple myeloma patients,” Dr Garfall said.
*Information in the abstract differs from that presented at the meeting.
Harnessing immune defense to treat Candida infection

An upset in the body’s natural balance of gut bacteria that may lead to life-threatening bloodstream infections can be reversed by enhancing an
immune response, according to research published in Nature Medicine.
Researchers found that a transcription factor known as HIF-1α works with LL-37, a naturally occurring antibiotic, to kill the infection-causing fungi Candida albicans.
And this response can be enhanced with a drug called L-mimosine.
The researchers noted that Candida albicans can be lethal if it invades the bloodstream from the gut. And stem cell transplant recipients and immunosuppressed cancer patients have a high risk for this type of infection.
“For a cancer patient with a Candida bloodstream infection, the fatality rate is about 30%, [and] Candida is the number 1 fungal pathogen,” said study author Andrew Koh, MD, of the University of Texas Southwestern Medical Center in Dallas.
With that in mind, he and his colleagues set out to determine how the body’s natural immune defense system might be enhanced to fight a Candida infection. By studying how mice infected with Candida responded in different scenarios, the team found their answer.
“The commensal bacteria stimulate gut tissue to make a transcription factor and a natural antibiotic, which then kills the Candida fungus,” Dr Koh explained.
“When we gave the mice a pharmacologic agent called L-mimosine that stimulates the transcription factor, the agent knocked down Candida 100-fold, which translated into a 50% reduction in mortality from invasive Candida infection.”
Specifically, the researchers found that enhancing the transcription factor HIF-1α with L-mimosine led to increased production of the natural antibiotic peptide LL-37, which, in turn, killed the fungi. L-mimosine is a natural product derived from seeds of the koa haole tree that is known to boost HIF-1α activity.
The study also suggested that certain gut bacteria—Clostridial Firmicutes and Bacteroidetes—may be important in producing short-chain fatty acids that help fight infection.
The researchers said more work is needed to pinpoint the optimal method of inducing the body’s gut defense system, whether through use of an agent like L-mimosine or by administering short-chain fatty acids such as vinegar.
“Can we modulate the gut system to maintain balance so that it never gets to the point of pathogens invading the bloodstream?” Dr Koh asked. “Boosting [gastrointestinal] mucosal immune effectors to reduce fungal burden may be the key to tipping the balance back toward normal and preventing invasive fungal disease.” ![]()

An upset in the body’s natural balance of gut bacteria that may lead to life-threatening bloodstream infections can be reversed by enhancing an
immune response, according to research published in Nature Medicine.
Researchers found that a transcription factor known as HIF-1α works with LL-37, a naturally occurring antibiotic, to kill the infection-causing fungi Candida albicans.
And this response can be enhanced with a drug called L-mimosine.
The researchers noted that Candida albicans can be lethal if it invades the bloodstream from the gut. And stem cell transplant recipients and immunosuppressed cancer patients have a high risk for this type of infection.
“For a cancer patient with a Candida bloodstream infection, the fatality rate is about 30%, [and] Candida is the number 1 fungal pathogen,” said study author Andrew Koh, MD, of the University of Texas Southwestern Medical Center in Dallas.
With that in mind, he and his colleagues set out to determine how the body’s natural immune defense system might be enhanced to fight a Candida infection. By studying how mice infected with Candida responded in different scenarios, the team found their answer.
“The commensal bacteria stimulate gut tissue to make a transcription factor and a natural antibiotic, which then kills the Candida fungus,” Dr Koh explained.
“When we gave the mice a pharmacologic agent called L-mimosine that stimulates the transcription factor, the agent knocked down Candida 100-fold, which translated into a 50% reduction in mortality from invasive Candida infection.”
Specifically, the researchers found that enhancing the transcription factor HIF-1α with L-mimosine led to increased production of the natural antibiotic peptide LL-37, which, in turn, killed the fungi. L-mimosine is a natural product derived from seeds of the koa haole tree that is known to boost HIF-1α activity.
The study also suggested that certain gut bacteria—Clostridial Firmicutes and Bacteroidetes—may be important in producing short-chain fatty acids that help fight infection.
The researchers said more work is needed to pinpoint the optimal method of inducing the body’s gut defense system, whether through use of an agent like L-mimosine or by administering short-chain fatty acids such as vinegar.
“Can we modulate the gut system to maintain balance so that it never gets to the point of pathogens invading the bloodstream?” Dr Koh asked. “Boosting [gastrointestinal] mucosal immune effectors to reduce fungal burden may be the key to tipping the balance back toward normal and preventing invasive fungal disease.” ![]()

An upset in the body’s natural balance of gut bacteria that may lead to life-threatening bloodstream infections can be reversed by enhancing an
immune response, according to research published in Nature Medicine.
Researchers found that a transcription factor known as HIF-1α works with LL-37, a naturally occurring antibiotic, to kill the infection-causing fungi Candida albicans.
And this response can be enhanced with a drug called L-mimosine.
The researchers noted that Candida albicans can be lethal if it invades the bloodstream from the gut. And stem cell transplant recipients and immunosuppressed cancer patients have a high risk for this type of infection.
“For a cancer patient with a Candida bloodstream infection, the fatality rate is about 30%, [and] Candida is the number 1 fungal pathogen,” said study author Andrew Koh, MD, of the University of Texas Southwestern Medical Center in Dallas.
With that in mind, he and his colleagues set out to determine how the body’s natural immune defense system might be enhanced to fight a Candida infection. By studying how mice infected with Candida responded in different scenarios, the team found their answer.
“The commensal bacteria stimulate gut tissue to make a transcription factor and a natural antibiotic, which then kills the Candida fungus,” Dr Koh explained.
“When we gave the mice a pharmacologic agent called L-mimosine that stimulates the transcription factor, the agent knocked down Candida 100-fold, which translated into a 50% reduction in mortality from invasive Candida infection.”
Specifically, the researchers found that enhancing the transcription factor HIF-1α with L-mimosine led to increased production of the natural antibiotic peptide LL-37, which, in turn, killed the fungi. L-mimosine is a natural product derived from seeds of the koa haole tree that is known to boost HIF-1α activity.
The study also suggested that certain gut bacteria—Clostridial Firmicutes and Bacteroidetes—may be important in producing short-chain fatty acids that help fight infection.
The researchers said more work is needed to pinpoint the optimal method of inducing the body’s gut defense system, whether through use of an agent like L-mimosine or by administering short-chain fatty acids such as vinegar.
“Can we modulate the gut system to maintain balance so that it never gets to the point of pathogens invading the bloodstream?” Dr Koh asked. “Boosting [gastrointestinal] mucosal immune effectors to reduce fungal burden may be the key to tipping the balance back toward normal and preventing invasive fungal disease.” ![]()
Follow-up PET/CT has ‘clinical value’ in NHL
BALTIMORE—Post-treatment PET/CT scans may be necessary for some patients with non-Hodgkin lymphoma (NHL), according to a presentation at the 2015 SNMMI Annual Meeting.
“[T]he role of post-treatment PET/CT has been controversial,” said Mehdi Taghipour, MD, of Johns Hopkins University in Baltimore, Maryland.
“Our study proves that 39% of follow-up PET/CT scans added clinical value, which represents a significant improvement in NHL patient care.”
Dr Taghipour presented the details of this study at the meeting as abstract 599.
He and his colleagues had examined 560 PET/CT scans from 204 patients. Imaging was performed 6 months or more after the patient completed primary therapy.
The researchers assessed the value of follow-up PET/CT by conducting statistical analyses to determine changes in patient management and evaluated the accuracy of these scans compared to either histopathology or an additional 6-month follow-up.
Results showed that the sensitivity of PET/CT for detecting relapsed NHL was 95.1%, and the specificity was 90.5%. Positive and negative predictive values were 84.5% and 97.1%, respectively.
The overall accuracy of follow-up PET/CT with the common imaging agent fluorodeoxyglucose (FDG) was 92.1%.
Follow-up PET/CT led to changes in patient management in 17% of cases, and new treatments were initiated after 15.7% of scans. More than 69% of scans were performed without prior clinical suspicion of recurrence, and 30.7% of scans were ordered because of suspected disease.
More than 22% of follow-up scans showed suspected disease when there was no clinical suspicion for disease recurrence, and the presence of disease was ruled out in 17.4% of scans where the treating physician had suspected recurrence prior to the scan. ![]()

BALTIMORE—Post-treatment PET/CT scans may be necessary for some patients with non-Hodgkin lymphoma (NHL), according to a presentation at the 2015 SNMMI Annual Meeting.
“[T]he role of post-treatment PET/CT has been controversial,” said Mehdi Taghipour, MD, of Johns Hopkins University in Baltimore, Maryland.
“Our study proves that 39% of follow-up PET/CT scans added clinical value, which represents a significant improvement in NHL patient care.”
Dr Taghipour presented the details of this study at the meeting as abstract 599.
He and his colleagues had examined 560 PET/CT scans from 204 patients. Imaging was performed 6 months or more after the patient completed primary therapy.
The researchers assessed the value of follow-up PET/CT by conducting statistical analyses to determine changes in patient management and evaluated the accuracy of these scans compared to either histopathology or an additional 6-month follow-up.
Results showed that the sensitivity of PET/CT for detecting relapsed NHL was 95.1%, and the specificity was 90.5%. Positive and negative predictive values were 84.5% and 97.1%, respectively.
The overall accuracy of follow-up PET/CT with the common imaging agent fluorodeoxyglucose (FDG) was 92.1%.
Follow-up PET/CT led to changes in patient management in 17% of cases, and new treatments were initiated after 15.7% of scans. More than 69% of scans were performed without prior clinical suspicion of recurrence, and 30.7% of scans were ordered because of suspected disease.
More than 22% of follow-up scans showed suspected disease when there was no clinical suspicion for disease recurrence, and the presence of disease was ruled out in 17.4% of scans where the treating physician had suspected recurrence prior to the scan. ![]()

BALTIMORE—Post-treatment PET/CT scans may be necessary for some patients with non-Hodgkin lymphoma (NHL), according to a presentation at the 2015 SNMMI Annual Meeting.
“[T]he role of post-treatment PET/CT has been controversial,” said Mehdi Taghipour, MD, of Johns Hopkins University in Baltimore, Maryland.
“Our study proves that 39% of follow-up PET/CT scans added clinical value, which represents a significant improvement in NHL patient care.”
Dr Taghipour presented the details of this study at the meeting as abstract 599.
He and his colleagues had examined 560 PET/CT scans from 204 patients. Imaging was performed 6 months or more after the patient completed primary therapy.
The researchers assessed the value of follow-up PET/CT by conducting statistical analyses to determine changes in patient management and evaluated the accuracy of these scans compared to either histopathology or an additional 6-month follow-up.
Results showed that the sensitivity of PET/CT for detecting relapsed NHL was 95.1%, and the specificity was 90.5%. Positive and negative predictive values were 84.5% and 97.1%, respectively.
The overall accuracy of follow-up PET/CT with the common imaging agent fluorodeoxyglucose (FDG) was 92.1%.
Follow-up PET/CT led to changes in patient management in 17% of cases, and new treatments were initiated after 15.7% of scans. More than 69% of scans were performed without prior clinical suspicion of recurrence, and 30.7% of scans were ordered because of suspected disease.
More than 22% of follow-up scans showed suspected disease when there was no clinical suspicion for disease recurrence, and the presence of disease was ruled out in 17.4% of scans where the treating physician had suspected recurrence prior to the scan. ![]()




