User login
Gadolinium and nephrogenic systemic fibrosis: The evidence of things not seen
Now faith is the substance of things hoped for, the evidence of things not seen.
—HEBREWS 11:1
Since the first case appeared in 1997,1 nephrogenic systemic fibrosis (NSF) has been detected with increasing frequency in patients with chronic kidney disease. Recognition that this condition affects more than just the skin led to the change in its name from “nephrogenic fibrosing dermopathy” to “nephrogenic systemic fibrosis.”
In this issue, Issa and colleagues2 review this devastating new disease and discuss its association with gadolinium exposure.
NSF RESEMBLES OTHER FIBROSING DISORDERS
The clinical presentation of NSF most closely resembles that of scleromyxedema or scleroderma.1 However, the face is spared in patients with NSF except for yellow plaques on the sclerae, a frequent finding. Monoclonal gammopathy (which may be associated with scleromyxedema) and Raynaud’s phenomenon (which often is associated with scleroderma) usually are absent in NSF.3
A set of histologic findings differentiates NSF from other fibrosing disorders. Skin biopsy reveals fibrosis and elastosis, often with mucin deposition. If NSF is suspected, immunohistochemical stains for CD34, CD45RO, and type I procollagen should be performed to look for dermal spindle cells (presumably “circulating fibrocytes”) coexpressing these markers. Histiocytic cells and dermal dendrocytes expressing CD68 and factor XIIIa have also been described in NSF skin lesions, but other inflammatory cells usually are absent.4 However, the histologic changes of NSF are difficult to distinguish from those of scleromyxedema.5
Thus, as with scleroderma, the diagnosis of NSF remains clinical. Skin biopsy, even of an affected area, occasionally may yield non-diagnostic findings. Histologic findings serve to confirm the diagnosis of NSF in the appropriate clinical setting.
RISK FACTORS FOR NSF: POSSIBLE ASCERTAINMENT BIAS
Renal dysfunction
Because cases of NSF have been searched for only in patients with chronic kidney disease, reported cases have been found only in this patient population. A major limitation of most published case series is that cases have been gathered from among those with histologic confirmation of NSF, and “controls” have been gathered from the remainder of the population receiving dialysis treatment without confirmation by physical examination of the absence of cutaneous changes of NSF.
Most cases have been found in those with stage 5 chronic kidney disease (creatinine clearance < 15 mL/min or requiring dialysis). However, cases have been described in patients with stage 4 chronic kidney disease (creatinine clearance 15–29 mL/min) and, occasionally, in those with lesser degrees of impaired renal function.
Despite the ascertainment bias in identifying cases, this greater prevalence of NSF with lesser renal function suggests a role for renal dysfunction in the pathogenesis of NSF.
Gadolinium exposure
To date, nearly all patients who have developed NSF have had known exposure to gadolinium-containing contrast agents. Gadolinium has been found in tissue of patients with NSF,6,7 yielding the postulate that gadolinium drives tissue fibrosis.
More patients with chronic kidney disease who developed NSF had been exposed to gadodiamide (Omniscan) than to other gadolinium-containing contrast agents, leading to the hypothesis that less-stable gadolinium-chelate complexes release greater amounts of free gadolinium, which then deposits in tissue and triggers fibrosis. However, it has not yet been determined that the gadolinium deposited in tissue is in the free form and not bound to chelate. Furthermore, this attractive hypothesis must be tempered by the recognition that NSF also has developed after exposure to gadopentetate dimeglumine (Magnevist), a more stable gadolinium-chelate complex than gadodiamide.8 The greater number of patients who have developed NSF after gadodiamide exposure may reflect the relative use of these contrast agents in radiology practice.
It is important to be aware that gadolinium-containing contrast agents are used in more than just magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA). Because gadolinium also blocks transmission of x-rays, radiologists occasionally have used gadolinium-containing contrast agents for angiography, venography, fistulography, and computed tomography in patients for whom use of iodinated contrast agents is contraindicated. Thus, a patient with chronic kidney disease may have received a gadolinium-containing contrast agent even if no magnetic resonance study had been performed.
Assessment of tissue gadolinium content may confirm prior exposure to a gadolinium-containing contrast agent if the patient does not recall having undergone an imaging study. In the one report that claims the development of NSF in two patients without prior gadolinium exposure, tissue was not assessed for gadolinium content.9
No study has yet been performed to assess the relative prevalence of NSF among patients with different stages of chronic kidney disease who have been exposed to gadolinium-containing contrast agents. Thus, it is impossible to ascertain a threshold of renal dysfunction above which the use of gadolinium-containing contrast agents might be safe.
In 90 patients with stage 5 chronic kidney disease, we found that 30% of those who previously had undergone gadolinium-enhanced imaging studies developed cutaneous changes of NSF; the relative risk of developing these skin changes after exposure to a gadolinium-containing contrast agent was 10.7 (95% confidence interval 1.5–6.9).8
Thus, it is essential that guidelines for the use of these contrast agents be formulated and implemented. Caution must be observed when administering a gadolinium-containing contrast agent to a patient with any degree of renal dysfunction. These patients must be informed of the possible risk of developing NSF, and appropriate follow-up must be conducted to assess for potential changes of NSF.
Other possible risk factors
Not all patients with chronic kidney disease who are exposed to gadolinium-containing contrast agents develop NSF: factors other than the degree of renal dysfunction must be involved in the pathogenesis of this condition.
Exposure to medications commonly taken by patients with chronic kidney disease, such as erythropoietin10 and iron supplements,11 has been suggested as a contributing factor. However, these medications are so widely used that this exposure is unlikely to explain why some patients develop NSF after receiving gadolinium-containing contrast agents and others do not.
Interestingly, lanthanum carbonate (Fosrenol) was approved by the US Food and Drug Administration in 2004 for use as a phosphate binder in patients with stage 5 chronic kidney disease. Since lanthanum and gadolinium both are rare earth metals of the lanthanide series, one might speculate that lanthanum deposition in tissue could produce similar changes or could potentiate those induced by gadolinium.
Future prospective case-control studies need to address risk factors for the development of NSF.
EFFECTIVE TREATMENT NEEDED
Because NSF imposes a markedly increased rate of death and devastating morbidity,8 efforts must be directed toward preventing its development and treating those who already are affected. So far, no treatment has been universally effective in reversing the fibrotic changes of NSF. Potentially effective therapeutic agents must be identified and studied in these patients.
Although performing hemodialysis promptly after the use of a gadolinium-containing contrast agent would appear to be a prudent clinical practice, there are no data to suggest that it is effective in preventing NSF. If free gadolinium disassociates from its chelate and deposits rapidly in tissue, it is unclear that hemodialysis could be performed soon enough to prevent this deposition. Furthermore, hemodialysis is not without associated potential risks and morbidity, especially in people with chronic kidney disease who are not already receiving hemodialysis. Thus, at present, avoiding the use of gadolinium-containing contrast agents in patients with chronic kidney disease appears to be the best preventive strategy.
A NAME CHANGE
Over the past decade, much has been learned about the clinical manifestations, course, and pathogenesis of NSF. However, the term “nephrogenic” in the name of this disease is misleading, in that this fibrosing disorder is not caused by the kidneys. Although some degree of renal dysfunction appears to be necessary for NSF to develop, the presence of gadolinium in tissue seems to drive fibrosis. Thus, it is time that “nephrogenic systemic fibrosis” be renamed more precisely as “gadolinium-associated systemic fibrosis” or “GASF.”
- Cowper SE, Robin HS, Steinberg SM, Su LD, Gupta S, LeBoit PE. Scleromyxoedema-like cutaneous diseases in renal-dialysis patients. Lancet 2000; 356:1000–1001.
- Issa N, Poggio E, Fatica R, Patel R, Ruggieri PM, Heyka RJ. Nephrogenic systemic fibrosis and its association with gadolinium exposure during MRI. Cleve Clin J Med 2008; 75:95–111.
- Moschella SL, Kay J, Mackool BT, Liu V. Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Case 35-2004. A 68-year-old man with end-stage renal disease and thickening of the skin. N Engl J Med 2004; 351:2219–2227.
- Cowper SE, Su LD, Bhawan J, Robin HS, LeBoit PE. Nephrogenic fibrosing dermopathy. Am J Dermatopathol 2001; 23:383–393.
- Kucher C, Xu X, Pasha T, Elenitsas R. Histopathologic comparison of nephrogenic fibrosing dermopathy and scleromyxedema. J Cutan Pathol 2005; 32:484–490.
- High WA, Ayers RA, Chandler J, Zito G, Cowper SE. Gadolinium is detectable within the tissue of patients with nephrogenic systemic fibrosis. J Am Acad Dermatol 2007; 56:21–26.
- Boyd AS, Zic JA, Abraham JL. Gadolinium deposition in nephrogenic fibrosing dermopathy. J Am Acad Dermatol 2007; 56:27–30.
- Todd DJ, Kagan A, Chibnik LB, Kay J. Cutaneous changes of nephrogenic systemic fibrosis: predictor of early mortality and association with gadolinium exposure. Arthritis Rheum 2007; 56:3433–3441.
- Wahba IM, Simpson EL, White K. Gadolinium is not the only trigger for nephrogenic systemic fibrosis: insights from two cases and review of the recent literature. Am J Transplant 2007; 7:2425–2432.
- Swaminathan S, Ahmed I, McCarthy JT, et al. Nephrogenic fibrosing dermopathy and high–dose erythropoietin therapy. Ann Intern Med 2006; 145:234–235.
- Swaminathan S, Horn TD, Pellowski D, et al. Nephrogenic systemic fibrosis, gadolinium, and iron mobilization. N Engl J Med 2007; 357:720–722.
Now faith is the substance of things hoped for, the evidence of things not seen.
—HEBREWS 11:1
Since the first case appeared in 1997,1 nephrogenic systemic fibrosis (NSF) has been detected with increasing frequency in patients with chronic kidney disease. Recognition that this condition affects more than just the skin led to the change in its name from “nephrogenic fibrosing dermopathy” to “nephrogenic systemic fibrosis.”
In this issue, Issa and colleagues2 review this devastating new disease and discuss its association with gadolinium exposure.
NSF RESEMBLES OTHER FIBROSING DISORDERS
The clinical presentation of NSF most closely resembles that of scleromyxedema or scleroderma.1 However, the face is spared in patients with NSF except for yellow plaques on the sclerae, a frequent finding. Monoclonal gammopathy (which may be associated with scleromyxedema) and Raynaud’s phenomenon (which often is associated with scleroderma) usually are absent in NSF.3
A set of histologic findings differentiates NSF from other fibrosing disorders. Skin biopsy reveals fibrosis and elastosis, often with mucin deposition. If NSF is suspected, immunohistochemical stains for CD34, CD45RO, and type I procollagen should be performed to look for dermal spindle cells (presumably “circulating fibrocytes”) coexpressing these markers. Histiocytic cells and dermal dendrocytes expressing CD68 and factor XIIIa have also been described in NSF skin lesions, but other inflammatory cells usually are absent.4 However, the histologic changes of NSF are difficult to distinguish from those of scleromyxedema.5
Thus, as with scleroderma, the diagnosis of NSF remains clinical. Skin biopsy, even of an affected area, occasionally may yield non-diagnostic findings. Histologic findings serve to confirm the diagnosis of NSF in the appropriate clinical setting.
RISK FACTORS FOR NSF: POSSIBLE ASCERTAINMENT BIAS
Renal dysfunction
Because cases of NSF have been searched for only in patients with chronic kidney disease, reported cases have been found only in this patient population. A major limitation of most published case series is that cases have been gathered from among those with histologic confirmation of NSF, and “controls” have been gathered from the remainder of the population receiving dialysis treatment without confirmation by physical examination of the absence of cutaneous changes of NSF.
Most cases have been found in those with stage 5 chronic kidney disease (creatinine clearance < 15 mL/min or requiring dialysis). However, cases have been described in patients with stage 4 chronic kidney disease (creatinine clearance 15–29 mL/min) and, occasionally, in those with lesser degrees of impaired renal function.
Despite the ascertainment bias in identifying cases, this greater prevalence of NSF with lesser renal function suggests a role for renal dysfunction in the pathogenesis of NSF.
Gadolinium exposure
To date, nearly all patients who have developed NSF have had known exposure to gadolinium-containing contrast agents. Gadolinium has been found in tissue of patients with NSF,6,7 yielding the postulate that gadolinium drives tissue fibrosis.
More patients with chronic kidney disease who developed NSF had been exposed to gadodiamide (Omniscan) than to other gadolinium-containing contrast agents, leading to the hypothesis that less-stable gadolinium-chelate complexes release greater amounts of free gadolinium, which then deposits in tissue and triggers fibrosis. However, it has not yet been determined that the gadolinium deposited in tissue is in the free form and not bound to chelate. Furthermore, this attractive hypothesis must be tempered by the recognition that NSF also has developed after exposure to gadopentetate dimeglumine (Magnevist), a more stable gadolinium-chelate complex than gadodiamide.8 The greater number of patients who have developed NSF after gadodiamide exposure may reflect the relative use of these contrast agents in radiology practice.
It is important to be aware that gadolinium-containing contrast agents are used in more than just magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA). Because gadolinium also blocks transmission of x-rays, radiologists occasionally have used gadolinium-containing contrast agents for angiography, venography, fistulography, and computed tomography in patients for whom use of iodinated contrast agents is contraindicated. Thus, a patient with chronic kidney disease may have received a gadolinium-containing contrast agent even if no magnetic resonance study had been performed.
Assessment of tissue gadolinium content may confirm prior exposure to a gadolinium-containing contrast agent if the patient does not recall having undergone an imaging study. In the one report that claims the development of NSF in two patients without prior gadolinium exposure, tissue was not assessed for gadolinium content.9
No study has yet been performed to assess the relative prevalence of NSF among patients with different stages of chronic kidney disease who have been exposed to gadolinium-containing contrast agents. Thus, it is impossible to ascertain a threshold of renal dysfunction above which the use of gadolinium-containing contrast agents might be safe.
In 90 patients with stage 5 chronic kidney disease, we found that 30% of those who previously had undergone gadolinium-enhanced imaging studies developed cutaneous changes of NSF; the relative risk of developing these skin changes after exposure to a gadolinium-containing contrast agent was 10.7 (95% confidence interval 1.5–6.9).8
Thus, it is essential that guidelines for the use of these contrast agents be formulated and implemented. Caution must be observed when administering a gadolinium-containing contrast agent to a patient with any degree of renal dysfunction. These patients must be informed of the possible risk of developing NSF, and appropriate follow-up must be conducted to assess for potential changes of NSF.
Other possible risk factors
Not all patients with chronic kidney disease who are exposed to gadolinium-containing contrast agents develop NSF: factors other than the degree of renal dysfunction must be involved in the pathogenesis of this condition.
Exposure to medications commonly taken by patients with chronic kidney disease, such as erythropoietin10 and iron supplements,11 has been suggested as a contributing factor. However, these medications are so widely used that this exposure is unlikely to explain why some patients develop NSF after receiving gadolinium-containing contrast agents and others do not.
Interestingly, lanthanum carbonate (Fosrenol) was approved by the US Food and Drug Administration in 2004 for use as a phosphate binder in patients with stage 5 chronic kidney disease. Since lanthanum and gadolinium both are rare earth metals of the lanthanide series, one might speculate that lanthanum deposition in tissue could produce similar changes or could potentiate those induced by gadolinium.
Future prospective case-control studies need to address risk factors for the development of NSF.
EFFECTIVE TREATMENT NEEDED
Because NSF imposes a markedly increased rate of death and devastating morbidity,8 efforts must be directed toward preventing its development and treating those who already are affected. So far, no treatment has been universally effective in reversing the fibrotic changes of NSF. Potentially effective therapeutic agents must be identified and studied in these patients.
Although performing hemodialysis promptly after the use of a gadolinium-containing contrast agent would appear to be a prudent clinical practice, there are no data to suggest that it is effective in preventing NSF. If free gadolinium disassociates from its chelate and deposits rapidly in tissue, it is unclear that hemodialysis could be performed soon enough to prevent this deposition. Furthermore, hemodialysis is not without associated potential risks and morbidity, especially in people with chronic kidney disease who are not already receiving hemodialysis. Thus, at present, avoiding the use of gadolinium-containing contrast agents in patients with chronic kidney disease appears to be the best preventive strategy.
A NAME CHANGE
Over the past decade, much has been learned about the clinical manifestations, course, and pathogenesis of NSF. However, the term “nephrogenic” in the name of this disease is misleading, in that this fibrosing disorder is not caused by the kidneys. Although some degree of renal dysfunction appears to be necessary for NSF to develop, the presence of gadolinium in tissue seems to drive fibrosis. Thus, it is time that “nephrogenic systemic fibrosis” be renamed more precisely as “gadolinium-associated systemic fibrosis” or “GASF.”
Now faith is the substance of things hoped for, the evidence of things not seen.
—HEBREWS 11:1
Since the first case appeared in 1997,1 nephrogenic systemic fibrosis (NSF) has been detected with increasing frequency in patients with chronic kidney disease. Recognition that this condition affects more than just the skin led to the change in its name from “nephrogenic fibrosing dermopathy” to “nephrogenic systemic fibrosis.”
In this issue, Issa and colleagues2 review this devastating new disease and discuss its association with gadolinium exposure.
NSF RESEMBLES OTHER FIBROSING DISORDERS
The clinical presentation of NSF most closely resembles that of scleromyxedema or scleroderma.1 However, the face is spared in patients with NSF except for yellow plaques on the sclerae, a frequent finding. Monoclonal gammopathy (which may be associated with scleromyxedema) and Raynaud’s phenomenon (which often is associated with scleroderma) usually are absent in NSF.3
A set of histologic findings differentiates NSF from other fibrosing disorders. Skin biopsy reveals fibrosis and elastosis, often with mucin deposition. If NSF is suspected, immunohistochemical stains for CD34, CD45RO, and type I procollagen should be performed to look for dermal spindle cells (presumably “circulating fibrocytes”) coexpressing these markers. Histiocytic cells and dermal dendrocytes expressing CD68 and factor XIIIa have also been described in NSF skin lesions, but other inflammatory cells usually are absent.4 However, the histologic changes of NSF are difficult to distinguish from those of scleromyxedema.5
Thus, as with scleroderma, the diagnosis of NSF remains clinical. Skin biopsy, even of an affected area, occasionally may yield non-diagnostic findings. Histologic findings serve to confirm the diagnosis of NSF in the appropriate clinical setting.
RISK FACTORS FOR NSF: POSSIBLE ASCERTAINMENT BIAS
Renal dysfunction
Because cases of NSF have been searched for only in patients with chronic kidney disease, reported cases have been found only in this patient population. A major limitation of most published case series is that cases have been gathered from among those with histologic confirmation of NSF, and “controls” have been gathered from the remainder of the population receiving dialysis treatment without confirmation by physical examination of the absence of cutaneous changes of NSF.
Most cases have been found in those with stage 5 chronic kidney disease (creatinine clearance < 15 mL/min or requiring dialysis). However, cases have been described in patients with stage 4 chronic kidney disease (creatinine clearance 15–29 mL/min) and, occasionally, in those with lesser degrees of impaired renal function.
Despite the ascertainment bias in identifying cases, this greater prevalence of NSF with lesser renal function suggests a role for renal dysfunction in the pathogenesis of NSF.
Gadolinium exposure
To date, nearly all patients who have developed NSF have had known exposure to gadolinium-containing contrast agents. Gadolinium has been found in tissue of patients with NSF,6,7 yielding the postulate that gadolinium drives tissue fibrosis.
More patients with chronic kidney disease who developed NSF had been exposed to gadodiamide (Omniscan) than to other gadolinium-containing contrast agents, leading to the hypothesis that less-stable gadolinium-chelate complexes release greater amounts of free gadolinium, which then deposits in tissue and triggers fibrosis. However, it has not yet been determined that the gadolinium deposited in tissue is in the free form and not bound to chelate. Furthermore, this attractive hypothesis must be tempered by the recognition that NSF also has developed after exposure to gadopentetate dimeglumine (Magnevist), a more stable gadolinium-chelate complex than gadodiamide.8 The greater number of patients who have developed NSF after gadodiamide exposure may reflect the relative use of these contrast agents in radiology practice.
It is important to be aware that gadolinium-containing contrast agents are used in more than just magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA). Because gadolinium also blocks transmission of x-rays, radiologists occasionally have used gadolinium-containing contrast agents for angiography, venography, fistulography, and computed tomography in patients for whom use of iodinated contrast agents is contraindicated. Thus, a patient with chronic kidney disease may have received a gadolinium-containing contrast agent even if no magnetic resonance study had been performed.
Assessment of tissue gadolinium content may confirm prior exposure to a gadolinium-containing contrast agent if the patient does not recall having undergone an imaging study. In the one report that claims the development of NSF in two patients without prior gadolinium exposure, tissue was not assessed for gadolinium content.9
No study has yet been performed to assess the relative prevalence of NSF among patients with different stages of chronic kidney disease who have been exposed to gadolinium-containing contrast agents. Thus, it is impossible to ascertain a threshold of renal dysfunction above which the use of gadolinium-containing contrast agents might be safe.
In 90 patients with stage 5 chronic kidney disease, we found that 30% of those who previously had undergone gadolinium-enhanced imaging studies developed cutaneous changes of NSF; the relative risk of developing these skin changes after exposure to a gadolinium-containing contrast agent was 10.7 (95% confidence interval 1.5–6.9).8
Thus, it is essential that guidelines for the use of these contrast agents be formulated and implemented. Caution must be observed when administering a gadolinium-containing contrast agent to a patient with any degree of renal dysfunction. These patients must be informed of the possible risk of developing NSF, and appropriate follow-up must be conducted to assess for potential changes of NSF.
Other possible risk factors
Not all patients with chronic kidney disease who are exposed to gadolinium-containing contrast agents develop NSF: factors other than the degree of renal dysfunction must be involved in the pathogenesis of this condition.
Exposure to medications commonly taken by patients with chronic kidney disease, such as erythropoietin10 and iron supplements,11 has been suggested as a contributing factor. However, these medications are so widely used that this exposure is unlikely to explain why some patients develop NSF after receiving gadolinium-containing contrast agents and others do not.
Interestingly, lanthanum carbonate (Fosrenol) was approved by the US Food and Drug Administration in 2004 for use as a phosphate binder in patients with stage 5 chronic kidney disease. Since lanthanum and gadolinium both are rare earth metals of the lanthanide series, one might speculate that lanthanum deposition in tissue could produce similar changes or could potentiate those induced by gadolinium.
Future prospective case-control studies need to address risk factors for the development of NSF.
EFFECTIVE TREATMENT NEEDED
Because NSF imposes a markedly increased rate of death and devastating morbidity,8 efforts must be directed toward preventing its development and treating those who already are affected. So far, no treatment has been universally effective in reversing the fibrotic changes of NSF. Potentially effective therapeutic agents must be identified and studied in these patients.
Although performing hemodialysis promptly after the use of a gadolinium-containing contrast agent would appear to be a prudent clinical practice, there are no data to suggest that it is effective in preventing NSF. If free gadolinium disassociates from its chelate and deposits rapidly in tissue, it is unclear that hemodialysis could be performed soon enough to prevent this deposition. Furthermore, hemodialysis is not without associated potential risks and morbidity, especially in people with chronic kidney disease who are not already receiving hemodialysis. Thus, at present, avoiding the use of gadolinium-containing contrast agents in patients with chronic kidney disease appears to be the best preventive strategy.
A NAME CHANGE
Over the past decade, much has been learned about the clinical manifestations, course, and pathogenesis of NSF. However, the term “nephrogenic” in the name of this disease is misleading, in that this fibrosing disorder is not caused by the kidneys. Although some degree of renal dysfunction appears to be necessary for NSF to develop, the presence of gadolinium in tissue seems to drive fibrosis. Thus, it is time that “nephrogenic systemic fibrosis” be renamed more precisely as “gadolinium-associated systemic fibrosis” or “GASF.”
- Cowper SE, Robin HS, Steinberg SM, Su LD, Gupta S, LeBoit PE. Scleromyxoedema-like cutaneous diseases in renal-dialysis patients. Lancet 2000; 356:1000–1001.
- Issa N, Poggio E, Fatica R, Patel R, Ruggieri PM, Heyka RJ. Nephrogenic systemic fibrosis and its association with gadolinium exposure during MRI. Cleve Clin J Med 2008; 75:95–111.
- Moschella SL, Kay J, Mackool BT, Liu V. Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Case 35-2004. A 68-year-old man with end-stage renal disease and thickening of the skin. N Engl J Med 2004; 351:2219–2227.
- Cowper SE, Su LD, Bhawan J, Robin HS, LeBoit PE. Nephrogenic fibrosing dermopathy. Am J Dermatopathol 2001; 23:383–393.
- Kucher C, Xu X, Pasha T, Elenitsas R. Histopathologic comparison of nephrogenic fibrosing dermopathy and scleromyxedema. J Cutan Pathol 2005; 32:484–490.
- High WA, Ayers RA, Chandler J, Zito G, Cowper SE. Gadolinium is detectable within the tissue of patients with nephrogenic systemic fibrosis. J Am Acad Dermatol 2007; 56:21–26.
- Boyd AS, Zic JA, Abraham JL. Gadolinium deposition in nephrogenic fibrosing dermopathy. J Am Acad Dermatol 2007; 56:27–30.
- Todd DJ, Kagan A, Chibnik LB, Kay J. Cutaneous changes of nephrogenic systemic fibrosis: predictor of early mortality and association with gadolinium exposure. Arthritis Rheum 2007; 56:3433–3441.
- Wahba IM, Simpson EL, White K. Gadolinium is not the only trigger for nephrogenic systemic fibrosis: insights from two cases and review of the recent literature. Am J Transplant 2007; 7:2425–2432.
- Swaminathan S, Ahmed I, McCarthy JT, et al. Nephrogenic fibrosing dermopathy and high–dose erythropoietin therapy. Ann Intern Med 2006; 145:234–235.
- Swaminathan S, Horn TD, Pellowski D, et al. Nephrogenic systemic fibrosis, gadolinium, and iron mobilization. N Engl J Med 2007; 357:720–722.
- Cowper SE, Robin HS, Steinberg SM, Su LD, Gupta S, LeBoit PE. Scleromyxoedema-like cutaneous diseases in renal-dialysis patients. Lancet 2000; 356:1000–1001.
- Issa N, Poggio E, Fatica R, Patel R, Ruggieri PM, Heyka RJ. Nephrogenic systemic fibrosis and its association with gadolinium exposure during MRI. Cleve Clin J Med 2008; 75:95–111.
- Moschella SL, Kay J, Mackool BT, Liu V. Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Case 35-2004. A 68-year-old man with end-stage renal disease and thickening of the skin. N Engl J Med 2004; 351:2219–2227.
- Cowper SE, Su LD, Bhawan J, Robin HS, LeBoit PE. Nephrogenic fibrosing dermopathy. Am J Dermatopathol 2001; 23:383–393.
- Kucher C, Xu X, Pasha T, Elenitsas R. Histopathologic comparison of nephrogenic fibrosing dermopathy and scleromyxedema. J Cutan Pathol 2005; 32:484–490.
- High WA, Ayers RA, Chandler J, Zito G, Cowper SE. Gadolinium is detectable within the tissue of patients with nephrogenic systemic fibrosis. J Am Acad Dermatol 2007; 56:21–26.
- Boyd AS, Zic JA, Abraham JL. Gadolinium deposition in nephrogenic fibrosing dermopathy. J Am Acad Dermatol 2007; 56:27–30.
- Todd DJ, Kagan A, Chibnik LB, Kay J. Cutaneous changes of nephrogenic systemic fibrosis: predictor of early mortality and association with gadolinium exposure. Arthritis Rheum 2007; 56:3433–3441.
- Wahba IM, Simpson EL, White K. Gadolinium is not the only trigger for nephrogenic systemic fibrosis: insights from two cases and review of the recent literature. Am J Transplant 2007; 7:2425–2432.
- Swaminathan S, Ahmed I, McCarthy JT, et al. Nephrogenic fibrosing dermopathy and high–dose erythropoietin therapy. Ann Intern Med 2006; 145:234–235.
- Swaminathan S, Horn TD, Pellowski D, et al. Nephrogenic systemic fibrosis, gadolinium, and iron mobilization. N Engl J Med 2007; 357:720–722.
Nephrogenic systemic fibrosis and its association with gadolinium exposure during MRI
The use of gadolinium as a contrast agent in magnetic resonance imaging (MRI) in patients with impaired kidney function has come under scrutiny because of recent reports of a potential association between its use and nephrogenic systemic fibrosis (NSF).
This entity was first identified in the United States in 1997. Cowper et al1 in 2000 described 15 hemodialysis patients who developed thickening and hardening of the skin with brawny hyperpigmentation, papules, and subcutaneous nodules on the extremities.
This “new disease” was initially called “nephrogenic fibrosing dermopathy,” as it was exclusively seen in patients with renal impairment and was thought to affect only the skin and subcutaneous tissue. With growing evidence of the extent and pathogenicity of the fibrosis in visceral organs, the nomenclature was changed to NSF, to better reflect the systemic nature of the disease.
PRESENTATION: MILD TO DEVASTATING
NSF has thus far been reported only in patients with renal impairment, most of whom were dialysis-dependent. It does not seem to be more common in one sex or the other, in any age range, or in any ethnic group. It can range in severity from mild to a devastating scleroderma-like systemic fibrosing disorder.
The heart, lungs, skeletal muscle, and diaphragm can also be involved, sometimes leading to serious complications and death.4–6
The disease is usually progressive and unremitting. Mendoza et al,7 in a review of 12 cases of NSF, reported that the disease had a progressive course in 6 patients, of whom 3 died within 2 years and 3 were ultimately confined to a wheelchair. More severe findings and rapid progression of the skin disease are associated with a poor prognosis.
Todd et al8 prospectively examined 186 dialysis patients to look for possible NSF. Of those with skin changes consistent with NSF, 48% died within 2 years, compared with 20% of those without these skin changes. Cardiovascular causes accounted for 58% of the deaths in patients with cutaneous changes of NSF and for 48% of the deaths in patients without these changes. Most of the excess deaths occurred within 6 months after the skin examination, suggesting an increased risk for early death in patients with skin changes suggestive of NSF.
DIAGNOSIS OF NSF IS CLINICAL
At presentation, NSF is frequently misdiagnosed and treated as cellulitis or edema. However, now that subspecialists—especially dermatologists, rheumatologists, and nephrologists—are becoming more aware of it, the correct diagnosis is being made earlier.
NSF should be suspected in any patient with underlying renal dysfunction—especially if on dialysis and if he or she has received a gadolinium contrast agent during MRI—who develops scleroderma-like cutaneous lesions affecting the distal extremities. Because most health care providers are still unfamiliar with this emerging disease, patients with renal impairment and suspected NSF should be referred to a rheumatologist or dermatologist to confirm the diagnosis, which is mainly entertained on a clinical basis. There is no laboratory biomarker for NSF.
A deep incisional skin biopsy may aid in the diagnosis. Due to the regional distribution of the disease, sampling error may occur, and repeat biopsy is warranted if the initial biopsy is nondiagnostic but the clinical picture suggests NSF.
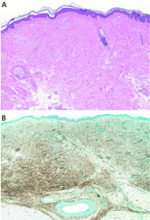
Histopathologic examination typically shows lesions containing proliferation of dermal spindle cells, thick collagen bundles with surrounding clefts, and a variable amount of mucin and elastic fibers.2 A characteristic and almost pathognomonic staining profile is the immunohistochemical identification of CD34 reactivity in the fibroblast-like cells (Figure 3). Cells expressing CD34 are normally found in the umbilical cord, the bone marrow (as pluripotential hematopoietic stem cells), and in the vascular endothelium. How they come to be in the skin is still speculative, but their presence suggests that circulating fibrocytes migrate from the bone marrow and deposit in the skin and other organs.9,10
Pulmonary function testing can be done to rule out lung involvement and transthoracic two-dimensional echocardiography can be done to rule out possible cardiomyopathy if these conditions are suggested by examination at the time of diagnosis.7 Muscle biopsy is not necessary to determine the extent of systemic involvement, since the findings do not necessarily correlate with other systemic involvement.
DIFFERENTIAL DIAGNOSIS
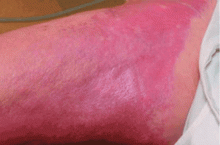
An important diagnostic feature of NSF is that it spares the face, a finding derived from all reported and confirmed cases of NSF (Figure 2). In contrast, scleromyxedema, systemic scleroderma, and morphea often involve the face.
Scleromyxedema is often associated with monoclonal gammopathy (usually an immunoglobulin G lambda paraproteinemia) whereas NSF is not.
Scleroderma is supported by the findings of Raynaud’s phenomenon, antinuclear antibodies, and either anticentromere or anti-DNA topoisomerase I (Scl-70) antibodies, but the absence of these antibodies does not necessarily rule it out.
Eosinophilic fasciitis is diagnosed on the basis of histologic examination of a deep wedge skin biopsy specimen that includes fascia.
Other diagnoses that should be considered include amyloidosis and calciphylaxis.
ASSOCIATION WITH GADOLINIUM: WHAT IS THE EVIDENCE?
Case series
The association of gadolinium use with NSF has been described in several case reports and case series.
Grobner11 reported that administration of gadodiamide (Omniscan, a gadolinium compound) for MRI was associated with NSF in five patients on chronic hemodialysis who had end-stage renal disease. Their ages ranged from 43 to 74 years, and they had been on dialysis from 10 to 58 months. The time of onset of NSF ranged from 2 to 4 weeks after exposure to gadodiamide.
Marckmann et al12 reported that NSF developed in 13 (3.5%) of 370 patients with severe kidney disease who received gadodiamide. Five of the 13 patients had stage 5 (advanced) chronic kidney disease and were not yet on renal replacement therapy, 7 were on hemodialysis, and 1 was on peritoneal dialysis. The time of onset ranged from 2 to 75 days (median 25 days) after exposure.
Kuo et al13 similarly estimated the incidence of NSF at approximately 3% in patients with severe renal failure who receive intravenous gadolinium-based contrast material for MRI.
Broome et al14 reported that 12 patients developed NSF within 2 to 11 weeks after receiving gadodiamide. Eight of the 12 patients had end-stage renal disease and were on hemodialysis; the other 4 patients had acute kidney injury attributed to hepatorenal syndrome, and 3 of these 4 patients were on hemodialysis.
Khurana et al15 reported that 6 patients on hemodialysis developed NSF from 2 weeks to 2 months after receiving a dose of gadodiamide of between 0.11 and 0.36 mmol/kg. These doses are high, and the findings suggest an association between the gadolinium dose and NSF. The dose approved by the US Food and Drug Administration (FDA) is only 0.1 mmol/kg, and the use of gadolinium is approved only in MRI. However, higher doses (0.3–0.4 mmol/kg) are widely used in practice for better imaging quality in magnetic resonance angiography (MRA).
Deo et al16 reported 3 cases of NSF in 87 patients with end-stage renal disease who underwent 123 radiologic studies with gadolinium. No patient with end-stage renal disease who was not exposed to gadolinium developed NSF, and the association between exposure to gadolinium and the subsequent development of NSF was statistically significant (P = .006). The authors concluded that each gadolinium study presented a 2.4% risk of NSF in end-stage renal disease patients.
This retrospective study is flawed by not having been cross-sectional or case-controlled, since the other 84 patients who received gadolinium were not examined at all to establish the absence of NSF.
Case-control studies
More evidence of association of NSF with gadolinium exposure comes from other reports.
Physicians in St. Louis, MO,17 identified 33 cases of NSF and performed a case-control study, matching each of 19 of the patients (for whom data were available and who met their entry criteria) with 3 controls. They found that exposure to gadolinium was independently associated with the development of NSF.
Sadowski et al18 reported that 13 patients with biopsy-confirmed NSF all had been exposed to gadodiamide and one had been exposed to gadobenate (MultiHANCE) in addition to gadodiamide. All 13 patients had renal insufficiency, with an estimated glomerular filtration rate (GFR) less than 60 mL/minute/1.73 m2. The investigators compared this group with a control group of patients with renal insufficiency who did not develop NSF. The NSF group had more proinflammatory events (P < .001) and more gadolinium-contrast-enhanced MRI examinations per patient (P = .002) than the control group.
Marckmann et al19 compared 19 patients who had histologically proven cases of NSF and 19 sex- and age-matched controls; all 38 patients had chronic kidney disease and had been exposed to gadolinium. Patients with NSF had received higher cumulative doses of gadodiamide and higher doses of erythropoietin and had higher serum concentrations of ionized calcium and phosphate than did their controls, as did patients with severe NSF compared with those with nonsevere NSF.
Comment. All the above reports are limited by their study design and suffer from recognition bias because not all of the patients with severe renal insufficiency who were exposed to gadolinium were examined for possible asymptomatic skin changes that might be characteristic of NSF. Therefore, it is impossible to be certain that all of the patients classified as not having NSF truly did not have it or did not subsequently develop it. Furthermore, the reports lacked standardized diagnostic criteria. Hence, the real prevalence and incidence of NSF are difficult to determine.
A cross-sectional study
As mentioned above, Todd et al8 examined 186 dialysis patients for cutaneous changes of NSF (using a scoring system based on hyper-pigmentation, hardening, and tethering of skin on the extremities). Patients who had been exposed to gadolinium had a higher risk of developing these skin changes than did nonexposed patients (odds ratio 14.7, 95% confidence interval 1.9–117.0). More importantly, the investigators found cutaneous changes of NSF in 25 (13%) of the 186 patients, 4 of whom had prior skin biopsies available for review, each revealing the histologic changes of NSF. This study suggests that NSF may be more prevalent than previously thought.
Is kidney dysfunction always present?
All the reported patients with NSF had underlying renal impairment. The renal dysfunction ranged from acute kidney injury to advanced chronic kidney disease (estimated GFR < 30 mL/minute/1.73 m2) and end-stage renal disease on renal replacement therapy, ie, hemodialysis or peritoneal dialysis. The incidence of NSF does not seem to be related to the cause of the underlying kidney disease.
What other diseases or comorbidities can be associated with NSF?
It is still unclear why not every patient with advanced renal failure develops NSF after exposure to gadolinium.
A variety of complex diseases and conditions have been reported to be associated with NSF, with no clear-cut evidence of causality or trigger. These include hypercoagulability states, thrombotic events, surgical procedures (especially those with reconstructive vascular components), calciphylaxis, kidney transplantation, hepatic disease (hepatorenal syndrome, liver transplantation, and hepatitis B and C), idiopathic pulmonary fibrosis, systemic lupus erythematosus, hypothyroidism, elevated serum ionized calcium or serum phosphate, hyperparathyroidism, and metabolic acidosis. A possible explanation is that most of these conditions are associated with an increased use of MRI or MRA testing (eg, in the workup for kidney or liver transplantation).
Many drugs have also been reported to be associated with NSF, including high-dose erythropoietin,20 sevelamer (Renagel),21 and, conversely, lack of angiotensin-converting enzyme inhibitor therapy,22 but none of these findings has been reproduced to date.
GADOLINIUM CHARACTERISTICS AND PHARMACOKINETICS
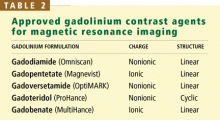
Gadolinium is a rare-earth lanthanide metallic element (atomic number 64) that is used in MRI and MRA because of its paramagnetic properties that enhance the quality of imaging. Its ionic form (Gd3+) is highly toxic if injected intravenously, so it is typically bound to a “chelate” to decrease its toxicity.23 The chelate stabilizes Gd3+ and thereby prevents its dissociation in vivo. These Gd-chelates can be classified (Table 2) according to their charge (ionic vs nonionic) and their structure (linear vs cyclic).
Most of the reported cases of NSF have been in patients who received gadodiamide, a nonionic, linear agent. Why gadodiamide has the highest rates of association with NSF is still unclear; perhaps it is simply the most widely used agent. Also, linear Gd compounds may be less stable and more likely to dissociate in vivo. The updated FDA Public Health Advisory in May 2007 warned against the use of all gadolinium-containing contrast agents for MRI, not just gadodiamide.
After intravenous injection, Gd-chelate equilibrates rapidly (within 2 hours) in the extracellular space. Very little of it enters into cells or binds to proteins. It is eliminated unchanged in the glomerular filtrate with no tubular secretion. In a study by Joffe et al,24 the elimination half-life of gadodiamide in patients with severely reduced renal function was considerably longer than in healthy volunteers (34.3 hours ± 22.9 vs 1.3 hours ± 0.25).
Since gadolinium compounds are not protein-bound and have a limited volume of distribution, they are typically removed by hemodialysis. Joffe et al found that an average of 65% of the gadodiamide was removed in a single hemodialysis session. However, they did not describe the specific features of the hemodialysis session, and it took four hemodialysis treatments to remove 99% of a single dose of gadolinium.24 A dialysis membrane with high permeability (large pores) seems to increase the clearance of the Gd-chelate during hemodialysis.25
Peritoneal dialysis may not remove gadolinium as effectively: Joffe et al24 reported that after 22 days of continuous ambulatory peritoneal dialysis, only 69% of the total amount of gadodiamide had been excreted, suggesting a very low peritoneal clearance.
SPECULATIVE PATHOGENESIS
Although a causal relationship between gadolinium use in patients with renal dysfunction and NSF has not been definitively established, the data derived from case reports assuredly raise this suspicion. Furthermore, on biopsy, gadolinium can be found in the skin of patients with NSF, adding evidence of causality.26–28
The mechanism by which Gd3+ might trigger NSF is still not understood. A plausible speculation is that if renal function is reduced, the half-life of the Gd-chelate molecule is significantly increased, as is the chance of Gd3+ dissociating from its chelate, leading to increased tissue exposure. Vascular trauma and endothelial dysfunction may allow free Gd3+ to enter tissues more easily, where macrophages phagocytose the metal, produce local profibrotic cytokines, and send out signals that recruit circulating fibrocytes to the tissues. Once in tissues, circulating fibrocytes induce a fibrosing process that is indistinguishable from normal scar formation.29
TREATMENTS LACK DATA
There is no consistently successful treatment for NSF.
In isolated reports, successful kidney transplantation slowed the skin fibrosis, but these findings need to be confirmed.30,31 Data from case reports should be interpreted very cautiously, as they are by nature sporadic and anecdotal. Moreover most of the reports of NSF were published on Web sites or as editorials and did not undergo exhaustive peer review. Because the evidence is weak, kidney transplantation should not be recommended as a treatment for NSF.
Oral steroids, plasmapheresis, extracorporeal photopheresis, thalidomide, topical ultraviolet-A therapy, and other treatments have yielded very conflicting results, with only anecdotal improvement of symptoms. In a recent case report,32 the use of intravenous sodium thiosulfate in addition to aggressive physical therapy provided some benefit by reducing the pain and improving the skin lesions.
Because of the lack of strong evidence of efficacy, we cannot advocate the use of any of these treatments until larger clinical trial results are available. Aggressive physical therapy along with appropriate pain control may have benefits and should be offered to all patients suffering from NSF.
Avoid gadolinium exposure in patients with renal insufficiency
The FDA33 recently asked manufacturers to include a new boxed warning on the product labeling of all gadolinium-based contrast agents (Magnevist, MultiHance, Omniscan, Opti-MARK, ProHance), due to risk of NSF in patients with acute or chronic severe renal insufficiency (GFR < 30 mL/minute/1.73 m2) and in patients with acute renal insufficiency of any severity due to hepatorenal syndrome or in the perioperative liver transplantation period.
For the time being, gadolinium should be contraindicated in patients with acute kidney injury and chronic kidney disease stages 4 and 5 and in those who are on renal replacement therapy (either hemodialysis or peritoneal dialysis). If an MRI study with gadolinium-based contrast is absolutely required in a patient with end-stage renal disease or advanced chronic kidney disease, an agent other than gadodiamide should be used in the lowest possible dose.
Will hemodialysis prevent NSF?
In a patient who is already on hemodialysis, it seems prudent to perform hemodialysis soon after gadolinium exposure and again the day after exposure to increase gadolinium elimination. However, to date, there are no data to support the theory that doing this will prevent NSF.
Because peritoneal dialysis has been reported to clear gadolinium poorly, use of gadolinium is contraindicated. If gadolinium is absolutely needed, either more-aggressive peritoneal dialysis (keeping the abdomen “wet”) or temporary hemodialysis may be considered.
For patients with advanced chronic kidney disease who are not yet on renal replacement therapy, the use of gadolinium is contraindicated, and hemodialysis should not be empirically recommended after gadolinium exposure because we have no evidence to support its utility and because hemodialysis may cause harm.
Nephrology consultation should be considered before any gadolinium use in a patient with impaired renal function, whether acute or chronic.
- Cowper SE, Robin HS, Steinberg SM, Su LD, Gupta S, LeBoit PE. Scleromyxoedema-like cutaneous diseases in renal-dialysis patients. Lancet 2000; 356:1000–1001.
- Galan A, Cowper SE, Bucala R. Nephrogenic systemic fibrosis (nephrogenic fibrosing dermopathy). Curr Opin Rheumatol 2006; 18:614–617.
- Cowper SE. Nephrogenic fibrosing dermopathy: the first 6 years. Curr Opin Rheumatol 2003; 15:785–790.
- Ting WW, Stone MS, Madison KC, Kurtz K. Nephrogenic fibrosing dermopathy with systemic involvement. Arch Dermatol 2003; 139:903–906.
- Kucher C, Steere J, Elenitsas R, Siegel DL, Xu X. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis with diaphragmatic involvement in a patient with respiratory failure. J Am Acad Dermatol 2006; 54:S31–S34.
- Jimenez SA, Artlett CM, Sandorfi N, et al. Dialysis-associated systemic fibrosis (nephrogenic fibrosing dermopathy): study of inflammatory cells and transforming growth factor beta1 expression in affected skin. Arthritis Rheum 2004; 50:2660–2666.
- Mendoza FA, Artlett CM, Sandorfi N, Latinis K, Piera-Velazquez S, Jimenez SA. Description of 12 cases of nephrogenic fibrosing dermopathy and review of the literature. Semin Arthritis Rheum 2006; 35:238–249.
- Todd DJ, Kagan A, Chibnik LB, Kay J. Cutaneous changes of nephrogenic systemic fibrosis: predictor of early mortality and association with gadolinium exposure. Arthritis Rheum 2007; 56:3433–3441.
- Cowper SE, Bucala R, Leboit PE. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis—setting the record straight. Semin Arthritis Rheum 2006; 35:208–210.
- Quan TE, Cowper S, Wu SP, Bockenstedt LK, Bucala R. Circulating fibrocytes: collagen-secreting cells of the peripheral blood. Int J Biochem Cell Biol 2004; 36:598–606.
- Grobner T. Gadolinium—a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis? Nephrol Dial Transplant 2006; 21:1104–1108.
- Marckmann P, Skov L, Rossen K, et al. Nephrogenic systemic fibrosis: suspected causative role of gadodiamide used for contrast-enhanced magnetic resonance imaging. J Am Soc Nephrol 2006; 17:2359–2362.
- Kuo PH, Kanal E, Abu-Alfa AK, Cowper SE. Gadolinium-based MR contrast agents and nephrogenic systemic fibrosis. Radiology 2007; 242:647–649.
- Broome DR, Girguis MS, Baron PW, Cottrell AC, Kjellin I, Kirk GA. Gadodiamide-associated nephrogenic systemic fibrosis: why radiologists should be concerned. AJR Am J Roentgenol 2007; 188:586–592.
- Khurana A, Runge VM, Narayanan M, Greene JF, Nickel AE. Nephrogenic systemic fibrosis: a review of 6 cases temporally related to gadodiamide injection (Omniscan). Invest Radiol 2007; 42:139–145.
- Deo A, Fogel M, Cowper SE. Nephrogenic systemic fibrosis: a population study examining the relationship of disease development to gadolinium exposure. Clin J Am Soc Nephrol 2007; 2:264–267.
- US Centers for Disease Control and Prevention (CDC). Nephrogenic fibrosing dermopathy associated with exposure to gadolinium-containing contrast agents—St. Louis, Missouri, 2002–2006. MMWR Morb Mortal Wkly Rep 2007; 56:137–141.
- Sadowski EA, Bennett LK, Chan MR, et al. Nephrogenic systemic fibrosis: risk factors and incidence estimation. Radiology 2007; 243:148–157.
- Marckmann P, Skov L, Rossen K, Heaf JG, Thomsen HS. Case-control study of gadodiamide-related nephrogenic systemic fibrosis. Nephrol Dial Transplant 2007 May 4; e-pub ahead of print.
- Swaminathan S, Ahmed I, McCarthy JT, et al. Nephrogenic fibrosing dermopathy and high-dose erythropoietin therapy. Ann Intern Med 2006; 145:234–235.
- Jain SM, Wesson S, Hassanein A, et al. Nephrogenic fibrosing dermopathy in pediatric patients. Pediatr Nephrol 2004; 19:467–470.
- Fazeli A, Lio PA, Liu V. Nephrogenic fibrosing dermopathy: are ACE inhibitors the missing link? (Letter). Arch Dermatol 2004; 140:1401.
- Bellin MF. MR contrast agents, the old and the new. Eur J Radiol 2006; 60:314–323.
- Joffe P, Thomsen HS, Meusel M. Pharmacokinetics of gadodiamide injection in patients with severe renal insufficiency and patients undergoing hemodialysis or continuous ambulatory peritoneal dialysis. Acad Radiol 1998; 5:491–502.
- Ueda J, Furukawa T, Higashino K, et al. Permeability of iodinated and MR contrast media through two types of hemodialysis membrane. Eur J Radiol 1999; 31:76–80.
- Boyd AS, Zic JA, Abraham JL. Gadolinium deposition in nephrogenic fibrosing dermopathy. J Am Acad Dermatol 2007; 56:27–30.
- High WA, Ayers RA, Chandler J, Zito G, Cowper SE. Gadolinium is detectable within the tissue of patients with nephrogenic systemic fibrosis. J Am Acad Dermatol 2007; 56:21–26.
- High WA, Ayers RA, Cowper SE. Gadolinium is quantifiable within the tissue of patients with nephrogenic systemic fibrosis, J Am Acad Dermatol 2007; 56:710–712.
- Perazella MA. Nephrogenic systemic fibrosis, kidney disease, and gadolinium: is there a link? Clin J Am Soc Nephrol 2007; 2:200–202.
- Cowper SE. Nephrogenic systemic fibrosis: The nosological and conceptual evolution of nephrogenic fibrosing dermopathy. Am J Kidney Dis 2005; 46:763–765.
- Jan F, Segal JM, Dyer J, LeBoit P, Siegfried E, Frieden IJ. Nephrogenic fibrosing dermopathy: two pediatric cases. J Pediatr 2003; 143:678–681.
- Yerram P, Saab G, Karuparthi PR, Hayden MR, Khanna R. Nephrogenic systemic fibrosis: a mysterious disease in patients with renal failure—role of gadolinium-based contrast media in causation and the beneficial effect of intravenous sodium thiosulfate. Clin J Am Soc Nephrol 2007; 2:258–263.
- US Food and Drug Administration. Accessed 01/03/08. http://www.fda.gov/cder/drug/InfoSheets/HCP/gcca_200705.htm.
The use of gadolinium as a contrast agent in magnetic resonance imaging (MRI) in patients with impaired kidney function has come under scrutiny because of recent reports of a potential association between its use and nephrogenic systemic fibrosis (NSF).
This entity was first identified in the United States in 1997. Cowper et al1 in 2000 described 15 hemodialysis patients who developed thickening and hardening of the skin with brawny hyperpigmentation, papules, and subcutaneous nodules on the extremities.
This “new disease” was initially called “nephrogenic fibrosing dermopathy,” as it was exclusively seen in patients with renal impairment and was thought to affect only the skin and subcutaneous tissue. With growing evidence of the extent and pathogenicity of the fibrosis in visceral organs, the nomenclature was changed to NSF, to better reflect the systemic nature of the disease.
PRESENTATION: MILD TO DEVASTATING
NSF has thus far been reported only in patients with renal impairment, most of whom were dialysis-dependent. It does not seem to be more common in one sex or the other, in any age range, or in any ethnic group. It can range in severity from mild to a devastating scleroderma-like systemic fibrosing disorder.
The heart, lungs, skeletal muscle, and diaphragm can also be involved, sometimes leading to serious complications and death.4–6
The disease is usually progressive and unremitting. Mendoza et al,7 in a review of 12 cases of NSF, reported that the disease had a progressive course in 6 patients, of whom 3 died within 2 years and 3 were ultimately confined to a wheelchair. More severe findings and rapid progression of the skin disease are associated with a poor prognosis.
Todd et al8 prospectively examined 186 dialysis patients to look for possible NSF. Of those with skin changes consistent with NSF, 48% died within 2 years, compared with 20% of those without these skin changes. Cardiovascular causes accounted for 58% of the deaths in patients with cutaneous changes of NSF and for 48% of the deaths in patients without these changes. Most of the excess deaths occurred within 6 months after the skin examination, suggesting an increased risk for early death in patients with skin changes suggestive of NSF.
DIAGNOSIS OF NSF IS CLINICAL
At presentation, NSF is frequently misdiagnosed and treated as cellulitis or edema. However, now that subspecialists—especially dermatologists, rheumatologists, and nephrologists—are becoming more aware of it, the correct diagnosis is being made earlier.
NSF should be suspected in any patient with underlying renal dysfunction—especially if on dialysis and if he or she has received a gadolinium contrast agent during MRI—who develops scleroderma-like cutaneous lesions affecting the distal extremities. Because most health care providers are still unfamiliar with this emerging disease, patients with renal impairment and suspected NSF should be referred to a rheumatologist or dermatologist to confirm the diagnosis, which is mainly entertained on a clinical basis. There is no laboratory biomarker for NSF.
A deep incisional skin biopsy may aid in the diagnosis. Due to the regional distribution of the disease, sampling error may occur, and repeat biopsy is warranted if the initial biopsy is nondiagnostic but the clinical picture suggests NSF.
Histopathologic examination typically shows lesions containing proliferation of dermal spindle cells, thick collagen bundles with surrounding clefts, and a variable amount of mucin and elastic fibers.2 A characteristic and almost pathognomonic staining profile is the immunohistochemical identification of CD34 reactivity in the fibroblast-like cells (Figure 3). Cells expressing CD34 are normally found in the umbilical cord, the bone marrow (as pluripotential hematopoietic stem cells), and in the vascular endothelium. How they come to be in the skin is still speculative, but their presence suggests that circulating fibrocytes migrate from the bone marrow and deposit in the skin and other organs.9,10
Pulmonary function testing can be done to rule out lung involvement and transthoracic two-dimensional echocardiography can be done to rule out possible cardiomyopathy if these conditions are suggested by examination at the time of diagnosis.7 Muscle biopsy is not necessary to determine the extent of systemic involvement, since the findings do not necessarily correlate with other systemic involvement.
DIFFERENTIAL DIAGNOSIS
An important diagnostic feature of NSF is that it spares the face, a finding derived from all reported and confirmed cases of NSF (Figure 2). In contrast, scleromyxedema, systemic scleroderma, and morphea often involve the face.
Scleromyxedema is often associated with monoclonal gammopathy (usually an immunoglobulin G lambda paraproteinemia) whereas NSF is not.
Scleroderma is supported by the findings of Raynaud’s phenomenon, antinuclear antibodies, and either anticentromere or anti-DNA topoisomerase I (Scl-70) antibodies, but the absence of these antibodies does not necessarily rule it out.
Eosinophilic fasciitis is diagnosed on the basis of histologic examination of a deep wedge skin biopsy specimen that includes fascia.
Other diagnoses that should be considered include amyloidosis and calciphylaxis.
ASSOCIATION WITH GADOLINIUM: WHAT IS THE EVIDENCE?
Case series
The association of gadolinium use with NSF has been described in several case reports and case series.
Grobner11 reported that administration of gadodiamide (Omniscan, a gadolinium compound) for MRI was associated with NSF in five patients on chronic hemodialysis who had end-stage renal disease. Their ages ranged from 43 to 74 years, and they had been on dialysis from 10 to 58 months. The time of onset of NSF ranged from 2 to 4 weeks after exposure to gadodiamide.
Marckmann et al12 reported that NSF developed in 13 (3.5%) of 370 patients with severe kidney disease who received gadodiamide. Five of the 13 patients had stage 5 (advanced) chronic kidney disease and were not yet on renal replacement therapy, 7 were on hemodialysis, and 1 was on peritoneal dialysis. The time of onset ranged from 2 to 75 days (median 25 days) after exposure.
Kuo et al13 similarly estimated the incidence of NSF at approximately 3% in patients with severe renal failure who receive intravenous gadolinium-based contrast material for MRI.
Broome et al14 reported that 12 patients developed NSF within 2 to 11 weeks after receiving gadodiamide. Eight of the 12 patients had end-stage renal disease and were on hemodialysis; the other 4 patients had acute kidney injury attributed to hepatorenal syndrome, and 3 of these 4 patients were on hemodialysis.
Khurana et al15 reported that 6 patients on hemodialysis developed NSF from 2 weeks to 2 months after receiving a dose of gadodiamide of between 0.11 and 0.36 mmol/kg. These doses are high, and the findings suggest an association between the gadolinium dose and NSF. The dose approved by the US Food and Drug Administration (FDA) is only 0.1 mmol/kg, and the use of gadolinium is approved only in MRI. However, higher doses (0.3–0.4 mmol/kg) are widely used in practice for better imaging quality in magnetic resonance angiography (MRA).
Deo et al16 reported 3 cases of NSF in 87 patients with end-stage renal disease who underwent 123 radiologic studies with gadolinium. No patient with end-stage renal disease who was not exposed to gadolinium developed NSF, and the association between exposure to gadolinium and the subsequent development of NSF was statistically significant (P = .006). The authors concluded that each gadolinium study presented a 2.4% risk of NSF in end-stage renal disease patients.
This retrospective study is flawed by not having been cross-sectional or case-controlled, since the other 84 patients who received gadolinium were not examined at all to establish the absence of NSF.
Case-control studies
More evidence of association of NSF with gadolinium exposure comes from other reports.
Physicians in St. Louis, MO,17 identified 33 cases of NSF and performed a case-control study, matching each of 19 of the patients (for whom data were available and who met their entry criteria) with 3 controls. They found that exposure to gadolinium was independently associated with the development of NSF.
Sadowski et al18 reported that 13 patients with biopsy-confirmed NSF all had been exposed to gadodiamide and one had been exposed to gadobenate (MultiHANCE) in addition to gadodiamide. All 13 patients had renal insufficiency, with an estimated glomerular filtration rate (GFR) less than 60 mL/minute/1.73 m2. The investigators compared this group with a control group of patients with renal insufficiency who did not develop NSF. The NSF group had more proinflammatory events (P < .001) and more gadolinium-contrast-enhanced MRI examinations per patient (P = .002) than the control group.
Marckmann et al19 compared 19 patients who had histologically proven cases of NSF and 19 sex- and age-matched controls; all 38 patients had chronic kidney disease and had been exposed to gadolinium. Patients with NSF had received higher cumulative doses of gadodiamide and higher doses of erythropoietin and had higher serum concentrations of ionized calcium and phosphate than did their controls, as did patients with severe NSF compared with those with nonsevere NSF.
Comment. All the above reports are limited by their study design and suffer from recognition bias because not all of the patients with severe renal insufficiency who were exposed to gadolinium were examined for possible asymptomatic skin changes that might be characteristic of NSF. Therefore, it is impossible to be certain that all of the patients classified as not having NSF truly did not have it or did not subsequently develop it. Furthermore, the reports lacked standardized diagnostic criteria. Hence, the real prevalence and incidence of NSF are difficult to determine.
A cross-sectional study
As mentioned above, Todd et al8 examined 186 dialysis patients for cutaneous changes of NSF (using a scoring system based on hyper-pigmentation, hardening, and tethering of skin on the extremities). Patients who had been exposed to gadolinium had a higher risk of developing these skin changes than did nonexposed patients (odds ratio 14.7, 95% confidence interval 1.9–117.0). More importantly, the investigators found cutaneous changes of NSF in 25 (13%) of the 186 patients, 4 of whom had prior skin biopsies available for review, each revealing the histologic changes of NSF. This study suggests that NSF may be more prevalent than previously thought.
Is kidney dysfunction always present?
All the reported patients with NSF had underlying renal impairment. The renal dysfunction ranged from acute kidney injury to advanced chronic kidney disease (estimated GFR < 30 mL/minute/1.73 m2) and end-stage renal disease on renal replacement therapy, ie, hemodialysis or peritoneal dialysis. The incidence of NSF does not seem to be related to the cause of the underlying kidney disease.
What other diseases or comorbidities can be associated with NSF?
It is still unclear why not every patient with advanced renal failure develops NSF after exposure to gadolinium.
A variety of complex diseases and conditions have been reported to be associated with NSF, with no clear-cut evidence of causality or trigger. These include hypercoagulability states, thrombotic events, surgical procedures (especially those with reconstructive vascular components), calciphylaxis, kidney transplantation, hepatic disease (hepatorenal syndrome, liver transplantation, and hepatitis B and C), idiopathic pulmonary fibrosis, systemic lupus erythematosus, hypothyroidism, elevated serum ionized calcium or serum phosphate, hyperparathyroidism, and metabolic acidosis. A possible explanation is that most of these conditions are associated with an increased use of MRI or MRA testing (eg, in the workup for kidney or liver transplantation).
Many drugs have also been reported to be associated with NSF, including high-dose erythropoietin,20 sevelamer (Renagel),21 and, conversely, lack of angiotensin-converting enzyme inhibitor therapy,22 but none of these findings has been reproduced to date.
GADOLINIUM CHARACTERISTICS AND PHARMACOKINETICS
Gadolinium is a rare-earth lanthanide metallic element (atomic number 64) that is used in MRI and MRA because of its paramagnetic properties that enhance the quality of imaging. Its ionic form (Gd3+) is highly toxic if injected intravenously, so it is typically bound to a “chelate” to decrease its toxicity.23 The chelate stabilizes Gd3+ and thereby prevents its dissociation in vivo. These Gd-chelates can be classified (Table 2) according to their charge (ionic vs nonionic) and their structure (linear vs cyclic).
Most of the reported cases of NSF have been in patients who received gadodiamide, a nonionic, linear agent. Why gadodiamide has the highest rates of association with NSF is still unclear; perhaps it is simply the most widely used agent. Also, linear Gd compounds may be less stable and more likely to dissociate in vivo. The updated FDA Public Health Advisory in May 2007 warned against the use of all gadolinium-containing contrast agents for MRI, not just gadodiamide.
After intravenous injection, Gd-chelate equilibrates rapidly (within 2 hours) in the extracellular space. Very little of it enters into cells or binds to proteins. It is eliminated unchanged in the glomerular filtrate with no tubular secretion. In a study by Joffe et al,24 the elimination half-life of gadodiamide in patients with severely reduced renal function was considerably longer than in healthy volunteers (34.3 hours ± 22.9 vs 1.3 hours ± 0.25).
Since gadolinium compounds are not protein-bound and have a limited volume of distribution, they are typically removed by hemodialysis. Joffe et al found that an average of 65% of the gadodiamide was removed in a single hemodialysis session. However, they did not describe the specific features of the hemodialysis session, and it took four hemodialysis treatments to remove 99% of a single dose of gadolinium.24 A dialysis membrane with high permeability (large pores) seems to increase the clearance of the Gd-chelate during hemodialysis.25
Peritoneal dialysis may not remove gadolinium as effectively: Joffe et al24 reported that after 22 days of continuous ambulatory peritoneal dialysis, only 69% of the total amount of gadodiamide had been excreted, suggesting a very low peritoneal clearance.
SPECULATIVE PATHOGENESIS
Although a causal relationship between gadolinium use in patients with renal dysfunction and NSF has not been definitively established, the data derived from case reports assuredly raise this suspicion. Furthermore, on biopsy, gadolinium can be found in the skin of patients with NSF, adding evidence of causality.26–28
The mechanism by which Gd3+ might trigger NSF is still not understood. A plausible speculation is that if renal function is reduced, the half-life of the Gd-chelate molecule is significantly increased, as is the chance of Gd3+ dissociating from its chelate, leading to increased tissue exposure. Vascular trauma and endothelial dysfunction may allow free Gd3+ to enter tissues more easily, where macrophages phagocytose the metal, produce local profibrotic cytokines, and send out signals that recruit circulating fibrocytes to the tissues. Once in tissues, circulating fibrocytes induce a fibrosing process that is indistinguishable from normal scar formation.29
TREATMENTS LACK DATA
There is no consistently successful treatment for NSF.
In isolated reports, successful kidney transplantation slowed the skin fibrosis, but these findings need to be confirmed.30,31 Data from case reports should be interpreted very cautiously, as they are by nature sporadic and anecdotal. Moreover most of the reports of NSF were published on Web sites or as editorials and did not undergo exhaustive peer review. Because the evidence is weak, kidney transplantation should not be recommended as a treatment for NSF.
Oral steroids, plasmapheresis, extracorporeal photopheresis, thalidomide, topical ultraviolet-A therapy, and other treatments have yielded very conflicting results, with only anecdotal improvement of symptoms. In a recent case report,32 the use of intravenous sodium thiosulfate in addition to aggressive physical therapy provided some benefit by reducing the pain and improving the skin lesions.
Because of the lack of strong evidence of efficacy, we cannot advocate the use of any of these treatments until larger clinical trial results are available. Aggressive physical therapy along with appropriate pain control may have benefits and should be offered to all patients suffering from NSF.
Avoid gadolinium exposure in patients with renal insufficiency
The FDA33 recently asked manufacturers to include a new boxed warning on the product labeling of all gadolinium-based contrast agents (Magnevist, MultiHance, Omniscan, Opti-MARK, ProHance), due to risk of NSF in patients with acute or chronic severe renal insufficiency (GFR < 30 mL/minute/1.73 m2) and in patients with acute renal insufficiency of any severity due to hepatorenal syndrome or in the perioperative liver transplantation period.
For the time being, gadolinium should be contraindicated in patients with acute kidney injury and chronic kidney disease stages 4 and 5 and in those who are on renal replacement therapy (either hemodialysis or peritoneal dialysis). If an MRI study with gadolinium-based contrast is absolutely required in a patient with end-stage renal disease or advanced chronic kidney disease, an agent other than gadodiamide should be used in the lowest possible dose.
Will hemodialysis prevent NSF?
In a patient who is already on hemodialysis, it seems prudent to perform hemodialysis soon after gadolinium exposure and again the day after exposure to increase gadolinium elimination. However, to date, there are no data to support the theory that doing this will prevent NSF.
Because peritoneal dialysis has been reported to clear gadolinium poorly, use of gadolinium is contraindicated. If gadolinium is absolutely needed, either more-aggressive peritoneal dialysis (keeping the abdomen “wet”) or temporary hemodialysis may be considered.
For patients with advanced chronic kidney disease who are not yet on renal replacement therapy, the use of gadolinium is contraindicated, and hemodialysis should not be empirically recommended after gadolinium exposure because we have no evidence to support its utility and because hemodialysis may cause harm.
Nephrology consultation should be considered before any gadolinium use in a patient with impaired renal function, whether acute or chronic.
The use of gadolinium as a contrast agent in magnetic resonance imaging (MRI) in patients with impaired kidney function has come under scrutiny because of recent reports of a potential association between its use and nephrogenic systemic fibrosis (NSF).
This entity was first identified in the United States in 1997. Cowper et al1 in 2000 described 15 hemodialysis patients who developed thickening and hardening of the skin with brawny hyperpigmentation, papules, and subcutaneous nodules on the extremities.
This “new disease” was initially called “nephrogenic fibrosing dermopathy,” as it was exclusively seen in patients with renal impairment and was thought to affect only the skin and subcutaneous tissue. With growing evidence of the extent and pathogenicity of the fibrosis in visceral organs, the nomenclature was changed to NSF, to better reflect the systemic nature of the disease.
PRESENTATION: MILD TO DEVASTATING
NSF has thus far been reported only in patients with renal impairment, most of whom were dialysis-dependent. It does not seem to be more common in one sex or the other, in any age range, or in any ethnic group. It can range in severity from mild to a devastating scleroderma-like systemic fibrosing disorder.
The heart, lungs, skeletal muscle, and diaphragm can also be involved, sometimes leading to serious complications and death.4–6
The disease is usually progressive and unremitting. Mendoza et al,7 in a review of 12 cases of NSF, reported that the disease had a progressive course in 6 patients, of whom 3 died within 2 years and 3 were ultimately confined to a wheelchair. More severe findings and rapid progression of the skin disease are associated with a poor prognosis.
Todd et al8 prospectively examined 186 dialysis patients to look for possible NSF. Of those with skin changes consistent with NSF, 48% died within 2 years, compared with 20% of those without these skin changes. Cardiovascular causes accounted for 58% of the deaths in patients with cutaneous changes of NSF and for 48% of the deaths in patients without these changes. Most of the excess deaths occurred within 6 months after the skin examination, suggesting an increased risk for early death in patients with skin changes suggestive of NSF.
DIAGNOSIS OF NSF IS CLINICAL
At presentation, NSF is frequently misdiagnosed and treated as cellulitis or edema. However, now that subspecialists—especially dermatologists, rheumatologists, and nephrologists—are becoming more aware of it, the correct diagnosis is being made earlier.
NSF should be suspected in any patient with underlying renal dysfunction—especially if on dialysis and if he or she has received a gadolinium contrast agent during MRI—who develops scleroderma-like cutaneous lesions affecting the distal extremities. Because most health care providers are still unfamiliar with this emerging disease, patients with renal impairment and suspected NSF should be referred to a rheumatologist or dermatologist to confirm the diagnosis, which is mainly entertained on a clinical basis. There is no laboratory biomarker for NSF.
A deep incisional skin biopsy may aid in the diagnosis. Due to the regional distribution of the disease, sampling error may occur, and repeat biopsy is warranted if the initial biopsy is nondiagnostic but the clinical picture suggests NSF.
Histopathologic examination typically shows lesions containing proliferation of dermal spindle cells, thick collagen bundles with surrounding clefts, and a variable amount of mucin and elastic fibers.2 A characteristic and almost pathognomonic staining profile is the immunohistochemical identification of CD34 reactivity in the fibroblast-like cells (Figure 3). Cells expressing CD34 are normally found in the umbilical cord, the bone marrow (as pluripotential hematopoietic stem cells), and in the vascular endothelium. How they come to be in the skin is still speculative, but their presence suggests that circulating fibrocytes migrate from the bone marrow and deposit in the skin and other organs.9,10
Pulmonary function testing can be done to rule out lung involvement and transthoracic two-dimensional echocardiography can be done to rule out possible cardiomyopathy if these conditions are suggested by examination at the time of diagnosis.7 Muscle biopsy is not necessary to determine the extent of systemic involvement, since the findings do not necessarily correlate with other systemic involvement.
DIFFERENTIAL DIAGNOSIS
An important diagnostic feature of NSF is that it spares the face, a finding derived from all reported and confirmed cases of NSF (Figure 2). In contrast, scleromyxedema, systemic scleroderma, and morphea often involve the face.
Scleromyxedema is often associated with monoclonal gammopathy (usually an immunoglobulin G lambda paraproteinemia) whereas NSF is not.
Scleroderma is supported by the findings of Raynaud’s phenomenon, antinuclear antibodies, and either anticentromere or anti-DNA topoisomerase I (Scl-70) antibodies, but the absence of these antibodies does not necessarily rule it out.
Eosinophilic fasciitis is diagnosed on the basis of histologic examination of a deep wedge skin biopsy specimen that includes fascia.
Other diagnoses that should be considered include amyloidosis and calciphylaxis.
ASSOCIATION WITH GADOLINIUM: WHAT IS THE EVIDENCE?
Case series
The association of gadolinium use with NSF has been described in several case reports and case series.
Grobner11 reported that administration of gadodiamide (Omniscan, a gadolinium compound) for MRI was associated with NSF in five patients on chronic hemodialysis who had end-stage renal disease. Their ages ranged from 43 to 74 years, and they had been on dialysis from 10 to 58 months. The time of onset of NSF ranged from 2 to 4 weeks after exposure to gadodiamide.
Marckmann et al12 reported that NSF developed in 13 (3.5%) of 370 patients with severe kidney disease who received gadodiamide. Five of the 13 patients had stage 5 (advanced) chronic kidney disease and were not yet on renal replacement therapy, 7 were on hemodialysis, and 1 was on peritoneal dialysis. The time of onset ranged from 2 to 75 days (median 25 days) after exposure.
Kuo et al13 similarly estimated the incidence of NSF at approximately 3% in patients with severe renal failure who receive intravenous gadolinium-based contrast material for MRI.
Broome et al14 reported that 12 patients developed NSF within 2 to 11 weeks after receiving gadodiamide. Eight of the 12 patients had end-stage renal disease and were on hemodialysis; the other 4 patients had acute kidney injury attributed to hepatorenal syndrome, and 3 of these 4 patients were on hemodialysis.
Khurana et al15 reported that 6 patients on hemodialysis developed NSF from 2 weeks to 2 months after receiving a dose of gadodiamide of between 0.11 and 0.36 mmol/kg. These doses are high, and the findings suggest an association between the gadolinium dose and NSF. The dose approved by the US Food and Drug Administration (FDA) is only 0.1 mmol/kg, and the use of gadolinium is approved only in MRI. However, higher doses (0.3–0.4 mmol/kg) are widely used in practice for better imaging quality in magnetic resonance angiography (MRA).
Deo et al16 reported 3 cases of NSF in 87 patients with end-stage renal disease who underwent 123 radiologic studies with gadolinium. No patient with end-stage renal disease who was not exposed to gadolinium developed NSF, and the association between exposure to gadolinium and the subsequent development of NSF was statistically significant (P = .006). The authors concluded that each gadolinium study presented a 2.4% risk of NSF in end-stage renal disease patients.
This retrospective study is flawed by not having been cross-sectional or case-controlled, since the other 84 patients who received gadolinium were not examined at all to establish the absence of NSF.
Case-control studies
More evidence of association of NSF with gadolinium exposure comes from other reports.
Physicians in St. Louis, MO,17 identified 33 cases of NSF and performed a case-control study, matching each of 19 of the patients (for whom data were available and who met their entry criteria) with 3 controls. They found that exposure to gadolinium was independently associated with the development of NSF.
Sadowski et al18 reported that 13 patients with biopsy-confirmed NSF all had been exposed to gadodiamide and one had been exposed to gadobenate (MultiHANCE) in addition to gadodiamide. All 13 patients had renal insufficiency, with an estimated glomerular filtration rate (GFR) less than 60 mL/minute/1.73 m2. The investigators compared this group with a control group of patients with renal insufficiency who did not develop NSF. The NSF group had more proinflammatory events (P < .001) and more gadolinium-contrast-enhanced MRI examinations per patient (P = .002) than the control group.
Marckmann et al19 compared 19 patients who had histologically proven cases of NSF and 19 sex- and age-matched controls; all 38 patients had chronic kidney disease and had been exposed to gadolinium. Patients with NSF had received higher cumulative doses of gadodiamide and higher doses of erythropoietin and had higher serum concentrations of ionized calcium and phosphate than did their controls, as did patients with severe NSF compared with those with nonsevere NSF.
Comment. All the above reports are limited by their study design and suffer from recognition bias because not all of the patients with severe renal insufficiency who were exposed to gadolinium were examined for possible asymptomatic skin changes that might be characteristic of NSF. Therefore, it is impossible to be certain that all of the patients classified as not having NSF truly did not have it or did not subsequently develop it. Furthermore, the reports lacked standardized diagnostic criteria. Hence, the real prevalence and incidence of NSF are difficult to determine.
A cross-sectional study
As mentioned above, Todd et al8 examined 186 dialysis patients for cutaneous changes of NSF (using a scoring system based on hyper-pigmentation, hardening, and tethering of skin on the extremities). Patients who had been exposed to gadolinium had a higher risk of developing these skin changes than did nonexposed patients (odds ratio 14.7, 95% confidence interval 1.9–117.0). More importantly, the investigators found cutaneous changes of NSF in 25 (13%) of the 186 patients, 4 of whom had prior skin biopsies available for review, each revealing the histologic changes of NSF. This study suggests that NSF may be more prevalent than previously thought.
Is kidney dysfunction always present?
All the reported patients with NSF had underlying renal impairment. The renal dysfunction ranged from acute kidney injury to advanced chronic kidney disease (estimated GFR < 30 mL/minute/1.73 m2) and end-stage renal disease on renal replacement therapy, ie, hemodialysis or peritoneal dialysis. The incidence of NSF does not seem to be related to the cause of the underlying kidney disease.
What other diseases or comorbidities can be associated with NSF?
It is still unclear why not every patient with advanced renal failure develops NSF after exposure to gadolinium.
A variety of complex diseases and conditions have been reported to be associated with NSF, with no clear-cut evidence of causality or trigger. These include hypercoagulability states, thrombotic events, surgical procedures (especially those with reconstructive vascular components), calciphylaxis, kidney transplantation, hepatic disease (hepatorenal syndrome, liver transplantation, and hepatitis B and C), idiopathic pulmonary fibrosis, systemic lupus erythematosus, hypothyroidism, elevated serum ionized calcium or serum phosphate, hyperparathyroidism, and metabolic acidosis. A possible explanation is that most of these conditions are associated with an increased use of MRI or MRA testing (eg, in the workup for kidney or liver transplantation).
Many drugs have also been reported to be associated with NSF, including high-dose erythropoietin,20 sevelamer (Renagel),21 and, conversely, lack of angiotensin-converting enzyme inhibitor therapy,22 but none of these findings has been reproduced to date.
GADOLINIUM CHARACTERISTICS AND PHARMACOKINETICS
Gadolinium is a rare-earth lanthanide metallic element (atomic number 64) that is used in MRI and MRA because of its paramagnetic properties that enhance the quality of imaging. Its ionic form (Gd3+) is highly toxic if injected intravenously, so it is typically bound to a “chelate” to decrease its toxicity.23 The chelate stabilizes Gd3+ and thereby prevents its dissociation in vivo. These Gd-chelates can be classified (Table 2) according to their charge (ionic vs nonionic) and their structure (linear vs cyclic).
Most of the reported cases of NSF have been in patients who received gadodiamide, a nonionic, linear agent. Why gadodiamide has the highest rates of association with NSF is still unclear; perhaps it is simply the most widely used agent. Also, linear Gd compounds may be less stable and more likely to dissociate in vivo. The updated FDA Public Health Advisory in May 2007 warned against the use of all gadolinium-containing contrast agents for MRI, not just gadodiamide.
After intravenous injection, Gd-chelate equilibrates rapidly (within 2 hours) in the extracellular space. Very little of it enters into cells or binds to proteins. It is eliminated unchanged in the glomerular filtrate with no tubular secretion. In a study by Joffe et al,24 the elimination half-life of gadodiamide in patients with severely reduced renal function was considerably longer than in healthy volunteers (34.3 hours ± 22.9 vs 1.3 hours ± 0.25).
Since gadolinium compounds are not protein-bound and have a limited volume of distribution, they are typically removed by hemodialysis. Joffe et al found that an average of 65% of the gadodiamide was removed in a single hemodialysis session. However, they did not describe the specific features of the hemodialysis session, and it took four hemodialysis treatments to remove 99% of a single dose of gadolinium.24 A dialysis membrane with high permeability (large pores) seems to increase the clearance of the Gd-chelate during hemodialysis.25
Peritoneal dialysis may not remove gadolinium as effectively: Joffe et al24 reported that after 22 days of continuous ambulatory peritoneal dialysis, only 69% of the total amount of gadodiamide had been excreted, suggesting a very low peritoneal clearance.
SPECULATIVE PATHOGENESIS
Although a causal relationship between gadolinium use in patients with renal dysfunction and NSF has not been definitively established, the data derived from case reports assuredly raise this suspicion. Furthermore, on biopsy, gadolinium can be found in the skin of patients with NSF, adding evidence of causality.26–28
The mechanism by which Gd3+ might trigger NSF is still not understood. A plausible speculation is that if renal function is reduced, the half-life of the Gd-chelate molecule is significantly increased, as is the chance of Gd3+ dissociating from its chelate, leading to increased tissue exposure. Vascular trauma and endothelial dysfunction may allow free Gd3+ to enter tissues more easily, where macrophages phagocytose the metal, produce local profibrotic cytokines, and send out signals that recruit circulating fibrocytes to the tissues. Once in tissues, circulating fibrocytes induce a fibrosing process that is indistinguishable from normal scar formation.29
TREATMENTS LACK DATA
There is no consistently successful treatment for NSF.
In isolated reports, successful kidney transplantation slowed the skin fibrosis, but these findings need to be confirmed.30,31 Data from case reports should be interpreted very cautiously, as they are by nature sporadic and anecdotal. Moreover most of the reports of NSF were published on Web sites or as editorials and did not undergo exhaustive peer review. Because the evidence is weak, kidney transplantation should not be recommended as a treatment for NSF.
Oral steroids, plasmapheresis, extracorporeal photopheresis, thalidomide, topical ultraviolet-A therapy, and other treatments have yielded very conflicting results, with only anecdotal improvement of symptoms. In a recent case report,32 the use of intravenous sodium thiosulfate in addition to aggressive physical therapy provided some benefit by reducing the pain and improving the skin lesions.
Because of the lack of strong evidence of efficacy, we cannot advocate the use of any of these treatments until larger clinical trial results are available. Aggressive physical therapy along with appropriate pain control may have benefits and should be offered to all patients suffering from NSF.
Avoid gadolinium exposure in patients with renal insufficiency
The FDA33 recently asked manufacturers to include a new boxed warning on the product labeling of all gadolinium-based contrast agents (Magnevist, MultiHance, Omniscan, Opti-MARK, ProHance), due to risk of NSF in patients with acute or chronic severe renal insufficiency (GFR < 30 mL/minute/1.73 m2) and in patients with acute renal insufficiency of any severity due to hepatorenal syndrome or in the perioperative liver transplantation period.
For the time being, gadolinium should be contraindicated in patients with acute kidney injury and chronic kidney disease stages 4 and 5 and in those who are on renal replacement therapy (either hemodialysis or peritoneal dialysis). If an MRI study with gadolinium-based contrast is absolutely required in a patient with end-stage renal disease or advanced chronic kidney disease, an agent other than gadodiamide should be used in the lowest possible dose.
Will hemodialysis prevent NSF?
In a patient who is already on hemodialysis, it seems prudent to perform hemodialysis soon after gadolinium exposure and again the day after exposure to increase gadolinium elimination. However, to date, there are no data to support the theory that doing this will prevent NSF.
Because peritoneal dialysis has been reported to clear gadolinium poorly, use of gadolinium is contraindicated. If gadolinium is absolutely needed, either more-aggressive peritoneal dialysis (keeping the abdomen “wet”) or temporary hemodialysis may be considered.
For patients with advanced chronic kidney disease who are not yet on renal replacement therapy, the use of gadolinium is contraindicated, and hemodialysis should not be empirically recommended after gadolinium exposure because we have no evidence to support its utility and because hemodialysis may cause harm.
Nephrology consultation should be considered before any gadolinium use in a patient with impaired renal function, whether acute or chronic.
- Cowper SE, Robin HS, Steinberg SM, Su LD, Gupta S, LeBoit PE. Scleromyxoedema-like cutaneous diseases in renal-dialysis patients. Lancet 2000; 356:1000–1001.
- Galan A, Cowper SE, Bucala R. Nephrogenic systemic fibrosis (nephrogenic fibrosing dermopathy). Curr Opin Rheumatol 2006; 18:614–617.
- Cowper SE. Nephrogenic fibrosing dermopathy: the first 6 years. Curr Opin Rheumatol 2003; 15:785–790.
- Ting WW, Stone MS, Madison KC, Kurtz K. Nephrogenic fibrosing dermopathy with systemic involvement. Arch Dermatol 2003; 139:903–906.
- Kucher C, Steere J, Elenitsas R, Siegel DL, Xu X. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis with diaphragmatic involvement in a patient with respiratory failure. J Am Acad Dermatol 2006; 54:S31–S34.
- Jimenez SA, Artlett CM, Sandorfi N, et al. Dialysis-associated systemic fibrosis (nephrogenic fibrosing dermopathy): study of inflammatory cells and transforming growth factor beta1 expression in affected skin. Arthritis Rheum 2004; 50:2660–2666.
- Mendoza FA, Artlett CM, Sandorfi N, Latinis K, Piera-Velazquez S, Jimenez SA. Description of 12 cases of nephrogenic fibrosing dermopathy and review of the literature. Semin Arthritis Rheum 2006; 35:238–249.
- Todd DJ, Kagan A, Chibnik LB, Kay J. Cutaneous changes of nephrogenic systemic fibrosis: predictor of early mortality and association with gadolinium exposure. Arthritis Rheum 2007; 56:3433–3441.
- Cowper SE, Bucala R, Leboit PE. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis—setting the record straight. Semin Arthritis Rheum 2006; 35:208–210.
- Quan TE, Cowper S, Wu SP, Bockenstedt LK, Bucala R. Circulating fibrocytes: collagen-secreting cells of the peripheral blood. Int J Biochem Cell Biol 2004; 36:598–606.
- Grobner T. Gadolinium—a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis? Nephrol Dial Transplant 2006; 21:1104–1108.
- Marckmann P, Skov L, Rossen K, et al. Nephrogenic systemic fibrosis: suspected causative role of gadodiamide used for contrast-enhanced magnetic resonance imaging. J Am Soc Nephrol 2006; 17:2359–2362.
- Kuo PH, Kanal E, Abu-Alfa AK, Cowper SE. Gadolinium-based MR contrast agents and nephrogenic systemic fibrosis. Radiology 2007; 242:647–649.
- Broome DR, Girguis MS, Baron PW, Cottrell AC, Kjellin I, Kirk GA. Gadodiamide-associated nephrogenic systemic fibrosis: why radiologists should be concerned. AJR Am J Roentgenol 2007; 188:586–592.
- Khurana A, Runge VM, Narayanan M, Greene JF, Nickel AE. Nephrogenic systemic fibrosis: a review of 6 cases temporally related to gadodiamide injection (Omniscan). Invest Radiol 2007; 42:139–145.
- Deo A, Fogel M, Cowper SE. Nephrogenic systemic fibrosis: a population study examining the relationship of disease development to gadolinium exposure. Clin J Am Soc Nephrol 2007; 2:264–267.
- US Centers for Disease Control and Prevention (CDC). Nephrogenic fibrosing dermopathy associated with exposure to gadolinium-containing contrast agents—St. Louis, Missouri, 2002–2006. MMWR Morb Mortal Wkly Rep 2007; 56:137–141.
- Sadowski EA, Bennett LK, Chan MR, et al. Nephrogenic systemic fibrosis: risk factors and incidence estimation. Radiology 2007; 243:148–157.
- Marckmann P, Skov L, Rossen K, Heaf JG, Thomsen HS. Case-control study of gadodiamide-related nephrogenic systemic fibrosis. Nephrol Dial Transplant 2007 May 4; e-pub ahead of print.
- Swaminathan S, Ahmed I, McCarthy JT, et al. Nephrogenic fibrosing dermopathy and high-dose erythropoietin therapy. Ann Intern Med 2006; 145:234–235.
- Jain SM, Wesson S, Hassanein A, et al. Nephrogenic fibrosing dermopathy in pediatric patients. Pediatr Nephrol 2004; 19:467–470.
- Fazeli A, Lio PA, Liu V. Nephrogenic fibrosing dermopathy: are ACE inhibitors the missing link? (Letter). Arch Dermatol 2004; 140:1401.
- Bellin MF. MR contrast agents, the old and the new. Eur J Radiol 2006; 60:314–323.
- Joffe P, Thomsen HS, Meusel M. Pharmacokinetics of gadodiamide injection in patients with severe renal insufficiency and patients undergoing hemodialysis or continuous ambulatory peritoneal dialysis. Acad Radiol 1998; 5:491–502.
- Ueda J, Furukawa T, Higashino K, et al. Permeability of iodinated and MR contrast media through two types of hemodialysis membrane. Eur J Radiol 1999; 31:76–80.
- Boyd AS, Zic JA, Abraham JL. Gadolinium deposition in nephrogenic fibrosing dermopathy. J Am Acad Dermatol 2007; 56:27–30.
- High WA, Ayers RA, Chandler J, Zito G, Cowper SE. Gadolinium is detectable within the tissue of patients with nephrogenic systemic fibrosis. J Am Acad Dermatol 2007; 56:21–26.
- High WA, Ayers RA, Cowper SE. Gadolinium is quantifiable within the tissue of patients with nephrogenic systemic fibrosis, J Am Acad Dermatol 2007; 56:710–712.
- Perazella MA. Nephrogenic systemic fibrosis, kidney disease, and gadolinium: is there a link? Clin J Am Soc Nephrol 2007; 2:200–202.
- Cowper SE. Nephrogenic systemic fibrosis: The nosological and conceptual evolution of nephrogenic fibrosing dermopathy. Am J Kidney Dis 2005; 46:763–765.
- Jan F, Segal JM, Dyer J, LeBoit P, Siegfried E, Frieden IJ. Nephrogenic fibrosing dermopathy: two pediatric cases. J Pediatr 2003; 143:678–681.
- Yerram P, Saab G, Karuparthi PR, Hayden MR, Khanna R. Nephrogenic systemic fibrosis: a mysterious disease in patients with renal failure—role of gadolinium-based contrast media in causation and the beneficial effect of intravenous sodium thiosulfate. Clin J Am Soc Nephrol 2007; 2:258–263.
- US Food and Drug Administration. Accessed 01/03/08. http://www.fda.gov/cder/drug/InfoSheets/HCP/gcca_200705.htm.
- Cowper SE, Robin HS, Steinberg SM, Su LD, Gupta S, LeBoit PE. Scleromyxoedema-like cutaneous diseases in renal-dialysis patients. Lancet 2000; 356:1000–1001.
- Galan A, Cowper SE, Bucala R. Nephrogenic systemic fibrosis (nephrogenic fibrosing dermopathy). Curr Opin Rheumatol 2006; 18:614–617.
- Cowper SE. Nephrogenic fibrosing dermopathy: the first 6 years. Curr Opin Rheumatol 2003; 15:785–790.
- Ting WW, Stone MS, Madison KC, Kurtz K. Nephrogenic fibrosing dermopathy with systemic involvement. Arch Dermatol 2003; 139:903–906.
- Kucher C, Steere J, Elenitsas R, Siegel DL, Xu X. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis with diaphragmatic involvement in a patient with respiratory failure. J Am Acad Dermatol 2006; 54:S31–S34.
- Jimenez SA, Artlett CM, Sandorfi N, et al. Dialysis-associated systemic fibrosis (nephrogenic fibrosing dermopathy): study of inflammatory cells and transforming growth factor beta1 expression in affected skin. Arthritis Rheum 2004; 50:2660–2666.
- Mendoza FA, Artlett CM, Sandorfi N, Latinis K, Piera-Velazquez S, Jimenez SA. Description of 12 cases of nephrogenic fibrosing dermopathy and review of the literature. Semin Arthritis Rheum 2006; 35:238–249.
- Todd DJ, Kagan A, Chibnik LB, Kay J. Cutaneous changes of nephrogenic systemic fibrosis: predictor of early mortality and association with gadolinium exposure. Arthritis Rheum 2007; 56:3433–3441.
- Cowper SE, Bucala R, Leboit PE. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis—setting the record straight. Semin Arthritis Rheum 2006; 35:208–210.
- Quan TE, Cowper S, Wu SP, Bockenstedt LK, Bucala R. Circulating fibrocytes: collagen-secreting cells of the peripheral blood. Int J Biochem Cell Biol 2004; 36:598–606.
- Grobner T. Gadolinium—a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis? Nephrol Dial Transplant 2006; 21:1104–1108.
- Marckmann P, Skov L, Rossen K, et al. Nephrogenic systemic fibrosis: suspected causative role of gadodiamide used for contrast-enhanced magnetic resonance imaging. J Am Soc Nephrol 2006; 17:2359–2362.
- Kuo PH, Kanal E, Abu-Alfa AK, Cowper SE. Gadolinium-based MR contrast agents and nephrogenic systemic fibrosis. Radiology 2007; 242:647–649.
- Broome DR, Girguis MS, Baron PW, Cottrell AC, Kjellin I, Kirk GA. Gadodiamide-associated nephrogenic systemic fibrosis: why radiologists should be concerned. AJR Am J Roentgenol 2007; 188:586–592.
- Khurana A, Runge VM, Narayanan M, Greene JF, Nickel AE. Nephrogenic systemic fibrosis: a review of 6 cases temporally related to gadodiamide injection (Omniscan). Invest Radiol 2007; 42:139–145.
- Deo A, Fogel M, Cowper SE. Nephrogenic systemic fibrosis: a population study examining the relationship of disease development to gadolinium exposure. Clin J Am Soc Nephrol 2007; 2:264–267.
- US Centers for Disease Control and Prevention (CDC). Nephrogenic fibrosing dermopathy associated with exposure to gadolinium-containing contrast agents—St. Louis, Missouri, 2002–2006. MMWR Morb Mortal Wkly Rep 2007; 56:137–141.
- Sadowski EA, Bennett LK, Chan MR, et al. Nephrogenic systemic fibrosis: risk factors and incidence estimation. Radiology 2007; 243:148–157.
- Marckmann P, Skov L, Rossen K, Heaf JG, Thomsen HS. Case-control study of gadodiamide-related nephrogenic systemic fibrosis. Nephrol Dial Transplant 2007 May 4; e-pub ahead of print.
- Swaminathan S, Ahmed I, McCarthy JT, et al. Nephrogenic fibrosing dermopathy and high-dose erythropoietin therapy. Ann Intern Med 2006; 145:234–235.
- Jain SM, Wesson S, Hassanein A, et al. Nephrogenic fibrosing dermopathy in pediatric patients. Pediatr Nephrol 2004; 19:467–470.
- Fazeli A, Lio PA, Liu V. Nephrogenic fibrosing dermopathy: are ACE inhibitors the missing link? (Letter). Arch Dermatol 2004; 140:1401.
- Bellin MF. MR contrast agents, the old and the new. Eur J Radiol 2006; 60:314–323.
- Joffe P, Thomsen HS, Meusel M. Pharmacokinetics of gadodiamide injection in patients with severe renal insufficiency and patients undergoing hemodialysis or continuous ambulatory peritoneal dialysis. Acad Radiol 1998; 5:491–502.
- Ueda J, Furukawa T, Higashino K, et al. Permeability of iodinated and MR contrast media through two types of hemodialysis membrane. Eur J Radiol 1999; 31:76–80.
- Boyd AS, Zic JA, Abraham JL. Gadolinium deposition in nephrogenic fibrosing dermopathy. J Am Acad Dermatol 2007; 56:27–30.
- High WA, Ayers RA, Chandler J, Zito G, Cowper SE. Gadolinium is detectable within the tissue of patients with nephrogenic systemic fibrosis. J Am Acad Dermatol 2007; 56:21–26.
- High WA, Ayers RA, Cowper SE. Gadolinium is quantifiable within the tissue of patients with nephrogenic systemic fibrosis, J Am Acad Dermatol 2007; 56:710–712.
- Perazella MA. Nephrogenic systemic fibrosis, kidney disease, and gadolinium: is there a link? Clin J Am Soc Nephrol 2007; 2:200–202.
- Cowper SE. Nephrogenic systemic fibrosis: The nosological and conceptual evolution of nephrogenic fibrosing dermopathy. Am J Kidney Dis 2005; 46:763–765.
- Jan F, Segal JM, Dyer J, LeBoit P, Siegfried E, Frieden IJ. Nephrogenic fibrosing dermopathy: two pediatric cases. J Pediatr 2003; 143:678–681.
- Yerram P, Saab G, Karuparthi PR, Hayden MR, Khanna R. Nephrogenic systemic fibrosis: a mysterious disease in patients with renal failure—role of gadolinium-based contrast media in causation and the beneficial effect of intravenous sodium thiosulfate. Clin J Am Soc Nephrol 2007; 2:258–263.
- US Food and Drug Administration. Accessed 01/03/08. http://www.fda.gov/cder/drug/InfoSheets/HCP/gcca_200705.htm.
KEY POINTS
- NSF seems to arise in roughly 3% of patients with renal insufficiency who receive gadolinium, although the data are somewhat sketchy and the true incidence might be higher if the NSF is specifically looked for.
- Manufacturers of all available gadolinium contrast agents now must include a boxed warning about the risk of NSF in patients with acute or chronic severe renal insufficiency (glomerular filtration rate < 30 mL/minute/1.73 m2) and in patients with acute renal insufficiency of any severity due to hepatorenal syndrome or in the perioperative liver transplantation period.
- As yet, we have no effective treatment for NSF. If the patient is already on hemodialysis, it may be reasonable to perform hemodialysis immediately after exposure to gadolinium and again the next day.
Do COX-2 inhibitors worsen renal function?
No, COX-2 inhibitors, as a class, do not worsen renal function for those without renal disease. Celecoxib is the only COX-2 inhibitor available, and it is associated with a lower risk of renal dysfunction and hypertension when compared with controls. Available data do not allow for adjusted risk assessment for patients with preexisting renal disease on COX-2 inhibitors (strength of recommendation [SOR]: A, based on meta-analysis).
Use celecoxib cautiously in patients at risk of serious complications
Vincent LO, MD
San Joaquin Family Medicine Residency, French Camp, Calif
Recent studies have raised concerns about the safety of this class of medication. For example, rofecoxib was linked with increased cardiovascular events, leading to it being pulled from the market.1 The claim of decreased gastrointestinal bleeding with long-term use of COX-2 inhibitors has also been questioned.2
Although this Clinical Inquiry concludes that celecoxib does not appear to worsen renal function, it should still be used with caution for patients who are elderly, hospitalized, or at risk of developing serious complications such as acute renal failure, heart failure, and gastrointestinal bleeding.
Evidence summary
A 2006 meta-analysis, including 114 trials and 116,094 patients randomized to either cyclooxygenase-2 (COX-2) inhibitor or control (placebo, nonsteroidal anti-inflammatory drug [NSAID], or mixed), indicated that the COX-2 inhibitors, as a class, had no effect on renal endpoints.3 Trials were reviewed for data on renal endpoints, including peripheral edema, hypertension, and renal dysfunction (defined as significant worsening of serum urea or creatinine, or clinical evidence of kidney disease and renal failure).
When viewed separately, rofecoxib (Vioxx) was associated with a composite relative risk (RR) of 1.53 (95% confidence interval [CI], 1.33–1.76) for all renal endpoints compared with controls. In contrast, the composite RR for the same endpoints among patients taking celecoxib (Celebrex) was 0.97 (95% CI, 0.84–1.12), indicating no effect on renal function. In fact, for the specific outcomes of hypertension and renal dysfunction, celecoxib was associated with a decreased risk compared with controls (TABLE).3
Stratified analysis by type of control (placebo, alternate NSAID, or mixed) yielded consistent results; rofecoxib was uniquely associated with adverse renal outcomes. No effect on renal function was noted for celecoxib compared with the same controls: the RR for adverse renal effects was 0.87 (95% CI, 0.55–1.38), 0.93 (95% CI, 0.70–1.23), and 1.26 (95% CI, 0.94–1.69) for celecoxib vs placebo, NSAID, and mixed controls, respectively. Statistical analysis for heterogeneity showed that the variation in effects on renal function among the COX-2 inhibitors was more likely due to actual differences than due to chance (heterogeneity [I2]=57%; P<.001).
Data were not available to assess the effect of COX-2 agents on patients with pre-existing renal disease, primarily because trials reporting abnormal renal function at baseline were excluded from this meta-analysis.
A recent randomized controlled trial compared standard dosing of diclofenac (75 mg twice daily) and ibuprofen (800 mg 3 times daily) with high-dose celecoxib (400 mg twice daily) for patients with normal kidney function being treated for osteoarthritis and rheumatoid arthritis.4 The mean increase in serum creatinine in the celecoxib arm was less than that noted in the diclofenac controls (0.009 mg/dL vs 0.027 mg/dL; P<.05; number needed to harm [NNH]=56). No difference in mean serum creatinine was seen among those patients using ibuprofen (800 mg 3 times daily) compared with those using high-dose celecoxib.
This evidence further supports the safety of celecoxib vs standard NSAIDs with respect to renal dysfunction.
Recommendations from others The American Pain Society 2002 guideline recommends acetaminophen for mild pain from osteoarthritis.5 For moderate to severe pain and inflammation, a COX-2 inhibitor was the first choice, unless there is significant risk of hypertension or kidney disorder. For active rheumatoid arthritis, the addition of a COX-2 agent to disease-modifying anti-rheumatic drugs (DMARDs) is advised unless there is uncontrolled hypertension or renal disease.6 However, these recommendations came out before the data on the cardiovascular effects of some COX-2 inhibitors.
The American College of rheumatology recommends the use of a COX-2 agent for osteoarthritis or pain unresponsive to acetaminophen. Their 2000 guidelines warn that due to potential renal toxicity, COX-2 inhibitors should not be used for patients with severe renal insufficiency, and used with caution in cases of mild to moderate renal insufficiency.
In 2005, these guidelines were amended to include the recommendation that patients with increased cardiovascular risk be cautioned about the risks associated with COX-2 inhibitor use.7
TABLE
Celecoxib is associated with a decreased risk of hypertension and renal dysfunction
| CELECOXIB | ROFECOXIB | |
|---|---|---|
| Hypertension | 0.83 (95% CI, 0.71–0.97) | 1.55 (95% CI, 1.29–1.85) |
| Peripheral edema | 1.09 (95% CI, 0.91–1.31) | 1.43 (95% CI, 1.23–1.66) |
| Renal dysfunction | 0.61 (95% CI, 0.40–0.94) | 2.31 (95% CI, 1.05–5.07) |
| Source: Zhang J, Ding EL, Song Y, JAMA 2006.3 | ||
1. Bresalier RS, Sandler RS, Quan H, et al. Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med 2005;352:1092-1102.
2. Hippisley-Cox J, Coupland C, Logan R. Risk of adverse gastrointestinal outcomes in patients taking cyclo-oxygenase-2 inhibitors or conventional non-steroidal anti-inflammatory drugs: population based nested case-control analysis. BMJ 2005;331:1310-1316.
3. Zhang J, Ding EL, Song Y. Adverse effects of cyclooxygenase 2 inhibitors on renal and arrhythmia events. JAMA 2006;296:1619-1632.
4. Whelton A, Lefkowith JL, West CR, Verburg KM. Cardiorenal effects of celecoxib as compared with the nonsteroidal anti-inflammatory drugs diclofenac and ibuprofen. Kidney Intl. 2006;70:1495-1502.
5. Simon LS, Lipman AG, Jacox AK, et al. Pain in Osteoarthritis, Rheumatoid Arthritis and Juvenile Chronic Arthritis. 2nd ed. Glenview, Ill: American Pain society; 2002.
6. American College of Rheumatology (ACR) sub-committee on Osteoarthritis Guidelines. recommendations for the medical management of osteoarthritis of the hip and knee: 2000 update. American College of Rheumatology Subcommittee on osteoarthritis Guidelines. Arthritis Rheum. 2000;43:1905-1915.
7. American College of Rheumatology Subcommittee on rheumatoid Arthritis Guidelines. Guidelines for the management of rheumatoid arthritis: 2002 update. Arthritis Rheum. 2002;46:328-346.
No, COX-2 inhibitors, as a class, do not worsen renal function for those without renal disease. Celecoxib is the only COX-2 inhibitor available, and it is associated with a lower risk of renal dysfunction and hypertension when compared with controls. Available data do not allow for adjusted risk assessment for patients with preexisting renal disease on COX-2 inhibitors (strength of recommendation [SOR]: A, based on meta-analysis).
Use celecoxib cautiously in patients at risk of serious complications
Vincent LO, MD
San Joaquin Family Medicine Residency, French Camp, Calif
Recent studies have raised concerns about the safety of this class of medication. For example, rofecoxib was linked with increased cardiovascular events, leading to it being pulled from the market.1 The claim of decreased gastrointestinal bleeding with long-term use of COX-2 inhibitors has also been questioned.2
Although this Clinical Inquiry concludes that celecoxib does not appear to worsen renal function, it should still be used with caution for patients who are elderly, hospitalized, or at risk of developing serious complications such as acute renal failure, heart failure, and gastrointestinal bleeding.
Evidence summary
A 2006 meta-analysis, including 114 trials and 116,094 patients randomized to either cyclooxygenase-2 (COX-2) inhibitor or control (placebo, nonsteroidal anti-inflammatory drug [NSAID], or mixed), indicated that the COX-2 inhibitors, as a class, had no effect on renal endpoints.3 Trials were reviewed for data on renal endpoints, including peripheral edema, hypertension, and renal dysfunction (defined as significant worsening of serum urea or creatinine, or clinical evidence of kidney disease and renal failure).
When viewed separately, rofecoxib (Vioxx) was associated with a composite relative risk (RR) of 1.53 (95% confidence interval [CI], 1.33–1.76) for all renal endpoints compared with controls. In contrast, the composite RR for the same endpoints among patients taking celecoxib (Celebrex) was 0.97 (95% CI, 0.84–1.12), indicating no effect on renal function. In fact, for the specific outcomes of hypertension and renal dysfunction, celecoxib was associated with a decreased risk compared with controls (TABLE).3
Stratified analysis by type of control (placebo, alternate NSAID, or mixed) yielded consistent results; rofecoxib was uniquely associated with adverse renal outcomes. No effect on renal function was noted for celecoxib compared with the same controls: the RR for adverse renal effects was 0.87 (95% CI, 0.55–1.38), 0.93 (95% CI, 0.70–1.23), and 1.26 (95% CI, 0.94–1.69) for celecoxib vs placebo, NSAID, and mixed controls, respectively. Statistical analysis for heterogeneity showed that the variation in effects on renal function among the COX-2 inhibitors was more likely due to actual differences than due to chance (heterogeneity [I2]=57%; P<.001).
Data were not available to assess the effect of COX-2 agents on patients with pre-existing renal disease, primarily because trials reporting abnormal renal function at baseline were excluded from this meta-analysis.
A recent randomized controlled trial compared standard dosing of diclofenac (75 mg twice daily) and ibuprofen (800 mg 3 times daily) with high-dose celecoxib (400 mg twice daily) for patients with normal kidney function being treated for osteoarthritis and rheumatoid arthritis.4 The mean increase in serum creatinine in the celecoxib arm was less than that noted in the diclofenac controls (0.009 mg/dL vs 0.027 mg/dL; P<.05; number needed to harm [NNH]=56). No difference in mean serum creatinine was seen among those patients using ibuprofen (800 mg 3 times daily) compared with those using high-dose celecoxib.
This evidence further supports the safety of celecoxib vs standard NSAIDs with respect to renal dysfunction.
Recommendations from others The American Pain Society 2002 guideline recommends acetaminophen for mild pain from osteoarthritis.5 For moderate to severe pain and inflammation, a COX-2 inhibitor was the first choice, unless there is significant risk of hypertension or kidney disorder. For active rheumatoid arthritis, the addition of a COX-2 agent to disease-modifying anti-rheumatic drugs (DMARDs) is advised unless there is uncontrolled hypertension or renal disease.6 However, these recommendations came out before the data on the cardiovascular effects of some COX-2 inhibitors.
The American College of rheumatology recommends the use of a COX-2 agent for osteoarthritis or pain unresponsive to acetaminophen. Their 2000 guidelines warn that due to potential renal toxicity, COX-2 inhibitors should not be used for patients with severe renal insufficiency, and used with caution in cases of mild to moderate renal insufficiency.
In 2005, these guidelines were amended to include the recommendation that patients with increased cardiovascular risk be cautioned about the risks associated with COX-2 inhibitor use.7
TABLE
Celecoxib is associated with a decreased risk of hypertension and renal dysfunction
| CELECOXIB | ROFECOXIB | |
|---|---|---|
| Hypertension | 0.83 (95% CI, 0.71–0.97) | 1.55 (95% CI, 1.29–1.85) |
| Peripheral edema | 1.09 (95% CI, 0.91–1.31) | 1.43 (95% CI, 1.23–1.66) |
| Renal dysfunction | 0.61 (95% CI, 0.40–0.94) | 2.31 (95% CI, 1.05–5.07) |
| Source: Zhang J, Ding EL, Song Y, JAMA 2006.3 | ||
No, COX-2 inhibitors, as a class, do not worsen renal function for those without renal disease. Celecoxib is the only COX-2 inhibitor available, and it is associated with a lower risk of renal dysfunction and hypertension when compared with controls. Available data do not allow for adjusted risk assessment for patients with preexisting renal disease on COX-2 inhibitors (strength of recommendation [SOR]: A, based on meta-analysis).
Use celecoxib cautiously in patients at risk of serious complications
Vincent LO, MD
San Joaquin Family Medicine Residency, French Camp, Calif
Recent studies have raised concerns about the safety of this class of medication. For example, rofecoxib was linked with increased cardiovascular events, leading to it being pulled from the market.1 The claim of decreased gastrointestinal bleeding with long-term use of COX-2 inhibitors has also been questioned.2
Although this Clinical Inquiry concludes that celecoxib does not appear to worsen renal function, it should still be used with caution for patients who are elderly, hospitalized, or at risk of developing serious complications such as acute renal failure, heart failure, and gastrointestinal bleeding.
Evidence summary
A 2006 meta-analysis, including 114 trials and 116,094 patients randomized to either cyclooxygenase-2 (COX-2) inhibitor or control (placebo, nonsteroidal anti-inflammatory drug [NSAID], or mixed), indicated that the COX-2 inhibitors, as a class, had no effect on renal endpoints.3 Trials were reviewed for data on renal endpoints, including peripheral edema, hypertension, and renal dysfunction (defined as significant worsening of serum urea or creatinine, or clinical evidence of kidney disease and renal failure).
When viewed separately, rofecoxib (Vioxx) was associated with a composite relative risk (RR) of 1.53 (95% confidence interval [CI], 1.33–1.76) for all renal endpoints compared with controls. In contrast, the composite RR for the same endpoints among patients taking celecoxib (Celebrex) was 0.97 (95% CI, 0.84–1.12), indicating no effect on renal function. In fact, for the specific outcomes of hypertension and renal dysfunction, celecoxib was associated with a decreased risk compared with controls (TABLE).3
Stratified analysis by type of control (placebo, alternate NSAID, or mixed) yielded consistent results; rofecoxib was uniquely associated with adverse renal outcomes. No effect on renal function was noted for celecoxib compared with the same controls: the RR for adverse renal effects was 0.87 (95% CI, 0.55–1.38), 0.93 (95% CI, 0.70–1.23), and 1.26 (95% CI, 0.94–1.69) for celecoxib vs placebo, NSAID, and mixed controls, respectively. Statistical analysis for heterogeneity showed that the variation in effects on renal function among the COX-2 inhibitors was more likely due to actual differences than due to chance (heterogeneity [I2]=57%; P<.001).
Data were not available to assess the effect of COX-2 agents on patients with pre-existing renal disease, primarily because trials reporting abnormal renal function at baseline were excluded from this meta-analysis.
A recent randomized controlled trial compared standard dosing of diclofenac (75 mg twice daily) and ibuprofen (800 mg 3 times daily) with high-dose celecoxib (400 mg twice daily) for patients with normal kidney function being treated for osteoarthritis and rheumatoid arthritis.4 The mean increase in serum creatinine in the celecoxib arm was less than that noted in the diclofenac controls (0.009 mg/dL vs 0.027 mg/dL; P<.05; number needed to harm [NNH]=56). No difference in mean serum creatinine was seen among those patients using ibuprofen (800 mg 3 times daily) compared with those using high-dose celecoxib.
This evidence further supports the safety of celecoxib vs standard NSAIDs with respect to renal dysfunction.
Recommendations from others The American Pain Society 2002 guideline recommends acetaminophen for mild pain from osteoarthritis.5 For moderate to severe pain and inflammation, a COX-2 inhibitor was the first choice, unless there is significant risk of hypertension or kidney disorder. For active rheumatoid arthritis, the addition of a COX-2 agent to disease-modifying anti-rheumatic drugs (DMARDs) is advised unless there is uncontrolled hypertension or renal disease.6 However, these recommendations came out before the data on the cardiovascular effects of some COX-2 inhibitors.
The American College of rheumatology recommends the use of a COX-2 agent for osteoarthritis or pain unresponsive to acetaminophen. Their 2000 guidelines warn that due to potential renal toxicity, COX-2 inhibitors should not be used for patients with severe renal insufficiency, and used with caution in cases of mild to moderate renal insufficiency.
In 2005, these guidelines were amended to include the recommendation that patients with increased cardiovascular risk be cautioned about the risks associated with COX-2 inhibitor use.7
TABLE
Celecoxib is associated with a decreased risk of hypertension and renal dysfunction
| CELECOXIB | ROFECOXIB | |
|---|---|---|
| Hypertension | 0.83 (95% CI, 0.71–0.97) | 1.55 (95% CI, 1.29–1.85) |
| Peripheral edema | 1.09 (95% CI, 0.91–1.31) | 1.43 (95% CI, 1.23–1.66) |
| Renal dysfunction | 0.61 (95% CI, 0.40–0.94) | 2.31 (95% CI, 1.05–5.07) |
| Source: Zhang J, Ding EL, Song Y, JAMA 2006.3 | ||
1. Bresalier RS, Sandler RS, Quan H, et al. Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med 2005;352:1092-1102.
2. Hippisley-Cox J, Coupland C, Logan R. Risk of adverse gastrointestinal outcomes in patients taking cyclo-oxygenase-2 inhibitors or conventional non-steroidal anti-inflammatory drugs: population based nested case-control analysis. BMJ 2005;331:1310-1316.
3. Zhang J, Ding EL, Song Y. Adverse effects of cyclooxygenase 2 inhibitors on renal and arrhythmia events. JAMA 2006;296:1619-1632.
4. Whelton A, Lefkowith JL, West CR, Verburg KM. Cardiorenal effects of celecoxib as compared with the nonsteroidal anti-inflammatory drugs diclofenac and ibuprofen. Kidney Intl. 2006;70:1495-1502.
5. Simon LS, Lipman AG, Jacox AK, et al. Pain in Osteoarthritis, Rheumatoid Arthritis and Juvenile Chronic Arthritis. 2nd ed. Glenview, Ill: American Pain society; 2002.
6. American College of Rheumatology (ACR) sub-committee on Osteoarthritis Guidelines. recommendations for the medical management of osteoarthritis of the hip and knee: 2000 update. American College of Rheumatology Subcommittee on osteoarthritis Guidelines. Arthritis Rheum. 2000;43:1905-1915.
7. American College of Rheumatology Subcommittee on rheumatoid Arthritis Guidelines. Guidelines for the management of rheumatoid arthritis: 2002 update. Arthritis Rheum. 2002;46:328-346.
1. Bresalier RS, Sandler RS, Quan H, et al. Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. N Engl J Med 2005;352:1092-1102.
2. Hippisley-Cox J, Coupland C, Logan R. Risk of adverse gastrointestinal outcomes in patients taking cyclo-oxygenase-2 inhibitors or conventional non-steroidal anti-inflammatory drugs: population based nested case-control analysis. BMJ 2005;331:1310-1316.
3. Zhang J, Ding EL, Song Y. Adverse effects of cyclooxygenase 2 inhibitors on renal and arrhythmia events. JAMA 2006;296:1619-1632.
4. Whelton A, Lefkowith JL, West CR, Verburg KM. Cardiorenal effects of celecoxib as compared with the nonsteroidal anti-inflammatory drugs diclofenac and ibuprofen. Kidney Intl. 2006;70:1495-1502.
5. Simon LS, Lipman AG, Jacox AK, et al. Pain in Osteoarthritis, Rheumatoid Arthritis and Juvenile Chronic Arthritis. 2nd ed. Glenview, Ill: American Pain society; 2002.
6. American College of Rheumatology (ACR) sub-committee on Osteoarthritis Guidelines. recommendations for the medical management of osteoarthritis of the hip and knee: 2000 update. American College of Rheumatology Subcommittee on osteoarthritis Guidelines. Arthritis Rheum. 2000;43:1905-1915.
7. American College of Rheumatology Subcommittee on rheumatoid Arthritis Guidelines. Guidelines for the management of rheumatoid arthritis: 2002 update. Arthritis Rheum. 2002;46:328-346.
Evidence-based answers from the Family Physicians Inquiries Network
What is adequate hypertension control? Having your dinner and dessert too
Medical Urology for the Primary Care Provider
Supplement Editors:
Matt T. Rosenberg, MD, and Milton M. Lakin, MD
Contents
The recognition of urologic disease is a primary care issue, whether we've know it or not
Matt T. Rosenberg, MD, and Milton M. Lakin, MD
Screening for urologic malignancies in primary care: Pros, cons, and recommendations
Andrew J. Stephenson, MD; Louis Kuritzky, MD; and Steven C. Campbell, MD, PhD
Benign prostatic hyperplasia: When to 'watch and wait,' when and how to treat
Albert Levy, MD, and George P. Samraj, MD
Overactive bladder: Recognition requires vigilance for symptoms
Matt T. Rosenberg, MD; Diane K. Newman, RNC, MSN, CRNP; Christopher T. Tallman, BS; and Shari A. Page, CFNP
Erectile dysfunction: A sentinel marker for cardiovascular disease in primary care
Martin M. Miner, MD, and Louis Kuritzky, MD
Evolving issues in male hypogonadism: Evaluation, management, and related cormorbidities
Martin M. Miner, MD, and Richard Sadovsky, MD
Identifying and treating premature ejaculation: Importance of the sexual history
Richard E. Payne, MD, and Richard Sadovsky, MD
Interstitial cystitis/painful bladder syndrome: Symptom recognition is key to early identification, treatment
Matt T. Rosenberg, MD; Diane K. Newman, RNC, MSN, CRNP; and Shari A. Page CFNP
Prostatitis: Infection, neuromuscular disorder, or pain syndrome? Proper patient classification is key
Jeannette Potts, MD, and Richard E. Payne, MD
Supplement Editors:
Matt T. Rosenberg, MD, and Milton M. Lakin, MD
Contents
The recognition of urologic disease is a primary care issue, whether we've know it or not
Matt T. Rosenberg, MD, and Milton M. Lakin, MD
Screening for urologic malignancies in primary care: Pros, cons, and recommendations
Andrew J. Stephenson, MD; Louis Kuritzky, MD; and Steven C. Campbell, MD, PhD
Benign prostatic hyperplasia: When to 'watch and wait,' when and how to treat
Albert Levy, MD, and George P. Samraj, MD
Overactive bladder: Recognition requires vigilance for symptoms
Matt T. Rosenberg, MD; Diane K. Newman, RNC, MSN, CRNP; Christopher T. Tallman, BS; and Shari A. Page, CFNP
Erectile dysfunction: A sentinel marker for cardiovascular disease in primary care
Martin M. Miner, MD, and Louis Kuritzky, MD
Evolving issues in male hypogonadism: Evaluation, management, and related cormorbidities
Martin M. Miner, MD, and Richard Sadovsky, MD
Identifying and treating premature ejaculation: Importance of the sexual history
Richard E. Payne, MD, and Richard Sadovsky, MD
Interstitial cystitis/painful bladder syndrome: Symptom recognition is key to early identification, treatment
Matt T. Rosenberg, MD; Diane K. Newman, RNC, MSN, CRNP; and Shari A. Page CFNP
Prostatitis: Infection, neuromuscular disorder, or pain syndrome? Proper patient classification is key
Jeannette Potts, MD, and Richard E. Payne, MD
Supplement Editors:
Matt T. Rosenberg, MD, and Milton M. Lakin, MD
Contents
The recognition of urologic disease is a primary care issue, whether we've know it or not
Matt T. Rosenberg, MD, and Milton M. Lakin, MD
Screening for urologic malignancies in primary care: Pros, cons, and recommendations
Andrew J. Stephenson, MD; Louis Kuritzky, MD; and Steven C. Campbell, MD, PhD
Benign prostatic hyperplasia: When to 'watch and wait,' when and how to treat
Albert Levy, MD, and George P. Samraj, MD
Overactive bladder: Recognition requires vigilance for symptoms
Matt T. Rosenberg, MD; Diane K. Newman, RNC, MSN, CRNP; Christopher T. Tallman, BS; and Shari A. Page, CFNP
Erectile dysfunction: A sentinel marker for cardiovascular disease in primary care
Martin M. Miner, MD, and Louis Kuritzky, MD
Evolving issues in male hypogonadism: Evaluation, management, and related cormorbidities
Martin M. Miner, MD, and Richard Sadovsky, MD
Identifying and treating premature ejaculation: Importance of the sexual history
Richard E. Payne, MD, and Richard Sadovsky, MD
Interstitial cystitis/painful bladder syndrome: Symptom recognition is key to early identification, treatment
Matt T. Rosenberg, MD; Diane K. Newman, RNC, MSN, CRNP; and Shari A. Page CFNP
Prostatitis: Infection, neuromuscular disorder, or pain syndrome? Proper patient classification is key
Jeannette Potts, MD, and Richard E. Payne, MD