User login
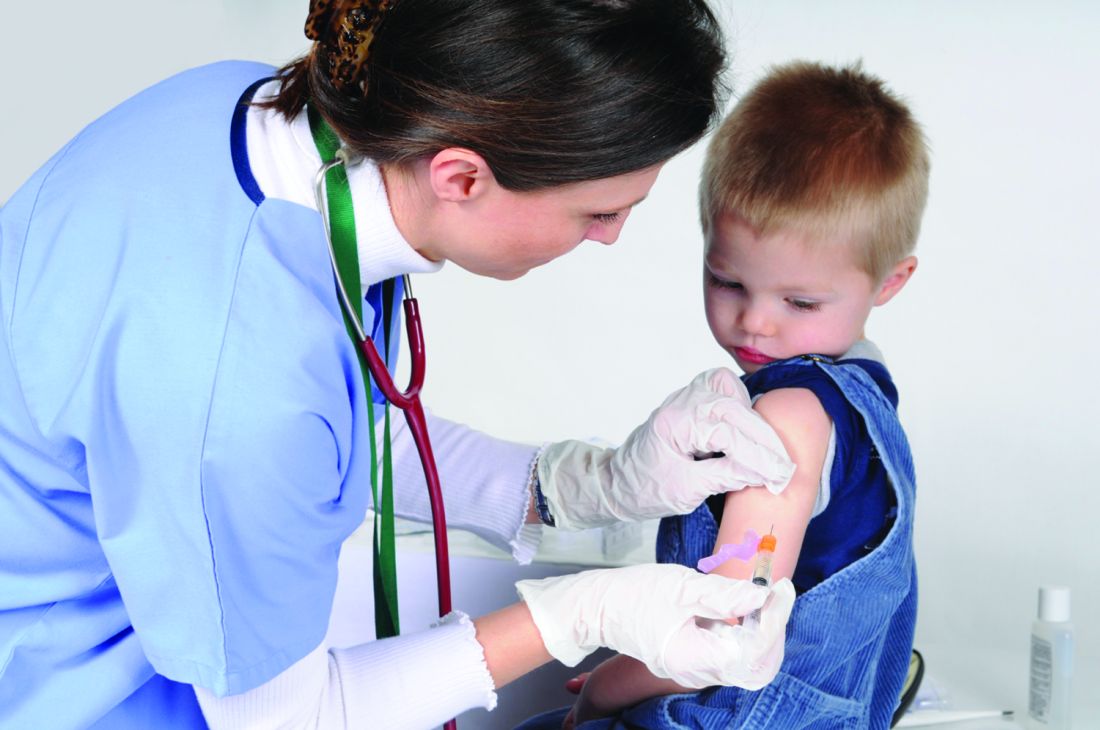
C-reactive protein testing reduced antibiotic prescribing in patients with COPD exacerbation
, according to a recent randomized, controlled trial.
Point-of-care C-reactive protein (CRP) testing led to fewer antibiotic prescriptions at the initial consultation, according to investigators participating in the PACE study, a multicenter, open-label trial of more than 600 patients with COPD enrolled at one of 86 general practices in the United Kingdom.
Patient-reported antibiotic use over the next 4 weeks was more than 20 percentage points lower for the group managed with the point-of-care strategy, compared with those who received usual care, according to the investigators, led by Christopher C. Butler, FMedSci, of the Nuffield Department of Primary Care Health Sciences at the University of Oxford (England).
Less antibiotic use and fewer prescriptions did not compromise patient-reported, disease-specific quality of life, added Dr. Butler and colleagues. Their report appears in the New England Journal of Medicine.
In the United States and in Europe, more than 80% of COPD patients with acute exacerbations will receive an antibiotic prescription, according to Dr. Butler and coauthors.
“Although many patients who have acute exacerbations of COPD are helped by these treatments, others are not,” wrote the investigators, noting that in one hospital-based study, about one in five such exacerbations were thought to be due to noninfectious causes.
The present study included patients at least 40 years of age who presented to a primary care practice with an acute exacerbation and at least one of the three Anthonisen criteria (increased dyspnea, sputum production, and sputum purulence) intended to guide antibiotic therapy in COPD. A total of 325 were randomly assigned to the CRP testing group, and 324 to a group that received just usual care.
Antibiotic use was reported by fewer patients in the CRP testing group, compared with the usual-care group (57.0% vs. 77.4%; adjusted odds ratio, 0.31, 95% confidence interval, 0.20-0.47), the investigators reported.
Only 47.7% of patients in the CRP-guided group received antibiotic prescriptions at the initial consultation, vs. 69.7% of patients in the usual care group.
Hospitalizations over 6 months of follow-up were reported for 8.6% and 9.3% of patients in the CRP-guided and usual-care groups, respectively, while diagnoses of pneumonia were recorded for 3.0% and 4.0%. There was no clinically important difference between groups in the rate of antibiotic-related adverse effects.
“The evidence from our trial suggests that CRP-guided antibiotic prescribing for COPD exacerbations in primary care clinics may reduce patient-reported use of antibiotics and the prescribing of antibiotics by clinicians,” Dr. Butler and colleagues said in a discussion of these results.
Findings from the study by Dr. Butler and colleagues are “compelling enough” to support C-reactive protein (CRP) testing to guide antibiotic use in patient who have acute exacerbations of COPD, wrote the authors of an accompanying editorial.
“The trial achieved its objective, which was to show that CRP testing safely reduces antibiotic use,” stated Allan S. Brett, MD, and Majdi N. Al-Hasan, MB,BS, of the department of medicine at the University of South Carolina, Columbia.
Point-of-care testing of CRP could be applied even more broadly in clinical practice, Dr. Brett and Dr. Al-Hasan wrote, since testing has been shown to reduce prescribing of antibiotics for suspected lower respiratory tract infections and other common presentations in patients with no COPD.
“Whether primary care practices in the United States would embrace point-of-care CRP testing is another matter, given the regulatory requirements for in-office laboratory testing and uncertainty about reimbursement,” they noted.
Reduced antibiotic prescribing in patients with COPD likely has certain benefits, including reducing risk of Clostridioides difficile colitis, according to the authors.
By contrast, the current study did not determine which COPD patients might benefit from antibiotics, if any, nor which antibiotic might be warranted for those patients.
The study was supported by the Health Technology Assessment Program of the UK National Institute for Health Research. Dr. Butler reported disclosures related to Roche Molecular Systems and Roche Molecular Diagnostics, among others.
SOURCE: Butler CC et al. N Engl J Med. 2019 Jul 10;381:111-20. doi: 10.1056/NEJMoa1803185.
, according to a recent randomized, controlled trial.
Point-of-care C-reactive protein (CRP) testing led to fewer antibiotic prescriptions at the initial consultation, according to investigators participating in the PACE study, a multicenter, open-label trial of more than 600 patients with COPD enrolled at one of 86 general practices in the United Kingdom.
Patient-reported antibiotic use over the next 4 weeks was more than 20 percentage points lower for the group managed with the point-of-care strategy, compared with those who received usual care, according to the investigators, led by Christopher C. Butler, FMedSci, of the Nuffield Department of Primary Care Health Sciences at the University of Oxford (England).
Less antibiotic use and fewer prescriptions did not compromise patient-reported, disease-specific quality of life, added Dr. Butler and colleagues. Their report appears in the New England Journal of Medicine.
In the United States and in Europe, more than 80% of COPD patients with acute exacerbations will receive an antibiotic prescription, according to Dr. Butler and coauthors.
“Although many patients who have acute exacerbations of COPD are helped by these treatments, others are not,” wrote the investigators, noting that in one hospital-based study, about one in five such exacerbations were thought to be due to noninfectious causes.
The present study included patients at least 40 years of age who presented to a primary care practice with an acute exacerbation and at least one of the three Anthonisen criteria (increased dyspnea, sputum production, and sputum purulence) intended to guide antibiotic therapy in COPD. A total of 325 were randomly assigned to the CRP testing group, and 324 to a group that received just usual care.
Antibiotic use was reported by fewer patients in the CRP testing group, compared with the usual-care group (57.0% vs. 77.4%; adjusted odds ratio, 0.31, 95% confidence interval, 0.20-0.47), the investigators reported.
Only 47.7% of patients in the CRP-guided group received antibiotic prescriptions at the initial consultation, vs. 69.7% of patients in the usual care group.
Hospitalizations over 6 months of follow-up were reported for 8.6% and 9.3% of patients in the CRP-guided and usual-care groups, respectively, while diagnoses of pneumonia were recorded for 3.0% and 4.0%. There was no clinically important difference between groups in the rate of antibiotic-related adverse effects.
“The evidence from our trial suggests that CRP-guided antibiotic prescribing for COPD exacerbations in primary care clinics may reduce patient-reported use of antibiotics and the prescribing of antibiotics by clinicians,” Dr. Butler and colleagues said in a discussion of these results.
Findings from the study by Dr. Butler and colleagues are “compelling enough” to support C-reactive protein (CRP) testing to guide antibiotic use in patient who have acute exacerbations of COPD, wrote the authors of an accompanying editorial.
“The trial achieved its objective, which was to show that CRP testing safely reduces antibiotic use,” stated Allan S. Brett, MD, and Majdi N. Al-Hasan, MB,BS, of the department of medicine at the University of South Carolina, Columbia.
Point-of-care testing of CRP could be applied even more broadly in clinical practice, Dr. Brett and Dr. Al-Hasan wrote, since testing has been shown to reduce prescribing of antibiotics for suspected lower respiratory tract infections and other common presentations in patients with no COPD.
“Whether primary care practices in the United States would embrace point-of-care CRP testing is another matter, given the regulatory requirements for in-office laboratory testing and uncertainty about reimbursement,” they noted.
Reduced antibiotic prescribing in patients with COPD likely has certain benefits, including reducing risk of Clostridioides difficile colitis, according to the authors.
By contrast, the current study did not determine which COPD patients might benefit from antibiotics, if any, nor which antibiotic might be warranted for those patients.
The study was supported by the Health Technology Assessment Program of the UK National Institute for Health Research. Dr. Butler reported disclosures related to Roche Molecular Systems and Roche Molecular Diagnostics, among others.
SOURCE: Butler CC et al. N Engl J Med. 2019 Jul 10;381:111-20. doi: 10.1056/NEJMoa1803185.
, according to a recent randomized, controlled trial.
Point-of-care C-reactive protein (CRP) testing led to fewer antibiotic prescriptions at the initial consultation, according to investigators participating in the PACE study, a multicenter, open-label trial of more than 600 patients with COPD enrolled at one of 86 general practices in the United Kingdom.
Patient-reported antibiotic use over the next 4 weeks was more than 20 percentage points lower for the group managed with the point-of-care strategy, compared with those who received usual care, according to the investigators, led by Christopher C. Butler, FMedSci, of the Nuffield Department of Primary Care Health Sciences at the University of Oxford (England).
Less antibiotic use and fewer prescriptions did not compromise patient-reported, disease-specific quality of life, added Dr. Butler and colleagues. Their report appears in the New England Journal of Medicine.
In the United States and in Europe, more than 80% of COPD patients with acute exacerbations will receive an antibiotic prescription, according to Dr. Butler and coauthors.
“Although many patients who have acute exacerbations of COPD are helped by these treatments, others are not,” wrote the investigators, noting that in one hospital-based study, about one in five such exacerbations were thought to be due to noninfectious causes.
The present study included patients at least 40 years of age who presented to a primary care practice with an acute exacerbation and at least one of the three Anthonisen criteria (increased dyspnea, sputum production, and sputum purulence) intended to guide antibiotic therapy in COPD. A total of 325 were randomly assigned to the CRP testing group, and 324 to a group that received just usual care.
Antibiotic use was reported by fewer patients in the CRP testing group, compared with the usual-care group (57.0% vs. 77.4%; adjusted odds ratio, 0.31, 95% confidence interval, 0.20-0.47), the investigators reported.
Only 47.7% of patients in the CRP-guided group received antibiotic prescriptions at the initial consultation, vs. 69.7% of patients in the usual care group.
Hospitalizations over 6 months of follow-up were reported for 8.6% and 9.3% of patients in the CRP-guided and usual-care groups, respectively, while diagnoses of pneumonia were recorded for 3.0% and 4.0%. There was no clinically important difference between groups in the rate of antibiotic-related adverse effects.
“The evidence from our trial suggests that CRP-guided antibiotic prescribing for COPD exacerbations in primary care clinics may reduce patient-reported use of antibiotics and the prescribing of antibiotics by clinicians,” Dr. Butler and colleagues said in a discussion of these results.
Findings from the study by Dr. Butler and colleagues are “compelling enough” to support C-reactive protein (CRP) testing to guide antibiotic use in patient who have acute exacerbations of COPD, wrote the authors of an accompanying editorial.
“The trial achieved its objective, which was to show that CRP testing safely reduces antibiotic use,” stated Allan S. Brett, MD, and Majdi N. Al-Hasan, MB,BS, of the department of medicine at the University of South Carolina, Columbia.
Point-of-care testing of CRP could be applied even more broadly in clinical practice, Dr. Brett and Dr. Al-Hasan wrote, since testing has been shown to reduce prescribing of antibiotics for suspected lower respiratory tract infections and other common presentations in patients with no COPD.
“Whether primary care practices in the United States would embrace point-of-care CRP testing is another matter, given the regulatory requirements for in-office laboratory testing and uncertainty about reimbursement,” they noted.
Reduced antibiotic prescribing in patients with COPD likely has certain benefits, including reducing risk of Clostridioides difficile colitis, according to the authors.
By contrast, the current study did not determine which COPD patients might benefit from antibiotics, if any, nor which antibiotic might be warranted for those patients.
The study was supported by the Health Technology Assessment Program of the UK National Institute for Health Research. Dr. Butler reported disclosures related to Roche Molecular Systems and Roche Molecular Diagnostics, among others.
SOURCE: Butler CC et al. N Engl J Med. 2019 Jul 10;381:111-20. doi: 10.1056/NEJMoa1803185.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Parent education improves pediatric influenza vaccination rates
according to a randomized clinical trial published in Pediatrics.
Vanessa P. Scott, MD, MS, of Columbia University, New York, and colleagues randomized 400 parent-child dyads into any of three arms: receiving a handout based on national data, receiving a handout based on local data, or receiving usual care. This convenience sample was drawn from two pediatric clinics in New York between August 2016 and March 2017.
After adjustment for parents’ education level, the trial found that parents who received either handout were significantly more likely than were those receiving usual care to vaccinate their children by the end of season (75% and 65%, respectively; adjusted odds ratio, 1.68; 95% confidence interval, 1.06-2.67), but the effects of any intervention versus those of usual care on vaccination on day of visit were not statistically significant (59% vs. 53%; aOR, 1.36; 95% CI, 0.89-2.09).The researchers had hoped that using a targeted approach based on local data would increase vaccine receipt, but that was not seen in the results.
They did find that, across all three arms in the trial, baseline parental intent to vaccinate (likely versus unlikely) was associated with vaccination rates: Both vaccination on clinic visit day (70% vs. 22%; aOR, 8.38; 95% CI, 4.85-14.34) and vaccination by end of season (87% vs. 29%; aOR, 18.26; 95% CI, 9.94-33.52) were affected.
Strengths of the study included the randomized, controlled design and assessment of baseline factors, such as intention to vaccinate, to reduce confounding effects. Limitations included use of a convenience sample, which could have introduced selection bias.
One author was an unremunerated coinvestigator of an unrelated trial that received an investigator-initiated grant from the Pfizer Medical Education Group. Two authors were funded by other grants, but no potential conflicts of interests to disclose were indicated by any of the authors in this study.
SOURCE: Scott VP et al. Pediatrics. 2019. doi: 10.1542/peds.2018-2580.
according to a randomized clinical trial published in Pediatrics.
Vanessa P. Scott, MD, MS, of Columbia University, New York, and colleagues randomized 400 parent-child dyads into any of three arms: receiving a handout based on national data, receiving a handout based on local data, or receiving usual care. This convenience sample was drawn from two pediatric clinics in New York between August 2016 and March 2017.
After adjustment for parents’ education level, the trial found that parents who received either handout were significantly more likely than were those receiving usual care to vaccinate their children by the end of season (75% and 65%, respectively; adjusted odds ratio, 1.68; 95% confidence interval, 1.06-2.67), but the effects of any intervention versus those of usual care on vaccination on day of visit were not statistically significant (59% vs. 53%; aOR, 1.36; 95% CI, 0.89-2.09).The researchers had hoped that using a targeted approach based on local data would increase vaccine receipt, but that was not seen in the results.
They did find that, across all three arms in the trial, baseline parental intent to vaccinate (likely versus unlikely) was associated with vaccination rates: Both vaccination on clinic visit day (70% vs. 22%; aOR, 8.38; 95% CI, 4.85-14.34) and vaccination by end of season (87% vs. 29%; aOR, 18.26; 95% CI, 9.94-33.52) were affected.
Strengths of the study included the randomized, controlled design and assessment of baseline factors, such as intention to vaccinate, to reduce confounding effects. Limitations included use of a convenience sample, which could have introduced selection bias.
One author was an unremunerated coinvestigator of an unrelated trial that received an investigator-initiated grant from the Pfizer Medical Education Group. Two authors were funded by other grants, but no potential conflicts of interests to disclose were indicated by any of the authors in this study.
SOURCE: Scott VP et al. Pediatrics. 2019. doi: 10.1542/peds.2018-2580.
according to a randomized clinical trial published in Pediatrics.
Vanessa P. Scott, MD, MS, of Columbia University, New York, and colleagues randomized 400 parent-child dyads into any of three arms: receiving a handout based on national data, receiving a handout based on local data, or receiving usual care. This convenience sample was drawn from two pediatric clinics in New York between August 2016 and March 2017.
After adjustment for parents’ education level, the trial found that parents who received either handout were significantly more likely than were those receiving usual care to vaccinate their children by the end of season (75% and 65%, respectively; adjusted odds ratio, 1.68; 95% confidence interval, 1.06-2.67), but the effects of any intervention versus those of usual care on vaccination on day of visit were not statistically significant (59% vs. 53%; aOR, 1.36; 95% CI, 0.89-2.09).The researchers had hoped that using a targeted approach based on local data would increase vaccine receipt, but that was not seen in the results.
They did find that, across all three arms in the trial, baseline parental intent to vaccinate (likely versus unlikely) was associated with vaccination rates: Both vaccination on clinic visit day (70% vs. 22%; aOR, 8.38; 95% CI, 4.85-14.34) and vaccination by end of season (87% vs. 29%; aOR, 18.26; 95% CI, 9.94-33.52) were affected.
Strengths of the study included the randomized, controlled design and assessment of baseline factors, such as intention to vaccinate, to reduce confounding effects. Limitations included use of a convenience sample, which could have introduced selection bias.
One author was an unremunerated coinvestigator of an unrelated trial that received an investigator-initiated grant from the Pfizer Medical Education Group. Two authors were funded by other grants, but no potential conflicts of interests to disclose were indicated by any of the authors in this study.
SOURCE: Scott VP et al. Pediatrics. 2019. doi: 10.1542/peds.2018-2580.
FROM PEDIATRICS
No reduction in PE risk with vena cava filters after severe injury
MELBOURNE – Use of a prophylactic vena cava filter to trap blood clots in severely injured patients does not appear to reduce the risk of pulmonary embolism or death, according to data presented at the International Society on Thrombosis and Haemostasis congress.
The researchers reported the outcomes of a multicenter, controlled trial in which 240 severely injured patients with a contraindication to anticoagulants were randomized to receive a vena cava filter within 72 hours of admission, or no filter. The findings were published simultaneously in the New England Journal of Medicine.
The study showed no significant differences between the filter and no-filter groups in the primary outcome of a composite of symptomatic pulmonary embolism or death from any cause at 90 days after enrollment (13.9% vs. 14.4% respectively, P = .98).
In a prespecified subgroup analysis, researchers examined patients who survived 7 days after injury and did not receive prophylactic anticoagulation in those 7 days. Among this group of patients, none of those who received the vena cava filter experienced a symptomatic pulmonary embolism between day 8 and day 90, but five patients (14.7%) in the no-filter group did.
Filters were left in place for a median duration of 27 days (11-90 days). Among the 122 patients who received a filter – which included two patients in the control group – researchers found trapped thrombi in the filter in six patients.
Transfusion requirements, and the incidence of major and nonmajor bleeding and leg deep vein thrombosis, were similar between the filter and no-filter groups. Seven patients in the filter group (5.7%) required more than one attempt to remove the filter, and in one patient the filter had to be removed surgically.
Kwok M. Ho, PhD, of the department of intensive care medicine at Royal Perth Hospital, Australia, and coauthors wrote that while vena cava filters are widely used in trauma centers to prevent pulmonary embolism in patients at high risk of bleeding, there are conflicting recommendations regarding their use, and most studies so far have been observational.
“Given the cost and risks associated with a vena cava filter, our data suggest that there is no urgency to insert the filter in patients who can be treated with prophylactic anticoagulation within 7 days after injury,” they wrote. “Unnecessary insertion of a vena cava filter has the potential to cause harm.”
However, they noted that patients with multiple, large intracranial hematomas were particularly at risk from bleeding with anticoagulant therapy, and therefore may benefit from the use of a vena cava filter.
The Medical Research Foundation of Royal Perth Hospital and the Western Australian Department of Health funded the study. Dr. Ho reported funding from the Western Australian Department of Health and the Raine Medical Research Foundation to conduct the study, as well as serving as an adviser to Medtronic and Cardinal Health.
SOURCE: Ho KM et al. N Engl J Med. 2019 Jul 7. doi: 10.156/NEJMoa1806515.
MELBOURNE – Use of a prophylactic vena cava filter to trap blood clots in severely injured patients does not appear to reduce the risk of pulmonary embolism or death, according to data presented at the International Society on Thrombosis and Haemostasis congress.
The researchers reported the outcomes of a multicenter, controlled trial in which 240 severely injured patients with a contraindication to anticoagulants were randomized to receive a vena cava filter within 72 hours of admission, or no filter. The findings were published simultaneously in the New England Journal of Medicine.
The study showed no significant differences between the filter and no-filter groups in the primary outcome of a composite of symptomatic pulmonary embolism or death from any cause at 90 days after enrollment (13.9% vs. 14.4% respectively, P = .98).
In a prespecified subgroup analysis, researchers examined patients who survived 7 days after injury and did not receive prophylactic anticoagulation in those 7 days. Among this group of patients, none of those who received the vena cava filter experienced a symptomatic pulmonary embolism between day 8 and day 90, but five patients (14.7%) in the no-filter group did.
Filters were left in place for a median duration of 27 days (11-90 days). Among the 122 patients who received a filter – which included two patients in the control group – researchers found trapped thrombi in the filter in six patients.
Transfusion requirements, and the incidence of major and nonmajor bleeding and leg deep vein thrombosis, were similar between the filter and no-filter groups. Seven patients in the filter group (5.7%) required more than one attempt to remove the filter, and in one patient the filter had to be removed surgically.
Kwok M. Ho, PhD, of the department of intensive care medicine at Royal Perth Hospital, Australia, and coauthors wrote that while vena cava filters are widely used in trauma centers to prevent pulmonary embolism in patients at high risk of bleeding, there are conflicting recommendations regarding their use, and most studies so far have been observational.
“Given the cost and risks associated with a vena cava filter, our data suggest that there is no urgency to insert the filter in patients who can be treated with prophylactic anticoagulation within 7 days after injury,” they wrote. “Unnecessary insertion of a vena cava filter has the potential to cause harm.”
However, they noted that patients with multiple, large intracranial hematomas were particularly at risk from bleeding with anticoagulant therapy, and therefore may benefit from the use of a vena cava filter.
The Medical Research Foundation of Royal Perth Hospital and the Western Australian Department of Health funded the study. Dr. Ho reported funding from the Western Australian Department of Health and the Raine Medical Research Foundation to conduct the study, as well as serving as an adviser to Medtronic and Cardinal Health.
SOURCE: Ho KM et al. N Engl J Med. 2019 Jul 7. doi: 10.156/NEJMoa1806515.
MELBOURNE – Use of a prophylactic vena cava filter to trap blood clots in severely injured patients does not appear to reduce the risk of pulmonary embolism or death, according to data presented at the International Society on Thrombosis and Haemostasis congress.
The researchers reported the outcomes of a multicenter, controlled trial in which 240 severely injured patients with a contraindication to anticoagulants were randomized to receive a vena cava filter within 72 hours of admission, or no filter. The findings were published simultaneously in the New England Journal of Medicine.
The study showed no significant differences between the filter and no-filter groups in the primary outcome of a composite of symptomatic pulmonary embolism or death from any cause at 90 days after enrollment (13.9% vs. 14.4% respectively, P = .98).
In a prespecified subgroup analysis, researchers examined patients who survived 7 days after injury and did not receive prophylactic anticoagulation in those 7 days. Among this group of patients, none of those who received the vena cava filter experienced a symptomatic pulmonary embolism between day 8 and day 90, but five patients (14.7%) in the no-filter group did.
Filters were left in place for a median duration of 27 days (11-90 days). Among the 122 patients who received a filter – which included two patients in the control group – researchers found trapped thrombi in the filter in six patients.
Transfusion requirements, and the incidence of major and nonmajor bleeding and leg deep vein thrombosis, were similar between the filter and no-filter groups. Seven patients in the filter group (5.7%) required more than one attempt to remove the filter, and in one patient the filter had to be removed surgically.
Kwok M. Ho, PhD, of the department of intensive care medicine at Royal Perth Hospital, Australia, and coauthors wrote that while vena cava filters are widely used in trauma centers to prevent pulmonary embolism in patients at high risk of bleeding, there are conflicting recommendations regarding their use, and most studies so far have been observational.
“Given the cost and risks associated with a vena cava filter, our data suggest that there is no urgency to insert the filter in patients who can be treated with prophylactic anticoagulation within 7 days after injury,” they wrote. “Unnecessary insertion of a vena cava filter has the potential to cause harm.”
However, they noted that patients with multiple, large intracranial hematomas were particularly at risk from bleeding with anticoagulant therapy, and therefore may benefit from the use of a vena cava filter.
The Medical Research Foundation of Royal Perth Hospital and the Western Australian Department of Health funded the study. Dr. Ho reported funding from the Western Australian Department of Health and the Raine Medical Research Foundation to conduct the study, as well as serving as an adviser to Medtronic and Cardinal Health.
SOURCE: Ho KM et al. N Engl J Med. 2019 Jul 7. doi: 10.156/NEJMoa1806515.
REPORTING FROM 2019 ISTH CONGRESS
Sicker COPD patients may be more likely to initiate arformoterol
according to study to identify predictors of its use. In addition to being sicker, being treated by a pulmonologist rather than a primary care physician and being white were factors that increased a patient’s likelihood of receiving nebulized arformoterol.
Patients less likely to receive the nebulized version of this long-acting beta2 adrenoreceptor agonist (LABA) were African Americans, patients with psychiatric comorbidities, and patients eligible for both Medicare and Medicaid.
“Studies have shown that 40% to 71% of Medicare beneficiaries receive no maintenance treatment for COPD. Although a recent longitudinal study on Medicare populations reported that use of maintenance medications has been improving, in general, it is recognized that Medicare beneficiaries with COPD remain undertreated,” Todd P. Gilmer, PhD, from the department of family medicine and public health at the University of California, San Diego, and colleagues wrote.
The investigators identified patients with COPD using Medicare administrative data; of these patients, 11,887 were arformoterol users, and 450,178 were control patients who did not use arformoterol. Patients were included in the study if they had at least one claim for COPD medication and were continuously enrolled in Medicare Parts A, B, and D. The cohort consisted of mostly white women aged 70 years or older, and 47% were dual-eligible to receive both Medicare and Medicaid benefits. A subgroup of 1,778 arformoterol users were also identified for analysis who were hospitalized and discharged within 30 days of using arformoterol, as well as a subgroup of 21,910 control patients with hospitalizations.
The researchers found COPD-related hospitalization (odds ratio, 1.31; 95% confidence interval, 1.24-1.39; P less than .001), exacerbation (OR, 1.33; 95% CI, 1.26-1.41; P less than .001), use of a systemic corticosteroid (OR, 1.50; 95% CI, 1.43-1.57; P less than .001) or methylxanthine (OR, 1.37; 95% CI, 1.28-1.47; P less than .001), use of oxygen therapy (OR, 2.01; 95% CI, 1.93-2.09; P less than .001), pulmonologist care (OR, 1.40; 95% CI, 1.34-1.46; P less than .001), and respiratory therapist care (OR, 1.23; 95% CI, 1.11-1.36; P less than .001) strongly predicted arformoterol use, while racial/ethnic minority status, psychiatric comorbidity (OR, 0.65; 95% CI, 0.56-0.76; P less than .001), acquired immune deficiency syndrome (OR, 0.69; 95% CI, 0.52-0.94; P less than .01), and dual-eligibility for Medicare and Medicaid (OR, 0.73; 95% CI, 0.70-0.77; P less than .001) lowered the odds of arformoterol use (P less than .001). In the subgroup of patients with hospitalizations, COPD-related admission (OR, 1.83; 95% CI, 1.55-2.14; P less than .001), exacerbation (OR, 2.62; 95% CI, 1.88-3.63; P less than .001)m and inpatient care from a pulmonologist (OR, 1.78; 95% CI, 1.58-2.01; P less than .001) predicted arformoterol use.
“Given the results of this study, increasing access to nebulized maintenance therapy is warranted for select populations with COPD including racial/ethnic minorities, the dual-eligible, and those with certain comorbidities, such as psychiatric disorders,” Dr. Gilmer and colleagues wrote in their study. “Future studies are needed to explore the optimal time to initiate nebulized maintenance therapy, and the potential differential impact of early versus late initiation on patient outcomes.”
Researchers noted that, although their results may seem initially counterintuitive given that COPD has a higher prevalence in men, 56% of the beneficiaries in their Medicare data were women who were 65 years or older, and the results are consistent with other studies that show similar gender distribution findings for maintenance treatment patterns among COPD patients receiving Medicare.
“Since most Medicare beneficiaries with COPD are older than 70 years of age, the higher percentage of women than men in our two cohorts can be explained by the age distributions that ensued as a result of applying our various inclusion and exclusion criteria,” they said.
This study was funded by Sunovian. Dr. Gilmer and one coauthor are paid employees of University of California San Diego, which receives research funding from Advance Health Solutions. Another coauthor is an advisory board member for Advance Health Solutions and a consultant for GlaxoSmithKline, Boehringer-Ingelheim, Astra Zeneca, Novartis, and Pulmonix. Two other coauthors are paid employees of Advance Health Solutions, and another is a paid employee of Sunovion.
SOURCE: Gilmer TP et al. COPD. 2019 Jun 19. doi: 10.1080/15412555.2019.1618256.
according to study to identify predictors of its use. In addition to being sicker, being treated by a pulmonologist rather than a primary care physician and being white were factors that increased a patient’s likelihood of receiving nebulized arformoterol.
Patients less likely to receive the nebulized version of this long-acting beta2 adrenoreceptor agonist (LABA) were African Americans, patients with psychiatric comorbidities, and patients eligible for both Medicare and Medicaid.
“Studies have shown that 40% to 71% of Medicare beneficiaries receive no maintenance treatment for COPD. Although a recent longitudinal study on Medicare populations reported that use of maintenance medications has been improving, in general, it is recognized that Medicare beneficiaries with COPD remain undertreated,” Todd P. Gilmer, PhD, from the department of family medicine and public health at the University of California, San Diego, and colleagues wrote.
The investigators identified patients with COPD using Medicare administrative data; of these patients, 11,887 were arformoterol users, and 450,178 were control patients who did not use arformoterol. Patients were included in the study if they had at least one claim for COPD medication and were continuously enrolled in Medicare Parts A, B, and D. The cohort consisted of mostly white women aged 70 years or older, and 47% were dual-eligible to receive both Medicare and Medicaid benefits. A subgroup of 1,778 arformoterol users were also identified for analysis who were hospitalized and discharged within 30 days of using arformoterol, as well as a subgroup of 21,910 control patients with hospitalizations.
The researchers found COPD-related hospitalization (odds ratio, 1.31; 95% confidence interval, 1.24-1.39; P less than .001), exacerbation (OR, 1.33; 95% CI, 1.26-1.41; P less than .001), use of a systemic corticosteroid (OR, 1.50; 95% CI, 1.43-1.57; P less than .001) or methylxanthine (OR, 1.37; 95% CI, 1.28-1.47; P less than .001), use of oxygen therapy (OR, 2.01; 95% CI, 1.93-2.09; P less than .001), pulmonologist care (OR, 1.40; 95% CI, 1.34-1.46; P less than .001), and respiratory therapist care (OR, 1.23; 95% CI, 1.11-1.36; P less than .001) strongly predicted arformoterol use, while racial/ethnic minority status, psychiatric comorbidity (OR, 0.65; 95% CI, 0.56-0.76; P less than .001), acquired immune deficiency syndrome (OR, 0.69; 95% CI, 0.52-0.94; P less than .01), and dual-eligibility for Medicare and Medicaid (OR, 0.73; 95% CI, 0.70-0.77; P less than .001) lowered the odds of arformoterol use (P less than .001). In the subgroup of patients with hospitalizations, COPD-related admission (OR, 1.83; 95% CI, 1.55-2.14; P less than .001), exacerbation (OR, 2.62; 95% CI, 1.88-3.63; P less than .001)m and inpatient care from a pulmonologist (OR, 1.78; 95% CI, 1.58-2.01; P less than .001) predicted arformoterol use.
“Given the results of this study, increasing access to nebulized maintenance therapy is warranted for select populations with COPD including racial/ethnic minorities, the dual-eligible, and those with certain comorbidities, such as psychiatric disorders,” Dr. Gilmer and colleagues wrote in their study. “Future studies are needed to explore the optimal time to initiate nebulized maintenance therapy, and the potential differential impact of early versus late initiation on patient outcomes.”
Researchers noted that, although their results may seem initially counterintuitive given that COPD has a higher prevalence in men, 56% of the beneficiaries in their Medicare data were women who were 65 years or older, and the results are consistent with other studies that show similar gender distribution findings for maintenance treatment patterns among COPD patients receiving Medicare.
“Since most Medicare beneficiaries with COPD are older than 70 years of age, the higher percentage of women than men in our two cohorts can be explained by the age distributions that ensued as a result of applying our various inclusion and exclusion criteria,” they said.
This study was funded by Sunovian. Dr. Gilmer and one coauthor are paid employees of University of California San Diego, which receives research funding from Advance Health Solutions. Another coauthor is an advisory board member for Advance Health Solutions and a consultant for GlaxoSmithKline, Boehringer-Ingelheim, Astra Zeneca, Novartis, and Pulmonix. Two other coauthors are paid employees of Advance Health Solutions, and another is a paid employee of Sunovion.
SOURCE: Gilmer TP et al. COPD. 2019 Jun 19. doi: 10.1080/15412555.2019.1618256.
according to study to identify predictors of its use. In addition to being sicker, being treated by a pulmonologist rather than a primary care physician and being white were factors that increased a patient’s likelihood of receiving nebulized arformoterol.
Patients less likely to receive the nebulized version of this long-acting beta2 adrenoreceptor agonist (LABA) were African Americans, patients with psychiatric comorbidities, and patients eligible for both Medicare and Medicaid.
“Studies have shown that 40% to 71% of Medicare beneficiaries receive no maintenance treatment for COPD. Although a recent longitudinal study on Medicare populations reported that use of maintenance medications has been improving, in general, it is recognized that Medicare beneficiaries with COPD remain undertreated,” Todd P. Gilmer, PhD, from the department of family medicine and public health at the University of California, San Diego, and colleagues wrote.
The investigators identified patients with COPD using Medicare administrative data; of these patients, 11,887 were arformoterol users, and 450,178 were control patients who did not use arformoterol. Patients were included in the study if they had at least one claim for COPD medication and were continuously enrolled in Medicare Parts A, B, and D. The cohort consisted of mostly white women aged 70 years or older, and 47% were dual-eligible to receive both Medicare and Medicaid benefits. A subgroup of 1,778 arformoterol users were also identified for analysis who were hospitalized and discharged within 30 days of using arformoterol, as well as a subgroup of 21,910 control patients with hospitalizations.
The researchers found COPD-related hospitalization (odds ratio, 1.31; 95% confidence interval, 1.24-1.39; P less than .001), exacerbation (OR, 1.33; 95% CI, 1.26-1.41; P less than .001), use of a systemic corticosteroid (OR, 1.50; 95% CI, 1.43-1.57; P less than .001) or methylxanthine (OR, 1.37; 95% CI, 1.28-1.47; P less than .001), use of oxygen therapy (OR, 2.01; 95% CI, 1.93-2.09; P less than .001), pulmonologist care (OR, 1.40; 95% CI, 1.34-1.46; P less than .001), and respiratory therapist care (OR, 1.23; 95% CI, 1.11-1.36; P less than .001) strongly predicted arformoterol use, while racial/ethnic minority status, psychiatric comorbidity (OR, 0.65; 95% CI, 0.56-0.76; P less than .001), acquired immune deficiency syndrome (OR, 0.69; 95% CI, 0.52-0.94; P less than .01), and dual-eligibility for Medicare and Medicaid (OR, 0.73; 95% CI, 0.70-0.77; P less than .001) lowered the odds of arformoterol use (P less than .001). In the subgroup of patients with hospitalizations, COPD-related admission (OR, 1.83; 95% CI, 1.55-2.14; P less than .001), exacerbation (OR, 2.62; 95% CI, 1.88-3.63; P less than .001)m and inpatient care from a pulmonologist (OR, 1.78; 95% CI, 1.58-2.01; P less than .001) predicted arformoterol use.
“Given the results of this study, increasing access to nebulized maintenance therapy is warranted for select populations with COPD including racial/ethnic minorities, the dual-eligible, and those with certain comorbidities, such as psychiatric disorders,” Dr. Gilmer and colleagues wrote in their study. “Future studies are needed to explore the optimal time to initiate nebulized maintenance therapy, and the potential differential impact of early versus late initiation on patient outcomes.”
Researchers noted that, although their results may seem initially counterintuitive given that COPD has a higher prevalence in men, 56% of the beneficiaries in their Medicare data were women who were 65 years or older, and the results are consistent with other studies that show similar gender distribution findings for maintenance treatment patterns among COPD patients receiving Medicare.
“Since most Medicare beneficiaries with COPD are older than 70 years of age, the higher percentage of women than men in our two cohorts can be explained by the age distributions that ensued as a result of applying our various inclusion and exclusion criteria,” they said.
This study was funded by Sunovian. Dr. Gilmer and one coauthor are paid employees of University of California San Diego, which receives research funding from Advance Health Solutions. Another coauthor is an advisory board member for Advance Health Solutions and a consultant for GlaxoSmithKline, Boehringer-Ingelheim, Astra Zeneca, Novartis, and Pulmonix. Two other coauthors are paid employees of Advance Health Solutions, and another is a paid employee of Sunovion.
SOURCE: Gilmer TP et al. COPD. 2019 Jun 19. doi: 10.1080/15412555.2019.1618256.
FROM COPD: JOURNAL OF CHRONIC OBSTRUCTIVE PULMONARY DISEASE
Study: Most patients hospitalized with pneumonia receive excessive antibiotics
Two-thirds of patients hospitalized with pneumonia received an excess duration of antibiotics, according to a recent study of more than 6,000 patients.
.
The findings bolster a growing body of evidence showing that short-course therapy for pneumonia is safe and that longer durations are not only unnecessary, but “potentially harmful,” said Valerie M. Vaughn, MD, assistant professor of medicine at the University of Michigan, Ann Arbor, and coinvestigators.
“Reducing excess treatment durations should be a top priority for antibiotic stewardship nationally,” the investigators wrote in their report, which appears in the Annals of Internal Medicine.
The primary analysis of their retrospective cohort study included 6,481 individuals with pneumonia treated at 43 hospitals participating in a statewide quality initiative designed to improve care for hospitalized medical patients at risk of adverse events. About half of the patients were women, and the median age was 70 years. Nearly 60% had severe pneumonia.
The primary outcome of the study was the rate of excess antibiotic therapy duration beyond the shortest expected treatment duration consistent with guidelines. Patients with community-acquired pneumonia (CAP), representing about three-quarters of the study cohort, were expected to have a treatment duration of at least 5 days, while patients with health care–acquired pneumonia (HCAP) were expected to have at least 7 days of treatment.
Overall, 4,391 patients (67.8%) had antibiotic courses longer than the shortest effective duration, with a median duration of 8 days, and a median excess duration of 2 days, the researchers noted.
The great majority of excess days (93.2%) were due to antibiotic prescribed at discharge, according to Dr. Vaughn and colleagues.
Excess treatment duration was not linked to any improvement in 30-day mortality, readmission rates, or subsequent emergency department visits, they found.
In a telephone call at 30 days, 38% of patients treated to excess said they had gone to the doctor for an antibiotic-associated adverse event, compared with 31% who received appropriate-length courses (P = .003).
Odds of a patient-reported adverse event were increased by 5% for every excess treatment day, the investigators wrote.
Taken together, these findings have implications for patient care, research efforts, and future guidelines, according to Dr. Vaughn and coinvestigators.
“The next iteration of CAP and HCAP guidelines should explicitly recommend (rather than imply) that providers prescribe the shortest effective duration,” they said in a discussion of their study results.
Dr. Vaughn reported no disclosures related to the study. Coauthors reported grants from Blue Cross Blue Shield of Michigan and the Agency for Healthcare Research and Quality, personal fees from Wiley Publishing, and royalties from Wolters Kluwer Publishing and Oxford University Press, among other disclosures.
SOURCE: Vaughn VM et al. Ann Intern Med. 2019;171:153-63. doi: 10.7326/M18-3640.
This study by Vaughn and colleagues adds “valuable insight” to an already considerable body of evidence showing that shorter durations of antibiotic therapy are effective and limit potential harm due to adverse effects, authors of an accompanying editorial said.
“After dozens of randomized, controlled trials and more than a decade since the initial clarion call to move to short-course therapy, it is time to adapt clinical practice for diseases that have been studied and adopt the mantra ‘shorter is better,’ ” Brad Spellberg, MD, and Louis B. Rice, MD, wrote in their editorial.
“It is time for regulatory agencies, payers, and professional societies to align themselves with the overwhelming data and assist in converting practice patterns to short-course therapy,” the authors said.
Brad Spellberg, MD, is with the Los Angeles County–University of Southern California Medical Center, and Louis B. Rice, MD, is with Rhode Island Hospital, Brown University, Providence, R.I. Their editorial appears in Annals of Internal Medicine. The authors reported disclosures outside the submitted work from Alexion, Paratek, TheoremDx, Acurx, Shionogi, Merck, Motif, BioAIM, Mycomed, and ExBaq (Dr. Spellberg); and Zavante Pharmaceuticals and Macrolide (Dr. Rice).
This study by Vaughn and colleagues adds “valuable insight” to an already considerable body of evidence showing that shorter durations of antibiotic therapy are effective and limit potential harm due to adverse effects, authors of an accompanying editorial said.
“After dozens of randomized, controlled trials and more than a decade since the initial clarion call to move to short-course therapy, it is time to adapt clinical practice for diseases that have been studied and adopt the mantra ‘shorter is better,’ ” Brad Spellberg, MD, and Louis B. Rice, MD, wrote in their editorial.
“It is time for regulatory agencies, payers, and professional societies to align themselves with the overwhelming data and assist in converting practice patterns to short-course therapy,” the authors said.
Brad Spellberg, MD, is with the Los Angeles County–University of Southern California Medical Center, and Louis B. Rice, MD, is with Rhode Island Hospital, Brown University, Providence, R.I. Their editorial appears in Annals of Internal Medicine. The authors reported disclosures outside the submitted work from Alexion, Paratek, TheoremDx, Acurx, Shionogi, Merck, Motif, BioAIM, Mycomed, and ExBaq (Dr. Spellberg); and Zavante Pharmaceuticals and Macrolide (Dr. Rice).
This study by Vaughn and colleagues adds “valuable insight” to an already considerable body of evidence showing that shorter durations of antibiotic therapy are effective and limit potential harm due to adverse effects, authors of an accompanying editorial said.
“After dozens of randomized, controlled trials and more than a decade since the initial clarion call to move to short-course therapy, it is time to adapt clinical practice for diseases that have been studied and adopt the mantra ‘shorter is better,’ ” Brad Spellberg, MD, and Louis B. Rice, MD, wrote in their editorial.
“It is time for regulatory agencies, payers, and professional societies to align themselves with the overwhelming data and assist in converting practice patterns to short-course therapy,” the authors said.
Brad Spellberg, MD, is with the Los Angeles County–University of Southern California Medical Center, and Louis B. Rice, MD, is with Rhode Island Hospital, Brown University, Providence, R.I. Their editorial appears in Annals of Internal Medicine. The authors reported disclosures outside the submitted work from Alexion, Paratek, TheoremDx, Acurx, Shionogi, Merck, Motif, BioAIM, Mycomed, and ExBaq (Dr. Spellberg); and Zavante Pharmaceuticals and Macrolide (Dr. Rice).
Two-thirds of patients hospitalized with pneumonia received an excess duration of antibiotics, according to a recent study of more than 6,000 patients.
.
The findings bolster a growing body of evidence showing that short-course therapy for pneumonia is safe and that longer durations are not only unnecessary, but “potentially harmful,” said Valerie M. Vaughn, MD, assistant professor of medicine at the University of Michigan, Ann Arbor, and coinvestigators.
“Reducing excess treatment durations should be a top priority for antibiotic stewardship nationally,” the investigators wrote in their report, which appears in the Annals of Internal Medicine.
The primary analysis of their retrospective cohort study included 6,481 individuals with pneumonia treated at 43 hospitals participating in a statewide quality initiative designed to improve care for hospitalized medical patients at risk of adverse events. About half of the patients were women, and the median age was 70 years. Nearly 60% had severe pneumonia.
The primary outcome of the study was the rate of excess antibiotic therapy duration beyond the shortest expected treatment duration consistent with guidelines. Patients with community-acquired pneumonia (CAP), representing about three-quarters of the study cohort, were expected to have a treatment duration of at least 5 days, while patients with health care–acquired pneumonia (HCAP) were expected to have at least 7 days of treatment.
Overall, 4,391 patients (67.8%) had antibiotic courses longer than the shortest effective duration, with a median duration of 8 days, and a median excess duration of 2 days, the researchers noted.
The great majority of excess days (93.2%) were due to antibiotic prescribed at discharge, according to Dr. Vaughn and colleagues.
Excess treatment duration was not linked to any improvement in 30-day mortality, readmission rates, or subsequent emergency department visits, they found.
In a telephone call at 30 days, 38% of patients treated to excess said they had gone to the doctor for an antibiotic-associated adverse event, compared with 31% who received appropriate-length courses (P = .003).
Odds of a patient-reported adverse event were increased by 5% for every excess treatment day, the investigators wrote.
Taken together, these findings have implications for patient care, research efforts, and future guidelines, according to Dr. Vaughn and coinvestigators.
“The next iteration of CAP and HCAP guidelines should explicitly recommend (rather than imply) that providers prescribe the shortest effective duration,” they said in a discussion of their study results.
Dr. Vaughn reported no disclosures related to the study. Coauthors reported grants from Blue Cross Blue Shield of Michigan and the Agency for Healthcare Research and Quality, personal fees from Wiley Publishing, and royalties from Wolters Kluwer Publishing and Oxford University Press, among other disclosures.
SOURCE: Vaughn VM et al. Ann Intern Med. 2019;171:153-63. doi: 10.7326/M18-3640.
Two-thirds of patients hospitalized with pneumonia received an excess duration of antibiotics, according to a recent study of more than 6,000 patients.
.
The findings bolster a growing body of evidence showing that short-course therapy for pneumonia is safe and that longer durations are not only unnecessary, but “potentially harmful,” said Valerie M. Vaughn, MD, assistant professor of medicine at the University of Michigan, Ann Arbor, and coinvestigators.
“Reducing excess treatment durations should be a top priority for antibiotic stewardship nationally,” the investigators wrote in their report, which appears in the Annals of Internal Medicine.
The primary analysis of their retrospective cohort study included 6,481 individuals with pneumonia treated at 43 hospitals participating in a statewide quality initiative designed to improve care for hospitalized medical patients at risk of adverse events. About half of the patients were women, and the median age was 70 years. Nearly 60% had severe pneumonia.
The primary outcome of the study was the rate of excess antibiotic therapy duration beyond the shortest expected treatment duration consistent with guidelines. Patients with community-acquired pneumonia (CAP), representing about three-quarters of the study cohort, were expected to have a treatment duration of at least 5 days, while patients with health care–acquired pneumonia (HCAP) were expected to have at least 7 days of treatment.
Overall, 4,391 patients (67.8%) had antibiotic courses longer than the shortest effective duration, with a median duration of 8 days, and a median excess duration of 2 days, the researchers noted.
The great majority of excess days (93.2%) were due to antibiotic prescribed at discharge, according to Dr. Vaughn and colleagues.
Excess treatment duration was not linked to any improvement in 30-day mortality, readmission rates, or subsequent emergency department visits, they found.
In a telephone call at 30 days, 38% of patients treated to excess said they had gone to the doctor for an antibiotic-associated adverse event, compared with 31% who received appropriate-length courses (P = .003).
Odds of a patient-reported adverse event were increased by 5% for every excess treatment day, the investigators wrote.
Taken together, these findings have implications for patient care, research efforts, and future guidelines, according to Dr. Vaughn and coinvestigators.
“The next iteration of CAP and HCAP guidelines should explicitly recommend (rather than imply) that providers prescribe the shortest effective duration,” they said in a discussion of their study results.
Dr. Vaughn reported no disclosures related to the study. Coauthors reported grants from Blue Cross Blue Shield of Michigan and the Agency for Healthcare Research and Quality, personal fees from Wiley Publishing, and royalties from Wolters Kluwer Publishing and Oxford University Press, among other disclosures.
SOURCE: Vaughn VM et al. Ann Intern Med. 2019;171:153-63. doi: 10.7326/M18-3640.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point: Excessive antibiotic therapy was common among patients hospitalized with pneumonia and linked to an increase in patient-reported adverse events.
Major finding: Two-thirds (67.8%) of patients had antibiotic courses longer than the shortest effective duration.
Study details: Retrospective cohort study of 6,481 individuals with pneumonia treated at 43 hospitals participating in a statewide quality initiative.
Disclosures: Study authors reported grants from Blue Cross Blue Shield of Michigan and the Agency for Healthcare Research and Quality, personal fees from Wiley Publishing, and royalties from Wolters Kluwer Publishing and Oxford University Press, among other disclosures.
Source: Vaughn VM et al. Ann Intern Med. 2019;171:153-63. doi: 10.7326/M18-3640.
Acquired MMR immunity doesn’t last to age 1 year
according to results of a study in Vaccine.
María José Cilleruelo, PhD, of Hospital Universitario Puerta de Hierro in Majadahonda, Spain, and colleagues showed that, although most infants acquire some protective antibodies against MMR from their mothers during gestation, most have lost this protection as early as 3 months of age. This single-center, observational, prospective study was conducted between October 2013 and December 2014, and it began with 146 mother-child pairs, with 99 remaining in follow-up at 3 months, 77 at 6 months, 63 at 9 months, and 30 at 12 months. For measles, 88% of newborns were seropositive, but only 19% were at 3 months; for mumps, 70% of newborns were seropositive, but only 11% were at 3 months; and for rubella, 91% of newborns were seropositive, but only 13% were at 3 months. No infants were seropositive for mumps or rubella at 9 months, and only 2% were for measles. No infants were seropositive for any of these viruses by 12 months of age.
The investigators noted that, given Spain (where the study was conducted) is a country that gives the first MMR vaccine at 12 months of life, these declining titers can leave most infants vulnerable to those viruses before then.
“We suggest that it may be worth considering administering the first dose of MMR vaccine before 12 months of age,” the investigators concluded, although they advised studies be undertaken into the efficacy and safety of administration of that vaccine in infants younger than 12 months. They noted that the biggest limitation of this study was the high percentage of loss to follow-up, which limited the statistical power to make comparisons.
The study was funded by the Fondo de Investigación Sanitaria, and one of the authors was funded by CIBER de Epidemiología y Salud Pública. The authors declared that there are no conflicts of interest.
SOURCE: Cilleruelo MJ et al. Vaccine. 2019;37:4164-71.
according to results of a study in Vaccine.
María José Cilleruelo, PhD, of Hospital Universitario Puerta de Hierro in Majadahonda, Spain, and colleagues showed that, although most infants acquire some protective antibodies against MMR from their mothers during gestation, most have lost this protection as early as 3 months of age. This single-center, observational, prospective study was conducted between October 2013 and December 2014, and it began with 146 mother-child pairs, with 99 remaining in follow-up at 3 months, 77 at 6 months, 63 at 9 months, and 30 at 12 months. For measles, 88% of newborns were seropositive, but only 19% were at 3 months; for mumps, 70% of newborns were seropositive, but only 11% were at 3 months; and for rubella, 91% of newborns were seropositive, but only 13% were at 3 months. No infants were seropositive for mumps or rubella at 9 months, and only 2% were for measles. No infants were seropositive for any of these viruses by 12 months of age.
The investigators noted that, given Spain (where the study was conducted) is a country that gives the first MMR vaccine at 12 months of life, these declining titers can leave most infants vulnerable to those viruses before then.
“We suggest that it may be worth considering administering the first dose of MMR vaccine before 12 months of age,” the investigators concluded, although they advised studies be undertaken into the efficacy and safety of administration of that vaccine in infants younger than 12 months. They noted that the biggest limitation of this study was the high percentage of loss to follow-up, which limited the statistical power to make comparisons.
The study was funded by the Fondo de Investigación Sanitaria, and one of the authors was funded by CIBER de Epidemiología y Salud Pública. The authors declared that there are no conflicts of interest.
SOURCE: Cilleruelo MJ et al. Vaccine. 2019;37:4164-71.
according to results of a study in Vaccine.
María José Cilleruelo, PhD, of Hospital Universitario Puerta de Hierro in Majadahonda, Spain, and colleagues showed that, although most infants acquire some protective antibodies against MMR from their mothers during gestation, most have lost this protection as early as 3 months of age. This single-center, observational, prospective study was conducted between October 2013 and December 2014, and it began with 146 mother-child pairs, with 99 remaining in follow-up at 3 months, 77 at 6 months, 63 at 9 months, and 30 at 12 months. For measles, 88% of newborns were seropositive, but only 19% were at 3 months; for mumps, 70% of newborns were seropositive, but only 11% were at 3 months; and for rubella, 91% of newborns were seropositive, but only 13% were at 3 months. No infants were seropositive for mumps or rubella at 9 months, and only 2% were for measles. No infants were seropositive for any of these viruses by 12 months of age.
The investigators noted that, given Spain (where the study was conducted) is a country that gives the first MMR vaccine at 12 months of life, these declining titers can leave most infants vulnerable to those viruses before then.
“We suggest that it may be worth considering administering the first dose of MMR vaccine before 12 months of age,” the investigators concluded, although they advised studies be undertaken into the efficacy and safety of administration of that vaccine in infants younger than 12 months. They noted that the biggest limitation of this study was the high percentage of loss to follow-up, which limited the statistical power to make comparisons.
The study was funded by the Fondo de Investigación Sanitaria, and one of the authors was funded by CIBER de Epidemiología y Salud Pública. The authors declared that there are no conflicts of interest.
SOURCE: Cilleruelo MJ et al. Vaccine. 2019;37:4164-71.
FROM VACCINE
AASM hypopnea definition best for detecting OSA cases, study finds
SAN DIEGO – The prevalence of obstructive sleep apnea (OSA) is substantially lower using the Centers for Medicare & Medicaid Services apnea-hypopnea index definition of OSA than using the one recommended by the American Academy of Sleep Medicine.
In addition,
The findings come from an analysis which set out to assess the relationship between OSA and hypertension using the AASM-recommended definition and the 2018 American Heart Association/American College of Cardiology blood pressure guidelines, and to determine if there is an association between hypertension and OSA among individuals who did not meet the CMS definition of OSA.
“Given the substantial morbidity associated with hypertension, these results suggest that universal adoption of the AASM AHI definition would be a reasonable step in ensuring appropriate diagnosis and treatment of OSA,” lead study author Stuart F. Quan, MD, said at the annual meeting of the Associated Professional Sleep Societies.
Dr. Quan, of the division of sleep and circadian disorders at Brigham and Women’s Hospital in Boston, noted that a number of studies have demonstrated that OSA is a risk factor for hypertension and a variety of other medical conditions. “Rightly or wrongly, the most important metric for determining whether OSA is present and determining its severity, is the apnea-hypopnea index,” he said. “It’s the most common metric used for determining OSA severity, and mostly importantly, Medicare and some other insurers use this metric to determine whether a person is eligible for treatment. If a person falls above the line, they can get continuous positive airway pressure, for example. If they’re below the line, that’s too bad; they don’t have OSA insofar as the insurance company is concerned.”
There is no controversy as to what constitutes apnea, he continued, but some disagreement exists on the definition of hypopnea. The AASM recommends using a 3% oxygen desaturation or an arousal, while Medicare uses a definition of hypopnea requiring only a 4% oxygen desaturation. Hypertension definitions have changed recently as well. Before 2018, the definition of hypertension was greater than 140/90 mm Hg for people younger than age 65 years and 150/80 mm Hg for people age 65 years and older. In 2018, the AHA and ACC changed the hypertension guidelines, defining normal as less than 120/80 mm Hg.
“Previous studies linking OSA and hypertension used older definitions, but to my knowledge there are no current studies examining the association between OSA and hypertension using new definitions,” Dr. Quan said.
He reported on results from an analysis of 6,307 participants in the Sleep Heart Health Study who underwent home polysomnography. Their AHI defined by a 3% oxygen desaturation or an arousal was classified into four categories of OSA severity: fewer than 5 events per hour (normal sleep), 5-14 events per hour (mild sleep apnea), 15-29 events per hour (moderate sleep apnea), and 30 or more events per hour (severe sleep apnea).
The researchers used three definitions of dichotomous BP elevation: elevated (greater than 120/80 mm Hg or use of hypertension medications [meds]), stage 1 (greater than 130/80 mm Hg or meds), or stage 2 (greater than 140/90 mm Hg or meds). They used logistic regression to assess the association between elevated BP and/or hypertension and OSA severity, controlling for demographics and body mass index. Additional analyses utilized multiple linear regression to determine the relationship between natural log AHI and systolic and diastolic BP, controlling for the same covariates.
For all definitions of elevated BP, increasing OSA severity was associated with greater likelihood of an elevated or hypertensive status in fully adjusted models. Specifically, the odds ratios among those with elevated BP was 1.30 (95% confidence interval, 1.10-1.54), 1.41 (95% CI, 1.15-1.72), and 1.69 (95% CI, 1.32-2.17) for mild, moderate, and severe sleep apnea, respectively. The ORs among those with stage 1 BP was 1.27 (95% CI, 1.09-1.49), 1.36 (95% CI, 1.13-1.63), 1.58 (95% CI, 1.27-1.97) for mild, moderate, and severe sleep apnea, while the OR among those with stage 2 BP was 1.07 (95% CI, 0.92-1.26), 1.22 (95% CI, 1.02-1.45), 1.38 (95% CI, 1.12-1.69) for mild, moderate, and severe sleep apnea. Linear regression found that AHI was associated with both systolic and diastolic BP in fully adjusted models.
“Using the AASM and CMS AHI definitions, increasing severity of AHI is associated with greater likelihood of having an elevated blood pressure or hypertension,” Dr. Quan concluded. “However, the prevalence of OSA was substantially lower using the CMS definition of OSA. In fact, 218 of these individuals had moderate to severe OSA when the AASM definition was applied.”
He characterized the study as “a practical analysis, a way to help identify patients who might benefit from treatment. This is not the issue of whether the science of 3% AHI is better than 4%.”
The Sleep Heart Health Study was supported by the National Heart, Lung, and Blood Institute. Dr. Quan reported that he helped draft the AASM AHI recommendations but had no other relevant disclosures.
SOURCE: Quan SF et al. SLEEP 2019, Abstract 0501.
SAN DIEGO – The prevalence of obstructive sleep apnea (OSA) is substantially lower using the Centers for Medicare & Medicaid Services apnea-hypopnea index definition of OSA than using the one recommended by the American Academy of Sleep Medicine.
In addition,
The findings come from an analysis which set out to assess the relationship between OSA and hypertension using the AASM-recommended definition and the 2018 American Heart Association/American College of Cardiology blood pressure guidelines, and to determine if there is an association between hypertension and OSA among individuals who did not meet the CMS definition of OSA.
“Given the substantial morbidity associated with hypertension, these results suggest that universal adoption of the AASM AHI definition would be a reasonable step in ensuring appropriate diagnosis and treatment of OSA,” lead study author Stuart F. Quan, MD, said at the annual meeting of the Associated Professional Sleep Societies.
Dr. Quan, of the division of sleep and circadian disorders at Brigham and Women’s Hospital in Boston, noted that a number of studies have demonstrated that OSA is a risk factor for hypertension and a variety of other medical conditions. “Rightly or wrongly, the most important metric for determining whether OSA is present and determining its severity, is the apnea-hypopnea index,” he said. “It’s the most common metric used for determining OSA severity, and mostly importantly, Medicare and some other insurers use this metric to determine whether a person is eligible for treatment. If a person falls above the line, they can get continuous positive airway pressure, for example. If they’re below the line, that’s too bad; they don’t have OSA insofar as the insurance company is concerned.”
There is no controversy as to what constitutes apnea, he continued, but some disagreement exists on the definition of hypopnea. The AASM recommends using a 3% oxygen desaturation or an arousal, while Medicare uses a definition of hypopnea requiring only a 4% oxygen desaturation. Hypertension definitions have changed recently as well. Before 2018, the definition of hypertension was greater than 140/90 mm Hg for people younger than age 65 years and 150/80 mm Hg for people age 65 years and older. In 2018, the AHA and ACC changed the hypertension guidelines, defining normal as less than 120/80 mm Hg.
“Previous studies linking OSA and hypertension used older definitions, but to my knowledge there are no current studies examining the association between OSA and hypertension using new definitions,” Dr. Quan said.
He reported on results from an analysis of 6,307 participants in the Sleep Heart Health Study who underwent home polysomnography. Their AHI defined by a 3% oxygen desaturation or an arousal was classified into four categories of OSA severity: fewer than 5 events per hour (normal sleep), 5-14 events per hour (mild sleep apnea), 15-29 events per hour (moderate sleep apnea), and 30 or more events per hour (severe sleep apnea).
The researchers used three definitions of dichotomous BP elevation: elevated (greater than 120/80 mm Hg or use of hypertension medications [meds]), stage 1 (greater than 130/80 mm Hg or meds), or stage 2 (greater than 140/90 mm Hg or meds). They used logistic regression to assess the association between elevated BP and/or hypertension and OSA severity, controlling for demographics and body mass index. Additional analyses utilized multiple linear regression to determine the relationship between natural log AHI and systolic and diastolic BP, controlling for the same covariates.
For all definitions of elevated BP, increasing OSA severity was associated with greater likelihood of an elevated or hypertensive status in fully adjusted models. Specifically, the odds ratios among those with elevated BP was 1.30 (95% confidence interval, 1.10-1.54), 1.41 (95% CI, 1.15-1.72), and 1.69 (95% CI, 1.32-2.17) for mild, moderate, and severe sleep apnea, respectively. The ORs among those with stage 1 BP was 1.27 (95% CI, 1.09-1.49), 1.36 (95% CI, 1.13-1.63), 1.58 (95% CI, 1.27-1.97) for mild, moderate, and severe sleep apnea, while the OR among those with stage 2 BP was 1.07 (95% CI, 0.92-1.26), 1.22 (95% CI, 1.02-1.45), 1.38 (95% CI, 1.12-1.69) for mild, moderate, and severe sleep apnea. Linear regression found that AHI was associated with both systolic and diastolic BP in fully adjusted models.
“Using the AASM and CMS AHI definitions, increasing severity of AHI is associated with greater likelihood of having an elevated blood pressure or hypertension,” Dr. Quan concluded. “However, the prevalence of OSA was substantially lower using the CMS definition of OSA. In fact, 218 of these individuals had moderate to severe OSA when the AASM definition was applied.”
He characterized the study as “a practical analysis, a way to help identify patients who might benefit from treatment. This is not the issue of whether the science of 3% AHI is better than 4%.”
The Sleep Heart Health Study was supported by the National Heart, Lung, and Blood Institute. Dr. Quan reported that he helped draft the AASM AHI recommendations but had no other relevant disclosures.
SOURCE: Quan SF et al. SLEEP 2019, Abstract 0501.
SAN DIEGO – The prevalence of obstructive sleep apnea (OSA) is substantially lower using the Centers for Medicare & Medicaid Services apnea-hypopnea index definition of OSA than using the one recommended by the American Academy of Sleep Medicine.
In addition,
The findings come from an analysis which set out to assess the relationship between OSA and hypertension using the AASM-recommended definition and the 2018 American Heart Association/American College of Cardiology blood pressure guidelines, and to determine if there is an association between hypertension and OSA among individuals who did not meet the CMS definition of OSA.
“Given the substantial morbidity associated with hypertension, these results suggest that universal adoption of the AASM AHI definition would be a reasonable step in ensuring appropriate diagnosis and treatment of OSA,” lead study author Stuart F. Quan, MD, said at the annual meeting of the Associated Professional Sleep Societies.
Dr. Quan, of the division of sleep and circadian disorders at Brigham and Women’s Hospital in Boston, noted that a number of studies have demonstrated that OSA is a risk factor for hypertension and a variety of other medical conditions. “Rightly or wrongly, the most important metric for determining whether OSA is present and determining its severity, is the apnea-hypopnea index,” he said. “It’s the most common metric used for determining OSA severity, and mostly importantly, Medicare and some other insurers use this metric to determine whether a person is eligible for treatment. If a person falls above the line, they can get continuous positive airway pressure, for example. If they’re below the line, that’s too bad; they don’t have OSA insofar as the insurance company is concerned.”
There is no controversy as to what constitutes apnea, he continued, but some disagreement exists on the definition of hypopnea. The AASM recommends using a 3% oxygen desaturation or an arousal, while Medicare uses a definition of hypopnea requiring only a 4% oxygen desaturation. Hypertension definitions have changed recently as well. Before 2018, the definition of hypertension was greater than 140/90 mm Hg for people younger than age 65 years and 150/80 mm Hg for people age 65 years and older. In 2018, the AHA and ACC changed the hypertension guidelines, defining normal as less than 120/80 mm Hg.
“Previous studies linking OSA and hypertension used older definitions, but to my knowledge there are no current studies examining the association between OSA and hypertension using new definitions,” Dr. Quan said.
He reported on results from an analysis of 6,307 participants in the Sleep Heart Health Study who underwent home polysomnography. Their AHI defined by a 3% oxygen desaturation or an arousal was classified into four categories of OSA severity: fewer than 5 events per hour (normal sleep), 5-14 events per hour (mild sleep apnea), 15-29 events per hour (moderate sleep apnea), and 30 or more events per hour (severe sleep apnea).
The researchers used three definitions of dichotomous BP elevation: elevated (greater than 120/80 mm Hg or use of hypertension medications [meds]), stage 1 (greater than 130/80 mm Hg or meds), or stage 2 (greater than 140/90 mm Hg or meds). They used logistic regression to assess the association between elevated BP and/or hypertension and OSA severity, controlling for demographics and body mass index. Additional analyses utilized multiple linear regression to determine the relationship between natural log AHI and systolic and diastolic BP, controlling for the same covariates.
For all definitions of elevated BP, increasing OSA severity was associated with greater likelihood of an elevated or hypertensive status in fully adjusted models. Specifically, the odds ratios among those with elevated BP was 1.30 (95% confidence interval, 1.10-1.54), 1.41 (95% CI, 1.15-1.72), and 1.69 (95% CI, 1.32-2.17) for mild, moderate, and severe sleep apnea, respectively. The ORs among those with stage 1 BP was 1.27 (95% CI, 1.09-1.49), 1.36 (95% CI, 1.13-1.63), 1.58 (95% CI, 1.27-1.97) for mild, moderate, and severe sleep apnea, while the OR among those with stage 2 BP was 1.07 (95% CI, 0.92-1.26), 1.22 (95% CI, 1.02-1.45), 1.38 (95% CI, 1.12-1.69) for mild, moderate, and severe sleep apnea. Linear regression found that AHI was associated with both systolic and diastolic BP in fully adjusted models.
“Using the AASM and CMS AHI definitions, increasing severity of AHI is associated with greater likelihood of having an elevated blood pressure or hypertension,” Dr. Quan concluded. “However, the prevalence of OSA was substantially lower using the CMS definition of OSA. In fact, 218 of these individuals had moderate to severe OSA when the AASM definition was applied.”
He characterized the study as “a practical analysis, a way to help identify patients who might benefit from treatment. This is not the issue of whether the science of 3% AHI is better than 4%.”
The Sleep Heart Health Study was supported by the National Heart, Lung, and Blood Institute. Dr. Quan reported that he helped draft the AASM AHI recommendations but had no other relevant disclosures.
SOURCE: Quan SF et al. SLEEP 2019, Abstract 0501.
REPORTING FROM SLEEP 2019
Sleepiest OSA patients have worse CV outcomes
SAN ANTONIO – Patients with obstructive sleep apnea who complain of feeling tired when they wake up, being sleepy during the day, and have a high score on the Epworth Sleepiness Scale face an increased risk for cardiovascular disease, results from a population-based analysis suggest.
“OSA is a highly heterogeneous disease, with multiple clinical presentations and consequences,” the study’s first author, Diego R. Mazzotti, PhD, said at the annual meeting of the Associated Professional Sleep Societies. “These patients also have diverse comorbidities, and there are arbitrary severity definitions and variable therapeutic responses. It’s difficult to lump these patients together.”
Symptom subtypes of OSA were originally described in the Icelandic Sleep Apnea Cohort, and defined as excessively sleepy, minimally symptomatic, and disturbed sleep (Eur Respir J. 2014; 44[6]:1600-7). These distinct clusters were identified based on symptom experiences and the existence of major comorbidities. “This concept is more popular today, trying to identify symptom clusters, or groups of individuals, that share similar polysomnographic data, and then compare differences in prevalence or incidence of cardiovascular disease,” said Dr. Mazzotti, a research associate at the University of Pennsylvania, Philadelphia. “That’s a concept that needs to be moving forward.”
Dr. Mazzotti and colleagues set out to determine if OSA symptom subtypes are present in the Sleep Heart Health Study, a multicenter, prospective, community-based cohort of individuals aged 40 years and older designed to assess the cardiovascular (CV) consequences of OSA. They also wanted to know if there is additional evidence of the relevance of OSA symptom subtypes, particularly with respect to cardiovascular disease .
Participant-reported symptoms, such as difficulty falling and staying asleep, snoring, fatigue, drowsy driving and daytime sleepiness, and responses to the Epworth Sleepiness Scale were used to determine the patient’s subtype. Assessments including questionnaires and in-home polysomnography were conducted at baseline (between 1995 and 1998) and follow-up (between 2001 and 2003), while CV outcomes were assessed until the end of follow-up (between 2008 and 2011).
In all, 1,207 patients from the Sleep Heart Health Study met criteria for moderate to severe OSA (apnea-hypopnea index, or AHI, of 15 or greater) and were included in the final analysis. They were followed for a mean of 12 years. Based on the clustering of symptoms, the researchers identified four OSA symptom subtypes: disturbed sleep (12%), minimally symptomatic (33%), excessively sleepy (17%), and moderately sleepy (38%) – proportions that were similar to those observed in prior studies.
The disturbed sleep subtype presented with increased prevalence of “insomnialike” symptoms, such as difficulty initiating or maintaining sleep, according to Dr. Mazzotti. “On the other hand, the excessively sleepy subtype presented with a very high prevalence of several symptoms related to excessive daytime sleepiness, while the moderately sleepy showed a moderately high prevalence of such symptoms, but not as much when compared to the excessively sleepy subtype,” he explained. “Finally, the minimally symptomatic subtype was found to have the lowest prevalence of all investigated symptoms, suggesting that these patients have low symptom burden. They do not complain as much, even though they have moderate-to-severe OSA.”
Next, Dr. Mazzotti and colleagues used Kaplan-Meier survival analysis and Cox proportional hazards models to evaluate whether subtypes were associated with incident coronary heart disease (CHD), heart failure, and CV disease, including CV mortality. Similar analyses were performed comparing each symptom subtype with 2,830 individuals without OSA (AHI less than 5).
Compared with other subtypes, the excessively sleepy group had a more than threefold increased odds of prevalent heart failure, after adjustment for other CV risk factors. They also had a 1.7- to 2.3-fold increased risk for incident CV disease (P less than .001), CHD (P = .015) and heart failure (P = 0.018), after adjustment for other CV risk factors.
“Compared to individuals without OSA, the excessively sleepy subtype is the only subtype with increased risk of incident CV disease and CHD,” Dr. Mazzotti said. “It is possible that excessively sleepy OSA patients are more likely to benefit from CPAP therapy in preventing CV disease.” These results were published online earlier this year (Am J Respir Crit Care Med. 2019 Feb 15. doi: 10.1164/rccm.201808-1509OC).
Dr. Mazzotti reported having no financial disclosures.
SOURCE: Mazzotti D et al. SLEEP 2019, Abstract 0586.
SAN ANTONIO – Patients with obstructive sleep apnea who complain of feeling tired when they wake up, being sleepy during the day, and have a high score on the Epworth Sleepiness Scale face an increased risk for cardiovascular disease, results from a population-based analysis suggest.
“OSA is a highly heterogeneous disease, with multiple clinical presentations and consequences,” the study’s first author, Diego R. Mazzotti, PhD, said at the annual meeting of the Associated Professional Sleep Societies. “These patients also have diverse comorbidities, and there are arbitrary severity definitions and variable therapeutic responses. It’s difficult to lump these patients together.”
Symptom subtypes of OSA were originally described in the Icelandic Sleep Apnea Cohort, and defined as excessively sleepy, minimally symptomatic, and disturbed sleep (Eur Respir J. 2014; 44[6]:1600-7). These distinct clusters were identified based on symptom experiences and the existence of major comorbidities. “This concept is more popular today, trying to identify symptom clusters, or groups of individuals, that share similar polysomnographic data, and then compare differences in prevalence or incidence of cardiovascular disease,” said Dr. Mazzotti, a research associate at the University of Pennsylvania, Philadelphia. “That’s a concept that needs to be moving forward.”
Dr. Mazzotti and colleagues set out to determine if OSA symptom subtypes are present in the Sleep Heart Health Study, a multicenter, prospective, community-based cohort of individuals aged 40 years and older designed to assess the cardiovascular (CV) consequences of OSA. They also wanted to know if there is additional evidence of the relevance of OSA symptom subtypes, particularly with respect to cardiovascular disease .
Participant-reported symptoms, such as difficulty falling and staying asleep, snoring, fatigue, drowsy driving and daytime sleepiness, and responses to the Epworth Sleepiness Scale were used to determine the patient’s subtype. Assessments including questionnaires and in-home polysomnography were conducted at baseline (between 1995 and 1998) and follow-up (between 2001 and 2003), while CV outcomes were assessed until the end of follow-up (between 2008 and 2011).
In all, 1,207 patients from the Sleep Heart Health Study met criteria for moderate to severe OSA (apnea-hypopnea index, or AHI, of 15 or greater) and were included in the final analysis. They were followed for a mean of 12 years. Based on the clustering of symptoms, the researchers identified four OSA symptom subtypes: disturbed sleep (12%), minimally symptomatic (33%), excessively sleepy (17%), and moderately sleepy (38%) – proportions that were similar to those observed in prior studies.
The disturbed sleep subtype presented with increased prevalence of “insomnialike” symptoms, such as difficulty initiating or maintaining sleep, according to Dr. Mazzotti. “On the other hand, the excessively sleepy subtype presented with a very high prevalence of several symptoms related to excessive daytime sleepiness, while the moderately sleepy showed a moderately high prevalence of such symptoms, but not as much when compared to the excessively sleepy subtype,” he explained. “Finally, the minimally symptomatic subtype was found to have the lowest prevalence of all investigated symptoms, suggesting that these patients have low symptom burden. They do not complain as much, even though they have moderate-to-severe OSA.”
Next, Dr. Mazzotti and colleagues used Kaplan-Meier survival analysis and Cox proportional hazards models to evaluate whether subtypes were associated with incident coronary heart disease (CHD), heart failure, and CV disease, including CV mortality. Similar analyses were performed comparing each symptom subtype with 2,830 individuals without OSA (AHI less than 5).
Compared with other subtypes, the excessively sleepy group had a more than threefold increased odds of prevalent heart failure, after adjustment for other CV risk factors. They also had a 1.7- to 2.3-fold increased risk for incident CV disease (P less than .001), CHD (P = .015) and heart failure (P = 0.018), after adjustment for other CV risk factors.
“Compared to individuals without OSA, the excessively sleepy subtype is the only subtype with increased risk of incident CV disease and CHD,” Dr. Mazzotti said. “It is possible that excessively sleepy OSA patients are more likely to benefit from CPAP therapy in preventing CV disease.” These results were published online earlier this year (Am J Respir Crit Care Med. 2019 Feb 15. doi: 10.1164/rccm.201808-1509OC).
Dr. Mazzotti reported having no financial disclosures.
SOURCE: Mazzotti D et al. SLEEP 2019, Abstract 0586.
SAN ANTONIO – Patients with obstructive sleep apnea who complain of feeling tired when they wake up, being sleepy during the day, and have a high score on the Epworth Sleepiness Scale face an increased risk for cardiovascular disease, results from a population-based analysis suggest.
“OSA is a highly heterogeneous disease, with multiple clinical presentations and consequences,” the study’s first author, Diego R. Mazzotti, PhD, said at the annual meeting of the Associated Professional Sleep Societies. “These patients also have diverse comorbidities, and there are arbitrary severity definitions and variable therapeutic responses. It’s difficult to lump these patients together.”
Symptom subtypes of OSA were originally described in the Icelandic Sleep Apnea Cohort, and defined as excessively sleepy, minimally symptomatic, and disturbed sleep (Eur Respir J. 2014; 44[6]:1600-7). These distinct clusters were identified based on symptom experiences and the existence of major comorbidities. “This concept is more popular today, trying to identify symptom clusters, or groups of individuals, that share similar polysomnographic data, and then compare differences in prevalence or incidence of cardiovascular disease,” said Dr. Mazzotti, a research associate at the University of Pennsylvania, Philadelphia. “That’s a concept that needs to be moving forward.”
Dr. Mazzotti and colleagues set out to determine if OSA symptom subtypes are present in the Sleep Heart Health Study, a multicenter, prospective, community-based cohort of individuals aged 40 years and older designed to assess the cardiovascular (CV) consequences of OSA. They also wanted to know if there is additional evidence of the relevance of OSA symptom subtypes, particularly with respect to cardiovascular disease .
Participant-reported symptoms, such as difficulty falling and staying asleep, snoring, fatigue, drowsy driving and daytime sleepiness, and responses to the Epworth Sleepiness Scale were used to determine the patient’s subtype. Assessments including questionnaires and in-home polysomnography were conducted at baseline (between 1995 and 1998) and follow-up (between 2001 and 2003), while CV outcomes were assessed until the end of follow-up (between 2008 and 2011).
In all, 1,207 patients from the Sleep Heart Health Study met criteria for moderate to severe OSA (apnea-hypopnea index, or AHI, of 15 or greater) and were included in the final analysis. They were followed for a mean of 12 years. Based on the clustering of symptoms, the researchers identified four OSA symptom subtypes: disturbed sleep (12%), minimally symptomatic (33%), excessively sleepy (17%), and moderately sleepy (38%) – proportions that were similar to those observed in prior studies.
The disturbed sleep subtype presented with increased prevalence of “insomnialike” symptoms, such as difficulty initiating or maintaining sleep, according to Dr. Mazzotti. “On the other hand, the excessively sleepy subtype presented with a very high prevalence of several symptoms related to excessive daytime sleepiness, while the moderately sleepy showed a moderately high prevalence of such symptoms, but not as much when compared to the excessively sleepy subtype,” he explained. “Finally, the minimally symptomatic subtype was found to have the lowest prevalence of all investigated symptoms, suggesting that these patients have low symptom burden. They do not complain as much, even though they have moderate-to-severe OSA.”
Next, Dr. Mazzotti and colleagues used Kaplan-Meier survival analysis and Cox proportional hazards models to evaluate whether subtypes were associated with incident coronary heart disease (CHD), heart failure, and CV disease, including CV mortality. Similar analyses were performed comparing each symptom subtype with 2,830 individuals without OSA (AHI less than 5).
Compared with other subtypes, the excessively sleepy group had a more than threefold increased odds of prevalent heart failure, after adjustment for other CV risk factors. They also had a 1.7- to 2.3-fold increased risk for incident CV disease (P less than .001), CHD (P = .015) and heart failure (P = 0.018), after adjustment for other CV risk factors.
“Compared to individuals without OSA, the excessively sleepy subtype is the only subtype with increased risk of incident CV disease and CHD,” Dr. Mazzotti said. “It is possible that excessively sleepy OSA patients are more likely to benefit from CPAP therapy in preventing CV disease.” These results were published online earlier this year (Am J Respir Crit Care Med. 2019 Feb 15. doi: 10.1164/rccm.201808-1509OC).
Dr. Mazzotti reported having no financial disclosures.
SOURCE: Mazzotti D et al. SLEEP 2019, Abstract 0586.
REPORTING FROM SLEEP 2019
Risk of cardiac events jumps after COPD exacerbation
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
FROM RESPIROLOGY
Evidence supports accuracy of COPD diagnosis tool
The ratio of the forced expiratory volume in 1 second to the forced vital capacity (FEV1:FVC) at the recommended threshold of 0.70 effectively diagnosed individuals at risk for clinically significant COPD, a longitudinal study of more than 24,000 individuals has found.
Guidelines from respiratory societies have long recommended a diagnosis of airflow obstruction when the FEV1:FVC is less than 0.70, but no rigorous, population-based studies have been conducted to support this recommendation, wrote Surya P. Bhatt, MD, of the University of Alabama at Birmingham, and colleagues.
“The selection of a threshold for defining airflow obstruction has major implications for patient care and public health as the prevalence of airflow obstruction can vary by as much as 33% depending on which threshold is selected,” they said.
In a study published in JAMA, the researchers reviewed data from 24,207 participants in the National Heart, Lung, and Blood Institute Pooled Cohorts Study to assess the accuracy of different thresholds in predicting COPD events in a large, multiethnic, U.S. population. All participants underwent spirometry; the average age at spirometry was 63 years, and 54% of the patients were women. Patients were enrolled during 1987-2000 and received follow-up longitudinally through 2016.
Overall, 3,925 participants experienced COPD-related events during an average of 15 years of follow-up (more than 340,757 person-years). These events included 3,563 hospitalizations and 447 deaths related to COPD.
The researchers compared three thresholds for FEV1:FVC ratios: a fixed optimal threshold of 0.71, a lower limit of normal (LLN) defined as 0.034, and the currently recommended 0.70.
The optimal 0.71 was not significantly different from the recommended 0.70 but was significantly more accurate than the LLN of 0.034. In addition, the 0.70 value was the optimal predictor in a subgroup analysis of ever-smokers and in multivariate analysis.
The findings were limited by several factors including the use of prebronchodilator spirometry, lack of adjustment for medication use, and limitation of outcomes to COPD mortality or clinical events mainly caused by COPD, which might exclude patients with mild to moderate disease, the researchers noted.
However, ” to help clinicians identify patients at increased risk for significant COPD, they said.
Lead author Dr. Bhatt disclosed a National Institutes of Health grant, consulting fees from Sunovion and research funds from Proterix Bio. The study was supported by grants from multiple agencies of the National Institutes of Health, including the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging.
The ratio of the forced expiratory volume in 1 second to the forced vital capacity (FEV1:FVC) at the recommended threshold of 0.70 effectively diagnosed individuals at risk for clinically significant COPD, a longitudinal study of more than 24,000 individuals has found.
Guidelines from respiratory societies have long recommended a diagnosis of airflow obstruction when the FEV1:FVC is less than 0.70, but no rigorous, population-based studies have been conducted to support this recommendation, wrote Surya P. Bhatt, MD, of the University of Alabama at Birmingham, and colleagues.
“The selection of a threshold for defining airflow obstruction has major implications for patient care and public health as the prevalence of airflow obstruction can vary by as much as 33% depending on which threshold is selected,” they said.
In a study published in JAMA, the researchers reviewed data from 24,207 participants in the National Heart, Lung, and Blood Institute Pooled Cohorts Study to assess the accuracy of different thresholds in predicting COPD events in a large, multiethnic, U.S. population. All participants underwent spirometry; the average age at spirometry was 63 years, and 54% of the patients were women. Patients were enrolled during 1987-2000 and received follow-up longitudinally through 2016.
Overall, 3,925 participants experienced COPD-related events during an average of 15 years of follow-up (more than 340,757 person-years). These events included 3,563 hospitalizations and 447 deaths related to COPD.
The researchers compared three thresholds for FEV1:FVC ratios: a fixed optimal threshold of 0.71, a lower limit of normal (LLN) defined as 0.034, and the currently recommended 0.70.
The optimal 0.71 was not significantly different from the recommended 0.70 but was significantly more accurate than the LLN of 0.034. In addition, the 0.70 value was the optimal predictor in a subgroup analysis of ever-smokers and in multivariate analysis.
The findings were limited by several factors including the use of prebronchodilator spirometry, lack of adjustment for medication use, and limitation of outcomes to COPD mortality or clinical events mainly caused by COPD, which might exclude patients with mild to moderate disease, the researchers noted.
However, ” to help clinicians identify patients at increased risk for significant COPD, they said.
Lead author Dr. Bhatt disclosed a National Institutes of Health grant, consulting fees from Sunovion and research funds from Proterix Bio. The study was supported by grants from multiple agencies of the National Institutes of Health, including the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging.
The ratio of the forced expiratory volume in 1 second to the forced vital capacity (FEV1:FVC) at the recommended threshold of 0.70 effectively diagnosed individuals at risk for clinically significant COPD, a longitudinal study of more than 24,000 individuals has found.
Guidelines from respiratory societies have long recommended a diagnosis of airflow obstruction when the FEV1:FVC is less than 0.70, but no rigorous, population-based studies have been conducted to support this recommendation, wrote Surya P. Bhatt, MD, of the University of Alabama at Birmingham, and colleagues.
“The selection of a threshold for defining airflow obstruction has major implications for patient care and public health as the prevalence of airflow obstruction can vary by as much as 33% depending on which threshold is selected,” they said.
In a study published in JAMA, the researchers reviewed data from 24,207 participants in the National Heart, Lung, and Blood Institute Pooled Cohorts Study to assess the accuracy of different thresholds in predicting COPD events in a large, multiethnic, U.S. population. All participants underwent spirometry; the average age at spirometry was 63 years, and 54% of the patients were women. Patients were enrolled during 1987-2000 and received follow-up longitudinally through 2016.
Overall, 3,925 participants experienced COPD-related events during an average of 15 years of follow-up (more than 340,757 person-years). These events included 3,563 hospitalizations and 447 deaths related to COPD.
The researchers compared three thresholds for FEV1:FVC ratios: a fixed optimal threshold of 0.71, a lower limit of normal (LLN) defined as 0.034, and the currently recommended 0.70.
The optimal 0.71 was not significantly different from the recommended 0.70 but was significantly more accurate than the LLN of 0.034. In addition, the 0.70 value was the optimal predictor in a subgroup analysis of ever-smokers and in multivariate analysis.
The findings were limited by several factors including the use of prebronchodilator spirometry, lack of adjustment for medication use, and limitation of outcomes to COPD mortality or clinical events mainly caused by COPD, which might exclude patients with mild to moderate disease, the researchers noted.
However, ” to help clinicians identify patients at increased risk for significant COPD, they said.
Lead author Dr. Bhatt disclosed a National Institutes of Health grant, consulting fees from Sunovion and research funds from Proterix Bio. The study was supported by grants from multiple agencies of the National Institutes of Health, including the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging.
FROM JAMA