User login
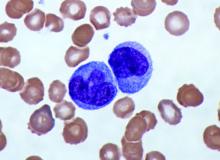
Elevated monocyte count predicts poor outcomes in idiopathic pulmonary fibrosis
, including hypertrophic cardiomyopathy, systemic sclerosis, and myelofibrosis, according to research published in The Lancet Respiratory Medicine.
The data indicate that “a single threshold value of absolute monocyte counts of 0.95 K/mcL could be used to identify high-risk patients with a fibrotic disease,” said Madeleine K. D. Scott, a researcher at Stanford (Calif.) University, and coauthors. The results “suggest that monocyte count should be incorporated into the clinical assessment” and may “enable more conscientious allocation of scarce resources, including lung transplantations,” they said.
While other published biomarkers – including gene panels and multicytokine signatures – may be expensive and not readily available, “absolute monocyte count is routinely measured as part of a complete blood count, an inexpensive test used in clinical practice worldwide,” the authors said.
Further study of monocytes’ mechanistic role in fibrosis ultimately could point to new treatment approaches.
A retrospective multicenter cohort study
To assess whether immune cells may identify patients with idiopathic pulmonary fibrosis at greater risk of poor outcomes, Ms. Scott and her collaborators conducted a retrospective multicenter cohort study.
They first analyzed transcriptome data from 120 peripheral blood mononuclear cell samples of patients with idiopathic pulmonary fibrosis, which they obtained from the Gene Expression Omnibus at the National Center for Biotechnology Information. They used statistical deconvolution to estimate percentages of 13 immune cell types and examined their associations with transplant-free survival. Their discovery analysis found that estimated CD14+ classical monocyte percentages above the mean correlated with shorter transplant-free survival times (hazard ratio, 1.82), but percentages of T cells and B cells did not.
The researchers then validated these results using samples from patients with idiopathic pulmonary fibrosis in two independent cohorts. In the COMET validation cohort, which included 45 patients with idiopathic pulmonary fibrosis whose monocyte counts were measured using flow cytometry, higher monocyte counts were significantly associated with greater risk of disease progression. In the Yale cohort, which included 15 patients with idiopathic pulmonary fibrosis, the 6 patients who were classified as high risk on the basis of a 52-gene signature had more CD14+ monocytes than the 9 low-risk patients did.
In addition, Ms. Scott and her collaborators looked at complete blood count values in the electronic health records of 45,068 patients with idiopathic pulmonary fibrosis, systemic sclerosis, hypertrophic cardiomyopathy, or myelofibrosis in Stanford, Northwestern, Vanderbilt, and Optum Clinformatics Data Mart cohorts.
Among patients in the COMET, Stanford, and Northwestern datasets, monocyte counts of 0.95 K/mcL or greater were associated with mortality after adjustment for forced vital capacity (HR, 2.47) and the gender, age, and physiology index (HR, 2.06). Data from 7,459 patients with idiopathic pulmonary fibrosis “showed that patients with monocyte counts of 0.95 K/mcL or greater were at increased risk of mortality with lung transplantation as a censoring event, after adjusting for age at diagnosis and sex” in the Stanford (HR, 2.30), Vanderbilt (HR, 1.52), and Optum (HR, 1.74) cohorts. “Likewise, higher absolute monocyte count was associated with shortened survival in patients with hypertrophic cardiomyopathy across all three cohorts, and in patients with systemic sclerosis or myelofibrosis in two of the three cohorts,” the researchers said.
The study was funded by grants from the Bill & Melinda Gates Foundation, U.S. National Institute of Allergy and Infectious Diseases, and U.S. National Library of Medicine. Ms. Scott had no competing interests. Coauthors disclosed grants, compensation, and support from foundations, agencies, and companies.
SOURCE: Scott MKD et al. Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600(18)30508-3.
The study by Scott et al. provides evidence that monocyte count may be a “novel, simple, and inexpensive prognostic biomarker in idiopathic pulmonary fibrosis,” according to an accompanying editorial.
Progress has been made in the treatment of idiopathic pulmonary fibrosis, but patient prognosis remains “challenging to predict,” wrote Michael Kreuter, MD, of University of Heidelberg, Germany, and Toby M. Maher, MB, MSc, PhD, of Royal Brompton Hospital in London and Imperial College London. “One lesson that can be learned from other respiratory disorders is that routinely measured cellular biomarkers, such as blood eosinophil counts in chronic obstructive pulmonary disease (COPD), can predict treatment responses” (Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600[19]30050-5).
Increased blood monocyte counts in idiopathic pulmonary fibrosis may reflect disease activity, which “could explain the outcome differences,” said Dr. Kreuter and Dr. Maher. “As highlighted by the investigators themselves, before introducing assessment of monocyte counts as part of routine clinical care for individuals with idiopathic pulmonary fibrosis, the limitations of this research should be taken into account. These include uncertainty around diagnosis and disease severity in a substantial subset of the patients, and the unknown effect of medical therapies (including corticosteroids and immunosuppressant and antifibrotic drugs) on monocyte counts and prognosis.” Researchers should validate the clinical value of blood monocyte counts in existing and future cohorts and evaluate the biomarker in clinical trials.
The editorialists have received compensation and funding from various pharmaceutical companies.
The study by Scott et al. provides evidence that monocyte count may be a “novel, simple, and inexpensive prognostic biomarker in idiopathic pulmonary fibrosis,” according to an accompanying editorial.
Progress has been made in the treatment of idiopathic pulmonary fibrosis, but patient prognosis remains “challenging to predict,” wrote Michael Kreuter, MD, of University of Heidelberg, Germany, and Toby M. Maher, MB, MSc, PhD, of Royal Brompton Hospital in London and Imperial College London. “One lesson that can be learned from other respiratory disorders is that routinely measured cellular biomarkers, such as blood eosinophil counts in chronic obstructive pulmonary disease (COPD), can predict treatment responses” (Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600[19]30050-5).
Increased blood monocyte counts in idiopathic pulmonary fibrosis may reflect disease activity, which “could explain the outcome differences,” said Dr. Kreuter and Dr. Maher. “As highlighted by the investigators themselves, before introducing assessment of monocyte counts as part of routine clinical care for individuals with idiopathic pulmonary fibrosis, the limitations of this research should be taken into account. These include uncertainty around diagnosis and disease severity in a substantial subset of the patients, and the unknown effect of medical therapies (including corticosteroids and immunosuppressant and antifibrotic drugs) on monocyte counts and prognosis.” Researchers should validate the clinical value of blood monocyte counts in existing and future cohorts and evaluate the biomarker in clinical trials.
The editorialists have received compensation and funding from various pharmaceutical companies.
The study by Scott et al. provides evidence that monocyte count may be a “novel, simple, and inexpensive prognostic biomarker in idiopathic pulmonary fibrosis,” according to an accompanying editorial.
Progress has been made in the treatment of idiopathic pulmonary fibrosis, but patient prognosis remains “challenging to predict,” wrote Michael Kreuter, MD, of University of Heidelberg, Germany, and Toby M. Maher, MB, MSc, PhD, of Royal Brompton Hospital in London and Imperial College London. “One lesson that can be learned from other respiratory disorders is that routinely measured cellular biomarkers, such as blood eosinophil counts in chronic obstructive pulmonary disease (COPD), can predict treatment responses” (Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600[19]30050-5).
Increased blood monocyte counts in idiopathic pulmonary fibrosis may reflect disease activity, which “could explain the outcome differences,” said Dr. Kreuter and Dr. Maher. “As highlighted by the investigators themselves, before introducing assessment of monocyte counts as part of routine clinical care for individuals with idiopathic pulmonary fibrosis, the limitations of this research should be taken into account. These include uncertainty around diagnosis and disease severity in a substantial subset of the patients, and the unknown effect of medical therapies (including corticosteroids and immunosuppressant and antifibrotic drugs) on monocyte counts and prognosis.” Researchers should validate the clinical value of blood monocyte counts in existing and future cohorts and evaluate the biomarker in clinical trials.
The editorialists have received compensation and funding from various pharmaceutical companies.
, including hypertrophic cardiomyopathy, systemic sclerosis, and myelofibrosis, according to research published in The Lancet Respiratory Medicine.
The data indicate that “a single threshold value of absolute monocyte counts of 0.95 K/mcL could be used to identify high-risk patients with a fibrotic disease,” said Madeleine K. D. Scott, a researcher at Stanford (Calif.) University, and coauthors. The results “suggest that monocyte count should be incorporated into the clinical assessment” and may “enable more conscientious allocation of scarce resources, including lung transplantations,” they said.
While other published biomarkers – including gene panels and multicytokine signatures – may be expensive and not readily available, “absolute monocyte count is routinely measured as part of a complete blood count, an inexpensive test used in clinical practice worldwide,” the authors said.
Further study of monocytes’ mechanistic role in fibrosis ultimately could point to new treatment approaches.
A retrospective multicenter cohort study
To assess whether immune cells may identify patients with idiopathic pulmonary fibrosis at greater risk of poor outcomes, Ms. Scott and her collaborators conducted a retrospective multicenter cohort study.
They first analyzed transcriptome data from 120 peripheral blood mononuclear cell samples of patients with idiopathic pulmonary fibrosis, which they obtained from the Gene Expression Omnibus at the National Center for Biotechnology Information. They used statistical deconvolution to estimate percentages of 13 immune cell types and examined their associations with transplant-free survival. Their discovery analysis found that estimated CD14+ classical monocyte percentages above the mean correlated with shorter transplant-free survival times (hazard ratio, 1.82), but percentages of T cells and B cells did not.
The researchers then validated these results using samples from patients with idiopathic pulmonary fibrosis in two independent cohorts. In the COMET validation cohort, which included 45 patients with idiopathic pulmonary fibrosis whose monocyte counts were measured using flow cytometry, higher monocyte counts were significantly associated with greater risk of disease progression. In the Yale cohort, which included 15 patients with idiopathic pulmonary fibrosis, the 6 patients who were classified as high risk on the basis of a 52-gene signature had more CD14+ monocytes than the 9 low-risk patients did.
In addition, Ms. Scott and her collaborators looked at complete blood count values in the electronic health records of 45,068 patients with idiopathic pulmonary fibrosis, systemic sclerosis, hypertrophic cardiomyopathy, or myelofibrosis in Stanford, Northwestern, Vanderbilt, and Optum Clinformatics Data Mart cohorts.
Among patients in the COMET, Stanford, and Northwestern datasets, monocyte counts of 0.95 K/mcL or greater were associated with mortality after adjustment for forced vital capacity (HR, 2.47) and the gender, age, and physiology index (HR, 2.06). Data from 7,459 patients with idiopathic pulmonary fibrosis “showed that patients with monocyte counts of 0.95 K/mcL or greater were at increased risk of mortality with lung transplantation as a censoring event, after adjusting for age at diagnosis and sex” in the Stanford (HR, 2.30), Vanderbilt (HR, 1.52), and Optum (HR, 1.74) cohorts. “Likewise, higher absolute monocyte count was associated with shortened survival in patients with hypertrophic cardiomyopathy across all three cohorts, and in patients with systemic sclerosis or myelofibrosis in two of the three cohorts,” the researchers said.
The study was funded by grants from the Bill & Melinda Gates Foundation, U.S. National Institute of Allergy and Infectious Diseases, and U.S. National Library of Medicine. Ms. Scott had no competing interests. Coauthors disclosed grants, compensation, and support from foundations, agencies, and companies.
SOURCE: Scott MKD et al. Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600(18)30508-3.
, including hypertrophic cardiomyopathy, systemic sclerosis, and myelofibrosis, according to research published in The Lancet Respiratory Medicine.
The data indicate that “a single threshold value of absolute monocyte counts of 0.95 K/mcL could be used to identify high-risk patients with a fibrotic disease,” said Madeleine K. D. Scott, a researcher at Stanford (Calif.) University, and coauthors. The results “suggest that monocyte count should be incorporated into the clinical assessment” and may “enable more conscientious allocation of scarce resources, including lung transplantations,” they said.
While other published biomarkers – including gene panels and multicytokine signatures – may be expensive and not readily available, “absolute monocyte count is routinely measured as part of a complete blood count, an inexpensive test used in clinical practice worldwide,” the authors said.
Further study of monocytes’ mechanistic role in fibrosis ultimately could point to new treatment approaches.
A retrospective multicenter cohort study
To assess whether immune cells may identify patients with idiopathic pulmonary fibrosis at greater risk of poor outcomes, Ms. Scott and her collaborators conducted a retrospective multicenter cohort study.
They first analyzed transcriptome data from 120 peripheral blood mononuclear cell samples of patients with idiopathic pulmonary fibrosis, which they obtained from the Gene Expression Omnibus at the National Center for Biotechnology Information. They used statistical deconvolution to estimate percentages of 13 immune cell types and examined their associations with transplant-free survival. Their discovery analysis found that estimated CD14+ classical monocyte percentages above the mean correlated with shorter transplant-free survival times (hazard ratio, 1.82), but percentages of T cells and B cells did not.
The researchers then validated these results using samples from patients with idiopathic pulmonary fibrosis in two independent cohorts. In the COMET validation cohort, which included 45 patients with idiopathic pulmonary fibrosis whose monocyte counts were measured using flow cytometry, higher monocyte counts were significantly associated with greater risk of disease progression. In the Yale cohort, which included 15 patients with idiopathic pulmonary fibrosis, the 6 patients who were classified as high risk on the basis of a 52-gene signature had more CD14+ monocytes than the 9 low-risk patients did.
In addition, Ms. Scott and her collaborators looked at complete blood count values in the electronic health records of 45,068 patients with idiopathic pulmonary fibrosis, systemic sclerosis, hypertrophic cardiomyopathy, or myelofibrosis in Stanford, Northwestern, Vanderbilt, and Optum Clinformatics Data Mart cohorts.
Among patients in the COMET, Stanford, and Northwestern datasets, monocyte counts of 0.95 K/mcL or greater were associated with mortality after adjustment for forced vital capacity (HR, 2.47) and the gender, age, and physiology index (HR, 2.06). Data from 7,459 patients with idiopathic pulmonary fibrosis “showed that patients with monocyte counts of 0.95 K/mcL or greater were at increased risk of mortality with lung transplantation as a censoring event, after adjusting for age at diagnosis and sex” in the Stanford (HR, 2.30), Vanderbilt (HR, 1.52), and Optum (HR, 1.74) cohorts. “Likewise, higher absolute monocyte count was associated with shortened survival in patients with hypertrophic cardiomyopathy across all three cohorts, and in patients with systemic sclerosis or myelofibrosis in two of the three cohorts,” the researchers said.
The study was funded by grants from the Bill & Melinda Gates Foundation, U.S. National Institute of Allergy and Infectious Diseases, and U.S. National Library of Medicine. Ms. Scott had no competing interests. Coauthors disclosed grants, compensation, and support from foundations, agencies, and companies.
SOURCE: Scott MKD et al. Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600(18)30508-3.
FROM THE LANCET RESPIRATORY MEDICINE
Key clinical point: An increased monocyte count predicts poor outcomes among patients with idiopathic pulmonary fibrosis and other fibrotic diseases.
Major finding: Among patients in three cohorts, monocyte counts of 0.95 K/mcL or greater were associated with mortality after adjustment for forced vital capacity (hazard ratio, 2.47) and the gender, age, and physiology index (HR, 2.06).
Study details: A retrospective analysis of data from 7,000 patients with idiopathic pulmonary fibrosis from five independent cohorts.
Disclosures: The study was funded by grants from the Bill & Melinda Gates Foundation, U.S. National Institute of Allergy and Infectious Diseases, and U.S. National Library of Medicine. Ms. Scott had no competing interests. Coauthors disclosed grants, compensation, and support from foundations, agencies, and companies.
Source: Scott MKD et al. Lancet Respir Med. 2019 Jun. doi: 10.1016/S2213-2600(18)30508-3.
CF drug picks up indication for children as young as 6
to include children as young as 6 years old who have cystic fibrosis.
The drug was approved in 2018 for patients aged 12 years and older who have the most common cause of the disease, two alleles for the F508del mutation in the gene that codes for the cystic fibrosis transmembrane conductance regulator (CFTR) protein, or at least one other CFTR mutation responsive to the combination, as listed in labeling.
The original approval was based on three phase 3, double blind, placebo-controlled trials, which demonstrated improvements in lung function and other key measures of the disease. One trial that found a 6.8% mean improvement in lung function testing over placebo at 8 weeks, and another that found a 4% improvement at 24 weeks, with fewer respiratory exacerbations and improved respiratory-related quality of life. A third trial in patients without the indicated genetic mutations was ended early for futility.
The efficacy in children under 12 years was extrapolated from those trials, plus an open-label study that found similar effects.
Labeling warns of elevated liver enzymes and cataracts in children, and notes that the drug should be taken with food that contains fat. Labeling also recommends against use with strong cytochrome P450 3A4 (CYP3A) inducers – rifampin, phenobarbital, St. John’s wort, among others – because they might reduce efficacy, and against use with CYP3A inhibitors – ketoconazole, clarithromycin, Seville oranges, grapefruit juice, etc. – because of the risk of increased exposure.
The most common side effects are headache, nausea, sinus congestion, and dizziness. The FDA has cleared a CF gene test to check for the required mutations. Symdeko is marketed by Vertex Pharmaceuticals.
to include children as young as 6 years old who have cystic fibrosis.
The drug was approved in 2018 for patients aged 12 years and older who have the most common cause of the disease, two alleles for the F508del mutation in the gene that codes for the cystic fibrosis transmembrane conductance regulator (CFTR) protein, or at least one other CFTR mutation responsive to the combination, as listed in labeling.
The original approval was based on three phase 3, double blind, placebo-controlled trials, which demonstrated improvements in lung function and other key measures of the disease. One trial that found a 6.8% mean improvement in lung function testing over placebo at 8 weeks, and another that found a 4% improvement at 24 weeks, with fewer respiratory exacerbations and improved respiratory-related quality of life. A third trial in patients without the indicated genetic mutations was ended early for futility.
The efficacy in children under 12 years was extrapolated from those trials, plus an open-label study that found similar effects.
Labeling warns of elevated liver enzymes and cataracts in children, and notes that the drug should be taken with food that contains fat. Labeling also recommends against use with strong cytochrome P450 3A4 (CYP3A) inducers – rifampin, phenobarbital, St. John’s wort, among others – because they might reduce efficacy, and against use with CYP3A inhibitors – ketoconazole, clarithromycin, Seville oranges, grapefruit juice, etc. – because of the risk of increased exposure.
The most common side effects are headache, nausea, sinus congestion, and dizziness. The FDA has cleared a CF gene test to check for the required mutations. Symdeko is marketed by Vertex Pharmaceuticals.
to include children as young as 6 years old who have cystic fibrosis.
The drug was approved in 2018 for patients aged 12 years and older who have the most common cause of the disease, two alleles for the F508del mutation in the gene that codes for the cystic fibrosis transmembrane conductance regulator (CFTR) protein, or at least one other CFTR mutation responsive to the combination, as listed in labeling.
The original approval was based on three phase 3, double blind, placebo-controlled trials, which demonstrated improvements in lung function and other key measures of the disease. One trial that found a 6.8% mean improvement in lung function testing over placebo at 8 weeks, and another that found a 4% improvement at 24 weeks, with fewer respiratory exacerbations and improved respiratory-related quality of life. A third trial in patients without the indicated genetic mutations was ended early for futility.
The efficacy in children under 12 years was extrapolated from those trials, plus an open-label study that found similar effects.
Labeling warns of elevated liver enzymes and cataracts in children, and notes that the drug should be taken with food that contains fat. Labeling also recommends against use with strong cytochrome P450 3A4 (CYP3A) inducers – rifampin, phenobarbital, St. John’s wort, among others – because they might reduce efficacy, and against use with CYP3A inhibitors – ketoconazole, clarithromycin, Seville oranges, grapefruit juice, etc. – because of the risk of increased exposure.
The most common side effects are headache, nausea, sinus congestion, and dizziness. The FDA has cleared a CF gene test to check for the required mutations. Symdeko is marketed by Vertex Pharmaceuticals.
Cannabis vaping among teens tied to tobacco use
and that practice is associated with cigars, waterpipe and e-cigarette use, findings from a survey of nearly 3,000 adolescents have shown.
“Although the prevalence of e-cigarette use among youth has increased dramatically in the past decade, little epidemiologic data exist on the prevalence of using e-cigarette devices or other specialised devices to vaporise (‘vape’) cannabis in the form of hash oil, tetrahydrocannabinol (THC) wax or oil, or dried cannabis buds or leaves,” wrote Sarah D. Kowitt, PhD, of the University of North Carolina, Chapel Hill, and colleagues. “This is surprising given that (1) cannabis (also referred to as marijuana) and e-cigarettes are the most commonly used substances by adolescents in the USA, (2) evidence exists that adolescents dual use both tobacco e-cigarettes and cannabis, and (3) longitudinal research suggests that use of e-cigarettes is associated with progression to use of cannabis.”
In a study published in BMJ Open, the researchers used data from the 2017 North Carolina Youth Tobacco Survey, a school-based survey of students in grades 6-12. The study population included 2,835 adolescents in grades 9-12.
Overall, 9.6% of students reported ever vaping cannabis. In multivariate analysis, cannabis vaping was significantly more likely among adolescents who reported using e-cigarettes (adjusted odds ratio 3.18), cigars (aOR 3.76), or water pipes (aOR 2.32) in the past 30 days, compared with peers who didn’t use tobacco.
The researchers found no significant association between smokeless tobacco use or traditional cigarette use in the past 30 days and vaping cannabis.
In a bivariate analysis, vaping cannabis was significantly more common among males vs. females (11% vs. 8.2%) and among non-Hispanic white students (11.3%), Hispanic students (10.5%), and other non-Hispanic students (11.8%) compared with non-Hispanic black students (5.0%).
In addition, prevalence of cannabis vaping increased with grade level, from 4.7% of 9th graders to 15.5% of 12th graders.
The health impacts of vaping cannabis are not well researched, but the researchers note that among the potential safety issues are earlier initiation of tobacco or cannabis use, concomitant tobacco and cannabis use, increased frequency of use or misuse of tobacco or cannabis, or increased potency of cannabis.
The results of the study were limited by several factors including the use of data only from the state of North Carolina, the lack of data on frequency or current vaping cannabis behavior, lack of data on specific products, and lack of data on whether teens used specialized devices or e-cigarettes for cannabis vaping. However, the findings are consistent with studies on prevalence of cannabis vaping in other states such as Connecticut and California. “No studies to our knowledge have examined how adolescents who vape cannabis use other specific tobacco products (i.e., cigarettes, cigars, waterpipe, smokeless tobacco),” the researchers wrote.
The findings confirm that a large number of adolescents who use tobacco products have vaped cannabis as well, and this growing public health issue “is likely to affect and be affected by tobacco control and cannabis policies in states and at the federal level in the USA,” the researchers concluded.
“Increased research investigating how youth use e-cigarette devices for other purposes beyond vaping nicotine, like the current study, is needed,” they added.
The study was supported in part by the National Cancer Institute and the Food and Drug Administration’s Center for Tobacco Products. The researchers had no financial conflicts to disclose.
SOURCE: Kowitt SD et al. BMJ Open. 2019 Jun 13. doi: 10.1136/bmjopen-2018-028535.
and that practice is associated with cigars, waterpipe and e-cigarette use, findings from a survey of nearly 3,000 adolescents have shown.
“Although the prevalence of e-cigarette use among youth has increased dramatically in the past decade, little epidemiologic data exist on the prevalence of using e-cigarette devices or other specialised devices to vaporise (‘vape’) cannabis in the form of hash oil, tetrahydrocannabinol (THC) wax or oil, or dried cannabis buds or leaves,” wrote Sarah D. Kowitt, PhD, of the University of North Carolina, Chapel Hill, and colleagues. “This is surprising given that (1) cannabis (also referred to as marijuana) and e-cigarettes are the most commonly used substances by adolescents in the USA, (2) evidence exists that adolescents dual use both tobacco e-cigarettes and cannabis, and (3) longitudinal research suggests that use of e-cigarettes is associated with progression to use of cannabis.”
In a study published in BMJ Open, the researchers used data from the 2017 North Carolina Youth Tobacco Survey, a school-based survey of students in grades 6-12. The study population included 2,835 adolescents in grades 9-12.
Overall, 9.6% of students reported ever vaping cannabis. In multivariate analysis, cannabis vaping was significantly more likely among adolescents who reported using e-cigarettes (adjusted odds ratio 3.18), cigars (aOR 3.76), or water pipes (aOR 2.32) in the past 30 days, compared with peers who didn’t use tobacco.
The researchers found no significant association between smokeless tobacco use or traditional cigarette use in the past 30 days and vaping cannabis.
In a bivariate analysis, vaping cannabis was significantly more common among males vs. females (11% vs. 8.2%) and among non-Hispanic white students (11.3%), Hispanic students (10.5%), and other non-Hispanic students (11.8%) compared with non-Hispanic black students (5.0%).
In addition, prevalence of cannabis vaping increased with grade level, from 4.7% of 9th graders to 15.5% of 12th graders.
The health impacts of vaping cannabis are not well researched, but the researchers note that among the potential safety issues are earlier initiation of tobacco or cannabis use, concomitant tobacco and cannabis use, increased frequency of use or misuse of tobacco or cannabis, or increased potency of cannabis.
The results of the study were limited by several factors including the use of data only from the state of North Carolina, the lack of data on frequency or current vaping cannabis behavior, lack of data on specific products, and lack of data on whether teens used specialized devices or e-cigarettes for cannabis vaping. However, the findings are consistent with studies on prevalence of cannabis vaping in other states such as Connecticut and California. “No studies to our knowledge have examined how adolescents who vape cannabis use other specific tobacco products (i.e., cigarettes, cigars, waterpipe, smokeless tobacco),” the researchers wrote.
The findings confirm that a large number of adolescents who use tobacco products have vaped cannabis as well, and this growing public health issue “is likely to affect and be affected by tobacco control and cannabis policies in states and at the federal level in the USA,” the researchers concluded.
“Increased research investigating how youth use e-cigarette devices for other purposes beyond vaping nicotine, like the current study, is needed,” they added.
The study was supported in part by the National Cancer Institute and the Food and Drug Administration’s Center for Tobacco Products. The researchers had no financial conflicts to disclose.
SOURCE: Kowitt SD et al. BMJ Open. 2019 Jun 13. doi: 10.1136/bmjopen-2018-028535.
and that practice is associated with cigars, waterpipe and e-cigarette use, findings from a survey of nearly 3,000 adolescents have shown.
“Although the prevalence of e-cigarette use among youth has increased dramatically in the past decade, little epidemiologic data exist on the prevalence of using e-cigarette devices or other specialised devices to vaporise (‘vape’) cannabis in the form of hash oil, tetrahydrocannabinol (THC) wax or oil, or dried cannabis buds or leaves,” wrote Sarah D. Kowitt, PhD, of the University of North Carolina, Chapel Hill, and colleagues. “This is surprising given that (1) cannabis (also referred to as marijuana) and e-cigarettes are the most commonly used substances by adolescents in the USA, (2) evidence exists that adolescents dual use both tobacco e-cigarettes and cannabis, and (3) longitudinal research suggests that use of e-cigarettes is associated with progression to use of cannabis.”
In a study published in BMJ Open, the researchers used data from the 2017 North Carolina Youth Tobacco Survey, a school-based survey of students in grades 6-12. The study population included 2,835 adolescents in grades 9-12.
Overall, 9.6% of students reported ever vaping cannabis. In multivariate analysis, cannabis vaping was significantly more likely among adolescents who reported using e-cigarettes (adjusted odds ratio 3.18), cigars (aOR 3.76), or water pipes (aOR 2.32) in the past 30 days, compared with peers who didn’t use tobacco.
The researchers found no significant association between smokeless tobacco use or traditional cigarette use in the past 30 days and vaping cannabis.
In a bivariate analysis, vaping cannabis was significantly more common among males vs. females (11% vs. 8.2%) and among non-Hispanic white students (11.3%), Hispanic students (10.5%), and other non-Hispanic students (11.8%) compared with non-Hispanic black students (5.0%).
In addition, prevalence of cannabis vaping increased with grade level, from 4.7% of 9th graders to 15.5% of 12th graders.
The health impacts of vaping cannabis are not well researched, but the researchers note that among the potential safety issues are earlier initiation of tobacco or cannabis use, concomitant tobacco and cannabis use, increased frequency of use or misuse of tobacco or cannabis, or increased potency of cannabis.
The results of the study were limited by several factors including the use of data only from the state of North Carolina, the lack of data on frequency or current vaping cannabis behavior, lack of data on specific products, and lack of data on whether teens used specialized devices or e-cigarettes for cannabis vaping. However, the findings are consistent with studies on prevalence of cannabis vaping in other states such as Connecticut and California. “No studies to our knowledge have examined how adolescents who vape cannabis use other specific tobacco products (i.e., cigarettes, cigars, waterpipe, smokeless tobacco),” the researchers wrote.
The findings confirm that a large number of adolescents who use tobacco products have vaped cannabis as well, and this growing public health issue “is likely to affect and be affected by tobacco control and cannabis policies in states and at the federal level in the USA,” the researchers concluded.
“Increased research investigating how youth use e-cigarette devices for other purposes beyond vaping nicotine, like the current study, is needed,” they added.
The study was supported in part by the National Cancer Institute and the Food and Drug Administration’s Center for Tobacco Products. The researchers had no financial conflicts to disclose.
SOURCE: Kowitt SD et al. BMJ Open. 2019 Jun 13. doi: 10.1136/bmjopen-2018-028535.
FROM BMJ OPEN
Key clinical point: Use of tobacco products was significantly associated with cannabis vaping in teens.
Major finding: Approximately 10% of adolescents reported vaping cannabis.
Study details: The data come from a survey of 2,835 adolescents in North Carolina.
Disclosures: The study was supported in part by the National Cancer Institute and the FDA Center for Tobacco Products. The researchers had no financial conflicts to disclose.
Source: Kowitt SD et al. BMJ Open. 2019 Jun 13. doi: 10.1136/bmjopen-2018-028535.
Children with Down syndrome may need more screening for sleep-disordered breathing
because the condition frequently persists and recurs.
“Current screening recommendations to assess for SDB at a particular age may not be adequate in this population,” the authors of the study stated, adding that “persistence/recurrence of SDB is not easily predicted.”
The study, led by Joy Nehme, BSc, of Children’s Hospital of Eastern Ontario and the University of Ottawa, was published in Pediatric Pulmonology.
According to the study, research suggests that 43%-66% of children with Down syndrome have SDB, a category that encompasses sleep apnea (both obstructive and central) and hypoventilation. Those numbers are several times higher than the prevalence of SDB in children in the general population (1%-5%).
“Because SDB is associated with cardiometabolic and neurocognitive morbidity, its prompt and accurate diagnosis is important,” the researchers wrote. However, diagnosis requires a sleep study, which is not always performed although the American Academy of Pediatrics recommends children with Down syndrome undergo one by age 4.
Treatments include adenotonsillectomy (considered first-line), positive airway pressure, and lingual tonsillectomy.
The study aims to fill in gaps in knowledge about the condition over the long term since “there is little available literature on the trajectory of SDB in children and youth with Down syndrome over time.”
The researchers launched a retrospective study of 560 children with Down syndrome who were treated from 2004 to 2015 at Children’s Hospital of Eastern Ontario. Of those, 120 showed signs of SDB and underwent sleep studies (48% male, median age 6.6 years [range 4.5-10.5], median total apnea‐hypopnea index events per hour = 3.4 [1.6-10.8]).
Of the 120 children, 67 (56%) had obstructive-mixed SDB, 9 (8%) had central sleep apnea, and 5 (4%) had hypoventilation. The others (39, 32%) had no SDB.
Fifty-four children underwent at least two sleep studies during the period of the study, with at least one undergoing seven.
Researchers found weak, nonsignificant evidence that SDB persistence/occurrence varied by age (odds ratio per year = 1.15; 95% confidence interval, 0.96-1.41; P = .13).
As for treatment, adenotonsillectomy was most common, although “previous studies have ... shown that moderate to severe OSA in children with Down syndrome is likely to persist after a tonsillectomy.”
In regard to obstructive sleep apnea (OSA) specifically, the authors wrote, “our study ... showed that OSA‐SDB persisted or recurred in the vast majority of children. Further, persistence/recurrence could not be predicted by clinical features or SDB severity in our study. This, therefore, highlights the need for serial longitudinal screening for SDB in this population and for follow‐up PSG to ensure the success of treatment interventions.”
No study funding was reported. The study authors reported no disclosures.
SOURCE: Nehme J et al. Pediatr Pulmonol. 2019 Jun 6. doi: 10.1002/ppul.24380.
because the condition frequently persists and recurs.
“Current screening recommendations to assess for SDB at a particular age may not be adequate in this population,” the authors of the study stated, adding that “persistence/recurrence of SDB is not easily predicted.”
The study, led by Joy Nehme, BSc, of Children’s Hospital of Eastern Ontario and the University of Ottawa, was published in Pediatric Pulmonology.
According to the study, research suggests that 43%-66% of children with Down syndrome have SDB, a category that encompasses sleep apnea (both obstructive and central) and hypoventilation. Those numbers are several times higher than the prevalence of SDB in children in the general population (1%-5%).
“Because SDB is associated with cardiometabolic and neurocognitive morbidity, its prompt and accurate diagnosis is important,” the researchers wrote. However, diagnosis requires a sleep study, which is not always performed although the American Academy of Pediatrics recommends children with Down syndrome undergo one by age 4.
Treatments include adenotonsillectomy (considered first-line), positive airway pressure, and lingual tonsillectomy.
The study aims to fill in gaps in knowledge about the condition over the long term since “there is little available literature on the trajectory of SDB in children and youth with Down syndrome over time.”
The researchers launched a retrospective study of 560 children with Down syndrome who were treated from 2004 to 2015 at Children’s Hospital of Eastern Ontario. Of those, 120 showed signs of SDB and underwent sleep studies (48% male, median age 6.6 years [range 4.5-10.5], median total apnea‐hypopnea index events per hour = 3.4 [1.6-10.8]).
Of the 120 children, 67 (56%) had obstructive-mixed SDB, 9 (8%) had central sleep apnea, and 5 (4%) had hypoventilation. The others (39, 32%) had no SDB.
Fifty-four children underwent at least two sleep studies during the period of the study, with at least one undergoing seven.
Researchers found weak, nonsignificant evidence that SDB persistence/occurrence varied by age (odds ratio per year = 1.15; 95% confidence interval, 0.96-1.41; P = .13).
As for treatment, adenotonsillectomy was most common, although “previous studies have ... shown that moderate to severe OSA in children with Down syndrome is likely to persist after a tonsillectomy.”
In regard to obstructive sleep apnea (OSA) specifically, the authors wrote, “our study ... showed that OSA‐SDB persisted or recurred in the vast majority of children. Further, persistence/recurrence could not be predicted by clinical features or SDB severity in our study. This, therefore, highlights the need for serial longitudinal screening for SDB in this population and for follow‐up PSG to ensure the success of treatment interventions.”
No study funding was reported. The study authors reported no disclosures.
SOURCE: Nehme J et al. Pediatr Pulmonol. 2019 Jun 6. doi: 10.1002/ppul.24380.
because the condition frequently persists and recurs.
“Current screening recommendations to assess for SDB at a particular age may not be adequate in this population,” the authors of the study stated, adding that “persistence/recurrence of SDB is not easily predicted.”
The study, led by Joy Nehme, BSc, of Children’s Hospital of Eastern Ontario and the University of Ottawa, was published in Pediatric Pulmonology.
According to the study, research suggests that 43%-66% of children with Down syndrome have SDB, a category that encompasses sleep apnea (both obstructive and central) and hypoventilation. Those numbers are several times higher than the prevalence of SDB in children in the general population (1%-5%).
“Because SDB is associated with cardiometabolic and neurocognitive morbidity, its prompt and accurate diagnosis is important,” the researchers wrote. However, diagnosis requires a sleep study, which is not always performed although the American Academy of Pediatrics recommends children with Down syndrome undergo one by age 4.
Treatments include adenotonsillectomy (considered first-line), positive airway pressure, and lingual tonsillectomy.
The study aims to fill in gaps in knowledge about the condition over the long term since “there is little available literature on the trajectory of SDB in children and youth with Down syndrome over time.”
The researchers launched a retrospective study of 560 children with Down syndrome who were treated from 2004 to 2015 at Children’s Hospital of Eastern Ontario. Of those, 120 showed signs of SDB and underwent sleep studies (48% male, median age 6.6 years [range 4.5-10.5], median total apnea‐hypopnea index events per hour = 3.4 [1.6-10.8]).
Of the 120 children, 67 (56%) had obstructive-mixed SDB, 9 (8%) had central sleep apnea, and 5 (4%) had hypoventilation. The others (39, 32%) had no SDB.
Fifty-four children underwent at least two sleep studies during the period of the study, with at least one undergoing seven.
Researchers found weak, nonsignificant evidence that SDB persistence/occurrence varied by age (odds ratio per year = 1.15; 95% confidence interval, 0.96-1.41; P = .13).
As for treatment, adenotonsillectomy was most common, although “previous studies have ... shown that moderate to severe OSA in children with Down syndrome is likely to persist after a tonsillectomy.”
In regard to obstructive sleep apnea (OSA) specifically, the authors wrote, “our study ... showed that OSA‐SDB persisted or recurred in the vast majority of children. Further, persistence/recurrence could not be predicted by clinical features or SDB severity in our study. This, therefore, highlights the need for serial longitudinal screening for SDB in this population and for follow‐up PSG to ensure the success of treatment interventions.”
No study funding was reported. The study authors reported no disclosures.
SOURCE: Nehme J et al. Pediatr Pulmonol. 2019 Jun 6. doi: 10.1002/ppul.24380.
FROM PEDIATRIC PULMONOLOGY
Key clinical point: Sleep-disordered breathing (SDB) can be persistent and recurrent in children with Down syndrome, and long-term monitoring is warranted.
Major finding: SDB persistence/recurrence did not vary by age (odds ratio per year = 1.15; 95% confidence interval, 0.96-1.41; P = .13).
Study details: Retrospective cohort analysis of 120 children with Down syndrome tested via sleep study at least once for SDB (48% male, median age 6.6 years).
Disclosures: No study funding or author disclosures were reported.
Source: Nehme J et al. Pediatr Pulmonol. 2019 Jun 6. doi: 10.1002/ppul.24380.
COPD rates reflect current smoking prevalence
, according to a Centers for Disease Control and Prevention analysis of respondents to a behavioral risk factor survey.
“Population-based strategies for smoking prevention and control have the potential to decrease the prevalence of COPD in the United States,” wrote Anne G. Wheaton, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and coauthors. The study was published in the Morbidity and Mortality Weekly Report.
Dr. Wheaton and her fellow researchers analyzed data from 418,378 adult respondents to the 2017 Behavioral Risk Factor Surveillance System survey. Responses came from all 50 states and Washington, D.C.; respondents who had smoked less than 100 lifetime cigarettes were categorized as “never smoked,” while those who had smoked at least 100 cigarettes but no longer smoked were categorized as “former smokers.” Anyone who had smoked at least 100 cigarettes and currently smoked was categorized as a “current smoker.”
The age-adjusted prevalence of COPD among U.S. adults was 6.2% (95% confidence interval, 6.0%-6.3%) in 2017. Current cigarette smokers had a prevalence of 15.2% (95% CI, 14.7%-15.7%); this dipped to 7.6% (95% CI, 7.3%-8.0%) among former smokers and 2.8% (95% CI, 2.7%-2.9%) among adults who had never smoked. Patterns were visible within states: Current smokers had a state-level prevalence of COPD that was strongly correlated with state-level current smoking prevalence (Pearson correlation coefficient, 0.69; P less than .001). State-level COPD prevalence among former smokers (Pearson correlation coefficient, 0.71; P less than .001) and those who never smoked (Pearson correlation coefficient, 0.64; P less than .001) were also strongly correlated with the current smoking prevalence, indicating secondhand smoke as a risk factor for COPD.
The coauthors acknowledged the study’s limitations, including relying on self-reporting for both COPD and smoking status. They also noted that there was no way to measure exposure to secondhand smoke, other indoor or outdoor air pollutants, or respiratory infection history, “all of which might contribute to COPD risk.”
No conflicts of interest were reported.
SOURCE: Wheaton AG et al. MMWR Morb Mortal Wkly Rep. 2019 Jun 21;68(24):533-8.
, according to a Centers for Disease Control and Prevention analysis of respondents to a behavioral risk factor survey.
“Population-based strategies for smoking prevention and control have the potential to decrease the prevalence of COPD in the United States,” wrote Anne G. Wheaton, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and coauthors. The study was published in the Morbidity and Mortality Weekly Report.
Dr. Wheaton and her fellow researchers analyzed data from 418,378 adult respondents to the 2017 Behavioral Risk Factor Surveillance System survey. Responses came from all 50 states and Washington, D.C.; respondents who had smoked less than 100 lifetime cigarettes were categorized as “never smoked,” while those who had smoked at least 100 cigarettes but no longer smoked were categorized as “former smokers.” Anyone who had smoked at least 100 cigarettes and currently smoked was categorized as a “current smoker.”
The age-adjusted prevalence of COPD among U.S. adults was 6.2% (95% confidence interval, 6.0%-6.3%) in 2017. Current cigarette smokers had a prevalence of 15.2% (95% CI, 14.7%-15.7%); this dipped to 7.6% (95% CI, 7.3%-8.0%) among former smokers and 2.8% (95% CI, 2.7%-2.9%) among adults who had never smoked. Patterns were visible within states: Current smokers had a state-level prevalence of COPD that was strongly correlated with state-level current smoking prevalence (Pearson correlation coefficient, 0.69; P less than .001). State-level COPD prevalence among former smokers (Pearson correlation coefficient, 0.71; P less than .001) and those who never smoked (Pearson correlation coefficient, 0.64; P less than .001) were also strongly correlated with the current smoking prevalence, indicating secondhand smoke as a risk factor for COPD.
The coauthors acknowledged the study’s limitations, including relying on self-reporting for both COPD and smoking status. They also noted that there was no way to measure exposure to secondhand smoke, other indoor or outdoor air pollutants, or respiratory infection history, “all of which might contribute to COPD risk.”
No conflicts of interest were reported.
SOURCE: Wheaton AG et al. MMWR Morb Mortal Wkly Rep. 2019 Jun 21;68(24):533-8.
, according to a Centers for Disease Control and Prevention analysis of respondents to a behavioral risk factor survey.
“Population-based strategies for smoking prevention and control have the potential to decrease the prevalence of COPD in the United States,” wrote Anne G. Wheaton, PhD, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion and coauthors. The study was published in the Morbidity and Mortality Weekly Report.
Dr. Wheaton and her fellow researchers analyzed data from 418,378 adult respondents to the 2017 Behavioral Risk Factor Surveillance System survey. Responses came from all 50 states and Washington, D.C.; respondents who had smoked less than 100 lifetime cigarettes were categorized as “never smoked,” while those who had smoked at least 100 cigarettes but no longer smoked were categorized as “former smokers.” Anyone who had smoked at least 100 cigarettes and currently smoked was categorized as a “current smoker.”
The age-adjusted prevalence of COPD among U.S. adults was 6.2% (95% confidence interval, 6.0%-6.3%) in 2017. Current cigarette smokers had a prevalence of 15.2% (95% CI, 14.7%-15.7%); this dipped to 7.6% (95% CI, 7.3%-8.0%) among former smokers and 2.8% (95% CI, 2.7%-2.9%) among adults who had never smoked. Patterns were visible within states: Current smokers had a state-level prevalence of COPD that was strongly correlated with state-level current smoking prevalence (Pearson correlation coefficient, 0.69; P less than .001). State-level COPD prevalence among former smokers (Pearson correlation coefficient, 0.71; P less than .001) and those who never smoked (Pearson correlation coefficient, 0.64; P less than .001) were also strongly correlated with the current smoking prevalence, indicating secondhand smoke as a risk factor for COPD.
The coauthors acknowledged the study’s limitations, including relying on self-reporting for both COPD and smoking status. They also noted that there was no way to measure exposure to secondhand smoke, other indoor or outdoor air pollutants, or respiratory infection history, “all of which might contribute to COPD risk.”
No conflicts of interest were reported.
SOURCE: Wheaton AG et al. MMWR Morb Mortal Wkly Rep. 2019 Jun 21;68(24):533-8.
FROM MMWR
Vaping among teens increased significantly from 2017 to 2018
according to data from national cross-sectional surveys.
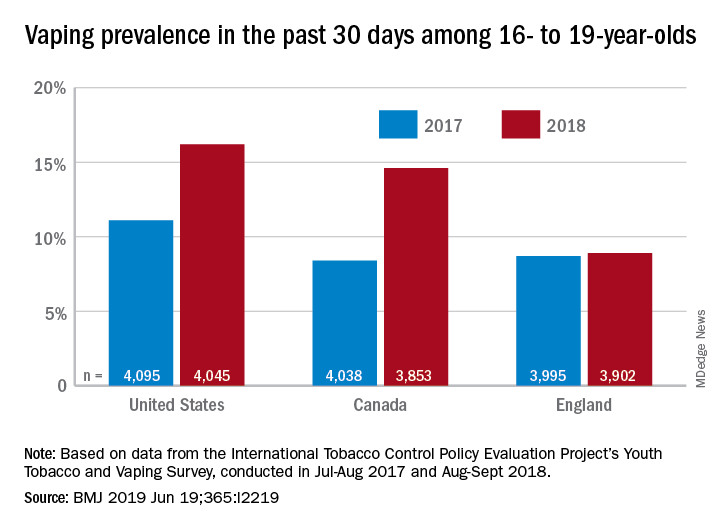
The prevalence of vaping in the past 30 days rose from 11% to 16% in the United States and from 8% to 14.6% in Canada, while use in England showed an nonsignificant increase of 8.7% to 8.9%, David Hammond, PhD, of the University of Waterloo (Canada) and associates said in the BMJ.
Embedded in those U.S. and Canadian increases is the recent evolution of the vaping market brought about by “the growth of JUUL e-cigarettes and similar products [that use] benzoic acid and nicotine salt technology to deliver higher concentrations of nicotine than conventional e-cigarettes,” they explained.
In England, the JUUL system is limited to less than half the nicotine concentration, at 20 mg/mL, compared with more than 50 mg/mL in the United States and Canada, and it was not available at all types of retail outlets at the time of the surveys. That situation changed in March 2019, when the company expanded to convenience stores, the investigators noted.
In the United States, JUUL was the second-most popular product among past–30-day vapers who had a usual brand in 2017, with 9% reporting use. In 2018, JUUL was the most popular brand and use was up to 28%. In Canada, the brand was not among the top five in 2017, but was third in 2018 at 10% in those who reported vaping in the past 30 days. The leading Canadian brand in 2018 was Smok, which released a nicotine-salt version in March of 2018, Dr. Hammond and associates reported.
“Before 2018, there was relatively little evidence of regular vaping among adolescents that might be indicative of nicotine addiction; however, the emergence of JUUL and nicotine salt–based products might signal a change,” they wrote.
The International Tobacco Control Policy Evaluation Project’s Youth Tobacco and Vaping Survey was conducted online in each country in two waves – July to August 2017 and August to September 2018 – with a sample size of approximately 12,000 for each.
The study was funded by the U.S. National Institutes of Health. Dr. Hammond is supported by a Canadian Institutes of Health Research–Public Health Agency of Canada applied public health research chair. The investigators said that they had no other financial disclosures to report, but several have served as paid witnesses in legal challenges against tobacco companies.
SOURCE: Hammond D et al. BMJ 2019 Jun 19. doi: 10.1136/bmj.l2219.
according to data from national cross-sectional surveys.

The prevalence of vaping in the past 30 days rose from 11% to 16% in the United States and from 8% to 14.6% in Canada, while use in England showed an nonsignificant increase of 8.7% to 8.9%, David Hammond, PhD, of the University of Waterloo (Canada) and associates said in the BMJ.
Embedded in those U.S. and Canadian increases is the recent evolution of the vaping market brought about by “the growth of JUUL e-cigarettes and similar products [that use] benzoic acid and nicotine salt technology to deliver higher concentrations of nicotine than conventional e-cigarettes,” they explained.
In England, the JUUL system is limited to less than half the nicotine concentration, at 20 mg/mL, compared with more than 50 mg/mL in the United States and Canada, and it was not available at all types of retail outlets at the time of the surveys. That situation changed in March 2019, when the company expanded to convenience stores, the investigators noted.
In the United States, JUUL was the second-most popular product among past–30-day vapers who had a usual brand in 2017, with 9% reporting use. In 2018, JUUL was the most popular brand and use was up to 28%. In Canada, the brand was not among the top five in 2017, but was third in 2018 at 10% in those who reported vaping in the past 30 days. The leading Canadian brand in 2018 was Smok, which released a nicotine-salt version in March of 2018, Dr. Hammond and associates reported.
“Before 2018, there was relatively little evidence of regular vaping among adolescents that might be indicative of nicotine addiction; however, the emergence of JUUL and nicotine salt–based products might signal a change,” they wrote.
The International Tobacco Control Policy Evaluation Project’s Youth Tobacco and Vaping Survey was conducted online in each country in two waves – July to August 2017 and August to September 2018 – with a sample size of approximately 12,000 for each.
The study was funded by the U.S. National Institutes of Health. Dr. Hammond is supported by a Canadian Institutes of Health Research–Public Health Agency of Canada applied public health research chair. The investigators said that they had no other financial disclosures to report, but several have served as paid witnesses in legal challenges against tobacco companies.
SOURCE: Hammond D et al. BMJ 2019 Jun 19. doi: 10.1136/bmj.l2219.
according to data from national cross-sectional surveys.

The prevalence of vaping in the past 30 days rose from 11% to 16% in the United States and from 8% to 14.6% in Canada, while use in England showed an nonsignificant increase of 8.7% to 8.9%, David Hammond, PhD, of the University of Waterloo (Canada) and associates said in the BMJ.
Embedded in those U.S. and Canadian increases is the recent evolution of the vaping market brought about by “the growth of JUUL e-cigarettes and similar products [that use] benzoic acid and nicotine salt technology to deliver higher concentrations of nicotine than conventional e-cigarettes,” they explained.
In England, the JUUL system is limited to less than half the nicotine concentration, at 20 mg/mL, compared with more than 50 mg/mL in the United States and Canada, and it was not available at all types of retail outlets at the time of the surveys. That situation changed in March 2019, when the company expanded to convenience stores, the investigators noted.
In the United States, JUUL was the second-most popular product among past–30-day vapers who had a usual brand in 2017, with 9% reporting use. In 2018, JUUL was the most popular brand and use was up to 28%. In Canada, the brand was not among the top five in 2017, but was third in 2018 at 10% in those who reported vaping in the past 30 days. The leading Canadian brand in 2018 was Smok, which released a nicotine-salt version in March of 2018, Dr. Hammond and associates reported.
“Before 2018, there was relatively little evidence of regular vaping among adolescents that might be indicative of nicotine addiction; however, the emergence of JUUL and nicotine salt–based products might signal a change,” they wrote.
The International Tobacco Control Policy Evaluation Project’s Youth Tobacco and Vaping Survey was conducted online in each country in two waves – July to August 2017 and August to September 2018 – with a sample size of approximately 12,000 for each.
The study was funded by the U.S. National Institutes of Health. Dr. Hammond is supported by a Canadian Institutes of Health Research–Public Health Agency of Canada applied public health research chair. The investigators said that they had no other financial disclosures to report, but several have served as paid witnesses in legal challenges against tobacco companies.
SOURCE: Hammond D et al. BMJ 2019 Jun 19. doi: 10.1136/bmj.l2219.
FROM THE BMJ
Key clinical point: Recent increases in vaping prevalence among teens “might be indicative of nicotine addiction.”
Major finding: Vaping prevalence increased from 11% to 16% in the United States and from 8% to 14.6% in Canada.
Study details: Two waves of a national, cross-sectional survey that included approximately 12,000 respondents each.
Disclosures: The study was funded by the U.S. National Institutes of Health. Dr. Hammond is supported by a Canadian Institutes of Health Research–Public Health Agency of Canada applied public health research chair. The investigators said that they had no other financial disclosures to report, but several have served as paid witnesses in legal challenges against tobacco companies.
Source: Hammond D et al. BMJ. 2019 Jun 19. doi: 10.1136/bmj.l2219.
Experts agree on routine lung disease screening in systemic sclerosis
MADRID – The for early detection, monitoring, and, when warranted, treatment, Anna-Maria Hoffmann-Vold, MD, PhD, reported at the European Congress of Rheumatology.
“Everyone with systemic sclerosis needs to be screened because this is the most important risk factor for ILD,” said Dr. Hoffmann-Vold, a clinical scientist in the division of rheumatology at the University of Oslo and head of scleroderma research at Oslo University Hospital.
Although the frequency of screening is not specified based on the opinion that this should be based on risk factors and other clinical characteristics, there was unanimous agreement that lung function tests do not represent an adequate screening tool or method for assessing ILD severity. Rather, the recommendations make clear that lung function studies are adjunctive to high-resolution computed tomography (HRCT).
“HRCT is the primary tool for evaluating ILD, but there was 100% agreement that assessment should include more than one measure, including lung function tests and clinical assessment,” Dr. Hoffmann-Vold reported.
There was a strong opinion that the numerous potential biomarkers described for ILD, although promising, are not yet ready for clinical use.
In developing these new recommendations, 95 potential statements were considered by the panel of 27 rheumatologists, pulmonologists, and others with experience in this field. A Delphi process was used for members of the panel to identify areas of agreement to produce consensus statements.
The result has been more than 50 statements issued in six major domains. These include statements on risk factors, appropriate methodology for diagnosis and severity assessment, when to initiate therapy, and when and how to initiate treatment escalation.
“We want to increase clinician awareness and provide standardized guidance for evaluating patients for the presence and medical management of ILD-SSc,” Dr. Hoffmann-Vold explained.
ILD occurs in about half of all patients with systemic sclerosis. Among these, approximately one out of three will experience lung disease progression. Although these high prevalence rates are well recognized and associated with high morbidity and mortality, Dr. Hoffmann-Vold said that there has been uncertainty about how to screen systemic sclerosis patients for ILD and what steps to take when it was found. It is this uncertainty that prompted the present initiative.
The consensus recommendations are an initial step to guide clinicians, but Dr. Hoffmann-Vold noted that the many statements are based on expert opinion, suggesting more studies are needed to compare strategies for objective severity grading and prediction of which patients are most at risk for ILD progression.
“There are still huge knowledge gaps we need to fill,” she stated. Still, she believes these recommendations represent progress in this field. While they are likely “to increase the standard of care” for those who develop ILD-SSc, they also have identified where to concentrate further research.
Dr. Hoffmann-Vold reported financial relationships with Actelion, Boehringer Ingelheim, and GlaxoSmithKline.
SOURCE: Hoffmann-Vold A-M et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):104, Abstract OPO064, doi: 10.1136/annrheumdis-2019-eular.3225.
MADRID – The for early detection, monitoring, and, when warranted, treatment, Anna-Maria Hoffmann-Vold, MD, PhD, reported at the European Congress of Rheumatology.
“Everyone with systemic sclerosis needs to be screened because this is the most important risk factor for ILD,” said Dr. Hoffmann-Vold, a clinical scientist in the division of rheumatology at the University of Oslo and head of scleroderma research at Oslo University Hospital.
Although the frequency of screening is not specified based on the opinion that this should be based on risk factors and other clinical characteristics, there was unanimous agreement that lung function tests do not represent an adequate screening tool or method for assessing ILD severity. Rather, the recommendations make clear that lung function studies are adjunctive to high-resolution computed tomography (HRCT).
“HRCT is the primary tool for evaluating ILD, but there was 100% agreement that assessment should include more than one measure, including lung function tests and clinical assessment,” Dr. Hoffmann-Vold reported.
There was a strong opinion that the numerous potential biomarkers described for ILD, although promising, are not yet ready for clinical use.
In developing these new recommendations, 95 potential statements were considered by the panel of 27 rheumatologists, pulmonologists, and others with experience in this field. A Delphi process was used for members of the panel to identify areas of agreement to produce consensus statements.
The result has been more than 50 statements issued in six major domains. These include statements on risk factors, appropriate methodology for diagnosis and severity assessment, when to initiate therapy, and when and how to initiate treatment escalation.
“We want to increase clinician awareness and provide standardized guidance for evaluating patients for the presence and medical management of ILD-SSc,” Dr. Hoffmann-Vold explained.
ILD occurs in about half of all patients with systemic sclerosis. Among these, approximately one out of three will experience lung disease progression. Although these high prevalence rates are well recognized and associated with high morbidity and mortality, Dr. Hoffmann-Vold said that there has been uncertainty about how to screen systemic sclerosis patients for ILD and what steps to take when it was found. It is this uncertainty that prompted the present initiative.
The consensus recommendations are an initial step to guide clinicians, but Dr. Hoffmann-Vold noted that the many statements are based on expert opinion, suggesting more studies are needed to compare strategies for objective severity grading and prediction of which patients are most at risk for ILD progression.
“There are still huge knowledge gaps we need to fill,” she stated. Still, she believes these recommendations represent progress in this field. While they are likely “to increase the standard of care” for those who develop ILD-SSc, they also have identified where to concentrate further research.
Dr. Hoffmann-Vold reported financial relationships with Actelion, Boehringer Ingelheim, and GlaxoSmithKline.
SOURCE: Hoffmann-Vold A-M et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):104, Abstract OPO064, doi: 10.1136/annrheumdis-2019-eular.3225.
MADRID – The for early detection, monitoring, and, when warranted, treatment, Anna-Maria Hoffmann-Vold, MD, PhD, reported at the European Congress of Rheumatology.
“Everyone with systemic sclerosis needs to be screened because this is the most important risk factor for ILD,” said Dr. Hoffmann-Vold, a clinical scientist in the division of rheumatology at the University of Oslo and head of scleroderma research at Oslo University Hospital.
Although the frequency of screening is not specified based on the opinion that this should be based on risk factors and other clinical characteristics, there was unanimous agreement that lung function tests do not represent an adequate screening tool or method for assessing ILD severity. Rather, the recommendations make clear that lung function studies are adjunctive to high-resolution computed tomography (HRCT).
“HRCT is the primary tool for evaluating ILD, but there was 100% agreement that assessment should include more than one measure, including lung function tests and clinical assessment,” Dr. Hoffmann-Vold reported.
There was a strong opinion that the numerous potential biomarkers described for ILD, although promising, are not yet ready for clinical use.
In developing these new recommendations, 95 potential statements were considered by the panel of 27 rheumatologists, pulmonologists, and others with experience in this field. A Delphi process was used for members of the panel to identify areas of agreement to produce consensus statements.
The result has been more than 50 statements issued in six major domains. These include statements on risk factors, appropriate methodology for diagnosis and severity assessment, when to initiate therapy, and when and how to initiate treatment escalation.
“We want to increase clinician awareness and provide standardized guidance for evaluating patients for the presence and medical management of ILD-SSc,” Dr. Hoffmann-Vold explained.
ILD occurs in about half of all patients with systemic sclerosis. Among these, approximately one out of three will experience lung disease progression. Although these high prevalence rates are well recognized and associated with high morbidity and mortality, Dr. Hoffmann-Vold said that there has been uncertainty about how to screen systemic sclerosis patients for ILD and what steps to take when it was found. It is this uncertainty that prompted the present initiative.
The consensus recommendations are an initial step to guide clinicians, but Dr. Hoffmann-Vold noted that the many statements are based on expert opinion, suggesting more studies are needed to compare strategies for objective severity grading and prediction of which patients are most at risk for ILD progression.
“There are still huge knowledge gaps we need to fill,” she stated. Still, she believes these recommendations represent progress in this field. While they are likely “to increase the standard of care” for those who develop ILD-SSc, they also have identified where to concentrate further research.
Dr. Hoffmann-Vold reported financial relationships with Actelion, Boehringer Ingelheim, and GlaxoSmithKline.
SOURCE: Hoffmann-Vold A-M et al. Ann Rheum Dis. Jun 2019;78(Suppl 2):104, Abstract OPO064, doi: 10.1136/annrheumdis-2019-eular.3225.
REPORTING FROM EULAR 2019 Congress
Adjuvant corticosteroids in hospitalized patients with CAP
When is it appropriate to treat?
Case
A 55-year-old male with a history of tobacco use disorder presents with 2 days of productive cough, fever, chills, and mild shortness of breath. T 38.4, HR 89, RR 32, BP 100/65, 02 sat 86% on room air. Exam reveals diminished breath sounds and positive egophony over the right lung base. WBC is 16,000 and BUN 22. Chest x-ray reveals right lower lobe consolidation. He is given ceftriaxone and azithromycin.
Brief overview of the issue
Community-acquired pneumonia (CAP) is the leading cause of infectious disease–related death in the United States. Mortality associated with CAP is estimated at 57,000 deaths annually and occurs largely in patients requiring hospitalization.1 The 30-day mortality rate in patients who are hospitalized for CAP is approximately 10%-12%.2 After discharge from the hospital, about 18% of patients are readmitted within 30 days.3 An excessive inflammatory cytokine response may be a major contributor to the high mortality rate in CAP and systemic corticosteroids may reduce the inflammatory response from the infection by down-regulating this proinflammatory cytokine production.
Almost all of the major decisions regarding management of CAP, including diagnostic and treatment issues, revolve around the initial assessment of severity of illness. Between 40% and 60% of patients who present to the emergency department with CAP are admitted4 and approximately 10% of hospitalized patients with CAP require ICU admission.5 Validated instruments such as CURB-65, the pneumonia severity index (PSI), and guidelines from the Infectious Diseases Society of America (IDSA)/American Thoracic Society (ATS) may predict severity of illness but should always be supplemented with physician determination of subjective factors when determining treatment.5 Although there is no census definition of severe pneumonia, studies generally define the condition in the following order of preference: PSI score of IV or V followed by CURB-65 score of two or greater. If these scoring modalities were not available, the IDSA/ATS criteria was used (1 major or 3 minor). Others define severe CAP as pneumonia requiring supportive therapy within a critical care environment.
Overview of the data
The use of corticosteroids in addition to antibiotics in the treatment of CAP was proposed as early as the 1950s and yet only in the last decade has the body of evidence grown significantly.5 There is evidence that corticosteroids suppress inflammation without acutely impairing the immune response as evidenced by a rapid and sustained decrease in circulating inflammatory markers such as C-reactive protein and interleukin 6 and no effect on the anti-inflammatory interleukin 10.6 Within the last year, three meta-analyses, one by the Cochrane Library, one by the IDSA, and a third in the American Journal of Emergency Medicine, addressed the role of routine low dose (20-60 mg of prednisone or equivalent), short-course (3-7 days) systemic corticosteroids in hospitalized patients with CAP of varying severities.
The Cochrane meta-analysis, the largest and most recent dataset, included 13 trials with a combined 1,954 adult patients and found that corticosteroids significantly lowered mortality in hospitalized patients with severe CAP with a number needed to treat of 19.7 In this group with severe CAP, mortality was lowered from 13% to 8% and there were significantly fewer episodes of respiratory failure and shock with the addition of corticosteroids. No effect was seen on mortality in patients with less severe CAP. In those patients who received adjuvant corticosteroids, length of hospital stay decreased by 3 days, regardless of CAP severity.7
The IDSA meta-analysis was similar and included 1,506 patients from six trials.8 In contrast with the Cochrane study, this analysis found corticosteroids did not significantly lower mortality in patients with severe CAP but did reduce time to clinical stability and length of hospital stay by over 1 day. This study also found significantly more CAP-related, 30-day rehospitalizations (5% vs. 3%; defined as recurrent pneumonia, other infection, pleuritic pain, adverse cardiovascular event, or diarrhea) in patient with non-severe CAP treated with corticosteroids.
The study in the American Journal of Emergency Medicine involved ten trials involving more than 700 patients admitted with severe CAP and found in-hospital mortality was cut in half (RR 0.49) and length of hospital stay was reduced when patients were treated with corticosteroids in addition to standard antibiotic therapy.9
In 2015, two randomized clinical trials, one in the Lancet and the other in JAMA, and a meta-analysis in Annals of Internal Medicine assessed the impact of adjuvant corticosteroids in the treatment of hospitalized patients with CAP. The Lancet study of 785 patients hospitalized with CAP of any severity found shortened time to clinical stability (3.0 vs. 4.4 days) as defined by stable vital signs, improved oral intake, and normalized mental status for greater than 24 hours when oral prednisone 50 mg for 7 days was added to standard therapy.10 Patients in the treatment group were also discharged 1 day earlier compared with the placebo control group.
The study in JAMA was small, with only 100 patients at three teaching hospitals in Spain, but found that patients hospitalized with severe CAP and high inflammatory response based on elevated C-reactive protein were less likely to experience a treatment failure, defined as shock, mechanical ventilation, death, or radiographic progression, when intravenous methylprednisolone 0.5 mg/kg was added to standard antibiotic therapy.11
Finally, the meta-analysis in Annals of Internal Medicine assessed 13 randomized controlled placebo trials of 1,974 patients and found that adjuvant corticosteroids in a dose of 20-60 mg of prednisone or equivalent total daily dose significantly lowered mortality in patients with severe CAP and incidence of respiratory distress syndrome, and need for mechanical ventilation in all patients hospitalized with CAP.12
Importantly, nearly all of the described studies showed a significantly higher incidence of hyperglycemia in patients who received corticosteroids.
Application of the data to our patients
The benefit of adjuvant corticosteroids is most clear in hospitalized patients with severe CAP. Recent, strong evidence supports decreased mortality, decreased time to clinical stability, and decreased length of stay in our patient, with severe CAP, if treated with 20-60 mg of prednisone or equivalent total daily dose for 3-7 days. For patients with non-severe CAP, we suggest taking a risk-benefit approach based on other comorbidities, as the risk for CAP-related rehospitalizations may be higher.
For patients with underlying lung disease, specifically COPD or reactive airway disease, we suggest a low threshold for adding corticosteroids. This approach is more anecdotal than data driven, though corticosteroids are a mainstay of treatment for COPD exacerbations and a retrospective analysis of more than 20,000 hospitalized children with CAP and wheezing revealed decreased length of stay with corticosteroid treatment.13 Furthermore, a number of the studies described above included patients with COPD. Our threshold rises significantly in patients with poorly controlled diabetes mellitus.
Bottom line
For patients hospitalized with severe community-acquired pneumonia, recent evidence supports the use of low dose, short-course, systemic corticosteroids in addition to standard therapy.
Dr. Parsons is an assistant professor at the University of Virginia and a hospitalist at the University of Virginia Medical Center in Charlottesville, Va. Dr. Miller is an assistant professor at the University of Virginia and a hospitalist at the University of Virginia Medical Center. Dr. Hoke is Associate Director of Hospital Medicine and Faculty Development at the University of Virginia.
References
1. Ramirez J et al. Adults hospitalized with pneumonia in the United States: Incidence, epidemiology, and mortality. Clin Infect Dis. 2017 Dec 1:65(11):1806-12.
2. Musher D et al. Community-acquired pneumonia: Review article. N Engl J Med. 2014 Oct 23;371:1619-28.
3. Wunderink R et al. Community-aquired pneumonia: Clinical practice. N Engl J Med. 2014 Feb 6;370:543-51.
4. Mandell L et al. Infectious Diseases Society America/American Thoracic Society Consensus Guidelines on the Management of Community-Acquired Pneumonia in Adults. Clin Infect Dis. 2007;44:S27-72.
5. Wagner HN et al. The effect of hydrocortisone upon the course of pneumococcal pneumonia treated with penicillin. Bull Johns Hopkins Hosp. 1956;98:197-215.
6. Polverino E et al. Systemic corticosteroids for community-acquired pneumonia: Reasons for use and lack of benefit on outcome. Respirology. 2013. Feb;18(2):263-71 (https://doi.org/10.1111/resp.12013).
7. Stern A et al. Corticosteroids for pneumonia. Cochrane Database Syst Rev. 2017 Dec 13; 12:CD007720 (https://doi.org/10.1002/14651858.CD007720.pub3).
8. Briel M et al. Corticosteroids in patients hospitalized with community-acquired pneumonia: Systematic review and individual patient data meta-analysis. Clin Infect Dis. 2018 Feb 1;66:346 (https://doi.org/10.1093/cid/cix801).
9. Wu W-F et al. Efficacy of corticosteroid treatment for severe community-acquired pneumonia: A meta-analysis. Am J Emerg Med. 2017 Jul 15; [e-pub] (http://dx.doi.org/10.1016/j.ajem.2017.07.050).
10. Blum CA et al. Adjunct prednisone therapy for patients with community-acquired pneumonia: A multicentre, double-blind, randomised, placebo-controlled trial. Lancet 2015 Jan 18; [e-pub ahead of print] (http://dx.doi.org/10.1016/S0140-6736[14]62447-8).
11. Torres A et al. Effect of corticosteroids on treatment failure among hospitalized patients with severe community-acquired pneumonia and high inflammatory response: A randomized clinical trial. JAMA 2015 Feb 17; 313:677 (http://dx.doi.org/10.1001/jama.2015.88).
12. Siemieniuk RAC et al. Corticosteroid therapy for patients hospitalized with community-acquired pneumonia: A systematic review and meta-analysis. Ann Intern Med. 2015 Oct 6;163:519 (http://dx.doi.org/10.7326/M15-0715).
13. Simon LH et al. Management of community-acquired pneumonia in hospitalized children. Current Treat Options Peds (2015) 1:59 (https://doi:.org/10.1007/s40746-014-0011-3).
Key points
• For patients hospitalized with severe CAP, recent evidence supports the use of low-dose, short-course, systemic corticosteroids in addition to standard therapy.
• Among hospitalized patients with non-severe CAP, the benefit is not well defined. Studies suggest these patients may benefit from reduced time to clinical stability and reduced length of hospital stay. However, they may be at risk for significantly more CAP-related, 30-day rehospitalizations and hyperglycemia.
• Further prospective, randomized controlled studies are needed to further delineate the patient population who will most benefit from adjunctive corticosteroids use, including dose and duration of treatment.
QUIZ
Which of the following is FALSE regarding community acquired pneumonia?
A. CAP is the leading cause of infectious disease related death in the United States.
B. An excessive inflammatory cytokine response may contribute to the high mortality rate in CAP.
C. Adjunctive steroid therapy has been shown to decrease mortality in all patients with CAP.
D. Hyperglycemia occurs more frequently in patients receiving steroid therapy.
E. Reasons to avoid adjunctive steroid therapy in CAP include low risk for mortality, poorly controlled diabetes, suspected viral or fungal etiology, and elevated risk for gastrointestinal bleeding.
ANSWER: C. The patient population that may benefit most from the use of adjuvant corticosteroids is poorly defined. However, in patients with severe pneumonia, the use of adjuvant steroids has been shown to decrease mortality, time to clinical stability, and length of stay.
Additional reading
Siemieniuk RAC et al. Corticosteroid therapy for patients hospitalized with community-acquired pneumonia: A systematic review and meta-analysis. Ann Intern Med. 2015 Oct 6; 163:519. (http://dx.doi.org/10.7326/M15-0715).
Briel M et al. Corticosteroids in patients hospitalized with community-acquired Pneumonia: Systematic review and individual patient data meta-analysis. Clin Infect Dis. 2018 Feb 1; 66:346 (https://doi.org/10.1093/cid/cix801).
Blum CA et al. Adjunct prednisone therapy for patients with community-acquired pneumonia: A multicentre, double-blind, randomised, placebo-controlled trial. Lancet. 2015 Jan 18; [e-pub ahead of print]. (http://dx.doi.org/10.1016/S0140-6736(14)62447-8).
Feldman C et al. Corticosteroids in the adjunctive therapy of community-acquired pneumonia: an appraisal of recent meta-analyses of clinical trials. J Thorac Dis. 2016 Mar; 8(3):E162-E171.
Wan YD et al. Efficacy and safety of corticosteroids for community-acquired pneumonia: A systemic review and meta-analysis. Chest. 2016 Jan;149(1):209-19.
When is it appropriate to treat?
When is it appropriate to treat?
Case
A 55-year-old male with a history of tobacco use disorder presents with 2 days of productive cough, fever, chills, and mild shortness of breath. T 38.4, HR 89, RR 32, BP 100/65, 02 sat 86% on room air. Exam reveals diminished breath sounds and positive egophony over the right lung base. WBC is 16,000 and BUN 22. Chest x-ray reveals right lower lobe consolidation. He is given ceftriaxone and azithromycin.
Brief overview of the issue
Community-acquired pneumonia (CAP) is the leading cause of infectious disease–related death in the United States. Mortality associated with CAP is estimated at 57,000 deaths annually and occurs largely in patients requiring hospitalization.1 The 30-day mortality rate in patients who are hospitalized for CAP is approximately 10%-12%.2 After discharge from the hospital, about 18% of patients are readmitted within 30 days.3 An excessive inflammatory cytokine response may be a major contributor to the high mortality rate in CAP and systemic corticosteroids may reduce the inflammatory response from the infection by down-regulating this proinflammatory cytokine production.
Almost all of the major decisions regarding management of CAP, including diagnostic and treatment issues, revolve around the initial assessment of severity of illness. Between 40% and 60% of patients who present to the emergency department with CAP are admitted4 and approximately 10% of hospitalized patients with CAP require ICU admission.5 Validated instruments such as CURB-65, the pneumonia severity index (PSI), and guidelines from the Infectious Diseases Society of America (IDSA)/American Thoracic Society (ATS) may predict severity of illness but should always be supplemented with physician determination of subjective factors when determining treatment.5 Although there is no census definition of severe pneumonia, studies generally define the condition in the following order of preference: PSI score of IV or V followed by CURB-65 score of two or greater. If these scoring modalities were not available, the IDSA/ATS criteria was used (1 major or 3 minor). Others define severe CAP as pneumonia requiring supportive therapy within a critical care environment.
Overview of the data
The use of corticosteroids in addition to antibiotics in the treatment of CAP was proposed as early as the 1950s and yet only in the last decade has the body of evidence grown significantly.5 There is evidence that corticosteroids suppress inflammation without acutely impairing the immune response as evidenced by a rapid and sustained decrease in circulating inflammatory markers such as C-reactive protein and interleukin 6 and no effect on the anti-inflammatory interleukin 10.6 Within the last year, three meta-analyses, one by the Cochrane Library, one by the IDSA, and a third in the American Journal of Emergency Medicine, addressed the role of routine low dose (20-60 mg of prednisone or equivalent), short-course (3-7 days) systemic corticosteroids in hospitalized patients with CAP of varying severities.
The Cochrane meta-analysis, the largest and most recent dataset, included 13 trials with a combined 1,954 adult patients and found that corticosteroids significantly lowered mortality in hospitalized patients with severe CAP with a number needed to treat of 19.7 In this group with severe CAP, mortality was lowered from 13% to 8% and there were significantly fewer episodes of respiratory failure and shock with the addition of corticosteroids. No effect was seen on mortality in patients with less severe CAP. In those patients who received adjuvant corticosteroids, length of hospital stay decreased by 3 days, regardless of CAP severity.7
The IDSA meta-analysis was similar and included 1,506 patients from six trials.8 In contrast with the Cochrane study, this analysis found corticosteroids did not significantly lower mortality in patients with severe CAP but did reduce time to clinical stability and length of hospital stay by over 1 day. This study also found significantly more CAP-related, 30-day rehospitalizations (5% vs. 3%; defined as recurrent pneumonia, other infection, pleuritic pain, adverse cardiovascular event, or diarrhea) in patient with non-severe CAP treated with corticosteroids.
The study in the American Journal of Emergency Medicine involved ten trials involving more than 700 patients admitted with severe CAP and found in-hospital mortality was cut in half (RR 0.49) and length of hospital stay was reduced when patients were treated with corticosteroids in addition to standard antibiotic therapy.9
In 2015, two randomized clinical trials, one in the Lancet and the other in JAMA, and a meta-analysis in Annals of Internal Medicine assessed the impact of adjuvant corticosteroids in the treatment of hospitalized patients with CAP. The Lancet study of 785 patients hospitalized with CAP of any severity found shortened time to clinical stability (3.0 vs. 4.4 days) as defined by stable vital signs, improved oral intake, and normalized mental status for greater than 24 hours when oral prednisone 50 mg for 7 days was added to standard therapy.10 Patients in the treatment group were also discharged 1 day earlier compared with the placebo control group.
The study in JAMA was small, with only 100 patients at three teaching hospitals in Spain, but found that patients hospitalized with severe CAP and high inflammatory response based on elevated C-reactive protein were less likely to experience a treatment failure, defined as shock, mechanical ventilation, death, or radiographic progression, when intravenous methylprednisolone 0.5 mg/kg was added to standard antibiotic therapy.11
Finally, the meta-analysis in Annals of Internal Medicine assessed 13 randomized controlled placebo trials of 1,974 patients and found that adjuvant corticosteroids in a dose of 20-60 mg of prednisone or equivalent total daily dose significantly lowered mortality in patients with severe CAP and incidence of respiratory distress syndrome, and need for mechanical ventilation in all patients hospitalized with CAP.12
Importantly, nearly all of the described studies showed a significantly higher incidence of hyperglycemia in patients who received corticosteroids.
Application of the data to our patients
The benefit of adjuvant corticosteroids is most clear in hospitalized patients with severe CAP. Recent, strong evidence supports decreased mortality, decreased time to clinical stability, and decreased length of stay in our patient, with severe CAP, if treated with 20-60 mg of prednisone or equivalent total daily dose for 3-7 days. For patients with non-severe CAP, we suggest taking a risk-benefit approach based on other comorbidities, as the risk for CAP-related rehospitalizations may be higher.
For patients with underlying lung disease, specifically COPD or reactive airway disease, we suggest a low threshold for adding corticosteroids. This approach is more anecdotal than data driven, though corticosteroids are a mainstay of treatment for COPD exacerbations and a retrospective analysis of more than 20,000 hospitalized children with CAP and wheezing revealed decreased length of stay with corticosteroid treatment.13 Furthermore, a number of the studies described above included patients with COPD. Our threshold rises significantly in patients with poorly controlled diabetes mellitus.
Bottom line
For patients hospitalized with severe community-acquired pneumonia, recent evidence supports the use of low dose, short-course, systemic corticosteroids in addition to standard therapy.
Dr. Parsons is an assistant professor at the University of Virginia and a hospitalist at the University of Virginia Medical Center in Charlottesville, Va. Dr. Miller is an assistant professor at the University of Virginia and a hospitalist at the University of Virginia Medical Center. Dr. Hoke is Associate Director of Hospital Medicine and Faculty Development at the University of Virginia.
References
1. Ramirez J et al. Adults hospitalized with pneumonia in the United States: Incidence, epidemiology, and mortality. Clin Infect Dis. 2017 Dec 1:65(11):1806-12.
2. Musher D et al. Community-acquired pneumonia: Review article. N Engl J Med. 2014 Oct 23;371:1619-28.
3. Wunderink R et al. Community-aquired pneumonia: Clinical practice. N Engl J Med. 2014 Feb 6;370:543-51.
4. Mandell L et al. Infectious Diseases Society America/American Thoracic Society Consensus Guidelines on the Management of Community-Acquired Pneumonia in Adults. Clin Infect Dis. 2007;44:S27-72.
5. Wagner HN et al. The effect of hydrocortisone upon the course of pneumococcal pneumonia treated with penicillin. Bull Johns Hopkins Hosp. 1956;98:197-215.
6. Polverino E et al. Systemic corticosteroids for community-acquired pneumonia: Reasons for use and lack of benefit on outcome. Respirology. 2013. Feb;18(2):263-71 (https://doi.org/10.1111/resp.12013).
7. Stern A et al. Corticosteroids for pneumonia. Cochrane Database Syst Rev. 2017 Dec 13; 12:CD007720 (https://doi.org/10.1002/14651858.CD007720.pub3).
8. Briel M et al. Corticosteroids in patients hospitalized with community-acquired pneumonia: Systematic review and individual patient data meta-analysis. Clin Infect Dis. 2018 Feb 1;66:346 (https://doi.org/10.1093/cid/cix801).
9. Wu W-F et al. Efficacy of corticosteroid treatment for severe community-acquired pneumonia: A meta-analysis. Am J Emerg Med. 2017 Jul 15; [e-pub] (http://dx.doi.org/10.1016/j.ajem.2017.07.050).
10. Blum CA et al. Adjunct prednisone therapy for patients with community-acquired pneumonia: A multicentre, double-blind, randomised, placebo-controlled trial. Lancet 2015 Jan 18; [e-pub ahead of print] (http://dx.doi.org/10.1016/S0140-6736[14]62447-8).
11. Torres A et al. Effect of corticosteroids on treatment failure among hospitalized patients with severe community-acquired pneumonia and high inflammatory response: A randomized clinical trial. JAMA 2015 Feb 17; 313:677 (http://dx.doi.org/10.1001/jama.2015.88).
12. Siemieniuk RAC et al. Corticosteroid therapy for patients hospitalized with community-acquired pneumonia: A systematic review and meta-analysis. Ann Intern Med. 2015 Oct 6;163:519 (http://dx.doi.org/10.7326/M15-0715).
13. Simon LH et al. Management of community-acquired pneumonia in hospitalized children. Current Treat Options Peds (2015) 1:59 (https://doi:.org/10.1007/s40746-014-0011-3).
Key points
• For patients hospitalized with severe CAP, recent evidence supports the use of low-dose, short-course, systemic corticosteroids in addition to standard therapy.
• Among hospitalized patients with non-severe CAP, the benefit is not well defined. Studies suggest these patients may benefit from reduced time to clinical stability and reduced length of hospital stay. However, they may be at risk for significantly more CAP-related, 30-day rehospitalizations and hyperglycemia.
• Further prospective, randomized controlled studies are needed to further delineate the patient population who will most benefit from adjunctive corticosteroids use, including dose and duration of treatment.
QUIZ
Which of the following is FALSE regarding community acquired pneumonia?
A. CAP is the leading cause of infectious disease related death in the United States.
B. An excessive inflammatory cytokine response may contribute to the high mortality rate in CAP.
C. Adjunctive steroid therapy has been shown to decrease mortality in all patients with CAP.
D. Hyperglycemia occurs more frequently in patients receiving steroid therapy.
E. Reasons to avoid adjunctive steroid therapy in CAP include low risk for mortality, poorly controlled diabetes, suspected viral or fungal etiology, and elevated risk for gastrointestinal bleeding.
ANSWER: C. The patient population that may benefit most from the use of adjuvant corticosteroids is poorly defined. However, in patients with severe pneumonia, the use of adjuvant steroids has been shown to decrease mortality, time to clinical stability, and length of stay.
Additional reading
Siemieniuk RAC et al. Corticosteroid therapy for patients hospitalized with community-acquired pneumonia: A systematic review and meta-analysis. Ann Intern Med. 2015 Oct 6; 163:519. (http://dx.doi.org/10.7326/M15-0715).
Briel M et al. Corticosteroids in patients hospitalized with community-acquired Pneumonia: Systematic review and individual patient data meta-analysis. Clin Infect Dis. 2018 Feb 1; 66:346 (https://doi.org/10.1093/cid/cix801).
Blum CA et al. Adjunct prednisone therapy for patients with community-acquired pneumonia: A multicentre, double-blind, randomised, placebo-controlled trial. Lancet. 2015 Jan 18; [e-pub ahead of print]. (http://dx.doi.org/10.1016/S0140-6736(14)62447-8).
Feldman C et al. Corticosteroids in the adjunctive therapy of community-acquired pneumonia: an appraisal of recent meta-analyses of clinical trials. J Thorac Dis. 2016 Mar; 8(3):E162-E171.
Wan YD et al. Efficacy and safety of corticosteroids for community-acquired pneumonia: A systemic review and meta-analysis. Chest. 2016 Jan;149(1):209-19.
Case
A 55-year-old male with a history of tobacco use disorder presents with 2 days of productive cough, fever, chills, and mild shortness of breath. T 38.4, HR 89, RR 32, BP 100/65, 02 sat 86% on room air. Exam reveals diminished breath sounds and positive egophony over the right lung base. WBC is 16,000 and BUN 22. Chest x-ray reveals right lower lobe consolidation. He is given ceftriaxone and azithromycin.
Brief overview of the issue
Community-acquired pneumonia (CAP) is the leading cause of infectious disease–related death in the United States. Mortality associated with CAP is estimated at 57,000 deaths annually and occurs largely in patients requiring hospitalization.1 The 30-day mortality rate in patients who are hospitalized for CAP is approximately 10%-12%.2 After discharge from the hospital, about 18% of patients are readmitted within 30 days.3 An excessive inflammatory cytokine response may be a major contributor to the high mortality rate in CAP and systemic corticosteroids may reduce the inflammatory response from the infection by down-regulating this proinflammatory cytokine production.
Almost all of the major decisions regarding management of CAP, including diagnostic and treatment issues, revolve around the initial assessment of severity of illness. Between 40% and 60% of patients who present to the emergency department with CAP are admitted4 and approximately 10% of hospitalized patients with CAP require ICU admission.5 Validated instruments such as CURB-65, the pneumonia severity index (PSI), and guidelines from the Infectious Diseases Society of America (IDSA)/American Thoracic Society (ATS) may predict severity of illness but should always be supplemented with physician determination of subjective factors when determining treatment.5 Although there is no census definition of severe pneumonia, studies generally define the condition in the following order of preference: PSI score of IV or V followed by CURB-65 score of two or greater. If these scoring modalities were not available, the IDSA/ATS criteria was used (1 major or 3 minor). Others define severe CAP as pneumonia requiring supportive therapy within a critical care environment.
Overview of the data
The use of corticosteroids in addition to antibiotics in the treatment of CAP was proposed as early as the 1950s and yet only in the last decade has the body of evidence grown significantly.5 There is evidence that corticosteroids suppress inflammation without acutely impairing the immune response as evidenced by a rapid and sustained decrease in circulating inflammatory markers such as C-reactive protein and interleukin 6 and no effect on the anti-inflammatory interleukin 10.6 Within the last year, three meta-analyses, one by the Cochrane Library, one by the IDSA, and a third in the American Journal of Emergency Medicine, addressed the role of routine low dose (20-60 mg of prednisone or equivalent), short-course (3-7 days) systemic corticosteroids in hospitalized patients with CAP of varying severities.
The Cochrane meta-analysis, the largest and most recent dataset, included 13 trials with a combined 1,954 adult patients and found that corticosteroids significantly lowered mortality in hospitalized patients with severe CAP with a number needed to treat of 19.7 In this group with severe CAP, mortality was lowered from 13% to 8% and there were significantly fewer episodes of respiratory failure and shock with the addition of corticosteroids. No effect was seen on mortality in patients with less severe CAP. In those patients who received adjuvant corticosteroids, length of hospital stay decreased by 3 days, regardless of CAP severity.7
The IDSA meta-analysis was similar and included 1,506 patients from six trials.8 In contrast with the Cochrane study, this analysis found corticosteroids did not significantly lower mortality in patients with severe CAP but did reduce time to clinical stability and length of hospital stay by over 1 day. This study also found significantly more CAP-related, 30-day rehospitalizations (5% vs. 3%; defined as recurrent pneumonia, other infection, pleuritic pain, adverse cardiovascular event, or diarrhea) in patient with non-severe CAP treated with corticosteroids.
The study in the American Journal of Emergency Medicine involved ten trials involving more than 700 patients admitted with severe CAP and found in-hospital mortality was cut in half (RR 0.49) and length of hospital stay was reduced when patients were treated with corticosteroids in addition to standard antibiotic therapy.9
In 2015, two randomized clinical trials, one in the Lancet and the other in JAMA, and a meta-analysis in Annals of Internal Medicine assessed the impact of adjuvant corticosteroids in the treatment of hospitalized patients with CAP. The Lancet study of 785 patients hospitalized with CAP of any severity found shortened time to clinical stability (3.0 vs. 4.4 days) as defined by stable vital signs, improved oral intake, and normalized mental status for greater than 24 hours when oral prednisone 50 mg for 7 days was added to standard therapy.10 Patients in the treatment group were also discharged 1 day earlier compared with the placebo control group.
The study in JAMA was small, with only 100 patients at three teaching hospitals in Spain, but found that patients hospitalized with severe CAP and high inflammatory response based on elevated C-reactive protein were less likely to experience a treatment failure, defined as shock, mechanical ventilation, death, or radiographic progression, when intravenous methylprednisolone 0.5 mg/kg was added to standard antibiotic therapy.11
Finally, the meta-analysis in Annals of Internal Medicine assessed 13 randomized controlled placebo trials of 1,974 patients and found that adjuvant corticosteroids in a dose of 20-60 mg of prednisone or equivalent total daily dose significantly lowered mortality in patients with severe CAP and incidence of respiratory distress syndrome, and need for mechanical ventilation in all patients hospitalized with CAP.12
Importantly, nearly all of the described studies showed a significantly higher incidence of hyperglycemia in patients who received corticosteroids.
Application of the data to our patients
The benefit of adjuvant corticosteroids is most clear in hospitalized patients with severe CAP. Recent, strong evidence supports decreased mortality, decreased time to clinical stability, and decreased length of stay in our patient, with severe CAP, if treated with 20-60 mg of prednisone or equivalent total daily dose for 3-7 days. For patients with non-severe CAP, we suggest taking a risk-benefit approach based on other comorbidities, as the risk for CAP-related rehospitalizations may be higher.
For patients with underlying lung disease, specifically COPD or reactive airway disease, we suggest a low threshold for adding corticosteroids. This approach is more anecdotal than data driven, though corticosteroids are a mainstay of treatment for COPD exacerbations and a retrospective analysis of more than 20,000 hospitalized children with CAP and wheezing revealed decreased length of stay with corticosteroid treatment.13 Furthermore, a number of the studies described above included patients with COPD. Our threshold rises significantly in patients with poorly controlled diabetes mellitus.
Bottom line
For patients hospitalized with severe community-acquired pneumonia, recent evidence supports the use of low dose, short-course, systemic corticosteroids in addition to standard therapy.
Dr. Parsons is an assistant professor at the University of Virginia and a hospitalist at the University of Virginia Medical Center in Charlottesville, Va. Dr. Miller is an assistant professor at the University of Virginia and a hospitalist at the University of Virginia Medical Center. Dr. Hoke is Associate Director of Hospital Medicine and Faculty Development at the University of Virginia.
References
1. Ramirez J et al. Adults hospitalized with pneumonia in the United States: Incidence, epidemiology, and mortality. Clin Infect Dis. 2017 Dec 1:65(11):1806-12.
2. Musher D et al. Community-acquired pneumonia: Review article. N Engl J Med. 2014 Oct 23;371:1619-28.
3. Wunderink R et al. Community-aquired pneumonia: Clinical practice. N Engl J Med. 2014 Feb 6;370:543-51.
4. Mandell L et al. Infectious Diseases Society America/American Thoracic Society Consensus Guidelines on the Management of Community-Acquired Pneumonia in Adults. Clin Infect Dis. 2007;44:S27-72.
5. Wagner HN et al. The effect of hydrocortisone upon the course of pneumococcal pneumonia treated with penicillin. Bull Johns Hopkins Hosp. 1956;98:197-215.
6. Polverino E et al. Systemic corticosteroids for community-acquired pneumonia: Reasons for use and lack of benefit on outcome. Respirology. 2013. Feb;18(2):263-71 (https://doi.org/10.1111/resp.12013).
7. Stern A et al. Corticosteroids for pneumonia. Cochrane Database Syst Rev. 2017 Dec 13; 12:CD007720 (https://doi.org/10.1002/14651858.CD007720.pub3).
8. Briel M et al. Corticosteroids in patients hospitalized with community-acquired pneumonia: Systematic review and individual patient data meta-analysis. Clin Infect Dis. 2018 Feb 1;66:346 (https://doi.org/10.1093/cid/cix801).
9. Wu W-F et al. Efficacy of corticosteroid treatment for severe community-acquired pneumonia: A meta-analysis. Am J Emerg Med. 2017 Jul 15; [e-pub] (http://dx.doi.org/10.1016/j.ajem.2017.07.050).
10. Blum CA et al. Adjunct prednisone therapy for patients with community-acquired pneumonia: A multicentre, double-blind, randomised, placebo-controlled trial. Lancet 2015 Jan 18; [e-pub ahead of print] (http://dx.doi.org/10.1016/S0140-6736[14]62447-8).
11. Torres A et al. Effect of corticosteroids on treatment failure among hospitalized patients with severe community-acquired pneumonia and high inflammatory response: A randomized clinical trial. JAMA 2015 Feb 17; 313:677 (http://dx.doi.org/10.1001/jama.2015.88).
12. Siemieniuk RAC et al. Corticosteroid therapy for patients hospitalized with community-acquired pneumonia: A systematic review and meta-analysis. Ann Intern Med. 2015 Oct 6;163:519 (http://dx.doi.org/10.7326/M15-0715).
13. Simon LH et al. Management of community-acquired pneumonia in hospitalized children. Current Treat Options Peds (2015) 1:59 (https://doi:.org/10.1007/s40746-014-0011-3).
Key points
• For patients hospitalized with severe CAP, recent evidence supports the use of low-dose, short-course, systemic corticosteroids in addition to standard therapy.
• Among hospitalized patients with non-severe CAP, the benefit is not well defined. Studies suggest these patients may benefit from reduced time to clinical stability and reduced length of hospital stay. However, they may be at risk for significantly more CAP-related, 30-day rehospitalizations and hyperglycemia.
• Further prospective, randomized controlled studies are needed to further delineate the patient population who will most benefit from adjunctive corticosteroids use, including dose and duration of treatment.
QUIZ
Which of the following is FALSE regarding community acquired pneumonia?
A. CAP is the leading cause of infectious disease related death in the United States.
B. An excessive inflammatory cytokine response may contribute to the high mortality rate in CAP.
C. Adjunctive steroid therapy has been shown to decrease mortality in all patients with CAP.
D. Hyperglycemia occurs more frequently in patients receiving steroid therapy.
E. Reasons to avoid adjunctive steroid therapy in CAP include low risk for mortality, poorly controlled diabetes, suspected viral or fungal etiology, and elevated risk for gastrointestinal bleeding.
ANSWER: C. The patient population that may benefit most from the use of adjuvant corticosteroids is poorly defined. However, in patients with severe pneumonia, the use of adjuvant steroids has been shown to decrease mortality, time to clinical stability, and length of stay.
Additional reading
Siemieniuk RAC et al. Corticosteroid therapy for patients hospitalized with community-acquired pneumonia: A systematic review and meta-analysis. Ann Intern Med. 2015 Oct 6; 163:519. (http://dx.doi.org/10.7326/M15-0715).
Briel M et al. Corticosteroids in patients hospitalized with community-acquired Pneumonia: Systematic review and individual patient data meta-analysis. Clin Infect Dis. 2018 Feb 1; 66:346 (https://doi.org/10.1093/cid/cix801).
Blum CA et al. Adjunct prednisone therapy for patients with community-acquired pneumonia: A multicentre, double-blind, randomised, placebo-controlled trial. Lancet. 2015 Jan 18; [e-pub ahead of print]. (http://dx.doi.org/10.1016/S0140-6736(14)62447-8).
Feldman C et al. Corticosteroids in the adjunctive therapy of community-acquired pneumonia: an appraisal of recent meta-analyses of clinical trials. J Thorac Dis. 2016 Mar; 8(3):E162-E171.
Wan YD et al. Efficacy and safety of corticosteroids for community-acquired pneumonia: A systemic review and meta-analysis. Chest. 2016 Jan;149(1):209-19.
COPD exacerbations associated with poor sleep quality
in an 18-month prospective study of 480 patients.
“Poor sleep quality in COPD has previously been associated with reduced health-related quality of life and reduced physical activity during the day,” wrote Matthew Shorofsky, MD, of McGill University, Montreal, and associates. Their report is in CHEST. “However, to our knowledge, this is the first population-based longitudinal study evaluating exacerbation risk in relation to subjective sleep disturbances and assessing previously diagnosed and undiagnosed COPD.”
The study included participants enrolled in the Canadian Respiratory Research Network and the Canadian Cohort Obstructive Lung Disease (CanCOLD) study who had COPD, available baseline PSQI scores, and 18 months of follow-up data. The PSQI includes 19 questions on sleep quality, latency, duration, efficiency, disturbances, use of sleep medications, and daytime dysfunction. Total score ranges between 0 and 21, and a score above 5 is considered poor sleep. Online patient surveys and quarterly phone interviews were used to track symptom-based exacerbations (at least 48 hours of increased dyspnea, sputum volume, or sputum purulence) and event-based exacerbations (a symptom-based exacerbation plus the use antibiotics or corticosteroids or health services).
At baseline, 203 patients met the PSQI threshold for poor sleep quality. During follow-up, 185 patients had at least one COPD exacerbation. Poor sleep at baseline was significantly more prevalent among patients with symptoms-based COPD exacerbations (50.3%) than among patients without symptoms-based exacerbations (37.3%; P = .01). Poor baseline sleep quality remained a significant risk factor for symptom-based exacerbations of COPD even after the researchers accounted for the effect of age, gender, body mass index, smoking, depression, angina, baseline inhaled respiratory medications, forced expiratory volume in 1 second %predicted, and modified Medical Research Council (mMRC) dyspnea scale (adjusted risk ratio, 1.09; 95% confidence interval, 1.01-1.18; P =.02).
Patients with at least one symptomatic exacerbation of COPD were significantly more likely to meet the threshold for poor sleep quality on the Pittsburgh Sleep Quality Index and have significantly higher median PSQI scores compared with patients without exacerbations (6.0 [interquartile range, 3.0 to 8.0] vs. 5.0 [2.0 to 7.0]; P = .01). Poor baseline sleep quality also was associated with event-based exacerbations and with a shorter time to symptoms-based exacerbations. Sleep disturbances, such as rising to void or experiencing respiratory issues or pain during sleep, correlated most strongly with symptoms-based exacerbations.
Several factors could explain the link between poor sleep quality and COPD exacerbations, the investigators wrote. Patients with inadequately controlled COPD have more frequent and unstable respiratory symptoms, which could disrupt sleep either directly or indirectly (secondary to medication use or anxiety, for example). Conversely, sleep disruption can impede immune function and increase systemic inflammation, which might worsen COPD control and increase exacerbation risk. Poor sleep can impair memory and cognition, “potentially fostering medication nonadherence and symptom flare-up, especially in the older COPD population.” Although the link is poorly understood, patients with COPD often have comorbid obstructive sleep apnea (OSA), which is associated with COPD exacerbations, the researchers wrote. Treating OSA is associated with improved COPD morbidity and fewer exacerbations and hospitalizations.
The researchers acknowledged limitations to their study design. “Individuals with asthma or other obstructive lung diseases could not be definitively excluded; methacholine challenges were not performed. However, analyses excluding self-reported asthma were consistent with our main results. Second, because definitions of COPD exacerbation vary among studies, comparison may be limited, but CanCOLD used a standard definition, as recommended by GOLD.”
The CanCOLD study has received funding from the Canadian Respiratory Research Network, Astra Zeneca Canada, Boehringer Ingelheim Canada, GlaxoSmithKline Canada, Novartis, Merck Nycomed, Pfizer Canada, and Theratechnologies. Dr. Shorofsky had no disclosures. Several coinvestigators reported ties to GlaxoSmithKline, Novartis, Boehringer Ingelheim, Merck, Almirall, and Theratechnologies.
SOURCE: Shorofsky M et al. CHEST. 2019 May 28. doi: 10.1016/j.chest.2019.04.132.
in an 18-month prospective study of 480 patients.
“Poor sleep quality in COPD has previously been associated with reduced health-related quality of life and reduced physical activity during the day,” wrote Matthew Shorofsky, MD, of McGill University, Montreal, and associates. Their report is in CHEST. “However, to our knowledge, this is the first population-based longitudinal study evaluating exacerbation risk in relation to subjective sleep disturbances and assessing previously diagnosed and undiagnosed COPD.”
The study included participants enrolled in the Canadian Respiratory Research Network and the Canadian Cohort Obstructive Lung Disease (CanCOLD) study who had COPD, available baseline PSQI scores, and 18 months of follow-up data. The PSQI includes 19 questions on sleep quality, latency, duration, efficiency, disturbances, use of sleep medications, and daytime dysfunction. Total score ranges between 0 and 21, and a score above 5 is considered poor sleep. Online patient surveys and quarterly phone interviews were used to track symptom-based exacerbations (at least 48 hours of increased dyspnea, sputum volume, or sputum purulence) and event-based exacerbations (a symptom-based exacerbation plus the use antibiotics or corticosteroids or health services).
At baseline, 203 patients met the PSQI threshold for poor sleep quality. During follow-up, 185 patients had at least one COPD exacerbation. Poor sleep at baseline was significantly more prevalent among patients with symptoms-based COPD exacerbations (50.3%) than among patients without symptoms-based exacerbations (37.3%; P = .01). Poor baseline sleep quality remained a significant risk factor for symptom-based exacerbations of COPD even after the researchers accounted for the effect of age, gender, body mass index, smoking, depression, angina, baseline inhaled respiratory medications, forced expiratory volume in 1 second %predicted, and modified Medical Research Council (mMRC) dyspnea scale (adjusted risk ratio, 1.09; 95% confidence interval, 1.01-1.18; P =.02).
Patients with at least one symptomatic exacerbation of COPD were significantly more likely to meet the threshold for poor sleep quality on the Pittsburgh Sleep Quality Index and have significantly higher median PSQI scores compared with patients without exacerbations (6.0 [interquartile range, 3.0 to 8.0] vs. 5.0 [2.0 to 7.0]; P = .01). Poor baseline sleep quality also was associated with event-based exacerbations and with a shorter time to symptoms-based exacerbations. Sleep disturbances, such as rising to void or experiencing respiratory issues or pain during sleep, correlated most strongly with symptoms-based exacerbations.
Several factors could explain the link between poor sleep quality and COPD exacerbations, the investigators wrote. Patients with inadequately controlled COPD have more frequent and unstable respiratory symptoms, which could disrupt sleep either directly or indirectly (secondary to medication use or anxiety, for example). Conversely, sleep disruption can impede immune function and increase systemic inflammation, which might worsen COPD control and increase exacerbation risk. Poor sleep can impair memory and cognition, “potentially fostering medication nonadherence and symptom flare-up, especially in the older COPD population.” Although the link is poorly understood, patients with COPD often have comorbid obstructive sleep apnea (OSA), which is associated with COPD exacerbations, the researchers wrote. Treating OSA is associated with improved COPD morbidity and fewer exacerbations and hospitalizations.
The researchers acknowledged limitations to their study design. “Individuals with asthma or other obstructive lung diseases could not be definitively excluded; methacholine challenges were not performed. However, analyses excluding self-reported asthma were consistent with our main results. Second, because definitions of COPD exacerbation vary among studies, comparison may be limited, but CanCOLD used a standard definition, as recommended by GOLD.”
The CanCOLD study has received funding from the Canadian Respiratory Research Network, Astra Zeneca Canada, Boehringer Ingelheim Canada, GlaxoSmithKline Canada, Novartis, Merck Nycomed, Pfizer Canada, and Theratechnologies. Dr. Shorofsky had no disclosures. Several coinvestigators reported ties to GlaxoSmithKline, Novartis, Boehringer Ingelheim, Merck, Almirall, and Theratechnologies.
SOURCE: Shorofsky M et al. CHEST. 2019 May 28. doi: 10.1016/j.chest.2019.04.132.
in an 18-month prospective study of 480 patients.
“Poor sleep quality in COPD has previously been associated with reduced health-related quality of life and reduced physical activity during the day,” wrote Matthew Shorofsky, MD, of McGill University, Montreal, and associates. Their report is in CHEST. “However, to our knowledge, this is the first population-based longitudinal study evaluating exacerbation risk in relation to subjective sleep disturbances and assessing previously diagnosed and undiagnosed COPD.”
The study included participants enrolled in the Canadian Respiratory Research Network and the Canadian Cohort Obstructive Lung Disease (CanCOLD) study who had COPD, available baseline PSQI scores, and 18 months of follow-up data. The PSQI includes 19 questions on sleep quality, latency, duration, efficiency, disturbances, use of sleep medications, and daytime dysfunction. Total score ranges between 0 and 21, and a score above 5 is considered poor sleep. Online patient surveys and quarterly phone interviews were used to track symptom-based exacerbations (at least 48 hours of increased dyspnea, sputum volume, or sputum purulence) and event-based exacerbations (a symptom-based exacerbation plus the use antibiotics or corticosteroids or health services).
At baseline, 203 patients met the PSQI threshold for poor sleep quality. During follow-up, 185 patients had at least one COPD exacerbation. Poor sleep at baseline was significantly more prevalent among patients with symptoms-based COPD exacerbations (50.3%) than among patients without symptoms-based exacerbations (37.3%; P = .01). Poor baseline sleep quality remained a significant risk factor for symptom-based exacerbations of COPD even after the researchers accounted for the effect of age, gender, body mass index, smoking, depression, angina, baseline inhaled respiratory medications, forced expiratory volume in 1 second %predicted, and modified Medical Research Council (mMRC) dyspnea scale (adjusted risk ratio, 1.09; 95% confidence interval, 1.01-1.18; P =.02).
Patients with at least one symptomatic exacerbation of COPD were significantly more likely to meet the threshold for poor sleep quality on the Pittsburgh Sleep Quality Index and have significantly higher median PSQI scores compared with patients without exacerbations (6.0 [interquartile range, 3.0 to 8.0] vs. 5.0 [2.0 to 7.0]; P = .01). Poor baseline sleep quality also was associated with event-based exacerbations and with a shorter time to symptoms-based exacerbations. Sleep disturbances, such as rising to void or experiencing respiratory issues or pain during sleep, correlated most strongly with symptoms-based exacerbations.
Several factors could explain the link between poor sleep quality and COPD exacerbations, the investigators wrote. Patients with inadequately controlled COPD have more frequent and unstable respiratory symptoms, which could disrupt sleep either directly or indirectly (secondary to medication use or anxiety, for example). Conversely, sleep disruption can impede immune function and increase systemic inflammation, which might worsen COPD control and increase exacerbation risk. Poor sleep can impair memory and cognition, “potentially fostering medication nonadherence and symptom flare-up, especially in the older COPD population.” Although the link is poorly understood, patients with COPD often have comorbid obstructive sleep apnea (OSA), which is associated with COPD exacerbations, the researchers wrote. Treating OSA is associated with improved COPD morbidity and fewer exacerbations and hospitalizations.
The researchers acknowledged limitations to their study design. “Individuals with asthma or other obstructive lung diseases could not be definitively excluded; methacholine challenges were not performed. However, analyses excluding self-reported asthma were consistent with our main results. Second, because definitions of COPD exacerbation vary among studies, comparison may be limited, but CanCOLD used a standard definition, as recommended by GOLD.”
The CanCOLD study has received funding from the Canadian Respiratory Research Network, Astra Zeneca Canada, Boehringer Ingelheim Canada, GlaxoSmithKline Canada, Novartis, Merck Nycomed, Pfizer Canada, and Theratechnologies. Dr. Shorofsky had no disclosures. Several coinvestigators reported ties to GlaxoSmithKline, Novartis, Boehringer Ingelheim, Merck, Almirall, and Theratechnologies.
SOURCE: Shorofsky M et al. CHEST. 2019 May 28. doi: 10.1016/j.chest.2019.04.132.
FROM CHEST
Rivaroxaban tied to higher GI bleeding than other NOACs
SAN DIEGO – Patients on rivaroxaban had significantly higher rates of GI bleeding, compared with those taking apixaban or dabigatran, results from a large population-based study showed.
“This may be due to the fact that rivaroxaban is administered as a single daily dose as opposed to the other two non–vitamin K anticoagulants [NOACs], which are given twice daily,” lead study author Arnar B. Ingason said at the annual Digestive Disease Week. “This may lead to a greater variance in plasma drug concentration, making these patients more susceptible to bleeding.”
Mr. Ingason, a medical student at the University of Iceland, Reykjavik, said that although several studies have compared warfarin with NOACs, it remains unclear which NOAC has the most favorable GI profile. In an effort to improve the research in this area, he and his associates performed a nationwide, population-based study during March 2014–Jan. 2018 to compare the GI bleeding risk of patients receiving rivaroxaban to that of a combined pool of patients receiving either apixaban or dabigatran. They drew from the Icelandic Medicine Registry, which contains all outpatient drug prescriptions in the country. Next, the researchers linked the personal identification numbers of patients to the Landspitali University diagnoses registry, which includes more than 90% of all patients hospitalized for GI bleeding. They used 1:1 nearest neighbor propensity score for matching and Kaplan-Meier survival estimates and Cox regression to compare rates of GI bleeding. The study outcome of interest was any clinically relevant GI bleeding.
Mr. Ingason reported that the baseline characteristics were similar between the rivaroxaban group and the apixaban/dabigatran group. They matched for several variables, including age, sex, Charlson score, the proportion being anticoagulant naive, moderate to severe renal disease, moderate to severe liver disease, any prior bleeding, and any prior thrombotic events.
During the study period, 3,473 patients received rivaroxaban, 1,901 received apixaban, and 1,086 received dabigatran. After propensity score matching, the researchers compared 2,635 patients who received rivaroxaban with 2,365 patients who received either apixaban or dabigatran. They found that patients in the rivaroxaban group had significantly higher rates of GI bleeding, compared with in the apixaban/dabigatran group (1.2 and. 0.6 events per 100 patient-years, respectively). This yielded a hazard ratio of 2.02, “which means that patients receiving rivaroxaban are twice as likely to get GI bleeding compared to patients on apixaban or dabigatran,” Mr. Ingason said. When the researchers examined the entire unmatched cohort of patients, the rivaroxaban group also had significantly higher rates of GI bleeding, compared with the apixaban/dabigatran group (1.0 and 0.6 events per 100 patient-years; HR, 1.75).
Mr. Ingason and his colleagues observed that patients in the rivaroxaban group had higher rates of GI bleeding, compared with the apixaban/dabigatran group, during the entire follow-up period. At the end of year 4, the rivaroxaban group had a 4% cumulative event rate of GI bleeding, compared with 1.8% for the apixaban/dabigatran group, a highly significant difference at P = .0057).
When a meeting attendee asked Mr. Ingason why patients taking apixaban or dabigatran were combined into one group, he said that it was done to increase the power of their study. “Our theory was that rivaroxaban was different because it is administered as a single daily dose, while the others are given twice daily,” he said. The researchers reported having no financial disclosures.
SAN DIEGO – Patients on rivaroxaban had significantly higher rates of GI bleeding, compared with those taking apixaban or dabigatran, results from a large population-based study showed.
“This may be due to the fact that rivaroxaban is administered as a single daily dose as opposed to the other two non–vitamin K anticoagulants [NOACs], which are given twice daily,” lead study author Arnar B. Ingason said at the annual Digestive Disease Week. “This may lead to a greater variance in plasma drug concentration, making these patients more susceptible to bleeding.”
Mr. Ingason, a medical student at the University of Iceland, Reykjavik, said that although several studies have compared warfarin with NOACs, it remains unclear which NOAC has the most favorable GI profile. In an effort to improve the research in this area, he and his associates performed a nationwide, population-based study during March 2014–Jan. 2018 to compare the GI bleeding risk of patients receiving rivaroxaban to that of a combined pool of patients receiving either apixaban or dabigatran. They drew from the Icelandic Medicine Registry, which contains all outpatient drug prescriptions in the country. Next, the researchers linked the personal identification numbers of patients to the Landspitali University diagnoses registry, which includes more than 90% of all patients hospitalized for GI bleeding. They used 1:1 nearest neighbor propensity score for matching and Kaplan-Meier survival estimates and Cox regression to compare rates of GI bleeding. The study outcome of interest was any clinically relevant GI bleeding.
Mr. Ingason reported that the baseline characteristics were similar between the rivaroxaban group and the apixaban/dabigatran group. They matched for several variables, including age, sex, Charlson score, the proportion being anticoagulant naive, moderate to severe renal disease, moderate to severe liver disease, any prior bleeding, and any prior thrombotic events.
During the study period, 3,473 patients received rivaroxaban, 1,901 received apixaban, and 1,086 received dabigatran. After propensity score matching, the researchers compared 2,635 patients who received rivaroxaban with 2,365 patients who received either apixaban or dabigatran. They found that patients in the rivaroxaban group had significantly higher rates of GI bleeding, compared with in the apixaban/dabigatran group (1.2 and. 0.6 events per 100 patient-years, respectively). This yielded a hazard ratio of 2.02, “which means that patients receiving rivaroxaban are twice as likely to get GI bleeding compared to patients on apixaban or dabigatran,” Mr. Ingason said. When the researchers examined the entire unmatched cohort of patients, the rivaroxaban group also had significantly higher rates of GI bleeding, compared with the apixaban/dabigatran group (1.0 and 0.6 events per 100 patient-years; HR, 1.75).
Mr. Ingason and his colleagues observed that patients in the rivaroxaban group had higher rates of GI bleeding, compared with the apixaban/dabigatran group, during the entire follow-up period. At the end of year 4, the rivaroxaban group had a 4% cumulative event rate of GI bleeding, compared with 1.8% for the apixaban/dabigatran group, a highly significant difference at P = .0057).
When a meeting attendee asked Mr. Ingason why patients taking apixaban or dabigatran were combined into one group, he said that it was done to increase the power of their study. “Our theory was that rivaroxaban was different because it is administered as a single daily dose, while the others are given twice daily,” he said. The researchers reported having no financial disclosures.
SAN DIEGO – Patients on rivaroxaban had significantly higher rates of GI bleeding, compared with those taking apixaban or dabigatran, results from a large population-based study showed.
“This may be due to the fact that rivaroxaban is administered as a single daily dose as opposed to the other two non–vitamin K anticoagulants [NOACs], which are given twice daily,” lead study author Arnar B. Ingason said at the annual Digestive Disease Week. “This may lead to a greater variance in plasma drug concentration, making these patients more susceptible to bleeding.”
Mr. Ingason, a medical student at the University of Iceland, Reykjavik, said that although several studies have compared warfarin with NOACs, it remains unclear which NOAC has the most favorable GI profile. In an effort to improve the research in this area, he and his associates performed a nationwide, population-based study during March 2014–Jan. 2018 to compare the GI bleeding risk of patients receiving rivaroxaban to that of a combined pool of patients receiving either apixaban or dabigatran. They drew from the Icelandic Medicine Registry, which contains all outpatient drug prescriptions in the country. Next, the researchers linked the personal identification numbers of patients to the Landspitali University diagnoses registry, which includes more than 90% of all patients hospitalized for GI bleeding. They used 1:1 nearest neighbor propensity score for matching and Kaplan-Meier survival estimates and Cox regression to compare rates of GI bleeding. The study outcome of interest was any clinically relevant GI bleeding.
Mr. Ingason reported that the baseline characteristics were similar between the rivaroxaban group and the apixaban/dabigatran group. They matched for several variables, including age, sex, Charlson score, the proportion being anticoagulant naive, moderate to severe renal disease, moderate to severe liver disease, any prior bleeding, and any prior thrombotic events.
During the study period, 3,473 patients received rivaroxaban, 1,901 received apixaban, and 1,086 received dabigatran. After propensity score matching, the researchers compared 2,635 patients who received rivaroxaban with 2,365 patients who received either apixaban or dabigatran. They found that patients in the rivaroxaban group had significantly higher rates of GI bleeding, compared with in the apixaban/dabigatran group (1.2 and. 0.6 events per 100 patient-years, respectively). This yielded a hazard ratio of 2.02, “which means that patients receiving rivaroxaban are twice as likely to get GI bleeding compared to patients on apixaban or dabigatran,” Mr. Ingason said. When the researchers examined the entire unmatched cohort of patients, the rivaroxaban group also had significantly higher rates of GI bleeding, compared with the apixaban/dabigatran group (1.0 and 0.6 events per 100 patient-years; HR, 1.75).
Mr. Ingason and his colleagues observed that patients in the rivaroxaban group had higher rates of GI bleeding, compared with the apixaban/dabigatran group, during the entire follow-up period. At the end of year 4, the rivaroxaban group had a 4% cumulative event rate of GI bleeding, compared with 1.8% for the apixaban/dabigatran group, a highly significant difference at P = .0057).
When a meeting attendee asked Mr. Ingason why patients taking apixaban or dabigatran were combined into one group, he said that it was done to increase the power of their study. “Our theory was that rivaroxaban was different because it is administered as a single daily dose, while the others are given twice daily,” he said. The researchers reported having no financial disclosures.
REPORTING FROM DDW 2019