User login
Vulvar Inflammatory Dermatoses: New Approaches for Diagnosis and Treatment
Vulvar dermatoses continue to be an overlooked aspect of medical care, highlighting the necessity for enhanced diagnosis and management of these conditions. Here, we address recent advancements in understanding vulvar inflammatory dermatoses other than lichen sclerosus (LS), which was discussed in a prior Guest Editorial1—specifically vulvovaginal lichen planus (VLP), plasma cell vulvitis (PCV), and vulvar lichen simplex chronicus (LSC).
Vulvar Inflammatory Skin Disease and Quality of Life
There is an increased awareness of the impact vulvar skin disease has on quality of life and its association with anxiety and depression.2-5 Evaluating the burden of vulvar dermatoses remains an active area of research due to its significance in monitoring disease progression and assessing therapeutic effectiveness. Despite the existence of various dermatology quality-of-life assessment tools, many fail to adequately capture the unique impacts of vulvovaginal diseases, such as sexual or urinary dysfunction. The vulvar quality of life index, which was developed and validated by Saunderson et al6 in 2020, consists of a 15-item questionnaire spanning 4 domains: symptoms, anxiety, activities of daily living, and sexuality. This tool has been utilized to gauge treatment response in vulvar conditions and to compare disease burden of various vulvar dermatoses.7,8 Moving forward, integrating this tool into clinical studies on vulvar skin disease holds promise for enhancing our understanding and management of these conditions.
Vulvovaginal Lichen Planus
Vulvovaginal lichen planus is unique among several prevalent vulvar inflammatory skin disorders encountered by dermatologists—primarily due to its erosive form, which can extend to the vagina, resulting in noninfectious vaginitis and potential vaginal stenosis.9,10 Managing VLP poses a notable challenge, even when it is confined to the vulva, as it often proves resistant to topical therapies.11
Evaluation for Vaginal Mucosal Disease—In contrast to LS, which typically spares the vaginal mucosa, VLP can involve mucosal sites.9,12,13 Therefore, it is imperative that all patients with a diagnosis of vulvar VLP undergo evaluation for potential vaginal involvement through speculum examination, wet mount, or vaginal biopsy. Strategies to manage vaginal involvement include use of dilators and pelvic floor physical therapy, lysis of adhesions (if present), topical estrogen, and intravaginal corticosteroids—all tailored to the severity of the disease.9,11,14
Management of VLP—Approximately 20% to 40% of patients with VLP may require systemic therapy for disease management, including those who are younger, those of non-White ethnicity, and those presenting with vulvar pruritus.11 Various systemic immunosuppressants have been used for VLP, with a recent retrospective study revealing similar response rates for both methotrexate and mycophenolate mofetil in the treatment of VLP.15 Another retrospective study found hydroxychloroquine to be safe and effective for VLP but noted a slow onset of action, with approximately 70% responding at 9 months following initiation of therapy.16
Recent attention has shifted to use of targeted therapies for VLP. For instance, apremilast has shown efficacy in a single-center, nonrandomized, open-label pilot study.17 Tildrakizumab, an IL-23 inhibitor, demonstrated efficacy in a case series involving 24 patients with VLP.18 Moreover, recent case reports and series have highlighted the potential of oral Janus kinase (JAK) inhibitors, such as tofacitinib, in VLP treatment.19 Clinical trials are ongoing to evaluate the safety and efficacy of topical ruxolitinib and deucravacitinib (a tyrosine kinase 2 inhibitor) in VLP.20-22 Systemic therapies for VLP currently are used off label, emphasizing the need for future randomized controlled trials to ascertain the optimal therapies for patients affected by erosive and nonerosive forms of this disease.
Plasma Cell Vulvitis
Plasma cell vulvitis is a chronic inflammatory disorder with an unknown etiology that some consider to be a variant of VLP.23 Others have observed an overlap with desquamative inflammatory vaginitis, categorizing PCV as a hemorrhagic vestibulovaginitis.24 Although its classification as a distinct entity remains under scrutiny, studies indicate a predilection for the nonkeratinized or partially keratinized vulva. A systematic review outlining common clinical findings reported that the most common anatomic sites included the vulvar vestibule, periurethral area, and labia minora.23 Additionally, reports have emphasized the association between PCV and other inflammatory vulvar skin conditions, including LS.25
Clinical Variants of PCV—A retrospective review proposed 2 clinical phenotypes for PCV: (1) primary non–lichen-associated PCV and (2) secondary lichen-associated PCV, which is linked to LS.26 The primary form is reported to be restricted to the vestibule, and the authors considered this a vulvar counterpart of atrophic vaginitis due to estrogen deficiency (now known as postmenopausal genitourinary syndrome). The secondary phenotype more commonly involved the vestibular and extravestibular epithelium.26
Management of PCV—Recognizing PCV in the context of LS may be important for identifying comorbid conditions and guiding treatment. However, evidence-based guidelines for PCV treatment are lacking. Commonly reported treatment modalities include clobetasol ointment 0.05% and tacrolimus ointment 0.1%.23 Successful treatment with hydrocortisone suppositories alternating with estradiol vaginal cream was reported in a recent case series.27 Crisaborole also has been reported as a treatment in 1 case of PCV.28 A recent case report found abrocitinib to be effective for the treatment of plasma cell balanitis in the setting of male genital LS,29 but there are limited data on the use of JAK inhibitors for PCV. Further research is necessary to ascertain the incidence, prevalence, clinical subtypes, and optimal management strategies for PCV to effectively treat patients with this condition.
Vulvar LSC
Similar to extragenital LSC, the evaluation of vulvar LSC should prioritize identification of underlying etiologies that contribute to the itch-scratch cycle, which may include psoriasis, atopic dermatitis, neurologic conditions, and allergic or irritant contact dermatitis.30,31 Although treatment strategies may vary based on underlying conditions, we will concentrate on updates in managing vulvar LSC and pruritus associated with an atopic diathesis or resulting from chronic contact dermatitis, which is prevalent in vulvar skin areas. Finally, we highlight some emerging vulvar allergens for consideration in clinical practice.
Management of Vulvar LSC—The advent of targeted therapies, including biologics and small-molecule inhibitors, for atopic dermatitis and prurigo nodularis in recent years presents potential options for treatment of individuals with vulvar LSC. However, studies on the use of these therapies specifically for vulvar LSC are limited, necessitating thorough discussions with patients. Given the debilitating nature of vulvar pruritus that may be seen in vulvar LSC and the potential inadequacy of topical steroids as monotherapy, systemic therapies may serve as alternative options for patients with refractory disease.30
Dupilumab, a dual inhibitor of IL-4 and IL-13 signaling, has shown rapid and sustained disease improvement in patients with atopic dermatitis, prurigo nodularis, and pruritus.32,33 Although data on its role in managing vulvar LSC are scarce, a recent case series reported improvement of vulvar pruritus with dupilumab.34 Similarly, tralokinumab, an IL-13 inhibitor approved by the US Food and Drug Administration (FDA) for atopic dermatitis, has shown efficacy in prurigo nodularis35 and may benefit patients with vulvar LSC, though studies on cutaneous outcomes in those with genital involvement specifically are lacking. Oral JAK inhibitors such as upadacitinib and abrocitinib—both FDA approved for atopic dermatitis—have demonstrated efficacy in treating LSC and itch, potentially serving as management options for vulvar LSC in cases resistant to topical steroids or in which steroid atrophy or other steroid adverse effects may preclude continued use of such agents.36,37 Finally, IL-31 inhibitors such as nemolizumab, which reduced the signs and symptoms of prurigo nodularis in a recent phase 3 clinical trial, may hold utility in addressing vulvar LSC and associated pruritus.38
The topical JAK inhibitor ruxolitinib, which is FDA approved for atopic dermatitis and vitiligo, holds promise for managing LSC on vulvar skin while mitigating the risk for steroid-induced atrophy.39 Additionally, nonsteroidal topicals including roflumilast cream 0.3% and tapinarof cream 1%, both FDA approved for psoriasis, are being evaluated in studies for their safety and efficacy in atopic dermatitis.40,41 These agents may have the potential to improve signs and symptoms of vulvar LSC, but further studies are necessary.
Vulvar Allergens and LSC—When assessing patients with vulvar LSC, it is crucial to recognize that allergic contact dermatitis is a common primary vulvar dermatosis but can coexist with other vulvar dermatoses such as LS.13,30 The vulvar skin’s susceptibly to allergic contact dermatitis is attributed to factors such as a higher ratio of antigen-presenting cells in the vulvar skin, the nonkeratinized nature of certain sites, and frequent contact with potential allergens.42,43 Therefore, incorporating patch testing into the diagnostic process should be considered when evaluating patients with vulvar skin conditions.43
A systemic review identified multiple vulvar allergens, including metals, topical medicaments, fragrances, preservatives, cosmetic constituents, and rubber components that led to contact dermatitis.44 Moreover, a recent analysis of topical preparations recommended by women with LS on social media found a high prevalence of known vulvar allergens in these agents, including botanical extracts/spices.45 Personal-care wipes marketed for vulvar care and hygiene are known to contain a variety of allergens, with a recent study finding numerous allergens in commercially available wipes including fragrances, scented botanicals in the form of essences, oils, fruit juices, and vitamin E.46 These findings underscore the importance of considering potential allergens when caring for patients with vulvar LSC and counseling patients about the potential allergens in many commercially available products that may be recommended on social media sites or by other sources.
Final Thoughts
Vulvar inflammatory dermatoses are becoming increasingly recognized, and there is a need to develop more effective diagnostic and treatment approaches. Recent literature has shed light on some of the challenges in the management of VLP, particularly its resistance to topical therapies and the importance of assessing and managing both cutaneous and vaginal involvement. Efforts have been made to refine the classification of PCV, with studies suggesting a variant that coexists with LS. Although evidence for vulvar-specific treatment of LSC is limited, the emergence of biologics and small-molecule inhibitors that are FDA approved for atopic dermatitis and prurigo nodularis offer promise for certain cases of vulvar LSC and vulvar pruritus. Moreover, recent developments in steroid-sparing topical agents warrant further investigation for their potential efficacy in treating vulvar LSC and possibly other vulvar inflammatory conditions in the future.
- Nguyen B, Kraus C. Vulvar lichen sclerosus: what’s new? Cutis. 2024;113:104-106. doi:10.12788/cutis.0967
- Van De Nieuwenhof HP, Meeuwis KAP, Nieboer TE, et al. The effect of vulvar lichen sclerosus on quality of life and sexual functioning. J Psychosom Obstet Gynaecol. 2010;31:279-284. doi:10.3109/0167482X.2010.507890
- Ranum A, Pearson DR. The impact of genital lichen sclerosus and lichen planus on quality of life: a review. Int J Womens Dermatol. 2022;8:E042. doi:10.1097/JW9.0000000000000042
- Messele F, Hinchee-Rodriguez K, Kraus CN. Vulvar dermatoses and depression: a systematic review of vulvar lichen sclerosus, lichen planus, and lichen simplex chronicus. JAAD Int. 2024;15:15-20. doi:10.1016/j.jdin.2023.10.009
- Choi UE, Nicholson RC, Agrawal P, et al. Involvement of vulva in lichen sclerosus increases the risk of antidepressant and benzodiazepine prescriptions for psychiatric disorder diagnoses. Int J Impot Res. Published online November 16, 2023. doi:10.1038/s41443-023-00793-3
- Saunderson R, Harris V, Yeh R, et al. Vulvar quality of life index (VQLI)—a simple tool to measure quality of life in patients with vulvar disease. Australas J Dermatol. 2020;61:152-157. doi:10.1111/ajd.13235
- Wu M, Kherlopian A, Wijaya M, et al. Quality of life impact and treatment response in vulval disease: comparison of 3 common conditions using the Vulval Quality of Life Index. Australas J Dermatol. 2022;63:E320-E328. doi:10.1111/ajd.13898
- Kherlopian A, Fischer G. Comparing quality of life in women with vulvovaginal lichen planus treated with topical and systemic treatments using the vulvar quality of life index. Australas J Dermatol. 2023;64:E125-E134. doi:10.1111/ajd.14032
- Cooper SM, Haefner HK, Abrahams-Gessel S, et al. Vulvovaginal lichen planus treatment: a survey of current practices. Arch Dermatol. 2008;144:1520-1521. doi:10.1001/archderm.144.11.1520
- Chow MR, Gill N, Alzahrani F, et al. Vulvar lichen planus–induced vulvovaginal stenosis: a case report and review of the literature. SAGE Open Med Case Rep. 2023;11:2050313X231164216. doi:10.1177/2050313X231164216
- Kherlopian A, Fischer G. Identifying predictors of systemic immunosuppressive treatment of vulvovaginal lichen planus: a retrospective cohort study of 122 women. Australas J Dermatol. 2022;63:335-343. doi:10.1111/ajd.13851
- Dunaway S, Tyler K, Kaffenberger, J. Update on treatments for erosive vulvovaginal lichen planus. Int J Dermatol. 2020;59:297-302. doi:10.1111/ijd.14692
- Mauskar MM, Marathe, K, Venkatesan A, et al. Vulvar diseases: conditions in adults and children. J Am Acad Dermatol. 2020;82:1287-1298. doi:10.1016/j.jaad.2019.10.077
- Hinchee-Rodriguez K, Duong A, Kraus CN. Local management strategies for inflammatory vaginitis in dermatologic conditions: suppositories, dilators, and estrogen replacement. JAAD Int. 2022;9:137-138. doi:10.1016/j.jdin.2022.09.004
- Hrin ML, Bowers NL, Feldman SR, et al. Mycophenolate mofetil versus methotrexate for vulvar lichen planus: a 10-year retrospective cohort study demonstrates comparable efficacy and tolerability. J Am Acad Dermatol. 2022;87:436-438. doi:10.1016/j.jaad.2021.08.061
- Vermeer HAB, Rashid H, Esajas MD, et al. The use of hydroxychloroquine as a systemic treatment in erosive lichen planus of the vulva and vagina. Br J Dermatol. 2021;185:201-203. doi:10.1111/bjd.19870
- Skullerud KH, Gjersvik P, Pripp AH, et al. Apremilast for genital erosive lichen planus in women (the AP-GELP Study): study protocol for a randomised placebo-controlled clinical trial. Trials. 2021;22:469. doi:10.1186/s13063-021-05428-w
- Kherlopian A, Fischer G. Successful treatment of vulvovaginal lichen planus with tildrakizumab: a case series of 24 patients. Australas J Dermatol. 2022;63:251-255. doi:10.1111/ajd.13793
- Kassels A, Edwards L, Kraus CN. Treatment of erosive vulvovaginal lichen planus with tofacitinib: a case series. JAAD Case Rep. 2023;40:14-18. doi:10.1016/j.jdcr.2023.08.001
- Wijaya M, Fischer G, Saunderson RB. The efficacy and safety of deucravacitinib compared to methotrexate, in patients with vulvar lichen planus who have failed topical therapy with potent corticosteroids: a study protocol for a single-centre double-blinded randomised controlled trial. Trials. 2024;25:181. doi:10.1186/s13063-024-08022-y
- Brumfiel CM, Patel MH, Severson KJ, et al. Ruxolitinib cream in the treatment of cutaneous lichen planus: a prospective, open-label study. J Invest Dermatol. 2022;142:2109-2116.e4. doi:10.1016/j.jid.2022.01.015
- A study to evaluate the efficacy and safety of ruxolitinib cream in participants with cutaneous lichen planus. ClinicalTrials.gov identifier: NCT05593432. Updated March 12, 2024. Accessed July 12, 2024. https://clinicaltrials.gov/study/NCT05593432
- Sattler S, Elsensohn AN, Mauskar MM, et al. Plasma cell vulvitis: a systematic review. Int J Womens Dermatol. 2021;7:756-762. doi:10.1016/j.ijwd.2021.04.005
- Song M, Day T, Kliman L, et al. Desquamative inflammatory vaginitis and plasma cell vulvitis represent a spectrum of hemorrhagic vestibulovaginitis. J Low Genit Tract Dis. 2022;26:60-67. doi:10.1097/LGT.0000000000000637
- Saeed L, Lee BA, Kraus CN. Tender solitary lesion in vulvar lichen sclerosus. JAAD Case Rep. 2022;23:61-63. doi:10.1016/j.jdcr.2022.01.038
- Wendling J, Plantier F, Moyal-Barracco M. Plasma cell vulvitis: a classification into two clinical phenotypes. J Low Genit Tract Dis. 2023;27:384-389. doi:10.1097/LGT.0000000000000771
- Prestwood CA, Granberry R, Rutherford A, et al. Successful treatment of plasma cell vulvitis: a case series. JAAD Case Rep. 2022;19:37-40. doi:10.1016/j.jdcr.2021.10.023
- He Y, Xu M, Wu M, et al. A case of plasma cell vulvitis successfully treated with crisaborole. J Dermatol. Published online April 1, 2024. doi:10.1111/1346-8138.17205
- Xiong X, Chen R, Wang L, et al. Treatment of plasma cell balanitis associated with male genital lichen sclerosus using abrocitinib. JAAD Case Rep. 2024;46:85-88. doi:10.1016/j.jdcr.2024.02.010
- Stewart KMA. Clinical care of vulvar pruritus, with emphasis on one common cause, lichen simplex chronicus. Dermatol Clin. 2010;28:669-680. doi:10.1016/j.det.2010.08.004
- Rimoin LP, Kwatra SG, Yosipovitch G. Female-specific pruritus from childhood to postmenopause: clinical features, hormonal factors, and treatment considerations. Dermatol Ther. 2013;26:157-167. doi:10.1111/dth.12034
- Simpson EL, Bieber T, Guttman-Yassky E, et al; SOLO 1 and SOLO 2 Investigators. Two phase 3 trials of dupilumab versus placebo in atopic dermatitis. N Engl J Med. 2016;375:2335-2348. doi:10.1056/NEJMoa1610020
- Yosipovitch G, Mollanazar N, Ständer S, et al. Dupilumab in patients with prurigo nodularis: two randomized, double-blind, placebo-controlled phase 3 trials. Nat Med. 2023;29:1180-1190. doi:10.1038/s41591-023-02320-9
- Gosch M, Cash S, Pichardo R. Vulvar pruritus improved with dupilumab. JSM Sexual Med. 2023;7:1104.
- Pezzolo E, Gambardella A, Guanti M, et al. Tralokinumab shows clinical improvement in patients with prurigo nodularis-like phenotype atopic dermatitis: a multicenter, prospective, open-label case series study. J Am Acad Dermatol. 2023;89:430-432. doi:10.1016/j.jaad.2023.04.056
- Simpson EL, Sinclair R, Forman S, et al. Efficacy and safety of abrocitinib in adults and adolescents with moderate-to-severe atopic dermatitis (JADE MONO-1): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet. 2020;396:255-266. doi:10.1016/S0140-6736(20)30732-7
- Simpson EL, Papp KA, Blauvelt A, et al. Efficacy and safety of upadacitinib in patients with moderate to severe atopic dermatitis: analysis of follow-up data from the Measure Up 1 and Measure Up 2 randomized clinical trials. JAMA Dermatol. 2022;158:404-413. doi:10.1001/jamadermatol.2022.0029
- Kwatra SG, Yosipovitch G, Legat FJ, et al. Phase 3 trial of nemolizumab in patients with prurigo nodularis. N Engl J Med. 2023;389:1579-1589. doi:10.1056/NEJMoa2301333
- Papp K, Szepietowski JC, Kircik L, et al. Long-term safety and disease control with ruxolitinib cream in atopic dermatitis: results from two phase 3 studies. J Am Acad Dermatol. 2023;88:1008-1016. doi:10.1016/j.jaad.2022.09.060
- Lebwohl MG, Kircik LH, Moore AY, et al. Effect of roflumilast cream vs vehicle cream on chronic plaque psoriasis: the DERMIS-1 and DERMIS-2 randomized clinical trials. JAMA. 2022;328:1073-1084. doi:10.1001/jama.2022.15632
- Lebwohl MG, Gold LS, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- O’Gorman SM, Torgerson RR. Allergic contact dermatitis of the vulva. Dermatitis. 2013;24:64-72. doi:10.1097/DER.0b013e318284da33
- Woodruff CM, Trivedi MK, Botto N, et al. Allergic contact dermatitis of the vulva. Dermatitis. 2018;29:233-243. doi:10.1097/DER.0000000000000339
- Vandeweege S, Debaene B, Lapeere H, et al. A systematic review of allergic and irritant contact dermatitis of the vulva: the most important allergens/irritants and the role of patch testing. Contact Dermatitis. 2023;88:249-262. doi:10.1111/cod.14258
- Luu Y, Admani S. Vulvar allergens in topical preparations recommended on social media: a cross-sectional analysis of Facebook groups for lichen sclerosus. Int J Womens Dermatol. 2023;9:E097. doi:10.1097/JW9.0000000000000097
- Newton J, Richardson S, van Oosbre AM, et al. A cross-sectional study of contact allergens in feminine hygiene wipes: a possible cause of vulvar contact dermatitis. Int J Womens Dermatol. 2022;8:E060. doi:10.1097/JW9.0000000000000060
Vulvar dermatoses continue to be an overlooked aspect of medical care, highlighting the necessity for enhanced diagnosis and management of these conditions. Here, we address recent advancements in understanding vulvar inflammatory dermatoses other than lichen sclerosus (LS), which was discussed in a prior Guest Editorial1—specifically vulvovaginal lichen planus (VLP), plasma cell vulvitis (PCV), and vulvar lichen simplex chronicus (LSC).
Vulvar Inflammatory Skin Disease and Quality of Life
There is an increased awareness of the impact vulvar skin disease has on quality of life and its association with anxiety and depression.2-5 Evaluating the burden of vulvar dermatoses remains an active area of research due to its significance in monitoring disease progression and assessing therapeutic effectiveness. Despite the existence of various dermatology quality-of-life assessment tools, many fail to adequately capture the unique impacts of vulvovaginal diseases, such as sexual or urinary dysfunction. The vulvar quality of life index, which was developed and validated by Saunderson et al6 in 2020, consists of a 15-item questionnaire spanning 4 domains: symptoms, anxiety, activities of daily living, and sexuality. This tool has been utilized to gauge treatment response in vulvar conditions and to compare disease burden of various vulvar dermatoses.7,8 Moving forward, integrating this tool into clinical studies on vulvar skin disease holds promise for enhancing our understanding and management of these conditions.
Vulvovaginal Lichen Planus
Vulvovaginal lichen planus is unique among several prevalent vulvar inflammatory skin disorders encountered by dermatologists—primarily due to its erosive form, which can extend to the vagina, resulting in noninfectious vaginitis and potential vaginal stenosis.9,10 Managing VLP poses a notable challenge, even when it is confined to the vulva, as it often proves resistant to topical therapies.11
Evaluation for Vaginal Mucosal Disease—In contrast to LS, which typically spares the vaginal mucosa, VLP can involve mucosal sites.9,12,13 Therefore, it is imperative that all patients with a diagnosis of vulvar VLP undergo evaluation for potential vaginal involvement through speculum examination, wet mount, or vaginal biopsy. Strategies to manage vaginal involvement include use of dilators and pelvic floor physical therapy, lysis of adhesions (if present), topical estrogen, and intravaginal corticosteroids—all tailored to the severity of the disease.9,11,14
Management of VLP—Approximately 20% to 40% of patients with VLP may require systemic therapy for disease management, including those who are younger, those of non-White ethnicity, and those presenting with vulvar pruritus.11 Various systemic immunosuppressants have been used for VLP, with a recent retrospective study revealing similar response rates for both methotrexate and mycophenolate mofetil in the treatment of VLP.15 Another retrospective study found hydroxychloroquine to be safe and effective for VLP but noted a slow onset of action, with approximately 70% responding at 9 months following initiation of therapy.16
Recent attention has shifted to use of targeted therapies for VLP. For instance, apremilast has shown efficacy in a single-center, nonrandomized, open-label pilot study.17 Tildrakizumab, an IL-23 inhibitor, demonstrated efficacy in a case series involving 24 patients with VLP.18 Moreover, recent case reports and series have highlighted the potential of oral Janus kinase (JAK) inhibitors, such as tofacitinib, in VLP treatment.19 Clinical trials are ongoing to evaluate the safety and efficacy of topical ruxolitinib and deucravacitinib (a tyrosine kinase 2 inhibitor) in VLP.20-22 Systemic therapies for VLP currently are used off label, emphasizing the need for future randomized controlled trials to ascertain the optimal therapies for patients affected by erosive and nonerosive forms of this disease.
Plasma Cell Vulvitis
Plasma cell vulvitis is a chronic inflammatory disorder with an unknown etiology that some consider to be a variant of VLP.23 Others have observed an overlap with desquamative inflammatory vaginitis, categorizing PCV as a hemorrhagic vestibulovaginitis.24 Although its classification as a distinct entity remains under scrutiny, studies indicate a predilection for the nonkeratinized or partially keratinized vulva. A systematic review outlining common clinical findings reported that the most common anatomic sites included the vulvar vestibule, periurethral area, and labia minora.23 Additionally, reports have emphasized the association between PCV and other inflammatory vulvar skin conditions, including LS.25
Clinical Variants of PCV—A retrospective review proposed 2 clinical phenotypes for PCV: (1) primary non–lichen-associated PCV and (2) secondary lichen-associated PCV, which is linked to LS.26 The primary form is reported to be restricted to the vestibule, and the authors considered this a vulvar counterpart of atrophic vaginitis due to estrogen deficiency (now known as postmenopausal genitourinary syndrome). The secondary phenotype more commonly involved the vestibular and extravestibular epithelium.26
Management of PCV—Recognizing PCV in the context of LS may be important for identifying comorbid conditions and guiding treatment. However, evidence-based guidelines for PCV treatment are lacking. Commonly reported treatment modalities include clobetasol ointment 0.05% and tacrolimus ointment 0.1%.23 Successful treatment with hydrocortisone suppositories alternating with estradiol vaginal cream was reported in a recent case series.27 Crisaborole also has been reported as a treatment in 1 case of PCV.28 A recent case report found abrocitinib to be effective for the treatment of plasma cell balanitis in the setting of male genital LS,29 but there are limited data on the use of JAK inhibitors for PCV. Further research is necessary to ascertain the incidence, prevalence, clinical subtypes, and optimal management strategies for PCV to effectively treat patients with this condition.
Vulvar LSC
Similar to extragenital LSC, the evaluation of vulvar LSC should prioritize identification of underlying etiologies that contribute to the itch-scratch cycle, which may include psoriasis, atopic dermatitis, neurologic conditions, and allergic or irritant contact dermatitis.30,31 Although treatment strategies may vary based on underlying conditions, we will concentrate on updates in managing vulvar LSC and pruritus associated with an atopic diathesis or resulting from chronic contact dermatitis, which is prevalent in vulvar skin areas. Finally, we highlight some emerging vulvar allergens for consideration in clinical practice.
Management of Vulvar LSC—The advent of targeted therapies, including biologics and small-molecule inhibitors, for atopic dermatitis and prurigo nodularis in recent years presents potential options for treatment of individuals with vulvar LSC. However, studies on the use of these therapies specifically for vulvar LSC are limited, necessitating thorough discussions with patients. Given the debilitating nature of vulvar pruritus that may be seen in vulvar LSC and the potential inadequacy of topical steroids as monotherapy, systemic therapies may serve as alternative options for patients with refractory disease.30
Dupilumab, a dual inhibitor of IL-4 and IL-13 signaling, has shown rapid and sustained disease improvement in patients with atopic dermatitis, prurigo nodularis, and pruritus.32,33 Although data on its role in managing vulvar LSC are scarce, a recent case series reported improvement of vulvar pruritus with dupilumab.34 Similarly, tralokinumab, an IL-13 inhibitor approved by the US Food and Drug Administration (FDA) for atopic dermatitis, has shown efficacy in prurigo nodularis35 and may benefit patients with vulvar LSC, though studies on cutaneous outcomes in those with genital involvement specifically are lacking. Oral JAK inhibitors such as upadacitinib and abrocitinib—both FDA approved for atopic dermatitis—have demonstrated efficacy in treating LSC and itch, potentially serving as management options for vulvar LSC in cases resistant to topical steroids or in which steroid atrophy or other steroid adverse effects may preclude continued use of such agents.36,37 Finally, IL-31 inhibitors such as nemolizumab, which reduced the signs and symptoms of prurigo nodularis in a recent phase 3 clinical trial, may hold utility in addressing vulvar LSC and associated pruritus.38
The topical JAK inhibitor ruxolitinib, which is FDA approved for atopic dermatitis and vitiligo, holds promise for managing LSC on vulvar skin while mitigating the risk for steroid-induced atrophy.39 Additionally, nonsteroidal topicals including roflumilast cream 0.3% and tapinarof cream 1%, both FDA approved for psoriasis, are being evaluated in studies for their safety and efficacy in atopic dermatitis.40,41 These agents may have the potential to improve signs and symptoms of vulvar LSC, but further studies are necessary.
Vulvar Allergens and LSC—When assessing patients with vulvar LSC, it is crucial to recognize that allergic contact dermatitis is a common primary vulvar dermatosis but can coexist with other vulvar dermatoses such as LS.13,30 The vulvar skin’s susceptibly to allergic contact dermatitis is attributed to factors such as a higher ratio of antigen-presenting cells in the vulvar skin, the nonkeratinized nature of certain sites, and frequent contact with potential allergens.42,43 Therefore, incorporating patch testing into the diagnostic process should be considered when evaluating patients with vulvar skin conditions.43
A systemic review identified multiple vulvar allergens, including metals, topical medicaments, fragrances, preservatives, cosmetic constituents, and rubber components that led to contact dermatitis.44 Moreover, a recent analysis of topical preparations recommended by women with LS on social media found a high prevalence of known vulvar allergens in these agents, including botanical extracts/spices.45 Personal-care wipes marketed for vulvar care and hygiene are known to contain a variety of allergens, with a recent study finding numerous allergens in commercially available wipes including fragrances, scented botanicals in the form of essences, oils, fruit juices, and vitamin E.46 These findings underscore the importance of considering potential allergens when caring for patients with vulvar LSC and counseling patients about the potential allergens in many commercially available products that may be recommended on social media sites or by other sources.
Final Thoughts
Vulvar inflammatory dermatoses are becoming increasingly recognized, and there is a need to develop more effective diagnostic and treatment approaches. Recent literature has shed light on some of the challenges in the management of VLP, particularly its resistance to topical therapies and the importance of assessing and managing both cutaneous and vaginal involvement. Efforts have been made to refine the classification of PCV, with studies suggesting a variant that coexists with LS. Although evidence for vulvar-specific treatment of LSC is limited, the emergence of biologics and small-molecule inhibitors that are FDA approved for atopic dermatitis and prurigo nodularis offer promise for certain cases of vulvar LSC and vulvar pruritus. Moreover, recent developments in steroid-sparing topical agents warrant further investigation for their potential efficacy in treating vulvar LSC and possibly other vulvar inflammatory conditions in the future.
Vulvar dermatoses continue to be an overlooked aspect of medical care, highlighting the necessity for enhanced diagnosis and management of these conditions. Here, we address recent advancements in understanding vulvar inflammatory dermatoses other than lichen sclerosus (LS), which was discussed in a prior Guest Editorial1—specifically vulvovaginal lichen planus (VLP), plasma cell vulvitis (PCV), and vulvar lichen simplex chronicus (LSC).
Vulvar Inflammatory Skin Disease and Quality of Life
There is an increased awareness of the impact vulvar skin disease has on quality of life and its association with anxiety and depression.2-5 Evaluating the burden of vulvar dermatoses remains an active area of research due to its significance in monitoring disease progression and assessing therapeutic effectiveness. Despite the existence of various dermatology quality-of-life assessment tools, many fail to adequately capture the unique impacts of vulvovaginal diseases, such as sexual or urinary dysfunction. The vulvar quality of life index, which was developed and validated by Saunderson et al6 in 2020, consists of a 15-item questionnaire spanning 4 domains: symptoms, anxiety, activities of daily living, and sexuality. This tool has been utilized to gauge treatment response in vulvar conditions and to compare disease burden of various vulvar dermatoses.7,8 Moving forward, integrating this tool into clinical studies on vulvar skin disease holds promise for enhancing our understanding and management of these conditions.
Vulvovaginal Lichen Planus
Vulvovaginal lichen planus is unique among several prevalent vulvar inflammatory skin disorders encountered by dermatologists—primarily due to its erosive form, which can extend to the vagina, resulting in noninfectious vaginitis and potential vaginal stenosis.9,10 Managing VLP poses a notable challenge, even when it is confined to the vulva, as it often proves resistant to topical therapies.11
Evaluation for Vaginal Mucosal Disease—In contrast to LS, which typically spares the vaginal mucosa, VLP can involve mucosal sites.9,12,13 Therefore, it is imperative that all patients with a diagnosis of vulvar VLP undergo evaluation for potential vaginal involvement through speculum examination, wet mount, or vaginal biopsy. Strategies to manage vaginal involvement include use of dilators and pelvic floor physical therapy, lysis of adhesions (if present), topical estrogen, and intravaginal corticosteroids—all tailored to the severity of the disease.9,11,14
Management of VLP—Approximately 20% to 40% of patients with VLP may require systemic therapy for disease management, including those who are younger, those of non-White ethnicity, and those presenting with vulvar pruritus.11 Various systemic immunosuppressants have been used for VLP, with a recent retrospective study revealing similar response rates for both methotrexate and mycophenolate mofetil in the treatment of VLP.15 Another retrospective study found hydroxychloroquine to be safe and effective for VLP but noted a slow onset of action, with approximately 70% responding at 9 months following initiation of therapy.16
Recent attention has shifted to use of targeted therapies for VLP. For instance, apremilast has shown efficacy in a single-center, nonrandomized, open-label pilot study.17 Tildrakizumab, an IL-23 inhibitor, demonstrated efficacy in a case series involving 24 patients with VLP.18 Moreover, recent case reports and series have highlighted the potential of oral Janus kinase (JAK) inhibitors, such as tofacitinib, in VLP treatment.19 Clinical trials are ongoing to evaluate the safety and efficacy of topical ruxolitinib and deucravacitinib (a tyrosine kinase 2 inhibitor) in VLP.20-22 Systemic therapies for VLP currently are used off label, emphasizing the need for future randomized controlled trials to ascertain the optimal therapies for patients affected by erosive and nonerosive forms of this disease.
Plasma Cell Vulvitis
Plasma cell vulvitis is a chronic inflammatory disorder with an unknown etiology that some consider to be a variant of VLP.23 Others have observed an overlap with desquamative inflammatory vaginitis, categorizing PCV as a hemorrhagic vestibulovaginitis.24 Although its classification as a distinct entity remains under scrutiny, studies indicate a predilection for the nonkeratinized or partially keratinized vulva. A systematic review outlining common clinical findings reported that the most common anatomic sites included the vulvar vestibule, periurethral area, and labia minora.23 Additionally, reports have emphasized the association between PCV and other inflammatory vulvar skin conditions, including LS.25
Clinical Variants of PCV—A retrospective review proposed 2 clinical phenotypes for PCV: (1) primary non–lichen-associated PCV and (2) secondary lichen-associated PCV, which is linked to LS.26 The primary form is reported to be restricted to the vestibule, and the authors considered this a vulvar counterpart of atrophic vaginitis due to estrogen deficiency (now known as postmenopausal genitourinary syndrome). The secondary phenotype more commonly involved the vestibular and extravestibular epithelium.26
Management of PCV—Recognizing PCV in the context of LS may be important for identifying comorbid conditions and guiding treatment. However, evidence-based guidelines for PCV treatment are lacking. Commonly reported treatment modalities include clobetasol ointment 0.05% and tacrolimus ointment 0.1%.23 Successful treatment with hydrocortisone suppositories alternating with estradiol vaginal cream was reported in a recent case series.27 Crisaborole also has been reported as a treatment in 1 case of PCV.28 A recent case report found abrocitinib to be effective for the treatment of plasma cell balanitis in the setting of male genital LS,29 but there are limited data on the use of JAK inhibitors for PCV. Further research is necessary to ascertain the incidence, prevalence, clinical subtypes, and optimal management strategies for PCV to effectively treat patients with this condition.
Vulvar LSC
Similar to extragenital LSC, the evaluation of vulvar LSC should prioritize identification of underlying etiologies that contribute to the itch-scratch cycle, which may include psoriasis, atopic dermatitis, neurologic conditions, and allergic or irritant contact dermatitis.30,31 Although treatment strategies may vary based on underlying conditions, we will concentrate on updates in managing vulvar LSC and pruritus associated with an atopic diathesis or resulting from chronic contact dermatitis, which is prevalent in vulvar skin areas. Finally, we highlight some emerging vulvar allergens for consideration in clinical practice.
Management of Vulvar LSC—The advent of targeted therapies, including biologics and small-molecule inhibitors, for atopic dermatitis and prurigo nodularis in recent years presents potential options for treatment of individuals with vulvar LSC. However, studies on the use of these therapies specifically for vulvar LSC are limited, necessitating thorough discussions with patients. Given the debilitating nature of vulvar pruritus that may be seen in vulvar LSC and the potential inadequacy of topical steroids as monotherapy, systemic therapies may serve as alternative options for patients with refractory disease.30
Dupilumab, a dual inhibitor of IL-4 and IL-13 signaling, has shown rapid and sustained disease improvement in patients with atopic dermatitis, prurigo nodularis, and pruritus.32,33 Although data on its role in managing vulvar LSC are scarce, a recent case series reported improvement of vulvar pruritus with dupilumab.34 Similarly, tralokinumab, an IL-13 inhibitor approved by the US Food and Drug Administration (FDA) for atopic dermatitis, has shown efficacy in prurigo nodularis35 and may benefit patients with vulvar LSC, though studies on cutaneous outcomes in those with genital involvement specifically are lacking. Oral JAK inhibitors such as upadacitinib and abrocitinib—both FDA approved for atopic dermatitis—have demonstrated efficacy in treating LSC and itch, potentially serving as management options for vulvar LSC in cases resistant to topical steroids or in which steroid atrophy or other steroid adverse effects may preclude continued use of such agents.36,37 Finally, IL-31 inhibitors such as nemolizumab, which reduced the signs and symptoms of prurigo nodularis in a recent phase 3 clinical trial, may hold utility in addressing vulvar LSC and associated pruritus.38
The topical JAK inhibitor ruxolitinib, which is FDA approved for atopic dermatitis and vitiligo, holds promise for managing LSC on vulvar skin while mitigating the risk for steroid-induced atrophy.39 Additionally, nonsteroidal topicals including roflumilast cream 0.3% and tapinarof cream 1%, both FDA approved for psoriasis, are being evaluated in studies for their safety and efficacy in atopic dermatitis.40,41 These agents may have the potential to improve signs and symptoms of vulvar LSC, but further studies are necessary.
Vulvar Allergens and LSC—When assessing patients with vulvar LSC, it is crucial to recognize that allergic contact dermatitis is a common primary vulvar dermatosis but can coexist with other vulvar dermatoses such as LS.13,30 The vulvar skin’s susceptibly to allergic contact dermatitis is attributed to factors such as a higher ratio of antigen-presenting cells in the vulvar skin, the nonkeratinized nature of certain sites, and frequent contact with potential allergens.42,43 Therefore, incorporating patch testing into the diagnostic process should be considered when evaluating patients with vulvar skin conditions.43
A systemic review identified multiple vulvar allergens, including metals, topical medicaments, fragrances, preservatives, cosmetic constituents, and rubber components that led to contact dermatitis.44 Moreover, a recent analysis of topical preparations recommended by women with LS on social media found a high prevalence of known vulvar allergens in these agents, including botanical extracts/spices.45 Personal-care wipes marketed for vulvar care and hygiene are known to contain a variety of allergens, with a recent study finding numerous allergens in commercially available wipes including fragrances, scented botanicals in the form of essences, oils, fruit juices, and vitamin E.46 These findings underscore the importance of considering potential allergens when caring for patients with vulvar LSC and counseling patients about the potential allergens in many commercially available products that may be recommended on social media sites or by other sources.
Final Thoughts
Vulvar inflammatory dermatoses are becoming increasingly recognized, and there is a need to develop more effective diagnostic and treatment approaches. Recent literature has shed light on some of the challenges in the management of VLP, particularly its resistance to topical therapies and the importance of assessing and managing both cutaneous and vaginal involvement. Efforts have been made to refine the classification of PCV, with studies suggesting a variant that coexists with LS. Although evidence for vulvar-specific treatment of LSC is limited, the emergence of biologics and small-molecule inhibitors that are FDA approved for atopic dermatitis and prurigo nodularis offer promise for certain cases of vulvar LSC and vulvar pruritus. Moreover, recent developments in steroid-sparing topical agents warrant further investigation for their potential efficacy in treating vulvar LSC and possibly other vulvar inflammatory conditions in the future.
- Nguyen B, Kraus C. Vulvar lichen sclerosus: what’s new? Cutis. 2024;113:104-106. doi:10.12788/cutis.0967
- Van De Nieuwenhof HP, Meeuwis KAP, Nieboer TE, et al. The effect of vulvar lichen sclerosus on quality of life and sexual functioning. J Psychosom Obstet Gynaecol. 2010;31:279-284. doi:10.3109/0167482X.2010.507890
- Ranum A, Pearson DR. The impact of genital lichen sclerosus and lichen planus on quality of life: a review. Int J Womens Dermatol. 2022;8:E042. doi:10.1097/JW9.0000000000000042
- Messele F, Hinchee-Rodriguez K, Kraus CN. Vulvar dermatoses and depression: a systematic review of vulvar lichen sclerosus, lichen planus, and lichen simplex chronicus. JAAD Int. 2024;15:15-20. doi:10.1016/j.jdin.2023.10.009
- Choi UE, Nicholson RC, Agrawal P, et al. Involvement of vulva in lichen sclerosus increases the risk of antidepressant and benzodiazepine prescriptions for psychiatric disorder diagnoses. Int J Impot Res. Published online November 16, 2023. doi:10.1038/s41443-023-00793-3
- Saunderson R, Harris V, Yeh R, et al. Vulvar quality of life index (VQLI)—a simple tool to measure quality of life in patients with vulvar disease. Australas J Dermatol. 2020;61:152-157. doi:10.1111/ajd.13235
- Wu M, Kherlopian A, Wijaya M, et al. Quality of life impact and treatment response in vulval disease: comparison of 3 common conditions using the Vulval Quality of Life Index. Australas J Dermatol. 2022;63:E320-E328. doi:10.1111/ajd.13898
- Kherlopian A, Fischer G. Comparing quality of life in women with vulvovaginal lichen planus treated with topical and systemic treatments using the vulvar quality of life index. Australas J Dermatol. 2023;64:E125-E134. doi:10.1111/ajd.14032
- Cooper SM, Haefner HK, Abrahams-Gessel S, et al. Vulvovaginal lichen planus treatment: a survey of current practices. Arch Dermatol. 2008;144:1520-1521. doi:10.1001/archderm.144.11.1520
- Chow MR, Gill N, Alzahrani F, et al. Vulvar lichen planus–induced vulvovaginal stenosis: a case report and review of the literature. SAGE Open Med Case Rep. 2023;11:2050313X231164216. doi:10.1177/2050313X231164216
- Kherlopian A, Fischer G. Identifying predictors of systemic immunosuppressive treatment of vulvovaginal lichen planus: a retrospective cohort study of 122 women. Australas J Dermatol. 2022;63:335-343. doi:10.1111/ajd.13851
- Dunaway S, Tyler K, Kaffenberger, J. Update on treatments for erosive vulvovaginal lichen planus. Int J Dermatol. 2020;59:297-302. doi:10.1111/ijd.14692
- Mauskar MM, Marathe, K, Venkatesan A, et al. Vulvar diseases: conditions in adults and children. J Am Acad Dermatol. 2020;82:1287-1298. doi:10.1016/j.jaad.2019.10.077
- Hinchee-Rodriguez K, Duong A, Kraus CN. Local management strategies for inflammatory vaginitis in dermatologic conditions: suppositories, dilators, and estrogen replacement. JAAD Int. 2022;9:137-138. doi:10.1016/j.jdin.2022.09.004
- Hrin ML, Bowers NL, Feldman SR, et al. Mycophenolate mofetil versus methotrexate for vulvar lichen planus: a 10-year retrospective cohort study demonstrates comparable efficacy and tolerability. J Am Acad Dermatol. 2022;87:436-438. doi:10.1016/j.jaad.2021.08.061
- Vermeer HAB, Rashid H, Esajas MD, et al. The use of hydroxychloroquine as a systemic treatment in erosive lichen planus of the vulva and vagina. Br J Dermatol. 2021;185:201-203. doi:10.1111/bjd.19870
- Skullerud KH, Gjersvik P, Pripp AH, et al. Apremilast for genital erosive lichen planus in women (the AP-GELP Study): study protocol for a randomised placebo-controlled clinical trial. Trials. 2021;22:469. doi:10.1186/s13063-021-05428-w
- Kherlopian A, Fischer G. Successful treatment of vulvovaginal lichen planus with tildrakizumab: a case series of 24 patients. Australas J Dermatol. 2022;63:251-255. doi:10.1111/ajd.13793
- Kassels A, Edwards L, Kraus CN. Treatment of erosive vulvovaginal lichen planus with tofacitinib: a case series. JAAD Case Rep. 2023;40:14-18. doi:10.1016/j.jdcr.2023.08.001
- Wijaya M, Fischer G, Saunderson RB. The efficacy and safety of deucravacitinib compared to methotrexate, in patients with vulvar lichen planus who have failed topical therapy with potent corticosteroids: a study protocol for a single-centre double-blinded randomised controlled trial. Trials. 2024;25:181. doi:10.1186/s13063-024-08022-y
- Brumfiel CM, Patel MH, Severson KJ, et al. Ruxolitinib cream in the treatment of cutaneous lichen planus: a prospective, open-label study. J Invest Dermatol. 2022;142:2109-2116.e4. doi:10.1016/j.jid.2022.01.015
- A study to evaluate the efficacy and safety of ruxolitinib cream in participants with cutaneous lichen planus. ClinicalTrials.gov identifier: NCT05593432. Updated March 12, 2024. Accessed July 12, 2024. https://clinicaltrials.gov/study/NCT05593432
- Sattler S, Elsensohn AN, Mauskar MM, et al. Plasma cell vulvitis: a systematic review. Int J Womens Dermatol. 2021;7:756-762. doi:10.1016/j.ijwd.2021.04.005
- Song M, Day T, Kliman L, et al. Desquamative inflammatory vaginitis and plasma cell vulvitis represent a spectrum of hemorrhagic vestibulovaginitis. J Low Genit Tract Dis. 2022;26:60-67. doi:10.1097/LGT.0000000000000637
- Saeed L, Lee BA, Kraus CN. Tender solitary lesion in vulvar lichen sclerosus. JAAD Case Rep. 2022;23:61-63. doi:10.1016/j.jdcr.2022.01.038
- Wendling J, Plantier F, Moyal-Barracco M. Plasma cell vulvitis: a classification into two clinical phenotypes. J Low Genit Tract Dis. 2023;27:384-389. doi:10.1097/LGT.0000000000000771
- Prestwood CA, Granberry R, Rutherford A, et al. Successful treatment of plasma cell vulvitis: a case series. JAAD Case Rep. 2022;19:37-40. doi:10.1016/j.jdcr.2021.10.023
- He Y, Xu M, Wu M, et al. A case of plasma cell vulvitis successfully treated with crisaborole. J Dermatol. Published online April 1, 2024. doi:10.1111/1346-8138.17205
- Xiong X, Chen R, Wang L, et al. Treatment of plasma cell balanitis associated with male genital lichen sclerosus using abrocitinib. JAAD Case Rep. 2024;46:85-88. doi:10.1016/j.jdcr.2024.02.010
- Stewart KMA. Clinical care of vulvar pruritus, with emphasis on one common cause, lichen simplex chronicus. Dermatol Clin. 2010;28:669-680. doi:10.1016/j.det.2010.08.004
- Rimoin LP, Kwatra SG, Yosipovitch G. Female-specific pruritus from childhood to postmenopause: clinical features, hormonal factors, and treatment considerations. Dermatol Ther. 2013;26:157-167. doi:10.1111/dth.12034
- Simpson EL, Bieber T, Guttman-Yassky E, et al; SOLO 1 and SOLO 2 Investigators. Two phase 3 trials of dupilumab versus placebo in atopic dermatitis. N Engl J Med. 2016;375:2335-2348. doi:10.1056/NEJMoa1610020
- Yosipovitch G, Mollanazar N, Ständer S, et al. Dupilumab in patients with prurigo nodularis: two randomized, double-blind, placebo-controlled phase 3 trials. Nat Med. 2023;29:1180-1190. doi:10.1038/s41591-023-02320-9
- Gosch M, Cash S, Pichardo R. Vulvar pruritus improved with dupilumab. JSM Sexual Med. 2023;7:1104.
- Pezzolo E, Gambardella A, Guanti M, et al. Tralokinumab shows clinical improvement in patients with prurigo nodularis-like phenotype atopic dermatitis: a multicenter, prospective, open-label case series study. J Am Acad Dermatol. 2023;89:430-432. doi:10.1016/j.jaad.2023.04.056
- Simpson EL, Sinclair R, Forman S, et al. Efficacy and safety of abrocitinib in adults and adolescents with moderate-to-severe atopic dermatitis (JADE MONO-1): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet. 2020;396:255-266. doi:10.1016/S0140-6736(20)30732-7
- Simpson EL, Papp KA, Blauvelt A, et al. Efficacy and safety of upadacitinib in patients with moderate to severe atopic dermatitis: analysis of follow-up data from the Measure Up 1 and Measure Up 2 randomized clinical trials. JAMA Dermatol. 2022;158:404-413. doi:10.1001/jamadermatol.2022.0029
- Kwatra SG, Yosipovitch G, Legat FJ, et al. Phase 3 trial of nemolizumab in patients with prurigo nodularis. N Engl J Med. 2023;389:1579-1589. doi:10.1056/NEJMoa2301333
- Papp K, Szepietowski JC, Kircik L, et al. Long-term safety and disease control with ruxolitinib cream in atopic dermatitis: results from two phase 3 studies. J Am Acad Dermatol. 2023;88:1008-1016. doi:10.1016/j.jaad.2022.09.060
- Lebwohl MG, Kircik LH, Moore AY, et al. Effect of roflumilast cream vs vehicle cream on chronic plaque psoriasis: the DERMIS-1 and DERMIS-2 randomized clinical trials. JAMA. 2022;328:1073-1084. doi:10.1001/jama.2022.15632
- Lebwohl MG, Gold LS, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- O’Gorman SM, Torgerson RR. Allergic contact dermatitis of the vulva. Dermatitis. 2013;24:64-72. doi:10.1097/DER.0b013e318284da33
- Woodruff CM, Trivedi MK, Botto N, et al. Allergic contact dermatitis of the vulva. Dermatitis. 2018;29:233-243. doi:10.1097/DER.0000000000000339
- Vandeweege S, Debaene B, Lapeere H, et al. A systematic review of allergic and irritant contact dermatitis of the vulva: the most important allergens/irritants and the role of patch testing. Contact Dermatitis. 2023;88:249-262. doi:10.1111/cod.14258
- Luu Y, Admani S. Vulvar allergens in topical preparations recommended on social media: a cross-sectional analysis of Facebook groups for lichen sclerosus. Int J Womens Dermatol. 2023;9:E097. doi:10.1097/JW9.0000000000000097
- Newton J, Richardson S, van Oosbre AM, et al. A cross-sectional study of contact allergens in feminine hygiene wipes: a possible cause of vulvar contact dermatitis. Int J Womens Dermatol. 2022;8:E060. doi:10.1097/JW9.0000000000000060
- Nguyen B, Kraus C. Vulvar lichen sclerosus: what’s new? Cutis. 2024;113:104-106. doi:10.12788/cutis.0967
- Van De Nieuwenhof HP, Meeuwis KAP, Nieboer TE, et al. The effect of vulvar lichen sclerosus on quality of life and sexual functioning. J Psychosom Obstet Gynaecol. 2010;31:279-284. doi:10.3109/0167482X.2010.507890
- Ranum A, Pearson DR. The impact of genital lichen sclerosus and lichen planus on quality of life: a review. Int J Womens Dermatol. 2022;8:E042. doi:10.1097/JW9.0000000000000042
- Messele F, Hinchee-Rodriguez K, Kraus CN. Vulvar dermatoses and depression: a systematic review of vulvar lichen sclerosus, lichen planus, and lichen simplex chronicus. JAAD Int. 2024;15:15-20. doi:10.1016/j.jdin.2023.10.009
- Choi UE, Nicholson RC, Agrawal P, et al. Involvement of vulva in lichen sclerosus increases the risk of antidepressant and benzodiazepine prescriptions for psychiatric disorder diagnoses. Int J Impot Res. Published online November 16, 2023. doi:10.1038/s41443-023-00793-3
- Saunderson R, Harris V, Yeh R, et al. Vulvar quality of life index (VQLI)—a simple tool to measure quality of life in patients with vulvar disease. Australas J Dermatol. 2020;61:152-157. doi:10.1111/ajd.13235
- Wu M, Kherlopian A, Wijaya M, et al. Quality of life impact and treatment response in vulval disease: comparison of 3 common conditions using the Vulval Quality of Life Index. Australas J Dermatol. 2022;63:E320-E328. doi:10.1111/ajd.13898
- Kherlopian A, Fischer G. Comparing quality of life in women with vulvovaginal lichen planus treated with topical and systemic treatments using the vulvar quality of life index. Australas J Dermatol. 2023;64:E125-E134. doi:10.1111/ajd.14032
- Cooper SM, Haefner HK, Abrahams-Gessel S, et al. Vulvovaginal lichen planus treatment: a survey of current practices. Arch Dermatol. 2008;144:1520-1521. doi:10.1001/archderm.144.11.1520
- Chow MR, Gill N, Alzahrani F, et al. Vulvar lichen planus–induced vulvovaginal stenosis: a case report and review of the literature. SAGE Open Med Case Rep. 2023;11:2050313X231164216. doi:10.1177/2050313X231164216
- Kherlopian A, Fischer G. Identifying predictors of systemic immunosuppressive treatment of vulvovaginal lichen planus: a retrospective cohort study of 122 women. Australas J Dermatol. 2022;63:335-343. doi:10.1111/ajd.13851
- Dunaway S, Tyler K, Kaffenberger, J. Update on treatments for erosive vulvovaginal lichen planus. Int J Dermatol. 2020;59:297-302. doi:10.1111/ijd.14692
- Mauskar MM, Marathe, K, Venkatesan A, et al. Vulvar diseases: conditions in adults and children. J Am Acad Dermatol. 2020;82:1287-1298. doi:10.1016/j.jaad.2019.10.077
- Hinchee-Rodriguez K, Duong A, Kraus CN. Local management strategies for inflammatory vaginitis in dermatologic conditions: suppositories, dilators, and estrogen replacement. JAAD Int. 2022;9:137-138. doi:10.1016/j.jdin.2022.09.004
- Hrin ML, Bowers NL, Feldman SR, et al. Mycophenolate mofetil versus methotrexate for vulvar lichen planus: a 10-year retrospective cohort study demonstrates comparable efficacy and tolerability. J Am Acad Dermatol. 2022;87:436-438. doi:10.1016/j.jaad.2021.08.061
- Vermeer HAB, Rashid H, Esajas MD, et al. The use of hydroxychloroquine as a systemic treatment in erosive lichen planus of the vulva and vagina. Br J Dermatol. 2021;185:201-203. doi:10.1111/bjd.19870
- Skullerud KH, Gjersvik P, Pripp AH, et al. Apremilast for genital erosive lichen planus in women (the AP-GELP Study): study protocol for a randomised placebo-controlled clinical trial. Trials. 2021;22:469. doi:10.1186/s13063-021-05428-w
- Kherlopian A, Fischer G. Successful treatment of vulvovaginal lichen planus with tildrakizumab: a case series of 24 patients. Australas J Dermatol. 2022;63:251-255. doi:10.1111/ajd.13793
- Kassels A, Edwards L, Kraus CN. Treatment of erosive vulvovaginal lichen planus with tofacitinib: a case series. JAAD Case Rep. 2023;40:14-18. doi:10.1016/j.jdcr.2023.08.001
- Wijaya M, Fischer G, Saunderson RB. The efficacy and safety of deucravacitinib compared to methotrexate, in patients with vulvar lichen planus who have failed topical therapy with potent corticosteroids: a study protocol for a single-centre double-blinded randomised controlled trial. Trials. 2024;25:181. doi:10.1186/s13063-024-08022-y
- Brumfiel CM, Patel MH, Severson KJ, et al. Ruxolitinib cream in the treatment of cutaneous lichen planus: a prospective, open-label study. J Invest Dermatol. 2022;142:2109-2116.e4. doi:10.1016/j.jid.2022.01.015
- A study to evaluate the efficacy and safety of ruxolitinib cream in participants with cutaneous lichen planus. ClinicalTrials.gov identifier: NCT05593432. Updated March 12, 2024. Accessed July 12, 2024. https://clinicaltrials.gov/study/NCT05593432
- Sattler S, Elsensohn AN, Mauskar MM, et al. Plasma cell vulvitis: a systematic review. Int J Womens Dermatol. 2021;7:756-762. doi:10.1016/j.ijwd.2021.04.005
- Song M, Day T, Kliman L, et al. Desquamative inflammatory vaginitis and plasma cell vulvitis represent a spectrum of hemorrhagic vestibulovaginitis. J Low Genit Tract Dis. 2022;26:60-67. doi:10.1097/LGT.0000000000000637
- Saeed L, Lee BA, Kraus CN. Tender solitary lesion in vulvar lichen sclerosus. JAAD Case Rep. 2022;23:61-63. doi:10.1016/j.jdcr.2022.01.038
- Wendling J, Plantier F, Moyal-Barracco M. Plasma cell vulvitis: a classification into two clinical phenotypes. J Low Genit Tract Dis. 2023;27:384-389. doi:10.1097/LGT.0000000000000771
- Prestwood CA, Granberry R, Rutherford A, et al. Successful treatment of plasma cell vulvitis: a case series. JAAD Case Rep. 2022;19:37-40. doi:10.1016/j.jdcr.2021.10.023
- He Y, Xu M, Wu M, et al. A case of plasma cell vulvitis successfully treated with crisaborole. J Dermatol. Published online April 1, 2024. doi:10.1111/1346-8138.17205
- Xiong X, Chen R, Wang L, et al. Treatment of plasma cell balanitis associated with male genital lichen sclerosus using abrocitinib. JAAD Case Rep. 2024;46:85-88. doi:10.1016/j.jdcr.2024.02.010
- Stewart KMA. Clinical care of vulvar pruritus, with emphasis on one common cause, lichen simplex chronicus. Dermatol Clin. 2010;28:669-680. doi:10.1016/j.det.2010.08.004
- Rimoin LP, Kwatra SG, Yosipovitch G. Female-specific pruritus from childhood to postmenopause: clinical features, hormonal factors, and treatment considerations. Dermatol Ther. 2013;26:157-167. doi:10.1111/dth.12034
- Simpson EL, Bieber T, Guttman-Yassky E, et al; SOLO 1 and SOLO 2 Investigators. Two phase 3 trials of dupilumab versus placebo in atopic dermatitis. N Engl J Med. 2016;375:2335-2348. doi:10.1056/NEJMoa1610020
- Yosipovitch G, Mollanazar N, Ständer S, et al. Dupilumab in patients with prurigo nodularis: two randomized, double-blind, placebo-controlled phase 3 trials. Nat Med. 2023;29:1180-1190. doi:10.1038/s41591-023-02320-9
- Gosch M, Cash S, Pichardo R. Vulvar pruritus improved with dupilumab. JSM Sexual Med. 2023;7:1104.
- Pezzolo E, Gambardella A, Guanti M, et al. Tralokinumab shows clinical improvement in patients with prurigo nodularis-like phenotype atopic dermatitis: a multicenter, prospective, open-label case series study. J Am Acad Dermatol. 2023;89:430-432. doi:10.1016/j.jaad.2023.04.056
- Simpson EL, Sinclair R, Forman S, et al. Efficacy and safety of abrocitinib in adults and adolescents with moderate-to-severe atopic dermatitis (JADE MONO-1): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet. 2020;396:255-266. doi:10.1016/S0140-6736(20)30732-7
- Simpson EL, Papp KA, Blauvelt A, et al. Efficacy and safety of upadacitinib in patients with moderate to severe atopic dermatitis: analysis of follow-up data from the Measure Up 1 and Measure Up 2 randomized clinical trials. JAMA Dermatol. 2022;158:404-413. doi:10.1001/jamadermatol.2022.0029
- Kwatra SG, Yosipovitch G, Legat FJ, et al. Phase 3 trial of nemolizumab in patients with prurigo nodularis. N Engl J Med. 2023;389:1579-1589. doi:10.1056/NEJMoa2301333
- Papp K, Szepietowski JC, Kircik L, et al. Long-term safety and disease control with ruxolitinib cream in atopic dermatitis: results from two phase 3 studies. J Am Acad Dermatol. 2023;88:1008-1016. doi:10.1016/j.jaad.2022.09.060
- Lebwohl MG, Kircik LH, Moore AY, et al. Effect of roflumilast cream vs vehicle cream on chronic plaque psoriasis: the DERMIS-1 and DERMIS-2 randomized clinical trials. JAMA. 2022;328:1073-1084. doi:10.1001/jama.2022.15632
- Lebwohl MG, Gold LS, Strober B, et al. Phase 3 trials of tapinarof cream for plaque psoriasis. N Engl J Med. 2021;385:2219-2229. doi:10.1056/NEJMoa2103629
- O’Gorman SM, Torgerson RR. Allergic contact dermatitis of the vulva. Dermatitis. 2013;24:64-72. doi:10.1097/DER.0b013e318284da33
- Woodruff CM, Trivedi MK, Botto N, et al. Allergic contact dermatitis of the vulva. Dermatitis. 2018;29:233-243. doi:10.1097/DER.0000000000000339
- Vandeweege S, Debaene B, Lapeere H, et al. A systematic review of allergic and irritant contact dermatitis of the vulva: the most important allergens/irritants and the role of patch testing. Contact Dermatitis. 2023;88:249-262. doi:10.1111/cod.14258
- Luu Y, Admani S. Vulvar allergens in topical preparations recommended on social media: a cross-sectional analysis of Facebook groups for lichen sclerosus. Int J Womens Dermatol. 2023;9:E097. doi:10.1097/JW9.0000000000000097
- Newton J, Richardson S, van Oosbre AM, et al. A cross-sectional study of contact allergens in feminine hygiene wipes: a possible cause of vulvar contact dermatitis. Int J Womens Dermatol. 2022;8:E060. doi:10.1097/JW9.0000000000000060
Insights From the 2020-2021 Dermatology Residency Match
To the Editor:
Data from the program director survey of the National Resident Matching Program offer key insights into the 2021 dermatology application process.1,2 Examination of data from the 2020 (N=12) and 2021 (N=17) program director survey regarding interviewing applicants revealed that specialty-specific letters of recommendation (LORs), personal prior knowledge of an applicant, and personal statement increased in importance by 17%, 7.4%, and 17%, respectively, whereas away rotations within the department decreased in importance by 44.9% (Table).1,2 Interestingly, for ranking applicants, programs decreased their emphasis on specialty-specific LORs by 25.8% and away rotations within the department by 22.7% and increased emphasis on personal statements by 14.7% and personal prior knowledge of an applicant by 0.8% from 2020 to 2021 (Table).1,2 These findings align with the prior recommendation to limit away rotations; data are contradictory—when comparing factors for interviewing as compared to ranking applicants—for specialty-specific LORs.
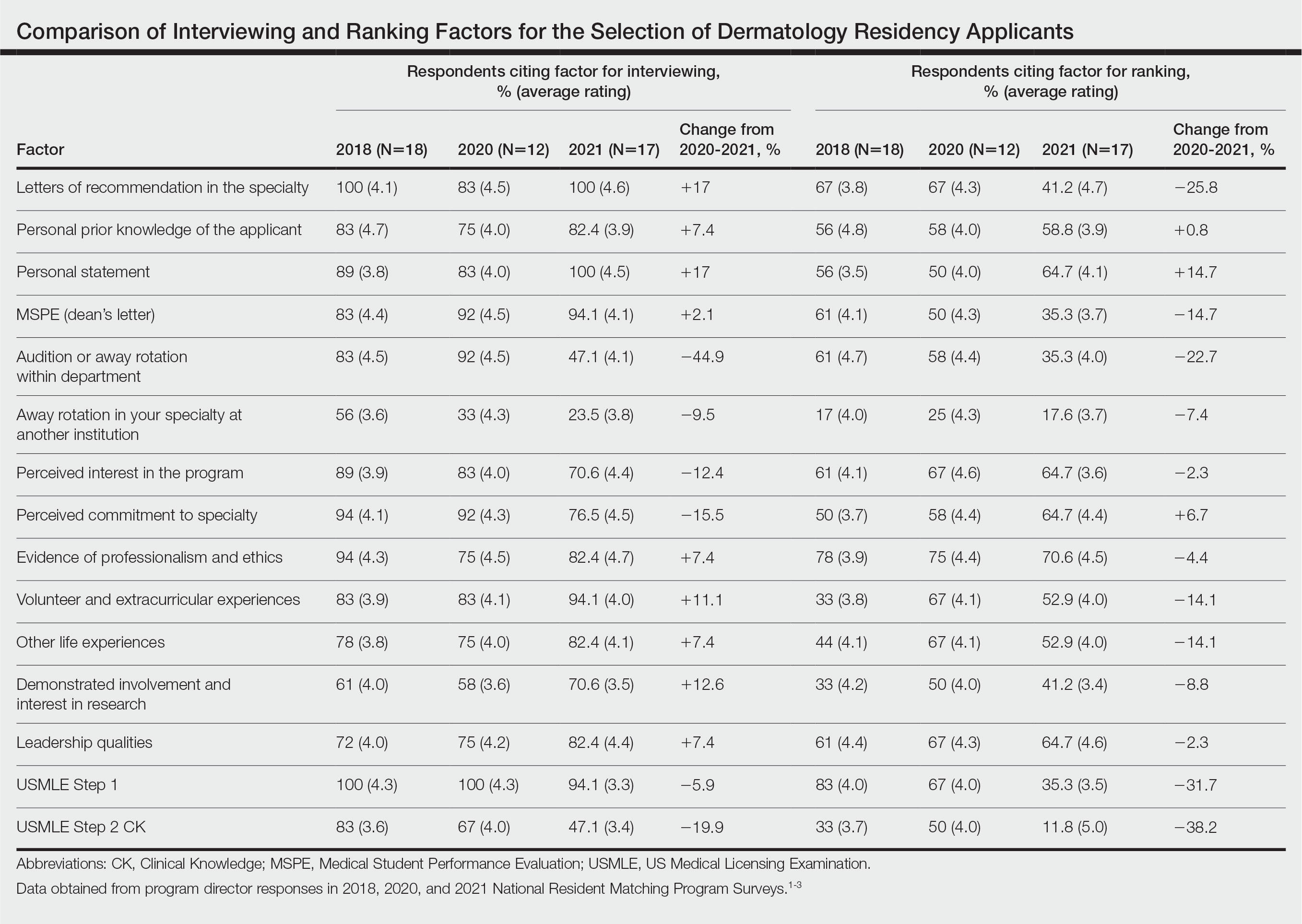
We further compared data from the otolaryngology cycle, which implemented preference signaling by which an applicant can signal their interest in a particular residency program in the 2021 Match, to data from dermatology with no preference signaling. A 90% probability of matching is estimated to require approximately 8 or 9 interviews for dermatology or 12 interviews for otolaryngology for MD senior students in 2020.4 In prior dermatology application cycles, the most highly qualified candidates constituted 7% to 21% of all applicants but were estimated to receive half of all interviews, causing a maldistribution of interviews.5,6
For the 2021 otolaryngology match, the Society of University Otolaryngologists implemented a novel preference signaling system that allowed candidates to show interest in programs by sending 5 preferences, or tokens.7 Recent data reports from the otolaryngology cycle demonstrated at least a 2-fold increase in the rate of receiving an interview invitation for signaled programs compared to the closest nonsignaled program if applicants were provided an additional token.7 Regarding overall applicant competitiveness (ie, dividing participants into quartiles based on their competitiveness), the highest increase in the overall rate of interview invitations (3.5 [total invitations/total applications]) was demonstrated for fourth-quartile (ie, “lowest quartile”) applicants compared with the increase in the overall rate of interview invitations seen in other quartiles (first quartile, an increase of 2.3; second quartile, an increase of 2.6; and third quartile, an increase of 2.4).7 We look forward to seeing the impact of preference signaling on the results of the 2022 dermatology cycle.
Despite changes in the interviewing process to accommodate COVID-19 pandemic safety recommendations, the overall dermatology postgraduate year (PGY) 2 fill rate remained unchanged from 2018 (98.6%) to 2021 (98.7%). Zero PGY-1 positions and 5 PGY-2 positions were unfilled in the 2021 Main Residency Match compared to 1 unfilled PGY-1 position and 4 unfilled PGY-2 positions in 2018.8 The coordinated interview invitation release, holistic review of applications, increased number of rankings, and virtual interviews might have helped offset potential obstacles imparted by inability to complete away rotations, inability to obtain LORs, and conducting interviews virtually.5
A limitation of our analysis is the low response rate of program directors to National Resident Matching Program surveys.
These strategies—holistic application review and coordinated interview release—may be considered in future cycles given their convenience and negligible impact on the dermatology match rate. For example, virtual interviews relieve the financial and time burdens of in-person interviews—approximately $10,000 for each US senior applicant—thus potentially allowing for a more equitable matching process.3 Inversely, in-person interviews allow participants to effectively network and form more meaningful connections while obtaining a better understanding of facilities and surrounding locales. As such, the medical community should continue to come to a consensus on the optimal format to host interviews.
- Results of the 2021 NRMP Program Director Survey. National Resident Matching Program. August 2021. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2021/11/2021-PD-Survey-Report-for-WWW.pdf
- Results of the 2020 NRMP Program Director Survey. National Resident Matching Program. August 2020. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2022/01/2020-PD-Survey.pdf
- Rojek NW, Shinkai K, Fett N. Dermatology faculty and residents’ perspectives on the dermatology residency application process: a nationwide survey. J Am Acad Dermatol. 2018;79:157-159. doi:10.1016/j.jaad.2018.01.00
- Charting Outcomes in the Match: Senior Students of U.S. MD Medical Schools. National Resident Matching Program. July 2020. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2021/08/Charting-Outcomes-in-the-Match-2020_MD-Senior_final.pdf
- Thatiparthi A, Martin A, Liu J, et al. Preliminary outcomes of 2020-2021 dermatology residency application cycle and adverse effects of COVID-19. J Am Acad Dermatol. 2021;84:e263-e264. doi:10.1016/j.jaad.2021.03.034
- Hammoud MM, Standiford T, Carmody JB. Potential implications of COVID-19 for the 2020-2021 residency application cycle. JAMA. 2020;324:29-30. doi:10.1001/jama.2020.8911
- Interview offer rate with/without ENTSignaling. Society of University Otolaryngologists. Updated July 19, 2022. Accessed December 12, 2022. https://opdo-hns.org/mpage/signaling-updates
- Results and Data: 2021 Main Residency Match. National Resident Matching Program. May 2021. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2021/08/MRM-Results_and-Data_2021.pdf
To the Editor:
Data from the program director survey of the National Resident Matching Program offer key insights into the 2021 dermatology application process.1,2 Examination of data from the 2020 (N=12) and 2021 (N=17) program director survey regarding interviewing applicants revealed that specialty-specific letters of recommendation (LORs), personal prior knowledge of an applicant, and personal statement increased in importance by 17%, 7.4%, and 17%, respectively, whereas away rotations within the department decreased in importance by 44.9% (Table).1,2 Interestingly, for ranking applicants, programs decreased their emphasis on specialty-specific LORs by 25.8% and away rotations within the department by 22.7% and increased emphasis on personal statements by 14.7% and personal prior knowledge of an applicant by 0.8% from 2020 to 2021 (Table).1,2 These findings align with the prior recommendation to limit away rotations; data are contradictory—when comparing factors for interviewing as compared to ranking applicants—for specialty-specific LORs.

We further compared data from the otolaryngology cycle, which implemented preference signaling by which an applicant can signal their interest in a particular residency program in the 2021 Match, to data from dermatology with no preference signaling. A 90% probability of matching is estimated to require approximately 8 or 9 interviews for dermatology or 12 interviews for otolaryngology for MD senior students in 2020.4 In prior dermatology application cycles, the most highly qualified candidates constituted 7% to 21% of all applicants but were estimated to receive half of all interviews, causing a maldistribution of interviews.5,6
For the 2021 otolaryngology match, the Society of University Otolaryngologists implemented a novel preference signaling system that allowed candidates to show interest in programs by sending 5 preferences, or tokens.7 Recent data reports from the otolaryngology cycle demonstrated at least a 2-fold increase in the rate of receiving an interview invitation for signaled programs compared to the closest nonsignaled program if applicants were provided an additional token.7 Regarding overall applicant competitiveness (ie, dividing participants into quartiles based on their competitiveness), the highest increase in the overall rate of interview invitations (3.5 [total invitations/total applications]) was demonstrated for fourth-quartile (ie, “lowest quartile”) applicants compared with the increase in the overall rate of interview invitations seen in other quartiles (first quartile, an increase of 2.3; second quartile, an increase of 2.6; and third quartile, an increase of 2.4).7 We look forward to seeing the impact of preference signaling on the results of the 2022 dermatology cycle.
Despite changes in the interviewing process to accommodate COVID-19 pandemic safety recommendations, the overall dermatology postgraduate year (PGY) 2 fill rate remained unchanged from 2018 (98.6%) to 2021 (98.7%). Zero PGY-1 positions and 5 PGY-2 positions were unfilled in the 2021 Main Residency Match compared to 1 unfilled PGY-1 position and 4 unfilled PGY-2 positions in 2018.8 The coordinated interview invitation release, holistic review of applications, increased number of rankings, and virtual interviews might have helped offset potential obstacles imparted by inability to complete away rotations, inability to obtain LORs, and conducting interviews virtually.5
A limitation of our analysis is the low response rate of program directors to National Resident Matching Program surveys.
These strategies—holistic application review and coordinated interview release—may be considered in future cycles given their convenience and negligible impact on the dermatology match rate. For example, virtual interviews relieve the financial and time burdens of in-person interviews—approximately $10,000 for each US senior applicant—thus potentially allowing for a more equitable matching process.3 Inversely, in-person interviews allow participants to effectively network and form more meaningful connections while obtaining a better understanding of facilities and surrounding locales. As such, the medical community should continue to come to a consensus on the optimal format to host interviews.
To the Editor:
Data from the program director survey of the National Resident Matching Program offer key insights into the 2021 dermatology application process.1,2 Examination of data from the 2020 (N=12) and 2021 (N=17) program director survey regarding interviewing applicants revealed that specialty-specific letters of recommendation (LORs), personal prior knowledge of an applicant, and personal statement increased in importance by 17%, 7.4%, and 17%, respectively, whereas away rotations within the department decreased in importance by 44.9% (Table).1,2 Interestingly, for ranking applicants, programs decreased their emphasis on specialty-specific LORs by 25.8% and away rotations within the department by 22.7% and increased emphasis on personal statements by 14.7% and personal prior knowledge of an applicant by 0.8% from 2020 to 2021 (Table).1,2 These findings align with the prior recommendation to limit away rotations; data are contradictory—when comparing factors for interviewing as compared to ranking applicants—for specialty-specific LORs.

We further compared data from the otolaryngology cycle, which implemented preference signaling by which an applicant can signal their interest in a particular residency program in the 2021 Match, to data from dermatology with no preference signaling. A 90% probability of matching is estimated to require approximately 8 or 9 interviews for dermatology or 12 interviews for otolaryngology for MD senior students in 2020.4 In prior dermatology application cycles, the most highly qualified candidates constituted 7% to 21% of all applicants but were estimated to receive half of all interviews, causing a maldistribution of interviews.5,6
For the 2021 otolaryngology match, the Society of University Otolaryngologists implemented a novel preference signaling system that allowed candidates to show interest in programs by sending 5 preferences, or tokens.7 Recent data reports from the otolaryngology cycle demonstrated at least a 2-fold increase in the rate of receiving an interview invitation for signaled programs compared to the closest nonsignaled program if applicants were provided an additional token.7 Regarding overall applicant competitiveness (ie, dividing participants into quartiles based on their competitiveness), the highest increase in the overall rate of interview invitations (3.5 [total invitations/total applications]) was demonstrated for fourth-quartile (ie, “lowest quartile”) applicants compared with the increase in the overall rate of interview invitations seen in other quartiles (first quartile, an increase of 2.3; second quartile, an increase of 2.6; and third quartile, an increase of 2.4).7 We look forward to seeing the impact of preference signaling on the results of the 2022 dermatology cycle.
Despite changes in the interviewing process to accommodate COVID-19 pandemic safety recommendations, the overall dermatology postgraduate year (PGY) 2 fill rate remained unchanged from 2018 (98.6%) to 2021 (98.7%). Zero PGY-1 positions and 5 PGY-2 positions were unfilled in the 2021 Main Residency Match compared to 1 unfilled PGY-1 position and 4 unfilled PGY-2 positions in 2018.8 The coordinated interview invitation release, holistic review of applications, increased number of rankings, and virtual interviews might have helped offset potential obstacles imparted by inability to complete away rotations, inability to obtain LORs, and conducting interviews virtually.5
A limitation of our analysis is the low response rate of program directors to National Resident Matching Program surveys.
These strategies—holistic application review and coordinated interview release—may be considered in future cycles given their convenience and negligible impact on the dermatology match rate. For example, virtual interviews relieve the financial and time burdens of in-person interviews—approximately $10,000 for each US senior applicant—thus potentially allowing for a more equitable matching process.3 Inversely, in-person interviews allow participants to effectively network and form more meaningful connections while obtaining a better understanding of facilities and surrounding locales. As such, the medical community should continue to come to a consensus on the optimal format to host interviews.
- Results of the 2021 NRMP Program Director Survey. National Resident Matching Program. August 2021. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2021/11/2021-PD-Survey-Report-for-WWW.pdf
- Results of the 2020 NRMP Program Director Survey. National Resident Matching Program. August 2020. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2022/01/2020-PD-Survey.pdf
- Rojek NW, Shinkai K, Fett N. Dermatology faculty and residents’ perspectives on the dermatology residency application process: a nationwide survey. J Am Acad Dermatol. 2018;79:157-159. doi:10.1016/j.jaad.2018.01.00
- Charting Outcomes in the Match: Senior Students of U.S. MD Medical Schools. National Resident Matching Program. July 2020. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2021/08/Charting-Outcomes-in-the-Match-2020_MD-Senior_final.pdf
- Thatiparthi A, Martin A, Liu J, et al. Preliminary outcomes of 2020-2021 dermatology residency application cycle and adverse effects of COVID-19. J Am Acad Dermatol. 2021;84:e263-e264. doi:10.1016/j.jaad.2021.03.034
- Hammoud MM, Standiford T, Carmody JB. Potential implications of COVID-19 for the 2020-2021 residency application cycle. JAMA. 2020;324:29-30. doi:10.1001/jama.2020.8911
- Interview offer rate with/without ENTSignaling. Society of University Otolaryngologists. Updated July 19, 2022. Accessed December 12, 2022. https://opdo-hns.org/mpage/signaling-updates
- Results and Data: 2021 Main Residency Match. National Resident Matching Program. May 2021. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2021/08/MRM-Results_and-Data_2021.pdf
- Results of the 2021 NRMP Program Director Survey. National Resident Matching Program. August 2021. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2021/11/2021-PD-Survey-Report-for-WWW.pdf
- Results of the 2020 NRMP Program Director Survey. National Resident Matching Program. August 2020. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2022/01/2020-PD-Survey.pdf
- Rojek NW, Shinkai K, Fett N. Dermatology faculty and residents’ perspectives on the dermatology residency application process: a nationwide survey. J Am Acad Dermatol. 2018;79:157-159. doi:10.1016/j.jaad.2018.01.00
- Charting Outcomes in the Match: Senior Students of U.S. MD Medical Schools. National Resident Matching Program. July 2020. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2021/08/Charting-Outcomes-in-the-Match-2020_MD-Senior_final.pdf
- Thatiparthi A, Martin A, Liu J, et al. Preliminary outcomes of 2020-2021 dermatology residency application cycle and adverse effects of COVID-19. J Am Acad Dermatol. 2021;84:e263-e264. doi:10.1016/j.jaad.2021.03.034
- Hammoud MM, Standiford T, Carmody JB. Potential implications of COVID-19 for the 2020-2021 residency application cycle. JAMA. 2020;324:29-30. doi:10.1001/jama.2020.8911
- Interview offer rate with/without ENTSignaling. Society of University Otolaryngologists. Updated July 19, 2022. Accessed December 12, 2022. https://opdo-hns.org/mpage/signaling-updates
- Results and Data: 2021 Main Residency Match. National Resident Matching Program. May 2021. Accessed December 6, 2021. https://www.nrmp.org/wp-content/uploads/2021/08/MRM-Results_and-Data_2021.pdf
PRACTICE POINTS
- Although there have been numerous changes to the dermatology interview process due to the COVID-19 pandemic, the overall fill rate for postgraduate year 2 positions remained unchanged from 2018 (prepandemic) to 2021 (postpandemic).
- Strategies to accommodate new safety recommendations for interviews may reduce the financial burden (approximately $10,000 for each senior applicant) and time constraints on applicants. These strategies should be considered for implementation in future cycles.