User login
The Alarm Burden of Excess Continuous Pulse Oximetry Monitoring Among Patients With Bronchiolitis
Practice guidelines discourage continuous pulse oximetry (SpO2) monitoring of patients with bronchiolitis who are not receiving supplemental oxygen.1,2 Overuse of SpO2 monitoring in this patient population has been associated with increased length of stay, unnecessary oxygen therapy, and excess hospital costs, without measurable patient benefit.3-5 In spite of this evidence base and expert guidance, nearly half of the more than 100,000 infants admitted for bronchiolitis each year receive excess SpO2 monitoring.6,7
Bronchiolitis guidelines suggest that guideline-discordant SpO2 monitoring may result in excess alarms, which disrupt families’ sleep and engender alarm fatigue among staff.1 Pediatric nurses receive up to 155 alarms per monitored patient per day.8,9 Frequent alarms are associated with slower nurse response times10,11 and increased nurse subjective workload.12
Methods
Cohort
We retrospectively evaluated SpO2 monitoring patterns and alarm rates of children 0 to 24 months old admitted to a general pediatrics service at a tertiary care children’s hospital. We included patients who had a discharge diagnosis of bronchiolitis (International Classification of Diseases, Tenth Revision codes J45x, T17.2x, T17.3x, T17.4x, T17.5x, T17.8x, T17.9x, A37xx, J04x, J05x, J05.1x, J69.0x, J69.1x, J69.8x) between November 24, 2019, and January 21, 2020, the period of time during which alarm data and monitor data were concurrently available for analysis. In order to conservatively assure applicability of clinical practice guidelines, we excluded patients with discharge diagnoses that included other respiratory conditions (eg, reactive airway disease), patients with complex chronic conditions (CCC) as defined by the CCC version 2 classification system,13 and patients with intensive care unit (ICU) stays during the admission.
Time
Flowsheet data detailing nursing respiratory assessments were extracted from the electronic health record (EHR) database (Clarity, Epic Systems). Using previously validated methodology,14 we identified minutes during which patients received supplemental oxygen or high-flow nasal cannula (supplemental oxygen) based on the documented fraction of inspired oxygen (FiO2), flow rate, and support devices. We then identified the final discontinuation of respiratory support during the hospital admission, and censored the 60 minutes after final discontinuation of supplemental oxygen based upon recent monitoring guidelines.2 Minutes up to an hour after supplemental oxygen discontinuation were classified as receiving supplemental oxygen and not included in our analysis. Only minutes between the end of the censored period and hospital discharge were used in the analysis. For patients who never received respiratory support during the admission, we censored the first 60 minutes of the admission and analyzed the remainder of their stay.
SpO2 Monitoring
We used device-integrated, physiologic-monitor, vital sign data sent each minute from the General Electric monitor network to the EHR to identify minutes during which patients were connected to physiologic monitors and transmitting signals from SpO2 sensors. We extracted minute-level SpO2 data from the hospital clinical data warehouse (CDW). Minutes in which SpO2 data were present were classified as “monitored,” an approach previously validated using in-person observation.14
To categorize time as “not receiving supplemental oxygen and continuously monitored (guideline-discordant monitoring),” or “not receiving supplemental oxygen and not continuously monitored (guideline-concordant intermittent measurement),” we evaluated the percent of minutes within an hour during which the patient received SpO2 monitoring and applied an a priori conservative rule. Hours during which ≥90% of minutes had SpO2 monitoring data were classified as “continuously monitored.” Hours during which ≤10% of minutes had SpO2 monitoring data were classified as “intermittently measured.” Hours during which 11% to 89% of minutes included monitor data were excluded from further analysis. The number of continuously monitored hours was tabulated for each patient. The median number of continuously monitored hours was computed; results were stratified by prior receipt of respiratory support.
Alarm Counts
Minute-level monitor alarm counts (the absolute number of abnormal vital signs that triggered a monitor to alarm) were extracted from the CDW. Alarm counts were tabulated for each patient hour. For each patient, the alarm rate (total number of alarms divided by time) was computed for continuously monitored and intermittently measured time. Results were stratified by prior receipt of respiratory support.
The study was reviewed by the institutional review board and determined to meet exemption criteria.
Results
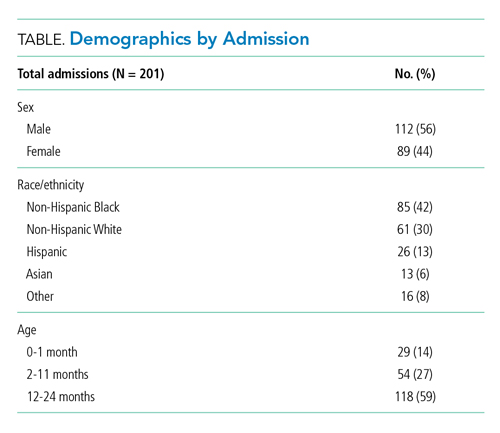
Our cohort included 201 admissions by 197 unique patients (Table). We evaluated 4402 hours that occurred ≥60 minutes following final discontinuation of supplemental oxygen, the time period during which guidelines discourage routine use of continuous SpO2 monitoring. This represented a median of 19 hours (interquartile range [IQR], 14-25) per admission. We excluded 474 hours (11%) that could not be classified as either continuously or intermittently measured.
During time ≥60 minutes following discontinuation of supplemental oxygen, our cohort experienced 1537 hours of guideline-discordant continuous monitoring, a median of 6 hours (IQR, 3-12) per admission. Patients experienced a median of 12 hours (IQR, 5-17) of intermittent measurement. Among patients who received supplemental oxygen, 91% experienced guideline-discordant continuous SpO2 monitoring, as compared to 68% of patients who did not receive supplemental oxygen. Among those who received guideline-discordant continuous SpO2 monitoring, the duration of this monitoring did not differ significantly between those who had received supplemental oxygen during the admission and those who had not.
During classifiable time ≥60 minutes following discontinuation of supplemental oxygen, our cohort experienced 14,371 alarms; 77% (11,101) of these alarms were generated during periods of guideline-discordant continuous monitoring. The median hourly alarm rate during these periods of guideline-discordant continuous monitoring was 6.7 alarms per hour (IQR, 2.1-12.3), representing a median of 35 (IQR, 10-81) additional alarms per patient. During periods of guideline-concordant intermittent measurement, the median hourly alarm rate was 0.5 (IQR, 0.1-0.8), with a median of 5 (IQR, 1-13) alarms per patient.
Those who received supplemental oxygen earlier in the admission had higher alarm rates during continuously monitored time (7.3 per hour [IQR, 2.7-12.7]) than patients who had not received supplemental oxygen (3.3 per hour [IQR, 0.6-11.8]), likely reflecting clinical differences between these patient populations. The most frequent alarm type among continuously monitored patients who had previously received supplemental oxygen was “SpO2 low.”
Discussion
Across 4402 patient hours, guideline-discordant continuous SpO2 monitoring of patients with bronchiolitis resulted in 11,101 alarms, at a rate of approximately 1 additional alarm every 9 minutes. Patients in our cohort received a median of 6 hours of guideline-discordant monitoring, which imposes a significant alarm burden that is potentially modifiable using targeted reduction strategies.15
Transient, self-resolved hypoxemia is a common feature of bronchiolitis and likely of little clinical consequence.16 Therefore, this rate of hypoxemia alarms is not unexpected. Though we evaluated only the period of time following final discontinuation of respiratory support, this finding is in keeping with the literature associating excess physiologic monitoring of patients with bronchiolitis with unnecessary oxygen therapy and increased length of stay,3-5 largely because clinicians may feel compelled to respond to hypoxemia alarms with either supplemental oxygen or longer monitoring.
Our findings must be contextualized in light of the limitations of our approach. We did not evaluate nurse workload associated with guideline-discordant continuous SpO2 monitoring. Prior work conducted by our lab has demonstrated that when nurses experience more than 40 alarms within a 2-hour period, their measured subjective workload increases to a degree associated with missing important tasks, threatening the quality and safety of the care they deliver.12,17 Given that nurses care for multiple patients, it is likely that the excess alarms introduced by guideline-discordant continuous monitoring contribute to increased nurse workload and alarm fatigue.
Similarly, we could not evaluate whether the alarms nurses experienced were actionable. Although our lab has previously reported that ≥99% of alarms occurring on non-ICU pediatric wards are nonactionable,10,11 it is possible that some of the alarms during guideline-discordant monitoring periods required action. However, it is unlikely that any life-sustaining actions were taken because (1) we only evaluated time >60 minutes after final discontinuation of supplemental oxygen, so by definition none of these alarms required treatment with supplemental oxygen, and (2) none of the patients we included received ICU care during their admission.
The avoidable alarm burden identified in our analysis suggests that eliminating continuous SpO2 monitoring overuse in bronchiolitis will likely reduce nurses’ workload and alarm fatigue in hospital settings that care for children with bronchiolitis.
1. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
2. Schondelmeyer AC, Dewan ML, Brady PW, et al. Cardiorespiratory and pulse oximetry monitoring in hospitalized children: a Delphi process. Pediatrics. 2020;146(2):e20193336. https://doi.org/10.1542/peds.2019-3336
3. Cunningham S, Rodriguez A, Boyd KA, McIntosh E, Lewis SC, BIDS Collaborators Group. Bronchiolitis of Infancy Discharge Study (BIDS): A multicentre, parallel-group, double-blind, randomised controlled, equivalence trial with economic evaluation. Health Technol Assess. 2015;19(71):i-xxiii, 1-172. https://doi.org/10.3310/hta19710
4. McCulloh R, Koster M, Ralston S, et al. Use of intermittent vs continuous pulse oximetry for nonhypoxemic infants and young children hospitalized for bronchiolitis: a randomized clinical trial. JAMA Pediatr. 2015;169(10):898-904. https://doi.org/10.1001/jamapediatrics.2015.1746
5. Schuh S, Freedman S, Coates A, et al. Effect of oximetry on hospitalization in bronchiolitis: a randomized clinical trial. JAMA. 2014;312(7):712-718. https://doi.org/10.1001/jama.2014.8637
6. Fujiogi M, Goto T, Yasunaga H, et al. Trends in bronchiolitis hospitalizations in the United States: 2000–2016. Pediatrics. 2019;144(6):e20192614. https://doi.org/10.1542/peds.2019-2614
7. Bonafide CP, Xiao R, Brady PW, et al. Prevalence of continuous pulse oximetry monitoring in hospitalized children with bronchiolitis not requiring supplemental oxygen. JAMA. 2020;323(15):1467-1477. https://doi.org/10.1001/jama.2020.2998
8. Schondelmeyer AC, Brady PW, Goel VV, et al. Physiologic monitor alarm rates at 5 children’s hospitals. J Hosp Med. 2018;13(6):396-398. https://doi.org/10.12788/jhm.2918
9. Schondelmeyer AC, Bonafide CP, Goel VV, et al. The frequency of physiologic monitor alarms in a children’s hospital. J Hosp Med. 2016;11(11):796-798. https://doi.org/10.1002/jhm.2612
10. Bonafide CP, Lin R, Zander M, et al. Association between exposure to nonactionable physiologic monitor alarms and response time in a children’s hospital. J Hosp Med. 2015;10(6):345-351. https://doi.org/10.1002/jhm.2331
11. Bonafide CP, Localio AR, Holmes JH, et al. Video analysis of factors associated with response time to physiologic monitor alarms in a children’s hospital. JAMA Pediatr. 2017;171(6):524-531. https://doi.org/10.1001/jamapediatrics.2016.5123
12. Rasooly IR, Kern-Goldberger AS, Xiao R, et al. Physiologic monitor alarm burden and nurses’ subjective workload in a children’s hospital. Hosp Pediatr. 2021;11(7):703-710. https://doi.org/10.1542/hpeds.2020-003509
13. Feudtner C, Feinstein JA, Zhong W, Hall M, Dai D. Pediatric complex chronic conditions classification system version 2: updated for ICD-10 and complex medical technology dependence and transplantation. BMC Pediatr. 2014;14:199. https://doi.org/10.1186/1471-2431-14-199
14. Kern-Goldberger AS, Rasooly IR, Luo B, et al. EHR-integrated monitor data to measure pulse oximetry use in bronchiolitis. Hosp Pediatr. 2021;11(10):1073-1082. https://doi.org/10.1542/hpeds.2021-005894
15. Schondelmeyer AC, Bettencourt AP, Xiao R, et al. Evaluation of an educational outreach and audit and feedback program to reduce continuous pulse oximetry use in hospitalized infants with stable bronchiolitis. JAMA Netw Open. 2021;4(9):e2122826. https://doi.org/10.1001/jamanetworkopen.2021.22826
16. Principi T, Coates AL, Parkin PC, Stephens D, DaSilva Z, Schuh S. Effect of oxygen desaturations on subsequent medical visits in infants discharged from the emergency department with bronchiolitis. JAMA Pediatr. 2016;170(6):602-608. https://doi.org/10.1001/jamapediatrics.2016.0114
17. Tubbs-Cooley HL, Mara CA, Carle AC, Mark BA, Pickler RH. Association of nurse workload with missed nursing care in the neonatal intensive care unit. JAMA Pediatr. 2019;173(1):44-51. https://doi.org/10.1001/jamapediatrics.2018.3619
Practice guidelines discourage continuous pulse oximetry (SpO2) monitoring of patients with bronchiolitis who are not receiving supplemental oxygen.1,2 Overuse of SpO2 monitoring in this patient population has been associated with increased length of stay, unnecessary oxygen therapy, and excess hospital costs, without measurable patient benefit.3-5 In spite of this evidence base and expert guidance, nearly half of the more than 100,000 infants admitted for bronchiolitis each year receive excess SpO2 monitoring.6,7
Bronchiolitis guidelines suggest that guideline-discordant SpO2 monitoring may result in excess alarms, which disrupt families’ sleep and engender alarm fatigue among staff.1 Pediatric nurses receive up to 155 alarms per monitored patient per day.8,9 Frequent alarms are associated with slower nurse response times10,11 and increased nurse subjective workload.12
Methods
Cohort
We retrospectively evaluated SpO2 monitoring patterns and alarm rates of children 0 to 24 months old admitted to a general pediatrics service at a tertiary care children’s hospital. We included patients who had a discharge diagnosis of bronchiolitis (International Classification of Diseases, Tenth Revision codes J45x, T17.2x, T17.3x, T17.4x, T17.5x, T17.8x, T17.9x, A37xx, J04x, J05x, J05.1x, J69.0x, J69.1x, J69.8x) between November 24, 2019, and January 21, 2020, the period of time during which alarm data and monitor data were concurrently available for analysis. In order to conservatively assure applicability of clinical practice guidelines, we excluded patients with discharge diagnoses that included other respiratory conditions (eg, reactive airway disease), patients with complex chronic conditions (CCC) as defined by the CCC version 2 classification system,13 and patients with intensive care unit (ICU) stays during the admission.
Time
Flowsheet data detailing nursing respiratory assessments were extracted from the electronic health record (EHR) database (Clarity, Epic Systems). Using previously validated methodology,14 we identified minutes during which patients received supplemental oxygen or high-flow nasal cannula (supplemental oxygen) based on the documented fraction of inspired oxygen (FiO2), flow rate, and support devices. We then identified the final discontinuation of respiratory support during the hospital admission, and censored the 60 minutes after final discontinuation of supplemental oxygen based upon recent monitoring guidelines.2 Minutes up to an hour after supplemental oxygen discontinuation were classified as receiving supplemental oxygen and not included in our analysis. Only minutes between the end of the censored period and hospital discharge were used in the analysis. For patients who never received respiratory support during the admission, we censored the first 60 minutes of the admission and analyzed the remainder of their stay.
SpO2 Monitoring
We used device-integrated, physiologic-monitor, vital sign data sent each minute from the General Electric monitor network to the EHR to identify minutes during which patients were connected to physiologic monitors and transmitting signals from SpO2 sensors. We extracted minute-level SpO2 data from the hospital clinical data warehouse (CDW). Minutes in which SpO2 data were present were classified as “monitored,” an approach previously validated using in-person observation.14
To categorize time as “not receiving supplemental oxygen and continuously monitored (guideline-discordant monitoring),” or “not receiving supplemental oxygen and not continuously monitored (guideline-concordant intermittent measurement),” we evaluated the percent of minutes within an hour during which the patient received SpO2 monitoring and applied an a priori conservative rule. Hours during which ≥90% of minutes had SpO2 monitoring data were classified as “continuously monitored.” Hours during which ≤10% of minutes had SpO2 monitoring data were classified as “intermittently measured.” Hours during which 11% to 89% of minutes included monitor data were excluded from further analysis. The number of continuously monitored hours was tabulated for each patient. The median number of continuously monitored hours was computed; results were stratified by prior receipt of respiratory support.
Alarm Counts
Minute-level monitor alarm counts (the absolute number of abnormal vital signs that triggered a monitor to alarm) were extracted from the CDW. Alarm counts were tabulated for each patient hour. For each patient, the alarm rate (total number of alarms divided by time) was computed for continuously monitored and intermittently measured time. Results were stratified by prior receipt of respiratory support.
The study was reviewed by the institutional review board and determined to meet exemption criteria.
Results
Our cohort included 201 admissions by 197 unique patients (Table). We evaluated 4402 hours that occurred ≥60 minutes following final discontinuation of supplemental oxygen, the time period during which guidelines discourage routine use of continuous SpO2 monitoring. This represented a median of 19 hours (interquartile range [IQR], 14-25) per admission. We excluded 474 hours (11%) that could not be classified as either continuously or intermittently measured.

During time ≥60 minutes following discontinuation of supplemental oxygen, our cohort experienced 1537 hours of guideline-discordant continuous monitoring, a median of 6 hours (IQR, 3-12) per admission. Patients experienced a median of 12 hours (IQR, 5-17) of intermittent measurement. Among patients who received supplemental oxygen, 91% experienced guideline-discordant continuous SpO2 monitoring, as compared to 68% of patients who did not receive supplemental oxygen. Among those who received guideline-discordant continuous SpO2 monitoring, the duration of this monitoring did not differ significantly between those who had received supplemental oxygen during the admission and those who had not.
During classifiable time ≥60 minutes following discontinuation of supplemental oxygen, our cohort experienced 14,371 alarms; 77% (11,101) of these alarms were generated during periods of guideline-discordant continuous monitoring. The median hourly alarm rate during these periods of guideline-discordant continuous monitoring was 6.7 alarms per hour (IQR, 2.1-12.3), representing a median of 35 (IQR, 10-81) additional alarms per patient. During periods of guideline-concordant intermittent measurement, the median hourly alarm rate was 0.5 (IQR, 0.1-0.8), with a median of 5 (IQR, 1-13) alarms per patient.
Those who received supplemental oxygen earlier in the admission had higher alarm rates during continuously monitored time (7.3 per hour [IQR, 2.7-12.7]) than patients who had not received supplemental oxygen (3.3 per hour [IQR, 0.6-11.8]), likely reflecting clinical differences between these patient populations. The most frequent alarm type among continuously monitored patients who had previously received supplemental oxygen was “SpO2 low.”
Discussion
Across 4402 patient hours, guideline-discordant continuous SpO2 monitoring of patients with bronchiolitis resulted in 11,101 alarms, at a rate of approximately 1 additional alarm every 9 minutes. Patients in our cohort received a median of 6 hours of guideline-discordant monitoring, which imposes a significant alarm burden that is potentially modifiable using targeted reduction strategies.15
Transient, self-resolved hypoxemia is a common feature of bronchiolitis and likely of little clinical consequence.16 Therefore, this rate of hypoxemia alarms is not unexpected. Though we evaluated only the period of time following final discontinuation of respiratory support, this finding is in keeping with the literature associating excess physiologic monitoring of patients with bronchiolitis with unnecessary oxygen therapy and increased length of stay,3-5 largely because clinicians may feel compelled to respond to hypoxemia alarms with either supplemental oxygen or longer monitoring.
Our findings must be contextualized in light of the limitations of our approach. We did not evaluate nurse workload associated with guideline-discordant continuous SpO2 monitoring. Prior work conducted by our lab has demonstrated that when nurses experience more than 40 alarms within a 2-hour period, their measured subjective workload increases to a degree associated with missing important tasks, threatening the quality and safety of the care they deliver.12,17 Given that nurses care for multiple patients, it is likely that the excess alarms introduced by guideline-discordant continuous monitoring contribute to increased nurse workload and alarm fatigue.
Similarly, we could not evaluate whether the alarms nurses experienced were actionable. Although our lab has previously reported that ≥99% of alarms occurring on non-ICU pediatric wards are nonactionable,10,11 it is possible that some of the alarms during guideline-discordant monitoring periods required action. However, it is unlikely that any life-sustaining actions were taken because (1) we only evaluated time >60 minutes after final discontinuation of supplemental oxygen, so by definition none of these alarms required treatment with supplemental oxygen, and (2) none of the patients we included received ICU care during their admission.
The avoidable alarm burden identified in our analysis suggests that eliminating continuous SpO2 monitoring overuse in bronchiolitis will likely reduce nurses’ workload and alarm fatigue in hospital settings that care for children with bronchiolitis.
Practice guidelines discourage continuous pulse oximetry (SpO2) monitoring of patients with bronchiolitis who are not receiving supplemental oxygen.1,2 Overuse of SpO2 monitoring in this patient population has been associated with increased length of stay, unnecessary oxygen therapy, and excess hospital costs, without measurable patient benefit.3-5 In spite of this evidence base and expert guidance, nearly half of the more than 100,000 infants admitted for bronchiolitis each year receive excess SpO2 monitoring.6,7
Bronchiolitis guidelines suggest that guideline-discordant SpO2 monitoring may result in excess alarms, which disrupt families’ sleep and engender alarm fatigue among staff.1 Pediatric nurses receive up to 155 alarms per monitored patient per day.8,9 Frequent alarms are associated with slower nurse response times10,11 and increased nurse subjective workload.12
Methods
Cohort
We retrospectively evaluated SpO2 monitoring patterns and alarm rates of children 0 to 24 months old admitted to a general pediatrics service at a tertiary care children’s hospital. We included patients who had a discharge diagnosis of bronchiolitis (International Classification of Diseases, Tenth Revision codes J45x, T17.2x, T17.3x, T17.4x, T17.5x, T17.8x, T17.9x, A37xx, J04x, J05x, J05.1x, J69.0x, J69.1x, J69.8x) between November 24, 2019, and January 21, 2020, the period of time during which alarm data and monitor data were concurrently available for analysis. In order to conservatively assure applicability of clinical practice guidelines, we excluded patients with discharge diagnoses that included other respiratory conditions (eg, reactive airway disease), patients with complex chronic conditions (CCC) as defined by the CCC version 2 classification system,13 and patients with intensive care unit (ICU) stays during the admission.
Time
Flowsheet data detailing nursing respiratory assessments were extracted from the electronic health record (EHR) database (Clarity, Epic Systems). Using previously validated methodology,14 we identified minutes during which patients received supplemental oxygen or high-flow nasal cannula (supplemental oxygen) based on the documented fraction of inspired oxygen (FiO2), flow rate, and support devices. We then identified the final discontinuation of respiratory support during the hospital admission, and censored the 60 minutes after final discontinuation of supplemental oxygen based upon recent monitoring guidelines.2 Minutes up to an hour after supplemental oxygen discontinuation were classified as receiving supplemental oxygen and not included in our analysis. Only minutes between the end of the censored period and hospital discharge were used in the analysis. For patients who never received respiratory support during the admission, we censored the first 60 minutes of the admission and analyzed the remainder of their stay.
SpO2 Monitoring
We used device-integrated, physiologic-monitor, vital sign data sent each minute from the General Electric monitor network to the EHR to identify minutes during which patients were connected to physiologic monitors and transmitting signals from SpO2 sensors. We extracted minute-level SpO2 data from the hospital clinical data warehouse (CDW). Minutes in which SpO2 data were present were classified as “monitored,” an approach previously validated using in-person observation.14
To categorize time as “not receiving supplemental oxygen and continuously monitored (guideline-discordant monitoring),” or “not receiving supplemental oxygen and not continuously monitored (guideline-concordant intermittent measurement),” we evaluated the percent of minutes within an hour during which the patient received SpO2 monitoring and applied an a priori conservative rule. Hours during which ≥90% of minutes had SpO2 monitoring data were classified as “continuously monitored.” Hours during which ≤10% of minutes had SpO2 monitoring data were classified as “intermittently measured.” Hours during which 11% to 89% of minutes included monitor data were excluded from further analysis. The number of continuously monitored hours was tabulated for each patient. The median number of continuously monitored hours was computed; results were stratified by prior receipt of respiratory support.
Alarm Counts
Minute-level monitor alarm counts (the absolute number of abnormal vital signs that triggered a monitor to alarm) were extracted from the CDW. Alarm counts were tabulated for each patient hour. For each patient, the alarm rate (total number of alarms divided by time) was computed for continuously monitored and intermittently measured time. Results were stratified by prior receipt of respiratory support.
The study was reviewed by the institutional review board and determined to meet exemption criteria.
Results
Our cohort included 201 admissions by 197 unique patients (Table). We evaluated 4402 hours that occurred ≥60 minutes following final discontinuation of supplemental oxygen, the time period during which guidelines discourage routine use of continuous SpO2 monitoring. This represented a median of 19 hours (interquartile range [IQR], 14-25) per admission. We excluded 474 hours (11%) that could not be classified as either continuously or intermittently measured.

During time ≥60 minutes following discontinuation of supplemental oxygen, our cohort experienced 1537 hours of guideline-discordant continuous monitoring, a median of 6 hours (IQR, 3-12) per admission. Patients experienced a median of 12 hours (IQR, 5-17) of intermittent measurement. Among patients who received supplemental oxygen, 91% experienced guideline-discordant continuous SpO2 monitoring, as compared to 68% of patients who did not receive supplemental oxygen. Among those who received guideline-discordant continuous SpO2 monitoring, the duration of this monitoring did not differ significantly between those who had received supplemental oxygen during the admission and those who had not.
During classifiable time ≥60 minutes following discontinuation of supplemental oxygen, our cohort experienced 14,371 alarms; 77% (11,101) of these alarms were generated during periods of guideline-discordant continuous monitoring. The median hourly alarm rate during these periods of guideline-discordant continuous monitoring was 6.7 alarms per hour (IQR, 2.1-12.3), representing a median of 35 (IQR, 10-81) additional alarms per patient. During periods of guideline-concordant intermittent measurement, the median hourly alarm rate was 0.5 (IQR, 0.1-0.8), with a median of 5 (IQR, 1-13) alarms per patient.
Those who received supplemental oxygen earlier in the admission had higher alarm rates during continuously monitored time (7.3 per hour [IQR, 2.7-12.7]) than patients who had not received supplemental oxygen (3.3 per hour [IQR, 0.6-11.8]), likely reflecting clinical differences between these patient populations. The most frequent alarm type among continuously monitored patients who had previously received supplemental oxygen was “SpO2 low.”
Discussion
Across 4402 patient hours, guideline-discordant continuous SpO2 monitoring of patients with bronchiolitis resulted in 11,101 alarms, at a rate of approximately 1 additional alarm every 9 minutes. Patients in our cohort received a median of 6 hours of guideline-discordant monitoring, which imposes a significant alarm burden that is potentially modifiable using targeted reduction strategies.15
Transient, self-resolved hypoxemia is a common feature of bronchiolitis and likely of little clinical consequence.16 Therefore, this rate of hypoxemia alarms is not unexpected. Though we evaluated only the period of time following final discontinuation of respiratory support, this finding is in keeping with the literature associating excess physiologic monitoring of patients with bronchiolitis with unnecessary oxygen therapy and increased length of stay,3-5 largely because clinicians may feel compelled to respond to hypoxemia alarms with either supplemental oxygen or longer monitoring.
Our findings must be contextualized in light of the limitations of our approach. We did not evaluate nurse workload associated with guideline-discordant continuous SpO2 monitoring. Prior work conducted by our lab has demonstrated that when nurses experience more than 40 alarms within a 2-hour period, their measured subjective workload increases to a degree associated with missing important tasks, threatening the quality and safety of the care they deliver.12,17 Given that nurses care for multiple patients, it is likely that the excess alarms introduced by guideline-discordant continuous monitoring contribute to increased nurse workload and alarm fatigue.
Similarly, we could not evaluate whether the alarms nurses experienced were actionable. Although our lab has previously reported that ≥99% of alarms occurring on non-ICU pediatric wards are nonactionable,10,11 it is possible that some of the alarms during guideline-discordant monitoring periods required action. However, it is unlikely that any life-sustaining actions were taken because (1) we only evaluated time >60 minutes after final discontinuation of supplemental oxygen, so by definition none of these alarms required treatment with supplemental oxygen, and (2) none of the patients we included received ICU care during their admission.
The avoidable alarm burden identified in our analysis suggests that eliminating continuous SpO2 monitoring overuse in bronchiolitis will likely reduce nurses’ workload and alarm fatigue in hospital settings that care for children with bronchiolitis.
1. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
2. Schondelmeyer AC, Dewan ML, Brady PW, et al. Cardiorespiratory and pulse oximetry monitoring in hospitalized children: a Delphi process. Pediatrics. 2020;146(2):e20193336. https://doi.org/10.1542/peds.2019-3336
3. Cunningham S, Rodriguez A, Boyd KA, McIntosh E, Lewis SC, BIDS Collaborators Group. Bronchiolitis of Infancy Discharge Study (BIDS): A multicentre, parallel-group, double-blind, randomised controlled, equivalence trial with economic evaluation. Health Technol Assess. 2015;19(71):i-xxiii, 1-172. https://doi.org/10.3310/hta19710
4. McCulloh R, Koster M, Ralston S, et al. Use of intermittent vs continuous pulse oximetry for nonhypoxemic infants and young children hospitalized for bronchiolitis: a randomized clinical trial. JAMA Pediatr. 2015;169(10):898-904. https://doi.org/10.1001/jamapediatrics.2015.1746
5. Schuh S, Freedman S, Coates A, et al. Effect of oximetry on hospitalization in bronchiolitis: a randomized clinical trial. JAMA. 2014;312(7):712-718. https://doi.org/10.1001/jama.2014.8637
6. Fujiogi M, Goto T, Yasunaga H, et al. Trends in bronchiolitis hospitalizations in the United States: 2000–2016. Pediatrics. 2019;144(6):e20192614. https://doi.org/10.1542/peds.2019-2614
7. Bonafide CP, Xiao R, Brady PW, et al. Prevalence of continuous pulse oximetry monitoring in hospitalized children with bronchiolitis not requiring supplemental oxygen. JAMA. 2020;323(15):1467-1477. https://doi.org/10.1001/jama.2020.2998
8. Schondelmeyer AC, Brady PW, Goel VV, et al. Physiologic monitor alarm rates at 5 children’s hospitals. J Hosp Med. 2018;13(6):396-398. https://doi.org/10.12788/jhm.2918
9. Schondelmeyer AC, Bonafide CP, Goel VV, et al. The frequency of physiologic monitor alarms in a children’s hospital. J Hosp Med. 2016;11(11):796-798. https://doi.org/10.1002/jhm.2612
10. Bonafide CP, Lin R, Zander M, et al. Association between exposure to nonactionable physiologic monitor alarms and response time in a children’s hospital. J Hosp Med. 2015;10(6):345-351. https://doi.org/10.1002/jhm.2331
11. Bonafide CP, Localio AR, Holmes JH, et al. Video analysis of factors associated with response time to physiologic monitor alarms in a children’s hospital. JAMA Pediatr. 2017;171(6):524-531. https://doi.org/10.1001/jamapediatrics.2016.5123
12. Rasooly IR, Kern-Goldberger AS, Xiao R, et al. Physiologic monitor alarm burden and nurses’ subjective workload in a children’s hospital. Hosp Pediatr. 2021;11(7):703-710. https://doi.org/10.1542/hpeds.2020-003509
13. Feudtner C, Feinstein JA, Zhong W, Hall M, Dai D. Pediatric complex chronic conditions classification system version 2: updated for ICD-10 and complex medical technology dependence and transplantation. BMC Pediatr. 2014;14:199. https://doi.org/10.1186/1471-2431-14-199
14. Kern-Goldberger AS, Rasooly IR, Luo B, et al. EHR-integrated monitor data to measure pulse oximetry use in bronchiolitis. Hosp Pediatr. 2021;11(10):1073-1082. https://doi.org/10.1542/hpeds.2021-005894
15. Schondelmeyer AC, Bettencourt AP, Xiao R, et al. Evaluation of an educational outreach and audit and feedback program to reduce continuous pulse oximetry use in hospitalized infants with stable bronchiolitis. JAMA Netw Open. 2021;4(9):e2122826. https://doi.org/10.1001/jamanetworkopen.2021.22826
16. Principi T, Coates AL, Parkin PC, Stephens D, DaSilva Z, Schuh S. Effect of oxygen desaturations on subsequent medical visits in infants discharged from the emergency department with bronchiolitis. JAMA Pediatr. 2016;170(6):602-608. https://doi.org/10.1001/jamapediatrics.2016.0114
17. Tubbs-Cooley HL, Mara CA, Carle AC, Mark BA, Pickler RH. Association of nurse workload with missed nursing care in the neonatal intensive care unit. JAMA Pediatr. 2019;173(1):44-51. https://doi.org/10.1001/jamapediatrics.2018.3619
1. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
2. Schondelmeyer AC, Dewan ML, Brady PW, et al. Cardiorespiratory and pulse oximetry monitoring in hospitalized children: a Delphi process. Pediatrics. 2020;146(2):e20193336. https://doi.org/10.1542/peds.2019-3336
3. Cunningham S, Rodriguez A, Boyd KA, McIntosh E, Lewis SC, BIDS Collaborators Group. Bronchiolitis of Infancy Discharge Study (BIDS): A multicentre, parallel-group, double-blind, randomised controlled, equivalence trial with economic evaluation. Health Technol Assess. 2015;19(71):i-xxiii, 1-172. https://doi.org/10.3310/hta19710
4. McCulloh R, Koster M, Ralston S, et al. Use of intermittent vs continuous pulse oximetry for nonhypoxemic infants and young children hospitalized for bronchiolitis: a randomized clinical trial. JAMA Pediatr. 2015;169(10):898-904. https://doi.org/10.1001/jamapediatrics.2015.1746
5. Schuh S, Freedman S, Coates A, et al. Effect of oximetry on hospitalization in bronchiolitis: a randomized clinical trial. JAMA. 2014;312(7):712-718. https://doi.org/10.1001/jama.2014.8637
6. Fujiogi M, Goto T, Yasunaga H, et al. Trends in bronchiolitis hospitalizations in the United States: 2000–2016. Pediatrics. 2019;144(6):e20192614. https://doi.org/10.1542/peds.2019-2614
7. Bonafide CP, Xiao R, Brady PW, et al. Prevalence of continuous pulse oximetry monitoring in hospitalized children with bronchiolitis not requiring supplemental oxygen. JAMA. 2020;323(15):1467-1477. https://doi.org/10.1001/jama.2020.2998
8. Schondelmeyer AC, Brady PW, Goel VV, et al. Physiologic monitor alarm rates at 5 children’s hospitals. J Hosp Med. 2018;13(6):396-398. https://doi.org/10.12788/jhm.2918
9. Schondelmeyer AC, Bonafide CP, Goel VV, et al. The frequency of physiologic monitor alarms in a children’s hospital. J Hosp Med. 2016;11(11):796-798. https://doi.org/10.1002/jhm.2612
10. Bonafide CP, Lin R, Zander M, et al. Association between exposure to nonactionable physiologic monitor alarms and response time in a children’s hospital. J Hosp Med. 2015;10(6):345-351. https://doi.org/10.1002/jhm.2331
11. Bonafide CP, Localio AR, Holmes JH, et al. Video analysis of factors associated with response time to physiologic monitor alarms in a children’s hospital. JAMA Pediatr. 2017;171(6):524-531. https://doi.org/10.1001/jamapediatrics.2016.5123
12. Rasooly IR, Kern-Goldberger AS, Xiao R, et al. Physiologic monitor alarm burden and nurses’ subjective workload in a children’s hospital. Hosp Pediatr. 2021;11(7):703-710. https://doi.org/10.1542/hpeds.2020-003509
13. Feudtner C, Feinstein JA, Zhong W, Hall M, Dai D. Pediatric complex chronic conditions classification system version 2: updated for ICD-10 and complex medical technology dependence and transplantation. BMC Pediatr. 2014;14:199. https://doi.org/10.1186/1471-2431-14-199
14. Kern-Goldberger AS, Rasooly IR, Luo B, et al. EHR-integrated monitor data to measure pulse oximetry use in bronchiolitis. Hosp Pediatr. 2021;11(10):1073-1082. https://doi.org/10.1542/hpeds.2021-005894
15. Schondelmeyer AC, Bettencourt AP, Xiao R, et al. Evaluation of an educational outreach and audit and feedback program to reduce continuous pulse oximetry use in hospitalized infants with stable bronchiolitis. JAMA Netw Open. 2021;4(9):e2122826. https://doi.org/10.1001/jamanetworkopen.2021.22826
16. Principi T, Coates AL, Parkin PC, Stephens D, DaSilva Z, Schuh S. Effect of oxygen desaturations on subsequent medical visits in infants discharged from the emergency department with bronchiolitis. JAMA Pediatr. 2016;170(6):602-608. https://doi.org/10.1001/jamapediatrics.2016.0114
17. Tubbs-Cooley HL, Mara CA, Carle AC, Mark BA, Pickler RH. Association of nurse workload with missed nursing care in the neonatal intensive care unit. JAMA Pediatr. 2019;173(1):44-51. https://doi.org/10.1001/jamapediatrics.2018.3619
© 2021 Society of Hospital Medicine
Preserving Margins to Promote Missions: COVID-19’s Toll on US Children’s Hospitals
Since the onset of the COVID-19 pandemic, the proclivity of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) for adults and its relative sparing of pediatric populations has been well characterized. Accordingly, policymakers have devoted significant attention to SARS-CoV-2’s impact on adult hospitals. Less consideration, however, has been given to children’s hospitals, which responded to the pandemic by suspending noncritical care encounters, conserving personal protective equipment, and implementing alternative care models.1 While important, these strategic decisions may threaten the financial health of children’s hospitals.
In this issue of the Journal of Hospital Medicine, Synhorst et al1 describe the impact of COVID-19 on US children’s hospitals.The authors utilized the Children’s Hospital Association’s PROSPECT database to compare year-over-year trends in healthcare encounters and hospital charges before and during the COVID-19 pandemic at 26 tertiary hospitals. The analysis focused on the first wave of COVID-19 in the United States from February through June 2020.
The results are staggering. Compared with 2019, the authors found significant decreases in healthcare encounters for all children’s hospitals beginning in March 2020, with a nadir in mid-April (corresponding to the first peak in adult hospitalizations). Inpatient bed days, emergency department (ED) visits, and surgeries decreased by a median of 36%, 65%, and 77%, respectively, per hospital during the nadir. Charges from February 1 to June 30, 2020, decreased by a median 24% per children’s hospital as compared to 2019—corresponding to a median $276 million decrease in charges per hospital. A quarter of hospitals faced more than $400 million in lost charges.1
Why do these trends matter? Large decreases in utilization and associated charges likely represent significant unmet demand for child healthcare for both acute and chronic disease management. For example, with limited in-person evaluation available at the onset of illness, caregivers are presenting to EDs with sicker children.2 With a shift to virtual care, clinicians may miss signs of child abuse from violence in the home—which can escalate during isolation.3 Children with chronic conditions may also be left without surveillance mechanisms, which may partly explain the autumn 2020 surge in acute mental health-related ED presentations.4 Furthermore, telemedicine may exacerbate care inequities for vulnerable populations lacking resources and/or English proficiency.
There is also a larger policy perspective to consider in evaluating these data: Because children’s hospitals largely operate in a fee-for-service reimbursement model, they often rely on marginal revenues to support mission-driven programming. In other words, revenue streams from profitable care segments (eg, elective surgeries) often help sustain institutional platforms operating at a loss, such as community safety net programs. Consequently, threats to marginal revenues can place mission-driven programming in jeopardy of being reduced or terminated.
The Synhorst et al1 study was limited to hospital charges, which likely overestimate revenue losses based on actual reimbursements. Yet, this is the first study to quantify COVID-19’s financial toll on children’s hospitals, and charges offer a reasonable proxy for balance sheet trends. Thus, it is safe to assume that most hospitals incurred substantial losses during the 2020 fiscal year. Unfortunately, as the authors highlight, these losses differentially impacted hospitals based on existing resources1—so some hospitals were likely forced to cut programs or reduce staff in an effort to return to profitability. In this way, COVID-19 has exposed the fragility of the fee-for-service model that children’s hospitals rely on for both patients and staff.
Children’s hospitals and the services they provide are essential to the health and well-being of children. The critical losses sustained by children’s hospitals due to COVID-19 threaten their ability to promote child health in the near and long term, with the greatest risk to vulnerable populations. Policymakers must act now to preserve these essential services for children.
1. Synhorst D, Hall M, Thurm C, et al. Healthcare encounter and financial impact of COVID-19 on children’s hospitals. J Hosp Med. 2021;16(4):223-226. https://doi.org/10.12788/jhm.3572
2. Chaiyachati BH, Agawu A, Zorc JJ, Balamuth F. Trends in pediatric emergency department utilization after institution of coronavirus disease-19 mandatory social distancing. J Pediatr. 2020;226:274-277.e1. https://doi.org/10.1016/j.jpeds.2020.07.048
3. Humphreys KL, Myint MT, Zeanah CH. Increased risk for family violence during the COVID-19 pandemic. Pediatrics. 2020;146(1):e20200982. https://doi.org/10.1542/peds.2020-0982
4. Leeb RT, Bitsko RH, Radhakrishnan L, Martinez P, Njai R, Holland KM. Mental health-related emergency department visits among children aged <18 years during the COVID-19 pandemic—United States, January 1–October 17, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1675-1680. https://doi.org/10.15585/mmwr.mm6945a3
Since the onset of the COVID-19 pandemic, the proclivity of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) for adults and its relative sparing of pediatric populations has been well characterized. Accordingly, policymakers have devoted significant attention to SARS-CoV-2’s impact on adult hospitals. Less consideration, however, has been given to children’s hospitals, which responded to the pandemic by suspending noncritical care encounters, conserving personal protective equipment, and implementing alternative care models.1 While important, these strategic decisions may threaten the financial health of children’s hospitals.
In this issue of the Journal of Hospital Medicine, Synhorst et al1 describe the impact of COVID-19 on US children’s hospitals.The authors utilized the Children’s Hospital Association’s PROSPECT database to compare year-over-year trends in healthcare encounters and hospital charges before and during the COVID-19 pandemic at 26 tertiary hospitals. The analysis focused on the first wave of COVID-19 in the United States from February through June 2020.
The results are staggering. Compared with 2019, the authors found significant decreases in healthcare encounters for all children’s hospitals beginning in March 2020, with a nadir in mid-April (corresponding to the first peak in adult hospitalizations). Inpatient bed days, emergency department (ED) visits, and surgeries decreased by a median of 36%, 65%, and 77%, respectively, per hospital during the nadir. Charges from February 1 to June 30, 2020, decreased by a median 24% per children’s hospital as compared to 2019—corresponding to a median $276 million decrease in charges per hospital. A quarter of hospitals faced more than $400 million in lost charges.1
Why do these trends matter? Large decreases in utilization and associated charges likely represent significant unmet demand for child healthcare for both acute and chronic disease management. For example, with limited in-person evaluation available at the onset of illness, caregivers are presenting to EDs with sicker children.2 With a shift to virtual care, clinicians may miss signs of child abuse from violence in the home—which can escalate during isolation.3 Children with chronic conditions may also be left without surveillance mechanisms, which may partly explain the autumn 2020 surge in acute mental health-related ED presentations.4 Furthermore, telemedicine may exacerbate care inequities for vulnerable populations lacking resources and/or English proficiency.
There is also a larger policy perspective to consider in evaluating these data: Because children’s hospitals largely operate in a fee-for-service reimbursement model, they often rely on marginal revenues to support mission-driven programming. In other words, revenue streams from profitable care segments (eg, elective surgeries) often help sustain institutional platforms operating at a loss, such as community safety net programs. Consequently, threats to marginal revenues can place mission-driven programming in jeopardy of being reduced or terminated.
The Synhorst et al1 study was limited to hospital charges, which likely overestimate revenue losses based on actual reimbursements. Yet, this is the first study to quantify COVID-19’s financial toll on children’s hospitals, and charges offer a reasonable proxy for balance sheet trends. Thus, it is safe to assume that most hospitals incurred substantial losses during the 2020 fiscal year. Unfortunately, as the authors highlight, these losses differentially impacted hospitals based on existing resources1—so some hospitals were likely forced to cut programs or reduce staff in an effort to return to profitability. In this way, COVID-19 has exposed the fragility of the fee-for-service model that children’s hospitals rely on for both patients and staff.
Children’s hospitals and the services they provide are essential to the health and well-being of children. The critical losses sustained by children’s hospitals due to COVID-19 threaten their ability to promote child health in the near and long term, with the greatest risk to vulnerable populations. Policymakers must act now to preserve these essential services for children.
Since the onset of the COVID-19 pandemic, the proclivity of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) for adults and its relative sparing of pediatric populations has been well characterized. Accordingly, policymakers have devoted significant attention to SARS-CoV-2’s impact on adult hospitals. Less consideration, however, has been given to children’s hospitals, which responded to the pandemic by suspending noncritical care encounters, conserving personal protective equipment, and implementing alternative care models.1 While important, these strategic decisions may threaten the financial health of children’s hospitals.
In this issue of the Journal of Hospital Medicine, Synhorst et al1 describe the impact of COVID-19 on US children’s hospitals.The authors utilized the Children’s Hospital Association’s PROSPECT database to compare year-over-year trends in healthcare encounters and hospital charges before and during the COVID-19 pandemic at 26 tertiary hospitals. The analysis focused on the first wave of COVID-19 in the United States from February through June 2020.
The results are staggering. Compared with 2019, the authors found significant decreases in healthcare encounters for all children’s hospitals beginning in March 2020, with a nadir in mid-April (corresponding to the first peak in adult hospitalizations). Inpatient bed days, emergency department (ED) visits, and surgeries decreased by a median of 36%, 65%, and 77%, respectively, per hospital during the nadir. Charges from February 1 to June 30, 2020, decreased by a median 24% per children’s hospital as compared to 2019—corresponding to a median $276 million decrease in charges per hospital. A quarter of hospitals faced more than $400 million in lost charges.1
Why do these trends matter? Large decreases in utilization and associated charges likely represent significant unmet demand for child healthcare for both acute and chronic disease management. For example, with limited in-person evaluation available at the onset of illness, caregivers are presenting to EDs with sicker children.2 With a shift to virtual care, clinicians may miss signs of child abuse from violence in the home—which can escalate during isolation.3 Children with chronic conditions may also be left without surveillance mechanisms, which may partly explain the autumn 2020 surge in acute mental health-related ED presentations.4 Furthermore, telemedicine may exacerbate care inequities for vulnerable populations lacking resources and/or English proficiency.
There is also a larger policy perspective to consider in evaluating these data: Because children’s hospitals largely operate in a fee-for-service reimbursement model, they often rely on marginal revenues to support mission-driven programming. In other words, revenue streams from profitable care segments (eg, elective surgeries) often help sustain institutional platforms operating at a loss, such as community safety net programs. Consequently, threats to marginal revenues can place mission-driven programming in jeopardy of being reduced or terminated.
The Synhorst et al1 study was limited to hospital charges, which likely overestimate revenue losses based on actual reimbursements. Yet, this is the first study to quantify COVID-19’s financial toll on children’s hospitals, and charges offer a reasonable proxy for balance sheet trends. Thus, it is safe to assume that most hospitals incurred substantial losses during the 2020 fiscal year. Unfortunately, as the authors highlight, these losses differentially impacted hospitals based on existing resources1—so some hospitals were likely forced to cut programs or reduce staff in an effort to return to profitability. In this way, COVID-19 has exposed the fragility of the fee-for-service model that children’s hospitals rely on for both patients and staff.
Children’s hospitals and the services they provide are essential to the health and well-being of children. The critical losses sustained by children’s hospitals due to COVID-19 threaten their ability to promote child health in the near and long term, with the greatest risk to vulnerable populations. Policymakers must act now to preserve these essential services for children.
1. Synhorst D, Hall M, Thurm C, et al. Healthcare encounter and financial impact of COVID-19 on children’s hospitals. J Hosp Med. 2021;16(4):223-226. https://doi.org/10.12788/jhm.3572
2. Chaiyachati BH, Agawu A, Zorc JJ, Balamuth F. Trends in pediatric emergency department utilization after institution of coronavirus disease-19 mandatory social distancing. J Pediatr. 2020;226:274-277.e1. https://doi.org/10.1016/j.jpeds.2020.07.048
3. Humphreys KL, Myint MT, Zeanah CH. Increased risk for family violence during the COVID-19 pandemic. Pediatrics. 2020;146(1):e20200982. https://doi.org/10.1542/peds.2020-0982
4. Leeb RT, Bitsko RH, Radhakrishnan L, Martinez P, Njai R, Holland KM. Mental health-related emergency department visits among children aged <18 years during the COVID-19 pandemic—United States, January 1–October 17, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1675-1680. https://doi.org/10.15585/mmwr.mm6945a3
1. Synhorst D, Hall M, Thurm C, et al. Healthcare encounter and financial impact of COVID-19 on children’s hospitals. J Hosp Med. 2021;16(4):223-226. https://doi.org/10.12788/jhm.3572
2. Chaiyachati BH, Agawu A, Zorc JJ, Balamuth F. Trends in pediatric emergency department utilization after institution of coronavirus disease-19 mandatory social distancing. J Pediatr. 2020;226:274-277.e1. https://doi.org/10.1016/j.jpeds.2020.07.048
3. Humphreys KL, Myint MT, Zeanah CH. Increased risk for family violence during the COVID-19 pandemic. Pediatrics. 2020;146(1):e20200982. https://doi.org/10.1542/peds.2020-0982
4. Leeb RT, Bitsko RH, Radhakrishnan L, Martinez P, Njai R, Holland KM. Mental health-related emergency department visits among children aged <18 years during the COVID-19 pandemic—United States, January 1–October 17, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1675-1680. https://doi.org/10.15585/mmwr.mm6945a3
© 2021 Society of Hospital Medicine
Barriers and Facilitators to Guideline-Adherent Pulse Oximetry Use in Bronchiolitis
Continuous pulse oximetry monitoring (cSpO2) in children with bronchiolitis is associated with increased rates of hospital admission, longer lengths of stay, more frequent treatment with supplemental oxygen, alarm fatigue, and higher hospital cost. There is no evidence that it improves clinical outcomes.1-7 The safety of reducing cSpO2 for stable bronchiolitis patients (ie, those who are clinically well and not requiring supplemental oxygen) has been assessed in quality improvement initiatives8-10 and a randomized controlled trial.2 These studies showed no increase in intensive care unit transfers, codes, or readmissions associated with reduced cSpO2. Current national guidelines from the American Academy of Pediatrics5 and the Society of Hospital Medicine Choosing Wisely in Pediatric Hospital Medicine workgroup4 support limiting monitoring of children with bronchiolitis. Despite this, the practice of cSpO2 in stable bronchiolitis patients off supplemental oxygen remains widespread.11,12
Deimplementation, defined as reducing or stopping low-value or ineffective healthcare practices,13,14 is a discrete focus area within implementation science. Deimplementation research involves the reduction of unnecessary and overused services for which there is potential for harm or no benefit.15,16 In pediatrics, there are a number of potential targets for deimplementation,4,17-20 including cSpO2 for stable infants with bronchiolitis, but efforts to reduce low-value practices have met limited success to date. 21,22
Implementation science offers rigorous methods for advancing the development and evaluation of strategies for deimplementation.23 In particular, implementation science frameworks can facilitate our understanding of relevant contextual factors that may hinder or help efforts to deimplement low-value practices. To develop broadly applicable strategies to reduce monitoring overuse, it is important to understand the barriers, facilitators, and contextual factors (eg, clinical, political, interpersonal) that contribute to guideline-discordant cSpO2 in hospitalized bronchiolitis patients. Further, the process by which one can develop a rigorous understanding of these factors and how they may impact deimplementation efforts could generalize to other scenarios in pediatrics where overuse remains an issue.
The goal of this study was to use semistructured interviews, informed by an established implementation science framework, specifically the Consolidated Framework for Implementation Research (CFIR),24 to (1) identify barriers and facilitators to deimplementing unnecessary cSpO2, and (2) develop strategies to deimplement cSpO2 in a multicenter cohort of hospital-based clinician and administrative stakeholders.
METHODS
Study Setting
This multicenter qualitative study using semistructured interviews took place within the Eliminating Monitor Overuse (EMO) SpO2 study. The EMO SpO2 study established rates of cSpO2 in bronchiolitis patients not receiving supplemental oxygen or not receiving room air flow at 56 hospitals across the United States and in Canada from December 1, 2018, through March 31, 2019.12 The study identified hospital-level risk-adjusted cSpO2 rates ranging from 6% to 82%. A description of the EMO SpO2 study methods25 and its findings12 have been published elsewhere.
Participants
We approached EMO study site principal investigators at 12 hospitals: the two highest- and two lowest-use hospitals within three hospital types (ie, freestanding children’s hospitals, children’s hospitals within large general hospitals, and community hospitals). We collaborated with the participating site principal investigators (n = 12), who were primarily hospitalist physicians in leadership roles, to recruit a purposive sample of additional stakeholders including bedside nurses (n = 12), hospitalist physicians (n = 15), respiratory therapists (n = 9), and hospital administrators (n = 8) to participate in semistructured interviews. Interviews were conducted until we achieved thematic saturation within each stakeholder group and within the high and low performing strata (total 56 interviews). Participants were asked to self-report basic demographic information (see Appendix, interview guide) as required by the study funder and to allow us to comment on the representativeness of the participant group.
Procedure
The interview guide was informed by the CFIR, a comprehensive framework detailing contextual factors that require consideration when planning for the implementation of a health service intervention. Table 1 details the CFIR domains with study-related examples. The interview guide (Appendix) provided limited clinical context apart from the age, diagnosis, and oxygen requirement for the population of interest to promote a broad array of responses and to avoid anchoring on specific clinical scenarios. Interviews were conducted by master’s degree or doctoral-level research coordinators with qualitative interviewing experience and supervised by a medical anthropologist and qualitative methods expert (F.K.B.). Prior to engaging in audio recorded phone interviews, the interviewer explained the risks and benefits of participating. Participants were compensated $50. Audio recordings were transcribed, deidentified, and uploaded to NVivo 12 Plus (QSR International) for data management.

The Institutional Review Boards of Children’s Hospital of Philadelphia, Pennsylvania, and the University of Pennsylvania in Philadelphia determined that the study met eligibility criteria for IRB exemption.
Data Analysis
Using an integrated approach to codebook development,26 a priori codes were developed using constructs from the CFIR. Additional codes were added by the research team following a close reading of the first five transcripts.27,28 Each code was defined, including decision rules for its application. Two research coordinators independently coded each transcript. Using the intercoder reliability function within NVivo, the coders established strong interrater reliability accordance scores (
RESULTS
Barriers and facilitators to deimplementation were identified in multiple domains of the CFIR: outer setting, inner setting, characteristics of the individuals, and intervention characteristics (Table 1). Participants also suggested strategies to facilitate deimplementation in response to some identified barriers. See Table 2 for participant demographics and Table 3 for illustrative participant quotations.

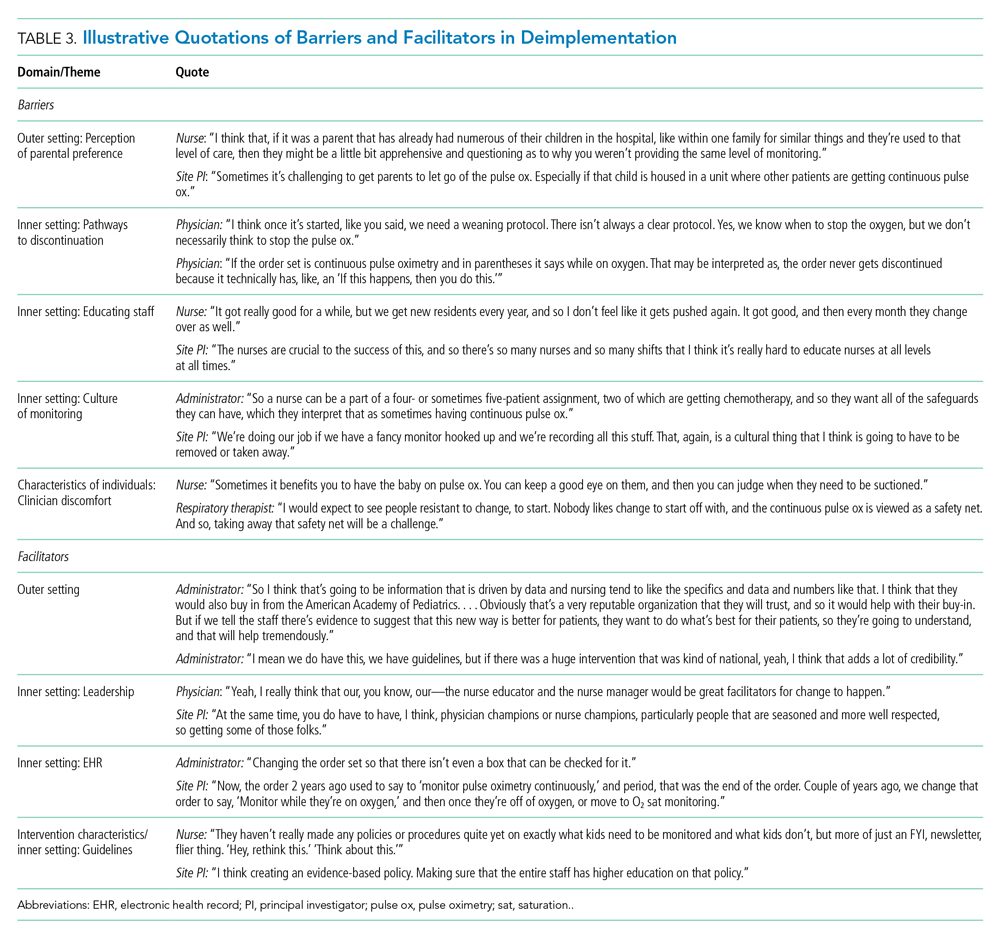
Barriers
Outer Setting: Clinician Perceptions of Parental Discomfort With Discontinuing Monitoring
Participants mentioned parental preferences as a barrier to discontinuing cSpO2, noting that parents seem to take comfort in watching the numbers on the monitor screen and are reluctant to have it withdrawn. Clinicians noted that parents sometimes put the monitor back on their child after a clinician removed it or have expressed concern that their unmonitored child was not receiving the same level of care as other patients who were being monitored. In these scenarios, clinicians reported they have found it helpful to educate caregivers about when cSpO2 is and is not appropriate.
Inner Setting: Unclear or Nonexistent Guideline to Discontinue cSpO2
Guidelines to discontinue cSpO2 reportedly did not exist at all institutions. If a guideline did exist, lack of clarity or conflicting guidelines about when to use oxygen presented a barrier. Participants suggested that a clear guideline or additional oversight to ensure all clinicians are informed of the procedure for discontinuing cSpO2 may help prevent miscommunication. Participants noted that their electronic health record (EHR) order sets commonly included cSpO2 orders and that removing that option would facilitate deimplementation.
Inner Setting: Difficulty Educating All Staff
Participants noted difficulty with incorporating education about discontinuing cSpO2 to all clinicians, particularly to those who are nightshift only or to rotating staff or trainees. This created barriers for frequent re-education because these staff are not familiar with the policies and procedures of the unit, which is crucial to developing a culture that supports the deimplementation of cSpO2. Participants suggested that recurring education about procedures for discontinuing cSpO2 should target trainees, new nurses, and overnight nurses. This would help to ensure that the guideline is uniformly followed.
Inner Setting: Culture of High cSpO2 Use
Participants from high-use sites discussed a culture driven by readily available monitoring features or an expectation that monitoring indicates higher-quality care. Participants from low-use sites discussed increased cSpO2 driven by clinicians who were accustomed to caring for higher-acuity patients, for whom continuous monitoring is likely appropriate, and were simultaneously caring for stable bronchiolitis patients.
Some suggested that visual cues would be useful to clinicians to sustain awareness about a cSpO2 deimplementation guideline. It was also suggested that audit and feedback techniques like posting unit deimplementation statistics and creating a competition among units by posting unit performance could facilitate deimplementation. Additionally, some noted that visual aids in common spaces would be useful to remind clinicians and to engage caregivers about discontinuing cSpO2.
Characteristics of Individuals: Clinician Discomfort Discontinuing cSpO2
One frequently cited barrier across participants is that cSpO2 provides “peace of mind” to alert clinicians to patients with low oxygen saturations that might otherwise be missed. Participants identified that clinician discomfort with reducing cSpO2 may be driven by inexperienced clinicians less familiar with the bronchiolitis disease process, such as trainees, new nurses, or rotating clinicians unaccustomed to pediatric care. Trainees and new nurses were perceived as being more likely to work at night when there are fewer clinicians to provide patient care. Additionally, participants perceived that night shift clinicians favored cSpO2 because they could measure vital signs without waking patients and families.
Clinicians discussed that discontinuing cSpO2 would require alternative methods for assessing patient status, particularly for night shift nurses. Participants suggested strategies including changes to pulse oximetry assessment procedures to include more frequent “spot checks,” incorporation of assessments during sleep events (eg, naps) to ensure the patient does not experience desaturations during sleep, and training nurses to become more comfortable with suctioning patients. Suggestions also included education on the typical features of transient oxygen desaturations in otherwise stable patients with bronchiolitis2 to bolster clinical confidence for clinicians unfamiliar with caring for bronchiolitis patients. Participants perceived that education about appropriate vs inappropriate use may help to empower clinicians to employ cSpO2 appropriately.
Facilitators
Outer Setting: Standards and Evidence From Research, Professional Organizations, and Leaders in the Field
Many participants expressed the importance of consistent guidelines that are advocated by thought leaders in the field, supported by robust evidence, and consistent with approaches at peer hospitals. The more authoritative support a guideline has, the more comfortable people are adopting it and taking it seriously. Additionally, consistent education about guidelines was desired. Participants noted that all clinicians should be receiving education related to the American Academy of Pediatrics (AAP) Bronchiolitis and Choosing Wisely® guidelines, ranging from a one-time update to annually. Continual updates and re-education sessions for clinicians who shared evidence about how cSpO2 deimplementation could improve the quality of patient care by shortening hospital length of stay and lowering cost were suggested strategies.
Inner Setting: Leadership
Participants noted that successful deimplementation depends upon the presence of a champion or educator who will be able to lead the institutional charge in making practice change. This is typically an individual who is trusted at the institution, experienced in their field, or already doing implementation work. This could be either a single individual (champion) or a team. The most commonly noted clinician roles to engage in a leadership role or team were physicians and nurses.
Participants noted that a change in related clinical care pathways or EHR order sets would require cooperation from multiple clinical disciplines, administrators, and information technology leaders and explained that messaging and education about the value of the change would facilitate buy-in from those clinicians.
Inner Setting: EHR Support for Guidelines
Participants often endorsed the use of an order set within the EHR that supports guidelines and includes reminders to decrease cSpO2. These reminders could come up when supplemental oxygen is discontinued or occur regularly throughout the patient’s stay to prompt the clinician to consider discontinuing cSpO2.
Intervention Characteristics/Inner Setting: Clear Bronchiolitis Guidelines
The presence of a well-articulated hospital policy that delineates the appropriate and inappropriate use of cSpO2 in bronchiolitis was mentioned as another facilitator of deimplementation.
DISCUSSION
Results of this qualitative study of stakeholders across hospitals with high and low cSpO2 use illustrated the complexities involved with deimplementation of cSpO2 in pediatric patients hospitalized with bronchiolitis. We identified numerous barriers spanning the CFIR constructs, including unclear or absent guidelines for stopping cSpO2, clinician knowledge and comfort with bronchiolitis disease features, and unit culture. This suggests that multicomponent strategies that target various domains and a variety of stakeholders are needed to deimplement cSpO2 use for stable bronchiolitis patients. Participants also identified facilitators, including clear cSpO2 guidelines, supportive leaders and champions, and EHR modifications, that provide insight into strategies that may help sites reduce their use of cSpO2. Additionally, participants also provided concrete, actionable suggestions for ways to reduce unnecessary monitoring that will be useful in informing promising deimplementation strategies for subsequent trials.
The importance of having specific and well-known guidelines from trusted sources, such as the AAP, about cSpO2 and bronchiolitis treatment that are thoughtfully integrated in the EHR came through in multiple themes of our analysis. Prior studies on the effect of guidelines on clinical practice have suggested that rigorously designed guidelines can positively impact practice.29 Participants also noted that cSpO2 guidelines should be authoritative and that knowledge of guideline adoption by peer institutions was a facilitator of adoption. Usability issues negatively impact clinicians’ ability to follow guidelines.30 Further, prior studies have demonstrated that EHR integration of guidelines can change practice.31-33 Based on our findings, incorporating clear guidelines into commonly used formats, such as EHR order sets, could be an important deimplementation tool for cSpO2 in stable bronchiolitis patients.
Education about and awareness of cSpO2 guidelines was described as an important facilitator for appropriate cSpO2 use and was suggested as a potential deimplementation strategy. Participants noted that educational need may vary by stakeholder group. For example, education may facilitate obtaining buy-in from hospital leaders, which is necessary to support changes to the EHR. Education incorporating information on the typical features of bronchiolitis and examples of appropriate and inappropriate cSpO2 use was suggested for clinical team members. The limitations of education as a stand-alone deimplementation strategy were also noted, and participants highlighted challenges such as time needed for education and the need for ongoing education for rotating trainees. Inner and outer setting barriers, such as a perceived “culture of high pulse oximetryuse” and patient and family expectations, could also make education less effective as a stand-alone strategy. That—coupled with evidence that education and training alone are generally insufficient for producing reliable, sustained behavior change34,35—suggests that a multifaceted approach will be important.
Our respondents consider parental perceptions and preference in their practice, which provides nuance to recent studies suggesting that parents prefer continuous monitors when their child is hospitalized with bronchiolitis. Chi et al described the impact of a brief educational intervention on parental preferences for monitoring children hospitalized for bronchiolitis.36 This work suggests that educational interventions aimed at families should be considered in future (de)implementation studies because they may indirectly impact clinician behavior. Future studies should directly assess parental discomfort with discontinuing monitoring. Participants highlighted the link between knowledge and confidence in caring for typical bronchiolitis patients and monitoring practice, perceiving that less experienced clinicians are more likely to rely on cSpO2. Participants at high-use sites emphasized the expectation that monitoring should occur during hospitalizations. This reflection is particularly pertinent for bronchiolitis, a disease characterized by frequent, self-resolving desaturations even after hospital discharge.3 This may reinforce a perceived need to capture and react to these desaturation events even though they are expected in bronchiolitis and can occur in healthy infants.37 Some participants suggested that continuous monitoring be replaced with “nap tests” (ie, assessment for desaturations during a nap prior to discharge); however, like cSpO2 in stable infants with bronchiolitis, this is another low-value practice. Otherwise healthy infants with mild to moderate disease are unlikely to subsequently worsen after showing signs of clinical improvement.38 Nap tests are likely to lead to infants who are clinically improving being placed unnecessarily back on oxygen in reaction to the transient desaturations. Participants’ perception about the importance of cSpO2 in bronchiolitis management, despite evidence suggesting it is a low-value practice, underscores the importance of not simply telling clinicians to stop cSpO2. Employing strategies that replace continuous monitoring with another acceptable and feasible alternative (eg, regular clinician assessments including intermittent pulse oximetry checks) should be considered when planning for deimplementation.39
Previous studies indicate that continuous monitoring can affect clinician decision-making, independent of other factors,6,40 despite limited evidence that continuous monitors improve patient outcomes.1-7 Studies have demonstrated noticeable increase in admissions based purely on pulse oximetry values,40 with no evidence that this type of admission changes outcomes for bronchiolitis patients.6 One previous, single-center study identified inexperience as a potential driver for monitor use,41 and studies in adult populations have suggested that clinicians overestimate the value that continuous monitoring contributes to patient care,42,43 which promotes guideline-discordant use. Our study provides novel insight into the issue of monitoring in bronchiolitis. Our results suggest that there is a need to shift organizational cultures around monitoring (which likely vary based on a range of factors) and that educational strategies addressing typical disease course, especially desaturations, in bronchiolitis will be an essential component in any deimplementation effort.
This study is strengthened by its sample of diverse stakeholder groups from multiple US health systems. Additionally, we interviewed individuals at sites with high cSpO2 rates and at sites with low rates, as well as from community hospitals, children’s hospitals within general hospitals, and freestanding children’s hospitals, which allows us to understand barriers high-use sites encounter and facilitators of lower cSpO2 rates at low-use sites. We also employed an interview approach informed by an established implementation science framework. Nonetheless, several limitations exist. First, participants at low-use sites did not necessarily have direct experience with a previous deimplementation effort to reduce cSpO2. Additionally, participants were predominantly White and female; more diverse perspectives would strengthen confidence in the generalizability of our findings. While thematic saturation was achieved within each stakeholder group and within the high- and low-use strata, we interviewed fewer administrators and respiratory therapists relative to other stakeholder groups. Nevertheless, our conclusions were validated by our interdisciplinary stakeholder panel. As noted by participants, family preferences may influence clinician practice, and parents were not interviewed for this study. The information gleaned from the present study will inform the development of strategies to deimplement unnecessary cSpO2 in pediatric hospitals, which we aim to rigorously evaluate in a future trial.
CONCLUSION
We identified barriers and facilitators to deimplementation of cSpO2 for stable patients with bronchiolitis across children’s hospitals with high and low utilization of cSpO2. These themes map to multiple CFIR domains and, along with participant-suggested strategies, can directly inform an approach to cSpO2 deimplementation in a range of inpatient settings. Based on these data, future deimplementation efforts should focus on clear protocols for use and discontinuation of cSpO2, EHR changes, and regular bronchiolitis education for hospital staff that emphasizes reducing unnecessary cSpO2 utilization.
ACKNOWLEDGMENTS
We acknowledge the NHLBI scientists who contributed their expertise to this project as part of the U01 Cooperative Agreement funding mechanism as federal employees conducting their official job duties: Lora Reineck, MD, MS, Karen Bienstock, MS, and Cheryl Boyce, PhD. We thank the Executive Council of the Pediatric Research in Inpatient Settings (PRIS) Network for their contributions to the early scientific development of this project. The Network assessed a Collaborative Support Fee for access to the hospitals and support of this project. We thank the PRIS Network collaborators for their major contributions to data collection measuring utilization to identify the hospitals we subsequently chose for this project. We thank Claire Bocage and the Mixed Methods Research Lab for major help in data management and data analysis.
1. Cunningham S, Rodriguez A, Adams T, et al; Bronchiolitis of Infancy Discharge Study (BIDS) group. Oxygen saturation targets in infants with bronchiolitis (BIDS): a double-blind, randomised, equivalence trial. Lancet. 2015;386(9998):1041-1048. https://doi.org/10.1016/s0140-6736(15)00163-4
2. McCulloh R, Koster M, Ralston S, et al. Use of intermittent vs continuous pulse oximetry for nonhypoxemic infants and young children hospitalized for bronchiolitis: a randomized clinical trial. JAMA Pediatr. 2015;169(10):898-904. https://doi.org/10.1001/jamapediatrics.2015.1746
3. Principi T, Coates AL, Parkin PC, Stephens D, DaSilva Z, Schuh S. Effect of oxygen desaturations on subsequent medical visits in infants discharged from the emergency department with bronchiolitis. JAMA Pediatr. 2016;170(6):602-608. https://doi.org/10.1001/jamapediatrics.2016.0114
4. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. https://doi.org/10.1002/jhm.2064
5. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
6. Schuh S, Freedman S, Coates A, et al. Effect of oximetry on hospitalization in bronchiolitis: a randomized clinical trial. JAMA. 2014;312(7):712-718. https://doi.org/10.1001/jama.2014.8637
7. Schuh S, Kwong JC, Holder L, Graves E, Macdonald EM, Finkelstein Y. Predictors of critical care and mortality in bronchiolitis after emergency department discharge. J Pediatr. 2018;199:217-222 e211. https://doi.org/10.1016/j.jpeds.2018.04.010
8. Schondelmeyer AC, Simmons JM, Statile AM, et al. Using quality improvement to reduce continuous pulse oximetry use in children with wheezing. Pediatrics. 2015;135(4):e1044-e1051. https://doi.org/10.1542/peds.2014-2295
9. Mittal S, Marlowe L, Blakeslee S, et al. Successful use of quality improvement methodology to reduce inpatient length of stay in bronchiolitis through judicious use of intermittent pulse oximetry. Hosp Pediatr. 2019;9(2):73-78. https://doi.org/10.1542/hpeds.2018-0023
10. Heneghan M, Hart J, Dewan M, et al. No Cause for Alarm: Decreasing inappropriate pulse oximetry use in bronchiolitis. Hosp Pediatr. 2018;8(2):109-111. https://doi.org/10.1542/hpeds.2017-0126
11. Ralston S, Garber M, Narang S, et al. Decreasing unnecessary utilization in acute bronchiolitis care: results from the value in inpatient pediatrics network. J Hosp Med. 2013;8(1):25-30. https://doi.org/10.1002/jhm.1982
12. Bonafide CP, Xiao R, Brady PW, et al. Prevalence of continuous pulse oximetry monitoring in hospitalized children with bronchiolitis not requiring supplemental oxygen. JAMA. 2020;323(15):1467-1477. https://doi.org/10.1001/jama.2020.2998
13. van Bodegom-Vos L, Davidoff F, Marang-van de Mheen PJ. Implementation and de-implementation: two sides of the same coin? BMJ Qual Saf. 2017;26(6):495-501. https://doi.org/10.1136/bmjqs-2016-005473
14. McKay VR, Morshed AB, Brownson RC, Proctor EK, Prusaczyk B. Letting go: conceptualizing intervention de-implementation in public health and social service settings. Am J Community Psychol. 2018;62(1-2):189-202. https://doi.org/10.1002/ajcp.12258
15. Brownlee S, Chalkidou K, Doust J, et al. Evidence for overuse of medical services around the world. Lancet. 2017;390(10090):156-168. https://doi.org/10.1016/s0140-6736(16)32585-5
16. Chassin MR, Galvin RW. The urgent need to improve health care quality. Institute of Medicine National roundtable on health care quality. JAMA. 1998;280(11):1000-1005. https://doi.org/10.1001/jama.280.11.1000
17. Coon ER, Young PC, Quinonez RA, Morgan DJ, Dhruva SS, Schroeder AR. 2017 update on pediatric medical overuse: a review. JAMA Pediatr. 2018;172(5):482-486. https://doi.org/10.1001/jamapediatrics.2017.5752
18. Schuh S, Babl FE, Dalziel SR, et al; Pediatric Emergency Research Networks (PERN). Practice variation in acute bronchiolitis: a Pediatric Emergency Research Networks study. Pediatrics. 2017;140(6):e20170842. https://doi.org/10.1542/peds.2017-0842
19. Lewis-de Los Angeles WW, Thurm C, Hersh AL, et al. Trends in intravenous antibiotic duration for urinary tract infections in young infants. Pediatrics. 2017;140(6):e20171021. https://doi.org/10.1542/peds.2017-1021
20. Parikh K, Hall M, Mittal V, et al. Establishing benchmarks for the hospitalized care of children with asthma, bronchiolitis, and pneumonia. Pediatrics. 2014;134(3):555-562. https://doi.org/10.1542/peds.2014-1052
21. Ralston SL, Garber MD, Rice-Conboy E, et al; Value in Inpatient Pediatrics Network Quality Collaborative for Improving Hospital Compliance with AAP Bronchiolitis Guideline (BQIP). A multicenter collaborative to reduce unnecessary care in inpatient bronchiolitis. Pediatrics. 2016;137(1):e20150851. https://doi.org/10.1542/peds.2015-0851
22. Reyes MA, Etinger V, Hall M, et al. Impact of the Choosing Wisely((R)) Campaign recommendations for hospitalized children on clinical practice: trends from 2008 to 2017. J Hosp Med. 2020;15(2):68-74. https://doi.org/10.12788/jhm.3291
23. Norton WE, Chambers DA. Unpacking the complexities of de-implementing inappropriate health interventions. Implement Sci. 2020;15(1):2. https://doi.org/10.1186/s13012-019-0960-9
24. Damschroder LJ, Aron DC, Keith RE, Kirsh SR, Alexander JA, Lowery JC. Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci. 2009;4:50. https://doi.org/10.1186/1748-5908-4-50
25. Rasooly IR, Beidas RS, Wolk CB, et al. Measuring overuse of continuous pulse oximetry in bronchiolitis and developing strategies for large-scale deimplementation: study protocol for a feasibility trial. Pilot Feasibility Stud. 2019;5:68. https://doi.org/10.1186/s40814-019-0453-2
26. Bradley EH, Curry LA, Devers KJ. Qualitative data analysis for health services research: developing taxonomy, themes, and theory. Health Serv Res. 2007;42(4):1758-1772. https://doi.org/10.1111/j.1475-6773.2006.00684.x
27. Glaser BG, Strauss AL. The Discovery of Grounded Theory: Strategies for Qualitative Research. Aldine Pub. Co.; 1967.
28. Charmaz K. Grounded Theory: Objectivist and Constructivist Methods. In: Denzin NK, Lincoln Y, eds. Handbook of Qualitative Research. 2nd ed. Sage Publications; 2000:509-535.
29. Grimshaw JM, Russell IT. Effect of clinical guidelines on medical practice: a systematic review of rigorous evaluations. Lancet. 1993;342(8883):1317-1322. https://doi.org/10.1016/0140-6736(93)92244-n
30. Cabana MD, Rand CS, Powe NR, et al. Why don’t physicians follow clinical practice guidelines? a framework for improvement. JAMA. 1999;282(15):1458-1465. https://doi.org/10.1001/jama.282.15.1458
31. Dressler R, Dryer MM, Coletti C, Mahoney D, Doorey AJ. Altering overuse of cardiac telemetry in non-intensive care unit settings by hardwiring the use of American Heart Association guidelines. JAMA Intern Med. 2014;174(11):1852-1854. https://doi.org/10.1001/jamainternmed.2014.4491
32. Forrest CB, Fiks AG, Bailey LC, et al. Improving adherence to otitis media guidelines with clinical decision support and physician feedback. Pediatrics. 2013;131(4):e1071-e1081. https://doi.org/10.1542/peds.2012-1988
33. Fiks AG, Grundmeier RW, Mayne S, et al. Effectiveness of decision support for families, clinicians, or both on HPV vaccine receipt. Pediatrics. 2013;131(6):1114-1124. https://doi.org/10.1542/peds.2012-3122
34. Nolan T, Resar R, Griffin F, Gordon AB. Improving the Reliability of Health Care. Institute for Healthcare Improvement; 2004. http://www.ihi.org/resources/Pages/IHIWhitePapers/ImprovingtheReliabilityofHealthCare.aspx
35. Beidas RS, Kendall PC. Training Therapists in evidence-based practice: a critical review of studies from a systems-contextual perspective. Clin Psychol (New York). 2010;17(1):1-30. https://doi.org/10.1111/j.1468-2850.2009.01187.x
36. Chi KW, Coon ER, Destino L, Schroeder AR. Parental perspectives on continuous pulse oximetry use in bronchiolitis hospitalizations. Pediatrics. 2020;146(2):e20200130. https://doi.org/10.1542/peds.2020-0130
37. Hunt CE, Corwin MJ, Lister G, et al. Longitudinal assessment of hemoglobin oxygen saturation in healthy infants during the first 6 months of age. Collaborative Home Infant Monitoring Evaluation (CHIME) Study Group. J Pediatr. 1999;135(5):580-586. https://doi.org/10.1016/s0022-3476(99)70056-9
38. Mansbach JM, Clark S, Piedra PA, et al; MARC-30 Investigators. Hospital course and discharge criteria for children hospitalized with bronchiolitis. J Hosp Med. 2015;10(4):205-211. https://doi.org/10.1002/jhm.2318
39. Burton C, Williams L, Bucknall T, et al. Understanding how and why de-implementation works in health and care: research protocol for a realist synthesis of evidence. Syst Rev. 2019;8(1):194. https://doi.org/10.1186/s13643-019-1111-840. Mallory MD, Shay DK, Garrett J, Bordley WC. Bronchiolitis management preferences and the influence of pulse oximetry and respiratory rate on the decision to admit. Pediatrics. 2003;111(1):e45-51. https://doi.org/10.1542/peds.111.1.e45.
41. Schondelmeyer AC, Jenkins AM, Allison B, et al. Factors influencing use of continuous physiologic monitors for hospitalized pediatric patients. Hosp Pediatr. 2019;9(6):423-428. https://doi.org/10.1542/hpeds.2019-0007
42. Najafi N, Auerbach A. Use and outcomes of telemetry monitoring on a medicine service. Arch Intern Med. 2012;172(17):1349-1350. https://doi.org/10.1001/archinternmed.2012.3163
43. Estrada CA, Rosman HS, Prasad NK, et al. Role of telemetry monitoring in the non-intensive care unit. Am J Cardiol. 1995;76(12):960-965. https://doi.org/10.1016/s0002-9149(99)80270-7
Continuous pulse oximetry monitoring (cSpO2) in children with bronchiolitis is associated with increased rates of hospital admission, longer lengths of stay, more frequent treatment with supplemental oxygen, alarm fatigue, and higher hospital cost. There is no evidence that it improves clinical outcomes.1-7 The safety of reducing cSpO2 for stable bronchiolitis patients (ie, those who are clinically well and not requiring supplemental oxygen) has been assessed in quality improvement initiatives8-10 and a randomized controlled trial.2 These studies showed no increase in intensive care unit transfers, codes, or readmissions associated with reduced cSpO2. Current national guidelines from the American Academy of Pediatrics5 and the Society of Hospital Medicine Choosing Wisely in Pediatric Hospital Medicine workgroup4 support limiting monitoring of children with bronchiolitis. Despite this, the practice of cSpO2 in stable bronchiolitis patients off supplemental oxygen remains widespread.11,12
Deimplementation, defined as reducing or stopping low-value or ineffective healthcare practices,13,14 is a discrete focus area within implementation science. Deimplementation research involves the reduction of unnecessary and overused services for which there is potential for harm or no benefit.15,16 In pediatrics, there are a number of potential targets for deimplementation,4,17-20 including cSpO2 for stable infants with bronchiolitis, but efforts to reduce low-value practices have met limited success to date. 21,22
Implementation science offers rigorous methods for advancing the development and evaluation of strategies for deimplementation.23 In particular, implementation science frameworks can facilitate our understanding of relevant contextual factors that may hinder or help efforts to deimplement low-value practices. To develop broadly applicable strategies to reduce monitoring overuse, it is important to understand the barriers, facilitators, and contextual factors (eg, clinical, political, interpersonal) that contribute to guideline-discordant cSpO2 in hospitalized bronchiolitis patients. Further, the process by which one can develop a rigorous understanding of these factors and how they may impact deimplementation efforts could generalize to other scenarios in pediatrics where overuse remains an issue.
The goal of this study was to use semistructured interviews, informed by an established implementation science framework, specifically the Consolidated Framework for Implementation Research (CFIR),24 to (1) identify barriers and facilitators to deimplementing unnecessary cSpO2, and (2) develop strategies to deimplement cSpO2 in a multicenter cohort of hospital-based clinician and administrative stakeholders.
METHODS
Study Setting
This multicenter qualitative study using semistructured interviews took place within the Eliminating Monitor Overuse (EMO) SpO2 study. The EMO SpO2 study established rates of cSpO2 in bronchiolitis patients not receiving supplemental oxygen or not receiving room air flow at 56 hospitals across the United States and in Canada from December 1, 2018, through March 31, 2019.12 The study identified hospital-level risk-adjusted cSpO2 rates ranging from 6% to 82%. A description of the EMO SpO2 study methods25 and its findings12 have been published elsewhere.
Participants
We approached EMO study site principal investigators at 12 hospitals: the two highest- and two lowest-use hospitals within three hospital types (ie, freestanding children’s hospitals, children’s hospitals within large general hospitals, and community hospitals). We collaborated with the participating site principal investigators (n = 12), who were primarily hospitalist physicians in leadership roles, to recruit a purposive sample of additional stakeholders including bedside nurses (n = 12), hospitalist physicians (n = 15), respiratory therapists (n = 9), and hospital administrators (n = 8) to participate in semistructured interviews. Interviews were conducted until we achieved thematic saturation within each stakeholder group and within the high and low performing strata (total 56 interviews). Participants were asked to self-report basic demographic information (see Appendix, interview guide) as required by the study funder and to allow us to comment on the representativeness of the participant group.
Procedure
The interview guide was informed by the CFIR, a comprehensive framework detailing contextual factors that require consideration when planning for the implementation of a health service intervention. Table 1 details the CFIR domains with study-related examples. The interview guide (Appendix) provided limited clinical context apart from the age, diagnosis, and oxygen requirement for the population of interest to promote a broad array of responses and to avoid anchoring on specific clinical scenarios. Interviews were conducted by master’s degree or doctoral-level research coordinators with qualitative interviewing experience and supervised by a medical anthropologist and qualitative methods expert (F.K.B.). Prior to engaging in audio recorded phone interviews, the interviewer explained the risks and benefits of participating. Participants were compensated $50. Audio recordings were transcribed, deidentified, and uploaded to NVivo 12 Plus (QSR International) for data management.

The Institutional Review Boards of Children’s Hospital of Philadelphia, Pennsylvania, and the University of Pennsylvania in Philadelphia determined that the study met eligibility criteria for IRB exemption.
Data Analysis
Using an integrated approach to codebook development,26 a priori codes were developed using constructs from the CFIR. Additional codes were added by the research team following a close reading of the first five transcripts.27,28 Each code was defined, including decision rules for its application. Two research coordinators independently coded each transcript. Using the intercoder reliability function within NVivo, the coders established strong interrater reliability accordance scores (
RESULTS
Barriers and facilitators to deimplementation were identified in multiple domains of the CFIR: outer setting, inner setting, characteristics of the individuals, and intervention characteristics (Table 1). Participants also suggested strategies to facilitate deimplementation in response to some identified barriers. See Table 2 for participant demographics and Table 3 for illustrative participant quotations.


Barriers
Outer Setting: Clinician Perceptions of Parental Discomfort With Discontinuing Monitoring
Participants mentioned parental preferences as a barrier to discontinuing cSpO2, noting that parents seem to take comfort in watching the numbers on the monitor screen and are reluctant to have it withdrawn. Clinicians noted that parents sometimes put the monitor back on their child after a clinician removed it or have expressed concern that their unmonitored child was not receiving the same level of care as other patients who were being monitored. In these scenarios, clinicians reported they have found it helpful to educate caregivers about when cSpO2 is and is not appropriate.
Inner Setting: Unclear or Nonexistent Guideline to Discontinue cSpO2
Guidelines to discontinue cSpO2 reportedly did not exist at all institutions. If a guideline did exist, lack of clarity or conflicting guidelines about when to use oxygen presented a barrier. Participants suggested that a clear guideline or additional oversight to ensure all clinicians are informed of the procedure for discontinuing cSpO2 may help prevent miscommunication. Participants noted that their electronic health record (EHR) order sets commonly included cSpO2 orders and that removing that option would facilitate deimplementation.
Inner Setting: Difficulty Educating All Staff
Participants noted difficulty with incorporating education about discontinuing cSpO2 to all clinicians, particularly to those who are nightshift only or to rotating staff or trainees. This created barriers for frequent re-education because these staff are not familiar with the policies and procedures of the unit, which is crucial to developing a culture that supports the deimplementation of cSpO2. Participants suggested that recurring education about procedures for discontinuing cSpO2 should target trainees, new nurses, and overnight nurses. This would help to ensure that the guideline is uniformly followed.
Inner Setting: Culture of High cSpO2 Use
Participants from high-use sites discussed a culture driven by readily available monitoring features or an expectation that monitoring indicates higher-quality care. Participants from low-use sites discussed increased cSpO2 driven by clinicians who were accustomed to caring for higher-acuity patients, for whom continuous monitoring is likely appropriate, and were simultaneously caring for stable bronchiolitis patients.
Some suggested that visual cues would be useful to clinicians to sustain awareness about a cSpO2 deimplementation guideline. It was also suggested that audit and feedback techniques like posting unit deimplementation statistics and creating a competition among units by posting unit performance could facilitate deimplementation. Additionally, some noted that visual aids in common spaces would be useful to remind clinicians and to engage caregivers about discontinuing cSpO2.
Characteristics of Individuals: Clinician Discomfort Discontinuing cSpO2
One frequently cited barrier across participants is that cSpO2 provides “peace of mind” to alert clinicians to patients with low oxygen saturations that might otherwise be missed. Participants identified that clinician discomfort with reducing cSpO2 may be driven by inexperienced clinicians less familiar with the bronchiolitis disease process, such as trainees, new nurses, or rotating clinicians unaccustomed to pediatric care. Trainees and new nurses were perceived as being more likely to work at night when there are fewer clinicians to provide patient care. Additionally, participants perceived that night shift clinicians favored cSpO2 because they could measure vital signs without waking patients and families.
Clinicians discussed that discontinuing cSpO2 would require alternative methods for assessing patient status, particularly for night shift nurses. Participants suggested strategies including changes to pulse oximetry assessment procedures to include more frequent “spot checks,” incorporation of assessments during sleep events (eg, naps) to ensure the patient does not experience desaturations during sleep, and training nurses to become more comfortable with suctioning patients. Suggestions also included education on the typical features of transient oxygen desaturations in otherwise stable patients with bronchiolitis2 to bolster clinical confidence for clinicians unfamiliar with caring for bronchiolitis patients. Participants perceived that education about appropriate vs inappropriate use may help to empower clinicians to employ cSpO2 appropriately.
Facilitators
Outer Setting: Standards and Evidence From Research, Professional Organizations, and Leaders in the Field
Many participants expressed the importance of consistent guidelines that are advocated by thought leaders in the field, supported by robust evidence, and consistent with approaches at peer hospitals. The more authoritative support a guideline has, the more comfortable people are adopting it and taking it seriously. Additionally, consistent education about guidelines was desired. Participants noted that all clinicians should be receiving education related to the American Academy of Pediatrics (AAP) Bronchiolitis and Choosing Wisely® guidelines, ranging from a one-time update to annually. Continual updates and re-education sessions for clinicians who shared evidence about how cSpO2 deimplementation could improve the quality of patient care by shortening hospital length of stay and lowering cost were suggested strategies.
Inner Setting: Leadership
Participants noted that successful deimplementation depends upon the presence of a champion or educator who will be able to lead the institutional charge in making practice change. This is typically an individual who is trusted at the institution, experienced in their field, or already doing implementation work. This could be either a single individual (champion) or a team. The most commonly noted clinician roles to engage in a leadership role or team were physicians and nurses.
Participants noted that a change in related clinical care pathways or EHR order sets would require cooperation from multiple clinical disciplines, administrators, and information technology leaders and explained that messaging and education about the value of the change would facilitate buy-in from those clinicians.
Inner Setting: EHR Support for Guidelines
Participants often endorsed the use of an order set within the EHR that supports guidelines and includes reminders to decrease cSpO2. These reminders could come up when supplemental oxygen is discontinued or occur regularly throughout the patient’s stay to prompt the clinician to consider discontinuing cSpO2.
Intervention Characteristics/Inner Setting: Clear Bronchiolitis Guidelines
The presence of a well-articulated hospital policy that delineates the appropriate and inappropriate use of cSpO2 in bronchiolitis was mentioned as another facilitator of deimplementation.
DISCUSSION
Results of this qualitative study of stakeholders across hospitals with high and low cSpO2 use illustrated the complexities involved with deimplementation of cSpO2 in pediatric patients hospitalized with bronchiolitis. We identified numerous barriers spanning the CFIR constructs, including unclear or absent guidelines for stopping cSpO2, clinician knowledge and comfort with bronchiolitis disease features, and unit culture. This suggests that multicomponent strategies that target various domains and a variety of stakeholders are needed to deimplement cSpO2 use for stable bronchiolitis patients. Participants also identified facilitators, including clear cSpO2 guidelines, supportive leaders and champions, and EHR modifications, that provide insight into strategies that may help sites reduce their use of cSpO2. Additionally, participants also provided concrete, actionable suggestions for ways to reduce unnecessary monitoring that will be useful in informing promising deimplementation strategies for subsequent trials.
The importance of having specific and well-known guidelines from trusted sources, such as the AAP, about cSpO2 and bronchiolitis treatment that are thoughtfully integrated in the EHR came through in multiple themes of our analysis. Prior studies on the effect of guidelines on clinical practice have suggested that rigorously designed guidelines can positively impact practice.29 Participants also noted that cSpO2 guidelines should be authoritative and that knowledge of guideline adoption by peer institutions was a facilitator of adoption. Usability issues negatively impact clinicians’ ability to follow guidelines.30 Further, prior studies have demonstrated that EHR integration of guidelines can change practice.31-33 Based on our findings, incorporating clear guidelines into commonly used formats, such as EHR order sets, could be an important deimplementation tool for cSpO2 in stable bronchiolitis patients.
Education about and awareness of cSpO2 guidelines was described as an important facilitator for appropriate cSpO2 use and was suggested as a potential deimplementation strategy. Participants noted that educational need may vary by stakeholder group. For example, education may facilitate obtaining buy-in from hospital leaders, which is necessary to support changes to the EHR. Education incorporating information on the typical features of bronchiolitis and examples of appropriate and inappropriate cSpO2 use was suggested for clinical team members. The limitations of education as a stand-alone deimplementation strategy were also noted, and participants highlighted challenges such as time needed for education and the need for ongoing education for rotating trainees. Inner and outer setting barriers, such as a perceived “culture of high pulse oximetryuse” and patient and family expectations, could also make education less effective as a stand-alone strategy. That—coupled with evidence that education and training alone are generally insufficient for producing reliable, sustained behavior change34,35—suggests that a multifaceted approach will be important.
Our respondents consider parental perceptions and preference in their practice, which provides nuance to recent studies suggesting that parents prefer continuous monitors when their child is hospitalized with bronchiolitis. Chi et al described the impact of a brief educational intervention on parental preferences for monitoring children hospitalized for bronchiolitis.36 This work suggests that educational interventions aimed at families should be considered in future (de)implementation studies because they may indirectly impact clinician behavior. Future studies should directly assess parental discomfort with discontinuing monitoring. Participants highlighted the link between knowledge and confidence in caring for typical bronchiolitis patients and monitoring practice, perceiving that less experienced clinicians are more likely to rely on cSpO2. Participants at high-use sites emphasized the expectation that monitoring should occur during hospitalizations. This reflection is particularly pertinent for bronchiolitis, a disease characterized by frequent, self-resolving desaturations even after hospital discharge.3 This may reinforce a perceived need to capture and react to these desaturation events even though they are expected in bronchiolitis and can occur in healthy infants.37 Some participants suggested that continuous monitoring be replaced with “nap tests” (ie, assessment for desaturations during a nap prior to discharge); however, like cSpO2 in stable infants with bronchiolitis, this is another low-value practice. Otherwise healthy infants with mild to moderate disease are unlikely to subsequently worsen after showing signs of clinical improvement.38 Nap tests are likely to lead to infants who are clinically improving being placed unnecessarily back on oxygen in reaction to the transient desaturations. Participants’ perception about the importance of cSpO2 in bronchiolitis management, despite evidence suggesting it is a low-value practice, underscores the importance of not simply telling clinicians to stop cSpO2. Employing strategies that replace continuous monitoring with another acceptable and feasible alternative (eg, regular clinician assessments including intermittent pulse oximetry checks) should be considered when planning for deimplementation.39
Previous studies indicate that continuous monitoring can affect clinician decision-making, independent of other factors,6,40 despite limited evidence that continuous monitors improve patient outcomes.1-7 Studies have demonstrated noticeable increase in admissions based purely on pulse oximetry values,40 with no evidence that this type of admission changes outcomes for bronchiolitis patients.6 One previous, single-center study identified inexperience as a potential driver for monitor use,41 and studies in adult populations have suggested that clinicians overestimate the value that continuous monitoring contributes to patient care,42,43 which promotes guideline-discordant use. Our study provides novel insight into the issue of monitoring in bronchiolitis. Our results suggest that there is a need to shift organizational cultures around monitoring (which likely vary based on a range of factors) and that educational strategies addressing typical disease course, especially desaturations, in bronchiolitis will be an essential component in any deimplementation effort.
This study is strengthened by its sample of diverse stakeholder groups from multiple US health systems. Additionally, we interviewed individuals at sites with high cSpO2 rates and at sites with low rates, as well as from community hospitals, children’s hospitals within general hospitals, and freestanding children’s hospitals, which allows us to understand barriers high-use sites encounter and facilitators of lower cSpO2 rates at low-use sites. We also employed an interview approach informed by an established implementation science framework. Nonetheless, several limitations exist. First, participants at low-use sites did not necessarily have direct experience with a previous deimplementation effort to reduce cSpO2. Additionally, participants were predominantly White and female; more diverse perspectives would strengthen confidence in the generalizability of our findings. While thematic saturation was achieved within each stakeholder group and within the high- and low-use strata, we interviewed fewer administrators and respiratory therapists relative to other stakeholder groups. Nevertheless, our conclusions were validated by our interdisciplinary stakeholder panel. As noted by participants, family preferences may influence clinician practice, and parents were not interviewed for this study. The information gleaned from the present study will inform the development of strategies to deimplement unnecessary cSpO2 in pediatric hospitals, which we aim to rigorously evaluate in a future trial.
CONCLUSION
We identified barriers and facilitators to deimplementation of cSpO2 for stable patients with bronchiolitis across children’s hospitals with high and low utilization of cSpO2. These themes map to multiple CFIR domains and, along with participant-suggested strategies, can directly inform an approach to cSpO2 deimplementation in a range of inpatient settings. Based on these data, future deimplementation efforts should focus on clear protocols for use and discontinuation of cSpO2, EHR changes, and regular bronchiolitis education for hospital staff that emphasizes reducing unnecessary cSpO2 utilization.
ACKNOWLEDGMENTS
We acknowledge the NHLBI scientists who contributed their expertise to this project as part of the U01 Cooperative Agreement funding mechanism as federal employees conducting their official job duties: Lora Reineck, MD, MS, Karen Bienstock, MS, and Cheryl Boyce, PhD. We thank the Executive Council of the Pediatric Research in Inpatient Settings (PRIS) Network for their contributions to the early scientific development of this project. The Network assessed a Collaborative Support Fee for access to the hospitals and support of this project. We thank the PRIS Network collaborators for their major contributions to data collection measuring utilization to identify the hospitals we subsequently chose for this project. We thank Claire Bocage and the Mixed Methods Research Lab for major help in data management and data analysis.
Continuous pulse oximetry monitoring (cSpO2) in children with bronchiolitis is associated with increased rates of hospital admission, longer lengths of stay, more frequent treatment with supplemental oxygen, alarm fatigue, and higher hospital cost. There is no evidence that it improves clinical outcomes.1-7 The safety of reducing cSpO2 for stable bronchiolitis patients (ie, those who are clinically well and not requiring supplemental oxygen) has been assessed in quality improvement initiatives8-10 and a randomized controlled trial.2 These studies showed no increase in intensive care unit transfers, codes, or readmissions associated with reduced cSpO2. Current national guidelines from the American Academy of Pediatrics5 and the Society of Hospital Medicine Choosing Wisely in Pediatric Hospital Medicine workgroup4 support limiting monitoring of children with bronchiolitis. Despite this, the practice of cSpO2 in stable bronchiolitis patients off supplemental oxygen remains widespread.11,12
Deimplementation, defined as reducing or stopping low-value or ineffective healthcare practices,13,14 is a discrete focus area within implementation science. Deimplementation research involves the reduction of unnecessary and overused services for which there is potential for harm or no benefit.15,16 In pediatrics, there are a number of potential targets for deimplementation,4,17-20 including cSpO2 for stable infants with bronchiolitis, but efforts to reduce low-value practices have met limited success to date. 21,22
Implementation science offers rigorous methods for advancing the development and evaluation of strategies for deimplementation.23 In particular, implementation science frameworks can facilitate our understanding of relevant contextual factors that may hinder or help efforts to deimplement low-value practices. To develop broadly applicable strategies to reduce monitoring overuse, it is important to understand the barriers, facilitators, and contextual factors (eg, clinical, political, interpersonal) that contribute to guideline-discordant cSpO2 in hospitalized bronchiolitis patients. Further, the process by which one can develop a rigorous understanding of these factors and how they may impact deimplementation efforts could generalize to other scenarios in pediatrics where overuse remains an issue.
The goal of this study was to use semistructured interviews, informed by an established implementation science framework, specifically the Consolidated Framework for Implementation Research (CFIR),24 to (1) identify barriers and facilitators to deimplementing unnecessary cSpO2, and (2) develop strategies to deimplement cSpO2 in a multicenter cohort of hospital-based clinician and administrative stakeholders.
METHODS
Study Setting
This multicenter qualitative study using semistructured interviews took place within the Eliminating Monitor Overuse (EMO) SpO2 study. The EMO SpO2 study established rates of cSpO2 in bronchiolitis patients not receiving supplemental oxygen or not receiving room air flow at 56 hospitals across the United States and in Canada from December 1, 2018, through March 31, 2019.12 The study identified hospital-level risk-adjusted cSpO2 rates ranging from 6% to 82%. A description of the EMO SpO2 study methods25 and its findings12 have been published elsewhere.
Participants
We approached EMO study site principal investigators at 12 hospitals: the two highest- and two lowest-use hospitals within three hospital types (ie, freestanding children’s hospitals, children’s hospitals within large general hospitals, and community hospitals). We collaborated with the participating site principal investigators (n = 12), who were primarily hospitalist physicians in leadership roles, to recruit a purposive sample of additional stakeholders including bedside nurses (n = 12), hospitalist physicians (n = 15), respiratory therapists (n = 9), and hospital administrators (n = 8) to participate in semistructured interviews. Interviews were conducted until we achieved thematic saturation within each stakeholder group and within the high and low performing strata (total 56 interviews). Participants were asked to self-report basic demographic information (see Appendix, interview guide) as required by the study funder and to allow us to comment on the representativeness of the participant group.
Procedure
The interview guide was informed by the CFIR, a comprehensive framework detailing contextual factors that require consideration when planning for the implementation of a health service intervention. Table 1 details the CFIR domains with study-related examples. The interview guide (Appendix) provided limited clinical context apart from the age, diagnosis, and oxygen requirement for the population of interest to promote a broad array of responses and to avoid anchoring on specific clinical scenarios. Interviews were conducted by master’s degree or doctoral-level research coordinators with qualitative interviewing experience and supervised by a medical anthropologist and qualitative methods expert (F.K.B.). Prior to engaging in audio recorded phone interviews, the interviewer explained the risks and benefits of participating. Participants were compensated $50. Audio recordings were transcribed, deidentified, and uploaded to NVivo 12 Plus (QSR International) for data management.

The Institutional Review Boards of Children’s Hospital of Philadelphia, Pennsylvania, and the University of Pennsylvania in Philadelphia determined that the study met eligibility criteria for IRB exemption.
Data Analysis
Using an integrated approach to codebook development,26 a priori codes were developed using constructs from the CFIR. Additional codes were added by the research team following a close reading of the first five transcripts.27,28 Each code was defined, including decision rules for its application. Two research coordinators independently coded each transcript. Using the intercoder reliability function within NVivo, the coders established strong interrater reliability accordance scores (
RESULTS
Barriers and facilitators to deimplementation were identified in multiple domains of the CFIR: outer setting, inner setting, characteristics of the individuals, and intervention characteristics (Table 1). Participants also suggested strategies to facilitate deimplementation in response to some identified barriers. See Table 2 for participant demographics and Table 3 for illustrative participant quotations.


Barriers
Outer Setting: Clinician Perceptions of Parental Discomfort With Discontinuing Monitoring
Participants mentioned parental preferences as a barrier to discontinuing cSpO2, noting that parents seem to take comfort in watching the numbers on the monitor screen and are reluctant to have it withdrawn. Clinicians noted that parents sometimes put the monitor back on their child after a clinician removed it or have expressed concern that their unmonitored child was not receiving the same level of care as other patients who were being monitored. In these scenarios, clinicians reported they have found it helpful to educate caregivers about when cSpO2 is and is not appropriate.
Inner Setting: Unclear or Nonexistent Guideline to Discontinue cSpO2
Guidelines to discontinue cSpO2 reportedly did not exist at all institutions. If a guideline did exist, lack of clarity or conflicting guidelines about when to use oxygen presented a barrier. Participants suggested that a clear guideline or additional oversight to ensure all clinicians are informed of the procedure for discontinuing cSpO2 may help prevent miscommunication. Participants noted that their electronic health record (EHR) order sets commonly included cSpO2 orders and that removing that option would facilitate deimplementation.
Inner Setting: Difficulty Educating All Staff
Participants noted difficulty with incorporating education about discontinuing cSpO2 to all clinicians, particularly to those who are nightshift only or to rotating staff or trainees. This created barriers for frequent re-education because these staff are not familiar with the policies and procedures of the unit, which is crucial to developing a culture that supports the deimplementation of cSpO2. Participants suggested that recurring education about procedures for discontinuing cSpO2 should target trainees, new nurses, and overnight nurses. This would help to ensure that the guideline is uniformly followed.
Inner Setting: Culture of High cSpO2 Use
Participants from high-use sites discussed a culture driven by readily available monitoring features or an expectation that monitoring indicates higher-quality care. Participants from low-use sites discussed increased cSpO2 driven by clinicians who were accustomed to caring for higher-acuity patients, for whom continuous monitoring is likely appropriate, and were simultaneously caring for stable bronchiolitis patients.
Some suggested that visual cues would be useful to clinicians to sustain awareness about a cSpO2 deimplementation guideline. It was also suggested that audit and feedback techniques like posting unit deimplementation statistics and creating a competition among units by posting unit performance could facilitate deimplementation. Additionally, some noted that visual aids in common spaces would be useful to remind clinicians and to engage caregivers about discontinuing cSpO2.
Characteristics of Individuals: Clinician Discomfort Discontinuing cSpO2
One frequently cited barrier across participants is that cSpO2 provides “peace of mind” to alert clinicians to patients with low oxygen saturations that might otherwise be missed. Participants identified that clinician discomfort with reducing cSpO2 may be driven by inexperienced clinicians less familiar with the bronchiolitis disease process, such as trainees, new nurses, or rotating clinicians unaccustomed to pediatric care. Trainees and new nurses were perceived as being more likely to work at night when there are fewer clinicians to provide patient care. Additionally, participants perceived that night shift clinicians favored cSpO2 because they could measure vital signs without waking patients and families.
Clinicians discussed that discontinuing cSpO2 would require alternative methods for assessing patient status, particularly for night shift nurses. Participants suggested strategies including changes to pulse oximetry assessment procedures to include more frequent “spot checks,” incorporation of assessments during sleep events (eg, naps) to ensure the patient does not experience desaturations during sleep, and training nurses to become more comfortable with suctioning patients. Suggestions also included education on the typical features of transient oxygen desaturations in otherwise stable patients with bronchiolitis2 to bolster clinical confidence for clinicians unfamiliar with caring for bronchiolitis patients. Participants perceived that education about appropriate vs inappropriate use may help to empower clinicians to employ cSpO2 appropriately.
Facilitators
Outer Setting: Standards and Evidence From Research, Professional Organizations, and Leaders in the Field
Many participants expressed the importance of consistent guidelines that are advocated by thought leaders in the field, supported by robust evidence, and consistent with approaches at peer hospitals. The more authoritative support a guideline has, the more comfortable people are adopting it and taking it seriously. Additionally, consistent education about guidelines was desired. Participants noted that all clinicians should be receiving education related to the American Academy of Pediatrics (AAP) Bronchiolitis and Choosing Wisely® guidelines, ranging from a one-time update to annually. Continual updates and re-education sessions for clinicians who shared evidence about how cSpO2 deimplementation could improve the quality of patient care by shortening hospital length of stay and lowering cost were suggested strategies.
Inner Setting: Leadership
Participants noted that successful deimplementation depends upon the presence of a champion or educator who will be able to lead the institutional charge in making practice change. This is typically an individual who is trusted at the institution, experienced in their field, or already doing implementation work. This could be either a single individual (champion) or a team. The most commonly noted clinician roles to engage in a leadership role or team were physicians and nurses.
Participants noted that a change in related clinical care pathways or EHR order sets would require cooperation from multiple clinical disciplines, administrators, and information technology leaders and explained that messaging and education about the value of the change would facilitate buy-in from those clinicians.
Inner Setting: EHR Support for Guidelines
Participants often endorsed the use of an order set within the EHR that supports guidelines and includes reminders to decrease cSpO2. These reminders could come up when supplemental oxygen is discontinued or occur regularly throughout the patient’s stay to prompt the clinician to consider discontinuing cSpO2.
Intervention Characteristics/Inner Setting: Clear Bronchiolitis Guidelines
The presence of a well-articulated hospital policy that delineates the appropriate and inappropriate use of cSpO2 in bronchiolitis was mentioned as another facilitator of deimplementation.
DISCUSSION
Results of this qualitative study of stakeholders across hospitals with high and low cSpO2 use illustrated the complexities involved with deimplementation of cSpO2 in pediatric patients hospitalized with bronchiolitis. We identified numerous barriers spanning the CFIR constructs, including unclear or absent guidelines for stopping cSpO2, clinician knowledge and comfort with bronchiolitis disease features, and unit culture. This suggests that multicomponent strategies that target various domains and a variety of stakeholders are needed to deimplement cSpO2 use for stable bronchiolitis patients. Participants also identified facilitators, including clear cSpO2 guidelines, supportive leaders and champions, and EHR modifications, that provide insight into strategies that may help sites reduce their use of cSpO2. Additionally, participants also provided concrete, actionable suggestions for ways to reduce unnecessary monitoring that will be useful in informing promising deimplementation strategies for subsequent trials.
The importance of having specific and well-known guidelines from trusted sources, such as the AAP, about cSpO2 and bronchiolitis treatment that are thoughtfully integrated in the EHR came through in multiple themes of our analysis. Prior studies on the effect of guidelines on clinical practice have suggested that rigorously designed guidelines can positively impact practice.29 Participants also noted that cSpO2 guidelines should be authoritative and that knowledge of guideline adoption by peer institutions was a facilitator of adoption. Usability issues negatively impact clinicians’ ability to follow guidelines.30 Further, prior studies have demonstrated that EHR integration of guidelines can change practice.31-33 Based on our findings, incorporating clear guidelines into commonly used formats, such as EHR order sets, could be an important deimplementation tool for cSpO2 in stable bronchiolitis patients.
Education about and awareness of cSpO2 guidelines was described as an important facilitator for appropriate cSpO2 use and was suggested as a potential deimplementation strategy. Participants noted that educational need may vary by stakeholder group. For example, education may facilitate obtaining buy-in from hospital leaders, which is necessary to support changes to the EHR. Education incorporating information on the typical features of bronchiolitis and examples of appropriate and inappropriate cSpO2 use was suggested for clinical team members. The limitations of education as a stand-alone deimplementation strategy were also noted, and participants highlighted challenges such as time needed for education and the need for ongoing education for rotating trainees. Inner and outer setting barriers, such as a perceived “culture of high pulse oximetryuse” and patient and family expectations, could also make education less effective as a stand-alone strategy. That—coupled with evidence that education and training alone are generally insufficient for producing reliable, sustained behavior change34,35—suggests that a multifaceted approach will be important.
Our respondents consider parental perceptions and preference in their practice, which provides nuance to recent studies suggesting that parents prefer continuous monitors when their child is hospitalized with bronchiolitis. Chi et al described the impact of a brief educational intervention on parental preferences for monitoring children hospitalized for bronchiolitis.36 This work suggests that educational interventions aimed at families should be considered in future (de)implementation studies because they may indirectly impact clinician behavior. Future studies should directly assess parental discomfort with discontinuing monitoring. Participants highlighted the link between knowledge and confidence in caring for typical bronchiolitis patients and monitoring practice, perceiving that less experienced clinicians are more likely to rely on cSpO2. Participants at high-use sites emphasized the expectation that monitoring should occur during hospitalizations. This reflection is particularly pertinent for bronchiolitis, a disease characterized by frequent, self-resolving desaturations even after hospital discharge.3 This may reinforce a perceived need to capture and react to these desaturation events even though they are expected in bronchiolitis and can occur in healthy infants.37 Some participants suggested that continuous monitoring be replaced with “nap tests” (ie, assessment for desaturations during a nap prior to discharge); however, like cSpO2 in stable infants with bronchiolitis, this is another low-value practice. Otherwise healthy infants with mild to moderate disease are unlikely to subsequently worsen after showing signs of clinical improvement.38 Nap tests are likely to lead to infants who are clinically improving being placed unnecessarily back on oxygen in reaction to the transient desaturations. Participants’ perception about the importance of cSpO2 in bronchiolitis management, despite evidence suggesting it is a low-value practice, underscores the importance of not simply telling clinicians to stop cSpO2. Employing strategies that replace continuous monitoring with another acceptable and feasible alternative (eg, regular clinician assessments including intermittent pulse oximetry checks) should be considered when planning for deimplementation.39
Previous studies indicate that continuous monitoring can affect clinician decision-making, independent of other factors,6,40 despite limited evidence that continuous monitors improve patient outcomes.1-7 Studies have demonstrated noticeable increase in admissions based purely on pulse oximetry values,40 with no evidence that this type of admission changes outcomes for bronchiolitis patients.6 One previous, single-center study identified inexperience as a potential driver for monitor use,41 and studies in adult populations have suggested that clinicians overestimate the value that continuous monitoring contributes to patient care,42,43 which promotes guideline-discordant use. Our study provides novel insight into the issue of monitoring in bronchiolitis. Our results suggest that there is a need to shift organizational cultures around monitoring (which likely vary based on a range of factors) and that educational strategies addressing typical disease course, especially desaturations, in bronchiolitis will be an essential component in any deimplementation effort.
This study is strengthened by its sample of diverse stakeholder groups from multiple US health systems. Additionally, we interviewed individuals at sites with high cSpO2 rates and at sites with low rates, as well as from community hospitals, children’s hospitals within general hospitals, and freestanding children’s hospitals, which allows us to understand barriers high-use sites encounter and facilitators of lower cSpO2 rates at low-use sites. We also employed an interview approach informed by an established implementation science framework. Nonetheless, several limitations exist. First, participants at low-use sites did not necessarily have direct experience with a previous deimplementation effort to reduce cSpO2. Additionally, participants were predominantly White and female; more diverse perspectives would strengthen confidence in the generalizability of our findings. While thematic saturation was achieved within each stakeholder group and within the high- and low-use strata, we interviewed fewer administrators and respiratory therapists relative to other stakeholder groups. Nevertheless, our conclusions were validated by our interdisciplinary stakeholder panel. As noted by participants, family preferences may influence clinician practice, and parents were not interviewed for this study. The information gleaned from the present study will inform the development of strategies to deimplement unnecessary cSpO2 in pediatric hospitals, which we aim to rigorously evaluate in a future trial.
CONCLUSION
We identified barriers and facilitators to deimplementation of cSpO2 for stable patients with bronchiolitis across children’s hospitals with high and low utilization of cSpO2. These themes map to multiple CFIR domains and, along with participant-suggested strategies, can directly inform an approach to cSpO2 deimplementation in a range of inpatient settings. Based on these data, future deimplementation efforts should focus on clear protocols for use and discontinuation of cSpO2, EHR changes, and regular bronchiolitis education for hospital staff that emphasizes reducing unnecessary cSpO2 utilization.
ACKNOWLEDGMENTS
We acknowledge the NHLBI scientists who contributed their expertise to this project as part of the U01 Cooperative Agreement funding mechanism as federal employees conducting their official job duties: Lora Reineck, MD, MS, Karen Bienstock, MS, and Cheryl Boyce, PhD. We thank the Executive Council of the Pediatric Research in Inpatient Settings (PRIS) Network for their contributions to the early scientific development of this project. The Network assessed a Collaborative Support Fee for access to the hospitals and support of this project. We thank the PRIS Network collaborators for their major contributions to data collection measuring utilization to identify the hospitals we subsequently chose for this project. We thank Claire Bocage and the Mixed Methods Research Lab for major help in data management and data analysis.
1. Cunningham S, Rodriguez A, Adams T, et al; Bronchiolitis of Infancy Discharge Study (BIDS) group. Oxygen saturation targets in infants with bronchiolitis (BIDS): a double-blind, randomised, equivalence trial. Lancet. 2015;386(9998):1041-1048. https://doi.org/10.1016/s0140-6736(15)00163-4
2. McCulloh R, Koster M, Ralston S, et al. Use of intermittent vs continuous pulse oximetry for nonhypoxemic infants and young children hospitalized for bronchiolitis: a randomized clinical trial. JAMA Pediatr. 2015;169(10):898-904. https://doi.org/10.1001/jamapediatrics.2015.1746
3. Principi T, Coates AL, Parkin PC, Stephens D, DaSilva Z, Schuh S. Effect of oxygen desaturations on subsequent medical visits in infants discharged from the emergency department with bronchiolitis. JAMA Pediatr. 2016;170(6):602-608. https://doi.org/10.1001/jamapediatrics.2016.0114
4. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. https://doi.org/10.1002/jhm.2064
5. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
6. Schuh S, Freedman S, Coates A, et al. Effect of oximetry on hospitalization in bronchiolitis: a randomized clinical trial. JAMA. 2014;312(7):712-718. https://doi.org/10.1001/jama.2014.8637
7. Schuh S, Kwong JC, Holder L, Graves E, Macdonald EM, Finkelstein Y. Predictors of critical care and mortality in bronchiolitis after emergency department discharge. J Pediatr. 2018;199:217-222 e211. https://doi.org/10.1016/j.jpeds.2018.04.010
8. Schondelmeyer AC, Simmons JM, Statile AM, et al. Using quality improvement to reduce continuous pulse oximetry use in children with wheezing. Pediatrics. 2015;135(4):e1044-e1051. https://doi.org/10.1542/peds.2014-2295
9. Mittal S, Marlowe L, Blakeslee S, et al. Successful use of quality improvement methodology to reduce inpatient length of stay in bronchiolitis through judicious use of intermittent pulse oximetry. Hosp Pediatr. 2019;9(2):73-78. https://doi.org/10.1542/hpeds.2018-0023
10. Heneghan M, Hart J, Dewan M, et al. No Cause for Alarm: Decreasing inappropriate pulse oximetry use in bronchiolitis. Hosp Pediatr. 2018;8(2):109-111. https://doi.org/10.1542/hpeds.2017-0126
11. Ralston S, Garber M, Narang S, et al. Decreasing unnecessary utilization in acute bronchiolitis care: results from the value in inpatient pediatrics network. J Hosp Med. 2013;8(1):25-30. https://doi.org/10.1002/jhm.1982
12. Bonafide CP, Xiao R, Brady PW, et al. Prevalence of continuous pulse oximetry monitoring in hospitalized children with bronchiolitis not requiring supplemental oxygen. JAMA. 2020;323(15):1467-1477. https://doi.org/10.1001/jama.2020.2998
13. van Bodegom-Vos L, Davidoff F, Marang-van de Mheen PJ. Implementation and de-implementation: two sides of the same coin? BMJ Qual Saf. 2017;26(6):495-501. https://doi.org/10.1136/bmjqs-2016-005473
14. McKay VR, Morshed AB, Brownson RC, Proctor EK, Prusaczyk B. Letting go: conceptualizing intervention de-implementation in public health and social service settings. Am J Community Psychol. 2018;62(1-2):189-202. https://doi.org/10.1002/ajcp.12258
15. Brownlee S, Chalkidou K, Doust J, et al. Evidence for overuse of medical services around the world. Lancet. 2017;390(10090):156-168. https://doi.org/10.1016/s0140-6736(16)32585-5
16. Chassin MR, Galvin RW. The urgent need to improve health care quality. Institute of Medicine National roundtable on health care quality. JAMA. 1998;280(11):1000-1005. https://doi.org/10.1001/jama.280.11.1000
17. Coon ER, Young PC, Quinonez RA, Morgan DJ, Dhruva SS, Schroeder AR. 2017 update on pediatric medical overuse: a review. JAMA Pediatr. 2018;172(5):482-486. https://doi.org/10.1001/jamapediatrics.2017.5752
18. Schuh S, Babl FE, Dalziel SR, et al; Pediatric Emergency Research Networks (PERN). Practice variation in acute bronchiolitis: a Pediatric Emergency Research Networks study. Pediatrics. 2017;140(6):e20170842. https://doi.org/10.1542/peds.2017-0842
19. Lewis-de Los Angeles WW, Thurm C, Hersh AL, et al. Trends in intravenous antibiotic duration for urinary tract infections in young infants. Pediatrics. 2017;140(6):e20171021. https://doi.org/10.1542/peds.2017-1021
20. Parikh K, Hall M, Mittal V, et al. Establishing benchmarks for the hospitalized care of children with asthma, bronchiolitis, and pneumonia. Pediatrics. 2014;134(3):555-562. https://doi.org/10.1542/peds.2014-1052
21. Ralston SL, Garber MD, Rice-Conboy E, et al; Value in Inpatient Pediatrics Network Quality Collaborative for Improving Hospital Compliance with AAP Bronchiolitis Guideline (BQIP). A multicenter collaborative to reduce unnecessary care in inpatient bronchiolitis. Pediatrics. 2016;137(1):e20150851. https://doi.org/10.1542/peds.2015-0851
22. Reyes MA, Etinger V, Hall M, et al. Impact of the Choosing Wisely((R)) Campaign recommendations for hospitalized children on clinical practice: trends from 2008 to 2017. J Hosp Med. 2020;15(2):68-74. https://doi.org/10.12788/jhm.3291
23. Norton WE, Chambers DA. Unpacking the complexities of de-implementing inappropriate health interventions. Implement Sci. 2020;15(1):2. https://doi.org/10.1186/s13012-019-0960-9
24. Damschroder LJ, Aron DC, Keith RE, Kirsh SR, Alexander JA, Lowery JC. Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci. 2009;4:50. https://doi.org/10.1186/1748-5908-4-50
25. Rasooly IR, Beidas RS, Wolk CB, et al. Measuring overuse of continuous pulse oximetry in bronchiolitis and developing strategies for large-scale deimplementation: study protocol for a feasibility trial. Pilot Feasibility Stud. 2019;5:68. https://doi.org/10.1186/s40814-019-0453-2
26. Bradley EH, Curry LA, Devers KJ. Qualitative data analysis for health services research: developing taxonomy, themes, and theory. Health Serv Res. 2007;42(4):1758-1772. https://doi.org/10.1111/j.1475-6773.2006.00684.x
27. Glaser BG, Strauss AL. The Discovery of Grounded Theory: Strategies for Qualitative Research. Aldine Pub. Co.; 1967.
28. Charmaz K. Grounded Theory: Objectivist and Constructivist Methods. In: Denzin NK, Lincoln Y, eds. Handbook of Qualitative Research. 2nd ed. Sage Publications; 2000:509-535.
29. Grimshaw JM, Russell IT. Effect of clinical guidelines on medical practice: a systematic review of rigorous evaluations. Lancet. 1993;342(8883):1317-1322. https://doi.org/10.1016/0140-6736(93)92244-n
30. Cabana MD, Rand CS, Powe NR, et al. Why don’t physicians follow clinical practice guidelines? a framework for improvement. JAMA. 1999;282(15):1458-1465. https://doi.org/10.1001/jama.282.15.1458
31. Dressler R, Dryer MM, Coletti C, Mahoney D, Doorey AJ. Altering overuse of cardiac telemetry in non-intensive care unit settings by hardwiring the use of American Heart Association guidelines. JAMA Intern Med. 2014;174(11):1852-1854. https://doi.org/10.1001/jamainternmed.2014.4491
32. Forrest CB, Fiks AG, Bailey LC, et al. Improving adherence to otitis media guidelines with clinical decision support and physician feedback. Pediatrics. 2013;131(4):e1071-e1081. https://doi.org/10.1542/peds.2012-1988
33. Fiks AG, Grundmeier RW, Mayne S, et al. Effectiveness of decision support for families, clinicians, or both on HPV vaccine receipt. Pediatrics. 2013;131(6):1114-1124. https://doi.org/10.1542/peds.2012-3122
34. Nolan T, Resar R, Griffin F, Gordon AB. Improving the Reliability of Health Care. Institute for Healthcare Improvement; 2004. http://www.ihi.org/resources/Pages/IHIWhitePapers/ImprovingtheReliabilityofHealthCare.aspx
35. Beidas RS, Kendall PC. Training Therapists in evidence-based practice: a critical review of studies from a systems-contextual perspective. Clin Psychol (New York). 2010;17(1):1-30. https://doi.org/10.1111/j.1468-2850.2009.01187.x
36. Chi KW, Coon ER, Destino L, Schroeder AR. Parental perspectives on continuous pulse oximetry use in bronchiolitis hospitalizations. Pediatrics. 2020;146(2):e20200130. https://doi.org/10.1542/peds.2020-0130
37. Hunt CE, Corwin MJ, Lister G, et al. Longitudinal assessment of hemoglobin oxygen saturation in healthy infants during the first 6 months of age. Collaborative Home Infant Monitoring Evaluation (CHIME) Study Group. J Pediatr. 1999;135(5):580-586. https://doi.org/10.1016/s0022-3476(99)70056-9
38. Mansbach JM, Clark S, Piedra PA, et al; MARC-30 Investigators. Hospital course and discharge criteria for children hospitalized with bronchiolitis. J Hosp Med. 2015;10(4):205-211. https://doi.org/10.1002/jhm.2318
39. Burton C, Williams L, Bucknall T, et al. Understanding how and why de-implementation works in health and care: research protocol for a realist synthesis of evidence. Syst Rev. 2019;8(1):194. https://doi.org/10.1186/s13643-019-1111-840. Mallory MD, Shay DK, Garrett J, Bordley WC. Bronchiolitis management preferences and the influence of pulse oximetry and respiratory rate on the decision to admit. Pediatrics. 2003;111(1):e45-51. https://doi.org/10.1542/peds.111.1.e45.
41. Schondelmeyer AC, Jenkins AM, Allison B, et al. Factors influencing use of continuous physiologic monitors for hospitalized pediatric patients. Hosp Pediatr. 2019;9(6):423-428. https://doi.org/10.1542/hpeds.2019-0007
42. Najafi N, Auerbach A. Use and outcomes of telemetry monitoring on a medicine service. Arch Intern Med. 2012;172(17):1349-1350. https://doi.org/10.1001/archinternmed.2012.3163
43. Estrada CA, Rosman HS, Prasad NK, et al. Role of telemetry monitoring in the non-intensive care unit. Am J Cardiol. 1995;76(12):960-965. https://doi.org/10.1016/s0002-9149(99)80270-7
1. Cunningham S, Rodriguez A, Adams T, et al; Bronchiolitis of Infancy Discharge Study (BIDS) group. Oxygen saturation targets in infants with bronchiolitis (BIDS): a double-blind, randomised, equivalence trial. Lancet. 2015;386(9998):1041-1048. https://doi.org/10.1016/s0140-6736(15)00163-4
2. McCulloh R, Koster M, Ralston S, et al. Use of intermittent vs continuous pulse oximetry for nonhypoxemic infants and young children hospitalized for bronchiolitis: a randomized clinical trial. JAMA Pediatr. 2015;169(10):898-904. https://doi.org/10.1001/jamapediatrics.2015.1746
3. Principi T, Coates AL, Parkin PC, Stephens D, DaSilva Z, Schuh S. Effect of oxygen desaturations on subsequent medical visits in infants discharged from the emergency department with bronchiolitis. JAMA Pediatr. 2016;170(6):602-608. https://doi.org/10.1001/jamapediatrics.2016.0114
4. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. https://doi.org/10.1002/jhm.2064
5. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
6. Schuh S, Freedman S, Coates A, et al. Effect of oximetry on hospitalization in bronchiolitis: a randomized clinical trial. JAMA. 2014;312(7):712-718. https://doi.org/10.1001/jama.2014.8637
7. Schuh S, Kwong JC, Holder L, Graves E, Macdonald EM, Finkelstein Y. Predictors of critical care and mortality in bronchiolitis after emergency department discharge. J Pediatr. 2018;199:217-222 e211. https://doi.org/10.1016/j.jpeds.2018.04.010
8. Schondelmeyer AC, Simmons JM, Statile AM, et al. Using quality improvement to reduce continuous pulse oximetry use in children with wheezing. Pediatrics. 2015;135(4):e1044-e1051. https://doi.org/10.1542/peds.2014-2295
9. Mittal S, Marlowe L, Blakeslee S, et al. Successful use of quality improvement methodology to reduce inpatient length of stay in bronchiolitis through judicious use of intermittent pulse oximetry. Hosp Pediatr. 2019;9(2):73-78. https://doi.org/10.1542/hpeds.2018-0023
10. Heneghan M, Hart J, Dewan M, et al. No Cause for Alarm: Decreasing inappropriate pulse oximetry use in bronchiolitis. Hosp Pediatr. 2018;8(2):109-111. https://doi.org/10.1542/hpeds.2017-0126
11. Ralston S, Garber M, Narang S, et al. Decreasing unnecessary utilization in acute bronchiolitis care: results from the value in inpatient pediatrics network. J Hosp Med. 2013;8(1):25-30. https://doi.org/10.1002/jhm.1982
12. Bonafide CP, Xiao R, Brady PW, et al. Prevalence of continuous pulse oximetry monitoring in hospitalized children with bronchiolitis not requiring supplemental oxygen. JAMA. 2020;323(15):1467-1477. https://doi.org/10.1001/jama.2020.2998
13. van Bodegom-Vos L, Davidoff F, Marang-van de Mheen PJ. Implementation and de-implementation: two sides of the same coin? BMJ Qual Saf. 2017;26(6):495-501. https://doi.org/10.1136/bmjqs-2016-005473
14. McKay VR, Morshed AB, Brownson RC, Proctor EK, Prusaczyk B. Letting go: conceptualizing intervention de-implementation in public health and social service settings. Am J Community Psychol. 2018;62(1-2):189-202. https://doi.org/10.1002/ajcp.12258
15. Brownlee S, Chalkidou K, Doust J, et al. Evidence for overuse of medical services around the world. Lancet. 2017;390(10090):156-168. https://doi.org/10.1016/s0140-6736(16)32585-5
16. Chassin MR, Galvin RW. The urgent need to improve health care quality. Institute of Medicine National roundtable on health care quality. JAMA. 1998;280(11):1000-1005. https://doi.org/10.1001/jama.280.11.1000
17. Coon ER, Young PC, Quinonez RA, Morgan DJ, Dhruva SS, Schroeder AR. 2017 update on pediatric medical overuse: a review. JAMA Pediatr. 2018;172(5):482-486. https://doi.org/10.1001/jamapediatrics.2017.5752
18. Schuh S, Babl FE, Dalziel SR, et al; Pediatric Emergency Research Networks (PERN). Practice variation in acute bronchiolitis: a Pediatric Emergency Research Networks study. Pediatrics. 2017;140(6):e20170842. https://doi.org/10.1542/peds.2017-0842
19. Lewis-de Los Angeles WW, Thurm C, Hersh AL, et al. Trends in intravenous antibiotic duration for urinary tract infections in young infants. Pediatrics. 2017;140(6):e20171021. https://doi.org/10.1542/peds.2017-1021
20. Parikh K, Hall M, Mittal V, et al. Establishing benchmarks for the hospitalized care of children with asthma, bronchiolitis, and pneumonia. Pediatrics. 2014;134(3):555-562. https://doi.org/10.1542/peds.2014-1052
21. Ralston SL, Garber MD, Rice-Conboy E, et al; Value in Inpatient Pediatrics Network Quality Collaborative for Improving Hospital Compliance with AAP Bronchiolitis Guideline (BQIP). A multicenter collaborative to reduce unnecessary care in inpatient bronchiolitis. Pediatrics. 2016;137(1):e20150851. https://doi.org/10.1542/peds.2015-0851
22. Reyes MA, Etinger V, Hall M, et al. Impact of the Choosing Wisely((R)) Campaign recommendations for hospitalized children on clinical practice: trends from 2008 to 2017. J Hosp Med. 2020;15(2):68-74. https://doi.org/10.12788/jhm.3291
23. Norton WE, Chambers DA. Unpacking the complexities of de-implementing inappropriate health interventions. Implement Sci. 2020;15(1):2. https://doi.org/10.1186/s13012-019-0960-9
24. Damschroder LJ, Aron DC, Keith RE, Kirsh SR, Alexander JA, Lowery JC. Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci. 2009;4:50. https://doi.org/10.1186/1748-5908-4-50
25. Rasooly IR, Beidas RS, Wolk CB, et al. Measuring overuse of continuous pulse oximetry in bronchiolitis and developing strategies for large-scale deimplementation: study protocol for a feasibility trial. Pilot Feasibility Stud. 2019;5:68. https://doi.org/10.1186/s40814-019-0453-2
26. Bradley EH, Curry LA, Devers KJ. Qualitative data analysis for health services research: developing taxonomy, themes, and theory. Health Serv Res. 2007;42(4):1758-1772. https://doi.org/10.1111/j.1475-6773.2006.00684.x
27. Glaser BG, Strauss AL. The Discovery of Grounded Theory: Strategies for Qualitative Research. Aldine Pub. Co.; 1967.
28. Charmaz K. Grounded Theory: Objectivist and Constructivist Methods. In: Denzin NK, Lincoln Y, eds. Handbook of Qualitative Research. 2nd ed. Sage Publications; 2000:509-535.
29. Grimshaw JM, Russell IT. Effect of clinical guidelines on medical practice: a systematic review of rigorous evaluations. Lancet. 1993;342(8883):1317-1322. https://doi.org/10.1016/0140-6736(93)92244-n
30. Cabana MD, Rand CS, Powe NR, et al. Why don’t physicians follow clinical practice guidelines? a framework for improvement. JAMA. 1999;282(15):1458-1465. https://doi.org/10.1001/jama.282.15.1458
31. Dressler R, Dryer MM, Coletti C, Mahoney D, Doorey AJ. Altering overuse of cardiac telemetry in non-intensive care unit settings by hardwiring the use of American Heart Association guidelines. JAMA Intern Med. 2014;174(11):1852-1854. https://doi.org/10.1001/jamainternmed.2014.4491
32. Forrest CB, Fiks AG, Bailey LC, et al. Improving adherence to otitis media guidelines with clinical decision support and physician feedback. Pediatrics. 2013;131(4):e1071-e1081. https://doi.org/10.1542/peds.2012-1988
33. Fiks AG, Grundmeier RW, Mayne S, et al. Effectiveness of decision support for families, clinicians, or both on HPV vaccine receipt. Pediatrics. 2013;131(6):1114-1124. https://doi.org/10.1542/peds.2012-3122
34. Nolan T, Resar R, Griffin F, Gordon AB. Improving the Reliability of Health Care. Institute for Healthcare Improvement; 2004. http://www.ihi.org/resources/Pages/IHIWhitePapers/ImprovingtheReliabilityofHealthCare.aspx
35. Beidas RS, Kendall PC. Training Therapists in evidence-based practice: a critical review of studies from a systems-contextual perspective. Clin Psychol (New York). 2010;17(1):1-30. https://doi.org/10.1111/j.1468-2850.2009.01187.x
36. Chi KW, Coon ER, Destino L, Schroeder AR. Parental perspectives on continuous pulse oximetry use in bronchiolitis hospitalizations. Pediatrics. 2020;146(2):e20200130. https://doi.org/10.1542/peds.2020-0130
37. Hunt CE, Corwin MJ, Lister G, et al. Longitudinal assessment of hemoglobin oxygen saturation in healthy infants during the first 6 months of age. Collaborative Home Infant Monitoring Evaluation (CHIME) Study Group. J Pediatr. 1999;135(5):580-586. https://doi.org/10.1016/s0022-3476(99)70056-9
38. Mansbach JM, Clark S, Piedra PA, et al; MARC-30 Investigators. Hospital course and discharge criteria for children hospitalized with bronchiolitis. J Hosp Med. 2015;10(4):205-211. https://doi.org/10.1002/jhm.2318
39. Burton C, Williams L, Bucknall T, et al. Understanding how and why de-implementation works in health and care: research protocol for a realist synthesis of evidence. Syst Rev. 2019;8(1):194. https://doi.org/10.1186/s13643-019-1111-840. Mallory MD, Shay DK, Garrett J, Bordley WC. Bronchiolitis management preferences and the influence of pulse oximetry and respiratory rate on the decision to admit. Pediatrics. 2003;111(1):e45-51. https://doi.org/10.1542/peds.111.1.e45.
41. Schondelmeyer AC, Jenkins AM, Allison B, et al. Factors influencing use of continuous physiologic monitors for hospitalized pediatric patients. Hosp Pediatr. 2019;9(6):423-428. https://doi.org/10.1542/hpeds.2019-0007
42. Najafi N, Auerbach A. Use and outcomes of telemetry monitoring on a medicine service. Arch Intern Med. 2012;172(17):1349-1350. https://doi.org/10.1001/archinternmed.2012.3163
43. Estrada CA, Rosman HS, Prasad NK, et al. Role of telemetry monitoring in the non-intensive care unit. Am J Cardiol. 1995;76(12):960-965. https://doi.org/10.1016/s0002-9149(99)80270-7
© 2021 Society of Hospital Medicine
Email: [email protected]; Telephone: 513-803-9158; Twitter: @SchondelmeyerMD.
Association Between Bronchiolitis Patient Volume and Continuous Pulse Oximetry Monitoring in 25 Hospitals
Continuous pulse oximetry monitoring in children with bronchiolitis who don’t require supplemental oxygen is discouraged by practice guidelines and is recognized as a form of medical overuse.1-3 This practice can be associated with negative outcomes, including prolonged length of stay,4-6 increased cost of hospitalization,7 and alarm fatigue among nurses.8 Despite initiatives to reduce continuous pulse oximetry monitoring in stable patients with bronchiolitis,1,2 wide practice variation exists between hospitals.9,10 Previous studies have shown that higher prevalence of inpatient bronchiolitis admissions is associated with decreased utilization of unnecessary interventions.11 However, the relationship between pulse oximetry use and bronchiolitis prevalence has not been studied. The objective of this study is to test the hypothesis that hospital units with lower proportions of patients admitted for bronchiolitis and those with fewer general pediatrics patients relative to subspecialty patients would have higher rates of pulse oximetry overuse.
METHODS
Study Design
We conducted a substudy of the Pediatric Research in Inpatient Settings (PRIS) Network’s Eliminating Monitoring Overuse (EMO) pulse oximetry study,10,12 a 56-hospital cross-sectional study that used direct observation to measure the prevalence of continuous pulse oximetry monitoring in hospitalized infants with bronchiolitis who did not require supplemental oxygen between December 1, 2018, through March 31, 2019. This substudy was not included as part of the original aims of the project and was proposed as a separate analysis during data collection. For US sites, the Institutional Review Board (IRB) at Children’s Hospital of Philadelphia approved the study and served as the central IRB. The Research Ethics Board at University of Calgary also approved the study.
Site Selection
Hospitals with at least 60 observations were eligible for inclusion. Of the 32 hospitals that conducted the minimum observations, 25 agreed to participate (21 free-standing children’s hospitals, 3 children’s hospitals within general hospitals, and 1 community hospital).
Patient Population
The parent study included patients aged 8 weeks through 23 months with a primary diagnosis of bronchiolitis. Patients were included only if they were not receiving supplemental oxygen or nasal cannula flow at the time of data collection. The inclusion and exclusion criteria were for both the parent study and the substudy. Further inclusion and exclusion criteria have been described previously.10,12
Data Collection
In order to ascertain continuous pulse oximetry monitoring status, staff at each hospital performed observational rounds by walking to the bedside of each patient who met inclusion criteria. Additional methodology for the parent study has been published elsewhere.10,12
Bronchiolitis Admission Volume by Unit
Collaborators at each hospital gathered bronchiolitis census data from each unit that admitted patients with bronchiolitis. Units were identified prior to data collection and were characterized at the institution level based on previous local definitions. Each site was responsible for using institution-specific data collection methods for determining bronchiolitis and total admissions on each unit (eg, departmental reports or directly querying admissions data using International Classification of Diseases, Tenth Revision, diagnosis codes for bronchiolitis) over the same period as the parent study. Following data analysis, bronchiolitis admission burden was classified into five categories, based on less than 10%, 10% to less than 20%, 20% to less than 30%, 30% to less than 40%, or 40% or more of total admissions having a primary discharge diagnosis of bronchiolitis during the study period. This categorization allowed investigators to determine whether there was a dose-dependent response among categories.
Unit Composition
Site investigators also completed a survey identifying which patients were admitted to each unit (eg, general pediatrics only, medical subspecialty, surgical). Based on these results, units were further classified into seven types (Appendix Table). For the final analysis, units caring exclusively for general pediatrics patients were compared to all other unit types.
Analysis
Bronchiolitis admission burden and unit composition data were combined with observations of pulse oximetry monitoring use of patients not requiring supplemental oxygen from the parent study. We determined unadjusted observed monitoring proportions for each unit’s bronchiolitis admission burden category across all 25 hospitals. This was calculated as a simple proportion of the total number of observations during which patients were continuously monitored divided by the total number of observations performed within each unit’s admission category. We then calculated unadjusted odds ratios using the 40% and higher bronchiolitis admission burden category as a reference. We calculated similar proportions and odds ratios for the dichotomous unit composition variable. Next, we used mixed-effects logistic regression with a random intercept for each hospital to allow for differences in baseline monitoring rates, which varied widely between hospitals (2% to 92%),10 to calculate adjusted odds ratios for the unit’s admission category and unit’s composition. We also adjusted for the same covariates used in the primary study’s analysis (Table).10
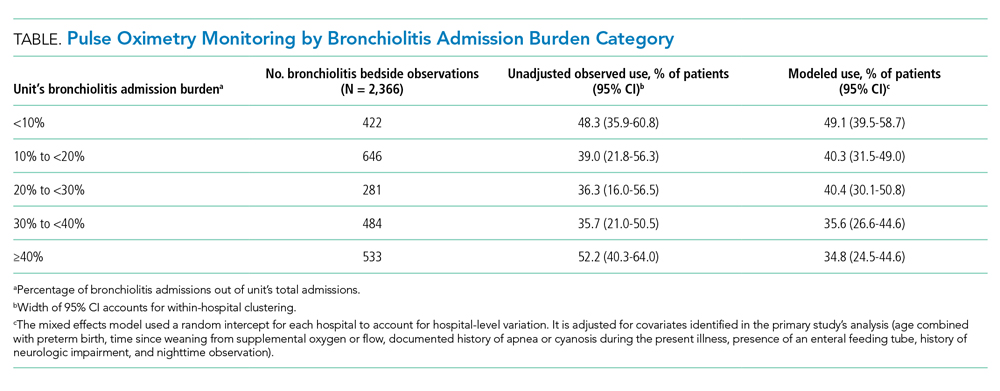
RESULTS
We analyzed 2,366 observations of bronchiolitis patients from 25 hospitals. Most observations were concentrated in freestanding children’s hospitals (89%), and 50% were from hospitals with more than 250 pediatric beds. Observations were well distributed among the five categories of admit burden (Table).
In unadjusted regression, the relationship between admission burden and rate of pulse oximetry use did not appear to be dose-dependent, and 95% CIs were wide. We then analyzed the data accounting for baseline differences in hospital monitoring rates and adjusted for the covariates significantly associated with continuous pulse oximetry monitoring in the primary study’s analysis with use of a mixed-effects model. As shown in the Table, low-burden units in which bronchiolitis constituted less than 10% of total admissions had a 2.16-fold increased odds of unnecessary pulse oximetry monitoring compared to high-burden units in which bronchiolitis constituted 40% or more of total admissions (95% CI, 1.27-3.69; P = .01).
In examining the subspecialty unit composition, 596 observations (25.2%) were conducted on units exclusively caring for general pediatrics patients. In the mixed-effects model adjusted for bronchiolitis admission burden and the covariates used in the study’s primary analysis, units exclusively caring for general pediatrics patients did not have significantly different independent odds of pulse oximetry monitoring use compared to units with a mixed patient population (OR 1.01; 95% CI, 0.71-1.45; P = .95) (Appendix Table).
DISCUSSION
In this multicenter observational study of children hospitalized with bronchiolitis not concurrently receiving supplemental oxygen, units that only occasionally cared for bronchiolitis patients appeared to be more likely to overuse continuous pulse oximetry during bronchiolitis hospitalizations.
This finding was not immediately apparent when examining the raw data because of wide hospital-level variation in continuous pulse oximetry monitoring use. However, when the high degree of hospital-level variation in baseline overuse was accounted for with use of a random intercept for each hospital in the mixed-effects model, units that cared for higher proportions of bronchiolitis patients had significantly lower odds of continuous pulse oximetry monitoring use compared to units that cared for these infants infrequently.
As many institutions have subspecialized units to cultivate nursing expertise for care of certain diseases and patient populations, we hypothesized that units caring primarily for children on general pediatrics units would also have lower rates of monitoring overuse compared to mixed units. Interestingly, these units did not perform better, likely because potential cultural factors that might contribute to differences in monitoring are accounted for by bronchiolitis admission burden.
Our findings build on prior literature by demonstrating that unit-level, as well as hospital-level, factors appear to drive overuse in healthcare. A prior single-site retrospective cohort study demonstrated an association between higher prevalence of inpatient bronchiolitis and decreased use of unnecessary interventions such as laboratory and radiographic testing, as well as steroid and antibiotic administration.11 Although study of the relationship between volume and quality is not new to healthcare, to our knowledge, this study is the first to examine the relationship between pulse oximetry overuse in bronchiolitis and unit-level factors like admission burden and subspecialty composition.
There are several limitations. First, because the study population included only children not receiving supplemental oxygen, both the parent study and this substudy assumed that all observed use of pulse oximetry monitoring was overuse. In some cases, however, there may have been other compelling clinical reasons, institutional policies, or differences in pulse oximetry availability that were not captured during data collection or in our adjusted model. Second, hospitals used convenience sampling. It is possible this resulted in samples that were not representative of each unit’s underlying patient population or monitoring practice. In addition, not all of the 32 eligible sites were able to provide data related to hospital admissions at the unit level and thus are not included in our analysis. This remains a potential source of hospital-level selection bias.
CONCLUSION
These findings demonstrate that high bronchiolitis admission burden correlates with lower rates of unnecessary pulse oximetry monitoring in bronchiolitis. We speculate that these outcomes might reflect differing degrees of nursing comfort, expertise, and unit-level norms in caring for bronchiolitis patients, although our study was not designed to establish underlying causes. Identification of operating principles that underpin low pulse oximetry monitoring on high-burden units will provide guidance for decreasing unnecessary monitoring and will inform future studies seeking ways to discourage continuous pulse oximetry monitoring in low-risk infants. Given the institutional variation in monitoring rates, future studies examining both institution-wide and unit-level interventions will be necessary to decrease unnecessary pulse oximetry monitoring in bronchiolitis. Furthermore, these findings may be relevant to studying care quality in other disease processes, with bronchiolitis serving as a model illness for overuse.
Acknowledgments
The authors acknowledge the National Heart, Lung, and Blood Institute of the National Institutes of Health scientists who contributed their expertise to this project as part of the U01 Cooperative Agreement funding mechanism as federal employees conducting their official job duties: Lora Reineck, MD, MS, Karen Bienstock, MS, and Cheryl Boyce, PhD. The authors thank the executive council of the Pediatric Research in Inpatient Settings Network for their contributions to the early scientific development of this project. The network assessed a Collaborative Support Fee for access to the hospitals and support of this project.
The authors thank the PRIS Network collaborators for their major contributions to data collection (see Appendix).
1. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
2. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: Five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. https://doi.org/10.1002/jhm.2064
3. Quinonez RA, Coon ER, Schroeder AR, Moyer VA. When technology creates uncertainty: pulse oximetry and overdiagnosis of hypoxaemia in bronchiolitis. BMJ. 2017;358:j3850. https://doi.org/10.1136/bmj.j3850
4. Cunningham S, Rodriguez A, Adams T, et al; Bronchiolitis of Infancy Discharge Study (BIDS) group. Oxygen saturation targets in infants with bronchiolitis (BIDS): a double-blind, randomised, equivalence trial. Lancet. 2015;386(9998):1041-1048. https://doi.org/10.1016/s0140-6736(15)00163-4
5. Schroeder AR, Marmor AK, Pantell RH, Newman TB. Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527-530. https://doi.org/10.1001/archpedi.158.6.527
6. Cunningham S, McMurray A. Observational study of two oxygen saturation targets for discharge in bronchiolitis. Arch Dis Child. 2012;97(4):361-363. https://doi.org/10.1136/adc.2010.205211
7. Cunningham S, Rodriguez A, Boyd KA, McIntosh E, Lewis SC; BIDS Collaborators Group. Bronchiolitis of Infancy Discharge Study (BIDS): a multicentre, parallel-group, double-blind, randomised controlled, equivalence trial with economic evaluation. Health Technol Assess. 2015;19(71):i-172. https://doi.org/10.3310/hta19710
8. Bonafide CP, Lin R, Zander M, et al. Association between exposure to nonactionable physiologic monitor alarms and response time in a children’s hospital. J Hosp Med. 2015;10(6):345-351. https://doi.org/10.1002/jhm.2331
9. Ralston SL, Garber MD, Rice-Conboy E, et al. A multicenter collaborative to reduce unnecessary care in inpatient bronchiolitis. Pediatrics. 2016;137(1):e20150851. https://doi.org/10.1542/peds.2015-0851
10. Bonafide CP, Xiao R, Brady PW, et al; for the Pediatric Research in Inpatient Settings (PRIS) Network. Prevalence of continuous pulse oximetry monitoring in hospitalized children with bronchiolitis not requiring supplemental oxygen. JAMA. 2020;323(15):1467-1477. https://doi.org/10.1001/jama.2020.2998
11. Van Cleve WC, Christakis DA. Unnecessary care for bronchiolitis decreases with increasing inpatient prevalence of bronchiolitis. Pediatrics. 2011;128(5):e1106-e1112. https://doi.org/10.1542/peds.2011-0655
12. Rasooly IR, Beidas RS, Wolk CB, et al. Measuring overuse of continuous pulse oximetry in bronchiolitis and developing strategies for large-scale deimplementation: study protocol for a feasibility trial. Pilot Feasibility Stud. 2019;5(1):68. https://doi.org/10.1186/s40814-019-0453-2
Continuous pulse oximetry monitoring in children with bronchiolitis who don’t require supplemental oxygen is discouraged by practice guidelines and is recognized as a form of medical overuse.1-3 This practice can be associated with negative outcomes, including prolonged length of stay,4-6 increased cost of hospitalization,7 and alarm fatigue among nurses.8 Despite initiatives to reduce continuous pulse oximetry monitoring in stable patients with bronchiolitis,1,2 wide practice variation exists between hospitals.9,10 Previous studies have shown that higher prevalence of inpatient bronchiolitis admissions is associated with decreased utilization of unnecessary interventions.11 However, the relationship between pulse oximetry use and bronchiolitis prevalence has not been studied. The objective of this study is to test the hypothesis that hospital units with lower proportions of patients admitted for bronchiolitis and those with fewer general pediatrics patients relative to subspecialty patients would have higher rates of pulse oximetry overuse.
METHODS
Study Design
We conducted a substudy of the Pediatric Research in Inpatient Settings (PRIS) Network’s Eliminating Monitoring Overuse (EMO) pulse oximetry study,10,12 a 56-hospital cross-sectional study that used direct observation to measure the prevalence of continuous pulse oximetry monitoring in hospitalized infants with bronchiolitis who did not require supplemental oxygen between December 1, 2018, through March 31, 2019. This substudy was not included as part of the original aims of the project and was proposed as a separate analysis during data collection. For US sites, the Institutional Review Board (IRB) at Children’s Hospital of Philadelphia approved the study and served as the central IRB. The Research Ethics Board at University of Calgary also approved the study.
Site Selection
Hospitals with at least 60 observations were eligible for inclusion. Of the 32 hospitals that conducted the minimum observations, 25 agreed to participate (21 free-standing children’s hospitals, 3 children’s hospitals within general hospitals, and 1 community hospital).
Patient Population
The parent study included patients aged 8 weeks through 23 months with a primary diagnosis of bronchiolitis. Patients were included only if they were not receiving supplemental oxygen or nasal cannula flow at the time of data collection. The inclusion and exclusion criteria were for both the parent study and the substudy. Further inclusion and exclusion criteria have been described previously.10,12
Data Collection
In order to ascertain continuous pulse oximetry monitoring status, staff at each hospital performed observational rounds by walking to the bedside of each patient who met inclusion criteria. Additional methodology for the parent study has been published elsewhere.10,12
Bronchiolitis Admission Volume by Unit
Collaborators at each hospital gathered bronchiolitis census data from each unit that admitted patients with bronchiolitis. Units were identified prior to data collection and were characterized at the institution level based on previous local definitions. Each site was responsible for using institution-specific data collection methods for determining bronchiolitis and total admissions on each unit (eg, departmental reports or directly querying admissions data using International Classification of Diseases, Tenth Revision, diagnosis codes for bronchiolitis) over the same period as the parent study. Following data analysis, bronchiolitis admission burden was classified into five categories, based on less than 10%, 10% to less than 20%, 20% to less than 30%, 30% to less than 40%, or 40% or more of total admissions having a primary discharge diagnosis of bronchiolitis during the study period. This categorization allowed investigators to determine whether there was a dose-dependent response among categories.
Unit Composition
Site investigators also completed a survey identifying which patients were admitted to each unit (eg, general pediatrics only, medical subspecialty, surgical). Based on these results, units were further classified into seven types (Appendix Table). For the final analysis, units caring exclusively for general pediatrics patients were compared to all other unit types.
Analysis
Bronchiolitis admission burden and unit composition data were combined with observations of pulse oximetry monitoring use of patients not requiring supplemental oxygen from the parent study. We determined unadjusted observed monitoring proportions for each unit’s bronchiolitis admission burden category across all 25 hospitals. This was calculated as a simple proportion of the total number of observations during which patients were continuously monitored divided by the total number of observations performed within each unit’s admission category. We then calculated unadjusted odds ratios using the 40% and higher bronchiolitis admission burden category as a reference. We calculated similar proportions and odds ratios for the dichotomous unit composition variable. Next, we used mixed-effects logistic regression with a random intercept for each hospital to allow for differences in baseline monitoring rates, which varied widely between hospitals (2% to 92%),10 to calculate adjusted odds ratios for the unit’s admission category and unit’s composition. We also adjusted for the same covariates used in the primary study’s analysis (Table).10

RESULTS
We analyzed 2,366 observations of bronchiolitis patients from 25 hospitals. Most observations were concentrated in freestanding children’s hospitals (89%), and 50% were from hospitals with more than 250 pediatric beds. Observations were well distributed among the five categories of admit burden (Table).
In unadjusted regression, the relationship between admission burden and rate of pulse oximetry use did not appear to be dose-dependent, and 95% CIs were wide. We then analyzed the data accounting for baseline differences in hospital monitoring rates and adjusted for the covariates significantly associated with continuous pulse oximetry monitoring in the primary study’s analysis with use of a mixed-effects model. As shown in the Table, low-burden units in which bronchiolitis constituted less than 10% of total admissions had a 2.16-fold increased odds of unnecessary pulse oximetry monitoring compared to high-burden units in which bronchiolitis constituted 40% or more of total admissions (95% CI, 1.27-3.69; P = .01).
In examining the subspecialty unit composition, 596 observations (25.2%) were conducted on units exclusively caring for general pediatrics patients. In the mixed-effects model adjusted for bronchiolitis admission burden and the covariates used in the study’s primary analysis, units exclusively caring for general pediatrics patients did not have significantly different independent odds of pulse oximetry monitoring use compared to units with a mixed patient population (OR 1.01; 95% CI, 0.71-1.45; P = .95) (Appendix Table).
DISCUSSION
In this multicenter observational study of children hospitalized with bronchiolitis not concurrently receiving supplemental oxygen, units that only occasionally cared for bronchiolitis patients appeared to be more likely to overuse continuous pulse oximetry during bronchiolitis hospitalizations.
This finding was not immediately apparent when examining the raw data because of wide hospital-level variation in continuous pulse oximetry monitoring use. However, when the high degree of hospital-level variation in baseline overuse was accounted for with use of a random intercept for each hospital in the mixed-effects model, units that cared for higher proportions of bronchiolitis patients had significantly lower odds of continuous pulse oximetry monitoring use compared to units that cared for these infants infrequently.
As many institutions have subspecialized units to cultivate nursing expertise for care of certain diseases and patient populations, we hypothesized that units caring primarily for children on general pediatrics units would also have lower rates of monitoring overuse compared to mixed units. Interestingly, these units did not perform better, likely because potential cultural factors that might contribute to differences in monitoring are accounted for by bronchiolitis admission burden.
Our findings build on prior literature by demonstrating that unit-level, as well as hospital-level, factors appear to drive overuse in healthcare. A prior single-site retrospective cohort study demonstrated an association between higher prevalence of inpatient bronchiolitis and decreased use of unnecessary interventions such as laboratory and radiographic testing, as well as steroid and antibiotic administration.11 Although study of the relationship between volume and quality is not new to healthcare, to our knowledge, this study is the first to examine the relationship between pulse oximetry overuse in bronchiolitis and unit-level factors like admission burden and subspecialty composition.
There are several limitations. First, because the study population included only children not receiving supplemental oxygen, both the parent study and this substudy assumed that all observed use of pulse oximetry monitoring was overuse. In some cases, however, there may have been other compelling clinical reasons, institutional policies, or differences in pulse oximetry availability that were not captured during data collection or in our adjusted model. Second, hospitals used convenience sampling. It is possible this resulted in samples that were not representative of each unit’s underlying patient population or monitoring practice. In addition, not all of the 32 eligible sites were able to provide data related to hospital admissions at the unit level and thus are not included in our analysis. This remains a potential source of hospital-level selection bias.
CONCLUSION
These findings demonstrate that high bronchiolitis admission burden correlates with lower rates of unnecessary pulse oximetry monitoring in bronchiolitis. We speculate that these outcomes might reflect differing degrees of nursing comfort, expertise, and unit-level norms in caring for bronchiolitis patients, although our study was not designed to establish underlying causes. Identification of operating principles that underpin low pulse oximetry monitoring on high-burden units will provide guidance for decreasing unnecessary monitoring and will inform future studies seeking ways to discourage continuous pulse oximetry monitoring in low-risk infants. Given the institutional variation in monitoring rates, future studies examining both institution-wide and unit-level interventions will be necessary to decrease unnecessary pulse oximetry monitoring in bronchiolitis. Furthermore, these findings may be relevant to studying care quality in other disease processes, with bronchiolitis serving as a model illness for overuse.
Acknowledgments
The authors acknowledge the National Heart, Lung, and Blood Institute of the National Institutes of Health scientists who contributed their expertise to this project as part of the U01 Cooperative Agreement funding mechanism as federal employees conducting their official job duties: Lora Reineck, MD, MS, Karen Bienstock, MS, and Cheryl Boyce, PhD. The authors thank the executive council of the Pediatric Research in Inpatient Settings Network for their contributions to the early scientific development of this project. The network assessed a Collaborative Support Fee for access to the hospitals and support of this project.
The authors thank the PRIS Network collaborators for their major contributions to data collection (see Appendix).
Continuous pulse oximetry monitoring in children with bronchiolitis who don’t require supplemental oxygen is discouraged by practice guidelines and is recognized as a form of medical overuse.1-3 This practice can be associated with negative outcomes, including prolonged length of stay,4-6 increased cost of hospitalization,7 and alarm fatigue among nurses.8 Despite initiatives to reduce continuous pulse oximetry monitoring in stable patients with bronchiolitis,1,2 wide practice variation exists between hospitals.9,10 Previous studies have shown that higher prevalence of inpatient bronchiolitis admissions is associated with decreased utilization of unnecessary interventions.11 However, the relationship between pulse oximetry use and bronchiolitis prevalence has not been studied. The objective of this study is to test the hypothesis that hospital units with lower proportions of patients admitted for bronchiolitis and those with fewer general pediatrics patients relative to subspecialty patients would have higher rates of pulse oximetry overuse.
METHODS
Study Design
We conducted a substudy of the Pediatric Research in Inpatient Settings (PRIS) Network’s Eliminating Monitoring Overuse (EMO) pulse oximetry study,10,12 a 56-hospital cross-sectional study that used direct observation to measure the prevalence of continuous pulse oximetry monitoring in hospitalized infants with bronchiolitis who did not require supplemental oxygen between December 1, 2018, through March 31, 2019. This substudy was not included as part of the original aims of the project and was proposed as a separate analysis during data collection. For US sites, the Institutional Review Board (IRB) at Children’s Hospital of Philadelphia approved the study and served as the central IRB. The Research Ethics Board at University of Calgary also approved the study.
Site Selection
Hospitals with at least 60 observations were eligible for inclusion. Of the 32 hospitals that conducted the minimum observations, 25 agreed to participate (21 free-standing children’s hospitals, 3 children’s hospitals within general hospitals, and 1 community hospital).
Patient Population
The parent study included patients aged 8 weeks through 23 months with a primary diagnosis of bronchiolitis. Patients were included only if they were not receiving supplemental oxygen or nasal cannula flow at the time of data collection. The inclusion and exclusion criteria were for both the parent study and the substudy. Further inclusion and exclusion criteria have been described previously.10,12
Data Collection
In order to ascertain continuous pulse oximetry monitoring status, staff at each hospital performed observational rounds by walking to the bedside of each patient who met inclusion criteria. Additional methodology for the parent study has been published elsewhere.10,12
Bronchiolitis Admission Volume by Unit
Collaborators at each hospital gathered bronchiolitis census data from each unit that admitted patients with bronchiolitis. Units were identified prior to data collection and were characterized at the institution level based on previous local definitions. Each site was responsible for using institution-specific data collection methods for determining bronchiolitis and total admissions on each unit (eg, departmental reports or directly querying admissions data using International Classification of Diseases, Tenth Revision, diagnosis codes for bronchiolitis) over the same period as the parent study. Following data analysis, bronchiolitis admission burden was classified into five categories, based on less than 10%, 10% to less than 20%, 20% to less than 30%, 30% to less than 40%, or 40% or more of total admissions having a primary discharge diagnosis of bronchiolitis during the study period. This categorization allowed investigators to determine whether there was a dose-dependent response among categories.
Unit Composition
Site investigators also completed a survey identifying which patients were admitted to each unit (eg, general pediatrics only, medical subspecialty, surgical). Based on these results, units were further classified into seven types (Appendix Table). For the final analysis, units caring exclusively for general pediatrics patients were compared to all other unit types.
Analysis
Bronchiolitis admission burden and unit composition data were combined with observations of pulse oximetry monitoring use of patients not requiring supplemental oxygen from the parent study. We determined unadjusted observed monitoring proportions for each unit’s bronchiolitis admission burden category across all 25 hospitals. This was calculated as a simple proportion of the total number of observations during which patients were continuously monitored divided by the total number of observations performed within each unit’s admission category. We then calculated unadjusted odds ratios using the 40% and higher bronchiolitis admission burden category as a reference. We calculated similar proportions and odds ratios for the dichotomous unit composition variable. Next, we used mixed-effects logistic regression with a random intercept for each hospital to allow for differences in baseline monitoring rates, which varied widely between hospitals (2% to 92%),10 to calculate adjusted odds ratios for the unit’s admission category and unit’s composition. We also adjusted for the same covariates used in the primary study’s analysis (Table).10

RESULTS
We analyzed 2,366 observations of bronchiolitis patients from 25 hospitals. Most observations were concentrated in freestanding children’s hospitals (89%), and 50% were from hospitals with more than 250 pediatric beds. Observations were well distributed among the five categories of admit burden (Table).
In unadjusted regression, the relationship between admission burden and rate of pulse oximetry use did not appear to be dose-dependent, and 95% CIs were wide. We then analyzed the data accounting for baseline differences in hospital monitoring rates and adjusted for the covariates significantly associated with continuous pulse oximetry monitoring in the primary study’s analysis with use of a mixed-effects model. As shown in the Table, low-burden units in which bronchiolitis constituted less than 10% of total admissions had a 2.16-fold increased odds of unnecessary pulse oximetry monitoring compared to high-burden units in which bronchiolitis constituted 40% or more of total admissions (95% CI, 1.27-3.69; P = .01).
In examining the subspecialty unit composition, 596 observations (25.2%) were conducted on units exclusively caring for general pediatrics patients. In the mixed-effects model adjusted for bronchiolitis admission burden and the covariates used in the study’s primary analysis, units exclusively caring for general pediatrics patients did not have significantly different independent odds of pulse oximetry monitoring use compared to units with a mixed patient population (OR 1.01; 95% CI, 0.71-1.45; P = .95) (Appendix Table).
DISCUSSION
In this multicenter observational study of children hospitalized with bronchiolitis not concurrently receiving supplemental oxygen, units that only occasionally cared for bronchiolitis patients appeared to be more likely to overuse continuous pulse oximetry during bronchiolitis hospitalizations.
This finding was not immediately apparent when examining the raw data because of wide hospital-level variation in continuous pulse oximetry monitoring use. However, when the high degree of hospital-level variation in baseline overuse was accounted for with use of a random intercept for each hospital in the mixed-effects model, units that cared for higher proportions of bronchiolitis patients had significantly lower odds of continuous pulse oximetry monitoring use compared to units that cared for these infants infrequently.
As many institutions have subspecialized units to cultivate nursing expertise for care of certain diseases and patient populations, we hypothesized that units caring primarily for children on general pediatrics units would also have lower rates of monitoring overuse compared to mixed units. Interestingly, these units did not perform better, likely because potential cultural factors that might contribute to differences in monitoring are accounted for by bronchiolitis admission burden.
Our findings build on prior literature by demonstrating that unit-level, as well as hospital-level, factors appear to drive overuse in healthcare. A prior single-site retrospective cohort study demonstrated an association between higher prevalence of inpatient bronchiolitis and decreased use of unnecessary interventions such as laboratory and radiographic testing, as well as steroid and antibiotic administration.11 Although study of the relationship between volume and quality is not new to healthcare, to our knowledge, this study is the first to examine the relationship between pulse oximetry overuse in bronchiolitis and unit-level factors like admission burden and subspecialty composition.
There are several limitations. First, because the study population included only children not receiving supplemental oxygen, both the parent study and this substudy assumed that all observed use of pulse oximetry monitoring was overuse. In some cases, however, there may have been other compelling clinical reasons, institutional policies, or differences in pulse oximetry availability that were not captured during data collection or in our adjusted model. Second, hospitals used convenience sampling. It is possible this resulted in samples that were not representative of each unit’s underlying patient population or monitoring practice. In addition, not all of the 32 eligible sites were able to provide data related to hospital admissions at the unit level and thus are not included in our analysis. This remains a potential source of hospital-level selection bias.
CONCLUSION
These findings demonstrate that high bronchiolitis admission burden correlates with lower rates of unnecessary pulse oximetry monitoring in bronchiolitis. We speculate that these outcomes might reflect differing degrees of nursing comfort, expertise, and unit-level norms in caring for bronchiolitis patients, although our study was not designed to establish underlying causes. Identification of operating principles that underpin low pulse oximetry monitoring on high-burden units will provide guidance for decreasing unnecessary monitoring and will inform future studies seeking ways to discourage continuous pulse oximetry monitoring in low-risk infants. Given the institutional variation in monitoring rates, future studies examining both institution-wide and unit-level interventions will be necessary to decrease unnecessary pulse oximetry monitoring in bronchiolitis. Furthermore, these findings may be relevant to studying care quality in other disease processes, with bronchiolitis serving as a model illness for overuse.
Acknowledgments
The authors acknowledge the National Heart, Lung, and Blood Institute of the National Institutes of Health scientists who contributed their expertise to this project as part of the U01 Cooperative Agreement funding mechanism as federal employees conducting their official job duties: Lora Reineck, MD, MS, Karen Bienstock, MS, and Cheryl Boyce, PhD. The authors thank the executive council of the Pediatric Research in Inpatient Settings Network for their contributions to the early scientific development of this project. The network assessed a Collaborative Support Fee for access to the hospitals and support of this project.
The authors thank the PRIS Network collaborators for their major contributions to data collection (see Appendix).
1. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
2. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: Five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. https://doi.org/10.1002/jhm.2064
3. Quinonez RA, Coon ER, Schroeder AR, Moyer VA. When technology creates uncertainty: pulse oximetry and overdiagnosis of hypoxaemia in bronchiolitis. BMJ. 2017;358:j3850. https://doi.org/10.1136/bmj.j3850
4. Cunningham S, Rodriguez A, Adams T, et al; Bronchiolitis of Infancy Discharge Study (BIDS) group. Oxygen saturation targets in infants with bronchiolitis (BIDS): a double-blind, randomised, equivalence trial. Lancet. 2015;386(9998):1041-1048. https://doi.org/10.1016/s0140-6736(15)00163-4
5. Schroeder AR, Marmor AK, Pantell RH, Newman TB. Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527-530. https://doi.org/10.1001/archpedi.158.6.527
6. Cunningham S, McMurray A. Observational study of two oxygen saturation targets for discharge in bronchiolitis. Arch Dis Child. 2012;97(4):361-363. https://doi.org/10.1136/adc.2010.205211
7. Cunningham S, Rodriguez A, Boyd KA, McIntosh E, Lewis SC; BIDS Collaborators Group. Bronchiolitis of Infancy Discharge Study (BIDS): a multicentre, parallel-group, double-blind, randomised controlled, equivalence trial with economic evaluation. Health Technol Assess. 2015;19(71):i-172. https://doi.org/10.3310/hta19710
8. Bonafide CP, Lin R, Zander M, et al. Association between exposure to nonactionable physiologic monitor alarms and response time in a children’s hospital. J Hosp Med. 2015;10(6):345-351. https://doi.org/10.1002/jhm.2331
9. Ralston SL, Garber MD, Rice-Conboy E, et al. A multicenter collaborative to reduce unnecessary care in inpatient bronchiolitis. Pediatrics. 2016;137(1):e20150851. https://doi.org/10.1542/peds.2015-0851
10. Bonafide CP, Xiao R, Brady PW, et al; for the Pediatric Research in Inpatient Settings (PRIS) Network. Prevalence of continuous pulse oximetry monitoring in hospitalized children with bronchiolitis not requiring supplemental oxygen. JAMA. 2020;323(15):1467-1477. https://doi.org/10.1001/jama.2020.2998
11. Van Cleve WC, Christakis DA. Unnecessary care for bronchiolitis decreases with increasing inpatient prevalence of bronchiolitis. Pediatrics. 2011;128(5):e1106-e1112. https://doi.org/10.1542/peds.2011-0655
12. Rasooly IR, Beidas RS, Wolk CB, et al. Measuring overuse of continuous pulse oximetry in bronchiolitis and developing strategies for large-scale deimplementation: study protocol for a feasibility trial. Pilot Feasibility Stud. 2019;5(1):68. https://doi.org/10.1186/s40814-019-0453-2
1. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
2. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: Five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. https://doi.org/10.1002/jhm.2064
3. Quinonez RA, Coon ER, Schroeder AR, Moyer VA. When technology creates uncertainty: pulse oximetry and overdiagnosis of hypoxaemia in bronchiolitis. BMJ. 2017;358:j3850. https://doi.org/10.1136/bmj.j3850
4. Cunningham S, Rodriguez A, Adams T, et al; Bronchiolitis of Infancy Discharge Study (BIDS) group. Oxygen saturation targets in infants with bronchiolitis (BIDS): a double-blind, randomised, equivalence trial. Lancet. 2015;386(9998):1041-1048. https://doi.org/10.1016/s0140-6736(15)00163-4
5. Schroeder AR, Marmor AK, Pantell RH, Newman TB. Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527-530. https://doi.org/10.1001/archpedi.158.6.527
6. Cunningham S, McMurray A. Observational study of two oxygen saturation targets for discharge in bronchiolitis. Arch Dis Child. 2012;97(4):361-363. https://doi.org/10.1136/adc.2010.205211
7. Cunningham S, Rodriguez A, Boyd KA, McIntosh E, Lewis SC; BIDS Collaborators Group. Bronchiolitis of Infancy Discharge Study (BIDS): a multicentre, parallel-group, double-blind, randomised controlled, equivalence trial with economic evaluation. Health Technol Assess. 2015;19(71):i-172. https://doi.org/10.3310/hta19710
8. Bonafide CP, Lin R, Zander M, et al. Association between exposure to nonactionable physiologic monitor alarms and response time in a children’s hospital. J Hosp Med. 2015;10(6):345-351. https://doi.org/10.1002/jhm.2331
9. Ralston SL, Garber MD, Rice-Conboy E, et al. A multicenter collaborative to reduce unnecessary care in inpatient bronchiolitis. Pediatrics. 2016;137(1):e20150851. https://doi.org/10.1542/peds.2015-0851
10. Bonafide CP, Xiao R, Brady PW, et al; for the Pediatric Research in Inpatient Settings (PRIS) Network. Prevalence of continuous pulse oximetry monitoring in hospitalized children with bronchiolitis not requiring supplemental oxygen. JAMA. 2020;323(15):1467-1477. https://doi.org/10.1001/jama.2020.2998
11. Van Cleve WC, Christakis DA. Unnecessary care for bronchiolitis decreases with increasing inpatient prevalence of bronchiolitis. Pediatrics. 2011;128(5):e1106-e1112. https://doi.org/10.1542/peds.2011-0655
12. Rasooly IR, Beidas RS, Wolk CB, et al. Measuring overuse of continuous pulse oximetry in bronchiolitis and developing strategies for large-scale deimplementation: study protocol for a feasibility trial. Pilot Feasibility Stud. 2019;5(1):68. https://doi.org/10.1186/s40814-019-0453-2
© 2020 Society of Hospital Medicine
Validity of Continuous Pulse Oximetry Orders for Identification of Actual Monitoring Status in Bronchiolitis
As part of improvement collaboratives that aimed to reduce overuse of continuous pulse oximetry in children hospitalized with bronchiolitis, researchers used the presence of an active order for it as a proxy for the actual use of such monitoring.1,2 With use of this proxy, investigators on a national study documented a high burden of continuous oximetry overuse (86.5% before quality improvement interventions and 45.5% after),1 but the validity of orders in representing actual monitoring practice is unknown. If the presence of an active pulse oximetry order accurately identifies infants on monitors, electronic health record data could inform epidemiologic estimates of monitoring overuse and measure the success of quality improvement and deimplementation interventions. Alternatively, if nurses commonly begin and/or discontinue pulse oximetry without updated orders, a pulse oximetry order would not be an accurate proxy, and additional data capture methods (eg, bedside observation or data capture from bedside monitors) would be needed.
Understanding the validity of orders for detection of actual use is critical because continuous pulse oximetry monitoring is considered an overused practice in pediatric acute viral bronchiolitis,3 and national guidelines recommend against its use in low-risk hospitalized children.4,5 Continuous monitoring may identify trivial, self-resolving oxygen desaturation and its use is not associated with improved outcomes.6-9 When self-resolving desaturations are treated with additional supplemental oxygen, hospital stays may be unnecessarily prolonged.10 In order to reduce unnecessary continuous pulse oximetry use, measurement of the extent of the overused practice is necessary. In this 56-hospital study,11 we aimed to determine the validity of using active continuous pulse oximetry orders instead of bedside observation of actual monitor use.
METHODS
Design
In this multicenter, repeated cross-sectional study, investigators used direct bedside observation to determine continuous pulse oximetry monitor use and then assessed whether an active continuous monitoring order was present in the electronic health record. The study took place during one bronchiolitis season, December 1, 2018, through March 31, 2019.
Setting and Patients
Investigators at 56 freestanding children’s hospitals, children’s hospitals within general hospitals, and community hospitals in the Pediatric Research in Inpatient Settings (PRIS) Network collected data on infants aged 8 weeks to 23 months who were hospitalized with bronchiolitis. As this work was a substudy of the larger Eliminating Monitor Overuse study, only infants not currently receiving supplemental oxygen were included.11 Investigators observed eligible infants outside of the intensive care unit on general hospital medicine units. We excluded infants born premature (documented prematurity of <28 weeks’ gestation or documented “premature” without a gestational age listed), as well as those with a home oxygen requirement, cyanotic congenital heart disease, pulmonary hypertension, tracheostomy, primary neuromuscular disease, immunodeficiency, or cancer.
Data Collection
Investigators used the electronic health record to identify eligible infants. Investigators entered patient rooms to confirm the infant was not on supplemental oxygen (hence confirming eligibility for the study) and determine if continuous pulse oximetry was actively in use by examining the monitor display for a pulse oximetry waveform. Investigators then confirmed if active orders for pulse oximetry were present in the patient’s chart. Per study design, site investigators aimed to observe approximately half of eligible infants during the day (10
Analysis
We excluded patients with conditional orders (eg, monitored only when certain conditions exist, such as when asleep) because of the time-varying and wide range of conditions that could be specified. Furthermore, conditional orders would not be useful as proxies to measure oximetry use because investigators would still need additional data (eg, bedside observation) to determine current monitoring status.
We calculated the sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of active orders using the reference standard of direct bedside observation, as well as corresponding 95% CIs that accounted for within-hospital clustering. We calculated these test characteristics overall and as stratified across four age groups: 8 weeks to 5 months, 6 months to 11 months, 12 months to 17 months, and 18 months to 23 months. We also calculated the test characteristics for each hospital. We decided a priori that a PPV and NPV of 80% would represent a reasonable threshold to use active orders as a proxy in multicenter research. For hospital-level analyses we included only hospitals with 60 or more total observations and more than 15 observations with active orders for PPV and more than 15 observations without active orders for NPV. We used Stata (StataCorp LLC, College Station, Texas) version 15.1 for analysis.
For US sites, the Institutional Review Board (IRB) at Children’s Hospital of Philadelphia approved the study as the single reviewing IRB, and the remaining US sites established reliance agreements with the reviewing IRB. Research Ethics Boards at the Canadian sites (University of Calgary and The Hospital for Sick Children) also reviewed and approved the study. All sites granted waivers of consent, assent, parental permission, and HIPAA authorization.
RESULTS
Investigators completed 3,612 observations in 56 hospitals. This included 33 freestanding children’s hospitals, 14 hospitals within large general hospitals, and 9 community hospitals. Of 3,612 completed observations, on 631 occasions (17%) patients had conditional orders (eg, continuous monitoring only when sleeping) and were excluded from further analysis.
Most pulse oximetry–monitored infants did not have an active monitoring order (670 out of 1,309; sensitivity of 49%). Test characteristics, stratified by age group, are presented in the Table. Across all observations, the overall PPV was 77% (95% CI, 72-82), and the overall NPV was 69% (95% CI, 61-77). Variation of all test characteristics across age group was small (eg, the sensitivity ranged from 43% to 51%).
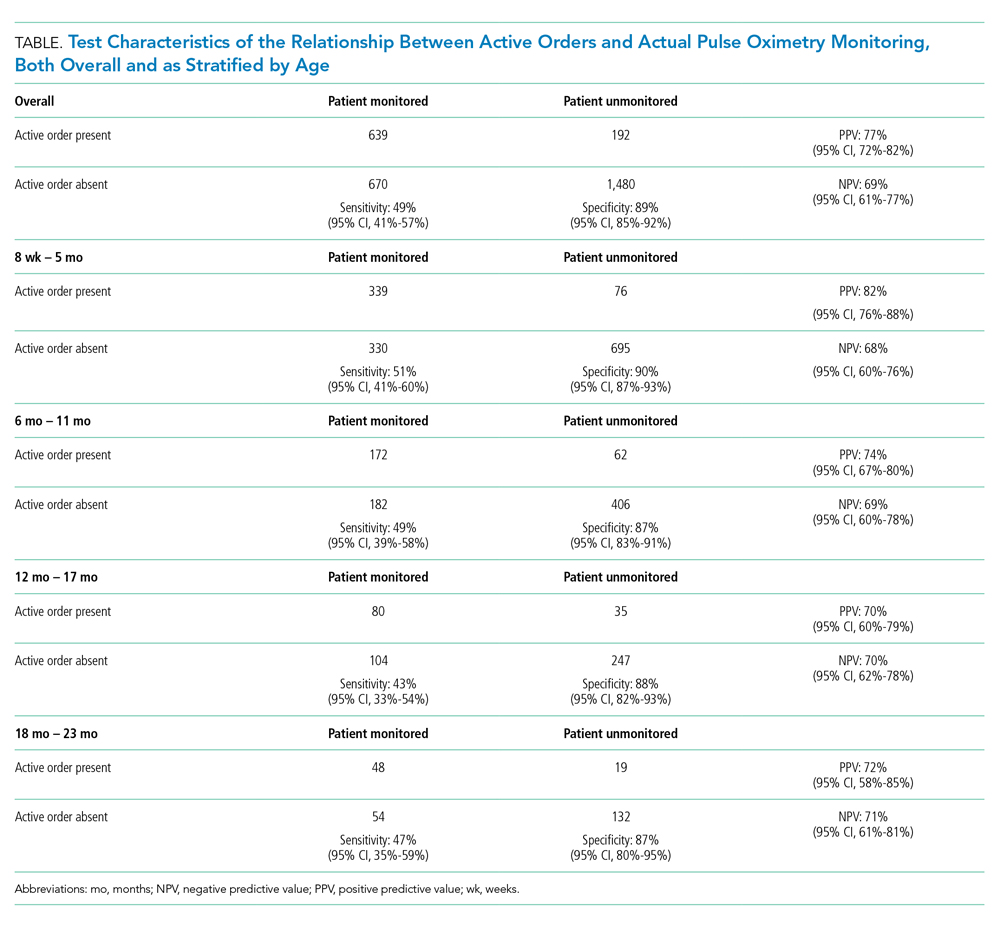
With inclusion of only those hospitals with sufficient observations, hospital-level variation in the PPV and NPV of using active orders was substantial (PPV range of 48% to 96% and NPV range of 30% to 98%). Only two hospitals had both a PPV and NPV for using monitor orders that exceeded the 80% threshold.
DISCUSSION
Active continuous pulse oximetry orders did not accurately represent actual monitoring status in this study. Monitoring orders alone frequently misrepresent true monitoring status and, as such, should be interpreted with caution in research or quality improvement activities. If more valid estimates of monitoring use and overuse are needed, potential measurement options include direct observation, as used in our study, as well as the use of more complex data streams such as the output of monitoring devices or pulse oximetry data in the electronic health record. In only two of the hospitals, using active continuous monitoring orders was a reasonable proxy for detecting actual monitor use. Monitoring orders could potentially be validly used for deimplementation efforts at those centers; other hospitals could consider targeted improvement efforts (eg, morning huddles examining the discordance between monitoring orders and monitoring status) to improve the accuracy of using continuous pulse oximetry orders.
We acknowledge several limitations of this study. Site investigators employed a convenience sampling approach, so it is possible that some investigators observed sicker or less sick infants. Although the PRIS network includes a geographically diverse group of North American hospitals, community hospitals were underrepresented in this study. Our results hence generalize more precisely to freestanding children’s hospitals than to community hospitals. We did not observe infants currently on supplemental oxygen, so we do not know to what degree using orders is valid in that context. We did not collect data on why actual monitoring status differed from monitoring orders and hence cannot quantify to what extent different factors (eg, nurse belief that monitors are a safety net or infants inadvertently left on monitors after a spot check pulse oximetry reading) contributed to this discordance. Finally, our study only examined one electronic health record variable—the presence of an active order. It may be that other variables in the health record (eg, minute-by-minute pulse oximetry values in a vital sign flowsheet) are much better proxies of actual continuous monitor use.
CONCLUSION
Using an active order for continuous pulse oximetry has poor sensitivity, PPV, and NPV for detecting true monitoring status at the bedside. Teams intending to measure the actual use of pulse oximetry should be aware of the limitations of using active orders alone as an accurate measure of pulse oximetry monitoring.
Acknowledgments
We thank the NHLBI scientists who contributed to this project as part of the U01 Cooperative Agreement funding mechanism: Lora Reineck, MD, MS, Karen Bienstock, MS, and Cheryl Boyce, PhD.
We thank the Executive Council of the PRIS Network for their contributions to the early scientific development of this project. We thank the PRIS site investigators for their major contributions to the Eliminating Monitor Overuse (EMO) Study data collection. Each listed collaborator is a group author for the PRIS Network in this manuscript. Their names can be found in the online supplemental information.
1. Ralston SL, Garber MD, Rice-Conboy E, et al. A multicenter collaborative to reduce unnecessary care in inpatient bronchiolitis. Pediatrics. 2016;137(1). https://doi.org/10.1542/peds.2015-0851
2. Mittal S, Marlowe L, Blakeslee S, et al. Successful use of quality improvement methodology to reduce inpatient length of stay in bronchiolitis through judicious use of intermittent pulse oximetry. Hosp Pediatr. 2019;9(2):73-78. https://doi.org/10.1542/hpeds.2018-0023
3. Quinonez RA, Coon ER, Schroeder AR, Moyer VA. When technology creates uncertainty: pulse oximetry and overdiagnosis of hypoxaemia in bronchiolitis. BMJ. 2017;358:j3850. https://doi.org/10.1136/bmj.j3850
4. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. https://doi.org/10.1002/jhm.2064
5. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
6. Principi T, Coates AL, Parkin PC, Stephens D, DaSilva Z, Schuh S. Effect of oxygen desaturations on subsequent medical visits in infants discharged from the emergency department with bronchiolitis. JAMA Pediatr. 2016;170(6):602-608. https://doi.org/10.1001/jamapediatrics.2016.0114
7. Cunningham S, Rodriguez A, Adams T, et al. Oxygen saturation targets in infants with bronchiolitis (BIDS): a double-blind, randomised, equivalence trial. Lancet. 2015;386(9998):1041-1048. https://doi.org/10.1016/s0140-6736(15)00163-4
8. McCulloh R, Koster M, Ralston S, et al. Use of intermittent vs continuous pulse oximetry for nonhypoxemic infants and young children hospitalized for bronchiolitis: a randomized clinical trial. JAMA Pediatr. 2015;169(10):898-904. https://doi.org/10.1001/jamapediatrics.2015.1746
9. Schuh S, Freedman S, Coates A, et al. Effect of oximetry on hospitalization in bronchiolitis: a randomized clinical trial. JAMA. 2014;312(7):712-718. https://doi.org/10.1001/jama.2014.8637
10. Schroeder AR, Marmor AK, Pantell RH, Newman TB. Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527-530. https://doi.org/10.1001/archpedi.158.6.527
11. Rasooly IR, Beidas RS, Wolk CB, et al. Measuring overuse of continuous pulse oximetry in bronchiolitis and developing strategies for large-scale deimplementation: study protocol for a feasibility trial. Pilot Feasibility Stud. 2019;5:68. https://doi.org/10.1186/s40814-019-0453-2
As part of improvement collaboratives that aimed to reduce overuse of continuous pulse oximetry in children hospitalized with bronchiolitis, researchers used the presence of an active order for it as a proxy for the actual use of such monitoring.1,2 With use of this proxy, investigators on a national study documented a high burden of continuous oximetry overuse (86.5% before quality improvement interventions and 45.5% after),1 but the validity of orders in representing actual monitoring practice is unknown. If the presence of an active pulse oximetry order accurately identifies infants on monitors, electronic health record data could inform epidemiologic estimates of monitoring overuse and measure the success of quality improvement and deimplementation interventions. Alternatively, if nurses commonly begin and/or discontinue pulse oximetry without updated orders, a pulse oximetry order would not be an accurate proxy, and additional data capture methods (eg, bedside observation or data capture from bedside monitors) would be needed.
Understanding the validity of orders for detection of actual use is critical because continuous pulse oximetry monitoring is considered an overused practice in pediatric acute viral bronchiolitis,3 and national guidelines recommend against its use in low-risk hospitalized children.4,5 Continuous monitoring may identify trivial, self-resolving oxygen desaturation and its use is not associated with improved outcomes.6-9 When self-resolving desaturations are treated with additional supplemental oxygen, hospital stays may be unnecessarily prolonged.10 In order to reduce unnecessary continuous pulse oximetry use, measurement of the extent of the overused practice is necessary. In this 56-hospital study,11 we aimed to determine the validity of using active continuous pulse oximetry orders instead of bedside observation of actual monitor use.
METHODS
Design
In this multicenter, repeated cross-sectional study, investigators used direct bedside observation to determine continuous pulse oximetry monitor use and then assessed whether an active continuous monitoring order was present in the electronic health record. The study took place during one bronchiolitis season, December 1, 2018, through March 31, 2019.
Setting and Patients
Investigators at 56 freestanding children’s hospitals, children’s hospitals within general hospitals, and community hospitals in the Pediatric Research in Inpatient Settings (PRIS) Network collected data on infants aged 8 weeks to 23 months who were hospitalized with bronchiolitis. As this work was a substudy of the larger Eliminating Monitor Overuse study, only infants not currently receiving supplemental oxygen were included.11 Investigators observed eligible infants outside of the intensive care unit on general hospital medicine units. We excluded infants born premature (documented prematurity of <28 weeks’ gestation or documented “premature” without a gestational age listed), as well as those with a home oxygen requirement, cyanotic congenital heart disease, pulmonary hypertension, tracheostomy, primary neuromuscular disease, immunodeficiency, or cancer.
Data Collection
Investigators used the electronic health record to identify eligible infants. Investigators entered patient rooms to confirm the infant was not on supplemental oxygen (hence confirming eligibility for the study) and determine if continuous pulse oximetry was actively in use by examining the monitor display for a pulse oximetry waveform. Investigators then confirmed if active orders for pulse oximetry were present in the patient’s chart. Per study design, site investigators aimed to observe approximately half of eligible infants during the day (10
Analysis
We excluded patients with conditional orders (eg, monitored only when certain conditions exist, such as when asleep) because of the time-varying and wide range of conditions that could be specified. Furthermore, conditional orders would not be useful as proxies to measure oximetry use because investigators would still need additional data (eg, bedside observation) to determine current monitoring status.
We calculated the sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of active orders using the reference standard of direct bedside observation, as well as corresponding 95% CIs that accounted for within-hospital clustering. We calculated these test characteristics overall and as stratified across four age groups: 8 weeks to 5 months, 6 months to 11 months, 12 months to 17 months, and 18 months to 23 months. We also calculated the test characteristics for each hospital. We decided a priori that a PPV and NPV of 80% would represent a reasonable threshold to use active orders as a proxy in multicenter research. For hospital-level analyses we included only hospitals with 60 or more total observations and more than 15 observations with active orders for PPV and more than 15 observations without active orders for NPV. We used Stata (StataCorp LLC, College Station, Texas) version 15.1 for analysis.
For US sites, the Institutional Review Board (IRB) at Children’s Hospital of Philadelphia approved the study as the single reviewing IRB, and the remaining US sites established reliance agreements with the reviewing IRB. Research Ethics Boards at the Canadian sites (University of Calgary and The Hospital for Sick Children) also reviewed and approved the study. All sites granted waivers of consent, assent, parental permission, and HIPAA authorization.
RESULTS
Investigators completed 3,612 observations in 56 hospitals. This included 33 freestanding children’s hospitals, 14 hospitals within large general hospitals, and 9 community hospitals. Of 3,612 completed observations, on 631 occasions (17%) patients had conditional orders (eg, continuous monitoring only when sleeping) and were excluded from further analysis.
Most pulse oximetry–monitored infants did not have an active monitoring order (670 out of 1,309; sensitivity of 49%). Test characteristics, stratified by age group, are presented in the Table. Across all observations, the overall PPV was 77% (95% CI, 72-82), and the overall NPV was 69% (95% CI, 61-77). Variation of all test characteristics across age group was small (eg, the sensitivity ranged from 43% to 51%).

With inclusion of only those hospitals with sufficient observations, hospital-level variation in the PPV and NPV of using active orders was substantial (PPV range of 48% to 96% and NPV range of 30% to 98%). Only two hospitals had both a PPV and NPV for using monitor orders that exceeded the 80% threshold.
DISCUSSION
Active continuous pulse oximetry orders did not accurately represent actual monitoring status in this study. Monitoring orders alone frequently misrepresent true monitoring status and, as such, should be interpreted with caution in research or quality improvement activities. If more valid estimates of monitoring use and overuse are needed, potential measurement options include direct observation, as used in our study, as well as the use of more complex data streams such as the output of monitoring devices or pulse oximetry data in the electronic health record. In only two of the hospitals, using active continuous monitoring orders was a reasonable proxy for detecting actual monitor use. Monitoring orders could potentially be validly used for deimplementation efforts at those centers; other hospitals could consider targeted improvement efforts (eg, morning huddles examining the discordance between monitoring orders and monitoring status) to improve the accuracy of using continuous pulse oximetry orders.
We acknowledge several limitations of this study. Site investigators employed a convenience sampling approach, so it is possible that some investigators observed sicker or less sick infants. Although the PRIS network includes a geographically diverse group of North American hospitals, community hospitals were underrepresented in this study. Our results hence generalize more precisely to freestanding children’s hospitals than to community hospitals. We did not observe infants currently on supplemental oxygen, so we do not know to what degree using orders is valid in that context. We did not collect data on why actual monitoring status differed from monitoring orders and hence cannot quantify to what extent different factors (eg, nurse belief that monitors are a safety net or infants inadvertently left on monitors after a spot check pulse oximetry reading) contributed to this discordance. Finally, our study only examined one electronic health record variable—the presence of an active order. It may be that other variables in the health record (eg, minute-by-minute pulse oximetry values in a vital sign flowsheet) are much better proxies of actual continuous monitor use.
CONCLUSION
Using an active order for continuous pulse oximetry has poor sensitivity, PPV, and NPV for detecting true monitoring status at the bedside. Teams intending to measure the actual use of pulse oximetry should be aware of the limitations of using active orders alone as an accurate measure of pulse oximetry monitoring.
Acknowledgments
We thank the NHLBI scientists who contributed to this project as part of the U01 Cooperative Agreement funding mechanism: Lora Reineck, MD, MS, Karen Bienstock, MS, and Cheryl Boyce, PhD.
We thank the Executive Council of the PRIS Network for their contributions to the early scientific development of this project. We thank the PRIS site investigators for their major contributions to the Eliminating Monitor Overuse (EMO) Study data collection. Each listed collaborator is a group author for the PRIS Network in this manuscript. Their names can be found in the online supplemental information.
As part of improvement collaboratives that aimed to reduce overuse of continuous pulse oximetry in children hospitalized with bronchiolitis, researchers used the presence of an active order for it as a proxy for the actual use of such monitoring.1,2 With use of this proxy, investigators on a national study documented a high burden of continuous oximetry overuse (86.5% before quality improvement interventions and 45.5% after),1 but the validity of orders in representing actual monitoring practice is unknown. If the presence of an active pulse oximetry order accurately identifies infants on monitors, electronic health record data could inform epidemiologic estimates of monitoring overuse and measure the success of quality improvement and deimplementation interventions. Alternatively, if nurses commonly begin and/or discontinue pulse oximetry without updated orders, a pulse oximetry order would not be an accurate proxy, and additional data capture methods (eg, bedside observation or data capture from bedside monitors) would be needed.
Understanding the validity of orders for detection of actual use is critical because continuous pulse oximetry monitoring is considered an overused practice in pediatric acute viral bronchiolitis,3 and national guidelines recommend against its use in low-risk hospitalized children.4,5 Continuous monitoring may identify trivial, self-resolving oxygen desaturation and its use is not associated with improved outcomes.6-9 When self-resolving desaturations are treated with additional supplemental oxygen, hospital stays may be unnecessarily prolonged.10 In order to reduce unnecessary continuous pulse oximetry use, measurement of the extent of the overused practice is necessary. In this 56-hospital study,11 we aimed to determine the validity of using active continuous pulse oximetry orders instead of bedside observation of actual monitor use.
METHODS
Design
In this multicenter, repeated cross-sectional study, investigators used direct bedside observation to determine continuous pulse oximetry monitor use and then assessed whether an active continuous monitoring order was present in the electronic health record. The study took place during one bronchiolitis season, December 1, 2018, through March 31, 2019.
Setting and Patients
Investigators at 56 freestanding children’s hospitals, children’s hospitals within general hospitals, and community hospitals in the Pediatric Research in Inpatient Settings (PRIS) Network collected data on infants aged 8 weeks to 23 months who were hospitalized with bronchiolitis. As this work was a substudy of the larger Eliminating Monitor Overuse study, only infants not currently receiving supplemental oxygen were included.11 Investigators observed eligible infants outside of the intensive care unit on general hospital medicine units. We excluded infants born premature (documented prematurity of <28 weeks’ gestation or documented “premature” without a gestational age listed), as well as those with a home oxygen requirement, cyanotic congenital heart disease, pulmonary hypertension, tracheostomy, primary neuromuscular disease, immunodeficiency, or cancer.
Data Collection
Investigators used the electronic health record to identify eligible infants. Investigators entered patient rooms to confirm the infant was not on supplemental oxygen (hence confirming eligibility for the study) and determine if continuous pulse oximetry was actively in use by examining the monitor display for a pulse oximetry waveform. Investigators then confirmed if active orders for pulse oximetry were present in the patient’s chart. Per study design, site investigators aimed to observe approximately half of eligible infants during the day (10
Analysis
We excluded patients with conditional orders (eg, monitored only when certain conditions exist, such as when asleep) because of the time-varying and wide range of conditions that could be specified. Furthermore, conditional orders would not be useful as proxies to measure oximetry use because investigators would still need additional data (eg, bedside observation) to determine current monitoring status.
We calculated the sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of active orders using the reference standard of direct bedside observation, as well as corresponding 95% CIs that accounted for within-hospital clustering. We calculated these test characteristics overall and as stratified across four age groups: 8 weeks to 5 months, 6 months to 11 months, 12 months to 17 months, and 18 months to 23 months. We also calculated the test characteristics for each hospital. We decided a priori that a PPV and NPV of 80% would represent a reasonable threshold to use active orders as a proxy in multicenter research. For hospital-level analyses we included only hospitals with 60 or more total observations and more than 15 observations with active orders for PPV and more than 15 observations without active orders for NPV. We used Stata (StataCorp LLC, College Station, Texas) version 15.1 for analysis.
For US sites, the Institutional Review Board (IRB) at Children’s Hospital of Philadelphia approved the study as the single reviewing IRB, and the remaining US sites established reliance agreements with the reviewing IRB. Research Ethics Boards at the Canadian sites (University of Calgary and The Hospital for Sick Children) also reviewed and approved the study. All sites granted waivers of consent, assent, parental permission, and HIPAA authorization.
RESULTS
Investigators completed 3,612 observations in 56 hospitals. This included 33 freestanding children’s hospitals, 14 hospitals within large general hospitals, and 9 community hospitals. Of 3,612 completed observations, on 631 occasions (17%) patients had conditional orders (eg, continuous monitoring only when sleeping) and were excluded from further analysis.
Most pulse oximetry–monitored infants did not have an active monitoring order (670 out of 1,309; sensitivity of 49%). Test characteristics, stratified by age group, are presented in the Table. Across all observations, the overall PPV was 77% (95% CI, 72-82), and the overall NPV was 69% (95% CI, 61-77). Variation of all test characteristics across age group was small (eg, the sensitivity ranged from 43% to 51%).

With inclusion of only those hospitals with sufficient observations, hospital-level variation in the PPV and NPV of using active orders was substantial (PPV range of 48% to 96% and NPV range of 30% to 98%). Only two hospitals had both a PPV and NPV for using monitor orders that exceeded the 80% threshold.
DISCUSSION
Active continuous pulse oximetry orders did not accurately represent actual monitoring status in this study. Monitoring orders alone frequently misrepresent true monitoring status and, as such, should be interpreted with caution in research or quality improvement activities. If more valid estimates of monitoring use and overuse are needed, potential measurement options include direct observation, as used in our study, as well as the use of more complex data streams such as the output of monitoring devices or pulse oximetry data in the electronic health record. In only two of the hospitals, using active continuous monitoring orders was a reasonable proxy for detecting actual monitor use. Monitoring orders could potentially be validly used for deimplementation efforts at those centers; other hospitals could consider targeted improvement efforts (eg, morning huddles examining the discordance between monitoring orders and monitoring status) to improve the accuracy of using continuous pulse oximetry orders.
We acknowledge several limitations of this study. Site investigators employed a convenience sampling approach, so it is possible that some investigators observed sicker or less sick infants. Although the PRIS network includes a geographically diverse group of North American hospitals, community hospitals were underrepresented in this study. Our results hence generalize more precisely to freestanding children’s hospitals than to community hospitals. We did not observe infants currently on supplemental oxygen, so we do not know to what degree using orders is valid in that context. We did not collect data on why actual monitoring status differed from monitoring orders and hence cannot quantify to what extent different factors (eg, nurse belief that monitors are a safety net or infants inadvertently left on monitors after a spot check pulse oximetry reading) contributed to this discordance. Finally, our study only examined one electronic health record variable—the presence of an active order. It may be that other variables in the health record (eg, minute-by-minute pulse oximetry values in a vital sign flowsheet) are much better proxies of actual continuous monitor use.
CONCLUSION
Using an active order for continuous pulse oximetry has poor sensitivity, PPV, and NPV for detecting true monitoring status at the bedside. Teams intending to measure the actual use of pulse oximetry should be aware of the limitations of using active orders alone as an accurate measure of pulse oximetry monitoring.
Acknowledgments
We thank the NHLBI scientists who contributed to this project as part of the U01 Cooperative Agreement funding mechanism: Lora Reineck, MD, MS, Karen Bienstock, MS, and Cheryl Boyce, PhD.
We thank the Executive Council of the PRIS Network for their contributions to the early scientific development of this project. We thank the PRIS site investigators for their major contributions to the Eliminating Monitor Overuse (EMO) Study data collection. Each listed collaborator is a group author for the PRIS Network in this manuscript. Their names can be found in the online supplemental information.
1. Ralston SL, Garber MD, Rice-Conboy E, et al. A multicenter collaborative to reduce unnecessary care in inpatient bronchiolitis. Pediatrics. 2016;137(1). https://doi.org/10.1542/peds.2015-0851
2. Mittal S, Marlowe L, Blakeslee S, et al. Successful use of quality improvement methodology to reduce inpatient length of stay in bronchiolitis through judicious use of intermittent pulse oximetry. Hosp Pediatr. 2019;9(2):73-78. https://doi.org/10.1542/hpeds.2018-0023
3. Quinonez RA, Coon ER, Schroeder AR, Moyer VA. When technology creates uncertainty: pulse oximetry and overdiagnosis of hypoxaemia in bronchiolitis. BMJ. 2017;358:j3850. https://doi.org/10.1136/bmj.j3850
4. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. https://doi.org/10.1002/jhm.2064
5. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
6. Principi T, Coates AL, Parkin PC, Stephens D, DaSilva Z, Schuh S. Effect of oxygen desaturations on subsequent medical visits in infants discharged from the emergency department with bronchiolitis. JAMA Pediatr. 2016;170(6):602-608. https://doi.org/10.1001/jamapediatrics.2016.0114
7. Cunningham S, Rodriguez A, Adams T, et al. Oxygen saturation targets in infants with bronchiolitis (BIDS): a double-blind, randomised, equivalence trial. Lancet. 2015;386(9998):1041-1048. https://doi.org/10.1016/s0140-6736(15)00163-4
8. McCulloh R, Koster M, Ralston S, et al. Use of intermittent vs continuous pulse oximetry for nonhypoxemic infants and young children hospitalized for bronchiolitis: a randomized clinical trial. JAMA Pediatr. 2015;169(10):898-904. https://doi.org/10.1001/jamapediatrics.2015.1746
9. Schuh S, Freedman S, Coates A, et al. Effect of oximetry on hospitalization in bronchiolitis: a randomized clinical trial. JAMA. 2014;312(7):712-718. https://doi.org/10.1001/jama.2014.8637
10. Schroeder AR, Marmor AK, Pantell RH, Newman TB. Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527-530. https://doi.org/10.1001/archpedi.158.6.527
11. Rasooly IR, Beidas RS, Wolk CB, et al. Measuring overuse of continuous pulse oximetry in bronchiolitis and developing strategies for large-scale deimplementation: study protocol for a feasibility trial. Pilot Feasibility Stud. 2019;5:68. https://doi.org/10.1186/s40814-019-0453-2
1. Ralston SL, Garber MD, Rice-Conboy E, et al. A multicenter collaborative to reduce unnecessary care in inpatient bronchiolitis. Pediatrics. 2016;137(1). https://doi.org/10.1542/peds.2015-0851
2. Mittal S, Marlowe L, Blakeslee S, et al. Successful use of quality improvement methodology to reduce inpatient length of stay in bronchiolitis through judicious use of intermittent pulse oximetry. Hosp Pediatr. 2019;9(2):73-78. https://doi.org/10.1542/hpeds.2018-0023
3. Quinonez RA, Coon ER, Schroeder AR, Moyer VA. When technology creates uncertainty: pulse oximetry and overdiagnosis of hypoxaemia in bronchiolitis. BMJ. 2017;358:j3850. https://doi.org/10.1136/bmj.j3850
4. Quinonez RA, Garber MD, Schroeder AR, et al. Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med. 2013;8(9):479-485. https://doi.org/10.1002/jhm.2064
5. Ralston SL, Lieberthal AS, Meissner HC, et al. Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics. 2014;134(5):e1474-e1502. https://doi.org/10.1542/peds.2014-2742
6. Principi T, Coates AL, Parkin PC, Stephens D, DaSilva Z, Schuh S. Effect of oxygen desaturations on subsequent medical visits in infants discharged from the emergency department with bronchiolitis. JAMA Pediatr. 2016;170(6):602-608. https://doi.org/10.1001/jamapediatrics.2016.0114
7. Cunningham S, Rodriguez A, Adams T, et al. Oxygen saturation targets in infants with bronchiolitis (BIDS): a double-blind, randomised, equivalence trial. Lancet. 2015;386(9998):1041-1048. https://doi.org/10.1016/s0140-6736(15)00163-4
8. McCulloh R, Koster M, Ralston S, et al. Use of intermittent vs continuous pulse oximetry for nonhypoxemic infants and young children hospitalized for bronchiolitis: a randomized clinical trial. JAMA Pediatr. 2015;169(10):898-904. https://doi.org/10.1001/jamapediatrics.2015.1746
9. Schuh S, Freedman S, Coates A, et al. Effect of oximetry on hospitalization in bronchiolitis: a randomized clinical trial. JAMA. 2014;312(7):712-718. https://doi.org/10.1001/jama.2014.8637
10. Schroeder AR, Marmor AK, Pantell RH, Newman TB. Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527-530. https://doi.org/10.1001/archpedi.158.6.527
11. Rasooly IR, Beidas RS, Wolk CB, et al. Measuring overuse of continuous pulse oximetry in bronchiolitis and developing strategies for large-scale deimplementation: study protocol for a feasibility trial. Pilot Feasibility Stud. 2019;5:68. https://doi.org/10.1186/s40814-019-0453-2
© 2020 Society of Hospital Medicine
Secure Text Messaging in Healthcare: Latent Threats and Opportunities to Improve Patient Safety
UNINTENDED CONSEQUENCES
Over the past two decades, physicians and nurses practicing in hospital settings have faced an onslaught of challenges in communication, an area frequently cited as critical to providing safe and effective care to patients.1-3 Communication needs have increased significantly as hospitalized patients have become more acute, complex, and technology-dependent, requiring larger healthcare teams comprising subspecialists across multiple disciplines spread across increasingly larger inpatient facilities.4 During this same period, the evolution of mobile phones has led to dramatic shifts in personal communication patterns, with asynchronous text messaging replacing verbal communication.5-7
In response to both the changing communication needs of clinicians and shifting cultural conventions, healthcare systems and providers alike have viewed text messaging as a solution to these growing communication problems. In fact, an entire industry has developed around “secure” and “Health Insurance Portability and Accountability Act (HIPAA)-compliant” text messaging platforms, which we will refer to below as secure text messaging systems (STMS). These systems offer benefits over carrier-based text messaging given their focus on the healthcare environment and HIPAA compliance. However, hospitals’ rapid adoption of these systems has outpaced our abilities to surveil, recognize, and understand the unintended consequences of transitioning to STMS communication in the hospital setting where failures in communication can be catastrophic. Below, we highlight three critical areas of concern encountered at our institutions and offer five potential mitigating strategies (Table).
CRITICAL AREAS OF CONCERN
Text Messaging is a Form of Alarm Fatigue
Text messaging renders clinicians vulnerable to a unique form of alarm fatigue. The burden of alarm fatigue has been well described in the literature and applies to interruptions to workflow in the electronic medical record and sensory alerts in clinical settings.8,9 Text messaging serves as yet another interruption for healthcare providers. Without a framework to triage urgent versus nonurgent messages, a clinician can become inundated with information and miss critical messages. This can lead to delayed or incorrect responses and impede patient care. System design and implementation can also contribute to this phenomenon. For example, a text message analysis at one center identified how system and workflow design resulted in all messages to an intensive care unit team being routed to a single physician’s phone.10 This design left the singular physician at risk of information and task overload and at the mercy of endless interruptive alerts. Although this can occur with any communication system, it has been well demonstrated that adopting STMS correlates with an increased frequency of messaging, leading to an increase in interruptive alerts, which may have implications for patient safety.11 This type of systems failure is silent unless proactively identified or revealed through a retrospective review of a resulting safety event.
Text Messaging Inappropriately Replaces Critical Communications that Should Happen in Person or by Phone
Text messaging has de-emphasized interpersonal communication skills and behaviors critical for quality and safety in hospital-based care. This concern emerges alongside evidence suggesting that new generations of physician trainees have profoundly different communication habits, preferences, and skillsets based on their experience in a text-heavy, asynchronous world of communication.12 There is reason to worry that reliance on text messaging in healthcare leads to similar alterations in relationships and collaboration as it has in our broader cultural context.13 Academic medical centers in particular should attempt to mitigate the loss of profound and formative learning that occurs during face-to-face encounters between providers of different disciplines, experience levels, and specialties.
Text Messaging Increases the Risk of Communication Error
Finally, text messaging appears to be highly vulnerable to communication errors in the healthcare setting. Prior work emphasizes the importance of nonverbal communication in face-to-face and even voice-to-voice interactions, highlighting the loss of fidelity when using text-only methods to communicate.1 Furthermore, the asynchronous nature of text messaging grants little room for clarification of minor misunderstandings that often arise in text-only communication through minor alterations in punctuation or automatic spelling corrections, a frequent occurrence when using medical terminology. Although a seasoned physician may be able to piece together the issues that deserve further clarification, young residents may be more hesitant to ask clarifying questions and determine the right course of action due to clinical inexperience.
PROPOSED SOLUTIONS
Deliberate Design and Implementation
A recent systematic review identified a lack of high-quality evidence evaluating the impact of mobile technologies on communication and teamwork in hospital settings.14 This paucity of understanding renders communication via STMS in the healthcare setting uniquely vulnerable to latent safety threats unless the design and implementation of these systems are purposeful and proactive.
These concerns led us to postulate that deliberate and proactive implementation of these systems, rather than passive adoption, is needed in the healthcare environment. We propose a number of approaches and interventions that may guide institutions as they seek to implement STMS or redesign communication in the inpatient setting. At the core of these proposals lies an important tension: can implementation of STMS occur in isolation or should the arrival of these systems prompt an overhaul of an institution’s clinical communication system and culture?15
Proactive Surveillance
Surveillance is one proactive method for healthcare systems to understand where and how the implementation of STMS might lead to safety threats. From a quantitative standpoint, understanding the burden of messaging for each user across the system can reveal the clinical roles in the system that are particularly vulnerable to alert fatigue or information overload. Quality assurance monitoring of critical roles in the hospital (ie, airway emergency team, rapid response teams) could be conducted to ensure accurate directory listings at all times. Associating conversations with events, from serious safety events to near misses, could help leaders understand when and how text messaging contributes to safety events and create actionable learnings for safety learning systems.
Standardized Communication
A standardized language eliminates the burden of individuals to parse and translate each individual text message. A standardized algorithm for language, urgency, and expectations (ie, response before escalation) would help define the interaction in the clinical setting.16 Moving toward standardized, meaningful “quick messages,” one of our centers has implemented a campaign to “stick to the FACS,” where the following four standard quick messages are available for users: (1) “FYI no response needed,” (2) “ACTION needed within X min,” (3) “CONCERN can we talk or meet,” and (4) “STAT immediate response required.” These quick messages, developed with frontline stakeholders, represent the majority of requests exchanged by providers, and help standardize expectations and task prioritization.
Targeted Training
Targeted training and culture change efforts might help institutions counteract the broader impact of asynchronous messaging on communication skills and behaviors. Highlighting the contrast between clinical and casual communication with an emphasis on examples, scenarios, or role-playing has the potential to emphasize why and how clinical communication with STMS requires a careful, deliberate approach. For instance, safety culture training at one of our institutions features a scenario that illustrates the potential for miscommunication and missed connection between a nurse and a physician on the wards. The scenario gives way to discussion between participants about the shortcomings of text messaging and allows the facilitator to segue into the “dos and don’ts” of text messaging and when a phone call might be more appropriate.
Innovate
Finally, creatively harnessing the technology and data underlying these STMS may uncover methods to identify and mitigate communication errors in real time. For instance, using trigger methods to create a “ripple in the pond,” whereby a floor nurse reaching out with an urgent text automatically loops in the charge nurse of the unit. Building a chatbot or a virtual assistant functionality by leveraging user behavior patterns and natural language processing to provide text-based guidance to users might help busy clinicians connect to the key decision-makers on their team. For example, in response to an unanswered text, a virtual assistant might reach out to the waiting provider as follows: “you texted the resident 20 minutes ago and they haven’t replied, would you like to call the fellow instead?” The data-rich nature of these systems implies that they are ripe for automated solutions that can respond to behavioral- or text-based patterns to augment the existing operation and safety infrastructure.
CONCLUSION
The transition of healthcare communication systems toward STMS is already well underway. These systems, despite their flaws, are undoubtedly an improvement over legacy paging systems and, if properly implemented, offer several benefits to large healthcare systems. However, the communication needs in the healthcare setting are vastly different from the personal communication needs in everyday text messaging. As clinicians at the forefront of these transitions, we have the opportunity to critically assess the unique communication requirements in our hospital settings and help shape the way STMS are implemented in our hospitals. Pausing to deliberate about the limitations and the vulnerabilities of the current messaging systems for our acute clinical needs, including how they impact training and education, will allow us to proactively design and implement better communication systems that improve patient safety.
1. Sutcliffe KM, Lewton E, Rosenthal MM. Communication failures: an insidious contributor to medical mishaps. Acad Med. 2004;79(2):186-194. https://doi.org/10.1097/00001888-200402000-00019.
2. Ash JS, Berg M, Coiera E. Some unintended consequences of information technology in health care: The nature of patient care information system-related errors. J Am Med Inform Assoc. 2004;11(2):104-112. https://doi.org/10.1197/jamia.M1471.
3. Coiera E. When conversation is better than computation. J Am Med Inform Assoc. 2000;7(3):277-286. https://doi.org/10.1136/jamia.2000.0070277.
4. Simon TD, Berry J, Feudtner C, et al. Children with complex chronic conditions in inpatient hospital settings in the United States. Pediatrics. 2010;126(4):647-655. https://doi.org/10.1542/peds.2009-3266.
5. The Nielsen Company. In U.S., SMS Text Messaging Tops Mobile Phone Calling. https://www.nielsen.com/us/en/insights/article/2008/in-us-text-messaging-tops-mobile-phone-calling/. Accessed July 22, 2019.
6. The Nielsen Company. New Mobile Obsession in U.S. Teens Triple Data Usage. The Nielsen Company. Published 2011. Accessed July 22, 2019.
7. The Nielsen Company. U.S. Teen Mobile Report Calling Yesterday, Texting Today, Using Apps Tomorrow. The Nielsen Company. https://www.nielsen.com/us/en/insights/article/2010/u-s-teen-mobile-report-calling-yesterday-texting-today-using-apps-tomorrow/. Accessed July 22, 2019.
8. Sendelbach S, Funk M. Alarm fatigue: a patient safety concern. AACN Adv Crit Care. 2013;24(4):378-386; quiz 387-378.
9. Paine CW, Goel VV, Ely E, et al. Systematic review of physiologic monitor alarm characteristics and pragmatic interventions to reduce alarm frequency. J Hosp Med. 2016;11(2):136-144. https://doi.org/10.1002/jhm.2520.
10. Hagedorn PA, Kirkendall ES, Spooner SA, Mohan V. Inpatient communication networks: leveraging secure text-messaging platforms to gain insight into inpatient communication systems. Appl Clin Inform. 2019;10(3):471-478. https://doi.org/10.1055/s-0039-1692401.
11. Westbrook JI, Coiera E, Dunsmuir WT, et al. The impact of interruptions on clinical task completion. Qual Saf Health Care. 2010;19(4):284-289. https://doi.org/10.1136/qshc.2009.039255.
12. Castells M. The Rise of the Network Society. 2nd ed. Malden, MA: Wiley-Blackwell; 2010.
13. Lo V, Wu RC, Morra D, Lee L, Reeves S. The use of smartphones in general and internal medicine units: A boon or a bane to the promotion of interprofessional collaboration? J Interprof Care. 2012;26(4):276-282. https://doi.org/10.3109/13561820.2012.663013.
14. Martin G, Khajuria A, Arora S, King D, Ashrafian H, Darzi A. The impact of mobile technology on teamwork and communication in hospitals: a systematic review. J Am Med Inform Assn. 2019;26(4):339-355. https://doi.org/10.1093/jamia/ocy175.
15. Liu X, Sutton PR, McKenna R, et al. Evaluation of secure messaging applications for a health care system: a case study. Appl Clin Inform. 2019;10(1):140-150. https://doi.org/10.1055/s-0039-1678607.
16. Weigert RM, Schmitz AH, Soung PJ, Porada K, Weisgerber MC. Improving standardization of paging communication using quality improvement methodology. Pediatrics. 2019;143(4). https://doi.org/10.1542/peds.2018-1362.
UNINTENDED CONSEQUENCES
Over the past two decades, physicians and nurses practicing in hospital settings have faced an onslaught of challenges in communication, an area frequently cited as critical to providing safe and effective care to patients.1-3 Communication needs have increased significantly as hospitalized patients have become more acute, complex, and technology-dependent, requiring larger healthcare teams comprising subspecialists across multiple disciplines spread across increasingly larger inpatient facilities.4 During this same period, the evolution of mobile phones has led to dramatic shifts in personal communication patterns, with asynchronous text messaging replacing verbal communication.5-7
In response to both the changing communication needs of clinicians and shifting cultural conventions, healthcare systems and providers alike have viewed text messaging as a solution to these growing communication problems. In fact, an entire industry has developed around “secure” and “Health Insurance Portability and Accountability Act (HIPAA)-compliant” text messaging platforms, which we will refer to below as secure text messaging systems (STMS). These systems offer benefits over carrier-based text messaging given their focus on the healthcare environment and HIPAA compliance. However, hospitals’ rapid adoption of these systems has outpaced our abilities to surveil, recognize, and understand the unintended consequences of transitioning to STMS communication in the hospital setting where failures in communication can be catastrophic. Below, we highlight three critical areas of concern encountered at our institutions and offer five potential mitigating strategies (Table).
CRITICAL AREAS OF CONCERN
Text Messaging is a Form of Alarm Fatigue
Text messaging renders clinicians vulnerable to a unique form of alarm fatigue. The burden of alarm fatigue has been well described in the literature and applies to interruptions to workflow in the electronic medical record and sensory alerts in clinical settings.8,9 Text messaging serves as yet another interruption for healthcare providers. Without a framework to triage urgent versus nonurgent messages, a clinician can become inundated with information and miss critical messages. This can lead to delayed or incorrect responses and impede patient care. System design and implementation can also contribute to this phenomenon. For example, a text message analysis at one center identified how system and workflow design resulted in all messages to an intensive care unit team being routed to a single physician’s phone.10 This design left the singular physician at risk of information and task overload and at the mercy of endless interruptive alerts. Although this can occur with any communication system, it has been well demonstrated that adopting STMS correlates with an increased frequency of messaging, leading to an increase in interruptive alerts, which may have implications for patient safety.11 This type of systems failure is silent unless proactively identified or revealed through a retrospective review of a resulting safety event.
Text Messaging Inappropriately Replaces Critical Communications that Should Happen in Person or by Phone
Text messaging has de-emphasized interpersonal communication skills and behaviors critical for quality and safety in hospital-based care. This concern emerges alongside evidence suggesting that new generations of physician trainees have profoundly different communication habits, preferences, and skillsets based on their experience in a text-heavy, asynchronous world of communication.12 There is reason to worry that reliance on text messaging in healthcare leads to similar alterations in relationships and collaboration as it has in our broader cultural context.13 Academic medical centers in particular should attempt to mitigate the loss of profound and formative learning that occurs during face-to-face encounters between providers of different disciplines, experience levels, and specialties.
Text Messaging Increases the Risk of Communication Error
Finally, text messaging appears to be highly vulnerable to communication errors in the healthcare setting. Prior work emphasizes the importance of nonverbal communication in face-to-face and even voice-to-voice interactions, highlighting the loss of fidelity when using text-only methods to communicate.1 Furthermore, the asynchronous nature of text messaging grants little room for clarification of minor misunderstandings that often arise in text-only communication through minor alterations in punctuation or automatic spelling corrections, a frequent occurrence when using medical terminology. Although a seasoned physician may be able to piece together the issues that deserve further clarification, young residents may be more hesitant to ask clarifying questions and determine the right course of action due to clinical inexperience.
PROPOSED SOLUTIONS
Deliberate Design and Implementation
A recent systematic review identified a lack of high-quality evidence evaluating the impact of mobile technologies on communication and teamwork in hospital settings.14 This paucity of understanding renders communication via STMS in the healthcare setting uniquely vulnerable to latent safety threats unless the design and implementation of these systems are purposeful and proactive.
These concerns led us to postulate that deliberate and proactive implementation of these systems, rather than passive adoption, is needed in the healthcare environment. We propose a number of approaches and interventions that may guide institutions as they seek to implement STMS or redesign communication in the inpatient setting. At the core of these proposals lies an important tension: can implementation of STMS occur in isolation or should the arrival of these systems prompt an overhaul of an institution’s clinical communication system and culture?15
Proactive Surveillance
Surveillance is one proactive method for healthcare systems to understand where and how the implementation of STMS might lead to safety threats. From a quantitative standpoint, understanding the burden of messaging for each user across the system can reveal the clinical roles in the system that are particularly vulnerable to alert fatigue or information overload. Quality assurance monitoring of critical roles in the hospital (ie, airway emergency team, rapid response teams) could be conducted to ensure accurate directory listings at all times. Associating conversations with events, from serious safety events to near misses, could help leaders understand when and how text messaging contributes to safety events and create actionable learnings for safety learning systems.
Standardized Communication
A standardized language eliminates the burden of individuals to parse and translate each individual text message. A standardized algorithm for language, urgency, and expectations (ie, response before escalation) would help define the interaction in the clinical setting.16 Moving toward standardized, meaningful “quick messages,” one of our centers has implemented a campaign to “stick to the FACS,” where the following four standard quick messages are available for users: (1) “FYI no response needed,” (2) “ACTION needed within X min,” (3) “CONCERN can we talk or meet,” and (4) “STAT immediate response required.” These quick messages, developed with frontline stakeholders, represent the majority of requests exchanged by providers, and help standardize expectations and task prioritization.
Targeted Training
Targeted training and culture change efforts might help institutions counteract the broader impact of asynchronous messaging on communication skills and behaviors. Highlighting the contrast between clinical and casual communication with an emphasis on examples, scenarios, or role-playing has the potential to emphasize why and how clinical communication with STMS requires a careful, deliberate approach. For instance, safety culture training at one of our institutions features a scenario that illustrates the potential for miscommunication and missed connection between a nurse and a physician on the wards. The scenario gives way to discussion between participants about the shortcomings of text messaging and allows the facilitator to segue into the “dos and don’ts” of text messaging and when a phone call might be more appropriate.
Innovate
Finally, creatively harnessing the technology and data underlying these STMS may uncover methods to identify and mitigate communication errors in real time. For instance, using trigger methods to create a “ripple in the pond,” whereby a floor nurse reaching out with an urgent text automatically loops in the charge nurse of the unit. Building a chatbot or a virtual assistant functionality by leveraging user behavior patterns and natural language processing to provide text-based guidance to users might help busy clinicians connect to the key decision-makers on their team. For example, in response to an unanswered text, a virtual assistant might reach out to the waiting provider as follows: “you texted the resident 20 minutes ago and they haven’t replied, would you like to call the fellow instead?” The data-rich nature of these systems implies that they are ripe for automated solutions that can respond to behavioral- or text-based patterns to augment the existing operation and safety infrastructure.
CONCLUSION
The transition of healthcare communication systems toward STMS is already well underway. These systems, despite their flaws, are undoubtedly an improvement over legacy paging systems and, if properly implemented, offer several benefits to large healthcare systems. However, the communication needs in the healthcare setting are vastly different from the personal communication needs in everyday text messaging. As clinicians at the forefront of these transitions, we have the opportunity to critically assess the unique communication requirements in our hospital settings and help shape the way STMS are implemented in our hospitals. Pausing to deliberate about the limitations and the vulnerabilities of the current messaging systems for our acute clinical needs, including how they impact training and education, will allow us to proactively design and implement better communication systems that improve patient safety.
UNINTENDED CONSEQUENCES
Over the past two decades, physicians and nurses practicing in hospital settings have faced an onslaught of challenges in communication, an area frequently cited as critical to providing safe and effective care to patients.1-3 Communication needs have increased significantly as hospitalized patients have become more acute, complex, and technology-dependent, requiring larger healthcare teams comprising subspecialists across multiple disciplines spread across increasingly larger inpatient facilities.4 During this same period, the evolution of mobile phones has led to dramatic shifts in personal communication patterns, with asynchronous text messaging replacing verbal communication.5-7
In response to both the changing communication needs of clinicians and shifting cultural conventions, healthcare systems and providers alike have viewed text messaging as a solution to these growing communication problems. In fact, an entire industry has developed around “secure” and “Health Insurance Portability and Accountability Act (HIPAA)-compliant” text messaging platforms, which we will refer to below as secure text messaging systems (STMS). These systems offer benefits over carrier-based text messaging given their focus on the healthcare environment and HIPAA compliance. However, hospitals’ rapid adoption of these systems has outpaced our abilities to surveil, recognize, and understand the unintended consequences of transitioning to STMS communication in the hospital setting where failures in communication can be catastrophic. Below, we highlight three critical areas of concern encountered at our institutions and offer five potential mitigating strategies (Table).
CRITICAL AREAS OF CONCERN
Text Messaging is a Form of Alarm Fatigue
Text messaging renders clinicians vulnerable to a unique form of alarm fatigue. The burden of alarm fatigue has been well described in the literature and applies to interruptions to workflow in the electronic medical record and sensory alerts in clinical settings.8,9 Text messaging serves as yet another interruption for healthcare providers. Without a framework to triage urgent versus nonurgent messages, a clinician can become inundated with information and miss critical messages. This can lead to delayed or incorrect responses and impede patient care. System design and implementation can also contribute to this phenomenon. For example, a text message analysis at one center identified how system and workflow design resulted in all messages to an intensive care unit team being routed to a single physician’s phone.10 This design left the singular physician at risk of information and task overload and at the mercy of endless interruptive alerts. Although this can occur with any communication system, it has been well demonstrated that adopting STMS correlates with an increased frequency of messaging, leading to an increase in interruptive alerts, which may have implications for patient safety.11 This type of systems failure is silent unless proactively identified or revealed through a retrospective review of a resulting safety event.
Text Messaging Inappropriately Replaces Critical Communications that Should Happen in Person or by Phone
Text messaging has de-emphasized interpersonal communication skills and behaviors critical for quality and safety in hospital-based care. This concern emerges alongside evidence suggesting that new generations of physician trainees have profoundly different communication habits, preferences, and skillsets based on their experience in a text-heavy, asynchronous world of communication.12 There is reason to worry that reliance on text messaging in healthcare leads to similar alterations in relationships and collaboration as it has in our broader cultural context.13 Academic medical centers in particular should attempt to mitigate the loss of profound and formative learning that occurs during face-to-face encounters between providers of different disciplines, experience levels, and specialties.
Text Messaging Increases the Risk of Communication Error
Finally, text messaging appears to be highly vulnerable to communication errors in the healthcare setting. Prior work emphasizes the importance of nonverbal communication in face-to-face and even voice-to-voice interactions, highlighting the loss of fidelity when using text-only methods to communicate.1 Furthermore, the asynchronous nature of text messaging grants little room for clarification of minor misunderstandings that often arise in text-only communication through minor alterations in punctuation or automatic spelling corrections, a frequent occurrence when using medical terminology. Although a seasoned physician may be able to piece together the issues that deserve further clarification, young residents may be more hesitant to ask clarifying questions and determine the right course of action due to clinical inexperience.
PROPOSED SOLUTIONS
Deliberate Design and Implementation
A recent systematic review identified a lack of high-quality evidence evaluating the impact of mobile technologies on communication and teamwork in hospital settings.14 This paucity of understanding renders communication via STMS in the healthcare setting uniquely vulnerable to latent safety threats unless the design and implementation of these systems are purposeful and proactive.
These concerns led us to postulate that deliberate and proactive implementation of these systems, rather than passive adoption, is needed in the healthcare environment. We propose a number of approaches and interventions that may guide institutions as they seek to implement STMS or redesign communication in the inpatient setting. At the core of these proposals lies an important tension: can implementation of STMS occur in isolation or should the arrival of these systems prompt an overhaul of an institution’s clinical communication system and culture?15
Proactive Surveillance
Surveillance is one proactive method for healthcare systems to understand where and how the implementation of STMS might lead to safety threats. From a quantitative standpoint, understanding the burden of messaging for each user across the system can reveal the clinical roles in the system that are particularly vulnerable to alert fatigue or information overload. Quality assurance monitoring of critical roles in the hospital (ie, airway emergency team, rapid response teams) could be conducted to ensure accurate directory listings at all times. Associating conversations with events, from serious safety events to near misses, could help leaders understand when and how text messaging contributes to safety events and create actionable learnings for safety learning systems.
Standardized Communication
A standardized language eliminates the burden of individuals to parse and translate each individual text message. A standardized algorithm for language, urgency, and expectations (ie, response before escalation) would help define the interaction in the clinical setting.16 Moving toward standardized, meaningful “quick messages,” one of our centers has implemented a campaign to “stick to the FACS,” where the following four standard quick messages are available for users: (1) “FYI no response needed,” (2) “ACTION needed within X min,” (3) “CONCERN can we talk or meet,” and (4) “STAT immediate response required.” These quick messages, developed with frontline stakeholders, represent the majority of requests exchanged by providers, and help standardize expectations and task prioritization.
Targeted Training
Targeted training and culture change efforts might help institutions counteract the broader impact of asynchronous messaging on communication skills and behaviors. Highlighting the contrast between clinical and casual communication with an emphasis on examples, scenarios, or role-playing has the potential to emphasize why and how clinical communication with STMS requires a careful, deliberate approach. For instance, safety culture training at one of our institutions features a scenario that illustrates the potential for miscommunication and missed connection between a nurse and a physician on the wards. The scenario gives way to discussion between participants about the shortcomings of text messaging and allows the facilitator to segue into the “dos and don’ts” of text messaging and when a phone call might be more appropriate.
Innovate
Finally, creatively harnessing the technology and data underlying these STMS may uncover methods to identify and mitigate communication errors in real time. For instance, using trigger methods to create a “ripple in the pond,” whereby a floor nurse reaching out with an urgent text automatically loops in the charge nurse of the unit. Building a chatbot or a virtual assistant functionality by leveraging user behavior patterns and natural language processing to provide text-based guidance to users might help busy clinicians connect to the key decision-makers on their team. For example, in response to an unanswered text, a virtual assistant might reach out to the waiting provider as follows: “you texted the resident 20 minutes ago and they haven’t replied, would you like to call the fellow instead?” The data-rich nature of these systems implies that they are ripe for automated solutions that can respond to behavioral- or text-based patterns to augment the existing operation and safety infrastructure.
CONCLUSION
The transition of healthcare communication systems toward STMS is already well underway. These systems, despite their flaws, are undoubtedly an improvement over legacy paging systems and, if properly implemented, offer several benefits to large healthcare systems. However, the communication needs in the healthcare setting are vastly different from the personal communication needs in everyday text messaging. As clinicians at the forefront of these transitions, we have the opportunity to critically assess the unique communication requirements in our hospital settings and help shape the way STMS are implemented in our hospitals. Pausing to deliberate about the limitations and the vulnerabilities of the current messaging systems for our acute clinical needs, including how they impact training and education, will allow us to proactively design and implement better communication systems that improve patient safety.
1. Sutcliffe KM, Lewton E, Rosenthal MM. Communication failures: an insidious contributor to medical mishaps. Acad Med. 2004;79(2):186-194. https://doi.org/10.1097/00001888-200402000-00019.
2. Ash JS, Berg M, Coiera E. Some unintended consequences of information technology in health care: The nature of patient care information system-related errors. J Am Med Inform Assoc. 2004;11(2):104-112. https://doi.org/10.1197/jamia.M1471.
3. Coiera E. When conversation is better than computation. J Am Med Inform Assoc. 2000;7(3):277-286. https://doi.org/10.1136/jamia.2000.0070277.
4. Simon TD, Berry J, Feudtner C, et al. Children with complex chronic conditions in inpatient hospital settings in the United States. Pediatrics. 2010;126(4):647-655. https://doi.org/10.1542/peds.2009-3266.
5. The Nielsen Company. In U.S., SMS Text Messaging Tops Mobile Phone Calling. https://www.nielsen.com/us/en/insights/article/2008/in-us-text-messaging-tops-mobile-phone-calling/. Accessed July 22, 2019.
6. The Nielsen Company. New Mobile Obsession in U.S. Teens Triple Data Usage. The Nielsen Company. Published 2011. Accessed July 22, 2019.
7. The Nielsen Company. U.S. Teen Mobile Report Calling Yesterday, Texting Today, Using Apps Tomorrow. The Nielsen Company. https://www.nielsen.com/us/en/insights/article/2010/u-s-teen-mobile-report-calling-yesterday-texting-today-using-apps-tomorrow/. Accessed July 22, 2019.
8. Sendelbach S, Funk M. Alarm fatigue: a patient safety concern. AACN Adv Crit Care. 2013;24(4):378-386; quiz 387-378.
9. Paine CW, Goel VV, Ely E, et al. Systematic review of physiologic monitor alarm characteristics and pragmatic interventions to reduce alarm frequency. J Hosp Med. 2016;11(2):136-144. https://doi.org/10.1002/jhm.2520.
10. Hagedorn PA, Kirkendall ES, Spooner SA, Mohan V. Inpatient communication networks: leveraging secure text-messaging platforms to gain insight into inpatient communication systems. Appl Clin Inform. 2019;10(3):471-478. https://doi.org/10.1055/s-0039-1692401.
11. Westbrook JI, Coiera E, Dunsmuir WT, et al. The impact of interruptions on clinical task completion. Qual Saf Health Care. 2010;19(4):284-289. https://doi.org/10.1136/qshc.2009.039255.
12. Castells M. The Rise of the Network Society. 2nd ed. Malden, MA: Wiley-Blackwell; 2010.
13. Lo V, Wu RC, Morra D, Lee L, Reeves S. The use of smartphones in general and internal medicine units: A boon or a bane to the promotion of interprofessional collaboration? J Interprof Care. 2012;26(4):276-282. https://doi.org/10.3109/13561820.2012.663013.
14. Martin G, Khajuria A, Arora S, King D, Ashrafian H, Darzi A. The impact of mobile technology on teamwork and communication in hospitals: a systematic review. J Am Med Inform Assn. 2019;26(4):339-355. https://doi.org/10.1093/jamia/ocy175.
15. Liu X, Sutton PR, McKenna R, et al. Evaluation of secure messaging applications for a health care system: a case study. Appl Clin Inform. 2019;10(1):140-150. https://doi.org/10.1055/s-0039-1678607.
16. Weigert RM, Schmitz AH, Soung PJ, Porada K, Weisgerber MC. Improving standardization of paging communication using quality improvement methodology. Pediatrics. 2019;143(4). https://doi.org/10.1542/peds.2018-1362.
1. Sutcliffe KM, Lewton E, Rosenthal MM. Communication failures: an insidious contributor to medical mishaps. Acad Med. 2004;79(2):186-194. https://doi.org/10.1097/00001888-200402000-00019.
2. Ash JS, Berg M, Coiera E. Some unintended consequences of information technology in health care: The nature of patient care information system-related errors. J Am Med Inform Assoc. 2004;11(2):104-112. https://doi.org/10.1197/jamia.M1471.
3. Coiera E. When conversation is better than computation. J Am Med Inform Assoc. 2000;7(3):277-286. https://doi.org/10.1136/jamia.2000.0070277.
4. Simon TD, Berry J, Feudtner C, et al. Children with complex chronic conditions in inpatient hospital settings in the United States. Pediatrics. 2010;126(4):647-655. https://doi.org/10.1542/peds.2009-3266.
5. The Nielsen Company. In U.S., SMS Text Messaging Tops Mobile Phone Calling. https://www.nielsen.com/us/en/insights/article/2008/in-us-text-messaging-tops-mobile-phone-calling/. Accessed July 22, 2019.
6. The Nielsen Company. New Mobile Obsession in U.S. Teens Triple Data Usage. The Nielsen Company. Published 2011. Accessed July 22, 2019.
7. The Nielsen Company. U.S. Teen Mobile Report Calling Yesterday, Texting Today, Using Apps Tomorrow. The Nielsen Company. https://www.nielsen.com/us/en/insights/article/2010/u-s-teen-mobile-report-calling-yesterday-texting-today-using-apps-tomorrow/. Accessed July 22, 2019.
8. Sendelbach S, Funk M. Alarm fatigue: a patient safety concern. AACN Adv Crit Care. 2013;24(4):378-386; quiz 387-378.
9. Paine CW, Goel VV, Ely E, et al. Systematic review of physiologic monitor alarm characteristics and pragmatic interventions to reduce alarm frequency. J Hosp Med. 2016;11(2):136-144. https://doi.org/10.1002/jhm.2520.
10. Hagedorn PA, Kirkendall ES, Spooner SA, Mohan V. Inpatient communication networks: leveraging secure text-messaging platforms to gain insight into inpatient communication systems. Appl Clin Inform. 2019;10(3):471-478. https://doi.org/10.1055/s-0039-1692401.
11. Westbrook JI, Coiera E, Dunsmuir WT, et al. The impact of interruptions on clinical task completion. Qual Saf Health Care. 2010;19(4):284-289. https://doi.org/10.1136/qshc.2009.039255.
12. Castells M. The Rise of the Network Society. 2nd ed. Malden, MA: Wiley-Blackwell; 2010.
13. Lo V, Wu RC, Morra D, Lee L, Reeves S. The use of smartphones in general and internal medicine units: A boon or a bane to the promotion of interprofessional collaboration? J Interprof Care. 2012;26(4):276-282. https://doi.org/10.3109/13561820.2012.663013.
14. Martin G, Khajuria A, Arora S, King D, Ashrafian H, Darzi A. The impact of mobile technology on teamwork and communication in hospitals: a systematic review. J Am Med Inform Assn. 2019;26(4):339-355. https://doi.org/10.1093/jamia/ocy175.
15. Liu X, Sutton PR, McKenna R, et al. Evaluation of secure messaging applications for a health care system: a case study. Appl Clin Inform. 2019;10(1):140-150. https://doi.org/10.1055/s-0039-1678607.
16. Weigert RM, Schmitz AH, Soung PJ, Porada K, Weisgerber MC. Improving standardization of paging communication using quality improvement methodology. Pediatrics. 2019;143(4). https://doi.org/10.1542/peds.2018-1362.
© 2019 Society of Hospital Medicine
Thinking Aloud: How Nurses Rationalize Responses to Monitor Alarms
In the past five years, it has become increasingly apparent that hospital physiologic monitoring systems are not functioning optimally for children. On pediatric wards, 26%-48% of children are continuously monitored, and these children generate between 42 and 155 alarms per day.1 Just 1% or fewer are considered actionable or informative, slowing nurses’ response times and placing patients at risk of delayed recognition of life-threatening events.2,3 While some factors associated with alarm response times have been elucidated,3 in order to design safe and effective monitoring systems, further work is needed to understand the complex decision-making process that nurses face when encountering alarms outside a patient’s room. It is in this area that Schondelmeyer and colleagues strive to enhance our understanding in this issue of the Journal of Hospital Medicine.4
Schondelmeyer et al. conducted a single-center, observational study using mixed methods in a general pediatric unit. Trained observers shadowed nine nurses one to four times each, during which nurses were asked to “think aloud” as they managed physiologic monitor alarms, rationalizing their decisions about how and why they might respond for the observer to document. Observers accumulated 61 patient-hours of observation before investigators halted data collection because new insights about alarm responses were no longer emerging from the data (thematic saturation).
Nurses thought aloud about 207 alarms during the study, which the investigators estimated comprised about one third of the alarms that occurred during observation periods. Most of the 207 occurred while the nurse was already in the patient’s room, where a response decision is uncomplicated. More interesting were the 45 alarms heard while outside the patient’s room, where nurses face the complex decision of whether to interrupt their current tasks and respond or delay their response and assume the associated risk of nonresponse to a potentially deteriorating patient. Of the 45 alarms, nurses went into the room to evaluate the patient 15 times and, after doing so, reported that five of the 15 warranted in-person responses to address technical issues with the monitor, clinical issues, or patients’ comfort. Reassuring clinical contexts—such as presence of the medical team or family in the room and recent patient assessments—were the reasons most commonly provided to explain alarm nonresponse.
This study has two key limitations. First, the authors designed the study to observe nurses’ responses until thematic saturation was achieved. However, the small sample size (nine nurses, 45 out-of-room alarms) could raise questions about whether sufficient data were captured to make broadly generalizable conclusions, given the diverse range of patients, families, and clinical scenarios nurses encounter on an inpatient unit. Second, by instructing nurse participants to verbalize their rationale for response or nonresponse, investigators essentially asked nurses to override the “Type 1”, heuristic-based reasoning5 that research suggests regulates nursing responses to alarms when adapting to circumstances requiring high cognitive demand or a heavy workload.3 While innovative, it is possible that this approach prevented the investigators from fully achieving their stated objective of describing how bedside nurses think about and act upon alarms.
Nonetheless, the findings by Schondelmeyer and colleagues extend our emerging understanding of why alarm responses are disconcertingly slow. Nursing staff’s dismissal of monitor alarms that are discordant with a reassuring patient evaluation underscores the imperative to reduce nuisance alarms. Furthermore, the explicit statements justifying alarm nonresponse because of the presence of family members build upon prior findings of longer response times when family members are at the bedside3 and invite a provocative question: how would family members feel if they knew that they were being entrusted as a foundational component of safety monitoring in the hospital? In their recently published study conducted at the same hospital,6 Schondelmeyer’s team elicited perceptions that families are deeply concerned about staff nonresponse to alarms—as one nurse stated, parents “wonder what’s going on when no one comes in.” While there is a valuable role for integrating families into efforts to overcome threats to patient safety, as has been achieved with family error reporting7 and communication on family-centered rounds,8 this must occur in a structured, explicit, and deliberate manner, with families engaged as key stakeholders.
In summary, while Schondelmeyer and colleagues may not have exposed the depth of implicit thinking that governs nurses’ responses to alarms, they have highlighted the high-stakes decisions that nurses confront on a daily basis in an environment with exceedingly high alarm rates and low alarm actionability. The authors cite staff education among potential solutions to improve the safety of continuous monitoring, but such an intervention cannot be effective in a system that places impossible burdens on nurses. An openly family centered and multidisciplinary approach to reengineering the system for monitoring hospitalized children is needed to enable nurses to respond quickly and accurately to patients at risk of clinical deterioration.
Disclosures
The authors report no conflicts of interest.
1. Schondelmeyer AC, Brady PW, Goel VV, et al. Physiologic monitor alarm rates at 5 children’s hospitals. J Hosp Med. 2018;13(6):396-398. https://doi.org/10.12788/jhm.2918.
2. Bonafide CP, Lin R, Zander M, et al. Association between exposure to nonactionable physiologic monitor alarms and response time in a children’s hospital. J Hosp Med. 2015;10(6):345-351. https://doi.org/10.1002/jhm.2331.
3. Bonafide CP, Localio AR, Holmes JH, et al. Video analysis of factors associated with response time to physiologic monitor alarms in a children’s hospital. JAMA Pediatr. 2017;171(6):524-531. https://doi.org/10.1001/jamapediatrics.2016.5123.
4. Schondelmeyer A, Daraiseh NM, Allison B, et al. Nurse responses to physiologic monitor alarms on a general pediatric unit. J Hosp Med. 2019;14(10):602-606. https://doi.org/10.12788/jhm.3234.
5. Croskerry P. A universal model of diagnostic reasoning. Acad Med. 2009;84(8):1022-1028. https://doi.org/10.1097/ACM.0b013e3181ace703.
6. Schondelmeyer AC, Jenkins AM, Allison B, et al. Factors influencing use of continuous physiologic monitors for hospitalized pediatric patients. Hosp Pediatr. 2019;9(6):423-428. https://doi.org/10.1542/hpeds.2019-0007.
7. Khan A, Coffey M, Litterer KP, et al. Families as partners in hospital error and adverse event surveillance. JAMA Pediatr. 2017;171(4):372-381. https://doi.org/10.1001/jamapediatrics.2016.4812.
8. Khan A, Spector ND, Baird JD, et al. Patient safety after implementation of a coproduced family centered communication programme: multicenter before and after intervention study. BMJ. 2018;363:k4764. https://doi.org/10.1136/bmj.k4764.
In the past five years, it has become increasingly apparent that hospital physiologic monitoring systems are not functioning optimally for children. On pediatric wards, 26%-48% of children are continuously monitored, and these children generate between 42 and 155 alarms per day.1 Just 1% or fewer are considered actionable or informative, slowing nurses’ response times and placing patients at risk of delayed recognition of life-threatening events.2,3 While some factors associated with alarm response times have been elucidated,3 in order to design safe and effective monitoring systems, further work is needed to understand the complex decision-making process that nurses face when encountering alarms outside a patient’s room. It is in this area that Schondelmeyer and colleagues strive to enhance our understanding in this issue of the Journal of Hospital Medicine.4
Schondelmeyer et al. conducted a single-center, observational study using mixed methods in a general pediatric unit. Trained observers shadowed nine nurses one to four times each, during which nurses were asked to “think aloud” as they managed physiologic monitor alarms, rationalizing their decisions about how and why they might respond for the observer to document. Observers accumulated 61 patient-hours of observation before investigators halted data collection because new insights about alarm responses were no longer emerging from the data (thematic saturation).
Nurses thought aloud about 207 alarms during the study, which the investigators estimated comprised about one third of the alarms that occurred during observation periods. Most of the 207 occurred while the nurse was already in the patient’s room, where a response decision is uncomplicated. More interesting were the 45 alarms heard while outside the patient’s room, where nurses face the complex decision of whether to interrupt their current tasks and respond or delay their response and assume the associated risk of nonresponse to a potentially deteriorating patient. Of the 45 alarms, nurses went into the room to evaluate the patient 15 times and, after doing so, reported that five of the 15 warranted in-person responses to address technical issues with the monitor, clinical issues, or patients’ comfort. Reassuring clinical contexts—such as presence of the medical team or family in the room and recent patient assessments—were the reasons most commonly provided to explain alarm nonresponse.
This study has two key limitations. First, the authors designed the study to observe nurses’ responses until thematic saturation was achieved. However, the small sample size (nine nurses, 45 out-of-room alarms) could raise questions about whether sufficient data were captured to make broadly generalizable conclusions, given the diverse range of patients, families, and clinical scenarios nurses encounter on an inpatient unit. Second, by instructing nurse participants to verbalize their rationale for response or nonresponse, investigators essentially asked nurses to override the “Type 1”, heuristic-based reasoning5 that research suggests regulates nursing responses to alarms when adapting to circumstances requiring high cognitive demand or a heavy workload.3 While innovative, it is possible that this approach prevented the investigators from fully achieving their stated objective of describing how bedside nurses think about and act upon alarms.
Nonetheless, the findings by Schondelmeyer and colleagues extend our emerging understanding of why alarm responses are disconcertingly slow. Nursing staff’s dismissal of monitor alarms that are discordant with a reassuring patient evaluation underscores the imperative to reduce nuisance alarms. Furthermore, the explicit statements justifying alarm nonresponse because of the presence of family members build upon prior findings of longer response times when family members are at the bedside3 and invite a provocative question: how would family members feel if they knew that they were being entrusted as a foundational component of safety monitoring in the hospital? In their recently published study conducted at the same hospital,6 Schondelmeyer’s team elicited perceptions that families are deeply concerned about staff nonresponse to alarms—as one nurse stated, parents “wonder what’s going on when no one comes in.” While there is a valuable role for integrating families into efforts to overcome threats to patient safety, as has been achieved with family error reporting7 and communication on family-centered rounds,8 this must occur in a structured, explicit, and deliberate manner, with families engaged as key stakeholders.
In summary, while Schondelmeyer and colleagues may not have exposed the depth of implicit thinking that governs nurses’ responses to alarms, they have highlighted the high-stakes decisions that nurses confront on a daily basis in an environment with exceedingly high alarm rates and low alarm actionability. The authors cite staff education among potential solutions to improve the safety of continuous monitoring, but such an intervention cannot be effective in a system that places impossible burdens on nurses. An openly family centered and multidisciplinary approach to reengineering the system for monitoring hospitalized children is needed to enable nurses to respond quickly and accurately to patients at risk of clinical deterioration.
Disclosures
The authors report no conflicts of interest.
In the past five years, it has become increasingly apparent that hospital physiologic monitoring systems are not functioning optimally for children. On pediatric wards, 26%-48% of children are continuously monitored, and these children generate between 42 and 155 alarms per day.1 Just 1% or fewer are considered actionable or informative, slowing nurses’ response times and placing patients at risk of delayed recognition of life-threatening events.2,3 While some factors associated with alarm response times have been elucidated,3 in order to design safe and effective monitoring systems, further work is needed to understand the complex decision-making process that nurses face when encountering alarms outside a patient’s room. It is in this area that Schondelmeyer and colleagues strive to enhance our understanding in this issue of the Journal of Hospital Medicine.4
Schondelmeyer et al. conducted a single-center, observational study using mixed methods in a general pediatric unit. Trained observers shadowed nine nurses one to four times each, during which nurses were asked to “think aloud” as they managed physiologic monitor alarms, rationalizing their decisions about how and why they might respond for the observer to document. Observers accumulated 61 patient-hours of observation before investigators halted data collection because new insights about alarm responses were no longer emerging from the data (thematic saturation).
Nurses thought aloud about 207 alarms during the study, which the investigators estimated comprised about one third of the alarms that occurred during observation periods. Most of the 207 occurred while the nurse was already in the patient’s room, where a response decision is uncomplicated. More interesting were the 45 alarms heard while outside the patient’s room, where nurses face the complex decision of whether to interrupt their current tasks and respond or delay their response and assume the associated risk of nonresponse to a potentially deteriorating patient. Of the 45 alarms, nurses went into the room to evaluate the patient 15 times and, after doing so, reported that five of the 15 warranted in-person responses to address technical issues with the monitor, clinical issues, or patients’ comfort. Reassuring clinical contexts—such as presence of the medical team or family in the room and recent patient assessments—were the reasons most commonly provided to explain alarm nonresponse.
This study has two key limitations. First, the authors designed the study to observe nurses’ responses until thematic saturation was achieved. However, the small sample size (nine nurses, 45 out-of-room alarms) could raise questions about whether sufficient data were captured to make broadly generalizable conclusions, given the diverse range of patients, families, and clinical scenarios nurses encounter on an inpatient unit. Second, by instructing nurse participants to verbalize their rationale for response or nonresponse, investigators essentially asked nurses to override the “Type 1”, heuristic-based reasoning5 that research suggests regulates nursing responses to alarms when adapting to circumstances requiring high cognitive demand or a heavy workload.3 While innovative, it is possible that this approach prevented the investigators from fully achieving their stated objective of describing how bedside nurses think about and act upon alarms.
Nonetheless, the findings by Schondelmeyer and colleagues extend our emerging understanding of why alarm responses are disconcertingly slow. Nursing staff’s dismissal of monitor alarms that are discordant with a reassuring patient evaluation underscores the imperative to reduce nuisance alarms. Furthermore, the explicit statements justifying alarm nonresponse because of the presence of family members build upon prior findings of longer response times when family members are at the bedside3 and invite a provocative question: how would family members feel if they knew that they were being entrusted as a foundational component of safety monitoring in the hospital? In their recently published study conducted at the same hospital,6 Schondelmeyer’s team elicited perceptions that families are deeply concerned about staff nonresponse to alarms—as one nurse stated, parents “wonder what’s going on when no one comes in.” While there is a valuable role for integrating families into efforts to overcome threats to patient safety, as has been achieved with family error reporting7 and communication on family-centered rounds,8 this must occur in a structured, explicit, and deliberate manner, with families engaged as key stakeholders.
In summary, while Schondelmeyer and colleagues may not have exposed the depth of implicit thinking that governs nurses’ responses to alarms, they have highlighted the high-stakes decisions that nurses confront on a daily basis in an environment with exceedingly high alarm rates and low alarm actionability. The authors cite staff education among potential solutions to improve the safety of continuous monitoring, but such an intervention cannot be effective in a system that places impossible burdens on nurses. An openly family centered and multidisciplinary approach to reengineering the system for monitoring hospitalized children is needed to enable nurses to respond quickly and accurately to patients at risk of clinical deterioration.
Disclosures
The authors report no conflicts of interest.
1. Schondelmeyer AC, Brady PW, Goel VV, et al. Physiologic monitor alarm rates at 5 children’s hospitals. J Hosp Med. 2018;13(6):396-398. https://doi.org/10.12788/jhm.2918.
2. Bonafide CP, Lin R, Zander M, et al. Association between exposure to nonactionable physiologic monitor alarms and response time in a children’s hospital. J Hosp Med. 2015;10(6):345-351. https://doi.org/10.1002/jhm.2331.
3. Bonafide CP, Localio AR, Holmes JH, et al. Video analysis of factors associated with response time to physiologic monitor alarms in a children’s hospital. JAMA Pediatr. 2017;171(6):524-531. https://doi.org/10.1001/jamapediatrics.2016.5123.
4. Schondelmeyer A, Daraiseh NM, Allison B, et al. Nurse responses to physiologic monitor alarms on a general pediatric unit. J Hosp Med. 2019;14(10):602-606. https://doi.org/10.12788/jhm.3234.
5. Croskerry P. A universal model of diagnostic reasoning. Acad Med. 2009;84(8):1022-1028. https://doi.org/10.1097/ACM.0b013e3181ace703.
6. Schondelmeyer AC, Jenkins AM, Allison B, et al. Factors influencing use of continuous physiologic monitors for hospitalized pediatric patients. Hosp Pediatr. 2019;9(6):423-428. https://doi.org/10.1542/hpeds.2019-0007.
7. Khan A, Coffey M, Litterer KP, et al. Families as partners in hospital error and adverse event surveillance. JAMA Pediatr. 2017;171(4):372-381. https://doi.org/10.1001/jamapediatrics.2016.4812.
8. Khan A, Spector ND, Baird JD, et al. Patient safety after implementation of a coproduced family centered communication programme: multicenter before and after intervention study. BMJ. 2018;363:k4764. https://doi.org/10.1136/bmj.k4764.
1. Schondelmeyer AC, Brady PW, Goel VV, et al. Physiologic monitor alarm rates at 5 children’s hospitals. J Hosp Med. 2018;13(6):396-398. https://doi.org/10.12788/jhm.2918.
2. Bonafide CP, Lin R, Zander M, et al. Association between exposure to nonactionable physiologic monitor alarms and response time in a children’s hospital. J Hosp Med. 2015;10(6):345-351. https://doi.org/10.1002/jhm.2331.
3. Bonafide CP, Localio AR, Holmes JH, et al. Video analysis of factors associated with response time to physiologic monitor alarms in a children’s hospital. JAMA Pediatr. 2017;171(6):524-531. https://doi.org/10.1001/jamapediatrics.2016.5123.
4. Schondelmeyer A, Daraiseh NM, Allison B, et al. Nurse responses to physiologic monitor alarms on a general pediatric unit. J Hosp Med. 2019;14(10):602-606. https://doi.org/10.12788/jhm.3234.
5. Croskerry P. A universal model of diagnostic reasoning. Acad Med. 2009;84(8):1022-1028. https://doi.org/10.1097/ACM.0b013e3181ace703.
6. Schondelmeyer AC, Jenkins AM, Allison B, et al. Factors influencing use of continuous physiologic monitors for hospitalized pediatric patients. Hosp Pediatr. 2019;9(6):423-428. https://doi.org/10.1542/hpeds.2019-0007.
7. Khan A, Coffey M, Litterer KP, et al. Families as partners in hospital error and adverse event surveillance. JAMA Pediatr. 2017;171(4):372-381. https://doi.org/10.1001/jamapediatrics.2016.4812.
8. Khan A, Spector ND, Baird JD, et al. Patient safety after implementation of a coproduced family centered communication programme: multicenter before and after intervention study. BMJ. 2018;363:k4764. https://doi.org/10.1136/bmj.k4764.
© 2019 Society of Hospital Medicine
