User login
Lucas Franki is an associate editor for MDedge News, and has been with the company since 2014. He has a BA in English from Penn State University and is an Eagle Scout.
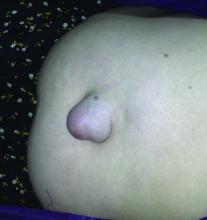
Umbilical hernia repair during pregnancy safe, but often serious
Umbilical hernia repair during pregnancy is rare and safe, but more than half of surgeries required incarceration or strangulation repair, according to Dr. I.N. Haskins and associates.
A total of 126 pregnant women underwent umbilical hernia repair from 2005 to 2014, according to data collected from the American College of Surgeons National Surgical Quality Improvement Program. All but six women underwent open surgery, and of these 120 patients, 71 had umbilical hernia incarceration or strangulation at the time of surgery.
“Additional studies are needed to determine the long-term recurrence rate of umbilical hernia repairs performed in pregnant patients and the effects of surgical intervention and approach on the fetus,” the investigators concluded.
Find the study in Hernia (doi: 10.1007/s10029-017-1633-8).
Umbilical hernia repair during pregnancy is rare and safe, but more than half of surgeries required incarceration or strangulation repair, according to Dr. I.N. Haskins and associates.
A total of 126 pregnant women underwent umbilical hernia repair from 2005 to 2014, according to data collected from the American College of Surgeons National Surgical Quality Improvement Program. All but six women underwent open surgery, and of these 120 patients, 71 had umbilical hernia incarceration or strangulation at the time of surgery.
“Additional studies are needed to determine the long-term recurrence rate of umbilical hernia repairs performed in pregnant patients and the effects of surgical intervention and approach on the fetus,” the investigators concluded.
Find the study in Hernia (doi: 10.1007/s10029-017-1633-8).
Umbilical hernia repair during pregnancy is rare and safe, but more than half of surgeries required incarceration or strangulation repair, according to Dr. I.N. Haskins and associates.
A total of 126 pregnant women underwent umbilical hernia repair from 2005 to 2014, according to data collected from the American College of Surgeons National Surgical Quality Improvement Program. All but six women underwent open surgery, and of these 120 patients, 71 had umbilical hernia incarceration or strangulation at the time of surgery.
“Additional studies are needed to determine the long-term recurrence rate of umbilical hernia repairs performed in pregnant patients and the effects of surgical intervention and approach on the fetus,” the investigators concluded.
Find the study in Hernia (doi: 10.1007/s10029-017-1633-8).
FROM HERNIA
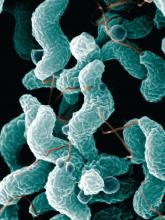
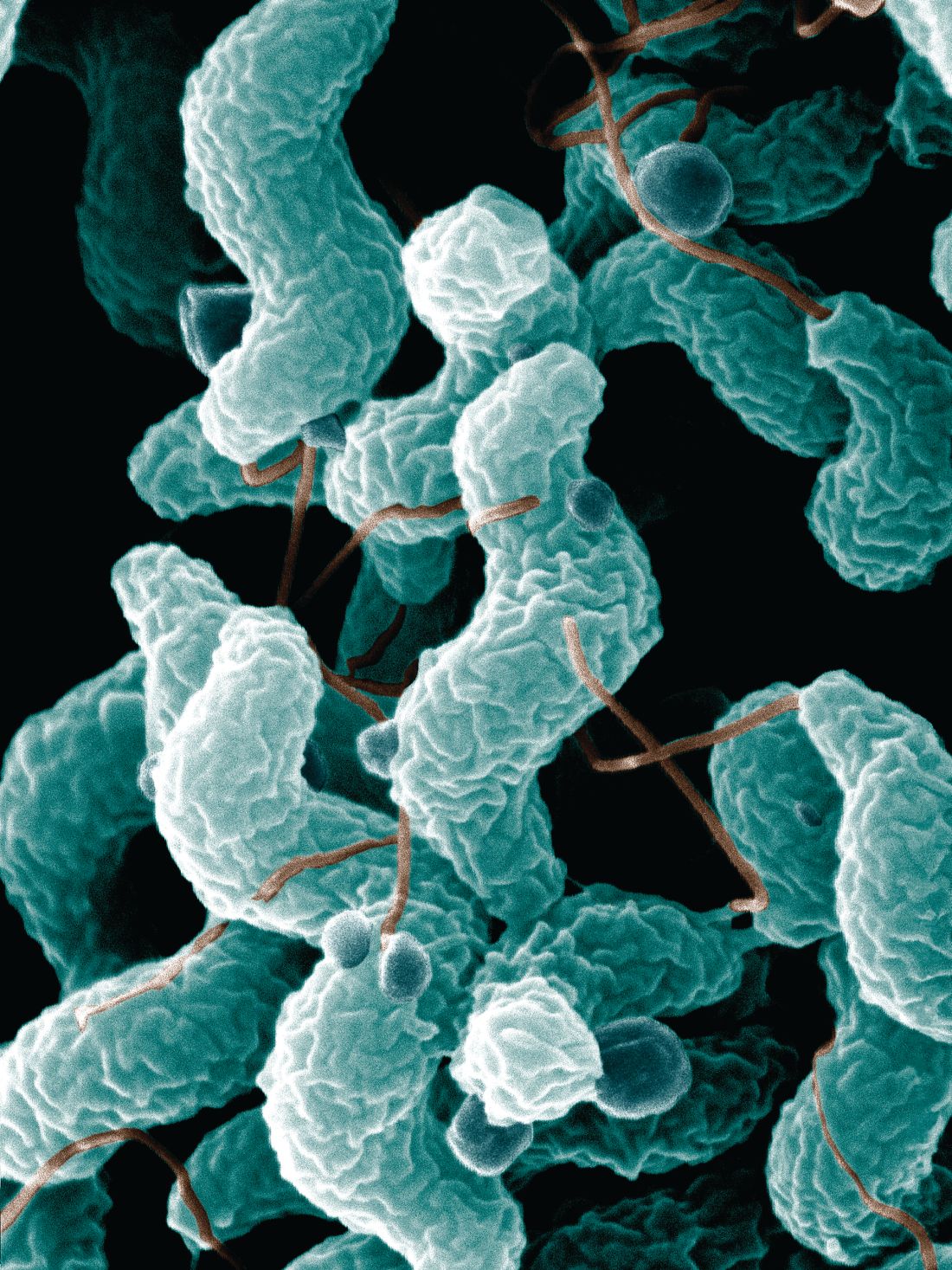
Campylobacteriosis incidence rises in U.S. from 2004 to 2012
Incidence of campylobacteriosis increased significantly in the United States from 2004 to 2012, according to Aimee Geissler, PhD, and her associates.
A total of 303,520 cases of campylobacteriosis were reported during the study period, with the average incidence rate growing from 10.5 cases per 100,000 persons during 2004-2006 to 12.7 cases per 100,000 persons during 2010-2012, an increase of 21%. The median number of Camplyobacter outbreaks doubled from 28 during 2004-2006 to 56 during 2010-2012; in total, 347 outbreaks were reported. Campylobacteriosis is the nation’s most common bacterial diarrheal illness.
The study findings “underscore the importance of standardizing national surveillance for campylobacteriosis, which is important in understanding the burden of infection, better describing geographic variations and differences among species, elucidating risk factors, and targeting prevention and control measures,” the investigators concluded.
Find the full study in Clinical Infectious Diseases (2017 Jul 20. doi: 10.1093/cid/cix624).
Incidence of campylobacteriosis increased significantly in the United States from 2004 to 2012, according to Aimee Geissler, PhD, and her associates.
A total of 303,520 cases of campylobacteriosis were reported during the study period, with the average incidence rate growing from 10.5 cases per 100,000 persons during 2004-2006 to 12.7 cases per 100,000 persons during 2010-2012, an increase of 21%. The median number of Camplyobacter outbreaks doubled from 28 during 2004-2006 to 56 during 2010-2012; in total, 347 outbreaks were reported. Campylobacteriosis is the nation’s most common bacterial diarrheal illness.
The study findings “underscore the importance of standardizing national surveillance for campylobacteriosis, which is important in understanding the burden of infection, better describing geographic variations and differences among species, elucidating risk factors, and targeting prevention and control measures,” the investigators concluded.
Find the full study in Clinical Infectious Diseases (2017 Jul 20. doi: 10.1093/cid/cix624).
Incidence of campylobacteriosis increased significantly in the United States from 2004 to 2012, according to Aimee Geissler, PhD, and her associates.
A total of 303,520 cases of campylobacteriosis were reported during the study period, with the average incidence rate growing from 10.5 cases per 100,000 persons during 2004-2006 to 12.7 cases per 100,000 persons during 2010-2012, an increase of 21%. The median number of Camplyobacter outbreaks doubled from 28 during 2004-2006 to 56 during 2010-2012; in total, 347 outbreaks were reported. Campylobacteriosis is the nation’s most common bacterial diarrheal illness.
The study findings “underscore the importance of standardizing national surveillance for campylobacteriosis, which is important in understanding the burden of infection, better describing geographic variations and differences among species, elucidating risk factors, and targeting prevention and control measures,” the investigators concluded.
Find the full study in Clinical Infectious Diseases (2017 Jul 20. doi: 10.1093/cid/cix624).
FROM CLINICAL INFECTIOUS DISEASES
Fujifilm issues recall to update ED-530XT duodenoscopes
Fujifilm has issued an Urgent Medical Device Correction and Removal notification for all ED-530XT duodenoscopes, according to a Safety Alert from the Food and Drug Administration.
The recall, initiated voluntarily by Fujifilm, includes replacement of the ED-530XT forceps elevator mechanism including the O-ring seal, replacement of the distal end cap, and new operation manuals. The FDA authorized the changes on July 21, 2017.
“Reprocessing is a detailed, multistep process to clean and disinfect or sterilize reusable devices. The FDA has been working with duodenoscope manufacturers as they modify and validate their reprocessing instructions to further enhance the safety margin of their devices and show with a high degree of assurance that their reprocessing instructions, when followed correctly, effectively clean and disinfect the duodenoscopes,” the FDA said in the press release.
Find the full Safety Alert on the FDA website.
The AGA Center for GI Innovation and Technology is committed to working towards zero device-associated infections and will keep you apprised of any future updates. Learn more at www.gastro.org/CGIT.
Fujifilm has issued an Urgent Medical Device Correction and Removal notification for all ED-530XT duodenoscopes, according to a Safety Alert from the Food and Drug Administration.
The recall, initiated voluntarily by Fujifilm, includes replacement of the ED-530XT forceps elevator mechanism including the O-ring seal, replacement of the distal end cap, and new operation manuals. The FDA authorized the changes on July 21, 2017.
“Reprocessing is a detailed, multistep process to clean and disinfect or sterilize reusable devices. The FDA has been working with duodenoscope manufacturers as they modify and validate their reprocessing instructions to further enhance the safety margin of their devices and show with a high degree of assurance that their reprocessing instructions, when followed correctly, effectively clean and disinfect the duodenoscopes,” the FDA said in the press release.
Find the full Safety Alert on the FDA website.
The AGA Center for GI Innovation and Technology is committed to working towards zero device-associated infections and will keep you apprised of any future updates. Learn more at www.gastro.org/CGIT.
Fujifilm has issued an Urgent Medical Device Correction and Removal notification for all ED-530XT duodenoscopes, according to a Safety Alert from the Food and Drug Administration.
The recall, initiated voluntarily by Fujifilm, includes replacement of the ED-530XT forceps elevator mechanism including the O-ring seal, replacement of the distal end cap, and new operation manuals. The FDA authorized the changes on July 21, 2017.
“Reprocessing is a detailed, multistep process to clean and disinfect or sterilize reusable devices. The FDA has been working with duodenoscope manufacturers as they modify and validate their reprocessing instructions to further enhance the safety margin of their devices and show with a high degree of assurance that their reprocessing instructions, when followed correctly, effectively clean and disinfect the duodenoscopes,” the FDA said in the press release.
Find the full Safety Alert on the FDA website.
The AGA Center for GI Innovation and Technology is committed to working towards zero device-associated infections and will keep you apprised of any future updates. Learn more at www.gastro.org/CGIT.
NLR useful for predicting 1-year mortality in PBC patients
An elevated baseline neutrophil-to-lymphocyte ratio (NLR) was associated with a poor 1-year mortality rate in hospitalized primary biliary cholangitis (PBC) patients, according to Lin Lin, MD, of Tianjin (China) Medical University General Hospital and the Tianjin Institute of Digestive Diseases and associates.
A retrospective analysis of 88 PBC patients hospitalized between June 2009 and January 2014 was performed for the study. NLR was a significant predictor of survival, with an odds ratio of 1.5, a sensitivity of 100%, and a specificity of 67.1%. A baseline NLR value of 2.18 was selected as the cutoff for 1-year mortality. Of the 33 patients above this value at initial hospitalization, 6 died, whereas none of the 55 patients below this value died.
The results of the retrospective study were confirmed in a prospective 1-year cohort that included 63 people with PBC. The patients with a baseline NLR of less than 2.18 had significantly longer survival times than those who had a baseline NLR of 2.18 or higher.
“NLR – an affordable, widely available and reproducible index – is closely related to short-term mortality in patients with PBC. Further studies are warranted to externally cross-validate our findings in other populations,” the investigators concluded.
Find the full study in BMJ Open (2017. doi: 10.1136/bmjopen-2016-015304).
An elevated baseline neutrophil-to-lymphocyte ratio (NLR) was associated with a poor 1-year mortality rate in hospitalized primary biliary cholangitis (PBC) patients, according to Lin Lin, MD, of Tianjin (China) Medical University General Hospital and the Tianjin Institute of Digestive Diseases and associates.
A retrospective analysis of 88 PBC patients hospitalized between June 2009 and January 2014 was performed for the study. NLR was a significant predictor of survival, with an odds ratio of 1.5, a sensitivity of 100%, and a specificity of 67.1%. A baseline NLR value of 2.18 was selected as the cutoff for 1-year mortality. Of the 33 patients above this value at initial hospitalization, 6 died, whereas none of the 55 patients below this value died.
The results of the retrospective study were confirmed in a prospective 1-year cohort that included 63 people with PBC. The patients with a baseline NLR of less than 2.18 had significantly longer survival times than those who had a baseline NLR of 2.18 or higher.
“NLR – an affordable, widely available and reproducible index – is closely related to short-term mortality in patients with PBC. Further studies are warranted to externally cross-validate our findings in other populations,” the investigators concluded.
Find the full study in BMJ Open (2017. doi: 10.1136/bmjopen-2016-015304).
An elevated baseline neutrophil-to-lymphocyte ratio (NLR) was associated with a poor 1-year mortality rate in hospitalized primary biliary cholangitis (PBC) patients, according to Lin Lin, MD, of Tianjin (China) Medical University General Hospital and the Tianjin Institute of Digestive Diseases and associates.
A retrospective analysis of 88 PBC patients hospitalized between June 2009 and January 2014 was performed for the study. NLR was a significant predictor of survival, with an odds ratio of 1.5, a sensitivity of 100%, and a specificity of 67.1%. A baseline NLR value of 2.18 was selected as the cutoff for 1-year mortality. Of the 33 patients above this value at initial hospitalization, 6 died, whereas none of the 55 patients below this value died.
The results of the retrospective study were confirmed in a prospective 1-year cohort that included 63 people with PBC. The patients with a baseline NLR of less than 2.18 had significantly longer survival times than those who had a baseline NLR of 2.18 or higher.
“NLR – an affordable, widely available and reproducible index – is closely related to short-term mortality in patients with PBC. Further studies are warranted to externally cross-validate our findings in other populations,” the investigators concluded.
Find the full study in BMJ Open (2017. doi: 10.1136/bmjopen-2016-015304).
FROM BMJ OPEN
Fujifilm issues recall to update ED-530XT duodenoscopes
Fujifilm has issued an Urgent Medical Device Correction and Removal notification for all ED-530XT duodenoscopes, according to a Safety Alert from the Food and Drug Administration.
The recall, initiated voluntarily by Fujifilm, includes replacement of the ED-530XT forceps elevator mechanism including the O-ring seal, replacement of the distal end cap, and new operation manuals. The FDA authorized the changes on July 21, 2017.
“Reprocessing is a detailed, multistep process to clean and disinfect or sterilize reusable devices. The FDA has been working with duodenoscope manufacturers as they modify and validate their reprocessing instructions to further enhance the safety margin of their devices and show with a high degree of assurance that their reprocessing instructions, when followed correctly, effectively clean and disinfect the duodenoscopes,” the FDA said in the press release.
Find the full Safety Alert on the FDA website.
Fujifilm has issued an Urgent Medical Device Correction and Removal notification for all ED-530XT duodenoscopes, according to a Safety Alert from the Food and Drug Administration.
The recall, initiated voluntarily by Fujifilm, includes replacement of the ED-530XT forceps elevator mechanism including the O-ring seal, replacement of the distal end cap, and new operation manuals. The FDA authorized the changes on July 21, 2017.
“Reprocessing is a detailed, multistep process to clean and disinfect or sterilize reusable devices. The FDA has been working with duodenoscope manufacturers as they modify and validate their reprocessing instructions to further enhance the safety margin of their devices and show with a high degree of assurance that their reprocessing instructions, when followed correctly, effectively clean and disinfect the duodenoscopes,” the FDA said in the press release.
Find the full Safety Alert on the FDA website.
Fujifilm has issued an Urgent Medical Device Correction and Removal notification for all ED-530XT duodenoscopes, according to a Safety Alert from the Food and Drug Administration.
The recall, initiated voluntarily by Fujifilm, includes replacement of the ED-530XT forceps elevator mechanism including the O-ring seal, replacement of the distal end cap, and new operation manuals. The FDA authorized the changes on July 21, 2017.
“Reprocessing is a detailed, multistep process to clean and disinfect or sterilize reusable devices. The FDA has been working with duodenoscope manufacturers as they modify and validate their reprocessing instructions to further enhance the safety margin of their devices and show with a high degree of assurance that their reprocessing instructions, when followed correctly, effectively clean and disinfect the duodenoscopes,” the FDA said in the press release.
Find the full Safety Alert on the FDA website.
Pegboard test useful in upper-extremity motor assessments in Parkinson’s
Pegboard tests are an effective way to assess the progression of upper-extremity motor deficits in patients with early-stage Parkinson’s disease (PD), according to a research letter from Sebastian Heitzel, PhD, and his associates.
As part of the prospective MODEP study, motor performance of early-stage PD patients, midstage PD patients, and a healthy control group were assessed biannually over a 4-year period using the pegboard test. E-PD and M-PD patients performed significantly worse on all pegboard tasks than did people in the control group.
Movement Disorder Society Unified Parkinson’s Disease Rating Scale part III scores worsened in E-PD patients, compared with controls, but slightly improved in M-PD patients.
“Pegboard is cost- and time-effective, does not require medically trained staff, and provides objective and reliable markers of upper-extremity motor deficits,” the investigators noted.
Find the full research letter in Movement Disorders (2017. doi: 10.1002/mds.27062).
Pegboard tests are an effective way to assess the progression of upper-extremity motor deficits in patients with early-stage Parkinson’s disease (PD), according to a research letter from Sebastian Heitzel, PhD, and his associates.
As part of the prospective MODEP study, motor performance of early-stage PD patients, midstage PD patients, and a healthy control group were assessed biannually over a 4-year period using the pegboard test. E-PD and M-PD patients performed significantly worse on all pegboard tasks than did people in the control group.
Movement Disorder Society Unified Parkinson’s Disease Rating Scale part III scores worsened in E-PD patients, compared with controls, but slightly improved in M-PD patients.
“Pegboard is cost- and time-effective, does not require medically trained staff, and provides objective and reliable markers of upper-extremity motor deficits,” the investigators noted.
Find the full research letter in Movement Disorders (2017. doi: 10.1002/mds.27062).
Pegboard tests are an effective way to assess the progression of upper-extremity motor deficits in patients with early-stage Parkinson’s disease (PD), according to a research letter from Sebastian Heitzel, PhD, and his associates.
As part of the prospective MODEP study, motor performance of early-stage PD patients, midstage PD patients, and a healthy control group were assessed biannually over a 4-year period using the pegboard test. E-PD and M-PD patients performed significantly worse on all pegboard tasks than did people in the control group.
Movement Disorder Society Unified Parkinson’s Disease Rating Scale part III scores worsened in E-PD patients, compared with controls, but slightly improved in M-PD patients.
“Pegboard is cost- and time-effective, does not require medically trained staff, and provides objective and reliable markers of upper-extremity motor deficits,” the investigators noted.
Find the full research letter in Movement Disorders (2017. doi: 10.1002/mds.27062).
FROM MOVEMENT DISORDERS
Clinical trial: Mesh Type in Ventral Hernia Repair
The Mesh Type in Ventral Hernia Repair trial is an interventional study currently recruiting patients scheduled for open ventral hernia repair.
Half of ventral hernia repairs utilize synthetic mesh, while the other half use biologic mesh. There is currently little solid evidence that one mesh type is better than the other, although the study investigators hypothesize that biologic mesh is superior to synthetic.
Patients will be included in the trial if they are scheduled for open ventral hernia repair at LBJ General Hospital in Houston and are at least 18 years old. Patients will be excluded if they have an active infection, are unlikely to survive the next 2 years, are individuals in whom a prosthetic would not normally be placed, or are unlikely to follow up.
The primary endpoint goal is zero complications 1 year after the operation. Secondary endpoint goals include patient-centered outcomes, cost, Dindo-Clavien complications (grades I-IV), and to be complication free 3 years after the operation.
Recruitment began on March 27, 2017, and the study is expected to include 50 people. The primary study endpoint will be completed March 31, 2019, with the full study being completed March 31, 2022.
Find more information at the study page on Clinicaltrials.gov.
The Mesh Type in Ventral Hernia Repair trial is an interventional study currently recruiting patients scheduled for open ventral hernia repair.
Half of ventral hernia repairs utilize synthetic mesh, while the other half use biologic mesh. There is currently little solid evidence that one mesh type is better than the other, although the study investigators hypothesize that biologic mesh is superior to synthetic.
Patients will be included in the trial if they are scheduled for open ventral hernia repair at LBJ General Hospital in Houston and are at least 18 years old. Patients will be excluded if they have an active infection, are unlikely to survive the next 2 years, are individuals in whom a prosthetic would not normally be placed, or are unlikely to follow up.
The primary endpoint goal is zero complications 1 year after the operation. Secondary endpoint goals include patient-centered outcomes, cost, Dindo-Clavien complications (grades I-IV), and to be complication free 3 years after the operation.
Recruitment began on March 27, 2017, and the study is expected to include 50 people. The primary study endpoint will be completed March 31, 2019, with the full study being completed March 31, 2022.
Find more information at the study page on Clinicaltrials.gov.
The Mesh Type in Ventral Hernia Repair trial is an interventional study currently recruiting patients scheduled for open ventral hernia repair.
Half of ventral hernia repairs utilize synthetic mesh, while the other half use biologic mesh. There is currently little solid evidence that one mesh type is better than the other, although the study investigators hypothesize that biologic mesh is superior to synthetic.
Patients will be included in the trial if they are scheduled for open ventral hernia repair at LBJ General Hospital in Houston and are at least 18 years old. Patients will be excluded if they have an active infection, are unlikely to survive the next 2 years, are individuals in whom a prosthetic would not normally be placed, or are unlikely to follow up.
The primary endpoint goal is zero complications 1 year after the operation. Secondary endpoint goals include patient-centered outcomes, cost, Dindo-Clavien complications (grades I-IV), and to be complication free 3 years after the operation.
Recruitment began on March 27, 2017, and the study is expected to include 50 people. The primary study endpoint will be completed March 31, 2019, with the full study being completed March 31, 2022.
Find more information at the study page on Clinicaltrials.gov.
SUMMARY FROM CLINICALTRIALS.GOV
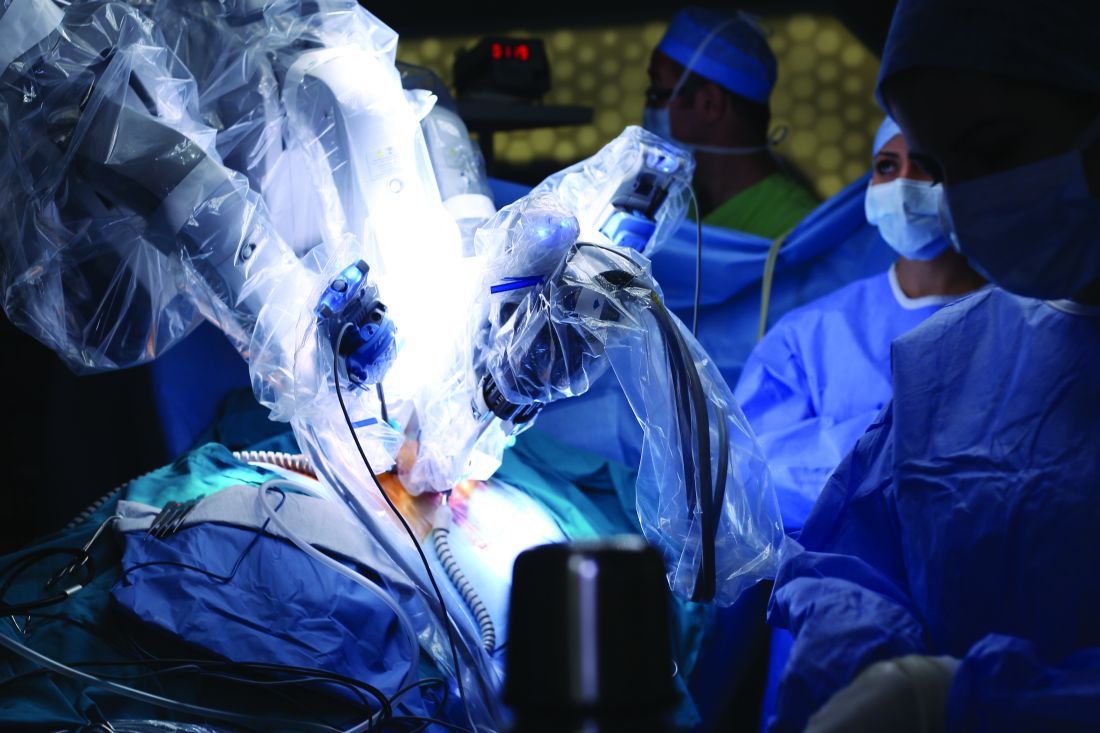
Robotic-assisted IHR causes fewer complications in obese patients
Obese people undergoing inguinal hernia repair experienced fewer complications when the surgery was robotic assisted, compared to open repairs, according to Ramachandra Kolachalam, MD, and his associates.
A total of 148 robotic-assisted repairs (RHRs) and 113 open repairs were included in the study. Of open repair (OHR) patients, 11.5% experienced postoperative complications post discharge, compared with only 2.7% of RHR patients. OHR patients also had lower rates of concomitant procedures (16.8% vs. 29.7%) and bilateral repairs (11.5% vs. 35.1%). Morbidity rates did not differ significantly between the groups.
“Robotic-assisted inguinal hernia repair could lead to increased acceptance of minimally invasive hernia repair with the associated clinical benefits to patients, including those who are obese with higher comorbidities and higher American Surgery Association scores. A prospective study of obesity in RHR is warranted to confirm our findings,” the investigators concluded.
Find the full study in Surgical Endoscopy (2017. doi: 10.1007/s00464-017-5665-z).
Obese people undergoing inguinal hernia repair experienced fewer complications when the surgery was robotic assisted, compared to open repairs, according to Ramachandra Kolachalam, MD, and his associates.
A total of 148 robotic-assisted repairs (RHRs) and 113 open repairs were included in the study. Of open repair (OHR) patients, 11.5% experienced postoperative complications post discharge, compared with only 2.7% of RHR patients. OHR patients also had lower rates of concomitant procedures (16.8% vs. 29.7%) and bilateral repairs (11.5% vs. 35.1%). Morbidity rates did not differ significantly between the groups.
“Robotic-assisted inguinal hernia repair could lead to increased acceptance of minimally invasive hernia repair with the associated clinical benefits to patients, including those who are obese with higher comorbidities and higher American Surgery Association scores. A prospective study of obesity in RHR is warranted to confirm our findings,” the investigators concluded.
Find the full study in Surgical Endoscopy (2017. doi: 10.1007/s00464-017-5665-z).
Obese people undergoing inguinal hernia repair experienced fewer complications when the surgery was robotic assisted, compared to open repairs, according to Ramachandra Kolachalam, MD, and his associates.
A total of 148 robotic-assisted repairs (RHRs) and 113 open repairs were included in the study. Of open repair (OHR) patients, 11.5% experienced postoperative complications post discharge, compared with only 2.7% of RHR patients. OHR patients also had lower rates of concomitant procedures (16.8% vs. 29.7%) and bilateral repairs (11.5% vs. 35.1%). Morbidity rates did not differ significantly between the groups.
“Robotic-assisted inguinal hernia repair could lead to increased acceptance of minimally invasive hernia repair with the associated clinical benefits to patients, including those who are obese with higher comorbidities and higher American Surgery Association scores. A prospective study of obesity in RHR is warranted to confirm our findings,” the investigators concluded.
Find the full study in Surgical Endoscopy (2017. doi: 10.1007/s00464-017-5665-z).
FROM SURGICAL ENDOSCOPY
FDA approves new panel to identify mCRC patients for panitumumab treatment
The Food and Drug Administration has approved the Praxis Extended RAS Panel for the identification of metastatic colorectal cancer patients who can be treated with panitumumab.
The Praxis Extended RAS Panel is able to detect 56 specific mutations in the RAS genes of mCRC patients, and is the first next generation sequencing test approved by the FDA capable of testing more than one RAS gene mutation. If RAS mutations are not detected, then panitumumab is indicated, and if a mutation is detected, panitumumab is not indicated, according to the FDA statement.
“Panitumumab’s product labeling has been modified to align the indication for panitumumab and intended use for the Praxis Extended RAS Panel,” the FDA noted.
The Food and Drug Administration has approved the Praxis Extended RAS Panel for the identification of metastatic colorectal cancer patients who can be treated with panitumumab.
The Praxis Extended RAS Panel is able to detect 56 specific mutations in the RAS genes of mCRC patients, and is the first next generation sequencing test approved by the FDA capable of testing more than one RAS gene mutation. If RAS mutations are not detected, then panitumumab is indicated, and if a mutation is detected, panitumumab is not indicated, according to the FDA statement.
“Panitumumab’s product labeling has been modified to align the indication for panitumumab and intended use for the Praxis Extended RAS Panel,” the FDA noted.
The Food and Drug Administration has approved the Praxis Extended RAS Panel for the identification of metastatic colorectal cancer patients who can be treated with panitumumab.
The Praxis Extended RAS Panel is able to detect 56 specific mutations in the RAS genes of mCRC patients, and is the first next generation sequencing test approved by the FDA capable of testing more than one RAS gene mutation. If RAS mutations are not detected, then panitumumab is indicated, and if a mutation is detected, panitumumab is not indicated, according to the FDA statement.
“Panitumumab’s product labeling has been modified to align the indication for panitumumab and intended use for the Praxis Extended RAS Panel,” the FDA noted.
FDA approves betrixaban for VTE prophylaxis
Betrixaban, a factor Xa inhibitor, has been approved for the prophylaxis of venous thromboembolism (VTE) in at-risk adult patients hospitalized with an acute illness, according to an announcement from the Food and Drug Administration.
Approval was based on results from a randomized, double-blind clinical trial in which over 7,000 hospitalized patients at risk for VTE received either extended-duration betrixaban (35-42 days) or short duration enoxaparin (6-14 days), a low molecular weight heparin administered subcutaneously. The rate of deep vein thrombosis, nonfatal pulmonary embolism, or VTE-related death was 4.4% among patients receiving betrixaban and 6% among patients receiving enoxaparin (relative risk, 0.75; 95% confidence interval: 0.61, 0.91).
The recommended dosage for betrixaban is 80 mg per day for 35-42 days at the same time every day with food, after a dose of 160 mg on the first day of treatment.
Betrixaban will be marketed as Bevyxxa by Portola.
Find the full FDA announcement and prescribing information on the FDA website.
Betrixaban, a factor Xa inhibitor, has been approved for the prophylaxis of venous thromboembolism (VTE) in at-risk adult patients hospitalized with an acute illness, according to an announcement from the Food and Drug Administration.
Approval was based on results from a randomized, double-blind clinical trial in which over 7,000 hospitalized patients at risk for VTE received either extended-duration betrixaban (35-42 days) or short duration enoxaparin (6-14 days), a low molecular weight heparin administered subcutaneously. The rate of deep vein thrombosis, nonfatal pulmonary embolism, or VTE-related death was 4.4% among patients receiving betrixaban and 6% among patients receiving enoxaparin (relative risk, 0.75; 95% confidence interval: 0.61, 0.91).
The recommended dosage for betrixaban is 80 mg per day for 35-42 days at the same time every day with food, after a dose of 160 mg on the first day of treatment.
Betrixaban will be marketed as Bevyxxa by Portola.
Find the full FDA announcement and prescribing information on the FDA website.
Betrixaban, a factor Xa inhibitor, has been approved for the prophylaxis of venous thromboembolism (VTE) in at-risk adult patients hospitalized with an acute illness, according to an announcement from the Food and Drug Administration.
Approval was based on results from a randomized, double-blind clinical trial in which over 7,000 hospitalized patients at risk for VTE received either extended-duration betrixaban (35-42 days) or short duration enoxaparin (6-14 days), a low molecular weight heparin administered subcutaneously. The rate of deep vein thrombosis, nonfatal pulmonary embolism, or VTE-related death was 4.4% among patients receiving betrixaban and 6% among patients receiving enoxaparin (relative risk, 0.75; 95% confidence interval: 0.61, 0.91).
The recommended dosage for betrixaban is 80 mg per day for 35-42 days at the same time every day with food, after a dose of 160 mg on the first day of treatment.
Betrixaban will be marketed as Bevyxxa by Portola.
Find the full FDA announcement and prescribing information on the FDA website.