User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Vitamin D supplements do not lower risk of fractures
compared with placebo, according to results from an ancillary study of the Vitamin D and Omega-3 Trial (VITAL).
The data showed that taking 2,000 IU of supplemental vitamin D each day without coadministered calcium did not have a significant effect on nonvertebral fractures (hazard ratio, 0.97; P = .50), hip fractures (HR, 1.01; P = .96), or total fractures (HR, 0.98; P = .70), compared with taking placebo, among individuals who did not have osteoporosis, vitamin D deficiency, or low bone mass, report Meryl S. LeBoff, MD, a professor of medicine at Harvard Medical School and chief of the calcium and bone section at Brigham and Women’s Hospital, both in Boston, and colleagues.
The findings were published online in the New England Journal of Medicine.
Prior randomized, controlled trials have presented conflicting findings. Some have shown that there is some benefit to supplemental vitamin D, whereas others have shown no effect or even harm with regard to risk of fractures, Dr. LeBoff noted.
“Because of the conflicting data at the time, we tested this hypothesis in an effort to advance science and understanding of the effects of vitamin D on bone. In a previous study, we did not see an effect of supplemental vitamin D on bone density in a subcohort from the VITAL trial,” Dr. LeBoff said in an interview.
“We previously reported that vitamin D, about 2,000 units per day, did not increase bone density, nor did it affect bone structure, according to PQCT [peripheral quantitative CT]. So that was an indicator that since bone density is a surrogate marker of fractures, there may not be an effect on fractures,” she added.
These results should dispel any idea that vitamin D alone could significantly reduce fracture rates in the general population, noted Steven R. Cummings, MD, of the University of California, San Francisco, and Clifford Rosen, MD, of Maine Medical Center Research Institute, Scarborough, in an accompanying editorial.
“Adding those findings to previous reports from VITAL and other trials showing the lack of an effect for preventing numerous conditions suggests that providers should stop screening for 25-hydroxyvitamin D levels or recommending vitamin D supplements, and people should stop taking vitamin D supplements to prevent major diseases or extend life,” the editorialists wrote.
The researchers assessed 25,871 participants from all 50 states during a median follow-up time of 5.3 years. Participants were randomly assigned in a 1:1 ratio to receive placebo or vitamin D.
The mean age of the participants was 67.1 years; 50.6% of the study cohort were women, and 20.2% of the cohort were Black. Participants did not have low bone mass, vitamin D deficiency, or osteoporosis.
Participants agreed not to supplement their dietary intake with more than 1,200 mg of calcium each day and no more than 800 IU of vitamin D each day.
Participants filled out detailed surveys to evaluate baseline prescription drug use, demographic factors, medical history, and the consumption of supplements, such as fish oil, calcium, and vitamin D, during the run-in stage. Yearly surveys were used to assess side effects, adherence to the investigation protocol, falls, fractures, physical activity, osteoporosis and associated risk factors, onset of major illness, and the use of nontrial prescription drugs and supplements, such as vitamin D and calcium.
The researchers adjudicated incident fracture data using a centralized medical record review. To approximate the therapeutic effect in intention-to-treat analyses, they used proportional-hazard models.
Notably, outcomes were similar for the placebo and vitamin D groups with regard to incident kidney stones and hypercalcemia.
The effect of vitamin D supplementation was not modified by baseline parameters such as race or ethnicity, sex, body mass index, age, or blood 25-hydroxyvitamin D levels.
Dr. Cummings and Dr. Rosen pointed out that these findings, along with other VITAL trial data, show that no subgroups classified on the basis of baseline 25-hydroxyvitamin D levels, including those with levels less than 20 ng/mL, benefited from vitamin supplementation.
“There is no justification for measuring 25-hydroxyvitamin D in the general population or treating to a target serum level. A 25-hydroxyvitamin D level might be a useful diagnostic test for some patients with conditions that may be due to or that may cause severe deficiency,” the editorialists noted.
Except with regard to select patients, such as individuals living in nursing homes who have limited sun exposure, the use of the terms “vitamin D deficiency” and “vitamin D “insufficiency” should now be reevaluated, Dr. Rosen and Dr. Cummings wrote.
The study’s limitations include its assessment of only one dosage of vitamin D supplementation and a lack of adjustment for multiplicity, exploratory, parent trial, or secondary endpoints, the researchers noted.
The number of participants who had vitamin D deficiency was limited, owing to ethical and feasibility concerns regarding these patients. The data are not generalizable to individuals who are older and institutionalized or those who have osteomalacia or osteoporosis, the researchers wrote.
Expert commentary
“The interpretation of this [study] to me is that vitamin D is not for everybody,” said Baha Arafah, MD, professor of medicine at Case Western Reserve University and chief of the division of endocrinology at University Hospital, both in Cleveland, who was not involved in the study.
“This is not the final word; I would suggest that you don’t throw vitamin D at everybody. I would use markers of bone formation as a better measure to determine whether they need vitamin D or not, specifically looking at parathyroid hormone,” Dr. Arafah said in an interview.
Dr. Arafah pointed out that these data do not mean that clinicians should stop thinking about vitamin D altogether. “I think that would be the wrong message to read. If you read through the article, you will find that there are people who do need vitamin D; people who are deficient do need vitamin D. There’s no question that excessive or extreme vitamin D deficiency can lead to other things, specifically, osteomalacia, weak bones, [and] poor mineralization, so we are not totally out of the woods at this time.”
The ancillary study of the VITAL trial was sponsored by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Pharmavite donated the vitamin D 3 supplements used in the trial. Dr. LeBoff reported that she holds stock in Amgen. Cummings reported receiving personal fees and nonfinancial support from Amgen outside the submitted work. Dr. Rosen is associate editor of the New England Journal of Medicine. Dr. Arafah reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
compared with placebo, according to results from an ancillary study of the Vitamin D and Omega-3 Trial (VITAL).
The data showed that taking 2,000 IU of supplemental vitamin D each day without coadministered calcium did not have a significant effect on nonvertebral fractures (hazard ratio, 0.97; P = .50), hip fractures (HR, 1.01; P = .96), or total fractures (HR, 0.98; P = .70), compared with taking placebo, among individuals who did not have osteoporosis, vitamin D deficiency, or low bone mass, report Meryl S. LeBoff, MD, a professor of medicine at Harvard Medical School and chief of the calcium and bone section at Brigham and Women’s Hospital, both in Boston, and colleagues.
The findings were published online in the New England Journal of Medicine.
Prior randomized, controlled trials have presented conflicting findings. Some have shown that there is some benefit to supplemental vitamin D, whereas others have shown no effect or even harm with regard to risk of fractures, Dr. LeBoff noted.
“Because of the conflicting data at the time, we tested this hypothesis in an effort to advance science and understanding of the effects of vitamin D on bone. In a previous study, we did not see an effect of supplemental vitamin D on bone density in a subcohort from the VITAL trial,” Dr. LeBoff said in an interview.
“We previously reported that vitamin D, about 2,000 units per day, did not increase bone density, nor did it affect bone structure, according to PQCT [peripheral quantitative CT]. So that was an indicator that since bone density is a surrogate marker of fractures, there may not be an effect on fractures,” she added.
These results should dispel any idea that vitamin D alone could significantly reduce fracture rates in the general population, noted Steven R. Cummings, MD, of the University of California, San Francisco, and Clifford Rosen, MD, of Maine Medical Center Research Institute, Scarborough, in an accompanying editorial.
“Adding those findings to previous reports from VITAL and other trials showing the lack of an effect for preventing numerous conditions suggests that providers should stop screening for 25-hydroxyvitamin D levels or recommending vitamin D supplements, and people should stop taking vitamin D supplements to prevent major diseases or extend life,” the editorialists wrote.
The researchers assessed 25,871 participants from all 50 states during a median follow-up time of 5.3 years. Participants were randomly assigned in a 1:1 ratio to receive placebo or vitamin D.
The mean age of the participants was 67.1 years; 50.6% of the study cohort were women, and 20.2% of the cohort were Black. Participants did not have low bone mass, vitamin D deficiency, or osteoporosis.
Participants agreed not to supplement their dietary intake with more than 1,200 mg of calcium each day and no more than 800 IU of vitamin D each day.
Participants filled out detailed surveys to evaluate baseline prescription drug use, demographic factors, medical history, and the consumption of supplements, such as fish oil, calcium, and vitamin D, during the run-in stage. Yearly surveys were used to assess side effects, adherence to the investigation protocol, falls, fractures, physical activity, osteoporosis and associated risk factors, onset of major illness, and the use of nontrial prescription drugs and supplements, such as vitamin D and calcium.
The researchers adjudicated incident fracture data using a centralized medical record review. To approximate the therapeutic effect in intention-to-treat analyses, they used proportional-hazard models.
Notably, outcomes were similar for the placebo and vitamin D groups with regard to incident kidney stones and hypercalcemia.
The effect of vitamin D supplementation was not modified by baseline parameters such as race or ethnicity, sex, body mass index, age, or blood 25-hydroxyvitamin D levels.
Dr. Cummings and Dr. Rosen pointed out that these findings, along with other VITAL trial data, show that no subgroups classified on the basis of baseline 25-hydroxyvitamin D levels, including those with levels less than 20 ng/mL, benefited from vitamin supplementation.
“There is no justification for measuring 25-hydroxyvitamin D in the general population or treating to a target serum level. A 25-hydroxyvitamin D level might be a useful diagnostic test for some patients with conditions that may be due to or that may cause severe deficiency,” the editorialists noted.
Except with regard to select patients, such as individuals living in nursing homes who have limited sun exposure, the use of the terms “vitamin D deficiency” and “vitamin D “insufficiency” should now be reevaluated, Dr. Rosen and Dr. Cummings wrote.
The study’s limitations include its assessment of only one dosage of vitamin D supplementation and a lack of adjustment for multiplicity, exploratory, parent trial, or secondary endpoints, the researchers noted.
The number of participants who had vitamin D deficiency was limited, owing to ethical and feasibility concerns regarding these patients. The data are not generalizable to individuals who are older and institutionalized or those who have osteomalacia or osteoporosis, the researchers wrote.
Expert commentary
“The interpretation of this [study] to me is that vitamin D is not for everybody,” said Baha Arafah, MD, professor of medicine at Case Western Reserve University and chief of the division of endocrinology at University Hospital, both in Cleveland, who was not involved in the study.
“This is not the final word; I would suggest that you don’t throw vitamin D at everybody. I would use markers of bone formation as a better measure to determine whether they need vitamin D or not, specifically looking at parathyroid hormone,” Dr. Arafah said in an interview.
Dr. Arafah pointed out that these data do not mean that clinicians should stop thinking about vitamin D altogether. “I think that would be the wrong message to read. If you read through the article, you will find that there are people who do need vitamin D; people who are deficient do need vitamin D. There’s no question that excessive or extreme vitamin D deficiency can lead to other things, specifically, osteomalacia, weak bones, [and] poor mineralization, so we are not totally out of the woods at this time.”
The ancillary study of the VITAL trial was sponsored by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Pharmavite donated the vitamin D 3 supplements used in the trial. Dr. LeBoff reported that she holds stock in Amgen. Cummings reported receiving personal fees and nonfinancial support from Amgen outside the submitted work. Dr. Rosen is associate editor of the New England Journal of Medicine. Dr. Arafah reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
compared with placebo, according to results from an ancillary study of the Vitamin D and Omega-3 Trial (VITAL).
The data showed that taking 2,000 IU of supplemental vitamin D each day without coadministered calcium did not have a significant effect on nonvertebral fractures (hazard ratio, 0.97; P = .50), hip fractures (HR, 1.01; P = .96), or total fractures (HR, 0.98; P = .70), compared with taking placebo, among individuals who did not have osteoporosis, vitamin D deficiency, or low bone mass, report Meryl S. LeBoff, MD, a professor of medicine at Harvard Medical School and chief of the calcium and bone section at Brigham and Women’s Hospital, both in Boston, and colleagues.
The findings were published online in the New England Journal of Medicine.
Prior randomized, controlled trials have presented conflicting findings. Some have shown that there is some benefit to supplemental vitamin D, whereas others have shown no effect or even harm with regard to risk of fractures, Dr. LeBoff noted.
“Because of the conflicting data at the time, we tested this hypothesis in an effort to advance science and understanding of the effects of vitamin D on bone. In a previous study, we did not see an effect of supplemental vitamin D on bone density in a subcohort from the VITAL trial,” Dr. LeBoff said in an interview.
“We previously reported that vitamin D, about 2,000 units per day, did not increase bone density, nor did it affect bone structure, according to PQCT [peripheral quantitative CT]. So that was an indicator that since bone density is a surrogate marker of fractures, there may not be an effect on fractures,” she added.
These results should dispel any idea that vitamin D alone could significantly reduce fracture rates in the general population, noted Steven R. Cummings, MD, of the University of California, San Francisco, and Clifford Rosen, MD, of Maine Medical Center Research Institute, Scarborough, in an accompanying editorial.
“Adding those findings to previous reports from VITAL and other trials showing the lack of an effect for preventing numerous conditions suggests that providers should stop screening for 25-hydroxyvitamin D levels or recommending vitamin D supplements, and people should stop taking vitamin D supplements to prevent major diseases or extend life,” the editorialists wrote.
The researchers assessed 25,871 participants from all 50 states during a median follow-up time of 5.3 years. Participants were randomly assigned in a 1:1 ratio to receive placebo or vitamin D.
The mean age of the participants was 67.1 years; 50.6% of the study cohort were women, and 20.2% of the cohort were Black. Participants did not have low bone mass, vitamin D deficiency, or osteoporosis.
Participants agreed not to supplement their dietary intake with more than 1,200 mg of calcium each day and no more than 800 IU of vitamin D each day.
Participants filled out detailed surveys to evaluate baseline prescription drug use, demographic factors, medical history, and the consumption of supplements, such as fish oil, calcium, and vitamin D, during the run-in stage. Yearly surveys were used to assess side effects, adherence to the investigation protocol, falls, fractures, physical activity, osteoporosis and associated risk factors, onset of major illness, and the use of nontrial prescription drugs and supplements, such as vitamin D and calcium.
The researchers adjudicated incident fracture data using a centralized medical record review. To approximate the therapeutic effect in intention-to-treat analyses, they used proportional-hazard models.
Notably, outcomes were similar for the placebo and vitamin D groups with regard to incident kidney stones and hypercalcemia.
The effect of vitamin D supplementation was not modified by baseline parameters such as race or ethnicity, sex, body mass index, age, or blood 25-hydroxyvitamin D levels.
Dr. Cummings and Dr. Rosen pointed out that these findings, along with other VITAL trial data, show that no subgroups classified on the basis of baseline 25-hydroxyvitamin D levels, including those with levels less than 20 ng/mL, benefited from vitamin supplementation.
“There is no justification for measuring 25-hydroxyvitamin D in the general population or treating to a target serum level. A 25-hydroxyvitamin D level might be a useful diagnostic test for some patients with conditions that may be due to or that may cause severe deficiency,” the editorialists noted.
Except with regard to select patients, such as individuals living in nursing homes who have limited sun exposure, the use of the terms “vitamin D deficiency” and “vitamin D “insufficiency” should now be reevaluated, Dr. Rosen and Dr. Cummings wrote.
The study’s limitations include its assessment of only one dosage of vitamin D supplementation and a lack of adjustment for multiplicity, exploratory, parent trial, or secondary endpoints, the researchers noted.
The number of participants who had vitamin D deficiency was limited, owing to ethical and feasibility concerns regarding these patients. The data are not generalizable to individuals who are older and institutionalized or those who have osteomalacia or osteoporosis, the researchers wrote.
Expert commentary
“The interpretation of this [study] to me is that vitamin D is not for everybody,” said Baha Arafah, MD, professor of medicine at Case Western Reserve University and chief of the division of endocrinology at University Hospital, both in Cleveland, who was not involved in the study.
“This is not the final word; I would suggest that you don’t throw vitamin D at everybody. I would use markers of bone formation as a better measure to determine whether they need vitamin D or not, specifically looking at parathyroid hormone,” Dr. Arafah said in an interview.
Dr. Arafah pointed out that these data do not mean that clinicians should stop thinking about vitamin D altogether. “I think that would be the wrong message to read. If you read through the article, you will find that there are people who do need vitamin D; people who are deficient do need vitamin D. There’s no question that excessive or extreme vitamin D deficiency can lead to other things, specifically, osteomalacia, weak bones, [and] poor mineralization, so we are not totally out of the woods at this time.”
The ancillary study of the VITAL trial was sponsored by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Pharmavite donated the vitamin D 3 supplements used in the trial. Dr. LeBoff reported that she holds stock in Amgen. Cummings reported receiving personal fees and nonfinancial support from Amgen outside the submitted work. Dr. Rosen is associate editor of the New England Journal of Medicine. Dr. Arafah reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Medicare advantage tied to less use of pricey diabetes drugs
U.S. Medicare beneficiaries with type 2 diabetes who had health coverage through a Medicare Advantage (MA) plan received treatment with an sodium-glucose cotransporter 2 inhibitor or glucagonlike peptide–1 receptor agonist significantly less often than patients with traditional fee-for-service (FFS) Medicare coverage in 2014-2019, according to a study of more than 411,000 patients.
concluded Utibe R. Essien, MD, and coauthors in a study published in Diabetes Care.
The report comes as the U.S. Congress is looking closely at the MA program and evidence that insurance companies that provide these policies sometimes impose inappropriate barriers on enrolled beneficiaries by denying or limiting access to treatments and interventions in ways that run counter to Medicare’s coverage policies.
According to Representative Diana DeGette (D-Colo.), who chaired a hearing on MA plans on June 28 by the House of Representatives’ Energy and Commerce Subcommittee on Oversight and Investigations, beneficiaries who are covered through an MA plan “do not always get the care that they are entitled to.”
The study by Dr. Essien and colleagues also documents some positives of care delivered through MA plans for patients with type 2 diabetes, compared with what FFS Medicare beneficiaries generally receive, such as significantly higher rates of screening for nephropathy and ophthalmologic disorders, and foot examinations.
But the apparently dampened use of SGLT2 inhibitors and GLP-1 receptor agonists among MA beneficiaries stand out as notable shortcomings, Dr. Essien maintained.
Cost containment may limit use
“The differences in health outcomes and in treatments in MA plans are important to highlight,” Dr. Essien said in an interview. “We worry that the cost-containment challenges [associated with MA plans] may be limiting use of these newer treatments.”
The study was based on 2014-2019 data from the Diabetes Collaborative Registry, which collects information from more than 5,000 U.S. clinicians whose practices include patients with diabetes, as well as claims data recorded by the Centers for Medicare and Medicaid Services during 2014-2017.
The main analysis focused on 345,911 Medicare beneficiaries with diabetes, which included 34% with MA coverage and 66% with FFS coverage. The two subgroups had similar ages, about 75 years old, and roughly half were women in both subgroups. The rate at which both subgroups received statin treatment was nearly the same: 72% for those with MA coverage and 71% for those with FFS Medicare.
But MA beneficiaries differed from those with FFS coverage in several other ways. MA beneficiaries had a higher prevalence of Medicaid eligibility than the FFS group (20% vs 12%) and lower rates of consultations with cardiologists (41% vs. 45%) or endocrinologists (7% vs. 10%).
Some of the positive differences in the care received by MA beneficiaries, compared with FFS beneficiaries, after adjustment for potential clinical and sociodemographic confounders, included:
- Screening for nephropathy, at a significant 14% higher relative rate.
- Screening for ophthalmologic disorders, at a significant 8% higher relative rate.
- Undergoing a diabetic foot examination, at a significant 13% higher relative rate.
- Receiving smoking-cessation counseling, at a significant 5% higher relative rate.
- Receiving treatment with an ACE inhibitor or angiotensin-receptor blocker (87% vs. 81%).
- More consistently receiving treatment with metformin, with rates of 72% versus 69% in 2017.
However, these positive differences were accompanied by these relative shortcomings for those with MA, compared with FFS coverage:
- Lower rates of treatment with an SGLT2 inhibitor (5.4% vs. 6.7%), a significant 9% relative difference after adjustment.
- Lower rates of treatment with a GLP-1 agonist (6.9% vs. 9.0%), a significant 20% relative difference after adjustment.
- Higher average levels of LDL cholesterol (81.5 vs. 78.9 mg/dL), a significantly higher average hemoglobin A1c level (7.1% vs. 7.0%), and a trend toward a lower prevalence of blood pressure control (70.3% vs. 71.5%).
Researchers also highlight that the lower rate at which people with MA coverage received SGLT2 inhibitors or GLP-1 agonists was consistent in patients with established cardiovascular or kidney disease, for whom these agents are particularly recommended.
In addition, a secondary analysis of data for another 65,000 Medicare beneficiaries in 2018 and 2019 showed the disparity in use of agents from these two drug classes continued.
Low systemic use of SGLT2 inhibitors, GLP-1 agonists
Dr. Essien acknowledged that, even in people with FFS Medicare coverage, use of SGLT2 inhibitors and GLP-1 agonists was low, but the difference between those with MA coverage is “important.”
Researchers offered four factors that might drive reduced prescribing of agents from these two classes for patients with type 2 diabetes with MA coverage: cost-containment strategies put in place by MA plans; the lower rate of consultations with specialists (cardiologists and endocrinologists); possible exclusion of clinicians from MA provider networks who tend to prescribe these higher-price agents; and lower household incomes of people with MA plans, which may lead to cost-related nonadherence.
Most SGLT2 inhibitors have an average retail cost of about $6,000/year, and some GLP-1 agonists cost more than $10,000/year.
In general, MA coverage includes more oversight of care and its cost than occurs with FFS coverage, noted Dr. Essien, an internal medicine physician at the University of Pittsburgh and a researcher at the Center for Health Equity Research and Promotion of the VA Pittsburgh Healthcare System.
“Incentives for using these more expensive treatments may not be there in MA plans,” he explained. Overcoming cost-related barriers is a challenge that will require “bold policy changes,” as well as better education of clinicians so they make correct treatment decisions, and of patients to resolve possible concerns about treatment safety.
Rep. DeGette hinted during her remarks at the June hearing that policy changes may be coming from Congress.
“Our seniors and their doctors should not be required to jump through numerous hoops to get coverage for straightforward and medically necessary procedures,” she said.
The study received no commercial funding. Dr. Essien reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
U.S. Medicare beneficiaries with type 2 diabetes who had health coverage through a Medicare Advantage (MA) plan received treatment with an sodium-glucose cotransporter 2 inhibitor or glucagonlike peptide–1 receptor agonist significantly less often than patients with traditional fee-for-service (FFS) Medicare coverage in 2014-2019, according to a study of more than 411,000 patients.
concluded Utibe R. Essien, MD, and coauthors in a study published in Diabetes Care.
The report comes as the U.S. Congress is looking closely at the MA program and evidence that insurance companies that provide these policies sometimes impose inappropriate barriers on enrolled beneficiaries by denying or limiting access to treatments and interventions in ways that run counter to Medicare’s coverage policies.
According to Representative Diana DeGette (D-Colo.), who chaired a hearing on MA plans on June 28 by the House of Representatives’ Energy and Commerce Subcommittee on Oversight and Investigations, beneficiaries who are covered through an MA plan “do not always get the care that they are entitled to.”
The study by Dr. Essien and colleagues also documents some positives of care delivered through MA plans for patients with type 2 diabetes, compared with what FFS Medicare beneficiaries generally receive, such as significantly higher rates of screening for nephropathy and ophthalmologic disorders, and foot examinations.
But the apparently dampened use of SGLT2 inhibitors and GLP-1 receptor agonists among MA beneficiaries stand out as notable shortcomings, Dr. Essien maintained.
Cost containment may limit use
“The differences in health outcomes and in treatments in MA plans are important to highlight,” Dr. Essien said in an interview. “We worry that the cost-containment challenges [associated with MA plans] may be limiting use of these newer treatments.”
The study was based on 2014-2019 data from the Diabetes Collaborative Registry, which collects information from more than 5,000 U.S. clinicians whose practices include patients with diabetes, as well as claims data recorded by the Centers for Medicare and Medicaid Services during 2014-2017.
The main analysis focused on 345,911 Medicare beneficiaries with diabetes, which included 34% with MA coverage and 66% with FFS coverage. The two subgroups had similar ages, about 75 years old, and roughly half were women in both subgroups. The rate at which both subgroups received statin treatment was nearly the same: 72% for those with MA coverage and 71% for those with FFS Medicare.
But MA beneficiaries differed from those with FFS coverage in several other ways. MA beneficiaries had a higher prevalence of Medicaid eligibility than the FFS group (20% vs 12%) and lower rates of consultations with cardiologists (41% vs. 45%) or endocrinologists (7% vs. 10%).
Some of the positive differences in the care received by MA beneficiaries, compared with FFS beneficiaries, after adjustment for potential clinical and sociodemographic confounders, included:
- Screening for nephropathy, at a significant 14% higher relative rate.
- Screening for ophthalmologic disorders, at a significant 8% higher relative rate.
- Undergoing a diabetic foot examination, at a significant 13% higher relative rate.
- Receiving smoking-cessation counseling, at a significant 5% higher relative rate.
- Receiving treatment with an ACE inhibitor or angiotensin-receptor blocker (87% vs. 81%).
- More consistently receiving treatment with metformin, with rates of 72% versus 69% in 2017.
However, these positive differences were accompanied by these relative shortcomings for those with MA, compared with FFS coverage:
- Lower rates of treatment with an SGLT2 inhibitor (5.4% vs. 6.7%), a significant 9% relative difference after adjustment.
- Lower rates of treatment with a GLP-1 agonist (6.9% vs. 9.0%), a significant 20% relative difference after adjustment.
- Higher average levels of LDL cholesterol (81.5 vs. 78.9 mg/dL), a significantly higher average hemoglobin A1c level (7.1% vs. 7.0%), and a trend toward a lower prevalence of blood pressure control (70.3% vs. 71.5%).
Researchers also highlight that the lower rate at which people with MA coverage received SGLT2 inhibitors or GLP-1 agonists was consistent in patients with established cardiovascular or kidney disease, for whom these agents are particularly recommended.
In addition, a secondary analysis of data for another 65,000 Medicare beneficiaries in 2018 and 2019 showed the disparity in use of agents from these two drug classes continued.
Low systemic use of SGLT2 inhibitors, GLP-1 agonists
Dr. Essien acknowledged that, even in people with FFS Medicare coverage, use of SGLT2 inhibitors and GLP-1 agonists was low, but the difference between those with MA coverage is “important.”
Researchers offered four factors that might drive reduced prescribing of agents from these two classes for patients with type 2 diabetes with MA coverage: cost-containment strategies put in place by MA plans; the lower rate of consultations with specialists (cardiologists and endocrinologists); possible exclusion of clinicians from MA provider networks who tend to prescribe these higher-price agents; and lower household incomes of people with MA plans, which may lead to cost-related nonadherence.
Most SGLT2 inhibitors have an average retail cost of about $6,000/year, and some GLP-1 agonists cost more than $10,000/year.
In general, MA coverage includes more oversight of care and its cost than occurs with FFS coverage, noted Dr. Essien, an internal medicine physician at the University of Pittsburgh and a researcher at the Center for Health Equity Research and Promotion of the VA Pittsburgh Healthcare System.
“Incentives for using these more expensive treatments may not be there in MA plans,” he explained. Overcoming cost-related barriers is a challenge that will require “bold policy changes,” as well as better education of clinicians so they make correct treatment decisions, and of patients to resolve possible concerns about treatment safety.
Rep. DeGette hinted during her remarks at the June hearing that policy changes may be coming from Congress.
“Our seniors and their doctors should not be required to jump through numerous hoops to get coverage for straightforward and medically necessary procedures,” she said.
The study received no commercial funding. Dr. Essien reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
U.S. Medicare beneficiaries with type 2 diabetes who had health coverage through a Medicare Advantage (MA) plan received treatment with an sodium-glucose cotransporter 2 inhibitor or glucagonlike peptide–1 receptor agonist significantly less often than patients with traditional fee-for-service (FFS) Medicare coverage in 2014-2019, according to a study of more than 411,000 patients.
concluded Utibe R. Essien, MD, and coauthors in a study published in Diabetes Care.
The report comes as the U.S. Congress is looking closely at the MA program and evidence that insurance companies that provide these policies sometimes impose inappropriate barriers on enrolled beneficiaries by denying or limiting access to treatments and interventions in ways that run counter to Medicare’s coverage policies.
According to Representative Diana DeGette (D-Colo.), who chaired a hearing on MA plans on June 28 by the House of Representatives’ Energy and Commerce Subcommittee on Oversight and Investigations, beneficiaries who are covered through an MA plan “do not always get the care that they are entitled to.”
The study by Dr. Essien and colleagues also documents some positives of care delivered through MA plans for patients with type 2 diabetes, compared with what FFS Medicare beneficiaries generally receive, such as significantly higher rates of screening for nephropathy and ophthalmologic disorders, and foot examinations.
But the apparently dampened use of SGLT2 inhibitors and GLP-1 receptor agonists among MA beneficiaries stand out as notable shortcomings, Dr. Essien maintained.
Cost containment may limit use
“The differences in health outcomes and in treatments in MA plans are important to highlight,” Dr. Essien said in an interview. “We worry that the cost-containment challenges [associated with MA plans] may be limiting use of these newer treatments.”
The study was based on 2014-2019 data from the Diabetes Collaborative Registry, which collects information from more than 5,000 U.S. clinicians whose practices include patients with diabetes, as well as claims data recorded by the Centers for Medicare and Medicaid Services during 2014-2017.
The main analysis focused on 345,911 Medicare beneficiaries with diabetes, which included 34% with MA coverage and 66% with FFS coverage. The two subgroups had similar ages, about 75 years old, and roughly half were women in both subgroups. The rate at which both subgroups received statin treatment was nearly the same: 72% for those with MA coverage and 71% for those with FFS Medicare.
But MA beneficiaries differed from those with FFS coverage in several other ways. MA beneficiaries had a higher prevalence of Medicaid eligibility than the FFS group (20% vs 12%) and lower rates of consultations with cardiologists (41% vs. 45%) or endocrinologists (7% vs. 10%).
Some of the positive differences in the care received by MA beneficiaries, compared with FFS beneficiaries, after adjustment for potential clinical and sociodemographic confounders, included:
- Screening for nephropathy, at a significant 14% higher relative rate.
- Screening for ophthalmologic disorders, at a significant 8% higher relative rate.
- Undergoing a diabetic foot examination, at a significant 13% higher relative rate.
- Receiving smoking-cessation counseling, at a significant 5% higher relative rate.
- Receiving treatment with an ACE inhibitor or angiotensin-receptor blocker (87% vs. 81%).
- More consistently receiving treatment with metformin, with rates of 72% versus 69% in 2017.
However, these positive differences were accompanied by these relative shortcomings for those with MA, compared with FFS coverage:
- Lower rates of treatment with an SGLT2 inhibitor (5.4% vs. 6.7%), a significant 9% relative difference after adjustment.
- Lower rates of treatment with a GLP-1 agonist (6.9% vs. 9.0%), a significant 20% relative difference after adjustment.
- Higher average levels of LDL cholesterol (81.5 vs. 78.9 mg/dL), a significantly higher average hemoglobin A1c level (7.1% vs. 7.0%), and a trend toward a lower prevalence of blood pressure control (70.3% vs. 71.5%).
Researchers also highlight that the lower rate at which people with MA coverage received SGLT2 inhibitors or GLP-1 agonists was consistent in patients with established cardiovascular or kidney disease, for whom these agents are particularly recommended.
In addition, a secondary analysis of data for another 65,000 Medicare beneficiaries in 2018 and 2019 showed the disparity in use of agents from these two drug classes continued.
Low systemic use of SGLT2 inhibitors, GLP-1 agonists
Dr. Essien acknowledged that, even in people with FFS Medicare coverage, use of SGLT2 inhibitors and GLP-1 agonists was low, but the difference between those with MA coverage is “important.”
Researchers offered four factors that might drive reduced prescribing of agents from these two classes for patients with type 2 diabetes with MA coverage: cost-containment strategies put in place by MA plans; the lower rate of consultations with specialists (cardiologists and endocrinologists); possible exclusion of clinicians from MA provider networks who tend to prescribe these higher-price agents; and lower household incomes of people with MA plans, which may lead to cost-related nonadherence.
Most SGLT2 inhibitors have an average retail cost of about $6,000/year, and some GLP-1 agonists cost more than $10,000/year.
In general, MA coverage includes more oversight of care and its cost than occurs with FFS coverage, noted Dr. Essien, an internal medicine physician at the University of Pittsburgh and a researcher at the Center for Health Equity Research and Promotion of the VA Pittsburgh Healthcare System.
“Incentives for using these more expensive treatments may not be there in MA plans,” he explained. Overcoming cost-related barriers is a challenge that will require “bold policy changes,” as well as better education of clinicians so they make correct treatment decisions, and of patients to resolve possible concerns about treatment safety.
Rep. DeGette hinted during her remarks at the June hearing that policy changes may be coming from Congress.
“Our seniors and their doctors should not be required to jump through numerous hoops to get coverage for straightforward and medically necessary procedures,” she said.
The study received no commercial funding. Dr. Essien reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM DIABETES CARE
Fourth patient cleared of HIV after stem cell transplant for blood cancer
MONTREAL – from a naturally HIV-resistant donor, U.S. researchers announced at a meeting of the International AIDS Society.
The man received the transplant nearly 3.5 years ago. Since discontinuation of antiretroviral therapy (ART) more than 17 months ago, he has shown no evidence of HIV-1 RNA rebound and no detectable HIV-1 DNA, reported lead investigator Jana K. Dickter, MD, associate clinical professor in the division of infectious diseases at City of Hope, a Duarte, Calif.–based stem cell transplantation center for patients with blood cancers and patients with HIV/blood cancer.
Known as the City of Hope (COH) patient, he is different from the three previously reported patients in that “he was the oldest person to successfully undergo a stem cell transplant with HIV and leukemia and then achieve remission from both conditions,” Dr. Dickter said during a press briefing for the meeting. “He has been living with HIV the longest of any of the patients to date – more than 31 years prior to transplant – and he had also received the least immunosuppressive preparative regimen prior to transplant,” she added.
She said that, like the three previous patients, known as the Berlin, London, and New York patients, the COH patient received a transplant from a donor with natural resistance to HIV because of a rare CCR5-delta 32 mutation.
Dr. Dickter and her coinvestigators used the term “remission” but went further, suggesting that an “HIV cure is feasible” after transplant, given this and the previous cases.
“It’s a bit early to say the patient is cured, but they are clearly in remission,” said Sharon Lewin, MD, president-elect of the International AIDS Society, which runs the meeting. Nevertheless, Dr. Lewin, professor of medicine at the University of Melbourne and director of the Peter Doherty Institute for Infection and Immunity, in Melbourne, acknowledged that cure is “very likely.”
“Two of the previously reported patients have been off ART for long periods of time – Berlin, 12 years (until Timothy’s death in 2020); London, 4 years – and both had far more extensive investigations to try and find intact virus, including very large blood draws, tissue biopsies, etc. For the New York and now this COH patient, the duration off ART has been much shorter. ... But given the prior cases, it is very likely that the New York and COH patients are indeed cured. But I think it’s too early to make that call, hence my preference to use the word, ‘remission,’ “ she told this news organization.
“Although a transplant is not an option for most people with HIV, these cases are still interesting, still inspiring, and help illuminate the search for a cure,” she added.
Dr. Dickter acknowledged that the complexity of stem cell transplant procedures and their potential for significant side effects make them unsuitable as treatment options for most people with HIV, although she said the COH case is evidence that some HIV patients with blood cancers may not need such intensive pretransplant conditioning regimens.
The COH patient received a reduced-intensity fludarabine and melphalan regimen that had been designed at Dr. Dickter’s center “for older and less fit patients to make transplantation more tolerable,” she said. In addition, the graft-vs.-host disease prophylaxis that the COH patient received included only tacrolimus and sirolimus, whereas the previous patients received additional immunosuppressive therapies, and some also had undergone total body irradiation.
Dr. Dickter has disclosed no relevant financial relationships. Dr. Lewin has relationships with AbbVie, BMS, Esfam, Genentech, Gilead, Immunocore, Merck, Vaxxinity, and Viiv.
A version of this article first appeared on Medscape.com.
MONTREAL – from a naturally HIV-resistant donor, U.S. researchers announced at a meeting of the International AIDS Society.
The man received the transplant nearly 3.5 years ago. Since discontinuation of antiretroviral therapy (ART) more than 17 months ago, he has shown no evidence of HIV-1 RNA rebound and no detectable HIV-1 DNA, reported lead investigator Jana K. Dickter, MD, associate clinical professor in the division of infectious diseases at City of Hope, a Duarte, Calif.–based stem cell transplantation center for patients with blood cancers and patients with HIV/blood cancer.
Known as the City of Hope (COH) patient, he is different from the three previously reported patients in that “he was the oldest person to successfully undergo a stem cell transplant with HIV and leukemia and then achieve remission from both conditions,” Dr. Dickter said during a press briefing for the meeting. “He has been living with HIV the longest of any of the patients to date – more than 31 years prior to transplant – and he had also received the least immunosuppressive preparative regimen prior to transplant,” she added.
She said that, like the three previous patients, known as the Berlin, London, and New York patients, the COH patient received a transplant from a donor with natural resistance to HIV because of a rare CCR5-delta 32 mutation.
Dr. Dickter and her coinvestigators used the term “remission” but went further, suggesting that an “HIV cure is feasible” after transplant, given this and the previous cases.
“It’s a bit early to say the patient is cured, but they are clearly in remission,” said Sharon Lewin, MD, president-elect of the International AIDS Society, which runs the meeting. Nevertheless, Dr. Lewin, professor of medicine at the University of Melbourne and director of the Peter Doherty Institute for Infection and Immunity, in Melbourne, acknowledged that cure is “very likely.”
“Two of the previously reported patients have been off ART for long periods of time – Berlin, 12 years (until Timothy’s death in 2020); London, 4 years – and both had far more extensive investigations to try and find intact virus, including very large blood draws, tissue biopsies, etc. For the New York and now this COH patient, the duration off ART has been much shorter. ... But given the prior cases, it is very likely that the New York and COH patients are indeed cured. But I think it’s too early to make that call, hence my preference to use the word, ‘remission,’ “ she told this news organization.
“Although a transplant is not an option for most people with HIV, these cases are still interesting, still inspiring, and help illuminate the search for a cure,” she added.
Dr. Dickter acknowledged that the complexity of stem cell transplant procedures and their potential for significant side effects make them unsuitable as treatment options for most people with HIV, although she said the COH case is evidence that some HIV patients with blood cancers may not need such intensive pretransplant conditioning regimens.
The COH patient received a reduced-intensity fludarabine and melphalan regimen that had been designed at Dr. Dickter’s center “for older and less fit patients to make transplantation more tolerable,” she said. In addition, the graft-vs.-host disease prophylaxis that the COH patient received included only tacrolimus and sirolimus, whereas the previous patients received additional immunosuppressive therapies, and some also had undergone total body irradiation.
Dr. Dickter has disclosed no relevant financial relationships. Dr. Lewin has relationships with AbbVie, BMS, Esfam, Genentech, Gilead, Immunocore, Merck, Vaxxinity, and Viiv.
A version of this article first appeared on Medscape.com.
MONTREAL – from a naturally HIV-resistant donor, U.S. researchers announced at a meeting of the International AIDS Society.
The man received the transplant nearly 3.5 years ago. Since discontinuation of antiretroviral therapy (ART) more than 17 months ago, he has shown no evidence of HIV-1 RNA rebound and no detectable HIV-1 DNA, reported lead investigator Jana K. Dickter, MD, associate clinical professor in the division of infectious diseases at City of Hope, a Duarte, Calif.–based stem cell transplantation center for patients with blood cancers and patients with HIV/blood cancer.
Known as the City of Hope (COH) patient, he is different from the three previously reported patients in that “he was the oldest person to successfully undergo a stem cell transplant with HIV and leukemia and then achieve remission from both conditions,” Dr. Dickter said during a press briefing for the meeting. “He has been living with HIV the longest of any of the patients to date – more than 31 years prior to transplant – and he had also received the least immunosuppressive preparative regimen prior to transplant,” she added.
She said that, like the three previous patients, known as the Berlin, London, and New York patients, the COH patient received a transplant from a donor with natural resistance to HIV because of a rare CCR5-delta 32 mutation.
Dr. Dickter and her coinvestigators used the term “remission” but went further, suggesting that an “HIV cure is feasible” after transplant, given this and the previous cases.
“It’s a bit early to say the patient is cured, but they are clearly in remission,” said Sharon Lewin, MD, president-elect of the International AIDS Society, which runs the meeting. Nevertheless, Dr. Lewin, professor of medicine at the University of Melbourne and director of the Peter Doherty Institute for Infection and Immunity, in Melbourne, acknowledged that cure is “very likely.”
“Two of the previously reported patients have been off ART for long periods of time – Berlin, 12 years (until Timothy’s death in 2020); London, 4 years – and both had far more extensive investigations to try and find intact virus, including very large blood draws, tissue biopsies, etc. For the New York and now this COH patient, the duration off ART has been much shorter. ... But given the prior cases, it is very likely that the New York and COH patients are indeed cured. But I think it’s too early to make that call, hence my preference to use the word, ‘remission,’ “ she told this news organization.
“Although a transplant is not an option for most people with HIV, these cases are still interesting, still inspiring, and help illuminate the search for a cure,” she added.
Dr. Dickter acknowledged that the complexity of stem cell transplant procedures and their potential for significant side effects make them unsuitable as treatment options for most people with HIV, although she said the COH case is evidence that some HIV patients with blood cancers may not need such intensive pretransplant conditioning regimens.
The COH patient received a reduced-intensity fludarabine and melphalan regimen that had been designed at Dr. Dickter’s center “for older and less fit patients to make transplantation more tolerable,” she said. In addition, the graft-vs.-host disease prophylaxis that the COH patient received included only tacrolimus and sirolimus, whereas the previous patients received additional immunosuppressive therapies, and some also had undergone total body irradiation.
Dr. Dickter has disclosed no relevant financial relationships. Dr. Lewin has relationships with AbbVie, BMS, Esfam, Genentech, Gilead, Immunocore, Merck, Vaxxinity, and Viiv.
A version of this article first appeared on Medscape.com.
AT AIDS 2022
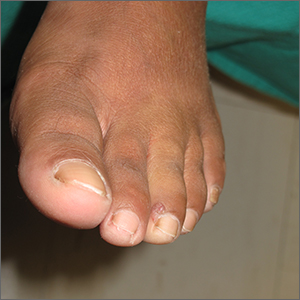
Toe growth
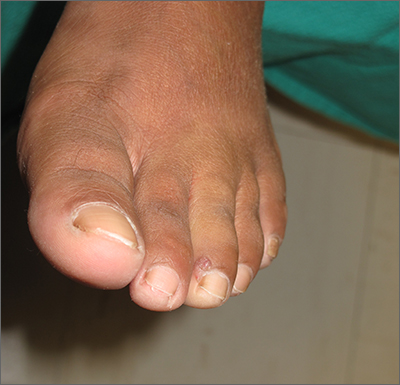
Shave biopsy was consistent with a solitary periungual angiofibroma, often termed a Koenen tumor. These can manifest as a soft pink papule (as with this patient), sometimes with a distal keratinaceous tip. At times, the nail bed and nail plate may be deformed because of the angiofibroma.
Periungual angiofibromas can occur sporadically in children and adults, it was a solitary finding in this case. Importantly, periungual angiofibromas may also occur as a visible sign of a multisystem genetic disorder known as tuberous sclerosis complex (TSC). TSC causes benign tumors to develop throughout the body (eg, skin, brain, heart, lungs). The condition can be mild or lead to serious disabilities, including seizures and developmental delays.
In isolation, periungual angiofibromas are benign but occasionally hurt or bleed from light trauma. In such cases, or for cosmetic reasons, patients may seek treatments. Complete excision of the lesion may include the affected portion of the nail bed or matrix. This is more easily repaired when the lesion is on the lateral nail fold, facilitating an en bloc fusiform excision and matrixectomy.1 Surgical excision of a lesion in the mid-proximal nail fold is much more likely to result in long-term nail deformity. Electrosurgery and various laser modalities have been successful as less invasive removal options.2 While expensive, topical sirolimus 1% has been used successfully in sporadic angiofibromas and those associated with TSC.
The patient in this case underwent lateral nail fold excision with complete removal of the tumor and repair with a side-to-side closure.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Tisa LM, Iurcotta A. Solitary periungual angiofibroma. An unusual case report. J Am Podiatr Med Assoc. 1993;83:679-80. doi: 10.7547/87507315-83-12-679
2. Boixeda P, Sánchez-Miralles E, Azaña JM, et al. CO2, argon, and pulsed dye laser treatment of angiofibromas. J Dermatol Surg Oncol. 1994;20:808-812. doi: 10.1111/j.1524- 4725.1994.tb03709.x

Shave biopsy was consistent with a solitary periungual angiofibroma, often termed a Koenen tumor. These can manifest as a soft pink papule (as with this patient), sometimes with a distal keratinaceous tip. At times, the nail bed and nail plate may be deformed because of the angiofibroma.
Periungual angiofibromas can occur sporadically in children and adults, it was a solitary finding in this case. Importantly, periungual angiofibromas may also occur as a visible sign of a multisystem genetic disorder known as tuberous sclerosis complex (TSC). TSC causes benign tumors to develop throughout the body (eg, skin, brain, heart, lungs). The condition can be mild or lead to serious disabilities, including seizures and developmental delays.
In isolation, periungual angiofibromas are benign but occasionally hurt or bleed from light trauma. In such cases, or for cosmetic reasons, patients may seek treatments. Complete excision of the lesion may include the affected portion of the nail bed or matrix. This is more easily repaired when the lesion is on the lateral nail fold, facilitating an en bloc fusiform excision and matrixectomy.1 Surgical excision of a lesion in the mid-proximal nail fold is much more likely to result in long-term nail deformity. Electrosurgery and various laser modalities have been successful as less invasive removal options.2 While expensive, topical sirolimus 1% has been used successfully in sporadic angiofibromas and those associated with TSC.
The patient in this case underwent lateral nail fold excision with complete removal of the tumor and repair with a side-to-side closure.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

Shave biopsy was consistent with a solitary periungual angiofibroma, often termed a Koenen tumor. These can manifest as a soft pink papule (as with this patient), sometimes with a distal keratinaceous tip. At times, the nail bed and nail plate may be deformed because of the angiofibroma.
Periungual angiofibromas can occur sporadically in children and adults, it was a solitary finding in this case. Importantly, periungual angiofibromas may also occur as a visible sign of a multisystem genetic disorder known as tuberous sclerosis complex (TSC). TSC causes benign tumors to develop throughout the body (eg, skin, brain, heart, lungs). The condition can be mild or lead to serious disabilities, including seizures and developmental delays.
In isolation, periungual angiofibromas are benign but occasionally hurt or bleed from light trauma. In such cases, or for cosmetic reasons, patients may seek treatments. Complete excision of the lesion may include the affected portion of the nail bed or matrix. This is more easily repaired when the lesion is on the lateral nail fold, facilitating an en bloc fusiform excision and matrixectomy.1 Surgical excision of a lesion in the mid-proximal nail fold is much more likely to result in long-term nail deformity. Electrosurgery and various laser modalities have been successful as less invasive removal options.2 While expensive, topical sirolimus 1% has been used successfully in sporadic angiofibromas and those associated with TSC.
The patient in this case underwent lateral nail fold excision with complete removal of the tumor and repair with a side-to-side closure.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Tisa LM, Iurcotta A. Solitary periungual angiofibroma. An unusual case report. J Am Podiatr Med Assoc. 1993;83:679-80. doi: 10.7547/87507315-83-12-679
2. Boixeda P, Sánchez-Miralles E, Azaña JM, et al. CO2, argon, and pulsed dye laser treatment of angiofibromas. J Dermatol Surg Oncol. 1994;20:808-812. doi: 10.1111/j.1524- 4725.1994.tb03709.x
1. Tisa LM, Iurcotta A. Solitary periungual angiofibroma. An unusual case report. J Am Podiatr Med Assoc. 1993;83:679-80. doi: 10.7547/87507315-83-12-679
2. Boixeda P, Sánchez-Miralles E, Azaña JM, et al. CO2, argon, and pulsed dye laser treatment of angiofibromas. J Dermatol Surg Oncol. 1994;20:808-812. doi: 10.1111/j.1524- 4725.1994.tb03709.x
Meta-analysis evaluates probiotic vs drug intervention in IBS
Key clinical point: Both probiotic and drug interventions decreased the persistence of irritable bowel syndrome (IBS) symptoms and abdominal pain scores compared with placebo in patients with IBS.
Major finding: Compared with placebo, both probiotic and drug interventions improved the persistence of IBS symptoms (relative risk [RR] 0.68; 95% CI 0.51-0.92, and RR 0.87; 95% CI 0.81-0.92, respectively) and abdominal pain scores (standardized mean difference [SMD] −0.35; 95% CI −0.56 to −0.14, and SMD −0.10; 95% CI −0.20 to 0.00, respectively).
Study details Findings are from a systematic review and meta-analysis of 32 randomized controlled trials including adults with IBS.
Disclosures: This study did not receive financial support. The authors declared being scientific advisory board members or serving as consultants for various sources.
Source: van der Geest AM et al. Comparing probiotic and drug interventions in irritable bowel syndrome: A meta-analysis of randomised controlled trials. Benef Microbes. 2022 (Jul 18). Doi: 10.3920/BM2021.0123
Key clinical point: Both probiotic and drug interventions decreased the persistence of irritable bowel syndrome (IBS) symptoms and abdominal pain scores compared with placebo in patients with IBS.
Major finding: Compared with placebo, both probiotic and drug interventions improved the persistence of IBS symptoms (relative risk [RR] 0.68; 95% CI 0.51-0.92, and RR 0.87; 95% CI 0.81-0.92, respectively) and abdominal pain scores (standardized mean difference [SMD] −0.35; 95% CI −0.56 to −0.14, and SMD −0.10; 95% CI −0.20 to 0.00, respectively).
Study details Findings are from a systematic review and meta-analysis of 32 randomized controlled trials including adults with IBS.
Disclosures: This study did not receive financial support. The authors declared being scientific advisory board members or serving as consultants for various sources.
Source: van der Geest AM et al. Comparing probiotic and drug interventions in irritable bowel syndrome: A meta-analysis of randomised controlled trials. Benef Microbes. 2022 (Jul 18). Doi: 10.3920/BM2021.0123
Key clinical point: Both probiotic and drug interventions decreased the persistence of irritable bowel syndrome (IBS) symptoms and abdominal pain scores compared with placebo in patients with IBS.
Major finding: Compared with placebo, both probiotic and drug interventions improved the persistence of IBS symptoms (relative risk [RR] 0.68; 95% CI 0.51-0.92, and RR 0.87; 95% CI 0.81-0.92, respectively) and abdominal pain scores (standardized mean difference [SMD] −0.35; 95% CI −0.56 to −0.14, and SMD −0.10; 95% CI −0.20 to 0.00, respectively).
Study details Findings are from a systematic review and meta-analysis of 32 randomized controlled trials including adults with IBS.
Disclosures: This study did not receive financial support. The authors declared being scientific advisory board members or serving as consultants for various sources.
Source: van der Geest AM et al. Comparing probiotic and drug interventions in irritable bowel syndrome: A meta-analysis of randomised controlled trials. Benef Microbes. 2022 (Jul 18). Doi: 10.3920/BM2021.0123
Fecal microbiota transplantation shows promise in patients with refractory IBS
Key clinical point: Fecal microbiota transplantation (FMT) reduced disease severity, improved microbiome α-diversity, and demonstrated a favorable safety profile in patients with refractory irritable bowel syndrome (IBS).
Major finding: The IBS severity index improved significantly at 12 weeks after FMT (P = .001), with 58.8% of patients being responders (≥50-point decrease in IBS severity index). Responders showed a significant increase in the median α-diversity index (P = .017), increased abundance of Neisseria and Akkermansia bacteria, and decreased abundance of Desulfovibrio and Delftia bacteria. No severe adverse events were reported.
Study details Findings are from a prospective study including 17 patients with refractory IBS who received FMT under colonoscopy.
Disclosures: This study was partly supported by the Japan Society for the Promotion of Science. The authors declared no conflicts of interest.
Source: Hamazaki M et al. Fecal microbiota transplantation in the treatment of irritable bowel syndrome: A single-center prospective study in Japan. BMC Gastroenterol. 2022;22:342 (Jul 14). Doi: 10.1186/s12876-022-02408-5
Key clinical point: Fecal microbiota transplantation (FMT) reduced disease severity, improved microbiome α-diversity, and demonstrated a favorable safety profile in patients with refractory irritable bowel syndrome (IBS).
Major finding: The IBS severity index improved significantly at 12 weeks after FMT (P = .001), with 58.8% of patients being responders (≥50-point decrease in IBS severity index). Responders showed a significant increase in the median α-diversity index (P = .017), increased abundance of Neisseria and Akkermansia bacteria, and decreased abundance of Desulfovibrio and Delftia bacteria. No severe adverse events were reported.
Study details Findings are from a prospective study including 17 patients with refractory IBS who received FMT under colonoscopy.
Disclosures: This study was partly supported by the Japan Society for the Promotion of Science. The authors declared no conflicts of interest.
Source: Hamazaki M et al. Fecal microbiota transplantation in the treatment of irritable bowel syndrome: A single-center prospective study in Japan. BMC Gastroenterol. 2022;22:342 (Jul 14). Doi: 10.1186/s12876-022-02408-5
Key clinical point: Fecal microbiota transplantation (FMT) reduced disease severity, improved microbiome α-diversity, and demonstrated a favorable safety profile in patients with refractory irritable bowel syndrome (IBS).
Major finding: The IBS severity index improved significantly at 12 weeks after FMT (P = .001), with 58.8% of patients being responders (≥50-point decrease in IBS severity index). Responders showed a significant increase in the median α-diversity index (P = .017), increased abundance of Neisseria and Akkermansia bacteria, and decreased abundance of Desulfovibrio and Delftia bacteria. No severe adverse events were reported.
Study details Findings are from a prospective study including 17 patients with refractory IBS who received FMT under colonoscopy.
Disclosures: This study was partly supported by the Japan Society for the Promotion of Science. The authors declared no conflicts of interest.
Source: Hamazaki M et al. Fecal microbiota transplantation in the treatment of irritable bowel syndrome: A single-center prospective study in Japan. BMC Gastroenterol. 2022;22:342 (Jul 14). Doi: 10.1186/s12876-022-02408-5
Irritable bowel syndrome severely hampers work and daily living activities
Key clinical point: Patients with irritable bowel syndrome (IBS) report a substantial impact of IBS on their abilities to work and perform daily activities.
Major finding: Overall, 10.5% of patients reported being unemployed, partly because of their condition, with the median level of absenteeism, presenteeism, and overall work impairment among employed patients being 0.0% (interquartile range [IQR] 0.0%-2.9%), 35.0% (IQR, 20.0%-60.0%), and 30.0% (IQR, 10.0%-60.0%), respectively. Moreover, 91.0% and 56.3% of patients reported that IBS symptoms interfered with their daily and social leisure activities, respectively.
Study details: This was a questionnaire-based study including 752 patients with Rome IV-defined IBS.
Disclosures: This study was supported by Tillotts Pharma UK Ltd. The authors declared no conflicts of interest.
Source: Goodoory VC et al. Impact of Rome IV irritable bowel syndrome on work and activities of daily living .Aliment Pharmacol Ther. 2022 (Jul 6). Doi: 10.1111/apt.17132
Key clinical point: Patients with irritable bowel syndrome (IBS) report a substantial impact of IBS on their abilities to work and perform daily activities.
Major finding: Overall, 10.5% of patients reported being unemployed, partly because of their condition, with the median level of absenteeism, presenteeism, and overall work impairment among employed patients being 0.0% (interquartile range [IQR] 0.0%-2.9%), 35.0% (IQR, 20.0%-60.0%), and 30.0% (IQR, 10.0%-60.0%), respectively. Moreover, 91.0% and 56.3% of patients reported that IBS symptoms interfered with their daily and social leisure activities, respectively.
Study details: This was a questionnaire-based study including 752 patients with Rome IV-defined IBS.
Disclosures: This study was supported by Tillotts Pharma UK Ltd. The authors declared no conflicts of interest.
Source: Goodoory VC et al. Impact of Rome IV irritable bowel syndrome on work and activities of daily living .Aliment Pharmacol Ther. 2022 (Jul 6). Doi: 10.1111/apt.17132
Key clinical point: Patients with irritable bowel syndrome (IBS) report a substantial impact of IBS on their abilities to work and perform daily activities.
Major finding: Overall, 10.5% of patients reported being unemployed, partly because of their condition, with the median level of absenteeism, presenteeism, and overall work impairment among employed patients being 0.0% (interquartile range [IQR] 0.0%-2.9%), 35.0% (IQR, 20.0%-60.0%), and 30.0% (IQR, 10.0%-60.0%), respectively. Moreover, 91.0% and 56.3% of patients reported that IBS symptoms interfered with their daily and social leisure activities, respectively.
Study details: This was a questionnaire-based study including 752 patients with Rome IV-defined IBS.
Disclosures: This study was supported by Tillotts Pharma UK Ltd. The authors declared no conflicts of interest.
Source: Goodoory VC et al. Impact of Rome IV irritable bowel syndrome on work and activities of daily living .Aliment Pharmacol Ther. 2022 (Jul 6). Doi: 10.1111/apt.17132
Probiotics improve stool consistency and are well tolerated in IBS-C
Key clinical point: In patients with constipation-predominant irritable bowel syndrome (IBS-C), short-term treatment with probiotics vs placebo led to a significant improvement in stool consistency and increased the number of fecal bacteria without increasing adverse events.
Major finding: Probiotics vs placebo significantly improved stool consistency (mean difference [MD] 0.71; P < .05) in 8 weeks and increased the number of fecal Bifidobacterium (MD 1.76; P < .05) and Lactobacillus (MD 1.69; P < .05) in 4 weeks, although the effects were not significant with longer treatment duration. Adverse effects were not significantly different between the probiotic and placebo groups.
Study details Findings are from a systematic review and meta-analysis of 10 randomized controlled trials including 757 patients with IBS-C.
Disclosures: This study was supported by the National Natural Science Foundation of China, the Gansu Special Project of Soft Science, and others. The authors declared no conflicts of interest.
Source: Shang X et al. Effectiveness and safety of probiotics for patients with constipation-predominant irritable bowel syndrome: A systematic review and meta-analysis of 10 randomized controlled trials. Nutrients. 2022;14(12):2482 (Jun 15). Doi: 10.3390/nu14122482
Key clinical point: In patients with constipation-predominant irritable bowel syndrome (IBS-C), short-term treatment with probiotics vs placebo led to a significant improvement in stool consistency and increased the number of fecal bacteria without increasing adverse events.
Major finding: Probiotics vs placebo significantly improved stool consistency (mean difference [MD] 0.71; P < .05) in 8 weeks and increased the number of fecal Bifidobacterium (MD 1.76; P < .05) and Lactobacillus (MD 1.69; P < .05) in 4 weeks, although the effects were not significant with longer treatment duration. Adverse effects were not significantly different between the probiotic and placebo groups.
Study details Findings are from a systematic review and meta-analysis of 10 randomized controlled trials including 757 patients with IBS-C.
Disclosures: This study was supported by the National Natural Science Foundation of China, the Gansu Special Project of Soft Science, and others. The authors declared no conflicts of interest.
Source: Shang X et al. Effectiveness and safety of probiotics for patients with constipation-predominant irritable bowel syndrome: A systematic review and meta-analysis of 10 randomized controlled trials. Nutrients. 2022;14(12):2482 (Jun 15). Doi: 10.3390/nu14122482
Key clinical point: In patients with constipation-predominant irritable bowel syndrome (IBS-C), short-term treatment with probiotics vs placebo led to a significant improvement in stool consistency and increased the number of fecal bacteria without increasing adverse events.
Major finding: Probiotics vs placebo significantly improved stool consistency (mean difference [MD] 0.71; P < .05) in 8 weeks and increased the number of fecal Bifidobacterium (MD 1.76; P < .05) and Lactobacillus (MD 1.69; P < .05) in 4 weeks, although the effects were not significant with longer treatment duration. Adverse effects were not significantly different between the probiotic and placebo groups.
Study details Findings are from a systematic review and meta-analysis of 10 randomized controlled trials including 757 patients with IBS-C.
Disclosures: This study was supported by the National Natural Science Foundation of China, the Gansu Special Project of Soft Science, and others. The authors declared no conflicts of interest.
Source: Shang X et al. Effectiveness and safety of probiotics for patients with constipation-predominant irritable bowel syndrome: A systematic review and meta-analysis of 10 randomized controlled trials. Nutrients. 2022;14(12):2482 (Jun 15). Doi: 10.3390/nu14122482
Food supplements improve standard treatment efficacy in IBS
Key clinical point: Food supplements containing standardized menthol, limonene, and gingerol content significantly improved the efficacy of standard treatment in patients with irritable bowel syndrome (IBS) or IBS/functional dyspepsia (FD) without causing any obvious side effects.
Major finding: At visit 1 (before treatment), the median 7×7 questionnaire score of all patients indicated moderate illness. However, at visit 3, the score range indicated borderline illness in patients who received the food supplement and mild illness in those who received placebo (mean difference, −2.68; P = .009). No side effects were reported.
Study details Findings are from a randomized controlled trial including 56 patients with IBS or IBS/FD who were randomly assigned to receive food supplements or placebo in addition to the standard treatment regimen for 30 days.
Disclosures: This study did not receive any funding. The authors declared no conflicts of interest.
Source: Ivashkin VT et al. Efficacy and safety of a food supplement with standardized menthol, limonene, and gingerol content in patients with irritable bowel syndrome: A double-blind, randomized, placebo-controlled trial. PloS One. 2022;17(6):e0263880 (Jun 15). Doi: 10.1371/journal.pone.0263880
Key clinical point: Food supplements containing standardized menthol, limonene, and gingerol content significantly improved the efficacy of standard treatment in patients with irritable bowel syndrome (IBS) or IBS/functional dyspepsia (FD) without causing any obvious side effects.
Major finding: At visit 1 (before treatment), the median 7×7 questionnaire score of all patients indicated moderate illness. However, at visit 3, the score range indicated borderline illness in patients who received the food supplement and mild illness in those who received placebo (mean difference, −2.68; P = .009). No side effects were reported.
Study details Findings are from a randomized controlled trial including 56 patients with IBS or IBS/FD who were randomly assigned to receive food supplements or placebo in addition to the standard treatment regimen for 30 days.
Disclosures: This study did not receive any funding. The authors declared no conflicts of interest.
Source: Ivashkin VT et al. Efficacy and safety of a food supplement with standardized menthol, limonene, and gingerol content in patients with irritable bowel syndrome: A double-blind, randomized, placebo-controlled trial. PloS One. 2022;17(6):e0263880 (Jun 15). Doi: 10.1371/journal.pone.0263880
Key clinical point: Food supplements containing standardized menthol, limonene, and gingerol content significantly improved the efficacy of standard treatment in patients with irritable bowel syndrome (IBS) or IBS/functional dyspepsia (FD) without causing any obvious side effects.
Major finding: At visit 1 (before treatment), the median 7×7 questionnaire score of all patients indicated moderate illness. However, at visit 3, the score range indicated borderline illness in patients who received the food supplement and mild illness in those who received placebo (mean difference, −2.68; P = .009). No side effects were reported.
Study details Findings are from a randomized controlled trial including 56 patients with IBS or IBS/FD who were randomly assigned to receive food supplements or placebo in addition to the standard treatment regimen for 30 days.
Disclosures: This study did not receive any funding. The authors declared no conflicts of interest.
Source: Ivashkin VT et al. Efficacy and safety of a food supplement with standardized menthol, limonene, and gingerol content in patients with irritable bowel syndrome: A double-blind, randomized, placebo-controlled trial. PloS One. 2022;17(6):e0263880 (Jun 15). Doi: 10.1371/journal.pone.0263880
Serum zinc levels, poor mood states, and QoL in diarrhea-predominant IBS
Key clinical point: Patients with diarrhea-predominant irritable bowel syndrome (IBS-D) vs healthy controls had lower serum zinc levels and poor scores for the quality of life (QoL) and physical and psychological health, and higher scores for depression, anxiety, and stress.
Major finding: Patients with IBS-D vs healthy controls reported significantly lower serum zinc levels (P = .001); mean scores for overall QoL and general health (53.48 vs 72.74; P ≤ .001), physical health (59.59 vs 71.30; P ≤ .001), and psychological health (55.80 vs 65.22; P ≤ .001); and higher mean scores for depression (P = .014), anxiety (P = .005), and stress (P = .001). Higher dietary zinc intake improved body image in patients with IBS-D (r 0.266; P = .044) without significantly influencing other parameters.
Study details: This was a case-control study including patients newly diagnosed with IBS-D (n = 61) and matched healthy controls (n = 61).
Disclosures: This study was supported by the Vice-Chancellor for Research of Isfahan University of Medical Sciences. The authors declared no conflicts of interest.
Source: Rezazadegan M et al. Sci Rep. 2022 Jun 29. doi: 10.1038/s41598-022-15080-2.
Key clinical point: Patients with diarrhea-predominant irritable bowel syndrome (IBS-D) vs healthy controls had lower serum zinc levels and poor scores for the quality of life (QoL) and physical and psychological health, and higher scores for depression, anxiety, and stress.
Major finding: Patients with IBS-D vs healthy controls reported significantly lower serum zinc levels (P = .001); mean scores for overall QoL and general health (53.48 vs 72.74; P ≤ .001), physical health (59.59 vs 71.30; P ≤ .001), and psychological health (55.80 vs 65.22; P ≤ .001); and higher mean scores for depression (P = .014), anxiety (P = .005), and stress (P = .001). Higher dietary zinc intake improved body image in patients with IBS-D (r 0.266; P = .044) without significantly influencing other parameters.
Study details: This was a case-control study including patients newly diagnosed with IBS-D (n = 61) and matched healthy controls (n = 61).
Disclosures: This study was supported by the Vice-Chancellor for Research of Isfahan University of Medical Sciences. The authors declared no conflicts of interest.
Source: Rezazadegan M et al. Sci Rep. 2022 Jun 29. doi: 10.1038/s41598-022-15080-2.
Key clinical point: Patients with diarrhea-predominant irritable bowel syndrome (IBS-D) vs healthy controls had lower serum zinc levels and poor scores for the quality of life (QoL) and physical and psychological health, and higher scores for depression, anxiety, and stress.
Major finding: Patients with IBS-D vs healthy controls reported significantly lower serum zinc levels (P = .001); mean scores for overall QoL and general health (53.48 vs 72.74; P ≤ .001), physical health (59.59 vs 71.30; P ≤ .001), and psychological health (55.80 vs 65.22; P ≤ .001); and higher mean scores for depression (P = .014), anxiety (P = .005), and stress (P = .001). Higher dietary zinc intake improved body image in patients with IBS-D (r 0.266; P = .044) without significantly influencing other parameters.
Study details: This was a case-control study including patients newly diagnosed with IBS-D (n = 61) and matched healthy controls (n = 61).
Disclosures: This study was supported by the Vice-Chancellor for Research of Isfahan University of Medical Sciences. The authors declared no conflicts of interest.
Source: Rezazadegan M et al. Sci Rep. 2022 Jun 29. doi: 10.1038/s41598-022-15080-2.