User login
EMERGENCY MEDICINE is a practical, peer-reviewed monthly publication and Web site that meets the educational needs of emergency clinicians and urgent care clinicians for their practice.
More fatalities in heart transplant patients with COVID-19
COVID-19 infection is associated with a high risk for mortality in heart transplant (HT) recipients, a new case series suggests.
Investigators looked at data on 28 patients with a confirmed diagnosis of COVID-19 who received a HT between March 1, 2020, and April 24, 2020 and found a case-fatality rate of 25%.
“The high case fatality in our case series should alert physicians to the vulnerability of heart transplant recipients during the COVID-19 pandemic,” senior author Nir Uriel, MD, MSc, professor of medicine at Columbia University, New York, said in an interview.
“These patients require extra precautions to prevent the development of infection,” said Dr. Uriel, who is also a cardiologist at New York Presbyterian/Columbia University Irving Medical Center.
The study was published online May 13 in JAMA Cardiology.
Similar presentation
HT recipients can have several comorbidities after the procedure, including hypertension, diabetes, cardiac allograft vasculopathy, and ongoing immunosuppression, all of which can place them at risk for infection and adverse outcomes with COVID-19 infection, the authors wrote.
The researchers therefore embarked on a case series looking at 28 HT recipients with COVID-19 infection (median age, 64.0 years; interquartile range, 53.5-70.5; 79% male) to “describe the outcomes of recipients of HT who are chronically immunosuppressed and develop COVID-19 and raise important questions about the role of the immune system in the process.”
The median time from HT to study period was 8.6 (IQR, 4.2-14.5) years. Most patients had numerous comorbidities.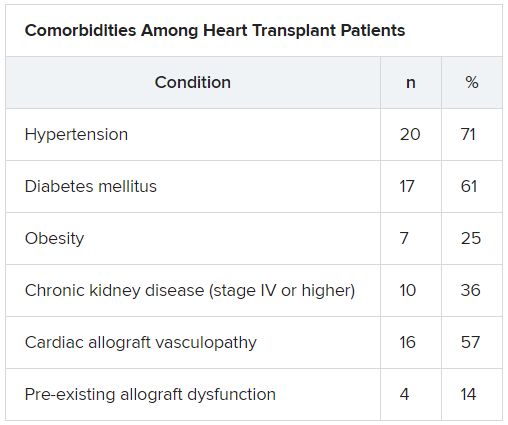
“The presentation of COVID-19 was similar to nontransplant patients with fever, dyspnea, cough, and GI symptoms,” Dr. Uriel reported.
No protective effect
Twenty-two patients (79%) required admission to the hospital, seven of whom (25%) required admission to the ICU and mechanical ventilation.
Despite the presence of immunosuppressive therapy, all patients had significant elevation of inflammatory biomarkers (median peak high-sensitivity C-reactive protein [hs-CRP], 11.83 mg/dL; IQR, 7.44-19.26; median peak interleukin [IL]-6, 105 pg/mL; IQR, 38-296).
Three-quarters had myocardial injury, with a median high-sensitivity troponin T of 0.055 (0.0205 - 0.1345) ng/mL.
Treatments of COVID-19 included hydroxychloroquine (18 patients; 78%), high-dose corticosteroids (eight patients; 47%), and IL-6 receptor antagonists (six patients; 26%).
Moreover, during hospitalization, mycophenolate mofetil was discontinued in most (70%) patients, and one-quarter had a reduction in their calcineurin inhibitor dose.
“Heart transplant recipients generally require more intense immunosuppressive therapy than most other solid organ transplant recipients, and this high baseline immunosuppression increases their propensity to develop infections and their likelihood of experiencing severe manifestations of infections,” Dr. Uriel commented.
“With COVID-19, in which the body’s inflammatory reaction appears to play a role in disease severity, there has been a question of whether immunosuppression may offer a protective effect,” he continued.
“This case series suggests that this is not the case, although this would need to be confirmed in larger studies,” he said.
Low threshold
Among the 22 patients who were admitted to the hospital, half were discharged home and four (18%) were still hospitalized at the end of the study.
Of the seven patients who died, two died at the study center, and five died in an outside institution.
“In the HT population, social distancing (or isolation), strict use of masks when in public, proper handwashing, and sanitization of surfaces are of paramount importance in the prevention of COVID-19 infection,” Dr. Uriel stated.
“In addition, we have restricted these patients’ contact with the hospital as much as possible during the pandemic,” he said.
However, “there should be a low threshold to hospitalize heart transplant patients who develop infection with COVID-19. Furthermore, in our series, outcomes were better for patients hospitalized at the transplant center; therefore, strong consideration should be given to transferring HT patients when hospitalized at another hospital,” he added.
The authors emphasized that COVID-19 patients “will require ongoing monitoring in the recovery phase, as an immunosuppression regimen is reintroduced and the consequences to the allograft itself become apparent.”
Vulnerable population
Commenting on the study, Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston, suggested that “in epidemiological terms, [the findings] might not look as bad as the way they are reflected in the paper.”
Given that Columbia is “one of the larger heart transplant centers in the U.S., following probably 1,000 patients, having only 22 out of perhaps thousands whom they transplanted or are actively following would actually represent a low serious infection rate,” said Dr. Mehra, who is also the executive director of the Center for Advanced Heart Disease at Brigham and Women’s Hospital and a professor of medicine at Harvard Medical School, also in Boston.
“We must not forget to emphasize that, when assessing these case fatality rates, we must look at the entire population at risk, not only the handful that we were able to observe,” explained Dr. Mehra, who was not involved with the study.
Moreover, the patients were “older and had comorbidities, with poor underlying kidney function and other complications, and underlying coronary artery disease in the transplanted heart,” so “it would not surprise me that they had such a high fatality rate, since they had a high degree of vulnerability,” he said.
Dr. Mehra, who is also the editor-in-chief of the Journal of Heart and Lung Transplantation, said that the journal has received manuscripts still in the review process that suggest different fatality rates than those found in the current case series.
However, he acknowledged that, because these are patients with serious vulnerability due to underlying heart disease, “you can’t be lackadaisical and need to do everything to decrease this vulnerability.”
The authors noted that, although their study did not show a protective effect from immunosuppression against COVID-19, further studies are needed to assess each individual immunosuppressive agent and provide a definitive answer.
The study was supported by a grant to one of the investigators from the National Heart, Lung, and Blood Institute. Dr. Uriel reports no relevant financial relationships. The other authors’ disclosures are listed in the publication. Dr. Mehra reports no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
COVID-19 infection is associated with a high risk for mortality in heart transplant (HT) recipients, a new case series suggests.
Investigators looked at data on 28 patients with a confirmed diagnosis of COVID-19 who received a HT between March 1, 2020, and April 24, 2020 and found a case-fatality rate of 25%.
“The high case fatality in our case series should alert physicians to the vulnerability of heart transplant recipients during the COVID-19 pandemic,” senior author Nir Uriel, MD, MSc, professor of medicine at Columbia University, New York, said in an interview.
“These patients require extra precautions to prevent the development of infection,” said Dr. Uriel, who is also a cardiologist at New York Presbyterian/Columbia University Irving Medical Center.
The study was published online May 13 in JAMA Cardiology.
Similar presentation
HT recipients can have several comorbidities after the procedure, including hypertension, diabetes, cardiac allograft vasculopathy, and ongoing immunosuppression, all of which can place them at risk for infection and adverse outcomes with COVID-19 infection, the authors wrote.
The researchers therefore embarked on a case series looking at 28 HT recipients with COVID-19 infection (median age, 64.0 years; interquartile range, 53.5-70.5; 79% male) to “describe the outcomes of recipients of HT who are chronically immunosuppressed and develop COVID-19 and raise important questions about the role of the immune system in the process.”
The median time from HT to study period was 8.6 (IQR, 4.2-14.5) years. Most patients had numerous comorbidities.
“The presentation of COVID-19 was similar to nontransplant patients with fever, dyspnea, cough, and GI symptoms,” Dr. Uriel reported.
No protective effect
Twenty-two patients (79%) required admission to the hospital, seven of whom (25%) required admission to the ICU and mechanical ventilation.
Despite the presence of immunosuppressive therapy, all patients had significant elevation of inflammatory biomarkers (median peak high-sensitivity C-reactive protein [hs-CRP], 11.83 mg/dL; IQR, 7.44-19.26; median peak interleukin [IL]-6, 105 pg/mL; IQR, 38-296).
Three-quarters had myocardial injury, with a median high-sensitivity troponin T of 0.055 (0.0205 - 0.1345) ng/mL.
Treatments of COVID-19 included hydroxychloroquine (18 patients; 78%), high-dose corticosteroids (eight patients; 47%), and IL-6 receptor antagonists (six patients; 26%).
Moreover, during hospitalization, mycophenolate mofetil was discontinued in most (70%) patients, and one-quarter had a reduction in their calcineurin inhibitor dose.
“Heart transplant recipients generally require more intense immunosuppressive therapy than most other solid organ transplant recipients, and this high baseline immunosuppression increases their propensity to develop infections and their likelihood of experiencing severe manifestations of infections,” Dr. Uriel commented.
“With COVID-19, in which the body’s inflammatory reaction appears to play a role in disease severity, there has been a question of whether immunosuppression may offer a protective effect,” he continued.
“This case series suggests that this is not the case, although this would need to be confirmed in larger studies,” he said.
Low threshold
Among the 22 patients who were admitted to the hospital, half were discharged home and four (18%) were still hospitalized at the end of the study.
Of the seven patients who died, two died at the study center, and five died in an outside institution.
“In the HT population, social distancing (or isolation), strict use of masks when in public, proper handwashing, and sanitization of surfaces are of paramount importance in the prevention of COVID-19 infection,” Dr. Uriel stated.
“In addition, we have restricted these patients’ contact with the hospital as much as possible during the pandemic,” he said.
However, “there should be a low threshold to hospitalize heart transplant patients who develop infection with COVID-19. Furthermore, in our series, outcomes were better for patients hospitalized at the transplant center; therefore, strong consideration should be given to transferring HT patients when hospitalized at another hospital,” he added.
The authors emphasized that COVID-19 patients “will require ongoing monitoring in the recovery phase, as an immunosuppression regimen is reintroduced and the consequences to the allograft itself become apparent.”
Vulnerable population
Commenting on the study, Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston, suggested that “in epidemiological terms, [the findings] might not look as bad as the way they are reflected in the paper.”
Given that Columbia is “one of the larger heart transplant centers in the U.S., following probably 1,000 patients, having only 22 out of perhaps thousands whom they transplanted or are actively following would actually represent a low serious infection rate,” said Dr. Mehra, who is also the executive director of the Center for Advanced Heart Disease at Brigham and Women’s Hospital and a professor of medicine at Harvard Medical School, also in Boston.
“We must not forget to emphasize that, when assessing these case fatality rates, we must look at the entire population at risk, not only the handful that we were able to observe,” explained Dr. Mehra, who was not involved with the study.
Moreover, the patients were “older and had comorbidities, with poor underlying kidney function and other complications, and underlying coronary artery disease in the transplanted heart,” so “it would not surprise me that they had such a high fatality rate, since they had a high degree of vulnerability,” he said.
Dr. Mehra, who is also the editor-in-chief of the Journal of Heart and Lung Transplantation, said that the journal has received manuscripts still in the review process that suggest different fatality rates than those found in the current case series.
However, he acknowledged that, because these are patients with serious vulnerability due to underlying heart disease, “you can’t be lackadaisical and need to do everything to decrease this vulnerability.”
The authors noted that, although their study did not show a protective effect from immunosuppression against COVID-19, further studies are needed to assess each individual immunosuppressive agent and provide a definitive answer.
The study was supported by a grant to one of the investigators from the National Heart, Lung, and Blood Institute. Dr. Uriel reports no relevant financial relationships. The other authors’ disclosures are listed in the publication. Dr. Mehra reports no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
COVID-19 infection is associated with a high risk for mortality in heart transplant (HT) recipients, a new case series suggests.
Investigators looked at data on 28 patients with a confirmed diagnosis of COVID-19 who received a HT between March 1, 2020, and April 24, 2020 and found a case-fatality rate of 25%.
“The high case fatality in our case series should alert physicians to the vulnerability of heart transplant recipients during the COVID-19 pandemic,” senior author Nir Uriel, MD, MSc, professor of medicine at Columbia University, New York, said in an interview.
“These patients require extra precautions to prevent the development of infection,” said Dr. Uriel, who is also a cardiologist at New York Presbyterian/Columbia University Irving Medical Center.
The study was published online May 13 in JAMA Cardiology.
Similar presentation
HT recipients can have several comorbidities after the procedure, including hypertension, diabetes, cardiac allograft vasculopathy, and ongoing immunosuppression, all of which can place them at risk for infection and adverse outcomes with COVID-19 infection, the authors wrote.
The researchers therefore embarked on a case series looking at 28 HT recipients with COVID-19 infection (median age, 64.0 years; interquartile range, 53.5-70.5; 79% male) to “describe the outcomes of recipients of HT who are chronically immunosuppressed and develop COVID-19 and raise important questions about the role of the immune system in the process.”
The median time from HT to study period was 8.6 (IQR, 4.2-14.5) years. Most patients had numerous comorbidities.
“The presentation of COVID-19 was similar to nontransplant patients with fever, dyspnea, cough, and GI symptoms,” Dr. Uriel reported.
No protective effect
Twenty-two patients (79%) required admission to the hospital, seven of whom (25%) required admission to the ICU and mechanical ventilation.
Despite the presence of immunosuppressive therapy, all patients had significant elevation of inflammatory biomarkers (median peak high-sensitivity C-reactive protein [hs-CRP], 11.83 mg/dL; IQR, 7.44-19.26; median peak interleukin [IL]-6, 105 pg/mL; IQR, 38-296).
Three-quarters had myocardial injury, with a median high-sensitivity troponin T of 0.055 (0.0205 - 0.1345) ng/mL.
Treatments of COVID-19 included hydroxychloroquine (18 patients; 78%), high-dose corticosteroids (eight patients; 47%), and IL-6 receptor antagonists (six patients; 26%).
Moreover, during hospitalization, mycophenolate mofetil was discontinued in most (70%) patients, and one-quarter had a reduction in their calcineurin inhibitor dose.
“Heart transplant recipients generally require more intense immunosuppressive therapy than most other solid organ transplant recipients, and this high baseline immunosuppression increases their propensity to develop infections and their likelihood of experiencing severe manifestations of infections,” Dr. Uriel commented.
“With COVID-19, in which the body’s inflammatory reaction appears to play a role in disease severity, there has been a question of whether immunosuppression may offer a protective effect,” he continued.
“This case series suggests that this is not the case, although this would need to be confirmed in larger studies,” he said.
Low threshold
Among the 22 patients who were admitted to the hospital, half were discharged home and four (18%) were still hospitalized at the end of the study.
Of the seven patients who died, two died at the study center, and five died in an outside institution.
“In the HT population, social distancing (or isolation), strict use of masks when in public, proper handwashing, and sanitization of surfaces are of paramount importance in the prevention of COVID-19 infection,” Dr. Uriel stated.
“In addition, we have restricted these patients’ contact with the hospital as much as possible during the pandemic,” he said.
However, “there should be a low threshold to hospitalize heart transplant patients who develop infection with COVID-19. Furthermore, in our series, outcomes were better for patients hospitalized at the transplant center; therefore, strong consideration should be given to transferring HT patients when hospitalized at another hospital,” he added.
The authors emphasized that COVID-19 patients “will require ongoing monitoring in the recovery phase, as an immunosuppression regimen is reintroduced and the consequences to the allograft itself become apparent.”
Vulnerable population
Commenting on the study, Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston, suggested that “in epidemiological terms, [the findings] might not look as bad as the way they are reflected in the paper.”
Given that Columbia is “one of the larger heart transplant centers in the U.S., following probably 1,000 patients, having only 22 out of perhaps thousands whom they transplanted or are actively following would actually represent a low serious infection rate,” said Dr. Mehra, who is also the executive director of the Center for Advanced Heart Disease at Brigham and Women’s Hospital and a professor of medicine at Harvard Medical School, also in Boston.
“We must not forget to emphasize that, when assessing these case fatality rates, we must look at the entire population at risk, not only the handful that we were able to observe,” explained Dr. Mehra, who was not involved with the study.
Moreover, the patients were “older and had comorbidities, with poor underlying kidney function and other complications, and underlying coronary artery disease in the transplanted heart,” so “it would not surprise me that they had such a high fatality rate, since they had a high degree of vulnerability,” he said.
Dr. Mehra, who is also the editor-in-chief of the Journal of Heart and Lung Transplantation, said that the journal has received manuscripts still in the review process that suggest different fatality rates than those found in the current case series.
However, he acknowledged that, because these are patients with serious vulnerability due to underlying heart disease, “you can’t be lackadaisical and need to do everything to decrease this vulnerability.”
The authors noted that, although their study did not show a protective effect from immunosuppression against COVID-19, further studies are needed to assess each individual immunosuppressive agent and provide a definitive answer.
The study was supported by a grant to one of the investigators from the National Heart, Lung, and Blood Institute. Dr. Uriel reports no relevant financial relationships. The other authors’ disclosures are listed in the publication. Dr. Mehra reports no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Deprescribing hypertension meds can be safe in older patients
Some patients aged 80 years or older can potentially cut back on their number of antihypertensive meds, under physician guidance, without an important loss of blood pressure (BP) control, researchers concluded based on their randomized multicenter trial.
Deprescription of one of at least two antihypertensive meds in such patients was found noninferior to usual care in keeping systolic BP below 150 mm Hg at 12 weeks, in the study that randomly assigned only patients who were considered appropriate for BP-med reduction by their primary care physicians.
Major trials that have shaped some contemporary hypertension guidelines, notably SPRINT, in general have not included such older patients with hypertension along with other chronic conditions, such as diabetes or a history of stroke. So “it’s difficult to know whether their data are relevant for frail, multimorbid patients. In fact, the guidelines say you should use some clinical judgment when applying the results of SPRINT to the kind of patients seen in clinical practice,” James P. Sheppard, PhD, of University of Oxford (England) said in an interview.
The current study, called Optimising Treatment for Mild Systolic Hypertension in the Elderly (OPTIMISE), entered “patients in whom the benefits of taking blood pressure-lowering treatments might start to be outweighed by the potential harms,” Dr. Sheppard said.
The trial is meant to provide something of an otherwise-scant evidence base for how to deprescribe antihypertensive medications, said Dr. Sheppard, who is lead author on the report published May 25 in JAMA.
Of the trial’s 282 patients randomly assigned to the drug-reduction group, 86.4% reached the primary endpoint goal of systolic BP less than 150 mm Hg, compared with 87.7% of the 287 patients on usual care, a difference which in adjusted analysis met the predetermined standard for noninferiority.
The intervention group reduced its number of antihypertensive agents by a mean of 0.6 per patient, which the authors described as “a modest reduction.” However, they noted, drugs that were taken away could be reintroduced as judged necessary by the physicians, yet most of the group sustained their reductions until the end of the 12 weeks.
Had the primary endpoint instead specified a threshold of 130 mm Hg for BP control, which is more consistent with SPRINT and some guidelines in the United States, “the deprescribing strategy would have failed to be considered noninferior to usual care” as calculated by the OPTIMISE authors themselves, observed an accompanying editorial.
The 150 mm Hg threshold chosen by the trialists for the primary endpoint, therefore, “was somewhat of a low bar,” wrote Eric D. Peterson, MD, MPH, of Duke University, Durham, N.C., and Michael W. Rich, MD, of Washington University School of Medicine, St. Louis, Mo.
“Here in the UK it wouldn’t be considered a low bar,” Dr. Sheppard said in an interview. The National Institute for Health and Care Excellence guidelines in Britain “recommends that you treat people over the age of 18 regardless of whether they have any other conditions and to 150 mm Hg systolic.”
The study’s general practitioners, he said, “did what we told them to do, and as a result, two-thirds of the patients were able to reduce their medications. If we had a lower threshold for treatment, it’s possible that more patients might have had medications reintroduced. I think you still could have potentially ended up with a noninferior result.”
Participating physicians were instructed to enroll only “patients who, in their opinion, might potentially benefit from medication reduction due to one or more of the following existing characteristics: polypharmacy, comorbidity, nonadherence or dislike of medicines, or frailty,” the report notes.
They chose which antihypertensives would be dropped for each patient and “were given a medication reduction algorithm to assist with this decision.” Physicians also followed a guide for monitoring for safety issues and were told to reintroduce medications if systolic BP exceeded 150 mm Hg or diastolic BP rose above 90 mm Hg for more than 1 week or in the event of adverse events or signs of accelerated hypertension, the group wrote.
In the deprescription group, the mean systolic BP rose 4.3 points from baseline to 12 weeks, from 129.4 to 133.7 mm Hg. For those given usual care, mean systolic BP went from 130.5 to 130.8 mm Hg. Adjusted, the mean change in systolic BP was 3.4 mm Hg greater (P = .005) in the intervention group. The corresponding adjusted mean change in diastolic BP was a 2.2 mm Hg increase in the intervention group (P = .001).
Although the difference seems minimal, wrote Dr. Peterson and Dr. Rich, “such differences in BP can potentially lead to long-term differences in outcomes at the population level.”
Also, they pointed out, only about 10% of patients screened for enrollment actually entered the study, which brings into question the study’s generalizability, and “patients in the trial had relatively well-controlled BP at baseline.”
Dr. Sheppard said patients in the original screened population, taken from a national database, were directly invited to participate en masse by conventional mail, based on broad inclusion criteria. Far more than the number needed were invited, and nearly all of those excluded from the study had simply not responded to the invitation.
As for greater increases in systolic and diastolic pressures in the deprescribing group, the OPTIMISE authors acknowledged that “caution should be exercised when adopting this approach in routine clinical practice.”
His own view, Dr. Sheppard said, “is that there are some patients who will definitely benefit from intensive blood pressure lowering like you saw in the SPRINT trial. And there’s other patients who will benefit from deprescribing and having a slightly higher target. Those sorts of things very much need to be individualized at the patient level.”
And ideally, he added, clinicians in practice should probably be even more selective in choosing patients for a deprescribing strategy, “and focus on people who are at the highest risk of adverse events.”
Dr. Sheppard has disclosed no relevant financial relationships; disclosures for the other authors are in the report. Dr. Peterson disclosed receiving personal fees from Cerner and Livongo and grants and personal fees from AstraZeneca, Janssen, and Amgen; Dr. Rick has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Some patients aged 80 years or older can potentially cut back on their number of antihypertensive meds, under physician guidance, without an important loss of blood pressure (BP) control, researchers concluded based on their randomized multicenter trial.
Deprescription of one of at least two antihypertensive meds in such patients was found noninferior to usual care in keeping systolic BP below 150 mm Hg at 12 weeks, in the study that randomly assigned only patients who were considered appropriate for BP-med reduction by their primary care physicians.
Major trials that have shaped some contemporary hypertension guidelines, notably SPRINT, in general have not included such older patients with hypertension along with other chronic conditions, such as diabetes or a history of stroke. So “it’s difficult to know whether their data are relevant for frail, multimorbid patients. In fact, the guidelines say you should use some clinical judgment when applying the results of SPRINT to the kind of patients seen in clinical practice,” James P. Sheppard, PhD, of University of Oxford (England) said in an interview.
The current study, called Optimising Treatment for Mild Systolic Hypertension in the Elderly (OPTIMISE), entered “patients in whom the benefits of taking blood pressure-lowering treatments might start to be outweighed by the potential harms,” Dr. Sheppard said.
The trial is meant to provide something of an otherwise-scant evidence base for how to deprescribe antihypertensive medications, said Dr. Sheppard, who is lead author on the report published May 25 in JAMA.
Of the trial’s 282 patients randomly assigned to the drug-reduction group, 86.4% reached the primary endpoint goal of systolic BP less than 150 mm Hg, compared with 87.7% of the 287 patients on usual care, a difference which in adjusted analysis met the predetermined standard for noninferiority.
The intervention group reduced its number of antihypertensive agents by a mean of 0.6 per patient, which the authors described as “a modest reduction.” However, they noted, drugs that were taken away could be reintroduced as judged necessary by the physicians, yet most of the group sustained their reductions until the end of the 12 weeks.
Had the primary endpoint instead specified a threshold of 130 mm Hg for BP control, which is more consistent with SPRINT and some guidelines in the United States, “the deprescribing strategy would have failed to be considered noninferior to usual care” as calculated by the OPTIMISE authors themselves, observed an accompanying editorial.
The 150 mm Hg threshold chosen by the trialists for the primary endpoint, therefore, “was somewhat of a low bar,” wrote Eric D. Peterson, MD, MPH, of Duke University, Durham, N.C., and Michael W. Rich, MD, of Washington University School of Medicine, St. Louis, Mo.
“Here in the UK it wouldn’t be considered a low bar,” Dr. Sheppard said in an interview. The National Institute for Health and Care Excellence guidelines in Britain “recommends that you treat people over the age of 18 regardless of whether they have any other conditions and to 150 mm Hg systolic.”
The study’s general practitioners, he said, “did what we told them to do, and as a result, two-thirds of the patients were able to reduce their medications. If we had a lower threshold for treatment, it’s possible that more patients might have had medications reintroduced. I think you still could have potentially ended up with a noninferior result.”
Participating physicians were instructed to enroll only “patients who, in their opinion, might potentially benefit from medication reduction due to one or more of the following existing characteristics: polypharmacy, comorbidity, nonadherence or dislike of medicines, or frailty,” the report notes.
They chose which antihypertensives would be dropped for each patient and “were given a medication reduction algorithm to assist with this decision.” Physicians also followed a guide for monitoring for safety issues and were told to reintroduce medications if systolic BP exceeded 150 mm Hg or diastolic BP rose above 90 mm Hg for more than 1 week or in the event of adverse events or signs of accelerated hypertension, the group wrote.
In the deprescription group, the mean systolic BP rose 4.3 points from baseline to 12 weeks, from 129.4 to 133.7 mm Hg. For those given usual care, mean systolic BP went from 130.5 to 130.8 mm Hg. Adjusted, the mean change in systolic BP was 3.4 mm Hg greater (P = .005) in the intervention group. The corresponding adjusted mean change in diastolic BP was a 2.2 mm Hg increase in the intervention group (P = .001).
Although the difference seems minimal, wrote Dr. Peterson and Dr. Rich, “such differences in BP can potentially lead to long-term differences in outcomes at the population level.”
Also, they pointed out, only about 10% of patients screened for enrollment actually entered the study, which brings into question the study’s generalizability, and “patients in the trial had relatively well-controlled BP at baseline.”
Dr. Sheppard said patients in the original screened population, taken from a national database, were directly invited to participate en masse by conventional mail, based on broad inclusion criteria. Far more than the number needed were invited, and nearly all of those excluded from the study had simply not responded to the invitation.
As for greater increases in systolic and diastolic pressures in the deprescribing group, the OPTIMISE authors acknowledged that “caution should be exercised when adopting this approach in routine clinical practice.”
His own view, Dr. Sheppard said, “is that there are some patients who will definitely benefit from intensive blood pressure lowering like you saw in the SPRINT trial. And there’s other patients who will benefit from deprescribing and having a slightly higher target. Those sorts of things very much need to be individualized at the patient level.”
And ideally, he added, clinicians in practice should probably be even more selective in choosing patients for a deprescribing strategy, “and focus on people who are at the highest risk of adverse events.”
Dr. Sheppard has disclosed no relevant financial relationships; disclosures for the other authors are in the report. Dr. Peterson disclosed receiving personal fees from Cerner and Livongo and grants and personal fees from AstraZeneca, Janssen, and Amgen; Dr. Rick has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Some patients aged 80 years or older can potentially cut back on their number of antihypertensive meds, under physician guidance, without an important loss of blood pressure (BP) control, researchers concluded based on their randomized multicenter trial.
Deprescription of one of at least two antihypertensive meds in such patients was found noninferior to usual care in keeping systolic BP below 150 mm Hg at 12 weeks, in the study that randomly assigned only patients who were considered appropriate for BP-med reduction by their primary care physicians.
Major trials that have shaped some contemporary hypertension guidelines, notably SPRINT, in general have not included such older patients with hypertension along with other chronic conditions, such as diabetes or a history of stroke. So “it’s difficult to know whether their data are relevant for frail, multimorbid patients. In fact, the guidelines say you should use some clinical judgment when applying the results of SPRINT to the kind of patients seen in clinical practice,” James P. Sheppard, PhD, of University of Oxford (England) said in an interview.
The current study, called Optimising Treatment for Mild Systolic Hypertension in the Elderly (OPTIMISE), entered “patients in whom the benefits of taking blood pressure-lowering treatments might start to be outweighed by the potential harms,” Dr. Sheppard said.
The trial is meant to provide something of an otherwise-scant evidence base for how to deprescribe antihypertensive medications, said Dr. Sheppard, who is lead author on the report published May 25 in JAMA.
Of the trial’s 282 patients randomly assigned to the drug-reduction group, 86.4% reached the primary endpoint goal of systolic BP less than 150 mm Hg, compared with 87.7% of the 287 patients on usual care, a difference which in adjusted analysis met the predetermined standard for noninferiority.
The intervention group reduced its number of antihypertensive agents by a mean of 0.6 per patient, which the authors described as “a modest reduction.” However, they noted, drugs that were taken away could be reintroduced as judged necessary by the physicians, yet most of the group sustained their reductions until the end of the 12 weeks.
Had the primary endpoint instead specified a threshold of 130 mm Hg for BP control, which is more consistent with SPRINT and some guidelines in the United States, “the deprescribing strategy would have failed to be considered noninferior to usual care” as calculated by the OPTIMISE authors themselves, observed an accompanying editorial.
The 150 mm Hg threshold chosen by the trialists for the primary endpoint, therefore, “was somewhat of a low bar,” wrote Eric D. Peterson, MD, MPH, of Duke University, Durham, N.C., and Michael W. Rich, MD, of Washington University School of Medicine, St. Louis, Mo.
“Here in the UK it wouldn’t be considered a low bar,” Dr. Sheppard said in an interview. The National Institute for Health and Care Excellence guidelines in Britain “recommends that you treat people over the age of 18 regardless of whether they have any other conditions and to 150 mm Hg systolic.”
The study’s general practitioners, he said, “did what we told them to do, and as a result, two-thirds of the patients were able to reduce their medications. If we had a lower threshold for treatment, it’s possible that more patients might have had medications reintroduced. I think you still could have potentially ended up with a noninferior result.”
Participating physicians were instructed to enroll only “patients who, in their opinion, might potentially benefit from medication reduction due to one or more of the following existing characteristics: polypharmacy, comorbidity, nonadherence or dislike of medicines, or frailty,” the report notes.
They chose which antihypertensives would be dropped for each patient and “were given a medication reduction algorithm to assist with this decision.” Physicians also followed a guide for monitoring for safety issues and were told to reintroduce medications if systolic BP exceeded 150 mm Hg or diastolic BP rose above 90 mm Hg for more than 1 week or in the event of adverse events or signs of accelerated hypertension, the group wrote.
In the deprescription group, the mean systolic BP rose 4.3 points from baseline to 12 weeks, from 129.4 to 133.7 mm Hg. For those given usual care, mean systolic BP went from 130.5 to 130.8 mm Hg. Adjusted, the mean change in systolic BP was 3.4 mm Hg greater (P = .005) in the intervention group. The corresponding adjusted mean change in diastolic BP was a 2.2 mm Hg increase in the intervention group (P = .001).
Although the difference seems minimal, wrote Dr. Peterson and Dr. Rich, “such differences in BP can potentially lead to long-term differences in outcomes at the population level.”
Also, they pointed out, only about 10% of patients screened for enrollment actually entered the study, which brings into question the study’s generalizability, and “patients in the trial had relatively well-controlled BP at baseline.”
Dr. Sheppard said patients in the original screened population, taken from a national database, were directly invited to participate en masse by conventional mail, based on broad inclusion criteria. Far more than the number needed were invited, and nearly all of those excluded from the study had simply not responded to the invitation.
As for greater increases in systolic and diastolic pressures in the deprescribing group, the OPTIMISE authors acknowledged that “caution should be exercised when adopting this approach in routine clinical practice.”
His own view, Dr. Sheppard said, “is that there are some patients who will definitely benefit from intensive blood pressure lowering like you saw in the SPRINT trial. And there’s other patients who will benefit from deprescribing and having a slightly higher target. Those sorts of things very much need to be individualized at the patient level.”
And ideally, he added, clinicians in practice should probably be even more selective in choosing patients for a deprescribing strategy, “and focus on people who are at the highest risk of adverse events.”
Dr. Sheppard has disclosed no relevant financial relationships; disclosures for the other authors are in the report. Dr. Peterson disclosed receiving personal fees from Cerner and Livongo and grants and personal fees from AstraZeneca, Janssen, and Amgen; Dr. Rick has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FDA okays emergency use for Impella RP in COVID-19 right heart failure
The Food and Drug Administration issued an emergency use authorization for use of the Impella RP heart pump system in COVID-19 patients with right heart failure or decompensation, Abiomed announced June 1.
“Based on extrapolation of data from the approved indication and reported clinical experience, FDA has concluded that the Impella RP may be effective at providing temporary right ventricular support for the treatment of acute right heart failure or decompensation caused by COVID-19 complications, including PE [pulmonary embolism],” the letter noted.
It cited, for example, use of the temporary heart pump in a 59-year-old woman suffering from COVID-19 who went into right ventricular failure and became hypotensive after an acute PE was removed. After placement of the device, the patient experienced a “dramatic and immediate” improvement in arterial pressure and the device was removed on the fifth day, according to Amir Kaki, MD, and Ted Schreiber, MD, of Ascension St. John Hospital, Detroit, whose review of the case has been posted online.
“Acute pulmonary embolism is clearly being recognized as a life-threatening manifestation of COVID-19. Impella RP is an important tool to help cardiologists save lives during this pandemic,” Dr. Kaki said in the letter. “As we have demonstrated in our series of patients, early recognition of right ventricular dysfunction and early placement of the Impella RP for patients who are hypotensive can be lifesaving.”
Other data cited in support of the Impella RP emergency use authorization (EUA) include a 2019 series of hemodynamically unstable patients with PE in Japan and a 2017 case report of a 47-year-old man with right ventricular failure, profound shock, and a massive PE.
The FDA granted premarket approval of the Impella RP system in 2017 to provide temporary right ventricular support for up to 14 days in patients with a body surface area of at least 1.5 m2 who develop acute right heart failure or decompensation following left ventricular assist device implantation, MI, heart transplant, or open-heart surgery.
The EUA indication for the Impella RP system is to provide temporary right ventricular support for up to 14 days in critical care patients with a body surface area of at least 1.5 m2 for the treatment of acute right heart failure or decompensation caused by complications related to COVID-19, including PE.
The Impella RP is authorized only for emergency use under the EUA and only for the duration of the circumstances justifying use of EUAs, the letter noted.
Last year, concerns were raised about off-indication use after interim results from a postapproval study suggested a higher risk for death than seen in premarket studies treated with the temporary heart pump.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration issued an emergency use authorization for use of the Impella RP heart pump system in COVID-19 patients with right heart failure or decompensation, Abiomed announced June 1.
“Based on extrapolation of data from the approved indication and reported clinical experience, FDA has concluded that the Impella RP may be effective at providing temporary right ventricular support for the treatment of acute right heart failure or decompensation caused by COVID-19 complications, including PE [pulmonary embolism],” the letter noted.
It cited, for example, use of the temporary heart pump in a 59-year-old woman suffering from COVID-19 who went into right ventricular failure and became hypotensive after an acute PE was removed. After placement of the device, the patient experienced a “dramatic and immediate” improvement in arterial pressure and the device was removed on the fifth day, according to Amir Kaki, MD, and Ted Schreiber, MD, of Ascension St. John Hospital, Detroit, whose review of the case has been posted online.
“Acute pulmonary embolism is clearly being recognized as a life-threatening manifestation of COVID-19. Impella RP is an important tool to help cardiologists save lives during this pandemic,” Dr. Kaki said in the letter. “As we have demonstrated in our series of patients, early recognition of right ventricular dysfunction and early placement of the Impella RP for patients who are hypotensive can be lifesaving.”
Other data cited in support of the Impella RP emergency use authorization (EUA) include a 2019 series of hemodynamically unstable patients with PE in Japan and a 2017 case report of a 47-year-old man with right ventricular failure, profound shock, and a massive PE.
The FDA granted premarket approval of the Impella RP system in 2017 to provide temporary right ventricular support for up to 14 days in patients with a body surface area of at least 1.5 m2 who develop acute right heart failure or decompensation following left ventricular assist device implantation, MI, heart transplant, or open-heart surgery.
The EUA indication for the Impella RP system is to provide temporary right ventricular support for up to 14 days in critical care patients with a body surface area of at least 1.5 m2 for the treatment of acute right heart failure or decompensation caused by complications related to COVID-19, including PE.
The Impella RP is authorized only for emergency use under the EUA and only for the duration of the circumstances justifying use of EUAs, the letter noted.
Last year, concerns were raised about off-indication use after interim results from a postapproval study suggested a higher risk for death than seen in premarket studies treated with the temporary heart pump.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration issued an emergency use authorization for use of the Impella RP heart pump system in COVID-19 patients with right heart failure or decompensation, Abiomed announced June 1.
“Based on extrapolation of data from the approved indication and reported clinical experience, FDA has concluded that the Impella RP may be effective at providing temporary right ventricular support for the treatment of acute right heart failure or decompensation caused by COVID-19 complications, including PE [pulmonary embolism],” the letter noted.
It cited, for example, use of the temporary heart pump in a 59-year-old woman suffering from COVID-19 who went into right ventricular failure and became hypotensive after an acute PE was removed. After placement of the device, the patient experienced a “dramatic and immediate” improvement in arterial pressure and the device was removed on the fifth day, according to Amir Kaki, MD, and Ted Schreiber, MD, of Ascension St. John Hospital, Detroit, whose review of the case has been posted online.
“Acute pulmonary embolism is clearly being recognized as a life-threatening manifestation of COVID-19. Impella RP is an important tool to help cardiologists save lives during this pandemic,” Dr. Kaki said in the letter. “As we have demonstrated in our series of patients, early recognition of right ventricular dysfunction and early placement of the Impella RP for patients who are hypotensive can be lifesaving.”
Other data cited in support of the Impella RP emergency use authorization (EUA) include a 2019 series of hemodynamically unstable patients with PE in Japan and a 2017 case report of a 47-year-old man with right ventricular failure, profound shock, and a massive PE.
The FDA granted premarket approval of the Impella RP system in 2017 to provide temporary right ventricular support for up to 14 days in patients with a body surface area of at least 1.5 m2 who develop acute right heart failure or decompensation following left ventricular assist device implantation, MI, heart transplant, or open-heart surgery.
The EUA indication for the Impella RP system is to provide temporary right ventricular support for up to 14 days in critical care patients with a body surface area of at least 1.5 m2 for the treatment of acute right heart failure or decompensation caused by complications related to COVID-19, including PE.
The Impella RP is authorized only for emergency use under the EUA and only for the duration of the circumstances justifying use of EUAs, the letter noted.
Last year, concerns were raised about off-indication use after interim results from a postapproval study suggested a higher risk for death than seen in premarket studies treated with the temporary heart pump.
A version of this article originally appeared on Medscape.com.
Latest from ISCHEMIA: Worse outcomes in patients with intermediate left main disease on CCTA
Patients in the landmark ISCHEMIA trial with intermediate left main disease had a greater extent of coronary artery disease on invasive angiography, indicating greater atherosclerotic burden. They also had worse prognosis with a higher risk of cardiovascular events.
“Many times, we are looking at results as to whether patients have left main disease or not,” Sripal Bangalore, MD, said during the Society for Cardiovascular Angiography & Interventions virtual annual scientific sessions. “Here, we are showing that it’s not black and white; there are shades of gray. If a patient has intermediate left main disease, the prognosis is worse. That’s very important information we need to convey to our referrals also, because many times they may just look at the bottom line and say, ‘there is no left main disease.’ But here, we’re seeing that even having intermediate left main disease has significantly worse prognosis. We need to take that seriously.”
Prior studies show that patients with significant left main disease (LMD; defined as 50% or greater stenosis on coronary CT angiography [CCTA]) have a high risk of cardiovascular events and guidelines recommend revascularization to improve survival, said Dr. Bangalore, an interventional cardiologist at New York University Langone Health. However, the impact of intermediate LMD (defined as 25%-49% stenosis on CCTA) on outcomes is unclear.
Members of the ISCHEMIA (International Study of Comparative Health Effectiveness with Medical and Invasive Approaches) research group randomized 5,179 participants to an initial invasive or conservative strategy. The main results showed that immediate revascularization in patients with stable ischemic heart disease provided no reduction in cardiovascular endpoints through 4 years of follow-up, compared with initial optimal medical therapy alone.
‘Discordance’ revealed in imaging modalities
For the current analysis, named the ISCHEMIA Intermediate LM Substudy, those who underwent coronary CCTA comprise the LMD substudy cohort. The objective was to evaluate clinical and quality of life outcomes in patients with and without intermediate left main disease on coronary CT and to evaluate the impact of treatment strategy on those outcomes across subgroups.
At baseline, these patients were categorized into those with and without intermediate LMD as determined by a core lab. Patients with LMD of 50% or greater, those with prior coronary artery bypass graft surgery, and those with nonevaluable or missing data on LM stenosis were excluded.
Among the 3,913 ISCHEMIA participants who underwent CCTA, 3,699 satisfied the inclusion criteria. Of these patients, 962 (26%) had intermediate LMD and 2,737 (74%) did not.
The researchers observed no significant differences in baseline characteristics between patients with and without LMD. However, patients with intermediate LMD tended to be older, and a greater proportion had hypertension and diabetes. Stress test characteristics were also similar between patients with and without LMD. However, patients with intermediate LMD tended toward a greater severity of severe ischemia.
This was also true for anatomic disease on CCTA. A higher proportion of patients with intermediate LMD had triple-vessel disease (61%-62%, compared with 36%-40% along those without intermediate LMD). In addition, a higher proportion of patients with intermediate LMD had stenosis in the proximal left anterior artery descending (LAD) artery (65% vs. 39% among those without intermediate LMD).
On analysis limited to 1,846 patients who underwent invasive angiography treatment in the main ISCHEMIA trial, 7% of those who were categorized into the intermediate LMD group were found to have LMD disease of 50% or greater, compared with 1.4% of patients who were categorized as not having intermediate LMD. “This goes to show this discordance between the two modalities [CCTA and coronary angiography], and I think we have to be careful,” said Dr. Bangalore, who also directs NYU Langone’s Cardiac Catheterization Laboratory. “There may be patients with left main disease, even if the CCTA says it’s not at 25%-29% [stenosis].”
The researchers found that, among patients who underwent invasive angiography, a greater proportion of those who were categorized into the LMD group had proximal LAD disease (43% vs. 33% among those who were categorized into the nonintermediate LMD group), triple-vessel disease (47% vs. 35%), a greater extent of coronary artery disease as denoted by a higher SYNTAX score (21 vs. 15), and a higher proportion underwent coronary artery bypass graft surgery (32% vs. 18%).
Intermediate LMD linked to worse outcomes
After the researchers adjusted for baseline differences between the two groups in overall substudy cohort, they found that intermediate LMD severity was an independent predictor of the primary composite endpoint of cardiovascular death, MI, hospitalization for unstable angina, heart failure, and resuscitated cardiac arrest (hazard ratio, 1.31; P = .0123); cardiovascular death/MI/stroke (HR, 1.30; P = .0143); procedural primary MI (HR, 1.64; P = .0487); heart failure (HR, 2.06; P = .0239); and stroke (HR, 1.82, P = .0362).
“We then looked to see if there is a treatment difference, a treatment effect based on whether patients had intermediate LMD,” Dr. Bangalore said. “Most of the P values were not significant. The results are very consistent with what we saw in the main analysis: not a significant difference between invasive and conservative strategy. We do see some differences, though. An invasive strategy was associated with a significantly higher risk of procedural MI [2.9% vs. 1.5%], but a significantly lower risk of nonprocedural MI [–6.4% vs. –2%].”
Dr. Bangalore added that there was significant benefit of the invasive strategy in reducing angina and improving quality of life based on the Seattle Angina Questionnaire-7. “This result was durable up to 48 months of follow-up, whether the patient had intermediate left main disease or not. These results were dependent on baseline angina status. The benefit of invasive strategy was mainly in patients who had daily, weekly, and monthly angina, and no benefit in patients with no angina; there was no interaction based on intermediate left main status.”
Dr. Bangalore emphasized that the original ISCHEMIA trial excluded patients with severe left main disease by design. “But patients with intermediate left main disease in ISCHEMIA tended to have a greater extent of coronary artery disease, indicating greater atherosclerotic burden. I don’t think that’s any surprise. They had a worse prognosis with higher risk of cardiovascular events but similar quality of life, including angina-specific quality of life.”
The key clinical message, he said, is that patients with intermediate LMD face an increased risk of cardiovascular events. “I think we have to be aggressive in trying to reduce their risk with medical therapy, etc.,” he said. “If they are symptomatic, ISCHEMIA tells us that patients have two options. They can choose an invasive strategy, because clearly there is a benefit. You have a significant benefit at making you feel better and potentially reducing the risk of spontaneous MI over a period of time. Or, you can try medical therapy first. If you do see some left main disease, it’s showing the general burden of atherosclerosis disease in those patients. I think that’s the critical message, that we have to be very aggressive with these patients.”
A call for more imaging studies
An invited panelist, Timothy D. Henry, MD, said that the results of the ISCHEMIA substudy should stimulate further research. “With an intermediate lesion, clearly the interventional group did better, and it wasn’t symptom related,” said Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research and Education at the Christ Hospital in Cincinnati. “So even if you do medical therapy, you’re not going to really find it out. In my mind, this should stimulate us to do more imaging of the left main that are moderate lesions, and follow this up as an independent study. I think this is a really important finding.”
ISCHEMIA was supported by grants from the National Heart, Lung, and Blood Institute. Dr. Bangalore disclosed that he is a member of the advisory board and/or a board member for Meril, SMT, Pfizer, Amgen, Biotronik, and Abbott. He also is a consultant for Reata Pharmaceuticals.
SOURCE: Bangalore S et al. SCAI 2020, Abstract 11656.
Patients in the landmark ISCHEMIA trial with intermediate left main disease had a greater extent of coronary artery disease on invasive angiography, indicating greater atherosclerotic burden. They also had worse prognosis with a higher risk of cardiovascular events.
“Many times, we are looking at results as to whether patients have left main disease or not,” Sripal Bangalore, MD, said during the Society for Cardiovascular Angiography & Interventions virtual annual scientific sessions. “Here, we are showing that it’s not black and white; there are shades of gray. If a patient has intermediate left main disease, the prognosis is worse. That’s very important information we need to convey to our referrals also, because many times they may just look at the bottom line and say, ‘there is no left main disease.’ But here, we’re seeing that even having intermediate left main disease has significantly worse prognosis. We need to take that seriously.”
Prior studies show that patients with significant left main disease (LMD; defined as 50% or greater stenosis on coronary CT angiography [CCTA]) have a high risk of cardiovascular events and guidelines recommend revascularization to improve survival, said Dr. Bangalore, an interventional cardiologist at New York University Langone Health. However, the impact of intermediate LMD (defined as 25%-49% stenosis on CCTA) on outcomes is unclear.
Members of the ISCHEMIA (International Study of Comparative Health Effectiveness with Medical and Invasive Approaches) research group randomized 5,179 participants to an initial invasive or conservative strategy. The main results showed that immediate revascularization in patients with stable ischemic heart disease provided no reduction in cardiovascular endpoints through 4 years of follow-up, compared with initial optimal medical therapy alone.
‘Discordance’ revealed in imaging modalities
For the current analysis, named the ISCHEMIA Intermediate LM Substudy, those who underwent coronary CCTA comprise the LMD substudy cohort. The objective was to evaluate clinical and quality of life outcomes in patients with and without intermediate left main disease on coronary CT and to evaluate the impact of treatment strategy on those outcomes across subgroups.
At baseline, these patients were categorized into those with and without intermediate LMD as determined by a core lab. Patients with LMD of 50% or greater, those with prior coronary artery bypass graft surgery, and those with nonevaluable or missing data on LM stenosis were excluded.
Among the 3,913 ISCHEMIA participants who underwent CCTA, 3,699 satisfied the inclusion criteria. Of these patients, 962 (26%) had intermediate LMD and 2,737 (74%) did not.
The researchers observed no significant differences in baseline characteristics between patients with and without LMD. However, patients with intermediate LMD tended to be older, and a greater proportion had hypertension and diabetes. Stress test characteristics were also similar between patients with and without LMD. However, patients with intermediate LMD tended toward a greater severity of severe ischemia.
This was also true for anatomic disease on CCTA. A higher proportion of patients with intermediate LMD had triple-vessel disease (61%-62%, compared with 36%-40% along those without intermediate LMD). In addition, a higher proportion of patients with intermediate LMD had stenosis in the proximal left anterior artery descending (LAD) artery (65% vs. 39% among those without intermediate LMD).
On analysis limited to 1,846 patients who underwent invasive angiography treatment in the main ISCHEMIA trial, 7% of those who were categorized into the intermediate LMD group were found to have LMD disease of 50% or greater, compared with 1.4% of patients who were categorized as not having intermediate LMD. “This goes to show this discordance between the two modalities [CCTA and coronary angiography], and I think we have to be careful,” said Dr. Bangalore, who also directs NYU Langone’s Cardiac Catheterization Laboratory. “There may be patients with left main disease, even if the CCTA says it’s not at 25%-29% [stenosis].”
The researchers found that, among patients who underwent invasive angiography, a greater proportion of those who were categorized into the LMD group had proximal LAD disease (43% vs. 33% among those who were categorized into the nonintermediate LMD group), triple-vessel disease (47% vs. 35%), a greater extent of coronary artery disease as denoted by a higher SYNTAX score (21 vs. 15), and a higher proportion underwent coronary artery bypass graft surgery (32% vs. 18%).
Intermediate LMD linked to worse outcomes
After the researchers adjusted for baseline differences between the two groups in overall substudy cohort, they found that intermediate LMD severity was an independent predictor of the primary composite endpoint of cardiovascular death, MI, hospitalization for unstable angina, heart failure, and resuscitated cardiac arrest (hazard ratio, 1.31; P = .0123); cardiovascular death/MI/stroke (HR, 1.30; P = .0143); procedural primary MI (HR, 1.64; P = .0487); heart failure (HR, 2.06; P = .0239); and stroke (HR, 1.82, P = .0362).
“We then looked to see if there is a treatment difference, a treatment effect based on whether patients had intermediate LMD,” Dr. Bangalore said. “Most of the P values were not significant. The results are very consistent with what we saw in the main analysis: not a significant difference between invasive and conservative strategy. We do see some differences, though. An invasive strategy was associated with a significantly higher risk of procedural MI [2.9% vs. 1.5%], but a significantly lower risk of nonprocedural MI [–6.4% vs. –2%].”
Dr. Bangalore added that there was significant benefit of the invasive strategy in reducing angina and improving quality of life based on the Seattle Angina Questionnaire-7. “This result was durable up to 48 months of follow-up, whether the patient had intermediate left main disease or not. These results were dependent on baseline angina status. The benefit of invasive strategy was mainly in patients who had daily, weekly, and monthly angina, and no benefit in patients with no angina; there was no interaction based on intermediate left main status.”
Dr. Bangalore emphasized that the original ISCHEMIA trial excluded patients with severe left main disease by design. “But patients with intermediate left main disease in ISCHEMIA tended to have a greater extent of coronary artery disease, indicating greater atherosclerotic burden. I don’t think that’s any surprise. They had a worse prognosis with higher risk of cardiovascular events but similar quality of life, including angina-specific quality of life.”
The key clinical message, he said, is that patients with intermediate LMD face an increased risk of cardiovascular events. “I think we have to be aggressive in trying to reduce their risk with medical therapy, etc.,” he said. “If they are symptomatic, ISCHEMIA tells us that patients have two options. They can choose an invasive strategy, because clearly there is a benefit. You have a significant benefit at making you feel better and potentially reducing the risk of spontaneous MI over a period of time. Or, you can try medical therapy first. If you do see some left main disease, it’s showing the general burden of atherosclerosis disease in those patients. I think that’s the critical message, that we have to be very aggressive with these patients.”
A call for more imaging studies
An invited panelist, Timothy D. Henry, MD, said that the results of the ISCHEMIA substudy should stimulate further research. “With an intermediate lesion, clearly the interventional group did better, and it wasn’t symptom related,” said Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research and Education at the Christ Hospital in Cincinnati. “So even if you do medical therapy, you’re not going to really find it out. In my mind, this should stimulate us to do more imaging of the left main that are moderate lesions, and follow this up as an independent study. I think this is a really important finding.”
ISCHEMIA was supported by grants from the National Heart, Lung, and Blood Institute. Dr. Bangalore disclosed that he is a member of the advisory board and/or a board member for Meril, SMT, Pfizer, Amgen, Biotronik, and Abbott. He also is a consultant for Reata Pharmaceuticals.
SOURCE: Bangalore S et al. SCAI 2020, Abstract 11656.
Patients in the landmark ISCHEMIA trial with intermediate left main disease had a greater extent of coronary artery disease on invasive angiography, indicating greater atherosclerotic burden. They also had worse prognosis with a higher risk of cardiovascular events.
“Many times, we are looking at results as to whether patients have left main disease or not,” Sripal Bangalore, MD, said during the Society for Cardiovascular Angiography & Interventions virtual annual scientific sessions. “Here, we are showing that it’s not black and white; there are shades of gray. If a patient has intermediate left main disease, the prognosis is worse. That’s very important information we need to convey to our referrals also, because many times they may just look at the bottom line and say, ‘there is no left main disease.’ But here, we’re seeing that even having intermediate left main disease has significantly worse prognosis. We need to take that seriously.”
Prior studies show that patients with significant left main disease (LMD; defined as 50% or greater stenosis on coronary CT angiography [CCTA]) have a high risk of cardiovascular events and guidelines recommend revascularization to improve survival, said Dr. Bangalore, an interventional cardiologist at New York University Langone Health. However, the impact of intermediate LMD (defined as 25%-49% stenosis on CCTA) on outcomes is unclear.
Members of the ISCHEMIA (International Study of Comparative Health Effectiveness with Medical and Invasive Approaches) research group randomized 5,179 participants to an initial invasive or conservative strategy. The main results showed that immediate revascularization in patients with stable ischemic heart disease provided no reduction in cardiovascular endpoints through 4 years of follow-up, compared with initial optimal medical therapy alone.
‘Discordance’ revealed in imaging modalities
For the current analysis, named the ISCHEMIA Intermediate LM Substudy, those who underwent coronary CCTA comprise the LMD substudy cohort. The objective was to evaluate clinical and quality of life outcomes in patients with and without intermediate left main disease on coronary CT and to evaluate the impact of treatment strategy on those outcomes across subgroups.
At baseline, these patients were categorized into those with and without intermediate LMD as determined by a core lab. Patients with LMD of 50% or greater, those with prior coronary artery bypass graft surgery, and those with nonevaluable or missing data on LM stenosis were excluded.
Among the 3,913 ISCHEMIA participants who underwent CCTA, 3,699 satisfied the inclusion criteria. Of these patients, 962 (26%) had intermediate LMD and 2,737 (74%) did not.
The researchers observed no significant differences in baseline characteristics between patients with and without LMD. However, patients with intermediate LMD tended to be older, and a greater proportion had hypertension and diabetes. Stress test characteristics were also similar between patients with and without LMD. However, patients with intermediate LMD tended toward a greater severity of severe ischemia.
This was also true for anatomic disease on CCTA. A higher proportion of patients with intermediate LMD had triple-vessel disease (61%-62%, compared with 36%-40% along those without intermediate LMD). In addition, a higher proportion of patients with intermediate LMD had stenosis in the proximal left anterior artery descending (LAD) artery (65% vs. 39% among those without intermediate LMD).
On analysis limited to 1,846 patients who underwent invasive angiography treatment in the main ISCHEMIA trial, 7% of those who were categorized into the intermediate LMD group were found to have LMD disease of 50% or greater, compared with 1.4% of patients who were categorized as not having intermediate LMD. “This goes to show this discordance between the two modalities [CCTA and coronary angiography], and I think we have to be careful,” said Dr. Bangalore, who also directs NYU Langone’s Cardiac Catheterization Laboratory. “There may be patients with left main disease, even if the CCTA says it’s not at 25%-29% [stenosis].”
The researchers found that, among patients who underwent invasive angiography, a greater proportion of those who were categorized into the LMD group had proximal LAD disease (43% vs. 33% among those who were categorized into the nonintermediate LMD group), triple-vessel disease (47% vs. 35%), a greater extent of coronary artery disease as denoted by a higher SYNTAX score (21 vs. 15), and a higher proportion underwent coronary artery bypass graft surgery (32% vs. 18%).
Intermediate LMD linked to worse outcomes
After the researchers adjusted for baseline differences between the two groups in overall substudy cohort, they found that intermediate LMD severity was an independent predictor of the primary composite endpoint of cardiovascular death, MI, hospitalization for unstable angina, heart failure, and resuscitated cardiac arrest (hazard ratio, 1.31; P = .0123); cardiovascular death/MI/stroke (HR, 1.30; P = .0143); procedural primary MI (HR, 1.64; P = .0487); heart failure (HR, 2.06; P = .0239); and stroke (HR, 1.82, P = .0362).
“We then looked to see if there is a treatment difference, a treatment effect based on whether patients had intermediate LMD,” Dr. Bangalore said. “Most of the P values were not significant. The results are very consistent with what we saw in the main analysis: not a significant difference between invasive and conservative strategy. We do see some differences, though. An invasive strategy was associated with a significantly higher risk of procedural MI [2.9% vs. 1.5%], but a significantly lower risk of nonprocedural MI [–6.4% vs. –2%].”
Dr. Bangalore added that there was significant benefit of the invasive strategy in reducing angina and improving quality of life based on the Seattle Angina Questionnaire-7. “This result was durable up to 48 months of follow-up, whether the patient had intermediate left main disease or not. These results were dependent on baseline angina status. The benefit of invasive strategy was mainly in patients who had daily, weekly, and monthly angina, and no benefit in patients with no angina; there was no interaction based on intermediate left main status.”
Dr. Bangalore emphasized that the original ISCHEMIA trial excluded patients with severe left main disease by design. “But patients with intermediate left main disease in ISCHEMIA tended to have a greater extent of coronary artery disease, indicating greater atherosclerotic burden. I don’t think that’s any surprise. They had a worse prognosis with higher risk of cardiovascular events but similar quality of life, including angina-specific quality of life.”
The key clinical message, he said, is that patients with intermediate LMD face an increased risk of cardiovascular events. “I think we have to be aggressive in trying to reduce their risk with medical therapy, etc.,” he said. “If they are symptomatic, ISCHEMIA tells us that patients have two options. They can choose an invasive strategy, because clearly there is a benefit. You have a significant benefit at making you feel better and potentially reducing the risk of spontaneous MI over a period of time. Or, you can try medical therapy first. If you do see some left main disease, it’s showing the general burden of atherosclerosis disease in those patients. I think that’s the critical message, that we have to be very aggressive with these patients.”
A call for more imaging studies
An invited panelist, Timothy D. Henry, MD, said that the results of the ISCHEMIA substudy should stimulate further research. “With an intermediate lesion, clearly the interventional group did better, and it wasn’t symptom related,” said Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research and Education at the Christ Hospital in Cincinnati. “So even if you do medical therapy, you’re not going to really find it out. In my mind, this should stimulate us to do more imaging of the left main that are moderate lesions, and follow this up as an independent study. I think this is a really important finding.”
ISCHEMIA was supported by grants from the National Heart, Lung, and Blood Institute. Dr. Bangalore disclosed that he is a member of the advisory board and/or a board member for Meril, SMT, Pfizer, Amgen, Biotronik, and Abbott. He also is a consultant for Reata Pharmaceuticals.
SOURCE: Bangalore S et al. SCAI 2020, Abstract 11656.
FROM SCAI 2020
FDA recalls extended-release metformin due to NDMA impurities
The US Food and Drug Administration (FDA) has recommended voluntary recall of certain extended-release (ER) versions of metformin because testing has revealed excessive levels of N-nitrosodimethylamine (NDMA) in these products.
Metformin is the most commonly prescribed drug used to treat type 2 diabetes worldwide.
NDMA is a contaminant with the potential to be carcinogenic if there is exposure to above-acceptable levels over the long-term.
Five pharmaceutical firms in particular are being contacted by the FDA with notices (posted on the FDA website) recommending they voluntarily recall their products. At the time of writing, only one was listed, Apotex Corp and its metformin hydrochloride ER tablets, USP 500 mg.
The recall does not apply to immediate-release metformin products, the most commonly prescribed ones for diabetes, the agency stresses.
It also recommends that clinicians continue to prescribe metformin when clinically appropriate.
In late 2019, the FDA announced it had become aware of NDMA in some metformin products in other countries. The agency immediately began testing to determine whether the metformin in the US supply was at risk, as part of the ongoing investigation into nitrosamine impurities across medication types, which included recalls of hypertension and heartburn medications within the past 2 years.
By February 2020, the agency had identified very low levels of NDMA in some samples, but at that time, no FDA-tested sample of metformin exceeded the acceptable intake limit for NDMA, as reported by Medscape Medical News.
“Now that we have identified some metformin products that do not meet our standards, we’re taking action. As we have been doing since this impurity was first identified, we will communicate as new scientific information becomes available and will take further action, if appropriate,” Patrizia Cavazzoni, MD, acting director of the FDA Center for Drug Evaluation and Research, said in a press release.
Requests for recall apply only to affected products
The recall was instigated after the FDA became aware of reports of higher levels of NDMA in certain ER formulations of metformin through a citizen petition filed by a private laboratory. The agency confirmed unacceptable NDMA levels in some, but not all, of those lots.
“In other instances, our laboratory detected NDMA in lots that the private laboratory did not,” it notes.
The FDA says it is working closely with manufacturers of the recalled tablets to identify the source of the NDMA impurity and ensure appropriate testing is carried out.
Elevated levels of NDMA have been found in some finished-dose tablets of the ER formulations but NDMA has not been detected in samples of the metformin active pharmaceutical ingredient.
The FDA also stresses there are many other additional manufacturers that supply metformin ER products to much of the US market, and they are not being asked to recall their products.
Work is also ongoing to determine whether the drug recalls will result in shortages, and if so, the agency says it will collaborate with manufacturers to prevent or reduce any impact of shortages.
“We understand that patients may have concerns about possible impurities in their medicines and want to assure the public that we have been looking closely at this problem over many months in order to provide patients and healthcare professionals with clear and accurate answers,” Cavazzoni said.
For more information about NDMA, visit the FDA nitrosamines web page.
This article first appeared on Medscape.com.
The US Food and Drug Administration (FDA) has recommended voluntary recall of certain extended-release (ER) versions of metformin because testing has revealed excessive levels of N-nitrosodimethylamine (NDMA) in these products.
Metformin is the most commonly prescribed drug used to treat type 2 diabetes worldwide.
NDMA is a contaminant with the potential to be carcinogenic if there is exposure to above-acceptable levels over the long-term.
Five pharmaceutical firms in particular are being contacted by the FDA with notices (posted on the FDA website) recommending they voluntarily recall their products. At the time of writing, only one was listed, Apotex Corp and its metformin hydrochloride ER tablets, USP 500 mg.
The recall does not apply to immediate-release metformin products, the most commonly prescribed ones for diabetes, the agency stresses.
It also recommends that clinicians continue to prescribe metformin when clinically appropriate.
In late 2019, the FDA announced it had become aware of NDMA in some metformin products in other countries. The agency immediately began testing to determine whether the metformin in the US supply was at risk, as part of the ongoing investigation into nitrosamine impurities across medication types, which included recalls of hypertension and heartburn medications within the past 2 years.
By February 2020, the agency had identified very low levels of NDMA in some samples, but at that time, no FDA-tested sample of metformin exceeded the acceptable intake limit for NDMA, as reported by Medscape Medical News.
“Now that we have identified some metformin products that do not meet our standards, we’re taking action. As we have been doing since this impurity was first identified, we will communicate as new scientific information becomes available and will take further action, if appropriate,” Patrizia Cavazzoni, MD, acting director of the FDA Center for Drug Evaluation and Research, said in a press release.
Requests for recall apply only to affected products
The recall was instigated after the FDA became aware of reports of higher levels of NDMA in certain ER formulations of metformin through a citizen petition filed by a private laboratory. The agency confirmed unacceptable NDMA levels in some, but not all, of those lots.
“In other instances, our laboratory detected NDMA in lots that the private laboratory did not,” it notes.
The FDA says it is working closely with manufacturers of the recalled tablets to identify the source of the NDMA impurity and ensure appropriate testing is carried out.
Elevated levels of NDMA have been found in some finished-dose tablets of the ER formulations but NDMA has not been detected in samples of the metformin active pharmaceutical ingredient.
The FDA also stresses there are many other additional manufacturers that supply metformin ER products to much of the US market, and they are not being asked to recall their products.
Work is also ongoing to determine whether the drug recalls will result in shortages, and if so, the agency says it will collaborate with manufacturers to prevent or reduce any impact of shortages.
“We understand that patients may have concerns about possible impurities in their medicines and want to assure the public that we have been looking closely at this problem over many months in order to provide patients and healthcare professionals with clear and accurate answers,” Cavazzoni said.
For more information about NDMA, visit the FDA nitrosamines web page.
This article first appeared on Medscape.com.
The US Food and Drug Administration (FDA) has recommended voluntary recall of certain extended-release (ER) versions of metformin because testing has revealed excessive levels of N-nitrosodimethylamine (NDMA) in these products.
Metformin is the most commonly prescribed drug used to treat type 2 diabetes worldwide.
NDMA is a contaminant with the potential to be carcinogenic if there is exposure to above-acceptable levels over the long-term.
Five pharmaceutical firms in particular are being contacted by the FDA with notices (posted on the FDA website) recommending they voluntarily recall their products. At the time of writing, only one was listed, Apotex Corp and its metformin hydrochloride ER tablets, USP 500 mg.
The recall does not apply to immediate-release metformin products, the most commonly prescribed ones for diabetes, the agency stresses.
It also recommends that clinicians continue to prescribe metformin when clinically appropriate.
In late 2019, the FDA announced it had become aware of NDMA in some metformin products in other countries. The agency immediately began testing to determine whether the metformin in the US supply was at risk, as part of the ongoing investigation into nitrosamine impurities across medication types, which included recalls of hypertension and heartburn medications within the past 2 years.
By February 2020, the agency had identified very low levels of NDMA in some samples, but at that time, no FDA-tested sample of metformin exceeded the acceptable intake limit for NDMA, as reported by Medscape Medical News.
“Now that we have identified some metformin products that do not meet our standards, we’re taking action. As we have been doing since this impurity was first identified, we will communicate as new scientific information becomes available and will take further action, if appropriate,” Patrizia Cavazzoni, MD, acting director of the FDA Center for Drug Evaluation and Research, said in a press release.
Requests for recall apply only to affected products
The recall was instigated after the FDA became aware of reports of higher levels of NDMA in certain ER formulations of metformin through a citizen petition filed by a private laboratory. The agency confirmed unacceptable NDMA levels in some, but not all, of those lots.
“In other instances, our laboratory detected NDMA in lots that the private laboratory did not,” it notes.
The FDA says it is working closely with manufacturers of the recalled tablets to identify the source of the NDMA impurity and ensure appropriate testing is carried out.
Elevated levels of NDMA have been found in some finished-dose tablets of the ER formulations but NDMA has not been detected in samples of the metformin active pharmaceutical ingredient.
The FDA also stresses there are many other additional manufacturers that supply metformin ER products to much of the US market, and they are not being asked to recall their products.
Work is also ongoing to determine whether the drug recalls will result in shortages, and if so, the agency says it will collaborate with manufacturers to prevent or reduce any impact of shortages.
“We understand that patients may have concerns about possible impurities in their medicines and want to assure the public that we have been looking closely at this problem over many months in order to provide patients and healthcare professionals with clear and accurate answers,” Cavazzoni said.
For more information about NDMA, visit the FDA nitrosamines web page.
This article first appeared on Medscape.com.
Patients find CAC more persuasive than ASCVD risk score for statin decisions
Patients who received a protocol-driven recommendation to initiate statin therapy for primary prevention of cardiovascular disease based upon their CT angiography coronary artery calcium score were twice as likely to actually start on the drug than those whose recommendation was guided by the American College of Cardiology/American Heart Association Pooled Cohort Equations Risk Calculator, according to the results of the randomized CorCal Vanguard study.
These results suggest that patients – and their primary care physicians – find the conventional method of screening for cardiovascular risk using the Pooled Cohort Equations to estimate the 10-year risk of MI or stroke, as recommended in ACC/AHA guidelines, to be less persuasive than screening for the presence or absence of actual disease as captured by CT angiography images and the associated coronary artery calcium (CAC) score, Joseph B. Muhlestein, MD, said at the joint scientific sessions of the ACC and the World Heart Federation. The meeting was conducted online after its cancellation because of the COVID-19 pandemic.
The CorCal Vanguard study included 601 patients with an average baseline LDL cholesterol of 120 mg/dL, an average age of 60 years, and no history of cardiovascular disease, diabetes, or prior statin therapy. They were randomized to decision-making regarding statin therapy based on either the ACC/AHA guideline–endorsed Pooled Cohort Equations, which use an estimated 10-year risk of 7.5% or more as the threshold for statin initiation, or their CAC score.
If a patient’s CAC score was 0, the recommendation was against starting a statin. Everyone with a CAC greater than 100 received a recommendation for high-intensity statin therapy. And for those with a CAC of 1-100, the decision defaulted to the results of the Pooled Cohort Equations. The screening results were provided to a patient’s primary physician so they could engage in joint decision-making regarding initiation of statin therapy. Adherence to a screening-based recommendation to start on a statin was assessed at 3 and 12 months of follow-up, explained Dr. Muhlestein, a cardiologist at the Intermountain Medical Center Heart Institute in Salt Lake City.
He noted that CorCal Vanguard was merely a feasibility study. Based on the study results he presented at ACC 2020, the full 9,000-patient CorCal primary prevention trial is now enrolling participants. CorCal is the first randomized trial to pit the Pooled Cohort Equations against the CAC score in a large study looking for differences in downstream clinical outcomes.
The rationale for this line of clinical research lies in the known limitations of the ACC/AHA risk calculator. “It may overestimate risk in some populations, patients aren’t always adherent to Pooled Cohort Equations Risk Calculator recommendations, and it doesn’t include novel risk markers such as C-reactive protein that some consider important for risk assessment. And the big question: Should we continue risk screening to determine potential benefit from drug therapy, or should we switch to disease screening?” the cardiologist commented.
The CorCal Vanguard results
A recommendation to start statin therapy was made in 48% of patients in the Pooled Cohort Equations group, versus 36% of the group randomized to CAC. However, only 17% of patients in the Pooled Cohort Equations group actually initiated a statin, a significantly lower rate than the 26% figure in the CAC arm. Fully 70% of patients who received a recommendation to start taking a statin on the basis of their CAC score actually did so, compared to just 36% of those whose recommendation was based upon their Pooled Cohort Equations Risk Calculator.
At 3 months of follow-up, 61% of patients who received an initial recommendation to start statin therapy based upon their CAC screening were actually taking a statin, compared with 41% of those whose recommendation was based upon the Pooled Cohort Equations. At 12 months, the figures were 64% and 49%.
In both groups, at 12 months of follow-up, the No. 1 reason patients weren’t taking a statin as recommended was that their personal physician had advised against it or never prescribed it. That accounted for roughly half of the nonadherence. Another quarter was because of a preference to try lifestyle change first. Fear of drug side effects was a less common reason.
Putting the CorCal Vanguard study results in perspective, Dr. Muhlestein observed that, prior to the screening study, none of the participants had ever been on a statin, yet 37% of them were found by one screening method or the other to be at high cardiovascular risk. Of those high-risk patients, 51% actually initiated statin therapy and the majority of them were still taking their medication 12 months later.
“That has to be a good thing. It emphasizes what can be done when proactive primary prevention is practiced,” the cardiologist said.
He reported having no financial conflicts regarding the CorCal study, which was funded by a grant from the Dell Loy Hansen Cardiovascular Research Fund.
SOURCE: Muhlestein JB et al. ACC 2020, Abstract 909-12.
Patients who received a protocol-driven recommendation to initiate statin therapy for primary prevention of cardiovascular disease based upon their CT angiography coronary artery calcium score were twice as likely to actually start on the drug than those whose recommendation was guided by the American College of Cardiology/American Heart Association Pooled Cohort Equations Risk Calculator, according to the results of the randomized CorCal Vanguard study.
These results suggest that patients – and their primary care physicians – find the conventional method of screening for cardiovascular risk using the Pooled Cohort Equations to estimate the 10-year risk of MI or stroke, as recommended in ACC/AHA guidelines, to be less persuasive than screening for the presence or absence of actual disease as captured by CT angiography images and the associated coronary artery calcium (CAC) score, Joseph B. Muhlestein, MD, said at the joint scientific sessions of the ACC and the World Heart Federation. The meeting was conducted online after its cancellation because of the COVID-19 pandemic.
The CorCal Vanguard study included 601 patients with an average baseline LDL cholesterol of 120 mg/dL, an average age of 60 years, and no history of cardiovascular disease, diabetes, or prior statin therapy. They were randomized to decision-making regarding statin therapy based on either the ACC/AHA guideline–endorsed Pooled Cohort Equations, which use an estimated 10-year risk of 7.5% or more as the threshold for statin initiation, or their CAC score.
If a patient’s CAC score was 0, the recommendation was against starting a statin. Everyone with a CAC greater than 100 received a recommendation for high-intensity statin therapy. And for those with a CAC of 1-100, the decision defaulted to the results of the Pooled Cohort Equations. The screening results were provided to a patient’s primary physician so they could engage in joint decision-making regarding initiation of statin therapy. Adherence to a screening-based recommendation to start on a statin was assessed at 3 and 12 months of follow-up, explained Dr. Muhlestein, a cardiologist at the Intermountain Medical Center Heart Institute in Salt Lake City.
He noted that CorCal Vanguard was merely a feasibility study. Based on the study results he presented at ACC 2020, the full 9,000-patient CorCal primary prevention trial is now enrolling participants. CorCal is the first randomized trial to pit the Pooled Cohort Equations against the CAC score in a large study looking for differences in downstream clinical outcomes.
The rationale for this line of clinical research lies in the known limitations of the ACC/AHA risk calculator. “It may overestimate risk in some populations, patients aren’t always adherent to Pooled Cohort Equations Risk Calculator recommendations, and it doesn’t include novel risk markers such as C-reactive protein that some consider important for risk assessment. And the big question: Should we continue risk screening to determine potential benefit from drug therapy, or should we switch to disease screening?” the cardiologist commented.
The CorCal Vanguard results
A recommendation to start statin therapy was made in 48% of patients in the Pooled Cohort Equations group, versus 36% of the group randomized to CAC. However, only 17% of patients in the Pooled Cohort Equations group actually initiated a statin, a significantly lower rate than the 26% figure in the CAC arm. Fully 70% of patients who received a recommendation to start taking a statin on the basis of their CAC score actually did so, compared to just 36% of those whose recommendation was based upon their Pooled Cohort Equations Risk Calculator.
At 3 months of follow-up, 61% of patients who received an initial recommendation to start statin therapy based upon their CAC screening were actually taking a statin, compared with 41% of those whose recommendation was based upon the Pooled Cohort Equations. At 12 months, the figures were 64% and 49%.
In both groups, at 12 months of follow-up, the No. 1 reason patients weren’t taking a statin as recommended was that their personal physician had advised against it or never prescribed it. That accounted for roughly half of the nonadherence. Another quarter was because of a preference to try lifestyle change first. Fear of drug side effects was a less common reason.
Putting the CorCal Vanguard study results in perspective, Dr. Muhlestein observed that, prior to the screening study, none of the participants had ever been on a statin, yet 37% of them were found by one screening method or the other to be at high cardiovascular risk. Of those high-risk patients, 51% actually initiated statin therapy and the majority of them were still taking their medication 12 months later.
“That has to be a good thing. It emphasizes what can be done when proactive primary prevention is practiced,” the cardiologist said.
He reported having no financial conflicts regarding the CorCal study, which was funded by a grant from the Dell Loy Hansen Cardiovascular Research Fund.
SOURCE: Muhlestein JB et al. ACC 2020, Abstract 909-12.
Patients who received a protocol-driven recommendation to initiate statin therapy for primary prevention of cardiovascular disease based upon their CT angiography coronary artery calcium score were twice as likely to actually start on the drug than those whose recommendation was guided by the American College of Cardiology/American Heart Association Pooled Cohort Equations Risk Calculator, according to the results of the randomized CorCal Vanguard study.
These results suggest that patients – and their primary care physicians – find the conventional method of screening for cardiovascular risk using the Pooled Cohort Equations to estimate the 10-year risk of MI or stroke, as recommended in ACC/AHA guidelines, to be less persuasive than screening for the presence or absence of actual disease as captured by CT angiography images and the associated coronary artery calcium (CAC) score, Joseph B. Muhlestein, MD, said at the joint scientific sessions of the ACC and the World Heart Federation. The meeting was conducted online after its cancellation because of the COVID-19 pandemic.
The CorCal Vanguard study included 601 patients with an average baseline LDL cholesterol of 120 mg/dL, an average age of 60 years, and no history of cardiovascular disease, diabetes, or prior statin therapy. They were randomized to decision-making regarding statin therapy based on either the ACC/AHA guideline–endorsed Pooled Cohort Equations, which use an estimated 10-year risk of 7.5% or more as the threshold for statin initiation, or their CAC score.
If a patient’s CAC score was 0, the recommendation was against starting a statin. Everyone with a CAC greater than 100 received a recommendation for high-intensity statin therapy. And for those with a CAC of 1-100, the decision defaulted to the results of the Pooled Cohort Equations. The screening results were provided to a patient’s primary physician so they could engage in joint decision-making regarding initiation of statin therapy. Adherence to a screening-based recommendation to start on a statin was assessed at 3 and 12 months of follow-up, explained Dr. Muhlestein, a cardiologist at the Intermountain Medical Center Heart Institute in Salt Lake City.
He noted that CorCal Vanguard was merely a feasibility study. Based on the study results he presented at ACC 2020, the full 9,000-patient CorCal primary prevention trial is now enrolling participants. CorCal is the first randomized trial to pit the Pooled Cohort Equations against the CAC score in a large study looking for differences in downstream clinical outcomes.
The rationale for this line of clinical research lies in the known limitations of the ACC/AHA risk calculator. “It may overestimate risk in some populations, patients aren’t always adherent to Pooled Cohort Equations Risk Calculator recommendations, and it doesn’t include novel risk markers such as C-reactive protein that some consider important for risk assessment. And the big question: Should we continue risk screening to determine potential benefit from drug therapy, or should we switch to disease screening?” the cardiologist commented.
The CorCal Vanguard results
A recommendation to start statin therapy was made in 48% of patients in the Pooled Cohort Equations group, versus 36% of the group randomized to CAC. However, only 17% of patients in the Pooled Cohort Equations group actually initiated a statin, a significantly lower rate than the 26% figure in the CAC arm. Fully 70% of patients who received a recommendation to start taking a statin on the basis of their CAC score actually did so, compared to just 36% of those whose recommendation was based upon their Pooled Cohort Equations Risk Calculator.
At 3 months of follow-up, 61% of patients who received an initial recommendation to start statin therapy based upon their CAC screening were actually taking a statin, compared with 41% of those whose recommendation was based upon the Pooled Cohort Equations. At 12 months, the figures were 64% and 49%.
In both groups, at 12 months of follow-up, the No. 1 reason patients weren’t taking a statin as recommended was that their personal physician had advised against it or never prescribed it. That accounted for roughly half of the nonadherence. Another quarter was because of a preference to try lifestyle change first. Fear of drug side effects was a less common reason.
Putting the CorCal Vanguard study results in perspective, Dr. Muhlestein observed that, prior to the screening study, none of the participants had ever been on a statin, yet 37% of them were found by one screening method or the other to be at high cardiovascular risk. Of those high-risk patients, 51% actually initiated statin therapy and the majority of them were still taking their medication 12 months later.
“That has to be a good thing. It emphasizes what can be done when proactive primary prevention is practiced,” the cardiologist said.
He reported having no financial conflicts regarding the CorCal study, which was funded by a grant from the Dell Loy Hansen Cardiovascular Research Fund.
SOURCE: Muhlestein JB et al. ACC 2020, Abstract 909-12.
FROM ACC 2020
More from REDUCE-IT: Icosapent ethyl cuts revascularization by a third
A new analysis from the REDUCE-IT trial has shown that the high-strength eicosapentaenoic acid product icosapent ethyl (Vascepa; Amarin) reduced the number of revascularization procedures by more than one-third in statin-treated patients whose triglyceride levels were elevated and who were at increased cardiovascular risk.
The new data were presented at the Society for Cardiovascular Angiography & Interventions virtual annual scientific sessions.
REDUCE-IT, a multicenter, double-blind, placebo-controlled trial, randomly assigned statin-treated patients whose triglyceride levels were elevated (135-499 mg/dL), whose LDL cholesterol levels were controlled (41-100 mg/dL), and who had established cardiovascular disease or diabetes plus risk factors to receive either icosapent ethyl 4 g daily or placebo.
The primary composite and other cardiovascular endpoints were substantially reduced. Prespecified analyses examined all coronary revascularizations, recurrent revascularizations, and revascularization subtypes.
“Compared with placebo, icosapent ethyl 4 g/day significantly reduced first and total revascularization events by 34% and 36%, respectively,” REDUCE-IT investigator Benjamin Peterson, MD, of Brigham and Women’s Hospital, Boston, concluded during his presentation.
This reduction was consistent with respect to urgent, emergent, and elective revascularization procedures overall, as well as percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG) individually, he reported.
“Prior therapies aimed at patients with elevated triglycerides have not demonstrated a consistent benefit in reducing coronary revascularization, and to the best of our knowledge, this is the first non–LDL cholesterol intervention in a major randomized trial in which statin-treated patients underwent fewer CABG surgeries,” Dr. Peterson stated.
“These data highlight the substantial impact of icosapent ethyl on the underlying atherothrombotic burden in the at-risk REDUCE-IT population,” he added.
Detailed results showed that the percentage of patients who underwent first revascularizations was 9.2% with icosapent ethyl versus 13.3% with placebo (hazard ratio, 0.66; P < .0001; number needed to treat, 25).
Similar reductions were observed in total (first and subsequent) revascularizations (risk ratio, 0.64; P < .0001) and across urgent, emergent, and elective revascularizations. Icosapent ethyl significantly reduced the need for PCI (HR, 0.68; P < .0001) and CABG (HR, 0.61; P = .0005).
The moderator of a SCAI press conference, Kirk Garratt, MD, of the Center for Heart and Vascular Health at Christiana Care Health System in Wilmington, Del., said that “this is an impressive impact. I couldn’t count the number of zeros in the P value.”
Timothy Henry, MD, of Christ Hospital in Cincinnati, said that REDUCE-IT was an important trial. “It showed a very impressive effect on revascularizations along with all the other benefits.” But he suggested that the uptake in usage of icosapent ethyl in the United States has been slow, and he asked what could be done to enhance this.
REDUCE-IT senior investigator Deepak Bhatt, MD, replied that the product was only approved for the REDUCE-IT indication in December 2019, and he suggested that initial uptake may have been affected by the current COVID-19 pandemic.
“This new REDUCE-IT indication ― patients with established cardiovascular disease or diabetes plus risk factors who have moderately elevated triglycerides and controlled LDL ― includes a lot of patients, between 15% and 50% of all cardiovascular patients,” he said.
“We wanted to present this revascularization data at the SCAI meeting, as it is superimportant that interventional cardiologists know about this. Interventionalists have now taken ownership of LDL and make sure patients are on statins, and we hope they will now do the same thing for triglycerides,” Dr. Bhatt commented.
Dr. Henry agreed. “It should be a simple thing to take all secondary prevention patients with eligible triglycerides and be aggressive with this new therapy.”
Dr. Bhatt noted that a cost-effectiveness analysis of the REDUCE-IT trial that was presented at last year’s American Heart Association meeting “has shown the drug to be highly cost effective and actually cost saving at the current list price.”
Asked what the mechanism of benefit is, Dr. Bhatt said that “there has been good basic science showing that EPA [eicosapentaenoic acid] stabilizes cell membranes and reduces plaque vulnerability and progression.”
REDUCE-IT was sponsored by Amarin. Brigham and Women’s Hospital receives research funding from Amarin for Dr. Bhatt’s role as chair of the trial.
A version of this article originally appeared on Medscape.com.
A new analysis from the REDUCE-IT trial has shown that the high-strength eicosapentaenoic acid product icosapent ethyl (Vascepa; Amarin) reduced the number of revascularization procedures by more than one-third in statin-treated patients whose triglyceride levels were elevated and who were at increased cardiovascular risk.
The new data were presented at the Society for Cardiovascular Angiography & Interventions virtual annual scientific sessions.
REDUCE-IT, a multicenter, double-blind, placebo-controlled trial, randomly assigned statin-treated patients whose triglyceride levels were elevated (135-499 mg/dL), whose LDL cholesterol levels were controlled (41-100 mg/dL), and who had established cardiovascular disease or diabetes plus risk factors to receive either icosapent ethyl 4 g daily or placebo.
The primary composite and other cardiovascular endpoints were substantially reduced. Prespecified analyses examined all coronary revascularizations, recurrent revascularizations, and revascularization subtypes.
“Compared with placebo, icosapent ethyl 4 g/day significantly reduced first and total revascularization events by 34% and 36%, respectively,” REDUCE-IT investigator Benjamin Peterson, MD, of Brigham and Women’s Hospital, Boston, concluded during his presentation.
This reduction was consistent with respect to urgent, emergent, and elective revascularization procedures overall, as well as percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG) individually, he reported.
“Prior therapies aimed at patients with elevated triglycerides have not demonstrated a consistent benefit in reducing coronary revascularization, and to the best of our knowledge, this is the first non–LDL cholesterol intervention in a major randomized trial in which statin-treated patients underwent fewer CABG surgeries,” Dr. Peterson stated.
“These data highlight the substantial impact of icosapent ethyl on the underlying atherothrombotic burden in the at-risk REDUCE-IT population,” he added.
Detailed results showed that the percentage of patients who underwent first revascularizations was 9.2% with icosapent ethyl versus 13.3% with placebo (hazard ratio, 0.66; P < .0001; number needed to treat, 25).
Similar reductions were observed in total (first and subsequent) revascularizations (risk ratio, 0.64; P < .0001) and across urgent, emergent, and elective revascularizations. Icosapent ethyl significantly reduced the need for PCI (HR, 0.68; P < .0001) and CABG (HR, 0.61; P = .0005).
The moderator of a SCAI press conference, Kirk Garratt, MD, of the Center for Heart and Vascular Health at Christiana Care Health System in Wilmington, Del., said that “this is an impressive impact. I couldn’t count the number of zeros in the P value.”
Timothy Henry, MD, of Christ Hospital in Cincinnati, said that REDUCE-IT was an important trial. “It showed a very impressive effect on revascularizations along with all the other benefits.” But he suggested that the uptake in usage of icosapent ethyl in the United States has been slow, and he asked what could be done to enhance this.
REDUCE-IT senior investigator Deepak Bhatt, MD, replied that the product was only approved for the REDUCE-IT indication in December 2019, and he suggested that initial uptake may have been affected by the current COVID-19 pandemic.
“This new REDUCE-IT indication ― patients with established cardiovascular disease or diabetes plus risk factors who have moderately elevated triglycerides and controlled LDL ― includes a lot of patients, between 15% and 50% of all cardiovascular patients,” he said.
“We wanted to present this revascularization data at the SCAI meeting, as it is superimportant that interventional cardiologists know about this. Interventionalists have now taken ownership of LDL and make sure patients are on statins, and we hope they will now do the same thing for triglycerides,” Dr. Bhatt commented.
Dr. Henry agreed. “It should be a simple thing to take all secondary prevention patients with eligible triglycerides and be aggressive with this new therapy.”
Dr. Bhatt noted that a cost-effectiveness analysis of the REDUCE-IT trial that was presented at last year’s American Heart Association meeting “has shown the drug to be highly cost effective and actually cost saving at the current list price.”
Asked what the mechanism of benefit is, Dr. Bhatt said that “there has been good basic science showing that EPA [eicosapentaenoic acid] stabilizes cell membranes and reduces plaque vulnerability and progression.”
REDUCE-IT was sponsored by Amarin. Brigham and Women’s Hospital receives research funding from Amarin for Dr. Bhatt’s role as chair of the trial.
A version of this article originally appeared on Medscape.com.
A new analysis from the REDUCE-IT trial has shown that the high-strength eicosapentaenoic acid product icosapent ethyl (Vascepa; Amarin) reduced the number of revascularization procedures by more than one-third in statin-treated patients whose triglyceride levels were elevated and who were at increased cardiovascular risk.
The new data were presented at the Society for Cardiovascular Angiography & Interventions virtual annual scientific sessions.
REDUCE-IT, a multicenter, double-blind, placebo-controlled trial, randomly assigned statin-treated patients whose triglyceride levels were elevated (135-499 mg/dL), whose LDL cholesterol levels were controlled (41-100 mg/dL), and who had established cardiovascular disease or diabetes plus risk factors to receive either icosapent ethyl 4 g daily or placebo.
The primary composite and other cardiovascular endpoints were substantially reduced. Prespecified analyses examined all coronary revascularizations, recurrent revascularizations, and revascularization subtypes.
“Compared with placebo, icosapent ethyl 4 g/day significantly reduced first and total revascularization events by 34% and 36%, respectively,” REDUCE-IT investigator Benjamin Peterson, MD, of Brigham and Women’s Hospital, Boston, concluded during his presentation.
This reduction was consistent with respect to urgent, emergent, and elective revascularization procedures overall, as well as percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG) individually, he reported.
“Prior therapies aimed at patients with elevated triglycerides have not demonstrated a consistent benefit in reducing coronary revascularization, and to the best of our knowledge, this is the first non–LDL cholesterol intervention in a major randomized trial in which statin-treated patients underwent fewer CABG surgeries,” Dr. Peterson stated.
“These data highlight the substantial impact of icosapent ethyl on the underlying atherothrombotic burden in the at-risk REDUCE-IT population,” he added.
Detailed results showed that the percentage of patients who underwent first revascularizations was 9.2% with icosapent ethyl versus 13.3% with placebo (hazard ratio, 0.66; P < .0001; number needed to treat, 25).
Similar reductions were observed in total (first and subsequent) revascularizations (risk ratio, 0.64; P < .0001) and across urgent, emergent, and elective revascularizations. Icosapent ethyl significantly reduced the need for PCI (HR, 0.68; P < .0001) and CABG (HR, 0.61; P = .0005).
The moderator of a SCAI press conference, Kirk Garratt, MD, of the Center for Heart and Vascular Health at Christiana Care Health System in Wilmington, Del., said that “this is an impressive impact. I couldn’t count the number of zeros in the P value.”
Timothy Henry, MD, of Christ Hospital in Cincinnati, said that REDUCE-IT was an important trial. “It showed a very impressive effect on revascularizations along with all the other benefits.” But he suggested that the uptake in usage of icosapent ethyl in the United States has been slow, and he asked what could be done to enhance this.
REDUCE-IT senior investigator Deepak Bhatt, MD, replied that the product was only approved for the REDUCE-IT indication in December 2019, and he suggested that initial uptake may have been affected by the current COVID-19 pandemic.
“This new REDUCE-IT indication ― patients with established cardiovascular disease or diabetes plus risk factors who have moderately elevated triglycerides and controlled LDL ― includes a lot of patients, between 15% and 50% of all cardiovascular patients,” he said.
“We wanted to present this revascularization data at the SCAI meeting, as it is superimportant that interventional cardiologists know about this. Interventionalists have now taken ownership of LDL and make sure patients are on statins, and we hope they will now do the same thing for triglycerides,” Dr. Bhatt commented.
Dr. Henry agreed. “It should be a simple thing to take all secondary prevention patients with eligible triglycerides and be aggressive with this new therapy.”
Dr. Bhatt noted that a cost-effectiveness analysis of the REDUCE-IT trial that was presented at last year’s American Heart Association meeting “has shown the drug to be highly cost effective and actually cost saving at the current list price.”
Asked what the mechanism of benefit is, Dr. Bhatt said that “there has been good basic science showing that EPA [eicosapentaenoic acid] stabilizes cell membranes and reduces plaque vulnerability and progression.”
REDUCE-IT was sponsored by Amarin. Brigham and Women’s Hospital receives research funding from Amarin for Dr. Bhatt’s role as chair of the trial.
A version of this article originally appeared on Medscape.com.
‘The story unfolding is worrisome’ for diabetes and COVID-19
The American Diabetes Association has dedicated a whole section of its journal, Diabetes Care, to the topic of “Diabetes and COVID-19,” publishing a range of articles with new data to help guide physicians in caring for patients.
“Certain groups are more vulnerable to COVID-19, notably older people and those with underlying medical conditions. Because diabetes is one of the conditions associated with high risk, the diabetes community urgently needs to know more about COVID-19 and its effects on people with diabetes,” an introductory commentary noted.
Entitled “COVID-19 in people with diabetes: Urgently needed lessons from early reports,” the commentary is penned by the journal’s editor-in-chief, Matthew Riddle, MD, of Oregon Health & Science University, Portland, and colleagues.
Also writing in the same issue, William T. Cefalu, MD, and colleagues from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) noted it is known that the SARS-CoV-2 virus enters cells via the angiotensin-converting enzyme 2 (ACE-2) receptor. The ACE-2 receptor is known to be in the lungs and upper respiratory tract, “but we also know that it is expressed in other tissues such as heart, small and large intestines, and pancreas,” they wrote, and also “in the kidney.”
Hence, there are emerging reports of acute kidney injury resulting from COVID-19, as well as the impact on many other endocrine/metabolic and gastrointestinal outcomes.
“Pilot clinical studies (observational and interventional) are needed that will support the understanding or treatment of COVID-19–related diseases within the mission of the NIDDK,” they stated.
Although rapidly collected, data “offer important clues”
Some of the new ground covered in the journal articles includes an analysis of COVID-19 outcomes by type of glucose-lowering medication; remote glucose monitoring in hospitalized patients with COVID-19; a suggested approach to cardiovascular risk management in the COVID-19 era, as already reported by Medscape Medical News; and the diagnosis and management of gestational diabetes during the pandemic.
Other articles provide new data for previously reported phenomena, including obesity as a risk factor for worse COVID-19 outcomes and the role of inpatient glycemic control on COVID-19 outcomes.
“The data reported in these articles were rapidly collected and analyzed, in most cases under urgent and stressful conditions,” Dr. Riddle and colleagues cautioned. “Thus, some of the analyses are understandably limited due to missing data, incomplete follow-up, and inability to identify infected but asymptomatic patients.”
Even so, they wrote, some points are clear. “The consistency of findings in these rapidly published reports is reassuring in terms of scientific validity, but the story unfolding is worrisome.”
Specifically, while diabetes does not appear to increase the likelihood of SARS-CoV-2 infection, progression to severe illness is more likely in people with diabetes and COVID-19: They are two to three times as likely to require intensive care, and to die, compared with those infected but without diabetes.
“Neither the mechanisms underlying the increased risk nor the best interventions to limit it have yet been defined, but the studies in this collection of articles offer important clues,” Dr. Riddle and colleagues wrote.
Existing insulin use linked to COVID-19 death risk
One of the articles is a retrospective study of 904 hospitalized COVID-19 patients by Yuchen Chen, MD, of the Huazhong University of Science and Technology, Wuhan, China, and colleagues.
Among the 136 patients with diabetes, risk factors for mortality included older age (adjusted odds ratio, 1.09 per year increase; P = .001) elevated C-reactive protein (aOR, 1.12; P = .043), and insulin use (aOR, 3.58; P = .009).
“Attention needs to be paid to patients with diabetes and COVID-19 who use insulin,” the Chinese authors wrote. “Whether this was due to effects of insulin itself or to characteristics of the patients for whom it was prescribed is not clear,” Dr. Riddle and colleagues noted.
Dr. Chen and colleagues also found no difference in clinical outcomes between those diabetes patients with COVID-19 who were taking an ACE inhibitor or angiotensin II type I receptor blocker, compared with those who did not, which supports existing recommendations to continue use of this type of medication.
Remote glucose monitoring a novel tool for COVID-19 isolation
Another publication, by Gilat Shehav-Zaltzman of Sheba Medical Center, Tel Hashomer, Israel, and colleagues, describes the use of remote continuous glucose monitoring (CGM) in two hospitalized COVID-19 patients who were in isolation – one with type 1 diabetes and the other with type 2 diabetes – treated with basal-bolus insulin.
Using Medtronic CGM systems, the hospital staff was able to view patients’ real-time data uploaded to the Web from computer terminals in virus-free areas outside the patients’ rooms. The hospital’s endocrinology team had trained the intensive care staff on how to replace the sensors weekly and calibrate them twice daily.
“Converting a personal CGM system originally designed for diabetes self-management to team-based, real-time remote glucose monitoring offers a novel tool for inpatient diabetes control in COVID-19 isolation facilities,” the authors wrote.
“Such a solution in addition to ongoing remotely monitored clinical parameters (such as pulse rate, electrocardiogram, and oxygen saturation) adds to quality of diabetes care while minimizing risk of staff exposure and burden,” they observed.
Dr. Riddle and colleagues concurred: “Newer methods of remotely monitoring glucose patterns could be uniquely helpful.”
Key question: Does glycemic management make a difference?
With regard to the important issue of in-hospital control of glucose, Celestino Sardu, MD, PhD, of the University of Campania Luigi Vanvitelli, Naples, Italy, and colleagues reported on 59 patients hospitalized with confirmed COVID-19 and moderately severe pneumonia.
They were categorized as normoglycemic (n = 34) or hyperglycemic (n = 25), as well as with or without diabetes, on the basis of a diagnosis preceding the current illness. Of the 25 patients with hyperglycemia, 15 patients were treated with insulin infusion and 10 patients were not.
In a risk-adjusted analysis, both patients with hyperglycemia and patients with diabetes had a higher risk of severe disease than did those without diabetes and with normoglycemia. Patients with hyperglycemia treated with insulin infusion had a lower risk of severe disease than did patients who didn’t receive an insulin infusion.
And although they noted limitations, the authors wrote, “Our data evidenced that optimal glucose control in the immediate postadmission period for almost 18 days was associated with a significant reduction of inflammatory cytokines and procoagulative status.”
Dr. Riddle and colleagues wrote that the findings of this unrandomized comparison were interpreted “as suggesting that insulin infusion may improve outcomes.”
“If the benefits of seeking excellent glycemic control by this means are confirmed, close monitoring of glucose levels will be essential.”
More on obesity and COVID-19, this time from China
Because it has become increasingly clear that obesity is a risk factor for severe COVID-19, new data from China – where this was less apparent initially – support observations in Europe and the United States.
An article by Qingxian Cai, PhD, of Southern University of Science and Technology, Shenzhen, Guangdong, China, and colleagues looks at this. They found that, among 383 hospitalized patients with COVID-19, the 41 patients with obesity (defined as a body mass index ≥ 28 kg/m2) were significantly more likely to progress to severe disease compared with the 203 patients classified as having normal weight (BMI, 18.5-23.9), with an odds ratio of 3.4.
A similar finding comes from Feng Gao, MD, PhD, of the First Affiliated Hospital of Wenzhou (China) Medical University and colleagues, who studied 75 patients hospitalized with confirmed COVID-19 and obesity (defined as a BMI > 25 in this Asian population) to 75 patients without obesity matched by age and sex. After adjustment for clinical characteristics including the presence of diabetes, those with obesity had a threefold greater risk of progression to severe or critical COVID-19 status, with a nearly linear relationship.
Emerging from the crisis: Protect the vulnerable, increase knowledge base
As the research community emerges from the crisis, “there should be renewed efforts for multidisciplinary research ... aimed at greatly increasing the knowledge base to understand how ... the current COVID-19 threat” affects “both healthy people and people with chronic diseases and conditions,” Dr. Cefalu and colleagues concluded in their commentary.
Dr. Riddle and coauthors agreed: “We will enter a longer interval in which we must continue to support the most vulnerable populations – especially older people, those with diabetes or obesity, and those who lack the resources to limit day-to-day exposure to infection. We hope a growing sense of community will help in this task.”
Dr. Riddle has reported receiving research grant support through Oregon Health & Science University from AstraZeneca, Eli Lilly, and Novo Nordisk, and honoraria for consulting from Adocia, AstraZeneca, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Sanofi, and Theracos. Dr. Cefalu has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The American Diabetes Association has dedicated a whole section of its journal, Diabetes Care, to the topic of “Diabetes and COVID-19,” publishing a range of articles with new data to help guide physicians in caring for patients.
“Certain groups are more vulnerable to COVID-19, notably older people and those with underlying medical conditions. Because diabetes is one of the conditions associated with high risk, the diabetes community urgently needs to know more about COVID-19 and its effects on people with diabetes,” an introductory commentary noted.
Entitled “COVID-19 in people with diabetes: Urgently needed lessons from early reports,” the commentary is penned by the journal’s editor-in-chief, Matthew Riddle, MD, of Oregon Health & Science University, Portland, and colleagues.
Also writing in the same issue, William T. Cefalu, MD, and colleagues from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) noted it is known that the SARS-CoV-2 virus enters cells via the angiotensin-converting enzyme 2 (ACE-2) receptor. The ACE-2 receptor is known to be in the lungs and upper respiratory tract, “but we also know that it is expressed in other tissues such as heart, small and large intestines, and pancreas,” they wrote, and also “in the kidney.”
Hence, there are emerging reports of acute kidney injury resulting from COVID-19, as well as the impact on many other endocrine/metabolic and gastrointestinal outcomes.
“Pilot clinical studies (observational and interventional) are needed that will support the understanding or treatment of COVID-19–related diseases within the mission of the NIDDK,” they stated.
Although rapidly collected, data “offer important clues”
Some of the new ground covered in the journal articles includes an analysis of COVID-19 outcomes by type of glucose-lowering medication; remote glucose monitoring in hospitalized patients with COVID-19; a suggested approach to cardiovascular risk management in the COVID-19 era, as already reported by Medscape Medical News; and the diagnosis and management of gestational diabetes during the pandemic.
Other articles provide new data for previously reported phenomena, including obesity as a risk factor for worse COVID-19 outcomes and the role of inpatient glycemic control on COVID-19 outcomes.
“The data reported in these articles were rapidly collected and analyzed, in most cases under urgent and stressful conditions,” Dr. Riddle and colleagues cautioned. “Thus, some of the analyses are understandably limited due to missing data, incomplete follow-up, and inability to identify infected but asymptomatic patients.”
Even so, they wrote, some points are clear. “The consistency of findings in these rapidly published reports is reassuring in terms of scientific validity, but the story unfolding is worrisome.”
Specifically, while diabetes does not appear to increase the likelihood of SARS-CoV-2 infection, progression to severe illness is more likely in people with diabetes and COVID-19: They are two to three times as likely to require intensive care, and to die, compared with those infected but without diabetes.
“Neither the mechanisms underlying the increased risk nor the best interventions to limit it have yet been defined, but the studies in this collection of articles offer important clues,” Dr. Riddle and colleagues wrote.
Existing insulin use linked to COVID-19 death risk
One of the articles is a retrospective study of 904 hospitalized COVID-19 patients by Yuchen Chen, MD, of the Huazhong University of Science and Technology, Wuhan, China, and colleagues.
Among the 136 patients with diabetes, risk factors for mortality included older age (adjusted odds ratio, 1.09 per year increase; P = .001) elevated C-reactive protein (aOR, 1.12; P = .043), and insulin use (aOR, 3.58; P = .009).
“Attention needs to be paid to patients with diabetes and COVID-19 who use insulin,” the Chinese authors wrote. “Whether this was due to effects of insulin itself or to characteristics of the patients for whom it was prescribed is not clear,” Dr. Riddle and colleagues noted.
Dr. Chen and colleagues also found no difference in clinical outcomes between those diabetes patients with COVID-19 who were taking an ACE inhibitor or angiotensin II type I receptor blocker, compared with those who did not, which supports existing recommendations to continue use of this type of medication.
Remote glucose monitoring a novel tool for COVID-19 isolation
Another publication, by Gilat Shehav-Zaltzman of Sheba Medical Center, Tel Hashomer, Israel, and colleagues, describes the use of remote continuous glucose monitoring (CGM) in two hospitalized COVID-19 patients who were in isolation – one with type 1 diabetes and the other with type 2 diabetes – treated with basal-bolus insulin.
Using Medtronic CGM systems, the hospital staff was able to view patients’ real-time data uploaded to the Web from computer terminals in virus-free areas outside the patients’ rooms. The hospital’s endocrinology team had trained the intensive care staff on how to replace the sensors weekly and calibrate them twice daily.
“Converting a personal CGM system originally designed for diabetes self-management to team-based, real-time remote glucose monitoring offers a novel tool for inpatient diabetes control in COVID-19 isolation facilities,” the authors wrote.
“Such a solution in addition to ongoing remotely monitored clinical parameters (such as pulse rate, electrocardiogram, and oxygen saturation) adds to quality of diabetes care while minimizing risk of staff exposure and burden,” they observed.
Dr. Riddle and colleagues concurred: “Newer methods of remotely monitoring glucose patterns could be uniquely helpful.”
Key question: Does glycemic management make a difference?
With regard to the important issue of in-hospital control of glucose, Celestino Sardu, MD, PhD, of the University of Campania Luigi Vanvitelli, Naples, Italy, and colleagues reported on 59 patients hospitalized with confirmed COVID-19 and moderately severe pneumonia.
They were categorized as normoglycemic (n = 34) or hyperglycemic (n = 25), as well as with or without diabetes, on the basis of a diagnosis preceding the current illness. Of the 25 patients with hyperglycemia, 15 patients were treated with insulin infusion and 10 patients were not.
In a risk-adjusted analysis, both patients with hyperglycemia and patients with diabetes had a higher risk of severe disease than did those without diabetes and with normoglycemia. Patients with hyperglycemia treated with insulin infusion had a lower risk of severe disease than did patients who didn’t receive an insulin infusion.
And although they noted limitations, the authors wrote, “Our data evidenced that optimal glucose control in the immediate postadmission period for almost 18 days was associated with a significant reduction of inflammatory cytokines and procoagulative status.”
Dr. Riddle and colleagues wrote that the findings of this unrandomized comparison were interpreted “as suggesting that insulin infusion may improve outcomes.”
“If the benefits of seeking excellent glycemic control by this means are confirmed, close monitoring of glucose levels will be essential.”
More on obesity and COVID-19, this time from China
Because it has become increasingly clear that obesity is a risk factor for severe COVID-19, new data from China – where this was less apparent initially – support observations in Europe and the United States.
An article by Qingxian Cai, PhD, of Southern University of Science and Technology, Shenzhen, Guangdong, China, and colleagues looks at this. They found that, among 383 hospitalized patients with COVID-19, the 41 patients with obesity (defined as a body mass index ≥ 28 kg/m2) were significantly more likely to progress to severe disease compared with the 203 patients classified as having normal weight (BMI, 18.5-23.9), with an odds ratio of 3.4.
A similar finding comes from Feng Gao, MD, PhD, of the First Affiliated Hospital of Wenzhou (China) Medical University and colleagues, who studied 75 patients hospitalized with confirmed COVID-19 and obesity (defined as a BMI > 25 in this Asian population) to 75 patients without obesity matched by age and sex. After adjustment for clinical characteristics including the presence of diabetes, those with obesity had a threefold greater risk of progression to severe or critical COVID-19 status, with a nearly linear relationship.
Emerging from the crisis: Protect the vulnerable, increase knowledge base
As the research community emerges from the crisis, “there should be renewed efforts for multidisciplinary research ... aimed at greatly increasing the knowledge base to understand how ... the current COVID-19 threat” affects “both healthy people and people with chronic diseases and conditions,” Dr. Cefalu and colleagues concluded in their commentary.
Dr. Riddle and coauthors agreed: “We will enter a longer interval in which we must continue to support the most vulnerable populations – especially older people, those with diabetes or obesity, and those who lack the resources to limit day-to-day exposure to infection. We hope a growing sense of community will help in this task.”
Dr. Riddle has reported receiving research grant support through Oregon Health & Science University from AstraZeneca, Eli Lilly, and Novo Nordisk, and honoraria for consulting from Adocia, AstraZeneca, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Sanofi, and Theracos. Dr. Cefalu has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The American Diabetes Association has dedicated a whole section of its journal, Diabetes Care, to the topic of “Diabetes and COVID-19,” publishing a range of articles with new data to help guide physicians in caring for patients.
“Certain groups are more vulnerable to COVID-19, notably older people and those with underlying medical conditions. Because diabetes is one of the conditions associated with high risk, the diabetes community urgently needs to know more about COVID-19 and its effects on people with diabetes,” an introductory commentary noted.
Entitled “COVID-19 in people with diabetes: Urgently needed lessons from early reports,” the commentary is penned by the journal’s editor-in-chief, Matthew Riddle, MD, of Oregon Health & Science University, Portland, and colleagues.
Also writing in the same issue, William T. Cefalu, MD, and colleagues from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) noted it is known that the SARS-CoV-2 virus enters cells via the angiotensin-converting enzyme 2 (ACE-2) receptor. The ACE-2 receptor is known to be in the lungs and upper respiratory tract, “but we also know that it is expressed in other tissues such as heart, small and large intestines, and pancreas,” they wrote, and also “in the kidney.”
Hence, there are emerging reports of acute kidney injury resulting from COVID-19, as well as the impact on many other endocrine/metabolic and gastrointestinal outcomes.
“Pilot clinical studies (observational and interventional) are needed that will support the understanding or treatment of COVID-19–related diseases within the mission of the NIDDK,” they stated.
Although rapidly collected, data “offer important clues”
Some of the new ground covered in the journal articles includes an analysis of COVID-19 outcomes by type of glucose-lowering medication; remote glucose monitoring in hospitalized patients with COVID-19; a suggested approach to cardiovascular risk management in the COVID-19 era, as already reported by Medscape Medical News; and the diagnosis and management of gestational diabetes during the pandemic.
Other articles provide new data for previously reported phenomena, including obesity as a risk factor for worse COVID-19 outcomes and the role of inpatient glycemic control on COVID-19 outcomes.
“The data reported in these articles were rapidly collected and analyzed, in most cases under urgent and stressful conditions,” Dr. Riddle and colleagues cautioned. “Thus, some of the analyses are understandably limited due to missing data, incomplete follow-up, and inability to identify infected but asymptomatic patients.”
Even so, they wrote, some points are clear. “The consistency of findings in these rapidly published reports is reassuring in terms of scientific validity, but the story unfolding is worrisome.”
Specifically, while diabetes does not appear to increase the likelihood of SARS-CoV-2 infection, progression to severe illness is more likely in people with diabetes and COVID-19: They are two to three times as likely to require intensive care, and to die, compared with those infected but without diabetes.
“Neither the mechanisms underlying the increased risk nor the best interventions to limit it have yet been defined, but the studies in this collection of articles offer important clues,” Dr. Riddle and colleagues wrote.
Existing insulin use linked to COVID-19 death risk
One of the articles is a retrospective study of 904 hospitalized COVID-19 patients by Yuchen Chen, MD, of the Huazhong University of Science and Technology, Wuhan, China, and colleagues.
Among the 136 patients with diabetes, risk factors for mortality included older age (adjusted odds ratio, 1.09 per year increase; P = .001) elevated C-reactive protein (aOR, 1.12; P = .043), and insulin use (aOR, 3.58; P = .009).
“Attention needs to be paid to patients with diabetes and COVID-19 who use insulin,” the Chinese authors wrote. “Whether this was due to effects of insulin itself or to characteristics of the patients for whom it was prescribed is not clear,” Dr. Riddle and colleagues noted.
Dr. Chen and colleagues also found no difference in clinical outcomes between those diabetes patients with COVID-19 who were taking an ACE inhibitor or angiotensin II type I receptor blocker, compared with those who did not, which supports existing recommendations to continue use of this type of medication.
Remote glucose monitoring a novel tool for COVID-19 isolation
Another publication, by Gilat Shehav-Zaltzman of Sheba Medical Center, Tel Hashomer, Israel, and colleagues, describes the use of remote continuous glucose monitoring (CGM) in two hospitalized COVID-19 patients who were in isolation – one with type 1 diabetes and the other with type 2 diabetes – treated with basal-bolus insulin.
Using Medtronic CGM systems, the hospital staff was able to view patients’ real-time data uploaded to the Web from computer terminals in virus-free areas outside the patients’ rooms. The hospital’s endocrinology team had trained the intensive care staff on how to replace the sensors weekly and calibrate them twice daily.
“Converting a personal CGM system originally designed for diabetes self-management to team-based, real-time remote glucose monitoring offers a novel tool for inpatient diabetes control in COVID-19 isolation facilities,” the authors wrote.
“Such a solution in addition to ongoing remotely monitored clinical parameters (such as pulse rate, electrocardiogram, and oxygen saturation) adds to quality of diabetes care while minimizing risk of staff exposure and burden,” they observed.
Dr. Riddle and colleagues concurred: “Newer methods of remotely monitoring glucose patterns could be uniquely helpful.”
Key question: Does glycemic management make a difference?
With regard to the important issue of in-hospital control of glucose, Celestino Sardu, MD, PhD, of the University of Campania Luigi Vanvitelli, Naples, Italy, and colleagues reported on 59 patients hospitalized with confirmed COVID-19 and moderately severe pneumonia.
They were categorized as normoglycemic (n = 34) or hyperglycemic (n = 25), as well as with or without diabetes, on the basis of a diagnosis preceding the current illness. Of the 25 patients with hyperglycemia, 15 patients were treated with insulin infusion and 10 patients were not.
In a risk-adjusted analysis, both patients with hyperglycemia and patients with diabetes had a higher risk of severe disease than did those without diabetes and with normoglycemia. Patients with hyperglycemia treated with insulin infusion had a lower risk of severe disease than did patients who didn’t receive an insulin infusion.
And although they noted limitations, the authors wrote, “Our data evidenced that optimal glucose control in the immediate postadmission period for almost 18 days was associated with a significant reduction of inflammatory cytokines and procoagulative status.”
Dr. Riddle and colleagues wrote that the findings of this unrandomized comparison were interpreted “as suggesting that insulin infusion may improve outcomes.”
“If the benefits of seeking excellent glycemic control by this means are confirmed, close monitoring of glucose levels will be essential.”
More on obesity and COVID-19, this time from China
Because it has become increasingly clear that obesity is a risk factor for severe COVID-19, new data from China – where this was less apparent initially – support observations in Europe and the United States.
An article by Qingxian Cai, PhD, of Southern University of Science and Technology, Shenzhen, Guangdong, China, and colleagues looks at this. They found that, among 383 hospitalized patients with COVID-19, the 41 patients with obesity (defined as a body mass index ≥ 28 kg/m2) were significantly more likely to progress to severe disease compared with the 203 patients classified as having normal weight (BMI, 18.5-23.9), with an odds ratio of 3.4.
A similar finding comes from Feng Gao, MD, PhD, of the First Affiliated Hospital of Wenzhou (China) Medical University and colleagues, who studied 75 patients hospitalized with confirmed COVID-19 and obesity (defined as a BMI > 25 in this Asian population) to 75 patients without obesity matched by age and sex. After adjustment for clinical characteristics including the presence of diabetes, those with obesity had a threefold greater risk of progression to severe or critical COVID-19 status, with a nearly linear relationship.
Emerging from the crisis: Protect the vulnerable, increase knowledge base
As the research community emerges from the crisis, “there should be renewed efforts for multidisciplinary research ... aimed at greatly increasing the knowledge base to understand how ... the current COVID-19 threat” affects “both healthy people and people with chronic diseases and conditions,” Dr. Cefalu and colleagues concluded in their commentary.
Dr. Riddle and coauthors agreed: “We will enter a longer interval in which we must continue to support the most vulnerable populations – especially older people, those with diabetes or obesity, and those who lack the resources to limit day-to-day exposure to infection. We hope a growing sense of community will help in this task.”
Dr. Riddle has reported receiving research grant support through Oregon Health & Science University from AstraZeneca, Eli Lilly, and Novo Nordisk, and honoraria for consulting from Adocia, AstraZeneca, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Sanofi, and Theracos. Dr. Cefalu has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
More evidence hydroxychloroquine is ineffective, harmful in COVID-19
Hydroxychloroquine and chloroquine, with or without azithromycin or clarithromycin, offer no benefit in treating patients with COVID-19 and, instead, are associated with ventricular arrhythmias and higher rates of mortality, according to a major new international study.
In the largest observational study of its kind, including close to 100,000 people in 671 hospitals on six continents, investigators compared outcomes in 15,000 patients with COVID-19 treated with hydroxychloroquine and chloroquine alone or in combination with a macrolide with 80,000 control patients with COVID-19 not receiving these agents.
Treatment with any of these medications, either alone or in combination, was associated with increased death during hospitalization; compared with about 10% in control group patients, mortality rates ranged from more than 16% to almost 24% in the treated groups.
Patients treated with hydroxychloroquine plus a macrolide showed the highest rates of serious cardiac arrhythmias, and, even after accounting for demographic factors and comorbidities, this combination was found to be associated with a more than 5-fold increase in the risk of developing a serious arrhythmia while in the hospital.
“In this real-world study, the biggest yet, we looked at 100,000 patients [with COVID-19] across six continents and found not the slightest hint of benefits and only risks, and the data is pretty straightforward,” study coauthor Frank Ruschitzka, MD, director of the Heart Center at University Hospital, Zürich, said in an interview. The study was published online May 22 in The Lancet.
‘Inconclusive’ evidence
The absence of an effective treatment for COVID-19 has led to the “repurposing” of the antimalarial drug chloroquine and its analogue hydroxychloroquine, which is used for treating autoimmune disease, but this approach is based on anecdotal evidence or open-label randomized trials that have been “largely inconclusive,” the authors wrote.
Additional agents used to treat COVID-19 are second-generation macrolides (azithromycin or clarithromycin), in combination with chloroquine or hydroxychloroquine, “despite limited evidence” and the risk for ventricular arrhythmias, the authors noted.
“Our primary question was whether there was any associated benefits of the use of hydroxychloroquine, chloroquine, or a combined regimen with macrolides in treating COVID-19, and — if there was no benefit — would there be harm?” lead author Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine, Brigham and Women’s Hospital, Boston, said in an interview.
The investigators used data from a multinational registry comprising 671 hospitals that included patients (n = 96,032; mean age 53.8 years; 46.3% female) who had been hospitalized between Dec. 20, 2019, and April 14, 2020, with confirmed COVID-19 infection.
They also collected data about demographics, underlying comorbidities, and medical history, and medications that patients were taking at baseline.
Patients receiving treatment (n = 14,888) were divided into four groups: those receiving chloroquine alone (n = 1,868), those receiving chloroquine with a macrolide (n = 3,783), those receiving hydroxychloroquine alone (n = 3,016) and those receiving hydroxychloroquine with a macrolide (n = 6,221).
The remaining patients not treated with these regimens (n = 81,144) were regarded as the control group.
Most patients (65.9%) came from North America, followed by Europe (17.39%), Asia (7.9%), Africa (4.6%), South America (3.7%), and Australia (0.6%). Most (66.9%) were white, followed by patients of Asian origin (14.1%), black patients (9.4%), and Hispanic patients (6.2%).
Comorbidities and underlying conditions included obesity, hyperlipidemia, and hypertension in about 30%.
Comorbidities and underlying conditions
The investigators conducted multiple analyses to control for confounding variables, including Cox proportional hazards regression and propensity score matching analyses.
“In an observational study, there is always a chance of residual confounding, which is why we did propensity score based matched analyses,” Dr. Ruschitzka explained.
No significant differences were found in distribution of demographics and comorbidities between the groups.
As good as it gets
“We found no benefit in any of the four treatment regimens for hospitalized patients with COVID-19, but we did notice higher rates of death and serious ventricular arrhythmias in these patients, compared to the controls,” Dr. Mehra reported.
Of the patients in the control group, roughly 9.3% died during their hospitalization, compared with 16.4% of patients treated with chloroquine alone, 18.0% of those treated with hydroxychloroquine alone, 22.2% of those treated with chloroquine and a macrolide, and 23.8% of those treated with hydroxychloroquine and a macrolide.
After accounting for confounding variables, the researchers estimated that the excess mortality risk attributable to use of the drug regimen ranged from 34% to 45%.
Patients treated with any of the four regimens sustained more serious arrhythmias, compared with those in the control group (0.35), with the biggest increase seen in the group treated with the combination of hydroxychloroquine plus a macrolide (8.1%), followed by chloroquine with a macrolide (6.5%), hydroxychloroquine alone (6.1%), and chloroquine alone (4.3%).
“We were fairly reassured that, although the study was observational, the signals were robust and consistent across all regions of the world in diverse populations, and we did not see any muting of that signal, depending on region,” Dr. Mehra said.
“Two months ago, we were all scratching our heads about how to treat patients with COVID-19, and then came a drug [hydroxychloroquine] with some anecdotal evidence, but now we have 2 months more experience, and we looked to science to provide some answer,” Dr. Ruschitzka said.
“Although this was not a randomized, controlled trial, so we do not have a definite answer, the data provided in this [large, multinational] real-world study is as good as it gets and the best data we have,” he concluded.
“Let the science speak for itself”
Commenting on the study in an interview, Christian Funck-Brentano, MD, from the Hospital Pitié-Salpêtrière and Sorbonne University, both in Paris, said that, although the study is observational and therefore not as reliable as a randomized controlled trial, it is “nevertheless well-documented, studied a huge amount of people, and utilized several sensitivity methods, all of which showed the same results.”
Dr. Funck-Brentano, who is the coauthor of an accompanying editorial in The Lancet and was not involved with the study, said that “we now have no evidence that hydroxychloroquine and chloroquine alone or in combination with a macrolide do any good and we have potential evidence that they do harm and kill people.”
Also commenting on the study in an interview, David Holtgrave, PhD, dean of the School of Public Health at the State University of New York at Albany, said that, “while no one observational study alone would lead to a firm clinical recommendation, I think it is helpful for physicians and public health officials to be aware of the findings of the peer-reviewed observational studies to date and the National Institutes of Health COVID-19 treatment guidelines and the Food and Drug Administration’s statement of drug safety concern about hydroxychloroquine to inform their decision-making as we await the results of randomized clinical trials of these drugs for the treatment of COVID-19,” said Dr. Holtgrave, who was not involved with the study.
He added that, to his knowledge, there are “still no published studies of prophylactic use of these drugs to prevent COVID-19.”
Dr. Mehra emphasized that a cardinal principle of practicing medicine is “first do no harm” and “even in situations where you believe a desperate disease calls for desperate measures, responsible physicians should take a step back and ask if we are doing harm, and until we can say we aren’t, I don’t think it’s wise to push something like this in the absence of good efficacy data.”
Dr. Ruschitzka added that those who are encouraging the use of these agents “should review their decision based on today’s data and let the science speak for itself.”
The study was supported by the William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston. Dr. Mehra reported personal fees from Abbott, Medtronic, Janssen, Mesoblast, Portola, Bayer, Baim Institute for Clinical Research, NuPulseCV, FineHeart, Leviticus, Roivant, and Triple Gene. Dr. Ruschitzka was paid for time spent as a committee member for clinical trials, advisory boards, other forms of consulting, and lectures or presentations; these payments were made directly to the University of Zürich and no personal payments were received in relation to these trials or other activities. Dr. Funck-Brentano, his coauthor, and Dr. Holtgrave declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Hydroxychloroquine and chloroquine, with or without azithromycin or clarithromycin, offer no benefit in treating patients with COVID-19 and, instead, are associated with ventricular arrhythmias and higher rates of mortality, according to a major new international study.
In the largest observational study of its kind, including close to 100,000 people in 671 hospitals on six continents, investigators compared outcomes in 15,000 patients with COVID-19 treated with hydroxychloroquine and chloroquine alone or in combination with a macrolide with 80,000 control patients with COVID-19 not receiving these agents.
Treatment with any of these medications, either alone or in combination, was associated with increased death during hospitalization; compared with about 10% in control group patients, mortality rates ranged from more than 16% to almost 24% in the treated groups.
Patients treated with hydroxychloroquine plus a macrolide showed the highest rates of serious cardiac arrhythmias, and, even after accounting for demographic factors and comorbidities, this combination was found to be associated with a more than 5-fold increase in the risk of developing a serious arrhythmia while in the hospital.
“In this real-world study, the biggest yet, we looked at 100,000 patients [with COVID-19] across six continents and found not the slightest hint of benefits and only risks, and the data is pretty straightforward,” study coauthor Frank Ruschitzka, MD, director of the Heart Center at University Hospital, Zürich, said in an interview. The study was published online May 22 in The Lancet.
‘Inconclusive’ evidence
The absence of an effective treatment for COVID-19 has led to the “repurposing” of the antimalarial drug chloroquine and its analogue hydroxychloroquine, which is used for treating autoimmune disease, but this approach is based on anecdotal evidence or open-label randomized trials that have been “largely inconclusive,” the authors wrote.
Additional agents used to treat COVID-19 are second-generation macrolides (azithromycin or clarithromycin), in combination with chloroquine or hydroxychloroquine, “despite limited evidence” and the risk for ventricular arrhythmias, the authors noted.
“Our primary question was whether there was any associated benefits of the use of hydroxychloroquine, chloroquine, or a combined regimen with macrolides in treating COVID-19, and — if there was no benefit — would there be harm?” lead author Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine, Brigham and Women’s Hospital, Boston, said in an interview.
The investigators used data from a multinational registry comprising 671 hospitals that included patients (n = 96,032; mean age 53.8 years; 46.3% female) who had been hospitalized between Dec. 20, 2019, and April 14, 2020, with confirmed COVID-19 infection.
They also collected data about demographics, underlying comorbidities, and medical history, and medications that patients were taking at baseline.
Patients receiving treatment (n = 14,888) were divided into four groups: those receiving chloroquine alone (n = 1,868), those receiving chloroquine with a macrolide (n = 3,783), those receiving hydroxychloroquine alone (n = 3,016) and those receiving hydroxychloroquine with a macrolide (n = 6,221).
The remaining patients not treated with these regimens (n = 81,144) were regarded as the control group.
Most patients (65.9%) came from North America, followed by Europe (17.39%), Asia (7.9%), Africa (4.6%), South America (3.7%), and Australia (0.6%). Most (66.9%) were white, followed by patients of Asian origin (14.1%), black patients (9.4%), and Hispanic patients (6.2%).
Comorbidities and underlying conditions included obesity, hyperlipidemia, and hypertension in about 30%.
Comorbidities and underlying conditions
The investigators conducted multiple analyses to control for confounding variables, including Cox proportional hazards regression and propensity score matching analyses.
“In an observational study, there is always a chance of residual confounding, which is why we did propensity score based matched analyses,” Dr. Ruschitzka explained.
No significant differences were found in distribution of demographics and comorbidities between the groups.
As good as it gets
“We found no benefit in any of the four treatment regimens for hospitalized patients with COVID-19, but we did notice higher rates of death and serious ventricular arrhythmias in these patients, compared to the controls,” Dr. Mehra reported.
Of the patients in the control group, roughly 9.3% died during their hospitalization, compared with 16.4% of patients treated with chloroquine alone, 18.0% of those treated with hydroxychloroquine alone, 22.2% of those treated with chloroquine and a macrolide, and 23.8% of those treated with hydroxychloroquine and a macrolide.
After accounting for confounding variables, the researchers estimated that the excess mortality risk attributable to use of the drug regimen ranged from 34% to 45%.
Patients treated with any of the four regimens sustained more serious arrhythmias, compared with those in the control group (0.35), with the biggest increase seen in the group treated with the combination of hydroxychloroquine plus a macrolide (8.1%), followed by chloroquine with a macrolide (6.5%), hydroxychloroquine alone (6.1%), and chloroquine alone (4.3%).
“We were fairly reassured that, although the study was observational, the signals were robust and consistent across all regions of the world in diverse populations, and we did not see any muting of that signal, depending on region,” Dr. Mehra said.
“Two months ago, we were all scratching our heads about how to treat patients with COVID-19, and then came a drug [hydroxychloroquine] with some anecdotal evidence, but now we have 2 months more experience, and we looked to science to provide some answer,” Dr. Ruschitzka said.
“Although this was not a randomized, controlled trial, so we do not have a definite answer, the data provided in this [large, multinational] real-world study is as good as it gets and the best data we have,” he concluded.
“Let the science speak for itself”
Commenting on the study in an interview, Christian Funck-Brentano, MD, from the Hospital Pitié-Salpêtrière and Sorbonne University, both in Paris, said that, although the study is observational and therefore not as reliable as a randomized controlled trial, it is “nevertheless well-documented, studied a huge amount of people, and utilized several sensitivity methods, all of which showed the same results.”
Dr. Funck-Brentano, who is the coauthor of an accompanying editorial in The Lancet and was not involved with the study, said that “we now have no evidence that hydroxychloroquine and chloroquine alone or in combination with a macrolide do any good and we have potential evidence that they do harm and kill people.”
Also commenting on the study in an interview, David Holtgrave, PhD, dean of the School of Public Health at the State University of New York at Albany, said that, “while no one observational study alone would lead to a firm clinical recommendation, I think it is helpful for physicians and public health officials to be aware of the findings of the peer-reviewed observational studies to date and the National Institutes of Health COVID-19 treatment guidelines and the Food and Drug Administration’s statement of drug safety concern about hydroxychloroquine to inform their decision-making as we await the results of randomized clinical trials of these drugs for the treatment of COVID-19,” said Dr. Holtgrave, who was not involved with the study.
He added that, to his knowledge, there are “still no published studies of prophylactic use of these drugs to prevent COVID-19.”
Dr. Mehra emphasized that a cardinal principle of practicing medicine is “first do no harm” and “even in situations where you believe a desperate disease calls for desperate measures, responsible physicians should take a step back and ask if we are doing harm, and until we can say we aren’t, I don’t think it’s wise to push something like this in the absence of good efficacy data.”
Dr. Ruschitzka added that those who are encouraging the use of these agents “should review their decision based on today’s data and let the science speak for itself.”
The study was supported by the William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston. Dr. Mehra reported personal fees from Abbott, Medtronic, Janssen, Mesoblast, Portola, Bayer, Baim Institute for Clinical Research, NuPulseCV, FineHeart, Leviticus, Roivant, and Triple Gene. Dr. Ruschitzka was paid for time spent as a committee member for clinical trials, advisory boards, other forms of consulting, and lectures or presentations; these payments were made directly to the University of Zürich and no personal payments were received in relation to these trials or other activities. Dr. Funck-Brentano, his coauthor, and Dr. Holtgrave declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Hydroxychloroquine and chloroquine, with or without azithromycin or clarithromycin, offer no benefit in treating patients with COVID-19 and, instead, are associated with ventricular arrhythmias and higher rates of mortality, according to a major new international study.
In the largest observational study of its kind, including close to 100,000 people in 671 hospitals on six continents, investigators compared outcomes in 15,000 patients with COVID-19 treated with hydroxychloroquine and chloroquine alone or in combination with a macrolide with 80,000 control patients with COVID-19 not receiving these agents.
Treatment with any of these medications, either alone or in combination, was associated with increased death during hospitalization; compared with about 10% in control group patients, mortality rates ranged from more than 16% to almost 24% in the treated groups.
Patients treated with hydroxychloroquine plus a macrolide showed the highest rates of serious cardiac arrhythmias, and, even after accounting for demographic factors and comorbidities, this combination was found to be associated with a more than 5-fold increase in the risk of developing a serious arrhythmia while in the hospital.
“In this real-world study, the biggest yet, we looked at 100,000 patients [with COVID-19] across six continents and found not the slightest hint of benefits and only risks, and the data is pretty straightforward,” study coauthor Frank Ruschitzka, MD, director of the Heart Center at University Hospital, Zürich, said in an interview. The study was published online May 22 in The Lancet.
‘Inconclusive’ evidence
The absence of an effective treatment for COVID-19 has led to the “repurposing” of the antimalarial drug chloroquine and its analogue hydroxychloroquine, which is used for treating autoimmune disease, but this approach is based on anecdotal evidence or open-label randomized trials that have been “largely inconclusive,” the authors wrote.
Additional agents used to treat COVID-19 are second-generation macrolides (azithromycin or clarithromycin), in combination with chloroquine or hydroxychloroquine, “despite limited evidence” and the risk for ventricular arrhythmias, the authors noted.
“Our primary question was whether there was any associated benefits of the use of hydroxychloroquine, chloroquine, or a combined regimen with macrolides in treating COVID-19, and — if there was no benefit — would there be harm?” lead author Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine, Brigham and Women’s Hospital, Boston, said in an interview.
The investigators used data from a multinational registry comprising 671 hospitals that included patients (n = 96,032; mean age 53.8 years; 46.3% female) who had been hospitalized between Dec. 20, 2019, and April 14, 2020, with confirmed COVID-19 infection.
They also collected data about demographics, underlying comorbidities, and medical history, and medications that patients were taking at baseline.
Patients receiving treatment (n = 14,888) were divided into four groups: those receiving chloroquine alone (n = 1,868), those receiving chloroquine with a macrolide (n = 3,783), those receiving hydroxychloroquine alone (n = 3,016) and those receiving hydroxychloroquine with a macrolide (n = 6,221).
The remaining patients not treated with these regimens (n = 81,144) were regarded as the control group.
Most patients (65.9%) came from North America, followed by Europe (17.39%), Asia (7.9%), Africa (4.6%), South America (3.7%), and Australia (0.6%). Most (66.9%) were white, followed by patients of Asian origin (14.1%), black patients (9.4%), and Hispanic patients (6.2%).
Comorbidities and underlying conditions included obesity, hyperlipidemia, and hypertension in about 30%.
Comorbidities and underlying conditions
The investigators conducted multiple analyses to control for confounding variables, including Cox proportional hazards regression and propensity score matching analyses.
“In an observational study, there is always a chance of residual confounding, which is why we did propensity score based matched analyses,” Dr. Ruschitzka explained.
No significant differences were found in distribution of demographics and comorbidities between the groups.
As good as it gets
“We found no benefit in any of the four treatment regimens for hospitalized patients with COVID-19, but we did notice higher rates of death and serious ventricular arrhythmias in these patients, compared to the controls,” Dr. Mehra reported.
Of the patients in the control group, roughly 9.3% died during their hospitalization, compared with 16.4% of patients treated with chloroquine alone, 18.0% of those treated with hydroxychloroquine alone, 22.2% of those treated with chloroquine and a macrolide, and 23.8% of those treated with hydroxychloroquine and a macrolide.
After accounting for confounding variables, the researchers estimated that the excess mortality risk attributable to use of the drug regimen ranged from 34% to 45%.
Patients treated with any of the four regimens sustained more serious arrhythmias, compared with those in the control group (0.35), with the biggest increase seen in the group treated with the combination of hydroxychloroquine plus a macrolide (8.1%), followed by chloroquine with a macrolide (6.5%), hydroxychloroquine alone (6.1%), and chloroquine alone (4.3%).
“We were fairly reassured that, although the study was observational, the signals were robust and consistent across all regions of the world in diverse populations, and we did not see any muting of that signal, depending on region,” Dr. Mehra said.
“Two months ago, we were all scratching our heads about how to treat patients with COVID-19, and then came a drug [hydroxychloroquine] with some anecdotal evidence, but now we have 2 months more experience, and we looked to science to provide some answer,” Dr. Ruschitzka said.
“Although this was not a randomized, controlled trial, so we do not have a definite answer, the data provided in this [large, multinational] real-world study is as good as it gets and the best data we have,” he concluded.
“Let the science speak for itself”
Commenting on the study in an interview, Christian Funck-Brentano, MD, from the Hospital Pitié-Salpêtrière and Sorbonne University, both in Paris, said that, although the study is observational and therefore not as reliable as a randomized controlled trial, it is “nevertheless well-documented, studied a huge amount of people, and utilized several sensitivity methods, all of which showed the same results.”
Dr. Funck-Brentano, who is the coauthor of an accompanying editorial in The Lancet and was not involved with the study, said that “we now have no evidence that hydroxychloroquine and chloroquine alone or in combination with a macrolide do any good and we have potential evidence that they do harm and kill people.”
Also commenting on the study in an interview, David Holtgrave, PhD, dean of the School of Public Health at the State University of New York at Albany, said that, “while no one observational study alone would lead to a firm clinical recommendation, I think it is helpful for physicians and public health officials to be aware of the findings of the peer-reviewed observational studies to date and the National Institutes of Health COVID-19 treatment guidelines and the Food and Drug Administration’s statement of drug safety concern about hydroxychloroquine to inform their decision-making as we await the results of randomized clinical trials of these drugs for the treatment of COVID-19,” said Dr. Holtgrave, who was not involved with the study.
He added that, to his knowledge, there are “still no published studies of prophylactic use of these drugs to prevent COVID-19.”
Dr. Mehra emphasized that a cardinal principle of practicing medicine is “first do no harm” and “even in situations where you believe a desperate disease calls for desperate measures, responsible physicians should take a step back and ask if we are doing harm, and until we can say we aren’t, I don’t think it’s wise to push something like this in the absence of good efficacy data.”
Dr. Ruschitzka added that those who are encouraging the use of these agents “should review their decision based on today’s data and let the science speak for itself.”
The study was supported by the William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston. Dr. Mehra reported personal fees from Abbott, Medtronic, Janssen, Mesoblast, Portola, Bayer, Baim Institute for Clinical Research, NuPulseCV, FineHeart, Leviticus, Roivant, and Triple Gene. Dr. Ruschitzka was paid for time spent as a committee member for clinical trials, advisory boards, other forms of consulting, and lectures or presentations; these payments were made directly to the University of Zürich and no personal payments were received in relation to these trials or other activities. Dr. Funck-Brentano, his coauthor, and Dr. Holtgrave declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
ACE inhibitors and severe COVID-19: Protective in older patients?
.
In addition, a new meta-analysis of all the available data on the use of ACE inhibitors and angiotensin-receptor blockers (ARBs) in COVID-19–infected patients has concluded that these drugs are not associated with more severe disease and do not increase susceptibility to infection.
The observational study, which was published on the MedRxiv preprint server on May 19 and has not yet been peer reviewed, was conducted by the health insurance company United Heath Group and by Yale University, New Haven, Conn.
The investigators analyzed data from 10,000 patients from across the United States who had tested positive for COVID-19, who were enrolled in Medicare Advantage insurance plans or were commercially insured, and who had received a prescription for one or more antihypertensive medications.
Results showed that the use of ACE inhibitors was associated with an almost 40% lower risk for COVID-19 hospitalization for older people enrolled in Medicare Advantage plans. No such benefit was seen in the younger commercially insured patients or in either group with ARBs.
At a telephone media briefing on the study, senior investigator Harlan M. Krumholz, MD, said: “We don’t believe this is enough info to change practice, but we do think this is an interesting and intriguing result.
“These findings merit a clinical trial to formally test whether ACE inhibitors – which are cheap, widely available, and well-tolerated drugs – can reduce hospitalization of patients infected with COVID-19,” added Dr. Krumholz, professor of medicine at Yale and director of the Yale New Haven Hospital Center for Outcomes Research.
A pragmatic clinical trial is now being planned. In this trial, 10,000 older people who test positive for COVID-19 will be randomly assigned to receive either a low dose of an ACE inhibitor or placebo. It is hoped that recruitment for the trial will begin in June of 2020. It is open to all eligible Americans who are older than 50 years, who test negative for COVID-19, and who are not taking medications for hypertension. Prospective patients can sign up at a dedicated website.
The randomized trial, also conducted by United Health Group and Yale, is said to be “one of the first virtual COVID-19 clinical trials to be launched at scale.”
For the observational study, the researchers identified 2,263 people who were receiving medication for hypertension and who tested positive for COVID-19. Of these, approximately two-thirds were older, Medicare Advantage enrollees; one-third were younger, commercially insured individuals.
In a propensity score–matched analysis, the investigators matched 441 patients who were taking ACE inhibitors to 441 patients who were taking other antihypertensive agents; and 412 patients who were receiving an ARB to 412 patients who were receiving other antihypertensive agents.
Results showed that during a median of 30 days after testing positive, 12.7% of the cohort were hospitalized for COVID-19. In propensity score–matched analyses, neither ACE inhibitors (hazard ratio [HR], 0.77; P = .18) nor ARBs (HR, 0.88; P =.48) were significantly associated with risk for hospitalization.
However, in analyses stratified by the insurance group, ACE inhibitors (but not ARBs) were associated with a significant lower risk for hospitalization among the Medicare group (HR, 0.61; P = .02) but not among the commercially insured group (HR, 2.14; P = .12).
A second study examined outcomes of 7,933 individuals with hypertension who were hospitalized with COVID-19 (92% of these patients were Medicare Advantage enrollees). Of these, 14.2% died, 59.5% survived to discharge, and 26.3% underwent ongoing hospitalization. In propensity score–matched analyses, use of neither an ACE inhibitor (HR, 0.97; P = .74) nor an ARB (HR, 1.15; P = .15) was associated with risk of in-hospital mortality.
The researchers said their findings are consistent with prior evidence from randomized clinical trials suggesting a reduced risk for pneumonia with ACE inhibitors that is not observed with ARBs.
They also cited some preclinical evidence that they said suggests a possible protective role for ACE inhibitors in COVID-19: that ACE inhibitors, but not ARBs, are associated with the upregulation of ACE2 receptors, which modulate the local interactions of the renin-angiotensin-aldosterone system in the lung tissue.
“The presence of ACE2 receptors, therefore, exerts a protective effect against the development of acute lung injury in infections with SARS coronaviruses, which lead to dysregulation of these mechanisms and endothelial damage,” they added. “Further, our observations do not support theoretical concerns of adverse outcomes due to enhanced virulence of SARS coronaviruses due to overexpression of ACE2 receptors in cell cultures – an indirect binding site for these viruses.”
The authors also noted that their findings have “important implications” for four ongoing randomized trials of ACE inhibitors/ARBs in COVID-19, “as none of them align with the observations of our study.”
They pointed out that of the four ongoing trials, three are testing the use of ACE inhibitors or ARBs in the treatment of hospitalized COVID-19 patients, and one is testing the use of a 10-day course of ARBs after a positive SARS-CoV-2 test to prevent hospitalization.
Experts cautious
However, two cardiovascular experts who were asked to comment on this latest study were not overly optimistic about the data.
Michael A. Weber, MD, professor of medicine at the State University of New York, Brooklyn, said: “This report adds to the growing number of observational studies that show varying effects of ACE inhibitors and ARBs in increasing or decreasing hospitalizations for COVID-19 and the likelihood of in-hospital mortality. Overall, this new report differs from others in the remarkable effects of insurance coverage: In particular, for ACE inhibitors, there was a 40% reduction in fatal events in Medicare patients but a twofold increase in patients using commercial insurance – albeit the test for heterogeneity when comparing the two groups did not quite reach statistical significance.
“In essence, these authors are saying that ACE inhibitors are highly protective in patients aged 65 or older but bordering on harmful in patients aged below 65. I agree that it’s worthwhile to check this finding in a prospective trial ... but this hypothesis does seem to be a reach.”
Dr. Weber noted that both ACE inhibitors and ARBs increase the level of the ACE2 enzyme to which the COVID-19 virus binds in the lungs.
“The ACE inhibitors do so by inhibiting the enzyme’s action and thus stimulate further enzyme production; the ARBs block the effects of angiotensin II, which results in high angiotensin II levels that also upregulate ACE2 production,” he said. “Perhaps the ACE inhibitors, by binding to the ACE enzyme, can in some way interfere with the enzyme’s uptake of the COVID virus and thus provide some measure of clinical protection. This is possible, but why would this effect be apparent only in older people?”
John McMurray, MD, professor of medical cardiology at the University of Glasgow, Scotland, added: “This looks like a subgroup of a subgroup type analysis based on small numbers of events – I think there were only 77 hospitalizations among the 722 patients treated with an ACE inhibitor, and the Medicare Advantage subgroup was only 581 of those 722 patients.
“The hazard ratio had wide 95% CI [confidence interval] and a modest P value,” Dr. McMurray added. “So yes, interesting and hypothesis-generating, but not definitive.”
New meta-analysis
The new meta-analysis of all data so far available on ACE inhibitor and ARB use for patients with COVID-19 was published online in Annals of Internal Medicine on May 15.
The analysis is a living, systematic review with ongoing literature surveillance and critical appraisal, which will be updated as new data become available. It included 14 observational studies.
The authors, led by Katherine M. Mackey, MD, VA Portland Health Care System, Oregon, concluded: “High-certainty evidence suggests that ACE-inhibitor or ARB use is not associated with more severe COVID-19 disease, and moderate certainty evidence suggested no association between use of these medications and positive SARS-CoV-2 test results among symptomatic patients. Whether these medications increase the risk for mild or asymptomatic disease or are beneficial in COVID-19 treatment remains uncertain.”
In an accompanying editorial, William G. Kussmaul III, MD, Drexel University, Philadelphia, said that initial fears that these drugs may be harmful for patients with COVID-19 now seem to have been unfounded.
“We now have reasonable reassurance that drugs that alter the renin-angiotensin system do not pose substantial threats as either COVID-19 risk factors or severity multipliers,” he wrote.
A version of this article originally appeared on Medscape.com.
.
In addition, a new meta-analysis of all the available data on the use of ACE inhibitors and angiotensin-receptor blockers (ARBs) in COVID-19–infected patients has concluded that these drugs are not associated with more severe disease and do not increase susceptibility to infection.
The observational study, which was published on the MedRxiv preprint server on May 19 and has not yet been peer reviewed, was conducted by the health insurance company United Heath Group and by Yale University, New Haven, Conn.
The investigators analyzed data from 10,000 patients from across the United States who had tested positive for COVID-19, who were enrolled in Medicare Advantage insurance plans or were commercially insured, and who had received a prescription for one or more antihypertensive medications.
Results showed that the use of ACE inhibitors was associated with an almost 40% lower risk for COVID-19 hospitalization for older people enrolled in Medicare Advantage plans. No such benefit was seen in the younger commercially insured patients or in either group with ARBs.
At a telephone media briefing on the study, senior investigator Harlan M. Krumholz, MD, said: “We don’t believe this is enough info to change practice, but we do think this is an interesting and intriguing result.
“These findings merit a clinical trial to formally test whether ACE inhibitors – which are cheap, widely available, and well-tolerated drugs – can reduce hospitalization of patients infected with COVID-19,” added Dr. Krumholz, professor of medicine at Yale and director of the Yale New Haven Hospital Center for Outcomes Research.
A pragmatic clinical trial is now being planned. In this trial, 10,000 older people who test positive for COVID-19 will be randomly assigned to receive either a low dose of an ACE inhibitor or placebo. It is hoped that recruitment for the trial will begin in June of 2020. It is open to all eligible Americans who are older than 50 years, who test negative for COVID-19, and who are not taking medications for hypertension. Prospective patients can sign up at a dedicated website.
The randomized trial, also conducted by United Health Group and Yale, is said to be “one of the first virtual COVID-19 clinical trials to be launched at scale.”
For the observational study, the researchers identified 2,263 people who were receiving medication for hypertension and who tested positive for COVID-19. Of these, approximately two-thirds were older, Medicare Advantage enrollees; one-third were younger, commercially insured individuals.
In a propensity score–matched analysis, the investigators matched 441 patients who were taking ACE inhibitors to 441 patients who were taking other antihypertensive agents; and 412 patients who were receiving an ARB to 412 patients who were receiving other antihypertensive agents.
Results showed that during a median of 30 days after testing positive, 12.7% of the cohort were hospitalized for COVID-19. In propensity score–matched analyses, neither ACE inhibitors (hazard ratio [HR], 0.77; P = .18) nor ARBs (HR, 0.88; P =.48) were significantly associated with risk for hospitalization.
However, in analyses stratified by the insurance group, ACE inhibitors (but not ARBs) were associated with a significant lower risk for hospitalization among the Medicare group (HR, 0.61; P = .02) but not among the commercially insured group (HR, 2.14; P = .12).
A second study examined outcomes of 7,933 individuals with hypertension who were hospitalized with COVID-19 (92% of these patients were Medicare Advantage enrollees). Of these, 14.2% died, 59.5% survived to discharge, and 26.3% underwent ongoing hospitalization. In propensity score–matched analyses, use of neither an ACE inhibitor (HR, 0.97; P = .74) nor an ARB (HR, 1.15; P = .15) was associated with risk of in-hospital mortality.
The researchers said their findings are consistent with prior evidence from randomized clinical trials suggesting a reduced risk for pneumonia with ACE inhibitors that is not observed with ARBs.
They also cited some preclinical evidence that they said suggests a possible protective role for ACE inhibitors in COVID-19: that ACE inhibitors, but not ARBs, are associated with the upregulation of ACE2 receptors, which modulate the local interactions of the renin-angiotensin-aldosterone system in the lung tissue.
“The presence of ACE2 receptors, therefore, exerts a protective effect against the development of acute lung injury in infections with SARS coronaviruses, which lead to dysregulation of these mechanisms and endothelial damage,” they added. “Further, our observations do not support theoretical concerns of adverse outcomes due to enhanced virulence of SARS coronaviruses due to overexpression of ACE2 receptors in cell cultures – an indirect binding site for these viruses.”
The authors also noted that their findings have “important implications” for four ongoing randomized trials of ACE inhibitors/ARBs in COVID-19, “as none of them align with the observations of our study.”
They pointed out that of the four ongoing trials, three are testing the use of ACE inhibitors or ARBs in the treatment of hospitalized COVID-19 patients, and one is testing the use of a 10-day course of ARBs after a positive SARS-CoV-2 test to prevent hospitalization.
Experts cautious
However, two cardiovascular experts who were asked to comment on this latest study were not overly optimistic about the data.
Michael A. Weber, MD, professor of medicine at the State University of New York, Brooklyn, said: “This report adds to the growing number of observational studies that show varying effects of ACE inhibitors and ARBs in increasing or decreasing hospitalizations for COVID-19 and the likelihood of in-hospital mortality. Overall, this new report differs from others in the remarkable effects of insurance coverage: In particular, for ACE inhibitors, there was a 40% reduction in fatal events in Medicare patients but a twofold increase in patients using commercial insurance – albeit the test for heterogeneity when comparing the two groups did not quite reach statistical significance.
“In essence, these authors are saying that ACE inhibitors are highly protective in patients aged 65 or older but bordering on harmful in patients aged below 65. I agree that it’s worthwhile to check this finding in a prospective trial ... but this hypothesis does seem to be a reach.”
Dr. Weber noted that both ACE inhibitors and ARBs increase the level of the ACE2 enzyme to which the COVID-19 virus binds in the lungs.
“The ACE inhibitors do so by inhibiting the enzyme’s action and thus stimulate further enzyme production; the ARBs block the effects of angiotensin II, which results in high angiotensin II levels that also upregulate ACE2 production,” he said. “Perhaps the ACE inhibitors, by binding to the ACE enzyme, can in some way interfere with the enzyme’s uptake of the COVID virus and thus provide some measure of clinical protection. This is possible, but why would this effect be apparent only in older people?”
John McMurray, MD, professor of medical cardiology at the University of Glasgow, Scotland, added: “This looks like a subgroup of a subgroup type analysis based on small numbers of events – I think there were only 77 hospitalizations among the 722 patients treated with an ACE inhibitor, and the Medicare Advantage subgroup was only 581 of those 722 patients.
“The hazard ratio had wide 95% CI [confidence interval] and a modest P value,” Dr. McMurray added. “So yes, interesting and hypothesis-generating, but not definitive.”
New meta-analysis
The new meta-analysis of all data so far available on ACE inhibitor and ARB use for patients with COVID-19 was published online in Annals of Internal Medicine on May 15.
The analysis is a living, systematic review with ongoing literature surveillance and critical appraisal, which will be updated as new data become available. It included 14 observational studies.
The authors, led by Katherine M. Mackey, MD, VA Portland Health Care System, Oregon, concluded: “High-certainty evidence suggests that ACE-inhibitor or ARB use is not associated with more severe COVID-19 disease, and moderate certainty evidence suggested no association between use of these medications and positive SARS-CoV-2 test results among symptomatic patients. Whether these medications increase the risk for mild or asymptomatic disease or are beneficial in COVID-19 treatment remains uncertain.”
In an accompanying editorial, William G. Kussmaul III, MD, Drexel University, Philadelphia, said that initial fears that these drugs may be harmful for patients with COVID-19 now seem to have been unfounded.
“We now have reasonable reassurance that drugs that alter the renin-angiotensin system do not pose substantial threats as either COVID-19 risk factors or severity multipliers,” he wrote.
A version of this article originally appeared on Medscape.com.
.
In addition, a new meta-analysis of all the available data on the use of ACE inhibitors and angiotensin-receptor blockers (ARBs) in COVID-19–infected patients has concluded that these drugs are not associated with more severe disease and do not increase susceptibility to infection.
The observational study, which was published on the MedRxiv preprint server on May 19 and has not yet been peer reviewed, was conducted by the health insurance company United Heath Group and by Yale University, New Haven, Conn.
The investigators analyzed data from 10,000 patients from across the United States who had tested positive for COVID-19, who were enrolled in Medicare Advantage insurance plans or were commercially insured, and who had received a prescription for one or more antihypertensive medications.
Results showed that the use of ACE inhibitors was associated with an almost 40% lower risk for COVID-19 hospitalization for older people enrolled in Medicare Advantage plans. No such benefit was seen in the younger commercially insured patients or in either group with ARBs.
At a telephone media briefing on the study, senior investigator Harlan M. Krumholz, MD, said: “We don’t believe this is enough info to change practice, but we do think this is an interesting and intriguing result.
“These findings merit a clinical trial to formally test whether ACE inhibitors – which are cheap, widely available, and well-tolerated drugs – can reduce hospitalization of patients infected with COVID-19,” added Dr. Krumholz, professor of medicine at Yale and director of the Yale New Haven Hospital Center for Outcomes Research.
A pragmatic clinical trial is now being planned. In this trial, 10,000 older people who test positive for COVID-19 will be randomly assigned to receive either a low dose of an ACE inhibitor or placebo. It is hoped that recruitment for the trial will begin in June of 2020. It is open to all eligible Americans who are older than 50 years, who test negative for COVID-19, and who are not taking medications for hypertension. Prospective patients can sign up at a dedicated website.
The randomized trial, also conducted by United Health Group and Yale, is said to be “one of the first virtual COVID-19 clinical trials to be launched at scale.”
For the observational study, the researchers identified 2,263 people who were receiving medication for hypertension and who tested positive for COVID-19. Of these, approximately two-thirds were older, Medicare Advantage enrollees; one-third were younger, commercially insured individuals.
In a propensity score–matched analysis, the investigators matched 441 patients who were taking ACE inhibitors to 441 patients who were taking other antihypertensive agents; and 412 patients who were receiving an ARB to 412 patients who were receiving other antihypertensive agents.
Results showed that during a median of 30 days after testing positive, 12.7% of the cohort were hospitalized for COVID-19. In propensity score–matched analyses, neither ACE inhibitors (hazard ratio [HR], 0.77; P = .18) nor ARBs (HR, 0.88; P =.48) were significantly associated with risk for hospitalization.
However, in analyses stratified by the insurance group, ACE inhibitors (but not ARBs) were associated with a significant lower risk for hospitalization among the Medicare group (HR, 0.61; P = .02) but not among the commercially insured group (HR, 2.14; P = .12).
A second study examined outcomes of 7,933 individuals with hypertension who were hospitalized with COVID-19 (92% of these patients were Medicare Advantage enrollees). Of these, 14.2% died, 59.5% survived to discharge, and 26.3% underwent ongoing hospitalization. In propensity score–matched analyses, use of neither an ACE inhibitor (HR, 0.97; P = .74) nor an ARB (HR, 1.15; P = .15) was associated with risk of in-hospital mortality.
The researchers said their findings are consistent with prior evidence from randomized clinical trials suggesting a reduced risk for pneumonia with ACE inhibitors that is not observed with ARBs.
They also cited some preclinical evidence that they said suggests a possible protective role for ACE inhibitors in COVID-19: that ACE inhibitors, but not ARBs, are associated with the upregulation of ACE2 receptors, which modulate the local interactions of the renin-angiotensin-aldosterone system in the lung tissue.
“The presence of ACE2 receptors, therefore, exerts a protective effect against the development of acute lung injury in infections with SARS coronaviruses, which lead to dysregulation of these mechanisms and endothelial damage,” they added. “Further, our observations do not support theoretical concerns of adverse outcomes due to enhanced virulence of SARS coronaviruses due to overexpression of ACE2 receptors in cell cultures – an indirect binding site for these viruses.”
The authors also noted that their findings have “important implications” for four ongoing randomized trials of ACE inhibitors/ARBs in COVID-19, “as none of them align with the observations of our study.”
They pointed out that of the four ongoing trials, three are testing the use of ACE inhibitors or ARBs in the treatment of hospitalized COVID-19 patients, and one is testing the use of a 10-day course of ARBs after a positive SARS-CoV-2 test to prevent hospitalization.
Experts cautious
However, two cardiovascular experts who were asked to comment on this latest study were not overly optimistic about the data.
Michael A. Weber, MD, professor of medicine at the State University of New York, Brooklyn, said: “This report adds to the growing number of observational studies that show varying effects of ACE inhibitors and ARBs in increasing or decreasing hospitalizations for COVID-19 and the likelihood of in-hospital mortality. Overall, this new report differs from others in the remarkable effects of insurance coverage: In particular, for ACE inhibitors, there was a 40% reduction in fatal events in Medicare patients but a twofold increase in patients using commercial insurance – albeit the test for heterogeneity when comparing the two groups did not quite reach statistical significance.
“In essence, these authors are saying that ACE inhibitors are highly protective in patients aged 65 or older but bordering on harmful in patients aged below 65. I agree that it’s worthwhile to check this finding in a prospective trial ... but this hypothesis does seem to be a reach.”
Dr. Weber noted that both ACE inhibitors and ARBs increase the level of the ACE2 enzyme to which the COVID-19 virus binds in the lungs.
“The ACE inhibitors do so by inhibiting the enzyme’s action and thus stimulate further enzyme production; the ARBs block the effects of angiotensin II, which results in high angiotensin II levels that also upregulate ACE2 production,” he said. “Perhaps the ACE inhibitors, by binding to the ACE enzyme, can in some way interfere with the enzyme’s uptake of the COVID virus and thus provide some measure of clinical protection. This is possible, but why would this effect be apparent only in older people?”
John McMurray, MD, professor of medical cardiology at the University of Glasgow, Scotland, added: “This looks like a subgroup of a subgroup type analysis based on small numbers of events – I think there were only 77 hospitalizations among the 722 patients treated with an ACE inhibitor, and the Medicare Advantage subgroup was only 581 of those 722 patients.
“The hazard ratio had wide 95% CI [confidence interval] and a modest P value,” Dr. McMurray added. “So yes, interesting and hypothesis-generating, but not definitive.”
New meta-analysis
The new meta-analysis of all data so far available on ACE inhibitor and ARB use for patients with COVID-19 was published online in Annals of Internal Medicine on May 15.
The analysis is a living, systematic review with ongoing literature surveillance and critical appraisal, which will be updated as new data become available. It included 14 observational studies.
The authors, led by Katherine M. Mackey, MD, VA Portland Health Care System, Oregon, concluded: “High-certainty evidence suggests that ACE-inhibitor or ARB use is not associated with more severe COVID-19 disease, and moderate certainty evidence suggested no association between use of these medications and positive SARS-CoV-2 test results among symptomatic patients. Whether these medications increase the risk for mild or asymptomatic disease or are beneficial in COVID-19 treatment remains uncertain.”
In an accompanying editorial, William G. Kussmaul III, MD, Drexel University, Philadelphia, said that initial fears that these drugs may be harmful for patients with COVID-19 now seem to have been unfounded.
“We now have reasonable reassurance that drugs that alter the renin-angiotensin system do not pose substantial threats as either COVID-19 risk factors or severity multipliers,” he wrote.
A version of this article originally appeared on Medscape.com.








