User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Treating Family: Ethicist Discusses Whether It’s Appropriate
This transcript has been edited for clarity.
There’s a very interesting story in the medical press. A few years ago, a plastic surgeon named Edmond Cabbabe was preparing to do a follow-up cosmetic procedure on his wife at Mercy Hospital South, which is a big hospital in the St. Louis, Missouri, area.
He put her on the operating schedule, and he had done that when he had performed the original operation on her. On the day of the surgery, he got a call from the hospital saying the procedure was canceled. They said that the hospital’s policy, maybe a new one, would not allow doctors to operate on family members.
This physician was a past president of the Missouri State Medical Association. I think he was also on the board or president of the American Medical Association (AMA) Foundation. This was a physician not only in a skilled area where he felt confident he could take care of his wife, but also someone who was prominent in medical politics and medical policy.
The AMA forever has had a policy that says don’t treat relatives. This physician basically said, I think that policy is too restrictive, too cautious, and it doesn’t make much sense to continue to say that you can’t treat family and friends.
By implication, he was saying, I know exactly what I’m doing in my field and I know exactly what I’m doing with her procedure. I should have a right to perform it. I think I do a great job and I’d be best for her.
If you look at medical boards, every once in a while in some state, someone is brought up on a charge of doing different things with family members and saying that they’re going to get censured. They don’t usually lose their license, but they get a reprimand or get told that is just not ethical to do.
I think, in the long run, the policy about not treating your family and friends makes sense. The problem is, as is well known from the social sciences and psychology, people get biased when they deal with those they care about, love, and hold close to them.
It’s hard for the doctor to be objective when dealing with people that they really like or love. It’s also difficult for patients because they may not want to bring up something or they are uncomfortable talking with a doctor who’s a family member or close friend. They may not want to complain. They may be a little bit embarrassed about things. It just adds an emotional edge, I think, that’s difficult.
All that said, do I know doctors who regularly prescribe, say, an ointment for something that’s itchy or some kind of a pill when allergy season breaks out? I do. Do I think they’re acting in a horribly unethical manner? I don’t.
You need some judgment here. There are absolutely minor things where objectivity, fear, and anxiety are not in play. You’re going to be able to prescribe the routine thing for the routine itch without worrying too much about whether it’s a stranger, a friend, or your daughter.
What sorts of things am I really talking about when I say that minor variability ought to be allowed? It’s one thing when someone has poison ivy and they’re going to need some kind of standard medicine to treat it. A very different area that’s much more dangerous, and one I would avoid, is in the mental health field, and for that matter, the pain field.
It’s tempting to say: “Oh, my relative is just having a bad time. I’ll give her a little bit of antidepressant medicine,” or “They seem to be having pain after an operation or something, and I’m going to give them a little bit of pain meds just to get them through.”
Those areas are flying red flags. It’s easy to abuse and easy for someone to become a user and manipulate a friend or a doctor who’s a relative into getting things that another doctor wouldn’t be giving. I think that’s the space where you’ve got to exercise extreme caution.
Time and again, when those people get called up in front of the boards for treating relatives, it’s in those spaces of mental health, anxiety, and pain control. Again, when you know that there’s a likelihood of abuse, I think that’s the place where the line has to hold. Don’t treat the relative. Don’t treat the friend.
At the end of the day, I wouldn’t change the AMA policy. I think we should keep it in place and morally try to discourage doctors from caring for those they’re close to or they have emotional ties to.
At the same time, as with all ethical situations, there has to be a little bit of wiggle room for those super-minor cases where it just makes sense to say: “You don’t have to go find somebody else to do this. I can prescribe this ointment or this minor thing for you. No one’s objectivity is going to be soured, and you’re not going to feel in any way at risk because I’m going to prescribe this for you.”
Common sense ought to prevail. The default position is don’t do it; however, maybe with a tiny bit of space for what’s minor, what’s routine, and what really does just save people some inconvenience, there I might just give a little.
Dr. Caplan, Director, Division of Medical Ethics, New York University Langone Medical Center, New York City, has disclosed relationships with Johnson & Johnson’s Panel for Compassionate Drug Use and Medscape.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
There’s a very interesting story in the medical press. A few years ago, a plastic surgeon named Edmond Cabbabe was preparing to do a follow-up cosmetic procedure on his wife at Mercy Hospital South, which is a big hospital in the St. Louis, Missouri, area.
He put her on the operating schedule, and he had done that when he had performed the original operation on her. On the day of the surgery, he got a call from the hospital saying the procedure was canceled. They said that the hospital’s policy, maybe a new one, would not allow doctors to operate on family members.
This physician was a past president of the Missouri State Medical Association. I think he was also on the board or president of the American Medical Association (AMA) Foundation. This was a physician not only in a skilled area where he felt confident he could take care of his wife, but also someone who was prominent in medical politics and medical policy.
The AMA forever has had a policy that says don’t treat relatives. This physician basically said, I think that policy is too restrictive, too cautious, and it doesn’t make much sense to continue to say that you can’t treat family and friends.
By implication, he was saying, I know exactly what I’m doing in my field and I know exactly what I’m doing with her procedure. I should have a right to perform it. I think I do a great job and I’d be best for her.
If you look at medical boards, every once in a while in some state, someone is brought up on a charge of doing different things with family members and saying that they’re going to get censured. They don’t usually lose their license, but they get a reprimand or get told that is just not ethical to do.
I think, in the long run, the policy about not treating your family and friends makes sense. The problem is, as is well known from the social sciences and psychology, people get biased when they deal with those they care about, love, and hold close to them.
It’s hard for the doctor to be objective when dealing with people that they really like or love. It’s also difficult for patients because they may not want to bring up something or they are uncomfortable talking with a doctor who’s a family member or close friend. They may not want to complain. They may be a little bit embarrassed about things. It just adds an emotional edge, I think, that’s difficult.
All that said, do I know doctors who regularly prescribe, say, an ointment for something that’s itchy or some kind of a pill when allergy season breaks out? I do. Do I think they’re acting in a horribly unethical manner? I don’t.
You need some judgment here. There are absolutely minor things where objectivity, fear, and anxiety are not in play. You’re going to be able to prescribe the routine thing for the routine itch without worrying too much about whether it’s a stranger, a friend, or your daughter.
What sorts of things am I really talking about when I say that minor variability ought to be allowed? It’s one thing when someone has poison ivy and they’re going to need some kind of standard medicine to treat it. A very different area that’s much more dangerous, and one I would avoid, is in the mental health field, and for that matter, the pain field.
It’s tempting to say: “Oh, my relative is just having a bad time. I’ll give her a little bit of antidepressant medicine,” or “They seem to be having pain after an operation or something, and I’m going to give them a little bit of pain meds just to get them through.”
Those areas are flying red flags. It’s easy to abuse and easy for someone to become a user and manipulate a friend or a doctor who’s a relative into getting things that another doctor wouldn’t be giving. I think that’s the space where you’ve got to exercise extreme caution.
Time and again, when those people get called up in front of the boards for treating relatives, it’s in those spaces of mental health, anxiety, and pain control. Again, when you know that there’s a likelihood of abuse, I think that’s the place where the line has to hold. Don’t treat the relative. Don’t treat the friend.
At the end of the day, I wouldn’t change the AMA policy. I think we should keep it in place and morally try to discourage doctors from caring for those they’re close to or they have emotional ties to.
At the same time, as with all ethical situations, there has to be a little bit of wiggle room for those super-minor cases where it just makes sense to say: “You don’t have to go find somebody else to do this. I can prescribe this ointment or this minor thing for you. No one’s objectivity is going to be soured, and you’re not going to feel in any way at risk because I’m going to prescribe this for you.”
Common sense ought to prevail. The default position is don’t do it; however, maybe with a tiny bit of space for what’s minor, what’s routine, and what really does just save people some inconvenience, there I might just give a little.
Dr. Caplan, Director, Division of Medical Ethics, New York University Langone Medical Center, New York City, has disclosed relationships with Johnson & Johnson’s Panel for Compassionate Drug Use and Medscape.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
There’s a very interesting story in the medical press. A few years ago, a plastic surgeon named Edmond Cabbabe was preparing to do a follow-up cosmetic procedure on his wife at Mercy Hospital South, which is a big hospital in the St. Louis, Missouri, area.
He put her on the operating schedule, and he had done that when he had performed the original operation on her. On the day of the surgery, he got a call from the hospital saying the procedure was canceled. They said that the hospital’s policy, maybe a new one, would not allow doctors to operate on family members.
This physician was a past president of the Missouri State Medical Association. I think he was also on the board or president of the American Medical Association (AMA) Foundation. This was a physician not only in a skilled area where he felt confident he could take care of his wife, but also someone who was prominent in medical politics and medical policy.
The AMA forever has had a policy that says don’t treat relatives. This physician basically said, I think that policy is too restrictive, too cautious, and it doesn’t make much sense to continue to say that you can’t treat family and friends.
By implication, he was saying, I know exactly what I’m doing in my field and I know exactly what I’m doing with her procedure. I should have a right to perform it. I think I do a great job and I’d be best for her.
If you look at medical boards, every once in a while in some state, someone is brought up on a charge of doing different things with family members and saying that they’re going to get censured. They don’t usually lose their license, but they get a reprimand or get told that is just not ethical to do.
I think, in the long run, the policy about not treating your family and friends makes sense. The problem is, as is well known from the social sciences and psychology, people get biased when they deal with those they care about, love, and hold close to them.
It’s hard for the doctor to be objective when dealing with people that they really like or love. It’s also difficult for patients because they may not want to bring up something or they are uncomfortable talking with a doctor who’s a family member or close friend. They may not want to complain. They may be a little bit embarrassed about things. It just adds an emotional edge, I think, that’s difficult.
All that said, do I know doctors who regularly prescribe, say, an ointment for something that’s itchy or some kind of a pill when allergy season breaks out? I do. Do I think they’re acting in a horribly unethical manner? I don’t.
You need some judgment here. There are absolutely minor things where objectivity, fear, and anxiety are not in play. You’re going to be able to prescribe the routine thing for the routine itch without worrying too much about whether it’s a stranger, a friend, or your daughter.
What sorts of things am I really talking about when I say that minor variability ought to be allowed? It’s one thing when someone has poison ivy and they’re going to need some kind of standard medicine to treat it. A very different area that’s much more dangerous, and one I would avoid, is in the mental health field, and for that matter, the pain field.
It’s tempting to say: “Oh, my relative is just having a bad time. I’ll give her a little bit of antidepressant medicine,” or “They seem to be having pain after an operation or something, and I’m going to give them a little bit of pain meds just to get them through.”
Those areas are flying red flags. It’s easy to abuse and easy for someone to become a user and manipulate a friend or a doctor who’s a relative into getting things that another doctor wouldn’t be giving. I think that’s the space where you’ve got to exercise extreme caution.
Time and again, when those people get called up in front of the boards for treating relatives, it’s in those spaces of mental health, anxiety, and pain control. Again, when you know that there’s a likelihood of abuse, I think that’s the place where the line has to hold. Don’t treat the relative. Don’t treat the friend.
At the end of the day, I wouldn’t change the AMA policy. I think we should keep it in place and morally try to discourage doctors from caring for those they’re close to or they have emotional ties to.
At the same time, as with all ethical situations, there has to be a little bit of wiggle room for those super-minor cases where it just makes sense to say: “You don’t have to go find somebody else to do this. I can prescribe this ointment or this minor thing for you. No one’s objectivity is going to be soured, and you’re not going to feel in any way at risk because I’m going to prescribe this for you.”
Common sense ought to prevail. The default position is don’t do it; however, maybe with a tiny bit of space for what’s minor, what’s routine, and what really does just save people some inconvenience, there I might just give a little.
Dr. Caplan, Director, Division of Medical Ethics, New York University Langone Medical Center, New York City, has disclosed relationships with Johnson & Johnson’s Panel for Compassionate Drug Use and Medscape.
A version of this article first appeared on Medscape.com.
Are You Using the Correct Medication or a Look-Alike?
Five years have passed since the member states of the World Health Organization (WHO) gathered at the 72nd World Health Assembly and decided that September 17 should be recognized as World Patient Safety Day, acknowledging it as a global health priority.
WHO data indicate the following findings related to medical safety:
- One in 10 patients is harmed while receiving healthcare, and 3 million die as a result.
- More than half of these incidents could be prevented.
- Indirect costs could amount to several billion US dollars annually.
Given the magnitude of preventable harm related to medication use, in 2017, the WHO launched the third Global Patient Safety Challenge: Medication Without Harm with the goal of reducing serious and preventable harm related to medication by 50%. In addition, considering the volume of medication packages prescribed in 2023 by physicians in Spain’s National Health System, it is necessary to understand the most common types of medication errors to provide an effective and efficient response.
According to Spain’s Institute for Safe Medication Practices (ISMP), the 10 types of medication errors detected in 2020 with the most serious consequences were the following:
- Errors due to omission or delay in medication.
- Administration of medication to the wrong patient.
- Errors related to allergies or known adverse effects of medications.
- Dosing errors in pediatric patients.
- Errors due to similarities in the labeling or packaging of marketed medications.
- Errors associated with the lack of use of smart infusion pumps.
- Errors due to accidental administration of neuromuscular blocking agents.
- Incorrect intravenous administration of oral liquid medications.
- Errors in medication reconciliation upon hospital admission and discharge.
- Errors due to patient misunderstandings regarding medication use.
I would like to focus on the fifth item, errors due to similarities in the labeling or packaging of marketed medications.
Medications with similar names or with similar labeling or packaging are known as “look alike–sound alike” medications. They are estimated to account for between 6.2% and 14.7% of all medication errors. Confusion can arise due to spelling and phonetic similarities.
As shown in bulletin no. 50 of the ISMP, difficulties in distinguishing different medications or different presentations of the same medication due to similar packaging and labeling have frequently been associated with reported incidents.
Most cases involve either medications marketed by the same laboratory with a design based on brand image or different medications marketed by different laboratories in screen-printed ampoules used in the same settings.
In 2020, the ISMP published 11 new cases of labeling or packaging that may promote errors on its website. It reported 49 incidents to the Spanish Agency for Medicines and Medical Devices.
Shortages caused by the COVID-19 pandemic have further contributed to these incidents, as healthcare facilities sometimes had to change the medications they usually acquired and purchase whatever was available, without being able to select products that would not be confused with existing medications in the facility.
The ISMP recommends the following general practices for healthcare institutions, professionals, and patients to prevent these errors:
- Develop short lists of easily confused medication names and distribute them among all healthcare professionals.
- Prioritize medication names by active ingredient instead of brand name.
- For similar names, highlight the differences in capital letters, eg, DOBUTamine, DOPamine.
- For similar active ingredients, use brand names.
- Avoid placing similar medications near each other.
- Prescribe all medications electronically to minimize the risk of selecting the wrong medication.
- Make manual prescriptions legible, with clearly written dosages and pharmaceutical forms.
- Encourage patients to actively participate in their treatment and consult a clinician if they have any questions about the medications they are receiving.
- Raise awareness among patients, family members, and caregivers about the issues caused by medication name confusion and inform them about how to avoid these errors.
- Instruct patients to focus on and always use the active ingredient name as an identifying element for the medications they are taking.
- Review treatments with patients to ensure they know the medications they are taking.
Julia María Ruiz Redondo is the regional nursing advisor inspector of Spanish Society of General and Family Physicians of Castilla-La Mancha (SEMG-CLM), coordinator of the National Working Group on Public Health in the SEMG, and director of the international public health master’s degree at TECH Technological University. This article is the result of an editorial collaboration between the SEMG and Univadis, which you can access here.
This story was translated from Univadis Spain, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Five years have passed since the member states of the World Health Organization (WHO) gathered at the 72nd World Health Assembly and decided that September 17 should be recognized as World Patient Safety Day, acknowledging it as a global health priority.
WHO data indicate the following findings related to medical safety:
- One in 10 patients is harmed while receiving healthcare, and 3 million die as a result.
- More than half of these incidents could be prevented.
- Indirect costs could amount to several billion US dollars annually.
Given the magnitude of preventable harm related to medication use, in 2017, the WHO launched the third Global Patient Safety Challenge: Medication Without Harm with the goal of reducing serious and preventable harm related to medication by 50%. In addition, considering the volume of medication packages prescribed in 2023 by physicians in Spain’s National Health System, it is necessary to understand the most common types of medication errors to provide an effective and efficient response.
According to Spain’s Institute for Safe Medication Practices (ISMP), the 10 types of medication errors detected in 2020 with the most serious consequences were the following:
- Errors due to omission or delay in medication.
- Administration of medication to the wrong patient.
- Errors related to allergies or known adverse effects of medications.
- Dosing errors in pediatric patients.
- Errors due to similarities in the labeling or packaging of marketed medications.
- Errors associated with the lack of use of smart infusion pumps.
- Errors due to accidental administration of neuromuscular blocking agents.
- Incorrect intravenous administration of oral liquid medications.
- Errors in medication reconciliation upon hospital admission and discharge.
- Errors due to patient misunderstandings regarding medication use.
I would like to focus on the fifth item, errors due to similarities in the labeling or packaging of marketed medications.
Medications with similar names or with similar labeling or packaging are known as “look alike–sound alike” medications. They are estimated to account for between 6.2% and 14.7% of all medication errors. Confusion can arise due to spelling and phonetic similarities.
As shown in bulletin no. 50 of the ISMP, difficulties in distinguishing different medications or different presentations of the same medication due to similar packaging and labeling have frequently been associated with reported incidents.
Most cases involve either medications marketed by the same laboratory with a design based on brand image or different medications marketed by different laboratories in screen-printed ampoules used in the same settings.
In 2020, the ISMP published 11 new cases of labeling or packaging that may promote errors on its website. It reported 49 incidents to the Spanish Agency for Medicines and Medical Devices.
Shortages caused by the COVID-19 pandemic have further contributed to these incidents, as healthcare facilities sometimes had to change the medications they usually acquired and purchase whatever was available, without being able to select products that would not be confused with existing medications in the facility.
The ISMP recommends the following general practices for healthcare institutions, professionals, and patients to prevent these errors:
- Develop short lists of easily confused medication names and distribute them among all healthcare professionals.
- Prioritize medication names by active ingredient instead of brand name.
- For similar names, highlight the differences in capital letters, eg, DOBUTamine, DOPamine.
- For similar active ingredients, use brand names.
- Avoid placing similar medications near each other.
- Prescribe all medications electronically to minimize the risk of selecting the wrong medication.
- Make manual prescriptions legible, with clearly written dosages and pharmaceutical forms.
- Encourage patients to actively participate in their treatment and consult a clinician if they have any questions about the medications they are receiving.
- Raise awareness among patients, family members, and caregivers about the issues caused by medication name confusion and inform them about how to avoid these errors.
- Instruct patients to focus on and always use the active ingredient name as an identifying element for the medications they are taking.
- Review treatments with patients to ensure they know the medications they are taking.
Julia María Ruiz Redondo is the regional nursing advisor inspector of Spanish Society of General and Family Physicians of Castilla-La Mancha (SEMG-CLM), coordinator of the National Working Group on Public Health in the SEMG, and director of the international public health master’s degree at TECH Technological University. This article is the result of an editorial collaboration between the SEMG and Univadis, which you can access here.
This story was translated from Univadis Spain, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Five years have passed since the member states of the World Health Organization (WHO) gathered at the 72nd World Health Assembly and decided that September 17 should be recognized as World Patient Safety Day, acknowledging it as a global health priority.
WHO data indicate the following findings related to medical safety:
- One in 10 patients is harmed while receiving healthcare, and 3 million die as a result.
- More than half of these incidents could be prevented.
- Indirect costs could amount to several billion US dollars annually.
Given the magnitude of preventable harm related to medication use, in 2017, the WHO launched the third Global Patient Safety Challenge: Medication Without Harm with the goal of reducing serious and preventable harm related to medication by 50%. In addition, considering the volume of medication packages prescribed in 2023 by physicians in Spain’s National Health System, it is necessary to understand the most common types of medication errors to provide an effective and efficient response.
According to Spain’s Institute for Safe Medication Practices (ISMP), the 10 types of medication errors detected in 2020 with the most serious consequences were the following:
- Errors due to omission or delay in medication.
- Administration of medication to the wrong patient.
- Errors related to allergies or known adverse effects of medications.
- Dosing errors in pediatric patients.
- Errors due to similarities in the labeling or packaging of marketed medications.
- Errors associated with the lack of use of smart infusion pumps.
- Errors due to accidental administration of neuromuscular blocking agents.
- Incorrect intravenous administration of oral liquid medications.
- Errors in medication reconciliation upon hospital admission and discharge.
- Errors due to patient misunderstandings regarding medication use.
I would like to focus on the fifth item, errors due to similarities in the labeling or packaging of marketed medications.
Medications with similar names or with similar labeling or packaging are known as “look alike–sound alike” medications. They are estimated to account for between 6.2% and 14.7% of all medication errors. Confusion can arise due to spelling and phonetic similarities.
As shown in bulletin no. 50 of the ISMP, difficulties in distinguishing different medications or different presentations of the same medication due to similar packaging and labeling have frequently been associated with reported incidents.
Most cases involve either medications marketed by the same laboratory with a design based on brand image or different medications marketed by different laboratories in screen-printed ampoules used in the same settings.
In 2020, the ISMP published 11 new cases of labeling or packaging that may promote errors on its website. It reported 49 incidents to the Spanish Agency for Medicines and Medical Devices.
Shortages caused by the COVID-19 pandemic have further contributed to these incidents, as healthcare facilities sometimes had to change the medications they usually acquired and purchase whatever was available, without being able to select products that would not be confused with existing medications in the facility.
The ISMP recommends the following general practices for healthcare institutions, professionals, and patients to prevent these errors:
- Develop short lists of easily confused medication names and distribute them among all healthcare professionals.
- Prioritize medication names by active ingredient instead of brand name.
- For similar names, highlight the differences in capital letters, eg, DOBUTamine, DOPamine.
- For similar active ingredients, use brand names.
- Avoid placing similar medications near each other.
- Prescribe all medications electronically to minimize the risk of selecting the wrong medication.
- Make manual prescriptions legible, with clearly written dosages and pharmaceutical forms.
- Encourage patients to actively participate in their treatment and consult a clinician if they have any questions about the medications they are receiving.
- Raise awareness among patients, family members, and caregivers about the issues caused by medication name confusion and inform them about how to avoid these errors.
- Instruct patients to focus on and always use the active ingredient name as an identifying element for the medications they are taking.
- Review treatments with patients to ensure they know the medications they are taking.
Julia María Ruiz Redondo is the regional nursing advisor inspector of Spanish Society of General and Family Physicians of Castilla-La Mancha (SEMG-CLM), coordinator of the National Working Group on Public Health in the SEMG, and director of the international public health master’s degree at TECH Technological University. This article is the result of an editorial collaboration between the SEMG and Univadis, which you can access here.
This story was translated from Univadis Spain, which is part of the Medscape professional network, using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Locally Acquired Dengue Case Confirmed in California
A case of locally acquired dengue fever has been confirmed in a resident of Baldwin Park, California, according to a press release from the Los Angeles County Department of Public Health.
“Dengue is the most common insect-borne viral infection in the world, with a wide geographic spread; we know that we have mosquitoes capable of carrying and transmitting the virus in the United States already, and Los Angeles county is a major epicenter for international travel and trade,” James Lawler, MD, associate director for International Programs and Innovation at the Global Center for Health Security and professor in the Infectious Diseases Division at the University of Nebraska Medical Center, Omaha, Nebraska, said in an interview.
Although the patient had no known history of travel to a dengue-endemic area, the potential risk for widespread transmission of the virus in the Los Angeles County area remains low, and no additional suspected cases of locally acquired dengue have been identified, according to the release. However, the recent cases highlight the need for vigilance on the part of the public to reduce transmission of mosquito-borne infections, the public health department noted.
Most cases of dengue occur in people who have traveled to areas where the disease is more common, mainly tropical and subtropical areas, according to the press release. However, the types of mosquitoes that spread dengue exist in parts of the United States, so locally acquired infections can occur.
The Centers for Disease Control and Prevention (CDC) issued an official health advisory in June 2024 about an increased risk for dengue infections in the United States. According to the advisory, 745 cases of dengue were identified in US travelers to endemic areas between January 1, 2024, and June 24, 2024.
The CDC advises clinicians to maintain a high level of suspicion for dengue among individuals with fever and recent travel to areas with frequent dengue transmission, but also to consider locally acquired disease in areas of mosquito vectors.
In clinical practice, dengue may be difficult to differentiate from other febrile systemic infections, Dr. Lawler noted. “Joint pain, low back pain, and headache (often retro-orbital) are common and can be severe, and a rash often appears several days into illness,” he noted.
Do not delay treatment in suspected cases while waiting for test results, the CDC emphasized in the advisory. Food and Drug Administration–approved tests for dengue include RT-PCR and IgM antibody tests or NS1 and IgM antibody tests.
“Severe dengue can be life-threatening and progress to a hemorrhagic fever-like syndrome, and patients with severe dengue should be cared for on a high-acuity or intensive care setting, with close monitoring of labs and fluid status,” Dr. Lawler told this news organization.
The World Health Organization has published guidelines for the management of dengue, which Dr. Lawler strongly recommends to clinicians in the rare event that they are facing a severe case. The treatment for dengue is supportive care, according to the CDC; a vaccine that was deemed safe and effective is no longer being manufactured because of low demand.
Most symptoms last for 2-7 days, and most patients recover within a week, but approximately 1 in 20 may develop severe disease, according to the Los Angeles County Department of Public Health.
Approximately one quarter of dengue infections are symptomatic, and clinicians should know the signs of progression to severe disease, which include abdominal pain or tenderness, persistent vomiting, clinical fluid accumulation, mucosal bleeding, lethargy or restlessness, and liver enlargement, according to the CDC.
Local Dengue Not Unexpected
“Sadly, I am not surprised at another locally acquired case of dengue fever in the United States,” said Dr. Lawler. “We also have seen a trend of more historically tropical, insect-borne diseases popping up with locally acquired cases in the United States,” he noted.
Dr. Lawler suggested that “the erosion of state and local public health” is a major contributor to the increase in dengue cases. For more than 100 years, activities of state and local public health officials had significantly curtailed mosquito-borne diseases through aggressive control programs, “but we seem to be losing ground over the last several years,” he said.
“Locally acquired dengue cases are still rare in the United States,” he added. “However, people can protect themselves against dengue and more common arthropod-borne infections by taking precautions to cover up and wear insect repellent while outdoors.”
In addition, the Los Angeles County Department of Public Health emphasized in its press release that local residents reduce their risk for contact with mosquitoes by removing areas of standing water on their property and ensuring well-fitted screens on doors and windows.
Dr. Lawler had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
A case of locally acquired dengue fever has been confirmed in a resident of Baldwin Park, California, according to a press release from the Los Angeles County Department of Public Health.
“Dengue is the most common insect-borne viral infection in the world, with a wide geographic spread; we know that we have mosquitoes capable of carrying and transmitting the virus in the United States already, and Los Angeles county is a major epicenter for international travel and trade,” James Lawler, MD, associate director for International Programs and Innovation at the Global Center for Health Security and professor in the Infectious Diseases Division at the University of Nebraska Medical Center, Omaha, Nebraska, said in an interview.
Although the patient had no known history of travel to a dengue-endemic area, the potential risk for widespread transmission of the virus in the Los Angeles County area remains low, and no additional suspected cases of locally acquired dengue have been identified, according to the release. However, the recent cases highlight the need for vigilance on the part of the public to reduce transmission of mosquito-borne infections, the public health department noted.
Most cases of dengue occur in people who have traveled to areas where the disease is more common, mainly tropical and subtropical areas, according to the press release. However, the types of mosquitoes that spread dengue exist in parts of the United States, so locally acquired infections can occur.
The Centers for Disease Control and Prevention (CDC) issued an official health advisory in June 2024 about an increased risk for dengue infections in the United States. According to the advisory, 745 cases of dengue were identified in US travelers to endemic areas between January 1, 2024, and June 24, 2024.
The CDC advises clinicians to maintain a high level of suspicion for dengue among individuals with fever and recent travel to areas with frequent dengue transmission, but also to consider locally acquired disease in areas of mosquito vectors.
In clinical practice, dengue may be difficult to differentiate from other febrile systemic infections, Dr. Lawler noted. “Joint pain, low back pain, and headache (often retro-orbital) are common and can be severe, and a rash often appears several days into illness,” he noted.
Do not delay treatment in suspected cases while waiting for test results, the CDC emphasized in the advisory. Food and Drug Administration–approved tests for dengue include RT-PCR and IgM antibody tests or NS1 and IgM antibody tests.
“Severe dengue can be life-threatening and progress to a hemorrhagic fever-like syndrome, and patients with severe dengue should be cared for on a high-acuity or intensive care setting, with close monitoring of labs and fluid status,” Dr. Lawler told this news organization.
The World Health Organization has published guidelines for the management of dengue, which Dr. Lawler strongly recommends to clinicians in the rare event that they are facing a severe case. The treatment for dengue is supportive care, according to the CDC; a vaccine that was deemed safe and effective is no longer being manufactured because of low demand.
Most symptoms last for 2-7 days, and most patients recover within a week, but approximately 1 in 20 may develop severe disease, according to the Los Angeles County Department of Public Health.
Approximately one quarter of dengue infections are symptomatic, and clinicians should know the signs of progression to severe disease, which include abdominal pain or tenderness, persistent vomiting, clinical fluid accumulation, mucosal bleeding, lethargy or restlessness, and liver enlargement, according to the CDC.
Local Dengue Not Unexpected
“Sadly, I am not surprised at another locally acquired case of dengue fever in the United States,” said Dr. Lawler. “We also have seen a trend of more historically tropical, insect-borne diseases popping up with locally acquired cases in the United States,” he noted.
Dr. Lawler suggested that “the erosion of state and local public health” is a major contributor to the increase in dengue cases. For more than 100 years, activities of state and local public health officials had significantly curtailed mosquito-borne diseases through aggressive control programs, “but we seem to be losing ground over the last several years,” he said.
“Locally acquired dengue cases are still rare in the United States,” he added. “However, people can protect themselves against dengue and more common arthropod-borne infections by taking precautions to cover up and wear insect repellent while outdoors.”
In addition, the Los Angeles County Department of Public Health emphasized in its press release that local residents reduce their risk for contact with mosquitoes by removing areas of standing water on their property and ensuring well-fitted screens on doors and windows.
Dr. Lawler had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
A case of locally acquired dengue fever has been confirmed in a resident of Baldwin Park, California, according to a press release from the Los Angeles County Department of Public Health.
“Dengue is the most common insect-borne viral infection in the world, with a wide geographic spread; we know that we have mosquitoes capable of carrying and transmitting the virus in the United States already, and Los Angeles county is a major epicenter for international travel and trade,” James Lawler, MD, associate director for International Programs and Innovation at the Global Center for Health Security and professor in the Infectious Diseases Division at the University of Nebraska Medical Center, Omaha, Nebraska, said in an interview.
Although the patient had no known history of travel to a dengue-endemic area, the potential risk for widespread transmission of the virus in the Los Angeles County area remains low, and no additional suspected cases of locally acquired dengue have been identified, according to the release. However, the recent cases highlight the need for vigilance on the part of the public to reduce transmission of mosquito-borne infections, the public health department noted.
Most cases of dengue occur in people who have traveled to areas where the disease is more common, mainly tropical and subtropical areas, according to the press release. However, the types of mosquitoes that spread dengue exist in parts of the United States, so locally acquired infections can occur.
The Centers for Disease Control and Prevention (CDC) issued an official health advisory in June 2024 about an increased risk for dengue infections in the United States. According to the advisory, 745 cases of dengue were identified in US travelers to endemic areas between January 1, 2024, and June 24, 2024.
The CDC advises clinicians to maintain a high level of suspicion for dengue among individuals with fever and recent travel to areas with frequent dengue transmission, but also to consider locally acquired disease in areas of mosquito vectors.
In clinical practice, dengue may be difficult to differentiate from other febrile systemic infections, Dr. Lawler noted. “Joint pain, low back pain, and headache (often retro-orbital) are common and can be severe, and a rash often appears several days into illness,” he noted.
Do not delay treatment in suspected cases while waiting for test results, the CDC emphasized in the advisory. Food and Drug Administration–approved tests for dengue include RT-PCR and IgM antibody tests or NS1 and IgM antibody tests.
“Severe dengue can be life-threatening and progress to a hemorrhagic fever-like syndrome, and patients with severe dengue should be cared for on a high-acuity or intensive care setting, with close monitoring of labs and fluid status,” Dr. Lawler told this news organization.
The World Health Organization has published guidelines for the management of dengue, which Dr. Lawler strongly recommends to clinicians in the rare event that they are facing a severe case. The treatment for dengue is supportive care, according to the CDC; a vaccine that was deemed safe and effective is no longer being manufactured because of low demand.
Most symptoms last for 2-7 days, and most patients recover within a week, but approximately 1 in 20 may develop severe disease, according to the Los Angeles County Department of Public Health.
Approximately one quarter of dengue infections are symptomatic, and clinicians should know the signs of progression to severe disease, which include abdominal pain or tenderness, persistent vomiting, clinical fluid accumulation, mucosal bleeding, lethargy or restlessness, and liver enlargement, according to the CDC.
Local Dengue Not Unexpected
“Sadly, I am not surprised at another locally acquired case of dengue fever in the United States,” said Dr. Lawler. “We also have seen a trend of more historically tropical, insect-borne diseases popping up with locally acquired cases in the United States,” he noted.
Dr. Lawler suggested that “the erosion of state and local public health” is a major contributor to the increase in dengue cases. For more than 100 years, activities of state and local public health officials had significantly curtailed mosquito-borne diseases through aggressive control programs, “but we seem to be losing ground over the last several years,” he said.
“Locally acquired dengue cases are still rare in the United States,” he added. “However, people can protect themselves against dengue and more common arthropod-borne infections by taking precautions to cover up and wear insect repellent while outdoors.”
In addition, the Los Angeles County Department of Public Health emphasized in its press release that local residents reduce their risk for contact with mosquitoes by removing areas of standing water on their property and ensuring well-fitted screens on doors and windows.
Dr. Lawler had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
‘Reform School’ for Pharmacy Benefit Managers: How Might Legislation Help Patients?
The term “reform school” is a bit outdated. It used to refer to institutions where young offenders were sent instead of prison. Some argue that pharmacy benefit managers (PBMs) should bypass reform school and go straight to prison. “PBM reform” has become a ubiquitous term, encompassing any legislative or regulatory efforts aimed at curbing PBMs’ bad behavior. When discussing PBM reform, it’s crucial to understand the various segments of the healthcare system affected by PBMs. This complexity often makes it challenging to determine what these reform packages would actually achieve and who they would benefit.
Pharmacists have long been vocal critics of PBMs, and while their issues are extremely important, it is essential to remember that the ultimate victims of PBM misconduct, in terms of access to care, are patients. At some point, we will all be patients, making this issue universally relevant. It has been quite challenging to follow federal legislation on this topic as these packages attempt to address a number of bad behaviors by PBMs affecting a variety of victims. This discussion will examine those reforms that would directly improve patient’s access to available and affordable medications.
Policy Categories of PBM Reform
There are five policy categories of PBM reform legislation overall, including three that have the greatest potential to directly address patient needs. The first is patient access to medications (utilization management, copay assistance, prior authorization, etc.), followed by delinking drug list prices from PBM income and pass-through of price concessions from the manufacturer. The remaining two categories involve transparency and pharmacy-facing reform, both of which are very important. However, this discussion will revolve around the first three categories. It should be noted that many of the legislation packages addressing the categories of patient access, delinking, and pass-through also include transparency issues, particularly as they relate to pharmacy-facing issues.
Patient Access to Medications — Step Therapy Legislation
One of the major obstacles to patient access to medications is the use of PBM utilization management tools such as step therapy (“fail first”), prior authorizations, nonmedical switching, and formulary exclusions. These tools dictate when patients can obtain necessary medications and for how long patients who are stable on their current treatments can remain on them.
While many states have enacted step therapy reforms to prevent stable patients from being whip-sawed between medications that maximize PBM profits (often labeled as “savings”), these state protections apply only to state-regulated health plans. These include fully insured health plans and those offered through the Affordable Care Act’s Health Insurance Marketplace. It also includes state employees, state corrections, and, in some cases, state labor unions. State legislation does not extend to patients covered by employer self-insured health plans, called ERISA plans for the federal law that governs employee benefit plans, the Employee Retirement Income Security Act. These ERISA plans include nearly 35 million people nationwide.
This is where the Safe Step Act (S.652/H.R.2630) becomes crucial, as it allows employees to request exceptions to harmful fail-first protocols. The bill has gained significant momentum, having been reported out of the Senate HELP Committee and discussed in House markups. The Safe Step Act would mandate that an exception to a step therapy protocol must be granted if:
- The required treatment has been ineffective
- The treatment is expected to be ineffective, and delaying effective treatment would lead to irreversible consequences
- The treatment will cause or is likely to cause an adverse reaction
- The treatment is expected to prevent the individual from performing daily activities or occupational responsibilities
- The individual is stable on their current prescription drugs
- There are other circumstances as determined by the Employee Benefits Security Administration
This legislation is vital for ensuring that patients have timely access to the medications they need without unnecessary delays or disruptions.
Patient Access to Medications — Prior Authorizations
Another significant issue affecting patient access to medications is prior authorizations (PAs). According to an American Medical Association survey, nearly one in four physicians (24%) report that a PA has led to a serious adverse event for a patient in their care. In rheumatology, PAs often result in delays in care (even for those initially approved) and a significant increase in steroid usage. In particular, PAs in Medicare Advantage (MA) plans are harmful to Medicare beneficiaries.
The Improving Seniors’ Timely Access to Care Act (H.R.8702 / S.4532) aims to reform PAs used in MA plans, making the process more efficient and transparent to improve access to care for seniors. Unfortunately, it does not cover Part D drugs and may only cover Part B drugs depending on the MA plan’s benefit package. Here are the key provisions of the act:
- Electronic PA: Implementing real-time decisions for routinely approved items and services.
- Transparency: Requiring annual publication of PA information, such as the percentage of requests approved and the average response time.
- Quality and Timeliness Standards: The Centers for Medicare & Medicaid Services (CMS) will set standards for the quality and timeliness of PA determinations.
- Streamlining Approvals: Simplifying the approval process and reducing the time allowed for health plans to consider PA requests.
This bill passed the House in September 2022 but stalled in the Senate because of an unfavorable Congressional Budget Office score. CMS has since finalized portions of this bill via regulation, zeroing out the CBO score and increasing the chances of its passage.
Delinking Drug Prices from PBM Income and Pass-Through of Price Concessions
Affordability is a crucial aspect of accessibility, especially when it comes to medications. Over the years, we’ve learned that PBMs often favor placing the highest list price drugs on formularies because the rebates and various fees they receive from manufacturers are based on a percentage of the list price. In other words, the higher the medication’s price, the more money the PBM makes.
This practice is evident in both commercial and government formularies, where brand-name drugs are often preferred, while lower-priced generics are either excluded or placed on higher tiers. As a result, while major PBMs benefit from these rebates and fees, patients continue to pay their cost share based on the list price of the medication.
To improve the affordability of medications, a key aspect of PBM reform should be to disincentivize PBMs from selecting higher-priced medications and/or require the pass-through of manufacturer price concessions to patients.
Several major PBM reform bills are currently being considered that address either the delinking of price concessions from the list price of the drug or some form of pass-through of these concessions. These reforms are essential to ensure that patients can access affordable medications without being burdened by inflated costs.
The legislation includes the Pharmacy Benefit Manager Reform Act (S.1339); the Modernizing & Ensuring PBM Accountability Act (S.2973); the Better Mental Health Care, Lower Cost Drugs, and Extenders Act (S.3430); the Protecting Patients Against PBM Abuses Act (H.R. 2880); the DRUG Act (S.2474 / H.R.6283); and the Share the Savings with Seniors Act (S.2474 / H.R.5376).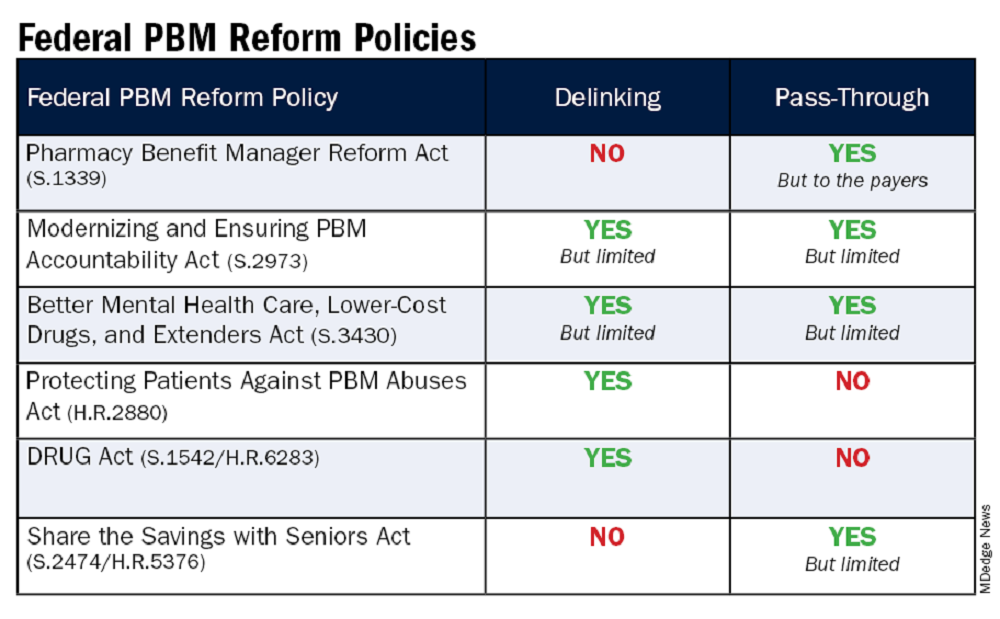
As with all legislation, there are limitations and compromises in each of these. However, these bills are a good first step in addressing PBM remuneration (rebates and fees) based on the list price of the drug and/or passing through to the patient the benefit of manufacturer price concessions. By focusing on key areas like utilization management, delinking drug prices from PBM income, and allowing patients to directly benefit from manufacturer price concessions, we can work toward a more equitable and efficient healthcare system. Reigning in PBM bad behavior is a challenge, but the potential benefits for patient care and access make it a crucial fight worth pursuing.
Please help in efforts to improve patients’ access to available and affordable medications by contacting your representatives in Congress to impart to them the importance of passing legislation. The CSRO’s legislative map tool can help to inform you of the latest information on these and other bills and assist you in engaging with your representatives on them.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of Advocacy and Government Affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. She has no relevant conflicts of interest to disclose. You can reach her at [email protected].
The term “reform school” is a bit outdated. It used to refer to institutions where young offenders were sent instead of prison. Some argue that pharmacy benefit managers (PBMs) should bypass reform school and go straight to prison. “PBM reform” has become a ubiquitous term, encompassing any legislative or regulatory efforts aimed at curbing PBMs’ bad behavior. When discussing PBM reform, it’s crucial to understand the various segments of the healthcare system affected by PBMs. This complexity often makes it challenging to determine what these reform packages would actually achieve and who they would benefit.
Pharmacists have long been vocal critics of PBMs, and while their issues are extremely important, it is essential to remember that the ultimate victims of PBM misconduct, in terms of access to care, are patients. At some point, we will all be patients, making this issue universally relevant. It has been quite challenging to follow federal legislation on this topic as these packages attempt to address a number of bad behaviors by PBMs affecting a variety of victims. This discussion will examine those reforms that would directly improve patient’s access to available and affordable medications.
Policy Categories of PBM Reform
There are five policy categories of PBM reform legislation overall, including three that have the greatest potential to directly address patient needs. The first is patient access to medications (utilization management, copay assistance, prior authorization, etc.), followed by delinking drug list prices from PBM income and pass-through of price concessions from the manufacturer. The remaining two categories involve transparency and pharmacy-facing reform, both of which are very important. However, this discussion will revolve around the first three categories. It should be noted that many of the legislation packages addressing the categories of patient access, delinking, and pass-through also include transparency issues, particularly as they relate to pharmacy-facing issues.
Patient Access to Medications — Step Therapy Legislation
One of the major obstacles to patient access to medications is the use of PBM utilization management tools such as step therapy (“fail first”), prior authorizations, nonmedical switching, and formulary exclusions. These tools dictate when patients can obtain necessary medications and for how long patients who are stable on their current treatments can remain on them.
While many states have enacted step therapy reforms to prevent stable patients from being whip-sawed between medications that maximize PBM profits (often labeled as “savings”), these state protections apply only to state-regulated health plans. These include fully insured health plans and those offered through the Affordable Care Act’s Health Insurance Marketplace. It also includes state employees, state corrections, and, in some cases, state labor unions. State legislation does not extend to patients covered by employer self-insured health plans, called ERISA plans for the federal law that governs employee benefit plans, the Employee Retirement Income Security Act. These ERISA plans include nearly 35 million people nationwide.
This is where the Safe Step Act (S.652/H.R.2630) becomes crucial, as it allows employees to request exceptions to harmful fail-first protocols. The bill has gained significant momentum, having been reported out of the Senate HELP Committee and discussed in House markups. The Safe Step Act would mandate that an exception to a step therapy protocol must be granted if:
- The required treatment has been ineffective
- The treatment is expected to be ineffective, and delaying effective treatment would lead to irreversible consequences
- The treatment will cause or is likely to cause an adverse reaction
- The treatment is expected to prevent the individual from performing daily activities or occupational responsibilities
- The individual is stable on their current prescription drugs
- There are other circumstances as determined by the Employee Benefits Security Administration
This legislation is vital for ensuring that patients have timely access to the medications they need without unnecessary delays or disruptions.
Patient Access to Medications — Prior Authorizations
Another significant issue affecting patient access to medications is prior authorizations (PAs). According to an American Medical Association survey, nearly one in four physicians (24%) report that a PA has led to a serious adverse event for a patient in their care. In rheumatology, PAs often result in delays in care (even for those initially approved) and a significant increase in steroid usage. In particular, PAs in Medicare Advantage (MA) plans are harmful to Medicare beneficiaries.
The Improving Seniors’ Timely Access to Care Act (H.R.8702 / S.4532) aims to reform PAs used in MA plans, making the process more efficient and transparent to improve access to care for seniors. Unfortunately, it does not cover Part D drugs and may only cover Part B drugs depending on the MA plan’s benefit package. Here are the key provisions of the act:
- Electronic PA: Implementing real-time decisions for routinely approved items and services.
- Transparency: Requiring annual publication of PA information, such as the percentage of requests approved and the average response time.
- Quality and Timeliness Standards: The Centers for Medicare & Medicaid Services (CMS) will set standards for the quality and timeliness of PA determinations.
- Streamlining Approvals: Simplifying the approval process and reducing the time allowed for health plans to consider PA requests.
This bill passed the House in September 2022 but stalled in the Senate because of an unfavorable Congressional Budget Office score. CMS has since finalized portions of this bill via regulation, zeroing out the CBO score and increasing the chances of its passage.
Delinking Drug Prices from PBM Income and Pass-Through of Price Concessions
Affordability is a crucial aspect of accessibility, especially when it comes to medications. Over the years, we’ve learned that PBMs often favor placing the highest list price drugs on formularies because the rebates and various fees they receive from manufacturers are based on a percentage of the list price. In other words, the higher the medication’s price, the more money the PBM makes.
This practice is evident in both commercial and government formularies, where brand-name drugs are often preferred, while lower-priced generics are either excluded or placed on higher tiers. As a result, while major PBMs benefit from these rebates and fees, patients continue to pay their cost share based on the list price of the medication.
To improve the affordability of medications, a key aspect of PBM reform should be to disincentivize PBMs from selecting higher-priced medications and/or require the pass-through of manufacturer price concessions to patients.
Several major PBM reform bills are currently being considered that address either the delinking of price concessions from the list price of the drug or some form of pass-through of these concessions. These reforms are essential to ensure that patients can access affordable medications without being burdened by inflated costs.
The legislation includes the Pharmacy Benefit Manager Reform Act (S.1339); the Modernizing & Ensuring PBM Accountability Act (S.2973); the Better Mental Health Care, Lower Cost Drugs, and Extenders Act (S.3430); the Protecting Patients Against PBM Abuses Act (H.R. 2880); the DRUG Act (S.2474 / H.R.6283); and the Share the Savings with Seniors Act (S.2474 / H.R.5376).
As with all legislation, there are limitations and compromises in each of these. However, these bills are a good first step in addressing PBM remuneration (rebates and fees) based on the list price of the drug and/or passing through to the patient the benefit of manufacturer price concessions. By focusing on key areas like utilization management, delinking drug prices from PBM income, and allowing patients to directly benefit from manufacturer price concessions, we can work toward a more equitable and efficient healthcare system. Reigning in PBM bad behavior is a challenge, but the potential benefits for patient care and access make it a crucial fight worth pursuing.
Please help in efforts to improve patients’ access to available and affordable medications by contacting your representatives in Congress to impart to them the importance of passing legislation. The CSRO’s legislative map tool can help to inform you of the latest information on these and other bills and assist you in engaging with your representatives on them.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of Advocacy and Government Affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. She has no relevant conflicts of interest to disclose. You can reach her at [email protected].
The term “reform school” is a bit outdated. It used to refer to institutions where young offenders were sent instead of prison. Some argue that pharmacy benefit managers (PBMs) should bypass reform school and go straight to prison. “PBM reform” has become a ubiquitous term, encompassing any legislative or regulatory efforts aimed at curbing PBMs’ bad behavior. When discussing PBM reform, it’s crucial to understand the various segments of the healthcare system affected by PBMs. This complexity often makes it challenging to determine what these reform packages would actually achieve and who they would benefit.
Pharmacists have long been vocal critics of PBMs, and while their issues are extremely important, it is essential to remember that the ultimate victims of PBM misconduct, in terms of access to care, are patients. At some point, we will all be patients, making this issue universally relevant. It has been quite challenging to follow federal legislation on this topic as these packages attempt to address a number of bad behaviors by PBMs affecting a variety of victims. This discussion will examine those reforms that would directly improve patient’s access to available and affordable medications.
Policy Categories of PBM Reform
There are five policy categories of PBM reform legislation overall, including three that have the greatest potential to directly address patient needs. The first is patient access to medications (utilization management, copay assistance, prior authorization, etc.), followed by delinking drug list prices from PBM income and pass-through of price concessions from the manufacturer. The remaining two categories involve transparency and pharmacy-facing reform, both of which are very important. However, this discussion will revolve around the first three categories. It should be noted that many of the legislation packages addressing the categories of patient access, delinking, and pass-through also include transparency issues, particularly as they relate to pharmacy-facing issues.
Patient Access to Medications — Step Therapy Legislation
One of the major obstacles to patient access to medications is the use of PBM utilization management tools such as step therapy (“fail first”), prior authorizations, nonmedical switching, and formulary exclusions. These tools dictate when patients can obtain necessary medications and for how long patients who are stable on their current treatments can remain on them.
While many states have enacted step therapy reforms to prevent stable patients from being whip-sawed between medications that maximize PBM profits (often labeled as “savings”), these state protections apply only to state-regulated health plans. These include fully insured health plans and those offered through the Affordable Care Act’s Health Insurance Marketplace. It also includes state employees, state corrections, and, in some cases, state labor unions. State legislation does not extend to patients covered by employer self-insured health plans, called ERISA plans for the federal law that governs employee benefit plans, the Employee Retirement Income Security Act. These ERISA plans include nearly 35 million people nationwide.
This is where the Safe Step Act (S.652/H.R.2630) becomes crucial, as it allows employees to request exceptions to harmful fail-first protocols. The bill has gained significant momentum, having been reported out of the Senate HELP Committee and discussed in House markups. The Safe Step Act would mandate that an exception to a step therapy protocol must be granted if:
- The required treatment has been ineffective
- The treatment is expected to be ineffective, and delaying effective treatment would lead to irreversible consequences
- The treatment will cause or is likely to cause an adverse reaction
- The treatment is expected to prevent the individual from performing daily activities or occupational responsibilities
- The individual is stable on their current prescription drugs
- There are other circumstances as determined by the Employee Benefits Security Administration
This legislation is vital for ensuring that patients have timely access to the medications they need without unnecessary delays or disruptions.
Patient Access to Medications — Prior Authorizations
Another significant issue affecting patient access to medications is prior authorizations (PAs). According to an American Medical Association survey, nearly one in four physicians (24%) report that a PA has led to a serious adverse event for a patient in their care. In rheumatology, PAs often result in delays in care (even for those initially approved) and a significant increase in steroid usage. In particular, PAs in Medicare Advantage (MA) plans are harmful to Medicare beneficiaries.
The Improving Seniors’ Timely Access to Care Act (H.R.8702 / S.4532) aims to reform PAs used in MA plans, making the process more efficient and transparent to improve access to care for seniors. Unfortunately, it does not cover Part D drugs and may only cover Part B drugs depending on the MA plan’s benefit package. Here are the key provisions of the act:
- Electronic PA: Implementing real-time decisions for routinely approved items and services.
- Transparency: Requiring annual publication of PA information, such as the percentage of requests approved and the average response time.
- Quality and Timeliness Standards: The Centers for Medicare & Medicaid Services (CMS) will set standards for the quality and timeliness of PA determinations.
- Streamlining Approvals: Simplifying the approval process and reducing the time allowed for health plans to consider PA requests.
This bill passed the House in September 2022 but stalled in the Senate because of an unfavorable Congressional Budget Office score. CMS has since finalized portions of this bill via regulation, zeroing out the CBO score and increasing the chances of its passage.
Delinking Drug Prices from PBM Income and Pass-Through of Price Concessions
Affordability is a crucial aspect of accessibility, especially when it comes to medications. Over the years, we’ve learned that PBMs often favor placing the highest list price drugs on formularies because the rebates and various fees they receive from manufacturers are based on a percentage of the list price. In other words, the higher the medication’s price, the more money the PBM makes.
This practice is evident in both commercial and government formularies, where brand-name drugs are often preferred, while lower-priced generics are either excluded or placed on higher tiers. As a result, while major PBMs benefit from these rebates and fees, patients continue to pay their cost share based on the list price of the medication.
To improve the affordability of medications, a key aspect of PBM reform should be to disincentivize PBMs from selecting higher-priced medications and/or require the pass-through of manufacturer price concessions to patients.
Several major PBM reform bills are currently being considered that address either the delinking of price concessions from the list price of the drug or some form of pass-through of these concessions. These reforms are essential to ensure that patients can access affordable medications without being burdened by inflated costs.
The legislation includes the Pharmacy Benefit Manager Reform Act (S.1339); the Modernizing & Ensuring PBM Accountability Act (S.2973); the Better Mental Health Care, Lower Cost Drugs, and Extenders Act (S.3430); the Protecting Patients Against PBM Abuses Act (H.R. 2880); the DRUG Act (S.2474 / H.R.6283); and the Share the Savings with Seniors Act (S.2474 / H.R.5376).
As with all legislation, there are limitations and compromises in each of these. However, these bills are a good first step in addressing PBM remuneration (rebates and fees) based on the list price of the drug and/or passing through to the patient the benefit of manufacturer price concessions. By focusing on key areas like utilization management, delinking drug prices from PBM income, and allowing patients to directly benefit from manufacturer price concessions, we can work toward a more equitable and efficient healthcare system. Reigning in PBM bad behavior is a challenge, but the potential benefits for patient care and access make it a crucial fight worth pursuing.
Please help in efforts to improve patients’ access to available and affordable medications by contacting your representatives in Congress to impart to them the importance of passing legislation. The CSRO’s legislative map tool can help to inform you of the latest information on these and other bills and assist you in engaging with your representatives on them.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of Advocacy and Government Affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. She has no relevant conflicts of interest to disclose. You can reach her at [email protected].
Oropouche Virus
The pediatrician’s first patient of the day was a 15-year-old boy complaining of fever, chills, and profound arthralgias. His exam, including a careful assessment of his joints, yielded no clues, and the pediatrician was ready to diagnose this as a routine viral illness. An additional bit of history provided by the patient’s mother prompted the pediatrician to pause and reconsider.
“A week ago, we returned from a visit to Cuba,” the mother reported. “Could this be Oropouche virus infection?”
Oropouche virus disease is an arboviral disease caused by the Oropouche virus (OROV). It is transmitted to humans through midge or mosquito bites. Although largely unknown to most United States clinicians until recently, this vector-borne virus is not new. The first human Oropouche virus infection was identified in Trinidad and Tobago in 1955 and since then, there have been intermittent outbreaks in the Amazon region. In recent months, though, the epidemiology of Oropouche virus infections has changed. Infections are being identified in new geographic areas, including Cuba. According to the Pan American Health Organization, 506 cases of Oropouche virus infection have been identified in Cuba since May 27, 2024.
Two deaths from Oropouche virus infection have been reported in previously healthy people. Evolving data suggests adverse outcomes associated with vertical transmission during pregnancy. One fetal death and child with congenital anomalies have been reported in Brazil. Additional fetal deaths, miscarriages, and congenital anomalies are under investigation.
Travel-associated cases have been reported in the United States. As of September 10, 2024, 52 Oropouche virus disease cases had been reported from five states in the United States. The Centers for Disease Control and Prevention confirmed that the first 31 of these cases were travelers returning from Cuba. The CDC issued a health advisory on August 16, 2024: Increased Oropouche Virus Activity and Associated Risk to Travelers.
The pediatrician quickly reviewed the signs and symptoms of Oropouche virus infection. Disease typically presents as an abrupt onset of fever, severe headache, chills, myalgia, and arthralgia 3 to 10 days after the bite of infected mosquito. Some patients develop a maculopapular rash that starts on the trunk and spreads to the extremities. Meningitis and encephalitis develop in less than 1 in 20 people. The symptoms of Oropouche virus infection overlap with those of other arboviruses such as dengue, chikungunya, and Zika viruses. The disease can also mimic malaria or rickettsial infection. Approximately 60% of people with Oropouche virus infection experience a recurrence of symptoms within days to weeks of the initial resolution of symptoms.
Testing for Oropouche virus infection is available through the CDC’s Arbovirus Diagnostic Laboratory. In people who are acutely ill, reverse transcription-polymerase chain reaction testing can be used to identify the virus in serum and cerebrospinal fluid. Serologic testing is also available for people who have been symptomatic for at least 6 days.
The pediatrician contacted his local health department to discuss the possibility of Oropouche virus infection. After reviewing the case definition, public health authorities recommended laboratory testing for Oropouche virus, dengue, and Zika virus.
Back in the exam room, the pediatrician provided anticipatory guidance to the patient and his mother. There are no antiviral medications to treat Oropouche virus infection, so the pediatrician recommended supportive care, including acetaminophen for fever and pain. He also advised avoiding aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs) until dengue could be ruled out to reduce the risk of bleeding. After confirming that no one else in the home was sick with similar symptoms, he counseled about prevention strategies.
To date, transmission of Oropouche virus in the United States has not been documented, but vectors potentially capable of transmitting the virus are present in some areas of the United States. When people who are infected with Oropouche are bitten, they can spread the virus through their blood to biting midges or mosquitoes. The insects can then spread the virus to other people. To reduce to potential for local transmission, people who are sick with suspected Oropouche virus infection are advised to avoid biting-midge and mosquito bites for the first week of their illness. Any person who has recently traveled to an area where Oropouche virus transmission is occurring should also avoid insect bites for 3 weeks after returning home to account for the potential incubation period of the virus. This includes wearing an EPA-registered insect repellent.
A suspect case is a patient who has been in an area with documented or suspected OROV circulation* within 2 weeks of initial symptom onset (as patients may experience recurrent symptoms) and the following:
- Abrupt onset of reported fever, headache, and one or more of the following: myalgia, arthralgia, photophobia, retro-orbital/eye pain, or signs and symptoms of neuroinvasive disease (eg, stiff neck, altered mental status, seizures, limb weakness, or cerebrospinal fluid pleocytosis).
- Tested negative for other possible diseases, in particular dengue.†
- Absence of a more likely clinical explanation.
*If concern exists for local transmission in a nonendemic area, consider if the patient shared an exposure location with a person with confirmed OROV infection, lives in an area where travel-related cases have been identified, or has known vector exposure (eg, mosquitoes or biting midges).
†If strong suspicion of OROV disease exists based on the patient’s clinical features and history of travel to an area with virus circulation, do not wait on negative testing before sending specimens to CDC.
Adapted from: Centers for Disease Control and Prevention. Response to Oropouche Virus Disease Cases in U.S. States and Territories in the Americas. Available at: https.//www.cdc.gov/oropouche/media/pdfs/2024/09/response-to-oropouche-virus-disease.pdf
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta and Gilead. Email her at [email protected]. (Also [email protected])
The pediatrician’s first patient of the day was a 15-year-old boy complaining of fever, chills, and profound arthralgias. His exam, including a careful assessment of his joints, yielded no clues, and the pediatrician was ready to diagnose this as a routine viral illness. An additional bit of history provided by the patient’s mother prompted the pediatrician to pause and reconsider.
“A week ago, we returned from a visit to Cuba,” the mother reported. “Could this be Oropouche virus infection?”
Oropouche virus disease is an arboviral disease caused by the Oropouche virus (OROV). It is transmitted to humans through midge or mosquito bites. Although largely unknown to most United States clinicians until recently, this vector-borne virus is not new. The first human Oropouche virus infection was identified in Trinidad and Tobago in 1955 and since then, there have been intermittent outbreaks in the Amazon region. In recent months, though, the epidemiology of Oropouche virus infections has changed. Infections are being identified in new geographic areas, including Cuba. According to the Pan American Health Organization, 506 cases of Oropouche virus infection have been identified in Cuba since May 27, 2024.
Two deaths from Oropouche virus infection have been reported in previously healthy people. Evolving data suggests adverse outcomes associated with vertical transmission during pregnancy. One fetal death and child with congenital anomalies have been reported in Brazil. Additional fetal deaths, miscarriages, and congenital anomalies are under investigation.
Travel-associated cases have been reported in the United States. As of September 10, 2024, 52 Oropouche virus disease cases had been reported from five states in the United States. The Centers for Disease Control and Prevention confirmed that the first 31 of these cases were travelers returning from Cuba. The CDC issued a health advisory on August 16, 2024: Increased Oropouche Virus Activity and Associated Risk to Travelers.
The pediatrician quickly reviewed the signs and symptoms of Oropouche virus infection. Disease typically presents as an abrupt onset of fever, severe headache, chills, myalgia, and arthralgia 3 to 10 days after the bite of infected mosquito. Some patients develop a maculopapular rash that starts on the trunk and spreads to the extremities. Meningitis and encephalitis develop in less than 1 in 20 people. The symptoms of Oropouche virus infection overlap with those of other arboviruses such as dengue, chikungunya, and Zika viruses. The disease can also mimic malaria or rickettsial infection. Approximately 60% of people with Oropouche virus infection experience a recurrence of symptoms within days to weeks of the initial resolution of symptoms.
Testing for Oropouche virus infection is available through the CDC’s Arbovirus Diagnostic Laboratory. In people who are acutely ill, reverse transcription-polymerase chain reaction testing can be used to identify the virus in serum and cerebrospinal fluid. Serologic testing is also available for people who have been symptomatic for at least 6 days.
The pediatrician contacted his local health department to discuss the possibility of Oropouche virus infection. After reviewing the case definition, public health authorities recommended laboratory testing for Oropouche virus, dengue, and Zika virus.
Back in the exam room, the pediatrician provided anticipatory guidance to the patient and his mother. There are no antiviral medications to treat Oropouche virus infection, so the pediatrician recommended supportive care, including acetaminophen for fever and pain. He also advised avoiding aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs) until dengue could be ruled out to reduce the risk of bleeding. After confirming that no one else in the home was sick with similar symptoms, he counseled about prevention strategies.
To date, transmission of Oropouche virus in the United States has not been documented, but vectors potentially capable of transmitting the virus are present in some areas of the United States. When people who are infected with Oropouche are bitten, they can spread the virus through their blood to biting midges or mosquitoes. The insects can then spread the virus to other people. To reduce to potential for local transmission, people who are sick with suspected Oropouche virus infection are advised to avoid biting-midge and mosquito bites for the first week of their illness. Any person who has recently traveled to an area where Oropouche virus transmission is occurring should also avoid insect bites for 3 weeks after returning home to account for the potential incubation period of the virus. This includes wearing an EPA-registered insect repellent.
A suspect case is a patient who has been in an area with documented or suspected OROV circulation* within 2 weeks of initial symptom onset (as patients may experience recurrent symptoms) and the following:
- Abrupt onset of reported fever, headache, and one or more of the following: myalgia, arthralgia, photophobia, retro-orbital/eye pain, or signs and symptoms of neuroinvasive disease (eg, stiff neck, altered mental status, seizures, limb weakness, or cerebrospinal fluid pleocytosis).
- Tested negative for other possible diseases, in particular dengue.†
- Absence of a more likely clinical explanation.
*If concern exists for local transmission in a nonendemic area, consider if the patient shared an exposure location with a person with confirmed OROV infection, lives in an area where travel-related cases have been identified, or has known vector exposure (eg, mosquitoes or biting midges).
†If strong suspicion of OROV disease exists based on the patient’s clinical features and history of travel to an area with virus circulation, do not wait on negative testing before sending specimens to CDC.
Adapted from: Centers for Disease Control and Prevention. Response to Oropouche Virus Disease Cases in U.S. States and Territories in the Americas. Available at: https.//www.cdc.gov/oropouche/media/pdfs/2024/09/response-to-oropouche-virus-disease.pdf
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta and Gilead. Email her at [email protected]. (Also [email protected])
The pediatrician’s first patient of the day was a 15-year-old boy complaining of fever, chills, and profound arthralgias. His exam, including a careful assessment of his joints, yielded no clues, and the pediatrician was ready to diagnose this as a routine viral illness. An additional bit of history provided by the patient’s mother prompted the pediatrician to pause and reconsider.
“A week ago, we returned from a visit to Cuba,” the mother reported. “Could this be Oropouche virus infection?”
Oropouche virus disease is an arboviral disease caused by the Oropouche virus (OROV). It is transmitted to humans through midge or mosquito bites. Although largely unknown to most United States clinicians until recently, this vector-borne virus is not new. The first human Oropouche virus infection was identified in Trinidad and Tobago in 1955 and since then, there have been intermittent outbreaks in the Amazon region. In recent months, though, the epidemiology of Oropouche virus infections has changed. Infections are being identified in new geographic areas, including Cuba. According to the Pan American Health Organization, 506 cases of Oropouche virus infection have been identified in Cuba since May 27, 2024.
Two deaths from Oropouche virus infection have been reported in previously healthy people. Evolving data suggests adverse outcomes associated with vertical transmission during pregnancy. One fetal death and child with congenital anomalies have been reported in Brazil. Additional fetal deaths, miscarriages, and congenital anomalies are under investigation.
Travel-associated cases have been reported in the United States. As of September 10, 2024, 52 Oropouche virus disease cases had been reported from five states in the United States. The Centers for Disease Control and Prevention confirmed that the first 31 of these cases were travelers returning from Cuba. The CDC issued a health advisory on August 16, 2024: Increased Oropouche Virus Activity and Associated Risk to Travelers.
The pediatrician quickly reviewed the signs and symptoms of Oropouche virus infection. Disease typically presents as an abrupt onset of fever, severe headache, chills, myalgia, and arthralgia 3 to 10 days after the bite of infected mosquito. Some patients develop a maculopapular rash that starts on the trunk and spreads to the extremities. Meningitis and encephalitis develop in less than 1 in 20 people. The symptoms of Oropouche virus infection overlap with those of other arboviruses such as dengue, chikungunya, and Zika viruses. The disease can also mimic malaria or rickettsial infection. Approximately 60% of people with Oropouche virus infection experience a recurrence of symptoms within days to weeks of the initial resolution of symptoms.
Testing for Oropouche virus infection is available through the CDC’s Arbovirus Diagnostic Laboratory. In people who are acutely ill, reverse transcription-polymerase chain reaction testing can be used to identify the virus in serum and cerebrospinal fluid. Serologic testing is also available for people who have been symptomatic for at least 6 days.
The pediatrician contacted his local health department to discuss the possibility of Oropouche virus infection. After reviewing the case definition, public health authorities recommended laboratory testing for Oropouche virus, dengue, and Zika virus.
Back in the exam room, the pediatrician provided anticipatory guidance to the patient and his mother. There are no antiviral medications to treat Oropouche virus infection, so the pediatrician recommended supportive care, including acetaminophen for fever and pain. He also advised avoiding aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs) until dengue could be ruled out to reduce the risk of bleeding. After confirming that no one else in the home was sick with similar symptoms, he counseled about prevention strategies.
To date, transmission of Oropouche virus in the United States has not been documented, but vectors potentially capable of transmitting the virus are present in some areas of the United States. When people who are infected with Oropouche are bitten, they can spread the virus through their blood to biting midges or mosquitoes. The insects can then spread the virus to other people. To reduce to potential for local transmission, people who are sick with suspected Oropouche virus infection are advised to avoid biting-midge and mosquito bites for the first week of their illness. Any person who has recently traveled to an area where Oropouche virus transmission is occurring should also avoid insect bites for 3 weeks after returning home to account for the potential incubation period of the virus. This includes wearing an EPA-registered insect repellent.
A suspect case is a patient who has been in an area with documented or suspected OROV circulation* within 2 weeks of initial symptom onset (as patients may experience recurrent symptoms) and the following:
- Abrupt onset of reported fever, headache, and one or more of the following: myalgia, arthralgia, photophobia, retro-orbital/eye pain, or signs and symptoms of neuroinvasive disease (eg, stiff neck, altered mental status, seizures, limb weakness, or cerebrospinal fluid pleocytosis).
- Tested negative for other possible diseases, in particular dengue.†
- Absence of a more likely clinical explanation.
*If concern exists for local transmission in a nonendemic area, consider if the patient shared an exposure location with a person with confirmed OROV infection, lives in an area where travel-related cases have been identified, or has known vector exposure (eg, mosquitoes or biting midges).
†If strong suspicion of OROV disease exists based on the patient’s clinical features and history of travel to an area with virus circulation, do not wait on negative testing before sending specimens to CDC.
Adapted from: Centers for Disease Control and Prevention. Response to Oropouche Virus Disease Cases in U.S. States and Territories in the Americas. Available at: https.//www.cdc.gov/oropouche/media/pdfs/2024/09/response-to-oropouche-virus-disease.pdf
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022-2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta and Gilead. Email her at [email protected]. (Also [email protected])
Could a Virus Reverse Antibiotic Resistance?
Peering through his microscope in 1910, Franco-Canadian microbiologist Félix d’Hérelle noticed some “clear spots” in his bacterial cultures, an anomaly that turned out to be viruses preying on the bacteria. Years later, Mr. d’Hérelle would come to use these viruses, which he called bacteriophages, to treat patients plagued with dysentery after World War I.
But now, with bacteria evolving resistance to more and more antibiotics, phage therapy is drawing a second look from researchers — sometimes with a novel twist. Instead of simply using the phages to kill bacteria directly, the new strategy aims to catch the bacteria in an evolutionary dilemma — one in which they cannot evade phages and antibiotics simultaneously.
This plan, which uses something called “phage steering,” has shown promising results in initial tests, but the scope of its usefulness remains to be proven.
There’s certainly need to find new ways to respond to bacterial infections. More than 70% of hospital-acquired bacterial infections in the United States are resistant to at least one type of antibiotic. And some pathogens, such as Acinetobacter, Pseudomonas, Escherichia coli, and Klebsiella — classified by the World Health Organization as some of the biggest threats to human health — are resistant to multiple antibiotics. In 2019, antibacterial resistance was linked to 4.95 million deaths globally, heightening the call for more effective treatment options.
One of the ways that bacteria can evolve resistance to antibiotics is by using structures in their membranes that are designed to move unwanted molecules out of the cell. By modifying these “efflux pumps” to recognize the antibiotic, bacteria can eliminate the drug before it poisons them.
As it turns out, some phages appear to use these same efflux pumps to invade the bacterial cell. The phage presumably attaches its tail to the outer portion of the pump protein, like a key slipping into a lock, and then injects its genetic material into the cell. This lucky coincidence led Paul Turner, PhD, an evolutionary biologist at Yale University, New Haven, Connecticut, to suggest that treating a patient with phages and antibiotics simultaneously could trap bacteria in a no-win situation: If they evolve to modify their efflux pumps so the phage can’t bind, the pumps will no longer expel antibiotics, and the bacteria will lose their resistance. But if they retain their antibiotic resistance, the phages will kill them, as Dr. Turner and colleagues explained in the 2023 Annual Review of Virology.
The result, in other words, is a two-pronged attack, said Michael Hochberg, PhD, an evolutionary biologist at the French National Centre for Scientific Research who studies how to prevent the evolution of bacterial resistance. “It’s kind of like a crisscross effect.” The same principle can target other bacterial molecules that play a dual role in resistance to viruses and antibiotics.
Turner tested this hypothesis on multidrug-resistant Pseudomonas aeruginosa, which causes dangerous infections, especially in healthcare settings. This bacterium has four efflux pumps involved in antibiotic resistance, and Dr. Turner predicted that if he could find a phage that used one of the pumps as a way into the cell, the bacterium would be forced to slam the door on the phage by mutating the receptor — thereby impeding its ability to pump out antibiotics.
Sampling from the environment, Dr. Turner’s team collected 42 phage strains that infect P aeruginosa. Out of all the phages, one, OMKO1, bound to an efflux pump, making it the perfect candidate for the experiment.
The researchers then cultured antibiotic-resistant P aeruginosa together with OMKO1, hoping this would force the bacterium to modify its efflux pump to resist the phage. They exposed these phage-resistant bacteria, as well as their normal, phage-sensitive counterparts, to four antibiotics the bacteria had been resistant to: tetracycline, erythromycin, ciprofloxacin, and ceftazidime.
As the theory predicted, the bacteria that had evolved resistance to the phage were more sensitive to the antibiotics than those that had not been exposed to the phage. This suggests that the bacteria had, indeed, been forced to lose their antibiotic resistance through their need to fight off the phage.
Other researchers have also shown that phage steering can resensitize bacteria to common antibiotics they’d become resistant to. One study, by an international research team, showed that a phage called Phab24 can be used to restore sensitivity to the antibiotic colistin in Acinetobacter baumannii, which causes life-threatening diseases.
In a second study, researchers at Monash University in Australia sampled infectious bacteria from patients. They found that several phages, including strains known as phi-FG02 and phi-CO01, were already present in some of the samples, and that A baumannii bacteria exposed to the phages had inactivated a gene that helps create the microbe’s important outer layer, or capsule. This layer serves as the entry point for the phages, but it also helps the bacterium to form biofilms that keep out antibiotics — so removing the layer rendered A baumannii susceptible to several antibiotics that it was previously resistant to.
In a third study, researchers from the University of Liverpool discovered that, when a P aeruginosa strain that was resistant to all antibiotics was exposed to phages, the bacterium became sensitive to two antibiotics that were otherwise considered ineffective against P aeruginosa.
Dr. Turner’s team has used phage steering in dozens of cases of personalized therapy in clinical settings, said Benjamin Chan, PhD, a microbiologist at Yale University who works with Dr. Turner. The results, many still unpublished, have been promising so far. Nonrespiratory infections are relatively easy to clear off, and lung infections, which the phage steering approach wouldn’t be expected to eradicate completely, often show some improvement.
“I would say that we have been quite successful in using phage steering to treat difficult-to-manage infections, reducing antimicrobial resistance in many cases,” he said. But he notes that it is sometimes difficult to determine whether phage steering really was responsible for the cures.
Devil in the details
Phage therapy may not work for all antibiotic-resistant bacteria, said molecular biologist Graham Hatfull, PhD, of the University of Pittsburgh in Pennsylvania. That’s because phages are very host specific, and for most phages, no one knows what target they bind to on the bacterial cell surface. For phage steering to work against antibiotic resistance, the phage has to bind to a molecule that’s involved in that resistance — and it’s not clear how often that fortuitous coincidence occurs.
Jason Gill, PhD, who studies bacteriophage biology at Texas A&M University, College Station, said that it is not easy to predict if a phage will induce antibiotic sensitivity. So you always have to hunt for the right virus each time.
Dr. Gill knows from experience how complicated the approach can get. He was part of a team of researchers and doctors who used phages to treat a patient with a multidrug-resistant A baumannii infection. Less than 4 days after the team administered phages intravenously and through the skin, the patient woke up from a coma and became responsive to the previously ineffective antibiotic minocycline — a striking success.
But when Dr. Gill tried a similar experiment in cell cultures, he got a different result. The A baumannii developed resistance to the phages, but they also maintained their resistance to minocycline. “There’s not a complete mechanistic understanding,” said Dr. Gill. “The linkage between phage resistance and antibiotic sensitivity probably varies by bacterial strain, phage and antibiotic.” That means phage steering may not always work.
Dr. Turner, for his part, pointed out another potential problem: That phages could work too well. If phage therapy kills large amounts of bacteria and deposits their remains in the bloodstream quickly, for example, this could trigger septic shock in patients. Scientists do not yet know how to address this problem.
Another concern is that doctors have less precise control over phages than antibiotics. “Phages can mutate, they can adapt, they have a genome,” said Dr. Hochberg. Safety concerns, he notes, are one factor inhibiting the routine use of phage therapy in countries like the United States, restricting it to case-by-case applications such as Dr. Turner and Dr. Chan’s.
Phage therapy may have been too high-tech for the 1940s, and even today, scientists grapple with how to use it. What we need now, said Dr. Turner, are rigorous experiments that will teach us how to make it work.
This article originally appeared in Knowable Magazine on September 09, 2024. Knowable Magazine is an independent journalistic endeavor from Annual Reviews, a nonprofit publisher dedicated to synthesizing and integrating knowledge for the progress of science and the benefit of society. Sign up for Knowable Magazine’s newsletter. A version of this article appeared on Medscape.com.
Peering through his microscope in 1910, Franco-Canadian microbiologist Félix d’Hérelle noticed some “clear spots” in his bacterial cultures, an anomaly that turned out to be viruses preying on the bacteria. Years later, Mr. d’Hérelle would come to use these viruses, which he called bacteriophages, to treat patients plagued with dysentery after World War I.
But now, with bacteria evolving resistance to more and more antibiotics, phage therapy is drawing a second look from researchers — sometimes with a novel twist. Instead of simply using the phages to kill bacteria directly, the new strategy aims to catch the bacteria in an evolutionary dilemma — one in which they cannot evade phages and antibiotics simultaneously.
This plan, which uses something called “phage steering,” has shown promising results in initial tests, but the scope of its usefulness remains to be proven.
There’s certainly need to find new ways to respond to bacterial infections. More than 70% of hospital-acquired bacterial infections in the United States are resistant to at least one type of antibiotic. And some pathogens, such as Acinetobacter, Pseudomonas, Escherichia coli, and Klebsiella — classified by the World Health Organization as some of the biggest threats to human health — are resistant to multiple antibiotics. In 2019, antibacterial resistance was linked to 4.95 million deaths globally, heightening the call for more effective treatment options.
One of the ways that bacteria can evolve resistance to antibiotics is by using structures in their membranes that are designed to move unwanted molecules out of the cell. By modifying these “efflux pumps” to recognize the antibiotic, bacteria can eliminate the drug before it poisons them.
As it turns out, some phages appear to use these same efflux pumps to invade the bacterial cell. The phage presumably attaches its tail to the outer portion of the pump protein, like a key slipping into a lock, and then injects its genetic material into the cell. This lucky coincidence led Paul Turner, PhD, an evolutionary biologist at Yale University, New Haven, Connecticut, to suggest that treating a patient with phages and antibiotics simultaneously could trap bacteria in a no-win situation: If they evolve to modify their efflux pumps so the phage can’t bind, the pumps will no longer expel antibiotics, and the bacteria will lose their resistance. But if they retain their antibiotic resistance, the phages will kill them, as Dr. Turner and colleagues explained in the 2023 Annual Review of Virology.
The result, in other words, is a two-pronged attack, said Michael Hochberg, PhD, an evolutionary biologist at the French National Centre for Scientific Research who studies how to prevent the evolution of bacterial resistance. “It’s kind of like a crisscross effect.” The same principle can target other bacterial molecules that play a dual role in resistance to viruses and antibiotics.
Turner tested this hypothesis on multidrug-resistant Pseudomonas aeruginosa, which causes dangerous infections, especially in healthcare settings. This bacterium has four efflux pumps involved in antibiotic resistance, and Dr. Turner predicted that if he could find a phage that used one of the pumps as a way into the cell, the bacterium would be forced to slam the door on the phage by mutating the receptor — thereby impeding its ability to pump out antibiotics.
Sampling from the environment, Dr. Turner’s team collected 42 phage strains that infect P aeruginosa. Out of all the phages, one, OMKO1, bound to an efflux pump, making it the perfect candidate for the experiment.
The researchers then cultured antibiotic-resistant P aeruginosa together with OMKO1, hoping this would force the bacterium to modify its efflux pump to resist the phage. They exposed these phage-resistant bacteria, as well as their normal, phage-sensitive counterparts, to four antibiotics the bacteria had been resistant to: tetracycline, erythromycin, ciprofloxacin, and ceftazidime.
As the theory predicted, the bacteria that had evolved resistance to the phage were more sensitive to the antibiotics than those that had not been exposed to the phage. This suggests that the bacteria had, indeed, been forced to lose their antibiotic resistance through their need to fight off the phage.
Other researchers have also shown that phage steering can resensitize bacteria to common antibiotics they’d become resistant to. One study, by an international research team, showed that a phage called Phab24 can be used to restore sensitivity to the antibiotic colistin in Acinetobacter baumannii, which causes life-threatening diseases.
In a second study, researchers at Monash University in Australia sampled infectious bacteria from patients. They found that several phages, including strains known as phi-FG02 and phi-CO01, were already present in some of the samples, and that A baumannii bacteria exposed to the phages had inactivated a gene that helps create the microbe’s important outer layer, or capsule. This layer serves as the entry point for the phages, but it also helps the bacterium to form biofilms that keep out antibiotics — so removing the layer rendered A baumannii susceptible to several antibiotics that it was previously resistant to.
In a third study, researchers from the University of Liverpool discovered that, when a P aeruginosa strain that was resistant to all antibiotics was exposed to phages, the bacterium became sensitive to two antibiotics that were otherwise considered ineffective against P aeruginosa.
Dr. Turner’s team has used phage steering in dozens of cases of personalized therapy in clinical settings, said Benjamin Chan, PhD, a microbiologist at Yale University who works with Dr. Turner. The results, many still unpublished, have been promising so far. Nonrespiratory infections are relatively easy to clear off, and lung infections, which the phage steering approach wouldn’t be expected to eradicate completely, often show some improvement.
“I would say that we have been quite successful in using phage steering to treat difficult-to-manage infections, reducing antimicrobial resistance in many cases,” he said. But he notes that it is sometimes difficult to determine whether phage steering really was responsible for the cures.
Devil in the details
Phage therapy may not work for all antibiotic-resistant bacteria, said molecular biologist Graham Hatfull, PhD, of the University of Pittsburgh in Pennsylvania. That’s because phages are very host specific, and for most phages, no one knows what target they bind to on the bacterial cell surface. For phage steering to work against antibiotic resistance, the phage has to bind to a molecule that’s involved in that resistance — and it’s not clear how often that fortuitous coincidence occurs.
Jason Gill, PhD, who studies bacteriophage biology at Texas A&M University, College Station, said that it is not easy to predict if a phage will induce antibiotic sensitivity. So you always have to hunt for the right virus each time.
Dr. Gill knows from experience how complicated the approach can get. He was part of a team of researchers and doctors who used phages to treat a patient with a multidrug-resistant A baumannii infection. Less than 4 days after the team administered phages intravenously and through the skin, the patient woke up from a coma and became responsive to the previously ineffective antibiotic minocycline — a striking success.
But when Dr. Gill tried a similar experiment in cell cultures, he got a different result. The A baumannii developed resistance to the phages, but they also maintained their resistance to minocycline. “There’s not a complete mechanistic understanding,” said Dr. Gill. “The linkage between phage resistance and antibiotic sensitivity probably varies by bacterial strain, phage and antibiotic.” That means phage steering may not always work.
Dr. Turner, for his part, pointed out another potential problem: That phages could work too well. If phage therapy kills large amounts of bacteria and deposits their remains in the bloodstream quickly, for example, this could trigger septic shock in patients. Scientists do not yet know how to address this problem.
Another concern is that doctors have less precise control over phages than antibiotics. “Phages can mutate, they can adapt, they have a genome,” said Dr. Hochberg. Safety concerns, he notes, are one factor inhibiting the routine use of phage therapy in countries like the United States, restricting it to case-by-case applications such as Dr. Turner and Dr. Chan’s.
Phage therapy may have been too high-tech for the 1940s, and even today, scientists grapple with how to use it. What we need now, said Dr. Turner, are rigorous experiments that will teach us how to make it work.
This article originally appeared in Knowable Magazine on September 09, 2024. Knowable Magazine is an independent journalistic endeavor from Annual Reviews, a nonprofit publisher dedicated to synthesizing and integrating knowledge for the progress of science and the benefit of society. Sign up for Knowable Magazine’s newsletter. A version of this article appeared on Medscape.com.
Peering through his microscope in 1910, Franco-Canadian microbiologist Félix d’Hérelle noticed some “clear spots” in his bacterial cultures, an anomaly that turned out to be viruses preying on the bacteria. Years later, Mr. d’Hérelle would come to use these viruses, which he called bacteriophages, to treat patients plagued with dysentery after World War I.
But now, with bacteria evolving resistance to more and more antibiotics, phage therapy is drawing a second look from researchers — sometimes with a novel twist. Instead of simply using the phages to kill bacteria directly, the new strategy aims to catch the bacteria in an evolutionary dilemma — one in which they cannot evade phages and antibiotics simultaneously.
This plan, which uses something called “phage steering,” has shown promising results in initial tests, but the scope of its usefulness remains to be proven.
There’s certainly need to find new ways to respond to bacterial infections. More than 70% of hospital-acquired bacterial infections in the United States are resistant to at least one type of antibiotic. And some pathogens, such as Acinetobacter, Pseudomonas, Escherichia coli, and Klebsiella — classified by the World Health Organization as some of the biggest threats to human health — are resistant to multiple antibiotics. In 2019, antibacterial resistance was linked to 4.95 million deaths globally, heightening the call for more effective treatment options.
One of the ways that bacteria can evolve resistance to antibiotics is by using structures in their membranes that are designed to move unwanted molecules out of the cell. By modifying these “efflux pumps” to recognize the antibiotic, bacteria can eliminate the drug before it poisons them.
As it turns out, some phages appear to use these same efflux pumps to invade the bacterial cell. The phage presumably attaches its tail to the outer portion of the pump protein, like a key slipping into a lock, and then injects its genetic material into the cell. This lucky coincidence led Paul Turner, PhD, an evolutionary biologist at Yale University, New Haven, Connecticut, to suggest that treating a patient with phages and antibiotics simultaneously could trap bacteria in a no-win situation: If they evolve to modify their efflux pumps so the phage can’t bind, the pumps will no longer expel antibiotics, and the bacteria will lose their resistance. But if they retain their antibiotic resistance, the phages will kill them, as Dr. Turner and colleagues explained in the 2023 Annual Review of Virology.
The result, in other words, is a two-pronged attack, said Michael Hochberg, PhD, an evolutionary biologist at the French National Centre for Scientific Research who studies how to prevent the evolution of bacterial resistance. “It’s kind of like a crisscross effect.” The same principle can target other bacterial molecules that play a dual role in resistance to viruses and antibiotics.
Turner tested this hypothesis on multidrug-resistant Pseudomonas aeruginosa, which causes dangerous infections, especially in healthcare settings. This bacterium has four efflux pumps involved in antibiotic resistance, and Dr. Turner predicted that if he could find a phage that used one of the pumps as a way into the cell, the bacterium would be forced to slam the door on the phage by mutating the receptor — thereby impeding its ability to pump out antibiotics.
Sampling from the environment, Dr. Turner’s team collected 42 phage strains that infect P aeruginosa. Out of all the phages, one, OMKO1, bound to an efflux pump, making it the perfect candidate for the experiment.
The researchers then cultured antibiotic-resistant P aeruginosa together with OMKO1, hoping this would force the bacterium to modify its efflux pump to resist the phage. They exposed these phage-resistant bacteria, as well as their normal, phage-sensitive counterparts, to four antibiotics the bacteria had been resistant to: tetracycline, erythromycin, ciprofloxacin, and ceftazidime.
As the theory predicted, the bacteria that had evolved resistance to the phage were more sensitive to the antibiotics than those that had not been exposed to the phage. This suggests that the bacteria had, indeed, been forced to lose their antibiotic resistance through their need to fight off the phage.
Other researchers have also shown that phage steering can resensitize bacteria to common antibiotics they’d become resistant to. One study, by an international research team, showed that a phage called Phab24 can be used to restore sensitivity to the antibiotic colistin in Acinetobacter baumannii, which causes life-threatening diseases.
In a second study, researchers at Monash University in Australia sampled infectious bacteria from patients. They found that several phages, including strains known as phi-FG02 and phi-CO01, were already present in some of the samples, and that A baumannii bacteria exposed to the phages had inactivated a gene that helps create the microbe’s important outer layer, or capsule. This layer serves as the entry point for the phages, but it also helps the bacterium to form biofilms that keep out antibiotics — so removing the layer rendered A baumannii susceptible to several antibiotics that it was previously resistant to.
In a third study, researchers from the University of Liverpool discovered that, when a P aeruginosa strain that was resistant to all antibiotics was exposed to phages, the bacterium became sensitive to two antibiotics that were otherwise considered ineffective against P aeruginosa.
Dr. Turner’s team has used phage steering in dozens of cases of personalized therapy in clinical settings, said Benjamin Chan, PhD, a microbiologist at Yale University who works with Dr. Turner. The results, many still unpublished, have been promising so far. Nonrespiratory infections are relatively easy to clear off, and lung infections, which the phage steering approach wouldn’t be expected to eradicate completely, often show some improvement.
“I would say that we have been quite successful in using phage steering to treat difficult-to-manage infections, reducing antimicrobial resistance in many cases,” he said. But he notes that it is sometimes difficult to determine whether phage steering really was responsible for the cures.
Devil in the details
Phage therapy may not work for all antibiotic-resistant bacteria, said molecular biologist Graham Hatfull, PhD, of the University of Pittsburgh in Pennsylvania. That’s because phages are very host specific, and for most phages, no one knows what target they bind to on the bacterial cell surface. For phage steering to work against antibiotic resistance, the phage has to bind to a molecule that’s involved in that resistance — and it’s not clear how often that fortuitous coincidence occurs.
Jason Gill, PhD, who studies bacteriophage biology at Texas A&M University, College Station, said that it is not easy to predict if a phage will induce antibiotic sensitivity. So you always have to hunt for the right virus each time.
Dr. Gill knows from experience how complicated the approach can get. He was part of a team of researchers and doctors who used phages to treat a patient with a multidrug-resistant A baumannii infection. Less than 4 days after the team administered phages intravenously and through the skin, the patient woke up from a coma and became responsive to the previously ineffective antibiotic minocycline — a striking success.
But when Dr. Gill tried a similar experiment in cell cultures, he got a different result. The A baumannii developed resistance to the phages, but they also maintained their resistance to minocycline. “There’s not a complete mechanistic understanding,” said Dr. Gill. “The linkage between phage resistance and antibiotic sensitivity probably varies by bacterial strain, phage and antibiotic.” That means phage steering may not always work.
Dr. Turner, for his part, pointed out another potential problem: That phages could work too well. If phage therapy kills large amounts of bacteria and deposits their remains in the bloodstream quickly, for example, this could trigger septic shock in patients. Scientists do not yet know how to address this problem.
Another concern is that doctors have less precise control over phages than antibiotics. “Phages can mutate, they can adapt, they have a genome,” said Dr. Hochberg. Safety concerns, he notes, are one factor inhibiting the routine use of phage therapy in countries like the United States, restricting it to case-by-case applications such as Dr. Turner and Dr. Chan’s.
Phage therapy may have been too high-tech for the 1940s, and even today, scientists grapple with how to use it. What we need now, said Dr. Turner, are rigorous experiments that will teach us how to make it work.
This article originally appeared in Knowable Magazine on September 09, 2024. Knowable Magazine is an independent journalistic endeavor from Annual Reviews, a nonprofit publisher dedicated to synthesizing and integrating knowledge for the progress of science and the benefit of society. Sign up for Knowable Magazine’s newsletter. A version of this article appeared on Medscape.com.
UVA Defends Medical School Dean, Hospital CEO After Docs Call for Their Removal
The University of Virginia (UVA) is defending the CEO of its health system and its medical school dean in the wake of a very public call for their removal.
At least 128 members of the University of Virginia faculty who are employed by both the medical school and the UVA Physicians Group wrote to the UVA Board of Visitors and its peer-elected faculty leaders, expressing no confidence in K. Craig Kent, MD, CEO of UVA Health and executive vice president for health affairs, and Melina Kibbe, MD, dean of the medical school and chief health affairs officer.
Dr. Kibbe, a vascular surgeon and researcher, is also the editor in chief of JAMA Surgery.
“We call for the immediate removal of Craig Kent and Melina Kibbe,” wrote the physicians.
The letter alleged that patient safety was compromised because doctors, nurses, and other staff were pressured to abstain from reporting safety concerns and that physicians had been hired “despite concerns regarding integrity and quality.” Those who raised safety concerns faced “explicit and implicit threats and retaliation,” including delays and denials of promotion and tenure, said the letter.
The September 5 letter did not include signatures. The authors said that names were being protected, but that they would share the names with a limited audience.
UVA President Jim Ryan took issue with the notion that the signees were anonymous. He said in his own letter to medical school faculty that some of the accusations were about matters that had already been addressed or that were being worked on. As far as allegations that he was not previously aware of, “we will do our best to investigate,” he said.
The faculty who signed the letter “have besmirched the reputations of not just Melina and Craig,” wrote Mr. Ryan. “They have unfairly — and I trust unwittingly — cast a shadow over the great work of the entire health system and medical school.”
The authors claimed that reports about bullying and harassment of trainees had been “suppressed, minimized, and subsequently altered.”
And they said that spending on leadership was prioritized over addressing clinical and technical staff shortages. Whistleblowers who reported fraud were not protected, and clinicians were pressured to modify patient records to “obfuscate adverse outcomes and boost productivity metrics,” they wrote.
The 128 members of the UVA Physicians Group who signed the letter represent about 10% of the 1400 medical school faculty members.
It is not the first time that Dr. Kent has been given a vote of no confidence. In 2017, when he was the dean of the College of Medicine at the Ohio State University, Dr. Kent was accused in a “no confidence” letter from 25 physicians and faculty of helping to undermine the school’s mission and taking actions that led to resignations and early retirements of many staff, the Columbus Dispatch reported.
William G. Crutchfield Jr., a member of the UVA Health System Board, defended Dr. Kent and Dr. Kibbe in a lengthy statement shared with this news organization. He said that UVA Health’s four hospitals had received “A” ratings for safety, and that the system has a 5.1% turnover rate compared with a national average of 8.3%.
Dr. Kent and Dr. Kibbe have recruited faculty from top academic medical centers, Mr. Crutchfield wrote.
“If our work environment were so toxic, these people would not have joined our faculty,” he wrote.
Mr. Crutchfield credited Dr. Kent and Dr. Kibbe with crafting a new 10-year strategic plan and for hiring a chief strategy officer to lead the plan — a move that replaced “expensive outside consultants.”
Mr. Ryan said in his letter that his inbox “is overflowing with testimonials from some of the 1200-plus faculty who did not sign the letter, who attest that the health system today — under Melina and Craig’s leadership — is in the best shape it has ever been in, and that they have addressed changes that have needed to be made for more than two decades.”
A request to see some of these positive testimonials was not answered by press time.
Mr. Crutchfield, like Mr. Ryan, said that the letter writers were doing more harm than good.
“If a small cabal of people hiding behind anonymity can force outstanding leaders out of UVA, it will make it extremely difficult to recruit outstanding new physicians, nurses, technicians, and administrators,” he wrote.
A version of this article first appeared on Medscape.com.
The University of Virginia (UVA) is defending the CEO of its health system and its medical school dean in the wake of a very public call for their removal.
At least 128 members of the University of Virginia faculty who are employed by both the medical school and the UVA Physicians Group wrote to the UVA Board of Visitors and its peer-elected faculty leaders, expressing no confidence in K. Craig Kent, MD, CEO of UVA Health and executive vice president for health affairs, and Melina Kibbe, MD, dean of the medical school and chief health affairs officer.
Dr. Kibbe, a vascular surgeon and researcher, is also the editor in chief of JAMA Surgery.
“We call for the immediate removal of Craig Kent and Melina Kibbe,” wrote the physicians.
The letter alleged that patient safety was compromised because doctors, nurses, and other staff were pressured to abstain from reporting safety concerns and that physicians had been hired “despite concerns regarding integrity and quality.” Those who raised safety concerns faced “explicit and implicit threats and retaliation,” including delays and denials of promotion and tenure, said the letter.
The September 5 letter did not include signatures. The authors said that names were being protected, but that they would share the names with a limited audience.
UVA President Jim Ryan took issue with the notion that the signees were anonymous. He said in his own letter to medical school faculty that some of the accusations were about matters that had already been addressed or that were being worked on. As far as allegations that he was not previously aware of, “we will do our best to investigate,” he said.
The faculty who signed the letter “have besmirched the reputations of not just Melina and Craig,” wrote Mr. Ryan. “They have unfairly — and I trust unwittingly — cast a shadow over the great work of the entire health system and medical school.”
The authors claimed that reports about bullying and harassment of trainees had been “suppressed, minimized, and subsequently altered.”
And they said that spending on leadership was prioritized over addressing clinical and technical staff shortages. Whistleblowers who reported fraud were not protected, and clinicians were pressured to modify patient records to “obfuscate adverse outcomes and boost productivity metrics,” they wrote.
The 128 members of the UVA Physicians Group who signed the letter represent about 10% of the 1400 medical school faculty members.
It is not the first time that Dr. Kent has been given a vote of no confidence. In 2017, when he was the dean of the College of Medicine at the Ohio State University, Dr. Kent was accused in a “no confidence” letter from 25 physicians and faculty of helping to undermine the school’s mission and taking actions that led to resignations and early retirements of many staff, the Columbus Dispatch reported.
William G. Crutchfield Jr., a member of the UVA Health System Board, defended Dr. Kent and Dr. Kibbe in a lengthy statement shared with this news organization. He said that UVA Health’s four hospitals had received “A” ratings for safety, and that the system has a 5.1% turnover rate compared with a national average of 8.3%.
Dr. Kent and Dr. Kibbe have recruited faculty from top academic medical centers, Mr. Crutchfield wrote.
“If our work environment were so toxic, these people would not have joined our faculty,” he wrote.
Mr. Crutchfield credited Dr. Kent and Dr. Kibbe with crafting a new 10-year strategic plan and for hiring a chief strategy officer to lead the plan — a move that replaced “expensive outside consultants.”
Mr. Ryan said in his letter that his inbox “is overflowing with testimonials from some of the 1200-plus faculty who did not sign the letter, who attest that the health system today — under Melina and Craig’s leadership — is in the best shape it has ever been in, and that they have addressed changes that have needed to be made for more than two decades.”
A request to see some of these positive testimonials was not answered by press time.
Mr. Crutchfield, like Mr. Ryan, said that the letter writers were doing more harm than good.
“If a small cabal of people hiding behind anonymity can force outstanding leaders out of UVA, it will make it extremely difficult to recruit outstanding new physicians, nurses, technicians, and administrators,” he wrote.
A version of this article first appeared on Medscape.com.
The University of Virginia (UVA) is defending the CEO of its health system and its medical school dean in the wake of a very public call for their removal.
At least 128 members of the University of Virginia faculty who are employed by both the medical school and the UVA Physicians Group wrote to the UVA Board of Visitors and its peer-elected faculty leaders, expressing no confidence in K. Craig Kent, MD, CEO of UVA Health and executive vice president for health affairs, and Melina Kibbe, MD, dean of the medical school and chief health affairs officer.
Dr. Kibbe, a vascular surgeon and researcher, is also the editor in chief of JAMA Surgery.
“We call for the immediate removal of Craig Kent and Melina Kibbe,” wrote the physicians.
The letter alleged that patient safety was compromised because doctors, nurses, and other staff were pressured to abstain from reporting safety concerns and that physicians had been hired “despite concerns regarding integrity and quality.” Those who raised safety concerns faced “explicit and implicit threats and retaliation,” including delays and denials of promotion and tenure, said the letter.
The September 5 letter did not include signatures. The authors said that names were being protected, but that they would share the names with a limited audience.
UVA President Jim Ryan took issue with the notion that the signees were anonymous. He said in his own letter to medical school faculty that some of the accusations were about matters that had already been addressed or that were being worked on. As far as allegations that he was not previously aware of, “we will do our best to investigate,” he said.
The faculty who signed the letter “have besmirched the reputations of not just Melina and Craig,” wrote Mr. Ryan. “They have unfairly — and I trust unwittingly — cast a shadow over the great work of the entire health system and medical school.”
The authors claimed that reports about bullying and harassment of trainees had been “suppressed, minimized, and subsequently altered.”
And they said that spending on leadership was prioritized over addressing clinical and technical staff shortages. Whistleblowers who reported fraud were not protected, and clinicians were pressured to modify patient records to “obfuscate adverse outcomes and boost productivity metrics,” they wrote.
The 128 members of the UVA Physicians Group who signed the letter represent about 10% of the 1400 medical school faculty members.
It is not the first time that Dr. Kent has been given a vote of no confidence. In 2017, when he was the dean of the College of Medicine at the Ohio State University, Dr. Kent was accused in a “no confidence” letter from 25 physicians and faculty of helping to undermine the school’s mission and taking actions that led to resignations and early retirements of many staff, the Columbus Dispatch reported.
William G. Crutchfield Jr., a member of the UVA Health System Board, defended Dr. Kent and Dr. Kibbe in a lengthy statement shared with this news organization. He said that UVA Health’s four hospitals had received “A” ratings for safety, and that the system has a 5.1% turnover rate compared with a national average of 8.3%.
Dr. Kent and Dr. Kibbe have recruited faculty from top academic medical centers, Mr. Crutchfield wrote.
“If our work environment were so toxic, these people would not have joined our faculty,” he wrote.
Mr. Crutchfield credited Dr. Kent and Dr. Kibbe with crafting a new 10-year strategic plan and for hiring a chief strategy officer to lead the plan — a move that replaced “expensive outside consultants.”
Mr. Ryan said in his letter that his inbox “is overflowing with testimonials from some of the 1200-plus faculty who did not sign the letter, who attest that the health system today — under Melina and Craig’s leadership — is in the best shape it has ever been in, and that they have addressed changes that have needed to be made for more than two decades.”
A request to see some of these positive testimonials was not answered by press time.
Mr. Crutchfield, like Mr. Ryan, said that the letter writers were doing more harm than good.
“If a small cabal of people hiding behind anonymity can force outstanding leaders out of UVA, it will make it extremely difficult to recruit outstanding new physicians, nurses, technicians, and administrators,” he wrote.
A version of this article first appeared on Medscape.com.
New COVID-19 Vaccines That Target KP.2 Variant Available
New COVID-19 vaccines formulated for better protection against the currently circulating variants have been approved by the US Food and Drug Administration.
The COVID vaccines available this fall have been updated to better match the currently circulating COVID strains, said William Schaffner, MD, professor of medicine in the Division of Infectious Diseases at Vanderbilt University, Nashville, Tennessee, in an interview.
“The Pfizer and Moderna vaccines — both mRNA vaccines — target the KP.2 variant, while the Novavax vaccine targets the JN.1 variant, which is a predecessor to KP.2,” said Dr. Schaffner, who also serves as a spokesperson for the National Foundation for Infectious Diseases. “The Novavax vaccine is a protein adjuvant vaccine made in a more traditional fashion and may appeal to those who remain hesitant about receiving an mRNA vaccine,” he explained. However, , he said.
Who Needs It?
“The CDC’s Advisory Committee on Immunization Practices (ACIP) continues to recommend that everyone in the United States who is age 6 months and older receive the updated COVID vaccine this fall, along with influenza vaccine,” Dr. Schaffner said.
“This was not a surprise because COVID will produce a sizable winter outbreak,” he predicted. Although older people and those who have chronic medical conditions such as heart or lung disease, diabetes, or other immunocompromising conditions suffer the most serious impact of COVID, he said. “The virus can strike anyone, even the young and healthy.” The risk for long COVID persists as well, he pointed out.
The ACIP recommendation is endorsed by the American Academy of Pediatrics and other professional organizations, Dr. Shaffner said.
A frequently asked question is whether the COVID and flu vaccines can be given at the same time, and the answer is yes, according to a statement from the Centers for Disease Control and Prevention (CDC).
“The optimal time to be vaccinated is late September and anytime during October in order to get the benefit of protection through the winter,” Dr. Schaffner said.
As with earlier versions of the COVID-19 vaccine, side effects vary from person to person. Reported side effects of the updated vaccine are similar to those seen with earlier versions and may include injection site pain, redness and swelling, fatigue, headache, muscle pain, chills, nausea, and fever, but most of these are short-lived, according to the CDC.
Clinical Guidance
The CDC’s clinical guidance for COVID-19 vaccination outlines more specific guidance for vaccination based on age, vaccination history, and immunocompromised status and will be updated as needed.
A notable difference in the latest guidance is the recommendation of only one shot for adults aged 65 years and older who are NOT moderately or severely immunocompromised. For those who are moderately or severely immunocompromised, the CDC recommends two to three doses of the same brand of vaccine.
Dr. Schaffner strongly encouraged clinicians to recommend the COVID-19 vaccination for all eligible patients. “COVID is a nasty virus that can cause serious disease in anyone,” and protection from previous vaccination or prior infection has likely waned, he said.
Dr. Schaffner also encouraged healthcare professionals and their families to lead by example. “We should all be vaccinated and let our patients know that we are vaccinated and that we want all our patents to be protected,” he said.
The updated COVID-19 vaccination recommendations have become much simpler for clinicians and patients, with a single messenger RNA (mRNA) vaccine required for anyone older than 5 years, said David J. Cennimo, MD, associate professor of medicine and pediatrics in the Division of Infectious Disease at Rutgers New Jersey Medical School, Newark, New Jersey, in an interview.
“The recommendations are a bit more complex for children under 5 years old receiving their first vaccination; they require two to three doses depending on the brand,” he said. “It is important to review the latest recommendations to plan the doses with the correct interval timing. Considering the doses may be 3-4 weeks apart, start early,” he advised.
One-Time Dosing
Although the updated mRNA vaccine is currently recommended as a one-time dose, Dr. Cennimo said he can envision a scenario later in the season when a second dose is recommended for the elderly and those at high risk for severe illness. Dr. Cennimo said that he strongly agrees with the recommendations that everyone aged 6 months and older receive an updated COVID-19 vaccine. Older age remains the prime risk factor, but anyone can become infected, he said.
Predicting a prime time to get vaccinated is tricky because no one knows when the expected rise in winter cases will occur, said Dr. Cennimo.
“We know from years of flu vaccine data that some number of people who delay the vaccine will never return and will miss protection,” he said. Therefore, delaying vaccination is not recommended. Dr. Cennimo plans to follow his habit of getting vaccinated in early October. “I anticipate the maximal effectiveness of the vaccine will carry me through the winter,” he said.
Data support the safety and effectiveness for both flu and COVID vaccines if they are given together, and some research on earlier versions of COVID vaccines suggested that receiving flu and COVID vaccines together might increase the antibody response against COVID, but similar studies of the updated version have not been done, Dr. Cennimo said.
Clinicians may have to overcome the barrier of COVID fatigue to encourage vaccination, Dr. Cennimo said. Many people say they “want it to be over,” he said, but SARS-CoV-2, established as a viral respiratory infection, shows no signs of disappearing. In addition, new data continue to show higher mortality associated with COVID-19 than with influenza, he said.
“We need to explain to our patients that COVID-19 is still here and is still dangerous. The yearly influenza vaccination campaigns should have established and normalized the idea of an updated vaccine targeted for the season’s predicated strains is expected,” he emphasized. “We now have years of safety data behind these vaccines, and we need to make a strong recommendation for this protection,” he said.
COVID-19 vaccines are covered by private insurance, as well as by Medicare and Medicaid, according to the CDC. Vaccination for uninsured children is covered through the Vaccines for Children Program.
A version of this article first appeared on Medscape.com.
New COVID-19 vaccines formulated for better protection against the currently circulating variants have been approved by the US Food and Drug Administration.
The COVID vaccines available this fall have been updated to better match the currently circulating COVID strains, said William Schaffner, MD, professor of medicine in the Division of Infectious Diseases at Vanderbilt University, Nashville, Tennessee, in an interview.
“The Pfizer and Moderna vaccines — both mRNA vaccines — target the KP.2 variant, while the Novavax vaccine targets the JN.1 variant, which is a predecessor to KP.2,” said Dr. Schaffner, who also serves as a spokesperson for the National Foundation for Infectious Diseases. “The Novavax vaccine is a protein adjuvant vaccine made in a more traditional fashion and may appeal to those who remain hesitant about receiving an mRNA vaccine,” he explained. However, , he said.
Who Needs It?
“The CDC’s Advisory Committee on Immunization Practices (ACIP) continues to recommend that everyone in the United States who is age 6 months and older receive the updated COVID vaccine this fall, along with influenza vaccine,” Dr. Schaffner said.
“This was not a surprise because COVID will produce a sizable winter outbreak,” he predicted. Although older people and those who have chronic medical conditions such as heart or lung disease, diabetes, or other immunocompromising conditions suffer the most serious impact of COVID, he said. “The virus can strike anyone, even the young and healthy.” The risk for long COVID persists as well, he pointed out.
The ACIP recommendation is endorsed by the American Academy of Pediatrics and other professional organizations, Dr. Shaffner said.
A frequently asked question is whether the COVID and flu vaccines can be given at the same time, and the answer is yes, according to a statement from the Centers for Disease Control and Prevention (CDC).
“The optimal time to be vaccinated is late September and anytime during October in order to get the benefit of protection through the winter,” Dr. Schaffner said.
As with earlier versions of the COVID-19 vaccine, side effects vary from person to person. Reported side effects of the updated vaccine are similar to those seen with earlier versions and may include injection site pain, redness and swelling, fatigue, headache, muscle pain, chills, nausea, and fever, but most of these are short-lived, according to the CDC.
Clinical Guidance
The CDC’s clinical guidance for COVID-19 vaccination outlines more specific guidance for vaccination based on age, vaccination history, and immunocompromised status and will be updated as needed.
A notable difference in the latest guidance is the recommendation of only one shot for adults aged 65 years and older who are NOT moderately or severely immunocompromised. For those who are moderately or severely immunocompromised, the CDC recommends two to three doses of the same brand of vaccine.
Dr. Schaffner strongly encouraged clinicians to recommend the COVID-19 vaccination for all eligible patients. “COVID is a nasty virus that can cause serious disease in anyone,” and protection from previous vaccination or prior infection has likely waned, he said.
Dr. Schaffner also encouraged healthcare professionals and their families to lead by example. “We should all be vaccinated and let our patients know that we are vaccinated and that we want all our patents to be protected,” he said.
The updated COVID-19 vaccination recommendations have become much simpler for clinicians and patients, with a single messenger RNA (mRNA) vaccine required for anyone older than 5 years, said David J. Cennimo, MD, associate professor of medicine and pediatrics in the Division of Infectious Disease at Rutgers New Jersey Medical School, Newark, New Jersey, in an interview.
“The recommendations are a bit more complex for children under 5 years old receiving their first vaccination; they require two to three doses depending on the brand,” he said. “It is important to review the latest recommendations to plan the doses with the correct interval timing. Considering the doses may be 3-4 weeks apart, start early,” he advised.
One-Time Dosing
Although the updated mRNA vaccine is currently recommended as a one-time dose, Dr. Cennimo said he can envision a scenario later in the season when a second dose is recommended for the elderly and those at high risk for severe illness. Dr. Cennimo said that he strongly agrees with the recommendations that everyone aged 6 months and older receive an updated COVID-19 vaccine. Older age remains the prime risk factor, but anyone can become infected, he said.
Predicting a prime time to get vaccinated is tricky because no one knows when the expected rise in winter cases will occur, said Dr. Cennimo.
“We know from years of flu vaccine data that some number of people who delay the vaccine will never return and will miss protection,” he said. Therefore, delaying vaccination is not recommended. Dr. Cennimo plans to follow his habit of getting vaccinated in early October. “I anticipate the maximal effectiveness of the vaccine will carry me through the winter,” he said.
Data support the safety and effectiveness for both flu and COVID vaccines if they are given together, and some research on earlier versions of COVID vaccines suggested that receiving flu and COVID vaccines together might increase the antibody response against COVID, but similar studies of the updated version have not been done, Dr. Cennimo said.
Clinicians may have to overcome the barrier of COVID fatigue to encourage vaccination, Dr. Cennimo said. Many people say they “want it to be over,” he said, but SARS-CoV-2, established as a viral respiratory infection, shows no signs of disappearing. In addition, new data continue to show higher mortality associated with COVID-19 than with influenza, he said.
“We need to explain to our patients that COVID-19 is still here and is still dangerous. The yearly influenza vaccination campaigns should have established and normalized the idea of an updated vaccine targeted for the season’s predicated strains is expected,” he emphasized. “We now have years of safety data behind these vaccines, and we need to make a strong recommendation for this protection,” he said.
COVID-19 vaccines are covered by private insurance, as well as by Medicare and Medicaid, according to the CDC. Vaccination for uninsured children is covered through the Vaccines for Children Program.
A version of this article first appeared on Medscape.com.
New COVID-19 vaccines formulated for better protection against the currently circulating variants have been approved by the US Food and Drug Administration.
The COVID vaccines available this fall have been updated to better match the currently circulating COVID strains, said William Schaffner, MD, professor of medicine in the Division of Infectious Diseases at Vanderbilt University, Nashville, Tennessee, in an interview.
“The Pfizer and Moderna vaccines — both mRNA vaccines — target the KP.2 variant, while the Novavax vaccine targets the JN.1 variant, which is a predecessor to KP.2,” said Dr. Schaffner, who also serves as a spokesperson for the National Foundation for Infectious Diseases. “The Novavax vaccine is a protein adjuvant vaccine made in a more traditional fashion and may appeal to those who remain hesitant about receiving an mRNA vaccine,” he explained. However, , he said.
Who Needs It?
“The CDC’s Advisory Committee on Immunization Practices (ACIP) continues to recommend that everyone in the United States who is age 6 months and older receive the updated COVID vaccine this fall, along with influenza vaccine,” Dr. Schaffner said.
“This was not a surprise because COVID will produce a sizable winter outbreak,” he predicted. Although older people and those who have chronic medical conditions such as heart or lung disease, diabetes, or other immunocompromising conditions suffer the most serious impact of COVID, he said. “The virus can strike anyone, even the young and healthy.” The risk for long COVID persists as well, he pointed out.
The ACIP recommendation is endorsed by the American Academy of Pediatrics and other professional organizations, Dr. Shaffner said.
A frequently asked question is whether the COVID and flu vaccines can be given at the same time, and the answer is yes, according to a statement from the Centers for Disease Control and Prevention (CDC).
“The optimal time to be vaccinated is late September and anytime during October in order to get the benefit of protection through the winter,” Dr. Schaffner said.
As with earlier versions of the COVID-19 vaccine, side effects vary from person to person. Reported side effects of the updated vaccine are similar to those seen with earlier versions and may include injection site pain, redness and swelling, fatigue, headache, muscle pain, chills, nausea, and fever, but most of these are short-lived, according to the CDC.
Clinical Guidance
The CDC’s clinical guidance for COVID-19 vaccination outlines more specific guidance for vaccination based on age, vaccination history, and immunocompromised status and will be updated as needed.
A notable difference in the latest guidance is the recommendation of only one shot for adults aged 65 years and older who are NOT moderately or severely immunocompromised. For those who are moderately or severely immunocompromised, the CDC recommends two to three doses of the same brand of vaccine.
Dr. Schaffner strongly encouraged clinicians to recommend the COVID-19 vaccination for all eligible patients. “COVID is a nasty virus that can cause serious disease in anyone,” and protection from previous vaccination or prior infection has likely waned, he said.
Dr. Schaffner also encouraged healthcare professionals and their families to lead by example. “We should all be vaccinated and let our patients know that we are vaccinated and that we want all our patents to be protected,” he said.
The updated COVID-19 vaccination recommendations have become much simpler for clinicians and patients, with a single messenger RNA (mRNA) vaccine required for anyone older than 5 years, said David J. Cennimo, MD, associate professor of medicine and pediatrics in the Division of Infectious Disease at Rutgers New Jersey Medical School, Newark, New Jersey, in an interview.
“The recommendations are a bit more complex for children under 5 years old receiving their first vaccination; they require two to three doses depending on the brand,” he said. “It is important to review the latest recommendations to plan the doses with the correct interval timing. Considering the doses may be 3-4 weeks apart, start early,” he advised.
One-Time Dosing
Although the updated mRNA vaccine is currently recommended as a one-time dose, Dr. Cennimo said he can envision a scenario later in the season when a second dose is recommended for the elderly and those at high risk for severe illness. Dr. Cennimo said that he strongly agrees with the recommendations that everyone aged 6 months and older receive an updated COVID-19 vaccine. Older age remains the prime risk factor, but anyone can become infected, he said.
Predicting a prime time to get vaccinated is tricky because no one knows when the expected rise in winter cases will occur, said Dr. Cennimo.
“We know from years of flu vaccine data that some number of people who delay the vaccine will never return and will miss protection,” he said. Therefore, delaying vaccination is not recommended. Dr. Cennimo plans to follow his habit of getting vaccinated in early October. “I anticipate the maximal effectiveness of the vaccine will carry me through the winter,” he said.
Data support the safety and effectiveness for both flu and COVID vaccines if they are given together, and some research on earlier versions of COVID vaccines suggested that receiving flu and COVID vaccines together might increase the antibody response against COVID, but similar studies of the updated version have not been done, Dr. Cennimo said.
Clinicians may have to overcome the barrier of COVID fatigue to encourage vaccination, Dr. Cennimo said. Many people say they “want it to be over,” he said, but SARS-CoV-2, established as a viral respiratory infection, shows no signs of disappearing. In addition, new data continue to show higher mortality associated with COVID-19 than with influenza, he said.
“We need to explain to our patients that COVID-19 is still here and is still dangerous. The yearly influenza vaccination campaigns should have established and normalized the idea of an updated vaccine targeted for the season’s predicated strains is expected,” he emphasized. “We now have years of safety data behind these vaccines, and we need to make a strong recommendation for this protection,” he said.
COVID-19 vaccines are covered by private insurance, as well as by Medicare and Medicaid, according to the CDC. Vaccination for uninsured children is covered through the Vaccines for Children Program.
A version of this article first appeared on Medscape.com.
Why More Doctors Are Joining Unions
With huge shifts over the past decade in the way doctors are employed — half of all doctors now work for a health system or large medical group — the idea of unionizing is not only being explored but gaining traction within the profession. In fact, 8% of the physician workforce (or 70,000 physicians) belong to a union, according to statistics gathered in 2022.
Exact numbers are hard to come by, and, interestingly, although the American Medical Association (AMA) “ supports the right of physicians to engage in collective bargaining,” the organization doesn’t track union membership among physicians, according to an AMA spokesperson.
Forming a Union
One challenge is that forming a union is not only time-consuming but also difficult, owing to several barriers. For starters, the laws dictating unionization differ by state, and the rules governing unionization vary if a hospital is public or private. If there’s enough momentum from doctors leading unionization efforts, approval from hospital leaders is required before an official election can be requested from the National Labor Relations Board.
That said, for doctors who are in a union — the two most popular are the Union of American Physicians and Dentists and the Doctors Council branch of the Service Employees International Union (SEIU)—the benefits are immense, especially because union members can focus on what matters, such as providing the best patient care possible.
, reported WBUR in Boston.
Belonging Matters
“When you build a relationship with your patients, it’s special, and that connection isn’t replaceable,” said Nicholas VenOsdel, MD, a pediatrician at Allina Health Primary Care in Hastings, Minnesota, and a union member of the Doctors Council. “However, a lot of us have felt like that hasn’t been respected as the climate of healthcare has changed so fast.”
In fact, autonomy over how much time doctors spend with patients is driving a lot of interest in unionization.
“We don’t necessarily have that autonomy now,” said Amber Higgins, MD, an emergency physician and an obstetrician at ChristianaCare, a hospital network in Newark, Delaware, and a member of the Doctors Council. “There are so many other demands, whether it’s billing, patient documentation, or other demands from the employer, and all of that takes time away from patient care.”
Another primary driver of physician unionization is the physician burnout epidemic. Physicians collectively complain that they spend more time on electronic health record documentation and bureaucratic administration. Yet if unions can improve these working conditions, the benefit to physicians and their patients would be a welcome change.
Union members are bullish and believe that having a cohesive voice will make a difference.
“We need to use our collective voices to get back to focusing on patient care instead of staring at a computer screen for 80% of the day,” Dr. Higgins told this news organization. “So much of medicine involves getting to the correct diagnosis, listening to patients, observing them, and building a relationship with them. We need time to build that.”
With corporate consolidation and a profit-driven mandate by healthcare systems, doctors are increasingly frustrated and feel that their voices haven’t been heard enough when it comes to issues like workplace safety, working hours, and benefits, said Stuart Bussey, MD, JD, a family practice physician and president of the Union of American Physicians and Dentists in Sacramento, California.
However, he adds that urging doctors to join together to fight for a better working environment hasn’t been easy.
“Doctors are individualists, and they don’t know how to work in packs like hospital administrators do,” said Dr. Bussey. “They’re hard to organize, but I want them to understand that unless they join hands, sign petitions, and speak as one voice, they’re going to lose out on an amazing opportunity.”
Overcoming Misperceptions About Unions
One barrier to doctors getting involved is the sentiment that unions might do the opposite of what’s intended — that is, they might further reduce a doctor’s autonomy and work flexibility. Or there may be a perception that the drive to join a union is predicated on making more money.
Though he’s now in a union, Dr. VenOsdel, who has been in a hospital-based practice for 7 years, admits that he initially felt very differently about unions than he does today.
“Even though I have family members in healthcare unions, I had a neutral to even slightly negative view of unions,” said Dr. VenOsdel. “It took me working directly with the Minnesota Nurses Association and the Doctors Council to learn the other side of the story.”
Armed with more information, he began lobbying for stricter rules about how his state’s large healthcare systems were closing hospitals and ending much-needed community services.
“I remember standing at the Capitol in Minnesota and telling one of the members that I once felt negatively about unions,” he added. “I realized then that I only knew what employers were telling me via such things as emails about strikes — that information was all being shared from the employers’ perspective.”
The other misperception is that unions only exist to argue against management, including against colleagues who are also part of the management structure, said Dr. Higgins.
“Some doctors perceive being in a union as ‘how can those same leaders also be in a union,’” she said. She feels that they currently don’t have leadership representing them that can help with such things as restructuring their support teams or getting them help with certain tasks. “That’s another way unions can help.”
Social Justice Plays a Role
For Dr. VenOsdel, being part of a union has helped him return to what he calls the “art” of medicine.
“Philosophically, the union gave me an option for change in what felt like a hopeless situation,” he said. “It wasn’t just that I was tossing the keys to someone else and saying, ‘I can’t fix this.’ Instead, we’re taking the reins back and fixing things ourselves.”
Bussey argues that as the uneven balance between administrators and providers in many healthcare organizations grows, the time to consider forming a union is now.
“We’re in a $4 trillion medical industrial revolution,” he said. “Administrators and bureaucrats are multiplying 30-fold times vs providers, and most of that $4 trillion supports things that don’t contribute to the doctor-patient relationship.”
Furthermore, union proponents say that where a one-on-one relationship between doctor and patient once existed, that has now been “triangulated” to include administrators.
“We’ve lost power in every way,” Dr. Bussey said. “We have the degrees, the liability, and the knowledge — we should have more power to make our workplaces safer and better.”
Ultimately, for some unionized doctors, the very holding of a union card is rooted in supporting social justice issues.
“When doctors realize how powerful a tool a union can be for social justice and change, this will alter perceptions of unions within our profession,” Dr. VenOsdel said. “Our union helps give us a voice to stand up for other staff who aren’t unionized and, most importantly, to stand up for the patients who need us.”
A version of this article first appeared on Medscape.com.
With huge shifts over the past decade in the way doctors are employed — half of all doctors now work for a health system or large medical group — the idea of unionizing is not only being explored but gaining traction within the profession. In fact, 8% of the physician workforce (or 70,000 physicians) belong to a union, according to statistics gathered in 2022.
Exact numbers are hard to come by, and, interestingly, although the American Medical Association (AMA) “ supports the right of physicians to engage in collective bargaining,” the organization doesn’t track union membership among physicians, according to an AMA spokesperson.
Forming a Union
One challenge is that forming a union is not only time-consuming but also difficult, owing to several barriers. For starters, the laws dictating unionization differ by state, and the rules governing unionization vary if a hospital is public or private. If there’s enough momentum from doctors leading unionization efforts, approval from hospital leaders is required before an official election can be requested from the National Labor Relations Board.
That said, for doctors who are in a union — the two most popular are the Union of American Physicians and Dentists and the Doctors Council branch of the Service Employees International Union (SEIU)—the benefits are immense, especially because union members can focus on what matters, such as providing the best patient care possible.
, reported WBUR in Boston.
Belonging Matters
“When you build a relationship with your patients, it’s special, and that connection isn’t replaceable,” said Nicholas VenOsdel, MD, a pediatrician at Allina Health Primary Care in Hastings, Minnesota, and a union member of the Doctors Council. “However, a lot of us have felt like that hasn’t been respected as the climate of healthcare has changed so fast.”
In fact, autonomy over how much time doctors spend with patients is driving a lot of interest in unionization.
“We don’t necessarily have that autonomy now,” said Amber Higgins, MD, an emergency physician and an obstetrician at ChristianaCare, a hospital network in Newark, Delaware, and a member of the Doctors Council. “There are so many other demands, whether it’s billing, patient documentation, or other demands from the employer, and all of that takes time away from patient care.”
Another primary driver of physician unionization is the physician burnout epidemic. Physicians collectively complain that they spend more time on electronic health record documentation and bureaucratic administration. Yet if unions can improve these working conditions, the benefit to physicians and their patients would be a welcome change.
Union members are bullish and believe that having a cohesive voice will make a difference.
“We need to use our collective voices to get back to focusing on patient care instead of staring at a computer screen for 80% of the day,” Dr. Higgins told this news organization. “So much of medicine involves getting to the correct diagnosis, listening to patients, observing them, and building a relationship with them. We need time to build that.”
With corporate consolidation and a profit-driven mandate by healthcare systems, doctors are increasingly frustrated and feel that their voices haven’t been heard enough when it comes to issues like workplace safety, working hours, and benefits, said Stuart Bussey, MD, JD, a family practice physician and president of the Union of American Physicians and Dentists in Sacramento, California.
However, he adds that urging doctors to join together to fight for a better working environment hasn’t been easy.
“Doctors are individualists, and they don’t know how to work in packs like hospital administrators do,” said Dr. Bussey. “They’re hard to organize, but I want them to understand that unless they join hands, sign petitions, and speak as one voice, they’re going to lose out on an amazing opportunity.”
Overcoming Misperceptions About Unions
One barrier to doctors getting involved is the sentiment that unions might do the opposite of what’s intended — that is, they might further reduce a doctor’s autonomy and work flexibility. Or there may be a perception that the drive to join a union is predicated on making more money.
Though he’s now in a union, Dr. VenOsdel, who has been in a hospital-based practice for 7 years, admits that he initially felt very differently about unions than he does today.
“Even though I have family members in healthcare unions, I had a neutral to even slightly negative view of unions,” said Dr. VenOsdel. “It took me working directly with the Minnesota Nurses Association and the Doctors Council to learn the other side of the story.”
Armed with more information, he began lobbying for stricter rules about how his state’s large healthcare systems were closing hospitals and ending much-needed community services.
“I remember standing at the Capitol in Minnesota and telling one of the members that I once felt negatively about unions,” he added. “I realized then that I only knew what employers were telling me via such things as emails about strikes — that information was all being shared from the employers’ perspective.”
The other misperception is that unions only exist to argue against management, including against colleagues who are also part of the management structure, said Dr. Higgins.
“Some doctors perceive being in a union as ‘how can those same leaders also be in a union,’” she said. She feels that they currently don’t have leadership representing them that can help with such things as restructuring their support teams or getting them help with certain tasks. “That’s another way unions can help.”
Social Justice Plays a Role
For Dr. VenOsdel, being part of a union has helped him return to what he calls the “art” of medicine.
“Philosophically, the union gave me an option for change in what felt like a hopeless situation,” he said. “It wasn’t just that I was tossing the keys to someone else and saying, ‘I can’t fix this.’ Instead, we’re taking the reins back and fixing things ourselves.”
Bussey argues that as the uneven balance between administrators and providers in many healthcare organizations grows, the time to consider forming a union is now.
“We’re in a $4 trillion medical industrial revolution,” he said. “Administrators and bureaucrats are multiplying 30-fold times vs providers, and most of that $4 trillion supports things that don’t contribute to the doctor-patient relationship.”
Furthermore, union proponents say that where a one-on-one relationship between doctor and patient once existed, that has now been “triangulated” to include administrators.
“We’ve lost power in every way,” Dr. Bussey said. “We have the degrees, the liability, and the knowledge — we should have more power to make our workplaces safer and better.”
Ultimately, for some unionized doctors, the very holding of a union card is rooted in supporting social justice issues.
“When doctors realize how powerful a tool a union can be for social justice and change, this will alter perceptions of unions within our profession,” Dr. VenOsdel said. “Our union helps give us a voice to stand up for other staff who aren’t unionized and, most importantly, to stand up for the patients who need us.”
A version of this article first appeared on Medscape.com.
With huge shifts over the past decade in the way doctors are employed — half of all doctors now work for a health system or large medical group — the idea of unionizing is not only being explored but gaining traction within the profession. In fact, 8% of the physician workforce (or 70,000 physicians) belong to a union, according to statistics gathered in 2022.
Exact numbers are hard to come by, and, interestingly, although the American Medical Association (AMA) “ supports the right of physicians to engage in collective bargaining,” the organization doesn’t track union membership among physicians, according to an AMA spokesperson.
Forming a Union
One challenge is that forming a union is not only time-consuming but also difficult, owing to several barriers. For starters, the laws dictating unionization differ by state, and the rules governing unionization vary if a hospital is public or private. If there’s enough momentum from doctors leading unionization efforts, approval from hospital leaders is required before an official election can be requested from the National Labor Relations Board.
That said, for doctors who are in a union — the two most popular are the Union of American Physicians and Dentists and the Doctors Council branch of the Service Employees International Union (SEIU)—the benefits are immense, especially because union members can focus on what matters, such as providing the best patient care possible.
, reported WBUR in Boston.
Belonging Matters
“When you build a relationship with your patients, it’s special, and that connection isn’t replaceable,” said Nicholas VenOsdel, MD, a pediatrician at Allina Health Primary Care in Hastings, Minnesota, and a union member of the Doctors Council. “However, a lot of us have felt like that hasn’t been respected as the climate of healthcare has changed so fast.”
In fact, autonomy over how much time doctors spend with patients is driving a lot of interest in unionization.
“We don’t necessarily have that autonomy now,” said Amber Higgins, MD, an emergency physician and an obstetrician at ChristianaCare, a hospital network in Newark, Delaware, and a member of the Doctors Council. “There are so many other demands, whether it’s billing, patient documentation, or other demands from the employer, and all of that takes time away from patient care.”
Another primary driver of physician unionization is the physician burnout epidemic. Physicians collectively complain that they spend more time on electronic health record documentation and bureaucratic administration. Yet if unions can improve these working conditions, the benefit to physicians and their patients would be a welcome change.
Union members are bullish and believe that having a cohesive voice will make a difference.
“We need to use our collective voices to get back to focusing on patient care instead of staring at a computer screen for 80% of the day,” Dr. Higgins told this news organization. “So much of medicine involves getting to the correct diagnosis, listening to patients, observing them, and building a relationship with them. We need time to build that.”
With corporate consolidation and a profit-driven mandate by healthcare systems, doctors are increasingly frustrated and feel that their voices haven’t been heard enough when it comes to issues like workplace safety, working hours, and benefits, said Stuart Bussey, MD, JD, a family practice physician and president of the Union of American Physicians and Dentists in Sacramento, California.
However, he adds that urging doctors to join together to fight for a better working environment hasn’t been easy.
“Doctors are individualists, and they don’t know how to work in packs like hospital administrators do,” said Dr. Bussey. “They’re hard to organize, but I want them to understand that unless they join hands, sign petitions, and speak as one voice, they’re going to lose out on an amazing opportunity.”
Overcoming Misperceptions About Unions
One barrier to doctors getting involved is the sentiment that unions might do the opposite of what’s intended — that is, they might further reduce a doctor’s autonomy and work flexibility. Or there may be a perception that the drive to join a union is predicated on making more money.
Though he’s now in a union, Dr. VenOsdel, who has been in a hospital-based practice for 7 years, admits that he initially felt very differently about unions than he does today.
“Even though I have family members in healthcare unions, I had a neutral to even slightly negative view of unions,” said Dr. VenOsdel. “It took me working directly with the Minnesota Nurses Association and the Doctors Council to learn the other side of the story.”
Armed with more information, he began lobbying for stricter rules about how his state’s large healthcare systems were closing hospitals and ending much-needed community services.
“I remember standing at the Capitol in Minnesota and telling one of the members that I once felt negatively about unions,” he added. “I realized then that I only knew what employers were telling me via such things as emails about strikes — that information was all being shared from the employers’ perspective.”
The other misperception is that unions only exist to argue against management, including against colleagues who are also part of the management structure, said Dr. Higgins.
“Some doctors perceive being in a union as ‘how can those same leaders also be in a union,’” she said. She feels that they currently don’t have leadership representing them that can help with such things as restructuring their support teams or getting them help with certain tasks. “That’s another way unions can help.”
Social Justice Plays a Role
For Dr. VenOsdel, being part of a union has helped him return to what he calls the “art” of medicine.
“Philosophically, the union gave me an option for change in what felt like a hopeless situation,” he said. “It wasn’t just that I was tossing the keys to someone else and saying, ‘I can’t fix this.’ Instead, we’re taking the reins back and fixing things ourselves.”
Bussey argues that as the uneven balance between administrators and providers in many healthcare organizations grows, the time to consider forming a union is now.
“We’re in a $4 trillion medical industrial revolution,” he said. “Administrators and bureaucrats are multiplying 30-fold times vs providers, and most of that $4 trillion supports things that don’t contribute to the doctor-patient relationship.”
Furthermore, union proponents say that where a one-on-one relationship between doctor and patient once existed, that has now been “triangulated” to include administrators.
“We’ve lost power in every way,” Dr. Bussey said. “We have the degrees, the liability, and the knowledge — we should have more power to make our workplaces safer and better.”
Ultimately, for some unionized doctors, the very holding of a union card is rooted in supporting social justice issues.
“When doctors realize how powerful a tool a union can be for social justice and change, this will alter perceptions of unions within our profession,” Dr. VenOsdel said. “Our union helps give us a voice to stand up for other staff who aren’t unionized and, most importantly, to stand up for the patients who need us.”
A version of this article first appeared on Medscape.com.
Updated COVID Vaccines: Who Should Get One, and When?
This transcript has been edited for clarity.
Two updated mRNA COVID vaccines, one by Moderna and the other by Pfizer, have been authorized or approved by the US Food and Drug Administration (FDA) for those aged 6 months or older.
Both vaccines target Omicron’s KP.2 strain of the JN.1 lineage. An updated protein-based version by Novavax, also directed at JN.1, has been authorized, but only for those aged 12 years or older.
The Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices recommends a dose of the 2024-2025 updated COVID vaccine for everyone aged 6 months or older. This includes people who have never been vaccinated against COVID, those who have been vaccinated, as well as people with previous COVID infection.
The big question is when, and FDA and CDC have set some parameters. For mRNA updated vaccines, patients should wait at least 2 months after their last dose of any COVID vaccine before getting a dose of the updated vaccine.
If the patient has recently had COVID, the wait time is even longer: Patients can wait 3 months after a COVID infection to be vaccinated, but they don’t have to. FDA’s instructions for the Novavax version are not as straightforward. People can get an updated Novavax dose at least 2 months after their last mRNA COVID vaccine dose, or at least 2 months after completing a Novavax two-dose primary series. Those two Novavax doses should be given at least 3 weeks apart.
Patients can personalize their vaccine plan. They will have the greatest protection in the first few weeks to months after a vaccine, after which antibodies tend to wane. It is a good idea to time vaccination so that protection peaks at big events like weddings and major meetings.
If patients decide to wait, they run the risk of getting a COVID infection. Also keep in mind which variants are circulating and the amount of local activity. Right now, there is a lot of COVID going around, and most of it is related to JN.1, the target of this year’s updated vaccine. If patients decide to wait, they should avoid crowded indoor settings or wear a high-quality mask for some protection.
Here’s the bottom line: Most people (more than 95%) have some degree of COVID protection from previous infection, vaccination, or both. But if they haven’t recently had COVID infection and didn’t get a dose of last year’s vaccine, they are sitting ducks for getting sick without updated protection. The best way to stay well is to get a dose of the updated vaccine as soon as possible. This is especially true for those in high-risk groups or who are near someone who is high risk.
Two thirds of COVID hospitalizations are in those aged 65 or older. Hospitalization rates are highest for those aged 75 or older and among infants under 6 months of age. These babies are too young to be vaccinated, but maternal vaccination during pregnancy and breastfeeding can help protect them.
We’re still seeing racial and ethnic disparities in COVID-related hospitalizations, which are highest among American Indians, Alaska Natives, and Black populations. People with immunocompromising conditions, those with chronic medical conditions, and people living in long-term care facilities are also at greater risk. Unlike last year, additional mRNA doses are not recommended for those aged 65 or older at this time, but that could change.
Since 2020, we’ve come a long way in our fight against COVID, but the battle is still on. In 2023, nearly a million people were hospitalized from COVID. More than 75,000 died. COVID vaccines help protect us from severe disease, hospitalization, and death.
Let’s face it — we all have booster fatigue, but COVID is now endemic. It’s here to stay, and it’s much safer to update antibody protection with vaccination than with infection. Another benefit of getting vaccinated is that it decreases the chance of getting long COVID. The uptake of last year’s COVID vaccine was abysmal; only about 23% of adults and 14% of children received it.
But this is a new year and a new vaccine. Please make sure your patients understand that the virus has changed a lot. Antibodies we built from previous infection and previous vaccination don’t work as well against these new variants. Antibody levels wane over time, so even if they missed the last few vaccine doses without getting sick, they really should consider getting a dose of this new vaccine. The 2024-2025 updated COVID vaccine is the best way to catch up, update their immunity, and keep them protected.
Furthermore, we are now entering respiratory virus season, which means we need to think about, recommend, and administer three shots if indicated: COVID, flu, and RSV. Now is the time. Patients can get all three at the same time, in the same visit, if they choose to do so.
Your recommendation as a physician is powerful. Adults and children who receive a healthcare provider recommendation are more likely to get vaccinated.
Dr. Fryhofer is an adjunct clinical associate professor of medicine, Emory University School of Medicine, Atlanta, Georgia. She reported conflicts of interest with the American Medical Association, the Medical Association of Atlanta, the American College of Physicians, and Medscape.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Two updated mRNA COVID vaccines, one by Moderna and the other by Pfizer, have been authorized or approved by the US Food and Drug Administration (FDA) for those aged 6 months or older.
Both vaccines target Omicron’s KP.2 strain of the JN.1 lineage. An updated protein-based version by Novavax, also directed at JN.1, has been authorized, but only for those aged 12 years or older.
The Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices recommends a dose of the 2024-2025 updated COVID vaccine for everyone aged 6 months or older. This includes people who have never been vaccinated against COVID, those who have been vaccinated, as well as people with previous COVID infection.
The big question is when, and FDA and CDC have set some parameters. For mRNA updated vaccines, patients should wait at least 2 months after their last dose of any COVID vaccine before getting a dose of the updated vaccine.
If the patient has recently had COVID, the wait time is even longer: Patients can wait 3 months after a COVID infection to be vaccinated, but they don’t have to. FDA’s instructions for the Novavax version are not as straightforward. People can get an updated Novavax dose at least 2 months after their last mRNA COVID vaccine dose, or at least 2 months after completing a Novavax two-dose primary series. Those two Novavax doses should be given at least 3 weeks apart.
Patients can personalize their vaccine plan. They will have the greatest protection in the first few weeks to months after a vaccine, after which antibodies tend to wane. It is a good idea to time vaccination so that protection peaks at big events like weddings and major meetings.
If patients decide to wait, they run the risk of getting a COVID infection. Also keep in mind which variants are circulating and the amount of local activity. Right now, there is a lot of COVID going around, and most of it is related to JN.1, the target of this year’s updated vaccine. If patients decide to wait, they should avoid crowded indoor settings or wear a high-quality mask for some protection.
Here’s the bottom line: Most people (more than 95%) have some degree of COVID protection from previous infection, vaccination, or both. But if they haven’t recently had COVID infection and didn’t get a dose of last year’s vaccine, they are sitting ducks for getting sick without updated protection. The best way to stay well is to get a dose of the updated vaccine as soon as possible. This is especially true for those in high-risk groups or who are near someone who is high risk.
Two thirds of COVID hospitalizations are in those aged 65 or older. Hospitalization rates are highest for those aged 75 or older and among infants under 6 months of age. These babies are too young to be vaccinated, but maternal vaccination during pregnancy and breastfeeding can help protect them.
We’re still seeing racial and ethnic disparities in COVID-related hospitalizations, which are highest among American Indians, Alaska Natives, and Black populations. People with immunocompromising conditions, those with chronic medical conditions, and people living in long-term care facilities are also at greater risk. Unlike last year, additional mRNA doses are not recommended for those aged 65 or older at this time, but that could change.
Since 2020, we’ve come a long way in our fight against COVID, but the battle is still on. In 2023, nearly a million people were hospitalized from COVID. More than 75,000 died. COVID vaccines help protect us from severe disease, hospitalization, and death.
Let’s face it — we all have booster fatigue, but COVID is now endemic. It’s here to stay, and it’s much safer to update antibody protection with vaccination than with infection. Another benefit of getting vaccinated is that it decreases the chance of getting long COVID. The uptake of last year’s COVID vaccine was abysmal; only about 23% of adults and 14% of children received it.
But this is a new year and a new vaccine. Please make sure your patients understand that the virus has changed a lot. Antibodies we built from previous infection and previous vaccination don’t work as well against these new variants. Antibody levels wane over time, so even if they missed the last few vaccine doses without getting sick, they really should consider getting a dose of this new vaccine. The 2024-2025 updated COVID vaccine is the best way to catch up, update their immunity, and keep them protected.
Furthermore, we are now entering respiratory virus season, which means we need to think about, recommend, and administer three shots if indicated: COVID, flu, and RSV. Now is the time. Patients can get all three at the same time, in the same visit, if they choose to do so.
Your recommendation as a physician is powerful. Adults and children who receive a healthcare provider recommendation are more likely to get vaccinated.
Dr. Fryhofer is an adjunct clinical associate professor of medicine, Emory University School of Medicine, Atlanta, Georgia. She reported conflicts of interest with the American Medical Association, the Medical Association of Atlanta, the American College of Physicians, and Medscape.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Two updated mRNA COVID vaccines, one by Moderna and the other by Pfizer, have been authorized or approved by the US Food and Drug Administration (FDA) for those aged 6 months or older.
Both vaccines target Omicron’s KP.2 strain of the JN.1 lineage. An updated protein-based version by Novavax, also directed at JN.1, has been authorized, but only for those aged 12 years or older.
The Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices recommends a dose of the 2024-2025 updated COVID vaccine for everyone aged 6 months or older. This includes people who have never been vaccinated against COVID, those who have been vaccinated, as well as people with previous COVID infection.
The big question is when, and FDA and CDC have set some parameters. For mRNA updated vaccines, patients should wait at least 2 months after their last dose of any COVID vaccine before getting a dose of the updated vaccine.
If the patient has recently had COVID, the wait time is even longer: Patients can wait 3 months after a COVID infection to be vaccinated, but they don’t have to. FDA’s instructions for the Novavax version are not as straightforward. People can get an updated Novavax dose at least 2 months after their last mRNA COVID vaccine dose, or at least 2 months after completing a Novavax two-dose primary series. Those two Novavax doses should be given at least 3 weeks apart.
Patients can personalize their vaccine plan. They will have the greatest protection in the first few weeks to months after a vaccine, after which antibodies tend to wane. It is a good idea to time vaccination so that protection peaks at big events like weddings and major meetings.
If patients decide to wait, they run the risk of getting a COVID infection. Also keep in mind which variants are circulating and the amount of local activity. Right now, there is a lot of COVID going around, and most of it is related to JN.1, the target of this year’s updated vaccine. If patients decide to wait, they should avoid crowded indoor settings or wear a high-quality mask for some protection.
Here’s the bottom line: Most people (more than 95%) have some degree of COVID protection from previous infection, vaccination, or both. But if they haven’t recently had COVID infection and didn’t get a dose of last year’s vaccine, they are sitting ducks for getting sick without updated protection. The best way to stay well is to get a dose of the updated vaccine as soon as possible. This is especially true for those in high-risk groups or who are near someone who is high risk.
Two thirds of COVID hospitalizations are in those aged 65 or older. Hospitalization rates are highest for those aged 75 or older and among infants under 6 months of age. These babies are too young to be vaccinated, but maternal vaccination during pregnancy and breastfeeding can help protect them.
We’re still seeing racial and ethnic disparities in COVID-related hospitalizations, which are highest among American Indians, Alaska Natives, and Black populations. People with immunocompromising conditions, those with chronic medical conditions, and people living in long-term care facilities are also at greater risk. Unlike last year, additional mRNA doses are not recommended for those aged 65 or older at this time, but that could change.
Since 2020, we’ve come a long way in our fight against COVID, but the battle is still on. In 2023, nearly a million people were hospitalized from COVID. More than 75,000 died. COVID vaccines help protect us from severe disease, hospitalization, and death.
Let’s face it — we all have booster fatigue, but COVID is now endemic. It’s here to stay, and it’s much safer to update antibody protection with vaccination than with infection. Another benefit of getting vaccinated is that it decreases the chance of getting long COVID. The uptake of last year’s COVID vaccine was abysmal; only about 23% of adults and 14% of children received it.
But this is a new year and a new vaccine. Please make sure your patients understand that the virus has changed a lot. Antibodies we built from previous infection and previous vaccination don’t work as well against these new variants. Antibody levels wane over time, so even if they missed the last few vaccine doses without getting sick, they really should consider getting a dose of this new vaccine. The 2024-2025 updated COVID vaccine is the best way to catch up, update their immunity, and keep them protected.
Furthermore, we are now entering respiratory virus season, which means we need to think about, recommend, and administer three shots if indicated: COVID, flu, and RSV. Now is the time. Patients can get all three at the same time, in the same visit, if they choose to do so.
Your recommendation as a physician is powerful. Adults and children who receive a healthcare provider recommendation are more likely to get vaccinated.
Dr. Fryhofer is an adjunct clinical associate professor of medicine, Emory University School of Medicine, Atlanta, Georgia. She reported conflicts of interest with the American Medical Association, the Medical Association of Atlanta, the American College of Physicians, and Medscape.
A version of this article first appeared on Medscape.com.


