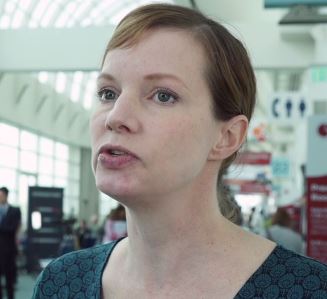User login
FLYER: Four cycles of R-CHOP as good as six in low-risk DLBCL
SAN DIEGO – A shortened regimen of four cycles of rituximab plus CHOP chemotherapy was noninferior in efficacy to the standard six cycles of R-CHOP in patients aged under age 60 years with favorable-risk diffuse large B-cell lymphoma (DLBCL), and the truncated regimen was associated with about a one-third reduction in nonhematologic adverse events, investigators in the FLYER trial reported.
Among 588 evaluable patients aged younger than 60 years with favorable-prognosis diffuse DLBCL, there were no significant differences in either progression-free survival (PFS), event-free survival, or overall survival (OS) between patients who were randomly assigned to therapy with four cycles of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone), compared with patients assigned to six cycles, reported Viola Poeschel, MD, of Saarland University in Homburg, Germany.
“Six cycles of R-CHOP led to a higher toxicity with respect to leukocytopenia and anemia, both of any grades and also of grades 3 to 4, compared to four cycles of R-CHOP,” she said in a briefing at the annual meeting of the American Society of Hematology.
For younger patients with favorable-prognosis DLBCL – defined as an age-adjusted International Prognostic Index score of 0 and low tumor burden (less than 7.5 cm) – four cycles of R-CHOP can be a new standard of care, Dr. Poeschel said.
The investigators were prompted to look at the question of a shorter R-CHOP regimen by the results of the MInT trial, in which a subpopulation of favorable-prognosis DLBCL had a 3-year PFS rate of 89% (Lancet Oncol. 2006 May;7[5]379-91). The FLYER trial was designed as a noninferiority study to see whether in a similar group of patients reducing the number of R-CHOP cycles could maintain efficacy while reducing toxicity.
At a median follow-up of 66 months, the PFS rate, the primary endpoint, was 94% in the R-CHOP 6 group, compared with 96% for R-CHOP 4.
“As the lower limit of the 95% confidence interval of our experimental arm was 94%, it is shown that it is definitely noninferior to the standard arm, six cycles of R-CHOP,” Dr. Poeschel said.
Similarly, the rate of 3-year OS was 98% in the six-cycle group, compared with 99% in the four-cycle group, and the survival curves were virtually superimposable out to more than 10 years of follow-up.
R-CHOP 6 was associated with more frequent hematologic adverse events, compared with R-CHOP 4, with leukopenia of any grade occurring in 237 versus 171 patients, respectively, and grade 3 or 4 events occurring in 110 versus 80 patients, respectively.
Any grade anemia occurred in 172 patients assigned to six cycles versus 107 assigned to four cycles. Rates of grade 3-4 anemia and thrombocytopenia of any grade or of grade 3-4 were similar between the groups.
Nonhematologic adverse events of any grade or of grade 3 or 4 that were more frequent with R-CHOP 6 versus R-CHOP 4 included all events considered together, paresthesias, nausea, infection, vomiting, and mucositis.
As noted before, the total number of nonhematologic adverse events was reduced by about one-third.
“We are certainly always looking for ways to make treatments easier for our patients to reduce adverse effects, and certainly for this subgroup of patients it appears that we can make their treatment shorter and have less burden but equivalent efficacy,” commented David Steensma, MD, from the Dana-Farber Cancer Institute/Harvard Cancer Center in Boston, who moderated the briefing.
Dr. Steensma and Dr. Poeschel both cautioned that the results of the study pertain only to those patients with DLBCL who are younger and have favorable-prognosis disease.
“We can’t extend it to other subtypes of large cell lymphoma, but that’s always a laudable goal, so I think this will immediately influence clinical practice,” Dr. Steensma said.
The study was sponsored by the German High-Grade Non-Hodgkin’s Lymphoma Study Group. Dr. Poeschel reporteed travel grants from Roche and Amgen. Dr. Steensma reported no disclosures relevant to the study.
SOURCE: Poeschel V et al. ASH 2018, Abstract 781.
SAN DIEGO – A shortened regimen of four cycles of rituximab plus CHOP chemotherapy was noninferior in efficacy to the standard six cycles of R-CHOP in patients aged under age 60 years with favorable-risk diffuse large B-cell lymphoma (DLBCL), and the truncated regimen was associated with about a one-third reduction in nonhematologic adverse events, investigators in the FLYER trial reported.
Among 588 evaluable patients aged younger than 60 years with favorable-prognosis diffuse DLBCL, there were no significant differences in either progression-free survival (PFS), event-free survival, or overall survival (OS) between patients who were randomly assigned to therapy with four cycles of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone), compared with patients assigned to six cycles, reported Viola Poeschel, MD, of Saarland University in Homburg, Germany.
“Six cycles of R-CHOP led to a higher toxicity with respect to leukocytopenia and anemia, both of any grades and also of grades 3 to 4, compared to four cycles of R-CHOP,” she said in a briefing at the annual meeting of the American Society of Hematology.
For younger patients with favorable-prognosis DLBCL – defined as an age-adjusted International Prognostic Index score of 0 and low tumor burden (less than 7.5 cm) – four cycles of R-CHOP can be a new standard of care, Dr. Poeschel said.
The investigators were prompted to look at the question of a shorter R-CHOP regimen by the results of the MInT trial, in which a subpopulation of favorable-prognosis DLBCL had a 3-year PFS rate of 89% (Lancet Oncol. 2006 May;7[5]379-91). The FLYER trial was designed as a noninferiority study to see whether in a similar group of patients reducing the number of R-CHOP cycles could maintain efficacy while reducing toxicity.
At a median follow-up of 66 months, the PFS rate, the primary endpoint, was 94% in the R-CHOP 6 group, compared with 96% for R-CHOP 4.
“As the lower limit of the 95% confidence interval of our experimental arm was 94%, it is shown that it is definitely noninferior to the standard arm, six cycles of R-CHOP,” Dr. Poeschel said.
Similarly, the rate of 3-year OS was 98% in the six-cycle group, compared with 99% in the four-cycle group, and the survival curves were virtually superimposable out to more than 10 years of follow-up.
R-CHOP 6 was associated with more frequent hematologic adverse events, compared with R-CHOP 4, with leukopenia of any grade occurring in 237 versus 171 patients, respectively, and grade 3 or 4 events occurring in 110 versus 80 patients, respectively.
Any grade anemia occurred in 172 patients assigned to six cycles versus 107 assigned to four cycles. Rates of grade 3-4 anemia and thrombocytopenia of any grade or of grade 3-4 were similar between the groups.
Nonhematologic adverse events of any grade or of grade 3 or 4 that were more frequent with R-CHOP 6 versus R-CHOP 4 included all events considered together, paresthesias, nausea, infection, vomiting, and mucositis.
As noted before, the total number of nonhematologic adverse events was reduced by about one-third.
“We are certainly always looking for ways to make treatments easier for our patients to reduce adverse effects, and certainly for this subgroup of patients it appears that we can make their treatment shorter and have less burden but equivalent efficacy,” commented David Steensma, MD, from the Dana-Farber Cancer Institute/Harvard Cancer Center in Boston, who moderated the briefing.
Dr. Steensma and Dr. Poeschel both cautioned that the results of the study pertain only to those patients with DLBCL who are younger and have favorable-prognosis disease.
“We can’t extend it to other subtypes of large cell lymphoma, but that’s always a laudable goal, so I think this will immediately influence clinical practice,” Dr. Steensma said.
The study was sponsored by the German High-Grade Non-Hodgkin’s Lymphoma Study Group. Dr. Poeschel reporteed travel grants from Roche and Amgen. Dr. Steensma reported no disclosures relevant to the study.
SOURCE: Poeschel V et al. ASH 2018, Abstract 781.
SAN DIEGO – A shortened regimen of four cycles of rituximab plus CHOP chemotherapy was noninferior in efficacy to the standard six cycles of R-CHOP in patients aged under age 60 years with favorable-risk diffuse large B-cell lymphoma (DLBCL), and the truncated regimen was associated with about a one-third reduction in nonhematologic adverse events, investigators in the FLYER trial reported.
Among 588 evaluable patients aged younger than 60 years with favorable-prognosis diffuse DLBCL, there were no significant differences in either progression-free survival (PFS), event-free survival, or overall survival (OS) between patients who were randomly assigned to therapy with four cycles of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone), compared with patients assigned to six cycles, reported Viola Poeschel, MD, of Saarland University in Homburg, Germany.
“Six cycles of R-CHOP led to a higher toxicity with respect to leukocytopenia and anemia, both of any grades and also of grades 3 to 4, compared to four cycles of R-CHOP,” she said in a briefing at the annual meeting of the American Society of Hematology.
For younger patients with favorable-prognosis DLBCL – defined as an age-adjusted International Prognostic Index score of 0 and low tumor burden (less than 7.5 cm) – four cycles of R-CHOP can be a new standard of care, Dr. Poeschel said.
The investigators were prompted to look at the question of a shorter R-CHOP regimen by the results of the MInT trial, in which a subpopulation of favorable-prognosis DLBCL had a 3-year PFS rate of 89% (Lancet Oncol. 2006 May;7[5]379-91). The FLYER trial was designed as a noninferiority study to see whether in a similar group of patients reducing the number of R-CHOP cycles could maintain efficacy while reducing toxicity.
At a median follow-up of 66 months, the PFS rate, the primary endpoint, was 94% in the R-CHOP 6 group, compared with 96% for R-CHOP 4.
“As the lower limit of the 95% confidence interval of our experimental arm was 94%, it is shown that it is definitely noninferior to the standard arm, six cycles of R-CHOP,” Dr. Poeschel said.
Similarly, the rate of 3-year OS was 98% in the six-cycle group, compared with 99% in the four-cycle group, and the survival curves were virtually superimposable out to more than 10 years of follow-up.
R-CHOP 6 was associated with more frequent hematologic adverse events, compared with R-CHOP 4, with leukopenia of any grade occurring in 237 versus 171 patients, respectively, and grade 3 or 4 events occurring in 110 versus 80 patients, respectively.
Any grade anemia occurred in 172 patients assigned to six cycles versus 107 assigned to four cycles. Rates of grade 3-4 anemia and thrombocytopenia of any grade or of grade 3-4 were similar between the groups.
Nonhematologic adverse events of any grade or of grade 3 or 4 that were more frequent with R-CHOP 6 versus R-CHOP 4 included all events considered together, paresthesias, nausea, infection, vomiting, and mucositis.
As noted before, the total number of nonhematologic adverse events was reduced by about one-third.
“We are certainly always looking for ways to make treatments easier for our patients to reduce adverse effects, and certainly for this subgroup of patients it appears that we can make their treatment shorter and have less burden but equivalent efficacy,” commented David Steensma, MD, from the Dana-Farber Cancer Institute/Harvard Cancer Center in Boston, who moderated the briefing.
Dr. Steensma and Dr. Poeschel both cautioned that the results of the study pertain only to those patients with DLBCL who are younger and have favorable-prognosis disease.
“We can’t extend it to other subtypes of large cell lymphoma, but that’s always a laudable goal, so I think this will immediately influence clinical practice,” Dr. Steensma said.
The study was sponsored by the German High-Grade Non-Hodgkin’s Lymphoma Study Group. Dr. Poeschel reporteed travel grants from Roche and Amgen. Dr. Steensma reported no disclosures relevant to the study.
SOURCE: Poeschel V et al. ASH 2018, Abstract 781.
REPORTING FROM ASH 2018
Key clinical point: Four cycles of R-CHOP was noninferior to six cycles in younger patients with favorable-prognosis diffuse large B-cell lymphoma.
Major finding: R-CHOP 4 was noninferior to R-CHOP 6 for the primary progression-free survival endpoint.
Study details: A randomized trial in 588 patients with favorable-prognosis diffuse large B-cell lymphoma.
Disclosures: The study was sponsored by the German High-Grade Non-Hodgkin’s Lymphoma Study Group. Dr. Poeschel reporteed travel grants from Roche and Amgen. Dr. Steensma reported no disclosures relevant to the study.
Source: Poeschel V et al. ASH 2018, Abstract 781.
Stem cell transplant after CAR T cells may reduce B-ALL relapse risk
SAN DIEGO – A hematopoietic cell transplant following chimeric antigen receptor (CAR) T-cell therapy for B-cell acute lymphocytic leukemia (B-ALL) may reduce late relapse risk in certain patients, a retrospective analysis suggests.
Corinne Summers, MD, of Seattle Children’s Hospital, and her colleagues evaluated the potential benefits of allogeneic hematopoietic cell transplant (HCT) in 50 pediatric and young adult B-ALL patients who had sustained leukemic remission after receiving SCRI-CAR19v1, a CD19-specific CAR T-cell product.
Leukemia-free survival was significantly improved for patients with no history of HCT who received CD19 CAR T-cell therapy followed by consolidative HCT, Dr. Summers reported at the annual meeting of the American Society of Hematology.
However, the benefits of consolidative HCT are unclear for patients with a history of HCT, Dr. Summers said at the meeting, noting that larger studies are needed.
In her video interview at ASH 2018, Dr. Summers talked more about the challenges of late leukemic relapse and the potential role of HCT after CAR T-cell therapy.
Dr. Summers reported no disclosures related to her presentation.
SAN DIEGO – A hematopoietic cell transplant following chimeric antigen receptor (CAR) T-cell therapy for B-cell acute lymphocytic leukemia (B-ALL) may reduce late relapse risk in certain patients, a retrospective analysis suggests.
Corinne Summers, MD, of Seattle Children’s Hospital, and her colleagues evaluated the potential benefits of allogeneic hematopoietic cell transplant (HCT) in 50 pediatric and young adult B-ALL patients who had sustained leukemic remission after receiving SCRI-CAR19v1, a CD19-specific CAR T-cell product.
Leukemia-free survival was significantly improved for patients with no history of HCT who received CD19 CAR T-cell therapy followed by consolidative HCT, Dr. Summers reported at the annual meeting of the American Society of Hematology.
However, the benefits of consolidative HCT are unclear for patients with a history of HCT, Dr. Summers said at the meeting, noting that larger studies are needed.
In her video interview at ASH 2018, Dr. Summers talked more about the challenges of late leukemic relapse and the potential role of HCT after CAR T-cell therapy.
Dr. Summers reported no disclosures related to her presentation.
SAN DIEGO – A hematopoietic cell transplant following chimeric antigen receptor (CAR) T-cell therapy for B-cell acute lymphocytic leukemia (B-ALL) may reduce late relapse risk in certain patients, a retrospective analysis suggests.
Corinne Summers, MD, of Seattle Children’s Hospital, and her colleagues evaluated the potential benefits of allogeneic hematopoietic cell transplant (HCT) in 50 pediatric and young adult B-ALL patients who had sustained leukemic remission after receiving SCRI-CAR19v1, a CD19-specific CAR T-cell product.
Leukemia-free survival was significantly improved for patients with no history of HCT who received CD19 CAR T-cell therapy followed by consolidative HCT, Dr. Summers reported at the annual meeting of the American Society of Hematology.
However, the benefits of consolidative HCT are unclear for patients with a history of HCT, Dr. Summers said at the meeting, noting that larger studies are needed.
In her video interview at ASH 2018, Dr. Summers talked more about the challenges of late leukemic relapse and the potential role of HCT after CAR T-cell therapy.
Dr. Summers reported no disclosures related to her presentation.
REPORTING FROM ASH 2018
Novel conditioning and familial haploidentical SCT well tolerated, cured 90% of patients
SAN DIEGO – A novel myeloablative regimen along with familial haploidentical stem cell transplantation was well tolerated and cured 90% of 19 young patients with high-risk sickle cell disease who underwent the procedure, according to Mitchell S. Cairo, MD.
The approach involved parental donors who were partial matches (as opposed to human leukocyte antigen [HLA]–matched sibling donors), CD34 enrichment, and mononuclear cell add-back (2 x 105 CD3/kg). The treatment resulted in a low cumulative incidence of acute and chronic graft-versus-host disease (6.2% and 6.7%, respectively) and stable to improved pulmonary and cardiac function. Patients also experienced significantly improved neurocognition and health-related quality of life at 2-year follow-up, Dr. Cairo of New York Medical College, Valhalla, reported at the annual meeting of the American Society of Hematology.
In a video interview, Dr. Cairo described the study, the potential benefits of familial haploidentical transplantation, and future directions.
“We have a 1-year 90% survival rate, and ... with a median follow-up now of 3 years with this approach, no patient has signs or symptoms of sickle cell disease,” he said. While the standard of care is “still to use an HLA-matched sibling donor that doesn’t have sickle cell disease,” this novel approach could benefit the five of six patients who don’t have such a donor.
The risks appear similar with the two approaches, but “more numbers will be needed to confirm this preliminary finding,” he said.
A second Food and Drug Administration–supported study with patients aged up to age 35 years (vs. 21 years in the current study) and with lower doses of the conditioning regimen to potentially reduce the risk of late adverse effects is underway, he said.
This study was supported by an FDA grant. Dr. Cairo reported receiving research funding from Janssen.
SAN DIEGO – A novel myeloablative regimen along with familial haploidentical stem cell transplantation was well tolerated and cured 90% of 19 young patients with high-risk sickle cell disease who underwent the procedure, according to Mitchell S. Cairo, MD.
The approach involved parental donors who were partial matches (as opposed to human leukocyte antigen [HLA]–matched sibling donors), CD34 enrichment, and mononuclear cell add-back (2 x 105 CD3/kg). The treatment resulted in a low cumulative incidence of acute and chronic graft-versus-host disease (6.2% and 6.7%, respectively) and stable to improved pulmonary and cardiac function. Patients also experienced significantly improved neurocognition and health-related quality of life at 2-year follow-up, Dr. Cairo of New York Medical College, Valhalla, reported at the annual meeting of the American Society of Hematology.
In a video interview, Dr. Cairo described the study, the potential benefits of familial haploidentical transplantation, and future directions.
“We have a 1-year 90% survival rate, and ... with a median follow-up now of 3 years with this approach, no patient has signs or symptoms of sickle cell disease,” he said. While the standard of care is “still to use an HLA-matched sibling donor that doesn’t have sickle cell disease,” this novel approach could benefit the five of six patients who don’t have such a donor.
The risks appear similar with the two approaches, but “more numbers will be needed to confirm this preliminary finding,” he said.
A second Food and Drug Administration–supported study with patients aged up to age 35 years (vs. 21 years in the current study) and with lower doses of the conditioning regimen to potentially reduce the risk of late adverse effects is underway, he said.
This study was supported by an FDA grant. Dr. Cairo reported receiving research funding from Janssen.
SAN DIEGO – A novel myeloablative regimen along with familial haploidentical stem cell transplantation was well tolerated and cured 90% of 19 young patients with high-risk sickle cell disease who underwent the procedure, according to Mitchell S. Cairo, MD.
The approach involved parental donors who were partial matches (as opposed to human leukocyte antigen [HLA]–matched sibling donors), CD34 enrichment, and mononuclear cell add-back (2 x 105 CD3/kg). The treatment resulted in a low cumulative incidence of acute and chronic graft-versus-host disease (6.2% and 6.7%, respectively) and stable to improved pulmonary and cardiac function. Patients also experienced significantly improved neurocognition and health-related quality of life at 2-year follow-up, Dr. Cairo of New York Medical College, Valhalla, reported at the annual meeting of the American Society of Hematology.
In a video interview, Dr. Cairo described the study, the potential benefits of familial haploidentical transplantation, and future directions.
“We have a 1-year 90% survival rate, and ... with a median follow-up now of 3 years with this approach, no patient has signs or symptoms of sickle cell disease,” he said. While the standard of care is “still to use an HLA-matched sibling donor that doesn’t have sickle cell disease,” this novel approach could benefit the five of six patients who don’t have such a donor.
The risks appear similar with the two approaches, but “more numbers will be needed to confirm this preliminary finding,” he said.
A second Food and Drug Administration–supported study with patients aged up to age 35 years (vs. 21 years in the current study) and with lower doses of the conditioning regimen to potentially reduce the risk of late adverse effects is underway, he said.
This study was supported by an FDA grant. Dr. Cairo reported receiving research funding from Janssen.
REPORTING FROM ASH 2018
New treatments promise sickle cell “cure” for all ages
SAN DIEGO –
“There is an opportunity to cure your disease no matter what age you are,” Dr. Osunkwo, medical director of the sickle cell program at Levine Cancer Institute at Atrium Health in Charlotte, N.C., said in a video interview at the annual meeting of the American Society of Hematology. “Sickle cell disease is now a disease of all ages and the treatments have to be treatments for everybody of all ages, not just for children.”
Dr. Osunkwo was the moderator of a press conference highlighting top research in sickle cell disease at ASH 2018. She pointed to findings from first-in-human trials of gene therapy using a lentiviral vector targeting BCL11A to reverse the sickle cell phenotype, as well as a study examining familial haploidentical stem cell transplantation with CD34 enrichment and mononuclear add-back in high-risk patients.
These two studies show parallel progress in curative therapies and are complementary, Dr. Osunkwo said. Improvements in transplants, and specifically in how patients are prepared and managed for them, will have a benefit in gene therapy.
But there are many other sickle cell disease studies being presented at ASH this year, she noted.
“There’s a recognition that sickle cell has been an understudied, underresourced, underexposed population,” she said. “And the suffering and the magnitude of medical problems is huge and it finally has bubbled up to the surface.”
Dr. Osunkwo reported being on advisory committees for Novartis and Pfizer and on the speaker’s bureau for Novartis. She has received honoraria from Terumo BCT and funding from the Health Resources and Services Administration and the Patient-Centered Outcomes Research Institute.
SAN DIEGO –
“There is an opportunity to cure your disease no matter what age you are,” Dr. Osunkwo, medical director of the sickle cell program at Levine Cancer Institute at Atrium Health in Charlotte, N.C., said in a video interview at the annual meeting of the American Society of Hematology. “Sickle cell disease is now a disease of all ages and the treatments have to be treatments for everybody of all ages, not just for children.”
Dr. Osunkwo was the moderator of a press conference highlighting top research in sickle cell disease at ASH 2018. She pointed to findings from first-in-human trials of gene therapy using a lentiviral vector targeting BCL11A to reverse the sickle cell phenotype, as well as a study examining familial haploidentical stem cell transplantation with CD34 enrichment and mononuclear add-back in high-risk patients.
These two studies show parallel progress in curative therapies and are complementary, Dr. Osunkwo said. Improvements in transplants, and specifically in how patients are prepared and managed for them, will have a benefit in gene therapy.
But there are many other sickle cell disease studies being presented at ASH this year, she noted.
“There’s a recognition that sickle cell has been an understudied, underresourced, underexposed population,” she said. “And the suffering and the magnitude of medical problems is huge and it finally has bubbled up to the surface.”
Dr. Osunkwo reported being on advisory committees for Novartis and Pfizer and on the speaker’s bureau for Novartis. She has received honoraria from Terumo BCT and funding from the Health Resources and Services Administration and the Patient-Centered Outcomes Research Institute.
SAN DIEGO –
“There is an opportunity to cure your disease no matter what age you are,” Dr. Osunkwo, medical director of the sickle cell program at Levine Cancer Institute at Atrium Health in Charlotte, N.C., said in a video interview at the annual meeting of the American Society of Hematology. “Sickle cell disease is now a disease of all ages and the treatments have to be treatments for everybody of all ages, not just for children.”
Dr. Osunkwo was the moderator of a press conference highlighting top research in sickle cell disease at ASH 2018. She pointed to findings from first-in-human trials of gene therapy using a lentiviral vector targeting BCL11A to reverse the sickle cell phenotype, as well as a study examining familial haploidentical stem cell transplantation with CD34 enrichment and mononuclear add-back in high-risk patients.
These two studies show parallel progress in curative therapies and are complementary, Dr. Osunkwo said. Improvements in transplants, and specifically in how patients are prepared and managed for them, will have a benefit in gene therapy.
But there are many other sickle cell disease studies being presented at ASH this year, she noted.
“There’s a recognition that sickle cell has been an understudied, underresourced, underexposed population,” she said. “And the suffering and the magnitude of medical problems is huge and it finally has bubbled up to the surface.”
Dr. Osunkwo reported being on advisory committees for Novartis and Pfizer and on the speaker’s bureau for Novartis. She has received honoraria from Terumo BCT and funding from the Health Resources and Services Administration and the Patient-Centered Outcomes Research Institute.
REPORTING FROM ASH 2018
FLYER: R-CHOP 4 safer, as effective for low-risk DLBCL patients under 60
SAN DIEGO – Patients aged younger than 60 years with favorable-prognosis diffuse large B-cell lymphoma who were randomly assigned to therapy with four cycles of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) had progression-free, event-free, and overall survival rates comparable with those of patients assigned to six cycles, investigators in the FLYER trial reported.
The four-cycle regimen was associated with a marked reduction in adverse events, with an overall drop in nonhematologic malignancies of approximately one-third compared with the six-cycle regimen.
For younger patients with favorable-prognosis DLBCL – defined as an age-adjusted International Prognostic Index score of 0 and low tumor burden (less than 7.5 cm) – four cycles of R-CHOP can be a new standard of care.
In this video interview at the annual meeting of the American Society of Hematology, Viola Poeschel, MD, of Saarland University in Homburg, Germany, describes the patient population who may benefit from shorter duration therapy.
SAN DIEGO – Patients aged younger than 60 years with favorable-prognosis diffuse large B-cell lymphoma who were randomly assigned to therapy with four cycles of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) had progression-free, event-free, and overall survival rates comparable with those of patients assigned to six cycles, investigators in the FLYER trial reported.
The four-cycle regimen was associated with a marked reduction in adverse events, with an overall drop in nonhematologic malignancies of approximately one-third compared with the six-cycle regimen.
For younger patients with favorable-prognosis DLBCL – defined as an age-adjusted International Prognostic Index score of 0 and low tumor burden (less than 7.5 cm) – four cycles of R-CHOP can be a new standard of care.
In this video interview at the annual meeting of the American Society of Hematology, Viola Poeschel, MD, of Saarland University in Homburg, Germany, describes the patient population who may benefit from shorter duration therapy.
SAN DIEGO – Patients aged younger than 60 years with favorable-prognosis diffuse large B-cell lymphoma who were randomly assigned to therapy with four cycles of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) had progression-free, event-free, and overall survival rates comparable with those of patients assigned to six cycles, investigators in the FLYER trial reported.
The four-cycle regimen was associated with a marked reduction in adverse events, with an overall drop in nonhematologic malignancies of approximately one-third compared with the six-cycle regimen.
For younger patients with favorable-prognosis DLBCL – defined as an age-adjusted International Prognostic Index score of 0 and low tumor burden (less than 7.5 cm) – four cycles of R-CHOP can be a new standard of care.
In this video interview at the annual meeting of the American Society of Hematology, Viola Poeschel, MD, of Saarland University in Homburg, Germany, describes the patient population who may benefit from shorter duration therapy.
REPORTING FROM ASH 2018
Sickle cell disease phenotype reversed by gene therapy
SAN DIEGO – An adult with sickle cell disease has had significant remissions in symptoms and a near elimination of transfusion requirements after receiving an infusion of autologous stem cells genetically modified to simultaneously induce the fetal form of hemoglobin and decrease sickle hemoglobin.
In a first-in-human, proof-of-concept study, transduction of hematopoietic stem cells with a lentiviral vector targeted against the gamma globin repressor BCL11A in erythroid cells led to rapid induction of fetal hemoglobin and a reversal of the sickle cell disease (SCD) phenotype in the early phase of stem cell reconstitution, reported Erica B. Esrick, MD, from the Dana-Farber/Boston Children’s Cancer and Blood Disorders Center in Boston.
“The potential advantage of this approach over the gene-addition strategy of gene therapy is that we can harness the physiologic switch machinery that exists in the cell to simultaneously increase fetal hemoglobin and decrease sickle hemoglobin,” she said at a briefing prior to her presentation at the annual meeting of the American Society of Hematology.
Several research groups are developing autologous gene therapy for beta-hemoglobinopathies, including the use of CRISPR-Cas9 technology to mimic a rare, naturally occurring mutation that causes the fetal type of hemoglobin to persist into adulthood in some patients with SCD and beta-thalassemia.
Dr. Esrick and her colleagues are trying a different approach: Using gene therapy to knock down BCL11A expression to induce gamma globin expression.
For the treatment, autologous hematopoietic stem cells are collected from patients following mobilization with plerixafor. The cells are then transduced with a lentiviral vector consisting of a novel short hairpin RNA embedded in an endogenous micro-RNA. The investigators refer to the construct as a shmiR (“schmeer”). The construct is designed to be erythroid specific, with BCL11A knocked down only in the red cell lineage, to avoid potential off-target effects of the therapy.
Following stem cell collection and transduction, patients undergo conditioning with busulfan prior to infusion of the modified stem cells.
In three patients treated thus far, the process has been shown to be highly efficient, with approximately 96% of treated cells transduced.
In the patient mentioned before, neutrophil engraftment was confirmed on day 22 after transfusion of the modified cells. He experienced adverse events that were consistent with myeloablative conditioning, but no adverse events associated with the modified cells.
During 6 months of follow-up the patient did not experience SCD-related pain, respiratory events, or neurologic events, and did not have anemia, with a total hemoglobin of 11 g/dL at 6 months. He has not required any transfusions since engraftment.
Patients in the trial will be followed for 2 years, and then will be enrolled in a 15-year follow-up study designed to evaluate the safety and the durability of therapy.
Dr. Esrick reported receiving honoraria from Bluebird Bio, maker of the short hairpin RNA construct used in the trial.
SOURCE: Esrick EB et al. ASH 2018, Abstract 1023.
SAN DIEGO – An adult with sickle cell disease has had significant remissions in symptoms and a near elimination of transfusion requirements after receiving an infusion of autologous stem cells genetically modified to simultaneously induce the fetal form of hemoglobin and decrease sickle hemoglobin.
In a first-in-human, proof-of-concept study, transduction of hematopoietic stem cells with a lentiviral vector targeted against the gamma globin repressor BCL11A in erythroid cells led to rapid induction of fetal hemoglobin and a reversal of the sickle cell disease (SCD) phenotype in the early phase of stem cell reconstitution, reported Erica B. Esrick, MD, from the Dana-Farber/Boston Children’s Cancer and Blood Disorders Center in Boston.
“The potential advantage of this approach over the gene-addition strategy of gene therapy is that we can harness the physiologic switch machinery that exists in the cell to simultaneously increase fetal hemoglobin and decrease sickle hemoglobin,” she said at a briefing prior to her presentation at the annual meeting of the American Society of Hematology.
Several research groups are developing autologous gene therapy for beta-hemoglobinopathies, including the use of CRISPR-Cas9 technology to mimic a rare, naturally occurring mutation that causes the fetal type of hemoglobin to persist into adulthood in some patients with SCD and beta-thalassemia.
Dr. Esrick and her colleagues are trying a different approach: Using gene therapy to knock down BCL11A expression to induce gamma globin expression.
For the treatment, autologous hematopoietic stem cells are collected from patients following mobilization with plerixafor. The cells are then transduced with a lentiviral vector consisting of a novel short hairpin RNA embedded in an endogenous micro-RNA. The investigators refer to the construct as a shmiR (“schmeer”). The construct is designed to be erythroid specific, with BCL11A knocked down only in the red cell lineage, to avoid potential off-target effects of the therapy.
Following stem cell collection and transduction, patients undergo conditioning with busulfan prior to infusion of the modified stem cells.
In three patients treated thus far, the process has been shown to be highly efficient, with approximately 96% of treated cells transduced.
In the patient mentioned before, neutrophil engraftment was confirmed on day 22 after transfusion of the modified cells. He experienced adverse events that were consistent with myeloablative conditioning, but no adverse events associated with the modified cells.
During 6 months of follow-up the patient did not experience SCD-related pain, respiratory events, or neurologic events, and did not have anemia, with a total hemoglobin of 11 g/dL at 6 months. He has not required any transfusions since engraftment.
Patients in the trial will be followed for 2 years, and then will be enrolled in a 15-year follow-up study designed to evaluate the safety and the durability of therapy.
Dr. Esrick reported receiving honoraria from Bluebird Bio, maker of the short hairpin RNA construct used in the trial.
SOURCE: Esrick EB et al. ASH 2018, Abstract 1023.
SAN DIEGO – An adult with sickle cell disease has had significant remissions in symptoms and a near elimination of transfusion requirements after receiving an infusion of autologous stem cells genetically modified to simultaneously induce the fetal form of hemoglobin and decrease sickle hemoglobin.
In a first-in-human, proof-of-concept study, transduction of hematopoietic stem cells with a lentiviral vector targeted against the gamma globin repressor BCL11A in erythroid cells led to rapid induction of fetal hemoglobin and a reversal of the sickle cell disease (SCD) phenotype in the early phase of stem cell reconstitution, reported Erica B. Esrick, MD, from the Dana-Farber/Boston Children’s Cancer and Blood Disorders Center in Boston.
“The potential advantage of this approach over the gene-addition strategy of gene therapy is that we can harness the physiologic switch machinery that exists in the cell to simultaneously increase fetal hemoglobin and decrease sickle hemoglobin,” she said at a briefing prior to her presentation at the annual meeting of the American Society of Hematology.
Several research groups are developing autologous gene therapy for beta-hemoglobinopathies, including the use of CRISPR-Cas9 technology to mimic a rare, naturally occurring mutation that causes the fetal type of hemoglobin to persist into adulthood in some patients with SCD and beta-thalassemia.
Dr. Esrick and her colleagues are trying a different approach: Using gene therapy to knock down BCL11A expression to induce gamma globin expression.
For the treatment, autologous hematopoietic stem cells are collected from patients following mobilization with plerixafor. The cells are then transduced with a lentiviral vector consisting of a novel short hairpin RNA embedded in an endogenous micro-RNA. The investigators refer to the construct as a shmiR (“schmeer”). The construct is designed to be erythroid specific, with BCL11A knocked down only in the red cell lineage, to avoid potential off-target effects of the therapy.
Following stem cell collection and transduction, patients undergo conditioning with busulfan prior to infusion of the modified stem cells.
In three patients treated thus far, the process has been shown to be highly efficient, with approximately 96% of treated cells transduced.
In the patient mentioned before, neutrophil engraftment was confirmed on day 22 after transfusion of the modified cells. He experienced adverse events that were consistent with myeloablative conditioning, but no adverse events associated with the modified cells.
During 6 months of follow-up the patient did not experience SCD-related pain, respiratory events, or neurologic events, and did not have anemia, with a total hemoglobin of 11 g/dL at 6 months. He has not required any transfusions since engraftment.
Patients in the trial will be followed for 2 years, and then will be enrolled in a 15-year follow-up study designed to evaluate the safety and the durability of therapy.
Dr. Esrick reported receiving honoraria from Bluebird Bio, maker of the short hairpin RNA construct used in the trial.
SOURCE: Esrick EB et al. ASH 2018, Abstract 1023.
REPORTING FROM ASH 2018
Key clinical point: Gene therapy to induce the fetal form of hemoglobin reversed the symptoms of sickle cell disease in an adult patient.
Major finding: During 6 months of follow-up the patient did not experience sickle cell disease–related pain, respiratory events, or neurologic events, and did not have anemia.
Study details: A first-in-human study in seven adults with sickle cell disease.
Disclosures: Dr. Esrick reported receiving honoraria from Bluebird Bio, maker of the short hairpin RNA construct used in the trial.
Source: Esrick EB et al. ASH 2018, Abstract 1023.
Daily hydroxyurea effective, safe for African children
SAN DIEGO – Daily hydroxyurea treatment for sickle cell disease is feasible, safe, and effective for children in sub-Saharan Africa, according to the results of a large open-label, phase 1-2, international trial.
Hydroxyurea was associated with reduced rates of malaria and other infections, resulting in improved survival, according to Léon Tshilolo, MD, PhD, of Centre Hospitalier Monkole in Kinshasa, the Democratic Republic of the Congo.
“Based on that data, we believe that wider access to hydroxyurea for sickle cell anemia has the potential to save millions of lives in Africa,” Dr. Tshilolo said in a press conference at the annual meeting of the American Society of Hematology.
Use of hydroxyurea has been limited in Africa because of cost, access issues, and challenges associated with laboratory monitoring, according to the researchers.
Moreover, most of the data on the efficacy of hydroxyurea come from studies conducted in the United States, Europe, and other high-income settings, said the study’s senior author Russell E. Ware, MD, PhD, of Cincinnati Children’s Hospital Center.
“Now that there’s data in an African setting, I think this will go a long way to advancing [hydroxyurea therapy] and encouraging governments, organizations, and pharmaceutical companies to bring it in,” Dr. Ware said in an interview.
In the study by Dr. Ware, Dr. Tshilolo, and their colleagues, 606 children in four sub-Saharan African countries completed a 2-month pretreatment screening phase designed to capture baseline clinical and laboratory data. The children, who were aged 1-10 years, were started at 15-20 mg/kg of hydroxyurea for 6 months, followed by escalation to the maximum tolerated dose.
With a median of 2.5 years of treatment, treated children experienced less pain and anemia, fewer cases of malaria and other infections, and lower rates of transfusions and death versus rates observed in the pretreatment screening phase of the trial.
The rate of vasoocclusive pain during hydroxyurea treatment was 44.6 events per 100 patient-years, compared with 98.3 events per 100 patient-years in the pretreatment period (incidence rate ratio, 0.45; 95% confidence interval, 0.37-0.56), according to data simultaneously published in the New England Journal of Medicine.
Malaria infection rates were 22.9 events per 100 patient-years in the hydroxyurea treatment period versus 46.9 events in the pretreatment period (IRR, 0.49; 95% CI, 0.37-0.66). Rates of nonmalaria infections were 90.0 events per 100 patient-years in the hydroxyurea treatment period versus 142.5 events per 100 patient-years in the pretreatment period (IRR, 0.62; 95% CI, 0.53-0.72).
Dr. Tshilolo said investigators were “encouraged” by the reduced infection rates, particularly in light of previous concerns that hydroxyurea could suppress the immune system and put children at risk for malaria.
Death rates were 1.1 per 100 patient-years in the hydroxyurea group and 3.6 per 100 patient-years in the pretreatment period (IR, 0.30; 95% CI, 0.10-0.88). Dose-limiting toxic events occurred in 5.1% of the children, which was below the protocol-specified threshold for safety, Dr. Tshilolo added.
Dr. Tshilolo reported grants from the National Institutes of Health/National Heart, Lung, and Blood Institute and Cincinnati Children’s Research Foundation, along with nonfinancial support from Bristol-Myers Squibb. Dr. Ware reported grants from the NIH/NHLBI and Bristol-Myers Squibb.
SOURCE: Tshilolo L et al. ASH 2018, Abstract 3.
SAN DIEGO – Daily hydroxyurea treatment for sickle cell disease is feasible, safe, and effective for children in sub-Saharan Africa, according to the results of a large open-label, phase 1-2, international trial.
Hydroxyurea was associated with reduced rates of malaria and other infections, resulting in improved survival, according to Léon Tshilolo, MD, PhD, of Centre Hospitalier Monkole in Kinshasa, the Democratic Republic of the Congo.
“Based on that data, we believe that wider access to hydroxyurea for sickle cell anemia has the potential to save millions of lives in Africa,” Dr. Tshilolo said in a press conference at the annual meeting of the American Society of Hematology.
Use of hydroxyurea has been limited in Africa because of cost, access issues, and challenges associated with laboratory monitoring, according to the researchers.
Moreover, most of the data on the efficacy of hydroxyurea come from studies conducted in the United States, Europe, and other high-income settings, said the study’s senior author Russell E. Ware, MD, PhD, of Cincinnati Children’s Hospital Center.
“Now that there’s data in an African setting, I think this will go a long way to advancing [hydroxyurea therapy] and encouraging governments, organizations, and pharmaceutical companies to bring it in,” Dr. Ware said in an interview.
In the study by Dr. Ware, Dr. Tshilolo, and their colleagues, 606 children in four sub-Saharan African countries completed a 2-month pretreatment screening phase designed to capture baseline clinical and laboratory data. The children, who were aged 1-10 years, were started at 15-20 mg/kg of hydroxyurea for 6 months, followed by escalation to the maximum tolerated dose.
With a median of 2.5 years of treatment, treated children experienced less pain and anemia, fewer cases of malaria and other infections, and lower rates of transfusions and death versus rates observed in the pretreatment screening phase of the trial.
The rate of vasoocclusive pain during hydroxyurea treatment was 44.6 events per 100 patient-years, compared with 98.3 events per 100 patient-years in the pretreatment period (incidence rate ratio, 0.45; 95% confidence interval, 0.37-0.56), according to data simultaneously published in the New England Journal of Medicine.
Malaria infection rates were 22.9 events per 100 patient-years in the hydroxyurea treatment period versus 46.9 events in the pretreatment period (IRR, 0.49; 95% CI, 0.37-0.66). Rates of nonmalaria infections were 90.0 events per 100 patient-years in the hydroxyurea treatment period versus 142.5 events per 100 patient-years in the pretreatment period (IRR, 0.62; 95% CI, 0.53-0.72).
Dr. Tshilolo said investigators were “encouraged” by the reduced infection rates, particularly in light of previous concerns that hydroxyurea could suppress the immune system and put children at risk for malaria.
Death rates were 1.1 per 100 patient-years in the hydroxyurea group and 3.6 per 100 patient-years in the pretreatment period (IR, 0.30; 95% CI, 0.10-0.88). Dose-limiting toxic events occurred in 5.1% of the children, which was below the protocol-specified threshold for safety, Dr. Tshilolo added.
Dr. Tshilolo reported grants from the National Institutes of Health/National Heart, Lung, and Blood Institute and Cincinnati Children’s Research Foundation, along with nonfinancial support from Bristol-Myers Squibb. Dr. Ware reported grants from the NIH/NHLBI and Bristol-Myers Squibb.
SOURCE: Tshilolo L et al. ASH 2018, Abstract 3.
SAN DIEGO – Daily hydroxyurea treatment for sickle cell disease is feasible, safe, and effective for children in sub-Saharan Africa, according to the results of a large open-label, phase 1-2, international trial.
Hydroxyurea was associated with reduced rates of malaria and other infections, resulting in improved survival, according to Léon Tshilolo, MD, PhD, of Centre Hospitalier Monkole in Kinshasa, the Democratic Republic of the Congo.
“Based on that data, we believe that wider access to hydroxyurea for sickle cell anemia has the potential to save millions of lives in Africa,” Dr. Tshilolo said in a press conference at the annual meeting of the American Society of Hematology.
Use of hydroxyurea has been limited in Africa because of cost, access issues, and challenges associated with laboratory monitoring, according to the researchers.
Moreover, most of the data on the efficacy of hydroxyurea come from studies conducted in the United States, Europe, and other high-income settings, said the study’s senior author Russell E. Ware, MD, PhD, of Cincinnati Children’s Hospital Center.
“Now that there’s data in an African setting, I think this will go a long way to advancing [hydroxyurea therapy] and encouraging governments, organizations, and pharmaceutical companies to bring it in,” Dr. Ware said in an interview.
In the study by Dr. Ware, Dr. Tshilolo, and their colleagues, 606 children in four sub-Saharan African countries completed a 2-month pretreatment screening phase designed to capture baseline clinical and laboratory data. The children, who were aged 1-10 years, were started at 15-20 mg/kg of hydroxyurea for 6 months, followed by escalation to the maximum tolerated dose.
With a median of 2.5 years of treatment, treated children experienced less pain and anemia, fewer cases of malaria and other infections, and lower rates of transfusions and death versus rates observed in the pretreatment screening phase of the trial.
The rate of vasoocclusive pain during hydroxyurea treatment was 44.6 events per 100 patient-years, compared with 98.3 events per 100 patient-years in the pretreatment period (incidence rate ratio, 0.45; 95% confidence interval, 0.37-0.56), according to data simultaneously published in the New England Journal of Medicine.
Malaria infection rates were 22.9 events per 100 patient-years in the hydroxyurea treatment period versus 46.9 events in the pretreatment period (IRR, 0.49; 95% CI, 0.37-0.66). Rates of nonmalaria infections were 90.0 events per 100 patient-years in the hydroxyurea treatment period versus 142.5 events per 100 patient-years in the pretreatment period (IRR, 0.62; 95% CI, 0.53-0.72).
Dr. Tshilolo said investigators were “encouraged” by the reduced infection rates, particularly in light of previous concerns that hydroxyurea could suppress the immune system and put children at risk for malaria.
Death rates were 1.1 per 100 patient-years in the hydroxyurea group and 3.6 per 100 patient-years in the pretreatment period (IR, 0.30; 95% CI, 0.10-0.88). Dose-limiting toxic events occurred in 5.1% of the children, which was below the protocol-specified threshold for safety, Dr. Tshilolo added.
Dr. Tshilolo reported grants from the National Institutes of Health/National Heart, Lung, and Blood Institute and Cincinnati Children’s Research Foundation, along with nonfinancial support from Bristol-Myers Squibb. Dr. Ware reported grants from the NIH/NHLBI and Bristol-Myers Squibb.
SOURCE: Tshilolo L et al. ASH 2018, Abstract 3.
REPORTING FROM ASH 2018
Key clinical point: Daily hydroxyurea treatment in sub-Saharan African children with sickle cell disease is feasible, safe, and effective, and has the additional benefit of reducing their rates of malaria and nonmalaria infections.
Major finding: Malaria infection rates were 22.9 versus 46.9 events per 100 patient-years in the hydroxyurea treatment period and pretreatment period, respectively (incidence rate ratio, 0.49; 95% CI, 0.37-0.66).
Study details: A phase 1-2, international, open-label trial including 606 children in four sub-Saharan African countries who completed a 2-month pretreatment screening phase and went on to receive hydroxyurea.
Disclosures: Dr. Tshilolo reported grants from the National Institutes of Health/National Heart, Lung, and Blood Institute and Cincinnati Children’s Research Foundation, along with nonfinancial support from Bristol-Myers Squibb. Dr. Ware reported grants from the NIH/NHLBI and Bristol-Myers Squibb.
Source: Tshilolo L et al. ASH 2018, Abstract 3.
Flipping the fetal hemoglobin switch reverses sickle cell symptoms
SAN DIEGO – Researchers were able to “flip the switch” from the adult to fetal form of hemoglobin using autologous stem cells genetically modified to simultaneously induce the fetal form of hemoglobin and decrease sickle hemoglobin.
The advance was announced by investigators at the Dana-Farber Cancer Institute and Boston Children’s Hospital at the annual meeting of the American Society of Hematology. At 6 months of follow-up, one adult patient in the proof-of-concept study has experienced a reversal of the sickle cell phenotype, with no pain episodes or respiratory or neurologic events.
The fetal form of hemoglobin is known to be protective against the signs and symptoms of sickle cell disease, but apart from a few rare exceptions, people with the disorder begin to experience debilitating symptoms as levels of the fetal form begin to decline in early childhood and levels of the adult form of hemoglobin steadily rise.
In this video interview, Erica B. Esrick, MD, from the Dana-Farber/Boston Children’s Cancer and Blood Disorders Center, describes the novel approach of using RNA interference to knock down a repressor that suppresses expression of gamma globin in sickle cell disease.
SAN DIEGO – Researchers were able to “flip the switch” from the adult to fetal form of hemoglobin using autologous stem cells genetically modified to simultaneously induce the fetal form of hemoglobin and decrease sickle hemoglobin.
The advance was announced by investigators at the Dana-Farber Cancer Institute and Boston Children’s Hospital at the annual meeting of the American Society of Hematology. At 6 months of follow-up, one adult patient in the proof-of-concept study has experienced a reversal of the sickle cell phenotype, with no pain episodes or respiratory or neurologic events.
The fetal form of hemoglobin is known to be protective against the signs and symptoms of sickle cell disease, but apart from a few rare exceptions, people with the disorder begin to experience debilitating symptoms as levels of the fetal form begin to decline in early childhood and levels of the adult form of hemoglobin steadily rise.
In this video interview, Erica B. Esrick, MD, from the Dana-Farber/Boston Children’s Cancer and Blood Disorders Center, describes the novel approach of using RNA interference to knock down a repressor that suppresses expression of gamma globin in sickle cell disease.
SAN DIEGO – Researchers were able to “flip the switch” from the adult to fetal form of hemoglobin using autologous stem cells genetically modified to simultaneously induce the fetal form of hemoglobin and decrease sickle hemoglobin.
The advance was announced by investigators at the Dana-Farber Cancer Institute and Boston Children’s Hospital at the annual meeting of the American Society of Hematology. At 6 months of follow-up, one adult patient in the proof-of-concept study has experienced a reversal of the sickle cell phenotype, with no pain episodes or respiratory or neurologic events.
The fetal form of hemoglobin is known to be protective against the signs and symptoms of sickle cell disease, but apart from a few rare exceptions, people with the disorder begin to experience debilitating symptoms as levels of the fetal form begin to decline in early childhood and levels of the adult form of hemoglobin steadily rise.
In this video interview, Erica B. Esrick, MD, from the Dana-Farber/Boston Children’s Cancer and Blood Disorders Center, describes the novel approach of using RNA interference to knock down a repressor that suppresses expression of gamma globin in sickle cell disease.
REPORTING FROM ASH 2018
Your guide to ASH 2018: Abstracts to watch
With more than 3,000 scientific abstracts at the 2018 annual meeting of the American Society of Hematology, it can be tough to figure out what research is most relevant to practice. But the editorial advisory board of Hematology News is making it easier this year with their picks for what to watch and why.
Lymphomas
Brian T. Hill, MD, of the Cleveland Clinic, offered his top picks in lymphoma research. Results of the phase 3 international Alliance North American Intergroup Study A041202 will be presented during the ASH plenary session at 2 p.m. PT on Sunday, Dec. 2 in Hall AB of the San Diego Convention Center (Abstract 6). The study compared bendamustine plus rituximab with ibrutinib and the combination of ibrutinib plus rituximab to see if the ibrutinib-containing therapies would have superior progression-free survival (PFS) in chronic lymphocytic leukemia (CLL), compared with chemoimmunotherapy. Results indicate that ibrutinib had superior PFS in older patients with CLL and could be a standard of care in this population.
The study is worth watching because it is the first report of a head-to-head trial of chemotherapy versus ibrutinib for first-line treatment of CLL, Dr. Hill said.
Two more studies offer important reports of “real world” experiences with chimeric antigen receptor (CAR) T-cell therapy.
In one multicenter retrospective study, researchers evaluated the outcomes of axicabtagene ciloleucel (axi-cel) CAR T-cell therapy for relapsed/refractory aggressive B-cell lymphoma when it is used a standard care. The researchers will report that 30-day responses in the real-world setting were comparable to the best responses seen in the ZUMA-1 trial. The full results will be reported at 9:30 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 91).
Another retrospective analysis looked at the use of axi-cell and revealed some critical differences from ZUMA-1, specifically the overall response rate (ORR) and complete response (CR) rate were lower than those reported in the pivotal clinical trial. The findings will be reported at 9:45 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 92).
Researchers will also present the unblinded results from the ECHELON-2 study, which compared the efficacy and safety of brentuximab vedotin in combination with cyclophosphamide, doxorubicin, and prednisone (CHP) versus standard CHOP for the treatment of patients with peripheral T-cell lymphoma. The results will be presented at 6:15 p.m. PT on Monday, Dec. 3 in room 6F of the San Diego convention center (Abstract 997).
Previously reported blinded pooled data showed that the treatment was well tolerated with 3-year PFS of 53% and OS of 73%.
“This should be a new standard of care for T-cell lymphomas,” Dr. Hill said.
CAR T-cell therapy
There are a number of abstracts featuring the latest results on CAR T-cell therapy. Helen Heslop, MD, of Baylor College of Medicine, Houston, recommended an updated analysis from the ELIANA study, which looked at the efficacy and safety of tisagenlecleucel in for children and young adults with relapsed/refractory acute lymphoblastic leukemia (ALL).
“Longer-term follow-up of the ELIANA study shows encouraging remission-duration data in pediatric and young adults with ALL without additional therapy,” Dr. Heslop said.
The findings will be presented at 4:30 p.m. PT on Monday, Dec. 3 in room 6A at the San Diego Convention Center (Abstract 895).
Another notable presentation will feature results from a phase 1B/2 trial evaluating infusion of CAR T cells targeting the CD30 molecule and encoding the CD28 endodomain (CD30.CAR-Ts) after lymphodepleting chemotherapy in patients with relapsed or refractory CD30+ Hodgkin lymphoma and non-Hodgkin lymphoma.
The researchers will report that there was a significant PFS advances for who received the highest dose level of the CAR T treatment, combined with bendamustine and fludarabine.
The study will be presented at 11 a.m. PT on Monday, Dec. 3 in room 6F at the San Diego Convention Center (Abstract 681).
Dr. Heslop also recommends another study being presented in the same session, which also shows encouraging results with CD30.CAR-Ts. Dr. Heslop is one of the co-investigators on the phase 1 RELY-30 trial, which is evaluating the efficacy of CD30.CAR-Ts after lymphodepleting chemotherapy. Preliminary results suggest a substantial improvement in efficacy. The findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in room 6F of the San Diego Convention Center (Abstract 680).
MDS/MPN
Vikas Gupta, MD, of Princess Margaret Cancer Center in Toronto, highlighted three abstracts to watch in the areas of myelodysplastic syndrome (MDS) and myeloproliferative neoplasms (MPN).
The phase 3 Medalist trial is a randomized double-blind placebo controlled study of luspatercept to treatment anemia in patients with MDS with ring sideroblasts who require red blood cell transfusion. The researchers will report significantly reduced transfusion burdens for luspatercept, compared with placebo.
“This is a practice-changing, pivotal trial in the field of MDS for the treatment of anemia,” Dr. Gupta said.
The findings will be presented at 2 p.m. PT on Sunday, Dec. 2 during the plenary session in Hall AB in the San Diego Convention Center (Abstract 1).
Also during the Sunday plenary session is a presentation on MPN therapy (Abstract 4). Researchers will present data on secreted mutant calreticulins as rogue cytokines trigger thrombopoietin receptor (TpoR) activation, specifically in CALR-mutated cells.
“This study investigates in to the mechanistic oncogenetic aspects of mutant calreticulin, and has potential for therapeutic approaches in the future,” Dr. Gupta said.
The ASH meeting will also feature the final analysis of the MPN-RC 112 consortium trial of pegylated interferon alfa-2a versus hydroxyurea for the treatment of high-risk polycythemia vera (PV) and essential thrombocythemia (ET). The researchers will report that the CR rates at 12 and 24 months were similar in patients treated with pegylated interferon alfa-2a and hydroxyurea, but pegylated interferon alfa-2a was associated with a higher rate of serious toxicities.
“There is a continuous debate on optimal first-line cytoreductive therapy for high risk PV/ET, and this is one of the first randomized study to answer this question,” Dr. Gupta said.
The findings will be presented at 7 a.m. PT on Monday, Dec. 3 in Grand Hall D at the Manchester Grand Hyatt San Diego (Abstract 577).
AML
For attendees interested in the latest developments in acute myeloid leukemia, Thomas Fischer, MD, of Otto-von-Guericke-University Magdeburg (Germany), highlighted three don’t-miss sessions.
In an analysis of a large cohort of FLT3-ITD mutated AML patients in the RATIFY trial, researchers looked at the prognostic impact of ITD insertion site.
“Interestingly, in this large cohort of 452 FLT3-ITD mutated AML, the negative prognostic impact of beta1-sheet insertion site of FLT3-ITD could be confirmed,” Dr. Fischer said. “Further analysis of a potential predictive effect on outcome of midostaurin treatment is ongoing and will be very interesting.”
The findings will be presented at 5 p.m. PT on Sunday, Dec. 2 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 435).
Another notable presentation features results from the phase 2 RADIUS trial, a randomized study comparing standard of care, with and without midostaurin, after allogeneic stem cell transplant in FLT3-ITD–mutated AML.
“Here, efficacy and toxicity of midostaurin was investigated in a [minimal residual disease] situation post-alloSCT,” Dr. Fischer said. “Interestingly, adding midostaurin to standard of care reduced the risk of relapse at 18 months post-alloSCT by 46%.”
The complete findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 662).
Dr. Fischer singled out another study looking at the efficacy and safety of single-agent quizartinib in patients with FLT3-ITD mutated AML. In this large, randomized trial the researchers noted a significant improvement in CR rates and survival benefit with the single agent FLT3 inhibitors, compared with salvage chemotherapy for patients with relapsed/refractory mutated AML.
The findings will be presented at 8 a.m. on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 563).
Notable posters
Iberia Romina Sosa, MD, PhD, of Baylor College of Medicine in Houston, suggested several posters worth visiting in the areas of thrombosis and bleeding.
Poster 1134 looks at the TNF-alpha driven inflammation and mitochondrial dysfunction in the platelet hyperreactivity of aging and MPN.
How do you know if your therapy for thrombotic thrombocytopenic purpura is working? Poster 3736 examines the measurement of cell-derived microparticles as a possible tool to monitor response to therapy.
You don’t have to be taking aspirin to have a bleeding profile characteristic with consumption of a cyclooxygenase inhibitor. Poster 1156 provides a first report of a platelet function disorder caused by autosomal recessive inheritance of PTGS1.
Poster 2477 takes a closer look at fitusiran, an antithrombin inhibitor, which improves thrombin generation in patients with hemophilia A or B. Protocol amendments for safety monitoring move fitusiran to phase 3 trials, Dr. Sosa said.
With more than 3,000 scientific abstracts at the 2018 annual meeting of the American Society of Hematology, it can be tough to figure out what research is most relevant to practice. But the editorial advisory board of Hematology News is making it easier this year with their picks for what to watch and why.
Lymphomas
Brian T. Hill, MD, of the Cleveland Clinic, offered his top picks in lymphoma research. Results of the phase 3 international Alliance North American Intergroup Study A041202 will be presented during the ASH plenary session at 2 p.m. PT on Sunday, Dec. 2 in Hall AB of the San Diego Convention Center (Abstract 6). The study compared bendamustine plus rituximab with ibrutinib and the combination of ibrutinib plus rituximab to see if the ibrutinib-containing therapies would have superior progression-free survival (PFS) in chronic lymphocytic leukemia (CLL), compared with chemoimmunotherapy. Results indicate that ibrutinib had superior PFS in older patients with CLL and could be a standard of care in this population.
The study is worth watching because it is the first report of a head-to-head trial of chemotherapy versus ibrutinib for first-line treatment of CLL, Dr. Hill said.
Two more studies offer important reports of “real world” experiences with chimeric antigen receptor (CAR) T-cell therapy.
In one multicenter retrospective study, researchers evaluated the outcomes of axicabtagene ciloleucel (axi-cel) CAR T-cell therapy for relapsed/refractory aggressive B-cell lymphoma when it is used a standard care. The researchers will report that 30-day responses in the real-world setting were comparable to the best responses seen in the ZUMA-1 trial. The full results will be reported at 9:30 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 91).
Another retrospective analysis looked at the use of axi-cell and revealed some critical differences from ZUMA-1, specifically the overall response rate (ORR) and complete response (CR) rate were lower than those reported in the pivotal clinical trial. The findings will be reported at 9:45 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 92).
Researchers will also present the unblinded results from the ECHELON-2 study, which compared the efficacy and safety of brentuximab vedotin in combination with cyclophosphamide, doxorubicin, and prednisone (CHP) versus standard CHOP for the treatment of patients with peripheral T-cell lymphoma. The results will be presented at 6:15 p.m. PT on Monday, Dec. 3 in room 6F of the San Diego convention center (Abstract 997).
Previously reported blinded pooled data showed that the treatment was well tolerated with 3-year PFS of 53% and OS of 73%.
“This should be a new standard of care for T-cell lymphomas,” Dr. Hill said.
CAR T-cell therapy
There are a number of abstracts featuring the latest results on CAR T-cell therapy. Helen Heslop, MD, of Baylor College of Medicine, Houston, recommended an updated analysis from the ELIANA study, which looked at the efficacy and safety of tisagenlecleucel in for children and young adults with relapsed/refractory acute lymphoblastic leukemia (ALL).
“Longer-term follow-up of the ELIANA study shows encouraging remission-duration data in pediatric and young adults with ALL without additional therapy,” Dr. Heslop said.
The findings will be presented at 4:30 p.m. PT on Monday, Dec. 3 in room 6A at the San Diego Convention Center (Abstract 895).
Another notable presentation will feature results from a phase 1B/2 trial evaluating infusion of CAR T cells targeting the CD30 molecule and encoding the CD28 endodomain (CD30.CAR-Ts) after lymphodepleting chemotherapy in patients with relapsed or refractory CD30+ Hodgkin lymphoma and non-Hodgkin lymphoma.
The researchers will report that there was a significant PFS advances for who received the highest dose level of the CAR T treatment, combined with bendamustine and fludarabine.
The study will be presented at 11 a.m. PT on Monday, Dec. 3 in room 6F at the San Diego Convention Center (Abstract 681).
Dr. Heslop also recommends another study being presented in the same session, which also shows encouraging results with CD30.CAR-Ts. Dr. Heslop is one of the co-investigators on the phase 1 RELY-30 trial, which is evaluating the efficacy of CD30.CAR-Ts after lymphodepleting chemotherapy. Preliminary results suggest a substantial improvement in efficacy. The findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in room 6F of the San Diego Convention Center (Abstract 680).
MDS/MPN
Vikas Gupta, MD, of Princess Margaret Cancer Center in Toronto, highlighted three abstracts to watch in the areas of myelodysplastic syndrome (MDS) and myeloproliferative neoplasms (MPN).
The phase 3 Medalist trial is a randomized double-blind placebo controlled study of luspatercept to treatment anemia in patients with MDS with ring sideroblasts who require red blood cell transfusion. The researchers will report significantly reduced transfusion burdens for luspatercept, compared with placebo.
“This is a practice-changing, pivotal trial in the field of MDS for the treatment of anemia,” Dr. Gupta said.
The findings will be presented at 2 p.m. PT on Sunday, Dec. 2 during the plenary session in Hall AB in the San Diego Convention Center (Abstract 1).
Also during the Sunday plenary session is a presentation on MPN therapy (Abstract 4). Researchers will present data on secreted mutant calreticulins as rogue cytokines trigger thrombopoietin receptor (TpoR) activation, specifically in CALR-mutated cells.
“This study investigates in to the mechanistic oncogenetic aspects of mutant calreticulin, and has potential for therapeutic approaches in the future,” Dr. Gupta said.
The ASH meeting will also feature the final analysis of the MPN-RC 112 consortium trial of pegylated interferon alfa-2a versus hydroxyurea for the treatment of high-risk polycythemia vera (PV) and essential thrombocythemia (ET). The researchers will report that the CR rates at 12 and 24 months were similar in patients treated with pegylated interferon alfa-2a and hydroxyurea, but pegylated interferon alfa-2a was associated with a higher rate of serious toxicities.
“There is a continuous debate on optimal first-line cytoreductive therapy for high risk PV/ET, and this is one of the first randomized study to answer this question,” Dr. Gupta said.
The findings will be presented at 7 a.m. PT on Monday, Dec. 3 in Grand Hall D at the Manchester Grand Hyatt San Diego (Abstract 577).
AML
For attendees interested in the latest developments in acute myeloid leukemia, Thomas Fischer, MD, of Otto-von-Guericke-University Magdeburg (Germany), highlighted three don’t-miss sessions.
In an analysis of a large cohort of FLT3-ITD mutated AML patients in the RATIFY trial, researchers looked at the prognostic impact of ITD insertion site.
“Interestingly, in this large cohort of 452 FLT3-ITD mutated AML, the negative prognostic impact of beta1-sheet insertion site of FLT3-ITD could be confirmed,” Dr. Fischer said. “Further analysis of a potential predictive effect on outcome of midostaurin treatment is ongoing and will be very interesting.”
The findings will be presented at 5 p.m. PT on Sunday, Dec. 2 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 435).
Another notable presentation features results from the phase 2 RADIUS trial, a randomized study comparing standard of care, with and without midostaurin, after allogeneic stem cell transplant in FLT3-ITD–mutated AML.
“Here, efficacy and toxicity of midostaurin was investigated in a [minimal residual disease] situation post-alloSCT,” Dr. Fischer said. “Interestingly, adding midostaurin to standard of care reduced the risk of relapse at 18 months post-alloSCT by 46%.”
The complete findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 662).
Dr. Fischer singled out another study looking at the efficacy and safety of single-agent quizartinib in patients with FLT3-ITD mutated AML. In this large, randomized trial the researchers noted a significant improvement in CR rates and survival benefit with the single agent FLT3 inhibitors, compared with salvage chemotherapy for patients with relapsed/refractory mutated AML.
The findings will be presented at 8 a.m. on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 563).
Notable posters
Iberia Romina Sosa, MD, PhD, of Baylor College of Medicine in Houston, suggested several posters worth visiting in the areas of thrombosis and bleeding.
Poster 1134 looks at the TNF-alpha driven inflammation and mitochondrial dysfunction in the platelet hyperreactivity of aging and MPN.
How do you know if your therapy for thrombotic thrombocytopenic purpura is working? Poster 3736 examines the measurement of cell-derived microparticles as a possible tool to monitor response to therapy.
You don’t have to be taking aspirin to have a bleeding profile characteristic with consumption of a cyclooxygenase inhibitor. Poster 1156 provides a first report of a platelet function disorder caused by autosomal recessive inheritance of PTGS1.
Poster 2477 takes a closer look at fitusiran, an antithrombin inhibitor, which improves thrombin generation in patients with hemophilia A or B. Protocol amendments for safety monitoring move fitusiran to phase 3 trials, Dr. Sosa said.
With more than 3,000 scientific abstracts at the 2018 annual meeting of the American Society of Hematology, it can be tough to figure out what research is most relevant to practice. But the editorial advisory board of Hematology News is making it easier this year with their picks for what to watch and why.
Lymphomas
Brian T. Hill, MD, of the Cleveland Clinic, offered his top picks in lymphoma research. Results of the phase 3 international Alliance North American Intergroup Study A041202 will be presented during the ASH plenary session at 2 p.m. PT on Sunday, Dec. 2 in Hall AB of the San Diego Convention Center (Abstract 6). The study compared bendamustine plus rituximab with ibrutinib and the combination of ibrutinib plus rituximab to see if the ibrutinib-containing therapies would have superior progression-free survival (PFS) in chronic lymphocytic leukemia (CLL), compared with chemoimmunotherapy. Results indicate that ibrutinib had superior PFS in older patients with CLL and could be a standard of care in this population.
The study is worth watching because it is the first report of a head-to-head trial of chemotherapy versus ibrutinib for first-line treatment of CLL, Dr. Hill said.
Two more studies offer important reports of “real world” experiences with chimeric antigen receptor (CAR) T-cell therapy.
In one multicenter retrospective study, researchers evaluated the outcomes of axicabtagene ciloleucel (axi-cel) CAR T-cell therapy for relapsed/refractory aggressive B-cell lymphoma when it is used a standard care. The researchers will report that 30-day responses in the real-world setting were comparable to the best responses seen in the ZUMA-1 trial. The full results will be reported at 9:30 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 91).
Another retrospective analysis looked at the use of axi-cell and revealed some critical differences from ZUMA-1, specifically the overall response rate (ORR) and complete response (CR) rate were lower than those reported in the pivotal clinical trial. The findings will be reported at 9:45 a.m. PT on Saturday, Dec. 1 in Pacific Ballroom 20 of the Marriott Marquis San Diego Marina (Abstract 92).
Researchers will also present the unblinded results from the ECHELON-2 study, which compared the efficacy and safety of brentuximab vedotin in combination with cyclophosphamide, doxorubicin, and prednisone (CHP) versus standard CHOP for the treatment of patients with peripheral T-cell lymphoma. The results will be presented at 6:15 p.m. PT on Monday, Dec. 3 in room 6F of the San Diego convention center (Abstract 997).
Previously reported blinded pooled data showed that the treatment was well tolerated with 3-year PFS of 53% and OS of 73%.
“This should be a new standard of care for T-cell lymphomas,” Dr. Hill said.
CAR T-cell therapy
There are a number of abstracts featuring the latest results on CAR T-cell therapy. Helen Heslop, MD, of Baylor College of Medicine, Houston, recommended an updated analysis from the ELIANA study, which looked at the efficacy and safety of tisagenlecleucel in for children and young adults with relapsed/refractory acute lymphoblastic leukemia (ALL).
“Longer-term follow-up of the ELIANA study shows encouraging remission-duration data in pediatric and young adults with ALL without additional therapy,” Dr. Heslop said.
The findings will be presented at 4:30 p.m. PT on Monday, Dec. 3 in room 6A at the San Diego Convention Center (Abstract 895).
Another notable presentation will feature results from a phase 1B/2 trial evaluating infusion of CAR T cells targeting the CD30 molecule and encoding the CD28 endodomain (CD30.CAR-Ts) after lymphodepleting chemotherapy in patients with relapsed or refractory CD30+ Hodgkin lymphoma and non-Hodgkin lymphoma.
The researchers will report that there was a significant PFS advances for who received the highest dose level of the CAR T treatment, combined with bendamustine and fludarabine.
The study will be presented at 11 a.m. PT on Monday, Dec. 3 in room 6F at the San Diego Convention Center (Abstract 681).
Dr. Heslop also recommends another study being presented in the same session, which also shows encouraging results with CD30.CAR-Ts. Dr. Heslop is one of the co-investigators on the phase 1 RELY-30 trial, which is evaluating the efficacy of CD30.CAR-Ts after lymphodepleting chemotherapy. Preliminary results suggest a substantial improvement in efficacy. The findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in room 6F of the San Diego Convention Center (Abstract 680).
MDS/MPN
Vikas Gupta, MD, of Princess Margaret Cancer Center in Toronto, highlighted three abstracts to watch in the areas of myelodysplastic syndrome (MDS) and myeloproliferative neoplasms (MPN).
The phase 3 Medalist trial is a randomized double-blind placebo controlled study of luspatercept to treatment anemia in patients with MDS with ring sideroblasts who require red blood cell transfusion. The researchers will report significantly reduced transfusion burdens for luspatercept, compared with placebo.
“This is a practice-changing, pivotal trial in the field of MDS for the treatment of anemia,” Dr. Gupta said.
The findings will be presented at 2 p.m. PT on Sunday, Dec. 2 during the plenary session in Hall AB in the San Diego Convention Center (Abstract 1).
Also during the Sunday plenary session is a presentation on MPN therapy (Abstract 4). Researchers will present data on secreted mutant calreticulins as rogue cytokines trigger thrombopoietin receptor (TpoR) activation, specifically in CALR-mutated cells.
“This study investigates in to the mechanistic oncogenetic aspects of mutant calreticulin, and has potential for therapeutic approaches in the future,” Dr. Gupta said.
The ASH meeting will also feature the final analysis of the MPN-RC 112 consortium trial of pegylated interferon alfa-2a versus hydroxyurea for the treatment of high-risk polycythemia vera (PV) and essential thrombocythemia (ET). The researchers will report that the CR rates at 12 and 24 months were similar in patients treated with pegylated interferon alfa-2a and hydroxyurea, but pegylated interferon alfa-2a was associated with a higher rate of serious toxicities.
“There is a continuous debate on optimal first-line cytoreductive therapy for high risk PV/ET, and this is one of the first randomized study to answer this question,” Dr. Gupta said.
The findings will be presented at 7 a.m. PT on Monday, Dec. 3 in Grand Hall D at the Manchester Grand Hyatt San Diego (Abstract 577).
AML
For attendees interested in the latest developments in acute myeloid leukemia, Thomas Fischer, MD, of Otto-von-Guericke-University Magdeburg (Germany), highlighted three don’t-miss sessions.
In an analysis of a large cohort of FLT3-ITD mutated AML patients in the RATIFY trial, researchers looked at the prognostic impact of ITD insertion site.
“Interestingly, in this large cohort of 452 FLT3-ITD mutated AML, the negative prognostic impact of beta1-sheet insertion site of FLT3-ITD could be confirmed,” Dr. Fischer said. “Further analysis of a potential predictive effect on outcome of midostaurin treatment is ongoing and will be very interesting.”
The findings will be presented at 5 p.m. PT on Sunday, Dec. 2 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 435).
Another notable presentation features results from the phase 2 RADIUS trial, a randomized study comparing standard of care, with and without midostaurin, after allogeneic stem cell transplant in FLT3-ITD–mutated AML.
“Here, efficacy and toxicity of midostaurin was investigated in a [minimal residual disease] situation post-alloSCT,” Dr. Fischer said. “Interestingly, adding midostaurin to standard of care reduced the risk of relapse at 18 months post-alloSCT by 46%.”
The complete findings will be presented at 10:45 a.m. PT on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 662).
Dr. Fischer singled out another study looking at the efficacy and safety of single-agent quizartinib in patients with FLT3-ITD mutated AML. In this large, randomized trial the researchers noted a significant improvement in CR rates and survival benefit with the single agent FLT3 inhibitors, compared with salvage chemotherapy for patients with relapsed/refractory mutated AML.
The findings will be presented at 8 a.m. on Monday, Dec. 3 in Seaport Ballroom F at the Manchester Grand Hyatt San Diego (Abstract 563).
Notable posters
Iberia Romina Sosa, MD, PhD, of Baylor College of Medicine in Houston, suggested several posters worth visiting in the areas of thrombosis and bleeding.
Poster 1134 looks at the TNF-alpha driven inflammation and mitochondrial dysfunction in the platelet hyperreactivity of aging and MPN.
How do you know if your therapy for thrombotic thrombocytopenic purpura is working? Poster 3736 examines the measurement of cell-derived microparticles as a possible tool to monitor response to therapy.
You don’t have to be taking aspirin to have a bleeding profile characteristic with consumption of a cyclooxygenase inhibitor. Poster 1156 provides a first report of a platelet function disorder caused by autosomal recessive inheritance of PTGS1.
Poster 2477 takes a closer look at fitusiran, an antithrombin inhibitor, which improves thrombin generation in patients with hemophilia A or B. Protocol amendments for safety monitoring move fitusiran to phase 3 trials, Dr. Sosa said.
ASH 2018 coming attractions look at the big picture
In the closest thing the medical world has to movie trailers, the American Society of Hematology held a press conference offering
Shorter R-CHOP regimen for DLBCL
Under the heading “Big Trials, Big Results” will be data from the FLYER trial, a phase 3, randomized, deescalation trial in 592 patients aged 18-60 years with favorable-prognosis diffuse large B-cell lymphoma. The investigators report that both progression-free survival and overall survival with four cycles of R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisone) were noninferior to those for patients treated with six cycles of R-CHOP (abstract 781).
Ibrutinib mastery in CLL
Also on the program are results of a study showing that ibrutinib (Imbruvica), either alone or in combination with rituximab, is associated with superior progression-free survival than bendamustine and rituximab in older patients with chronic lymphocytic leukemia (CLL).
The trial, the Alliance North American Intergroup Study A041202 (abstract 6) is the first major trial to pit ibrutinib against the modern standard of immunochemotherapy rather than the older standard of chlorambucil, Dr. Brodsky noted.
Anemia support in beta-thalassemia, MDS
In nonmalignant disease, investigators in the randomized, phase 3 BELIEVE trial are reporting results of their study showing that the first-in-class erythroid maturation agent luspatercept was associated with significant reductions in the need for RBC transfusion in adults with transfusion-dependent beta-thalassemia.
The investigators report that the experimental agent was “generally well tolerated” (abstract 163).
“Beyond a proof of principle, [this is] certainly a very exciting advancement in this group of patients who otherwise had very few treatment options,” said Alexis A. Thompson, MD, associate director of equity and minority health at the Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago, and the current ASH president.
Dr. Thompson also highlighted the MEDALIST trial (abstract 1), a phase 3, randomized study showing that luspatercept significantly reduced transfusion burden, compared with placebo, in patients with anemia caused by very low–, low-, or intermediate-risk myelodysplastic syndrome with ring sideroblasts who require RBC transfusions.
“This group of patients were individuals who were refractory or were not responders or did not tolerate erythropoietic stimulating agents and therefore were requiring regular transfusion,” Dr. Thompson said.
Worth the wait
The late-breaking abstract program was stretched from the usual six abstracts to seven this year because of the unusually high quality of the science, Dr. Brodsky said.
Among these star attractions are results of a phase 3, randomized study of daratumumab (Darzalex) plus lenalidomide and dexamethasone versus lenalidomide-dexamethasone alone for patients with newly diagnosed multiple myeloma who are ineligible for transplant.
The investigators found that adding daratumumab reduced the risk of disease progression or death by close to 50%, supporting the combination as a new standard of care in these patients, according to Thierry Facon, MD, from the Hospital Claude Huriez in Lille, France, and colleagues (abstract LBA-2).
Two other late-breakers deal with CLL. The first, a randomized, phase 3 study of ibrutinib-based therapy versus standard fludarabine, cyclophosphamide, and rituximab chemoimmunotherapy in younger patients with untreated CLL, found that ibrutinib and rituximab provided significantly better progression-free survival and overall survival (abstract LBA-4).
“These findings have immediate practice-changing implications and establish ibrutinib-based therapy as the most efficacious first-line therapy for patients with CLL,” wrote Tait D. Shanafelt, MD, from Stanford (Calif.) University, and colleagues.
On a less positive note, Australian researchers report their discovery of a recurrent mutation in BCL2 that confers resistance to venetoclax (Venclexta) in patients with progressive CLL (abstract LBA-7).
“This mutation provides new insights into the pathobiology of venetoclax resistance and provides a potential biomarker of impending clinical relapse,” wrote Piers Blombery, MBBS, from the University of Melbourne, and colleagues.
Finally, investigators from children’s hospitals in the United States and Europe report promising findings on the safety and efficacy of emapalumab for the treatment of patients with the rare genetic disorder primary hemophagocytic lymphohistiocytosis (HLH).
The drug, newly approved by the Food and Drug Administration, was able to control HLH’s hyperinflammatory activity, and allowed a substantial proportion of patients to survive to hematopoietic stem cell transplantation, the investigators said (abstract LBA-6).
In the closest thing the medical world has to movie trailers, the American Society of Hematology held a press conference offering
Shorter R-CHOP regimen for DLBCL
Under the heading “Big Trials, Big Results” will be data from the FLYER trial, a phase 3, randomized, deescalation trial in 592 patients aged 18-60 years with favorable-prognosis diffuse large B-cell lymphoma. The investigators report that both progression-free survival and overall survival with four cycles of R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisone) were noninferior to those for patients treated with six cycles of R-CHOP (abstract 781).
Ibrutinib mastery in CLL
Also on the program are results of a study showing that ibrutinib (Imbruvica), either alone or in combination with rituximab, is associated with superior progression-free survival than bendamustine and rituximab in older patients with chronic lymphocytic leukemia (CLL).
The trial, the Alliance North American Intergroup Study A041202 (abstract 6) is the first major trial to pit ibrutinib against the modern standard of immunochemotherapy rather than the older standard of chlorambucil, Dr. Brodsky noted.
Anemia support in beta-thalassemia, MDS
In nonmalignant disease, investigators in the randomized, phase 3 BELIEVE trial are reporting results of their study showing that the first-in-class erythroid maturation agent luspatercept was associated with significant reductions in the need for RBC transfusion in adults with transfusion-dependent beta-thalassemia.
The investigators report that the experimental agent was “generally well tolerated” (abstract 163).
“Beyond a proof of principle, [this is] certainly a very exciting advancement in this group of patients who otherwise had very few treatment options,” said Alexis A. Thompson, MD, associate director of equity and minority health at the Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago, and the current ASH president.
Dr. Thompson also highlighted the MEDALIST trial (abstract 1), a phase 3, randomized study showing that luspatercept significantly reduced transfusion burden, compared with placebo, in patients with anemia caused by very low–, low-, or intermediate-risk myelodysplastic syndrome with ring sideroblasts who require RBC transfusions.
“This group of patients were individuals who were refractory or were not responders or did not tolerate erythropoietic stimulating agents and therefore were requiring regular transfusion,” Dr. Thompson said.
Worth the wait
The late-breaking abstract program was stretched from the usual six abstracts to seven this year because of the unusually high quality of the science, Dr. Brodsky said.
Among these star attractions are results of a phase 3, randomized study of daratumumab (Darzalex) plus lenalidomide and dexamethasone versus lenalidomide-dexamethasone alone for patients with newly diagnosed multiple myeloma who are ineligible for transplant.
The investigators found that adding daratumumab reduced the risk of disease progression or death by close to 50%, supporting the combination as a new standard of care in these patients, according to Thierry Facon, MD, from the Hospital Claude Huriez in Lille, France, and colleagues (abstract LBA-2).
Two other late-breakers deal with CLL. The first, a randomized, phase 3 study of ibrutinib-based therapy versus standard fludarabine, cyclophosphamide, and rituximab chemoimmunotherapy in younger patients with untreated CLL, found that ibrutinib and rituximab provided significantly better progression-free survival and overall survival (abstract LBA-4).
“These findings have immediate practice-changing implications and establish ibrutinib-based therapy as the most efficacious first-line therapy for patients with CLL,” wrote Tait D. Shanafelt, MD, from Stanford (Calif.) University, and colleagues.
On a less positive note, Australian researchers report their discovery of a recurrent mutation in BCL2 that confers resistance to venetoclax (Venclexta) in patients with progressive CLL (abstract LBA-7).
“This mutation provides new insights into the pathobiology of venetoclax resistance and provides a potential biomarker of impending clinical relapse,” wrote Piers Blombery, MBBS, from the University of Melbourne, and colleagues.
Finally, investigators from children’s hospitals in the United States and Europe report promising findings on the safety and efficacy of emapalumab for the treatment of patients with the rare genetic disorder primary hemophagocytic lymphohistiocytosis (HLH).
The drug, newly approved by the Food and Drug Administration, was able to control HLH’s hyperinflammatory activity, and allowed a substantial proportion of patients to survive to hematopoietic stem cell transplantation, the investigators said (abstract LBA-6).
In the closest thing the medical world has to movie trailers, the American Society of Hematology held a press conference offering
Shorter R-CHOP regimen for DLBCL
Under the heading “Big Trials, Big Results” will be data from the FLYER trial, a phase 3, randomized, deescalation trial in 592 patients aged 18-60 years with favorable-prognosis diffuse large B-cell lymphoma. The investigators report that both progression-free survival and overall survival with four cycles of R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisone) were noninferior to those for patients treated with six cycles of R-CHOP (abstract 781).
Ibrutinib mastery in CLL
Also on the program are results of a study showing that ibrutinib (Imbruvica), either alone or in combination with rituximab, is associated with superior progression-free survival than bendamustine and rituximab in older patients with chronic lymphocytic leukemia (CLL).
The trial, the Alliance North American Intergroup Study A041202 (abstract 6) is the first major trial to pit ibrutinib against the modern standard of immunochemotherapy rather than the older standard of chlorambucil, Dr. Brodsky noted.
Anemia support in beta-thalassemia, MDS
In nonmalignant disease, investigators in the randomized, phase 3 BELIEVE trial are reporting results of their study showing that the first-in-class erythroid maturation agent luspatercept was associated with significant reductions in the need for RBC transfusion in adults with transfusion-dependent beta-thalassemia.
The investigators report that the experimental agent was “generally well tolerated” (abstract 163).
“Beyond a proof of principle, [this is] certainly a very exciting advancement in this group of patients who otherwise had very few treatment options,” said Alexis A. Thompson, MD, associate director of equity and minority health at the Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago, and the current ASH president.
Dr. Thompson also highlighted the MEDALIST trial (abstract 1), a phase 3, randomized study showing that luspatercept significantly reduced transfusion burden, compared with placebo, in patients with anemia caused by very low–, low-, or intermediate-risk myelodysplastic syndrome with ring sideroblasts who require RBC transfusions.
“This group of patients were individuals who were refractory or were not responders or did not tolerate erythropoietic stimulating agents and therefore were requiring regular transfusion,” Dr. Thompson said.
Worth the wait
The late-breaking abstract program was stretched from the usual six abstracts to seven this year because of the unusually high quality of the science, Dr. Brodsky said.
Among these star attractions are results of a phase 3, randomized study of daratumumab (Darzalex) plus lenalidomide and dexamethasone versus lenalidomide-dexamethasone alone for patients with newly diagnosed multiple myeloma who are ineligible for transplant.
The investigators found that adding daratumumab reduced the risk of disease progression or death by close to 50%, supporting the combination as a new standard of care in these patients, according to Thierry Facon, MD, from the Hospital Claude Huriez in Lille, France, and colleagues (abstract LBA-2).
Two other late-breakers deal with CLL. The first, a randomized, phase 3 study of ibrutinib-based therapy versus standard fludarabine, cyclophosphamide, and rituximab chemoimmunotherapy in younger patients with untreated CLL, found that ibrutinib and rituximab provided significantly better progression-free survival and overall survival (abstract LBA-4).
“These findings have immediate practice-changing implications and establish ibrutinib-based therapy as the most efficacious first-line therapy for patients with CLL,” wrote Tait D. Shanafelt, MD, from Stanford (Calif.) University, and colleagues.
On a less positive note, Australian researchers report their discovery of a recurrent mutation in BCL2 that confers resistance to venetoclax (Venclexta) in patients with progressive CLL (abstract LBA-7).
“This mutation provides new insights into the pathobiology of venetoclax resistance and provides a potential biomarker of impending clinical relapse,” wrote Piers Blombery, MBBS, from the University of Melbourne, and colleagues.
Finally, investigators from children’s hospitals in the United States and Europe report promising findings on the safety and efficacy of emapalumab for the treatment of patients with the rare genetic disorder primary hemophagocytic lymphohistiocytosis (HLH).
The drug, newly approved by the Food and Drug Administration, was able to control HLH’s hyperinflammatory activity, and allowed a substantial proportion of patients to survive to hematopoietic stem cell transplantation, the investigators said (abstract LBA-6).