User login
Medicaid expansion associated with lower cardiovascular mortality
Counties in states that expanded Medicaid coverage under the Affordable Care Act have experienced a significantly smaller increase in cardiovascular mortality rates among middle-aged adults, compared with counties in states that did not expand coverage, according to findings from a new study.
In expansion-state counties, the change in cardiovascular mortality was stable between the pre-expansion (2010-2013) and postexpansion (2014-2016) periods, at 146.5-146.4 deaths per 100,000 residents per year, compared with mortality rates in nonexpansion counties during the same periods (176.3-180.9 deaths per 100,000), Sameed Ahmed M. Khatana, MD, and colleagues wrote in JAMA Cardiology.
“After accounting for demographic, clinical, and economic differences, counties in expansion states had 4.3 fewer deaths per 100,000 residents per year from cardiovascular causes after Medicaid expansion than if they had followed the same trends as counties in nonexpansion states,” Dr. Khatana, of the University of Pennsylvania, Philadelphia, and colleagues wrote..
That translated into 2,039 fewer total deaths per year in residents aged between 45 and 64 years from cardiovascular causes after Medicaid expansion, the authors noted.
In all, 29 states, plus Washington, D.C., were included in the expansion group, and 19 states were in the nonexpansion (control) group. During the study period, from 2010 to 2016, the number of expansion counties ranged between 912 and 931, and for the nonexpansion counties, between 985 and 1,029. About half of the residents in each group were women. The percentage of black residents was lower in expansion states, but the percentage of Hispanic residents did not differ. Compared with nonexpansion counties, expansion counties also had a lower prevalence of diabetes (8.5% vs. 9.7% in the nonexpansion counties), obesity (26.2% vs. 29.1%), and smoking (17.1 vs. 18.9%); a lower mean percentage of poor residents (14.4% vs 16.6%; all with P less than .001); and a higher median household income.
Expansion counties also fared better when it came to health insurance coverage. In 2010, 14.6% of their residents had no coverage, compared with 19.5% of residents in nonexpansion counties. During the study period, the decrease in the percentage of middle-aged residents without health coverage was larger in expansion than in nonexpansion counties (7.3% vs. 5.6%, respectively), as was the decrease in low-income residents without coverage (19.8% vs. 13.5%).
However, the authors cautioned that, given the observational nature of the study, they were “not able to make a causal association between expansion of Medicaid eligibility and differences in the cardiovascular mortality rates between the two groups of counties. It is possible that there were other unmeasured time varying factors that can explain the observed association.”
Despite that limitation of the study, which observed adults in all income categories and was not limited to low-income residents, the researchers noted that, given the association between Medicaid expansion and cardiovascular mortality rates, as well as the “high burden of cardiovascular risk factors among individuals without insurance and those with lower socioeconomic status,” policy makers might consider the results in future discussions about changes to eligibility for and expansion of Medicaid.
Dr. Khatana is supported by a grant from the National Institutes of Health. Two authors reported relationships with drug companies outside of the reported study; the rest of the authors had no disclosures to report.
SOURCE: Khatana SAM et al. JAMA Cardiol. 2019 Jun 5. doi: 10.1001/jamacardio.2019.1651.
Counties in states that expanded Medicaid coverage under the Affordable Care Act have experienced a significantly smaller increase in cardiovascular mortality rates among middle-aged adults, compared with counties in states that did not expand coverage, according to findings from a new study.
In expansion-state counties, the change in cardiovascular mortality was stable between the pre-expansion (2010-2013) and postexpansion (2014-2016) periods, at 146.5-146.4 deaths per 100,000 residents per year, compared with mortality rates in nonexpansion counties during the same periods (176.3-180.9 deaths per 100,000), Sameed Ahmed M. Khatana, MD, and colleagues wrote in JAMA Cardiology.
“After accounting for demographic, clinical, and economic differences, counties in expansion states had 4.3 fewer deaths per 100,000 residents per year from cardiovascular causes after Medicaid expansion than if they had followed the same trends as counties in nonexpansion states,” Dr. Khatana, of the University of Pennsylvania, Philadelphia, and colleagues wrote..
That translated into 2,039 fewer total deaths per year in residents aged between 45 and 64 years from cardiovascular causes after Medicaid expansion, the authors noted.
In all, 29 states, plus Washington, D.C., were included in the expansion group, and 19 states were in the nonexpansion (control) group. During the study period, from 2010 to 2016, the number of expansion counties ranged between 912 and 931, and for the nonexpansion counties, between 985 and 1,029. About half of the residents in each group were women. The percentage of black residents was lower in expansion states, but the percentage of Hispanic residents did not differ. Compared with nonexpansion counties, expansion counties also had a lower prevalence of diabetes (8.5% vs. 9.7% in the nonexpansion counties), obesity (26.2% vs. 29.1%), and smoking (17.1 vs. 18.9%); a lower mean percentage of poor residents (14.4% vs 16.6%; all with P less than .001); and a higher median household income.
Expansion counties also fared better when it came to health insurance coverage. In 2010, 14.6% of their residents had no coverage, compared with 19.5% of residents in nonexpansion counties. During the study period, the decrease in the percentage of middle-aged residents without health coverage was larger in expansion than in nonexpansion counties (7.3% vs. 5.6%, respectively), as was the decrease in low-income residents without coverage (19.8% vs. 13.5%).
However, the authors cautioned that, given the observational nature of the study, they were “not able to make a causal association between expansion of Medicaid eligibility and differences in the cardiovascular mortality rates between the two groups of counties. It is possible that there were other unmeasured time varying factors that can explain the observed association.”
Despite that limitation of the study, which observed adults in all income categories and was not limited to low-income residents, the researchers noted that, given the association between Medicaid expansion and cardiovascular mortality rates, as well as the “high burden of cardiovascular risk factors among individuals without insurance and those with lower socioeconomic status,” policy makers might consider the results in future discussions about changes to eligibility for and expansion of Medicaid.
Dr. Khatana is supported by a grant from the National Institutes of Health. Two authors reported relationships with drug companies outside of the reported study; the rest of the authors had no disclosures to report.
SOURCE: Khatana SAM et al. JAMA Cardiol. 2019 Jun 5. doi: 10.1001/jamacardio.2019.1651.
Counties in states that expanded Medicaid coverage under the Affordable Care Act have experienced a significantly smaller increase in cardiovascular mortality rates among middle-aged adults, compared with counties in states that did not expand coverage, according to findings from a new study.
In expansion-state counties, the change in cardiovascular mortality was stable between the pre-expansion (2010-2013) and postexpansion (2014-2016) periods, at 146.5-146.4 deaths per 100,000 residents per year, compared with mortality rates in nonexpansion counties during the same periods (176.3-180.9 deaths per 100,000), Sameed Ahmed M. Khatana, MD, and colleagues wrote in JAMA Cardiology.
“After accounting for demographic, clinical, and economic differences, counties in expansion states had 4.3 fewer deaths per 100,000 residents per year from cardiovascular causes after Medicaid expansion than if they had followed the same trends as counties in nonexpansion states,” Dr. Khatana, of the University of Pennsylvania, Philadelphia, and colleagues wrote..
That translated into 2,039 fewer total deaths per year in residents aged between 45 and 64 years from cardiovascular causes after Medicaid expansion, the authors noted.
In all, 29 states, plus Washington, D.C., were included in the expansion group, and 19 states were in the nonexpansion (control) group. During the study period, from 2010 to 2016, the number of expansion counties ranged between 912 and 931, and for the nonexpansion counties, between 985 and 1,029. About half of the residents in each group were women. The percentage of black residents was lower in expansion states, but the percentage of Hispanic residents did not differ. Compared with nonexpansion counties, expansion counties also had a lower prevalence of diabetes (8.5% vs. 9.7% in the nonexpansion counties), obesity (26.2% vs. 29.1%), and smoking (17.1 vs. 18.9%); a lower mean percentage of poor residents (14.4% vs 16.6%; all with P less than .001); and a higher median household income.
Expansion counties also fared better when it came to health insurance coverage. In 2010, 14.6% of their residents had no coverage, compared with 19.5% of residents in nonexpansion counties. During the study period, the decrease in the percentage of middle-aged residents without health coverage was larger in expansion than in nonexpansion counties (7.3% vs. 5.6%, respectively), as was the decrease in low-income residents without coverage (19.8% vs. 13.5%).
However, the authors cautioned that, given the observational nature of the study, they were “not able to make a causal association between expansion of Medicaid eligibility and differences in the cardiovascular mortality rates between the two groups of counties. It is possible that there were other unmeasured time varying factors that can explain the observed association.”
Despite that limitation of the study, which observed adults in all income categories and was not limited to low-income residents, the researchers noted that, given the association between Medicaid expansion and cardiovascular mortality rates, as well as the “high burden of cardiovascular risk factors among individuals without insurance and those with lower socioeconomic status,” policy makers might consider the results in future discussions about changes to eligibility for and expansion of Medicaid.
Dr. Khatana is supported by a grant from the National Institutes of Health. Two authors reported relationships with drug companies outside of the reported study; the rest of the authors had no disclosures to report.
SOURCE: Khatana SAM et al. JAMA Cardiol. 2019 Jun 5. doi: 10.1001/jamacardio.2019.1651.
FROM JAMA CARDIOLOGY
Key clinical point:
Major finding: Counties in expansion states had 4.3 fewer deaths from cardiovascular causes per 100,000 residents per year after Medicaid expansion, compared with counties in nonexpansion states.
Study details: In this longitudinal, observational study from 2010 to 2016, researchers used a difference-in-difference approach with county-level data for adults from 48 states (excluding Massachusetts and Wisconsin) and Washington, D.C., who were aged between 45 and 64 years. The county-level data were obtained from the Centers for Disease Control and Prevention’s Online Data for Epidemiologic Research mortality database.
Disclosures: Dr. Khatana is supported by a grant from the National Institutes of Health. Two authors reported relationships with drug companies outside of the reported study; the rest of the authors had no disclosures to report.
Source: Khatana SAM et al. JAMA Cardiol. 2019 Jun 5. doi: 10.1001/jamacardio.2019.1651.
Focus on science, not format
How JHM is improving the author experience
“No hassle” new manuscript submission process
Many authors have experienced the frustration of formatting a manuscript for submission to a medical journal. The process is time consuming and each journal has different requirements. This means that if you decide to submit your manuscript to one journal and later decide that another journal is a better fit, you may spend an hour (or several hours) reformatting to meet the new journal’s unique requirements.
To allow authors to spend more time on what matters to them, we’re pleased to introduce our “No Hassle” process for initial original research and brief report manuscript submissions to the Journal of Hospital Medicine. Our goal is to eliminate unnecessary and burdensome steps in the manuscript submission process. Thus, we have relaxed formatting requirements for initial manuscript submissions. Any conventional and readable manuscript format and reference style is acceptable.
Tables and figures can be embedded in the main document file or uploaded individually, depending on your preference. Funding and disclosures should be included on the title page but there is no need to submit completed disclosure or copyright forms unless we request a manuscript revision.
Timely decisions
We have all experienced the agony of waiting months on end for a journal to make a decision about our manuscript. The review process itself can take many months (or even longer). Furthermore, a manuscript may not be published for many more months (or even longer) following acceptance. At the Journal of Hospital Medicine, we commit to making timely decisions and publishing your accepted manuscript as fast as we can.
We currently reject approximately half of all original research and brief report manuscript submissions without formal peer review. We do this for two reasons. First, we want to ensure that we’re not overburdening our peer reviewers so we only ask them to review manuscripts that we are seriously considering for publication. Second, we want to ensure that we’re being respectful of our authors’ time. If we are unlikely to publish a manuscript based on lower priority scores assigned by me, as editor-in-chief, or other journal editors, we don’t want to subject your manuscript to a lengthy peer review, but would rather return the manuscript to you quickly for timely submission elsewhere.
Here are data that support our timely decision making:
- 1.3 days = our average time from manuscript submission to rejection without formal peer review (median, less than one day).
- 23 days = our average time from manuscript submission to first decision for manuscripts sent for peer review.
We also are working to improve our time to publication. Our goal is to publish accepted manuscripts within 120 days from initial submission to publication, and within 60 days from acceptance to publication.
Dissemination
Finally, little public knowledge is gleaned from medical research unless the study is published and widely read. The Journal of Hospital Medicine is at the leading edge of helping authors disseminate their work to a broader audience. Of course, we produce press releases and distribute those to many media outlets in partnership with the Society of Hospital Medicine. We also leverage social media to promote your article through tweets, visual abstracts, and, more recently, comics or graphic medicine abstracts. Some articles are even discussed on #JHMChat, our twitter-based journal club. This work is led by our exceptional Digital Media Editors, Dr. Vineet Arora (@FutureDocs), Dr. Charlie Wray (@WrayCharles), and Dr. Grace Farris (@gracefarris).
In summary, we are committed to making the Journal of Hospital Medicine even more author friendly. To that end, we’re making it easy for authors to submit their work, making timely disposition decisions, and facilitating dissemination of the work we publish.
Dr. Shah is chief metrics officer and director of the division of hospital medicine at Cincinnati Children’s Hospital Medical Center. He is the current editor-in-chief of the Journal of Hospital Medicine.
How JHM is improving the author experience
How JHM is improving the author experience
“No hassle” new manuscript submission process
Many authors have experienced the frustration of formatting a manuscript for submission to a medical journal. The process is time consuming and each journal has different requirements. This means that if you decide to submit your manuscript to one journal and later decide that another journal is a better fit, you may spend an hour (or several hours) reformatting to meet the new journal’s unique requirements.
To allow authors to spend more time on what matters to them, we’re pleased to introduce our “No Hassle” process for initial original research and brief report manuscript submissions to the Journal of Hospital Medicine. Our goal is to eliminate unnecessary and burdensome steps in the manuscript submission process. Thus, we have relaxed formatting requirements for initial manuscript submissions. Any conventional and readable manuscript format and reference style is acceptable.
Tables and figures can be embedded in the main document file or uploaded individually, depending on your preference. Funding and disclosures should be included on the title page but there is no need to submit completed disclosure or copyright forms unless we request a manuscript revision.
Timely decisions
We have all experienced the agony of waiting months on end for a journal to make a decision about our manuscript. The review process itself can take many months (or even longer). Furthermore, a manuscript may not be published for many more months (or even longer) following acceptance. At the Journal of Hospital Medicine, we commit to making timely decisions and publishing your accepted manuscript as fast as we can.
We currently reject approximately half of all original research and brief report manuscript submissions without formal peer review. We do this for two reasons. First, we want to ensure that we’re not overburdening our peer reviewers so we only ask them to review manuscripts that we are seriously considering for publication. Second, we want to ensure that we’re being respectful of our authors’ time. If we are unlikely to publish a manuscript based on lower priority scores assigned by me, as editor-in-chief, or other journal editors, we don’t want to subject your manuscript to a lengthy peer review, but would rather return the manuscript to you quickly for timely submission elsewhere.
Here are data that support our timely decision making:
- 1.3 days = our average time from manuscript submission to rejection without formal peer review (median, less than one day).
- 23 days = our average time from manuscript submission to first decision for manuscripts sent for peer review.
We also are working to improve our time to publication. Our goal is to publish accepted manuscripts within 120 days from initial submission to publication, and within 60 days from acceptance to publication.
Dissemination
Finally, little public knowledge is gleaned from medical research unless the study is published and widely read. The Journal of Hospital Medicine is at the leading edge of helping authors disseminate their work to a broader audience. Of course, we produce press releases and distribute those to many media outlets in partnership with the Society of Hospital Medicine. We also leverage social media to promote your article through tweets, visual abstracts, and, more recently, comics or graphic medicine abstracts. Some articles are even discussed on #JHMChat, our twitter-based journal club. This work is led by our exceptional Digital Media Editors, Dr. Vineet Arora (@FutureDocs), Dr. Charlie Wray (@WrayCharles), and Dr. Grace Farris (@gracefarris).
In summary, we are committed to making the Journal of Hospital Medicine even more author friendly. To that end, we’re making it easy for authors to submit their work, making timely disposition decisions, and facilitating dissemination of the work we publish.
Dr. Shah is chief metrics officer and director of the division of hospital medicine at Cincinnati Children’s Hospital Medical Center. He is the current editor-in-chief of the Journal of Hospital Medicine.
“No hassle” new manuscript submission process
Many authors have experienced the frustration of formatting a manuscript for submission to a medical journal. The process is time consuming and each journal has different requirements. This means that if you decide to submit your manuscript to one journal and later decide that another journal is a better fit, you may spend an hour (or several hours) reformatting to meet the new journal’s unique requirements.
To allow authors to spend more time on what matters to them, we’re pleased to introduce our “No Hassle” process for initial original research and brief report manuscript submissions to the Journal of Hospital Medicine. Our goal is to eliminate unnecessary and burdensome steps in the manuscript submission process. Thus, we have relaxed formatting requirements for initial manuscript submissions. Any conventional and readable manuscript format and reference style is acceptable.
Tables and figures can be embedded in the main document file or uploaded individually, depending on your preference. Funding and disclosures should be included on the title page but there is no need to submit completed disclosure or copyright forms unless we request a manuscript revision.
Timely decisions
We have all experienced the agony of waiting months on end for a journal to make a decision about our manuscript. The review process itself can take many months (or even longer). Furthermore, a manuscript may not be published for many more months (or even longer) following acceptance. At the Journal of Hospital Medicine, we commit to making timely decisions and publishing your accepted manuscript as fast as we can.
We currently reject approximately half of all original research and brief report manuscript submissions without formal peer review. We do this for two reasons. First, we want to ensure that we’re not overburdening our peer reviewers so we only ask them to review manuscripts that we are seriously considering for publication. Second, we want to ensure that we’re being respectful of our authors’ time. If we are unlikely to publish a manuscript based on lower priority scores assigned by me, as editor-in-chief, or other journal editors, we don’t want to subject your manuscript to a lengthy peer review, but would rather return the manuscript to you quickly for timely submission elsewhere.
Here are data that support our timely decision making:
- 1.3 days = our average time from manuscript submission to rejection without formal peer review (median, less than one day).
- 23 days = our average time from manuscript submission to first decision for manuscripts sent for peer review.
We also are working to improve our time to publication. Our goal is to publish accepted manuscripts within 120 days from initial submission to publication, and within 60 days from acceptance to publication.
Dissemination
Finally, little public knowledge is gleaned from medical research unless the study is published and widely read. The Journal of Hospital Medicine is at the leading edge of helping authors disseminate their work to a broader audience. Of course, we produce press releases and distribute those to many media outlets in partnership with the Society of Hospital Medicine. We also leverage social media to promote your article through tweets, visual abstracts, and, more recently, comics or graphic medicine abstracts. Some articles are even discussed on #JHMChat, our twitter-based journal club. This work is led by our exceptional Digital Media Editors, Dr. Vineet Arora (@FutureDocs), Dr. Charlie Wray (@WrayCharles), and Dr. Grace Farris (@gracefarris).
In summary, we are committed to making the Journal of Hospital Medicine even more author friendly. To that end, we’re making it easy for authors to submit their work, making timely disposition decisions, and facilitating dissemination of the work we publish.
Dr. Shah is chief metrics officer and director of the division of hospital medicine at Cincinnati Children’s Hospital Medical Center. He is the current editor-in-chief of the Journal of Hospital Medicine.
Lipoprotein(a) levels can guide CV risk assessment and treatment
Lipoprotein(a) is an independent risk factor for atherosclerotic cardiovascular disease–related events, and plasma levels of Lp(a) could help refine risk assessment and influence treatment decisions, say the authors of a scientific statement from the National Lipid Association.
Don P. Wilson, MD, of Cook Children’s Medical Center, Fort Worth, Tex., and coauthors reviewed the evidence around testing of Lp(a) in clinical practice and its use in guiding treatment for both primary and secondary prevention. Their report is in the Journal of Clinical Lipidology.
Prospective, population-based studies point to a clear link between high Lp(a) levels and high risk of myocardial infarction, coronary heart disease, coronary artery stenosis, carotid stenosis, valvular aortic stenosis, ischemic stroke, cardiovascular mortality, and all-cause mortality, the authors wrote. This association was independent of the effect of other risk factors, including LDL cholesterol.
However, existing Lp(a) assays have not been globally standardized, and there is only incomplete evidence for age, sex, or ethnicity-specific cutoff points for high risk.
The authors suggested Lp(a) levels greater than 50 mg/dL (100 nmol/L) could be considered a risk factor that justifies the initiation of statin therapy. However ,they pointed out this level corresponded to the 80th population percentile in predominantly white populations, while in African American populations the equivalent cutoff was around 150 nmol/L.
On the issue of whom to test for Lp(a) serum levels, the authors said testing could reasonably be used to refine risk assessment for atherosclerotic cardiovascular disease in adults with first-degree relatives who experienced premature atherosclerotic cardiovascular disease, those with a personal history of the disease, or in those with severe hypercholesterolemia or suspected familial hypercholesterolemia.
However, statin therapy does not decrease Lp(a) levels, and there is also evidence that patients with high Lp(a) levels may not show as much LDL-C lowering in response to statin therapy.
“There is a lack of current evidence demonstrating that lowering Lp(a), independently of LDL-C, reduces ASCVD events in individuals with established ASCVD,” the authors wrote. “It appears that large absolute reductions in Lp(a) may be needed to demonstrate a significant clinical benefit.”
Despite this, the authors argued that in primary prevention, it was reasonable to use a Lp(a) level greater than 50 mg/dL (100 nmol/L) as a “risk-enhancing factor,” and in high-risk or very-high-risk patients with elevated LDL-C, it could prompt use of more intensive therapies.
Five authors disclosed honorarium or advisory board positions with the pharmaceutical sector. No other conflicts of interest were declared.
SOURCE: Wilson D et al. J Clin Lipidol. 2019 May 17. doi: 10.1016/j.jacl.2019.04.010.
Lipoprotein(a) is an independent risk factor for atherosclerotic cardiovascular disease–related events, and plasma levels of Lp(a) could help refine risk assessment and influence treatment decisions, say the authors of a scientific statement from the National Lipid Association.
Don P. Wilson, MD, of Cook Children’s Medical Center, Fort Worth, Tex., and coauthors reviewed the evidence around testing of Lp(a) in clinical practice and its use in guiding treatment for both primary and secondary prevention. Their report is in the Journal of Clinical Lipidology.
Prospective, population-based studies point to a clear link between high Lp(a) levels and high risk of myocardial infarction, coronary heart disease, coronary artery stenosis, carotid stenosis, valvular aortic stenosis, ischemic stroke, cardiovascular mortality, and all-cause mortality, the authors wrote. This association was independent of the effect of other risk factors, including LDL cholesterol.
However, existing Lp(a) assays have not been globally standardized, and there is only incomplete evidence for age, sex, or ethnicity-specific cutoff points for high risk.
The authors suggested Lp(a) levels greater than 50 mg/dL (100 nmol/L) could be considered a risk factor that justifies the initiation of statin therapy. However ,they pointed out this level corresponded to the 80th population percentile in predominantly white populations, while in African American populations the equivalent cutoff was around 150 nmol/L.
On the issue of whom to test for Lp(a) serum levels, the authors said testing could reasonably be used to refine risk assessment for atherosclerotic cardiovascular disease in adults with first-degree relatives who experienced premature atherosclerotic cardiovascular disease, those with a personal history of the disease, or in those with severe hypercholesterolemia or suspected familial hypercholesterolemia.
However, statin therapy does not decrease Lp(a) levels, and there is also evidence that patients with high Lp(a) levels may not show as much LDL-C lowering in response to statin therapy.
“There is a lack of current evidence demonstrating that lowering Lp(a), independently of LDL-C, reduces ASCVD events in individuals with established ASCVD,” the authors wrote. “It appears that large absolute reductions in Lp(a) may be needed to demonstrate a significant clinical benefit.”
Despite this, the authors argued that in primary prevention, it was reasonable to use a Lp(a) level greater than 50 mg/dL (100 nmol/L) as a “risk-enhancing factor,” and in high-risk or very-high-risk patients with elevated LDL-C, it could prompt use of more intensive therapies.
Five authors disclosed honorarium or advisory board positions with the pharmaceutical sector. No other conflicts of interest were declared.
SOURCE: Wilson D et al. J Clin Lipidol. 2019 May 17. doi: 10.1016/j.jacl.2019.04.010.
Lipoprotein(a) is an independent risk factor for atherosclerotic cardiovascular disease–related events, and plasma levels of Lp(a) could help refine risk assessment and influence treatment decisions, say the authors of a scientific statement from the National Lipid Association.
Don P. Wilson, MD, of Cook Children’s Medical Center, Fort Worth, Tex., and coauthors reviewed the evidence around testing of Lp(a) in clinical practice and its use in guiding treatment for both primary and secondary prevention. Their report is in the Journal of Clinical Lipidology.
Prospective, population-based studies point to a clear link between high Lp(a) levels and high risk of myocardial infarction, coronary heart disease, coronary artery stenosis, carotid stenosis, valvular aortic stenosis, ischemic stroke, cardiovascular mortality, and all-cause mortality, the authors wrote. This association was independent of the effect of other risk factors, including LDL cholesterol.
However, existing Lp(a) assays have not been globally standardized, and there is only incomplete evidence for age, sex, or ethnicity-specific cutoff points for high risk.
The authors suggested Lp(a) levels greater than 50 mg/dL (100 nmol/L) could be considered a risk factor that justifies the initiation of statin therapy. However ,they pointed out this level corresponded to the 80th population percentile in predominantly white populations, while in African American populations the equivalent cutoff was around 150 nmol/L.
On the issue of whom to test for Lp(a) serum levels, the authors said testing could reasonably be used to refine risk assessment for atherosclerotic cardiovascular disease in adults with first-degree relatives who experienced premature atherosclerotic cardiovascular disease, those with a personal history of the disease, or in those with severe hypercholesterolemia or suspected familial hypercholesterolemia.
However, statin therapy does not decrease Lp(a) levels, and there is also evidence that patients with high Lp(a) levels may not show as much LDL-C lowering in response to statin therapy.
“There is a lack of current evidence demonstrating that lowering Lp(a), independently of LDL-C, reduces ASCVD events in individuals with established ASCVD,” the authors wrote. “It appears that large absolute reductions in Lp(a) may be needed to demonstrate a significant clinical benefit.”
Despite this, the authors argued that in primary prevention, it was reasonable to use a Lp(a) level greater than 50 mg/dL (100 nmol/L) as a “risk-enhancing factor,” and in high-risk or very-high-risk patients with elevated LDL-C, it could prompt use of more intensive therapies.
Five authors disclosed honorarium or advisory board positions with the pharmaceutical sector. No other conflicts of interest were declared.
SOURCE: Wilson D et al. J Clin Lipidol. 2019 May 17. doi: 10.1016/j.jacl.2019.04.010.
FROM THE JOURNAL OF CLINICAL LIPIDOLOGY
DOACs surpass warfarin in low-weight AFib patients
SAN FRANCISCO – The direct-acting anticoagulants, as a class, were more effective and at least as safe as warfarin in low-weight and very-low-weight patients with atrial fibrillation in an adjusted analysis of real-world outcomes data from more than 21,000 Korean patients.
The analysis also showed that the direct-acting oral anticoagulants (DOACs) had the best safety and efficacy on low-weight patients when used at the labeled dosages, with blunted efficacy and safety at dosages that either exceeded or fell short of labeled levels, So-Ryoung Lee, MD, said at the annual scientific sessions of the Heart Rhythm Society.
The overall superiority of DOACs by both efficacy and safety also generally extended to the subgroup of very-low-weight patients, those with weights of less than 50 kg. In this subgroup, which was 28% of the total population studied, the composite adverse event outcome occurred 33% less often among patients treated with a DOAC relative to patients treated with warfarin, a statistically significant difference, said Dr. Lee, a cardiologist at Seoul (South Korea) National University Hospital. Among all patients with weights of 60 kg (132 pounds) or less, the composite outcome occurred 34% less often in the DOAC-treated patients relative to the warfarin-treated patients, also a statistically significant difference.
Dr. Lee and colleagues used a Korean National Health Insurance database that included information on more than 600,000 adults with atrial fibrillation (AFib) as of January 2013. The researchers whittled this down to 21,678 patients who began for the first time treatment with an oral anticoagulant starting during or after January 2014; had no history of a stroke, intracranial hemorrhage, or gastrointestinal bleed; and weighed no more than 60 kg. This cohort included 7,575 (35%) who received warfarin treatment, and 14,103 (65%) who received a DOAC. Within the DOAC-treated group, 42% received rivaroxaban (Xarelto), 26% dabigatran (Pradaxa), 24% apixaban (Eliquis), and 8% edoxaban (Savaysa).
To account for baseline differences in demographics and comorbidities between the patients treated with a DOAC and those who received warfarin, Dr. Lee and her associates did propensity score adjustment, which resulted in similar cohorts of 6,692 patients treated with warfarin and 12,810 patients treated with a DOAC. The average age of these patients was 73 years, a third were men, and the average body mass index was just over 22 kg/m2.
The events that the researchers tallied during follow-up through December 2016 included rates of all-cause death, ischemic stroke, intracranial hemorrhage, hospitalization for GI bleeding, hospitalization for major bleeding, and the composite of these five outcomes.
In the propensity-score adjusted full cohort of all patients who weighed 60 kg or less, the rate of each of these five outcomes, as well as the composite outcome, occurred statistically significantly less often among the DOAC-treated patients than in those on warfarin. The reductions ranged from a 41% lower incidence of ischemic stroke on DOAC treatment compared with warfarin treatment, to an 18% reduced rate of hospitalization for a GI bleed, compared with warfarin-treated patients. In the subgroup of patients who weighed less than 50 kg (110 pounds), the reductions ranged from a 41% cut in ischemic stroke on a DOAC compared with warfarin to a 24% relative reduction in the rate of hospitalization for a major bleed, a difference that just reached statistical significance. The outcome of hospitalization for a GI bleed showed no significant between-group difference among very-low-weight patients, but the rates of intracranial hemorrhage and all-cause death also showed statistically significant lower rates among DOAC-treated patients.
Nearly two-thirds of the patients on a DOAC received the label-appropriate dose of the drug, but 31% received a dosage that was below the labeled level while 4% received a dosage above the labeled level. An analysis that divided the NOAC patients by the appropriateness of their treatment dosages showed that patients on the correct dosages fared best. For example, in the total cohort of patients who weighed 60 kg or less, those on the correct DOAC dosage had a 9.1% rate of the combined endpoint. Patients on a low DOAC dosage did about as well as did the patients on warfarin, with a combined event rate of 11.6% in each of these subgroups. The worst outcomes occurred among the small number of patients on an inappropriately-high DOAC dosage, with a combined event rate of 15.4%. The researchers found a similar pattern among patients who weighed less than 50 kg.
Dr. Lee had no disclosures.
SOURCE: Lee SR et al. HRS 2019, Abstract S-AB30-05.
SAN FRANCISCO – The direct-acting anticoagulants, as a class, were more effective and at least as safe as warfarin in low-weight and very-low-weight patients with atrial fibrillation in an adjusted analysis of real-world outcomes data from more than 21,000 Korean patients.
The analysis also showed that the direct-acting oral anticoagulants (DOACs) had the best safety and efficacy on low-weight patients when used at the labeled dosages, with blunted efficacy and safety at dosages that either exceeded or fell short of labeled levels, So-Ryoung Lee, MD, said at the annual scientific sessions of the Heart Rhythm Society.
The overall superiority of DOACs by both efficacy and safety also generally extended to the subgroup of very-low-weight patients, those with weights of less than 50 kg. In this subgroup, which was 28% of the total population studied, the composite adverse event outcome occurred 33% less often among patients treated with a DOAC relative to patients treated with warfarin, a statistically significant difference, said Dr. Lee, a cardiologist at Seoul (South Korea) National University Hospital. Among all patients with weights of 60 kg (132 pounds) or less, the composite outcome occurred 34% less often in the DOAC-treated patients relative to the warfarin-treated patients, also a statistically significant difference.
Dr. Lee and colleagues used a Korean National Health Insurance database that included information on more than 600,000 adults with atrial fibrillation (AFib) as of January 2013. The researchers whittled this down to 21,678 patients who began for the first time treatment with an oral anticoagulant starting during or after January 2014; had no history of a stroke, intracranial hemorrhage, or gastrointestinal bleed; and weighed no more than 60 kg. This cohort included 7,575 (35%) who received warfarin treatment, and 14,103 (65%) who received a DOAC. Within the DOAC-treated group, 42% received rivaroxaban (Xarelto), 26% dabigatran (Pradaxa), 24% apixaban (Eliquis), and 8% edoxaban (Savaysa).
To account for baseline differences in demographics and comorbidities between the patients treated with a DOAC and those who received warfarin, Dr. Lee and her associates did propensity score adjustment, which resulted in similar cohorts of 6,692 patients treated with warfarin and 12,810 patients treated with a DOAC. The average age of these patients was 73 years, a third were men, and the average body mass index was just over 22 kg/m2.
The events that the researchers tallied during follow-up through December 2016 included rates of all-cause death, ischemic stroke, intracranial hemorrhage, hospitalization for GI bleeding, hospitalization for major bleeding, and the composite of these five outcomes.
In the propensity-score adjusted full cohort of all patients who weighed 60 kg or less, the rate of each of these five outcomes, as well as the composite outcome, occurred statistically significantly less often among the DOAC-treated patients than in those on warfarin. The reductions ranged from a 41% lower incidence of ischemic stroke on DOAC treatment compared with warfarin treatment, to an 18% reduced rate of hospitalization for a GI bleed, compared with warfarin-treated patients. In the subgroup of patients who weighed less than 50 kg (110 pounds), the reductions ranged from a 41% cut in ischemic stroke on a DOAC compared with warfarin to a 24% relative reduction in the rate of hospitalization for a major bleed, a difference that just reached statistical significance. The outcome of hospitalization for a GI bleed showed no significant between-group difference among very-low-weight patients, but the rates of intracranial hemorrhage and all-cause death also showed statistically significant lower rates among DOAC-treated patients.
Nearly two-thirds of the patients on a DOAC received the label-appropriate dose of the drug, but 31% received a dosage that was below the labeled level while 4% received a dosage above the labeled level. An analysis that divided the NOAC patients by the appropriateness of their treatment dosages showed that patients on the correct dosages fared best. For example, in the total cohort of patients who weighed 60 kg or less, those on the correct DOAC dosage had a 9.1% rate of the combined endpoint. Patients on a low DOAC dosage did about as well as did the patients on warfarin, with a combined event rate of 11.6% in each of these subgroups. The worst outcomes occurred among the small number of patients on an inappropriately-high DOAC dosage, with a combined event rate of 15.4%. The researchers found a similar pattern among patients who weighed less than 50 kg.
Dr. Lee had no disclosures.
SOURCE: Lee SR et al. HRS 2019, Abstract S-AB30-05.
SAN FRANCISCO – The direct-acting anticoagulants, as a class, were more effective and at least as safe as warfarin in low-weight and very-low-weight patients with atrial fibrillation in an adjusted analysis of real-world outcomes data from more than 21,000 Korean patients.
The analysis also showed that the direct-acting oral anticoagulants (DOACs) had the best safety and efficacy on low-weight patients when used at the labeled dosages, with blunted efficacy and safety at dosages that either exceeded or fell short of labeled levels, So-Ryoung Lee, MD, said at the annual scientific sessions of the Heart Rhythm Society.
The overall superiority of DOACs by both efficacy and safety also generally extended to the subgroup of very-low-weight patients, those with weights of less than 50 kg. In this subgroup, which was 28% of the total population studied, the composite adverse event outcome occurred 33% less often among patients treated with a DOAC relative to patients treated with warfarin, a statistically significant difference, said Dr. Lee, a cardiologist at Seoul (South Korea) National University Hospital. Among all patients with weights of 60 kg (132 pounds) or less, the composite outcome occurred 34% less often in the DOAC-treated patients relative to the warfarin-treated patients, also a statistically significant difference.
Dr. Lee and colleagues used a Korean National Health Insurance database that included information on more than 600,000 adults with atrial fibrillation (AFib) as of January 2013. The researchers whittled this down to 21,678 patients who began for the first time treatment with an oral anticoagulant starting during or after January 2014; had no history of a stroke, intracranial hemorrhage, or gastrointestinal bleed; and weighed no more than 60 kg. This cohort included 7,575 (35%) who received warfarin treatment, and 14,103 (65%) who received a DOAC. Within the DOAC-treated group, 42% received rivaroxaban (Xarelto), 26% dabigatran (Pradaxa), 24% apixaban (Eliquis), and 8% edoxaban (Savaysa).
To account for baseline differences in demographics and comorbidities between the patients treated with a DOAC and those who received warfarin, Dr. Lee and her associates did propensity score adjustment, which resulted in similar cohorts of 6,692 patients treated with warfarin and 12,810 patients treated with a DOAC. The average age of these patients was 73 years, a third were men, and the average body mass index was just over 22 kg/m2.
The events that the researchers tallied during follow-up through December 2016 included rates of all-cause death, ischemic stroke, intracranial hemorrhage, hospitalization for GI bleeding, hospitalization for major bleeding, and the composite of these five outcomes.
In the propensity-score adjusted full cohort of all patients who weighed 60 kg or less, the rate of each of these five outcomes, as well as the composite outcome, occurred statistically significantly less often among the DOAC-treated patients than in those on warfarin. The reductions ranged from a 41% lower incidence of ischemic stroke on DOAC treatment compared with warfarin treatment, to an 18% reduced rate of hospitalization for a GI bleed, compared with warfarin-treated patients. In the subgroup of patients who weighed less than 50 kg (110 pounds), the reductions ranged from a 41% cut in ischemic stroke on a DOAC compared with warfarin to a 24% relative reduction in the rate of hospitalization for a major bleed, a difference that just reached statistical significance. The outcome of hospitalization for a GI bleed showed no significant between-group difference among very-low-weight patients, but the rates of intracranial hemorrhage and all-cause death also showed statistically significant lower rates among DOAC-treated patients.
Nearly two-thirds of the patients on a DOAC received the label-appropriate dose of the drug, but 31% received a dosage that was below the labeled level while 4% received a dosage above the labeled level. An analysis that divided the NOAC patients by the appropriateness of their treatment dosages showed that patients on the correct dosages fared best. For example, in the total cohort of patients who weighed 60 kg or less, those on the correct DOAC dosage had a 9.1% rate of the combined endpoint. Patients on a low DOAC dosage did about as well as did the patients on warfarin, with a combined event rate of 11.6% in each of these subgroups. The worst outcomes occurred among the small number of patients on an inappropriately-high DOAC dosage, with a combined event rate of 15.4%. The researchers found a similar pattern among patients who weighed less than 50 kg.
Dr. Lee had no disclosures.
SOURCE: Lee SR et al. HRS 2019, Abstract S-AB30-05.
REPORTING FROM HEART RHYTHM 2019
Tick-borne disease has become a national issue
Pennsylvania had more reported cases of tick-borne disease from 2004 to 2016 than any other state, but these diseases are becoming a national threat, according to the Centers for Disease Control and Prevention.
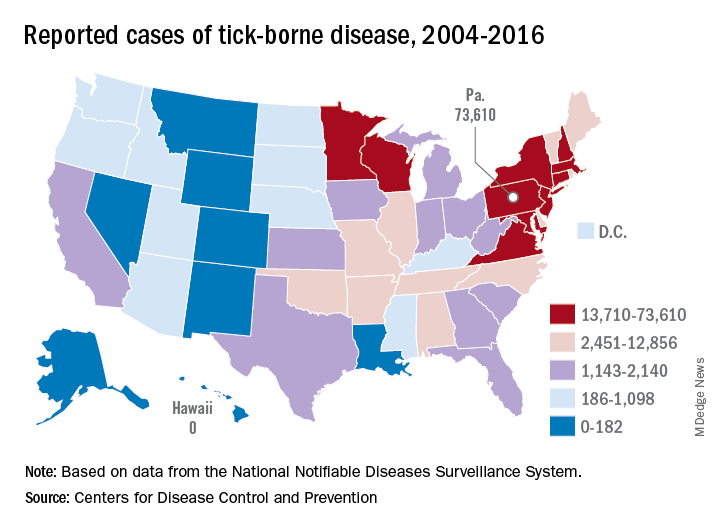
There were 73,000 cases reported in Pennsylvania over that period, and tick-borne diseases, including Lyme disease, anaplasmosis/ehrlichiosis, spotted fever rickettsiosis, babesiosis, tularemia, and Powassan virus, among others, affected almost 492,000 people nationwide, with Lyme disease representing the majority of cases, the CDC said in a Vital Signs report.
Although it’s no surprise that Pennsylvania, New York, and Connecticut were tick-borne disease hot spots, non-Northeastern states like Virginia, Wisconsin, and Minnesota also were among the top 10 in cases. States even further away from the Northeast can be found in the next 10: Arkansas had more than 7,000 cases in 13 years, and Oklahoma had over 4,600 cases, data from the National Notifiable Diseases Surveillance System show.
Nationally, the number of cases more than doubled from 23,000 in 2004 to 49,000 in 2016, and tick-borne disease hit every state except Hawaii. Over that same time, seven new tick-borne pathogens were discovered or introduced into the United States, the CDC reported.
“Local and state health departments and vector control organizations face increasing demands to respond to these threats,” the CDC said, but “more than 80% of vector control organizations report needing improvement in one or more of five core competencies, such as testing for pesticide resistance [and using] data to drive local decisions about vector control.”
Pennsylvania had more reported cases of tick-borne disease from 2004 to 2016 than any other state, but these diseases are becoming a national threat, according to the Centers for Disease Control and Prevention.

There were 73,000 cases reported in Pennsylvania over that period, and tick-borne diseases, including Lyme disease, anaplasmosis/ehrlichiosis, spotted fever rickettsiosis, babesiosis, tularemia, and Powassan virus, among others, affected almost 492,000 people nationwide, with Lyme disease representing the majority of cases, the CDC said in a Vital Signs report.
Although it’s no surprise that Pennsylvania, New York, and Connecticut were tick-borne disease hot spots, non-Northeastern states like Virginia, Wisconsin, and Minnesota also were among the top 10 in cases. States even further away from the Northeast can be found in the next 10: Arkansas had more than 7,000 cases in 13 years, and Oklahoma had over 4,600 cases, data from the National Notifiable Diseases Surveillance System show.
Nationally, the number of cases more than doubled from 23,000 in 2004 to 49,000 in 2016, and tick-borne disease hit every state except Hawaii. Over that same time, seven new tick-borne pathogens were discovered or introduced into the United States, the CDC reported.
“Local and state health departments and vector control organizations face increasing demands to respond to these threats,” the CDC said, but “more than 80% of vector control organizations report needing improvement in one or more of five core competencies, such as testing for pesticide resistance [and using] data to drive local decisions about vector control.”
Pennsylvania had more reported cases of tick-borne disease from 2004 to 2016 than any other state, but these diseases are becoming a national threat, according to the Centers for Disease Control and Prevention.

There were 73,000 cases reported in Pennsylvania over that period, and tick-borne diseases, including Lyme disease, anaplasmosis/ehrlichiosis, spotted fever rickettsiosis, babesiosis, tularemia, and Powassan virus, among others, affected almost 492,000 people nationwide, with Lyme disease representing the majority of cases, the CDC said in a Vital Signs report.
Although it’s no surprise that Pennsylvania, New York, and Connecticut were tick-borne disease hot spots, non-Northeastern states like Virginia, Wisconsin, and Minnesota also were among the top 10 in cases. States even further away from the Northeast can be found in the next 10: Arkansas had more than 7,000 cases in 13 years, and Oklahoma had over 4,600 cases, data from the National Notifiable Diseases Surveillance System show.
Nationally, the number of cases more than doubled from 23,000 in 2004 to 49,000 in 2016, and tick-borne disease hit every state except Hawaii. Over that same time, seven new tick-borne pathogens were discovered or introduced into the United States, the CDC reported.
“Local and state health departments and vector control organizations face increasing demands to respond to these threats,” the CDC said, but “more than 80% of vector control organizations report needing improvement in one or more of five core competencies, such as testing for pesticide resistance [and using] data to drive local decisions about vector control.”
A warning song to keep our children safe
Pay heed to “The House of the Rising Sun”
“There is a house in New Orleans. They call the Rising Sun. And it’s been the ruin of many a poor boy. And, God, I know I’m one.”
The 1960s rock band the Animals will tell you a tale to convince you to get vaccinated. Don’t believe me? Follow along.
The first hints of the song “House of the Rising Sun” rolled out of the hills of Appalachia.
Somewhere in the Golden Triangle, far away from New Orleans, where Virginia, Kentucky, and Tennessee rise in quiet desolation, a warning song about a tailor and a drunk emerged. Sometime around the Civil War, a hint of a tune began. Over the next century, it evolved, until it became cemented in rock culture 50 years ago by The Animals, existing as the version played most commonly today.
In the mid-19th century, medicine shows rambled through the South, stopping in places like Noetown or Daisy. The small towns would empty out for the day to see the entertainers, singers, and jugglers perform. Hundreds gathered in the hot summer day, the entertainment solely a pretext for the traveling doctors to sell their wares, the snake oil, and cure-alls, as well as various patent medicines.
These were isolated towns, with no deliveries, few visitors, and the railroad yet to arrive. Frequently, the only news from outside came from these caravans of entertainers and con men who swept into town. They were like Professor Marvel from The Wizard of Oz, or a current-day Dr. Oz, luring the crowd with false advertising, selling colored water, and then disappearing before you realized you were duped. Today, traveling doctors of the same ilk convince parents to not vaccinate their children, tell them to visit stem cell centers that claim false cures, and offer them a shiny object with one hand while taking their cash with the other.
Yet, there was a positive development in the wake of these patent medicine shows: the entertainment lingered. New songs traveled the same journeys as these medicine shows – new earworms that would then be warbled in the local bars, while doing chores around the barn, or simply during walks on the Appalachian trails.
In 1937, Alan Lomax arrived in Noetown, Ky., with a microphone and an acetate record and recorded the voice of 16-year-old Georgia Turner singing “House of the Rising Sun.” She didn’t know where she heard that song, but most likely picked it up at the medicine show.
One of those singers was Clarence Ashley, who would croon about the Rising Sun Blues. He sang with Doc Cloud and Doc Hauer, who offered tonics for whatever ailed you. Perhaps Georgia Turner heard the song in the early 1900s as well. Her 1937 version contains the lyrics most closely related to the Animals’ tune.
Lomax spent the 1940s gathering songs around the Appalachian South. He put these songs into a songbook and spread them throughout the country. He would also return to New York City and gather in a room with legendary folk singers. They would hear these new lyrics, new sounds, and make them their own.
In that room would be Lead Belly, Pete Seeger, Woody Guthrie, and Josh White, the fathers of folk music. The music Lomax pulled out of the mountains in small towns would become new again in the guitars and harmonicas of the Greenwich Village singers and musicians. Pete Seeger performed with the Weavers, named because they would weave songs from the past into new versions.
“House of the Rising Sun” was woven into the folk music landscape, evolving and growing. Josh White is credited with changing the song from a major key into the minor key we know today. Bob Dylan sang a version. And then in 1964, Eric Burdon and The Animals released their version, which became the standard. An arpeggio guitar opening, the rhythm sped up, a louder sound, and that minor key provides an emotional wallop for this warning song.
Numerous covers followed, including a beautiful version of “Amazing Grace”, sung to the tune of “House of the Rising Sun” by the Blind Boys of Alabama.
The song endures for its melody as well as for its lyrics. This was a warning song, a universal song, “not to do what I have done.” The small towns in Kentucky may have heard of the sinful ways of New Orleans and would spread the message with these songs to avoid the brothels, the drink, and the broken marriages that would reverberate with visits to the Crescent City.
“House of the Rising Sun” is one of the most covered songs, traveling wide and far, no longer with the need for a medicine show. It was a pivotal moment in rock ‘n roll, turning folk music into rock music. The Animals became huge because of this song, and their version became the standard on which all subsequent covers based their version. It made Bob Dylan’s older version seem quaint.
The song has been in my head for a while now. My wife is hoping writing about it will keep it from being played in our household any more. There are various reasons it has been resonating with me, including the following:
- It traces the origins of folk music and the importance of people like Lomax and Guthrie to collect and save Americana.
- The magic of musical evolution – a reminder of how art is built on the work of those who came before, each version with its unique personality.
- The release of “House of the Rising Sun” was a seminal, transformative moment when folk became rock music.
- The lasting power of warning songs.
- The hucksters that enabled this song to be kept alive.
That last one has really stuck with me. The medicine shows are an important part of American history. For instance, Coca-Cola started as one of those patent medicines; it was one of the many concoctions of the Atlanta pharmacist John Stith Pemberton, sold to treat all that ails us. Dr. Pepper, too, was a medicine in a sugary bottle – another that often contained alcohol or cocaine. Society wants a cure-all, and the marketing and selling done during these medicine shows offered placebos.
The hucksters exist in various forms today, selling detoxifications, magic diet cures, psychic powers of healing, or convincing parents that their kids don’t need vaccines. We need a warning song that goes viral to keep our children safe. We are blessed to be in a world without smallpox, almost rid of polio, and we have the knowledge and opportunity to rid the world of other preventable illnesses. Measles was declared eliminated in the United States in 2000; now, outbreaks emerge in every news cycle.
The CDC admits they have not been targeting misinformation well. How can we spread the science, the truth, the message faster than the lies? Better marketing? The answer may be through stories and narratives and song, with the backing of good science. “House of the Rising Sun” is a warning song. Maybe we need more. We need that deep history, that long trail to remind us of the world before vaccines, when everyone knew someone, either in their own household or next door, who succumbed to one of the childhood illnesses.
Let the “House of the Rising Sun” play on. Create a new version, and let that message reverberate, too.
Tell your children; they need to be vaccinated.
Dr. Messler is a hospitalist at Morton Plant Hospitalist group in Clearwater, Fla. He previously chaired SHM’s Quality and Patient Safety Committee and has been active in several SHM mentoring programs, most recently with Project BOOST and Glycemic Control. This article appeared originally in SHM's official blog The Hospital Leader. Read more recent posts here.
Pay heed to “The House of the Rising Sun”
Pay heed to “The House of the Rising Sun”
“There is a house in New Orleans. They call the Rising Sun. And it’s been the ruin of many a poor boy. And, God, I know I’m one.”
The 1960s rock band the Animals will tell you a tale to convince you to get vaccinated. Don’t believe me? Follow along.
The first hints of the song “House of the Rising Sun” rolled out of the hills of Appalachia.
Somewhere in the Golden Triangle, far away from New Orleans, where Virginia, Kentucky, and Tennessee rise in quiet desolation, a warning song about a tailor and a drunk emerged. Sometime around the Civil War, a hint of a tune began. Over the next century, it evolved, until it became cemented in rock culture 50 years ago by The Animals, existing as the version played most commonly today.
In the mid-19th century, medicine shows rambled through the South, stopping in places like Noetown or Daisy. The small towns would empty out for the day to see the entertainers, singers, and jugglers perform. Hundreds gathered in the hot summer day, the entertainment solely a pretext for the traveling doctors to sell their wares, the snake oil, and cure-alls, as well as various patent medicines.
These were isolated towns, with no deliveries, few visitors, and the railroad yet to arrive. Frequently, the only news from outside came from these caravans of entertainers and con men who swept into town. They were like Professor Marvel from The Wizard of Oz, or a current-day Dr. Oz, luring the crowd with false advertising, selling colored water, and then disappearing before you realized you were duped. Today, traveling doctors of the same ilk convince parents to not vaccinate their children, tell them to visit stem cell centers that claim false cures, and offer them a shiny object with one hand while taking their cash with the other.
Yet, there was a positive development in the wake of these patent medicine shows: the entertainment lingered. New songs traveled the same journeys as these medicine shows – new earworms that would then be warbled in the local bars, while doing chores around the barn, or simply during walks on the Appalachian trails.
In 1937, Alan Lomax arrived in Noetown, Ky., with a microphone and an acetate record and recorded the voice of 16-year-old Georgia Turner singing “House of the Rising Sun.” She didn’t know where she heard that song, but most likely picked it up at the medicine show.
One of those singers was Clarence Ashley, who would croon about the Rising Sun Blues. He sang with Doc Cloud and Doc Hauer, who offered tonics for whatever ailed you. Perhaps Georgia Turner heard the song in the early 1900s as well. Her 1937 version contains the lyrics most closely related to the Animals’ tune.
Lomax spent the 1940s gathering songs around the Appalachian South. He put these songs into a songbook and spread them throughout the country. He would also return to New York City and gather in a room with legendary folk singers. They would hear these new lyrics, new sounds, and make them their own.
In that room would be Lead Belly, Pete Seeger, Woody Guthrie, and Josh White, the fathers of folk music. The music Lomax pulled out of the mountains in small towns would become new again in the guitars and harmonicas of the Greenwich Village singers and musicians. Pete Seeger performed with the Weavers, named because they would weave songs from the past into new versions.
“House of the Rising Sun” was woven into the folk music landscape, evolving and growing. Josh White is credited with changing the song from a major key into the minor key we know today. Bob Dylan sang a version. And then in 1964, Eric Burdon and The Animals released their version, which became the standard. An arpeggio guitar opening, the rhythm sped up, a louder sound, and that minor key provides an emotional wallop for this warning song.
Numerous covers followed, including a beautiful version of “Amazing Grace”, sung to the tune of “House of the Rising Sun” by the Blind Boys of Alabama.
The song endures for its melody as well as for its lyrics. This was a warning song, a universal song, “not to do what I have done.” The small towns in Kentucky may have heard of the sinful ways of New Orleans and would spread the message with these songs to avoid the brothels, the drink, and the broken marriages that would reverberate with visits to the Crescent City.
“House of the Rising Sun” is one of the most covered songs, traveling wide and far, no longer with the need for a medicine show. It was a pivotal moment in rock ‘n roll, turning folk music into rock music. The Animals became huge because of this song, and their version became the standard on which all subsequent covers based their version. It made Bob Dylan’s older version seem quaint.
The song has been in my head for a while now. My wife is hoping writing about it will keep it from being played in our household any more. There are various reasons it has been resonating with me, including the following:
- It traces the origins of folk music and the importance of people like Lomax and Guthrie to collect and save Americana.
- The magic of musical evolution – a reminder of how art is built on the work of those who came before, each version with its unique personality.
- The release of “House of the Rising Sun” was a seminal, transformative moment when folk became rock music.
- The lasting power of warning songs.
- The hucksters that enabled this song to be kept alive.
That last one has really stuck with me. The medicine shows are an important part of American history. For instance, Coca-Cola started as one of those patent medicines; it was one of the many concoctions of the Atlanta pharmacist John Stith Pemberton, sold to treat all that ails us. Dr. Pepper, too, was a medicine in a sugary bottle – another that often contained alcohol or cocaine. Society wants a cure-all, and the marketing and selling done during these medicine shows offered placebos.
The hucksters exist in various forms today, selling detoxifications, magic diet cures, psychic powers of healing, or convincing parents that their kids don’t need vaccines. We need a warning song that goes viral to keep our children safe. We are blessed to be in a world without smallpox, almost rid of polio, and we have the knowledge and opportunity to rid the world of other preventable illnesses. Measles was declared eliminated in the United States in 2000; now, outbreaks emerge in every news cycle.
The CDC admits they have not been targeting misinformation well. How can we spread the science, the truth, the message faster than the lies? Better marketing? The answer may be through stories and narratives and song, with the backing of good science. “House of the Rising Sun” is a warning song. Maybe we need more. We need that deep history, that long trail to remind us of the world before vaccines, when everyone knew someone, either in their own household or next door, who succumbed to one of the childhood illnesses.
Let the “House of the Rising Sun” play on. Create a new version, and let that message reverberate, too.
Tell your children; they need to be vaccinated.
Dr. Messler is a hospitalist at Morton Plant Hospitalist group in Clearwater, Fla. He previously chaired SHM’s Quality and Patient Safety Committee and has been active in several SHM mentoring programs, most recently with Project BOOST and Glycemic Control. This article appeared originally in SHM's official blog The Hospital Leader. Read more recent posts here.
“There is a house in New Orleans. They call the Rising Sun. And it’s been the ruin of many a poor boy. And, God, I know I’m one.”
The 1960s rock band the Animals will tell you a tale to convince you to get vaccinated. Don’t believe me? Follow along.
The first hints of the song “House of the Rising Sun” rolled out of the hills of Appalachia.
Somewhere in the Golden Triangle, far away from New Orleans, where Virginia, Kentucky, and Tennessee rise in quiet desolation, a warning song about a tailor and a drunk emerged. Sometime around the Civil War, a hint of a tune began. Over the next century, it evolved, until it became cemented in rock culture 50 years ago by The Animals, existing as the version played most commonly today.
In the mid-19th century, medicine shows rambled through the South, stopping in places like Noetown or Daisy. The small towns would empty out for the day to see the entertainers, singers, and jugglers perform. Hundreds gathered in the hot summer day, the entertainment solely a pretext for the traveling doctors to sell their wares, the snake oil, and cure-alls, as well as various patent medicines.
These were isolated towns, with no deliveries, few visitors, and the railroad yet to arrive. Frequently, the only news from outside came from these caravans of entertainers and con men who swept into town. They were like Professor Marvel from The Wizard of Oz, or a current-day Dr. Oz, luring the crowd with false advertising, selling colored water, and then disappearing before you realized you were duped. Today, traveling doctors of the same ilk convince parents to not vaccinate their children, tell them to visit stem cell centers that claim false cures, and offer them a shiny object with one hand while taking their cash with the other.
Yet, there was a positive development in the wake of these patent medicine shows: the entertainment lingered. New songs traveled the same journeys as these medicine shows – new earworms that would then be warbled in the local bars, while doing chores around the barn, or simply during walks on the Appalachian trails.
In 1937, Alan Lomax arrived in Noetown, Ky., with a microphone and an acetate record and recorded the voice of 16-year-old Georgia Turner singing “House of the Rising Sun.” She didn’t know where she heard that song, but most likely picked it up at the medicine show.
One of those singers was Clarence Ashley, who would croon about the Rising Sun Blues. He sang with Doc Cloud and Doc Hauer, who offered tonics for whatever ailed you. Perhaps Georgia Turner heard the song in the early 1900s as well. Her 1937 version contains the lyrics most closely related to the Animals’ tune.
Lomax spent the 1940s gathering songs around the Appalachian South. He put these songs into a songbook and spread them throughout the country. He would also return to New York City and gather in a room with legendary folk singers. They would hear these new lyrics, new sounds, and make them their own.
In that room would be Lead Belly, Pete Seeger, Woody Guthrie, and Josh White, the fathers of folk music. The music Lomax pulled out of the mountains in small towns would become new again in the guitars and harmonicas of the Greenwich Village singers and musicians. Pete Seeger performed with the Weavers, named because they would weave songs from the past into new versions.
“House of the Rising Sun” was woven into the folk music landscape, evolving and growing. Josh White is credited with changing the song from a major key into the minor key we know today. Bob Dylan sang a version. And then in 1964, Eric Burdon and The Animals released their version, which became the standard. An arpeggio guitar opening, the rhythm sped up, a louder sound, and that minor key provides an emotional wallop for this warning song.
Numerous covers followed, including a beautiful version of “Amazing Grace”, sung to the tune of “House of the Rising Sun” by the Blind Boys of Alabama.
The song endures for its melody as well as for its lyrics. This was a warning song, a universal song, “not to do what I have done.” The small towns in Kentucky may have heard of the sinful ways of New Orleans and would spread the message with these songs to avoid the brothels, the drink, and the broken marriages that would reverberate with visits to the Crescent City.
“House of the Rising Sun” is one of the most covered songs, traveling wide and far, no longer with the need for a medicine show. It was a pivotal moment in rock ‘n roll, turning folk music into rock music. The Animals became huge because of this song, and their version became the standard on which all subsequent covers based their version. It made Bob Dylan’s older version seem quaint.
The song has been in my head for a while now. My wife is hoping writing about it will keep it from being played in our household any more. There are various reasons it has been resonating with me, including the following:
- It traces the origins of folk music and the importance of people like Lomax and Guthrie to collect and save Americana.
- The magic of musical evolution – a reminder of how art is built on the work of those who came before, each version with its unique personality.
- The release of “House of the Rising Sun” was a seminal, transformative moment when folk became rock music.
- The lasting power of warning songs.
- The hucksters that enabled this song to be kept alive.
That last one has really stuck with me. The medicine shows are an important part of American history. For instance, Coca-Cola started as one of those patent medicines; it was one of the many concoctions of the Atlanta pharmacist John Stith Pemberton, sold to treat all that ails us. Dr. Pepper, too, was a medicine in a sugary bottle – another that often contained alcohol or cocaine. Society wants a cure-all, and the marketing and selling done during these medicine shows offered placebos.
The hucksters exist in various forms today, selling detoxifications, magic diet cures, psychic powers of healing, or convincing parents that their kids don’t need vaccines. We need a warning song that goes viral to keep our children safe. We are blessed to be in a world without smallpox, almost rid of polio, and we have the knowledge and opportunity to rid the world of other preventable illnesses. Measles was declared eliminated in the United States in 2000; now, outbreaks emerge in every news cycle.
The CDC admits they have not been targeting misinformation well. How can we spread the science, the truth, the message faster than the lies? Better marketing? The answer may be through stories and narratives and song, with the backing of good science. “House of the Rising Sun” is a warning song. Maybe we need more. We need that deep history, that long trail to remind us of the world before vaccines, when everyone knew someone, either in their own household or next door, who succumbed to one of the childhood illnesses.
Let the “House of the Rising Sun” play on. Create a new version, and let that message reverberate, too.
Tell your children; they need to be vaccinated.
Dr. Messler is a hospitalist at Morton Plant Hospitalist group in Clearwater, Fla. He previously chaired SHM’s Quality and Patient Safety Committee and has been active in several SHM mentoring programs, most recently with Project BOOST and Glycemic Control. This article appeared originally in SHM's official blog The Hospital Leader. Read more recent posts here.
FDA approves new treatment for hospital-acquired, ventilator-associated bacterial pneumonia
authorizing it for the treatment of both hospital-acquired and ventilator-associated bacterial pneumonia.
The new indication is for patients 18 years and older. It was based on results of a multinational, double-blind study that compared Zerbaxa with a different antibacterial drug in 726 patients hospitalized with hospital-acquired/ventilator-associated bacterial pneumonia. Mortality and cure rates were similar in the Zerbaxa and comparator groups.
The most common adverse events observed in the trial were elevated liver enzyme levels, renal impairment or failure, and diarrhea. Patients with hypersensitivity to beta-lactam drugs should not be receive Zerbaxa.
“A key global challenge we face as a public health agency is addressing the threat of antimicrobial-resistant infections. Hospital-acquired and ventilator-associated bacterial pneumonia are serious infections that can result in death in some patients. ... That’s why, among our other efforts to address antimicrobial resistance, we’re focused on facilitating the development of safe and effective new treatments to give patients more options to fight life-threatening infections,” said Amy Abernethy, MD, PhD, the FDA’s principal deputy commissioner.
Zerbaxa was initially approved in 2014 for treatment of complicated intra-abdominal and urinary tract infections.
Find the full press release on the FDA website.
authorizing it for the treatment of both hospital-acquired and ventilator-associated bacterial pneumonia.
The new indication is for patients 18 years and older. It was based on results of a multinational, double-blind study that compared Zerbaxa with a different antibacterial drug in 726 patients hospitalized with hospital-acquired/ventilator-associated bacterial pneumonia. Mortality and cure rates were similar in the Zerbaxa and comparator groups.
The most common adverse events observed in the trial were elevated liver enzyme levels, renal impairment or failure, and diarrhea. Patients with hypersensitivity to beta-lactam drugs should not be receive Zerbaxa.
“A key global challenge we face as a public health agency is addressing the threat of antimicrobial-resistant infections. Hospital-acquired and ventilator-associated bacterial pneumonia are serious infections that can result in death in some patients. ... That’s why, among our other efforts to address antimicrobial resistance, we’re focused on facilitating the development of safe and effective new treatments to give patients more options to fight life-threatening infections,” said Amy Abernethy, MD, PhD, the FDA’s principal deputy commissioner.
Zerbaxa was initially approved in 2014 for treatment of complicated intra-abdominal and urinary tract infections.
Find the full press release on the FDA website.
authorizing it for the treatment of both hospital-acquired and ventilator-associated bacterial pneumonia.
The new indication is for patients 18 years and older. It was based on results of a multinational, double-blind study that compared Zerbaxa with a different antibacterial drug in 726 patients hospitalized with hospital-acquired/ventilator-associated bacterial pneumonia. Mortality and cure rates were similar in the Zerbaxa and comparator groups.
The most common adverse events observed in the trial were elevated liver enzyme levels, renal impairment or failure, and diarrhea. Patients with hypersensitivity to beta-lactam drugs should not be receive Zerbaxa.
“A key global challenge we face as a public health agency is addressing the threat of antimicrobial-resistant infections. Hospital-acquired and ventilator-associated bacterial pneumonia are serious infections that can result in death in some patients. ... That’s why, among our other efforts to address antimicrobial resistance, we’re focused on facilitating the development of safe and effective new treatments to give patients more options to fight life-threatening infections,” said Amy Abernethy, MD, PhD, the FDA’s principal deputy commissioner.
Zerbaxa was initially approved in 2014 for treatment of complicated intra-abdominal and urinary tract infections.
Find the full press release on the FDA website.
Why you should re-credential with Medicare as a hospitalist
CMS needs a better database of hospitalist information
In April 2017, the Centers for Medicare and Medicaid Services implemented the new physician specialty code C6, specifically for hospitalists. There has been a lot of confusion about what this means and some uncertainty about why clinicians should bother to use it.
Some folks thought initially that it was a new CPT code they could use to bill hospitalist services, which might recognize the increased intensity of services hospitalists often provide to their hospitalized patients compared to many traditional internal medicine and family medicine primary care physicians. Others thought it was a code that was added to the HCFA 1500 billing form somewhere to designate that the service was provided by a hospitalist.
Neither is true. The C6 physician specialty code is one of a large number of such codes used by physicians to designate their primary physician specialty when they enroll with Medicare via the PECOS online enrollment system. It describes the unique type of medicine practiced by the enrolling physician and is used by the CMS both for claims processing purposes and for “programmatic” purposes (whatever that means).
It doesn’t change how your claim is processed or how much you get paid. So why bother going through the laborious process of re-credentialing with CMS via PECOS just to change your specialty code? Well, I believe there are several ways in which the C6 specialty code provides value – both to you and to the specialty of hospital medicine.
Reduce concurrent care denials
First, it distinguishes you from a general internal medicine or general family medicine practitioner by recognizing “hospitalist” as a distinct specialty. This can be valuable from a financial perspective because it may reduce the risk that claims for your services might be denied due to “concurrent care” by another provider in the same specialty on the same calendar day.
And it’s not just a general internist or family medicine physician that you might run into concurrent care trouble with. I’ve seen situations where doctors completed critical care or cardiology fellowships but never got around to re-credentialing with Medicare in their new specialty, so their claims still showed up with an “internal medicine” physician specialty code, resulting in denied “concurrent care” claims for either the hospitalist or the specialist.
While Medicare may still see unnecessary overlap between services provided by you and an internal medicine or family physician to the same patient on the same calendar day, you can make a better argument that your services were unique and complementary to (not duplicative of) the services of others if you are credentialed as a hospitalist.
Ensure “apples to apples” comparisons
A second reason to re-credential as a hospitalist is to ensure that when the CMS looks at the services you are providing and the CPT codes you are selecting, it is comparing you to an appropriate peer group for compliance purposes.
The mix of CPT codes reported by hospitalists in the SHM State of Hospital Medicine Survey has historically tilted toward higher-level care than has the mix of CPT codes reported by the CMS for internal medicine or family medicine physicians. But last year when Medicare released the utilization of evaluation and management services by specialty for calendar year 2017, CPT utilization was shown separately for hospitalists for the first time!
The volume of services reported for physicians credentialed as hospitalists was very small relative to the volume of inpatient services provided by internal medicine and family medicine physicians, but the distribution of inpatient admission, subsequent visit, and discharge codes for hospitalists closely mirrored those reported by SHM in its 2018 State of Hospital Medicine Report (see graphic).
If you’re going to be targeted in a RAC audit for the high proportion of 99233s you bill, you want to be sure the CMS is looking at your performance compared to those who are truly your peers, caring for patients of the same type and complexity.
Improve CMS data used for research purposes
Finally, the ability of academic hospitalists and other health services researchers to utilize Medicare claims data to better understand the care provided by hospitalists and its impact on the overall health care system will be significantly enhanced by a more robust presence of physicians who have identified themselves as hospitalists in the PECOS credentialing system.
We care for the majority of patients in most hospitals these days, yet “hospitalists” billed only 2,009,869 inpatient subsequent visits (CPT codes 99231, 99232, and 99233) in 2017 compared to 25,903,829 billed by internal medicine physicians and 4,678,111 billed by family medicine physicians. And regardless of what you think about using claims data as a proxy for health care services and quality, it’s undeniably the best data set we currently have.
So, let’s work together to build a bigger, better database of hospitalist information at the CMS. I urge you to go to your credentialing folks today and find out how you can work with them to get yourself re-credentialed in PECOS using the C6 “hospitalist” physician specialty.
Ms. Flores is a partner at Nelson Flores Hospital Medicine Consultants, La Quinta, Calif. She serves on SHM’s Practice Analysis and Annual Meeting Committees, and helps to coordinate SHM’s bi-annual State of Hospital Medicine Survey. This article appeared originally in SHM's official blog The Hospital Leader. Read more recent posts here.
CMS needs a better database of hospitalist information
CMS needs a better database of hospitalist information
In April 2017, the Centers for Medicare and Medicaid Services implemented the new physician specialty code C6, specifically for hospitalists. There has been a lot of confusion about what this means and some uncertainty about why clinicians should bother to use it.
Some folks thought initially that it was a new CPT code they could use to bill hospitalist services, which might recognize the increased intensity of services hospitalists often provide to their hospitalized patients compared to many traditional internal medicine and family medicine primary care physicians. Others thought it was a code that was added to the HCFA 1500 billing form somewhere to designate that the service was provided by a hospitalist.
Neither is true. The C6 physician specialty code is one of a large number of such codes used by physicians to designate their primary physician specialty when they enroll with Medicare via the PECOS online enrollment system. It describes the unique type of medicine practiced by the enrolling physician and is used by the CMS both for claims processing purposes and for “programmatic” purposes (whatever that means).
It doesn’t change how your claim is processed or how much you get paid. So why bother going through the laborious process of re-credentialing with CMS via PECOS just to change your specialty code? Well, I believe there are several ways in which the C6 specialty code provides value – both to you and to the specialty of hospital medicine.
Reduce concurrent care denials
First, it distinguishes you from a general internal medicine or general family medicine practitioner by recognizing “hospitalist” as a distinct specialty. This can be valuable from a financial perspective because it may reduce the risk that claims for your services might be denied due to “concurrent care” by another provider in the same specialty on the same calendar day.
And it’s not just a general internist or family medicine physician that you might run into concurrent care trouble with. I’ve seen situations where doctors completed critical care or cardiology fellowships but never got around to re-credentialing with Medicare in their new specialty, so their claims still showed up with an “internal medicine” physician specialty code, resulting in denied “concurrent care” claims for either the hospitalist or the specialist.
While Medicare may still see unnecessary overlap between services provided by you and an internal medicine or family physician to the same patient on the same calendar day, you can make a better argument that your services were unique and complementary to (not duplicative of) the services of others if you are credentialed as a hospitalist.
Ensure “apples to apples” comparisons
A second reason to re-credential as a hospitalist is to ensure that when the CMS looks at the services you are providing and the CPT codes you are selecting, it is comparing you to an appropriate peer group for compliance purposes.
The mix of CPT codes reported by hospitalists in the SHM State of Hospital Medicine Survey has historically tilted toward higher-level care than has the mix of CPT codes reported by the CMS for internal medicine or family medicine physicians. But last year when Medicare released the utilization of evaluation and management services by specialty for calendar year 2017, CPT utilization was shown separately for hospitalists for the first time!
The volume of services reported for physicians credentialed as hospitalists was very small relative to the volume of inpatient services provided by internal medicine and family medicine physicians, but the distribution of inpatient admission, subsequent visit, and discharge codes for hospitalists closely mirrored those reported by SHM in its 2018 State of Hospital Medicine Report (see graphic).
If you’re going to be targeted in a RAC audit for the high proportion of 99233s you bill, you want to be sure the CMS is looking at your performance compared to those who are truly your peers, caring for patients of the same type and complexity.
Improve CMS data used for research purposes
Finally, the ability of academic hospitalists and other health services researchers to utilize Medicare claims data to better understand the care provided by hospitalists and its impact on the overall health care system will be significantly enhanced by a more robust presence of physicians who have identified themselves as hospitalists in the PECOS credentialing system.
We care for the majority of patients in most hospitals these days, yet “hospitalists” billed only 2,009,869 inpatient subsequent visits (CPT codes 99231, 99232, and 99233) in 2017 compared to 25,903,829 billed by internal medicine physicians and 4,678,111 billed by family medicine physicians. And regardless of what you think about using claims data as a proxy for health care services and quality, it’s undeniably the best data set we currently have.
So, let’s work together to build a bigger, better database of hospitalist information at the CMS. I urge you to go to your credentialing folks today and find out how you can work with them to get yourself re-credentialed in PECOS using the C6 “hospitalist” physician specialty.
Ms. Flores is a partner at Nelson Flores Hospital Medicine Consultants, La Quinta, Calif. She serves on SHM’s Practice Analysis and Annual Meeting Committees, and helps to coordinate SHM’s bi-annual State of Hospital Medicine Survey. This article appeared originally in SHM's official blog The Hospital Leader. Read more recent posts here.
In April 2017, the Centers for Medicare and Medicaid Services implemented the new physician specialty code C6, specifically for hospitalists. There has been a lot of confusion about what this means and some uncertainty about why clinicians should bother to use it.
Some folks thought initially that it was a new CPT code they could use to bill hospitalist services, which might recognize the increased intensity of services hospitalists often provide to their hospitalized patients compared to many traditional internal medicine and family medicine primary care physicians. Others thought it was a code that was added to the HCFA 1500 billing form somewhere to designate that the service was provided by a hospitalist.
Neither is true. The C6 physician specialty code is one of a large number of such codes used by physicians to designate their primary physician specialty when they enroll with Medicare via the PECOS online enrollment system. It describes the unique type of medicine practiced by the enrolling physician and is used by the CMS both for claims processing purposes and for “programmatic” purposes (whatever that means).
It doesn’t change how your claim is processed or how much you get paid. So why bother going through the laborious process of re-credentialing with CMS via PECOS just to change your specialty code? Well, I believe there are several ways in which the C6 specialty code provides value – both to you and to the specialty of hospital medicine.
Reduce concurrent care denials
First, it distinguishes you from a general internal medicine or general family medicine practitioner by recognizing “hospitalist” as a distinct specialty. This can be valuable from a financial perspective because it may reduce the risk that claims for your services might be denied due to “concurrent care” by another provider in the same specialty on the same calendar day.
And it’s not just a general internist or family medicine physician that you might run into concurrent care trouble with. I’ve seen situations where doctors completed critical care or cardiology fellowships but never got around to re-credentialing with Medicare in their new specialty, so their claims still showed up with an “internal medicine” physician specialty code, resulting in denied “concurrent care” claims for either the hospitalist or the specialist.
While Medicare may still see unnecessary overlap between services provided by you and an internal medicine or family physician to the same patient on the same calendar day, you can make a better argument that your services were unique and complementary to (not duplicative of) the services of others if you are credentialed as a hospitalist.
Ensure “apples to apples” comparisons
A second reason to re-credential as a hospitalist is to ensure that when the CMS looks at the services you are providing and the CPT codes you are selecting, it is comparing you to an appropriate peer group for compliance purposes.
The mix of CPT codes reported by hospitalists in the SHM State of Hospital Medicine Survey has historically tilted toward higher-level care than has the mix of CPT codes reported by the CMS for internal medicine or family medicine physicians. But last year when Medicare released the utilization of evaluation and management services by specialty for calendar year 2017, CPT utilization was shown separately for hospitalists for the first time!
The volume of services reported for physicians credentialed as hospitalists was very small relative to the volume of inpatient services provided by internal medicine and family medicine physicians, but the distribution of inpatient admission, subsequent visit, and discharge codes for hospitalists closely mirrored those reported by SHM in its 2018 State of Hospital Medicine Report (see graphic).
If you’re going to be targeted in a RAC audit for the high proportion of 99233s you bill, you want to be sure the CMS is looking at your performance compared to those who are truly your peers, caring for patients of the same type and complexity.
Improve CMS data used for research purposes
Finally, the ability of academic hospitalists and other health services researchers to utilize Medicare claims data to better understand the care provided by hospitalists and its impact on the overall health care system will be significantly enhanced by a more robust presence of physicians who have identified themselves as hospitalists in the PECOS credentialing system.
We care for the majority of patients in most hospitals these days, yet “hospitalists” billed only 2,009,869 inpatient subsequent visits (CPT codes 99231, 99232, and 99233) in 2017 compared to 25,903,829 billed by internal medicine physicians and 4,678,111 billed by family medicine physicians. And regardless of what you think about using claims data as a proxy for health care services and quality, it’s undeniably the best data set we currently have.
So, let’s work together to build a bigger, better database of hospitalist information at the CMS. I urge you to go to your credentialing folks today and find out how you can work with them to get yourself re-credentialed in PECOS using the C6 “hospitalist” physician specialty.
Ms. Flores is a partner at Nelson Flores Hospital Medicine Consultants, La Quinta, Calif. She serves on SHM’s Practice Analysis and Annual Meeting Committees, and helps to coordinate SHM’s bi-annual State of Hospital Medicine Survey. This article appeared originally in SHM's official blog The Hospital Leader. Read more recent posts here.
Hospitalists can help alleviate rising drug costs
Four key actions providers can take
Because of the increasing costs of prescription drugs and medical therapies, many patients are unable to afford the treatment they need that could improve their health or even save their lives. In the United States, drug manufacturers can set their own prices – a policy that has resulted in overall medicine costs being far higher than in other places around the globe. Increasingly, insurers are passing the costs along to patients through higher deductibles, and pharmaceutical companies are making record profits.
Something needs to change in order to achieve the right balance between maintaining pharmaceutical innovation and ensuring patients have proper access to treatments they need. Waiting for legislation, regulation, or the courts is not an effective short-term solution. Instead, hospitalists can take immediate actions to help by alleviating the costs for as many patients possible.
Historical context
Many might be wondering how prescription costs became so imbalanced in the first place. Here are a few important factors that played a role in the dramatic price increase of pharmaceuticals:
Entrance of generic drugs: Around 2012 the entrance of generic drugs caused major unexpected competition in the medical industry. During this time, many insurers were promoting the generic drugs and not allowing brand names to be covered when a generic substitute was available.
“Orphan drugs” and manufacturer pricing: In 2014, 33 new brand-name drugs were launched in the United States, and only 8 had a direct price competitor at the time they were introduced. In addition, manufacturers were free to set their prices. Over the past decade, introductory prices for brand name drugs have reached unprecedented levels. Furthermore, manufacturers use the patent protections to increase their prices every year, even when no significant improvements have been made to the drug.
Expiring patents: According to research, there are 182 drugs that no longer have patent protection or any associated generics available. This creates opportunities for manufacturers to maintain patent-era pricing or even engage in price gouging.
Lack of robust competition: Several high-priced blockbuster drugs hit the market to treat serious diseases, most of which do not have generic brand substitutes, which leaves only one option for patients – and it’s usually not affordable. According to research, more than 500 drugs have only one marketed generic. In addition, manufacturer mergers and acquisitions have occurred, which has led to a more concentrated and less competitive market for pricing.
Stricter Food and Drug Administration policies: American consumers have access to the safest and most advanced pharmaceutical system in the world, which requires several trials and testing before the drug can be approved and brought to the market. Despite the benefits of these strict procedures, the downside means higher costs for the brand and manufacturer that they will want to recoup through the price of the drug on the market.
Number of new drugs allowed to enter the market: New drugs that enter the market in the United States do so more quickly than in most other countries. Research shows the U.S. pharmaceutical market contributes to 45% of the global pharmaceutical market. The $76 billion in research and development that pharmaceutical companies claim overlooks the ways that U.S. employers and taxpayers pay for at least 44% through tax subsidies and credits. What makes it worse is that research shows most corporate research and development is directed at minimally innovative new drugs, using the system to secure patents and charge monopoly prices.
Compared with other high-income countries, the United States spends the most per capita on prescription drugs. While insured U.S. patients often pay little or nothing for generic prescriptions, they can be billed tens of thousands of dollars for certain high-priced medicines. The United States has the highest rate of insured patients skipping or not filling prescriptions because of cost. For example, the price of EpiPens, a drug delivery system that is crucial for persons experiencing life-threatening allergic reactions, has increased more than $500 in just 9 years.
How to alleviate rising drug costs
The good news is that hospitalists can do something about the high costs of pharmaceuticals.
Understand and offer alternative ways for drug intake: Many patients admitted to a hospital with severe infections are initially started with intravenous medications. Although conversion from intravenous to oral therapy is inappropriate for a patient who is critically ill or has an inability to absorb oral medications, every hospital will have a certain number of patients who are eligible for a switch from intravenous to oral therapy.
The World Health Organization (WHO) reports that the irrational use of medicines is a major problem worldwide, including antibiotics. Switching from IV to oral enables one to select a cheaper or older antibiotic that is as effective as the IV antibiotic. However, this requires breaking the belief that many physicians still have that IV medications’ bioavailability is stronger and creates less susceptibility to the illness reoccurring in the patient. For many medications, essentially the same amount of drug is found in the blood when given intravenously or orally. In addition, research has shown several benefits beyond cost reduction for oral over IV, such as earlier discharge and reduced risk of infections.
Limit unnecessary antibiotic prescriptions and consider antibiotics stewardship programs: The Center for Disease Control reports that one in three (47 million) antibiotic prescriptions are unnecessary. Most of these unnecessary antibiotics are prescribed for respiratory conditions caused by viruses including common colds, viral sore throats, bronchitis, and sinus and ear infections that do not respond to antibiotics. Although the White House released The National Action Plan for Combating Antibiotic-Resistant Bacteria (CARB) in 2015, which set a goal of reducing inappropriate outpatient antibiotic use by at least half by 2020, hospitalists can still do more by being extremely cautious with prescribing drugs to patients. Use appropriate consultants whenever necessary to suggest the right drug. For example, consider an infectious disease specialist to suggest the appropriate type and length of time for an antibiotic. In addition, hospital-based programs dedicated to improving antibiotic use, known as antibiotic stewardship programs (ASPs), have been shown to optimize the treatment of infections and reduce adverse events associated with antibiotic use.
Review labs and vitals carefully and encourage a higher level of patient care beyond the digital tools available: Studies have shown an oversight in an exam (a “miss”) can result in real consequences, including death. Our $3.4 trillion health care system is responsible for more than a quarter of a million deaths per year because of medical error. Much of that is a result of poorly coordinated care, poor communication, patients falling through the cracks, or knowledge not being transferred. “True clinical judgment is more than addressing the avalanche of blood work, imaging, and lab tests; it is about using human skills to understand where the patient is in the trajectory of a life and the disease, what the nature of the patient’s family and social circumstances is, and how much they want done,” wrote Dr. Abraham Verghese in the New York Times in 2018 (“How Tech Can Turn Doctors into Clerical Workers”). This also means understanding whether the patient is on any other type of medication and, as a result, having knowledge of possible consequences for drug interactions. Always look for safe medications or discontinue the use of any unnecessary drugs the patient is currently taking.
Allow pharmacies to automatically substitute less expensive equivalent drugs: When prescribing pharmaceuticals for patients, determine if there are any substitutes that can help alleviate costs while delivering equivalent care to the patient. This requires excellent ongoing communication with pharmacists and understanding the substitutes available, as well as any side effects or consequences.
Hospitalists can make a difference
There are many variables that play a role in rising pharmaceutical costs in the United States. One of the most significant is that there are no strategies in place to control pricing of drugs and the profits made by the pharmaceutical companies.
Although finding new drugs that can cure major life-threatening diseases or illnesses is important, so is ensuring that more patients have access to such drugs at a reasonable cost. While there are several ways that the government can and should help with enabling and supporting this balance, it most likely requires such large changes that it will take a long time. As a result, it is important for hospitalists to find effective short-term solutions that can be implemented right away to alleviate the rising costs of pharmaceuticals and provide proper patient care regardless of their economic status – all of which requires better research, analysis, and comparison before prescribing treatment to patients.
Dr. Kasarla is a hospitalist with APOGEE Physicians at Wise Surgical at Parkway in Fort Worth, Tex. He did his internal medicine residency at Mercy Hospital & Medical Center, Chicago. Contact him at [email protected]. Dr. Devireddy is a hospitalist based at Sri Ramachandra Medical Centre, Porur, Tamilnadu, India. Contact her at [email protected].
FURTHER READING
Olson and Sheiner (2017). “The Hutchins Center Explains: Prescription drug spending” Brooking.edu
Lo, Chris (2018). “Cost control: drug pricing policies around the world,” Pharmaceutical-Technology.com
Center for Disease Control and Prevention. (2016). 1 in 3 antibiotic prescriptions unnecessary. Retrieved Jan 31, 2019, from https://www.cdc.gov/media/releases/2016/p0503-unnecessary-prescriptions.html
Verghese, Abraham (2018). “How Tech Can Turn Doctors Into Clerical Workers” NYTimes.Com
Waxman, Corr, Martin et al (2017). “Getting to the Root of High Prescription Drug Prices” Commonwealthfund.org
American Council on Science and Health. (2018). Government Is The Big Reason EpiPen And Other Generics Are So Expensive. Retrieved Jan 31, 2019, from https://www.acsh.org/news/2018/06/23/government-big-reason-epipen-and-other-generics-are-so-expensive-13114
Statista. (2018). U.S. Pharmaceutical Industry – Statistics & Facts. Retrieved Jan 31, 2019, from https://www.statista.com/topics/1719/pharmaceutical-industry/
Four key actions providers can take
Four key actions providers can take
Because of the increasing costs of prescription drugs and medical therapies, many patients are unable to afford the treatment they need that could improve their health or even save their lives. In the United States, drug manufacturers can set their own prices – a policy that has resulted in overall medicine costs being far higher than in other places around the globe. Increasingly, insurers are passing the costs along to patients through higher deductibles, and pharmaceutical companies are making record profits.
Something needs to change in order to achieve the right balance between maintaining pharmaceutical innovation and ensuring patients have proper access to treatments they need. Waiting for legislation, regulation, or the courts is not an effective short-term solution. Instead, hospitalists can take immediate actions to help by alleviating the costs for as many patients possible.
Historical context
Many might be wondering how prescription costs became so imbalanced in the first place. Here are a few important factors that played a role in the dramatic price increase of pharmaceuticals:
Entrance of generic drugs: Around 2012 the entrance of generic drugs caused major unexpected competition in the medical industry. During this time, many insurers were promoting the generic drugs and not allowing brand names to be covered when a generic substitute was available.
“Orphan drugs” and manufacturer pricing: In 2014, 33 new brand-name drugs were launched in the United States, and only 8 had a direct price competitor at the time they were introduced. In addition, manufacturers were free to set their prices. Over the past decade, introductory prices for brand name drugs have reached unprecedented levels. Furthermore, manufacturers use the patent protections to increase their prices every year, even when no significant improvements have been made to the drug.
Expiring patents: According to research, there are 182 drugs that no longer have patent protection or any associated generics available. This creates opportunities for manufacturers to maintain patent-era pricing or even engage in price gouging.
Lack of robust competition: Several high-priced blockbuster drugs hit the market to treat serious diseases, most of which do not have generic brand substitutes, which leaves only one option for patients – and it’s usually not affordable. According to research, more than 500 drugs have only one marketed generic. In addition, manufacturer mergers and acquisitions have occurred, which has led to a more concentrated and less competitive market for pricing.
Stricter Food and Drug Administration policies: American consumers have access to the safest and most advanced pharmaceutical system in the world, which requires several trials and testing before the drug can be approved and brought to the market. Despite the benefits of these strict procedures, the downside means higher costs for the brand and manufacturer that they will want to recoup through the price of the drug on the market.
Number of new drugs allowed to enter the market: New drugs that enter the market in the United States do so more quickly than in most other countries. Research shows the U.S. pharmaceutical market contributes to 45% of the global pharmaceutical market. The $76 billion in research and development that pharmaceutical companies claim overlooks the ways that U.S. employers and taxpayers pay for at least 44% through tax subsidies and credits. What makes it worse is that research shows most corporate research and development is directed at minimally innovative new drugs, using the system to secure patents and charge monopoly prices.
Compared with other high-income countries, the United States spends the most per capita on prescription drugs. While insured U.S. patients often pay little or nothing for generic prescriptions, they can be billed tens of thousands of dollars for certain high-priced medicines. The United States has the highest rate of insured patients skipping or not filling prescriptions because of cost. For example, the price of EpiPens, a drug delivery system that is crucial for persons experiencing life-threatening allergic reactions, has increased more than $500 in just 9 years.
How to alleviate rising drug costs
The good news is that hospitalists can do something about the high costs of pharmaceuticals.
Understand and offer alternative ways for drug intake: Many patients admitted to a hospital with severe infections are initially started with intravenous medications. Although conversion from intravenous to oral therapy is inappropriate for a patient who is critically ill or has an inability to absorb oral medications, every hospital will have a certain number of patients who are eligible for a switch from intravenous to oral therapy.
The World Health Organization (WHO) reports that the irrational use of medicines is a major problem worldwide, including antibiotics. Switching from IV to oral enables one to select a cheaper or older antibiotic that is as effective as the IV antibiotic. However, this requires breaking the belief that many physicians still have that IV medications’ bioavailability is stronger and creates less susceptibility to the illness reoccurring in the patient. For many medications, essentially the same amount of drug is found in the blood when given intravenously or orally. In addition, research has shown several benefits beyond cost reduction for oral over IV, such as earlier discharge and reduced risk of infections.
Limit unnecessary antibiotic prescriptions and consider antibiotics stewardship programs: The Center for Disease Control reports that one in three (47 million) antibiotic prescriptions are unnecessary. Most of these unnecessary antibiotics are prescribed for respiratory conditions caused by viruses including common colds, viral sore throats, bronchitis, and sinus and ear infections that do not respond to antibiotics. Although the White House released The National Action Plan for Combating Antibiotic-Resistant Bacteria (CARB) in 2015, which set a goal of reducing inappropriate outpatient antibiotic use by at least half by 2020, hospitalists can still do more by being extremely cautious with prescribing drugs to patients. Use appropriate consultants whenever necessary to suggest the right drug. For example, consider an infectious disease specialist to suggest the appropriate type and length of time for an antibiotic. In addition, hospital-based programs dedicated to improving antibiotic use, known as antibiotic stewardship programs (ASPs), have been shown to optimize the treatment of infections and reduce adverse events associated with antibiotic use.
Review labs and vitals carefully and encourage a higher level of patient care beyond the digital tools available: Studies have shown an oversight in an exam (a “miss”) can result in real consequences, including death. Our $3.4 trillion health care system is responsible for more than a quarter of a million deaths per year because of medical error. Much of that is a result of poorly coordinated care, poor communication, patients falling through the cracks, or knowledge not being transferred. “True clinical judgment is more than addressing the avalanche of blood work, imaging, and lab tests; it is about using human skills to understand where the patient is in the trajectory of a life and the disease, what the nature of the patient’s family and social circumstances is, and how much they want done,” wrote Dr. Abraham Verghese in the New York Times in 2018 (“How Tech Can Turn Doctors into Clerical Workers”). This also means understanding whether the patient is on any other type of medication and, as a result, having knowledge of possible consequences for drug interactions. Always look for safe medications or discontinue the use of any unnecessary drugs the patient is currently taking.
Allow pharmacies to automatically substitute less expensive equivalent drugs: When prescribing pharmaceuticals for patients, determine if there are any substitutes that can help alleviate costs while delivering equivalent care to the patient. This requires excellent ongoing communication with pharmacists and understanding the substitutes available, as well as any side effects or consequences.
Hospitalists can make a difference
There are many variables that play a role in rising pharmaceutical costs in the United States. One of the most significant is that there are no strategies in place to control pricing of drugs and the profits made by the pharmaceutical companies.
Although finding new drugs that can cure major life-threatening diseases or illnesses is important, so is ensuring that more patients have access to such drugs at a reasonable cost. While there are several ways that the government can and should help with enabling and supporting this balance, it most likely requires such large changes that it will take a long time. As a result, it is important for hospitalists to find effective short-term solutions that can be implemented right away to alleviate the rising costs of pharmaceuticals and provide proper patient care regardless of their economic status – all of which requires better research, analysis, and comparison before prescribing treatment to patients.
Dr. Kasarla is a hospitalist with APOGEE Physicians at Wise Surgical at Parkway in Fort Worth, Tex. He did his internal medicine residency at Mercy Hospital & Medical Center, Chicago. Contact him at [email protected]. Dr. Devireddy is a hospitalist based at Sri Ramachandra Medical Centre, Porur, Tamilnadu, India. Contact her at [email protected].
FURTHER READING
Olson and Sheiner (2017). “The Hutchins Center Explains: Prescription drug spending” Brooking.edu
Lo, Chris (2018). “Cost control: drug pricing policies around the world,” Pharmaceutical-Technology.com
Center for Disease Control and Prevention. (2016). 1 in 3 antibiotic prescriptions unnecessary. Retrieved Jan 31, 2019, from https://www.cdc.gov/media/releases/2016/p0503-unnecessary-prescriptions.html
Verghese, Abraham (2018). “How Tech Can Turn Doctors Into Clerical Workers” NYTimes.Com
Waxman, Corr, Martin et al (2017). “Getting to the Root of High Prescription Drug Prices” Commonwealthfund.org
American Council on Science and Health. (2018). Government Is The Big Reason EpiPen And Other Generics Are So Expensive. Retrieved Jan 31, 2019, from https://www.acsh.org/news/2018/06/23/government-big-reason-epipen-and-other-generics-are-so-expensive-13114
Statista. (2018). U.S. Pharmaceutical Industry – Statistics & Facts. Retrieved Jan 31, 2019, from https://www.statista.com/topics/1719/pharmaceutical-industry/
Because of the increasing costs of prescription drugs and medical therapies, many patients are unable to afford the treatment they need that could improve their health or even save their lives. In the United States, drug manufacturers can set their own prices – a policy that has resulted in overall medicine costs being far higher than in other places around the globe. Increasingly, insurers are passing the costs along to patients through higher deductibles, and pharmaceutical companies are making record profits.
Something needs to change in order to achieve the right balance between maintaining pharmaceutical innovation and ensuring patients have proper access to treatments they need. Waiting for legislation, regulation, or the courts is not an effective short-term solution. Instead, hospitalists can take immediate actions to help by alleviating the costs for as many patients possible.
Historical context
Many might be wondering how prescription costs became so imbalanced in the first place. Here are a few important factors that played a role in the dramatic price increase of pharmaceuticals:
Entrance of generic drugs: Around 2012 the entrance of generic drugs caused major unexpected competition in the medical industry. During this time, many insurers were promoting the generic drugs and not allowing brand names to be covered when a generic substitute was available.
“Orphan drugs” and manufacturer pricing: In 2014, 33 new brand-name drugs were launched in the United States, and only 8 had a direct price competitor at the time they were introduced. In addition, manufacturers were free to set their prices. Over the past decade, introductory prices for brand name drugs have reached unprecedented levels. Furthermore, manufacturers use the patent protections to increase their prices every year, even when no significant improvements have been made to the drug.
Expiring patents: According to research, there are 182 drugs that no longer have patent protection or any associated generics available. This creates opportunities for manufacturers to maintain patent-era pricing or even engage in price gouging.
Lack of robust competition: Several high-priced blockbuster drugs hit the market to treat serious diseases, most of which do not have generic brand substitutes, which leaves only one option for patients – and it’s usually not affordable. According to research, more than 500 drugs have only one marketed generic. In addition, manufacturer mergers and acquisitions have occurred, which has led to a more concentrated and less competitive market for pricing.
Stricter Food and Drug Administration policies: American consumers have access to the safest and most advanced pharmaceutical system in the world, which requires several trials and testing before the drug can be approved and brought to the market. Despite the benefits of these strict procedures, the downside means higher costs for the brand and manufacturer that they will want to recoup through the price of the drug on the market.
Number of new drugs allowed to enter the market: New drugs that enter the market in the United States do so more quickly than in most other countries. Research shows the U.S. pharmaceutical market contributes to 45% of the global pharmaceutical market. The $76 billion in research and development that pharmaceutical companies claim overlooks the ways that U.S. employers and taxpayers pay for at least 44% through tax subsidies and credits. What makes it worse is that research shows most corporate research and development is directed at minimally innovative new drugs, using the system to secure patents and charge monopoly prices.
Compared with other high-income countries, the United States spends the most per capita on prescription drugs. While insured U.S. patients often pay little or nothing for generic prescriptions, they can be billed tens of thousands of dollars for certain high-priced medicines. The United States has the highest rate of insured patients skipping or not filling prescriptions because of cost. For example, the price of EpiPens, a drug delivery system that is crucial for persons experiencing life-threatening allergic reactions, has increased more than $500 in just 9 years.
How to alleviate rising drug costs
The good news is that hospitalists can do something about the high costs of pharmaceuticals.
Understand and offer alternative ways for drug intake: Many patients admitted to a hospital with severe infections are initially started with intravenous medications. Although conversion from intravenous to oral therapy is inappropriate for a patient who is critically ill or has an inability to absorb oral medications, every hospital will have a certain number of patients who are eligible for a switch from intravenous to oral therapy.
The World Health Organization (WHO) reports that the irrational use of medicines is a major problem worldwide, including antibiotics. Switching from IV to oral enables one to select a cheaper or older antibiotic that is as effective as the IV antibiotic. However, this requires breaking the belief that many physicians still have that IV medications’ bioavailability is stronger and creates less susceptibility to the illness reoccurring in the patient. For many medications, essentially the same amount of drug is found in the blood when given intravenously or orally. In addition, research has shown several benefits beyond cost reduction for oral over IV, such as earlier discharge and reduced risk of infections.
Limit unnecessary antibiotic prescriptions and consider antibiotics stewardship programs: The Center for Disease Control reports that one in three (47 million) antibiotic prescriptions are unnecessary. Most of these unnecessary antibiotics are prescribed for respiratory conditions caused by viruses including common colds, viral sore throats, bronchitis, and sinus and ear infections that do not respond to antibiotics. Although the White House released The National Action Plan for Combating Antibiotic-Resistant Bacteria (CARB) in 2015, which set a goal of reducing inappropriate outpatient antibiotic use by at least half by 2020, hospitalists can still do more by being extremely cautious with prescribing drugs to patients. Use appropriate consultants whenever necessary to suggest the right drug. For example, consider an infectious disease specialist to suggest the appropriate type and length of time for an antibiotic. In addition, hospital-based programs dedicated to improving antibiotic use, known as antibiotic stewardship programs (ASPs), have been shown to optimize the treatment of infections and reduce adverse events associated with antibiotic use.
Review labs and vitals carefully and encourage a higher level of patient care beyond the digital tools available: Studies have shown an oversight in an exam (a “miss”) can result in real consequences, including death. Our $3.4 trillion health care system is responsible for more than a quarter of a million deaths per year because of medical error. Much of that is a result of poorly coordinated care, poor communication, patients falling through the cracks, or knowledge not being transferred. “True clinical judgment is more than addressing the avalanche of blood work, imaging, and lab tests; it is about using human skills to understand where the patient is in the trajectory of a life and the disease, what the nature of the patient’s family and social circumstances is, and how much they want done,” wrote Dr. Abraham Verghese in the New York Times in 2018 (“How Tech Can Turn Doctors into Clerical Workers”). This also means understanding whether the patient is on any other type of medication and, as a result, having knowledge of possible consequences for drug interactions. Always look for safe medications or discontinue the use of any unnecessary drugs the patient is currently taking.
Allow pharmacies to automatically substitute less expensive equivalent drugs: When prescribing pharmaceuticals for patients, determine if there are any substitutes that can help alleviate costs while delivering equivalent care to the patient. This requires excellent ongoing communication with pharmacists and understanding the substitutes available, as well as any side effects or consequences.
Hospitalists can make a difference
There are many variables that play a role in rising pharmaceutical costs in the United States. One of the most significant is that there are no strategies in place to control pricing of drugs and the profits made by the pharmaceutical companies.
Although finding new drugs that can cure major life-threatening diseases or illnesses is important, so is ensuring that more patients have access to such drugs at a reasonable cost. While there are several ways that the government can and should help with enabling and supporting this balance, it most likely requires such large changes that it will take a long time. As a result, it is important for hospitalists to find effective short-term solutions that can be implemented right away to alleviate the rising costs of pharmaceuticals and provide proper patient care regardless of their economic status – all of which requires better research, analysis, and comparison before prescribing treatment to patients.
Dr. Kasarla is a hospitalist with APOGEE Physicians at Wise Surgical at Parkway in Fort Worth, Tex. He did his internal medicine residency at Mercy Hospital & Medical Center, Chicago. Contact him at [email protected]. Dr. Devireddy is a hospitalist based at Sri Ramachandra Medical Centre, Porur, Tamilnadu, India. Contact her at [email protected].
FURTHER READING
Olson and Sheiner (2017). “The Hutchins Center Explains: Prescription drug spending” Brooking.edu
Lo, Chris (2018). “Cost control: drug pricing policies around the world,” Pharmaceutical-Technology.com
Center for Disease Control and Prevention. (2016). 1 in 3 antibiotic prescriptions unnecessary. Retrieved Jan 31, 2019, from https://www.cdc.gov/media/releases/2016/p0503-unnecessary-prescriptions.html
Verghese, Abraham (2018). “How Tech Can Turn Doctors Into Clerical Workers” NYTimes.Com
Waxman, Corr, Martin et al (2017). “Getting to the Root of High Prescription Drug Prices” Commonwealthfund.org
American Council on Science and Health. (2018). Government Is The Big Reason EpiPen And Other Generics Are So Expensive. Retrieved Jan 31, 2019, from https://www.acsh.org/news/2018/06/23/government-big-reason-epipen-and-other-generics-are-so-expensive-13114
Statista. (2018). U.S. Pharmaceutical Industry – Statistics & Facts. Retrieved Jan 31, 2019, from https://www.statista.com/topics/1719/pharmaceutical-industry/
HM19: Things we do for no reason
The pediatric version
Presenters
Amit K. Pahwa, MD, FAAP; Nicola Orlov, MD, MPH, FAAP
Workshop title
Things we do for no reason (pediatrics)
Session summary
As he began by stating the Institute of Medicine definition of high-value care (HVC), Amit K. Pahwa, MD, of Johns Hopkins Medicine, Baltimore, described HVC as the best care for the patient, with the optimal result for the circumstances, at the right price. But few pediatric residency programs provide education regarding HVC, with only 11% providing a formal HVC curriculum, as found by a survey of pediatric program directors and chief residents in a study published in 2017.
Dr. Pahwa then provided examples of cases in which HVC could be optimized, including reducing rebound bilirubin levels in neonatal hyperbilirubinemia, giving nasogastric feeds instead of IV hydration in bronchiolitis, reducing unnecessary vital sign checks, and providing apple juice supplemented with liquids of choice instead of more expensive oral electrolyte solutions.
Nicola Orlov, MD, of the University of Chicago presented another illustrative case which highlighted the need to reduce vital sign frequency when appropriate. This was linked to her work at Comer Children’s Hospital on reducing nighttime sleep disruptions in hospitalized children, as part of the SIESTA (Sleep for Inpatients: Empowering Staff to Act) study. This led to a significant reduction in nurse/physician interruptions during the study period.
Key takeaways for HM
- High-value care (HVC) is a key focus of systems improvement in the field of pediatric hospital medicine.
- Educational efforts for all levels of learners is inadequate currently, and needs to be augmented.
- Quality improvement projects to promote HVC can lead not only to reduced costs, but improved quality and patient experience.
Dr. Chang is a pediatric hospitalist at Baystate Children’s Hospital in Springfield, Mass., and is the pediatric editor of The Hospitalist.
The pediatric version
The pediatric version
Presenters
Amit K. Pahwa, MD, FAAP; Nicola Orlov, MD, MPH, FAAP
Workshop title
Things we do for no reason (pediatrics)
Session summary
As he began by stating the Institute of Medicine definition of high-value care (HVC), Amit K. Pahwa, MD, of Johns Hopkins Medicine, Baltimore, described HVC as the best care for the patient, with the optimal result for the circumstances, at the right price. But few pediatric residency programs provide education regarding HVC, with only 11% providing a formal HVC curriculum, as found by a survey of pediatric program directors and chief residents in a study published in 2017.
Dr. Pahwa then provided examples of cases in which HVC could be optimized, including reducing rebound bilirubin levels in neonatal hyperbilirubinemia, giving nasogastric feeds instead of IV hydration in bronchiolitis, reducing unnecessary vital sign checks, and providing apple juice supplemented with liquids of choice instead of more expensive oral electrolyte solutions.
Nicola Orlov, MD, of the University of Chicago presented another illustrative case which highlighted the need to reduce vital sign frequency when appropriate. This was linked to her work at Comer Children’s Hospital on reducing nighttime sleep disruptions in hospitalized children, as part of the SIESTA (Sleep for Inpatients: Empowering Staff to Act) study. This led to a significant reduction in nurse/physician interruptions during the study period.
Key takeaways for HM
- High-value care (HVC) is a key focus of systems improvement in the field of pediatric hospital medicine.
- Educational efforts for all levels of learners is inadequate currently, and needs to be augmented.
- Quality improvement projects to promote HVC can lead not only to reduced costs, but improved quality and patient experience.
Dr. Chang is a pediatric hospitalist at Baystate Children’s Hospital in Springfield, Mass., and is the pediatric editor of The Hospitalist.
Presenters
Amit K. Pahwa, MD, FAAP; Nicola Orlov, MD, MPH, FAAP
Workshop title
Things we do for no reason (pediatrics)
Session summary
As he began by stating the Institute of Medicine definition of high-value care (HVC), Amit K. Pahwa, MD, of Johns Hopkins Medicine, Baltimore, described HVC as the best care for the patient, with the optimal result for the circumstances, at the right price. But few pediatric residency programs provide education regarding HVC, with only 11% providing a formal HVC curriculum, as found by a survey of pediatric program directors and chief residents in a study published in 2017.
Dr. Pahwa then provided examples of cases in which HVC could be optimized, including reducing rebound bilirubin levels in neonatal hyperbilirubinemia, giving nasogastric feeds instead of IV hydration in bronchiolitis, reducing unnecessary vital sign checks, and providing apple juice supplemented with liquids of choice instead of more expensive oral electrolyte solutions.
Nicola Orlov, MD, of the University of Chicago presented another illustrative case which highlighted the need to reduce vital sign frequency when appropriate. This was linked to her work at Comer Children’s Hospital on reducing nighttime sleep disruptions in hospitalized children, as part of the SIESTA (Sleep for Inpatients: Empowering Staff to Act) study. This led to a significant reduction in nurse/physician interruptions during the study period.
Key takeaways for HM
- High-value care (HVC) is a key focus of systems improvement in the field of pediatric hospital medicine.
- Educational efforts for all levels of learners is inadequate currently, and needs to be augmented.
- Quality improvement projects to promote HVC can lead not only to reduced costs, but improved quality and patient experience.
Dr. Chang is a pediatric hospitalist at Baystate Children’s Hospital in Springfield, Mass., and is the pediatric editor of The Hospitalist.












