User login
Preserving the VBAC alternative: 8 pearls
Is vaginal birth after cesarean an endangered procedure? Most women who attempt a trial of labor after cesarean have good outcomes, and those at high risk for adverse events, who should be excluded, are becoming increasingly better defined. Yet many physicians eschew this option altogether because of serious concerns about the safety of trial of labor after cesarean.1
If VBAC is to play a role in obstetrical management in the 21st century, we will need to improve our ability to distinguish these 2 populations:
- women with a low risk for complications and a high likelihood of a successful trial of labor, and
- women at high risk for adverse outcomes.
Of many reports documenting risk factors for the most feared complication, uterine rupture, none are from randomized controlled studies of trial of labor after cesarean versus elective repeat cesarean (and such reports are unlikely to be forthcoming). Therefore, we must depend on less rigorously obtained information. Nonetheless, we do have a wealth of data to guide us.
The 8 “pearls” in this article summarize what we know from the available data on vaginal birth after cesarean.
- The repeat cesarean rate varies by prior indication for cesarean: lowest for breech; highest for failure to progress.
- Induced labor has a higher rate of repeat cesarean than spontaneous labor.
- Maternal obesity and fetal macrosomia lower the success rate.
- Induction with oxytocin is associated with an increased risk of uterine rupture, but oxytocin can be used judiciously for augmentation of labor.
- Prostaglandins should not be used for cervical ripening or induction.
- Having more than 1 prior cesarean increases risk of uterine rupture.
- Interdelivery intervals of up to 18 months and maybe even 24 months are associated with an increased risk for uterine rupture. Women should be discouraged from becoming pregnant for at least 9 months, and possibly up to 15 months, after cesarean, if they are contemplating a trial of labor after cesarean for their next delivery.
- Uterine rupture is 5 times less likely in women who have had a vaginal delivery either before or after a prior cesarean delivery.
Pearl 1VBAC rate varies by prior indication
The overall rate of successful vaginal delivery for all women attempting trial of labor after cesarean varies from approximately 60% to 80%.
We showed, however, that the repeat cesarean rate varies by prior indication for cesarean. The rate for those whose prior cesarean was due to breech presentation was 13.9%, which approximated the cesarean rate among nulliparas during the study period (13.5%). The highest rate of repeat cesarean, 37.3%, was for those whose prior cesarean was for failure to progress.2
Pearl 2Repeat cesarean rate is higher with induced labor
Failed induction is a risk for all gravidas having labor induced, regardless of any prior cesarean deliveries. It is especially common among women with an unripe cervix.
Among women with a prior cesarean, labor induction has an approximately 10% increased rate of repeat cesarean, compared with those undergoing spontaneous labor.3
Pearl 3Successful VBAC rate is lower with maternal obesity or fetal macrosomia
Recent evidence suggests that both maternal and fetal weights influence the success of trial of labor after cesarean.
Maternal obesity is associated with a decreased success rate for trial of labor after prior cesarean delivery, but the magnitude of this risk is not well characterized: A success rate of 13% for morbidly obese women has been documented4; more recently, however, a success rate of 57% was reported.5 These studies are limited by their small numbers.
Fetal macrosomia. For pregnancies with fetuses weighing greater than 4,000 g, the literature notes VBAC success rates of 40% to 60%.6,7
The factors most important for counseling a woman about obstetrical management after a prior cesarean are her likelihood of success from trial of labor, and the risks and benefits of both trial of labor and an elective repeat cesarean.
The findings noted in this article call for a scoring system that will more precisely define the risk for an individual patient contemplating trial of labor after prior cesarean.
Over the past 3 decades, multiple studies have attempted to predict the success of trial of labor after cesarean. Flamm and Geiger19 reported a 10-fold predicted difference in the rate of cesarean based on a scoring system incorporating:
- maternal age,
- prior vaginal delivery,
- prior indication for cesarean, and
- the intrapartum assessment of cervical dilation and effacement.
Others have used additional variables including:
- estimated fetal weight,
- gestational age,
- prepregnancy body mass index,
- maternal weight gain, and
- induction of labor.20-23
With further investigation and better identification of those at highest and lowest risk, we will be better able to counsel each patient on her individual risks for a uterine rupture during a trial of labor after prior cesarean delivery.
Pearl 4Risk of rupture is greater with oxytocin induction
Women with a prior cesarean delivery face an increased risk of uterine rupture with labor induction.8,9 Zelop et al8 demonstrated that labor induction with oxytocin is associated with a 4- to 5-fold increased risk of uterine rupture compared to spontaneous labor.
Lydon-Rochelle et al9 reported an increased risk of uterine rupture for those in spontaneous labor and those induced without prostaglandins, compared with women opting for elective repeat cesarean. The odds ratios for patients with spontaneous labor (3.3; 95% confidence interval [CI] 1.8-6.0) and for those with labor induced without prostaglandins (4.9; 95% CI 2.4-9.7) were not statistically significantly different.
Recent trials have suggested that induction of labor is not associated with uterine rupture, though these studies are limited by relatively low numbers of patients. Delaney and Young10 reported rates of uterine rupture of 0.7% for those with induced labor as compared to 0.3% for those with spontaneous labor (P = 0.1). By combining these studies, we see a statistically significant increased rate of uterine rupture approximately twice that of those in spontaneous labor.11
Oxytocin can be used judiciously for augmentation of labor for women with prior cesarean delivery, as it is not associated with an increased risk for uterine rupture in these cases.8
Pearl 5Prostaglandins should not be used for cervical ripening or induction
For patients with a prior cesarean delivery, prostaglandins used for cervical ripening are associated with a significantly higher rate of uterine rupture compared with repeat cesarean, and with either spontaneous labor or induction with oxytocin alone.9
Lydon-Rochelle et al9 demonstrated a 15.6 relative risk (95% CI 8.1-30.0) for uterine rupture among women having labor induced with prostaglandins, compared with those undergoing elective repeat cesarean.
Women with a prior vaginal delivery were 5 times less likely to suffer uterine rupture than those with no prior vaginal deliveries.
At the beginning of the 20th century, Cragin wrote a dictum that is still invoked: “Once a cesarean, always a cesarean.”24
Not often mentioned, however, is that he went on to discuss a patient who had 3 successful vaginal births after cesarean “without difficulty.”24
For much of the 20th century, vaginal birth after cesarean (VBAC) was the exception rather than the rule. Then, 25 years ago, the National Institutes of Health advocated a trial of labor for women who had a prior cesarean delivery. During this time, VBAC was greatly encouraged, and the rate of trials of labor after cesarean began to increase.25 Thus, 15 years ago, the cesarean delivery rate in the United States began to fall after an unprecedented rise during the previous decades.
A few years later, however, this trend ceased and the cesarean delivery rate once again began to rise. This switch can be attributed to both an increase in primary cesarean deliveries and a decrease in the VBAC rate.26 It coincides with published data on uterine rupture associated with a trial of labor after cesarean: Two case series published in 1991 together documented 20 uterine ruptures with 4 perinatal deaths, 4 neonates with neurological impairment, and 3 women who underwent hysterectomy due to the event.27,28
Further study is needed to more precisely identify those women at high risk for uterine rupture and low risk of success of a trial of labor, and also—perhaps more importantly—those women with a very low risk of uterine rupture and a high likelihood of success with a trial of labor. Perhaps such additional research will help reverse the current malpractice climate, which is influencing the move by many physicians away from VBAC.
Pearl 6More than 1 prior cesarean increases risk of rupture
The presence of multiple prior cesarean scars places a woman at greater risk for uterine rupture during trial of labor. This risk is likely 3 to 5 times higher than for patients with only 1 prior cesarean delivery.13
Pearl 7Risk of rupture is increased with interdelivery interval of up to 18-24 months
Women who have undergone a cesarean delivery and are contemplating a future trial of labor should be discouraged from becoming pregnant for at least 9 months, possibly up to 15 months.
Interdelivery interval has been shown to be an important contributor to the risk for uterine rupture during trial of labor after cesarean.12-14 Esposito et al14 were the first to note an increased risk of uterine scar failure—including both symptomatic uterine ruptures and asymptomatic uterine scar dehiscences—for those with an interpregnancy interval of less than 6 months.
We showed an increased risk for uterine rupture with an odds ratio of 3.0 (95% CI 1.2-7.2) for those with interdelivery intervals of up to 18 months compared with those who had interdelivery intervals of 19 months or longer.15 More recently, Bujold et al16 confirmed these findings: They found the odds ratio for uterine rupture to be 2.7 (95% CI 1.1- 6.5) for those with an interdelivery interval of up to 24 months.
In a smaller study, Huang et al17 suggested that the success of trial of labor after cesarean may also be lower for those with interdelivery intervals of up to 18 months.
Pearl 8A vaginal delivery before or after prior cesarean lowers risk of rupture
Patients with a prior vaginal delivery are at significantly lower risk for uterine rupture than those without.
We published a study evaluating women with 1 prior cesarean delivery and either a preceding vaginal delivery or a previous VBAC. Our data suggest that women with a prior vaginal delivery were 5 times less likely to experience uterine rupture than those with no prior vaginal deliveries, either before or after the prior cesarean (odds ratio 0.2; 95% CI 0.04-0.8).18
Summary and recommendations
To continue to use vaginal birth after cesarean as an obstetrical practice, we must be better able to identify patients at high and low risk for complications from this procedure, and those who have the greatest chance for success. Women with a nonrecurring indication for the prior cesarean (eg, breech) have the best chance for success. Those with recurring indications for the prior cesarean (eg, failure to progress, morbidly obese women, those with macrosomic fetuses, and those with short interdelivery intervals) may have lower success rates.
Patients contemplating a future trial of labor should consider avoiding pregnancy for at least 9 to 15 months after cesarean delivery.
How can we reduce the risk of uterine rupture in women who are considering a trial of labor after prior cesarean delivery?
- We should not give these women prostaglandins for cervical ripening.
- We must consider allowing a trial of labor for those in spontaneous labor, and be more hesitant about inducing the labor in these patients.
- Women with multiple prior cesareans may also benefit from avoiding a trial of labor.
- Having patients avoid pregnancy for at least 9 months, and maybe up to 15 months, after a cesarean delivery could also assist with decreasing our rate of uterine rupture.
- Keep in mind that those with prior vaginal delivery have a much lower rate of uterine rupture.
The authors report no financial relationships relevant to this article.
1. Greene MF. Vaginal delivery after cesarean section—is the risk acceptable? N Engl J Med. 2001;345:54-55.
2. Shipp TD, Zelop CM, Repke JT, Cohen A, Caughey AB, Lieberman E. Labor after previous cesarean: influence of prior indication and parity. Obstet Gynecol. 2000;95:913-916.
3. Zelop CM, Shipp TD, Cohen A, Repke JT, Lieberman E. Trial of labor after 40 weeks’ gestation in women with prior cesarean. Obstet Gynecol. 2001;97:391-393.
4. Chauhan SP, Magann EF, Carroll CS, Barrilleaux PS, Scardo JA, Martin, Jr. JN. Mode of delivery for the morbidly obese with prior cesarean delivery: vaginal versus repeat cesarean section. Am J Obstet Gynecol. 2001;185:349-354.
5. Edwards RK, Harnsberger DS, Johnson IM, Treloar RW, Cruz AC. Deciding on route of delivery for obese women with a prior cesarean delivery. Am J Obstet Gynecol. 2003;189:385-390.
6. Zelop CM, Shipp TD, Repke JT, Cohen A, Lieberman E. Outcomes of trial of labor following previous cesarean delivery among women with fetuses weighing >4000 g. Am J Obstet Gynecol. 2001;185:903-905.
7. Elkousy MA, Sammel M, Stevens E, Peipert JF, Macones G. The effect of birth weight on vaginal birth after cesarean delivery success rates. Am J Obstet Gynecol. 2003;188:824-830.
8. Zelop CM, Shipp TD, Repke JT, Cohen A, Caughey AB, Lieberman E. Uterine rupture during induced or augmented labor in gravid women with one prior cesarean delivery. Am J Obstet Gynecol. 1999;181:882-886.
9. Lydon-Rochelle M, Holt VL, Easterling TR, Martin DP. Risk of uterine rupture during labor among women with a prior cesarean delivery. N Engl J Med. 2001;345:3-8.
10. Delaney T, Young DC. Spontaneous versus induced labor after a previous cesarean delivery. Obstet Gynecol. 2003;102:39-44.
11. Shipp TD. Prostaglandin E2, oxytocin may raise risk of rupture after previous cesarean. OBG Management. March 2004;16:14-16.
12. The American College of Obstetricians and Gynecologists Committee on Obstetric Practice Induction of labor for vaginal birth after cesarean delivery. ACOG Committee Opinion No. 271. Washington DC: ACOG; 2002.
13. Caughey AB, Shipp TD, Repke JT, Zelop CM, Cohen A, Lieberman E. Rate of uterine rupture during a trial of labor in women with one or two prior cesarean deliveries. Am J Obstet Gynecol. 1999;181:872-876.
14. Esposito MA, Menihan CA, Malee MP. Association of interpregnancy interval with uterine scar failure in labor: a case-control study. Am J Obstet Gynecol. 2000;183:1180-1183.
15. Shipp TD, Zelop CM, Repke JT, Cohen A, Lieberman E. Interdelivery interval and risk of symptomatic uterine rupture. Obstet Gynecol. 2001;97:175-177.
16. Bujold E, Mehta SH, Bujold C, Gauthier RJ. Interdelivery interval and uterine rupture. Am J Obstet Gynecol. 2002;187:1199-1202.
17. Huang WH, Nakashima DK, Rumney PJ, Keegan KA, Jr, Chan K. Interdelivery interval and the success of vaginal birth after cesarean delivery. Obstet Gynecol. 2002;99:41-44.
18. Zelop CM, Shipp TD, Repke JT, Cohen A, Lieberman E. Effect of previous vaginal delivery on the risk of uterine rupture during a subsequent trial of labor. Am J Obstet Gynecol. 2000;183:1184-1186.
19. Flamm BL, Geiger AM. Vaginal birth after cesarean delivery: an admission scoring system. Obstet Gynecol. 1997;90:907-910.
20. Dhall K, Mittal SC, Grover V, Dhall GI. Childbirth following primary cesarean section—evaluation of a scoring system. Int J Gynaecol Obstet. 1987;25:199-205.
21. Pickhardt MG, Martin JN, Jr, Meydrech EF, et al. Vaginal birth after cesarean delivery: are there useful and valid predictors of success or failure? Am J Obstet Gynecol. 1992;166:1811-1819.
22. Troyer LR, Parisi VM. Obstetric parameters affecting success in a trial of labor: designation of a scoring system. Am J Obstet Gynecol. 1992;167:1099-1104.
23. Macones GA, Hausman N, Edelstein R, Stamilio DM, Marder SJ. Predicting outcomes of trials of labor in women attempting vaginal birth after cesarean delivery: a comparison of multivariate methods with neural networks. Am J Obstet Gynecol. 2001;184:409-413.
24. Cragin EB. Conservatism in obstetrics. New York Med J. 1916;104:1-3.
25. Harer WB, Jr. Vaginal birth after cesarean delivery: current status. JAMA. 2002;287:2627-2630.
26. National Vital Statistics Reports, Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics, National Vital Statistics System. Births: Final data for 2001. December 2002;51(2).-
27. Scott JR. Mandatory trial of labor after cesarean delivery: an alternative viewpoint. Obstet Gynecol. 1991;77:811-814.
28. Jones RO, Nagashima AW, Hartnett-Goodman MM, Goodlin RC. Rupture of low transverse cesarean scars during trial of labor. Obstet Gynecol. 1991;77:815-817.
Is vaginal birth after cesarean an endangered procedure? Most women who attempt a trial of labor after cesarean have good outcomes, and those at high risk for adverse events, who should be excluded, are becoming increasingly better defined. Yet many physicians eschew this option altogether because of serious concerns about the safety of trial of labor after cesarean.1
If VBAC is to play a role in obstetrical management in the 21st century, we will need to improve our ability to distinguish these 2 populations:
- women with a low risk for complications and a high likelihood of a successful trial of labor, and
- women at high risk for adverse outcomes.
Of many reports documenting risk factors for the most feared complication, uterine rupture, none are from randomized controlled studies of trial of labor after cesarean versus elective repeat cesarean (and such reports are unlikely to be forthcoming). Therefore, we must depend on less rigorously obtained information. Nonetheless, we do have a wealth of data to guide us.
The 8 “pearls” in this article summarize what we know from the available data on vaginal birth after cesarean.
- The repeat cesarean rate varies by prior indication for cesarean: lowest for breech; highest for failure to progress.
- Induced labor has a higher rate of repeat cesarean than spontaneous labor.
- Maternal obesity and fetal macrosomia lower the success rate.
- Induction with oxytocin is associated with an increased risk of uterine rupture, but oxytocin can be used judiciously for augmentation of labor.
- Prostaglandins should not be used for cervical ripening or induction.
- Having more than 1 prior cesarean increases risk of uterine rupture.
- Interdelivery intervals of up to 18 months and maybe even 24 months are associated with an increased risk for uterine rupture. Women should be discouraged from becoming pregnant for at least 9 months, and possibly up to 15 months, after cesarean, if they are contemplating a trial of labor after cesarean for their next delivery.
- Uterine rupture is 5 times less likely in women who have had a vaginal delivery either before or after a prior cesarean delivery.
Pearl 1VBAC rate varies by prior indication
The overall rate of successful vaginal delivery for all women attempting trial of labor after cesarean varies from approximately 60% to 80%.
We showed, however, that the repeat cesarean rate varies by prior indication for cesarean. The rate for those whose prior cesarean was due to breech presentation was 13.9%, which approximated the cesarean rate among nulliparas during the study period (13.5%). The highest rate of repeat cesarean, 37.3%, was for those whose prior cesarean was for failure to progress.2
Pearl 2Repeat cesarean rate is higher with induced labor
Failed induction is a risk for all gravidas having labor induced, regardless of any prior cesarean deliveries. It is especially common among women with an unripe cervix.
Among women with a prior cesarean, labor induction has an approximately 10% increased rate of repeat cesarean, compared with those undergoing spontaneous labor.3
Pearl 3Successful VBAC rate is lower with maternal obesity or fetal macrosomia
Recent evidence suggests that both maternal and fetal weights influence the success of trial of labor after cesarean.
Maternal obesity is associated with a decreased success rate for trial of labor after prior cesarean delivery, but the magnitude of this risk is not well characterized: A success rate of 13% for morbidly obese women has been documented4; more recently, however, a success rate of 57% was reported.5 These studies are limited by their small numbers.
Fetal macrosomia. For pregnancies with fetuses weighing greater than 4,000 g, the literature notes VBAC success rates of 40% to 60%.6,7
The factors most important for counseling a woman about obstetrical management after a prior cesarean are her likelihood of success from trial of labor, and the risks and benefits of both trial of labor and an elective repeat cesarean.
The findings noted in this article call for a scoring system that will more precisely define the risk for an individual patient contemplating trial of labor after prior cesarean.
Over the past 3 decades, multiple studies have attempted to predict the success of trial of labor after cesarean. Flamm and Geiger19 reported a 10-fold predicted difference in the rate of cesarean based on a scoring system incorporating:
- maternal age,
- prior vaginal delivery,
- prior indication for cesarean, and
- the intrapartum assessment of cervical dilation and effacement.
Others have used additional variables including:
- estimated fetal weight,
- gestational age,
- prepregnancy body mass index,
- maternal weight gain, and
- induction of labor.20-23
With further investigation and better identification of those at highest and lowest risk, we will be better able to counsel each patient on her individual risks for a uterine rupture during a trial of labor after prior cesarean delivery.
Pearl 4Risk of rupture is greater with oxytocin induction
Women with a prior cesarean delivery face an increased risk of uterine rupture with labor induction.8,9 Zelop et al8 demonstrated that labor induction with oxytocin is associated with a 4- to 5-fold increased risk of uterine rupture compared to spontaneous labor.
Lydon-Rochelle et al9 reported an increased risk of uterine rupture for those in spontaneous labor and those induced without prostaglandins, compared with women opting for elective repeat cesarean. The odds ratios for patients with spontaneous labor (3.3; 95% confidence interval [CI] 1.8-6.0) and for those with labor induced without prostaglandins (4.9; 95% CI 2.4-9.7) were not statistically significantly different.
Recent trials have suggested that induction of labor is not associated with uterine rupture, though these studies are limited by relatively low numbers of patients. Delaney and Young10 reported rates of uterine rupture of 0.7% for those with induced labor as compared to 0.3% for those with spontaneous labor (P = 0.1). By combining these studies, we see a statistically significant increased rate of uterine rupture approximately twice that of those in spontaneous labor.11
Oxytocin can be used judiciously for augmentation of labor for women with prior cesarean delivery, as it is not associated with an increased risk for uterine rupture in these cases.8
Pearl 5Prostaglandins should not be used for cervical ripening or induction
For patients with a prior cesarean delivery, prostaglandins used for cervical ripening are associated with a significantly higher rate of uterine rupture compared with repeat cesarean, and with either spontaneous labor or induction with oxytocin alone.9
Lydon-Rochelle et al9 demonstrated a 15.6 relative risk (95% CI 8.1-30.0) for uterine rupture among women having labor induced with prostaglandins, compared with those undergoing elective repeat cesarean.
Women with a prior vaginal delivery were 5 times less likely to suffer uterine rupture than those with no prior vaginal deliveries.
At the beginning of the 20th century, Cragin wrote a dictum that is still invoked: “Once a cesarean, always a cesarean.”24
Not often mentioned, however, is that he went on to discuss a patient who had 3 successful vaginal births after cesarean “without difficulty.”24
For much of the 20th century, vaginal birth after cesarean (VBAC) was the exception rather than the rule. Then, 25 years ago, the National Institutes of Health advocated a trial of labor for women who had a prior cesarean delivery. During this time, VBAC was greatly encouraged, and the rate of trials of labor after cesarean began to increase.25 Thus, 15 years ago, the cesarean delivery rate in the United States began to fall after an unprecedented rise during the previous decades.
A few years later, however, this trend ceased and the cesarean delivery rate once again began to rise. This switch can be attributed to both an increase in primary cesarean deliveries and a decrease in the VBAC rate.26 It coincides with published data on uterine rupture associated with a trial of labor after cesarean: Two case series published in 1991 together documented 20 uterine ruptures with 4 perinatal deaths, 4 neonates with neurological impairment, and 3 women who underwent hysterectomy due to the event.27,28
Further study is needed to more precisely identify those women at high risk for uterine rupture and low risk of success of a trial of labor, and also—perhaps more importantly—those women with a very low risk of uterine rupture and a high likelihood of success with a trial of labor. Perhaps such additional research will help reverse the current malpractice climate, which is influencing the move by many physicians away from VBAC.
Pearl 6More than 1 prior cesarean increases risk of rupture
The presence of multiple prior cesarean scars places a woman at greater risk for uterine rupture during trial of labor. This risk is likely 3 to 5 times higher than for patients with only 1 prior cesarean delivery.13
Pearl 7Risk of rupture is increased with interdelivery interval of up to 18-24 months
Women who have undergone a cesarean delivery and are contemplating a future trial of labor should be discouraged from becoming pregnant for at least 9 months, possibly up to 15 months.
Interdelivery interval has been shown to be an important contributor to the risk for uterine rupture during trial of labor after cesarean.12-14 Esposito et al14 were the first to note an increased risk of uterine scar failure—including both symptomatic uterine ruptures and asymptomatic uterine scar dehiscences—for those with an interpregnancy interval of less than 6 months.
We showed an increased risk for uterine rupture with an odds ratio of 3.0 (95% CI 1.2-7.2) for those with interdelivery intervals of up to 18 months compared with those who had interdelivery intervals of 19 months or longer.15 More recently, Bujold et al16 confirmed these findings: They found the odds ratio for uterine rupture to be 2.7 (95% CI 1.1- 6.5) for those with an interdelivery interval of up to 24 months.
In a smaller study, Huang et al17 suggested that the success of trial of labor after cesarean may also be lower for those with interdelivery intervals of up to 18 months.
Pearl 8A vaginal delivery before or after prior cesarean lowers risk of rupture
Patients with a prior vaginal delivery are at significantly lower risk for uterine rupture than those without.
We published a study evaluating women with 1 prior cesarean delivery and either a preceding vaginal delivery or a previous VBAC. Our data suggest that women with a prior vaginal delivery were 5 times less likely to experience uterine rupture than those with no prior vaginal deliveries, either before or after the prior cesarean (odds ratio 0.2; 95% CI 0.04-0.8).18
Summary and recommendations
To continue to use vaginal birth after cesarean as an obstetrical practice, we must be better able to identify patients at high and low risk for complications from this procedure, and those who have the greatest chance for success. Women with a nonrecurring indication for the prior cesarean (eg, breech) have the best chance for success. Those with recurring indications for the prior cesarean (eg, failure to progress, morbidly obese women, those with macrosomic fetuses, and those with short interdelivery intervals) may have lower success rates.
Patients contemplating a future trial of labor should consider avoiding pregnancy for at least 9 to 15 months after cesarean delivery.
How can we reduce the risk of uterine rupture in women who are considering a trial of labor after prior cesarean delivery?
- We should not give these women prostaglandins for cervical ripening.
- We must consider allowing a trial of labor for those in spontaneous labor, and be more hesitant about inducing the labor in these patients.
- Women with multiple prior cesareans may also benefit from avoiding a trial of labor.
- Having patients avoid pregnancy for at least 9 months, and maybe up to 15 months, after a cesarean delivery could also assist with decreasing our rate of uterine rupture.
- Keep in mind that those with prior vaginal delivery have a much lower rate of uterine rupture.
The authors report no financial relationships relevant to this article.
Is vaginal birth after cesarean an endangered procedure? Most women who attempt a trial of labor after cesarean have good outcomes, and those at high risk for adverse events, who should be excluded, are becoming increasingly better defined. Yet many physicians eschew this option altogether because of serious concerns about the safety of trial of labor after cesarean.1
If VBAC is to play a role in obstetrical management in the 21st century, we will need to improve our ability to distinguish these 2 populations:
- women with a low risk for complications and a high likelihood of a successful trial of labor, and
- women at high risk for adverse outcomes.
Of many reports documenting risk factors for the most feared complication, uterine rupture, none are from randomized controlled studies of trial of labor after cesarean versus elective repeat cesarean (and such reports are unlikely to be forthcoming). Therefore, we must depend on less rigorously obtained information. Nonetheless, we do have a wealth of data to guide us.
The 8 “pearls” in this article summarize what we know from the available data on vaginal birth after cesarean.
- The repeat cesarean rate varies by prior indication for cesarean: lowest for breech; highest for failure to progress.
- Induced labor has a higher rate of repeat cesarean than spontaneous labor.
- Maternal obesity and fetal macrosomia lower the success rate.
- Induction with oxytocin is associated with an increased risk of uterine rupture, but oxytocin can be used judiciously for augmentation of labor.
- Prostaglandins should not be used for cervical ripening or induction.
- Having more than 1 prior cesarean increases risk of uterine rupture.
- Interdelivery intervals of up to 18 months and maybe even 24 months are associated with an increased risk for uterine rupture. Women should be discouraged from becoming pregnant for at least 9 months, and possibly up to 15 months, after cesarean, if they are contemplating a trial of labor after cesarean for their next delivery.
- Uterine rupture is 5 times less likely in women who have had a vaginal delivery either before or after a prior cesarean delivery.
Pearl 1VBAC rate varies by prior indication
The overall rate of successful vaginal delivery for all women attempting trial of labor after cesarean varies from approximately 60% to 80%.
We showed, however, that the repeat cesarean rate varies by prior indication for cesarean. The rate for those whose prior cesarean was due to breech presentation was 13.9%, which approximated the cesarean rate among nulliparas during the study period (13.5%). The highest rate of repeat cesarean, 37.3%, was for those whose prior cesarean was for failure to progress.2
Pearl 2Repeat cesarean rate is higher with induced labor
Failed induction is a risk for all gravidas having labor induced, regardless of any prior cesarean deliveries. It is especially common among women with an unripe cervix.
Among women with a prior cesarean, labor induction has an approximately 10% increased rate of repeat cesarean, compared with those undergoing spontaneous labor.3
Pearl 3Successful VBAC rate is lower with maternal obesity or fetal macrosomia
Recent evidence suggests that both maternal and fetal weights influence the success of trial of labor after cesarean.
Maternal obesity is associated with a decreased success rate for trial of labor after prior cesarean delivery, but the magnitude of this risk is not well characterized: A success rate of 13% for morbidly obese women has been documented4; more recently, however, a success rate of 57% was reported.5 These studies are limited by their small numbers.
Fetal macrosomia. For pregnancies with fetuses weighing greater than 4,000 g, the literature notes VBAC success rates of 40% to 60%.6,7
The factors most important for counseling a woman about obstetrical management after a prior cesarean are her likelihood of success from trial of labor, and the risks and benefits of both trial of labor and an elective repeat cesarean.
The findings noted in this article call for a scoring system that will more precisely define the risk for an individual patient contemplating trial of labor after prior cesarean.
Over the past 3 decades, multiple studies have attempted to predict the success of trial of labor after cesarean. Flamm and Geiger19 reported a 10-fold predicted difference in the rate of cesarean based on a scoring system incorporating:
- maternal age,
- prior vaginal delivery,
- prior indication for cesarean, and
- the intrapartum assessment of cervical dilation and effacement.
Others have used additional variables including:
- estimated fetal weight,
- gestational age,
- prepregnancy body mass index,
- maternal weight gain, and
- induction of labor.20-23
With further investigation and better identification of those at highest and lowest risk, we will be better able to counsel each patient on her individual risks for a uterine rupture during a trial of labor after prior cesarean delivery.
Pearl 4Risk of rupture is greater with oxytocin induction
Women with a prior cesarean delivery face an increased risk of uterine rupture with labor induction.8,9 Zelop et al8 demonstrated that labor induction with oxytocin is associated with a 4- to 5-fold increased risk of uterine rupture compared to spontaneous labor.
Lydon-Rochelle et al9 reported an increased risk of uterine rupture for those in spontaneous labor and those induced without prostaglandins, compared with women opting for elective repeat cesarean. The odds ratios for patients with spontaneous labor (3.3; 95% confidence interval [CI] 1.8-6.0) and for those with labor induced without prostaglandins (4.9; 95% CI 2.4-9.7) were not statistically significantly different.
Recent trials have suggested that induction of labor is not associated with uterine rupture, though these studies are limited by relatively low numbers of patients. Delaney and Young10 reported rates of uterine rupture of 0.7% for those with induced labor as compared to 0.3% for those with spontaneous labor (P = 0.1). By combining these studies, we see a statistically significant increased rate of uterine rupture approximately twice that of those in spontaneous labor.11
Oxytocin can be used judiciously for augmentation of labor for women with prior cesarean delivery, as it is not associated with an increased risk for uterine rupture in these cases.8
Pearl 5Prostaglandins should not be used for cervical ripening or induction
For patients with a prior cesarean delivery, prostaglandins used for cervical ripening are associated with a significantly higher rate of uterine rupture compared with repeat cesarean, and with either spontaneous labor or induction with oxytocin alone.9
Lydon-Rochelle et al9 demonstrated a 15.6 relative risk (95% CI 8.1-30.0) for uterine rupture among women having labor induced with prostaglandins, compared with those undergoing elective repeat cesarean.
Women with a prior vaginal delivery were 5 times less likely to suffer uterine rupture than those with no prior vaginal deliveries.
At the beginning of the 20th century, Cragin wrote a dictum that is still invoked: “Once a cesarean, always a cesarean.”24
Not often mentioned, however, is that he went on to discuss a patient who had 3 successful vaginal births after cesarean “without difficulty.”24
For much of the 20th century, vaginal birth after cesarean (VBAC) was the exception rather than the rule. Then, 25 years ago, the National Institutes of Health advocated a trial of labor for women who had a prior cesarean delivery. During this time, VBAC was greatly encouraged, and the rate of trials of labor after cesarean began to increase.25 Thus, 15 years ago, the cesarean delivery rate in the United States began to fall after an unprecedented rise during the previous decades.
A few years later, however, this trend ceased and the cesarean delivery rate once again began to rise. This switch can be attributed to both an increase in primary cesarean deliveries and a decrease in the VBAC rate.26 It coincides with published data on uterine rupture associated with a trial of labor after cesarean: Two case series published in 1991 together documented 20 uterine ruptures with 4 perinatal deaths, 4 neonates with neurological impairment, and 3 women who underwent hysterectomy due to the event.27,28
Further study is needed to more precisely identify those women at high risk for uterine rupture and low risk of success of a trial of labor, and also—perhaps more importantly—those women with a very low risk of uterine rupture and a high likelihood of success with a trial of labor. Perhaps such additional research will help reverse the current malpractice climate, which is influencing the move by many physicians away from VBAC.
Pearl 6More than 1 prior cesarean increases risk of rupture
The presence of multiple prior cesarean scars places a woman at greater risk for uterine rupture during trial of labor. This risk is likely 3 to 5 times higher than for patients with only 1 prior cesarean delivery.13
Pearl 7Risk of rupture is increased with interdelivery interval of up to 18-24 months
Women who have undergone a cesarean delivery and are contemplating a future trial of labor should be discouraged from becoming pregnant for at least 9 months, possibly up to 15 months.
Interdelivery interval has been shown to be an important contributor to the risk for uterine rupture during trial of labor after cesarean.12-14 Esposito et al14 were the first to note an increased risk of uterine scar failure—including both symptomatic uterine ruptures and asymptomatic uterine scar dehiscences—for those with an interpregnancy interval of less than 6 months.
We showed an increased risk for uterine rupture with an odds ratio of 3.0 (95% CI 1.2-7.2) for those with interdelivery intervals of up to 18 months compared with those who had interdelivery intervals of 19 months or longer.15 More recently, Bujold et al16 confirmed these findings: They found the odds ratio for uterine rupture to be 2.7 (95% CI 1.1- 6.5) for those with an interdelivery interval of up to 24 months.
In a smaller study, Huang et al17 suggested that the success of trial of labor after cesarean may also be lower for those with interdelivery intervals of up to 18 months.
Pearl 8A vaginal delivery before or after prior cesarean lowers risk of rupture
Patients with a prior vaginal delivery are at significantly lower risk for uterine rupture than those without.
We published a study evaluating women with 1 prior cesarean delivery and either a preceding vaginal delivery or a previous VBAC. Our data suggest that women with a prior vaginal delivery were 5 times less likely to experience uterine rupture than those with no prior vaginal deliveries, either before or after the prior cesarean (odds ratio 0.2; 95% CI 0.04-0.8).18
Summary and recommendations
To continue to use vaginal birth after cesarean as an obstetrical practice, we must be better able to identify patients at high and low risk for complications from this procedure, and those who have the greatest chance for success. Women with a nonrecurring indication for the prior cesarean (eg, breech) have the best chance for success. Those with recurring indications for the prior cesarean (eg, failure to progress, morbidly obese women, those with macrosomic fetuses, and those with short interdelivery intervals) may have lower success rates.
Patients contemplating a future trial of labor should consider avoiding pregnancy for at least 9 to 15 months after cesarean delivery.
How can we reduce the risk of uterine rupture in women who are considering a trial of labor after prior cesarean delivery?
- We should not give these women prostaglandins for cervical ripening.
- We must consider allowing a trial of labor for those in spontaneous labor, and be more hesitant about inducing the labor in these patients.
- Women with multiple prior cesareans may also benefit from avoiding a trial of labor.
- Having patients avoid pregnancy for at least 9 months, and maybe up to 15 months, after a cesarean delivery could also assist with decreasing our rate of uterine rupture.
- Keep in mind that those with prior vaginal delivery have a much lower rate of uterine rupture.
The authors report no financial relationships relevant to this article.
1. Greene MF. Vaginal delivery after cesarean section—is the risk acceptable? N Engl J Med. 2001;345:54-55.
2. Shipp TD, Zelop CM, Repke JT, Cohen A, Caughey AB, Lieberman E. Labor after previous cesarean: influence of prior indication and parity. Obstet Gynecol. 2000;95:913-916.
3. Zelop CM, Shipp TD, Cohen A, Repke JT, Lieberman E. Trial of labor after 40 weeks’ gestation in women with prior cesarean. Obstet Gynecol. 2001;97:391-393.
4. Chauhan SP, Magann EF, Carroll CS, Barrilleaux PS, Scardo JA, Martin, Jr. JN. Mode of delivery for the morbidly obese with prior cesarean delivery: vaginal versus repeat cesarean section. Am J Obstet Gynecol. 2001;185:349-354.
5. Edwards RK, Harnsberger DS, Johnson IM, Treloar RW, Cruz AC. Deciding on route of delivery for obese women with a prior cesarean delivery. Am J Obstet Gynecol. 2003;189:385-390.
6. Zelop CM, Shipp TD, Repke JT, Cohen A, Lieberman E. Outcomes of trial of labor following previous cesarean delivery among women with fetuses weighing >4000 g. Am J Obstet Gynecol. 2001;185:903-905.
7. Elkousy MA, Sammel M, Stevens E, Peipert JF, Macones G. The effect of birth weight on vaginal birth after cesarean delivery success rates. Am J Obstet Gynecol. 2003;188:824-830.
8. Zelop CM, Shipp TD, Repke JT, Cohen A, Caughey AB, Lieberman E. Uterine rupture during induced or augmented labor in gravid women with one prior cesarean delivery. Am J Obstet Gynecol. 1999;181:882-886.
9. Lydon-Rochelle M, Holt VL, Easterling TR, Martin DP. Risk of uterine rupture during labor among women with a prior cesarean delivery. N Engl J Med. 2001;345:3-8.
10. Delaney T, Young DC. Spontaneous versus induced labor after a previous cesarean delivery. Obstet Gynecol. 2003;102:39-44.
11. Shipp TD. Prostaglandin E2, oxytocin may raise risk of rupture after previous cesarean. OBG Management. March 2004;16:14-16.
12. The American College of Obstetricians and Gynecologists Committee on Obstetric Practice Induction of labor for vaginal birth after cesarean delivery. ACOG Committee Opinion No. 271. Washington DC: ACOG; 2002.
13. Caughey AB, Shipp TD, Repke JT, Zelop CM, Cohen A, Lieberman E. Rate of uterine rupture during a trial of labor in women with one or two prior cesarean deliveries. Am J Obstet Gynecol. 1999;181:872-876.
14. Esposito MA, Menihan CA, Malee MP. Association of interpregnancy interval with uterine scar failure in labor: a case-control study. Am J Obstet Gynecol. 2000;183:1180-1183.
15. Shipp TD, Zelop CM, Repke JT, Cohen A, Lieberman E. Interdelivery interval and risk of symptomatic uterine rupture. Obstet Gynecol. 2001;97:175-177.
16. Bujold E, Mehta SH, Bujold C, Gauthier RJ. Interdelivery interval and uterine rupture. Am J Obstet Gynecol. 2002;187:1199-1202.
17. Huang WH, Nakashima DK, Rumney PJ, Keegan KA, Jr, Chan K. Interdelivery interval and the success of vaginal birth after cesarean delivery. Obstet Gynecol. 2002;99:41-44.
18. Zelop CM, Shipp TD, Repke JT, Cohen A, Lieberman E. Effect of previous vaginal delivery on the risk of uterine rupture during a subsequent trial of labor. Am J Obstet Gynecol. 2000;183:1184-1186.
19. Flamm BL, Geiger AM. Vaginal birth after cesarean delivery: an admission scoring system. Obstet Gynecol. 1997;90:907-910.
20. Dhall K, Mittal SC, Grover V, Dhall GI. Childbirth following primary cesarean section—evaluation of a scoring system. Int J Gynaecol Obstet. 1987;25:199-205.
21. Pickhardt MG, Martin JN, Jr, Meydrech EF, et al. Vaginal birth after cesarean delivery: are there useful and valid predictors of success or failure? Am J Obstet Gynecol. 1992;166:1811-1819.
22. Troyer LR, Parisi VM. Obstetric parameters affecting success in a trial of labor: designation of a scoring system. Am J Obstet Gynecol. 1992;167:1099-1104.
23. Macones GA, Hausman N, Edelstein R, Stamilio DM, Marder SJ. Predicting outcomes of trials of labor in women attempting vaginal birth after cesarean delivery: a comparison of multivariate methods with neural networks. Am J Obstet Gynecol. 2001;184:409-413.
24. Cragin EB. Conservatism in obstetrics. New York Med J. 1916;104:1-3.
25. Harer WB, Jr. Vaginal birth after cesarean delivery: current status. JAMA. 2002;287:2627-2630.
26. National Vital Statistics Reports, Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics, National Vital Statistics System. Births: Final data for 2001. December 2002;51(2).-
27. Scott JR. Mandatory trial of labor after cesarean delivery: an alternative viewpoint. Obstet Gynecol. 1991;77:811-814.
28. Jones RO, Nagashima AW, Hartnett-Goodman MM, Goodlin RC. Rupture of low transverse cesarean scars during trial of labor. Obstet Gynecol. 1991;77:815-817.
1. Greene MF. Vaginal delivery after cesarean section—is the risk acceptable? N Engl J Med. 2001;345:54-55.
2. Shipp TD, Zelop CM, Repke JT, Cohen A, Caughey AB, Lieberman E. Labor after previous cesarean: influence of prior indication and parity. Obstet Gynecol. 2000;95:913-916.
3. Zelop CM, Shipp TD, Cohen A, Repke JT, Lieberman E. Trial of labor after 40 weeks’ gestation in women with prior cesarean. Obstet Gynecol. 2001;97:391-393.
4. Chauhan SP, Magann EF, Carroll CS, Barrilleaux PS, Scardo JA, Martin, Jr. JN. Mode of delivery for the morbidly obese with prior cesarean delivery: vaginal versus repeat cesarean section. Am J Obstet Gynecol. 2001;185:349-354.
5. Edwards RK, Harnsberger DS, Johnson IM, Treloar RW, Cruz AC. Deciding on route of delivery for obese women with a prior cesarean delivery. Am J Obstet Gynecol. 2003;189:385-390.
6. Zelop CM, Shipp TD, Repke JT, Cohen A, Lieberman E. Outcomes of trial of labor following previous cesarean delivery among women with fetuses weighing >4000 g. Am J Obstet Gynecol. 2001;185:903-905.
7. Elkousy MA, Sammel M, Stevens E, Peipert JF, Macones G. The effect of birth weight on vaginal birth after cesarean delivery success rates. Am J Obstet Gynecol. 2003;188:824-830.
8. Zelop CM, Shipp TD, Repke JT, Cohen A, Caughey AB, Lieberman E. Uterine rupture during induced or augmented labor in gravid women with one prior cesarean delivery. Am J Obstet Gynecol. 1999;181:882-886.
9. Lydon-Rochelle M, Holt VL, Easterling TR, Martin DP. Risk of uterine rupture during labor among women with a prior cesarean delivery. N Engl J Med. 2001;345:3-8.
10. Delaney T, Young DC. Spontaneous versus induced labor after a previous cesarean delivery. Obstet Gynecol. 2003;102:39-44.
11. Shipp TD. Prostaglandin E2, oxytocin may raise risk of rupture after previous cesarean. OBG Management. March 2004;16:14-16.
12. The American College of Obstetricians and Gynecologists Committee on Obstetric Practice Induction of labor for vaginal birth after cesarean delivery. ACOG Committee Opinion No. 271. Washington DC: ACOG; 2002.
13. Caughey AB, Shipp TD, Repke JT, Zelop CM, Cohen A, Lieberman E. Rate of uterine rupture during a trial of labor in women with one or two prior cesarean deliveries. Am J Obstet Gynecol. 1999;181:872-876.
14. Esposito MA, Menihan CA, Malee MP. Association of interpregnancy interval with uterine scar failure in labor: a case-control study. Am J Obstet Gynecol. 2000;183:1180-1183.
15. Shipp TD, Zelop CM, Repke JT, Cohen A, Lieberman E. Interdelivery interval and risk of symptomatic uterine rupture. Obstet Gynecol. 2001;97:175-177.
16. Bujold E, Mehta SH, Bujold C, Gauthier RJ. Interdelivery interval and uterine rupture. Am J Obstet Gynecol. 2002;187:1199-1202.
17. Huang WH, Nakashima DK, Rumney PJ, Keegan KA, Jr, Chan K. Interdelivery interval and the success of vaginal birth after cesarean delivery. Obstet Gynecol. 2002;99:41-44.
18. Zelop CM, Shipp TD, Repke JT, Cohen A, Lieberman E. Effect of previous vaginal delivery on the risk of uterine rupture during a subsequent trial of labor. Am J Obstet Gynecol. 2000;183:1184-1186.
19. Flamm BL, Geiger AM. Vaginal birth after cesarean delivery: an admission scoring system. Obstet Gynecol. 1997;90:907-910.
20. Dhall K, Mittal SC, Grover V, Dhall GI. Childbirth following primary cesarean section—evaluation of a scoring system. Int J Gynaecol Obstet. 1987;25:199-205.
21. Pickhardt MG, Martin JN, Jr, Meydrech EF, et al. Vaginal birth after cesarean delivery: are there useful and valid predictors of success or failure? Am J Obstet Gynecol. 1992;166:1811-1819.
22. Troyer LR, Parisi VM. Obstetric parameters affecting success in a trial of labor: designation of a scoring system. Am J Obstet Gynecol. 1992;167:1099-1104.
23. Macones GA, Hausman N, Edelstein R, Stamilio DM, Marder SJ. Predicting outcomes of trials of labor in women attempting vaginal birth after cesarean delivery: a comparison of multivariate methods with neural networks. Am J Obstet Gynecol. 2001;184:409-413.
24. Cragin EB. Conservatism in obstetrics. New York Med J. 1916;104:1-3.
25. Harer WB, Jr. Vaginal birth after cesarean delivery: current status. JAMA. 2002;287:2627-2630.
26. National Vital Statistics Reports, Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics, National Vital Statistics System. Births: Final data for 2001. December 2002;51(2).-
27. Scott JR. Mandatory trial of labor after cesarean delivery: an alternative viewpoint. Obstet Gynecol. 1991;77:811-814.
28. Jones RO, Nagashima AW, Hartnett-Goodman MM, Goodlin RC. Rupture of low transverse cesarean scars during trial of labor. Obstet Gynecol. 1991;77:815-817.
Smoking Cessation for Patients with Psychotic Disorders
Redesigning the Fall Incident Report
Prognostic Value of Computer Electrocardiography in Veteran Outpatients
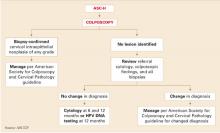
• Improved cytology reports • New guidelines: ASC-US triage, HPV testing • ALTS findings • Breakthrough on HPV vaccine
A 2001 adjustment in the terminology used to report cytology results was the first of 4 recent advancements that, in sum, have fundamentally changed how we interpret and follow-up Pap smears. Another breakthrough in cervical disease reveals clear potential for a vaccine for human papillomavirus (HPV). This Update on Cervical Disease reviews these pivotal developments:
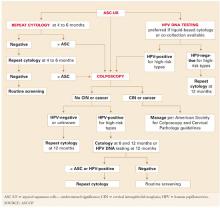
- The 2001 revision of the Bethesda System—also known as Bethesda 3—for reporting cervical cytologic results.
- The 2001 consensus guidelines on managing women with abnormal cytology and cer-vical cancer precursors.
- Interim guidance on the use of HPV DNA testing as an adjunct to cervical cytology for screening.
- Findings of the National Cancer Institute’s ASCUS/LSIL Triage Study (ALTS).
- A proof-of-principle trial demonstrating the potential clinical utility of a vaccine for HPV type 16 (HPV-16) in young adults.
New terms aim to reduce unnecessary repeat Paps
Solomon D, Davey D, Kurman R, et al. The 2001 Bethesda System: terminology for reporting results of cervical cytology. JAMA. 2002;287:2114-2119.
By deleting a few categories, changing several definitions, and adding new information to the report, the new Bethesda System for reporting results of screening cytology has created a simpler, more precise, and more helpful report.
Elements of the widely utilized 1991 system were believed to cause confusion and lead to unnecessary repeat testing, as well as undue concerns and expenses; these have been refined. Key original features that served well, however, remain. For example, results are still shown in 3 distinct sections: specimen adequacy; general interpretation of normal or abnormal; and specific interpretation, or type of abnormality, if present.
Simpler classifications with notes added when indicated are a step forward from the standpoint of physicians and patients alike.
- Specimen adequacy is now either “satisfactory” or “unsatisfactory” for evaluation. The confusing category “satisfactory but limited by …” was eliminated because it seemed to lead to many unnecessary repeat Pap tests. Under the old system, specimens were categorized as “satisfactory but limited by …” for any number of reasons, including lack of a transformation-zone component, partially obscuring blood, inflammation, or poor presentation. Such specimens are now classified as “satisfactory for evaluation,” and a statement describing any flaws in the specimen is added.
- “Benign cellular changes,” a classification that confused patients, was also appropriately eliminated. Cases that fell into that category under the old system are now classified as “negative for intraepithelial lesion or malignancy” (ie, normal), and include a statement explaining that organisms, reparative changes, radiation effect, atrophy, or other conditions are present.
ASCUS was changed to “atypical squamous cells” (ASC), with 2 subcategories: “undetermined significance” (ASC-US) and “suggestive of a high-grade squamous intraepithelial lesion” (ASC-H) (Atypical squamous cells: The case for HPV testing”
A 2001 adjustment in the terminology used to report cytology results was the first of 4 recent advancements that, in sum, have fundamentally changed how we interpret and follow-up Pap smears. Another breakthrough in cervical disease reveals clear potential for a vaccine for human papillomavirus (HPV). This Update on Cervical Disease reviews these pivotal developments:
- The 2001 revision of the Bethesda System—also known as Bethesda 3—for reporting cervical cytologic results.
- The 2001 consensus guidelines on managing women with abnormal cytology and cer-vical cancer precursors.
- Interim guidance on the use of HPV DNA testing as an adjunct to cervical cytology for screening.
- Findings of the National Cancer Institute’s ASCUS/LSIL Triage Study (ALTS).
- A proof-of-principle trial demonstrating the potential clinical utility of a vaccine for HPV type 16 (HPV-16) in young adults.
New terms aim to reduce unnecessary repeat Paps
Solomon D, Davey D, Kurman R, et al. The 2001 Bethesda System: terminology for reporting results of cervical cytology. JAMA. 2002;287:2114-2119.
By deleting a few categories, changing several definitions, and adding new information to the report, the new Bethesda System for reporting results of screening cytology has created a simpler, more precise, and more helpful report.
Elements of the widely utilized 1991 system were believed to cause confusion and lead to unnecessary repeat testing, as well as undue concerns and expenses; these have been refined. Key original features that served well, however, remain. For example, results are still shown in 3 distinct sections: specimen adequacy; general interpretation of normal or abnormal; and specific interpretation, or type of abnormality, if present.
Simpler classifications with notes added when indicated are a step forward from the standpoint of physicians and patients alike.
- Specimen adequacy is now either “satisfactory” or “unsatisfactory” for evaluation. The confusing category “satisfactory but limited by …” was eliminated because it seemed to lead to many unnecessary repeat Pap tests. Under the old system, specimens were categorized as “satisfactory but limited by …” for any number of reasons, including lack of a transformation-zone component, partially obscuring blood, inflammation, or poor presentation. Such specimens are now classified as “satisfactory for evaluation,” and a statement describing any flaws in the specimen is added.
- “Benign cellular changes,” a classification that confused patients, was also appropriately eliminated. Cases that fell into that category under the old system are now classified as “negative for intraepithelial lesion or malignancy” (ie, normal), and include a statement explaining that organisms, reparative changes, radiation effect, atrophy, or other conditions are present.
ASCUS was changed to “atypical squamous cells” (ASC), with 2 subcategories: “undetermined significance” (ASC-US) and “suggestive of a high-grade squamous intraepithelial lesion” (ASC-H) (Atypical squamous cells: The case for HPV testing”
A 2001 adjustment in the terminology used to report cytology results was the first of 4 recent advancements that, in sum, have fundamentally changed how we interpret and follow-up Pap smears. Another breakthrough in cervical disease reveals clear potential for a vaccine for human papillomavirus (HPV). This Update on Cervical Disease reviews these pivotal developments:
- The 2001 revision of the Bethesda System—also known as Bethesda 3—for reporting cervical cytologic results.
- The 2001 consensus guidelines on managing women with abnormal cytology and cer-vical cancer precursors.
- Interim guidance on the use of HPV DNA testing as an adjunct to cervical cytology for screening.
- Findings of the National Cancer Institute’s ASCUS/LSIL Triage Study (ALTS).
- A proof-of-principle trial demonstrating the potential clinical utility of a vaccine for HPV type 16 (HPV-16) in young adults.
New terms aim to reduce unnecessary repeat Paps
Solomon D, Davey D, Kurman R, et al. The 2001 Bethesda System: terminology for reporting results of cervical cytology. JAMA. 2002;287:2114-2119.
By deleting a few categories, changing several definitions, and adding new information to the report, the new Bethesda System for reporting results of screening cytology has created a simpler, more precise, and more helpful report.
Elements of the widely utilized 1991 system were believed to cause confusion and lead to unnecessary repeat testing, as well as undue concerns and expenses; these have been refined. Key original features that served well, however, remain. For example, results are still shown in 3 distinct sections: specimen adequacy; general interpretation of normal or abnormal; and specific interpretation, or type of abnormality, if present.
Simpler classifications with notes added when indicated are a step forward from the standpoint of physicians and patients alike.
- Specimen adequacy is now either “satisfactory” or “unsatisfactory” for evaluation. The confusing category “satisfactory but limited by …” was eliminated because it seemed to lead to many unnecessary repeat Pap tests. Under the old system, specimens were categorized as “satisfactory but limited by …” for any number of reasons, including lack of a transformation-zone component, partially obscuring blood, inflammation, or poor presentation. Such specimens are now classified as “satisfactory for evaluation,” and a statement describing any flaws in the specimen is added.
- “Benign cellular changes,” a classification that confused patients, was also appropriately eliminated. Cases that fell into that category under the old system are now classified as “negative for intraepithelial lesion or malignancy” (ie, normal), and include a statement explaining that organisms, reparative changes, radiation effect, atrophy, or other conditions are present.
ASCUS was changed to “atypical squamous cells” (ASC), with 2 subcategories: “undetermined significance” (ASC-US) and “suggestive of a high-grade squamous intraepithelial lesion” (ASC-H) (Atypical squamous cells: The case for HPV testing”
Fetal pulse oximetry: 8 vital questions
- The value of this new technology might not be so much the prediction of acidosis but identification of the well-oxygenated fetus so that labor may be safely continued in the presence of a concerning—but not ominous—fetal heart rate tracing.
- The only randomized study published so far did not determine whether clinical decisions can be based solely on fetal pulse oximetry. The investigators did suggest that sensitivity and specificity for metabolic acidemia was improved in the intervention group—a promising appraisal, in contrast with previous observational data.
When a teenage nullipara underwent labor induction for preeclampsia at 37 weeks, she was given epidural analgesia and seizure prophylaxis with magnesium sulfate. Her electronic fetal heart rate (FHR) tracing was initially reassuring, with only occasional variable decelerations, but subsequently revealed a baseline of 140 beats per minute (bpm), minimal to absent variability, no accelerations, and variable decelerations to 90 bpm with rapid return to baseline.
The tracing was interpreted as nonreassuring, and a fetal pulse oximeter was inserted. It revealed a fetal oxygen saturation rate between 45% and 50%, and labor was allowed to continue. After 3.5 hours in the second stage, the patient was delivered by outlet forceps. Her infant had Apgar scores of 8 at 1 minute and 9 at 5 minutes. The umbilical arterial pH was 7.25, and base excess was–4.9.
Fetal pulse oximetry made it possible to manage this case without resorting to emergent cesarean. But is this noninvasive technology truly a step forward in intrapartum assessment of fetal well-being?
We describe what the evidence (a single randomized study and a number of observational studies) reveals about these questions:
- How accurately does fetal pulse oximetry reflect the fetal condition?
- What is the critical threshold for fetal oxygen desaturation?
- Is a single reading reliable?
- Does oximetry correlate with acid-base status?
- Does the combination of oximetry and electronic monitoring improve accuracy?
- Will fetal pulse oximetry improve neonatal outcomes?
- How precise is it?
- Is it easy to use?
Needed: Effective adjunct to electronic monitoring
Except in the chronically hypoxic fetus (which is affected by the time labor begins), the pathophysiology of acute intrapartum events is a continuum, from hypoxemia to respiratory acidosis to metabolic acidosis and, ultimately, clinical impairment. The goal of intrapartum surveillance is to detect fetal hypoxemia before it progresses to asphyxia and perinatal mortality or long-term morbidity.
Although it is approved as an adjunct to electronic fetal monitoring (EFM), fetal pulse oximetry has gained only sporadic use since it became available in the United States in 2000—even though EFM has proved disappointing as a tool for predicting fetal hypoxia. Only about 10% of US obstetrical units had fetal pulse oximetry technology as of 2002.1
Clinicians began questioning the reliability of subjective interpretation of fetal heart tracings soon after EFM went into general use. Thirty years later, a meta-analysis of 12 randomized clinical trials involving 58,855 gravidas cast doubt on the benefits of EFM,2 which is associated with an increase in operative deliveries as a result of high sensitivity but low specificity in predicting fetal hypoxia and acidosis.
FDA approval was based on sole randomized trial
The only commercially available fetal oximetry sensor, the Nellcor N-400 (Nellcor, Pleasanton, Calif), obtained US Food and Drug Administration (FDA) approval as an adjunct to EFM when the latter indicates a nonreassuring FHR pattern. That approval was based on the only randomized study3 of fetal pulse oximetry conducted, which involved 1,010 women with predefined nonreassuring FHR patterns in labor.
Goal: Reduced cesarean rate with comparable outcomes. Investigators hypothesized that adjunctive fetal oximetry would improve assessment and reduce the cesarean rate without altering neonatal outcome. Indeed, in the oximetry group, the rate of cesarean delivery performed for a nonreassuring FHR tracing (4.5% versus 10.2%; P = .007) was significantly reduced. Other findings:
- Same neonatal outcomes, with no significant differences between the 2 groups.
- Higher cesarean rate for dystocia in the intervention group, offsetting any advantage in the overall cesarean delivery rate (29% versus 26%). This unexpected increase in cesarean deliveries raises several possibilities:
- Given the unblinded design, it is possible that clinicians, circumspect of the pulse oximetry, continued to perform cesareans for nonreassuring FHR, but labeled the indication for surgery differently. The validity of the dystocia diagnosis was discredited by a subsequent partogram analysis that showed a similar rate of arrested labor in both groups.
- A nonreassuring FHR in conditions of normal fetal oxygenation is predictive of dystocia. Previous randomized studies of EFM have suggested the same thing.4
- Dystocia is the consequence of the device itself. Anecdotal observations suggest a higher rate of persistent occiput posterior positions with fetal oximetry.
Other trials underway. The ongoing Fetal Oximetry (FOX) trial of the National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network, involving 10,000 nulliparous participants, is comparing cesarean delivery rates and safety outcomes in patients monitored for FHR plus pulse oximetry with a group in which the clinicians are blinded to the pulse oximetry readings. Another randomized controlled trial of fetal pulse oximetry is underway in Australia.
Potential for increased costs. The American College of Obstetricians and Gynecologists (ACOG) has raised concerns about the potential increase in costs without demonstrable improvement in outcome.5 ACOG has not endorsed fetal pulse oximetry for general practice.
Question 1How accurately does pulse oximetry reflect the fetal condition?
It yields only indirect information on the partial pressure of oxygen in the blood and no data on perfusion or acid-base status.
In other clinical settings, oxygen saturation is not an acceptable substitute for arterial blood gas analysis. The pulse oximeter is not a hemoximeter—only that device directly and reliably determines blood oxygen saturation by spectrophotometry.6 Even the calculated oxygen saturation values provided automatically by modern blood gas analyzers are inaccurate.7
Studies report varying results. In a comparison8 of fetal oxygen saturation by hemoximetry in a fetal scalp blood (FSB) sample and fetal arterial oxyhemoglobin saturation (FSpO2) by pulse oximetry immediately before the blood sampling, the FSpO2 medians were always higher than the FSB hemoximetry saturation—which led to false-negative results in hypoxic babies.
In animal studies, pulse oximetry correlated well with simultaneously measured arterial oxygen saturation (r = 0.98, P = .01),9 but data from human studies are inconsistent. While McNamara et al10 reported good correlation between FSpO2 measurements and umbilical artery blood oxygen saturation at birth (r = 0.59, P <.001), Langer et al11 found no relationship between FSpO2 levels determined during pushing efforts and oxygen saturation in umbilical vein blood at birth.
Possible reasons for the ambiguous findings:
- differences in practice, such as use of umbilical venous versus arterial blood, or measurement during pushing versus between pushes,
- different intervals from FSpO2 reading to umbilical blood sampling, or
- incomparable groups, such as all women in labor versus those with abnormal FHR.
Limitations. Fetal pulse oximetry measures arterial oxygen saturation during the systolic pulse wave in the skin microcirculation at head level. In the fetus, this is part of the preductal circulation, with oxygen saturation levels somewhere between umbilical arterial and umbilical venous blood oxygen saturation.
Theoretically, FSpO2 should be closer to FSB than to umbilical blood. Although FSB samples consist of capillary blood, which is not exactly central arterial blood, the differences are small, at least in the neonate.12 In the intrapartum period, however, several variables with unknown effect may weaken relationships:
- different intervals between the last oximetry signal and blood sampling after delivery
- differences in local tissue perfusion status13
- perfusion changes during fetal compromise, as the fetus centralizes its blood flow, with vasoconstriction in the skin circulation
Question 2What is the critical threshold for fetal oxygen desaturation?
Human studies indicate that an FSpO2 of 33% is approximately the 10th percentile on the normal distribution, and an FSpO2 of 29% to 30% represents the third to fifth percentiles in normal-outcome labor.14 Studies in catheterized fetal sheep suggest that the level below which metabolic acidosis can be anticipated is an FSpO2 of about 30%.15
The 30% threshold also is supported by prospective human data from a multicenter trial.16 According to those data, an FSpO2 of less than 30% has 100% sensitivity in predicting an FSB pH below 7.20. FSpO2 of less than 30% also correlated with a lack of variability on the FHR tracing.17
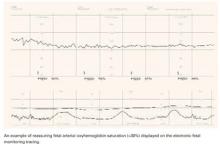
The cutoff of 30% should not be interpreted as an indication of fetal distress, however. Rather, it represents a threshold below which increasing fetal acidosis will be encountered ( FIGURE 1). Oxygen saturation is a dynamic biologic parameter with broad variation.
FIGURE 1 Tracking fetal arterial oxyhemoglobin saturation
Question 3Is a single reading reliable?
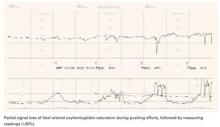
The normal fetus has a remarkable capacity to compensate for transient episodes of desaturation. Thus, a single reading cannot reflect the fetal condition; the trend in FSpO2 must be taken into account. Research indicates only FSpO2 levels below 30% for more than 2 minutes18 or more than 10 minutes19 are likely to be associated with intrapartum acidosis.
ACOG has raised concerns about the potential increase in costs without demonstrable improvement in outcome.
Gorenberg et al20 retrospectively correlated FSpO2 with umbilical artery pH and found that neither the 30% threshold alone nor the duration of FSpO2 below 30% correlated with fetal acidemia (pH below 7.20). Rather, the repetition of such episodes was more predictive. The authors concluded that more than 10 episodes of FSpO2below 30% would overcome the ability of the fetus to compensate.
The study was underpowered to detect a significant difference in acidemia, and did not allow sufficient observation time to detect the natural progression of hypoxia to metabolic acidosis, a better indicator of fetal compromise. Additional research is needed.
Question 4Does oximetry correlate with acid-base status?
Many of the studies mentioned here assumed a correlation. Whenever oxygen saturation in the umbilical artery is 30% or more, acidosis (pH below 7.13) in the same blood is rare—only 1%.21 However, the correlation between fetal pulse oximetry values and acid-base status is much weaker.8.
Leszczynska-Gorzelak et al22 found no relationship between FSpO2 levels in the first or second stage of labor and pH or partial pressure of oxygen in umbilical vein blood at delivery. Other investigators concluded similarly, considering intrapartum FSpO2 of limited use for predicting acidosis at birth, irrespective of FSpO2 cutoff.23,24
Rijnders et al24 found no significant correlation between fetal scalp or umbilical artery blood pH and mean FSpO2 for the last 30 minutes before sampling (r = 0.02, P = .9). Even the lowest FSpO2 level did not correlate with arterial pH (r = .04, P = .84). None of the study’s 3 cases of umbilical pH below 7.05 would have been detected using the mean FSpO2 before delivery, and only 1 would have been detected using the lowest FSpO2.
In another multicenter study involving the Nellcor system in 164 cases with abnormal FHR, a correlation between oximetry and FSB sampling (r = 0.29, P < .01) was noted in the first stage of labor, but second-stage FSpO2 readings did not correlate with oxygen saturation, partial pressure of oxygen, pH, or bicarbonate level in the umbilical artery at birth.25
An observational series26 of 128 fetuses with nonreassuring FHR patterns concluded that fetal distress was insufficiently identified by oximetry. Only 2 of the 11 cases with umbilical artery pH below 7.20 were detected by pulse oximetry recordings below 30% during the last 30 minutes of the second stage, and out of 5 cases with hypoxic readings in the second stage, only 2 were acidotic at birth. The calculated sensitivity was 18%, specificity 92%, positive predictive value (PPV) 40%, and negative predictive value (NPV) 80%. A low Apgar score was never predicted by fetal pulse oximetry.
Others used the same Nellcor system over the final 30 minutes of labor and a cutoff for umbilical blood acidemia of pH below 7.13 and reported similar numbers: sensitivity 28%, specificity 94%, PPV 40%, and NPV 80%.23
Vitoratos et al27 analyzed FSpO2 readings in active labor (not limited to the last 30 minutes before delivery) and obtained somewhat better values: sensitivity 72%, specificity 93%, PPV 61.5%, and NPV 95.8% for an umbilical artery blood pH below 7.15.
The impression that the validity of fetal pulse oximetry is higher in earlier labor than in the second stage is supported by data from Stiller et al. 28 Leszczynska-Gorzelak et al29 found a significant decrease in mean FSpO2 from the first stage to the second stage of normal labor (51.9% versus 43.8%, P < .001), and Dildy et al14 noted a similar difference upon analyzing 160 normal labors (59% versus 53%), but other studies failed to verify such differences.25,30
Observational studies had unrealistic pH cutoff. All the evidence presented thus far on the validity of fetal pulse oximetry in predicting acidemia is based on observational data. A common deficiency is the unrealistic cutoff for pathologic fetal acidemia—a pH of less than 7.13 to 7.20—when it is widely accepted that “pathologic fetal acidemia” reflects an umbilical artery blood pH below 7.31 Even in this group, two thirds of neonates are unaffected by morbidity.
Need to identify metabolic acidosis. It also is accepted that the presence of a metabolic component to fetal acidemia may be as important—if not more important—than a single pH cutoff.31 Only a few human studies of pulse oximetry have distinguished between respiratory and metabolic acidemia. When they did, intrapartum fetal pulse oximetry was unable to predict umbilical artery base excess.23,25
The only randomized study failed to determine whether clinical decisions can be based solely on fetal pulse oximetry. 3 The investigators did suggest that sensitivity and specificity for metabolic acidemia was improved in the intervention group—a promising appraisal, in contrast with previous observational data.
In the study, 7 neonates (3 in the intervention group and 4 controls) had umbilical artery blood pH below 7. All 4 controls had vaginal delivery. There also were 6 cases of elevated base excess (ie, -16 mEq/L or below) among controls. None were recorded in the intervention group, and the 3 cases of acidemia were recognized antepartum and led to cesareans.
Unfortunately, the study design did not guarantee that patient management was based exclusively on EFM with or without fetal pulse oximetry. Vibro-acoustic stimulation or FSB sampling was required before proceeding to cesarean delivery in both groups.
It appears that the negative predictive value of fetal oximetry is of greater practical value than other attributes.
When FSpO2 was less than 30% for the entire interval between 2 contractions, or was unobtainable, the physician was supposed to revert to interpretation of EFM. When that was persistently nonreassuring, the physician was given the option of scalp stimulation or FSB sampling. Thus, it was not determined whether clinical decisions can be based exclusively on fetal pulse oximetry. Schmidt et al26 suggested that such exclusive application of fetal pulse oximetry might actually jeopardize fetal health.
Question 5 Does the combination of oximetry and EFM improve accuracy?
Fetal pulse oximetry was not used independently in any of the studies discussed here, but in association with EFM, which has a sensitivity for fetal acidosis of 93%, specificity of 29%, PPV of 2.6%, and NPV of 99.5%.32
From a statistical point of view, whenever 2 evaluation methods with the same endpoint (fetal acidosis) are combined, sensitivity decreases while specificity increases, theoretically resulting in less unnecessary intervention. That is exactly what investigators have reported: sensitivity as low as 18%26 for fetal oximetry, and specificity as high as 94%.23 However, the value of this new technology might not be so much the prediction of acidosis but identification of the well-oxygenated fetus so that labor may be safely continued in the presence of a concerning—but not ominous—FHR tracing.
Fetal pulse oximetry employs principles of optical spectrophotometry and plethysmography to provide information on the percentage of oxygen bound to hemoglobin. Oxyhemoglobin (oxygenated hemoglobin) and deoxyhemoglobin (hemoglobin without oxygen) absorb red and infrared light differently: more red absorption by deoxyhemoglobin, and more infrared absorption by oxyhemoglobin.
By measuring the relative absorption at each wavelength, the fraction of hemoglobin that carries oxygen can be determined. The arterial oxygen saturation is expressed as a percentage. The technology has been refined to measure fetal arterial oxyhemoglobin saturation during labor.
Pulse oximetry sensors must be calibrated for fetal biological values. In the fetus, normal oxygen saturation is much lower than in the adult or neonate; hemoglobin has a higher affinity for oxygen and is in higher concentration; and there are more capillaries per unit of tissue, higher cardiac output, and a higher heart rate.
In the adult or neonate, pulse oximetry sensors can be attached to fingers, toes, ears, or the bridge of the nose, but such stable placement is not feasible in utero. Further, good contact between sensor and fetal skin is a prerequisite for avoidance of artifacts. This last aspect has presented a sizeable challenge.
Fetal sensors measure reflected light. There is disagreement about the merits of the 2 sensor types, reflectance and transmission. Both include 2 light emitters (for red and infrared light) and a detector. In the transmission sensor (the adult or neonatal type), the light produced by the light-emitting diodes (LED) is picked up by the detector after traversing the interposed tissues. Since tissue interposition is not possible in the fetus, most fetal studies have used reflectance sensors, in which the LED and detector are placed side by side, and the light to be analyzed is reflected by the tissues. This design adds variance depending on the light’s depth of tissue penetration and device position changes.
Placement of the sensor. The Nellcor N-400 includes a reflectance sensor housed in a smooth, pliable head that is advanced through the cervix with the aid of a handle. The handle has a removable stylet to stiffen it during placement.
The sensor is placed against the fetal temple, cheek, or forehead and is held in place by the uterine wall. Placement is similar to that of an internal pressure catheter. Once the stylet is removed, it should not be reinserted.
Because the sensor usually descends and rotates with the fetal head, displacements are frequent and adjustments in sensor placement may be necessary. Placement adjustments can be attempted without the stylet and, if unsuccessful, a new device can be inserted. The Nellcor sensor is not reusable.
The prerequisites for insertion are dilatation of at least 2 cm, ruptured membranes, cephalic presentation, single fetus, gestational age of at least 36 weeks, and no placenta previa.
The manufacturer reports that active genital herpes, HIV, and hepatitis B or E seropositivity preclude fetal pulse oximetry monitoring.
Placement may be impossible when the presenting part is at high station (-3 or above) or low station (+2 or below).
The Nellcor N-400 system has been commercially available in many European countries since 1995, and in Canada since 1998. It was approved for sale in the United States in early 2003.
Question 6Will it improve neonatal outcomes?
Neonatal outcome is the ultimate endpoint in obstetrical care. In the randomized trial by Garite et al,3 there was no difference in neonatal outcome between the groups using or not using fetal pulse oximetry. According to Chua et al,33 FSpO2 levels measured even 10 minutes before delivery have no relation to neonatal outcome.
Leszczynska-Gorzelak et al22 believe FSpO2 is more predictive of neonatal outcome in the first stage than the second. However, Apgar score had no relationship with FSpO2readings in the first or second stage. Butterwegge34 reported 6 cases of FSpO2 below 30% for more than 30 minutes, all with good neonatal outcome, and Alshimmiri et al23 noted that only normal FSpO2 correlates with fetal well-being. Thus, it appears that the NPV of fetal oximetry is of greater practical value than other attributes.
Question 7How precise is it?
The Nellcor system monitors the quality of FSpO2 measurement; no value is displayed if the signal lacks the characteristics of a fetal arterial plethysmographic curve or if contact between sensor and skin is insufficient. Because of fetal movements and other artifacts, posting time is always less than 100%.
In the French multicenter study,25 the mean reliable signal time in the first stage of labor was only 64.7%—even less in the second stage (54%). Signal retention was 67% in the randomized trial by Garite et al. 3
Many artifacts may impede signal acquisition and impact the reliability of a reading:
- The sensor’s position on the fetal head. For example, the difference in FSpO2 readings between the forehead and occiput may be as much as 13.4%. (The sensor is designed to go against the fetal cheek, but may move around.)
- Incomplete sensor-to-skin contact, such as with high fetal head station, -2 or above.
- Marked caput formation.
- Increased intrauterine pressure accompanying contractions, especially at presentation stations of +2 or below (FIGURE 2). FSpO2 monitoring requires detection of fetal pulses, which may be undetectable when the surrounding pressure is high, resulting in a loss of signal.
- Interposition of vernix or fetal hair.
- Presence of meconium, which behaves like a red-light filter, altering the ratio of red to infrared light and resulting in artificially low values.35 This theoretical concern is rejected by Yam et al,36 who did not observe any effect of meconium on FSpO2 values. (When the amniotic fluid is meconium-stained, Carbonne et al37 showed that fetal oximetry is a better predictor of meconium aspiration syndrome than FSB sampling.) The data on the influence of meconium on FSpO2 readings remain contradictory.
All these conditions may impair precision and contribute to poor sensitivity.
FIGURE 2 A weakening signal during pushing
Question 8Is it easy to use?
An Australian survey38 assessed clinicians’ perceptions during placement of the oximetry sensor. Ease of placement was rated as good or excellent in 71% of cases, and the patient’s comfort was rated as good or excellent in 90% of cases. Chua et al39 reported a mean insertion time of 90 seconds, with a reliable signal obtained within 5 minutes in 87% of placements. The French multicenter study25 mentioned earlier concluded that the procedure is satisfactory and easier than FSB sampling. The device itself was harmless to both mother and fetus.40
Potential research directions
Fetal pulse oximetry may be an effective tool in clinical scenarios such as:
- fetal arrhythmias with uninterpretable FHR tracing
- fetal tachycardia associated with maternal fever, thyrotoxicosis, or fetal supraventricular tachycardia, when distinguishing other contributions to tachycardia may be difficult
- fetal bradycardia caused by a complete heart block, which may render EFM undecipherable
- when amnioinfusion is attempted for variable decelerations and it is necessary to differentiate a nonreassuring FHR tracing related to transient in utero stress (eg, umbilical cord compressions) from ominous tracings
Another area not yet addressed is cost-effectiveness beyond the immediate direct costs (approximately $11,000 for the monitor and $150 for each disposable sensor). Also uncertain is whether laboring women will accept the device (how disturbing or invasive it is perceived to be) and how acceptable or applicable it is outside tertiary institutions.
Dr. Vidaeff reports no financial relationships relevant to this article. Dr. Ramin reports grant support from the US National Institutes of Health.
1. Dildy GA. A guest editorial: fetal pulse oximetry. Obstet Gynecol Surv. 2003;58:225-226.
2. Thacker SB, Stroup DF, Peterson HB. Efficacy and safety of intrapartum electronic fetal monitoring: an update. Obstet Gynecol. 1995;86:613-620.
3. Garite TJ, Dildy GA, McNamara H, et al. A multicenter controlled trial of fetal pulse oximetry in the intrapartum management of nonreassuring fetal heart rate patterns. Am J Obstet Gynecol. 2000;183:1049-1058.
4. Haverkamp AD, Orleans M, Langendoerfer S, et al. A controlled trial of the differential effects of continuous fetal heart rate monitoring in labor: a randomized trial. Am J Obstet Gynecol. 1979;134:399-412.
5. American College of Obstetricians and Gynecologists. Committee opinion: fetal pulse oximetry. Number 258, September 2001. Obstet Gynecol. 2001;98:523-524.
6. Zijlstra WG, Buursma A, Zwart A. Performance of an automated six-wavelength photometer (radiometer OSM3) for routine measurement of hemoglobin derivatives. Clin Chem. 1988;34:149-152.
7. Porath M, Sinah P, Dudenhausen JW, Luttkus AK. Systematic instrumental errors between oxygen saturation analysers in fetal blood during deep hypoxemia. Clin Chim Acta. 2001;307:151-157.
8. Luttkus AK, Lübke M, Büscher U, Porath M, Dudenhausen JW. Accuracy of fetal pulse oximetry. Acta Obstet Gynecol Scand. 2002;81:417-423.
9. Carter AM, Stiller R, König V, Jorgensen JS, Svendsen P, Huch R. Calibration of a reflectance pulse oximeter in fetal lambs for arterial oxygen saturations below 70%. J Soc Gynecol Invest. 1998;5:255-259.
10. McNamara H, Cung CD, Lilford R, Johnson N. Do fetal pulse oximetry readings at delivery correlate with cord blood oxygenation and acidemia? Br J Obstet Gynaecol. 1992;99:735-738.
11. Langer B, Boudier E, Haddad J, Pain L, Schlaeder G. Fetal pulse oximetry during labor of 62 patients. Fetal Diagn Ther. 1996;11:37-45.
12. Harrison AM, Lynch JM, Dean JM, Witte MK. Comparison of simultaneously obtained arterial and capillary blood gases in pediatric intensive care unit patients. Crit Care Med. 1997;25:1904-1908.
13. Nijland R, Jongsma HW, Nijhuis JG, Oeseburg B. Accuracy of fetal pulse oximetry and pitfalls in measurements. Eur J Obstet Gynecol Reprod Biol. 1997;72:S21-S27.
14. Dildy GA, van den Berg PP, Katz M, et al. Intrapartum fetal pulse oximetry: fetal oxygen saturation trends during labor and relation to delivery outcome. Am J Obstet Gynecol. 1994;171:679-684.
15. Nijland R, Jongsma H, Nijhuis J, van den Berg P, Oeseburg B. Arterial saturation in relation to metabolic acidosis in fetal lambs. Am J Obstet Gynecol. 1995;172:810-819.
16. Kühnert M, Seelbach-Göbel B, Butterwegge M. Predictive agreement between fetal arterial oxygen saturation and fetal scalp pH: results of the German multicenter study. Am J Obstet Gynecol. 1998;178:330-335.
17. Salamalekis E, Thomopoulos P, Giannaris D, et al. Computerised intrapartum diagnosis of fetal hypoxia based on heart rate monitoring and fetal pulse oximetry recordings utilizing wavelet analysis and neural networks. Br J Obstet Gynaecol. 2002;109:1137-1142.
18. Bloom SL, Swindle RG, McIntire DD, Leveno KJ. Fetal pulse oximetry: duration of desaturation and intrapartum outcome. Obstet Gynecol. 1999;93:1036-1040.
19. Seelbach-Göbel B, Heupel M, Kühnert M, et al. The prediction of fetal acidosis by means of intrapartum fetal pulse oximetry. Am J Obstet Gynecol. 1999;180:73-81.
20. Gorenberg DM, Patillo C, Hendi P, Rumney PJ, Garite TJ. Fetal pulse oximetry: correlation between oxygen desaturation, duration, and frequency and neonatal outcomes. Am J Obstet Gynecol. 2003;189:136-138.
21. Dildy GA, Thorp JA, Yeast JD, Clark SL. The relationship between oxygen saturation and pH in umbilical blood: implications for intrapartum fetal oxygen saturation monitoring. Am J Obstet Gynecol. 1996;175:682-687.
22. Leszczynska-Gorzelak B, Poniedzialek-Czajkowska E, Oleszczuk J. Fetal blood saturation during the 1st and 2nd stage of labor and its relation to the neonatal outcome. Gynecol Obstet Invest. 2002;54:159-163.
23. Alshimmiri M, Bocking AD, Gagnon R, Natale R, Richardson BS. Prediction of umbilical artery base excess by intrapartum fetal oxygen saturation monitoring. Am J Obstet Gynecol. 1997;177:775-779.
24. Rijnders RJ, Mol BW, Reuwer PJ, Drogtrop AP, Vernooij MM, Visser GH. Is the correlation between fetal oxygen saturation and blood pH sufficient for the use of fetal pulse oximetry? J Matern Fetal Neonatal Med. 2002;11:80-83.
25. Goffinet F, Langer B, Carbonne B, et al. Multicenter study on the clinical value of fetal pulse oximetry. I. Methodologic evaluation. The French Study Group on Fetal Pulse Oximetry. Am J Obstet Gynecol. 1997;177:1238-1246.
26. Schmidt S, Koslowski S, Sierra F, et al. Clinical usefulness of pulse oximetry in the fetus with non-reassuring heart rate pattern? J Perinat Med. 2000;28:298-305.
27. Vitoratos N, Salamalekis E, Saloum J, Makrakis E, Creatsas G. Abnormal fetal heart rate patterns during the active phase of labor and the value of fetal oxygen saturation. J Matern Fetal Neonatal Med. 2002;11:46-49.
28. Stiller R, von Mering R, König V, et al. How well does reflectance pulse oximetry reflect intrapartum fetal acidosis? Am J Obstet Gynecol. 2002;186:1351-1357.
29. Leszczynska-Gorzelak B, Poniedzialek-Czajkowska E, Zych I, Grzechnik M, Oleszczuk J. The usefulness of the intrapartum fetal pulse oximetry in anticipating the neonatal outcome. Ginekol Pol. 2001;72:1183-1188.
30. Nikolov A, Dimitrov A, Vakrilkova L, Iarkova N. Fetal oxygen saturation during normal delivery. Akush Ginekol. 2000;40:3-6.
31. Goldaber KG, Gilstrap LC III, Leveno K, Dax JS, McIntire DD. Pathologic fetal acidemia. Obstet Gynecol. 1991;78:1103-1107.
32. Low JA, Victory R, Derrick EJ. Predictive value of electronic fetal monitoring for intra-partum fetal asphyxia and metabolic acidosis. Obstet Gynecol. 1999;93:285-291.
33. Chua S, Yeong SM, Razvi K, Arulkumaran S. Fetal oxygen saturation during labour. Br J Obstet Gynaecol. 1997;104:1080-1083.
34. Butterwegge M. Fetal pulse oximetry and non-reassuring heart rate. Eur J Obstet Gynecol Reprod Biol. 1997;72:S63-S66.
35. Johnson N, Johnson VA, Bannister J, McNamara H. The effect of meconium on neonatal and fetal reflectance pulse oximetry. J Perinat Med. 1990;18:351-355.
36. Yam J, Chua S, Arulkumaran S. Intrapartum pulse oximetry: Part 1: Principles and technical issues. Obstet Gynecol Surv. 2000;55:163-172.
37. Carbonne B, Cudeville C, Sivan H, Cabrol D, Papiernik E. Fetal oxygen saturation measured by pulse oximetry during labor with clear and meconium-stained amniotic fluid. Eur J Obstet Gynecol Reprod Biol. 1997;72:S51-S55.
38. East CE, Colditz PB. Clinicians’ perceptions of placing a fetal oximetry sensor. J Qual Clin Pract. 2000;20:161-163.
39. Chua S, Yam J, Razvi K, Yeong SM, Arulkumaran S. Intrapartum fetal oxygen saturation monitoring in a busy labour ward. Eur J Obstet Gynecol Reprod Biol. 1999;82:185-189.
40. Luttkus AK, Friedmann W, Thomas S, Dimer JA, Dudenhausen JW. The safety of fetal pulse oximetry in parturients requiring fetal scalp blood sample. Obstet Gynecol. 1997;90:533-537.
- The value of this new technology might not be so much the prediction of acidosis but identification of the well-oxygenated fetus so that labor may be safely continued in the presence of a concerning—but not ominous—fetal heart rate tracing.
- The only randomized study published so far did not determine whether clinical decisions can be based solely on fetal pulse oximetry. The investigators did suggest that sensitivity and specificity for metabolic acidemia was improved in the intervention group—a promising appraisal, in contrast with previous observational data.
When a teenage nullipara underwent labor induction for preeclampsia at 37 weeks, she was given epidural analgesia and seizure prophylaxis with magnesium sulfate. Her electronic fetal heart rate (FHR) tracing was initially reassuring, with only occasional variable decelerations, but subsequently revealed a baseline of 140 beats per minute (bpm), minimal to absent variability, no accelerations, and variable decelerations to 90 bpm with rapid return to baseline.
The tracing was interpreted as nonreassuring, and a fetal pulse oximeter was inserted. It revealed a fetal oxygen saturation rate between 45% and 50%, and labor was allowed to continue. After 3.5 hours in the second stage, the patient was delivered by outlet forceps. Her infant had Apgar scores of 8 at 1 minute and 9 at 5 minutes. The umbilical arterial pH was 7.25, and base excess was–4.9.
Fetal pulse oximetry made it possible to manage this case without resorting to emergent cesarean. But is this noninvasive technology truly a step forward in intrapartum assessment of fetal well-being?
We describe what the evidence (a single randomized study and a number of observational studies) reveals about these questions:
- How accurately does fetal pulse oximetry reflect the fetal condition?
- What is the critical threshold for fetal oxygen desaturation?
- Is a single reading reliable?
- Does oximetry correlate with acid-base status?
- Does the combination of oximetry and electronic monitoring improve accuracy?
- Will fetal pulse oximetry improve neonatal outcomes?
- How precise is it?
- Is it easy to use?
Needed: Effective adjunct to electronic monitoring
Except in the chronically hypoxic fetus (which is affected by the time labor begins), the pathophysiology of acute intrapartum events is a continuum, from hypoxemia to respiratory acidosis to metabolic acidosis and, ultimately, clinical impairment. The goal of intrapartum surveillance is to detect fetal hypoxemia before it progresses to asphyxia and perinatal mortality or long-term morbidity.
Although it is approved as an adjunct to electronic fetal monitoring (EFM), fetal pulse oximetry has gained only sporadic use since it became available in the United States in 2000—even though EFM has proved disappointing as a tool for predicting fetal hypoxia. Only about 10% of US obstetrical units had fetal pulse oximetry technology as of 2002.1
Clinicians began questioning the reliability of subjective interpretation of fetal heart tracings soon after EFM went into general use. Thirty years later, a meta-analysis of 12 randomized clinical trials involving 58,855 gravidas cast doubt on the benefits of EFM,2 which is associated with an increase in operative deliveries as a result of high sensitivity but low specificity in predicting fetal hypoxia and acidosis.
FDA approval was based on sole randomized trial
The only commercially available fetal oximetry sensor, the Nellcor N-400 (Nellcor, Pleasanton, Calif), obtained US Food and Drug Administration (FDA) approval as an adjunct to EFM when the latter indicates a nonreassuring FHR pattern. That approval was based on the only randomized study3 of fetal pulse oximetry conducted, which involved 1,010 women with predefined nonreassuring FHR patterns in labor.
Goal: Reduced cesarean rate with comparable outcomes. Investigators hypothesized that adjunctive fetal oximetry would improve assessment and reduce the cesarean rate without altering neonatal outcome. Indeed, in the oximetry group, the rate of cesarean delivery performed for a nonreassuring FHR tracing (4.5% versus 10.2%; P = .007) was significantly reduced. Other findings:
- Same neonatal outcomes, with no significant differences between the 2 groups.
- Higher cesarean rate for dystocia in the intervention group, offsetting any advantage in the overall cesarean delivery rate (29% versus 26%). This unexpected increase in cesarean deliveries raises several possibilities:
- Given the unblinded design, it is possible that clinicians, circumspect of the pulse oximetry, continued to perform cesareans for nonreassuring FHR, but labeled the indication for surgery differently. The validity of the dystocia diagnosis was discredited by a subsequent partogram analysis that showed a similar rate of arrested labor in both groups.
- A nonreassuring FHR in conditions of normal fetal oxygenation is predictive of dystocia. Previous randomized studies of EFM have suggested the same thing.4
- Dystocia is the consequence of the device itself. Anecdotal observations suggest a higher rate of persistent occiput posterior positions with fetal oximetry.
Other trials underway. The ongoing Fetal Oximetry (FOX) trial of the National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network, involving 10,000 nulliparous participants, is comparing cesarean delivery rates and safety outcomes in patients monitored for FHR plus pulse oximetry with a group in which the clinicians are blinded to the pulse oximetry readings. Another randomized controlled trial of fetal pulse oximetry is underway in Australia.
Potential for increased costs. The American College of Obstetricians and Gynecologists (ACOG) has raised concerns about the potential increase in costs without demonstrable improvement in outcome.5 ACOG has not endorsed fetal pulse oximetry for general practice.
Question 1How accurately does pulse oximetry reflect the fetal condition?
It yields only indirect information on the partial pressure of oxygen in the blood and no data on perfusion or acid-base status.
In other clinical settings, oxygen saturation is not an acceptable substitute for arterial blood gas analysis. The pulse oximeter is not a hemoximeter—only that device directly and reliably determines blood oxygen saturation by spectrophotometry.6 Even the calculated oxygen saturation values provided automatically by modern blood gas analyzers are inaccurate.7
Studies report varying results. In a comparison8 of fetal oxygen saturation by hemoximetry in a fetal scalp blood (FSB) sample and fetal arterial oxyhemoglobin saturation (FSpO2) by pulse oximetry immediately before the blood sampling, the FSpO2 medians were always higher than the FSB hemoximetry saturation—which led to false-negative results in hypoxic babies.
In animal studies, pulse oximetry correlated well with simultaneously measured arterial oxygen saturation (r = 0.98, P = .01),9 but data from human studies are inconsistent. While McNamara et al10 reported good correlation between FSpO2 measurements and umbilical artery blood oxygen saturation at birth (r = 0.59, P <.001), Langer et al11 found no relationship between FSpO2 levels determined during pushing efforts and oxygen saturation in umbilical vein blood at birth.
Possible reasons for the ambiguous findings:
- differences in practice, such as use of umbilical venous versus arterial blood, or measurement during pushing versus between pushes,
- different intervals from FSpO2 reading to umbilical blood sampling, or
- incomparable groups, such as all women in labor versus those with abnormal FHR.
Limitations. Fetal pulse oximetry measures arterial oxygen saturation during the systolic pulse wave in the skin microcirculation at head level. In the fetus, this is part of the preductal circulation, with oxygen saturation levels somewhere between umbilical arterial and umbilical venous blood oxygen saturation.
Theoretically, FSpO2 should be closer to FSB than to umbilical blood. Although FSB samples consist of capillary blood, which is not exactly central arterial blood, the differences are small, at least in the neonate.12 In the intrapartum period, however, several variables with unknown effect may weaken relationships:
- different intervals between the last oximetry signal and blood sampling after delivery
- differences in local tissue perfusion status13
- perfusion changes during fetal compromise, as the fetus centralizes its blood flow, with vasoconstriction in the skin circulation
Question 2What is the critical threshold for fetal oxygen desaturation?
Human studies indicate that an FSpO2 of 33% is approximately the 10th percentile on the normal distribution, and an FSpO2 of 29% to 30% represents the third to fifth percentiles in normal-outcome labor.14 Studies in catheterized fetal sheep suggest that the level below which metabolic acidosis can be anticipated is an FSpO2 of about 30%.15
The 30% threshold also is supported by prospective human data from a multicenter trial.16 According to those data, an FSpO2 of less than 30% has 100% sensitivity in predicting an FSB pH below 7.20. FSpO2 of less than 30% also correlated with a lack of variability on the FHR tracing.17
The cutoff of 30% should not be interpreted as an indication of fetal distress, however. Rather, it represents a threshold below which increasing fetal acidosis will be encountered ( FIGURE 1). Oxygen saturation is a dynamic biologic parameter with broad variation.
FIGURE 1 Tracking fetal arterial oxyhemoglobin saturation
Question 3Is a single reading reliable?
The normal fetus has a remarkable capacity to compensate for transient episodes of desaturation. Thus, a single reading cannot reflect the fetal condition; the trend in FSpO2 must be taken into account. Research indicates only FSpO2 levels below 30% for more than 2 minutes18 or more than 10 minutes19 are likely to be associated with intrapartum acidosis.
ACOG has raised concerns about the potential increase in costs without demonstrable improvement in outcome.
Gorenberg et al20 retrospectively correlated FSpO2 with umbilical artery pH and found that neither the 30% threshold alone nor the duration of FSpO2 below 30% correlated with fetal acidemia (pH below 7.20). Rather, the repetition of such episodes was more predictive. The authors concluded that more than 10 episodes of FSpO2below 30% would overcome the ability of the fetus to compensate.
The study was underpowered to detect a significant difference in acidemia, and did not allow sufficient observation time to detect the natural progression of hypoxia to metabolic acidosis, a better indicator of fetal compromise. Additional research is needed.
Question 4Does oximetry correlate with acid-base status?
Many of the studies mentioned here assumed a correlation. Whenever oxygen saturation in the umbilical artery is 30% or more, acidosis (pH below 7.13) in the same blood is rare—only 1%.21 However, the correlation between fetal pulse oximetry values and acid-base status is much weaker.8.
Leszczynska-Gorzelak et al22 found no relationship between FSpO2 levels in the first or second stage of labor and pH or partial pressure of oxygen in umbilical vein blood at delivery. Other investigators concluded similarly, considering intrapartum FSpO2 of limited use for predicting acidosis at birth, irrespective of FSpO2 cutoff.23,24
Rijnders et al24 found no significant correlation between fetal scalp or umbilical artery blood pH and mean FSpO2 for the last 30 minutes before sampling (r = 0.02, P = .9). Even the lowest FSpO2 level did not correlate with arterial pH (r = .04, P = .84). None of the study’s 3 cases of umbilical pH below 7.05 would have been detected using the mean FSpO2 before delivery, and only 1 would have been detected using the lowest FSpO2.
In another multicenter study involving the Nellcor system in 164 cases with abnormal FHR, a correlation between oximetry and FSB sampling (r = 0.29, P < .01) was noted in the first stage of labor, but second-stage FSpO2 readings did not correlate with oxygen saturation, partial pressure of oxygen, pH, or bicarbonate level in the umbilical artery at birth.25
An observational series26 of 128 fetuses with nonreassuring FHR patterns concluded that fetal distress was insufficiently identified by oximetry. Only 2 of the 11 cases with umbilical artery pH below 7.20 were detected by pulse oximetry recordings below 30% during the last 30 minutes of the second stage, and out of 5 cases with hypoxic readings in the second stage, only 2 were acidotic at birth. The calculated sensitivity was 18%, specificity 92%, positive predictive value (PPV) 40%, and negative predictive value (NPV) 80%. A low Apgar score was never predicted by fetal pulse oximetry.
Others used the same Nellcor system over the final 30 minutes of labor and a cutoff for umbilical blood acidemia of pH below 7.13 and reported similar numbers: sensitivity 28%, specificity 94%, PPV 40%, and NPV 80%.23
Vitoratos et al27 analyzed FSpO2 readings in active labor (not limited to the last 30 minutes before delivery) and obtained somewhat better values: sensitivity 72%, specificity 93%, PPV 61.5%, and NPV 95.8% for an umbilical artery blood pH below 7.15.
The impression that the validity of fetal pulse oximetry is higher in earlier labor than in the second stage is supported by data from Stiller et al. 28 Leszczynska-Gorzelak et al29 found a significant decrease in mean FSpO2 from the first stage to the second stage of normal labor (51.9% versus 43.8%, P < .001), and Dildy et al14 noted a similar difference upon analyzing 160 normal labors (59% versus 53%), but other studies failed to verify such differences.25,30
Observational studies had unrealistic pH cutoff. All the evidence presented thus far on the validity of fetal pulse oximetry in predicting acidemia is based on observational data. A common deficiency is the unrealistic cutoff for pathologic fetal acidemia—a pH of less than 7.13 to 7.20—when it is widely accepted that “pathologic fetal acidemia” reflects an umbilical artery blood pH below 7.31 Even in this group, two thirds of neonates are unaffected by morbidity.
Need to identify metabolic acidosis. It also is accepted that the presence of a metabolic component to fetal acidemia may be as important—if not more important—than a single pH cutoff.31 Only a few human studies of pulse oximetry have distinguished between respiratory and metabolic acidemia. When they did, intrapartum fetal pulse oximetry was unable to predict umbilical artery base excess.23,25
The only randomized study failed to determine whether clinical decisions can be based solely on fetal pulse oximetry. 3 The investigators did suggest that sensitivity and specificity for metabolic acidemia was improved in the intervention group—a promising appraisal, in contrast with previous observational data.
In the study, 7 neonates (3 in the intervention group and 4 controls) had umbilical artery blood pH below 7. All 4 controls had vaginal delivery. There also were 6 cases of elevated base excess (ie, -16 mEq/L or below) among controls. None were recorded in the intervention group, and the 3 cases of acidemia were recognized antepartum and led to cesareans.
Unfortunately, the study design did not guarantee that patient management was based exclusively on EFM with or without fetal pulse oximetry. Vibro-acoustic stimulation or FSB sampling was required before proceeding to cesarean delivery in both groups.
It appears that the negative predictive value of fetal oximetry is of greater practical value than other attributes.
When FSpO2 was less than 30% for the entire interval between 2 contractions, or was unobtainable, the physician was supposed to revert to interpretation of EFM. When that was persistently nonreassuring, the physician was given the option of scalp stimulation or FSB sampling. Thus, it was not determined whether clinical decisions can be based exclusively on fetal pulse oximetry. Schmidt et al26 suggested that such exclusive application of fetal pulse oximetry might actually jeopardize fetal health.
Question 5 Does the combination of oximetry and EFM improve accuracy?
Fetal pulse oximetry was not used independently in any of the studies discussed here, but in association with EFM, which has a sensitivity for fetal acidosis of 93%, specificity of 29%, PPV of 2.6%, and NPV of 99.5%.32
From a statistical point of view, whenever 2 evaluation methods with the same endpoint (fetal acidosis) are combined, sensitivity decreases while specificity increases, theoretically resulting in less unnecessary intervention. That is exactly what investigators have reported: sensitivity as low as 18%26 for fetal oximetry, and specificity as high as 94%.23 However, the value of this new technology might not be so much the prediction of acidosis but identification of the well-oxygenated fetus so that labor may be safely continued in the presence of a concerning—but not ominous—FHR tracing.
Fetal pulse oximetry employs principles of optical spectrophotometry and plethysmography to provide information on the percentage of oxygen bound to hemoglobin. Oxyhemoglobin (oxygenated hemoglobin) and deoxyhemoglobin (hemoglobin without oxygen) absorb red and infrared light differently: more red absorption by deoxyhemoglobin, and more infrared absorption by oxyhemoglobin.
By measuring the relative absorption at each wavelength, the fraction of hemoglobin that carries oxygen can be determined. The arterial oxygen saturation is expressed as a percentage. The technology has been refined to measure fetal arterial oxyhemoglobin saturation during labor.
Pulse oximetry sensors must be calibrated for fetal biological values. In the fetus, normal oxygen saturation is much lower than in the adult or neonate; hemoglobin has a higher affinity for oxygen and is in higher concentration; and there are more capillaries per unit of tissue, higher cardiac output, and a higher heart rate.
In the adult or neonate, pulse oximetry sensors can be attached to fingers, toes, ears, or the bridge of the nose, but such stable placement is not feasible in utero. Further, good contact between sensor and fetal skin is a prerequisite for avoidance of artifacts. This last aspect has presented a sizeable challenge.
Fetal sensors measure reflected light. There is disagreement about the merits of the 2 sensor types, reflectance and transmission. Both include 2 light emitters (for red and infrared light) and a detector. In the transmission sensor (the adult or neonatal type), the light produced by the light-emitting diodes (LED) is picked up by the detector after traversing the interposed tissues. Since tissue interposition is not possible in the fetus, most fetal studies have used reflectance sensors, in which the LED and detector are placed side by side, and the light to be analyzed is reflected by the tissues. This design adds variance depending on the light’s depth of tissue penetration and device position changes.
Placement of the sensor. The Nellcor N-400 includes a reflectance sensor housed in a smooth, pliable head that is advanced through the cervix with the aid of a handle. The handle has a removable stylet to stiffen it during placement.
The sensor is placed against the fetal temple, cheek, or forehead and is held in place by the uterine wall. Placement is similar to that of an internal pressure catheter. Once the stylet is removed, it should not be reinserted.
Because the sensor usually descends and rotates with the fetal head, displacements are frequent and adjustments in sensor placement may be necessary. Placement adjustments can be attempted without the stylet and, if unsuccessful, a new device can be inserted. The Nellcor sensor is not reusable.
The prerequisites for insertion are dilatation of at least 2 cm, ruptured membranes, cephalic presentation, single fetus, gestational age of at least 36 weeks, and no placenta previa.
The manufacturer reports that active genital herpes, HIV, and hepatitis B or E seropositivity preclude fetal pulse oximetry monitoring.
Placement may be impossible when the presenting part is at high station (-3 or above) or low station (+2 or below).
The Nellcor N-400 system has been commercially available in many European countries since 1995, and in Canada since 1998. It was approved for sale in the United States in early 2003.
Question 6Will it improve neonatal outcomes?
Neonatal outcome is the ultimate endpoint in obstetrical care. In the randomized trial by Garite et al,3 there was no difference in neonatal outcome between the groups using or not using fetal pulse oximetry. According to Chua et al,33 FSpO2 levels measured even 10 minutes before delivery have no relation to neonatal outcome.
Leszczynska-Gorzelak et al22 believe FSpO2 is more predictive of neonatal outcome in the first stage than the second. However, Apgar score had no relationship with FSpO2readings in the first or second stage. Butterwegge34 reported 6 cases of FSpO2 below 30% for more than 30 minutes, all with good neonatal outcome, and Alshimmiri et al23 noted that only normal FSpO2 correlates with fetal well-being. Thus, it appears that the NPV of fetal oximetry is of greater practical value than other attributes.
Question 7How precise is it?
The Nellcor system monitors the quality of FSpO2 measurement; no value is displayed if the signal lacks the characteristics of a fetal arterial plethysmographic curve or if contact between sensor and skin is insufficient. Because of fetal movements and other artifacts, posting time is always less than 100%.
In the French multicenter study,25 the mean reliable signal time in the first stage of labor was only 64.7%—even less in the second stage (54%). Signal retention was 67% in the randomized trial by Garite et al. 3
Many artifacts may impede signal acquisition and impact the reliability of a reading:
- The sensor’s position on the fetal head. For example, the difference in FSpO2 readings between the forehead and occiput may be as much as 13.4%. (The sensor is designed to go against the fetal cheek, but may move around.)
- Incomplete sensor-to-skin contact, such as with high fetal head station, -2 or above.
- Marked caput formation.
- Increased intrauterine pressure accompanying contractions, especially at presentation stations of +2 or below (FIGURE 2). FSpO2 monitoring requires detection of fetal pulses, which may be undetectable when the surrounding pressure is high, resulting in a loss of signal.
- Interposition of vernix or fetal hair.
- Presence of meconium, which behaves like a red-light filter, altering the ratio of red to infrared light and resulting in artificially low values.35 This theoretical concern is rejected by Yam et al,36 who did not observe any effect of meconium on FSpO2 values. (When the amniotic fluid is meconium-stained, Carbonne et al37 showed that fetal oximetry is a better predictor of meconium aspiration syndrome than FSB sampling.) The data on the influence of meconium on FSpO2 readings remain contradictory.
All these conditions may impair precision and contribute to poor sensitivity.
FIGURE 2 A weakening signal during pushing
Question 8Is it easy to use?
An Australian survey38 assessed clinicians’ perceptions during placement of the oximetry sensor. Ease of placement was rated as good or excellent in 71% of cases, and the patient’s comfort was rated as good or excellent in 90% of cases. Chua et al39 reported a mean insertion time of 90 seconds, with a reliable signal obtained within 5 minutes in 87% of placements. The French multicenter study25 mentioned earlier concluded that the procedure is satisfactory and easier than FSB sampling. The device itself was harmless to both mother and fetus.40
Potential research directions
Fetal pulse oximetry may be an effective tool in clinical scenarios such as:
- fetal arrhythmias with uninterpretable FHR tracing
- fetal tachycardia associated with maternal fever, thyrotoxicosis, or fetal supraventricular tachycardia, when distinguishing other contributions to tachycardia may be difficult
- fetal bradycardia caused by a complete heart block, which may render EFM undecipherable
- when amnioinfusion is attempted for variable decelerations and it is necessary to differentiate a nonreassuring FHR tracing related to transient in utero stress (eg, umbilical cord compressions) from ominous tracings
Another area not yet addressed is cost-effectiveness beyond the immediate direct costs (approximately $11,000 for the monitor and $150 for each disposable sensor). Also uncertain is whether laboring women will accept the device (how disturbing or invasive it is perceived to be) and how acceptable or applicable it is outside tertiary institutions.
Dr. Vidaeff reports no financial relationships relevant to this article. Dr. Ramin reports grant support from the US National Institutes of Health.
- The value of this new technology might not be so much the prediction of acidosis but identification of the well-oxygenated fetus so that labor may be safely continued in the presence of a concerning—but not ominous—fetal heart rate tracing.
- The only randomized study published so far did not determine whether clinical decisions can be based solely on fetal pulse oximetry. The investigators did suggest that sensitivity and specificity for metabolic acidemia was improved in the intervention group—a promising appraisal, in contrast with previous observational data.
When a teenage nullipara underwent labor induction for preeclampsia at 37 weeks, she was given epidural analgesia and seizure prophylaxis with magnesium sulfate. Her electronic fetal heart rate (FHR) tracing was initially reassuring, with only occasional variable decelerations, but subsequently revealed a baseline of 140 beats per minute (bpm), minimal to absent variability, no accelerations, and variable decelerations to 90 bpm with rapid return to baseline.
The tracing was interpreted as nonreassuring, and a fetal pulse oximeter was inserted. It revealed a fetal oxygen saturation rate between 45% and 50%, and labor was allowed to continue. After 3.5 hours in the second stage, the patient was delivered by outlet forceps. Her infant had Apgar scores of 8 at 1 minute and 9 at 5 minutes. The umbilical arterial pH was 7.25, and base excess was–4.9.
Fetal pulse oximetry made it possible to manage this case without resorting to emergent cesarean. But is this noninvasive technology truly a step forward in intrapartum assessment of fetal well-being?
We describe what the evidence (a single randomized study and a number of observational studies) reveals about these questions:
- How accurately does fetal pulse oximetry reflect the fetal condition?
- What is the critical threshold for fetal oxygen desaturation?
- Is a single reading reliable?
- Does oximetry correlate with acid-base status?
- Does the combination of oximetry and electronic monitoring improve accuracy?
- Will fetal pulse oximetry improve neonatal outcomes?
- How precise is it?
- Is it easy to use?
Needed: Effective adjunct to electronic monitoring
Except in the chronically hypoxic fetus (which is affected by the time labor begins), the pathophysiology of acute intrapartum events is a continuum, from hypoxemia to respiratory acidosis to metabolic acidosis and, ultimately, clinical impairment. The goal of intrapartum surveillance is to detect fetal hypoxemia before it progresses to asphyxia and perinatal mortality or long-term morbidity.
Although it is approved as an adjunct to electronic fetal monitoring (EFM), fetal pulse oximetry has gained only sporadic use since it became available in the United States in 2000—even though EFM has proved disappointing as a tool for predicting fetal hypoxia. Only about 10% of US obstetrical units had fetal pulse oximetry technology as of 2002.1
Clinicians began questioning the reliability of subjective interpretation of fetal heart tracings soon after EFM went into general use. Thirty years later, a meta-analysis of 12 randomized clinical trials involving 58,855 gravidas cast doubt on the benefits of EFM,2 which is associated with an increase in operative deliveries as a result of high sensitivity but low specificity in predicting fetal hypoxia and acidosis.
FDA approval was based on sole randomized trial
The only commercially available fetal oximetry sensor, the Nellcor N-400 (Nellcor, Pleasanton, Calif), obtained US Food and Drug Administration (FDA) approval as an adjunct to EFM when the latter indicates a nonreassuring FHR pattern. That approval was based on the only randomized study3 of fetal pulse oximetry conducted, which involved 1,010 women with predefined nonreassuring FHR patterns in labor.
Goal: Reduced cesarean rate with comparable outcomes. Investigators hypothesized that adjunctive fetal oximetry would improve assessment and reduce the cesarean rate without altering neonatal outcome. Indeed, in the oximetry group, the rate of cesarean delivery performed for a nonreassuring FHR tracing (4.5% versus 10.2%; P = .007) was significantly reduced. Other findings:
- Same neonatal outcomes, with no significant differences between the 2 groups.
- Higher cesarean rate for dystocia in the intervention group, offsetting any advantage in the overall cesarean delivery rate (29% versus 26%). This unexpected increase in cesarean deliveries raises several possibilities:
- Given the unblinded design, it is possible that clinicians, circumspect of the pulse oximetry, continued to perform cesareans for nonreassuring FHR, but labeled the indication for surgery differently. The validity of the dystocia diagnosis was discredited by a subsequent partogram analysis that showed a similar rate of arrested labor in both groups.
- A nonreassuring FHR in conditions of normal fetal oxygenation is predictive of dystocia. Previous randomized studies of EFM have suggested the same thing.4
- Dystocia is the consequence of the device itself. Anecdotal observations suggest a higher rate of persistent occiput posterior positions with fetal oximetry.
Other trials underway. The ongoing Fetal Oximetry (FOX) trial of the National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network, involving 10,000 nulliparous participants, is comparing cesarean delivery rates and safety outcomes in patients monitored for FHR plus pulse oximetry with a group in which the clinicians are blinded to the pulse oximetry readings. Another randomized controlled trial of fetal pulse oximetry is underway in Australia.
Potential for increased costs. The American College of Obstetricians and Gynecologists (ACOG) has raised concerns about the potential increase in costs without demonstrable improvement in outcome.5 ACOG has not endorsed fetal pulse oximetry for general practice.
Question 1How accurately does pulse oximetry reflect the fetal condition?
It yields only indirect information on the partial pressure of oxygen in the blood and no data on perfusion or acid-base status.
In other clinical settings, oxygen saturation is not an acceptable substitute for arterial blood gas analysis. The pulse oximeter is not a hemoximeter—only that device directly and reliably determines blood oxygen saturation by spectrophotometry.6 Even the calculated oxygen saturation values provided automatically by modern blood gas analyzers are inaccurate.7
Studies report varying results. In a comparison8 of fetal oxygen saturation by hemoximetry in a fetal scalp blood (FSB) sample and fetal arterial oxyhemoglobin saturation (FSpO2) by pulse oximetry immediately before the blood sampling, the FSpO2 medians were always higher than the FSB hemoximetry saturation—which led to false-negative results in hypoxic babies.
In animal studies, pulse oximetry correlated well with simultaneously measured arterial oxygen saturation (r = 0.98, P = .01),9 but data from human studies are inconsistent. While McNamara et al10 reported good correlation between FSpO2 measurements and umbilical artery blood oxygen saturation at birth (r = 0.59, P <.001), Langer et al11 found no relationship between FSpO2 levels determined during pushing efforts and oxygen saturation in umbilical vein blood at birth.
Possible reasons for the ambiguous findings:
- differences in practice, such as use of umbilical venous versus arterial blood, or measurement during pushing versus between pushes,
- different intervals from FSpO2 reading to umbilical blood sampling, or
- incomparable groups, such as all women in labor versus those with abnormal FHR.
Limitations. Fetal pulse oximetry measures arterial oxygen saturation during the systolic pulse wave in the skin microcirculation at head level. In the fetus, this is part of the preductal circulation, with oxygen saturation levels somewhere between umbilical arterial and umbilical venous blood oxygen saturation.
Theoretically, FSpO2 should be closer to FSB than to umbilical blood. Although FSB samples consist of capillary blood, which is not exactly central arterial blood, the differences are small, at least in the neonate.12 In the intrapartum period, however, several variables with unknown effect may weaken relationships:
- different intervals between the last oximetry signal and blood sampling after delivery
- differences in local tissue perfusion status13
- perfusion changes during fetal compromise, as the fetus centralizes its blood flow, with vasoconstriction in the skin circulation
Question 2What is the critical threshold for fetal oxygen desaturation?
Human studies indicate that an FSpO2 of 33% is approximately the 10th percentile on the normal distribution, and an FSpO2 of 29% to 30% represents the third to fifth percentiles in normal-outcome labor.14 Studies in catheterized fetal sheep suggest that the level below which metabolic acidosis can be anticipated is an FSpO2 of about 30%.15
The 30% threshold also is supported by prospective human data from a multicenter trial.16 According to those data, an FSpO2 of less than 30% has 100% sensitivity in predicting an FSB pH below 7.20. FSpO2 of less than 30% also correlated with a lack of variability on the FHR tracing.17
The cutoff of 30% should not be interpreted as an indication of fetal distress, however. Rather, it represents a threshold below which increasing fetal acidosis will be encountered ( FIGURE 1). Oxygen saturation is a dynamic biologic parameter with broad variation.
FIGURE 1 Tracking fetal arterial oxyhemoglobin saturation
Question 3Is a single reading reliable?
The normal fetus has a remarkable capacity to compensate for transient episodes of desaturation. Thus, a single reading cannot reflect the fetal condition; the trend in FSpO2 must be taken into account. Research indicates only FSpO2 levels below 30% for more than 2 minutes18 or more than 10 minutes19 are likely to be associated with intrapartum acidosis.
ACOG has raised concerns about the potential increase in costs without demonstrable improvement in outcome.
Gorenberg et al20 retrospectively correlated FSpO2 with umbilical artery pH and found that neither the 30% threshold alone nor the duration of FSpO2 below 30% correlated with fetal acidemia (pH below 7.20). Rather, the repetition of such episodes was more predictive. The authors concluded that more than 10 episodes of FSpO2below 30% would overcome the ability of the fetus to compensate.
The study was underpowered to detect a significant difference in acidemia, and did not allow sufficient observation time to detect the natural progression of hypoxia to metabolic acidosis, a better indicator of fetal compromise. Additional research is needed.
Question 4Does oximetry correlate with acid-base status?
Many of the studies mentioned here assumed a correlation. Whenever oxygen saturation in the umbilical artery is 30% or more, acidosis (pH below 7.13) in the same blood is rare—only 1%.21 However, the correlation between fetal pulse oximetry values and acid-base status is much weaker.8.
Leszczynska-Gorzelak et al22 found no relationship between FSpO2 levels in the first or second stage of labor and pH or partial pressure of oxygen in umbilical vein blood at delivery. Other investigators concluded similarly, considering intrapartum FSpO2 of limited use for predicting acidosis at birth, irrespective of FSpO2 cutoff.23,24
Rijnders et al24 found no significant correlation between fetal scalp or umbilical artery blood pH and mean FSpO2 for the last 30 minutes before sampling (r = 0.02, P = .9). Even the lowest FSpO2 level did not correlate with arterial pH (r = .04, P = .84). None of the study’s 3 cases of umbilical pH below 7.05 would have been detected using the mean FSpO2 before delivery, and only 1 would have been detected using the lowest FSpO2.
In another multicenter study involving the Nellcor system in 164 cases with abnormal FHR, a correlation between oximetry and FSB sampling (r = 0.29, P < .01) was noted in the first stage of labor, but second-stage FSpO2 readings did not correlate with oxygen saturation, partial pressure of oxygen, pH, or bicarbonate level in the umbilical artery at birth.25
An observational series26 of 128 fetuses with nonreassuring FHR patterns concluded that fetal distress was insufficiently identified by oximetry. Only 2 of the 11 cases with umbilical artery pH below 7.20 were detected by pulse oximetry recordings below 30% during the last 30 minutes of the second stage, and out of 5 cases with hypoxic readings in the second stage, only 2 were acidotic at birth. The calculated sensitivity was 18%, specificity 92%, positive predictive value (PPV) 40%, and negative predictive value (NPV) 80%. A low Apgar score was never predicted by fetal pulse oximetry.
Others used the same Nellcor system over the final 30 minutes of labor and a cutoff for umbilical blood acidemia of pH below 7.13 and reported similar numbers: sensitivity 28%, specificity 94%, PPV 40%, and NPV 80%.23
Vitoratos et al27 analyzed FSpO2 readings in active labor (not limited to the last 30 minutes before delivery) and obtained somewhat better values: sensitivity 72%, specificity 93%, PPV 61.5%, and NPV 95.8% for an umbilical artery blood pH below 7.15.
The impression that the validity of fetal pulse oximetry is higher in earlier labor than in the second stage is supported by data from Stiller et al. 28 Leszczynska-Gorzelak et al29 found a significant decrease in mean FSpO2 from the first stage to the second stage of normal labor (51.9% versus 43.8%, P < .001), and Dildy et al14 noted a similar difference upon analyzing 160 normal labors (59% versus 53%), but other studies failed to verify such differences.25,30
Observational studies had unrealistic pH cutoff. All the evidence presented thus far on the validity of fetal pulse oximetry in predicting acidemia is based on observational data. A common deficiency is the unrealistic cutoff for pathologic fetal acidemia—a pH of less than 7.13 to 7.20—when it is widely accepted that “pathologic fetal acidemia” reflects an umbilical artery blood pH below 7.31 Even in this group, two thirds of neonates are unaffected by morbidity.
Need to identify metabolic acidosis. It also is accepted that the presence of a metabolic component to fetal acidemia may be as important—if not more important—than a single pH cutoff.31 Only a few human studies of pulse oximetry have distinguished between respiratory and metabolic acidemia. When they did, intrapartum fetal pulse oximetry was unable to predict umbilical artery base excess.23,25
The only randomized study failed to determine whether clinical decisions can be based solely on fetal pulse oximetry. 3 The investigators did suggest that sensitivity and specificity for metabolic acidemia was improved in the intervention group—a promising appraisal, in contrast with previous observational data.
In the study, 7 neonates (3 in the intervention group and 4 controls) had umbilical artery blood pH below 7. All 4 controls had vaginal delivery. There also were 6 cases of elevated base excess (ie, -16 mEq/L or below) among controls. None were recorded in the intervention group, and the 3 cases of acidemia were recognized antepartum and led to cesareans.
Unfortunately, the study design did not guarantee that patient management was based exclusively on EFM with or without fetal pulse oximetry. Vibro-acoustic stimulation or FSB sampling was required before proceeding to cesarean delivery in both groups.
It appears that the negative predictive value of fetal oximetry is of greater practical value than other attributes.
When FSpO2 was less than 30% for the entire interval between 2 contractions, or was unobtainable, the physician was supposed to revert to interpretation of EFM. When that was persistently nonreassuring, the physician was given the option of scalp stimulation or FSB sampling. Thus, it was not determined whether clinical decisions can be based exclusively on fetal pulse oximetry. Schmidt et al26 suggested that such exclusive application of fetal pulse oximetry might actually jeopardize fetal health.
Question 5 Does the combination of oximetry and EFM improve accuracy?
Fetal pulse oximetry was not used independently in any of the studies discussed here, but in association with EFM, which has a sensitivity for fetal acidosis of 93%, specificity of 29%, PPV of 2.6%, and NPV of 99.5%.32
From a statistical point of view, whenever 2 evaluation methods with the same endpoint (fetal acidosis) are combined, sensitivity decreases while specificity increases, theoretically resulting in less unnecessary intervention. That is exactly what investigators have reported: sensitivity as low as 18%26 for fetal oximetry, and specificity as high as 94%.23 However, the value of this new technology might not be so much the prediction of acidosis but identification of the well-oxygenated fetus so that labor may be safely continued in the presence of a concerning—but not ominous—FHR tracing.
Fetal pulse oximetry employs principles of optical spectrophotometry and plethysmography to provide information on the percentage of oxygen bound to hemoglobin. Oxyhemoglobin (oxygenated hemoglobin) and deoxyhemoglobin (hemoglobin without oxygen) absorb red and infrared light differently: more red absorption by deoxyhemoglobin, and more infrared absorption by oxyhemoglobin.
By measuring the relative absorption at each wavelength, the fraction of hemoglobin that carries oxygen can be determined. The arterial oxygen saturation is expressed as a percentage. The technology has been refined to measure fetal arterial oxyhemoglobin saturation during labor.
Pulse oximetry sensors must be calibrated for fetal biological values. In the fetus, normal oxygen saturation is much lower than in the adult or neonate; hemoglobin has a higher affinity for oxygen and is in higher concentration; and there are more capillaries per unit of tissue, higher cardiac output, and a higher heart rate.
In the adult or neonate, pulse oximetry sensors can be attached to fingers, toes, ears, or the bridge of the nose, but such stable placement is not feasible in utero. Further, good contact between sensor and fetal skin is a prerequisite for avoidance of artifacts. This last aspect has presented a sizeable challenge.
Fetal sensors measure reflected light. There is disagreement about the merits of the 2 sensor types, reflectance and transmission. Both include 2 light emitters (for red and infrared light) and a detector. In the transmission sensor (the adult or neonatal type), the light produced by the light-emitting diodes (LED) is picked up by the detector after traversing the interposed tissues. Since tissue interposition is not possible in the fetus, most fetal studies have used reflectance sensors, in which the LED and detector are placed side by side, and the light to be analyzed is reflected by the tissues. This design adds variance depending on the light’s depth of tissue penetration and device position changes.
Placement of the sensor. The Nellcor N-400 includes a reflectance sensor housed in a smooth, pliable head that is advanced through the cervix with the aid of a handle. The handle has a removable stylet to stiffen it during placement.
The sensor is placed against the fetal temple, cheek, or forehead and is held in place by the uterine wall. Placement is similar to that of an internal pressure catheter. Once the stylet is removed, it should not be reinserted.
Because the sensor usually descends and rotates with the fetal head, displacements are frequent and adjustments in sensor placement may be necessary. Placement adjustments can be attempted without the stylet and, if unsuccessful, a new device can be inserted. The Nellcor sensor is not reusable.
The prerequisites for insertion are dilatation of at least 2 cm, ruptured membranes, cephalic presentation, single fetus, gestational age of at least 36 weeks, and no placenta previa.
The manufacturer reports that active genital herpes, HIV, and hepatitis B or E seropositivity preclude fetal pulse oximetry monitoring.
Placement may be impossible when the presenting part is at high station (-3 or above) or low station (+2 or below).
The Nellcor N-400 system has been commercially available in many European countries since 1995, and in Canada since 1998. It was approved for sale in the United States in early 2003.
Question 6Will it improve neonatal outcomes?
Neonatal outcome is the ultimate endpoint in obstetrical care. In the randomized trial by Garite et al,3 there was no difference in neonatal outcome between the groups using or not using fetal pulse oximetry. According to Chua et al,33 FSpO2 levels measured even 10 minutes before delivery have no relation to neonatal outcome.
Leszczynska-Gorzelak et al22 believe FSpO2 is more predictive of neonatal outcome in the first stage than the second. However, Apgar score had no relationship with FSpO2readings in the first or second stage. Butterwegge34 reported 6 cases of FSpO2 below 30% for more than 30 minutes, all with good neonatal outcome, and Alshimmiri et al23 noted that only normal FSpO2 correlates with fetal well-being. Thus, it appears that the NPV of fetal oximetry is of greater practical value than other attributes.
Question 7How precise is it?
The Nellcor system monitors the quality of FSpO2 measurement; no value is displayed if the signal lacks the characteristics of a fetal arterial plethysmographic curve or if contact between sensor and skin is insufficient. Because of fetal movements and other artifacts, posting time is always less than 100%.
In the French multicenter study,25 the mean reliable signal time in the first stage of labor was only 64.7%—even less in the second stage (54%). Signal retention was 67% in the randomized trial by Garite et al. 3
Many artifacts may impede signal acquisition and impact the reliability of a reading:
- The sensor’s position on the fetal head. For example, the difference in FSpO2 readings between the forehead and occiput may be as much as 13.4%. (The sensor is designed to go against the fetal cheek, but may move around.)
- Incomplete sensor-to-skin contact, such as with high fetal head station, -2 or above.
- Marked caput formation.
- Increased intrauterine pressure accompanying contractions, especially at presentation stations of +2 or below (FIGURE 2). FSpO2 monitoring requires detection of fetal pulses, which may be undetectable when the surrounding pressure is high, resulting in a loss of signal.
- Interposition of vernix or fetal hair.
- Presence of meconium, which behaves like a red-light filter, altering the ratio of red to infrared light and resulting in artificially low values.35 This theoretical concern is rejected by Yam et al,36 who did not observe any effect of meconium on FSpO2 values. (When the amniotic fluid is meconium-stained, Carbonne et al37 showed that fetal oximetry is a better predictor of meconium aspiration syndrome than FSB sampling.) The data on the influence of meconium on FSpO2 readings remain contradictory.
All these conditions may impair precision and contribute to poor sensitivity.
FIGURE 2 A weakening signal during pushing
Question 8Is it easy to use?
An Australian survey38 assessed clinicians’ perceptions during placement of the oximetry sensor. Ease of placement was rated as good or excellent in 71% of cases, and the patient’s comfort was rated as good or excellent in 90% of cases. Chua et al39 reported a mean insertion time of 90 seconds, with a reliable signal obtained within 5 minutes in 87% of placements. The French multicenter study25 mentioned earlier concluded that the procedure is satisfactory and easier than FSB sampling. The device itself was harmless to both mother and fetus.40
Potential research directions
Fetal pulse oximetry may be an effective tool in clinical scenarios such as:
- fetal arrhythmias with uninterpretable FHR tracing
- fetal tachycardia associated with maternal fever, thyrotoxicosis, or fetal supraventricular tachycardia, when distinguishing other contributions to tachycardia may be difficult
- fetal bradycardia caused by a complete heart block, which may render EFM undecipherable
- when amnioinfusion is attempted for variable decelerations and it is necessary to differentiate a nonreassuring FHR tracing related to transient in utero stress (eg, umbilical cord compressions) from ominous tracings
Another area not yet addressed is cost-effectiveness beyond the immediate direct costs (approximately $11,000 for the monitor and $150 for each disposable sensor). Also uncertain is whether laboring women will accept the device (how disturbing or invasive it is perceived to be) and how acceptable or applicable it is outside tertiary institutions.
Dr. Vidaeff reports no financial relationships relevant to this article. Dr. Ramin reports grant support from the US National Institutes of Health.
1. Dildy GA. A guest editorial: fetal pulse oximetry. Obstet Gynecol Surv. 2003;58:225-226.
2. Thacker SB, Stroup DF, Peterson HB. Efficacy and safety of intrapartum electronic fetal monitoring: an update. Obstet Gynecol. 1995;86:613-620.
3. Garite TJ, Dildy GA, McNamara H, et al. A multicenter controlled trial of fetal pulse oximetry in the intrapartum management of nonreassuring fetal heart rate patterns. Am J Obstet Gynecol. 2000;183:1049-1058.
4. Haverkamp AD, Orleans M, Langendoerfer S, et al. A controlled trial of the differential effects of continuous fetal heart rate monitoring in labor: a randomized trial. Am J Obstet Gynecol. 1979;134:399-412.
5. American College of Obstetricians and Gynecologists. Committee opinion: fetal pulse oximetry. Number 258, September 2001. Obstet Gynecol. 2001;98:523-524.
6. Zijlstra WG, Buursma A, Zwart A. Performance of an automated six-wavelength photometer (radiometer OSM3) for routine measurement of hemoglobin derivatives. Clin Chem. 1988;34:149-152.
7. Porath M, Sinah P, Dudenhausen JW, Luttkus AK. Systematic instrumental errors between oxygen saturation analysers in fetal blood during deep hypoxemia. Clin Chim Acta. 2001;307:151-157.
8. Luttkus AK, Lübke M, Büscher U, Porath M, Dudenhausen JW. Accuracy of fetal pulse oximetry. Acta Obstet Gynecol Scand. 2002;81:417-423.
9. Carter AM, Stiller R, König V, Jorgensen JS, Svendsen P, Huch R. Calibration of a reflectance pulse oximeter in fetal lambs for arterial oxygen saturations below 70%. J Soc Gynecol Invest. 1998;5:255-259.
10. McNamara H, Cung CD, Lilford R, Johnson N. Do fetal pulse oximetry readings at delivery correlate with cord blood oxygenation and acidemia? Br J Obstet Gynaecol. 1992;99:735-738.
11. Langer B, Boudier E, Haddad J, Pain L, Schlaeder G. Fetal pulse oximetry during labor of 62 patients. Fetal Diagn Ther. 1996;11:37-45.
12. Harrison AM, Lynch JM, Dean JM, Witte MK. Comparison of simultaneously obtained arterial and capillary blood gases in pediatric intensive care unit patients. Crit Care Med. 1997;25:1904-1908.
13. Nijland R, Jongsma HW, Nijhuis JG, Oeseburg B. Accuracy of fetal pulse oximetry and pitfalls in measurements. Eur J Obstet Gynecol Reprod Biol. 1997;72:S21-S27.
14. Dildy GA, van den Berg PP, Katz M, et al. Intrapartum fetal pulse oximetry: fetal oxygen saturation trends during labor and relation to delivery outcome. Am J Obstet Gynecol. 1994;171:679-684.
15. Nijland R, Jongsma H, Nijhuis J, van den Berg P, Oeseburg B. Arterial saturation in relation to metabolic acidosis in fetal lambs. Am J Obstet Gynecol. 1995;172:810-819.
16. Kühnert M, Seelbach-Göbel B, Butterwegge M. Predictive agreement between fetal arterial oxygen saturation and fetal scalp pH: results of the German multicenter study. Am J Obstet Gynecol. 1998;178:330-335.
17. Salamalekis E, Thomopoulos P, Giannaris D, et al. Computerised intrapartum diagnosis of fetal hypoxia based on heart rate monitoring and fetal pulse oximetry recordings utilizing wavelet analysis and neural networks. Br J Obstet Gynaecol. 2002;109:1137-1142.
18. Bloom SL, Swindle RG, McIntire DD, Leveno KJ. Fetal pulse oximetry: duration of desaturation and intrapartum outcome. Obstet Gynecol. 1999;93:1036-1040.
19. Seelbach-Göbel B, Heupel M, Kühnert M, et al. The prediction of fetal acidosis by means of intrapartum fetal pulse oximetry. Am J Obstet Gynecol. 1999;180:73-81.
20. Gorenberg DM, Patillo C, Hendi P, Rumney PJ, Garite TJ. Fetal pulse oximetry: correlation between oxygen desaturation, duration, and frequency and neonatal outcomes. Am J Obstet Gynecol. 2003;189:136-138.
21. Dildy GA, Thorp JA, Yeast JD, Clark SL. The relationship between oxygen saturation and pH in umbilical blood: implications for intrapartum fetal oxygen saturation monitoring. Am J Obstet Gynecol. 1996;175:682-687.
22. Leszczynska-Gorzelak B, Poniedzialek-Czajkowska E, Oleszczuk J. Fetal blood saturation during the 1st and 2nd stage of labor and its relation to the neonatal outcome. Gynecol Obstet Invest. 2002;54:159-163.
23. Alshimmiri M, Bocking AD, Gagnon R, Natale R, Richardson BS. Prediction of umbilical artery base excess by intrapartum fetal oxygen saturation monitoring. Am J Obstet Gynecol. 1997;177:775-779.
24. Rijnders RJ, Mol BW, Reuwer PJ, Drogtrop AP, Vernooij MM, Visser GH. Is the correlation between fetal oxygen saturation and blood pH sufficient for the use of fetal pulse oximetry? J Matern Fetal Neonatal Med. 2002;11:80-83.
25. Goffinet F, Langer B, Carbonne B, et al. Multicenter study on the clinical value of fetal pulse oximetry. I. Methodologic evaluation. The French Study Group on Fetal Pulse Oximetry. Am J Obstet Gynecol. 1997;177:1238-1246.
26. Schmidt S, Koslowski S, Sierra F, et al. Clinical usefulness of pulse oximetry in the fetus with non-reassuring heart rate pattern? J Perinat Med. 2000;28:298-305.
27. Vitoratos N, Salamalekis E, Saloum J, Makrakis E, Creatsas G. Abnormal fetal heart rate patterns during the active phase of labor and the value of fetal oxygen saturation. J Matern Fetal Neonatal Med. 2002;11:46-49.
28. Stiller R, von Mering R, König V, et al. How well does reflectance pulse oximetry reflect intrapartum fetal acidosis? Am J Obstet Gynecol. 2002;186:1351-1357.
29. Leszczynska-Gorzelak B, Poniedzialek-Czajkowska E, Zych I, Grzechnik M, Oleszczuk J. The usefulness of the intrapartum fetal pulse oximetry in anticipating the neonatal outcome. Ginekol Pol. 2001;72:1183-1188.
30. Nikolov A, Dimitrov A, Vakrilkova L, Iarkova N. Fetal oxygen saturation during normal delivery. Akush Ginekol. 2000;40:3-6.
31. Goldaber KG, Gilstrap LC III, Leveno K, Dax JS, McIntire DD. Pathologic fetal acidemia. Obstet Gynecol. 1991;78:1103-1107.
32. Low JA, Victory R, Derrick EJ. Predictive value of electronic fetal monitoring for intra-partum fetal asphyxia and metabolic acidosis. Obstet Gynecol. 1999;93:285-291.
33. Chua S, Yeong SM, Razvi K, Arulkumaran S. Fetal oxygen saturation during labour. Br J Obstet Gynaecol. 1997;104:1080-1083.
34. Butterwegge M. Fetal pulse oximetry and non-reassuring heart rate. Eur J Obstet Gynecol Reprod Biol. 1997;72:S63-S66.
35. Johnson N, Johnson VA, Bannister J, McNamara H. The effect of meconium on neonatal and fetal reflectance pulse oximetry. J Perinat Med. 1990;18:351-355.
36. Yam J, Chua S, Arulkumaran S. Intrapartum pulse oximetry: Part 1: Principles and technical issues. Obstet Gynecol Surv. 2000;55:163-172.
37. Carbonne B, Cudeville C, Sivan H, Cabrol D, Papiernik E. Fetal oxygen saturation measured by pulse oximetry during labor with clear and meconium-stained amniotic fluid. Eur J Obstet Gynecol Reprod Biol. 1997;72:S51-S55.
38. East CE, Colditz PB. Clinicians’ perceptions of placing a fetal oximetry sensor. J Qual Clin Pract. 2000;20:161-163.
39. Chua S, Yam J, Razvi K, Yeong SM, Arulkumaran S. Intrapartum fetal oxygen saturation monitoring in a busy labour ward. Eur J Obstet Gynecol Reprod Biol. 1999;82:185-189.
40. Luttkus AK, Friedmann W, Thomas S, Dimer JA, Dudenhausen JW. The safety of fetal pulse oximetry in parturients requiring fetal scalp blood sample. Obstet Gynecol. 1997;90:533-537.
1. Dildy GA. A guest editorial: fetal pulse oximetry. Obstet Gynecol Surv. 2003;58:225-226.
2. Thacker SB, Stroup DF, Peterson HB. Efficacy and safety of intrapartum electronic fetal monitoring: an update. Obstet Gynecol. 1995;86:613-620.
3. Garite TJ, Dildy GA, McNamara H, et al. A multicenter controlled trial of fetal pulse oximetry in the intrapartum management of nonreassuring fetal heart rate patterns. Am J Obstet Gynecol. 2000;183:1049-1058.
4. Haverkamp AD, Orleans M, Langendoerfer S, et al. A controlled trial of the differential effects of continuous fetal heart rate monitoring in labor: a randomized trial. Am J Obstet Gynecol. 1979;134:399-412.
5. American College of Obstetricians and Gynecologists. Committee opinion: fetal pulse oximetry. Number 258, September 2001. Obstet Gynecol. 2001;98:523-524.
6. Zijlstra WG, Buursma A, Zwart A. Performance of an automated six-wavelength photometer (radiometer OSM3) for routine measurement of hemoglobin derivatives. Clin Chem. 1988;34:149-152.
7. Porath M, Sinah P, Dudenhausen JW, Luttkus AK. Systematic instrumental errors between oxygen saturation analysers in fetal blood during deep hypoxemia. Clin Chim Acta. 2001;307:151-157.
8. Luttkus AK, Lübke M, Büscher U, Porath M, Dudenhausen JW. Accuracy of fetal pulse oximetry. Acta Obstet Gynecol Scand. 2002;81:417-423.
9. Carter AM, Stiller R, König V, Jorgensen JS, Svendsen P, Huch R. Calibration of a reflectance pulse oximeter in fetal lambs for arterial oxygen saturations below 70%. J Soc Gynecol Invest. 1998;5:255-259.
10. McNamara H, Cung CD, Lilford R, Johnson N. Do fetal pulse oximetry readings at delivery correlate with cord blood oxygenation and acidemia? Br J Obstet Gynaecol. 1992;99:735-738.
11. Langer B, Boudier E, Haddad J, Pain L, Schlaeder G. Fetal pulse oximetry during labor of 62 patients. Fetal Diagn Ther. 1996;11:37-45.
12. Harrison AM, Lynch JM, Dean JM, Witte MK. Comparison of simultaneously obtained arterial and capillary blood gases in pediatric intensive care unit patients. Crit Care Med. 1997;25:1904-1908.
13. Nijland R, Jongsma HW, Nijhuis JG, Oeseburg B. Accuracy of fetal pulse oximetry and pitfalls in measurements. Eur J Obstet Gynecol Reprod Biol. 1997;72:S21-S27.
14. Dildy GA, van den Berg PP, Katz M, et al. Intrapartum fetal pulse oximetry: fetal oxygen saturation trends during labor and relation to delivery outcome. Am J Obstet Gynecol. 1994;171:679-684.
15. Nijland R, Jongsma H, Nijhuis J, van den Berg P, Oeseburg B. Arterial saturation in relation to metabolic acidosis in fetal lambs. Am J Obstet Gynecol. 1995;172:810-819.
16. Kühnert M, Seelbach-Göbel B, Butterwegge M. Predictive agreement between fetal arterial oxygen saturation and fetal scalp pH: results of the German multicenter study. Am J Obstet Gynecol. 1998;178:330-335.
17. Salamalekis E, Thomopoulos P, Giannaris D, et al. Computerised intrapartum diagnosis of fetal hypoxia based on heart rate monitoring and fetal pulse oximetry recordings utilizing wavelet analysis and neural networks. Br J Obstet Gynaecol. 2002;109:1137-1142.
18. Bloom SL, Swindle RG, McIntire DD, Leveno KJ. Fetal pulse oximetry: duration of desaturation and intrapartum outcome. Obstet Gynecol. 1999;93:1036-1040.
19. Seelbach-Göbel B, Heupel M, Kühnert M, et al. The prediction of fetal acidosis by means of intrapartum fetal pulse oximetry. Am J Obstet Gynecol. 1999;180:73-81.
20. Gorenberg DM, Patillo C, Hendi P, Rumney PJ, Garite TJ. Fetal pulse oximetry: correlation between oxygen desaturation, duration, and frequency and neonatal outcomes. Am J Obstet Gynecol. 2003;189:136-138.
21. Dildy GA, Thorp JA, Yeast JD, Clark SL. The relationship between oxygen saturation and pH in umbilical blood: implications for intrapartum fetal oxygen saturation monitoring. Am J Obstet Gynecol. 1996;175:682-687.
22. Leszczynska-Gorzelak B, Poniedzialek-Czajkowska E, Oleszczuk J. Fetal blood saturation during the 1st and 2nd stage of labor and its relation to the neonatal outcome. Gynecol Obstet Invest. 2002;54:159-163.
23. Alshimmiri M, Bocking AD, Gagnon R, Natale R, Richardson BS. Prediction of umbilical artery base excess by intrapartum fetal oxygen saturation monitoring. Am J Obstet Gynecol. 1997;177:775-779.
24. Rijnders RJ, Mol BW, Reuwer PJ, Drogtrop AP, Vernooij MM, Visser GH. Is the correlation between fetal oxygen saturation and blood pH sufficient for the use of fetal pulse oximetry? J Matern Fetal Neonatal Med. 2002;11:80-83.
25. Goffinet F, Langer B, Carbonne B, et al. Multicenter study on the clinical value of fetal pulse oximetry. I. Methodologic evaluation. The French Study Group on Fetal Pulse Oximetry. Am J Obstet Gynecol. 1997;177:1238-1246.
26. Schmidt S, Koslowski S, Sierra F, et al. Clinical usefulness of pulse oximetry in the fetus with non-reassuring heart rate pattern? J Perinat Med. 2000;28:298-305.
27. Vitoratos N, Salamalekis E, Saloum J, Makrakis E, Creatsas G. Abnormal fetal heart rate patterns during the active phase of labor and the value of fetal oxygen saturation. J Matern Fetal Neonatal Med. 2002;11:46-49.
28. Stiller R, von Mering R, König V, et al. How well does reflectance pulse oximetry reflect intrapartum fetal acidosis? Am J Obstet Gynecol. 2002;186:1351-1357.
29. Leszczynska-Gorzelak B, Poniedzialek-Czajkowska E, Zych I, Grzechnik M, Oleszczuk J. The usefulness of the intrapartum fetal pulse oximetry in anticipating the neonatal outcome. Ginekol Pol. 2001;72:1183-1188.
30. Nikolov A, Dimitrov A, Vakrilkova L, Iarkova N. Fetal oxygen saturation during normal delivery. Akush Ginekol. 2000;40:3-6.
31. Goldaber KG, Gilstrap LC III, Leveno K, Dax JS, McIntire DD. Pathologic fetal acidemia. Obstet Gynecol. 1991;78:1103-1107.
32. Low JA, Victory R, Derrick EJ. Predictive value of electronic fetal monitoring for intra-partum fetal asphyxia and metabolic acidosis. Obstet Gynecol. 1999;93:285-291.
33. Chua S, Yeong SM, Razvi K, Arulkumaran S. Fetal oxygen saturation during labour. Br J Obstet Gynaecol. 1997;104:1080-1083.
34. Butterwegge M. Fetal pulse oximetry and non-reassuring heart rate. Eur J Obstet Gynecol Reprod Biol. 1997;72:S63-S66.
35. Johnson N, Johnson VA, Bannister J, McNamara H. The effect of meconium on neonatal and fetal reflectance pulse oximetry. J Perinat Med. 1990;18:351-355.
36. Yam J, Chua S, Arulkumaran S. Intrapartum pulse oximetry: Part 1: Principles and technical issues. Obstet Gynecol Surv. 2000;55:163-172.
37. Carbonne B, Cudeville C, Sivan H, Cabrol D, Papiernik E. Fetal oxygen saturation measured by pulse oximetry during labor with clear and meconium-stained amniotic fluid. Eur J Obstet Gynecol Reprod Biol. 1997;72:S51-S55.
38. East CE, Colditz PB. Clinicians’ perceptions of placing a fetal oximetry sensor. J Qual Clin Pract. 2000;20:161-163.
39. Chua S, Yam J, Razvi K, Yeong SM, Arulkumaran S. Intrapartum fetal oxygen saturation monitoring in a busy labour ward. Eur J Obstet Gynecol Reprod Biol. 1999;82:185-189.
40. Luttkus AK, Friedmann W, Thomas S, Dimer JA, Dudenhausen JW. The safety of fetal pulse oximetry in parturients requiring fetal scalp blood sample. Obstet Gynecol. 1997;90:533-537.
Atypical squamous cells: The case for HPV testing
- Management by immediate colposcopy, repeat cytology, or HPV testing is acceptable for ASC-US, but testing for HPV is preferred when the Pap test is liquid-based.
- The sensitivity of HPV triage for high-grade CIN is essentially equivalent to colposcopy, and reduces the need for colposcopy by half.
- HPV testing is a good option for follow-up after treatment with cryosurgery, loop electrosurgical excision procedure, laser, or cold-knife conization.
What’s the best management strategy for the roughly 2 to 3 million women each yearUpdate on Cervical Disease, for commentary by Thomas C. Wright, Jr, MD, Department of Pathology, College of Physicians and Surgeons of Columbia University.The difference is even more pronounced when the cumulative 2-year detection rate for CIN 2,3 is added in for women referred for HPV-positive ASCUS but not found to have CIN 2,3 at initial colposcopy. That rate rises from 20.1% at initial colposcopy to 26.9% at 2 years.8 Although many experts consider even HPV-positive ASCUS of minimal risk, few would consider a risk of high-grade disease exceeding 1 in 4 to be minimal. In fact, 39% of the total CIN 2,3 cases reported from a routine screening population were detected following triage of ASCUS, and fully 69% were from all equivocal and low-grade Pap diagnoses.9
TABLE 1
Risk of cervical intraepithelial neoplasia grade 2 or greater at initial colposcopy
| ASCUS | ||||
|---|---|---|---|---|
| STUDY | HPV TEST | HPV–POSITIVE | HPV–NEGATIVE | TOTAL RISK FOR ALL ASCUS |
| Cox6 | Hybrid capture 1 (expanded first–generation test) | 17% (14/81) | 0.74% (1/136) | 6.9% (15/217) |
| Manos7 | Hybrid capture 2 | 15% (45/300) | 1.2% (6/498) | 6.4% (51/798) |
| Solomon4 (ALTS) | Hybrid capture 2 | 18% (195/1,087) | 1.1% (13/1,175) | 9.2% (208/2,262) |
| ALTS = ASCUS/LSIL Triage Study; ASCUS = atypical squamous cells of undetermined significance; HPV = human papillomavirus | ||||
Bethesda 3 redefines ASCUS
The third Bethesda System workshop took place in May 2001 with the aim of evaluating and updating earlier terminology.10 It began by eliminating the words “of undetermined significance” from the overall ASCUS category, which is now called simply “atypical squamous cells,” or ASC. Most subcategories of the former ASCUS were eliminated as well. (Note: Within this article, the acronyms ASCUS and ASC-US are both used to describe atypical squamous cells of undetermined significance. The latter acronym reflects usage and guidelines developed after the third Bethesda workshop.)
Now the ASC classification is broken down into 2 distinct groups:
Atypical squamous cells–undetermined significance, or ASC-US. This new subcategory includes cells previously termed “favor reactive” but not relegated by the pathologist to normal, as well as cells previously in the “unqualified” and “favor HPV” or “favor low-grade squamous intraepithelial lesion (LSIL)” subcategories.
Atypical squamous cells–cannot rule out high-grade squamous intraepithelial lesions, or ASC-H. This category includes atypical cells difficult to distinguish from high-grade cells but not definitive for that classification. Women with such Pap tests are at greater risk for high-risk HPV and histologic CIN 2,3 (TABLE 2).
Evidence-based guidelines reflect Bethesda 3 changes. By the time of Bethesda 3, extensive new data on the management of abnormal cytology was available, including but not limited to data from ALTS, making it possible to create evidence-based guidelines on management of abnormal cervical cytology and CIN. These guidelines were developed in 2001 at a consensus conference hosted by the American Society for Colposcopy and Cervical Pathology (ASCCP),11 with input from 29 professional organizations, federal agencies, and national and international health organizations.
The entire set recommendations for all types of abnormal Pap tests were published in the April 24, 2002 issue of the Journal of the American Medical Association, and management recommendations for histologically proven CIN were published in the July 2003 American Journal of Obstetrics and Gynecology and the July 2003 Journal of Lower Genital Tract Disease. The management algorithms for both cytology and histology can be downloaded from http://ASCCP.org.
TABLE 2
Comparison of risk for high-risk HPV and CIN grade 2,3, by Pap results
| HISTOLOGY | |||||
|---|---|---|---|---|---|
| PAP TEST | HIGH-RISK HPV | CIN 2 OR GREATER | CIN 3 | ||
| ASC-US | 63% | 12% | 5% | ||
| ASC-H | 86% | 40% | 24% | ||
| HSIL | 99% | 59% | 38% | ||
| Data from Sherman et al29 | |||||
| ASC-US = atypical squamous cells–undetermined significance; | |||||
| ASC-H = Atypical squamous cells–cannot rule out high-grade squamous intraepithelial lesion; CIN = cervical intraepithelial neoplasia; HPV = human papillomavirus; HSIL = high-grade squamous intraepithelial lesion | |||||
All 3 triage options safe, effective
An evidence-based review found all 3 options safe and effective.11 Therefore, management by immediate colposcopy, repeat cytology, or HPV testing is acceptable for ASCUS, but testing for HPV is preferred when the Pap test is liquid-based (FIGURE 1).
Liquid-based cytology (ThinPrep; Cytyc, Boxborough, Mass and SurePath, Raleigh-Durham, NC) has several advantages. For example, residual cells in the fluid can be tested for HPV, eliminating a return visit.
Immediate colposcopy: Low predictive value, high anxiety and expense. Proponents of immediate colposcopy for all women with ASC-US argue that this would theoretically detect all CIN 2,3 and cancer. However, the positive predictive value of this approach will always be extremely low due to the low rate (6.4% to 11.9%) of CIN 2,3 in women with ASCUS.4,6,7 What’s more, the cost and anxiety generated by immediate colposcopy are high.12
2 repeat cytologies: Sensitivity, cost issues. This approach requires at least 2 repeat, optimized (liquid-based) Pap tests to equal the sensitivity of a single HPV test. This, compounded with the high rate of repeat abnormal cytology requiring colposcopic evaluation, means repeat cytology is unlikely to be cost-competitive with HPV testing.4,13
- Cervical cytology as a triage option. Cytology has been a good screening test, but its comparatively low sensitivity (51% to 83%) and poor reproducibility reduces its value as a triage test.13-17 For example, in ALTS, of 1,473 repeat Paps originally read as ASCUS by good clinical pathologists, only 633 were reread as ASCUS when 2-of-3 agreement was obtained in a blinded review by an expert panel of pathologists.16 In other words, 840 (57%) were reread as something other than ASCUS. Most were downgraded to normal.
- The sensitivity of the HPV test in detecting CIN 2,3 was 92.4%. This rate was matched only by 2 repeat Pap tests, provided the threshold for referral to colposcopy was ASCUS or greater.17 At this threshold, 95% of the CIN 2,3 was detected with repeat Pap testing, but only after an average of 8 to 12 months. This contrasts with the immediate reassurance provided by the initial HPV test.
- ALTS did not evaluate repeat conventional Pap smears. Nor do the guidelines differentiate between conventional and liquid-based methods in the number of follow-up Pap tests required for reassurance, despite consensus that the sensitivity of liquid-based cytology is better than that of the conventional “dry slide.”
- Any woman with a repeat Pap result of ASC-US or greater should be referred to colposcopy. Referral at a threshold of LSIL or greater would result in far fewer colposcopies, but has not been shown to be sufficiently sensitive for CIN 2,3.17
HPV testing identifies clear risk. Any objective test that initially indicates which women with ASC-US are at risk for CIN 2,3 and which are not—either now or in the future—should confer a major advantage.
HPV-positive women are clearly at risk, justifying the anxiety and cost of colposcopic referral, while HPV-negative women may be reassured (FIGURE 2). Also, ALTS data showed HPV triage is essentially equivalent to immediate colposcopy in sensitivity for high-grade CIN, while halving colposcopic referrals.17,18
Because low-risk HPV types do not cause CIN 3 or cancer, the HPV test should document only high-risk types.11 The only HPV test approved by the US Food and Drug Administration (Hybrid Capture 2, Digene, Gaithersburg, Md) includes both low- and high-risk HPV panels. For cost savings, the laboratory can be asked to use only the high-risk panel. All positive high-risk HPV cases should be referred to colposcopy.
FIGURE 1 3 triage options for management of ASC-US
FIGURE 2 Management of atypical squamous cells–cannot exclude high–grade squamous intraepithelial lesions (ASC-H)
Some high-grade lesions are still overlooked
A single HPV test or 2 repeat liquid-based Pap tests with a colposcopy-referral threshold of any findings of ASC-US or greater have similar sensitivity for CIN 2,3.17,18
The guidelines state that women who undergo immediate colposcopy with negative results or who have a negative initial HPV test should undergo a follow-up Pap test in 12 months. Note that the guidelines do not state that these women can return directly to routine screening. The reason: In some settings, “routine” screening is at 2- or 3-year intervals, and some risk still exists—albeit minimal—for missed CIN 2,3.
For example, 1 of 83 cases of CIN 2,3 were missed by HPV testing in the study by Manos et al.7 In ALTS, that number was 1 in 90,4 and in the study by Cox et al6 it was 1 in 136. Further, colposcopy did not initially detect 25% of the cumulative high-grade lesions detected over 2 years of follow-up in ALTS.17
In contrast, the recommendation for women with 2 repeat normal Pap tests is to return to “routine screening.” This inexplicably departs from the 12-month repeat Pap testing urged for women with negative results on the other 2 triage options, despite a similar risk of missed high-grade disease.
- In my opinion, all 3 scenarios should be managed by repeat Pap testing in 12 months.
Reducing referrals to colposcopy
If all women returned as directed for repeat cytology, more of them would be referred to colposcopy by repeat abnormal Pap tests at the ASC-US threshold than by testing positive for high-risk HPV types. In ALTS, 53% tested positive for high-risk HPV and were referred to colposcopy, compared with 67% who had an abnormal Pap test on the first or second repeat (these women also had 1 or 2 more office visits prior to referral to colposcopy.).
No difference for conventional smears. All the advantages of HPV testing in the triage of women with ASC-US persist when the initial referral Pap test is a conventional smear. The only exception is that HPV testing would require the patient to return for a repeat office visit. An alternative would be co-collecting an HPV-test sample at the time of the primary screening Pap test.
One major health-maintenance organization collects a separate sample from all women when the routine conventional Pap test is obtained using a standard Hybrid Capture 2 HPV test kit. The HPV-testing samples are then held until the results of the Pap smear are reported. For women reported to have ASCUS, the samples are sent to the lab for HPV testing; the remaining samples (approximately 95% in most practices) are discarded as medical waste. The cost of each discarded kit is approximately $1. Modeling has found this approach to be cost-effective.19
Postcolposcopy management
Many clinicians are concerned that women referred for the evaluation of HPV-positive ASC-US and found not to have CIN or other manifestations of HPV at colposcopy have a “false-positive” HPV test. However, although there are occasional HPV tests that misclassify a low-risk HPV type as high-risk, actual false-positive tests are very rare.
The 2-year ALTS longitudinal data provide the best information on what to expect when a woman with HPV-positive ASC-US or LSIL is found at colposcopy to have no CIN or to have only CIN 1 that is subsequently managed expectantly.8
The cumulative risk of CIN 2,3 over the 2 years was nearly equivalent for women referred initially for LSIL (27.6%) and for women referred for HPV-positive ASCUS (26.7%), further verifying that management should be similar. Two thirds of the CIN 2,3 was detected at initial colposcopy, and the remaining one third during the postcolposcopy 2-year follow-up.
The risk for subsequent detection of high-grade CIN was nearly identical for all women initially found not to have CIN 2,3 regardless of whether CIN 1 was detected at initial colposcopy, whether the colposcopy was initially completely normal, or whether there were changes that were biopsied and found not to have CIN (risk for CIN 2,3 was 13%, 11.3%, and 11.7% respectively).
Hence, all women referred for evaluation of HPV-positive ASC-US or LSIL and not treated for CIN 2,3 require similar diligent follow-up.
A single HPV test at 12 months detected 92% of all CIN 2,3 found over the 24-month follow-up; 55% tested HPV-positive and were referred to colposcopy.20 Repeat liquid-based cytology at 6 and 12 months referred to colposcopy 63% of women (using a threshold of a repeat Pap test of ASCUS or greater). Cumulative sensitivity of 2 repeat cytologies for CIN 2,3 was slightly less (88%). Combining a repeat Pap test with an HPV test did not increase sensitivity, but did significantly increase referral to colposcopy.
An HPV test alone at 12 months might be the most efficient test for identifying women with CIN 2,3 after colposcopy.20 Further support for this approach can be found in the substantial body of evidence showing that only persistent HPV progresses to CIN 321 and that testing for high-risk HPV detects most CIN 3.4,17,20
The ASCCP guidelines for women referred for either HPV-positive ASC-US or LSIL and found not to have CIN 2,3 or greater at initial colposcopy recommend either HPV testing at 12 months or repeat cytology at 6 and 12 months (FIGURE 1).11,22
Posttreatment follow-up. The ASCCP treatment guidelines also list HPV testing as an acceptable option for follow-up after treatment with cryosurgery, loop electrosurgical excision procedure, laser, or cold-knife conization,22 since there is substantial evidence that women successfully treated for CIN become HPV-negative, whereas women with persistent disease remain HPV-positive.23-25
A posttreatment HPV test should be performed no sooner than 6 months following the procedure, as it takes time for the patient to return to HPV-negative status. A positive HPV test is an indication for colposcopy. However, the guidelines advise against basing repeat treatment on a positive HPV test alone without documentation of persistent CIN.22
Other options for posttreatment surveillance include either repeat cytology or a combination of Pap testing and colposcopy at 4- to 6-month intervals until at least 3 cytologic results are “negative for squamous intraepithelial lesion or malignancy.”22
Annual cytologic follow-up is recommended thereafter. During that follow-up, any abnormal Pap test (ASC-US or greater) should be referred to colposcopy.
Managing ASC-US in special populations
Management of ASC-US may differ from the general recommendations when the patient is postmenopausal or immunosuppressed. However, there are no differences in the management guidelines during pregnancy.
HPV-negative postmenopausal patients. All 3 management options—immediate colposcopy, repeat cytology, and HPV DNA testing—are acceptable for postmenopausal women with ASC-US.11 However, estrogen deficiency is a common cause of ASC-US and is responsible for increasing rates of HPV-negative ASC-US in this age group despite high sensitivity of HPV testing for CIN 2 and 3.5
- Treatment with vaginal estrogen cream followed by repeat cytology approximately 1 week after completing the regimen is an option for postmenopausal women with ASC-US. This approach also may be helpful for perimenopausal women and for women of any age on progestin-only contraception who have clinical or cytologic evidence of atrophy.
- Women with ASC-US or greater on repeat cytology should be referred for colposcopy, whereas women with normal repeat cytology should have a second Pap test in 4 to 6 months. Repeating the course of vaginal estrogen prior to each Pap test may be helpful when atrophy is likely to persist. After 2 normal repeat Pap tests, the patient can return to routine screening.
Refer all immunosuppressed women for colposcopy. The management of ASC-US in HIV-infected women is particularly problematic because the rates of ASC-US and HPV detection are 2 to 3 times greater than in HIV-negative women. In addition, the risk of CIN 2 and 3 is much higher.26 HPV testing as a triage for ASC-US is not efficient in immunosuppressed women because the majority of ASC-US Pap tests in these women are HPV-positive.
ASCCP recommends colposcopy referral of all immunosuppressed women with ASCUS Pap test, regardless of their CD4 count, HIV viral load, or anti-retroviral therapy.26
Managing ASC-H: First, colposcopy
Clearly, women with ASC-H test results face a greater risk for CIN 2,3 and should be referred for immediate colposcopy.
The ASC-H designation is uncommon, reported in 0.27% to 0.6% of all Pap tests,27,28 or approximately 1 in 10 Pap smears read as ASC.
In ALTS, HPV testing and histology results were compared for women with Pap tests categorized as equivocal LSIL (ASCUSL), ASCUS-H, and high-grade squamous intraepithelial lesion (HSIL) (TABLE 2).29 High-risk HPV DNA was detected in 86% of ASCUS-H liquid-based Pap tests and 69.8% of ASCUS-H conventional smears. CIN 2,3 was found in 40% of liquid-based ASCUS-H smears and in 27.2% of conventional ASCUSH smears. A 3-year retrospective review of ASC-H with follow-up at Johns Hopkins Medical Institutions determined that 49% of patients had no CIN or glandular lesions.28 Of the 51% with CIN, approximately half the lesions were CIN 1 and half were CIN 2,3.
Further management depends on whether CIN is detected (FIGURE 3). If no CIN is found, the ASCCP guidelines recommend that cytology, colposcopy, and histology be reviewed. If there is a change in the diagnosis—eg, if the Pap interpretation is revised to HSIL—the patient should be managed accordingly.11
If there is no change, the patient should be followed with repeat cytology at 6-and 12-month intervals or HPV testing at 12 months. Women having any repeat abnormal Pap test at a threshold of ASC-US or greater or a positive HPV test should undergo repeat colposcopy.
ASC-H is of greater risk than ASC-US, but it is not as risky as HSIL. Therefore, a surgical excision procedure in the absence of documented CIN 2,3 would not normally be indicated.11
FIGURE 3 Management of atypical squamous cells–cannot exclude high–grade squamous intraepithelial lesions (ASC-H)
HPV test as triage option would mean retooling the system
Cytologic management systems have traditionally involved follow-up by repeat cytology, colposcopy, and, when necessary, treatment. Adding another triage option—HPV testing—requires that this system be retooled.
The labs. This is not difficult when the laboratory interpreting the liquid-based Pap test is the same lab that performs the “reflex” HPV test, as this allows the ASC-US Pap test to be reported as HPV-negative or HPV-positive. However, if the HPV test must be performed in a separate reference laboratory, the results of the Pap and HPV tests will arrive separately, and the clinician must collate the 2 reports before relaying the result to the patient.
The patients. Remember than an HPV test is a test for a sexually transmitted disease. (So is the Pap test, although it has not traditionally been considered as such.) For that reason, I give all patients a written explanation of the rationale behind testing ASC-US Pap tests for HPV. This explanation includes 2 check-off options at the bottom of the sheet where patients can indicate whether they would prefer HPV testing or one of the other follow-up options.
Most patients elect the HPV option. Our Pap test requisitions also have a check-off portion that allows us to notify the lab of patients who wants an HPV test if the Pap is interpreted as ASC-US.
The office staff. Whenever a new test or procedure is introduced, it is of primary importance that the office staff responsible for completing critical information on the requisition form is adequately trained. This involves knowing when and how to order the test and how to complete insurance information and clinical history on the Pap requisition—including the correct International Classification of Diseases, Ninth Revision code—to ensure that the HPV test is covered by the patient’s insurer.
Clinicians must understand the usually benign nature of HPV infection. Reporting a positive HPV test in a manner that is not unduly concerning requires reassuring and nonjudgmental communication of the results based on a broad understanding of the usually low-risk natural history of the virus, yet fosters responsible follow-up.
Why all HPV-positive ASC-US requires diligent follow-up
The recently released ASCCP guidelines recognize HPV testing as an option in the management of ASC results, including:
- initial management of ASC-US,
- postcolposcopy management of ASC-H or HPV-positive ASC Pap tests found to be normal or to have CIN 1, and
- posttreatment follow-up.
For each indication, the HPV test identifies women most likely to have CIN (HPV-positive) and those likely to have benign processes not related to HPV (HPV-negative).
New longitudinal data verify that women with HPV-positive ASC-US continue at risk for detection of CIN 2,3 (about 12% overall), whether the original colposcopic finding was normal or CIN 1.8 Therefore, they need continued diligent follow-up.
Dr. Cox serves on the Speaker’s Bureaus of Cytyc Corporation and Digene Corporation.
1. Kurman RJ, Henson DE, Berbst AL, Noller KL, Schiffman MH. Interim guidelines for the management of abnormal cervical cytology. JAMA. 1994;271:1866-1869.
2. Ferenczy A. Viral testing for genital human papillomavirus infections: recent progress and clinical potentials. Int J Gynecol Cancer. 1995;5:321-328.
3. Schiffman M, Adrianza ME. ASCUS-LSIL Triage Study. Design, methods and characteristics of trial participants. Acta Cytol. 2000;44:726-742.
4. Solomon D, Schiffman M, Tarone R, et al. Comparison of three management strategies for patients with atypical squamous cells of undetermined significance: baseline results from a randomized trial. J Natl Cancer Inst. 2001;93:293-299.
5. Sherman ME, Schiffman M, Cox JT, et al. Effects of age and human papilloma viral load on colposcopy triage: data from the randomized Atypical Squamous Cells of Undetermined Significance/Low-Grade Squamous Intraepithelial Lesion Triage Study (ALTS). J Natl Cancer Inst. 2002;94(2):102-107.
6. Cox JT, Lorincz AT, Schiffman MH, Sherman ME, Cullen A, Kurman RJ. Human papillomavirus testing by hybrid capture appears to be useful in triaging women with a cytologic diagnosis of atypical squamous cells of undetermined significance. Am J Obstet Gynecol. 1995;172:946-954.
7. Manos MM, Kinney WK, Hurley LB, et al. Identifying women with cervical neoplasia: using human papillomavirus DNA testing for equivocal Papanicolaou results. JAMA. 1999;281:1605-1610.
8. Cox JT, Schiffman M, Solomon D, et al. Prospective follow-up suggests similar risk of subsequent cervical intraepithelial neoplasia grade 2 or 3 among women with cervical intraepithelial neoplasia grade 1 or negative colposcopy and directed biopsy. Am J Obstet Gynecol. 2003;188:1406-1412.
9. Kinney WK, Manos MM, Hurley LB, Ransley JE. Where’s the high-grade cervical neoplasia? The importance of the minimally abnormal Papanicolaou diagnosis. Obstet Gynecol. 1998;91:973-976.
10. Solomon D, Davey D, Kurman R, et al. The 2001 Bethesda System: terminology for reporting results of cervical cytology. JAMA. 2002;287:2114-2119.
11. Wright TC, Jr, Cox JT, Massad LS, et al. 2001 Consensus Guidelines for the management of women with cervical cytological abnormalities. JAMA. 2002;287:2120-2129.
12. Jones MH, Singer A, Jenkins D. The mildly abnormal cervical smear: patient awareness and choice of management. J Royal Soc Med. 1996;89:257.-
13. Wright TC, Lorincz AT, Ferris DG, et al. Reflex human papillomavirus deoxyribonucleic acid testing in women with abnormal Pap smears. Am J Obstet Gynecol. 1998;178:926-966.
14. Fahey MT, Irwig L, Macaskill P. Meta-analysis of Pap test accuracy. Am J Epidemiol. 1995;141:680-689.
15. Agency for Health Care Policy and Research. Evidence Report/Technology Assessment #5: Evaluation of Cervical Cytology. Rockville, Md: AHCPR; January 1999.
16. Stoler MH, Schiffman M. Atypical Squamous Cells of Undetermined Significance—Low-grade Squamous Intraepithelial Lesion Triage Study (ALTS) Group. Interobserver reproducibility of cervical cytologic and histologic interpretations: realistic estimates from the ASCUS-LSIL Triage Study. JAMA. 2001;285:1500-1505.
17. ASCUS-LSIL Triage Study (ALTS) Group. Results of a randomized trial on the management of cytology interpretations of atypical squamous cells of undetermined significance. Am J Obstet Gynecol. 2003;188:1383-1392.
18. Schiffman M, Solomon D. Findings to date from the ASCUS-LSIL Triage Study (ALTS). Arch Pathol Lab Med. 2003;127:946-949.
19. Kim JJ, Wright TC, Goldie SJ. Cost-effectiveness of alternative triage strategies for atypical squamous cells of undetermined significance. JAMA. 2002;287:2382-2390.
20. Guido R, Schiffman M, Solomon D, et al. Postcolposcopy management strategies for women referred with low-grade squamous intraepithelial lesions or human papillomavirus DNA-positive atypical squamous cells of undetermined significance: a two-year prospective study. Am J Obstet Gynecol. 2003;188:1401-1405.
21. Nobbenhuis M, Walboomers JM, Helmerhorst TI, Rozendaal L. Relation of human papillomavirus status to cervical lesions and consequences for cervical-cancer screening: a prospective study. Lancet. 1999;354:20.-
22. Wright TC, Jr, Cox JT, Massad LS, et al. 2001 consensus guidelines for the management of women with cervical intraepithelial neoplasia. Am J Obstet Gynecol. 2003;189:295-304.
23. Paraskevaidis E, Koliopoulos G, Alamanos Y, Malamou-Mitsi V, Lolis ED, Kitchener HC. Human papillomavirus testing and the outcome of treatment for cervical intraepithelial neoplasia. Obstet Gynecol. 2001;98:833-836.
24. Nobbenhuis MA, Meijer CJ, van den Brule AJ, et al. Addition of high-risk HPV testing improves the current guidelines on follow-up after treatment for cervical intraepithelial neoplasia. Br J Cancer. 2001;84:796-801.
25. Jain S, Tseng CJ, Horng SG, Soong YK, Pao CC. Negative predictive value of human papillomavirus test following conization of the cervix uteri. Gynecol Oncol. 2001;82:177-180.
26. Massad LS, Ahdieh L, Benning L, et al. Evolution of cervical abnormalities among women with HIV-1: evidence from surveillance cytology in the women’s interagency HIV study. J Acquir Immune Defic Syndr. 2001;27:432-442.
27. Selvaggi SM. Reporting of atypical squamous cells, cannot exclude a high-grade squamous intraepithelial lesion (ASC-H) on cervical samples: is it significant? Diagn Cytopathol. 2003;29:38-41.
28. Alli PM, Ali SZ. Atypical squamous cells of undetermined significance—rule out high-grade squamous intraepithelial lesion: cytopathologic characteristics and clinical correlates. Diagn Cytopathol. 2003;28:308-312.
29. Sherman ME, Solomon D, Schiffman M, et al. A comparison of equivocal LSIL and equivocal HSIL cervical cytology in the ASCUS LSIL Triage Study. Am J Clin Pathol. 2001;116:386-394.
- Management by immediate colposcopy, repeat cytology, or HPV testing is acceptable for ASC-US, but testing for HPV is preferred when the Pap test is liquid-based.
- The sensitivity of HPV triage for high-grade CIN is essentially equivalent to colposcopy, and reduces the need for colposcopy by half.
- HPV testing is a good option for follow-up after treatment with cryosurgery, loop electrosurgical excision procedure, laser, or cold-knife conization.
What’s the best management strategy for the roughly 2 to 3 million women each yearUpdate on Cervical Disease, for commentary by Thomas C. Wright, Jr, MD, Department of Pathology, College of Physicians and Surgeons of Columbia University.The difference is even more pronounced when the cumulative 2-year detection rate for CIN 2,3 is added in for women referred for HPV-positive ASCUS but not found to have CIN 2,3 at initial colposcopy. That rate rises from 20.1% at initial colposcopy to 26.9% at 2 years.8 Although many experts consider even HPV-positive ASCUS of minimal risk, few would consider a risk of high-grade disease exceeding 1 in 4 to be minimal. In fact, 39% of the total CIN 2,3 cases reported from a routine screening population were detected following triage of ASCUS, and fully 69% were from all equivocal and low-grade Pap diagnoses.9
TABLE 1
Risk of cervical intraepithelial neoplasia grade 2 or greater at initial colposcopy
| ASCUS | ||||
|---|---|---|---|---|
| STUDY | HPV TEST | HPV–POSITIVE | HPV–NEGATIVE | TOTAL RISK FOR ALL ASCUS |
| Cox6 | Hybrid capture 1 (expanded first–generation test) | 17% (14/81) | 0.74% (1/136) | 6.9% (15/217) |
| Manos7 | Hybrid capture 2 | 15% (45/300) | 1.2% (6/498) | 6.4% (51/798) |
| Solomon4 (ALTS) | Hybrid capture 2 | 18% (195/1,087) | 1.1% (13/1,175) | 9.2% (208/2,262) |
| ALTS = ASCUS/LSIL Triage Study; ASCUS = atypical squamous cells of undetermined significance; HPV = human papillomavirus | ||||
Bethesda 3 redefines ASCUS
The third Bethesda System workshop took place in May 2001 with the aim of evaluating and updating earlier terminology.10 It began by eliminating the words “of undetermined significance” from the overall ASCUS category, which is now called simply “atypical squamous cells,” or ASC. Most subcategories of the former ASCUS were eliminated as well. (Note: Within this article, the acronyms ASCUS and ASC-US are both used to describe atypical squamous cells of undetermined significance. The latter acronym reflects usage and guidelines developed after the third Bethesda workshop.)
Now the ASC classification is broken down into 2 distinct groups:
Atypical squamous cells–undetermined significance, or ASC-US. This new subcategory includes cells previously termed “favor reactive” but not relegated by the pathologist to normal, as well as cells previously in the “unqualified” and “favor HPV” or “favor low-grade squamous intraepithelial lesion (LSIL)” subcategories.
Atypical squamous cells–cannot rule out high-grade squamous intraepithelial lesions, or ASC-H. This category includes atypical cells difficult to distinguish from high-grade cells but not definitive for that classification. Women with such Pap tests are at greater risk for high-risk HPV and histologic CIN 2,3 (TABLE 2).
Evidence-based guidelines reflect Bethesda 3 changes. By the time of Bethesda 3, extensive new data on the management of abnormal cytology was available, including but not limited to data from ALTS, making it possible to create evidence-based guidelines on management of abnormal cervical cytology and CIN. These guidelines were developed in 2001 at a consensus conference hosted by the American Society for Colposcopy and Cervical Pathology (ASCCP),11 with input from 29 professional organizations, federal agencies, and national and international health organizations.
The entire set recommendations for all types of abnormal Pap tests were published in the April 24, 2002 issue of the Journal of the American Medical Association, and management recommendations for histologically proven CIN were published in the July 2003 American Journal of Obstetrics and Gynecology and the July 2003 Journal of Lower Genital Tract Disease. The management algorithms for both cytology and histology can be downloaded from http://ASCCP.org.
TABLE 2
Comparison of risk for high-risk HPV and CIN grade 2,3, by Pap results
| HISTOLOGY | |||||
|---|---|---|---|---|---|
| PAP TEST | HIGH-RISK HPV | CIN 2 OR GREATER | CIN 3 | ||
| ASC-US | 63% | 12% | 5% | ||
| ASC-H | 86% | 40% | 24% | ||
| HSIL | 99% | 59% | 38% | ||
| Data from Sherman et al29 | |||||
| ASC-US = atypical squamous cells–undetermined significance; | |||||
| ASC-H = Atypical squamous cells–cannot rule out high-grade squamous intraepithelial lesion; CIN = cervical intraepithelial neoplasia; HPV = human papillomavirus; HSIL = high-grade squamous intraepithelial lesion | |||||
All 3 triage options safe, effective
An evidence-based review found all 3 options safe and effective.11 Therefore, management by immediate colposcopy, repeat cytology, or HPV testing is acceptable for ASCUS, but testing for HPV is preferred when the Pap test is liquid-based (FIGURE 1).
Liquid-based cytology (ThinPrep; Cytyc, Boxborough, Mass and SurePath, Raleigh-Durham, NC) has several advantages. For example, residual cells in the fluid can be tested for HPV, eliminating a return visit.
Immediate colposcopy: Low predictive value, high anxiety and expense. Proponents of immediate colposcopy for all women with ASC-US argue that this would theoretically detect all CIN 2,3 and cancer. However, the positive predictive value of this approach will always be extremely low due to the low rate (6.4% to 11.9%) of CIN 2,3 in women with ASCUS.4,6,7 What’s more, the cost and anxiety generated by immediate colposcopy are high.12
2 repeat cytologies: Sensitivity, cost issues. This approach requires at least 2 repeat, optimized (liquid-based) Pap tests to equal the sensitivity of a single HPV test. This, compounded with the high rate of repeat abnormal cytology requiring colposcopic evaluation, means repeat cytology is unlikely to be cost-competitive with HPV testing.4,13
- Cervical cytology as a triage option. Cytology has been a good screening test, but its comparatively low sensitivity (51% to 83%) and poor reproducibility reduces its value as a triage test.13-17 For example, in ALTS, of 1,473 repeat Paps originally read as ASCUS by good clinical pathologists, only 633 were reread as ASCUS when 2-of-3 agreement was obtained in a blinded review by an expert panel of pathologists.16 In other words, 840 (57%) were reread as something other than ASCUS. Most were downgraded to normal.
- The sensitivity of the HPV test in detecting CIN 2,3 was 92.4%. This rate was matched only by 2 repeat Pap tests, provided the threshold for referral to colposcopy was ASCUS or greater.17 At this threshold, 95% of the CIN 2,3 was detected with repeat Pap testing, but only after an average of 8 to 12 months. This contrasts with the immediate reassurance provided by the initial HPV test.
- ALTS did not evaluate repeat conventional Pap smears. Nor do the guidelines differentiate between conventional and liquid-based methods in the number of follow-up Pap tests required for reassurance, despite consensus that the sensitivity of liquid-based cytology is better than that of the conventional “dry slide.”
- Any woman with a repeat Pap result of ASC-US or greater should be referred to colposcopy. Referral at a threshold of LSIL or greater would result in far fewer colposcopies, but has not been shown to be sufficiently sensitive for CIN 2,3.17
HPV testing identifies clear risk. Any objective test that initially indicates which women with ASC-US are at risk for CIN 2,3 and which are not—either now or in the future—should confer a major advantage.
HPV-positive women are clearly at risk, justifying the anxiety and cost of colposcopic referral, while HPV-negative women may be reassured (FIGURE 2). Also, ALTS data showed HPV triage is essentially equivalent to immediate colposcopy in sensitivity for high-grade CIN, while halving colposcopic referrals.17,18
Because low-risk HPV types do not cause CIN 3 or cancer, the HPV test should document only high-risk types.11 The only HPV test approved by the US Food and Drug Administration (Hybrid Capture 2, Digene, Gaithersburg, Md) includes both low- and high-risk HPV panels. For cost savings, the laboratory can be asked to use only the high-risk panel. All positive high-risk HPV cases should be referred to colposcopy.
FIGURE 1 3 triage options for management of ASC-US
FIGURE 2 Management of atypical squamous cells–cannot exclude high–grade squamous intraepithelial lesions (ASC-H)
Some high-grade lesions are still overlooked
A single HPV test or 2 repeat liquid-based Pap tests with a colposcopy-referral threshold of any findings of ASC-US or greater have similar sensitivity for CIN 2,3.17,18
The guidelines state that women who undergo immediate colposcopy with negative results or who have a negative initial HPV test should undergo a follow-up Pap test in 12 months. Note that the guidelines do not state that these women can return directly to routine screening. The reason: In some settings, “routine” screening is at 2- or 3-year intervals, and some risk still exists—albeit minimal—for missed CIN 2,3.
For example, 1 of 83 cases of CIN 2,3 were missed by HPV testing in the study by Manos et al.7 In ALTS, that number was 1 in 90,4 and in the study by Cox et al6 it was 1 in 136. Further, colposcopy did not initially detect 25% of the cumulative high-grade lesions detected over 2 years of follow-up in ALTS.17
In contrast, the recommendation for women with 2 repeat normal Pap tests is to return to “routine screening.” This inexplicably departs from the 12-month repeat Pap testing urged for women with negative results on the other 2 triage options, despite a similar risk of missed high-grade disease.
- In my opinion, all 3 scenarios should be managed by repeat Pap testing in 12 months.
Reducing referrals to colposcopy
If all women returned as directed for repeat cytology, more of them would be referred to colposcopy by repeat abnormal Pap tests at the ASC-US threshold than by testing positive for high-risk HPV types. In ALTS, 53% tested positive for high-risk HPV and were referred to colposcopy, compared with 67% who had an abnormal Pap test on the first or second repeat (these women also had 1 or 2 more office visits prior to referral to colposcopy.).
No difference for conventional smears. All the advantages of HPV testing in the triage of women with ASC-US persist when the initial referral Pap test is a conventional smear. The only exception is that HPV testing would require the patient to return for a repeat office visit. An alternative would be co-collecting an HPV-test sample at the time of the primary screening Pap test.
One major health-maintenance organization collects a separate sample from all women when the routine conventional Pap test is obtained using a standard Hybrid Capture 2 HPV test kit. The HPV-testing samples are then held until the results of the Pap smear are reported. For women reported to have ASCUS, the samples are sent to the lab for HPV testing; the remaining samples (approximately 95% in most practices) are discarded as medical waste. The cost of each discarded kit is approximately $1. Modeling has found this approach to be cost-effective.19
Postcolposcopy management
Many clinicians are concerned that women referred for the evaluation of HPV-positive ASC-US and found not to have CIN or other manifestations of HPV at colposcopy have a “false-positive” HPV test. However, although there are occasional HPV tests that misclassify a low-risk HPV type as high-risk, actual false-positive tests are very rare.
The 2-year ALTS longitudinal data provide the best information on what to expect when a woman with HPV-positive ASC-US or LSIL is found at colposcopy to have no CIN or to have only CIN 1 that is subsequently managed expectantly.8
The cumulative risk of CIN 2,3 over the 2 years was nearly equivalent for women referred initially for LSIL (27.6%) and for women referred for HPV-positive ASCUS (26.7%), further verifying that management should be similar. Two thirds of the CIN 2,3 was detected at initial colposcopy, and the remaining one third during the postcolposcopy 2-year follow-up.
The risk for subsequent detection of high-grade CIN was nearly identical for all women initially found not to have CIN 2,3 regardless of whether CIN 1 was detected at initial colposcopy, whether the colposcopy was initially completely normal, or whether there were changes that were biopsied and found not to have CIN (risk for CIN 2,3 was 13%, 11.3%, and 11.7% respectively).
Hence, all women referred for evaluation of HPV-positive ASC-US or LSIL and not treated for CIN 2,3 require similar diligent follow-up.
A single HPV test at 12 months detected 92% of all CIN 2,3 found over the 24-month follow-up; 55% tested HPV-positive and were referred to colposcopy.20 Repeat liquid-based cytology at 6 and 12 months referred to colposcopy 63% of women (using a threshold of a repeat Pap test of ASCUS or greater). Cumulative sensitivity of 2 repeat cytologies for CIN 2,3 was slightly less (88%). Combining a repeat Pap test with an HPV test did not increase sensitivity, but did significantly increase referral to colposcopy.
An HPV test alone at 12 months might be the most efficient test for identifying women with CIN 2,3 after colposcopy.20 Further support for this approach can be found in the substantial body of evidence showing that only persistent HPV progresses to CIN 321 and that testing for high-risk HPV detects most CIN 3.4,17,20
The ASCCP guidelines for women referred for either HPV-positive ASC-US or LSIL and found not to have CIN 2,3 or greater at initial colposcopy recommend either HPV testing at 12 months or repeat cytology at 6 and 12 months (FIGURE 1).11,22
Posttreatment follow-up. The ASCCP treatment guidelines also list HPV testing as an acceptable option for follow-up after treatment with cryosurgery, loop electrosurgical excision procedure, laser, or cold-knife conization,22 since there is substantial evidence that women successfully treated for CIN become HPV-negative, whereas women with persistent disease remain HPV-positive.23-25
A posttreatment HPV test should be performed no sooner than 6 months following the procedure, as it takes time for the patient to return to HPV-negative status. A positive HPV test is an indication for colposcopy. However, the guidelines advise against basing repeat treatment on a positive HPV test alone without documentation of persistent CIN.22
Other options for posttreatment surveillance include either repeat cytology or a combination of Pap testing and colposcopy at 4- to 6-month intervals until at least 3 cytologic results are “negative for squamous intraepithelial lesion or malignancy.”22
Annual cytologic follow-up is recommended thereafter. During that follow-up, any abnormal Pap test (ASC-US or greater) should be referred to colposcopy.
Managing ASC-US in special populations
Management of ASC-US may differ from the general recommendations when the patient is postmenopausal or immunosuppressed. However, there are no differences in the management guidelines during pregnancy.
HPV-negative postmenopausal patients. All 3 management options—immediate colposcopy, repeat cytology, and HPV DNA testing—are acceptable for postmenopausal women with ASC-US.11 However, estrogen deficiency is a common cause of ASC-US and is responsible for increasing rates of HPV-negative ASC-US in this age group despite high sensitivity of HPV testing for CIN 2 and 3.5
- Treatment with vaginal estrogen cream followed by repeat cytology approximately 1 week after completing the regimen is an option for postmenopausal women with ASC-US. This approach also may be helpful for perimenopausal women and for women of any age on progestin-only contraception who have clinical or cytologic evidence of atrophy.
- Women with ASC-US or greater on repeat cytology should be referred for colposcopy, whereas women with normal repeat cytology should have a second Pap test in 4 to 6 months. Repeating the course of vaginal estrogen prior to each Pap test may be helpful when atrophy is likely to persist. After 2 normal repeat Pap tests, the patient can return to routine screening.
Refer all immunosuppressed women for colposcopy. The management of ASC-US in HIV-infected women is particularly problematic because the rates of ASC-US and HPV detection are 2 to 3 times greater than in HIV-negative women. In addition, the risk of CIN 2 and 3 is much higher.26 HPV testing as a triage for ASC-US is not efficient in immunosuppressed women because the majority of ASC-US Pap tests in these women are HPV-positive.
ASCCP recommends colposcopy referral of all immunosuppressed women with ASCUS Pap test, regardless of their CD4 count, HIV viral load, or anti-retroviral therapy.26
Managing ASC-H: First, colposcopy
Clearly, women with ASC-H test results face a greater risk for CIN 2,3 and should be referred for immediate colposcopy.
The ASC-H designation is uncommon, reported in 0.27% to 0.6% of all Pap tests,27,28 or approximately 1 in 10 Pap smears read as ASC.
In ALTS, HPV testing and histology results were compared for women with Pap tests categorized as equivocal LSIL (ASCUSL), ASCUS-H, and high-grade squamous intraepithelial lesion (HSIL) (TABLE 2).29 High-risk HPV DNA was detected in 86% of ASCUS-H liquid-based Pap tests and 69.8% of ASCUS-H conventional smears. CIN 2,3 was found in 40% of liquid-based ASCUS-H smears and in 27.2% of conventional ASCUSH smears. A 3-year retrospective review of ASC-H with follow-up at Johns Hopkins Medical Institutions determined that 49% of patients had no CIN or glandular lesions.28 Of the 51% with CIN, approximately half the lesions were CIN 1 and half were CIN 2,3.
Further management depends on whether CIN is detected (FIGURE 3). If no CIN is found, the ASCCP guidelines recommend that cytology, colposcopy, and histology be reviewed. If there is a change in the diagnosis—eg, if the Pap interpretation is revised to HSIL—the patient should be managed accordingly.11
If there is no change, the patient should be followed with repeat cytology at 6-and 12-month intervals or HPV testing at 12 months. Women having any repeat abnormal Pap test at a threshold of ASC-US or greater or a positive HPV test should undergo repeat colposcopy.
ASC-H is of greater risk than ASC-US, but it is not as risky as HSIL. Therefore, a surgical excision procedure in the absence of documented CIN 2,3 would not normally be indicated.11
FIGURE 3 Management of atypical squamous cells–cannot exclude high–grade squamous intraepithelial lesions (ASC-H)
HPV test as triage option would mean retooling the system
Cytologic management systems have traditionally involved follow-up by repeat cytology, colposcopy, and, when necessary, treatment. Adding another triage option—HPV testing—requires that this system be retooled.
The labs. This is not difficult when the laboratory interpreting the liquid-based Pap test is the same lab that performs the “reflex” HPV test, as this allows the ASC-US Pap test to be reported as HPV-negative or HPV-positive. However, if the HPV test must be performed in a separate reference laboratory, the results of the Pap and HPV tests will arrive separately, and the clinician must collate the 2 reports before relaying the result to the patient.
The patients. Remember than an HPV test is a test for a sexually transmitted disease. (So is the Pap test, although it has not traditionally been considered as such.) For that reason, I give all patients a written explanation of the rationale behind testing ASC-US Pap tests for HPV. This explanation includes 2 check-off options at the bottom of the sheet where patients can indicate whether they would prefer HPV testing or one of the other follow-up options.
Most patients elect the HPV option. Our Pap test requisitions also have a check-off portion that allows us to notify the lab of patients who wants an HPV test if the Pap is interpreted as ASC-US.
The office staff. Whenever a new test or procedure is introduced, it is of primary importance that the office staff responsible for completing critical information on the requisition form is adequately trained. This involves knowing when and how to order the test and how to complete insurance information and clinical history on the Pap requisition—including the correct International Classification of Diseases, Ninth Revision code—to ensure that the HPV test is covered by the patient’s insurer.
Clinicians must understand the usually benign nature of HPV infection. Reporting a positive HPV test in a manner that is not unduly concerning requires reassuring and nonjudgmental communication of the results based on a broad understanding of the usually low-risk natural history of the virus, yet fosters responsible follow-up.
Why all HPV-positive ASC-US requires diligent follow-up
The recently released ASCCP guidelines recognize HPV testing as an option in the management of ASC results, including:
- initial management of ASC-US,
- postcolposcopy management of ASC-H or HPV-positive ASC Pap tests found to be normal or to have CIN 1, and
- posttreatment follow-up.
For each indication, the HPV test identifies women most likely to have CIN (HPV-positive) and those likely to have benign processes not related to HPV (HPV-negative).
New longitudinal data verify that women with HPV-positive ASC-US continue at risk for detection of CIN 2,3 (about 12% overall), whether the original colposcopic finding was normal or CIN 1.8 Therefore, they need continued diligent follow-up.
Dr. Cox serves on the Speaker’s Bureaus of Cytyc Corporation and Digene Corporation.
- Management by immediate colposcopy, repeat cytology, or HPV testing is acceptable for ASC-US, but testing for HPV is preferred when the Pap test is liquid-based.
- The sensitivity of HPV triage for high-grade CIN is essentially equivalent to colposcopy, and reduces the need for colposcopy by half.
- HPV testing is a good option for follow-up after treatment with cryosurgery, loop electrosurgical excision procedure, laser, or cold-knife conization.
What’s the best management strategy for the roughly 2 to 3 million women each yearUpdate on Cervical Disease, for commentary by Thomas C. Wright, Jr, MD, Department of Pathology, College of Physicians and Surgeons of Columbia University.The difference is even more pronounced when the cumulative 2-year detection rate for CIN 2,3 is added in for women referred for HPV-positive ASCUS but not found to have CIN 2,3 at initial colposcopy. That rate rises from 20.1% at initial colposcopy to 26.9% at 2 years.8 Although many experts consider even HPV-positive ASCUS of minimal risk, few would consider a risk of high-grade disease exceeding 1 in 4 to be minimal. In fact, 39% of the total CIN 2,3 cases reported from a routine screening population were detected following triage of ASCUS, and fully 69% were from all equivocal and low-grade Pap diagnoses.9
TABLE 1
Risk of cervical intraepithelial neoplasia grade 2 or greater at initial colposcopy
| ASCUS | ||||
|---|---|---|---|---|
| STUDY | HPV TEST | HPV–POSITIVE | HPV–NEGATIVE | TOTAL RISK FOR ALL ASCUS |
| Cox6 | Hybrid capture 1 (expanded first–generation test) | 17% (14/81) | 0.74% (1/136) | 6.9% (15/217) |
| Manos7 | Hybrid capture 2 | 15% (45/300) | 1.2% (6/498) | 6.4% (51/798) |
| Solomon4 (ALTS) | Hybrid capture 2 | 18% (195/1,087) | 1.1% (13/1,175) | 9.2% (208/2,262) |
| ALTS = ASCUS/LSIL Triage Study; ASCUS = atypical squamous cells of undetermined significance; HPV = human papillomavirus | ||||
Bethesda 3 redefines ASCUS
The third Bethesda System workshop took place in May 2001 with the aim of evaluating and updating earlier terminology.10 It began by eliminating the words “of undetermined significance” from the overall ASCUS category, which is now called simply “atypical squamous cells,” or ASC. Most subcategories of the former ASCUS were eliminated as well. (Note: Within this article, the acronyms ASCUS and ASC-US are both used to describe atypical squamous cells of undetermined significance. The latter acronym reflects usage and guidelines developed after the third Bethesda workshop.)
Now the ASC classification is broken down into 2 distinct groups:
Atypical squamous cells–undetermined significance, or ASC-US. This new subcategory includes cells previously termed “favor reactive” but not relegated by the pathologist to normal, as well as cells previously in the “unqualified” and “favor HPV” or “favor low-grade squamous intraepithelial lesion (LSIL)” subcategories.
Atypical squamous cells–cannot rule out high-grade squamous intraepithelial lesions, or ASC-H. This category includes atypical cells difficult to distinguish from high-grade cells but not definitive for that classification. Women with such Pap tests are at greater risk for high-risk HPV and histologic CIN 2,3 (TABLE 2).
Evidence-based guidelines reflect Bethesda 3 changes. By the time of Bethesda 3, extensive new data on the management of abnormal cytology was available, including but not limited to data from ALTS, making it possible to create evidence-based guidelines on management of abnormal cervical cytology and CIN. These guidelines were developed in 2001 at a consensus conference hosted by the American Society for Colposcopy and Cervical Pathology (ASCCP),11 with input from 29 professional organizations, federal agencies, and national and international health organizations.
The entire set recommendations for all types of abnormal Pap tests were published in the April 24, 2002 issue of the Journal of the American Medical Association, and management recommendations for histologically proven CIN were published in the July 2003 American Journal of Obstetrics and Gynecology and the July 2003 Journal of Lower Genital Tract Disease. The management algorithms for both cytology and histology can be downloaded from http://ASCCP.org.
TABLE 2
Comparison of risk for high-risk HPV and CIN grade 2,3, by Pap results
| HISTOLOGY | |||||
|---|---|---|---|---|---|
| PAP TEST | HIGH-RISK HPV | CIN 2 OR GREATER | CIN 3 | ||
| ASC-US | 63% | 12% | 5% | ||
| ASC-H | 86% | 40% | 24% | ||
| HSIL | 99% | 59% | 38% | ||
| Data from Sherman et al29 | |||||
| ASC-US = atypical squamous cells–undetermined significance; | |||||
| ASC-H = Atypical squamous cells–cannot rule out high-grade squamous intraepithelial lesion; CIN = cervical intraepithelial neoplasia; HPV = human papillomavirus; HSIL = high-grade squamous intraepithelial lesion | |||||
All 3 triage options safe, effective
An evidence-based review found all 3 options safe and effective.11 Therefore, management by immediate colposcopy, repeat cytology, or HPV testing is acceptable for ASCUS, but testing for HPV is preferred when the Pap test is liquid-based (FIGURE 1).
Liquid-based cytology (ThinPrep; Cytyc, Boxborough, Mass and SurePath, Raleigh-Durham, NC) has several advantages. For example, residual cells in the fluid can be tested for HPV, eliminating a return visit.
Immediate colposcopy: Low predictive value, high anxiety and expense. Proponents of immediate colposcopy for all women with ASC-US argue that this would theoretically detect all CIN 2,3 and cancer. However, the positive predictive value of this approach will always be extremely low due to the low rate (6.4% to 11.9%) of CIN 2,3 in women with ASCUS.4,6,7 What’s more, the cost and anxiety generated by immediate colposcopy are high.12
2 repeat cytologies: Sensitivity, cost issues. This approach requires at least 2 repeat, optimized (liquid-based) Pap tests to equal the sensitivity of a single HPV test. This, compounded with the high rate of repeat abnormal cytology requiring colposcopic evaluation, means repeat cytology is unlikely to be cost-competitive with HPV testing.4,13
- Cervical cytology as a triage option. Cytology has been a good screening test, but its comparatively low sensitivity (51% to 83%) and poor reproducibility reduces its value as a triage test.13-17 For example, in ALTS, of 1,473 repeat Paps originally read as ASCUS by good clinical pathologists, only 633 were reread as ASCUS when 2-of-3 agreement was obtained in a blinded review by an expert panel of pathologists.16 In other words, 840 (57%) were reread as something other than ASCUS. Most were downgraded to normal.
- The sensitivity of the HPV test in detecting CIN 2,3 was 92.4%. This rate was matched only by 2 repeat Pap tests, provided the threshold for referral to colposcopy was ASCUS or greater.17 At this threshold, 95% of the CIN 2,3 was detected with repeat Pap testing, but only after an average of 8 to 12 months. This contrasts with the immediate reassurance provided by the initial HPV test.
- ALTS did not evaluate repeat conventional Pap smears. Nor do the guidelines differentiate between conventional and liquid-based methods in the number of follow-up Pap tests required for reassurance, despite consensus that the sensitivity of liquid-based cytology is better than that of the conventional “dry slide.”
- Any woman with a repeat Pap result of ASC-US or greater should be referred to colposcopy. Referral at a threshold of LSIL or greater would result in far fewer colposcopies, but has not been shown to be sufficiently sensitive for CIN 2,3.17
HPV testing identifies clear risk. Any objective test that initially indicates which women with ASC-US are at risk for CIN 2,3 and which are not—either now or in the future—should confer a major advantage.
HPV-positive women are clearly at risk, justifying the anxiety and cost of colposcopic referral, while HPV-negative women may be reassured (FIGURE 2). Also, ALTS data showed HPV triage is essentially equivalent to immediate colposcopy in sensitivity for high-grade CIN, while halving colposcopic referrals.17,18
Because low-risk HPV types do not cause CIN 3 or cancer, the HPV test should document only high-risk types.11 The only HPV test approved by the US Food and Drug Administration (Hybrid Capture 2, Digene, Gaithersburg, Md) includes both low- and high-risk HPV panels. For cost savings, the laboratory can be asked to use only the high-risk panel. All positive high-risk HPV cases should be referred to colposcopy.
FIGURE 1 3 triage options for management of ASC-US
FIGURE 2 Management of atypical squamous cells–cannot exclude high–grade squamous intraepithelial lesions (ASC-H)
Some high-grade lesions are still overlooked
A single HPV test or 2 repeat liquid-based Pap tests with a colposcopy-referral threshold of any findings of ASC-US or greater have similar sensitivity for CIN 2,3.17,18
The guidelines state that women who undergo immediate colposcopy with negative results or who have a negative initial HPV test should undergo a follow-up Pap test in 12 months. Note that the guidelines do not state that these women can return directly to routine screening. The reason: In some settings, “routine” screening is at 2- or 3-year intervals, and some risk still exists—albeit minimal—for missed CIN 2,3.
For example, 1 of 83 cases of CIN 2,3 were missed by HPV testing in the study by Manos et al.7 In ALTS, that number was 1 in 90,4 and in the study by Cox et al6 it was 1 in 136. Further, colposcopy did not initially detect 25% of the cumulative high-grade lesions detected over 2 years of follow-up in ALTS.17
In contrast, the recommendation for women with 2 repeat normal Pap tests is to return to “routine screening.” This inexplicably departs from the 12-month repeat Pap testing urged for women with negative results on the other 2 triage options, despite a similar risk of missed high-grade disease.
- In my opinion, all 3 scenarios should be managed by repeat Pap testing in 12 months.
Reducing referrals to colposcopy
If all women returned as directed for repeat cytology, more of them would be referred to colposcopy by repeat abnormal Pap tests at the ASC-US threshold than by testing positive for high-risk HPV types. In ALTS, 53% tested positive for high-risk HPV and were referred to colposcopy, compared with 67% who had an abnormal Pap test on the first or second repeat (these women also had 1 or 2 more office visits prior to referral to colposcopy.).
No difference for conventional smears. All the advantages of HPV testing in the triage of women with ASC-US persist when the initial referral Pap test is a conventional smear. The only exception is that HPV testing would require the patient to return for a repeat office visit. An alternative would be co-collecting an HPV-test sample at the time of the primary screening Pap test.
One major health-maintenance organization collects a separate sample from all women when the routine conventional Pap test is obtained using a standard Hybrid Capture 2 HPV test kit. The HPV-testing samples are then held until the results of the Pap smear are reported. For women reported to have ASCUS, the samples are sent to the lab for HPV testing; the remaining samples (approximately 95% in most practices) are discarded as medical waste. The cost of each discarded kit is approximately $1. Modeling has found this approach to be cost-effective.19
Postcolposcopy management
Many clinicians are concerned that women referred for the evaluation of HPV-positive ASC-US and found not to have CIN or other manifestations of HPV at colposcopy have a “false-positive” HPV test. However, although there are occasional HPV tests that misclassify a low-risk HPV type as high-risk, actual false-positive tests are very rare.
The 2-year ALTS longitudinal data provide the best information on what to expect when a woman with HPV-positive ASC-US or LSIL is found at colposcopy to have no CIN or to have only CIN 1 that is subsequently managed expectantly.8
The cumulative risk of CIN 2,3 over the 2 years was nearly equivalent for women referred initially for LSIL (27.6%) and for women referred for HPV-positive ASCUS (26.7%), further verifying that management should be similar. Two thirds of the CIN 2,3 was detected at initial colposcopy, and the remaining one third during the postcolposcopy 2-year follow-up.
The risk for subsequent detection of high-grade CIN was nearly identical for all women initially found not to have CIN 2,3 regardless of whether CIN 1 was detected at initial colposcopy, whether the colposcopy was initially completely normal, or whether there were changes that were biopsied and found not to have CIN (risk for CIN 2,3 was 13%, 11.3%, and 11.7% respectively).
Hence, all women referred for evaluation of HPV-positive ASC-US or LSIL and not treated for CIN 2,3 require similar diligent follow-up.
A single HPV test at 12 months detected 92% of all CIN 2,3 found over the 24-month follow-up; 55% tested HPV-positive and were referred to colposcopy.20 Repeat liquid-based cytology at 6 and 12 months referred to colposcopy 63% of women (using a threshold of a repeat Pap test of ASCUS or greater). Cumulative sensitivity of 2 repeat cytologies for CIN 2,3 was slightly less (88%). Combining a repeat Pap test with an HPV test did not increase sensitivity, but did significantly increase referral to colposcopy.
An HPV test alone at 12 months might be the most efficient test for identifying women with CIN 2,3 after colposcopy.20 Further support for this approach can be found in the substantial body of evidence showing that only persistent HPV progresses to CIN 321 and that testing for high-risk HPV detects most CIN 3.4,17,20
The ASCCP guidelines for women referred for either HPV-positive ASC-US or LSIL and found not to have CIN 2,3 or greater at initial colposcopy recommend either HPV testing at 12 months or repeat cytology at 6 and 12 months (FIGURE 1).11,22
Posttreatment follow-up. The ASCCP treatment guidelines also list HPV testing as an acceptable option for follow-up after treatment with cryosurgery, loop electrosurgical excision procedure, laser, or cold-knife conization,22 since there is substantial evidence that women successfully treated for CIN become HPV-negative, whereas women with persistent disease remain HPV-positive.23-25
A posttreatment HPV test should be performed no sooner than 6 months following the procedure, as it takes time for the patient to return to HPV-negative status. A positive HPV test is an indication for colposcopy. However, the guidelines advise against basing repeat treatment on a positive HPV test alone without documentation of persistent CIN.22
Other options for posttreatment surveillance include either repeat cytology or a combination of Pap testing and colposcopy at 4- to 6-month intervals until at least 3 cytologic results are “negative for squamous intraepithelial lesion or malignancy.”22
Annual cytologic follow-up is recommended thereafter. During that follow-up, any abnormal Pap test (ASC-US or greater) should be referred to colposcopy.
Managing ASC-US in special populations
Management of ASC-US may differ from the general recommendations when the patient is postmenopausal or immunosuppressed. However, there are no differences in the management guidelines during pregnancy.
HPV-negative postmenopausal patients. All 3 management options—immediate colposcopy, repeat cytology, and HPV DNA testing—are acceptable for postmenopausal women with ASC-US.11 However, estrogen deficiency is a common cause of ASC-US and is responsible for increasing rates of HPV-negative ASC-US in this age group despite high sensitivity of HPV testing for CIN 2 and 3.5
- Treatment with vaginal estrogen cream followed by repeat cytology approximately 1 week after completing the regimen is an option for postmenopausal women with ASC-US. This approach also may be helpful for perimenopausal women and for women of any age on progestin-only contraception who have clinical or cytologic evidence of atrophy.
- Women with ASC-US or greater on repeat cytology should be referred for colposcopy, whereas women with normal repeat cytology should have a second Pap test in 4 to 6 months. Repeating the course of vaginal estrogen prior to each Pap test may be helpful when atrophy is likely to persist. After 2 normal repeat Pap tests, the patient can return to routine screening.
Refer all immunosuppressed women for colposcopy. The management of ASC-US in HIV-infected women is particularly problematic because the rates of ASC-US and HPV detection are 2 to 3 times greater than in HIV-negative women. In addition, the risk of CIN 2 and 3 is much higher.26 HPV testing as a triage for ASC-US is not efficient in immunosuppressed women because the majority of ASC-US Pap tests in these women are HPV-positive.
ASCCP recommends colposcopy referral of all immunosuppressed women with ASCUS Pap test, regardless of their CD4 count, HIV viral load, or anti-retroviral therapy.26
Managing ASC-H: First, colposcopy
Clearly, women with ASC-H test results face a greater risk for CIN 2,3 and should be referred for immediate colposcopy.
The ASC-H designation is uncommon, reported in 0.27% to 0.6% of all Pap tests,27,28 or approximately 1 in 10 Pap smears read as ASC.
In ALTS, HPV testing and histology results were compared for women with Pap tests categorized as equivocal LSIL (ASCUSL), ASCUS-H, and high-grade squamous intraepithelial lesion (HSIL) (TABLE 2).29 High-risk HPV DNA was detected in 86% of ASCUS-H liquid-based Pap tests and 69.8% of ASCUS-H conventional smears. CIN 2,3 was found in 40% of liquid-based ASCUS-H smears and in 27.2% of conventional ASCUSH smears. A 3-year retrospective review of ASC-H with follow-up at Johns Hopkins Medical Institutions determined that 49% of patients had no CIN or glandular lesions.28 Of the 51% with CIN, approximately half the lesions were CIN 1 and half were CIN 2,3.
Further management depends on whether CIN is detected (FIGURE 3). If no CIN is found, the ASCCP guidelines recommend that cytology, colposcopy, and histology be reviewed. If there is a change in the diagnosis—eg, if the Pap interpretation is revised to HSIL—the patient should be managed accordingly.11
If there is no change, the patient should be followed with repeat cytology at 6-and 12-month intervals or HPV testing at 12 months. Women having any repeat abnormal Pap test at a threshold of ASC-US or greater or a positive HPV test should undergo repeat colposcopy.
ASC-H is of greater risk than ASC-US, but it is not as risky as HSIL. Therefore, a surgical excision procedure in the absence of documented CIN 2,3 would not normally be indicated.11
FIGURE 3 Management of atypical squamous cells–cannot exclude high–grade squamous intraepithelial lesions (ASC-H)
HPV test as triage option would mean retooling the system
Cytologic management systems have traditionally involved follow-up by repeat cytology, colposcopy, and, when necessary, treatment. Adding another triage option—HPV testing—requires that this system be retooled.
The labs. This is not difficult when the laboratory interpreting the liquid-based Pap test is the same lab that performs the “reflex” HPV test, as this allows the ASC-US Pap test to be reported as HPV-negative or HPV-positive. However, if the HPV test must be performed in a separate reference laboratory, the results of the Pap and HPV tests will arrive separately, and the clinician must collate the 2 reports before relaying the result to the patient.
The patients. Remember than an HPV test is a test for a sexually transmitted disease. (So is the Pap test, although it has not traditionally been considered as such.) For that reason, I give all patients a written explanation of the rationale behind testing ASC-US Pap tests for HPV. This explanation includes 2 check-off options at the bottom of the sheet where patients can indicate whether they would prefer HPV testing or one of the other follow-up options.
Most patients elect the HPV option. Our Pap test requisitions also have a check-off portion that allows us to notify the lab of patients who wants an HPV test if the Pap is interpreted as ASC-US.
The office staff. Whenever a new test or procedure is introduced, it is of primary importance that the office staff responsible for completing critical information on the requisition form is adequately trained. This involves knowing when and how to order the test and how to complete insurance information and clinical history on the Pap requisition—including the correct International Classification of Diseases, Ninth Revision code—to ensure that the HPV test is covered by the patient’s insurer.
Clinicians must understand the usually benign nature of HPV infection. Reporting a positive HPV test in a manner that is not unduly concerning requires reassuring and nonjudgmental communication of the results based on a broad understanding of the usually low-risk natural history of the virus, yet fosters responsible follow-up.
Why all HPV-positive ASC-US requires diligent follow-up
The recently released ASCCP guidelines recognize HPV testing as an option in the management of ASC results, including:
- initial management of ASC-US,
- postcolposcopy management of ASC-H or HPV-positive ASC Pap tests found to be normal or to have CIN 1, and
- posttreatment follow-up.
For each indication, the HPV test identifies women most likely to have CIN (HPV-positive) and those likely to have benign processes not related to HPV (HPV-negative).
New longitudinal data verify that women with HPV-positive ASC-US continue at risk for detection of CIN 2,3 (about 12% overall), whether the original colposcopic finding was normal or CIN 1.8 Therefore, they need continued diligent follow-up.
Dr. Cox serves on the Speaker’s Bureaus of Cytyc Corporation and Digene Corporation.
1. Kurman RJ, Henson DE, Berbst AL, Noller KL, Schiffman MH. Interim guidelines for the management of abnormal cervical cytology. JAMA. 1994;271:1866-1869.
2. Ferenczy A. Viral testing for genital human papillomavirus infections: recent progress and clinical potentials. Int J Gynecol Cancer. 1995;5:321-328.
3. Schiffman M, Adrianza ME. ASCUS-LSIL Triage Study. Design, methods and characteristics of trial participants. Acta Cytol. 2000;44:726-742.
4. Solomon D, Schiffman M, Tarone R, et al. Comparison of three management strategies for patients with atypical squamous cells of undetermined significance: baseline results from a randomized trial. J Natl Cancer Inst. 2001;93:293-299.
5. Sherman ME, Schiffman M, Cox JT, et al. Effects of age and human papilloma viral load on colposcopy triage: data from the randomized Atypical Squamous Cells of Undetermined Significance/Low-Grade Squamous Intraepithelial Lesion Triage Study (ALTS). J Natl Cancer Inst. 2002;94(2):102-107.
6. Cox JT, Lorincz AT, Schiffman MH, Sherman ME, Cullen A, Kurman RJ. Human papillomavirus testing by hybrid capture appears to be useful in triaging women with a cytologic diagnosis of atypical squamous cells of undetermined significance. Am J Obstet Gynecol. 1995;172:946-954.
7. Manos MM, Kinney WK, Hurley LB, et al. Identifying women with cervical neoplasia: using human papillomavirus DNA testing for equivocal Papanicolaou results. JAMA. 1999;281:1605-1610.
8. Cox JT, Schiffman M, Solomon D, et al. Prospective follow-up suggests similar risk of subsequent cervical intraepithelial neoplasia grade 2 or 3 among women with cervical intraepithelial neoplasia grade 1 or negative colposcopy and directed biopsy. Am J Obstet Gynecol. 2003;188:1406-1412.
9. Kinney WK, Manos MM, Hurley LB, Ransley JE. Where’s the high-grade cervical neoplasia? The importance of the minimally abnormal Papanicolaou diagnosis. Obstet Gynecol. 1998;91:973-976.
10. Solomon D, Davey D, Kurman R, et al. The 2001 Bethesda System: terminology for reporting results of cervical cytology. JAMA. 2002;287:2114-2119.
11. Wright TC, Jr, Cox JT, Massad LS, et al. 2001 Consensus Guidelines for the management of women with cervical cytological abnormalities. JAMA. 2002;287:2120-2129.
12. Jones MH, Singer A, Jenkins D. The mildly abnormal cervical smear: patient awareness and choice of management. J Royal Soc Med. 1996;89:257.-
13. Wright TC, Lorincz AT, Ferris DG, et al. Reflex human papillomavirus deoxyribonucleic acid testing in women with abnormal Pap smears. Am J Obstet Gynecol. 1998;178:926-966.
14. Fahey MT, Irwig L, Macaskill P. Meta-analysis of Pap test accuracy. Am J Epidemiol. 1995;141:680-689.
15. Agency for Health Care Policy and Research. Evidence Report/Technology Assessment #5: Evaluation of Cervical Cytology. Rockville, Md: AHCPR; January 1999.
16. Stoler MH, Schiffman M. Atypical Squamous Cells of Undetermined Significance—Low-grade Squamous Intraepithelial Lesion Triage Study (ALTS) Group. Interobserver reproducibility of cervical cytologic and histologic interpretations: realistic estimates from the ASCUS-LSIL Triage Study. JAMA. 2001;285:1500-1505.
17. ASCUS-LSIL Triage Study (ALTS) Group. Results of a randomized trial on the management of cytology interpretations of atypical squamous cells of undetermined significance. Am J Obstet Gynecol. 2003;188:1383-1392.
18. Schiffman M, Solomon D. Findings to date from the ASCUS-LSIL Triage Study (ALTS). Arch Pathol Lab Med. 2003;127:946-949.
19. Kim JJ, Wright TC, Goldie SJ. Cost-effectiveness of alternative triage strategies for atypical squamous cells of undetermined significance. JAMA. 2002;287:2382-2390.
20. Guido R, Schiffman M, Solomon D, et al. Postcolposcopy management strategies for women referred with low-grade squamous intraepithelial lesions or human papillomavirus DNA-positive atypical squamous cells of undetermined significance: a two-year prospective study. Am J Obstet Gynecol. 2003;188:1401-1405.
21. Nobbenhuis M, Walboomers JM, Helmerhorst TI, Rozendaal L. Relation of human papillomavirus status to cervical lesions and consequences for cervical-cancer screening: a prospective study. Lancet. 1999;354:20.-
22. Wright TC, Jr, Cox JT, Massad LS, et al. 2001 consensus guidelines for the management of women with cervical intraepithelial neoplasia. Am J Obstet Gynecol. 2003;189:295-304.
23. Paraskevaidis E, Koliopoulos G, Alamanos Y, Malamou-Mitsi V, Lolis ED, Kitchener HC. Human papillomavirus testing and the outcome of treatment for cervical intraepithelial neoplasia. Obstet Gynecol. 2001;98:833-836.
24. Nobbenhuis MA, Meijer CJ, van den Brule AJ, et al. Addition of high-risk HPV testing improves the current guidelines on follow-up after treatment for cervical intraepithelial neoplasia. Br J Cancer. 2001;84:796-801.
25. Jain S, Tseng CJ, Horng SG, Soong YK, Pao CC. Negative predictive value of human papillomavirus test following conization of the cervix uteri. Gynecol Oncol. 2001;82:177-180.
26. Massad LS, Ahdieh L, Benning L, et al. Evolution of cervical abnormalities among women with HIV-1: evidence from surveillance cytology in the women’s interagency HIV study. J Acquir Immune Defic Syndr. 2001;27:432-442.
27. Selvaggi SM. Reporting of atypical squamous cells, cannot exclude a high-grade squamous intraepithelial lesion (ASC-H) on cervical samples: is it significant? Diagn Cytopathol. 2003;29:38-41.
28. Alli PM, Ali SZ. Atypical squamous cells of undetermined significance—rule out high-grade squamous intraepithelial lesion: cytopathologic characteristics and clinical correlates. Diagn Cytopathol. 2003;28:308-312.
29. Sherman ME, Solomon D, Schiffman M, et al. A comparison of equivocal LSIL and equivocal HSIL cervical cytology in the ASCUS LSIL Triage Study. Am J Clin Pathol. 2001;116:386-394.
1. Kurman RJ, Henson DE, Berbst AL, Noller KL, Schiffman MH. Interim guidelines for the management of abnormal cervical cytology. JAMA. 1994;271:1866-1869.
2. Ferenczy A. Viral testing for genital human papillomavirus infections: recent progress and clinical potentials. Int J Gynecol Cancer. 1995;5:321-328.
3. Schiffman M, Adrianza ME. ASCUS-LSIL Triage Study. Design, methods and characteristics of trial participants. Acta Cytol. 2000;44:726-742.
4. Solomon D, Schiffman M, Tarone R, et al. Comparison of three management strategies for patients with atypical squamous cells of undetermined significance: baseline results from a randomized trial. J Natl Cancer Inst. 2001;93:293-299.
5. Sherman ME, Schiffman M, Cox JT, et al. Effects of age and human papilloma viral load on colposcopy triage: data from the randomized Atypical Squamous Cells of Undetermined Significance/Low-Grade Squamous Intraepithelial Lesion Triage Study (ALTS). J Natl Cancer Inst. 2002;94(2):102-107.
6. Cox JT, Lorincz AT, Schiffman MH, Sherman ME, Cullen A, Kurman RJ. Human papillomavirus testing by hybrid capture appears to be useful in triaging women with a cytologic diagnosis of atypical squamous cells of undetermined significance. Am J Obstet Gynecol. 1995;172:946-954.
7. Manos MM, Kinney WK, Hurley LB, et al. Identifying women with cervical neoplasia: using human papillomavirus DNA testing for equivocal Papanicolaou results. JAMA. 1999;281:1605-1610.
8. Cox JT, Schiffman M, Solomon D, et al. Prospective follow-up suggests similar risk of subsequent cervical intraepithelial neoplasia grade 2 or 3 among women with cervical intraepithelial neoplasia grade 1 or negative colposcopy and directed biopsy. Am J Obstet Gynecol. 2003;188:1406-1412.
9. Kinney WK, Manos MM, Hurley LB, Ransley JE. Where’s the high-grade cervical neoplasia? The importance of the minimally abnormal Papanicolaou diagnosis. Obstet Gynecol. 1998;91:973-976.
10. Solomon D, Davey D, Kurman R, et al. The 2001 Bethesda System: terminology for reporting results of cervical cytology. JAMA. 2002;287:2114-2119.
11. Wright TC, Jr, Cox JT, Massad LS, et al. 2001 Consensus Guidelines for the management of women with cervical cytological abnormalities. JAMA. 2002;287:2120-2129.
12. Jones MH, Singer A, Jenkins D. The mildly abnormal cervical smear: patient awareness and choice of management. J Royal Soc Med. 1996;89:257.-
13. Wright TC, Lorincz AT, Ferris DG, et al. Reflex human papillomavirus deoxyribonucleic acid testing in women with abnormal Pap smears. Am J Obstet Gynecol. 1998;178:926-966.
14. Fahey MT, Irwig L, Macaskill P. Meta-analysis of Pap test accuracy. Am J Epidemiol. 1995;141:680-689.
15. Agency for Health Care Policy and Research. Evidence Report/Technology Assessment #5: Evaluation of Cervical Cytology. Rockville, Md: AHCPR; January 1999.
16. Stoler MH, Schiffman M. Atypical Squamous Cells of Undetermined Significance—Low-grade Squamous Intraepithelial Lesion Triage Study (ALTS) Group. Interobserver reproducibility of cervical cytologic and histologic interpretations: realistic estimates from the ASCUS-LSIL Triage Study. JAMA. 2001;285:1500-1505.
17. ASCUS-LSIL Triage Study (ALTS) Group. Results of a randomized trial on the management of cytology interpretations of atypical squamous cells of undetermined significance. Am J Obstet Gynecol. 2003;188:1383-1392.
18. Schiffman M, Solomon D. Findings to date from the ASCUS-LSIL Triage Study (ALTS). Arch Pathol Lab Med. 2003;127:946-949.
19. Kim JJ, Wright TC, Goldie SJ. Cost-effectiveness of alternative triage strategies for atypical squamous cells of undetermined significance. JAMA. 2002;287:2382-2390.
20. Guido R, Schiffman M, Solomon D, et al. Postcolposcopy management strategies for women referred with low-grade squamous intraepithelial lesions or human papillomavirus DNA-positive atypical squamous cells of undetermined significance: a two-year prospective study. Am J Obstet Gynecol. 2003;188:1401-1405.
21. Nobbenhuis M, Walboomers JM, Helmerhorst TI, Rozendaal L. Relation of human papillomavirus status to cervical lesions and consequences for cervical-cancer screening: a prospective study. Lancet. 1999;354:20.-
22. Wright TC, Jr, Cox JT, Massad LS, et al. 2001 consensus guidelines for the management of women with cervical intraepithelial neoplasia. Am J Obstet Gynecol. 2003;189:295-304.
23. Paraskevaidis E, Koliopoulos G, Alamanos Y, Malamou-Mitsi V, Lolis ED, Kitchener HC. Human papillomavirus testing and the outcome of treatment for cervical intraepithelial neoplasia. Obstet Gynecol. 2001;98:833-836.
24. Nobbenhuis MA, Meijer CJ, van den Brule AJ, et al. Addition of high-risk HPV testing improves the current guidelines on follow-up after treatment for cervical intraepithelial neoplasia. Br J Cancer. 2001;84:796-801.
25. Jain S, Tseng CJ, Horng SG, Soong YK, Pao CC. Negative predictive value of human papillomavirus test following conization of the cervix uteri. Gynecol Oncol. 2001;82:177-180.
26. Massad LS, Ahdieh L, Benning L, et al. Evolution of cervical abnormalities among women with HIV-1: evidence from surveillance cytology in the women’s interagency HIV study. J Acquir Immune Defic Syndr. 2001;27:432-442.
27. Selvaggi SM. Reporting of atypical squamous cells, cannot exclude a high-grade squamous intraepithelial lesion (ASC-H) on cervical samples: is it significant? Diagn Cytopathol. 2003;29:38-41.
28. Alli PM, Ali SZ. Atypical squamous cells of undetermined significance—rule out high-grade squamous intraepithelial lesion: cytopathologic characteristics and clinical correlates. Diagn Cytopathol. 2003;28:308-312.
29. Sherman ME, Solomon D, Schiffman M, et al. A comparison of equivocal LSIL and equivocal HSIL cervical cytology in the ASCUS LSIL Triage Study. Am J Clin Pathol. 2001;116:386-394.
What VA Providers Really Think About Clinical Practice Guidelines
Cryopreservation impact will rival the Pill Breakthroughs in genetic diagnosis Does ART insurance reduce multiple births?
Advancements during the year 2003 propelled infertility therapy further than ever along its 20-year evolution toward extensive use of assisted reproductive technologies (ART) and the introduction of diagnostic genetics. Refreshingly, social and ethical implications were viewed with greater recognition that we must deal effectively with the repercussions of technical progress.
Basic evaluation still key; early referral appropriate. For the practicing Ob/Gyn already confused by the bewildering and growing array of options presented to patients, these changes do not mean that a basic infertility evaluation and primary therapy are inappropriate. However, early referral to a trusted, experienced infertility specialist before invasive diagnostic studies such as laparoscopy or aggressive therapies is now most appropriate.
This Update reviews trends in reproductive medicine that herald both clinical and social transformations.
Oocyte cryopreservation: Ready for prime time
Porcu E. Human oocyte cryopreservation: from basic research to clinical application. Tap Pharmaceuticals Endowed Lecture presented at: American Society for Reproductive Medicine 59th Annual Meeting; October 15, 2003; San Antonio, Tex.
Growing concern over the past decade about age-related infertility due to progressive loss of oocyte number and quality has prompted rapidly expanding use of donated oocytes from younger women. Due to technical difficulties in cryopreserving oocytes, it has not been clinically feasible to save eggs from young women for later use or for donation to other women from “egg banks,” as has been done with sperm for many years.
In a presentation on the research program in oocyte freezing at the University of Bologna, Italy, Dr. Eleonora Porcu elaborated on the last decade’s progress. She described progressively improving clinical pregnancy rates from cryopreserved eggs, and commented that clinical experience to date is sufficient to begin offering this option to women generally.
A growing number of centers are offering oocyte cryopreservation, although outcome data for specific centers are limited and no national aggregate data are yet available.
Cryopreservation will beat the biological clock. Although immediate clinical use of this technology will be directed to young women at risk for loss of ovarian function due to cancer therapy or oophorectomy, the social implications could be comparable to the introduction of effective contraception in the 1960s.
Just as oral contraceptives allowed women to delay childbearing and yet be sexually active, oocyte cryopreservation may allow women to delay childbearing without fear that the “biological clock” will strike midnight before they have had children.
More older mothers, more concerns. While demand for this service is undetermined, and many biological and financial issues remain to be delineated, this process will continue the trend to later childbearing and older mothers, with all their attendant social and medical concerns.
Prepare to provide referral services. It will be increasingly important for physicians to be aware of the availability of such services and to have a ready source of referral for their patients interested in pursuing oocyte cryopreservation.
On the horizon: Unlimited supply of gametes. In a scientifically related but much less clinically applicable presentation at the same meeting, Susan Rothmann, Roger Gosden, and Pasquale Patrizio described the status of research in germ-line stem cells and reviewed the evidence for oogonial stem cells in human ovaries and the efforts to isolate and cryopreserve such stem cells.
Although much work needs to be done before spermatogonial and oogonial stem cells can be isolated and stored from patients, this work holds the promise that storage of germ-line stem cells may provide an unlimited future supply of gametes for use in later reproduction.
Related Reading
Porcu E. Oocyte freezing. Semin Reprod Med. 2001;19:221-230.
Advances in genetic diagnosis will enable transfer of ‘normal’ embryos only
Wilton L, Voullaire L, Sargeant P, Williamson R, McBain J. Preimplantation aneuploidy screening using comparative genomic hybridization or fluorescence in situ hybridization of embryos from patients with recurrent implantation failure. Fertil Steril. 2003;80:860-868.
One of the hottest topics today in reproductive medicine is the increasing availability of preimplantation diagnosis to determine if embryos are genetically normal. This Australian study compared 2 methods of genetic evaluation for their sensitivity in identifying abnormalities and in predicting subsequent implantation and pregnancy rates. The investigators concluded that comparative genomic hybridization was more effective than fluorescence in situ hybridization in identifying chromosomally normal embryos.
Improved techniques will be universally applied, to maximize the likelihood of a positive outcome from any intervention.
Improved techniques will be used for all ART embryos. Although the specifics of individual laboratory techniques are of limited interest to the general Ob/Gyn, the study emphasizes a growing trend in ART that will have far-reaching consequences. Currently, preimplantation genetic diagnosis is offered to couples with known genetic defects and to an increasing number of older couples at greater risk of producing embryos with genetic abnormalities.
As the techniques improve in reliability, applicability, and cost-effectiveness, they will be universally applied to all embryos resulting from ART procedures, to the extent that only genetically “normal” embryos will be transferred. This has the potential to drive all infertility therapy in the direction of ART, to maximize the likelihood of a positive outcome from any intervention.
Surgery, superovulation will dwindle. The necessity of infertility surgery will continue to diminish and use of superovulation as an isolated therapy will disappear, since the number and quality of resulting fetuses cannot be controlled.
CDC study: Insured patients may opt for fewer embryos
Reynolds MA, Schieve LA, Jeng G, Peterson HB. Does insurance coverage decrease the risk for multiple births associated with assisted reproductive technology? Fertil Steril. 2003;80:16-23.
Multiple births have soared over the past generation, primarily because of ART, specifically superovulation and in vitro fertilization. The initial emphasis was simply to achieve pregnancy, but now, the medical and financial risks related to multiple gestation are a target of concern. Strategies to decrease multiple pregnancies and yet maintain high pregnancy rates are now a health policy priority.
CDC examines assisted reproduction outcomes in insured vs noninsured states. This CDC study compared ART practices and outcomes in 3 states with mandated ART insurance coverage (Illinois, Massachusetts, and Rhode Island) to 3 states without such mandated coverage (Indiana, Michigan, and New Jersey). Outcome measures were number of embryos transferred, multiple-birth rate, triplet or higher order birth rate, and triplet or higher order gestation rate. The study utilized data from the national registry for 1998.
A smaller proportion of procedures included transfer of 3 or more embryos in 2 of the 3 states with mandated insurance (Massachusetts, 64% and Rhode Island, 74%) than in the noninsurance states (82%), and the multiple-birth rate was lower in 1 of the mandated states (Massachusetts, 38%) than in the nonmandated states (43%). A trend toward fewer triplet or higher order births was seen in all mandated states, but was statistically significant only in Massachusetts.
Implication: Insurance may be a tool to reduce multiple births. While the magnitude of the differences in the number of embryos transferred and the multiple-birth rate was not great, this study does suggest that insurance coverage may be an effective way to modify practice patterns and outcomes. Higher order multiple-birth rates are substantially lower in countries with national health insurance and restrictions on the number of embryos transferred. In the United States, where most states have not mandated ART insurance and there is no restriction on number of embryos transferred, the decision and risks are assumed by the patient and practitioner.
The natural tendency is to maximize the yield from an expensive procedure, and accept the increased risk.
Without insurance, the motivation is maximum yield. Unlike health decisions where cost is less central, the natural tendency is to try to maximize the yield from a single expensive procedure and accept the increased risk. This study suggests that reducing personal financial risk can modify those decisions.
Would long-term savings offset higher insurance costs? The move to find ways to reduce the rate of multiple gestations is welcome news for the practicing Ob/Gyn. While any attempt to increase health-care coverage may seem counterintuitive in an era of escalating health costs, a subsequent reduction in the multiple-birth rate would provide long-term savings by reducing the number of multiple gestations and their well-documented increase in premature deliveries and long-term costs.
Be alert for cost-analysis studies in the upcoming year.
Dr. Randolph reports no affiliations or financial arrangements with any companies whose products are mentioned in this article.
Advancements during the year 2003 propelled infertility therapy further than ever along its 20-year evolution toward extensive use of assisted reproductive technologies (ART) and the introduction of diagnostic genetics. Refreshingly, social and ethical implications were viewed with greater recognition that we must deal effectively with the repercussions of technical progress.
Basic evaluation still key; early referral appropriate. For the practicing Ob/Gyn already confused by the bewildering and growing array of options presented to patients, these changes do not mean that a basic infertility evaluation and primary therapy are inappropriate. However, early referral to a trusted, experienced infertility specialist before invasive diagnostic studies such as laparoscopy or aggressive therapies is now most appropriate.
This Update reviews trends in reproductive medicine that herald both clinical and social transformations.
Oocyte cryopreservation: Ready for prime time
Porcu E. Human oocyte cryopreservation: from basic research to clinical application. Tap Pharmaceuticals Endowed Lecture presented at: American Society for Reproductive Medicine 59th Annual Meeting; October 15, 2003; San Antonio, Tex.
Growing concern over the past decade about age-related infertility due to progressive loss of oocyte number and quality has prompted rapidly expanding use of donated oocytes from younger women. Due to technical difficulties in cryopreserving oocytes, it has not been clinically feasible to save eggs from young women for later use or for donation to other women from “egg banks,” as has been done with sperm for many years.
In a presentation on the research program in oocyte freezing at the University of Bologna, Italy, Dr. Eleonora Porcu elaborated on the last decade’s progress. She described progressively improving clinical pregnancy rates from cryopreserved eggs, and commented that clinical experience to date is sufficient to begin offering this option to women generally.
A growing number of centers are offering oocyte cryopreservation, although outcome data for specific centers are limited and no national aggregate data are yet available.
Cryopreservation will beat the biological clock. Although immediate clinical use of this technology will be directed to young women at risk for loss of ovarian function due to cancer therapy or oophorectomy, the social implications could be comparable to the introduction of effective contraception in the 1960s.
Just as oral contraceptives allowed women to delay childbearing and yet be sexually active, oocyte cryopreservation may allow women to delay childbearing without fear that the “biological clock” will strike midnight before they have had children.
More older mothers, more concerns. While demand for this service is undetermined, and many biological and financial issues remain to be delineated, this process will continue the trend to later childbearing and older mothers, with all their attendant social and medical concerns.
Prepare to provide referral services. It will be increasingly important for physicians to be aware of the availability of such services and to have a ready source of referral for their patients interested in pursuing oocyte cryopreservation.
On the horizon: Unlimited supply of gametes. In a scientifically related but much less clinically applicable presentation at the same meeting, Susan Rothmann, Roger Gosden, and Pasquale Patrizio described the status of research in germ-line stem cells and reviewed the evidence for oogonial stem cells in human ovaries and the efforts to isolate and cryopreserve such stem cells.
Although much work needs to be done before spermatogonial and oogonial stem cells can be isolated and stored from patients, this work holds the promise that storage of germ-line stem cells may provide an unlimited future supply of gametes for use in later reproduction.
Related Reading
Porcu E. Oocyte freezing. Semin Reprod Med. 2001;19:221-230.
Advances in genetic diagnosis will enable transfer of ‘normal’ embryos only
Wilton L, Voullaire L, Sargeant P, Williamson R, McBain J. Preimplantation aneuploidy screening using comparative genomic hybridization or fluorescence in situ hybridization of embryos from patients with recurrent implantation failure. Fertil Steril. 2003;80:860-868.
One of the hottest topics today in reproductive medicine is the increasing availability of preimplantation diagnosis to determine if embryos are genetically normal. This Australian study compared 2 methods of genetic evaluation for their sensitivity in identifying abnormalities and in predicting subsequent implantation and pregnancy rates. The investigators concluded that comparative genomic hybridization was more effective than fluorescence in situ hybridization in identifying chromosomally normal embryos.
Improved techniques will be universally applied, to maximize the likelihood of a positive outcome from any intervention.
Improved techniques will be used for all ART embryos. Although the specifics of individual laboratory techniques are of limited interest to the general Ob/Gyn, the study emphasizes a growing trend in ART that will have far-reaching consequences. Currently, preimplantation genetic diagnosis is offered to couples with known genetic defects and to an increasing number of older couples at greater risk of producing embryos with genetic abnormalities.
As the techniques improve in reliability, applicability, and cost-effectiveness, they will be universally applied to all embryos resulting from ART procedures, to the extent that only genetically “normal” embryos will be transferred. This has the potential to drive all infertility therapy in the direction of ART, to maximize the likelihood of a positive outcome from any intervention.
Surgery, superovulation will dwindle. The necessity of infertility surgery will continue to diminish and use of superovulation as an isolated therapy will disappear, since the number and quality of resulting fetuses cannot be controlled.
CDC study: Insured patients may opt for fewer embryos
Reynolds MA, Schieve LA, Jeng G, Peterson HB. Does insurance coverage decrease the risk for multiple births associated with assisted reproductive technology? Fertil Steril. 2003;80:16-23.
Multiple births have soared over the past generation, primarily because of ART, specifically superovulation and in vitro fertilization. The initial emphasis was simply to achieve pregnancy, but now, the medical and financial risks related to multiple gestation are a target of concern. Strategies to decrease multiple pregnancies and yet maintain high pregnancy rates are now a health policy priority.
CDC examines assisted reproduction outcomes in insured vs noninsured states. This CDC study compared ART practices and outcomes in 3 states with mandated ART insurance coverage (Illinois, Massachusetts, and Rhode Island) to 3 states without such mandated coverage (Indiana, Michigan, and New Jersey). Outcome measures were number of embryos transferred, multiple-birth rate, triplet or higher order birth rate, and triplet or higher order gestation rate. The study utilized data from the national registry for 1998.
A smaller proportion of procedures included transfer of 3 or more embryos in 2 of the 3 states with mandated insurance (Massachusetts, 64% and Rhode Island, 74%) than in the noninsurance states (82%), and the multiple-birth rate was lower in 1 of the mandated states (Massachusetts, 38%) than in the nonmandated states (43%). A trend toward fewer triplet or higher order births was seen in all mandated states, but was statistically significant only in Massachusetts.
Implication: Insurance may be a tool to reduce multiple births. While the magnitude of the differences in the number of embryos transferred and the multiple-birth rate was not great, this study does suggest that insurance coverage may be an effective way to modify practice patterns and outcomes. Higher order multiple-birth rates are substantially lower in countries with national health insurance and restrictions on the number of embryos transferred. In the United States, where most states have not mandated ART insurance and there is no restriction on number of embryos transferred, the decision and risks are assumed by the patient and practitioner.
The natural tendency is to maximize the yield from an expensive procedure, and accept the increased risk.
Without insurance, the motivation is maximum yield. Unlike health decisions where cost is less central, the natural tendency is to try to maximize the yield from a single expensive procedure and accept the increased risk. This study suggests that reducing personal financial risk can modify those decisions.
Would long-term savings offset higher insurance costs? The move to find ways to reduce the rate of multiple gestations is welcome news for the practicing Ob/Gyn. While any attempt to increase health-care coverage may seem counterintuitive in an era of escalating health costs, a subsequent reduction in the multiple-birth rate would provide long-term savings by reducing the number of multiple gestations and their well-documented increase in premature deliveries and long-term costs.
Be alert for cost-analysis studies in the upcoming year.
Dr. Randolph reports no affiliations or financial arrangements with any companies whose products are mentioned in this article.
Advancements during the year 2003 propelled infertility therapy further than ever along its 20-year evolution toward extensive use of assisted reproductive technologies (ART) and the introduction of diagnostic genetics. Refreshingly, social and ethical implications were viewed with greater recognition that we must deal effectively with the repercussions of technical progress.
Basic evaluation still key; early referral appropriate. For the practicing Ob/Gyn already confused by the bewildering and growing array of options presented to patients, these changes do not mean that a basic infertility evaluation and primary therapy are inappropriate. However, early referral to a trusted, experienced infertility specialist before invasive diagnostic studies such as laparoscopy or aggressive therapies is now most appropriate.
This Update reviews trends in reproductive medicine that herald both clinical and social transformations.
Oocyte cryopreservation: Ready for prime time
Porcu E. Human oocyte cryopreservation: from basic research to clinical application. Tap Pharmaceuticals Endowed Lecture presented at: American Society for Reproductive Medicine 59th Annual Meeting; October 15, 2003; San Antonio, Tex.
Growing concern over the past decade about age-related infertility due to progressive loss of oocyte number and quality has prompted rapidly expanding use of donated oocytes from younger women. Due to technical difficulties in cryopreserving oocytes, it has not been clinically feasible to save eggs from young women for later use or for donation to other women from “egg banks,” as has been done with sperm for many years.
In a presentation on the research program in oocyte freezing at the University of Bologna, Italy, Dr. Eleonora Porcu elaborated on the last decade’s progress. She described progressively improving clinical pregnancy rates from cryopreserved eggs, and commented that clinical experience to date is sufficient to begin offering this option to women generally.
A growing number of centers are offering oocyte cryopreservation, although outcome data for specific centers are limited and no national aggregate data are yet available.
Cryopreservation will beat the biological clock. Although immediate clinical use of this technology will be directed to young women at risk for loss of ovarian function due to cancer therapy or oophorectomy, the social implications could be comparable to the introduction of effective contraception in the 1960s.
Just as oral contraceptives allowed women to delay childbearing and yet be sexually active, oocyte cryopreservation may allow women to delay childbearing without fear that the “biological clock” will strike midnight before they have had children.
More older mothers, more concerns. While demand for this service is undetermined, and many biological and financial issues remain to be delineated, this process will continue the trend to later childbearing and older mothers, with all their attendant social and medical concerns.
Prepare to provide referral services. It will be increasingly important for physicians to be aware of the availability of such services and to have a ready source of referral for their patients interested in pursuing oocyte cryopreservation.
On the horizon: Unlimited supply of gametes. In a scientifically related but much less clinically applicable presentation at the same meeting, Susan Rothmann, Roger Gosden, and Pasquale Patrizio described the status of research in germ-line stem cells and reviewed the evidence for oogonial stem cells in human ovaries and the efforts to isolate and cryopreserve such stem cells.
Although much work needs to be done before spermatogonial and oogonial stem cells can be isolated and stored from patients, this work holds the promise that storage of germ-line stem cells may provide an unlimited future supply of gametes for use in later reproduction.
Related Reading
Porcu E. Oocyte freezing. Semin Reprod Med. 2001;19:221-230.
Advances in genetic diagnosis will enable transfer of ‘normal’ embryos only
Wilton L, Voullaire L, Sargeant P, Williamson R, McBain J. Preimplantation aneuploidy screening using comparative genomic hybridization or fluorescence in situ hybridization of embryos from patients with recurrent implantation failure. Fertil Steril. 2003;80:860-868.
One of the hottest topics today in reproductive medicine is the increasing availability of preimplantation diagnosis to determine if embryos are genetically normal. This Australian study compared 2 methods of genetic evaluation for their sensitivity in identifying abnormalities and in predicting subsequent implantation and pregnancy rates. The investigators concluded that comparative genomic hybridization was more effective than fluorescence in situ hybridization in identifying chromosomally normal embryos.
Improved techniques will be universally applied, to maximize the likelihood of a positive outcome from any intervention.
Improved techniques will be used for all ART embryos. Although the specifics of individual laboratory techniques are of limited interest to the general Ob/Gyn, the study emphasizes a growing trend in ART that will have far-reaching consequences. Currently, preimplantation genetic diagnosis is offered to couples with known genetic defects and to an increasing number of older couples at greater risk of producing embryos with genetic abnormalities.
As the techniques improve in reliability, applicability, and cost-effectiveness, they will be universally applied to all embryos resulting from ART procedures, to the extent that only genetically “normal” embryos will be transferred. This has the potential to drive all infertility therapy in the direction of ART, to maximize the likelihood of a positive outcome from any intervention.
Surgery, superovulation will dwindle. The necessity of infertility surgery will continue to diminish and use of superovulation as an isolated therapy will disappear, since the number and quality of resulting fetuses cannot be controlled.
CDC study: Insured patients may opt for fewer embryos
Reynolds MA, Schieve LA, Jeng G, Peterson HB. Does insurance coverage decrease the risk for multiple births associated with assisted reproductive technology? Fertil Steril. 2003;80:16-23.
Multiple births have soared over the past generation, primarily because of ART, specifically superovulation and in vitro fertilization. The initial emphasis was simply to achieve pregnancy, but now, the medical and financial risks related to multiple gestation are a target of concern. Strategies to decrease multiple pregnancies and yet maintain high pregnancy rates are now a health policy priority.
CDC examines assisted reproduction outcomes in insured vs noninsured states. This CDC study compared ART practices and outcomes in 3 states with mandated ART insurance coverage (Illinois, Massachusetts, and Rhode Island) to 3 states without such mandated coverage (Indiana, Michigan, and New Jersey). Outcome measures were number of embryos transferred, multiple-birth rate, triplet or higher order birth rate, and triplet or higher order gestation rate. The study utilized data from the national registry for 1998.
A smaller proportion of procedures included transfer of 3 or more embryos in 2 of the 3 states with mandated insurance (Massachusetts, 64% and Rhode Island, 74%) than in the noninsurance states (82%), and the multiple-birth rate was lower in 1 of the mandated states (Massachusetts, 38%) than in the nonmandated states (43%). A trend toward fewer triplet or higher order births was seen in all mandated states, but was statistically significant only in Massachusetts.
Implication: Insurance may be a tool to reduce multiple births. While the magnitude of the differences in the number of embryos transferred and the multiple-birth rate was not great, this study does suggest that insurance coverage may be an effective way to modify practice patterns and outcomes. Higher order multiple-birth rates are substantially lower in countries with national health insurance and restrictions on the number of embryos transferred. In the United States, where most states have not mandated ART insurance and there is no restriction on number of embryos transferred, the decision and risks are assumed by the patient and practitioner.
The natural tendency is to maximize the yield from an expensive procedure, and accept the increased risk.
Without insurance, the motivation is maximum yield. Unlike health decisions where cost is less central, the natural tendency is to try to maximize the yield from a single expensive procedure and accept the increased risk. This study suggests that reducing personal financial risk can modify those decisions.
Would long-term savings offset higher insurance costs? The move to find ways to reduce the rate of multiple gestations is welcome news for the practicing Ob/Gyn. While any attempt to increase health-care coverage may seem counterintuitive in an era of escalating health costs, a subsequent reduction in the multiple-birth rate would provide long-term savings by reducing the number of multiple gestations and their well-documented increase in premature deliveries and long-term costs.
Be alert for cost-analysis studies in the upcoming year.
Dr. Randolph reports no affiliations or financial arrangements with any companies whose products are mentioned in this article.
Electronic fetal monitoring: The difficulty of linking patterns with outcomes
- High-risk pattern. Fetuses at greatest risk include those with marked bradycardia, recurrent late and variable decelerations, and absent variability.
- Normal pattern. In contrast, a normal baseline rate with normal, moderate variability and accelerations, and absence of periodic patterns (decelerations) predicts fetal well-being.
- Tips on interpretation. Deciphering fetal heart rate patterns entails making observations over time. Many patterns acquire increased significance when there is a trend toward persistent, significant departures from baseline with decreased variability, loss of accelerations, or persistent episodic or periodic decelerations, particularly with loss of variability.
True or false: Electronic fetal monitoring reduces the incidence of cerebral palsy and infant morbidity and mortality.
Unfortunately, the statement is false, although patients continue to believe it is true. As a result, a large proportion of obstetrics liability cases center on electronic fetal monitoring (EFM): 43% of all lawsuits alleging obstetric malpractice, 52% of cases involving a stillborn fetus or neonatal death, and 66% of cases involving a neurologically impaired infant.1
Although the reliability and validity of EFM in reducing perinatal morbidity and mortality leave much to be desired, they could be greatly improved by increasing our understanding of the physiologic causes of patterns, establishing formal definitions for the various fetal heart rate (FHR) patterns, and developing specific recommendations for “abnormal” patterns.
Once these goals are attained, the question of whether EFM can be used to prevent asphyxial brain damage can be more adequately addressed.
This article discusses the following topics:
- EFM patterns and their importance,
- key research findings to date, and
- how to identify patients at greatest risk of an adverse outcome.
Unfulfilled hopes based on flawed assumption
Evaluation of the FHR in labor—first by auscultation and then by continuous EFM—has long held promise of improved outcomes. As early as the 1800s, auscultated Electronic fetal monitoring: The difficulty of linking patterns with outcomes FHR decelerations and prolonged bradycardias were seen as signs of fetal distress in labor. When criteria were being developed for obstetric intervention with forceps in the late 1800s, tachycardia was included as an indication, building the foundation for contemporary definitions of nonreassuring FHR patterns.
Many decades later, abnormal FHR patterns correlated with low Apgar scores and neonatal death (although the correlation between certain patterns and poor outcomes was weak) in the findings of the American Collaborative Perinatal Study,2 which reviewed 25,000 births monitored using intermittent auscultation from 1959 through 1965.
The largely flawed assumption that most cases of cerebral palsy are caused by intrapartum asphyxia led to a concerted effort to decrease the incidence of asphyxia through EFM. By the 1970s, the expectation was that EFM would decrease perinatal morbidity and mortality.
Since its introduction into clinical practice in the early 1970s, EFM has become increasingly common; its use grew from 61% of women giving birth in 1989 to 84% in 1998.3
Findings of randomized controlled trials. The first randomized controlled trial4 of EFM in labor, in 1976, demonstrated no improvement in outcome but a significant increase in surgical births; later trials showed no decrease in perinatal mortality or cerebral palsy.
The 1985 Dublin trial,5 which scored the highest in study design in a later meta-analysis, showed a slight protective effect of EFM overall.
A 1995 meta-analysis6 of 12 randomized controlled trials encompassing 58,555 pregnancies identified 9 outcomes, including Apgar scores, perinatal mortality, and neonatal seizures. EFM had a consistent impact only on neonatal seizures, and had no measurable effect on morbidity and mortality. Overall, 1.1% of neonates in the auscultated group had seizures, compared with 0.8% in the EFM group. Operative deliveries also increased in the EFM group.
EFM effects on neonatal outcomes. Freeman7 summarized the gains of EFM from 1972 to 1990 as follows:
- no effect on cerebral palsy,
- no effect on neonatal morbidity, and
- no improvement in neurologic outcomes in premature infants.
In 1995, Vintzileos and colleagues8 studied fetal acidemia in 680 auscultated labors and compared them with 739 monitored by EFM, using umbilical artery pH of less than 7.15 as an outcome measure. EFM was superior to auscultation, with better sensitivity and higher positive and negative predictive value.
Identification of patterns still rests on visual interpretation
In 1997, after a workshop to develop standardized, unambiguous definitions for fetal heart tracings, the National Institute for Child Health and Human Development (NICHD) issued guidelines for interpreting EFM9; these focus on visual interpretation, though computerized analyses are being developed. The definitions emphasize intrapartum evaluation, and the major patterns are categorized as baseline, periodic, or episodic.
Variability is defined by amplitude and includes short-term (beat to beat) and long-term variability, with no distinction between the two. Sinusoidal patterns are excluded from the definition.
According to NICHD guidelines, variabilities can be defined as follows:
- amplitude undetectable = absent variability;
- amplitude of 0 to 5 beats per minute (bpm) = minimal variability;
- 5 to 25 bpm = moderate variability; and
- amplitude exceeding 25 bpm = marked variability.
Loss of variability is the most sensitive indicator of fetal acidemia, cerebral asphyxia, and myocardial depression, when it is associated with other periodic patterns.10
Baseline rate. Baseline heart rate is the approximate mean rounded to 5 bpm during a 10-minute interval, excluding periodic or episodic changes, periods of marked variability, and segments of the baseline that differ by more than 25 bpm. The baseline must continue for 2 minutes during that 10-minute period or it is considered indeterminate. A normal baseline generally ranges from 110 to 160 bpm. Below 110 bpm is bradycardia, and above 160 bpm is tachycardia.
Tachycardia and bradycardia indicate changes in the baseline rate.
Causes of tachycardia include maternal fever, sympathomimetics, and fetal arrhythmia. Tachycardia with a loss in variability and recurrent late or variable decelerations may indicate fetal acidemia.
In contrast, bradycardia ranging between 80 and 110 bpm does not indicate fetal hypoxia if the variability is retained, but a rate less than 80 bpm may indicate fetal hypoxia. The fetus may demonstrate terminal bradycardia associated with prolonged head or cord compression, but may tolerate this as long as variability is maintained. Sinusoidal patterns are excluded.
Decelerations are quantified by depth of the nadir (below the baseline) in bpm, with the duration measured in minutes or seconds from the beginning to the end of the deceleration (FIGURES 1 and 2). Those that occur more than 50% of the time are recurrent or periodic.
Acceleration is an abrupt change above baseline. Before 32 weeks, accelerations are defined as 10 or more bpm over baseline for 10 seconds or more. Peak acceleration is a minimum of 15 bpm above baseline, with a duration ranging from 15 seconds to 2 minutes before return to baseline. A prolonged acceleration continues for more than 2 minutes; if it lasts more than 10 minutes, it is considered a change in baseline.
Periodic and episodic patterns. Periodic patterns include early, late, and variable decelerations associated with uterine contractions. Episodic patterns are those not associated with uterine contractions.
A late deceleration is a visually apparent gradual decrease below the baseline for 30 seconds (at the nadir) that is delayed in timing relative to the uterine contraction. The recovery also is delayed relative to the end of the uterine contraction. Late decelerations that maintain variability are thought to indicate a well-oxygenated fetus and are neurogenic in origin,11 but late decelerations without variability are thought to represent possible asphyxia. In fetuses with prolonged placental insufficiency, these smooth late decelerations are thought to reflect decreased cerebral and myocardial function.
Early decelerations are visually apparent decreases of 30 seconds (to the nadir) that start and end with a uterine contraction. They are not associated with significant fetal acidemia.
Variable decelerations are abrupt and visually apparent decelerations of 15 bpm below the baseline; they last more than 15 seconds to the nadir and less than 2 minutes overall. Variable decelerations are generally due to head compression, vagal stimulation, or cord compression. When variable decelerations are persistent and severe, tachycardia, delayed return to baseline, or decreased variability may occur, reflecting fetal acidemia.12
Prolonged late and severe variable decelerations may lead to fetal hypoxia. A duration between 2 and 10 minutes indicates a prolonged deceleration. A change of 10 minutes or more indicates a change in baseline.
Absent or severe variability with persistent late and prolonged decelerations is generally believed to be ominous and may correlate with hypoxia of such severity that the fetal central nervous system may have already been damaged. Surrogate markers for intrapartum asphyxia include mixed acidemia at birth, low Apgar score (less than 3 at 5 minutes), seizures within 24 hours, and multiorgan dysfunction.
High-risk pattern. Fetuses at greatest risk exhibit marked bradycardia, recurrent late and variable decelerations, and absent variability.
Normal pattern. In contrast, a normal baseline with moderate variability and accelerations and absence of periodic patterns (decelerations) is predictive of fetal well-being.
Tips on interpretation. Deciphering EFM patterns entails making observations over time. Many patterns acquire increased significance when there is a trend toward persistent, significant departures from baseline with decreased variability, loss of accelerations, or persistent episodic or periodic decelerations, particularly with loss of variability.
FIGURE 2 Severe deceleration
A severe deceleration with variability. When variable decelerations are persistent and severe, fetal acidemia may be present.
Human factors
Agreement between observers. The reliability and reproducibility of EFM interpretation have been reviewed, but prospective studies to evaluate them are lacking. Interobserver agreement is reasonably high for the baseline rate, accelerations, and decelerations, but lower for variable decelerations and lower still for variability. Computers have been investigated for standardizing EFM interpretation.
Competency of physicians and nurses. A study14 assessing competency in the evaluation of EFM tracings surveyed 43 Ob/Gyn training programs.
Researchers found that the majority (79%) use clinical experience along with structured lectures (87%) to train residents and fellows. Perinatal morbidity and mortality conferences were used by 85% of programs, and maintenance of EFM skills involved both clinical experience and case studies.
Although registered nurses typically undergo annual competency evaluation in EFM, physician competency is not formally evaluated. Thus, some other mechanism for ensuring expertise would be helpful.
Unreliable link to outcomes
The correlation between FHR patterns and neonatal outcomes is difficult to establish. The causative event in 70% of neonatal encephalopathy cases is believed to have occurred before the onset of labor13; therefore, few cases of cerebral palsy and neonatal encephalopathy are amenable to interventions during labor. Abnormal patterns that do not allow sufficient time for intervention also may have poor outcomes.
Fetuses that have intrapartum asphyxia usually have abnormal heart rate patterns. However, most women with abnormal FHR patterns give birth to neonates with normal Apgar scores and outcomes.
The central tenet of the 2003 monograph13 by the American College of Obstetricians and Gynecologists is that in order for an intrapartum event to progress to cerebral palsy, the pathway must proceed through neonatal encephalopathy. However, only 30% of neonatal encephalopathy cases are due to events during labor. Further, epilepsy, mental retardation, and attention deficit disorder are not caused by birth asphyxia.
A 99% false-positive rate may result when nonreassuring EFM tracings are used to predict cerebral palsy. For this reason, there is still considerable disagreement about whether EFM can reduce the incidence of cerebral palsy. Using the criteria for establishing acute hypoxic-ischemic insult (TABLE 1) and for determining that intrapartum events were sufficient to cause cerebral palsy (TABLE 2), only 9% of cerebral palsy cases are attributable to possible birth asphyxia.11
Most cases of catastrophic hypoxia ultimately lead to fetal or neonatal death; a minority of infants survive and develop cerebral palsy. The pattern of neurologic injury following acute catastrophic hypoxic-ischemic insult can involve the basal ganglia and thalami; it may be different from the pattern that commonly follows chronic insult, which involves primarily the cerebral cortex and subcortical white matter.
TABLE 1
Criteria for establishing acute hypoxic-ischemic insult
| Metabolic acidosis evident in fetal umbilical cord arterial blood at delivery (pH <7.0 or base excess ≥ 12 mmol/L) |
| Early onset of severe or moderate neonatal encephalopathy in infants delivered at ≥ 34 weeks’ gestation |
| Cerebral palsy of the spastic quadriplegic or dyskinetic type |
| Exclusion of other identifiable causes |
| Data from American College of Obstetricians and Gynecologists13 |
TABLE 2
Criteria to establish intrapartum event as cause of cerebral palsy
| Sentinel hypoxic event occurring immediately before or during labor |
| Sudden sustained fetal bradycardia or absence of fetal heart rate variability in presence of persistent late or variable decelerations |
| Apgar score of less than 3 at 5 minutes |
| Onset of multisystem involvement within 72 hours of birth |
| Early imaging studies showing evidence of acute nonfocal abnormality. |
| Data from American College of Obstetricians and Gynecologists13 |
Little time to react during labor
Severe intrapartum hypoxic-ischemic insult may be detected by EFM but, in some cases, the reaction time to prevent fetal brain injury is very short. These intrapartum events include umbilical cord prolapse, shoulder dystocia, uterine rupture, and maternal cardiopulmonary arrest. With uterine rupture, asphyxia may occur as quickly as 10 minutes after onset of bradycardia when the prolonged deceleration is preceded by severe late decelerations of 30 to 90 minutes. The average time from onset of prolonged bradycardia has been reported as 13±6.5 minutes.13
The 30-minute rule. Because mortality and morbidity may depend on how much time elapses from onset of the catastrophic event to delivery, ACOG developed guidelines for timing emergent cesarean. The 30-minute rule13 advises that a cesarean be performed within 30 minutes of the decision to proceed. Unfortunately, in many cases, acute hypoxic-ischemic insult occurs in less than 30 minutes.
Many hospitals and obstetric services no longer offer vaginal birth after cesarean (VBAC) if they cannot guarantee that anesthesia, nursing, pediatric, and operating room services will be available within this time frame. However, other causes of sudden hypoxia—besides the small chance of uterine rupture with VBAC—may be neither predictable nor preventable.
General recommendations
In cases of nonreassuring FHR tracings, the options include maternal positioning to achieve uterine displacement; hydration; correction of hypotension, which may be useful when regional anesthetics are used; tocolytic use for treating a uterine contraction; maternal oxygen; amnio-infusion for thick meconium in the presence of a nonreassuring FHR tracing; and assessment and correction of the oxytocin dose when augmentation or induction of labor is occurring.
The authors report no financial relationships relevant to this article.
1. American College of Obstetricians and Gynecologists. 1999 Survey of Professional Liability: Claims Data Tabulations. Washington, DC: ACOG; 1999.
2. Benson RC, Schubeck F, Deutschberger J, et al. Fetal heart rate as a predictor of fetal distress: a report from the collaborative project. Obstet Gynecol. 1968;32:259-266.
3. Curtin SC, Matthews TJ. US obstetric procedures, 1998. Birth. 2000;27:136-138.
4. Havercamp AD, Thompson HE, McFee JG, Cetrulo C. The evaluation of continuous fetal heart rate monitoring in high-risk pregnancy. Am J Obstet Gynecol. 1976;125(3):310-320.
5. MacDonald D, Grant A, Sheridan-Pereira M, et al. The Dublin randomized control trial of intrapartum fetal heart rate monitoring. Am J Obstet Gynecol. 1985;152:524-529.
6. Thacker SB, Stroup DF, Peterson HB. Efficacy and safety of intrapartum electronic fetal monitoring: an update. Obstet Gynecol. 1995;86:613-620.
7. Freeman RK. Intrapartum electronic fetal monitoring—a disappointing story. N Engl J Med. 1990;322:624-626.
8. Vintzileos AM, Nochimson DJ, Antsaklis A, Varvarigos I, Guzman ER, Knuppel RA. Comparison of intrapartum electronic fetal heart rate monitoring versus intermittent auscultation in detecting fetal acidemia at birth. Am J Obstet Gynecol. 1995;173:1021-1024.
9. National Institute of Child Health and Human Development Research Planning Workshop. Electronic fetal heart rate monitoring: research guidelines for interpretation. Am J Obstet Gynecol. 1997;177:1385-1390.
10. Court DJ, Parer JT. Experimental studies in fetal asphyxial heart rate interpretation. In: Nathanielsz PW, Parer JT, eds. Research in Perinatal Medicine. Ithaca, NY: Perinatology Press; 1985;114-167.
11. Paul RH, Suidan AH, Yen S, et al. Clinical fetal monitoring III: the evaluation and significance of intrapartum fetal heart rate variability. Am J Obstet Gynecol. 1975;123:206-210.
12. Ball RH, Parer JT. The physiologic mechanism of variable decelerations. AmJ Obstet Gynecol. 1992;166:1683-1689.
13. American College of Obstetricians and Gynecologists. Neonatal Encephalopathy and Cerebral Palsy: Defining the Pathogenesis and Pathophysiology. Washington, DC: ACOG; 2003.
14. Murphy AA, Halamek LP, Lyell DJ, et al. Training and competency assessment in electronic fetal monitoring: a national survey. Obstet Gynecol. 2003;101:1243-1248.
- High-risk pattern. Fetuses at greatest risk include those with marked bradycardia, recurrent late and variable decelerations, and absent variability.
- Normal pattern. In contrast, a normal baseline rate with normal, moderate variability and accelerations, and absence of periodic patterns (decelerations) predicts fetal well-being.
- Tips on interpretation. Deciphering fetal heart rate patterns entails making observations over time. Many patterns acquire increased significance when there is a trend toward persistent, significant departures from baseline with decreased variability, loss of accelerations, or persistent episodic or periodic decelerations, particularly with loss of variability.
True or false: Electronic fetal monitoring reduces the incidence of cerebral palsy and infant morbidity and mortality.
Unfortunately, the statement is false, although patients continue to believe it is true. As a result, a large proportion of obstetrics liability cases center on electronic fetal monitoring (EFM): 43% of all lawsuits alleging obstetric malpractice, 52% of cases involving a stillborn fetus or neonatal death, and 66% of cases involving a neurologically impaired infant.1
Although the reliability and validity of EFM in reducing perinatal morbidity and mortality leave much to be desired, they could be greatly improved by increasing our understanding of the physiologic causes of patterns, establishing formal definitions for the various fetal heart rate (FHR) patterns, and developing specific recommendations for “abnormal” patterns.
Once these goals are attained, the question of whether EFM can be used to prevent asphyxial brain damage can be more adequately addressed.
This article discusses the following topics:
- EFM patterns and their importance,
- key research findings to date, and
- how to identify patients at greatest risk of an adverse outcome.
Unfulfilled hopes based on flawed assumption
Evaluation of the FHR in labor—first by auscultation and then by continuous EFM—has long held promise of improved outcomes. As early as the 1800s, auscultated Electronic fetal monitoring: The difficulty of linking patterns with outcomes FHR decelerations and prolonged bradycardias were seen as signs of fetal distress in labor. When criteria were being developed for obstetric intervention with forceps in the late 1800s, tachycardia was included as an indication, building the foundation for contemporary definitions of nonreassuring FHR patterns.
Many decades later, abnormal FHR patterns correlated with low Apgar scores and neonatal death (although the correlation between certain patterns and poor outcomes was weak) in the findings of the American Collaborative Perinatal Study,2 which reviewed 25,000 births monitored using intermittent auscultation from 1959 through 1965.
The largely flawed assumption that most cases of cerebral palsy are caused by intrapartum asphyxia led to a concerted effort to decrease the incidence of asphyxia through EFM. By the 1970s, the expectation was that EFM would decrease perinatal morbidity and mortality.
Since its introduction into clinical practice in the early 1970s, EFM has become increasingly common; its use grew from 61% of women giving birth in 1989 to 84% in 1998.3
Findings of randomized controlled trials. The first randomized controlled trial4 of EFM in labor, in 1976, demonstrated no improvement in outcome but a significant increase in surgical births; later trials showed no decrease in perinatal mortality or cerebral palsy.
The 1985 Dublin trial,5 which scored the highest in study design in a later meta-analysis, showed a slight protective effect of EFM overall.
A 1995 meta-analysis6 of 12 randomized controlled trials encompassing 58,555 pregnancies identified 9 outcomes, including Apgar scores, perinatal mortality, and neonatal seizures. EFM had a consistent impact only on neonatal seizures, and had no measurable effect on morbidity and mortality. Overall, 1.1% of neonates in the auscultated group had seizures, compared with 0.8% in the EFM group. Operative deliveries also increased in the EFM group.
EFM effects on neonatal outcomes. Freeman7 summarized the gains of EFM from 1972 to 1990 as follows:
- no effect on cerebral palsy,
- no effect on neonatal morbidity, and
- no improvement in neurologic outcomes in premature infants.
In 1995, Vintzileos and colleagues8 studied fetal acidemia in 680 auscultated labors and compared them with 739 monitored by EFM, using umbilical artery pH of less than 7.15 as an outcome measure. EFM was superior to auscultation, with better sensitivity and higher positive and negative predictive value.
Identification of patterns still rests on visual interpretation
In 1997, after a workshop to develop standardized, unambiguous definitions for fetal heart tracings, the National Institute for Child Health and Human Development (NICHD) issued guidelines for interpreting EFM9; these focus on visual interpretation, though computerized analyses are being developed. The definitions emphasize intrapartum evaluation, and the major patterns are categorized as baseline, periodic, or episodic.
Variability is defined by amplitude and includes short-term (beat to beat) and long-term variability, with no distinction between the two. Sinusoidal patterns are excluded from the definition.
According to NICHD guidelines, variabilities can be defined as follows:
- amplitude undetectable = absent variability;
- amplitude of 0 to 5 beats per minute (bpm) = minimal variability;
- 5 to 25 bpm = moderate variability; and
- amplitude exceeding 25 bpm = marked variability.
Loss of variability is the most sensitive indicator of fetal acidemia, cerebral asphyxia, and myocardial depression, when it is associated with other periodic patterns.10
Baseline rate. Baseline heart rate is the approximate mean rounded to 5 bpm during a 10-minute interval, excluding periodic or episodic changes, periods of marked variability, and segments of the baseline that differ by more than 25 bpm. The baseline must continue for 2 minutes during that 10-minute period or it is considered indeterminate. A normal baseline generally ranges from 110 to 160 bpm. Below 110 bpm is bradycardia, and above 160 bpm is tachycardia.
Tachycardia and bradycardia indicate changes in the baseline rate.
Causes of tachycardia include maternal fever, sympathomimetics, and fetal arrhythmia. Tachycardia with a loss in variability and recurrent late or variable decelerations may indicate fetal acidemia.
In contrast, bradycardia ranging between 80 and 110 bpm does not indicate fetal hypoxia if the variability is retained, but a rate less than 80 bpm may indicate fetal hypoxia. The fetus may demonstrate terminal bradycardia associated with prolonged head or cord compression, but may tolerate this as long as variability is maintained. Sinusoidal patterns are excluded.
Decelerations are quantified by depth of the nadir (below the baseline) in bpm, with the duration measured in minutes or seconds from the beginning to the end of the deceleration (FIGURES 1 and 2). Those that occur more than 50% of the time are recurrent or periodic.
Acceleration is an abrupt change above baseline. Before 32 weeks, accelerations are defined as 10 or more bpm over baseline for 10 seconds or more. Peak acceleration is a minimum of 15 bpm above baseline, with a duration ranging from 15 seconds to 2 minutes before return to baseline. A prolonged acceleration continues for more than 2 minutes; if it lasts more than 10 minutes, it is considered a change in baseline.
Periodic and episodic patterns. Periodic patterns include early, late, and variable decelerations associated with uterine contractions. Episodic patterns are those not associated with uterine contractions.
A late deceleration is a visually apparent gradual decrease below the baseline for 30 seconds (at the nadir) that is delayed in timing relative to the uterine contraction. The recovery also is delayed relative to the end of the uterine contraction. Late decelerations that maintain variability are thought to indicate a well-oxygenated fetus and are neurogenic in origin,11 but late decelerations without variability are thought to represent possible asphyxia. In fetuses with prolonged placental insufficiency, these smooth late decelerations are thought to reflect decreased cerebral and myocardial function.
Early decelerations are visually apparent decreases of 30 seconds (to the nadir) that start and end with a uterine contraction. They are not associated with significant fetal acidemia.
Variable decelerations are abrupt and visually apparent decelerations of 15 bpm below the baseline; they last more than 15 seconds to the nadir and less than 2 minutes overall. Variable decelerations are generally due to head compression, vagal stimulation, or cord compression. When variable decelerations are persistent and severe, tachycardia, delayed return to baseline, or decreased variability may occur, reflecting fetal acidemia.12
Prolonged late and severe variable decelerations may lead to fetal hypoxia. A duration between 2 and 10 minutes indicates a prolonged deceleration. A change of 10 minutes or more indicates a change in baseline.
Absent or severe variability with persistent late and prolonged decelerations is generally believed to be ominous and may correlate with hypoxia of such severity that the fetal central nervous system may have already been damaged. Surrogate markers for intrapartum asphyxia include mixed acidemia at birth, low Apgar score (less than 3 at 5 minutes), seizures within 24 hours, and multiorgan dysfunction.
High-risk pattern. Fetuses at greatest risk exhibit marked bradycardia, recurrent late and variable decelerations, and absent variability.
Normal pattern. In contrast, a normal baseline with moderate variability and accelerations and absence of periodic patterns (decelerations) is predictive of fetal well-being.
Tips on interpretation. Deciphering EFM patterns entails making observations over time. Many patterns acquire increased significance when there is a trend toward persistent, significant departures from baseline with decreased variability, loss of accelerations, or persistent episodic or periodic decelerations, particularly with loss of variability.
FIGURE 2 Severe deceleration
A severe deceleration with variability. When variable decelerations are persistent and severe, fetal acidemia may be present.
Human factors
Agreement between observers. The reliability and reproducibility of EFM interpretation have been reviewed, but prospective studies to evaluate them are lacking. Interobserver agreement is reasonably high for the baseline rate, accelerations, and decelerations, but lower for variable decelerations and lower still for variability. Computers have been investigated for standardizing EFM interpretation.
Competency of physicians and nurses. A study14 assessing competency in the evaluation of EFM tracings surveyed 43 Ob/Gyn training programs.
Researchers found that the majority (79%) use clinical experience along with structured lectures (87%) to train residents and fellows. Perinatal morbidity and mortality conferences were used by 85% of programs, and maintenance of EFM skills involved both clinical experience and case studies.
Although registered nurses typically undergo annual competency evaluation in EFM, physician competency is not formally evaluated. Thus, some other mechanism for ensuring expertise would be helpful.
Unreliable link to outcomes
The correlation between FHR patterns and neonatal outcomes is difficult to establish. The causative event in 70% of neonatal encephalopathy cases is believed to have occurred before the onset of labor13; therefore, few cases of cerebral palsy and neonatal encephalopathy are amenable to interventions during labor. Abnormal patterns that do not allow sufficient time for intervention also may have poor outcomes.
Fetuses that have intrapartum asphyxia usually have abnormal heart rate patterns. However, most women with abnormal FHR patterns give birth to neonates with normal Apgar scores and outcomes.
The central tenet of the 2003 monograph13 by the American College of Obstetricians and Gynecologists is that in order for an intrapartum event to progress to cerebral palsy, the pathway must proceed through neonatal encephalopathy. However, only 30% of neonatal encephalopathy cases are due to events during labor. Further, epilepsy, mental retardation, and attention deficit disorder are not caused by birth asphyxia.
A 99% false-positive rate may result when nonreassuring EFM tracings are used to predict cerebral palsy. For this reason, there is still considerable disagreement about whether EFM can reduce the incidence of cerebral palsy. Using the criteria for establishing acute hypoxic-ischemic insult (TABLE 1) and for determining that intrapartum events were sufficient to cause cerebral palsy (TABLE 2), only 9% of cerebral palsy cases are attributable to possible birth asphyxia.11
Most cases of catastrophic hypoxia ultimately lead to fetal or neonatal death; a minority of infants survive and develop cerebral palsy. The pattern of neurologic injury following acute catastrophic hypoxic-ischemic insult can involve the basal ganglia and thalami; it may be different from the pattern that commonly follows chronic insult, which involves primarily the cerebral cortex and subcortical white matter.
TABLE 1
Criteria for establishing acute hypoxic-ischemic insult
| Metabolic acidosis evident in fetal umbilical cord arterial blood at delivery (pH <7.0 or base excess ≥ 12 mmol/L) |
| Early onset of severe or moderate neonatal encephalopathy in infants delivered at ≥ 34 weeks’ gestation |
| Cerebral palsy of the spastic quadriplegic or dyskinetic type |
| Exclusion of other identifiable causes |
| Data from American College of Obstetricians and Gynecologists13 |
TABLE 2
Criteria to establish intrapartum event as cause of cerebral palsy
| Sentinel hypoxic event occurring immediately before or during labor |
| Sudden sustained fetal bradycardia or absence of fetal heart rate variability in presence of persistent late or variable decelerations |
| Apgar score of less than 3 at 5 minutes |
| Onset of multisystem involvement within 72 hours of birth |
| Early imaging studies showing evidence of acute nonfocal abnormality. |
| Data from American College of Obstetricians and Gynecologists13 |
Little time to react during labor
Severe intrapartum hypoxic-ischemic insult may be detected by EFM but, in some cases, the reaction time to prevent fetal brain injury is very short. These intrapartum events include umbilical cord prolapse, shoulder dystocia, uterine rupture, and maternal cardiopulmonary arrest. With uterine rupture, asphyxia may occur as quickly as 10 minutes after onset of bradycardia when the prolonged deceleration is preceded by severe late decelerations of 30 to 90 minutes. The average time from onset of prolonged bradycardia has been reported as 13±6.5 minutes.13
The 30-minute rule. Because mortality and morbidity may depend on how much time elapses from onset of the catastrophic event to delivery, ACOG developed guidelines for timing emergent cesarean. The 30-minute rule13 advises that a cesarean be performed within 30 minutes of the decision to proceed. Unfortunately, in many cases, acute hypoxic-ischemic insult occurs in less than 30 minutes.
Many hospitals and obstetric services no longer offer vaginal birth after cesarean (VBAC) if they cannot guarantee that anesthesia, nursing, pediatric, and operating room services will be available within this time frame. However, other causes of sudden hypoxia—besides the small chance of uterine rupture with VBAC—may be neither predictable nor preventable.
General recommendations
In cases of nonreassuring FHR tracings, the options include maternal positioning to achieve uterine displacement; hydration; correction of hypotension, which may be useful when regional anesthetics are used; tocolytic use for treating a uterine contraction; maternal oxygen; amnio-infusion for thick meconium in the presence of a nonreassuring FHR tracing; and assessment and correction of the oxytocin dose when augmentation or induction of labor is occurring.
The authors report no financial relationships relevant to this article.
- High-risk pattern. Fetuses at greatest risk include those with marked bradycardia, recurrent late and variable decelerations, and absent variability.
- Normal pattern. In contrast, a normal baseline rate with normal, moderate variability and accelerations, and absence of periodic patterns (decelerations) predicts fetal well-being.
- Tips on interpretation. Deciphering fetal heart rate patterns entails making observations over time. Many patterns acquire increased significance when there is a trend toward persistent, significant departures from baseline with decreased variability, loss of accelerations, or persistent episodic or periodic decelerations, particularly with loss of variability.
True or false: Electronic fetal monitoring reduces the incidence of cerebral palsy and infant morbidity and mortality.
Unfortunately, the statement is false, although patients continue to believe it is true. As a result, a large proportion of obstetrics liability cases center on electronic fetal monitoring (EFM): 43% of all lawsuits alleging obstetric malpractice, 52% of cases involving a stillborn fetus or neonatal death, and 66% of cases involving a neurologically impaired infant.1
Although the reliability and validity of EFM in reducing perinatal morbidity and mortality leave much to be desired, they could be greatly improved by increasing our understanding of the physiologic causes of patterns, establishing formal definitions for the various fetal heart rate (FHR) patterns, and developing specific recommendations for “abnormal” patterns.
Once these goals are attained, the question of whether EFM can be used to prevent asphyxial brain damage can be more adequately addressed.
This article discusses the following topics:
- EFM patterns and their importance,
- key research findings to date, and
- how to identify patients at greatest risk of an adverse outcome.
Unfulfilled hopes based on flawed assumption
Evaluation of the FHR in labor—first by auscultation and then by continuous EFM—has long held promise of improved outcomes. As early as the 1800s, auscultated Electronic fetal monitoring: The difficulty of linking patterns with outcomes FHR decelerations and prolonged bradycardias were seen as signs of fetal distress in labor. When criteria were being developed for obstetric intervention with forceps in the late 1800s, tachycardia was included as an indication, building the foundation for contemporary definitions of nonreassuring FHR patterns.
Many decades later, abnormal FHR patterns correlated with low Apgar scores and neonatal death (although the correlation between certain patterns and poor outcomes was weak) in the findings of the American Collaborative Perinatal Study,2 which reviewed 25,000 births monitored using intermittent auscultation from 1959 through 1965.
The largely flawed assumption that most cases of cerebral palsy are caused by intrapartum asphyxia led to a concerted effort to decrease the incidence of asphyxia through EFM. By the 1970s, the expectation was that EFM would decrease perinatal morbidity and mortality.
Since its introduction into clinical practice in the early 1970s, EFM has become increasingly common; its use grew from 61% of women giving birth in 1989 to 84% in 1998.3
Findings of randomized controlled trials. The first randomized controlled trial4 of EFM in labor, in 1976, demonstrated no improvement in outcome but a significant increase in surgical births; later trials showed no decrease in perinatal mortality or cerebral palsy.
The 1985 Dublin trial,5 which scored the highest in study design in a later meta-analysis, showed a slight protective effect of EFM overall.
A 1995 meta-analysis6 of 12 randomized controlled trials encompassing 58,555 pregnancies identified 9 outcomes, including Apgar scores, perinatal mortality, and neonatal seizures. EFM had a consistent impact only on neonatal seizures, and had no measurable effect on morbidity and mortality. Overall, 1.1% of neonates in the auscultated group had seizures, compared with 0.8% in the EFM group. Operative deliveries also increased in the EFM group.
EFM effects on neonatal outcomes. Freeman7 summarized the gains of EFM from 1972 to 1990 as follows:
- no effect on cerebral palsy,
- no effect on neonatal morbidity, and
- no improvement in neurologic outcomes in premature infants.
In 1995, Vintzileos and colleagues8 studied fetal acidemia in 680 auscultated labors and compared them with 739 monitored by EFM, using umbilical artery pH of less than 7.15 as an outcome measure. EFM was superior to auscultation, with better sensitivity and higher positive and negative predictive value.
Identification of patterns still rests on visual interpretation
In 1997, after a workshop to develop standardized, unambiguous definitions for fetal heart tracings, the National Institute for Child Health and Human Development (NICHD) issued guidelines for interpreting EFM9; these focus on visual interpretation, though computerized analyses are being developed. The definitions emphasize intrapartum evaluation, and the major patterns are categorized as baseline, periodic, or episodic.
Variability is defined by amplitude and includes short-term (beat to beat) and long-term variability, with no distinction between the two. Sinusoidal patterns are excluded from the definition.
According to NICHD guidelines, variabilities can be defined as follows:
- amplitude undetectable = absent variability;
- amplitude of 0 to 5 beats per minute (bpm) = minimal variability;
- 5 to 25 bpm = moderate variability; and
- amplitude exceeding 25 bpm = marked variability.
Loss of variability is the most sensitive indicator of fetal acidemia, cerebral asphyxia, and myocardial depression, when it is associated with other periodic patterns.10
Baseline rate. Baseline heart rate is the approximate mean rounded to 5 bpm during a 10-minute interval, excluding periodic or episodic changes, periods of marked variability, and segments of the baseline that differ by more than 25 bpm. The baseline must continue for 2 minutes during that 10-minute period or it is considered indeterminate. A normal baseline generally ranges from 110 to 160 bpm. Below 110 bpm is bradycardia, and above 160 bpm is tachycardia.
Tachycardia and bradycardia indicate changes in the baseline rate.
Causes of tachycardia include maternal fever, sympathomimetics, and fetal arrhythmia. Tachycardia with a loss in variability and recurrent late or variable decelerations may indicate fetal acidemia.
In contrast, bradycardia ranging between 80 and 110 bpm does not indicate fetal hypoxia if the variability is retained, but a rate less than 80 bpm may indicate fetal hypoxia. The fetus may demonstrate terminal bradycardia associated with prolonged head or cord compression, but may tolerate this as long as variability is maintained. Sinusoidal patterns are excluded.
Decelerations are quantified by depth of the nadir (below the baseline) in bpm, with the duration measured in minutes or seconds from the beginning to the end of the deceleration (FIGURES 1 and 2). Those that occur more than 50% of the time are recurrent or periodic.
Acceleration is an abrupt change above baseline. Before 32 weeks, accelerations are defined as 10 or more bpm over baseline for 10 seconds or more. Peak acceleration is a minimum of 15 bpm above baseline, with a duration ranging from 15 seconds to 2 minutes before return to baseline. A prolonged acceleration continues for more than 2 minutes; if it lasts more than 10 minutes, it is considered a change in baseline.
Periodic and episodic patterns. Periodic patterns include early, late, and variable decelerations associated with uterine contractions. Episodic patterns are those not associated with uterine contractions.
A late deceleration is a visually apparent gradual decrease below the baseline for 30 seconds (at the nadir) that is delayed in timing relative to the uterine contraction. The recovery also is delayed relative to the end of the uterine contraction. Late decelerations that maintain variability are thought to indicate a well-oxygenated fetus and are neurogenic in origin,11 but late decelerations without variability are thought to represent possible asphyxia. In fetuses with prolonged placental insufficiency, these smooth late decelerations are thought to reflect decreased cerebral and myocardial function.
Early decelerations are visually apparent decreases of 30 seconds (to the nadir) that start and end with a uterine contraction. They are not associated with significant fetal acidemia.
Variable decelerations are abrupt and visually apparent decelerations of 15 bpm below the baseline; they last more than 15 seconds to the nadir and less than 2 minutes overall. Variable decelerations are generally due to head compression, vagal stimulation, or cord compression. When variable decelerations are persistent and severe, tachycardia, delayed return to baseline, or decreased variability may occur, reflecting fetal acidemia.12
Prolonged late and severe variable decelerations may lead to fetal hypoxia. A duration between 2 and 10 minutes indicates a prolonged deceleration. A change of 10 minutes or more indicates a change in baseline.
Absent or severe variability with persistent late and prolonged decelerations is generally believed to be ominous and may correlate with hypoxia of such severity that the fetal central nervous system may have already been damaged. Surrogate markers for intrapartum asphyxia include mixed acidemia at birth, low Apgar score (less than 3 at 5 minutes), seizures within 24 hours, and multiorgan dysfunction.
High-risk pattern. Fetuses at greatest risk exhibit marked bradycardia, recurrent late and variable decelerations, and absent variability.
Normal pattern. In contrast, a normal baseline with moderate variability and accelerations and absence of periodic patterns (decelerations) is predictive of fetal well-being.
Tips on interpretation. Deciphering EFM patterns entails making observations over time. Many patterns acquire increased significance when there is a trend toward persistent, significant departures from baseline with decreased variability, loss of accelerations, or persistent episodic or periodic decelerations, particularly with loss of variability.
FIGURE 2 Severe deceleration
A severe deceleration with variability. When variable decelerations are persistent and severe, fetal acidemia may be present.
Human factors
Agreement between observers. The reliability and reproducibility of EFM interpretation have been reviewed, but prospective studies to evaluate them are lacking. Interobserver agreement is reasonably high for the baseline rate, accelerations, and decelerations, but lower for variable decelerations and lower still for variability. Computers have been investigated for standardizing EFM interpretation.
Competency of physicians and nurses. A study14 assessing competency in the evaluation of EFM tracings surveyed 43 Ob/Gyn training programs.
Researchers found that the majority (79%) use clinical experience along with structured lectures (87%) to train residents and fellows. Perinatal morbidity and mortality conferences were used by 85% of programs, and maintenance of EFM skills involved both clinical experience and case studies.
Although registered nurses typically undergo annual competency evaluation in EFM, physician competency is not formally evaluated. Thus, some other mechanism for ensuring expertise would be helpful.
Unreliable link to outcomes
The correlation between FHR patterns and neonatal outcomes is difficult to establish. The causative event in 70% of neonatal encephalopathy cases is believed to have occurred before the onset of labor13; therefore, few cases of cerebral palsy and neonatal encephalopathy are amenable to interventions during labor. Abnormal patterns that do not allow sufficient time for intervention also may have poor outcomes.
Fetuses that have intrapartum asphyxia usually have abnormal heart rate patterns. However, most women with abnormal FHR patterns give birth to neonates with normal Apgar scores and outcomes.
The central tenet of the 2003 monograph13 by the American College of Obstetricians and Gynecologists is that in order for an intrapartum event to progress to cerebral palsy, the pathway must proceed through neonatal encephalopathy. However, only 30% of neonatal encephalopathy cases are due to events during labor. Further, epilepsy, mental retardation, and attention deficit disorder are not caused by birth asphyxia.
A 99% false-positive rate may result when nonreassuring EFM tracings are used to predict cerebral palsy. For this reason, there is still considerable disagreement about whether EFM can reduce the incidence of cerebral palsy. Using the criteria for establishing acute hypoxic-ischemic insult (TABLE 1) and for determining that intrapartum events were sufficient to cause cerebral palsy (TABLE 2), only 9% of cerebral palsy cases are attributable to possible birth asphyxia.11
Most cases of catastrophic hypoxia ultimately lead to fetal or neonatal death; a minority of infants survive and develop cerebral palsy. The pattern of neurologic injury following acute catastrophic hypoxic-ischemic insult can involve the basal ganglia and thalami; it may be different from the pattern that commonly follows chronic insult, which involves primarily the cerebral cortex and subcortical white matter.
TABLE 1
Criteria for establishing acute hypoxic-ischemic insult
| Metabolic acidosis evident in fetal umbilical cord arterial blood at delivery (pH <7.0 or base excess ≥ 12 mmol/L) |
| Early onset of severe or moderate neonatal encephalopathy in infants delivered at ≥ 34 weeks’ gestation |
| Cerebral palsy of the spastic quadriplegic or dyskinetic type |
| Exclusion of other identifiable causes |
| Data from American College of Obstetricians and Gynecologists13 |
TABLE 2
Criteria to establish intrapartum event as cause of cerebral palsy
| Sentinel hypoxic event occurring immediately before or during labor |
| Sudden sustained fetal bradycardia or absence of fetal heart rate variability in presence of persistent late or variable decelerations |
| Apgar score of less than 3 at 5 minutes |
| Onset of multisystem involvement within 72 hours of birth |
| Early imaging studies showing evidence of acute nonfocal abnormality. |
| Data from American College of Obstetricians and Gynecologists13 |
Little time to react during labor
Severe intrapartum hypoxic-ischemic insult may be detected by EFM but, in some cases, the reaction time to prevent fetal brain injury is very short. These intrapartum events include umbilical cord prolapse, shoulder dystocia, uterine rupture, and maternal cardiopulmonary arrest. With uterine rupture, asphyxia may occur as quickly as 10 minutes after onset of bradycardia when the prolonged deceleration is preceded by severe late decelerations of 30 to 90 minutes. The average time from onset of prolonged bradycardia has been reported as 13±6.5 minutes.13
The 30-minute rule. Because mortality and morbidity may depend on how much time elapses from onset of the catastrophic event to delivery, ACOG developed guidelines for timing emergent cesarean. The 30-minute rule13 advises that a cesarean be performed within 30 minutes of the decision to proceed. Unfortunately, in many cases, acute hypoxic-ischemic insult occurs in less than 30 minutes.
Many hospitals and obstetric services no longer offer vaginal birth after cesarean (VBAC) if they cannot guarantee that anesthesia, nursing, pediatric, and operating room services will be available within this time frame. However, other causes of sudden hypoxia—besides the small chance of uterine rupture with VBAC—may be neither predictable nor preventable.
General recommendations
In cases of nonreassuring FHR tracings, the options include maternal positioning to achieve uterine displacement; hydration; correction of hypotension, which may be useful when regional anesthetics are used; tocolytic use for treating a uterine contraction; maternal oxygen; amnio-infusion for thick meconium in the presence of a nonreassuring FHR tracing; and assessment and correction of the oxytocin dose when augmentation or induction of labor is occurring.
The authors report no financial relationships relevant to this article.
1. American College of Obstetricians and Gynecologists. 1999 Survey of Professional Liability: Claims Data Tabulations. Washington, DC: ACOG; 1999.
2. Benson RC, Schubeck F, Deutschberger J, et al. Fetal heart rate as a predictor of fetal distress: a report from the collaborative project. Obstet Gynecol. 1968;32:259-266.
3. Curtin SC, Matthews TJ. US obstetric procedures, 1998. Birth. 2000;27:136-138.
4. Havercamp AD, Thompson HE, McFee JG, Cetrulo C. The evaluation of continuous fetal heart rate monitoring in high-risk pregnancy. Am J Obstet Gynecol. 1976;125(3):310-320.
5. MacDonald D, Grant A, Sheridan-Pereira M, et al. The Dublin randomized control trial of intrapartum fetal heart rate monitoring. Am J Obstet Gynecol. 1985;152:524-529.
6. Thacker SB, Stroup DF, Peterson HB. Efficacy and safety of intrapartum electronic fetal monitoring: an update. Obstet Gynecol. 1995;86:613-620.
7. Freeman RK. Intrapartum electronic fetal monitoring—a disappointing story. N Engl J Med. 1990;322:624-626.
8. Vintzileos AM, Nochimson DJ, Antsaklis A, Varvarigos I, Guzman ER, Knuppel RA. Comparison of intrapartum electronic fetal heart rate monitoring versus intermittent auscultation in detecting fetal acidemia at birth. Am J Obstet Gynecol. 1995;173:1021-1024.
9. National Institute of Child Health and Human Development Research Planning Workshop. Electronic fetal heart rate monitoring: research guidelines for interpretation. Am J Obstet Gynecol. 1997;177:1385-1390.
10. Court DJ, Parer JT. Experimental studies in fetal asphyxial heart rate interpretation. In: Nathanielsz PW, Parer JT, eds. Research in Perinatal Medicine. Ithaca, NY: Perinatology Press; 1985;114-167.
11. Paul RH, Suidan AH, Yen S, et al. Clinical fetal monitoring III: the evaluation and significance of intrapartum fetal heart rate variability. Am J Obstet Gynecol. 1975;123:206-210.
12. Ball RH, Parer JT. The physiologic mechanism of variable decelerations. AmJ Obstet Gynecol. 1992;166:1683-1689.
13. American College of Obstetricians and Gynecologists. Neonatal Encephalopathy and Cerebral Palsy: Defining the Pathogenesis and Pathophysiology. Washington, DC: ACOG; 2003.
14. Murphy AA, Halamek LP, Lyell DJ, et al. Training and competency assessment in electronic fetal monitoring: a national survey. Obstet Gynecol. 2003;101:1243-1248.
1. American College of Obstetricians and Gynecologists. 1999 Survey of Professional Liability: Claims Data Tabulations. Washington, DC: ACOG; 1999.
2. Benson RC, Schubeck F, Deutschberger J, et al. Fetal heart rate as a predictor of fetal distress: a report from the collaborative project. Obstet Gynecol. 1968;32:259-266.
3. Curtin SC, Matthews TJ. US obstetric procedures, 1998. Birth. 2000;27:136-138.
4. Havercamp AD, Thompson HE, McFee JG, Cetrulo C. The evaluation of continuous fetal heart rate monitoring in high-risk pregnancy. Am J Obstet Gynecol. 1976;125(3):310-320.
5. MacDonald D, Grant A, Sheridan-Pereira M, et al. The Dublin randomized control trial of intrapartum fetal heart rate monitoring. Am J Obstet Gynecol. 1985;152:524-529.
6. Thacker SB, Stroup DF, Peterson HB. Efficacy and safety of intrapartum electronic fetal monitoring: an update. Obstet Gynecol. 1995;86:613-620.
7. Freeman RK. Intrapartum electronic fetal monitoring—a disappointing story. N Engl J Med. 1990;322:624-626.
8. Vintzileos AM, Nochimson DJ, Antsaklis A, Varvarigos I, Guzman ER, Knuppel RA. Comparison of intrapartum electronic fetal heart rate monitoring versus intermittent auscultation in detecting fetal acidemia at birth. Am J Obstet Gynecol. 1995;173:1021-1024.
9. National Institute of Child Health and Human Development Research Planning Workshop. Electronic fetal heart rate monitoring: research guidelines for interpretation. Am J Obstet Gynecol. 1997;177:1385-1390.
10. Court DJ, Parer JT. Experimental studies in fetal asphyxial heart rate interpretation. In: Nathanielsz PW, Parer JT, eds. Research in Perinatal Medicine. Ithaca, NY: Perinatology Press; 1985;114-167.
11. Paul RH, Suidan AH, Yen S, et al. Clinical fetal monitoring III: the evaluation and significance of intrapartum fetal heart rate variability. Am J Obstet Gynecol. 1975;123:206-210.
12. Ball RH, Parer JT. The physiologic mechanism of variable decelerations. AmJ Obstet Gynecol. 1992;166:1683-1689.
13. American College of Obstetricians and Gynecologists. Neonatal Encephalopathy and Cerebral Palsy: Defining the Pathogenesis and Pathophysiology. Washington, DC: ACOG; 2003.
14. Murphy AA, Halamek LP, Lyell DJ, et al. Training and competency assessment in electronic fetal monitoring: a national survey. Obstet Gynecol. 2003;101:1243-1248.