User login
‘Tour de force’ study reveals therapeutic targets in 38% of cancer patients
The effort is the National Cancer Institute Molecular Analysis for Therapy Choice (NCI-MATCH) trial. For this study, researchers performed next-generation sequencing on tumor biopsy specimens to identify therapeutically actionable molecular alterations in patients with “underexplored” cancer types.
The trial included 5,954 patients with cancers that had progressed on standard treatments or rare cancers for which there is no standard treatment. If actionable alterations were found in these patients, they could receive new drugs in development that showed promise in other clinical trials or drugs that were approved by the Food and Drug Administration to treat at least one cancer type.
Data newly reported in the Journal of Clinical Oncology showed that 37.6% of patients had alterations that could be matched to targeted drugs, and 17.8% of patients were assigned to targeted treatment. Multiple actionable tumor mutations were seen in 11.9% of specimens, and resistance-conferring mutations were seen in 71.3% of specimens.
“The bottom line from this report is that next-generation sequencing is an efficient way to identify both approved and promising investigational therapies. For this reason, it should be considered standard of care for patients with advanced cancers,” said study chair Keith T. Flaherty, MD, director of the Henri and Belinda Termeer Center for Targeted Therapy at Massachusetts General Hospital Cancer Center in Boston.
“This study sets the benchmark for the ‘actionability’ of next-generation sequencing,” Dr. Flaherty added. “We expect this number [of actionable alterations] will continue to rise steadily as further advances are made in the development of therapies that target some of the genetic alterations for which we could not offer treatment options in NCI-MATCH.”
Relapsed/refractory vs. primary tumors
The NCI-MATCH researchers focused on the most commonly found genetic alterations and performed biopsies at study entry to provide the most accurate picture of the genetic landscape of relapsed/refractory cancer patients. That makes this cohort distinct from The Cancer Genome Atlas (TCGA), a database of patients with mostly untreated primary tumors, and other published cohorts that include genetic analysis of primary tumors and biopsies from the time of initial metastatic recurrence.
The researchers compared the tumor gene makeup of NCI-MATCH and TCGA patients with seven cancer types – breast, bile duct, cervix, colorectal, lung, pancreas, and prostate.
“Perhaps the biggest surprise was the relatively minimal change in the genetic alterations found in these relapsed/refractory patients, compared to primary tumors,” Dr. Flaherty said. “These findings suggest that it is very reasonable to perform next-generation sequencing at the time of initial metastatic cancer diagnosis and to rely on those findings for the purposes of considering FDA-approved therapies and clinical trial participation.”
Multiple alterations and resistance
The complex genetics of cancers has led researchers to explore combinations of targeted and other therapies to address multiple defects at the same time.
“Not surprisingly, the most common collision of multiple genetic alterations within the same tumor was in the commonly altered tumor suppressor genes: TP53, APC, and PTEN,” Dr. Flaherty said.
“An increasing body of evidence supports a role for loss-of-function alterations in these genes to confer resistance to many targeted therapies,” he added. “While we don’t have targeted therapies yet established to directly counter this form of therapeutic resistance, we hypothesize that various types of combination therapy may be able to indirectly undercut resistance and enhance the benefit of many targeted therapies.”
The NCI-MATCH researchers will continue to mine this large dataset to better understand the many small, genetically defined cancer subpopulations.
“We will continue to report the outcome of the individual treatment subprotocols, and combining this genetic analysis with those outcomes will likely inform the next clinical trials,” Dr. Flaherty said.
Actionable mutations make a difference
Precision oncology experts agree that NCI-MATCH results are impressive and add a fuller appreciation that actionable mutations make a clinical difference.
“This is a powerful, extremely well-designed study, a tour de force of collaborative science,” said Stephen Gruber, MD, PhD, director of the Center for Precision Medicine at City of Hope National Medical Center in Duarte, Calif.
“The future holds even more promise,” he added. “Our ability to interrogate the genomic landscape of cancer is improving rapidly. Tumor testing helps get the right drug to the right tumor faster than a guidelines-based approach from historical data of combination chemotherapy. This is a likely game changer for the way oncologists will practice in the future, especially as we learn more results of subset trials. The NCI-MATCH researchers have taken a laser-focused look at the current data, but we now know we can look far more comprehensively at genomic profiles of tumors.”
From the viewpoint of the practicing oncologist, co-occurring resistance mutations make a difference in defining what combinations are likely and, more importantly, less likely to be effective. “When we see two mutations and one is likely to confer resistance, we can make a choice to avoid a drug that is not likely to work,” Dr. Gruber said.
“The NCI-MATCH trial allows both approved and investigational agents, which expands the possibility of matching patients to newer agents. This is especially relevant if there are no FDA-approved drugs yet for some molecular aberrations,” said Lillian L. Siu, MD, a senior medical oncologist at the Princess Margaret Cancer Centre in Toronto. “This trial enables such evaluations under the auspice of a clinical trial to provide important knowledge.”
Both experts agree that in-depth biological interrogations of cancer will move the field of precision oncology forward. Dr. Gruber said that “studies have not yet fully addressed the power of germline genetic testing of DNA. Inherited susceptibility will drive therapeutic choices – for example, PARP inhibitors that access homologous recombination deficiency for breast, ovarian, and prostate cancer. We will learn more about treatment choices for those cancers.”
Dr. Siu added: “I truly believe that liquid biopsies [circulating tumor DNA] will help us perform target-drug matching in a less invasive way. We need to explore beyond the genome to look at the transcriptome, proteome, epigenome, and immunome, among others. It is likely that multiomic predictors are going to be able to identify more therapeutic options compared to single genomic predictors.”
Dr. Flaherty noted that all tumor samples from patients assigned to treatment are being subjected to whole-exome sequencing to further the discovery of the genetic features of responsive and nonresponsive tumors.
NCI-MATCH was funded by the National Cancer Institute. Dr. Flaherty disclosed relationships with Clovis Oncology, Loxo, X4 Pharma, and many other companies. His coauthors disclosed many conflicts as well. Dr. Gruber is cofounder of Brogent International. Dr. Siu disclosed relationships with Agios, Treadwell Therapeutics, Merck, Pfizer, and many other companies.
SOURCE: Flaherty KT et al. J Clin Oncol. 2020 Oct 13. doi: 10.1200/JCO.19.03010.
The effort is the National Cancer Institute Molecular Analysis for Therapy Choice (NCI-MATCH) trial. For this study, researchers performed next-generation sequencing on tumor biopsy specimens to identify therapeutically actionable molecular alterations in patients with “underexplored” cancer types.
The trial included 5,954 patients with cancers that had progressed on standard treatments or rare cancers for which there is no standard treatment. If actionable alterations were found in these patients, they could receive new drugs in development that showed promise in other clinical trials or drugs that were approved by the Food and Drug Administration to treat at least one cancer type.
Data newly reported in the Journal of Clinical Oncology showed that 37.6% of patients had alterations that could be matched to targeted drugs, and 17.8% of patients were assigned to targeted treatment. Multiple actionable tumor mutations were seen in 11.9% of specimens, and resistance-conferring mutations were seen in 71.3% of specimens.
“The bottom line from this report is that next-generation sequencing is an efficient way to identify both approved and promising investigational therapies. For this reason, it should be considered standard of care for patients with advanced cancers,” said study chair Keith T. Flaherty, MD, director of the Henri and Belinda Termeer Center for Targeted Therapy at Massachusetts General Hospital Cancer Center in Boston.
“This study sets the benchmark for the ‘actionability’ of next-generation sequencing,” Dr. Flaherty added. “We expect this number [of actionable alterations] will continue to rise steadily as further advances are made in the development of therapies that target some of the genetic alterations for which we could not offer treatment options in NCI-MATCH.”
Relapsed/refractory vs. primary tumors
The NCI-MATCH researchers focused on the most commonly found genetic alterations and performed biopsies at study entry to provide the most accurate picture of the genetic landscape of relapsed/refractory cancer patients. That makes this cohort distinct from The Cancer Genome Atlas (TCGA), a database of patients with mostly untreated primary tumors, and other published cohorts that include genetic analysis of primary tumors and biopsies from the time of initial metastatic recurrence.
The researchers compared the tumor gene makeup of NCI-MATCH and TCGA patients with seven cancer types – breast, bile duct, cervix, colorectal, lung, pancreas, and prostate.
“Perhaps the biggest surprise was the relatively minimal change in the genetic alterations found in these relapsed/refractory patients, compared to primary tumors,” Dr. Flaherty said. “These findings suggest that it is very reasonable to perform next-generation sequencing at the time of initial metastatic cancer diagnosis and to rely on those findings for the purposes of considering FDA-approved therapies and clinical trial participation.”
Multiple alterations and resistance
The complex genetics of cancers has led researchers to explore combinations of targeted and other therapies to address multiple defects at the same time.
“Not surprisingly, the most common collision of multiple genetic alterations within the same tumor was in the commonly altered tumor suppressor genes: TP53, APC, and PTEN,” Dr. Flaherty said.
“An increasing body of evidence supports a role for loss-of-function alterations in these genes to confer resistance to many targeted therapies,” he added. “While we don’t have targeted therapies yet established to directly counter this form of therapeutic resistance, we hypothesize that various types of combination therapy may be able to indirectly undercut resistance and enhance the benefit of many targeted therapies.”
The NCI-MATCH researchers will continue to mine this large dataset to better understand the many small, genetically defined cancer subpopulations.
“We will continue to report the outcome of the individual treatment subprotocols, and combining this genetic analysis with those outcomes will likely inform the next clinical trials,” Dr. Flaherty said.
Actionable mutations make a difference
Precision oncology experts agree that NCI-MATCH results are impressive and add a fuller appreciation that actionable mutations make a clinical difference.
“This is a powerful, extremely well-designed study, a tour de force of collaborative science,” said Stephen Gruber, MD, PhD, director of the Center for Precision Medicine at City of Hope National Medical Center in Duarte, Calif.
“The future holds even more promise,” he added. “Our ability to interrogate the genomic landscape of cancer is improving rapidly. Tumor testing helps get the right drug to the right tumor faster than a guidelines-based approach from historical data of combination chemotherapy. This is a likely game changer for the way oncologists will practice in the future, especially as we learn more results of subset trials. The NCI-MATCH researchers have taken a laser-focused look at the current data, but we now know we can look far more comprehensively at genomic profiles of tumors.”
From the viewpoint of the practicing oncologist, co-occurring resistance mutations make a difference in defining what combinations are likely and, more importantly, less likely to be effective. “When we see two mutations and one is likely to confer resistance, we can make a choice to avoid a drug that is not likely to work,” Dr. Gruber said.
“The NCI-MATCH trial allows both approved and investigational agents, which expands the possibility of matching patients to newer agents. This is especially relevant if there are no FDA-approved drugs yet for some molecular aberrations,” said Lillian L. Siu, MD, a senior medical oncologist at the Princess Margaret Cancer Centre in Toronto. “This trial enables such evaluations under the auspice of a clinical trial to provide important knowledge.”
Both experts agree that in-depth biological interrogations of cancer will move the field of precision oncology forward. Dr. Gruber said that “studies have not yet fully addressed the power of germline genetic testing of DNA. Inherited susceptibility will drive therapeutic choices – for example, PARP inhibitors that access homologous recombination deficiency for breast, ovarian, and prostate cancer. We will learn more about treatment choices for those cancers.”
Dr. Siu added: “I truly believe that liquid biopsies [circulating tumor DNA] will help us perform target-drug matching in a less invasive way. We need to explore beyond the genome to look at the transcriptome, proteome, epigenome, and immunome, among others. It is likely that multiomic predictors are going to be able to identify more therapeutic options compared to single genomic predictors.”
Dr. Flaherty noted that all tumor samples from patients assigned to treatment are being subjected to whole-exome sequencing to further the discovery of the genetic features of responsive and nonresponsive tumors.
NCI-MATCH was funded by the National Cancer Institute. Dr. Flaherty disclosed relationships with Clovis Oncology, Loxo, X4 Pharma, and many other companies. His coauthors disclosed many conflicts as well. Dr. Gruber is cofounder of Brogent International. Dr. Siu disclosed relationships with Agios, Treadwell Therapeutics, Merck, Pfizer, and many other companies.
SOURCE: Flaherty KT et al. J Clin Oncol. 2020 Oct 13. doi: 10.1200/JCO.19.03010.
The effort is the National Cancer Institute Molecular Analysis for Therapy Choice (NCI-MATCH) trial. For this study, researchers performed next-generation sequencing on tumor biopsy specimens to identify therapeutically actionable molecular alterations in patients with “underexplored” cancer types.
The trial included 5,954 patients with cancers that had progressed on standard treatments or rare cancers for which there is no standard treatment. If actionable alterations were found in these patients, they could receive new drugs in development that showed promise in other clinical trials or drugs that were approved by the Food and Drug Administration to treat at least one cancer type.
Data newly reported in the Journal of Clinical Oncology showed that 37.6% of patients had alterations that could be matched to targeted drugs, and 17.8% of patients were assigned to targeted treatment. Multiple actionable tumor mutations were seen in 11.9% of specimens, and resistance-conferring mutations were seen in 71.3% of specimens.
“The bottom line from this report is that next-generation sequencing is an efficient way to identify both approved and promising investigational therapies. For this reason, it should be considered standard of care for patients with advanced cancers,” said study chair Keith T. Flaherty, MD, director of the Henri and Belinda Termeer Center for Targeted Therapy at Massachusetts General Hospital Cancer Center in Boston.
“This study sets the benchmark for the ‘actionability’ of next-generation sequencing,” Dr. Flaherty added. “We expect this number [of actionable alterations] will continue to rise steadily as further advances are made in the development of therapies that target some of the genetic alterations for which we could not offer treatment options in NCI-MATCH.”
Relapsed/refractory vs. primary tumors
The NCI-MATCH researchers focused on the most commonly found genetic alterations and performed biopsies at study entry to provide the most accurate picture of the genetic landscape of relapsed/refractory cancer patients. That makes this cohort distinct from The Cancer Genome Atlas (TCGA), a database of patients with mostly untreated primary tumors, and other published cohorts that include genetic analysis of primary tumors and biopsies from the time of initial metastatic recurrence.
The researchers compared the tumor gene makeup of NCI-MATCH and TCGA patients with seven cancer types – breast, bile duct, cervix, colorectal, lung, pancreas, and prostate.
“Perhaps the biggest surprise was the relatively minimal change in the genetic alterations found in these relapsed/refractory patients, compared to primary tumors,” Dr. Flaherty said. “These findings suggest that it is very reasonable to perform next-generation sequencing at the time of initial metastatic cancer diagnosis and to rely on those findings for the purposes of considering FDA-approved therapies and clinical trial participation.”
Multiple alterations and resistance
The complex genetics of cancers has led researchers to explore combinations of targeted and other therapies to address multiple defects at the same time.
“Not surprisingly, the most common collision of multiple genetic alterations within the same tumor was in the commonly altered tumor suppressor genes: TP53, APC, and PTEN,” Dr. Flaherty said.
“An increasing body of evidence supports a role for loss-of-function alterations in these genes to confer resistance to many targeted therapies,” he added. “While we don’t have targeted therapies yet established to directly counter this form of therapeutic resistance, we hypothesize that various types of combination therapy may be able to indirectly undercut resistance and enhance the benefit of many targeted therapies.”
The NCI-MATCH researchers will continue to mine this large dataset to better understand the many small, genetically defined cancer subpopulations.
“We will continue to report the outcome of the individual treatment subprotocols, and combining this genetic analysis with those outcomes will likely inform the next clinical trials,” Dr. Flaherty said.
Actionable mutations make a difference
Precision oncology experts agree that NCI-MATCH results are impressive and add a fuller appreciation that actionable mutations make a clinical difference.
“This is a powerful, extremely well-designed study, a tour de force of collaborative science,” said Stephen Gruber, MD, PhD, director of the Center for Precision Medicine at City of Hope National Medical Center in Duarte, Calif.
“The future holds even more promise,” he added. “Our ability to interrogate the genomic landscape of cancer is improving rapidly. Tumor testing helps get the right drug to the right tumor faster than a guidelines-based approach from historical data of combination chemotherapy. This is a likely game changer for the way oncologists will practice in the future, especially as we learn more results of subset trials. The NCI-MATCH researchers have taken a laser-focused look at the current data, but we now know we can look far more comprehensively at genomic profiles of tumors.”
From the viewpoint of the practicing oncologist, co-occurring resistance mutations make a difference in defining what combinations are likely and, more importantly, less likely to be effective. “When we see two mutations and one is likely to confer resistance, we can make a choice to avoid a drug that is not likely to work,” Dr. Gruber said.
“The NCI-MATCH trial allows both approved and investigational agents, which expands the possibility of matching patients to newer agents. This is especially relevant if there are no FDA-approved drugs yet for some molecular aberrations,” said Lillian L. Siu, MD, a senior medical oncologist at the Princess Margaret Cancer Centre in Toronto. “This trial enables such evaluations under the auspice of a clinical trial to provide important knowledge.”
Both experts agree that in-depth biological interrogations of cancer will move the field of precision oncology forward. Dr. Gruber said that “studies have not yet fully addressed the power of germline genetic testing of DNA. Inherited susceptibility will drive therapeutic choices – for example, PARP inhibitors that access homologous recombination deficiency for breast, ovarian, and prostate cancer. We will learn more about treatment choices for those cancers.”
Dr. Siu added: “I truly believe that liquid biopsies [circulating tumor DNA] will help us perform target-drug matching in a less invasive way. We need to explore beyond the genome to look at the transcriptome, proteome, epigenome, and immunome, among others. It is likely that multiomic predictors are going to be able to identify more therapeutic options compared to single genomic predictors.”
Dr. Flaherty noted that all tumor samples from patients assigned to treatment are being subjected to whole-exome sequencing to further the discovery of the genetic features of responsive and nonresponsive tumors.
NCI-MATCH was funded by the National Cancer Institute. Dr. Flaherty disclosed relationships with Clovis Oncology, Loxo, X4 Pharma, and many other companies. His coauthors disclosed many conflicts as well. Dr. Gruber is cofounder of Brogent International. Dr. Siu disclosed relationships with Agios, Treadwell Therapeutics, Merck, Pfizer, and many other companies.
SOURCE: Flaherty KT et al. J Clin Oncol. 2020 Oct 13. doi: 10.1200/JCO.19.03010.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Lower BP and better tumor control with drug combo?
It’s not ready for the clinic, but new research suggests that angiotensin receptor II blockers (ARBs) widely used to treat hypertension may improve responses to cancer immunotherapy agents targeted against the programmed death-1/ligand-1 (PD-1/PD-L1) pathway.
That conclusion comes from an observational study of 597 patients with more than 3 dozen different cancer types treated in clinical trials at the US National Institutes of Health. Investigators found that both objective response rates and 3-year overall survival (OS) rates were significantly higher for patients treated with a PD-1/PD-L1 inhibitor who were on ARBs, compared with patients who weren’t taking the antihypertensive agents.
An association was also seen between higher ORR and OS rates for patients taking ACE inhibitors, but it was not statistically significant, reported Julius Strauss, MD, from the Center for Cancer Research at the National Cancer Institute in Bethesda, Md.
All study patients received PD-1/PD-L1 inhibitors, and the ORR for patients treated with ARBs was 33.8%, compared with 19.5% for those treated with ACE inhibitors, and 17% for those who took neither drug. The respective complete response (CR) rates were 11.3%, 3.7%, and 3.1%.
Strauss discussed the data during an online briefing prior to his presentation of the findings during the 32nd EORTC-NCI-AACR Symposium on Molecular Targets and Cancer Therapeutics, which is taking place virtually.
Several early studies have suggested that angiotensin II, in addition to its effect on blood pressure, can also affect cancer growth by leading to downstream production of two proteins: vascular endothelial growth factor (VEGF) and transforming growth factor–beta (TGF-beta), he explained.
“Both of these [proteins] have been linked to cancer growth and cancer resistance to immune system attack,” Strauss observed.
He also discussed the mechanics of possible effects. Angiotensin II increases VEGF and TGF-beta through binding to the AT1 receptor, but has the opposite effect when it binds to the AT2 receptor, resulting in a decrease in both of the growth factors, he added.
ACE inhibitors prevent the conversion of angiotensin I to angiotensin II, with the result being that the drugs indirectly block both the AT1 and AT2 receptors.
In contrast, ARBs block only the AT1 receptor and leave the AT2 counter-regulatory receptor alone, said Strauss.
More data, including on overall survival
Strauss and colleagues examined whether ACE inhibitors and/or ARBs could have an effect on the response to PD-1/PD-L1 immune checkpoint inhibitors delivered with or without other immunotherapies, such as anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) checkpoint inhibitors, or targeted agents such as tyrosine kinase inhibitors (TKIs).
They pooled data on 597 patients receiving PD-1/PD-L1 inhibitors in clinical trials for various cancers, including 71 receiving concomitant ARBs, 82 receiving an ACE inhibitor, and 444 who were not receiving either class of antihypertensives.
The above-mentioned improvement in ORR with ARBs compared with patients not receiving the drug was statistically significant (P = .001), as was the improvement in CR rates (P = .002). In contrast, neither ORR nor CR were significantly better with patients on ACE inhibitors compared with patients not taking these drugs.
In multiple regression analysis controlling for age, gender, body mass index (BMI), tumor type, and additional therapies given, the superior ORR and CR rates with ARBs remained (P = .039 and .002, respectively), while there continued to be no significant additional benefit with ACE inhibitors.
The median overall survival was 35.2 months for patients on ARBs, 26.2 months for those on ACE inhibitors, and 18.8 months for patients on neither drug. The respective 3-year OS rates were 48.1%, 37.2%, and 31.5%, with the difference between the ARB and no-drug groups being significant (P = .0078).
In regression analysis controlling for the factors mentioned before, the OS advantage with ARBs but not ACE inhibitors remained significant (P = .006 for ARBs, and .078 for ACE inhibitors).
Strauss emphasized that further study is needed to determine if AT1 blockade can improve outcomes when combined anti-PD-1/PD-L1-based therapy.
It might be reasonable for patients who are taking ACE inhibitors to control blood pressure and are also receiving immunotherapy with a PD-1/PD-L1 inhibitor to be switched to an ARB if it is deemed safe and if further research bears it out, said Strauss in response to a question from Medscape Medical News.
Hypothesis-generating study
Meeting cochair Emiliano Calvo, MD, PhD, from Hospital de Madrid Norte Sanchinarro in Madrid, who attended the media briefing but was not involved in the study, commented that hypothesis-generating research using drugs already on the market for other indications adds important value to cancer therapy.
James Gulley, MD, PhD, from the Center for Cancer Research at the NCI, also a meeting cochair, agreed with Calvo.
“Thinking about utilizing the data that already exists to really get hypothesis-generating questions, it also opens up the possibility for real-world data, real-world evidence from these big datasets from [electronic medical records] that we could really interrogate and understand what we might see and get these hypothesis-generating findings that we could then prospectively evaluate,” Gulley said.
The research was funded by the National Cancer Institute. Strauss and Gulley are National Cancer Institute employees. Calvo disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
It’s not ready for the clinic, but new research suggests that angiotensin receptor II blockers (ARBs) widely used to treat hypertension may improve responses to cancer immunotherapy agents targeted against the programmed death-1/ligand-1 (PD-1/PD-L1) pathway.
That conclusion comes from an observational study of 597 patients with more than 3 dozen different cancer types treated in clinical trials at the US National Institutes of Health. Investigators found that both objective response rates and 3-year overall survival (OS) rates were significantly higher for patients treated with a PD-1/PD-L1 inhibitor who were on ARBs, compared with patients who weren’t taking the antihypertensive agents.
An association was also seen between higher ORR and OS rates for patients taking ACE inhibitors, but it was not statistically significant, reported Julius Strauss, MD, from the Center for Cancer Research at the National Cancer Institute in Bethesda, Md.
All study patients received PD-1/PD-L1 inhibitors, and the ORR for patients treated with ARBs was 33.8%, compared with 19.5% for those treated with ACE inhibitors, and 17% for those who took neither drug. The respective complete response (CR) rates were 11.3%, 3.7%, and 3.1%.
Strauss discussed the data during an online briefing prior to his presentation of the findings during the 32nd EORTC-NCI-AACR Symposium on Molecular Targets and Cancer Therapeutics, which is taking place virtually.
Several early studies have suggested that angiotensin II, in addition to its effect on blood pressure, can also affect cancer growth by leading to downstream production of two proteins: vascular endothelial growth factor (VEGF) and transforming growth factor–beta (TGF-beta), he explained.
“Both of these [proteins] have been linked to cancer growth and cancer resistance to immune system attack,” Strauss observed.
He also discussed the mechanics of possible effects. Angiotensin II increases VEGF and TGF-beta through binding to the AT1 receptor, but has the opposite effect when it binds to the AT2 receptor, resulting in a decrease in both of the growth factors, he added.
ACE inhibitors prevent the conversion of angiotensin I to angiotensin II, with the result being that the drugs indirectly block both the AT1 and AT2 receptors.
In contrast, ARBs block only the AT1 receptor and leave the AT2 counter-regulatory receptor alone, said Strauss.
More data, including on overall survival
Strauss and colleagues examined whether ACE inhibitors and/or ARBs could have an effect on the response to PD-1/PD-L1 immune checkpoint inhibitors delivered with or without other immunotherapies, such as anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) checkpoint inhibitors, or targeted agents such as tyrosine kinase inhibitors (TKIs).
They pooled data on 597 patients receiving PD-1/PD-L1 inhibitors in clinical trials for various cancers, including 71 receiving concomitant ARBs, 82 receiving an ACE inhibitor, and 444 who were not receiving either class of antihypertensives.
The above-mentioned improvement in ORR with ARBs compared with patients not receiving the drug was statistically significant (P = .001), as was the improvement in CR rates (P = .002). In contrast, neither ORR nor CR were significantly better with patients on ACE inhibitors compared with patients not taking these drugs.
In multiple regression analysis controlling for age, gender, body mass index (BMI), tumor type, and additional therapies given, the superior ORR and CR rates with ARBs remained (P = .039 and .002, respectively), while there continued to be no significant additional benefit with ACE inhibitors.
The median overall survival was 35.2 months for patients on ARBs, 26.2 months for those on ACE inhibitors, and 18.8 months for patients on neither drug. The respective 3-year OS rates were 48.1%, 37.2%, and 31.5%, with the difference between the ARB and no-drug groups being significant (P = .0078).
In regression analysis controlling for the factors mentioned before, the OS advantage with ARBs but not ACE inhibitors remained significant (P = .006 for ARBs, and .078 for ACE inhibitors).
Strauss emphasized that further study is needed to determine if AT1 blockade can improve outcomes when combined anti-PD-1/PD-L1-based therapy.
It might be reasonable for patients who are taking ACE inhibitors to control blood pressure and are also receiving immunotherapy with a PD-1/PD-L1 inhibitor to be switched to an ARB if it is deemed safe and if further research bears it out, said Strauss in response to a question from Medscape Medical News.
Hypothesis-generating study
Meeting cochair Emiliano Calvo, MD, PhD, from Hospital de Madrid Norte Sanchinarro in Madrid, who attended the media briefing but was not involved in the study, commented that hypothesis-generating research using drugs already on the market for other indications adds important value to cancer therapy.
James Gulley, MD, PhD, from the Center for Cancer Research at the NCI, also a meeting cochair, agreed with Calvo.
“Thinking about utilizing the data that already exists to really get hypothesis-generating questions, it also opens up the possibility for real-world data, real-world evidence from these big datasets from [electronic medical records] that we could really interrogate and understand what we might see and get these hypothesis-generating findings that we could then prospectively evaluate,” Gulley said.
The research was funded by the National Cancer Institute. Strauss and Gulley are National Cancer Institute employees. Calvo disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
It’s not ready for the clinic, but new research suggests that angiotensin receptor II blockers (ARBs) widely used to treat hypertension may improve responses to cancer immunotherapy agents targeted against the programmed death-1/ligand-1 (PD-1/PD-L1) pathway.
That conclusion comes from an observational study of 597 patients with more than 3 dozen different cancer types treated in clinical trials at the US National Institutes of Health. Investigators found that both objective response rates and 3-year overall survival (OS) rates were significantly higher for patients treated with a PD-1/PD-L1 inhibitor who were on ARBs, compared with patients who weren’t taking the antihypertensive agents.
An association was also seen between higher ORR and OS rates for patients taking ACE inhibitors, but it was not statistically significant, reported Julius Strauss, MD, from the Center for Cancer Research at the National Cancer Institute in Bethesda, Md.
All study patients received PD-1/PD-L1 inhibitors, and the ORR for patients treated with ARBs was 33.8%, compared with 19.5% for those treated with ACE inhibitors, and 17% for those who took neither drug. The respective complete response (CR) rates were 11.3%, 3.7%, and 3.1%.
Strauss discussed the data during an online briefing prior to his presentation of the findings during the 32nd EORTC-NCI-AACR Symposium on Molecular Targets and Cancer Therapeutics, which is taking place virtually.
Several early studies have suggested that angiotensin II, in addition to its effect on blood pressure, can also affect cancer growth by leading to downstream production of two proteins: vascular endothelial growth factor (VEGF) and transforming growth factor–beta (TGF-beta), he explained.
“Both of these [proteins] have been linked to cancer growth and cancer resistance to immune system attack,” Strauss observed.
He also discussed the mechanics of possible effects. Angiotensin II increases VEGF and TGF-beta through binding to the AT1 receptor, but has the opposite effect when it binds to the AT2 receptor, resulting in a decrease in both of the growth factors, he added.
ACE inhibitors prevent the conversion of angiotensin I to angiotensin II, with the result being that the drugs indirectly block both the AT1 and AT2 receptors.
In contrast, ARBs block only the AT1 receptor and leave the AT2 counter-regulatory receptor alone, said Strauss.
More data, including on overall survival
Strauss and colleagues examined whether ACE inhibitors and/or ARBs could have an effect on the response to PD-1/PD-L1 immune checkpoint inhibitors delivered with or without other immunotherapies, such as anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) checkpoint inhibitors, or targeted agents such as tyrosine kinase inhibitors (TKIs).
They pooled data on 597 patients receiving PD-1/PD-L1 inhibitors in clinical trials for various cancers, including 71 receiving concomitant ARBs, 82 receiving an ACE inhibitor, and 444 who were not receiving either class of antihypertensives.
The above-mentioned improvement in ORR with ARBs compared with patients not receiving the drug was statistically significant (P = .001), as was the improvement in CR rates (P = .002). In contrast, neither ORR nor CR were significantly better with patients on ACE inhibitors compared with patients not taking these drugs.
In multiple regression analysis controlling for age, gender, body mass index (BMI), tumor type, and additional therapies given, the superior ORR and CR rates with ARBs remained (P = .039 and .002, respectively), while there continued to be no significant additional benefit with ACE inhibitors.
The median overall survival was 35.2 months for patients on ARBs, 26.2 months for those on ACE inhibitors, and 18.8 months for patients on neither drug. The respective 3-year OS rates were 48.1%, 37.2%, and 31.5%, with the difference between the ARB and no-drug groups being significant (P = .0078).
In regression analysis controlling for the factors mentioned before, the OS advantage with ARBs but not ACE inhibitors remained significant (P = .006 for ARBs, and .078 for ACE inhibitors).
Strauss emphasized that further study is needed to determine if AT1 blockade can improve outcomes when combined anti-PD-1/PD-L1-based therapy.
It might be reasonable for patients who are taking ACE inhibitors to control blood pressure and are also receiving immunotherapy with a PD-1/PD-L1 inhibitor to be switched to an ARB if it is deemed safe and if further research bears it out, said Strauss in response to a question from Medscape Medical News.
Hypothesis-generating study
Meeting cochair Emiliano Calvo, MD, PhD, from Hospital de Madrid Norte Sanchinarro in Madrid, who attended the media briefing but was not involved in the study, commented that hypothesis-generating research using drugs already on the market for other indications adds important value to cancer therapy.
James Gulley, MD, PhD, from the Center for Cancer Research at the NCI, also a meeting cochair, agreed with Calvo.
“Thinking about utilizing the data that already exists to really get hypothesis-generating questions, it also opens up the possibility for real-world data, real-world evidence from these big datasets from [electronic medical records] that we could really interrogate and understand what we might see and get these hypothesis-generating findings that we could then prospectively evaluate,” Gulley said.
The research was funded by the National Cancer Institute. Strauss and Gulley are National Cancer Institute employees. Calvo disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Thermography plus software shows efficacy for breast cancer screening
Sensitivity and area under the curve (AUC) analyses of thermography that is combined with diagnostic software demonstrate “the efficacy of the tool for breast cancer screening,” concludes an observational, comparative study from India published online Oct. 1 in JCO Global Oncology, a publication of the American Society of Clinical Oncology.
Siva Teja Kakileti of Niramai Health Analytix, Koramangala, Bangalore, India, and colleagues said that the product, Thermalytix, is potentially a good fit for low- and middle-income countries because it is portable and provides automated quantitative analysis of thermal images – and thus can be conducted by technicians with “minimal training.”
Conventional thermography involves manual interpretation of complex thermal images, which “often results in erroneous results owing to subjectivity,” said the study authors.
That manual interpretation of thermal images might involve looking at 200 color shades, which is “high cognitive overload for the thermographer,” explained Mr. Kakileti in an interview.
However, an American mammography expert who was approached for comment dismissed thermography – even with the new twist of software-aided diagnostic scoring by Thermalytix – as wholly inappropriate for the detection of early breast cancer, owing to inherent limitations.
“Thermal imaging of any type has no value in finding early breast cancer,” Daniel Kopans, MD, of Harvard University and Massachusetts General Hospital, both in Boston, said in an interview. He said that thermal imaging only detects heat on the skin and perhaps a few millimeters beneath the skin and thus misses deeper cancers, the heat from which is carried away by the vascular system.
The new study included 470 women who presented for breast screening at two centers in Bangalore, India. A total of 238 women had symptoms such as breast lump, nipple discharge, skin changes, or breast pain; the remaining 232 women were asymptomatic.
All participants underwent a Thermalytix test and one or more standard-of-care tests for breast cancer screening (such as mammography, ultrasonography, biopsy, fine-needle aspiration, or elastography). A total of 78 women, or 16.6% of the group overall, were diagnosed with a malignancy. For the overall group of 470 women, Thermalytix had a sensitivity of 91.02% (symptomatic, 89.85%; asymptomatic,100%) and a specificity of 82.39% (symptomatic, 69.04%; asymptomatic, 92.41%) in detection of breast malignancy. Thermalytix showed an overall AUC of 0.90, with an AUC of 0.82 for symptomatic and 0.98 for asymptomatic women.
The study authors characterized both the sensitivity and AUC as “high.”
The results from the study, which the authors characterized as preliminary, encouraged the study sponsor, Niramai, to start planning a large-scale, multicountry trial.
But Dr. Kopans, who serves as a consultant to DART, which produces digital breast tomosynthesis units in China, suggested that this research will be fruitless. “Thermal imaging seems to raise its head every few years since it is passive, but it does not work and is a waste of money,” Dr. Kopans reiterated.
“Its use can be dangerous by dissuading women from being screened with mammography, which has been proven to save lives,” he stressed.
Thermalytix compared with mammography
Investigators also compared screening results in the subset of 242 women who underwent both Thermalytix and mammography. Results showed that Thermalytix had a higher sensitivity than did mammography (91.23% vs. 85.96%), but mammography had a higher specificity than Thermalytix did (94.05% vs. 68.65%).
In the asymptomatic group who underwent both tests (n = 95), four cancers were detected, and Thermalytix demonstrated superior sensitivity than mammography (100% vs. 50%), Mr. Kakileti and colleagues state.
Thermalytix evaluates vascularity variations too
In the subset of 228 women who did not undergo mammography (owing to dense breasts, younger age, or other reasons), Thermalytix detected tumors in all but 3 of 21 patients who went on to be diagnosed with breast cancer. The authors state that, because their artificial intelligence–based analysis uses vascularity, as well as temperature variations on the skin, to complement hot-spot detection, it is able to detect small lesions.
In the current study, 24 malignant tumors were less than 2 cm in diameter, and Thermalytix was able to identify 17 of the tumors as positive, for a 71% sensitivity rate for T1 tumors. This compared with a 68% sensitivity rate for mammography for detecting the same T1 tumors. Thermalytix also showed promising results in women younger than 40 years, for whom screening mammography is not usually recommended. The automated test picked up all 11 tumors eventually diagnosed in this younger cohort.
“Thermalytix is a portable, noninvasive, radiation-free test that has shown promising results in this preliminary study,” the investigators wrote, “[and] it can be an affordable and scalable method of screening in remote areas,” they added.
“We believe that Thermalytix ... is poised to be a promising modality for breast cancer screening,” Mr. Kakileti and colleagues summarized.
The FDA warns about thermography in place of mammography
The US Food and Drug Administration fairly recently warned against the use of thermography as an alternative to mammography for breast cancer screening or diagnosis, noting that it has received reports that facilities where thermography is offered often provide false information about the technology that can mislead patients into believing that it is either an alternative to or a better option than mammography.
Dr. Kopans says that other groups have invested in thermography research. “The Israelis spent millions working on a similar approach that didn’t work,” he commented.
The new software from Thermalytix, which is derived from artificial intelligence, is a “gimmick,” says the Boston radiologist. “If the basic information is not there, a computer cannot find it,” he stated, referring to what he believes are deeper-tissue tumors that are inaccessible to heat-detecting technology.
Mr. Kakileti is an employee of Nirami Health Analytix and owns stock and has filed patents with the company. Other investigators are also employed by the same company or receive research and other funding or have patents filed by the company as well. Dr. Kopans serves as a consultant to DART, which produces digital breast tomosynthesis units in China.
A version of this article originally appeared on Medscape.com.
Sensitivity and area under the curve (AUC) analyses of thermography that is combined with diagnostic software demonstrate “the efficacy of the tool for breast cancer screening,” concludes an observational, comparative study from India published online Oct. 1 in JCO Global Oncology, a publication of the American Society of Clinical Oncology.
Siva Teja Kakileti of Niramai Health Analytix, Koramangala, Bangalore, India, and colleagues said that the product, Thermalytix, is potentially a good fit for low- and middle-income countries because it is portable and provides automated quantitative analysis of thermal images – and thus can be conducted by technicians with “minimal training.”
Conventional thermography involves manual interpretation of complex thermal images, which “often results in erroneous results owing to subjectivity,” said the study authors.
That manual interpretation of thermal images might involve looking at 200 color shades, which is “high cognitive overload for the thermographer,” explained Mr. Kakileti in an interview.
However, an American mammography expert who was approached for comment dismissed thermography – even with the new twist of software-aided diagnostic scoring by Thermalytix – as wholly inappropriate for the detection of early breast cancer, owing to inherent limitations.
“Thermal imaging of any type has no value in finding early breast cancer,” Daniel Kopans, MD, of Harvard University and Massachusetts General Hospital, both in Boston, said in an interview. He said that thermal imaging only detects heat on the skin and perhaps a few millimeters beneath the skin and thus misses deeper cancers, the heat from which is carried away by the vascular system.
The new study included 470 women who presented for breast screening at two centers in Bangalore, India. A total of 238 women had symptoms such as breast lump, nipple discharge, skin changes, or breast pain; the remaining 232 women were asymptomatic.
All participants underwent a Thermalytix test and one or more standard-of-care tests for breast cancer screening (such as mammography, ultrasonography, biopsy, fine-needle aspiration, or elastography). A total of 78 women, or 16.6% of the group overall, were diagnosed with a malignancy. For the overall group of 470 women, Thermalytix had a sensitivity of 91.02% (symptomatic, 89.85%; asymptomatic,100%) and a specificity of 82.39% (symptomatic, 69.04%; asymptomatic, 92.41%) in detection of breast malignancy. Thermalytix showed an overall AUC of 0.90, with an AUC of 0.82 for symptomatic and 0.98 for asymptomatic women.
The study authors characterized both the sensitivity and AUC as “high.”
The results from the study, which the authors characterized as preliminary, encouraged the study sponsor, Niramai, to start planning a large-scale, multicountry trial.
But Dr. Kopans, who serves as a consultant to DART, which produces digital breast tomosynthesis units in China, suggested that this research will be fruitless. “Thermal imaging seems to raise its head every few years since it is passive, but it does not work and is a waste of money,” Dr. Kopans reiterated.
“Its use can be dangerous by dissuading women from being screened with mammography, which has been proven to save lives,” he stressed.
Thermalytix compared with mammography
Investigators also compared screening results in the subset of 242 women who underwent both Thermalytix and mammography. Results showed that Thermalytix had a higher sensitivity than did mammography (91.23% vs. 85.96%), but mammography had a higher specificity than Thermalytix did (94.05% vs. 68.65%).
In the asymptomatic group who underwent both tests (n = 95), four cancers were detected, and Thermalytix demonstrated superior sensitivity than mammography (100% vs. 50%), Mr. Kakileti and colleagues state.
Thermalytix evaluates vascularity variations too
In the subset of 228 women who did not undergo mammography (owing to dense breasts, younger age, or other reasons), Thermalytix detected tumors in all but 3 of 21 patients who went on to be diagnosed with breast cancer. The authors state that, because their artificial intelligence–based analysis uses vascularity, as well as temperature variations on the skin, to complement hot-spot detection, it is able to detect small lesions.
In the current study, 24 malignant tumors were less than 2 cm in diameter, and Thermalytix was able to identify 17 of the tumors as positive, for a 71% sensitivity rate for T1 tumors. This compared with a 68% sensitivity rate for mammography for detecting the same T1 tumors. Thermalytix also showed promising results in women younger than 40 years, for whom screening mammography is not usually recommended. The automated test picked up all 11 tumors eventually diagnosed in this younger cohort.
“Thermalytix is a portable, noninvasive, radiation-free test that has shown promising results in this preliminary study,” the investigators wrote, “[and] it can be an affordable and scalable method of screening in remote areas,” they added.
“We believe that Thermalytix ... is poised to be a promising modality for breast cancer screening,” Mr. Kakileti and colleagues summarized.
The FDA warns about thermography in place of mammography
The US Food and Drug Administration fairly recently warned against the use of thermography as an alternative to mammography for breast cancer screening or diagnosis, noting that it has received reports that facilities where thermography is offered often provide false information about the technology that can mislead patients into believing that it is either an alternative to or a better option than mammography.
Dr. Kopans says that other groups have invested in thermography research. “The Israelis spent millions working on a similar approach that didn’t work,” he commented.
The new software from Thermalytix, which is derived from artificial intelligence, is a “gimmick,” says the Boston radiologist. “If the basic information is not there, a computer cannot find it,” he stated, referring to what he believes are deeper-tissue tumors that are inaccessible to heat-detecting technology.
Mr. Kakileti is an employee of Nirami Health Analytix and owns stock and has filed patents with the company. Other investigators are also employed by the same company or receive research and other funding or have patents filed by the company as well. Dr. Kopans serves as a consultant to DART, which produces digital breast tomosynthesis units in China.
A version of this article originally appeared on Medscape.com.
Sensitivity and area under the curve (AUC) analyses of thermography that is combined with diagnostic software demonstrate “the efficacy of the tool for breast cancer screening,” concludes an observational, comparative study from India published online Oct. 1 in JCO Global Oncology, a publication of the American Society of Clinical Oncology.
Siva Teja Kakileti of Niramai Health Analytix, Koramangala, Bangalore, India, and colleagues said that the product, Thermalytix, is potentially a good fit for low- and middle-income countries because it is portable and provides automated quantitative analysis of thermal images – and thus can be conducted by technicians with “minimal training.”
Conventional thermography involves manual interpretation of complex thermal images, which “often results in erroneous results owing to subjectivity,” said the study authors.
That manual interpretation of thermal images might involve looking at 200 color shades, which is “high cognitive overload for the thermographer,” explained Mr. Kakileti in an interview.
However, an American mammography expert who was approached for comment dismissed thermography – even with the new twist of software-aided diagnostic scoring by Thermalytix – as wholly inappropriate for the detection of early breast cancer, owing to inherent limitations.
“Thermal imaging of any type has no value in finding early breast cancer,” Daniel Kopans, MD, of Harvard University and Massachusetts General Hospital, both in Boston, said in an interview. He said that thermal imaging only detects heat on the skin and perhaps a few millimeters beneath the skin and thus misses deeper cancers, the heat from which is carried away by the vascular system.
The new study included 470 women who presented for breast screening at two centers in Bangalore, India. A total of 238 women had symptoms such as breast lump, nipple discharge, skin changes, or breast pain; the remaining 232 women were asymptomatic.
All participants underwent a Thermalytix test and one or more standard-of-care tests for breast cancer screening (such as mammography, ultrasonography, biopsy, fine-needle aspiration, or elastography). A total of 78 women, or 16.6% of the group overall, were diagnosed with a malignancy. For the overall group of 470 women, Thermalytix had a sensitivity of 91.02% (symptomatic, 89.85%; asymptomatic,100%) and a specificity of 82.39% (symptomatic, 69.04%; asymptomatic, 92.41%) in detection of breast malignancy. Thermalytix showed an overall AUC of 0.90, with an AUC of 0.82 for symptomatic and 0.98 for asymptomatic women.
The study authors characterized both the sensitivity and AUC as “high.”
The results from the study, which the authors characterized as preliminary, encouraged the study sponsor, Niramai, to start planning a large-scale, multicountry trial.
But Dr. Kopans, who serves as a consultant to DART, which produces digital breast tomosynthesis units in China, suggested that this research will be fruitless. “Thermal imaging seems to raise its head every few years since it is passive, but it does not work and is a waste of money,” Dr. Kopans reiterated.
“Its use can be dangerous by dissuading women from being screened with mammography, which has been proven to save lives,” he stressed.
Thermalytix compared with mammography
Investigators also compared screening results in the subset of 242 women who underwent both Thermalytix and mammography. Results showed that Thermalytix had a higher sensitivity than did mammography (91.23% vs. 85.96%), but mammography had a higher specificity than Thermalytix did (94.05% vs. 68.65%).
In the asymptomatic group who underwent both tests (n = 95), four cancers were detected, and Thermalytix demonstrated superior sensitivity than mammography (100% vs. 50%), Mr. Kakileti and colleagues state.
Thermalytix evaluates vascularity variations too
In the subset of 228 women who did not undergo mammography (owing to dense breasts, younger age, or other reasons), Thermalytix detected tumors in all but 3 of 21 patients who went on to be diagnosed with breast cancer. The authors state that, because their artificial intelligence–based analysis uses vascularity, as well as temperature variations on the skin, to complement hot-spot detection, it is able to detect small lesions.
In the current study, 24 malignant tumors were less than 2 cm in diameter, and Thermalytix was able to identify 17 of the tumors as positive, for a 71% sensitivity rate for T1 tumors. This compared with a 68% sensitivity rate for mammography for detecting the same T1 tumors. Thermalytix also showed promising results in women younger than 40 years, for whom screening mammography is not usually recommended. The automated test picked up all 11 tumors eventually diagnosed in this younger cohort.
“Thermalytix is a portable, noninvasive, radiation-free test that has shown promising results in this preliminary study,” the investigators wrote, “[and] it can be an affordable and scalable method of screening in remote areas,” they added.
“We believe that Thermalytix ... is poised to be a promising modality for breast cancer screening,” Mr. Kakileti and colleagues summarized.
The FDA warns about thermography in place of mammography
The US Food and Drug Administration fairly recently warned against the use of thermography as an alternative to mammography for breast cancer screening or diagnosis, noting that it has received reports that facilities where thermography is offered often provide false information about the technology that can mislead patients into believing that it is either an alternative to or a better option than mammography.
Dr. Kopans says that other groups have invested in thermography research. “The Israelis spent millions working on a similar approach that didn’t work,” he commented.
The new software from Thermalytix, which is derived from artificial intelligence, is a “gimmick,” says the Boston radiologist. “If the basic information is not there, a computer cannot find it,” he stated, referring to what he believes are deeper-tissue tumors that are inaccessible to heat-detecting technology.
Mr. Kakileti is an employee of Nirami Health Analytix and owns stock and has filed patents with the company. Other investigators are also employed by the same company or receive research and other funding or have patents filed by the company as well. Dr. Kopans serves as a consultant to DART, which produces digital breast tomosynthesis units in China.
A version of this article originally appeared on Medscape.com.
Are oncologists ready to confront a second wave of COVID-19?
Canceled appointments, postponed surgeries, and delayed cancer diagnoses – all are a recipe for exhaustion for oncologists around the world, struggling to reach and treat their patients during the pandemic. Physicians and their teams felt the pain as COVID-19 took its initial march around the globe.
“We saw the distress of people with cancer who could no longer get to anyone on the phone. Their medical visit was usually canceled. Their radiotherapy session was postponed or modified, and chemotherapy postponed,” says Axel Kahn, MD, chairman of the board of directors of La Ligue Nationale Contre le Cancer (National League Against Cancer). “In the vast majority of cases, cancer treatment can be postponed or readjusted, without affecting the patient’s chances of survival, but there has been a lot of anxiety because the patients do not know that.”
The stay-at-home factor was one that played out across many months during the first wave.
“I believe that the ‘stay-home’ message that we transmitted was rigorously followed by patients who should have come to the emergency room much earlier and who, therefore, were admitted with a much more deteriorated general condition than in non-COVID-19 times,” says Benjamín Domingo Arrué, MD, from the department of medical oncology at Hospital Universitari i Politècnic La Fe in Valencia, Spain.
And in Brazil, some of the impact from the initial hit of COVID-19 on oncology is only now being felt, according to Laura Testa, MD, head of breast medical oncology, Instituto do Câncer do Estado de São Paulo.
“We are starting to see a lot of cancer cases that didn’t show up at the beginning of the pandemic, but now they are arriving to us already in advanced stages,” she said. “These patients need hospital care. If the situation worsens and goes back to what we saw at the peak of the curve, I fear the public system won’t be able to treat properly the oncology patients that need hospital care and the patients with cancer who also have COVID-19.”
But even as health care worker fatigue and concerns linger, oncologists say that what they have learned in the last 6 months has helped them prepare as COVID-19 cases increase and a second global wave kicks up.
Lessons from the first wave
In the United States, COVID-19 hit different regions at different times and to different degrees. One of the areas hit first was Seattle.
“We jumped on top of this, we were evidence based, we put things in place very, very quickly,” said Julie Gralow, MD, professor at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle.
“We did a really good job keeping COVID out of our cancer centers,” Dr. Gralow said. “We learned how to be super safe, and to keep symptomatic people out of the building, and to limit the extra people they could bring with them. It’s all about the number of contacts you have.”
The story was different, though, for oncologists in several other countries, and sometimes it varied immensely within each nation.
“We treated fewer patients with cancer during the first wave,” says Dirk Arnold, MD, medical director of the Asklepios Tumor Center Hamburg (Germany), in an interview. “In part, this was because staff were quarantined and because we had a completely different infrastructure in all of the hospitals. But also fewer patients with cancer came to the clinic at all. A lot of resources were directed toward COVID-19.”
In Spain, telemedicine helped keep up with visits, but other areas felt the effect of COVID-19 patient loads.
“At least in the oncology department of our center, we have practically maintained 100% of visits, mostly by telephone,” says Dr. Arrué, “but the reality is that our country has not yet been prepared for telemedicine.”
Laura Mezquita, MD, of the department of medical oncology at Hospital Clinic de Barcelona, describes a more dramatic situation: “We have seen how some of our patients, especially with metastatic disease, have been dismissed for intensive care and life-support treatments, as well as specific treatments against COVID-19 (tocilizumab, remdesivir, etc.) due to the general health collapse of the former wave,” she said. She adds that specific oncologic populations, such as those with thoracic tumors, have been more affected.
Distress among oncologists
Many oncologists are still feeling stressed and fatigued after the first wave, just as a second string of outbreaks is on its way.
A survey presented at last month’s ESMO 2020 Congress found that, in July-August, moral distress was reported by one-third of the oncologists who responded, and more than half reported a feeling of exhaustion.
“The tiredness and team exhaustion is noticeable,” said Dr. Arnold. “We recently had a task force discussion about what will happen when we have a second wave and how the department and our services will adapt. It was clear that those who were at the very front in the first wave had only a limited desire to do that again in the second wave.”
Another concern: COVID-19’s effect on staffing levels.
“We have a population of young caregivers who are affected by the COVID-19 disease with an absenteeism rate that is quite unprecedented,” said Sophie Beaupère, general delegate of Unicancer since January.
She said that, in general, the absenteeism rate in the cancer centers averages 5%-6%, depending on the year. But that rate is now skyrocketing.
Stop-start cycle for surgery
As caregivers quarantined around the world, more than 10% of patients with cancer had treatment canceled or delayed during the first wave of the pandemic, according to another survey from ESMO, involving 109 oncologists from 18 countries.
Difficulties were reported for surgeries by 34% of the centers, but also difficulties with delivering chemotherapy (22% of centers), radiotherapy (13.7%), and therapy with checkpoint inhibitors (9.1%), monoclonal antibodies (9%), and oral targeted therapy (3.7%).
Stopping surgery is a real concern in France, noted Dr. Kahn, the National League Against Cancer chair. He says that in regions that were badly hit by COVID-19, “it was not possible to have access to the operating room for people who absolutely needed surgery; for example, patients with lung cancer that was still operable. Most of the recovery rooms were mobilized for resuscitation.”
There may be some solutions, suggested Thierry Breton, director general of the National Institute of Cancer in France. “We are getting prepared, with the health ministry, for a possible increase in hospital tension, which would lead to a situation where we would have to reschedule operations. Nationally, regionally, and locally, we are seeing how we can resume and prioritize surgeries that have not been done.”
Delays in cancer diagnosis
While COVID-19 affected treatment, many oncologists say the major impact of the first wave was a delay in diagnosing cancer. Some of this was a result of the suspension of cancer screening programs, but there was also fear among the general public about visiting clinics and hospitals during a pandemic.
“We didn’t do so well with cancer during the first wave here in the U.K.,” said Karol Sikora, PhD, MBBChir, professor of cancer medicine and founding dean at the University of Buckingham Medical School, London. “Cancer diagnostic pathways virtually stalled partly because patients didn’t seek help, but getting scans and biopsies was also very difficult. Even patients referred urgently under the ‘2-weeks-wait’ rule were turned down.”
In France, “the delay in diagnosis is indisputable,” said Dr. Kahn. “About 50% of the cancer diagnoses one would expect during this period were missed.”
“I am worried that there remains a major traffic jam that has not been caught up with, and, in the meantime, the health crisis is worsening,” he added.
In Seattle, Dr. Gralow said the first COVID-19 wave had little impact on treatment for breast cancer, but it was in screening for breast cancer “where things really got messed up.”
“Even though we’ve been fully ramped up again,” she said, concerns remain. To ensure that screening mammography is maintained, “we have spaced out the visits to keep our waiting rooms less populated, with a longer time between using the machine so we can clean it. To do this, we have extended operating hours and are now opening on Saturday.
“So we’re actually at 100% of our capacity, but I’m really nervous, though, that a lot of people put off their screening mammogram and aren’t going to come in and get it.
“Not only did people get the message to stay home and not do nonessential things, but I think a lot of people lost their health insurance when they lost their jobs,” she said, and without health insurance, they are not covered for cancer screening.
Looking ahead, with a plan
Many oncologists agree that access to care can and must be improved – and there were some positive moves.
“Some regimens changed during the first months of the pandemic, and I don’t see them going back to the way they were anytime soon,” said Dr. Testa. “The changes/adaptations that were made to minimize the chance of SARS-CoV-2 infection are still in place and will go on for a while. In this context, telemedicine helped a lot. The pandemic forced the stakeholders to step up and put it in place in March. And now it’s here to stay.”
The experience gained in the last several months has driven preparation for the next wave.
“We are not going to see the disorganization that we saw during the first wave,” said Florence Joly, MD, PhD, head of medical oncology at the Centre François Baclesse in Caen, France. “The difference between now and earlier this year is that COVID diagnostic tests are available. That was one of the problems in the first wave. We had no way to diagnose.”
On the East Coast of the United States, medical oncologist Charu Aggarwal, MD, MPH, is also optimistic: “I think we’re at a place where we can manage.”
“I believe if there was going to be a new wave of COVID-19 cases we would be: better psychologically prepared and better organized,” said Dr. Aggarwal, assistant professor of medicine in the hematology-oncology division at the University of Pennsylvania, Philadelphia. “We already have experience with all of the tools, we have telemedicine available, we have screening protocols available, we have testing, we are already universally masking, everyone’s hand-washing, so I do think that means we would be okay.”
Dr. Arnold agreed that “we are much better prepared than for the first wave, but … we have immense tasks in the area of patient management, the digitization of patient care, the clear allocation of resources when there is a second or third wave. In many areas of preparation, I believe, unfortunately, we are not as well positioned as we had actually hoped.”
The first wave of COVID hit cancer services in the United Kingdom particularly hard: One modeling study suggested that delays in cancer referrals will lead to thousands of additional deaths and tens of thousands of life-years lost.
“Cancer services are working at near normal levels now, but they are still fragile and could be severely compromised again if the NHS [National Health Service] gets flooded by COVID patients,” said Dr. Sikora.
The second wave may be different. “Although the number of infections has increased, the hospitalizations have only risen a little. Let’s see what happens,” he said in an interview. Since then, however, infections have continued to rise, and there has been an increase in hospitalizations. New social distancing measures in the United Kingdom were put into place on Oct. 12, with the aim of protecting the NHS from overload.
Dr. Arrué describes it this way: “The reality is that the ‘second wave’ has left behind the initial grief and shock that both patients and health professionals experienced when faced with something that, until now, we had only seen in the movies.” The second wave has led to new restrictions – including a partial lockdown since the beginning of October.
Dr. Aggarwal says her department recently had a conference with Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, about the impact of COVID-19 on oncology.
“I asked him what advice he’d give oncologists, and he said to go back to as much screening as you were doing previously as quickly as possible. That’s what must be relayed to our oncologists in the community – and also to primary care physicians – because they are often the ones who are ordering and championing the screening efforts.”
This article was originated by Aude Lecrubier, Medscape French edition, and developed by Zosia Chustecka, Medscape Oncology. With additional reporting by Kate Johnson, freelance medical journalist, Claudia Gottschling for Medscape Germany, Leoleli Schwartz for Medscape em português, Tim Locke for Medscape United Kingdom, and Carla Nieto Martínez, freelance medical journalist for Medscape Spanish edition.
This article first appeared on Medscape.com.
Canceled appointments, postponed surgeries, and delayed cancer diagnoses – all are a recipe for exhaustion for oncologists around the world, struggling to reach and treat their patients during the pandemic. Physicians and their teams felt the pain as COVID-19 took its initial march around the globe.
“We saw the distress of people with cancer who could no longer get to anyone on the phone. Their medical visit was usually canceled. Their radiotherapy session was postponed or modified, and chemotherapy postponed,” says Axel Kahn, MD, chairman of the board of directors of La Ligue Nationale Contre le Cancer (National League Against Cancer). “In the vast majority of cases, cancer treatment can be postponed or readjusted, without affecting the patient’s chances of survival, but there has been a lot of anxiety because the patients do not know that.”
The stay-at-home factor was one that played out across many months during the first wave.
“I believe that the ‘stay-home’ message that we transmitted was rigorously followed by patients who should have come to the emergency room much earlier and who, therefore, were admitted with a much more deteriorated general condition than in non-COVID-19 times,” says Benjamín Domingo Arrué, MD, from the department of medical oncology at Hospital Universitari i Politècnic La Fe in Valencia, Spain.
And in Brazil, some of the impact from the initial hit of COVID-19 on oncology is only now being felt, according to Laura Testa, MD, head of breast medical oncology, Instituto do Câncer do Estado de São Paulo.
“We are starting to see a lot of cancer cases that didn’t show up at the beginning of the pandemic, but now they are arriving to us already in advanced stages,” she said. “These patients need hospital care. If the situation worsens and goes back to what we saw at the peak of the curve, I fear the public system won’t be able to treat properly the oncology patients that need hospital care and the patients with cancer who also have COVID-19.”
But even as health care worker fatigue and concerns linger, oncologists say that what they have learned in the last 6 months has helped them prepare as COVID-19 cases increase and a second global wave kicks up.
Lessons from the first wave
In the United States, COVID-19 hit different regions at different times and to different degrees. One of the areas hit first was Seattle.
“We jumped on top of this, we were evidence based, we put things in place very, very quickly,” said Julie Gralow, MD, professor at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle.
“We did a really good job keeping COVID out of our cancer centers,” Dr. Gralow said. “We learned how to be super safe, and to keep symptomatic people out of the building, and to limit the extra people they could bring with them. It’s all about the number of contacts you have.”
The story was different, though, for oncologists in several other countries, and sometimes it varied immensely within each nation.
“We treated fewer patients with cancer during the first wave,” says Dirk Arnold, MD, medical director of the Asklepios Tumor Center Hamburg (Germany), in an interview. “In part, this was because staff were quarantined and because we had a completely different infrastructure in all of the hospitals. But also fewer patients with cancer came to the clinic at all. A lot of resources were directed toward COVID-19.”
In Spain, telemedicine helped keep up with visits, but other areas felt the effect of COVID-19 patient loads.
“At least in the oncology department of our center, we have practically maintained 100% of visits, mostly by telephone,” says Dr. Arrué, “but the reality is that our country has not yet been prepared for telemedicine.”
Laura Mezquita, MD, of the department of medical oncology at Hospital Clinic de Barcelona, describes a more dramatic situation: “We have seen how some of our patients, especially with metastatic disease, have been dismissed for intensive care and life-support treatments, as well as specific treatments against COVID-19 (tocilizumab, remdesivir, etc.) due to the general health collapse of the former wave,” she said. She adds that specific oncologic populations, such as those with thoracic tumors, have been more affected.
Distress among oncologists
Many oncologists are still feeling stressed and fatigued after the first wave, just as a second string of outbreaks is on its way.
A survey presented at last month’s ESMO 2020 Congress found that, in July-August, moral distress was reported by one-third of the oncologists who responded, and more than half reported a feeling of exhaustion.
“The tiredness and team exhaustion is noticeable,” said Dr. Arnold. “We recently had a task force discussion about what will happen when we have a second wave and how the department and our services will adapt. It was clear that those who were at the very front in the first wave had only a limited desire to do that again in the second wave.”
Another concern: COVID-19’s effect on staffing levels.
“We have a population of young caregivers who are affected by the COVID-19 disease with an absenteeism rate that is quite unprecedented,” said Sophie Beaupère, general delegate of Unicancer since January.
She said that, in general, the absenteeism rate in the cancer centers averages 5%-6%, depending on the year. But that rate is now skyrocketing.
Stop-start cycle for surgery
As caregivers quarantined around the world, more than 10% of patients with cancer had treatment canceled or delayed during the first wave of the pandemic, according to another survey from ESMO, involving 109 oncologists from 18 countries.
Difficulties were reported for surgeries by 34% of the centers, but also difficulties with delivering chemotherapy (22% of centers), radiotherapy (13.7%), and therapy with checkpoint inhibitors (9.1%), monoclonal antibodies (9%), and oral targeted therapy (3.7%).
Stopping surgery is a real concern in France, noted Dr. Kahn, the National League Against Cancer chair. He says that in regions that were badly hit by COVID-19, “it was not possible to have access to the operating room for people who absolutely needed surgery; for example, patients with lung cancer that was still operable. Most of the recovery rooms were mobilized for resuscitation.”
There may be some solutions, suggested Thierry Breton, director general of the National Institute of Cancer in France. “We are getting prepared, with the health ministry, for a possible increase in hospital tension, which would lead to a situation where we would have to reschedule operations. Nationally, regionally, and locally, we are seeing how we can resume and prioritize surgeries that have not been done.”
Delays in cancer diagnosis
While COVID-19 affected treatment, many oncologists say the major impact of the first wave was a delay in diagnosing cancer. Some of this was a result of the suspension of cancer screening programs, but there was also fear among the general public about visiting clinics and hospitals during a pandemic.
“We didn’t do so well with cancer during the first wave here in the U.K.,” said Karol Sikora, PhD, MBBChir, professor of cancer medicine and founding dean at the University of Buckingham Medical School, London. “Cancer diagnostic pathways virtually stalled partly because patients didn’t seek help, but getting scans and biopsies was also very difficult. Even patients referred urgently under the ‘2-weeks-wait’ rule were turned down.”
In France, “the delay in diagnosis is indisputable,” said Dr. Kahn. “About 50% of the cancer diagnoses one would expect during this period were missed.”
“I am worried that there remains a major traffic jam that has not been caught up with, and, in the meantime, the health crisis is worsening,” he added.
In Seattle, Dr. Gralow said the first COVID-19 wave had little impact on treatment for breast cancer, but it was in screening for breast cancer “where things really got messed up.”
“Even though we’ve been fully ramped up again,” she said, concerns remain. To ensure that screening mammography is maintained, “we have spaced out the visits to keep our waiting rooms less populated, with a longer time between using the machine so we can clean it. To do this, we have extended operating hours and are now opening on Saturday.
“So we’re actually at 100% of our capacity, but I’m really nervous, though, that a lot of people put off their screening mammogram and aren’t going to come in and get it.
“Not only did people get the message to stay home and not do nonessential things, but I think a lot of people lost their health insurance when they lost their jobs,” she said, and without health insurance, they are not covered for cancer screening.
Looking ahead, with a plan
Many oncologists agree that access to care can and must be improved – and there were some positive moves.
“Some regimens changed during the first months of the pandemic, and I don’t see them going back to the way they were anytime soon,” said Dr. Testa. “The changes/adaptations that were made to minimize the chance of SARS-CoV-2 infection are still in place and will go on for a while. In this context, telemedicine helped a lot. The pandemic forced the stakeholders to step up and put it in place in March. And now it’s here to stay.”
The experience gained in the last several months has driven preparation for the next wave.
“We are not going to see the disorganization that we saw during the first wave,” said Florence Joly, MD, PhD, head of medical oncology at the Centre François Baclesse in Caen, France. “The difference between now and earlier this year is that COVID diagnostic tests are available. That was one of the problems in the first wave. We had no way to diagnose.”
On the East Coast of the United States, medical oncologist Charu Aggarwal, MD, MPH, is also optimistic: “I think we’re at a place where we can manage.”
“I believe if there was going to be a new wave of COVID-19 cases we would be: better psychologically prepared and better organized,” said Dr. Aggarwal, assistant professor of medicine in the hematology-oncology division at the University of Pennsylvania, Philadelphia. “We already have experience with all of the tools, we have telemedicine available, we have screening protocols available, we have testing, we are already universally masking, everyone’s hand-washing, so I do think that means we would be okay.”
Dr. Arnold agreed that “we are much better prepared than for the first wave, but … we have immense tasks in the area of patient management, the digitization of patient care, the clear allocation of resources when there is a second or third wave. In many areas of preparation, I believe, unfortunately, we are not as well positioned as we had actually hoped.”
The first wave of COVID hit cancer services in the United Kingdom particularly hard: One modeling study suggested that delays in cancer referrals will lead to thousands of additional deaths and tens of thousands of life-years lost.
“Cancer services are working at near normal levels now, but they are still fragile and could be severely compromised again if the NHS [National Health Service] gets flooded by COVID patients,” said Dr. Sikora.
The second wave may be different. “Although the number of infections has increased, the hospitalizations have only risen a little. Let’s see what happens,” he said in an interview. Since then, however, infections have continued to rise, and there has been an increase in hospitalizations. New social distancing measures in the United Kingdom were put into place on Oct. 12, with the aim of protecting the NHS from overload.
Dr. Arrué describes it this way: “The reality is that the ‘second wave’ has left behind the initial grief and shock that both patients and health professionals experienced when faced with something that, until now, we had only seen in the movies.” The second wave has led to new restrictions – including a partial lockdown since the beginning of October.
Dr. Aggarwal says her department recently had a conference with Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, about the impact of COVID-19 on oncology.
“I asked him what advice he’d give oncologists, and he said to go back to as much screening as you were doing previously as quickly as possible. That’s what must be relayed to our oncologists in the community – and also to primary care physicians – because they are often the ones who are ordering and championing the screening efforts.”
This article was originated by Aude Lecrubier, Medscape French edition, and developed by Zosia Chustecka, Medscape Oncology. With additional reporting by Kate Johnson, freelance medical journalist, Claudia Gottschling for Medscape Germany, Leoleli Schwartz for Medscape em português, Tim Locke for Medscape United Kingdom, and Carla Nieto Martínez, freelance medical journalist for Medscape Spanish edition.
This article first appeared on Medscape.com.
Canceled appointments, postponed surgeries, and delayed cancer diagnoses – all are a recipe for exhaustion for oncologists around the world, struggling to reach and treat their patients during the pandemic. Physicians and their teams felt the pain as COVID-19 took its initial march around the globe.
“We saw the distress of people with cancer who could no longer get to anyone on the phone. Their medical visit was usually canceled. Their radiotherapy session was postponed or modified, and chemotherapy postponed,” says Axel Kahn, MD, chairman of the board of directors of La Ligue Nationale Contre le Cancer (National League Against Cancer). “In the vast majority of cases, cancer treatment can be postponed or readjusted, without affecting the patient’s chances of survival, but there has been a lot of anxiety because the patients do not know that.”
The stay-at-home factor was one that played out across many months during the first wave.
“I believe that the ‘stay-home’ message that we transmitted was rigorously followed by patients who should have come to the emergency room much earlier and who, therefore, were admitted with a much more deteriorated general condition than in non-COVID-19 times,” says Benjamín Domingo Arrué, MD, from the department of medical oncology at Hospital Universitari i Politècnic La Fe in Valencia, Spain.
And in Brazil, some of the impact from the initial hit of COVID-19 on oncology is only now being felt, according to Laura Testa, MD, head of breast medical oncology, Instituto do Câncer do Estado de São Paulo.
“We are starting to see a lot of cancer cases that didn’t show up at the beginning of the pandemic, but now they are arriving to us already in advanced stages,” she said. “These patients need hospital care. If the situation worsens and goes back to what we saw at the peak of the curve, I fear the public system won’t be able to treat properly the oncology patients that need hospital care and the patients with cancer who also have COVID-19.”
But even as health care worker fatigue and concerns linger, oncologists say that what they have learned in the last 6 months has helped them prepare as COVID-19 cases increase and a second global wave kicks up.
Lessons from the first wave
In the United States, COVID-19 hit different regions at different times and to different degrees. One of the areas hit first was Seattle.
“We jumped on top of this, we were evidence based, we put things in place very, very quickly,” said Julie Gralow, MD, professor at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle.
“We did a really good job keeping COVID out of our cancer centers,” Dr. Gralow said. “We learned how to be super safe, and to keep symptomatic people out of the building, and to limit the extra people they could bring with them. It’s all about the number of contacts you have.”
The story was different, though, for oncologists in several other countries, and sometimes it varied immensely within each nation.
“We treated fewer patients with cancer during the first wave,” says Dirk Arnold, MD, medical director of the Asklepios Tumor Center Hamburg (Germany), in an interview. “In part, this was because staff were quarantined and because we had a completely different infrastructure in all of the hospitals. But also fewer patients with cancer came to the clinic at all. A lot of resources were directed toward COVID-19.”
In Spain, telemedicine helped keep up with visits, but other areas felt the effect of COVID-19 patient loads.
“At least in the oncology department of our center, we have practically maintained 100% of visits, mostly by telephone,” says Dr. Arrué, “but the reality is that our country has not yet been prepared for telemedicine.”
Laura Mezquita, MD, of the department of medical oncology at Hospital Clinic de Barcelona, describes a more dramatic situation: “We have seen how some of our patients, especially with metastatic disease, have been dismissed for intensive care and life-support treatments, as well as specific treatments against COVID-19 (tocilizumab, remdesivir, etc.) due to the general health collapse of the former wave,” she said. She adds that specific oncologic populations, such as those with thoracic tumors, have been more affected.
Distress among oncologists
Many oncologists are still feeling stressed and fatigued after the first wave, just as a second string of outbreaks is on its way.
A survey presented at last month’s ESMO 2020 Congress found that, in July-August, moral distress was reported by one-third of the oncologists who responded, and more than half reported a feeling of exhaustion.
“The tiredness and team exhaustion is noticeable,” said Dr. Arnold. “We recently had a task force discussion about what will happen when we have a second wave and how the department and our services will adapt. It was clear that those who were at the very front in the first wave had only a limited desire to do that again in the second wave.”
Another concern: COVID-19’s effect on staffing levels.
“We have a population of young caregivers who are affected by the COVID-19 disease with an absenteeism rate that is quite unprecedented,” said Sophie Beaupère, general delegate of Unicancer since January.
She said that, in general, the absenteeism rate in the cancer centers averages 5%-6%, depending on the year. But that rate is now skyrocketing.
Stop-start cycle for surgery
As caregivers quarantined around the world, more than 10% of patients with cancer had treatment canceled or delayed during the first wave of the pandemic, according to another survey from ESMO, involving 109 oncologists from 18 countries.
Difficulties were reported for surgeries by 34% of the centers, but also difficulties with delivering chemotherapy (22% of centers), radiotherapy (13.7%), and therapy with checkpoint inhibitors (9.1%), monoclonal antibodies (9%), and oral targeted therapy (3.7%).
Stopping surgery is a real concern in France, noted Dr. Kahn, the National League Against Cancer chair. He says that in regions that were badly hit by COVID-19, “it was not possible to have access to the operating room for people who absolutely needed surgery; for example, patients with lung cancer that was still operable. Most of the recovery rooms were mobilized for resuscitation.”
There may be some solutions, suggested Thierry Breton, director general of the National Institute of Cancer in France. “We are getting prepared, with the health ministry, for a possible increase in hospital tension, which would lead to a situation where we would have to reschedule operations. Nationally, regionally, and locally, we are seeing how we can resume and prioritize surgeries that have not been done.”
Delays in cancer diagnosis
While COVID-19 affected treatment, many oncologists say the major impact of the first wave was a delay in diagnosing cancer. Some of this was a result of the suspension of cancer screening programs, but there was also fear among the general public about visiting clinics and hospitals during a pandemic.
“We didn’t do so well with cancer during the first wave here in the U.K.,” said Karol Sikora, PhD, MBBChir, professor of cancer medicine and founding dean at the University of Buckingham Medical School, London. “Cancer diagnostic pathways virtually stalled partly because patients didn’t seek help, but getting scans and biopsies was also very difficult. Even patients referred urgently under the ‘2-weeks-wait’ rule were turned down.”
In France, “the delay in diagnosis is indisputable,” said Dr. Kahn. “About 50% of the cancer diagnoses one would expect during this period were missed.”
“I am worried that there remains a major traffic jam that has not been caught up with, and, in the meantime, the health crisis is worsening,” he added.
In Seattle, Dr. Gralow said the first COVID-19 wave had little impact on treatment for breast cancer, but it was in screening for breast cancer “where things really got messed up.”
“Even though we’ve been fully ramped up again,” she said, concerns remain. To ensure that screening mammography is maintained, “we have spaced out the visits to keep our waiting rooms less populated, with a longer time between using the machine so we can clean it. To do this, we have extended operating hours and are now opening on Saturday.
“So we’re actually at 100% of our capacity, but I’m really nervous, though, that a lot of people put off their screening mammogram and aren’t going to come in and get it.
“Not only did people get the message to stay home and not do nonessential things, but I think a lot of people lost their health insurance when they lost their jobs,” she said, and without health insurance, they are not covered for cancer screening.
Looking ahead, with a plan
Many oncologists agree that access to care can and must be improved – and there were some positive moves.
“Some regimens changed during the first months of the pandemic, and I don’t see them going back to the way they were anytime soon,” said Dr. Testa. “The changes/adaptations that were made to minimize the chance of SARS-CoV-2 infection are still in place and will go on for a while. In this context, telemedicine helped a lot. The pandemic forced the stakeholders to step up and put it in place in March. And now it’s here to stay.”
The experience gained in the last several months has driven preparation for the next wave.
“We are not going to see the disorganization that we saw during the first wave,” said Florence Joly, MD, PhD, head of medical oncology at the Centre François Baclesse in Caen, France. “The difference between now and earlier this year is that COVID diagnostic tests are available. That was one of the problems in the first wave. We had no way to diagnose.”
On the East Coast of the United States, medical oncologist Charu Aggarwal, MD, MPH, is also optimistic: “I think we’re at a place where we can manage.”
“I believe if there was going to be a new wave of COVID-19 cases we would be: better psychologically prepared and better organized,” said Dr. Aggarwal, assistant professor of medicine in the hematology-oncology division at the University of Pennsylvania, Philadelphia. “We already have experience with all of the tools, we have telemedicine available, we have screening protocols available, we have testing, we are already universally masking, everyone’s hand-washing, so I do think that means we would be okay.”
Dr. Arnold agreed that “we are much better prepared than for the first wave, but … we have immense tasks in the area of patient management, the digitization of patient care, the clear allocation of resources when there is a second or third wave. In many areas of preparation, I believe, unfortunately, we are not as well positioned as we had actually hoped.”
The first wave of COVID hit cancer services in the United Kingdom particularly hard: One modeling study suggested that delays in cancer referrals will lead to thousands of additional deaths and tens of thousands of life-years lost.
“Cancer services are working at near normal levels now, but they are still fragile and could be severely compromised again if the NHS [National Health Service] gets flooded by COVID patients,” said Dr. Sikora.
The second wave may be different. “Although the number of infections has increased, the hospitalizations have only risen a little. Let’s see what happens,” he said in an interview. Since then, however, infections have continued to rise, and there has been an increase in hospitalizations. New social distancing measures in the United Kingdom were put into place on Oct. 12, with the aim of protecting the NHS from overload.
Dr. Arrué describes it this way: “The reality is that the ‘second wave’ has left behind the initial grief and shock that both patients and health professionals experienced when faced with something that, until now, we had only seen in the movies.” The second wave has led to new restrictions – including a partial lockdown since the beginning of October.
Dr. Aggarwal says her department recently had a conference with Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, about the impact of COVID-19 on oncology.
“I asked him what advice he’d give oncologists, and he said to go back to as much screening as you were doing previously as quickly as possible. That’s what must be relayed to our oncologists in the community – and also to primary care physicians – because they are often the ones who are ordering and championing the screening efforts.”
This article was originated by Aude Lecrubier, Medscape French edition, and developed by Zosia Chustecka, Medscape Oncology. With additional reporting by Kate Johnson, freelance medical journalist, Claudia Gottschling for Medscape Germany, Leoleli Schwartz for Medscape em português, Tim Locke for Medscape United Kingdom, and Carla Nieto Martínez, freelance medical journalist for Medscape Spanish edition.
This article first appeared on Medscape.com.
It’s not time to abandon routine screening mammography in average-risk women in their 40s
In the 1970s and early 1980s, population-based screening mammography was studied in numerous randomized control trials (RCTs), with the primary outcome of reduced breast cancer mortality. Although technology and the sensitivity of mammography in the 1980s was somewhat rudimentary compared with current screening, a meta-analysis of these RCTs demonstrated a clear mortality benefit for screening mammography.1 As a result, widespread population-based mammography was introduced in the mid-1980s in the United States and has become a standard for breast cancer screening.
Since that time, few RCTs of screening mammography versus observation have been conducted because of the ethical challenges of entering women into such studies as well as the difficulty and expense of long-term follow-up to measure the effect of screening on breast cancer mortality. Without ongoing RCTs of mammography, retrospective, observational, and computer simulation trials of the efficacy and harms of screening mammography have been conducted using proxy measures of mortality (such as stage at diagnosis), and some have questioned the overall benefit of screening mammography.2,3
To further complicate this controversy, some national guidelines have recommended against routinely recommending screening mammography for women aged 40 to 49 based on concerns that the harms (callbacks, benign breast biopsies, overdiagnosis) exceed the potential benefits (earlier diagnosis, possible decrease in needed treatments, reduced breast cancer mortality).4 This has resulted in a confusing morass of national recommendations with uncertainty regarding the question of whether to routinely offer screening mammography for women in their 40s at average risk for breast cancer.4-6
Recently, to address this question Duffy and colleagues conducted a large RCT of women in their 40s to evaluate the long-term effect of mammography on breast cancer mortality.7 Here, I review the study in depth and offer some guidance to clinicians and women struggling with screening decisions.
Breast cancer mortality significantly lower in the screening group
The RCT, known as the UK Age trial, was conducted in England, Wales, and Scotland and enrolled 160,921 women from 1990 through 1997.7 Women were randomly assigned in a 2:1 ratio to observation or annual screening mammogram beginning at age 39–41 until age 48. (In the United Kingdom, all women are screened starting at age 50.) Study enrollees were followed for a median of 22.8 years, and the primary outcome was breast cancer mortality.
The study results showed a 25% relative risk (RR) reduction in breast cancer mortality at 10 years of follow-up in the mammography group compared with the unscreened women (83 breast cancer deaths in the mammography group vs 219 in the observation group [RR, 0.75; 95% confidence interval (CI), 0.58–0.97; P = .029]). Based on the prevalence of breast cancer in women in their 40s, this 25% relative risk reduction translates into approximately 1 less death per 1,000 women who undergo routine screening in their 40s.
While there was no additional significant mortality reduction beyond 10 years of follow-up, as noted mammography is offered routinely starting at age 50 to all women in the United Kingdom. The authors concluded that “reducing the lower age limit for screening from 50 to 40 years [of age] could potentially reduce breast cancer mortality.”
Was overdiagnosis a concern? Another finding in this trial was related to overdiagnosis of breast cancer in the screened group. Overdiagnosis refers to mammographic-only diagnosis (that is, no clinical findings) of nonaggressive breast cancer, which would remain indolent and not harm the patient. The study results demonstrated essentially no overdiagnosis in women screened at age 40 compared with the unscreened group.
Continue to: Large trial, long follow-up are key strengths...
Large trial, long follow-up are key strengths
The UK Age trial’s primary strength is its study design: a large population-based RCT that included diverse participants with the critical study outcome for cancer screening (mortality). The study’s long-term follow-up is another key strength, since breast cancer mortality typically occurs 7 to 10 years after diagnosis. In addition, results were available for 99.9% of the women enrolled in the trial (that is, only 0.1% of women were lost to follow-up). Interestingly, the demonstrated mortality reduction with screening mammography for women in their 40s validates the mortality benefit demonstrated in other large RCTs of women in their 40s.1
Another strong point is that the study addresses the issue of whether screening women in their 40s results in overdiagnosis compared with women who start screening in their 50s. Further, this study validates a prior observational study that mammographic findings of nonprogressive cancers do not disappear, so nonaggressive cancers that present on mammography in women in their 40s still would be detected when women start screening in their 50s.8
Study limitations should be noted
The study has several limitations. For example, significant improvements have been made in breast cancer treatments that may mitigate against the positive impact of screening mammography. The impact of changed breast cancer management over the past 20 years could not be addressed with this study’s design since women would have been treated in the 1990s. In addition, substantial improvements have occurred in breast cancer screening standards (2 views vs the single view used in the study) and technology since the 1990s. Current mammography includes nearly uniform use of either digital mammography (DM) or digital breast tomosynthesis (DBT), both of which improve breast cancer detection for women in their 40s compared with the older film-screen technology. In addition, DBT reduces false-positive results by approximately 40%, resulting in fewer callbacks and biopsies. While improved cancer detection and reduced false-positive results are seen with DM and DBT, whether these technology improvements result in improved breast cancer mortality has not yet been sufficiently studied.
Perhaps the most important limitation in this study is that the women did not undergo routine risk assessment before trial entry to assure that they all were at “average risk.” As a result, both high- and average-risk women would have been included in this population-based trial. Without risk stratification, it remains uncertain whether the reduction in breast cancer mortality disproportionately exists within a high-risk subgroup (such as breast cancer gene mutation carriers).
Finally, the cost efficacy of routine screening mammography for women in their 40s was not evaluated in this study.
The UK Age trial in perspective
The good news is that there is the clear evidence that breast cancer mortality rates (deaths per 100,000) have decreased by about 40% over the past 50 years, likely due to improvements in breast cancer treatment and routine screening mammography.9 Breast cancer mortality reduction is particularly important because breast cancer remains the most common cancer and is the second leading cause of cancer death in women in the United States. In the past decade, considerable debate has arisen arguing whether this reduction in breast cancer mortality is due to improved treatments, routine screening mammography, or both. Authors of a retrospective trial in Australia, recently reviewed in OBG Management, suggested that the majority of improvement is due to improvements in treatment.3,10 However, as the authors pointed out, due to the trial’s retrospective design, causality only can be inferred. The current UK Age trial does add to the numerous prospective trials demonstrating mortality benefit for mammography in women in their 40s.11
What remains a challenge for clinicians, and for women struggling with the mammography question, is the absence of risk assessment in these long-term RCT trials as well as in the large retrospective database studies. Without risk stratification, these studies treated all the study population as “average risk.” Because breast cancer risk assessment is sporadically performed in clinical practice and there are no published RCTs of screening mammography in risk-assessed “average risk” women in their 40s, it remains uncertain whether the women benefiting from screening in their 40s are in a high-risk group or whether women of average risk in this age group also are benefiting from routine screening mammography.
Continue to: What’s next: Incorporate routine risk assessment into clinical practice...
What’s next: Incorporate routine risk assessment into clinical practice
It is not time to abandon screening mammography for all women in their 40s. Rather, routine risk assessment should be performed using one of many available validated or widely tested tools, a recommendation supported by the American College of Obstetricians and Gynecologists, the National Comprehensive Cancer Network, and the US Preventive Services Task Force.5,6,12
Ideally, these tools can be incorporated into an electronic health record and prepopulated using already available patient data (such as age, reproductive risk factors, current medications, breast density if available, and family history). Prepopulating available data into breast cancer risk calculators would allow clinicians to spend time on counseling women regarding breast cancer risk and appropriate screening methods. The TABLE provides a summary of useful breast cancer risk calculators and includes comments about their utility and significant limitations and benefits. In addition to breast cancer risk, the more comprehensive risk calculators (Tyrer-Cuzick and BOADICEA) allow calculation of ovarian cancer risk and gene mutation risk.
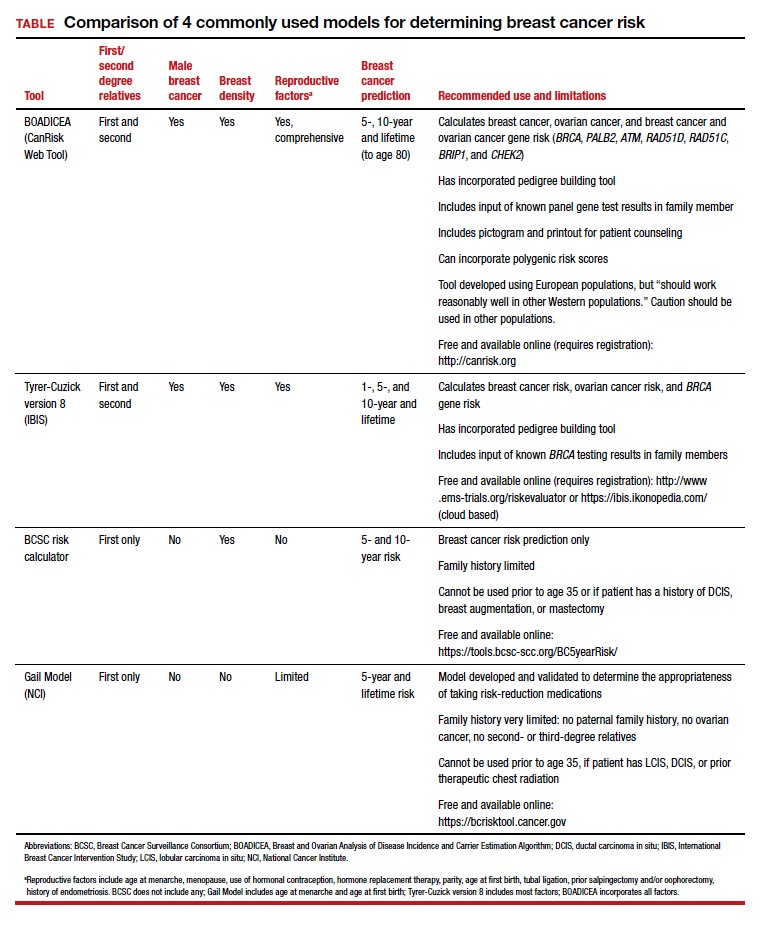
Routinely performing breast cancer risk assessment can guide discussions of screening mammography and can provide data for conducting a more individualized discussion on cancer genetic counseling and testing, risk reduction methods in high-risk women, and possible use of intensive breast cancer screening tools in identified high-risk women.
Ultimately, debating the question of whether all women should have routine breast cancer screening in their 40s should be passé. Ideally, all women should undergo breast cancer risk assessment in their 20s. Risk assessment results can then be used to guide the discussion of multiple potential interventions for women in their 40s (or earlier if appropriate), including routine screening mammography, cancer genetic counseling and testing in appropriate individuals, and intervention for women who are identified at high risk.
Absent breast cancer risk assessment, screening mammography still should be offered to women in their 40s, and the decision to proceed should be based on a discussion of risks, benefits, and the value the patient places on these factors.●
- Nelson HD, Fu R, Cantor A, et al. Effectiveness of breast cancer screening: systematic review and meta-analysis to update the 2009 US Preventive Services Task Force recommendation. Ann Intern Med. 2016;164:244-255.
- Bleyer A, Welch HG. Effect of three decades of screening mammography on breast-cancer incidence. N Engl J Med. 2012;367:1998-2005.
- Burton R, Stevenson C. Assessment of breast cancer mortality trends associated with mammographic screening and adjuvant therapy from 1986 to 2013 in the state of Victoria, Australia. JAMA Netw Open. 2020;3:e208249-e.
- Nelson HD, Cantor A, Humphrey L, et al. A systematic review to update the 2009 US Preventive Services Task Force recommendation. Evidence syntheses No. 124. AHRQ Publication No. 14-05201-EF-1. Rockville, MD: Agency for Healthcare Research and Quality; 2016.
- Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, version 3.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2018;16:1362-1389.
- ACOG Committee on Practice Bulletins–Gynecology. Breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130:e1-e16.
- Duffy SW, Vulkan D, Cuckle H, et al. Effect of mammographic screening from age 40 years on breast cancer mortality (UK Age trial): final results of a randomised, controlled trial. Lancet Oncol. 2020;21:1165-1172.
- Arleo EK, Monticciolo DL, Monsees B, et al. Persistent untreated screening-detected breast cancer: an argument against delaying screening or increasing the interval between screenings. J Am Coll Radiol. 2017;14:863-867.
- DeSantis CE, Ma J, Gaudet MM, et al. Breast cancer statistics, 2019. CA Cancer J Clin. 2019;69:438-451.
- Kaunitz AM. How effective is screening mammography for preventing breast cancer mortality? OBG Manag. 2020;32(8):17,49.
- Oeffinger KC, Fontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314:1599-1614.
- US Preventive Services Task Force; Owens DK, Davidson KW, Krist AH, et al. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer: US Preventive Services Task Force recommendation statement. JAMA. 2019;322:652-665.
In the 1970s and early 1980s, population-based screening mammography was studied in numerous randomized control trials (RCTs), with the primary outcome of reduced breast cancer mortality. Although technology and the sensitivity of mammography in the 1980s was somewhat rudimentary compared with current screening, a meta-analysis of these RCTs demonstrated a clear mortality benefit for screening mammography.1 As a result, widespread population-based mammography was introduced in the mid-1980s in the United States and has become a standard for breast cancer screening.
Since that time, few RCTs of screening mammography versus observation have been conducted because of the ethical challenges of entering women into such studies as well as the difficulty and expense of long-term follow-up to measure the effect of screening on breast cancer mortality. Without ongoing RCTs of mammography, retrospective, observational, and computer simulation trials of the efficacy and harms of screening mammography have been conducted using proxy measures of mortality (such as stage at diagnosis), and some have questioned the overall benefit of screening mammography.2,3
To further complicate this controversy, some national guidelines have recommended against routinely recommending screening mammography for women aged 40 to 49 based on concerns that the harms (callbacks, benign breast biopsies, overdiagnosis) exceed the potential benefits (earlier diagnosis, possible decrease in needed treatments, reduced breast cancer mortality).4 This has resulted in a confusing morass of national recommendations with uncertainty regarding the question of whether to routinely offer screening mammography for women in their 40s at average risk for breast cancer.4-6
Recently, to address this question Duffy and colleagues conducted a large RCT of women in their 40s to evaluate the long-term effect of mammography on breast cancer mortality.7 Here, I review the study in depth and offer some guidance to clinicians and women struggling with screening decisions.
Breast cancer mortality significantly lower in the screening group
The RCT, known as the UK Age trial, was conducted in England, Wales, and Scotland and enrolled 160,921 women from 1990 through 1997.7 Women were randomly assigned in a 2:1 ratio to observation or annual screening mammogram beginning at age 39–41 until age 48. (In the United Kingdom, all women are screened starting at age 50.) Study enrollees were followed for a median of 22.8 years, and the primary outcome was breast cancer mortality.
The study results showed a 25% relative risk (RR) reduction in breast cancer mortality at 10 years of follow-up in the mammography group compared with the unscreened women (83 breast cancer deaths in the mammography group vs 219 in the observation group [RR, 0.75; 95% confidence interval (CI), 0.58–0.97; P = .029]). Based on the prevalence of breast cancer in women in their 40s, this 25% relative risk reduction translates into approximately 1 less death per 1,000 women who undergo routine screening in their 40s.
While there was no additional significant mortality reduction beyond 10 years of follow-up, as noted mammography is offered routinely starting at age 50 to all women in the United Kingdom. The authors concluded that “reducing the lower age limit for screening from 50 to 40 years [of age] could potentially reduce breast cancer mortality.”
Was overdiagnosis a concern? Another finding in this trial was related to overdiagnosis of breast cancer in the screened group. Overdiagnosis refers to mammographic-only diagnosis (that is, no clinical findings) of nonaggressive breast cancer, which would remain indolent and not harm the patient. The study results demonstrated essentially no overdiagnosis in women screened at age 40 compared with the unscreened group.
Continue to: Large trial, long follow-up are key strengths...
Large trial, long follow-up are key strengths
The UK Age trial’s primary strength is its study design: a large population-based RCT that included diverse participants with the critical study outcome for cancer screening (mortality). The study’s long-term follow-up is another key strength, since breast cancer mortality typically occurs 7 to 10 years after diagnosis. In addition, results were available for 99.9% of the women enrolled in the trial (that is, only 0.1% of women were lost to follow-up). Interestingly, the demonstrated mortality reduction with screening mammography for women in their 40s validates the mortality benefit demonstrated in other large RCTs of women in their 40s.1
Another strong point is that the study addresses the issue of whether screening women in their 40s results in overdiagnosis compared with women who start screening in their 50s. Further, this study validates a prior observational study that mammographic findings of nonprogressive cancers do not disappear, so nonaggressive cancers that present on mammography in women in their 40s still would be detected when women start screening in their 50s.8
Study limitations should be noted
The study has several limitations. For example, significant improvements have been made in breast cancer treatments that may mitigate against the positive impact of screening mammography. The impact of changed breast cancer management over the past 20 years could not be addressed with this study’s design since women would have been treated in the 1990s. In addition, substantial improvements have occurred in breast cancer screening standards (2 views vs the single view used in the study) and technology since the 1990s. Current mammography includes nearly uniform use of either digital mammography (DM) or digital breast tomosynthesis (DBT), both of which improve breast cancer detection for women in their 40s compared with the older film-screen technology. In addition, DBT reduces false-positive results by approximately 40%, resulting in fewer callbacks and biopsies. While improved cancer detection and reduced false-positive results are seen with DM and DBT, whether these technology improvements result in improved breast cancer mortality has not yet been sufficiently studied.
Perhaps the most important limitation in this study is that the women did not undergo routine risk assessment before trial entry to assure that they all were at “average risk.” As a result, both high- and average-risk women would have been included in this population-based trial. Without risk stratification, it remains uncertain whether the reduction in breast cancer mortality disproportionately exists within a high-risk subgroup (such as breast cancer gene mutation carriers).
Finally, the cost efficacy of routine screening mammography for women in their 40s was not evaluated in this study.
The UK Age trial in perspective
The good news is that there is the clear evidence that breast cancer mortality rates (deaths per 100,000) have decreased by about 40% over the past 50 years, likely due to improvements in breast cancer treatment and routine screening mammography.9 Breast cancer mortality reduction is particularly important because breast cancer remains the most common cancer and is the second leading cause of cancer death in women in the United States. In the past decade, considerable debate has arisen arguing whether this reduction in breast cancer mortality is due to improved treatments, routine screening mammography, or both. Authors of a retrospective trial in Australia, recently reviewed in OBG Management, suggested that the majority of improvement is due to improvements in treatment.3,10 However, as the authors pointed out, due to the trial’s retrospective design, causality only can be inferred. The current UK Age trial does add to the numerous prospective trials demonstrating mortality benefit for mammography in women in their 40s.11
What remains a challenge for clinicians, and for women struggling with the mammography question, is the absence of risk assessment in these long-term RCT trials as well as in the large retrospective database studies. Without risk stratification, these studies treated all the study population as “average risk.” Because breast cancer risk assessment is sporadically performed in clinical practice and there are no published RCTs of screening mammography in risk-assessed “average risk” women in their 40s, it remains uncertain whether the women benefiting from screening in their 40s are in a high-risk group or whether women of average risk in this age group also are benefiting from routine screening mammography.
Continue to: What’s next: Incorporate routine risk assessment into clinical practice...
What’s next: Incorporate routine risk assessment into clinical practice
It is not time to abandon screening mammography for all women in their 40s. Rather, routine risk assessment should be performed using one of many available validated or widely tested tools, a recommendation supported by the American College of Obstetricians and Gynecologists, the National Comprehensive Cancer Network, and the US Preventive Services Task Force.5,6,12
Ideally, these tools can be incorporated into an electronic health record and prepopulated using already available patient data (such as age, reproductive risk factors, current medications, breast density if available, and family history). Prepopulating available data into breast cancer risk calculators would allow clinicians to spend time on counseling women regarding breast cancer risk and appropriate screening methods. The TABLE provides a summary of useful breast cancer risk calculators and includes comments about their utility and significant limitations and benefits. In addition to breast cancer risk, the more comprehensive risk calculators (Tyrer-Cuzick and BOADICEA) allow calculation of ovarian cancer risk and gene mutation risk.

Routinely performing breast cancer risk assessment can guide discussions of screening mammography and can provide data for conducting a more individualized discussion on cancer genetic counseling and testing, risk reduction methods in high-risk women, and possible use of intensive breast cancer screening tools in identified high-risk women.
Ultimately, debating the question of whether all women should have routine breast cancer screening in their 40s should be passé. Ideally, all women should undergo breast cancer risk assessment in their 20s. Risk assessment results can then be used to guide the discussion of multiple potential interventions for women in their 40s (or earlier if appropriate), including routine screening mammography, cancer genetic counseling and testing in appropriate individuals, and intervention for women who are identified at high risk.
Absent breast cancer risk assessment, screening mammography still should be offered to women in their 40s, and the decision to proceed should be based on a discussion of risks, benefits, and the value the patient places on these factors.●
In the 1970s and early 1980s, population-based screening mammography was studied in numerous randomized control trials (RCTs), with the primary outcome of reduced breast cancer mortality. Although technology and the sensitivity of mammography in the 1980s was somewhat rudimentary compared with current screening, a meta-analysis of these RCTs demonstrated a clear mortality benefit for screening mammography.1 As a result, widespread population-based mammography was introduced in the mid-1980s in the United States and has become a standard for breast cancer screening.
Since that time, few RCTs of screening mammography versus observation have been conducted because of the ethical challenges of entering women into such studies as well as the difficulty and expense of long-term follow-up to measure the effect of screening on breast cancer mortality. Without ongoing RCTs of mammography, retrospective, observational, and computer simulation trials of the efficacy and harms of screening mammography have been conducted using proxy measures of mortality (such as stage at diagnosis), and some have questioned the overall benefit of screening mammography.2,3
To further complicate this controversy, some national guidelines have recommended against routinely recommending screening mammography for women aged 40 to 49 based on concerns that the harms (callbacks, benign breast biopsies, overdiagnosis) exceed the potential benefits (earlier diagnosis, possible decrease in needed treatments, reduced breast cancer mortality).4 This has resulted in a confusing morass of national recommendations with uncertainty regarding the question of whether to routinely offer screening mammography for women in their 40s at average risk for breast cancer.4-6
Recently, to address this question Duffy and colleagues conducted a large RCT of women in their 40s to evaluate the long-term effect of mammography on breast cancer mortality.7 Here, I review the study in depth and offer some guidance to clinicians and women struggling with screening decisions.
Breast cancer mortality significantly lower in the screening group
The RCT, known as the UK Age trial, was conducted in England, Wales, and Scotland and enrolled 160,921 women from 1990 through 1997.7 Women were randomly assigned in a 2:1 ratio to observation or annual screening mammogram beginning at age 39–41 until age 48. (In the United Kingdom, all women are screened starting at age 50.) Study enrollees were followed for a median of 22.8 years, and the primary outcome was breast cancer mortality.
The study results showed a 25% relative risk (RR) reduction in breast cancer mortality at 10 years of follow-up in the mammography group compared with the unscreened women (83 breast cancer deaths in the mammography group vs 219 in the observation group [RR, 0.75; 95% confidence interval (CI), 0.58–0.97; P = .029]). Based on the prevalence of breast cancer in women in their 40s, this 25% relative risk reduction translates into approximately 1 less death per 1,000 women who undergo routine screening in their 40s.
While there was no additional significant mortality reduction beyond 10 years of follow-up, as noted mammography is offered routinely starting at age 50 to all women in the United Kingdom. The authors concluded that “reducing the lower age limit for screening from 50 to 40 years [of age] could potentially reduce breast cancer mortality.”
Was overdiagnosis a concern? Another finding in this trial was related to overdiagnosis of breast cancer in the screened group. Overdiagnosis refers to mammographic-only diagnosis (that is, no clinical findings) of nonaggressive breast cancer, which would remain indolent and not harm the patient. The study results demonstrated essentially no overdiagnosis in women screened at age 40 compared with the unscreened group.
Continue to: Large trial, long follow-up are key strengths...
Large trial, long follow-up are key strengths
The UK Age trial’s primary strength is its study design: a large population-based RCT that included diverse participants with the critical study outcome for cancer screening (mortality). The study’s long-term follow-up is another key strength, since breast cancer mortality typically occurs 7 to 10 years after diagnosis. In addition, results were available for 99.9% of the women enrolled in the trial (that is, only 0.1% of women were lost to follow-up). Interestingly, the demonstrated mortality reduction with screening mammography for women in their 40s validates the mortality benefit demonstrated in other large RCTs of women in their 40s.1
Another strong point is that the study addresses the issue of whether screening women in their 40s results in overdiagnosis compared with women who start screening in their 50s. Further, this study validates a prior observational study that mammographic findings of nonprogressive cancers do not disappear, so nonaggressive cancers that present on mammography in women in their 40s still would be detected when women start screening in their 50s.8
Study limitations should be noted
The study has several limitations. For example, significant improvements have been made in breast cancer treatments that may mitigate against the positive impact of screening mammography. The impact of changed breast cancer management over the past 20 years could not be addressed with this study’s design since women would have been treated in the 1990s. In addition, substantial improvements have occurred in breast cancer screening standards (2 views vs the single view used in the study) and technology since the 1990s. Current mammography includes nearly uniform use of either digital mammography (DM) or digital breast tomosynthesis (DBT), both of which improve breast cancer detection for women in their 40s compared with the older film-screen technology. In addition, DBT reduces false-positive results by approximately 40%, resulting in fewer callbacks and biopsies. While improved cancer detection and reduced false-positive results are seen with DM and DBT, whether these technology improvements result in improved breast cancer mortality has not yet been sufficiently studied.
Perhaps the most important limitation in this study is that the women did not undergo routine risk assessment before trial entry to assure that they all were at “average risk.” As a result, both high- and average-risk women would have been included in this population-based trial. Without risk stratification, it remains uncertain whether the reduction in breast cancer mortality disproportionately exists within a high-risk subgroup (such as breast cancer gene mutation carriers).
Finally, the cost efficacy of routine screening mammography for women in their 40s was not evaluated in this study.
The UK Age trial in perspective
The good news is that there is the clear evidence that breast cancer mortality rates (deaths per 100,000) have decreased by about 40% over the past 50 years, likely due to improvements in breast cancer treatment and routine screening mammography.9 Breast cancer mortality reduction is particularly important because breast cancer remains the most common cancer and is the second leading cause of cancer death in women in the United States. In the past decade, considerable debate has arisen arguing whether this reduction in breast cancer mortality is due to improved treatments, routine screening mammography, or both. Authors of a retrospective trial in Australia, recently reviewed in OBG Management, suggested that the majority of improvement is due to improvements in treatment.3,10 However, as the authors pointed out, due to the trial’s retrospective design, causality only can be inferred. The current UK Age trial does add to the numerous prospective trials demonstrating mortality benefit for mammography in women in their 40s.11
What remains a challenge for clinicians, and for women struggling with the mammography question, is the absence of risk assessment in these long-term RCT trials as well as in the large retrospective database studies. Without risk stratification, these studies treated all the study population as “average risk.” Because breast cancer risk assessment is sporadically performed in clinical practice and there are no published RCTs of screening mammography in risk-assessed “average risk” women in their 40s, it remains uncertain whether the women benefiting from screening in their 40s are in a high-risk group or whether women of average risk in this age group also are benefiting from routine screening mammography.
Continue to: What’s next: Incorporate routine risk assessment into clinical practice...
What’s next: Incorporate routine risk assessment into clinical practice
It is not time to abandon screening mammography for all women in their 40s. Rather, routine risk assessment should be performed using one of many available validated or widely tested tools, a recommendation supported by the American College of Obstetricians and Gynecologists, the National Comprehensive Cancer Network, and the US Preventive Services Task Force.5,6,12
Ideally, these tools can be incorporated into an electronic health record and prepopulated using already available patient data (such as age, reproductive risk factors, current medications, breast density if available, and family history). Prepopulating available data into breast cancer risk calculators would allow clinicians to spend time on counseling women regarding breast cancer risk and appropriate screening methods. The TABLE provides a summary of useful breast cancer risk calculators and includes comments about their utility and significant limitations and benefits. In addition to breast cancer risk, the more comprehensive risk calculators (Tyrer-Cuzick and BOADICEA) allow calculation of ovarian cancer risk and gene mutation risk.

Routinely performing breast cancer risk assessment can guide discussions of screening mammography and can provide data for conducting a more individualized discussion on cancer genetic counseling and testing, risk reduction methods in high-risk women, and possible use of intensive breast cancer screening tools in identified high-risk women.
Ultimately, debating the question of whether all women should have routine breast cancer screening in their 40s should be passé. Ideally, all women should undergo breast cancer risk assessment in their 20s. Risk assessment results can then be used to guide the discussion of multiple potential interventions for women in their 40s (or earlier if appropriate), including routine screening mammography, cancer genetic counseling and testing in appropriate individuals, and intervention for women who are identified at high risk.
Absent breast cancer risk assessment, screening mammography still should be offered to women in their 40s, and the decision to proceed should be based on a discussion of risks, benefits, and the value the patient places on these factors.●
- Nelson HD, Fu R, Cantor A, et al. Effectiveness of breast cancer screening: systematic review and meta-analysis to update the 2009 US Preventive Services Task Force recommendation. Ann Intern Med. 2016;164:244-255.
- Bleyer A, Welch HG. Effect of three decades of screening mammography on breast-cancer incidence. N Engl J Med. 2012;367:1998-2005.
- Burton R, Stevenson C. Assessment of breast cancer mortality trends associated with mammographic screening and adjuvant therapy from 1986 to 2013 in the state of Victoria, Australia. JAMA Netw Open. 2020;3:e208249-e.
- Nelson HD, Cantor A, Humphrey L, et al. A systematic review to update the 2009 US Preventive Services Task Force recommendation. Evidence syntheses No. 124. AHRQ Publication No. 14-05201-EF-1. Rockville, MD: Agency for Healthcare Research and Quality; 2016.
- Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, version 3.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2018;16:1362-1389.
- ACOG Committee on Practice Bulletins–Gynecology. Breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130:e1-e16.
- Duffy SW, Vulkan D, Cuckle H, et al. Effect of mammographic screening from age 40 years on breast cancer mortality (UK Age trial): final results of a randomised, controlled trial. Lancet Oncol. 2020;21:1165-1172.
- Arleo EK, Monticciolo DL, Monsees B, et al. Persistent untreated screening-detected breast cancer: an argument against delaying screening or increasing the interval between screenings. J Am Coll Radiol. 2017;14:863-867.
- DeSantis CE, Ma J, Gaudet MM, et al. Breast cancer statistics, 2019. CA Cancer J Clin. 2019;69:438-451.
- Kaunitz AM. How effective is screening mammography for preventing breast cancer mortality? OBG Manag. 2020;32(8):17,49.
- Oeffinger KC, Fontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314:1599-1614.
- US Preventive Services Task Force; Owens DK, Davidson KW, Krist AH, et al. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer: US Preventive Services Task Force recommendation statement. JAMA. 2019;322:652-665.
- Nelson HD, Fu R, Cantor A, et al. Effectiveness of breast cancer screening: systematic review and meta-analysis to update the 2009 US Preventive Services Task Force recommendation. Ann Intern Med. 2016;164:244-255.
- Bleyer A, Welch HG. Effect of three decades of screening mammography on breast-cancer incidence. N Engl J Med. 2012;367:1998-2005.
- Burton R, Stevenson C. Assessment of breast cancer mortality trends associated with mammographic screening and adjuvant therapy from 1986 to 2013 in the state of Victoria, Australia. JAMA Netw Open. 2020;3:e208249-e.
- Nelson HD, Cantor A, Humphrey L, et al. A systematic review to update the 2009 US Preventive Services Task Force recommendation. Evidence syntheses No. 124. AHRQ Publication No. 14-05201-EF-1. Rockville, MD: Agency for Healthcare Research and Quality; 2016.
- Bevers TB, Helvie M, Bonaccio E, et al. Breast cancer screening and diagnosis, version 3.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2018;16:1362-1389.
- ACOG Committee on Practice Bulletins–Gynecology. Breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130:e1-e16.
- Duffy SW, Vulkan D, Cuckle H, et al. Effect of mammographic screening from age 40 years on breast cancer mortality (UK Age trial): final results of a randomised, controlled trial. Lancet Oncol. 2020;21:1165-1172.
- Arleo EK, Monticciolo DL, Monsees B, et al. Persistent untreated screening-detected breast cancer: an argument against delaying screening or increasing the interval between screenings. J Am Coll Radiol. 2017;14:863-867.
- DeSantis CE, Ma J, Gaudet MM, et al. Breast cancer statistics, 2019. CA Cancer J Clin. 2019;69:438-451.
- Kaunitz AM. How effective is screening mammography for preventing breast cancer mortality? OBG Manag. 2020;32(8):17,49.
- Oeffinger KC, Fontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314:1599-1614.
- US Preventive Services Task Force; Owens DK, Davidson KW, Krist AH, et al. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer: US Preventive Services Task Force recommendation statement. JAMA. 2019;322:652-665.
Pathologic CR in HER2+ breast cancer predicts long-term survival
In fact, for the majority of women, pCR appears to be a marker of cure.
The trial was conducted among 455 women with HER2-positive breast cancer tumors measuring at least 2 cm who were randomized to neoadjuvant trastuzumab, lapatinib, or both drugs in combination, each together with paclitaxel, followed by more chemotherapy and more of the same targeted therapy after surgery.
Relative to trastuzumab alone, trastuzumab plus lapatinib improved rates of pCR, as shown by data published in The Lancet in 2012. However, the dual therapy did not significantly prolong event-free or overall survival, according to data published in The Lancet Oncology in 2014. Findings were similar in an update at a median follow-up of 6.7 years, published in the European Journal of Cancer in 2019.
Study investigator Paolo Nuciforo, MD, PhD, of the Vall d’Hebron Institute of Oncology in Barcelona, reported the trial’s final results, now at a median follow-up of 9.7 years, at the 12th European Breast Cancer Conference.
There were no significant differences in 9-year outcomes by specific HER2-targeted therapy. However, in a landmark analysis among women who were event free and still on follow-up 30 weeks after randomization, those achieving pCR with any of the therapies were 52% less likely to experience events and 63% less likely to die. Benefit was greatest in the subset of patients with hormone receptor–negative disease.
“The long-term follow-up confirms that, independent of the treatment regimen that we use – in this case, the dual blockade was with lapatinib, but similar results can be expected with other dual blockade – the pCR is a very robust surrogate biomarker of long-term survival,” Dr. Nuciforo commented in a press conference, noting that dual trastuzumab and pertuzumab has emerged as the standard of care.
“If we really pay attention to the curve, it’s maybe interesting to see that, after year 6, we actually don’t see any events in the pCR population. So this means that these patients are almost cured. We cannot say the word ‘cure’ in cancer, but it’s very reassuring to see the long-term survival analysis support the use of pCR as an endpoint,” he elaborated.
“Our results support the design of future trial concepts in HER2-positive early breast cancer which use pCR as an early efficacy readout of long-term benefit to escalate or deescalate therapy, particularly for hormone receptor–negative tumors,” Dr. Nuciforo concluded.
Support for current practice
“The study lends support for the current practice of risk-stratifying by pCR as well as making treatment decisions regarding T-DM1 [trastuzumab emtansine], and there hasn’t been a big change between 5-year and 9-year outcomes,” Lisa A. Carey, MD, of the University of North Carolina at Chapel Hill Lineberger Comprehensive Cancer Center, commented in an interview.
The lack of late events in the group with pCR technically meets the definition of cure, Dr. Carey said. “I think it speaks to the relatively early relapse risk in HER2-positive breast cancer and the impact of anti-HER2 therapy that carries forward. In general, these are findings similar to long-term findings of other trials and I suspect will be the same for any regimen.”
Although the analysis of dual lapatinib-trastuzumab therapy was underpowered, the trends seen align with favorable results in the adjuvant APHINITY trial (which combined trastuzumab with pertuzumab) and the neoadjuvant CALGB 40601 trial (which combined trastuzumab with lapatinib), according to Dr. Carey. “There has been a trend in every other study [of dual therapy] performed, so this is consistent.”
Study details
NeoALTTO is noteworthy for having the longest follow-up among all neoadjuvant studies of dual HER2 blockade in early breast cancer, Dr. Nuciforo said.
He reported no significant difference in survival between the treatment arms at 9 years.
The 9-year rate of event-free survival was 69% with lapatinib-trastuzumab, 63% with lapatinib alone, and 65% with trastuzumab alone. The corresponding 9-year rates of overall survival were 80%, 77%, and 76%, respectively.
However, there were significant differences in event-free and overall survival among women who achieved pCR and those who did not.
“pCR was achieved for almost twice as many patients treated with dual HER2 blockade, compared with patients in the single-agent arms,” Dr. Nuciforo pointed out. The pCR rate was 51.3% with lapatinib-trastuzumab, 24.7% with lapatinib alone, and 29.5% with trastuzumab alone.
Relative to peers who did not achieve pCR, women who did had better 9-year event-free survival (77% vs. 61%; adjusted hazard ratio, 0.48; P = .0008). The benefit was stronger in hormone receptor–negative disease (HR, 0.43; P = .002) than in hormone receptor–positive disease (HR, 0.60; P = .15).
The pattern was similar for overall survival at 9 years – 88% in those who achieved a pCR and 72% in those who did not (adjusted HR, 0.37; P = .0004). Again, greater benefit was seen in hormone receptor–negative disease (HR, 0.33; P = .002) than in hormone receptor–positive disease (HR, 0.44; P = .09).
“Biomarker-driven approaches may improve selection of those patients who are more likely to respond to anti-HER2 therapies,” Dr. Nuciforo proposed.
From 6 years onward, there were no additional fatal adverse events or nonfatal serious adverse events recorded, and no additional primary cardiac endpoints were recorded.
The study was funded by Novartis. Dr. Nuciforo and Dr. Carey disclosed no conflicts of interest.
SOURCE: Nuciforo P et al. EBCC-12 Virtual Conference, Abstract 23.
In fact, for the majority of women, pCR appears to be a marker of cure.
The trial was conducted among 455 women with HER2-positive breast cancer tumors measuring at least 2 cm who were randomized to neoadjuvant trastuzumab, lapatinib, or both drugs in combination, each together with paclitaxel, followed by more chemotherapy and more of the same targeted therapy after surgery.
Relative to trastuzumab alone, trastuzumab plus lapatinib improved rates of pCR, as shown by data published in The Lancet in 2012. However, the dual therapy did not significantly prolong event-free or overall survival, according to data published in The Lancet Oncology in 2014. Findings were similar in an update at a median follow-up of 6.7 years, published in the European Journal of Cancer in 2019.
Study investigator Paolo Nuciforo, MD, PhD, of the Vall d’Hebron Institute of Oncology in Barcelona, reported the trial’s final results, now at a median follow-up of 9.7 years, at the 12th European Breast Cancer Conference.
There were no significant differences in 9-year outcomes by specific HER2-targeted therapy. However, in a landmark analysis among women who were event free and still on follow-up 30 weeks after randomization, those achieving pCR with any of the therapies were 52% less likely to experience events and 63% less likely to die. Benefit was greatest in the subset of patients with hormone receptor–negative disease.
“The long-term follow-up confirms that, independent of the treatment regimen that we use – in this case, the dual blockade was with lapatinib, but similar results can be expected with other dual blockade – the pCR is a very robust surrogate biomarker of long-term survival,” Dr. Nuciforo commented in a press conference, noting that dual trastuzumab and pertuzumab has emerged as the standard of care.
“If we really pay attention to the curve, it’s maybe interesting to see that, after year 6, we actually don’t see any events in the pCR population. So this means that these patients are almost cured. We cannot say the word ‘cure’ in cancer, but it’s very reassuring to see the long-term survival analysis support the use of pCR as an endpoint,” he elaborated.
“Our results support the design of future trial concepts in HER2-positive early breast cancer which use pCR as an early efficacy readout of long-term benefit to escalate or deescalate therapy, particularly for hormone receptor–negative tumors,” Dr. Nuciforo concluded.
Support for current practice
“The study lends support for the current practice of risk-stratifying by pCR as well as making treatment decisions regarding T-DM1 [trastuzumab emtansine], and there hasn’t been a big change between 5-year and 9-year outcomes,” Lisa A. Carey, MD, of the University of North Carolina at Chapel Hill Lineberger Comprehensive Cancer Center, commented in an interview.
The lack of late events in the group with pCR technically meets the definition of cure, Dr. Carey said. “I think it speaks to the relatively early relapse risk in HER2-positive breast cancer and the impact of anti-HER2 therapy that carries forward. In general, these are findings similar to long-term findings of other trials and I suspect will be the same for any regimen.”
Although the analysis of dual lapatinib-trastuzumab therapy was underpowered, the trends seen align with favorable results in the adjuvant APHINITY trial (which combined trastuzumab with pertuzumab) and the neoadjuvant CALGB 40601 trial (which combined trastuzumab with lapatinib), according to Dr. Carey. “There has been a trend in every other study [of dual therapy] performed, so this is consistent.”
Study details
NeoALTTO is noteworthy for having the longest follow-up among all neoadjuvant studies of dual HER2 blockade in early breast cancer, Dr. Nuciforo said.
He reported no significant difference in survival between the treatment arms at 9 years.
The 9-year rate of event-free survival was 69% with lapatinib-trastuzumab, 63% with lapatinib alone, and 65% with trastuzumab alone. The corresponding 9-year rates of overall survival were 80%, 77%, and 76%, respectively.
However, there were significant differences in event-free and overall survival among women who achieved pCR and those who did not.
“pCR was achieved for almost twice as many patients treated with dual HER2 blockade, compared with patients in the single-agent arms,” Dr. Nuciforo pointed out. The pCR rate was 51.3% with lapatinib-trastuzumab, 24.7% with lapatinib alone, and 29.5% with trastuzumab alone.
Relative to peers who did not achieve pCR, women who did had better 9-year event-free survival (77% vs. 61%; adjusted hazard ratio, 0.48; P = .0008). The benefit was stronger in hormone receptor–negative disease (HR, 0.43; P = .002) than in hormone receptor–positive disease (HR, 0.60; P = .15).
The pattern was similar for overall survival at 9 years – 88% in those who achieved a pCR and 72% in those who did not (adjusted HR, 0.37; P = .0004). Again, greater benefit was seen in hormone receptor–negative disease (HR, 0.33; P = .002) than in hormone receptor–positive disease (HR, 0.44; P = .09).
“Biomarker-driven approaches may improve selection of those patients who are more likely to respond to anti-HER2 therapies,” Dr. Nuciforo proposed.
From 6 years onward, there were no additional fatal adverse events or nonfatal serious adverse events recorded, and no additional primary cardiac endpoints were recorded.
The study was funded by Novartis. Dr. Nuciforo and Dr. Carey disclosed no conflicts of interest.
SOURCE: Nuciforo P et al. EBCC-12 Virtual Conference, Abstract 23.
In fact, for the majority of women, pCR appears to be a marker of cure.
The trial was conducted among 455 women with HER2-positive breast cancer tumors measuring at least 2 cm who were randomized to neoadjuvant trastuzumab, lapatinib, or both drugs in combination, each together with paclitaxel, followed by more chemotherapy and more of the same targeted therapy after surgery.
Relative to trastuzumab alone, trastuzumab plus lapatinib improved rates of pCR, as shown by data published in The Lancet in 2012. However, the dual therapy did not significantly prolong event-free or overall survival, according to data published in The Lancet Oncology in 2014. Findings were similar in an update at a median follow-up of 6.7 years, published in the European Journal of Cancer in 2019.
Study investigator Paolo Nuciforo, MD, PhD, of the Vall d’Hebron Institute of Oncology in Barcelona, reported the trial’s final results, now at a median follow-up of 9.7 years, at the 12th European Breast Cancer Conference.
There were no significant differences in 9-year outcomes by specific HER2-targeted therapy. However, in a landmark analysis among women who were event free and still on follow-up 30 weeks after randomization, those achieving pCR with any of the therapies were 52% less likely to experience events and 63% less likely to die. Benefit was greatest in the subset of patients with hormone receptor–negative disease.
“The long-term follow-up confirms that, independent of the treatment regimen that we use – in this case, the dual blockade was with lapatinib, but similar results can be expected with other dual blockade – the pCR is a very robust surrogate biomarker of long-term survival,” Dr. Nuciforo commented in a press conference, noting that dual trastuzumab and pertuzumab has emerged as the standard of care.
“If we really pay attention to the curve, it’s maybe interesting to see that, after year 6, we actually don’t see any events in the pCR population. So this means that these patients are almost cured. We cannot say the word ‘cure’ in cancer, but it’s very reassuring to see the long-term survival analysis support the use of pCR as an endpoint,” he elaborated.
“Our results support the design of future trial concepts in HER2-positive early breast cancer which use pCR as an early efficacy readout of long-term benefit to escalate or deescalate therapy, particularly for hormone receptor–negative tumors,” Dr. Nuciforo concluded.
Support for current practice
“The study lends support for the current practice of risk-stratifying by pCR as well as making treatment decisions regarding T-DM1 [trastuzumab emtansine], and there hasn’t been a big change between 5-year and 9-year outcomes,” Lisa A. Carey, MD, of the University of North Carolina at Chapel Hill Lineberger Comprehensive Cancer Center, commented in an interview.
The lack of late events in the group with pCR technically meets the definition of cure, Dr. Carey said. “I think it speaks to the relatively early relapse risk in HER2-positive breast cancer and the impact of anti-HER2 therapy that carries forward. In general, these are findings similar to long-term findings of other trials and I suspect will be the same for any regimen.”
Although the analysis of dual lapatinib-trastuzumab therapy was underpowered, the trends seen align with favorable results in the adjuvant APHINITY trial (which combined trastuzumab with pertuzumab) and the neoadjuvant CALGB 40601 trial (which combined trastuzumab with lapatinib), according to Dr. Carey. “There has been a trend in every other study [of dual therapy] performed, so this is consistent.”
Study details
NeoALTTO is noteworthy for having the longest follow-up among all neoadjuvant studies of dual HER2 blockade in early breast cancer, Dr. Nuciforo said.
He reported no significant difference in survival between the treatment arms at 9 years.
The 9-year rate of event-free survival was 69% with lapatinib-trastuzumab, 63% with lapatinib alone, and 65% with trastuzumab alone. The corresponding 9-year rates of overall survival were 80%, 77%, and 76%, respectively.
However, there were significant differences in event-free and overall survival among women who achieved pCR and those who did not.
“pCR was achieved for almost twice as many patients treated with dual HER2 blockade, compared with patients in the single-agent arms,” Dr. Nuciforo pointed out. The pCR rate was 51.3% with lapatinib-trastuzumab, 24.7% with lapatinib alone, and 29.5% with trastuzumab alone.
Relative to peers who did not achieve pCR, women who did had better 9-year event-free survival (77% vs. 61%; adjusted hazard ratio, 0.48; P = .0008). The benefit was stronger in hormone receptor–negative disease (HR, 0.43; P = .002) than in hormone receptor–positive disease (HR, 0.60; P = .15).
The pattern was similar for overall survival at 9 years – 88% in those who achieved a pCR and 72% in those who did not (adjusted HR, 0.37; P = .0004). Again, greater benefit was seen in hormone receptor–negative disease (HR, 0.33; P = .002) than in hormone receptor–positive disease (HR, 0.44; P = .09).
“Biomarker-driven approaches may improve selection of those patients who are more likely to respond to anti-HER2 therapies,” Dr. Nuciforo proposed.
From 6 years onward, there were no additional fatal adverse events or nonfatal serious adverse events recorded, and no additional primary cardiac endpoints were recorded.
The study was funded by Novartis. Dr. Nuciforo and Dr. Carey disclosed no conflicts of interest.
SOURCE: Nuciforo P et al. EBCC-12 Virtual Conference, Abstract 23.
FROM EBCC-12 VIRTUAL CONFERENCE
Combined features of benign breast disease tied to breast cancer risk
“Benign breast disease is a key risk factor for breast cancer risk prediction,” commented presenting investigator Marta Román, PhD, of the Hospital del Mar Medical Research Institute in Barcelona. “Those women who have had a benign breast disease diagnosis have an increased risk that lasts for at least 20 years.”
To assess the combined influence of various attributes of benign breast disease, the investigators studied 629,087 women, aged 50-69 years, in Spain who underwent population-based mammographic breast cancer screening during 1994-2015 and did not have breast cancer at their prevalent (first) screen. The mean follow-up was 7.8 years.
Results showed that breast cancer risk was about three times higher for women with benign breast disease that was proliferative or that was detected on an incident screen, relative to peers with no benign breast disease. When combinations of factors were considered, breast cancer risk was most elevated – more than four times higher – for women with proliferative benign breast disease with atypia detected on an incident screen.
“We believe that these findings should be considered when discussing risk-based personalized screening strategies because these differences between prevalent and incident screens might be important if we want to personalize the screening, whether it’s the first time a woman comes to the screening program or a subsequent screen,” Dr. Román said.
Practice changing?
The study’s large size and population-based design, likely permitting capture of most biopsy results, are strengths, Mark David Pearlman, MD, of the University of Michigan, Ann Arbor, commented in an interview.
But its observational, retrospective nature opens the study up to biases, such as uncertainty as to how many women were symptomatic at the time of their mammogram and the likelihood of heightened monitoring after a biopsy showing hyperplasia, Dr. Pearlman cautioned.
“Moreover, the relative risk in this study for proliferative benign breast disease without atypia is substantially higher than prior observations of this group. This discrepancy was not discussed by the authors,” Dr. Pearlman said.
At present, women’s risk of breast cancer is predicted using well-validated models that include the question of prior breast biopsies, such as the Gail Model, the Tyrer-Cuzick model (IBIS tool), and the Breast and Ovarian Analysis of Disease Incidence and Carrier Estimation Algorithm, Dr. Pearlman noted.
“This study, without further validation within a model, would not change risk assessment,” he said, disagreeing with the investigators’ conclusions. “What I would say is that further study to determine how to use this observation to decide if any change in screening or management should occur would be more appropriate.”
Study details
The 629,087 women studied underwent 2,327,384 screens, Dr. Román reported. In total, screening detected 9,184 cases of benign breast disease and 9,431 breast cancers.
Breast cancer was diagnosed in 2.4% and 3.0% of women with benign breast disease detected on prevalent and incident screens, respectively, compared with 1.5% of women without any benign breast disease detected.
Elevation of breast cancer risk varied across benign breast disease subtype. Relative to peers without any benign disease, risk was significantly elevated for women with nonproliferative disease (adjusted hazard ratio, 1.95), proliferative disease without atypia (aHR, 3.19), and proliferative disease with atypia (aHR, 3.82).
Similarly, elevation of risk varied depending on the screening at which the benign disease was detected. Risk was significantly elevated when the disease was found at prevalent screens (aHR, 1.87) and more so when it was found at incident screens (aHR, 2.67).
There was no significant interaction of these two factors (P = .83). However, when combinations were considered, risk was highest for women with proliferative benign breast disease with atypia detected on incident screens (aHR, 4.35) or prevalent screens (aHR, 3.35), and women with proliferative benign breast disease without atypia detected on incident screens (aHR, 3.83).
This study was supported by grants from Instituto de Salud Carlos III FEDER and by the Research Network on Health Services in Chronic Diseases. Dr. Román and Dr. Pearlman disclosed no conflicts of interest.
SOURCE: Román M et al. EBCC-12 Virtual Conference, Abstract 15.
“Benign breast disease is a key risk factor for breast cancer risk prediction,” commented presenting investigator Marta Román, PhD, of the Hospital del Mar Medical Research Institute in Barcelona. “Those women who have had a benign breast disease diagnosis have an increased risk that lasts for at least 20 years.”
To assess the combined influence of various attributes of benign breast disease, the investigators studied 629,087 women, aged 50-69 years, in Spain who underwent population-based mammographic breast cancer screening during 1994-2015 and did not have breast cancer at their prevalent (first) screen. The mean follow-up was 7.8 years.
Results showed that breast cancer risk was about three times higher for women with benign breast disease that was proliferative or that was detected on an incident screen, relative to peers with no benign breast disease. When combinations of factors were considered, breast cancer risk was most elevated – more than four times higher – for women with proliferative benign breast disease with atypia detected on an incident screen.
“We believe that these findings should be considered when discussing risk-based personalized screening strategies because these differences between prevalent and incident screens might be important if we want to personalize the screening, whether it’s the first time a woman comes to the screening program or a subsequent screen,” Dr. Román said.
Practice changing?
The study’s large size and population-based design, likely permitting capture of most biopsy results, are strengths, Mark David Pearlman, MD, of the University of Michigan, Ann Arbor, commented in an interview.
But its observational, retrospective nature opens the study up to biases, such as uncertainty as to how many women were symptomatic at the time of their mammogram and the likelihood of heightened monitoring after a biopsy showing hyperplasia, Dr. Pearlman cautioned.
“Moreover, the relative risk in this study for proliferative benign breast disease without atypia is substantially higher than prior observations of this group. This discrepancy was not discussed by the authors,” Dr. Pearlman said.
At present, women’s risk of breast cancer is predicted using well-validated models that include the question of prior breast biopsies, such as the Gail Model, the Tyrer-Cuzick model (IBIS tool), and the Breast and Ovarian Analysis of Disease Incidence and Carrier Estimation Algorithm, Dr. Pearlman noted.
“This study, without further validation within a model, would not change risk assessment,” he said, disagreeing with the investigators’ conclusions. “What I would say is that further study to determine how to use this observation to decide if any change in screening or management should occur would be more appropriate.”
Study details
The 629,087 women studied underwent 2,327,384 screens, Dr. Román reported. In total, screening detected 9,184 cases of benign breast disease and 9,431 breast cancers.
Breast cancer was diagnosed in 2.4% and 3.0% of women with benign breast disease detected on prevalent and incident screens, respectively, compared with 1.5% of women without any benign breast disease detected.
Elevation of breast cancer risk varied across benign breast disease subtype. Relative to peers without any benign disease, risk was significantly elevated for women with nonproliferative disease (adjusted hazard ratio, 1.95), proliferative disease without atypia (aHR, 3.19), and proliferative disease with atypia (aHR, 3.82).
Similarly, elevation of risk varied depending on the screening at which the benign disease was detected. Risk was significantly elevated when the disease was found at prevalent screens (aHR, 1.87) and more so when it was found at incident screens (aHR, 2.67).
There was no significant interaction of these two factors (P = .83). However, when combinations were considered, risk was highest for women with proliferative benign breast disease with atypia detected on incident screens (aHR, 4.35) or prevalent screens (aHR, 3.35), and women with proliferative benign breast disease without atypia detected on incident screens (aHR, 3.83).
This study was supported by grants from Instituto de Salud Carlos III FEDER and by the Research Network on Health Services in Chronic Diseases. Dr. Román and Dr. Pearlman disclosed no conflicts of interest.
SOURCE: Román M et al. EBCC-12 Virtual Conference, Abstract 15.
“Benign breast disease is a key risk factor for breast cancer risk prediction,” commented presenting investigator Marta Román, PhD, of the Hospital del Mar Medical Research Institute in Barcelona. “Those women who have had a benign breast disease diagnosis have an increased risk that lasts for at least 20 years.”
To assess the combined influence of various attributes of benign breast disease, the investigators studied 629,087 women, aged 50-69 years, in Spain who underwent population-based mammographic breast cancer screening during 1994-2015 and did not have breast cancer at their prevalent (first) screen. The mean follow-up was 7.8 years.
Results showed that breast cancer risk was about three times higher for women with benign breast disease that was proliferative or that was detected on an incident screen, relative to peers with no benign breast disease. When combinations of factors were considered, breast cancer risk was most elevated – more than four times higher – for women with proliferative benign breast disease with atypia detected on an incident screen.
“We believe that these findings should be considered when discussing risk-based personalized screening strategies because these differences between prevalent and incident screens might be important if we want to personalize the screening, whether it’s the first time a woman comes to the screening program or a subsequent screen,” Dr. Román said.
Practice changing?
The study’s large size and population-based design, likely permitting capture of most biopsy results, are strengths, Mark David Pearlman, MD, of the University of Michigan, Ann Arbor, commented in an interview.
But its observational, retrospective nature opens the study up to biases, such as uncertainty as to how many women were symptomatic at the time of their mammogram and the likelihood of heightened monitoring after a biopsy showing hyperplasia, Dr. Pearlman cautioned.
“Moreover, the relative risk in this study for proliferative benign breast disease without atypia is substantially higher than prior observations of this group. This discrepancy was not discussed by the authors,” Dr. Pearlman said.
At present, women’s risk of breast cancer is predicted using well-validated models that include the question of prior breast biopsies, such as the Gail Model, the Tyrer-Cuzick model (IBIS tool), and the Breast and Ovarian Analysis of Disease Incidence and Carrier Estimation Algorithm, Dr. Pearlman noted.
“This study, without further validation within a model, would not change risk assessment,” he said, disagreeing with the investigators’ conclusions. “What I would say is that further study to determine how to use this observation to decide if any change in screening or management should occur would be more appropriate.”
Study details
The 629,087 women studied underwent 2,327,384 screens, Dr. Román reported. In total, screening detected 9,184 cases of benign breast disease and 9,431 breast cancers.
Breast cancer was diagnosed in 2.4% and 3.0% of women with benign breast disease detected on prevalent and incident screens, respectively, compared with 1.5% of women without any benign breast disease detected.
Elevation of breast cancer risk varied across benign breast disease subtype. Relative to peers without any benign disease, risk was significantly elevated for women with nonproliferative disease (adjusted hazard ratio, 1.95), proliferative disease without atypia (aHR, 3.19), and proliferative disease with atypia (aHR, 3.82).
Similarly, elevation of risk varied depending on the screening at which the benign disease was detected. Risk was significantly elevated when the disease was found at prevalent screens (aHR, 1.87) and more so when it was found at incident screens (aHR, 2.67).
There was no significant interaction of these two factors (P = .83). However, when combinations were considered, risk was highest for women with proliferative benign breast disease with atypia detected on incident screens (aHR, 4.35) or prevalent screens (aHR, 3.35), and women with proliferative benign breast disease without atypia detected on incident screens (aHR, 3.83).
This study was supported by grants from Instituto de Salud Carlos III FEDER and by the Research Network on Health Services in Chronic Diseases. Dr. Román and Dr. Pearlman disclosed no conflicts of interest.
SOURCE: Román M et al. EBCC-12 Virtual Conference, Abstract 15.
FROM EBCC-12 VIRTUAL CONFERENCE
Gene signature found similarly prognostic in ILC and IDC
ILC is enriched with features indicating low proliferative activity, noted investigator Otto Metzger, MD, of the Dana Farber Cancer Institute in Boston.
“Data from retrospective series have indicated no benefit with adjuvant chemotherapy for patients diagnosed with early-stage ILC,” he said. “It’s fair to say that chemotherapy decisions for patients with ILC remain controversial.”
With this in mind, Dr. Metzger and colleagues analyzed data for 5,313 women who underwent surgery for early-stage breast cancer (node-negative or up to three positive lymph nodes) and were risk-stratified to receive or skip adjuvant chemotherapy based on both clinical risk and the MammaPrint score for genomic risk. Fully 44% of women with ILC had discordant clinical and genomic risks.
With a median follow-up of 8.7 years, the 5-year rate of distant metastasis–free survival among all patients classified as genomic high risk was 92.1% in women with IDC and 88.1% in women with ILC, with overlapping 95% confidence intervals. Rates of distant metastasis–free survival for patients with genomic low risk were 96.4% in women with IDC and 96.6% in women with ILC, again with confidence intervals that overlapped.
The pattern was essentially the same for overall survival, and results carried over into 8-year outcomes as well.
“We believe that MammaPrint is a clinically useful test for patients diagnosed with ILC,” Dr. Metzger said. “There are similar survival outcomes for ILC and IDC when matched by genomic risk. This is an important message.”
It should be standard to omit chemotherapy for patients who have ILC classified as high clinical risk but low genomic risk by MammaPrint, Dr. Metzger recommended. “By contrast, MammaPrint should facilitate chemotherapy treatment decisions for patients diagnosed with ILC and high-risk MammaPrint,” he said.
Prognostic, but predictive?
“This is a well-designed prospective multicenter trial and provides the best evidence to date that MammaPrint is an important prognostic tool for ILC,” Todd Tuttle, MD, of University of Minnesota in Minneapolis, said in an interview.
Dr. Tuttle said he mainly uses the MammaPrint test and the OncoType 21-gene recurrence score to estimate prognosis for his patients with ILC.
These new data establish that MammaPrint is prognostic in ILC, but the value of MammaPrint’s genomic high risk result for making the decision about chemotherapy is still unclear, according to Dr. Tuttle.
“I don’t think we know whether MammaPrint can predict the benefit of chemotherapy for patients with stage I or II ILC,” he elaborated. “We need further high-quality studies such as this one to determine the best treatment strategies for ILC, which is a difficult breast cancer.”
Study details
Of the 5,313 patients studied, 487 had ILC (255 classic and 232 variant) and 4,826 had IDC according to central pathology assessment, Dr. Metzger reported.
MammaPrint classified 39% of the IDC group and 16% of the ILC group (10% of those with classic disease and 23% of those with variant disease) as genomically high risk for recurrence. The Adjuvant! Online tool classified 48.3% of ILC and 51.5% of IDC patients as clinically high risk.
Among the 44% of women with ILC having discordant genomic and clinical risk, discordance was usually due to the combination of low genomic risk and high clinical risk, seen in 38%.
The curves for 5-year distant metastasis–free survival stratified by genomic risk essentially overlapped for the IDC and ILC groups. Furthermore, there was no significant interaction of histologic type and genomic risk on this outcome (P = .547).
The 5-year rate of overall survival among women with genomic high risk was 95.6% in the IDC group and 93.5% in the ILC group. Among women with genomic low risk, 5-year overall survival was 98.1% in the IDC group and 97.7% in the ILC group, again with overlapping confidence intervals within each risk category.
The study was funded with support from the Breast Cancer Research Foundation. Dr. Metzger disclosed consulting fees from AbbVie, Genentech, Roche, and Pfizer. Dr. Tuttle disclosed no conflicts of interest.
SOURCE: Metzger O et al. EBCC-12 Virtual Conference. Abstract 6.
ILC is enriched with features indicating low proliferative activity, noted investigator Otto Metzger, MD, of the Dana Farber Cancer Institute in Boston.
“Data from retrospective series have indicated no benefit with adjuvant chemotherapy for patients diagnosed with early-stage ILC,” he said. “It’s fair to say that chemotherapy decisions for patients with ILC remain controversial.”
With this in mind, Dr. Metzger and colleagues analyzed data for 5,313 women who underwent surgery for early-stage breast cancer (node-negative or up to three positive lymph nodes) and were risk-stratified to receive or skip adjuvant chemotherapy based on both clinical risk and the MammaPrint score for genomic risk. Fully 44% of women with ILC had discordant clinical and genomic risks.
With a median follow-up of 8.7 years, the 5-year rate of distant metastasis–free survival among all patients classified as genomic high risk was 92.1% in women with IDC and 88.1% in women with ILC, with overlapping 95% confidence intervals. Rates of distant metastasis–free survival for patients with genomic low risk were 96.4% in women with IDC and 96.6% in women with ILC, again with confidence intervals that overlapped.
The pattern was essentially the same for overall survival, and results carried over into 8-year outcomes as well.
“We believe that MammaPrint is a clinically useful test for patients diagnosed with ILC,” Dr. Metzger said. “There are similar survival outcomes for ILC and IDC when matched by genomic risk. This is an important message.”
It should be standard to omit chemotherapy for patients who have ILC classified as high clinical risk but low genomic risk by MammaPrint, Dr. Metzger recommended. “By contrast, MammaPrint should facilitate chemotherapy treatment decisions for patients diagnosed with ILC and high-risk MammaPrint,” he said.
Prognostic, but predictive?
“This is a well-designed prospective multicenter trial and provides the best evidence to date that MammaPrint is an important prognostic tool for ILC,” Todd Tuttle, MD, of University of Minnesota in Minneapolis, said in an interview.
Dr. Tuttle said he mainly uses the MammaPrint test and the OncoType 21-gene recurrence score to estimate prognosis for his patients with ILC.
These new data establish that MammaPrint is prognostic in ILC, but the value of MammaPrint’s genomic high risk result for making the decision about chemotherapy is still unclear, according to Dr. Tuttle.
“I don’t think we know whether MammaPrint can predict the benefit of chemotherapy for patients with stage I or II ILC,” he elaborated. “We need further high-quality studies such as this one to determine the best treatment strategies for ILC, which is a difficult breast cancer.”
Study details
Of the 5,313 patients studied, 487 had ILC (255 classic and 232 variant) and 4,826 had IDC according to central pathology assessment, Dr. Metzger reported.
MammaPrint classified 39% of the IDC group and 16% of the ILC group (10% of those with classic disease and 23% of those with variant disease) as genomically high risk for recurrence. The Adjuvant! Online tool classified 48.3% of ILC and 51.5% of IDC patients as clinically high risk.
Among the 44% of women with ILC having discordant genomic and clinical risk, discordance was usually due to the combination of low genomic risk and high clinical risk, seen in 38%.
The curves for 5-year distant metastasis–free survival stratified by genomic risk essentially overlapped for the IDC and ILC groups. Furthermore, there was no significant interaction of histologic type and genomic risk on this outcome (P = .547).
The 5-year rate of overall survival among women with genomic high risk was 95.6% in the IDC group and 93.5% in the ILC group. Among women with genomic low risk, 5-year overall survival was 98.1% in the IDC group and 97.7% in the ILC group, again with overlapping confidence intervals within each risk category.
The study was funded with support from the Breast Cancer Research Foundation. Dr. Metzger disclosed consulting fees from AbbVie, Genentech, Roche, and Pfizer. Dr. Tuttle disclosed no conflicts of interest.
SOURCE: Metzger O et al. EBCC-12 Virtual Conference. Abstract 6.
ILC is enriched with features indicating low proliferative activity, noted investigator Otto Metzger, MD, of the Dana Farber Cancer Institute in Boston.
“Data from retrospective series have indicated no benefit with adjuvant chemotherapy for patients diagnosed with early-stage ILC,” he said. “It’s fair to say that chemotherapy decisions for patients with ILC remain controversial.”
With this in mind, Dr. Metzger and colleagues analyzed data for 5,313 women who underwent surgery for early-stage breast cancer (node-negative or up to three positive lymph nodes) and were risk-stratified to receive or skip adjuvant chemotherapy based on both clinical risk and the MammaPrint score for genomic risk. Fully 44% of women with ILC had discordant clinical and genomic risks.
With a median follow-up of 8.7 years, the 5-year rate of distant metastasis–free survival among all patients classified as genomic high risk was 92.1% in women with IDC and 88.1% in women with ILC, with overlapping 95% confidence intervals. Rates of distant metastasis–free survival for patients with genomic low risk were 96.4% in women with IDC and 96.6% in women with ILC, again with confidence intervals that overlapped.
The pattern was essentially the same for overall survival, and results carried over into 8-year outcomes as well.
“We believe that MammaPrint is a clinically useful test for patients diagnosed with ILC,” Dr. Metzger said. “There are similar survival outcomes for ILC and IDC when matched by genomic risk. This is an important message.”
It should be standard to omit chemotherapy for patients who have ILC classified as high clinical risk but low genomic risk by MammaPrint, Dr. Metzger recommended. “By contrast, MammaPrint should facilitate chemotherapy treatment decisions for patients diagnosed with ILC and high-risk MammaPrint,” he said.
Prognostic, but predictive?
“This is a well-designed prospective multicenter trial and provides the best evidence to date that MammaPrint is an important prognostic tool for ILC,” Todd Tuttle, MD, of University of Minnesota in Minneapolis, said in an interview.
Dr. Tuttle said he mainly uses the MammaPrint test and the OncoType 21-gene recurrence score to estimate prognosis for his patients with ILC.
These new data establish that MammaPrint is prognostic in ILC, but the value of MammaPrint’s genomic high risk result for making the decision about chemotherapy is still unclear, according to Dr. Tuttle.
“I don’t think we know whether MammaPrint can predict the benefit of chemotherapy for patients with stage I or II ILC,” he elaborated. “We need further high-quality studies such as this one to determine the best treatment strategies for ILC, which is a difficult breast cancer.”
Study details
Of the 5,313 patients studied, 487 had ILC (255 classic and 232 variant) and 4,826 had IDC according to central pathology assessment, Dr. Metzger reported.
MammaPrint classified 39% of the IDC group and 16% of the ILC group (10% of those with classic disease and 23% of those with variant disease) as genomically high risk for recurrence. The Adjuvant! Online tool classified 48.3% of ILC and 51.5% of IDC patients as clinically high risk.
Among the 44% of women with ILC having discordant genomic and clinical risk, discordance was usually due to the combination of low genomic risk and high clinical risk, seen in 38%.
The curves for 5-year distant metastasis–free survival stratified by genomic risk essentially overlapped for the IDC and ILC groups. Furthermore, there was no significant interaction of histologic type and genomic risk on this outcome (P = .547).
The 5-year rate of overall survival among women with genomic high risk was 95.6% in the IDC group and 93.5% in the ILC group. Among women with genomic low risk, 5-year overall survival was 98.1% in the IDC group and 97.7% in the ILC group, again with overlapping confidence intervals within each risk category.
The study was funded with support from the Breast Cancer Research Foundation. Dr. Metzger disclosed consulting fees from AbbVie, Genentech, Roche, and Pfizer. Dr. Tuttle disclosed no conflicts of interest.
SOURCE: Metzger O et al. EBCC-12 Virtual Conference. Abstract 6.
FROM EBCC-12 VIRTUAL CONFERENCE
Study advances personalized treatment for older breast cancer patients
Findings from the study were reported at the 12th European Breast Cancer Conference.
“Primary endocrine therapy is usually reserved for older, less fit, and frail women. Rates of use vary widely,” noted investigator Lynda Wyld, MBChB, PhD, of the University of Sheffield (England).
“Although there is no set threshold for who is suitable, some women are undoubtedly over- and undertreated for their breast cancer,” she added.
Dr. Wyld and colleagues undertook the Age Gap study among women older than 70 years with breast cancer recruited from 56 U.K. breast units during 2013-2018.
The main goals were to determine which women can be safely offered primary endocrine therapy as nonstandard care and to develop and test a tool to help women in this age group make treatment decisions.
The first component of the study was a multicenter, prospective cohort study of women with ER+ disease who were eligible for surgery. Results showed that breast cancer–specific mortality was greater with primary endocrine therapy than with surgery in the entire cohort. However, breast cancer–specific mortality was lower with primary endocrine therapy than with surgery in a cohort matched with propensity scores to achieve similar age, fitness, and frailty.
The second component of the study was a cluster-randomized controlled trial of women with operable breast cancer, most of whom had ER+ disease. Results showed that a decision support tool increased awareness of treatment options and readiness to decide. The tool also altered treatment choices, prompting a larger share of patients with ER+ disease to choose primary endocrine therapy.
Prospective cohort study
The prospective observational study was conducted in 2,854 women with ER+ disease who were eligible for surgery and treated in usual practice. Most women (n = 2,354) were treated with surgery (followed by antiestrogen therapy), while the rest received primary endocrine therapy (n = 500).
In the entire cohort, patients undergoing surgery were younger, had a lower level of comorbidity, and were less often frail. But these characteristics were generally similar in a propensity-matched cohort of 672 patients.
At a median follow-up of 52 months, overall and breast cancer–specific survival were significantly poorer with primary endocrine therapy versus surgery in the entire cohort but not in the propensity-matched cohort.
In the entire cohort, the breast cancer–specific mortality was 9.5% with primary endocrine therapy and 4.9% with surgery. In the propensity-matched cohort, breast cancer–specific mortality was 3.1% and 6.6%, respectively.
The overall mortality was 41.8% with primary endocrine therapy and 14.6% with surgery in the entire cohort, but the gap narrowed to 34.5% and 25.6%, respectively, in the propensity-matched cohort.
In the latter, “although there is a slight divergence in overall survival and it’s likely that with longer-term follow-up this will become significant, at the moment, it isn’t,” Dr. Wyld commented.
Curves for breast cancer–specific survival basically overlapped until 5 years, when surgery started to show an advantage. The rate of locoregional recurrence or progression was low and not significantly different by treatment.
None of the women in the entire cohort died from surgery. “But it’s worth bearing in mind that these were all women selected for surgery, who were thought to be fit for it by their surgeons. The least fit women in this cohort will have obviously been offered primary endocrine therapy,” Dr. Wyld cautioned.
Although 19% of patients had a surgical complication, only 2.1% had a systemic surgical complication.
Cluster-randomized controlled trial
In the cluster-randomized controlled trial, researchers compared a decision support tool to usual care. The tool was developed using U.K. registry data from almost 30,000 older women and input from women in this age group on their preferred format and method of presentation, according to Dr. Wyld.
The tool consists of an algorithm available to clinicians online (for input of tumor stage and biology, comorbidities, and functional status) plus a booklet and outcome sheets for patients to take home after discussions that can be personalized to their particulars.
Intention-to-treat analyses were based on 1,339 patients with operable breast cancer, 1,161 of whom had ER+ disease. Per-protocol analyses were based on the subset of 449 patients who were offered a choice between surgery and primary endocrine therapy, presumably because they were less fit and frailer.
Results showed that, at 6 months, mean scores for global quality of life on the EORTC questionnaire did not differ between decision support and usual care in the intention-to-treat population (69.0 vs. 68.9; P = .900), but scores were more favorable with decision support in the per-protocol population (70.7 vs. 66.8; P = .044).
The tool also altered treatment choices, with a larger share of ER+ patients choosing primary endocrine therapy (21.0% vs. 15.4%; P = .029) but still having similar disease outcomes.
Although ER+ patients in the decision support group more often selected primary endocrine therapy, at a median follow-up of 36 months, the groups did not differ significantly on overall survival, cause-specific survival, or time to recurrence in either intention-to-treat or per-protocol analyses.
Larger shares of women in the decision support group reported that they had adequate knowledge about the treatment options available to them (94% vs. 74%), were aware of the advantages and disadvantages of each option (91% vs. 76%), knew which option they preferred (96% vs. 91%), and were ready to make a decision (99% vs. 90%).
Applying results to practice
“Most women over the age of 70 are relatively fit, and the aim should be to treat them with surgery,” Dr. Wyld said. “For the less fit, a point is reached where the oncology benefits of surgery disappear and surgery may just cause harm. This threshold appears to be for women in their mid-80s with moderate to poor health.”
“Use of the Age Gap online tool may enhance shared decision-making for these women while increasing knowledge. And whilst it does seem to increase the use of primary endocrine therapy, this does not seem to have an adverse impact on survival at 36 months of follow-up,” she added.
“The study by Dr. Wyld and colleagues adds to the available literature regarding the scenarios in which some treatments may be omitted without impacting overall survival in older women with breast cancer,” Lesly A. Dossett, MD, of Michigan Medicine in Ann Arbor, commented in an interview.
In her own practice, Dr. Dossett emphasizes the generally favorable prognosis for older women with hormone receptor–positive breast cancer, she said. However, tools that help communicate risk and clarify the value of various therapies are welcome.
“The decision support tool appears to be a promising tool in helping to avoid treatments that are unlikely to benefit older women with breast cancer,” Dr. Dossett said. “The results will be widely applicable, as there is growing recognition that this patient population is at risk for overtreatment.”
The study was funded by the U.K. National Institute for Health Research programme grant for applied research. Dr. Wyld and Dr. Dossett said they had no relevant conflicts of interest.
SOURCES: Wyld L et al. EBCC-12 Virtual Congress. Abstract 8A and Abstract 8B.
Findings from the study were reported at the 12th European Breast Cancer Conference.
“Primary endocrine therapy is usually reserved for older, less fit, and frail women. Rates of use vary widely,” noted investigator Lynda Wyld, MBChB, PhD, of the University of Sheffield (England).
“Although there is no set threshold for who is suitable, some women are undoubtedly over- and undertreated for their breast cancer,” she added.
Dr. Wyld and colleagues undertook the Age Gap study among women older than 70 years with breast cancer recruited from 56 U.K. breast units during 2013-2018.
The main goals were to determine which women can be safely offered primary endocrine therapy as nonstandard care and to develop and test a tool to help women in this age group make treatment decisions.
The first component of the study was a multicenter, prospective cohort study of women with ER+ disease who were eligible for surgery. Results showed that breast cancer–specific mortality was greater with primary endocrine therapy than with surgery in the entire cohort. However, breast cancer–specific mortality was lower with primary endocrine therapy than with surgery in a cohort matched with propensity scores to achieve similar age, fitness, and frailty.
The second component of the study was a cluster-randomized controlled trial of women with operable breast cancer, most of whom had ER+ disease. Results showed that a decision support tool increased awareness of treatment options and readiness to decide. The tool also altered treatment choices, prompting a larger share of patients with ER+ disease to choose primary endocrine therapy.
Prospective cohort study
The prospective observational study was conducted in 2,854 women with ER+ disease who were eligible for surgery and treated in usual practice. Most women (n = 2,354) were treated with surgery (followed by antiestrogen therapy), while the rest received primary endocrine therapy (n = 500).
In the entire cohort, patients undergoing surgery were younger, had a lower level of comorbidity, and were less often frail. But these characteristics were generally similar in a propensity-matched cohort of 672 patients.
At a median follow-up of 52 months, overall and breast cancer–specific survival were significantly poorer with primary endocrine therapy versus surgery in the entire cohort but not in the propensity-matched cohort.
In the entire cohort, the breast cancer–specific mortality was 9.5% with primary endocrine therapy and 4.9% with surgery. In the propensity-matched cohort, breast cancer–specific mortality was 3.1% and 6.6%, respectively.
The overall mortality was 41.8% with primary endocrine therapy and 14.6% with surgery in the entire cohort, but the gap narrowed to 34.5% and 25.6%, respectively, in the propensity-matched cohort.
In the latter, “although there is a slight divergence in overall survival and it’s likely that with longer-term follow-up this will become significant, at the moment, it isn’t,” Dr. Wyld commented.
Curves for breast cancer–specific survival basically overlapped until 5 years, when surgery started to show an advantage. The rate of locoregional recurrence or progression was low and not significantly different by treatment.
None of the women in the entire cohort died from surgery. “But it’s worth bearing in mind that these were all women selected for surgery, who were thought to be fit for it by their surgeons. The least fit women in this cohort will have obviously been offered primary endocrine therapy,” Dr. Wyld cautioned.
Although 19% of patients had a surgical complication, only 2.1% had a systemic surgical complication.
Cluster-randomized controlled trial
In the cluster-randomized controlled trial, researchers compared a decision support tool to usual care. The tool was developed using U.K. registry data from almost 30,000 older women and input from women in this age group on their preferred format and method of presentation, according to Dr. Wyld.
The tool consists of an algorithm available to clinicians online (for input of tumor stage and biology, comorbidities, and functional status) plus a booklet and outcome sheets for patients to take home after discussions that can be personalized to their particulars.
Intention-to-treat analyses were based on 1,339 patients with operable breast cancer, 1,161 of whom had ER+ disease. Per-protocol analyses were based on the subset of 449 patients who were offered a choice between surgery and primary endocrine therapy, presumably because they were less fit and frailer.
Results showed that, at 6 months, mean scores for global quality of life on the EORTC questionnaire did not differ between decision support and usual care in the intention-to-treat population (69.0 vs. 68.9; P = .900), but scores were more favorable with decision support in the per-protocol population (70.7 vs. 66.8; P = .044).
The tool also altered treatment choices, with a larger share of ER+ patients choosing primary endocrine therapy (21.0% vs. 15.4%; P = .029) but still having similar disease outcomes.
Although ER+ patients in the decision support group more often selected primary endocrine therapy, at a median follow-up of 36 months, the groups did not differ significantly on overall survival, cause-specific survival, or time to recurrence in either intention-to-treat or per-protocol analyses.
Larger shares of women in the decision support group reported that they had adequate knowledge about the treatment options available to them (94% vs. 74%), were aware of the advantages and disadvantages of each option (91% vs. 76%), knew which option they preferred (96% vs. 91%), and were ready to make a decision (99% vs. 90%).
Applying results to practice
“Most women over the age of 70 are relatively fit, and the aim should be to treat them with surgery,” Dr. Wyld said. “For the less fit, a point is reached where the oncology benefits of surgery disappear and surgery may just cause harm. This threshold appears to be for women in their mid-80s with moderate to poor health.”
“Use of the Age Gap online tool may enhance shared decision-making for these women while increasing knowledge. And whilst it does seem to increase the use of primary endocrine therapy, this does not seem to have an adverse impact on survival at 36 months of follow-up,” she added.
“The study by Dr. Wyld and colleagues adds to the available literature regarding the scenarios in which some treatments may be omitted without impacting overall survival in older women with breast cancer,” Lesly A. Dossett, MD, of Michigan Medicine in Ann Arbor, commented in an interview.
In her own practice, Dr. Dossett emphasizes the generally favorable prognosis for older women with hormone receptor–positive breast cancer, she said. However, tools that help communicate risk and clarify the value of various therapies are welcome.
“The decision support tool appears to be a promising tool in helping to avoid treatments that are unlikely to benefit older women with breast cancer,” Dr. Dossett said. “The results will be widely applicable, as there is growing recognition that this patient population is at risk for overtreatment.”
The study was funded by the U.K. National Institute for Health Research programme grant for applied research. Dr. Wyld and Dr. Dossett said they had no relevant conflicts of interest.
SOURCES: Wyld L et al. EBCC-12 Virtual Congress. Abstract 8A and Abstract 8B.
Findings from the study were reported at the 12th European Breast Cancer Conference.
“Primary endocrine therapy is usually reserved for older, less fit, and frail women. Rates of use vary widely,” noted investigator Lynda Wyld, MBChB, PhD, of the University of Sheffield (England).
“Although there is no set threshold for who is suitable, some women are undoubtedly over- and undertreated for their breast cancer,” she added.
Dr. Wyld and colleagues undertook the Age Gap study among women older than 70 years with breast cancer recruited from 56 U.K. breast units during 2013-2018.
The main goals were to determine which women can be safely offered primary endocrine therapy as nonstandard care and to develop and test a tool to help women in this age group make treatment decisions.
The first component of the study was a multicenter, prospective cohort study of women with ER+ disease who were eligible for surgery. Results showed that breast cancer–specific mortality was greater with primary endocrine therapy than with surgery in the entire cohort. However, breast cancer–specific mortality was lower with primary endocrine therapy than with surgery in a cohort matched with propensity scores to achieve similar age, fitness, and frailty.
The second component of the study was a cluster-randomized controlled trial of women with operable breast cancer, most of whom had ER+ disease. Results showed that a decision support tool increased awareness of treatment options and readiness to decide. The tool also altered treatment choices, prompting a larger share of patients with ER+ disease to choose primary endocrine therapy.
Prospective cohort study
The prospective observational study was conducted in 2,854 women with ER+ disease who were eligible for surgery and treated in usual practice. Most women (n = 2,354) were treated with surgery (followed by antiestrogen therapy), while the rest received primary endocrine therapy (n = 500).
In the entire cohort, patients undergoing surgery were younger, had a lower level of comorbidity, and were less often frail. But these characteristics were generally similar in a propensity-matched cohort of 672 patients.
At a median follow-up of 52 months, overall and breast cancer–specific survival were significantly poorer with primary endocrine therapy versus surgery in the entire cohort but not in the propensity-matched cohort.
In the entire cohort, the breast cancer–specific mortality was 9.5% with primary endocrine therapy and 4.9% with surgery. In the propensity-matched cohort, breast cancer–specific mortality was 3.1% and 6.6%, respectively.
The overall mortality was 41.8% with primary endocrine therapy and 14.6% with surgery in the entire cohort, but the gap narrowed to 34.5% and 25.6%, respectively, in the propensity-matched cohort.
In the latter, “although there is a slight divergence in overall survival and it’s likely that with longer-term follow-up this will become significant, at the moment, it isn’t,” Dr. Wyld commented.
Curves for breast cancer–specific survival basically overlapped until 5 years, when surgery started to show an advantage. The rate of locoregional recurrence or progression was low and not significantly different by treatment.
None of the women in the entire cohort died from surgery. “But it’s worth bearing in mind that these were all women selected for surgery, who were thought to be fit for it by their surgeons. The least fit women in this cohort will have obviously been offered primary endocrine therapy,” Dr. Wyld cautioned.
Although 19% of patients had a surgical complication, only 2.1% had a systemic surgical complication.
Cluster-randomized controlled trial
In the cluster-randomized controlled trial, researchers compared a decision support tool to usual care. The tool was developed using U.K. registry data from almost 30,000 older women and input from women in this age group on their preferred format and method of presentation, according to Dr. Wyld.
The tool consists of an algorithm available to clinicians online (for input of tumor stage and biology, comorbidities, and functional status) plus a booklet and outcome sheets for patients to take home after discussions that can be personalized to their particulars.
Intention-to-treat analyses were based on 1,339 patients with operable breast cancer, 1,161 of whom had ER+ disease. Per-protocol analyses were based on the subset of 449 patients who were offered a choice between surgery and primary endocrine therapy, presumably because they were less fit and frailer.
Results showed that, at 6 months, mean scores for global quality of life on the EORTC questionnaire did not differ between decision support and usual care in the intention-to-treat population (69.0 vs. 68.9; P = .900), but scores were more favorable with decision support in the per-protocol population (70.7 vs. 66.8; P = .044).
The tool also altered treatment choices, with a larger share of ER+ patients choosing primary endocrine therapy (21.0% vs. 15.4%; P = .029) but still having similar disease outcomes.
Although ER+ patients in the decision support group more often selected primary endocrine therapy, at a median follow-up of 36 months, the groups did not differ significantly on overall survival, cause-specific survival, or time to recurrence in either intention-to-treat or per-protocol analyses.
Larger shares of women in the decision support group reported that they had adequate knowledge about the treatment options available to them (94% vs. 74%), were aware of the advantages and disadvantages of each option (91% vs. 76%), knew which option they preferred (96% vs. 91%), and were ready to make a decision (99% vs. 90%).
Applying results to practice
“Most women over the age of 70 are relatively fit, and the aim should be to treat them with surgery,” Dr. Wyld said. “For the less fit, a point is reached where the oncology benefits of surgery disappear and surgery may just cause harm. This threshold appears to be for women in their mid-80s with moderate to poor health.”
“Use of the Age Gap online tool may enhance shared decision-making for these women while increasing knowledge. And whilst it does seem to increase the use of primary endocrine therapy, this does not seem to have an adverse impact on survival at 36 months of follow-up,” she added.
“The study by Dr. Wyld and colleagues adds to the available literature regarding the scenarios in which some treatments may be omitted without impacting overall survival in older women with breast cancer,” Lesly A. Dossett, MD, of Michigan Medicine in Ann Arbor, commented in an interview.
In her own practice, Dr. Dossett emphasizes the generally favorable prognosis for older women with hormone receptor–positive breast cancer, she said. However, tools that help communicate risk and clarify the value of various therapies are welcome.
“The decision support tool appears to be a promising tool in helping to avoid treatments that are unlikely to benefit older women with breast cancer,” Dr. Dossett said. “The results will be widely applicable, as there is growing recognition that this patient population is at risk for overtreatment.”
The study was funded by the U.K. National Institute for Health Research programme grant for applied research. Dr. Wyld and Dr. Dossett said they had no relevant conflicts of interest.
SOURCES: Wyld L et al. EBCC-12 Virtual Congress. Abstract 8A and Abstract 8B.
FROM EBCC-12 VIRTUAL CONFERENCE
Breast Cancer Journal Scan: October 2020
Screening mammography has led to decreased breast cancer-specific mortality, and both digital mammography (DM) and digital breast tomosynthesis (DBT) are available modalities. A study by Lowry and colleagues evaluated DM and DBT performance in over 1,500,000 women age 40-79 without a prior history of breast cancer and demonstrated greater DBT benefit on initial screening exam. DBT benefit persisted on subsequent screening for women with heterogeneously dense breasts and scattered fibroglandular density, while no improvement in recall or cancer detection rates was seen for women with extremely dense breasts with DBT on subsequent exams. A physician survey showed 30% utilization of DBT, with higher uptake in academic settings and those with higher number of breast imagers and mammography units. Interestingly, 16% of respondents used mammographic density as a criterion to select patients to undergo DBT. Guidelines to help determine which women benefit from DBT would be a useful asset to clinicians and help optimize resources.
Although the majority of breast cancers are detected by screening mammography, a significant proportion are first noticed by a patient. Interval breast cancers, those detected between a normal mammogram and next scheduled mammogram, have more unfavorable features and worse survival compared with those detected by screening. Niraula et al found that interval breast cancers accounted for approximately 20% of cases, were over 6 times more likely to be higher grade, nearly 3 times more likely to be estrogen receptor-negative, and had a hazard ratio of 3.5 for breast cancer-specific mortality compared to screening-detected breast cancers. These findings are not entirely surprising as tumors with more aggressive biology are expected to have a faster onset and progression. Development of more personalized screening strategies may help address breast cancer heterogeneity.
Breast cancer diagnosed in women ≥70 years of age tends to be early stage and hormone receptor (HR)-positive. These cancers carry an excellent prognosis, and omission of routine sentinel lymph node biopsy (SLNB) and post-lumpectomy radiotherapy (assuming endocrine therapy is given) are acceptable strategies. However, these modalities are still utilized at fairly high rates nationally. Wang and colleagues conducted a qualitative study in women ≥70 years of age without a diagnosis of breast cancer, to evaluate treatment preferences in the setting of a hypothetical diagnosis of low-risk HR-positive breast cancer. A total of 40% stated they would elect to undergo SLNB, regarding the procedure as low-risk and providing prognostic information. Most women (73%) would choose to avoid radiation, due to perception of risk/benefit ratio and inconvenience. This study highlights the importance of effective communication regarding the excellent prognosis of these cancers in older women, and that de-escalation strategies are presented to reduce overtreatment and potential harms while achieving similar benefit.
Higher rates of genetic mutations (non-BRCA 1/2) have been observed in patients with breast cancer and another primary cancer compared to those with single primary breast cancer. Maxwell et al demonstrated rates of 7-9% compared to 4-5% for those with multiple primary breast cancer and single breast cancer, respectively. Further, they showed gene mutations (other than BRCA) are found in up to 25% of patients with breast cancer and another primary with their first breast cancer diagnosed ≤30 years old. Genetic testing is not a one-size fits all method and many patients are offered multigene panel testing. A multidisciplinary approach is key to identifying patients at higher risk, implementing effective screening and hopefully preventing future cancer development.
Erin Roesch, MD
The Cleveland Clinic
References:
Hardesty LA, Kreidler SM, Glueck DH. Digital breast tomosynthesis utilization in the United States: A survey of physician members of the society of breast imaging. J Am Coll Radiol 2016; 11S:R67-R73.
Bellio G, Marion R, Giudici F, Kus S, Tonutti M, Zanconati F, Bortul M. Interval breast cancer versus screen-detected cancer: comparison of clinicopathologic characteristics in a single-center analysis. Clin Breast Cancer. 2017;17:564-71.
Piccinin C, Panchal S, Watkins N, Kim, RH. An update on genetic risk assessment and prevention: the role of genetic testing panels in breast cancer. Expert Rev Anticancer Ther. 2019; 19:787-801.
Screening mammography has led to decreased breast cancer-specific mortality, and both digital mammography (DM) and digital breast tomosynthesis (DBT) are available modalities. A study by Lowry and colleagues evaluated DM and DBT performance in over 1,500,000 women age 40-79 without a prior history of breast cancer and demonstrated greater DBT benefit on initial screening exam. DBT benefit persisted on subsequent screening for women with heterogeneously dense breasts and scattered fibroglandular density, while no improvement in recall or cancer detection rates was seen for women with extremely dense breasts with DBT on subsequent exams. A physician survey showed 30% utilization of DBT, with higher uptake in academic settings and those with higher number of breast imagers and mammography units. Interestingly, 16% of respondents used mammographic density as a criterion to select patients to undergo DBT. Guidelines to help determine which women benefit from DBT would be a useful asset to clinicians and help optimize resources.
Although the majority of breast cancers are detected by screening mammography, a significant proportion are first noticed by a patient. Interval breast cancers, those detected between a normal mammogram and next scheduled mammogram, have more unfavorable features and worse survival compared with those detected by screening. Niraula et al found that interval breast cancers accounted for approximately 20% of cases, were over 6 times more likely to be higher grade, nearly 3 times more likely to be estrogen receptor-negative, and had a hazard ratio of 3.5 for breast cancer-specific mortality compared to screening-detected breast cancers. These findings are not entirely surprising as tumors with more aggressive biology are expected to have a faster onset and progression. Development of more personalized screening strategies may help address breast cancer heterogeneity.
Breast cancer diagnosed in women ≥70 years of age tends to be early stage and hormone receptor (HR)-positive. These cancers carry an excellent prognosis, and omission of routine sentinel lymph node biopsy (SLNB) and post-lumpectomy radiotherapy (assuming endocrine therapy is given) are acceptable strategies. However, these modalities are still utilized at fairly high rates nationally. Wang and colleagues conducted a qualitative study in women ≥70 years of age without a diagnosis of breast cancer, to evaluate treatment preferences in the setting of a hypothetical diagnosis of low-risk HR-positive breast cancer. A total of 40% stated they would elect to undergo SLNB, regarding the procedure as low-risk and providing prognostic information. Most women (73%) would choose to avoid radiation, due to perception of risk/benefit ratio and inconvenience. This study highlights the importance of effective communication regarding the excellent prognosis of these cancers in older women, and that de-escalation strategies are presented to reduce overtreatment and potential harms while achieving similar benefit.
Higher rates of genetic mutations (non-BRCA 1/2) have been observed in patients with breast cancer and another primary cancer compared to those with single primary breast cancer. Maxwell et al demonstrated rates of 7-9% compared to 4-5% for those with multiple primary breast cancer and single breast cancer, respectively. Further, they showed gene mutations (other than BRCA) are found in up to 25% of patients with breast cancer and another primary with their first breast cancer diagnosed ≤30 years old. Genetic testing is not a one-size fits all method and many patients are offered multigene panel testing. A multidisciplinary approach is key to identifying patients at higher risk, implementing effective screening and hopefully preventing future cancer development.
Erin Roesch, MD
The Cleveland Clinic
References:
Hardesty LA, Kreidler SM, Glueck DH. Digital breast tomosynthesis utilization in the United States: A survey of physician members of the society of breast imaging. J Am Coll Radiol 2016; 11S:R67-R73.
Bellio G, Marion R, Giudici F, Kus S, Tonutti M, Zanconati F, Bortul M. Interval breast cancer versus screen-detected cancer: comparison of clinicopathologic characteristics in a single-center analysis. Clin Breast Cancer. 2017;17:564-71.
Piccinin C, Panchal S, Watkins N, Kim, RH. An update on genetic risk assessment and prevention: the role of genetic testing panels in breast cancer. Expert Rev Anticancer Ther. 2019; 19:787-801.
Screening mammography has led to decreased breast cancer-specific mortality, and both digital mammography (DM) and digital breast tomosynthesis (DBT) are available modalities. A study by Lowry and colleagues evaluated DM and DBT performance in over 1,500,000 women age 40-79 without a prior history of breast cancer and demonstrated greater DBT benefit on initial screening exam. DBT benefit persisted on subsequent screening for women with heterogeneously dense breasts and scattered fibroglandular density, while no improvement in recall or cancer detection rates was seen for women with extremely dense breasts with DBT on subsequent exams. A physician survey showed 30% utilization of DBT, with higher uptake in academic settings and those with higher number of breast imagers and mammography units. Interestingly, 16% of respondents used mammographic density as a criterion to select patients to undergo DBT. Guidelines to help determine which women benefit from DBT would be a useful asset to clinicians and help optimize resources.
Although the majority of breast cancers are detected by screening mammography, a significant proportion are first noticed by a patient. Interval breast cancers, those detected between a normal mammogram and next scheduled mammogram, have more unfavorable features and worse survival compared with those detected by screening. Niraula et al found that interval breast cancers accounted for approximately 20% of cases, were over 6 times more likely to be higher grade, nearly 3 times more likely to be estrogen receptor-negative, and had a hazard ratio of 3.5 for breast cancer-specific mortality compared to screening-detected breast cancers. These findings are not entirely surprising as tumors with more aggressive biology are expected to have a faster onset and progression. Development of more personalized screening strategies may help address breast cancer heterogeneity.
Breast cancer diagnosed in women ≥70 years of age tends to be early stage and hormone receptor (HR)-positive. These cancers carry an excellent prognosis, and omission of routine sentinel lymph node biopsy (SLNB) and post-lumpectomy radiotherapy (assuming endocrine therapy is given) are acceptable strategies. However, these modalities are still utilized at fairly high rates nationally. Wang and colleagues conducted a qualitative study in women ≥70 years of age without a diagnosis of breast cancer, to evaluate treatment preferences in the setting of a hypothetical diagnosis of low-risk HR-positive breast cancer. A total of 40% stated they would elect to undergo SLNB, regarding the procedure as low-risk and providing prognostic information. Most women (73%) would choose to avoid radiation, due to perception of risk/benefit ratio and inconvenience. This study highlights the importance of effective communication regarding the excellent prognosis of these cancers in older women, and that de-escalation strategies are presented to reduce overtreatment and potential harms while achieving similar benefit.
Higher rates of genetic mutations (non-BRCA 1/2) have been observed in patients with breast cancer and another primary cancer compared to those with single primary breast cancer. Maxwell et al demonstrated rates of 7-9% compared to 4-5% for those with multiple primary breast cancer and single breast cancer, respectively. Further, they showed gene mutations (other than BRCA) are found in up to 25% of patients with breast cancer and another primary with their first breast cancer diagnosed ≤30 years old. Genetic testing is not a one-size fits all method and many patients are offered multigene panel testing. A multidisciplinary approach is key to identifying patients at higher risk, implementing effective screening and hopefully preventing future cancer development.
Erin Roesch, MD
The Cleveland Clinic
References:
Hardesty LA, Kreidler SM, Glueck DH. Digital breast tomosynthesis utilization in the United States: A survey of physician members of the society of breast imaging. J Am Coll Radiol 2016; 11S:R67-R73.
Bellio G, Marion R, Giudici F, Kus S, Tonutti M, Zanconati F, Bortul M. Interval breast cancer versus screen-detected cancer: comparison of clinicopathologic characteristics in a single-center analysis. Clin Breast Cancer. 2017;17:564-71.
Piccinin C, Panchal S, Watkins N, Kim, RH. An update on genetic risk assessment and prevention: the role of genetic testing panels in breast cancer. Expert Rev Anticancer Ther. 2019; 19:787-801.