User login
Breast cancer recurrence risk substantial after endocrine treatment
Women who stop adjuvant endocrine therapy after 5 years are still at substantial risk of distant recurrence over the next 15 years, even if their tumors were small, according to results of a recent meta-analysis of 88 clinical trials.
For women with T1N0 disease, the annual rate of distant recurrence was approximately 1% each year during 5-20 years, resulting in a cumulative risk of distant recurrence of 13%, authors of the meta-analysis reported (N Engl J Med. 2017 Nov. 8. doi: 10.1056/NEJMoa1701830).
Tumor diameter and nodal status was associated with the risk of distant recurrence during the later years and was approximately additive, with the risk increasing from 13% for T1N0 to 41% for T2N4–9 disease, wrote investigator Hongchao Pan, PhD, of the Nuffield Department of Population Health, University of Oxford, England, and his coauthors.
“Recognition of the magnitude of the long-term risks of ER-positive disease can help women and their health care professionals decide whether to extend therapy beyond 5 years and whether to persist if adverse events occur,” the authors wrote in the report.
The meta-analysis by Dr. Pan and his colleagues included 62,923 women with ER-positive breast cancer who were free of disease after 5 years of scheduled endocrine therapy.
They had hoped to identify a subgroup of women with a recurrence risks so small that the risk of additional side effects caused by extending endocrine therapy would outweigh any potential benefits of that additional treatment. However, the finding of measurable risk even in the women with T1N0 disease led them to recommend that extending endocrine therapy at least be considered for all patients.
“An absolute reduction of a few percentage points in the risk of distant metastases over the next 15 years might well be possible even for such low-risk women, with correspondingly greater absolute benefits for women with larger tumors or node-positive disease,” they wrote.
Whether reducing risk translates into improved survival remains to be seen.
As of now, “reliable trial evidence is not yet available” to confirm the clinical benefit of extending endocrine therapy beyond 5 years, the authors noted.
Cancer Research UK and others funded the study. Senior author Daniel F. Hayes, MD, reported grant support from Eli Lilly, Janssen Research & Development, Veridex, Puma, Pfizer, and AstraZeneca, among other disclosures. Full disclosures for all authors were provided on the NEJM website.
“This study reaffirms the potential for recurrences very late after the original diagnosis, an observation made with other datasets as well. This pattern of recurrence is most consistent with hormone-sensitive breast cancer,” William J. Gradishar, MD, said in an interview.
Dr. William J. Gradishar is the Betsy Bramsen Professor of Breast Oncology & professor of medicine at Northwestern University, Chicago.
“This study reaffirms the potential for recurrences very late after the original diagnosis, an observation made with other datasets as well. This pattern of recurrence is most consistent with hormone-sensitive breast cancer,” William J. Gradishar, MD, said in an interview.
Dr. William J. Gradishar is the Betsy Bramsen Professor of Breast Oncology & professor of medicine at Northwestern University, Chicago.
“This study reaffirms the potential for recurrences very late after the original diagnosis, an observation made with other datasets as well. This pattern of recurrence is most consistent with hormone-sensitive breast cancer,” William J. Gradishar, MD, said in an interview.
Dr. William J. Gradishar is the Betsy Bramsen Professor of Breast Oncology & professor of medicine at Northwestern University, Chicago.
Women who stop adjuvant endocrine therapy after 5 years are still at substantial risk of distant recurrence over the next 15 years, even if their tumors were small, according to results of a recent meta-analysis of 88 clinical trials.
For women with T1N0 disease, the annual rate of distant recurrence was approximately 1% each year during 5-20 years, resulting in a cumulative risk of distant recurrence of 13%, authors of the meta-analysis reported (N Engl J Med. 2017 Nov. 8. doi: 10.1056/NEJMoa1701830).
Tumor diameter and nodal status was associated with the risk of distant recurrence during the later years and was approximately additive, with the risk increasing from 13% for T1N0 to 41% for T2N4–9 disease, wrote investigator Hongchao Pan, PhD, of the Nuffield Department of Population Health, University of Oxford, England, and his coauthors.
“Recognition of the magnitude of the long-term risks of ER-positive disease can help women and their health care professionals decide whether to extend therapy beyond 5 years and whether to persist if adverse events occur,” the authors wrote in the report.
The meta-analysis by Dr. Pan and his colleagues included 62,923 women with ER-positive breast cancer who were free of disease after 5 years of scheduled endocrine therapy.
They had hoped to identify a subgroup of women with a recurrence risks so small that the risk of additional side effects caused by extending endocrine therapy would outweigh any potential benefits of that additional treatment. However, the finding of measurable risk even in the women with T1N0 disease led them to recommend that extending endocrine therapy at least be considered for all patients.
“An absolute reduction of a few percentage points in the risk of distant metastases over the next 15 years might well be possible even for such low-risk women, with correspondingly greater absolute benefits for women with larger tumors or node-positive disease,” they wrote.
Whether reducing risk translates into improved survival remains to be seen.
As of now, “reliable trial evidence is not yet available” to confirm the clinical benefit of extending endocrine therapy beyond 5 years, the authors noted.
Cancer Research UK and others funded the study. Senior author Daniel F. Hayes, MD, reported grant support from Eli Lilly, Janssen Research & Development, Veridex, Puma, Pfizer, and AstraZeneca, among other disclosures. Full disclosures for all authors were provided on the NEJM website.
Women who stop adjuvant endocrine therapy after 5 years are still at substantial risk of distant recurrence over the next 15 years, even if their tumors were small, according to results of a recent meta-analysis of 88 clinical trials.
For women with T1N0 disease, the annual rate of distant recurrence was approximately 1% each year during 5-20 years, resulting in a cumulative risk of distant recurrence of 13%, authors of the meta-analysis reported (N Engl J Med. 2017 Nov. 8. doi: 10.1056/NEJMoa1701830).
Tumor diameter and nodal status was associated with the risk of distant recurrence during the later years and was approximately additive, with the risk increasing from 13% for T1N0 to 41% for T2N4–9 disease, wrote investigator Hongchao Pan, PhD, of the Nuffield Department of Population Health, University of Oxford, England, and his coauthors.
“Recognition of the magnitude of the long-term risks of ER-positive disease can help women and their health care professionals decide whether to extend therapy beyond 5 years and whether to persist if adverse events occur,” the authors wrote in the report.
The meta-analysis by Dr. Pan and his colleagues included 62,923 women with ER-positive breast cancer who were free of disease after 5 years of scheduled endocrine therapy.
They had hoped to identify a subgroup of women with a recurrence risks so small that the risk of additional side effects caused by extending endocrine therapy would outweigh any potential benefits of that additional treatment. However, the finding of measurable risk even in the women with T1N0 disease led them to recommend that extending endocrine therapy at least be considered for all patients.
“An absolute reduction of a few percentage points in the risk of distant metastases over the next 15 years might well be possible even for such low-risk women, with correspondingly greater absolute benefits for women with larger tumors or node-positive disease,” they wrote.
Whether reducing risk translates into improved survival remains to be seen.
As of now, “reliable trial evidence is not yet available” to confirm the clinical benefit of extending endocrine therapy beyond 5 years, the authors noted.
Cancer Research UK and others funded the study. Senior author Daniel F. Hayes, MD, reported grant support from Eli Lilly, Janssen Research & Development, Veridex, Puma, Pfizer, and AstraZeneca, among other disclosures. Full disclosures for all authors were provided on the NEJM website.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Among women with early-stage, estrogen-receptor (ER)–positive breast cancer who stop adjuvant endocrine therapy after 5 years, distant recurrences happened at a steady rate over the ensuing 15 years.
Major finding: Distant recurrence risk ranged from 10% to 41%, depending on tumor diameter and nodal status (TN) and tumor grade.
Data source: A meta-analysis of 88 trials including 62,923 women with ER-positive breast cancer who were disease free after 5 years of scheduled endocrine therapy.
Disclosures: The study was funded by Cancer Research UK and others. Senior author Daniel F. Hayes, MD, reported grant support from Eli Lilly, Janssen Research & Development, Veridex, Puma, Pfizer, and AstraZeneca, among other disclosures. Full disclosures for all authors were provided on the NEJM website.
Seven days of opioids adequate for most hernia and other general surgery procedures
A 7-day limit on the initial opioid prescription may be sufficient for many common general surgery procedures, including hernia surgery and gynecologic procedures, findings of a large retrospective study suggest.
Rebecca E. Scully, MD, of the Center for Surgery and Public Health at Brigham and Women’s Hospital in Boston, and her associates examined opioid pain medication prescriptions and refills from records of the Military Health System Data Repository and the TRICARE insurance program of 215,140 opioid-naive patients. These patients were aged 18-64 years who underwent either cholecystectomy, appendectomy, inguinal hernia repair, anterior cruciate ligament reconstruction, rotator cuff tear repair, discectomy, mastectomy, or hysterectomy (JAMA Surg. 2017. doi: 10.1001/jamasurg.2017.3132). Only 20% of the covered individuals are active members of the U.S. military. The mean age was 40 years; 50% were male, and 60% were white.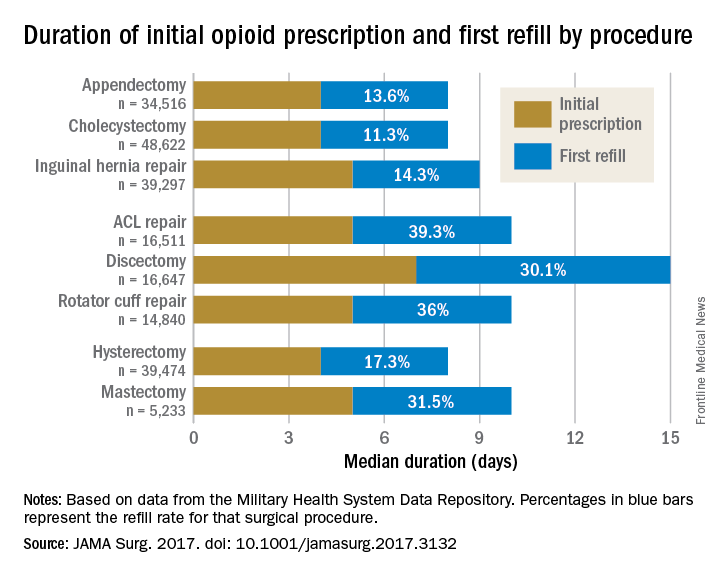
For appendectomy, cholecystectomy, and hysterectomy, the prescription was a median 4 days. For inguinal hernia repair, anterior cruciate ligament repair, rotator cuff repair, and mastectomy, the initial prescription was for 5 days. For discectomy, the median was 7 days.
Refill rates were the least at 11.3% for cholecystectomy and the most at 39.3% after anterior cruciate ligament repair. The time after the initial prescription until a refill was a median 6 days for appendectomy, cholecystectomy, and inguinal hernia repair, compared with a median 10 days for discectomy. The median duration of a refill prescription was 4 days for appendectomy, cholecystectomy, hernia repair, and hysterectomy versus 8 days for discectomy.
“Although 7 days appears to be more than adequate for many patients undergoing common general surgery and gynecologic procedures, prescription lengths likely should be extended to 10 days, particularly after common neurosurgical and musculoskeletal procedures, recognizing that as many as 40% of patients may still require one refill at a 7-day limit,” Dr. Scully and her associates said.
Although this study did not include rates of unused prescriptions or use of nonopioid pain relievers such as acetaminophen or NSAIDs, it did include a large population considered to be nationally representative “in many respects,” and it included a variety of procedures for which patients are commonly discharged to home, the researchers said.
The study was funded in part by the Department of Defense/Henry M. Jackson Foundation. The investigators had no conflict of interests. Adil H. Haider, MD, MPH, is deputy editor of JAMA Surgery, but he was not involved in any of the decisions regarding review of the manuscript or its acceptance.
A 7-day limit on the initial opioid prescription may be sufficient for many common general surgery procedures, including hernia surgery and gynecologic procedures, findings of a large retrospective study suggest.
Rebecca E. Scully, MD, of the Center for Surgery and Public Health at Brigham and Women’s Hospital in Boston, and her associates examined opioid pain medication prescriptions and refills from records of the Military Health System Data Repository and the TRICARE insurance program of 215,140 opioid-naive patients. These patients were aged 18-64 years who underwent either cholecystectomy, appendectomy, inguinal hernia repair, anterior cruciate ligament reconstruction, rotator cuff tear repair, discectomy, mastectomy, or hysterectomy (JAMA Surg. 2017. doi: 10.1001/jamasurg.2017.3132). Only 20% of the covered individuals are active members of the U.S. military. The mean age was 40 years; 50% were male, and 60% were white.
For appendectomy, cholecystectomy, and hysterectomy, the prescription was a median 4 days. For inguinal hernia repair, anterior cruciate ligament repair, rotator cuff repair, and mastectomy, the initial prescription was for 5 days. For discectomy, the median was 7 days.
Refill rates were the least at 11.3% for cholecystectomy and the most at 39.3% after anterior cruciate ligament repair. The time after the initial prescription until a refill was a median 6 days for appendectomy, cholecystectomy, and inguinal hernia repair, compared with a median 10 days for discectomy. The median duration of a refill prescription was 4 days for appendectomy, cholecystectomy, hernia repair, and hysterectomy versus 8 days for discectomy.
“Although 7 days appears to be more than adequate for many patients undergoing common general surgery and gynecologic procedures, prescription lengths likely should be extended to 10 days, particularly after common neurosurgical and musculoskeletal procedures, recognizing that as many as 40% of patients may still require one refill at a 7-day limit,” Dr. Scully and her associates said.
Although this study did not include rates of unused prescriptions or use of nonopioid pain relievers such as acetaminophen or NSAIDs, it did include a large population considered to be nationally representative “in many respects,” and it included a variety of procedures for which patients are commonly discharged to home, the researchers said.
The study was funded in part by the Department of Defense/Henry M. Jackson Foundation. The investigators had no conflict of interests. Adil H. Haider, MD, MPH, is deputy editor of JAMA Surgery, but he was not involved in any of the decisions regarding review of the manuscript or its acceptance.
A 7-day limit on the initial opioid prescription may be sufficient for many common general surgery procedures, including hernia surgery and gynecologic procedures, findings of a large retrospective study suggest.
Rebecca E. Scully, MD, of the Center for Surgery and Public Health at Brigham and Women’s Hospital in Boston, and her associates examined opioid pain medication prescriptions and refills from records of the Military Health System Data Repository and the TRICARE insurance program of 215,140 opioid-naive patients. These patients were aged 18-64 years who underwent either cholecystectomy, appendectomy, inguinal hernia repair, anterior cruciate ligament reconstruction, rotator cuff tear repair, discectomy, mastectomy, or hysterectomy (JAMA Surg. 2017. doi: 10.1001/jamasurg.2017.3132). Only 20% of the covered individuals are active members of the U.S. military. The mean age was 40 years; 50% were male, and 60% were white.
For appendectomy, cholecystectomy, and hysterectomy, the prescription was a median 4 days. For inguinal hernia repair, anterior cruciate ligament repair, rotator cuff repair, and mastectomy, the initial prescription was for 5 days. For discectomy, the median was 7 days.
Refill rates were the least at 11.3% for cholecystectomy and the most at 39.3% after anterior cruciate ligament repair. The time after the initial prescription until a refill was a median 6 days for appendectomy, cholecystectomy, and inguinal hernia repair, compared with a median 10 days for discectomy. The median duration of a refill prescription was 4 days for appendectomy, cholecystectomy, hernia repair, and hysterectomy versus 8 days for discectomy.
“Although 7 days appears to be more than adequate for many patients undergoing common general surgery and gynecologic procedures, prescription lengths likely should be extended to 10 days, particularly after common neurosurgical and musculoskeletal procedures, recognizing that as many as 40% of patients may still require one refill at a 7-day limit,” Dr. Scully and her associates said.
Although this study did not include rates of unused prescriptions or use of nonopioid pain relievers such as acetaminophen or NSAIDs, it did include a large population considered to be nationally representative “in many respects,” and it included a variety of procedures for which patients are commonly discharged to home, the researchers said.
The study was funded in part by the Department of Defense/Henry M. Jackson Foundation. The investigators had no conflict of interests. Adil H. Haider, MD, MPH, is deputy editor of JAMA Surgery, but he was not involved in any of the decisions regarding review of the manuscript or its acceptance.
FROM JAMA SURGERY
Key clinical point:
Major finding: The initial opioid prescription was a median 4 days for appendectomy and cholecystectomy, a median 5 days for inguinal hernia repair and anterior cruciate ligament and rotator cuff repair, and a median 7 days for discectomy.
Data source: A study of opioid prescriptions in 215,140 surgery patients aged 18-64 years.
Disclosures: The study was funded in part by the Department of Defense/Henry M. Jackson Foundation. The investigators had no conflict of interests. Adil H. Haider, MD, MPH, is deputy editor of JAMA Surgery, but he was not involved in any of the decisions regarding review of the manuscript or its acceptance.
New persistent opioid use common after cancer surgery
New and persistent opioid use is a common complication of surgery in patients with early-stage cancer, according to results of a retrospective cohort study.
The risk of new persistent opioid use was 10.4% (95% confidence interval, 10.1%-10.7%) among patients undergoing curative-intent cancer surgery, according to the report, which was based on examination of 68,463 deidentified insurance claims from employer health plans from 2010 to 2014.
“This problem requires changes to prescribing guidelines and patient counseling during the surveillance and survivorship phases of care,” wrote Jay Soong-Jin Lee, MD, and his colleagues at the University of Michigan, Ann Arbor (J Clin Oncol. 2017 Oct 19. doi: 10.1200/JCO.2017.74.1363).
One year after the surgery, patients who developed new persistent opioid use were still filling prescriptions at high daily opioid doses, equivalent to six hydrocodone 5-mg tablets per day, according Dr. Lee and his colleagues.
“This dose is similar to intermittent and chronic opioid users [in the insurance claim data], suggesting that patients with new persistent opioid use may transition to chronic opioid use,” they said in the study report.
Adjuvant chemotherapy was a “strong risk factor” for new persistent opioid use, they added, though use was still common among patients who had no adjuvant chemotherapy. Rates of new persistent opioid use ranged from 15% to 21% for adjuvant therapy patient groups, compared with 7%-11% for no advjuvant therapy, data show.
Previous studies suggested a 6%-8% risk of new persistent opioid use among surgical patients, but those studies either did not focus on cancer patients or excluded them entirely, Dr. Lee and his coauthors noted.
Strategies are needed to combat new persistent opioid use after curative-intent surgery, they added.
They recommended further study to develop evidence-based guidelines to reduce excessive opioid prescribing and screening tools to identify at-risk patients (e.g., those with psychosocial factors).
Surgeons should be more active in counseling patients on the potential risks of opioids and how to keep use to a minimum after surgery, they added.
“Given the high risk of new persistent opioid use in this population, physicians should consider universal precautions … including educating patients on safe use, storage, and disposal,” they wrote.
Dr. Lee disclosed no relationships relevant to the study, while several coauthors reported relationships with Neuros Medical, Merck, and Anesthesia Associates of Ann Arbor.
New and persistent opioid use is a common complication of surgery in patients with early-stage cancer, according to results of a retrospective cohort study.
The risk of new persistent opioid use was 10.4% (95% confidence interval, 10.1%-10.7%) among patients undergoing curative-intent cancer surgery, according to the report, which was based on examination of 68,463 deidentified insurance claims from employer health plans from 2010 to 2014.
“This problem requires changes to prescribing guidelines and patient counseling during the surveillance and survivorship phases of care,” wrote Jay Soong-Jin Lee, MD, and his colleagues at the University of Michigan, Ann Arbor (J Clin Oncol. 2017 Oct 19. doi: 10.1200/JCO.2017.74.1363).
One year after the surgery, patients who developed new persistent opioid use were still filling prescriptions at high daily opioid doses, equivalent to six hydrocodone 5-mg tablets per day, according Dr. Lee and his colleagues.
“This dose is similar to intermittent and chronic opioid users [in the insurance claim data], suggesting that patients with new persistent opioid use may transition to chronic opioid use,” they said in the study report.
Adjuvant chemotherapy was a “strong risk factor” for new persistent opioid use, they added, though use was still common among patients who had no adjuvant chemotherapy. Rates of new persistent opioid use ranged from 15% to 21% for adjuvant therapy patient groups, compared with 7%-11% for no advjuvant therapy, data show.
Previous studies suggested a 6%-8% risk of new persistent opioid use among surgical patients, but those studies either did not focus on cancer patients or excluded them entirely, Dr. Lee and his coauthors noted.
Strategies are needed to combat new persistent opioid use after curative-intent surgery, they added.
They recommended further study to develop evidence-based guidelines to reduce excessive opioid prescribing and screening tools to identify at-risk patients (e.g., those with psychosocial factors).
Surgeons should be more active in counseling patients on the potential risks of opioids and how to keep use to a minimum after surgery, they added.
“Given the high risk of new persistent opioid use in this population, physicians should consider universal precautions … including educating patients on safe use, storage, and disposal,” they wrote.
Dr. Lee disclosed no relationships relevant to the study, while several coauthors reported relationships with Neuros Medical, Merck, and Anesthesia Associates of Ann Arbor.
New and persistent opioid use is a common complication of surgery in patients with early-stage cancer, according to results of a retrospective cohort study.
The risk of new persistent opioid use was 10.4% (95% confidence interval, 10.1%-10.7%) among patients undergoing curative-intent cancer surgery, according to the report, which was based on examination of 68,463 deidentified insurance claims from employer health plans from 2010 to 2014.
“This problem requires changes to prescribing guidelines and patient counseling during the surveillance and survivorship phases of care,” wrote Jay Soong-Jin Lee, MD, and his colleagues at the University of Michigan, Ann Arbor (J Clin Oncol. 2017 Oct 19. doi: 10.1200/JCO.2017.74.1363).
One year after the surgery, patients who developed new persistent opioid use were still filling prescriptions at high daily opioid doses, equivalent to six hydrocodone 5-mg tablets per day, according Dr. Lee and his colleagues.
“This dose is similar to intermittent and chronic opioid users [in the insurance claim data], suggesting that patients with new persistent opioid use may transition to chronic opioid use,” they said in the study report.
Adjuvant chemotherapy was a “strong risk factor” for new persistent opioid use, they added, though use was still common among patients who had no adjuvant chemotherapy. Rates of new persistent opioid use ranged from 15% to 21% for adjuvant therapy patient groups, compared with 7%-11% for no advjuvant therapy, data show.
Previous studies suggested a 6%-8% risk of new persistent opioid use among surgical patients, but those studies either did not focus on cancer patients or excluded them entirely, Dr. Lee and his coauthors noted.
Strategies are needed to combat new persistent opioid use after curative-intent surgery, they added.
They recommended further study to develop evidence-based guidelines to reduce excessive opioid prescribing and screening tools to identify at-risk patients (e.g., those with psychosocial factors).
Surgeons should be more active in counseling patients on the potential risks of opioids and how to keep use to a minimum after surgery, they added.
“Given the high risk of new persistent opioid use in this population, physicians should consider universal precautions … including educating patients on safe use, storage, and disposal,” they wrote.
Dr. Lee disclosed no relationships relevant to the study, while several coauthors reported relationships with Neuros Medical, Merck, and Anesthesia Associates of Ann Arbor.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Prescribing guidelines and patient counseling need to change to combat new persistent opioid use, which authors confirmed is a common problem in patients undergoing surgery for early-stage cancer.
Major finding: The risk of new persistent opioid use was 10.4% (95% CI, 10.1%-10.7%) among patients undergoing curative-intent cancer surgery.
Data source: Retrospective cohort study based on examination of deidentified insurance claims from employer health plans from 2010 to 2014.
Disclosures: First author Jay Soong-Jin Lee, MD, had no relationships to disclose. Coauthors reported relationships with Neuros Medical and Merck and Anesthesia Associates of Ann Arbor.
Breast cancer screening: Is the controversy of benefits versus harms resolved?
Breast cancer is the most common cancer and the second leading cause of cancer death in women in the United States, with an estimated 252,710 new cases and 40,610 deaths in 2017.1 Breast cancer mortality is prevented by the use of regular screening mammography, as demonstrated by randomized controlled trials (20% reduction), incidence-based mortality studies (38% to 40% reduction), and service screening studies (48% to 49% reduction).2
Controversy continues, however, on when to start mammography screening, when to stop screening, and the frequency with which screening should be performed for women at average risk for breast cancer. Indeed, 3 national recommendations—written by the American College of Obstetricians and Gynecologists (ACOG), the American Cancer Society (ACS), and the US Preventive Services Task Force (USPSTF)—offer different guidelines for mammography screening (TABLE 1).2–4

There are 2 principal reasons for the controversy over screening:
- mammography has both benefits and harms, and individuals place differential weight on the importance of these relative to each other
- randomized controlled trials on screening mammography did not include all of the starting age, stopping age, and screening intervals that are included in screening recommendations.
New comparison of recommendations
An ongoing project funded by the National Cancer Institute, known as the Cancer Intervention and Surveillance Modeling Network (CISNET), models different starting and stopping ages and screening intervals for mammography to assess their impact on both benefits (mortality improvement, life-years gained) and harms (callbacks, benign breast biopsies). Recently, Arleo and colleagues used CISNET model data to compare the breast cancer screening recommendations from ACOG, the ACS, and the USPSTF, focusing on the differential effect on benefits and harms.5
Benefits vs harms of screening in perspective
Without question, the principal goal of cancer screening strategies is to effectively and efficiently reduce cancer mortality. Because mammography screening has both benefits and harms, a clear understanding of the relative frequency of these events among the different screening recommendations should be an important element in patient counseling.
Based on CISNET-modeled estimates, TABLE 2, illustrates the differences in both benefits and harms of the 3 screening strategies. With all strategies, there is a clear benefit in both fewer breast cancer–related deaths and life-years gained per 1,000 women screened.
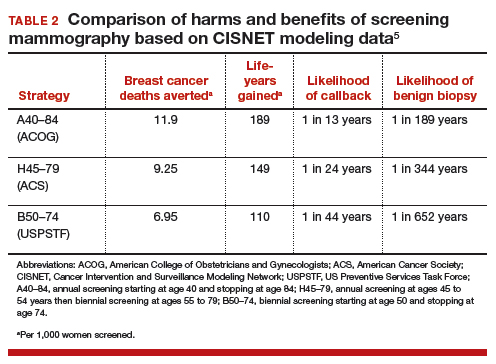
The greatest benefit is seen in the A40–84 group, that is, women who undergo the most intensive screening strategy with annual screening starting at age 40 and ending at age 84 (ACOG) compared with the USPSTF’s least intensive screening strategy, B50–74, which includes biennial screening starting at age 50 and stopping at age 74; benefits of the ACS’s H45–79 strategy (annual screening at ages 45 to 54 years then biennial screening at ages 55 to 79) were in-between. Not surprisingly, the A40–84 screening strategy was also associated with the most harms, with more recalls and benign breast biopsies; the least harms occurred with the USPSTF strategy, with the ACS strategy again in-between in terms of harms.
Related articles:
Breast density and optimal screening for breast cancer
To further demonstrate differences between the 3 strategies, CISNET also modeled results by looking at all women born in a single birth year cohort (1960) who were still alive at age 40 (2.468 million women). The modeling estimates the number of women who would die from breast cancer without screening mammography and compares that with the number of women who would die from breast cancer using any of the 3 screening strategies. Using this 1960 birth year cohort analysis, there would be approximately 12,000 fewer breast cancer deaths using the ACOG-recommended screening strategy compared with the USPSTF-recommended approach.4
These data show that while there are more harms associated with the most intense screening recommendation, the less frequent screening recommendations will result in higher mortality and more life-years lost. It is reasonable to assume that most patients would value mortality reduction and life-years gained over a likelihood of more benign biopsies or callbacks. As a result, each of the guidelines recommends that by age 40, women at average risk for breast cancer should be counseled and offered mammography screening based on their personal values.
Read about how Dr. Pearlman counsels his patients on screening.
My counseling approach on screening
Notably, the Women’s Preventive Services Initiative recommends that average risk women initiate mammography screening no earlier than age 40 and no later than age 50.6 This creates more flexibility around starting time for screening. In the population of women that I personally counsel, we discuss that fewer women (1 in 68) will experience breast cancer in their 40s compared with in their 50s (1 in 43); therefore as a population, more women will benefit from screening mammography in their 50s. However, there is clear evidence of mortality benefit for a woman in either decade should she develop breast cancer.
We also discuss that the frequency of harms is fairly comparable in either decade, but women who choose to start screening at age 50 will obviously not experience any callbacks or screening-associated benign breast biopsies in their 40s. With this understanding of benefits and harms, most (but not all) average risk women in my practice choose to start screening at age 40.
Related articles:
Breast cancer screening: My practices and response to the USPSTF guidelines
Be mindful of study limitations
The study by Arleo and colleagues has several weaknesses.5
Simulation studies/computer models have limitations. They are only as accurate as the assumptions that are used in the model. However, CISNET modeling has the benefit of having 6 different models with different assumptions on mortality, efficacy of mammography, and efficacy of treatment, and Arleo and colleagues’ analysis takes the mean of these 6 different models.5 It is reassuring to know that the modeling results are consistent with virtually all studies that show that annual screening mammography has a mortality benefit for women in their 40s.
Cost differences are not included. The actual cost of differences between the strategies is difficult to calculate and was not analyzed in this study. While it is easy to calculate the “front end” costs in a study like this (for example, how many more mammograms or biopsies in the different strategies), it is very difficult to calculate the “back end” costs (such as avoided chemotherapy or end-of-life care).
Overtreatment and overdiagnosis have been discussed extensively with regard to the different screening strategies. For example, approximately 80% of women with ductal carcinoma in situ (DCIS) have these tumors detected on screening mammography, and DCIS is not an obligate precursor to invasive breast cancer. Because the natural history of DCIS cannot be predicted, treatment is recommended for all women with DCIS, even though many of these tumors will remain indolent and never cause harm. As a result, concerns have been raised that more intensive screening strategies may result in more overdiagnosis and overtreatment compared with less intensive strategies.
Increasingly, this argument has been questioned, since the prevailing thought is that DCIS does not regress or disappear on mammography. In other words, if DCIS is present at age 40, it will be detected whenever screening starts (age 40, 45, or 50), and age of starting screening or the screening interval will not impact overdiagnosis or overtreatment.7
Related articles:
More than one-third of tumors found on breast cancer screening represent overdiagnosis
Counsel patients, offer screening at age 40
While 3 different breast cancer mammography screening strategies are recommended in the United States, the study by Arleo and colleagues suggests that based on CISNET data, the A40–84 strategy appears to be the most effective at reducing breast cancer mortality and resulting in the most life-years gained. This strategy also requires the most lifetime mammograms and results in the most callbacks and benign biopsies. Women should be offered annual screening mammography starting at age 40 and should start no later than age 50 after receiving counseling about benefits and harms.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Cancer Facts & Figures 2017. American Cancer Society website. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf. Accessed October 4, 2017.
- Oeffinger KC, Frontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599–1614.
- American College of Obstetricians and Gynecologists Committee on Practice Bulletins–Gynecology. ACOG Practice Bulletin No. 179: Breast cancer risk assessment and screening in average risk women. Obstet Gynecol. 2017;130(1):e1–e16.
- Siu AL; US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164(4):279–296.
- Arleo EK, Hendrick RE, Helvie MA, Sickles EA. Comparison of recommendations for screening mammography using CISNET models. Cancer. 2017;123(19):3673–3680.
- Women’s Preventive Services Initiative. Breast cancer screening for average-risk women. https://www.womenspreventivehealth.org/recommendations/breast-cancer-screening-for-average-risk-women/. Published 2016. Accessed October 4, 2017.
- Arleo EK, Monticciolo DL, Monsees B, McGinty G, Sickles EA. Persistent untreated screening-detected breast cancer: an argument against delaying screening or increasing the interval between screenings. J Am Coll Radiol. 2017;14(7):863–867.
Breast cancer is the most common cancer and the second leading cause of cancer death in women in the United States, with an estimated 252,710 new cases and 40,610 deaths in 2017.1 Breast cancer mortality is prevented by the use of regular screening mammography, as demonstrated by randomized controlled trials (20% reduction), incidence-based mortality studies (38% to 40% reduction), and service screening studies (48% to 49% reduction).2
Controversy continues, however, on when to start mammography screening, when to stop screening, and the frequency with which screening should be performed for women at average risk for breast cancer. Indeed, 3 national recommendations—written by the American College of Obstetricians and Gynecologists (ACOG), the American Cancer Society (ACS), and the US Preventive Services Task Force (USPSTF)—offer different guidelines for mammography screening (TABLE 1).2–4

There are 2 principal reasons for the controversy over screening:
- mammography has both benefits and harms, and individuals place differential weight on the importance of these relative to each other
- randomized controlled trials on screening mammography did not include all of the starting age, stopping age, and screening intervals that are included in screening recommendations.
New comparison of recommendations
An ongoing project funded by the National Cancer Institute, known as the Cancer Intervention and Surveillance Modeling Network (CISNET), models different starting and stopping ages and screening intervals for mammography to assess their impact on both benefits (mortality improvement, life-years gained) and harms (callbacks, benign breast biopsies). Recently, Arleo and colleagues used CISNET model data to compare the breast cancer screening recommendations from ACOG, the ACS, and the USPSTF, focusing on the differential effect on benefits and harms.5
Benefits vs harms of screening in perspective
Without question, the principal goal of cancer screening strategies is to effectively and efficiently reduce cancer mortality. Because mammography screening has both benefits and harms, a clear understanding of the relative frequency of these events among the different screening recommendations should be an important element in patient counseling.
Based on CISNET-modeled estimates, TABLE 2, illustrates the differences in both benefits and harms of the 3 screening strategies. With all strategies, there is a clear benefit in both fewer breast cancer–related deaths and life-years gained per 1,000 women screened.

The greatest benefit is seen in the A40–84 group, that is, women who undergo the most intensive screening strategy with annual screening starting at age 40 and ending at age 84 (ACOG) compared with the USPSTF’s least intensive screening strategy, B50–74, which includes biennial screening starting at age 50 and stopping at age 74; benefits of the ACS’s H45–79 strategy (annual screening at ages 45 to 54 years then biennial screening at ages 55 to 79) were in-between. Not surprisingly, the A40–84 screening strategy was also associated with the most harms, with more recalls and benign breast biopsies; the least harms occurred with the USPSTF strategy, with the ACS strategy again in-between in terms of harms.
Related articles:
Breast density and optimal screening for breast cancer
To further demonstrate differences between the 3 strategies, CISNET also modeled results by looking at all women born in a single birth year cohort (1960) who were still alive at age 40 (2.468 million women). The modeling estimates the number of women who would die from breast cancer without screening mammography and compares that with the number of women who would die from breast cancer using any of the 3 screening strategies. Using this 1960 birth year cohort analysis, there would be approximately 12,000 fewer breast cancer deaths using the ACOG-recommended screening strategy compared with the USPSTF-recommended approach.4
These data show that while there are more harms associated with the most intense screening recommendation, the less frequent screening recommendations will result in higher mortality and more life-years lost. It is reasonable to assume that most patients would value mortality reduction and life-years gained over a likelihood of more benign biopsies or callbacks. As a result, each of the guidelines recommends that by age 40, women at average risk for breast cancer should be counseled and offered mammography screening based on their personal values.
Read about how Dr. Pearlman counsels his patients on screening.
My counseling approach on screening
Notably, the Women’s Preventive Services Initiative recommends that average risk women initiate mammography screening no earlier than age 40 and no later than age 50.6 This creates more flexibility around starting time for screening. In the population of women that I personally counsel, we discuss that fewer women (1 in 68) will experience breast cancer in their 40s compared with in their 50s (1 in 43); therefore as a population, more women will benefit from screening mammography in their 50s. However, there is clear evidence of mortality benefit for a woman in either decade should she develop breast cancer.
We also discuss that the frequency of harms is fairly comparable in either decade, but women who choose to start screening at age 50 will obviously not experience any callbacks or screening-associated benign breast biopsies in their 40s. With this understanding of benefits and harms, most (but not all) average risk women in my practice choose to start screening at age 40.
Related articles:
Breast cancer screening: My practices and response to the USPSTF guidelines
Be mindful of study limitations
The study by Arleo and colleagues has several weaknesses.5
Simulation studies/computer models have limitations. They are only as accurate as the assumptions that are used in the model. However, CISNET modeling has the benefit of having 6 different models with different assumptions on mortality, efficacy of mammography, and efficacy of treatment, and Arleo and colleagues’ analysis takes the mean of these 6 different models.5 It is reassuring to know that the modeling results are consistent with virtually all studies that show that annual screening mammography has a mortality benefit for women in their 40s.
Cost differences are not included. The actual cost of differences between the strategies is difficult to calculate and was not analyzed in this study. While it is easy to calculate the “front end” costs in a study like this (for example, how many more mammograms or biopsies in the different strategies), it is very difficult to calculate the “back end” costs (such as avoided chemotherapy or end-of-life care).
Overtreatment and overdiagnosis have been discussed extensively with regard to the different screening strategies. For example, approximately 80% of women with ductal carcinoma in situ (DCIS) have these tumors detected on screening mammography, and DCIS is not an obligate precursor to invasive breast cancer. Because the natural history of DCIS cannot be predicted, treatment is recommended for all women with DCIS, even though many of these tumors will remain indolent and never cause harm. As a result, concerns have been raised that more intensive screening strategies may result in more overdiagnosis and overtreatment compared with less intensive strategies.
Increasingly, this argument has been questioned, since the prevailing thought is that DCIS does not regress or disappear on mammography. In other words, if DCIS is present at age 40, it will be detected whenever screening starts (age 40, 45, or 50), and age of starting screening or the screening interval will not impact overdiagnosis or overtreatment.7
Related articles:
More than one-third of tumors found on breast cancer screening represent overdiagnosis
Counsel patients, offer screening at age 40
While 3 different breast cancer mammography screening strategies are recommended in the United States, the study by Arleo and colleagues suggests that based on CISNET data, the A40–84 strategy appears to be the most effective at reducing breast cancer mortality and resulting in the most life-years gained. This strategy also requires the most lifetime mammograms and results in the most callbacks and benign biopsies. Women should be offered annual screening mammography starting at age 40 and should start no later than age 50 after receiving counseling about benefits and harms.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Breast cancer is the most common cancer and the second leading cause of cancer death in women in the United States, with an estimated 252,710 new cases and 40,610 deaths in 2017.1 Breast cancer mortality is prevented by the use of regular screening mammography, as demonstrated by randomized controlled trials (20% reduction), incidence-based mortality studies (38% to 40% reduction), and service screening studies (48% to 49% reduction).2
Controversy continues, however, on when to start mammography screening, when to stop screening, and the frequency with which screening should be performed for women at average risk for breast cancer. Indeed, 3 national recommendations—written by the American College of Obstetricians and Gynecologists (ACOG), the American Cancer Society (ACS), and the US Preventive Services Task Force (USPSTF)—offer different guidelines for mammography screening (TABLE 1).2–4

There are 2 principal reasons for the controversy over screening:
- mammography has both benefits and harms, and individuals place differential weight on the importance of these relative to each other
- randomized controlled trials on screening mammography did not include all of the starting age, stopping age, and screening intervals that are included in screening recommendations.
New comparison of recommendations
An ongoing project funded by the National Cancer Institute, known as the Cancer Intervention and Surveillance Modeling Network (CISNET), models different starting and stopping ages and screening intervals for mammography to assess their impact on both benefits (mortality improvement, life-years gained) and harms (callbacks, benign breast biopsies). Recently, Arleo and colleagues used CISNET model data to compare the breast cancer screening recommendations from ACOG, the ACS, and the USPSTF, focusing on the differential effect on benefits and harms.5
Benefits vs harms of screening in perspective
Without question, the principal goal of cancer screening strategies is to effectively and efficiently reduce cancer mortality. Because mammography screening has both benefits and harms, a clear understanding of the relative frequency of these events among the different screening recommendations should be an important element in patient counseling.
Based on CISNET-modeled estimates, TABLE 2, illustrates the differences in both benefits and harms of the 3 screening strategies. With all strategies, there is a clear benefit in both fewer breast cancer–related deaths and life-years gained per 1,000 women screened.

The greatest benefit is seen in the A40–84 group, that is, women who undergo the most intensive screening strategy with annual screening starting at age 40 and ending at age 84 (ACOG) compared with the USPSTF’s least intensive screening strategy, B50–74, which includes biennial screening starting at age 50 and stopping at age 74; benefits of the ACS’s H45–79 strategy (annual screening at ages 45 to 54 years then biennial screening at ages 55 to 79) were in-between. Not surprisingly, the A40–84 screening strategy was also associated with the most harms, with more recalls and benign breast biopsies; the least harms occurred with the USPSTF strategy, with the ACS strategy again in-between in terms of harms.
Related articles:
Breast density and optimal screening for breast cancer
To further demonstrate differences between the 3 strategies, CISNET also modeled results by looking at all women born in a single birth year cohort (1960) who were still alive at age 40 (2.468 million women). The modeling estimates the number of women who would die from breast cancer without screening mammography and compares that with the number of women who would die from breast cancer using any of the 3 screening strategies. Using this 1960 birth year cohort analysis, there would be approximately 12,000 fewer breast cancer deaths using the ACOG-recommended screening strategy compared with the USPSTF-recommended approach.4
These data show that while there are more harms associated with the most intense screening recommendation, the less frequent screening recommendations will result in higher mortality and more life-years lost. It is reasonable to assume that most patients would value mortality reduction and life-years gained over a likelihood of more benign biopsies or callbacks. As a result, each of the guidelines recommends that by age 40, women at average risk for breast cancer should be counseled and offered mammography screening based on their personal values.
Read about how Dr. Pearlman counsels his patients on screening.
My counseling approach on screening
Notably, the Women’s Preventive Services Initiative recommends that average risk women initiate mammography screening no earlier than age 40 and no later than age 50.6 This creates more flexibility around starting time for screening. In the population of women that I personally counsel, we discuss that fewer women (1 in 68) will experience breast cancer in their 40s compared with in their 50s (1 in 43); therefore as a population, more women will benefit from screening mammography in their 50s. However, there is clear evidence of mortality benefit for a woman in either decade should she develop breast cancer.
We also discuss that the frequency of harms is fairly comparable in either decade, but women who choose to start screening at age 50 will obviously not experience any callbacks or screening-associated benign breast biopsies in their 40s. With this understanding of benefits and harms, most (but not all) average risk women in my practice choose to start screening at age 40.
Related articles:
Breast cancer screening: My practices and response to the USPSTF guidelines
Be mindful of study limitations
The study by Arleo and colleagues has several weaknesses.5
Simulation studies/computer models have limitations. They are only as accurate as the assumptions that are used in the model. However, CISNET modeling has the benefit of having 6 different models with different assumptions on mortality, efficacy of mammography, and efficacy of treatment, and Arleo and colleagues’ analysis takes the mean of these 6 different models.5 It is reassuring to know that the modeling results are consistent with virtually all studies that show that annual screening mammography has a mortality benefit for women in their 40s.
Cost differences are not included. The actual cost of differences between the strategies is difficult to calculate and was not analyzed in this study. While it is easy to calculate the “front end” costs in a study like this (for example, how many more mammograms or biopsies in the different strategies), it is very difficult to calculate the “back end” costs (such as avoided chemotherapy or end-of-life care).
Overtreatment and overdiagnosis have been discussed extensively with regard to the different screening strategies. For example, approximately 80% of women with ductal carcinoma in situ (DCIS) have these tumors detected on screening mammography, and DCIS is not an obligate precursor to invasive breast cancer. Because the natural history of DCIS cannot be predicted, treatment is recommended for all women with DCIS, even though many of these tumors will remain indolent and never cause harm. As a result, concerns have been raised that more intensive screening strategies may result in more overdiagnosis and overtreatment compared with less intensive strategies.
Increasingly, this argument has been questioned, since the prevailing thought is that DCIS does not regress or disappear on mammography. In other words, if DCIS is present at age 40, it will be detected whenever screening starts (age 40, 45, or 50), and age of starting screening or the screening interval will not impact overdiagnosis or overtreatment.7
Related articles:
More than one-third of tumors found on breast cancer screening represent overdiagnosis
Counsel patients, offer screening at age 40
While 3 different breast cancer mammography screening strategies are recommended in the United States, the study by Arleo and colleagues suggests that based on CISNET data, the A40–84 strategy appears to be the most effective at reducing breast cancer mortality and resulting in the most life-years gained. This strategy also requires the most lifetime mammograms and results in the most callbacks and benign biopsies. Women should be offered annual screening mammography starting at age 40 and should start no later than age 50 after receiving counseling about benefits and harms.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Cancer Facts & Figures 2017. American Cancer Society website. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf. Accessed October 4, 2017.
- Oeffinger KC, Frontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599–1614.
- American College of Obstetricians and Gynecologists Committee on Practice Bulletins–Gynecology. ACOG Practice Bulletin No. 179: Breast cancer risk assessment and screening in average risk women. Obstet Gynecol. 2017;130(1):e1–e16.
- Siu AL; US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164(4):279–296.
- Arleo EK, Hendrick RE, Helvie MA, Sickles EA. Comparison of recommendations for screening mammography using CISNET models. Cancer. 2017;123(19):3673–3680.
- Women’s Preventive Services Initiative. Breast cancer screening for average-risk women. https://www.womenspreventivehealth.org/recommendations/breast-cancer-screening-for-average-risk-women/. Published 2016. Accessed October 4, 2017.
- Arleo EK, Monticciolo DL, Monsees B, McGinty G, Sickles EA. Persistent untreated screening-detected breast cancer: an argument against delaying screening or increasing the interval between screenings. J Am Coll Radiol. 2017;14(7):863–867.
- Cancer Facts & Figures 2017. American Cancer Society website. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf. Accessed October 4, 2017.
- Oeffinger KC, Frontham ET, Etzioni R, et al; American Cancer Society. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599–1614.
- American College of Obstetricians and Gynecologists Committee on Practice Bulletins–Gynecology. ACOG Practice Bulletin No. 179: Breast cancer risk assessment and screening in average risk women. Obstet Gynecol. 2017;130(1):e1–e16.
- Siu AL; US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164(4):279–296.
- Arleo EK, Hendrick RE, Helvie MA, Sickles EA. Comparison of recommendations for screening mammography using CISNET models. Cancer. 2017;123(19):3673–3680.
- Women’s Preventive Services Initiative. Breast cancer screening for average-risk women. https://www.womenspreventivehealth.org/recommendations/breast-cancer-screening-for-average-risk-women/. Published 2016. Accessed October 4, 2017.
- Arleo EK, Monticciolo DL, Monsees B, McGinty G, Sickles EA. Persistent untreated screening-detected breast cancer: an argument against delaying screening or increasing the interval between screenings. J Am Coll Radiol. 2017;14(7):863–867.
FDA Approves Patient-Assisted Mammography
Women of all ages and sizes will be glad to know that they now have some say in the amount of pressure applied to the breast during a mammography. The FDA has cleared Senographe Pristina with Self-Compression, the first patient-assisted 2D digital mammography system.
Digital mammograms use a computer along with x-rays. During an exam with the new system, the technologist positions the patient and initiates compression, then guides the patient in using the handheld wireless remote control to adjust the compression to a comfortable level. The technologist makes the final decision on whether the compression is adequate.
A clinical validation demonstrated that the addition of a remote to allow self-compression did not negatively affect image quality. Nor did allowing the patient to help with adjustments make the exam take significantly longer.
Women of all ages and sizes will be glad to know that they now have some say in the amount of pressure applied to the breast during a mammography. The FDA has cleared Senographe Pristina with Self-Compression, the first patient-assisted 2D digital mammography system.
Digital mammograms use a computer along with x-rays. During an exam with the new system, the technologist positions the patient and initiates compression, then guides the patient in using the handheld wireless remote control to adjust the compression to a comfortable level. The technologist makes the final decision on whether the compression is adequate.
A clinical validation demonstrated that the addition of a remote to allow self-compression did not negatively affect image quality. Nor did allowing the patient to help with adjustments make the exam take significantly longer.
Women of all ages and sizes will be glad to know that they now have some say in the amount of pressure applied to the breast during a mammography. The FDA has cleared Senographe Pristina with Self-Compression, the first patient-assisted 2D digital mammography system.
Digital mammograms use a computer along with x-rays. During an exam with the new system, the technologist positions the patient and initiates compression, then guides the patient in using the handheld wireless remote control to adjust the compression to a comfortable level. The technologist makes the final decision on whether the compression is adequate.
A clinical validation demonstrated that the addition of a remote to allow self-compression did not negatively affect image quality. Nor did allowing the patient to help with adjustments make the exam take significantly longer.
Reconstruction becoming more common after mastectomy
The rate of breast reconstruction surgery for mastectomy increased 62% from 2009 to 2014, while the mastectomy rate itself “remained relatively stable,” according to the Agency for Healthcare Research and Quality.
The rate of breast reconstructions in hospitals and ambulatory surgery settings rose steadily over the 6-year period, going from 21.7 per 100,000 women in 2009 to 35.1 per 100,000 in 2014. Meanwhile, the mastectomy rate dipped from 90.1 in 2009 to 83.2 in 2010 but varied less than 10% over the 2009-2014 time period, reaching 88.4 per 100,000 women in 2014. To put those numbers in a different context, the ratio of reconstructions to mastectomies went from 24-to-100 in 2009 to 40-to-100 in 2014, the AHRQ reported in a Statistical Brief.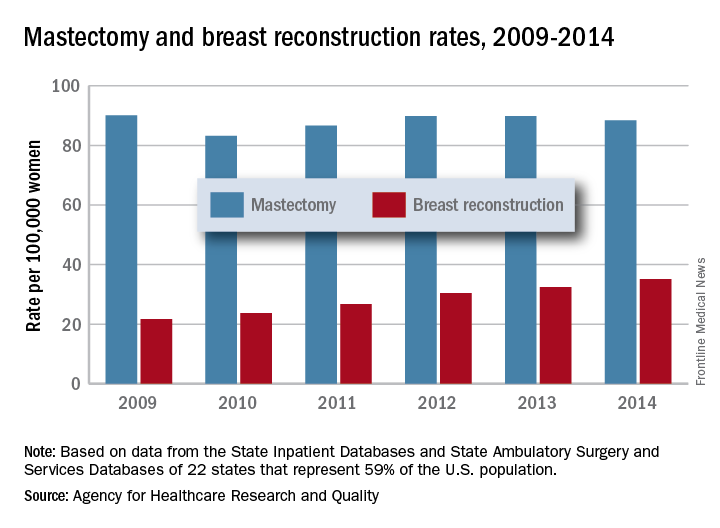
Those nonsimultaneous procedures were taking place much more often in ambulatory settings by 2014, as the rate of reconstructions at a separate visit increased 152% from 7.4 per 100,000 women in 2009 to 18.2. Simultaneous reconstructions in ambulatory settings were less common but increased at an even greater rate of 155%, going from 1.1 to 2.8 per 100,000 women. Inpatient reconstruction had little or no growth over the 6 years: Separate-visit procedures went from 6 to 6.8 and simultaneous reconstructions actually dropped from 7.4 per 100,000 women to 7.3, they reported.
The analysis was based on data from AHRQ State Inpatient Databases and State Ambulatory Surgery and Services Databases for 22 states that include 59% of the U.S. population: California, Colorado, Connecticut, Florida, Georgia, Iowa, Indiana, Maryland, Michigan, Minnesota, Missouri, Nebraska, New Jersey, New York, North Carolina, Ohio, South Carolina, South Dakota, Tennessee, Utah, Vermont, and Wisconsin.
The rate of breast reconstruction surgery for mastectomy increased 62% from 2009 to 2014, while the mastectomy rate itself “remained relatively stable,” according to the Agency for Healthcare Research and Quality.
The rate of breast reconstructions in hospitals and ambulatory surgery settings rose steadily over the 6-year period, going from 21.7 per 100,000 women in 2009 to 35.1 per 100,000 in 2014. Meanwhile, the mastectomy rate dipped from 90.1 in 2009 to 83.2 in 2010 but varied less than 10% over the 2009-2014 time period, reaching 88.4 per 100,000 women in 2014. To put those numbers in a different context, the ratio of reconstructions to mastectomies went from 24-to-100 in 2009 to 40-to-100 in 2014, the AHRQ reported in a Statistical Brief.
Those nonsimultaneous procedures were taking place much more often in ambulatory settings by 2014, as the rate of reconstructions at a separate visit increased 152% from 7.4 per 100,000 women in 2009 to 18.2. Simultaneous reconstructions in ambulatory settings were less common but increased at an even greater rate of 155%, going from 1.1 to 2.8 per 100,000 women. Inpatient reconstruction had little or no growth over the 6 years: Separate-visit procedures went from 6 to 6.8 and simultaneous reconstructions actually dropped from 7.4 per 100,000 women to 7.3, they reported.
The analysis was based on data from AHRQ State Inpatient Databases and State Ambulatory Surgery and Services Databases for 22 states that include 59% of the U.S. population: California, Colorado, Connecticut, Florida, Georgia, Iowa, Indiana, Maryland, Michigan, Minnesota, Missouri, Nebraska, New Jersey, New York, North Carolina, Ohio, South Carolina, South Dakota, Tennessee, Utah, Vermont, and Wisconsin.
The rate of breast reconstruction surgery for mastectomy increased 62% from 2009 to 2014, while the mastectomy rate itself “remained relatively stable,” according to the Agency for Healthcare Research and Quality.
The rate of breast reconstructions in hospitals and ambulatory surgery settings rose steadily over the 6-year period, going from 21.7 per 100,000 women in 2009 to 35.1 per 100,000 in 2014. Meanwhile, the mastectomy rate dipped from 90.1 in 2009 to 83.2 in 2010 but varied less than 10% over the 2009-2014 time period, reaching 88.4 per 100,000 women in 2014. To put those numbers in a different context, the ratio of reconstructions to mastectomies went from 24-to-100 in 2009 to 40-to-100 in 2014, the AHRQ reported in a Statistical Brief.
Those nonsimultaneous procedures were taking place much more often in ambulatory settings by 2014, as the rate of reconstructions at a separate visit increased 152% from 7.4 per 100,000 women in 2009 to 18.2. Simultaneous reconstructions in ambulatory settings were less common but increased at an even greater rate of 155%, going from 1.1 to 2.8 per 100,000 women. Inpatient reconstruction had little or no growth over the 6 years: Separate-visit procedures went from 6 to 6.8 and simultaneous reconstructions actually dropped from 7.4 per 100,000 women to 7.3, they reported.
The analysis was based on data from AHRQ State Inpatient Databases and State Ambulatory Surgery and Services Databases for 22 states that include 59% of the U.S. population: California, Colorado, Connecticut, Florida, Georgia, Iowa, Indiana, Maryland, Michigan, Minnesota, Missouri, Nebraska, New Jersey, New York, North Carolina, Ohio, South Carolina, South Dakota, Tennessee, Utah, Vermont, and Wisconsin.
ExteNET: Benefit of extended neratinib in HER2+ breast cancer sustained
MADRID – who received postoperative trastuzumab (Herceptin) and chemotherapy, long-term follow-up results from the ExteNET trial show.
In a planned intention-to-treat analysis at 5 years of follow-up, extended adjuvant therapy with the tyrosine kinase inhibitor neratinib was associated with a small but significant improvement in invasive disease-free survival (iDFS), compared with placebo, with most of the benefit occurring in women with hormone receptor–positive disease, reported Miguel Martin, MD, of the Gregorio Marañón Health Research Institute in Madrid.

Data from an earlier analysis of the trial supported the Food and Drug Administration’s decision to approve neratinib in the extended adjuvant setting in July 2017.
In the ExteNET trial, 2,840 women with early HER2-positive breast cancer who had undergone surgery and adjuvant treatment with trastuzumab and chemotherapy were stratified by nodal and hormone receptor status and by concurrent vs. sequential chemotherapy and trastuzumab, and were then randomly assigned to receive oral neratinib 240 mg/day for 1 year, or placebo. Analyses of iDFS were planned for 2 and 5 years, and an overall survival analysis was planned after 248 patient deaths had occurred. Overall survival data have not matured as yet, Dr. Martin noted.
Results of an unspecified 3-year analysis of the trial, presented at the San Antonio Breast Cancer Symposium in 2015, showed a continued benefit for the addition of neratinib, a finding that has now been extended out to 5 years.
At ESMO 2017, Dr. Martin presented data on all efficacy endpoints except overall survival in the intention-to-treat population.
By the cutoff date in March 2017, 2,117 of the original 2,840 patients (76%) gave consent for collection of additional data, including 1,028 who had been assigned to neratinib, and 1,089 assigned to placebo.
The 5-year iDFS rate was 90.2% for patients assigned to neratinib, compared with 87.7% for those assigned to placebo, an absolute difference of 2.5%. This translated into a hazard ratio favoring neratinib of 0.73 (P = .008). Neratinib was also significantly better than placebo for DFS in patients with ductal carcinoma in situ (89.7% vs. 86.8, HR, 0.71, P = .004).
However, there were no significant differences at 5 years between trial arms in either distant DFS, time to distant recurrence, or central nervous system recurrences. Dr. Martin noted that the although there were fewer CNS recurrences with neratinib (1.30% vs. 1.82%), the total number of cases was too small to detect a possible difference.
In a subgroup analysis, neratinib trended toward better performance in all categories, but was significantly better than placebo only among patients from Asia, Eastern Europe, and South America, and among patients with four or more positive lymph nodes.
An analysis of iDFS by hormone receptor status showed that for HR-positive patients, the 5-year iDFS rate was 91.2% with neratinib vs. 86.8% with placebo, translating into a hazard ratio of 0.60, P = .002). In contrast, iDFS rates were nearly identical among HR-negative patients, at 88.9% vs. 88.8%, respectively.
Following treatment discontinuation, there was no evidence of increased symptomatic cardiotoxicity or second primary malignancies vs. placebo, and no late-term consequences of neratinib-associated diarrhea, Dr. Martin said.
A separate poster on health-related quality of life, also presented at ESMO 2017, showed that patients assigned to neratinib had a drop in quality-of-life measures during the first month of treatment, possibly because of diarrhea, but then had a steady improvement toward baseline. There is an ongoing study to evaluate whether loperamide-based regimens can reduce or prevent neratinib-associated diarrhea, the investigators noted.
“In ExteNET, we’ve seen continued demonstration of clinically significant benefit, particularly in higher-risk, hormone receptor–positive disease, despite many limitations, with change in sponsor and initial plan for only 2 years of follow-up,” said Hope S. Rugo, MD, from the University of California, San Francisco, the invited discussant.
“Survival data is pending, and we’re looking forward to seeing that in 2019, but the reduction in distant events, although small, is still encouraging,” she said.
The trial is sponsored by Puma Biotechnology. Dr. Martin disclosed honoraria from Roche/Genentech, Novartis, Amgen, AstraZeneca, Pfizer, PharmaMar, and Lilly, and research grants from Roche and Novartis. Dr Rugo disclosed travel support from PUMA and Mylan, research support from Genentech/Roche, and honoraria from Biotheranostics. She also serves on the Oncology Practice Advisory Board.
MADRID – who received postoperative trastuzumab (Herceptin) and chemotherapy, long-term follow-up results from the ExteNET trial show.
In a planned intention-to-treat analysis at 5 years of follow-up, extended adjuvant therapy with the tyrosine kinase inhibitor neratinib was associated with a small but significant improvement in invasive disease-free survival (iDFS), compared with placebo, with most of the benefit occurring in women with hormone receptor–positive disease, reported Miguel Martin, MD, of the Gregorio Marañón Health Research Institute in Madrid.

Data from an earlier analysis of the trial supported the Food and Drug Administration’s decision to approve neratinib in the extended adjuvant setting in July 2017.
In the ExteNET trial, 2,840 women with early HER2-positive breast cancer who had undergone surgery and adjuvant treatment with trastuzumab and chemotherapy were stratified by nodal and hormone receptor status and by concurrent vs. sequential chemotherapy and trastuzumab, and were then randomly assigned to receive oral neratinib 240 mg/day for 1 year, or placebo. Analyses of iDFS were planned for 2 and 5 years, and an overall survival analysis was planned after 248 patient deaths had occurred. Overall survival data have not matured as yet, Dr. Martin noted.
Results of an unspecified 3-year analysis of the trial, presented at the San Antonio Breast Cancer Symposium in 2015, showed a continued benefit for the addition of neratinib, a finding that has now been extended out to 5 years.
At ESMO 2017, Dr. Martin presented data on all efficacy endpoints except overall survival in the intention-to-treat population.
By the cutoff date in March 2017, 2,117 of the original 2,840 patients (76%) gave consent for collection of additional data, including 1,028 who had been assigned to neratinib, and 1,089 assigned to placebo.
The 5-year iDFS rate was 90.2% for patients assigned to neratinib, compared with 87.7% for those assigned to placebo, an absolute difference of 2.5%. This translated into a hazard ratio favoring neratinib of 0.73 (P = .008). Neratinib was also significantly better than placebo for DFS in patients with ductal carcinoma in situ (89.7% vs. 86.8, HR, 0.71, P = .004).
However, there were no significant differences at 5 years between trial arms in either distant DFS, time to distant recurrence, or central nervous system recurrences. Dr. Martin noted that the although there were fewer CNS recurrences with neratinib (1.30% vs. 1.82%), the total number of cases was too small to detect a possible difference.
In a subgroup analysis, neratinib trended toward better performance in all categories, but was significantly better than placebo only among patients from Asia, Eastern Europe, and South America, and among patients with four or more positive lymph nodes.
An analysis of iDFS by hormone receptor status showed that for HR-positive patients, the 5-year iDFS rate was 91.2% with neratinib vs. 86.8% with placebo, translating into a hazard ratio of 0.60, P = .002). In contrast, iDFS rates were nearly identical among HR-negative patients, at 88.9% vs. 88.8%, respectively.
Following treatment discontinuation, there was no evidence of increased symptomatic cardiotoxicity or second primary malignancies vs. placebo, and no late-term consequences of neratinib-associated diarrhea, Dr. Martin said.
A separate poster on health-related quality of life, also presented at ESMO 2017, showed that patients assigned to neratinib had a drop in quality-of-life measures during the first month of treatment, possibly because of diarrhea, but then had a steady improvement toward baseline. There is an ongoing study to evaluate whether loperamide-based regimens can reduce or prevent neratinib-associated diarrhea, the investigators noted.
“In ExteNET, we’ve seen continued demonstration of clinically significant benefit, particularly in higher-risk, hormone receptor–positive disease, despite many limitations, with change in sponsor and initial plan for only 2 years of follow-up,” said Hope S. Rugo, MD, from the University of California, San Francisco, the invited discussant.
“Survival data is pending, and we’re looking forward to seeing that in 2019, but the reduction in distant events, although small, is still encouraging,” she said.
The trial is sponsored by Puma Biotechnology. Dr. Martin disclosed honoraria from Roche/Genentech, Novartis, Amgen, AstraZeneca, Pfizer, PharmaMar, and Lilly, and research grants from Roche and Novartis. Dr Rugo disclosed travel support from PUMA and Mylan, research support from Genentech/Roche, and honoraria from Biotheranostics. She also serves on the Oncology Practice Advisory Board.
MADRID – who received postoperative trastuzumab (Herceptin) and chemotherapy, long-term follow-up results from the ExteNET trial show.
In a planned intention-to-treat analysis at 5 years of follow-up, extended adjuvant therapy with the tyrosine kinase inhibitor neratinib was associated with a small but significant improvement in invasive disease-free survival (iDFS), compared with placebo, with most of the benefit occurring in women with hormone receptor–positive disease, reported Miguel Martin, MD, of the Gregorio Marañón Health Research Institute in Madrid.

Data from an earlier analysis of the trial supported the Food and Drug Administration’s decision to approve neratinib in the extended adjuvant setting in July 2017.
In the ExteNET trial, 2,840 women with early HER2-positive breast cancer who had undergone surgery and adjuvant treatment with trastuzumab and chemotherapy were stratified by nodal and hormone receptor status and by concurrent vs. sequential chemotherapy and trastuzumab, and were then randomly assigned to receive oral neratinib 240 mg/day for 1 year, or placebo. Analyses of iDFS were planned for 2 and 5 years, and an overall survival analysis was planned after 248 patient deaths had occurred. Overall survival data have not matured as yet, Dr. Martin noted.
Results of an unspecified 3-year analysis of the trial, presented at the San Antonio Breast Cancer Symposium in 2015, showed a continued benefit for the addition of neratinib, a finding that has now been extended out to 5 years.
At ESMO 2017, Dr. Martin presented data on all efficacy endpoints except overall survival in the intention-to-treat population.
By the cutoff date in March 2017, 2,117 of the original 2,840 patients (76%) gave consent for collection of additional data, including 1,028 who had been assigned to neratinib, and 1,089 assigned to placebo.
The 5-year iDFS rate was 90.2% for patients assigned to neratinib, compared with 87.7% for those assigned to placebo, an absolute difference of 2.5%. This translated into a hazard ratio favoring neratinib of 0.73 (P = .008). Neratinib was also significantly better than placebo for DFS in patients with ductal carcinoma in situ (89.7% vs. 86.8, HR, 0.71, P = .004).
However, there were no significant differences at 5 years between trial arms in either distant DFS, time to distant recurrence, or central nervous system recurrences. Dr. Martin noted that the although there were fewer CNS recurrences with neratinib (1.30% vs. 1.82%), the total number of cases was too small to detect a possible difference.
In a subgroup analysis, neratinib trended toward better performance in all categories, but was significantly better than placebo only among patients from Asia, Eastern Europe, and South America, and among patients with four or more positive lymph nodes.
An analysis of iDFS by hormone receptor status showed that for HR-positive patients, the 5-year iDFS rate was 91.2% with neratinib vs. 86.8% with placebo, translating into a hazard ratio of 0.60, P = .002). In contrast, iDFS rates were nearly identical among HR-negative patients, at 88.9% vs. 88.8%, respectively.
Following treatment discontinuation, there was no evidence of increased symptomatic cardiotoxicity or second primary malignancies vs. placebo, and no late-term consequences of neratinib-associated diarrhea, Dr. Martin said.
A separate poster on health-related quality of life, also presented at ESMO 2017, showed that patients assigned to neratinib had a drop in quality-of-life measures during the first month of treatment, possibly because of diarrhea, but then had a steady improvement toward baseline. There is an ongoing study to evaluate whether loperamide-based regimens can reduce or prevent neratinib-associated diarrhea, the investigators noted.
“In ExteNET, we’ve seen continued demonstration of clinically significant benefit, particularly in higher-risk, hormone receptor–positive disease, despite many limitations, with change in sponsor and initial plan for only 2 years of follow-up,” said Hope S. Rugo, MD, from the University of California, San Francisco, the invited discussant.
“Survival data is pending, and we’re looking forward to seeing that in 2019, but the reduction in distant events, although small, is still encouraging,” she said.
The trial is sponsored by Puma Biotechnology. Dr. Martin disclosed honoraria from Roche/Genentech, Novartis, Amgen, AstraZeneca, Pfizer, PharmaMar, and Lilly, and research grants from Roche and Novartis. Dr Rugo disclosed travel support from PUMA and Mylan, research support from Genentech/Roche, and honoraria from Biotheranostics. She also serves on the Oncology Practice Advisory Board.
AT ESMO 2017
Key clinical point: Neratinib after adjuvant trastuzumab and chemotherapy continued to show improved invasive disease-free survival at 5 years in women with early HER2+ breast cancer.
Major finding: The 5-year iDFS rate was 90.2% for patients assigned to neratinib, compared with 87.7% for those assigned to placebo.
Data source: 5-year follow-up of randomized phase 3 trial in 2,840 women with HER2+ breast cancer treated with surgery and adjuvant chemotherapy/trastuzumab.
Disclosures: The trial is sponsored by Puma Biotechnology. Dr. Martin disclosed honoraria from Roche/Genentech, Novartis, Amgen, AstraZeneca, Pfizer, PharmaMar, and Lilly, and research grants from Roche and Novartis. Dr Rugo disclosed travel support from PUMA and Mylan, research support from Genentech/Roche, and honoraria from Biotheranostics. She also serves on the Oncology Practice Advisory Board.
Hyperlipidemia diagnosis protects against breast cancer
BARCELONA – Women diagnosed with hyperlipidemia had a strikingly reduced risk of subsequently developing breast cancer in a big-data, case-control study, Paul R. Carter, MD, reported at the annual congress of the European Society of Cardiology.
Moreover, those baseline hyperlipidemic women who later got breast cancer had a 40% lower risk of all-cause mortality than did matched nonhyperlipidemic controls diagnosed with the malignancy during follow-up, according to Dr. Carter, a cardiology fellow at Cambridge (England) University.
The inference isn’t that hyperlipidemia somehow protects against the most common type of cancer in women. Indeed, preclinical evidence indicates high cholesterol drives several key steps in carcinogenesis. Rather, the strong implication is that the explanation for the observed preventive effect lies in the pleotropic effects of the statin therapy routinely prescribed in accordance with guidelines once women received the diagnosis of hyperlipidemia, he continued.
“The results of this study provide the strongest justification to date for a clinical trial evaluating the protective effect of statins in patients with breast cancer, and this is what we intend to do,” according to Dr. Carter.
He presented a retrospective longitudinal study of the Algorithm for Comorbidities, Associations, Length of Stay and Mortality database, comprising more than 1.2 million patients admitted for various reasons to selected hospitals in northern England during 2000-2014. This big-data study entailed recruitment of 16,043 women aged 40 years and older who were diagnosed with hyperlipidemia during their hospital stay along with an equal number of age-matched women with normal lipid levels. None of the participants had a breast cancer diagnosis at baseline.
The all-cause mortality rate in baseline hyperlipidemic women who later developed breast cancer was 27.4%, significantly lower than the 37.4% rate in normolipidemic women with breast cancer. This translated into an adjusted 40% relative risk reduction.
All-cause mortality occurred during follow-up in 13.7% of breast cancer–free women with baseline hyperlipidemia, compared with 23.6% of nonhyperlipidemic controls without breast cancer.
In an analysis adjusted for age, ethnicity, and the top-10 causes of death in the U.K., women with baseline hyperlipidemia were 40% less likely to die during follow-up than were women without high cholesterol.
Dr. Carter reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
BARCELONA – Women diagnosed with hyperlipidemia had a strikingly reduced risk of subsequently developing breast cancer in a big-data, case-control study, Paul R. Carter, MD, reported at the annual congress of the European Society of Cardiology.
Moreover, those baseline hyperlipidemic women who later got breast cancer had a 40% lower risk of all-cause mortality than did matched nonhyperlipidemic controls diagnosed with the malignancy during follow-up, according to Dr. Carter, a cardiology fellow at Cambridge (England) University.
The inference isn’t that hyperlipidemia somehow protects against the most common type of cancer in women. Indeed, preclinical evidence indicates high cholesterol drives several key steps in carcinogenesis. Rather, the strong implication is that the explanation for the observed preventive effect lies in the pleotropic effects of the statin therapy routinely prescribed in accordance with guidelines once women received the diagnosis of hyperlipidemia, he continued.
“The results of this study provide the strongest justification to date for a clinical trial evaluating the protective effect of statins in patients with breast cancer, and this is what we intend to do,” according to Dr. Carter.
He presented a retrospective longitudinal study of the Algorithm for Comorbidities, Associations, Length of Stay and Mortality database, comprising more than 1.2 million patients admitted for various reasons to selected hospitals in northern England during 2000-2014. This big-data study entailed recruitment of 16,043 women aged 40 years and older who were diagnosed with hyperlipidemia during their hospital stay along with an equal number of age-matched women with normal lipid levels. None of the participants had a breast cancer diagnosis at baseline.
The all-cause mortality rate in baseline hyperlipidemic women who later developed breast cancer was 27.4%, significantly lower than the 37.4% rate in normolipidemic women with breast cancer. This translated into an adjusted 40% relative risk reduction.
All-cause mortality occurred during follow-up in 13.7% of breast cancer–free women with baseline hyperlipidemia, compared with 23.6% of nonhyperlipidemic controls without breast cancer.
In an analysis adjusted for age, ethnicity, and the top-10 causes of death in the U.K., women with baseline hyperlipidemia were 40% less likely to die during follow-up than were women without high cholesterol.
Dr. Carter reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
BARCELONA – Women diagnosed with hyperlipidemia had a strikingly reduced risk of subsequently developing breast cancer in a big-data, case-control study, Paul R. Carter, MD, reported at the annual congress of the European Society of Cardiology.
Moreover, those baseline hyperlipidemic women who later got breast cancer had a 40% lower risk of all-cause mortality than did matched nonhyperlipidemic controls diagnosed with the malignancy during follow-up, according to Dr. Carter, a cardiology fellow at Cambridge (England) University.
The inference isn’t that hyperlipidemia somehow protects against the most common type of cancer in women. Indeed, preclinical evidence indicates high cholesterol drives several key steps in carcinogenesis. Rather, the strong implication is that the explanation for the observed preventive effect lies in the pleotropic effects of the statin therapy routinely prescribed in accordance with guidelines once women received the diagnosis of hyperlipidemia, he continued.
“The results of this study provide the strongest justification to date for a clinical trial evaluating the protective effect of statins in patients with breast cancer, and this is what we intend to do,” according to Dr. Carter.
He presented a retrospective longitudinal study of the Algorithm for Comorbidities, Associations, Length of Stay and Mortality database, comprising more than 1.2 million patients admitted for various reasons to selected hospitals in northern England during 2000-2014. This big-data study entailed recruitment of 16,043 women aged 40 years and older who were diagnosed with hyperlipidemia during their hospital stay along with an equal number of age-matched women with normal lipid levels. None of the participants had a breast cancer diagnosis at baseline.
The all-cause mortality rate in baseline hyperlipidemic women who later developed breast cancer was 27.4%, significantly lower than the 37.4% rate in normolipidemic women with breast cancer. This translated into an adjusted 40% relative risk reduction.
All-cause mortality occurred during follow-up in 13.7% of breast cancer–free women with baseline hyperlipidemia, compared with 23.6% of nonhyperlipidemic controls without breast cancer.
In an analysis adjusted for age, ethnicity, and the top-10 causes of death in the U.K., women with baseline hyperlipidemia were 40% less likely to die during follow-up than were women without high cholesterol.
Dr. Carter reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
AT THE ESC CONGRESS 2017
Key clinical point:
Major finding: The risk of subsequent development of breast cancer was one-third lower in women diagnosed with hyperlipidemia than in controls with normal lipid levels.
Data source: A retrospective longitudinal case-control study of 16,043 U.K. women aged 40 years or older when diagnosed with hyperlipidemia and an equal number of age-matched women with normal lipids.
Disclosures: The presenter reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
David Henry's JCSO podcast, September-October 2017
In this podcast, coinciding with breast cancer awareness month, Dr David Henry highlights an article by contributor Jane de Lartigue on recent advances in the use of targeted therapies in multiple breast cancer subtypes and another by a practicing oncologist on the positive impact of centralizing breast cancer care in an urban public hospital. Patient-reported outcomes are the focus a review on PROs in palliative and supportive interventions rural cancer patients and an original research report on findings on adverse events from systemic treatment of cancer and patient-reported quality of life. Also featured are two Community Translations columns on the approvals for atezolizumab for non–small-cell lung cancer and lenalidomide as standard of care for multiple myeloma in the maintenance setting, and two case reports on familial essential thrombocythemia associated with JAK2 V617F mutation in siblings and on managing tonsillar carcinoma with advanced radiation and chemotherapy techniques.
Listen to the podcast below.
In this podcast, coinciding with breast cancer awareness month, Dr David Henry highlights an article by contributor Jane de Lartigue on recent advances in the use of targeted therapies in multiple breast cancer subtypes and another by a practicing oncologist on the positive impact of centralizing breast cancer care in an urban public hospital. Patient-reported outcomes are the focus a review on PROs in palliative and supportive interventions rural cancer patients and an original research report on findings on adverse events from systemic treatment of cancer and patient-reported quality of life. Also featured are two Community Translations columns on the approvals for atezolizumab for non–small-cell lung cancer and lenalidomide as standard of care for multiple myeloma in the maintenance setting, and two case reports on familial essential thrombocythemia associated with JAK2 V617F mutation in siblings and on managing tonsillar carcinoma with advanced radiation and chemotherapy techniques.
Listen to the podcast below.
In this podcast, coinciding with breast cancer awareness month, Dr David Henry highlights an article by contributor Jane de Lartigue on recent advances in the use of targeted therapies in multiple breast cancer subtypes and another by a practicing oncologist on the positive impact of centralizing breast cancer care in an urban public hospital. Patient-reported outcomes are the focus a review on PROs in palliative and supportive interventions rural cancer patients and an original research report on findings on adverse events from systemic treatment of cancer and patient-reported quality of life. Also featured are two Community Translations columns on the approvals for atezolizumab for non–small-cell lung cancer and lenalidomide as standard of care for multiple myeloma in the maintenance setting, and two case reports on familial essential thrombocythemia associated with JAK2 V617F mutation in siblings and on managing tonsillar carcinoma with advanced radiation and chemotherapy techniques.
Listen to the podcast below.
FDA approves abemaciclib for advanced breast cancer
The Food and Drug Administration has approved abemaciclib (Verzenio) to be given in combination with fulvestrant, to treat patients who have hormone receptor (HR)–positive, human epidermal growth factor receptor 2 (HER2)–negative advanced or metastatic breast cancer that has progressed after taking endocrine therapy.
This is the third cyclin-dependent kinase 4/6 (CDK4/6) inhibitor approved for the treatment of advanced breast cancer. Palbociclib (Ibrance) was granted accelerated approval in February 2015, in combination with letrozole, for the treatment of HR-positive, HER2-negative advanced breast cancer as initial endocrine-based therapy in postmenopausal women. Ribociclib (Kisqali) was approved in March 2017, in combination with any aromatase inhibitor, also for the treatment of HR-positive, HER2-negative advanced breast cancer as initial endocrine-based therapy in postmenopausal women.
Approval of abemaciclib in combination with fulvestrant was based on a median progression-free survival of 16.4 months for patients taking abemaciclib with fulvestrant, compared with 9.3 months for patients taking a placebo with fulvestrant, in a randomized trial. All 669 patients in the trial had HR-positive, HER2-negative breast cancer that had progressed after treatment with endocrine therapy and had not received chemotherapy once the cancer had metastasized.
Approval of abemaciclib as a single agent was based on an overall response rate of 19.7% in a single-arm trial of 132 patients with HR-positive, HER2-negative breast cancer that had progressed after treatment with endocrine therapy and chemotherapy after the cancer metastasized.
Common side effects of abemaciclib include diarrhea, neutropenia, leukopenia, nausea, abdominal pain, infections, fatigue, anemia, decreased appetite, vomiting, and headache.
Serious side effects include diarrhea, neutropenia, elevated liver blood tests, and deep venous thrombosis/pulmonary embolism, the FDA said.
Approval was granted to Eli Lilly.
The Food and Drug Administration has approved abemaciclib (Verzenio) to be given in combination with fulvestrant, to treat patients who have hormone receptor (HR)–positive, human epidermal growth factor receptor 2 (HER2)–negative advanced or metastatic breast cancer that has progressed after taking endocrine therapy.
This is the third cyclin-dependent kinase 4/6 (CDK4/6) inhibitor approved for the treatment of advanced breast cancer. Palbociclib (Ibrance) was granted accelerated approval in February 2015, in combination with letrozole, for the treatment of HR-positive, HER2-negative advanced breast cancer as initial endocrine-based therapy in postmenopausal women. Ribociclib (Kisqali) was approved in March 2017, in combination with any aromatase inhibitor, also for the treatment of HR-positive, HER2-negative advanced breast cancer as initial endocrine-based therapy in postmenopausal women.
Approval of abemaciclib in combination with fulvestrant was based on a median progression-free survival of 16.4 months for patients taking abemaciclib with fulvestrant, compared with 9.3 months for patients taking a placebo with fulvestrant, in a randomized trial. All 669 patients in the trial had HR-positive, HER2-negative breast cancer that had progressed after treatment with endocrine therapy and had not received chemotherapy once the cancer had metastasized.
Approval of abemaciclib as a single agent was based on an overall response rate of 19.7% in a single-arm trial of 132 patients with HR-positive, HER2-negative breast cancer that had progressed after treatment with endocrine therapy and chemotherapy after the cancer metastasized.
Common side effects of abemaciclib include diarrhea, neutropenia, leukopenia, nausea, abdominal pain, infections, fatigue, anemia, decreased appetite, vomiting, and headache.
Serious side effects include diarrhea, neutropenia, elevated liver blood tests, and deep venous thrombosis/pulmonary embolism, the FDA said.
Approval was granted to Eli Lilly.
The Food and Drug Administration has approved abemaciclib (Verzenio) to be given in combination with fulvestrant, to treat patients who have hormone receptor (HR)–positive, human epidermal growth factor receptor 2 (HER2)–negative advanced or metastatic breast cancer that has progressed after taking endocrine therapy.
This is the third cyclin-dependent kinase 4/6 (CDK4/6) inhibitor approved for the treatment of advanced breast cancer. Palbociclib (Ibrance) was granted accelerated approval in February 2015, in combination with letrozole, for the treatment of HR-positive, HER2-negative advanced breast cancer as initial endocrine-based therapy in postmenopausal women. Ribociclib (Kisqali) was approved in March 2017, in combination with any aromatase inhibitor, also for the treatment of HR-positive, HER2-negative advanced breast cancer as initial endocrine-based therapy in postmenopausal women.
Approval of abemaciclib in combination with fulvestrant was based on a median progression-free survival of 16.4 months for patients taking abemaciclib with fulvestrant, compared with 9.3 months for patients taking a placebo with fulvestrant, in a randomized trial. All 669 patients in the trial had HR-positive, HER2-negative breast cancer that had progressed after treatment with endocrine therapy and had not received chemotherapy once the cancer had metastasized.
Approval of abemaciclib as a single agent was based on an overall response rate of 19.7% in a single-arm trial of 132 patients with HR-positive, HER2-negative breast cancer that had progressed after treatment with endocrine therapy and chemotherapy after the cancer metastasized.
Common side effects of abemaciclib include diarrhea, neutropenia, leukopenia, nausea, abdominal pain, infections, fatigue, anemia, decreased appetite, vomiting, and headache.
Serious side effects include diarrhea, neutropenia, elevated liver blood tests, and deep venous thrombosis/pulmonary embolism, the FDA said.
Approval was granted to Eli Lilly.