User login
VIDEO: Breast surgeons can and do provide genetic counseling to cancer patients
MIAMI BEACH – Breast surgeons can help relieve a “medical education crisis” by advising breast cancer patients about the implications of genetic testing results. The crisis continues to worsen given the remarkable rise in precision medicine and the limited number of genetic counselors in the United States, Patrick W. Whitworth, MD, said at the annual Miami Breast Cancer Conference, held by Physicians’ Education Resource.
Breast surgeons are qualified to help counsel patients, and, in fact, already order more than half of breast cancer genetic sequencing, a specialist survey reveals, said Dr. Whitworth, director of the Nashville (Tenn.) Breast Center, in a video interview.
At the same time, the one genotype–one phenotype paradigm for cancer genetics is rapidly shifting to testing patients for a whole panel of mutations simultaneously. Breast surgeons can partner with genetic counselors to support and expand the reach of this vital medical testing and counseling to more patients, he added.
Dr. Whitworth receives research grant support from and is a shareholder in Targeted Medical Education. He is also a shareholder in Advantage Consulting and Education.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
MIAMI BEACH – Breast surgeons can help relieve a “medical education crisis” by advising breast cancer patients about the implications of genetic testing results. The crisis continues to worsen given the remarkable rise in precision medicine and the limited number of genetic counselors in the United States, Patrick W. Whitworth, MD, said at the annual Miami Breast Cancer Conference, held by Physicians’ Education Resource.
Breast surgeons are qualified to help counsel patients, and, in fact, already order more than half of breast cancer genetic sequencing, a specialist survey reveals, said Dr. Whitworth, director of the Nashville (Tenn.) Breast Center, in a video interview.
At the same time, the one genotype–one phenotype paradigm for cancer genetics is rapidly shifting to testing patients for a whole panel of mutations simultaneously. Breast surgeons can partner with genetic counselors to support and expand the reach of this vital medical testing and counseling to more patients, he added.
Dr. Whitworth receives research grant support from and is a shareholder in Targeted Medical Education. He is also a shareholder in Advantage Consulting and Education.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
MIAMI BEACH – Breast surgeons can help relieve a “medical education crisis” by advising breast cancer patients about the implications of genetic testing results. The crisis continues to worsen given the remarkable rise in precision medicine and the limited number of genetic counselors in the United States, Patrick W. Whitworth, MD, said at the annual Miami Breast Cancer Conference, held by Physicians’ Education Resource.
Breast surgeons are qualified to help counsel patients, and, in fact, already order more than half of breast cancer genetic sequencing, a specialist survey reveals, said Dr. Whitworth, director of the Nashville (Tenn.) Breast Center, in a video interview.
At the same time, the one genotype–one phenotype paradigm for cancer genetics is rapidly shifting to testing patients for a whole panel of mutations simultaneously. Breast surgeons can partner with genetic counselors to support and expand the reach of this vital medical testing and counseling to more patients, he added.
Dr. Whitworth receives research grant support from and is a shareholder in Targeted Medical Education. He is also a shareholder in Advantage Consulting and Education.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT MBCC
VIDEO: Resistance to endocrine therapy a moving target
MIAMI BEACH – Hormonal ablation is a mainstay of therapy for women with hormone receptor–positive breast cancer. A significant proportion of patients, however, are either initially refractory to hormonal therapy or acquire resistance to it over time.
The difficulty for patients with breast cancer and for the physicians who treat them is that there are no simple answers to the question of which patients can continue to benefit from endocrine monotherapy. Are there adequate biomarkers for optimal follow-on therapy when a patient experiences disease progression, and what is the optimal sequence of targeted therapy with endocrine inhibitors, disrupters, or other agents?
In a video interview at the annual Miami Breast Cancer Conference, held by Physicians’ Education Resource, William J. Gradishar, MD, of Northwestern University, Chicago, discusses strategies for combating resistance to endocrine ablative therapy, and describes how new therapies and new treatment strategies are being incorporated into National Comprehensive Cancer Network breast cancer guidelines.
Dr. Gradishar reported having no clinical disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
MIAMI BEACH – Hormonal ablation is a mainstay of therapy for women with hormone receptor–positive breast cancer. A significant proportion of patients, however, are either initially refractory to hormonal therapy or acquire resistance to it over time.
The difficulty for patients with breast cancer and for the physicians who treat them is that there are no simple answers to the question of which patients can continue to benefit from endocrine monotherapy. Are there adequate biomarkers for optimal follow-on therapy when a patient experiences disease progression, and what is the optimal sequence of targeted therapy with endocrine inhibitors, disrupters, or other agents?
In a video interview at the annual Miami Breast Cancer Conference, held by Physicians’ Education Resource, William J. Gradishar, MD, of Northwestern University, Chicago, discusses strategies for combating resistance to endocrine ablative therapy, and describes how new therapies and new treatment strategies are being incorporated into National Comprehensive Cancer Network breast cancer guidelines.
Dr. Gradishar reported having no clinical disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
MIAMI BEACH – Hormonal ablation is a mainstay of therapy for women with hormone receptor–positive breast cancer. A significant proportion of patients, however, are either initially refractory to hormonal therapy or acquire resistance to it over time.
The difficulty for patients with breast cancer and for the physicians who treat them is that there are no simple answers to the question of which patients can continue to benefit from endocrine monotherapy. Are there adequate biomarkers for optimal follow-on therapy when a patient experiences disease progression, and what is the optimal sequence of targeted therapy with endocrine inhibitors, disrupters, or other agents?
In a video interview at the annual Miami Breast Cancer Conference, held by Physicians’ Education Resource, William J. Gradishar, MD, of Northwestern University, Chicago, discusses strategies for combating resistance to endocrine ablative therapy, and describes how new therapies and new treatment strategies are being incorporated into National Comprehensive Cancer Network breast cancer guidelines.
Dr. Gradishar reported having no clinical disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT MBCC
Posttreatment survivorship care needs of Spanish-speaking Latinas with breast cancer
After treatment, cancer patients transition to a survivorship phase, often with little information or support. Cancer survivors are at increased risk of recurrence, secondary cancers, comorbid conditions, and late treatment effects.1,2 However, many remain unaware of these risks and the options for managing them3 and face numerous unmet medical, psychosocial, and informational needs that can be addressed through survivorship care programs.4 Anxiety may increase as they lose their treatment team’s support while attempting to reestablish their lives.2 Patients need to know the long-term risks of cancer treatments, probabilities of recurrence and second cancers, effectiveness of surveillance and interventions for managing late effects and psychosocial concerns, and benefits of healthy lifestyles.2
Due to sociocultural and economic factors, Spanish-speaking Latina breast cancer survivors (SSBCS) suffer worse posttreatment health-related quality of life and more pain, fatigue, depressive symptoms, body image issues, and distress than their white counterparts.5-7 However, they are less likely to receive necessary cancer treatment, symptom management, and surveillance. For example, compared with whites, Latina breast cancer survivors receive less guideline-adherent treatment8 and follow-up care, including survivorship information.3,9 SSBCS, in particular have less access to survivorship information.10 Consequently, SSBCS are more likely to report unmet symptom management needs.11
Several breast cancer survivorship program trials have included Latinas,12,13 but their effectiveness has been demonstrated only for depressive symptoms or health worry. A comprehensive assessment of the posttreatment needs of SSBCS would provide a foundation for designing tailored survivorship interventions for this vulnerable group. This study aimed to identify the symptom management, psychosocial, and informational needs of SSBCS during the transition to survivorship from the perspectives of SSBCS and their cancer support providers and cancer physicians.
Methods
We sampled respondents within a 5-county area in Northern California to obtain multiple perspectives of the survivorship care needs of SSBCS using structured and in-depth methods: a telephone survey of SSBCS; semistructured interviews with SSBCS; semistructured interviews with cancer support providers serving SSBCS; and semistructured interviews with physicians providing cancer care for SSBCS. The study protocol was approved by the University of California San Francisco Committee on Human Research.
Sample and procedures
Structured telephone survey with SSBCS. The sample was drawn evenly from San Francisco General Hospital-University of California San Francisco primary care practices and SSBCS from a previous study who agreed to be re-contacted.14 The inclusion criteria were: completed active treatment (except adjuvant hormonal therapy) for nonmetastatic breast cancer within 10 years; living in one of the five counties; primarily Spanish-speaking; and self-identified as Latina. The exclusion criteria were: previous cancer except nonmelanoma skin cancer; terminal illness; or metastatic breast cancer. Study staff mailed potential participants a bilingual letter and information sheet, and bilingual opt-out postcard (6th grade reading level assessed by Flesch-Kincaid grade level statistic). Female bilingual-bicultural research associates conducted interviews of 20-30 minutes in Spanish after obtaining verbal consent. Participants were mailed $20. Surveys were conducted during March-November 2014.
Semistructured in-person interviews with SSBCS. Four community-based organizations (CBOs) in the targeted area providing cancer support services to Latinos agreed to recruit SSBCS for interviews. Inclusion criteria were identical to the survey. Patient navigators or support providers from CBOs contacted women by phone or in-person to invite them to an interview to assess their cancer survivorship needs. Women could choose a focus group or individual interview. With permission, names and contact information were given to study interviewers who called, explained the study, screened for eligibility, and scheduled an interview.
Recruitment was stratified by age (under or over age 50). We sampled women until saturation was achieved (no new themes emerged). Focus groups (90 minutes) were conducted at the CBOs. Individual interviews (45 minutes) were conducted in participants’ homes. Written informed consent was obtained. Participants were paid $50. Interviews were conducted during August-November, 2014, audiotaped, and transcribed.
Semistructured in-person interviews with cancer support providers and physicians. Investigators invited five cancer support providers (three patient navigators from three county hospitals, and two CBO directors of cancer psychosocial support services) and four physicians (three oncologists and one breast cancer surgeon from three county hospitals) to an in-person interview to identify SSBCS’ survivorship care needs. All agreed to participate. No further candidates were approached because saturation was achieved. We obtained written informed consent and 30-minute interviews were conducted in participants’ offices during August-October, 2014. Interviews were audiotaped and transcribed. Participants were paid $50.
Ethical approval. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent. Informed consent was obtained from all individual participants included in the study.
Measures
Structured telephone survey. Based on cancer survivor needs assessments,15 we assessed: physical and emotional symptoms; problems with sleep and memory/concentration; concerns about mortality, family, social isolation, intimacy, appearance; and healthy lifestyles. Items were adapted and translated into Spanish if needed, using forward/backward translation with team reconciliation. These questions used the introduction, “Now I am going to ask you if you have had any problems because of your cancer. In the past month, how much have you been bothered by …” with responses rated on a scale of 1-5 (1, Not at all; 5, A lot). For example, we asked, “In the past month, how much have you been bothered by fatigue?”
Regarding healthy lifestyles, we used the introduction, “Here are some changes women sometimes want to make after cancer. Would you like help with …?” For example, we asked, “Would you like help with getting more exercise?” We asked if they wanted help getting more exercise, eating healthier, managing stress, and doing meditation or yoga (Yes/No).
Semistructured interview guide for SSBCS. Participants were asked about their emotional and physical concerns when treatment ended, current cancer needs, symptoms or late effects, and issues related to relationships, family, employment, insurance, financial hardships, barriers to follow-up care, health behaviors, and survivorship program content. Sample questions are, “Have you had any symptoms or side effects related to your cancer or treatment?” and “What kinds of information do you feel you need now about your cancer or treatment?” A brief questionnaire assessed demographics.
Semistructured interview guide for cancer support providers and physicians. Support providers and physicians were asked about informational, psychosocial, and symptom management needs of SSBCS and recommended self-management content and formats. Sample questions are, “What kinds of information and support do you wish was available to help Spanish-speaking women take care of their health after treatment ends?” and “What do you think are the most pressing emotional needs of Spanish-speaking women after breast cancer treatment ends?” A brief questionnaire assessed demographics.
Analysis
Frequencies are reported for survey items. For questions about symptoms/concerns, we report the frequency of responding that they were bothered Somewhat/Quite a bit/A lot. For healthy lifestyles, we present the frequency answering Yes.
Verbatim semistructured interview transcripts were verified against audiotapes. Using QSR NVIVO software, transcripts were coded independently by two bilingual-bicultural investigators using a constant comparative method to generate coding categories for cancer survivorship needs.16 Coders started with themes specified by the interview guides, expanded them to represent the data, and discussed and reconciled coding discrepancies. Coding was compared by type of interview participant (survivor, support provider, or physician). Triangulation of survey and semistructured interviews occurred through team discussions to verify codes, themes, and implications for interventions.
Results
Telephone survey of SSBCS
Of the telephone survey sampling frame (N = 231), 118 individuals (51%) completed the interview, 37 (16%) were ineligible, 31 (13%) could not be reached, 22 (10%) had incorrect contact information, 19 (8%) refused to participate, and 4 (2%) were deceased. Mean age of the participants was 54.9 years (SD, 12.3); all were foreign-born, with more than half of Mexican origin; and most had less than a high-school education (Table 1). All had completed active treatment, and most (68%) were within 2 years of diagnosis.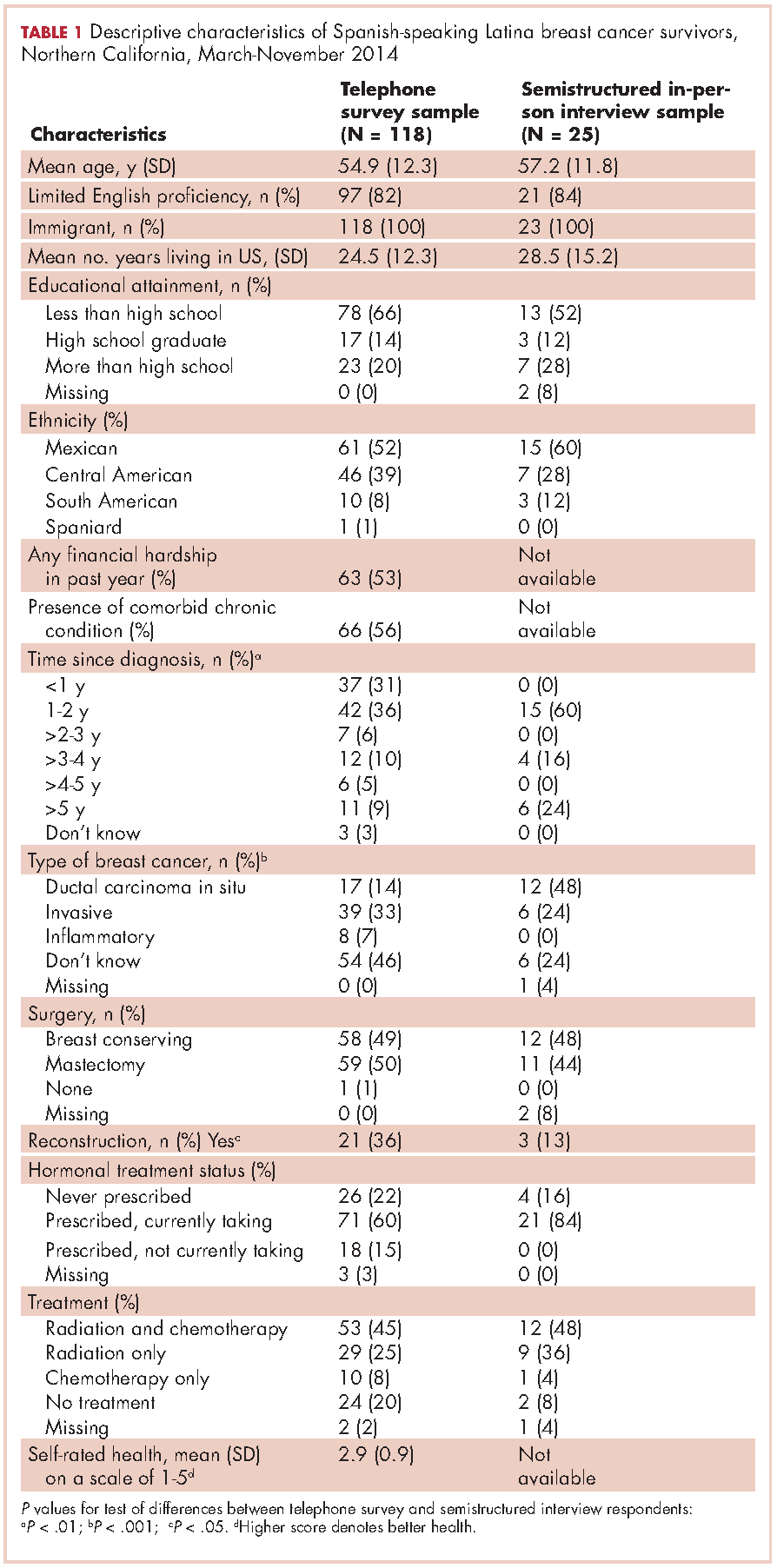
For symptom management needs (Table 2), the most prevalent (bothersome) symptoms (reported by more than 30%, in rank order) were joint pain, sleep problems, fatigue, hot flashes, numbness/tingling of extremities, and memory. Next most prevalent (reported by 20%-30%) were vaginal dryness, dry/itchy skin, dry nose/mouth, inability to concentrate, constipation, changes in urination, and shortness of breath.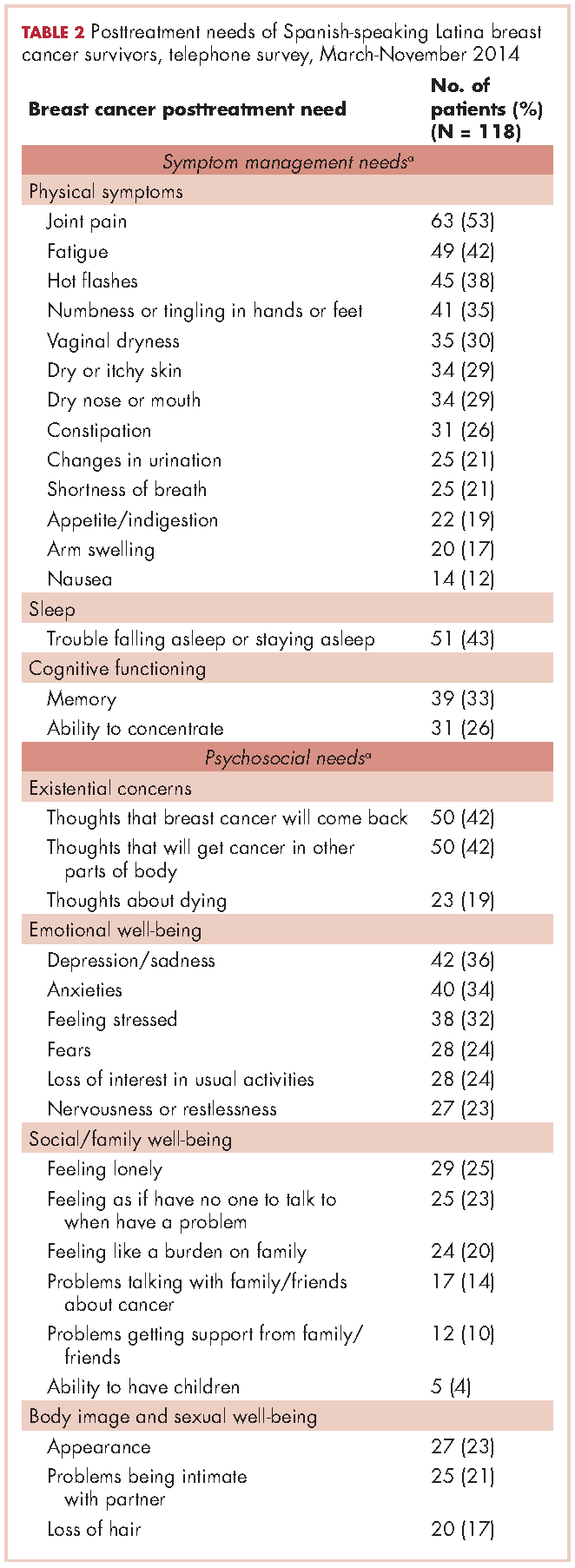

For psychosocial needs, fears of recurrence or new cancers were reported by 42%. Emotional symptoms reported by more than 30% were depression/sadness, anxieties, and feeling stressed. Next most prevalent (20%-30%) were fears, loss of interest in usual activities, and nervousness/restlessness. Social well-being concerns reported by 20%-30% of survivors were loneliness, having no one to talk to, and being a burden to their families. Body image and sexual problems reported by 20%-30% of survivors included appearance and problems being intimate with partners.
Regarding lifestyle, most of the participants said they wanted help with eating a healthier diet (74%), getting more exercise (69%), managing stress (63%), and doing yoga or meditation (55%).
Semistructured interviews
Twenty-five SSBCS completed semistructured interviews, 10 in individual interviews and 15 in one of two focus groups (one of 9 women older than 50 years; one of 6 women younger than 50). The telephone survey respondents were similar to semistructured interview respondents on all sociodemographic characteristics, but differed slightly on some clinical characteristics (Table 1). The telephone survey women had been more recently diagnosed (P < .01), were less likely to have ductal carcinoma in situ (P < .001), and more likely to have had reconstructive surgery
(P < .05).
Five cancer support providers and 4 physicians were interviewed. All support providers were Spanish-speaking Latinas with at least some college education. Cancer physicians were board certified. Two were men; two were white and two Asian; one was a breast surgeon and three were hematologists/oncologists; three spoke Spanish poorly/not at all and one spoke it fairly well.
Seven themes emerged from interviews: unmet physical symptom management needs; social support often ends when treatment ends; challenges resuming roles; sense of abandonment by health care system when treatment ends; need for formal transition from active treatment to follow-up care; fear of recurrence especially when obtaining follow-up care; and desire for information on late effects of initial treatments and side effects of hormonal treatments. We summarize results according to these themes.
Unmet physical symptom management needs. The main physical symptoms reported by survivors and physicians in interviews were arthralgia, menopausal symptoms, and neuropathy. Fatigue was reported only by survivors. Many survivors and several support providers expressed that symptoms were poorly managed and often ignored. One stated,
I have a lot of pain where I had surgery, it burns. I worry a lot about my arm because I have sacs of fluid. My doctor only says, ‘They will dissolve over the years.’ So, I don’t feel any support. (FocGrp1#6)
Survivors reported side effects of hormonal therapies, and felt that physicians downplayed these to prevent them from discontinuing medications.
Social support from family and friends often ends when treatment ends. Many survivors described a loss of support from family and friends who expected them to get back to “normal” once treatment ended. One said,
My sisters have told me to my face that there’s nothing wrong with me. So now when people ask me, I say, ‘I’m fine, thank God, I have nothing,’ even though I’m dying of pain and have all these pills to take. (Survivor#1025)
A support provider related,
The client was telling me that as she was getting closer to finishing her treatment, her husband was upset because he felt like all she was doing was focusing on the cancer. I think caregivers, family, spouses, and children out of their own sort of selfishness want this person to be well. (SuppProv#104)
A few survivors said that family bonds were strengthened after cancer and several reported lacking support because their families were in their home countries.
Challenges resuming roles, especially returning to work. Survivors, support providers, and physicians described challenges and few resources as women transition back to their normal roles. Survivors questioned their ability to return to work due to physically demanding occupations. One stated,
I would like information on how to take care of myself, how working can affect this side if I don’t take care of it. I clean houses and I need both hands. (Survivor#3012)
Survivors described how changes in memory affected daily chores and work performance. Support providers and physicians described the need for resources to aid with return to work and household responsibilities. One physician noted,
There are usually questions about how to go ahead and live their lives from that point forward. It’s a sort of reverse shock: going back to life as they know it. (Physician#004)
Support providers and physicians mentioned that women needed help with resuming intimate partner relations.
Sense of abandonment by health care system once active treatment ends. Survivors, support providers, and physicians reported a loss of support and sense of abandonment by the patients’ oncology team at the end of active treatment. One survivor stated,
Once they tell you to stop the pills, ‘You’re cured, there’s nothing wrong with you,’ the truth is that one feels, ‘Now what do I do? I have no one to help me.’ I felt very abandoned. (FocGrp1#5)
A provider said,
The support system falls apart once women complete treatment. They lose their entire support system at the medical level. They no longer have nurses checking in about symptoms and addressing anything that’s come up. They won’t have access to doctors unless they’re doing their screening. (SuppProv#101)
An oncologist, noting that this loss of support occurs when women face pressures of transitioning back to work or family obligations, commented,
So here’s a woman whose marriage is in turmoil, whose husband may even have left her during this, and now her clinic is leaving her and she’s on her own … that must be scary as hell because there’s nobody out there to support her. (Physician#002)
Need for formal transition from active treatment to follow-up care. Two themes emerged about transitioning from active treatment: transferring care from oncologists to primary care physicians (PCP); and issues of follow-up care (with oncologists or PCPs). Survivors felt lost in transitioning from specialty to primary care, or expressed apprehension seeing a PCP rather than a cancer specialist. One stated,
I have my doctor but she is not a specialist. She does what I tell her to and orders a mammogram every year. But, I don’t go to the oncologist anymore, and so I worry. With the specialists, I feel protected. (FocGrp1#5)
Physicians acknowledged the lack of a formal transition to primary care such as a survivorship care program.
Follow-up care issues were common. Physicians stressed that women needed to know how often to return for follow-up once active treatment ends and about recommended examinations and tests, especially when receiving hormonal therapy. Physicians indicated the need for patient education materials specific to patients’ treatments, for example, elevated risk of heart disease with certain chemotherapy agents. An oncologist expressed concern that PCPs are not prepared adequately about late effects and hormonal treatment side effects, and suggested providing summary notes for PCPs detailing these.
Survivors identified several barriers to follow-up care: lacking information on which symptoms merited a call to physicians; financial burden/limited health insurance; lacking appointment reminders; fear of examinations; and limited English proficiency. A survivor stated,
If you have insurance, you can make your appointment, see the doctor, and have your mammogram. I stopped taking my pills because I didn’t have insurance. I tried to get them again but they told me they would cost me a thousand dollars. (FocGrp1#5)
One oncologist suggested scheduling a follow-up appointment before patients leave treatment and calling patients who miss appointments.
Facilitators of regular follow-up care identified by survivors were physicians informing them about symptom monitoring and reporting, having a clinic contact person/navigator, being given a follow-up appointment, being assertive about one’s care, and physicians’ reinforcement of adherence to hormonal treatment and follow-up. According to support providers, a key facilitator was having a clinic contact person/navigator. Once treatment ended, support providers often served as the liaison between the patient and the physician, making them the first point of contact for symptom reporting.
Fear of recurrence especially when obtaining follow-up care. Fear of recurrence dominated survivor interviews. This fear was heightened at the time of follow-up examinations or when they experienced unusual pain. A survivor commented,
Every time I’m due for my mammogram, I can’t sleep, worrying. I lose sleep until I get the letter with my results. Then I feel at peace again. (FocGrp1#9)
Support providers discussed the need to provide reassurance to SSBCS to help them cope with fears of recurrence. Physicians expressed challenges in allaying fears of recurrence among SSBCS, requiring a lot of time when recommending follow-up mammograms.
Desire for information on late effects of treatments and side effects of hormonal therapies. All survivors expressed receiving insufficient information on potential symptoms and side effects. One stated,
Doctors only have five minutes. There has never been someone who gave me guidance like, ‘From now on you have to do this or you might get these symptoms now or in the future. (Survivor#6019)
They indicated uncertainty about what symptoms were “normal” and when symptoms merited a call to the physician. Several survivors reported being unaware that fatigue, arthralgia and neuropathy were side effects of breast cancer treatments until they reported these to physicians.
Physicians stressed the importance of women knowing about the elevated risk of future cancers, symptoms of recurrence, and seeking follow-up care if they experience symptoms that are out of the ordinary. Support providers felt that it was important to provide SSBCS with information on signs of recurrence and when to report these. However, providers expressed concern that giving women too much information might elevate their anxiety. A physician suggested,
It’s probably better to have a symptom list that’s short and relevant for the most common and catastrophic things, same thing with side effects … short to avoid overwhelming the patient. (Physician#001)
Hormonal treatments were of special concern. Survivors expressed a need for information on hormonal treatments and support providers stressed that this information is needed in simple Spanish. Several survivors indicated they stopped taking hormonal treatments due to side effects. One woman experienced severe headaches and heart palpitations, stopped taking the hormonal medication, felt better, and did not inform her physician until her next appointment. A support provider stated,
What I hear from a lot of women is that if side effects are too uncomfortable, they just stop it (hormonal treatment) without saying anything to the doctor. So more information about why they have to take it and that there is a good chance of recurrence is really important. (SuppProv#101)
Likewise, physicians indicated that SSBCS’ lack of information on hormonal treatments often resulted in nonadherence, emphasizing the need to reinforce adherence to prevent recurrence.
Conceptual framework of interventions
Based on triangulation of survey and interview results, we compiled a conceptual framework that includes needs identified, suggested components of a survivorship care intervention to address these needs, potential mediators by which such interventions could improve outcomes, and relevant outcomes (Figure). Survivorship care needs fell into four categories: symptom management, psychosocial, sense of abandonment by health care team, and healthy lifestyles. Survivorship care programs would provide skills training in symptom and stress management, and communicating with providers, family, friends, and coworkers. Mediators include increased self-efficacy, knowledge and perceived social support, ultimately leading to reduced distress (anxiety and depressive symptoms) and stress, and improved health-related quality of life.
Conclusions
Our study aimed to identify the most critical needs of SSBCS in the posttreatment survivorship phase to facilitate the design of survivorship interventions for this vulnerable group. SSBCS, cancer support providers, and cancer physicians reported substantial symptom management, psychosocial, and informational needs among this population. Results from surveys and open-ended interviews were remarkably consistent. Survivors, physicians, and support providers viewed transition out of active treatment as a time of increased psychosocial need and heightened vulnerability.
Our findings are consistent with needs assessments conducted in other breast cancer survivors. Similar to a study of rural white women with breast cancer, fear of recurrence was among the most common psychosocial concerns.17 Results of two studies that included white, African American and Latina breast cancer survivors were consistent with ours in finding that pain and fatigue were among the most persistent symptoms; in both studies, Latinas were more likely to report pain and a higher number of symptoms.7,18 The prevalence of sleep problems in our sample was identical to that reported in a sample of African American breast cancer survivors.19 Our findings of a high need for symptom management information and support, social support from family and friends, and self-management resources were similar to studies of other vulnerable breast cancer survivors.18,20
Our results suggest that it is critical for health care professionals to provide assistance with managing side effects and information to alleviate fears, and reinforce behaviors of symptom monitoring and reporting, and adherence to follow-up care and hormonal therapies. Yet this information is not being conveyed effectively and is complicated by the need to balance women’s need for information with minimizing anxiety when providing such information.
A limitation of our study is that most of our sample was Mexican origin and may not reflect experiences of Spanish-speaking Latinas of other national origin groups or outside of Northern California. Another limitation is the lack of an English-speaking comparison group, which would have permitted the identification of similarities and differences across language groups. Finally, we did not interview radiation oncologists who may have had opinions that are not represented here.
Survivorship care programs offer great promise for meeting patients’ informational and symptom management needs and improving well-being and communication with clinicians.21 Due to limited access to survivorship care information, financial hardships, and pressures from their families to resume their social roles, concerted efforts are needed to develop appropriate survivorship programs for SSBCS.22 Unique language, cultural and socioeconomic factors of Spanish-speaking Latinas require tailoring of cancer survivorship programs to best meet their needs.23 These programs need to provide psychosocial stress and symptom management assistance, simple information on recommended follow-up care, and healthy lifestyle and role reintegration strategies that account for their unique sociocultural contexts.
1. Danese MD, O’Malley C, Lindquist K, Gleeson M, Griffiths RI. An observational study of the prevalence and incidence of comorbid conditions in older women with breast cancer. Ann Oncol. 2012;23(7):1756-1765.
2. Hewitt M, Greenfield S, Stovall E, eds. From cancer patient to cancer survivor: lost in transition. Washington, DC: National Academy of Sciences; 2006.
3. Beckjord EB, Arora NK, McLaughlin W, Oakley-Girvan I, Hamilton AS, Hesse BW. Health-related information needs in a large and diverse sample of adult cancer survivors: implications for cancer care. J Cancer Surviv. 2008;2(3):179-189.
4. Hewitt ME, Bamundo A, Day R, Harvey C. Perspectives on posttreatment cancer care: qualitative research with survivors, nurses, and physicians. J Clin Oncol. 2007;25(16):2270-2273.
5. Ashing-Giwa KT, Tejero JS, Kim J, Padilla GV, Hellemann G. Examining predictive models of HRQOL in a population-based, multiethnic sample of women with breast carcinoma. Qual Life Res. 2007;16(3):413-428.
6. Clauser SB, Arora NK, Bellizzi KM, Haffer SC, Topor M, Hays RD. Disparities in HRQOL of cancer survivors and non-cancer managed care enrollees. Health Care Financ Rev. 2008;29(4):23-40.
7. Eversley R, Estrin D, Dibble S, Wardlaw L, Pedrosa M, Favila-Penney W. Posttreatment symptoms among ethnic minority breast cancer survivors. Oncol Nurs Forum. 2005;32(2):250-256.
8. Bickell NA, Wang JJ, Oluwole S, et al. Missed opportunities: racial disparities in adjuvant breast cancer treatment. J Clin Oncol. 2006;24(9):1357-1362.
9. Arora NK, Reeve BB, Hays RD, Clauser SB, Oakley-Girvan I. Assessment of quality of cancer-related follow-up care from the cancer survivor’s perspective. J Clin Oncol. 2011;29(10):1280-1289.
10. Janz NK, Mujahid MS, Hawley ST, Griggs JJ, Hamilton AS, Katz SJ. Racial/ethnic differences in adequacy of information and support for women with breast cancer. Cancer. 2008;113(5):1058-1067.
11. Yoon J, Malin JL, Tisnado DM, et al. Symptom management after breast cancer treatment: is it influenced by patient characteristics? Breast Cancer Res Treat. 2008;108(1):69-77.
12. Ashing K, Rosales M. A telephonic-based trial to reduce depressive symptoms among Latina breast cancer survivors. Psychooncology. 2014;23(5):507-515.
13. Hershman DL, Greenlee H, Awad D, et al. Randomized controlled trial of a clinic-based survivorship intervention following adjuvant therapy in breast cancer survivors. Breast Cancer Res Treat. 2013;138(3):795-806.
14. Napoles AM, Ortiz C, Santoyo-Olsson J, et al. Nuevo Amanecer: results of a randomized controlled trial of a community-based, peer-delivered stress management intervention to improve quality of life in Latinas with breast cancer. Am J Public Health. 2015;105(suppl 3):e55-63.
15. Rechis R, Reynolds KA, Beckjord EB, Nutt S, Burns RM, Schaefer JS. ‘I learned to live with it’ is not good enough: challenges reported by posttreatment cancer survivors in the Livestrong surveys. Austin, TX: Livestrong;2011.
16. Glaser BG, Strauss AL. The discovery of grounded theory: strategies for qualitative research. Hawthorne: Aldine Publishing Company; 1967.
17. Befort CA, Klemp J. Sequelae of breast cancer and the influence of menopausal status at diagnosis among rural breast cancer survivors. J Womens Health (Larchmt). 2011;20(9):1307-1313.
18. Fu OS, Crew KD, Jacobson JS, et al. Ethnicity and persistent symptom burden in breast cancer survivors. J Cancer Surviv. 2009;3(4):241-250.
19. Taylor TR, Huntley ED, Makambi K, et al. Understanding sleep disturbances in African-American breast cancer survivors: a pilot study. Psychooncology. 2012;21(8):896-902.
20. Adams N, Gisiger-Camata S, Hardy CM, Thomas TF, Jukkala A, Meneses K. Evaluating survivorship experiences and needs among rural African American breast cancer survivors. J Cancer Educ. October 24, 2015 [Epub ahead of print].
21. Blinder VS, Patil S, Thind A, et al. Return to work in low-income Latina and non-Latina white breast cancer survivors: a 3-year longitudinal study. Cancer. 2012;118(6):1664-1674.
22. Lopez-Class M, Perret-Gentil M, Kreling B, Caicedo L, Mandelblatt J, Graves KD. Quality of life among immigrant Latina breast cancer survivors: realities of culture and enhancing cancer care. J Cancer Educ. 2011;26(4):724-733.
23. Napoles-Springer AM, Ortiz C, O’Brien H, Diaz-Mendez M. Developing a culturally competent peer support intervention for Spanish-speaking Latinas with breast cancer. J Immigr Minor Health. 2009;11(4):268-280
After treatment, cancer patients transition to a survivorship phase, often with little information or support. Cancer survivors are at increased risk of recurrence, secondary cancers, comorbid conditions, and late treatment effects.1,2 However, many remain unaware of these risks and the options for managing them3 and face numerous unmet medical, psychosocial, and informational needs that can be addressed through survivorship care programs.4 Anxiety may increase as they lose their treatment team’s support while attempting to reestablish their lives.2 Patients need to know the long-term risks of cancer treatments, probabilities of recurrence and second cancers, effectiveness of surveillance and interventions for managing late effects and psychosocial concerns, and benefits of healthy lifestyles.2
Due to sociocultural and economic factors, Spanish-speaking Latina breast cancer survivors (SSBCS) suffer worse posttreatment health-related quality of life and more pain, fatigue, depressive symptoms, body image issues, and distress than their white counterparts.5-7 However, they are less likely to receive necessary cancer treatment, symptom management, and surveillance. For example, compared with whites, Latina breast cancer survivors receive less guideline-adherent treatment8 and follow-up care, including survivorship information.3,9 SSBCS, in particular have less access to survivorship information.10 Consequently, SSBCS are more likely to report unmet symptom management needs.11
Several breast cancer survivorship program trials have included Latinas,12,13 but their effectiveness has been demonstrated only for depressive symptoms or health worry. A comprehensive assessment of the posttreatment needs of SSBCS would provide a foundation for designing tailored survivorship interventions for this vulnerable group. This study aimed to identify the symptom management, psychosocial, and informational needs of SSBCS during the transition to survivorship from the perspectives of SSBCS and their cancer support providers and cancer physicians.
Methods
We sampled respondents within a 5-county area in Northern California to obtain multiple perspectives of the survivorship care needs of SSBCS using structured and in-depth methods: a telephone survey of SSBCS; semistructured interviews with SSBCS; semistructured interviews with cancer support providers serving SSBCS; and semistructured interviews with physicians providing cancer care for SSBCS. The study protocol was approved by the University of California San Francisco Committee on Human Research.
Sample and procedures
Structured telephone survey with SSBCS. The sample was drawn evenly from San Francisco General Hospital-University of California San Francisco primary care practices and SSBCS from a previous study who agreed to be re-contacted.14 The inclusion criteria were: completed active treatment (except adjuvant hormonal therapy) for nonmetastatic breast cancer within 10 years; living in one of the five counties; primarily Spanish-speaking; and self-identified as Latina. The exclusion criteria were: previous cancer except nonmelanoma skin cancer; terminal illness; or metastatic breast cancer. Study staff mailed potential participants a bilingual letter and information sheet, and bilingual opt-out postcard (6th grade reading level assessed by Flesch-Kincaid grade level statistic). Female bilingual-bicultural research associates conducted interviews of 20-30 minutes in Spanish after obtaining verbal consent. Participants were mailed $20. Surveys were conducted during March-November 2014.
Semistructured in-person interviews with SSBCS. Four community-based organizations (CBOs) in the targeted area providing cancer support services to Latinos agreed to recruit SSBCS for interviews. Inclusion criteria were identical to the survey. Patient navigators or support providers from CBOs contacted women by phone or in-person to invite them to an interview to assess their cancer survivorship needs. Women could choose a focus group or individual interview. With permission, names and contact information were given to study interviewers who called, explained the study, screened for eligibility, and scheduled an interview.
Recruitment was stratified by age (under or over age 50). We sampled women until saturation was achieved (no new themes emerged). Focus groups (90 minutes) were conducted at the CBOs. Individual interviews (45 minutes) were conducted in participants’ homes. Written informed consent was obtained. Participants were paid $50. Interviews were conducted during August-November, 2014, audiotaped, and transcribed.
Semistructured in-person interviews with cancer support providers and physicians. Investigators invited five cancer support providers (three patient navigators from three county hospitals, and two CBO directors of cancer psychosocial support services) and four physicians (three oncologists and one breast cancer surgeon from three county hospitals) to an in-person interview to identify SSBCS’ survivorship care needs. All agreed to participate. No further candidates were approached because saturation was achieved. We obtained written informed consent and 30-minute interviews were conducted in participants’ offices during August-October, 2014. Interviews were audiotaped and transcribed. Participants were paid $50.
Ethical approval. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent. Informed consent was obtained from all individual participants included in the study.
Measures
Structured telephone survey. Based on cancer survivor needs assessments,15 we assessed: physical and emotional symptoms; problems with sleep and memory/concentration; concerns about mortality, family, social isolation, intimacy, appearance; and healthy lifestyles. Items were adapted and translated into Spanish if needed, using forward/backward translation with team reconciliation. These questions used the introduction, “Now I am going to ask you if you have had any problems because of your cancer. In the past month, how much have you been bothered by …” with responses rated on a scale of 1-5 (1, Not at all; 5, A lot). For example, we asked, “In the past month, how much have you been bothered by fatigue?”
Regarding healthy lifestyles, we used the introduction, “Here are some changes women sometimes want to make after cancer. Would you like help with …?” For example, we asked, “Would you like help with getting more exercise?” We asked if they wanted help getting more exercise, eating healthier, managing stress, and doing meditation or yoga (Yes/No).
Semistructured interview guide for SSBCS. Participants were asked about their emotional and physical concerns when treatment ended, current cancer needs, symptoms or late effects, and issues related to relationships, family, employment, insurance, financial hardships, barriers to follow-up care, health behaviors, and survivorship program content. Sample questions are, “Have you had any symptoms or side effects related to your cancer or treatment?” and “What kinds of information do you feel you need now about your cancer or treatment?” A brief questionnaire assessed demographics.
Semistructured interview guide for cancer support providers and physicians. Support providers and physicians were asked about informational, psychosocial, and symptom management needs of SSBCS and recommended self-management content and formats. Sample questions are, “What kinds of information and support do you wish was available to help Spanish-speaking women take care of their health after treatment ends?” and “What do you think are the most pressing emotional needs of Spanish-speaking women after breast cancer treatment ends?” A brief questionnaire assessed demographics.
Analysis
Frequencies are reported for survey items. For questions about symptoms/concerns, we report the frequency of responding that they were bothered Somewhat/Quite a bit/A lot. For healthy lifestyles, we present the frequency answering Yes.
Verbatim semistructured interview transcripts were verified against audiotapes. Using QSR NVIVO software, transcripts were coded independently by two bilingual-bicultural investigators using a constant comparative method to generate coding categories for cancer survivorship needs.16 Coders started with themes specified by the interview guides, expanded them to represent the data, and discussed and reconciled coding discrepancies. Coding was compared by type of interview participant (survivor, support provider, or physician). Triangulation of survey and semistructured interviews occurred through team discussions to verify codes, themes, and implications for interventions.
Results
Telephone survey of SSBCS
Of the telephone survey sampling frame (N = 231), 118 individuals (51%) completed the interview, 37 (16%) were ineligible, 31 (13%) could not be reached, 22 (10%) had incorrect contact information, 19 (8%) refused to participate, and 4 (2%) were deceased. Mean age of the participants was 54.9 years (SD, 12.3); all were foreign-born, with more than half of Mexican origin; and most had less than a high-school education (Table 1). All had completed active treatment, and most (68%) were within 2 years of diagnosis.
For symptom management needs (Table 2), the most prevalent (bothersome) symptoms (reported by more than 30%, in rank order) were joint pain, sleep problems, fatigue, hot flashes, numbness/tingling of extremities, and memory. Next most prevalent (reported by 20%-30%) were vaginal dryness, dry/itchy skin, dry nose/mouth, inability to concentrate, constipation, changes in urination, and shortness of breath.

For psychosocial needs, fears of recurrence or new cancers were reported by 42%. Emotional symptoms reported by more than 30% were depression/sadness, anxieties, and feeling stressed. Next most prevalent (20%-30%) were fears, loss of interest in usual activities, and nervousness/restlessness. Social well-being concerns reported by 20%-30% of survivors were loneliness, having no one to talk to, and being a burden to their families. Body image and sexual problems reported by 20%-30% of survivors included appearance and problems being intimate with partners.
Regarding lifestyle, most of the participants said they wanted help with eating a healthier diet (74%), getting more exercise (69%), managing stress (63%), and doing yoga or meditation (55%).
Semistructured interviews
Twenty-five SSBCS completed semistructured interviews, 10 in individual interviews and 15 in one of two focus groups (one of 9 women older than 50 years; one of 6 women younger than 50). The telephone survey respondents were similar to semistructured interview respondents on all sociodemographic characteristics, but differed slightly on some clinical characteristics (Table 1). The telephone survey women had been more recently diagnosed (P < .01), were less likely to have ductal carcinoma in situ (P < .001), and more likely to have had reconstructive surgery
(P < .05).
Five cancer support providers and 4 physicians were interviewed. All support providers were Spanish-speaking Latinas with at least some college education. Cancer physicians were board certified. Two were men; two were white and two Asian; one was a breast surgeon and three were hematologists/oncologists; three spoke Spanish poorly/not at all and one spoke it fairly well.
Seven themes emerged from interviews: unmet physical symptom management needs; social support often ends when treatment ends; challenges resuming roles; sense of abandonment by health care system when treatment ends; need for formal transition from active treatment to follow-up care; fear of recurrence especially when obtaining follow-up care; and desire for information on late effects of initial treatments and side effects of hormonal treatments. We summarize results according to these themes.
Unmet physical symptom management needs. The main physical symptoms reported by survivors and physicians in interviews were arthralgia, menopausal symptoms, and neuropathy. Fatigue was reported only by survivors. Many survivors and several support providers expressed that symptoms were poorly managed and often ignored. One stated,
I have a lot of pain where I had surgery, it burns. I worry a lot about my arm because I have sacs of fluid. My doctor only says, ‘They will dissolve over the years.’ So, I don’t feel any support. (FocGrp1#6)
Survivors reported side effects of hormonal therapies, and felt that physicians downplayed these to prevent them from discontinuing medications.
Social support from family and friends often ends when treatment ends. Many survivors described a loss of support from family and friends who expected them to get back to “normal” once treatment ended. One said,
My sisters have told me to my face that there’s nothing wrong with me. So now when people ask me, I say, ‘I’m fine, thank God, I have nothing,’ even though I’m dying of pain and have all these pills to take. (Survivor#1025)
A support provider related,
The client was telling me that as she was getting closer to finishing her treatment, her husband was upset because he felt like all she was doing was focusing on the cancer. I think caregivers, family, spouses, and children out of their own sort of selfishness want this person to be well. (SuppProv#104)
A few survivors said that family bonds were strengthened after cancer and several reported lacking support because their families were in their home countries.
Challenges resuming roles, especially returning to work. Survivors, support providers, and physicians described challenges and few resources as women transition back to their normal roles. Survivors questioned their ability to return to work due to physically demanding occupations. One stated,
I would like information on how to take care of myself, how working can affect this side if I don’t take care of it. I clean houses and I need both hands. (Survivor#3012)
Survivors described how changes in memory affected daily chores and work performance. Support providers and physicians described the need for resources to aid with return to work and household responsibilities. One physician noted,
There are usually questions about how to go ahead and live their lives from that point forward. It’s a sort of reverse shock: going back to life as they know it. (Physician#004)
Support providers and physicians mentioned that women needed help with resuming intimate partner relations.
Sense of abandonment by health care system once active treatment ends. Survivors, support providers, and physicians reported a loss of support and sense of abandonment by the patients’ oncology team at the end of active treatment. One survivor stated,
Once they tell you to stop the pills, ‘You’re cured, there’s nothing wrong with you,’ the truth is that one feels, ‘Now what do I do? I have no one to help me.’ I felt very abandoned. (FocGrp1#5)
A provider said,
The support system falls apart once women complete treatment. They lose their entire support system at the medical level. They no longer have nurses checking in about symptoms and addressing anything that’s come up. They won’t have access to doctors unless they’re doing their screening. (SuppProv#101)
An oncologist, noting that this loss of support occurs when women face pressures of transitioning back to work or family obligations, commented,
So here’s a woman whose marriage is in turmoil, whose husband may even have left her during this, and now her clinic is leaving her and she’s on her own … that must be scary as hell because there’s nobody out there to support her. (Physician#002)
Need for formal transition from active treatment to follow-up care. Two themes emerged about transitioning from active treatment: transferring care from oncologists to primary care physicians (PCP); and issues of follow-up care (with oncologists or PCPs). Survivors felt lost in transitioning from specialty to primary care, or expressed apprehension seeing a PCP rather than a cancer specialist. One stated,
I have my doctor but she is not a specialist. She does what I tell her to and orders a mammogram every year. But, I don’t go to the oncologist anymore, and so I worry. With the specialists, I feel protected. (FocGrp1#5)
Physicians acknowledged the lack of a formal transition to primary care such as a survivorship care program.
Follow-up care issues were common. Physicians stressed that women needed to know how often to return for follow-up once active treatment ends and about recommended examinations and tests, especially when receiving hormonal therapy. Physicians indicated the need for patient education materials specific to patients’ treatments, for example, elevated risk of heart disease with certain chemotherapy agents. An oncologist expressed concern that PCPs are not prepared adequately about late effects and hormonal treatment side effects, and suggested providing summary notes for PCPs detailing these.
Survivors identified several barriers to follow-up care: lacking information on which symptoms merited a call to physicians; financial burden/limited health insurance; lacking appointment reminders; fear of examinations; and limited English proficiency. A survivor stated,
If you have insurance, you can make your appointment, see the doctor, and have your mammogram. I stopped taking my pills because I didn’t have insurance. I tried to get them again but they told me they would cost me a thousand dollars. (FocGrp1#5)
One oncologist suggested scheduling a follow-up appointment before patients leave treatment and calling patients who miss appointments.
Facilitators of regular follow-up care identified by survivors were physicians informing them about symptom monitoring and reporting, having a clinic contact person/navigator, being given a follow-up appointment, being assertive about one’s care, and physicians’ reinforcement of adherence to hormonal treatment and follow-up. According to support providers, a key facilitator was having a clinic contact person/navigator. Once treatment ended, support providers often served as the liaison between the patient and the physician, making them the first point of contact for symptom reporting.
Fear of recurrence especially when obtaining follow-up care. Fear of recurrence dominated survivor interviews. This fear was heightened at the time of follow-up examinations or when they experienced unusual pain. A survivor commented,
Every time I’m due for my mammogram, I can’t sleep, worrying. I lose sleep until I get the letter with my results. Then I feel at peace again. (FocGrp1#9)
Support providers discussed the need to provide reassurance to SSBCS to help them cope with fears of recurrence. Physicians expressed challenges in allaying fears of recurrence among SSBCS, requiring a lot of time when recommending follow-up mammograms.
Desire for information on late effects of treatments and side effects of hormonal therapies. All survivors expressed receiving insufficient information on potential symptoms and side effects. One stated,
Doctors only have five minutes. There has never been someone who gave me guidance like, ‘From now on you have to do this or you might get these symptoms now or in the future. (Survivor#6019)
They indicated uncertainty about what symptoms were “normal” and when symptoms merited a call to the physician. Several survivors reported being unaware that fatigue, arthralgia and neuropathy were side effects of breast cancer treatments until they reported these to physicians.
Physicians stressed the importance of women knowing about the elevated risk of future cancers, symptoms of recurrence, and seeking follow-up care if they experience symptoms that are out of the ordinary. Support providers felt that it was important to provide SSBCS with information on signs of recurrence and when to report these. However, providers expressed concern that giving women too much information might elevate their anxiety. A physician suggested,
It’s probably better to have a symptom list that’s short and relevant for the most common and catastrophic things, same thing with side effects … short to avoid overwhelming the patient. (Physician#001)
Hormonal treatments were of special concern. Survivors expressed a need for information on hormonal treatments and support providers stressed that this information is needed in simple Spanish. Several survivors indicated they stopped taking hormonal treatments due to side effects. One woman experienced severe headaches and heart palpitations, stopped taking the hormonal medication, felt better, and did not inform her physician until her next appointment. A support provider stated,
What I hear from a lot of women is that if side effects are too uncomfortable, they just stop it (hormonal treatment) without saying anything to the doctor. So more information about why they have to take it and that there is a good chance of recurrence is really important. (SuppProv#101)
Likewise, physicians indicated that SSBCS’ lack of information on hormonal treatments often resulted in nonadherence, emphasizing the need to reinforce adherence to prevent recurrence.
Conceptual framework of interventions
Based on triangulation of survey and interview results, we compiled a conceptual framework that includes needs identified, suggested components of a survivorship care intervention to address these needs, potential mediators by which such interventions could improve outcomes, and relevant outcomes (Figure). Survivorship care needs fell into four categories: symptom management, psychosocial, sense of abandonment by health care team, and healthy lifestyles. Survivorship care programs would provide skills training in symptom and stress management, and communicating with providers, family, friends, and coworkers. Mediators include increased self-efficacy, knowledge and perceived social support, ultimately leading to reduced distress (anxiety and depressive symptoms) and stress, and improved health-related quality of life.
Conclusions
Our study aimed to identify the most critical needs of SSBCS in the posttreatment survivorship phase to facilitate the design of survivorship interventions for this vulnerable group. SSBCS, cancer support providers, and cancer physicians reported substantial symptom management, psychosocial, and informational needs among this population. Results from surveys and open-ended interviews were remarkably consistent. Survivors, physicians, and support providers viewed transition out of active treatment as a time of increased psychosocial need and heightened vulnerability.
Our findings are consistent with needs assessments conducted in other breast cancer survivors. Similar to a study of rural white women with breast cancer, fear of recurrence was among the most common psychosocial concerns.17 Results of two studies that included white, African American and Latina breast cancer survivors were consistent with ours in finding that pain and fatigue were among the most persistent symptoms; in both studies, Latinas were more likely to report pain and a higher number of symptoms.7,18 The prevalence of sleep problems in our sample was identical to that reported in a sample of African American breast cancer survivors.19 Our findings of a high need for symptom management information and support, social support from family and friends, and self-management resources were similar to studies of other vulnerable breast cancer survivors.18,20
Our results suggest that it is critical for health care professionals to provide assistance with managing side effects and information to alleviate fears, and reinforce behaviors of symptom monitoring and reporting, and adherence to follow-up care and hormonal therapies. Yet this information is not being conveyed effectively and is complicated by the need to balance women’s need for information with minimizing anxiety when providing such information.
A limitation of our study is that most of our sample was Mexican origin and may not reflect experiences of Spanish-speaking Latinas of other national origin groups or outside of Northern California. Another limitation is the lack of an English-speaking comparison group, which would have permitted the identification of similarities and differences across language groups. Finally, we did not interview radiation oncologists who may have had opinions that are not represented here.
Survivorship care programs offer great promise for meeting patients’ informational and symptom management needs and improving well-being and communication with clinicians.21 Due to limited access to survivorship care information, financial hardships, and pressures from their families to resume their social roles, concerted efforts are needed to develop appropriate survivorship programs for SSBCS.22 Unique language, cultural and socioeconomic factors of Spanish-speaking Latinas require tailoring of cancer survivorship programs to best meet their needs.23 These programs need to provide psychosocial stress and symptom management assistance, simple information on recommended follow-up care, and healthy lifestyle and role reintegration strategies that account for their unique sociocultural contexts.
After treatment, cancer patients transition to a survivorship phase, often with little information or support. Cancer survivors are at increased risk of recurrence, secondary cancers, comorbid conditions, and late treatment effects.1,2 However, many remain unaware of these risks and the options for managing them3 and face numerous unmet medical, psychosocial, and informational needs that can be addressed through survivorship care programs.4 Anxiety may increase as they lose their treatment team’s support while attempting to reestablish their lives.2 Patients need to know the long-term risks of cancer treatments, probabilities of recurrence and second cancers, effectiveness of surveillance and interventions for managing late effects and psychosocial concerns, and benefits of healthy lifestyles.2
Due to sociocultural and economic factors, Spanish-speaking Latina breast cancer survivors (SSBCS) suffer worse posttreatment health-related quality of life and more pain, fatigue, depressive symptoms, body image issues, and distress than their white counterparts.5-7 However, they are less likely to receive necessary cancer treatment, symptom management, and surveillance. For example, compared with whites, Latina breast cancer survivors receive less guideline-adherent treatment8 and follow-up care, including survivorship information.3,9 SSBCS, in particular have less access to survivorship information.10 Consequently, SSBCS are more likely to report unmet symptom management needs.11
Several breast cancer survivorship program trials have included Latinas,12,13 but their effectiveness has been demonstrated only for depressive symptoms or health worry. A comprehensive assessment of the posttreatment needs of SSBCS would provide a foundation for designing tailored survivorship interventions for this vulnerable group. This study aimed to identify the symptom management, psychosocial, and informational needs of SSBCS during the transition to survivorship from the perspectives of SSBCS and their cancer support providers and cancer physicians.
Methods
We sampled respondents within a 5-county area in Northern California to obtain multiple perspectives of the survivorship care needs of SSBCS using structured and in-depth methods: a telephone survey of SSBCS; semistructured interviews with SSBCS; semistructured interviews with cancer support providers serving SSBCS; and semistructured interviews with physicians providing cancer care for SSBCS. The study protocol was approved by the University of California San Francisco Committee on Human Research.
Sample and procedures
Structured telephone survey with SSBCS. The sample was drawn evenly from San Francisco General Hospital-University of California San Francisco primary care practices and SSBCS from a previous study who agreed to be re-contacted.14 The inclusion criteria were: completed active treatment (except adjuvant hormonal therapy) for nonmetastatic breast cancer within 10 years; living in one of the five counties; primarily Spanish-speaking; and self-identified as Latina. The exclusion criteria were: previous cancer except nonmelanoma skin cancer; terminal illness; or metastatic breast cancer. Study staff mailed potential participants a bilingual letter and information sheet, and bilingual opt-out postcard (6th grade reading level assessed by Flesch-Kincaid grade level statistic). Female bilingual-bicultural research associates conducted interviews of 20-30 minutes in Spanish after obtaining verbal consent. Participants were mailed $20. Surveys were conducted during March-November 2014.
Semistructured in-person interviews with SSBCS. Four community-based organizations (CBOs) in the targeted area providing cancer support services to Latinos agreed to recruit SSBCS for interviews. Inclusion criteria were identical to the survey. Patient navigators or support providers from CBOs contacted women by phone or in-person to invite them to an interview to assess their cancer survivorship needs. Women could choose a focus group or individual interview. With permission, names and contact information were given to study interviewers who called, explained the study, screened for eligibility, and scheduled an interview.
Recruitment was stratified by age (under or over age 50). We sampled women until saturation was achieved (no new themes emerged). Focus groups (90 minutes) were conducted at the CBOs. Individual interviews (45 minutes) were conducted in participants’ homes. Written informed consent was obtained. Participants were paid $50. Interviews were conducted during August-November, 2014, audiotaped, and transcribed.
Semistructured in-person interviews with cancer support providers and physicians. Investigators invited five cancer support providers (three patient navigators from three county hospitals, and two CBO directors of cancer psychosocial support services) and four physicians (three oncologists and one breast cancer surgeon from three county hospitals) to an in-person interview to identify SSBCS’ survivorship care needs. All agreed to participate. No further candidates were approached because saturation was achieved. We obtained written informed consent and 30-minute interviews were conducted in participants’ offices during August-October, 2014. Interviews were audiotaped and transcribed. Participants were paid $50.
Ethical approval. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent. Informed consent was obtained from all individual participants included in the study.
Measures
Structured telephone survey. Based on cancer survivor needs assessments,15 we assessed: physical and emotional symptoms; problems with sleep and memory/concentration; concerns about mortality, family, social isolation, intimacy, appearance; and healthy lifestyles. Items were adapted and translated into Spanish if needed, using forward/backward translation with team reconciliation. These questions used the introduction, “Now I am going to ask you if you have had any problems because of your cancer. In the past month, how much have you been bothered by …” with responses rated on a scale of 1-5 (1, Not at all; 5, A lot). For example, we asked, “In the past month, how much have you been bothered by fatigue?”
Regarding healthy lifestyles, we used the introduction, “Here are some changes women sometimes want to make after cancer. Would you like help with …?” For example, we asked, “Would you like help with getting more exercise?” We asked if they wanted help getting more exercise, eating healthier, managing stress, and doing meditation or yoga (Yes/No).
Semistructured interview guide for SSBCS. Participants were asked about their emotional and physical concerns when treatment ended, current cancer needs, symptoms or late effects, and issues related to relationships, family, employment, insurance, financial hardships, barriers to follow-up care, health behaviors, and survivorship program content. Sample questions are, “Have you had any symptoms or side effects related to your cancer or treatment?” and “What kinds of information do you feel you need now about your cancer or treatment?” A brief questionnaire assessed demographics.
Semistructured interview guide for cancer support providers and physicians. Support providers and physicians were asked about informational, psychosocial, and symptom management needs of SSBCS and recommended self-management content and formats. Sample questions are, “What kinds of information and support do you wish was available to help Spanish-speaking women take care of their health after treatment ends?” and “What do you think are the most pressing emotional needs of Spanish-speaking women after breast cancer treatment ends?” A brief questionnaire assessed demographics.
Analysis
Frequencies are reported for survey items. For questions about symptoms/concerns, we report the frequency of responding that they were bothered Somewhat/Quite a bit/A lot. For healthy lifestyles, we present the frequency answering Yes.
Verbatim semistructured interview transcripts were verified against audiotapes. Using QSR NVIVO software, transcripts were coded independently by two bilingual-bicultural investigators using a constant comparative method to generate coding categories for cancer survivorship needs.16 Coders started with themes specified by the interview guides, expanded them to represent the data, and discussed and reconciled coding discrepancies. Coding was compared by type of interview participant (survivor, support provider, or physician). Triangulation of survey and semistructured interviews occurred through team discussions to verify codes, themes, and implications for interventions.
Results
Telephone survey of SSBCS
Of the telephone survey sampling frame (N = 231), 118 individuals (51%) completed the interview, 37 (16%) were ineligible, 31 (13%) could not be reached, 22 (10%) had incorrect contact information, 19 (8%) refused to participate, and 4 (2%) were deceased. Mean age of the participants was 54.9 years (SD, 12.3); all were foreign-born, with more than half of Mexican origin; and most had less than a high-school education (Table 1). All had completed active treatment, and most (68%) were within 2 years of diagnosis.
For symptom management needs (Table 2), the most prevalent (bothersome) symptoms (reported by more than 30%, in rank order) were joint pain, sleep problems, fatigue, hot flashes, numbness/tingling of extremities, and memory. Next most prevalent (reported by 20%-30%) were vaginal dryness, dry/itchy skin, dry nose/mouth, inability to concentrate, constipation, changes in urination, and shortness of breath.

For psychosocial needs, fears of recurrence or new cancers were reported by 42%. Emotional symptoms reported by more than 30% were depression/sadness, anxieties, and feeling stressed. Next most prevalent (20%-30%) were fears, loss of interest in usual activities, and nervousness/restlessness. Social well-being concerns reported by 20%-30% of survivors were loneliness, having no one to talk to, and being a burden to their families. Body image and sexual problems reported by 20%-30% of survivors included appearance and problems being intimate with partners.
Regarding lifestyle, most of the participants said they wanted help with eating a healthier diet (74%), getting more exercise (69%), managing stress (63%), and doing yoga or meditation (55%).
Semistructured interviews
Twenty-five SSBCS completed semistructured interviews, 10 in individual interviews and 15 in one of two focus groups (one of 9 women older than 50 years; one of 6 women younger than 50). The telephone survey respondents were similar to semistructured interview respondents on all sociodemographic characteristics, but differed slightly on some clinical characteristics (Table 1). The telephone survey women had been more recently diagnosed (P < .01), were less likely to have ductal carcinoma in situ (P < .001), and more likely to have had reconstructive surgery
(P < .05).
Five cancer support providers and 4 physicians were interviewed. All support providers were Spanish-speaking Latinas with at least some college education. Cancer physicians were board certified. Two were men; two were white and two Asian; one was a breast surgeon and three were hematologists/oncologists; three spoke Spanish poorly/not at all and one spoke it fairly well.
Seven themes emerged from interviews: unmet physical symptom management needs; social support often ends when treatment ends; challenges resuming roles; sense of abandonment by health care system when treatment ends; need for formal transition from active treatment to follow-up care; fear of recurrence especially when obtaining follow-up care; and desire for information on late effects of initial treatments and side effects of hormonal treatments. We summarize results according to these themes.
Unmet physical symptom management needs. The main physical symptoms reported by survivors and physicians in interviews were arthralgia, menopausal symptoms, and neuropathy. Fatigue was reported only by survivors. Many survivors and several support providers expressed that symptoms were poorly managed and often ignored. One stated,
I have a lot of pain where I had surgery, it burns. I worry a lot about my arm because I have sacs of fluid. My doctor only says, ‘They will dissolve over the years.’ So, I don’t feel any support. (FocGrp1#6)
Survivors reported side effects of hormonal therapies, and felt that physicians downplayed these to prevent them from discontinuing medications.
Social support from family and friends often ends when treatment ends. Many survivors described a loss of support from family and friends who expected them to get back to “normal” once treatment ended. One said,
My sisters have told me to my face that there’s nothing wrong with me. So now when people ask me, I say, ‘I’m fine, thank God, I have nothing,’ even though I’m dying of pain and have all these pills to take. (Survivor#1025)
A support provider related,
The client was telling me that as she was getting closer to finishing her treatment, her husband was upset because he felt like all she was doing was focusing on the cancer. I think caregivers, family, spouses, and children out of their own sort of selfishness want this person to be well. (SuppProv#104)
A few survivors said that family bonds were strengthened after cancer and several reported lacking support because their families were in their home countries.
Challenges resuming roles, especially returning to work. Survivors, support providers, and physicians described challenges and few resources as women transition back to their normal roles. Survivors questioned their ability to return to work due to physically demanding occupations. One stated,
I would like information on how to take care of myself, how working can affect this side if I don’t take care of it. I clean houses and I need both hands. (Survivor#3012)
Survivors described how changes in memory affected daily chores and work performance. Support providers and physicians described the need for resources to aid with return to work and household responsibilities. One physician noted,
There are usually questions about how to go ahead and live their lives from that point forward. It’s a sort of reverse shock: going back to life as they know it. (Physician#004)
Support providers and physicians mentioned that women needed help with resuming intimate partner relations.
Sense of abandonment by health care system once active treatment ends. Survivors, support providers, and physicians reported a loss of support and sense of abandonment by the patients’ oncology team at the end of active treatment. One survivor stated,
Once they tell you to stop the pills, ‘You’re cured, there’s nothing wrong with you,’ the truth is that one feels, ‘Now what do I do? I have no one to help me.’ I felt very abandoned. (FocGrp1#5)
A provider said,
The support system falls apart once women complete treatment. They lose their entire support system at the medical level. They no longer have nurses checking in about symptoms and addressing anything that’s come up. They won’t have access to doctors unless they’re doing their screening. (SuppProv#101)
An oncologist, noting that this loss of support occurs when women face pressures of transitioning back to work or family obligations, commented,
So here’s a woman whose marriage is in turmoil, whose husband may even have left her during this, and now her clinic is leaving her and she’s on her own … that must be scary as hell because there’s nobody out there to support her. (Physician#002)
Need for formal transition from active treatment to follow-up care. Two themes emerged about transitioning from active treatment: transferring care from oncologists to primary care physicians (PCP); and issues of follow-up care (with oncologists or PCPs). Survivors felt lost in transitioning from specialty to primary care, or expressed apprehension seeing a PCP rather than a cancer specialist. One stated,
I have my doctor but she is not a specialist. She does what I tell her to and orders a mammogram every year. But, I don’t go to the oncologist anymore, and so I worry. With the specialists, I feel protected. (FocGrp1#5)
Physicians acknowledged the lack of a formal transition to primary care such as a survivorship care program.
Follow-up care issues were common. Physicians stressed that women needed to know how often to return for follow-up once active treatment ends and about recommended examinations and tests, especially when receiving hormonal therapy. Physicians indicated the need for patient education materials specific to patients’ treatments, for example, elevated risk of heart disease with certain chemotherapy agents. An oncologist expressed concern that PCPs are not prepared adequately about late effects and hormonal treatment side effects, and suggested providing summary notes for PCPs detailing these.
Survivors identified several barriers to follow-up care: lacking information on which symptoms merited a call to physicians; financial burden/limited health insurance; lacking appointment reminders; fear of examinations; and limited English proficiency. A survivor stated,
If you have insurance, you can make your appointment, see the doctor, and have your mammogram. I stopped taking my pills because I didn’t have insurance. I tried to get them again but they told me they would cost me a thousand dollars. (FocGrp1#5)
One oncologist suggested scheduling a follow-up appointment before patients leave treatment and calling patients who miss appointments.
Facilitators of regular follow-up care identified by survivors were physicians informing them about symptom monitoring and reporting, having a clinic contact person/navigator, being given a follow-up appointment, being assertive about one’s care, and physicians’ reinforcement of adherence to hormonal treatment and follow-up. According to support providers, a key facilitator was having a clinic contact person/navigator. Once treatment ended, support providers often served as the liaison between the patient and the physician, making them the first point of contact for symptom reporting.
Fear of recurrence especially when obtaining follow-up care. Fear of recurrence dominated survivor interviews. This fear was heightened at the time of follow-up examinations or when they experienced unusual pain. A survivor commented,
Every time I’m due for my mammogram, I can’t sleep, worrying. I lose sleep until I get the letter with my results. Then I feel at peace again. (FocGrp1#9)
Support providers discussed the need to provide reassurance to SSBCS to help them cope with fears of recurrence. Physicians expressed challenges in allaying fears of recurrence among SSBCS, requiring a lot of time when recommending follow-up mammograms.
Desire for information on late effects of treatments and side effects of hormonal therapies. All survivors expressed receiving insufficient information on potential symptoms and side effects. One stated,
Doctors only have five minutes. There has never been someone who gave me guidance like, ‘From now on you have to do this or you might get these symptoms now or in the future. (Survivor#6019)
They indicated uncertainty about what symptoms were “normal” and when symptoms merited a call to the physician. Several survivors reported being unaware that fatigue, arthralgia and neuropathy were side effects of breast cancer treatments until they reported these to physicians.
Physicians stressed the importance of women knowing about the elevated risk of future cancers, symptoms of recurrence, and seeking follow-up care if they experience symptoms that are out of the ordinary. Support providers felt that it was important to provide SSBCS with information on signs of recurrence and when to report these. However, providers expressed concern that giving women too much information might elevate their anxiety. A physician suggested,
It’s probably better to have a symptom list that’s short and relevant for the most common and catastrophic things, same thing with side effects … short to avoid overwhelming the patient. (Physician#001)
Hormonal treatments were of special concern. Survivors expressed a need for information on hormonal treatments and support providers stressed that this information is needed in simple Spanish. Several survivors indicated they stopped taking hormonal treatments due to side effects. One woman experienced severe headaches and heart palpitations, stopped taking the hormonal medication, felt better, and did not inform her physician until her next appointment. A support provider stated,
What I hear from a lot of women is that if side effects are too uncomfortable, they just stop it (hormonal treatment) without saying anything to the doctor. So more information about why they have to take it and that there is a good chance of recurrence is really important. (SuppProv#101)
Likewise, physicians indicated that SSBCS’ lack of information on hormonal treatments often resulted in nonadherence, emphasizing the need to reinforce adherence to prevent recurrence.
Conceptual framework of interventions
Based on triangulation of survey and interview results, we compiled a conceptual framework that includes needs identified, suggested components of a survivorship care intervention to address these needs, potential mediators by which such interventions could improve outcomes, and relevant outcomes (Figure). Survivorship care needs fell into four categories: symptom management, psychosocial, sense of abandonment by health care team, and healthy lifestyles. Survivorship care programs would provide skills training in symptom and stress management, and communicating with providers, family, friends, and coworkers. Mediators include increased self-efficacy, knowledge and perceived social support, ultimately leading to reduced distress (anxiety and depressive symptoms) and stress, and improved health-related quality of life.
Conclusions
Our study aimed to identify the most critical needs of SSBCS in the posttreatment survivorship phase to facilitate the design of survivorship interventions for this vulnerable group. SSBCS, cancer support providers, and cancer physicians reported substantial symptom management, psychosocial, and informational needs among this population. Results from surveys and open-ended interviews were remarkably consistent. Survivors, physicians, and support providers viewed transition out of active treatment as a time of increased psychosocial need and heightened vulnerability.
Our findings are consistent with needs assessments conducted in other breast cancer survivors. Similar to a study of rural white women with breast cancer, fear of recurrence was among the most common psychosocial concerns.17 Results of two studies that included white, African American and Latina breast cancer survivors were consistent with ours in finding that pain and fatigue were among the most persistent symptoms; in both studies, Latinas were more likely to report pain and a higher number of symptoms.7,18 The prevalence of sleep problems in our sample was identical to that reported in a sample of African American breast cancer survivors.19 Our findings of a high need for symptom management information and support, social support from family and friends, and self-management resources were similar to studies of other vulnerable breast cancer survivors.18,20
Our results suggest that it is critical for health care professionals to provide assistance with managing side effects and information to alleviate fears, and reinforce behaviors of symptom monitoring and reporting, and adherence to follow-up care and hormonal therapies. Yet this information is not being conveyed effectively and is complicated by the need to balance women’s need for information with minimizing anxiety when providing such information.
A limitation of our study is that most of our sample was Mexican origin and may not reflect experiences of Spanish-speaking Latinas of other national origin groups or outside of Northern California. Another limitation is the lack of an English-speaking comparison group, which would have permitted the identification of similarities and differences across language groups. Finally, we did not interview radiation oncologists who may have had opinions that are not represented here.
Survivorship care programs offer great promise for meeting patients’ informational and symptom management needs and improving well-being and communication with clinicians.21 Due to limited access to survivorship care information, financial hardships, and pressures from their families to resume their social roles, concerted efforts are needed to develop appropriate survivorship programs for SSBCS.22 Unique language, cultural and socioeconomic factors of Spanish-speaking Latinas require tailoring of cancer survivorship programs to best meet their needs.23 These programs need to provide psychosocial stress and symptom management assistance, simple information on recommended follow-up care, and healthy lifestyle and role reintegration strategies that account for their unique sociocultural contexts.
1. Danese MD, O’Malley C, Lindquist K, Gleeson M, Griffiths RI. An observational study of the prevalence and incidence of comorbid conditions in older women with breast cancer. Ann Oncol. 2012;23(7):1756-1765.
2. Hewitt M, Greenfield S, Stovall E, eds. From cancer patient to cancer survivor: lost in transition. Washington, DC: National Academy of Sciences; 2006.
3. Beckjord EB, Arora NK, McLaughlin W, Oakley-Girvan I, Hamilton AS, Hesse BW. Health-related information needs in a large and diverse sample of adult cancer survivors: implications for cancer care. J Cancer Surviv. 2008;2(3):179-189.
4. Hewitt ME, Bamundo A, Day R, Harvey C. Perspectives on posttreatment cancer care: qualitative research with survivors, nurses, and physicians. J Clin Oncol. 2007;25(16):2270-2273.
5. Ashing-Giwa KT, Tejero JS, Kim J, Padilla GV, Hellemann G. Examining predictive models of HRQOL in a population-based, multiethnic sample of women with breast carcinoma. Qual Life Res. 2007;16(3):413-428.
6. Clauser SB, Arora NK, Bellizzi KM, Haffer SC, Topor M, Hays RD. Disparities in HRQOL of cancer survivors and non-cancer managed care enrollees. Health Care Financ Rev. 2008;29(4):23-40.
7. Eversley R, Estrin D, Dibble S, Wardlaw L, Pedrosa M, Favila-Penney W. Posttreatment symptoms among ethnic minority breast cancer survivors. Oncol Nurs Forum. 2005;32(2):250-256.
8. Bickell NA, Wang JJ, Oluwole S, et al. Missed opportunities: racial disparities in adjuvant breast cancer treatment. J Clin Oncol. 2006;24(9):1357-1362.
9. Arora NK, Reeve BB, Hays RD, Clauser SB, Oakley-Girvan I. Assessment of quality of cancer-related follow-up care from the cancer survivor’s perspective. J Clin Oncol. 2011;29(10):1280-1289.
10. Janz NK, Mujahid MS, Hawley ST, Griggs JJ, Hamilton AS, Katz SJ. Racial/ethnic differences in adequacy of information and support for women with breast cancer. Cancer. 2008;113(5):1058-1067.
11. Yoon J, Malin JL, Tisnado DM, et al. Symptom management after breast cancer treatment: is it influenced by patient characteristics? Breast Cancer Res Treat. 2008;108(1):69-77.
12. Ashing K, Rosales M. A telephonic-based trial to reduce depressive symptoms among Latina breast cancer survivors. Psychooncology. 2014;23(5):507-515.
13. Hershman DL, Greenlee H, Awad D, et al. Randomized controlled trial of a clinic-based survivorship intervention following adjuvant therapy in breast cancer survivors. Breast Cancer Res Treat. 2013;138(3):795-806.
14. Napoles AM, Ortiz C, Santoyo-Olsson J, et al. Nuevo Amanecer: results of a randomized controlled trial of a community-based, peer-delivered stress management intervention to improve quality of life in Latinas with breast cancer. Am J Public Health. 2015;105(suppl 3):e55-63.
15. Rechis R, Reynolds KA, Beckjord EB, Nutt S, Burns RM, Schaefer JS. ‘I learned to live with it’ is not good enough: challenges reported by posttreatment cancer survivors in the Livestrong surveys. Austin, TX: Livestrong;2011.
16. Glaser BG, Strauss AL. The discovery of grounded theory: strategies for qualitative research. Hawthorne: Aldine Publishing Company; 1967.
17. Befort CA, Klemp J. Sequelae of breast cancer and the influence of menopausal status at diagnosis among rural breast cancer survivors. J Womens Health (Larchmt). 2011;20(9):1307-1313.
18. Fu OS, Crew KD, Jacobson JS, et al. Ethnicity and persistent symptom burden in breast cancer survivors. J Cancer Surviv. 2009;3(4):241-250.
19. Taylor TR, Huntley ED, Makambi K, et al. Understanding sleep disturbances in African-American breast cancer survivors: a pilot study. Psychooncology. 2012;21(8):896-902.
20. Adams N, Gisiger-Camata S, Hardy CM, Thomas TF, Jukkala A, Meneses K. Evaluating survivorship experiences and needs among rural African American breast cancer survivors. J Cancer Educ. October 24, 2015 [Epub ahead of print].
21. Blinder VS, Patil S, Thind A, et al. Return to work in low-income Latina and non-Latina white breast cancer survivors: a 3-year longitudinal study. Cancer. 2012;118(6):1664-1674.
22. Lopez-Class M, Perret-Gentil M, Kreling B, Caicedo L, Mandelblatt J, Graves KD. Quality of life among immigrant Latina breast cancer survivors: realities of culture and enhancing cancer care. J Cancer Educ. 2011;26(4):724-733.
23. Napoles-Springer AM, Ortiz C, O’Brien H, Diaz-Mendez M. Developing a culturally competent peer support intervention for Spanish-speaking Latinas with breast cancer. J Immigr Minor Health. 2009;11(4):268-280
1. Danese MD, O’Malley C, Lindquist K, Gleeson M, Griffiths RI. An observational study of the prevalence and incidence of comorbid conditions in older women with breast cancer. Ann Oncol. 2012;23(7):1756-1765.
2. Hewitt M, Greenfield S, Stovall E, eds. From cancer patient to cancer survivor: lost in transition. Washington, DC: National Academy of Sciences; 2006.
3. Beckjord EB, Arora NK, McLaughlin W, Oakley-Girvan I, Hamilton AS, Hesse BW. Health-related information needs in a large and diverse sample of adult cancer survivors: implications for cancer care. J Cancer Surviv. 2008;2(3):179-189.
4. Hewitt ME, Bamundo A, Day R, Harvey C. Perspectives on posttreatment cancer care: qualitative research with survivors, nurses, and physicians. J Clin Oncol. 2007;25(16):2270-2273.
5. Ashing-Giwa KT, Tejero JS, Kim J, Padilla GV, Hellemann G. Examining predictive models of HRQOL in a population-based, multiethnic sample of women with breast carcinoma. Qual Life Res. 2007;16(3):413-428.
6. Clauser SB, Arora NK, Bellizzi KM, Haffer SC, Topor M, Hays RD. Disparities in HRQOL of cancer survivors and non-cancer managed care enrollees. Health Care Financ Rev. 2008;29(4):23-40.
7. Eversley R, Estrin D, Dibble S, Wardlaw L, Pedrosa M, Favila-Penney W. Posttreatment symptoms among ethnic minority breast cancer survivors. Oncol Nurs Forum. 2005;32(2):250-256.
8. Bickell NA, Wang JJ, Oluwole S, et al. Missed opportunities: racial disparities in adjuvant breast cancer treatment. J Clin Oncol. 2006;24(9):1357-1362.
9. Arora NK, Reeve BB, Hays RD, Clauser SB, Oakley-Girvan I. Assessment of quality of cancer-related follow-up care from the cancer survivor’s perspective. J Clin Oncol. 2011;29(10):1280-1289.
10. Janz NK, Mujahid MS, Hawley ST, Griggs JJ, Hamilton AS, Katz SJ. Racial/ethnic differences in adequacy of information and support for women with breast cancer. Cancer. 2008;113(5):1058-1067.
11. Yoon J, Malin JL, Tisnado DM, et al. Symptom management after breast cancer treatment: is it influenced by patient characteristics? Breast Cancer Res Treat. 2008;108(1):69-77.
12. Ashing K, Rosales M. A telephonic-based trial to reduce depressive symptoms among Latina breast cancer survivors. Psychooncology. 2014;23(5):507-515.
13. Hershman DL, Greenlee H, Awad D, et al. Randomized controlled trial of a clinic-based survivorship intervention following adjuvant therapy in breast cancer survivors. Breast Cancer Res Treat. 2013;138(3):795-806.
14. Napoles AM, Ortiz C, Santoyo-Olsson J, et al. Nuevo Amanecer: results of a randomized controlled trial of a community-based, peer-delivered stress management intervention to improve quality of life in Latinas with breast cancer. Am J Public Health. 2015;105(suppl 3):e55-63.
15. Rechis R, Reynolds KA, Beckjord EB, Nutt S, Burns RM, Schaefer JS. ‘I learned to live with it’ is not good enough: challenges reported by posttreatment cancer survivors in the Livestrong surveys. Austin, TX: Livestrong;2011.
16. Glaser BG, Strauss AL. The discovery of grounded theory: strategies for qualitative research. Hawthorne: Aldine Publishing Company; 1967.
17. Befort CA, Klemp J. Sequelae of breast cancer and the influence of menopausal status at diagnosis among rural breast cancer survivors. J Womens Health (Larchmt). 2011;20(9):1307-1313.
18. Fu OS, Crew KD, Jacobson JS, et al. Ethnicity and persistent symptom burden in breast cancer survivors. J Cancer Surviv. 2009;3(4):241-250.
19. Taylor TR, Huntley ED, Makambi K, et al. Understanding sleep disturbances in African-American breast cancer survivors: a pilot study. Psychooncology. 2012;21(8):896-902.
20. Adams N, Gisiger-Camata S, Hardy CM, Thomas TF, Jukkala A, Meneses K. Evaluating survivorship experiences and needs among rural African American breast cancer survivors. J Cancer Educ. October 24, 2015 [Epub ahead of print].
21. Blinder VS, Patil S, Thind A, et al. Return to work in low-income Latina and non-Latina white breast cancer survivors: a 3-year longitudinal study. Cancer. 2012;118(6):1664-1674.
22. Lopez-Class M, Perret-Gentil M, Kreling B, Caicedo L, Mandelblatt J, Graves KD. Quality of life among immigrant Latina breast cancer survivors: realities of culture and enhancing cancer care. J Cancer Educ. 2011;26(4):724-733.
23. Napoles-Springer AM, Ortiz C, O’Brien H, Diaz-Mendez M. Developing a culturally competent peer support intervention for Spanish-speaking Latinas with breast cancer. J Immigr Minor Health. 2009;11(4):268-280
Miami Breast Cancer Symposium to explore treatment controversies
FROM MBCC (FRONTLINE MEDICAL NEWS) – Oncology Practice will be on site this coming week at the Miami Breast Cancer Symposium in Miami Beach, reporting on the latest in multidisciplinary management of breast cancer patients. Sessions will highlight updates in systemic therapy, immunology and immunotherapy, and overcoming therapeutic resistance, as well as explore treatment controversies, such as omitting anthracyclines, extended-field radiation therapy, and who should be receiving extended hormonal therapy. The symposium, hosted by Physicians’ Education Resource, begins Thursday, March 9. Our team will provide daily coverage of the following presentations and more:
Overcoming Resistance to HER2 Targeted Therapy
Mark D. Pegram, MD
Overcoming Resistance to Endocrine Ablative Therapy
William J. Gradishar, MD
Medical Crossfire: Is Extended Field Radiation Therapy Ready for Prime Time?
Lawrence J. Solin, MD, & Thomas A. Buchholz, MD
Raising the Therapeutic Index for HER2+ Targeted Therapy: Can We Safely Omit
Anthracyclines?
Sara Hurvitz, MD
Update on Neoadjuvant Treatment Strategies in HER2+ Breast Cancer
Debu Tripathy, MD
Update on PARP Inhibitors in Breast Cancer
Kimberly L. Blackwell, MD
Update on Immunology and Immunotherapy in Breast Cancer
Elizabeth Mittendorf, MD, PhD
Medical Crossfire: Is Extended Hormonal Therapy Required for Everyone?
William J. Gradishar, MD, & Sara Hurvitz, MD
FROM MBCC (FRONTLINE MEDICAL NEWS) – Oncology Practice will be on site this coming week at the Miami Breast Cancer Symposium in Miami Beach, reporting on the latest in multidisciplinary management of breast cancer patients. Sessions will highlight updates in systemic therapy, immunology and immunotherapy, and overcoming therapeutic resistance, as well as explore treatment controversies, such as omitting anthracyclines, extended-field radiation therapy, and who should be receiving extended hormonal therapy. The symposium, hosted by Physicians’ Education Resource, begins Thursday, March 9. Our team will provide daily coverage of the following presentations and more:
Overcoming Resistance to HER2 Targeted Therapy
Mark D. Pegram, MD
Overcoming Resistance to Endocrine Ablative Therapy
William J. Gradishar, MD
Medical Crossfire: Is Extended Field Radiation Therapy Ready for Prime Time?
Lawrence J. Solin, MD, & Thomas A. Buchholz, MD
Raising the Therapeutic Index for HER2+ Targeted Therapy: Can We Safely Omit
Anthracyclines?
Sara Hurvitz, MD
Update on Neoadjuvant Treatment Strategies in HER2+ Breast Cancer
Debu Tripathy, MD
Update on PARP Inhibitors in Breast Cancer
Kimberly L. Blackwell, MD
Update on Immunology and Immunotherapy in Breast Cancer
Elizabeth Mittendorf, MD, PhD
Medical Crossfire: Is Extended Hormonal Therapy Required for Everyone?
William J. Gradishar, MD, & Sara Hurvitz, MD
FROM MBCC (FRONTLINE MEDICAL NEWS) – Oncology Practice will be on site this coming week at the Miami Breast Cancer Symposium in Miami Beach, reporting on the latest in multidisciplinary management of breast cancer patients. Sessions will highlight updates in systemic therapy, immunology and immunotherapy, and overcoming therapeutic resistance, as well as explore treatment controversies, such as omitting anthracyclines, extended-field radiation therapy, and who should be receiving extended hormonal therapy. The symposium, hosted by Physicians’ Education Resource, begins Thursday, March 9. Our team will provide daily coverage of the following presentations and more:
Overcoming Resistance to HER2 Targeted Therapy
Mark D. Pegram, MD
Overcoming Resistance to Endocrine Ablative Therapy
William J. Gradishar, MD
Medical Crossfire: Is Extended Field Radiation Therapy Ready for Prime Time?
Lawrence J. Solin, MD, & Thomas A. Buchholz, MD
Raising the Therapeutic Index for HER2+ Targeted Therapy: Can We Safely Omit
Anthracyclines?
Sara Hurvitz, MD
Update on Neoadjuvant Treatment Strategies in HER2+ Breast Cancer
Debu Tripathy, MD
Update on PARP Inhibitors in Breast Cancer
Kimberly L. Blackwell, MD
Update on Immunology and Immunotherapy in Breast Cancer
Elizabeth Mittendorf, MD, PhD
Medical Crossfire: Is Extended Hormonal Therapy Required for Everyone?
William J. Gradishar, MD, & Sara Hurvitz, MD
Breast cancer mortality mapped for 2017
U.S. breast cancer mortality will be an estimated 25.3 per 100,000 females in 2017, with the highest state rates in the East and the lowest in the West and Midwest.
Approximately 40,600 breast cancer deaths are predicted for the year in the United States by the American Cancer Society in its Cancer Facts & Figures 2017, based on 2000-2014 data from the National Center for Health Statistics. With the U.S. population currently around 321 million, that works out to a completely unadjusted death rate of 25.3 per 100,000 women. Doing a little more math puts the highest death rate (29.8) in West Virginia and the lowest (18.8) in Utah.
The incidence rate for female breast cancer was 123.3 per 100,000 for 2009-2013, with that figure age adjusted to the 2000 U.S. standard population, according to data from the North American Association of Central Cancer Registries, the ACS reported.
Wyoming had the lowest incidence over that time period (109.6 per 100,000), and New Hampshire had the highest at 138.1. Utah had the fifth-lowest incidence in that period at 112.7, but West Virginia, the state with the highest estimated mortality for 2017, had a relatively low incidence of 114.4 in 2009-2013, the ACS said.
U.S. breast cancer mortality will be an estimated 25.3 per 100,000 females in 2017, with the highest state rates in the East and the lowest in the West and Midwest.
Approximately 40,600 breast cancer deaths are predicted for the year in the United States by the American Cancer Society in its Cancer Facts & Figures 2017, based on 2000-2014 data from the National Center for Health Statistics. With the U.S. population currently around 321 million, that works out to a completely unadjusted death rate of 25.3 per 100,000 women. Doing a little more math puts the highest death rate (29.8) in West Virginia and the lowest (18.8) in Utah.
The incidence rate for female breast cancer was 123.3 per 100,000 for 2009-2013, with that figure age adjusted to the 2000 U.S. standard population, according to data from the North American Association of Central Cancer Registries, the ACS reported.
Wyoming had the lowest incidence over that time period (109.6 per 100,000), and New Hampshire had the highest at 138.1. Utah had the fifth-lowest incidence in that period at 112.7, but West Virginia, the state with the highest estimated mortality for 2017, had a relatively low incidence of 114.4 in 2009-2013, the ACS said.
U.S. breast cancer mortality will be an estimated 25.3 per 100,000 females in 2017, with the highest state rates in the East and the lowest in the West and Midwest.
Approximately 40,600 breast cancer deaths are predicted for the year in the United States by the American Cancer Society in its Cancer Facts & Figures 2017, based on 2000-2014 data from the National Center for Health Statistics. With the U.S. population currently around 321 million, that works out to a completely unadjusted death rate of 25.3 per 100,000 women. Doing a little more math puts the highest death rate (29.8) in West Virginia and the lowest (18.8) in Utah.
The incidence rate for female breast cancer was 123.3 per 100,000 for 2009-2013, with that figure age adjusted to the 2000 U.S. standard population, according to data from the North American Association of Central Cancer Registries, the ACS reported.
Wyoming had the lowest incidence over that time period (109.6 per 100,000), and New Hampshire had the highest at 138.1. Utah had the fifth-lowest incidence in that period at 112.7, but West Virginia, the state with the highest estimated mortality for 2017, had a relatively low incidence of 114.4 in 2009-2013, the ACS said.
‘Strong evidence’ links obesity to cancers
There is strong evidence linking adiposity to esophageal adenocarcinoma, multiple myeloma, and cancer of the colon, rectum, biliary tract, pancreas, endometrium, kidney, and postmenopausal breast, according to the authors of an umbrella review published in the Feb. 28 edition of the BMJ.
“Several meta-analyses support the link between obesity and cancer, but substantial heterogeneity exists between studies,” wrote Maria Kyrgiou, MD, of Imperial College London and her coauthors. “The reported associations may be causal, but they may also be flawed, as inherent study biases such as residual confounding and selective reporting of positive results may exaggerate the effect of obesity on cancer.”
In this umbrella review, researchers analyzed 49 papers that included a total of 204 meta-analyses, which in turn summarized 2,179 individual study estimates from 507 unique cohort or case-control studies.
When researchers applied a threshold for significance of P less than .000001, the summary random effects were significant in 35 meta-analyses; 31 of these found increased risk with adiposity of esophageal adenocarcinoma, multiple myeloma, and cancers of the colon, rectum, liver, biliary tract system (cancers of gallbladder, extrahepatic bile duct, and ampulla of Vater), pancreas, postmenopausal breast, endometrium, and kidney.
“The effect of obesity on the incidence and mortality of cancer is well recognized and was evident in our umbrella review, with approximately 77% of the included meta-analyses reporting a nominally statistically significant summary random effects estimate,” the authors reported.
Overall, the summary estimates were similar between men and women for esophageal adenocarcinoma, esophageal squamous cell carcinoma, multiple myeloma, leukemia, and gastric, lung, kidney, and thyroid cancers.
However, men had a 30% higher risk of colon cancer per 5-kg/m2 increase of body mass index, compared with a 9% increase in risk in women for the same rise in BMI. Men also showed an increased risk of melanoma with increasing BMI, whereas women did not.
Women who had never used hormone therapy showed an 11% increase in the risk of postmenopausal breast cancer with each 5 kg of weight gained. Similarly, each 0.1 increase in waist-to-hip ratio in these women was associated with a 21% increase in the risk of endometrial cancer.
The analysis also revealed an inverse relationship in four meta-analyses for esophageal squamous cell carcinoma and lung cancer.
The authors said their findings agree with those of the World Cancer Research Fund, which currently states there is a convincing causal relationship with obesity for esophageal adenocarcinoma and cancers of the pancreas, colorectum, postmenopausal breast, endometrium, kidney, and liver.
While this umbrella analysis did not find strong evidence for an association with liver cancer, the authors said the evidence was “highly suggestive” but suffered from small study effects, excess significance bias, and substantial heterogeneity between studies.
“To draw firmer conclusions, we need prospective studies and large consortiums with better assessment of the changing nature of body fatness and with comprehensive standardized reporting of analyses,” they wrote. “As obesity becomes one of the greatest public health problems worldwide, evidence of the strength of the associations between obesity and cancer may allow finer selection of people at high risk, who could be selected for personalized primary and secondary prevention strategies.”
The study was supported by the Genesis Research Trust, Sigrid Jusélius Fellowship, the World Cancer Research Fund International Regular Grant Programme, Ovarian Cancer Action, the Imperial Experimental Cancer Medicine Centre, the Cancer Research UK Imperial Centre, Imperial Healthcare NHS Trust NIHR BRC. No relevant conflicts of interest were declared.
There is strong evidence linking adiposity to esophageal adenocarcinoma, multiple myeloma, and cancer of the colon, rectum, biliary tract, pancreas, endometrium, kidney, and postmenopausal breast, according to the authors of an umbrella review published in the Feb. 28 edition of the BMJ.
“Several meta-analyses support the link between obesity and cancer, but substantial heterogeneity exists between studies,” wrote Maria Kyrgiou, MD, of Imperial College London and her coauthors. “The reported associations may be causal, but they may also be flawed, as inherent study biases such as residual confounding and selective reporting of positive results may exaggerate the effect of obesity on cancer.”
In this umbrella review, researchers analyzed 49 papers that included a total of 204 meta-analyses, which in turn summarized 2,179 individual study estimates from 507 unique cohort or case-control studies.
When researchers applied a threshold for significance of P less than .000001, the summary random effects were significant in 35 meta-analyses; 31 of these found increased risk with adiposity of esophageal adenocarcinoma, multiple myeloma, and cancers of the colon, rectum, liver, biliary tract system (cancers of gallbladder, extrahepatic bile duct, and ampulla of Vater), pancreas, postmenopausal breast, endometrium, and kidney.
“The effect of obesity on the incidence and mortality of cancer is well recognized and was evident in our umbrella review, with approximately 77% of the included meta-analyses reporting a nominally statistically significant summary random effects estimate,” the authors reported.
Overall, the summary estimates were similar between men and women for esophageal adenocarcinoma, esophageal squamous cell carcinoma, multiple myeloma, leukemia, and gastric, lung, kidney, and thyroid cancers.
However, men had a 30% higher risk of colon cancer per 5-kg/m2 increase of body mass index, compared with a 9% increase in risk in women for the same rise in BMI. Men also showed an increased risk of melanoma with increasing BMI, whereas women did not.
Women who had never used hormone therapy showed an 11% increase in the risk of postmenopausal breast cancer with each 5 kg of weight gained. Similarly, each 0.1 increase in waist-to-hip ratio in these women was associated with a 21% increase in the risk of endometrial cancer.
The analysis also revealed an inverse relationship in four meta-analyses for esophageal squamous cell carcinoma and lung cancer.
The authors said their findings agree with those of the World Cancer Research Fund, which currently states there is a convincing causal relationship with obesity for esophageal adenocarcinoma and cancers of the pancreas, colorectum, postmenopausal breast, endometrium, kidney, and liver.
While this umbrella analysis did not find strong evidence for an association with liver cancer, the authors said the evidence was “highly suggestive” but suffered from small study effects, excess significance bias, and substantial heterogeneity between studies.
“To draw firmer conclusions, we need prospective studies and large consortiums with better assessment of the changing nature of body fatness and with comprehensive standardized reporting of analyses,” they wrote. “As obesity becomes one of the greatest public health problems worldwide, evidence of the strength of the associations between obesity and cancer may allow finer selection of people at high risk, who could be selected for personalized primary and secondary prevention strategies.”
The study was supported by the Genesis Research Trust, Sigrid Jusélius Fellowship, the World Cancer Research Fund International Regular Grant Programme, Ovarian Cancer Action, the Imperial Experimental Cancer Medicine Centre, the Cancer Research UK Imperial Centre, Imperial Healthcare NHS Trust NIHR BRC. No relevant conflicts of interest were declared.
There is strong evidence linking adiposity to esophageal adenocarcinoma, multiple myeloma, and cancer of the colon, rectum, biliary tract, pancreas, endometrium, kidney, and postmenopausal breast, according to the authors of an umbrella review published in the Feb. 28 edition of the BMJ.
“Several meta-analyses support the link between obesity and cancer, but substantial heterogeneity exists between studies,” wrote Maria Kyrgiou, MD, of Imperial College London and her coauthors. “The reported associations may be causal, but they may also be flawed, as inherent study biases such as residual confounding and selective reporting of positive results may exaggerate the effect of obesity on cancer.”
In this umbrella review, researchers analyzed 49 papers that included a total of 204 meta-analyses, which in turn summarized 2,179 individual study estimates from 507 unique cohort or case-control studies.
When researchers applied a threshold for significance of P less than .000001, the summary random effects were significant in 35 meta-analyses; 31 of these found increased risk with adiposity of esophageal adenocarcinoma, multiple myeloma, and cancers of the colon, rectum, liver, biliary tract system (cancers of gallbladder, extrahepatic bile duct, and ampulla of Vater), pancreas, postmenopausal breast, endometrium, and kidney.
“The effect of obesity on the incidence and mortality of cancer is well recognized and was evident in our umbrella review, with approximately 77% of the included meta-analyses reporting a nominally statistically significant summary random effects estimate,” the authors reported.
Overall, the summary estimates were similar between men and women for esophageal adenocarcinoma, esophageal squamous cell carcinoma, multiple myeloma, leukemia, and gastric, lung, kidney, and thyroid cancers.
However, men had a 30% higher risk of colon cancer per 5-kg/m2 increase of body mass index, compared with a 9% increase in risk in women for the same rise in BMI. Men also showed an increased risk of melanoma with increasing BMI, whereas women did not.
Women who had never used hormone therapy showed an 11% increase in the risk of postmenopausal breast cancer with each 5 kg of weight gained. Similarly, each 0.1 increase in waist-to-hip ratio in these women was associated with a 21% increase in the risk of endometrial cancer.
The analysis also revealed an inverse relationship in four meta-analyses for esophageal squamous cell carcinoma and lung cancer.
The authors said their findings agree with those of the World Cancer Research Fund, which currently states there is a convincing causal relationship with obesity for esophageal adenocarcinoma and cancers of the pancreas, colorectum, postmenopausal breast, endometrium, kidney, and liver.
While this umbrella analysis did not find strong evidence for an association with liver cancer, the authors said the evidence was “highly suggestive” but suffered from small study effects, excess significance bias, and substantial heterogeneity between studies.
“To draw firmer conclusions, we need prospective studies and large consortiums with better assessment of the changing nature of body fatness and with comprehensive standardized reporting of analyses,” they wrote. “As obesity becomes one of the greatest public health problems worldwide, evidence of the strength of the associations between obesity and cancer may allow finer selection of people at high risk, who could be selected for personalized primary and secondary prevention strategies.”
The study was supported by the Genesis Research Trust, Sigrid Jusélius Fellowship, the World Cancer Research Fund International Regular Grant Programme, Ovarian Cancer Action, the Imperial Experimental Cancer Medicine Centre, the Cancer Research UK Imperial Centre, Imperial Healthcare NHS Trust NIHR BRC. No relevant conflicts of interest were declared.
FROM BMJ
Key clinical point: An umbrella analysis of systematic reviews has found strong evidence linking adiposity to a range of cancers including esophageal adenocarcinoma, and cancer of the colon, kidney, and pancreas.
Major finding: Adiposity is significantly associated with cancers of the esophagus, colon, rectum, biliary tract, pancreas, endometrium, kidney, postmenopausal breast, and to multiple myeloma.
Data source: An umbrella review of 204 meta-analyses.
Disclosures: The study was supported by the Genesis Research Trust, Sigrid Jusélius Fellowship, the World Cancer Research Fund International Regular Grant Programme, Ovarian Cancer Action, the Imperial Experimental Cancer Medicine Centre, the Cancer Research UK Imperial Centre, Imperial Healthcare NHS Trust NIHR BRC. No relevant conflicts of interest were declared.
More than one-third of tumors found on breast cancer screening represent overdiagnosis
The purpose of screening mammography is to detect tumors when they are small and nonpalpable in order to prevent more advanced breast tumors in women. Overdiagnosis, which leads to unnecessary treatment, refers to screen-detected tumors that will not lead to symptoms. Overdiagnosis cannot be measured directly and, therefore, understanding this concept is problematic for both women and clinicians.
Related article:
Women’s Preventive Services Initiative Guidelines provide consensus for practicing ObGyns
Observations from other types of cancer screening put overdiagnosis in perspective
To help us grasp the overall issue of overdiagnosis, we can consider screening mammography alongside cervical cancer screening and colon cancer screening. For instance, screening with cervical cytology has reduced the incidence of and mortality from invasive cervical cancer.1 Likewise, colonoscopy repeatedly has been found to reduce colon cancer mortality.2,3 Decades of media messaging have emphasized the benefits of screening mammograms.4 However, and in contrast with cervical cytology and colonoscopy, screening mammography has not reduced the incidence of breast cancer presenting with metastatic (advanced) disease.5 Likewise, as the Danish authors of a recent study published in Annals of Internal Medicine point out, screening mammography has not achieved the promised reduction in breast cancer mortality.
New data from Denmark highlight overdiagnosis concerns
Jørgensen and colleagues conducted a cohort study to estimate the incidence of screen-detected tumors that would not become clinically relevant (overdiagnosis) among women aged 35 to 84 years between 1980 and 2010 in Denmark.6 This country offers a particularly well-suited backdrop for a study of overdiagnosis because biennial screening mammography was introduced by region beginning in the early 1990s. By 2007, one-fifth of the country’s female population aged 50 to 69 years were invited to participate. In the following years, screening became universal for Danish women in this age group.
For the study, researchers identified the size of all invasive breast cancer tumors diagnosed over the study period and then compared the incidence rates of advanced tumors (more than 20-mm in size at detection) with nonadvanced tumors in screened and unscreened Danish regions. The investigators took into account regional differences not related to screening by assessing the trends in diagnosis of advanced and nonadvanced tumors in screened and unscreened regions among women older and younger than those screened. This gave them a better estimate of the incidence of overdiagnosis.6
Jørgensen and colleagues found that breast cancer screening resulted in an increase in the incidence of nonadvanced tumors, but that it did not reduce the incidence of advanced tumors. They estimated that 39% of the invasive tumors found among women aged 50 to 69 were overdiagnosed.6
These Danish study results, that more than one-third of screen-detected tumors represent overdiagnosis, are similar to those found for studies conducted in the United States and other countries.7,8 The lengthy follow-up after initiation of screening and the assessment of trends in unscreened women represent strengths of the study by Jørgensen and colleagues, and speak to concerns voiced by those skeptical of reported overdiagnosis incidence rates.9
Although breast cancer mortality is declining, the lion’s share of this decline has resulted from improvements in systemic therapy rather than from screening mammography. Widespread screening mammography has resulted in a scenario in which women are more likely to have a breast cancer that was overdiagnosed than in having earlier detection of a tumor destined to grow larger.5 In the future, by targeting higher-risk women, screening may result in a better benefit:risk ratio. However, and as pointed out by Otis Brawley, MD, Chief Medical and Scientific Officer of the American Cancer Society, we must acknowledge that overdiagnosis is common, the benefits of screening have been overstated, and some patients considered as “cured” from breast cancer have in fact been harmed by unneeded treatment.10
Related article:
No surprises from the USPSTF with new guidance on screening mammography
My breast cancer screening approach
As Brawley indicates, we should not abandon screening.10 I continue to recommend screening based on US Preventive Services Taskforce guidance, beginning biennial screens at age 50.11 I also recognize that some women prefer earlier and more frequent screens, while others may prefer less frequent or even no screening.
- Nieminen P, Kallio M, Hakama M. The effect of mass screening on incidence and mortality of squamous and adenocarcinoma of cervix uteri. Obstet Gynecol. 1995;85(6):1017-1021.
- Baxter NN, Goldwasser MA, Paszat LF, Saskin R, Urbach DR, Rabeneck L. Association of colonoscopy and death from colorectal cancer. Ann Intern Med. 2009;150(1):1-8.
- Singh H, Nugent Z, Demers AA, Kliewer EV, Mahmud SM, Bernstein CN. The reduction in colorectal cancer mortality after colonoscopy varies by site of the cancer. Gastroenterology. 2010;139(4):1128-1137.
- Orenstein P. Our feel-good war on breast cancer. New York Times website. http://www.nytimes.com/2013/04/28/magazine/our-feel-good-war-on-breast-cancer.html?pagewanted=all& _r=0. Published April 25, 2013. Accessed February 21, 2017.
- Welch HG, Gorski DH, Albertsen PC. Trends in metastatic breast and prostate cancer. N Engl J Med. 2016;374(8):596.
- Jørgensen KJ, Gøtzsche PC, Kalager M, Zahl PH. Breast cancer screening in Denmark: a cohort study of tumor size and overdiagnosis. Ann Intern Med. 2017 Jan 10. doi:10.7326/M16-0270.
- Welch HG, Prorok PC, O'Malley AJ, Kramer BS. Breast-cancer tumor size, overdiagnosis, and mammography screening effectiveness. N Engl J Med. 2016;375(15):1438-1447.
- Autier P, Boniol M, Middleton R, et al. Advanced breast cancer incidence following population-based mammographic screening. Ann Oncol. 2011;22(8):1726-1735.
- Kopans DB. Breast-cancer tumor size and screening effectiveness. N Engl J Med. 2017;376(1):93-94.
- Brawley OW. Accepting the existence of breast cancer overdiagnosis [published online ahead of print January 10, 2017]. Ann Intern Med. doi:10.7326/M16-2850.
- Nelson HD, Tyne K, Naik A, Bougatsos C, Chan BK, Humphrey L. Screening for breast cancer: an update for the U.S. Preventive Services Task Force. Ann Intern Med. 2009;151(10):727-737.
The purpose of screening mammography is to detect tumors when they are small and nonpalpable in order to prevent more advanced breast tumors in women. Overdiagnosis, which leads to unnecessary treatment, refers to screen-detected tumors that will not lead to symptoms. Overdiagnosis cannot be measured directly and, therefore, understanding this concept is problematic for both women and clinicians.
Related article:
Women’s Preventive Services Initiative Guidelines provide consensus for practicing ObGyns
Observations from other types of cancer screening put overdiagnosis in perspective
To help us grasp the overall issue of overdiagnosis, we can consider screening mammography alongside cervical cancer screening and colon cancer screening. For instance, screening with cervical cytology has reduced the incidence of and mortality from invasive cervical cancer.1 Likewise, colonoscopy repeatedly has been found to reduce colon cancer mortality.2,3 Decades of media messaging have emphasized the benefits of screening mammograms.4 However, and in contrast with cervical cytology and colonoscopy, screening mammography has not reduced the incidence of breast cancer presenting with metastatic (advanced) disease.5 Likewise, as the Danish authors of a recent study published in Annals of Internal Medicine point out, screening mammography has not achieved the promised reduction in breast cancer mortality.
New data from Denmark highlight overdiagnosis concerns
Jørgensen and colleagues conducted a cohort study to estimate the incidence of screen-detected tumors that would not become clinically relevant (overdiagnosis) among women aged 35 to 84 years between 1980 and 2010 in Denmark.6 This country offers a particularly well-suited backdrop for a study of overdiagnosis because biennial screening mammography was introduced by region beginning in the early 1990s. By 2007, one-fifth of the country’s female population aged 50 to 69 years were invited to participate. In the following years, screening became universal for Danish women in this age group.
For the study, researchers identified the size of all invasive breast cancer tumors diagnosed over the study period and then compared the incidence rates of advanced tumors (more than 20-mm in size at detection) with nonadvanced tumors in screened and unscreened Danish regions. The investigators took into account regional differences not related to screening by assessing the trends in diagnosis of advanced and nonadvanced tumors in screened and unscreened regions among women older and younger than those screened. This gave them a better estimate of the incidence of overdiagnosis.6
Jørgensen and colleagues found that breast cancer screening resulted in an increase in the incidence of nonadvanced tumors, but that it did not reduce the incidence of advanced tumors. They estimated that 39% of the invasive tumors found among women aged 50 to 69 were overdiagnosed.6
These Danish study results, that more than one-third of screen-detected tumors represent overdiagnosis, are similar to those found for studies conducted in the United States and other countries.7,8 The lengthy follow-up after initiation of screening and the assessment of trends in unscreened women represent strengths of the study by Jørgensen and colleagues, and speak to concerns voiced by those skeptical of reported overdiagnosis incidence rates.9
Although breast cancer mortality is declining, the lion’s share of this decline has resulted from improvements in systemic therapy rather than from screening mammography. Widespread screening mammography has resulted in a scenario in which women are more likely to have a breast cancer that was overdiagnosed than in having earlier detection of a tumor destined to grow larger.5 In the future, by targeting higher-risk women, screening may result in a better benefit:risk ratio. However, and as pointed out by Otis Brawley, MD, Chief Medical and Scientific Officer of the American Cancer Society, we must acknowledge that overdiagnosis is common, the benefits of screening have been overstated, and some patients considered as “cured” from breast cancer have in fact been harmed by unneeded treatment.10
Related article:
No surprises from the USPSTF with new guidance on screening mammography
My breast cancer screening approach
As Brawley indicates, we should not abandon screening.10 I continue to recommend screening based on US Preventive Services Taskforce guidance, beginning biennial screens at age 50.11 I also recognize that some women prefer earlier and more frequent screens, while others may prefer less frequent or even no screening.
The purpose of screening mammography is to detect tumors when they are small and nonpalpable in order to prevent more advanced breast tumors in women. Overdiagnosis, which leads to unnecessary treatment, refers to screen-detected tumors that will not lead to symptoms. Overdiagnosis cannot be measured directly and, therefore, understanding this concept is problematic for both women and clinicians.
Related article:
Women’s Preventive Services Initiative Guidelines provide consensus for practicing ObGyns
Observations from other types of cancer screening put overdiagnosis in perspective
To help us grasp the overall issue of overdiagnosis, we can consider screening mammography alongside cervical cancer screening and colon cancer screening. For instance, screening with cervical cytology has reduced the incidence of and mortality from invasive cervical cancer.1 Likewise, colonoscopy repeatedly has been found to reduce colon cancer mortality.2,3 Decades of media messaging have emphasized the benefits of screening mammograms.4 However, and in contrast with cervical cytology and colonoscopy, screening mammography has not reduced the incidence of breast cancer presenting with metastatic (advanced) disease.5 Likewise, as the Danish authors of a recent study published in Annals of Internal Medicine point out, screening mammography has not achieved the promised reduction in breast cancer mortality.
New data from Denmark highlight overdiagnosis concerns
Jørgensen and colleagues conducted a cohort study to estimate the incidence of screen-detected tumors that would not become clinically relevant (overdiagnosis) among women aged 35 to 84 years between 1980 and 2010 in Denmark.6 This country offers a particularly well-suited backdrop for a study of overdiagnosis because biennial screening mammography was introduced by region beginning in the early 1990s. By 2007, one-fifth of the country’s female population aged 50 to 69 years were invited to participate. In the following years, screening became universal for Danish women in this age group.
For the study, researchers identified the size of all invasive breast cancer tumors diagnosed over the study period and then compared the incidence rates of advanced tumors (more than 20-mm in size at detection) with nonadvanced tumors in screened and unscreened Danish regions. The investigators took into account regional differences not related to screening by assessing the trends in diagnosis of advanced and nonadvanced tumors in screened and unscreened regions among women older and younger than those screened. This gave them a better estimate of the incidence of overdiagnosis.6
Jørgensen and colleagues found that breast cancer screening resulted in an increase in the incidence of nonadvanced tumors, but that it did not reduce the incidence of advanced tumors. They estimated that 39% of the invasive tumors found among women aged 50 to 69 were overdiagnosed.6
These Danish study results, that more than one-third of screen-detected tumors represent overdiagnosis, are similar to those found for studies conducted in the United States and other countries.7,8 The lengthy follow-up after initiation of screening and the assessment of trends in unscreened women represent strengths of the study by Jørgensen and colleagues, and speak to concerns voiced by those skeptical of reported overdiagnosis incidence rates.9
Although breast cancer mortality is declining, the lion’s share of this decline has resulted from improvements in systemic therapy rather than from screening mammography. Widespread screening mammography has resulted in a scenario in which women are more likely to have a breast cancer that was overdiagnosed than in having earlier detection of a tumor destined to grow larger.5 In the future, by targeting higher-risk women, screening may result in a better benefit:risk ratio. However, and as pointed out by Otis Brawley, MD, Chief Medical and Scientific Officer of the American Cancer Society, we must acknowledge that overdiagnosis is common, the benefits of screening have been overstated, and some patients considered as “cured” from breast cancer have in fact been harmed by unneeded treatment.10
Related article:
No surprises from the USPSTF with new guidance on screening mammography
My breast cancer screening approach
As Brawley indicates, we should not abandon screening.10 I continue to recommend screening based on US Preventive Services Taskforce guidance, beginning biennial screens at age 50.11 I also recognize that some women prefer earlier and more frequent screens, while others may prefer less frequent or even no screening.
- Nieminen P, Kallio M, Hakama M. The effect of mass screening on incidence and mortality of squamous and adenocarcinoma of cervix uteri. Obstet Gynecol. 1995;85(6):1017-1021.
- Baxter NN, Goldwasser MA, Paszat LF, Saskin R, Urbach DR, Rabeneck L. Association of colonoscopy and death from colorectal cancer. Ann Intern Med. 2009;150(1):1-8.
- Singh H, Nugent Z, Demers AA, Kliewer EV, Mahmud SM, Bernstein CN. The reduction in colorectal cancer mortality after colonoscopy varies by site of the cancer. Gastroenterology. 2010;139(4):1128-1137.
- Orenstein P. Our feel-good war on breast cancer. New York Times website. http://www.nytimes.com/2013/04/28/magazine/our-feel-good-war-on-breast-cancer.html?pagewanted=all& _r=0. Published April 25, 2013. Accessed February 21, 2017.
- Welch HG, Gorski DH, Albertsen PC. Trends in metastatic breast and prostate cancer. N Engl J Med. 2016;374(8):596.
- Jørgensen KJ, Gøtzsche PC, Kalager M, Zahl PH. Breast cancer screening in Denmark: a cohort study of tumor size and overdiagnosis. Ann Intern Med. 2017 Jan 10. doi:10.7326/M16-0270.
- Welch HG, Prorok PC, O'Malley AJ, Kramer BS. Breast-cancer tumor size, overdiagnosis, and mammography screening effectiveness. N Engl J Med. 2016;375(15):1438-1447.
- Autier P, Boniol M, Middleton R, et al. Advanced breast cancer incidence following population-based mammographic screening. Ann Oncol. 2011;22(8):1726-1735.
- Kopans DB. Breast-cancer tumor size and screening effectiveness. N Engl J Med. 2017;376(1):93-94.
- Brawley OW. Accepting the existence of breast cancer overdiagnosis [published online ahead of print January 10, 2017]. Ann Intern Med. doi:10.7326/M16-2850.
- Nelson HD, Tyne K, Naik A, Bougatsos C, Chan BK, Humphrey L. Screening for breast cancer: an update for the U.S. Preventive Services Task Force. Ann Intern Med. 2009;151(10):727-737.
- Nieminen P, Kallio M, Hakama M. The effect of mass screening on incidence and mortality of squamous and adenocarcinoma of cervix uteri. Obstet Gynecol. 1995;85(6):1017-1021.
- Baxter NN, Goldwasser MA, Paszat LF, Saskin R, Urbach DR, Rabeneck L. Association of colonoscopy and death from colorectal cancer. Ann Intern Med. 2009;150(1):1-8.
- Singh H, Nugent Z, Demers AA, Kliewer EV, Mahmud SM, Bernstein CN. The reduction in colorectal cancer mortality after colonoscopy varies by site of the cancer. Gastroenterology. 2010;139(4):1128-1137.
- Orenstein P. Our feel-good war on breast cancer. New York Times website. http://www.nytimes.com/2013/04/28/magazine/our-feel-good-war-on-breast-cancer.html?pagewanted=all& _r=0. Published April 25, 2013. Accessed February 21, 2017.
- Welch HG, Gorski DH, Albertsen PC. Trends in metastatic breast and prostate cancer. N Engl J Med. 2016;374(8):596.
- Jørgensen KJ, Gøtzsche PC, Kalager M, Zahl PH. Breast cancer screening in Denmark: a cohort study of tumor size and overdiagnosis. Ann Intern Med. 2017 Jan 10. doi:10.7326/M16-0270.
- Welch HG, Prorok PC, O'Malley AJ, Kramer BS. Breast-cancer tumor size, overdiagnosis, and mammography screening effectiveness. N Engl J Med. 2016;375(15):1438-1447.
- Autier P, Boniol M, Middleton R, et al. Advanced breast cancer incidence following population-based mammographic screening. Ann Oncol. 2011;22(8):1726-1735.
- Kopans DB. Breast-cancer tumor size and screening effectiveness. N Engl J Med. 2017;376(1):93-94.
- Brawley OW. Accepting the existence of breast cancer overdiagnosis [published online ahead of print January 10, 2017]. Ann Intern Med. doi:10.7326/M16-2850.
- Nelson HD, Tyne K, Naik A, Bougatsos C, Chan BK, Humphrey L. Screening for breast cancer: an update for the U.S. Preventive Services Task Force. Ann Intern Med. 2009;151(10):727-737.
Oral contraceptive use confers long-term cancer protection
New findings from a cohort study with more than 4 decades of follow-up show that, while women who have ever used combined oral contraceptives see an increased risk of breast and cervical cancer, the risk disappears within about 5 years after stopping, but a protective effect against colorectal, endometrial, and ovarian cancer persists for more than 30 years.
The findings provide an update to the General Practitioners’ Oral Contraception Study of a United Kingdom cohort recruited in the late 1960s.
The mean age was 70.2 years, most were white, and the mean follow-up was 40.7 years. Women who had used the pill did so a mean 3.66 years and used older, higher-estrogen formulations.
Compared with never users, users of oral contraception had a nonsignificant 4% reduced risk of any cancer. The incidence rate ratio for breast cancer was similar between ever users and nonusers (IRR 1.04; 99% CI, 0.91-1.17). Women who had used OCs saw significant reductions in colorectal (IRR, 0.81; 99% CI, 0.66-0.99), endometrial (IRR, 0.66; 99% CI, 0.48-0.89), ovarian (IRR, 0.67; 99% CI, 0.50-0.89), and lymphatic and hematopoietic cancers (IRR, 0.74; 0.58-0.94), compared with never users.
Lung cancer incidence was increased among ever users of OCs, but only in women who smoked at the time of recruitment.
“There was no evidence of new cancer risks appearing later in life among women who had used oral contraceptives,” the researchers wrote. “Thus, the overall balance of cancer risk among past users of oral contraceptives was neutral with the increased risks counterbalanced by the endometrial, ovarian, and colorectal cancer benefits that persist at least 30 years.”
The results, the researchers wrote, “provide strong evidence that most women do not expose themselves to long-term cancer harm if they choose to use oral contraception, indeed many are likely to be protected.”
The study was funded by the Royal College of General Practitioners, Medical Research Council, Imperial Cancer Research Fund, British Heart Foundation, Schering AG, Schering Health Care, Wyeth Ayerst International, Ortho Cilag, and Searle. The researchers reported having no conflicts of interest.
New findings from a cohort study with more than 4 decades of follow-up show that, while women who have ever used combined oral contraceptives see an increased risk of breast and cervical cancer, the risk disappears within about 5 years after stopping, but a protective effect against colorectal, endometrial, and ovarian cancer persists for more than 30 years.
The findings provide an update to the General Practitioners’ Oral Contraception Study of a United Kingdom cohort recruited in the late 1960s.
The mean age was 70.2 years, most were white, and the mean follow-up was 40.7 years. Women who had used the pill did so a mean 3.66 years and used older, higher-estrogen formulations.
Compared with never users, users of oral contraception had a nonsignificant 4% reduced risk of any cancer. The incidence rate ratio for breast cancer was similar between ever users and nonusers (IRR 1.04; 99% CI, 0.91-1.17). Women who had used OCs saw significant reductions in colorectal (IRR, 0.81; 99% CI, 0.66-0.99), endometrial (IRR, 0.66; 99% CI, 0.48-0.89), ovarian (IRR, 0.67; 99% CI, 0.50-0.89), and lymphatic and hematopoietic cancers (IRR, 0.74; 0.58-0.94), compared with never users.
Lung cancer incidence was increased among ever users of OCs, but only in women who smoked at the time of recruitment.
“There was no evidence of new cancer risks appearing later in life among women who had used oral contraceptives,” the researchers wrote. “Thus, the overall balance of cancer risk among past users of oral contraceptives was neutral with the increased risks counterbalanced by the endometrial, ovarian, and colorectal cancer benefits that persist at least 30 years.”
The results, the researchers wrote, “provide strong evidence that most women do not expose themselves to long-term cancer harm if they choose to use oral contraception, indeed many are likely to be protected.”
The study was funded by the Royal College of General Practitioners, Medical Research Council, Imperial Cancer Research Fund, British Heart Foundation, Schering AG, Schering Health Care, Wyeth Ayerst International, Ortho Cilag, and Searle. The researchers reported having no conflicts of interest.
New findings from a cohort study with more than 4 decades of follow-up show that, while women who have ever used combined oral contraceptives see an increased risk of breast and cervical cancer, the risk disappears within about 5 years after stopping, but a protective effect against colorectal, endometrial, and ovarian cancer persists for more than 30 years.
The findings provide an update to the General Practitioners’ Oral Contraception Study of a United Kingdom cohort recruited in the late 1960s.
The mean age was 70.2 years, most were white, and the mean follow-up was 40.7 years. Women who had used the pill did so a mean 3.66 years and used older, higher-estrogen formulations.
Compared with never users, users of oral contraception had a nonsignificant 4% reduced risk of any cancer. The incidence rate ratio for breast cancer was similar between ever users and nonusers (IRR 1.04; 99% CI, 0.91-1.17). Women who had used OCs saw significant reductions in colorectal (IRR, 0.81; 99% CI, 0.66-0.99), endometrial (IRR, 0.66; 99% CI, 0.48-0.89), ovarian (IRR, 0.67; 99% CI, 0.50-0.89), and lymphatic and hematopoietic cancers (IRR, 0.74; 0.58-0.94), compared with never users.
Lung cancer incidence was increased among ever users of OCs, but only in women who smoked at the time of recruitment.
“There was no evidence of new cancer risks appearing later in life among women who had used oral contraceptives,” the researchers wrote. “Thus, the overall balance of cancer risk among past users of oral contraceptives was neutral with the increased risks counterbalanced by the endometrial, ovarian, and colorectal cancer benefits that persist at least 30 years.”
The results, the researchers wrote, “provide strong evidence that most women do not expose themselves to long-term cancer harm if they choose to use oral contraception, indeed many are likely to be protected.”
The study was funded by the Royal College of General Practitioners, Medical Research Council, Imperial Cancer Research Fund, British Heart Foundation, Schering AG, Schering Health Care, Wyeth Ayerst International, Ortho Cilag, and Searle. The researchers reported having no conflicts of interest.
FROM THE AMERICAN JOURNAL OF OBSTETRICS AND GYNECOLOGY
Key clinical point:
Major finding: At about 40 years of follow-up, women who had ever used combined OCs saw reduced incidence of colorectal (IRR, 0.81), endometrial (IRR, 0.66), ovarian (IRR, 0.67), and lymphatic and hematopoietic cancer (IRR, 0.74), compared with never users.
Data source: A prospective cohort study originally enrolling 46,000 women who were followed for up to 44 years.
Disclosures: The study was funded by the Royal College of General Practitioners, Medical Research Council, Imperial Cancer Research Fund, British Heart Foundation, Schering AG, Schering Health Care, Wyeth Ayerst International, Ortho Cilag, and Searle. The researchers reported having no conflicts of interest.
Scalp cooling reduces hair loss in 50% or more of women in separate studies
Scalp cooling resulted in significant reductions in hair loss in about half of all women who were treated before, during, and after chemotherapy for breast cancer in both the Scalp Cooling Alopecia Prevention (SCALP) randomized clinical trial and in a multicenter prospective cohort study.
However, the effects of the reduced alopecia on quality of life measures were mixed, according to the findings of the studies, which were published online in JAMA.
The findings of the multicenter SCALP trial are from a planned interim analysis of data from 95 women with breast cancer who were undergoing chemotherapy and who were randomized to undergo scalp cooling using the Orbis Paxman Hair Loss Prevention System (OPHLPS), and 47 controls. Successful hair preservation occurred in 50.5% of those in the scalp cooling group, compared with 0% of those in the control group – results which led to early study termination, reported Julie Nangia, MD, of Baylor College of Medicine, Houston, and her colleagues (JAMA 2017 Feb 14. doi: 10.1001/jama.2016.20939).
No serious adverse device-related events occurred in the cooling group, but there also were no significant differences between the groups with respect to changes in quality of life scales from baseline to chemotherapy cycle 4, the investigators found.
Study subjects were women with a mean age of 52.6 years who were receiving anthracycline-based chemotherapy (36%) or taxane-based chemotherapy (64%). Successful hair preservation was defined as grade 0 or 1 based on the Common Terminology Criteria for Adverse Events version 4.1 scale, representing no hair loss or less than 50% hair loss not requiring a wig, respectively. Five women had grade 0 hair loss, and 43 had grade 1 hair loss.
Quality of life was measured using the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire-Core 30 (EORTC-QLQ-30), the Hospital Anxiety and Depression Scale (HADS), and a summary scale of the Body Image Scale. Changes in emotional and social functioning as measured using the EORTC-QLQ-30 did not differ between the groups, including among those with and without hair preservation, after four treatment cycles, and HADS anxiety and depression summary scores were normal both at baseline and after four cycles in both groups, regardless of hair preservation.
Patients will be followed for 5 years to assess for safety and overall survival, the investigators noted.
During a presentation of these findings at the San Antonio Breast Cancer Symposium in December, Dr. Nangia noted that the maker of the OPHLPS is seeking Food and Drug Administration clearance based on the findings, and if approved, the system would compete with the DigniCap (Dignitana AB), which has already received Food and Drug Administration clearance.*
The DigniCap was the device evaluated (at the time under an FDA investigational device exemption) in the prospective cohort study also published in JAMA.
In that study, 106 women receiving adjuvant or neoadjuvant chemotherapy for stage 1 or II breast cancer were treated with scalp cooling between August 2013 and October 2014, and, along with 16 control subjects, were followed for a median of 29.5 months. Self-estimated hair loss at 4 weeks after the last chemotherapy dose was 50% or less, based on the Dean scale (score of 0-2) in 67 of 101 evaluable patients in the scalp cooling group, vs. 0 of 16 in the control group, reported Dr. Hope S. Rugo, MD, of the Helen Diller Family Comprehensive Cancer Center, San Francisco, and her colleagues.
Five patients had no hair loss, and 62 had less than 50% hair loss, the investigators said (JAMA. 2017 Feb 14. doi: 10.1001/jama.2016.21038).
Three of five quality of life measures, as assessed using the EORTC-QLQ, were significantly better at 1 month after the end of chemotherapy vs. at baseline in the cooling group, compared with the control group. For example, 27.3% vs. 56.3% of treatment and control subjects, respectively, reported feeling less physically attractive. The results were similar among those with 50% or less hair loss vs. controls, they noted.
Adverse events associated with cooling included mild headache in four patients. Three patients discontinued treatment due to feeling cold.
The mean age of the women was 53 years. None of those in the treatment group received anthracyclines, thus further research is needed to assess scalp cooling outcomes after treatment with anthracycline regimens, the investigators said, noting that additional research is also needed to assess long-term measures of alopecia and adverse effects. Patients will be followed for a total of 5 years.
Taken together, the findings of these two studies suggest that increased use of scalp cooling is warranted, as it has the potential to both reduce a troublesome side effect of chemotherapy and to remove a common concern – and sometimes a deterrent – among women considering chemotherapy, according to Dawn L. Hershman, MD, of the Herbert Irving Comprehensive Cancer Center at Columbia University.
In an editorial, Dr. Hershman noted that adjuvant chemotherapy reduces the 10-year relative risk of death from breast cancer by about 35%, but that “a substantial number of women who are advised to undergo chemotherapy choose not to receive treatment because of concerns about adverse effects.”
About 50% of patients consider hair loss the most traumatic aspect of chemotherapy, and about 8% said they would decline chemotherapy because of concerns about hair loss, she said.
“Therefore an intervention that could reduce the adverse effects of chemotherapy may lead to improvement in the initiation and completion of therapy, in quality of life, and in survival outcomes,” she wrote (JAMA. 2017 Feb 14;317[6]:587-8).
At face value, the “reassuringly similar” findings from these two studies appear to represent a major step forward for improving the quality of life for individuals with cancer, she added, explaining that while the quality of life data suggest a limited effect, they should be interpreted with caution as the overall effects of the patients’ diagnoses, surgery, and treatment may have influenced the patient-reported outcomes, diminishing the likelihood of detecting differences in quality of life associated with lower rates of alopecia.
Further, the unblinded nature of the intervention may also bias patient-reported outcomes results, she said, adding that “better measures may be needed to capture the effect of treatments on outcomes that are meaningful to patients so that important adverse effects are fully captured in comparative clinical trials.”
Although questions about cost and coverage of scalp cooling remain, Dr. Hershman concluded that until chemotherapy is no longer necessary and some of the distressing adverse effects of cancer treatment can be avoided, interventions such as scalp cooling that can reduce or eliminate toxic effects will help ease the distress and may thereby improve outcomes for patients with breast cancer.
In a separate editorial, Howard (Jack) West, MD, of the Swedish Cancer Institute in Seattle, further notes that a “lingering concern” with respect to scalp cooling is “the speculated potential for increased scalp metastases ... owing to poor local circulation of chemotherapy.”
However, this has not been observed in studies to date, he noted (JAMA. 2017 Feb 14. doi: 10.1001/jamaoncol.2017.0051).
“While some may argue that we need long-term data on timing and patterns of recurrence as well as overall survival to ensure that there is no increased risk of scalp metastases or otherwise compromised outcomes for scalp cooling, there is no evidence thus far to suggest this,” he said, concluding that “it is arguable that growing attention on interventions to reduce chemotherapy-induced alopecia are reaching an inflection point that justifies far more widespread adoption.”
The SCALP trial was supported by Paxman Coolers Ltd. Dr. Nangia reported having no disclosures. The study by Dr. Rugo et al. was funded in part by Dignitana AB, the Laszlo Tauber Family Foundation, the Anne Moore Breast Cancer Research Fund, and the Friedman Family Foundation. Dr. Rugo, Dr. Hershman, and Dr. West reported having no disclosures.
*Correction, 4/5/17: An earlier version of this article misstated the device's FDA status.
Scalp cooling resulted in significant reductions in hair loss in about half of all women who were treated before, during, and after chemotherapy for breast cancer in both the Scalp Cooling Alopecia Prevention (SCALP) randomized clinical trial and in a multicenter prospective cohort study.
However, the effects of the reduced alopecia on quality of life measures were mixed, according to the findings of the studies, which were published online in JAMA.
The findings of the multicenter SCALP trial are from a planned interim analysis of data from 95 women with breast cancer who were undergoing chemotherapy and who were randomized to undergo scalp cooling using the Orbis Paxman Hair Loss Prevention System (OPHLPS), and 47 controls. Successful hair preservation occurred in 50.5% of those in the scalp cooling group, compared with 0% of those in the control group – results which led to early study termination, reported Julie Nangia, MD, of Baylor College of Medicine, Houston, and her colleagues (JAMA 2017 Feb 14. doi: 10.1001/jama.2016.20939).
No serious adverse device-related events occurred in the cooling group, but there also were no significant differences between the groups with respect to changes in quality of life scales from baseline to chemotherapy cycle 4, the investigators found.
Study subjects were women with a mean age of 52.6 years who were receiving anthracycline-based chemotherapy (36%) or taxane-based chemotherapy (64%). Successful hair preservation was defined as grade 0 or 1 based on the Common Terminology Criteria for Adverse Events version 4.1 scale, representing no hair loss or less than 50% hair loss not requiring a wig, respectively. Five women had grade 0 hair loss, and 43 had grade 1 hair loss.
Quality of life was measured using the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire-Core 30 (EORTC-QLQ-30), the Hospital Anxiety and Depression Scale (HADS), and a summary scale of the Body Image Scale. Changes in emotional and social functioning as measured using the EORTC-QLQ-30 did not differ between the groups, including among those with and without hair preservation, after four treatment cycles, and HADS anxiety and depression summary scores were normal both at baseline and after four cycles in both groups, regardless of hair preservation.
Patients will be followed for 5 years to assess for safety and overall survival, the investigators noted.
During a presentation of these findings at the San Antonio Breast Cancer Symposium in December, Dr. Nangia noted that the maker of the OPHLPS is seeking Food and Drug Administration clearance based on the findings, and if approved, the system would compete with the DigniCap (Dignitana AB), which has already received Food and Drug Administration clearance.*
The DigniCap was the device evaluated (at the time under an FDA investigational device exemption) in the prospective cohort study also published in JAMA.
In that study, 106 women receiving adjuvant or neoadjuvant chemotherapy for stage 1 or II breast cancer were treated with scalp cooling between August 2013 and October 2014, and, along with 16 control subjects, were followed for a median of 29.5 months. Self-estimated hair loss at 4 weeks after the last chemotherapy dose was 50% or less, based on the Dean scale (score of 0-2) in 67 of 101 evaluable patients in the scalp cooling group, vs. 0 of 16 in the control group, reported Dr. Hope S. Rugo, MD, of the Helen Diller Family Comprehensive Cancer Center, San Francisco, and her colleagues.
Five patients had no hair loss, and 62 had less than 50% hair loss, the investigators said (JAMA. 2017 Feb 14. doi: 10.1001/jama.2016.21038).
Three of five quality of life measures, as assessed using the EORTC-QLQ, were significantly better at 1 month after the end of chemotherapy vs. at baseline in the cooling group, compared with the control group. For example, 27.3% vs. 56.3% of treatment and control subjects, respectively, reported feeling less physically attractive. The results were similar among those with 50% or less hair loss vs. controls, they noted.
Adverse events associated with cooling included mild headache in four patients. Three patients discontinued treatment due to feeling cold.
The mean age of the women was 53 years. None of those in the treatment group received anthracyclines, thus further research is needed to assess scalp cooling outcomes after treatment with anthracycline regimens, the investigators said, noting that additional research is also needed to assess long-term measures of alopecia and adverse effects. Patients will be followed for a total of 5 years.
Taken together, the findings of these two studies suggest that increased use of scalp cooling is warranted, as it has the potential to both reduce a troublesome side effect of chemotherapy and to remove a common concern – and sometimes a deterrent – among women considering chemotherapy, according to Dawn L. Hershman, MD, of the Herbert Irving Comprehensive Cancer Center at Columbia University.
In an editorial, Dr. Hershman noted that adjuvant chemotherapy reduces the 10-year relative risk of death from breast cancer by about 35%, but that “a substantial number of women who are advised to undergo chemotherapy choose not to receive treatment because of concerns about adverse effects.”
About 50% of patients consider hair loss the most traumatic aspect of chemotherapy, and about 8% said they would decline chemotherapy because of concerns about hair loss, she said.
“Therefore an intervention that could reduce the adverse effects of chemotherapy may lead to improvement in the initiation and completion of therapy, in quality of life, and in survival outcomes,” she wrote (JAMA. 2017 Feb 14;317[6]:587-8).
At face value, the “reassuringly similar” findings from these two studies appear to represent a major step forward for improving the quality of life for individuals with cancer, she added, explaining that while the quality of life data suggest a limited effect, they should be interpreted with caution as the overall effects of the patients’ diagnoses, surgery, and treatment may have influenced the patient-reported outcomes, diminishing the likelihood of detecting differences in quality of life associated with lower rates of alopecia.
Further, the unblinded nature of the intervention may also bias patient-reported outcomes results, she said, adding that “better measures may be needed to capture the effect of treatments on outcomes that are meaningful to patients so that important adverse effects are fully captured in comparative clinical trials.”
Although questions about cost and coverage of scalp cooling remain, Dr. Hershman concluded that until chemotherapy is no longer necessary and some of the distressing adverse effects of cancer treatment can be avoided, interventions such as scalp cooling that can reduce or eliminate toxic effects will help ease the distress and may thereby improve outcomes for patients with breast cancer.
In a separate editorial, Howard (Jack) West, MD, of the Swedish Cancer Institute in Seattle, further notes that a “lingering concern” with respect to scalp cooling is “the speculated potential for increased scalp metastases ... owing to poor local circulation of chemotherapy.”
However, this has not been observed in studies to date, he noted (JAMA. 2017 Feb 14. doi: 10.1001/jamaoncol.2017.0051).
“While some may argue that we need long-term data on timing and patterns of recurrence as well as overall survival to ensure that there is no increased risk of scalp metastases or otherwise compromised outcomes for scalp cooling, there is no evidence thus far to suggest this,” he said, concluding that “it is arguable that growing attention on interventions to reduce chemotherapy-induced alopecia are reaching an inflection point that justifies far more widespread adoption.”
The SCALP trial was supported by Paxman Coolers Ltd. Dr. Nangia reported having no disclosures. The study by Dr. Rugo et al. was funded in part by Dignitana AB, the Laszlo Tauber Family Foundation, the Anne Moore Breast Cancer Research Fund, and the Friedman Family Foundation. Dr. Rugo, Dr. Hershman, and Dr. West reported having no disclosures.
*Correction, 4/5/17: An earlier version of this article misstated the device's FDA status.
Scalp cooling resulted in significant reductions in hair loss in about half of all women who were treated before, during, and after chemotherapy for breast cancer in both the Scalp Cooling Alopecia Prevention (SCALP) randomized clinical trial and in a multicenter prospective cohort study.
However, the effects of the reduced alopecia on quality of life measures were mixed, according to the findings of the studies, which were published online in JAMA.
The findings of the multicenter SCALP trial are from a planned interim analysis of data from 95 women with breast cancer who were undergoing chemotherapy and who were randomized to undergo scalp cooling using the Orbis Paxman Hair Loss Prevention System (OPHLPS), and 47 controls. Successful hair preservation occurred in 50.5% of those in the scalp cooling group, compared with 0% of those in the control group – results which led to early study termination, reported Julie Nangia, MD, of Baylor College of Medicine, Houston, and her colleagues (JAMA 2017 Feb 14. doi: 10.1001/jama.2016.20939).
No serious adverse device-related events occurred in the cooling group, but there also were no significant differences between the groups with respect to changes in quality of life scales from baseline to chemotherapy cycle 4, the investigators found.
Study subjects were women with a mean age of 52.6 years who were receiving anthracycline-based chemotherapy (36%) or taxane-based chemotherapy (64%). Successful hair preservation was defined as grade 0 or 1 based on the Common Terminology Criteria for Adverse Events version 4.1 scale, representing no hair loss or less than 50% hair loss not requiring a wig, respectively. Five women had grade 0 hair loss, and 43 had grade 1 hair loss.
Quality of life was measured using the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire-Core 30 (EORTC-QLQ-30), the Hospital Anxiety and Depression Scale (HADS), and a summary scale of the Body Image Scale. Changes in emotional and social functioning as measured using the EORTC-QLQ-30 did not differ between the groups, including among those with and without hair preservation, after four treatment cycles, and HADS anxiety and depression summary scores were normal both at baseline and after four cycles in both groups, regardless of hair preservation.
Patients will be followed for 5 years to assess for safety and overall survival, the investigators noted.
During a presentation of these findings at the San Antonio Breast Cancer Symposium in December, Dr. Nangia noted that the maker of the OPHLPS is seeking Food and Drug Administration clearance based on the findings, and if approved, the system would compete with the DigniCap (Dignitana AB), which has already received Food and Drug Administration clearance.*
The DigniCap was the device evaluated (at the time under an FDA investigational device exemption) in the prospective cohort study also published in JAMA.
In that study, 106 women receiving adjuvant or neoadjuvant chemotherapy for stage 1 or II breast cancer were treated with scalp cooling between August 2013 and October 2014, and, along with 16 control subjects, were followed for a median of 29.5 months. Self-estimated hair loss at 4 weeks after the last chemotherapy dose was 50% or less, based on the Dean scale (score of 0-2) in 67 of 101 evaluable patients in the scalp cooling group, vs. 0 of 16 in the control group, reported Dr. Hope S. Rugo, MD, of the Helen Diller Family Comprehensive Cancer Center, San Francisco, and her colleagues.
Five patients had no hair loss, and 62 had less than 50% hair loss, the investigators said (JAMA. 2017 Feb 14. doi: 10.1001/jama.2016.21038).
Three of five quality of life measures, as assessed using the EORTC-QLQ, were significantly better at 1 month after the end of chemotherapy vs. at baseline in the cooling group, compared with the control group. For example, 27.3% vs. 56.3% of treatment and control subjects, respectively, reported feeling less physically attractive. The results were similar among those with 50% or less hair loss vs. controls, they noted.
Adverse events associated with cooling included mild headache in four patients. Three patients discontinued treatment due to feeling cold.
The mean age of the women was 53 years. None of those in the treatment group received anthracyclines, thus further research is needed to assess scalp cooling outcomes after treatment with anthracycline regimens, the investigators said, noting that additional research is also needed to assess long-term measures of alopecia and adverse effects. Patients will be followed for a total of 5 years.
Taken together, the findings of these two studies suggest that increased use of scalp cooling is warranted, as it has the potential to both reduce a troublesome side effect of chemotherapy and to remove a common concern – and sometimes a deterrent – among women considering chemotherapy, according to Dawn L. Hershman, MD, of the Herbert Irving Comprehensive Cancer Center at Columbia University.
In an editorial, Dr. Hershman noted that adjuvant chemotherapy reduces the 10-year relative risk of death from breast cancer by about 35%, but that “a substantial number of women who are advised to undergo chemotherapy choose not to receive treatment because of concerns about adverse effects.”
About 50% of patients consider hair loss the most traumatic aspect of chemotherapy, and about 8% said they would decline chemotherapy because of concerns about hair loss, she said.
“Therefore an intervention that could reduce the adverse effects of chemotherapy may lead to improvement in the initiation and completion of therapy, in quality of life, and in survival outcomes,” she wrote (JAMA. 2017 Feb 14;317[6]:587-8).
At face value, the “reassuringly similar” findings from these two studies appear to represent a major step forward for improving the quality of life for individuals with cancer, she added, explaining that while the quality of life data suggest a limited effect, they should be interpreted with caution as the overall effects of the patients’ diagnoses, surgery, and treatment may have influenced the patient-reported outcomes, diminishing the likelihood of detecting differences in quality of life associated with lower rates of alopecia.
Further, the unblinded nature of the intervention may also bias patient-reported outcomes results, she said, adding that “better measures may be needed to capture the effect of treatments on outcomes that are meaningful to patients so that important adverse effects are fully captured in comparative clinical trials.”
Although questions about cost and coverage of scalp cooling remain, Dr. Hershman concluded that until chemotherapy is no longer necessary and some of the distressing adverse effects of cancer treatment can be avoided, interventions such as scalp cooling that can reduce or eliminate toxic effects will help ease the distress and may thereby improve outcomes for patients with breast cancer.
In a separate editorial, Howard (Jack) West, MD, of the Swedish Cancer Institute in Seattle, further notes that a “lingering concern” with respect to scalp cooling is “the speculated potential for increased scalp metastases ... owing to poor local circulation of chemotherapy.”
However, this has not been observed in studies to date, he noted (JAMA. 2017 Feb 14. doi: 10.1001/jamaoncol.2017.0051).
“While some may argue that we need long-term data on timing and patterns of recurrence as well as overall survival to ensure that there is no increased risk of scalp metastases or otherwise compromised outcomes for scalp cooling, there is no evidence thus far to suggest this,” he said, concluding that “it is arguable that growing attention on interventions to reduce chemotherapy-induced alopecia are reaching an inflection point that justifies far more widespread adoption.”
The SCALP trial was supported by Paxman Coolers Ltd. Dr. Nangia reported having no disclosures. The study by Dr. Rugo et al. was funded in part by Dignitana AB, the Laszlo Tauber Family Foundation, the Anne Moore Breast Cancer Research Fund, and the Friedman Family Foundation. Dr. Rugo, Dr. Hershman, and Dr. West reported having no disclosures.
*Correction, 4/5/17: An earlier version of this article misstated the device's FDA status.
FROM JAMA
Key clinical point:
Major finding: About half of the treated women in both studies experienced reduced or no hair loss during chemotherapy.
Data source: A randomized clinical trial involving 142 women, and a prospective cohort study involving 122 women.
Disclosures: The SCALP trial was supported by Paxman Coolers Ltd. Dr. Nangia reported having no disclosures. The study by Dr. Rugo et al. was funded in part by Dignitana AB, the Laszlo Tauber Family Foundation, the Anne Moore Breast Cancer Research Fund, and the Friedman Family Foundation. Dr. Rugo, Dr. Hershman, and Dr. West reported having no disclosures.