User login
Lipid-lowering meds benefit some breast cancer patients
Patients with early-stage, hormone-receptor positive breast cancer who used cholesterol-lowering medication at baseline of a long-term randomized study had more beneficial tumor characteristics and improved outcomes, compared with nonusers.
The findings come from an observation study of a randomized, phase III, double-blind trial conducted by the Breast International Group (BIG) known as BIG 1-98, which enrolled 8,010 postmenopausal women with early-stage, hormone receptor–positive invasive breast cancer from 1998 to 2003. As reported online in the Feb. 13, 2017 issue of the Journal of Clinical Oncology, researchers measured systemic levels of total cholesterol and use of cholesterol-lowering medication at study entry and every 6 months up to 5.5 years. Endpoints of interest were disease-free survival, breast cancer–free interval, and distant recurrence–free interval.
Of the 789 patients who initiated cholesterol-lowering medication during endocrine therapy, most were assigned to letrozole monotherapy (318), followed by sequential tamoxifen-letrozole (189), letrozole-tamoxifen (176), and tamoxifen monotherapy (106).
The results showed that initiation of cholesterol-lowering medication during endocrine therapy was related to improved disease-free survival (hazard ratio 0.79; P = .01), breast cancer–free interval (HR, 0.76; P = .02), and distant recurrence–free interval (HR, 0.74; P = .03).
“The evidence from our observational study warrants consideration of a large, prospective, randomized clinical trial to confirm the value of CLM concomitant with endocrine treatment of breast cancer,” corresponding author Signe Borgquist, MD, PhD, of the division of oncology and pathology at Lund University, Sweden, and her associates concluded. “Further elucidation of the effect upon outcome of the clinical interaction between CLM and endocrine agents – both widely used by patients with breast cancer – will provide exclusive insight to future trial designs.”
The BIG 1-98 trial was supported by Novartis and coordinated by the International Breast Cancer Study Group (IBCSG). Of the 17 study authors, 7 reported having relevant financial disclosures.
[email protected]
Patients with early-stage, hormone-receptor positive breast cancer who used cholesterol-lowering medication at baseline of a long-term randomized study had more beneficial tumor characteristics and improved outcomes, compared with nonusers.
The findings come from an observation study of a randomized, phase III, double-blind trial conducted by the Breast International Group (BIG) known as BIG 1-98, which enrolled 8,010 postmenopausal women with early-stage, hormone receptor–positive invasive breast cancer from 1998 to 2003. As reported online in the Feb. 13, 2017 issue of the Journal of Clinical Oncology, researchers measured systemic levels of total cholesterol and use of cholesterol-lowering medication at study entry and every 6 months up to 5.5 years. Endpoints of interest were disease-free survival, breast cancer–free interval, and distant recurrence–free interval.
Of the 789 patients who initiated cholesterol-lowering medication during endocrine therapy, most were assigned to letrozole monotherapy (318), followed by sequential tamoxifen-letrozole (189), letrozole-tamoxifen (176), and tamoxifen monotherapy (106).
The results showed that initiation of cholesterol-lowering medication during endocrine therapy was related to improved disease-free survival (hazard ratio 0.79; P = .01), breast cancer–free interval (HR, 0.76; P = .02), and distant recurrence–free interval (HR, 0.74; P = .03).
“The evidence from our observational study warrants consideration of a large, prospective, randomized clinical trial to confirm the value of CLM concomitant with endocrine treatment of breast cancer,” corresponding author Signe Borgquist, MD, PhD, of the division of oncology and pathology at Lund University, Sweden, and her associates concluded. “Further elucidation of the effect upon outcome of the clinical interaction between CLM and endocrine agents – both widely used by patients with breast cancer – will provide exclusive insight to future trial designs.”
The BIG 1-98 trial was supported by Novartis and coordinated by the International Breast Cancer Study Group (IBCSG). Of the 17 study authors, 7 reported having relevant financial disclosures.
[email protected]
Patients with early-stage, hormone-receptor positive breast cancer who used cholesterol-lowering medication at baseline of a long-term randomized study had more beneficial tumor characteristics and improved outcomes, compared with nonusers.
The findings come from an observation study of a randomized, phase III, double-blind trial conducted by the Breast International Group (BIG) known as BIG 1-98, which enrolled 8,010 postmenopausal women with early-stage, hormone receptor–positive invasive breast cancer from 1998 to 2003. As reported online in the Feb. 13, 2017 issue of the Journal of Clinical Oncology, researchers measured systemic levels of total cholesterol and use of cholesterol-lowering medication at study entry and every 6 months up to 5.5 years. Endpoints of interest were disease-free survival, breast cancer–free interval, and distant recurrence–free interval.
Of the 789 patients who initiated cholesterol-lowering medication during endocrine therapy, most were assigned to letrozole monotherapy (318), followed by sequential tamoxifen-letrozole (189), letrozole-tamoxifen (176), and tamoxifen monotherapy (106).
The results showed that initiation of cholesterol-lowering medication during endocrine therapy was related to improved disease-free survival (hazard ratio 0.79; P = .01), breast cancer–free interval (HR, 0.76; P = .02), and distant recurrence–free interval (HR, 0.74; P = .03).
“The evidence from our observational study warrants consideration of a large, prospective, randomized clinical trial to confirm the value of CLM concomitant with endocrine treatment of breast cancer,” corresponding author Signe Borgquist, MD, PhD, of the division of oncology and pathology at Lund University, Sweden, and her associates concluded. “Further elucidation of the effect upon outcome of the clinical interaction between CLM and endocrine agents – both widely used by patients with breast cancer – will provide exclusive insight to future trial designs.”
The BIG 1-98 trial was supported by Novartis and coordinated by the International Breast Cancer Study Group (IBCSG). Of the 17 study authors, 7 reported having relevant financial disclosures.
[email protected]
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Are Breast Cancer Patients Satisfied With Their Care?
Japan has a universal health care system with low copays and short wait times for appointments, including those with specialists. Yet patient satisfaction scores are low compared with those of other countries. Researchers from Juntendo Urayasu Hospital, a university hospital in a Tokyo suburb, conducted a study of 214 patients with breast cancer to find out which aspects of radiation oncology care might affect patient satisfaction. The survey included questions about overall treatment, time from diagnosis to treatment start, wait times in the hospital, and length of consultations.
In general, levels of satisfaction were high. However, wait time was significantly negatively associated with both overall satisfaction and satisfaction with the radiation oncologist. Wait time was just under an hour for an average 11-minute consultation. Although this was longer than the “notorious” Japanese situation of a “3 hours wait and 3 minutes consultation,” the researchers say, “We expect that an international audience will appreciate that even 11 minutes is an exceptionally short duration for a consultation visit with a specialist in radiation oncology.”
They note, though, a reasonable caveat. Anyone can walk into their hospital and, for an additional fee, see a specialist on the day they want, which can lead to extended wait times from sheer congestion. Their hospital’s chief breast cancer surgeon sees 60 to 70 patients a day; the radiation oncologist treats 500 to 600 patients a year without resident or trainee support. This situation is typical of Japanese university hospitals, the researchers add.
Related: Breast Cancer Treatment Among Rural and Urban Women at the Veterans Health Administration
Importantly, for Japanese patients, the researchers also included questions to measure patients’ opinions about sharing how they felt with their physicians. The level of sharing correlated with satisfaction, but the researchers point out that in Japan sharing feelings remains “challenging.” Their findings suggest, they say, that if this were improved, patients’ satisfaction might increase.
Japan has a universal health care system with low copays and short wait times for appointments, including those with specialists. Yet patient satisfaction scores are low compared with those of other countries. Researchers from Juntendo Urayasu Hospital, a university hospital in a Tokyo suburb, conducted a study of 214 patients with breast cancer to find out which aspects of radiation oncology care might affect patient satisfaction. The survey included questions about overall treatment, time from diagnosis to treatment start, wait times in the hospital, and length of consultations.
In general, levels of satisfaction were high. However, wait time was significantly negatively associated with both overall satisfaction and satisfaction with the radiation oncologist. Wait time was just under an hour for an average 11-minute consultation. Although this was longer than the “notorious” Japanese situation of a “3 hours wait and 3 minutes consultation,” the researchers say, “We expect that an international audience will appreciate that even 11 minutes is an exceptionally short duration for a consultation visit with a specialist in radiation oncology.”
They note, though, a reasonable caveat. Anyone can walk into their hospital and, for an additional fee, see a specialist on the day they want, which can lead to extended wait times from sheer congestion. Their hospital’s chief breast cancer surgeon sees 60 to 70 patients a day; the radiation oncologist treats 500 to 600 patients a year without resident or trainee support. This situation is typical of Japanese university hospitals, the researchers add.
Related: Breast Cancer Treatment Among Rural and Urban Women at the Veterans Health Administration
Importantly, for Japanese patients, the researchers also included questions to measure patients’ opinions about sharing how they felt with their physicians. The level of sharing correlated with satisfaction, but the researchers point out that in Japan sharing feelings remains “challenging.” Their findings suggest, they say, that if this were improved, patients’ satisfaction might increase.
Japan has a universal health care system with low copays and short wait times for appointments, including those with specialists. Yet patient satisfaction scores are low compared with those of other countries. Researchers from Juntendo Urayasu Hospital, a university hospital in a Tokyo suburb, conducted a study of 214 patients with breast cancer to find out which aspects of radiation oncology care might affect patient satisfaction. The survey included questions about overall treatment, time from diagnosis to treatment start, wait times in the hospital, and length of consultations.
In general, levels of satisfaction were high. However, wait time was significantly negatively associated with both overall satisfaction and satisfaction with the radiation oncologist. Wait time was just under an hour for an average 11-minute consultation. Although this was longer than the “notorious” Japanese situation of a “3 hours wait and 3 minutes consultation,” the researchers say, “We expect that an international audience will appreciate that even 11 minutes is an exceptionally short duration for a consultation visit with a specialist in radiation oncology.”
They note, though, a reasonable caveat. Anyone can walk into their hospital and, for an additional fee, see a specialist on the day they want, which can lead to extended wait times from sheer congestion. Their hospital’s chief breast cancer surgeon sees 60 to 70 patients a day; the radiation oncologist treats 500 to 600 patients a year without resident or trainee support. This situation is typical of Japanese university hospitals, the researchers add.
Related: Breast Cancer Treatment Among Rural and Urban Women at the Veterans Health Administration
Importantly, for Japanese patients, the researchers also included questions to measure patients’ opinions about sharing how they felt with their physicians. The level of sharing correlated with satisfaction, but the researchers point out that in Japan sharing feelings remains “challenging.” Their findings suggest, they say, that if this were improved, patients’ satisfaction might increase.
Optimal adjuvant endocrine therapy use in breast cancer remains elusive
Appropriate use of adjuvant endocrine therapy for breast cancer improved over a 10-year period, but optimal use has not been achieved, according to findings from a retrospective review of more than 980,000 women with stages I-III breast cancer.
As a result, an estimated 14,630 lives were unnecessarily lost during the study period, according to Bobby M. Daly, MD, of Memorial Sloan Kettering Cancer Center in New York, and his colleagues.
Of the 981,729 women in the National Cancer Database from Jan. 1, 2004, to Dec. 31, 2013, who received all or part of their care at the reporting institution and who met eligibility criteria, 818,435 had hormone receptor positive (HR+) disease and 163,294 had hormone receptor negative (HR-) disease.
The percentage of HR+ patients receiving adjuvant endocrine therapy (AET) increased from 69.8% in 2004 to 82.4% in 2013 (annual percentage change, 1.51%), and the percentage of HR- patients decreased from 5.2% to 3.4% during the same time period (annual percentage change, -0.17%), the authors reported online Feb. 2 in JAMA Oncology (2017 Feb 2. doi: 10.1001jamaoncol.2016.6380).
Notably, receipt of AET varied significantly by age, race, geographic location, and receptor status. For example, more than 80% of those aged 50-69 years received AET, compared with 79.1% of those younger than age 40 years and 60.5% of those 80 years or older. African American and Hispanic patients were less likely than non-Hispanic white patients to receive AET (76.4% and 75.9% vs. 79.0%, respectively). The latter finding could be an important contributing factor to the racial disparity in breast cancer survival, the authors noted.
Facility factors also played a role in AET receipt; the rate of receipt varied substantially by facility volume and geographic location.
“We found that facilities in west south central states and low-volume institutions were more likely to misuse and underuse AET,” the investigators wrote, noting that these deficits could affect breast cancer mortality, as geographic differences in such mortality are well documented.
AET receipt also varied based on hormone receptor status, tumor size, and local treatment. Surgery and radiotherapy were the factors most significantly associated with appropriate AET receipt, with only 45% of those who underwent lumpectomy without radiotherapy receiving AET, compared with 90.1% of those receiving postmastectomy radiotherapy, 85.5% in those with postlumpectomy radiotherapy, and 73.2% of those with mastectomy alone.
Women included in this study were age 18 years or older (mean age, 60.8 years). Those undergoing surgery but receiving no neoadjuvant systemic treatment were eligible. Those with prior cancer diagnoses or with missing hormone receptor or AET status were excluded.
Based on recent trial results, the American Society of Clinical Oncology updated treatment guidelines in 2016 to recommend ovarian suppression for 5 years in combination with AET for high-risk premenopausal women, and AET alone both for women with stage I breast cancers that don’t warrant chemotherapy and for node-negative cancers of 1.0 cm or less.
“Similarly, the National Quality Forum (cancer measure 0220) endorsed tamoxifen or a third-generation aromatase inhibitor (considered or administered) within 1 year of diagnosis as a marker of quality care for patients with HR+ American Joint Committee on Cancer stage T1cN0M0, II, or III disease,” the researchers wrote. Studies demonstrated benefit with AET even in those with node-negative cancers of 1.0 cm or less, they added.
Improving adherence to these guidelines has lifesaving potential. In the current cohort, receipt of AET was associated with a 29% relative risk reduction in mortality, after adjusting for numerous patient, disease, and facility-related factors. This suggests that if all women with HR+ disease received AET in concordance with guidelines, 14,630 more lives would have been saved over the 10-year study period, the investigators said.
As for approaches that could help improve the appropriate use of AET, the findings of this cohort study support those from previous studies suggesting that a team-based approach is of benefit.
The finding that local treatments are key factors associated with appropriate AET use suggests that patients who undergo radiotherapy may be more likely to receive standard-of-care therapy in general, the authors explained, adding that “with more physicians involved in a patient’s care, these patients would be more likely to be recommended for guideline concordant care.
“Facilitation of multidisciplinary team-based care may help optimize guideline-concordant treatment by ensuring patients are not lost to follow-up and are recommended for evidence-based care,” the study authors concluded.
Their hope is that with the coming launch of the Medicare Access and Chip Reauthorization Act and value-based reimbursement, efforts will be made to close the quality gap affecting patients in certain age groups, racial minority groups, and geographic regions, and to thereby prevent the loss of lives.
MACRA may boost outcomes
In an interview, Dr. Daly said he believes the coming changes with respect to value-based reimbursement will indeed have an important impact on outcomes.
“The oncology care model, for example, mandates that physicians document that they are providing guideline-concordant care,” said Dr. Daly, assistant attending physician at Memorial Sloan Kettering Cancer Center. “There are also new technologies such as oncology clinical pathways ... that also try to ensure that all patients are receiving care according to guidelines.”
Further, the increasing use of team-based approaches to care and the incorporation of “tumor boards” might explain the growth in optimal AET usage seen in this cohort.
Dr. Daly said he was surprised to find that certain patient groups were being left behind, such as those with estrogen receptor-negative/progesterone receptor-positive disease, African American patients, and younger and older patients, who were less likely to receive AET.
“I think that helps us also focus on patient populations we can target to make sure they are receiving optimal care,” he said, stressing that the findings have important policy implications for figuring out why those patients are being left behind, and raising the standard of care for all patients.
“We are making great strides to providing appropriate guideline-concordant care for breast cancer patients, but there’s still room to improve,” he said.
Dr. Daly serves as a director of and receives compensation from Quadrant Holdings. Frontline Medical News is a subsidiary of Quadrant Holdings. Dr. Daly also reported financial relationships with CVS Health, Johnson & Johnson, McKesson, and Walgreens Boots Alliance.
Appropriate use of adjuvant endocrine therapy for breast cancer improved over a 10-year period, but optimal use has not been achieved, according to findings from a retrospective review of more than 980,000 women with stages I-III breast cancer.
As a result, an estimated 14,630 lives were unnecessarily lost during the study period, according to Bobby M. Daly, MD, of Memorial Sloan Kettering Cancer Center in New York, and his colleagues.
Of the 981,729 women in the National Cancer Database from Jan. 1, 2004, to Dec. 31, 2013, who received all or part of their care at the reporting institution and who met eligibility criteria, 818,435 had hormone receptor positive (HR+) disease and 163,294 had hormone receptor negative (HR-) disease.
The percentage of HR+ patients receiving adjuvant endocrine therapy (AET) increased from 69.8% in 2004 to 82.4% in 2013 (annual percentage change, 1.51%), and the percentage of HR- patients decreased from 5.2% to 3.4% during the same time period (annual percentage change, -0.17%), the authors reported online Feb. 2 in JAMA Oncology (2017 Feb 2. doi: 10.1001jamaoncol.2016.6380).
Notably, receipt of AET varied significantly by age, race, geographic location, and receptor status. For example, more than 80% of those aged 50-69 years received AET, compared with 79.1% of those younger than age 40 years and 60.5% of those 80 years or older. African American and Hispanic patients were less likely than non-Hispanic white patients to receive AET (76.4% and 75.9% vs. 79.0%, respectively). The latter finding could be an important contributing factor to the racial disparity in breast cancer survival, the authors noted.
Facility factors also played a role in AET receipt; the rate of receipt varied substantially by facility volume and geographic location.
“We found that facilities in west south central states and low-volume institutions were more likely to misuse and underuse AET,” the investigators wrote, noting that these deficits could affect breast cancer mortality, as geographic differences in such mortality are well documented.
AET receipt also varied based on hormone receptor status, tumor size, and local treatment. Surgery and radiotherapy were the factors most significantly associated with appropriate AET receipt, with only 45% of those who underwent lumpectomy without radiotherapy receiving AET, compared with 90.1% of those receiving postmastectomy radiotherapy, 85.5% in those with postlumpectomy radiotherapy, and 73.2% of those with mastectomy alone.
Women included in this study were age 18 years or older (mean age, 60.8 years). Those undergoing surgery but receiving no neoadjuvant systemic treatment were eligible. Those with prior cancer diagnoses or with missing hormone receptor or AET status were excluded.
Based on recent trial results, the American Society of Clinical Oncology updated treatment guidelines in 2016 to recommend ovarian suppression for 5 years in combination with AET for high-risk premenopausal women, and AET alone both for women with stage I breast cancers that don’t warrant chemotherapy and for node-negative cancers of 1.0 cm or less.
“Similarly, the National Quality Forum (cancer measure 0220) endorsed tamoxifen or a third-generation aromatase inhibitor (considered or administered) within 1 year of diagnosis as a marker of quality care for patients with HR+ American Joint Committee on Cancer stage T1cN0M0, II, or III disease,” the researchers wrote. Studies demonstrated benefit with AET even in those with node-negative cancers of 1.0 cm or less, they added.
Improving adherence to these guidelines has lifesaving potential. In the current cohort, receipt of AET was associated with a 29% relative risk reduction in mortality, after adjusting for numerous patient, disease, and facility-related factors. This suggests that if all women with HR+ disease received AET in concordance with guidelines, 14,630 more lives would have been saved over the 10-year study period, the investigators said.
As for approaches that could help improve the appropriate use of AET, the findings of this cohort study support those from previous studies suggesting that a team-based approach is of benefit.
The finding that local treatments are key factors associated with appropriate AET use suggests that patients who undergo radiotherapy may be more likely to receive standard-of-care therapy in general, the authors explained, adding that “with more physicians involved in a patient’s care, these patients would be more likely to be recommended for guideline concordant care.
“Facilitation of multidisciplinary team-based care may help optimize guideline-concordant treatment by ensuring patients are not lost to follow-up and are recommended for evidence-based care,” the study authors concluded.
Their hope is that with the coming launch of the Medicare Access and Chip Reauthorization Act and value-based reimbursement, efforts will be made to close the quality gap affecting patients in certain age groups, racial minority groups, and geographic regions, and to thereby prevent the loss of lives.
MACRA may boost outcomes
In an interview, Dr. Daly said he believes the coming changes with respect to value-based reimbursement will indeed have an important impact on outcomes.
“The oncology care model, for example, mandates that physicians document that they are providing guideline-concordant care,” said Dr. Daly, assistant attending physician at Memorial Sloan Kettering Cancer Center. “There are also new technologies such as oncology clinical pathways ... that also try to ensure that all patients are receiving care according to guidelines.”
Further, the increasing use of team-based approaches to care and the incorporation of “tumor boards” might explain the growth in optimal AET usage seen in this cohort.
Dr. Daly said he was surprised to find that certain patient groups were being left behind, such as those with estrogen receptor-negative/progesterone receptor-positive disease, African American patients, and younger and older patients, who were less likely to receive AET.
“I think that helps us also focus on patient populations we can target to make sure they are receiving optimal care,” he said, stressing that the findings have important policy implications for figuring out why those patients are being left behind, and raising the standard of care for all patients.
“We are making great strides to providing appropriate guideline-concordant care for breast cancer patients, but there’s still room to improve,” he said.
Dr. Daly serves as a director of and receives compensation from Quadrant Holdings. Frontline Medical News is a subsidiary of Quadrant Holdings. Dr. Daly also reported financial relationships with CVS Health, Johnson & Johnson, McKesson, and Walgreens Boots Alliance.
Appropriate use of adjuvant endocrine therapy for breast cancer improved over a 10-year period, but optimal use has not been achieved, according to findings from a retrospective review of more than 980,000 women with stages I-III breast cancer.
As a result, an estimated 14,630 lives were unnecessarily lost during the study period, according to Bobby M. Daly, MD, of Memorial Sloan Kettering Cancer Center in New York, and his colleagues.
Of the 981,729 women in the National Cancer Database from Jan. 1, 2004, to Dec. 31, 2013, who received all or part of their care at the reporting institution and who met eligibility criteria, 818,435 had hormone receptor positive (HR+) disease and 163,294 had hormone receptor negative (HR-) disease.
The percentage of HR+ patients receiving adjuvant endocrine therapy (AET) increased from 69.8% in 2004 to 82.4% in 2013 (annual percentage change, 1.51%), and the percentage of HR- patients decreased from 5.2% to 3.4% during the same time period (annual percentage change, -0.17%), the authors reported online Feb. 2 in JAMA Oncology (2017 Feb 2. doi: 10.1001jamaoncol.2016.6380).
Notably, receipt of AET varied significantly by age, race, geographic location, and receptor status. For example, more than 80% of those aged 50-69 years received AET, compared with 79.1% of those younger than age 40 years and 60.5% of those 80 years or older. African American and Hispanic patients were less likely than non-Hispanic white patients to receive AET (76.4% and 75.9% vs. 79.0%, respectively). The latter finding could be an important contributing factor to the racial disparity in breast cancer survival, the authors noted.
Facility factors also played a role in AET receipt; the rate of receipt varied substantially by facility volume and geographic location.
“We found that facilities in west south central states and low-volume institutions were more likely to misuse and underuse AET,” the investigators wrote, noting that these deficits could affect breast cancer mortality, as geographic differences in such mortality are well documented.
AET receipt also varied based on hormone receptor status, tumor size, and local treatment. Surgery and radiotherapy were the factors most significantly associated with appropriate AET receipt, with only 45% of those who underwent lumpectomy without radiotherapy receiving AET, compared with 90.1% of those receiving postmastectomy radiotherapy, 85.5% in those with postlumpectomy radiotherapy, and 73.2% of those with mastectomy alone.
Women included in this study were age 18 years or older (mean age, 60.8 years). Those undergoing surgery but receiving no neoadjuvant systemic treatment were eligible. Those with prior cancer diagnoses or with missing hormone receptor or AET status were excluded.
Based on recent trial results, the American Society of Clinical Oncology updated treatment guidelines in 2016 to recommend ovarian suppression for 5 years in combination with AET for high-risk premenopausal women, and AET alone both for women with stage I breast cancers that don’t warrant chemotherapy and for node-negative cancers of 1.0 cm or less.
“Similarly, the National Quality Forum (cancer measure 0220) endorsed tamoxifen or a third-generation aromatase inhibitor (considered or administered) within 1 year of diagnosis as a marker of quality care for patients with HR+ American Joint Committee on Cancer stage T1cN0M0, II, or III disease,” the researchers wrote. Studies demonstrated benefit with AET even in those with node-negative cancers of 1.0 cm or less, they added.
Improving adherence to these guidelines has lifesaving potential. In the current cohort, receipt of AET was associated with a 29% relative risk reduction in mortality, after adjusting for numerous patient, disease, and facility-related factors. This suggests that if all women with HR+ disease received AET in concordance with guidelines, 14,630 more lives would have been saved over the 10-year study period, the investigators said.
As for approaches that could help improve the appropriate use of AET, the findings of this cohort study support those from previous studies suggesting that a team-based approach is of benefit.
The finding that local treatments are key factors associated with appropriate AET use suggests that patients who undergo radiotherapy may be more likely to receive standard-of-care therapy in general, the authors explained, adding that “with more physicians involved in a patient’s care, these patients would be more likely to be recommended for guideline concordant care.
“Facilitation of multidisciplinary team-based care may help optimize guideline-concordant treatment by ensuring patients are not lost to follow-up and are recommended for evidence-based care,” the study authors concluded.
Their hope is that with the coming launch of the Medicare Access and Chip Reauthorization Act and value-based reimbursement, efforts will be made to close the quality gap affecting patients in certain age groups, racial minority groups, and geographic regions, and to thereby prevent the loss of lives.
MACRA may boost outcomes
In an interview, Dr. Daly said he believes the coming changes with respect to value-based reimbursement will indeed have an important impact on outcomes.
“The oncology care model, for example, mandates that physicians document that they are providing guideline-concordant care,” said Dr. Daly, assistant attending physician at Memorial Sloan Kettering Cancer Center. “There are also new technologies such as oncology clinical pathways ... that also try to ensure that all patients are receiving care according to guidelines.”
Further, the increasing use of team-based approaches to care and the incorporation of “tumor boards” might explain the growth in optimal AET usage seen in this cohort.
Dr. Daly said he was surprised to find that certain patient groups were being left behind, such as those with estrogen receptor-negative/progesterone receptor-positive disease, African American patients, and younger and older patients, who were less likely to receive AET.
“I think that helps us also focus on patient populations we can target to make sure they are receiving optimal care,” he said, stressing that the findings have important policy implications for figuring out why those patients are being left behind, and raising the standard of care for all patients.
“We are making great strides to providing appropriate guideline-concordant care for breast cancer patients, but there’s still room to improve,” he said.
Dr. Daly serves as a director of and receives compensation from Quadrant Holdings. Frontline Medical News is a subsidiary of Quadrant Holdings. Dr. Daly also reported financial relationships with CVS Health, Johnson & Johnson, McKesson, and Walgreens Boots Alliance.
FROM JAMA ONCOLOGY
Key clinical point:
Major finding: Receipt of AET was associated with a 29% relative risk reduction in mortality; use of AET in concordance with guidelines would have saved an estimated 14,630 additional lives over 10 years.
Data source: A retrospective cohort study of 981,729 women with breast cancer.
Disclosures: Dr. Daly serves as a director of and receives compensation from Quadrant Holdings. Frontline Medical News is a subsidiary of Quadrant Holdings. Dr. Daly also reported financial relationships with CVS Health, Johnson & Johnson, McKesson, and Walgreens Boots Alliance.
START again shows safety of hypofractionated doses in early breast cancer
AMSTERDAM – Analyses of patient-reported outcomes in the UK START A and B trials show that radiation doses greater than 2 Gy to the supraclavicular fossa or axilla are safe and are associated with acceptable near- and late-term toxicities, trial investigators reported.
The START trials compared standard adjuvant radiation doses and schedules with accelerated, hypofractionated doses in women with early breast cancer.
At the annual San Antonio Breast Cancer Symposium in 2012, lead investigator John Yarnold, MD, professor of clinical oncology at the Institute of Cancer Research in London, reported that a 40-Gy, 15-fraction regimen was associated with fewer adverse events over 10 years of follow-up compared with a 50-Gy, 25-fraction regimen that was at the time the standard in the United States and the United Kingdom.
Here at an annual congress sponsored by the European Cancer Organisation, Dr. Yarnold reported that among 479 patients in the two trials who had lymphatic radiation, there were no significant differences in arm or shoulder pain, swelling, motion problems, or shoulder stiffness between patients who received conventional 2-Gy fractions for a total of 50 Gy, or lower total doses of radiation divided into fractions larger than 2 Gy.
“A post hoc analysis of outcomes in 479 patients treated with radiotherapy fractions larger than 2 Gy to the supraclavicular fossa and/or the axilla raise no concerns in terms of safety or patient acceptability, and it was for this reason that this schedule has been the standard for locoregional radiotherapy in the UK for some years now,” he said.
START (Standardization of Breast Radiotherapy) A enrolled 2,236 patients from 35 UK centers with breast cancer stages T 1-3, nodal stage N0-1, and no distant metastases (M0) and randomly assigned them to 50 Gy in 25 fractions of 2 Gy each over 5 weeks, 39.0 Gy in 13 fractions of 3.0 Gy over 5 weeks, and 41.6 Gy in 13 fractions of 3.2 Gy over 5 weeks.
In START B, 2,215 women were randomly assigned to the 50 Gy or 40 Gy regimens.
As reported in 2012, over a median follow-up of 9.3 years in START A, the 41.6-Gy regimen was similar in its adverse events profile compared with the 50-Gy dose (hazard ratio [HR] 0.94, and the lower-dose regimen also was comparable in efficacy, as measured by the rate of locoregional tumor relapse.
There were numerically but not significantly more relapses in the 39-Gy dose, however.
In START B, the 40-Gy dose was associated with significantly fewer adverse events over a median of 9.9 years of follow-up (HR 0.77). Efficacy was similar, although there was a nonsignificant trend toward superiority for the hypofractionated regimen.
In the post hoc analysis, the investigators looked at adverse events in 479 total patients in START A and B, 365 of whom had completed patient-reported outcome measures on the European Organization for Research and Treatment of Cancer (EORTC) Breast Cancer–Specific Quality of Life Questionnaire (QLQ-BR23).
A total of 262 patients in START A, and 103 in START B completed the questionnaires at 0, 6, 12, 24, and 60 months. Of these patients. The majority in each arm had radiation to the supraclavicular fossa only, and a minority had axilla radiation alone or in addition to supraclavicular fossa radiation.
In START B, 5-year patient reported outcomes of severity of 3 or 4 on a 4-point scale were low in each treatment arm, with arm or hand swelling reported in 10% of patients on 50 Gy, and 6% of patients on 40 Gy, and shoulder stiffness reported in 12% on the 50-Gy dose, vs. 15% on the 40-Gy dose.
Among all 479 patients with clinical assessments at 10 years, there were also no significant differences in either arm edema or shoulder stiffness between patients who received 50 Gy and those who received other, hypofractionated in each of the two trials.
The findings lend support to the Danish Breast Cancer Cooperative Group’s SKAGEN Trial 1, which is evaluating moderately hypofractionated locoregional adjuvant radiation therapy of early breast cancer combined with a simultaneous integrated boost in patients with an indication for boost, Dr. Yarnold said.
The UK START trials were supported by Cancer Research UK, the Medical Research Council, and the National Cancer Research Institute. Dr. Yarnold reported having no conflicts of interest.
AMSTERDAM – Analyses of patient-reported outcomes in the UK START A and B trials show that radiation doses greater than 2 Gy to the supraclavicular fossa or axilla are safe and are associated with acceptable near- and late-term toxicities, trial investigators reported.
The START trials compared standard adjuvant radiation doses and schedules with accelerated, hypofractionated doses in women with early breast cancer.
At the annual San Antonio Breast Cancer Symposium in 2012, lead investigator John Yarnold, MD, professor of clinical oncology at the Institute of Cancer Research in London, reported that a 40-Gy, 15-fraction regimen was associated with fewer adverse events over 10 years of follow-up compared with a 50-Gy, 25-fraction regimen that was at the time the standard in the United States and the United Kingdom.
Here at an annual congress sponsored by the European Cancer Organisation, Dr. Yarnold reported that among 479 patients in the two trials who had lymphatic radiation, there were no significant differences in arm or shoulder pain, swelling, motion problems, or shoulder stiffness between patients who received conventional 2-Gy fractions for a total of 50 Gy, or lower total doses of radiation divided into fractions larger than 2 Gy.
“A post hoc analysis of outcomes in 479 patients treated with radiotherapy fractions larger than 2 Gy to the supraclavicular fossa and/or the axilla raise no concerns in terms of safety or patient acceptability, and it was for this reason that this schedule has been the standard for locoregional radiotherapy in the UK for some years now,” he said.
START (Standardization of Breast Radiotherapy) A enrolled 2,236 patients from 35 UK centers with breast cancer stages T 1-3, nodal stage N0-1, and no distant metastases (M0) and randomly assigned them to 50 Gy in 25 fractions of 2 Gy each over 5 weeks, 39.0 Gy in 13 fractions of 3.0 Gy over 5 weeks, and 41.6 Gy in 13 fractions of 3.2 Gy over 5 weeks.
In START B, 2,215 women were randomly assigned to the 50 Gy or 40 Gy regimens.
As reported in 2012, over a median follow-up of 9.3 years in START A, the 41.6-Gy regimen was similar in its adverse events profile compared with the 50-Gy dose (hazard ratio [HR] 0.94, and the lower-dose regimen also was comparable in efficacy, as measured by the rate of locoregional tumor relapse.
There were numerically but not significantly more relapses in the 39-Gy dose, however.
In START B, the 40-Gy dose was associated with significantly fewer adverse events over a median of 9.9 years of follow-up (HR 0.77). Efficacy was similar, although there was a nonsignificant trend toward superiority for the hypofractionated regimen.
In the post hoc analysis, the investigators looked at adverse events in 479 total patients in START A and B, 365 of whom had completed patient-reported outcome measures on the European Organization for Research and Treatment of Cancer (EORTC) Breast Cancer–Specific Quality of Life Questionnaire (QLQ-BR23).
A total of 262 patients in START A, and 103 in START B completed the questionnaires at 0, 6, 12, 24, and 60 months. Of these patients. The majority in each arm had radiation to the supraclavicular fossa only, and a minority had axilla radiation alone or in addition to supraclavicular fossa radiation.
In START B, 5-year patient reported outcomes of severity of 3 or 4 on a 4-point scale were low in each treatment arm, with arm or hand swelling reported in 10% of patients on 50 Gy, and 6% of patients on 40 Gy, and shoulder stiffness reported in 12% on the 50-Gy dose, vs. 15% on the 40-Gy dose.
Among all 479 patients with clinical assessments at 10 years, there were also no significant differences in either arm edema or shoulder stiffness between patients who received 50 Gy and those who received other, hypofractionated in each of the two trials.
The findings lend support to the Danish Breast Cancer Cooperative Group’s SKAGEN Trial 1, which is evaluating moderately hypofractionated locoregional adjuvant radiation therapy of early breast cancer combined with a simultaneous integrated boost in patients with an indication for boost, Dr. Yarnold said.
The UK START trials were supported by Cancer Research UK, the Medical Research Council, and the National Cancer Research Institute. Dr. Yarnold reported having no conflicts of interest.
AMSTERDAM – Analyses of patient-reported outcomes in the UK START A and B trials show that radiation doses greater than 2 Gy to the supraclavicular fossa or axilla are safe and are associated with acceptable near- and late-term toxicities, trial investigators reported.
The START trials compared standard adjuvant radiation doses and schedules with accelerated, hypofractionated doses in women with early breast cancer.
At the annual San Antonio Breast Cancer Symposium in 2012, lead investigator John Yarnold, MD, professor of clinical oncology at the Institute of Cancer Research in London, reported that a 40-Gy, 15-fraction regimen was associated with fewer adverse events over 10 years of follow-up compared with a 50-Gy, 25-fraction regimen that was at the time the standard in the United States and the United Kingdom.
Here at an annual congress sponsored by the European Cancer Organisation, Dr. Yarnold reported that among 479 patients in the two trials who had lymphatic radiation, there were no significant differences in arm or shoulder pain, swelling, motion problems, or shoulder stiffness between patients who received conventional 2-Gy fractions for a total of 50 Gy, or lower total doses of radiation divided into fractions larger than 2 Gy.
“A post hoc analysis of outcomes in 479 patients treated with radiotherapy fractions larger than 2 Gy to the supraclavicular fossa and/or the axilla raise no concerns in terms of safety or patient acceptability, and it was for this reason that this schedule has been the standard for locoregional radiotherapy in the UK for some years now,” he said.
START (Standardization of Breast Radiotherapy) A enrolled 2,236 patients from 35 UK centers with breast cancer stages T 1-3, nodal stage N0-1, and no distant metastases (M0) and randomly assigned them to 50 Gy in 25 fractions of 2 Gy each over 5 weeks, 39.0 Gy in 13 fractions of 3.0 Gy over 5 weeks, and 41.6 Gy in 13 fractions of 3.2 Gy over 5 weeks.
In START B, 2,215 women were randomly assigned to the 50 Gy or 40 Gy regimens.
As reported in 2012, over a median follow-up of 9.3 years in START A, the 41.6-Gy regimen was similar in its adverse events profile compared with the 50-Gy dose (hazard ratio [HR] 0.94, and the lower-dose regimen also was comparable in efficacy, as measured by the rate of locoregional tumor relapse.
There were numerically but not significantly more relapses in the 39-Gy dose, however.
In START B, the 40-Gy dose was associated with significantly fewer adverse events over a median of 9.9 years of follow-up (HR 0.77). Efficacy was similar, although there was a nonsignificant trend toward superiority for the hypofractionated regimen.
In the post hoc analysis, the investigators looked at adverse events in 479 total patients in START A and B, 365 of whom had completed patient-reported outcome measures on the European Organization for Research and Treatment of Cancer (EORTC) Breast Cancer–Specific Quality of Life Questionnaire (QLQ-BR23).
A total of 262 patients in START A, and 103 in START B completed the questionnaires at 0, 6, 12, 24, and 60 months. Of these patients. The majority in each arm had radiation to the supraclavicular fossa only, and a minority had axilla radiation alone or in addition to supraclavicular fossa radiation.
In START B, 5-year patient reported outcomes of severity of 3 or 4 on a 4-point scale were low in each treatment arm, with arm or hand swelling reported in 10% of patients on 50 Gy, and 6% of patients on 40 Gy, and shoulder stiffness reported in 12% on the 50-Gy dose, vs. 15% on the 40-Gy dose.
Among all 479 patients with clinical assessments at 10 years, there were also no significant differences in either arm edema or shoulder stiffness between patients who received 50 Gy and those who received other, hypofractionated in each of the two trials.
The findings lend support to the Danish Breast Cancer Cooperative Group’s SKAGEN Trial 1, which is evaluating moderately hypofractionated locoregional adjuvant radiation therapy of early breast cancer combined with a simultaneous integrated boost in patients with an indication for boost, Dr. Yarnold said.
The UK START trials were supported by Cancer Research UK, the Medical Research Council, and the National Cancer Research Institute. Dr. Yarnold reported having no conflicts of interest.
AT ECCO2017
Key clinical point: Additional follow-up supports continued safety of hypofractionated radiation in early breast cancer.
Major finding: There were no significant differences in either patient-reported outcomes at 5 years or clinical assessments at 10 years in patients treated at 40-Gy or 50-Gy doses.
Data source: Post hoc analysis of 479 patients treated in the UK START A and B trials.
Disclosures: The UK START trials were supported by Cancer Research UK, the Medical Research Council, and the National Cancer Research Institute. Dr. Yarnold reported having no conflicts of interest..
Survival better with breast-conserving therapy for early cancers
AMSTERDAM – In real-life practice, women with early, localized breast cancer who underwent breast conserving therapy had better breast cancer–specific and overall survival compared with women who underwent mastectomy, according to investigators in the Netherlands.
Among nearly 130,000 patients treated over two different time periods, breast-conserving surgery and radiation (BCT) was associated with superior survival for women older than 50, patients who did not receive adjuvant chemotherapy, and those with comorbidities – irrespective of either hormonal or human epidermal growth factor receptor 2 (HER2) status, reported Mirelle Lagendijk, MD, of Erasmus Medical Center Cancer Institute in Rotterdam, the Netherlands.
For patients 50 and younger, overall survival (OS), but not breast cancer–specific survival (BCSS), was superior with the more conservative approach.
“Breast conserving therapy in these identified subgroups seems to be the preferable treatment when both treatments are optional,” Dr. Lagendijk said at an annual congress sponsored by the European Cancer Organisation.
Although recent observational studies have shown survival with BCT to be at least equivalent for women with early stage disease, there is still a lack of sufficient data on BCSS, potential confounders such as systemic therapies and comorbidities, and on the relative effects of BCT or mastectomy on subgroups, she said.
The investigators drew data from the Netherlands Cancer Registry on 129,692 patients with early, primary invasive breast cancer without metastases other than to regional lymph nodes (T1-2NO-2MO).
They compared BCT to mastectomy for BCSS and OS in the population as a whole and in subgroups based on prognostic factors. They controlled for age, tumor and nodal stage, comorbidities, systemic therapy, hormone receptor and HER2 status, differentiation grade, morphology, year of treatment, axillary lymph node dissection, and contralateral breast cancer.
They divided patients into two treatment time periods. The older cohort consisted of 60,381 patients treated from 1999 through 2005, 48% of whom underwent mastectomy, with a median follow-up of 11.1 years, and 52% of whom had BCT, with a median follow-up of 12 years.
The more recent cohort consisted of 69,311 patients, 40% of whom had mastectomy with a median follow-up of 5.9 years, and 60% of whom had BCT with a median follow-up of 6.1 years.
In both time periods, deaths from all causes were lower among patients treated with BCT. In the older cohort, 13,960 of 28,968 patients (48.2%) who underwent mastectomy had died, compared with 8,915 of 31,413 patients (28.4%) who underwent BCT. In the more recent cohort, 5,504 of 27,731 (19.8%) of patients who had mastectomies had died, compared with 3,702 of 41,580 (8.9%) who underwent BCT.
“Irrespective of the time cohort and irrespective of the treatment, around 50% of the events were breast cancer related,” Dr. Lagendijk said.
BCSS was superior with BCT in each time cohort (log-rank P less than .001 for each). In the earlier cohort, BCT was significantly superior for BCSS across all disease stages; in the later cohort, it was significant for all but stages T1N1 and T1-2N2.
BCSS was superior for patients in all age categories in the early cohort, and for patients 50 and older in the later cohort.
“The final stratification performed for comorbidities present in the patients evaluated showed, surprisingly, that especially for those patients with comorbidity, there was significantly better breast cancer-specific survival when treated by breast conserving therapy as compared to a mastectomy,” Dr. Lagendijk said.
The investigators acknowledged that the study was limited by its retrospective design, potential confounding by severity, and the inability to show causal relationship between survival and treatment type.
Dutch health agencies sponsored the study. Dr. Lagendijk and Dr. Naredi reported no conflicts of interest.
AMSTERDAM – In real-life practice, women with early, localized breast cancer who underwent breast conserving therapy had better breast cancer–specific and overall survival compared with women who underwent mastectomy, according to investigators in the Netherlands.
Among nearly 130,000 patients treated over two different time periods, breast-conserving surgery and radiation (BCT) was associated with superior survival for women older than 50, patients who did not receive adjuvant chemotherapy, and those with comorbidities – irrespective of either hormonal or human epidermal growth factor receptor 2 (HER2) status, reported Mirelle Lagendijk, MD, of Erasmus Medical Center Cancer Institute in Rotterdam, the Netherlands.
For patients 50 and younger, overall survival (OS), but not breast cancer–specific survival (BCSS), was superior with the more conservative approach.
“Breast conserving therapy in these identified subgroups seems to be the preferable treatment when both treatments are optional,” Dr. Lagendijk said at an annual congress sponsored by the European Cancer Organisation.
Although recent observational studies have shown survival with BCT to be at least equivalent for women with early stage disease, there is still a lack of sufficient data on BCSS, potential confounders such as systemic therapies and comorbidities, and on the relative effects of BCT or mastectomy on subgroups, she said.
The investigators drew data from the Netherlands Cancer Registry on 129,692 patients with early, primary invasive breast cancer without metastases other than to regional lymph nodes (T1-2NO-2MO).
They compared BCT to mastectomy for BCSS and OS in the population as a whole and in subgroups based on prognostic factors. They controlled for age, tumor and nodal stage, comorbidities, systemic therapy, hormone receptor and HER2 status, differentiation grade, morphology, year of treatment, axillary lymph node dissection, and contralateral breast cancer.
They divided patients into two treatment time periods. The older cohort consisted of 60,381 patients treated from 1999 through 2005, 48% of whom underwent mastectomy, with a median follow-up of 11.1 years, and 52% of whom had BCT, with a median follow-up of 12 years.
The more recent cohort consisted of 69,311 patients, 40% of whom had mastectomy with a median follow-up of 5.9 years, and 60% of whom had BCT with a median follow-up of 6.1 years.
In both time periods, deaths from all causes were lower among patients treated with BCT. In the older cohort, 13,960 of 28,968 patients (48.2%) who underwent mastectomy had died, compared with 8,915 of 31,413 patients (28.4%) who underwent BCT. In the more recent cohort, 5,504 of 27,731 (19.8%) of patients who had mastectomies had died, compared with 3,702 of 41,580 (8.9%) who underwent BCT.
“Irrespective of the time cohort and irrespective of the treatment, around 50% of the events were breast cancer related,” Dr. Lagendijk said.
BCSS was superior with BCT in each time cohort (log-rank P less than .001 for each). In the earlier cohort, BCT was significantly superior for BCSS across all disease stages; in the later cohort, it was significant for all but stages T1N1 and T1-2N2.
BCSS was superior for patients in all age categories in the early cohort, and for patients 50 and older in the later cohort.
“The final stratification performed for comorbidities present in the patients evaluated showed, surprisingly, that especially for those patients with comorbidity, there was significantly better breast cancer-specific survival when treated by breast conserving therapy as compared to a mastectomy,” Dr. Lagendijk said.
The investigators acknowledged that the study was limited by its retrospective design, potential confounding by severity, and the inability to show causal relationship between survival and treatment type.
Dutch health agencies sponsored the study. Dr. Lagendijk and Dr. Naredi reported no conflicts of interest.
AMSTERDAM – In real-life practice, women with early, localized breast cancer who underwent breast conserving therapy had better breast cancer–specific and overall survival compared with women who underwent mastectomy, according to investigators in the Netherlands.
Among nearly 130,000 patients treated over two different time periods, breast-conserving surgery and radiation (BCT) was associated with superior survival for women older than 50, patients who did not receive adjuvant chemotherapy, and those with comorbidities – irrespective of either hormonal or human epidermal growth factor receptor 2 (HER2) status, reported Mirelle Lagendijk, MD, of Erasmus Medical Center Cancer Institute in Rotterdam, the Netherlands.
For patients 50 and younger, overall survival (OS), but not breast cancer–specific survival (BCSS), was superior with the more conservative approach.
“Breast conserving therapy in these identified subgroups seems to be the preferable treatment when both treatments are optional,” Dr. Lagendijk said at an annual congress sponsored by the European Cancer Organisation.
Although recent observational studies have shown survival with BCT to be at least equivalent for women with early stage disease, there is still a lack of sufficient data on BCSS, potential confounders such as systemic therapies and comorbidities, and on the relative effects of BCT or mastectomy on subgroups, she said.
The investigators drew data from the Netherlands Cancer Registry on 129,692 patients with early, primary invasive breast cancer without metastases other than to regional lymph nodes (T1-2NO-2MO).
They compared BCT to mastectomy for BCSS and OS in the population as a whole and in subgroups based on prognostic factors. They controlled for age, tumor and nodal stage, comorbidities, systemic therapy, hormone receptor and HER2 status, differentiation grade, morphology, year of treatment, axillary lymph node dissection, and contralateral breast cancer.
They divided patients into two treatment time periods. The older cohort consisted of 60,381 patients treated from 1999 through 2005, 48% of whom underwent mastectomy, with a median follow-up of 11.1 years, and 52% of whom had BCT, with a median follow-up of 12 years.
The more recent cohort consisted of 69,311 patients, 40% of whom had mastectomy with a median follow-up of 5.9 years, and 60% of whom had BCT with a median follow-up of 6.1 years.
In both time periods, deaths from all causes were lower among patients treated with BCT. In the older cohort, 13,960 of 28,968 patients (48.2%) who underwent mastectomy had died, compared with 8,915 of 31,413 patients (28.4%) who underwent BCT. In the more recent cohort, 5,504 of 27,731 (19.8%) of patients who had mastectomies had died, compared with 3,702 of 41,580 (8.9%) who underwent BCT.
“Irrespective of the time cohort and irrespective of the treatment, around 50% of the events were breast cancer related,” Dr. Lagendijk said.
BCSS was superior with BCT in each time cohort (log-rank P less than .001 for each). In the earlier cohort, BCT was significantly superior for BCSS across all disease stages; in the later cohort, it was significant for all but stages T1N1 and T1-2N2.
BCSS was superior for patients in all age categories in the early cohort, and for patients 50 and older in the later cohort.
“The final stratification performed for comorbidities present in the patients evaluated showed, surprisingly, that especially for those patients with comorbidity, there was significantly better breast cancer-specific survival when treated by breast conserving therapy as compared to a mastectomy,” Dr. Lagendijk said.
The investigators acknowledged that the study was limited by its retrospective design, potential confounding by severity, and the inability to show causal relationship between survival and treatment type.
Dutch health agencies sponsored the study. Dr. Lagendijk and Dr. Naredi reported no conflicts of interest.
AT ECCO2017
Key clinical point: Breast cancer–specific survival and overall survival were better among women who had breast-conserving therapy (BCT) compared with mastectomy.
Major finding: BCT was associated with superior survival for women older than 50, patients who did not receive adjuvant chemotherapy, and those with comorbidities.
Data source: Retrospective registry data study of 129,692 women treated for early breast cancer in the Netherlands during 1999-2005 and 2006-2012.
Disclosures: Dutch health agencies sponsored the study. Dr. Langendijk and Dr. Naredi reported no conflicts of interest.
Axial radiotherapy noninferior to cALND in early invasive breast cancer
AMSTERDAM – Axillary radiotherapy appears to be a safe and effective alternative to completion axillary lymph node dissection (cALND) for selected patients who have early invasive breast cancer with sentinel lymph node metastasis, a randomized phase III trial showed.
After a mean of just over 8 years of follow-up, there were no significant differences in breast cancer recurrence, overall survival (OS), disease-free survival (DFS), or breast cancer deaths between patients treated with cALND or axillary radiotherapy, reported Akos Savolt, MD, PhD, of the National Institute of Oncology in Budapest.
“This trial has changed our everyday practice about the optimal care of the axilla,” he said at an annual congress sponsored by the European Cancer Organisation.
An estimated 25%-50% of patients with positive sentinel lymph nodes will have disease that extends to other lymph nodes, and for these patients, cALND is the standard of care.
But patients for whom metastasis is limited to the sentinel lymph node are unlikely to benefit from more extensive dissections, and for these patients, the proven benefits of cALND must be weighed against the significant complications associated with the procedure, including lymphedema, arm pain, nerve injury, shoulder dysfunction, and paresthesias, Dr. Savolt noted.
The OTOASOR (Optimal Treatment of the Axilla – Surgery or Radiotherapy) trial was a single-center study designed to see whether axillary radiotherapy could be noninferior to cALND for preventing recurrence and breast cancer deaths.
From mid-2002 through mid-2009,the investigators enrolled women with primary invasive breast cancer (tumors 3 cm or smaller and no clinically detected lymph node metastases), and randomized them prior to surgery to receive either cALND or axillary radiotherapy at a dose of 50 Gy. Patients also received adjuvant therapy as per institutional guidelines.
A total of 474 patients were evaluable for follow-up: 244 assigned to cALND and 230 assigned to radiotherapy. In all, 94 patients assigned to cALND (38.5%) were found to have additional lymph node metastases.
At a mean follow-up of 97 months, 2% of women in the cALND group had experienced an axillary recurrence (the primary endpoint), compared with 1.7% in the axillary radiation arm.
Overall survival was also similar between the groups, at 77.9% vs. 84.8%, respectively, as was disease-free survival, at 72.1% and 77.4%; neither comparison yielded statistically significant results.
There were also no between-group differences in the percentage of patients alive with recurrence, breast cancer deaths (13.9% of patients in the cALND arm vs. 8.7 in the radiation arm), or deaths from other causes (8.2% vs. 6.5%, respectively).
In contrast, however, 15.3% of patients assigned to cALND reported lymphedema, paresthesia, swelling, arm pain, or shoulder mobility problems, compared with 4.7% treated with radiotherapy. There were no significant differences in quality of life as assessed by standard instruments, however.
The study was supported by the Hungarian National Institute of Oncology. Dr. Savolt and colleagues reported no competing interests.
AMSTERDAM – Axillary radiotherapy appears to be a safe and effective alternative to completion axillary lymph node dissection (cALND) for selected patients who have early invasive breast cancer with sentinel lymph node metastasis, a randomized phase III trial showed.
After a mean of just over 8 years of follow-up, there were no significant differences in breast cancer recurrence, overall survival (OS), disease-free survival (DFS), or breast cancer deaths between patients treated with cALND or axillary radiotherapy, reported Akos Savolt, MD, PhD, of the National Institute of Oncology in Budapest.
“This trial has changed our everyday practice about the optimal care of the axilla,” he said at an annual congress sponsored by the European Cancer Organisation.
An estimated 25%-50% of patients with positive sentinel lymph nodes will have disease that extends to other lymph nodes, and for these patients, cALND is the standard of care.
But patients for whom metastasis is limited to the sentinel lymph node are unlikely to benefit from more extensive dissections, and for these patients, the proven benefits of cALND must be weighed against the significant complications associated with the procedure, including lymphedema, arm pain, nerve injury, shoulder dysfunction, and paresthesias, Dr. Savolt noted.
The OTOASOR (Optimal Treatment of the Axilla – Surgery or Radiotherapy) trial was a single-center study designed to see whether axillary radiotherapy could be noninferior to cALND for preventing recurrence and breast cancer deaths.
From mid-2002 through mid-2009,the investigators enrolled women with primary invasive breast cancer (tumors 3 cm or smaller and no clinically detected lymph node metastases), and randomized them prior to surgery to receive either cALND or axillary radiotherapy at a dose of 50 Gy. Patients also received adjuvant therapy as per institutional guidelines.
A total of 474 patients were evaluable for follow-up: 244 assigned to cALND and 230 assigned to radiotherapy. In all, 94 patients assigned to cALND (38.5%) were found to have additional lymph node metastases.
At a mean follow-up of 97 months, 2% of women in the cALND group had experienced an axillary recurrence (the primary endpoint), compared with 1.7% in the axillary radiation arm.
Overall survival was also similar between the groups, at 77.9% vs. 84.8%, respectively, as was disease-free survival, at 72.1% and 77.4%; neither comparison yielded statistically significant results.
There were also no between-group differences in the percentage of patients alive with recurrence, breast cancer deaths (13.9% of patients in the cALND arm vs. 8.7 in the radiation arm), or deaths from other causes (8.2% vs. 6.5%, respectively).
In contrast, however, 15.3% of patients assigned to cALND reported lymphedema, paresthesia, swelling, arm pain, or shoulder mobility problems, compared with 4.7% treated with radiotherapy. There were no significant differences in quality of life as assessed by standard instruments, however.
The study was supported by the Hungarian National Institute of Oncology. Dr. Savolt and colleagues reported no competing interests.
AMSTERDAM – Axillary radiotherapy appears to be a safe and effective alternative to completion axillary lymph node dissection (cALND) for selected patients who have early invasive breast cancer with sentinel lymph node metastasis, a randomized phase III trial showed.
After a mean of just over 8 years of follow-up, there were no significant differences in breast cancer recurrence, overall survival (OS), disease-free survival (DFS), or breast cancer deaths between patients treated with cALND or axillary radiotherapy, reported Akos Savolt, MD, PhD, of the National Institute of Oncology in Budapest.
“This trial has changed our everyday practice about the optimal care of the axilla,” he said at an annual congress sponsored by the European Cancer Organisation.
An estimated 25%-50% of patients with positive sentinel lymph nodes will have disease that extends to other lymph nodes, and for these patients, cALND is the standard of care.
But patients for whom metastasis is limited to the sentinel lymph node are unlikely to benefit from more extensive dissections, and for these patients, the proven benefits of cALND must be weighed against the significant complications associated with the procedure, including lymphedema, arm pain, nerve injury, shoulder dysfunction, and paresthesias, Dr. Savolt noted.
The OTOASOR (Optimal Treatment of the Axilla – Surgery or Radiotherapy) trial was a single-center study designed to see whether axillary radiotherapy could be noninferior to cALND for preventing recurrence and breast cancer deaths.
From mid-2002 through mid-2009,the investigators enrolled women with primary invasive breast cancer (tumors 3 cm or smaller and no clinically detected lymph node metastases), and randomized them prior to surgery to receive either cALND or axillary radiotherapy at a dose of 50 Gy. Patients also received adjuvant therapy as per institutional guidelines.
A total of 474 patients were evaluable for follow-up: 244 assigned to cALND and 230 assigned to radiotherapy. In all, 94 patients assigned to cALND (38.5%) were found to have additional lymph node metastases.
At a mean follow-up of 97 months, 2% of women in the cALND group had experienced an axillary recurrence (the primary endpoint), compared with 1.7% in the axillary radiation arm.
Overall survival was also similar between the groups, at 77.9% vs. 84.8%, respectively, as was disease-free survival, at 72.1% and 77.4%; neither comparison yielded statistically significant results.
There were also no between-group differences in the percentage of patients alive with recurrence, breast cancer deaths (13.9% of patients in the cALND arm vs. 8.7 in the radiation arm), or deaths from other causes (8.2% vs. 6.5%, respectively).
In contrast, however, 15.3% of patients assigned to cALND reported lymphedema, paresthesia, swelling, arm pain, or shoulder mobility problems, compared with 4.7% treated with radiotherapy. There were no significant differences in quality of life as assessed by standard instruments, however.
The study was supported by the Hungarian National Institute of Oncology. Dr. Savolt and colleagues reported no competing interests.
Key clinical point: Women with early breast cancer with only sentinel lymph node involvement may be able to be spared morbidity from axillary dissection.
Major finding: Axillary radiotherapy was noninferior to completion axillary node dissection for recurrence, overall survival, and disease-free survival.
Data source: A randomized, single-center phase III trial in 474 women with early invasive breast cancer.
Disclosures: The study was supported by the Hungarian National Institute of Oncology. Dr. Savolt and colleagues reported no competing interests.
Women’s Preventive Services Initiative Guidelines provide consensus for practicing ObGyns
The Patient Protection and Affordable Care Act of 2010 (ACA) intended that women have access to critical preventive health services without a copay or deductible. The Institute of Medicine (IOM) was asked to help identify those critical preventive women’s health services. In 2011, the IOM Committee on Preventive Services for Women recommended that all women have access to 9 preventive services, among them1:
- screening for gestational diabetes mellitus (GDM)
- human papilloma virus testing
- contraceptive methods and counseling
- well-woman visits.
The Health Resources and Services Administration (HRSA) of the US Department of Health and Human Services agreed to update the recommended preventive services every 5 years.
In March 2016, HRSA entered into a 5-year cooperative agreement with the American College of Obstetricians and Gynecologists (ACOG) to update the guidelines and to develop additional recommendations to enhance women’s health.2 ACOG launched the Women’s Preventive Services Initiative (WPSI) to develop the 2016 update.
The 5-year grant with HRSA will address many more preventive health services for women across their lifespan as well as implementation strategies so that women receive consistent and appropriate care, regardless of the health care provider’s specialty. The WPSI recognizes that the selection of a provider for well-woman care will be determined as much by a woman’s needs and preferences as by her access to health care services and health plan availability.
The WPSI draft recommendations were released for public comment in September 2016,2 and HRSA approved the recommendations in December 2016.3 In this editorial, I provide a look at which organizations comprise the WPSI and a summary of the 9 recommended preventive health services.
Who makes up the Women’s Preventive Services Initiative?
The WPSI is a collaboration between professional societies and consumer organizations. The goal of the WPSI is “to promote health over the course of a woman’s lifetime through disease prevention and preventive healthcare.” The WPSI advisory panel provides oversight to the effort and the multidisciplinary steering committee develops the recommendations. The WPSI advisory panel includes leaders and experts from 4 major professional organizations, whose members provide the majority of women’s health care in the United States:
- ACOG
- American College of Physicians (ACP)
- American Academy of Family Physicians (AAFP)
- National Association of Nurse Practitioners in Women’s Health (NPWH).
The multidisciplinary steering committee includes the members of the advisory panel, representatives from 17 professional and consumer organizations, a patient representative, and representatives from 6 federal agencies. The WPSI is currently chaired by Jeanne Conry, MD, PhD, past president of ACOG. The steering committee used evidence-based best practices to develop the guidelines and relied heavily on the foundation provided by the 2011 IOM report.1

The 9 WPSI recommendations
Much of the text below is directly quoted from the final recommendations. When a recommendation is paraphrased it is not placed in quotations.
Recommendation 1: Breast cancer screening for average-risk women
“Average-risk women should initiate mammography screening for breast cancer no earlier than age 40 and no later than age 50 years. Screeningmammography should occur at least biennially and as frequently as annually. Screening should continue through at least age 74 years and age alone should not be the basis to stop screening.”
Decisions about when to initiate screening for women between 40 and 50 years of age, how often to screen, and when to stop screening should be based on shared decision making involving the woman and her clinician.
Recommendation 2: Breastfeeding services and supplies
Women should be provided “comprehensive lactation support services including counseling, education and breast feeding equipment and supplies during the antenatal, perinatal, and postpartum periods.” These services will support the successful initiation and maintenance of breastfeeding. Women should have access to double electric breast pumps.
Recommendation 3: Screening for cervical cancer
Average-risk women should initiate cervical cancer screening with cervical cytology at age 21 years and have cervical cytology testing every 3 years from 21 to 29 years of age. “Cotesting with cytology and human papillomavirus (HPV) testing is not recommended for women younger than 30 years. Women aged 30 to 65 years should be screened with cytology and HPV testing every 5 years or cytology alone every 3 years.” Women who have received the HPV vaccine should be screened using these guidelines. Cervical cancer screening is not recommended for women younger than 21 years or older than 65 years who have had adequate prior screening and are not at high risk for cervical cancer. Cervical cancer screening is also not recommended for women who have had a hysterectomy with removal of the cervix and no personal history of cervical intraepithelial neoplasia grade 2 or 3 within the past 20 years.
Recommendation 4: Contraception
Adolescent and adult women should have access to the full range of US Food and Drug Administration–approved female-controlled contraceptives to prevent unintended pregnancy and improve birth outcomes. Multiple visits with a clinician may be needed to select an optimal contraceptive.
Recommendation 5: Screening for gestational diabetes mellitus
Pregnant women should be screened for GDM between 24 and 28 weeks’ gestation to prevent adverse birth outcomes. Screening should be performed with a “50 gm oral glucose challenge test followed by a 3-hour 100 gm oral glucose tolerance test” if the results on the initial oral glucose tolerance test are abnormal. This testing sequence has high sensitivity and specificity. Women with risk factors for diabetes mellitus should be screened for diabetes at the first prenatal visit using current best clinical practice.
Recommendation 6: Screening for human immunodeficiency virus (HIV) infection
Adolescents and women should receive education and risk assessment for HIV annually and should be tested for HIV at least once during their lifetime. Based on assessed risk, screening annually may be appropriate. “Screening for HIV is recommended for all pregnant women upon initiation of prenatal care with retesting during pregnancy based on risk factors. Rapid HIV testing is recommended for pregnant women who present in active labor with an undocumented HIV status.” Risk-based screening does not identify approximately 20% of HIV-infected people. Hence screening annually may be reasonable.
Recommendation 7: Screening for interpersonal and domestic violence
All adolescents and women should be screened annually for both interpersonal violence (IPV) and domestic violence (DV). Intervention services should be available to all adolescents and women. IPV and DV are prevalent problems, and they are often undetected by clinicians. Hence annual screening is recommended.
Recommendation 8: Counseling for sexually transmitted infections
Adolescents and women should be assessed for sexually transmittedinfection (STI) risk. Risk factors include:
- “age younger than 25 years,
- a recent STI,
- a new sex partner,
- multiple partners,
- a partner with concurrent partners,
- a partner with an STI, and
- a lack of or inconsistent condom use.”
Women at increased risk for an STI should receive behavioral counseling.
Recommendation 9: Well-woman preventive visits
Women should “receive at least one preventive care visit per year beginning in adolescence and continuing across the lifespan to ensure that the recommended preventive services including preconception and many services necessary for prenatal and interconception care are obtained. The primary purpose of these visits is the delivery and coordination of recommended preventive services as determined by age and risk factors.”
- Abridged guidelines for the Women's Preventive Services Initiative can be found here: http://www.womenspreventivehealth.org/wp-content/uploads/2017/01/WPSI_2016AbridgedReport.pdf.
- Evidence-based summaries and appendices are available at this link: http://www.womenspreventivehealth.org/wp-content/uploads/2016/12/Evidence-Summaries-and-Appendices.pdf.
I plan on using these recommendations to guide my practice
Historically, many high-profile expert professional groups have developed their own women’s health services guidelines. The proliferation of conflicting guidelines confused both patients and clinicians. Dueling guidelines likely undermine public health because they result in confusion among patients and inconsistent care across the many disciplines that provide medical services to women.
The proliferation of conflicting guidelines for mammography screening for breast cancer is a good example of how dueling guidelines can undermine public health (TABLE).4−7 The WPSI has done a great service to women and clinicians by creating a shared framework for consistently providing critical services across a woman’s entire life. I plan on using these recommendations to guide my practice. Patients and clinicians will greatly benefit from the exceptionally thoughtful women’s preventive services guidelines provided by the WPSI.
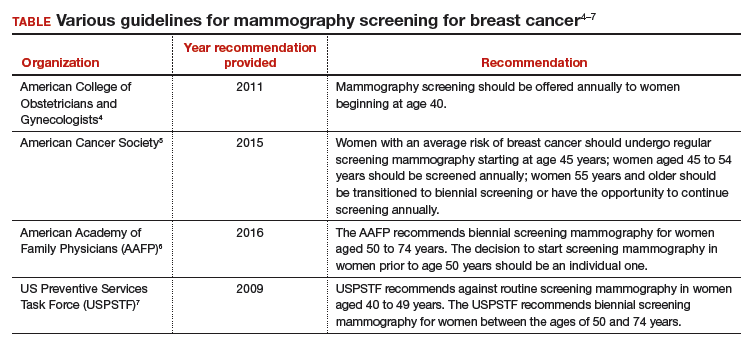
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Institute of Medicine. Clinical preventive services for women: closing the gaps. Washington DC: The National Academies Press; 2011. http://nap.edu/13181. Accessed January 16, 2017.
- American Congress of Obstetricians and Gynecologists (ACOG). Women's Preventive Services Initiative (WPSI). http://www.acog.org/About-ACOG/ACOG-Departments/Annual-Womens-Health-Care/Womens-Preventive-Services-Initiative. Accessed January 16, 2017.
- Health Resources and Services Administration website. Women's preventive services guidelines. https://www.hrsa.gov/womensguidelines/. Accessed January 16, 2017.
- American College of Obstetricians and Gynecologists. Practice bulletin no. 122: breast cancer screening. Obstet Gynecol. 2011;118(2 pt 1):372-382.
- Oeffinger KC, Fontham ET, Etzioni R, et al. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599-1614.
- American Academy of Family Physicians website. Clinical preventive service recommendation: breast cancer. http://www.aafp.org/patient-care/clinical-recommendations/all/breast-cancer.html. Accessed January 16, 2017.
- US Preventive Services Task Force. Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;151(10):716-726.
The Patient Protection and Affordable Care Act of 2010 (ACA) intended that women have access to critical preventive health services without a copay or deductible. The Institute of Medicine (IOM) was asked to help identify those critical preventive women’s health services. In 2011, the IOM Committee on Preventive Services for Women recommended that all women have access to 9 preventive services, among them1:
- screening for gestational diabetes mellitus (GDM)
- human papilloma virus testing
- contraceptive methods and counseling
- well-woman visits.
The Health Resources and Services Administration (HRSA) of the US Department of Health and Human Services agreed to update the recommended preventive services every 5 years.
In March 2016, HRSA entered into a 5-year cooperative agreement with the American College of Obstetricians and Gynecologists (ACOG) to update the guidelines and to develop additional recommendations to enhance women’s health.2 ACOG launched the Women’s Preventive Services Initiative (WPSI) to develop the 2016 update.
The 5-year grant with HRSA will address many more preventive health services for women across their lifespan as well as implementation strategies so that women receive consistent and appropriate care, regardless of the health care provider’s specialty. The WPSI recognizes that the selection of a provider for well-woman care will be determined as much by a woman’s needs and preferences as by her access to health care services and health plan availability.
The WPSI draft recommendations were released for public comment in September 2016,2 and HRSA approved the recommendations in December 2016.3 In this editorial, I provide a look at which organizations comprise the WPSI and a summary of the 9 recommended preventive health services.
Who makes up the Women’s Preventive Services Initiative?
The WPSI is a collaboration between professional societies and consumer organizations. The goal of the WPSI is “to promote health over the course of a woman’s lifetime through disease prevention and preventive healthcare.” The WPSI advisory panel provides oversight to the effort and the multidisciplinary steering committee develops the recommendations. The WPSI advisory panel includes leaders and experts from 4 major professional organizations, whose members provide the majority of women’s health care in the United States:
- ACOG
- American College of Physicians (ACP)
- American Academy of Family Physicians (AAFP)
- National Association of Nurse Practitioners in Women’s Health (NPWH).
The multidisciplinary steering committee includes the members of the advisory panel, representatives from 17 professional and consumer organizations, a patient representative, and representatives from 6 federal agencies. The WPSI is currently chaired by Jeanne Conry, MD, PhD, past president of ACOG. The steering committee used evidence-based best practices to develop the guidelines and relied heavily on the foundation provided by the 2011 IOM report.1

The 9 WPSI recommendations
Much of the text below is directly quoted from the final recommendations. When a recommendation is paraphrased it is not placed in quotations.
Recommendation 1: Breast cancer screening for average-risk women
“Average-risk women should initiate mammography screening for breast cancer no earlier than age 40 and no later than age 50 years. Screeningmammography should occur at least biennially and as frequently as annually. Screening should continue through at least age 74 years and age alone should not be the basis to stop screening.”
Decisions about when to initiate screening for women between 40 and 50 years of age, how often to screen, and when to stop screening should be based on shared decision making involving the woman and her clinician.
Recommendation 2: Breastfeeding services and supplies
Women should be provided “comprehensive lactation support services including counseling, education and breast feeding equipment and supplies during the antenatal, perinatal, and postpartum periods.” These services will support the successful initiation and maintenance of breastfeeding. Women should have access to double electric breast pumps.
Recommendation 3: Screening for cervical cancer
Average-risk women should initiate cervical cancer screening with cervical cytology at age 21 years and have cervical cytology testing every 3 years from 21 to 29 years of age. “Cotesting with cytology and human papillomavirus (HPV) testing is not recommended for women younger than 30 years. Women aged 30 to 65 years should be screened with cytology and HPV testing every 5 years or cytology alone every 3 years.” Women who have received the HPV vaccine should be screened using these guidelines. Cervical cancer screening is not recommended for women younger than 21 years or older than 65 years who have had adequate prior screening and are not at high risk for cervical cancer. Cervical cancer screening is also not recommended for women who have had a hysterectomy with removal of the cervix and no personal history of cervical intraepithelial neoplasia grade 2 or 3 within the past 20 years.
Recommendation 4: Contraception
Adolescent and adult women should have access to the full range of US Food and Drug Administration–approved female-controlled contraceptives to prevent unintended pregnancy and improve birth outcomes. Multiple visits with a clinician may be needed to select an optimal contraceptive.
Recommendation 5: Screening for gestational diabetes mellitus
Pregnant women should be screened for GDM between 24 and 28 weeks’ gestation to prevent adverse birth outcomes. Screening should be performed with a “50 gm oral glucose challenge test followed by a 3-hour 100 gm oral glucose tolerance test” if the results on the initial oral glucose tolerance test are abnormal. This testing sequence has high sensitivity and specificity. Women with risk factors for diabetes mellitus should be screened for diabetes at the first prenatal visit using current best clinical practice.
Recommendation 6: Screening for human immunodeficiency virus (HIV) infection
Adolescents and women should receive education and risk assessment for HIV annually and should be tested for HIV at least once during their lifetime. Based on assessed risk, screening annually may be appropriate. “Screening for HIV is recommended for all pregnant women upon initiation of prenatal care with retesting during pregnancy based on risk factors. Rapid HIV testing is recommended for pregnant women who present in active labor with an undocumented HIV status.” Risk-based screening does not identify approximately 20% of HIV-infected people. Hence screening annually may be reasonable.
Recommendation 7: Screening for interpersonal and domestic violence
All adolescents and women should be screened annually for both interpersonal violence (IPV) and domestic violence (DV). Intervention services should be available to all adolescents and women. IPV and DV are prevalent problems, and they are often undetected by clinicians. Hence annual screening is recommended.
Recommendation 8: Counseling for sexually transmitted infections
Adolescents and women should be assessed for sexually transmittedinfection (STI) risk. Risk factors include:
- “age younger than 25 years,
- a recent STI,
- a new sex partner,
- multiple partners,
- a partner with concurrent partners,
- a partner with an STI, and
- a lack of or inconsistent condom use.”
Women at increased risk for an STI should receive behavioral counseling.
Recommendation 9: Well-woman preventive visits
Women should “receive at least one preventive care visit per year beginning in adolescence and continuing across the lifespan to ensure that the recommended preventive services including preconception and many services necessary for prenatal and interconception care are obtained. The primary purpose of these visits is the delivery and coordination of recommended preventive services as determined by age and risk factors.”
- Abridged guidelines for the Women's Preventive Services Initiative can be found here: http://www.womenspreventivehealth.org/wp-content/uploads/2017/01/WPSI_2016AbridgedReport.pdf.
- Evidence-based summaries and appendices are available at this link: http://www.womenspreventivehealth.org/wp-content/uploads/2016/12/Evidence-Summaries-and-Appendices.pdf.
I plan on using these recommendations to guide my practice
Historically, many high-profile expert professional groups have developed their own women’s health services guidelines. The proliferation of conflicting guidelines confused both patients and clinicians. Dueling guidelines likely undermine public health because they result in confusion among patients and inconsistent care across the many disciplines that provide medical services to women.
The proliferation of conflicting guidelines for mammography screening for breast cancer is a good example of how dueling guidelines can undermine public health (TABLE).4−7 The WPSI has done a great service to women and clinicians by creating a shared framework for consistently providing critical services across a woman’s entire life. I plan on using these recommendations to guide my practice. Patients and clinicians will greatly benefit from the exceptionally thoughtful women’s preventive services guidelines provided by the WPSI.

Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
The Patient Protection and Affordable Care Act of 2010 (ACA) intended that women have access to critical preventive health services without a copay or deductible. The Institute of Medicine (IOM) was asked to help identify those critical preventive women’s health services. In 2011, the IOM Committee on Preventive Services for Women recommended that all women have access to 9 preventive services, among them1:
- screening for gestational diabetes mellitus (GDM)
- human papilloma virus testing
- contraceptive methods and counseling
- well-woman visits.
The Health Resources and Services Administration (HRSA) of the US Department of Health and Human Services agreed to update the recommended preventive services every 5 years.
In March 2016, HRSA entered into a 5-year cooperative agreement with the American College of Obstetricians and Gynecologists (ACOG) to update the guidelines and to develop additional recommendations to enhance women’s health.2 ACOG launched the Women’s Preventive Services Initiative (WPSI) to develop the 2016 update.
The 5-year grant with HRSA will address many more preventive health services for women across their lifespan as well as implementation strategies so that women receive consistent and appropriate care, regardless of the health care provider’s specialty. The WPSI recognizes that the selection of a provider for well-woman care will be determined as much by a woman’s needs and preferences as by her access to health care services and health plan availability.
The WPSI draft recommendations were released for public comment in September 2016,2 and HRSA approved the recommendations in December 2016.3 In this editorial, I provide a look at which organizations comprise the WPSI and a summary of the 9 recommended preventive health services.
Who makes up the Women’s Preventive Services Initiative?
The WPSI is a collaboration between professional societies and consumer organizations. The goal of the WPSI is “to promote health over the course of a woman’s lifetime through disease prevention and preventive healthcare.” The WPSI advisory panel provides oversight to the effort and the multidisciplinary steering committee develops the recommendations. The WPSI advisory panel includes leaders and experts from 4 major professional organizations, whose members provide the majority of women’s health care in the United States:
- ACOG
- American College of Physicians (ACP)
- American Academy of Family Physicians (AAFP)
- National Association of Nurse Practitioners in Women’s Health (NPWH).
The multidisciplinary steering committee includes the members of the advisory panel, representatives from 17 professional and consumer organizations, a patient representative, and representatives from 6 federal agencies. The WPSI is currently chaired by Jeanne Conry, MD, PhD, past president of ACOG. The steering committee used evidence-based best practices to develop the guidelines and relied heavily on the foundation provided by the 2011 IOM report.1

The 9 WPSI recommendations
Much of the text below is directly quoted from the final recommendations. When a recommendation is paraphrased it is not placed in quotations.
Recommendation 1: Breast cancer screening for average-risk women
“Average-risk women should initiate mammography screening for breast cancer no earlier than age 40 and no later than age 50 years. Screeningmammography should occur at least biennially and as frequently as annually. Screening should continue through at least age 74 years and age alone should not be the basis to stop screening.”
Decisions about when to initiate screening for women between 40 and 50 years of age, how often to screen, and when to stop screening should be based on shared decision making involving the woman and her clinician.
Recommendation 2: Breastfeeding services and supplies
Women should be provided “comprehensive lactation support services including counseling, education and breast feeding equipment and supplies during the antenatal, perinatal, and postpartum periods.” These services will support the successful initiation and maintenance of breastfeeding. Women should have access to double electric breast pumps.
Recommendation 3: Screening for cervical cancer
Average-risk women should initiate cervical cancer screening with cervical cytology at age 21 years and have cervical cytology testing every 3 years from 21 to 29 years of age. “Cotesting with cytology and human papillomavirus (HPV) testing is not recommended for women younger than 30 years. Women aged 30 to 65 years should be screened with cytology and HPV testing every 5 years or cytology alone every 3 years.” Women who have received the HPV vaccine should be screened using these guidelines. Cervical cancer screening is not recommended for women younger than 21 years or older than 65 years who have had adequate prior screening and are not at high risk for cervical cancer. Cervical cancer screening is also not recommended for women who have had a hysterectomy with removal of the cervix and no personal history of cervical intraepithelial neoplasia grade 2 or 3 within the past 20 years.
Recommendation 4: Contraception
Adolescent and adult women should have access to the full range of US Food and Drug Administration–approved female-controlled contraceptives to prevent unintended pregnancy and improve birth outcomes. Multiple visits with a clinician may be needed to select an optimal contraceptive.
Recommendation 5: Screening for gestational diabetes mellitus
Pregnant women should be screened for GDM between 24 and 28 weeks’ gestation to prevent adverse birth outcomes. Screening should be performed with a “50 gm oral glucose challenge test followed by a 3-hour 100 gm oral glucose tolerance test” if the results on the initial oral glucose tolerance test are abnormal. This testing sequence has high sensitivity and specificity. Women with risk factors for diabetes mellitus should be screened for diabetes at the first prenatal visit using current best clinical practice.
Recommendation 6: Screening for human immunodeficiency virus (HIV) infection
Adolescents and women should receive education and risk assessment for HIV annually and should be tested for HIV at least once during their lifetime. Based on assessed risk, screening annually may be appropriate. “Screening for HIV is recommended for all pregnant women upon initiation of prenatal care with retesting during pregnancy based on risk factors. Rapid HIV testing is recommended for pregnant women who present in active labor with an undocumented HIV status.” Risk-based screening does not identify approximately 20% of HIV-infected people. Hence screening annually may be reasonable.
Recommendation 7: Screening for interpersonal and domestic violence
All adolescents and women should be screened annually for both interpersonal violence (IPV) and domestic violence (DV). Intervention services should be available to all adolescents and women. IPV and DV are prevalent problems, and they are often undetected by clinicians. Hence annual screening is recommended.
Recommendation 8: Counseling for sexually transmitted infections
Adolescents and women should be assessed for sexually transmittedinfection (STI) risk. Risk factors include:
- “age younger than 25 years,
- a recent STI,
- a new sex partner,
- multiple partners,
- a partner with concurrent partners,
- a partner with an STI, and
- a lack of or inconsistent condom use.”
Women at increased risk for an STI should receive behavioral counseling.
Recommendation 9: Well-woman preventive visits
Women should “receive at least one preventive care visit per year beginning in adolescence and continuing across the lifespan to ensure that the recommended preventive services including preconception and many services necessary for prenatal and interconception care are obtained. The primary purpose of these visits is the delivery and coordination of recommended preventive services as determined by age and risk factors.”
- Abridged guidelines for the Women's Preventive Services Initiative can be found here: http://www.womenspreventivehealth.org/wp-content/uploads/2017/01/WPSI_2016AbridgedReport.pdf.
- Evidence-based summaries and appendices are available at this link: http://www.womenspreventivehealth.org/wp-content/uploads/2016/12/Evidence-Summaries-and-Appendices.pdf.
I plan on using these recommendations to guide my practice
Historically, many high-profile expert professional groups have developed their own women’s health services guidelines. The proliferation of conflicting guidelines confused both patients and clinicians. Dueling guidelines likely undermine public health because they result in confusion among patients and inconsistent care across the many disciplines that provide medical services to women.
The proliferation of conflicting guidelines for mammography screening for breast cancer is a good example of how dueling guidelines can undermine public health (TABLE).4−7 The WPSI has done a great service to women and clinicians by creating a shared framework for consistently providing critical services across a woman’s entire life. I plan on using these recommendations to guide my practice. Patients and clinicians will greatly benefit from the exceptionally thoughtful women’s preventive services guidelines provided by the WPSI.

Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Institute of Medicine. Clinical preventive services for women: closing the gaps. Washington DC: The National Academies Press; 2011. http://nap.edu/13181. Accessed January 16, 2017.
- American Congress of Obstetricians and Gynecologists (ACOG). Women's Preventive Services Initiative (WPSI). http://www.acog.org/About-ACOG/ACOG-Departments/Annual-Womens-Health-Care/Womens-Preventive-Services-Initiative. Accessed January 16, 2017.
- Health Resources and Services Administration website. Women's preventive services guidelines. https://www.hrsa.gov/womensguidelines/. Accessed January 16, 2017.
- American College of Obstetricians and Gynecologists. Practice bulletin no. 122: breast cancer screening. Obstet Gynecol. 2011;118(2 pt 1):372-382.
- Oeffinger KC, Fontham ET, Etzioni R, et al. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599-1614.
- American Academy of Family Physicians website. Clinical preventive service recommendation: breast cancer. http://www.aafp.org/patient-care/clinical-recommendations/all/breast-cancer.html. Accessed January 16, 2017.
- US Preventive Services Task Force. Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;151(10):716-726.
- Institute of Medicine. Clinical preventive services for women: closing the gaps. Washington DC: The National Academies Press; 2011. http://nap.edu/13181. Accessed January 16, 2017.
- American Congress of Obstetricians and Gynecologists (ACOG). Women's Preventive Services Initiative (WPSI). http://www.acog.org/About-ACOG/ACOG-Departments/Annual-Womens-Health-Care/Womens-Preventive-Services-Initiative. Accessed January 16, 2017.
- Health Resources and Services Administration website. Women's preventive services guidelines. https://www.hrsa.gov/womensguidelines/. Accessed January 16, 2017.
- American College of Obstetricians and Gynecologists. Practice bulletin no. 122: breast cancer screening. Obstet Gynecol. 2011;118(2 pt 1):372-382.
- Oeffinger KC, Fontham ET, Etzioni R, et al. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599-1614.
- American Academy of Family Physicians website. Clinical preventive service recommendation: breast cancer. http://www.aafp.org/patient-care/clinical-recommendations/all/breast-cancer.html. Accessed January 16, 2017.
- US Preventive Services Task Force. Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;151(10):716-726.
Residual disease burden is prognostic across breast cancer phenotypes
The Residual Cancer Burden (RCB), a standardized measure of residual disease in pathologic resection specimens following neoadjuvant chemotherapy, was found to be prognostic of long-term survival across all three phenotypic subtypes when it was applied to five breast cancer cohorts totaling 1,158 patients from a single institution, investigators report in the Journal of Clinical Oncology.
If the findings of this retrospective cohort analysis are validated in other cohorts, it would indicate that assessing patients’ RCB index could add “meaningful information to pretreatement clinical and pathologic information and posttreatment yp stage [American Joint Commission on Cancer stage],” said W. Fraser Symmans, MD, of the University of Texas M.D. Anderson Cancer Center, Houston, and his associates.
Residual disease is categorized into four groups: an index of zero (RCB-0) reflects a complete pathologic response to neoadjuvant treatment, RCB-I indicates minimal residual disease, RCB-II indicates moderate residual disease, and RCB-III indicates extensive residual disease. The investigators reviewed pathology specimens to determine the RCB index in a cohort of 219 patients followed for 13 years, a cohort of 262 patients followed for 9 years, a cohort of 342 patients followed for 7 years, a cohort of 132 patients followed for more than 16 years, and a cohort of 203 patients followed for 7 years.
They found that the RCB index predicted the risk of relapse or death across all five cohorts, regardless of other clinical and pathologic variables such as tumor stage or grade, patient age, and type of surgery (J Clin Oncol. 2017 Jan 30. doi: 10.1200/JCO.2015.63.1010).
RCB also was predictive regardless of whether patients had triple-negative disease, HR-positive/HER2-negative disease, HER2-positive disease treated with paclitaxel plus combined fluorouracil, doxorubicin, and cyclophosphamide alone, or HER2-positive disease treated with paclitaxel plus combined fluorouracil, doxorubicin, and cyclophosphamide plus trastuzumab.
In two especially high-risk groups of patients – those with triple-negative breast cancer and those with HER2-positive breast cancer – RCB was the only or the most important predictor of survival. Approximately half of the patients with triple-negative disease had an index of RCB-0 or RCB-I and a good prognosis, while those with an RCB-II or RCB-III index had poor survival, Dr. Symmans and his associates said.
The authors use their main finding – that RCB index provides additional and independent prognostic information to yp stage and other clinical factors – to support their opinion that clinicians should be provided this information.
Sibylle Loibl, MD, is with the German Breast Group in Neu-Isenburg, Germany. Carsten Denkert, MD, is with the German Breast Group and with Charite University Hospital, Berlin. They reported having no relevant financial disclosures. Dr. Loibl and Dr. Denkert made these remarks in an editorial accompanying Dr. Symmans’ report (J Clin Oncol. 2017 Jan 30. doi: 10.1200/JCO.2016.71.3503).
The authors use their main finding – that RCB index provides additional and independent prognostic information to yp stage and other clinical factors – to support their opinion that clinicians should be provided this information.
Sibylle Loibl, MD, is with the German Breast Group in Neu-Isenburg, Germany. Carsten Denkert, MD, is with the German Breast Group and with Charite University Hospital, Berlin. They reported having no relevant financial disclosures. Dr. Loibl and Dr. Denkert made these remarks in an editorial accompanying Dr. Symmans’ report (J Clin Oncol. 2017 Jan 30. doi: 10.1200/JCO.2016.71.3503).
The authors use their main finding – that RCB index provides additional and independent prognostic information to yp stage and other clinical factors – to support their opinion that clinicians should be provided this information.
Sibylle Loibl, MD, is with the German Breast Group in Neu-Isenburg, Germany. Carsten Denkert, MD, is with the German Breast Group and with Charite University Hospital, Berlin. They reported having no relevant financial disclosures. Dr. Loibl and Dr. Denkert made these remarks in an editorial accompanying Dr. Symmans’ report (J Clin Oncol. 2017 Jan 30. doi: 10.1200/JCO.2016.71.3503).
The Residual Cancer Burden (RCB), a standardized measure of residual disease in pathologic resection specimens following neoadjuvant chemotherapy, was found to be prognostic of long-term survival across all three phenotypic subtypes when it was applied to five breast cancer cohorts totaling 1,158 patients from a single institution, investigators report in the Journal of Clinical Oncology.
If the findings of this retrospective cohort analysis are validated in other cohorts, it would indicate that assessing patients’ RCB index could add “meaningful information to pretreatement clinical and pathologic information and posttreatment yp stage [American Joint Commission on Cancer stage],” said W. Fraser Symmans, MD, of the University of Texas M.D. Anderson Cancer Center, Houston, and his associates.
Residual disease is categorized into four groups: an index of zero (RCB-0) reflects a complete pathologic response to neoadjuvant treatment, RCB-I indicates minimal residual disease, RCB-II indicates moderate residual disease, and RCB-III indicates extensive residual disease. The investigators reviewed pathology specimens to determine the RCB index in a cohort of 219 patients followed for 13 years, a cohort of 262 patients followed for 9 years, a cohort of 342 patients followed for 7 years, a cohort of 132 patients followed for more than 16 years, and a cohort of 203 patients followed for 7 years.
They found that the RCB index predicted the risk of relapse or death across all five cohorts, regardless of other clinical and pathologic variables such as tumor stage or grade, patient age, and type of surgery (J Clin Oncol. 2017 Jan 30. doi: 10.1200/JCO.2015.63.1010).
RCB also was predictive regardless of whether patients had triple-negative disease, HR-positive/HER2-negative disease, HER2-positive disease treated with paclitaxel plus combined fluorouracil, doxorubicin, and cyclophosphamide alone, or HER2-positive disease treated with paclitaxel plus combined fluorouracil, doxorubicin, and cyclophosphamide plus trastuzumab.
In two especially high-risk groups of patients – those with triple-negative breast cancer and those with HER2-positive breast cancer – RCB was the only or the most important predictor of survival. Approximately half of the patients with triple-negative disease had an index of RCB-0 or RCB-I and a good prognosis, while those with an RCB-II or RCB-III index had poor survival, Dr. Symmans and his associates said.
The Residual Cancer Burden (RCB), a standardized measure of residual disease in pathologic resection specimens following neoadjuvant chemotherapy, was found to be prognostic of long-term survival across all three phenotypic subtypes when it was applied to five breast cancer cohorts totaling 1,158 patients from a single institution, investigators report in the Journal of Clinical Oncology.
If the findings of this retrospective cohort analysis are validated in other cohorts, it would indicate that assessing patients’ RCB index could add “meaningful information to pretreatement clinical and pathologic information and posttreatment yp stage [American Joint Commission on Cancer stage],” said W. Fraser Symmans, MD, of the University of Texas M.D. Anderson Cancer Center, Houston, and his associates.
Residual disease is categorized into four groups: an index of zero (RCB-0) reflects a complete pathologic response to neoadjuvant treatment, RCB-I indicates minimal residual disease, RCB-II indicates moderate residual disease, and RCB-III indicates extensive residual disease. The investigators reviewed pathology specimens to determine the RCB index in a cohort of 219 patients followed for 13 years, a cohort of 262 patients followed for 9 years, a cohort of 342 patients followed for 7 years, a cohort of 132 patients followed for more than 16 years, and a cohort of 203 patients followed for 7 years.
They found that the RCB index predicted the risk of relapse or death across all five cohorts, regardless of other clinical and pathologic variables such as tumor stage or grade, patient age, and type of surgery (J Clin Oncol. 2017 Jan 30. doi: 10.1200/JCO.2015.63.1010).
RCB also was predictive regardless of whether patients had triple-negative disease, HR-positive/HER2-negative disease, HER2-positive disease treated with paclitaxel plus combined fluorouracil, doxorubicin, and cyclophosphamide alone, or HER2-positive disease treated with paclitaxel plus combined fluorouracil, doxorubicin, and cyclophosphamide plus trastuzumab.
In two especially high-risk groups of patients – those with triple-negative breast cancer and those with HER2-positive breast cancer – RCB was the only or the most important predictor of survival. Approximately half of the patients with triple-negative disease had an index of RCB-0 or RCB-I and a good prognosis, while those with an RCB-II or RCB-III index had poor survival, Dr. Symmans and his associates said.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: The RCB measure is prognostic of long-term survival across all three phenotypic subtypes of breast cancer.
Major finding: The RCB index predicted the risk of relapse or death across five cohorts at a single institution, regardless of other clinical and pathologic variables such as tumor stage or grade, patient age, and type of surgery.
Data source: A retrospective cohort study assessing the RCB’s ability to predict long-term survival using data from five breast cancer cohorts (1,158 patients) with 6-17 years of follow-up.
Disclosures: This study was supported by the Department of Defense Congressionally Directed Funds for Breast Cancer Research, the Breast Cancer Research Foundation, Susan G. Komen for the Cure, and the Nellie B. Connally Breast Center at M.D. Anderson Cancer Center. Dr. Symmans reported ties to ISIS Pharmaceuticals, Nuvera Biosciences, Affymetrix, Celgene, Genentech, and AbbVie, and his associates reported ties to numerous industry sources.
Left ventricle dose predicts heart events after BC radiation
The risk of acute coronary events following radiotherapy for breast cancer is better predicted by the volume of the left ventricle that received 5 Gy than by the mean dose of radiation to the heart, according to a Dutch investigation of 910 women who underwent radiation treatment following breast-conserving surgery.
The finding follows up a 2013 report that found that the risk of acute coronary events (ACE) after breast cancer (BC) radiation could be predicted by the mean radiation heart dose (MHD), the presence of cardiac risk factors, and age (N Engl J Med. 2013 Mar 14;368[11]:987-98. doi: 0.1056/NEJMoa1209825).
The new study validated those findings, but also found that risk prediction was better when mean heart dose (MHD) was replaced by the volume of the left ventricle receiving 5 Gy (LV-V5); the substitution improved the c-statistic to 0.80 (95% confidence interval, 0.72-0.88). Using a weighted ACE risk score based on baseline diabetes, hypertension, and ischemic event history – instead of the risk factor yes-or-no approach from 2013 – further improved predictive power, with a c-statistic of 0.83 (95% CI, 0.75-0.91). Anything over a c-statistic of 0.8 is considered strong; 0.5 is chance, 1.0 is perfect prediction.
For instance, a 70-year-old woman with an LV-V5 of 50% and no cardiac risk factors had an excess ACE risk in the new system of 2.52% within 9 years of radiotherapy (RT). If she had a history of ischemic heart disease, the excess risk increased to 8.42%, the investigators said (J Clin Oncol. 2017 Jan 17. doi: 10.1200/JCO.2016.69.8480).
“Model performance was significantly improved by replacing MHD with LV-V5 and using the weighted ACE risk score.” However, “because we were not able to externally validate the LV-V5 model, this model” requires validation “before it can be used in routine clinical practice,” said investigators, led by Veerle van den Bogaard, MD, of the University of Groningen, the Netherlands.
The women were a median of 59 years old, and they were followed for a median of 7.6 years, with a range of 0.1-10.1 years. Radiation dose information was derived from CT planning scans. The median MHD was 2.37 Gy.
Thirty patients (3.3%) had an ACE, defined as myocardial infarction, coronary revascularization, or death due to ischemic heart disease; 17 had events in the first 5 years. The 5- and 9-year cumulative ACE incidences were 1.9% and 3.9%. Ten of the 30 women died from their cardiac complication.
The model predicted a cumulative ACE incidence at 9 years of 3.5%, which was in line with the observed rate of 3.9%. The excess cumulative risk related to RT was 1.13%. Overall, about 10 patients had an ACE that could be attributed to RT. The cumulative incidence of ACE increased by 16.5% per Gy (95% CI, 0.6-35.0; P = .042). The findings were consistent with the 2013 study.
ACE incidence was not significantly associated with the maximum dose of radiation to the heart.
LV-V5 was the most important prognostic dose-volume parameter associated with the cumulative incidence of ACE, with a hazard ratio of 1.016 (95% CI, 1.002-1.030; P = .016). “Because of this strong association, we chose to include LV-V5 in the model,” the investigators said.
There was no external funding. The lead investigator had no disclosures, but two authors reported institutional research funding from Philips, Roche, and other companies. One was an advisor and speaker for IBA.
The limitations of this study are substantial. As the authors note, their work was based on only a small number of events, and their length of follow-up prevented them from being able to examine risk in the period of 10 years and beyond.
Also, the likelihood is high that the risk of radiation-induced heart disease can be reduced below that seen in the study. Cardiac exposure in many recent studies has been lower. Techniques such as deep inspiration breath holding or treatment in the prone position often can further decrease this exposure. Several randomized trials comparing partial-breast with whole-breast irradiation have found equal local control rates with both approaches, strongly suggesting that judicious individualization of field borders and cardiac blocks are unlikely to compromise outcome for selected patients treated with partial mastectomy.
Nonetheless, at some point, compromising coverage of the breast, chest wall, or nodal target volumes because an arbitrary threshold of a cardiac dose-volume parameter has been exceeded may be dangerous. Computer programs can likely be created to perform calculations of an individual’s excess risk of cardiac events or death in relation to dose-volume and clinical parameters, although I suspect it will be some years before they provide sufficiently validated, narrow estimates of risk to be clinically useful. Perhaps by then we will also have better ways to explain such sobering decisions to patients.
Abram Recht, MD , is a professor of radiation oncology at Harvard Medical School, Boston. He is also an advisor for CareCore and US Oncology, and receives institutional research funding form Genomic Health. He made his comments in an editorial (J Clin Oncol. 2017 Jan 17. doi: 0.1200/JCO.2016.71.4113 ).
The limitations of this study are substantial. As the authors note, their work was based on only a small number of events, and their length of follow-up prevented them from being able to examine risk in the period of 10 years and beyond.
Also, the likelihood is high that the risk of radiation-induced heart disease can be reduced below that seen in the study. Cardiac exposure in many recent studies has been lower. Techniques such as deep inspiration breath holding or treatment in the prone position often can further decrease this exposure. Several randomized trials comparing partial-breast with whole-breast irradiation have found equal local control rates with both approaches, strongly suggesting that judicious individualization of field borders and cardiac blocks are unlikely to compromise outcome for selected patients treated with partial mastectomy.
Nonetheless, at some point, compromising coverage of the breast, chest wall, or nodal target volumes because an arbitrary threshold of a cardiac dose-volume parameter has been exceeded may be dangerous. Computer programs can likely be created to perform calculations of an individual’s excess risk of cardiac events or death in relation to dose-volume and clinical parameters, although I suspect it will be some years before they provide sufficiently validated, narrow estimates of risk to be clinically useful. Perhaps by then we will also have better ways to explain such sobering decisions to patients.
Abram Recht, MD , is a professor of radiation oncology at Harvard Medical School, Boston. He is also an advisor for CareCore and US Oncology, and receives institutional research funding form Genomic Health. He made his comments in an editorial (J Clin Oncol. 2017 Jan 17. doi: 0.1200/JCO.2016.71.4113 ).
The limitations of this study are substantial. As the authors note, their work was based on only a small number of events, and their length of follow-up prevented them from being able to examine risk in the period of 10 years and beyond.
Also, the likelihood is high that the risk of radiation-induced heart disease can be reduced below that seen in the study. Cardiac exposure in many recent studies has been lower. Techniques such as deep inspiration breath holding or treatment in the prone position often can further decrease this exposure. Several randomized trials comparing partial-breast with whole-breast irradiation have found equal local control rates with both approaches, strongly suggesting that judicious individualization of field borders and cardiac blocks are unlikely to compromise outcome for selected patients treated with partial mastectomy.
Nonetheless, at some point, compromising coverage of the breast, chest wall, or nodal target volumes because an arbitrary threshold of a cardiac dose-volume parameter has been exceeded may be dangerous. Computer programs can likely be created to perform calculations of an individual’s excess risk of cardiac events or death in relation to dose-volume and clinical parameters, although I suspect it will be some years before they provide sufficiently validated, narrow estimates of risk to be clinically useful. Perhaps by then we will also have better ways to explain such sobering decisions to patients.
Abram Recht, MD , is a professor of radiation oncology at Harvard Medical School, Boston. He is also an advisor for CareCore and US Oncology, and receives institutional research funding form Genomic Health. He made his comments in an editorial (J Clin Oncol. 2017 Jan 17. doi: 0.1200/JCO.2016.71.4113 ).
The risk of acute coronary events following radiotherapy for breast cancer is better predicted by the volume of the left ventricle that received 5 Gy than by the mean dose of radiation to the heart, according to a Dutch investigation of 910 women who underwent radiation treatment following breast-conserving surgery.
The finding follows up a 2013 report that found that the risk of acute coronary events (ACE) after breast cancer (BC) radiation could be predicted by the mean radiation heart dose (MHD), the presence of cardiac risk factors, and age (N Engl J Med. 2013 Mar 14;368[11]:987-98. doi: 0.1056/NEJMoa1209825).
The new study validated those findings, but also found that risk prediction was better when mean heart dose (MHD) was replaced by the volume of the left ventricle receiving 5 Gy (LV-V5); the substitution improved the c-statistic to 0.80 (95% confidence interval, 0.72-0.88). Using a weighted ACE risk score based on baseline diabetes, hypertension, and ischemic event history – instead of the risk factor yes-or-no approach from 2013 – further improved predictive power, with a c-statistic of 0.83 (95% CI, 0.75-0.91). Anything over a c-statistic of 0.8 is considered strong; 0.5 is chance, 1.0 is perfect prediction.
For instance, a 70-year-old woman with an LV-V5 of 50% and no cardiac risk factors had an excess ACE risk in the new system of 2.52% within 9 years of radiotherapy (RT). If she had a history of ischemic heart disease, the excess risk increased to 8.42%, the investigators said (J Clin Oncol. 2017 Jan 17. doi: 10.1200/JCO.2016.69.8480).
“Model performance was significantly improved by replacing MHD with LV-V5 and using the weighted ACE risk score.” However, “because we were not able to externally validate the LV-V5 model, this model” requires validation “before it can be used in routine clinical practice,” said investigators, led by Veerle van den Bogaard, MD, of the University of Groningen, the Netherlands.
The women were a median of 59 years old, and they were followed for a median of 7.6 years, with a range of 0.1-10.1 years. Radiation dose information was derived from CT planning scans. The median MHD was 2.37 Gy.
Thirty patients (3.3%) had an ACE, defined as myocardial infarction, coronary revascularization, or death due to ischemic heart disease; 17 had events in the first 5 years. The 5- and 9-year cumulative ACE incidences were 1.9% and 3.9%. Ten of the 30 women died from their cardiac complication.
The model predicted a cumulative ACE incidence at 9 years of 3.5%, which was in line with the observed rate of 3.9%. The excess cumulative risk related to RT was 1.13%. Overall, about 10 patients had an ACE that could be attributed to RT. The cumulative incidence of ACE increased by 16.5% per Gy (95% CI, 0.6-35.0; P = .042). The findings were consistent with the 2013 study.
ACE incidence was not significantly associated with the maximum dose of radiation to the heart.
LV-V5 was the most important prognostic dose-volume parameter associated with the cumulative incidence of ACE, with a hazard ratio of 1.016 (95% CI, 1.002-1.030; P = .016). “Because of this strong association, we chose to include LV-V5 in the model,” the investigators said.
There was no external funding. The lead investigator had no disclosures, but two authors reported institutional research funding from Philips, Roche, and other companies. One was an advisor and speaker for IBA.
The risk of acute coronary events following radiotherapy for breast cancer is better predicted by the volume of the left ventricle that received 5 Gy than by the mean dose of radiation to the heart, according to a Dutch investigation of 910 women who underwent radiation treatment following breast-conserving surgery.
The finding follows up a 2013 report that found that the risk of acute coronary events (ACE) after breast cancer (BC) radiation could be predicted by the mean radiation heart dose (MHD), the presence of cardiac risk factors, and age (N Engl J Med. 2013 Mar 14;368[11]:987-98. doi: 0.1056/NEJMoa1209825).
The new study validated those findings, but also found that risk prediction was better when mean heart dose (MHD) was replaced by the volume of the left ventricle receiving 5 Gy (LV-V5); the substitution improved the c-statistic to 0.80 (95% confidence interval, 0.72-0.88). Using a weighted ACE risk score based on baseline diabetes, hypertension, and ischemic event history – instead of the risk factor yes-or-no approach from 2013 – further improved predictive power, with a c-statistic of 0.83 (95% CI, 0.75-0.91). Anything over a c-statistic of 0.8 is considered strong; 0.5 is chance, 1.0 is perfect prediction.
For instance, a 70-year-old woman with an LV-V5 of 50% and no cardiac risk factors had an excess ACE risk in the new system of 2.52% within 9 years of radiotherapy (RT). If she had a history of ischemic heart disease, the excess risk increased to 8.42%, the investigators said (J Clin Oncol. 2017 Jan 17. doi: 10.1200/JCO.2016.69.8480).
“Model performance was significantly improved by replacing MHD with LV-V5 and using the weighted ACE risk score.” However, “because we were not able to externally validate the LV-V5 model, this model” requires validation “before it can be used in routine clinical practice,” said investigators, led by Veerle van den Bogaard, MD, of the University of Groningen, the Netherlands.
The women were a median of 59 years old, and they were followed for a median of 7.6 years, with a range of 0.1-10.1 years. Radiation dose information was derived from CT planning scans. The median MHD was 2.37 Gy.
Thirty patients (3.3%) had an ACE, defined as myocardial infarction, coronary revascularization, or death due to ischemic heart disease; 17 had events in the first 5 years. The 5- and 9-year cumulative ACE incidences were 1.9% and 3.9%. Ten of the 30 women died from their cardiac complication.
The model predicted a cumulative ACE incidence at 9 years of 3.5%, which was in line with the observed rate of 3.9%. The excess cumulative risk related to RT was 1.13%. Overall, about 10 patients had an ACE that could be attributed to RT. The cumulative incidence of ACE increased by 16.5% per Gy (95% CI, 0.6-35.0; P = .042). The findings were consistent with the 2013 study.
ACE incidence was not significantly associated with the maximum dose of radiation to the heart.
LV-V5 was the most important prognostic dose-volume parameter associated with the cumulative incidence of ACE, with a hazard ratio of 1.016 (95% CI, 1.002-1.030; P = .016). “Because of this strong association, we chose to include LV-V5 in the model,” the investigators said.
There was no external funding. The lead investigator had no disclosures, but two authors reported institutional research funding from Philips, Roche, and other companies. One was an advisor and speaker for IBA.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point:
Major finding: Replacing the mean heart dose with the volume of the left ventricle receiving 5 Gy improves the predictive c-statistic to 0.80.
Data source: Investigation of 910 women who underwent radiation treatment following breast-conserving surgery.
Disclosures: There was no external funding. The lead investigator had no disclosures, but two authors reported institutional research funding from Philips, Roche, and other companies. One was an advisor and speaker for IBA.
Circulating microRNAs may predict breast cancer treatment response
SAN ANTONIO – Circulating microRNAs may predict treatment response in HER2-positive breast cancer patients treated with neoadjuvant therapy, according to findings from the randomized phase III NeoALTTO trial.
Specifically, four circulating tumor microRNA signatures were found to predict pathologic complete response to specific treatments at specific time points in a “testing set” of patients from the study, Serena Di Cosimo, MD, of Istituto Nazionale dei Tumori, Milan, reported at the San Antonio Breast Cancer Symposium.
Overall results from the trial, which were reported in 2012 (The Lancet. 379[9816]:633-40) showed that the combination of lapatinib and trastuzumab significantly improved rates of pathologic complete response, compared with either drug alone in patients with HER2-positive early breast cancer, and the authors concluded that dual inhibition of HER2 might be a valid approach in such patients in the neoadjuvant treatment setting.
For the current analysis of NeoALTTO data, microRNA from plasma samples from 435 women – 141 treated with lapatinib, 151 treated with trastuzumab, and 143 treated with a combination of the two – were monitored longitudinally, as NeoALTTO “was done thinking about future translational studies, so blood samples were collected prospectively at baseline, after 2 weeks of treatment, at surgery, and at the time of relapse,” she noted.
Potential microRNAs associated with pathologic complete response were identified for each treatment arm and time point, and a multivariate model was used to identify specific signatures and their predictive capability.
A total of 30 microRNAs and 6 microRNA signatures were found to be predict pathologic complete response in a “training population,” and the predictive ability of 4 of those was confirmed in the testing set for lapatinib at baseline (area under the curve [AUC] = .86), lapatinib after 2 weeks (AUC = .71), trastuzumab after 2 weeks (AUC=.81), and lapatinib and trastuzumab after 2 weeks (AUC = .67), Dr. Di Cosimo said.
“Results obtained early post treatment are of special value,” she said, explaining that “women with unfavorable microRNA signatures can be expected to have poor response after just 2 weeks of treatment.”
The findings are of note because a significant proportion of breast cancer patients treated in the neoadjuvant setting do not achieve pathologic complete response and have an increased risk of relapse after surgery. Further, there is a lack of reliable predictors of response to help guide therapy in clinical practice, Dr. Ci Cosimo said.
“This is the first evidence of the potential of circulating microRNAs to discriminate between responsive and unresponsive HER2+ breast cancer patients,” she said.
At present, the signatures, overall, have been associated with pathologic complete response, but not event-free survival. However, one microRNA signature (140-5p) appeared to be associated with event-free survival after 2 weeks among patients treated with trastuzumab, she said, adding that functional studies are ongoing to investigate the biological role of microRNAs, and independent validation studies are planned.
Dr. Di Cosimo reported having no disclosures.
SAN ANTONIO – Circulating microRNAs may predict treatment response in HER2-positive breast cancer patients treated with neoadjuvant therapy, according to findings from the randomized phase III NeoALTTO trial.
Specifically, four circulating tumor microRNA signatures were found to predict pathologic complete response to specific treatments at specific time points in a “testing set” of patients from the study, Serena Di Cosimo, MD, of Istituto Nazionale dei Tumori, Milan, reported at the San Antonio Breast Cancer Symposium.
Overall results from the trial, which were reported in 2012 (The Lancet. 379[9816]:633-40) showed that the combination of lapatinib and trastuzumab significantly improved rates of pathologic complete response, compared with either drug alone in patients with HER2-positive early breast cancer, and the authors concluded that dual inhibition of HER2 might be a valid approach in such patients in the neoadjuvant treatment setting.
For the current analysis of NeoALTTO data, microRNA from plasma samples from 435 women – 141 treated with lapatinib, 151 treated with trastuzumab, and 143 treated with a combination of the two – were monitored longitudinally, as NeoALTTO “was done thinking about future translational studies, so blood samples were collected prospectively at baseline, after 2 weeks of treatment, at surgery, and at the time of relapse,” she noted.
Potential microRNAs associated with pathologic complete response were identified for each treatment arm and time point, and a multivariate model was used to identify specific signatures and their predictive capability.
A total of 30 microRNAs and 6 microRNA signatures were found to be predict pathologic complete response in a “training population,” and the predictive ability of 4 of those was confirmed in the testing set for lapatinib at baseline (area under the curve [AUC] = .86), lapatinib after 2 weeks (AUC = .71), trastuzumab after 2 weeks (AUC=.81), and lapatinib and trastuzumab after 2 weeks (AUC = .67), Dr. Di Cosimo said.
“Results obtained early post treatment are of special value,” she said, explaining that “women with unfavorable microRNA signatures can be expected to have poor response after just 2 weeks of treatment.”
The findings are of note because a significant proportion of breast cancer patients treated in the neoadjuvant setting do not achieve pathologic complete response and have an increased risk of relapse after surgery. Further, there is a lack of reliable predictors of response to help guide therapy in clinical practice, Dr. Ci Cosimo said.
“This is the first evidence of the potential of circulating microRNAs to discriminate between responsive and unresponsive HER2+ breast cancer patients,” she said.
At present, the signatures, overall, have been associated with pathologic complete response, but not event-free survival. However, one microRNA signature (140-5p) appeared to be associated with event-free survival after 2 weeks among patients treated with trastuzumab, she said, adding that functional studies are ongoing to investigate the biological role of microRNAs, and independent validation studies are planned.
Dr. Di Cosimo reported having no disclosures.
SAN ANTONIO – Circulating microRNAs may predict treatment response in HER2-positive breast cancer patients treated with neoadjuvant therapy, according to findings from the randomized phase III NeoALTTO trial.
Specifically, four circulating tumor microRNA signatures were found to predict pathologic complete response to specific treatments at specific time points in a “testing set” of patients from the study, Serena Di Cosimo, MD, of Istituto Nazionale dei Tumori, Milan, reported at the San Antonio Breast Cancer Symposium.
Overall results from the trial, which were reported in 2012 (The Lancet. 379[9816]:633-40) showed that the combination of lapatinib and trastuzumab significantly improved rates of pathologic complete response, compared with either drug alone in patients with HER2-positive early breast cancer, and the authors concluded that dual inhibition of HER2 might be a valid approach in such patients in the neoadjuvant treatment setting.
For the current analysis of NeoALTTO data, microRNA from plasma samples from 435 women – 141 treated with lapatinib, 151 treated with trastuzumab, and 143 treated with a combination of the two – were monitored longitudinally, as NeoALTTO “was done thinking about future translational studies, so blood samples were collected prospectively at baseline, after 2 weeks of treatment, at surgery, and at the time of relapse,” she noted.
Potential microRNAs associated with pathologic complete response were identified for each treatment arm and time point, and a multivariate model was used to identify specific signatures and their predictive capability.
A total of 30 microRNAs and 6 microRNA signatures were found to be predict pathologic complete response in a “training population,” and the predictive ability of 4 of those was confirmed in the testing set for lapatinib at baseline (area under the curve [AUC] = .86), lapatinib after 2 weeks (AUC = .71), trastuzumab after 2 weeks (AUC=.81), and lapatinib and trastuzumab after 2 weeks (AUC = .67), Dr. Di Cosimo said.
“Results obtained early post treatment are of special value,” she said, explaining that “women with unfavorable microRNA signatures can be expected to have poor response after just 2 weeks of treatment.”
The findings are of note because a significant proportion of breast cancer patients treated in the neoadjuvant setting do not achieve pathologic complete response and have an increased risk of relapse after surgery. Further, there is a lack of reliable predictors of response to help guide therapy in clinical practice, Dr. Ci Cosimo said.
“This is the first evidence of the potential of circulating microRNAs to discriminate between responsive and unresponsive HER2+ breast cancer patients,” she said.
At present, the signatures, overall, have been associated with pathologic complete response, but not event-free survival. However, one microRNA signature (140-5p) appeared to be associated with event-free survival after 2 weeks among patients treated with trastuzumab, she said, adding that functional studies are ongoing to investigate the biological role of microRNAs, and independent validation studies are planned.
Dr. Di Cosimo reported having no disclosures.
AT SABCS 2016
Key clinical point:
Major finding: A total of 30 microRNAs and 6 microRNA signatures were found to predict pathologic complete response in a “training population,” and the predictive ability of 4 of those was confirmed in the testing set.
Data source: An analysis of data from the randomized phase III NeoALTTO trial.
Disclosures: Dr. Di Cosimo reported having no disclosures.