User login
Fulvestrant/everolimus improves PFS in HR+, HER2– advanced breast cancer
SAN ANTONIO – Adding everolimus to fulvestrant doubled median progression-free survival among postmenopausal women with hormone-receptor positive, human epidermal growth factor receptor 2–negative (HER2-negative) metastatic breast cancer resistant to therapy with an aromatase inhibitor [AI] in the PrECOG 0102 study.
In the randomized phase II trial, the combination of the mammalian target of rapamycin (mTOR) inhibitor everolimus (Afinitor) with the selective estrogen receptor down-regulator [SERD] fulvestrant (Faslodex) was associated with a median progression-free survival of 10.4 months, compared with 5.1 months for fulvestrant plus placebo, reported Noah S. Kornblum, MD, of Montefiore Einstein Center for Cancer Care, New York.
This study provides additional evidence that adding everolimus to anti-estrogen therapy in AI-resistant disease improves clinical outcomes,” he said at the San Antonio Breast Cancer Symposium.
Most women with hormone receptor–positive breast cancer treated with an AI will eventually develop resistance to these agents. Strategies for overcoming resistance include the addition of everolimus to a steroid AI, exemestane (Aromasin), as in the BOLERO-2 trial.
“Another strategy for overcoming AI resistance is by more completely blocking estrogen-receptor signaling through the use of a selective estrogen receptor down-regulator, which may result in more complete blockade of the ER signaling pathway than a steroidal AI such as exemestane,” Dr. Kornblum said.
To test this hypothesis, the investigators enrolled 131 postmenopausal women with inoperable locally advanced or metastatic hormone receptor–positive, HER2-negative breast cancer resistant to AIs.
AI resistance was defined as relapse while receiving adjuvant AI therapy, and/or progression after one or more AIs for metastatic disease. The patients could have had no more than one prior chemotherapy regimen for metastatic disease.
The patients were stratified by Eastern Cooperative Oncology Group performance status, presence of measurable disease, and prior chemotherapy status, and were then randomized to receive either high-dose fulvestrant (500 mg on day 1 and 15 of cycle 1, and then on day 1 of cycles 2-12) plus oral everolimus 10 mg/day, or fulvestrant and placebo.
The trial had an induction phase, in which patients were treated until evidence of progressive disease or unacceptable toxicity for a maximum of 12 28-day cycles, and a continuation phase in which patients who had neither disease progression nor experienced unacceptable toxicities could have their data unblinded and could continue on fulvestrant/everolimus.
The trial did not include the use of corticosteroid-containing mouthwash for prevention of treatment-associated stomatitis, because the trial was designed before the evidence of the benefit of such prophylaxis became public, Dr. Kornblum said.
As noted before, the primary endpoint of PFS by investigator assessment was significantly better with the combination, at 10.4 vs. 5.1 months for the fulvestrant/placebo group. The hazard ratio was 0.60 (P = .02).
There was no difference in overall survival (OS), however. Median OS was 24.8 months among patients treated with everolimus, compared with not yet reached in the placebo arm (not statistically significant).
The combination was associated with more grade 3 adverse events than fulvestrant/placebo (48% vs. 14%, respectively). The most common grade 3 adverse events occurring in more than 5% of patients were stomatitis, pneumonitis, fatigue and hyperglycemia. Overall, the safety profile of the combination was consistent with that seen in BOLERO-2, Dr. Kornblum said.
In all, 10% of patients assigned to the combination and 12% assigned to placebo withdrew from the study because of adverse events; these patients were included in the analysis, which was by intention-to-treat.
Regarding future directions, Dr. Kornblum noted that “our study was completed prior to the availability of the CD4/6 inhibitors, which are effective added to both first-line AI therapy or second-line fulvestrant in AI-resistant disease. On the other hand, mTOR inhibitors are effective as second line therapy in AI-resistant disease, but possibly not as first-line therapy.”
As only a few patients in the study received a prior CDK4/6 inhibitor, “further work is required to define whether prior CDk4/6 inhibitor therapy would impact response to combination mTOR-inhibitor/SERD therapy,” he added.
Potential role for the combination?
Following the presentation, SABCS fixture Steven “Vogl, New York” Vogl, MD, asked what to do when fulvestrant-based therapy fails.*
“I’m interested in what to do after progression. Let’s say the patient gets fulvestrant, gets everolimus, has a nice response – 9 months later, the tumor gets worse. We’re all pretty sure we shouldn’t continue the fulvestrant. I would love PrECOG to do a study in those patients of giving megestrol or megestrol/everolimus, and see if the everolimus extended beyond progression does the patient some good, as does trastuzumab,” he said.
“Sounds like a good idea. I’m game, let’s roll up our sleeves and do it together,” Dr. Kornblum replied.
The study was sponsored by PrECOG with financial support from Novartis. Dr. Kornblum reported having no conflicts of interest.
Correction 12/8/16: An earlier version of this article misstated Dr. Steven Vogl's name.
SAN ANTONIO – Adding everolimus to fulvestrant doubled median progression-free survival among postmenopausal women with hormone-receptor positive, human epidermal growth factor receptor 2–negative (HER2-negative) metastatic breast cancer resistant to therapy with an aromatase inhibitor [AI] in the PrECOG 0102 study.
In the randomized phase II trial, the combination of the mammalian target of rapamycin (mTOR) inhibitor everolimus (Afinitor) with the selective estrogen receptor down-regulator [SERD] fulvestrant (Faslodex) was associated with a median progression-free survival of 10.4 months, compared with 5.1 months for fulvestrant plus placebo, reported Noah S. Kornblum, MD, of Montefiore Einstein Center for Cancer Care, New York.
This study provides additional evidence that adding everolimus to anti-estrogen therapy in AI-resistant disease improves clinical outcomes,” he said at the San Antonio Breast Cancer Symposium.
Most women with hormone receptor–positive breast cancer treated with an AI will eventually develop resistance to these agents. Strategies for overcoming resistance include the addition of everolimus to a steroid AI, exemestane (Aromasin), as in the BOLERO-2 trial.
“Another strategy for overcoming AI resistance is by more completely blocking estrogen-receptor signaling through the use of a selective estrogen receptor down-regulator, which may result in more complete blockade of the ER signaling pathway than a steroidal AI such as exemestane,” Dr. Kornblum said.
To test this hypothesis, the investigators enrolled 131 postmenopausal women with inoperable locally advanced or metastatic hormone receptor–positive, HER2-negative breast cancer resistant to AIs.
AI resistance was defined as relapse while receiving adjuvant AI therapy, and/or progression after one or more AIs for metastatic disease. The patients could have had no more than one prior chemotherapy regimen for metastatic disease.
The patients were stratified by Eastern Cooperative Oncology Group performance status, presence of measurable disease, and prior chemotherapy status, and were then randomized to receive either high-dose fulvestrant (500 mg on day 1 and 15 of cycle 1, and then on day 1 of cycles 2-12) plus oral everolimus 10 mg/day, or fulvestrant and placebo.
The trial had an induction phase, in which patients were treated until evidence of progressive disease or unacceptable toxicity for a maximum of 12 28-day cycles, and a continuation phase in which patients who had neither disease progression nor experienced unacceptable toxicities could have their data unblinded and could continue on fulvestrant/everolimus.
The trial did not include the use of corticosteroid-containing mouthwash for prevention of treatment-associated stomatitis, because the trial was designed before the evidence of the benefit of such prophylaxis became public, Dr. Kornblum said.
As noted before, the primary endpoint of PFS by investigator assessment was significantly better with the combination, at 10.4 vs. 5.1 months for the fulvestrant/placebo group. The hazard ratio was 0.60 (P = .02).
There was no difference in overall survival (OS), however. Median OS was 24.8 months among patients treated with everolimus, compared with not yet reached in the placebo arm (not statistically significant).
The combination was associated with more grade 3 adverse events than fulvestrant/placebo (48% vs. 14%, respectively). The most common grade 3 adverse events occurring in more than 5% of patients were stomatitis, pneumonitis, fatigue and hyperglycemia. Overall, the safety profile of the combination was consistent with that seen in BOLERO-2, Dr. Kornblum said.
In all, 10% of patients assigned to the combination and 12% assigned to placebo withdrew from the study because of adverse events; these patients were included in the analysis, which was by intention-to-treat.
Regarding future directions, Dr. Kornblum noted that “our study was completed prior to the availability of the CD4/6 inhibitors, which are effective added to both first-line AI therapy or second-line fulvestrant in AI-resistant disease. On the other hand, mTOR inhibitors are effective as second line therapy in AI-resistant disease, but possibly not as first-line therapy.”
As only a few patients in the study received a prior CDK4/6 inhibitor, “further work is required to define whether prior CDk4/6 inhibitor therapy would impact response to combination mTOR-inhibitor/SERD therapy,” he added.
Potential role for the combination?
Following the presentation, SABCS fixture Steven “Vogl, New York” Vogl, MD, asked what to do when fulvestrant-based therapy fails.*
“I’m interested in what to do after progression. Let’s say the patient gets fulvestrant, gets everolimus, has a nice response – 9 months later, the tumor gets worse. We’re all pretty sure we shouldn’t continue the fulvestrant. I would love PrECOG to do a study in those patients of giving megestrol or megestrol/everolimus, and see if the everolimus extended beyond progression does the patient some good, as does trastuzumab,” he said.
“Sounds like a good idea. I’m game, let’s roll up our sleeves and do it together,” Dr. Kornblum replied.
The study was sponsored by PrECOG with financial support from Novartis. Dr. Kornblum reported having no conflicts of interest.
Correction 12/8/16: An earlier version of this article misstated Dr. Steven Vogl's name.
SAN ANTONIO – Adding everolimus to fulvestrant doubled median progression-free survival among postmenopausal women with hormone-receptor positive, human epidermal growth factor receptor 2–negative (HER2-negative) metastatic breast cancer resistant to therapy with an aromatase inhibitor [AI] in the PrECOG 0102 study.
In the randomized phase II trial, the combination of the mammalian target of rapamycin (mTOR) inhibitor everolimus (Afinitor) with the selective estrogen receptor down-regulator [SERD] fulvestrant (Faslodex) was associated with a median progression-free survival of 10.4 months, compared with 5.1 months for fulvestrant plus placebo, reported Noah S. Kornblum, MD, of Montefiore Einstein Center for Cancer Care, New York.
This study provides additional evidence that adding everolimus to anti-estrogen therapy in AI-resistant disease improves clinical outcomes,” he said at the San Antonio Breast Cancer Symposium.
Most women with hormone receptor–positive breast cancer treated with an AI will eventually develop resistance to these agents. Strategies for overcoming resistance include the addition of everolimus to a steroid AI, exemestane (Aromasin), as in the BOLERO-2 trial.
“Another strategy for overcoming AI resistance is by more completely blocking estrogen-receptor signaling through the use of a selective estrogen receptor down-regulator, which may result in more complete blockade of the ER signaling pathway than a steroidal AI such as exemestane,” Dr. Kornblum said.
To test this hypothesis, the investigators enrolled 131 postmenopausal women with inoperable locally advanced or metastatic hormone receptor–positive, HER2-negative breast cancer resistant to AIs.
AI resistance was defined as relapse while receiving adjuvant AI therapy, and/or progression after one or more AIs for metastatic disease. The patients could have had no more than one prior chemotherapy regimen for metastatic disease.
The patients were stratified by Eastern Cooperative Oncology Group performance status, presence of measurable disease, and prior chemotherapy status, and were then randomized to receive either high-dose fulvestrant (500 mg on day 1 and 15 of cycle 1, and then on day 1 of cycles 2-12) plus oral everolimus 10 mg/day, or fulvestrant and placebo.
The trial had an induction phase, in which patients were treated until evidence of progressive disease or unacceptable toxicity for a maximum of 12 28-day cycles, and a continuation phase in which patients who had neither disease progression nor experienced unacceptable toxicities could have their data unblinded and could continue on fulvestrant/everolimus.
The trial did not include the use of corticosteroid-containing mouthwash for prevention of treatment-associated stomatitis, because the trial was designed before the evidence of the benefit of such prophylaxis became public, Dr. Kornblum said.
As noted before, the primary endpoint of PFS by investigator assessment was significantly better with the combination, at 10.4 vs. 5.1 months for the fulvestrant/placebo group. The hazard ratio was 0.60 (P = .02).
There was no difference in overall survival (OS), however. Median OS was 24.8 months among patients treated with everolimus, compared with not yet reached in the placebo arm (not statistically significant).
The combination was associated with more grade 3 adverse events than fulvestrant/placebo (48% vs. 14%, respectively). The most common grade 3 adverse events occurring in more than 5% of patients were stomatitis, pneumonitis, fatigue and hyperglycemia. Overall, the safety profile of the combination was consistent with that seen in BOLERO-2, Dr. Kornblum said.
In all, 10% of patients assigned to the combination and 12% assigned to placebo withdrew from the study because of adverse events; these patients were included in the analysis, which was by intention-to-treat.
Regarding future directions, Dr. Kornblum noted that “our study was completed prior to the availability of the CD4/6 inhibitors, which are effective added to both first-line AI therapy or second-line fulvestrant in AI-resistant disease. On the other hand, mTOR inhibitors are effective as second line therapy in AI-resistant disease, but possibly not as first-line therapy.”
As only a few patients in the study received a prior CDK4/6 inhibitor, “further work is required to define whether prior CDk4/6 inhibitor therapy would impact response to combination mTOR-inhibitor/SERD therapy,” he added.
Potential role for the combination?
Following the presentation, SABCS fixture Steven “Vogl, New York” Vogl, MD, asked what to do when fulvestrant-based therapy fails.*
“I’m interested in what to do after progression. Let’s say the patient gets fulvestrant, gets everolimus, has a nice response – 9 months later, the tumor gets worse. We’re all pretty sure we shouldn’t continue the fulvestrant. I would love PrECOG to do a study in those patients of giving megestrol or megestrol/everolimus, and see if the everolimus extended beyond progression does the patient some good, as does trastuzumab,” he said.
“Sounds like a good idea. I’m game, let’s roll up our sleeves and do it together,” Dr. Kornblum replied.
The study was sponsored by PrECOG with financial support from Novartis. Dr. Kornblum reported having no conflicts of interest.
Correction 12/8/16: An earlier version of this article misstated Dr. Steven Vogl's name.
AT SABCS 2016
Key clinical point: This study provides further evidence of the benefits of adding an mTOR inhibitor to anti-hormonal therapy in postmenopausal women with advanced breast cancer resistant to aromatase inhibitors.
Major finding: Fulvestrant/everolimus was associated with progression-free survival of 10.4 months, vs. 5.1 months for fulvestrant/placebo.
Data source: Randomized phase II trial of 131 women with hormone-receptor positive, HER2-negative locally advanced of metastatic breast cancer resistant to aromatase inhibitors.
Disclosures: The study was sponsored by PrECOG with financial support from Novartis. Dr. Kornblum reported having no conflicts of interest.
VIDEO: Investigator discusses fulvestrant/everolimus combo
SAN ANTONIO – Most women with hormone receptor–positive breast cancer treated with an aromatase inhibitor will eventually develop resistance to these agents. Strategies for overcoming resistance include the addition of everolimus (Affinitor) to a steroidal aromatase inhibitor (AI) such as exemestane (Aromasin), as in the BOLERO-2 trial.
Alternatively, blocking estrogen-receptor signaling through the use of a selective estrogen receptor down regulator, such as fulvestrant (Faslodex), may result in more complete blockade of the ER signaling pathway than would a steroidal AI such as exemestane.
In this video interview at the San Antonio Breast Cancer Symposium, Noah S. Kornblum, MD, of the Montefiore-Einstein Center for Cancer Care, New York, discusses findings from the phase II PrECOG 0102 trial comparing a combination of fulvestrant and everolimus to fulvestrant and placebo for the treatment of postmenopausal women with hormone receptor–positive, HER2-negative breast cancer resistant to AI therapy.
The combination was associated with a median progression-free survival of 10.4 months, compared with 5.1 months for fulvestrant plus placebo (hazard ratio, 0.60; P = .02).
Dr. Kornblum says that the study provides additional evidence for adding everolimus to anti-estrogen therapy in AI-resistant disease.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – Most women with hormone receptor–positive breast cancer treated with an aromatase inhibitor will eventually develop resistance to these agents. Strategies for overcoming resistance include the addition of everolimus (Affinitor) to a steroidal aromatase inhibitor (AI) such as exemestane (Aromasin), as in the BOLERO-2 trial.
Alternatively, blocking estrogen-receptor signaling through the use of a selective estrogen receptor down regulator, such as fulvestrant (Faslodex), may result in more complete blockade of the ER signaling pathway than would a steroidal AI such as exemestane.
In this video interview at the San Antonio Breast Cancer Symposium, Noah S. Kornblum, MD, of the Montefiore-Einstein Center for Cancer Care, New York, discusses findings from the phase II PrECOG 0102 trial comparing a combination of fulvestrant and everolimus to fulvestrant and placebo for the treatment of postmenopausal women with hormone receptor–positive, HER2-negative breast cancer resistant to AI therapy.
The combination was associated with a median progression-free survival of 10.4 months, compared with 5.1 months for fulvestrant plus placebo (hazard ratio, 0.60; P = .02).
Dr. Kornblum says that the study provides additional evidence for adding everolimus to anti-estrogen therapy in AI-resistant disease.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN ANTONIO – Most women with hormone receptor–positive breast cancer treated with an aromatase inhibitor will eventually develop resistance to these agents. Strategies for overcoming resistance include the addition of everolimus (Affinitor) to a steroidal aromatase inhibitor (AI) such as exemestane (Aromasin), as in the BOLERO-2 trial.
Alternatively, blocking estrogen-receptor signaling through the use of a selective estrogen receptor down regulator, such as fulvestrant (Faslodex), may result in more complete blockade of the ER signaling pathway than would a steroidal AI such as exemestane.
In this video interview at the San Antonio Breast Cancer Symposium, Noah S. Kornblum, MD, of the Montefiore-Einstein Center for Cancer Care, New York, discusses findings from the phase II PrECOG 0102 trial comparing a combination of fulvestrant and everolimus to fulvestrant and placebo for the treatment of postmenopausal women with hormone receptor–positive, HER2-negative breast cancer resistant to AI therapy.
The combination was associated with a median progression-free survival of 10.4 months, compared with 5.1 months for fulvestrant plus placebo (hazard ratio, 0.60; P = .02).
Dr. Kornblum says that the study provides additional evidence for adding everolimus to anti-estrogen therapy in AI-resistant disease.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT SABCS 2016
Younger age, greater anxiety, affect pain after breast reconstruction
Younger age, bilateral procedure, and presurgery depression and anxiety are among the significant contributors to pain after breast reconstruction surgery, according to findings from a prospective study of 2,207 women who underwent several types of breast reconstruction procedures.
Although breast reconstruction is an important element of breast cancer treatment, and most acute postoperative pain resolves with time and pain management intervention, some patients suffer from persistent, severe postoperative pain that increases their risk for long-term pain and clinical morbidity, wrote Anita R. Kulkarni, MD, of Memorial Sloan-Kettering Cancer Center, New York, and her colleagues.
Overall, greater acute postoperative pain was significantly associated with younger age, bilateral procedures, higher levels of preoperative pain, and higher levels of preoperative anxiety and depression, the researchers said.
“Moreover, the degree of patients’ self-reported preoperative anxiety and depressive symptoms both appeared to bear a linear relationship with the magnitude of increased postoperative pain at 1 week” on the MPQ sensory pain rating, they noted.
Similarly, lower scores of physical well-being based on the BREAST-Q physical well-being scale were significantly associated with younger age, bilateral procedures, immediate reconstruction, and higher levels of preoperative pain, anxiety, and depression.
The average age of the women was 50 years, and 87% were white. Most patients (69%) had tissue expander (TE)/implant reconstruction procedures; 93% were immediate reconstruction; and 53% were bilateral reconstruction. The majority (90%) underwent surgery as part of breast cancer treatment.
“The comparative effect of procedure type on postoperative pain showed variable results across our multiple pain scales,” the researchers said.
The patients who underwent autologous flap reconstruction procedures – pedicled transverse rectus abdominis myocutaneous flap (PTRAMS), deep inferior epigastric perforator (DIEP), or superficial inferior epigastric artery (SIEA) – reported less-severe postoperative pain than did those who had TE/Implant procedures. Specifically, patients with SIEA and DIEP procedures reported significantly less pain 1 week after surgery than did those who had TE/Implant procedures, based on the MPQ sensory scale. In addition, patients with PTRAMS reported significantly less postoperative pain 1 week after surgery than did those who had TE/Implant procedures based on the NPRS scale. All three flap procedures were significantly associated with less-severe postoperative pain, compared with TE/Implant procedures.
The findings support data from previous studies that identified preoperative pain, mood disturbance, and age as factors for increased risk of acute postoperative pain, the researchers noted. In addition, “the findings have important implications for the identification of women who might be at risk for significant early postoperative pain following breast reconstruction, as acute postoperative pain is associated with poor functional outcomes, diminished quality of life, and the risk for development of persistent postsurgical pain,” they wrote.
The study results were limited by several factors, including the lack of standardization for postoperative pain treatment regimens, variability in the timing of preoperative assessment, and nonresponder bias, the researchers said.
However, “Once high-risk patients are identified, appropriate referrals can be considered to facilitate careful monitoring of postsurgical pain for selected patients,” they said. “Early intervention can give clinicians the opportunity to reduce postoperative morbidity and improve patients’ surgical experience and satisfaction with breast reconstruction outcome,” they added.
The researchers had no financial conflicts to disclose. The study was supported in part by a grant from the National Institutes of Health/National Cancer Institute.
Younger age, bilateral procedure, and presurgery depression and anxiety are among the significant contributors to pain after breast reconstruction surgery, according to findings from a prospective study of 2,207 women who underwent several types of breast reconstruction procedures.
Although breast reconstruction is an important element of breast cancer treatment, and most acute postoperative pain resolves with time and pain management intervention, some patients suffer from persistent, severe postoperative pain that increases their risk for long-term pain and clinical morbidity, wrote Anita R. Kulkarni, MD, of Memorial Sloan-Kettering Cancer Center, New York, and her colleagues.
Overall, greater acute postoperative pain was significantly associated with younger age, bilateral procedures, higher levels of preoperative pain, and higher levels of preoperative anxiety and depression, the researchers said.
“Moreover, the degree of patients’ self-reported preoperative anxiety and depressive symptoms both appeared to bear a linear relationship with the magnitude of increased postoperative pain at 1 week” on the MPQ sensory pain rating, they noted.
Similarly, lower scores of physical well-being based on the BREAST-Q physical well-being scale were significantly associated with younger age, bilateral procedures, immediate reconstruction, and higher levels of preoperative pain, anxiety, and depression.
The average age of the women was 50 years, and 87% were white. Most patients (69%) had tissue expander (TE)/implant reconstruction procedures; 93% were immediate reconstruction; and 53% were bilateral reconstruction. The majority (90%) underwent surgery as part of breast cancer treatment.
“The comparative effect of procedure type on postoperative pain showed variable results across our multiple pain scales,” the researchers said.
The patients who underwent autologous flap reconstruction procedures – pedicled transverse rectus abdominis myocutaneous flap (PTRAMS), deep inferior epigastric perforator (DIEP), or superficial inferior epigastric artery (SIEA) – reported less-severe postoperative pain than did those who had TE/Implant procedures. Specifically, patients with SIEA and DIEP procedures reported significantly less pain 1 week after surgery than did those who had TE/Implant procedures, based on the MPQ sensory scale. In addition, patients with PTRAMS reported significantly less postoperative pain 1 week after surgery than did those who had TE/Implant procedures based on the NPRS scale. All three flap procedures were significantly associated with less-severe postoperative pain, compared with TE/Implant procedures.
The findings support data from previous studies that identified preoperative pain, mood disturbance, and age as factors for increased risk of acute postoperative pain, the researchers noted. In addition, “the findings have important implications for the identification of women who might be at risk for significant early postoperative pain following breast reconstruction, as acute postoperative pain is associated with poor functional outcomes, diminished quality of life, and the risk for development of persistent postsurgical pain,” they wrote.
The study results were limited by several factors, including the lack of standardization for postoperative pain treatment regimens, variability in the timing of preoperative assessment, and nonresponder bias, the researchers said.
However, “Once high-risk patients are identified, appropriate referrals can be considered to facilitate careful monitoring of postsurgical pain for selected patients,” they said. “Early intervention can give clinicians the opportunity to reduce postoperative morbidity and improve patients’ surgical experience and satisfaction with breast reconstruction outcome,” they added.
The researchers had no financial conflicts to disclose. The study was supported in part by a grant from the National Institutes of Health/National Cancer Institute.
Younger age, bilateral procedure, and presurgery depression and anxiety are among the significant contributors to pain after breast reconstruction surgery, according to findings from a prospective study of 2,207 women who underwent several types of breast reconstruction procedures.
Although breast reconstruction is an important element of breast cancer treatment, and most acute postoperative pain resolves with time and pain management intervention, some patients suffer from persistent, severe postoperative pain that increases their risk for long-term pain and clinical morbidity, wrote Anita R. Kulkarni, MD, of Memorial Sloan-Kettering Cancer Center, New York, and her colleagues.
Overall, greater acute postoperative pain was significantly associated with younger age, bilateral procedures, higher levels of preoperative pain, and higher levels of preoperative anxiety and depression, the researchers said.
“Moreover, the degree of patients’ self-reported preoperative anxiety and depressive symptoms both appeared to bear a linear relationship with the magnitude of increased postoperative pain at 1 week” on the MPQ sensory pain rating, they noted.
Similarly, lower scores of physical well-being based on the BREAST-Q physical well-being scale were significantly associated with younger age, bilateral procedures, immediate reconstruction, and higher levels of preoperative pain, anxiety, and depression.
The average age of the women was 50 years, and 87% were white. Most patients (69%) had tissue expander (TE)/implant reconstruction procedures; 93% were immediate reconstruction; and 53% were bilateral reconstruction. The majority (90%) underwent surgery as part of breast cancer treatment.
“The comparative effect of procedure type on postoperative pain showed variable results across our multiple pain scales,” the researchers said.
The patients who underwent autologous flap reconstruction procedures – pedicled transverse rectus abdominis myocutaneous flap (PTRAMS), deep inferior epigastric perforator (DIEP), or superficial inferior epigastric artery (SIEA) – reported less-severe postoperative pain than did those who had TE/Implant procedures. Specifically, patients with SIEA and DIEP procedures reported significantly less pain 1 week after surgery than did those who had TE/Implant procedures, based on the MPQ sensory scale. In addition, patients with PTRAMS reported significantly less postoperative pain 1 week after surgery than did those who had TE/Implant procedures based on the NPRS scale. All three flap procedures were significantly associated with less-severe postoperative pain, compared with TE/Implant procedures.
The findings support data from previous studies that identified preoperative pain, mood disturbance, and age as factors for increased risk of acute postoperative pain, the researchers noted. In addition, “the findings have important implications for the identification of women who might be at risk for significant early postoperative pain following breast reconstruction, as acute postoperative pain is associated with poor functional outcomes, diminished quality of life, and the risk for development of persistent postsurgical pain,” they wrote.
The study results were limited by several factors, including the lack of standardization for postoperative pain treatment regimens, variability in the timing of preoperative assessment, and nonresponder bias, the researchers said.
However, “Once high-risk patients are identified, appropriate referrals can be considered to facilitate careful monitoring of postsurgical pain for selected patients,” they said. “Early intervention can give clinicians the opportunity to reduce postoperative morbidity and improve patients’ surgical experience and satisfaction with breast reconstruction outcome,” they added.
The researchers had no financial conflicts to disclose. The study was supported in part by a grant from the National Institutes of Health/National Cancer Institute.
FROM THE JOURNAL OF PLASTIC, RECONSTRUCTIVE & AESTHETIC SURGERY OPEN
Key clinical point: Identifying women at increased risk for severe acute postoperative pain allows clinicians the opportunity to reduce postoperative morbidity and improve patients’ surgical experience.
Major finding: Younger age (P = .002), bilateral procedures (P less than .0001), higher levels of preoperative pain (P less than .0001), and higher levels of preoperative anxiety (P less than .01) were among the strongest factors significantly associated with acute postoperative pain.
Data source: A prospective study of 2,207 women who underwent several types of breast reconstruction procedures.
Disclosures: The researchers had no financial conflicts to disclose. The study was supported in part by a grant from the National Institutes of Health/National Cancer Institute.
Halogenated anesthetic linked to less chronic postop mastectomy pain
The use of halogenated agents for anesthetic during a mastectomy operation may be associated with a lower incidence of long-term chronic postmastectomy pain (CPMP), according to a paper published in the the Journal of Clinical Anesthesia.
The retrospective cross-sectional survey of 128 women who underwent mastectomy with axillary lymph node dissection set out to determine whether the anesthetic or analgesic regimen used perioperatively had any impact on the risk of long-term chronic postoperative pain.
Overall, 43.8% of the women reported chronic pain, and nearly half of these showed neuropathic characteristics with an ID Pain score greater than or equal to 2 (J Clin Anesthesia. 2016;33:20-25. doi: 10.1016/j.jclinane.2015.07.010).
Those who were given a halogenated agent for anesthesia during the operation – 64% of patients in the survey - had a significant 19% lower incidence of chronic long-term postoperative mastectomy pain (95% CI, 0.70-0.95; P = .012).
Arnaud Steyaert, MD, and colleagues at the Catholic University of Louvain (Belgium) described this result as surprising, noting that sevoflurane use was recently found to be a risk factor for chronic pain after breast cancer surgery.
“An explanation for this discrepancy could be that the influence of sevoflurane on the development of CPMP depends on the other components of the anesthetic regimen,” the authors wrote, pointing out that the aforementioned study included the use of remifentanil in all patients, which can trigger acute opioid-induced hyperalgesia and chronic pain after surgery.
Apart from this effect, the authors said they did not see any impact from other analgesics – which included sufentanil, ketamine, clonidine, NSAIDs, and/or magnesium sulfate – on the risk of long-term chronic pain. However, patients treated with piritramide in the recovery room did have a significant 30% greater risk of chronic postoperative pain.
The study also found that patients who needed strong opioids in the postanesthesia care unit had a 30% higher risk of chronic long-term pain (95% CI, 1.11-1.53). “This was expected, as more intense acute postoperative pain is a known risk factor for developing chronic postsurgical pain, including CPMP,” the authors wrote.
Patients who had received adjuvant chemotherapy had a 32% higher incidence of chronic long-term pain, but there was no increase in risk associated with adjuvant radiotherapy. Both are known to cause neurotoxicity and therefore neuropathic pain, the authors commented.
No conflicts of interest were declared.
The use of halogenated agents for anesthetic during a mastectomy operation may be associated with a lower incidence of long-term chronic postmastectomy pain (CPMP), according to a paper published in the the Journal of Clinical Anesthesia.
The retrospective cross-sectional survey of 128 women who underwent mastectomy with axillary lymph node dissection set out to determine whether the anesthetic or analgesic regimen used perioperatively had any impact on the risk of long-term chronic postoperative pain.
Overall, 43.8% of the women reported chronic pain, and nearly half of these showed neuropathic characteristics with an ID Pain score greater than or equal to 2 (J Clin Anesthesia. 2016;33:20-25. doi: 10.1016/j.jclinane.2015.07.010).
Those who were given a halogenated agent for anesthesia during the operation – 64% of patients in the survey - had a significant 19% lower incidence of chronic long-term postoperative mastectomy pain (95% CI, 0.70-0.95; P = .012).
Arnaud Steyaert, MD, and colleagues at the Catholic University of Louvain (Belgium) described this result as surprising, noting that sevoflurane use was recently found to be a risk factor for chronic pain after breast cancer surgery.
“An explanation for this discrepancy could be that the influence of sevoflurane on the development of CPMP depends on the other components of the anesthetic regimen,” the authors wrote, pointing out that the aforementioned study included the use of remifentanil in all patients, which can trigger acute opioid-induced hyperalgesia and chronic pain after surgery.
Apart from this effect, the authors said they did not see any impact from other analgesics – which included sufentanil, ketamine, clonidine, NSAIDs, and/or magnesium sulfate – on the risk of long-term chronic pain. However, patients treated with piritramide in the recovery room did have a significant 30% greater risk of chronic postoperative pain.
The study also found that patients who needed strong opioids in the postanesthesia care unit had a 30% higher risk of chronic long-term pain (95% CI, 1.11-1.53). “This was expected, as more intense acute postoperative pain is a known risk factor for developing chronic postsurgical pain, including CPMP,” the authors wrote.
Patients who had received adjuvant chemotherapy had a 32% higher incidence of chronic long-term pain, but there was no increase in risk associated with adjuvant radiotherapy. Both are known to cause neurotoxicity and therefore neuropathic pain, the authors commented.
No conflicts of interest were declared.
The use of halogenated agents for anesthetic during a mastectomy operation may be associated with a lower incidence of long-term chronic postmastectomy pain (CPMP), according to a paper published in the the Journal of Clinical Anesthesia.
The retrospective cross-sectional survey of 128 women who underwent mastectomy with axillary lymph node dissection set out to determine whether the anesthetic or analgesic regimen used perioperatively had any impact on the risk of long-term chronic postoperative pain.
Overall, 43.8% of the women reported chronic pain, and nearly half of these showed neuropathic characteristics with an ID Pain score greater than or equal to 2 (J Clin Anesthesia. 2016;33:20-25. doi: 10.1016/j.jclinane.2015.07.010).
Those who were given a halogenated agent for anesthesia during the operation – 64% of patients in the survey - had a significant 19% lower incidence of chronic long-term postoperative mastectomy pain (95% CI, 0.70-0.95; P = .012).
Arnaud Steyaert, MD, and colleagues at the Catholic University of Louvain (Belgium) described this result as surprising, noting that sevoflurane use was recently found to be a risk factor for chronic pain after breast cancer surgery.
“An explanation for this discrepancy could be that the influence of sevoflurane on the development of CPMP depends on the other components of the anesthetic regimen,” the authors wrote, pointing out that the aforementioned study included the use of remifentanil in all patients, which can trigger acute opioid-induced hyperalgesia and chronic pain after surgery.
Apart from this effect, the authors said they did not see any impact from other analgesics – which included sufentanil, ketamine, clonidine, NSAIDs, and/or magnesium sulfate – on the risk of long-term chronic pain. However, patients treated with piritramide in the recovery room did have a significant 30% greater risk of chronic postoperative pain.
The study also found that patients who needed strong opioids in the postanesthesia care unit had a 30% higher risk of chronic long-term pain (95% CI, 1.11-1.53). “This was expected, as more intense acute postoperative pain is a known risk factor for developing chronic postsurgical pain, including CPMP,” the authors wrote.
Patients who had received adjuvant chemotherapy had a 32% higher incidence of chronic long-term pain, but there was no increase in risk associated with adjuvant radiotherapy. Both are known to cause neurotoxicity and therefore neuropathic pain, the authors commented.
No conflicts of interest were declared.
FROM THE JOURNAL OF CLINICAL ANESTHESIA
Key clinical point: The use of halogenated agents for anesthetic during a mastectomy operation may be associated with a lower incidence of long-term chronic postmastectomy pain.
Major finding: Patients given a halogenated agent for anesthesia during a mastectomy had a significant 19% lower incidence of chronic long-term postoperative mastectomy pain.
Data source: A retrospective cross-sectional survey.
Disclosures: No conflicts of interest were declared.
Biosimilar trastuzumab shows similar efficacy
A trastuzumab biosimilar drug has shown an equivalent response, compared with trastuzumab, in the treatment of ERBB2 (HER2)-positive metastatic breast cancer, according to the results of a randomized double-blind controlled trial.
The anti-ERBB2 humanized monoclonal antibody trastuzumab in combination with chemotherapy has been found in numerous trials to significantly improve progression-free survival and overall survival in women with ERBB2-positive metastatic breast, compared with chemotherapy alone.
“With impending patent expiration of some biological agents, development of biosimilars has become a high priority for drug developers and health authorities throughout the world to provide access to high-quality alternatives,” the authors wrote. “A biosimilar drug is a biological product that is highly similar to a licensed biological product, with no clinically meaningful differences in terms of safety, purity, or potency.”
In a phase III multicenter trial, 500 women with ERBB2 (HER2)-positive metastatic breast cancer, recruited from 95 sites in Europe, Africa, South America, and Asia, were randomized 1:1 to intravenous infusions of trastuzumab or a biosimilar labeled MYL-1401O, with both arms also receiving taxane therapy.
At 24 weeks, the overall response rate (ORR) was not significantly different between the biosimilar and trastuzumab groups (69.6% vs. 64.0%; ORR ratio, 1.09; 90% confidence interval, 0.974-1.211) and within the predefined equivalence boundaries, the investigators report (JAMA. 2016 Dec 1. doi: 10.1001/jama.2016.18305).
By week 48, both groups also showed no significant differences in time to tumor progression, progression-free survival (44.3% vs. 44.7%), or overall survival (89.1% vs. 85.1%).
Pharmacokinetic analysis showed the mean concentrations of trastuzumab were similar for the two treatments, and minimum drug concentrations were also comparable at week 16 of treatment.
“This confirmatory efficacy and safety study was the last step in the multistep process to demonstrate similarity of a trastuzumab biosimilar and was adequately powered to demonstrate equivalence with trastuzumab,” the authors wrote. “The results of this study are consistent with the physicochemical and functional similarity shown in vitro and in vivo and with the similar pharmacokinetics shown in healthy participants between the candidate biosimilar and trastuzumab.”
This consistency also extended to adverse events. Almost all participants in both the biosimilar and the trastuzumab groups reported at least one adverse event, which included neutropenia (57.5% vs. 53.3%), peripheral neuropathy (23.1% vs. 24.8%), and diarrhea (20.6% vs. 20.7%).
“A biosimilar treatment option may increase global access to biological cancer therapies, provided, among other issues, that the price of the biosimilar is sufficiently inexpensive to enable women in non–high-income countries to access this therapy,” the authors wrote.
However, they pointed out that the stepwise development program for biosimilar drugs tended to use shorter end-points – 24 weeks for the primary endpoint and 48 weeks for secondary endpoints in this particular study. By 48 weeks, more than 50% of patients had not shown progression, suggesting that the medians for efficacy parameters may have been longer with a longer data cut-off.
“The choice of the 24-week evaluation period for part 1 of this study was related to the ability to analyze the ORR as a short-term measure of clinical activity and safety directly related to the combination of taxanes with trastuzumab and the proposed biosimilar as first-line treatment.”
The study was funded and sponsored by Mylan, which manufactured the biosimilar drug, and Biocon Research Limited. Four authors declared stock in Mylan, two declared consulting fees from Mylan and one also declared stock in Biocon Research Limited. One author declared research and travel support from other pharmaceutical companies.
The availability of a biosimilar agent that achieves equivalent clinical outcomes at lower cost could enable many patients with breast cancer to have access to a therapy that may improve survival. Moreover, given the large number of patients with breast cancer, widespread use of this trastuzumab biosimilar evaluated by Rugo et al. (if approved for use by the U.S. FDA, the European Medicines Agency, and other regulatory agencies) also could have financial implications for the manufacturer of this product.
In announcing their FDA submission for the proposed trastuzumab biosimilar, the sponsors of the trial by Rugo et al. have expressed their “shared commitment to increasing access to these critical medicines worldwide” and indicated that “this advancement in the U.S. will enable us to enhance access to this affordable therapy to larger patient pools.” Ultimately, to fulfill these pledges, the manufacturers must ensure that the pricing of this biosimilar product is responsible and fair and provides access to this important therapy at an affordable price.
Howard Bauchner, MD, is editor in chief of JAMA, Phil B. Fontanarosa, MD, MBA, is executive editor of JAMA, and Robert M. Golub, MD, is deputy editor of JAMA. These comments are taken from an accompanying editorial (JAMA. 2016 Dec 1. doi: 10.1001/jama.2016.18743). No conflicts of interest were declared.
The availability of a biosimilar agent that achieves equivalent clinical outcomes at lower cost could enable many patients with breast cancer to have access to a therapy that may improve survival. Moreover, given the large number of patients with breast cancer, widespread use of this trastuzumab biosimilar evaluated by Rugo et al. (if approved for use by the U.S. FDA, the European Medicines Agency, and other regulatory agencies) also could have financial implications for the manufacturer of this product.
In announcing their FDA submission for the proposed trastuzumab biosimilar, the sponsors of the trial by Rugo et al. have expressed their “shared commitment to increasing access to these critical medicines worldwide” and indicated that “this advancement in the U.S. will enable us to enhance access to this affordable therapy to larger patient pools.” Ultimately, to fulfill these pledges, the manufacturers must ensure that the pricing of this biosimilar product is responsible and fair and provides access to this important therapy at an affordable price.
Howard Bauchner, MD, is editor in chief of JAMA, Phil B. Fontanarosa, MD, MBA, is executive editor of JAMA, and Robert M. Golub, MD, is deputy editor of JAMA. These comments are taken from an accompanying editorial (JAMA. 2016 Dec 1. doi: 10.1001/jama.2016.18743). No conflicts of interest were declared.
The availability of a biosimilar agent that achieves equivalent clinical outcomes at lower cost could enable many patients with breast cancer to have access to a therapy that may improve survival. Moreover, given the large number of patients with breast cancer, widespread use of this trastuzumab biosimilar evaluated by Rugo et al. (if approved for use by the U.S. FDA, the European Medicines Agency, and other regulatory agencies) also could have financial implications for the manufacturer of this product.
In announcing their FDA submission for the proposed trastuzumab biosimilar, the sponsors of the trial by Rugo et al. have expressed their “shared commitment to increasing access to these critical medicines worldwide” and indicated that “this advancement in the U.S. will enable us to enhance access to this affordable therapy to larger patient pools.” Ultimately, to fulfill these pledges, the manufacturers must ensure that the pricing of this biosimilar product is responsible and fair and provides access to this important therapy at an affordable price.
Howard Bauchner, MD, is editor in chief of JAMA, Phil B. Fontanarosa, MD, MBA, is executive editor of JAMA, and Robert M. Golub, MD, is deputy editor of JAMA. These comments are taken from an accompanying editorial (JAMA. 2016 Dec 1. doi: 10.1001/jama.2016.18743). No conflicts of interest were declared.
A trastuzumab biosimilar drug has shown an equivalent response, compared with trastuzumab, in the treatment of ERBB2 (HER2)-positive metastatic breast cancer, according to the results of a randomized double-blind controlled trial.
The anti-ERBB2 humanized monoclonal antibody trastuzumab in combination with chemotherapy has been found in numerous trials to significantly improve progression-free survival and overall survival in women with ERBB2-positive metastatic breast, compared with chemotherapy alone.
“With impending patent expiration of some biological agents, development of biosimilars has become a high priority for drug developers and health authorities throughout the world to provide access to high-quality alternatives,” the authors wrote. “A biosimilar drug is a biological product that is highly similar to a licensed biological product, with no clinically meaningful differences in terms of safety, purity, or potency.”
In a phase III multicenter trial, 500 women with ERBB2 (HER2)-positive metastatic breast cancer, recruited from 95 sites in Europe, Africa, South America, and Asia, were randomized 1:1 to intravenous infusions of trastuzumab or a biosimilar labeled MYL-1401O, with both arms also receiving taxane therapy.
At 24 weeks, the overall response rate (ORR) was not significantly different between the biosimilar and trastuzumab groups (69.6% vs. 64.0%; ORR ratio, 1.09; 90% confidence interval, 0.974-1.211) and within the predefined equivalence boundaries, the investigators report (JAMA. 2016 Dec 1. doi: 10.1001/jama.2016.18305).
By week 48, both groups also showed no significant differences in time to tumor progression, progression-free survival (44.3% vs. 44.7%), or overall survival (89.1% vs. 85.1%).
Pharmacokinetic analysis showed the mean concentrations of trastuzumab were similar for the two treatments, and minimum drug concentrations were also comparable at week 16 of treatment.
“This confirmatory efficacy and safety study was the last step in the multistep process to demonstrate similarity of a trastuzumab biosimilar and was adequately powered to demonstrate equivalence with trastuzumab,” the authors wrote. “The results of this study are consistent with the physicochemical and functional similarity shown in vitro and in vivo and with the similar pharmacokinetics shown in healthy participants between the candidate biosimilar and trastuzumab.”
This consistency also extended to adverse events. Almost all participants in both the biosimilar and the trastuzumab groups reported at least one adverse event, which included neutropenia (57.5% vs. 53.3%), peripheral neuropathy (23.1% vs. 24.8%), and diarrhea (20.6% vs. 20.7%).
“A biosimilar treatment option may increase global access to biological cancer therapies, provided, among other issues, that the price of the biosimilar is sufficiently inexpensive to enable women in non–high-income countries to access this therapy,” the authors wrote.
However, they pointed out that the stepwise development program for biosimilar drugs tended to use shorter end-points – 24 weeks for the primary endpoint and 48 weeks for secondary endpoints in this particular study. By 48 weeks, more than 50% of patients had not shown progression, suggesting that the medians for efficacy parameters may have been longer with a longer data cut-off.
“The choice of the 24-week evaluation period for part 1 of this study was related to the ability to analyze the ORR as a short-term measure of clinical activity and safety directly related to the combination of taxanes with trastuzumab and the proposed biosimilar as first-line treatment.”
The study was funded and sponsored by Mylan, which manufactured the biosimilar drug, and Biocon Research Limited. Four authors declared stock in Mylan, two declared consulting fees from Mylan and one also declared stock in Biocon Research Limited. One author declared research and travel support from other pharmaceutical companies.
A trastuzumab biosimilar drug has shown an equivalent response, compared with trastuzumab, in the treatment of ERBB2 (HER2)-positive metastatic breast cancer, according to the results of a randomized double-blind controlled trial.
The anti-ERBB2 humanized monoclonal antibody trastuzumab in combination with chemotherapy has been found in numerous trials to significantly improve progression-free survival and overall survival in women with ERBB2-positive metastatic breast, compared with chemotherapy alone.
“With impending patent expiration of some biological agents, development of biosimilars has become a high priority for drug developers and health authorities throughout the world to provide access to high-quality alternatives,” the authors wrote. “A biosimilar drug is a biological product that is highly similar to a licensed biological product, with no clinically meaningful differences in terms of safety, purity, or potency.”
In a phase III multicenter trial, 500 women with ERBB2 (HER2)-positive metastatic breast cancer, recruited from 95 sites in Europe, Africa, South America, and Asia, were randomized 1:1 to intravenous infusions of trastuzumab or a biosimilar labeled MYL-1401O, with both arms also receiving taxane therapy.
At 24 weeks, the overall response rate (ORR) was not significantly different between the biosimilar and trastuzumab groups (69.6% vs. 64.0%; ORR ratio, 1.09; 90% confidence interval, 0.974-1.211) and within the predefined equivalence boundaries, the investigators report (JAMA. 2016 Dec 1. doi: 10.1001/jama.2016.18305).
By week 48, both groups also showed no significant differences in time to tumor progression, progression-free survival (44.3% vs. 44.7%), or overall survival (89.1% vs. 85.1%).
Pharmacokinetic analysis showed the mean concentrations of trastuzumab were similar for the two treatments, and minimum drug concentrations were also comparable at week 16 of treatment.
“This confirmatory efficacy and safety study was the last step in the multistep process to demonstrate similarity of a trastuzumab biosimilar and was adequately powered to demonstrate equivalence with trastuzumab,” the authors wrote. “The results of this study are consistent with the physicochemical and functional similarity shown in vitro and in vivo and with the similar pharmacokinetics shown in healthy participants between the candidate biosimilar and trastuzumab.”
This consistency also extended to adverse events. Almost all participants in both the biosimilar and the trastuzumab groups reported at least one adverse event, which included neutropenia (57.5% vs. 53.3%), peripheral neuropathy (23.1% vs. 24.8%), and diarrhea (20.6% vs. 20.7%).
“A biosimilar treatment option may increase global access to biological cancer therapies, provided, among other issues, that the price of the biosimilar is sufficiently inexpensive to enable women in non–high-income countries to access this therapy,” the authors wrote.
However, they pointed out that the stepwise development program for biosimilar drugs tended to use shorter end-points – 24 weeks for the primary endpoint and 48 weeks for secondary endpoints in this particular study. By 48 weeks, more than 50% of patients had not shown progression, suggesting that the medians for efficacy parameters may have been longer with a longer data cut-off.
“The choice of the 24-week evaluation period for part 1 of this study was related to the ability to analyze the ORR as a short-term measure of clinical activity and safety directly related to the combination of taxanes with trastuzumab and the proposed biosimilar as first-line treatment.”
The study was funded and sponsored by Mylan, which manufactured the biosimilar drug, and Biocon Research Limited. Four authors declared stock in Mylan, two declared consulting fees from Mylan and one also declared stock in Biocon Research Limited. One author declared research and travel support from other pharmaceutical companies.
FROM JAMA
Key clinical point: A trastuzumab biosimilar drug has shown an equivalent response, compared with trastuzumab, in the treatment of ERBB2 (HER2)-positive metastatic breast cancer.
Major finding: Patients treated with biosimilar trastuzumab showed no significant differences in response rate, progression, and survival, compared with those treated with trastuzumab.
Data source: Randomized, double-blind phase III controlled trial in 500 women with ERBB2 (HER2)-positive metastatic breast cancer.
Disclosures: The study was funded and sponsored by Mylan, which manufactured the biosimilar drug, and Biocon Research Limited. Four authors declared stock in Mylan, two declared consulting fees from Mylan, and one also declared stock in Biocon Research Limited. One author declared research and travel support from other pharmaceutical companies.
SABCS 2016: Top picks from Dr. Hope S. Rugo and Dr. William J. Gradishar
Oncology Practice associate editors Dr. Hope S. Rugo and Dr. William J. Gradishar reveal several anticipated studies from the 39th annual San Antonio Breast Cancer Symposium, set to begin on Wednesday, Dec. 7:
• S1-02. PrECOG 0102. A randomized, double-blind, phase II trial of fulvestrant plus everolimus or placebo in postmenopausal women with hormone receptor (HR)–positive, human epidermal growth factor receptor 2 (HER2)–negative metastatic breast cancer resistant to aromatase inhibitor therapy
• S1-03. First results from the multicenter phase III DATA study comparing 3 vs. 6 years of anastrozole after 2-3 years of tamoxifen in postmenopausal women with HR–positive early breast cancer.
• S1-04. Optimal duration of extended letrozole treatment after 5 years of adjuvant endocrine therapy; results of the randomized phase III IDEAL trial (BOOG 2006-05).
• S1-05. A randomized, double-blind, placebo-controlled clinical trial to evaluate extended adjuvant endocrine therapy (5 years of letrozole) in postmenopausal women with HR–positive breast cancer who have completed previous adjuvant endocrine therapy: initial results of NRG oncology/NSABP B-42.
• S1-08. Prognostic associations of tumor-infiltrating lymphocytes in metastatic HER2-positive breast cancer treated with trastuzumab and pertuzumab: a secondary analysis of the CLEOPATRA study.
• S2-03. Does BRCA status affect outcome in young breast cancer patients? Results from the Prospective Study of Outcomes in Sporadic versus Hereditary Breast Cancer (POSH).
• S2-05. Efficacy and tolerability of veliparib (ABT-888) in combination with carboplatin (C) and paclitaxel (P) vs. placebo (Plc)+C/P in patients with BRCA1 or BRCA2 mutations and metastatic breast cancer: a randomized, phase II study.
• S2-06. DNA repair deficiency biomarkers and MammaPrint High1/(ultra)High2 risk as predictors of veliparib/carboplatin response: results from the neoadjuvant I-SPY 2 trial for high-risk breast cancer.
• S3-02. Plasma microRNA levels for predicting therapeutic response to neoadjuvant treatment in HER2-positive breast cancer: results from Neo-ALTTO.
• S3-04. Primary analysis of PERTAIN: a randomized, two-arm, open-label, multicenter phase II trial assessing the efficacy and safety of pertuzumab given in combination with trastuzumab plus an aromatase inhibitor in first-line patients with HER2-positive and HR–positive metastatic or locally advanced breast cancer.
• S4-06. Biologic effects of abemaciclib in a phase II neoadjuvant study for postmenopausal patients with HR–positive, HER2-negative breast cancer.
• S4-07. BELLE-3: a phase III study of buparlisib + fulvestrant in postmenopausal women with HR-positive, HER2-negative, aromatase inhibitor–treated locally advanced or metastatic breast cancer, who progressed on or after mTOR inhibitor–based treatment.
• S5-02. Scalp Cooling Alopecia Prevention (SCALP) trial for patients with early-stage breast cancer.
Dr. Rugo is professor of medicine, University of California, San Francisco, and director, breast oncology and clinical trials education, UCSF Helen Diller Family Comprehensive Cancer Center. Dr. Gradishar is the Betsy Bramsen Professor of Breast Oncology, professor of medicine, and deputy chief in the division of hematology/oncology, Feinberg School of Medicine, Chicago; and deputy director for the Clinical Network and director of the Maggie Daley Center for Women’s Cancer Care, Robert H. Lurie Comprehensive Cancer Center Network of Northwestern University, Chicago.
Oncology Practice associate editors Dr. Hope S. Rugo and Dr. William J. Gradishar reveal several anticipated studies from the 39th annual San Antonio Breast Cancer Symposium, set to begin on Wednesday, Dec. 7:
• S1-02. PrECOG 0102. A randomized, double-blind, phase II trial of fulvestrant plus everolimus or placebo in postmenopausal women with hormone receptor (HR)–positive, human epidermal growth factor receptor 2 (HER2)–negative metastatic breast cancer resistant to aromatase inhibitor therapy
• S1-03. First results from the multicenter phase III DATA study comparing 3 vs. 6 years of anastrozole after 2-3 years of tamoxifen in postmenopausal women with HR–positive early breast cancer.
• S1-04. Optimal duration of extended letrozole treatment after 5 years of adjuvant endocrine therapy; results of the randomized phase III IDEAL trial (BOOG 2006-05).
• S1-05. A randomized, double-blind, placebo-controlled clinical trial to evaluate extended adjuvant endocrine therapy (5 years of letrozole) in postmenopausal women with HR–positive breast cancer who have completed previous adjuvant endocrine therapy: initial results of NRG oncology/NSABP B-42.
• S1-08. Prognostic associations of tumor-infiltrating lymphocytes in metastatic HER2-positive breast cancer treated with trastuzumab and pertuzumab: a secondary analysis of the CLEOPATRA study.
• S2-03. Does BRCA status affect outcome in young breast cancer patients? Results from the Prospective Study of Outcomes in Sporadic versus Hereditary Breast Cancer (POSH).
• S2-05. Efficacy and tolerability of veliparib (ABT-888) in combination with carboplatin (C) and paclitaxel (P) vs. placebo (Plc)+C/P in patients with BRCA1 or BRCA2 mutations and metastatic breast cancer: a randomized, phase II study.
• S2-06. DNA repair deficiency biomarkers and MammaPrint High1/(ultra)High2 risk as predictors of veliparib/carboplatin response: results from the neoadjuvant I-SPY 2 trial for high-risk breast cancer.
• S3-02. Plasma microRNA levels for predicting therapeutic response to neoadjuvant treatment in HER2-positive breast cancer: results from Neo-ALTTO.
• S3-04. Primary analysis of PERTAIN: a randomized, two-arm, open-label, multicenter phase II trial assessing the efficacy and safety of pertuzumab given in combination with trastuzumab plus an aromatase inhibitor in first-line patients with HER2-positive and HR–positive metastatic or locally advanced breast cancer.
• S4-06. Biologic effects of abemaciclib in a phase II neoadjuvant study for postmenopausal patients with HR–positive, HER2-negative breast cancer.
• S4-07. BELLE-3: a phase III study of buparlisib + fulvestrant in postmenopausal women with HR-positive, HER2-negative, aromatase inhibitor–treated locally advanced or metastatic breast cancer, who progressed on or after mTOR inhibitor–based treatment.
• S5-02. Scalp Cooling Alopecia Prevention (SCALP) trial for patients with early-stage breast cancer.
Dr. Rugo is professor of medicine, University of California, San Francisco, and director, breast oncology and clinical trials education, UCSF Helen Diller Family Comprehensive Cancer Center. Dr. Gradishar is the Betsy Bramsen Professor of Breast Oncology, professor of medicine, and deputy chief in the division of hematology/oncology, Feinberg School of Medicine, Chicago; and deputy director for the Clinical Network and director of the Maggie Daley Center for Women’s Cancer Care, Robert H. Lurie Comprehensive Cancer Center Network of Northwestern University, Chicago.
Oncology Practice associate editors Dr. Hope S. Rugo and Dr. William J. Gradishar reveal several anticipated studies from the 39th annual San Antonio Breast Cancer Symposium, set to begin on Wednesday, Dec. 7:
• S1-02. PrECOG 0102. A randomized, double-blind, phase II trial of fulvestrant plus everolimus or placebo in postmenopausal women with hormone receptor (HR)–positive, human epidermal growth factor receptor 2 (HER2)–negative metastatic breast cancer resistant to aromatase inhibitor therapy
• S1-03. First results from the multicenter phase III DATA study comparing 3 vs. 6 years of anastrozole after 2-3 years of tamoxifen in postmenopausal women with HR–positive early breast cancer.
• S1-04. Optimal duration of extended letrozole treatment after 5 years of adjuvant endocrine therapy; results of the randomized phase III IDEAL trial (BOOG 2006-05).
• S1-05. A randomized, double-blind, placebo-controlled clinical trial to evaluate extended adjuvant endocrine therapy (5 years of letrozole) in postmenopausal women with HR–positive breast cancer who have completed previous adjuvant endocrine therapy: initial results of NRG oncology/NSABP B-42.
• S1-08. Prognostic associations of tumor-infiltrating lymphocytes in metastatic HER2-positive breast cancer treated with trastuzumab and pertuzumab: a secondary analysis of the CLEOPATRA study.
• S2-03. Does BRCA status affect outcome in young breast cancer patients? Results from the Prospective Study of Outcomes in Sporadic versus Hereditary Breast Cancer (POSH).
• S2-05. Efficacy and tolerability of veliparib (ABT-888) in combination with carboplatin (C) and paclitaxel (P) vs. placebo (Plc)+C/P in patients with BRCA1 or BRCA2 mutations and metastatic breast cancer: a randomized, phase II study.
• S2-06. DNA repair deficiency biomarkers and MammaPrint High1/(ultra)High2 risk as predictors of veliparib/carboplatin response: results from the neoadjuvant I-SPY 2 trial for high-risk breast cancer.
• S3-02. Plasma microRNA levels for predicting therapeutic response to neoadjuvant treatment in HER2-positive breast cancer: results from Neo-ALTTO.
• S3-04. Primary analysis of PERTAIN: a randomized, two-arm, open-label, multicenter phase II trial assessing the efficacy and safety of pertuzumab given in combination with trastuzumab plus an aromatase inhibitor in first-line patients with HER2-positive and HR–positive metastatic or locally advanced breast cancer.
• S4-06. Biologic effects of abemaciclib in a phase II neoadjuvant study for postmenopausal patients with HR–positive, HER2-negative breast cancer.
• S4-07. BELLE-3: a phase III study of buparlisib + fulvestrant in postmenopausal women with HR-positive, HER2-negative, aromatase inhibitor–treated locally advanced or metastatic breast cancer, who progressed on or after mTOR inhibitor–based treatment.
• S5-02. Scalp Cooling Alopecia Prevention (SCALP) trial for patients with early-stage breast cancer.
Dr. Rugo is professor of medicine, University of California, San Francisco, and director, breast oncology and clinical trials education, UCSF Helen Diller Family Comprehensive Cancer Center. Dr. Gradishar is the Betsy Bramsen Professor of Breast Oncology, professor of medicine, and deputy chief in the division of hematology/oncology, Feinberg School of Medicine, Chicago; and deputy director for the Clinical Network and director of the Maggie Daley Center for Women’s Cancer Care, Robert H. Lurie Comprehensive Cancer Center Network of Northwestern University, Chicago.
FROM SABCS 2016
Patient with a breast mass: Why did she pursue litigation?
CASE: After routine mammography results, DCIS found
A 49-year-old woman (G2 P2002) with a history of fibrocystic breast disease presented with a left breast mass that she found a month ago on self-examination. The patient faithfully had obtained routine mammograms since age 40. This year, after reporting the mass and with spot films obtained as recommended by the radiologist, a new cluster of microcalcifications was identified on the report: “spot compression” assessment identified a 3-cm mass and noted “s/p breast augmentation.”
The radiologist interpreted the spot films to be benign. His report stated that “15% of breast cancers are not detected by mammogram and breast self-exam is recommended monthly from 40 years of age.”
The gynecologist recommended a 6-month follow up. When the patient complied, the radiologist’s report again noted calcifications believed to be nonmalignant. Six months later, the patient presented with bloody nipple discharge from her left breast with apparent “eczema-like” lesions on the areola. The patient noted that her “left implant felt different.”
The patient’s surgical history included breast augmentation “years ago.” Her family history was negative for breast cancer. Her medications included hormone therapy (conjugated estrogens 0.625 mg with medroxyprogesterone acetate 2.5 mg daily) for vaginal atrophy. Other medical conditions included irritable bowel syndrome (managed with diet), anxiety and mood swings (for which she was taking sertraline), decreased libido, and irregular vaginal bleeding (after the patient refused endometrial sampling, she was switched to oral contraceptives to address the problem). In addition, her hypertension was being treated with hydrochlorothiazide.
At the gynecologist’s suggestion, a dermatology consultation was obtained.
The dermatologist gave a diagnosis of Paget disease with high-grade ductal carcinoma-in-situ (DCIS). The interval from screening mammogram to DCIS diagnosis had been 8 months. The dermatologist referred the patient to a breast surgeon. A discussion ensued between the breast surgeon and the dermatologist concerning the difficulty of making a diagnosis of breast cancer in a woman with breast augmentation.
The patient underwent modified radical mastectomy, and histopathology revealed DCIS with clear margins; lymph nodes were negative.
The patient filed a malpractice suit against the gynecologist related to the delayed breast mass evaluation and management. She remained upset that when she called the gynecologist’s office to convey her concerns regarding the left nipple discharge and implant concerns, “she was blown off.” She felt there was a clear “failure to communicate on critical matters of her health.” She alleged that the gynecologist, not the dermatologist, should have referred her to a breast surgeon.
WHAT 'S THE VERDICT?
In the end, the patient decided not to pursue the lawsuit.
Related article:
Who is liable when a surgical error occurs?
Medical considerations
Breast cancer is the most common female malignancy, with 232,340 cases occurring annually in the United States. It is the second leading cause of cancer-related death in US women.1 For this case discussion, we review the role of breast cancer screening, including breast self-examination and mammography.
Is breast self-examination recommended?
Recently, medical care has evolved from “breast self-examination” (BSE) to “breast self-awareness.”2 The concept of BSE and concerns about it stem in part from “the Shanghai study.”3 In this prospective randomized trial, 266,064 female textile workers were randomly assigned to “rigorous and repetitive training in BSE” versus no instruction and no BSE performance. The former group had twice as many breast biopsies than the latter group (2,761 biopsies in the BSE group vs 1,505 in the control group). There was no difference in the number of breast cancers diagnosed among the groups—864 in the BSE group and 896 in the control group (relative risk [RR], 0.97; 95% confidence interval [CI], 0.88–1.06; P = .47). Other studies also support lack of efficacy regarding BSE.4
The potential for psychological harm, unnecessary biopsy, and additional imaging in association with false-positive findings is a concern2 (TABLE). The American College of Obstetricians and Gynecologists (ACOG) states, “breast self-examination may be appropriate for certain high-risk populations and for other women who chose to follow this approach.”2,5,6 The US Preventive Services Task Force (USPSTF) guidelines say that there is “insufficient evidence to assess risks vs benefits, including harms,” and recommends against teaching BSE.7 The American Cancer Society puts BSE in the “optional” category.2,8
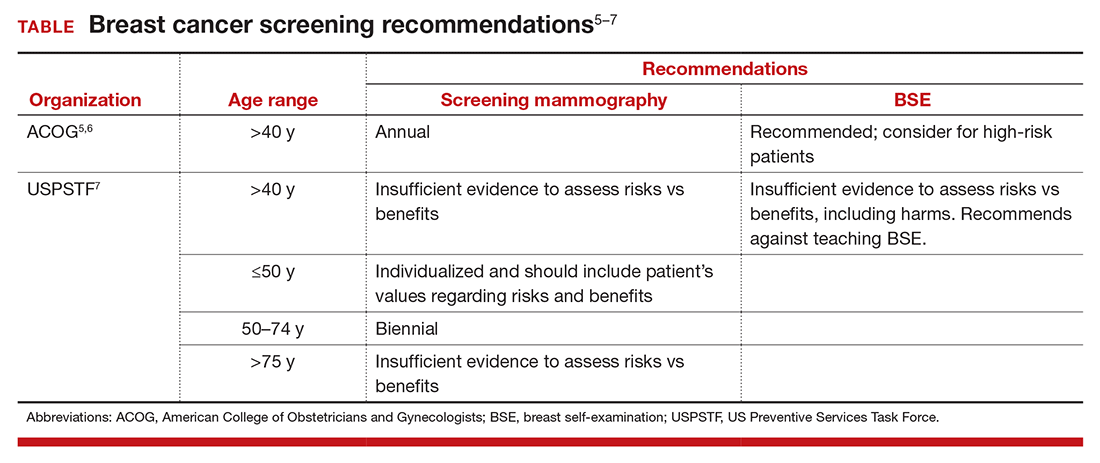
Is there a middle of the road strategy? Perhaps. The concept of breast-awareness was developed so that women understand how their breasts look and feel.2,5 The concept does not advocate monthly BSE. The Mayo Clinic reported that, of 592 breast cancers, 57% were detected following abnormal screening mammography, 30% by BSE and 14% by clinical examination by a clinician. Furthermore, 38% of women with a palpable abnormality had a normal mammogram within the preceding 13 months.9 McBride and colleagues aptly addressed this: “Healthcare providers can educate their patients that breast awareness, in essence, is a two-step process. First, it requires that women be familiar with their breasts and aware of new changes and, second, have an understanding of the implications of these changes which includes informing their health care provider promptly.”10 The concept of “know what is normal for you” as conveyed by The Susan G. Komen Foundation succinctly encourages communication with patients.2,11
Mammography
The latest technique in mammography is digital or 3D mammography, also known as tomosynthesis. The technique is similar to 2D mammography with the addition of digital cameras. A study published in the radiology literature noted that the 2 methods were equivalent.12 One possible advantage of 3D mammography is that the 3D images are stored in computer files and are more easily incorporated into the electronic medical record.
What about mammography after breast augmentation?
While breast augmentation is not associated with an increase in breast cancer,13 mammography following breast augmentation can be more difficult to interpret and may result in a delay in diagnosis. In a prospective study of asymptomatic women who were diagnosed with breast cancer, 137 had augmentation and 685 did not. Miglioretti and colleagues noted that the sensitivity of screening mammography was lower in the augmentation cohort.14 To enhance accuracy, breast implant displacement views (in which the breast tissue is pulled forward and the implant is displaced posteriorly to improve visualization) have been recommended.15 A retrospective review provides data reporting no effect on interpretation of mammograms following augmentation.16 The American Cancer Society recommends the same screening for women with implants as without implants, starting at age 40 years.17
Paget disease of the breast
Paget disease of the breast was first described by Sir James Paget in 1874. He also defined Paget disease of extramammary tissue, bone, vulva, and penis. Paget disease of the breast is a rare type of cancer in which the skin and nipple are involved frequently in association with DCIS or invasive breast cancer. The skin has an eczema-like appearance. Characteristic Paget (malignant) cells are large with clear cytoplasm (clear halo) and eccentric, hyperchromatic nuclei throughout the dermis. Assessment includes mammography and biopsy with immunohistochemical staining. Treatment varies by case and can include lumpectomy or mastectomy and chemotherapy and/or radiation therapy. Medications, including tamoxifen and anastrazole, have been recommended. Prognosis depends on nodal involvement. The disease is more common in women older than age 50.18
Related article:
The medicolegal considerations of interacting with your patients online
Legal issues: What was the gynecologist’s obligation?
The question remains, did the gynecologist have an obligation to obtain diagnostic mammography and ultrasound of the breast? Would it have been prudent for immediate referral to a breast surgeon? These are critical questions.
Negligence and the standard of care
The malpractice lawsuit against this gynecologist is based on negligence. In essence, it is a claim that the management provided fell below a standard of care that would be given by a reasonably careful and prudent gynecologist under the circumstances. This generally means that the care was less than the profession itself would find acceptable. Here the claim is essentially a diagnostic error claim, a common basis of malpractice.19 The delay in diagnosing breast cancer is a leading cause of malpractice claims against gynecologists.20,21
It is axiomatic that not all bad outcomes are the result of error, and not all errors are a result of an unacceptable standard of care. It is only bad outcomes resulting from careless or negligent errors that give rise to malpractice. But malpractice claims are often filed without knowing whether or not there was negligence—and, as we will see, many of those claims are without merit.
Was there negligence?
Ordinarily the plaintiff/patient must demonstrate that the care by the physician fell below the standard of care and, as a result, the patient suffered an injury. Stated another way, the plaintiff must show that the physician’s actions were unreasonable given all of the circumstances. In this case, the plaintiff must demonstrate that the gynecologist’s care was not appropriate. Failure to refer to another physician or provide additional testing is likely to be the major claim for negligence or inappropriate care.
Did that negligence cause injury?
Even if the plaintiff can demonstrate that the care was negligent, to prevail the plaintiff must also demonstrate that that negligence caused the injury—and that might be difficult.
What injury was caused by the delay in discovering the cancer? It did not apparently lead to the patient’s death. Can it be proven that the delay clearly shortened the patient’s life expectancy or required additional expensive and painful treatment? That may be difficult to demonstrate.
Causation. The causation factor could appear in many cancer or similar medical cases. On one hand, causation is a critical matter, but on the other hand, delay in treating cancer might have a very adverse effect on patients.
In recent years, about half the states have solved this dilemma by recognizing the concept of “loss of chance.”22 Essentially, this means that the health care provider, by delaying treatment, diminished the possibility that the patient would survive or recover fully.
There are significant variations among states in the loss-of-chance concept, many being quite technical.22 Thus, it is possible that a delay that reduced the chance of recovery or survival could be the injury and causal connection between the injury and the negligence. Even in states that recognize the loss-of-chance concept, the patient still must prove loss of chance.
Lessons learned from this case study
- Breast self-awareness has replaced (substituted) breast self-examination
- ACOG recommends breast mammography beginning at age 40 years
- Breast augmentation affects mammographic interpretation
- Prehaps if better communication had been provided initially, the patient would not have sought legal counsel or filed a weak suit
Related article:
Is the smartphone recording while the patient is under anesthesia?
Is this a strong malpractice case?
In the case presented here, it is not clear that the patient could show a meaningful loss of chance. If there is a delay in the breast cancer diagnosis, tumor doubling time would be an issue. While it is impossible to assess growth rate when a breast cancer is in its preclinical microscopic stage, doubling time can be 100 to 200 days. Therefore, it would take 20 years for the tumor to reach a 1- to 2-cm diameter. A log-normal distribution has been suggested for determining tumor growth.23
Although the facts in this case are sketchy, this does not look like a strong malpractice case. Given the expense, difficulty, and length of time it takes to pursue a malpractice case (especially for someone battling cancer), an obvious question is: Why would a patient file a lawsuit in these circumstances? There is no single answer to that, but the hope of getting rich is unlikely a primary motivation. Ironically, many malpractice cases are filed in which there was no error (or at least no negligent error).24
The search for what really happened, or why the bad event happened, is key. In other circumstances, it may be a desire for revenge or to protect other patients from similar bad results. Studies repeatedly have shown a somewhat limited correlation between negligent error and the decision to file a malpractice claim.24–26 In this case, the patient’s sense of being “blown off” during a particularly difficult time may represent the reason why she filed a malpractice lawsuit. Communication gaffes and poor physician-patient relationships undoubtedly contribute to medical malpractice claims.27,28 Improving communication with patients probably improves care, but it also almost certainly reduces the risk of a malpractice claim.29
Why a lawyer would accept this case is also unclear, but that is an issue for another day. Also for another day is the issue of product liability concerning breast implants. Those legal issues and related liability, primarily directed to the manufacturers of the implants, are interesting topics. They are also complex and will be the subject of a future article.
Finally, the patient’s decision to not pursue her lawsuit does not come as a surprise. A relatively small percentage of malpractice claims result in any meaningful financial recovery for the plaintiff. Few cases go to trial, and of those that do result in a verdict, about 75% of the verdicts are in favor of the physician.25,30 Many cases just fade away, either because the plaintiff never pursues them or because they are dismissed by a court at an early stage. Nonetheless, for the physician, even winning a malpractice case is disruptive and difficult. So in addition to ensuring careful, quality, and up-to-date care, a physician should seek to maintain good relationships and communication with patients to reduce the probability of even weak lawsuits being filed.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63(1):11–30.
- Mark K, Temkin S, Terplan M. Breast self-awareness: the evidence behind the euphemism. Obstet Gynecol. 2014;123(4):734–745.
- Thomas D, Gao D, Self S, et al. Randomized trial of breast self-examination in Shanghai: methodology and preliminary results. J Natl Cancer Inst. 1997;89(5):355–365.
- Jones S. Regular self-examination or clinical examination for early detection of breast cancer. Int J Epidemiol. 2008;37(6):1219.
- ACOG Committee on Practice Bulletins–Gynecology. ACOG Practice Bulletin No. 122. American College of Obstetricians and Gynecologists website. http://www.acog.org/Resources-And-Publications/Practice-Bulletins/Committee-on-Practice-Bulletins-Gynecology/Breast-Cancer-Screening. Published August 2011; reaffirmed 2014. Accessed November 10, 2016.
- ACOG Statement on breast cancer screening guidelines. American College of Obstetricians and Gynecologists website. http://www.acog.org/About-ACOG/News-Room/Statements/2016/ACOG-Statement-on-Breast-Cancer-Screening-Guidelines. Published January 11, 2016. Accessed November 10, 2016.
- Final Recommendation Statement: Breast Cancer: Screening. U.S. Preventive Services Task Force web site. https://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/breast-cancer-screening1. Published September 2016. Accessed November 10, 2016.
- American Cancer Society recommendations for early breast cancer detection in women without breast symptoms. American Cancer Society website. http://www.cancer.org/cancer/breastcancer/moreinformation/breastcancerearlydetection/breast-cancer-early-detection-acs-recs. Updated October 20, 2015. Accessed November 10, 2016.
- Mathis K, Hoskin T, Boughey J, et al. Palpable presentation of breast cancer persists in the era of screening mammography. J Am Coll Surg. 2010;210(3):314–318.
- MacBride M, Pruthi S, Bevers T. The evolution of breast self-examination to breast awareness. Breast J. 2012;18(6):641–643.
- Susan G. Komen Foundation. Breast self-awareness messages. http://ww5.komen.org/Breastcancer/Breastselfawareness.html. Accessed November 10, 2016.
- DelTurco MR, Mantellini P, Ciatto S, et al. Full-field digital versus screen-film mammography: comparative accuracy in concurrent screening cohorts. AJR. 2007;189(4):860–866.
- Susan G. Koman Foundation. Factors that do not increase breast cancer risk: Breast implants. https://ww5.komen.org/BreastCancer/FactorsThatDoNotIncreaseRisk.html. Updated October 28, 2016. Accessed November 10, 2016.
- Miglioretti D, Rutter, C, Geller B, et al. Effect of breast augmentation on the accuracy of mammography and cancer characteristics. JAMA. 2004;291(4):442–450.
- Eklund GW, Busby RC, Miller SH, Job JS. Improved imaging of the augmented breast. Am J Roentgenol. 1988;151(3):469–473.
- Kam K, Lee E, Pairwan S, et al. The effect of breast implants on mammogram outcomes. Am Surg. 2015;81(10):1053–1056.
- American Cancer Society. Mammograms and other imaging tests. http://www.cancer.org/acs/groups/cid/documents/webcontent/003178-pdf.pdf. Revised April 25, 2016. Accessed November 10, 2016.
- Dalberg K, Hellborg H, Warnberg F. Paget’s disease of the nipple in a population based cohort. Breast Cancer Res Treat. 2008;111(2):313–319.
- Saber Tehrani A, Lee H, Mathews S, et al. 25-year summary of US malpractice claims for diagnostic errors 1986–2010: an analysis from the National Practitioner Data Bank. BMJ Qual Saf. 2013;22(8):672–680.
- White AA, Pichert JW, Bledsoe SH, Irwin C, Entman SS. Cause and effect analysis of closed claims in obstetrics and gynecology. Obstet Gynecol. 2005;105(5 pt 1):1031–1038.
- Ward CJ, Green VL. Risk management and medico-legal issues in breast cancer. Clin Obstet Gynecol. 2016;59(2):439–446.
- Guest L, Schap D, Tran T. The “Loss of Chance” rule as a special category of damages in medical malpractice: a state-by-state analysis. J Legal Econ. 2015;21(2):53–107.
- Kuroishi T, Tominaga S, Morimoto T, et al. Tumor growth rate and prognosis of breast cancer mainly detected by mass screening. Jpn J Cancer Res. 1990;81(5):454–462.
- Localio AR, Lawthers AG, Brennan TA, et al. Relation between malpractice claims and adverse events due to negligence: results of the Harvard Medical Practice Study III. N Engl J Medicine. 1991;325(4):245–251.
- Gandhi TK, Kachalia A, Thomas EJ, et al. Missed and delayed diagnoses in the ambulatory setting: a study of closed malpractice claims. Ann Intern Med. 2006;145(7):488–496.
- Studdert DM, Mello MM, Gawande AA, et al. Claims, errors, and compensation payments in medical malpractice litigation. N Engl J Med. 2006;354(19):2024–2033.
- Huntington B, Kuhn N. Communication gaffes: a root cause of malpractice claims. Proc (Bayl Univ Med Cent). 2003;16(2):157–161.
- Moore PJ, Adler NE, Robertson PA. Medical malpractice: the effect of doctor-patient relations on medical patient perceptions and malpractice intentions. West J Med. 2000;173(4):244–250.
- Hickson GB, Jenkins AD. Identifying and addressing communication failures as a means of reducing unnecessary malpractice claims. NC Med J. 2007;68(5):362–364.
- Jena AB, Chandra A, Lakdawalla D, Seabury S. Outcomes of medical malpractice litigation against US physicians. Arch Intern Med. 2012;172(11):892–894.
CASE: After routine mammography results, DCIS found
A 49-year-old woman (G2 P2002) with a history of fibrocystic breast disease presented with a left breast mass that she found a month ago on self-examination. The patient faithfully had obtained routine mammograms since age 40. This year, after reporting the mass and with spot films obtained as recommended by the radiologist, a new cluster of microcalcifications was identified on the report: “spot compression” assessment identified a 3-cm mass and noted “s/p breast augmentation.”
The radiologist interpreted the spot films to be benign. His report stated that “15% of breast cancers are not detected by mammogram and breast self-exam is recommended monthly from 40 years of age.”
The gynecologist recommended a 6-month follow up. When the patient complied, the radiologist’s report again noted calcifications believed to be nonmalignant. Six months later, the patient presented with bloody nipple discharge from her left breast with apparent “eczema-like” lesions on the areola. The patient noted that her “left implant felt different.”
The patient’s surgical history included breast augmentation “years ago.” Her family history was negative for breast cancer. Her medications included hormone therapy (conjugated estrogens 0.625 mg with medroxyprogesterone acetate 2.5 mg daily) for vaginal atrophy. Other medical conditions included irritable bowel syndrome (managed with diet), anxiety and mood swings (for which she was taking sertraline), decreased libido, and irregular vaginal bleeding (after the patient refused endometrial sampling, she was switched to oral contraceptives to address the problem). In addition, her hypertension was being treated with hydrochlorothiazide.
At the gynecologist’s suggestion, a dermatology consultation was obtained.
The dermatologist gave a diagnosis of Paget disease with high-grade ductal carcinoma-in-situ (DCIS). The interval from screening mammogram to DCIS diagnosis had been 8 months. The dermatologist referred the patient to a breast surgeon. A discussion ensued between the breast surgeon and the dermatologist concerning the difficulty of making a diagnosis of breast cancer in a woman with breast augmentation.
The patient underwent modified radical mastectomy, and histopathology revealed DCIS with clear margins; lymph nodes were negative.
The patient filed a malpractice suit against the gynecologist related to the delayed breast mass evaluation and management. She remained upset that when she called the gynecologist’s office to convey her concerns regarding the left nipple discharge and implant concerns, “she was blown off.” She felt there was a clear “failure to communicate on critical matters of her health.” She alleged that the gynecologist, not the dermatologist, should have referred her to a breast surgeon.
WHAT 'S THE VERDICT?
In the end, the patient decided not to pursue the lawsuit.
Related article:
Who is liable when a surgical error occurs?
Medical considerations
Breast cancer is the most common female malignancy, with 232,340 cases occurring annually in the United States. It is the second leading cause of cancer-related death in US women.1 For this case discussion, we review the role of breast cancer screening, including breast self-examination and mammography.
Is breast self-examination recommended?
Recently, medical care has evolved from “breast self-examination” (BSE) to “breast self-awareness.”2 The concept of BSE and concerns about it stem in part from “the Shanghai study.”3 In this prospective randomized trial, 266,064 female textile workers were randomly assigned to “rigorous and repetitive training in BSE” versus no instruction and no BSE performance. The former group had twice as many breast biopsies than the latter group (2,761 biopsies in the BSE group vs 1,505 in the control group). There was no difference in the number of breast cancers diagnosed among the groups—864 in the BSE group and 896 in the control group (relative risk [RR], 0.97; 95% confidence interval [CI], 0.88–1.06; P = .47). Other studies also support lack of efficacy regarding BSE.4
The potential for psychological harm, unnecessary biopsy, and additional imaging in association with false-positive findings is a concern2 (TABLE). The American College of Obstetricians and Gynecologists (ACOG) states, “breast self-examination may be appropriate for certain high-risk populations and for other women who chose to follow this approach.”2,5,6 The US Preventive Services Task Force (USPSTF) guidelines say that there is “insufficient evidence to assess risks vs benefits, including harms,” and recommends against teaching BSE.7 The American Cancer Society puts BSE in the “optional” category.2,8

Is there a middle of the road strategy? Perhaps. The concept of breast-awareness was developed so that women understand how their breasts look and feel.2,5 The concept does not advocate monthly BSE. The Mayo Clinic reported that, of 592 breast cancers, 57% were detected following abnormal screening mammography, 30% by BSE and 14% by clinical examination by a clinician. Furthermore, 38% of women with a palpable abnormality had a normal mammogram within the preceding 13 months.9 McBride and colleagues aptly addressed this: “Healthcare providers can educate their patients that breast awareness, in essence, is a two-step process. First, it requires that women be familiar with their breasts and aware of new changes and, second, have an understanding of the implications of these changes which includes informing their health care provider promptly.”10 The concept of “know what is normal for you” as conveyed by The Susan G. Komen Foundation succinctly encourages communication with patients.2,11
Mammography
The latest technique in mammography is digital or 3D mammography, also known as tomosynthesis. The technique is similar to 2D mammography with the addition of digital cameras. A study published in the radiology literature noted that the 2 methods were equivalent.12 One possible advantage of 3D mammography is that the 3D images are stored in computer files and are more easily incorporated into the electronic medical record.
What about mammography after breast augmentation?
While breast augmentation is not associated with an increase in breast cancer,13 mammography following breast augmentation can be more difficult to interpret and may result in a delay in diagnosis. In a prospective study of asymptomatic women who were diagnosed with breast cancer, 137 had augmentation and 685 did not. Miglioretti and colleagues noted that the sensitivity of screening mammography was lower in the augmentation cohort.14 To enhance accuracy, breast implant displacement views (in which the breast tissue is pulled forward and the implant is displaced posteriorly to improve visualization) have been recommended.15 A retrospective review provides data reporting no effect on interpretation of mammograms following augmentation.16 The American Cancer Society recommends the same screening for women with implants as without implants, starting at age 40 years.17
Paget disease of the breast
Paget disease of the breast was first described by Sir James Paget in 1874. He also defined Paget disease of extramammary tissue, bone, vulva, and penis. Paget disease of the breast is a rare type of cancer in which the skin and nipple are involved frequently in association with DCIS or invasive breast cancer. The skin has an eczema-like appearance. Characteristic Paget (malignant) cells are large with clear cytoplasm (clear halo) and eccentric, hyperchromatic nuclei throughout the dermis. Assessment includes mammography and biopsy with immunohistochemical staining. Treatment varies by case and can include lumpectomy or mastectomy and chemotherapy and/or radiation therapy. Medications, including tamoxifen and anastrazole, have been recommended. Prognosis depends on nodal involvement. The disease is more common in women older than age 50.18
Related article:
The medicolegal considerations of interacting with your patients online
Legal issues: What was the gynecologist’s obligation?
The question remains, did the gynecologist have an obligation to obtain diagnostic mammography and ultrasound of the breast? Would it have been prudent for immediate referral to a breast surgeon? These are critical questions.
Negligence and the standard of care
The malpractice lawsuit against this gynecologist is based on negligence. In essence, it is a claim that the management provided fell below a standard of care that would be given by a reasonably careful and prudent gynecologist under the circumstances. This generally means that the care was less than the profession itself would find acceptable. Here the claim is essentially a diagnostic error claim, a common basis of malpractice.19 The delay in diagnosing breast cancer is a leading cause of malpractice claims against gynecologists.20,21
It is axiomatic that not all bad outcomes are the result of error, and not all errors are a result of an unacceptable standard of care. It is only bad outcomes resulting from careless or negligent errors that give rise to malpractice. But malpractice claims are often filed without knowing whether or not there was negligence—and, as we will see, many of those claims are without merit.
Was there negligence?
Ordinarily the plaintiff/patient must demonstrate that the care by the physician fell below the standard of care and, as a result, the patient suffered an injury. Stated another way, the plaintiff must show that the physician’s actions were unreasonable given all of the circumstances. In this case, the plaintiff must demonstrate that the gynecologist’s care was not appropriate. Failure to refer to another physician or provide additional testing is likely to be the major claim for negligence or inappropriate care.
Did that negligence cause injury?
Even if the plaintiff can demonstrate that the care was negligent, to prevail the plaintiff must also demonstrate that that negligence caused the injury—and that might be difficult.
What injury was caused by the delay in discovering the cancer? It did not apparently lead to the patient’s death. Can it be proven that the delay clearly shortened the patient’s life expectancy or required additional expensive and painful treatment? That may be difficult to demonstrate.
Causation. The causation factor could appear in many cancer or similar medical cases. On one hand, causation is a critical matter, but on the other hand, delay in treating cancer might have a very adverse effect on patients.
In recent years, about half the states have solved this dilemma by recognizing the concept of “loss of chance.”22 Essentially, this means that the health care provider, by delaying treatment, diminished the possibility that the patient would survive or recover fully.
There are significant variations among states in the loss-of-chance concept, many being quite technical.22 Thus, it is possible that a delay that reduced the chance of recovery or survival could be the injury and causal connection between the injury and the negligence. Even in states that recognize the loss-of-chance concept, the patient still must prove loss of chance.
Lessons learned from this case study
- Breast self-awareness has replaced (substituted) breast self-examination
- ACOG recommends breast mammography beginning at age 40 years
- Breast augmentation affects mammographic interpretation
- Prehaps if better communication had been provided initially, the patient would not have sought legal counsel or filed a weak suit
Related article:
Is the smartphone recording while the patient is under anesthesia?
Is this a strong malpractice case?
In the case presented here, it is not clear that the patient could show a meaningful loss of chance. If there is a delay in the breast cancer diagnosis, tumor doubling time would be an issue. While it is impossible to assess growth rate when a breast cancer is in its preclinical microscopic stage, doubling time can be 100 to 200 days. Therefore, it would take 20 years for the tumor to reach a 1- to 2-cm diameter. A log-normal distribution has been suggested for determining tumor growth.23
Although the facts in this case are sketchy, this does not look like a strong malpractice case. Given the expense, difficulty, and length of time it takes to pursue a malpractice case (especially for someone battling cancer), an obvious question is: Why would a patient file a lawsuit in these circumstances? There is no single answer to that, but the hope of getting rich is unlikely a primary motivation. Ironically, many malpractice cases are filed in which there was no error (or at least no negligent error).24
The search for what really happened, or why the bad event happened, is key. In other circumstances, it may be a desire for revenge or to protect other patients from similar bad results. Studies repeatedly have shown a somewhat limited correlation between negligent error and the decision to file a malpractice claim.24–26 In this case, the patient’s sense of being “blown off” during a particularly difficult time may represent the reason why she filed a malpractice lawsuit. Communication gaffes and poor physician-patient relationships undoubtedly contribute to medical malpractice claims.27,28 Improving communication with patients probably improves care, but it also almost certainly reduces the risk of a malpractice claim.29
Why a lawyer would accept this case is also unclear, but that is an issue for another day. Also for another day is the issue of product liability concerning breast implants. Those legal issues and related liability, primarily directed to the manufacturers of the implants, are interesting topics. They are also complex and will be the subject of a future article.
Finally, the patient’s decision to not pursue her lawsuit does not come as a surprise. A relatively small percentage of malpractice claims result in any meaningful financial recovery for the plaintiff. Few cases go to trial, and of those that do result in a verdict, about 75% of the verdicts are in favor of the physician.25,30 Many cases just fade away, either because the plaintiff never pursues them or because they are dismissed by a court at an early stage. Nonetheless, for the physician, even winning a malpractice case is disruptive and difficult. So in addition to ensuring careful, quality, and up-to-date care, a physician should seek to maintain good relationships and communication with patients to reduce the probability of even weak lawsuits being filed.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
CASE: After routine mammography results, DCIS found
A 49-year-old woman (G2 P2002) with a history of fibrocystic breast disease presented with a left breast mass that she found a month ago on self-examination. The patient faithfully had obtained routine mammograms since age 40. This year, after reporting the mass and with spot films obtained as recommended by the radiologist, a new cluster of microcalcifications was identified on the report: “spot compression” assessment identified a 3-cm mass and noted “s/p breast augmentation.”
The radiologist interpreted the spot films to be benign. His report stated that “15% of breast cancers are not detected by mammogram and breast self-exam is recommended monthly from 40 years of age.”
The gynecologist recommended a 6-month follow up. When the patient complied, the radiologist’s report again noted calcifications believed to be nonmalignant. Six months later, the patient presented with bloody nipple discharge from her left breast with apparent “eczema-like” lesions on the areola. The patient noted that her “left implant felt different.”
The patient’s surgical history included breast augmentation “years ago.” Her family history was negative for breast cancer. Her medications included hormone therapy (conjugated estrogens 0.625 mg with medroxyprogesterone acetate 2.5 mg daily) for vaginal atrophy. Other medical conditions included irritable bowel syndrome (managed with diet), anxiety and mood swings (for which she was taking sertraline), decreased libido, and irregular vaginal bleeding (after the patient refused endometrial sampling, she was switched to oral contraceptives to address the problem). In addition, her hypertension was being treated with hydrochlorothiazide.
At the gynecologist’s suggestion, a dermatology consultation was obtained.
The dermatologist gave a diagnosis of Paget disease with high-grade ductal carcinoma-in-situ (DCIS). The interval from screening mammogram to DCIS diagnosis had been 8 months. The dermatologist referred the patient to a breast surgeon. A discussion ensued between the breast surgeon and the dermatologist concerning the difficulty of making a diagnosis of breast cancer in a woman with breast augmentation.
The patient underwent modified radical mastectomy, and histopathology revealed DCIS with clear margins; lymph nodes were negative.
The patient filed a malpractice suit against the gynecologist related to the delayed breast mass evaluation and management. She remained upset that when she called the gynecologist’s office to convey her concerns regarding the left nipple discharge and implant concerns, “she was blown off.” She felt there was a clear “failure to communicate on critical matters of her health.” She alleged that the gynecologist, not the dermatologist, should have referred her to a breast surgeon.
WHAT 'S THE VERDICT?
In the end, the patient decided not to pursue the lawsuit.
Related article:
Who is liable when a surgical error occurs?
Medical considerations
Breast cancer is the most common female malignancy, with 232,340 cases occurring annually in the United States. It is the second leading cause of cancer-related death in US women.1 For this case discussion, we review the role of breast cancer screening, including breast self-examination and mammography.
Is breast self-examination recommended?
Recently, medical care has evolved from “breast self-examination” (BSE) to “breast self-awareness.”2 The concept of BSE and concerns about it stem in part from “the Shanghai study.”3 In this prospective randomized trial, 266,064 female textile workers were randomly assigned to “rigorous and repetitive training in BSE” versus no instruction and no BSE performance. The former group had twice as many breast biopsies than the latter group (2,761 biopsies in the BSE group vs 1,505 in the control group). There was no difference in the number of breast cancers diagnosed among the groups—864 in the BSE group and 896 in the control group (relative risk [RR], 0.97; 95% confidence interval [CI], 0.88–1.06; P = .47). Other studies also support lack of efficacy regarding BSE.4
The potential for psychological harm, unnecessary biopsy, and additional imaging in association with false-positive findings is a concern2 (TABLE). The American College of Obstetricians and Gynecologists (ACOG) states, “breast self-examination may be appropriate for certain high-risk populations and for other women who chose to follow this approach.”2,5,6 The US Preventive Services Task Force (USPSTF) guidelines say that there is “insufficient evidence to assess risks vs benefits, including harms,” and recommends against teaching BSE.7 The American Cancer Society puts BSE in the “optional” category.2,8

Is there a middle of the road strategy? Perhaps. The concept of breast-awareness was developed so that women understand how their breasts look and feel.2,5 The concept does not advocate monthly BSE. The Mayo Clinic reported that, of 592 breast cancers, 57% were detected following abnormal screening mammography, 30% by BSE and 14% by clinical examination by a clinician. Furthermore, 38% of women with a palpable abnormality had a normal mammogram within the preceding 13 months.9 McBride and colleagues aptly addressed this: “Healthcare providers can educate their patients that breast awareness, in essence, is a two-step process. First, it requires that women be familiar with their breasts and aware of new changes and, second, have an understanding of the implications of these changes which includes informing their health care provider promptly.”10 The concept of “know what is normal for you” as conveyed by The Susan G. Komen Foundation succinctly encourages communication with patients.2,11
Mammography
The latest technique in mammography is digital or 3D mammography, also known as tomosynthesis. The technique is similar to 2D mammography with the addition of digital cameras. A study published in the radiology literature noted that the 2 methods were equivalent.12 One possible advantage of 3D mammography is that the 3D images are stored in computer files and are more easily incorporated into the electronic medical record.
What about mammography after breast augmentation?
While breast augmentation is not associated with an increase in breast cancer,13 mammography following breast augmentation can be more difficult to interpret and may result in a delay in diagnosis. In a prospective study of asymptomatic women who were diagnosed with breast cancer, 137 had augmentation and 685 did not. Miglioretti and colleagues noted that the sensitivity of screening mammography was lower in the augmentation cohort.14 To enhance accuracy, breast implant displacement views (in which the breast tissue is pulled forward and the implant is displaced posteriorly to improve visualization) have been recommended.15 A retrospective review provides data reporting no effect on interpretation of mammograms following augmentation.16 The American Cancer Society recommends the same screening for women with implants as without implants, starting at age 40 years.17
Paget disease of the breast
Paget disease of the breast was first described by Sir James Paget in 1874. He also defined Paget disease of extramammary tissue, bone, vulva, and penis. Paget disease of the breast is a rare type of cancer in which the skin and nipple are involved frequently in association with DCIS or invasive breast cancer. The skin has an eczema-like appearance. Characteristic Paget (malignant) cells are large with clear cytoplasm (clear halo) and eccentric, hyperchromatic nuclei throughout the dermis. Assessment includes mammography and biopsy with immunohistochemical staining. Treatment varies by case and can include lumpectomy or mastectomy and chemotherapy and/or radiation therapy. Medications, including tamoxifen and anastrazole, have been recommended. Prognosis depends on nodal involvement. The disease is more common in women older than age 50.18
Related article:
The medicolegal considerations of interacting with your patients online
Legal issues: What was the gynecologist’s obligation?
The question remains, did the gynecologist have an obligation to obtain diagnostic mammography and ultrasound of the breast? Would it have been prudent for immediate referral to a breast surgeon? These are critical questions.
Negligence and the standard of care
The malpractice lawsuit against this gynecologist is based on negligence. In essence, it is a claim that the management provided fell below a standard of care that would be given by a reasonably careful and prudent gynecologist under the circumstances. This generally means that the care was less than the profession itself would find acceptable. Here the claim is essentially a diagnostic error claim, a common basis of malpractice.19 The delay in diagnosing breast cancer is a leading cause of malpractice claims against gynecologists.20,21
It is axiomatic that not all bad outcomes are the result of error, and not all errors are a result of an unacceptable standard of care. It is only bad outcomes resulting from careless or negligent errors that give rise to malpractice. But malpractice claims are often filed without knowing whether or not there was negligence—and, as we will see, many of those claims are without merit.
Was there negligence?
Ordinarily the plaintiff/patient must demonstrate that the care by the physician fell below the standard of care and, as a result, the patient suffered an injury. Stated another way, the plaintiff must show that the physician’s actions were unreasonable given all of the circumstances. In this case, the plaintiff must demonstrate that the gynecologist’s care was not appropriate. Failure to refer to another physician or provide additional testing is likely to be the major claim for negligence or inappropriate care.
Did that negligence cause injury?
Even if the plaintiff can demonstrate that the care was negligent, to prevail the plaintiff must also demonstrate that that negligence caused the injury—and that might be difficult.
What injury was caused by the delay in discovering the cancer? It did not apparently lead to the patient’s death. Can it be proven that the delay clearly shortened the patient’s life expectancy or required additional expensive and painful treatment? That may be difficult to demonstrate.
Causation. The causation factor could appear in many cancer or similar medical cases. On one hand, causation is a critical matter, but on the other hand, delay in treating cancer might have a very adverse effect on patients.
In recent years, about half the states have solved this dilemma by recognizing the concept of “loss of chance.”22 Essentially, this means that the health care provider, by delaying treatment, diminished the possibility that the patient would survive or recover fully.
There are significant variations among states in the loss-of-chance concept, many being quite technical.22 Thus, it is possible that a delay that reduced the chance of recovery or survival could be the injury and causal connection between the injury and the negligence. Even in states that recognize the loss-of-chance concept, the patient still must prove loss of chance.
Lessons learned from this case study
- Breast self-awareness has replaced (substituted) breast self-examination
- ACOG recommends breast mammography beginning at age 40 years
- Breast augmentation affects mammographic interpretation
- Prehaps if better communication had been provided initially, the patient would not have sought legal counsel or filed a weak suit
Related article:
Is the smartphone recording while the patient is under anesthesia?
Is this a strong malpractice case?
In the case presented here, it is not clear that the patient could show a meaningful loss of chance. If there is a delay in the breast cancer diagnosis, tumor doubling time would be an issue. While it is impossible to assess growth rate when a breast cancer is in its preclinical microscopic stage, doubling time can be 100 to 200 days. Therefore, it would take 20 years for the tumor to reach a 1- to 2-cm diameter. A log-normal distribution has been suggested for determining tumor growth.23
Although the facts in this case are sketchy, this does not look like a strong malpractice case. Given the expense, difficulty, and length of time it takes to pursue a malpractice case (especially for someone battling cancer), an obvious question is: Why would a patient file a lawsuit in these circumstances? There is no single answer to that, but the hope of getting rich is unlikely a primary motivation. Ironically, many malpractice cases are filed in which there was no error (or at least no negligent error).24
The search for what really happened, or why the bad event happened, is key. In other circumstances, it may be a desire for revenge or to protect other patients from similar bad results. Studies repeatedly have shown a somewhat limited correlation between negligent error and the decision to file a malpractice claim.24–26 In this case, the patient’s sense of being “blown off” during a particularly difficult time may represent the reason why she filed a malpractice lawsuit. Communication gaffes and poor physician-patient relationships undoubtedly contribute to medical malpractice claims.27,28 Improving communication with patients probably improves care, but it also almost certainly reduces the risk of a malpractice claim.29
Why a lawyer would accept this case is also unclear, but that is an issue for another day. Also for another day is the issue of product liability concerning breast implants. Those legal issues and related liability, primarily directed to the manufacturers of the implants, are interesting topics. They are also complex and will be the subject of a future article.
Finally, the patient’s decision to not pursue her lawsuit does not come as a surprise. A relatively small percentage of malpractice claims result in any meaningful financial recovery for the plaintiff. Few cases go to trial, and of those that do result in a verdict, about 75% of the verdicts are in favor of the physician.25,30 Many cases just fade away, either because the plaintiff never pursues them or because they are dismissed by a court at an early stage. Nonetheless, for the physician, even winning a malpractice case is disruptive and difficult. So in addition to ensuring careful, quality, and up-to-date care, a physician should seek to maintain good relationships and communication with patients to reduce the probability of even weak lawsuits being filed.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63(1):11–30.
- Mark K, Temkin S, Terplan M. Breast self-awareness: the evidence behind the euphemism. Obstet Gynecol. 2014;123(4):734–745.
- Thomas D, Gao D, Self S, et al. Randomized trial of breast self-examination in Shanghai: methodology and preliminary results. J Natl Cancer Inst. 1997;89(5):355–365.
- Jones S. Regular self-examination or clinical examination for early detection of breast cancer. Int J Epidemiol. 2008;37(6):1219.
- ACOG Committee on Practice Bulletins–Gynecology. ACOG Practice Bulletin No. 122. American College of Obstetricians and Gynecologists website. http://www.acog.org/Resources-And-Publications/Practice-Bulletins/Committee-on-Practice-Bulletins-Gynecology/Breast-Cancer-Screening. Published August 2011; reaffirmed 2014. Accessed November 10, 2016.
- ACOG Statement on breast cancer screening guidelines. American College of Obstetricians and Gynecologists website. http://www.acog.org/About-ACOG/News-Room/Statements/2016/ACOG-Statement-on-Breast-Cancer-Screening-Guidelines. Published January 11, 2016. Accessed November 10, 2016.
- Final Recommendation Statement: Breast Cancer: Screening. U.S. Preventive Services Task Force web site. https://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/breast-cancer-screening1. Published September 2016. Accessed November 10, 2016.
- American Cancer Society recommendations for early breast cancer detection in women without breast symptoms. American Cancer Society website. http://www.cancer.org/cancer/breastcancer/moreinformation/breastcancerearlydetection/breast-cancer-early-detection-acs-recs. Updated October 20, 2015. Accessed November 10, 2016.
- Mathis K, Hoskin T, Boughey J, et al. Palpable presentation of breast cancer persists in the era of screening mammography. J Am Coll Surg. 2010;210(3):314–318.
- MacBride M, Pruthi S, Bevers T. The evolution of breast self-examination to breast awareness. Breast J. 2012;18(6):641–643.
- Susan G. Komen Foundation. Breast self-awareness messages. http://ww5.komen.org/Breastcancer/Breastselfawareness.html. Accessed November 10, 2016.
- DelTurco MR, Mantellini P, Ciatto S, et al. Full-field digital versus screen-film mammography: comparative accuracy in concurrent screening cohorts. AJR. 2007;189(4):860–866.
- Susan G. Koman Foundation. Factors that do not increase breast cancer risk: Breast implants. https://ww5.komen.org/BreastCancer/FactorsThatDoNotIncreaseRisk.html. Updated October 28, 2016. Accessed November 10, 2016.
- Miglioretti D, Rutter, C, Geller B, et al. Effect of breast augmentation on the accuracy of mammography and cancer characteristics. JAMA. 2004;291(4):442–450.
- Eklund GW, Busby RC, Miller SH, Job JS. Improved imaging of the augmented breast. Am J Roentgenol. 1988;151(3):469–473.
- Kam K, Lee E, Pairwan S, et al. The effect of breast implants on mammogram outcomes. Am Surg. 2015;81(10):1053–1056.
- American Cancer Society. Mammograms and other imaging tests. http://www.cancer.org/acs/groups/cid/documents/webcontent/003178-pdf.pdf. Revised April 25, 2016. Accessed November 10, 2016.
- Dalberg K, Hellborg H, Warnberg F. Paget’s disease of the nipple in a population based cohort. Breast Cancer Res Treat. 2008;111(2):313–319.
- Saber Tehrani A, Lee H, Mathews S, et al. 25-year summary of US malpractice claims for diagnostic errors 1986–2010: an analysis from the National Practitioner Data Bank. BMJ Qual Saf. 2013;22(8):672–680.
- White AA, Pichert JW, Bledsoe SH, Irwin C, Entman SS. Cause and effect analysis of closed claims in obstetrics and gynecology. Obstet Gynecol. 2005;105(5 pt 1):1031–1038.
- Ward CJ, Green VL. Risk management and medico-legal issues in breast cancer. Clin Obstet Gynecol. 2016;59(2):439–446.
- Guest L, Schap D, Tran T. The “Loss of Chance” rule as a special category of damages in medical malpractice: a state-by-state analysis. J Legal Econ. 2015;21(2):53–107.
- Kuroishi T, Tominaga S, Morimoto T, et al. Tumor growth rate and prognosis of breast cancer mainly detected by mass screening. Jpn J Cancer Res. 1990;81(5):454–462.
- Localio AR, Lawthers AG, Brennan TA, et al. Relation between malpractice claims and adverse events due to negligence: results of the Harvard Medical Practice Study III. N Engl J Medicine. 1991;325(4):245–251.
- Gandhi TK, Kachalia A, Thomas EJ, et al. Missed and delayed diagnoses in the ambulatory setting: a study of closed malpractice claims. Ann Intern Med. 2006;145(7):488–496.
- Studdert DM, Mello MM, Gawande AA, et al. Claims, errors, and compensation payments in medical malpractice litigation. N Engl J Med. 2006;354(19):2024–2033.
- Huntington B, Kuhn N. Communication gaffes: a root cause of malpractice claims. Proc (Bayl Univ Med Cent). 2003;16(2):157–161.
- Moore PJ, Adler NE, Robertson PA. Medical malpractice: the effect of doctor-patient relations on medical patient perceptions and malpractice intentions. West J Med. 2000;173(4):244–250.
- Hickson GB, Jenkins AD. Identifying and addressing communication failures as a means of reducing unnecessary malpractice claims. NC Med J. 2007;68(5):362–364.
- Jena AB, Chandra A, Lakdawalla D, Seabury S. Outcomes of medical malpractice litigation against US physicians. Arch Intern Med. 2012;172(11):892–894.
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63(1):11–30.
- Mark K, Temkin S, Terplan M. Breast self-awareness: the evidence behind the euphemism. Obstet Gynecol. 2014;123(4):734–745.
- Thomas D, Gao D, Self S, et al. Randomized trial of breast self-examination in Shanghai: methodology and preliminary results. J Natl Cancer Inst. 1997;89(5):355–365.
- Jones S. Regular self-examination or clinical examination for early detection of breast cancer. Int J Epidemiol. 2008;37(6):1219.
- ACOG Committee on Practice Bulletins–Gynecology. ACOG Practice Bulletin No. 122. American College of Obstetricians and Gynecologists website. http://www.acog.org/Resources-And-Publications/Practice-Bulletins/Committee-on-Practice-Bulletins-Gynecology/Breast-Cancer-Screening. Published August 2011; reaffirmed 2014. Accessed November 10, 2016.
- ACOG Statement on breast cancer screening guidelines. American College of Obstetricians and Gynecologists website. http://www.acog.org/About-ACOG/News-Room/Statements/2016/ACOG-Statement-on-Breast-Cancer-Screening-Guidelines. Published January 11, 2016. Accessed November 10, 2016.
- Final Recommendation Statement: Breast Cancer: Screening. U.S. Preventive Services Task Force web site. https://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/breast-cancer-screening1. Published September 2016. Accessed November 10, 2016.
- American Cancer Society recommendations for early breast cancer detection in women without breast symptoms. American Cancer Society website. http://www.cancer.org/cancer/breastcancer/moreinformation/breastcancerearlydetection/breast-cancer-early-detection-acs-recs. Updated October 20, 2015. Accessed November 10, 2016.
- Mathis K, Hoskin T, Boughey J, et al. Palpable presentation of breast cancer persists in the era of screening mammography. J Am Coll Surg. 2010;210(3):314–318.
- MacBride M, Pruthi S, Bevers T. The evolution of breast self-examination to breast awareness. Breast J. 2012;18(6):641–643.
- Susan G. Komen Foundation. Breast self-awareness messages. http://ww5.komen.org/Breastcancer/Breastselfawareness.html. Accessed November 10, 2016.
- DelTurco MR, Mantellini P, Ciatto S, et al. Full-field digital versus screen-film mammography: comparative accuracy in concurrent screening cohorts. AJR. 2007;189(4):860–866.
- Susan G. Koman Foundation. Factors that do not increase breast cancer risk: Breast implants. https://ww5.komen.org/BreastCancer/FactorsThatDoNotIncreaseRisk.html. Updated October 28, 2016. Accessed November 10, 2016.
- Miglioretti D, Rutter, C, Geller B, et al. Effect of breast augmentation on the accuracy of mammography and cancer characteristics. JAMA. 2004;291(4):442–450.
- Eklund GW, Busby RC, Miller SH, Job JS. Improved imaging of the augmented breast. Am J Roentgenol. 1988;151(3):469–473.
- Kam K, Lee E, Pairwan S, et al. The effect of breast implants on mammogram outcomes. Am Surg. 2015;81(10):1053–1056.
- American Cancer Society. Mammograms and other imaging tests. http://www.cancer.org/acs/groups/cid/documents/webcontent/003178-pdf.pdf. Revised April 25, 2016. Accessed November 10, 2016.
- Dalberg K, Hellborg H, Warnberg F. Paget’s disease of the nipple in a population based cohort. Breast Cancer Res Treat. 2008;111(2):313–319.
- Saber Tehrani A, Lee H, Mathews S, et al. 25-year summary of US malpractice claims for diagnostic errors 1986–2010: an analysis from the National Practitioner Data Bank. BMJ Qual Saf. 2013;22(8):672–680.
- White AA, Pichert JW, Bledsoe SH, Irwin C, Entman SS. Cause and effect analysis of closed claims in obstetrics and gynecology. Obstet Gynecol. 2005;105(5 pt 1):1031–1038.
- Ward CJ, Green VL. Risk management and medico-legal issues in breast cancer. Clin Obstet Gynecol. 2016;59(2):439–446.
- Guest L, Schap D, Tran T. The “Loss of Chance” rule as a special category of damages in medical malpractice: a state-by-state analysis. J Legal Econ. 2015;21(2):53–107.
- Kuroishi T, Tominaga S, Morimoto T, et al. Tumor growth rate and prognosis of breast cancer mainly detected by mass screening. Jpn J Cancer Res. 1990;81(5):454–462.
- Localio AR, Lawthers AG, Brennan TA, et al. Relation between malpractice claims and adverse events due to negligence: results of the Harvard Medical Practice Study III. N Engl J Medicine. 1991;325(4):245–251.
- Gandhi TK, Kachalia A, Thomas EJ, et al. Missed and delayed diagnoses in the ambulatory setting: a study of closed malpractice claims. Ann Intern Med. 2006;145(7):488–496.
- Studdert DM, Mello MM, Gawande AA, et al. Claims, errors, and compensation payments in medical malpractice litigation. N Engl J Med. 2006;354(19):2024–2033.
- Huntington B, Kuhn N. Communication gaffes: a root cause of malpractice claims. Proc (Bayl Univ Med Cent). 2003;16(2):157–161.
- Moore PJ, Adler NE, Robertson PA. Medical malpractice: the effect of doctor-patient relations on medical patient perceptions and malpractice intentions. West J Med. 2000;173(4):244–250.
- Hickson GB, Jenkins AD. Identifying and addressing communication failures as a means of reducing unnecessary malpractice claims. NC Med J. 2007;68(5):362–364.
- Jena AB, Chandra A, Lakdawalla D, Seabury S. Outcomes of medical malpractice litigation against US physicians. Arch Intern Med. 2012;172(11):892–894.
DNA methylation markers prognostic for metastatic breast cancer
A novel panel of DNA methylation markers may aid in predicting survival outcomes in metastatic breast cancer (MBC), according to new findings.
Using a new quantitative assay known as cMethDNA, researchers found that a high cumulative methylation index (CMI) level, as measured with a six-gene panel after beginning a new treatment, was consistently associated with both progression-free survival and overall survival, as well as progressive disease.
Even though these results are encouraging, however, its clinical application is still unknown, they noted.
Epigenetic alterations are common in human malignancies, and DNA methylation is a form of epigenetic alteration that is heritable during DNA replication. The authors developed a highly sensitive, high-throughput quantitative multiplex methylation-specific polymerase chain reaction assay known as cMethDNA that was able to detect circulating cell-free methylated DNA.
In this study, 10 genes were tested from 141 women with MBC who were beginning a new systemic therapy and who received treatment at participating academic medical centers.
Among patients with a high CMI, the median progression-free survival and overall survival were significantly shorter (PFS, 2.1 months; OS, 12.3 months), compared with those with a low CMI (PFS, 5.8 months; OS, 21.7 months), Dr. Visvanathan and her associates reported (J Clin Oncol. 2016 Nov. 21 doi: 10.1200/JCO.2015.66.2080).
Upon multivariable analysis, a high CMI versus low CMI at 4 weeks was independently associated with poorer PFS (hazard ratio, 1.79; 95% CI, 1.23-2.60; P = .002) and OS (hazard ratio, 1.75; 95% CI, 1.21-2.54; P = .003). Rising CMI levels from baseline to week 4 also were associated with worse PFS (P less than .001) as well as a 4.6-fold increase in the risk of progressive disease at first restaging (OR, 4.58; 95% CI, 1.82-11.60; P = .001) when compared with either a drop in CMI or no change at all.
The CMI level at week 4 was observed to be a strong predictor of PFS and improved the prediction of the base model, even in the presence of circulating tumor cells (P = .004).
Dr. Visvanathan and her colleagues concluded that these findings “must be validated to determine the clinical usefulness of the cMethDNA assay for specific treatments and tumor phenotypes in patients with metastatic disease and early-stage breast cancer.”
The study was supported by the Avon Foundation for Women, the Breast Cancer Research Foundation, Janssen Diagnostics, the Rubenstein Family Fund, the Susan G. Komen Foundation, and National Institutes of Health Grants. Dr. Visvanathan has declared holding patents, royalties, and other intellectual property, and several coauthors also have declared relationships with industry.
A novel panel of DNA methylation markers may aid in predicting survival outcomes in metastatic breast cancer (MBC), according to new findings.
Using a new quantitative assay known as cMethDNA, researchers found that a high cumulative methylation index (CMI) level, as measured with a six-gene panel after beginning a new treatment, was consistently associated with both progression-free survival and overall survival, as well as progressive disease.
Even though these results are encouraging, however, its clinical application is still unknown, they noted.
Epigenetic alterations are common in human malignancies, and DNA methylation is a form of epigenetic alteration that is heritable during DNA replication. The authors developed a highly sensitive, high-throughput quantitative multiplex methylation-specific polymerase chain reaction assay known as cMethDNA that was able to detect circulating cell-free methylated DNA.
In this study, 10 genes were tested from 141 women with MBC who were beginning a new systemic therapy and who received treatment at participating academic medical centers.
Among patients with a high CMI, the median progression-free survival and overall survival were significantly shorter (PFS, 2.1 months; OS, 12.3 months), compared with those with a low CMI (PFS, 5.8 months; OS, 21.7 months), Dr. Visvanathan and her associates reported (J Clin Oncol. 2016 Nov. 21 doi: 10.1200/JCO.2015.66.2080).
Upon multivariable analysis, a high CMI versus low CMI at 4 weeks was independently associated with poorer PFS (hazard ratio, 1.79; 95% CI, 1.23-2.60; P = .002) and OS (hazard ratio, 1.75; 95% CI, 1.21-2.54; P = .003). Rising CMI levels from baseline to week 4 also were associated with worse PFS (P less than .001) as well as a 4.6-fold increase in the risk of progressive disease at first restaging (OR, 4.58; 95% CI, 1.82-11.60; P = .001) when compared with either a drop in CMI or no change at all.
The CMI level at week 4 was observed to be a strong predictor of PFS and improved the prediction of the base model, even in the presence of circulating tumor cells (P = .004).
Dr. Visvanathan and her colleagues concluded that these findings “must be validated to determine the clinical usefulness of the cMethDNA assay for specific treatments and tumor phenotypes in patients with metastatic disease and early-stage breast cancer.”
The study was supported by the Avon Foundation for Women, the Breast Cancer Research Foundation, Janssen Diagnostics, the Rubenstein Family Fund, the Susan G. Komen Foundation, and National Institutes of Health Grants. Dr. Visvanathan has declared holding patents, royalties, and other intellectual property, and several coauthors also have declared relationships with industry.
A novel panel of DNA methylation markers may aid in predicting survival outcomes in metastatic breast cancer (MBC), according to new findings.
Using a new quantitative assay known as cMethDNA, researchers found that a high cumulative methylation index (CMI) level, as measured with a six-gene panel after beginning a new treatment, was consistently associated with both progression-free survival and overall survival, as well as progressive disease.
Even though these results are encouraging, however, its clinical application is still unknown, they noted.
Epigenetic alterations are common in human malignancies, and DNA methylation is a form of epigenetic alteration that is heritable during DNA replication. The authors developed a highly sensitive, high-throughput quantitative multiplex methylation-specific polymerase chain reaction assay known as cMethDNA that was able to detect circulating cell-free methylated DNA.
In this study, 10 genes were tested from 141 women with MBC who were beginning a new systemic therapy and who received treatment at participating academic medical centers.
Among patients with a high CMI, the median progression-free survival and overall survival were significantly shorter (PFS, 2.1 months; OS, 12.3 months), compared with those with a low CMI (PFS, 5.8 months; OS, 21.7 months), Dr. Visvanathan and her associates reported (J Clin Oncol. 2016 Nov. 21 doi: 10.1200/JCO.2015.66.2080).
Upon multivariable analysis, a high CMI versus low CMI at 4 weeks was independently associated with poorer PFS (hazard ratio, 1.79; 95% CI, 1.23-2.60; P = .002) and OS (hazard ratio, 1.75; 95% CI, 1.21-2.54; P = .003). Rising CMI levels from baseline to week 4 also were associated with worse PFS (P less than .001) as well as a 4.6-fold increase in the risk of progressive disease at first restaging (OR, 4.58; 95% CI, 1.82-11.60; P = .001) when compared with either a drop in CMI or no change at all.
The CMI level at week 4 was observed to be a strong predictor of PFS and improved the prediction of the base model, even in the presence of circulating tumor cells (P = .004).
Dr. Visvanathan and her colleagues concluded that these findings “must be validated to determine the clinical usefulness of the cMethDNA assay for specific treatments and tumor phenotypes in patients with metastatic disease and early-stage breast cancer.”
The study was supported by the Avon Foundation for Women, the Breast Cancer Research Foundation, Janssen Diagnostics, the Rubenstein Family Fund, the Susan G. Komen Foundation, and National Institutes of Health Grants. Dr. Visvanathan has declared holding patents, royalties, and other intellectual property, and several coauthors also have declared relationships with industry.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point:
Major finding: Among patients with a high cumulative methylation index, the median PFS and OS were significantly shorter (PFS, 2.1 months; OS, 12.3 months), compared with those with a low CMI (PFS, 5.8 months; OS, 21.7 months).
Data source: An experimental study that evaluated 10 genes from samples obtained from 141 metastatic breast cancer patients at varying intervals.
Disclosures: The study was supported by the Avon Foundation for Women, the Breast Cancer Research Foundation, Janssen Diagnostics, the Rubenstein Family Fund, the Susan G. Komen Foundation, and National Institutes of Health Grants. Dr. Visvanathan has declared holding patents, royalties, and other intellectual property, and several coauthors also have declared relationships with industry.
ASTRO guidelines lower age thresholds for APBI
The American Society for Radiation Oncology has issued new guidelines recommending accelerated partial breast irradiation brachytherapy (APBI) as an alternative to whole breast irradiation (WBI) after surgery in early-stage breast cancer patients, and lowering the age range of patients considered suitable for the procedure to people 50 and older, from 60.
With APBI, localized radiation is delivered to the region around the excised tissue, reducing treatment time and sparing healthy tissue. APBI may also be considered for patients 40 and older, according to ASTRO, if they meet all of the pathologic criteria for suitability listed in the guidelines for patients 50 and above.
The guidelines represent the first ASTRO update on APBI since 2009. In addition to expanding the age range for APBI treatment, the guidelines add low-risk ductal carcinoma in situ as an indication. The guidelines also address intraoperative radiation therapy, or IORT, in which patients receive low-energy photon or electron radiation during surgery (Pract Rad Oncol. 2016 Nov. 17 doi: 10.1016/j.prro.2016.09.007).
While IORT is suitable for patients with invasive cancer eligible for APBI, the guidelines say, patients considering this option should be counseled about the risk of recurrence compared with standard treatment, and, with photon IORT, about potential toxicity risk requiring follow-up. Though more than 40 studies were considered by the ASTRO committee, including large randomized trials comparing APBI with WBI, the new recommendations represent “more of a tweak than a revolution,” said Jay Harris, MD, of Brigham and Women’s Hospital and Dana-Farber Cancer Institute, Boston, one of the guideline authors.
Dr. Harris noted in an interview that two important randomized controlled trials comparing APBI and WBI are still underway, with full follow-up results expected in 2-3 years, after which more definitive recommendations can be made. For the intraoperative radiation advice contained in the guidelines, “we had evidence from two trials looking at different approaches,” Dr. Harris said. “One has long-term data using an electron beam in the operating room – this group showed that that approach seems reasonable in patients that we at ASTRO considered suitable in general for APBI. The other approach is low-dose photon radiation, for which we have only short-term follow-up, making us more hesitant to endorse it.” As for the new recommendation sanctioning APBI for ductal carcinoma, “There’s a lot of variation [in protocols] across the country, compared with invasive cancer,” Dr. Harris said. “We’re kind of all over the map with DCIS. This guideline presents another option.”
The guidelines were sponsored by ASTRO; two authors disclosed financial relationships with firms that make radiologic technology.
The American Society for Radiation Oncology has issued new guidelines recommending accelerated partial breast irradiation brachytherapy (APBI) as an alternative to whole breast irradiation (WBI) after surgery in early-stage breast cancer patients, and lowering the age range of patients considered suitable for the procedure to people 50 and older, from 60.
With APBI, localized radiation is delivered to the region around the excised tissue, reducing treatment time and sparing healthy tissue. APBI may also be considered for patients 40 and older, according to ASTRO, if they meet all of the pathologic criteria for suitability listed in the guidelines for patients 50 and above.
The guidelines represent the first ASTRO update on APBI since 2009. In addition to expanding the age range for APBI treatment, the guidelines add low-risk ductal carcinoma in situ as an indication. The guidelines also address intraoperative radiation therapy, or IORT, in which patients receive low-energy photon or electron radiation during surgery (Pract Rad Oncol. 2016 Nov. 17 doi: 10.1016/j.prro.2016.09.007).
While IORT is suitable for patients with invasive cancer eligible for APBI, the guidelines say, patients considering this option should be counseled about the risk of recurrence compared with standard treatment, and, with photon IORT, about potential toxicity risk requiring follow-up. Though more than 40 studies were considered by the ASTRO committee, including large randomized trials comparing APBI with WBI, the new recommendations represent “more of a tweak than a revolution,” said Jay Harris, MD, of Brigham and Women’s Hospital and Dana-Farber Cancer Institute, Boston, one of the guideline authors.
Dr. Harris noted in an interview that two important randomized controlled trials comparing APBI and WBI are still underway, with full follow-up results expected in 2-3 years, after which more definitive recommendations can be made. For the intraoperative radiation advice contained in the guidelines, “we had evidence from two trials looking at different approaches,” Dr. Harris said. “One has long-term data using an electron beam in the operating room – this group showed that that approach seems reasonable in patients that we at ASTRO considered suitable in general for APBI. The other approach is low-dose photon radiation, for which we have only short-term follow-up, making us more hesitant to endorse it.” As for the new recommendation sanctioning APBI for ductal carcinoma, “There’s a lot of variation [in protocols] across the country, compared with invasive cancer,” Dr. Harris said. “We’re kind of all over the map with DCIS. This guideline presents another option.”
The guidelines were sponsored by ASTRO; two authors disclosed financial relationships with firms that make radiologic technology.
The American Society for Radiation Oncology has issued new guidelines recommending accelerated partial breast irradiation brachytherapy (APBI) as an alternative to whole breast irradiation (WBI) after surgery in early-stage breast cancer patients, and lowering the age range of patients considered suitable for the procedure to people 50 and older, from 60.
With APBI, localized radiation is delivered to the region around the excised tissue, reducing treatment time and sparing healthy tissue. APBI may also be considered for patients 40 and older, according to ASTRO, if they meet all of the pathologic criteria for suitability listed in the guidelines for patients 50 and above.
The guidelines represent the first ASTRO update on APBI since 2009. In addition to expanding the age range for APBI treatment, the guidelines add low-risk ductal carcinoma in situ as an indication. The guidelines also address intraoperative radiation therapy, or IORT, in which patients receive low-energy photon or electron radiation during surgery (Pract Rad Oncol. 2016 Nov. 17 doi: 10.1016/j.prro.2016.09.007).
While IORT is suitable for patients with invasive cancer eligible for APBI, the guidelines say, patients considering this option should be counseled about the risk of recurrence compared with standard treatment, and, with photon IORT, about potential toxicity risk requiring follow-up. Though more than 40 studies were considered by the ASTRO committee, including large randomized trials comparing APBI with WBI, the new recommendations represent “more of a tweak than a revolution,” said Jay Harris, MD, of Brigham and Women’s Hospital and Dana-Farber Cancer Institute, Boston, one of the guideline authors.
Dr. Harris noted in an interview that two important randomized controlled trials comparing APBI and WBI are still underway, with full follow-up results expected in 2-3 years, after which more definitive recommendations can be made. For the intraoperative radiation advice contained in the guidelines, “we had evidence from two trials looking at different approaches,” Dr. Harris said. “One has long-term data using an electron beam in the operating room – this group showed that that approach seems reasonable in patients that we at ASTRO considered suitable in general for APBI. The other approach is low-dose photon radiation, for which we have only short-term follow-up, making us more hesitant to endorse it.” As for the new recommendation sanctioning APBI for ductal carcinoma, “There’s a lot of variation [in protocols] across the country, compared with invasive cancer,” Dr. Harris said. “We’re kind of all over the map with DCIS. This guideline presents another option.”
The guidelines were sponsored by ASTRO; two authors disclosed financial relationships with firms that make radiologic technology.
FROM PRACTICAL RADIATION ONCOLOGY
Palbociclib extends PFS in advanced breast cancer
Adding the CDK inhibitor palbociclib to standard endocrine therapy (letrozole) extended progression-free survival in a manufacturer-sponsored phase III trial of advanced ER-positive, HER2-negative breast cancer, investigators reported in the New England Journal of Medicine.
Compared with placebo, palbociclib significantly improved progression-free survival regardless of patient age, patient performance status, site of metastasis, prior use of chemotherapy, prior use of endocrine therapy, length of the disease-free interval before progression, or the cancer’s histologic subtype. “The median progression-free survival of 24.8 months in [our study] is longer than that seen in other phase III studies involving women with advanced breast cancer. Whether this progression-free survival will result in longer overall survival is uncertain until further follow-up is completed,” wrote Richard S. Finn, MD, of the University of California, Los Angeles, and his associates.
The primary endpoint – time to disease progression or death – was 24.8 months with active treatment, compared with 14.5 months with placebo, for a hazard ratio of 0.58. The rate of disease progression or death was 43.7% in the palbociclib group, compared with 61.7% in the placebo group. In addition, the rate of objective tumor response was 42.1% with palbociclib, compared with 34.7% with placebo, they reported.
“Data on overall survival were immature at the time of this analysis of the primary end point, and the final overall survival analysis will be performed when a total of 390 deaths occur, per protocol and in agreement with regulatory agencies,” Dr. Finn and his associates said (N Engl J Med. 2016 Nov. 17. doi: 10.1056/NEJMoa1607303). As expected, the rate of hematologic adverse events was high with palbociclib. “Although the incidence of neutropenia of any grade in the palbobiclib/letrozole group was 79.5% in the current study, the incidence of febrile neutropenia was lower than 2%. In addition, the rate of permanent treatment discontinuation associated with an adverse event did not differ significantly between the two study groups, although dose reductions were more common with palbociclib than with placebo,” they added.
Myelotoxic effects have been managed successfully with appropriate supportive care and dose reductions. At the time of this analysis, 199 patients (44.8%) in the active-treatment group were still taking palbociclib and letrozole, the investigators said.
This study was supported by Pfizer. Dr. Finn reported receiving grants, personal fees, and other support from Pfizer, Bayer, Novartis, and Bristol-Myers Squibb. His associates reported ties to numerous industry sources.
Inhibition of CDK4 and CDK6, in combination with antiestrogens, is clearly the new standard of treatment for advanced ER-positive breast cancer.
Antonio C. Wolff, MD, is professor of oncology at Johns Hopkins University’s Sidney Kimmel Comprehensive Cancer Center, Baltimore, and executive officer of the Translational Breast Cancer Research Consortium. He reported ties to Pfizer. Dr. Wolff made these remarks in an editorial accompanying Dr. Finn’s report (N Engl J Med. 2016 Nov 17. doi: 10.1056/NEJMe1611926).
Inhibition of CDK4 and CDK6, in combination with antiestrogens, is clearly the new standard of treatment for advanced ER-positive breast cancer.
Antonio C. Wolff, MD, is professor of oncology at Johns Hopkins University’s Sidney Kimmel Comprehensive Cancer Center, Baltimore, and executive officer of the Translational Breast Cancer Research Consortium. He reported ties to Pfizer. Dr. Wolff made these remarks in an editorial accompanying Dr. Finn’s report (N Engl J Med. 2016 Nov 17. doi: 10.1056/NEJMe1611926).
Inhibition of CDK4 and CDK6, in combination with antiestrogens, is clearly the new standard of treatment for advanced ER-positive breast cancer.
Antonio C. Wolff, MD, is professor of oncology at Johns Hopkins University’s Sidney Kimmel Comprehensive Cancer Center, Baltimore, and executive officer of the Translational Breast Cancer Research Consortium. He reported ties to Pfizer. Dr. Wolff made these remarks in an editorial accompanying Dr. Finn’s report (N Engl J Med. 2016 Nov 17. doi: 10.1056/NEJMe1611926).
Adding the CDK inhibitor palbociclib to standard endocrine therapy (letrozole) extended progression-free survival in a manufacturer-sponsored phase III trial of advanced ER-positive, HER2-negative breast cancer, investigators reported in the New England Journal of Medicine.
Compared with placebo, palbociclib significantly improved progression-free survival regardless of patient age, patient performance status, site of metastasis, prior use of chemotherapy, prior use of endocrine therapy, length of the disease-free interval before progression, or the cancer’s histologic subtype. “The median progression-free survival of 24.8 months in [our study] is longer than that seen in other phase III studies involving women with advanced breast cancer. Whether this progression-free survival will result in longer overall survival is uncertain until further follow-up is completed,” wrote Richard S. Finn, MD, of the University of California, Los Angeles, and his associates.
The primary endpoint – time to disease progression or death – was 24.8 months with active treatment, compared with 14.5 months with placebo, for a hazard ratio of 0.58. The rate of disease progression or death was 43.7% in the palbociclib group, compared with 61.7% in the placebo group. In addition, the rate of objective tumor response was 42.1% with palbociclib, compared with 34.7% with placebo, they reported.
“Data on overall survival were immature at the time of this analysis of the primary end point, and the final overall survival analysis will be performed when a total of 390 deaths occur, per protocol and in agreement with regulatory agencies,” Dr. Finn and his associates said (N Engl J Med. 2016 Nov. 17. doi: 10.1056/NEJMoa1607303). As expected, the rate of hematologic adverse events was high with palbociclib. “Although the incidence of neutropenia of any grade in the palbobiclib/letrozole group was 79.5% in the current study, the incidence of febrile neutropenia was lower than 2%. In addition, the rate of permanent treatment discontinuation associated with an adverse event did not differ significantly between the two study groups, although dose reductions were more common with palbociclib than with placebo,” they added.
Myelotoxic effects have been managed successfully with appropriate supportive care and dose reductions. At the time of this analysis, 199 patients (44.8%) in the active-treatment group were still taking palbociclib and letrozole, the investigators said.
This study was supported by Pfizer. Dr. Finn reported receiving grants, personal fees, and other support from Pfizer, Bayer, Novartis, and Bristol-Myers Squibb. His associates reported ties to numerous industry sources.
Adding the CDK inhibitor palbociclib to standard endocrine therapy (letrozole) extended progression-free survival in a manufacturer-sponsored phase III trial of advanced ER-positive, HER2-negative breast cancer, investigators reported in the New England Journal of Medicine.
Compared with placebo, palbociclib significantly improved progression-free survival regardless of patient age, patient performance status, site of metastasis, prior use of chemotherapy, prior use of endocrine therapy, length of the disease-free interval before progression, or the cancer’s histologic subtype. “The median progression-free survival of 24.8 months in [our study] is longer than that seen in other phase III studies involving women with advanced breast cancer. Whether this progression-free survival will result in longer overall survival is uncertain until further follow-up is completed,” wrote Richard S. Finn, MD, of the University of California, Los Angeles, and his associates.
The primary endpoint – time to disease progression or death – was 24.8 months with active treatment, compared with 14.5 months with placebo, for a hazard ratio of 0.58. The rate of disease progression or death was 43.7% in the palbociclib group, compared with 61.7% in the placebo group. In addition, the rate of objective tumor response was 42.1% with palbociclib, compared with 34.7% with placebo, they reported.
“Data on overall survival were immature at the time of this analysis of the primary end point, and the final overall survival analysis will be performed when a total of 390 deaths occur, per protocol and in agreement with regulatory agencies,” Dr. Finn and his associates said (N Engl J Med. 2016 Nov. 17. doi: 10.1056/NEJMoa1607303). As expected, the rate of hematologic adverse events was high with palbociclib. “Although the incidence of neutropenia of any grade in the palbobiclib/letrozole group was 79.5% in the current study, the incidence of febrile neutropenia was lower than 2%. In addition, the rate of permanent treatment discontinuation associated with an adverse event did not differ significantly between the two study groups, although dose reductions were more common with palbociclib than with placebo,” they added.
Myelotoxic effects have been managed successfully with appropriate supportive care and dose reductions. At the time of this analysis, 199 patients (44.8%) in the active-treatment group were still taking palbociclib and letrozole, the investigators said.
This study was supported by Pfizer. Dr. Finn reported receiving grants, personal fees, and other support from Pfizer, Bayer, Novartis, and Bristol-Myers Squibb. His associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point:
Major finding: The primary endpoint – time to disease progression or death – was 24.8 months with active treatment, compared with 14.5 months with placebo, for an hazard ratio of 0.58.
Data source: An international, randomized, double-blind phase III clinical trial involving 666 women.
Disclosures: This study was supported by Pfizer. Dr. Finn reported receiving grants, personal fees, and other support from Pfizer, Bayer, Novartis, and Bristol-Myers Squibb. His associates reported ties to numerous industry sources.