User login
ACC 21 looks to repeat success despite pandemic headwinds
The American College of Cardiology pulled off an impressive all-virtual meeting in March 2020, less than 3 weeks after canceling its in-person event and just 2 weeks after COVID-19 was declared a national emergency.
Optimistic plans for the annual scientific sessions of the American College of Cardiology (ACC 2021) to be a March hybrid affair in Atlanta pivoted not once, but twice, as the pandemic evolved, with the date pushed back 2 full months, to May 15-17, and the format revised to fully virtual.
“While this meeting is being delivered virtually, I think you’ll see there have been benefits in the time to plan and also the lessons that ACC has learned in virtual education over the past year. This has come together to really create a robust educational and scientific agenda,” ACC 2021 chair Pamela B. Morris, MD, said in a press conference focused on the upcoming meeting.
Over the 3 days, there will be more than 200 education sessions, 10 guideline-specific sessions, and 11 learning pathways that include core areas, but also special topics, such as COVID-19 and the emerging cardio-obstetrics subspecialty.
The meeting will be delivered through a new virtual education program built to optimize real-time interaction between faculty members and attendees, she said. A dedicated portal on the platform will allow attendees to interact virtually, for example, with presenters of the nearly 3,000 ePosters and 420 moderated posters.
For those suffering from Zoom fatigue, the increasingly popular Heart2Heart stage talks have also been converted to podcasts, which cover topics like gender equity in cardiology, the evolving role of advanced practice professionals, and “one of my favorites: art as a tool for healing,” said Dr. Morris, from the Medical University of South Carolina, Charleston. “Those sessions are really not to be missed.”
Reconnecting is an underlying theme of the meeting but the great divider will not be ignored. COVID-19 will be the focus of two 90-minute Intensive Sessions on Saturday, May 15, the first kicking off at 10:30 a.m. ET, with the Bishop Keynote lecture on bringing health equity to the frontline of cardiovascular care, followed by lessons learned during the pandemic, how to conduct clinical trials, and vaccine development.
The second session, set for 12:15 p.m., continues the “silver linings” theme, with case presentations on advances in telehealth, myocardial involvement, and thrombosis in COVID. For those wanting more, 18 abstracts are on tap in a 2-hour Spotlight on Special Topics session beginning at 2:30 p.m.
Asked about the pandemic’s effect on bringing science to fruition this past year, Dr. Morris said there’s no question it’s slowed some of the progress the cardiology community had made but, like clinical practice, “we’ve also surmounted many of those obstacles.”
“I think research has rebounded,” she said. “Just in terms of the number of abstracts and the quality of abstracts that were submitted this year, I don’t think there’s any question that we are right on par with previous years.”
Indeed, 5,258 abstracts from 76 countries were submitted, with more than 3,400 chosen for oral and poster presentation, including 25 late-breaking clinical trials to be presented in five sessions.
The late-breaking presentations and discussions will be prerecorded but speakers and panelists have been invited to be present during the streaming to answer live any questions that may arise in the chat box, ACC 2021 vice chair Douglas Drachman, MD, Massachusetts General Hospital, Boston, said in an interview.
Late-breaking clinical trials
The Joint ACC/JACC Late-Breaking Clinical Trials I (Saturday, May 15, 9:00 a.m.–-10:00 a.m.) kicks off with PARADISE-MI, the first head-to-head comparison of an angiotensin receptor neprilysin inhibitor (ARNI) and an ACE inhibitor in patients with reduced ejection fractions (EFs) after MI but no history of heart failure (HF), studying 200 mg sacubitril/valsartan (Entresto) versus 5 mg of ramipril, both twice daily, in 5,669 patients.
Sacubitril/valsartan was initially approved for HF with reduced EF and added a new indication to treat some HF patients with preserved EF. Novartis, however, recently told investors that although numerical trends consistently favored the ARNI over the ACE inhibitor ramipril, the phase 3 study failed to meet the primary endpoint for efficacy superiority of reducing the risk for cardiovascular (CV) death and HF events after an acute MI.
Second up is ADAPTABLE, which looks to close a surprising evidence gap over whether 81 mg or 325 mg daily is the optimal dose of the ubiquitously prescribed aspirin for secondary prevention in high-risk patients with established atherosclerotic CV disease.
The open-label, randomized study will look at efficacy and major bleeding over roughly 4 years in 15,000 patients within PCORnet, the National Patient-centered Clinical Research Network, a partnership of clinical research, health plan research, and patient-powered networks created to streamline patient-reported outcomes research.
“This study will not only give important clinical information for us, practically speaking, whether we should prescribe lower- or higher-dose aspirin, but it may also serve as a template for future pragmatic clinical trial design in the real world,” Dr. Drachman said during the press conference.
Up next is the 4,812-patient Canadian LAAOS III, the largest trial to examine the efficacy of left atrial appendage occlusion for stroke prevention in patients with atrial fibrillation (AFib) already undergoing cardiac surgery. The primary outcome is the first occurrence of stroke or systemic arterial embolism over an average follow-up of 4 years.
Percutaneous closure of the left atrial appendage (LAA) has been shown to reduce stroke in AFib patients at high-risk of bleeding on systemic anticoagulation. But these devices can be expensive and studies haven’t included patients who also have valvular heart disease, a group that actually comprises more than half of patients undergoing cardiac surgery who also have AFib, he noted.
At the same time, surgical LAA closure studies have been small and have had very mixed results. “There isn’t a large-scale rigorous assessment out there for these patients undergoing surgery, so I think this is going to be fascinating to see,” Dr. Drachman said.
The session closes with ATLANTIS, which looks to shed some light on the role of anticoagulation therapy in patients after transcatheter aortic valve replacement (TAVR or TAVI). POPular TAVI, presented at ACC 2020, showed aspirin alone was the preferred antithrombotic therapy over aspirin plus clopidogrel (Plavix) in patients not on oral anticoagulants, but the optimal anticoagulation regimen remains unsettled.
The French open-label, 1,510-patient ATLANTIS trial examined whether the novel oral anticoagulant apixaban (Eliquis) is superior in preventing CV events after TAVR, compared with antiplatelet therapy in patients without an indication for anticoagulation and compared with vitamin K antagonists in those receiving anticoagulants.
An ATLANTIS 4D CT substudy of valve thrombosis is also slated for Saturday’s Featured Clinical Research 1 session at 12:15 p.m. to 1:45 p.m..
Sunday LBCTs
Dr. Drachman highlighted a series of other late-breaking studies, including the global DARE-19 trial testing the diabetes and HF drug dapagliflozin (Farxiga) given with local standard-of-care therapy for 30 days in hospitalized COVID-19 patients with CV, metabolic, or renal risk factors.
Although sodium-glucose cotransporter-2 inhibitors have been white-hot of late, top-line results reported last month show dapagliflozin failed to achieve statistical significance for the primary endpoints of reducing organ dysfunction and all-cause mortality and for improving recovery. Details will be presented in the Joint ACC/JAMA Late-Breaking Clinical Trials II (Sunday, May 16, 8:00 a.m.-9:30 a.m.).
Two trials, FLOWER-MI and RADIANCE-HTN TRIO, were singled out in the Joint ACC/New England Journal of Medicine Late-Breaking Clinical Trials III (Sunday, May 16, 10:45 a.m.-12:00 p.m.). FLOWER-MI examines whether fractional flow reserve (FFR) is better than angiography to guide complete multivessel revascularization in ST-elevation MI patients with at least 50% stenosis in at least one nonculprit lesion requiring percutaneous coronary intervention (PCI). Recent studies have shown the superiority of FFR-guided PCI for nonculprit lesions, compared with culprit lesion treatment-only, but this is the first time FFR- and angiography-guided PCI have been compared in STEMI patients.
RADIANCE-HTN TRIO already tipped its hand, with top-line results reported in late 2020 showing that the trial met its primary efficacy endpoint of greater reduction in daytime blood pressure over 2 months with the Paradise endovascular ultrasound renal denervation system, compared with a sham procedure, in 136 patients with resistant hypertension, importantly, after being given a single pill containing a calcium channel blocker, angiotensin II receptor blocker, and diuretic.
Renal denervation for hypertension has been making something of a comeback, with the 2018 RADIANCE-HTN SOLO reporting better ambulatory blood pressure control with the Paradise system than with a sham procedure in the absence of antihypertensive agents. The device has been granted breakthrough device designation from the Food and Drug Administration for the treatment of hypertensive patients who are unable to sufficiently respond to or are intolerant of antihypertensive therapy.
Monday LBCTs
In the Late-Breaking Clinical Trials IV session (Monday, May 17, 8 a.m.–9:30 a.m.), Drachman called out a secondary analysis from GALATIC-HF looking at the impact of EF on the therapeutic effect of omecamtiv mecarbil. In last year’s primary analysis, the selective cardiac myosin activator produced a modest but significant reduction in HF events or CV death in 8,232 patients with HF and an EF of 35% or less.
Rounding out the list is the Canadian CAPITAL CHILL study of moderate versus mild therapeutic hypothermia in out-of-hospital cardiac arrest, to be presented in the final Late-Breaking Clinical Trials V session (Monday, May 17, 10:45 a.m.–12:00 p.m.).
The double-blind trial sought to determine whether neurologic outcomes at 6 months are improved by targeting a core temperature of 31 ˚C versus 34 ˚C after the return of spontaneous circulation in comatose survivors of out-of-hospital cardiac arrest.
“For me, I think this could really change practice and has personal relevance from experience with cardiac arrest survivors that I’ve known and care for very deeply,” Dr. Drachman said in an interview. “I think that there’s a lot of opportunity here as well.”
Asked what other trials have the potential to change practice, Dr. Drachman said FLOWER-MI holds particular interest because it looks at how to manage patients with STEMI with multiple lesions at the point of care.
“We’ve gained a lot of clarity from several other prior clinical trials, but this will help to answer the question in a slightly different way of saying: can you eyeball it, can you look at the angiogram and say whether or not that other, nonculprit lesion ought to be treated in the same hospitalization or should you really be using a pressure wire,” he said. “For me as an interventionalist, this is really important because when you finish up doing an intervention on a patient it might be the middle of the night and the patient may be more or less stable, but you’ve already exposed them to the risk of a procedure, should you then move on and do another aspect of the procedure to interrogate with a pressure wire a remaining narrowing? I think that’s very important; that’ll help me make decisions on a day-to-day basis.”
Dr. Drachman also cited RADIANCE-HTN TRIO because it employs an endovascular technique to control blood pressure in patients with hypertension, specifically those resistant to multiple drugs.
During the press conference, Dr. Morris, a preventive cardiologist, put her money on the ADAPTABLE study of aspirin dosing, reiterating that the unique trial design could inform future research, and on Sunday’s 8:45 a.m. late-breaking post hoc analysis from the STRENGTH trial that looks to pick up where the controversy over omega-3 fatty acid preparations left off at last year’s American Heart Association meeting.
A lack of benefit on CV event rates reported with Epanova, a high-dose combination of eicosapentaenoic acid (EPA) and docosahexaenoic acid, led to a contentious debate over how to reconcile STRENGTH with the findings from REDUCE-IT, which showed a 25% relative risk reduction in major CV events with the EPA product icosapent ethyl (Vascepa).
STRENGTH investigator Steven Nissen, MD, Cleveland Clinic, and REDUCE-IT investigator and session panelist Deepak Bhatt, MD, Brigham and Women’s Hospital, Boston, will share the virtual stage at ACC 2021, but Dr. Morris said the “good news” is both researchers know one another very well and “will really be focusing on no political issues, just the omega-3 fatty levels in the bloodstream and what does that mean in either trial.
“This is not designed to be a debate, point counterpoint,” she added.
For that, as all cardiologists and journalists know, there will be the wild and woolly #CardioTwitter sphere.
A version of this article first appeared on Medscape.com.
The American College of Cardiology pulled off an impressive all-virtual meeting in March 2020, less than 3 weeks after canceling its in-person event and just 2 weeks after COVID-19 was declared a national emergency.
Optimistic plans for the annual scientific sessions of the American College of Cardiology (ACC 2021) to be a March hybrid affair in Atlanta pivoted not once, but twice, as the pandemic evolved, with the date pushed back 2 full months, to May 15-17, and the format revised to fully virtual.
“While this meeting is being delivered virtually, I think you’ll see there have been benefits in the time to plan and also the lessons that ACC has learned in virtual education over the past year. This has come together to really create a robust educational and scientific agenda,” ACC 2021 chair Pamela B. Morris, MD, said in a press conference focused on the upcoming meeting.
Over the 3 days, there will be more than 200 education sessions, 10 guideline-specific sessions, and 11 learning pathways that include core areas, but also special topics, such as COVID-19 and the emerging cardio-obstetrics subspecialty.
The meeting will be delivered through a new virtual education program built to optimize real-time interaction between faculty members and attendees, she said. A dedicated portal on the platform will allow attendees to interact virtually, for example, with presenters of the nearly 3,000 ePosters and 420 moderated posters.
For those suffering from Zoom fatigue, the increasingly popular Heart2Heart stage talks have also been converted to podcasts, which cover topics like gender equity in cardiology, the evolving role of advanced practice professionals, and “one of my favorites: art as a tool for healing,” said Dr. Morris, from the Medical University of South Carolina, Charleston. “Those sessions are really not to be missed.”
Reconnecting is an underlying theme of the meeting but the great divider will not be ignored. COVID-19 will be the focus of two 90-minute Intensive Sessions on Saturday, May 15, the first kicking off at 10:30 a.m. ET, with the Bishop Keynote lecture on bringing health equity to the frontline of cardiovascular care, followed by lessons learned during the pandemic, how to conduct clinical trials, and vaccine development.
The second session, set for 12:15 p.m., continues the “silver linings” theme, with case presentations on advances in telehealth, myocardial involvement, and thrombosis in COVID. For those wanting more, 18 abstracts are on tap in a 2-hour Spotlight on Special Topics session beginning at 2:30 p.m.
Asked about the pandemic’s effect on bringing science to fruition this past year, Dr. Morris said there’s no question it’s slowed some of the progress the cardiology community had made but, like clinical practice, “we’ve also surmounted many of those obstacles.”
“I think research has rebounded,” she said. “Just in terms of the number of abstracts and the quality of abstracts that were submitted this year, I don’t think there’s any question that we are right on par with previous years.”
Indeed, 5,258 abstracts from 76 countries were submitted, with more than 3,400 chosen for oral and poster presentation, including 25 late-breaking clinical trials to be presented in five sessions.
The late-breaking presentations and discussions will be prerecorded but speakers and panelists have been invited to be present during the streaming to answer live any questions that may arise in the chat box, ACC 2021 vice chair Douglas Drachman, MD, Massachusetts General Hospital, Boston, said in an interview.
Late-breaking clinical trials
The Joint ACC/JACC Late-Breaking Clinical Trials I (Saturday, May 15, 9:00 a.m.–-10:00 a.m.) kicks off with PARADISE-MI, the first head-to-head comparison of an angiotensin receptor neprilysin inhibitor (ARNI) and an ACE inhibitor in patients with reduced ejection fractions (EFs) after MI but no history of heart failure (HF), studying 200 mg sacubitril/valsartan (Entresto) versus 5 mg of ramipril, both twice daily, in 5,669 patients.
Sacubitril/valsartan was initially approved for HF with reduced EF and added a new indication to treat some HF patients with preserved EF. Novartis, however, recently told investors that although numerical trends consistently favored the ARNI over the ACE inhibitor ramipril, the phase 3 study failed to meet the primary endpoint for efficacy superiority of reducing the risk for cardiovascular (CV) death and HF events after an acute MI.
Second up is ADAPTABLE, which looks to close a surprising evidence gap over whether 81 mg or 325 mg daily is the optimal dose of the ubiquitously prescribed aspirin for secondary prevention in high-risk patients with established atherosclerotic CV disease.
The open-label, randomized study will look at efficacy and major bleeding over roughly 4 years in 15,000 patients within PCORnet, the National Patient-centered Clinical Research Network, a partnership of clinical research, health plan research, and patient-powered networks created to streamline patient-reported outcomes research.
“This study will not only give important clinical information for us, practically speaking, whether we should prescribe lower- or higher-dose aspirin, but it may also serve as a template for future pragmatic clinical trial design in the real world,” Dr. Drachman said during the press conference.
Up next is the 4,812-patient Canadian LAAOS III, the largest trial to examine the efficacy of left atrial appendage occlusion for stroke prevention in patients with atrial fibrillation (AFib) already undergoing cardiac surgery. The primary outcome is the first occurrence of stroke or systemic arterial embolism over an average follow-up of 4 years.
Percutaneous closure of the left atrial appendage (LAA) has been shown to reduce stroke in AFib patients at high-risk of bleeding on systemic anticoagulation. But these devices can be expensive and studies haven’t included patients who also have valvular heart disease, a group that actually comprises more than half of patients undergoing cardiac surgery who also have AFib, he noted.
At the same time, surgical LAA closure studies have been small and have had very mixed results. “There isn’t a large-scale rigorous assessment out there for these patients undergoing surgery, so I think this is going to be fascinating to see,” Dr. Drachman said.
The session closes with ATLANTIS, which looks to shed some light on the role of anticoagulation therapy in patients after transcatheter aortic valve replacement (TAVR or TAVI). POPular TAVI, presented at ACC 2020, showed aspirin alone was the preferred antithrombotic therapy over aspirin plus clopidogrel (Plavix) in patients not on oral anticoagulants, but the optimal anticoagulation regimen remains unsettled.
The French open-label, 1,510-patient ATLANTIS trial examined whether the novel oral anticoagulant apixaban (Eliquis) is superior in preventing CV events after TAVR, compared with antiplatelet therapy in patients without an indication for anticoagulation and compared with vitamin K antagonists in those receiving anticoagulants.
An ATLANTIS 4D CT substudy of valve thrombosis is also slated for Saturday’s Featured Clinical Research 1 session at 12:15 p.m. to 1:45 p.m..
Sunday LBCTs
Dr. Drachman highlighted a series of other late-breaking studies, including the global DARE-19 trial testing the diabetes and HF drug dapagliflozin (Farxiga) given with local standard-of-care therapy for 30 days in hospitalized COVID-19 patients with CV, metabolic, or renal risk factors.
Although sodium-glucose cotransporter-2 inhibitors have been white-hot of late, top-line results reported last month show dapagliflozin failed to achieve statistical significance for the primary endpoints of reducing organ dysfunction and all-cause mortality and for improving recovery. Details will be presented in the Joint ACC/JAMA Late-Breaking Clinical Trials II (Sunday, May 16, 8:00 a.m.-9:30 a.m.).
Two trials, FLOWER-MI and RADIANCE-HTN TRIO, were singled out in the Joint ACC/New England Journal of Medicine Late-Breaking Clinical Trials III (Sunday, May 16, 10:45 a.m.-12:00 p.m.). FLOWER-MI examines whether fractional flow reserve (FFR) is better than angiography to guide complete multivessel revascularization in ST-elevation MI patients with at least 50% stenosis in at least one nonculprit lesion requiring percutaneous coronary intervention (PCI). Recent studies have shown the superiority of FFR-guided PCI for nonculprit lesions, compared with culprit lesion treatment-only, but this is the first time FFR- and angiography-guided PCI have been compared in STEMI patients.
RADIANCE-HTN TRIO already tipped its hand, with top-line results reported in late 2020 showing that the trial met its primary efficacy endpoint of greater reduction in daytime blood pressure over 2 months with the Paradise endovascular ultrasound renal denervation system, compared with a sham procedure, in 136 patients with resistant hypertension, importantly, after being given a single pill containing a calcium channel blocker, angiotensin II receptor blocker, and diuretic.
Renal denervation for hypertension has been making something of a comeback, with the 2018 RADIANCE-HTN SOLO reporting better ambulatory blood pressure control with the Paradise system than with a sham procedure in the absence of antihypertensive agents. The device has been granted breakthrough device designation from the Food and Drug Administration for the treatment of hypertensive patients who are unable to sufficiently respond to or are intolerant of antihypertensive therapy.
Monday LBCTs
In the Late-Breaking Clinical Trials IV session (Monday, May 17, 8 a.m.–9:30 a.m.), Drachman called out a secondary analysis from GALATIC-HF looking at the impact of EF on the therapeutic effect of omecamtiv mecarbil. In last year’s primary analysis, the selective cardiac myosin activator produced a modest but significant reduction in HF events or CV death in 8,232 patients with HF and an EF of 35% or less.
Rounding out the list is the Canadian CAPITAL CHILL study of moderate versus mild therapeutic hypothermia in out-of-hospital cardiac arrest, to be presented in the final Late-Breaking Clinical Trials V session (Monday, May 17, 10:45 a.m.–12:00 p.m.).
The double-blind trial sought to determine whether neurologic outcomes at 6 months are improved by targeting a core temperature of 31 ˚C versus 34 ˚C after the return of spontaneous circulation in comatose survivors of out-of-hospital cardiac arrest.
“For me, I think this could really change practice and has personal relevance from experience with cardiac arrest survivors that I’ve known and care for very deeply,” Dr. Drachman said in an interview. “I think that there’s a lot of opportunity here as well.”
Asked what other trials have the potential to change practice, Dr. Drachman said FLOWER-MI holds particular interest because it looks at how to manage patients with STEMI with multiple lesions at the point of care.
“We’ve gained a lot of clarity from several other prior clinical trials, but this will help to answer the question in a slightly different way of saying: can you eyeball it, can you look at the angiogram and say whether or not that other, nonculprit lesion ought to be treated in the same hospitalization or should you really be using a pressure wire,” he said. “For me as an interventionalist, this is really important because when you finish up doing an intervention on a patient it might be the middle of the night and the patient may be more or less stable, but you’ve already exposed them to the risk of a procedure, should you then move on and do another aspect of the procedure to interrogate with a pressure wire a remaining narrowing? I think that’s very important; that’ll help me make decisions on a day-to-day basis.”
Dr. Drachman also cited RADIANCE-HTN TRIO because it employs an endovascular technique to control blood pressure in patients with hypertension, specifically those resistant to multiple drugs.
During the press conference, Dr. Morris, a preventive cardiologist, put her money on the ADAPTABLE study of aspirin dosing, reiterating that the unique trial design could inform future research, and on Sunday’s 8:45 a.m. late-breaking post hoc analysis from the STRENGTH trial that looks to pick up where the controversy over omega-3 fatty acid preparations left off at last year’s American Heart Association meeting.
A lack of benefit on CV event rates reported with Epanova, a high-dose combination of eicosapentaenoic acid (EPA) and docosahexaenoic acid, led to a contentious debate over how to reconcile STRENGTH with the findings from REDUCE-IT, which showed a 25% relative risk reduction in major CV events with the EPA product icosapent ethyl (Vascepa).
STRENGTH investigator Steven Nissen, MD, Cleveland Clinic, and REDUCE-IT investigator and session panelist Deepak Bhatt, MD, Brigham and Women’s Hospital, Boston, will share the virtual stage at ACC 2021, but Dr. Morris said the “good news” is both researchers know one another very well and “will really be focusing on no political issues, just the omega-3 fatty levels in the bloodstream and what does that mean in either trial.
“This is not designed to be a debate, point counterpoint,” she added.
For that, as all cardiologists and journalists know, there will be the wild and woolly #CardioTwitter sphere.
A version of this article first appeared on Medscape.com.
The American College of Cardiology pulled off an impressive all-virtual meeting in March 2020, less than 3 weeks after canceling its in-person event and just 2 weeks after COVID-19 was declared a national emergency.
Optimistic plans for the annual scientific sessions of the American College of Cardiology (ACC 2021) to be a March hybrid affair in Atlanta pivoted not once, but twice, as the pandemic evolved, with the date pushed back 2 full months, to May 15-17, and the format revised to fully virtual.
“While this meeting is being delivered virtually, I think you’ll see there have been benefits in the time to plan and also the lessons that ACC has learned in virtual education over the past year. This has come together to really create a robust educational and scientific agenda,” ACC 2021 chair Pamela B. Morris, MD, said in a press conference focused on the upcoming meeting.
Over the 3 days, there will be more than 200 education sessions, 10 guideline-specific sessions, and 11 learning pathways that include core areas, but also special topics, such as COVID-19 and the emerging cardio-obstetrics subspecialty.
The meeting will be delivered through a new virtual education program built to optimize real-time interaction between faculty members and attendees, she said. A dedicated portal on the platform will allow attendees to interact virtually, for example, with presenters of the nearly 3,000 ePosters and 420 moderated posters.
For those suffering from Zoom fatigue, the increasingly popular Heart2Heart stage talks have also been converted to podcasts, which cover topics like gender equity in cardiology, the evolving role of advanced practice professionals, and “one of my favorites: art as a tool for healing,” said Dr. Morris, from the Medical University of South Carolina, Charleston. “Those sessions are really not to be missed.”
Reconnecting is an underlying theme of the meeting but the great divider will not be ignored. COVID-19 will be the focus of two 90-minute Intensive Sessions on Saturday, May 15, the first kicking off at 10:30 a.m. ET, with the Bishop Keynote lecture on bringing health equity to the frontline of cardiovascular care, followed by lessons learned during the pandemic, how to conduct clinical trials, and vaccine development.
The second session, set for 12:15 p.m., continues the “silver linings” theme, with case presentations on advances in telehealth, myocardial involvement, and thrombosis in COVID. For those wanting more, 18 abstracts are on tap in a 2-hour Spotlight on Special Topics session beginning at 2:30 p.m.
Asked about the pandemic’s effect on bringing science to fruition this past year, Dr. Morris said there’s no question it’s slowed some of the progress the cardiology community had made but, like clinical practice, “we’ve also surmounted many of those obstacles.”
“I think research has rebounded,” she said. “Just in terms of the number of abstracts and the quality of abstracts that were submitted this year, I don’t think there’s any question that we are right on par with previous years.”
Indeed, 5,258 abstracts from 76 countries were submitted, with more than 3,400 chosen for oral and poster presentation, including 25 late-breaking clinical trials to be presented in five sessions.
The late-breaking presentations and discussions will be prerecorded but speakers and panelists have been invited to be present during the streaming to answer live any questions that may arise in the chat box, ACC 2021 vice chair Douglas Drachman, MD, Massachusetts General Hospital, Boston, said in an interview.
Late-breaking clinical trials
The Joint ACC/JACC Late-Breaking Clinical Trials I (Saturday, May 15, 9:00 a.m.–-10:00 a.m.) kicks off with PARADISE-MI, the first head-to-head comparison of an angiotensin receptor neprilysin inhibitor (ARNI) and an ACE inhibitor in patients with reduced ejection fractions (EFs) after MI but no history of heart failure (HF), studying 200 mg sacubitril/valsartan (Entresto) versus 5 mg of ramipril, both twice daily, in 5,669 patients.
Sacubitril/valsartan was initially approved for HF with reduced EF and added a new indication to treat some HF patients with preserved EF. Novartis, however, recently told investors that although numerical trends consistently favored the ARNI over the ACE inhibitor ramipril, the phase 3 study failed to meet the primary endpoint for efficacy superiority of reducing the risk for cardiovascular (CV) death and HF events after an acute MI.
Second up is ADAPTABLE, which looks to close a surprising evidence gap over whether 81 mg or 325 mg daily is the optimal dose of the ubiquitously prescribed aspirin for secondary prevention in high-risk patients with established atherosclerotic CV disease.
The open-label, randomized study will look at efficacy and major bleeding over roughly 4 years in 15,000 patients within PCORnet, the National Patient-centered Clinical Research Network, a partnership of clinical research, health plan research, and patient-powered networks created to streamline patient-reported outcomes research.
“This study will not only give important clinical information for us, practically speaking, whether we should prescribe lower- or higher-dose aspirin, but it may also serve as a template for future pragmatic clinical trial design in the real world,” Dr. Drachman said during the press conference.
Up next is the 4,812-patient Canadian LAAOS III, the largest trial to examine the efficacy of left atrial appendage occlusion for stroke prevention in patients with atrial fibrillation (AFib) already undergoing cardiac surgery. The primary outcome is the first occurrence of stroke or systemic arterial embolism over an average follow-up of 4 years.
Percutaneous closure of the left atrial appendage (LAA) has been shown to reduce stroke in AFib patients at high-risk of bleeding on systemic anticoagulation. But these devices can be expensive and studies haven’t included patients who also have valvular heart disease, a group that actually comprises more than half of patients undergoing cardiac surgery who also have AFib, he noted.
At the same time, surgical LAA closure studies have been small and have had very mixed results. “There isn’t a large-scale rigorous assessment out there for these patients undergoing surgery, so I think this is going to be fascinating to see,” Dr. Drachman said.
The session closes with ATLANTIS, which looks to shed some light on the role of anticoagulation therapy in patients after transcatheter aortic valve replacement (TAVR or TAVI). POPular TAVI, presented at ACC 2020, showed aspirin alone was the preferred antithrombotic therapy over aspirin plus clopidogrel (Plavix) in patients not on oral anticoagulants, but the optimal anticoagulation regimen remains unsettled.
The French open-label, 1,510-patient ATLANTIS trial examined whether the novel oral anticoagulant apixaban (Eliquis) is superior in preventing CV events after TAVR, compared with antiplatelet therapy in patients without an indication for anticoagulation and compared with vitamin K antagonists in those receiving anticoagulants.
An ATLANTIS 4D CT substudy of valve thrombosis is also slated for Saturday’s Featured Clinical Research 1 session at 12:15 p.m. to 1:45 p.m..
Sunday LBCTs
Dr. Drachman highlighted a series of other late-breaking studies, including the global DARE-19 trial testing the diabetes and HF drug dapagliflozin (Farxiga) given with local standard-of-care therapy for 30 days in hospitalized COVID-19 patients with CV, metabolic, or renal risk factors.
Although sodium-glucose cotransporter-2 inhibitors have been white-hot of late, top-line results reported last month show dapagliflozin failed to achieve statistical significance for the primary endpoints of reducing organ dysfunction and all-cause mortality and for improving recovery. Details will be presented in the Joint ACC/JAMA Late-Breaking Clinical Trials II (Sunday, May 16, 8:00 a.m.-9:30 a.m.).
Two trials, FLOWER-MI and RADIANCE-HTN TRIO, were singled out in the Joint ACC/New England Journal of Medicine Late-Breaking Clinical Trials III (Sunday, May 16, 10:45 a.m.-12:00 p.m.). FLOWER-MI examines whether fractional flow reserve (FFR) is better than angiography to guide complete multivessel revascularization in ST-elevation MI patients with at least 50% stenosis in at least one nonculprit lesion requiring percutaneous coronary intervention (PCI). Recent studies have shown the superiority of FFR-guided PCI for nonculprit lesions, compared with culprit lesion treatment-only, but this is the first time FFR- and angiography-guided PCI have been compared in STEMI patients.
RADIANCE-HTN TRIO already tipped its hand, with top-line results reported in late 2020 showing that the trial met its primary efficacy endpoint of greater reduction in daytime blood pressure over 2 months with the Paradise endovascular ultrasound renal denervation system, compared with a sham procedure, in 136 patients with resistant hypertension, importantly, after being given a single pill containing a calcium channel blocker, angiotensin II receptor blocker, and diuretic.
Renal denervation for hypertension has been making something of a comeback, with the 2018 RADIANCE-HTN SOLO reporting better ambulatory blood pressure control with the Paradise system than with a sham procedure in the absence of antihypertensive agents. The device has been granted breakthrough device designation from the Food and Drug Administration for the treatment of hypertensive patients who are unable to sufficiently respond to or are intolerant of antihypertensive therapy.
Monday LBCTs
In the Late-Breaking Clinical Trials IV session (Monday, May 17, 8 a.m.–9:30 a.m.), Drachman called out a secondary analysis from GALATIC-HF looking at the impact of EF on the therapeutic effect of omecamtiv mecarbil. In last year’s primary analysis, the selective cardiac myosin activator produced a modest but significant reduction in HF events or CV death in 8,232 patients with HF and an EF of 35% or less.
Rounding out the list is the Canadian CAPITAL CHILL study of moderate versus mild therapeutic hypothermia in out-of-hospital cardiac arrest, to be presented in the final Late-Breaking Clinical Trials V session (Monday, May 17, 10:45 a.m.–12:00 p.m.).
The double-blind trial sought to determine whether neurologic outcomes at 6 months are improved by targeting a core temperature of 31 ˚C versus 34 ˚C after the return of spontaneous circulation in comatose survivors of out-of-hospital cardiac arrest.
“For me, I think this could really change practice and has personal relevance from experience with cardiac arrest survivors that I’ve known and care for very deeply,” Dr. Drachman said in an interview. “I think that there’s a lot of opportunity here as well.”
Asked what other trials have the potential to change practice, Dr. Drachman said FLOWER-MI holds particular interest because it looks at how to manage patients with STEMI with multiple lesions at the point of care.
“We’ve gained a lot of clarity from several other prior clinical trials, but this will help to answer the question in a slightly different way of saying: can you eyeball it, can you look at the angiogram and say whether or not that other, nonculprit lesion ought to be treated in the same hospitalization or should you really be using a pressure wire,” he said. “For me as an interventionalist, this is really important because when you finish up doing an intervention on a patient it might be the middle of the night and the patient may be more or less stable, but you’ve already exposed them to the risk of a procedure, should you then move on and do another aspect of the procedure to interrogate with a pressure wire a remaining narrowing? I think that’s very important; that’ll help me make decisions on a day-to-day basis.”
Dr. Drachman also cited RADIANCE-HTN TRIO because it employs an endovascular technique to control blood pressure in patients with hypertension, specifically those resistant to multiple drugs.
During the press conference, Dr. Morris, a preventive cardiologist, put her money on the ADAPTABLE study of aspirin dosing, reiterating that the unique trial design could inform future research, and on Sunday’s 8:45 a.m. late-breaking post hoc analysis from the STRENGTH trial that looks to pick up where the controversy over omega-3 fatty acid preparations left off at last year’s American Heart Association meeting.
A lack of benefit on CV event rates reported with Epanova, a high-dose combination of eicosapentaenoic acid (EPA) and docosahexaenoic acid, led to a contentious debate over how to reconcile STRENGTH with the findings from REDUCE-IT, which showed a 25% relative risk reduction in major CV events with the EPA product icosapent ethyl (Vascepa).
STRENGTH investigator Steven Nissen, MD, Cleveland Clinic, and REDUCE-IT investigator and session panelist Deepak Bhatt, MD, Brigham and Women’s Hospital, Boston, will share the virtual stage at ACC 2021, but Dr. Morris said the “good news” is both researchers know one another very well and “will really be focusing on no political issues, just the omega-3 fatty levels in the bloodstream and what does that mean in either trial.
“This is not designed to be a debate, point counterpoint,” she added.
For that, as all cardiologists and journalists know, there will be the wild and woolly #CardioTwitter sphere.
A version of this article first appeared on Medscape.com.
FDA blazes path for ‘real-world’ evidence as proof of efficacy
In 2016, results from the LEADER trial of liraglutide in patients with type 2 diabetes helped jump-start awareness of the potential role of this new class of drugs, the glucagonlike peptide–1 receptor agonists, for reducing cardiovascular events. The randomized, placebo-controlled trial enrolled more than 9000 patients at more than 400 sites in over 30 countries, and took nearly 6 years from the start of patient enrollment to publication of the landmark results.
In December 2020, an independent team of researchers published results from a study with a design identical to LEADER, but used data that came not from a massive, global, years-long trial but from already-existing numbers culled from three large U.S. insurance claim databases. The result of this emulation using real-world data was virtually identical to what the actual trial showed, replicating both the direction and statistical significance of the original finding of the randomized, controlled trial (RCT).
What if research proved that this sort of RCT emulation could reliably be done on a regular basis? What might it mean for regulatory decisions on drugs and devices that historically have been based entirely on efficacy evidence from RCTs?
Making the most of a sea of observational data
Medicine in the United States has become increasingly awash in a sea of observational data collected from sources that include electronic health records, insurance claims, and increasingly, personal-health monitoring devices.
The Food and Drug Administration is now in the process of trying to figure out how it can legitimately harness this tsunami of real-world data to make efficacy decisions, essentially creating a new category of evidence to complement traditional data from randomized trials. It’s an opportunity that agency staff and their outside advisors have been keen to seize, especially given the soaring cost of prospective, randomized trials.
Recognition of this untapped resource in part led to a key initiative, among many others, included in the 21st Century Cures Act, passed in December 2016. Among the Act’s mandates was that, by the end of 2021, the FDA would issue guidance on when drug sponsors could use real-world evidence (RWE) to either help support a new indication for an already approved drug or help satisfy postapproval study requirements.
The initiative recognizes that this approach is not appropriate for initial drug approvals, which remain exclusively reliant on evidence from RCTs. Instead, it seems best suited to support expanding indications for already approved drugs.
Although FDA staff have made progress in identifying the challenges and broadening their understanding of how to best handle real-world data that come from observing patients in routine practice, agency leaders stress that this complex issue will likely not be fully resolved by their guidance to be published later this year. The FDA released a draft of the guidance in May 2019.
Can RWE be ‘credible and reliable?’
“Whether observational, nonrandomized data can become credible enough to use is what we’re talking about. These are possibilities that need to be explained and better understood,” said Robert Temple, MD, deputy director for clinical science of the FDA Center for Drug Evaluation and Research.
“Since the 1970s, the FDA has recognized historical controls as legitimate, so it’s possible [for RWE] to be credible. The big test is when is it credible and reliable enough [to assess efficacy]?” wondered Dr. Temple during a 2-day workshop on the topic held mid-February and organized by Duke University’s Margolis Center for Health Policy.
“We’re approaching an inflection point regarding how observational studies are generated and used, but our evidentiary standards will not lower, and it will be a case-by-case decision” by the agency as they review future RWE submissions, said John Concato, MD, the FDA’s associate director for real-world evidence, during the workshop.
“We are working toward guidance development, but also looking down the road to what we need to do to enable this,” said Dr. Concato. “It’s a complicated issue. If it was easy, it would have already been fixed.” He added that the agency will likely release a “portfolio” of guidance for submitting real-world data and RWE. Real-world data are raw information that, when analyzed, become RWE.
In short, the FDA seems headed toward guidance that won’t spell out a pathway that guarantees success using RWE but will at least open the door to consideration of this unprecedented application.
Not like flipping a switch
The guidance will not activate acceptance of RWE all at once. “It’s not like a light switch,” cautioned Adam Kroetsch, MPP, research director for biomedical innovation and regulatory policy at Duke-Margolis in Washington, D.C. “It’s an evolutionary process,” and the upcoming guidance will provide “just a little more clarity” on what sorts of best practices using RWE the FDA will find persuasive. “It’s hard for the FDA to clearly say what it’s looking for until they see some good examples,” Dr. Kroetsch said in an interview.
What will change is that drug sponsors can submit using RWE, and the FDA “will have a more open-minded view,” predicted Sebastian Schneeweiss, MD, ScD, a workshop participant and chief of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston. “For the first time, a law required [the FDA] to take a serious look” at observational data for efficacy assessment.
“The FDA has had a bias against using RWE for evidence of efficacy but has long used it to understand drug safety. Now the FDA is trying to wrap its arms around how to best use RWE” for efficacy decisions, said Joseph S. Ross, MD, another workshop participant and professor of medicine and public health at Yale University, New Haven, Conn.
The agency’s cautious approach is reassuring, Dr. Ross noted in an interview. “There was worry that the 21st Century Cures Act would open the door to allowing real-world data to be used in ways that weren’t very reliable. Very quickly, the FDA started trying to figure out the best ways to use these data in reasonable ways.”
Duplicating RCTs with RWE
To help better understand the potential use of RWE, the FDA sponsored several demonstration projects. Researchers presented results from three of these projects during the workshop in February. All three examined whether RWE, plugged into the design of an actual RCT, can produce roughly similar results when similar patients are used.
A generally consistent finding from the three demonstration projects was that “when the data are fit for purpose” the emulated or duplicated analyses with RWE “can come to similar conclusions” as the actual RCTs, said Dr. Schneeweiss, who leads one of the demonstration projects, RCT DUPLICATE.
At the workshop he reported results from RWE duplications of 20 different RCTs using insurance claims data from U.S. patients. The findings came from 10 duplications already reported in Circulation in December 2020 (including a duplication of the LEADER trial), and an additional 10 as yet unpublished RCT duplications. In the next few months, the researchers intend to assess a final group of 10 more RCT duplications.
Workshop participants also presented results from two other FDA demonstration projects: the OPERAND program run by the Multi-Regional Clinical Trials Center of Brigham and Women’s Hospital and Harvard; and the CERSI program based at Yale and the Mayo Clinic in Rochester, Minn. Both are smaller in scale than RCT DUPLICATE, incorporate lab data in addition to claims data, and in some cases test how well RWE can emulate RCTs that are not yet completed.
Collectively, results from these demonstration projects suggest that RWE can successfully emulate the results of an RCT, said Dr. Ross, a coinvestigator on the CERSI study. But the CERSI findings also highlighted how an RCT can fall short of clinical relevance.
“One of our most important findings was that RCTs don’t always represent real-world practice,” he said. His group attempted to replicate the 5,000-patient GRADE trial of four different drug options added to metformin in patients with type 2 diabetes. One of the four options included insulin glargine (Lantus), and the attempt to emulate the study with RWE hit the bump that no relevant real-world patients in their US claims database actually received the formulation.
That means the GRADE trial “is almost meaningless. It doesn’t reflect real-world practice,” Dr. Ross noted.
Results from the three demonstration projects “highlight the gaps we still have,” summed up Dr. Kroetsch. “They show where we need better data” from observational sources that function as well as data from RCTs.
Still, the demonstration project results are “an important step forward in establishing the validity of real-world evidence,” commented David Kerr, MBChB, an endocrinologist and director of research and innovation at the Sansum Diabetes Research Institute in Santa Barbara, Calif.
‘Target trials’ tether RWE
The target trial approach to designing an observational study is a key tool for boosting reliability and applicability of the results. The idea is to create a well-designed trial that could be the basis for a conventional RCT, and then use observational data to flesh out the target trial instead of collecting data from prospectively enrolled patients.
Designing observational studies that emulate target trials allows causal inferences, said Miguel A. Hernán, MD, DrPH, a professor of biostatistics and epidemiology at the Harvard School of Public Health, Boston. Plugging real-world data into the framework of an appropriately designed target trial substantially cuts the risk of a biased analysis, he explained during the workshop.
However, the approach has limitations. The target trial must be a pragmatic trial, and the approach does not work for placebo-controlled trials, although it can accommodate a usual-care control arm. It also usually precludes patient blinding, testing treatments not used in routine practice, and close monitoring of patients in ways that are uncommon in usual care.
The target trial approach received broad endorsement during the workshop as the future for observational studies destined for efficacy consideration by the FDA.
“The idea of prespecifying a target trial is a really fantastic place to start,” commented Robert Ball, MD, deputy director of the FDA Office of Surveillance and Epidemiology. “There is still a whole set of questions once the trial is prespecified, but prespecification would be a fantastic step forward,” he said during the workshop.
Participants also endorsed other important steps to boost the value of observational studies for regulatory reviews, including preregistering the study on a site such as clinicaltrials.gov; being fully transparent about the origins of observational data; using data that match the needs of the target trial; not reviewing the data in advance to avoid cherry picking and gaming the analysis; and reporting neutral or negative results when they occur, something often not currently done for observational analyses.
But although there was clear progress and much agreement among thought leaders at the workshop, FDA representatives stressed caution in moving forward.
“No easy answer”
“With more experience, we can learn what works and what doesn’t work in generating valid results from observational studies,” said Dr. Concato. “Although the observational results have upside potential, we need to learn more. There is no easy answer, no checklist for fit-for-use data, no off-the-shelf study design, and no ideal analytic method.”
Dr. Concato acknowledged that the FDA’s goal is clear given the 2016 legislation. “The FDA is embracing our obligations under the 21st Century Cures Act to evaluate use of real-world data and real-world evidence.”
He also suggested that researchers “shy away from a false dichotomy of RCTs or observational studies and instead think about how and when RCTs and observational studies can be designed and conducted to yield trustworthy results.” Dr. Concato’s solution: “a taxonomy of interventional or noninterventional studies.”
“The FDA is under enormous pressure to embrace real-world evidence, both because of the economics of running RCTs and because of the availability of new observational data from electronic health records, wearable devices, claims, etc.,” said Dr. Kerr, who did not participate in the workshop but coauthored an editorial that calls for using real-world data in regulatory decisions for drugs and devices for diabetes. These factors create an “irresistible force” spurring the FDA to consider observational, noninterventional data.
“I think the FDA really wants this to go forward,” Dr. Kerr added in an interview. “The FDA keeps telling us that clinical trials do not have enough women or patients from minority groups. Real-world data is a way to address that. This will not be the death of RCTs, but this work shines a light on the deficiencies of RCTs and how the deficiencies can be dealt with.”
Dr. Kroetsch has reported no relevant financial relationships. Dr. Schneeweiss has reported being a consultant to and holding equity in Aetion and receiving research funding from the FDA. Dr. Ross has reported receiving research funding from the FDA, Johnson & Johnson, and Medtronic. Dr. Hernán has reported being a consultant for Cytel. Dr. Kerr has reported being a consultant for Ascensia, EOFlow, Lifecare, Merck, Novo Nordisk, Roche Diagnostics, and Voluntis. Dr. Temple, Dr. Concato, and Dr. Ball are FDA employees.
A version of this article first appeared on Medscape.com.
In 2016, results from the LEADER trial of liraglutide in patients with type 2 diabetes helped jump-start awareness of the potential role of this new class of drugs, the glucagonlike peptide–1 receptor agonists, for reducing cardiovascular events. The randomized, placebo-controlled trial enrolled more than 9000 patients at more than 400 sites in over 30 countries, and took nearly 6 years from the start of patient enrollment to publication of the landmark results.
In December 2020, an independent team of researchers published results from a study with a design identical to LEADER, but used data that came not from a massive, global, years-long trial but from already-existing numbers culled from three large U.S. insurance claim databases. The result of this emulation using real-world data was virtually identical to what the actual trial showed, replicating both the direction and statistical significance of the original finding of the randomized, controlled trial (RCT).
What if research proved that this sort of RCT emulation could reliably be done on a regular basis? What might it mean for regulatory decisions on drugs and devices that historically have been based entirely on efficacy evidence from RCTs?
Making the most of a sea of observational data
Medicine in the United States has become increasingly awash in a sea of observational data collected from sources that include electronic health records, insurance claims, and increasingly, personal-health monitoring devices.
The Food and Drug Administration is now in the process of trying to figure out how it can legitimately harness this tsunami of real-world data to make efficacy decisions, essentially creating a new category of evidence to complement traditional data from randomized trials. It’s an opportunity that agency staff and their outside advisors have been keen to seize, especially given the soaring cost of prospective, randomized trials.
Recognition of this untapped resource in part led to a key initiative, among many others, included in the 21st Century Cures Act, passed in December 2016. Among the Act’s mandates was that, by the end of 2021, the FDA would issue guidance on when drug sponsors could use real-world evidence (RWE) to either help support a new indication for an already approved drug or help satisfy postapproval study requirements.
The initiative recognizes that this approach is not appropriate for initial drug approvals, which remain exclusively reliant on evidence from RCTs. Instead, it seems best suited to support expanding indications for already approved drugs.
Although FDA staff have made progress in identifying the challenges and broadening their understanding of how to best handle real-world data that come from observing patients in routine practice, agency leaders stress that this complex issue will likely not be fully resolved by their guidance to be published later this year. The FDA released a draft of the guidance in May 2019.
Can RWE be ‘credible and reliable?’
“Whether observational, nonrandomized data can become credible enough to use is what we’re talking about. These are possibilities that need to be explained and better understood,” said Robert Temple, MD, deputy director for clinical science of the FDA Center for Drug Evaluation and Research.
“Since the 1970s, the FDA has recognized historical controls as legitimate, so it’s possible [for RWE] to be credible. The big test is when is it credible and reliable enough [to assess efficacy]?” wondered Dr. Temple during a 2-day workshop on the topic held mid-February and organized by Duke University’s Margolis Center for Health Policy.
“We’re approaching an inflection point regarding how observational studies are generated and used, but our evidentiary standards will not lower, and it will be a case-by-case decision” by the agency as they review future RWE submissions, said John Concato, MD, the FDA’s associate director for real-world evidence, during the workshop.
“We are working toward guidance development, but also looking down the road to what we need to do to enable this,” said Dr. Concato. “It’s a complicated issue. If it was easy, it would have already been fixed.” He added that the agency will likely release a “portfolio” of guidance for submitting real-world data and RWE. Real-world data are raw information that, when analyzed, become RWE.
In short, the FDA seems headed toward guidance that won’t spell out a pathway that guarantees success using RWE but will at least open the door to consideration of this unprecedented application.
Not like flipping a switch
The guidance will not activate acceptance of RWE all at once. “It’s not like a light switch,” cautioned Adam Kroetsch, MPP, research director for biomedical innovation and regulatory policy at Duke-Margolis in Washington, D.C. “It’s an evolutionary process,” and the upcoming guidance will provide “just a little more clarity” on what sorts of best practices using RWE the FDA will find persuasive. “It’s hard for the FDA to clearly say what it’s looking for until they see some good examples,” Dr. Kroetsch said in an interview.
What will change is that drug sponsors can submit using RWE, and the FDA “will have a more open-minded view,” predicted Sebastian Schneeweiss, MD, ScD, a workshop participant and chief of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston. “For the first time, a law required [the FDA] to take a serious look” at observational data for efficacy assessment.
“The FDA has had a bias against using RWE for evidence of efficacy but has long used it to understand drug safety. Now the FDA is trying to wrap its arms around how to best use RWE” for efficacy decisions, said Joseph S. Ross, MD, another workshop participant and professor of medicine and public health at Yale University, New Haven, Conn.
The agency’s cautious approach is reassuring, Dr. Ross noted in an interview. “There was worry that the 21st Century Cures Act would open the door to allowing real-world data to be used in ways that weren’t very reliable. Very quickly, the FDA started trying to figure out the best ways to use these data in reasonable ways.”
Duplicating RCTs with RWE
To help better understand the potential use of RWE, the FDA sponsored several demonstration projects. Researchers presented results from three of these projects during the workshop in February. All three examined whether RWE, plugged into the design of an actual RCT, can produce roughly similar results when similar patients are used.
A generally consistent finding from the three demonstration projects was that “when the data are fit for purpose” the emulated or duplicated analyses with RWE “can come to similar conclusions” as the actual RCTs, said Dr. Schneeweiss, who leads one of the demonstration projects, RCT DUPLICATE.
At the workshop he reported results from RWE duplications of 20 different RCTs using insurance claims data from U.S. patients. The findings came from 10 duplications already reported in Circulation in December 2020 (including a duplication of the LEADER trial), and an additional 10 as yet unpublished RCT duplications. In the next few months, the researchers intend to assess a final group of 10 more RCT duplications.
Workshop participants also presented results from two other FDA demonstration projects: the OPERAND program run by the Multi-Regional Clinical Trials Center of Brigham and Women’s Hospital and Harvard; and the CERSI program based at Yale and the Mayo Clinic in Rochester, Minn. Both are smaller in scale than RCT DUPLICATE, incorporate lab data in addition to claims data, and in some cases test how well RWE can emulate RCTs that are not yet completed.
Collectively, results from these demonstration projects suggest that RWE can successfully emulate the results of an RCT, said Dr. Ross, a coinvestigator on the CERSI study. But the CERSI findings also highlighted how an RCT can fall short of clinical relevance.
“One of our most important findings was that RCTs don’t always represent real-world practice,” he said. His group attempted to replicate the 5,000-patient GRADE trial of four different drug options added to metformin in patients with type 2 diabetes. One of the four options included insulin glargine (Lantus), and the attempt to emulate the study with RWE hit the bump that no relevant real-world patients in their US claims database actually received the formulation.
That means the GRADE trial “is almost meaningless. It doesn’t reflect real-world practice,” Dr. Ross noted.
Results from the three demonstration projects “highlight the gaps we still have,” summed up Dr. Kroetsch. “They show where we need better data” from observational sources that function as well as data from RCTs.
Still, the demonstration project results are “an important step forward in establishing the validity of real-world evidence,” commented David Kerr, MBChB, an endocrinologist and director of research and innovation at the Sansum Diabetes Research Institute in Santa Barbara, Calif.
‘Target trials’ tether RWE
The target trial approach to designing an observational study is a key tool for boosting reliability and applicability of the results. The idea is to create a well-designed trial that could be the basis for a conventional RCT, and then use observational data to flesh out the target trial instead of collecting data from prospectively enrolled patients.
Designing observational studies that emulate target trials allows causal inferences, said Miguel A. Hernán, MD, DrPH, a professor of biostatistics and epidemiology at the Harvard School of Public Health, Boston. Plugging real-world data into the framework of an appropriately designed target trial substantially cuts the risk of a biased analysis, he explained during the workshop.
However, the approach has limitations. The target trial must be a pragmatic trial, and the approach does not work for placebo-controlled trials, although it can accommodate a usual-care control arm. It also usually precludes patient blinding, testing treatments not used in routine practice, and close monitoring of patients in ways that are uncommon in usual care.
The target trial approach received broad endorsement during the workshop as the future for observational studies destined for efficacy consideration by the FDA.
“The idea of prespecifying a target trial is a really fantastic place to start,” commented Robert Ball, MD, deputy director of the FDA Office of Surveillance and Epidemiology. “There is still a whole set of questions once the trial is prespecified, but prespecification would be a fantastic step forward,” he said during the workshop.
Participants also endorsed other important steps to boost the value of observational studies for regulatory reviews, including preregistering the study on a site such as clinicaltrials.gov; being fully transparent about the origins of observational data; using data that match the needs of the target trial; not reviewing the data in advance to avoid cherry picking and gaming the analysis; and reporting neutral or negative results when they occur, something often not currently done for observational analyses.
But although there was clear progress and much agreement among thought leaders at the workshop, FDA representatives stressed caution in moving forward.
“No easy answer”
“With more experience, we can learn what works and what doesn’t work in generating valid results from observational studies,” said Dr. Concato. “Although the observational results have upside potential, we need to learn more. There is no easy answer, no checklist for fit-for-use data, no off-the-shelf study design, and no ideal analytic method.”
Dr. Concato acknowledged that the FDA’s goal is clear given the 2016 legislation. “The FDA is embracing our obligations under the 21st Century Cures Act to evaluate use of real-world data and real-world evidence.”
He also suggested that researchers “shy away from a false dichotomy of RCTs or observational studies and instead think about how and when RCTs and observational studies can be designed and conducted to yield trustworthy results.” Dr. Concato’s solution: “a taxonomy of interventional or noninterventional studies.”
“The FDA is under enormous pressure to embrace real-world evidence, both because of the economics of running RCTs and because of the availability of new observational data from electronic health records, wearable devices, claims, etc.,” said Dr. Kerr, who did not participate in the workshop but coauthored an editorial that calls for using real-world data in regulatory decisions for drugs and devices for diabetes. These factors create an “irresistible force” spurring the FDA to consider observational, noninterventional data.
“I think the FDA really wants this to go forward,” Dr. Kerr added in an interview. “The FDA keeps telling us that clinical trials do not have enough women or patients from minority groups. Real-world data is a way to address that. This will not be the death of RCTs, but this work shines a light on the deficiencies of RCTs and how the deficiencies can be dealt with.”
Dr. Kroetsch has reported no relevant financial relationships. Dr. Schneeweiss has reported being a consultant to and holding equity in Aetion and receiving research funding from the FDA. Dr. Ross has reported receiving research funding from the FDA, Johnson & Johnson, and Medtronic. Dr. Hernán has reported being a consultant for Cytel. Dr. Kerr has reported being a consultant for Ascensia, EOFlow, Lifecare, Merck, Novo Nordisk, Roche Diagnostics, and Voluntis. Dr. Temple, Dr. Concato, and Dr. Ball are FDA employees.
A version of this article first appeared on Medscape.com.
In 2016, results from the LEADER trial of liraglutide in patients with type 2 diabetes helped jump-start awareness of the potential role of this new class of drugs, the glucagonlike peptide–1 receptor agonists, for reducing cardiovascular events. The randomized, placebo-controlled trial enrolled more than 9000 patients at more than 400 sites in over 30 countries, and took nearly 6 years from the start of patient enrollment to publication of the landmark results.
In December 2020, an independent team of researchers published results from a study with a design identical to LEADER, but used data that came not from a massive, global, years-long trial but from already-existing numbers culled from three large U.S. insurance claim databases. The result of this emulation using real-world data was virtually identical to what the actual trial showed, replicating both the direction and statistical significance of the original finding of the randomized, controlled trial (RCT).
What if research proved that this sort of RCT emulation could reliably be done on a regular basis? What might it mean for regulatory decisions on drugs and devices that historically have been based entirely on efficacy evidence from RCTs?
Making the most of a sea of observational data
Medicine in the United States has become increasingly awash in a sea of observational data collected from sources that include electronic health records, insurance claims, and increasingly, personal-health monitoring devices.
The Food and Drug Administration is now in the process of trying to figure out how it can legitimately harness this tsunami of real-world data to make efficacy decisions, essentially creating a new category of evidence to complement traditional data from randomized trials. It’s an opportunity that agency staff and their outside advisors have been keen to seize, especially given the soaring cost of prospective, randomized trials.
Recognition of this untapped resource in part led to a key initiative, among many others, included in the 21st Century Cures Act, passed in December 2016. Among the Act’s mandates was that, by the end of 2021, the FDA would issue guidance on when drug sponsors could use real-world evidence (RWE) to either help support a new indication for an already approved drug or help satisfy postapproval study requirements.
The initiative recognizes that this approach is not appropriate for initial drug approvals, which remain exclusively reliant on evidence from RCTs. Instead, it seems best suited to support expanding indications for already approved drugs.
Although FDA staff have made progress in identifying the challenges and broadening their understanding of how to best handle real-world data that come from observing patients in routine practice, agency leaders stress that this complex issue will likely not be fully resolved by their guidance to be published later this year. The FDA released a draft of the guidance in May 2019.
Can RWE be ‘credible and reliable?’
“Whether observational, nonrandomized data can become credible enough to use is what we’re talking about. These are possibilities that need to be explained and better understood,” said Robert Temple, MD, deputy director for clinical science of the FDA Center for Drug Evaluation and Research.
“Since the 1970s, the FDA has recognized historical controls as legitimate, so it’s possible [for RWE] to be credible. The big test is when is it credible and reliable enough [to assess efficacy]?” wondered Dr. Temple during a 2-day workshop on the topic held mid-February and organized by Duke University’s Margolis Center for Health Policy.
“We’re approaching an inflection point regarding how observational studies are generated and used, but our evidentiary standards will not lower, and it will be a case-by-case decision” by the agency as they review future RWE submissions, said John Concato, MD, the FDA’s associate director for real-world evidence, during the workshop.
“We are working toward guidance development, but also looking down the road to what we need to do to enable this,” said Dr. Concato. “It’s a complicated issue. If it was easy, it would have already been fixed.” He added that the agency will likely release a “portfolio” of guidance for submitting real-world data and RWE. Real-world data are raw information that, when analyzed, become RWE.
In short, the FDA seems headed toward guidance that won’t spell out a pathway that guarantees success using RWE but will at least open the door to consideration of this unprecedented application.
Not like flipping a switch
The guidance will not activate acceptance of RWE all at once. “It’s not like a light switch,” cautioned Adam Kroetsch, MPP, research director for biomedical innovation and regulatory policy at Duke-Margolis in Washington, D.C. “It’s an evolutionary process,” and the upcoming guidance will provide “just a little more clarity” on what sorts of best practices using RWE the FDA will find persuasive. “It’s hard for the FDA to clearly say what it’s looking for until they see some good examples,” Dr. Kroetsch said in an interview.
What will change is that drug sponsors can submit using RWE, and the FDA “will have a more open-minded view,” predicted Sebastian Schneeweiss, MD, ScD, a workshop participant and chief of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston. “For the first time, a law required [the FDA] to take a serious look” at observational data for efficacy assessment.
“The FDA has had a bias against using RWE for evidence of efficacy but has long used it to understand drug safety. Now the FDA is trying to wrap its arms around how to best use RWE” for efficacy decisions, said Joseph S. Ross, MD, another workshop participant and professor of medicine and public health at Yale University, New Haven, Conn.
The agency’s cautious approach is reassuring, Dr. Ross noted in an interview. “There was worry that the 21st Century Cures Act would open the door to allowing real-world data to be used in ways that weren’t very reliable. Very quickly, the FDA started trying to figure out the best ways to use these data in reasonable ways.”
Duplicating RCTs with RWE
To help better understand the potential use of RWE, the FDA sponsored several demonstration projects. Researchers presented results from three of these projects during the workshop in February. All three examined whether RWE, plugged into the design of an actual RCT, can produce roughly similar results when similar patients are used.
A generally consistent finding from the three demonstration projects was that “when the data are fit for purpose” the emulated or duplicated analyses with RWE “can come to similar conclusions” as the actual RCTs, said Dr. Schneeweiss, who leads one of the demonstration projects, RCT DUPLICATE.
At the workshop he reported results from RWE duplications of 20 different RCTs using insurance claims data from U.S. patients. The findings came from 10 duplications already reported in Circulation in December 2020 (including a duplication of the LEADER trial), and an additional 10 as yet unpublished RCT duplications. In the next few months, the researchers intend to assess a final group of 10 more RCT duplications.
Workshop participants also presented results from two other FDA demonstration projects: the OPERAND program run by the Multi-Regional Clinical Trials Center of Brigham and Women’s Hospital and Harvard; and the CERSI program based at Yale and the Mayo Clinic in Rochester, Minn. Both are smaller in scale than RCT DUPLICATE, incorporate lab data in addition to claims data, and in some cases test how well RWE can emulate RCTs that are not yet completed.
Collectively, results from these demonstration projects suggest that RWE can successfully emulate the results of an RCT, said Dr. Ross, a coinvestigator on the CERSI study. But the CERSI findings also highlighted how an RCT can fall short of clinical relevance.
“One of our most important findings was that RCTs don’t always represent real-world practice,” he said. His group attempted to replicate the 5,000-patient GRADE trial of four different drug options added to metformin in patients with type 2 diabetes. One of the four options included insulin glargine (Lantus), and the attempt to emulate the study with RWE hit the bump that no relevant real-world patients in their US claims database actually received the formulation.
That means the GRADE trial “is almost meaningless. It doesn’t reflect real-world practice,” Dr. Ross noted.
Results from the three demonstration projects “highlight the gaps we still have,” summed up Dr. Kroetsch. “They show where we need better data” from observational sources that function as well as data from RCTs.
Still, the demonstration project results are “an important step forward in establishing the validity of real-world evidence,” commented David Kerr, MBChB, an endocrinologist and director of research and innovation at the Sansum Diabetes Research Institute in Santa Barbara, Calif.
‘Target trials’ tether RWE
The target trial approach to designing an observational study is a key tool for boosting reliability and applicability of the results. The idea is to create a well-designed trial that could be the basis for a conventional RCT, and then use observational data to flesh out the target trial instead of collecting data from prospectively enrolled patients.
Designing observational studies that emulate target trials allows causal inferences, said Miguel A. Hernán, MD, DrPH, a professor of biostatistics and epidemiology at the Harvard School of Public Health, Boston. Plugging real-world data into the framework of an appropriately designed target trial substantially cuts the risk of a biased analysis, he explained during the workshop.
However, the approach has limitations. The target trial must be a pragmatic trial, and the approach does not work for placebo-controlled trials, although it can accommodate a usual-care control arm. It also usually precludes patient blinding, testing treatments not used in routine practice, and close monitoring of patients in ways that are uncommon in usual care.
The target trial approach received broad endorsement during the workshop as the future for observational studies destined for efficacy consideration by the FDA.
“The idea of prespecifying a target trial is a really fantastic place to start,” commented Robert Ball, MD, deputy director of the FDA Office of Surveillance and Epidemiology. “There is still a whole set of questions once the trial is prespecified, but prespecification would be a fantastic step forward,” he said during the workshop.
Participants also endorsed other important steps to boost the value of observational studies for regulatory reviews, including preregistering the study on a site such as clinicaltrials.gov; being fully transparent about the origins of observational data; using data that match the needs of the target trial; not reviewing the data in advance to avoid cherry picking and gaming the analysis; and reporting neutral or negative results when they occur, something often not currently done for observational analyses.
But although there was clear progress and much agreement among thought leaders at the workshop, FDA representatives stressed caution in moving forward.
“No easy answer”
“With more experience, we can learn what works and what doesn’t work in generating valid results from observational studies,” said Dr. Concato. “Although the observational results have upside potential, we need to learn more. There is no easy answer, no checklist for fit-for-use data, no off-the-shelf study design, and no ideal analytic method.”
Dr. Concato acknowledged that the FDA’s goal is clear given the 2016 legislation. “The FDA is embracing our obligations under the 21st Century Cures Act to evaluate use of real-world data and real-world evidence.”
He also suggested that researchers “shy away from a false dichotomy of RCTs or observational studies and instead think about how and when RCTs and observational studies can be designed and conducted to yield trustworthy results.” Dr. Concato’s solution: “a taxonomy of interventional or noninterventional studies.”
“The FDA is under enormous pressure to embrace real-world evidence, both because of the economics of running RCTs and because of the availability of new observational data from electronic health records, wearable devices, claims, etc.,” said Dr. Kerr, who did not participate in the workshop but coauthored an editorial that calls for using real-world data in regulatory decisions for drugs and devices for diabetes. These factors create an “irresistible force” spurring the FDA to consider observational, noninterventional data.
“I think the FDA really wants this to go forward,” Dr. Kerr added in an interview. “The FDA keeps telling us that clinical trials do not have enough women or patients from minority groups. Real-world data is a way to address that. This will not be the death of RCTs, but this work shines a light on the deficiencies of RCTs and how the deficiencies can be dealt with.”
Dr. Kroetsch has reported no relevant financial relationships. Dr. Schneeweiss has reported being a consultant to and holding equity in Aetion and receiving research funding from the FDA. Dr. Ross has reported receiving research funding from the FDA, Johnson & Johnson, and Medtronic. Dr. Hernán has reported being a consultant for Cytel. Dr. Kerr has reported being a consultant for Ascensia, EOFlow, Lifecare, Merck, Novo Nordisk, Roche Diagnostics, and Voluntis. Dr. Temple, Dr. Concato, and Dr. Ball are FDA employees.
A version of this article first appeared on Medscape.com.
Fresh look at ISCHEMIA bolsters conservative message in stable CAD
The more complicated a primary endpoint, the greater a puzzle it can be for clinicians to interpret the results. It’s likely even tougher for patients, who don’t help choose the events studied in clinical trials yet are increasingly sharing in the management decisions they influence.
That creates an opening for a more patient-centered take on one of cardiology’s most influential recent studies, ISCHEMIA, which bolsters the case for conservative, med-oriented management over a more invasive initial strategy for patients with stable coronary artery disease (CAD) and positive stress tests, researchers said.
The new, prespecified analysis replaced the trial’s conventional primary endpoint of major adverse cardiac events (MACE) with one based on “days alive out of hospital” (DAOH) and found an early advantage for the conservative approach, with caveats.
Those assigned to the conservative arm benefited with more out-of-hospital days throughout the next 2 years than those in the invasive-management group, owing to the latter’s protocol-mandated early cath-lab work-up with possible revascularization. The difference averaged more than 6 days for much of that time.
But DAOH evened out for the two groups by the fourth year in the analysis of more than 5,000 patients.
Protocol-determined cath procedures accounted for 61% of hospitalizations in the invasively managed group. A secondary DAOH analysis that excluded such required hospital days, also prespecified, showed no meaningful difference between the two strategies over the 4 years, noted the report published online May 3 in JAMA Cardiology.
DOAH is ‘very, very important’
The DAOH metric has been a far less common consideration in clinical trials, compared with clinical events, yet in some ways it is as “hard” a metric as mortality, encompasses a broader range of outcomes, and may matter more to patients, it’s been argued.
“The thing patients most value is time at home. So they don’t want to be in the hospital, they don’t want to be away from friends, they want to do recreation, or they may want to work,” lead author Harvey D. White, DSc, Green Lane Cardiovascular Services, Auckland (New Zealand) City Hospital, University of Auckland, told this news organization.
“When we need to talk to patients – and we do need to talk to patients – to have a days-out-of-hospital metric is very, very important,” he said. It is not only patient focused, it’s “meaningful in terms of the seriousness of events,” in that length of hospitalization tracks with clinical severity, observed Dr. White, who is slated to present the analysis May 17 during the virtual American College of Cardiology 2021 scientific sessions.
As previously reported, ISCHEMIA showed no significant effect on the primary endpoint of cardiovascular mortality, MI, or hospitalization for unstable angina, heart failure, or resuscitated cardiac arrest by assignment group over a median 3.2 years. Angina and quality of life measures were improved for patients in the invasive arm.
With an invasive initial strategy, “What we know now is that you get nothing of an advantage in terms of the composite endpoint, and you’re going to spend 6 days more in the hospital in the first 2 years, for largely no benefit,” Dr. White said.
That outlook may apply out to 4 years, the analysis suggests, but could conceivably change if DAOH is reassessed later as the ISCHEMIA follow-up continues for what is now a planned total of 10 years.
Meanwhile, the current findings could enhance doctor-patient discussions about the trade-offs between the two strategies for individuals whose considerations will vary.
“This is a very helpful measure to understand the burden of an approach to the patient,” observed E. Magnus Ohman, MD, an interventional cardiologist at Duke University, Durham, N.C., who was not involved in the trial.
With DAOH as an endpoint, “you as a clinician get another aspect of understanding of a treatment’s impact on a multitude of endpoints.” Days out of hospital, he noted, encompasses the effects of clinical events that often go into composite clinical endpoints – not death, but including nonfatal MI, stroke, need for revascularization, and cardiovascular hospitalization.
To patients with stable CAD who ask whether the invasive approach has merits in their case, the DAOH finding “helps you to say, well, at the end of the day, you will probably be spending an equal amount of time in the hospital. Your price up front is a little bit higher, but over time, the group who gets conservative treatment will catch up.”
The DAOH outcome also avoids the limitations of an endpoint based on time to first event, “not the least of which,” said Dr. White, is that it counts only the first of what might be multiple events of varying clinical impact. Misleadingly, “you can have an event that’s a small troponin rise, but that becomes more important in a person than dying the next day.”
The DAOH analysis was based on 5,179 patients from 37 countries who averaged 64 years of age and of whom 23% were women. The endpoint considered only overnight stays in hospitals, skilled nursing facilities, rehabilitation centers, and nursing homes.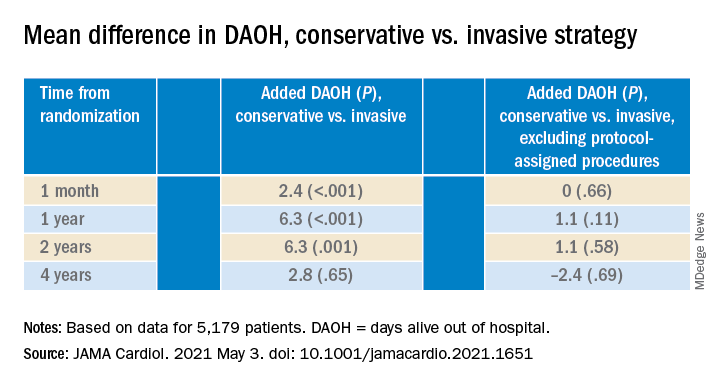
There were many more hospital or extended care facility stays overall in the invasive-management group, 4,002 versus 1,897 for those following the conservative strategy (P < .001), but the numbers flipped after excluding protocol-assigned procedures: 1,568 stays in the invasive group, compared with 1,897 (P = .001)
There were no associations between DAOH and Seattle Angina Questionnaire 7–Angina Frequency scores or DAOH interactions by age, sex, geographic region, or whether the patient had diabetes, prior MI, or heart failure, the report notes.
The primary ISCHEMIA analysis hinted at a possible long-term advantage for the invasive initial strategy in that event curves for the two arms crossed after 2-3 years, Dr. Ohman observed.
Based on that, for younger patients with stable CAD and ischemia at stress testing, “an investment of more hospital days early on might be worth it in the long run.” But ISCHEMIA, he said, “only suggests it, it doesn’t confirm it.”
The study was supported in part by grants from Arbor Pharmaceuticals and AstraZeneca. Devices or medications were provided by Abbott Vascular, Amgen, Arbor, AstraZeneca, Esperion, Medtronic, Merck Sharp & Dohme, Phillips, Omron Healthcare, and Sunovion. Dr. White disclosed receiving grants paid to his institution and fees for serving on a steering committee from Sanofi-Aventis, Regeneron, Eli Lilly, Omthera, American Regent, Eisai, DalCor, CSL Behring, Sanofi-Aventis Australia, and Esperion Therapeutics, and personal fees from Genentech and AstraZeneca. Dr. Ohman reported receiving grants from Abiomed and Cheisi USA, and consulting for Abiomed, Cara Therapeutics, Chiesi USA, Cytokinetics, Imbria Pharmaceuticals, Otsuka Pharmaceuticals, Milestone Pharmaceuticals, and XyloCor Therapeutics.
A version of this article first appeared on Medscape.com.
The more complicated a primary endpoint, the greater a puzzle it can be for clinicians to interpret the results. It’s likely even tougher for patients, who don’t help choose the events studied in clinical trials yet are increasingly sharing in the management decisions they influence.
That creates an opening for a more patient-centered take on one of cardiology’s most influential recent studies, ISCHEMIA, which bolsters the case for conservative, med-oriented management over a more invasive initial strategy for patients with stable coronary artery disease (CAD) and positive stress tests, researchers said.
The new, prespecified analysis replaced the trial’s conventional primary endpoint of major adverse cardiac events (MACE) with one based on “days alive out of hospital” (DAOH) and found an early advantage for the conservative approach, with caveats.
Those assigned to the conservative arm benefited with more out-of-hospital days throughout the next 2 years than those in the invasive-management group, owing to the latter’s protocol-mandated early cath-lab work-up with possible revascularization. The difference averaged more than 6 days for much of that time.
But DAOH evened out for the two groups by the fourth year in the analysis of more than 5,000 patients.
Protocol-determined cath procedures accounted for 61% of hospitalizations in the invasively managed group. A secondary DAOH analysis that excluded such required hospital days, also prespecified, showed no meaningful difference between the two strategies over the 4 years, noted the report published online May 3 in JAMA Cardiology.
DOAH is ‘very, very important’
The DAOH metric has been a far less common consideration in clinical trials, compared with clinical events, yet in some ways it is as “hard” a metric as mortality, encompasses a broader range of outcomes, and may matter more to patients, it’s been argued.
“The thing patients most value is time at home. So they don’t want to be in the hospital, they don’t want to be away from friends, they want to do recreation, or they may want to work,” lead author Harvey D. White, DSc, Green Lane Cardiovascular Services, Auckland (New Zealand) City Hospital, University of Auckland, told this news organization.
“When we need to talk to patients – and we do need to talk to patients – to have a days-out-of-hospital metric is very, very important,” he said. It is not only patient focused, it’s “meaningful in terms of the seriousness of events,” in that length of hospitalization tracks with clinical severity, observed Dr. White, who is slated to present the analysis May 17 during the virtual American College of Cardiology 2021 scientific sessions.
As previously reported, ISCHEMIA showed no significant effect on the primary endpoint of cardiovascular mortality, MI, or hospitalization for unstable angina, heart failure, or resuscitated cardiac arrest by assignment group over a median 3.2 years. Angina and quality of life measures were improved for patients in the invasive arm.
With an invasive initial strategy, “What we know now is that you get nothing of an advantage in terms of the composite endpoint, and you’re going to spend 6 days more in the hospital in the first 2 years, for largely no benefit,” Dr. White said.
That outlook may apply out to 4 years, the analysis suggests, but could conceivably change if DAOH is reassessed later as the ISCHEMIA follow-up continues for what is now a planned total of 10 years.
Meanwhile, the current findings could enhance doctor-patient discussions about the trade-offs between the two strategies for individuals whose considerations will vary.
“This is a very helpful measure to understand the burden of an approach to the patient,” observed E. Magnus Ohman, MD, an interventional cardiologist at Duke University, Durham, N.C., who was not involved in the trial.
With DAOH as an endpoint, “you as a clinician get another aspect of understanding of a treatment’s impact on a multitude of endpoints.” Days out of hospital, he noted, encompasses the effects of clinical events that often go into composite clinical endpoints – not death, but including nonfatal MI, stroke, need for revascularization, and cardiovascular hospitalization.
To patients with stable CAD who ask whether the invasive approach has merits in their case, the DAOH finding “helps you to say, well, at the end of the day, you will probably be spending an equal amount of time in the hospital. Your price up front is a little bit higher, but over time, the group who gets conservative treatment will catch up.”
The DAOH outcome also avoids the limitations of an endpoint based on time to first event, “not the least of which,” said Dr. White, is that it counts only the first of what might be multiple events of varying clinical impact. Misleadingly, “you can have an event that’s a small troponin rise, but that becomes more important in a person than dying the next day.”
The DAOH analysis was based on 5,179 patients from 37 countries who averaged 64 years of age and of whom 23% were women. The endpoint considered only overnight stays in hospitals, skilled nursing facilities, rehabilitation centers, and nursing homes.
There were many more hospital or extended care facility stays overall in the invasive-management group, 4,002 versus 1,897 for those following the conservative strategy (P < .001), but the numbers flipped after excluding protocol-assigned procedures: 1,568 stays in the invasive group, compared with 1,897 (P = .001)
There were no associations between DAOH and Seattle Angina Questionnaire 7–Angina Frequency scores or DAOH interactions by age, sex, geographic region, or whether the patient had diabetes, prior MI, or heart failure, the report notes.
The primary ISCHEMIA analysis hinted at a possible long-term advantage for the invasive initial strategy in that event curves for the two arms crossed after 2-3 years, Dr. Ohman observed.
Based on that, for younger patients with stable CAD and ischemia at stress testing, “an investment of more hospital days early on might be worth it in the long run.” But ISCHEMIA, he said, “only suggests it, it doesn’t confirm it.”
The study was supported in part by grants from Arbor Pharmaceuticals and AstraZeneca. Devices or medications were provided by Abbott Vascular, Amgen, Arbor, AstraZeneca, Esperion, Medtronic, Merck Sharp & Dohme, Phillips, Omron Healthcare, and Sunovion. Dr. White disclosed receiving grants paid to his institution and fees for serving on a steering committee from Sanofi-Aventis, Regeneron, Eli Lilly, Omthera, American Regent, Eisai, DalCor, CSL Behring, Sanofi-Aventis Australia, and Esperion Therapeutics, and personal fees from Genentech and AstraZeneca. Dr. Ohman reported receiving grants from Abiomed and Cheisi USA, and consulting for Abiomed, Cara Therapeutics, Chiesi USA, Cytokinetics, Imbria Pharmaceuticals, Otsuka Pharmaceuticals, Milestone Pharmaceuticals, and XyloCor Therapeutics.
A version of this article first appeared on Medscape.com.
The more complicated a primary endpoint, the greater a puzzle it can be for clinicians to interpret the results. It’s likely even tougher for patients, who don’t help choose the events studied in clinical trials yet are increasingly sharing in the management decisions they influence.
That creates an opening for a more patient-centered take on one of cardiology’s most influential recent studies, ISCHEMIA, which bolsters the case for conservative, med-oriented management over a more invasive initial strategy for patients with stable coronary artery disease (CAD) and positive stress tests, researchers said.
The new, prespecified analysis replaced the trial’s conventional primary endpoint of major adverse cardiac events (MACE) with one based on “days alive out of hospital” (DAOH) and found an early advantage for the conservative approach, with caveats.
Those assigned to the conservative arm benefited with more out-of-hospital days throughout the next 2 years than those in the invasive-management group, owing to the latter’s protocol-mandated early cath-lab work-up with possible revascularization. The difference averaged more than 6 days for much of that time.
But DAOH evened out for the two groups by the fourth year in the analysis of more than 5,000 patients.
Protocol-determined cath procedures accounted for 61% of hospitalizations in the invasively managed group. A secondary DAOH analysis that excluded such required hospital days, also prespecified, showed no meaningful difference between the two strategies over the 4 years, noted the report published online May 3 in JAMA Cardiology.
DOAH is ‘very, very important’
The DAOH metric has been a far less common consideration in clinical trials, compared with clinical events, yet in some ways it is as “hard” a metric as mortality, encompasses a broader range of outcomes, and may matter more to patients, it’s been argued.
“The thing patients most value is time at home. So they don’t want to be in the hospital, they don’t want to be away from friends, they want to do recreation, or they may want to work,” lead author Harvey D. White, DSc, Green Lane Cardiovascular Services, Auckland (New Zealand) City Hospital, University of Auckland, told this news organization.
“When we need to talk to patients – and we do need to talk to patients – to have a days-out-of-hospital metric is very, very important,” he said. It is not only patient focused, it’s “meaningful in terms of the seriousness of events,” in that length of hospitalization tracks with clinical severity, observed Dr. White, who is slated to present the analysis May 17 during the virtual American College of Cardiology 2021 scientific sessions.
As previously reported, ISCHEMIA showed no significant effect on the primary endpoint of cardiovascular mortality, MI, or hospitalization for unstable angina, heart failure, or resuscitated cardiac arrest by assignment group over a median 3.2 years. Angina and quality of life measures were improved for patients in the invasive arm.
With an invasive initial strategy, “What we know now is that you get nothing of an advantage in terms of the composite endpoint, and you’re going to spend 6 days more in the hospital in the first 2 years, for largely no benefit,” Dr. White said.
That outlook may apply out to 4 years, the analysis suggests, but could conceivably change if DAOH is reassessed later as the ISCHEMIA follow-up continues for what is now a planned total of 10 years.
Meanwhile, the current findings could enhance doctor-patient discussions about the trade-offs between the two strategies for individuals whose considerations will vary.
“This is a very helpful measure to understand the burden of an approach to the patient,” observed E. Magnus Ohman, MD, an interventional cardiologist at Duke University, Durham, N.C., who was not involved in the trial.
With DAOH as an endpoint, “you as a clinician get another aspect of understanding of a treatment’s impact on a multitude of endpoints.” Days out of hospital, he noted, encompasses the effects of clinical events that often go into composite clinical endpoints – not death, but including nonfatal MI, stroke, need for revascularization, and cardiovascular hospitalization.
To patients with stable CAD who ask whether the invasive approach has merits in their case, the DAOH finding “helps you to say, well, at the end of the day, you will probably be spending an equal amount of time in the hospital. Your price up front is a little bit higher, but over time, the group who gets conservative treatment will catch up.”
The DAOH outcome also avoids the limitations of an endpoint based on time to first event, “not the least of which,” said Dr. White, is that it counts only the first of what might be multiple events of varying clinical impact. Misleadingly, “you can have an event that’s a small troponin rise, but that becomes more important in a person than dying the next day.”
The DAOH analysis was based on 5,179 patients from 37 countries who averaged 64 years of age and of whom 23% were women. The endpoint considered only overnight stays in hospitals, skilled nursing facilities, rehabilitation centers, and nursing homes.
There were many more hospital or extended care facility stays overall in the invasive-management group, 4,002 versus 1,897 for those following the conservative strategy (P < .001), but the numbers flipped after excluding protocol-assigned procedures: 1,568 stays in the invasive group, compared with 1,897 (P = .001)
There were no associations between DAOH and Seattle Angina Questionnaire 7–Angina Frequency scores or DAOH interactions by age, sex, geographic region, or whether the patient had diabetes, prior MI, or heart failure, the report notes.
The primary ISCHEMIA analysis hinted at a possible long-term advantage for the invasive initial strategy in that event curves for the two arms crossed after 2-3 years, Dr. Ohman observed.
Based on that, for younger patients with stable CAD and ischemia at stress testing, “an investment of more hospital days early on might be worth it in the long run.” But ISCHEMIA, he said, “only suggests it, it doesn’t confirm it.”
The study was supported in part by grants from Arbor Pharmaceuticals and AstraZeneca. Devices or medications were provided by Abbott Vascular, Amgen, Arbor, AstraZeneca, Esperion, Medtronic, Merck Sharp & Dohme, Phillips, Omron Healthcare, and Sunovion. Dr. White disclosed receiving grants paid to his institution and fees for serving on a steering committee from Sanofi-Aventis, Regeneron, Eli Lilly, Omthera, American Regent, Eisai, DalCor, CSL Behring, Sanofi-Aventis Australia, and Esperion Therapeutics, and personal fees from Genentech and AstraZeneca. Dr. Ohman reported receiving grants from Abiomed and Cheisi USA, and consulting for Abiomed, Cara Therapeutics, Chiesi USA, Cytokinetics, Imbria Pharmaceuticals, Otsuka Pharmaceuticals, Milestone Pharmaceuticals, and XyloCor Therapeutics.
A version of this article first appeared on Medscape.com.
A ‘mess’ of a diagnosis: Is it type 2 MI or a nonischemic imposter?
Survival gains in the management of acute myocardial infarction in recent decades don’t apply to one increasingly common category of MI.
Type 2 MI, triggered by a surge in myocardial oxygen demand or a drop in its supply, is on the rise and might be more prognostically serious than the “classic” atherothrombotic type 1 form, for which there have been such impressive strides in therapy.
Strategies for assessing and treating type 2 MI and another condition it can resemble clinically – nonischemic myocardial injury – have been less rigorously explored and are far less settled.
That could be partly because recent iterations of the consensus-based universal definition of MI define type 1 MI primarily by the atherothrombotic process, whereas “demand” type 2 MI is characterized as secondary to other disorders. The list of potential primary conditions, cardiac and noncardiac, is long.
As a result, patients with type 1 MI are clinically well defined, but those with type 2 MI have so far defied efforts to be clinically characterized in a consistent way. However, recent efforts might change that, given growing appreciation that all-cause and cardiovascular (CV) mortality outcomes are actually worse for patients with type 2 MI.
“That’s because we have lots of treatments for type 1 MI. Type 2 and myocardial injury? We don’t know how to treat them,” David E. Newby, MD, PhD, University of Edinburgh, said in an interview.
Dr. Newby pointed to a widely cited 2018 publication, of which he is a coauthor, documenting 5-year outcomes of 2,122 patients with type 1 MI, type 2 MI, or nonischemic myocardial injury per the newly minted fourth universal definition.
Risk-factor profiles for patients with the latter two conditions contrasted with those of patients with type 1 MI, he observed. They were “a lot older,” were less likely to be smokers, had more hypertension and previous stroke, and a less prominent CV family history.
“So they’re a different beast,” Dr. Newby said. And their prognosis tended to be worse: all-cause mortality was about 62% for patients with type 2 MI and 72% with nonischemic myocardial injury, but only 37% for patients with type 1 MI. The difference between the two types of infarction was driven by an excess of noncardiovascular death after type 2 MI.
Mortality in patients with type 2 MI is “quite high, but it may well be a marker of the fact that you’ve got other serious diseases on board that are associated with poorer outcome,” he said.
Risk varies
The degree of risk in type 2 MI seems to vary with the underlying condition, a recent cohort study suggests. In about 3,800 patients with cardiac troponin (cTn) elevations qualifying as MI – a younger group; most were in their 30s and 40s – mortality at 10 years was 12% for those with type 1 MI, but 34% for those with type 2 MI and 46% for the remainder with nonischemic myocardial injury.
Underlying precipitating conditions varied widely among the patients with type 2 MI or nonischemic myocardial injury, and there was broad variation in mortality by etiology among those with type 2 MI. Sepsis and anemia entailed some of the highest risk, and hypertension and arrhythmias some of the lowest.
A prospective, community-based study of 5,460 patients with type 1 MI or type 2 MI reached a similar conclusion, but with a twist. Five-year all-cause mortality contrasted significantly between types of MI at 31% and 52%, respectively, but CV mortality rates were similar in this study.
Mortality in type 2 MI again varied by the precipitating etiology, suggesting that patients can be risk stratified according to pathophysiological mechanism behind their demand infarction, the authors concluded, “underscoring that type 2 MI is not a single entity, rather a group of phenotypic clusters.”
The usually high comorbidity burden and CV risk in patients with type 2 MI, one of those authors said in an interview, suggest there are “opportunities to see whether we can reduce that risk.”
Formal recommendations consistently say that, in patients with type 2 MI, “your first and foremost target should be to treat the underlying trigger and cause,” said Yader Sandoval, MD, Mayo Clinic, Rochester, Minn. That means such opportunities for further CV risk reduction tend to be “underappreciated.”
“In principle, treating the inciting cause of type 2 MI or the injury is important,” said James L. Januzzi, MD, Massachusetts General Hospital, Boston, in an interview, “but I feel quite strongly that there must be more that we can do for these folks.”
Dr. Januzzi is senior author on a recent analysis based on more than 200,000 admissions across the United States that saw a 43% lower risk for in-hospital death and 54% lower risk for 30-day MI readmission for patients with type 2 MI than those with type 1, adjusted for risk factors and comorbidities.
But, “it is important to emphasize that type 2 MI patients had a substantial risk for adverse outcome, nonetheless, and lack a clear management approach,” Dr. Januzzi and colleagues stated in their publication, as reported by this news organization.
“Due to the high rates of long-term cardiovascular events experienced by the frequently encountered type 2 MI patients,” they wrote, “identifying evidence-based therapies represents a major unmet need.”
That such patients tend to be sick with multiple comorbidities and have not yet been clinically well characterized, Dr. Januzzi said, “has stymied our ability to develop a treatment strategy.”
Role of the universal definitions
That challenge might in some ways be complicated by the universal definition, especially version 4, in which the definitions for type 1 MI, type 2 MI, and nonischemic myocardial injury are unified biochemically.
This version, published in 2018 in the European Heart Journal and Circulation, introduced a formal definition of myocardial injury, which was hailed as an innovation: cTn elevation to the 99th percentile of the upper limit of normal in a reference population.
It differentiates type 1 MI from type 2 MI by the separate pathophysiology of the ischemia – plaque rupture with intracoronary thrombosis and myocardial oxygen supply–demand mismatch, respectively. In both cases, however, there must be symptoms or objective evidence of ischemia. Absent signs of ischemia, the determination would be nonischemic myocardial injury.
Yet clinically and prognostically, type 2 MI and nonischemic myocardial injury in some ways are more similar to each other than either is to type 1 MI. Both occur secondary to other conditions across diverse clinical settings and can be a challenge to tell apart.
The universal definition’s perspective of the three events – so heavily dependent on cTn levels and myocardial ischemia – fails to account for the myriad complexities of individual patients in practice, some say, and so can muddle the process of risk assessment and therapy.
“Abnormal troponin identifies injury, but it doesn’t identify mechanism. Type 2 MI is highly prevalent, but there are other things that cause abnormal troponins,” Dr. Januzzi said. That’s why it’s important to explore and map out the clinical variables associated with the two conditions, to “understand who has a type 2 MI and who has cardiac injury. And believe it or not, it’s actually harder than it sounds to sort that out.”
“Practically speaking, the differentiation between these events is clinical,” Dr. Sandoval agreed. “There’s not always perfect agreement on what we’re calling what.”
Consequently, the universal definitions might categorize some events in ways that seem inconsistent from a management perspective. For example, they make a sharp distinction between coronary atherothrombotic and coronary nonatherothrombotic MI etiologies. Some clinicians would group MI caused by coronary spasm, coronary embolism, or spontaneous coronary artery dissection along with MI from coronary plaque rupture and thrombosis. But, Dr. Sandoval said, “even though these are coronary issues, they would fall into the type 2 bin.”
Also, about half of cases identified as type 2 MI are caused by tachyarrhythmias, which can elevate troponin and cause ECG changes and possibly symptoms resembling angina, Dr. Newby observed. “But that is completely different from other types of myocardial infarction, which are much more serious.”
So, “it’s a real mess of a diagnosis – acute myocardial injury, type 2 and type 1 MI – and it can be quite difficult to disentangle,” he said. “I think that the definition certainly has let us down.”
The diversity of type 2 MI clinical settings might also be a challenge. Myocardial injury according to cTn, with or without ischemia, occurs widely during critical illnesses and acute conditions, including respiratory distress, sepsis, internal bleeding, stroke, and pulmonary embolism.
Early in the COVID-19 pandemic, much was made of elevated troponin levels and myocarditis as an apparently frequent complication among hospitalized patients. “I raised my hand and said, we’ve been seeing abnormal troponins in people with influenza for 20 years,” Dr. Januzzi said. “Critical illness, infection, toxicity from drugs, from chemotherapy, from alcohol – there are all sorts of potential triggers of myocardial injury.”
Troponin ‘overdependence’
With many clinical settings in common and the presence or absence of myocardial ischemia to primarily distinguish them, type 2 MI and nonischemic myocardial injury both can be mistaken for the other. That can send management decisions in inappropriate directions.
A 2019 study looked at 633 cases that had been coded as type 2 MI at a major center and readjudicated them according to the fourth universal definition. Only 57% met all the type 2 criteria, 42% were reclassified as nonischemic myocardial injury, and a few were determined to have unstable angina.
“There’s overdependence on the easiest tool in the universal definition,” said Dr. Januzzi, a coauthor on that study. “Frequently people get seduced by the rise in a troponin value and immediately call it a myocardial infarction, lacking the other components of the universal definition that require evidence for coronary ischemia. That happens every day, where someone with an abnormal troponin is incorrectly branded as having an MI.”
It may not help that the current ICD-10-CM system features a diagnostic code for type 2 MI but not for myocardial injury.
“Instead, the new ICD-10-CM coding includes a proxy called ‘non-MI troponin elevation due to an underlying cause,’ ” wrote Kristian Thygesen, MD, DSc, and Allan S. Jaffe, MD, in a recent editorial. They caution against “using this code for myocardial injury because it is not specific for an elevated cTn value and could represent any abnormal laboratory measurements.” The code could be “misleading,” thereby worsening the potential for miscoding and “misattribution of MI diagnoses.”
That potential suggests there could be a growing population of patients who have been told they had an MI, which then becomes part of their medical record, when, actually, they experienced nonischemic myocardial injury.
“Having seen this occur,” Dr. Januzzi explained, “it affects people emotionally to think they’ve had an MI. Precision in diagnosis is important, which is why the universal definition is so valuable. If people would adhere to it more assiduously, we could reduce the frequency of people getting a misdiagnosis of MI when in fact they had injury.”
Still, he added, “if someone has an illness severe enough to cause myocardial injury, they’re at risk for a bad outcome regardless of whether they did or didn’t have an MI.”
The uncertain role of angiography
Angiography isn’t ordered nearly as often for patients ultimately diagnosed with type 2 MI or myocardial injury as for those with type 1 MI. Type 2 MI can hit some patients who have remained symptom free despite possibly unrecognized obstructive coronary artery disease (CAD) when myocardial demand is pushed past supply by a critical illness, tachyarrhythmia, or other acute conditions.
In such cases, “it’s reasonable to hypothesize that revascularization, something that really is not done in the vast majority of patients with type 2 MI, might actually be of benefit,” Dr. Januzzi said.
Whether these patients should routinely have angiography remains an open question. Without intervention, any newly identified obstructive CAD would continue to lurk in the background as a potential threat.
In efforts to differentiate type 2 MI from nonischemic injury, it can be “incredibly hard to know whether or not there’s actual ischemia in the mix. And that’s the only thing that defines the difference before taking an angiogram,” Derek P. Chew, MBBS, MPH, Flinders Medical Centre, Bedford Park, Australia, said in an interview.
Dr. Chew is principal investigator for the ongoing ACT-2 trial that is enrolling hospitalized, hemodynamically stable patients with cTn elevations but no suspicion of type 1 MI and “an unequivocal acute intercurrent diagnosis.” Qualifying diagnoses are prespecified on a list that includes sepsis, pneumonia, septicemia, a systemic inflammatory response, anemia, atrial tachycardia, acute kidney injury, and recent noncardiac surgery.
The patients are randomly assigned to a strategy of routine, usually invasive coronary angiography with discretionary revascularization, or to conservative care with noninvasive functional testing as appropriate. The sicker the patient, the greater the competing risk from other conditions and the less revascularization is likely to improve outcomes, Dr. Chew observed. Importantly, therefore, outcomes in the trial will be stratified by patient risk from comorbidities, measured with baseline GRACE and APACHE III scores.
Dr. Chew said the study aims to determine whether routine angiography is of benefit in patients at some identifiable level of risk, if not the whole range. One possible result, he said, is that there could be a risk-profile “sweet spot” associated with better outcomes in those assigned to angiography.
Enrollment in the trial started about 3 years ago, but “the process has been slow,” he said, because many potentially referring clinicians have a “bias on one side or another,” with about half of them preferring the angiography approach and the other half conservative management.
The unsettled role of drug therapy
With their often-complicated clinical profile, patients with type 2 MI or nonischemic myocardial injury tend to be medically undertreated, yet there is observational evidence they can benefit from familiar drug therapies.
In the previously noted cohort study of 3,800 younger patients with one of the three forms of myocardial injury, less than half of patients with type 2 MI received any form of CAD secondary prevention therapy at discharge, the researchers, with first author Avinainder Singh, MD, from Yale University, New Haven, Conn, wrote.
The finding, consistent with Dr. Newby’s study from 2018, suggests that “categorizing the type of MI in young subjects might inform long-term cardiovascular prognosis,” and “emphasizes the need to identify and implement secondary prevention strategies to mitigate the high rate of cardiovascular death in patients with type 2 MI,” they concluded.
Further, outcomes varied with the number of discharge CV meds in an older cohort of patients with myocardial injury. Those with type 2 MI or acute or chronic nonischemic myocardial injury were far less likely than patients with type 1 MI to be prescribed guideline-based drugs. Survival was greater for those on two or three classes of CV medications, compared with one or none, in patients with acute or chronic nonischemic injury.
The investigators urged that patients with nonischemic myocardial injury or type 2 MI “be treated with cardiovascular medication to a larger degree than what is done today.”
When there is documented CAD in patients with type 2 MI, “it would be reasonable to suggest that preventative secondary prevention approaches, such as such lipid-reduction therapy or aspirin, would be beneficial,” Dr. Sandoval said. “But the reality is, there are no randomized trials, there are no prospective studies. ACT-2 is one of the few and early studies that’s really trying to address this.”
“The great majority of these people are not going to the cath lab, but when they do, there seems to be a signal of potential benefit,” Dr. Januzzi said. “For someone with a type 2 MI, it’s quite possible revascularization might help. Then more long-term treatment with medications that are proven in randomized trials to reduce risk would be a very plausible intervention.”
“We’ve actually proposed a number of potential therapeutic interventions to explore, both in people with type 2 MI and in people with injury without MI,” he said. “They might include sodium glucose cotransporter 2 inhibitors. They might include antithrombotic therapy or more aggressive lipid lowering, possibly for the pleiotropic effects rather than the effects on atherosclerosis.”
Any such therapies that prove successful in well-designed trials could well earn both type 2 MI and nonischemic myocardial injury, neglected as disorders in their own right, the kind of respect in clinical care pathways that they likely deserve.
Dr. Newby has disclosed receiving consulting fees or honoraria from Eli Lilly, Roche, Toshiba, Jansen, Reckitt Benckiser Pharmaceuticals, Pfizer, AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, CellProthera, and Oncoarendi; and conducting research or receiving grants from Pfizer, AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Merck, Boehringer Ingelheim, and Inositec. Sandoval reports serving on an advisory board and as a speaker for Abbott Diagnostics and on an advisory board for Roche Diagnostics. Dr. Januzzi has disclosed receiving grant support from Novartis, Applied Therapeutics, and Innolife; consulting for Abbott Diagnostics, Janssen, Novartis, Quidel, and Roche Diagnostics; and serving on endpoint committees or data safety monitoring boards for trials supported by Abbott, AbbVie, Amgen, CVRx, Janssen, MyoKardia, and Takeda. Dr. Chew has reported receiving grants from AstraZeneca and Edwards Life Sciences. ACT-2 is sponsored by the National Medical and Health Research Council of Australia.
A version of this article first appeared on Medscape.com.
Survival gains in the management of acute myocardial infarction in recent decades don’t apply to one increasingly common category of MI.
Type 2 MI, triggered by a surge in myocardial oxygen demand or a drop in its supply, is on the rise and might be more prognostically serious than the “classic” atherothrombotic type 1 form, for which there have been such impressive strides in therapy.
Strategies for assessing and treating type 2 MI and another condition it can resemble clinically – nonischemic myocardial injury – have been less rigorously explored and are far less settled.
That could be partly because recent iterations of the consensus-based universal definition of MI define type 1 MI primarily by the atherothrombotic process, whereas “demand” type 2 MI is characterized as secondary to other disorders. The list of potential primary conditions, cardiac and noncardiac, is long.
As a result, patients with type 1 MI are clinically well defined, but those with type 2 MI have so far defied efforts to be clinically characterized in a consistent way. However, recent efforts might change that, given growing appreciation that all-cause and cardiovascular (CV) mortality outcomes are actually worse for patients with type 2 MI.
“That’s because we have lots of treatments for type 1 MI. Type 2 and myocardial injury? We don’t know how to treat them,” David E. Newby, MD, PhD, University of Edinburgh, said in an interview.
Dr. Newby pointed to a widely cited 2018 publication, of which he is a coauthor, documenting 5-year outcomes of 2,122 patients with type 1 MI, type 2 MI, or nonischemic myocardial injury per the newly minted fourth universal definition.
Risk-factor profiles for patients with the latter two conditions contrasted with those of patients with type 1 MI, he observed. They were “a lot older,” were less likely to be smokers, had more hypertension and previous stroke, and a less prominent CV family history.
“So they’re a different beast,” Dr. Newby said. And their prognosis tended to be worse: all-cause mortality was about 62% for patients with type 2 MI and 72% with nonischemic myocardial injury, but only 37% for patients with type 1 MI. The difference between the two types of infarction was driven by an excess of noncardiovascular death after type 2 MI.
Mortality in patients with type 2 MI is “quite high, but it may well be a marker of the fact that you’ve got other serious diseases on board that are associated with poorer outcome,” he said.
Risk varies
The degree of risk in type 2 MI seems to vary with the underlying condition, a recent cohort study suggests. In about 3,800 patients with cardiac troponin (cTn) elevations qualifying as MI – a younger group; most were in their 30s and 40s – mortality at 10 years was 12% for those with type 1 MI, but 34% for those with type 2 MI and 46% for the remainder with nonischemic myocardial injury.
Underlying precipitating conditions varied widely among the patients with type 2 MI or nonischemic myocardial injury, and there was broad variation in mortality by etiology among those with type 2 MI. Sepsis and anemia entailed some of the highest risk, and hypertension and arrhythmias some of the lowest.
A prospective, community-based study of 5,460 patients with type 1 MI or type 2 MI reached a similar conclusion, but with a twist. Five-year all-cause mortality contrasted significantly between types of MI at 31% and 52%, respectively, but CV mortality rates were similar in this study.
Mortality in type 2 MI again varied by the precipitating etiology, suggesting that patients can be risk stratified according to pathophysiological mechanism behind their demand infarction, the authors concluded, “underscoring that type 2 MI is not a single entity, rather a group of phenotypic clusters.”
The usually high comorbidity burden and CV risk in patients with type 2 MI, one of those authors said in an interview, suggest there are “opportunities to see whether we can reduce that risk.”
Formal recommendations consistently say that, in patients with type 2 MI, “your first and foremost target should be to treat the underlying trigger and cause,” said Yader Sandoval, MD, Mayo Clinic, Rochester, Minn. That means such opportunities for further CV risk reduction tend to be “underappreciated.”
“In principle, treating the inciting cause of type 2 MI or the injury is important,” said James L. Januzzi, MD, Massachusetts General Hospital, Boston, in an interview, “but I feel quite strongly that there must be more that we can do for these folks.”
Dr. Januzzi is senior author on a recent analysis based on more than 200,000 admissions across the United States that saw a 43% lower risk for in-hospital death and 54% lower risk for 30-day MI readmission for patients with type 2 MI than those with type 1, adjusted for risk factors and comorbidities.
But, “it is important to emphasize that type 2 MI patients had a substantial risk for adverse outcome, nonetheless, and lack a clear management approach,” Dr. Januzzi and colleagues stated in their publication, as reported by this news organization.
“Due to the high rates of long-term cardiovascular events experienced by the frequently encountered type 2 MI patients,” they wrote, “identifying evidence-based therapies represents a major unmet need.”
That such patients tend to be sick with multiple comorbidities and have not yet been clinically well characterized, Dr. Januzzi said, “has stymied our ability to develop a treatment strategy.”
Role of the universal definitions
That challenge might in some ways be complicated by the universal definition, especially version 4, in which the definitions for type 1 MI, type 2 MI, and nonischemic myocardial injury are unified biochemically.
This version, published in 2018 in the European Heart Journal and Circulation, introduced a formal definition of myocardial injury, which was hailed as an innovation: cTn elevation to the 99th percentile of the upper limit of normal in a reference population.
It differentiates type 1 MI from type 2 MI by the separate pathophysiology of the ischemia – plaque rupture with intracoronary thrombosis and myocardial oxygen supply–demand mismatch, respectively. In both cases, however, there must be symptoms or objective evidence of ischemia. Absent signs of ischemia, the determination would be nonischemic myocardial injury.
Yet clinically and prognostically, type 2 MI and nonischemic myocardial injury in some ways are more similar to each other than either is to type 1 MI. Both occur secondary to other conditions across diverse clinical settings and can be a challenge to tell apart.
The universal definition’s perspective of the three events – so heavily dependent on cTn levels and myocardial ischemia – fails to account for the myriad complexities of individual patients in practice, some say, and so can muddle the process of risk assessment and therapy.
“Abnormal troponin identifies injury, but it doesn’t identify mechanism. Type 2 MI is highly prevalent, but there are other things that cause abnormal troponins,” Dr. Januzzi said. That’s why it’s important to explore and map out the clinical variables associated with the two conditions, to “understand who has a type 2 MI and who has cardiac injury. And believe it or not, it’s actually harder than it sounds to sort that out.”
“Practically speaking, the differentiation between these events is clinical,” Dr. Sandoval agreed. “There’s not always perfect agreement on what we’re calling what.”
Consequently, the universal definitions might categorize some events in ways that seem inconsistent from a management perspective. For example, they make a sharp distinction between coronary atherothrombotic and coronary nonatherothrombotic MI etiologies. Some clinicians would group MI caused by coronary spasm, coronary embolism, or spontaneous coronary artery dissection along with MI from coronary plaque rupture and thrombosis. But, Dr. Sandoval said, “even though these are coronary issues, they would fall into the type 2 bin.”
Also, about half of cases identified as type 2 MI are caused by tachyarrhythmias, which can elevate troponin and cause ECG changes and possibly symptoms resembling angina, Dr. Newby observed. “But that is completely different from other types of myocardial infarction, which are much more serious.”
So, “it’s a real mess of a diagnosis – acute myocardial injury, type 2 and type 1 MI – and it can be quite difficult to disentangle,” he said. “I think that the definition certainly has let us down.”
The diversity of type 2 MI clinical settings might also be a challenge. Myocardial injury according to cTn, with or without ischemia, occurs widely during critical illnesses and acute conditions, including respiratory distress, sepsis, internal bleeding, stroke, and pulmonary embolism.
Early in the COVID-19 pandemic, much was made of elevated troponin levels and myocarditis as an apparently frequent complication among hospitalized patients. “I raised my hand and said, we’ve been seeing abnormal troponins in people with influenza for 20 years,” Dr. Januzzi said. “Critical illness, infection, toxicity from drugs, from chemotherapy, from alcohol – there are all sorts of potential triggers of myocardial injury.”
Troponin ‘overdependence’
With many clinical settings in common and the presence or absence of myocardial ischemia to primarily distinguish them, type 2 MI and nonischemic myocardial injury both can be mistaken for the other. That can send management decisions in inappropriate directions.
A 2019 study looked at 633 cases that had been coded as type 2 MI at a major center and readjudicated them according to the fourth universal definition. Only 57% met all the type 2 criteria, 42% were reclassified as nonischemic myocardial injury, and a few were determined to have unstable angina.
“There’s overdependence on the easiest tool in the universal definition,” said Dr. Januzzi, a coauthor on that study. “Frequently people get seduced by the rise in a troponin value and immediately call it a myocardial infarction, lacking the other components of the universal definition that require evidence for coronary ischemia. That happens every day, where someone with an abnormal troponin is incorrectly branded as having an MI.”
It may not help that the current ICD-10-CM system features a diagnostic code for type 2 MI but not for myocardial injury.
“Instead, the new ICD-10-CM coding includes a proxy called ‘non-MI troponin elevation due to an underlying cause,’ ” wrote Kristian Thygesen, MD, DSc, and Allan S. Jaffe, MD, in a recent editorial. They caution against “using this code for myocardial injury because it is not specific for an elevated cTn value and could represent any abnormal laboratory measurements.” The code could be “misleading,” thereby worsening the potential for miscoding and “misattribution of MI diagnoses.”
That potential suggests there could be a growing population of patients who have been told they had an MI, which then becomes part of their medical record, when, actually, they experienced nonischemic myocardial injury.
“Having seen this occur,” Dr. Januzzi explained, “it affects people emotionally to think they’ve had an MI. Precision in diagnosis is important, which is why the universal definition is so valuable. If people would adhere to it more assiduously, we could reduce the frequency of people getting a misdiagnosis of MI when in fact they had injury.”
Still, he added, “if someone has an illness severe enough to cause myocardial injury, they’re at risk for a bad outcome regardless of whether they did or didn’t have an MI.”
The uncertain role of angiography
Angiography isn’t ordered nearly as often for patients ultimately diagnosed with type 2 MI or myocardial injury as for those with type 1 MI. Type 2 MI can hit some patients who have remained symptom free despite possibly unrecognized obstructive coronary artery disease (CAD) when myocardial demand is pushed past supply by a critical illness, tachyarrhythmia, or other acute conditions.
In such cases, “it’s reasonable to hypothesize that revascularization, something that really is not done in the vast majority of patients with type 2 MI, might actually be of benefit,” Dr. Januzzi said.
Whether these patients should routinely have angiography remains an open question. Without intervention, any newly identified obstructive CAD would continue to lurk in the background as a potential threat.
In efforts to differentiate type 2 MI from nonischemic injury, it can be “incredibly hard to know whether or not there’s actual ischemia in the mix. And that’s the only thing that defines the difference before taking an angiogram,” Derek P. Chew, MBBS, MPH, Flinders Medical Centre, Bedford Park, Australia, said in an interview.
Dr. Chew is principal investigator for the ongoing ACT-2 trial that is enrolling hospitalized, hemodynamically stable patients with cTn elevations but no suspicion of type 1 MI and “an unequivocal acute intercurrent diagnosis.” Qualifying diagnoses are prespecified on a list that includes sepsis, pneumonia, septicemia, a systemic inflammatory response, anemia, atrial tachycardia, acute kidney injury, and recent noncardiac surgery.
The patients are randomly assigned to a strategy of routine, usually invasive coronary angiography with discretionary revascularization, or to conservative care with noninvasive functional testing as appropriate. The sicker the patient, the greater the competing risk from other conditions and the less revascularization is likely to improve outcomes, Dr. Chew observed. Importantly, therefore, outcomes in the trial will be stratified by patient risk from comorbidities, measured with baseline GRACE and APACHE III scores.
Dr. Chew said the study aims to determine whether routine angiography is of benefit in patients at some identifiable level of risk, if not the whole range. One possible result, he said, is that there could be a risk-profile “sweet spot” associated with better outcomes in those assigned to angiography.
Enrollment in the trial started about 3 years ago, but “the process has been slow,” he said, because many potentially referring clinicians have a “bias on one side or another,” with about half of them preferring the angiography approach and the other half conservative management.
The unsettled role of drug therapy
With their often-complicated clinical profile, patients with type 2 MI or nonischemic myocardial injury tend to be medically undertreated, yet there is observational evidence they can benefit from familiar drug therapies.
In the previously noted cohort study of 3,800 younger patients with one of the three forms of myocardial injury, less than half of patients with type 2 MI received any form of CAD secondary prevention therapy at discharge, the researchers, with first author Avinainder Singh, MD, from Yale University, New Haven, Conn, wrote.
The finding, consistent with Dr. Newby’s study from 2018, suggests that “categorizing the type of MI in young subjects might inform long-term cardiovascular prognosis,” and “emphasizes the need to identify and implement secondary prevention strategies to mitigate the high rate of cardiovascular death in patients with type 2 MI,” they concluded.
Further, outcomes varied with the number of discharge CV meds in an older cohort of patients with myocardial injury. Those with type 2 MI or acute or chronic nonischemic myocardial injury were far less likely than patients with type 1 MI to be prescribed guideline-based drugs. Survival was greater for those on two or three classes of CV medications, compared with one or none, in patients with acute or chronic nonischemic injury.
The investigators urged that patients with nonischemic myocardial injury or type 2 MI “be treated with cardiovascular medication to a larger degree than what is done today.”
When there is documented CAD in patients with type 2 MI, “it would be reasonable to suggest that preventative secondary prevention approaches, such as such lipid-reduction therapy or aspirin, would be beneficial,” Dr. Sandoval said. “But the reality is, there are no randomized trials, there are no prospective studies. ACT-2 is one of the few and early studies that’s really trying to address this.”
“The great majority of these people are not going to the cath lab, but when they do, there seems to be a signal of potential benefit,” Dr. Januzzi said. “For someone with a type 2 MI, it’s quite possible revascularization might help. Then more long-term treatment with medications that are proven in randomized trials to reduce risk would be a very plausible intervention.”
“We’ve actually proposed a number of potential therapeutic interventions to explore, both in people with type 2 MI and in people with injury without MI,” he said. “They might include sodium glucose cotransporter 2 inhibitors. They might include antithrombotic therapy or more aggressive lipid lowering, possibly for the pleiotropic effects rather than the effects on atherosclerosis.”
Any such therapies that prove successful in well-designed trials could well earn both type 2 MI and nonischemic myocardial injury, neglected as disorders in their own right, the kind of respect in clinical care pathways that they likely deserve.
Dr. Newby has disclosed receiving consulting fees or honoraria from Eli Lilly, Roche, Toshiba, Jansen, Reckitt Benckiser Pharmaceuticals, Pfizer, AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, CellProthera, and Oncoarendi; and conducting research or receiving grants from Pfizer, AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Merck, Boehringer Ingelheim, and Inositec. Sandoval reports serving on an advisory board and as a speaker for Abbott Diagnostics and on an advisory board for Roche Diagnostics. Dr. Januzzi has disclosed receiving grant support from Novartis, Applied Therapeutics, and Innolife; consulting for Abbott Diagnostics, Janssen, Novartis, Quidel, and Roche Diagnostics; and serving on endpoint committees or data safety monitoring boards for trials supported by Abbott, AbbVie, Amgen, CVRx, Janssen, MyoKardia, and Takeda. Dr. Chew has reported receiving grants from AstraZeneca and Edwards Life Sciences. ACT-2 is sponsored by the National Medical and Health Research Council of Australia.
A version of this article first appeared on Medscape.com.
Survival gains in the management of acute myocardial infarction in recent decades don’t apply to one increasingly common category of MI.
Type 2 MI, triggered by a surge in myocardial oxygen demand or a drop in its supply, is on the rise and might be more prognostically serious than the “classic” atherothrombotic type 1 form, for which there have been such impressive strides in therapy.
Strategies for assessing and treating type 2 MI and another condition it can resemble clinically – nonischemic myocardial injury – have been less rigorously explored and are far less settled.
That could be partly because recent iterations of the consensus-based universal definition of MI define type 1 MI primarily by the atherothrombotic process, whereas “demand” type 2 MI is characterized as secondary to other disorders. The list of potential primary conditions, cardiac and noncardiac, is long.
As a result, patients with type 1 MI are clinically well defined, but those with type 2 MI have so far defied efforts to be clinically characterized in a consistent way. However, recent efforts might change that, given growing appreciation that all-cause and cardiovascular (CV) mortality outcomes are actually worse for patients with type 2 MI.
“That’s because we have lots of treatments for type 1 MI. Type 2 and myocardial injury? We don’t know how to treat them,” David E. Newby, MD, PhD, University of Edinburgh, said in an interview.
Dr. Newby pointed to a widely cited 2018 publication, of which he is a coauthor, documenting 5-year outcomes of 2,122 patients with type 1 MI, type 2 MI, or nonischemic myocardial injury per the newly minted fourth universal definition.
Risk-factor profiles for patients with the latter two conditions contrasted with those of patients with type 1 MI, he observed. They were “a lot older,” were less likely to be smokers, had more hypertension and previous stroke, and a less prominent CV family history.
“So they’re a different beast,” Dr. Newby said. And their prognosis tended to be worse: all-cause mortality was about 62% for patients with type 2 MI and 72% with nonischemic myocardial injury, but only 37% for patients with type 1 MI. The difference between the two types of infarction was driven by an excess of noncardiovascular death after type 2 MI.
Mortality in patients with type 2 MI is “quite high, but it may well be a marker of the fact that you’ve got other serious diseases on board that are associated with poorer outcome,” he said.
Risk varies
The degree of risk in type 2 MI seems to vary with the underlying condition, a recent cohort study suggests. In about 3,800 patients with cardiac troponin (cTn) elevations qualifying as MI – a younger group; most were in their 30s and 40s – mortality at 10 years was 12% for those with type 1 MI, but 34% for those with type 2 MI and 46% for the remainder with nonischemic myocardial injury.
Underlying precipitating conditions varied widely among the patients with type 2 MI or nonischemic myocardial injury, and there was broad variation in mortality by etiology among those with type 2 MI. Sepsis and anemia entailed some of the highest risk, and hypertension and arrhythmias some of the lowest.
A prospective, community-based study of 5,460 patients with type 1 MI or type 2 MI reached a similar conclusion, but with a twist. Five-year all-cause mortality contrasted significantly between types of MI at 31% and 52%, respectively, but CV mortality rates were similar in this study.
Mortality in type 2 MI again varied by the precipitating etiology, suggesting that patients can be risk stratified according to pathophysiological mechanism behind their demand infarction, the authors concluded, “underscoring that type 2 MI is not a single entity, rather a group of phenotypic clusters.”
The usually high comorbidity burden and CV risk in patients with type 2 MI, one of those authors said in an interview, suggest there are “opportunities to see whether we can reduce that risk.”
Formal recommendations consistently say that, in patients with type 2 MI, “your first and foremost target should be to treat the underlying trigger and cause,” said Yader Sandoval, MD, Mayo Clinic, Rochester, Minn. That means such opportunities for further CV risk reduction tend to be “underappreciated.”
“In principle, treating the inciting cause of type 2 MI or the injury is important,” said James L. Januzzi, MD, Massachusetts General Hospital, Boston, in an interview, “but I feel quite strongly that there must be more that we can do for these folks.”
Dr. Januzzi is senior author on a recent analysis based on more than 200,000 admissions across the United States that saw a 43% lower risk for in-hospital death and 54% lower risk for 30-day MI readmission for patients with type 2 MI than those with type 1, adjusted for risk factors and comorbidities.
But, “it is important to emphasize that type 2 MI patients had a substantial risk for adverse outcome, nonetheless, and lack a clear management approach,” Dr. Januzzi and colleagues stated in their publication, as reported by this news organization.
“Due to the high rates of long-term cardiovascular events experienced by the frequently encountered type 2 MI patients,” they wrote, “identifying evidence-based therapies represents a major unmet need.”
That such patients tend to be sick with multiple comorbidities and have not yet been clinically well characterized, Dr. Januzzi said, “has stymied our ability to develop a treatment strategy.”
Role of the universal definitions
That challenge might in some ways be complicated by the universal definition, especially version 4, in which the definitions for type 1 MI, type 2 MI, and nonischemic myocardial injury are unified biochemically.
This version, published in 2018 in the European Heart Journal and Circulation, introduced a formal definition of myocardial injury, which was hailed as an innovation: cTn elevation to the 99th percentile of the upper limit of normal in a reference population.
It differentiates type 1 MI from type 2 MI by the separate pathophysiology of the ischemia – plaque rupture with intracoronary thrombosis and myocardial oxygen supply–demand mismatch, respectively. In both cases, however, there must be symptoms or objective evidence of ischemia. Absent signs of ischemia, the determination would be nonischemic myocardial injury.
Yet clinically and prognostically, type 2 MI and nonischemic myocardial injury in some ways are more similar to each other than either is to type 1 MI. Both occur secondary to other conditions across diverse clinical settings and can be a challenge to tell apart.
The universal definition’s perspective of the three events – so heavily dependent on cTn levels and myocardial ischemia – fails to account for the myriad complexities of individual patients in practice, some say, and so can muddle the process of risk assessment and therapy.
“Abnormal troponin identifies injury, but it doesn’t identify mechanism. Type 2 MI is highly prevalent, but there are other things that cause abnormal troponins,” Dr. Januzzi said. That’s why it’s important to explore and map out the clinical variables associated with the two conditions, to “understand who has a type 2 MI and who has cardiac injury. And believe it or not, it’s actually harder than it sounds to sort that out.”
“Practically speaking, the differentiation between these events is clinical,” Dr. Sandoval agreed. “There’s not always perfect agreement on what we’re calling what.”
Consequently, the universal definitions might categorize some events in ways that seem inconsistent from a management perspective. For example, they make a sharp distinction between coronary atherothrombotic and coronary nonatherothrombotic MI etiologies. Some clinicians would group MI caused by coronary spasm, coronary embolism, or spontaneous coronary artery dissection along with MI from coronary plaque rupture and thrombosis. But, Dr. Sandoval said, “even though these are coronary issues, they would fall into the type 2 bin.”
Also, about half of cases identified as type 2 MI are caused by tachyarrhythmias, which can elevate troponin and cause ECG changes and possibly symptoms resembling angina, Dr. Newby observed. “But that is completely different from other types of myocardial infarction, which are much more serious.”
So, “it’s a real mess of a diagnosis – acute myocardial injury, type 2 and type 1 MI – and it can be quite difficult to disentangle,” he said. “I think that the definition certainly has let us down.”
The diversity of type 2 MI clinical settings might also be a challenge. Myocardial injury according to cTn, with or without ischemia, occurs widely during critical illnesses and acute conditions, including respiratory distress, sepsis, internal bleeding, stroke, and pulmonary embolism.
Early in the COVID-19 pandemic, much was made of elevated troponin levels and myocarditis as an apparently frequent complication among hospitalized patients. “I raised my hand and said, we’ve been seeing abnormal troponins in people with influenza for 20 years,” Dr. Januzzi said. “Critical illness, infection, toxicity from drugs, from chemotherapy, from alcohol – there are all sorts of potential triggers of myocardial injury.”
Troponin ‘overdependence’
With many clinical settings in common and the presence or absence of myocardial ischemia to primarily distinguish them, type 2 MI and nonischemic myocardial injury both can be mistaken for the other. That can send management decisions in inappropriate directions.
A 2019 study looked at 633 cases that had been coded as type 2 MI at a major center and readjudicated them according to the fourth universal definition. Only 57% met all the type 2 criteria, 42% were reclassified as nonischemic myocardial injury, and a few were determined to have unstable angina.
“There’s overdependence on the easiest tool in the universal definition,” said Dr. Januzzi, a coauthor on that study. “Frequently people get seduced by the rise in a troponin value and immediately call it a myocardial infarction, lacking the other components of the universal definition that require evidence for coronary ischemia. That happens every day, where someone with an abnormal troponin is incorrectly branded as having an MI.”
It may not help that the current ICD-10-CM system features a diagnostic code for type 2 MI but not for myocardial injury.
“Instead, the new ICD-10-CM coding includes a proxy called ‘non-MI troponin elevation due to an underlying cause,’ ” wrote Kristian Thygesen, MD, DSc, and Allan S. Jaffe, MD, in a recent editorial. They caution against “using this code for myocardial injury because it is not specific for an elevated cTn value and could represent any abnormal laboratory measurements.” The code could be “misleading,” thereby worsening the potential for miscoding and “misattribution of MI diagnoses.”
That potential suggests there could be a growing population of patients who have been told they had an MI, which then becomes part of their medical record, when, actually, they experienced nonischemic myocardial injury.
“Having seen this occur,” Dr. Januzzi explained, “it affects people emotionally to think they’ve had an MI. Precision in diagnosis is important, which is why the universal definition is so valuable. If people would adhere to it more assiduously, we could reduce the frequency of people getting a misdiagnosis of MI when in fact they had injury.”
Still, he added, “if someone has an illness severe enough to cause myocardial injury, they’re at risk for a bad outcome regardless of whether they did or didn’t have an MI.”
The uncertain role of angiography
Angiography isn’t ordered nearly as often for patients ultimately diagnosed with type 2 MI or myocardial injury as for those with type 1 MI. Type 2 MI can hit some patients who have remained symptom free despite possibly unrecognized obstructive coronary artery disease (CAD) when myocardial demand is pushed past supply by a critical illness, tachyarrhythmia, or other acute conditions.
In such cases, “it’s reasonable to hypothesize that revascularization, something that really is not done in the vast majority of patients with type 2 MI, might actually be of benefit,” Dr. Januzzi said.
Whether these patients should routinely have angiography remains an open question. Without intervention, any newly identified obstructive CAD would continue to lurk in the background as a potential threat.
In efforts to differentiate type 2 MI from nonischemic injury, it can be “incredibly hard to know whether or not there’s actual ischemia in the mix. And that’s the only thing that defines the difference before taking an angiogram,” Derek P. Chew, MBBS, MPH, Flinders Medical Centre, Bedford Park, Australia, said in an interview.
Dr. Chew is principal investigator for the ongoing ACT-2 trial that is enrolling hospitalized, hemodynamically stable patients with cTn elevations but no suspicion of type 1 MI and “an unequivocal acute intercurrent diagnosis.” Qualifying diagnoses are prespecified on a list that includes sepsis, pneumonia, septicemia, a systemic inflammatory response, anemia, atrial tachycardia, acute kidney injury, and recent noncardiac surgery.
The patients are randomly assigned to a strategy of routine, usually invasive coronary angiography with discretionary revascularization, or to conservative care with noninvasive functional testing as appropriate. The sicker the patient, the greater the competing risk from other conditions and the less revascularization is likely to improve outcomes, Dr. Chew observed. Importantly, therefore, outcomes in the trial will be stratified by patient risk from comorbidities, measured with baseline GRACE and APACHE III scores.
Dr. Chew said the study aims to determine whether routine angiography is of benefit in patients at some identifiable level of risk, if not the whole range. One possible result, he said, is that there could be a risk-profile “sweet spot” associated with better outcomes in those assigned to angiography.
Enrollment in the trial started about 3 years ago, but “the process has been slow,” he said, because many potentially referring clinicians have a “bias on one side or another,” with about half of them preferring the angiography approach and the other half conservative management.
The unsettled role of drug therapy
With their often-complicated clinical profile, patients with type 2 MI or nonischemic myocardial injury tend to be medically undertreated, yet there is observational evidence they can benefit from familiar drug therapies.
In the previously noted cohort study of 3,800 younger patients with one of the three forms of myocardial injury, less than half of patients with type 2 MI received any form of CAD secondary prevention therapy at discharge, the researchers, with first author Avinainder Singh, MD, from Yale University, New Haven, Conn, wrote.
The finding, consistent with Dr. Newby’s study from 2018, suggests that “categorizing the type of MI in young subjects might inform long-term cardiovascular prognosis,” and “emphasizes the need to identify and implement secondary prevention strategies to mitigate the high rate of cardiovascular death in patients with type 2 MI,” they concluded.
Further, outcomes varied with the number of discharge CV meds in an older cohort of patients with myocardial injury. Those with type 2 MI or acute or chronic nonischemic myocardial injury were far less likely than patients with type 1 MI to be prescribed guideline-based drugs. Survival was greater for those on two or three classes of CV medications, compared with one or none, in patients with acute or chronic nonischemic injury.
The investigators urged that patients with nonischemic myocardial injury or type 2 MI “be treated with cardiovascular medication to a larger degree than what is done today.”
When there is documented CAD in patients with type 2 MI, “it would be reasonable to suggest that preventative secondary prevention approaches, such as such lipid-reduction therapy or aspirin, would be beneficial,” Dr. Sandoval said. “But the reality is, there are no randomized trials, there are no prospective studies. ACT-2 is one of the few and early studies that’s really trying to address this.”
“The great majority of these people are not going to the cath lab, but when they do, there seems to be a signal of potential benefit,” Dr. Januzzi said. “For someone with a type 2 MI, it’s quite possible revascularization might help. Then more long-term treatment with medications that are proven in randomized trials to reduce risk would be a very plausible intervention.”
“We’ve actually proposed a number of potential therapeutic interventions to explore, both in people with type 2 MI and in people with injury without MI,” he said. “They might include sodium glucose cotransporter 2 inhibitors. They might include antithrombotic therapy or more aggressive lipid lowering, possibly for the pleiotropic effects rather than the effects on atherosclerosis.”
Any such therapies that prove successful in well-designed trials could well earn both type 2 MI and nonischemic myocardial injury, neglected as disorders in their own right, the kind of respect in clinical care pathways that they likely deserve.
Dr. Newby has disclosed receiving consulting fees or honoraria from Eli Lilly, Roche, Toshiba, Jansen, Reckitt Benckiser Pharmaceuticals, Pfizer, AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, CellProthera, and Oncoarendi; and conducting research or receiving grants from Pfizer, AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Merck, Boehringer Ingelheim, and Inositec. Sandoval reports serving on an advisory board and as a speaker for Abbott Diagnostics and on an advisory board for Roche Diagnostics. Dr. Januzzi has disclosed receiving grant support from Novartis, Applied Therapeutics, and Innolife; consulting for Abbott Diagnostics, Janssen, Novartis, Quidel, and Roche Diagnostics; and serving on endpoint committees or data safety monitoring boards for trials supported by Abbott, AbbVie, Amgen, CVRx, Janssen, MyoKardia, and Takeda. Dr. Chew has reported receiving grants from AstraZeneca and Edwards Life Sciences. ACT-2 is sponsored by the National Medical and Health Research Council of Australia.
A version of this article first appeared on Medscape.com.
Finerenone scores second pivotal-trial success in patients with diabetic kidney disease
Finerenone, an investigational agent from a new drug class, just scored a second pivotal trial win after showing significant benefit for slowing progression of diabetic kidney disease in patients with type 2 diabetes in the FIDELIO-DKD pivotal trial with more than 5,700 patients.
Top-line results from FIGARO-DKD showed significant benefit for the primary endpoint of cardiovascular death and nonfatal cardiovascular disease endpoints in a placebo-controlled trial with about 7,400 patients with type 2 diabetes, reported Bayer, the company developing finerenone in statement released on May 10, 2021.
Based on the FIDELIO-DKD results, finerenone is currently under review by the U.S. Food and Drug Administration for marketing approval as a treatment for patients with type 2 diabetes and chronic kidney disease. FIDELIO-DKD, in addition to the primary endpoint that focused on slowing progression of diabetic kidney disease, had a secondary endpoint that assessed the combined incidence on treatment of cardiovascular death, or nonfatal episodes of stroke, MI, or hospitalization for heart failure. Results from the study published in 2020 in the New England Journal of Medicine showed that finerenone was safe and effective for both endpoints.
In the current study, FIGARO-DKD, run at more than 1,000 sites in 47 countries, these endpoints flipped. The primary outcome was a composite of cardiovascular death or nonfatal cardiovascular disease events, and the secondary outcome was prevention of DKD progression.
Other than stating the results significantly fulfilled FIGARO-DKD’s primary endpoint of reducing the incidence of combined cardiovascular disease endpoints, the release gave no further outcome details. The release noted that the enrolled patient cohort in FIGARO-DKD included more patients with earlier-stage chronic kidney disease, compared with FIDELIO-DKD.
Finerenone is a first-in-class investigational nonsteroidal, selective mineralocorticoid receptor antagonist (MRA). As an MRA it shares certain activities with the steroidal MRAs spironolactone and eplerenone. But the absence of a steroidal structure means that finerenone does not cause steroidal adverse effects such as gynecomastia. Results in FIDELIO-DKD showed that finerenone caused more hyperkalemia than placebo, but the level of hyperkalemia that it causes relative to spironolactone or eplerenone remains uncertain.
Finerenone, an investigational agent from a new drug class, just scored a second pivotal trial win after showing significant benefit for slowing progression of diabetic kidney disease in patients with type 2 diabetes in the FIDELIO-DKD pivotal trial with more than 5,700 patients.
Top-line results from FIGARO-DKD showed significant benefit for the primary endpoint of cardiovascular death and nonfatal cardiovascular disease endpoints in a placebo-controlled trial with about 7,400 patients with type 2 diabetes, reported Bayer, the company developing finerenone in statement released on May 10, 2021.
Based on the FIDELIO-DKD results, finerenone is currently under review by the U.S. Food and Drug Administration for marketing approval as a treatment for patients with type 2 diabetes and chronic kidney disease. FIDELIO-DKD, in addition to the primary endpoint that focused on slowing progression of diabetic kidney disease, had a secondary endpoint that assessed the combined incidence on treatment of cardiovascular death, or nonfatal episodes of stroke, MI, or hospitalization for heart failure. Results from the study published in 2020 in the New England Journal of Medicine showed that finerenone was safe and effective for both endpoints.
In the current study, FIGARO-DKD, run at more than 1,000 sites in 47 countries, these endpoints flipped. The primary outcome was a composite of cardiovascular death or nonfatal cardiovascular disease events, and the secondary outcome was prevention of DKD progression.
Other than stating the results significantly fulfilled FIGARO-DKD’s primary endpoint of reducing the incidence of combined cardiovascular disease endpoints, the release gave no further outcome details. The release noted that the enrolled patient cohort in FIGARO-DKD included more patients with earlier-stage chronic kidney disease, compared with FIDELIO-DKD.
Finerenone is a first-in-class investigational nonsteroidal, selective mineralocorticoid receptor antagonist (MRA). As an MRA it shares certain activities with the steroidal MRAs spironolactone and eplerenone. But the absence of a steroidal structure means that finerenone does not cause steroidal adverse effects such as gynecomastia. Results in FIDELIO-DKD showed that finerenone caused more hyperkalemia than placebo, but the level of hyperkalemia that it causes relative to spironolactone or eplerenone remains uncertain.
Finerenone, an investigational agent from a new drug class, just scored a second pivotal trial win after showing significant benefit for slowing progression of diabetic kidney disease in patients with type 2 diabetes in the FIDELIO-DKD pivotal trial with more than 5,700 patients.
Top-line results from FIGARO-DKD showed significant benefit for the primary endpoint of cardiovascular death and nonfatal cardiovascular disease endpoints in a placebo-controlled trial with about 7,400 patients with type 2 diabetes, reported Bayer, the company developing finerenone in statement released on May 10, 2021.
Based on the FIDELIO-DKD results, finerenone is currently under review by the U.S. Food and Drug Administration for marketing approval as a treatment for patients with type 2 diabetes and chronic kidney disease. FIDELIO-DKD, in addition to the primary endpoint that focused on slowing progression of diabetic kidney disease, had a secondary endpoint that assessed the combined incidence on treatment of cardiovascular death, or nonfatal episodes of stroke, MI, or hospitalization for heart failure. Results from the study published in 2020 in the New England Journal of Medicine showed that finerenone was safe and effective for both endpoints.
In the current study, FIGARO-DKD, run at more than 1,000 sites in 47 countries, these endpoints flipped. The primary outcome was a composite of cardiovascular death or nonfatal cardiovascular disease events, and the secondary outcome was prevention of DKD progression.
Other than stating the results significantly fulfilled FIGARO-DKD’s primary endpoint of reducing the incidence of combined cardiovascular disease endpoints, the release gave no further outcome details. The release noted that the enrolled patient cohort in FIGARO-DKD included more patients with earlier-stage chronic kidney disease, compared with FIDELIO-DKD.
Finerenone is a first-in-class investigational nonsteroidal, selective mineralocorticoid receptor antagonist (MRA). As an MRA it shares certain activities with the steroidal MRAs spironolactone and eplerenone. But the absence of a steroidal structure means that finerenone does not cause steroidal adverse effects such as gynecomastia. Results in FIDELIO-DKD showed that finerenone caused more hyperkalemia than placebo, but the level of hyperkalemia that it causes relative to spironolactone or eplerenone remains uncertain.
A simple new definition for ‘metabolically healthy obesity’?
Scientists have proposed a simple new definition for “metabolically healthy obesity” to identify individuals who do not have an increased risk of cardiovascular disease (CVD) death and total mortality.
The team – led by Anika Zembic, MPH, German Institute of Human Nutrition Potsdam-Rehbruecke, Nuthetal, Germany – performed an assessment of anthropometric and metabolic risk factors as well as mortality data from two cohorts that “yielded a simple definition to categorize participants with obesity as metabolically healthy or unhealthy.”
They defined “metabolically healthy” as systolic blood pressure <130 mm Hg and no use of blood pressure-lowering medication; waist-to-hip ratio <0.95 (in women) and <1.03 (in men); and no prevalent type 2 diabetes.
Based on this new definition, 42% of participants in the third U.S. National Health and Nutrition Examination Survey (NHANES-III) and 19% of participants in the UK Biobank study had metabolically healthy obesity and did not have an increased risk for CVD mortality and total mortality compared with individuals with metabolically healthy normal weight.
“People with a phenotype defined as metabolically unhealthy using this definition had significantly higher hazard ratios for [CVD] mortality and total mortality irrespective of body mass index category, and people with phenotypes defined as having metabolically healthy obesity displayed no increased risk,” the researchers noted in their article, published May 7 in JAMA Network Open.
“Our new definition may be important not only to stratify risk of mortality in people with obesity, but also in people with overweight and normal weight,” they concluded.
Thirty different definitions of ‘metabolically healthy obesity’
“To date, there is no universally accepted standard for defining [metabolically healthy obesity] and more than 30 different definitions have been used to operationalize the phenotypes in studies,” which may explain the “continued unresolved debate” about outcomes in patients with metabolically unhealthy obesity, Ayana K. April-Sanders, PhD, and Carlos J. Rodriguez, MD, MPH, from Albert Einstein College of Medicine, New York, wrote in an accompanying commentary.
The current study, they noted, suggests that waist-to-hip ratio is a better measure of central adiposity than waist circumference, and that the effect of dyslipidemia on CVD mortality may be weaker among individuals with obesity.
However, the findings may not be generalizable to other CVD outcomes, they cautioned.
And importantly, some individuals with metabolically healthy obesity will likely transition to unhealthy obesity over time due to weight gain, aging, and lack of physical activity.
Therefore, “the present study provides a prototype of how that definition can be derived, but more rigorous tests and evidence using similar techniques are needed, particularly in prospective studies,” according to Dr. April-Sanders and Dr. Rodriguez.
They call for more research to establish a standardized definition of metabolically healthy obesity and then, using that definition, to determine the prevalence of healthy and unhealthy obesity and identify factors that preserve healthy obesity.
Definition developed from NHANES cohort, validated in UK biobank
Ms. Zembic and colleagues explained that previous definitions for metabolically healthy obesity were mainly based on the absence of either metabolic syndrome or insulin resistance, but some individuals with obesity but without metabolic disease still have increased risks of CVD mortality and total mortality.
To develop a more precise definition of metabolically healthy obesity, the researchers analyzed data from 12,341 individuals in the United States who participated in NHANES-III, conducted between 1988 and 1994. The individuals were a mean age of 42 and 51% were women, and they were followed for an average of 14.5 years.
The researchers validated this definition using data from 374,079 individuals in the population-based UK Biobank cohort who were assessed in 2006 to 2010. Those individuals were a mean age of 56 and 55% were women, and they were followed for a mean of 7.8 years.
The combination of systolic blood pressure and waist-to-hip ratio had the strongest association with CVD mortality and total mortality, and the prevalence of type 2 diabetes was also associated with greater risk.
Regardless of BMI, all groups of metabolically unhealthy individuals had increased risks of CVD mortality and total mortality.
The study and some of the researchers were supported by grants from the German Federal Ministry of Education and Research.
A version of this article first appeared on Medscape.com.
Scientists have proposed a simple new definition for “metabolically healthy obesity” to identify individuals who do not have an increased risk of cardiovascular disease (CVD) death and total mortality.
The team – led by Anika Zembic, MPH, German Institute of Human Nutrition Potsdam-Rehbruecke, Nuthetal, Germany – performed an assessment of anthropometric and metabolic risk factors as well as mortality data from two cohorts that “yielded a simple definition to categorize participants with obesity as metabolically healthy or unhealthy.”
They defined “metabolically healthy” as systolic blood pressure <130 mm Hg and no use of blood pressure-lowering medication; waist-to-hip ratio <0.95 (in women) and <1.03 (in men); and no prevalent type 2 diabetes.
Based on this new definition, 42% of participants in the third U.S. National Health and Nutrition Examination Survey (NHANES-III) and 19% of participants in the UK Biobank study had metabolically healthy obesity and did not have an increased risk for CVD mortality and total mortality compared with individuals with metabolically healthy normal weight.
“People with a phenotype defined as metabolically unhealthy using this definition had significantly higher hazard ratios for [CVD] mortality and total mortality irrespective of body mass index category, and people with phenotypes defined as having metabolically healthy obesity displayed no increased risk,” the researchers noted in their article, published May 7 in JAMA Network Open.
“Our new definition may be important not only to stratify risk of mortality in people with obesity, but also in people with overweight and normal weight,” they concluded.
Thirty different definitions of ‘metabolically healthy obesity’
“To date, there is no universally accepted standard for defining [metabolically healthy obesity] and more than 30 different definitions have been used to operationalize the phenotypes in studies,” which may explain the “continued unresolved debate” about outcomes in patients with metabolically unhealthy obesity, Ayana K. April-Sanders, PhD, and Carlos J. Rodriguez, MD, MPH, from Albert Einstein College of Medicine, New York, wrote in an accompanying commentary.
The current study, they noted, suggests that waist-to-hip ratio is a better measure of central adiposity than waist circumference, and that the effect of dyslipidemia on CVD mortality may be weaker among individuals with obesity.
However, the findings may not be generalizable to other CVD outcomes, they cautioned.
And importantly, some individuals with metabolically healthy obesity will likely transition to unhealthy obesity over time due to weight gain, aging, and lack of physical activity.
Therefore, “the present study provides a prototype of how that definition can be derived, but more rigorous tests and evidence using similar techniques are needed, particularly in prospective studies,” according to Dr. April-Sanders and Dr. Rodriguez.
They call for more research to establish a standardized definition of metabolically healthy obesity and then, using that definition, to determine the prevalence of healthy and unhealthy obesity and identify factors that preserve healthy obesity.
Definition developed from NHANES cohort, validated in UK biobank
Ms. Zembic and colleagues explained that previous definitions for metabolically healthy obesity were mainly based on the absence of either metabolic syndrome or insulin resistance, but some individuals with obesity but without metabolic disease still have increased risks of CVD mortality and total mortality.
To develop a more precise definition of metabolically healthy obesity, the researchers analyzed data from 12,341 individuals in the United States who participated in NHANES-III, conducted between 1988 and 1994. The individuals were a mean age of 42 and 51% were women, and they were followed for an average of 14.5 years.
The researchers validated this definition using data from 374,079 individuals in the population-based UK Biobank cohort who were assessed in 2006 to 2010. Those individuals were a mean age of 56 and 55% were women, and they were followed for a mean of 7.8 years.
The combination of systolic blood pressure and waist-to-hip ratio had the strongest association with CVD mortality and total mortality, and the prevalence of type 2 diabetes was also associated with greater risk.
Regardless of BMI, all groups of metabolically unhealthy individuals had increased risks of CVD mortality and total mortality.
The study and some of the researchers were supported by grants from the German Federal Ministry of Education and Research.
A version of this article first appeared on Medscape.com.
Scientists have proposed a simple new definition for “metabolically healthy obesity” to identify individuals who do not have an increased risk of cardiovascular disease (CVD) death and total mortality.
The team – led by Anika Zembic, MPH, German Institute of Human Nutrition Potsdam-Rehbruecke, Nuthetal, Germany – performed an assessment of anthropometric and metabolic risk factors as well as mortality data from two cohorts that “yielded a simple definition to categorize participants with obesity as metabolically healthy or unhealthy.”
They defined “metabolically healthy” as systolic blood pressure <130 mm Hg and no use of blood pressure-lowering medication; waist-to-hip ratio <0.95 (in women) and <1.03 (in men); and no prevalent type 2 diabetes.
Based on this new definition, 42% of participants in the third U.S. National Health and Nutrition Examination Survey (NHANES-III) and 19% of participants in the UK Biobank study had metabolically healthy obesity and did not have an increased risk for CVD mortality and total mortality compared with individuals with metabolically healthy normal weight.
“People with a phenotype defined as metabolically unhealthy using this definition had significantly higher hazard ratios for [CVD] mortality and total mortality irrespective of body mass index category, and people with phenotypes defined as having metabolically healthy obesity displayed no increased risk,” the researchers noted in their article, published May 7 in JAMA Network Open.
“Our new definition may be important not only to stratify risk of mortality in people with obesity, but also in people with overweight and normal weight,” they concluded.
Thirty different definitions of ‘metabolically healthy obesity’
“To date, there is no universally accepted standard for defining [metabolically healthy obesity] and more than 30 different definitions have been used to operationalize the phenotypes in studies,” which may explain the “continued unresolved debate” about outcomes in patients with metabolically unhealthy obesity, Ayana K. April-Sanders, PhD, and Carlos J. Rodriguez, MD, MPH, from Albert Einstein College of Medicine, New York, wrote in an accompanying commentary.
The current study, they noted, suggests that waist-to-hip ratio is a better measure of central adiposity than waist circumference, and that the effect of dyslipidemia on CVD mortality may be weaker among individuals with obesity.
However, the findings may not be generalizable to other CVD outcomes, they cautioned.
And importantly, some individuals with metabolically healthy obesity will likely transition to unhealthy obesity over time due to weight gain, aging, and lack of physical activity.
Therefore, “the present study provides a prototype of how that definition can be derived, but more rigorous tests and evidence using similar techniques are needed, particularly in prospective studies,” according to Dr. April-Sanders and Dr. Rodriguez.
They call for more research to establish a standardized definition of metabolically healthy obesity and then, using that definition, to determine the prevalence of healthy and unhealthy obesity and identify factors that preserve healthy obesity.
Definition developed from NHANES cohort, validated in UK biobank
Ms. Zembic and colleagues explained that previous definitions for metabolically healthy obesity were mainly based on the absence of either metabolic syndrome or insulin resistance, but some individuals with obesity but without metabolic disease still have increased risks of CVD mortality and total mortality.
To develop a more precise definition of metabolically healthy obesity, the researchers analyzed data from 12,341 individuals in the United States who participated in NHANES-III, conducted between 1988 and 1994. The individuals were a mean age of 42 and 51% were women, and they were followed for an average of 14.5 years.
The researchers validated this definition using data from 374,079 individuals in the population-based UK Biobank cohort who were assessed in 2006 to 2010. Those individuals were a mean age of 56 and 55% were women, and they were followed for a mean of 7.8 years.
The combination of systolic blood pressure and waist-to-hip ratio had the strongest association with CVD mortality and total mortality, and the prevalence of type 2 diabetes was also associated with greater risk.
Regardless of BMI, all groups of metabolically unhealthy individuals had increased risks of CVD mortality and total mortality.
The study and some of the researchers were supported by grants from the German Federal Ministry of Education and Research.
A version of this article first appeared on Medscape.com.
Hypertension worsened by commonly used prescription meds
Nearly half of these American adults had hypertension, and in this subgroup, 18.5% reported using a prescription drug known to increase blood pressure. The most widely used class of agents with this effect was antidepressants, used by 8.7%; followed by nonsteroidal anti-inflammatory drugs (NSAIDs), used by 6.5%; steroids, 1.9%; estrogens, 1.7%; and several other agents each used by fewer than 1% of the study cohort, John Vitarello, MD, said during a press briefing on reports from the upcoming annual scientific sessions of the American College of Cardiology.
He and his associates estimated that this use of prescription drugs known to raise blood pressure could be what stands in the way of some 560,000-2.2 million Americans from having their hypertension under control, depending on the exact blood pressure impact that various pressure-increasing drugs have and presuming that half of those on blood pressure increasing agents could stop them and switch to alternative agents, said Dr. Vitarello, a researcher at Beth Israel Deaconess Medical Center in Boston.
He also highlighted that the study assessed only prescription drugs and did not examine OTC drug use, which may be especially relevant for the many people who regularly take NSAIDs.
“Clinicians should review the prescription and OTC drug use of patients with hypertension and consider stopping drugs that increase blood pressure or switching the patient to alternatives” that are blood pressure neutral, Dr. Vitarello said during the briefing. He cautioned that maintaining hypertensive patients on agents that raise their blood pressure can result in “prescribing cascades” where taking drugs that boost blood pressure results in need for intensified antihypertensive treatment.
An opportunity for NSAID alternatives
“This study hopefully raises awareness that there is a very high use of medications that increase blood pressure, and use of OTC agents could increase the rate even higher” said Eugene Yang, MD, a cardiologist and codirector of the Cardiovascular Wellness and Prevention Program of the University of Washington, Seattle. Substituting for certain antidepressant agents may often not be realistic, but an opportunity exists for reducing NSAID use, a class also linked with an increased risk for bleeding and other adverse effects, Dr. Yang said during the briefing. Minimizing use of NSAIDs including ibuprofen and naproxen use “is something to think about,” he suggested.
“The effect of NSAIDs on blood pressure is not well studied and can vary from person to person” noted Dr. Vitarello, who added that higher NSAID dosages and more prolonged use likely increase the risk for an adverse effect on blood pressure. One reasonable option is to encourage patients to use an alternative class of pain reliever such as acetaminophen.
It remains “a challenge” to discern differences in adverse blood pressure effects, and in all adverse cardiovascular effects among different NSAIDs, said Dr. Yang. Results from “some studies show that certain NSAIDs may be safer, but other studies did not. We need to be very careful using NSAIDs because, on average, they increase blood pressure by about 3 mm Hg. We need to be mindful to try to prescribe alternative agents, like acetaminophen.”
A decade of data from NHANES
The analysis run by Dr. Vitarello and associates used data from 27,599 American adults included in the NHANES during 2009-2018, and focused on the 44% who either had an average blood pressure measurement of at least 130/80 mm Hg or reported having ever been told by a clinician that they had hypertension. The NHANES assessments included the prescription medications taken by each participant. The prevalence of using at least one prescription drug known to raise blood pressure was 24% among women and 14% among men, and 4% of those with hypertension were on two or more pressure-increasing agents.
The researchers based their identification of pressure-increasing prescription drugs on the list included in the 2017 guideline for managing high blood pressure from the American College of Cardiology and American Heart Association. This list specifies that the antidepressants that raise blood pressure are the monoamine oxidase inhibitors, serotonin norepinephrine reuptake inhibitors, and tricyclic antidepressants.
Dr. Vitarello and Dr. Yang had no disclosures.
Nearly half of these American adults had hypertension, and in this subgroup, 18.5% reported using a prescription drug known to increase blood pressure. The most widely used class of agents with this effect was antidepressants, used by 8.7%; followed by nonsteroidal anti-inflammatory drugs (NSAIDs), used by 6.5%; steroids, 1.9%; estrogens, 1.7%; and several other agents each used by fewer than 1% of the study cohort, John Vitarello, MD, said during a press briefing on reports from the upcoming annual scientific sessions of the American College of Cardiology.
He and his associates estimated that this use of prescription drugs known to raise blood pressure could be what stands in the way of some 560,000-2.2 million Americans from having their hypertension under control, depending on the exact blood pressure impact that various pressure-increasing drugs have and presuming that half of those on blood pressure increasing agents could stop them and switch to alternative agents, said Dr. Vitarello, a researcher at Beth Israel Deaconess Medical Center in Boston.
He also highlighted that the study assessed only prescription drugs and did not examine OTC drug use, which may be especially relevant for the many people who regularly take NSAIDs.
“Clinicians should review the prescription and OTC drug use of patients with hypertension and consider stopping drugs that increase blood pressure or switching the patient to alternatives” that are blood pressure neutral, Dr. Vitarello said during the briefing. He cautioned that maintaining hypertensive patients on agents that raise their blood pressure can result in “prescribing cascades” where taking drugs that boost blood pressure results in need for intensified antihypertensive treatment.
An opportunity for NSAID alternatives
“This study hopefully raises awareness that there is a very high use of medications that increase blood pressure, and use of OTC agents could increase the rate even higher” said Eugene Yang, MD, a cardiologist and codirector of the Cardiovascular Wellness and Prevention Program of the University of Washington, Seattle. Substituting for certain antidepressant agents may often not be realistic, but an opportunity exists for reducing NSAID use, a class also linked with an increased risk for bleeding and other adverse effects, Dr. Yang said during the briefing. Minimizing use of NSAIDs including ibuprofen and naproxen use “is something to think about,” he suggested.
“The effect of NSAIDs on blood pressure is not well studied and can vary from person to person” noted Dr. Vitarello, who added that higher NSAID dosages and more prolonged use likely increase the risk for an adverse effect on blood pressure. One reasonable option is to encourage patients to use an alternative class of pain reliever such as acetaminophen.
It remains “a challenge” to discern differences in adverse blood pressure effects, and in all adverse cardiovascular effects among different NSAIDs, said Dr. Yang. Results from “some studies show that certain NSAIDs may be safer, but other studies did not. We need to be very careful using NSAIDs because, on average, they increase blood pressure by about 3 mm Hg. We need to be mindful to try to prescribe alternative agents, like acetaminophen.”
A decade of data from NHANES
The analysis run by Dr. Vitarello and associates used data from 27,599 American adults included in the NHANES during 2009-2018, and focused on the 44% who either had an average blood pressure measurement of at least 130/80 mm Hg or reported having ever been told by a clinician that they had hypertension. The NHANES assessments included the prescription medications taken by each participant. The prevalence of using at least one prescription drug known to raise blood pressure was 24% among women and 14% among men, and 4% of those with hypertension were on two or more pressure-increasing agents.
The researchers based their identification of pressure-increasing prescription drugs on the list included in the 2017 guideline for managing high blood pressure from the American College of Cardiology and American Heart Association. This list specifies that the antidepressants that raise blood pressure are the monoamine oxidase inhibitors, serotonin norepinephrine reuptake inhibitors, and tricyclic antidepressants.
Dr. Vitarello and Dr. Yang had no disclosures.
Nearly half of these American adults had hypertension, and in this subgroup, 18.5% reported using a prescription drug known to increase blood pressure. The most widely used class of agents with this effect was antidepressants, used by 8.7%; followed by nonsteroidal anti-inflammatory drugs (NSAIDs), used by 6.5%; steroids, 1.9%; estrogens, 1.7%; and several other agents each used by fewer than 1% of the study cohort, John Vitarello, MD, said during a press briefing on reports from the upcoming annual scientific sessions of the American College of Cardiology.
He and his associates estimated that this use of prescription drugs known to raise blood pressure could be what stands in the way of some 560,000-2.2 million Americans from having their hypertension under control, depending on the exact blood pressure impact that various pressure-increasing drugs have and presuming that half of those on blood pressure increasing agents could stop them and switch to alternative agents, said Dr. Vitarello, a researcher at Beth Israel Deaconess Medical Center in Boston.
He also highlighted that the study assessed only prescription drugs and did not examine OTC drug use, which may be especially relevant for the many people who regularly take NSAIDs.
“Clinicians should review the prescription and OTC drug use of patients with hypertension and consider stopping drugs that increase blood pressure or switching the patient to alternatives” that are blood pressure neutral, Dr. Vitarello said during the briefing. He cautioned that maintaining hypertensive patients on agents that raise their blood pressure can result in “prescribing cascades” where taking drugs that boost blood pressure results in need for intensified antihypertensive treatment.
An opportunity for NSAID alternatives
“This study hopefully raises awareness that there is a very high use of medications that increase blood pressure, and use of OTC agents could increase the rate even higher” said Eugene Yang, MD, a cardiologist and codirector of the Cardiovascular Wellness and Prevention Program of the University of Washington, Seattle. Substituting for certain antidepressant agents may often not be realistic, but an opportunity exists for reducing NSAID use, a class also linked with an increased risk for bleeding and other adverse effects, Dr. Yang said during the briefing. Minimizing use of NSAIDs including ibuprofen and naproxen use “is something to think about,” he suggested.
“The effect of NSAIDs on blood pressure is not well studied and can vary from person to person” noted Dr. Vitarello, who added that higher NSAID dosages and more prolonged use likely increase the risk for an adverse effect on blood pressure. One reasonable option is to encourage patients to use an alternative class of pain reliever such as acetaminophen.
It remains “a challenge” to discern differences in adverse blood pressure effects, and in all adverse cardiovascular effects among different NSAIDs, said Dr. Yang. Results from “some studies show that certain NSAIDs may be safer, but other studies did not. We need to be very careful using NSAIDs because, on average, they increase blood pressure by about 3 mm Hg. We need to be mindful to try to prescribe alternative agents, like acetaminophen.”
A decade of data from NHANES
The analysis run by Dr. Vitarello and associates used data from 27,599 American adults included in the NHANES during 2009-2018, and focused on the 44% who either had an average blood pressure measurement of at least 130/80 mm Hg or reported having ever been told by a clinician that they had hypertension. The NHANES assessments included the prescription medications taken by each participant. The prevalence of using at least one prescription drug known to raise blood pressure was 24% among women and 14% among men, and 4% of those with hypertension were on two or more pressure-increasing agents.
The researchers based their identification of pressure-increasing prescription drugs on the list included in the 2017 guideline for managing high blood pressure from the American College of Cardiology and American Heart Association. This list specifies that the antidepressants that raise blood pressure are the monoamine oxidase inhibitors, serotonin norepinephrine reuptake inhibitors, and tricyclic antidepressants.
Dr. Vitarello and Dr. Yang had no disclosures.
FROM ACC 2021
Patchy growth of TAVR programs leaves poorer communities behind
Inequities in the initial growth of transcatheter aortic valve replacement (TAVR) programs in American hospitals has led to less use of the transformative procedure in poorer communities, a new cross-sectional study suggests.
Using Medicare claims data, investigators identified 554 new TAVR programs created between January 2012 and December 2018.
Of these, 98% were established in metropolitan areas (>50,000 residents) and 53% were started in areas with preexisting TAVR programs, “thereby increasing the number of programs but not necessarily increasing the geographic availability of the procedure,” said study author Ashwin Nathan, MD, Hospital of the University of Pennsylvania, Philadelphia.
Only 11 programs were started in nonmetropolitan areas over the study period, he noted during the featured clinical research presentation at the Society for Cardiovascular Angiography and Interventions (SCAI) 2021 annual scientific sessions, held virtually this year.
Hospitals that established TAVR programs, compared with those that did not, cared for patients with higher median household incomes (difference, $1,305; P = .03) and from areas with better economic well-being based on the Distressed Communities Index (difference, –3.15 units; P < .01), and cared for fewer patients with dual eligibility for Medicaid (difference, –3.15%; P < .01).
When the investigators looked at rates of TAVR between the core-based statistical areas, there were fewer TAVR procedures per 100,000 Medicare beneficiaries in areas with more Medicaid dual-eligible patients (difference, –1.19% per 1% increase), lower average median household incomes (difference, –0.62% per $1,000 decrease), and more average community distress (difference, –0.35% per 1 unit increase; P < .01 for all).
“What we can conclude is that the increased number of TAVR programs that we found during the study period did not necessarily translate to increased access to TAVR ... Wealthy, more privileged patients had more access to TAVR by virtue of the hospitals that serve them,” Dr. Nathan said.
Future steps, he said, are to identify the role of race and ethnicity in inequitable access to TAVR, identify system- and patient-level barriers to access, and to develop and test solutions to address inequitable care.
Elaborating on the latter point during a discussion of the results, study coauthor Jay S. Giri, MD, MPH, also from the Hospital of the University of Pennsylvania, observed that although the data showed rural areas are left behind, not every part of an urban area acts like the area more generally.
As a result, they’re delving into the 25 largest urban areas and trying to disaggregate, based on both socioeconomic status and race within the area, whether inequities exist, he said. “Believe it or not, in some urban areas where there clearly is access – there might even be a dozen TAVR programs within a 25 mile radius – do some of those areas still act like rural areas that don’t have access? So more to come on that.”
Session comoderator Steven Yakubov, MD, MidWest Cardiology Research Foundation in Columbus, Ohio, said the results show TAVR programs tend to be developed in well-served areas but asked whether some of the responsibility falls on patients to seek medical attention. “Do we just not give enough education to patients on how to access care?”
Dr. Giri responded by highlighting the complexity of navigating from even being diagnosed with aortic stenosis to making it through a multidisciplinary TAVR evaluation.
“Individuals with increased health literacy and more means are more likely to make it through that gauntlet. But from a public health perspective, obviously, I’d argue that the onus is probably more on the medical community at large to figure out how to roll these programs out more widespread,” he said.
“It looked to us like market forces overwhelmingly seemed to drive the development of new TAVR programs over access to care considerations,” Dr. Giri added. “And just to point out, those market forces aren’t at the level of the device manufacturers, who are often maligned for cost. This is really about the market forces at the level of hospitals and health systems.”
Session comoderator Megan Coylewright, MD, MPH, Erlanger Heart and Lung Institute, Chattanooga, Tenn., said, “I think that’s really well stated,” and noted that physicians may bear some responsibility as well.
“From a physician responsibility, especially for structural heart, we tended to all aggregate together, all of us that have structural heart training or that have trained in certain institutions,” she said. “It’s certainly on us to continue to spread out and go to the communities in need to ensure access. I think, as Dr. Giri said, there are a lot of solutions and that needs to be the focus for the next couple of years.”
Dr. Nathan reported having no relevant disclosures. Dr. Giri reported serving as a principal investigator for a research study for Boston Scientific, Inari Medical, Abbott, and Recor Medical; consulting for Boston Scientific; and serving on an advisory board for Inari Medical.
A version of this article first appeared on Medscape.com.
Inequities in the initial growth of transcatheter aortic valve replacement (TAVR) programs in American hospitals has led to less use of the transformative procedure in poorer communities, a new cross-sectional study suggests.
Using Medicare claims data, investigators identified 554 new TAVR programs created between January 2012 and December 2018.
Of these, 98% were established in metropolitan areas (>50,000 residents) and 53% were started in areas with preexisting TAVR programs, “thereby increasing the number of programs but not necessarily increasing the geographic availability of the procedure,” said study author Ashwin Nathan, MD, Hospital of the University of Pennsylvania, Philadelphia.
Only 11 programs were started in nonmetropolitan areas over the study period, he noted during the featured clinical research presentation at the Society for Cardiovascular Angiography and Interventions (SCAI) 2021 annual scientific sessions, held virtually this year.
Hospitals that established TAVR programs, compared with those that did not, cared for patients with higher median household incomes (difference, $1,305; P = .03) and from areas with better economic well-being based on the Distressed Communities Index (difference, –3.15 units; P < .01), and cared for fewer patients with dual eligibility for Medicaid (difference, –3.15%; P < .01).
When the investigators looked at rates of TAVR between the core-based statistical areas, there were fewer TAVR procedures per 100,000 Medicare beneficiaries in areas with more Medicaid dual-eligible patients (difference, –1.19% per 1% increase), lower average median household incomes (difference, –0.62% per $1,000 decrease), and more average community distress (difference, –0.35% per 1 unit increase; P < .01 for all).
“What we can conclude is that the increased number of TAVR programs that we found during the study period did not necessarily translate to increased access to TAVR ... Wealthy, more privileged patients had more access to TAVR by virtue of the hospitals that serve them,” Dr. Nathan said.
Future steps, he said, are to identify the role of race and ethnicity in inequitable access to TAVR, identify system- and patient-level barriers to access, and to develop and test solutions to address inequitable care.
Elaborating on the latter point during a discussion of the results, study coauthor Jay S. Giri, MD, MPH, also from the Hospital of the University of Pennsylvania, observed that although the data showed rural areas are left behind, not every part of an urban area acts like the area more generally.
As a result, they’re delving into the 25 largest urban areas and trying to disaggregate, based on both socioeconomic status and race within the area, whether inequities exist, he said. “Believe it or not, in some urban areas where there clearly is access – there might even be a dozen TAVR programs within a 25 mile radius – do some of those areas still act like rural areas that don’t have access? So more to come on that.”
Session comoderator Steven Yakubov, MD, MidWest Cardiology Research Foundation in Columbus, Ohio, said the results show TAVR programs tend to be developed in well-served areas but asked whether some of the responsibility falls on patients to seek medical attention. “Do we just not give enough education to patients on how to access care?”
Dr. Giri responded by highlighting the complexity of navigating from even being diagnosed with aortic stenosis to making it through a multidisciplinary TAVR evaluation.
“Individuals with increased health literacy and more means are more likely to make it through that gauntlet. But from a public health perspective, obviously, I’d argue that the onus is probably more on the medical community at large to figure out how to roll these programs out more widespread,” he said.
“It looked to us like market forces overwhelmingly seemed to drive the development of new TAVR programs over access to care considerations,” Dr. Giri added. “And just to point out, those market forces aren’t at the level of the device manufacturers, who are often maligned for cost. This is really about the market forces at the level of hospitals and health systems.”
Session comoderator Megan Coylewright, MD, MPH, Erlanger Heart and Lung Institute, Chattanooga, Tenn., said, “I think that’s really well stated,” and noted that physicians may bear some responsibility as well.
“From a physician responsibility, especially for structural heart, we tended to all aggregate together, all of us that have structural heart training or that have trained in certain institutions,” she said. “It’s certainly on us to continue to spread out and go to the communities in need to ensure access. I think, as Dr. Giri said, there are a lot of solutions and that needs to be the focus for the next couple of years.”
Dr. Nathan reported having no relevant disclosures. Dr. Giri reported serving as a principal investigator for a research study for Boston Scientific, Inari Medical, Abbott, and Recor Medical; consulting for Boston Scientific; and serving on an advisory board for Inari Medical.
A version of this article first appeared on Medscape.com.
Inequities in the initial growth of transcatheter aortic valve replacement (TAVR) programs in American hospitals has led to less use of the transformative procedure in poorer communities, a new cross-sectional study suggests.
Using Medicare claims data, investigators identified 554 new TAVR programs created between January 2012 and December 2018.
Of these, 98% were established in metropolitan areas (>50,000 residents) and 53% were started in areas with preexisting TAVR programs, “thereby increasing the number of programs but not necessarily increasing the geographic availability of the procedure,” said study author Ashwin Nathan, MD, Hospital of the University of Pennsylvania, Philadelphia.
Only 11 programs were started in nonmetropolitan areas over the study period, he noted during the featured clinical research presentation at the Society for Cardiovascular Angiography and Interventions (SCAI) 2021 annual scientific sessions, held virtually this year.
Hospitals that established TAVR programs, compared with those that did not, cared for patients with higher median household incomes (difference, $1,305; P = .03) and from areas with better economic well-being based on the Distressed Communities Index (difference, –3.15 units; P < .01), and cared for fewer patients with dual eligibility for Medicaid (difference, –3.15%; P < .01).
When the investigators looked at rates of TAVR between the core-based statistical areas, there were fewer TAVR procedures per 100,000 Medicare beneficiaries in areas with more Medicaid dual-eligible patients (difference, –1.19% per 1% increase), lower average median household incomes (difference, –0.62% per $1,000 decrease), and more average community distress (difference, –0.35% per 1 unit increase; P < .01 for all).
“What we can conclude is that the increased number of TAVR programs that we found during the study period did not necessarily translate to increased access to TAVR ... Wealthy, more privileged patients had more access to TAVR by virtue of the hospitals that serve them,” Dr. Nathan said.
Future steps, he said, are to identify the role of race and ethnicity in inequitable access to TAVR, identify system- and patient-level barriers to access, and to develop and test solutions to address inequitable care.
Elaborating on the latter point during a discussion of the results, study coauthor Jay S. Giri, MD, MPH, also from the Hospital of the University of Pennsylvania, observed that although the data showed rural areas are left behind, not every part of an urban area acts like the area more generally.
As a result, they’re delving into the 25 largest urban areas and trying to disaggregate, based on both socioeconomic status and race within the area, whether inequities exist, he said. “Believe it or not, in some urban areas where there clearly is access – there might even be a dozen TAVR programs within a 25 mile radius – do some of those areas still act like rural areas that don’t have access? So more to come on that.”
Session comoderator Steven Yakubov, MD, MidWest Cardiology Research Foundation in Columbus, Ohio, said the results show TAVR programs tend to be developed in well-served areas but asked whether some of the responsibility falls on patients to seek medical attention. “Do we just not give enough education to patients on how to access care?”
Dr. Giri responded by highlighting the complexity of navigating from even being diagnosed with aortic stenosis to making it through a multidisciplinary TAVR evaluation.
“Individuals with increased health literacy and more means are more likely to make it through that gauntlet. But from a public health perspective, obviously, I’d argue that the onus is probably more on the medical community at large to figure out how to roll these programs out more widespread,” he said.
“It looked to us like market forces overwhelmingly seemed to drive the development of new TAVR programs over access to care considerations,” Dr. Giri added. “And just to point out, those market forces aren’t at the level of the device manufacturers, who are often maligned for cost. This is really about the market forces at the level of hospitals and health systems.”
Session comoderator Megan Coylewright, MD, MPH, Erlanger Heart and Lung Institute, Chattanooga, Tenn., said, “I think that’s really well stated,” and noted that physicians may bear some responsibility as well.
“From a physician responsibility, especially for structural heart, we tended to all aggregate together, all of us that have structural heart training or that have trained in certain institutions,” she said. “It’s certainly on us to continue to spread out and go to the communities in need to ensure access. I think, as Dr. Giri said, there are a lot of solutions and that needs to be the focus for the next couple of years.”
Dr. Nathan reported having no relevant disclosures. Dr. Giri reported serving as a principal investigator for a research study for Boston Scientific, Inari Medical, Abbott, and Recor Medical; consulting for Boston Scientific; and serving on an advisory board for Inari Medical.
A version of this article first appeared on Medscape.com.
AHA statement flags CV risk of hormonal cancer therapies
Hormonal therapies for the treatment of hormone-dependent breast and prostate cancer could raise the risk for myocardial infarction and stroke, and patients need to be closely monitored to allow early detection and treatment of cardiovascular disease (CVD), the American Heart Association says in a new scientific statement.
“The statement provides data on the risks of each type of hormonal therapy so clinicians can use it as a guide to help manage cardiovascular risks during cancer treatment,” Tochi Okwuosa, DO, chair of the writing group, said in a news release.
“A team-based approach to patient care that includes the oncology team, cardiologist, primary care clinician, dietitian, endocrinologist, and other health care professionals as appropriate is needed to work with each patient to manage and reduce the increased risk of heart disease and strokes associated with hormonal therapy in breast and prostate cancer treatment,” said Dr. Okwuosa, director of cardio-oncology services, Rush University Medical Center, Chicago.
The scientific statement was published online April 26 in Circulation: Genomic and Precision Medicine.
Hormone-dependent cancers, such as prostate and breast cancer, are the most common noncutaneous cancers in the United States and around the world. As hormonal therapies have markedly improved survival in these patients, CVD has emerged as a leading cause illness and death.
The increased CVD burden might be explained by the increasing average age of cancer survivors, leading to higher rates of age-related CV risk factors and coronary artery disease.
The writing group reviewed existing evidence from observational studies and randomized controlled trials on the cardiovascular impact of anticancer hormonal therapies.
Among the key findings:
- In patients with breast cancer, has been shown to increase the risk for venous thromboembolic events, but to have somewhat protective to neutral effects on CVD risk burden and CVD events. Conversely, aromatase inhibitors have been shown to increase the risk for CVD risk factors and events, including MI and stroke.
- Androgen-deprivation therapy for prostate cancer appears to increase the risk for CV events, although gonadotrophin-releasing hormone (GnRH) antagonists are associated with a lower risk for CV events than are GnRH agonists. The oral antiandrogens appear to be associated with increased CVD risk as well, particularly when used for complete androgen blockade as combination GnRH/anti-androgen therapy.
- The duration of hormonal therapies has a significant impact on CVD risk; the longer patients receive hormonal therapy, the greater the risk. More research is needed to better define the risks associated with duration of treatment.
- The data are mixed on the impact of preexisting CV risk factors and CVD on CV events associated with hormonal therapy. Although the presence of baseline CV risk factors and CVD can increase CV events associated with aromatase inhibitors, it is not clear that tamoxifen does.
- Studies suggest that patients with prostate cancer and baseline CVD and CV risk factors have increased rates of CV events when treated with androgen-deprivation therapy.
- Although the prolonged use of some hormonal therapies worsens CV risk factors and , the effects of the duration of therapy on CV events are less clear.
The writing group noted that there are no definitive guidelines for the monitoring and management of hormonal therapy-related CVD risks.
The authors encourage clinicians to be alert for worsening CV problems in those with preexisting heart disease or risk factors, and to recognize that even patients without preexisting CV problems are at higher risk because of their exposure to hormonal therapies.
“For patients who have two or more cardiovascular risk factors, it is likely that referral to a cardiologist would be appropriate prior to beginning hormone treatment. For patients already receiving hormonal therapies, a discussion with the oncology team can help to determine if a cardiology referral is recommended,” Dr. Okwuosa said in the news release.
This scientific statement was prepared by the volunteer writing group on behalf of the AHA Cardio-Oncology Subcommittee of the Council on Clinical Cardiology and the Council on Genomic and Precision Medicine; the Council on Arteriosclerosis, Thrombosis, and Vascular Biology; and the Council on Cardiovascular Radiology and Intervention.
The research had no commercial funding. Dr. Okwuosa has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Hormonal therapies for the treatment of hormone-dependent breast and prostate cancer could raise the risk for myocardial infarction and stroke, and patients need to be closely monitored to allow early detection and treatment of cardiovascular disease (CVD), the American Heart Association says in a new scientific statement.
“The statement provides data on the risks of each type of hormonal therapy so clinicians can use it as a guide to help manage cardiovascular risks during cancer treatment,” Tochi Okwuosa, DO, chair of the writing group, said in a news release.
“A team-based approach to patient care that includes the oncology team, cardiologist, primary care clinician, dietitian, endocrinologist, and other health care professionals as appropriate is needed to work with each patient to manage and reduce the increased risk of heart disease and strokes associated with hormonal therapy in breast and prostate cancer treatment,” said Dr. Okwuosa, director of cardio-oncology services, Rush University Medical Center, Chicago.
The scientific statement was published online April 26 in Circulation: Genomic and Precision Medicine.
Hormone-dependent cancers, such as prostate and breast cancer, are the most common noncutaneous cancers in the United States and around the world. As hormonal therapies have markedly improved survival in these patients, CVD has emerged as a leading cause illness and death.
The increased CVD burden might be explained by the increasing average age of cancer survivors, leading to higher rates of age-related CV risk factors and coronary artery disease.
The writing group reviewed existing evidence from observational studies and randomized controlled trials on the cardiovascular impact of anticancer hormonal therapies.
Among the key findings:
- In patients with breast cancer, has been shown to increase the risk for venous thromboembolic events, but to have somewhat protective to neutral effects on CVD risk burden and CVD events. Conversely, aromatase inhibitors have been shown to increase the risk for CVD risk factors and events, including MI and stroke.
- Androgen-deprivation therapy for prostate cancer appears to increase the risk for CV events, although gonadotrophin-releasing hormone (GnRH) antagonists are associated with a lower risk for CV events than are GnRH agonists. The oral antiandrogens appear to be associated with increased CVD risk as well, particularly when used for complete androgen blockade as combination GnRH/anti-androgen therapy.
- The duration of hormonal therapies has a significant impact on CVD risk; the longer patients receive hormonal therapy, the greater the risk. More research is needed to better define the risks associated with duration of treatment.
- The data are mixed on the impact of preexisting CV risk factors and CVD on CV events associated with hormonal therapy. Although the presence of baseline CV risk factors and CVD can increase CV events associated with aromatase inhibitors, it is not clear that tamoxifen does.
- Studies suggest that patients with prostate cancer and baseline CVD and CV risk factors have increased rates of CV events when treated with androgen-deprivation therapy.
- Although the prolonged use of some hormonal therapies worsens CV risk factors and , the effects of the duration of therapy on CV events are less clear.
The writing group noted that there are no definitive guidelines for the monitoring and management of hormonal therapy-related CVD risks.
The authors encourage clinicians to be alert for worsening CV problems in those with preexisting heart disease or risk factors, and to recognize that even patients without preexisting CV problems are at higher risk because of their exposure to hormonal therapies.
“For patients who have two or more cardiovascular risk factors, it is likely that referral to a cardiologist would be appropriate prior to beginning hormone treatment. For patients already receiving hormonal therapies, a discussion with the oncology team can help to determine if a cardiology referral is recommended,” Dr. Okwuosa said in the news release.
This scientific statement was prepared by the volunteer writing group on behalf of the AHA Cardio-Oncology Subcommittee of the Council on Clinical Cardiology and the Council on Genomic and Precision Medicine; the Council on Arteriosclerosis, Thrombosis, and Vascular Biology; and the Council on Cardiovascular Radiology and Intervention.
The research had no commercial funding. Dr. Okwuosa has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Hormonal therapies for the treatment of hormone-dependent breast and prostate cancer could raise the risk for myocardial infarction and stroke, and patients need to be closely monitored to allow early detection and treatment of cardiovascular disease (CVD), the American Heart Association says in a new scientific statement.
“The statement provides data on the risks of each type of hormonal therapy so clinicians can use it as a guide to help manage cardiovascular risks during cancer treatment,” Tochi Okwuosa, DO, chair of the writing group, said in a news release.
“A team-based approach to patient care that includes the oncology team, cardiologist, primary care clinician, dietitian, endocrinologist, and other health care professionals as appropriate is needed to work with each patient to manage and reduce the increased risk of heart disease and strokes associated with hormonal therapy in breast and prostate cancer treatment,” said Dr. Okwuosa, director of cardio-oncology services, Rush University Medical Center, Chicago.
The scientific statement was published online April 26 in Circulation: Genomic and Precision Medicine.
Hormone-dependent cancers, such as prostate and breast cancer, are the most common noncutaneous cancers in the United States and around the world. As hormonal therapies have markedly improved survival in these patients, CVD has emerged as a leading cause illness and death.
The increased CVD burden might be explained by the increasing average age of cancer survivors, leading to higher rates of age-related CV risk factors and coronary artery disease.
The writing group reviewed existing evidence from observational studies and randomized controlled trials on the cardiovascular impact of anticancer hormonal therapies.
Among the key findings:
- In patients with breast cancer, has been shown to increase the risk for venous thromboembolic events, but to have somewhat protective to neutral effects on CVD risk burden and CVD events. Conversely, aromatase inhibitors have been shown to increase the risk for CVD risk factors and events, including MI and stroke.
- Androgen-deprivation therapy for prostate cancer appears to increase the risk for CV events, although gonadotrophin-releasing hormone (GnRH) antagonists are associated with a lower risk for CV events than are GnRH agonists. The oral antiandrogens appear to be associated with increased CVD risk as well, particularly when used for complete androgen blockade as combination GnRH/anti-androgen therapy.
- The duration of hormonal therapies has a significant impact on CVD risk; the longer patients receive hormonal therapy, the greater the risk. More research is needed to better define the risks associated with duration of treatment.
- The data are mixed on the impact of preexisting CV risk factors and CVD on CV events associated with hormonal therapy. Although the presence of baseline CV risk factors and CVD can increase CV events associated with aromatase inhibitors, it is not clear that tamoxifen does.
- Studies suggest that patients with prostate cancer and baseline CVD and CV risk factors have increased rates of CV events when treated with androgen-deprivation therapy.
- Although the prolonged use of some hormonal therapies worsens CV risk factors and , the effects of the duration of therapy on CV events are less clear.
The writing group noted that there are no definitive guidelines for the monitoring and management of hormonal therapy-related CVD risks.
The authors encourage clinicians to be alert for worsening CV problems in those with preexisting heart disease or risk factors, and to recognize that even patients without preexisting CV problems are at higher risk because of their exposure to hormonal therapies.
“For patients who have two or more cardiovascular risk factors, it is likely that referral to a cardiologist would be appropriate prior to beginning hormone treatment. For patients already receiving hormonal therapies, a discussion with the oncology team can help to determine if a cardiology referral is recommended,” Dr. Okwuosa said in the news release.
This scientific statement was prepared by the volunteer writing group on behalf of the AHA Cardio-Oncology Subcommittee of the Council on Clinical Cardiology and the Council on Genomic and Precision Medicine; the Council on Arteriosclerosis, Thrombosis, and Vascular Biology; and the Council on Cardiovascular Radiology and Intervention.
The research had no commercial funding. Dr. Okwuosa has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Post–COVID-19 cardiac involvement in college athletes much rarer than thought
In a multicenter study conducted during September-December 2020, only 0.7% of 3,018 collegiate athletes who tested positive for SARS-CoV-2 infection were found to have definite, probable, or possible infection-related cardiac involvement.
None experienced an adverse cardiac event and only five (0.2%) required hospitalization for noncardiac complications of COVID-19.
“The take-home message is that cardiac involvement does not happen as much as we had initially feared. It’s in the range of 0.5% to 3%, depending on how you define cardiac involvement, which is not nothing, but it’s not the 30% or 50% that some early studies hinted at,” said Kimberly G. Harmon, MD, of the University of Washington, Seattle.
Dr. Harmon, along with Jeffrey A. Drezner, MD, also from UW, and Aaron L. Baggish, MD, of Massachusetts General Hospital, Boston, were co–primary investigators of the Outcomes Registry for Cardiac Conditions in Athletes (ORCCA) study. The group’s findings were published April 17 in Circulation.
Nearly 20,000 athletes tested
The researchers prospectively tested 19,378 athletes for SARS-CoV-2 infection from 42 U.S. colleges and universities during the study period. A total of 3,018 (16%; mean age, 20 years; 32% female) tested positive and underwent cardiac evaluation.
“We didn’t prescribe what the schools had to do in terms of cardiac evaluation, but most of these colleges are well resourced, and about 74% of athletes were evaluated using the triad testing strategy of 12-lead electrocardiography, cardiac troponin, and transthoracic echocardiography [TEE], with cardiac magnetic resonance [CMR ]when indicated,” explained Dr. Harmon. Only 198 athletes underwent primary screening with CMR.
Athletes were often tested multiple times for SARS-CoV-2 infection by participating institutions and were included in this study if they had any positive test and underwent postinfection cardiac screening.
The cohort includes athletes representing 26 distinct sporting disciplines, including American-style football (36%), basketball (9%), and cross country/track and field (8%). Most were asymptomatic or had only mild COVID-19 symptoms (33% and 29%, respectively).
‘Exercise appears to be protective’
Abnormal findings suggestive of SARS-CoV-2 cardiac involvement were detected by ECG in 0.7% of athletes (21 of 2,999), cardiac troponin elevation in 0.9% (24/2,719), and abnormal TTE findings in 0.9% (24/2,556).
The odds of having cardiac involvement was 3.1 times higher in athletes with cardiopulmonary symptoms.
“One thing we’ve seen in the literature and in this cohort, is that exercise appears to be protective to some extent from COVID-19. We had a lot of cases, but in the whole cohort, only five athletes were hospitalized with COVID and those were for noncardiac reasons,” said Dr. Harmon.
During a median clinical surveillance of 113 days, there was one (0.03%) adverse cardiac event likely unrelated to SARS-CoV-2 infection.
The diagnostic yield for probable or definite cardiac involvement was 6.7 times higher for a CMR obtained for clinical reasons (10.1%) versus a primary screening CMR (1.5%).
“This is data we desperately needed. Small, single-center studies early in the pandemic had indicated a higher prevalence of cardiac involvement, which led us to be very conservative about return-to-play in the early days,” said Jeffrey Lander, MD, who was not involved in the study.
The study is complementary, he noted, to one published in March that looked at professional athletes post–COVID-19 and also found cardiac pathology in fewer than 1%. The mean age in that study was 25 years.
“They saw a similarly low rate of cardiac involvement in professional athletes, and together with this study, it gives us new information that is also reassuring,” added Dr. Lander, codirector of sports cardiology at Saint Barnabas Medical Center in Livingston, N.J., an RWJBarnabas Health facility, and team cardiologist for Seton Hall University in South Orange, N.J.
Limit CMR to symptomatic athletes
“I think this data can be extended beyond the college athlete. And it’s fair to say to high school athletes and young recreational athletes who have had asymptomatic or mild infection, you probably don’t need further workup if you’re feeling fine,” suggested Dr. Harmon.
“For those with moderate or severe illness, then the triple screen protocol is a good idea, particularly if they are having any symptoms,” she added.
Dr. Lander agrees that athletes should be screened by appropriate providers before returning to sports, but that CMR should not be used routinely for return-to-play screening.
“We’ve never taken a group of, say, 1,000 college athletes who just recovered from the flu and done cardiac MRIs on them, so it’s a bit like opening Pandora’s box when it’s used too liberally. It’s difficult to assess if the findings are secondary to COVID infection or from something entirely unrelated,” he noted.
ORCCA is a collaboration of the American Heart Association and the American Medical Society for Sports Medicine to track COVID-19 cases among National Collegiate Athletic Association (NCAA) athletes. The current study was supported by a grant from the American Medical Society for Sports Medicine.
In a multicenter study conducted during September-December 2020, only 0.7% of 3,018 collegiate athletes who tested positive for SARS-CoV-2 infection were found to have definite, probable, or possible infection-related cardiac involvement.
None experienced an adverse cardiac event and only five (0.2%) required hospitalization for noncardiac complications of COVID-19.
“The take-home message is that cardiac involvement does not happen as much as we had initially feared. It’s in the range of 0.5% to 3%, depending on how you define cardiac involvement, which is not nothing, but it’s not the 30% or 50% that some early studies hinted at,” said Kimberly G. Harmon, MD, of the University of Washington, Seattle.
Dr. Harmon, along with Jeffrey A. Drezner, MD, also from UW, and Aaron L. Baggish, MD, of Massachusetts General Hospital, Boston, were co–primary investigators of the Outcomes Registry for Cardiac Conditions in Athletes (ORCCA) study. The group’s findings were published April 17 in Circulation.
Nearly 20,000 athletes tested
The researchers prospectively tested 19,378 athletes for SARS-CoV-2 infection from 42 U.S. colleges and universities during the study period. A total of 3,018 (16%; mean age, 20 years; 32% female) tested positive and underwent cardiac evaluation.
“We didn’t prescribe what the schools had to do in terms of cardiac evaluation, but most of these colleges are well resourced, and about 74% of athletes were evaluated using the triad testing strategy of 12-lead electrocardiography, cardiac troponin, and transthoracic echocardiography [TEE], with cardiac magnetic resonance [CMR ]when indicated,” explained Dr. Harmon. Only 198 athletes underwent primary screening with CMR.
Athletes were often tested multiple times for SARS-CoV-2 infection by participating institutions and were included in this study if they had any positive test and underwent postinfection cardiac screening.
The cohort includes athletes representing 26 distinct sporting disciplines, including American-style football (36%), basketball (9%), and cross country/track and field (8%). Most were asymptomatic or had only mild COVID-19 symptoms (33% and 29%, respectively).
‘Exercise appears to be protective’
Abnormal findings suggestive of SARS-CoV-2 cardiac involvement were detected by ECG in 0.7% of athletes (21 of 2,999), cardiac troponin elevation in 0.9% (24/2,719), and abnormal TTE findings in 0.9% (24/2,556).
The odds of having cardiac involvement was 3.1 times higher in athletes with cardiopulmonary symptoms.
“One thing we’ve seen in the literature and in this cohort, is that exercise appears to be protective to some extent from COVID-19. We had a lot of cases, but in the whole cohort, only five athletes were hospitalized with COVID and those were for noncardiac reasons,” said Dr. Harmon.
During a median clinical surveillance of 113 days, there was one (0.03%) adverse cardiac event likely unrelated to SARS-CoV-2 infection.
The diagnostic yield for probable or definite cardiac involvement was 6.7 times higher for a CMR obtained for clinical reasons (10.1%) versus a primary screening CMR (1.5%).
“This is data we desperately needed. Small, single-center studies early in the pandemic had indicated a higher prevalence of cardiac involvement, which led us to be very conservative about return-to-play in the early days,” said Jeffrey Lander, MD, who was not involved in the study.
The study is complementary, he noted, to one published in March that looked at professional athletes post–COVID-19 and also found cardiac pathology in fewer than 1%. The mean age in that study was 25 years.
“They saw a similarly low rate of cardiac involvement in professional athletes, and together with this study, it gives us new information that is also reassuring,” added Dr. Lander, codirector of sports cardiology at Saint Barnabas Medical Center in Livingston, N.J., an RWJBarnabas Health facility, and team cardiologist for Seton Hall University in South Orange, N.J.
Limit CMR to symptomatic athletes
“I think this data can be extended beyond the college athlete. And it’s fair to say to high school athletes and young recreational athletes who have had asymptomatic or mild infection, you probably don’t need further workup if you’re feeling fine,” suggested Dr. Harmon.
“For those with moderate or severe illness, then the triple screen protocol is a good idea, particularly if they are having any symptoms,” she added.
Dr. Lander agrees that athletes should be screened by appropriate providers before returning to sports, but that CMR should not be used routinely for return-to-play screening.
“We’ve never taken a group of, say, 1,000 college athletes who just recovered from the flu and done cardiac MRIs on them, so it’s a bit like opening Pandora’s box when it’s used too liberally. It’s difficult to assess if the findings are secondary to COVID infection or from something entirely unrelated,” he noted.
ORCCA is a collaboration of the American Heart Association and the American Medical Society for Sports Medicine to track COVID-19 cases among National Collegiate Athletic Association (NCAA) athletes. The current study was supported by a grant from the American Medical Society for Sports Medicine.
In a multicenter study conducted during September-December 2020, only 0.7% of 3,018 collegiate athletes who tested positive for SARS-CoV-2 infection were found to have definite, probable, or possible infection-related cardiac involvement.
None experienced an adverse cardiac event and only five (0.2%) required hospitalization for noncardiac complications of COVID-19.
“The take-home message is that cardiac involvement does not happen as much as we had initially feared. It’s in the range of 0.5% to 3%, depending on how you define cardiac involvement, which is not nothing, but it’s not the 30% or 50% that some early studies hinted at,” said Kimberly G. Harmon, MD, of the University of Washington, Seattle.
Dr. Harmon, along with Jeffrey A. Drezner, MD, also from UW, and Aaron L. Baggish, MD, of Massachusetts General Hospital, Boston, were co–primary investigators of the Outcomes Registry for Cardiac Conditions in Athletes (ORCCA) study. The group’s findings were published April 17 in Circulation.
Nearly 20,000 athletes tested
The researchers prospectively tested 19,378 athletes for SARS-CoV-2 infection from 42 U.S. colleges and universities during the study period. A total of 3,018 (16%; mean age, 20 years; 32% female) tested positive and underwent cardiac evaluation.
“We didn’t prescribe what the schools had to do in terms of cardiac evaluation, but most of these colleges are well resourced, and about 74% of athletes were evaluated using the triad testing strategy of 12-lead electrocardiography, cardiac troponin, and transthoracic echocardiography [TEE], with cardiac magnetic resonance [CMR ]when indicated,” explained Dr. Harmon. Only 198 athletes underwent primary screening with CMR.
Athletes were often tested multiple times for SARS-CoV-2 infection by participating institutions and were included in this study if they had any positive test and underwent postinfection cardiac screening.
The cohort includes athletes representing 26 distinct sporting disciplines, including American-style football (36%), basketball (9%), and cross country/track and field (8%). Most were asymptomatic or had only mild COVID-19 symptoms (33% and 29%, respectively).
‘Exercise appears to be protective’
Abnormal findings suggestive of SARS-CoV-2 cardiac involvement were detected by ECG in 0.7% of athletes (21 of 2,999), cardiac troponin elevation in 0.9% (24/2,719), and abnormal TTE findings in 0.9% (24/2,556).
The odds of having cardiac involvement was 3.1 times higher in athletes with cardiopulmonary symptoms.
“One thing we’ve seen in the literature and in this cohort, is that exercise appears to be protective to some extent from COVID-19. We had a lot of cases, but in the whole cohort, only five athletes were hospitalized with COVID and those were for noncardiac reasons,” said Dr. Harmon.
During a median clinical surveillance of 113 days, there was one (0.03%) adverse cardiac event likely unrelated to SARS-CoV-2 infection.
The diagnostic yield for probable or definite cardiac involvement was 6.7 times higher for a CMR obtained for clinical reasons (10.1%) versus a primary screening CMR (1.5%).
“This is data we desperately needed. Small, single-center studies early in the pandemic had indicated a higher prevalence of cardiac involvement, which led us to be very conservative about return-to-play in the early days,” said Jeffrey Lander, MD, who was not involved in the study.
The study is complementary, he noted, to one published in March that looked at professional athletes post–COVID-19 and also found cardiac pathology in fewer than 1%. The mean age in that study was 25 years.
“They saw a similarly low rate of cardiac involvement in professional athletes, and together with this study, it gives us new information that is also reassuring,” added Dr. Lander, codirector of sports cardiology at Saint Barnabas Medical Center in Livingston, N.J., an RWJBarnabas Health facility, and team cardiologist for Seton Hall University in South Orange, N.J.
Limit CMR to symptomatic athletes
“I think this data can be extended beyond the college athlete. And it’s fair to say to high school athletes and young recreational athletes who have had asymptomatic or mild infection, you probably don’t need further workup if you’re feeling fine,” suggested Dr. Harmon.
“For those with moderate or severe illness, then the triple screen protocol is a good idea, particularly if they are having any symptoms,” she added.
Dr. Lander agrees that athletes should be screened by appropriate providers before returning to sports, but that CMR should not be used routinely for return-to-play screening.
“We’ve never taken a group of, say, 1,000 college athletes who just recovered from the flu and done cardiac MRIs on them, so it’s a bit like opening Pandora’s box when it’s used too liberally. It’s difficult to assess if the findings are secondary to COVID infection or from something entirely unrelated,” he noted.
ORCCA is a collaboration of the American Heart Association and the American Medical Society for Sports Medicine to track COVID-19 cases among National Collegiate Athletic Association (NCAA) athletes. The current study was supported by a grant from the American Medical Society for Sports Medicine.
FROM CIRCULATION






