User login
COVID plus MI confers poor prognosis; 1 in 3 die in hospital
COVID-19 patients with ST-segment elevation MI (STEMI) represent a population with unique demographic and clinical features resulting in a high risk for mortality, according to initial findings from the North American Cardiovascular COVID-19 Myocardial Infarction (NACMI) Registry.
“This is the largest registry of COVID-positive patients presenting with STEMI [and] the results clearly illustrate the challenges and uniqueness of this patient population that deserves prompt and special attention,” study cochair Timothy Henry, MD, president-elect of the Society for Cardiovascular Angiography & Interventions, said in a news release.
The NACMI registry is a collaborative effort between the SCAI, the American College of Cardiology Interventional Council, and the Canadian Association of Interventional Cardiology.
“The rapid development of this ongoing, critically important prospective registry reflects the strong and unique collaboration of all three societies. It was gratifying to be part of this process and hopefully the results will improve the care of our patients and stimulate further research,” Dr. Henry said in the news release.
The registry has enrolled 1,185 patients presenting with STEMI at 64 sites across the United States and Canada. Participants include 230 COVID-positive STEMI patients; 495 STEMI patients suspected but ultimately confirmed not to have COVID-19; and 460 age-and sex-matched control STEMI patients treated prior to the pandemic who are part of the Midwest STEMI Consortium.
The initial findings from the registry were published online in the Journal of the American College of Cardiology.
Atypical symptoms may explain high death rate
The primary outcome – a composite of in-hospital death, stroke, recurrent MI, or repeat unplanned revascularization – occurred in 36% of COVID-positive patients, compared with 13% of COVID-negative patients and 5% of control patients (P < .001 relative to controls).
This difference was driven largely by a “very high” in-hospital death rate in COVID-positive patients, lead author Santiago Garcia, MD, Minneapolis Heart Institute Foundation, said in an interview.
The in-hospital death rate was 33% in COVID-positive patients, compared with 11% in COVID-negative patients and 4% in controls. Stroke also occurred more often in COVID-positive patients at 3% versus 2% in COVID-negative and 0% in controls.
These initial findings suggest that the combination of STEMI and COVID-19 infection “confers a poor prognosis, with one in three patients succumbing to the disease, even among patients selected for invasive angiography (28% mortality),” the investigators wrote.
The data also show that STEMI in COVID-positive patients disproportionately affects ethnic minorities (23% Hispanic and 24% Black) with diabetes, which was present in 46% of COVID-positive patients.
COVID-positive patients with STEMI are more likely to present with atypical symptoms such as dyspnea (54%), pulmonary infiltrates on chest x-ray (46%), and high-risk conditions such as cardiogenic shock (18%), “which may explain the high fatality rate,” Dr. Garcia said.
Despite these high-risk features, COVID-positive patients are less apt to undergo invasive angiography when compared with COVID-negative and control STEMI patients (78% vs. 96% vs. 100%).
The majority of patients (71%) who did under angiography received primary percutaneous coronary intervention (PPCI) with very small treatment delays (at 15 minutes) during the pandemic.
Another notable finding is that “many patients (23%) have ‘no culprit’ vessel and may represent different etiologies of ST-segment elevation including microemboli, myocarditis, Takotsubo cardiomyopathy,” Dr. Garcia said in an interview.
“In line with current guidelines, patients with suspected STEMI should be managed with PPCI, without delay while the safety of health care providers is ensured,” Ran Kornowski, MD, and Katia Orvin, MD, both with Rabin Medical Center, Petah Tikva, Israel, and Tel Aviv University, wrote in a linked editorial.
“In this case, PPCI should be performed routinely, even if the patient is presumed to have COVID-19, because PPCI should not be postponed. Confirmation of SARS-CoV-2 infection should not delay urgent decision management concerning reperfusion strategy,” they advised.
Looking ahead, Garcia said plans for the registry include determining predictors of in-hospital mortality and studying demographic and treatment trends as the pandemic continues with more virulent strains of the virus.
Various subgroup analyses are also planned as well as an independent angiographic and electrocardiographic core lab analysis. A comparative analysis of data from the US and Canada is also planned.
This work was supported by an ACC Accreditation Grant, Saskatchewan Health Research Foundation, and grants from Medtronic and Abbott Vascular to SCAI. Dr. Garcia has received institutional research grants from Edwards Lifesciences, BSCI, Medtronic, and Abbott Vascular; has served as a consultant for Medtronic and BSCI; and has served as a proctor for Edwards Lifesciences. Dr. Kornowski and Dr. Orvin disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID-19 patients with ST-segment elevation MI (STEMI) represent a population with unique demographic and clinical features resulting in a high risk for mortality, according to initial findings from the North American Cardiovascular COVID-19 Myocardial Infarction (NACMI) Registry.

“This is the largest registry of COVID-positive patients presenting with STEMI [and] the results clearly illustrate the challenges and uniqueness of this patient population that deserves prompt and special attention,” study cochair Timothy Henry, MD, president-elect of the Society for Cardiovascular Angiography & Interventions, said in a news release.
The NACMI registry is a collaborative effort between the SCAI, the American College of Cardiology Interventional Council, and the Canadian Association of Interventional Cardiology.
“The rapid development of this ongoing, critically important prospective registry reflects the strong and unique collaboration of all three societies. It was gratifying to be part of this process and hopefully the results will improve the care of our patients and stimulate further research,” Dr. Henry said in the news release.
The registry has enrolled 1,185 patients presenting with STEMI at 64 sites across the United States and Canada. Participants include 230 COVID-positive STEMI patients; 495 STEMI patients suspected but ultimately confirmed not to have COVID-19; and 460 age-and sex-matched control STEMI patients treated prior to the pandemic who are part of the Midwest STEMI Consortium.
The initial findings from the registry were published online in the Journal of the American College of Cardiology.
Atypical symptoms may explain high death rate
The primary outcome – a composite of in-hospital death, stroke, recurrent MI, or repeat unplanned revascularization – occurred in 36% of COVID-positive patients, compared with 13% of COVID-negative patients and 5% of control patients (P < .001 relative to controls).
This difference was driven largely by a “very high” in-hospital death rate in COVID-positive patients, lead author Santiago Garcia, MD, Minneapolis Heart Institute Foundation, said in an interview.
The in-hospital death rate was 33% in COVID-positive patients, compared with 11% in COVID-negative patients and 4% in controls. Stroke also occurred more often in COVID-positive patients at 3% versus 2% in COVID-negative and 0% in controls.
These initial findings suggest that the combination of STEMI and COVID-19 infection “confers a poor prognosis, with one in three patients succumbing to the disease, even among patients selected for invasive angiography (28% mortality),” the investigators wrote.
The data also show that STEMI in COVID-positive patients disproportionately affects ethnic minorities (23% Hispanic and 24% Black) with diabetes, which was present in 46% of COVID-positive patients.
COVID-positive patients with STEMI are more likely to present with atypical symptoms such as dyspnea (54%), pulmonary infiltrates on chest x-ray (46%), and high-risk conditions such as cardiogenic shock (18%), “which may explain the high fatality rate,” Dr. Garcia said.
Despite these high-risk features, COVID-positive patients are less apt to undergo invasive angiography when compared with COVID-negative and control STEMI patients (78% vs. 96% vs. 100%).
The majority of patients (71%) who did under angiography received primary percutaneous coronary intervention (PPCI) with very small treatment delays (at 15 minutes) during the pandemic.
Another notable finding is that “many patients (23%) have ‘no culprit’ vessel and may represent different etiologies of ST-segment elevation including microemboli, myocarditis, Takotsubo cardiomyopathy,” Dr. Garcia said in an interview.
“In line with current guidelines, patients with suspected STEMI should be managed with PPCI, without delay while the safety of health care providers is ensured,” Ran Kornowski, MD, and Katia Orvin, MD, both with Rabin Medical Center, Petah Tikva, Israel, and Tel Aviv University, wrote in a linked editorial.
“In this case, PPCI should be performed routinely, even if the patient is presumed to have COVID-19, because PPCI should not be postponed. Confirmation of SARS-CoV-2 infection should not delay urgent decision management concerning reperfusion strategy,” they advised.
Looking ahead, Garcia said plans for the registry include determining predictors of in-hospital mortality and studying demographic and treatment trends as the pandemic continues with more virulent strains of the virus.
Various subgroup analyses are also planned as well as an independent angiographic and electrocardiographic core lab analysis. A comparative analysis of data from the US and Canada is also planned.
This work was supported by an ACC Accreditation Grant, Saskatchewan Health Research Foundation, and grants from Medtronic and Abbott Vascular to SCAI. Dr. Garcia has received institutional research grants from Edwards Lifesciences, BSCI, Medtronic, and Abbott Vascular; has served as a consultant for Medtronic and BSCI; and has served as a proctor for Edwards Lifesciences. Dr. Kornowski and Dr. Orvin disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID-19 patients with ST-segment elevation MI (STEMI) represent a population with unique demographic and clinical features resulting in a high risk for mortality, according to initial findings from the North American Cardiovascular COVID-19 Myocardial Infarction (NACMI) Registry.

“This is the largest registry of COVID-positive patients presenting with STEMI [and] the results clearly illustrate the challenges and uniqueness of this patient population that deserves prompt and special attention,” study cochair Timothy Henry, MD, president-elect of the Society for Cardiovascular Angiography & Interventions, said in a news release.
The NACMI registry is a collaborative effort between the SCAI, the American College of Cardiology Interventional Council, and the Canadian Association of Interventional Cardiology.
“The rapid development of this ongoing, critically important prospective registry reflects the strong and unique collaboration of all three societies. It was gratifying to be part of this process and hopefully the results will improve the care of our patients and stimulate further research,” Dr. Henry said in the news release.
The registry has enrolled 1,185 patients presenting with STEMI at 64 sites across the United States and Canada. Participants include 230 COVID-positive STEMI patients; 495 STEMI patients suspected but ultimately confirmed not to have COVID-19; and 460 age-and sex-matched control STEMI patients treated prior to the pandemic who are part of the Midwest STEMI Consortium.
The initial findings from the registry were published online in the Journal of the American College of Cardiology.
Atypical symptoms may explain high death rate
The primary outcome – a composite of in-hospital death, stroke, recurrent MI, or repeat unplanned revascularization – occurred in 36% of COVID-positive patients, compared with 13% of COVID-negative patients and 5% of control patients (P < .001 relative to controls).
This difference was driven largely by a “very high” in-hospital death rate in COVID-positive patients, lead author Santiago Garcia, MD, Minneapolis Heart Institute Foundation, said in an interview.
The in-hospital death rate was 33% in COVID-positive patients, compared with 11% in COVID-negative patients and 4% in controls. Stroke also occurred more often in COVID-positive patients at 3% versus 2% in COVID-negative and 0% in controls.
These initial findings suggest that the combination of STEMI and COVID-19 infection “confers a poor prognosis, with one in three patients succumbing to the disease, even among patients selected for invasive angiography (28% mortality),” the investigators wrote.
The data also show that STEMI in COVID-positive patients disproportionately affects ethnic minorities (23% Hispanic and 24% Black) with diabetes, which was present in 46% of COVID-positive patients.
COVID-positive patients with STEMI are more likely to present with atypical symptoms such as dyspnea (54%), pulmonary infiltrates on chest x-ray (46%), and high-risk conditions such as cardiogenic shock (18%), “which may explain the high fatality rate,” Dr. Garcia said.
Despite these high-risk features, COVID-positive patients are less apt to undergo invasive angiography when compared with COVID-negative and control STEMI patients (78% vs. 96% vs. 100%).
The majority of patients (71%) who did under angiography received primary percutaneous coronary intervention (PPCI) with very small treatment delays (at 15 minutes) during the pandemic.
Another notable finding is that “many patients (23%) have ‘no culprit’ vessel and may represent different etiologies of ST-segment elevation including microemboli, myocarditis, Takotsubo cardiomyopathy,” Dr. Garcia said in an interview.
“In line with current guidelines, patients with suspected STEMI should be managed with PPCI, without delay while the safety of health care providers is ensured,” Ran Kornowski, MD, and Katia Orvin, MD, both with Rabin Medical Center, Petah Tikva, Israel, and Tel Aviv University, wrote in a linked editorial.
“In this case, PPCI should be performed routinely, even if the patient is presumed to have COVID-19, because PPCI should not be postponed. Confirmation of SARS-CoV-2 infection should not delay urgent decision management concerning reperfusion strategy,” they advised.
Looking ahead, Garcia said plans for the registry include determining predictors of in-hospital mortality and studying demographic and treatment trends as the pandemic continues with more virulent strains of the virus.
Various subgroup analyses are also planned as well as an independent angiographic and electrocardiographic core lab analysis. A comparative analysis of data from the US and Canada is also planned.
This work was supported by an ACC Accreditation Grant, Saskatchewan Health Research Foundation, and grants from Medtronic and Abbott Vascular to SCAI. Dr. Garcia has received institutional research grants from Edwards Lifesciences, BSCI, Medtronic, and Abbott Vascular; has served as a consultant for Medtronic and BSCI; and has served as a proctor for Edwards Lifesciences. Dr. Kornowski and Dr. Orvin disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cushing’s death rate ‘unacceptable,’ triple that of general population
Excess mortality among people with endogenous Cushing’s syndrome (CS) has declined in the past 20 years yet remains three times higher than in the general population, new research finds.
Among more than 90,000 individuals with endogenous CS, the overall proportion of mortality – defined as the ratio of the number of deaths from CS divided by the total number of CS patients – was 0.05, and the standardized mortality rate was an “unacceptable” three times that of the general population, Padiporn Limumpornpetch, MD, reported on March 20 at ENDO 2021: The Endocrine Society Annual Meeting.
Excess deaths were higher among those with adrenal CS, compared with those with Cushing’s disease. The most common causes of death among those with CS were cardiovascular diseases, cerebrovascular accident, infection, and malignancy, noted Dr. Limumpornpetch, of Songkla University, Hat Yai, Thailand, who is also a PhD student at the University of Leeds, United Kingdom.
“While mortality has improved since 2000, it is still significantly compromised compared to the background population ... The causes of death highlight the need for aggressive management of cardiovascular risk, prevention of thromboembolism, infection control, and a normalized cortisol level,” she said.
Asked to comment, Maria Fleseriu, MD, told this news organization that the new data show “we are making improvements in the care of patients with CS and thus outcomes, but we are not there yet ... This meta-analysis highlights the whole spectrum of acute and life-threatening complications in CS and their high prevalence, even before disease diagnosis and after successful surgery.”
She noted that although she wasn’t surprised by the overall results, “the improvement over time was indeed lower than I expected. However, interestingly here, the risk of mortality in adrenal Cushing’s was unexpectedly high despite patients with adrenal cancer being excluded.”
Dr. Fleseriu, who is director of the Pituitary Center at Oregon Health and Science University, Portland, advised, “Management of hyperglycemia and diabetes, hypertension, hypokalemia, hyperlipidemia, and other cardiovascular risk factors is generally undertaken in accordance with standard of clinical care.”
“But we should focus more on optimizing more aggressively this care in addition to the specific Cushing’s treatment,” she stressed.
In addition, she noted, “Medical therapy for CS may be needed even prior to surgery in severe and/or prolonged hypercortisolism to decrease complications ... We definitely need a multidisciplinary approach to address complications and etiologic treatment as well as the reduced long-term quality of life in patients with CS.”
Largest study in scale and scope of Cushing’s syndrome mortality
Endogenous Cushing’s syndrome occurs when the body overproduces cortisol. The most common cause of the latter is a tumor of the pituitary gland (Cushing’s disease), but another cause is a usually benign tumor of the adrenal glands (adrenal Cushing’s syndrome). Surgery is the mainstay of initial treatment of Cushing’s syndrome. If an operation to remove the tumor fails to cause remission, medications are available.
Prior to this new meta-analysis, there had been limited data on mortality among patients with endogenous CS. Research has mostly been limited to single-cohort studies. A previous systematic review/meta-analysis comprised only seven articles with 780 patients. All the studies were conducted prior to 2012, and most were limited to Cushing’s disease.
“In 2021, we lacked a detailed understanding of patient outcomes and mortality because of the rarity of Cushing’s syndrome,” Dr. Limumpornpetch noted.
The current meta-analysis included 91 articles that reported mortality among patients with endogenous CS. There was a total of 19,181 patients from 92 study cohorts, including 49 studies on CD (n = 14,971), 24 studies on adrenal CS (n = 2304), and 19 studies that included both (n = 1906).
Among 21 studies that reported standardized mortality rate (SMR) data, including 13 CD studies (n = 2160) and seven on adrenal CS (n = 1531), the overall increase in mortality compared to the background population was a significant 3.00 (range, 1.15-7.84).
This SMR was higher among patients with adrenal Cushing’s syndrome (3.3) versus Cushing’s disease (2.8) (P = .003) and among patients who had active disease (5.7) versus those whose disease was in remission (2.3) (P < .001).
The SMR was also worse among patients with Cushing’s disease with larger tumors (macroadenomas), at 7.4, than among patients with very small tumors (microadenomas), at 1.9 (P = .004).
The proportion of death was 0.05 for CS overall, with 0.04 for CD and 0.02 for adrenal adenomas.
Compared to studies published prior to the year 2000, more recent studies seem to reflect advances in treatment and care. The overall proportion of death for all CS cohorts dropped from 0.10 to 0.03 (P < .001); for all CD cohorts, it dropped from 0.14 to 0.03; and for adrenal CS cohorts, it dropped from 0.09 to 0.03 (P = .04).
Causes of death were cardiovascular diseases (29.5% of cases), cerebrovascular accident (11.5%), infection (10.5%), and malignancy (10.1%). Less common causes of death were gastrointestinal bleeding and acute pancreatitis (3.7%), active CS (3.5%), adrenal insufficiency (2.5%), suicide (2.5%), and surgery (1.6%).
Overall, in the CS groups, the proportion of deaths within 30 days of surgery dropped from 0.04 prior to 2000 to 0.01 since (P = .07). For CD, the proportion dropped from 0.02 to 0.01 (P = .25).
Preventing perioperative mortality: Consider thromboprophylaxis
Dr. Fleseriu told this news organization that she believes hypercoagulability is “the least recognized complication with a big role in mortality.” Because most of the perioperative mortality is due to venous thromboembolism and infections, “thromboprophylaxis should be considered for CS patients with severe hypercortisolism and/or postoperatively, based on individual risk factors of thromboembolism and bleeding.”
Recently, Dr. Fleseriu’s group showed in a single retrospective study that the risk for arterial and venous thromboembolic events among patients with CS was approximately 20%. Many patients experienced more than one event. Risk was higher 30 to 60 days postoperatively.
The odds ratio of venous thromoboembolism among patients with CS was 18 times higher than in the normal population.
“Due to the additional thrombotic risk of surgery or any invasive procedure, anticoagulation prophylaxis should be at least considered in all patients with Cushing’s syndrome and balanced with individual bleeding risk,” Dr. Fleseriu advised.
A recent Pituitary Society workshop discussed the management of complications of CS at length; proceedings will be published soon, she noted.
Dr. Limumpornpetch commented, “We look forward to the day when our interdisciplinary approach to managing these challenging patients can deliver outcomes similar to the background population.”
Dr. Limumpornpetch has disclosed no relevant financial relationships. Dr. Fleseriu has been a scientific consultant to Recordati, Sparrow, and Strongbridge and has received grants (inst) from Novartis and Strongbridge.
A version of this article first appeared on Medscape.com.
Excess mortality among people with endogenous Cushing’s syndrome (CS) has declined in the past 20 years yet remains three times higher than in the general population, new research finds.
Among more than 90,000 individuals with endogenous CS, the overall proportion of mortality – defined as the ratio of the number of deaths from CS divided by the total number of CS patients – was 0.05, and the standardized mortality rate was an “unacceptable” three times that of the general population, Padiporn Limumpornpetch, MD, reported on March 20 at ENDO 2021: The Endocrine Society Annual Meeting.
Excess deaths were higher among those with adrenal CS, compared with those with Cushing’s disease. The most common causes of death among those with CS were cardiovascular diseases, cerebrovascular accident, infection, and malignancy, noted Dr. Limumpornpetch, of Songkla University, Hat Yai, Thailand, who is also a PhD student at the University of Leeds, United Kingdom.
“While mortality has improved since 2000, it is still significantly compromised compared to the background population ... The causes of death highlight the need for aggressive management of cardiovascular risk, prevention of thromboembolism, infection control, and a normalized cortisol level,” she said.
Asked to comment, Maria Fleseriu, MD, told this news organization that the new data show “we are making improvements in the care of patients with CS and thus outcomes, but we are not there yet ... This meta-analysis highlights the whole spectrum of acute and life-threatening complications in CS and their high prevalence, even before disease diagnosis and after successful surgery.”
She noted that although she wasn’t surprised by the overall results, “the improvement over time was indeed lower than I expected. However, interestingly here, the risk of mortality in adrenal Cushing’s was unexpectedly high despite patients with adrenal cancer being excluded.”
Dr. Fleseriu, who is director of the Pituitary Center at Oregon Health and Science University, Portland, advised, “Management of hyperglycemia and diabetes, hypertension, hypokalemia, hyperlipidemia, and other cardiovascular risk factors is generally undertaken in accordance with standard of clinical care.”
“But we should focus more on optimizing more aggressively this care in addition to the specific Cushing’s treatment,” she stressed.
In addition, she noted, “Medical therapy for CS may be needed even prior to surgery in severe and/or prolonged hypercortisolism to decrease complications ... We definitely need a multidisciplinary approach to address complications and etiologic treatment as well as the reduced long-term quality of life in patients with CS.”
Largest study in scale and scope of Cushing’s syndrome mortality
Endogenous Cushing’s syndrome occurs when the body overproduces cortisol. The most common cause of the latter is a tumor of the pituitary gland (Cushing’s disease), but another cause is a usually benign tumor of the adrenal glands (adrenal Cushing’s syndrome). Surgery is the mainstay of initial treatment of Cushing’s syndrome. If an operation to remove the tumor fails to cause remission, medications are available.
Prior to this new meta-analysis, there had been limited data on mortality among patients with endogenous CS. Research has mostly been limited to single-cohort studies. A previous systematic review/meta-analysis comprised only seven articles with 780 patients. All the studies were conducted prior to 2012, and most were limited to Cushing’s disease.
“In 2021, we lacked a detailed understanding of patient outcomes and mortality because of the rarity of Cushing’s syndrome,” Dr. Limumpornpetch noted.
The current meta-analysis included 91 articles that reported mortality among patients with endogenous CS. There was a total of 19,181 patients from 92 study cohorts, including 49 studies on CD (n = 14,971), 24 studies on adrenal CS (n = 2304), and 19 studies that included both (n = 1906).
Among 21 studies that reported standardized mortality rate (SMR) data, including 13 CD studies (n = 2160) and seven on adrenal CS (n = 1531), the overall increase in mortality compared to the background population was a significant 3.00 (range, 1.15-7.84).
This SMR was higher among patients with adrenal Cushing’s syndrome (3.3) versus Cushing’s disease (2.8) (P = .003) and among patients who had active disease (5.7) versus those whose disease was in remission (2.3) (P < .001).
The SMR was also worse among patients with Cushing’s disease with larger tumors (macroadenomas), at 7.4, than among patients with very small tumors (microadenomas), at 1.9 (P = .004).
The proportion of death was 0.05 for CS overall, with 0.04 for CD and 0.02 for adrenal adenomas.
Compared to studies published prior to the year 2000, more recent studies seem to reflect advances in treatment and care. The overall proportion of death for all CS cohorts dropped from 0.10 to 0.03 (P < .001); for all CD cohorts, it dropped from 0.14 to 0.03; and for adrenal CS cohorts, it dropped from 0.09 to 0.03 (P = .04).
Causes of death were cardiovascular diseases (29.5% of cases), cerebrovascular accident (11.5%), infection (10.5%), and malignancy (10.1%). Less common causes of death were gastrointestinal bleeding and acute pancreatitis (3.7%), active CS (3.5%), adrenal insufficiency (2.5%), suicide (2.5%), and surgery (1.6%).
Overall, in the CS groups, the proportion of deaths within 30 days of surgery dropped from 0.04 prior to 2000 to 0.01 since (P = .07). For CD, the proportion dropped from 0.02 to 0.01 (P = .25).
Preventing perioperative mortality: Consider thromboprophylaxis
Dr. Fleseriu told this news organization that she believes hypercoagulability is “the least recognized complication with a big role in mortality.” Because most of the perioperative mortality is due to venous thromboembolism and infections, “thromboprophylaxis should be considered for CS patients with severe hypercortisolism and/or postoperatively, based on individual risk factors of thromboembolism and bleeding.”
Recently, Dr. Fleseriu’s group showed in a single retrospective study that the risk for arterial and venous thromboembolic events among patients with CS was approximately 20%. Many patients experienced more than one event. Risk was higher 30 to 60 days postoperatively.
The odds ratio of venous thromoboembolism among patients with CS was 18 times higher than in the normal population.
“Due to the additional thrombotic risk of surgery or any invasive procedure, anticoagulation prophylaxis should be at least considered in all patients with Cushing’s syndrome and balanced with individual bleeding risk,” Dr. Fleseriu advised.
A recent Pituitary Society workshop discussed the management of complications of CS at length; proceedings will be published soon, she noted.
Dr. Limumpornpetch commented, “We look forward to the day when our interdisciplinary approach to managing these challenging patients can deliver outcomes similar to the background population.”
Dr. Limumpornpetch has disclosed no relevant financial relationships. Dr. Fleseriu has been a scientific consultant to Recordati, Sparrow, and Strongbridge and has received grants (inst) from Novartis and Strongbridge.
A version of this article first appeared on Medscape.com.
Excess mortality among people with endogenous Cushing’s syndrome (CS) has declined in the past 20 years yet remains three times higher than in the general population, new research finds.
Among more than 90,000 individuals with endogenous CS, the overall proportion of mortality – defined as the ratio of the number of deaths from CS divided by the total number of CS patients – was 0.05, and the standardized mortality rate was an “unacceptable” three times that of the general population, Padiporn Limumpornpetch, MD, reported on March 20 at ENDO 2021: The Endocrine Society Annual Meeting.
Excess deaths were higher among those with adrenal CS, compared with those with Cushing’s disease. The most common causes of death among those with CS were cardiovascular diseases, cerebrovascular accident, infection, and malignancy, noted Dr. Limumpornpetch, of Songkla University, Hat Yai, Thailand, who is also a PhD student at the University of Leeds, United Kingdom.
“While mortality has improved since 2000, it is still significantly compromised compared to the background population ... The causes of death highlight the need for aggressive management of cardiovascular risk, prevention of thromboembolism, infection control, and a normalized cortisol level,” she said.
Asked to comment, Maria Fleseriu, MD, told this news organization that the new data show “we are making improvements in the care of patients with CS and thus outcomes, but we are not there yet ... This meta-analysis highlights the whole spectrum of acute and life-threatening complications in CS and their high prevalence, even before disease diagnosis and after successful surgery.”
She noted that although she wasn’t surprised by the overall results, “the improvement over time was indeed lower than I expected. However, interestingly here, the risk of mortality in adrenal Cushing’s was unexpectedly high despite patients with adrenal cancer being excluded.”
Dr. Fleseriu, who is director of the Pituitary Center at Oregon Health and Science University, Portland, advised, “Management of hyperglycemia and diabetes, hypertension, hypokalemia, hyperlipidemia, and other cardiovascular risk factors is generally undertaken in accordance with standard of clinical care.”
“But we should focus more on optimizing more aggressively this care in addition to the specific Cushing’s treatment,” she stressed.
In addition, she noted, “Medical therapy for CS may be needed even prior to surgery in severe and/or prolonged hypercortisolism to decrease complications ... We definitely need a multidisciplinary approach to address complications and etiologic treatment as well as the reduced long-term quality of life in patients with CS.”
Largest study in scale and scope of Cushing’s syndrome mortality
Endogenous Cushing’s syndrome occurs when the body overproduces cortisol. The most common cause of the latter is a tumor of the pituitary gland (Cushing’s disease), but another cause is a usually benign tumor of the adrenal glands (adrenal Cushing’s syndrome). Surgery is the mainstay of initial treatment of Cushing’s syndrome. If an operation to remove the tumor fails to cause remission, medications are available.
Prior to this new meta-analysis, there had been limited data on mortality among patients with endogenous CS. Research has mostly been limited to single-cohort studies. A previous systematic review/meta-analysis comprised only seven articles with 780 patients. All the studies were conducted prior to 2012, and most were limited to Cushing’s disease.
“In 2021, we lacked a detailed understanding of patient outcomes and mortality because of the rarity of Cushing’s syndrome,” Dr. Limumpornpetch noted.
The current meta-analysis included 91 articles that reported mortality among patients with endogenous CS. There was a total of 19,181 patients from 92 study cohorts, including 49 studies on CD (n = 14,971), 24 studies on adrenal CS (n = 2304), and 19 studies that included both (n = 1906).
Among 21 studies that reported standardized mortality rate (SMR) data, including 13 CD studies (n = 2160) and seven on adrenal CS (n = 1531), the overall increase in mortality compared to the background population was a significant 3.00 (range, 1.15-7.84).
This SMR was higher among patients with adrenal Cushing’s syndrome (3.3) versus Cushing’s disease (2.8) (P = .003) and among patients who had active disease (5.7) versus those whose disease was in remission (2.3) (P < .001).
The SMR was also worse among patients with Cushing’s disease with larger tumors (macroadenomas), at 7.4, than among patients with very small tumors (microadenomas), at 1.9 (P = .004).
The proportion of death was 0.05 for CS overall, with 0.04 for CD and 0.02 for adrenal adenomas.
Compared to studies published prior to the year 2000, more recent studies seem to reflect advances in treatment and care. The overall proportion of death for all CS cohorts dropped from 0.10 to 0.03 (P < .001); for all CD cohorts, it dropped from 0.14 to 0.03; and for adrenal CS cohorts, it dropped from 0.09 to 0.03 (P = .04).
Causes of death were cardiovascular diseases (29.5% of cases), cerebrovascular accident (11.5%), infection (10.5%), and malignancy (10.1%). Less common causes of death were gastrointestinal bleeding and acute pancreatitis (3.7%), active CS (3.5%), adrenal insufficiency (2.5%), suicide (2.5%), and surgery (1.6%).
Overall, in the CS groups, the proportion of deaths within 30 days of surgery dropped from 0.04 prior to 2000 to 0.01 since (P = .07). For CD, the proportion dropped from 0.02 to 0.01 (P = .25).
Preventing perioperative mortality: Consider thromboprophylaxis
Dr. Fleseriu told this news organization that she believes hypercoagulability is “the least recognized complication with a big role in mortality.” Because most of the perioperative mortality is due to venous thromboembolism and infections, “thromboprophylaxis should be considered for CS patients with severe hypercortisolism and/or postoperatively, based on individual risk factors of thromboembolism and bleeding.”
Recently, Dr. Fleseriu’s group showed in a single retrospective study that the risk for arterial and venous thromboembolic events among patients with CS was approximately 20%. Many patients experienced more than one event. Risk was higher 30 to 60 days postoperatively.
The odds ratio of venous thromoboembolism among patients with CS was 18 times higher than in the normal population.
“Due to the additional thrombotic risk of surgery or any invasive procedure, anticoagulation prophylaxis should be at least considered in all patients with Cushing’s syndrome and balanced with individual bleeding risk,” Dr. Fleseriu advised.
A recent Pituitary Society workshop discussed the management of complications of CS at length; proceedings will be published soon, she noted.
Dr. Limumpornpetch commented, “We look forward to the day when our interdisciplinary approach to managing these challenging patients can deliver outcomes similar to the background population.”
Dr. Limumpornpetch has disclosed no relevant financial relationships. Dr. Fleseriu has been a scientific consultant to Recordati, Sparrow, and Strongbridge and has received grants (inst) from Novartis and Strongbridge.
A version of this article first appeared on Medscape.com.
PTSD linked to ischemic heart disease
A study using data from Veterans Health Administration (VHA) electronic medical records shows a significant association between posttraumatic stress disorder (PTSD) among female veterans and an increased risk for incident ischemic heart disease (IHD).
The increased risk for IHD was highest among women younger than 40 with PTSD, and among racial and ethnic minorities.
“These women have been emerging as important targets for cardiovascular prevention, and our study suggests that PTSD may be an important psychosocial risk factor for IHD in these individuals,” wrote the researchers, led by Ramin Ebrahimi, MD, department of medicine, cardiology section, Veterans Affairs Greater Los Angeles Health Care System. “With the number of women veterans growing, it is critical to appreciate the health care needs of this relatively young and diverse patient population.”
The study results also have “important implications for earlier and more aggressive IHD risk assessment, monitoring and management in vulnerable women veterans,” they added. “Indeed, our findings support recent calls for cardiovascular risk screening in younger individuals and for the need to harness a broad range of clinicians who routinely treat younger women to maximize prevention efforts.”
The article was published online in JAMA Cardiology on March 17.
Increasing number of VHA users
“As an interventional cardiologist and the director of the cardiac catheterization laboratory, I noticed a significant number of the patients referred to the cath lab carried a diagnosis of posttraumatic stress disorder,” Dr. Ebrahimi said in an interview. “This intrigued me and started my journey into trying to understand how psychiatric disorders in general, and PTSD, may impact/interact with cardiovascular disorders,” he added.
The number of female veterans in the military has been increasing, and they now make up about 10% of the 20 million American veterans; that number is projected to exceed 2.2 million in the next 20 years, the authors wrote. Female veterans are also the fastest growing group of users of the VHA, they added.
IHD is the leading cause of death in women in the United States, despite the advancements in prevention and treatment. Although women are twice as likely to develop PTSD as are men, and it is even more likely in female veterans, much of the research has predominately been on male veterans, the authors wrote.
For this retrospective study, which used data from the VHA Corporate Data Warehouse, the authors examined a cohort of female veterans who were 18 years or older who had used the VHA health care system between Jan. 1, 2000, and Dec. 31, 2017.
Of the 828,997 female veterans, 151,030 had PTSD. Women excluded from the study were those who did not have any clinical encounters after their index visit, participants who had a diagnosis of IHD at or before the index visit, and those with incident IHD within 90 days of the index visit, allowing time between a PTSD diagnosis and IHD.
Propensity score matching on age at index visit, the number of previous visits, and the presence of traditional and female-specific cardiovascular risk factors, as well as mental and physical health conditions, was conducted to identify female veterans ever diagnosed with PTSD, who were matched in a 1:2 ratio to those never diagnosed with PTSD. In all, 132,923 women with PTSD and 265,846 women without PTSD were included, and data were analyzed for the period of Oct. 1, 2018, to Oct. 30, 2020.
IHD was defined as new-onset coronary artery disease, angina, or myocardial infarction–based ICD-9 and ICD-10 diagnostic codes. Age, race, and ethnicity were self-reported.
The analytic sample consisted of relatively young female veterans (mean [SD] age at baseline, 40.1 [12.2] years) of various races (White, 57.6%; Black, 29.8%) and ethnicities, the authors reported.
Of the 9,940 women who experienced incident IHD during follow-up, 5,559 did not have PTSD (2.1% of the overall population examined) and 4,381 had PTSD (3.3%). PTSD was significantly associated with an increased risk for IHD. Over the median follow-up of 4.9 years, female veterans with PTSD had a 44% higher rate of developing incident IHD compared with the female veterans without PTSD (hazard ratio [HR], 1.44; 95% confidence interval [CI], 1.38-1.50).
In addition, those with PTSD who developed IHD were younger at diagnosis (mean [SD] age, 55.5 [9.7]) than were patients without PTSD (mean [SD] age, 57.8 [10.7]). Effect sizes were largest in the group younger than 40 years (HR, 1.72; 95% CI, 1.55-1.90) and decreased for older participants (HR for those ≥60 years, 1.24; 95% CI, 1.12-1.38)
The authors found a 49% to 66% increase in risk for IHD associated with PTSD in Black women (HR, 1.49; 95% CI, 1.38-1.62) and those identified as non-White and non-Black (HR, 1.66; 95%, 1.33-2.08).
Women of all ethnic groups with PTSD were at higher risk of developing IHD, but this was especially true for Hispanic/Latina women (HR, 1.50; 95% CI 1.22-1.84), they noted.
The authors reported some limitations to their findings. The analytic sample could result in a lower ascertainment of certain conditions, such as psychiatric disorders, they wrote. Substance disorders were low in this study, possibly because of the younger age of female veterans in the sample. Because this study used VHA electronic medical records data, medical care outside of the VHA that was not paid for by the VHA could not be considered.
In addition, although this study used a large sample of female veterans, the findings cannot be generalized to female veterans outside of the VHA system, nonveteran women, or men, the researchers wrote.
A call to action
In an accompanying comment, Beth E. Cohen, MD, of the University of California, San Francisco, and the San Francisco Veterans Affairs Health Care System, points out that the physical implications for psychosocial conditions, including depression and PTSD, have been recognized for quite some time. For example, results of the INTERHEART case-control study of 30,000 people showed stress, depression, and stressful life events accounted for one-third the population-attributable risk for myocardial infarction.
As was also noted by Dr. Ebrahimi and colleagues, much of the current research has been on male veterans, yet types of trauma differ among genders; women experience higher rates of military sexual trauma but lower rates of combat trauma, Dr. Cohen wrote. The PTSD symptoms, trajectory, and biological effects can differ for women and men, as can the pathogenesis, presentation, and outcomes of cardiovascular disease (CVD).
These findings, she said, “are an important extension of the prior literature and represent the largest study in female veterans to date. Although methods differ across studies, the magnitude of risk associated with PTSD was consistent with that found in prior studies of male veterans and nonveteran samples.”
The assessment of age-specific risk is also a strength of the study, “and has implications for clinical practice, because PTSD-associated risk was greatest in a younger group in whom CVD may be overlooked.”
Dr. Cohen addressed the limitations outlined by the authors, including ascertainment bias, severity of PTSD symptoms, and their chronicity, but added that “even in the context of these limitations, this study illustrates the importance of PTSD to the health of women veterans and the additional work needed to reduce their CVD risk.”
Clinical questions remain, she added. Screens for PTSD are widely used in the VHA, yet no studies have examined whether screening or early detection decrease CVD risk. In addition, no evidence suggests that screening for or treatment of PTSD improves cardiovascular outcomes.
“Given the challenges of answering these questions in observational studies, it will be important to incorporate measures of CVD risk and outcomes in trials of behavioral and medical therapies for patients with PTSD,” she wrote.
She added that collaborations among multidisciplinary patient care teams will be important. “The findings of this study represent a call to action for this important work to understand the cardiovascular effects of PTSD and improve the health and well-being of women veterans,” Dr. Cohen concluded.
This research was supported by Investigator-Initiated Research Award from the Department of Defense U.S. Army Medical Research and Material Command Congressionally Directed Medical Research Programs (Dr. Ebrahimi) and in part by grants from the VA Informatics and Computing Infrastructure and the Offices of Research and Development at the Northport, Durham, and Greater Los Angeles Veterans Affairs medical centers. Dr. Ebrahimi reported receiving grants from the Department of Defense during the conduct of the study. Disclosures for other authors are available in the paper. Dr. Cohen reports no disclosures.
A version of this article first appeared on Medscape.com.
A study using data from Veterans Health Administration (VHA) electronic medical records shows a significant association between posttraumatic stress disorder (PTSD) among female veterans and an increased risk for incident ischemic heart disease (IHD).
The increased risk for IHD was highest among women younger than 40 with PTSD, and among racial and ethnic minorities.
“These women have been emerging as important targets for cardiovascular prevention, and our study suggests that PTSD may be an important psychosocial risk factor for IHD in these individuals,” wrote the researchers, led by Ramin Ebrahimi, MD, department of medicine, cardiology section, Veterans Affairs Greater Los Angeles Health Care System. “With the number of women veterans growing, it is critical to appreciate the health care needs of this relatively young and diverse patient population.”
The study results also have “important implications for earlier and more aggressive IHD risk assessment, monitoring and management in vulnerable women veterans,” they added. “Indeed, our findings support recent calls for cardiovascular risk screening in younger individuals and for the need to harness a broad range of clinicians who routinely treat younger women to maximize prevention efforts.”
The article was published online in JAMA Cardiology on March 17.
Increasing number of VHA users
“As an interventional cardiologist and the director of the cardiac catheterization laboratory, I noticed a significant number of the patients referred to the cath lab carried a diagnosis of posttraumatic stress disorder,” Dr. Ebrahimi said in an interview. “This intrigued me and started my journey into trying to understand how psychiatric disorders in general, and PTSD, may impact/interact with cardiovascular disorders,” he added.
The number of female veterans in the military has been increasing, and they now make up about 10% of the 20 million American veterans; that number is projected to exceed 2.2 million in the next 20 years, the authors wrote. Female veterans are also the fastest growing group of users of the VHA, they added.
IHD is the leading cause of death in women in the United States, despite the advancements in prevention and treatment. Although women are twice as likely to develop PTSD as are men, and it is even more likely in female veterans, much of the research has predominately been on male veterans, the authors wrote.
For this retrospective study, which used data from the VHA Corporate Data Warehouse, the authors examined a cohort of female veterans who were 18 years or older who had used the VHA health care system between Jan. 1, 2000, and Dec. 31, 2017.
Of the 828,997 female veterans, 151,030 had PTSD. Women excluded from the study were those who did not have any clinical encounters after their index visit, participants who had a diagnosis of IHD at or before the index visit, and those with incident IHD within 90 days of the index visit, allowing time between a PTSD diagnosis and IHD.
Propensity score matching on age at index visit, the number of previous visits, and the presence of traditional and female-specific cardiovascular risk factors, as well as mental and physical health conditions, was conducted to identify female veterans ever diagnosed with PTSD, who were matched in a 1:2 ratio to those never diagnosed with PTSD. In all, 132,923 women with PTSD and 265,846 women without PTSD were included, and data were analyzed for the period of Oct. 1, 2018, to Oct. 30, 2020.
IHD was defined as new-onset coronary artery disease, angina, or myocardial infarction–based ICD-9 and ICD-10 diagnostic codes. Age, race, and ethnicity were self-reported.
The analytic sample consisted of relatively young female veterans (mean [SD] age at baseline, 40.1 [12.2] years) of various races (White, 57.6%; Black, 29.8%) and ethnicities, the authors reported.
Of the 9,940 women who experienced incident IHD during follow-up, 5,559 did not have PTSD (2.1% of the overall population examined) and 4,381 had PTSD (3.3%). PTSD was significantly associated with an increased risk for IHD. Over the median follow-up of 4.9 years, female veterans with PTSD had a 44% higher rate of developing incident IHD compared with the female veterans without PTSD (hazard ratio [HR], 1.44; 95% confidence interval [CI], 1.38-1.50).
In addition, those with PTSD who developed IHD were younger at diagnosis (mean [SD] age, 55.5 [9.7]) than were patients without PTSD (mean [SD] age, 57.8 [10.7]). Effect sizes were largest in the group younger than 40 years (HR, 1.72; 95% CI, 1.55-1.90) and decreased for older participants (HR for those ≥60 years, 1.24; 95% CI, 1.12-1.38)
The authors found a 49% to 66% increase in risk for IHD associated with PTSD in Black women (HR, 1.49; 95% CI, 1.38-1.62) and those identified as non-White and non-Black (HR, 1.66; 95%, 1.33-2.08).
Women of all ethnic groups with PTSD were at higher risk of developing IHD, but this was especially true for Hispanic/Latina women (HR, 1.50; 95% CI 1.22-1.84), they noted.
The authors reported some limitations to their findings. The analytic sample could result in a lower ascertainment of certain conditions, such as psychiatric disorders, they wrote. Substance disorders were low in this study, possibly because of the younger age of female veterans in the sample. Because this study used VHA electronic medical records data, medical care outside of the VHA that was not paid for by the VHA could not be considered.
In addition, although this study used a large sample of female veterans, the findings cannot be generalized to female veterans outside of the VHA system, nonveteran women, or men, the researchers wrote.
A call to action
In an accompanying comment, Beth E. Cohen, MD, of the University of California, San Francisco, and the San Francisco Veterans Affairs Health Care System, points out that the physical implications for psychosocial conditions, including depression and PTSD, have been recognized for quite some time. For example, results of the INTERHEART case-control study of 30,000 people showed stress, depression, and stressful life events accounted for one-third the population-attributable risk for myocardial infarction.
As was also noted by Dr. Ebrahimi and colleagues, much of the current research has been on male veterans, yet types of trauma differ among genders; women experience higher rates of military sexual trauma but lower rates of combat trauma, Dr. Cohen wrote. The PTSD symptoms, trajectory, and biological effects can differ for women and men, as can the pathogenesis, presentation, and outcomes of cardiovascular disease (CVD).
These findings, she said, “are an important extension of the prior literature and represent the largest study in female veterans to date. Although methods differ across studies, the magnitude of risk associated with PTSD was consistent with that found in prior studies of male veterans and nonveteran samples.”
The assessment of age-specific risk is also a strength of the study, “and has implications for clinical practice, because PTSD-associated risk was greatest in a younger group in whom CVD may be overlooked.”
Dr. Cohen addressed the limitations outlined by the authors, including ascertainment bias, severity of PTSD symptoms, and their chronicity, but added that “even in the context of these limitations, this study illustrates the importance of PTSD to the health of women veterans and the additional work needed to reduce their CVD risk.”
Clinical questions remain, she added. Screens for PTSD are widely used in the VHA, yet no studies have examined whether screening or early detection decrease CVD risk. In addition, no evidence suggests that screening for or treatment of PTSD improves cardiovascular outcomes.
“Given the challenges of answering these questions in observational studies, it will be important to incorporate measures of CVD risk and outcomes in trials of behavioral and medical therapies for patients with PTSD,” she wrote.
She added that collaborations among multidisciplinary patient care teams will be important. “The findings of this study represent a call to action for this important work to understand the cardiovascular effects of PTSD and improve the health and well-being of women veterans,” Dr. Cohen concluded.
This research was supported by Investigator-Initiated Research Award from the Department of Defense U.S. Army Medical Research and Material Command Congressionally Directed Medical Research Programs (Dr. Ebrahimi) and in part by grants from the VA Informatics and Computing Infrastructure and the Offices of Research and Development at the Northport, Durham, and Greater Los Angeles Veterans Affairs medical centers. Dr. Ebrahimi reported receiving grants from the Department of Defense during the conduct of the study. Disclosures for other authors are available in the paper. Dr. Cohen reports no disclosures.
A version of this article first appeared on Medscape.com.
A study using data from Veterans Health Administration (VHA) electronic medical records shows a significant association between posttraumatic stress disorder (PTSD) among female veterans and an increased risk for incident ischemic heart disease (IHD).
The increased risk for IHD was highest among women younger than 40 with PTSD, and among racial and ethnic minorities.
“These women have been emerging as important targets for cardiovascular prevention, and our study suggests that PTSD may be an important psychosocial risk factor for IHD in these individuals,” wrote the researchers, led by Ramin Ebrahimi, MD, department of medicine, cardiology section, Veterans Affairs Greater Los Angeles Health Care System. “With the number of women veterans growing, it is critical to appreciate the health care needs of this relatively young and diverse patient population.”
The study results also have “important implications for earlier and more aggressive IHD risk assessment, monitoring and management in vulnerable women veterans,” they added. “Indeed, our findings support recent calls for cardiovascular risk screening in younger individuals and for the need to harness a broad range of clinicians who routinely treat younger women to maximize prevention efforts.”
The article was published online in JAMA Cardiology on March 17.
Increasing number of VHA users
“As an interventional cardiologist and the director of the cardiac catheterization laboratory, I noticed a significant number of the patients referred to the cath lab carried a diagnosis of posttraumatic stress disorder,” Dr. Ebrahimi said in an interview. “This intrigued me and started my journey into trying to understand how psychiatric disorders in general, and PTSD, may impact/interact with cardiovascular disorders,” he added.
The number of female veterans in the military has been increasing, and they now make up about 10% of the 20 million American veterans; that number is projected to exceed 2.2 million in the next 20 years, the authors wrote. Female veterans are also the fastest growing group of users of the VHA, they added.
IHD is the leading cause of death in women in the United States, despite the advancements in prevention and treatment. Although women are twice as likely to develop PTSD as are men, and it is even more likely in female veterans, much of the research has predominately been on male veterans, the authors wrote.
For this retrospective study, which used data from the VHA Corporate Data Warehouse, the authors examined a cohort of female veterans who were 18 years or older who had used the VHA health care system between Jan. 1, 2000, and Dec. 31, 2017.
Of the 828,997 female veterans, 151,030 had PTSD. Women excluded from the study were those who did not have any clinical encounters after their index visit, participants who had a diagnosis of IHD at or before the index visit, and those with incident IHD within 90 days of the index visit, allowing time between a PTSD diagnosis and IHD.
Propensity score matching on age at index visit, the number of previous visits, and the presence of traditional and female-specific cardiovascular risk factors, as well as mental and physical health conditions, was conducted to identify female veterans ever diagnosed with PTSD, who were matched in a 1:2 ratio to those never diagnosed with PTSD. In all, 132,923 women with PTSD and 265,846 women without PTSD were included, and data were analyzed for the period of Oct. 1, 2018, to Oct. 30, 2020.
IHD was defined as new-onset coronary artery disease, angina, or myocardial infarction–based ICD-9 and ICD-10 diagnostic codes. Age, race, and ethnicity were self-reported.
The analytic sample consisted of relatively young female veterans (mean [SD] age at baseline, 40.1 [12.2] years) of various races (White, 57.6%; Black, 29.8%) and ethnicities, the authors reported.
Of the 9,940 women who experienced incident IHD during follow-up, 5,559 did not have PTSD (2.1% of the overall population examined) and 4,381 had PTSD (3.3%). PTSD was significantly associated with an increased risk for IHD. Over the median follow-up of 4.9 years, female veterans with PTSD had a 44% higher rate of developing incident IHD compared with the female veterans without PTSD (hazard ratio [HR], 1.44; 95% confidence interval [CI], 1.38-1.50).
In addition, those with PTSD who developed IHD were younger at diagnosis (mean [SD] age, 55.5 [9.7]) than were patients without PTSD (mean [SD] age, 57.8 [10.7]). Effect sizes were largest in the group younger than 40 years (HR, 1.72; 95% CI, 1.55-1.90) and decreased for older participants (HR for those ≥60 years, 1.24; 95% CI, 1.12-1.38)
The authors found a 49% to 66% increase in risk for IHD associated with PTSD in Black women (HR, 1.49; 95% CI, 1.38-1.62) and those identified as non-White and non-Black (HR, 1.66; 95%, 1.33-2.08).
Women of all ethnic groups with PTSD were at higher risk of developing IHD, but this was especially true for Hispanic/Latina women (HR, 1.50; 95% CI 1.22-1.84), they noted.
The authors reported some limitations to their findings. The analytic sample could result in a lower ascertainment of certain conditions, such as psychiatric disorders, they wrote. Substance disorders were low in this study, possibly because of the younger age of female veterans in the sample. Because this study used VHA electronic medical records data, medical care outside of the VHA that was not paid for by the VHA could not be considered.
In addition, although this study used a large sample of female veterans, the findings cannot be generalized to female veterans outside of the VHA system, nonveteran women, or men, the researchers wrote.
A call to action
In an accompanying comment, Beth E. Cohen, MD, of the University of California, San Francisco, and the San Francisco Veterans Affairs Health Care System, points out that the physical implications for psychosocial conditions, including depression and PTSD, have been recognized for quite some time. For example, results of the INTERHEART case-control study of 30,000 people showed stress, depression, and stressful life events accounted for one-third the population-attributable risk for myocardial infarction.
As was also noted by Dr. Ebrahimi and colleagues, much of the current research has been on male veterans, yet types of trauma differ among genders; women experience higher rates of military sexual trauma but lower rates of combat trauma, Dr. Cohen wrote. The PTSD symptoms, trajectory, and biological effects can differ for women and men, as can the pathogenesis, presentation, and outcomes of cardiovascular disease (CVD).
These findings, she said, “are an important extension of the prior literature and represent the largest study in female veterans to date. Although methods differ across studies, the magnitude of risk associated with PTSD was consistent with that found in prior studies of male veterans and nonveteran samples.”
The assessment of age-specific risk is also a strength of the study, “and has implications for clinical practice, because PTSD-associated risk was greatest in a younger group in whom CVD may be overlooked.”
Dr. Cohen addressed the limitations outlined by the authors, including ascertainment bias, severity of PTSD symptoms, and their chronicity, but added that “even in the context of these limitations, this study illustrates the importance of PTSD to the health of women veterans and the additional work needed to reduce their CVD risk.”
Clinical questions remain, she added. Screens for PTSD are widely used in the VHA, yet no studies have examined whether screening or early detection decrease CVD risk. In addition, no evidence suggests that screening for or treatment of PTSD improves cardiovascular outcomes.
“Given the challenges of answering these questions in observational studies, it will be important to incorporate measures of CVD risk and outcomes in trials of behavioral and medical therapies for patients with PTSD,” she wrote.
She added that collaborations among multidisciplinary patient care teams will be important. “The findings of this study represent a call to action for this important work to understand the cardiovascular effects of PTSD and improve the health and well-being of women veterans,” Dr. Cohen concluded.
This research was supported by Investigator-Initiated Research Award from the Department of Defense U.S. Army Medical Research and Material Command Congressionally Directed Medical Research Programs (Dr. Ebrahimi) and in part by grants from the VA Informatics and Computing Infrastructure and the Offices of Research and Development at the Northport, Durham, and Greater Los Angeles Veterans Affairs medical centers. Dr. Ebrahimi reported receiving grants from the Department of Defense during the conduct of the study. Disclosures for other authors are available in the paper. Dr. Cohen reports no disclosures.
A version of this article first appeared on Medscape.com.
Severe obesity persists, takes high cardiovascular toll
In a U.K. cohort of more than 260,000 mostly middle-aged adults in primary care with overweight or obesity, body mass index remained relatively stable over a decade.
However, compared to overweight individuals, those with severe (class 3) obesity were more socioeconomically disadvantaged and had triple the risk for incident heart failure or all-cause or cardiovascular disease (CVD)–related mortality in a study published online April 15 in BMC Public Health.
“This is the first study to evaluate the long-term impact of overweight and obese individuals’ BMI trajectory on cardiovascular endpoints, heart failure, and mortality outcomes,” wrote Barbara Iyen, PhD, University of Nottingham, England, and colleagues.
The findings emphasize “the high cardiovascular toll exacted by continuing failure to tackle obesity, particularly among more socioeconomically deprived populations,” they warned.
“We have found that despite widespread efforts to prevent and manage obesity, the majority of adults who are overweight or obese in the general population continue to remain so in the long term,” Dr. Iyen said in a statement from her university.
“More effective policies and weight-management interventions are needed urgently to address this increasing burden and associated adverse health outcomes,” she stressed.
Invited to comment, Sadiya S. Khan, MD, Northwestern University, Chicago, said in an interview: “This research adds to the growing body of evidence [that] earlier and more intensive interventions for weight loss are necessary to promote cardiovascular health and reduce morbidity and mortality.
“Adjunctive pharmacotherapy and bariatric surgery are both options that should be considered in addition to intensive lifestyle interventions in overweight and obesity groups,” she added.
“I would always advocate for earlier prevention efforts focused on weight loss, because years lived with obesity are associated with future CVD, so every year counts,” Dr. Khan said.
Does BMI remain elevated, predict worse heart health?
Although obesity is a well-recognized risk factor for CVD, long-term changes in BMI and the impact of BMI on the risk for heart failure, CVD, and mortality have not been quantified among adults with overweight and obesity, Dr. Iyen and colleagues explained.
The researchers examined data from the UK Clinical Practice Research Datalink and secondary care and mortality records to determine BMI trajectories among adults with overweight or obesity and to quantify the risk for heart failure, CVD (defined as coronary heart disease, stroke, transient ischemic attack, or peripheral vascular disease, CVD-related mortality, and all-cause mortality.
They identified 264,230 adults with overweight or obesity who were seen in 790 primary care practices in the United Kingdom from 1999 to 2018 and who did not initially have heart failure or CVD and for whom baseline BMI measurements and at least one other BMI measurement 2, 5, 8, and 10 years later was available.
The researchers divided the cohort into four groups on the basis of initial BMI: overweight (36% of patients; mean BMI, 28.7 kg/m2); class 1 obesity (40%; mean BMI, 33.7 kg/m2); class 2 obesity (19%; mean BMI, 39.9 kg/m2), and class 3 obesity (5%; mean BMI, 49.1 kg/m2).
The mean age of the individuals was 50 years, and 64% were White. Race/ethnicity data were unavailable for 31%. Asian Indian, Asian, and Black patients comprised 5% of the cohort.
“Strong significant gradient in heart failure risk”
Compared to the overweight (reference) group, the severe-obesity group comprised a higher percentage of women (74% vs. 70%), and the prevalence of comorbidities and socioeconomic deprivation was higher.
BMI remained relatively stable in each BMI group. The mean BMI increase was 1.06 kg/m2 during a median follow-up of 10.9 years.
There were 30,400 incident cases of CVD, 7,662 incident cases of heart failure, and 24,022 deaths, of which 2,827 (11.8%) were from CVD.
The risk for heart failure and CVD-related or all-cause mortality increased with increasing obesity severity.
Compared with overweight individuals, those with class 3 obesity were at significantly increased risk for heart failure (hazard ratio [HR], 3.26), all-cause mortality (HR, 2.72), and CVD-related mortality (HR, 3.31) after adjustment for age, sex, and comorbidities (hypertension, type 2 diabetes, atrial fibrillation, and chronic kidney disease).
The risk for stroke/TIA or coronary heart disease was similar among those with severe obesity and the other individuals. The risk for PVD was significantly lower (HR, 0.73).
The reduced risk for PVD in the most severely obese group is similar to findings in the Framingham heart study, the authors noted, and may be due to underdiagnosis or differences in the underlying mechanism.
Compelling evidence of poor health outcomes associated with obesity
Study limitations include the fact that the findings may not be generalizable to other race/ethnicity groups, the lack of information on diet and exercise, and the fact that BMI was used as a surrogate of adiposity. As such, it does not account for an age-related decrease in heavier-than-fat muscle mass and differences between sexes and ethnic groups.
The finding of stable obesity over time accords with two smaller studies that included Canadian and American adults.
The current study did not uncover an obesity paradox, unlike some studies that included patients with preexisting CVD or a history of acute coronary events. Those studies reported better clinical outcomes among patients with overweight or obesity.
The current study included individuals who did not initially have CVD. Those with more severe obesity were younger than individuals with overweight at the time of the occurrence of incident CVD (age 64 vs. 66) and at the age of death (age 67 vs. age 75), which “provides compelling evidence of poor health outcomes associated with obesity,” the authors emphasized.
“Further research is ... needed to explore whether interventions to change BMI trajectories would have an impact on future CVD outcomes,” they concluded.
Dr. Iyen’s clinical academic lectureship is fully funded by the National Institute for Health Research (NIHR). The views expressed are those of the authors and are not necessarily those of the National Health Service, the NIHR, or the Department of Health and Social Care. Dr. Khan has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a U.K. cohort of more than 260,000 mostly middle-aged adults in primary care with overweight or obesity, body mass index remained relatively stable over a decade.
However, compared to overweight individuals, those with severe (class 3) obesity were more socioeconomically disadvantaged and had triple the risk for incident heart failure or all-cause or cardiovascular disease (CVD)–related mortality in a study published online April 15 in BMC Public Health.
“This is the first study to evaluate the long-term impact of overweight and obese individuals’ BMI trajectory on cardiovascular endpoints, heart failure, and mortality outcomes,” wrote Barbara Iyen, PhD, University of Nottingham, England, and colleagues.
The findings emphasize “the high cardiovascular toll exacted by continuing failure to tackle obesity, particularly among more socioeconomically deprived populations,” they warned.
“We have found that despite widespread efforts to prevent and manage obesity, the majority of adults who are overweight or obese in the general population continue to remain so in the long term,” Dr. Iyen said in a statement from her university.
“More effective policies and weight-management interventions are needed urgently to address this increasing burden and associated adverse health outcomes,” she stressed.
Invited to comment, Sadiya S. Khan, MD, Northwestern University, Chicago, said in an interview: “This research adds to the growing body of evidence [that] earlier and more intensive interventions for weight loss are necessary to promote cardiovascular health and reduce morbidity and mortality.
“Adjunctive pharmacotherapy and bariatric surgery are both options that should be considered in addition to intensive lifestyle interventions in overweight and obesity groups,” she added.
“I would always advocate for earlier prevention efforts focused on weight loss, because years lived with obesity are associated with future CVD, so every year counts,” Dr. Khan said.
Does BMI remain elevated, predict worse heart health?
Although obesity is a well-recognized risk factor for CVD, long-term changes in BMI and the impact of BMI on the risk for heart failure, CVD, and mortality have not been quantified among adults with overweight and obesity, Dr. Iyen and colleagues explained.
The researchers examined data from the UK Clinical Practice Research Datalink and secondary care and mortality records to determine BMI trajectories among adults with overweight or obesity and to quantify the risk for heart failure, CVD (defined as coronary heart disease, stroke, transient ischemic attack, or peripheral vascular disease, CVD-related mortality, and all-cause mortality.
They identified 264,230 adults with overweight or obesity who were seen in 790 primary care practices in the United Kingdom from 1999 to 2018 and who did not initially have heart failure or CVD and for whom baseline BMI measurements and at least one other BMI measurement 2, 5, 8, and 10 years later was available.
The researchers divided the cohort into four groups on the basis of initial BMI: overweight (36% of patients; mean BMI, 28.7 kg/m2); class 1 obesity (40%; mean BMI, 33.7 kg/m2); class 2 obesity (19%; mean BMI, 39.9 kg/m2), and class 3 obesity (5%; mean BMI, 49.1 kg/m2).
The mean age of the individuals was 50 years, and 64% were White. Race/ethnicity data were unavailable for 31%. Asian Indian, Asian, and Black patients comprised 5% of the cohort.
“Strong significant gradient in heart failure risk”
Compared to the overweight (reference) group, the severe-obesity group comprised a higher percentage of women (74% vs. 70%), and the prevalence of comorbidities and socioeconomic deprivation was higher.
BMI remained relatively stable in each BMI group. The mean BMI increase was 1.06 kg/m2 during a median follow-up of 10.9 years.
There were 30,400 incident cases of CVD, 7,662 incident cases of heart failure, and 24,022 deaths, of which 2,827 (11.8%) were from CVD.
The risk for heart failure and CVD-related or all-cause mortality increased with increasing obesity severity.
Compared with overweight individuals, those with class 3 obesity were at significantly increased risk for heart failure (hazard ratio [HR], 3.26), all-cause mortality (HR, 2.72), and CVD-related mortality (HR, 3.31) after adjustment for age, sex, and comorbidities (hypertension, type 2 diabetes, atrial fibrillation, and chronic kidney disease).
The risk for stroke/TIA or coronary heart disease was similar among those with severe obesity and the other individuals. The risk for PVD was significantly lower (HR, 0.73).
The reduced risk for PVD in the most severely obese group is similar to findings in the Framingham heart study, the authors noted, and may be due to underdiagnosis or differences in the underlying mechanism.
Compelling evidence of poor health outcomes associated with obesity
Study limitations include the fact that the findings may not be generalizable to other race/ethnicity groups, the lack of information on diet and exercise, and the fact that BMI was used as a surrogate of adiposity. As such, it does not account for an age-related decrease in heavier-than-fat muscle mass and differences between sexes and ethnic groups.
The finding of stable obesity over time accords with two smaller studies that included Canadian and American adults.
The current study did not uncover an obesity paradox, unlike some studies that included patients with preexisting CVD or a history of acute coronary events. Those studies reported better clinical outcomes among patients with overweight or obesity.
The current study included individuals who did not initially have CVD. Those with more severe obesity were younger than individuals with overweight at the time of the occurrence of incident CVD (age 64 vs. 66) and at the age of death (age 67 vs. age 75), which “provides compelling evidence of poor health outcomes associated with obesity,” the authors emphasized.
“Further research is ... needed to explore whether interventions to change BMI trajectories would have an impact on future CVD outcomes,” they concluded.
Dr. Iyen’s clinical academic lectureship is fully funded by the National Institute for Health Research (NIHR). The views expressed are those of the authors and are not necessarily those of the National Health Service, the NIHR, or the Department of Health and Social Care. Dr. Khan has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a U.K. cohort of more than 260,000 mostly middle-aged adults in primary care with overweight or obesity, body mass index remained relatively stable over a decade.
However, compared to overweight individuals, those with severe (class 3) obesity were more socioeconomically disadvantaged and had triple the risk for incident heart failure or all-cause or cardiovascular disease (CVD)–related mortality in a study published online April 15 in BMC Public Health.
“This is the first study to evaluate the long-term impact of overweight and obese individuals’ BMI trajectory on cardiovascular endpoints, heart failure, and mortality outcomes,” wrote Barbara Iyen, PhD, University of Nottingham, England, and colleagues.
The findings emphasize “the high cardiovascular toll exacted by continuing failure to tackle obesity, particularly among more socioeconomically deprived populations,” they warned.
“We have found that despite widespread efforts to prevent and manage obesity, the majority of adults who are overweight or obese in the general population continue to remain so in the long term,” Dr. Iyen said in a statement from her university.
“More effective policies and weight-management interventions are needed urgently to address this increasing burden and associated adverse health outcomes,” she stressed.
Invited to comment, Sadiya S. Khan, MD, Northwestern University, Chicago, said in an interview: “This research adds to the growing body of evidence [that] earlier and more intensive interventions for weight loss are necessary to promote cardiovascular health and reduce morbidity and mortality.
“Adjunctive pharmacotherapy and bariatric surgery are both options that should be considered in addition to intensive lifestyle interventions in overweight and obesity groups,” she added.
“I would always advocate for earlier prevention efforts focused on weight loss, because years lived with obesity are associated with future CVD, so every year counts,” Dr. Khan said.
Does BMI remain elevated, predict worse heart health?
Although obesity is a well-recognized risk factor for CVD, long-term changes in BMI and the impact of BMI on the risk for heart failure, CVD, and mortality have not been quantified among adults with overweight and obesity, Dr. Iyen and colleagues explained.
The researchers examined data from the UK Clinical Practice Research Datalink and secondary care and mortality records to determine BMI trajectories among adults with overweight or obesity and to quantify the risk for heart failure, CVD (defined as coronary heart disease, stroke, transient ischemic attack, or peripheral vascular disease, CVD-related mortality, and all-cause mortality.
They identified 264,230 adults with overweight or obesity who were seen in 790 primary care practices in the United Kingdom from 1999 to 2018 and who did not initially have heart failure or CVD and for whom baseline BMI measurements and at least one other BMI measurement 2, 5, 8, and 10 years later was available.
The researchers divided the cohort into four groups on the basis of initial BMI: overweight (36% of patients; mean BMI, 28.7 kg/m2); class 1 obesity (40%; mean BMI, 33.7 kg/m2); class 2 obesity (19%; mean BMI, 39.9 kg/m2), and class 3 obesity (5%; mean BMI, 49.1 kg/m2).
The mean age of the individuals was 50 years, and 64% were White. Race/ethnicity data were unavailable for 31%. Asian Indian, Asian, and Black patients comprised 5% of the cohort.
“Strong significant gradient in heart failure risk”
Compared to the overweight (reference) group, the severe-obesity group comprised a higher percentage of women (74% vs. 70%), and the prevalence of comorbidities and socioeconomic deprivation was higher.
BMI remained relatively stable in each BMI group. The mean BMI increase was 1.06 kg/m2 during a median follow-up of 10.9 years.
There were 30,400 incident cases of CVD, 7,662 incident cases of heart failure, and 24,022 deaths, of which 2,827 (11.8%) were from CVD.
The risk for heart failure and CVD-related or all-cause mortality increased with increasing obesity severity.
Compared with overweight individuals, those with class 3 obesity were at significantly increased risk for heart failure (hazard ratio [HR], 3.26), all-cause mortality (HR, 2.72), and CVD-related mortality (HR, 3.31) after adjustment for age, sex, and comorbidities (hypertension, type 2 diabetes, atrial fibrillation, and chronic kidney disease).
The risk for stroke/TIA or coronary heart disease was similar among those with severe obesity and the other individuals. The risk for PVD was significantly lower (HR, 0.73).
The reduced risk for PVD in the most severely obese group is similar to findings in the Framingham heart study, the authors noted, and may be due to underdiagnosis or differences in the underlying mechanism.
Compelling evidence of poor health outcomes associated with obesity
Study limitations include the fact that the findings may not be generalizable to other race/ethnicity groups, the lack of information on diet and exercise, and the fact that BMI was used as a surrogate of adiposity. As such, it does not account for an age-related decrease in heavier-than-fat muscle mass and differences between sexes and ethnic groups.
The finding of stable obesity over time accords with two smaller studies that included Canadian and American adults.
The current study did not uncover an obesity paradox, unlike some studies that included patients with preexisting CVD or a history of acute coronary events. Those studies reported better clinical outcomes among patients with overweight or obesity.
The current study included individuals who did not initially have CVD. Those with more severe obesity were younger than individuals with overweight at the time of the occurrence of incident CVD (age 64 vs. 66) and at the age of death (age 67 vs. age 75), which “provides compelling evidence of poor health outcomes associated with obesity,” the authors emphasized.
“Further research is ... needed to explore whether interventions to change BMI trajectories would have an impact on future CVD outcomes,” they concluded.
Dr. Iyen’s clinical academic lectureship is fully funded by the National Institute for Health Research (NIHR). The views expressed are those of the authors and are not necessarily those of the National Health Service, the NIHR, or the Department of Health and Social Care. Dr. Khan has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
HDL anti-inflammatory effects show prognostic potential
The high-density lipoprotein particle’s complexity as a mediator of cardiovascular risk was on display in a case-control study that, the researchers say, points to its anti-inflammatory capacity as potentially a worthy addition to standard CV risk assessments.
A measure of HDL anti-inflammatory capacity in a prospective community cohort was inversely related to future CV risk independent of HDL’s role in cholesterol transport, total cholesterol, and other established biomarkers, as well as any lipid-modifying therapy.
The current analysis “identified an impaired HDL anti-inflammatory capacity as a functional metric prospectively associated with increased cardiovascular risk in the general population,” observed the authors of the study, published April 12, 2021, in Circulation, led by Congzhuo Jia, MD, University of Groningen (the Netherlands).
“In contrast with the cholesterol efflux function of HDL that tracks moderately with HDL cholesterol levels,” they wrote, HDL anti-inflammatory capacity was not significantly correlated with actual levels of the lipoprotein or a major constituent, apolipoprotein A1 (apoA1). Nor was it correlated with levels of a more generalized inflammatory biomarker, C-reactive protein by high-sensitivity assay (hsCRP).
In a test of its independence as a prognosticator, HDL anti-inflammatory capacity significantly and meaningfully improved prediction of CV events in the study after it was added to the familiar Framingham risk equations.
Measurement of HDL anti-inflammatory properties, therefore, has the potential to improve current CV risk assessments in people without clinical heart disease, the authors proposed.
The study “adds to our understanding of the potential cardioprotective role of HDL,” Michael Miller, MD, University of Maryland, Baltimore, said in an interview.
“We’ve known for some time that HDL has anti-inflammatory properties in vitro, and my understanding is this is the first study to assess these anti-inflammatory properties in a clinical trial,” said Dr. Miller, who studies lipid metabolism and directs the Center for Preventive Cardiology at his center but isn’t an author of the report.
The study is part of a long line of research aiming to “untangle the complexities of HDL and try to get a better handle as to the properties that make it cardioprotective,” he said. For example, “high levels are not always associated with cardioprotection, and low levels don’t always imply increased risk.”
The current findings highlight a quality of HDL that might be prognostic but also independent of its concentrations, apoA1 content, or cholesterol efflux capacity, Dr. Miller noted. That makes HDL anti-inflammatory capacity a “promising feature” of HDL that, if confirmed in further studies, could potentially be brought into the mainstream for CV risk prediction. “But it’s too premature at this time.”
The study of participants in the population-based PREVEND cohort study compared 340 patients with a first CV event – CV death, ischemic heart disease, nonfatal MI, or coronary revascularization – over a median of about 10 years with the same number of participants without such events. The two cohorts of people from the same city in the Netherlands had been matched according to sex, smoking status, age, and HDL cholesterol levels at baseline.
No measured clinical or laboratory value, the group wrote, was significantly correlated with HDL anti-inflammatory capacity, defined here as ability to suppress vascular cell adhesion molecule-1 (VCAM-1) mRNA expression as induced by tumor necrosis factor–alpha in endothelial cells in vitro.
HDL anti-inflammatory capacity was significantly lower in the case cohort, compared with the control cohort (P < .001), and was inversely related to new CV events, at an odds ratio per 1 standard deviation of 0.74 (95% confidence interval, 0.61-0.90; P = .002). Covariate adjustments included body mass index; alcohol intake; diabetes and hypertension status; use of lipid-lowering medicine; levels of total cholesterol, apoA1, triglyceride, and hsCRP; and measures of renal function.
No significant association was seen between HDL anti-inflammatory capacity and cholesterol efflux capacity (coefficient of correlation, −0.02; P > .05). But both metrics were independently associated with CV disease events. The OR per 1 standard deviation was 0.74 (95% CI, 0.61-0.90; P = .002) for cholesterol efflux capacity and 0.66 (95% CI, 0.54-0.81; P < .001) for HDL anti-inflammatory capacity.
Adding HDL anti-inflammatory capacity to the Framingham risk score significantly improved its predictive power; its likelihood-ratio statistic rose from 10.50 to 20.40 (P = .002), the group wrote. The addition of cholesterol efflux capacity further elevated the risk score’s likelihood-ratio statistic to 32.84 (P = .0005).
The analysis has all the limitations of a case-control study, Dr. Miller said, but it does “show a potential reasonable association” between anti-inflammatory capacity and CV risk “that needs to be taken to the next level.”
For example, it could be explored in a controlled trial that tracks anti-inflammatory capacity in individuals who receive an intervention that is likely to improve the biomarker – such as weight loss, he proposed – and follows them for clinical outcomes.
“If you want to elevate the stature of the anti-inflammatory index,” Dr. Miller said, “you will need to show that it’s clinically meaningful.”
Dr. Jia reported no conflicts. Dr. Miller has no relevant disclosures.
A version of this article first appeared on Medscape.com.
The high-density lipoprotein particle’s complexity as a mediator of cardiovascular risk was on display in a case-control study that, the researchers say, points to its anti-inflammatory capacity as potentially a worthy addition to standard CV risk assessments.
A measure of HDL anti-inflammatory capacity in a prospective community cohort was inversely related to future CV risk independent of HDL’s role in cholesterol transport, total cholesterol, and other established biomarkers, as well as any lipid-modifying therapy.
The current analysis “identified an impaired HDL anti-inflammatory capacity as a functional metric prospectively associated with increased cardiovascular risk in the general population,” observed the authors of the study, published April 12, 2021, in Circulation, led by Congzhuo Jia, MD, University of Groningen (the Netherlands).
“In contrast with the cholesterol efflux function of HDL that tracks moderately with HDL cholesterol levels,” they wrote, HDL anti-inflammatory capacity was not significantly correlated with actual levels of the lipoprotein or a major constituent, apolipoprotein A1 (apoA1). Nor was it correlated with levels of a more generalized inflammatory biomarker, C-reactive protein by high-sensitivity assay (hsCRP).
In a test of its independence as a prognosticator, HDL anti-inflammatory capacity significantly and meaningfully improved prediction of CV events in the study after it was added to the familiar Framingham risk equations.
Measurement of HDL anti-inflammatory properties, therefore, has the potential to improve current CV risk assessments in people without clinical heart disease, the authors proposed.
The study “adds to our understanding of the potential cardioprotective role of HDL,” Michael Miller, MD, University of Maryland, Baltimore, said in an interview.
“We’ve known for some time that HDL has anti-inflammatory properties in vitro, and my understanding is this is the first study to assess these anti-inflammatory properties in a clinical trial,” said Dr. Miller, who studies lipid metabolism and directs the Center for Preventive Cardiology at his center but isn’t an author of the report.
The study is part of a long line of research aiming to “untangle the complexities of HDL and try to get a better handle as to the properties that make it cardioprotective,” he said. For example, “high levels are not always associated with cardioprotection, and low levels don’t always imply increased risk.”
The current findings highlight a quality of HDL that might be prognostic but also independent of its concentrations, apoA1 content, or cholesterol efflux capacity, Dr. Miller noted. That makes HDL anti-inflammatory capacity a “promising feature” of HDL that, if confirmed in further studies, could potentially be brought into the mainstream for CV risk prediction. “But it’s too premature at this time.”
The study of participants in the population-based PREVEND cohort study compared 340 patients with a first CV event – CV death, ischemic heart disease, nonfatal MI, or coronary revascularization – over a median of about 10 years with the same number of participants without such events. The two cohorts of people from the same city in the Netherlands had been matched according to sex, smoking status, age, and HDL cholesterol levels at baseline.
No measured clinical or laboratory value, the group wrote, was significantly correlated with HDL anti-inflammatory capacity, defined here as ability to suppress vascular cell adhesion molecule-1 (VCAM-1) mRNA expression as induced by tumor necrosis factor–alpha in endothelial cells in vitro.
HDL anti-inflammatory capacity was significantly lower in the case cohort, compared with the control cohort (P < .001), and was inversely related to new CV events, at an odds ratio per 1 standard deviation of 0.74 (95% confidence interval, 0.61-0.90; P = .002). Covariate adjustments included body mass index; alcohol intake; diabetes and hypertension status; use of lipid-lowering medicine; levels of total cholesterol, apoA1, triglyceride, and hsCRP; and measures of renal function.
No significant association was seen between HDL anti-inflammatory capacity and cholesterol efflux capacity (coefficient of correlation, −0.02; P > .05). But both metrics were independently associated with CV disease events. The OR per 1 standard deviation was 0.74 (95% CI, 0.61-0.90; P = .002) for cholesterol efflux capacity and 0.66 (95% CI, 0.54-0.81; P < .001) for HDL anti-inflammatory capacity.
Adding HDL anti-inflammatory capacity to the Framingham risk score significantly improved its predictive power; its likelihood-ratio statistic rose from 10.50 to 20.40 (P = .002), the group wrote. The addition of cholesterol efflux capacity further elevated the risk score’s likelihood-ratio statistic to 32.84 (P = .0005).
The analysis has all the limitations of a case-control study, Dr. Miller said, but it does “show a potential reasonable association” between anti-inflammatory capacity and CV risk “that needs to be taken to the next level.”
For example, it could be explored in a controlled trial that tracks anti-inflammatory capacity in individuals who receive an intervention that is likely to improve the biomarker – such as weight loss, he proposed – and follows them for clinical outcomes.
“If you want to elevate the stature of the anti-inflammatory index,” Dr. Miller said, “you will need to show that it’s clinically meaningful.”
Dr. Jia reported no conflicts. Dr. Miller has no relevant disclosures.
A version of this article first appeared on Medscape.com.
The high-density lipoprotein particle’s complexity as a mediator of cardiovascular risk was on display in a case-control study that, the researchers say, points to its anti-inflammatory capacity as potentially a worthy addition to standard CV risk assessments.
A measure of HDL anti-inflammatory capacity in a prospective community cohort was inversely related to future CV risk independent of HDL’s role in cholesterol transport, total cholesterol, and other established biomarkers, as well as any lipid-modifying therapy.
The current analysis “identified an impaired HDL anti-inflammatory capacity as a functional metric prospectively associated with increased cardiovascular risk in the general population,” observed the authors of the study, published April 12, 2021, in Circulation, led by Congzhuo Jia, MD, University of Groningen (the Netherlands).
“In contrast with the cholesterol efflux function of HDL that tracks moderately with HDL cholesterol levels,” they wrote, HDL anti-inflammatory capacity was not significantly correlated with actual levels of the lipoprotein or a major constituent, apolipoprotein A1 (apoA1). Nor was it correlated with levels of a more generalized inflammatory biomarker, C-reactive protein by high-sensitivity assay (hsCRP).
In a test of its independence as a prognosticator, HDL anti-inflammatory capacity significantly and meaningfully improved prediction of CV events in the study after it was added to the familiar Framingham risk equations.
Measurement of HDL anti-inflammatory properties, therefore, has the potential to improve current CV risk assessments in people without clinical heart disease, the authors proposed.
The study “adds to our understanding of the potential cardioprotective role of HDL,” Michael Miller, MD, University of Maryland, Baltimore, said in an interview.
“We’ve known for some time that HDL has anti-inflammatory properties in vitro, and my understanding is this is the first study to assess these anti-inflammatory properties in a clinical trial,” said Dr. Miller, who studies lipid metabolism and directs the Center for Preventive Cardiology at his center but isn’t an author of the report.
The study is part of a long line of research aiming to “untangle the complexities of HDL and try to get a better handle as to the properties that make it cardioprotective,” he said. For example, “high levels are not always associated with cardioprotection, and low levels don’t always imply increased risk.”
The current findings highlight a quality of HDL that might be prognostic but also independent of its concentrations, apoA1 content, or cholesterol efflux capacity, Dr. Miller noted. That makes HDL anti-inflammatory capacity a “promising feature” of HDL that, if confirmed in further studies, could potentially be brought into the mainstream for CV risk prediction. “But it’s too premature at this time.”
The study of participants in the population-based PREVEND cohort study compared 340 patients with a first CV event – CV death, ischemic heart disease, nonfatal MI, or coronary revascularization – over a median of about 10 years with the same number of participants without such events. The two cohorts of people from the same city in the Netherlands had been matched according to sex, smoking status, age, and HDL cholesterol levels at baseline.
No measured clinical or laboratory value, the group wrote, was significantly correlated with HDL anti-inflammatory capacity, defined here as ability to suppress vascular cell adhesion molecule-1 (VCAM-1) mRNA expression as induced by tumor necrosis factor–alpha in endothelial cells in vitro.
HDL anti-inflammatory capacity was significantly lower in the case cohort, compared with the control cohort (P < .001), and was inversely related to new CV events, at an odds ratio per 1 standard deviation of 0.74 (95% confidence interval, 0.61-0.90; P = .002). Covariate adjustments included body mass index; alcohol intake; diabetes and hypertension status; use of lipid-lowering medicine; levels of total cholesterol, apoA1, triglyceride, and hsCRP; and measures of renal function.
No significant association was seen between HDL anti-inflammatory capacity and cholesterol efflux capacity (coefficient of correlation, −0.02; P > .05). But both metrics were independently associated with CV disease events. The OR per 1 standard deviation was 0.74 (95% CI, 0.61-0.90; P = .002) for cholesterol efflux capacity and 0.66 (95% CI, 0.54-0.81; P < .001) for HDL anti-inflammatory capacity.
Adding HDL anti-inflammatory capacity to the Framingham risk score significantly improved its predictive power; its likelihood-ratio statistic rose from 10.50 to 20.40 (P = .002), the group wrote. The addition of cholesterol efflux capacity further elevated the risk score’s likelihood-ratio statistic to 32.84 (P = .0005).
The analysis has all the limitations of a case-control study, Dr. Miller said, but it does “show a potential reasonable association” between anti-inflammatory capacity and CV risk “that needs to be taken to the next level.”
For example, it could be explored in a controlled trial that tracks anti-inflammatory capacity in individuals who receive an intervention that is likely to improve the biomarker – such as weight loss, he proposed – and follows them for clinical outcomes.
“If you want to elevate the stature of the anti-inflammatory index,” Dr. Miller said, “you will need to show that it’s clinically meaningful.”
Dr. Jia reported no conflicts. Dr. Miller has no relevant disclosures.
A version of this article first appeared on Medscape.com.
Remote cardio visits expand access for underserved during COVID
Remote cardiology clinic visits during COVID-19 were used more often by certain traditionally underserved patient groups, but were also associated with less frequent testing and prescribing, new research shows.
“The COVID-19 pandemic has led to an unprecedented shift in ambulatory cardiovascular care from in-person to remote visits,” lead author Neal Yuan, MD, a cardiology fellow at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
Their findings were published online April 5 in JAMA Network Open.
“We wanted to explore whether the transition to remote visits was associated with disparities in how patients accessed care, and also how this transition affected diagnostic test ordering and medication prescribing,” Dr. Yuan said.
The researchers used electronic health records data for all ambulatory cardiology visits at an urban, multisite health system in Los Angeles County during two periods: April 1 to Dec. 31, 2019, the pre-COVID era; and April 1 to Dec. 31, 2020, the COVID era.
The investigators compared patient characteristics and frequencies of medication ordering and cardiology-specific testing across four visit types: pre-COVID in person, used as reference; COVID-era in person; COVID-era video; and COVID-era telephone.
The study looked at 176,781 ambulatory cardiology visits. Of these visits, 87,182 were conducted in person in the pre-COVID period; 74,498 were conducted in person in the COVID era; 4,720 were COVID-era video visits; and 10,381 were COVID-era telephone visits.
In the study cohort, 79,572 patients (45.0%) were female, 127,080 patients (71.9%) were non-Hispanic White, and the mean age was 68.1 years (standard deviation, 17.0).
Patients accessing COVID-era remote visits were more likely to be Asian, Black, or Hispanic, to have private insurance, and to have cardiovascular comorbidities, such as hypertension and heart failure.
Also, patients whose visits were conducted by video were significantly younger than patients whose visits were conducted in person or by telephone (P < .001).
In addition, the study found that clinicians ordered fewer diagnostic tests, such as electrocardiograms and echocardiograms, and were less likely to order any medication, in the pre-COVID era than during the COVID era.
“If you don’t have a patient in front of you, it’s much more difficult to get a physical exam or obtain reliable vital signs,” said Dr. Yuan. Communication can sometimes be difficult, often because of technical issues, like a bad connection. “You might be more reticent to get testing or to prescribe medications if you don’t feel confident knowing what the patient’s vital signs are.”
In addition, he added, “a lot of medications used in the cardiology setting require monitoring patients’ kidney function and electrolytes, and if you can’t do that reliably, you might be more cautious about prescribing those types of medications.”
An eye-opening study
Cardiologist Nieca Goldberg, MD, medical director of the New York University Langone womens’ heart program and spokesperson for the American Heart Association, recounted her experience with telemedicine at the height of the pandemic in New York, when everything, including medical outpatient offices, had to close.
“We were experienced with telemedicine because we had started a virtual urgent care program well ahead of the pandemic,” she said. “We started using that to screen people with potential COVID symptoms so that they wouldn’t have to come into the hospital, the medical center, or to the offices and expose people. We learned that it was great to have the telemedicine option from the infectious disease standpoint, and I did visits like that for my own patient population.”
An equally if not more important finding from the study is the fact that telemedicine increased access to care among traditionally underserved demographics, she said.
“This is eye-opening, that you can actually improve access to care by doing telemedicine visits. It was really important to see that telemedicine has added benefit to the way we can see people in the health care system.”
Telemedicine visits had a positive impact at a time when people were isolated at home, Dr. Goldberg said.
“It was a way for them to connect with their doctor and in some ways it was more personal,” she added. “I actually got to meet some of my patients’ family members. It was like making a remote house call.”
Stable cardiology patients can take their blood pressure at home, weigh themselves, and take their own pulse to give an excellent set of vital signs that will indicate how they are doing, said Dr. Goldberg.
“During a remote visit, we can talk to the patient and notice whether or not they are short of breath or coughing, but we can’t listen to their heart or do an EKG or any of the traditional cardiac testing. Still, for someone who is not having symptoms and is able to reliably monitor their blood pressure and weight, a remote visit is sufficient to give you a good sense of how that patient is doing,” she said. “We can talk to them about their medications, any potential side effects, and we can use their blood pressure information to adjust their medications.”
Many patients are becoming more savvy about using tech gadgets and devices to monitor their health.
“Some of my patients were using Apple watches and the Kardia app to address their heart rate. Many had purchased inexpensive pulse oximeters to check their oxygen during the pandemic, and that also reads the pulse,” Dr. Goldberg said.
In-person visits were reserved for symptomatic cardiac patients, she explained.
“Initially during the pandemic, we did mostly telemedicine visits and we organized the office so that each cardiologist would come in 1 day a week to take care of symptomatic cardiac patients. In that way, we were able to socially distance – they provided us with [personal protective equipment]; at NYU there was no problem with that – and nobody waited in the waiting room. To this day, office issues are more efficient and people are not waiting in the waiting room,” she added. “Telemedicine improves access to health care in populations where such access is limited.”
Dr. Yuan’s research is supported by a grant from the National Institutes of Health. Dr. Goldberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Remote cardiology clinic visits during COVID-19 were used more often by certain traditionally underserved patient groups, but were also associated with less frequent testing and prescribing, new research shows.
“The COVID-19 pandemic has led to an unprecedented shift in ambulatory cardiovascular care from in-person to remote visits,” lead author Neal Yuan, MD, a cardiology fellow at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
Their findings were published online April 5 in JAMA Network Open.
“We wanted to explore whether the transition to remote visits was associated with disparities in how patients accessed care, and also how this transition affected diagnostic test ordering and medication prescribing,” Dr. Yuan said.
The researchers used electronic health records data for all ambulatory cardiology visits at an urban, multisite health system in Los Angeles County during two periods: April 1 to Dec. 31, 2019, the pre-COVID era; and April 1 to Dec. 31, 2020, the COVID era.
The investigators compared patient characteristics and frequencies of medication ordering and cardiology-specific testing across four visit types: pre-COVID in person, used as reference; COVID-era in person; COVID-era video; and COVID-era telephone.
The study looked at 176,781 ambulatory cardiology visits. Of these visits, 87,182 were conducted in person in the pre-COVID period; 74,498 were conducted in person in the COVID era; 4,720 were COVID-era video visits; and 10,381 were COVID-era telephone visits.
In the study cohort, 79,572 patients (45.0%) were female, 127,080 patients (71.9%) were non-Hispanic White, and the mean age was 68.1 years (standard deviation, 17.0).
Patients accessing COVID-era remote visits were more likely to be Asian, Black, or Hispanic, to have private insurance, and to have cardiovascular comorbidities, such as hypertension and heart failure.
Also, patients whose visits were conducted by video were significantly younger than patients whose visits were conducted in person or by telephone (P < .001).
In addition, the study found that clinicians ordered fewer diagnostic tests, such as electrocardiograms and echocardiograms, and were less likely to order any medication, in the pre-COVID era than during the COVID era.
“If you don’t have a patient in front of you, it’s much more difficult to get a physical exam or obtain reliable vital signs,” said Dr. Yuan. Communication can sometimes be difficult, often because of technical issues, like a bad connection. “You might be more reticent to get testing or to prescribe medications if you don’t feel confident knowing what the patient’s vital signs are.”
In addition, he added, “a lot of medications used in the cardiology setting require monitoring patients’ kidney function and electrolytes, and if you can’t do that reliably, you might be more cautious about prescribing those types of medications.”
An eye-opening study
Cardiologist Nieca Goldberg, MD, medical director of the New York University Langone womens’ heart program and spokesperson for the American Heart Association, recounted her experience with telemedicine at the height of the pandemic in New York, when everything, including medical outpatient offices, had to close.
“We were experienced with telemedicine because we had started a virtual urgent care program well ahead of the pandemic,” she said. “We started using that to screen people with potential COVID symptoms so that they wouldn’t have to come into the hospital, the medical center, or to the offices and expose people. We learned that it was great to have the telemedicine option from the infectious disease standpoint, and I did visits like that for my own patient population.”
An equally if not more important finding from the study is the fact that telemedicine increased access to care among traditionally underserved demographics, she said.
“This is eye-opening, that you can actually improve access to care by doing telemedicine visits. It was really important to see that telemedicine has added benefit to the way we can see people in the health care system.”
Telemedicine visits had a positive impact at a time when people were isolated at home, Dr. Goldberg said.
“It was a way for them to connect with their doctor and in some ways it was more personal,” she added. “I actually got to meet some of my patients’ family members. It was like making a remote house call.”
Stable cardiology patients can take their blood pressure at home, weigh themselves, and take their own pulse to give an excellent set of vital signs that will indicate how they are doing, said Dr. Goldberg.
“During a remote visit, we can talk to the patient and notice whether or not they are short of breath or coughing, but we can’t listen to their heart or do an EKG or any of the traditional cardiac testing. Still, for someone who is not having symptoms and is able to reliably monitor their blood pressure and weight, a remote visit is sufficient to give you a good sense of how that patient is doing,” she said. “We can talk to them about their medications, any potential side effects, and we can use their blood pressure information to adjust their medications.”
Many patients are becoming more savvy about using tech gadgets and devices to monitor their health.
“Some of my patients were using Apple watches and the Kardia app to address their heart rate. Many had purchased inexpensive pulse oximeters to check their oxygen during the pandemic, and that also reads the pulse,” Dr. Goldberg said.
In-person visits were reserved for symptomatic cardiac patients, she explained.
“Initially during the pandemic, we did mostly telemedicine visits and we organized the office so that each cardiologist would come in 1 day a week to take care of symptomatic cardiac patients. In that way, we were able to socially distance – they provided us with [personal protective equipment]; at NYU there was no problem with that – and nobody waited in the waiting room. To this day, office issues are more efficient and people are not waiting in the waiting room,” she added. “Telemedicine improves access to health care in populations where such access is limited.”
Dr. Yuan’s research is supported by a grant from the National Institutes of Health. Dr. Goldberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Remote cardiology clinic visits during COVID-19 were used more often by certain traditionally underserved patient groups, but were also associated with less frequent testing and prescribing, new research shows.
“The COVID-19 pandemic has led to an unprecedented shift in ambulatory cardiovascular care from in-person to remote visits,” lead author Neal Yuan, MD, a cardiology fellow at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
Their findings were published online April 5 in JAMA Network Open.
“We wanted to explore whether the transition to remote visits was associated with disparities in how patients accessed care, and also how this transition affected diagnostic test ordering and medication prescribing,” Dr. Yuan said.
The researchers used electronic health records data for all ambulatory cardiology visits at an urban, multisite health system in Los Angeles County during two periods: April 1 to Dec. 31, 2019, the pre-COVID era; and April 1 to Dec. 31, 2020, the COVID era.
The investigators compared patient characteristics and frequencies of medication ordering and cardiology-specific testing across four visit types: pre-COVID in person, used as reference; COVID-era in person; COVID-era video; and COVID-era telephone.
The study looked at 176,781 ambulatory cardiology visits. Of these visits, 87,182 were conducted in person in the pre-COVID period; 74,498 were conducted in person in the COVID era; 4,720 were COVID-era video visits; and 10,381 were COVID-era telephone visits.
In the study cohort, 79,572 patients (45.0%) were female, 127,080 patients (71.9%) were non-Hispanic White, and the mean age was 68.1 years (standard deviation, 17.0).
Patients accessing COVID-era remote visits were more likely to be Asian, Black, or Hispanic, to have private insurance, and to have cardiovascular comorbidities, such as hypertension and heart failure.
Also, patients whose visits were conducted by video were significantly younger than patients whose visits were conducted in person or by telephone (P < .001).
In addition, the study found that clinicians ordered fewer diagnostic tests, such as electrocardiograms and echocardiograms, and were less likely to order any medication, in the pre-COVID era than during the COVID era.
“If you don’t have a patient in front of you, it’s much more difficult to get a physical exam or obtain reliable vital signs,” said Dr. Yuan. Communication can sometimes be difficult, often because of technical issues, like a bad connection. “You might be more reticent to get testing or to prescribe medications if you don’t feel confident knowing what the patient’s vital signs are.”
In addition, he added, “a lot of medications used in the cardiology setting require monitoring patients’ kidney function and electrolytes, and if you can’t do that reliably, you might be more cautious about prescribing those types of medications.”
An eye-opening study
Cardiologist Nieca Goldberg, MD, medical director of the New York University Langone womens’ heart program and spokesperson for the American Heart Association, recounted her experience with telemedicine at the height of the pandemic in New York, when everything, including medical outpatient offices, had to close.
“We were experienced with telemedicine because we had started a virtual urgent care program well ahead of the pandemic,” she said. “We started using that to screen people with potential COVID symptoms so that they wouldn’t have to come into the hospital, the medical center, or to the offices and expose people. We learned that it was great to have the telemedicine option from the infectious disease standpoint, and I did visits like that for my own patient population.”
An equally if not more important finding from the study is the fact that telemedicine increased access to care among traditionally underserved demographics, she said.
“This is eye-opening, that you can actually improve access to care by doing telemedicine visits. It was really important to see that telemedicine has added benefit to the way we can see people in the health care system.”
Telemedicine visits had a positive impact at a time when people were isolated at home, Dr. Goldberg said.
“It was a way for them to connect with their doctor and in some ways it was more personal,” she added. “I actually got to meet some of my patients’ family members. It was like making a remote house call.”
Stable cardiology patients can take their blood pressure at home, weigh themselves, and take their own pulse to give an excellent set of vital signs that will indicate how they are doing, said Dr. Goldberg.
“During a remote visit, we can talk to the patient and notice whether or not they are short of breath or coughing, but we can’t listen to their heart or do an EKG or any of the traditional cardiac testing. Still, for someone who is not having symptoms and is able to reliably monitor their blood pressure and weight, a remote visit is sufficient to give you a good sense of how that patient is doing,” she said. “We can talk to them about their medications, any potential side effects, and we can use their blood pressure information to adjust their medications.”
Many patients are becoming more savvy about using tech gadgets and devices to monitor their health.
“Some of my patients were using Apple watches and the Kardia app to address their heart rate. Many had purchased inexpensive pulse oximeters to check their oxygen during the pandemic, and that also reads the pulse,” Dr. Goldberg said.
In-person visits were reserved for symptomatic cardiac patients, she explained.
“Initially during the pandemic, we did mostly telemedicine visits and we organized the office so that each cardiologist would come in 1 day a week to take care of symptomatic cardiac patients. In that way, we were able to socially distance – they provided us with [personal protective equipment]; at NYU there was no problem with that – and nobody waited in the waiting room. To this day, office issues are more efficient and people are not waiting in the waiting room,” she added. “Telemedicine improves access to health care in populations where such access is limited.”
Dr. Yuan’s research is supported by a grant from the National Institutes of Health. Dr. Goldberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Novel antiplatelet drug: Hope for efficacy without bleeding?
A new antiplatelet drug with a completely novel mechanism of action may hold the promise of delivering the holy grail – reducing cardiac events without increasing bleeding. That is the hope behind the new class of drugs directed against the platelet collagen glycoprotein VI (GPVI) receptor.
A phase 2 trial with the first agent in this class, known as revacept (advanceCOR), showed no increase in bleeding with the product when added to standard dual-antiplatelet therapy for patients with stable ischemic heart disease undergoing elective percutaneous coronary intervention (PCI), despite the drug’s being used at a dose that has been shown to increase platelet inhibition.
Unfortunately, there was no reduction in the primary clinical efficacy endpoint, a myocardial injury surrogate, but the authors pointed out that the overall event rate was low, and they were hopeful that future trials in a higher-risk population will show efficacy.
The ISAR PLASTER study was published online on March 31 in JAMA Cardiology.
“This new drug is targeting the collagen in the extracellular matrix of atherosclerotic plaque rather than the platelets themselves. So, in theory, this agent should not cause an increase in bleeding,” study author Steffen Massberg, DrMed, said in an interview.
Dr. Massberg explained that revacept targets the binding site for platelets on collagen that is exposed on rupture of atherosclerotic plaques and is a major trigger of platelet activation.
“In contrast to aspirin and P2Y12 inhibitors, which target all platelets, revacept only binds to sites where there is ruptured plaque. But the platelets themselves otherwise have normal function, so regular coagulation processes should be unaffected,” he commented.
“While collagen also has a role in the coagulation process, it is more involved in atherosclerotic plaque rupture, and in animal studies, revacept was effective in preventing clot formation in large arteries but only had a small effect on bleeding,” Dr. Massberg added.
In the JAMA Cardiology article, the authors further elaborated that, when collagen is exposed during atherosclerotic plaque rupture, it binds platelet GPVI, the major platelet collagen receptor.
“Glycoprotein VI in turn mediates local platelet recruitment, activation, and aggregation. Glycoprotein VI is an attractive antiplatelet target because GPVI-mediated platelet response plays a central role during myocardial infarction and stroke but is less relevant in physiological hemostasis,” they wrote.
The researchers describe revacept as a dimeric, soluble fusion protein composed of the extracellular domain of the GPVI receptor and the human Fc-fragment. It competes with endogenous platelet GPVI for binding to exposed collagen fibers and inhibits collagen-mediated platelet adhesion and aggregation selectively at the site of plaque rupture.
In addition, revacept blocks binding of von Willebrand factor to collagen and inhibits von Willebrand factor–mediated platelet activation, they reported.
“As a lesion-directed drug, revacept does not interfere with the function of circulating platelets beyond the atherosclerotic lesion,” the authors said.
In animal studies and a phase 1 clinical trial, the drug was shown to inhibit atherothrombosis but to have little effect on systemic hemostasis or bleeding.
The current ISAR-PLASTER trial is the first study of the use of the agent for patients with coronary heart disease.
For the study, 334 patients with stable ischemic heart disease undergoing elective PCI were randomly assigned to receive a single intravenous infusion of revacept 160 mg, revacept 80 mg, or placebo prior to the start of PCI in addition to standard antithrombotic therapy.
The safety endpoint was bleeding of type 2-5, per Bleeding Academic Research Consortium (BARC) criteria, at 30 days.
Results showed no significant differences in the primary efficacy endpoint (the composite of death or myocardial injury, defined as an increase in high-sensitivity cardiac troponin T [hsTnT] to at least five times the upper limit of normal within 48 hours from randomization) between the revacept and placebo groups. The primary efficacy endpoint occurred in 24.4% of the revacept 160-mg group, 25.0% of the revacept 80-mg group, and 23.3% of the placebo group.
The high dose of revacept was associated with a small but significant reduction of high-concentration collagen-induced platelet aggregation, but adenosine 5-diphosphate–induced aggregation was not affected.
Revacept did not increase bleeding. Bleeding of BARC type 2 or higher at 30 days occurred in 5.0% of the 160-mg group, 5.9% of the 80-mg group, and 8.6% of the placebo group.
Dr. Massberg pointed out that one possible explanation for the lack of difference in the efficacy outcome was that the patients enrolled in the study were at low risk.
“The rate of major adverse cardiovascular events was very low (2.5% at 30 days), and this was a low-risk population undergoing elective PCI,” he commented.
The authors also pointed out that the five-times increase in hsTnT endpoint used in the current study has little prognostic impact.
In addition, Dr. Massberg noted that, in the stable situation, myocardial injury is mostly triggered by cholesterol embolism during PCI and side-branch occlusion due to distal plaque embolization, problems that are unlikely to respond to inhibition of GPVI-collagen interaction by revacept.
He suggested that better results may be achieved in patients with acute coronary syndrome (ACS). “In ACS patients, the myocardial injury is caused by ongoing thrombotic cascades, where the collagen-platelet interaction plays a much larger role, so in theory, this drug should show a greater effect in an ACS population.”
The researchers are now planning a larger phase 3 study in that group.
“I am still optimistic. I still believe it could work,” Dr. Massberg said. “The major aim for this study was safety and dosing. There was no difference in bleeding, so safety was supported,” he added.
The ISAR-PLASTER study was funded by the German Center for Cardiovascular Research, Deutsches Herzzentrum Munchen, the Federal Ministry of Education and Research, and advanceCOR (the manufacturer of revacept). One of the coauthors of the study is a cofounder of advanceCor.
A version of this article first appeared on Medscape.com.
A new antiplatelet drug with a completely novel mechanism of action may hold the promise of delivering the holy grail – reducing cardiac events without increasing bleeding. That is the hope behind the new class of drugs directed against the platelet collagen glycoprotein VI (GPVI) receptor.
A phase 2 trial with the first agent in this class, known as revacept (advanceCOR), showed no increase in bleeding with the product when added to standard dual-antiplatelet therapy for patients with stable ischemic heart disease undergoing elective percutaneous coronary intervention (PCI), despite the drug’s being used at a dose that has been shown to increase platelet inhibition.
Unfortunately, there was no reduction in the primary clinical efficacy endpoint, a myocardial injury surrogate, but the authors pointed out that the overall event rate was low, and they were hopeful that future trials in a higher-risk population will show efficacy.
The ISAR PLASTER study was published online on March 31 in JAMA Cardiology.
“This new drug is targeting the collagen in the extracellular matrix of atherosclerotic plaque rather than the platelets themselves. So, in theory, this agent should not cause an increase in bleeding,” study author Steffen Massberg, DrMed, said in an interview.
Dr. Massberg explained that revacept targets the binding site for platelets on collagen that is exposed on rupture of atherosclerotic plaques and is a major trigger of platelet activation.
“In contrast to aspirin and P2Y12 inhibitors, which target all platelets, revacept only binds to sites where there is ruptured plaque. But the platelets themselves otherwise have normal function, so regular coagulation processes should be unaffected,” he commented.
“While collagen also has a role in the coagulation process, it is more involved in atherosclerotic plaque rupture, and in animal studies, revacept was effective in preventing clot formation in large arteries but only had a small effect on bleeding,” Dr. Massberg added.
In the JAMA Cardiology article, the authors further elaborated that, when collagen is exposed during atherosclerotic plaque rupture, it binds platelet GPVI, the major platelet collagen receptor.
“Glycoprotein VI in turn mediates local platelet recruitment, activation, and aggregation. Glycoprotein VI is an attractive antiplatelet target because GPVI-mediated platelet response plays a central role during myocardial infarction and stroke but is less relevant in physiological hemostasis,” they wrote.
The researchers describe revacept as a dimeric, soluble fusion protein composed of the extracellular domain of the GPVI receptor and the human Fc-fragment. It competes with endogenous platelet GPVI for binding to exposed collagen fibers and inhibits collagen-mediated platelet adhesion and aggregation selectively at the site of plaque rupture.
In addition, revacept blocks binding of von Willebrand factor to collagen and inhibits von Willebrand factor–mediated platelet activation, they reported.
“As a lesion-directed drug, revacept does not interfere with the function of circulating platelets beyond the atherosclerotic lesion,” the authors said.
In animal studies and a phase 1 clinical trial, the drug was shown to inhibit atherothrombosis but to have little effect on systemic hemostasis or bleeding.
The current ISAR-PLASTER trial is the first study of the use of the agent for patients with coronary heart disease.
For the study, 334 patients with stable ischemic heart disease undergoing elective PCI were randomly assigned to receive a single intravenous infusion of revacept 160 mg, revacept 80 mg, or placebo prior to the start of PCI in addition to standard antithrombotic therapy.
The safety endpoint was bleeding of type 2-5, per Bleeding Academic Research Consortium (BARC) criteria, at 30 days.
Results showed no significant differences in the primary efficacy endpoint (the composite of death or myocardial injury, defined as an increase in high-sensitivity cardiac troponin T [hsTnT] to at least five times the upper limit of normal within 48 hours from randomization) between the revacept and placebo groups. The primary efficacy endpoint occurred in 24.4% of the revacept 160-mg group, 25.0% of the revacept 80-mg group, and 23.3% of the placebo group.
The high dose of revacept was associated with a small but significant reduction of high-concentration collagen-induced platelet aggregation, but adenosine 5-diphosphate–induced aggregation was not affected.
Revacept did not increase bleeding. Bleeding of BARC type 2 or higher at 30 days occurred in 5.0% of the 160-mg group, 5.9% of the 80-mg group, and 8.6% of the placebo group.
Dr. Massberg pointed out that one possible explanation for the lack of difference in the efficacy outcome was that the patients enrolled in the study were at low risk.
“The rate of major adverse cardiovascular events was very low (2.5% at 30 days), and this was a low-risk population undergoing elective PCI,” he commented.
The authors also pointed out that the five-times increase in hsTnT endpoint used in the current study has little prognostic impact.
In addition, Dr. Massberg noted that, in the stable situation, myocardial injury is mostly triggered by cholesterol embolism during PCI and side-branch occlusion due to distal plaque embolization, problems that are unlikely to respond to inhibition of GPVI-collagen interaction by revacept.
He suggested that better results may be achieved in patients with acute coronary syndrome (ACS). “In ACS patients, the myocardial injury is caused by ongoing thrombotic cascades, where the collagen-platelet interaction plays a much larger role, so in theory, this drug should show a greater effect in an ACS population.”
The researchers are now planning a larger phase 3 study in that group.
“I am still optimistic. I still believe it could work,” Dr. Massberg said. “The major aim for this study was safety and dosing. There was no difference in bleeding, so safety was supported,” he added.
The ISAR-PLASTER study was funded by the German Center for Cardiovascular Research, Deutsches Herzzentrum Munchen, the Federal Ministry of Education and Research, and advanceCOR (the manufacturer of revacept). One of the coauthors of the study is a cofounder of advanceCor.
A version of this article first appeared on Medscape.com.
A new antiplatelet drug with a completely novel mechanism of action may hold the promise of delivering the holy grail – reducing cardiac events without increasing bleeding. That is the hope behind the new class of drugs directed against the platelet collagen glycoprotein VI (GPVI) receptor.
A phase 2 trial with the first agent in this class, known as revacept (advanceCOR), showed no increase in bleeding with the product when added to standard dual-antiplatelet therapy for patients with stable ischemic heart disease undergoing elective percutaneous coronary intervention (PCI), despite the drug’s being used at a dose that has been shown to increase platelet inhibition.
Unfortunately, there was no reduction in the primary clinical efficacy endpoint, a myocardial injury surrogate, but the authors pointed out that the overall event rate was low, and they were hopeful that future trials in a higher-risk population will show efficacy.
The ISAR PLASTER study was published online on March 31 in JAMA Cardiology.
“This new drug is targeting the collagen in the extracellular matrix of atherosclerotic plaque rather than the platelets themselves. So, in theory, this agent should not cause an increase in bleeding,” study author Steffen Massberg, DrMed, said in an interview.
Dr. Massberg explained that revacept targets the binding site for platelets on collagen that is exposed on rupture of atherosclerotic plaques and is a major trigger of platelet activation.
“In contrast to aspirin and P2Y12 inhibitors, which target all platelets, revacept only binds to sites where there is ruptured plaque. But the platelets themselves otherwise have normal function, so regular coagulation processes should be unaffected,” he commented.
“While collagen also has a role in the coagulation process, it is more involved in atherosclerotic plaque rupture, and in animal studies, revacept was effective in preventing clot formation in large arteries but only had a small effect on bleeding,” Dr. Massberg added.
In the JAMA Cardiology article, the authors further elaborated that, when collagen is exposed during atherosclerotic plaque rupture, it binds platelet GPVI, the major platelet collagen receptor.
“Glycoprotein VI in turn mediates local platelet recruitment, activation, and aggregation. Glycoprotein VI is an attractive antiplatelet target because GPVI-mediated platelet response plays a central role during myocardial infarction and stroke but is less relevant in physiological hemostasis,” they wrote.
The researchers describe revacept as a dimeric, soluble fusion protein composed of the extracellular domain of the GPVI receptor and the human Fc-fragment. It competes with endogenous platelet GPVI for binding to exposed collagen fibers and inhibits collagen-mediated platelet adhesion and aggregation selectively at the site of plaque rupture.
In addition, revacept blocks binding of von Willebrand factor to collagen and inhibits von Willebrand factor–mediated platelet activation, they reported.
“As a lesion-directed drug, revacept does not interfere with the function of circulating platelets beyond the atherosclerotic lesion,” the authors said.
In animal studies and a phase 1 clinical trial, the drug was shown to inhibit atherothrombosis but to have little effect on systemic hemostasis or bleeding.
The current ISAR-PLASTER trial is the first study of the use of the agent for patients with coronary heart disease.
For the study, 334 patients with stable ischemic heart disease undergoing elective PCI were randomly assigned to receive a single intravenous infusion of revacept 160 mg, revacept 80 mg, or placebo prior to the start of PCI in addition to standard antithrombotic therapy.
The safety endpoint was bleeding of type 2-5, per Bleeding Academic Research Consortium (BARC) criteria, at 30 days.
Results showed no significant differences in the primary efficacy endpoint (the composite of death or myocardial injury, defined as an increase in high-sensitivity cardiac troponin T [hsTnT] to at least five times the upper limit of normal within 48 hours from randomization) between the revacept and placebo groups. The primary efficacy endpoint occurred in 24.4% of the revacept 160-mg group, 25.0% of the revacept 80-mg group, and 23.3% of the placebo group.
The high dose of revacept was associated with a small but significant reduction of high-concentration collagen-induced platelet aggregation, but adenosine 5-diphosphate–induced aggregation was not affected.
Revacept did not increase bleeding. Bleeding of BARC type 2 or higher at 30 days occurred in 5.0% of the 160-mg group, 5.9% of the 80-mg group, and 8.6% of the placebo group.
Dr. Massberg pointed out that one possible explanation for the lack of difference in the efficacy outcome was that the patients enrolled in the study were at low risk.
“The rate of major adverse cardiovascular events was very low (2.5% at 30 days), and this was a low-risk population undergoing elective PCI,” he commented.
The authors also pointed out that the five-times increase in hsTnT endpoint used in the current study has little prognostic impact.
In addition, Dr. Massberg noted that, in the stable situation, myocardial injury is mostly triggered by cholesterol embolism during PCI and side-branch occlusion due to distal plaque embolization, problems that are unlikely to respond to inhibition of GPVI-collagen interaction by revacept.
He suggested that better results may be achieved in patients with acute coronary syndrome (ACS). “In ACS patients, the myocardial injury is caused by ongoing thrombotic cascades, where the collagen-platelet interaction plays a much larger role, so in theory, this drug should show a greater effect in an ACS population.”
The researchers are now planning a larger phase 3 study in that group.
“I am still optimistic. I still believe it could work,” Dr. Massberg said. “The major aim for this study was safety and dosing. There was no difference in bleeding, so safety was supported,” he added.
The ISAR-PLASTER study was funded by the German Center for Cardiovascular Research, Deutsches Herzzentrum Munchen, the Federal Ministry of Education and Research, and advanceCOR (the manufacturer of revacept). One of the coauthors of the study is a cofounder of advanceCor.
A version of this article first appeared on Medscape.com.
Risk for erectile dysfunction sixfold higher in men with COVID-19
COVID-19 increases the risk of developing erectile dysfunction (ED) by nearly sixfold, according to data from the first study to investigate the association between ED and COVID-19 in young men in a real-life setting.
Men with ED are more than five times more likely to have COVID-19 (odds ratio, 5.27).
For men with a history of COVID-19, the odds ratio of developing ED was 5.66. The strength of the association remained after adjusting for factors considered to affect ED.
The study, which was led by Emmanuele A. Jannini, MD, professor of endocrinology and medical sexology, University of Rome Tor Vergata, was published on March 20 in Andrology.
‘Mask up to keep it up’
ED can be both a short-term and a long-term complication of COVID-19, Dr. Jannini suggests.
“When offered, men should have the COVID vaccination. It also gives a whole new meaning to wearing the mask – mask up to keep it up,” he said. “It could possibly have the added benefit of preventing sexual dysfunction.”
He points out that older age, diabetes, high body mass index, and smoking increase the risk of contracting COVID-19.
“These are the same as risk factors for ED. Results of our study agree with the pathophysiological mechanisms linking ED, endothelial dysfunction, and COVID-19. Basically, endothelial dysfunction is common in both conditions [COVID-10 and ED].
“We would like to find some sort of biomarker of endothelial dysfunction post COVID, because it seems that there are many sequelae that coexist for a long time after infection,” added Dr. Jannini. “Asking a patient if they have ED after COVID might provide a measure of systemic wellness.”
Allan Pacey, MD, professor of andrology at the University of Sheffield (England), welcomed the research, noting, “This seems to be a well-conducted study. However, at the moment, the relationship is just a correlation, and it might be that some of the comorbidities that increased the men’s chances of getting a significant COVID-19 infection may have also independently increased their chances of erectile dysfunction.
“But the authors offer a plausible mechanism by which COVID-19 may impact directly on erectile function,” agrees Dr. Pacey. However, “There’s more work to be done,” he said. “I’d also argue it’s a good reason for men to wear a mask, practice social distancing, and take the vaccine when it’s offered to them.”
Urologist John Mulhall, MD, from Memorial Sloan Kettering Cancer Center, New York, remarked, “It was a highly preliminary study, but the data are suggestive of a potential link between COVID-19 infection and ED.
“However, it raises enough questions such that further large, more long-term analyses are required to define causation. Future studies assessing testosterone levels and erectile hemodynamics will be needed to provide definite evidence of a causative link,» he stressed.
Erectile problems a ‘hallmark’ of systemic endothelial dysfunction
Prior research has suggested that asymptomatic COVID-19 could be associated with subclinical microvascular involvement with long-term cardiovascular effects.
“Indeed, COVID-19 is by all means an endothelial disease, in which systemic manifestations ... can potentially be due to alterations in the endothelial thrombotic/fibrinolytic balance,” emphasized Dr. Jannini. “In addition, endothelial cells express many of the cofactors used by SARS-CoV-2 to invade host cells.
“Erectile dysfunction has often been considered a hallmark of endothelial dysfunction, and as such, a potential association between ED and COVID-19 has also been postulated and underpinned the investigation in this study,” he explained.
The study was predicated on the fact that ED is often considered a clinical marker of impaired overall health status, which often features cardiovascular events at an early age. It aimed to investigate the bidirectional relationship between COVID-19 and ED. It asked whether ED could be a risk factor for contracting COVID-19 and whether having COVID-19 predisposes to developing ED.
“This would possibly suggest that men with ED, due to the underlying conditions which impair erectile response, could also be more susceptible to contracting COVID-19,” said Dr. Jannini.
Data were drawn from the Sex@COVID online survey, which was conducted from April 7 to May 4, 2020, in Italy. The survey included 6,821 participants aged 18 years or older (4,177 women; 2,644 men; mean age, 32.83 ± 11.24 years). Participants were stratified on the basis of marital status and sexual activity during lockdown. From these participants, 985 sexually active men were identified, among whom 25 (2.54%) reported having tested positive for COVID-19. These persons were matched with 75 COVID-19–negative men using propensity score matching in a 1:3 ratio.
The researchers used standardized psychometric tools to measure the effects of lockdown and social distancing on the intrapsychic, relational, and sexual health of the participants.
Erectile function was measured with the International Index of Erectile Function or the Sexual Health Inventory for Men, which are often used in clinical settings. In light of the two-way interaction between sexual activity and psychological well-being, results were adjusted for any influence of anxiety and depression, which were measured with recognized scales for use in patients with a history of COVID-19.
Results showed that the prevalence of ED was significantly higher among men who self-reported a history of COVID-19, compared with a matching COVID-negative population (28% vs. 9.33%; P = .027).
After adjusting for variables that are considered to have a bearing on the development of ED, such as psychological status, age, and BMI, the odds ratio for developing ED after having had COVID-19 was 5.66 (95% confidence interval, 1.50-24.01).
Similarly, after adjusting for age and BMI, men with ED were more likely to have COVID‐19 (OR, 5.27; 95% CI, 1.49-20.09).
The authors note that persons who experience “a sudden onset or worsening of ED might also consider precautionary quarantine or nasopharyngeal swab, as COVID‐19 might act as a potential initiating trigger for the onset of erectile impairment, or an aggravating factor for its progression to more severe forms.”
Similarly, patients who have ED “should consider their erectile impairment as a sign of possible underlying conditions that could increase the likelihood of suffering from COVID‐19,” they write.
Dr. Mulhall highlighted several limitations of the study, including its retrospective nature, recall bias associated with the use of online questionnaires, and the inclusion of COVID‐19 diagnoses that were based on the response to the survey rather than on testing with nasopharyngeal swabs. In addition, comorbidity data were incomplete, and there was no indication of duration after COVID-19 infection, the severity of COVID-19, or the severity of ED.
The authors have disclosed no relevant financial relationships. Dr. Pacey is chairman of the advisory committee of the U.K. National External Quality Assurance Schemes in Andrology, editor-in-chief of Human Fertility, trustee of the Progress Educational Trust, and trustee of the British Fertility Society (all unpaid). Dr. Mulhall has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID-19 increases the risk of developing erectile dysfunction (ED) by nearly sixfold, according to data from the first study to investigate the association between ED and COVID-19 in young men in a real-life setting.
Men with ED are more than five times more likely to have COVID-19 (odds ratio, 5.27).
For men with a history of COVID-19, the odds ratio of developing ED was 5.66. The strength of the association remained after adjusting for factors considered to affect ED.
The study, which was led by Emmanuele A. Jannini, MD, professor of endocrinology and medical sexology, University of Rome Tor Vergata, was published on March 20 in Andrology.
‘Mask up to keep it up’
ED can be both a short-term and a long-term complication of COVID-19, Dr. Jannini suggests.
“When offered, men should have the COVID vaccination. It also gives a whole new meaning to wearing the mask – mask up to keep it up,” he said. “It could possibly have the added benefit of preventing sexual dysfunction.”
He points out that older age, diabetes, high body mass index, and smoking increase the risk of contracting COVID-19.
“These are the same as risk factors for ED. Results of our study agree with the pathophysiological mechanisms linking ED, endothelial dysfunction, and COVID-19. Basically, endothelial dysfunction is common in both conditions [COVID-10 and ED].
“We would like to find some sort of biomarker of endothelial dysfunction post COVID, because it seems that there are many sequelae that coexist for a long time after infection,” added Dr. Jannini. “Asking a patient if they have ED after COVID might provide a measure of systemic wellness.”
Allan Pacey, MD, professor of andrology at the University of Sheffield (England), welcomed the research, noting, “This seems to be a well-conducted study. However, at the moment, the relationship is just a correlation, and it might be that some of the comorbidities that increased the men’s chances of getting a significant COVID-19 infection may have also independently increased their chances of erectile dysfunction.
“But the authors offer a plausible mechanism by which COVID-19 may impact directly on erectile function,” agrees Dr. Pacey. However, “There’s more work to be done,” he said. “I’d also argue it’s a good reason for men to wear a mask, practice social distancing, and take the vaccine when it’s offered to them.”
Urologist John Mulhall, MD, from Memorial Sloan Kettering Cancer Center, New York, remarked, “It was a highly preliminary study, but the data are suggestive of a potential link between COVID-19 infection and ED.
“However, it raises enough questions such that further large, more long-term analyses are required to define causation. Future studies assessing testosterone levels and erectile hemodynamics will be needed to provide definite evidence of a causative link,» he stressed.
Erectile problems a ‘hallmark’ of systemic endothelial dysfunction
Prior research has suggested that asymptomatic COVID-19 could be associated with subclinical microvascular involvement with long-term cardiovascular effects.
“Indeed, COVID-19 is by all means an endothelial disease, in which systemic manifestations ... can potentially be due to alterations in the endothelial thrombotic/fibrinolytic balance,” emphasized Dr. Jannini. “In addition, endothelial cells express many of the cofactors used by SARS-CoV-2 to invade host cells.
“Erectile dysfunction has often been considered a hallmark of endothelial dysfunction, and as such, a potential association between ED and COVID-19 has also been postulated and underpinned the investigation in this study,” he explained.
The study was predicated on the fact that ED is often considered a clinical marker of impaired overall health status, which often features cardiovascular events at an early age. It aimed to investigate the bidirectional relationship between COVID-19 and ED. It asked whether ED could be a risk factor for contracting COVID-19 and whether having COVID-19 predisposes to developing ED.
“This would possibly suggest that men with ED, due to the underlying conditions which impair erectile response, could also be more susceptible to contracting COVID-19,” said Dr. Jannini.
Data were drawn from the Sex@COVID online survey, which was conducted from April 7 to May 4, 2020, in Italy. The survey included 6,821 participants aged 18 years or older (4,177 women; 2,644 men; mean age, 32.83 ± 11.24 years). Participants were stratified on the basis of marital status and sexual activity during lockdown. From these participants, 985 sexually active men were identified, among whom 25 (2.54%) reported having tested positive for COVID-19. These persons were matched with 75 COVID-19–negative men using propensity score matching in a 1:3 ratio.
The researchers used standardized psychometric tools to measure the effects of lockdown and social distancing on the intrapsychic, relational, and sexual health of the participants.
Erectile function was measured with the International Index of Erectile Function or the Sexual Health Inventory for Men, which are often used in clinical settings. In light of the two-way interaction between sexual activity and psychological well-being, results were adjusted for any influence of anxiety and depression, which were measured with recognized scales for use in patients with a history of COVID-19.
Results showed that the prevalence of ED was significantly higher among men who self-reported a history of COVID-19, compared with a matching COVID-negative population (28% vs. 9.33%; P = .027).
After adjusting for variables that are considered to have a bearing on the development of ED, such as psychological status, age, and BMI, the odds ratio for developing ED after having had COVID-19 was 5.66 (95% confidence interval, 1.50-24.01).
Similarly, after adjusting for age and BMI, men with ED were more likely to have COVID‐19 (OR, 5.27; 95% CI, 1.49-20.09).
The authors note that persons who experience “a sudden onset or worsening of ED might also consider precautionary quarantine or nasopharyngeal swab, as COVID‐19 might act as a potential initiating trigger for the onset of erectile impairment, or an aggravating factor for its progression to more severe forms.”
Similarly, patients who have ED “should consider their erectile impairment as a sign of possible underlying conditions that could increase the likelihood of suffering from COVID‐19,” they write.
Dr. Mulhall highlighted several limitations of the study, including its retrospective nature, recall bias associated with the use of online questionnaires, and the inclusion of COVID‐19 diagnoses that were based on the response to the survey rather than on testing with nasopharyngeal swabs. In addition, comorbidity data were incomplete, and there was no indication of duration after COVID-19 infection, the severity of COVID-19, or the severity of ED.
The authors have disclosed no relevant financial relationships. Dr. Pacey is chairman of the advisory committee of the U.K. National External Quality Assurance Schemes in Andrology, editor-in-chief of Human Fertility, trustee of the Progress Educational Trust, and trustee of the British Fertility Society (all unpaid). Dr. Mulhall has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID-19 increases the risk of developing erectile dysfunction (ED) by nearly sixfold, according to data from the first study to investigate the association between ED and COVID-19 in young men in a real-life setting.
Men with ED are more than five times more likely to have COVID-19 (odds ratio, 5.27).
For men with a history of COVID-19, the odds ratio of developing ED was 5.66. The strength of the association remained after adjusting for factors considered to affect ED.
The study, which was led by Emmanuele A. Jannini, MD, professor of endocrinology and medical sexology, University of Rome Tor Vergata, was published on March 20 in Andrology.
‘Mask up to keep it up’
ED can be both a short-term and a long-term complication of COVID-19, Dr. Jannini suggests.
“When offered, men should have the COVID vaccination. It also gives a whole new meaning to wearing the mask – mask up to keep it up,” he said. “It could possibly have the added benefit of preventing sexual dysfunction.”
He points out that older age, diabetes, high body mass index, and smoking increase the risk of contracting COVID-19.
“These are the same as risk factors for ED. Results of our study agree with the pathophysiological mechanisms linking ED, endothelial dysfunction, and COVID-19. Basically, endothelial dysfunction is common in both conditions [COVID-10 and ED].
“We would like to find some sort of biomarker of endothelial dysfunction post COVID, because it seems that there are many sequelae that coexist for a long time after infection,” added Dr. Jannini. “Asking a patient if they have ED after COVID might provide a measure of systemic wellness.”
Allan Pacey, MD, professor of andrology at the University of Sheffield (England), welcomed the research, noting, “This seems to be a well-conducted study. However, at the moment, the relationship is just a correlation, and it might be that some of the comorbidities that increased the men’s chances of getting a significant COVID-19 infection may have also independently increased their chances of erectile dysfunction.
“But the authors offer a plausible mechanism by which COVID-19 may impact directly on erectile function,” agrees Dr. Pacey. However, “There’s more work to be done,” he said. “I’d also argue it’s a good reason for men to wear a mask, practice social distancing, and take the vaccine when it’s offered to them.”
Urologist John Mulhall, MD, from Memorial Sloan Kettering Cancer Center, New York, remarked, “It was a highly preliminary study, but the data are suggestive of a potential link between COVID-19 infection and ED.
“However, it raises enough questions such that further large, more long-term analyses are required to define causation. Future studies assessing testosterone levels and erectile hemodynamics will be needed to provide definite evidence of a causative link,» he stressed.
Erectile problems a ‘hallmark’ of systemic endothelial dysfunction
Prior research has suggested that asymptomatic COVID-19 could be associated with subclinical microvascular involvement with long-term cardiovascular effects.
“Indeed, COVID-19 is by all means an endothelial disease, in which systemic manifestations ... can potentially be due to alterations in the endothelial thrombotic/fibrinolytic balance,” emphasized Dr. Jannini. “In addition, endothelial cells express many of the cofactors used by SARS-CoV-2 to invade host cells.
“Erectile dysfunction has often been considered a hallmark of endothelial dysfunction, and as such, a potential association between ED and COVID-19 has also been postulated and underpinned the investigation in this study,” he explained.
The study was predicated on the fact that ED is often considered a clinical marker of impaired overall health status, which often features cardiovascular events at an early age. It aimed to investigate the bidirectional relationship between COVID-19 and ED. It asked whether ED could be a risk factor for contracting COVID-19 and whether having COVID-19 predisposes to developing ED.
“This would possibly suggest that men with ED, due to the underlying conditions which impair erectile response, could also be more susceptible to contracting COVID-19,” said Dr. Jannini.
Data were drawn from the Sex@COVID online survey, which was conducted from April 7 to May 4, 2020, in Italy. The survey included 6,821 participants aged 18 years or older (4,177 women; 2,644 men; mean age, 32.83 ± 11.24 years). Participants were stratified on the basis of marital status and sexual activity during lockdown. From these participants, 985 sexually active men were identified, among whom 25 (2.54%) reported having tested positive for COVID-19. These persons were matched with 75 COVID-19–negative men using propensity score matching in a 1:3 ratio.
The researchers used standardized psychometric tools to measure the effects of lockdown and social distancing on the intrapsychic, relational, and sexual health of the participants.
Erectile function was measured with the International Index of Erectile Function or the Sexual Health Inventory for Men, which are often used in clinical settings. In light of the two-way interaction between sexual activity and psychological well-being, results were adjusted for any influence of anxiety and depression, which were measured with recognized scales for use in patients with a history of COVID-19.
Results showed that the prevalence of ED was significantly higher among men who self-reported a history of COVID-19, compared with a matching COVID-negative population (28% vs. 9.33%; P = .027).
After adjusting for variables that are considered to have a bearing on the development of ED, such as psychological status, age, and BMI, the odds ratio for developing ED after having had COVID-19 was 5.66 (95% confidence interval, 1.50-24.01).
Similarly, after adjusting for age and BMI, men with ED were more likely to have COVID‐19 (OR, 5.27; 95% CI, 1.49-20.09).
The authors note that persons who experience “a sudden onset or worsening of ED might also consider precautionary quarantine or nasopharyngeal swab, as COVID‐19 might act as a potential initiating trigger for the onset of erectile impairment, or an aggravating factor for its progression to more severe forms.”
Similarly, patients who have ED “should consider their erectile impairment as a sign of possible underlying conditions that could increase the likelihood of suffering from COVID‐19,” they write.
Dr. Mulhall highlighted several limitations of the study, including its retrospective nature, recall bias associated with the use of online questionnaires, and the inclusion of COVID‐19 diagnoses that were based on the response to the survey rather than on testing with nasopharyngeal swabs. In addition, comorbidity data were incomplete, and there was no indication of duration after COVID-19 infection, the severity of COVID-19, or the severity of ED.
The authors have disclosed no relevant financial relationships. Dr. Pacey is chairman of the advisory committee of the U.K. National External Quality Assurance Schemes in Andrology, editor-in-chief of Human Fertility, trustee of the Progress Educational Trust, and trustee of the British Fertility Society (all unpaid). Dr. Mulhall has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cardiovascular disease remains leading cause of type 2 diabetes mortality
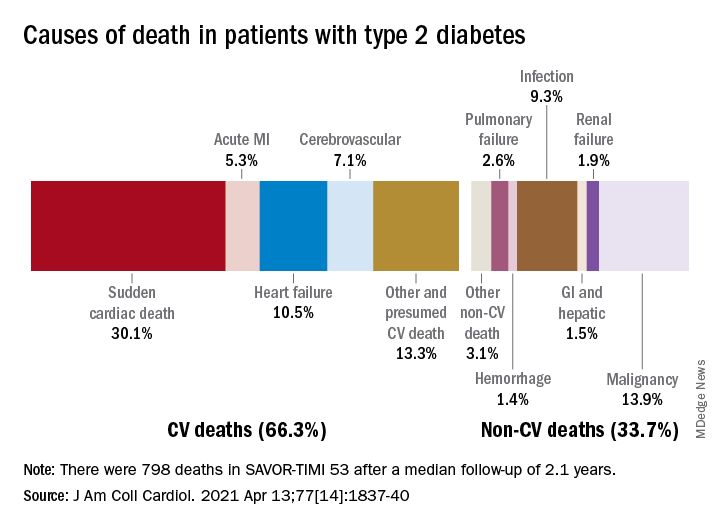
Two-thirds (66.3%) of all 798 deaths after a median 2.1 years of follow-up were caused by one of five cardiovascular (CV) conditions, with sudden cardiac death accounting for the largest share (30.1%) of the total, Ilaria Cavallari, MD, PhD, and associates said in the Journal of the American College of Cardiology.
Most common among the non-CV causes was malignancy at 13.9% of all deaths in a T2DM population at high/very high risk for CV disease (n = 16,492), followed by infection (9.3%), the members of the TIMI Study Group noted.
After variables independently associated with overall mortality were identified, a subdistribution of competing risks was constructed using a competing-risk analysis based on the proportional hazards model, they explained.
Prior heart failure was the clinical variable most associated with CV death and could, along with older age, worse glycemic control, prior CV events, peripheral artery disease, and kidney complications, “identify a subgroup of T2DM patients at high risk of mortality who are likely to achieve the greatest benefit from aggressive management of modifiable risk factors and newer glucose-lowering agents,” the investigators wrote.
It was a pair of laboratory measurements, however, that had the largest subdistribution hazard ratios. “Interestingly, the magnitude of associations of abnormal N-terminal pro–B-type natriuretic peptide [sHR, 2.82] and high-sensitivity troponin T [sHR, 2.46] measured in a stable population were greater than clinical variables in the prediction of all causes of death,” Dr. Cavallari and associates said.

Two-thirds (66.3%) of all 798 deaths after a median 2.1 years of follow-up were caused by one of five cardiovascular (CV) conditions, with sudden cardiac death accounting for the largest share (30.1%) of the total, Ilaria Cavallari, MD, PhD, and associates said in the Journal of the American College of Cardiology.
Most common among the non-CV causes was malignancy at 13.9% of all deaths in a T2DM population at high/very high risk for CV disease (n = 16,492), followed by infection (9.3%), the members of the TIMI Study Group noted.
After variables independently associated with overall mortality were identified, a subdistribution of competing risks was constructed using a competing-risk analysis based on the proportional hazards model, they explained.
Prior heart failure was the clinical variable most associated with CV death and could, along with older age, worse glycemic control, prior CV events, peripheral artery disease, and kidney complications, “identify a subgroup of T2DM patients at high risk of mortality who are likely to achieve the greatest benefit from aggressive management of modifiable risk factors and newer glucose-lowering agents,” the investigators wrote.
It was a pair of laboratory measurements, however, that had the largest subdistribution hazard ratios. “Interestingly, the magnitude of associations of abnormal N-terminal pro–B-type natriuretic peptide [sHR, 2.82] and high-sensitivity troponin T [sHR, 2.46] measured in a stable population were greater than clinical variables in the prediction of all causes of death,” Dr. Cavallari and associates said.

Two-thirds (66.3%) of all 798 deaths after a median 2.1 years of follow-up were caused by one of five cardiovascular (CV) conditions, with sudden cardiac death accounting for the largest share (30.1%) of the total, Ilaria Cavallari, MD, PhD, and associates said in the Journal of the American College of Cardiology.
Most common among the non-CV causes was malignancy at 13.9% of all deaths in a T2DM population at high/very high risk for CV disease (n = 16,492), followed by infection (9.3%), the members of the TIMI Study Group noted.
After variables independently associated with overall mortality were identified, a subdistribution of competing risks was constructed using a competing-risk analysis based on the proportional hazards model, they explained.
Prior heart failure was the clinical variable most associated with CV death and could, along with older age, worse glycemic control, prior CV events, peripheral artery disease, and kidney complications, “identify a subgroup of T2DM patients at high risk of mortality who are likely to achieve the greatest benefit from aggressive management of modifiable risk factors and newer glucose-lowering agents,” the investigators wrote.
It was a pair of laboratory measurements, however, that had the largest subdistribution hazard ratios. “Interestingly, the magnitude of associations of abnormal N-terminal pro–B-type natriuretic peptide [sHR, 2.82] and high-sensitivity troponin T [sHR, 2.46] measured in a stable population were greater than clinical variables in the prediction of all causes of death,” Dr. Cavallari and associates said.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
FDA okays new indication for alirocumab in homozygous FH
The Food and Drug Administration has approved alirocumab (Praluent, Regeneron Pharmaceuticals) injection as add-on therapy for adults with homozygous familial hypercholesterolemia, the agency announced.
The proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor was originally approved in the United States in 2015 as an adjunct to diet, alone or in combination with other lipid-lowering therapies, to reduce LDL cholesterol in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (FH).
Heterozygous FH is one of the most common genetic disorders, affecting 1 in every 200-500 people worldwide, whereas homozygous FH is very rare, affecting about 1 in 1 million people worldwide.
Alirocumab is also approved to reduce the risk of myocardial infarction, stroke, and unstable angina requiring hospitalization in adults with cardiovascular disease.
The new indication is based on a 12-week randomized trial in 45 adults who received 150 mg alirocumab every 2 weeks and 24 patients who received placebo, both on top of other therapies to reduce LDL cholesterol. At week 12, patients receiving alirocumab had an average 27% decrease in LDL cholesterol, compared with an average 9% increase among patients on placebo.
Common side effects of alirocumab are nasopharyngitis, injection-site reactions, and influenza. Serious hypersensitivity reactions have occurred among people taking alirocumab.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved alirocumab (Praluent, Regeneron Pharmaceuticals) injection as add-on therapy for adults with homozygous familial hypercholesterolemia, the agency announced.
The proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor was originally approved in the United States in 2015 as an adjunct to diet, alone or in combination with other lipid-lowering therapies, to reduce LDL cholesterol in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (FH).
Heterozygous FH is one of the most common genetic disorders, affecting 1 in every 200-500 people worldwide, whereas homozygous FH is very rare, affecting about 1 in 1 million people worldwide.
Alirocumab is also approved to reduce the risk of myocardial infarction, stroke, and unstable angina requiring hospitalization in adults with cardiovascular disease.
The new indication is based on a 12-week randomized trial in 45 adults who received 150 mg alirocumab every 2 weeks and 24 patients who received placebo, both on top of other therapies to reduce LDL cholesterol. At week 12, patients receiving alirocumab had an average 27% decrease in LDL cholesterol, compared with an average 9% increase among patients on placebo.
Common side effects of alirocumab are nasopharyngitis, injection-site reactions, and influenza. Serious hypersensitivity reactions have occurred among people taking alirocumab.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved alirocumab (Praluent, Regeneron Pharmaceuticals) injection as add-on therapy for adults with homozygous familial hypercholesterolemia, the agency announced.
The proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor was originally approved in the United States in 2015 as an adjunct to diet, alone or in combination with other lipid-lowering therapies, to reduce LDL cholesterol in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (FH).
Heterozygous FH is one of the most common genetic disorders, affecting 1 in every 200-500 people worldwide, whereas homozygous FH is very rare, affecting about 1 in 1 million people worldwide.
Alirocumab is also approved to reduce the risk of myocardial infarction, stroke, and unstable angina requiring hospitalization in adults with cardiovascular disease.
The new indication is based on a 12-week randomized trial in 45 adults who received 150 mg alirocumab every 2 weeks and 24 patients who received placebo, both on top of other therapies to reduce LDL cholesterol. At week 12, patients receiving alirocumab had an average 27% decrease in LDL cholesterol, compared with an average 9% increase among patients on placebo.
Common side effects of alirocumab are nasopharyngitis, injection-site reactions, and influenza. Serious hypersensitivity reactions have occurred among people taking alirocumab.
A version of this article first appeared on Medscape.com.


