User login
Deprescribe Low-Value Meds to Reduce Polypharmacy Harms
VANCOUVER, BRITISH COLUMBIA — While polypharmacy is inevitable for patients with multiple chronic diseases, not all medications improve patient-oriented outcomes, members of the Patients, Experience, Evidence, Research (PEER) team, a group of Canadian primary care professionals who develop evidence-based guidelines, told attendees at the Family Medicine Forum (FMF) 2024.
In a thought-provoking presentation called “Axe the Rx: Deprescribing Chronic Medications with PEER,” the panelists gave examples of medications that may be safely stopped or tapered, particularly for older adults “whose pill bag is heavier than their lunch bag.”
Curbing Cardiovascular Drugs
The 2021 Canadian Cardiovascular Society Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease in Adults call for reaching an LDL-C < 1.8 mmol/L in secondary cardiovascular prevention by potentially adding on medical therapies such as proprotein convertase subtilisin/kexin type 9 inhibitors or ezetimibe or both if that target is not reached with the maximal dosage of a statin.
But family physicians do not need to follow this guidance for their patients who have had a myocardial infarction, said Ontario family physician Jennifer Young, MD, a physician advisor in the Canadian College of Family Physicians’ Knowledge Experts and Tools Program.
Treating to below 1.8 mmol/L “means lab testing for the patients,” Young told this news organization. “It means increasing doses [of a statin] to try and get to that level.” If the patient is already on the highest dose of a statin, it means adding other medications that lower cholesterol.
“If that was translating into better outcomes like [preventing] death and another heart attack, then all of that extra effort would be worth it,” said Young. “But we don’t have evidence that it actually does have a benefit for outcomes like death and repeated heart attacks,” compared with putting them on a high dose of a potent statin.
Tapering Opioids
Before placing patients on an opioid taper, clinicians should first assess them for opioid use disorder (OUD), said Jessica Kirkwood, MD, assistant professor of family medicine at the University of Alberta in Edmonton, Canada. She suggested using the Prescription Opioid Misuse Index questionnaire to do so.
Clinicians should be much more careful in initiating a taper with patients with OUD, said Kirkwood. They must ensure that these patients are motivated to discontinue their opioids. “We’re losing 21 Canadians a day to the opioid crisis. We all know that cutting someone off their opioids and potentially having them seek opioids elsewhere through illicit means can be fatal.”
In addition, clinicians should spend more time counseling patients with OUD than those without, Kirkwood continued. They must explain to these patients how they are being tapered (eg, the intervals and doses) and highlight the benefits of a taper, such as reduced constipation. Opioid agonist therapy (such as methadone or buprenorphine) can be considered in these patients.
Some research has pointed to the importance of patient motivation as a factor in the success of opioid tapers, noted Kirkwood.
Deprescribing Benzodiazepines
Benzodiazepine receptor agonists, too, often can be deprescribed. These drugs should not be prescribed to promote sleep on a long-term basis. Yet clinicians commonly encounter patients who have been taking them for more than a year, said pharmacist Betsy Thomas, assistant adjunct professor of family medicine at the University of Alberta.
The medications “are usually fairly effective for the first couple of weeks to about a month, and then the benefits start to decrease, and we start to see more harms,” she said.
Some of the harms that have been associated with continued use of benzodiazepine receptor agonists include delayed reaction time and impaired cognition, which can affect the ability to drive, the risk for falls, and the risk for hip fractures, she noted. Some research suggests that these drugs are not an option for treating insomnia in patients aged 65 years or older.
Clinicians should encourage tapering the use of benzodiazepine receptor agonists to minimize dependence and transition patients to nonpharmacologic approaches such as cognitive behavioral therapy to manage insomnia, she said. A recent study demonstrated the efficacy of the intervention, and Thomas suggested that family physicians visit the mysleepwell.ca website for more information.
Young, Kirkwood, and Thomas reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER, BRITISH COLUMBIA — While polypharmacy is inevitable for patients with multiple chronic diseases, not all medications improve patient-oriented outcomes, members of the Patients, Experience, Evidence, Research (PEER) team, a group of Canadian primary care professionals who develop evidence-based guidelines, told attendees at the Family Medicine Forum (FMF) 2024.
In a thought-provoking presentation called “Axe the Rx: Deprescribing Chronic Medications with PEER,” the panelists gave examples of medications that may be safely stopped or tapered, particularly for older adults “whose pill bag is heavier than their lunch bag.”
Curbing Cardiovascular Drugs
The 2021 Canadian Cardiovascular Society Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease in Adults call for reaching an LDL-C < 1.8 mmol/L in secondary cardiovascular prevention by potentially adding on medical therapies such as proprotein convertase subtilisin/kexin type 9 inhibitors or ezetimibe or both if that target is not reached with the maximal dosage of a statin.
But family physicians do not need to follow this guidance for their patients who have had a myocardial infarction, said Ontario family physician Jennifer Young, MD, a physician advisor in the Canadian College of Family Physicians’ Knowledge Experts and Tools Program.
Treating to below 1.8 mmol/L “means lab testing for the patients,” Young told this news organization. “It means increasing doses [of a statin] to try and get to that level.” If the patient is already on the highest dose of a statin, it means adding other medications that lower cholesterol.
“If that was translating into better outcomes like [preventing] death and another heart attack, then all of that extra effort would be worth it,” said Young. “But we don’t have evidence that it actually does have a benefit for outcomes like death and repeated heart attacks,” compared with putting them on a high dose of a potent statin.
Tapering Opioids
Before placing patients on an opioid taper, clinicians should first assess them for opioid use disorder (OUD), said Jessica Kirkwood, MD, assistant professor of family medicine at the University of Alberta in Edmonton, Canada. She suggested using the Prescription Opioid Misuse Index questionnaire to do so.
Clinicians should be much more careful in initiating a taper with patients with OUD, said Kirkwood. They must ensure that these patients are motivated to discontinue their opioids. “We’re losing 21 Canadians a day to the opioid crisis. We all know that cutting someone off their opioids and potentially having them seek opioids elsewhere through illicit means can be fatal.”
In addition, clinicians should spend more time counseling patients with OUD than those without, Kirkwood continued. They must explain to these patients how they are being tapered (eg, the intervals and doses) and highlight the benefits of a taper, such as reduced constipation. Opioid agonist therapy (such as methadone or buprenorphine) can be considered in these patients.
Some research has pointed to the importance of patient motivation as a factor in the success of opioid tapers, noted Kirkwood.
Deprescribing Benzodiazepines
Benzodiazepine receptor agonists, too, often can be deprescribed. These drugs should not be prescribed to promote sleep on a long-term basis. Yet clinicians commonly encounter patients who have been taking them for more than a year, said pharmacist Betsy Thomas, assistant adjunct professor of family medicine at the University of Alberta.
The medications “are usually fairly effective for the first couple of weeks to about a month, and then the benefits start to decrease, and we start to see more harms,” she said.
Some of the harms that have been associated with continued use of benzodiazepine receptor agonists include delayed reaction time and impaired cognition, which can affect the ability to drive, the risk for falls, and the risk for hip fractures, she noted. Some research suggests that these drugs are not an option for treating insomnia in patients aged 65 years or older.
Clinicians should encourage tapering the use of benzodiazepine receptor agonists to minimize dependence and transition patients to nonpharmacologic approaches such as cognitive behavioral therapy to manage insomnia, she said. A recent study demonstrated the efficacy of the intervention, and Thomas suggested that family physicians visit the mysleepwell.ca website for more information.
Young, Kirkwood, and Thomas reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER, BRITISH COLUMBIA — While polypharmacy is inevitable for patients with multiple chronic diseases, not all medications improve patient-oriented outcomes, members of the Patients, Experience, Evidence, Research (PEER) team, a group of Canadian primary care professionals who develop evidence-based guidelines, told attendees at the Family Medicine Forum (FMF) 2024.
In a thought-provoking presentation called “Axe the Rx: Deprescribing Chronic Medications with PEER,” the panelists gave examples of medications that may be safely stopped or tapered, particularly for older adults “whose pill bag is heavier than their lunch bag.”
Curbing Cardiovascular Drugs
The 2021 Canadian Cardiovascular Society Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease in Adults call for reaching an LDL-C < 1.8 mmol/L in secondary cardiovascular prevention by potentially adding on medical therapies such as proprotein convertase subtilisin/kexin type 9 inhibitors or ezetimibe or both if that target is not reached with the maximal dosage of a statin.
But family physicians do not need to follow this guidance for their patients who have had a myocardial infarction, said Ontario family physician Jennifer Young, MD, a physician advisor in the Canadian College of Family Physicians’ Knowledge Experts and Tools Program.
Treating to below 1.8 mmol/L “means lab testing for the patients,” Young told this news organization. “It means increasing doses [of a statin] to try and get to that level.” If the patient is already on the highest dose of a statin, it means adding other medications that lower cholesterol.
“If that was translating into better outcomes like [preventing] death and another heart attack, then all of that extra effort would be worth it,” said Young. “But we don’t have evidence that it actually does have a benefit for outcomes like death and repeated heart attacks,” compared with putting them on a high dose of a potent statin.
Tapering Opioids
Before placing patients on an opioid taper, clinicians should first assess them for opioid use disorder (OUD), said Jessica Kirkwood, MD, assistant professor of family medicine at the University of Alberta in Edmonton, Canada. She suggested using the Prescription Opioid Misuse Index questionnaire to do so.
Clinicians should be much more careful in initiating a taper with patients with OUD, said Kirkwood. They must ensure that these patients are motivated to discontinue their opioids. “We’re losing 21 Canadians a day to the opioid crisis. We all know that cutting someone off their opioids and potentially having them seek opioids elsewhere through illicit means can be fatal.”
In addition, clinicians should spend more time counseling patients with OUD than those without, Kirkwood continued. They must explain to these patients how they are being tapered (eg, the intervals and doses) and highlight the benefits of a taper, such as reduced constipation. Opioid agonist therapy (such as methadone or buprenorphine) can be considered in these patients.
Some research has pointed to the importance of patient motivation as a factor in the success of opioid tapers, noted Kirkwood.
Deprescribing Benzodiazepines
Benzodiazepine receptor agonists, too, often can be deprescribed. These drugs should not be prescribed to promote sleep on a long-term basis. Yet clinicians commonly encounter patients who have been taking them for more than a year, said pharmacist Betsy Thomas, assistant adjunct professor of family medicine at the University of Alberta.
The medications “are usually fairly effective for the first couple of weeks to about a month, and then the benefits start to decrease, and we start to see more harms,” she said.
Some of the harms that have been associated with continued use of benzodiazepine receptor agonists include delayed reaction time and impaired cognition, which can affect the ability to drive, the risk for falls, and the risk for hip fractures, she noted. Some research suggests that these drugs are not an option for treating insomnia in patients aged 65 years or older.
Clinicians should encourage tapering the use of benzodiazepine receptor agonists to minimize dependence and transition patients to nonpharmacologic approaches such as cognitive behavioral therapy to manage insomnia, she said. A recent study demonstrated the efficacy of the intervention, and Thomas suggested that family physicians visit the mysleepwell.ca website for more information.
Young, Kirkwood, and Thomas reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM FMF 2024
Aliens, Ian McShane, and Heart Disease Risk
This transcript has been edited for clarity.
I was really struggling to think of a good analogy to explain the glaring problem of polygenic risk scores (PRS) this week. But I think I have it now. Go with me on this.
An alien spaceship parks itself, Independence Day style, above a local office building.
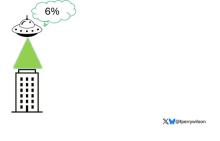
But unlike the aliens that gave such a hard time to Will Smith and Brent Spiner, these are benevolent, technologically superior guys. They shine a mysterious green light down on the building and then announce, maybe via telepathy, that 6% of the people in that building will have a heart attack in the next year.
They move on to the next building. “Five percent will have a heart attack in the next year.” And the next, 7%. And the next, 2%.
Let’s assume the aliens are entirely accurate. What do you do with this information?
Most of us would suggest that you find out who was in the buildings with the higher percentages. You check their cholesterol levels, get them to exercise more, do some stress tests, and so on.
But that said, you’d still be spending a lot of money on a bunch of people who were not going to have heart attacks. So, a crack team of spies — in my mind, this is definitely led by a grizzled Ian McShane — infiltrate the alien ship, steal this predictive ray gun, and start pointing it, not at buildings but at people.
In this scenario, one person could have a 10% chance of having a heart attack in the next year. Another person has a 50% chance. The aliens, seeing this, leave us one final message before flying into the great beyond: “No, you guys are doing it wrong.”
This week: The people and companies using an advanced predictive technology, PRS , wrong — and a study that shows just how problematic this is.
We all know that genes play a significant role in our health outcomes. Some diseases (Huntington disease, cystic fibrosis, sickle cell disease, hemochromatosis, and Duchenne muscular dystrophy, for example) are entirely driven by genetic mutations.
The vast majority of chronic diseases we face are not driven by genetics, but they may be enhanced by genetics. Coronary heart disease (CHD) is a prime example. There are clearly environmental risk factors, like smoking, that dramatically increase risk. But there are also genetic underpinnings; about half the risk for CHD comes from genetic variation, according to one study.
But in the case of those common diseases, it’s not one gene that leads to increased risk; it’s the aggregate effect of multiple risk genes, each contributing a small amount of risk to the final total.
The promise of PRS was based on this fact. Take the genome of an individual, identify all the risk genes, and integrate them into some final number that represents your genetic risk of developing CHD.
The way you derive a PRS is take a big group of people and sequence their genomes. Then, you see who develops the disease of interest — in this case, CHD. If the people who develop CHD are more likely to have a particular mutation, that mutation goes in the risk score. Risk scores can integrate tens, hundreds, even thousands of individual mutations to create that final score.
There are literally dozens of PRS for CHD. And there are companies that will calculate yours right now for a reasonable fee.
The accuracy of these scores is assessed at the population level. It’s the alien ray gun thing. Researchers apply the PRS to a big group of people and say 20% of them should develop CHD. If indeed 20% develop CHD, they say the score is accurate. And that’s true.
But what happens next is the problem. Companies and even doctors have been marketing PRS to individuals. And honestly, it sounds amazing. “We’ll use sophisticated techniques to analyze your genetic code and integrate the information to give you your personal risk for CHD.” Or dementia. Or other diseases. A lot of people would want to know this information.
It turns out, though, that this is where the system breaks down. And it is nicely illustrated by this study, appearing November 16 in JAMA.
The authors wanted to see how PRS, which are developed to predict disease in a group of people, work when applied to an individual.
They identified 48 previously published PRS for CHD. They applied those scores to more than 170,000 individuals across multiple genetic databases. And, by and large, the scores worked as advertised, at least across the entire group. The weighted accuracy of all 48 scores was around 78%. They aren’t perfect, of course. We wouldn’t expect them to be, since CHD is not entirely driven by genetics. But 78% accurate isn’t too bad.
But that accuracy is at the population level. At the level of the office building. At the individual level, it was a vastly different story.
This is best illustrated by this plot, which shows the score from 48 different PRS for CHD within the same person. A note here: It is arranged by the publication date of the risk score, but these were all assessed on a single blood sample at a single point in time in this study participant.
The individual scores are all over the map. Using one risk score gives an individual a risk that is near the 99th percentile — a ticking time bomb of CHD. Another score indicates a level of risk at the very bottom of the spectrum — highly reassuring. A bunch of scores fall somewhere in between. In other words, as a doctor, the risk I will discuss with this patient is more strongly determined by which PRS I happen to choose than by his actual genetic risk, whatever that is.
This may seem counterintuitive. All these risk scores were similarly accurate within a population; how can they all give different results to an individual? The answer is simpler than you may think. As long as a given score makes one extra good prediction for each extra bad prediction, its accuracy is not changed.
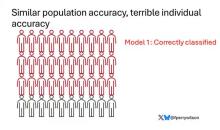
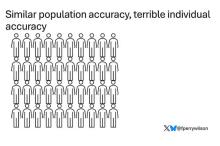
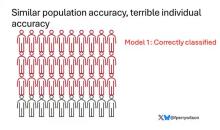
Let’s imagine we have a population of 40 people.
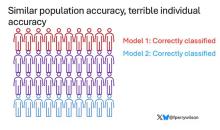
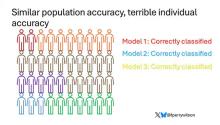
Risk score model 1 correctly classified 30 of them for 75% accuracy. Great.
Risk score model 2 also correctly classified 30 of our 40 individuals, for 75% accuracy. It’s just a different 30.
Risk score model 3 also correctly classified 30 of 40, but another different 30.
I’ve colored this to show you all the different overlaps. What you can see is that although each score has similar accuracy, the individual people have a bunch of different colors, indicating that some scores worked for them and some didn’t. That’s a real problem.
This has not stopped companies from advertising PRS for all sorts of diseases. Companies are even using PRS to decide which fetuses to implant during IVF therapy, which is a particularly egregiously wrong use of this technology that I have written about before.
How do you fix this? Our aliens tried to warn us. This is not how you are supposed to use this ray gun. You are supposed to use it to identify groups of people at higher risk to direct more resources to that group. That’s really all you can do.
It’s also possible that we need to match the risk score to the individual in a better way. This is likely driven by the fact that risk scores tend to work best in the populations in which they were developed, and many of them were developed in people of largely European ancestry.
It is worth noting that if a PRS had perfect accuracy at the population level, it would also necessarily have perfect accuracy at the individual level. But there aren’t any scores like that. It’s possible that combining various scores may increase the individual accuracy, but that hasn’t been demonstrated yet either.
Look, genetics is and will continue to play a major role in healthcare. At the same time, sequencing entire genomes is a technology that is ripe for hype and thus misuse. Or even abuse. Fundamentally, this JAMA study reminds us that accuracy in a population and accuracy in an individual are not the same. But more deeply, it reminds us that just because a technology is new or cool or expensive doesn’t mean it will work in the clinic.
Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I was really struggling to think of a good analogy to explain the glaring problem of polygenic risk scores (PRS) this week. But I think I have it now. Go with me on this.
An alien spaceship parks itself, Independence Day style, above a local office building.
But unlike the aliens that gave such a hard time to Will Smith and Brent Spiner, these are benevolent, technologically superior guys. They shine a mysterious green light down on the building and then announce, maybe via telepathy, that 6% of the people in that building will have a heart attack in the next year.
They move on to the next building. “Five percent will have a heart attack in the next year.” And the next, 7%. And the next, 2%.
Let’s assume the aliens are entirely accurate. What do you do with this information?
Most of us would suggest that you find out who was in the buildings with the higher percentages. You check their cholesterol levels, get them to exercise more, do some stress tests, and so on.
But that said, you’d still be spending a lot of money on a bunch of people who were not going to have heart attacks. So, a crack team of spies — in my mind, this is definitely led by a grizzled Ian McShane — infiltrate the alien ship, steal this predictive ray gun, and start pointing it, not at buildings but at people.
In this scenario, one person could have a 10% chance of having a heart attack in the next year. Another person has a 50% chance. The aliens, seeing this, leave us one final message before flying into the great beyond: “No, you guys are doing it wrong.”
This week: The people and companies using an advanced predictive technology, PRS , wrong — and a study that shows just how problematic this is.
We all know that genes play a significant role in our health outcomes. Some diseases (Huntington disease, cystic fibrosis, sickle cell disease, hemochromatosis, and Duchenne muscular dystrophy, for example) are entirely driven by genetic mutations.
The vast majority of chronic diseases we face are not driven by genetics, but they may be enhanced by genetics. Coronary heart disease (CHD) is a prime example. There are clearly environmental risk factors, like smoking, that dramatically increase risk. But there are also genetic underpinnings; about half the risk for CHD comes from genetic variation, according to one study.
But in the case of those common diseases, it’s not one gene that leads to increased risk; it’s the aggregate effect of multiple risk genes, each contributing a small amount of risk to the final total.
The promise of PRS was based on this fact. Take the genome of an individual, identify all the risk genes, and integrate them into some final number that represents your genetic risk of developing CHD.
The way you derive a PRS is take a big group of people and sequence their genomes. Then, you see who develops the disease of interest — in this case, CHD. If the people who develop CHD are more likely to have a particular mutation, that mutation goes in the risk score. Risk scores can integrate tens, hundreds, even thousands of individual mutations to create that final score.
There are literally dozens of PRS for CHD. And there are companies that will calculate yours right now for a reasonable fee.
The accuracy of these scores is assessed at the population level. It’s the alien ray gun thing. Researchers apply the PRS to a big group of people and say 20% of them should develop CHD. If indeed 20% develop CHD, they say the score is accurate. And that’s true.
But what happens next is the problem. Companies and even doctors have been marketing PRS to individuals. And honestly, it sounds amazing. “We’ll use sophisticated techniques to analyze your genetic code and integrate the information to give you your personal risk for CHD.” Or dementia. Or other diseases. A lot of people would want to know this information.
It turns out, though, that this is where the system breaks down. And it is nicely illustrated by this study, appearing November 16 in JAMA.
The authors wanted to see how PRS, which are developed to predict disease in a group of people, work when applied to an individual.
They identified 48 previously published PRS for CHD. They applied those scores to more than 170,000 individuals across multiple genetic databases. And, by and large, the scores worked as advertised, at least across the entire group. The weighted accuracy of all 48 scores was around 78%. They aren’t perfect, of course. We wouldn’t expect them to be, since CHD is not entirely driven by genetics. But 78% accurate isn’t too bad.
But that accuracy is at the population level. At the level of the office building. At the individual level, it was a vastly different story.
This is best illustrated by this plot, which shows the score from 48 different PRS for CHD within the same person. A note here: It is arranged by the publication date of the risk score, but these were all assessed on a single blood sample at a single point in time in this study participant.
The individual scores are all over the map. Using one risk score gives an individual a risk that is near the 99th percentile — a ticking time bomb of CHD. Another score indicates a level of risk at the very bottom of the spectrum — highly reassuring. A bunch of scores fall somewhere in between. In other words, as a doctor, the risk I will discuss with this patient is more strongly determined by which PRS I happen to choose than by his actual genetic risk, whatever that is.
This may seem counterintuitive. All these risk scores were similarly accurate within a population; how can they all give different results to an individual? The answer is simpler than you may think. As long as a given score makes one extra good prediction for each extra bad prediction, its accuracy is not changed.
Let’s imagine we have a population of 40 people.
Risk score model 1 correctly classified 30 of them for 75% accuracy. Great.
Risk score model 2 also correctly classified 30 of our 40 individuals, for 75% accuracy. It’s just a different 30.
Risk score model 3 also correctly classified 30 of 40, but another different 30.
I’ve colored this to show you all the different overlaps. What you can see is that although each score has similar accuracy, the individual people have a bunch of different colors, indicating that some scores worked for them and some didn’t. That’s a real problem.
This has not stopped companies from advertising PRS for all sorts of diseases. Companies are even using PRS to decide which fetuses to implant during IVF therapy, which is a particularly egregiously wrong use of this technology that I have written about before.
How do you fix this? Our aliens tried to warn us. This is not how you are supposed to use this ray gun. You are supposed to use it to identify groups of people at higher risk to direct more resources to that group. That’s really all you can do.
It’s also possible that we need to match the risk score to the individual in a better way. This is likely driven by the fact that risk scores tend to work best in the populations in which they were developed, and many of them were developed in people of largely European ancestry.
It is worth noting that if a PRS had perfect accuracy at the population level, it would also necessarily have perfect accuracy at the individual level. But there aren’t any scores like that. It’s possible that combining various scores may increase the individual accuracy, but that hasn’t been demonstrated yet either.
Look, genetics is and will continue to play a major role in healthcare. At the same time, sequencing entire genomes is a technology that is ripe for hype and thus misuse. Or even abuse. Fundamentally, this JAMA study reminds us that accuracy in a population and accuracy in an individual are not the same. But more deeply, it reminds us that just because a technology is new or cool or expensive doesn’t mean it will work in the clinic.
Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I was really struggling to think of a good analogy to explain the glaring problem of polygenic risk scores (PRS) this week. But I think I have it now. Go with me on this.
An alien spaceship parks itself, Independence Day style, above a local office building.
But unlike the aliens that gave such a hard time to Will Smith and Brent Spiner, these are benevolent, technologically superior guys. They shine a mysterious green light down on the building and then announce, maybe via telepathy, that 6% of the people in that building will have a heart attack in the next year.
They move on to the next building. “Five percent will have a heart attack in the next year.” And the next, 7%. And the next, 2%.
Let’s assume the aliens are entirely accurate. What do you do with this information?
Most of us would suggest that you find out who was in the buildings with the higher percentages. You check their cholesterol levels, get them to exercise more, do some stress tests, and so on.
But that said, you’d still be spending a lot of money on a bunch of people who were not going to have heart attacks. So, a crack team of spies — in my mind, this is definitely led by a grizzled Ian McShane — infiltrate the alien ship, steal this predictive ray gun, and start pointing it, not at buildings but at people.
In this scenario, one person could have a 10% chance of having a heart attack in the next year. Another person has a 50% chance. The aliens, seeing this, leave us one final message before flying into the great beyond: “No, you guys are doing it wrong.”
This week: The people and companies using an advanced predictive technology, PRS , wrong — and a study that shows just how problematic this is.
We all know that genes play a significant role in our health outcomes. Some diseases (Huntington disease, cystic fibrosis, sickle cell disease, hemochromatosis, and Duchenne muscular dystrophy, for example) are entirely driven by genetic mutations.
The vast majority of chronic diseases we face are not driven by genetics, but they may be enhanced by genetics. Coronary heart disease (CHD) is a prime example. There are clearly environmental risk factors, like smoking, that dramatically increase risk. But there are also genetic underpinnings; about half the risk for CHD comes from genetic variation, according to one study.
But in the case of those common diseases, it’s not one gene that leads to increased risk; it’s the aggregate effect of multiple risk genes, each contributing a small amount of risk to the final total.
The promise of PRS was based on this fact. Take the genome of an individual, identify all the risk genes, and integrate them into some final number that represents your genetic risk of developing CHD.
The way you derive a PRS is take a big group of people and sequence their genomes. Then, you see who develops the disease of interest — in this case, CHD. If the people who develop CHD are more likely to have a particular mutation, that mutation goes in the risk score. Risk scores can integrate tens, hundreds, even thousands of individual mutations to create that final score.
There are literally dozens of PRS for CHD. And there are companies that will calculate yours right now for a reasonable fee.
The accuracy of these scores is assessed at the population level. It’s the alien ray gun thing. Researchers apply the PRS to a big group of people and say 20% of them should develop CHD. If indeed 20% develop CHD, they say the score is accurate. And that’s true.
But what happens next is the problem. Companies and even doctors have been marketing PRS to individuals. And honestly, it sounds amazing. “We’ll use sophisticated techniques to analyze your genetic code and integrate the information to give you your personal risk for CHD.” Or dementia. Or other diseases. A lot of people would want to know this information.
It turns out, though, that this is where the system breaks down. And it is nicely illustrated by this study, appearing November 16 in JAMA.
The authors wanted to see how PRS, which are developed to predict disease in a group of people, work when applied to an individual.
They identified 48 previously published PRS for CHD. They applied those scores to more than 170,000 individuals across multiple genetic databases. And, by and large, the scores worked as advertised, at least across the entire group. The weighted accuracy of all 48 scores was around 78%. They aren’t perfect, of course. We wouldn’t expect them to be, since CHD is not entirely driven by genetics. But 78% accurate isn’t too bad.
But that accuracy is at the population level. At the level of the office building. At the individual level, it was a vastly different story.
This is best illustrated by this plot, which shows the score from 48 different PRS for CHD within the same person. A note here: It is arranged by the publication date of the risk score, but these were all assessed on a single blood sample at a single point in time in this study participant.
The individual scores are all over the map. Using one risk score gives an individual a risk that is near the 99th percentile — a ticking time bomb of CHD. Another score indicates a level of risk at the very bottom of the spectrum — highly reassuring. A bunch of scores fall somewhere in between. In other words, as a doctor, the risk I will discuss with this patient is more strongly determined by which PRS I happen to choose than by his actual genetic risk, whatever that is.
This may seem counterintuitive. All these risk scores were similarly accurate within a population; how can they all give different results to an individual? The answer is simpler than you may think. As long as a given score makes one extra good prediction for each extra bad prediction, its accuracy is not changed.
Let’s imagine we have a population of 40 people.
Risk score model 1 correctly classified 30 of them for 75% accuracy. Great.
Risk score model 2 also correctly classified 30 of our 40 individuals, for 75% accuracy. It’s just a different 30.
Risk score model 3 also correctly classified 30 of 40, but another different 30.
I’ve colored this to show you all the different overlaps. What you can see is that although each score has similar accuracy, the individual people have a bunch of different colors, indicating that some scores worked for them and some didn’t. That’s a real problem.
This has not stopped companies from advertising PRS for all sorts of diseases. Companies are even using PRS to decide which fetuses to implant during IVF therapy, which is a particularly egregiously wrong use of this technology that I have written about before.
How do you fix this? Our aliens tried to warn us. This is not how you are supposed to use this ray gun. You are supposed to use it to identify groups of people at higher risk to direct more resources to that group. That’s really all you can do.
It’s also possible that we need to match the risk score to the individual in a better way. This is likely driven by the fact that risk scores tend to work best in the populations in which they were developed, and many of them were developed in people of largely European ancestry.
It is worth noting that if a PRS had perfect accuracy at the population level, it would also necessarily have perfect accuracy at the individual level. But there aren’t any scores like that. It’s possible that combining various scores may increase the individual accuracy, but that hasn’t been demonstrated yet either.
Look, genetics is and will continue to play a major role in healthcare. At the same time, sequencing entire genomes is a technology that is ripe for hype and thus misuse. Or even abuse. Fundamentally, this JAMA study reminds us that accuracy in a population and accuracy in an individual are not the same. But more deeply, it reminds us that just because a technology is new or cool or expensive doesn’t mean it will work in the clinic.
Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Effect of Alirocumab Monotherapy vs Ezetimibe Plus Statin Therapy on LDL-C Lowering in Veterans With History of ASCVD
Atherosclerotic cardiovascular disease (ASCVD) is a significant cause of morbidity and mortality in the United States. ASCVD involves the buildup of cholesterol plaque in arteries and includes acute coronary syndrome, peripheral arterial disease, and events such as myocardial infarction and stroke.1 Cardiovascular disease (CVD) risk factors include high cholesterol levels, elevated blood pressure, insulin resistance, elevated blood glucose levels, smoking, poor dietary habits, and a sedentary lifestyle.2
According to the Centers for Disease Control and Prevention, about 86 million adults aged ≥ 20 years have total cholesterol levels > 200 mg/dL. More than half (54.5%) who could benefit are currently taking cholesterol-lowering medications.3 Controlling high cholesterol in American adults, especially veterans, is essential for reducing CVD morbidity and mortality.
The 2018 American College of Cardiology/American Heart Association (ACC/AHA) guideline recommends a low-density lipoprotein cholesterol (LDL-C) target goal of < 70 mg/dL for patients at high risk for ASCVD. Very high-risk ASCVD includes a history of multiple major ASCVD events or 1 major ASCVD event and multiple high-risk conditions (eg, age ≥ 65 years, smoking, or diabetes).4 Major ASCVD events include recent acute coronary syndrome (within the past 12 months), a history of myocardial infarction or ischemic stroke, and symptomatic peripheral artery disease.
The ACC/AHA guideline suggests that if the LDL-C level remains ≥ 70 mg/dL, adding ezetimibe (a dietary cholesterol absorption inhibitor) to maximally tolerated statin therapy is reasonable. If LDL-C levels remain ≥ 70 mg/dL, adding a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor, such as alirocumab, is reasonable.4 The US Departments of Veterans Affairs/US Department of Defense guidelines recommend using maximally tolerated statins and ezetimibe before PCSK9 inhibitors due to established long-term safety and reduction in CVD events.
Generic statins and ezetimibe are administered orally and widely available. In contrast, PCSK9 inhibitors have unknown long-term safety profiles, require subcutaneous injection once or twice monthly, and are significantly more expensive. They also require patient education on proper use while providing comparable or lesser relative risk reductions.2
These 3 classes of medication vary in their mechanisms of action to reduce LDL.5,6 Ezetimibe and several statin medications are included on the Veterans Affairs Sioux Falls Health Care System (VASFHCS) formulary and do not require review prior to prescribing. Alirocumab is available at VASFHCS but is restricted to patients with a history of ASCVD or a diagnosis of familial hypercholesterolemia, and who are receiving maximally tolerated statin and ezetimibe therapy but require further LDL-C lowering to reduce their ASCVD risk.
Studies have found ezetimibe monotherapy reduces LDL-C in patients with dyslipidemia by 18% after 12 weeks.7 One found that the percentage reduction in LDL-C was significantly greater (P < .001) with all doses of ezetimibe plus simvastatin (46% to 59%) compared with either atorvastatin 10 mg (37%) or simvastatin 20 mg (38%) monotherapy after 6 weeks.8
Although alirocumab can be added to other lipid therapies, most VASFHCS patients are prescribed alirocumab monotherapy. In the ODYSSEY CHOICE II study, patients were randomly assigned to receive either a placebo or alirocumab 150 mg every 4 weeks or alirocumab 75 mg every 2 weeks. The primary efficacy endpoint was LDL-C percentage change from baseline to week 24. In the alirocumab 150 mg every 4 weeks and 75 mg every 2 weeks groups, the least-squares mean LDL-C changes from baseline to week 24 were 51.7% and 53.5%, respectively, compared to a 4.7% increase in the placebo group (both groups P < .001 vs placebo). The authors also reported that alirocumab 150 mg every 4 weeks as monotherapy demonstrated a 47.4% reduction in LDL-C levels from baseline in a phase 1 study.9Although alirocumab monotherapy and ezetimibe plus statin therapy have been shown to effectively decrease LDL-C independently, a direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been assessed, to our knowledge. Understanding the differences in effectiveness and safety between these 2 regimens will be valuable for clinicians when selecting a medication regimen for veterans with a history of ASCVD.
METHODS
This retrospective, single-center chart review used VASFHCS Computerized Patient Record System (CPRS) and Joint Longitudinal Viewer (JLV) records to compare patients with a history of ASCVD events who were treated with alirocumab monotherapy or ezetimibe plus statin. The 2 groups were randomized in a 1:3 ratio. The primary endpoint was achieving LDL-C < 70 mg/dL after 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks. Secondary endpoints included the mean percentage change from baseline in total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), LDL-C, and triglycerides (TG) over 52 weeks. The incidence of ASCVD events during this period was also assessed. If LDL-C < 70 mg/dL was achieved > 1 time during each time frame, only 1 incident was counted for analysis. Safety was assessed based on the incidence of any adverse event (AE) that led to treatment discontinuation.
Patients were identified by screening the prescription fill history between October 1, 2019, and December 31, 2022. The 52-week data collection period was counted from the first available fill date. Additionally, the prior authorization drug request file from January 1, 2017, to December 31, 2022, was used to obtain a list of patients prescribed alirocumab. Patients were included if they were veterans aged ≥ 18 years and had a history of an ASCVD event, had a alirocumab monotherapy or ezetimibe plus statin prescription between October 1, 2019, and December 31, 2022, or had an approved prior authorization drug request for alirocumab between January 1, 2017, and December 31, 2022. Patients missing a baseline or follow-up lipid panel and those with concurrent use of alirocumab and ezetimibe and/or statin were excluded.
Baseline characteristics collected for patients included age, sex, race, weight, body mass index, lipid parameters (LDL-C, TC, HDL-C, and TG), dosing of each type of statin before adding ezetimibe, and use of any other antihyperlipidemic medication. We also collected histories of hypertension, hyperlipidemia, diabetes, chronic kidney disease, congestive heart failure, and smoking or tobacco use status. The baseline lipid panel was the most recent lipid panel documented before starting alirocumab or ezetimibe plus statin therapy. Follow-up lipid panel values were gathered at 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks following initiation of either therapy.
High-, moderate-, and low-intensity dosing of statin therapy and alirocumab dosing (75 mg every 2 weeks, 150 mg every 2 weeks, or 300 mg every 4 weeks) were recorded at the specified intervals. However, no patients in this study received the latter dosing regimen. ASCVD events and safety endpoints were recorded based on a review of clinical notes over the 52 weeks following the first available start date.
Statistical Analysis
The primary endpoint of achieving the LDL-C < 70 mg/dL goal from baseline to 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks after initiation was compared between alirocumab monotherapy and ezetimibe plus statin therapy using the χ² test. Mean percentage change from baseline in TC, HDL-C, LDL-C, and TG were compared using the independent t test. P < .05 was considered statistically significant. Incidence of ASCVD events and the safety endpoint (incidence of AEs leading to treatment discontinuation) were also compared using the χ² test. Continuous baseline characteristics were reported mean (SD) and nominal baseline characteristics were reported as a percentage.
RESULTS
There were 80 participants in this study: 20 in the alirocumab monotherapy group and 60 in the ezetimibe plus statin therapy group. More than 100 patients did not meet the prespecified inclusion criteria and were excluded. Mean (SD) age was 75 (8) years in the alirocumab group and 74 (8) years in the ezetimibe plus statin group. There was no significant differences in mean (SD) weight or mean (SD) body mass index. All study participants identified as White and male except for 2 patients in the ezetimibe plus statin therapy group whose race was not documented. Differences in lipid parameters were observed between groups, with mean baseline LDL-C, HDL-C, and TC higher in the alirocumab monotherapy group than in the ezetimibe plus statin therapy group, with significant differences in LDL-C and TC (Table 1).
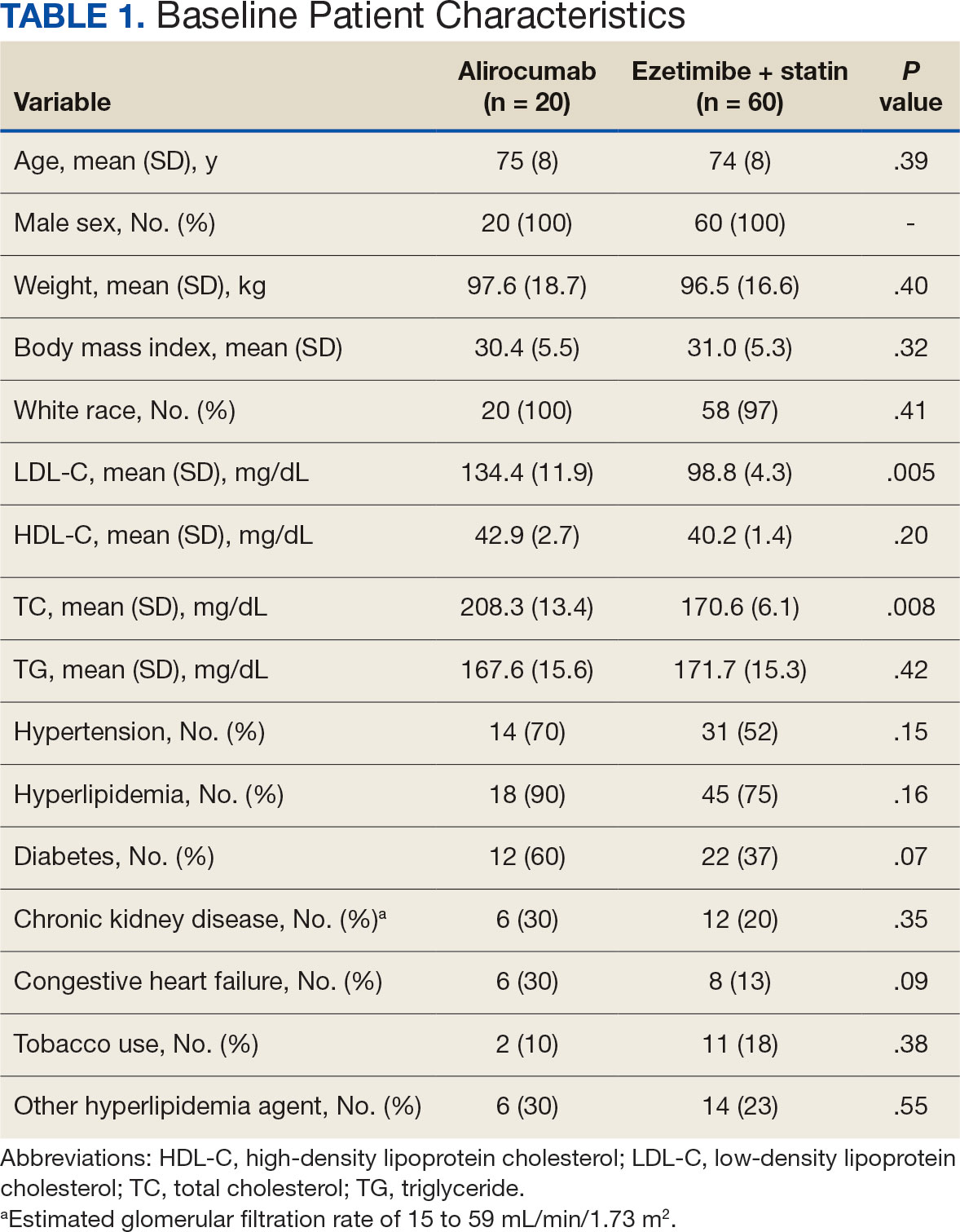
Fourteen patients (70%) in the alirocumab monotherapy group had hypertension, compared with 31 (52%) in the ezetimibe plus statin therapy group. In both groups, most patients had previously been diagnosed with hyperlipidemia. More patients (60%) in the alirocumab group had diabetes than in the ezetimibe plus statin therapy group (37%). The alirocumab monotherapy group also had a higher percentage of patients with diagnoses of congestive heart failure and used other antihyperlipidemic medications than in the ezetimibe plus statin therapy group. Five patients (25%) in the alirocumab monotherapy group and 12 patients (20%) in the ezetimibe plus statin therapy group took fish oil. In the ezetimibe plus statin therapy group, 2 patients (3%) took gemfibrozil, and 2 patients (3%) took fenofibrate. Six (30%) patients in the alirocumab monotherapy group and 12 (20%) patients in the ezetimibe plus statin therapy group had chronic kidney disease. Although the majority of patients in each group did not use tobacco products, there were more tobacco users in the ezetimibe plus statin therapy group.
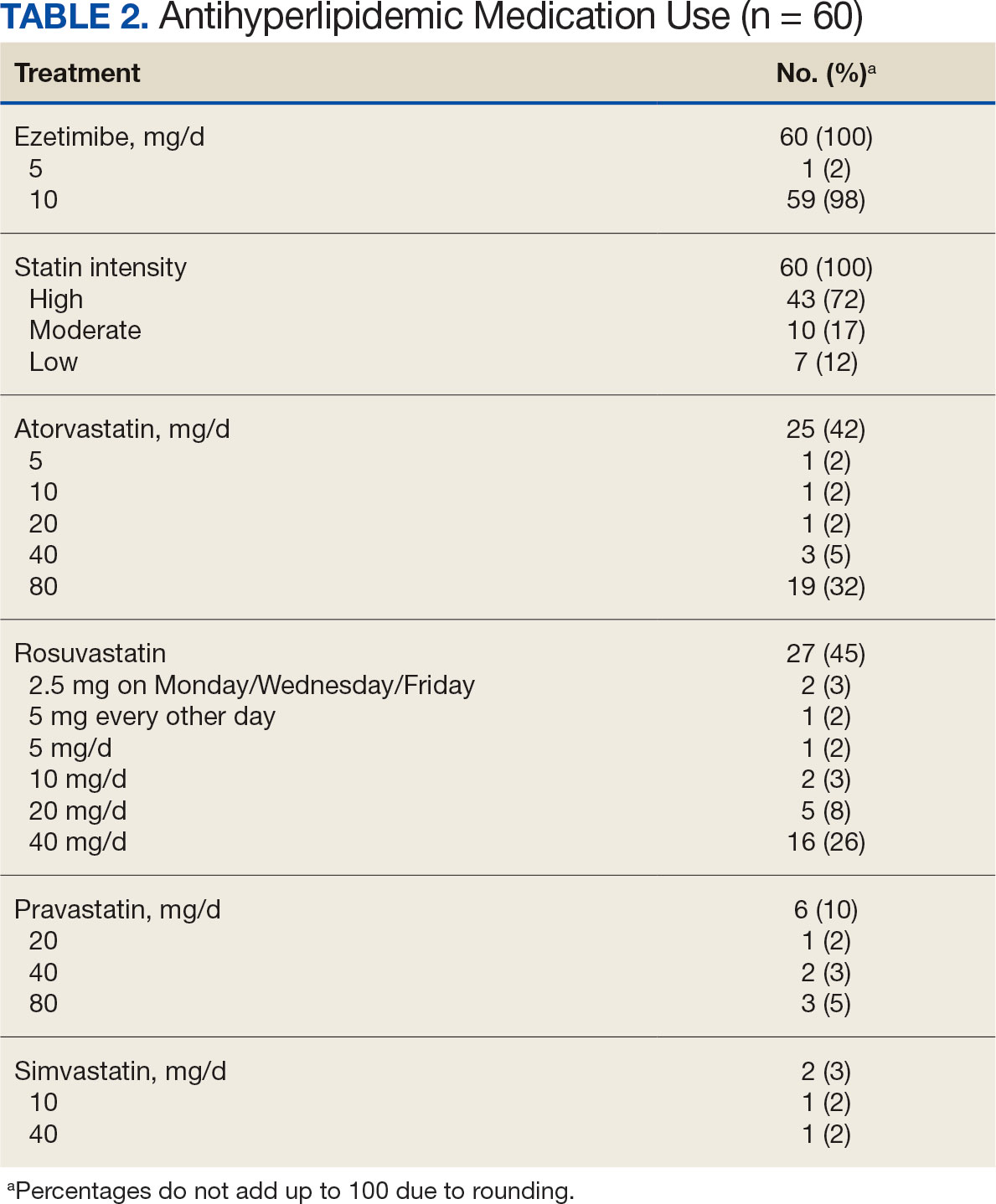
In the alirocumab monotherapy group, 15 patients (75%) were prescribed 75 mg every 2 weeks and 5 patients (25%) were prescribed 150 mg every 2 weeks. In the ezetimibe plus statin therapy group, 59 patients (98%) were prescribed ezetimibe 10 mg/d (Table 2). Forty-three patients (72%) were prescribed a high-intensity statin 10 received moderate-intensity (17%) and 7 received low-intensity statin (12%). Most patients were prescribed rosuvastatin (45%), followed by atorvastatin (42%), pravastatin (10%), and simvastatin (3%).
Primary Endpoint
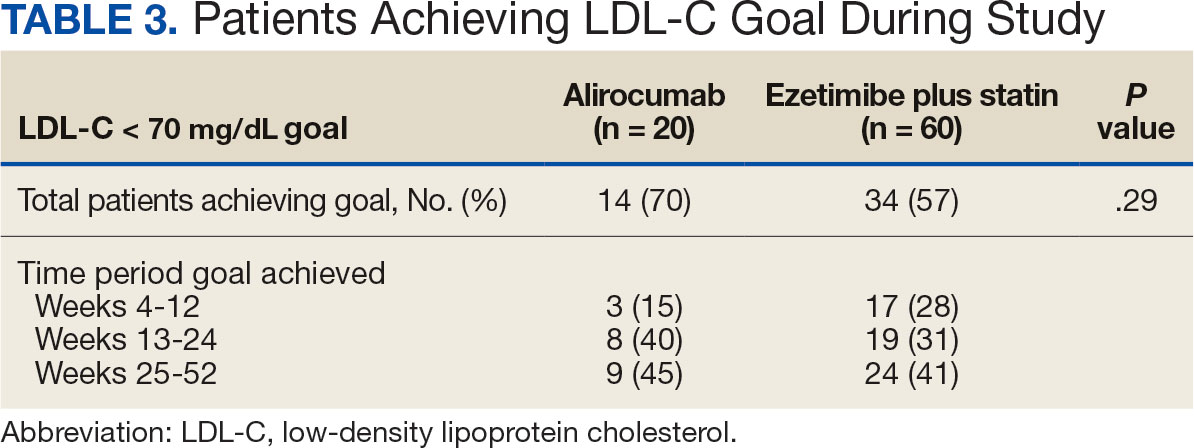
During the 52-week study, more patients met the LDL-C goal of < 70 mg/dL in the alirocumab monotherapy group (70%) than in the ezetimibe plus statin therapy group (57%); however, the difference was not significant (P = .29). Of the patients prescribed alirocumab monotherapy who achieved LDL-C < 70 mg/dL, 15% achieved this goal in 4 to 12 weeks, 40% in 13 to 24 weeks, and 45% in 25 to 52 weeks. In the ezetimibe plus statin therapy group, 28% of patients achieved LDL-C < 70 mg/dL in 4 to 12 weeks, 31% in 13 to 24 weeks, and 41% in 25 to 52 weeks (Table 3).
Secondary Endpoints
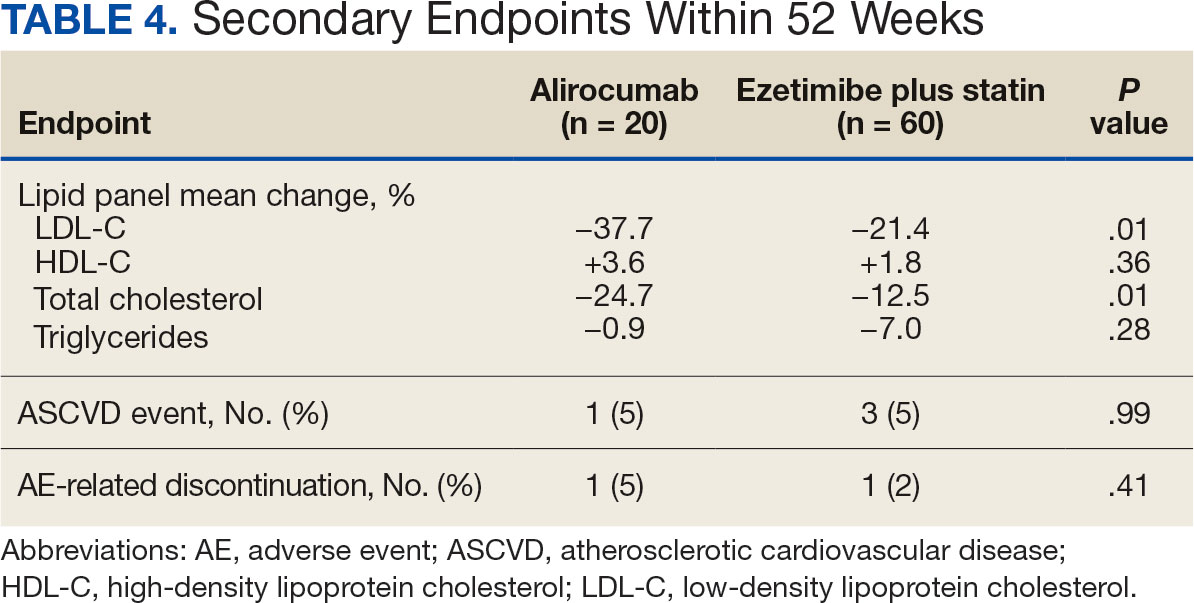
During weeks 4 to 52 of treatment, the mean percentage change decreased in LDL-C (37.7% vs 21.4%; P = .01), TC (24.7% vs 12.5%; P = .01), and TG (0.9% vs 7.0%; P = .28) in the alirocumab monotherapy group and the ezetimibe plus statin therapy group, respectively (Table 4). The mean percentage change increased in HDL-C by 3.6% in the alirocumab monotherapy group and 1.8% in the ezetimibe plus statin therapy group (P = .36). During the study, ASCVD events occurred in 1 patient (5%) in the alirocumab monotherapy group and 3 patients (5%) in the ezetimibe plus statin therapy group (P = .99). The patient in the alirocumab monotherapy group had unstable angina 1 month after taking alirocumab. One patient in the ezetimibe plus statin therapy group had coronary artery disease and 2 patients had coronary heart disease that required stents during the 52-week period. There was 1 patient in each group who reported an AE that led to treatment discontinuation (P = .41). One patient stopped alirocumab after a trial of 2 months due to intolerance, but no specific AE was reported in the CPRS. In the ezetimibe plus statin therapy group, 1 patient requested to discontinue ezetimibe after a trial of 3 months without a specific reason noted in the medical record.
DISCUSSION
This study found no statistically significant difference in the incidence of reaching an LDL-C goal of < 70 mg/dL after alirocumab monotherapy initiation compared with ezetimibe plus statin therapy. This occurred despite baseline LDL-C being lower in the ezetimibe plus statin therapy group, which required a smaller reduction in LDL-C to reach the primary goal. Most patients on alirocumab monotherapy were prescribed a lower initial dose of 75 mg every 2 weeks. Of those patients, 30% did not achieve the LDL-C goal < 70 mg/dL. Thus, a higher dose may have led to more patients achieving the LDL-C goal.
Secondary endpoints, including mean percentage change in HDL-C and TG and incidence of ASCVD events during 52 weeks of treatment, were not statistically significant. The mean percentage increase in HDL-C was negligible in both groups, while the mean percentage reduction in TG favored the ezetimibe plus statin therapy group. In the ezetimibe plus statin therapy group, patients who also took fenofibrate experienced a significant reduction in TG while none of the patients in the alirocumab group were prescribed fenofibrate. Although the alirocumab monotherapy group had a statistically significant greater reduction in LDL-C and TC compared with those prescribed ezetimibe plus statin, the mean baseline LDL-C and TC were significantly greater in the alirocumab monotherapy group, which could contribute to higher reductions in LDL-C and TC after alirocumab monotherapy.Based on the available literature, we expected greater reductions in LDL-C in both study groups compared with statin therapy alone.8,9 However, it was unclear whether the LDL-C and TC reductions were clinically significant.
Limitations
The study design did not permit randomization prior to the treatments, restricting our ability to account for some confounding factors, such as diet, exercise, other antihyperlipidemic medication, and medication adherence, which may have affected LDL-C, HDL-C, TG, and TC levels. Differences in baseline characteristics—particularly major risk factors, such as hypertension, diabetes, and tobacco use—also could have confounding affect on lipid levels and ASCVD events. Additionally, patients prescribed alirocumab monotherapy may have switched from statin or ezetimibe therapy, and the washout period was not reviewed or recorded, which could have affected the lipid panel results.
The small sample size of this study also may have limited the ability to detect significant differences between groups. A direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been performed, making it difficult to prospectively evaluate what sample size would be needed to power this study. A posthoc analysis was used to calculate power, which was found to be only 17%. Many patients were excluded due to a lack of laboratory results within the study period, contributing to the small sample size.
Another limitation was the reliance on documentation in CPRS and JLV. For example, having documentation of the specific AEs for the 2 patients who discontinued alirocumab or ezetimibe could have helped determine the severity of the AEs. Several patients were followed by non-VA clinicians, which could have contributed to limited documentation in the CPRS and JLV. It is difficult to draw any conclusions regarding ASCVD events and AEs that led to treatment discontinuation between alirocumab monotherapy and ezetimibe plus statin therapy based on the results of this retrospective study due to the limited number of events within the 52-week period.
CONCLUSIONS
This study found that there was no statistically significant difference in LDL-C reduction to < 70 mg/dL between alirocumab monotherapy and ezetimibe plus statin therapy in a small population of veterans with ASCVD, with a higher percentage of participants in both groups achieving that goal in 25 to 52 weeks. There also was no significant difference in percentage change in HDL-C or TG or in incidence of ASCVD events and AEs leading to treatment discontinuation. However, there was a statistically significant difference in percentage reduction for LDL-C and TC during 52 weeks of alirocumab monotherapy vs ezetimibe plus statin therapy.
Although there was no significant difference in LDL-C reduction to < 70 mg/dL, targeting this goal in patients with ASCVD is still clinically warranted. This study does not support a change in current VA criteria for use of alirocumab or a change in current guidelines for secondary prevention of ASCVD. Still, this study does indicate that the efficacy of alirocumab monotherapy is similar to that of ezetimibe plus statin therapy in patients with a history of ASCVD and may be useful in clinical settings when an alternative to ezetimibe plus statin therapy is needed. Alirocumab also may be more effective in lowering LDL-C and TC than ezetimibe plus statin therapy in veterans with ASCVD and could be added to statin therapy or ezetimibe when additional LDL-C or TC reduction is needed.
Lucchi T. Dyslipidemia and prevention of atherosclerotic cardiovascular disease in the elderly. Minerva Med. 2021;112:804-816. doi:10.23736/S0026-4806.21.07347-X
The Management of Dyslipidemia for Cardiovascular Risk Reduction Work Group. VA/DoD Clinical Practice Guideline for the Management of Dyslipidemia for Cardiovascular Risk Reduction. Version 4.0. June 2020. Accessed September 5, 2024. https://www.healthquality.va.gov/guidelines/CD/lipids/VADoDDyslipidemiaCPG5087212020.pdf
Centers for Disease Control and Prevention. High Cholesterol Facts. May 15, 2024. Accessed October 3, 2024. https://www.cdc.gov/cholesterol/data-research/facts-stats/index.html
Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143. doi:10.1161/CIR.0000000000000625
Vavlukis M, Vavlukis A. Statins alone or in combination with ezetimibe or PCSK9 inhibitors in atherosclerotic cardiovascular disease protection. IntechOpen. January 24, 2019. doi:10.5772/intechopen.82520
Alirocumab. Prescribing information. Regeneron Pharmaceuticals, Inc.; 2024. Accessed September 5, 2024. https://www.regeneron.com/downloads/praluent_pi.pdf
Pandor A, Ara RM, Tumur I, et al. Ezetimibe monotherapy for cholesterol lowering in 2,722 people: systematic review and meta-analysis of randomized controlled trials. J Intern Med. 2009;265(5):568-580. doi:10.1111/j.1365-2796.2008.02062.x
McKenney J, Ballantyne CM, Feldman TA, et al. LDL-C goal attainment with ezetimibe plus simvastatin coadministration vs atorvastatin or simvastatin monotherapy in patients at high risk of CHD. MedGenMed. 2005;7(3):3.
Stroes E, Guyton JR, Lepor N, et al. Efficacy and safety of alirocumab 150 mg every 4 weeks in patients with hypercholesterolemia not on statin therapy: the ODYSSEY CHOICE II study. J Am Heart Assoc. 2016;5(9):e003421. doi:10.1161/JAHA.116.003421
Atherosclerotic cardiovascular disease (ASCVD) is a significant cause of morbidity and mortality in the United States. ASCVD involves the buildup of cholesterol plaque in arteries and includes acute coronary syndrome, peripheral arterial disease, and events such as myocardial infarction and stroke.1 Cardiovascular disease (CVD) risk factors include high cholesterol levels, elevated blood pressure, insulin resistance, elevated blood glucose levels, smoking, poor dietary habits, and a sedentary lifestyle.2
According to the Centers for Disease Control and Prevention, about 86 million adults aged ≥ 20 years have total cholesterol levels > 200 mg/dL. More than half (54.5%) who could benefit are currently taking cholesterol-lowering medications.3 Controlling high cholesterol in American adults, especially veterans, is essential for reducing CVD morbidity and mortality.
The 2018 American College of Cardiology/American Heart Association (ACC/AHA) guideline recommends a low-density lipoprotein cholesterol (LDL-C) target goal of < 70 mg/dL for patients at high risk for ASCVD. Very high-risk ASCVD includes a history of multiple major ASCVD events or 1 major ASCVD event and multiple high-risk conditions (eg, age ≥ 65 years, smoking, or diabetes).4 Major ASCVD events include recent acute coronary syndrome (within the past 12 months), a history of myocardial infarction or ischemic stroke, and symptomatic peripheral artery disease.
The ACC/AHA guideline suggests that if the LDL-C level remains ≥ 70 mg/dL, adding ezetimibe (a dietary cholesterol absorption inhibitor) to maximally tolerated statin therapy is reasonable. If LDL-C levels remain ≥ 70 mg/dL, adding a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor, such as alirocumab, is reasonable.4 The US Departments of Veterans Affairs/US Department of Defense guidelines recommend using maximally tolerated statins and ezetimibe before PCSK9 inhibitors due to established long-term safety and reduction in CVD events.
Generic statins and ezetimibe are administered orally and widely available. In contrast, PCSK9 inhibitors have unknown long-term safety profiles, require subcutaneous injection once or twice monthly, and are significantly more expensive. They also require patient education on proper use while providing comparable or lesser relative risk reductions.2
These 3 classes of medication vary in their mechanisms of action to reduce LDL.5,6 Ezetimibe and several statin medications are included on the Veterans Affairs Sioux Falls Health Care System (VASFHCS) formulary and do not require review prior to prescribing. Alirocumab is available at VASFHCS but is restricted to patients with a history of ASCVD or a diagnosis of familial hypercholesterolemia, and who are receiving maximally tolerated statin and ezetimibe therapy but require further LDL-C lowering to reduce their ASCVD risk.
Studies have found ezetimibe monotherapy reduces LDL-C in patients with dyslipidemia by 18% after 12 weeks.7 One found that the percentage reduction in LDL-C was significantly greater (P < .001) with all doses of ezetimibe plus simvastatin (46% to 59%) compared with either atorvastatin 10 mg (37%) or simvastatin 20 mg (38%) monotherapy after 6 weeks.8
Although alirocumab can be added to other lipid therapies, most VASFHCS patients are prescribed alirocumab monotherapy. In the ODYSSEY CHOICE II study, patients were randomly assigned to receive either a placebo or alirocumab 150 mg every 4 weeks or alirocumab 75 mg every 2 weeks. The primary efficacy endpoint was LDL-C percentage change from baseline to week 24. In the alirocumab 150 mg every 4 weeks and 75 mg every 2 weeks groups, the least-squares mean LDL-C changes from baseline to week 24 were 51.7% and 53.5%, respectively, compared to a 4.7% increase in the placebo group (both groups P < .001 vs placebo). The authors also reported that alirocumab 150 mg every 4 weeks as monotherapy demonstrated a 47.4% reduction in LDL-C levels from baseline in a phase 1 study.9Although alirocumab monotherapy and ezetimibe plus statin therapy have been shown to effectively decrease LDL-C independently, a direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been assessed, to our knowledge. Understanding the differences in effectiveness and safety between these 2 regimens will be valuable for clinicians when selecting a medication regimen for veterans with a history of ASCVD.
METHODS
This retrospective, single-center chart review used VASFHCS Computerized Patient Record System (CPRS) and Joint Longitudinal Viewer (JLV) records to compare patients with a history of ASCVD events who were treated with alirocumab monotherapy or ezetimibe plus statin. The 2 groups were randomized in a 1:3 ratio. The primary endpoint was achieving LDL-C < 70 mg/dL after 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks. Secondary endpoints included the mean percentage change from baseline in total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), LDL-C, and triglycerides (TG) over 52 weeks. The incidence of ASCVD events during this period was also assessed. If LDL-C < 70 mg/dL was achieved > 1 time during each time frame, only 1 incident was counted for analysis. Safety was assessed based on the incidence of any adverse event (AE) that led to treatment discontinuation.
Patients were identified by screening the prescription fill history between October 1, 2019, and December 31, 2022. The 52-week data collection period was counted from the first available fill date. Additionally, the prior authorization drug request file from January 1, 2017, to December 31, 2022, was used to obtain a list of patients prescribed alirocumab. Patients were included if they were veterans aged ≥ 18 years and had a history of an ASCVD event, had a alirocumab monotherapy or ezetimibe plus statin prescription between October 1, 2019, and December 31, 2022, or had an approved prior authorization drug request for alirocumab between January 1, 2017, and December 31, 2022. Patients missing a baseline or follow-up lipid panel and those with concurrent use of alirocumab and ezetimibe and/or statin were excluded.
Baseline characteristics collected for patients included age, sex, race, weight, body mass index, lipid parameters (LDL-C, TC, HDL-C, and TG), dosing of each type of statin before adding ezetimibe, and use of any other antihyperlipidemic medication. We also collected histories of hypertension, hyperlipidemia, diabetes, chronic kidney disease, congestive heart failure, and smoking or tobacco use status. The baseline lipid panel was the most recent lipid panel documented before starting alirocumab or ezetimibe plus statin therapy. Follow-up lipid panel values were gathered at 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks following initiation of either therapy.
High-, moderate-, and low-intensity dosing of statin therapy and alirocumab dosing (75 mg every 2 weeks, 150 mg every 2 weeks, or 300 mg every 4 weeks) were recorded at the specified intervals. However, no patients in this study received the latter dosing regimen. ASCVD events and safety endpoints were recorded based on a review of clinical notes over the 52 weeks following the first available start date.
Statistical Analysis
The primary endpoint of achieving the LDL-C < 70 mg/dL goal from baseline to 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks after initiation was compared between alirocumab monotherapy and ezetimibe plus statin therapy using the χ² test. Mean percentage change from baseline in TC, HDL-C, LDL-C, and TG were compared using the independent t test. P < .05 was considered statistically significant. Incidence of ASCVD events and the safety endpoint (incidence of AEs leading to treatment discontinuation) were also compared using the χ² test. Continuous baseline characteristics were reported mean (SD) and nominal baseline characteristics were reported as a percentage.
RESULTS
There were 80 participants in this study: 20 in the alirocumab monotherapy group and 60 in the ezetimibe plus statin therapy group. More than 100 patients did not meet the prespecified inclusion criteria and were excluded. Mean (SD) age was 75 (8) years in the alirocumab group and 74 (8) years in the ezetimibe plus statin group. There was no significant differences in mean (SD) weight or mean (SD) body mass index. All study participants identified as White and male except for 2 patients in the ezetimibe plus statin therapy group whose race was not documented. Differences in lipid parameters were observed between groups, with mean baseline LDL-C, HDL-C, and TC higher in the alirocumab monotherapy group than in the ezetimibe plus statin therapy group, with significant differences in LDL-C and TC (Table 1).

Fourteen patients (70%) in the alirocumab monotherapy group had hypertension, compared with 31 (52%) in the ezetimibe plus statin therapy group. In both groups, most patients had previously been diagnosed with hyperlipidemia. More patients (60%) in the alirocumab group had diabetes than in the ezetimibe plus statin therapy group (37%). The alirocumab monotherapy group also had a higher percentage of patients with diagnoses of congestive heart failure and used other antihyperlipidemic medications than in the ezetimibe plus statin therapy group. Five patients (25%) in the alirocumab monotherapy group and 12 patients (20%) in the ezetimibe plus statin therapy group took fish oil. In the ezetimibe plus statin therapy group, 2 patients (3%) took gemfibrozil, and 2 patients (3%) took fenofibrate. Six (30%) patients in the alirocumab monotherapy group and 12 (20%) patients in the ezetimibe plus statin therapy group had chronic kidney disease. Although the majority of patients in each group did not use tobacco products, there were more tobacco users in the ezetimibe plus statin therapy group.
In the alirocumab monotherapy group, 15 patients (75%) were prescribed 75 mg every 2 weeks and 5 patients (25%) were prescribed 150 mg every 2 weeks. In the ezetimibe plus statin therapy group, 59 patients (98%) were prescribed ezetimibe 10 mg/d (Table 2). Forty-three patients (72%) were prescribed a high-intensity statin 10 received moderate-intensity (17%) and 7 received low-intensity statin (12%). Most patients were prescribed rosuvastatin (45%), followed by atorvastatin (42%), pravastatin (10%), and simvastatin (3%).

Primary Endpoint
During the 52-week study, more patients met the LDL-C goal of < 70 mg/dL in the alirocumab monotherapy group (70%) than in the ezetimibe plus statin therapy group (57%); however, the difference was not significant (P = .29). Of the patients prescribed alirocumab monotherapy who achieved LDL-C < 70 mg/dL, 15% achieved this goal in 4 to 12 weeks, 40% in 13 to 24 weeks, and 45% in 25 to 52 weeks. In the ezetimibe plus statin therapy group, 28% of patients achieved LDL-C < 70 mg/dL in 4 to 12 weeks, 31% in 13 to 24 weeks, and 41% in 25 to 52 weeks (Table 3).

Secondary Endpoints
During weeks 4 to 52 of treatment, the mean percentage change decreased in LDL-C (37.7% vs 21.4%; P = .01), TC (24.7% vs 12.5%; P = .01), and TG (0.9% vs 7.0%; P = .28) in the alirocumab monotherapy group and the ezetimibe plus statin therapy group, respectively (Table 4). The mean percentage change increased in HDL-C by 3.6% in the alirocumab monotherapy group and 1.8% in the ezetimibe plus statin therapy group (P = .36). During the study, ASCVD events occurred in 1 patient (5%) in the alirocumab monotherapy group and 3 patients (5%) in the ezetimibe plus statin therapy group (P = .99). The patient in the alirocumab monotherapy group had unstable angina 1 month after taking alirocumab. One patient in the ezetimibe plus statin therapy group had coronary artery disease and 2 patients had coronary heart disease that required stents during the 52-week period. There was 1 patient in each group who reported an AE that led to treatment discontinuation (P = .41). One patient stopped alirocumab after a trial of 2 months due to intolerance, but no specific AE was reported in the CPRS. In the ezetimibe plus statin therapy group, 1 patient requested to discontinue ezetimibe after a trial of 3 months without a specific reason noted in the medical record.

DISCUSSION
This study found no statistically significant difference in the incidence of reaching an LDL-C goal of < 70 mg/dL after alirocumab monotherapy initiation compared with ezetimibe plus statin therapy. This occurred despite baseline LDL-C being lower in the ezetimibe plus statin therapy group, which required a smaller reduction in LDL-C to reach the primary goal. Most patients on alirocumab monotherapy were prescribed a lower initial dose of 75 mg every 2 weeks. Of those patients, 30% did not achieve the LDL-C goal < 70 mg/dL. Thus, a higher dose may have led to more patients achieving the LDL-C goal.
Secondary endpoints, including mean percentage change in HDL-C and TG and incidence of ASCVD events during 52 weeks of treatment, were not statistically significant. The mean percentage increase in HDL-C was negligible in both groups, while the mean percentage reduction in TG favored the ezetimibe plus statin therapy group. In the ezetimibe plus statin therapy group, patients who also took fenofibrate experienced a significant reduction in TG while none of the patients in the alirocumab group were prescribed fenofibrate. Although the alirocumab monotherapy group had a statistically significant greater reduction in LDL-C and TC compared with those prescribed ezetimibe plus statin, the mean baseline LDL-C and TC were significantly greater in the alirocumab monotherapy group, which could contribute to higher reductions in LDL-C and TC after alirocumab monotherapy.Based on the available literature, we expected greater reductions in LDL-C in both study groups compared with statin therapy alone.8,9 However, it was unclear whether the LDL-C and TC reductions were clinically significant.
Limitations
The study design did not permit randomization prior to the treatments, restricting our ability to account for some confounding factors, such as diet, exercise, other antihyperlipidemic medication, and medication adherence, which may have affected LDL-C, HDL-C, TG, and TC levels. Differences in baseline characteristics—particularly major risk factors, such as hypertension, diabetes, and tobacco use—also could have confounding affect on lipid levels and ASCVD events. Additionally, patients prescribed alirocumab monotherapy may have switched from statin or ezetimibe therapy, and the washout period was not reviewed or recorded, which could have affected the lipid panel results.
The small sample size of this study also may have limited the ability to detect significant differences between groups. A direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been performed, making it difficult to prospectively evaluate what sample size would be needed to power this study. A posthoc analysis was used to calculate power, which was found to be only 17%. Many patients were excluded due to a lack of laboratory results within the study period, contributing to the small sample size.
Another limitation was the reliance on documentation in CPRS and JLV. For example, having documentation of the specific AEs for the 2 patients who discontinued alirocumab or ezetimibe could have helped determine the severity of the AEs. Several patients were followed by non-VA clinicians, which could have contributed to limited documentation in the CPRS and JLV. It is difficult to draw any conclusions regarding ASCVD events and AEs that led to treatment discontinuation between alirocumab monotherapy and ezetimibe plus statin therapy based on the results of this retrospective study due to the limited number of events within the 52-week period.
CONCLUSIONS
This study found that there was no statistically significant difference in LDL-C reduction to < 70 mg/dL between alirocumab monotherapy and ezetimibe plus statin therapy in a small population of veterans with ASCVD, with a higher percentage of participants in both groups achieving that goal in 25 to 52 weeks. There also was no significant difference in percentage change in HDL-C or TG or in incidence of ASCVD events and AEs leading to treatment discontinuation. However, there was a statistically significant difference in percentage reduction for LDL-C and TC during 52 weeks of alirocumab monotherapy vs ezetimibe plus statin therapy.
Although there was no significant difference in LDL-C reduction to < 70 mg/dL, targeting this goal in patients with ASCVD is still clinically warranted. This study does not support a change in current VA criteria for use of alirocumab or a change in current guidelines for secondary prevention of ASCVD. Still, this study does indicate that the efficacy of alirocumab monotherapy is similar to that of ezetimibe plus statin therapy in patients with a history of ASCVD and may be useful in clinical settings when an alternative to ezetimibe plus statin therapy is needed. Alirocumab also may be more effective in lowering LDL-C and TC than ezetimibe plus statin therapy in veterans with ASCVD and could be added to statin therapy or ezetimibe when additional LDL-C or TC reduction is needed.
Atherosclerotic cardiovascular disease (ASCVD) is a significant cause of morbidity and mortality in the United States. ASCVD involves the buildup of cholesterol plaque in arteries and includes acute coronary syndrome, peripheral arterial disease, and events such as myocardial infarction and stroke.1 Cardiovascular disease (CVD) risk factors include high cholesterol levels, elevated blood pressure, insulin resistance, elevated blood glucose levels, smoking, poor dietary habits, and a sedentary lifestyle.2
According to the Centers for Disease Control and Prevention, about 86 million adults aged ≥ 20 years have total cholesterol levels > 200 mg/dL. More than half (54.5%) who could benefit are currently taking cholesterol-lowering medications.3 Controlling high cholesterol in American adults, especially veterans, is essential for reducing CVD morbidity and mortality.
The 2018 American College of Cardiology/American Heart Association (ACC/AHA) guideline recommends a low-density lipoprotein cholesterol (LDL-C) target goal of < 70 mg/dL for patients at high risk for ASCVD. Very high-risk ASCVD includes a history of multiple major ASCVD events or 1 major ASCVD event and multiple high-risk conditions (eg, age ≥ 65 years, smoking, or diabetes).4 Major ASCVD events include recent acute coronary syndrome (within the past 12 months), a history of myocardial infarction or ischemic stroke, and symptomatic peripheral artery disease.
The ACC/AHA guideline suggests that if the LDL-C level remains ≥ 70 mg/dL, adding ezetimibe (a dietary cholesterol absorption inhibitor) to maximally tolerated statin therapy is reasonable. If LDL-C levels remain ≥ 70 mg/dL, adding a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor, such as alirocumab, is reasonable.4 The US Departments of Veterans Affairs/US Department of Defense guidelines recommend using maximally tolerated statins and ezetimibe before PCSK9 inhibitors due to established long-term safety and reduction in CVD events.
Generic statins and ezetimibe are administered orally and widely available. In contrast, PCSK9 inhibitors have unknown long-term safety profiles, require subcutaneous injection once or twice monthly, and are significantly more expensive. They also require patient education on proper use while providing comparable or lesser relative risk reductions.2
These 3 classes of medication vary in their mechanisms of action to reduce LDL.5,6 Ezetimibe and several statin medications are included on the Veterans Affairs Sioux Falls Health Care System (VASFHCS) formulary and do not require review prior to prescribing. Alirocumab is available at VASFHCS but is restricted to patients with a history of ASCVD or a diagnosis of familial hypercholesterolemia, and who are receiving maximally tolerated statin and ezetimibe therapy but require further LDL-C lowering to reduce their ASCVD risk.
Studies have found ezetimibe monotherapy reduces LDL-C in patients with dyslipidemia by 18% after 12 weeks.7 One found that the percentage reduction in LDL-C was significantly greater (P < .001) with all doses of ezetimibe plus simvastatin (46% to 59%) compared with either atorvastatin 10 mg (37%) or simvastatin 20 mg (38%) monotherapy after 6 weeks.8
Although alirocumab can be added to other lipid therapies, most VASFHCS patients are prescribed alirocumab monotherapy. In the ODYSSEY CHOICE II study, patients were randomly assigned to receive either a placebo or alirocumab 150 mg every 4 weeks or alirocumab 75 mg every 2 weeks. The primary efficacy endpoint was LDL-C percentage change from baseline to week 24. In the alirocumab 150 mg every 4 weeks and 75 mg every 2 weeks groups, the least-squares mean LDL-C changes from baseline to week 24 were 51.7% and 53.5%, respectively, compared to a 4.7% increase in the placebo group (both groups P < .001 vs placebo). The authors also reported that alirocumab 150 mg every 4 weeks as monotherapy demonstrated a 47.4% reduction in LDL-C levels from baseline in a phase 1 study.9Although alirocumab monotherapy and ezetimibe plus statin therapy have been shown to effectively decrease LDL-C independently, a direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been assessed, to our knowledge. Understanding the differences in effectiveness and safety between these 2 regimens will be valuable for clinicians when selecting a medication regimen for veterans with a history of ASCVD.
METHODS
This retrospective, single-center chart review used VASFHCS Computerized Patient Record System (CPRS) and Joint Longitudinal Viewer (JLV) records to compare patients with a history of ASCVD events who were treated with alirocumab monotherapy or ezetimibe plus statin. The 2 groups were randomized in a 1:3 ratio. The primary endpoint was achieving LDL-C < 70 mg/dL after 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks. Secondary endpoints included the mean percentage change from baseline in total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), LDL-C, and triglycerides (TG) over 52 weeks. The incidence of ASCVD events during this period was also assessed. If LDL-C < 70 mg/dL was achieved > 1 time during each time frame, only 1 incident was counted for analysis. Safety was assessed based on the incidence of any adverse event (AE) that led to treatment discontinuation.
Patients were identified by screening the prescription fill history between October 1, 2019, and December 31, 2022. The 52-week data collection period was counted from the first available fill date. Additionally, the prior authorization drug request file from January 1, 2017, to December 31, 2022, was used to obtain a list of patients prescribed alirocumab. Patients were included if they were veterans aged ≥ 18 years and had a history of an ASCVD event, had a alirocumab monotherapy or ezetimibe plus statin prescription between October 1, 2019, and December 31, 2022, or had an approved prior authorization drug request for alirocumab between January 1, 2017, and December 31, 2022. Patients missing a baseline or follow-up lipid panel and those with concurrent use of alirocumab and ezetimibe and/or statin were excluded.
Baseline characteristics collected for patients included age, sex, race, weight, body mass index, lipid parameters (LDL-C, TC, HDL-C, and TG), dosing of each type of statin before adding ezetimibe, and use of any other antihyperlipidemic medication. We also collected histories of hypertension, hyperlipidemia, diabetes, chronic kidney disease, congestive heart failure, and smoking or tobacco use status. The baseline lipid panel was the most recent lipid panel documented before starting alirocumab or ezetimibe plus statin therapy. Follow-up lipid panel values were gathered at 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks following initiation of either therapy.
High-, moderate-, and low-intensity dosing of statin therapy and alirocumab dosing (75 mg every 2 weeks, 150 mg every 2 weeks, or 300 mg every 4 weeks) were recorded at the specified intervals. However, no patients in this study received the latter dosing regimen. ASCVD events and safety endpoints were recorded based on a review of clinical notes over the 52 weeks following the first available start date.
Statistical Analysis
The primary endpoint of achieving the LDL-C < 70 mg/dL goal from baseline to 4 to 12 weeks, 13 to 24 weeks, and 25 to 52 weeks after initiation was compared between alirocumab monotherapy and ezetimibe plus statin therapy using the χ² test. Mean percentage change from baseline in TC, HDL-C, LDL-C, and TG were compared using the independent t test. P < .05 was considered statistically significant. Incidence of ASCVD events and the safety endpoint (incidence of AEs leading to treatment discontinuation) were also compared using the χ² test. Continuous baseline characteristics were reported mean (SD) and nominal baseline characteristics were reported as a percentage.
RESULTS
There were 80 participants in this study: 20 in the alirocumab monotherapy group and 60 in the ezetimibe plus statin therapy group. More than 100 patients did not meet the prespecified inclusion criteria and were excluded. Mean (SD) age was 75 (8) years in the alirocumab group and 74 (8) years in the ezetimibe plus statin group. There was no significant differences in mean (SD) weight or mean (SD) body mass index. All study participants identified as White and male except for 2 patients in the ezetimibe plus statin therapy group whose race was not documented. Differences in lipid parameters were observed between groups, with mean baseline LDL-C, HDL-C, and TC higher in the alirocumab monotherapy group than in the ezetimibe plus statin therapy group, with significant differences in LDL-C and TC (Table 1).

Fourteen patients (70%) in the alirocumab monotherapy group had hypertension, compared with 31 (52%) in the ezetimibe plus statin therapy group. In both groups, most patients had previously been diagnosed with hyperlipidemia. More patients (60%) in the alirocumab group had diabetes than in the ezetimibe plus statin therapy group (37%). The alirocumab monotherapy group also had a higher percentage of patients with diagnoses of congestive heart failure and used other antihyperlipidemic medications than in the ezetimibe plus statin therapy group. Five patients (25%) in the alirocumab monotherapy group and 12 patients (20%) in the ezetimibe plus statin therapy group took fish oil. In the ezetimibe plus statin therapy group, 2 patients (3%) took gemfibrozil, and 2 patients (3%) took fenofibrate. Six (30%) patients in the alirocumab monotherapy group and 12 (20%) patients in the ezetimibe plus statin therapy group had chronic kidney disease. Although the majority of patients in each group did not use tobacco products, there were more tobacco users in the ezetimibe plus statin therapy group.
In the alirocumab monotherapy group, 15 patients (75%) were prescribed 75 mg every 2 weeks and 5 patients (25%) were prescribed 150 mg every 2 weeks. In the ezetimibe plus statin therapy group, 59 patients (98%) were prescribed ezetimibe 10 mg/d (Table 2). Forty-three patients (72%) were prescribed a high-intensity statin 10 received moderate-intensity (17%) and 7 received low-intensity statin (12%). Most patients were prescribed rosuvastatin (45%), followed by atorvastatin (42%), pravastatin (10%), and simvastatin (3%).

Primary Endpoint
During the 52-week study, more patients met the LDL-C goal of < 70 mg/dL in the alirocumab monotherapy group (70%) than in the ezetimibe plus statin therapy group (57%); however, the difference was not significant (P = .29). Of the patients prescribed alirocumab monotherapy who achieved LDL-C < 70 mg/dL, 15% achieved this goal in 4 to 12 weeks, 40% in 13 to 24 weeks, and 45% in 25 to 52 weeks. In the ezetimibe plus statin therapy group, 28% of patients achieved LDL-C < 70 mg/dL in 4 to 12 weeks, 31% in 13 to 24 weeks, and 41% in 25 to 52 weeks (Table 3).

Secondary Endpoints
During weeks 4 to 52 of treatment, the mean percentage change decreased in LDL-C (37.7% vs 21.4%; P = .01), TC (24.7% vs 12.5%; P = .01), and TG (0.9% vs 7.0%; P = .28) in the alirocumab monotherapy group and the ezetimibe plus statin therapy group, respectively (Table 4). The mean percentage change increased in HDL-C by 3.6% in the alirocumab monotherapy group and 1.8% in the ezetimibe plus statin therapy group (P = .36). During the study, ASCVD events occurred in 1 patient (5%) in the alirocumab monotherapy group and 3 patients (5%) in the ezetimibe plus statin therapy group (P = .99). The patient in the alirocumab monotherapy group had unstable angina 1 month after taking alirocumab. One patient in the ezetimibe plus statin therapy group had coronary artery disease and 2 patients had coronary heart disease that required stents during the 52-week period. There was 1 patient in each group who reported an AE that led to treatment discontinuation (P = .41). One patient stopped alirocumab after a trial of 2 months due to intolerance, but no specific AE was reported in the CPRS. In the ezetimibe plus statin therapy group, 1 patient requested to discontinue ezetimibe after a trial of 3 months without a specific reason noted in the medical record.

DISCUSSION
This study found no statistically significant difference in the incidence of reaching an LDL-C goal of < 70 mg/dL after alirocumab monotherapy initiation compared with ezetimibe plus statin therapy. This occurred despite baseline LDL-C being lower in the ezetimibe plus statin therapy group, which required a smaller reduction in LDL-C to reach the primary goal. Most patients on alirocumab monotherapy were prescribed a lower initial dose of 75 mg every 2 weeks. Of those patients, 30% did not achieve the LDL-C goal < 70 mg/dL. Thus, a higher dose may have led to more patients achieving the LDL-C goal.
Secondary endpoints, including mean percentage change in HDL-C and TG and incidence of ASCVD events during 52 weeks of treatment, were not statistically significant. The mean percentage increase in HDL-C was negligible in both groups, while the mean percentage reduction in TG favored the ezetimibe plus statin therapy group. In the ezetimibe plus statin therapy group, patients who also took fenofibrate experienced a significant reduction in TG while none of the patients in the alirocumab group were prescribed fenofibrate. Although the alirocumab monotherapy group had a statistically significant greater reduction in LDL-C and TC compared with those prescribed ezetimibe plus statin, the mean baseline LDL-C and TC were significantly greater in the alirocumab monotherapy group, which could contribute to higher reductions in LDL-C and TC after alirocumab monotherapy.Based on the available literature, we expected greater reductions in LDL-C in both study groups compared with statin therapy alone.8,9 However, it was unclear whether the LDL-C and TC reductions were clinically significant.
Limitations
The study design did not permit randomization prior to the treatments, restricting our ability to account for some confounding factors, such as diet, exercise, other antihyperlipidemic medication, and medication adherence, which may have affected LDL-C, HDL-C, TG, and TC levels. Differences in baseline characteristics—particularly major risk factors, such as hypertension, diabetes, and tobacco use—also could have confounding affect on lipid levels and ASCVD events. Additionally, patients prescribed alirocumab monotherapy may have switched from statin or ezetimibe therapy, and the washout period was not reviewed or recorded, which could have affected the lipid panel results.
The small sample size of this study also may have limited the ability to detect significant differences between groups. A direct comparison of alirocumab monotherapy vs ezetimibe plus statin therapy has not been performed, making it difficult to prospectively evaluate what sample size would be needed to power this study. A posthoc analysis was used to calculate power, which was found to be only 17%. Many patients were excluded due to a lack of laboratory results within the study period, contributing to the small sample size.
Another limitation was the reliance on documentation in CPRS and JLV. For example, having documentation of the specific AEs for the 2 patients who discontinued alirocumab or ezetimibe could have helped determine the severity of the AEs. Several patients were followed by non-VA clinicians, which could have contributed to limited documentation in the CPRS and JLV. It is difficult to draw any conclusions regarding ASCVD events and AEs that led to treatment discontinuation between alirocumab monotherapy and ezetimibe plus statin therapy based on the results of this retrospective study due to the limited number of events within the 52-week period.
CONCLUSIONS
This study found that there was no statistically significant difference in LDL-C reduction to < 70 mg/dL between alirocumab monotherapy and ezetimibe plus statin therapy in a small population of veterans with ASCVD, with a higher percentage of participants in both groups achieving that goal in 25 to 52 weeks. There also was no significant difference in percentage change in HDL-C or TG or in incidence of ASCVD events and AEs leading to treatment discontinuation. However, there was a statistically significant difference in percentage reduction for LDL-C and TC during 52 weeks of alirocumab monotherapy vs ezetimibe plus statin therapy.
Although there was no significant difference in LDL-C reduction to < 70 mg/dL, targeting this goal in patients with ASCVD is still clinically warranted. This study does not support a change in current VA criteria for use of alirocumab or a change in current guidelines for secondary prevention of ASCVD. Still, this study does indicate that the efficacy of alirocumab monotherapy is similar to that of ezetimibe plus statin therapy in patients with a history of ASCVD and may be useful in clinical settings when an alternative to ezetimibe plus statin therapy is needed. Alirocumab also may be more effective in lowering LDL-C and TC than ezetimibe plus statin therapy in veterans with ASCVD and could be added to statin therapy or ezetimibe when additional LDL-C or TC reduction is needed.
Lucchi T. Dyslipidemia and prevention of atherosclerotic cardiovascular disease in the elderly. Minerva Med. 2021;112:804-816. doi:10.23736/S0026-4806.21.07347-X
The Management of Dyslipidemia for Cardiovascular Risk Reduction Work Group. VA/DoD Clinical Practice Guideline for the Management of Dyslipidemia for Cardiovascular Risk Reduction. Version 4.0. June 2020. Accessed September 5, 2024. https://www.healthquality.va.gov/guidelines/CD/lipids/VADoDDyslipidemiaCPG5087212020.pdf
Centers for Disease Control and Prevention. High Cholesterol Facts. May 15, 2024. Accessed October 3, 2024. https://www.cdc.gov/cholesterol/data-research/facts-stats/index.html
Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143. doi:10.1161/CIR.0000000000000625
Vavlukis M, Vavlukis A. Statins alone or in combination with ezetimibe or PCSK9 inhibitors in atherosclerotic cardiovascular disease protection. IntechOpen. January 24, 2019. doi:10.5772/intechopen.82520
Alirocumab. Prescribing information. Regeneron Pharmaceuticals, Inc.; 2024. Accessed September 5, 2024. https://www.regeneron.com/downloads/praluent_pi.pdf
Pandor A, Ara RM, Tumur I, et al. Ezetimibe monotherapy for cholesterol lowering in 2,722 people: systematic review and meta-analysis of randomized controlled trials. J Intern Med. 2009;265(5):568-580. doi:10.1111/j.1365-2796.2008.02062.x
McKenney J, Ballantyne CM, Feldman TA, et al. LDL-C goal attainment with ezetimibe plus simvastatin coadministration vs atorvastatin or simvastatin monotherapy in patients at high risk of CHD. MedGenMed. 2005;7(3):3.
Stroes E, Guyton JR, Lepor N, et al. Efficacy and safety of alirocumab 150 mg every 4 weeks in patients with hypercholesterolemia not on statin therapy: the ODYSSEY CHOICE II study. J Am Heart Assoc. 2016;5(9):e003421. doi:10.1161/JAHA.116.003421
Lucchi T. Dyslipidemia and prevention of atherosclerotic cardiovascular disease in the elderly. Minerva Med. 2021;112:804-816. doi:10.23736/S0026-4806.21.07347-X
The Management of Dyslipidemia for Cardiovascular Risk Reduction Work Group. VA/DoD Clinical Practice Guideline for the Management of Dyslipidemia for Cardiovascular Risk Reduction. Version 4.0. June 2020. Accessed September 5, 2024. https://www.healthquality.va.gov/guidelines/CD/lipids/VADoDDyslipidemiaCPG5087212020.pdf
Centers for Disease Control and Prevention. High Cholesterol Facts. May 15, 2024. Accessed October 3, 2024. https://www.cdc.gov/cholesterol/data-research/facts-stats/index.html
Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139:e1082-e1143. doi:10.1161/CIR.0000000000000625
Vavlukis M, Vavlukis A. Statins alone or in combination with ezetimibe or PCSK9 inhibitors in atherosclerotic cardiovascular disease protection. IntechOpen. January 24, 2019. doi:10.5772/intechopen.82520
Alirocumab. Prescribing information. Regeneron Pharmaceuticals, Inc.; 2024. Accessed September 5, 2024. https://www.regeneron.com/downloads/praluent_pi.pdf
Pandor A, Ara RM, Tumur I, et al. Ezetimibe monotherapy for cholesterol lowering in 2,722 people: systematic review and meta-analysis of randomized controlled trials. J Intern Med. 2009;265(5):568-580. doi:10.1111/j.1365-2796.2008.02062.x
McKenney J, Ballantyne CM, Feldman TA, et al. LDL-C goal attainment with ezetimibe plus simvastatin coadministration vs atorvastatin or simvastatin monotherapy in patients at high risk of CHD. MedGenMed. 2005;7(3):3.
Stroes E, Guyton JR, Lepor N, et al. Efficacy and safety of alirocumab 150 mg every 4 weeks in patients with hypercholesterolemia not on statin therapy: the ODYSSEY CHOICE II study. J Am Heart Assoc. 2016;5(9):e003421. doi:10.1161/JAHA.116.003421
Sitting for More Than 10 Hours Daily Ups Heart Disease Risk
TOPLINE:
Sedentary time exceeding 10.6 h/d is linked to an increased risk for atrial fibrillation, heart failure, myocardial infarction, and cardiovascular (CV) mortality, researchers found. The risk persists even in individuals who meet recommended physical activity levels.
METHODOLOGY:
- Researchers used a validated machine learning approach to investigate the relationships between sedentary behavior and the future risks for CV illness and mortality in 89,530 middle-aged and older adults (mean age, 62 years; 56% women) from the UK Biobank.
- Participants provided data from a wrist-worn triaxial accelerometer that recorded their movements over a period of 7 days.
- Machine learning algorithms classified accelerometer signals into four classes of activities: Sleep, sedentary behavior, light physical activity, and moderate to vigorous physical activity.
- Participants were followed up for a median of 8 years through linkage to national health-related datasets in England, Scotland, and Wales.
- The median sedentary time was 9.4 h/d.
TAKEAWAY:
- During the follow-up period, 3638 individuals (4.9%) experienced incident atrial fibrillation, 1854 (2.09%) developed incident heart failure, 1610 (1.84%) experienced incident myocardial infarction, and 846 (0.94%) died from cardiovascular causes.
- The risks for atrial fibrillation and myocardial infarction increased steadily with an increase in sedentary time, with sedentary time greater than 10.6 h/d showing a modest increase in risk for atrial fibrillation (hazard ratio [HR], 1.11; 95% CI, 1.01-1.21).
- The risks for heart failure and CV mortality were low until sedentary time surpassed approximately 10.6 h/d, after which they rose by 45% (HR, 1.45; 95% CI, 1.28-1.65) and 62% (HR, 1.62; 95% CI, 1.34-1.96), respectively.
- The associations were attenuated but remained significant for CV mortality (HR, 1.33; 95% CI: 1.07-1.64) in individuals who met the recommended levels for physical activity yet were sedentary for more than 10.6 h/d. Reallocating 30 minutes of sedentary time to other activities reduced the risk for heart failure (HR, 0.93; 95% CI, 0.90-0.96) among those who were sedentary more than 10.6 h/d.
IN PRACTICE:
The study “highlights a complex interplay between sedentary behavior and physical activity, ultimately suggesting that sedentary behavior remains relevant for CV disease risk even among individuals meeting sufficient” levels of activity, the researchers reported.
“Individuals should move more and be less sedentary to reduce CV risk. ... Being a ‘weekend warrior’ and meeting guideline levels of [moderate to vigorous physical activity] of 150 minutes/week will not completely abolish the deleterious effects of extended sedentary time of > 10.6 hours per day,” Charles B. Eaton, MD, MS, of the Warren Alpert Medical School of Brown University in Providence, Rhode Island, wrote in an editorial accompanying the journal article.
SOURCE:
The study was led by Ezimamaka Ajufo, MD, of Brigham and Women’s Hospital in Boston. It was published online on November 15, 2024, in the Journal of the American College of Cardiology.
LIMITATIONS:
Wrist-based accelerometers cannot assess specific contexts for sedentary behavior and may misclassify standing time as sedentary time, and these limitations may have affected the findings. Physical activity was measured for 1 week only, which might not have fully represented habitual activity patterns. The sample included predominantly White participants and was enriched for health and socioeconomic status, which may have limited the generalizability of the findings.
DISCLOSURES:
The authors disclosed receiving research support, grants, and research fellowships and collaborations from various institutions and pharmaceutical companies, as well as serving on their advisory boards.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Sedentary time exceeding 10.6 h/d is linked to an increased risk for atrial fibrillation, heart failure, myocardial infarction, and cardiovascular (CV) mortality, researchers found. The risk persists even in individuals who meet recommended physical activity levels.
METHODOLOGY:
- Researchers used a validated machine learning approach to investigate the relationships between sedentary behavior and the future risks for CV illness and mortality in 89,530 middle-aged and older adults (mean age, 62 years; 56% women) from the UK Biobank.
- Participants provided data from a wrist-worn triaxial accelerometer that recorded their movements over a period of 7 days.
- Machine learning algorithms classified accelerometer signals into four classes of activities: Sleep, sedentary behavior, light physical activity, and moderate to vigorous physical activity.
- Participants were followed up for a median of 8 years through linkage to national health-related datasets in England, Scotland, and Wales.
- The median sedentary time was 9.4 h/d.
TAKEAWAY:
- During the follow-up period, 3638 individuals (4.9%) experienced incident atrial fibrillation, 1854 (2.09%) developed incident heart failure, 1610 (1.84%) experienced incident myocardial infarction, and 846 (0.94%) died from cardiovascular causes.
- The risks for atrial fibrillation and myocardial infarction increased steadily with an increase in sedentary time, with sedentary time greater than 10.6 h/d showing a modest increase in risk for atrial fibrillation (hazard ratio [HR], 1.11; 95% CI, 1.01-1.21).
- The risks for heart failure and CV mortality were low until sedentary time surpassed approximately 10.6 h/d, after which they rose by 45% (HR, 1.45; 95% CI, 1.28-1.65) and 62% (HR, 1.62; 95% CI, 1.34-1.96), respectively.
- The associations were attenuated but remained significant for CV mortality (HR, 1.33; 95% CI: 1.07-1.64) in individuals who met the recommended levels for physical activity yet were sedentary for more than 10.6 h/d. Reallocating 30 minutes of sedentary time to other activities reduced the risk for heart failure (HR, 0.93; 95% CI, 0.90-0.96) among those who were sedentary more than 10.6 h/d.
IN PRACTICE:
The study “highlights a complex interplay between sedentary behavior and physical activity, ultimately suggesting that sedentary behavior remains relevant for CV disease risk even among individuals meeting sufficient” levels of activity, the researchers reported.
“Individuals should move more and be less sedentary to reduce CV risk. ... Being a ‘weekend warrior’ and meeting guideline levels of [moderate to vigorous physical activity] of 150 minutes/week will not completely abolish the deleterious effects of extended sedentary time of > 10.6 hours per day,” Charles B. Eaton, MD, MS, of the Warren Alpert Medical School of Brown University in Providence, Rhode Island, wrote in an editorial accompanying the journal article.
SOURCE:
The study was led by Ezimamaka Ajufo, MD, of Brigham and Women’s Hospital in Boston. It was published online on November 15, 2024, in the Journal of the American College of Cardiology.
LIMITATIONS:
Wrist-based accelerometers cannot assess specific contexts for sedentary behavior and may misclassify standing time as sedentary time, and these limitations may have affected the findings. Physical activity was measured for 1 week only, which might not have fully represented habitual activity patterns. The sample included predominantly White participants and was enriched for health and socioeconomic status, which may have limited the generalizability of the findings.
DISCLOSURES:
The authors disclosed receiving research support, grants, and research fellowships and collaborations from various institutions and pharmaceutical companies, as well as serving on their advisory boards.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Sedentary time exceeding 10.6 h/d is linked to an increased risk for atrial fibrillation, heart failure, myocardial infarction, and cardiovascular (CV) mortality, researchers found. The risk persists even in individuals who meet recommended physical activity levels.
METHODOLOGY:
- Researchers used a validated machine learning approach to investigate the relationships between sedentary behavior and the future risks for CV illness and mortality in 89,530 middle-aged and older adults (mean age, 62 years; 56% women) from the UK Biobank.
- Participants provided data from a wrist-worn triaxial accelerometer that recorded their movements over a period of 7 days.
- Machine learning algorithms classified accelerometer signals into four classes of activities: Sleep, sedentary behavior, light physical activity, and moderate to vigorous physical activity.
- Participants were followed up for a median of 8 years through linkage to national health-related datasets in England, Scotland, and Wales.
- The median sedentary time was 9.4 h/d.
TAKEAWAY:
- During the follow-up period, 3638 individuals (4.9%) experienced incident atrial fibrillation, 1854 (2.09%) developed incident heart failure, 1610 (1.84%) experienced incident myocardial infarction, and 846 (0.94%) died from cardiovascular causes.
- The risks for atrial fibrillation and myocardial infarction increased steadily with an increase in sedentary time, with sedentary time greater than 10.6 h/d showing a modest increase in risk for atrial fibrillation (hazard ratio [HR], 1.11; 95% CI, 1.01-1.21).
- The risks for heart failure and CV mortality were low until sedentary time surpassed approximately 10.6 h/d, after which they rose by 45% (HR, 1.45; 95% CI, 1.28-1.65) and 62% (HR, 1.62; 95% CI, 1.34-1.96), respectively.
- The associations were attenuated but remained significant for CV mortality (HR, 1.33; 95% CI: 1.07-1.64) in individuals who met the recommended levels for physical activity yet were sedentary for more than 10.6 h/d. Reallocating 30 minutes of sedentary time to other activities reduced the risk for heart failure (HR, 0.93; 95% CI, 0.90-0.96) among those who were sedentary more than 10.6 h/d.
IN PRACTICE:
The study “highlights a complex interplay between sedentary behavior and physical activity, ultimately suggesting that sedentary behavior remains relevant for CV disease risk even among individuals meeting sufficient” levels of activity, the researchers reported.
“Individuals should move more and be less sedentary to reduce CV risk. ... Being a ‘weekend warrior’ and meeting guideline levels of [moderate to vigorous physical activity] of 150 minutes/week will not completely abolish the deleterious effects of extended sedentary time of > 10.6 hours per day,” Charles B. Eaton, MD, MS, of the Warren Alpert Medical School of Brown University in Providence, Rhode Island, wrote in an editorial accompanying the journal article.
SOURCE:
The study was led by Ezimamaka Ajufo, MD, of Brigham and Women’s Hospital in Boston. It was published online on November 15, 2024, in the Journal of the American College of Cardiology.
LIMITATIONS:
Wrist-based accelerometers cannot assess specific contexts for sedentary behavior and may misclassify standing time as sedentary time, and these limitations may have affected the findings. Physical activity was measured for 1 week only, which might not have fully represented habitual activity patterns. The sample included predominantly White participants and was enriched for health and socioeconomic status, which may have limited the generalizability of the findings.
DISCLOSURES:
The authors disclosed receiving research support, grants, and research fellowships and collaborations from various institutions and pharmaceutical companies, as well as serving on their advisory boards.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
In IBD Patients, No Increased Risk for MACE Seen for JAK Inhibitors vs Anti-TNF
PHILADELPHIA — according to a study presented at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
In particular, 1.76% of patients taking JAKi and 1.94% of patients taking anti-TNF developed MACE. There also weren’t significant differences when comparing ulcerative colitis with Crohn’s disease, upadacitinib with tofacitinib, or JAKi with infliximab.
“IBD is associated with an increased risk of cardiovascular diseases, and with the emergence of JAK inhibitors and anti-TNF therapies, there is a concern about the increased risk of MACE,” said lead author Saqr Alsakarneh, MD, an internal medicine resident at the University of Missouri–Kansas City School of Medicine.
Previous randomized controlled trials have indicated increased risks of MACE with JAKi and anti-TNF agents, compared with placebo, but researchers haven’t conducted a head-to-head comparison, he said.
“A potential explanation for previous associations could be linked to immune modulation and inflammation that can increase coagulation risk, as well as fluctuation in disease severity while patients are on the medications, which can impact cardiovascular risk factors,” he added.
Alsakarneh and colleagues conducted a retrospective cohort study using the TriNetX database to identify adult patients with IBD who were treated with JAKi or anti-TNF therapy after diagnosis. After matching patients in the JAKi cohort with patients in the anti-TNF cohort, the research team looked for MACE and VTE within a year of medication initiation, as well as associations by age, sex, and IBD type.
Overall, 3740 patients in the JAKi cohort had a mean age of 43.1 and were 48.9% women and 75.3% White individuals, while 3,740 patients in the anti-TNF cohort had a mean age of 43 and were 48.9% women and 75.3% White individuals.
After excluding those with a history of a prior cardiovascular event, 57 patients (1.76%) in the JAKi cohort developed MACE, compared with 63 patients (1.94%) in the anti-TNF cohort. There weren’t significant differences between the groups in MACE (adjusted hazard ratio [aHR], 0.99) or VTE (aHR, 0.9).
Among patients aged ≥ 65, 25 patients (5.3%) in the JAKi cohort developed MACE, as compared with 30 patients (6.4%) in the anti-TNF cohort. There weren’t significant differences between the groups in MACE (aHR, 0.83) or VTE (aHR, 0.77).
In addition, there were no differences when comparing Crohn’s disease with ulcerative colitis for MACE (aHR, 1.69) or VTE (aHR, 0.85); upadacitinib with tofacitinib for MACE (aHR, 1.1) or VTE (aHR, 1.13); or JAKi medications with infliximab for MACE (aHR, 0.85) or VTE (aHR, 0.8).
Patients in the JAKi group were more likely to undergo intestinal resection surgery (aHR, 1.32), but there wasn’t a statistically significant difference in systematic corticosteroid use (aHR, 0.99).
The study limitations included the inability to assess for disease severity, dose-dependent risk for MACE or VTE, or long-term outcomes among the two cohorts, Alsakarneh said. Prospective controlled trials are needed to confirm findings.
“This is a wonderful study and nice to see. We presented the same thing at Digestive Disease Week that’s being confirmed in this data,” said Miguel Regueiro, MD, AGAF, chief of Cleveland Clinic’s Digestive Disease Institute in Ohio. Regueiro, who wasn’t involved with the study, attended the conference session.
“Looking ahead, all of us are wondering if the regulatory guidance by the FDA [Food and Drug Administration] is going to change the label so we don’t need to step through a TNF,” he said. “I think we’re seeing study after study showing safety or at least not an increased risk with JAK.”
The study was awarded an ACG Noteworthy Abstract. Alsakarneh and Regueiro reported no relevant disclosures.
A version of this article appeared on Medscape.com.
PHILADELPHIA — according to a study presented at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
In particular, 1.76% of patients taking JAKi and 1.94% of patients taking anti-TNF developed MACE. There also weren’t significant differences when comparing ulcerative colitis with Crohn’s disease, upadacitinib with tofacitinib, or JAKi with infliximab.
“IBD is associated with an increased risk of cardiovascular diseases, and with the emergence of JAK inhibitors and anti-TNF therapies, there is a concern about the increased risk of MACE,” said lead author Saqr Alsakarneh, MD, an internal medicine resident at the University of Missouri–Kansas City School of Medicine.
Previous randomized controlled trials have indicated increased risks of MACE with JAKi and anti-TNF agents, compared with placebo, but researchers haven’t conducted a head-to-head comparison, he said.
“A potential explanation for previous associations could be linked to immune modulation and inflammation that can increase coagulation risk, as well as fluctuation in disease severity while patients are on the medications, which can impact cardiovascular risk factors,” he added.
Alsakarneh and colleagues conducted a retrospective cohort study using the TriNetX database to identify adult patients with IBD who were treated with JAKi or anti-TNF therapy after diagnosis. After matching patients in the JAKi cohort with patients in the anti-TNF cohort, the research team looked for MACE and VTE within a year of medication initiation, as well as associations by age, sex, and IBD type.
Overall, 3740 patients in the JAKi cohort had a mean age of 43.1 and were 48.9% women and 75.3% White individuals, while 3,740 patients in the anti-TNF cohort had a mean age of 43 and were 48.9% women and 75.3% White individuals.
After excluding those with a history of a prior cardiovascular event, 57 patients (1.76%) in the JAKi cohort developed MACE, compared with 63 patients (1.94%) in the anti-TNF cohort. There weren’t significant differences between the groups in MACE (adjusted hazard ratio [aHR], 0.99) or VTE (aHR, 0.9).
Among patients aged ≥ 65, 25 patients (5.3%) in the JAKi cohort developed MACE, as compared with 30 patients (6.4%) in the anti-TNF cohort. There weren’t significant differences between the groups in MACE (aHR, 0.83) or VTE (aHR, 0.77).
In addition, there were no differences when comparing Crohn’s disease with ulcerative colitis for MACE (aHR, 1.69) or VTE (aHR, 0.85); upadacitinib with tofacitinib for MACE (aHR, 1.1) or VTE (aHR, 1.13); or JAKi medications with infliximab for MACE (aHR, 0.85) or VTE (aHR, 0.8).
Patients in the JAKi group were more likely to undergo intestinal resection surgery (aHR, 1.32), but there wasn’t a statistically significant difference in systematic corticosteroid use (aHR, 0.99).
The study limitations included the inability to assess for disease severity, dose-dependent risk for MACE or VTE, or long-term outcomes among the two cohorts, Alsakarneh said. Prospective controlled trials are needed to confirm findings.
“This is a wonderful study and nice to see. We presented the same thing at Digestive Disease Week that’s being confirmed in this data,” said Miguel Regueiro, MD, AGAF, chief of Cleveland Clinic’s Digestive Disease Institute in Ohio. Regueiro, who wasn’t involved with the study, attended the conference session.
“Looking ahead, all of us are wondering if the regulatory guidance by the FDA [Food and Drug Administration] is going to change the label so we don’t need to step through a TNF,” he said. “I think we’re seeing study after study showing safety or at least not an increased risk with JAK.”
The study was awarded an ACG Noteworthy Abstract. Alsakarneh and Regueiro reported no relevant disclosures.
A version of this article appeared on Medscape.com.
PHILADELPHIA — according to a study presented at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
In particular, 1.76% of patients taking JAKi and 1.94% of patients taking anti-TNF developed MACE. There also weren’t significant differences when comparing ulcerative colitis with Crohn’s disease, upadacitinib with tofacitinib, or JAKi with infliximab.
“IBD is associated with an increased risk of cardiovascular diseases, and with the emergence of JAK inhibitors and anti-TNF therapies, there is a concern about the increased risk of MACE,” said lead author Saqr Alsakarneh, MD, an internal medicine resident at the University of Missouri–Kansas City School of Medicine.
Previous randomized controlled trials have indicated increased risks of MACE with JAKi and anti-TNF agents, compared with placebo, but researchers haven’t conducted a head-to-head comparison, he said.
“A potential explanation for previous associations could be linked to immune modulation and inflammation that can increase coagulation risk, as well as fluctuation in disease severity while patients are on the medications, which can impact cardiovascular risk factors,” he added.
Alsakarneh and colleagues conducted a retrospective cohort study using the TriNetX database to identify adult patients with IBD who were treated with JAKi or anti-TNF therapy after diagnosis. After matching patients in the JAKi cohort with patients in the anti-TNF cohort, the research team looked for MACE and VTE within a year of medication initiation, as well as associations by age, sex, and IBD type.
Overall, 3740 patients in the JAKi cohort had a mean age of 43.1 and were 48.9% women and 75.3% White individuals, while 3,740 patients in the anti-TNF cohort had a mean age of 43 and were 48.9% women and 75.3% White individuals.
After excluding those with a history of a prior cardiovascular event, 57 patients (1.76%) in the JAKi cohort developed MACE, compared with 63 patients (1.94%) in the anti-TNF cohort. There weren’t significant differences between the groups in MACE (adjusted hazard ratio [aHR], 0.99) or VTE (aHR, 0.9).
Among patients aged ≥ 65, 25 patients (5.3%) in the JAKi cohort developed MACE, as compared with 30 patients (6.4%) in the anti-TNF cohort. There weren’t significant differences between the groups in MACE (aHR, 0.83) or VTE (aHR, 0.77).
In addition, there were no differences when comparing Crohn’s disease with ulcerative colitis for MACE (aHR, 1.69) or VTE (aHR, 0.85); upadacitinib with tofacitinib for MACE (aHR, 1.1) or VTE (aHR, 1.13); or JAKi medications with infliximab for MACE (aHR, 0.85) or VTE (aHR, 0.8).
Patients in the JAKi group were more likely to undergo intestinal resection surgery (aHR, 1.32), but there wasn’t a statistically significant difference in systematic corticosteroid use (aHR, 0.99).
The study limitations included the inability to assess for disease severity, dose-dependent risk for MACE or VTE, or long-term outcomes among the two cohorts, Alsakarneh said. Prospective controlled trials are needed to confirm findings.
“This is a wonderful study and nice to see. We presented the same thing at Digestive Disease Week that’s being confirmed in this data,” said Miguel Regueiro, MD, AGAF, chief of Cleveland Clinic’s Digestive Disease Institute in Ohio. Regueiro, who wasn’t involved with the study, attended the conference session.
“Looking ahead, all of us are wondering if the regulatory guidance by the FDA [Food and Drug Administration] is going to change the label so we don’t need to step through a TNF,” he said. “I think we’re seeing study after study showing safety or at least not an increased risk with JAK.”
The study was awarded an ACG Noteworthy Abstract. Alsakarneh and Regueiro reported no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM ACG 2024
New Pill Successfully Lowers Lp(a) Levels
Concentrations of Lp(a) cholesterol are genetically determined and remain steady throughout life. Levels of 125 nmol/L or higher promote clotting and inflammation, significantly increasing the risk for heart attack, stroke, aortic stenosis, and peripheral artery disease. This affects about 20% of the population, particularly people of Black African and South Asian descent.
There are currently no approved therapies that lower Lp(a), said study author Stephen Nicholls, MBBS, PhD, director of the Victorian Heart Institute at Monash University in Melbourne, Australia. Several injectable therapies are currently in clinical trials, but muvalaplin is the only oral option. The new drug lowers Lp(a) levels by disrupting the bond between the two parts of the Lp(a) particle.
The KRAKEN Trial
In the KRAKEN trial, 233 adults from around the world with very high Lp(a) levels (> 175 nmol/L) were randomized either to one of three daily doses of muvalaplin — 10, 60, or 240 mg — or to placebo for 12 weeks.
The researchers measured Lp(a) levels with a standard blood test and with a novel test designed to specifically measure levels of intact Lp(a) particles in the blood. In addition to Lp(a), the standard test detects one of its components, apolipoprotein A particles, that are bound to the drug, which can lead to an underestimation of Lp(a) reductions.
Lp(a) levels were up to 70.0% lower in the muvalaplin group than in the placebo group when measured with the traditional blood test and by up to 85.5% lower when measured with the new test. Approximately 82% of participants achieved an Lp(a) level lower than 125 nmol/L when measured with the traditional blood test, and 97% achieved that level when the new test was used. Patients who received either 60 or 240 mg of muvalaplin had similar reductions in Lp(a) levels, which were greater than the reductions seen in the 10 mg group. The drug was safe and generally well tolerated.
“This is a very reassuring phase 2 result,” Nicholls said when he presented the KRAKEN findings at the American Heart Association (AHA) Scientific Sessions 2024 in Chicago, which were simultaneously published online in JAMA. “It encourages the ongoing development of this agent.”
Lp(a) levels are not affected by changes in lifestyle or diet or by traditional lipid-lowering treatments like statins, said Erin Michos, MD, a cardiologist at the Johns Hopkins University School of Medicine in Baltimore, Maryland, who was not involved in the study.
And high Lp(a) levels confer significant cardiovascular risk even when other risks are reduced. So muvalaplin is “a highly promising approach to treat a previously untreatable disorder,” she explained.
Larger and longer studies, with more diverse patient populations, are needed to confirm the results and to determine whether reducing Lp(a) also improves cardiovascular outcomes, Michos pointed out.
“While muvalaplin appears to be an effective approach to lowering Lp(a) levels, we still need to study whether Lp(a) lowering will result in fewer heart attacks and strokes,” Nicholls added.
A version of this article appeared on Medscape.com.
Concentrations of Lp(a) cholesterol are genetically determined and remain steady throughout life. Levels of 125 nmol/L or higher promote clotting and inflammation, significantly increasing the risk for heart attack, stroke, aortic stenosis, and peripheral artery disease. This affects about 20% of the population, particularly people of Black African and South Asian descent.
There are currently no approved therapies that lower Lp(a), said study author Stephen Nicholls, MBBS, PhD, director of the Victorian Heart Institute at Monash University in Melbourne, Australia. Several injectable therapies are currently in clinical trials, but muvalaplin is the only oral option. The new drug lowers Lp(a) levels by disrupting the bond between the two parts of the Lp(a) particle.
The KRAKEN Trial
In the KRAKEN trial, 233 adults from around the world with very high Lp(a) levels (> 175 nmol/L) were randomized either to one of three daily doses of muvalaplin — 10, 60, or 240 mg — or to placebo for 12 weeks.
The researchers measured Lp(a) levels with a standard blood test and with a novel test designed to specifically measure levels of intact Lp(a) particles in the blood. In addition to Lp(a), the standard test detects one of its components, apolipoprotein A particles, that are bound to the drug, which can lead to an underestimation of Lp(a) reductions.
Lp(a) levels were up to 70.0% lower in the muvalaplin group than in the placebo group when measured with the traditional blood test and by up to 85.5% lower when measured with the new test. Approximately 82% of participants achieved an Lp(a) level lower than 125 nmol/L when measured with the traditional blood test, and 97% achieved that level when the new test was used. Patients who received either 60 or 240 mg of muvalaplin had similar reductions in Lp(a) levels, which were greater than the reductions seen in the 10 mg group. The drug was safe and generally well tolerated.
“This is a very reassuring phase 2 result,” Nicholls said when he presented the KRAKEN findings at the American Heart Association (AHA) Scientific Sessions 2024 in Chicago, which were simultaneously published online in JAMA. “It encourages the ongoing development of this agent.”
Lp(a) levels are not affected by changes in lifestyle or diet or by traditional lipid-lowering treatments like statins, said Erin Michos, MD, a cardiologist at the Johns Hopkins University School of Medicine in Baltimore, Maryland, who was not involved in the study.
And high Lp(a) levels confer significant cardiovascular risk even when other risks are reduced. So muvalaplin is “a highly promising approach to treat a previously untreatable disorder,” she explained.
Larger and longer studies, with more diverse patient populations, are needed to confirm the results and to determine whether reducing Lp(a) also improves cardiovascular outcomes, Michos pointed out.
“While muvalaplin appears to be an effective approach to lowering Lp(a) levels, we still need to study whether Lp(a) lowering will result in fewer heart attacks and strokes,” Nicholls added.
A version of this article appeared on Medscape.com.
Concentrations of Lp(a) cholesterol are genetically determined and remain steady throughout life. Levels of 125 nmol/L or higher promote clotting and inflammation, significantly increasing the risk for heart attack, stroke, aortic stenosis, and peripheral artery disease. This affects about 20% of the population, particularly people of Black African and South Asian descent.
There are currently no approved therapies that lower Lp(a), said study author Stephen Nicholls, MBBS, PhD, director of the Victorian Heart Institute at Monash University in Melbourne, Australia. Several injectable therapies are currently in clinical trials, but muvalaplin is the only oral option. The new drug lowers Lp(a) levels by disrupting the bond between the two parts of the Lp(a) particle.
The KRAKEN Trial
In the KRAKEN trial, 233 adults from around the world with very high Lp(a) levels (> 175 nmol/L) were randomized either to one of three daily doses of muvalaplin — 10, 60, or 240 mg — or to placebo for 12 weeks.
The researchers measured Lp(a) levels with a standard blood test and with a novel test designed to specifically measure levels of intact Lp(a) particles in the blood. In addition to Lp(a), the standard test detects one of its components, apolipoprotein A particles, that are bound to the drug, which can lead to an underestimation of Lp(a) reductions.
Lp(a) levels were up to 70.0% lower in the muvalaplin group than in the placebo group when measured with the traditional blood test and by up to 85.5% lower when measured with the new test. Approximately 82% of participants achieved an Lp(a) level lower than 125 nmol/L when measured with the traditional blood test, and 97% achieved that level when the new test was used. Patients who received either 60 or 240 mg of muvalaplin had similar reductions in Lp(a) levels, which were greater than the reductions seen in the 10 mg group. The drug was safe and generally well tolerated.
“This is a very reassuring phase 2 result,” Nicholls said when he presented the KRAKEN findings at the American Heart Association (AHA) Scientific Sessions 2024 in Chicago, which were simultaneously published online in JAMA. “It encourages the ongoing development of this agent.”
Lp(a) levels are not affected by changes in lifestyle or diet or by traditional lipid-lowering treatments like statins, said Erin Michos, MD, a cardiologist at the Johns Hopkins University School of Medicine in Baltimore, Maryland, who was not involved in the study.
And high Lp(a) levels confer significant cardiovascular risk even when other risks are reduced. So muvalaplin is “a highly promising approach to treat a previously untreatable disorder,” she explained.
Larger and longer studies, with more diverse patient populations, are needed to confirm the results and to determine whether reducing Lp(a) also improves cardiovascular outcomes, Michos pointed out.
“While muvalaplin appears to be an effective approach to lowering Lp(a) levels, we still need to study whether Lp(a) lowering will result in fewer heart attacks and strokes,” Nicholls added.
A version of this article appeared on Medscape.com.
Gout and SGLT2 Inhibitors: Evidence Points to Reduced Need for ULT, Flare Drugs
WASHINGTON — Use of sodium-glucose cotransporter 2 inhibitors (SGLT2i) reduced the need for urate-lowering therapy (ULT) and gout flare therapies in people who had both type 2 diabetes (T2D) and gout, new research has found.
Data from a large US claims database showed that SGLT2i use was associated with a 31% lower rate of initiation of ULT. “This provides further support for the use of SLGT2i therapy in patients with gout, particularly those with high-risk multimorbidity and polypharmacy,” Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said in his presentation of the data at the annual meeting of the American College of Rheumatology.
The first agent of the SGLT2i class, dapagliflozin, was initially approved in the United States a decade ago for treating T2D. Since then, several other “flozins” have become available, and some have also received additional indications for heart failure and albuminuric chronic kidney disease. Several prior studies have linked SGLT2i use with lower rates of gout flares as well as lower likelihood of developing gout in the first place, although not all studies have found this benefit.
Asked about the clinical implications of the new data, Challener said in an interview that “I don’t think we’re quite at the point where this is changing gout management per se, but this just helps us understand that [SGT2is] may have a role at some point, maybe as a combination on top of another agent. Or, in some patients, it really may be enough if they’re already on an SGLT2i where we don’t need to jump to adding allopurinol. Maybe they have tophi, but they were just started on an SGLT2i and they’re not flaring. Typically, you would start those patients on allopurinol, but you could potentially just monitor them if they were just started on one of those [SGLT2i] agents.”
Asked to comment, session moderator J. Antonio Aviña-Zubieta, MD, PhD, head of the Division of Rheumatology at the University of British Columbia, Vancouver, Canada, and senior scientist at Arthritis Research Canada, said in an interview: “What I can see possibly happening when there’s more evidence is that SGLT2is may be used or even become standard of care as an adjuvant therapy to decrease flares, and by that, decrease the risk of complications.”
Reductions in ULT, Flares, and Healthcare Visits
The new study used administrative health data from the multicenter TriNetX Diamond network of electronic medical record and claims data from 92 healthcare sites with 212 million patients. Among those with both T2D and gout who were not taking ULT at baseline, a total of 16,104 initiated SGLT2is and 16,046 initiated glucagon-like peptide 1 receptor agonists (GLP-1 RA).
Propensity score matching was conducted for demographics including age, race, and sex; comorbidities; use of emergency, inpatient, and critical care services; medications; labs; and body mass index. That yielded 11,800 individuals each in the SGLT2i and GLP-1 RA groups.
Over 5 years, 9.9% of the SGLT2i group vs 13.4% of those using GLP-1 RA had initiated ULT, a significant difference with a hazard ratio (HR) of 0.69 (95% CI, 0.64-0.75). The risk for initiation of colchicine for gout flares was 4.7% with SGLT2i vs 6.0% for GLP-1 RA — also a significant difference with an HR of 0.74 (0.65-0.83).
Medical visits for gout occurred in 28.0% vs 28.4% of patients, which also reached statistical significance (HR, 0.94; 95% CI, 0.89-0.99).
Aviña-Zubieta, an author of one of the previous studies finding a reduction in gout flares with SGLT2i, said, “many patients do not want to start gout therapy until they start having more acute attacks. ... So, for a lot of people, it’s a burden taking another pill to prevent one attack. But, if you don’t treat it over time, the attacks come more often. So, can we still delay the initiation of therapy? If you’re not having that many flares, you’re decreasing the burden of the disease and polypharmacy, which I think is the potential benefit in the long run if you already have an indication for the therapy for diabetes. ... These data are supporting that.”
Indeed, Challener said these data can help in counseling patients. “Taking your SGLT2i for your heart failure and your diabetes is also providing some benefit for your gout, and we know that there is also cardiac benefit when gout is controlled.”
Challener and Aviña-Zubieta had no disclosures.
A version of this article first appeared on Medscape.com.
WASHINGTON — Use of sodium-glucose cotransporter 2 inhibitors (SGLT2i) reduced the need for urate-lowering therapy (ULT) and gout flare therapies in people who had both type 2 diabetes (T2D) and gout, new research has found.
Data from a large US claims database showed that SGLT2i use was associated with a 31% lower rate of initiation of ULT. “This provides further support for the use of SLGT2i therapy in patients with gout, particularly those with high-risk multimorbidity and polypharmacy,” Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said in his presentation of the data at the annual meeting of the American College of Rheumatology.
The first agent of the SGLT2i class, dapagliflozin, was initially approved in the United States a decade ago for treating T2D. Since then, several other “flozins” have become available, and some have also received additional indications for heart failure and albuminuric chronic kidney disease. Several prior studies have linked SGLT2i use with lower rates of gout flares as well as lower likelihood of developing gout in the first place, although not all studies have found this benefit.
Asked about the clinical implications of the new data, Challener said in an interview that “I don’t think we’re quite at the point where this is changing gout management per se, but this just helps us understand that [SGT2is] may have a role at some point, maybe as a combination on top of another agent. Or, in some patients, it really may be enough if they’re already on an SGLT2i where we don’t need to jump to adding allopurinol. Maybe they have tophi, but they were just started on an SGLT2i and they’re not flaring. Typically, you would start those patients on allopurinol, but you could potentially just monitor them if they were just started on one of those [SGLT2i] agents.”
Asked to comment, session moderator J. Antonio Aviña-Zubieta, MD, PhD, head of the Division of Rheumatology at the University of British Columbia, Vancouver, Canada, and senior scientist at Arthritis Research Canada, said in an interview: “What I can see possibly happening when there’s more evidence is that SGLT2is may be used or even become standard of care as an adjuvant therapy to decrease flares, and by that, decrease the risk of complications.”
Reductions in ULT, Flares, and Healthcare Visits
The new study used administrative health data from the multicenter TriNetX Diamond network of electronic medical record and claims data from 92 healthcare sites with 212 million patients. Among those with both T2D and gout who were not taking ULT at baseline, a total of 16,104 initiated SGLT2is and 16,046 initiated glucagon-like peptide 1 receptor agonists (GLP-1 RA).
Propensity score matching was conducted for demographics including age, race, and sex; comorbidities; use of emergency, inpatient, and critical care services; medications; labs; and body mass index. That yielded 11,800 individuals each in the SGLT2i and GLP-1 RA groups.
Over 5 years, 9.9% of the SGLT2i group vs 13.4% of those using GLP-1 RA had initiated ULT, a significant difference with a hazard ratio (HR) of 0.69 (95% CI, 0.64-0.75). The risk for initiation of colchicine for gout flares was 4.7% with SGLT2i vs 6.0% for GLP-1 RA — also a significant difference with an HR of 0.74 (0.65-0.83).
Medical visits for gout occurred in 28.0% vs 28.4% of patients, which also reached statistical significance (HR, 0.94; 95% CI, 0.89-0.99).
Aviña-Zubieta, an author of one of the previous studies finding a reduction in gout flares with SGLT2i, said, “many patients do not want to start gout therapy until they start having more acute attacks. ... So, for a lot of people, it’s a burden taking another pill to prevent one attack. But, if you don’t treat it over time, the attacks come more often. So, can we still delay the initiation of therapy? If you’re not having that many flares, you’re decreasing the burden of the disease and polypharmacy, which I think is the potential benefit in the long run if you already have an indication for the therapy for diabetes. ... These data are supporting that.”
Indeed, Challener said these data can help in counseling patients. “Taking your SGLT2i for your heart failure and your diabetes is also providing some benefit for your gout, and we know that there is also cardiac benefit when gout is controlled.”
Challener and Aviña-Zubieta had no disclosures.
A version of this article first appeared on Medscape.com.
WASHINGTON — Use of sodium-glucose cotransporter 2 inhibitors (SGLT2i) reduced the need for urate-lowering therapy (ULT) and gout flare therapies in people who had both type 2 diabetes (T2D) and gout, new research has found.
Data from a large US claims database showed that SGLT2i use was associated with a 31% lower rate of initiation of ULT. “This provides further support for the use of SLGT2i therapy in patients with gout, particularly those with high-risk multimorbidity and polypharmacy,” Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said in his presentation of the data at the annual meeting of the American College of Rheumatology.
The first agent of the SGLT2i class, dapagliflozin, was initially approved in the United States a decade ago for treating T2D. Since then, several other “flozins” have become available, and some have also received additional indications for heart failure and albuminuric chronic kidney disease. Several prior studies have linked SGLT2i use with lower rates of gout flares as well as lower likelihood of developing gout in the first place, although not all studies have found this benefit.
Asked about the clinical implications of the new data, Challener said in an interview that “I don’t think we’re quite at the point where this is changing gout management per se, but this just helps us understand that [SGT2is] may have a role at some point, maybe as a combination on top of another agent. Or, in some patients, it really may be enough if they’re already on an SGLT2i where we don’t need to jump to adding allopurinol. Maybe they have tophi, but they were just started on an SGLT2i and they’re not flaring. Typically, you would start those patients on allopurinol, but you could potentially just monitor them if they were just started on one of those [SGLT2i] agents.”
Asked to comment, session moderator J. Antonio Aviña-Zubieta, MD, PhD, head of the Division of Rheumatology at the University of British Columbia, Vancouver, Canada, and senior scientist at Arthritis Research Canada, said in an interview: “What I can see possibly happening when there’s more evidence is that SGLT2is may be used or even become standard of care as an adjuvant therapy to decrease flares, and by that, decrease the risk of complications.”
Reductions in ULT, Flares, and Healthcare Visits
The new study used administrative health data from the multicenter TriNetX Diamond network of electronic medical record and claims data from 92 healthcare sites with 212 million patients. Among those with both T2D and gout who were not taking ULT at baseline, a total of 16,104 initiated SGLT2is and 16,046 initiated glucagon-like peptide 1 receptor agonists (GLP-1 RA).
Propensity score matching was conducted for demographics including age, race, and sex; comorbidities; use of emergency, inpatient, and critical care services; medications; labs; and body mass index. That yielded 11,800 individuals each in the SGLT2i and GLP-1 RA groups.
Over 5 years, 9.9% of the SGLT2i group vs 13.4% of those using GLP-1 RA had initiated ULT, a significant difference with a hazard ratio (HR) of 0.69 (95% CI, 0.64-0.75). The risk for initiation of colchicine for gout flares was 4.7% with SGLT2i vs 6.0% for GLP-1 RA — also a significant difference with an HR of 0.74 (0.65-0.83).
Medical visits for gout occurred in 28.0% vs 28.4% of patients, which also reached statistical significance (HR, 0.94; 95% CI, 0.89-0.99).
Aviña-Zubieta, an author of one of the previous studies finding a reduction in gout flares with SGLT2i, said, “many patients do not want to start gout therapy until they start having more acute attacks. ... So, for a lot of people, it’s a burden taking another pill to prevent one attack. But, if you don’t treat it over time, the attacks come more often. So, can we still delay the initiation of therapy? If you’re not having that many flares, you’re decreasing the burden of the disease and polypharmacy, which I think is the potential benefit in the long run if you already have an indication for the therapy for diabetes. ... These data are supporting that.”
Indeed, Challener said these data can help in counseling patients. “Taking your SGLT2i for your heart failure and your diabetes is also providing some benefit for your gout, and we know that there is also cardiac benefit when gout is controlled.”
Challener and Aviña-Zubieta had no disclosures.
A version of this article first appeared on Medscape.com.
FROM ACR 2024
An Epidemiologist’s Guide to Debunking Nutritional Research
You’re invited to a dinner party but you struggle to make small talk. Do not worry; that will invariably crop up over cocktails. Because all journalism has been reduced to listicles, here are four ways to seem clever at dinner parties.
1. The Predinner Cocktails: A Lesson in Reverse Causation
Wine connoisseurs sniff, swirl, and gently swish the wine in their mouths before spitting out and cleansing their palates to better appreciate the subtlety of each vintage. If you’re not an oenophile, no matter. Whenever somebody claims that moderate amounts of alcohol are good for your heart, this is your moment to pounce. Interject yourself in the conversation and tell everybody about reverse causation.
Reverse causation, also known as protopathic bias, involves misinterpreting the directionality of an association. You assume that X leads to Y, when in fact Y leads to X. Temporal paradoxes are useful plot devices in science fiction movies, but they have no place in medical research. In our bland world, cause must precede effect. As such, smoking leads to lung cancer; lung cancer doesn’t make you smoke more.
But with alcohol, directionality is less obvious. Many studies of alcohol and cardiovascular disease have demonstrated a U-shaped association, with risk being lowest among those who drink moderate amounts of alcohol (usually one to two drinks per day) and higher in those who drink more and also those who drink very little.
But one must ask why some people drink little or no alcohol. There is an important difference between former drinkers and never drinkers. Former drinkers cut back for a reason. More likely than not, the reason for this newfound sobriety was medical. A new cancer diagnosis, the emergence of atrial fibrillation, the development of diabetes, or rising blood pressure are all good reasons to reduce or eliminate alcohol. A cross-sectional study will fail to capture that alcohol consumption changes over time — people who now don’t drink may have imbibed alcohol in years past. It was not abstinence that led to an increased risk for heart disease; it was the increased risk for heart disease that led to abstinence.
You see the same phenomenon with the so-called obesity paradox. The idea that being a little overweight is good for you may appeal when you no longer fit into last year’s pants. But people who are underweight are so for a reason. Malnutrition, cachexia from cancer, or some other cause is almost certainly driving up the risk at the left-hand side of the U-shaped curve that makes the middle part seem better than it actually is.
Food consumption changes over time. A cross-sectional survey at one point in time cannot accurately capture past habits and distant exposures, especially for diseases such as heart disease and cancer that develop slowly over time. Studies on alcohol that try to overcome these shortcomings by eliminating former drinkers, or by using Mendelian randomization to better account for past exposure, do not show a cardiovascular benefit for moderate red wine drinking.
2. The Hors D’oeuvres — The Importance of RCTs
Now that you have made yourself the center of attention, it is time to cement your newfound reputation as a font of scientific knowledge. Most self-respecting hosts will serve smoked salmon as an amuse-bouche before the main meal. When someone mentions the health benefits of fish oils, you should take the opportunity to teach them about confounding.
Fish, especially cold-water fish from northern climates, have relatively high amounts of omega-3 fatty acids. Despite the plethora of observational studies suggesting a cardiovascular benefit, it’s now relatively clear that fish oil or omega-3 supplements have no medical benefit.
This will probably come as a shock to the worried well, but many studies, including VITAL and ASCEND, have demonstrated no cardiovascular or cancer benefit to supplementation with omega-3s. The reason is straightforward and explains why hormone replacement therapy, vitamin D, and myriad purported game-changers never panned out. Confounding is hard to overcome in observational research.
Prior to the publication of the Women’s Health Initiative (WHI) Study, hormone replacement therapy was routinely prescribed to postmenopausal women because numerous observational studies suggested a cardiovascular benefit. But with the publication of the WHI study, it became clear that much of that “benefit” was due to confounding. The women choosing to take hormones were more health conscious at baseline and healthier overall.
A similar phenomenon occurred during COVID. Patients with low serum vitamin D levels had worse outcomes, prompting many to suggest vitamin D supplementation as a possible treatment. Trials did not support the intervention because we’d overlooked the obvious. People with vitamin D deficiency have underlying health problems that contribute to the vitamin D deficiency. They are probably older, frailer, possibly with a poorer diet. No amount of statistical adjustment can account for all those differences, and some degree of residual confounding will always persist.
The only way to overcome confounding is with randomization. When patients are randomly assigned to one group or another, their baseline differences largely balance out if the randomization was performed properly and the groups were large enough. There is a role for observational research, such as in situations where ethics, cost, and practicality do not allow for a randomized controlled trial. But randomized controlled trials have largely put to rest the purported health benefits of over-the-counter fish oils, omega-3s, and vitamin D.
3. The Main Course — Absolute vs Relative Risk
When you get to the main course, all eyes will now be on you. You will almost certainly be called upon to pronounce on the harms or benefits of red meat consumption. Begin by regaling your guests with a little trivia. Ask them if they know the definition of red meat and white meat. When someone says pork is white meat, you can reveal that “pork, the other white meat,” was a marketing slogan with no scientific underpinning. Now that everyone is lulled into a stupefied silence, tell them that red meat comes from mammals and white meat comes from birds. As they process this revelation, you can now launch into the deeply mathematical concept of absolute vs relative risk.
Many etiquette books will caution against bringing up math at a dinner party. These books are wrong. Everyone finds math interesting if they are primed properly. For example, you can point to a study claiming that berries reduce cardiovascular risk in women. Even if true — and there is reason to be cautious, given the observational nature of the research — we need to understand what the authors meant by a 32% risk reduction. (Side note: It was a reduction in hazard, with a hazard ratio of 0.68 (95% CI, 0.49-0.96), but we won’t dwell on the difference between hazard ratios and risk ratios right now.)
This relative risk reduction has to be interpreted carefully. The authors divided the population into quintiles based on their consumption of anthocyanins (the antioxidant in blueberries and strawberries) and compared the bottom fifth (average consumption, 2.5 mg/d) with the top fifth (average consumption, 25 mg/d). The bottom quintile had 126 myocardial infarctions (MIs) over 324,793 patient-years compared with 59 MIs over 332,143 patient-years. Some quick math shows an approximate reduction from 39 to 18 MIs per 100,000 patient-years. Or to put it another way, you must get 4762 women to increase their berry consumption 10-fold for 1 year to prevent one heart attack. Feel free to show people how you calculated this number. They will be impressed by your head for numbers. It is nothing more than 39 minus 18, divided by 100,000, to get the absolute risk reduction. Take the reciprocal of this (ie, 1 divided by this number) to get the number needed to treat.
Describing risks in absolute terms or using number needed to treat (or harm) can help conceptualize statistics that are sometimes hard to wrap your head around.
4. Dessert — Funding
By the time the coffee is served, everyone will be hanging on to your every word. This is as it should be, and you should not be afraid of your newfound power and influence.
Dessert will probably involve some form of chocolate, possibly in cake format. (Anyone who serves fruit as dessert is not someone you should associate with.) Take the opportunity to tell your follow diners that chocolate is not actually good for you and will not boost brain performance.
The health benefits of chocolate are often repeated but rarely scrutinized. In fact, much of the scientific research purporting to show that chocolate is good for you did not actually study chocolate. It usually involved a cocoa bean extract because the chocolate manufacturing process destroys the supposedly health-promoting antioxidants in the cocoa bean. It is true that dark chocolate has more antioxidants than milk chocolate, and that the addition of milk to chocolate further inactivates the potentially healthy antioxidants. But the amount of sugar and fat that has to be added to chocolate to make it palatable precludes any serious consideration about health benefits. Dark chocolate may have less fat and sugar than milk chocolate, but it still has a lot.
But even the cocoa bean extract doesn’t seem to do much for your heart or your brain. The long-awaited COSMOS study was published with surprisingly little fanfare. The largest randomized controlled trial of chocolate (or rather cocoa bean extract) was supposed to settle the issue definitively.
COSMOS showed no cardiovascular or neurocognitive benefit to the cocoa bean extract. But the health halo of chocolate continues to be bolstered by many studies funded by chocolate manufacturers.
We are appropriately critical of the pharmaceutical industry’s involvement in drug research. However, we should not forget that any private entity is prone to the same self-interest regardless of its product’s tastiness. How many of you knew that there was an avocado lobby funding research? No matter how many industry-funded observational studies using surrogate endpoints are out there telling you that chocolate is healthy, a randomized trial with hard clinical endpoints such as COSMOS should generally win the day.
The Final Goodbyes — Summarizing Your Case
As the party slowly winds down and everyone is saddened that you will soon take your leave, synthesize everything you have taught them over the evening. Like movies, not all studies are good. Some are just bad. They can be prone to reverse causation or confounding, and they may report relative risks when absolute risks would be more telling. Reading research studies critically is essential for separating the wheat from the chaff. With the knowledge you have now imparted to your friends, they will be much better consumers of medical news, especially when it comes to food.
And they will no doubt thank you for it by never inviting you to another dinner party!
Labos, a cardiologist at Hôpital, Notre-Dame, Montreal, Quebec, Canada, has disclosed no relevant financial relationships. He has a degree in epidemiology.
A version of this article appeared on Medscape.com.
You’re invited to a dinner party but you struggle to make small talk. Do not worry; that will invariably crop up over cocktails. Because all journalism has been reduced to listicles, here are four ways to seem clever at dinner parties.
1. The Predinner Cocktails: A Lesson in Reverse Causation
Wine connoisseurs sniff, swirl, and gently swish the wine in their mouths before spitting out and cleansing their palates to better appreciate the subtlety of each vintage. If you’re not an oenophile, no matter. Whenever somebody claims that moderate amounts of alcohol are good for your heart, this is your moment to pounce. Interject yourself in the conversation and tell everybody about reverse causation.
Reverse causation, also known as protopathic bias, involves misinterpreting the directionality of an association. You assume that X leads to Y, when in fact Y leads to X. Temporal paradoxes are useful plot devices in science fiction movies, but they have no place in medical research. In our bland world, cause must precede effect. As such, smoking leads to lung cancer; lung cancer doesn’t make you smoke more.
But with alcohol, directionality is less obvious. Many studies of alcohol and cardiovascular disease have demonstrated a U-shaped association, with risk being lowest among those who drink moderate amounts of alcohol (usually one to two drinks per day) and higher in those who drink more and also those who drink very little.
But one must ask why some people drink little or no alcohol. There is an important difference between former drinkers and never drinkers. Former drinkers cut back for a reason. More likely than not, the reason for this newfound sobriety was medical. A new cancer diagnosis, the emergence of atrial fibrillation, the development of diabetes, or rising blood pressure are all good reasons to reduce or eliminate alcohol. A cross-sectional study will fail to capture that alcohol consumption changes over time — people who now don’t drink may have imbibed alcohol in years past. It was not abstinence that led to an increased risk for heart disease; it was the increased risk for heart disease that led to abstinence.
You see the same phenomenon with the so-called obesity paradox. The idea that being a little overweight is good for you may appeal when you no longer fit into last year’s pants. But people who are underweight are so for a reason. Malnutrition, cachexia from cancer, or some other cause is almost certainly driving up the risk at the left-hand side of the U-shaped curve that makes the middle part seem better than it actually is.
Food consumption changes over time. A cross-sectional survey at one point in time cannot accurately capture past habits and distant exposures, especially for diseases such as heart disease and cancer that develop slowly over time. Studies on alcohol that try to overcome these shortcomings by eliminating former drinkers, or by using Mendelian randomization to better account for past exposure, do not show a cardiovascular benefit for moderate red wine drinking.
2. The Hors D’oeuvres — The Importance of RCTs
Now that you have made yourself the center of attention, it is time to cement your newfound reputation as a font of scientific knowledge. Most self-respecting hosts will serve smoked salmon as an amuse-bouche before the main meal. When someone mentions the health benefits of fish oils, you should take the opportunity to teach them about confounding.
Fish, especially cold-water fish from northern climates, have relatively high amounts of omega-3 fatty acids. Despite the plethora of observational studies suggesting a cardiovascular benefit, it’s now relatively clear that fish oil or omega-3 supplements have no medical benefit.
This will probably come as a shock to the worried well, but many studies, including VITAL and ASCEND, have demonstrated no cardiovascular or cancer benefit to supplementation with omega-3s. The reason is straightforward and explains why hormone replacement therapy, vitamin D, and myriad purported game-changers never panned out. Confounding is hard to overcome in observational research.
Prior to the publication of the Women’s Health Initiative (WHI) Study, hormone replacement therapy was routinely prescribed to postmenopausal women because numerous observational studies suggested a cardiovascular benefit. But with the publication of the WHI study, it became clear that much of that “benefit” was due to confounding. The women choosing to take hormones were more health conscious at baseline and healthier overall.
A similar phenomenon occurred during COVID. Patients with low serum vitamin D levels had worse outcomes, prompting many to suggest vitamin D supplementation as a possible treatment. Trials did not support the intervention because we’d overlooked the obvious. People with vitamin D deficiency have underlying health problems that contribute to the vitamin D deficiency. They are probably older, frailer, possibly with a poorer diet. No amount of statistical adjustment can account for all those differences, and some degree of residual confounding will always persist.
The only way to overcome confounding is with randomization. When patients are randomly assigned to one group or another, their baseline differences largely balance out if the randomization was performed properly and the groups were large enough. There is a role for observational research, such as in situations where ethics, cost, and practicality do not allow for a randomized controlled trial. But randomized controlled trials have largely put to rest the purported health benefits of over-the-counter fish oils, omega-3s, and vitamin D.
3. The Main Course — Absolute vs Relative Risk
When you get to the main course, all eyes will now be on you. You will almost certainly be called upon to pronounce on the harms or benefits of red meat consumption. Begin by regaling your guests with a little trivia. Ask them if they know the definition of red meat and white meat. When someone says pork is white meat, you can reveal that “pork, the other white meat,” was a marketing slogan with no scientific underpinning. Now that everyone is lulled into a stupefied silence, tell them that red meat comes from mammals and white meat comes from birds. As they process this revelation, you can now launch into the deeply mathematical concept of absolute vs relative risk.
Many etiquette books will caution against bringing up math at a dinner party. These books are wrong. Everyone finds math interesting if they are primed properly. For example, you can point to a study claiming that berries reduce cardiovascular risk in women. Even if true — and there is reason to be cautious, given the observational nature of the research — we need to understand what the authors meant by a 32% risk reduction. (Side note: It was a reduction in hazard, with a hazard ratio of 0.68 (95% CI, 0.49-0.96), but we won’t dwell on the difference between hazard ratios and risk ratios right now.)
This relative risk reduction has to be interpreted carefully. The authors divided the population into quintiles based on their consumption of anthocyanins (the antioxidant in blueberries and strawberries) and compared the bottom fifth (average consumption, 2.5 mg/d) with the top fifth (average consumption, 25 mg/d). The bottom quintile had 126 myocardial infarctions (MIs) over 324,793 patient-years compared with 59 MIs over 332,143 patient-years. Some quick math shows an approximate reduction from 39 to 18 MIs per 100,000 patient-years. Or to put it another way, you must get 4762 women to increase their berry consumption 10-fold for 1 year to prevent one heart attack. Feel free to show people how you calculated this number. They will be impressed by your head for numbers. It is nothing more than 39 minus 18, divided by 100,000, to get the absolute risk reduction. Take the reciprocal of this (ie, 1 divided by this number) to get the number needed to treat.
Describing risks in absolute terms or using number needed to treat (or harm) can help conceptualize statistics that are sometimes hard to wrap your head around.
4. Dessert — Funding
By the time the coffee is served, everyone will be hanging on to your every word. This is as it should be, and you should not be afraid of your newfound power and influence.
Dessert will probably involve some form of chocolate, possibly in cake format. (Anyone who serves fruit as dessert is not someone you should associate with.) Take the opportunity to tell your follow diners that chocolate is not actually good for you and will not boost brain performance.
The health benefits of chocolate are often repeated but rarely scrutinized. In fact, much of the scientific research purporting to show that chocolate is good for you did not actually study chocolate. It usually involved a cocoa bean extract because the chocolate manufacturing process destroys the supposedly health-promoting antioxidants in the cocoa bean. It is true that dark chocolate has more antioxidants than milk chocolate, and that the addition of milk to chocolate further inactivates the potentially healthy antioxidants. But the amount of sugar and fat that has to be added to chocolate to make it palatable precludes any serious consideration about health benefits. Dark chocolate may have less fat and sugar than milk chocolate, but it still has a lot.
But even the cocoa bean extract doesn’t seem to do much for your heart or your brain. The long-awaited COSMOS study was published with surprisingly little fanfare. The largest randomized controlled trial of chocolate (or rather cocoa bean extract) was supposed to settle the issue definitively.
COSMOS showed no cardiovascular or neurocognitive benefit to the cocoa bean extract. But the health halo of chocolate continues to be bolstered by many studies funded by chocolate manufacturers.
We are appropriately critical of the pharmaceutical industry’s involvement in drug research. However, we should not forget that any private entity is prone to the same self-interest regardless of its product’s tastiness. How many of you knew that there was an avocado lobby funding research? No matter how many industry-funded observational studies using surrogate endpoints are out there telling you that chocolate is healthy, a randomized trial with hard clinical endpoints such as COSMOS should generally win the day.
The Final Goodbyes — Summarizing Your Case
As the party slowly winds down and everyone is saddened that you will soon take your leave, synthesize everything you have taught them over the evening. Like movies, not all studies are good. Some are just bad. They can be prone to reverse causation or confounding, and they may report relative risks when absolute risks would be more telling. Reading research studies critically is essential for separating the wheat from the chaff. With the knowledge you have now imparted to your friends, they will be much better consumers of medical news, especially when it comes to food.
And they will no doubt thank you for it by never inviting you to another dinner party!
Labos, a cardiologist at Hôpital, Notre-Dame, Montreal, Quebec, Canada, has disclosed no relevant financial relationships. He has a degree in epidemiology.
A version of this article appeared on Medscape.com.
You’re invited to a dinner party but you struggle to make small talk. Do not worry; that will invariably crop up over cocktails. Because all journalism has been reduced to listicles, here are four ways to seem clever at dinner parties.
1. The Predinner Cocktails: A Lesson in Reverse Causation
Wine connoisseurs sniff, swirl, and gently swish the wine in their mouths before spitting out and cleansing their palates to better appreciate the subtlety of each vintage. If you’re not an oenophile, no matter. Whenever somebody claims that moderate amounts of alcohol are good for your heart, this is your moment to pounce. Interject yourself in the conversation and tell everybody about reverse causation.
Reverse causation, also known as protopathic bias, involves misinterpreting the directionality of an association. You assume that X leads to Y, when in fact Y leads to X. Temporal paradoxes are useful plot devices in science fiction movies, but they have no place in medical research. In our bland world, cause must precede effect. As such, smoking leads to lung cancer; lung cancer doesn’t make you smoke more.
But with alcohol, directionality is less obvious. Many studies of alcohol and cardiovascular disease have demonstrated a U-shaped association, with risk being lowest among those who drink moderate amounts of alcohol (usually one to two drinks per day) and higher in those who drink more and also those who drink very little.
But one must ask why some people drink little or no alcohol. There is an important difference between former drinkers and never drinkers. Former drinkers cut back for a reason. More likely than not, the reason for this newfound sobriety was medical. A new cancer diagnosis, the emergence of atrial fibrillation, the development of diabetes, or rising blood pressure are all good reasons to reduce or eliminate alcohol. A cross-sectional study will fail to capture that alcohol consumption changes over time — people who now don’t drink may have imbibed alcohol in years past. It was not abstinence that led to an increased risk for heart disease; it was the increased risk for heart disease that led to abstinence.
You see the same phenomenon with the so-called obesity paradox. The idea that being a little overweight is good for you may appeal when you no longer fit into last year’s pants. But people who are underweight are so for a reason. Malnutrition, cachexia from cancer, or some other cause is almost certainly driving up the risk at the left-hand side of the U-shaped curve that makes the middle part seem better than it actually is.
Food consumption changes over time. A cross-sectional survey at one point in time cannot accurately capture past habits and distant exposures, especially for diseases such as heart disease and cancer that develop slowly over time. Studies on alcohol that try to overcome these shortcomings by eliminating former drinkers, or by using Mendelian randomization to better account for past exposure, do not show a cardiovascular benefit for moderate red wine drinking.
2. The Hors D’oeuvres — The Importance of RCTs
Now that you have made yourself the center of attention, it is time to cement your newfound reputation as a font of scientific knowledge. Most self-respecting hosts will serve smoked salmon as an amuse-bouche before the main meal. When someone mentions the health benefits of fish oils, you should take the opportunity to teach them about confounding.
Fish, especially cold-water fish from northern climates, have relatively high amounts of omega-3 fatty acids. Despite the plethora of observational studies suggesting a cardiovascular benefit, it’s now relatively clear that fish oil or omega-3 supplements have no medical benefit.
This will probably come as a shock to the worried well, but many studies, including VITAL and ASCEND, have demonstrated no cardiovascular or cancer benefit to supplementation with omega-3s. The reason is straightforward and explains why hormone replacement therapy, vitamin D, and myriad purported game-changers never panned out. Confounding is hard to overcome in observational research.
Prior to the publication of the Women’s Health Initiative (WHI) Study, hormone replacement therapy was routinely prescribed to postmenopausal women because numerous observational studies suggested a cardiovascular benefit. But with the publication of the WHI study, it became clear that much of that “benefit” was due to confounding. The women choosing to take hormones were more health conscious at baseline and healthier overall.
A similar phenomenon occurred during COVID. Patients with low serum vitamin D levels had worse outcomes, prompting many to suggest vitamin D supplementation as a possible treatment. Trials did not support the intervention because we’d overlooked the obvious. People with vitamin D deficiency have underlying health problems that contribute to the vitamin D deficiency. They are probably older, frailer, possibly with a poorer diet. No amount of statistical adjustment can account for all those differences, and some degree of residual confounding will always persist.
The only way to overcome confounding is with randomization. When patients are randomly assigned to one group or another, their baseline differences largely balance out if the randomization was performed properly and the groups were large enough. There is a role for observational research, such as in situations where ethics, cost, and practicality do not allow for a randomized controlled trial. But randomized controlled trials have largely put to rest the purported health benefits of over-the-counter fish oils, omega-3s, and vitamin D.
3. The Main Course — Absolute vs Relative Risk
When you get to the main course, all eyes will now be on you. You will almost certainly be called upon to pronounce on the harms or benefits of red meat consumption. Begin by regaling your guests with a little trivia. Ask them if they know the definition of red meat and white meat. When someone says pork is white meat, you can reveal that “pork, the other white meat,” was a marketing slogan with no scientific underpinning. Now that everyone is lulled into a stupefied silence, tell them that red meat comes from mammals and white meat comes from birds. As they process this revelation, you can now launch into the deeply mathematical concept of absolute vs relative risk.
Many etiquette books will caution against bringing up math at a dinner party. These books are wrong. Everyone finds math interesting if they are primed properly. For example, you can point to a study claiming that berries reduce cardiovascular risk in women. Even if true — and there is reason to be cautious, given the observational nature of the research — we need to understand what the authors meant by a 32% risk reduction. (Side note: It was a reduction in hazard, with a hazard ratio of 0.68 (95% CI, 0.49-0.96), but we won’t dwell on the difference between hazard ratios and risk ratios right now.)
This relative risk reduction has to be interpreted carefully. The authors divided the population into quintiles based on their consumption of anthocyanins (the antioxidant in blueberries and strawberries) and compared the bottom fifth (average consumption, 2.5 mg/d) with the top fifth (average consumption, 25 mg/d). The bottom quintile had 126 myocardial infarctions (MIs) over 324,793 patient-years compared with 59 MIs over 332,143 patient-years. Some quick math shows an approximate reduction from 39 to 18 MIs per 100,000 patient-years. Or to put it another way, you must get 4762 women to increase their berry consumption 10-fold for 1 year to prevent one heart attack. Feel free to show people how you calculated this number. They will be impressed by your head for numbers. It is nothing more than 39 minus 18, divided by 100,000, to get the absolute risk reduction. Take the reciprocal of this (ie, 1 divided by this number) to get the number needed to treat.
Describing risks in absolute terms or using number needed to treat (or harm) can help conceptualize statistics that are sometimes hard to wrap your head around.
4. Dessert — Funding
By the time the coffee is served, everyone will be hanging on to your every word. This is as it should be, and you should not be afraid of your newfound power and influence.
Dessert will probably involve some form of chocolate, possibly in cake format. (Anyone who serves fruit as dessert is not someone you should associate with.) Take the opportunity to tell your follow diners that chocolate is not actually good for you and will not boost brain performance.
The health benefits of chocolate are often repeated but rarely scrutinized. In fact, much of the scientific research purporting to show that chocolate is good for you did not actually study chocolate. It usually involved a cocoa bean extract because the chocolate manufacturing process destroys the supposedly health-promoting antioxidants in the cocoa bean. It is true that dark chocolate has more antioxidants than milk chocolate, and that the addition of milk to chocolate further inactivates the potentially healthy antioxidants. But the amount of sugar and fat that has to be added to chocolate to make it palatable precludes any serious consideration about health benefits. Dark chocolate may have less fat and sugar than milk chocolate, but it still has a lot.
But even the cocoa bean extract doesn’t seem to do much for your heart or your brain. The long-awaited COSMOS study was published with surprisingly little fanfare. The largest randomized controlled trial of chocolate (or rather cocoa bean extract) was supposed to settle the issue definitively.
COSMOS showed no cardiovascular or neurocognitive benefit to the cocoa bean extract. But the health halo of chocolate continues to be bolstered by many studies funded by chocolate manufacturers.
We are appropriately critical of the pharmaceutical industry’s involvement in drug research. However, we should not forget that any private entity is prone to the same self-interest regardless of its product’s tastiness. How many of you knew that there was an avocado lobby funding research? No matter how many industry-funded observational studies using surrogate endpoints are out there telling you that chocolate is healthy, a randomized trial with hard clinical endpoints such as COSMOS should generally win the day.
The Final Goodbyes — Summarizing Your Case
As the party slowly winds down and everyone is saddened that you will soon take your leave, synthesize everything you have taught them over the evening. Like movies, not all studies are good. Some are just bad. They can be prone to reverse causation or confounding, and they may report relative risks when absolute risks would be more telling. Reading research studies critically is essential for separating the wheat from the chaff. With the knowledge you have now imparted to your friends, they will be much better consumers of medical news, especially when it comes to food.
And they will no doubt thank you for it by never inviting you to another dinner party!
Labos, a cardiologist at Hôpital, Notre-Dame, Montreal, Quebec, Canada, has disclosed no relevant financial relationships. He has a degree in epidemiology.
A version of this article appeared on Medscape.com.
Vitamin D May Lower Blood Pressure in Seniors With Overweight
TOPLINE:
Supplementation with vitamin D and calcium can reduce systolic and diastolic blood pressure in older individuals with overweight, particularly in those with a body mass index (BMI) > 30 and those diagnosed with hypertension.
METHODOLOGY:
- Large cohort data have provided epidemiologic evidence linking vitamin D deficiency to a higher risk for cardiovascular disorders, including hypertension; however, evidence on the beneficial effects of vitamin D supplementation on blood pressure outcomes remains inconclusive.
- A post hoc analysis of a randomized controlled trial was conducted to investigate the effect of two doses of cholecalciferol (vitamin D3) on blood pressure in individuals aged 65 years or older with a BMI > 25 and serum vitamin D levels of 10-30 ng/mL.
- A total of 221 participants were recruited through outpatient departments, clinics, and advertisements in the greater Beirut area and received calcium supplementation in combination with either a low dose (600 IU/d, as recommended by the Institute of Medicine [IOM]) or a high dose (3750 IU/d) of vitamin D3.
- Blood pressure measurements were taken at baseline, 6 months, and 12 months using a SureSigns VS3 monitor.
- Participants were also stratified by BMI and hypertension status to assess the effects of vitamin D and calcium on blood pressure.
TAKEAWAY:
- Systolic and diastolic blood pressures were significantly reduced with vitamin D supplementation in the overall cohort (mean difference, 3.5 and 2.8 mm Hg, respectively; P = .005 and P = .002, respectively), with the effect more prominent in those in the high-dose vitamin D group.
- Participants with a BMI > 30 experienced reductions in both systolic and diastolic blood pressures in the overall cohort (P < .0001 and P = .01, respectively); although the systolic blood pressure was significantly reduced with both high- and low-dose vitamin D, the diastolic blood pressure decreased in the high-dose group only.
- Patients with hypertension benefited from all doses of vitamin D, regardless of the BMI.
- Systolic blood pressure at 6 and 12 months was significantly predicted by BMI and baseline systolic blood pressure measurements, although not by the dose of vitamin D received.
IN PRACTICE:
“Our study found vitamin D supplementation may decrease blood pressure in specific subgroups such as older people, people with obesity, and possibly those with low vitamin D levels,” said study author Ghada El-Hajj Fuleihan, MD, MPH, of the American University of Beirut Medical Center in Beirut, Lebanon, said in a news release. “High vitamin D doses compared to the IOM’s recommended daily dose did not provide additional health benefits.”
SOURCE:
This study was led by Maya Rahme, Department of Internal Medicine, Division of Endocrinology, Calcium Metabolism and Osteoporosis Program, World Health Organization Collaborating Center for Metabolic Bone Disorders, American University of Beirut Medical Center in Beirut, Lebanon. It was published online in Journal of the Endocrine Society.
LIMITATIONS:
This study’s limitations included the exploratory nature of the analyses and the low power of the subgroup analyses. Additionally, the study focused on older individuals who were sedentary and had overweight, many of whom had prediabetes — conditions known to influence blood pressure. The possible effect of calcium alone on blood pressure reduction was also unclear.
DISCLOSURES:
This study was supported by grants from the American University of Beirut, St Joseph University, and the Lebanese Council for National Scientific Research. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.
TOPLINE:
Supplementation with vitamin D and calcium can reduce systolic and diastolic blood pressure in older individuals with overweight, particularly in those with a body mass index (BMI) > 30 and those diagnosed with hypertension.
METHODOLOGY:
- Large cohort data have provided epidemiologic evidence linking vitamin D deficiency to a higher risk for cardiovascular disorders, including hypertension; however, evidence on the beneficial effects of vitamin D supplementation on blood pressure outcomes remains inconclusive.
- A post hoc analysis of a randomized controlled trial was conducted to investigate the effect of two doses of cholecalciferol (vitamin D3) on blood pressure in individuals aged 65 years or older with a BMI > 25 and serum vitamin D levels of 10-30 ng/mL.
- A total of 221 participants were recruited through outpatient departments, clinics, and advertisements in the greater Beirut area and received calcium supplementation in combination with either a low dose (600 IU/d, as recommended by the Institute of Medicine [IOM]) or a high dose (3750 IU/d) of vitamin D3.
- Blood pressure measurements were taken at baseline, 6 months, and 12 months using a SureSigns VS3 monitor.
- Participants were also stratified by BMI and hypertension status to assess the effects of vitamin D and calcium on blood pressure.
TAKEAWAY:
- Systolic and diastolic blood pressures were significantly reduced with vitamin D supplementation in the overall cohort (mean difference, 3.5 and 2.8 mm Hg, respectively; P = .005 and P = .002, respectively), with the effect more prominent in those in the high-dose vitamin D group.
- Participants with a BMI > 30 experienced reductions in both systolic and diastolic blood pressures in the overall cohort (P < .0001 and P = .01, respectively); although the systolic blood pressure was significantly reduced with both high- and low-dose vitamin D, the diastolic blood pressure decreased in the high-dose group only.
- Patients with hypertension benefited from all doses of vitamin D, regardless of the BMI.
- Systolic blood pressure at 6 and 12 months was significantly predicted by BMI and baseline systolic blood pressure measurements, although not by the dose of vitamin D received.
IN PRACTICE:
“Our study found vitamin D supplementation may decrease blood pressure in specific subgroups such as older people, people with obesity, and possibly those with low vitamin D levels,” said study author Ghada El-Hajj Fuleihan, MD, MPH, of the American University of Beirut Medical Center in Beirut, Lebanon, said in a news release. “High vitamin D doses compared to the IOM’s recommended daily dose did not provide additional health benefits.”
SOURCE:
This study was led by Maya Rahme, Department of Internal Medicine, Division of Endocrinology, Calcium Metabolism and Osteoporosis Program, World Health Organization Collaborating Center for Metabolic Bone Disorders, American University of Beirut Medical Center in Beirut, Lebanon. It was published online in Journal of the Endocrine Society.
LIMITATIONS:
This study’s limitations included the exploratory nature of the analyses and the low power of the subgroup analyses. Additionally, the study focused on older individuals who were sedentary and had overweight, many of whom had prediabetes — conditions known to influence blood pressure. The possible effect of calcium alone on blood pressure reduction was also unclear.
DISCLOSURES:
This study was supported by grants from the American University of Beirut, St Joseph University, and the Lebanese Council for National Scientific Research. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.
TOPLINE:
Supplementation with vitamin D and calcium can reduce systolic and diastolic blood pressure in older individuals with overweight, particularly in those with a body mass index (BMI) > 30 and those diagnosed with hypertension.
METHODOLOGY:
- Large cohort data have provided epidemiologic evidence linking vitamin D deficiency to a higher risk for cardiovascular disorders, including hypertension; however, evidence on the beneficial effects of vitamin D supplementation on blood pressure outcomes remains inconclusive.
- A post hoc analysis of a randomized controlled trial was conducted to investigate the effect of two doses of cholecalciferol (vitamin D3) on blood pressure in individuals aged 65 years or older with a BMI > 25 and serum vitamin D levels of 10-30 ng/mL.
- A total of 221 participants were recruited through outpatient departments, clinics, and advertisements in the greater Beirut area and received calcium supplementation in combination with either a low dose (600 IU/d, as recommended by the Institute of Medicine [IOM]) or a high dose (3750 IU/d) of vitamin D3.
- Blood pressure measurements were taken at baseline, 6 months, and 12 months using a SureSigns VS3 monitor.
- Participants were also stratified by BMI and hypertension status to assess the effects of vitamin D and calcium on blood pressure.
TAKEAWAY:
- Systolic and diastolic blood pressures were significantly reduced with vitamin D supplementation in the overall cohort (mean difference, 3.5 and 2.8 mm Hg, respectively; P = .005 and P = .002, respectively), with the effect more prominent in those in the high-dose vitamin D group.
- Participants with a BMI > 30 experienced reductions in both systolic and diastolic blood pressures in the overall cohort (P < .0001 and P = .01, respectively); although the systolic blood pressure was significantly reduced with both high- and low-dose vitamin D, the diastolic blood pressure decreased in the high-dose group only.
- Patients with hypertension benefited from all doses of vitamin D, regardless of the BMI.
- Systolic blood pressure at 6 and 12 months was significantly predicted by BMI and baseline systolic blood pressure measurements, although not by the dose of vitamin D received.
IN PRACTICE:
“Our study found vitamin D supplementation may decrease blood pressure in specific subgroups such as older people, people with obesity, and possibly those with low vitamin D levels,” said study author Ghada El-Hajj Fuleihan, MD, MPH, of the American University of Beirut Medical Center in Beirut, Lebanon, said in a news release. “High vitamin D doses compared to the IOM’s recommended daily dose did not provide additional health benefits.”
SOURCE:
This study was led by Maya Rahme, Department of Internal Medicine, Division of Endocrinology, Calcium Metabolism and Osteoporosis Program, World Health Organization Collaborating Center for Metabolic Bone Disorders, American University of Beirut Medical Center in Beirut, Lebanon. It was published online in Journal of the Endocrine Society.
LIMITATIONS:
This study’s limitations included the exploratory nature of the analyses and the low power of the subgroup analyses. Additionally, the study focused on older individuals who were sedentary and had overweight, many of whom had prediabetes — conditions known to influence blood pressure. The possible effect of calcium alone on blood pressure reduction was also unclear.
DISCLOSURES:
This study was supported by grants from the American University of Beirut, St Joseph University, and the Lebanese Council for National Scientific Research. No relevant conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.
PRECISE-DAPT Score Predicts GI Bleeding Risk Among Post-PCI Patients
PHILADELPHIA — Gastrointestinal (GI) bleeding after percutaneous coronary intervention (PCI) among patients on dual antiplatelet therapy (DAPT) remains risky in terms of morbidity and mortality, but the Predicting Bleeding Complications in Patients Undergoing Stent Implantation and Subsequent Dual Antiplatelet Therapy (PRECISE-DAPT) score could help predict that risk, according to a study presented at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
In a predominantly Hispanic population in Texas, 2.5% of post-PCI patients on DAPT had GI bleeding in the first year.
“Our study established that the PRECISE-DAPT score possesses a moderate predictive accuracy not only for overall bleeding risk but also specifically for gastrointestinal bleeding,” said lead author Jesus Guzman, MD, a gastroenterology fellow at the Texas Tech University Health Sciences Center El Paso.
Current guidelines from the American College of Cardiology and American Heart Association recommend DAPT for 6-12 months post-PCI, with consideration for shorter durations in patients with lower ischemic risks but higher bleeding risks.
“Interestingly, some of these patients were on DAPT for more than 2 years, which goes beyond the guidelines,” he said. “In this patient population, this has to do with them being lost to follow-up and getting reestablished, and they kept refilling their prescriptions.”
Guzman and colleagues conducted a retrospective cohort study of patients receiving DAPT after PCI from 2014 to 2021. They looked for GI bleeding rates at 1 year and across the duration of the study period, as well as endoscopic indications, findings, concurrent antiplatelet therapy, and the primary cause of bleeding.
In addition, the research team evaluated the predictive value of the PRECISE-DAPT score, which categorizes patients based on low risk (≤ 17), moderate risk (18-24), and high risk (≥ 25) for bleeding. The score aims to optimize the balance between bleeding and ischemic risks, Guzman said, by incorporating five factors: Age, creatinine clearance, hemoglobin, white blood cell count, and history of spontaneous bleeding.
Among 1067 patients, 563 (57.9%) received clopidogrel and 409 (42%) received ticagrelor. The overall cohort was 66.6% men, 77.1% Hispanic, and had a mean age of 62 years.
The GI bleeding rate was 2.5% at 1-year post-PCI among 27 patients and 3.7% for the study duration among 39 patients, with a median follow-up of 2.2 years.
Among the 39 GI bleeds, 41% were lower GI bleeds, 28% were upper GI bleeds, 15% were small bowel bleeds, and 15% were undetermined. The most frequent etiology was colon cancer, accounting for 18% of bleeds, followed by 15% for gastric ulcers, 10% for diverticular bleeds, and 10% for hemorrhoidal bleeds.
In general, analyses indicated no significant differences in GI bleeding between patients on clopidogrel (21.2%) and those on ticagrelor (19.2%).
However, the odds of GI bleeding were significantly higher in patients with high-risk PRECISE-DAPT scores (odds ratio [OR], 2.5) and moderate-risk scores (OR, 2.8) than in those with low-risk scores. The majority of patients without GI bleeding had scores < 17, whereas the majority of patients with GI bleeding had scores > 24. An optimal threshold for the PRECISE-DAPT score was identified as ≥ 19.
“When patients on DAPT present with GI bleeding, it can be a clinical conundrum for gastroenterologists and cardiologists, especially when it can be a life-or-death event, and stopping DAPT can increase risk of thrombosis,” said Jeff Taclob, MD, a hepatology fellow at The University of Tennessee Health Science Center in Memphis. Taclob, who wasn’t involved with the study, attended the conference session.
“In this population in El Paso, in particular, many patients don’t have adequate healthcare, may be lost to follow-up, and get their prescriptions filled elsewhere, such as Juárez, Mexico,” he said. “Then they come in with this life-threatening bleed, so we need to focus more on their risks.”
Paying attention to specific patient populations, cultures, and values remains important for patient communication and clinical decision-making, Taclob noted.
“In this population of older men, there’s often a macho persona where they don’t want to seek help,” he said. “DAPT criteria could differ in other populations, but here, the PRECISE-DAPT score appeared to help.”
The study was awarded the ACG Outstanding Research Award in the GI Bleeding Category (Trainee). Guzman and Taclob reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
PHILADELPHIA — Gastrointestinal (GI) bleeding after percutaneous coronary intervention (PCI) among patients on dual antiplatelet therapy (DAPT) remains risky in terms of morbidity and mortality, but the Predicting Bleeding Complications in Patients Undergoing Stent Implantation and Subsequent Dual Antiplatelet Therapy (PRECISE-DAPT) score could help predict that risk, according to a study presented at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
In a predominantly Hispanic population in Texas, 2.5% of post-PCI patients on DAPT had GI bleeding in the first year.
“Our study established that the PRECISE-DAPT score possesses a moderate predictive accuracy not only for overall bleeding risk but also specifically for gastrointestinal bleeding,” said lead author Jesus Guzman, MD, a gastroenterology fellow at the Texas Tech University Health Sciences Center El Paso.
Current guidelines from the American College of Cardiology and American Heart Association recommend DAPT for 6-12 months post-PCI, with consideration for shorter durations in patients with lower ischemic risks but higher bleeding risks.
“Interestingly, some of these patients were on DAPT for more than 2 years, which goes beyond the guidelines,” he said. “In this patient population, this has to do with them being lost to follow-up and getting reestablished, and they kept refilling their prescriptions.”
Guzman and colleagues conducted a retrospective cohort study of patients receiving DAPT after PCI from 2014 to 2021. They looked for GI bleeding rates at 1 year and across the duration of the study period, as well as endoscopic indications, findings, concurrent antiplatelet therapy, and the primary cause of bleeding.
In addition, the research team evaluated the predictive value of the PRECISE-DAPT score, which categorizes patients based on low risk (≤ 17), moderate risk (18-24), and high risk (≥ 25) for bleeding. The score aims to optimize the balance between bleeding and ischemic risks, Guzman said, by incorporating five factors: Age, creatinine clearance, hemoglobin, white blood cell count, and history of spontaneous bleeding.
Among 1067 patients, 563 (57.9%) received clopidogrel and 409 (42%) received ticagrelor. The overall cohort was 66.6% men, 77.1% Hispanic, and had a mean age of 62 years.
The GI bleeding rate was 2.5% at 1-year post-PCI among 27 patients and 3.7% for the study duration among 39 patients, with a median follow-up of 2.2 years.
Among the 39 GI bleeds, 41% were lower GI bleeds, 28% were upper GI bleeds, 15% were small bowel bleeds, and 15% were undetermined. The most frequent etiology was colon cancer, accounting for 18% of bleeds, followed by 15% for gastric ulcers, 10% for diverticular bleeds, and 10% for hemorrhoidal bleeds.
In general, analyses indicated no significant differences in GI bleeding between patients on clopidogrel (21.2%) and those on ticagrelor (19.2%).
However, the odds of GI bleeding were significantly higher in patients with high-risk PRECISE-DAPT scores (odds ratio [OR], 2.5) and moderate-risk scores (OR, 2.8) than in those with low-risk scores. The majority of patients without GI bleeding had scores < 17, whereas the majority of patients with GI bleeding had scores > 24. An optimal threshold for the PRECISE-DAPT score was identified as ≥ 19.
“When patients on DAPT present with GI bleeding, it can be a clinical conundrum for gastroenterologists and cardiologists, especially when it can be a life-or-death event, and stopping DAPT can increase risk of thrombosis,” said Jeff Taclob, MD, a hepatology fellow at The University of Tennessee Health Science Center in Memphis. Taclob, who wasn’t involved with the study, attended the conference session.
“In this population in El Paso, in particular, many patients don’t have adequate healthcare, may be lost to follow-up, and get their prescriptions filled elsewhere, such as Juárez, Mexico,” he said. “Then they come in with this life-threatening bleed, so we need to focus more on their risks.”
Paying attention to specific patient populations, cultures, and values remains important for patient communication and clinical decision-making, Taclob noted.
“In this population of older men, there’s often a macho persona where they don’t want to seek help,” he said. “DAPT criteria could differ in other populations, but here, the PRECISE-DAPT score appeared to help.”
The study was awarded the ACG Outstanding Research Award in the GI Bleeding Category (Trainee). Guzman and Taclob reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
PHILADELPHIA — Gastrointestinal (GI) bleeding after percutaneous coronary intervention (PCI) among patients on dual antiplatelet therapy (DAPT) remains risky in terms of morbidity and mortality, but the Predicting Bleeding Complications in Patients Undergoing Stent Implantation and Subsequent Dual Antiplatelet Therapy (PRECISE-DAPT) score could help predict that risk, according to a study presented at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
In a predominantly Hispanic population in Texas, 2.5% of post-PCI patients on DAPT had GI bleeding in the first year.
“Our study established that the PRECISE-DAPT score possesses a moderate predictive accuracy not only for overall bleeding risk but also specifically for gastrointestinal bleeding,” said lead author Jesus Guzman, MD, a gastroenterology fellow at the Texas Tech University Health Sciences Center El Paso.
Current guidelines from the American College of Cardiology and American Heart Association recommend DAPT for 6-12 months post-PCI, with consideration for shorter durations in patients with lower ischemic risks but higher bleeding risks.
“Interestingly, some of these patients were on DAPT for more than 2 years, which goes beyond the guidelines,” he said. “In this patient population, this has to do with them being lost to follow-up and getting reestablished, and they kept refilling their prescriptions.”
Guzman and colleagues conducted a retrospective cohort study of patients receiving DAPT after PCI from 2014 to 2021. They looked for GI bleeding rates at 1 year and across the duration of the study period, as well as endoscopic indications, findings, concurrent antiplatelet therapy, and the primary cause of bleeding.
In addition, the research team evaluated the predictive value of the PRECISE-DAPT score, which categorizes patients based on low risk (≤ 17), moderate risk (18-24), and high risk (≥ 25) for bleeding. The score aims to optimize the balance between bleeding and ischemic risks, Guzman said, by incorporating five factors: Age, creatinine clearance, hemoglobin, white blood cell count, and history of spontaneous bleeding.
Among 1067 patients, 563 (57.9%) received clopidogrel and 409 (42%) received ticagrelor. The overall cohort was 66.6% men, 77.1% Hispanic, and had a mean age of 62 years.
The GI bleeding rate was 2.5% at 1-year post-PCI among 27 patients and 3.7% for the study duration among 39 patients, with a median follow-up of 2.2 years.
Among the 39 GI bleeds, 41% were lower GI bleeds, 28% were upper GI bleeds, 15% were small bowel bleeds, and 15% were undetermined. The most frequent etiology was colon cancer, accounting for 18% of bleeds, followed by 15% for gastric ulcers, 10% for diverticular bleeds, and 10% for hemorrhoidal bleeds.
In general, analyses indicated no significant differences in GI bleeding between patients on clopidogrel (21.2%) and those on ticagrelor (19.2%).
However, the odds of GI bleeding were significantly higher in patients with high-risk PRECISE-DAPT scores (odds ratio [OR], 2.5) and moderate-risk scores (OR, 2.8) than in those with low-risk scores. The majority of patients without GI bleeding had scores < 17, whereas the majority of patients with GI bleeding had scores > 24. An optimal threshold for the PRECISE-DAPT score was identified as ≥ 19.
“When patients on DAPT present with GI bleeding, it can be a clinical conundrum for gastroenterologists and cardiologists, especially when it can be a life-or-death event, and stopping DAPT can increase risk of thrombosis,” said Jeff Taclob, MD, a hepatology fellow at The University of Tennessee Health Science Center in Memphis. Taclob, who wasn’t involved with the study, attended the conference session.
“In this population in El Paso, in particular, many patients don’t have adequate healthcare, may be lost to follow-up, and get their prescriptions filled elsewhere, such as Juárez, Mexico,” he said. “Then they come in with this life-threatening bleed, so we need to focus more on their risks.”
Paying attention to specific patient populations, cultures, and values remains important for patient communication and clinical decision-making, Taclob noted.
“In this population of older men, there’s often a macho persona where they don’t want to seek help,” he said. “DAPT criteria could differ in other populations, but here, the PRECISE-DAPT score appeared to help.”
The study was awarded the ACG Outstanding Research Award in the GI Bleeding Category (Trainee). Guzman and Taclob reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM ACG 2024