User login
Sternal SSIs, Mediastinitis Plummet Under Preop Decolonization Program
SAN DIEGO – Conducting preoperative nasal screening and decolonization of Staphylococcus aureus in patients undergoing cardiothoracic surgery led to a significant reduction in the rate of all sternal surgical site infections, including those attributable to S. aureus, results from a large single-center study showed.
"Staphylococcus aureus sternal surgical site infections [SSIs] are associated with significant morbidity and mortality," lead researcher Jennifer Madigan said in an interview following IDWeek 2012, the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society. "Multiple studies in the past have shown that screening and decolonization of S. aureus carriers are associated with a reduction in sternal SSIs."
One recent intervention that identified S. aureus nasal carriers concluded that S. aureus SSIs can be reduced by rapid screening and decolonization of nares on hospital admission (N. Engl. J. Med. 2010;362:9-17). "This study used [polymerase chain reaction testing] for identification of S. aureus nasal carriers, followed by treatment with mupirocin nasal ointment and chlorhexidine soap," said Ms. Madigan of the department of infection prevention and control at St. John Hospital and Medical Center, Detroit. "The results showed more than a 50% reduction in S. aureus infections."
Ms. Madigan and her associates compared the SSI rates 57 months before and 24 months after initiation of an S. aureus decolonization program for cardiothoracic surgery patients. For this program S. aureus nasal carriers were decolonized with mupirocin nasal ointment daily for 5 days and were asked to bathe with chlorhexidine gluconate rinse for 5 days immediately before surgery. The researchers reported results from 580 patients who were screened from April 2010 through March 2012. Of these patients, 118 (20%) tested positive for S. aureus colonization, including 34 (6%) who tested positive for methicillin-resistant S. aureus.
After the S. aureus decolonization program was initiated, the rate of postoperative sternal SSIs following coronary artery bypass grafting (CABG) decreased by 65% (from 76 infections per 1,416 cases before screening to 8 infections per 427 cases after screening; P = .0019), with a 75% drop in the number of mediastinitis cases (from 39 infections per 1,416 cases before screening to 3 infections per 427 cases after screening; P = .0106).
The researchers also found that sternal SSIs attributable to S. aureus dropped by 82% (from 39 infections per 1,416 cases before screening to 2 infections per 427 cases after screening; P = .0044), with S. aureus mediastinitis dropping by 87% (from 21 infections per 1,416 cases before screening to 1 infection per 427 cases after screening; P = .0337).
"We encourage hospitals that perform CABG surgeries to incorporate this [decolonization program] into their process," Ms. Madigan said. "The program is associated with significant reductions in infection, morbidity, and mortality. It provides a great tool to reduce the risk of patient harm. In addition, this may have a positive financial impact on hospitals as mediastinitis is no longer a reimbursable condition."
Ms. Madigan said that she had no relevant financial conflicts.
SAN DIEGO – Conducting preoperative nasal screening and decolonization of Staphylococcus aureus in patients undergoing cardiothoracic surgery led to a significant reduction in the rate of all sternal surgical site infections, including those attributable to S. aureus, results from a large single-center study showed.
"Staphylococcus aureus sternal surgical site infections [SSIs] are associated with significant morbidity and mortality," lead researcher Jennifer Madigan said in an interview following IDWeek 2012, the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society. "Multiple studies in the past have shown that screening and decolonization of S. aureus carriers are associated with a reduction in sternal SSIs."
One recent intervention that identified S. aureus nasal carriers concluded that S. aureus SSIs can be reduced by rapid screening and decolonization of nares on hospital admission (N. Engl. J. Med. 2010;362:9-17). "This study used [polymerase chain reaction testing] for identification of S. aureus nasal carriers, followed by treatment with mupirocin nasal ointment and chlorhexidine soap," said Ms. Madigan of the department of infection prevention and control at St. John Hospital and Medical Center, Detroit. "The results showed more than a 50% reduction in S. aureus infections."
Ms. Madigan and her associates compared the SSI rates 57 months before and 24 months after initiation of an S. aureus decolonization program for cardiothoracic surgery patients. For this program S. aureus nasal carriers were decolonized with mupirocin nasal ointment daily for 5 days and were asked to bathe with chlorhexidine gluconate rinse for 5 days immediately before surgery. The researchers reported results from 580 patients who were screened from April 2010 through March 2012. Of these patients, 118 (20%) tested positive for S. aureus colonization, including 34 (6%) who tested positive for methicillin-resistant S. aureus.
After the S. aureus decolonization program was initiated, the rate of postoperative sternal SSIs following coronary artery bypass grafting (CABG) decreased by 65% (from 76 infections per 1,416 cases before screening to 8 infections per 427 cases after screening; P = .0019), with a 75% drop in the number of mediastinitis cases (from 39 infections per 1,416 cases before screening to 3 infections per 427 cases after screening; P = .0106).
The researchers also found that sternal SSIs attributable to S. aureus dropped by 82% (from 39 infections per 1,416 cases before screening to 2 infections per 427 cases after screening; P = .0044), with S. aureus mediastinitis dropping by 87% (from 21 infections per 1,416 cases before screening to 1 infection per 427 cases after screening; P = .0337).
"We encourage hospitals that perform CABG surgeries to incorporate this [decolonization program] into their process," Ms. Madigan said. "The program is associated with significant reductions in infection, morbidity, and mortality. It provides a great tool to reduce the risk of patient harm. In addition, this may have a positive financial impact on hospitals as mediastinitis is no longer a reimbursable condition."
Ms. Madigan said that she had no relevant financial conflicts.
SAN DIEGO – Conducting preoperative nasal screening and decolonization of Staphylococcus aureus in patients undergoing cardiothoracic surgery led to a significant reduction in the rate of all sternal surgical site infections, including those attributable to S. aureus, results from a large single-center study showed.
"Staphylococcus aureus sternal surgical site infections [SSIs] are associated with significant morbidity and mortality," lead researcher Jennifer Madigan said in an interview following IDWeek 2012, the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society. "Multiple studies in the past have shown that screening and decolonization of S. aureus carriers are associated with a reduction in sternal SSIs."
One recent intervention that identified S. aureus nasal carriers concluded that S. aureus SSIs can be reduced by rapid screening and decolonization of nares on hospital admission (N. Engl. J. Med. 2010;362:9-17). "This study used [polymerase chain reaction testing] for identification of S. aureus nasal carriers, followed by treatment with mupirocin nasal ointment and chlorhexidine soap," said Ms. Madigan of the department of infection prevention and control at St. John Hospital and Medical Center, Detroit. "The results showed more than a 50% reduction in S. aureus infections."
Ms. Madigan and her associates compared the SSI rates 57 months before and 24 months after initiation of an S. aureus decolonization program for cardiothoracic surgery patients. For this program S. aureus nasal carriers were decolonized with mupirocin nasal ointment daily for 5 days and were asked to bathe with chlorhexidine gluconate rinse for 5 days immediately before surgery. The researchers reported results from 580 patients who were screened from April 2010 through March 2012. Of these patients, 118 (20%) tested positive for S. aureus colonization, including 34 (6%) who tested positive for methicillin-resistant S. aureus.
After the S. aureus decolonization program was initiated, the rate of postoperative sternal SSIs following coronary artery bypass grafting (CABG) decreased by 65% (from 76 infections per 1,416 cases before screening to 8 infections per 427 cases after screening; P = .0019), with a 75% drop in the number of mediastinitis cases (from 39 infections per 1,416 cases before screening to 3 infections per 427 cases after screening; P = .0106).
The researchers also found that sternal SSIs attributable to S. aureus dropped by 82% (from 39 infections per 1,416 cases before screening to 2 infections per 427 cases after screening; P = .0044), with S. aureus mediastinitis dropping by 87% (from 21 infections per 1,416 cases before screening to 1 infection per 427 cases after screening; P = .0337).
"We encourage hospitals that perform CABG surgeries to incorporate this [decolonization program] into their process," Ms. Madigan said. "The program is associated with significant reductions in infection, morbidity, and mortality. It provides a great tool to reduce the risk of patient harm. In addition, this may have a positive financial impact on hospitals as mediastinitis is no longer a reimbursable condition."
Ms. Madigan said that she had no relevant financial conflicts.
AT IDWEEK 2012
Major Finding: Following initiation of a preoperative S. aureus decolonization program, the rate of postoperative sternal SSIs following coronary artery bypass grafting dropped by 65%, and the rate of mediastinitis cases dropped by 75%.
Data Source: A single-center study of 580 cardiothoracic surgery patients who were screened from April 2010 through March 2012.
Disclosures: The researchers reported having no relevant financial conflicts.
FDA Expands Sapien Approval
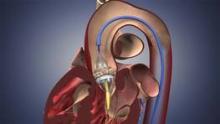
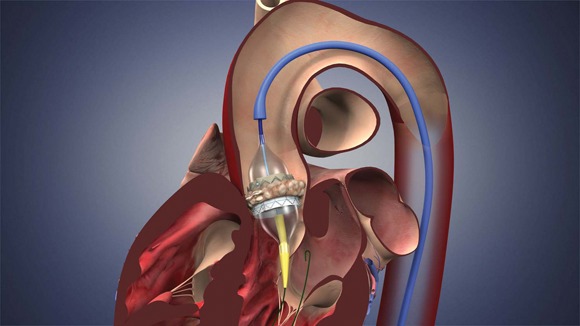
The Food and Drug Administration has expanded approval for the Sapien transcatheter heart valve for patients with aortic valve stenosis who are at high risk for serious surgical complications or death.
It is the second approval for the device, which is made by Edwards Lifesciences Corp. of Irvine, Calif. In November 2011, the FDA approved it for severe aortic valve stenosis in inoperable patients.
The indication for high-risk patients was reviewed by an FDA advisory panel in July. The majority of the panel indicated that the device was safe and effective in that group.
Both approvals were based on results from the PARTNER trial (N. Engl. J. Med. 2010;363:1597-607).
"Any procedure to replace the aortic valve carries the risk for serious complications, but for some patients with coexisting conditions or diseases, that risk may be especially high," Christy Foreman, director of the office of device evaluation at FDA’s Center for Devices and Radiological Health, said in a statement.
"The THV serves as an alternative for some very high-risk patients."
Larry L. Wood, Edwards’ corporate vice president, transcatheter heart valves, said in a statement: "It is extremely rewarding for us and our clinical partners to know that high-risk patients suffering from this often-debilitating disease will now have access to this life-saving therapy."
The Sapien THV is implanted at the site of the diseased valve using a catheter that is delivered either through a transfemoral or transapical approach. According to the Edwards statement, the transapical approach, which requires insertion through the ribs and myocardium, was not approved outside of clinical trials until now.
Transcatheter aortic valve replacement has become one of the driving forces in the heart valve market, which may hit $1.5 billion by 2016.
The new indication for the Sapien THV is expected to expand that market further. According to the FDA, the approved labeling indicates that a surgeon must determine whether a patient is eligible for the Sapien. But in fact, Edwards, the Centers for Medicare and Medicaid Services, and professional societies such as the American College of Cardiology and the Society of Thoracic Surgeons, have worked together to establish standards and requirements for "Heart Teams" that must be present during the procedure and evaluation of the patient by two cardiac surgeons.
There is an increased risk for major vascular complications and for stroke during the first month post implant. Thus, the device is contraindicated in patients who cannot tolerate anticoagulation/antiplatelet therapy.
Edwards will monitor safety and complication rates through the national Transcatheter Valve Therapy Registry.
The Food and Drug Administration has expanded approval for the Sapien transcatheter heart valve for patients with aortic valve stenosis who are at high risk for serious surgical complications or death.
It is the second approval for the device, which is made by Edwards Lifesciences Corp. of Irvine, Calif. In November 2011, the FDA approved it for severe aortic valve stenosis in inoperable patients.
The indication for high-risk patients was reviewed by an FDA advisory panel in July. The majority of the panel indicated that the device was safe and effective in that group.
Both approvals were based on results from the PARTNER trial (N. Engl. J. Med. 2010;363:1597-607).
"Any procedure to replace the aortic valve carries the risk for serious complications, but for some patients with coexisting conditions or diseases, that risk may be especially high," Christy Foreman, director of the office of device evaluation at FDA’s Center for Devices and Radiological Health, said in a statement.
"The THV serves as an alternative for some very high-risk patients."
Larry L. Wood, Edwards’ corporate vice president, transcatheter heart valves, said in a statement: "It is extremely rewarding for us and our clinical partners to know that high-risk patients suffering from this often-debilitating disease will now have access to this life-saving therapy."
The Sapien THV is implanted at the site of the diseased valve using a catheter that is delivered either through a transfemoral or transapical approach. According to the Edwards statement, the transapical approach, which requires insertion through the ribs and myocardium, was not approved outside of clinical trials until now.
Transcatheter aortic valve replacement has become one of the driving forces in the heart valve market, which may hit $1.5 billion by 2016.
The new indication for the Sapien THV is expected to expand that market further. According to the FDA, the approved labeling indicates that a surgeon must determine whether a patient is eligible for the Sapien. But in fact, Edwards, the Centers for Medicare and Medicaid Services, and professional societies such as the American College of Cardiology and the Society of Thoracic Surgeons, have worked together to establish standards and requirements for "Heart Teams" that must be present during the procedure and evaluation of the patient by two cardiac surgeons.
There is an increased risk for major vascular complications and for stroke during the first month post implant. Thus, the device is contraindicated in patients who cannot tolerate anticoagulation/antiplatelet therapy.
Edwards will monitor safety and complication rates through the national Transcatheter Valve Therapy Registry.
The Food and Drug Administration has expanded approval for the Sapien transcatheter heart valve for patients with aortic valve stenosis who are at high risk for serious surgical complications or death.
It is the second approval for the device, which is made by Edwards Lifesciences Corp. of Irvine, Calif. In November 2011, the FDA approved it for severe aortic valve stenosis in inoperable patients.
The indication for high-risk patients was reviewed by an FDA advisory panel in July. The majority of the panel indicated that the device was safe and effective in that group.
Both approvals were based on results from the PARTNER trial (N. Engl. J. Med. 2010;363:1597-607).
"Any procedure to replace the aortic valve carries the risk for serious complications, but for some patients with coexisting conditions or diseases, that risk may be especially high," Christy Foreman, director of the office of device evaluation at FDA’s Center for Devices and Radiological Health, said in a statement.
"The THV serves as an alternative for some very high-risk patients."
Larry L. Wood, Edwards’ corporate vice president, transcatheter heart valves, said in a statement: "It is extremely rewarding for us and our clinical partners to know that high-risk patients suffering from this often-debilitating disease will now have access to this life-saving therapy."
The Sapien THV is implanted at the site of the diseased valve using a catheter that is delivered either through a transfemoral or transapical approach. According to the Edwards statement, the transapical approach, which requires insertion through the ribs and myocardium, was not approved outside of clinical trials until now.
Transcatheter aortic valve replacement has become one of the driving forces in the heart valve market, which may hit $1.5 billion by 2016.
The new indication for the Sapien THV is expected to expand that market further. According to the FDA, the approved labeling indicates that a surgeon must determine whether a patient is eligible for the Sapien. But in fact, Edwards, the Centers for Medicare and Medicaid Services, and professional societies such as the American College of Cardiology and the Society of Thoracic Surgeons, have worked together to establish standards and requirements for "Heart Teams" that must be present during the procedure and evaluation of the patient by two cardiac surgeons.
There is an increased risk for major vascular complications and for stroke during the first month post implant. Thus, the device is contraindicated in patients who cannot tolerate anticoagulation/antiplatelet therapy.
Edwards will monitor safety and complication rates through the national Transcatheter Valve Therapy Registry.
Frailty Useful for TAVR Prognosis
Frailty was associated with a threefold increase in mortality risk 1 year after transcatheter aortic valve replacement, according to a single-center study published online Sept. 16 in JACC: Cardiovascular Interventions.
Researchers also found that there was no significant association between frailty status and the majority of post-TAVR procedural outcomes, suggesting that the current standard for patient selection is adequate, they wrote.
"We hope that the result will convince physicians to measure frailty," Dr. Philip Green, the study’s lead author, said in an interview. "Formally assessed frailty can be extremely useful for prognostic information."
Risk prediction for older adults undergoing cardiac surgery is somewhat tricky, said Dr. Green, because some of the well established measurement tools are based on studies that did not include many adults who were very old or at high risk.
"So the risk-prediction confidence intervals among the highest risk patients tend to get very wide," said Dr. Green, a fellow in cardiovascular medicine at Columbia University Medical Center, New York, where the study was conducted.
Frailty, which is the loss of resiliency and physiological reserve, helps predict the patients’ tolerance for certain procedures, their odds of survival, and their overall prognosis.
For an objective frailty score, Dr. Green said he and colleagues built on findings from previous studies and measured gait speed, grip strength, serum albumin, and activities of daily living to derive a frailty score in 159 very-high-risk patients with severe aortic stenosis who underwent TAVR at the Valve Center at Columbia University Medical Center/New York–Presbyterian Hospital.
They then broke down the patients into two groups based on their median frailty score: those who were frail and those who were not.
Patients’ mean age was 86 years, and half were men. Half of the patients had at least three comorbidities, although the frailty score was not associated with the number of comorbidities.
Overall, 76 patients had a frailty score higher than 5, and 83 had a score of 5 or less (considered not frail).
Patients whose frailty score was higher than 5 had longer hospital stays and were at a higher risk of in-hospital, life-threatening, or major bleeding events compared with the nonfrail group (JACC Cardiovasc. Interv. 2012;5:974-81).
Eight patients died during the first 30 days; however, frailty status was not associated with adverse periprocedural events such as vascular complications, stroke, or procedural mortality.
Meanwhile, 1-year follow-up showed that patients with a frailty score of more than 5 had a threefold increase in mortality after the procedure, compared with the nonfrail group (17 frail vs. 7 nonfrail; hazard ratio, 1.15).
"But it’s really important to distinguish between frailty and futility [of the procedure]," said Dr. Green. "Even the frail group had an 80% survival rate, and that suggests that even the most frail can tolerate and live for a long time after TAVR."
He added that the study did not address the patients’ quality of life, which could be a subject for another study.
The authors pointed out a few methodological issues.
For one, all the patients were carefully selected for TAVR, and hence it is not clear whether the findings can be generalized to unselected or lower-risk patients, or to patients who undergo surgical aortic valve replacement (SAVR).
Also, the components of the frailty score used are somewhat of a departure from the previously validated assessment tools.
"For our study, we had to raise the threshold for who’s frail," said Dr. Green. "We’re really talking about the frailest of the frail. Nevertheless we saw excellent outcome."
But for now, "the bottom line is measure frailty," advised Dr. Green. "Understand the functional status of your patients. Understand their abilities to perform activities and their nutritional status. It can shed light on patients who are thriving despite their heart disease and other comorbidities compared to those who are really limited on the basis of their diseases."
Dr. Green said that he had no relevant financial disclosures.
Frailty was associated with a threefold increase in mortality risk 1 year after transcatheter aortic valve replacement, according to a single-center study published online Sept. 16 in JACC: Cardiovascular Interventions.
Researchers also found that there was no significant association between frailty status and the majority of post-TAVR procedural outcomes, suggesting that the current standard for patient selection is adequate, they wrote.
"We hope that the result will convince physicians to measure frailty," Dr. Philip Green, the study’s lead author, said in an interview. "Formally assessed frailty can be extremely useful for prognostic information."
Risk prediction for older adults undergoing cardiac surgery is somewhat tricky, said Dr. Green, because some of the well established measurement tools are based on studies that did not include many adults who were very old or at high risk.
"So the risk-prediction confidence intervals among the highest risk patients tend to get very wide," said Dr. Green, a fellow in cardiovascular medicine at Columbia University Medical Center, New York, where the study was conducted.
Frailty, which is the loss of resiliency and physiological reserve, helps predict the patients’ tolerance for certain procedures, their odds of survival, and their overall prognosis.
For an objective frailty score, Dr. Green said he and colleagues built on findings from previous studies and measured gait speed, grip strength, serum albumin, and activities of daily living to derive a frailty score in 159 very-high-risk patients with severe aortic stenosis who underwent TAVR at the Valve Center at Columbia University Medical Center/New York–Presbyterian Hospital.
They then broke down the patients into two groups based on their median frailty score: those who were frail and those who were not.
Patients’ mean age was 86 years, and half were men. Half of the patients had at least three comorbidities, although the frailty score was not associated with the number of comorbidities.
Overall, 76 patients had a frailty score higher than 5, and 83 had a score of 5 or less (considered not frail).
Patients whose frailty score was higher than 5 had longer hospital stays and were at a higher risk of in-hospital, life-threatening, or major bleeding events compared with the nonfrail group (JACC Cardiovasc. Interv. 2012;5:974-81).
Eight patients died during the first 30 days; however, frailty status was not associated with adverse periprocedural events such as vascular complications, stroke, or procedural mortality.
Meanwhile, 1-year follow-up showed that patients with a frailty score of more than 5 had a threefold increase in mortality after the procedure, compared with the nonfrail group (17 frail vs. 7 nonfrail; hazard ratio, 1.15).
"But it’s really important to distinguish between frailty and futility [of the procedure]," said Dr. Green. "Even the frail group had an 80% survival rate, and that suggests that even the most frail can tolerate and live for a long time after TAVR."
He added that the study did not address the patients’ quality of life, which could be a subject for another study.
The authors pointed out a few methodological issues.
For one, all the patients were carefully selected for TAVR, and hence it is not clear whether the findings can be generalized to unselected or lower-risk patients, or to patients who undergo surgical aortic valve replacement (SAVR).
Also, the components of the frailty score used are somewhat of a departure from the previously validated assessment tools.
"For our study, we had to raise the threshold for who’s frail," said Dr. Green. "We’re really talking about the frailest of the frail. Nevertheless we saw excellent outcome."
But for now, "the bottom line is measure frailty," advised Dr. Green. "Understand the functional status of your patients. Understand their abilities to perform activities and their nutritional status. It can shed light on patients who are thriving despite their heart disease and other comorbidities compared to those who are really limited on the basis of their diseases."
Dr. Green said that he had no relevant financial disclosures.
Frailty was associated with a threefold increase in mortality risk 1 year after transcatheter aortic valve replacement, according to a single-center study published online Sept. 16 in JACC: Cardiovascular Interventions.
Researchers also found that there was no significant association between frailty status and the majority of post-TAVR procedural outcomes, suggesting that the current standard for patient selection is adequate, they wrote.
"We hope that the result will convince physicians to measure frailty," Dr. Philip Green, the study’s lead author, said in an interview. "Formally assessed frailty can be extremely useful for prognostic information."
Risk prediction for older adults undergoing cardiac surgery is somewhat tricky, said Dr. Green, because some of the well established measurement tools are based on studies that did not include many adults who were very old or at high risk.
"So the risk-prediction confidence intervals among the highest risk patients tend to get very wide," said Dr. Green, a fellow in cardiovascular medicine at Columbia University Medical Center, New York, where the study was conducted.
Frailty, which is the loss of resiliency and physiological reserve, helps predict the patients’ tolerance for certain procedures, their odds of survival, and their overall prognosis.
For an objective frailty score, Dr. Green said he and colleagues built on findings from previous studies and measured gait speed, grip strength, serum albumin, and activities of daily living to derive a frailty score in 159 very-high-risk patients with severe aortic stenosis who underwent TAVR at the Valve Center at Columbia University Medical Center/New York–Presbyterian Hospital.
They then broke down the patients into two groups based on their median frailty score: those who were frail and those who were not.
Patients’ mean age was 86 years, and half were men. Half of the patients had at least three comorbidities, although the frailty score was not associated with the number of comorbidities.
Overall, 76 patients had a frailty score higher than 5, and 83 had a score of 5 or less (considered not frail).
Patients whose frailty score was higher than 5 had longer hospital stays and were at a higher risk of in-hospital, life-threatening, or major bleeding events compared with the nonfrail group (JACC Cardiovasc. Interv. 2012;5:974-81).
Eight patients died during the first 30 days; however, frailty status was not associated with adverse periprocedural events such as vascular complications, stroke, or procedural mortality.
Meanwhile, 1-year follow-up showed that patients with a frailty score of more than 5 had a threefold increase in mortality after the procedure, compared with the nonfrail group (17 frail vs. 7 nonfrail; hazard ratio, 1.15).
"But it’s really important to distinguish between frailty and futility [of the procedure]," said Dr. Green. "Even the frail group had an 80% survival rate, and that suggests that even the most frail can tolerate and live for a long time after TAVR."
He added that the study did not address the patients’ quality of life, which could be a subject for another study.
The authors pointed out a few methodological issues.
For one, all the patients were carefully selected for TAVR, and hence it is not clear whether the findings can be generalized to unselected or lower-risk patients, or to patients who undergo surgical aortic valve replacement (SAVR).
Also, the components of the frailty score used are somewhat of a departure from the previously validated assessment tools.
"For our study, we had to raise the threshold for who’s frail," said Dr. Green. "We’re really talking about the frailest of the frail. Nevertheless we saw excellent outcome."
But for now, "the bottom line is measure frailty," advised Dr. Green. "Understand the functional status of your patients. Understand their abilities to perform activities and their nutritional status. It can shed light on patients who are thriving despite their heart disease and other comorbidities compared to those who are really limited on the basis of their diseases."
Dr. Green said that he had no relevant financial disclosures.
FROM JACC: CARDIOVASCULAR INTERVENTIONS
Major Finding: Frailty was associated with a threefold increase in mortality risk over the first year after transcatheter aortic valve replacement.
Data Source: Measurement of gait speed, grip strength, serum albumin, and activities of daily living to derive a frailty score in 159 very high risk patients with severe aortic stenosis who underwent TAVR at one U.S. center.
Disclosures: Dr. Green said that he had no relevant financial disclosures.
Real-World TAVI Outcomes Spot On With Clinical Trial Results
MUNICH – In-hospital mortality and complications following transcatheter aortic valve implantation in the real-world setting are comparable with those achieved in randomized controlled studies, according to data from the first large-scale national registry capturing surgical and catheter-based procedures.
"In high-risk patients, the in-hospital mortality with TAVI is at least as good, if not better, as with the surgical approach," said Dr. Christian Hamm, who presented data from 13,860 patients in the German Aortic Valve Registry (GARY during a hotline session at the annual congress of the European Society of Cardiology.
In-hospital mortality was 2.2% for conventional aortic valve replacement surgery alone, 4.6% for conventional surgery with coronary artery bypass grafting (CABG), 5.5% for TAVI using a transvascular approach, and 7.8% for TAVI with a transapical approach.
After adjusting for the expected higher risk profile of TAVI patients, the risk of in-hospital death was not increased with transvascular TAVI, compared with surgery alone, although there was a trend toward increased risk with transapical TAVI in both younger patients and those aged 75 years and older, said Dr. Hamm, medical director of the Kerckhoff Heart and Thorax Centre, Bad Nauheim, Germany.
When asked by the media whether the higher mortality may be diminishing use of the transapical approach, GARY coauthor Dr. Friedrich-Wilhelm Mohr said that similar rates have been seen in the source registry and that these patients tend to have slightly more comorbidities and vascular disease.
"The current data in 2012 do not show a pull back," said Dr. Mohr, medical director of the Leipzig (Germany) Heart Center. "It’s almost the same situation as here: two-thirds transfemoral and one-third transapical."
Consistent with earlier observations, the rate of cerebrovascular events was lowest in patients undergoing surgery without CABG at 2.2% and was "in the range of 3.5%" for all other groups, Dr. Hamm reported.
Discussant Dr. Olaf Wendler said it’s convincing that cerebrovascular complications are lower than in the landmark PARTNER (Placement of Aortic Transcatheter Valves) trial, which reported a 5% major stroke rate among TAVI patients at 30 days.
"This may be less of a problem than we thought about transcatheter-valve treatment in the past," he said. "However, again we don’t have all the details, definitions of the adverse events, and we don’t have a clear idea of how adverse events were adjudicated in this registry."
Rates of vascular complications and new onset atrioventricular (AV) blockage were highest after transvascular TAVI, while renal failure was most common after the transapical approach, said Dr. Wendler, professor at King’s College Hospital in London.
Specifically, vascular complications were reported in 12% of transvascular patients, compared with 2.5% for the transapical approach, 2% for surgery plus CABG, and 1% for surgery alone.
Residual postimplant aortic regurgitation, which has been linked to long-term TAVI outcome, "seemed to be excellent because more than 90% of patients had no or grade I regurgitation," Dr. Hamm said. Only 7% of transvascular and 3% of transapical patients had grade II regurgitation, and only 1% of transapical and no transvascular patients had grade III or IV regurgitation.
Although between-valve comparisons were limited since the CoreValve was not implanted transapically, grade I or II aortic regurgitation was more common with the CoreValve (70%) than with the Edwards prosthesis (45.7%).
Despite the increasing shift of TAVI in lower-risk patients, data from GARY show that TAVI is being performed predominantly in high-risk patients, as recommended in various guidelines, Dr. Hamm said.
Participation in GARY is voluntary, with 92 of 99 German centers currently taking part. As of July 2012, more than 26,000 patients were included, of whom 23% were TAVI patients.
The current analysis included 13,860 patients from 53 cardiac surgery units and 69 cardiology units. Of these patients, 6,523 underwent surgery alone, 3,462 surgery plus CABG, 2,694 transvascular TAVI, and 1,181 transapical TAVI.
At baseline, TAVI patients were significantly older than surgical patients (average of 81 years transvascular and 80.3 years transapical vs. 68 years surgical); had significantly more comorbidities including atrial fibrillation, hypertension, and diabetes; and were significantly more likely to have a left ventricular ejection fraction below 30%.
Roughly half of elderly patients more than 75 years of age now receive TAVI in Germany, which is quite impressive and by far a higher number than any other country worldwide, said Dr. Wendler.
German investigators also have developed a novel risk scoring system called the AKL score, which, unlike the logistic EuroSCORE or Society of Thoracic Surgeons (STS) risk score, is based only on patients with aortic valve disease.
"The EuroSCORE, as many of us know, is not very suitable in this scenario," Dr. Hamm said.
When the GARY authors compared observed vs. expected in-hospital mortality based on patients’ EuroSCORE, the observed risk for mortality was much lower than predicted by the EuroSCORE.
When the same analysis was performed using the AKL score, however, the observed and expected mortality outcomes were very similar, suggesting the "AKL score much better reflects the real outcome of the patients," he said.
Dr. Wendler remarked that in low-risk patients, however, the observed in-hospital mortality was higher than what was predicted, "questioning if this is the right treatment for this group of patients."
GARY is supported by the German Heart Foundation and unrestricted grants from heart valve manufacturers. Dr. Hamm disclosed honoraria from Medtronic and Edwards Lifesciences and participation in clinical trials with Medtronic, Edwards, Symetis, and JenaValve. Dr. Mohr reported no conflicts of interest. Dr. Wendler reported research ties with Edwards and consulting for Edwards and St. Jude Medical.
MUNICH – In-hospital mortality and complications following transcatheter aortic valve implantation in the real-world setting are comparable with those achieved in randomized controlled studies, according to data from the first large-scale national registry capturing surgical and catheter-based procedures.
"In high-risk patients, the in-hospital mortality with TAVI is at least as good, if not better, as with the surgical approach," said Dr. Christian Hamm, who presented data from 13,860 patients in the German Aortic Valve Registry (GARY during a hotline session at the annual congress of the European Society of Cardiology.
In-hospital mortality was 2.2% for conventional aortic valve replacement surgery alone, 4.6% for conventional surgery with coronary artery bypass grafting (CABG), 5.5% for TAVI using a transvascular approach, and 7.8% for TAVI with a transapical approach.
After adjusting for the expected higher risk profile of TAVI patients, the risk of in-hospital death was not increased with transvascular TAVI, compared with surgery alone, although there was a trend toward increased risk with transapical TAVI in both younger patients and those aged 75 years and older, said Dr. Hamm, medical director of the Kerckhoff Heart and Thorax Centre, Bad Nauheim, Germany.
When asked by the media whether the higher mortality may be diminishing use of the transapical approach, GARY coauthor Dr. Friedrich-Wilhelm Mohr said that similar rates have been seen in the source registry and that these patients tend to have slightly more comorbidities and vascular disease.
"The current data in 2012 do not show a pull back," said Dr. Mohr, medical director of the Leipzig (Germany) Heart Center. "It’s almost the same situation as here: two-thirds transfemoral and one-third transapical."
Consistent with earlier observations, the rate of cerebrovascular events was lowest in patients undergoing surgery without CABG at 2.2% and was "in the range of 3.5%" for all other groups, Dr. Hamm reported.
Discussant Dr. Olaf Wendler said it’s convincing that cerebrovascular complications are lower than in the landmark PARTNER (Placement of Aortic Transcatheter Valves) trial, which reported a 5% major stroke rate among TAVI patients at 30 days.
"This may be less of a problem than we thought about transcatheter-valve treatment in the past," he said. "However, again we don’t have all the details, definitions of the adverse events, and we don’t have a clear idea of how adverse events were adjudicated in this registry."
Rates of vascular complications and new onset atrioventricular (AV) blockage were highest after transvascular TAVI, while renal failure was most common after the transapical approach, said Dr. Wendler, professor at King’s College Hospital in London.
Specifically, vascular complications were reported in 12% of transvascular patients, compared with 2.5% for the transapical approach, 2% for surgery plus CABG, and 1% for surgery alone.
Residual postimplant aortic regurgitation, which has been linked to long-term TAVI outcome, "seemed to be excellent because more than 90% of patients had no or grade I regurgitation," Dr. Hamm said. Only 7% of transvascular and 3% of transapical patients had grade II regurgitation, and only 1% of transapical and no transvascular patients had grade III or IV regurgitation.
Although between-valve comparisons were limited since the CoreValve was not implanted transapically, grade I or II aortic regurgitation was more common with the CoreValve (70%) than with the Edwards prosthesis (45.7%).
Despite the increasing shift of TAVI in lower-risk patients, data from GARY show that TAVI is being performed predominantly in high-risk patients, as recommended in various guidelines, Dr. Hamm said.
Participation in GARY is voluntary, with 92 of 99 German centers currently taking part. As of July 2012, more than 26,000 patients were included, of whom 23% were TAVI patients.
The current analysis included 13,860 patients from 53 cardiac surgery units and 69 cardiology units. Of these patients, 6,523 underwent surgery alone, 3,462 surgery plus CABG, 2,694 transvascular TAVI, and 1,181 transapical TAVI.
At baseline, TAVI patients were significantly older than surgical patients (average of 81 years transvascular and 80.3 years transapical vs. 68 years surgical); had significantly more comorbidities including atrial fibrillation, hypertension, and diabetes; and were significantly more likely to have a left ventricular ejection fraction below 30%.
Roughly half of elderly patients more than 75 years of age now receive TAVI in Germany, which is quite impressive and by far a higher number than any other country worldwide, said Dr. Wendler.
German investigators also have developed a novel risk scoring system called the AKL score, which, unlike the logistic EuroSCORE or Society of Thoracic Surgeons (STS) risk score, is based only on patients with aortic valve disease.
"The EuroSCORE, as many of us know, is not very suitable in this scenario," Dr. Hamm said.
When the GARY authors compared observed vs. expected in-hospital mortality based on patients’ EuroSCORE, the observed risk for mortality was much lower than predicted by the EuroSCORE.
When the same analysis was performed using the AKL score, however, the observed and expected mortality outcomes were very similar, suggesting the "AKL score much better reflects the real outcome of the patients," he said.
Dr. Wendler remarked that in low-risk patients, however, the observed in-hospital mortality was higher than what was predicted, "questioning if this is the right treatment for this group of patients."
GARY is supported by the German Heart Foundation and unrestricted grants from heart valve manufacturers. Dr. Hamm disclosed honoraria from Medtronic and Edwards Lifesciences and participation in clinical trials with Medtronic, Edwards, Symetis, and JenaValve. Dr. Mohr reported no conflicts of interest. Dr. Wendler reported research ties with Edwards and consulting for Edwards and St. Jude Medical.
MUNICH – In-hospital mortality and complications following transcatheter aortic valve implantation in the real-world setting are comparable with those achieved in randomized controlled studies, according to data from the first large-scale national registry capturing surgical and catheter-based procedures.
"In high-risk patients, the in-hospital mortality with TAVI is at least as good, if not better, as with the surgical approach," said Dr. Christian Hamm, who presented data from 13,860 patients in the German Aortic Valve Registry (GARY during a hotline session at the annual congress of the European Society of Cardiology.
In-hospital mortality was 2.2% for conventional aortic valve replacement surgery alone, 4.6% for conventional surgery with coronary artery bypass grafting (CABG), 5.5% for TAVI using a transvascular approach, and 7.8% for TAVI with a transapical approach.
After adjusting for the expected higher risk profile of TAVI patients, the risk of in-hospital death was not increased with transvascular TAVI, compared with surgery alone, although there was a trend toward increased risk with transapical TAVI in both younger patients and those aged 75 years and older, said Dr. Hamm, medical director of the Kerckhoff Heart and Thorax Centre, Bad Nauheim, Germany.
When asked by the media whether the higher mortality may be diminishing use of the transapical approach, GARY coauthor Dr. Friedrich-Wilhelm Mohr said that similar rates have been seen in the source registry and that these patients tend to have slightly more comorbidities and vascular disease.
"The current data in 2012 do not show a pull back," said Dr. Mohr, medical director of the Leipzig (Germany) Heart Center. "It’s almost the same situation as here: two-thirds transfemoral and one-third transapical."
Consistent with earlier observations, the rate of cerebrovascular events was lowest in patients undergoing surgery without CABG at 2.2% and was "in the range of 3.5%" for all other groups, Dr. Hamm reported.
Discussant Dr. Olaf Wendler said it’s convincing that cerebrovascular complications are lower than in the landmark PARTNER (Placement of Aortic Transcatheter Valves) trial, which reported a 5% major stroke rate among TAVI patients at 30 days.
"This may be less of a problem than we thought about transcatheter-valve treatment in the past," he said. "However, again we don’t have all the details, definitions of the adverse events, and we don’t have a clear idea of how adverse events were adjudicated in this registry."
Rates of vascular complications and new onset atrioventricular (AV) blockage were highest after transvascular TAVI, while renal failure was most common after the transapical approach, said Dr. Wendler, professor at King’s College Hospital in London.
Specifically, vascular complications were reported in 12% of transvascular patients, compared with 2.5% for the transapical approach, 2% for surgery plus CABG, and 1% for surgery alone.
Residual postimplant aortic regurgitation, which has been linked to long-term TAVI outcome, "seemed to be excellent because more than 90% of patients had no or grade I regurgitation," Dr. Hamm said. Only 7% of transvascular and 3% of transapical patients had grade II regurgitation, and only 1% of transapical and no transvascular patients had grade III or IV regurgitation.
Although between-valve comparisons were limited since the CoreValve was not implanted transapically, grade I or II aortic regurgitation was more common with the CoreValve (70%) than with the Edwards prosthesis (45.7%).
Despite the increasing shift of TAVI in lower-risk patients, data from GARY show that TAVI is being performed predominantly in high-risk patients, as recommended in various guidelines, Dr. Hamm said.
Participation in GARY is voluntary, with 92 of 99 German centers currently taking part. As of July 2012, more than 26,000 patients were included, of whom 23% were TAVI patients.
The current analysis included 13,860 patients from 53 cardiac surgery units and 69 cardiology units. Of these patients, 6,523 underwent surgery alone, 3,462 surgery plus CABG, 2,694 transvascular TAVI, and 1,181 transapical TAVI.
At baseline, TAVI patients were significantly older than surgical patients (average of 81 years transvascular and 80.3 years transapical vs. 68 years surgical); had significantly more comorbidities including atrial fibrillation, hypertension, and diabetes; and were significantly more likely to have a left ventricular ejection fraction below 30%.
Roughly half of elderly patients more than 75 years of age now receive TAVI in Germany, which is quite impressive and by far a higher number than any other country worldwide, said Dr. Wendler.
German investigators also have developed a novel risk scoring system called the AKL score, which, unlike the logistic EuroSCORE or Society of Thoracic Surgeons (STS) risk score, is based only on patients with aortic valve disease.
"The EuroSCORE, as many of us know, is not very suitable in this scenario," Dr. Hamm said.
When the GARY authors compared observed vs. expected in-hospital mortality based on patients’ EuroSCORE, the observed risk for mortality was much lower than predicted by the EuroSCORE.
When the same analysis was performed using the AKL score, however, the observed and expected mortality outcomes were very similar, suggesting the "AKL score much better reflects the real outcome of the patients," he said.
Dr. Wendler remarked that in low-risk patients, however, the observed in-hospital mortality was higher than what was predicted, "questioning if this is the right treatment for this group of patients."
GARY is supported by the German Heart Foundation and unrestricted grants from heart valve manufacturers. Dr. Hamm disclosed honoraria from Medtronic and Edwards Lifesciences and participation in clinical trials with Medtronic, Edwards, Symetis, and JenaValve. Dr. Mohr reported no conflicts of interest. Dr. Wendler reported research ties with Edwards and consulting for Edwards and St. Jude Medical.
AT THE ANNUAL CONGRESS OF THE EUROPEAN SOCIETY OF CARDIOLOGY
Major Finding: In-hospital mortality was 2.2% for conventional aortic valve replacement surgery, 4.6% for conventional surgery with coronary artery bypass grafting, 5.5% for transvascular TAVI, and 7.8% for transapical TAVI.
Data Source: Data are from 13,860 patients treated with TAVI in 2011 in the German Aortic Valve Registry.
Disclosures: GARY is supported by the German Heart Foundation and unrestricted grants from heart valve manufacturers. Dr. Hamm disclosed honoraria from Medtronic and Edwards Lifesciences and participation in clinical trials with Medtronic, Edwards, Symetis, and JenaValve. Dr. Mohr reported no conflicts of interest. Dr. Wendler reported research ties with Edwards and consulting for Edwards and St. Jude Medical.
Early Lung Cancer Patients Living Longer After Radiotherapy
Median overall survival increased significantly among patients with stage I non–small cell lung cancer over the last decade – in particular, those treated with radiation therapy alone, according to an analysis of the Surveillance, Epidemiology, and End Results database.
The median survival for all treatment groups increased by 27%, from 44 months during 1999-2003 to 56 months during 2004-2008. For those treated with radiation alone – who would likely be the sickest patients since they would not have been considered candidates for surgery – median overall survival improved by 31%, from 16 to 21 months. Both changes were statistically significant (log rank P less than .0001).
"Stage I NSCLC [non–small cell lung cancer] patients who receive radiation therapy alone are surviving longer than they used to," Dr. Nirav S. Kapadia said in a press briefing from the Chicago Multidisciplinary Symposium in Thoracic Oncology.
A change in the survival of patients treated with surgery could not be detected, as median survival has not yet been reached, he and his coauthors reported.
Until recently, surgery has been the primary treatment for stage I NSCLC. However, as recent advances in radiotherapy (RT) such as stereotactic body radiation therapy have allowed dose escalation with more precise tumor targeting, the use of RT has increased, and outcomes appear to have improved over time, said Dr. Kapadia, a chief resident in the department of radiation oncology at the University of Michigan, Ann Arbor
The National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) database encompasses about 25% of the U.S. population. This study compared SEER data on 27,469 patients with NSCLC treated during 1999-2003 with data from 26,195 patients treated during 2004-2008.
During 1999-2003, 64% of patients were treated with primary surgery, 14% received RT alone, 20% had neither treatment, and 2% had unknown treatment. In the later era, 70% of patients underwent primary surgery, 13% received primary RT, 16% had neither surgery nor RT, and 1% had unknown treatment.
The proportion receiving surgery alone increased from 60% to 67% during the two time periods. Thus, the rates of surgery increased from the earlier to the later period, but there was no significant difference in the number of patients who received radiotherapy, either as an adjunct to surgery or as definitive therapy, noted Dr. Kapadia.
He expressed concern about the significant proportion of patients – 20% in the earlier period and 16% in the later – who did not receive surgery or radiation. "At least 16% of patients are still not getting the care that they need – care that could save their lives. We must identify the barriers to treatment so that every patient has hope for a cancer cure," he said in a statement.
For the entire study period, factors significantly associated with higher risk of death after primary RT or surgery included age, African American race, large cell or squamous histology, and being unmarried. Significant protective factors included female sex and race listed as "other."
Dr. Kapadia noted that RT is advantageous in that it is noninvasive and is done on an outpatient basis. Moreover, local control rates with radiotherapy among patients who are too sick to undergo surgery are now approaching those of surgery.
Ongoing "coin flip" studies are currently comparing outcomes of radiation versus surgery in patients who would otherwise be fit for surgery. "Those are going to be very exciting studies. ... But for right now I would say surgery is still the preferred modality, with a large body of evidence to support that statement," he said.
The symposium was sponsored by the American Society of Clinical Oncology, the American Society for Radiation Oncology, the International Association for the Study of Lung Cancer, and the University of Chicago.
Median overall survival increased significantly among patients with stage I non–small cell lung cancer over the last decade – in particular, those treated with radiation therapy alone, according to an analysis of the Surveillance, Epidemiology, and End Results database.
The median survival for all treatment groups increased by 27%, from 44 months during 1999-2003 to 56 months during 2004-2008. For those treated with radiation alone – who would likely be the sickest patients since they would not have been considered candidates for surgery – median overall survival improved by 31%, from 16 to 21 months. Both changes were statistically significant (log rank P less than .0001).
"Stage I NSCLC [non–small cell lung cancer] patients who receive radiation therapy alone are surviving longer than they used to," Dr. Nirav S. Kapadia said in a press briefing from the Chicago Multidisciplinary Symposium in Thoracic Oncology.
A change in the survival of patients treated with surgery could not be detected, as median survival has not yet been reached, he and his coauthors reported.
Until recently, surgery has been the primary treatment for stage I NSCLC. However, as recent advances in radiotherapy (RT) such as stereotactic body radiation therapy have allowed dose escalation with more precise tumor targeting, the use of RT has increased, and outcomes appear to have improved over time, said Dr. Kapadia, a chief resident in the department of radiation oncology at the University of Michigan, Ann Arbor
The National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) database encompasses about 25% of the U.S. population. This study compared SEER data on 27,469 patients with NSCLC treated during 1999-2003 with data from 26,195 patients treated during 2004-2008.
During 1999-2003, 64% of patients were treated with primary surgery, 14% received RT alone, 20% had neither treatment, and 2% had unknown treatment. In the later era, 70% of patients underwent primary surgery, 13% received primary RT, 16% had neither surgery nor RT, and 1% had unknown treatment.
The proportion receiving surgery alone increased from 60% to 67% during the two time periods. Thus, the rates of surgery increased from the earlier to the later period, but there was no significant difference in the number of patients who received radiotherapy, either as an adjunct to surgery or as definitive therapy, noted Dr. Kapadia.
He expressed concern about the significant proportion of patients – 20% in the earlier period and 16% in the later – who did not receive surgery or radiation. "At least 16% of patients are still not getting the care that they need – care that could save their lives. We must identify the barriers to treatment so that every patient has hope for a cancer cure," he said in a statement.
For the entire study period, factors significantly associated with higher risk of death after primary RT or surgery included age, African American race, large cell or squamous histology, and being unmarried. Significant protective factors included female sex and race listed as "other."
Dr. Kapadia noted that RT is advantageous in that it is noninvasive and is done on an outpatient basis. Moreover, local control rates with radiotherapy among patients who are too sick to undergo surgery are now approaching those of surgery.
Ongoing "coin flip" studies are currently comparing outcomes of radiation versus surgery in patients who would otherwise be fit for surgery. "Those are going to be very exciting studies. ... But for right now I would say surgery is still the preferred modality, with a large body of evidence to support that statement," he said.
The symposium was sponsored by the American Society of Clinical Oncology, the American Society for Radiation Oncology, the International Association for the Study of Lung Cancer, and the University of Chicago.
Median overall survival increased significantly among patients with stage I non–small cell lung cancer over the last decade – in particular, those treated with radiation therapy alone, according to an analysis of the Surveillance, Epidemiology, and End Results database.
The median survival for all treatment groups increased by 27%, from 44 months during 1999-2003 to 56 months during 2004-2008. For those treated with radiation alone – who would likely be the sickest patients since they would not have been considered candidates for surgery – median overall survival improved by 31%, from 16 to 21 months. Both changes were statistically significant (log rank P less than .0001).
"Stage I NSCLC [non–small cell lung cancer] patients who receive radiation therapy alone are surviving longer than they used to," Dr. Nirav S. Kapadia said in a press briefing from the Chicago Multidisciplinary Symposium in Thoracic Oncology.
A change in the survival of patients treated with surgery could not be detected, as median survival has not yet been reached, he and his coauthors reported.
Until recently, surgery has been the primary treatment for stage I NSCLC. However, as recent advances in radiotherapy (RT) such as stereotactic body radiation therapy have allowed dose escalation with more precise tumor targeting, the use of RT has increased, and outcomes appear to have improved over time, said Dr. Kapadia, a chief resident in the department of radiation oncology at the University of Michigan, Ann Arbor
The National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) database encompasses about 25% of the U.S. population. This study compared SEER data on 27,469 patients with NSCLC treated during 1999-2003 with data from 26,195 patients treated during 2004-2008.
During 1999-2003, 64% of patients were treated with primary surgery, 14% received RT alone, 20% had neither treatment, and 2% had unknown treatment. In the later era, 70% of patients underwent primary surgery, 13% received primary RT, 16% had neither surgery nor RT, and 1% had unknown treatment.
The proportion receiving surgery alone increased from 60% to 67% during the two time periods. Thus, the rates of surgery increased from the earlier to the later period, but there was no significant difference in the number of patients who received radiotherapy, either as an adjunct to surgery or as definitive therapy, noted Dr. Kapadia.
He expressed concern about the significant proportion of patients – 20% in the earlier period and 16% in the later – who did not receive surgery or radiation. "At least 16% of patients are still not getting the care that they need – care that could save their lives. We must identify the barriers to treatment so that every patient has hope for a cancer cure," he said in a statement.
For the entire study period, factors significantly associated with higher risk of death after primary RT or surgery included age, African American race, large cell or squamous histology, and being unmarried. Significant protective factors included female sex and race listed as "other."
Dr. Kapadia noted that RT is advantageous in that it is noninvasive and is done on an outpatient basis. Moreover, local control rates with radiotherapy among patients who are too sick to undergo surgery are now approaching those of surgery.
Ongoing "coin flip" studies are currently comparing outcomes of radiation versus surgery in patients who would otherwise be fit for surgery. "Those are going to be very exciting studies. ... But for right now I would say surgery is still the preferred modality, with a large body of evidence to support that statement," he said.
The symposium was sponsored by the American Society of Clinical Oncology, the American Society for Radiation Oncology, the International Association for the Study of Lung Cancer, and the University of Chicago.
FROM THE CHICAGO MULTIDISCIPLINARY SYMPOSIUM IN THORACIC ONCOLOGY
Major Finding: When stage I NSCLC was treated with radiation alone, median survival improved significantly, from 16 months during 1999-2003 to 21 months during 2004-2008.
Data Source: The findings come from an analysis of data from the National Cancer Institute’s Surveillance, Epidemiology, and End Results database.
Disclosures: Dr. Kapadia and his coauthors had no financial disclosures.
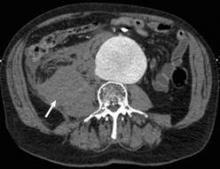
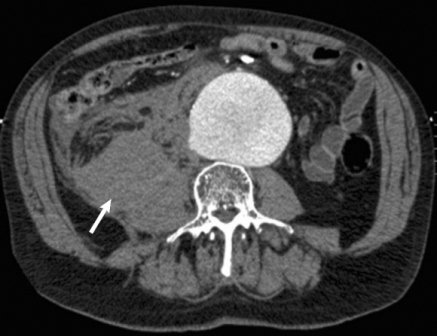
No Survival Benefit Seen for EVAR Over Open Surgery for Ruptured AAA
Endovascular repair of ruptured abdominal aortic aneurysms confers no acute or late mortality benefit over open surgery, according to the results of a retrospective study using propensity-score matching by researchers at the University of Pittsburgh Medical Center.
Given these results and the massive systemic change and resources needed to organize and sustain an endovascular program, widespread adoption and routine use of endovascular repair of ruptured AAA (REVAR) should be deferred until there is good evidence of its superiority, according to Dr. Jae-Sung Cho and his colleagues.
Currently, the majority of ruptured AAAs are repaired using open surgery, but there is increasing enthusiasm for REVAR based on results from case reports and single-center series that used a standard REVAR protocol, according to the researchers. Those results are suspect because of the problem of selection bias and limited follow-up, the investigators said.
In an attempt to clarify the situation, Dr. Cho and his colleagues reviewed 312 patients who underwent ruptured abdominal aortic aneurysm (rAAA) repair from January 2001 to November 2010 at the University of Pittsburgh Medical Center (UPMC), a regional tertiary referral hospital with a high volume of open and emergent aortic surgery cases, according to the report in the September issue of the Journal of Vascular Surgery (2012;56:614-19).
A total of 34 patients were excluded owing to prior open or endovascular repair or because they were symptomatic without actual rupture. The remaining 288 patients formed the basis of the analysis: 37 underwent REVAR and 251 were treated with open surgical repair (OSR).
The researchers used propensity score–based matching to reduce the confounding effects of covariate imbalance between the REVAR and OSR as well as potential selection bias. A 1:3 ratio was used in the matching because of the various sample sizes of the REVAR and OSR patients and their distributions of propensity scores, yielding 37 REVAR patients matched to 111 OSR patients for analysis.
The matched cohorts did not significantly differ according to sex (the majority were men: 70.3% of the REVAR group, 62.2% of the OSR group); age (around 75 years in both groups); or history of comorbidities, including chronic obstructive pulmonary disease, cerebrovascular accident, hypertension, peripheral vascular disease, diabetes, smoking, hemodialysis, and coronary artery disease.
The only significant demographic difference between the patient groups was presentation with hemodynamic instability, defined as a systolic blood pressure less than 80 mm Hg. Such patients were more likely to undergo OSR (44%, compared with 24% of patients who underwent REVAR).
The outcomes measured comprised operative mortality (defined as intraoperative, in-hospital, and 30-day mortality), postoperative morbidity, length of stay, and transfusion of blood products. Long-term mortality was determined by evaluating against the Social Security Death Index. Late survival was estimated by Kaplan-Meier methods.
Although operative time and blood replacement were higher with OSR, overall complication rates were not significantly different (54% with REVAR vs. 66% with OSR). However, tracheostomies (21% vs. 3%), myocardial infarction (38% vs. 18%), and acute tubular necrosis (47% vs. 21%) were all significantly higher with OSR.
"Operative mortality rates were similar," according to the authors: 22% with REVAR vs. 32% with OSR (odds ratio, 0.63 for REVAR; 95% confidence interval, 0.24-1.48), and the incidences of secondary interventions for aneurysm- or graft-related complications were identical at 22%.
Kaplan-Meier estimates of 1-, 2-, and 3-year survival rates were not significantly different at 50%, 50%, and 42%, respectively, for REVAR and 54%, 52%, and 47% for OSR (P = .66).
The investigators discussed these results within the scope of several prospective, randomized trials that have been conducted or are currently underway comparing REVAR with OSR. The Nottingham trial and the Amsterdam Acute Aneurysm (AJAX) trial also failed to demonstrate any survival benefits from REVAR. The ongoing IMPROVE trial should be large enough (600 randomized patients) and should be able to address the question of whether or not a REVAR first approach provides a survival benefit over OSR.
Widespread adoption of a REVAR first approach is attractive, according to the researchers, who detail the value of its minimally invasive nature and the lower rates of complications, as demonstrated in their own study.
"However, implementation of such a protocol involves massive systemic changes, such as stocking of endovascular stent grafts and auxiliaries, and around-the-clock availability of skilled endovascular, radiologic, and nursing teams. It is not pragmatic to undertake such systemic and systematic changes in the absence of clear evidence of REVAR’s superiority," they said. For these reasons, no such change was established at the UPMC system, which would represent "a monumental task in a variety of ways."
The limitations the authors pointed out were those inherent to a retrospective study with a relatively small sample size. In addition, there might have been a selection bias on the choice of treatment rendered. Propensity score–based matching can only remove overt bias; unlike randomization, it cannot remove hidden biases.
"There is still equipoise on what the best treatment for ruptured AAA is. The data [in the literature] are conflicting, and many studies showing improved outcomes with REVAR employed it preferentially for patients with favorable anatomy and hemodynamic stability. Preferential REVAR should be deferred until results of prospective, randomized trials are available and predictive factors for its success are identified. Some patients may be harmed by indiscriminate insistence on REVAR," Dr. Cho and his colleagues concluded.
The authors reported that they had no conflicts of interest with regard to their paper.

|
|
[The researchers’] experience indicates that, with expertise in both emergent endovascular aneurysm repair (EVAR) and OSR, the short-term mortality can be equally reduced in both groups, albeit the morbidity of ruptured EVAR is significantly lower. It would also appear that all 37 patients (100%) with ruptured EVAR and only 111 of 241 patients (46%) with ruptured OSR ... underwent propensity score–based analysis. ... I feel obliged to comment that their inability to include the remaining 54% of patients with ruptured OSR has resulted in a cumulative midterm Kaplan-Meier survival analysis that would strongly bias in favor of OSR.
Dr. Manish Mehta is a vascular surgeon with the Albany (N.Y.) Vascular Group. His remarks were part of an invited commentary (J. Vasc. Surg. 2012;56:620). Dr. Mehta did not disclose any potential conflicts of interest.

|
|
[The researchers’] experience indicates that, with expertise in both emergent endovascular aneurysm repair (EVAR) and OSR, the short-term mortality can be equally reduced in both groups, albeit the morbidity of ruptured EVAR is significantly lower. It would also appear that all 37 patients (100%) with ruptured EVAR and only 111 of 241 patients (46%) with ruptured OSR ... underwent propensity score–based analysis. ... I feel obliged to comment that their inability to include the remaining 54% of patients with ruptured OSR has resulted in a cumulative midterm Kaplan-Meier survival analysis that would strongly bias in favor of OSR.
Dr. Manish Mehta is a vascular surgeon with the Albany (N.Y.) Vascular Group. His remarks were part of an invited commentary (J. Vasc. Surg. 2012;56:620). Dr. Mehta did not disclose any potential conflicts of interest.

|
|
[The researchers’] experience indicates that, with expertise in both emergent endovascular aneurysm repair (EVAR) and OSR, the short-term mortality can be equally reduced in both groups, albeit the morbidity of ruptured EVAR is significantly lower. It would also appear that all 37 patients (100%) with ruptured EVAR and only 111 of 241 patients (46%) with ruptured OSR ... underwent propensity score–based analysis. ... I feel obliged to comment that their inability to include the remaining 54% of patients with ruptured OSR has resulted in a cumulative midterm Kaplan-Meier survival analysis that would strongly bias in favor of OSR.
Dr. Manish Mehta is a vascular surgeon with the Albany (N.Y.) Vascular Group. His remarks were part of an invited commentary (J. Vasc. Surg. 2012;56:620). Dr. Mehta did not disclose any potential conflicts of interest.
Endovascular repair of ruptured abdominal aortic aneurysms confers no acute or late mortality benefit over open surgery, according to the results of a retrospective study using propensity-score matching by researchers at the University of Pittsburgh Medical Center.
Given these results and the massive systemic change and resources needed to organize and sustain an endovascular program, widespread adoption and routine use of endovascular repair of ruptured AAA (REVAR) should be deferred until there is good evidence of its superiority, according to Dr. Jae-Sung Cho and his colleagues.
Currently, the majority of ruptured AAAs are repaired using open surgery, but there is increasing enthusiasm for REVAR based on results from case reports and single-center series that used a standard REVAR protocol, according to the researchers. Those results are suspect because of the problem of selection bias and limited follow-up, the investigators said.
In an attempt to clarify the situation, Dr. Cho and his colleagues reviewed 312 patients who underwent ruptured abdominal aortic aneurysm (rAAA) repair from January 2001 to November 2010 at the University of Pittsburgh Medical Center (UPMC), a regional tertiary referral hospital with a high volume of open and emergent aortic surgery cases, according to the report in the September issue of the Journal of Vascular Surgery (2012;56:614-19).
A total of 34 patients were excluded owing to prior open or endovascular repair or because they were symptomatic without actual rupture. The remaining 288 patients formed the basis of the analysis: 37 underwent REVAR and 251 were treated with open surgical repair (OSR).
The researchers used propensity score–based matching to reduce the confounding effects of covariate imbalance between the REVAR and OSR as well as potential selection bias. A 1:3 ratio was used in the matching because of the various sample sizes of the REVAR and OSR patients and their distributions of propensity scores, yielding 37 REVAR patients matched to 111 OSR patients for analysis.
The matched cohorts did not significantly differ according to sex (the majority were men: 70.3% of the REVAR group, 62.2% of the OSR group); age (around 75 years in both groups); or history of comorbidities, including chronic obstructive pulmonary disease, cerebrovascular accident, hypertension, peripheral vascular disease, diabetes, smoking, hemodialysis, and coronary artery disease.
The only significant demographic difference between the patient groups was presentation with hemodynamic instability, defined as a systolic blood pressure less than 80 mm Hg. Such patients were more likely to undergo OSR (44%, compared with 24% of patients who underwent REVAR).
The outcomes measured comprised operative mortality (defined as intraoperative, in-hospital, and 30-day mortality), postoperative morbidity, length of stay, and transfusion of blood products. Long-term mortality was determined by evaluating against the Social Security Death Index. Late survival was estimated by Kaplan-Meier methods.
Although operative time and blood replacement were higher with OSR, overall complication rates were not significantly different (54% with REVAR vs. 66% with OSR). However, tracheostomies (21% vs. 3%), myocardial infarction (38% vs. 18%), and acute tubular necrosis (47% vs. 21%) were all significantly higher with OSR.
"Operative mortality rates were similar," according to the authors: 22% with REVAR vs. 32% with OSR (odds ratio, 0.63 for REVAR; 95% confidence interval, 0.24-1.48), and the incidences of secondary interventions for aneurysm- or graft-related complications were identical at 22%.
Kaplan-Meier estimates of 1-, 2-, and 3-year survival rates were not significantly different at 50%, 50%, and 42%, respectively, for REVAR and 54%, 52%, and 47% for OSR (P = .66).
The investigators discussed these results within the scope of several prospective, randomized trials that have been conducted or are currently underway comparing REVAR with OSR. The Nottingham trial and the Amsterdam Acute Aneurysm (AJAX) trial also failed to demonstrate any survival benefits from REVAR. The ongoing IMPROVE trial should be large enough (600 randomized patients) and should be able to address the question of whether or not a REVAR first approach provides a survival benefit over OSR.
Widespread adoption of a REVAR first approach is attractive, according to the researchers, who detail the value of its minimally invasive nature and the lower rates of complications, as demonstrated in their own study.
"However, implementation of such a protocol involves massive systemic changes, such as stocking of endovascular stent grafts and auxiliaries, and around-the-clock availability of skilled endovascular, radiologic, and nursing teams. It is not pragmatic to undertake such systemic and systematic changes in the absence of clear evidence of REVAR’s superiority," they said. For these reasons, no such change was established at the UPMC system, which would represent "a monumental task in a variety of ways."
The limitations the authors pointed out were those inherent to a retrospective study with a relatively small sample size. In addition, there might have been a selection bias on the choice of treatment rendered. Propensity score–based matching can only remove overt bias; unlike randomization, it cannot remove hidden biases.
"There is still equipoise on what the best treatment for ruptured AAA is. The data [in the literature] are conflicting, and many studies showing improved outcomes with REVAR employed it preferentially for patients with favorable anatomy and hemodynamic stability. Preferential REVAR should be deferred until results of prospective, randomized trials are available and predictive factors for its success are identified. Some patients may be harmed by indiscriminate insistence on REVAR," Dr. Cho and his colleagues concluded.
The authors reported that they had no conflicts of interest with regard to their paper.
Endovascular repair of ruptured abdominal aortic aneurysms confers no acute or late mortality benefit over open surgery, according to the results of a retrospective study using propensity-score matching by researchers at the University of Pittsburgh Medical Center.
Given these results and the massive systemic change and resources needed to organize and sustain an endovascular program, widespread adoption and routine use of endovascular repair of ruptured AAA (REVAR) should be deferred until there is good evidence of its superiority, according to Dr. Jae-Sung Cho and his colleagues.
Currently, the majority of ruptured AAAs are repaired using open surgery, but there is increasing enthusiasm for REVAR based on results from case reports and single-center series that used a standard REVAR protocol, according to the researchers. Those results are suspect because of the problem of selection bias and limited follow-up, the investigators said.
In an attempt to clarify the situation, Dr. Cho and his colleagues reviewed 312 patients who underwent ruptured abdominal aortic aneurysm (rAAA) repair from January 2001 to November 2010 at the University of Pittsburgh Medical Center (UPMC), a regional tertiary referral hospital with a high volume of open and emergent aortic surgery cases, according to the report in the September issue of the Journal of Vascular Surgery (2012;56:614-19).
A total of 34 patients were excluded owing to prior open or endovascular repair or because they were symptomatic without actual rupture. The remaining 288 patients formed the basis of the analysis: 37 underwent REVAR and 251 were treated with open surgical repair (OSR).
The researchers used propensity score–based matching to reduce the confounding effects of covariate imbalance between the REVAR and OSR as well as potential selection bias. A 1:3 ratio was used in the matching because of the various sample sizes of the REVAR and OSR patients and their distributions of propensity scores, yielding 37 REVAR patients matched to 111 OSR patients for analysis.
The matched cohorts did not significantly differ according to sex (the majority were men: 70.3% of the REVAR group, 62.2% of the OSR group); age (around 75 years in both groups); or history of comorbidities, including chronic obstructive pulmonary disease, cerebrovascular accident, hypertension, peripheral vascular disease, diabetes, smoking, hemodialysis, and coronary artery disease.
The only significant demographic difference between the patient groups was presentation with hemodynamic instability, defined as a systolic blood pressure less than 80 mm Hg. Such patients were more likely to undergo OSR (44%, compared with 24% of patients who underwent REVAR).
The outcomes measured comprised operative mortality (defined as intraoperative, in-hospital, and 30-day mortality), postoperative morbidity, length of stay, and transfusion of blood products. Long-term mortality was determined by evaluating against the Social Security Death Index. Late survival was estimated by Kaplan-Meier methods.
Although operative time and blood replacement were higher with OSR, overall complication rates were not significantly different (54% with REVAR vs. 66% with OSR). However, tracheostomies (21% vs. 3%), myocardial infarction (38% vs. 18%), and acute tubular necrosis (47% vs. 21%) were all significantly higher with OSR.
"Operative mortality rates were similar," according to the authors: 22% with REVAR vs. 32% with OSR (odds ratio, 0.63 for REVAR; 95% confidence interval, 0.24-1.48), and the incidences of secondary interventions for aneurysm- or graft-related complications were identical at 22%.
Kaplan-Meier estimates of 1-, 2-, and 3-year survival rates were not significantly different at 50%, 50%, and 42%, respectively, for REVAR and 54%, 52%, and 47% for OSR (P = .66).
The investigators discussed these results within the scope of several prospective, randomized trials that have been conducted or are currently underway comparing REVAR with OSR. The Nottingham trial and the Amsterdam Acute Aneurysm (AJAX) trial also failed to demonstrate any survival benefits from REVAR. The ongoing IMPROVE trial should be large enough (600 randomized patients) and should be able to address the question of whether or not a REVAR first approach provides a survival benefit over OSR.
Widespread adoption of a REVAR first approach is attractive, according to the researchers, who detail the value of its minimally invasive nature and the lower rates of complications, as demonstrated in their own study.
"However, implementation of such a protocol involves massive systemic changes, such as stocking of endovascular stent grafts and auxiliaries, and around-the-clock availability of skilled endovascular, radiologic, and nursing teams. It is not pragmatic to undertake such systemic and systematic changes in the absence of clear evidence of REVAR’s superiority," they said. For these reasons, no such change was established at the UPMC system, which would represent "a monumental task in a variety of ways."
The limitations the authors pointed out were those inherent to a retrospective study with a relatively small sample size. In addition, there might have been a selection bias on the choice of treatment rendered. Propensity score–based matching can only remove overt bias; unlike randomization, it cannot remove hidden biases.
"There is still equipoise on what the best treatment for ruptured AAA is. The data [in the literature] are conflicting, and many studies showing improved outcomes with REVAR employed it preferentially for patients with favorable anatomy and hemodynamic stability. Preferential REVAR should be deferred until results of prospective, randomized trials are available and predictive factors for its success are identified. Some patients may be harmed by indiscriminate insistence on REVAR," Dr. Cho and his colleagues concluded.
The authors reported that they had no conflicts of interest with regard to their paper.
FROM THE JOURNAL OF VASCULAR SURGERY
Major Finding: Kaplan-Meier estimates of 1-, 2-, and 3-year survival rates were not significantly different, at 50%, 50%, and 42%, respectively, for REVAR and 54%, 52%, and 47% for open repair.
Data Source: Outcomes were reviewed for patients in a single center, retrospective, propensity score–matched study comprising 37 patients who underwent REVAR and 251 treated with open surgical repair.
Disclosures: The authors reported that they had no conflicts of interest with regard to their paper.
A-Fib Ablation Shown Safe During Cardiac Surgery
MUNICH – Left atrial ablative treatment for atrial fibrillation was safe when administered during open heart surgery in a multicenter, randomized trial of 224 patients who all had atrial fibrillation and required heart surgery for another reason.
The treatment was also effective, resulting in a significantly higher rate of sinus rhythm among the patients treated with ablation compared with the control patients who underwent open-heart surgery alone, Dr. Petr Widimsky reported at the annual congress of the European Society of Cardiology.
Some experts who heard the findings expressed some skepticism about the pattern of anti-arrhythmic effects and other atrial fibrillation–related outcomes during follow-up.
"The rhythm outcomes had some surprises. All the benefit was in patients with long-standing, persistent atrial fibrillation; that’s very surprising because it contrasts with previous findings from both catheter ablation and surgical ablation," said Dr. Gerhard Hindricks, a professor of medicine and director of the department of electrophysiology at the Heart Center of Leipzig (Germany) University.
"The secondary outcomes were also a surprise, because the atrial fibrillation response did not translate into a change in treatment" in the use of anti-arrhythmic drugs, and the two groups showed no differences in the 1-year rates of stroke, major bleeding, and all-cause mortality despite the reported different rates of continued AF. In an editorial that accompanied the published paper, Dr. Hindricks cited the inadequacy of assessing atrial arrhythmia at 1-year follow-up with 24-hour Holter ECG monitoring. "This follow-up regimen is by far not enough to generate reliable and solid results," Dr. Hindricks and his coauthor wrote (Eur. Heart J. 2012;33 [doi: 10.1093/eurheartj/ehs294]).
Despite these unexpected findings, "surgical ablation of atrial fibrillation should be cautiously indicated in asymptomatic patients scheduled for cardiac surgery," Dr. Hindricks said in comments he made at the meeting.
"This was the largest" of several randomized, prospective studies that have assessed ablation of atrial fibrillation during cardiac surgery, said Dr. Riccardo Cappato, director of the Center of Clinical Arrhythmia and Electrophysiology at the Policlinico San Donato in Milan. "The increased probability of maintaining sinus rhythm in patients with permanent atrial fibrillation does not seem to have been at the expense of higher perioperative risk exposure."
The PRAGUE-12 study enrolled 224 patients with AF who required cardiac surgery for coronary artery bypass, valve repair or replacement, or both at three centers in the Czech Republic and Slovakia during 2007-2011. The investigators randomized 117 patients to undergo atrial ablation during their surgery, and 107 who received no ablation with their surgery and served as controls. The surgeons could use whichever energy source for ablation they preferred, but 97% used cryoablation. The ablation-lesion set was the same for all patients in that treatment arm.
The patients averaged about 70 years old, and about 58% were men. Roughly half the patients had long-standing persistent AF, about a quarter had persistent AF, and the remaining quarter had paroxysmal AF.
All patients underwent open surgery using a median sternotomy with cardiopulmonary bypass and cardioplegic heart arrest. Adding ablation to the procedures increased the total surgical time by an average of 20 minutes, and increased the period of cardiopulmonary bypass and the cross-clamp time by an average of 28 minutes.
The primary safety end point was the combined rate of death, stroke, myocardial ischemia, or renal failure requiring dialysis at 30 days after surgery; this occurred in 10% of the patients who had ablation and 15% of the control patients, a nonsignificant difference, reported Dr. Widimsky, professor and head of the Cardiocenter of Charles University in Prague. Each component of the combined adverse event was similar between the two study arms. At 1 year after surgery, the combined adverse event rate was 41% in the patients treated with AF ablation and 40% in the controls.
The study’s primary efficacy end point was the prevalence of sinus rhythm in patients as measured by 24-hour Holter ECG monitoring, which occurred in 60% of the patients who underwent ablation and in 36% of the controls, a significant difference. This difference was driven by the outcome difference among the subgroup of patients who entered the study with long-standing persistent AF. In this subgroup, the proportion of patients in sinus rhythm when assessed after 1 year was 53% in the ablated group and 14% in the controls, a significant difference. In contrast, the prevalence of sinus rhythm at 1 year did not differ significantly between the two treatment arms in patients who entered the study with paroxysmal AF, or in those who entered with persistent AF.
Concurrent with the presentation of PRAGUE-12, the results also appeared in an article published online (Eur. Heart J. 2012;33 [doi: 10.1093/eurheartj/ehs290]).
Dr. Widimsky and his associates said that they had no disclosures.
When Dr. James L. Cox introduced the Cox-Maze III procedure for the surgical disruption of atrial fibrillation, the technique involved prolonged cutting and sewing, and the added time on cardiopulmonary bypass and cross-clamping this produced led to a lot of morbidity, including neurologic complications and renal failure.
The current study used a modified Maze procedure that primarily used cryoablation. Because no cutting and sewing were involved, the surgery was much easier, and it produced fewer complications. Surgeons are interested in an atrial fibrillation procedure that is safe and increases the likelihood of leading to sinus rhythm.

|
Mitchel L. Zoler/IMNG Medical Media
|
This study is important because it shows that this method can be used safely and it improves the end point of resting sinus rhythm. It is paradoxical that the main rhythm benefit was in patients with long-standing persistent atrial fibrillation, because usually that is harder to treat. But this may have been a methodological issue, because they measured the 1-year rhythm outcome without doing prolonged, continuous ECG monitoring. It’s hard to define success in patients with paroxysmal atrial fibrillation unless you monitor patients continuously for a week.
The main message from this study was that the ablation procedure was safe during cardiac surgery and did not add much time or complexity to the surgery. I think these results will increase the use of ablation during cardiac surgery for other reasons. It is a major contribution.
Miguel Sousa Uva, M.D., a cardiac surgeon at Hospital da Cruz Vermelha in Lisbon, made these comments in an interview. He said that he had no disclosures.
When Dr. James L. Cox introduced the Cox-Maze III procedure for the surgical disruption of atrial fibrillation, the technique involved prolonged cutting and sewing, and the added time on cardiopulmonary bypass and cross-clamping this produced led to a lot of morbidity, including neurologic complications and renal failure.
The current study used a modified Maze procedure that primarily used cryoablation. Because no cutting and sewing were involved, the surgery was much easier, and it produced fewer complications. Surgeons are interested in an atrial fibrillation procedure that is safe and increases the likelihood of leading to sinus rhythm.

|
Mitchel L. Zoler/IMNG Medical Media
|
This study is important because it shows that this method can be used safely and it improves the end point of resting sinus rhythm. It is paradoxical that the main rhythm benefit was in patients with long-standing persistent atrial fibrillation, because usually that is harder to treat. But this may have been a methodological issue, because they measured the 1-year rhythm outcome without doing prolonged, continuous ECG monitoring. It’s hard to define success in patients with paroxysmal atrial fibrillation unless you monitor patients continuously for a week.
The main message from this study was that the ablation procedure was safe during cardiac surgery and did not add much time or complexity to the surgery. I think these results will increase the use of ablation during cardiac surgery for other reasons. It is a major contribution.
Miguel Sousa Uva, M.D., a cardiac surgeon at Hospital da Cruz Vermelha in Lisbon, made these comments in an interview. He said that he had no disclosures.
When Dr. James L. Cox introduced the Cox-Maze III procedure for the surgical disruption of atrial fibrillation, the technique involved prolonged cutting and sewing, and the added time on cardiopulmonary bypass and cross-clamping this produced led to a lot of morbidity, including neurologic complications and renal failure.
The current study used a modified Maze procedure that primarily used cryoablation. Because no cutting and sewing were involved, the surgery was much easier, and it produced fewer complications. Surgeons are interested in an atrial fibrillation procedure that is safe and increases the likelihood of leading to sinus rhythm.

|
Mitchel L. Zoler/IMNG Medical Media
|
This study is important because it shows that this method can be used safely and it improves the end point of resting sinus rhythm. It is paradoxical that the main rhythm benefit was in patients with long-standing persistent atrial fibrillation, because usually that is harder to treat. But this may have been a methodological issue, because they measured the 1-year rhythm outcome without doing prolonged, continuous ECG monitoring. It’s hard to define success in patients with paroxysmal atrial fibrillation unless you monitor patients continuously for a week.
The main message from this study was that the ablation procedure was safe during cardiac surgery and did not add much time or complexity to the surgery. I think these results will increase the use of ablation during cardiac surgery for other reasons. It is a major contribution.
Miguel Sousa Uva, M.D., a cardiac surgeon at Hospital da Cruz Vermelha in Lisbon, made these comments in an interview. He said that he had no disclosures.
MUNICH – Left atrial ablative treatment for atrial fibrillation was safe when administered during open heart surgery in a multicenter, randomized trial of 224 patients who all had atrial fibrillation and required heart surgery for another reason.
The treatment was also effective, resulting in a significantly higher rate of sinus rhythm among the patients treated with ablation compared with the control patients who underwent open-heart surgery alone, Dr. Petr Widimsky reported at the annual congress of the European Society of Cardiology.
Some experts who heard the findings expressed some skepticism about the pattern of anti-arrhythmic effects and other atrial fibrillation–related outcomes during follow-up.
"The rhythm outcomes had some surprises. All the benefit was in patients with long-standing, persistent atrial fibrillation; that’s very surprising because it contrasts with previous findings from both catheter ablation and surgical ablation," said Dr. Gerhard Hindricks, a professor of medicine and director of the department of electrophysiology at the Heart Center of Leipzig (Germany) University.
"The secondary outcomes were also a surprise, because the atrial fibrillation response did not translate into a change in treatment" in the use of anti-arrhythmic drugs, and the two groups showed no differences in the 1-year rates of stroke, major bleeding, and all-cause mortality despite the reported different rates of continued AF. In an editorial that accompanied the published paper, Dr. Hindricks cited the inadequacy of assessing atrial arrhythmia at 1-year follow-up with 24-hour Holter ECG monitoring. "This follow-up regimen is by far not enough to generate reliable and solid results," Dr. Hindricks and his coauthor wrote (Eur. Heart J. 2012;33 [doi: 10.1093/eurheartj/ehs294]).
Despite these unexpected findings, "surgical ablation of atrial fibrillation should be cautiously indicated in asymptomatic patients scheduled for cardiac surgery," Dr. Hindricks said in comments he made at the meeting.
"This was the largest" of several randomized, prospective studies that have assessed ablation of atrial fibrillation during cardiac surgery, said Dr. Riccardo Cappato, director of the Center of Clinical Arrhythmia and Electrophysiology at the Policlinico San Donato in Milan. "The increased probability of maintaining sinus rhythm in patients with permanent atrial fibrillation does not seem to have been at the expense of higher perioperative risk exposure."
The PRAGUE-12 study enrolled 224 patients with AF who required cardiac surgery for coronary artery bypass, valve repair or replacement, or both at three centers in the Czech Republic and Slovakia during 2007-2011. The investigators randomized 117 patients to undergo atrial ablation during their surgery, and 107 who received no ablation with their surgery and served as controls. The surgeons could use whichever energy source for ablation they preferred, but 97% used cryoablation. The ablation-lesion set was the same for all patients in that treatment arm.
The patients averaged about 70 years old, and about 58% were men. Roughly half the patients had long-standing persistent AF, about a quarter had persistent AF, and the remaining quarter had paroxysmal AF.
All patients underwent open surgery using a median sternotomy with cardiopulmonary bypass and cardioplegic heart arrest. Adding ablation to the procedures increased the total surgical time by an average of 20 minutes, and increased the period of cardiopulmonary bypass and the cross-clamp time by an average of 28 minutes.
The primary safety end point was the combined rate of death, stroke, myocardial ischemia, or renal failure requiring dialysis at 30 days after surgery; this occurred in 10% of the patients who had ablation and 15% of the control patients, a nonsignificant difference, reported Dr. Widimsky, professor and head of the Cardiocenter of Charles University in Prague. Each component of the combined adverse event was similar between the two study arms. At 1 year after surgery, the combined adverse event rate was 41% in the patients treated with AF ablation and 40% in the controls.
The study’s primary efficacy end point was the prevalence of sinus rhythm in patients as measured by 24-hour Holter ECG monitoring, which occurred in 60% of the patients who underwent ablation and in 36% of the controls, a significant difference. This difference was driven by the outcome difference among the subgroup of patients who entered the study with long-standing persistent AF. In this subgroup, the proportion of patients in sinus rhythm when assessed after 1 year was 53% in the ablated group and 14% in the controls, a significant difference. In contrast, the prevalence of sinus rhythm at 1 year did not differ significantly between the two treatment arms in patients who entered the study with paroxysmal AF, or in those who entered with persistent AF.
Concurrent with the presentation of PRAGUE-12, the results also appeared in an article published online (Eur. Heart J. 2012;33 [doi: 10.1093/eurheartj/ehs290]).
Dr. Widimsky and his associates said that they had no disclosures.
MUNICH – Left atrial ablative treatment for atrial fibrillation was safe when administered during open heart surgery in a multicenter, randomized trial of 224 patients who all had atrial fibrillation and required heart surgery for another reason.
The treatment was also effective, resulting in a significantly higher rate of sinus rhythm among the patients treated with ablation compared with the control patients who underwent open-heart surgery alone, Dr. Petr Widimsky reported at the annual congress of the European Society of Cardiology.
Some experts who heard the findings expressed some skepticism about the pattern of anti-arrhythmic effects and other atrial fibrillation–related outcomes during follow-up.
"The rhythm outcomes had some surprises. All the benefit was in patients with long-standing, persistent atrial fibrillation; that’s very surprising because it contrasts with previous findings from both catheter ablation and surgical ablation," said Dr. Gerhard Hindricks, a professor of medicine and director of the department of electrophysiology at the Heart Center of Leipzig (Germany) University.
"The secondary outcomes were also a surprise, because the atrial fibrillation response did not translate into a change in treatment" in the use of anti-arrhythmic drugs, and the two groups showed no differences in the 1-year rates of stroke, major bleeding, and all-cause mortality despite the reported different rates of continued AF. In an editorial that accompanied the published paper, Dr. Hindricks cited the inadequacy of assessing atrial arrhythmia at 1-year follow-up with 24-hour Holter ECG monitoring. "This follow-up regimen is by far not enough to generate reliable and solid results," Dr. Hindricks and his coauthor wrote (Eur. Heart J. 2012;33 [doi: 10.1093/eurheartj/ehs294]).
Despite these unexpected findings, "surgical ablation of atrial fibrillation should be cautiously indicated in asymptomatic patients scheduled for cardiac surgery," Dr. Hindricks said in comments he made at the meeting.
"This was the largest" of several randomized, prospective studies that have assessed ablation of atrial fibrillation during cardiac surgery, said Dr. Riccardo Cappato, director of the Center of Clinical Arrhythmia and Electrophysiology at the Policlinico San Donato in Milan. "The increased probability of maintaining sinus rhythm in patients with permanent atrial fibrillation does not seem to have been at the expense of higher perioperative risk exposure."
The PRAGUE-12 study enrolled 224 patients with AF who required cardiac surgery for coronary artery bypass, valve repair or replacement, or both at three centers in the Czech Republic and Slovakia during 2007-2011. The investigators randomized 117 patients to undergo atrial ablation during their surgery, and 107 who received no ablation with their surgery and served as controls. The surgeons could use whichever energy source for ablation they preferred, but 97% used cryoablation. The ablation-lesion set was the same for all patients in that treatment arm.
The patients averaged about 70 years old, and about 58% were men. Roughly half the patients had long-standing persistent AF, about a quarter had persistent AF, and the remaining quarter had paroxysmal AF.
All patients underwent open surgery using a median sternotomy with cardiopulmonary bypass and cardioplegic heart arrest. Adding ablation to the procedures increased the total surgical time by an average of 20 minutes, and increased the period of cardiopulmonary bypass and the cross-clamp time by an average of 28 minutes.
The primary safety end point was the combined rate of death, stroke, myocardial ischemia, or renal failure requiring dialysis at 30 days after surgery; this occurred in 10% of the patients who had ablation and 15% of the control patients, a nonsignificant difference, reported Dr. Widimsky, professor and head of the Cardiocenter of Charles University in Prague. Each component of the combined adverse event was similar between the two study arms. At 1 year after surgery, the combined adverse event rate was 41% in the patients treated with AF ablation and 40% in the controls.
The study’s primary efficacy end point was the prevalence of sinus rhythm in patients as measured by 24-hour Holter ECG monitoring, which occurred in 60% of the patients who underwent ablation and in 36% of the controls, a significant difference. This difference was driven by the outcome difference among the subgroup of patients who entered the study with long-standing persistent AF. In this subgroup, the proportion of patients in sinus rhythm when assessed after 1 year was 53% in the ablated group and 14% in the controls, a significant difference. In contrast, the prevalence of sinus rhythm at 1 year did not differ significantly between the two treatment arms in patients who entered the study with paroxysmal AF, or in those who entered with persistent AF.
Concurrent with the presentation of PRAGUE-12, the results also appeared in an article published online (Eur. Heart J. 2012;33 [doi: 10.1093/eurheartj/ehs290]).
Dr. Widimsky and his associates said that they had no disclosures.
AT THE ANNUAL CONGRESS OF THE EUROPEAN SOCIETY OF CARDIOLOGY
Major Finding: After 30 days, the adverse event rate was 10% in cardiac surgery patients also undergoing ablation and 15% in patients without ablation, a difference that was not significant.
Data Source: PRAGUE-12 was a randomized trial that enrolled 224 patients with atrial fibrillation who also required cardiac surgery for another reason at three centers in the Czech Republic and Slovakia.
Disclosures: Dr. Widimsky and his associates said that they had no disclosures.
FAME 2 Favorable PCI Results Driven by Revascularization
The use of fractional flow reserve to guide percutaneous coronary intervention, along with best medical management, sharply reduced the need for urgent revascularization in patients with stable coronary artery disease and at least one physiologically significant lesion.
However, FFR-guided percutaneous coronary intervention (PCI) had little effect on deaths or myocardial infarctions, when compared with best medical management alone, according to the results of the FAME 2 (Fractional Flow Reserve vs. Angiography for Multivessel Evaluation 2) trial, which was conducted at 28 sites in Europe and North America and halted early.
The percentage of patients who had an MI, death, or urgent revascularization (the combined primary end point) was significantly lower in the PCI group than in the medical therapy group (4.3% vs.12.7%; hazard ratio with PCI, 0.32). This difference was driven by a 13-fold increase in the need for urgent revascularization in the medical-therapy group. Notably, the rate of death from any cause and the rate of MI did not differ significantly between the PCI group and the medical therapy group.
Importantly, patient recruitment was stopped on Jan. 15, 2012, at the recommendation of an independent data and safety monitoring board because of the highly significant difference in the incidence rates of the primary end point between the PCI and medical-therapy groups.
The results of the study were released in the New England Journal of Medicine on Aug. 28 to coincide with the presentation of the study at the annual congress of the European Society of Cardiology (N. Engl. J. Med. 2012 Aug. 28 [doi:10.1056/NEJMoa1205361]).
A total of 1,220 patients out a planned 1,832 were enrolled. Of those, 888 patients had at least one stenosis with an FFR of 0.80 or less: 447 patients were randomly assigned to FFR-guided PCI plus the best available medical therapy, and 441 patients to the best available medical therapy alone. The 332 patients with angiographically significant stenoses, but none with an FFR of 0.80 or less were enrolled in the registry and received the best available medical therapy alone. The mean duration of follow-up was 212 days.
Patients in stable condition who were appropriate candidates for PCI and who had angiographically assessed one-, two-, or three-vessel coronary artery disease suitable for PCI were included in the trial.
All patients were prescribed aspirin at a dose of 80-325 mg daily, metoprolol at a dose of 50 mg-200 mg daily (or any other beta-blocker), lisinopril (at least 5 mg daily, or another ACE inhibitor or an angiotensin receptor blocker)and atorvastatin (20-80 mg daily, or another statin).
All PCI patients were treated with second-generation drug-eluting stents.
Among the 56 patients who underwent urgent revascularization, the procedure was triggered by a MI in 12 patients (21%), by unstable angina accompanied by evidence of ischemia on ECG in 15 patients (27%), and by unstable angina diagnosed on the basis of clinical features in 29 patients (52%).
Patients in the PCI group were 86% less likely to undergo any revascularization and 83% less likely to undergo or nonurgent revascularization than were those in the medical therapy group.
The researchers identified several factors that may explain the differences between results in the present study and those in previous trials involving patients with stable coronary disease. "First, in previous trials in which various revascularization methods were compared with the best available medical therapy, patient enrollment was based primarily on angiographic findings, with or without noninvasive documentation of ischemia. It is likely that a sizable proportion of the patients had only limited ischemia," wrote lead investigator Dr. Bernard B. De Bruyne and his coinvestigators. Dr. De Bruyne is the codirector of the Cardiovascular Center at OLV Hospital in Aalst, Belgium.
"Even in the COURAGE (Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation) trial, in which noninvasive testing was performed in 85% of the patients, less than one-third of the patients had more than 10% ischemia on myocardial perfusion imaging. In daily clinical practice, less than half of patients undergo noninvasive stress testing before elective PCI. In the current trial, all the patients who underwent randomization had at least one functionally significant stenosis," they observed (N. Engl. J. Med. 2007;356:1503-16).
Second, PCI was performed only in lesions with an FFR of 0.80 or less. "This FFR-guided approach is associated with a better clinical outcome than that with PCI performed on the basis of angiographic results alone. These features probably explain the similarity of event rates between patients who were treated with PCI plus the best available medical therapy and patients with equivalent baseline characteristics but no functionally significant lesions who were enrolled in the registry and treated with the best available medical therapy alone," according to the investigators.
Third, second-generation drug-eluting stents were used in PCIs. This strategy is associated with a low number of repeat revascularizations. Finally, the primary end point included urgent revascularization, a component that was not included in the primary end point of previous trials.
The study was sponsored by St. Jude Medical, which makes the two pressure wires used in the trial. The company was involved in the collection and source verification of the data but not in the conduct of the trial. Dr. De Bruyne reported receiving consulting/honorarium and travel fees from St. Jude Medical. Most of the investigators had significant financial relationships with St. Jude Medical, as well as with other device and pharmaceutical companies. One author is an employee of St. Jude Medical.
The recommendation to terminate FAME 2 by the independent data and safety monitoring board at 7 months’ follow-up was based solely on one highly significant treatment difference. That difference was in the end point of urgent revascularization, which was performed in 49 patients in the group that received the best available medical therapy alone vs. 7 patients in the group that underwent percutaneous coronary intervention and also received the best available medical therapy, Dr. William E. Boden wrote in an editorial accompanying the FAME 2 report (N. Engl. J. Med. 2012 Aug. 28 [doi:10.1056/NEJMe1208620]).
"There were very few ‘hard’ events overall, with only four deaths (three in the medical-therapy group and one in the PCI group) and 29 myocardial infarctions (14 in the medical-therapy group and 15 in the PCI group). Of note, the definition of urgent revascularization was largely a clinical one and did not require evidence of ischemia or positive cardiac biomarkers in all patients; 29 of the 56 unplanned revascularizations (52%) were classified solely on the basis of clinical features, whereas in an exploratory subgroup analysis of the remaining 27 patients, there were fewer revascularizations triggered by a myocardial infarction or electrocardiographic evidence of ischemia in the PCI group than in the medical-therapy group," wrote Dr. Boden.
"Clearly, FFR [fractional flow reserve] holds potential promise for a more targeted approach to PCI that might be more clinically effective and cost effective than visually directed PCI for all angiographically significant stenoses," he continued. "Unfortunately, the early termination of the FAME 2 trial before full enrollment and follow-up were achieved, the neutral effects on the rate of death or myocardial infarction, and the lack of a significant, sustained treatment effect on the reduction of angina beyond 6 months leave more questions than answers."
The FAME 2 and COURAGE [Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation] trials are remarkably similar and showed that PCI reduced only the need for revascularization; in the COURAGE trial, there was a significant 40% reduction, whereas "neither the FAME 2 trial (with a mean 7 months of follow-up) nor the COURAGE trial (with a mean 55 months of follow-up) showed a benefit from PCI with respect to a reduction in the rate of death or myocardial infarction. The FAME 2 trial sought to establish the scientific basis for an FFR-guided PCI strategy for all functionally significant stenoses, but the results make this prospect somewhat unappealing," he wrote. Dr. Boden was the lead author of the COURAGE trial.
Current practice guidelines advocate the selective use of FFR to guide PCI decision making regarding borderline visual lesions (approximately 50%-70% stenosis). "It seems likely that the more routine use of FFR for all angiographically-significant stenoses would add considerable time, cost, and complexity to each PCI procedure and might also increase the risk of catheter-related complications such as coronary dissection and perforation," Dr. Boden pointed out.
Some of the uncertainty arising from FAME 2 may be resolved with the results of the ongoing ISCHEMIA (International Study of Comparative Health Effectiveness with Medical and Invasive Approaches) trial. The study is designed and powered to evaluate the long-term superiority of revascularization plus the best available medical therapy as compared with the best available medical therapy alone with respect to cardiovascular death or MI in patients with stable coronary artery disease and moderate-to-severe myocardial ischemia documented by means of noninvasive measures. "Until the results of ISCHEMIA are available, the case for a more durable clinical benefit of PCI beyond relief of angina or a reduction in the rate of subsequent revascularization is likely to remain both elusive and illusory," concluded Dr. Boden, who is a coprincipal investigator of ISCHEMIA.
Dr. Boden is the chief of medicine at the Albany Stratton Veterans Affairs Medical Center and vice chair of the department of medicine at Albany (N.Y.) Medical Center. He reported that he is a paid consultant for Arbor Pharmaceuticals and is a speaker for Abbott Laboratories and Gilead Sciences.
The recommendation to terminate FAME 2 by the independent data and safety monitoring board at 7 months’ follow-up was based solely on one highly significant treatment difference. That difference was in the end point of urgent revascularization, which was performed in 49 patients in the group that received the best available medical therapy alone vs. 7 patients in the group that underwent percutaneous coronary intervention and also received the best available medical therapy, Dr. William E. Boden wrote in an editorial accompanying the FAME 2 report (N. Engl. J. Med. 2012 Aug. 28 [doi:10.1056/NEJMe1208620]).
"There were very few ‘hard’ events overall, with only four deaths (three in the medical-therapy group and one in the PCI group) and 29 myocardial infarctions (14 in the medical-therapy group and 15 in the PCI group). Of note, the definition of urgent revascularization was largely a clinical one and did not require evidence of ischemia or positive cardiac biomarkers in all patients; 29 of the 56 unplanned revascularizations (52%) were classified solely on the basis of clinical features, whereas in an exploratory subgroup analysis of the remaining 27 patients, there were fewer revascularizations triggered by a myocardial infarction or electrocardiographic evidence of ischemia in the PCI group than in the medical-therapy group," wrote Dr. Boden.
"Clearly, FFR [fractional flow reserve] holds potential promise for a more targeted approach to PCI that might be more clinically effective and cost effective than visually directed PCI for all angiographically significant stenoses," he continued. "Unfortunately, the early termination of the FAME 2 trial before full enrollment and follow-up were achieved, the neutral effects on the rate of death or myocardial infarction, and the lack of a significant, sustained treatment effect on the reduction of angina beyond 6 months leave more questions than answers."
The FAME 2 and COURAGE [Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation] trials are remarkably similar and showed that PCI reduced only the need for revascularization; in the COURAGE trial, there was a significant 40% reduction, whereas "neither the FAME 2 trial (with a mean 7 months of follow-up) nor the COURAGE trial (with a mean 55 months of follow-up) showed a benefit from PCI with respect to a reduction in the rate of death or myocardial infarction. The FAME 2 trial sought to establish the scientific basis for an FFR-guided PCI strategy for all functionally significant stenoses, but the results make this prospect somewhat unappealing," he wrote. Dr. Boden was the lead author of the COURAGE trial.
Current practice guidelines advocate the selective use of FFR to guide PCI decision making regarding borderline visual lesions (approximately 50%-70% stenosis). "It seems likely that the more routine use of FFR for all angiographically-significant stenoses would add considerable time, cost, and complexity to each PCI procedure and might also increase the risk of catheter-related complications such as coronary dissection and perforation," Dr. Boden pointed out.
Some of the uncertainty arising from FAME 2 may be resolved with the results of the ongoing ISCHEMIA (International Study of Comparative Health Effectiveness with Medical and Invasive Approaches) trial. The study is designed and powered to evaluate the long-term superiority of revascularization plus the best available medical therapy as compared with the best available medical therapy alone with respect to cardiovascular death or MI in patients with stable coronary artery disease and moderate-to-severe myocardial ischemia documented by means of noninvasive measures. "Until the results of ISCHEMIA are available, the case for a more durable clinical benefit of PCI beyond relief of angina or a reduction in the rate of subsequent revascularization is likely to remain both elusive and illusory," concluded Dr. Boden, who is a coprincipal investigator of ISCHEMIA.
Dr. Boden is the chief of medicine at the Albany Stratton Veterans Affairs Medical Center and vice chair of the department of medicine at Albany (N.Y.) Medical Center. He reported that he is a paid consultant for Arbor Pharmaceuticals and is a speaker for Abbott Laboratories and Gilead Sciences.
The recommendation to terminate FAME 2 by the independent data and safety monitoring board at 7 months’ follow-up was based solely on one highly significant treatment difference. That difference was in the end point of urgent revascularization, which was performed in 49 patients in the group that received the best available medical therapy alone vs. 7 patients in the group that underwent percutaneous coronary intervention and also received the best available medical therapy, Dr. William E. Boden wrote in an editorial accompanying the FAME 2 report (N. Engl. J. Med. 2012 Aug. 28 [doi:10.1056/NEJMe1208620]).
"There were very few ‘hard’ events overall, with only four deaths (three in the medical-therapy group and one in the PCI group) and 29 myocardial infarctions (14 in the medical-therapy group and 15 in the PCI group). Of note, the definition of urgent revascularization was largely a clinical one and did not require evidence of ischemia or positive cardiac biomarkers in all patients; 29 of the 56 unplanned revascularizations (52%) were classified solely on the basis of clinical features, whereas in an exploratory subgroup analysis of the remaining 27 patients, there were fewer revascularizations triggered by a myocardial infarction or electrocardiographic evidence of ischemia in the PCI group than in the medical-therapy group," wrote Dr. Boden.
"Clearly, FFR [fractional flow reserve] holds potential promise for a more targeted approach to PCI that might be more clinically effective and cost effective than visually directed PCI for all angiographically significant stenoses," he continued. "Unfortunately, the early termination of the FAME 2 trial before full enrollment and follow-up were achieved, the neutral effects on the rate of death or myocardial infarction, and the lack of a significant, sustained treatment effect on the reduction of angina beyond 6 months leave more questions than answers."
The FAME 2 and COURAGE [Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation] trials are remarkably similar and showed that PCI reduced only the need for revascularization; in the COURAGE trial, there was a significant 40% reduction, whereas "neither the FAME 2 trial (with a mean 7 months of follow-up) nor the COURAGE trial (with a mean 55 months of follow-up) showed a benefit from PCI with respect to a reduction in the rate of death or myocardial infarction. The FAME 2 trial sought to establish the scientific basis for an FFR-guided PCI strategy for all functionally significant stenoses, but the results make this prospect somewhat unappealing," he wrote. Dr. Boden was the lead author of the COURAGE trial.
Current practice guidelines advocate the selective use of FFR to guide PCI decision making regarding borderline visual lesions (approximately 50%-70% stenosis). "It seems likely that the more routine use of FFR for all angiographically-significant stenoses would add considerable time, cost, and complexity to each PCI procedure and might also increase the risk of catheter-related complications such as coronary dissection and perforation," Dr. Boden pointed out.
Some of the uncertainty arising from FAME 2 may be resolved with the results of the ongoing ISCHEMIA (International Study of Comparative Health Effectiveness with Medical and Invasive Approaches) trial. The study is designed and powered to evaluate the long-term superiority of revascularization plus the best available medical therapy as compared with the best available medical therapy alone with respect to cardiovascular death or MI in patients with stable coronary artery disease and moderate-to-severe myocardial ischemia documented by means of noninvasive measures. "Until the results of ISCHEMIA are available, the case for a more durable clinical benefit of PCI beyond relief of angina or a reduction in the rate of subsequent revascularization is likely to remain both elusive and illusory," concluded Dr. Boden, who is a coprincipal investigator of ISCHEMIA.
Dr. Boden is the chief of medicine at the Albany Stratton Veterans Affairs Medical Center and vice chair of the department of medicine at Albany (N.Y.) Medical Center. He reported that he is a paid consultant for Arbor Pharmaceuticals and is a speaker for Abbott Laboratories and Gilead Sciences.
The use of fractional flow reserve to guide percutaneous coronary intervention, along with best medical management, sharply reduced the need for urgent revascularization in patients with stable coronary artery disease and at least one physiologically significant lesion.
However, FFR-guided percutaneous coronary intervention (PCI) had little effect on deaths or myocardial infarctions, when compared with best medical management alone, according to the results of the FAME 2 (Fractional Flow Reserve vs. Angiography for Multivessel Evaluation 2) trial, which was conducted at 28 sites in Europe and North America and halted early.
The percentage of patients who had an MI, death, or urgent revascularization (the combined primary end point) was significantly lower in the PCI group than in the medical therapy group (4.3% vs.12.7%; hazard ratio with PCI, 0.32). This difference was driven by a 13-fold increase in the need for urgent revascularization in the medical-therapy group. Notably, the rate of death from any cause and the rate of MI did not differ significantly between the PCI group and the medical therapy group.
Importantly, patient recruitment was stopped on Jan. 15, 2012, at the recommendation of an independent data and safety monitoring board because of the highly significant difference in the incidence rates of the primary end point between the PCI and medical-therapy groups.
The results of the study were released in the New England Journal of Medicine on Aug. 28 to coincide with the presentation of the study at the annual congress of the European Society of Cardiology (N. Engl. J. Med. 2012 Aug. 28 [doi:10.1056/NEJMoa1205361]).
A total of 1,220 patients out a planned 1,832 were enrolled. Of those, 888 patients had at least one stenosis with an FFR of 0.80 or less: 447 patients were randomly assigned to FFR-guided PCI plus the best available medical therapy, and 441 patients to the best available medical therapy alone. The 332 patients with angiographically significant stenoses, but none with an FFR of 0.80 or less were enrolled in the registry and received the best available medical therapy alone. The mean duration of follow-up was 212 days.
Patients in stable condition who were appropriate candidates for PCI and who had angiographically assessed one-, two-, or three-vessel coronary artery disease suitable for PCI were included in the trial.
All patients were prescribed aspirin at a dose of 80-325 mg daily, metoprolol at a dose of 50 mg-200 mg daily (or any other beta-blocker), lisinopril (at least 5 mg daily, or another ACE inhibitor or an angiotensin receptor blocker)and atorvastatin (20-80 mg daily, or another statin).
All PCI patients were treated with second-generation drug-eluting stents.
Among the 56 patients who underwent urgent revascularization, the procedure was triggered by a MI in 12 patients (21%), by unstable angina accompanied by evidence of ischemia on ECG in 15 patients (27%), and by unstable angina diagnosed on the basis of clinical features in 29 patients (52%).
Patients in the PCI group were 86% less likely to undergo any revascularization and 83% less likely to undergo or nonurgent revascularization than were those in the medical therapy group.
The researchers identified several factors that may explain the differences between results in the present study and those in previous trials involving patients with stable coronary disease. "First, in previous trials in which various revascularization methods were compared with the best available medical therapy, patient enrollment was based primarily on angiographic findings, with or without noninvasive documentation of ischemia. It is likely that a sizable proportion of the patients had only limited ischemia," wrote lead investigator Dr. Bernard B. De Bruyne and his coinvestigators. Dr. De Bruyne is the codirector of the Cardiovascular Center at OLV Hospital in Aalst, Belgium.
"Even in the COURAGE (Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation) trial, in which noninvasive testing was performed in 85% of the patients, less than one-third of the patients had more than 10% ischemia on myocardial perfusion imaging. In daily clinical practice, less than half of patients undergo noninvasive stress testing before elective PCI. In the current trial, all the patients who underwent randomization had at least one functionally significant stenosis," they observed (N. Engl. J. Med. 2007;356:1503-16).
Second, PCI was performed only in lesions with an FFR of 0.80 or less. "This FFR-guided approach is associated with a better clinical outcome than that with PCI performed on the basis of angiographic results alone. These features probably explain the similarity of event rates between patients who were treated with PCI plus the best available medical therapy and patients with equivalent baseline characteristics but no functionally significant lesions who were enrolled in the registry and treated with the best available medical therapy alone," according to the investigators.
Third, second-generation drug-eluting stents were used in PCIs. This strategy is associated with a low number of repeat revascularizations. Finally, the primary end point included urgent revascularization, a component that was not included in the primary end point of previous trials.
The study was sponsored by St. Jude Medical, which makes the two pressure wires used in the trial. The company was involved in the collection and source verification of the data but not in the conduct of the trial. Dr. De Bruyne reported receiving consulting/honorarium and travel fees from St. Jude Medical. Most of the investigators had significant financial relationships with St. Jude Medical, as well as with other device and pharmaceutical companies. One author is an employee of St. Jude Medical.
The use of fractional flow reserve to guide percutaneous coronary intervention, along with best medical management, sharply reduced the need for urgent revascularization in patients with stable coronary artery disease and at least one physiologically significant lesion.
However, FFR-guided percutaneous coronary intervention (PCI) had little effect on deaths or myocardial infarctions, when compared with best medical management alone, according to the results of the FAME 2 (Fractional Flow Reserve vs. Angiography for Multivessel Evaluation 2) trial, which was conducted at 28 sites in Europe and North America and halted early.
The percentage of patients who had an MI, death, or urgent revascularization (the combined primary end point) was significantly lower in the PCI group than in the medical therapy group (4.3% vs.12.7%; hazard ratio with PCI, 0.32). This difference was driven by a 13-fold increase in the need for urgent revascularization in the medical-therapy group. Notably, the rate of death from any cause and the rate of MI did not differ significantly between the PCI group and the medical therapy group.
Importantly, patient recruitment was stopped on Jan. 15, 2012, at the recommendation of an independent data and safety monitoring board because of the highly significant difference in the incidence rates of the primary end point between the PCI and medical-therapy groups.
The results of the study were released in the New England Journal of Medicine on Aug. 28 to coincide with the presentation of the study at the annual congress of the European Society of Cardiology (N. Engl. J. Med. 2012 Aug. 28 [doi:10.1056/NEJMoa1205361]).
A total of 1,220 patients out a planned 1,832 were enrolled. Of those, 888 patients had at least one stenosis with an FFR of 0.80 or less: 447 patients were randomly assigned to FFR-guided PCI plus the best available medical therapy, and 441 patients to the best available medical therapy alone. The 332 patients with angiographically significant stenoses, but none with an FFR of 0.80 or less were enrolled in the registry and received the best available medical therapy alone. The mean duration of follow-up was 212 days.
Patients in stable condition who were appropriate candidates for PCI and who had angiographically assessed one-, two-, or three-vessel coronary artery disease suitable for PCI were included in the trial.
All patients were prescribed aspirin at a dose of 80-325 mg daily, metoprolol at a dose of 50 mg-200 mg daily (or any other beta-blocker), lisinopril (at least 5 mg daily, or another ACE inhibitor or an angiotensin receptor blocker)and atorvastatin (20-80 mg daily, or another statin).
All PCI patients were treated with second-generation drug-eluting stents.
Among the 56 patients who underwent urgent revascularization, the procedure was triggered by a MI in 12 patients (21%), by unstable angina accompanied by evidence of ischemia on ECG in 15 patients (27%), and by unstable angina diagnosed on the basis of clinical features in 29 patients (52%).
Patients in the PCI group were 86% less likely to undergo any revascularization and 83% less likely to undergo or nonurgent revascularization than were those in the medical therapy group.
The researchers identified several factors that may explain the differences between results in the present study and those in previous trials involving patients with stable coronary disease. "First, in previous trials in which various revascularization methods were compared with the best available medical therapy, patient enrollment was based primarily on angiographic findings, with or without noninvasive documentation of ischemia. It is likely that a sizable proportion of the patients had only limited ischemia," wrote lead investigator Dr. Bernard B. De Bruyne and his coinvestigators. Dr. De Bruyne is the codirector of the Cardiovascular Center at OLV Hospital in Aalst, Belgium.
"Even in the COURAGE (Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation) trial, in which noninvasive testing was performed in 85% of the patients, less than one-third of the patients had more than 10% ischemia on myocardial perfusion imaging. In daily clinical practice, less than half of patients undergo noninvasive stress testing before elective PCI. In the current trial, all the patients who underwent randomization had at least one functionally significant stenosis," they observed (N. Engl. J. Med. 2007;356:1503-16).
Second, PCI was performed only in lesions with an FFR of 0.80 or less. "This FFR-guided approach is associated with a better clinical outcome than that with PCI performed on the basis of angiographic results alone. These features probably explain the similarity of event rates between patients who were treated with PCI plus the best available medical therapy and patients with equivalent baseline characteristics but no functionally significant lesions who were enrolled in the registry and treated with the best available medical therapy alone," according to the investigators.
Third, second-generation drug-eluting stents were used in PCIs. This strategy is associated with a low number of repeat revascularizations. Finally, the primary end point included urgent revascularization, a component that was not included in the primary end point of previous trials.
The study was sponsored by St. Jude Medical, which makes the two pressure wires used in the trial. The company was involved in the collection and source verification of the data but not in the conduct of the trial. Dr. De Bruyne reported receiving consulting/honorarium and travel fees from St. Jude Medical. Most of the investigators had significant financial relationships with St. Jude Medical, as well as with other device and pharmaceutical companies. One author is an employee of St. Jude Medical.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: The percentage of patients who had an MI, death, or urgent revascularization (the combined primary end point) was significantly lower in patients who underwent FFR-guided PCI than in patients who received best medical management alone (4.3% vs.12.7%; hazard ratio with PCI, 0.32). This difference was driven by a 13-fold increase in the need for urgent revascularization in the medical therapy group.
Data Source: Data are from the FAME 2 trial, in 1,220 stable patients who had angiographically assessed coronary artery disease and were appropriate candidates for PCI. The trial was stopped early because of a highly significant difference in the primary outcome.
Disclosures: The study was sponsored by St. Jude Medical. Dr. De Bruyne reported receiving consulting/honorarium and travel fees from St. Jude Medical. Most of the investigators had significant financial relationships with St. Jude Medical, as well as with other device and pharmaceutical companies. One author is an employee of St. Jude Medical.
Equally Low Thrombosis Seen With Zotarolimus-, Sirolimus-Eluting Stents
Rates of stent thrombosis were low and similar between patients implanted with zotarolimus-eluting stents and those with sirolimus-eluting stents in a large, randomized controlled trial.
Over a 3-year period, thrombosis occurred in less than 2% of each group. Furthermore, there were no significant between-group differences in overall mortality, myocardial infarction, or cardiac death, Dr. Edoardo Camenzind and colleagues reported in the August 27 online issue of the Lancet. The study results were simultaneously presented at the annual congress of the European Society of Cardiology.
The 1.4% rate of stent thrombosis noted with the zotarolimus stent matched the expected rate of 1.5%, but the rate with the sirolimus stent was lower than anticipated, with an expected rate of 2.5% and an observed rate of 1.8%, wrote Dr. Camenzind of the University of Geneva and his coauthors (Lancet Aug. 27, 2012 [doi: 10.1016/S0140-6736(12)61336-1]).
But the Patient Related Outcomes With Endeavor Versus Cypher Stenting Trial (PROTECT) did show some time-related differences between the two devices, finding more thromboses in the first year with the zotarolimus stent (Z-ES, Endeavor), but more in years 2 and 3 with the sirolimus stent (S-ES, Cypher).
"Interestingly, the pattern of events over time was distributed differentially, with both devices having the same incidence of stent thrombosis, but C-ES having fewer late, but more very late, stent thrombosis than Z-ES," the authors noted. "The higher incidence of late stent thrombosis in the Z-ES group was driven by an increased incidence of definite stent thrombosis."
The study enrolled 8,709 patients who underwent an elective, unplanned, or emergency stenting of coronary arteries. They were a mean of 63 years old. The most common reason for stent placement was acute myocardial infarction (26%). Other reasons for stenting included unstable or stable angina or silent ischemia.
At discharge, 96% were on dual antiplatelet therapy. This number had dropped to 88% at 1 year, 37% at 2 years, and 30% by 3 years.
At the end of follow-up, definite or probable stent thrombosis had occurred in 61 (1.4%) of the Z-ES group and in 75 (1.8%) of the S-ES group – not a significant difference. Nor were there significant differences in any of the main secondary end points of total death and nonfatal MI, or cardiac death and nonfatal MI.
The study revealed a distinct time-associated pattern of thrombosis. In the first month after the procedure, definite or probable thrombosis occurred in 31 of the Z-ES group and 26 of the S-ES group. From days 31 to 360, the numbers were 17 and 5, respectively. During the last 2 years of follow-up, there were 13 definite or probable thromboses in the Z-ES group and 44 in the S-ES group.
Over the entire study period, the Z-ES was associated with a lower incidence of definite stent thrombosis than the S-ES (0.7% vs. 1.2%), but the S-ES had a lower incidence of target vessel revascularization (7.1% vs. 8.2%). None of these differences were significant, however.
Those findings also showed time-related differences. During the first year of the study, significantly more target vessel revascularizations occurred in the Z-ES group (5.8% vs. 3.4%), but that difference was reversed in years 2-3 (4.0% S-ES vs. 2.9% Z-ES).
Although slight, the differences observed in PROTECT are still important, the investigators said: "Given the seriousness of clinical manifestation of stent thrombosis, typically death or myocardial infarction, any reduction has clinical relevance. Furthermore, hundreds of thousands of patients worldwide have been implanted with these devices."
Follow-up on these patients will continue for another 2 years. This "will show whether curves of definite and definite or probable stent thrombosis further diverge and will translate into differences in clinical safety outcomes," Dr. Camenzind and his associates said.
The study was funded by Medtronic, maker of the Endeavor stent. Dr. Camenzind had no financial disclosures but several of his coauthors noted receiving financial remuneration from Medtronic and from other pharmaceutical companies and device manufacturers.
Results of the PROTECT study are academic now, but perhaps still interesting to physicians caring for the millions of patients who still carry the zotarolimus- and sirolimus-eluting stents, Dr. Robert A. Byrne and his colleagues wrote in an accompanying editorial.
"Since the study began in 2007, a significant shift has occurred in interventional cardiology practice, such that both study devices have largely fallen out of use worldwide, being superseded by new devices with increased efficacy and decreased rates of stent thrombosis," wrote Dr. Byrne. "This shift undoubtedly limits the immediate clinical relevance of these findings."
Dr. Byrne and his coauthors also said that PROTECT investigators problematically searched too hard for any interesting tidbits in their secondary analyses.
"The authors report a negative clinical trial with no differences in the primary endpoint. This finding is supported by an absence of difference in the main secondary endpoints. For this reason, the value of all additional analyses presented in the article is restricted, and to highlight differences in the temporal distribution of stent thrombosis between the two devices is inadvisable. This analysis was not prespecified and the probability of a chance finding is high."
The time-driven conclusions can be seeing as a never-ending division problem, they suggested.
"If one notes that stent thrombosis beyond 1 year is higher with [the sirolimus-eluting stent] ... then one must also observe that the number of patients with stent thrombosis up to 1 year is higher with [the zotarolimus-eluting stent]. To take the argument further, one must then question whether it is preferable for a patient to suffer stent thrombosis during the first year or after it: reductio ad absurdum" – arguing on and on that the finding is true because a false result follows from its denial, they wrote.
Dr. Byrne is an interventional cardiologist at Deutsches Herzzentrum, Munich. He had no financial disclosures, but a coauthor disclosed that he has received lecture fees from Medtronic, as well as other device and pharmaceutical companies.
Results of the PROTECT study are academic now, but perhaps still interesting to physicians caring for the millions of patients who still carry the zotarolimus- and sirolimus-eluting stents, Dr. Robert A. Byrne and his colleagues wrote in an accompanying editorial.
"Since the study began in 2007, a significant shift has occurred in interventional cardiology practice, such that both study devices have largely fallen out of use worldwide, being superseded by new devices with increased efficacy and decreased rates of stent thrombosis," wrote Dr. Byrne. "This shift undoubtedly limits the immediate clinical relevance of these findings."
Dr. Byrne and his coauthors also said that PROTECT investigators problematically searched too hard for any interesting tidbits in their secondary analyses.
"The authors report a negative clinical trial with no differences in the primary endpoint. This finding is supported by an absence of difference in the main secondary endpoints. For this reason, the value of all additional analyses presented in the article is restricted, and to highlight differences in the temporal distribution of stent thrombosis between the two devices is inadvisable. This analysis was not prespecified and the probability of a chance finding is high."
The time-driven conclusions can be seeing as a never-ending division problem, they suggested.
"If one notes that stent thrombosis beyond 1 year is higher with [the sirolimus-eluting stent] ... then one must also observe that the number of patients with stent thrombosis up to 1 year is higher with [the zotarolimus-eluting stent]. To take the argument further, one must then question whether it is preferable for a patient to suffer stent thrombosis during the first year or after it: reductio ad absurdum" – arguing on and on that the finding is true because a false result follows from its denial, they wrote.
Dr. Byrne is an interventional cardiologist at Deutsches Herzzentrum, Munich. He had no financial disclosures, but a coauthor disclosed that he has received lecture fees from Medtronic, as well as other device and pharmaceutical companies.
Results of the PROTECT study are academic now, but perhaps still interesting to physicians caring for the millions of patients who still carry the zotarolimus- and sirolimus-eluting stents, Dr. Robert A. Byrne and his colleagues wrote in an accompanying editorial.
"Since the study began in 2007, a significant shift has occurred in interventional cardiology practice, such that both study devices have largely fallen out of use worldwide, being superseded by new devices with increased efficacy and decreased rates of stent thrombosis," wrote Dr. Byrne. "This shift undoubtedly limits the immediate clinical relevance of these findings."
Dr. Byrne and his coauthors also said that PROTECT investigators problematically searched too hard for any interesting tidbits in their secondary analyses.
"The authors report a negative clinical trial with no differences in the primary endpoint. This finding is supported by an absence of difference in the main secondary endpoints. For this reason, the value of all additional analyses presented in the article is restricted, and to highlight differences in the temporal distribution of stent thrombosis between the two devices is inadvisable. This analysis was not prespecified and the probability of a chance finding is high."
The time-driven conclusions can be seeing as a never-ending division problem, they suggested.
"If one notes that stent thrombosis beyond 1 year is higher with [the sirolimus-eluting stent] ... then one must also observe that the number of patients with stent thrombosis up to 1 year is higher with [the zotarolimus-eluting stent]. To take the argument further, one must then question whether it is preferable for a patient to suffer stent thrombosis during the first year or after it: reductio ad absurdum" – arguing on and on that the finding is true because a false result follows from its denial, they wrote.
Dr. Byrne is an interventional cardiologist at Deutsches Herzzentrum, Munich. He had no financial disclosures, but a coauthor disclosed that he has received lecture fees from Medtronic, as well as other device and pharmaceutical companies.
Rates of stent thrombosis were low and similar between patients implanted with zotarolimus-eluting stents and those with sirolimus-eluting stents in a large, randomized controlled trial.
Over a 3-year period, thrombosis occurred in less than 2% of each group. Furthermore, there were no significant between-group differences in overall mortality, myocardial infarction, or cardiac death, Dr. Edoardo Camenzind and colleagues reported in the August 27 online issue of the Lancet. The study results were simultaneously presented at the annual congress of the European Society of Cardiology.
The 1.4% rate of stent thrombosis noted with the zotarolimus stent matched the expected rate of 1.5%, but the rate with the sirolimus stent was lower than anticipated, with an expected rate of 2.5% and an observed rate of 1.8%, wrote Dr. Camenzind of the University of Geneva and his coauthors (Lancet Aug. 27, 2012 [doi: 10.1016/S0140-6736(12)61336-1]).
But the Patient Related Outcomes With Endeavor Versus Cypher Stenting Trial (PROTECT) did show some time-related differences between the two devices, finding more thromboses in the first year with the zotarolimus stent (Z-ES, Endeavor), but more in years 2 and 3 with the sirolimus stent (S-ES, Cypher).
"Interestingly, the pattern of events over time was distributed differentially, with both devices having the same incidence of stent thrombosis, but C-ES having fewer late, but more very late, stent thrombosis than Z-ES," the authors noted. "The higher incidence of late stent thrombosis in the Z-ES group was driven by an increased incidence of definite stent thrombosis."
The study enrolled 8,709 patients who underwent an elective, unplanned, or emergency stenting of coronary arteries. They were a mean of 63 years old. The most common reason for stent placement was acute myocardial infarction (26%). Other reasons for stenting included unstable or stable angina or silent ischemia.
At discharge, 96% were on dual antiplatelet therapy. This number had dropped to 88% at 1 year, 37% at 2 years, and 30% by 3 years.
At the end of follow-up, definite or probable stent thrombosis had occurred in 61 (1.4%) of the Z-ES group and in 75 (1.8%) of the S-ES group – not a significant difference. Nor were there significant differences in any of the main secondary end points of total death and nonfatal MI, or cardiac death and nonfatal MI.
The study revealed a distinct time-associated pattern of thrombosis. In the first month after the procedure, definite or probable thrombosis occurred in 31 of the Z-ES group and 26 of the S-ES group. From days 31 to 360, the numbers were 17 and 5, respectively. During the last 2 years of follow-up, there were 13 definite or probable thromboses in the Z-ES group and 44 in the S-ES group.
Over the entire study period, the Z-ES was associated with a lower incidence of definite stent thrombosis than the S-ES (0.7% vs. 1.2%), but the S-ES had a lower incidence of target vessel revascularization (7.1% vs. 8.2%). None of these differences were significant, however.
Those findings also showed time-related differences. During the first year of the study, significantly more target vessel revascularizations occurred in the Z-ES group (5.8% vs. 3.4%), but that difference was reversed in years 2-3 (4.0% S-ES vs. 2.9% Z-ES).
Although slight, the differences observed in PROTECT are still important, the investigators said: "Given the seriousness of clinical manifestation of stent thrombosis, typically death or myocardial infarction, any reduction has clinical relevance. Furthermore, hundreds of thousands of patients worldwide have been implanted with these devices."
Follow-up on these patients will continue for another 2 years. This "will show whether curves of definite and definite or probable stent thrombosis further diverge and will translate into differences in clinical safety outcomes," Dr. Camenzind and his associates said.
The study was funded by Medtronic, maker of the Endeavor stent. Dr. Camenzind had no financial disclosures but several of his coauthors noted receiving financial remuneration from Medtronic and from other pharmaceutical companies and device manufacturers.
Rates of stent thrombosis were low and similar between patients implanted with zotarolimus-eluting stents and those with sirolimus-eluting stents in a large, randomized controlled trial.
Over a 3-year period, thrombosis occurred in less than 2% of each group. Furthermore, there were no significant between-group differences in overall mortality, myocardial infarction, or cardiac death, Dr. Edoardo Camenzind and colleagues reported in the August 27 online issue of the Lancet. The study results were simultaneously presented at the annual congress of the European Society of Cardiology.
The 1.4% rate of stent thrombosis noted with the zotarolimus stent matched the expected rate of 1.5%, but the rate with the sirolimus stent was lower than anticipated, with an expected rate of 2.5% and an observed rate of 1.8%, wrote Dr. Camenzind of the University of Geneva and his coauthors (Lancet Aug. 27, 2012 [doi: 10.1016/S0140-6736(12)61336-1]).
But the Patient Related Outcomes With Endeavor Versus Cypher Stenting Trial (PROTECT) did show some time-related differences between the two devices, finding more thromboses in the first year with the zotarolimus stent (Z-ES, Endeavor), but more in years 2 and 3 with the sirolimus stent (S-ES, Cypher).
"Interestingly, the pattern of events over time was distributed differentially, with both devices having the same incidence of stent thrombosis, but C-ES having fewer late, but more very late, stent thrombosis than Z-ES," the authors noted. "The higher incidence of late stent thrombosis in the Z-ES group was driven by an increased incidence of definite stent thrombosis."
The study enrolled 8,709 patients who underwent an elective, unplanned, or emergency stenting of coronary arteries. They were a mean of 63 years old. The most common reason for stent placement was acute myocardial infarction (26%). Other reasons for stenting included unstable or stable angina or silent ischemia.
At discharge, 96% were on dual antiplatelet therapy. This number had dropped to 88% at 1 year, 37% at 2 years, and 30% by 3 years.
At the end of follow-up, definite or probable stent thrombosis had occurred in 61 (1.4%) of the Z-ES group and in 75 (1.8%) of the S-ES group – not a significant difference. Nor were there significant differences in any of the main secondary end points of total death and nonfatal MI, or cardiac death and nonfatal MI.
The study revealed a distinct time-associated pattern of thrombosis. In the first month after the procedure, definite or probable thrombosis occurred in 31 of the Z-ES group and 26 of the S-ES group. From days 31 to 360, the numbers were 17 and 5, respectively. During the last 2 years of follow-up, there were 13 definite or probable thromboses in the Z-ES group and 44 in the S-ES group.
Over the entire study period, the Z-ES was associated with a lower incidence of definite stent thrombosis than the S-ES (0.7% vs. 1.2%), but the S-ES had a lower incidence of target vessel revascularization (7.1% vs. 8.2%). None of these differences were significant, however.
Those findings also showed time-related differences. During the first year of the study, significantly more target vessel revascularizations occurred in the Z-ES group (5.8% vs. 3.4%), but that difference was reversed in years 2-3 (4.0% S-ES vs. 2.9% Z-ES).
Although slight, the differences observed in PROTECT are still important, the investigators said: "Given the seriousness of clinical manifestation of stent thrombosis, typically death or myocardial infarction, any reduction has clinical relevance. Furthermore, hundreds of thousands of patients worldwide have been implanted with these devices."
Follow-up on these patients will continue for another 2 years. This "will show whether curves of definite and definite or probable stent thrombosis further diverge and will translate into differences in clinical safety outcomes," Dr. Camenzind and his associates said.
The study was funded by Medtronic, maker of the Endeavor stent. Dr. Camenzind had no financial disclosures but several of his coauthors noted receiving financial remuneration from Medtronic and from other pharmaceutical companies and device manufacturers.
FROM THE LANCET
Major Finding: Over 3 years, the incidence of stent thrombosis was 1.4% with a zotarolimus-eluting device and 1.8% with a sirolumus-eluting stent.
Data Source: PROTECT was a randomized controlled trial involving 8,709 patients implanted with drug-eluting stents and followed for 3 years.
Disclosures: The study was funded by Medtronic. Dr. Camenzind had no financial disclosures, but several of his coauthors noted receiving financial remuneration from Medtronic and other pharmaceutical companies and device manufacturers.
ESC Guidelines Tackle TAVI
MUNICH – Transcatheter aortic valve implantation should not be performed in patients at intermediate risk for surgery, according to new European guidelines on the management of valvular heart disease.
"We strongly stress that today, TAVI should not be performed in patients at intermediate risk," said task force chair Dr. Alec Vahanian of the department of cardiology at Bichat Hospital, Paris.
This is the first time that TAVI has been addressed in the guidelines, a joint collaboration between the European Society of Cardiology and the European Association for Cardio-Thoracic Surgery.
The use of TAVI has mushroomed in recent years among high-risk patients with severe aortic stenosis and in those who are not candidates for surgery, but also has been performed in patients at lower risk.
The new guidelines update those issued in 2010, and were prompted by new evidence on risk stratification, diagnostic methods, therapeutic options, and the importance of the collaborative approach between cardiologists and cardiac surgeons, Dr. Vahanian said at the annual congress of the European Society of Cardiology.
"The key message – the core of the document – is to stress teamwork," he said.
The guidelines state that TAVI should be undertaken only with a multidisciplinary "heart team" including cardiologists, cardiac surgeons, and other specialists, if necessary, and only in hospitals with cardiac surgery on-site.
The indications for TAVI, based on results of the randomized PARTNER trial and large European registries, include patients with severe, symptomatic aortic stenosis (AS) who are not suitable for aortic valve replacement (AVR) as assessed by a heart team, and who are likely to gain improvement in their quality of life and are expected to live more than 1 year.
TAVI also should be considered in high-risk patients with severe symptomatic AS who may still be suitable for surgery, but in whom TAVI is favored by a heart team based on the individual risk profile and anatomic suitability, Dr. Vahanian said.
He reported a virtual laundry list of absolute and relative contraindications to TAVI, including an inadequate annulus size, defined as less than 10 mm or greater than 29 mm; thrombus in the left ventricle; active endocarditis; plaques with mobile thrombi in the ascending aorta or arch; inadequate vascular access for a transfemoral/subclavian approach; and a very low left ventricular ejection fraction of less than 20%.
Dr. Vahanian said AVR remains suitable for patients with severe symptomatic AS, including those undergoing coronary artery bypass surgery or surgery of the ascending aorta or another valve, as well as those who are suitable for TAVI, but in whom surgery is favored by a heart team. He observed that there was a great deal of debate among the task force members regarding surgery in symptomatic patients with low flow, low- gradient (defined as less than 40 mm Hg) aortic stenosis, and a normal ejection fraction.
"We have to be extremely cautious and treat only the patient while symptomatic and we are absolutely sure, if we can be sure, that the aortic disease is severe," he remarked.
Disclosures were not provided. The Organizing Committee assumed responsibility for ensuring that all potential conflicts of interest relevant to the program are declared to the participants.
MUNICH – Transcatheter aortic valve implantation should not be performed in patients at intermediate risk for surgery, according to new European guidelines on the management of valvular heart disease.
"We strongly stress that today, TAVI should not be performed in patients at intermediate risk," said task force chair Dr. Alec Vahanian of the department of cardiology at Bichat Hospital, Paris.
This is the first time that TAVI has been addressed in the guidelines, a joint collaboration between the European Society of Cardiology and the European Association for Cardio-Thoracic Surgery.
The use of TAVI has mushroomed in recent years among high-risk patients with severe aortic stenosis and in those who are not candidates for surgery, but also has been performed in patients at lower risk.
The new guidelines update those issued in 2010, and were prompted by new evidence on risk stratification, diagnostic methods, therapeutic options, and the importance of the collaborative approach between cardiologists and cardiac surgeons, Dr. Vahanian said at the annual congress of the European Society of Cardiology.
"The key message – the core of the document – is to stress teamwork," he said.
The guidelines state that TAVI should be undertaken only with a multidisciplinary "heart team" including cardiologists, cardiac surgeons, and other specialists, if necessary, and only in hospitals with cardiac surgery on-site.
The indications for TAVI, based on results of the randomized PARTNER trial and large European registries, include patients with severe, symptomatic aortic stenosis (AS) who are not suitable for aortic valve replacement (AVR) as assessed by a heart team, and who are likely to gain improvement in their quality of life and are expected to live more than 1 year.
TAVI also should be considered in high-risk patients with severe symptomatic AS who may still be suitable for surgery, but in whom TAVI is favored by a heart team based on the individual risk profile and anatomic suitability, Dr. Vahanian said.
He reported a virtual laundry list of absolute and relative contraindications to TAVI, including an inadequate annulus size, defined as less than 10 mm or greater than 29 mm; thrombus in the left ventricle; active endocarditis; plaques with mobile thrombi in the ascending aorta or arch; inadequate vascular access for a transfemoral/subclavian approach; and a very low left ventricular ejection fraction of less than 20%.
Dr. Vahanian said AVR remains suitable for patients with severe symptomatic AS, including those undergoing coronary artery bypass surgery or surgery of the ascending aorta or another valve, as well as those who are suitable for TAVI, but in whom surgery is favored by a heart team. He observed that there was a great deal of debate among the task force members regarding surgery in symptomatic patients with low flow, low- gradient (defined as less than 40 mm Hg) aortic stenosis, and a normal ejection fraction.
"We have to be extremely cautious and treat only the patient while symptomatic and we are absolutely sure, if we can be sure, that the aortic disease is severe," he remarked.
Disclosures were not provided. The Organizing Committee assumed responsibility for ensuring that all potential conflicts of interest relevant to the program are declared to the participants.
MUNICH – Transcatheter aortic valve implantation should not be performed in patients at intermediate risk for surgery, according to new European guidelines on the management of valvular heart disease.
"We strongly stress that today, TAVI should not be performed in patients at intermediate risk," said task force chair Dr. Alec Vahanian of the department of cardiology at Bichat Hospital, Paris.
This is the first time that TAVI has been addressed in the guidelines, a joint collaboration between the European Society of Cardiology and the European Association for Cardio-Thoracic Surgery.
The use of TAVI has mushroomed in recent years among high-risk patients with severe aortic stenosis and in those who are not candidates for surgery, but also has been performed in patients at lower risk.
The new guidelines update those issued in 2010, and were prompted by new evidence on risk stratification, diagnostic methods, therapeutic options, and the importance of the collaborative approach between cardiologists and cardiac surgeons, Dr. Vahanian said at the annual congress of the European Society of Cardiology.
"The key message – the core of the document – is to stress teamwork," he said.
The guidelines state that TAVI should be undertaken only with a multidisciplinary "heart team" including cardiologists, cardiac surgeons, and other specialists, if necessary, and only in hospitals with cardiac surgery on-site.
The indications for TAVI, based on results of the randomized PARTNER trial and large European registries, include patients with severe, symptomatic aortic stenosis (AS) who are not suitable for aortic valve replacement (AVR) as assessed by a heart team, and who are likely to gain improvement in their quality of life and are expected to live more than 1 year.
TAVI also should be considered in high-risk patients with severe symptomatic AS who may still be suitable for surgery, but in whom TAVI is favored by a heart team based on the individual risk profile and anatomic suitability, Dr. Vahanian said.
He reported a virtual laundry list of absolute and relative contraindications to TAVI, including an inadequate annulus size, defined as less than 10 mm or greater than 29 mm; thrombus in the left ventricle; active endocarditis; plaques with mobile thrombi in the ascending aorta or arch; inadequate vascular access for a transfemoral/subclavian approach; and a very low left ventricular ejection fraction of less than 20%.
Dr. Vahanian said AVR remains suitable for patients with severe symptomatic AS, including those undergoing coronary artery bypass surgery or surgery of the ascending aorta or another valve, as well as those who are suitable for TAVI, but in whom surgery is favored by a heart team. He observed that there was a great deal of debate among the task force members regarding surgery in symptomatic patients with low flow, low- gradient (defined as less than 40 mm Hg) aortic stenosis, and a normal ejection fraction.
"We have to be extremely cautious and treat only the patient while symptomatic and we are absolutely sure, if we can be sure, that the aortic disease is severe," he remarked.
Disclosures were not provided. The Organizing Committee assumed responsibility for ensuring that all potential conflicts of interest relevant to the program are declared to the participants.
AT THE ANNUAL CONGRESS OF THE EUROPEAN SOCIETY OF CARDIOLOGY