User login
Treat-to-target strategy ‘not ready for primetime’ in osteoporosis
“A treat-to-target approach is useful in the management of osteoporosis” was the motion proposed in a debate during the recent virtual American Society of Bone and Mineral Research (ASBMR) 2020 annual meeting, and when the votes came in, Michael McClung, MD, who argued against the motion, carried the day.
Agreement with the motion dropped from 63%-46% after McClung, of the Oregon Osteoporosis Center, Portland, put his views forward in opposition to those of Celia L. Gregson, PhD, University of Bristol (England), who argued for the motion on behalf of the European Calcified Tissue Society (ECTS).
Disagreement with the statement rose from 37% predebate to 54% in the postdebate audience polls.
“The debate is part education and part entertainment,” said Dr. McClung, who represented the ASBMR. “I could just as easily have made a strong argument for the motion,” he emphasized in an interview.
On the other hand, “had I been in the audience, as a member of ASBMR relying on data and evidence to make clinical decisions, I would have voted against the motion. As appealing as the strategy sounds, we don’t yet have the hard evidence to support its use nor is there a consensus about what an appropriate target should be, he noted.
Similarly, the debate comoderator and incoming ASBMR President, Suzanne M. Jan de Beur, MD, from Johns Hopkins University, Baltimore, said that a treat-to-target strategy for osteoporosis is an attractive idea, but there is no consensus on how to apply it nor evidence that it improves clinical outcomes.
Treat to target to guide osteoporosis therapy is like going “backwards”
In treat to target, the target – such as bone mineral density (BMD) (the most common one) – is identified before treatment is started, Dr. McClung explained (and as stated in a review article in the New England Journal of Medicine he coauthored on the topic).
“While treat to target has appealing concepts, using risk factors to guide therapy is almost backwards,” he said. “We can’t change bone density very much.”
Treat to target is “not quite ready for prime time,” he concluded in his rebuttal.
Invited to speculate on which of Dr. McClung’s arguments swayed the audience, Dr. Gregson conceded that with a treat-to-target strategy “there is too much focus on getting one target for the whole global population with osteoporosis.”
“This is an oversimplification of a complex disease, and it misses the main message that the target should be decided with the patient not for the patient, which means one can’t just have one rule for everyone. There has to be scope to have different targets for different people so that we can deliver individualized care.”
Also, she noted, “generally people don’t vote to change familiar systems.”
Arguments for treat to target
Dr. Gregson began her argument, however, by stating that treat to target “is now a feasible and useful approach in osteoporosis care.”
The main reasons for adopting this treatment strategy are as follows:
- It provides a proactive approach with a clear goal.
- It includes periodic treatment reassessment, which allows for prompt revisions to treatment.
- It can use targets to guide treatment timing and patient monitoring.
- It includes shared decision-making, the preferred method of patient care.
- It could improve treatment adherence through patient “buy-in” of the target.
- It can use targets to address the risk of rare side effects.
- It allows for sequential treatments, especially for patients at highest risk of fracture.
- It can include more patient-centered outcomes such as reduced , restored range of movement, and ability to live independently.
“Patients are not interested in their T-score. They are interested in pain,” said Dr. Gregson.
“Reduced fracture risk is a very important goal,” she emphasized. Patients “with osteoporosis and a high fracture risk have the most to gain from a treat-to-target approach.”
“Improved access to anabolic osteoporosis treatments mean achieving those goals or targets are now more achievable than ever,” she concluded.
Arguments against treat to target
“Do we truly have an appropriate, meaningful target for osteoporosis?” Dr. McClung began in his counterargument, which cast a seed of doubt in the minds of the audience.
Targets such as no fractures, fracture risk (FRAX score), bone turnover markers, and bone strength have limitations.
Moreover, “do we have treatment strategies to move patients to the chosen target?” he continued. “What is the evidence that a treat-to-target strategy provides better outcomes than our current treatment paradigm?”
After pointing out a lack of evidence that treat to target leads to better outcomes in osteoporosis, he did allow that “recent data about the relationship between treatment-related BMD values and current fracture risk are appreciated and welcomed.”
“However, a treat-to-target strategy will only be successful if the targets are individualized for each patient, those targets are attainable for most patients, and we have evidence that adopting this strategy improves clinical outcomes,” he summarized.
He then quoted his late wife Betsy Love McClung, RN, MN, who had said, “We don’t treat osteoporosis; we treat patients with osteoporosis.”
Dr. McClung wrapped up by stressing: “We should not treat T-scores or any other specific target. We should individualize our therapy based upon the patient’s risk of fracture and other clinical factors.”
As members of the ECTS and ASBMR, and “proud of our reputation of our societies as being scientifically based and driven,” Dr. McClung concluded, “recognizing that a treat-to-target strategy has appeal, we should certainly encourage more research and be attentive to those results.
“But we must hold off on the adoption of the strategy until we have evidence convincing us of its clinical value.”
When to use a treat-to-target strategy
However, “there are some specific situations where I use something like a treat-to-target strategy,” Dr. McClung conceded. “That is, I make decisions and recommendations to the patients about one drug rather than another because I want to maximize the improvement in their bone density.”
For example, “We have known for 15 years that denosumab results in greater increases in bone density than do bisphosphonates,” he continued.
“So I have used that information to make treatment decisions long before the term ‘treat to target’ entered the vocabulary of osteoporosis experts. I simply wanted to induce the largest possible gains in bone density – but I didn’t have a ‘target’ in mind.”
But for most patients, treatment decisions are made based on other factors, such as their fracture risk, he added. BMD is an important risk factor for fracture, but not as important as having had a recent fracture or being old and frail.
“Unfortunately, in most of today’s health systems, decisions about treatment are made on the basis of cost,” he continued. “More often than not, the health plan rules rather than optimal medical practice are the main guides to treatment decisions.”
According to Dr. Gregson, “in some instances, treat to target would be very helpful. I don’t think it will suit everyone, but I think we should have it in our portfolio of management approaches, and we should as an osteoporosis community be trained in its use.”
“Attractive idea, but ...”
Invited to weigh in, Dr. Jan de Beur noted that A1c, blood pressure, and LDL cholesterol targets are used to improve clinical outcomes in patients with diabetes, hypertension, and hyperlipidemia, respectively.
However, “treat to target for the treatment of low BMD is controversial because it is an attractive idea but without consensus on what the target should be and without evidence that treat to target improves clinical outcomes,” she reiterated.
“The potential benefits of treat to target are proactive, clear goals to achieve, shared decision-making with the patient, the possibility for improved adherence, justification for sequence treatments, and balancing risk of rare side effects.”
On the other hand, “barriers to operationalizing the treat-to-target concept is that there is lack of consensus on the target to be achieved [as any specific target may minimize other important risk factors],” she noted.
There is also a “lack of evidence that demonstrates improved clinical outcomes over choosing therapy based on fracture risk, and lack of ability to achieve the target with available therapies in those with very-low bone density,” she concluded.
Dr. McClung has reported receiving consulting fees from Amgen and Myovant and speaker honoraria from Amgen. Dr. Gregson and Dr. Jan de Beur have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
“A treat-to-target approach is useful in the management of osteoporosis” was the motion proposed in a debate during the recent virtual American Society of Bone and Mineral Research (ASBMR) 2020 annual meeting, and when the votes came in, Michael McClung, MD, who argued against the motion, carried the day.
Agreement with the motion dropped from 63%-46% after McClung, of the Oregon Osteoporosis Center, Portland, put his views forward in opposition to those of Celia L. Gregson, PhD, University of Bristol (England), who argued for the motion on behalf of the European Calcified Tissue Society (ECTS).
Disagreement with the statement rose from 37% predebate to 54% in the postdebate audience polls.
“The debate is part education and part entertainment,” said Dr. McClung, who represented the ASBMR. “I could just as easily have made a strong argument for the motion,” he emphasized in an interview.
On the other hand, “had I been in the audience, as a member of ASBMR relying on data and evidence to make clinical decisions, I would have voted against the motion. As appealing as the strategy sounds, we don’t yet have the hard evidence to support its use nor is there a consensus about what an appropriate target should be, he noted.
Similarly, the debate comoderator and incoming ASBMR President, Suzanne M. Jan de Beur, MD, from Johns Hopkins University, Baltimore, said that a treat-to-target strategy for osteoporosis is an attractive idea, but there is no consensus on how to apply it nor evidence that it improves clinical outcomes.
Treat to target to guide osteoporosis therapy is like going “backwards”
In treat to target, the target – such as bone mineral density (BMD) (the most common one) – is identified before treatment is started, Dr. McClung explained (and as stated in a review article in the New England Journal of Medicine he coauthored on the topic).
“While treat to target has appealing concepts, using risk factors to guide therapy is almost backwards,” he said. “We can’t change bone density very much.”
Treat to target is “not quite ready for prime time,” he concluded in his rebuttal.
Invited to speculate on which of Dr. McClung’s arguments swayed the audience, Dr. Gregson conceded that with a treat-to-target strategy “there is too much focus on getting one target for the whole global population with osteoporosis.”
“This is an oversimplification of a complex disease, and it misses the main message that the target should be decided with the patient not for the patient, which means one can’t just have one rule for everyone. There has to be scope to have different targets for different people so that we can deliver individualized care.”
Also, she noted, “generally people don’t vote to change familiar systems.”
Arguments for treat to target
Dr. Gregson began her argument, however, by stating that treat to target “is now a feasible and useful approach in osteoporosis care.”
The main reasons for adopting this treatment strategy are as follows:
- It provides a proactive approach with a clear goal.
- It includes periodic treatment reassessment, which allows for prompt revisions to treatment.
- It can use targets to guide treatment timing and patient monitoring.
- It includes shared decision-making, the preferred method of patient care.
- It could improve treatment adherence through patient “buy-in” of the target.
- It can use targets to address the risk of rare side effects.
- It allows for sequential treatments, especially for patients at highest risk of fracture.
- It can include more patient-centered outcomes such as reduced , restored range of movement, and ability to live independently.
“Patients are not interested in their T-score. They are interested in pain,” said Dr. Gregson.
“Reduced fracture risk is a very important goal,” she emphasized. Patients “with osteoporosis and a high fracture risk have the most to gain from a treat-to-target approach.”
“Improved access to anabolic osteoporosis treatments mean achieving those goals or targets are now more achievable than ever,” she concluded.
Arguments against treat to target
“Do we truly have an appropriate, meaningful target for osteoporosis?” Dr. McClung began in his counterargument, which cast a seed of doubt in the minds of the audience.
Targets such as no fractures, fracture risk (FRAX score), bone turnover markers, and bone strength have limitations.
Moreover, “do we have treatment strategies to move patients to the chosen target?” he continued. “What is the evidence that a treat-to-target strategy provides better outcomes than our current treatment paradigm?”
After pointing out a lack of evidence that treat to target leads to better outcomes in osteoporosis, he did allow that “recent data about the relationship between treatment-related BMD values and current fracture risk are appreciated and welcomed.”
“However, a treat-to-target strategy will only be successful if the targets are individualized for each patient, those targets are attainable for most patients, and we have evidence that adopting this strategy improves clinical outcomes,” he summarized.
He then quoted his late wife Betsy Love McClung, RN, MN, who had said, “We don’t treat osteoporosis; we treat patients with osteoporosis.”
Dr. McClung wrapped up by stressing: “We should not treat T-scores or any other specific target. We should individualize our therapy based upon the patient’s risk of fracture and other clinical factors.”
As members of the ECTS and ASBMR, and “proud of our reputation of our societies as being scientifically based and driven,” Dr. McClung concluded, “recognizing that a treat-to-target strategy has appeal, we should certainly encourage more research and be attentive to those results.
“But we must hold off on the adoption of the strategy until we have evidence convincing us of its clinical value.”
When to use a treat-to-target strategy
However, “there are some specific situations where I use something like a treat-to-target strategy,” Dr. McClung conceded. “That is, I make decisions and recommendations to the patients about one drug rather than another because I want to maximize the improvement in their bone density.”
For example, “We have known for 15 years that denosumab results in greater increases in bone density than do bisphosphonates,” he continued.
“So I have used that information to make treatment decisions long before the term ‘treat to target’ entered the vocabulary of osteoporosis experts. I simply wanted to induce the largest possible gains in bone density – but I didn’t have a ‘target’ in mind.”
But for most patients, treatment decisions are made based on other factors, such as their fracture risk, he added. BMD is an important risk factor for fracture, but not as important as having had a recent fracture or being old and frail.
“Unfortunately, in most of today’s health systems, decisions about treatment are made on the basis of cost,” he continued. “More often than not, the health plan rules rather than optimal medical practice are the main guides to treatment decisions.”
According to Dr. Gregson, “in some instances, treat to target would be very helpful. I don’t think it will suit everyone, but I think we should have it in our portfolio of management approaches, and we should as an osteoporosis community be trained in its use.”
“Attractive idea, but ...”
Invited to weigh in, Dr. Jan de Beur noted that A1c, blood pressure, and LDL cholesterol targets are used to improve clinical outcomes in patients with diabetes, hypertension, and hyperlipidemia, respectively.
However, “treat to target for the treatment of low BMD is controversial because it is an attractive idea but without consensus on what the target should be and without evidence that treat to target improves clinical outcomes,” she reiterated.
“The potential benefits of treat to target are proactive, clear goals to achieve, shared decision-making with the patient, the possibility for improved adherence, justification for sequence treatments, and balancing risk of rare side effects.”
On the other hand, “barriers to operationalizing the treat-to-target concept is that there is lack of consensus on the target to be achieved [as any specific target may minimize other important risk factors],” she noted.
There is also a “lack of evidence that demonstrates improved clinical outcomes over choosing therapy based on fracture risk, and lack of ability to achieve the target with available therapies in those with very-low bone density,” she concluded.
Dr. McClung has reported receiving consulting fees from Amgen and Myovant and speaker honoraria from Amgen. Dr. Gregson and Dr. Jan de Beur have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
“A treat-to-target approach is useful in the management of osteoporosis” was the motion proposed in a debate during the recent virtual American Society of Bone and Mineral Research (ASBMR) 2020 annual meeting, and when the votes came in, Michael McClung, MD, who argued against the motion, carried the day.
Agreement with the motion dropped from 63%-46% after McClung, of the Oregon Osteoporosis Center, Portland, put his views forward in opposition to those of Celia L. Gregson, PhD, University of Bristol (England), who argued for the motion on behalf of the European Calcified Tissue Society (ECTS).
Disagreement with the statement rose from 37% predebate to 54% in the postdebate audience polls.
“The debate is part education and part entertainment,” said Dr. McClung, who represented the ASBMR. “I could just as easily have made a strong argument for the motion,” he emphasized in an interview.
On the other hand, “had I been in the audience, as a member of ASBMR relying on data and evidence to make clinical decisions, I would have voted against the motion. As appealing as the strategy sounds, we don’t yet have the hard evidence to support its use nor is there a consensus about what an appropriate target should be, he noted.
Similarly, the debate comoderator and incoming ASBMR President, Suzanne M. Jan de Beur, MD, from Johns Hopkins University, Baltimore, said that a treat-to-target strategy for osteoporosis is an attractive idea, but there is no consensus on how to apply it nor evidence that it improves clinical outcomes.
Treat to target to guide osteoporosis therapy is like going “backwards”
In treat to target, the target – such as bone mineral density (BMD) (the most common one) – is identified before treatment is started, Dr. McClung explained (and as stated in a review article in the New England Journal of Medicine he coauthored on the topic).
“While treat to target has appealing concepts, using risk factors to guide therapy is almost backwards,” he said. “We can’t change bone density very much.”
Treat to target is “not quite ready for prime time,” he concluded in his rebuttal.
Invited to speculate on which of Dr. McClung’s arguments swayed the audience, Dr. Gregson conceded that with a treat-to-target strategy “there is too much focus on getting one target for the whole global population with osteoporosis.”
“This is an oversimplification of a complex disease, and it misses the main message that the target should be decided with the patient not for the patient, which means one can’t just have one rule for everyone. There has to be scope to have different targets for different people so that we can deliver individualized care.”
Also, she noted, “generally people don’t vote to change familiar systems.”
Arguments for treat to target
Dr. Gregson began her argument, however, by stating that treat to target “is now a feasible and useful approach in osteoporosis care.”
The main reasons for adopting this treatment strategy are as follows:
- It provides a proactive approach with a clear goal.
- It includes periodic treatment reassessment, which allows for prompt revisions to treatment.
- It can use targets to guide treatment timing and patient monitoring.
- It includes shared decision-making, the preferred method of patient care.
- It could improve treatment adherence through patient “buy-in” of the target.
- It can use targets to address the risk of rare side effects.
- It allows for sequential treatments, especially for patients at highest risk of fracture.
- It can include more patient-centered outcomes such as reduced , restored range of movement, and ability to live independently.
“Patients are not interested in their T-score. They are interested in pain,” said Dr. Gregson.
“Reduced fracture risk is a very important goal,” she emphasized. Patients “with osteoporosis and a high fracture risk have the most to gain from a treat-to-target approach.”
“Improved access to anabolic osteoporosis treatments mean achieving those goals or targets are now more achievable than ever,” she concluded.
Arguments against treat to target
“Do we truly have an appropriate, meaningful target for osteoporosis?” Dr. McClung began in his counterargument, which cast a seed of doubt in the minds of the audience.
Targets such as no fractures, fracture risk (FRAX score), bone turnover markers, and bone strength have limitations.
Moreover, “do we have treatment strategies to move patients to the chosen target?” he continued. “What is the evidence that a treat-to-target strategy provides better outcomes than our current treatment paradigm?”
After pointing out a lack of evidence that treat to target leads to better outcomes in osteoporosis, he did allow that “recent data about the relationship between treatment-related BMD values and current fracture risk are appreciated and welcomed.”
“However, a treat-to-target strategy will only be successful if the targets are individualized for each patient, those targets are attainable for most patients, and we have evidence that adopting this strategy improves clinical outcomes,” he summarized.
He then quoted his late wife Betsy Love McClung, RN, MN, who had said, “We don’t treat osteoporosis; we treat patients with osteoporosis.”
Dr. McClung wrapped up by stressing: “We should not treat T-scores or any other specific target. We should individualize our therapy based upon the patient’s risk of fracture and other clinical factors.”
As members of the ECTS and ASBMR, and “proud of our reputation of our societies as being scientifically based and driven,” Dr. McClung concluded, “recognizing that a treat-to-target strategy has appeal, we should certainly encourage more research and be attentive to those results.
“But we must hold off on the adoption of the strategy until we have evidence convincing us of its clinical value.”
When to use a treat-to-target strategy
However, “there are some specific situations where I use something like a treat-to-target strategy,” Dr. McClung conceded. “That is, I make decisions and recommendations to the patients about one drug rather than another because I want to maximize the improvement in their bone density.”
For example, “We have known for 15 years that denosumab results in greater increases in bone density than do bisphosphonates,” he continued.
“So I have used that information to make treatment decisions long before the term ‘treat to target’ entered the vocabulary of osteoporosis experts. I simply wanted to induce the largest possible gains in bone density – but I didn’t have a ‘target’ in mind.”
But for most patients, treatment decisions are made based on other factors, such as their fracture risk, he added. BMD is an important risk factor for fracture, but not as important as having had a recent fracture or being old and frail.
“Unfortunately, in most of today’s health systems, decisions about treatment are made on the basis of cost,” he continued. “More often than not, the health plan rules rather than optimal medical practice are the main guides to treatment decisions.”
According to Dr. Gregson, “in some instances, treat to target would be very helpful. I don’t think it will suit everyone, but I think we should have it in our portfolio of management approaches, and we should as an osteoporosis community be trained in its use.”
“Attractive idea, but ...”
Invited to weigh in, Dr. Jan de Beur noted that A1c, blood pressure, and LDL cholesterol targets are used to improve clinical outcomes in patients with diabetes, hypertension, and hyperlipidemia, respectively.
However, “treat to target for the treatment of low BMD is controversial because it is an attractive idea but without consensus on what the target should be and without evidence that treat to target improves clinical outcomes,” she reiterated.
“The potential benefits of treat to target are proactive, clear goals to achieve, shared decision-making with the patient, the possibility for improved adherence, justification for sequence treatments, and balancing risk of rare side effects.”
On the other hand, “barriers to operationalizing the treat-to-target concept is that there is lack of consensus on the target to be achieved [as any specific target may minimize other important risk factors],” she noted.
There is also a “lack of evidence that demonstrates improved clinical outcomes over choosing therapy based on fracture risk, and lack of ability to achieve the target with available therapies in those with very-low bone density,” she concluded.
Dr. McClung has reported receiving consulting fees from Amgen and Myovant and speaker honoraria from Amgen. Dr. Gregson and Dr. Jan de Beur have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM ASBMR 2020
More dairy lowers risk of falls, fractures in frail elderly
Consuming more milk, cheese, or yogurt might be a simple, low-cost way to boost bone health and prevent some falls and fractures in older people living in long-term care facilities, according to a new randomized study from Australia.
“Supplementation using dairy foods is likely to be an effective, safe, widely available, and low cost means of curtailing the public health burden of fractures,” said Sandra Iuliano, PhD, from the University of Melbourne, who presented the findings during the virtual American Society of Bone and Mineral Research 2020 annual meeting.
The researchers randomized 60 old-age institutions to provide residents with their usual menus or a diet with more milk, cheese, or yogurt for 2 years.
The residents with the altered menus increased their dairy consumption from 2 servings/day to 3.5 servings/day, which was reflected in a greater intake of calcium and protein, along with fewer falls, total fractures, and hip fractures than in the control group.
“This is the first randomized trial to show a benefit of dairy food intake on risk of fractures,” Walter Willett, MD, DrPH, professor of nutrition and epidemiology at the Harvard School of Public Health, Boston, said in an interview.
The results are “not surprising” because supplements of calcium plus vitamin D have reduced the risk of fractures in a similar population of older residents living in special living facilities, said Dr. Willett, coauthor of a recent review article, “Milk and Health,” published in the New England Journal of Medicine.
“It is important for everyone to have adequate intake of calcium and vitamin D,” he said. However, “it isn’t clear whether it is better to ensure this clinically by supplements, overall healthy diet, or extra dairy intake,” he added, noting that consuming the amount of dairy given in this Australian study is not environmentally sustainable.
Clifford Rosen, MD, professor of medicine, Tufts University, Boston, said in an interview that the Australian researchers studied the impact of increased dietary calcium and protein, not the impact of vitamin D via supplements.
“This is progress toward getting interventions to our most needy residents to prevent fractures – probably the most compelling data that we have had in a number of years,” he noted.
The current study shows “it’s not [the] vitamin D,” because the residents had initial low calcium levels but normal vitamin D levels. “For too long we’ve been stuck on the idea that it is [increasing] vitamin D in the elderly that causes a reduction in fractures,” said Dr. Rosen. “The data are not very supportive of it, but people continue to think that’s the most important element.”
On the other hand, the current study raises certain questions. “What we don’t know is, is it the calcium, or is it the protein, or the combination, that had an impact?”
Would upping dairy decrease falls?
Older adults living in institutions have a high risk of falls and fractures, including hip fractures, and “malnutrition is common,” said Dr. Iuliano during her presentation.
Prior studies have reported that such residents have a daily dietary calcium intake of 635 mg (half the recommended 1,300 mg), a protein intake of 0.8 g/kg body weight (less than the recommended 1 g/kg body weight), and a dairy intake of 1.5 servings (about a third of the recommended amount), she said.
The group hypothesized that upping dairy intake of elderly residents living in long-term care institutions would reduce the risk of fractures. They performed a 2-year cluster-randomized trial in 60 facilities in Melbourne and surrounding areas.
Half gave their 3,301 residents menus with a higher dairy content, and the other half gave their 3,894 residents (controls) the usual menus.
The residents in both groups had similar characteristics: they were a mean age of 87 years and 68% were women. A subgroup had blood tests and bone morphology studies at baseline and 1 year.
Researchers verified nutrient intake by analyzing the menus and doing plate waste analysis for a subgroup, and they determined the number of falls and fractures from incident and hospital x-ray reports, respectively.
One-third fewer fractures in the higher-dairy group
At the study start, residents in both groups had similar vitamin D levels (72 nmol/L) and bone morphology. They were consuming two servings of dairy food and drink a day, where a serving was 250 mL of milk (including lactose-free milk) or 200 g of yogurt or 40 g of cheese.
Their initial daily calcium intake was 650 mg, which stayed the same in the control group, but increased to >1100 mg in the intervention group.
Their initial daily protein intake was around 59 g, which remained the same in the control group, but increased to about 72 grams (1.1 g/kg body weight) in the intervention group.
At 2 years, the 1.5 servings/day increase in dairy intake in the control versus intervention group was associated with an 11% reduction in falls (62% vs. 57%), a 33% reduction in fractures (5.2% vs. 3.7%), a 46% reduction in hip fractures (2.4% vs. 1.3%), and no difference in mortality (28% in both groups).
The intervention was also associated with a slowing in bone loss and an increase in insulinlike growth factor–1.
Four dairy servings a day “is high”
Dr. Willett said that “it is reasonable for seniors to take one or two servings of dairy per day, but four servings per day, as in this study, is probably not necessary.”
Moreover, “dairy production has a major impact on greenhouse gas emissions, and even two servings per day would not be environmentally sustainable if everyone were to consume this amount,” he observed.
“Because the world is facing an existential threat from climate change, general advice to consume high amounts of dairy products would be irresponsible as we can get all essential nutrients from other sources,” he added. “That said, modest amounts of dairy foods, such as one to two servings per day could be reasonable. There is some suggestive evidence that dairy in the form of yogurt may have particular benefits.”
The study was funded by Melbourne University and various dietary councils. Dr. Iuliano reported receiving lecture fees from Abbott. Dr. Rosen and Dr. Willett reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Consuming more milk, cheese, or yogurt might be a simple, low-cost way to boost bone health and prevent some falls and fractures in older people living in long-term care facilities, according to a new randomized study from Australia.
“Supplementation using dairy foods is likely to be an effective, safe, widely available, and low cost means of curtailing the public health burden of fractures,” said Sandra Iuliano, PhD, from the University of Melbourne, who presented the findings during the virtual American Society of Bone and Mineral Research 2020 annual meeting.
The researchers randomized 60 old-age institutions to provide residents with their usual menus or a diet with more milk, cheese, or yogurt for 2 years.
The residents with the altered menus increased their dairy consumption from 2 servings/day to 3.5 servings/day, which was reflected in a greater intake of calcium and protein, along with fewer falls, total fractures, and hip fractures than in the control group.
“This is the first randomized trial to show a benefit of dairy food intake on risk of fractures,” Walter Willett, MD, DrPH, professor of nutrition and epidemiology at the Harvard School of Public Health, Boston, said in an interview.
The results are “not surprising” because supplements of calcium plus vitamin D have reduced the risk of fractures in a similar population of older residents living in special living facilities, said Dr. Willett, coauthor of a recent review article, “Milk and Health,” published in the New England Journal of Medicine.
“It is important for everyone to have adequate intake of calcium and vitamin D,” he said. However, “it isn’t clear whether it is better to ensure this clinically by supplements, overall healthy diet, or extra dairy intake,” he added, noting that consuming the amount of dairy given in this Australian study is not environmentally sustainable.
Clifford Rosen, MD, professor of medicine, Tufts University, Boston, said in an interview that the Australian researchers studied the impact of increased dietary calcium and protein, not the impact of vitamin D via supplements.
“This is progress toward getting interventions to our most needy residents to prevent fractures – probably the most compelling data that we have had in a number of years,” he noted.
The current study shows “it’s not [the] vitamin D,” because the residents had initial low calcium levels but normal vitamin D levels. “For too long we’ve been stuck on the idea that it is [increasing] vitamin D in the elderly that causes a reduction in fractures,” said Dr. Rosen. “The data are not very supportive of it, but people continue to think that’s the most important element.”
On the other hand, the current study raises certain questions. “What we don’t know is, is it the calcium, or is it the protein, or the combination, that had an impact?”
Would upping dairy decrease falls?
Older adults living in institutions have a high risk of falls and fractures, including hip fractures, and “malnutrition is common,” said Dr. Iuliano during her presentation.
Prior studies have reported that such residents have a daily dietary calcium intake of 635 mg (half the recommended 1,300 mg), a protein intake of 0.8 g/kg body weight (less than the recommended 1 g/kg body weight), and a dairy intake of 1.5 servings (about a third of the recommended amount), she said.
The group hypothesized that upping dairy intake of elderly residents living in long-term care institutions would reduce the risk of fractures. They performed a 2-year cluster-randomized trial in 60 facilities in Melbourne and surrounding areas.
Half gave their 3,301 residents menus with a higher dairy content, and the other half gave their 3,894 residents (controls) the usual menus.
The residents in both groups had similar characteristics: they were a mean age of 87 years and 68% were women. A subgroup had blood tests and bone morphology studies at baseline and 1 year.
Researchers verified nutrient intake by analyzing the menus and doing plate waste analysis for a subgroup, and they determined the number of falls and fractures from incident and hospital x-ray reports, respectively.
One-third fewer fractures in the higher-dairy group
At the study start, residents in both groups had similar vitamin D levels (72 nmol/L) and bone morphology. They were consuming two servings of dairy food and drink a day, where a serving was 250 mL of milk (including lactose-free milk) or 200 g of yogurt or 40 g of cheese.
Their initial daily calcium intake was 650 mg, which stayed the same in the control group, but increased to >1100 mg in the intervention group.
Their initial daily protein intake was around 59 g, which remained the same in the control group, but increased to about 72 grams (1.1 g/kg body weight) in the intervention group.
At 2 years, the 1.5 servings/day increase in dairy intake in the control versus intervention group was associated with an 11% reduction in falls (62% vs. 57%), a 33% reduction in fractures (5.2% vs. 3.7%), a 46% reduction in hip fractures (2.4% vs. 1.3%), and no difference in mortality (28% in both groups).
The intervention was also associated with a slowing in bone loss and an increase in insulinlike growth factor–1.
Four dairy servings a day “is high”
Dr. Willett said that “it is reasonable for seniors to take one or two servings of dairy per day, but four servings per day, as in this study, is probably not necessary.”
Moreover, “dairy production has a major impact on greenhouse gas emissions, and even two servings per day would not be environmentally sustainable if everyone were to consume this amount,” he observed.
“Because the world is facing an existential threat from climate change, general advice to consume high amounts of dairy products would be irresponsible as we can get all essential nutrients from other sources,” he added. “That said, modest amounts of dairy foods, such as one to two servings per day could be reasonable. There is some suggestive evidence that dairy in the form of yogurt may have particular benefits.”
The study was funded by Melbourne University and various dietary councils. Dr. Iuliano reported receiving lecture fees from Abbott. Dr. Rosen and Dr. Willett reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Consuming more milk, cheese, or yogurt might be a simple, low-cost way to boost bone health and prevent some falls and fractures in older people living in long-term care facilities, according to a new randomized study from Australia.
“Supplementation using dairy foods is likely to be an effective, safe, widely available, and low cost means of curtailing the public health burden of fractures,” said Sandra Iuliano, PhD, from the University of Melbourne, who presented the findings during the virtual American Society of Bone and Mineral Research 2020 annual meeting.
The researchers randomized 60 old-age institutions to provide residents with their usual menus or a diet with more milk, cheese, or yogurt for 2 years.
The residents with the altered menus increased their dairy consumption from 2 servings/day to 3.5 servings/day, which was reflected in a greater intake of calcium and protein, along with fewer falls, total fractures, and hip fractures than in the control group.
“This is the first randomized trial to show a benefit of dairy food intake on risk of fractures,” Walter Willett, MD, DrPH, professor of nutrition and epidemiology at the Harvard School of Public Health, Boston, said in an interview.
The results are “not surprising” because supplements of calcium plus vitamin D have reduced the risk of fractures in a similar population of older residents living in special living facilities, said Dr. Willett, coauthor of a recent review article, “Milk and Health,” published in the New England Journal of Medicine.
“It is important for everyone to have adequate intake of calcium and vitamin D,” he said. However, “it isn’t clear whether it is better to ensure this clinically by supplements, overall healthy diet, or extra dairy intake,” he added, noting that consuming the amount of dairy given in this Australian study is not environmentally sustainable.
Clifford Rosen, MD, professor of medicine, Tufts University, Boston, said in an interview that the Australian researchers studied the impact of increased dietary calcium and protein, not the impact of vitamin D via supplements.
“This is progress toward getting interventions to our most needy residents to prevent fractures – probably the most compelling data that we have had in a number of years,” he noted.
The current study shows “it’s not [the] vitamin D,” because the residents had initial low calcium levels but normal vitamin D levels. “For too long we’ve been stuck on the idea that it is [increasing] vitamin D in the elderly that causes a reduction in fractures,” said Dr. Rosen. “The data are not very supportive of it, but people continue to think that’s the most important element.”
On the other hand, the current study raises certain questions. “What we don’t know is, is it the calcium, or is it the protein, or the combination, that had an impact?”
Would upping dairy decrease falls?
Older adults living in institutions have a high risk of falls and fractures, including hip fractures, and “malnutrition is common,” said Dr. Iuliano during her presentation.
Prior studies have reported that such residents have a daily dietary calcium intake of 635 mg (half the recommended 1,300 mg), a protein intake of 0.8 g/kg body weight (less than the recommended 1 g/kg body weight), and a dairy intake of 1.5 servings (about a third of the recommended amount), she said.
The group hypothesized that upping dairy intake of elderly residents living in long-term care institutions would reduce the risk of fractures. They performed a 2-year cluster-randomized trial in 60 facilities in Melbourne and surrounding areas.
Half gave their 3,301 residents menus with a higher dairy content, and the other half gave their 3,894 residents (controls) the usual menus.
The residents in both groups had similar characteristics: they were a mean age of 87 years and 68% were women. A subgroup had blood tests and bone morphology studies at baseline and 1 year.
Researchers verified nutrient intake by analyzing the menus and doing plate waste analysis for a subgroup, and they determined the number of falls and fractures from incident and hospital x-ray reports, respectively.
One-third fewer fractures in the higher-dairy group
At the study start, residents in both groups had similar vitamin D levels (72 nmol/L) and bone morphology. They were consuming two servings of dairy food and drink a day, where a serving was 250 mL of milk (including lactose-free milk) or 200 g of yogurt or 40 g of cheese.
Their initial daily calcium intake was 650 mg, which stayed the same in the control group, but increased to >1100 mg in the intervention group.
Their initial daily protein intake was around 59 g, which remained the same in the control group, but increased to about 72 grams (1.1 g/kg body weight) in the intervention group.
At 2 years, the 1.5 servings/day increase in dairy intake in the control versus intervention group was associated with an 11% reduction in falls (62% vs. 57%), a 33% reduction in fractures (5.2% vs. 3.7%), a 46% reduction in hip fractures (2.4% vs. 1.3%), and no difference in mortality (28% in both groups).
The intervention was also associated with a slowing in bone loss and an increase in insulinlike growth factor–1.
Four dairy servings a day “is high”
Dr. Willett said that “it is reasonable for seniors to take one or two servings of dairy per day, but four servings per day, as in this study, is probably not necessary.”
Moreover, “dairy production has a major impact on greenhouse gas emissions, and even two servings per day would not be environmentally sustainable if everyone were to consume this amount,” he observed.
“Because the world is facing an existential threat from climate change, general advice to consume high amounts of dairy products would be irresponsible as we can get all essential nutrients from other sources,” he added. “That said, modest amounts of dairy foods, such as one to two servings per day could be reasonable. There is some suggestive evidence that dairy in the form of yogurt may have particular benefits.”
The study was funded by Melbourne University and various dietary councils. Dr. Iuliano reported receiving lecture fees from Abbott. Dr. Rosen and Dr. Willett reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM ASBMR 2020
Mega vitamin D harms bone in women, not men, without osteoporosis
“More is not necessarily better” when it comes to vitamin D supplements for women with adequate serum levels, new research suggests.
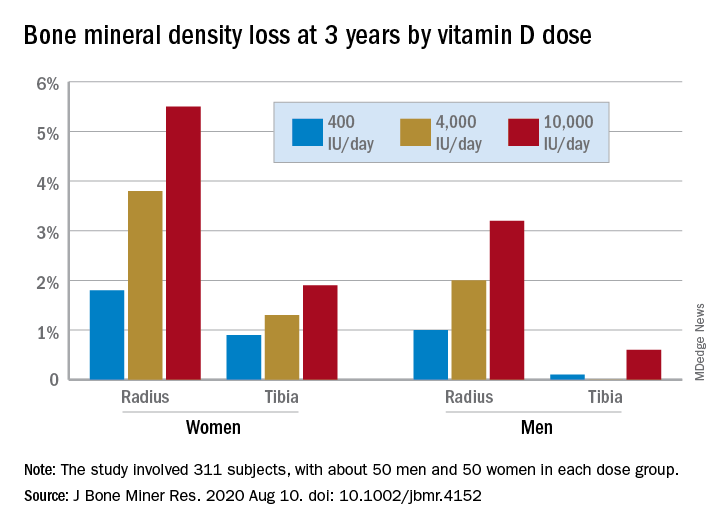
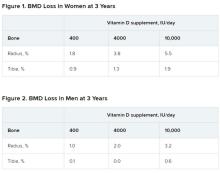
In a study of healthy 55- to 70-year-old women who took very-high-dose vitamin D supplements – either 4,000 IU/day or the previously identified “upper safe limit” of 10,000 IU/day – for 3 years had a significantly greater loss of total bone mineral density (BMD) at the radius and tibia than did women who took 400 IU/day. However, this effect was not seen in men. And the higher-dose vitamin D supplements did not improve bone strength in men or women.
But this was an exploratory post hoc analysis, and these were healthy community-dwelling adults with sufficient serum vitamin D levels (and no osteoporosis) at study entry, stressed lead researcher Lauren A. Burt, PhD, from the University of Calgary, in Alberta, Canada.
Dr. Burt presented these findings Sept. 11 at the virtual American Society of Bone and Mineral Research (ASBMR) 2020 annual meeting, and the study was also recently published online in the Journal of Bone and Mineral Research.
The results suggest that, “if you have normal bone density and adequate levels of vitamin D, there is no bone benefit in taking doses of vitamin D above the standard recommendations designed to prevent vitamin D deficiency, and doses at or above 4,000 IU/day might even be detrimental to bone, especially in females,” Dr. Burt said in an interview.
“These results are clinically relevant,” Dr. Burt and her coauthors wrote, “as vitamin D supplementation is widely administered to postmenopausal females for osteoporosis prevention.”
“Our findings do not support a benefit of high-dose vitamin D supplementation for bone health and raise the possibility of harm for females.”
Invited to comment, Meryl S. LeBoff, MD, of Harvard Medical School, Boston, said in an interview that this finding “warrants further research” because it is “important” to discover sex differences in bone responses to vitamin D.
“This doesn’t apply to osteoporosis”
Dr. LeBoff was lead author of a subanalysis of the Vitamin D and Omega-3 Trial (VITAL).
As she reported at last year’s ASBMR meeting, that analysis showed that, in healthy adults who did not have vitamin D insufficiency, taking vitamin D3 supplements for 2 years did not improve BMD, compared with placebo (recently published), nor was this linked with fewer fractures.
Dr. LeBoff pointed out that the current study investigated “very high doses of vitamin D” – at least double the 2,000 IU/day doses examined in VITAL.
Also, the serum vitamin D levels in this study were “above what we considered the upper normal limit for our assay in our hospital,” she noted, and there was no placebo control.
“We did not see any adverse effects of 2,000 IU/day vitamin D,” Dr. LeBoff stressed.
“At the same time, we didn’t see any significant benefits in terms of bone density because they already had achieved a normal level of vitamin D sufficient for bone.”
But “this doesn’t apply to patients with vitamin D deficiency, patients with osteoporosis, or low bone mass, in which case we would recommend vitamin D.”
Some patients take more vitamin D than they need because they think more is better, said LeBoff, but this study suggests “more is not necessarily better.”
“There’s been a concern for several years that too much vitamin D may be associated with increased fractures,” she emphasized.
Post hoc analysis
The current study analyzed new data from the Calgary Vitamin D study.
That study found no benefit in BMD or bone strength (JAMA. 2019;322[8]:736-45), contrary to the researchers’ hypothesis that high-dose vitamin D supplements would be associated with greater calcium absorption and parathyroid hormone suppression and, thus, reduced age-related bone loss (improved bone density and strength).
Instead, they found a negative dose-response relationship, which “should be regarded as hypothesis generating, requiring confirmation with further research,” they wrote.
The current study sought to determine if there were sex differences in the effect of vitamin D supplements on bone health in this population.
From October 2013 to December 2017, the Canada Vitamin D study enrolled 311 participants (53% male). To be eligible for the study, participants had to have serum 25-hydroxyvitamin D levels greater than 30 nmol/L and less than 125 nmol/L. They also needed to have adequate calcium intake (1,200 mg/day, as defined by the U.S. Institute of Medicine), or if not, they were instructed to take an appropriate calcium supplement dose.
Patients were randomized to receive 400, 4,000, or 10,000 IU/day of vitamin D3 cholecalciferol, given as 5 drops/day of liquid (Ddrops), with roughly 50 men and 50 women in each dose group.
Researchers selected the 400 IU/day dose as the comparator because the Institute of Medicine recommends a vitamin D intake of 600 IU/day for adults under age 70 years to provide the vitamin D needed for bone health. The typical Canadian diet includes 200-300 IU/day of vitamin D, so individuals would need a supplement of 400 IU/day to reach the recommended intake. The 4,000 IU/day dose is the recommended tolerable upper intake level, according to the Institute of Medicine. And the 10,000 IU/day dose is the tolerable upper intake level of vitamin D as identified in a review by Hathcock and colleagues (Am J Clin Nutr. 2007;85:6-18).
Participants underwent scans with high-resolution peripheral quantitative computed tomography (HR-pQCT) to measure total volumetric BMD at the radius and tibia at baseline, 6, 12, 24, and 36 months. Finite element analysis was used to estimate bone strength.
After 3 years, women had lost significantly more BMD at the radius after taking high-dose versus 400 IU/day of vitamin D. Losses in BMD at the tibia followed a similar trend but were smaller (Figure 1). There were no significant changes in this measure among men (Figure 2).
There were also no significant changes in bone strength among men or women.
Biological mechanism remains to be determined
Dr. LeBoff said a “possible biological explanation” for the findings is that “women, particularly when they are younger, lose more bone than men.”
“Postmenopausal females do lose bone at an accelerated rate compared with males,” Dr. Burt agreed, “but at the time the study was designed, there was no reason to believe that high-dose vitamin D supplementation would accelerate the problem.”
“The biological mechanism of the vitamin D–related bone loss needs further investigation,” Dr. Burt added, “but there are laboratory data suggesting that supraphysiologic doses of active metabolites of vitamin D may stimulate bone resorption.”
The study was funded by the Pure North S’Energy Foundation. Dr. Burt has reported no relevant financial relationships. Disclosures for the other authors are listed with the article. Dr. LeBoff has reported receiving grants from the National Institutes of Health for the VITAL analysis.
A version of this article originally appeared on Medscape.com.
“More is not necessarily better” when it comes to vitamin D supplements for women with adequate serum levels, new research suggests.

In a study of healthy 55- to 70-year-old women who took very-high-dose vitamin D supplements – either 4,000 IU/day or the previously identified “upper safe limit” of 10,000 IU/day – for 3 years had a significantly greater loss of total bone mineral density (BMD) at the radius and tibia than did women who took 400 IU/day. However, this effect was not seen in men. And the higher-dose vitamin D supplements did not improve bone strength in men or women.
But this was an exploratory post hoc analysis, and these were healthy community-dwelling adults with sufficient serum vitamin D levels (and no osteoporosis) at study entry, stressed lead researcher Lauren A. Burt, PhD, from the University of Calgary, in Alberta, Canada.
Dr. Burt presented these findings Sept. 11 at the virtual American Society of Bone and Mineral Research (ASBMR) 2020 annual meeting, and the study was also recently published online in the Journal of Bone and Mineral Research.
The results suggest that, “if you have normal bone density and adequate levels of vitamin D, there is no bone benefit in taking doses of vitamin D above the standard recommendations designed to prevent vitamin D deficiency, and doses at or above 4,000 IU/day might even be detrimental to bone, especially in females,” Dr. Burt said in an interview.
“These results are clinically relevant,” Dr. Burt and her coauthors wrote, “as vitamin D supplementation is widely administered to postmenopausal females for osteoporosis prevention.”
“Our findings do not support a benefit of high-dose vitamin D supplementation for bone health and raise the possibility of harm for females.”
Invited to comment, Meryl S. LeBoff, MD, of Harvard Medical School, Boston, said in an interview that this finding “warrants further research” because it is “important” to discover sex differences in bone responses to vitamin D.
“This doesn’t apply to osteoporosis”
Dr. LeBoff was lead author of a subanalysis of the Vitamin D and Omega-3 Trial (VITAL).
As she reported at last year’s ASBMR meeting, that analysis showed that, in healthy adults who did not have vitamin D insufficiency, taking vitamin D3 supplements for 2 years did not improve BMD, compared with placebo (recently published), nor was this linked with fewer fractures.
Dr. LeBoff pointed out that the current study investigated “very high doses of vitamin D” – at least double the 2,000 IU/day doses examined in VITAL.
Also, the serum vitamin D levels in this study were “above what we considered the upper normal limit for our assay in our hospital,” she noted, and there was no placebo control.
“We did not see any adverse effects of 2,000 IU/day vitamin D,” Dr. LeBoff stressed.
“At the same time, we didn’t see any significant benefits in terms of bone density because they already had achieved a normal level of vitamin D sufficient for bone.”
But “this doesn’t apply to patients with vitamin D deficiency, patients with osteoporosis, or low bone mass, in which case we would recommend vitamin D.”
Some patients take more vitamin D than they need because they think more is better, said LeBoff, but this study suggests “more is not necessarily better.”
“There’s been a concern for several years that too much vitamin D may be associated with increased fractures,” she emphasized.
Post hoc analysis
The current study analyzed new data from the Calgary Vitamin D study.
That study found no benefit in BMD or bone strength (JAMA. 2019;322[8]:736-45), contrary to the researchers’ hypothesis that high-dose vitamin D supplements would be associated with greater calcium absorption and parathyroid hormone suppression and, thus, reduced age-related bone loss (improved bone density and strength).
Instead, they found a negative dose-response relationship, which “should be regarded as hypothesis generating, requiring confirmation with further research,” they wrote.
The current study sought to determine if there were sex differences in the effect of vitamin D supplements on bone health in this population.
From October 2013 to December 2017, the Canada Vitamin D study enrolled 311 participants (53% male). To be eligible for the study, participants had to have serum 25-hydroxyvitamin D levels greater than 30 nmol/L and less than 125 nmol/L. They also needed to have adequate calcium intake (1,200 mg/day, as defined by the U.S. Institute of Medicine), or if not, they were instructed to take an appropriate calcium supplement dose.
Patients were randomized to receive 400, 4,000, or 10,000 IU/day of vitamin D3 cholecalciferol, given as 5 drops/day of liquid (Ddrops), with roughly 50 men and 50 women in each dose group.
Researchers selected the 400 IU/day dose as the comparator because the Institute of Medicine recommends a vitamin D intake of 600 IU/day for adults under age 70 years to provide the vitamin D needed for bone health. The typical Canadian diet includes 200-300 IU/day of vitamin D, so individuals would need a supplement of 400 IU/day to reach the recommended intake. The 4,000 IU/day dose is the recommended tolerable upper intake level, according to the Institute of Medicine. And the 10,000 IU/day dose is the tolerable upper intake level of vitamin D as identified in a review by Hathcock and colleagues (Am J Clin Nutr. 2007;85:6-18).
Participants underwent scans with high-resolution peripheral quantitative computed tomography (HR-pQCT) to measure total volumetric BMD at the radius and tibia at baseline, 6, 12, 24, and 36 months. Finite element analysis was used to estimate bone strength.
After 3 years, women had lost significantly more BMD at the radius after taking high-dose versus 400 IU/day of vitamin D. Losses in BMD at the tibia followed a similar trend but were smaller (Figure 1). There were no significant changes in this measure among men (Figure 2).
There were also no significant changes in bone strength among men or women.
Biological mechanism remains to be determined
Dr. LeBoff said a “possible biological explanation” for the findings is that “women, particularly when they are younger, lose more bone than men.”
“Postmenopausal females do lose bone at an accelerated rate compared with males,” Dr. Burt agreed, “but at the time the study was designed, there was no reason to believe that high-dose vitamin D supplementation would accelerate the problem.”
“The biological mechanism of the vitamin D–related bone loss needs further investigation,” Dr. Burt added, “but there are laboratory data suggesting that supraphysiologic doses of active metabolites of vitamin D may stimulate bone resorption.”
The study was funded by the Pure North S’Energy Foundation. Dr. Burt has reported no relevant financial relationships. Disclosures for the other authors are listed with the article. Dr. LeBoff has reported receiving grants from the National Institutes of Health for the VITAL analysis.
A version of this article originally appeared on Medscape.com.
“More is not necessarily better” when it comes to vitamin D supplements for women with adequate serum levels, new research suggests.

In a study of healthy 55- to 70-year-old women who took very-high-dose vitamin D supplements – either 4,000 IU/day or the previously identified “upper safe limit” of 10,000 IU/day – for 3 years had a significantly greater loss of total bone mineral density (BMD) at the radius and tibia than did women who took 400 IU/day. However, this effect was not seen in men. And the higher-dose vitamin D supplements did not improve bone strength in men or women.
But this was an exploratory post hoc analysis, and these were healthy community-dwelling adults with sufficient serum vitamin D levels (and no osteoporosis) at study entry, stressed lead researcher Lauren A. Burt, PhD, from the University of Calgary, in Alberta, Canada.
Dr. Burt presented these findings Sept. 11 at the virtual American Society of Bone and Mineral Research (ASBMR) 2020 annual meeting, and the study was also recently published online in the Journal of Bone and Mineral Research.
The results suggest that, “if you have normal bone density and adequate levels of vitamin D, there is no bone benefit in taking doses of vitamin D above the standard recommendations designed to prevent vitamin D deficiency, and doses at or above 4,000 IU/day might even be detrimental to bone, especially in females,” Dr. Burt said in an interview.
“These results are clinically relevant,” Dr. Burt and her coauthors wrote, “as vitamin D supplementation is widely administered to postmenopausal females for osteoporosis prevention.”
“Our findings do not support a benefit of high-dose vitamin D supplementation for bone health and raise the possibility of harm for females.”
Invited to comment, Meryl S. LeBoff, MD, of Harvard Medical School, Boston, said in an interview that this finding “warrants further research” because it is “important” to discover sex differences in bone responses to vitamin D.
“This doesn’t apply to osteoporosis”
Dr. LeBoff was lead author of a subanalysis of the Vitamin D and Omega-3 Trial (VITAL).
As she reported at last year’s ASBMR meeting, that analysis showed that, in healthy adults who did not have vitamin D insufficiency, taking vitamin D3 supplements for 2 years did not improve BMD, compared with placebo (recently published), nor was this linked with fewer fractures.
Dr. LeBoff pointed out that the current study investigated “very high doses of vitamin D” – at least double the 2,000 IU/day doses examined in VITAL.
Also, the serum vitamin D levels in this study were “above what we considered the upper normal limit for our assay in our hospital,” she noted, and there was no placebo control.
“We did not see any adverse effects of 2,000 IU/day vitamin D,” Dr. LeBoff stressed.
“At the same time, we didn’t see any significant benefits in terms of bone density because they already had achieved a normal level of vitamin D sufficient for bone.”
But “this doesn’t apply to patients with vitamin D deficiency, patients with osteoporosis, or low bone mass, in which case we would recommend vitamin D.”
Some patients take more vitamin D than they need because they think more is better, said LeBoff, but this study suggests “more is not necessarily better.”
“There’s been a concern for several years that too much vitamin D may be associated with increased fractures,” she emphasized.
Post hoc analysis
The current study analyzed new data from the Calgary Vitamin D study.
That study found no benefit in BMD or bone strength (JAMA. 2019;322[8]:736-45), contrary to the researchers’ hypothesis that high-dose vitamin D supplements would be associated with greater calcium absorption and parathyroid hormone suppression and, thus, reduced age-related bone loss (improved bone density and strength).
Instead, they found a negative dose-response relationship, which “should be regarded as hypothesis generating, requiring confirmation with further research,” they wrote.
The current study sought to determine if there were sex differences in the effect of vitamin D supplements on bone health in this population.
From October 2013 to December 2017, the Canada Vitamin D study enrolled 311 participants (53% male). To be eligible for the study, participants had to have serum 25-hydroxyvitamin D levels greater than 30 nmol/L and less than 125 nmol/L. They also needed to have adequate calcium intake (1,200 mg/day, as defined by the U.S. Institute of Medicine), or if not, they were instructed to take an appropriate calcium supplement dose.
Patients were randomized to receive 400, 4,000, or 10,000 IU/day of vitamin D3 cholecalciferol, given as 5 drops/day of liquid (Ddrops), with roughly 50 men and 50 women in each dose group.
Researchers selected the 400 IU/day dose as the comparator because the Institute of Medicine recommends a vitamin D intake of 600 IU/day for adults under age 70 years to provide the vitamin D needed for bone health. The typical Canadian diet includes 200-300 IU/day of vitamin D, so individuals would need a supplement of 400 IU/day to reach the recommended intake. The 4,000 IU/day dose is the recommended tolerable upper intake level, according to the Institute of Medicine. And the 10,000 IU/day dose is the tolerable upper intake level of vitamin D as identified in a review by Hathcock and colleagues (Am J Clin Nutr. 2007;85:6-18).
Participants underwent scans with high-resolution peripheral quantitative computed tomography (HR-pQCT) to measure total volumetric BMD at the radius and tibia at baseline, 6, 12, 24, and 36 months. Finite element analysis was used to estimate bone strength.
After 3 years, women had lost significantly more BMD at the radius after taking high-dose versus 400 IU/day of vitamin D. Losses in BMD at the tibia followed a similar trend but were smaller (Figure 1). There were no significant changes in this measure among men (Figure 2).
There were also no significant changes in bone strength among men or women.
Biological mechanism remains to be determined
Dr. LeBoff said a “possible biological explanation” for the findings is that “women, particularly when they are younger, lose more bone than men.”
“Postmenopausal females do lose bone at an accelerated rate compared with males,” Dr. Burt agreed, “but at the time the study was designed, there was no reason to believe that high-dose vitamin D supplementation would accelerate the problem.”
“The biological mechanism of the vitamin D–related bone loss needs further investigation,” Dr. Burt added, “but there are laboratory data suggesting that supraphysiologic doses of active metabolites of vitamin D may stimulate bone resorption.”
The study was funded by the Pure North S’Energy Foundation. Dr. Burt has reported no relevant financial relationships. Disclosures for the other authors are listed with the article. Dr. LeBoff has reported receiving grants from the National Institutes of Health for the VITAL analysis.
A version of this article originally appeared on Medscape.com.
More U.S. states cap insulin cost, but activists will ‘fight harder’
Twelve U.S. states have now passed laws aimed at making insulin more affordable – and more than 30 are considering such legislation – but they all have gaps that still put the cost of this basic and essential medication out of reach for many with diabetes.
The laws only apply to health insurance through state-regulated plans, and not to the majority of health plans that cover most Americans: Medicare, Medicaid, the Veterans Affairs health system, or self-funded employer-sponsored plans.
Overall, Hannah Crabtree, an activist who writes the blog Data for Insulin, estimates state laws that limit copays, deductibles, or other out-of-pocket costs for insulin cover an average of 27% of people with diabetes across the United States.
And while diabetes activists have applauded state actions, most want more help for the under- and uninsured.
“Our chapter will be fighting harder next legislative session for the uninsured,” said Mindie Hooley, the leader of the Utah #insulin4all chapter, which successfully lobbied legislators to pass a bill signed by the state’s governor on March 30.
“With so many losing their jobs because of the pandemic, there’s no better time than now to fight for these patients who don’t have insurance,” Ms. Hooley said in an interview.
The American Diabetes Association has also been lobbying for state caps as one of many avenues for making insulin more affordable, said Stephen Habbe, the ADA’s director for state government affairs.
One in four insulin users report rationing the medication, Mr. Habbe said.
The state laws “can really provide important relief in terms of affordability for their insulin costs, which we know can be critical in terms of preserving their life and helping to prevent complications that can potentially be disabling or even deadly,” he said in an interview.
Activists with T1 International, which created the #insulin4all campaign, are working nationwide to convince state legislators to back measures that limit out-of-pocket costs for insulin, or for other diabetes medications and supplies.
Colorado, Connecticut, Delaware, Illinois, Maine, New Hampshire, New Mexico, New York, Utah, Virginia, Washington, and West Virginia have enacted such limits, with caps ranging from $25 to $100.
Insulin makers unfazed, blame insurers, PBMs for high prices
The three insulin manufacturers in the United States – Eli Lilly, Novo Nordisk, and Sanofi– have not overtly fought against the laws, although in July, the Pharmaceutical Research and Manufacturers of America did sue to block a related Minnesota law that provides a free emergency supply of insulin.
And the nonprofit news organization FairWarning reported in August that a lobbyist from Eli Lilly had attempted to push a Tennessee legislator to keep the uninsured from being eligible for any out-of-pocket limits.
The insulin makers have also not lowered prices in response to the mounting number of state laws.
They see no need, said Tara O’Neill Hayes, director of human welfare policy at the American Action Forum, a center right–leaning Washington, D.C., think tank.
“You’re going to do what you can get away with,” Ms. O’Neill Hayes said in an interview. “To the extent that they can keep their prices high and people are still buying, they have limited incentives to lower those costs.”
The insulin market is dysfunctional, she added. “The increasing cost of insulin seems primarily to be the result of a lack of competition in the market and convoluted drug pricing and insurance practices,” Ms. O’Neill Hayes and colleagues wrote in a report in April on federal and state attempts to address insulin affordability.
Novo Nordisk, however, maintains that drugmakers are not solely to blame.
“Everyone in the health care system has a role to play in affordability,” said Ken Inchausti, Novo Nordisk’s senior director for corporate communications. State legislation “attempts to address a systemic issue in [U.S.] health care: How benefit design can make medicines unaffordable for many, especially for those in high-deductible health plans,” he said in an interview.
“Efforts to place copay caps on insurance plans covering insulin can certainly help lower out-of-pocket costs,” said Mr. Inchausti.
Sanofi spokesperson Jon Florio said the company supports actions that increase affordable access to insulin. However, “while we support capped copays, we feel this should not be limited to just one class of medicines,” he said. Mr. Florio also noted that Sanofi provides out-of-pocket caps to anyone with commercial insurance and that anyone without insurance can buy one or multiple Sanofi insulins for a fixed price of $99 per month, up to 10 boxes of pens and/or 10-mL vials.
And Sanofi will take part in the Centers for Medicare & Medicaid Services’ new insulin demonstration program. Starting in 2021, CMS will cap insulin copays at $35 for people in Part D plans that participate.
Eli Lilly spokesperson Brad Jacklin said the company “believes in the common goal of ensuring affordable access to insulin and other life-saving medicines because nobody should have to forgo or ration because of cost.”
Lilly supports efforts “that more directly affect patients’ cost-sharing based on their health care coverage,” he said. Insurers and pharmacy benefit managers (PBMs) should pass savings on to patients, Mr. Jacklin urged. Lilly caps some insulins at $35 for the uninsured or commercially insured. The company will also participate in the CMS program.
Meanwhile, a PhRMA-sponsored website www.letstalkaboutcost.org said that, because they do not share savings, insurers and PBMs are responsible for high insulin costs.
Manufacturer assistance programs for patients with diabetes and other chronic diseases, on the other hand, can save individuals $300-$500 a year, PhRMA said in August.
PBMs point back at insulin manufacturers
PBMs, however, point back at drug companies. “PBMs have been able to moderate insulin costs for most consumers with insurance,” said J.C. Scott, president and CEO of the Pharmaceutical Care Management Association, the PBM trade group, in a statement.
The rising cost of insulin is caused by a lack of competition and overuse of patent extensions, PCMA maintains.
Health insurers, which, in tandem with PBMs, give insulins formulary preference based on a discounted price, are most likely to feel the impact of laws limiting out-of-pocket costs.
If they have to make up the shortfall from a patient’s reduced payment for a prescription, they will likely raise premiums, said Ms. O’Neill Hayes.
And if patients pay the same price for insulin – regardless of who makes it – drugmakers won’t have much incentive to offer discounts or rebates for formulary placement, she said. Again, that would likely lead to higher premiums.
David Allen, a spokesperson for America’s Health Insurance Plans, said in an interview that AHIP believes lack of competition has driven up insulin prices.
“High prices for insulin correspond with high health insurance costs for insulin,” he said. When CMS starts requiring drugmakers to discount their insulins for Medicare that will allow “health plans to use those savings to reduce out-of-pocket [costs] for seniors.”
He did not respond to a question as to why health insurers were not already passing savings on to commercially insured patients, especially in states with out-of-pocket limits.
Mr. Allen did say that AHIP’s plans “stand ready to work with state policymakers to remove barriers to lower insulin prices for Americans.”
Utah savings hopefully saving lives already
In Utah, legislators tuned out the blame game, and instead were keen to listen to patients, who had many stories about how the high cost of insulin had hurt them, said Ms. Hooley.
She noted an estimated 50,000 Utahans rely on insulin to stay alive.
Ms. Hooley and her chapter convinced legislators to pass a bill that gives insurers the option to cap patient copays at $30 per month, or to put insulin on its lowest formulary tier and waive any patient deductible. That aspect of the law does not go into effect until January 2021, but insurers are already starting to move insulin to the lowest formulary tier.
That has helped some people immediately. One state resident said her most recent insulin prescription cost $7 – instead of the usual $200.
The uninsured are not left totally high and dry either. Starting June 1, anyone in the state could buy through a state bulk-purchasing program, which guaranteed a 60% discount.
Ms. Hooley said she’d recently heard about a patient who usually spent $300 per prescription but was able to buy insulin for $100 through the program.
“Although $100 is still too much, it is nice knowing the Utah Insulin Savings Program is saving lives,” Ms. Hooley concluded.
A version of this article originally appeared on Medscape.com.
Twelve U.S. states have now passed laws aimed at making insulin more affordable – and more than 30 are considering such legislation – but they all have gaps that still put the cost of this basic and essential medication out of reach for many with diabetes.
The laws only apply to health insurance through state-regulated plans, and not to the majority of health plans that cover most Americans: Medicare, Medicaid, the Veterans Affairs health system, or self-funded employer-sponsored plans.
Overall, Hannah Crabtree, an activist who writes the blog Data for Insulin, estimates state laws that limit copays, deductibles, or other out-of-pocket costs for insulin cover an average of 27% of people with diabetes across the United States.
And while diabetes activists have applauded state actions, most want more help for the under- and uninsured.
“Our chapter will be fighting harder next legislative session for the uninsured,” said Mindie Hooley, the leader of the Utah #insulin4all chapter, which successfully lobbied legislators to pass a bill signed by the state’s governor on March 30.
“With so many losing their jobs because of the pandemic, there’s no better time than now to fight for these patients who don’t have insurance,” Ms. Hooley said in an interview.
The American Diabetes Association has also been lobbying for state caps as one of many avenues for making insulin more affordable, said Stephen Habbe, the ADA’s director for state government affairs.
One in four insulin users report rationing the medication, Mr. Habbe said.
The state laws “can really provide important relief in terms of affordability for their insulin costs, which we know can be critical in terms of preserving their life and helping to prevent complications that can potentially be disabling or even deadly,” he said in an interview.
Activists with T1 International, which created the #insulin4all campaign, are working nationwide to convince state legislators to back measures that limit out-of-pocket costs for insulin, or for other diabetes medications and supplies.
Colorado, Connecticut, Delaware, Illinois, Maine, New Hampshire, New Mexico, New York, Utah, Virginia, Washington, and West Virginia have enacted such limits, with caps ranging from $25 to $100.
Insulin makers unfazed, blame insurers, PBMs for high prices
The three insulin manufacturers in the United States – Eli Lilly, Novo Nordisk, and Sanofi– have not overtly fought against the laws, although in July, the Pharmaceutical Research and Manufacturers of America did sue to block a related Minnesota law that provides a free emergency supply of insulin.
And the nonprofit news organization FairWarning reported in August that a lobbyist from Eli Lilly had attempted to push a Tennessee legislator to keep the uninsured from being eligible for any out-of-pocket limits.
The insulin makers have also not lowered prices in response to the mounting number of state laws.
They see no need, said Tara O’Neill Hayes, director of human welfare policy at the American Action Forum, a center right–leaning Washington, D.C., think tank.
“You’re going to do what you can get away with,” Ms. O’Neill Hayes said in an interview. “To the extent that they can keep their prices high and people are still buying, they have limited incentives to lower those costs.”
The insulin market is dysfunctional, she added. “The increasing cost of insulin seems primarily to be the result of a lack of competition in the market and convoluted drug pricing and insurance practices,” Ms. O’Neill Hayes and colleagues wrote in a report in April on federal and state attempts to address insulin affordability.
Novo Nordisk, however, maintains that drugmakers are not solely to blame.
“Everyone in the health care system has a role to play in affordability,” said Ken Inchausti, Novo Nordisk’s senior director for corporate communications. State legislation “attempts to address a systemic issue in [U.S.] health care: How benefit design can make medicines unaffordable for many, especially for those in high-deductible health plans,” he said in an interview.
“Efforts to place copay caps on insurance plans covering insulin can certainly help lower out-of-pocket costs,” said Mr. Inchausti.
Sanofi spokesperson Jon Florio said the company supports actions that increase affordable access to insulin. However, “while we support capped copays, we feel this should not be limited to just one class of medicines,” he said. Mr. Florio also noted that Sanofi provides out-of-pocket caps to anyone with commercial insurance and that anyone without insurance can buy one or multiple Sanofi insulins for a fixed price of $99 per month, up to 10 boxes of pens and/or 10-mL vials.
And Sanofi will take part in the Centers for Medicare & Medicaid Services’ new insulin demonstration program. Starting in 2021, CMS will cap insulin copays at $35 for people in Part D plans that participate.
Eli Lilly spokesperson Brad Jacklin said the company “believes in the common goal of ensuring affordable access to insulin and other life-saving medicines because nobody should have to forgo or ration because of cost.”
Lilly supports efforts “that more directly affect patients’ cost-sharing based on their health care coverage,” he said. Insurers and pharmacy benefit managers (PBMs) should pass savings on to patients, Mr. Jacklin urged. Lilly caps some insulins at $35 for the uninsured or commercially insured. The company will also participate in the CMS program.
Meanwhile, a PhRMA-sponsored website www.letstalkaboutcost.org said that, because they do not share savings, insurers and PBMs are responsible for high insulin costs.
Manufacturer assistance programs for patients with diabetes and other chronic diseases, on the other hand, can save individuals $300-$500 a year, PhRMA said in August.
PBMs point back at insulin manufacturers
PBMs, however, point back at drug companies. “PBMs have been able to moderate insulin costs for most consumers with insurance,” said J.C. Scott, president and CEO of the Pharmaceutical Care Management Association, the PBM trade group, in a statement.
The rising cost of insulin is caused by a lack of competition and overuse of patent extensions, PCMA maintains.
Health insurers, which, in tandem with PBMs, give insulins formulary preference based on a discounted price, are most likely to feel the impact of laws limiting out-of-pocket costs.
If they have to make up the shortfall from a patient’s reduced payment for a prescription, they will likely raise premiums, said Ms. O’Neill Hayes.
And if patients pay the same price for insulin – regardless of who makes it – drugmakers won’t have much incentive to offer discounts or rebates for formulary placement, she said. Again, that would likely lead to higher premiums.
David Allen, a spokesperson for America’s Health Insurance Plans, said in an interview that AHIP believes lack of competition has driven up insulin prices.
“High prices for insulin correspond with high health insurance costs for insulin,” he said. When CMS starts requiring drugmakers to discount their insulins for Medicare that will allow “health plans to use those savings to reduce out-of-pocket [costs] for seniors.”
He did not respond to a question as to why health insurers were not already passing savings on to commercially insured patients, especially in states with out-of-pocket limits.
Mr. Allen did say that AHIP’s plans “stand ready to work with state policymakers to remove barriers to lower insulin prices for Americans.”
Utah savings hopefully saving lives already
In Utah, legislators tuned out the blame game, and instead were keen to listen to patients, who had many stories about how the high cost of insulin had hurt them, said Ms. Hooley.
She noted an estimated 50,000 Utahans rely on insulin to stay alive.
Ms. Hooley and her chapter convinced legislators to pass a bill that gives insurers the option to cap patient copays at $30 per month, or to put insulin on its lowest formulary tier and waive any patient deductible. That aspect of the law does not go into effect until January 2021, but insurers are already starting to move insulin to the lowest formulary tier.
That has helped some people immediately. One state resident said her most recent insulin prescription cost $7 – instead of the usual $200.
The uninsured are not left totally high and dry either. Starting June 1, anyone in the state could buy through a state bulk-purchasing program, which guaranteed a 60% discount.
Ms. Hooley said she’d recently heard about a patient who usually spent $300 per prescription but was able to buy insulin for $100 through the program.
“Although $100 is still too much, it is nice knowing the Utah Insulin Savings Program is saving lives,” Ms. Hooley concluded.
A version of this article originally appeared on Medscape.com.
Twelve U.S. states have now passed laws aimed at making insulin more affordable – and more than 30 are considering such legislation – but they all have gaps that still put the cost of this basic and essential medication out of reach for many with diabetes.
The laws only apply to health insurance through state-regulated plans, and not to the majority of health plans that cover most Americans: Medicare, Medicaid, the Veterans Affairs health system, or self-funded employer-sponsored plans.
Overall, Hannah Crabtree, an activist who writes the blog Data for Insulin, estimates state laws that limit copays, deductibles, or other out-of-pocket costs for insulin cover an average of 27% of people with diabetes across the United States.
And while diabetes activists have applauded state actions, most want more help for the under- and uninsured.
“Our chapter will be fighting harder next legislative session for the uninsured,” said Mindie Hooley, the leader of the Utah #insulin4all chapter, which successfully lobbied legislators to pass a bill signed by the state’s governor on March 30.
“With so many losing their jobs because of the pandemic, there’s no better time than now to fight for these patients who don’t have insurance,” Ms. Hooley said in an interview.
The American Diabetes Association has also been lobbying for state caps as one of many avenues for making insulin more affordable, said Stephen Habbe, the ADA’s director for state government affairs.
One in four insulin users report rationing the medication, Mr. Habbe said.
The state laws “can really provide important relief in terms of affordability for their insulin costs, which we know can be critical in terms of preserving their life and helping to prevent complications that can potentially be disabling or even deadly,” he said in an interview.
Activists with T1 International, which created the #insulin4all campaign, are working nationwide to convince state legislators to back measures that limit out-of-pocket costs for insulin, or for other diabetes medications and supplies.
Colorado, Connecticut, Delaware, Illinois, Maine, New Hampshire, New Mexico, New York, Utah, Virginia, Washington, and West Virginia have enacted such limits, with caps ranging from $25 to $100.
Insulin makers unfazed, blame insurers, PBMs for high prices
The three insulin manufacturers in the United States – Eli Lilly, Novo Nordisk, and Sanofi– have not overtly fought against the laws, although in July, the Pharmaceutical Research and Manufacturers of America did sue to block a related Minnesota law that provides a free emergency supply of insulin.
And the nonprofit news organization FairWarning reported in August that a lobbyist from Eli Lilly had attempted to push a Tennessee legislator to keep the uninsured from being eligible for any out-of-pocket limits.
The insulin makers have also not lowered prices in response to the mounting number of state laws.
They see no need, said Tara O’Neill Hayes, director of human welfare policy at the American Action Forum, a center right–leaning Washington, D.C., think tank.
“You’re going to do what you can get away with,” Ms. O’Neill Hayes said in an interview. “To the extent that they can keep their prices high and people are still buying, they have limited incentives to lower those costs.”
The insulin market is dysfunctional, she added. “The increasing cost of insulin seems primarily to be the result of a lack of competition in the market and convoluted drug pricing and insurance practices,” Ms. O’Neill Hayes and colleagues wrote in a report in April on federal and state attempts to address insulin affordability.
Novo Nordisk, however, maintains that drugmakers are not solely to blame.
“Everyone in the health care system has a role to play in affordability,” said Ken Inchausti, Novo Nordisk’s senior director for corporate communications. State legislation “attempts to address a systemic issue in [U.S.] health care: How benefit design can make medicines unaffordable for many, especially for those in high-deductible health plans,” he said in an interview.
“Efforts to place copay caps on insurance plans covering insulin can certainly help lower out-of-pocket costs,” said Mr. Inchausti.
Sanofi spokesperson Jon Florio said the company supports actions that increase affordable access to insulin. However, “while we support capped copays, we feel this should not be limited to just one class of medicines,” he said. Mr. Florio also noted that Sanofi provides out-of-pocket caps to anyone with commercial insurance and that anyone without insurance can buy one or multiple Sanofi insulins for a fixed price of $99 per month, up to 10 boxes of pens and/or 10-mL vials.
And Sanofi will take part in the Centers for Medicare & Medicaid Services’ new insulin demonstration program. Starting in 2021, CMS will cap insulin copays at $35 for people in Part D plans that participate.
Eli Lilly spokesperson Brad Jacklin said the company “believes in the common goal of ensuring affordable access to insulin and other life-saving medicines because nobody should have to forgo or ration because of cost.”
Lilly supports efforts “that more directly affect patients’ cost-sharing based on their health care coverage,” he said. Insurers and pharmacy benefit managers (PBMs) should pass savings on to patients, Mr. Jacklin urged. Lilly caps some insulins at $35 for the uninsured or commercially insured. The company will also participate in the CMS program.
Meanwhile, a PhRMA-sponsored website www.letstalkaboutcost.org said that, because they do not share savings, insurers and PBMs are responsible for high insulin costs.
Manufacturer assistance programs for patients with diabetes and other chronic diseases, on the other hand, can save individuals $300-$500 a year, PhRMA said in August.
PBMs point back at insulin manufacturers
PBMs, however, point back at drug companies. “PBMs have been able to moderate insulin costs for most consumers with insurance,” said J.C. Scott, president and CEO of the Pharmaceutical Care Management Association, the PBM trade group, in a statement.
The rising cost of insulin is caused by a lack of competition and overuse of patent extensions, PCMA maintains.
Health insurers, which, in tandem with PBMs, give insulins formulary preference based on a discounted price, are most likely to feel the impact of laws limiting out-of-pocket costs.
If they have to make up the shortfall from a patient’s reduced payment for a prescription, they will likely raise premiums, said Ms. O’Neill Hayes.
And if patients pay the same price for insulin – regardless of who makes it – drugmakers won’t have much incentive to offer discounts or rebates for formulary placement, she said. Again, that would likely lead to higher premiums.
David Allen, a spokesperson for America’s Health Insurance Plans, said in an interview that AHIP believes lack of competition has driven up insulin prices.
“High prices for insulin correspond with high health insurance costs for insulin,” he said. When CMS starts requiring drugmakers to discount their insulins for Medicare that will allow “health plans to use those savings to reduce out-of-pocket [costs] for seniors.”
He did not respond to a question as to why health insurers were not already passing savings on to commercially insured patients, especially in states with out-of-pocket limits.
Mr. Allen did say that AHIP’s plans “stand ready to work with state policymakers to remove barriers to lower insulin prices for Americans.”
Utah savings hopefully saving lives already
In Utah, legislators tuned out the blame game, and instead were keen to listen to patients, who had many stories about how the high cost of insulin had hurt them, said Ms. Hooley.
She noted an estimated 50,000 Utahans rely on insulin to stay alive.
Ms. Hooley and her chapter convinced legislators to pass a bill that gives insurers the option to cap patient copays at $30 per month, or to put insulin on its lowest formulary tier and waive any patient deductible. That aspect of the law does not go into effect until January 2021, but insurers are already starting to move insulin to the lowest formulary tier.
That has helped some people immediately. One state resident said her most recent insulin prescription cost $7 – instead of the usual $200.
The uninsured are not left totally high and dry either. Starting June 1, anyone in the state could buy through a state bulk-purchasing program, which guaranteed a 60% discount.
Ms. Hooley said she’d recently heard about a patient who usually spent $300 per prescription but was able to buy insulin for $100 through the program.
“Although $100 is still too much, it is nice knowing the Utah Insulin Savings Program is saving lives,” Ms. Hooley concluded.
A version of this article originally appeared on Medscape.com.
FDA grants approval to weekly growth hormone for adults
The human growth hormone formulation somapacitan for adults with growth hormone deficiency was approved by the Food and Drug Administration on Sept. 1. .
Somapacitan contains an albumin-binding element attached to the growth hormone, causing the reversible binding to albumin proteins in the body. This reduces clearance and increases the half-life of the hormone. The formulation has previous demonstrated safety and efficacy in children with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgz310).
Growth hormone treatment can counter abdominal obesity, reduced lean body mass, fatigue, osteopenia, cardiovascular risks, and other manifestations of growth hormone deficiency in adults, but daily injections can be burdensome for patients. That makes long-acting versions attractive, but the lifelong nature of the treatment makes it important to characterize safety and tolerability.
The approval comes on the strength of a randomized, placebo-controlled phase 3 trial (REAL 1) of 300 adult patients in 17 countries with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgaa049). Participants had either never received growth hormone treatment, or had stopped taking one at least 6 months before starting the trial. Subjects received once-weekly somapacitan, once-weekly placebo, or daily somatropin, which is FDA approved.
The primary endpoint was percentage change of truncal fat, which is regulated by growth hormone, and can lead to medical problems. After 34 weeks, subjects in the somapacitan group experienced a 1.06% decrease in truncal fat, compared with a 0.47% increase in the placebo group (P = .009) and a 2.23% decrease in the daily somatropin group.
After 34 weeks, a 52-week extension trial began. The somapacitan group continued on the drug and the placebo group was offered somapacitan. Patients on daily somatropin were randomized to continue daily treatment with somatropin or to switch to somapacitan.
At the end of the extension trial, those taking somapacitan for the full 86-week duration had an average reduction of 1.52% in truncal fat. After 86 weeks, the somapacitan and daily somatropin groups had similar values for percentage change in visceral fat, lean body mass, or appendicular skeletal muscle mass.
Common side effects of somapacitan were back pain, joint paint, indigestion, a sleep disorder, dizziness, tonsillitis, swelling in the arms or lower legs, vomiting, adrenal insufficiency, hypertension, increase in blood creatine phosphokinase, weight increase, and anemia.
Somapacitan, marketed as Sogroya by Novo Nordisk, is contraindicated in patients with an allergy to the drug, as well as those with an active malignancy, diabetic eye disease where increases in blood sugars could lead to retinal damage, acute critical illness, or acute respiratory failure.
The FDA recommends that providers perform an eye examination before drug initiation, as well as periodically while the patient is taking the drug, to rule out preexisting papilledema. This could be a sign of intracranial hypertension, which could be caused or worsened by growth hormones.
The human growth hormone formulation somapacitan for adults with growth hormone deficiency was approved by the Food and Drug Administration on Sept. 1. .
Somapacitan contains an albumin-binding element attached to the growth hormone, causing the reversible binding to albumin proteins in the body. This reduces clearance and increases the half-life of the hormone. The formulation has previous demonstrated safety and efficacy in children with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgz310).
Growth hormone treatment can counter abdominal obesity, reduced lean body mass, fatigue, osteopenia, cardiovascular risks, and other manifestations of growth hormone deficiency in adults, but daily injections can be burdensome for patients. That makes long-acting versions attractive, but the lifelong nature of the treatment makes it important to characterize safety and tolerability.
The approval comes on the strength of a randomized, placebo-controlled phase 3 trial (REAL 1) of 300 adult patients in 17 countries with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgaa049). Participants had either never received growth hormone treatment, or had stopped taking one at least 6 months before starting the trial. Subjects received once-weekly somapacitan, once-weekly placebo, or daily somatropin, which is FDA approved.
The primary endpoint was percentage change of truncal fat, which is regulated by growth hormone, and can lead to medical problems. After 34 weeks, subjects in the somapacitan group experienced a 1.06% decrease in truncal fat, compared with a 0.47% increase in the placebo group (P = .009) and a 2.23% decrease in the daily somatropin group.
After 34 weeks, a 52-week extension trial began. The somapacitan group continued on the drug and the placebo group was offered somapacitan. Patients on daily somatropin were randomized to continue daily treatment with somatropin or to switch to somapacitan.
At the end of the extension trial, those taking somapacitan for the full 86-week duration had an average reduction of 1.52% in truncal fat. After 86 weeks, the somapacitan and daily somatropin groups had similar values for percentage change in visceral fat, lean body mass, or appendicular skeletal muscle mass.
Common side effects of somapacitan were back pain, joint paint, indigestion, a sleep disorder, dizziness, tonsillitis, swelling in the arms or lower legs, vomiting, adrenal insufficiency, hypertension, increase in blood creatine phosphokinase, weight increase, and anemia.
Somapacitan, marketed as Sogroya by Novo Nordisk, is contraindicated in patients with an allergy to the drug, as well as those with an active malignancy, diabetic eye disease where increases in blood sugars could lead to retinal damage, acute critical illness, or acute respiratory failure.
The FDA recommends that providers perform an eye examination before drug initiation, as well as periodically while the patient is taking the drug, to rule out preexisting papilledema. This could be a sign of intracranial hypertension, which could be caused or worsened by growth hormones.
The human growth hormone formulation somapacitan for adults with growth hormone deficiency was approved by the Food and Drug Administration on Sept. 1. .
Somapacitan contains an albumin-binding element attached to the growth hormone, causing the reversible binding to albumin proteins in the body. This reduces clearance and increases the half-life of the hormone. The formulation has previous demonstrated safety and efficacy in children with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgz310).
Growth hormone treatment can counter abdominal obesity, reduced lean body mass, fatigue, osteopenia, cardiovascular risks, and other manifestations of growth hormone deficiency in adults, but daily injections can be burdensome for patients. That makes long-acting versions attractive, but the lifelong nature of the treatment makes it important to characterize safety and tolerability.
The approval comes on the strength of a randomized, placebo-controlled phase 3 trial (REAL 1) of 300 adult patients in 17 countries with growth hormone deficiency (J Clin Endocrinol Metab. 2020 Apr 1. doi: 10.1210/clinem/dgaa049). Participants had either never received growth hormone treatment, or had stopped taking one at least 6 months before starting the trial. Subjects received once-weekly somapacitan, once-weekly placebo, or daily somatropin, which is FDA approved.
The primary endpoint was percentage change of truncal fat, which is regulated by growth hormone, and can lead to medical problems. After 34 weeks, subjects in the somapacitan group experienced a 1.06% decrease in truncal fat, compared with a 0.47% increase in the placebo group (P = .009) and a 2.23% decrease in the daily somatropin group.
After 34 weeks, a 52-week extension trial began. The somapacitan group continued on the drug and the placebo group was offered somapacitan. Patients on daily somatropin were randomized to continue daily treatment with somatropin or to switch to somapacitan.
At the end of the extension trial, those taking somapacitan for the full 86-week duration had an average reduction of 1.52% in truncal fat. After 86 weeks, the somapacitan and daily somatropin groups had similar values for percentage change in visceral fat, lean body mass, or appendicular skeletal muscle mass.
Common side effects of somapacitan were back pain, joint paint, indigestion, a sleep disorder, dizziness, tonsillitis, swelling in the arms or lower legs, vomiting, adrenal insufficiency, hypertension, increase in blood creatine phosphokinase, weight increase, and anemia.
Somapacitan, marketed as Sogroya by Novo Nordisk, is contraindicated in patients with an allergy to the drug, as well as those with an active malignancy, diabetic eye disease where increases in blood sugars could lead to retinal damage, acute critical illness, or acute respiratory failure.
The FDA recommends that providers perform an eye examination before drug initiation, as well as periodically while the patient is taking the drug, to rule out preexisting papilledema. This could be a sign of intracranial hypertension, which could be caused or worsened by growth hormones.
CagA-positive H. pylori patients at higher risk of osteoporosis, fracture
A new study has found that older patients who test positive for the cytotoxin associated gene-A (CagA) strain of Helicobacter pylori may be more at risk of both osteoporosis and fractures.
“Further studies will be required to replicate these findings in other cohorts and to better clarify the underlying pathogenetic mechanisms leading to increased bone fragility in subjects infected by CagA-positive H. pylori strains,” wrote Luigi Gennari, MD, PhD, of the University of Siena (Italy), and coauthors. The study was published in the Journal of Bone and Mineral Research.
To determine the effects of H. pylori on bone health and potential fracture risk, the researchers launched a population-based cohort study of 1,149 adults between the ages of 50 and 80 in Siena. The cohort comprised 174 males with an average (SD) age of 65.9 (plus or minus 6 years) and 975 females with an average age of 62.5 (plus or minus 6 years). All subjects were examined for H. pylori antibodies, and those who were infected were also examined for anti-CagA serum antibodies. As blood was sampled, bone mineral density (BMD) of the lumbar spine, femoral neck, total hip, and total body was measured via dual-energy x-ray absorptiometry.
In total, 53% of male participants and 49% of female participants tested positive for H. pylori, with CagA-positive strains found in 27% of males and 26% of females. No differences in infection rates were discovered in regard to socioeconomic status, age, weight, or height. Patients with normal BMD (45%), osteoporosis (51%), or osteopenia (49%) had similar prevalence of H. pylori infection, but CagA-positive strains were more frequently found in osteoporotic (30%) and osteopenic (26%) patients, compared to patients with normal BMD (21%, P < .01). CagA-positive female patients also had lower lumbar (0.950 g/cm2) and femoral (0.795 g/cm2) BMD, compared to CagA-negative (0.987 and 0.813 g/cm2) or H. pylori-negative women (0.997 and 0.821 g/cm2), respectively.
After an average follow-up period of 11.8 years, 199 nontraumatic fractures (72 vertebral and 127 nonvertebral) had occurred in 158 participants. Patients with CagA-positive strains of H. pylori had significantly increased risk of a clinical vertebral fracture (hazard ratio [HR], 5.27; 95% confidence interval, 2.23-12.63; P < .0001) or a nonvertebral incident fracture (HR, 2.09; 95% CI, 1.27-2.46; P < .01), compared to patients without H. pylori. After adjustment for age, sex, and body mass index, the risk among CagA-positive patients remained similarly significantly elevated for both vertebral (aHR, 4.78; 95% CI, 1.99-11.47; P < .0001) and nonvertebral fractures (aHR, 2.04; 95% CI, 1.22-3.41; P < .01).
The authors acknowledged their study’s limitations, including a cohort that was notably low in male participants, an inability to assess the effects of eradicating H. pylori on bone, and uncertainty as to which specific effects of H. pylori infection increase the risk of osteoporosis or fracture. Along those lines, they noted that an association between serum CagA antibody titer and gastric mucosal inflammation could lead to malabsorption of calcium, hypothesizing that antibody titer rather than antibody positivity “might be a more relevant marker for assessing the risk of bone fragility in patients affected by H. pylori infection.”
The study was supported in part by a grant from the Italian Association for Osteoporosis. The authors reported no potential conflicts of interest.
SOURCE: Gennari L et al. J Bone Miner Res. 2020 Aug 13. doi: 10.1002/jbmr.4162.
A new study has found that older patients who test positive for the cytotoxin associated gene-A (CagA) strain of Helicobacter pylori may be more at risk of both osteoporosis and fractures.
“Further studies will be required to replicate these findings in other cohorts and to better clarify the underlying pathogenetic mechanisms leading to increased bone fragility in subjects infected by CagA-positive H. pylori strains,” wrote Luigi Gennari, MD, PhD, of the University of Siena (Italy), and coauthors. The study was published in the Journal of Bone and Mineral Research.
To determine the effects of H. pylori on bone health and potential fracture risk, the researchers launched a population-based cohort study of 1,149 adults between the ages of 50 and 80 in Siena. The cohort comprised 174 males with an average (SD) age of 65.9 (plus or minus 6 years) and 975 females with an average age of 62.5 (plus or minus 6 years). All subjects were examined for H. pylori antibodies, and those who were infected were also examined for anti-CagA serum antibodies. As blood was sampled, bone mineral density (BMD) of the lumbar spine, femoral neck, total hip, and total body was measured via dual-energy x-ray absorptiometry.
In total, 53% of male participants and 49% of female participants tested positive for H. pylori, with CagA-positive strains found in 27% of males and 26% of females. No differences in infection rates were discovered in regard to socioeconomic status, age, weight, or height. Patients with normal BMD (45%), osteoporosis (51%), or osteopenia (49%) had similar prevalence of H. pylori infection, but CagA-positive strains were more frequently found in osteoporotic (30%) and osteopenic (26%) patients, compared to patients with normal BMD (21%, P < .01). CagA-positive female patients also had lower lumbar (0.950 g/cm2) and femoral (0.795 g/cm2) BMD, compared to CagA-negative (0.987 and 0.813 g/cm2) or H. pylori-negative women (0.997 and 0.821 g/cm2), respectively.
After an average follow-up period of 11.8 years, 199 nontraumatic fractures (72 vertebral and 127 nonvertebral) had occurred in 158 participants. Patients with CagA-positive strains of H. pylori had significantly increased risk of a clinical vertebral fracture (hazard ratio [HR], 5.27; 95% confidence interval, 2.23-12.63; P < .0001) or a nonvertebral incident fracture (HR, 2.09; 95% CI, 1.27-2.46; P < .01), compared to patients without H. pylori. After adjustment for age, sex, and body mass index, the risk among CagA-positive patients remained similarly significantly elevated for both vertebral (aHR, 4.78; 95% CI, 1.99-11.47; P < .0001) and nonvertebral fractures (aHR, 2.04; 95% CI, 1.22-3.41; P < .01).
The authors acknowledged their study’s limitations, including a cohort that was notably low in male participants, an inability to assess the effects of eradicating H. pylori on bone, and uncertainty as to which specific effects of H. pylori infection increase the risk of osteoporosis or fracture. Along those lines, they noted that an association between serum CagA antibody titer and gastric mucosal inflammation could lead to malabsorption of calcium, hypothesizing that antibody titer rather than antibody positivity “might be a more relevant marker for assessing the risk of bone fragility in patients affected by H. pylori infection.”
The study was supported in part by a grant from the Italian Association for Osteoporosis. The authors reported no potential conflicts of interest.
SOURCE: Gennari L et al. J Bone Miner Res. 2020 Aug 13. doi: 10.1002/jbmr.4162.
A new study has found that older patients who test positive for the cytotoxin associated gene-A (CagA) strain of Helicobacter pylori may be more at risk of both osteoporosis and fractures.
“Further studies will be required to replicate these findings in other cohorts and to better clarify the underlying pathogenetic mechanisms leading to increased bone fragility in subjects infected by CagA-positive H. pylori strains,” wrote Luigi Gennari, MD, PhD, of the University of Siena (Italy), and coauthors. The study was published in the Journal of Bone and Mineral Research.
To determine the effects of H. pylori on bone health and potential fracture risk, the researchers launched a population-based cohort study of 1,149 adults between the ages of 50 and 80 in Siena. The cohort comprised 174 males with an average (SD) age of 65.9 (plus or minus 6 years) and 975 females with an average age of 62.5 (plus or minus 6 years). All subjects were examined for H. pylori antibodies, and those who were infected were also examined for anti-CagA serum antibodies. As blood was sampled, bone mineral density (BMD) of the lumbar spine, femoral neck, total hip, and total body was measured via dual-energy x-ray absorptiometry.
In total, 53% of male participants and 49% of female participants tested positive for H. pylori, with CagA-positive strains found in 27% of males and 26% of females. No differences in infection rates were discovered in regard to socioeconomic status, age, weight, or height. Patients with normal BMD (45%), osteoporosis (51%), or osteopenia (49%) had similar prevalence of H. pylori infection, but CagA-positive strains were more frequently found in osteoporotic (30%) and osteopenic (26%) patients, compared to patients with normal BMD (21%, P < .01). CagA-positive female patients also had lower lumbar (0.950 g/cm2) and femoral (0.795 g/cm2) BMD, compared to CagA-negative (0.987 and 0.813 g/cm2) or H. pylori-negative women (0.997 and 0.821 g/cm2), respectively.
After an average follow-up period of 11.8 years, 199 nontraumatic fractures (72 vertebral and 127 nonvertebral) had occurred in 158 participants. Patients with CagA-positive strains of H. pylori had significantly increased risk of a clinical vertebral fracture (hazard ratio [HR], 5.27; 95% confidence interval, 2.23-12.63; P < .0001) or a nonvertebral incident fracture (HR, 2.09; 95% CI, 1.27-2.46; P < .01), compared to patients without H. pylori. After adjustment for age, sex, and body mass index, the risk among CagA-positive patients remained similarly significantly elevated for both vertebral (aHR, 4.78; 95% CI, 1.99-11.47; P < .0001) and nonvertebral fractures (aHR, 2.04; 95% CI, 1.22-3.41; P < .01).
The authors acknowledged their study’s limitations, including a cohort that was notably low in male participants, an inability to assess the effects of eradicating H. pylori on bone, and uncertainty as to which specific effects of H. pylori infection increase the risk of osteoporosis or fracture. Along those lines, they noted that an association between serum CagA antibody titer and gastric mucosal inflammation could lead to malabsorption of calcium, hypothesizing that antibody titer rather than antibody positivity “might be a more relevant marker for assessing the risk of bone fragility in patients affected by H. pylori infection.”
The study was supported in part by a grant from the Italian Association for Osteoporosis. The authors reported no potential conflicts of interest.
SOURCE: Gennari L et al. J Bone Miner Res. 2020 Aug 13. doi: 10.1002/jbmr.4162.
FROM THE JOURNAL OF BONE AND MINERAL RESEARCH
Obesity boosts risks in COVID-19 from diagnosis to death
A new analysis of existing research confirms a stark link between excess weight and COVID-19:
Obese patients faced the greatest bump in risk on the hospitalization front, with their odds of being admitted listed as 113% higher. The odds of diagnosis, ICU admission, and death were 46% higher (odds ratio [OR], 1.46; 95% confidence interval [CI], 1.30-1.65; P < .0001); 74% higher (OR, 1.74, CI, 1.46-2.08, P < .0001); 48% (OR, 1.48, CI, 1.22–1.80, P < .001, all pooled analyses and 95% CI), respectively. All differences were highly significantly different, investigators reported in a systematic review and meta-analysis published online Aug. 26 in Obesity Reviews.
“Essentially, these are pretty scary statistics,” nutrition researcher and study lead author Barry M. Popkin, PhD, of the University of North Carolina at Chapel Hill School of Public Health, said in an interview. “Other studies have talked about an increase in mortality, and we were thinking there’d be a little increase like 10% – nothing like 48%.”
According to the Johns Hopkins University of Medicine tracker, nearly 6 million people in the United States had been diagnosed with COVID-19 as of Aug. 30. The number of deaths had surpassed 183,000.
The authors of the new review launched their project to better understand the link between obesity and COVID-19 “all the way from being diagnosed to death,” Dr. Popkin said, adding that the meta-analysis is the largest of its kind to examine the link.
Dr. Popkin and colleagues analyzed 75 studies during January to June 2020 that tracked 399,461 patients (55% of whom were male) diagnosed with COVID-19. They found that 18 of 20 studies linked obesity with a 46% higher risk of diagnosis, but Dr. Popkin cautioned that this may be misleading. “I suspect it’s because they’re sicker and getting tested more for COVID,” he said. “I don’t think obesity enhances your likelihood of getting COVID. We don’t have a biological rationale for that.”
The researchers examined 19 studies that explored a link between obesity and hospitalization; all 19 found a higher risk of hospitalization in patients with obesity (pooled OR, 2.13). Twenty-one of 22 studies that looked at ICU admissions discovered a higher risk for patients with obesity (pooled OR, 1.74). And 27 of 35 studies that examined COVID-19 mortality found a higher death rate in patients with obesity (pooled OR, 1.48).
The review also looked at 14 studies that examined links between obesity and administration of invasive mechanical ventilation. All the studies found a higher risk for patients with obesity (pooled OR, 1.66; 95% CI, 1.38-1.99; P < .0001).
Could socioeconomic factors explain the difference in risk for people with obesity? It’s not clear. According to Dr. Popkin, most of the studies don’t examine factors such as income. While he believes physical factors are the key to the higher risk, he said “there’s clearly a social side to this.”
On the biological front, it appears that “the immune system is much weaker if you’re obese,” he said, and excess weight may worsen the course of a respiratory disease such as COVID-19 because of lung disorders such as sleep apnea.
In addition to highlighting inflammation and a weakened immune system, the review offers multiple explanations for why patients with obesity face worse outcomes in COVID-19. It may be more difficult for medical professionals to care for them in the hospital because of their weight, the authors wrote, and “obesity may also impair therapeutic treatments during COVID-19 infections.” The authors noted that ACE inhibitors may worsen COVID-19 in patients with type 2 diabetes.
The researchers noted that “potentially the vaccines developed to address COVID-19 will be less effective for individuals with obesity due to a weakened immune response.” They pointed to research that suggests T-cell responses are weaker and antibody titers wane at a faster rate in people with obesity who are vaccinated against influenza.
Pulmonologist Joshua L. Denson, MD, MS, of Tulane University, New Orleans, praised the review in an interview, but noted that some of the included studies have wide confidence intervals. One study that links COVID-19 to a sixfold higher mortality rate (OR, 6.29) has a confidence interval of 1.76-22.45.
Dr. Denson said he’s seen about 100 patients with COVID-19, and many are obese and have metabolic syndrome.
Like the authors of the study, he believes higher levels of inflammation play a crucial role in making these patients more vulnerable. “For whatever reason, the virus tends to really like that state. That’s driving these people to get sick,” he said.
Moving forward, Dr. Popkin urged physicians to redouble their efforts to warn patients about the risks of obesity and the importance of healthy eating. He also said COVID-19 vaccine researchers must stratify obese vs. nonobese subjects in clinical trials.
The review was funded by Bloomberg Philanthropies, the Carolina Population Center, World Bank, and Saudi Health Council. The review authors report no relevant disclosures. Dr. Denson reports no relevant disclosures.
SOURCE: Popkin BM et al. Obes Rev. 2020 Aug 26. doi: 10.1111/obr.13128.
A new analysis of existing research confirms a stark link between excess weight and COVID-19:
Obese patients faced the greatest bump in risk on the hospitalization front, with their odds of being admitted listed as 113% higher. The odds of diagnosis, ICU admission, and death were 46% higher (odds ratio [OR], 1.46; 95% confidence interval [CI], 1.30-1.65; P < .0001); 74% higher (OR, 1.74, CI, 1.46-2.08, P < .0001); 48% (OR, 1.48, CI, 1.22–1.80, P < .001, all pooled analyses and 95% CI), respectively. All differences were highly significantly different, investigators reported in a systematic review and meta-analysis published online Aug. 26 in Obesity Reviews.
“Essentially, these are pretty scary statistics,” nutrition researcher and study lead author Barry M. Popkin, PhD, of the University of North Carolina at Chapel Hill School of Public Health, said in an interview. “Other studies have talked about an increase in mortality, and we were thinking there’d be a little increase like 10% – nothing like 48%.”
According to the Johns Hopkins University of Medicine tracker, nearly 6 million people in the United States had been diagnosed with COVID-19 as of Aug. 30. The number of deaths had surpassed 183,000.
The authors of the new review launched their project to better understand the link between obesity and COVID-19 “all the way from being diagnosed to death,” Dr. Popkin said, adding that the meta-analysis is the largest of its kind to examine the link.
Dr. Popkin and colleagues analyzed 75 studies during January to June 2020 that tracked 399,461 patients (55% of whom were male) diagnosed with COVID-19. They found that 18 of 20 studies linked obesity with a 46% higher risk of diagnosis, but Dr. Popkin cautioned that this may be misleading. “I suspect it’s because they’re sicker and getting tested more for COVID,” he said. “I don’t think obesity enhances your likelihood of getting COVID. We don’t have a biological rationale for that.”
The researchers examined 19 studies that explored a link between obesity and hospitalization; all 19 found a higher risk of hospitalization in patients with obesity (pooled OR, 2.13). Twenty-one of 22 studies that looked at ICU admissions discovered a higher risk for patients with obesity (pooled OR, 1.74). And 27 of 35 studies that examined COVID-19 mortality found a higher death rate in patients with obesity (pooled OR, 1.48).
The review also looked at 14 studies that examined links between obesity and administration of invasive mechanical ventilation. All the studies found a higher risk for patients with obesity (pooled OR, 1.66; 95% CI, 1.38-1.99; P < .0001).
Could socioeconomic factors explain the difference in risk for people with obesity? It’s not clear. According to Dr. Popkin, most of the studies don’t examine factors such as income. While he believes physical factors are the key to the higher risk, he said “there’s clearly a social side to this.”
On the biological front, it appears that “the immune system is much weaker if you’re obese,” he said, and excess weight may worsen the course of a respiratory disease such as COVID-19 because of lung disorders such as sleep apnea.
In addition to highlighting inflammation and a weakened immune system, the review offers multiple explanations for why patients with obesity face worse outcomes in COVID-19. It may be more difficult for medical professionals to care for them in the hospital because of their weight, the authors wrote, and “obesity may also impair therapeutic treatments during COVID-19 infections.” The authors noted that ACE inhibitors may worsen COVID-19 in patients with type 2 diabetes.
The researchers noted that “potentially the vaccines developed to address COVID-19 will be less effective for individuals with obesity due to a weakened immune response.” They pointed to research that suggests T-cell responses are weaker and antibody titers wane at a faster rate in people with obesity who are vaccinated against influenza.
Pulmonologist Joshua L. Denson, MD, MS, of Tulane University, New Orleans, praised the review in an interview, but noted that some of the included studies have wide confidence intervals. One study that links COVID-19 to a sixfold higher mortality rate (OR, 6.29) has a confidence interval of 1.76-22.45.
Dr. Denson said he’s seen about 100 patients with COVID-19, and many are obese and have metabolic syndrome.
Like the authors of the study, he believes higher levels of inflammation play a crucial role in making these patients more vulnerable. “For whatever reason, the virus tends to really like that state. That’s driving these people to get sick,” he said.
Moving forward, Dr. Popkin urged physicians to redouble their efforts to warn patients about the risks of obesity and the importance of healthy eating. He also said COVID-19 vaccine researchers must stratify obese vs. nonobese subjects in clinical trials.
The review was funded by Bloomberg Philanthropies, the Carolina Population Center, World Bank, and Saudi Health Council. The review authors report no relevant disclosures. Dr. Denson reports no relevant disclosures.
SOURCE: Popkin BM et al. Obes Rev. 2020 Aug 26. doi: 10.1111/obr.13128.
A new analysis of existing research confirms a stark link between excess weight and COVID-19:
Obese patients faced the greatest bump in risk on the hospitalization front, with their odds of being admitted listed as 113% higher. The odds of diagnosis, ICU admission, and death were 46% higher (odds ratio [OR], 1.46; 95% confidence interval [CI], 1.30-1.65; P < .0001); 74% higher (OR, 1.74, CI, 1.46-2.08, P < .0001); 48% (OR, 1.48, CI, 1.22–1.80, P < .001, all pooled analyses and 95% CI), respectively. All differences were highly significantly different, investigators reported in a systematic review and meta-analysis published online Aug. 26 in Obesity Reviews.
“Essentially, these are pretty scary statistics,” nutrition researcher and study lead author Barry M. Popkin, PhD, of the University of North Carolina at Chapel Hill School of Public Health, said in an interview. “Other studies have talked about an increase in mortality, and we were thinking there’d be a little increase like 10% – nothing like 48%.”
According to the Johns Hopkins University of Medicine tracker, nearly 6 million people in the United States had been diagnosed with COVID-19 as of Aug. 30. The number of deaths had surpassed 183,000.
The authors of the new review launched their project to better understand the link between obesity and COVID-19 “all the way from being diagnosed to death,” Dr. Popkin said, adding that the meta-analysis is the largest of its kind to examine the link.
Dr. Popkin and colleagues analyzed 75 studies during January to June 2020 that tracked 399,461 patients (55% of whom were male) diagnosed with COVID-19. They found that 18 of 20 studies linked obesity with a 46% higher risk of diagnosis, but Dr. Popkin cautioned that this may be misleading. “I suspect it’s because they’re sicker and getting tested more for COVID,” he said. “I don’t think obesity enhances your likelihood of getting COVID. We don’t have a biological rationale for that.”
The researchers examined 19 studies that explored a link between obesity and hospitalization; all 19 found a higher risk of hospitalization in patients with obesity (pooled OR, 2.13). Twenty-one of 22 studies that looked at ICU admissions discovered a higher risk for patients with obesity (pooled OR, 1.74). And 27 of 35 studies that examined COVID-19 mortality found a higher death rate in patients with obesity (pooled OR, 1.48).
The review also looked at 14 studies that examined links between obesity and administration of invasive mechanical ventilation. All the studies found a higher risk for patients with obesity (pooled OR, 1.66; 95% CI, 1.38-1.99; P < .0001).
Could socioeconomic factors explain the difference in risk for people with obesity? It’s not clear. According to Dr. Popkin, most of the studies don’t examine factors such as income. While he believes physical factors are the key to the higher risk, he said “there’s clearly a social side to this.”
On the biological front, it appears that “the immune system is much weaker if you’re obese,” he said, and excess weight may worsen the course of a respiratory disease such as COVID-19 because of lung disorders such as sleep apnea.
In addition to highlighting inflammation and a weakened immune system, the review offers multiple explanations for why patients with obesity face worse outcomes in COVID-19. It may be more difficult for medical professionals to care for them in the hospital because of their weight, the authors wrote, and “obesity may also impair therapeutic treatments during COVID-19 infections.” The authors noted that ACE inhibitors may worsen COVID-19 in patients with type 2 diabetes.
The researchers noted that “potentially the vaccines developed to address COVID-19 will be less effective for individuals with obesity due to a weakened immune response.” They pointed to research that suggests T-cell responses are weaker and antibody titers wane at a faster rate in people with obesity who are vaccinated against influenza.
Pulmonologist Joshua L. Denson, MD, MS, of Tulane University, New Orleans, praised the review in an interview, but noted that some of the included studies have wide confidence intervals. One study that links COVID-19 to a sixfold higher mortality rate (OR, 6.29) has a confidence interval of 1.76-22.45.
Dr. Denson said he’s seen about 100 patients with COVID-19, and many are obese and have metabolic syndrome.
Like the authors of the study, he believes higher levels of inflammation play a crucial role in making these patients more vulnerable. “For whatever reason, the virus tends to really like that state. That’s driving these people to get sick,” he said.
Moving forward, Dr. Popkin urged physicians to redouble their efforts to warn patients about the risks of obesity and the importance of healthy eating. He also said COVID-19 vaccine researchers must stratify obese vs. nonobese subjects in clinical trials.
The review was funded by Bloomberg Philanthropies, the Carolina Population Center, World Bank, and Saudi Health Council. The review authors report no relevant disclosures. Dr. Denson reports no relevant disclosures.
SOURCE: Popkin BM et al. Obes Rev. 2020 Aug 26. doi: 10.1111/obr.13128.
FROM OBESITY REVIEWS
Gut bacteria linked to cardiovascular, other health conditions
Microorganisms in the human digestive tract are linked to 29 specific health conditions, including chronic obstructive pulmonary disease, high blood pressure, and type 2 diabetes, according to a genome analysis in more than 400,000 individuals.
Although previous studies have suggested a link between gut microbiota and diseases in humans, “the extent to which the human gut microbiome can be considered a determinant of disease and healthy aging remains unknown,” Hilde E. Groot, MD, of the University of Groningen (The Netherlands), said in a presentation at the virtual annual congress of the European Society of Cardiology.
To identify the spectrum of diseases linked to the gut microbiome, the researchers identified 422,417 unrelated adults of White British ancestry with genotype and matching genetic data. The average age of the participants was 57 years and 46% were male.
The researchers conducted a phenomewide association study including 35 distinct single-nucleotide polymorphisms (SNPs) that are known to influence the microbiome of the human gut.
Overall, seven SNPs were significantly associated with 29 disease outcomes including hypertension, type 2 diabetes, hypercholesterolemia, heart failure, renal failure, and osteoarthritis.
In addition, after a further sensitivity analysis using a Mendelian randomization (MR) approach, associations between Ruminococcus flavefaciens and hypertension and between Clostridium and platelet count might point to a causal link, the researchers said.
“Over the past few years, the amount of research concerning the human gut microbiome and the associations with health and disease has tremendously increased. However, most studies investigated one or a few traits. The strength of our study is the possibility to cover a wide range of traits simultaneously within one population,” Dr. Groot said in an interview.
“Our data support the hypothesis that the human gut microbiome is a complex system, involved in many pathophysiological mechanisms in the human body. So, our results are additional to earlier research and strengthen this hypothesis,” Dr. Groot added.
“Microbiota and their metabolites might be of importance in the interplay between overlapping pathophysiological processes, and could serve as potential therapeutic targets for the maintenance of health and prevention and treatment of cardiovascular diseases. However, before it is possible to give advice for the public and medical practice, further research is needed to study causality,” she emphasized.
“Currently, it is too soon to advise patients concerning their microbiome,” Dr. Groot noted. “However, genetic studies like ours might help other researchers to study causality between the gut microbiome and particular traits, which might potentially lead to new therapeutic targets. Next to genetic variants as a proxy, we’re currently studying the gut microbiome composition in myocardial infarction patients and healthy controls in a longitudinal setting.”
“Previous studies have suggested a potential link between the gut microbiome and the development of cardiovascular disease, type 2 diabetes mellitus, and other chronic disorders,” Carol Ann Remme, MD, of the Amsterdam University Medical Center, said in an interview. “However, it is challenging to study the effect of gut microbiome composition in large patient cohorts. As an alternative approach, the study authors showed in a very large population that genetic variants previously shown to influence gut microbiome composition were significantly associated with conditions such as hypertension, type 2 diabetes, hypercholesterolemia, and heart failure.”
The study is unique in that it employed a very large cohort of more than 400,000 individuals, which is typically required to be able to draw clear conclusions, Dr. Remme continued. “The authors were able to further refine their findings by linking genetic variants known to influence specific gut bacteria to some particular disorders,” she noted.
“It is becoming increasingly clear that an individual’s gut microbiome composition, which is defined by both genetic and environmental factors such as diet, may affect his/her susceptibility to certain diseases – including cardiovascular – in addition to disease progression and outcome,” said Dr. Remme. “This may ultimately lead to development of novel, personalized strategies for risk stratification in addition to potential preventive measures targeting the gut microbiome. I expect this area of research will become increasingly important in the coming years.”
The study received no outside funding. Dr. Groot and colleagues had no financial conflicts to disclose. Dr. Remme had no financial conflicts to disclose.
Microorganisms in the human digestive tract are linked to 29 specific health conditions, including chronic obstructive pulmonary disease, high blood pressure, and type 2 diabetes, according to a genome analysis in more than 400,000 individuals.
Although previous studies have suggested a link between gut microbiota and diseases in humans, “the extent to which the human gut microbiome can be considered a determinant of disease and healthy aging remains unknown,” Hilde E. Groot, MD, of the University of Groningen (The Netherlands), said in a presentation at the virtual annual congress of the European Society of Cardiology.
To identify the spectrum of diseases linked to the gut microbiome, the researchers identified 422,417 unrelated adults of White British ancestry with genotype and matching genetic data. The average age of the participants was 57 years and 46% were male.
The researchers conducted a phenomewide association study including 35 distinct single-nucleotide polymorphisms (SNPs) that are known to influence the microbiome of the human gut.
Overall, seven SNPs were significantly associated with 29 disease outcomes including hypertension, type 2 diabetes, hypercholesterolemia, heart failure, renal failure, and osteoarthritis.
In addition, after a further sensitivity analysis using a Mendelian randomization (MR) approach, associations between Ruminococcus flavefaciens and hypertension and between Clostridium and platelet count might point to a causal link, the researchers said.
“Over the past few years, the amount of research concerning the human gut microbiome and the associations with health and disease has tremendously increased. However, most studies investigated one or a few traits. The strength of our study is the possibility to cover a wide range of traits simultaneously within one population,” Dr. Groot said in an interview.
“Our data support the hypothesis that the human gut microbiome is a complex system, involved in many pathophysiological mechanisms in the human body. So, our results are additional to earlier research and strengthen this hypothesis,” Dr. Groot added.
“Microbiota and their metabolites might be of importance in the interplay between overlapping pathophysiological processes, and could serve as potential therapeutic targets for the maintenance of health and prevention and treatment of cardiovascular diseases. However, before it is possible to give advice for the public and medical practice, further research is needed to study causality,” she emphasized.
“Currently, it is too soon to advise patients concerning their microbiome,” Dr. Groot noted. “However, genetic studies like ours might help other researchers to study causality between the gut microbiome and particular traits, which might potentially lead to new therapeutic targets. Next to genetic variants as a proxy, we’re currently studying the gut microbiome composition in myocardial infarction patients and healthy controls in a longitudinal setting.”
“Previous studies have suggested a potential link between the gut microbiome and the development of cardiovascular disease, type 2 diabetes mellitus, and other chronic disorders,” Carol Ann Remme, MD, of the Amsterdam University Medical Center, said in an interview. “However, it is challenging to study the effect of gut microbiome composition in large patient cohorts. As an alternative approach, the study authors showed in a very large population that genetic variants previously shown to influence gut microbiome composition were significantly associated with conditions such as hypertension, type 2 diabetes, hypercholesterolemia, and heart failure.”
The study is unique in that it employed a very large cohort of more than 400,000 individuals, which is typically required to be able to draw clear conclusions, Dr. Remme continued. “The authors were able to further refine their findings by linking genetic variants known to influence specific gut bacteria to some particular disorders,” she noted.
“It is becoming increasingly clear that an individual’s gut microbiome composition, which is defined by both genetic and environmental factors such as diet, may affect his/her susceptibility to certain diseases – including cardiovascular – in addition to disease progression and outcome,” said Dr. Remme. “This may ultimately lead to development of novel, personalized strategies for risk stratification in addition to potential preventive measures targeting the gut microbiome. I expect this area of research will become increasingly important in the coming years.”
The study received no outside funding. Dr. Groot and colleagues had no financial conflicts to disclose. Dr. Remme had no financial conflicts to disclose.
Microorganisms in the human digestive tract are linked to 29 specific health conditions, including chronic obstructive pulmonary disease, high blood pressure, and type 2 diabetes, according to a genome analysis in more than 400,000 individuals.
Although previous studies have suggested a link between gut microbiota and diseases in humans, “the extent to which the human gut microbiome can be considered a determinant of disease and healthy aging remains unknown,” Hilde E. Groot, MD, of the University of Groningen (The Netherlands), said in a presentation at the virtual annual congress of the European Society of Cardiology.
To identify the spectrum of diseases linked to the gut microbiome, the researchers identified 422,417 unrelated adults of White British ancestry with genotype and matching genetic data. The average age of the participants was 57 years and 46% were male.
The researchers conducted a phenomewide association study including 35 distinct single-nucleotide polymorphisms (SNPs) that are known to influence the microbiome of the human gut.
Overall, seven SNPs were significantly associated with 29 disease outcomes including hypertension, type 2 diabetes, hypercholesterolemia, heart failure, renal failure, and osteoarthritis.
In addition, after a further sensitivity analysis using a Mendelian randomization (MR) approach, associations between Ruminococcus flavefaciens and hypertension and between Clostridium and platelet count might point to a causal link, the researchers said.
“Over the past few years, the amount of research concerning the human gut microbiome and the associations with health and disease has tremendously increased. However, most studies investigated one or a few traits. The strength of our study is the possibility to cover a wide range of traits simultaneously within one population,” Dr. Groot said in an interview.
“Our data support the hypothesis that the human gut microbiome is a complex system, involved in many pathophysiological mechanisms in the human body. So, our results are additional to earlier research and strengthen this hypothesis,” Dr. Groot added.
“Microbiota and their metabolites might be of importance in the interplay between overlapping pathophysiological processes, and could serve as potential therapeutic targets for the maintenance of health and prevention and treatment of cardiovascular diseases. However, before it is possible to give advice for the public and medical practice, further research is needed to study causality,” she emphasized.
“Currently, it is too soon to advise patients concerning their microbiome,” Dr. Groot noted. “However, genetic studies like ours might help other researchers to study causality between the gut microbiome and particular traits, which might potentially lead to new therapeutic targets. Next to genetic variants as a proxy, we’re currently studying the gut microbiome composition in myocardial infarction patients and healthy controls in a longitudinal setting.”
“Previous studies have suggested a potential link between the gut microbiome and the development of cardiovascular disease, type 2 diabetes mellitus, and other chronic disorders,” Carol Ann Remme, MD, of the Amsterdam University Medical Center, said in an interview. “However, it is challenging to study the effect of gut microbiome composition in large patient cohorts. As an alternative approach, the study authors showed in a very large population that genetic variants previously shown to influence gut microbiome composition were significantly associated with conditions such as hypertension, type 2 diabetes, hypercholesterolemia, and heart failure.”
The study is unique in that it employed a very large cohort of more than 400,000 individuals, which is typically required to be able to draw clear conclusions, Dr. Remme continued. “The authors were able to further refine their findings by linking genetic variants known to influence specific gut bacteria to some particular disorders,” she noted.
“It is becoming increasingly clear that an individual’s gut microbiome composition, which is defined by both genetic and environmental factors such as diet, may affect his/her susceptibility to certain diseases – including cardiovascular – in addition to disease progression and outcome,” said Dr. Remme. “This may ultimately lead to development of novel, personalized strategies for risk stratification in addition to potential preventive measures targeting the gut microbiome. I expect this area of research will become increasingly important in the coming years.”
The study received no outside funding. Dr. Groot and colleagues had no financial conflicts to disclose. Dr. Remme had no financial conflicts to disclose.
FROM ESC CONGRESS 2020
DAPA-CKD: SGLT2 inhibitor benefit extends to chronic kidney disease without diabetes
In the DAPA-CKD trial, treatment with the SGLT2 inhibitor dapagliflozin (Farxiga) cut the incidence of substantially worsened chronic kidney disease by an average of 39% compared with placebo when added to standard treatment, with a number needed to treat of 19 to prevent one primary outcome event after a median of 2.4 years.
The level of benefit was similar in both the one-third of enrolled patients without diabetes and in the two-thirds with diabetes, showing a statistically significant 50% cut in the primary endpoint among patients without diabetes, Hiddo J.L. Heerspink, MD, reported at the virtual annual congress of the European Society of Cardiology.
“We found that dapagliflozin delayed the initiation of dialysis, and reduced the number of deaths,” regardless of diabetes status, Dr. Heerspink, of University Medical Centre Groningen, the Netherlands, said during a press conference. “DAPA-CKD trial has shown dapagliflozin’s potential as a long-awaited new treatment for patients with chronic kidney disease.”
This finding ushers in a “completely new era in chronic kidney disease management,” said Janani Rangaswami, MD, a nephrologist and cardiorenal syndrome specialist at Einstein Medical Center in Philadelphia. “It’s good news” for these patients.
The results showed that dapagliflozin is the first “game changing” drug for chronic kidney disease in 2 decades, following the introduction of angiotensin converting enzyme inhibitors and angiotensin receptor blockers, she said in an interview. And given the consistency of the findings with the results from several other studies that documented meaningful renal protection by several different SGLT2 inhibitors, the results from this single trial also convincingly establish dapagliflozin as a standard-of-care agent to use on the types of patients the study enrolled, she said in an interview.
Representing many real-world patients
The DAPA-CKD trial enrolled 4,304 patients with albuminuria based on having a urinary albumin-to-creatinine ratio of at least 200 mg/g, and an estimated glomerular filtration rate (eGFR) of 25-75 mL/min per 1.73 m2 (with 90% of patients having an eGFR of less than 60 mL/min per 1.73 m2), and 97% were on treatment with a renin-angiotensin system–blocking drug. The primary endpoint was the combined rate of a drop in eGFR of at least 50% from baseline, progression to end stage renal disease, or renal or cardiovascular death; the between-group difference in this composite was driven primarily by both preserved eGFR and by prevention of end stage renal disease.
This represents both an appropriate target population, and meaningful endpoints, Dr. Rangaswami said. The study was “very representative of who we see in real-world practice,” a group that likely includes “hundreds of thousands” of U.S. patients with nondiabetic chronic kidney disease, she estimated.
Another notable finding was that 14% of the enrolled patients had eGFR values at baseline of 25-29 mL/min per 1.73 m2, pegging them as having stage 4 chronic kidney disease, and the median baseline eGFR was 43 mL/min per 1.73 m2, but dapagliflozin treatment was as safe and effective in these patients as it was in enrolled patients with a higher level of retained renal activity. This experience should give clinicians greater confidence about using dapagliflozin and other drugs in the sodium-glucose cotransporter (SGLT) 2 inhibitor class in patients with substantially depressed renal function, Dr. Rangaswami said.
“We now need to be more proactive about treating patients with more advanced kidney disease who can still benefit” from dapagliflozin treatment. “The sooner you intervene the better,” to slow further progression, but the new findings show “benefit even when treating patients with lower eGFRs. There is still hope to prevent or delay dialysis.”
A heart-kidney connection
Dapagliflozin treatment also cut all-cause mortality by a statistically significant, relative 31%, and another secondary-endpoint analysis showed a statistically significant 29% relative reduction in the rate of cardiovascular death or heart failure hospitalization, a benefit seen consistently in several prior studies of SGLT2 inhibitors, but possibly unexpected here because enrolled patients underwent no selection for a history of heart failure or any other cardiovascular disease. But the finding shouldn’t surprise, because “chronic kidney disease is an independent risk factor for cardiovascular disease across the board, and especially for heart failure,” noted Dr. Rangaswami.
“Heart and kidney disease is one big spectrum,” and the collected experience of several trials that have now proven the efficacy of SGLT2 inhibitors among patients with heart failure with reduced ejection fraction or with chronic kidney disease, regardless of their glycemic control, shows how broadly this drug class can benefit patients across the breadth of this spectrum, she said.
DAPA-CKD was funded by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Heerspink has been a consultant to and received research funding from AstraZeneca and from several other companies. Dr. Rangaswami had no disclosures.
In the DAPA-CKD trial, treatment with the SGLT2 inhibitor dapagliflozin (Farxiga) cut the incidence of substantially worsened chronic kidney disease by an average of 39% compared with placebo when added to standard treatment, with a number needed to treat of 19 to prevent one primary outcome event after a median of 2.4 years.
The level of benefit was similar in both the one-third of enrolled patients without diabetes and in the two-thirds with diabetes, showing a statistically significant 50% cut in the primary endpoint among patients without diabetes, Hiddo J.L. Heerspink, MD, reported at the virtual annual congress of the European Society of Cardiology.
“We found that dapagliflozin delayed the initiation of dialysis, and reduced the number of deaths,” regardless of diabetes status, Dr. Heerspink, of University Medical Centre Groningen, the Netherlands, said during a press conference. “DAPA-CKD trial has shown dapagliflozin’s potential as a long-awaited new treatment for patients with chronic kidney disease.”
This finding ushers in a “completely new era in chronic kidney disease management,” said Janani Rangaswami, MD, a nephrologist and cardiorenal syndrome specialist at Einstein Medical Center in Philadelphia. “It’s good news” for these patients.
The results showed that dapagliflozin is the first “game changing” drug for chronic kidney disease in 2 decades, following the introduction of angiotensin converting enzyme inhibitors and angiotensin receptor blockers, she said in an interview. And given the consistency of the findings with the results from several other studies that documented meaningful renal protection by several different SGLT2 inhibitors, the results from this single trial also convincingly establish dapagliflozin as a standard-of-care agent to use on the types of patients the study enrolled, she said in an interview.
Representing many real-world patients
The DAPA-CKD trial enrolled 4,304 patients with albuminuria based on having a urinary albumin-to-creatinine ratio of at least 200 mg/g, and an estimated glomerular filtration rate (eGFR) of 25-75 mL/min per 1.73 m2 (with 90% of patients having an eGFR of less than 60 mL/min per 1.73 m2), and 97% were on treatment with a renin-angiotensin system–blocking drug. The primary endpoint was the combined rate of a drop in eGFR of at least 50% from baseline, progression to end stage renal disease, or renal or cardiovascular death; the between-group difference in this composite was driven primarily by both preserved eGFR and by prevention of end stage renal disease.
This represents both an appropriate target population, and meaningful endpoints, Dr. Rangaswami said. The study was “very representative of who we see in real-world practice,” a group that likely includes “hundreds of thousands” of U.S. patients with nondiabetic chronic kidney disease, she estimated.
Another notable finding was that 14% of the enrolled patients had eGFR values at baseline of 25-29 mL/min per 1.73 m2, pegging them as having stage 4 chronic kidney disease, and the median baseline eGFR was 43 mL/min per 1.73 m2, but dapagliflozin treatment was as safe and effective in these patients as it was in enrolled patients with a higher level of retained renal activity. This experience should give clinicians greater confidence about using dapagliflozin and other drugs in the sodium-glucose cotransporter (SGLT) 2 inhibitor class in patients with substantially depressed renal function, Dr. Rangaswami said.
“We now need to be more proactive about treating patients with more advanced kidney disease who can still benefit” from dapagliflozin treatment. “The sooner you intervene the better,” to slow further progression, but the new findings show “benefit even when treating patients with lower eGFRs. There is still hope to prevent or delay dialysis.”
A heart-kidney connection
Dapagliflozin treatment also cut all-cause mortality by a statistically significant, relative 31%, and another secondary-endpoint analysis showed a statistically significant 29% relative reduction in the rate of cardiovascular death or heart failure hospitalization, a benefit seen consistently in several prior studies of SGLT2 inhibitors, but possibly unexpected here because enrolled patients underwent no selection for a history of heart failure or any other cardiovascular disease. But the finding shouldn’t surprise, because “chronic kidney disease is an independent risk factor for cardiovascular disease across the board, and especially for heart failure,” noted Dr. Rangaswami.
“Heart and kidney disease is one big spectrum,” and the collected experience of several trials that have now proven the efficacy of SGLT2 inhibitors among patients with heart failure with reduced ejection fraction or with chronic kidney disease, regardless of their glycemic control, shows how broadly this drug class can benefit patients across the breadth of this spectrum, she said.
DAPA-CKD was funded by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Heerspink has been a consultant to and received research funding from AstraZeneca and from several other companies. Dr. Rangaswami had no disclosures.
In the DAPA-CKD trial, treatment with the SGLT2 inhibitor dapagliflozin (Farxiga) cut the incidence of substantially worsened chronic kidney disease by an average of 39% compared with placebo when added to standard treatment, with a number needed to treat of 19 to prevent one primary outcome event after a median of 2.4 years.
The level of benefit was similar in both the one-third of enrolled patients without diabetes and in the two-thirds with diabetes, showing a statistically significant 50% cut in the primary endpoint among patients without diabetes, Hiddo J.L. Heerspink, MD, reported at the virtual annual congress of the European Society of Cardiology.
“We found that dapagliflozin delayed the initiation of dialysis, and reduced the number of deaths,” regardless of diabetes status, Dr. Heerspink, of University Medical Centre Groningen, the Netherlands, said during a press conference. “DAPA-CKD trial has shown dapagliflozin’s potential as a long-awaited new treatment for patients with chronic kidney disease.”
This finding ushers in a “completely new era in chronic kidney disease management,” said Janani Rangaswami, MD, a nephrologist and cardiorenal syndrome specialist at Einstein Medical Center in Philadelphia. “It’s good news” for these patients.
The results showed that dapagliflozin is the first “game changing” drug for chronic kidney disease in 2 decades, following the introduction of angiotensin converting enzyme inhibitors and angiotensin receptor blockers, she said in an interview. And given the consistency of the findings with the results from several other studies that documented meaningful renal protection by several different SGLT2 inhibitors, the results from this single trial also convincingly establish dapagliflozin as a standard-of-care agent to use on the types of patients the study enrolled, she said in an interview.
Representing many real-world patients
The DAPA-CKD trial enrolled 4,304 patients with albuminuria based on having a urinary albumin-to-creatinine ratio of at least 200 mg/g, and an estimated glomerular filtration rate (eGFR) of 25-75 mL/min per 1.73 m2 (with 90% of patients having an eGFR of less than 60 mL/min per 1.73 m2), and 97% were on treatment with a renin-angiotensin system–blocking drug. The primary endpoint was the combined rate of a drop in eGFR of at least 50% from baseline, progression to end stage renal disease, or renal or cardiovascular death; the between-group difference in this composite was driven primarily by both preserved eGFR and by prevention of end stage renal disease.
This represents both an appropriate target population, and meaningful endpoints, Dr. Rangaswami said. The study was “very representative of who we see in real-world practice,” a group that likely includes “hundreds of thousands” of U.S. patients with nondiabetic chronic kidney disease, she estimated.
Another notable finding was that 14% of the enrolled patients had eGFR values at baseline of 25-29 mL/min per 1.73 m2, pegging them as having stage 4 chronic kidney disease, and the median baseline eGFR was 43 mL/min per 1.73 m2, but dapagliflozin treatment was as safe and effective in these patients as it was in enrolled patients with a higher level of retained renal activity. This experience should give clinicians greater confidence about using dapagliflozin and other drugs in the sodium-glucose cotransporter (SGLT) 2 inhibitor class in patients with substantially depressed renal function, Dr. Rangaswami said.
“We now need to be more proactive about treating patients with more advanced kidney disease who can still benefit” from dapagliflozin treatment. “The sooner you intervene the better,” to slow further progression, but the new findings show “benefit even when treating patients with lower eGFRs. There is still hope to prevent or delay dialysis.”
A heart-kidney connection
Dapagliflozin treatment also cut all-cause mortality by a statistically significant, relative 31%, and another secondary-endpoint analysis showed a statistically significant 29% relative reduction in the rate of cardiovascular death or heart failure hospitalization, a benefit seen consistently in several prior studies of SGLT2 inhibitors, but possibly unexpected here because enrolled patients underwent no selection for a history of heart failure or any other cardiovascular disease. But the finding shouldn’t surprise, because “chronic kidney disease is an independent risk factor for cardiovascular disease across the board, and especially for heart failure,” noted Dr. Rangaswami.
“Heart and kidney disease is one big spectrum,” and the collected experience of several trials that have now proven the efficacy of SGLT2 inhibitors among patients with heart failure with reduced ejection fraction or with chronic kidney disease, regardless of their glycemic control, shows how broadly this drug class can benefit patients across the breadth of this spectrum, she said.
DAPA-CKD was funded by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Heerspink has been a consultant to and received research funding from AstraZeneca and from several other companies. Dr. Rangaswami had no disclosures.
FROM ESC CONGRESS 2020
EMPEROR-Reduced: Empagliflozin’s HFrEF benefit solidifies class effects
The SGLT2 inhibitor drug class solidified its role as a major, new treatment for patients with heart failure with reduced ejection fraction and no diabetes, with results from a second large, controlled trial showing clear efficacy and safety in this population.
Patients with heart failure with reduced ejection fraction (HFrEF) treated with the sodium glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) had a statistically significant 25% relative cut in their incidence of cardiovascular death or first heart failure hospitalization, compared with placebo-treated controls when added on top of standard HFrEF treatment, and this benefit was consistent regardless of whether the treated patients also had type 2 diabetes, Milton Packer, MD, reported at the virtual annual congress of the European Society of Cardiology.
This 25% drop in the primary endpoint with empagliflozin treatment in the EMPEROR-Reduced trial exactly matched the cut in incidence of cardiovascular death or heart failure hospitalization produced by treatment with a another SGLT2 inhibitor, dapagliflozin (Farxiga), in the DAPA-HF trial (N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
The performance of these two SGLT2 inhibitors was “incredibly consistent” across the their respective trials run in HFrEF patients with and without type 2 diabetes, and the combined evidence base of the two trials makes for “really compelling evidence” of both safety and efficacy that should prompt a change to U.S. practice, with both of these drugs forming a new cornerstone of HFrEF treatment, Dr. Packer said.
Results plant drug class firmly as HFrEF treatment
Dr. Packer stressed in his presentation that optimal treatment of patients with HFrEF now demands use of one of these two SGLT2 inhibitors, as well as sacubitril plus valsartan (Entresto), a beta-blocker, and a mineralocorticoid receptor antagonist, plus a diuretic as a fifth drug class for the many HFrEF patients who also need treatment for fluid overload. He further advocated for rapid introduction of these four cornerstone agents with proven survival benefits once a patient receives a HFrEF diagnosis, suggesting that sacubitril plus valsartan, an SGLT2 inhibitor, a beta-blocker, and a mineralocorticoid receptor antagonist could all be initiated within 6 weeks or less while acknowledging that optimal up-titration of the beta-blocker would likely take longer.
The order in which a patient starts these drugs shouldn’t matter, and there currently seems to be no evidence that clearly points toward using either dapagliflozin or empagliflozin over the other, Dr. Packer added.
In recognition of the importance of sending a message to heart failure clinicians about the newly proven efficacy of SGLT2 inhibitors in HFrEF patients, the American College of Cardiology and American Heart Association are now drafting an “expert decision pathway” to help clinicians as they enter this new prescribing space. This interim guidance should come out before the end of 2020, prior to release of fully revised HFrEF management guidelines in 2021, said Athena Poppas, MD, president of the ACC, in an interview.
“There is clearly need for education” that can help guide physicians who care for HFrEF patients on how to introduce an SGLT2 inhibitor along with the additional, lengthy list of drug classes proven to benefit these patients, noted Dr. Poppas, who is also a professor and chief of cardiology at the Brown University in Providence, R.I. Physicians may find that they need extra backup for successfully starting both sacubitril plus valsartan and an SGLT2 inhibitor in HFrEF patients because recent history has shown substantial pushback from third-party payers in reimbursing for these relatively expensive drugs, Dr. Poppas noted. She added that this is a problem that may be compounded when patients should ideally get both drug classes.
Physicians who care for heart failure patients have their own history of dragging their feet when adding new drugs to the regimens HFrEF patients receive. The angiotensin converting enzyme inhibitors and beta-blockers took about 17 years each to start reaching a majority of U.S. HFrEF patients, and sacubitril plus valsartan is now used on perhaps a quarter to a third of HFrEF patients despite receiving Food and Drug Administration approval for these patients in mid 2015, noted Christopher M. O’Connor, MD, a heart failure specialist and president of the Inova Heart and Vascular Institute in Fairfax, Va.
Despite dapagliflozin receiving FDA approval in May 2020 for treating HFrEF in patients without diabetes, early uptake in U.S. practice has been very slow, with findings from large U.S. patient registries suggesting that perhaps 1% of suitable HFrEF patients currently get the drug, estimated Dr. O’Connor in an interview.
Given how strong the evidence now is for benefit and safety from dapagliflozin and empagliflozin, it may take as little as 5 years to reach greater than 50% penetration of one of these drugs into U.S. HFrEF patient populations, suggested Dr. Packer, a distinguished scholar in cardiovascular science at Baylor University Medical Center in Dallas.
EMPEROR-Reduced outcomes
The road to routine use of these SGLT2 inhibitor drugs should be hastened by empagliflozin’s impressive performance in EMPEROR-Reduced, in which the drug scored highly significant benefits over placebo for the prespecified primary and two major secondary endpoints, one of which was a measure of preserved renal function.
The trial randomized 3,730 patients at 520 sites in 20 countries during 2017-2019 and followed them on treatment for a median of 16 months. All patients had a left ventricular ejection fraction of 40% or less, and roughly three-quarters had New York Heart Association (NYHA) class II function, nearly one-quarter had class III function, and fewer than 1% of patients fell into the class IV category.
The primary endpoint occurred in 19% of the empagliflozin-treated patients and in 25% of those who received placebo. Among the half of patients with diabetes in the trial, the relative risk reduction by empagliflozin compared with placebo was a statistically significant 28%; among those without diabetes, it was a statistically significant 22%. Concurrently with Dr. Packer’s report, the results appeared in an article posted online (N Engl J Med. 2020 Aug 29. doi: 10.1056/NEJMoa2022190).
The study also had two main prespecified secondary endpoints: the incidence of total hospitalizations for heart failure, both first and recurrent, which fell by 30% in the empagliflozin-treated patients, compared with placebo, and the rate of declining renal function during the 16 months of the study as measured by estimated glomerular filtration rate, which dropped by roughly 1 mL/min per 1.73 m2 among the empagliflozin recipients and by about 4 mL/min/ per 1.73 m2 in the placebo patients.
Treatment with empagliflozin also achieved a notable, statistically significant 50% drop in major adverse renal events, consistent with the performance of other drugs in the class.
“Renal protection is a big plus” of empagliflozin in this trial and from the other SGLT2 inhibitors in prior studies, noted Dr. O’Connor.
The EMPEROR-Reduced results also showed an important benefit for HFrEF patients from empagliflozin not previously seen as quickly with any other drug class, noted Dr. Packer. The SGLT2 inhibitor led to statistically a significant slowing in the progression of patients from NYHA class II function to class III, compared with placebo, and it also significantly promoted the recovery of patients from NYHA class III to class II, an effect that became apparent within the first month on treatment and a benefit that is a “big deal” for patients because it represents a “significant change in functional capacity.” This additional dimension of empagliflozin’s benefit “really impressed me,” Dr. Packer said.
EMPEROR-Reduced was funded by Boehringer Ingelheim and Eli Lilly, the companies that market empagliflozin. Dr. Packer has received personal fees from Boehringer Ingelheim and Eli Lilly and from several other companies. Dr. Poppas and Dr. O’Connor had no relevant disclosures.
SOURCE: Packer M. ESC 2020. N Engl J Med. 2020 Aug 29. doi: 10.1056/NEJMoa2022190.
The SGLT2 inhibitor drug class solidified its role as a major, new treatment for patients with heart failure with reduced ejection fraction and no diabetes, with results from a second large, controlled trial showing clear efficacy and safety in this population.
Patients with heart failure with reduced ejection fraction (HFrEF) treated with the sodium glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) had a statistically significant 25% relative cut in their incidence of cardiovascular death or first heart failure hospitalization, compared with placebo-treated controls when added on top of standard HFrEF treatment, and this benefit was consistent regardless of whether the treated patients also had type 2 diabetes, Milton Packer, MD, reported at the virtual annual congress of the European Society of Cardiology.
This 25% drop in the primary endpoint with empagliflozin treatment in the EMPEROR-Reduced trial exactly matched the cut in incidence of cardiovascular death or heart failure hospitalization produced by treatment with a another SGLT2 inhibitor, dapagliflozin (Farxiga), in the DAPA-HF trial (N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
The performance of these two SGLT2 inhibitors was “incredibly consistent” across the their respective trials run in HFrEF patients with and without type 2 diabetes, and the combined evidence base of the two trials makes for “really compelling evidence” of both safety and efficacy that should prompt a change to U.S. practice, with both of these drugs forming a new cornerstone of HFrEF treatment, Dr. Packer said.
Results plant drug class firmly as HFrEF treatment
Dr. Packer stressed in his presentation that optimal treatment of patients with HFrEF now demands use of one of these two SGLT2 inhibitors, as well as sacubitril plus valsartan (Entresto), a beta-blocker, and a mineralocorticoid receptor antagonist, plus a diuretic as a fifth drug class for the many HFrEF patients who also need treatment for fluid overload. He further advocated for rapid introduction of these four cornerstone agents with proven survival benefits once a patient receives a HFrEF diagnosis, suggesting that sacubitril plus valsartan, an SGLT2 inhibitor, a beta-blocker, and a mineralocorticoid receptor antagonist could all be initiated within 6 weeks or less while acknowledging that optimal up-titration of the beta-blocker would likely take longer.
The order in which a patient starts these drugs shouldn’t matter, and there currently seems to be no evidence that clearly points toward using either dapagliflozin or empagliflozin over the other, Dr. Packer added.
In recognition of the importance of sending a message to heart failure clinicians about the newly proven efficacy of SGLT2 inhibitors in HFrEF patients, the American College of Cardiology and American Heart Association are now drafting an “expert decision pathway” to help clinicians as they enter this new prescribing space. This interim guidance should come out before the end of 2020, prior to release of fully revised HFrEF management guidelines in 2021, said Athena Poppas, MD, president of the ACC, in an interview.
“There is clearly need for education” that can help guide physicians who care for HFrEF patients on how to introduce an SGLT2 inhibitor along with the additional, lengthy list of drug classes proven to benefit these patients, noted Dr. Poppas, who is also a professor and chief of cardiology at the Brown University in Providence, R.I. Physicians may find that they need extra backup for successfully starting both sacubitril plus valsartan and an SGLT2 inhibitor in HFrEF patients because recent history has shown substantial pushback from third-party payers in reimbursing for these relatively expensive drugs, Dr. Poppas noted. She added that this is a problem that may be compounded when patients should ideally get both drug classes.
Physicians who care for heart failure patients have their own history of dragging their feet when adding new drugs to the regimens HFrEF patients receive. The angiotensin converting enzyme inhibitors and beta-blockers took about 17 years each to start reaching a majority of U.S. HFrEF patients, and sacubitril plus valsartan is now used on perhaps a quarter to a third of HFrEF patients despite receiving Food and Drug Administration approval for these patients in mid 2015, noted Christopher M. O’Connor, MD, a heart failure specialist and president of the Inova Heart and Vascular Institute in Fairfax, Va.
Despite dapagliflozin receiving FDA approval in May 2020 for treating HFrEF in patients without diabetes, early uptake in U.S. practice has been very slow, with findings from large U.S. patient registries suggesting that perhaps 1% of suitable HFrEF patients currently get the drug, estimated Dr. O’Connor in an interview.
Given how strong the evidence now is for benefit and safety from dapagliflozin and empagliflozin, it may take as little as 5 years to reach greater than 50% penetration of one of these drugs into U.S. HFrEF patient populations, suggested Dr. Packer, a distinguished scholar in cardiovascular science at Baylor University Medical Center in Dallas.
EMPEROR-Reduced outcomes
The road to routine use of these SGLT2 inhibitor drugs should be hastened by empagliflozin’s impressive performance in EMPEROR-Reduced, in which the drug scored highly significant benefits over placebo for the prespecified primary and two major secondary endpoints, one of which was a measure of preserved renal function.
The trial randomized 3,730 patients at 520 sites in 20 countries during 2017-2019 and followed them on treatment for a median of 16 months. All patients had a left ventricular ejection fraction of 40% or less, and roughly three-quarters had New York Heart Association (NYHA) class II function, nearly one-quarter had class III function, and fewer than 1% of patients fell into the class IV category.
The primary endpoint occurred in 19% of the empagliflozin-treated patients and in 25% of those who received placebo. Among the half of patients with diabetes in the trial, the relative risk reduction by empagliflozin compared with placebo was a statistically significant 28%; among those without diabetes, it was a statistically significant 22%. Concurrently with Dr. Packer’s report, the results appeared in an article posted online (N Engl J Med. 2020 Aug 29. doi: 10.1056/NEJMoa2022190).
The study also had two main prespecified secondary endpoints: the incidence of total hospitalizations for heart failure, both first and recurrent, which fell by 30% in the empagliflozin-treated patients, compared with placebo, and the rate of declining renal function during the 16 months of the study as measured by estimated glomerular filtration rate, which dropped by roughly 1 mL/min per 1.73 m2 among the empagliflozin recipients and by about 4 mL/min/ per 1.73 m2 in the placebo patients.
Treatment with empagliflozin also achieved a notable, statistically significant 50% drop in major adverse renal events, consistent with the performance of other drugs in the class.
“Renal protection is a big plus” of empagliflozin in this trial and from the other SGLT2 inhibitors in prior studies, noted Dr. O’Connor.
The EMPEROR-Reduced results also showed an important benefit for HFrEF patients from empagliflozin not previously seen as quickly with any other drug class, noted Dr. Packer. The SGLT2 inhibitor led to statistically a significant slowing in the progression of patients from NYHA class II function to class III, compared with placebo, and it also significantly promoted the recovery of patients from NYHA class III to class II, an effect that became apparent within the first month on treatment and a benefit that is a “big deal” for patients because it represents a “significant change in functional capacity.” This additional dimension of empagliflozin’s benefit “really impressed me,” Dr. Packer said.
EMPEROR-Reduced was funded by Boehringer Ingelheim and Eli Lilly, the companies that market empagliflozin. Dr. Packer has received personal fees from Boehringer Ingelheim and Eli Lilly and from several other companies. Dr. Poppas and Dr. O’Connor had no relevant disclosures.
SOURCE: Packer M. ESC 2020. N Engl J Med. 2020 Aug 29. doi: 10.1056/NEJMoa2022190.
The SGLT2 inhibitor drug class solidified its role as a major, new treatment for patients with heart failure with reduced ejection fraction and no diabetes, with results from a second large, controlled trial showing clear efficacy and safety in this population.
Patients with heart failure with reduced ejection fraction (HFrEF) treated with the sodium glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) had a statistically significant 25% relative cut in their incidence of cardiovascular death or first heart failure hospitalization, compared with placebo-treated controls when added on top of standard HFrEF treatment, and this benefit was consistent regardless of whether the treated patients also had type 2 diabetes, Milton Packer, MD, reported at the virtual annual congress of the European Society of Cardiology.
This 25% drop in the primary endpoint with empagliflozin treatment in the EMPEROR-Reduced trial exactly matched the cut in incidence of cardiovascular death or heart failure hospitalization produced by treatment with a another SGLT2 inhibitor, dapagliflozin (Farxiga), in the DAPA-HF trial (N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
The performance of these two SGLT2 inhibitors was “incredibly consistent” across the their respective trials run in HFrEF patients with and without type 2 diabetes, and the combined evidence base of the two trials makes for “really compelling evidence” of both safety and efficacy that should prompt a change to U.S. practice, with both of these drugs forming a new cornerstone of HFrEF treatment, Dr. Packer said.
Results plant drug class firmly as HFrEF treatment
Dr. Packer stressed in his presentation that optimal treatment of patients with HFrEF now demands use of one of these two SGLT2 inhibitors, as well as sacubitril plus valsartan (Entresto), a beta-blocker, and a mineralocorticoid receptor antagonist, plus a diuretic as a fifth drug class for the many HFrEF patients who also need treatment for fluid overload. He further advocated for rapid introduction of these four cornerstone agents with proven survival benefits once a patient receives a HFrEF diagnosis, suggesting that sacubitril plus valsartan, an SGLT2 inhibitor, a beta-blocker, and a mineralocorticoid receptor antagonist could all be initiated within 6 weeks or less while acknowledging that optimal up-titration of the beta-blocker would likely take longer.
The order in which a patient starts these drugs shouldn’t matter, and there currently seems to be no evidence that clearly points toward using either dapagliflozin or empagliflozin over the other, Dr. Packer added.
In recognition of the importance of sending a message to heart failure clinicians about the newly proven efficacy of SGLT2 inhibitors in HFrEF patients, the American College of Cardiology and American Heart Association are now drafting an “expert decision pathway” to help clinicians as they enter this new prescribing space. This interim guidance should come out before the end of 2020, prior to release of fully revised HFrEF management guidelines in 2021, said Athena Poppas, MD, president of the ACC, in an interview.
“There is clearly need for education” that can help guide physicians who care for HFrEF patients on how to introduce an SGLT2 inhibitor along with the additional, lengthy list of drug classes proven to benefit these patients, noted Dr. Poppas, who is also a professor and chief of cardiology at the Brown University in Providence, R.I. Physicians may find that they need extra backup for successfully starting both sacubitril plus valsartan and an SGLT2 inhibitor in HFrEF patients because recent history has shown substantial pushback from third-party payers in reimbursing for these relatively expensive drugs, Dr. Poppas noted. She added that this is a problem that may be compounded when patients should ideally get both drug classes.
Physicians who care for heart failure patients have their own history of dragging their feet when adding new drugs to the regimens HFrEF patients receive. The angiotensin converting enzyme inhibitors and beta-blockers took about 17 years each to start reaching a majority of U.S. HFrEF patients, and sacubitril plus valsartan is now used on perhaps a quarter to a third of HFrEF patients despite receiving Food and Drug Administration approval for these patients in mid 2015, noted Christopher M. O’Connor, MD, a heart failure specialist and president of the Inova Heart and Vascular Institute in Fairfax, Va.
Despite dapagliflozin receiving FDA approval in May 2020 for treating HFrEF in patients without diabetes, early uptake in U.S. practice has been very slow, with findings from large U.S. patient registries suggesting that perhaps 1% of suitable HFrEF patients currently get the drug, estimated Dr. O’Connor in an interview.
Given how strong the evidence now is for benefit and safety from dapagliflozin and empagliflozin, it may take as little as 5 years to reach greater than 50% penetration of one of these drugs into U.S. HFrEF patient populations, suggested Dr. Packer, a distinguished scholar in cardiovascular science at Baylor University Medical Center in Dallas.
EMPEROR-Reduced outcomes
The road to routine use of these SGLT2 inhibitor drugs should be hastened by empagliflozin’s impressive performance in EMPEROR-Reduced, in which the drug scored highly significant benefits over placebo for the prespecified primary and two major secondary endpoints, one of which was a measure of preserved renal function.
The trial randomized 3,730 patients at 520 sites in 20 countries during 2017-2019 and followed them on treatment for a median of 16 months. All patients had a left ventricular ejection fraction of 40% or less, and roughly three-quarters had New York Heart Association (NYHA) class II function, nearly one-quarter had class III function, and fewer than 1% of patients fell into the class IV category.
The primary endpoint occurred in 19% of the empagliflozin-treated patients and in 25% of those who received placebo. Among the half of patients with diabetes in the trial, the relative risk reduction by empagliflozin compared with placebo was a statistically significant 28%; among those without diabetes, it was a statistically significant 22%. Concurrently with Dr. Packer’s report, the results appeared in an article posted online (N Engl J Med. 2020 Aug 29. doi: 10.1056/NEJMoa2022190).
The study also had two main prespecified secondary endpoints: the incidence of total hospitalizations for heart failure, both first and recurrent, which fell by 30% in the empagliflozin-treated patients, compared with placebo, and the rate of declining renal function during the 16 months of the study as measured by estimated glomerular filtration rate, which dropped by roughly 1 mL/min per 1.73 m2 among the empagliflozin recipients and by about 4 mL/min/ per 1.73 m2 in the placebo patients.
Treatment with empagliflozin also achieved a notable, statistically significant 50% drop in major adverse renal events, consistent with the performance of other drugs in the class.
“Renal protection is a big plus” of empagliflozin in this trial and from the other SGLT2 inhibitors in prior studies, noted Dr. O’Connor.
The EMPEROR-Reduced results also showed an important benefit for HFrEF patients from empagliflozin not previously seen as quickly with any other drug class, noted Dr. Packer. The SGLT2 inhibitor led to statistically a significant slowing in the progression of patients from NYHA class II function to class III, compared with placebo, and it also significantly promoted the recovery of patients from NYHA class III to class II, an effect that became apparent within the first month on treatment and a benefit that is a “big deal” for patients because it represents a “significant change in functional capacity.” This additional dimension of empagliflozin’s benefit “really impressed me,” Dr. Packer said.
EMPEROR-Reduced was funded by Boehringer Ingelheim and Eli Lilly, the companies that market empagliflozin. Dr. Packer has received personal fees from Boehringer Ingelheim and Eli Lilly and from several other companies. Dr. Poppas and Dr. O’Connor had no relevant disclosures.
SOURCE: Packer M. ESC 2020. N Engl J Med. 2020 Aug 29. doi: 10.1056/NEJMoa2022190.
FROM ESC CONGRESS 2020