User login
The multiple meanings of sex
Knowing the sex of a developing fetus is a common question many expectant parents ask at their prenatal appointments. While the sex of a fetus has minimal clinical significance to obstetrician/gynecologists, technology has made ascertaining the answer to this question much more accessible.
In addition to detecting certain genetic abnormalities, both noninvasive prenatal testing (NIPT) and preimplantation genetic testing (PGT) can discern the chromosomal sex of a fetus prior to birth. At the 20-week anatomy scan, the ultrasonographer can detect the presence of external genitalia to determine the sex. In fact, when a baby is first born, obstetrician/gynecologists are consistently asked “do I have a boy or a girl?” Assigning the sex of a newborn is one of the many tasks we complete in the delivery room. However, some of you reading this article would disagree.
“You cannot assign sex at birth.” “Sex is fixed, you cannot change biology.” These are examples of statements that frequent the comments section of my medical articles and plague professionals who treat gender diverse patients. I would argue, as would many biologists, scientists, and physicians, that these statements oversimplify biologic reality.
The term “sex” has multiple meanings: It can allude to the act of reproduction itself, but in the context of sexual determination and sexual differentiation, it can refer to the biologic and structural composition of a developing human. Within this paradigm, there exist three definitions for sex: chromosomal, gonadal, and phenotypic.
Chromosomal sex refers to the genetic makeup of a human, typically XX or XY chromosomes. There are also variations within this seemingly binary system. Embryos can have an extra sex chromosome, as seen in Klinefelter syndrome, which is characterized by XXY karyotype. Embryos can also be devoid of a sex chromosome, as observed in Turner’s syndrome, which is characterized by an XO karyotype. These variations can impact fertility and expression of secondary sexual characteristics as the type of sex chromosomes present results in primary sex determination, or the development of gonads.
Most often, individuals with a chromosomal makeup of XX are considered female and will subsequently develop ovaries that produce oocytes (eggs). Individuals with XY chromosomes are deemed male and will go on to develop testes, which are responsible for spermatogenesis (sperm production).
Gonadal sex is the presence of either testes or ovaries. The primary function of testes is to produce sperm for reproduction and to secrete testosterone, the primary male sex hormone. Similarly, ovaries produce oocytes and secrete estrogen as the primary female sex hormone. Gonads can be surgically removed either via orchiectomy (the removal of testes), or oophorectomy (the removal of ovaries) for a variety of reasons. There is no current medical technology that can replace the function of these structures, although patients can be placed on hormone replacement to counter the negative physiologic consequences resulting from their removal.
Secondary sex determination, or sexual differentiation, is the development of external genitalia and internal genital tracts because of the hormones produced from the gonads. At puberty, further differentiation occurs with the development of pubic and axillary hair and breast growth. This process determines phenotypic sex – the visible distinction between male and female.
When opponents of gender affirming care state that individuals cannot change sex, are they correct or false? The answer to this question is entirely dependent on which definition of sex they are using. Chromosomal? Gonadal? Phenotypic? It is an immutable fact that humans cannot change chromosomal sex. No one in the transgender community, either provider or patient, would dispute this. However, we can remove gonadal structures and alter phenotypic sex.
In fact, many cisgender individuals also revise their phenotypic sex when they undergo augmentation mammaplasty, penile enlargement, or vulvoplasty procedures for the exact same reason.
Circling back to the debate about whether we can “assign sex at birth,” it all depends on what definition of sex you are referencing. At birth, obstetrician/gynecologists most often look at the phenotypic sex and make assumptions about the genetic and gonadal sex based on the secondary sexual characteristics. So yes, we can, and we do assign sex at birth. However, in the case of intersex individuals, these physical characteristics may not align with their gonadal and chromosomal composition.
In the case of an infant that has a known XY karyotype prior to birth but a female phenotype at birth (as seen in a condition called complete androgen insensitivity syndrome), what sex should be assigned to that baby? Should the infant be raised male or female? A lot of unintended but significant harm has resulted from providers and parents trying to answer that very question. The mistreatment of intersex patients through forced and coercive medical and surgical treatments, often in infancy, should serve as a dark reminder that sex and gender are not as biologically binary as we would like to believe.
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa. She has no relevant disclosures.
References
Moore KL and Persaud TVN. The urogenital system. In: Before we are born: essentials of embryology and birth defects. 7th ed. Philadelphia: Saunders Elsevier;2008:163-89.
Standring S. Development of the urogenital system. In: Gray’s Anatomy, 42nd ed. Philadelphia: Elsevier;2021:341-64.
Escobar O et al. Pediatric endocrinology. In: Zitelli BJ, ed. Zitelli and Davis’ atlas of pediatric physical diagnosis 8th edition. Philadelphia: Elsevier;2023:342-81.
Knowing the sex of a developing fetus is a common question many expectant parents ask at their prenatal appointments. While the sex of a fetus has minimal clinical significance to obstetrician/gynecologists, technology has made ascertaining the answer to this question much more accessible.
In addition to detecting certain genetic abnormalities, both noninvasive prenatal testing (NIPT) and preimplantation genetic testing (PGT) can discern the chromosomal sex of a fetus prior to birth. At the 20-week anatomy scan, the ultrasonographer can detect the presence of external genitalia to determine the sex. In fact, when a baby is first born, obstetrician/gynecologists are consistently asked “do I have a boy or a girl?” Assigning the sex of a newborn is one of the many tasks we complete in the delivery room. However, some of you reading this article would disagree.
“You cannot assign sex at birth.” “Sex is fixed, you cannot change biology.” These are examples of statements that frequent the comments section of my medical articles and plague professionals who treat gender diverse patients. I would argue, as would many biologists, scientists, and physicians, that these statements oversimplify biologic reality.
The term “sex” has multiple meanings: It can allude to the act of reproduction itself, but in the context of sexual determination and sexual differentiation, it can refer to the biologic and structural composition of a developing human. Within this paradigm, there exist three definitions for sex: chromosomal, gonadal, and phenotypic.
Chromosomal sex refers to the genetic makeup of a human, typically XX or XY chromosomes. There are also variations within this seemingly binary system. Embryos can have an extra sex chromosome, as seen in Klinefelter syndrome, which is characterized by XXY karyotype. Embryos can also be devoid of a sex chromosome, as observed in Turner’s syndrome, which is characterized by an XO karyotype. These variations can impact fertility and expression of secondary sexual characteristics as the type of sex chromosomes present results in primary sex determination, or the development of gonads.
Most often, individuals with a chromosomal makeup of XX are considered female and will subsequently develop ovaries that produce oocytes (eggs). Individuals with XY chromosomes are deemed male and will go on to develop testes, which are responsible for spermatogenesis (sperm production).
Gonadal sex is the presence of either testes or ovaries. The primary function of testes is to produce sperm for reproduction and to secrete testosterone, the primary male sex hormone. Similarly, ovaries produce oocytes and secrete estrogen as the primary female sex hormone. Gonads can be surgically removed either via orchiectomy (the removal of testes), or oophorectomy (the removal of ovaries) for a variety of reasons. There is no current medical technology that can replace the function of these structures, although patients can be placed on hormone replacement to counter the negative physiologic consequences resulting from their removal.
Secondary sex determination, or sexual differentiation, is the development of external genitalia and internal genital tracts because of the hormones produced from the gonads. At puberty, further differentiation occurs with the development of pubic and axillary hair and breast growth. This process determines phenotypic sex – the visible distinction between male and female.
When opponents of gender affirming care state that individuals cannot change sex, are they correct or false? The answer to this question is entirely dependent on which definition of sex they are using. Chromosomal? Gonadal? Phenotypic? It is an immutable fact that humans cannot change chromosomal sex. No one in the transgender community, either provider or patient, would dispute this. However, we can remove gonadal structures and alter phenotypic sex.
In fact, many cisgender individuals also revise their phenotypic sex when they undergo augmentation mammaplasty, penile enlargement, or vulvoplasty procedures for the exact same reason.
Circling back to the debate about whether we can “assign sex at birth,” it all depends on what definition of sex you are referencing. At birth, obstetrician/gynecologists most often look at the phenotypic sex and make assumptions about the genetic and gonadal sex based on the secondary sexual characteristics. So yes, we can, and we do assign sex at birth. However, in the case of intersex individuals, these physical characteristics may not align with their gonadal and chromosomal composition.
In the case of an infant that has a known XY karyotype prior to birth but a female phenotype at birth (as seen in a condition called complete androgen insensitivity syndrome), what sex should be assigned to that baby? Should the infant be raised male or female? A lot of unintended but significant harm has resulted from providers and parents trying to answer that very question. The mistreatment of intersex patients through forced and coercive medical and surgical treatments, often in infancy, should serve as a dark reminder that sex and gender are not as biologically binary as we would like to believe.
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa. She has no relevant disclosures.
References
Moore KL and Persaud TVN. The urogenital system. In: Before we are born: essentials of embryology and birth defects. 7th ed. Philadelphia: Saunders Elsevier;2008:163-89.
Standring S. Development of the urogenital system. In: Gray’s Anatomy, 42nd ed. Philadelphia: Elsevier;2021:341-64.
Escobar O et al. Pediatric endocrinology. In: Zitelli BJ, ed. Zitelli and Davis’ atlas of pediatric physical diagnosis 8th edition. Philadelphia: Elsevier;2023:342-81.
Knowing the sex of a developing fetus is a common question many expectant parents ask at their prenatal appointments. While the sex of a fetus has minimal clinical significance to obstetrician/gynecologists, technology has made ascertaining the answer to this question much more accessible.
In addition to detecting certain genetic abnormalities, both noninvasive prenatal testing (NIPT) and preimplantation genetic testing (PGT) can discern the chromosomal sex of a fetus prior to birth. At the 20-week anatomy scan, the ultrasonographer can detect the presence of external genitalia to determine the sex. In fact, when a baby is first born, obstetrician/gynecologists are consistently asked “do I have a boy or a girl?” Assigning the sex of a newborn is one of the many tasks we complete in the delivery room. However, some of you reading this article would disagree.
“You cannot assign sex at birth.” “Sex is fixed, you cannot change biology.” These are examples of statements that frequent the comments section of my medical articles and plague professionals who treat gender diverse patients. I would argue, as would many biologists, scientists, and physicians, that these statements oversimplify biologic reality.
The term “sex” has multiple meanings: It can allude to the act of reproduction itself, but in the context of sexual determination and sexual differentiation, it can refer to the biologic and structural composition of a developing human. Within this paradigm, there exist three definitions for sex: chromosomal, gonadal, and phenotypic.
Chromosomal sex refers to the genetic makeup of a human, typically XX or XY chromosomes. There are also variations within this seemingly binary system. Embryos can have an extra sex chromosome, as seen in Klinefelter syndrome, which is characterized by XXY karyotype. Embryos can also be devoid of a sex chromosome, as observed in Turner’s syndrome, which is characterized by an XO karyotype. These variations can impact fertility and expression of secondary sexual characteristics as the type of sex chromosomes present results in primary sex determination, or the development of gonads.
Most often, individuals with a chromosomal makeup of XX are considered female and will subsequently develop ovaries that produce oocytes (eggs). Individuals with XY chromosomes are deemed male and will go on to develop testes, which are responsible for spermatogenesis (sperm production).
Gonadal sex is the presence of either testes or ovaries. The primary function of testes is to produce sperm for reproduction and to secrete testosterone, the primary male sex hormone. Similarly, ovaries produce oocytes and secrete estrogen as the primary female sex hormone. Gonads can be surgically removed either via orchiectomy (the removal of testes), or oophorectomy (the removal of ovaries) for a variety of reasons. There is no current medical technology that can replace the function of these structures, although patients can be placed on hormone replacement to counter the negative physiologic consequences resulting from their removal.
Secondary sex determination, or sexual differentiation, is the development of external genitalia and internal genital tracts because of the hormones produced from the gonads. At puberty, further differentiation occurs with the development of pubic and axillary hair and breast growth. This process determines phenotypic sex – the visible distinction between male and female.
When opponents of gender affirming care state that individuals cannot change sex, are they correct or false? The answer to this question is entirely dependent on which definition of sex they are using. Chromosomal? Gonadal? Phenotypic? It is an immutable fact that humans cannot change chromosomal sex. No one in the transgender community, either provider or patient, would dispute this. However, we can remove gonadal structures and alter phenotypic sex.
In fact, many cisgender individuals also revise their phenotypic sex when they undergo augmentation mammaplasty, penile enlargement, or vulvoplasty procedures for the exact same reason.
Circling back to the debate about whether we can “assign sex at birth,” it all depends on what definition of sex you are referencing. At birth, obstetrician/gynecologists most often look at the phenotypic sex and make assumptions about the genetic and gonadal sex based on the secondary sexual characteristics. So yes, we can, and we do assign sex at birth. However, in the case of intersex individuals, these physical characteristics may not align with their gonadal and chromosomal composition.
In the case of an infant that has a known XY karyotype prior to birth but a female phenotype at birth (as seen in a condition called complete androgen insensitivity syndrome), what sex should be assigned to that baby? Should the infant be raised male or female? A lot of unintended but significant harm has resulted from providers and parents trying to answer that very question. The mistreatment of intersex patients through forced and coercive medical and surgical treatments, often in infancy, should serve as a dark reminder that sex and gender are not as biologically binary as we would like to believe.
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa. She has no relevant disclosures.
References
Moore KL and Persaud TVN. The urogenital system. In: Before we are born: essentials of embryology and birth defects. 7th ed. Philadelphia: Saunders Elsevier;2008:163-89.
Standring S. Development of the urogenital system. In: Gray’s Anatomy, 42nd ed. Philadelphia: Elsevier;2021:341-64.
Escobar O et al. Pediatric endocrinology. In: Zitelli BJ, ed. Zitelli and Davis’ atlas of pediatric physical diagnosis 8th edition. Philadelphia: Elsevier;2023:342-81.
Digital tool clarifies menopause symptoms
GLASGOW – An interactive digital decision tool that individualizes menopause care received praise from primary care clinicians in the United Kingdom, who said it could improve patient care and streamline office visits.
“Access to hormone replacement therapy [HRT], as well as decision-making around treatment for menopausal symptoms, is often complicated by concerns around its safety, and there is still a knowledge and a confidence gap among health care professionals causing reluctance to prescribe HRT,” said Aini Kamal, MSc, from University College London. Ms. Kamal presented results of a survey about the tool at the annual meeting of the Royal College of General Practitioners.
For the study, Ms. Kamal, Daniel Reisel, MBBS, PhD, a gynecologist at UCL, and colleagues evaluated Wellspring with doctors, nurses, and pharmacists.
“Ensuring that women receive education around symptoms, so that they are empowered, is a key part of optimizing their care and sharing decision-making,” Dr. Reisel said in an interview. He added that U.K. primary care had seen an increase in cases of women presenting with symptoms associated with the perimenopause and menopause at a time when U.K. Members of Parliament are debating whether to make it mandatory for all women to have menopause check-up in their early 40s.
The online survey was completed by 280 participants, and respondents were primarily GPs with several years of relevant prescribing practice. Of those, 93% found information from national guidelines to be accurately presented in the tool, and 97% said they would recommend this decision aid to other health care professionals, Ms. Kamal reported.
Nearly all participants said they could see themselves using the tool with patients in the clinic or as an adjunct to virtual sessions. “This [finding] was particularly important because it demonstrates the clinical potential this tool has,” she said.
One consult, too many problems
Louise Newson, MBChB, who runs the U.K.’s largest menopause clinic, said primary care appointments are often time-pressured and follow a “’one problem-one consultation’” policy. As such, women are often thinking ‘Do I go with my joint pains, or my palpitations, tinnitus, or what?’ If a patient presents with tinnitus, a doctor might focus on the potential of an inner ear problem rather than a hormone deficiency, but I do know that if the woman is perimenopausal or menopausal, we often look to replace the missing hormones, and then if the tinnitus doesn’t improve we can revisit the ear problem.”
Dr. Newson noted that 17% of women in her clinics have had more than six GP visits in the year before she sees them, but in the year following, this figure drops to 1%. Acknowledging that a menopause consultation for a GP is time-consuming, Dr. Newson pointed out that taking time initially with the patient “means it will reduce the number of future consultations quickly, but more importantly, we also know that taking HRT reduces long-term risk of serious diseases, including heart disease and osteoporosis.”
The digital tool can be used by both doctors and patients to help women work through their symptoms and equip them with knowledge so their GP visits are more productive.
“When we see women who are empowered with knowledge [about menopause symptoms], then the consultations are quicker and essentially place the patient central to the discussion,” Dr. Newson said.
Ed Russell-Smith, MBChB, a GP in Scotland who moderated the session, said the tool “lays out a nicely structured approach and provides modern treatment options and resources for patients.”
However, he added “we also need to remember there are potential harms to be done from HRT too. It’s vitally important that while patients might see HRT as a panacea, doctors need to balance this with the risks involved for each individual. As a tool, I think Wellspring can help us in this respect to apply general principles to that patient and individualize treatment.”
Dr. Reisel, Dr. Newson, Ms. Kamal, and Dr. Russell-Smith disclosed no relevant financial relationships. The Wellspring Decision Aid was supported by UCL’s Institute for Women’s Health. The Newson Health clinic is fully private, but research is done via the nonprofit arm, which is supported by the clinic. There is no pharma involvement.
A version of this article first appeared on Medscape.com.
GLASGOW – An interactive digital decision tool that individualizes menopause care received praise from primary care clinicians in the United Kingdom, who said it could improve patient care and streamline office visits.
“Access to hormone replacement therapy [HRT], as well as decision-making around treatment for menopausal symptoms, is often complicated by concerns around its safety, and there is still a knowledge and a confidence gap among health care professionals causing reluctance to prescribe HRT,” said Aini Kamal, MSc, from University College London. Ms. Kamal presented results of a survey about the tool at the annual meeting of the Royal College of General Practitioners.
For the study, Ms. Kamal, Daniel Reisel, MBBS, PhD, a gynecologist at UCL, and colleagues evaluated Wellspring with doctors, nurses, and pharmacists.
“Ensuring that women receive education around symptoms, so that they are empowered, is a key part of optimizing their care and sharing decision-making,” Dr. Reisel said in an interview. He added that U.K. primary care had seen an increase in cases of women presenting with symptoms associated with the perimenopause and menopause at a time when U.K. Members of Parliament are debating whether to make it mandatory for all women to have menopause check-up in their early 40s.
The online survey was completed by 280 participants, and respondents were primarily GPs with several years of relevant prescribing practice. Of those, 93% found information from national guidelines to be accurately presented in the tool, and 97% said they would recommend this decision aid to other health care professionals, Ms. Kamal reported.
Nearly all participants said they could see themselves using the tool with patients in the clinic or as an adjunct to virtual sessions. “This [finding] was particularly important because it demonstrates the clinical potential this tool has,” she said.
One consult, too many problems
Louise Newson, MBChB, who runs the U.K.’s largest menopause clinic, said primary care appointments are often time-pressured and follow a “’one problem-one consultation’” policy. As such, women are often thinking ‘Do I go with my joint pains, or my palpitations, tinnitus, or what?’ If a patient presents with tinnitus, a doctor might focus on the potential of an inner ear problem rather than a hormone deficiency, but I do know that if the woman is perimenopausal or menopausal, we often look to replace the missing hormones, and then if the tinnitus doesn’t improve we can revisit the ear problem.”
Dr. Newson noted that 17% of women in her clinics have had more than six GP visits in the year before she sees them, but in the year following, this figure drops to 1%. Acknowledging that a menopause consultation for a GP is time-consuming, Dr. Newson pointed out that taking time initially with the patient “means it will reduce the number of future consultations quickly, but more importantly, we also know that taking HRT reduces long-term risk of serious diseases, including heart disease and osteoporosis.”
The digital tool can be used by both doctors and patients to help women work through their symptoms and equip them with knowledge so their GP visits are more productive.
“When we see women who are empowered with knowledge [about menopause symptoms], then the consultations are quicker and essentially place the patient central to the discussion,” Dr. Newson said.
Ed Russell-Smith, MBChB, a GP in Scotland who moderated the session, said the tool “lays out a nicely structured approach and provides modern treatment options and resources for patients.”
However, he added “we also need to remember there are potential harms to be done from HRT too. It’s vitally important that while patients might see HRT as a panacea, doctors need to balance this with the risks involved for each individual. As a tool, I think Wellspring can help us in this respect to apply general principles to that patient and individualize treatment.”
Dr. Reisel, Dr. Newson, Ms. Kamal, and Dr. Russell-Smith disclosed no relevant financial relationships. The Wellspring Decision Aid was supported by UCL’s Institute for Women’s Health. The Newson Health clinic is fully private, but research is done via the nonprofit arm, which is supported by the clinic. There is no pharma involvement.
A version of this article first appeared on Medscape.com.
GLASGOW – An interactive digital decision tool that individualizes menopause care received praise from primary care clinicians in the United Kingdom, who said it could improve patient care and streamline office visits.
“Access to hormone replacement therapy [HRT], as well as decision-making around treatment for menopausal symptoms, is often complicated by concerns around its safety, and there is still a knowledge and a confidence gap among health care professionals causing reluctance to prescribe HRT,” said Aini Kamal, MSc, from University College London. Ms. Kamal presented results of a survey about the tool at the annual meeting of the Royal College of General Practitioners.
For the study, Ms. Kamal, Daniel Reisel, MBBS, PhD, a gynecologist at UCL, and colleagues evaluated Wellspring with doctors, nurses, and pharmacists.
“Ensuring that women receive education around symptoms, so that they are empowered, is a key part of optimizing their care and sharing decision-making,” Dr. Reisel said in an interview. He added that U.K. primary care had seen an increase in cases of women presenting with symptoms associated with the perimenopause and menopause at a time when U.K. Members of Parliament are debating whether to make it mandatory for all women to have menopause check-up in their early 40s.
The online survey was completed by 280 participants, and respondents were primarily GPs with several years of relevant prescribing practice. Of those, 93% found information from national guidelines to be accurately presented in the tool, and 97% said they would recommend this decision aid to other health care professionals, Ms. Kamal reported.
Nearly all participants said they could see themselves using the tool with patients in the clinic or as an adjunct to virtual sessions. “This [finding] was particularly important because it demonstrates the clinical potential this tool has,” she said.
One consult, too many problems
Louise Newson, MBChB, who runs the U.K.’s largest menopause clinic, said primary care appointments are often time-pressured and follow a “’one problem-one consultation’” policy. As such, women are often thinking ‘Do I go with my joint pains, or my palpitations, tinnitus, or what?’ If a patient presents with tinnitus, a doctor might focus on the potential of an inner ear problem rather than a hormone deficiency, but I do know that if the woman is perimenopausal or menopausal, we often look to replace the missing hormones, and then if the tinnitus doesn’t improve we can revisit the ear problem.”
Dr. Newson noted that 17% of women in her clinics have had more than six GP visits in the year before she sees them, but in the year following, this figure drops to 1%. Acknowledging that a menopause consultation for a GP is time-consuming, Dr. Newson pointed out that taking time initially with the patient “means it will reduce the number of future consultations quickly, but more importantly, we also know that taking HRT reduces long-term risk of serious diseases, including heart disease and osteoporosis.”
The digital tool can be used by both doctors and patients to help women work through their symptoms and equip them with knowledge so their GP visits are more productive.
“When we see women who are empowered with knowledge [about menopause symptoms], then the consultations are quicker and essentially place the patient central to the discussion,” Dr. Newson said.
Ed Russell-Smith, MBChB, a GP in Scotland who moderated the session, said the tool “lays out a nicely structured approach and provides modern treatment options and resources for patients.”
However, he added “we also need to remember there are potential harms to be done from HRT too. It’s vitally important that while patients might see HRT as a panacea, doctors need to balance this with the risks involved for each individual. As a tool, I think Wellspring can help us in this respect to apply general principles to that patient and individualize treatment.”
Dr. Reisel, Dr. Newson, Ms. Kamal, and Dr. Russell-Smith disclosed no relevant financial relationships. The Wellspring Decision Aid was supported by UCL’s Institute for Women’s Health. The Newson Health clinic is fully private, but research is done via the nonprofit arm, which is supported by the clinic. There is no pharma involvement.
A version of this article first appeared on Medscape.com.
AT RCGP 2023
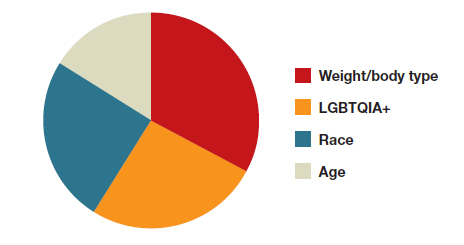
Why aren’t doctors managing pain during gynecologic procedures?
During a fellowship rotation in gynecology, Rebekah D. Fenton, MD, asked the attending physicians what pain management options they could offer patients for insertion of an intrauterine device (IUD). Their answer surprised her: None.
The research on the effectiveness of pain management techniques during the procedure were not strong enough to warrant providing potential relief.
But Dr. Fenton knew the attending physician was wrong: She’d received the drug lidocaine during a recent visit to her own ob.gyn. to get an IUD placed. The local anesthetic enabled her to avoid the experiences of many patients who often withstand debilitating cramping and pain during insertion, side effects that can last for hours after the procedure has ended.
By not teaching her how to administer pain treatment options such as lidocaine gel or injection, “they made the decision for me, whether I could give patients this option,” said Dr. Fenton, now an adolescent medicine specialist at Alivio Medical Center in Chicago.
As a result, patients undergoing IUD placements, biopsies, hysteroscopies, and pelvic exams are often subject to pain that could be mitigated.
Some research suggests simple numbing agents, including lidocaine, may induce less pain without the need for full anesthesia. But clinicians don’t always present these options.
During gynecologic procedures, the amount of pain a patient can expect is often downplayed by clinicians. Because every patient experiences the sensation differently, discussing options for pain management and the range of possible pain is paramount in building patient-clinician trust, and ultimately providing the best care for patients in the long run, according to Megan Wasson, DO, chair of the department of medical and surgical gynecology at Mayo Clinic Arizona in Phoenix.
“It comes down to shared decision-making so the patient is aware of the pain that should be expected and what avenue they want to go down,” Dr. Wasson said. “It’s not a one-size-fits-all.”
Lack of uniform protocols
The American College of Obstetricians and Gynecologists (ACOG) has clear guidelines for pain management during pregnancy and delivery but not for many routine gynecologic procedures. Some experts say not offering options for pain management based on lack of efficacy evidence can undermine a patient’s experience.
ACOG does have recommendations for reducing dilation pain during a hysteroscopy, including providing intravaginal misoprostol and estrogen. The organization also recommends performing a vaginoscopy instead if possible because the procedure is typically less painful than is a hysteroscopy.
For an IUD placement, ACOG states that the procedure “may cause temporary discomfort” and recommends that patients take over-the-counter pain relief before a procedure. The most recent clinical bulletin on the topic, published in 2016, states routine misoprostol is not recommended for IUD placement, although it may be considered with difficult insertions for management of pain.
A clinical inquiry published in 2020 outlined the efficacy of several pain options that practitioners can weigh with patients. The inquiry cited a 2019 meta-analysis of 38 studies that found lidocaine-prilocaine cream to be the most effective option for pain management during IUD placement, reducing insertion pain by nearly 30%. The inquiry concluded that a combination of 600 mcg of misoprostol and 4% lidocaine gel may be effective, while lower dosages of both drugs were not effective. A 2018 clinical trial cited in the analysis found that though a 20-cc 1% lidocaine paracervical block on its own did not reduce pain, the block mixed with sodium bicarbonate reduced pain during IUD insertion by 22%.
Some doctors make the decision to not use lidocaine without offering it to patients first, according to Dr. Fenton. Instead, clinicians should discuss any potential drawbacks, such as pain from administering the numbing agent with a needle or the procedure taking extra time while the patient waits for the lidocaine to kick in.
“That always felt unfair, to make that decision for [the patient],” Dr. Fenton said.
Often clinicians won’t know how a patient will respond to a procedure: A 2014 secondary analysis of a clinical trial compared how patients rated their pain after an IUD procedure to the amount of pain physicians perceived the procedure to cause. They found that the average pain scores patients reported were nearly twice as high as clinician expectations were.
ACOG’s guideline states that the evidence backing paracervical blocks and lidocaine to IUD insertion pain is controversial. The American College of Physicians also cites “low-quality evidence” to support patient reports of pain and discomfort during pelvic exams. Some studies have found up to 60% of women report these negative experiences.
The varying evidence highlights the need for a personalized approach – one that includes patients – to pain management for routine gynecological procedures.
“Usually patients are pretty good predictors,” said Lisa Bayer, MD, MPH, associate professor of obstetrics and gynecology at Oregon Health & Science University in Portland. “They can anticipate what different things are going to feel like based on previous experiences.”
Making patients part of the discussion
Clinicians should have open discussions with patients about their past experiences and current anxieties about a gynecologic procedure, according to Dr. Bayer.
“Part of it is just creating a really safe environment of trust as a medical provider,” she said.
A study published in 2016 of more than 800 patients undergoing oocyte retrieval, which has clear protocols for pain management, found that previous negative gynecologic experiences were significantly correlated to greater amounts of pain reported during the procedure.
If pain isn’t properly managed, patients may avoid care in the future, putting them at risk for unplanned pregnancies, skipped cancer screenings, and complications from undiagnosed conditions and infections, Dr. Bayer added. Clinician offices will not always have access to all pain management options, so making referrals to another physician who has access to the appropriate technique may be the best thing for the patient, Dr. Bayer said.
Downplaying the experience
Informing a patient that she will feel only a little discomfort during a procedure – when a clinician doesn’t know how exactly the patient will react – can also result in distrust.
When a clinician says, “ ’It’s only going to be a little cramp, it’s only going to be a little pinch,’ we know extreme pain is a possibility, we’ve seen it,” Dr. Fenton said. “But if we choose to disregard that [possibility], it feels invalidating for patients.”
Failing to fully explain the possible pain scale can also directly interfere with the procedure at hand.
“My first concern is if they aren’t anticipating the amount of pain they are going to experience, they may move; For biopsies and IUD insertions, we need them to be still,” Dr. Wasson said. “If they are unable to tolerate the procedure, we’ve put them through pain and not been able to accomplish the primary goal.”
Managing both pain and what patients can expect is even more crucial for adolescent and teenage patients who are often having their first gynecologic experience.
“We’re framing what these experiences look like,” Dr. Fenton said. “That means there are opportunities for creating a space that builds trust and security for the patients moving forward.”
A version of this article first appeared on Medscape.com.
During a fellowship rotation in gynecology, Rebekah D. Fenton, MD, asked the attending physicians what pain management options they could offer patients for insertion of an intrauterine device (IUD). Their answer surprised her: None.
The research on the effectiveness of pain management techniques during the procedure were not strong enough to warrant providing potential relief.
But Dr. Fenton knew the attending physician was wrong: She’d received the drug lidocaine during a recent visit to her own ob.gyn. to get an IUD placed. The local anesthetic enabled her to avoid the experiences of many patients who often withstand debilitating cramping and pain during insertion, side effects that can last for hours after the procedure has ended.
By not teaching her how to administer pain treatment options such as lidocaine gel or injection, “they made the decision for me, whether I could give patients this option,” said Dr. Fenton, now an adolescent medicine specialist at Alivio Medical Center in Chicago.
As a result, patients undergoing IUD placements, biopsies, hysteroscopies, and pelvic exams are often subject to pain that could be mitigated.
Some research suggests simple numbing agents, including lidocaine, may induce less pain without the need for full anesthesia. But clinicians don’t always present these options.
During gynecologic procedures, the amount of pain a patient can expect is often downplayed by clinicians. Because every patient experiences the sensation differently, discussing options for pain management and the range of possible pain is paramount in building patient-clinician trust, and ultimately providing the best care for patients in the long run, according to Megan Wasson, DO, chair of the department of medical and surgical gynecology at Mayo Clinic Arizona in Phoenix.
“It comes down to shared decision-making so the patient is aware of the pain that should be expected and what avenue they want to go down,” Dr. Wasson said. “It’s not a one-size-fits-all.”
Lack of uniform protocols
The American College of Obstetricians and Gynecologists (ACOG) has clear guidelines for pain management during pregnancy and delivery but not for many routine gynecologic procedures. Some experts say not offering options for pain management based on lack of efficacy evidence can undermine a patient’s experience.
ACOG does have recommendations for reducing dilation pain during a hysteroscopy, including providing intravaginal misoprostol and estrogen. The organization also recommends performing a vaginoscopy instead if possible because the procedure is typically less painful than is a hysteroscopy.
For an IUD placement, ACOG states that the procedure “may cause temporary discomfort” and recommends that patients take over-the-counter pain relief before a procedure. The most recent clinical bulletin on the topic, published in 2016, states routine misoprostol is not recommended for IUD placement, although it may be considered with difficult insertions for management of pain.
A clinical inquiry published in 2020 outlined the efficacy of several pain options that practitioners can weigh with patients. The inquiry cited a 2019 meta-analysis of 38 studies that found lidocaine-prilocaine cream to be the most effective option for pain management during IUD placement, reducing insertion pain by nearly 30%. The inquiry concluded that a combination of 600 mcg of misoprostol and 4% lidocaine gel may be effective, while lower dosages of both drugs were not effective. A 2018 clinical trial cited in the analysis found that though a 20-cc 1% lidocaine paracervical block on its own did not reduce pain, the block mixed with sodium bicarbonate reduced pain during IUD insertion by 22%.
Some doctors make the decision to not use lidocaine without offering it to patients first, according to Dr. Fenton. Instead, clinicians should discuss any potential drawbacks, such as pain from administering the numbing agent with a needle or the procedure taking extra time while the patient waits for the lidocaine to kick in.
“That always felt unfair, to make that decision for [the patient],” Dr. Fenton said.
Often clinicians won’t know how a patient will respond to a procedure: A 2014 secondary analysis of a clinical trial compared how patients rated their pain after an IUD procedure to the amount of pain physicians perceived the procedure to cause. They found that the average pain scores patients reported were nearly twice as high as clinician expectations were.
ACOG’s guideline states that the evidence backing paracervical blocks and lidocaine to IUD insertion pain is controversial. The American College of Physicians also cites “low-quality evidence” to support patient reports of pain and discomfort during pelvic exams. Some studies have found up to 60% of women report these negative experiences.
The varying evidence highlights the need for a personalized approach – one that includes patients – to pain management for routine gynecological procedures.
“Usually patients are pretty good predictors,” said Lisa Bayer, MD, MPH, associate professor of obstetrics and gynecology at Oregon Health & Science University in Portland. “They can anticipate what different things are going to feel like based on previous experiences.”
Making patients part of the discussion
Clinicians should have open discussions with patients about their past experiences and current anxieties about a gynecologic procedure, according to Dr. Bayer.
“Part of it is just creating a really safe environment of trust as a medical provider,” she said.
A study published in 2016 of more than 800 patients undergoing oocyte retrieval, which has clear protocols for pain management, found that previous negative gynecologic experiences were significantly correlated to greater amounts of pain reported during the procedure.
If pain isn’t properly managed, patients may avoid care in the future, putting them at risk for unplanned pregnancies, skipped cancer screenings, and complications from undiagnosed conditions and infections, Dr. Bayer added. Clinician offices will not always have access to all pain management options, so making referrals to another physician who has access to the appropriate technique may be the best thing for the patient, Dr. Bayer said.
Downplaying the experience
Informing a patient that she will feel only a little discomfort during a procedure – when a clinician doesn’t know how exactly the patient will react – can also result in distrust.
When a clinician says, “ ’It’s only going to be a little cramp, it’s only going to be a little pinch,’ we know extreme pain is a possibility, we’ve seen it,” Dr. Fenton said. “But if we choose to disregard that [possibility], it feels invalidating for patients.”
Failing to fully explain the possible pain scale can also directly interfere with the procedure at hand.
“My first concern is if they aren’t anticipating the amount of pain they are going to experience, they may move; For biopsies and IUD insertions, we need them to be still,” Dr. Wasson said. “If they are unable to tolerate the procedure, we’ve put them through pain and not been able to accomplish the primary goal.”
Managing both pain and what patients can expect is even more crucial for adolescent and teenage patients who are often having their first gynecologic experience.
“We’re framing what these experiences look like,” Dr. Fenton said. “That means there are opportunities for creating a space that builds trust and security for the patients moving forward.”
A version of this article first appeared on Medscape.com.
During a fellowship rotation in gynecology, Rebekah D. Fenton, MD, asked the attending physicians what pain management options they could offer patients for insertion of an intrauterine device (IUD). Their answer surprised her: None.
The research on the effectiveness of pain management techniques during the procedure were not strong enough to warrant providing potential relief.
But Dr. Fenton knew the attending physician was wrong: She’d received the drug lidocaine during a recent visit to her own ob.gyn. to get an IUD placed. The local anesthetic enabled her to avoid the experiences of many patients who often withstand debilitating cramping and pain during insertion, side effects that can last for hours after the procedure has ended.
By not teaching her how to administer pain treatment options such as lidocaine gel or injection, “they made the decision for me, whether I could give patients this option,” said Dr. Fenton, now an adolescent medicine specialist at Alivio Medical Center in Chicago.
As a result, patients undergoing IUD placements, biopsies, hysteroscopies, and pelvic exams are often subject to pain that could be mitigated.
Some research suggests simple numbing agents, including lidocaine, may induce less pain without the need for full anesthesia. But clinicians don’t always present these options.
During gynecologic procedures, the amount of pain a patient can expect is often downplayed by clinicians. Because every patient experiences the sensation differently, discussing options for pain management and the range of possible pain is paramount in building patient-clinician trust, and ultimately providing the best care for patients in the long run, according to Megan Wasson, DO, chair of the department of medical and surgical gynecology at Mayo Clinic Arizona in Phoenix.
“It comes down to shared decision-making so the patient is aware of the pain that should be expected and what avenue they want to go down,” Dr. Wasson said. “It’s not a one-size-fits-all.”
Lack of uniform protocols
The American College of Obstetricians and Gynecologists (ACOG) has clear guidelines for pain management during pregnancy and delivery but not for many routine gynecologic procedures. Some experts say not offering options for pain management based on lack of efficacy evidence can undermine a patient’s experience.
ACOG does have recommendations for reducing dilation pain during a hysteroscopy, including providing intravaginal misoprostol and estrogen. The organization also recommends performing a vaginoscopy instead if possible because the procedure is typically less painful than is a hysteroscopy.
For an IUD placement, ACOG states that the procedure “may cause temporary discomfort” and recommends that patients take over-the-counter pain relief before a procedure. The most recent clinical bulletin on the topic, published in 2016, states routine misoprostol is not recommended for IUD placement, although it may be considered with difficult insertions for management of pain.
A clinical inquiry published in 2020 outlined the efficacy of several pain options that practitioners can weigh with patients. The inquiry cited a 2019 meta-analysis of 38 studies that found lidocaine-prilocaine cream to be the most effective option for pain management during IUD placement, reducing insertion pain by nearly 30%. The inquiry concluded that a combination of 600 mcg of misoprostol and 4% lidocaine gel may be effective, while lower dosages of both drugs were not effective. A 2018 clinical trial cited in the analysis found that though a 20-cc 1% lidocaine paracervical block on its own did not reduce pain, the block mixed with sodium bicarbonate reduced pain during IUD insertion by 22%.
Some doctors make the decision to not use lidocaine without offering it to patients first, according to Dr. Fenton. Instead, clinicians should discuss any potential drawbacks, such as pain from administering the numbing agent with a needle or the procedure taking extra time while the patient waits for the lidocaine to kick in.
“That always felt unfair, to make that decision for [the patient],” Dr. Fenton said.
Often clinicians won’t know how a patient will respond to a procedure: A 2014 secondary analysis of a clinical trial compared how patients rated their pain after an IUD procedure to the amount of pain physicians perceived the procedure to cause. They found that the average pain scores patients reported were nearly twice as high as clinician expectations were.
ACOG’s guideline states that the evidence backing paracervical blocks and lidocaine to IUD insertion pain is controversial. The American College of Physicians also cites “low-quality evidence” to support patient reports of pain and discomfort during pelvic exams. Some studies have found up to 60% of women report these negative experiences.
The varying evidence highlights the need for a personalized approach – one that includes patients – to pain management for routine gynecological procedures.
“Usually patients are pretty good predictors,” said Lisa Bayer, MD, MPH, associate professor of obstetrics and gynecology at Oregon Health & Science University in Portland. “They can anticipate what different things are going to feel like based on previous experiences.”
Making patients part of the discussion
Clinicians should have open discussions with patients about their past experiences and current anxieties about a gynecologic procedure, according to Dr. Bayer.
“Part of it is just creating a really safe environment of trust as a medical provider,” she said.
A study published in 2016 of more than 800 patients undergoing oocyte retrieval, which has clear protocols for pain management, found that previous negative gynecologic experiences were significantly correlated to greater amounts of pain reported during the procedure.
If pain isn’t properly managed, patients may avoid care in the future, putting them at risk for unplanned pregnancies, skipped cancer screenings, and complications from undiagnosed conditions and infections, Dr. Bayer added. Clinician offices will not always have access to all pain management options, so making referrals to another physician who has access to the appropriate technique may be the best thing for the patient, Dr. Bayer said.
Downplaying the experience
Informing a patient that she will feel only a little discomfort during a procedure – when a clinician doesn’t know how exactly the patient will react – can also result in distrust.
When a clinician says, “ ’It’s only going to be a little cramp, it’s only going to be a little pinch,’ we know extreme pain is a possibility, we’ve seen it,” Dr. Fenton said. “But if we choose to disregard that [possibility], it feels invalidating for patients.”
Failing to fully explain the possible pain scale can also directly interfere with the procedure at hand.
“My first concern is if they aren’t anticipating the amount of pain they are going to experience, they may move; For biopsies and IUD insertions, we need them to be still,” Dr. Wasson said. “If they are unable to tolerate the procedure, we’ve put them through pain and not been able to accomplish the primary goal.”
Managing both pain and what patients can expect is even more crucial for adolescent and teenage patients who are often having their first gynecologic experience.
“We’re framing what these experiences look like,” Dr. Fenton said. “That means there are opportunities for creating a space that builds trust and security for the patients moving forward.”
A version of this article first appeared on Medscape.com.
Obesity boosts gestational diabetes risk in women with PCOS
In a population-based cohort study that included more than 1.2 million hospital live births, PCOS was associated with a 5% increase in risk for gestational diabetes. Almost 90% of this association was mediated by obesity.
“Women with PCOS are at higher risk, but it’s only 5% higher than the general population. However, that risk rises substantially with obesity,” senior author Maria P. Velez, MD, PhD, clinician-scientist and associate professor of obstetrics and gynecology at Queen’s University, Kingston, Ont., said in an interview. “Our study highlights the need for counseling our patients about the importance of weight optimization, ideally starting with lifestyle changes like diet and exercise.”The findings were published in the Journal of Obstetrics and Gynaecology Canada.
Major mediator
The estimated prevalence of PCOS is 8%-13%, and affected patients often present with anovulation, hyperandrogenism, obesity, metabolic syndrome, and infertility. Prepregnancy insulin resistance is common among women with PCOS and may play a major part in the pathogenesis of gestational diabetes. In addition, PCOS is often accompanied by excess weight gain; about 60% of women with PCOS are overweight or obese.
Previous research has shown that PCOS is a risk factor for gestational diabetes independent of obesity, while other research has shown that obesity has an important effect on this risk.
For the current study, the researchers used causal mediation analysis to elucidate more clearly the effect of obesity on the development of gestational diabetes among patients with PCOS. No previous study has used causal mediation analysis to examine this relationship.
Using data from linked universal health databases in Ontario, the researchers analyzed data on 1,268,901 births between 2006 and 2018. Of these births, 386,748 were associated with maternal PCOS.
The rate of gestational diabetes was higher among women with PCOS (60.2 per 1000 births), compared with women without PCOS (48.6 per 1,000 births). The finding resulted in an adjusted relative risk of 1.05. Obesity mediated 89.7% of this association.
“We hope that these data will inform preconception counseling and gestational diabetes screening in pregnant women with PCOS,” said Dr. Velez. “We have the data now to counsel our patients on the importance of weight management before pregnancy. But we need more resources, such as specialized clinics, to help these patients cope with managing their weight. We can tell our patients to work on their weight management, but they need much more support from the health care system.”
Results ‘not surprising’
Commenting on the study, Francine Hippolyte, MD, vice chair of obstetrics and gynecology at Long Island Jewish Medical Center, Katz Women’s Hospital, New Hyde Park, N.Y., said that the results are “not at all surprising.” Dr. Hippolyte was not involved in the research.
“We do know that PCOS is and should be treated as a metabolic syndrome. It’s a lot more than just infertility or changes or abnormalities with one’s menstrual cycle. It impacts a woman’s risk for diabetes, prediabetes, and abnormal lipid profile, regardless of whether or not she is obese,” said Dr. Hippolyte.
She agrees with the need for specialized clinics to help such vulnerable patients manage their weight.
“It would be great if insurances would cover things like nutritional counseling or have nutritionists on their roster so that patients can easily access that service. Many patients want to do right, especially preconceptually, but it is difficult without having access to resources. Unfortunately, as clinicians, we’re not as well versed in nutrition as we would like to be or should be, so we need a multidisciplinary approach. We need nutrition and weight loss clinics and proper services to really help these patients.”
The study was supported by the Canadian Institute of Health Research and ICES. Dr. Velez and Dr. Hippolyte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a population-based cohort study that included more than 1.2 million hospital live births, PCOS was associated with a 5% increase in risk for gestational diabetes. Almost 90% of this association was mediated by obesity.
“Women with PCOS are at higher risk, but it’s only 5% higher than the general population. However, that risk rises substantially with obesity,” senior author Maria P. Velez, MD, PhD, clinician-scientist and associate professor of obstetrics and gynecology at Queen’s University, Kingston, Ont., said in an interview. “Our study highlights the need for counseling our patients about the importance of weight optimization, ideally starting with lifestyle changes like diet and exercise.”The findings were published in the Journal of Obstetrics and Gynaecology Canada.
Major mediator
The estimated prevalence of PCOS is 8%-13%, and affected patients often present with anovulation, hyperandrogenism, obesity, metabolic syndrome, and infertility. Prepregnancy insulin resistance is common among women with PCOS and may play a major part in the pathogenesis of gestational diabetes. In addition, PCOS is often accompanied by excess weight gain; about 60% of women with PCOS are overweight or obese.
Previous research has shown that PCOS is a risk factor for gestational diabetes independent of obesity, while other research has shown that obesity has an important effect on this risk.
For the current study, the researchers used causal mediation analysis to elucidate more clearly the effect of obesity on the development of gestational diabetes among patients with PCOS. No previous study has used causal mediation analysis to examine this relationship.
Using data from linked universal health databases in Ontario, the researchers analyzed data on 1,268,901 births between 2006 and 2018. Of these births, 386,748 were associated with maternal PCOS.
The rate of gestational diabetes was higher among women with PCOS (60.2 per 1000 births), compared with women without PCOS (48.6 per 1,000 births). The finding resulted in an adjusted relative risk of 1.05. Obesity mediated 89.7% of this association.
“We hope that these data will inform preconception counseling and gestational diabetes screening in pregnant women with PCOS,” said Dr. Velez. “We have the data now to counsel our patients on the importance of weight management before pregnancy. But we need more resources, such as specialized clinics, to help these patients cope with managing their weight. We can tell our patients to work on their weight management, but they need much more support from the health care system.”
Results ‘not surprising’
Commenting on the study, Francine Hippolyte, MD, vice chair of obstetrics and gynecology at Long Island Jewish Medical Center, Katz Women’s Hospital, New Hyde Park, N.Y., said that the results are “not at all surprising.” Dr. Hippolyte was not involved in the research.
“We do know that PCOS is and should be treated as a metabolic syndrome. It’s a lot more than just infertility or changes or abnormalities with one’s menstrual cycle. It impacts a woman’s risk for diabetes, prediabetes, and abnormal lipid profile, regardless of whether or not she is obese,” said Dr. Hippolyte.
She agrees with the need for specialized clinics to help such vulnerable patients manage their weight.
“It would be great if insurances would cover things like nutritional counseling or have nutritionists on their roster so that patients can easily access that service. Many patients want to do right, especially preconceptually, but it is difficult without having access to resources. Unfortunately, as clinicians, we’re not as well versed in nutrition as we would like to be or should be, so we need a multidisciplinary approach. We need nutrition and weight loss clinics and proper services to really help these patients.”
The study was supported by the Canadian Institute of Health Research and ICES. Dr. Velez and Dr. Hippolyte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a population-based cohort study that included more than 1.2 million hospital live births, PCOS was associated with a 5% increase in risk for gestational diabetes. Almost 90% of this association was mediated by obesity.
“Women with PCOS are at higher risk, but it’s only 5% higher than the general population. However, that risk rises substantially with obesity,” senior author Maria P. Velez, MD, PhD, clinician-scientist and associate professor of obstetrics and gynecology at Queen’s University, Kingston, Ont., said in an interview. “Our study highlights the need for counseling our patients about the importance of weight optimization, ideally starting with lifestyle changes like diet and exercise.”The findings were published in the Journal of Obstetrics and Gynaecology Canada.
Major mediator
The estimated prevalence of PCOS is 8%-13%, and affected patients often present with anovulation, hyperandrogenism, obesity, metabolic syndrome, and infertility. Prepregnancy insulin resistance is common among women with PCOS and may play a major part in the pathogenesis of gestational diabetes. In addition, PCOS is often accompanied by excess weight gain; about 60% of women with PCOS are overweight or obese.
Previous research has shown that PCOS is a risk factor for gestational diabetes independent of obesity, while other research has shown that obesity has an important effect on this risk.
For the current study, the researchers used causal mediation analysis to elucidate more clearly the effect of obesity on the development of gestational diabetes among patients with PCOS. No previous study has used causal mediation analysis to examine this relationship.
Using data from linked universal health databases in Ontario, the researchers analyzed data on 1,268,901 births between 2006 and 2018. Of these births, 386,748 were associated with maternal PCOS.
The rate of gestational diabetes was higher among women with PCOS (60.2 per 1000 births), compared with women without PCOS (48.6 per 1,000 births). The finding resulted in an adjusted relative risk of 1.05. Obesity mediated 89.7% of this association.
“We hope that these data will inform preconception counseling and gestational diabetes screening in pregnant women with PCOS,” said Dr. Velez. “We have the data now to counsel our patients on the importance of weight management before pregnancy. But we need more resources, such as specialized clinics, to help these patients cope with managing their weight. We can tell our patients to work on their weight management, but they need much more support from the health care system.”
Results ‘not surprising’
Commenting on the study, Francine Hippolyte, MD, vice chair of obstetrics and gynecology at Long Island Jewish Medical Center, Katz Women’s Hospital, New Hyde Park, N.Y., said that the results are “not at all surprising.” Dr. Hippolyte was not involved in the research.
“We do know that PCOS is and should be treated as a metabolic syndrome. It’s a lot more than just infertility or changes or abnormalities with one’s menstrual cycle. It impacts a woman’s risk for diabetes, prediabetes, and abnormal lipid profile, regardless of whether or not she is obese,” said Dr. Hippolyte.
She agrees with the need for specialized clinics to help such vulnerable patients manage their weight.
“It would be great if insurances would cover things like nutritional counseling or have nutritionists on their roster so that patients can easily access that service. Many patients want to do right, especially preconceptually, but it is difficult without having access to resources. Unfortunately, as clinicians, we’re not as well versed in nutrition as we would like to be or should be, so we need a multidisciplinary approach. We need nutrition and weight loss clinics and proper services to really help these patients.”
The study was supported by the Canadian Institute of Health Research and ICES. Dr. Velez and Dr. Hippolyte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JOURNAL OF OBSTETRICS AND GYNAECOLOGY CANADA
Metabolic effects of estetrol are promising in postmenopausal women
PHILADELPHIA – presented at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
Participants taking estetrol experienced a decrease in hemoglobin A1c, fasting plasma glucose, total cholesterol, LDL and lipoprotein as well as an increase in HDL cholesterol, according to the findings presented by Wolf Utian, MD, PhD, DSC, a professor emeritus of reproductive biology at Case Western Reserve University, Cleveland, and medical director emeritus of the Menopause Society.
A separate poster at the conference from the same trial also reported significant improvements from estetrol in quality of life, including that related to vasomotor symptoms, and several psychosocial and sexual functioning areas.
E4 is already available as combination oral contraception and is now being considered for treating vasomotor symptoms, explained Chrisandra Shufelt, MD, professor and chair of general internal of medicine and associate director of the Women’s Health Research Center at Mayo Clinic Florida, who was not involved in the study.
Background on estetrol
E4 is a human fetal liver estrogen produced during pregnancy that’s synthesized from plants for pharmaceutical use, including as the oral contraceptive drospirenone, Dr. Utian told attendees. It’s classified as a native estrogen with selective tissue activity (NEST), he said.
“E4 is a completely different native estrogen with oral administration mimicking the benefits of transdermals and hence safe and effective,” Dr. Utian said in an interview. “It would be a significant new addition to the pharmaceutical armamentarium.”
Two phase 3 trials presented by Dr. Utian at the same conference last year found estetrol reduced the frequency and severity of moderate to severe vasomotor symptoms, and a previous phase 2 trial finding vasomotor and genitourinary symptom benefits suggested it had potential benefits for lipids, carbohydrate metabolism, and bone turnover.
“In summary, E4 at a daily dose of 15 mg exhibited estrogenic effects in the vagina, leading to improved vaginal health and reduced signs of atrophy, emerging as a promising treatment option not only for vasomotor symptoms but also for other significant menopausal symptoms,” Dr. Utian said. “E4 could offer comprehensive relief for women experiencing a range of menopause-related discomforts.”
Dr. Utian also referenced a 2017 trial in which estetrol positively impacted lipid profiles, “lowering low-density lipoprotein cholesterol, increasing high-density lipoprotein cholesterol, and showing minimal influence on triglycerides,” he said. “Importantly, estetrol was associated with a significant decrease in osteocalcin levels in the higher dose groups, suggesting a potential preventive effect on bone loss,” he added. A recent review of the overall evidence on estetrol suggests its use is “promising,” Dr. Utian noted.
Current trial
His current randomized controlled phase 3 trial included postmenopausal women ages 40-65 from 151 sites in 14 countries in Europe, Latin America, and North America, and Russia. Among the 640 participants in the trial, 213 women randomly received 15 mg of estetrol, 213 women received 20 mg of estetrol, and 214 women received a placebo every day for 3 months. All women without hysterectomies also received 200 mg of progesterone once daily for two weeks after completing the estetrol treatment to protect the endometrium.
Researchers took blood samples from the participants at baseline and week 12 to assess total cholesterol, LDL, HDL, the total cholesterol/HDL ratio, triglycerides, lipoprotein A, fasting plasma glucose, insulin, and A1c.
Compared with women in the placebo group, women in both the 15 mg and 20 mg groups saw a statistically significant decrease in lipoprotein A and in the ratio of total cholesterol to HDL, and a statistically significant increase in HDL. Only the women in the 15 mg group saw a statistically significant decrease in LDL and increase in triglycerides; an increase in triglycerides in the 20 mg group did not reach statistical significance.
Statistically significant decreases in fasting plasma glucose and A1c also occurred in both treatment groups, but a decrease in insulin levels and in the homeostasis model-assessment-estimated insulin resistance (HOMA-IR) seen in both treatment arms did not reach significance.
“While the mean changes after 12 weeks from baseline overall were small changes to the cholesterol and blood sugar profiles, they are clinically meaningful because it suggests that E4 does not have any adverse effects to these measures,” Dr. Shufelt said in an interview. “An advantage is that this gives us another hormone option for vasomotor symptoms since it is a native estrogen with selective tissue.”
It’s too early, however, to determine whether estetrol offers benefits in terms of its safety profile, compared with currently available therapies, Dr. Shufelt said.
”These findings of E4 are similar to how oral estradiol changes lipids, which finds an increase in high-density lipoprotein cholesterol, and decreases plasma concentrations of total and low-density lipoprotein cholesterol. an increase in HDL-C and triglycerides and decrease in LDL-C,” she said.
Poster findings also promising
For the findings reported in the poster, researchers assessed quality of life and the clinical meaningfulness of vasomotor symptoms’ reduction at baseline and 12 weeks using the Menopause-Specific Quality of Life (MENQOL) questionnaire and the Clinical Global Impression questionnaire, respectively. They also assessed women’s self-reported genitourinary symptoms, including vaginal dryness, pain during urination, vaginal pain and bleeding related to sex, and vaginal or vulvar irritation or itching. Most of these findings primarily confirmed previous positive effects from E4 in other trials.
Women in both the 15 mg and 20 mg estetrol groups reported a statistically significant improvement at 12 weeks, compared with placebo, in their total MENQOL score and in the vasomotor, psychosocial, and sexual functioning domain scores (P < .05). Those in the 20 mg group also had a statistically significant improvement in their physical domain score (P < .05).
Although numerical improvements in genitourinary symptoms occurred at 12 weeks across all three groups, the only statistically significant difference from baseline occurred in patients taking 15 mg of estetrol, who experienced a decrease in vaginal dryness and vaginal pain during sex (P = .0142 and P = .003, respectively).
The Clinical Global Impression questionnaire asked women at 4 and 12 weeks to rate on a seven-item Likert scale their response to this question: “Rate the total improvement, whether or not in your judgment it is due entirely to drug treatment. Compared to your condition at admission to the study, how much has it changed?” Responses of “very much improved” and “much improved” counted as a clinically meaningful difference.
Compared with 27.9% of patients in the placebo group, 52.9% of patients in the 15 mg group and 59.8% of patients in the 20 mg group rated the weekly frequency of moderate to severe vasomotor symptoms as “much improved” or “very much improved” at 4 weeks (P < .0001). At 12 weeks, those numbers rose to 47% in the placebo group, 73.3% in the 15 mg group and 77.8% in the 20 mg group (P < .0001).
The trial’s primary limitation at this point is having only a 12-week follow-up, Dr. Shufelt said, though a few other questions remain.
“Because the two phase 3 RCTs included hysterectomized and nonhysterectomized women, it was unclear how many women in the study had E4 alone versus E4 with progesterone, as that might play a role in both cholesterol and carbohydrate metabolism,” Dr. Shufelt said. “While baseline data was not presented, it would also be important to know baseline values for the women and confirm that none were on lipid-lowering medications.”
The research was funded by Estetra SRL, an affiliate of Mithra Pharmaceuticals. Dr. Utian is a member of the Mithra and Elektra Scientific Advisory Boards. Dr. Shufelt has no disclosures.
PHILADELPHIA – presented at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
Participants taking estetrol experienced a decrease in hemoglobin A1c, fasting plasma glucose, total cholesterol, LDL and lipoprotein as well as an increase in HDL cholesterol, according to the findings presented by Wolf Utian, MD, PhD, DSC, a professor emeritus of reproductive biology at Case Western Reserve University, Cleveland, and medical director emeritus of the Menopause Society.
A separate poster at the conference from the same trial also reported significant improvements from estetrol in quality of life, including that related to vasomotor symptoms, and several psychosocial and sexual functioning areas.
E4 is already available as combination oral contraception and is now being considered for treating vasomotor symptoms, explained Chrisandra Shufelt, MD, professor and chair of general internal of medicine and associate director of the Women’s Health Research Center at Mayo Clinic Florida, who was not involved in the study.
Background on estetrol
E4 is a human fetal liver estrogen produced during pregnancy that’s synthesized from plants for pharmaceutical use, including as the oral contraceptive drospirenone, Dr. Utian told attendees. It’s classified as a native estrogen with selective tissue activity (NEST), he said.
“E4 is a completely different native estrogen with oral administration mimicking the benefits of transdermals and hence safe and effective,” Dr. Utian said in an interview. “It would be a significant new addition to the pharmaceutical armamentarium.”
Two phase 3 trials presented by Dr. Utian at the same conference last year found estetrol reduced the frequency and severity of moderate to severe vasomotor symptoms, and a previous phase 2 trial finding vasomotor and genitourinary symptom benefits suggested it had potential benefits for lipids, carbohydrate metabolism, and bone turnover.
“In summary, E4 at a daily dose of 15 mg exhibited estrogenic effects in the vagina, leading to improved vaginal health and reduced signs of atrophy, emerging as a promising treatment option not only for vasomotor symptoms but also for other significant menopausal symptoms,” Dr. Utian said. “E4 could offer comprehensive relief for women experiencing a range of menopause-related discomforts.”
Dr. Utian also referenced a 2017 trial in which estetrol positively impacted lipid profiles, “lowering low-density lipoprotein cholesterol, increasing high-density lipoprotein cholesterol, and showing minimal influence on triglycerides,” he said. “Importantly, estetrol was associated with a significant decrease in osteocalcin levels in the higher dose groups, suggesting a potential preventive effect on bone loss,” he added. A recent review of the overall evidence on estetrol suggests its use is “promising,” Dr. Utian noted.
Current trial
His current randomized controlled phase 3 trial included postmenopausal women ages 40-65 from 151 sites in 14 countries in Europe, Latin America, and North America, and Russia. Among the 640 participants in the trial, 213 women randomly received 15 mg of estetrol, 213 women received 20 mg of estetrol, and 214 women received a placebo every day for 3 months. All women without hysterectomies also received 200 mg of progesterone once daily for two weeks after completing the estetrol treatment to protect the endometrium.
Researchers took blood samples from the participants at baseline and week 12 to assess total cholesterol, LDL, HDL, the total cholesterol/HDL ratio, triglycerides, lipoprotein A, fasting plasma glucose, insulin, and A1c.
Compared with women in the placebo group, women in both the 15 mg and 20 mg groups saw a statistically significant decrease in lipoprotein A and in the ratio of total cholesterol to HDL, and a statistically significant increase in HDL. Only the women in the 15 mg group saw a statistically significant decrease in LDL and increase in triglycerides; an increase in triglycerides in the 20 mg group did not reach statistical significance.
Statistically significant decreases in fasting plasma glucose and A1c also occurred in both treatment groups, but a decrease in insulin levels and in the homeostasis model-assessment-estimated insulin resistance (HOMA-IR) seen in both treatment arms did not reach significance.
“While the mean changes after 12 weeks from baseline overall were small changes to the cholesterol and blood sugar profiles, they are clinically meaningful because it suggests that E4 does not have any adverse effects to these measures,” Dr. Shufelt said in an interview. “An advantage is that this gives us another hormone option for vasomotor symptoms since it is a native estrogen with selective tissue.”
It’s too early, however, to determine whether estetrol offers benefits in terms of its safety profile, compared with currently available therapies, Dr. Shufelt said.
”These findings of E4 are similar to how oral estradiol changes lipids, which finds an increase in high-density lipoprotein cholesterol, and decreases plasma concentrations of total and low-density lipoprotein cholesterol. an increase in HDL-C and triglycerides and decrease in LDL-C,” she said.
Poster findings also promising
For the findings reported in the poster, researchers assessed quality of life and the clinical meaningfulness of vasomotor symptoms’ reduction at baseline and 12 weeks using the Menopause-Specific Quality of Life (MENQOL) questionnaire and the Clinical Global Impression questionnaire, respectively. They also assessed women’s self-reported genitourinary symptoms, including vaginal dryness, pain during urination, vaginal pain and bleeding related to sex, and vaginal or vulvar irritation or itching. Most of these findings primarily confirmed previous positive effects from E4 in other trials.
Women in both the 15 mg and 20 mg estetrol groups reported a statistically significant improvement at 12 weeks, compared with placebo, in their total MENQOL score and in the vasomotor, psychosocial, and sexual functioning domain scores (P < .05). Those in the 20 mg group also had a statistically significant improvement in their physical domain score (P < .05).
Although numerical improvements in genitourinary symptoms occurred at 12 weeks across all three groups, the only statistically significant difference from baseline occurred in patients taking 15 mg of estetrol, who experienced a decrease in vaginal dryness and vaginal pain during sex (P = .0142 and P = .003, respectively).
The Clinical Global Impression questionnaire asked women at 4 and 12 weeks to rate on a seven-item Likert scale their response to this question: “Rate the total improvement, whether or not in your judgment it is due entirely to drug treatment. Compared to your condition at admission to the study, how much has it changed?” Responses of “very much improved” and “much improved” counted as a clinically meaningful difference.
Compared with 27.9% of patients in the placebo group, 52.9% of patients in the 15 mg group and 59.8% of patients in the 20 mg group rated the weekly frequency of moderate to severe vasomotor symptoms as “much improved” or “very much improved” at 4 weeks (P < .0001). At 12 weeks, those numbers rose to 47% in the placebo group, 73.3% in the 15 mg group and 77.8% in the 20 mg group (P < .0001).
The trial’s primary limitation at this point is having only a 12-week follow-up, Dr. Shufelt said, though a few other questions remain.
“Because the two phase 3 RCTs included hysterectomized and nonhysterectomized women, it was unclear how many women in the study had E4 alone versus E4 with progesterone, as that might play a role in both cholesterol and carbohydrate metabolism,” Dr. Shufelt said. “While baseline data was not presented, it would also be important to know baseline values for the women and confirm that none were on lipid-lowering medications.”
The research was funded by Estetra SRL, an affiliate of Mithra Pharmaceuticals. Dr. Utian is a member of the Mithra and Elektra Scientific Advisory Boards. Dr. Shufelt has no disclosures.
PHILADELPHIA – presented at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
Participants taking estetrol experienced a decrease in hemoglobin A1c, fasting plasma glucose, total cholesterol, LDL and lipoprotein as well as an increase in HDL cholesterol, according to the findings presented by Wolf Utian, MD, PhD, DSC, a professor emeritus of reproductive biology at Case Western Reserve University, Cleveland, and medical director emeritus of the Menopause Society.
A separate poster at the conference from the same trial also reported significant improvements from estetrol in quality of life, including that related to vasomotor symptoms, and several psychosocial and sexual functioning areas.
E4 is already available as combination oral contraception and is now being considered for treating vasomotor symptoms, explained Chrisandra Shufelt, MD, professor and chair of general internal of medicine and associate director of the Women’s Health Research Center at Mayo Clinic Florida, who was not involved in the study.
Background on estetrol
E4 is a human fetal liver estrogen produced during pregnancy that’s synthesized from plants for pharmaceutical use, including as the oral contraceptive drospirenone, Dr. Utian told attendees. It’s classified as a native estrogen with selective tissue activity (NEST), he said.
“E4 is a completely different native estrogen with oral administration mimicking the benefits of transdermals and hence safe and effective,” Dr. Utian said in an interview. “It would be a significant new addition to the pharmaceutical armamentarium.”
Two phase 3 trials presented by Dr. Utian at the same conference last year found estetrol reduced the frequency and severity of moderate to severe vasomotor symptoms, and a previous phase 2 trial finding vasomotor and genitourinary symptom benefits suggested it had potential benefits for lipids, carbohydrate metabolism, and bone turnover.
“In summary, E4 at a daily dose of 15 mg exhibited estrogenic effects in the vagina, leading to improved vaginal health and reduced signs of atrophy, emerging as a promising treatment option not only for vasomotor symptoms but also for other significant menopausal symptoms,” Dr. Utian said. “E4 could offer comprehensive relief for women experiencing a range of menopause-related discomforts.”
Dr. Utian also referenced a 2017 trial in which estetrol positively impacted lipid profiles, “lowering low-density lipoprotein cholesterol, increasing high-density lipoprotein cholesterol, and showing minimal influence on triglycerides,” he said. “Importantly, estetrol was associated with a significant decrease in osteocalcin levels in the higher dose groups, suggesting a potential preventive effect on bone loss,” he added. A recent review of the overall evidence on estetrol suggests its use is “promising,” Dr. Utian noted.
Current trial
His current randomized controlled phase 3 trial included postmenopausal women ages 40-65 from 151 sites in 14 countries in Europe, Latin America, and North America, and Russia. Among the 640 participants in the trial, 213 women randomly received 15 mg of estetrol, 213 women received 20 mg of estetrol, and 214 women received a placebo every day for 3 months. All women without hysterectomies also received 200 mg of progesterone once daily for two weeks after completing the estetrol treatment to protect the endometrium.
Researchers took blood samples from the participants at baseline and week 12 to assess total cholesterol, LDL, HDL, the total cholesterol/HDL ratio, triglycerides, lipoprotein A, fasting plasma glucose, insulin, and A1c.
Compared with women in the placebo group, women in both the 15 mg and 20 mg groups saw a statistically significant decrease in lipoprotein A and in the ratio of total cholesterol to HDL, and a statistically significant increase in HDL. Only the women in the 15 mg group saw a statistically significant decrease in LDL and increase in triglycerides; an increase in triglycerides in the 20 mg group did not reach statistical significance.
Statistically significant decreases in fasting plasma glucose and A1c also occurred in both treatment groups, but a decrease in insulin levels and in the homeostasis model-assessment-estimated insulin resistance (HOMA-IR) seen in both treatment arms did not reach significance.
“While the mean changes after 12 weeks from baseline overall were small changes to the cholesterol and blood sugar profiles, they are clinically meaningful because it suggests that E4 does not have any adverse effects to these measures,” Dr. Shufelt said in an interview. “An advantage is that this gives us another hormone option for vasomotor symptoms since it is a native estrogen with selective tissue.”
It’s too early, however, to determine whether estetrol offers benefits in terms of its safety profile, compared with currently available therapies, Dr. Shufelt said.
”These findings of E4 are similar to how oral estradiol changes lipids, which finds an increase in high-density lipoprotein cholesterol, and decreases plasma concentrations of total and low-density lipoprotein cholesterol. an increase in HDL-C and triglycerides and decrease in LDL-C,” she said.
Poster findings also promising
For the findings reported in the poster, researchers assessed quality of life and the clinical meaningfulness of vasomotor symptoms’ reduction at baseline and 12 weeks using the Menopause-Specific Quality of Life (MENQOL) questionnaire and the Clinical Global Impression questionnaire, respectively. They also assessed women’s self-reported genitourinary symptoms, including vaginal dryness, pain during urination, vaginal pain and bleeding related to sex, and vaginal or vulvar irritation or itching. Most of these findings primarily confirmed previous positive effects from E4 in other trials.
Women in both the 15 mg and 20 mg estetrol groups reported a statistically significant improvement at 12 weeks, compared with placebo, in their total MENQOL score and in the vasomotor, psychosocial, and sexual functioning domain scores (P < .05). Those in the 20 mg group also had a statistically significant improvement in their physical domain score (P < .05).
Although numerical improvements in genitourinary symptoms occurred at 12 weeks across all three groups, the only statistically significant difference from baseline occurred in patients taking 15 mg of estetrol, who experienced a decrease in vaginal dryness and vaginal pain during sex (P = .0142 and P = .003, respectively).
The Clinical Global Impression questionnaire asked women at 4 and 12 weeks to rate on a seven-item Likert scale their response to this question: “Rate the total improvement, whether or not in your judgment it is due entirely to drug treatment. Compared to your condition at admission to the study, how much has it changed?” Responses of “very much improved” and “much improved” counted as a clinically meaningful difference.
Compared with 27.9% of patients in the placebo group, 52.9% of patients in the 15 mg group and 59.8% of patients in the 20 mg group rated the weekly frequency of moderate to severe vasomotor symptoms as “much improved” or “very much improved” at 4 weeks (P < .0001). At 12 weeks, those numbers rose to 47% in the placebo group, 73.3% in the 15 mg group and 77.8% in the 20 mg group (P < .0001).
The trial’s primary limitation at this point is having only a 12-week follow-up, Dr. Shufelt said, though a few other questions remain.
“Because the two phase 3 RCTs included hysterectomized and nonhysterectomized women, it was unclear how many women in the study had E4 alone versus E4 with progesterone, as that might play a role in both cholesterol and carbohydrate metabolism,” Dr. Shufelt said. “While baseline data was not presented, it would also be important to know baseline values for the women and confirm that none were on lipid-lowering medications.”
The research was funded by Estetra SRL, an affiliate of Mithra Pharmaceuticals. Dr. Utian is a member of the Mithra and Elektra Scientific Advisory Boards. Dr. Shufelt has no disclosures.
AT NAMS 2023
Have you asked your patients: What is your ideal outpatient gynecology experience?
There has been increasing awareness of a need for creating a more patient-centered experience with outpatient gynecology; however, very little data exist about what interventions are important to patients. Given social media’s ease of use and ability for widespread access to a diverse group of users, it has the potential to be a powerful tool for qualitative research questions without the difficulties of cost, transportation, transcription, etc. required of a focus group. Crowdsourced public opinion also has the advantage of producing qualitative metrics in the form of “likes” that, at scale, can provide a reliable measure of public support or engagement for a particular concept.1 Particularly for topics that are controversial or novel, X (formerly Twitter, and referred to as Twitter intermittently throughout this article based on the time the study was conducted), with 300 million monthly users,2 has become a popular tool for general and health care ̶ focused content and sentiment analysis.3,4 This study presents a qualitative analysis of themes from a crowdsourced request on Twitter to design the ideal outpatient gynecologic experience that subsequently went “viral”.5,6
When asked to design the optimized outpatient gynecology experience, social media users expressed:
- hospitality, comfort, and pain control as frequent themes
- preserving privacy and acknowledgement of voluntary nulliparity as frequent themes
- a desire for diverse imagery and representation related to race, LGBTQIA+ themes, age, and weight/body type within the office setting
- a call for a sense of psychological safety within gynecology
Why the need for our research question on patient-centered gyn care
While the body of literature on patient-centered health care has grown rapidly in recent years, a patient-centered outpatient gynecology experience has not yet been described in the medical literature.
Patient-centered office design, driven by cultural sensitivity, has been shown in other studies to be both appreciated by established patients and a viable business strategy to attract new patients.7 Topics such as pain control, trauma-informed care in gynecologyclinics,8 and diverse representation in patient materials and illustrations9 have been popular topics in medicine and in the lay press. Our primary aim in our research was to utilize feedback from the question posed to quantify and rank patient-centered interventions in a gynecology office. These themes and others that emerged in our analysis were used to suggest b
What we asked social media users. The survey query to social media users, “I have the opportunity to design my office from scratch. I’m asking women: How would you design/optimize a visit to the gynecologist’s office?” was crowd-sourced via Twitter on December 5, 2021.5 Given a robust response to the query, it provided an opportunity for a qualitative research study exploring social media users’ perspectives on optimizing outpatient gynecologic care, although the original question was not planned for research utilization.
What we found
By December 27, 2021, the original tweet had earned 9,411 likes; 2,143 retweets; and 3,400 replies. Of this group, we analyzed 131 tweets, all of which had 100 or greater likes on Twitter at the time of the review. The majority of analyzed tweets earned between 100 ̶ 500 likes (75/131; 57.3%), while 22.9% (30/131) had 501 ̶ 1,000 likes, 11.5% (15/131) had >2,000 likes, and 8.4% (11/131) had 1,001 ̶ 1,999 likes.
Identified themes within the tweets analyzed included: medical education, comfort improvements, continuity of care, disability accommodations/accessibility, economic accessibility, nonbinary/transgender care and inclusivity, general layout/floorplan, hospitality, aid for intimate partner violence, childcare accessibility, multi-disciplinary care access, pain/anxiety control, sensitivity toward pregnancy loss/fertility issues, privacy issues, professionalism, representation (subdivided into race, LGBTQIA+, age, and weight/body type), trauma-informed care, and acknowledgement of voluntary nulliparity/support for reproductive choices (TABLE 1). TABLE 2 lists examples of popular tweets by selected themes.
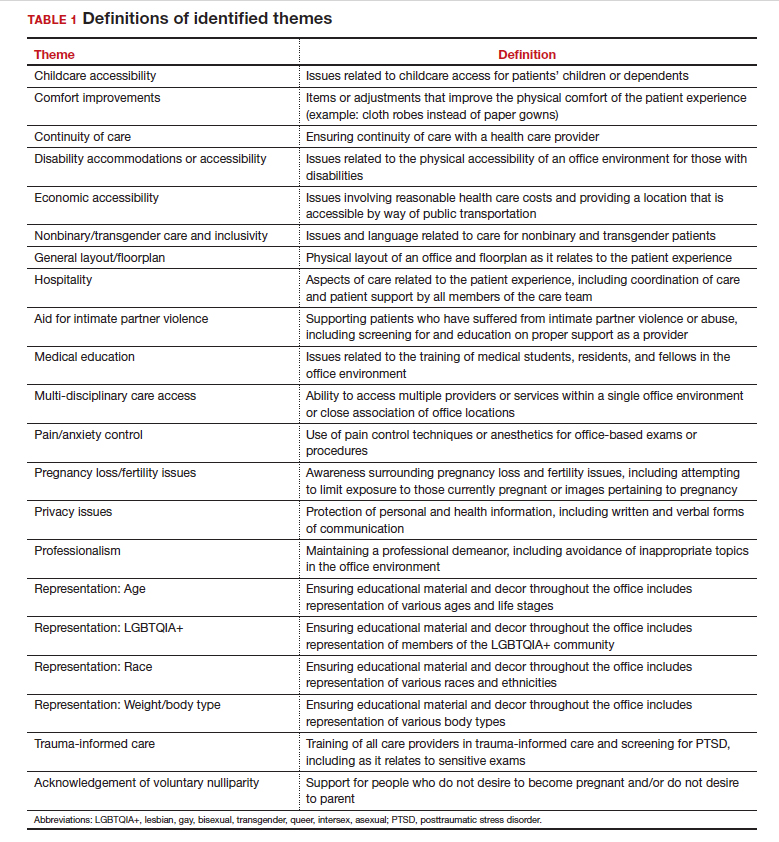
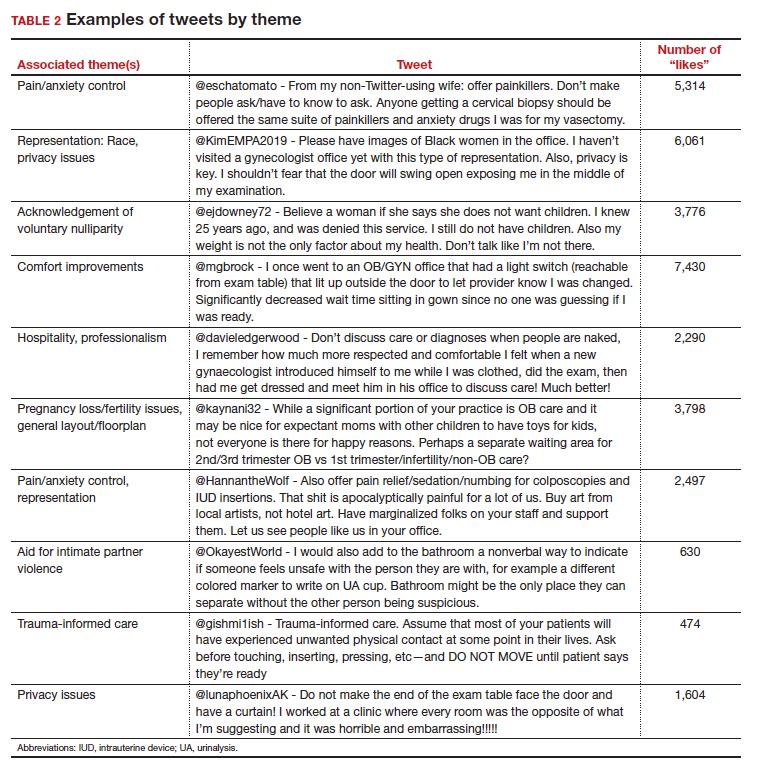
Frequent themes. The most frequently occurring themes within the 131 analyzed tweets (FIGURE 1) were:
- hospitality (77 occurrences)
- comfort improvements (75 occurrences)
- general layout/floorplan (75 occurrences)
- pain/anxiety control (55 occurrences)
- representation (53 occurrences).
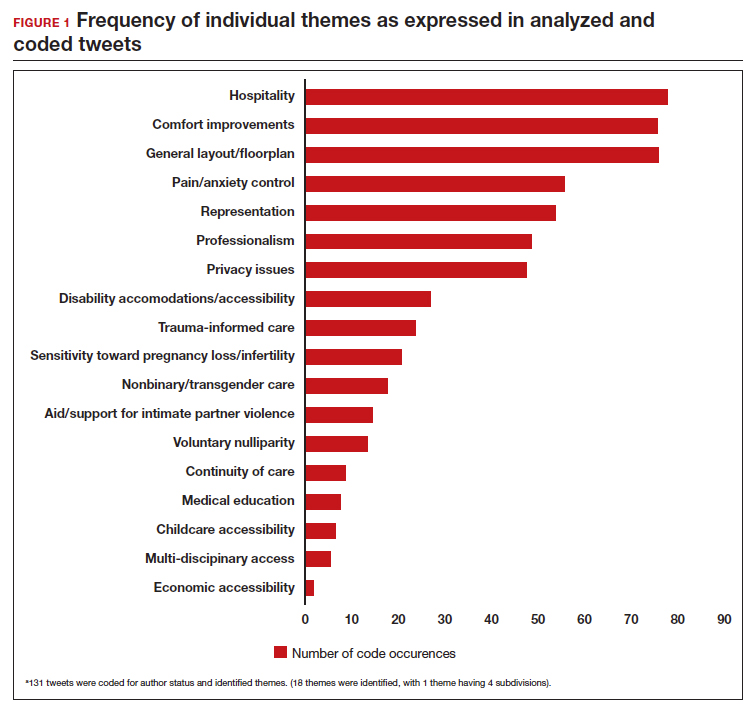
Popular themes. Defined as those with more than 1,000 likes at the time of analysis (FIGURE 2), the most popular themes included:
- privacy issues (48.5% of related tweets with >1,000 likes)
- voluntary nulliparity (37.0% of related tweets with >1,000 likes)
- general layout/floorplan (33.4% of related tweets with >1,000 likes)
- representation (32.1% of related tweets with >1,000 likes)
- hospitality (31.3% of related tweets with >1,000 likes).
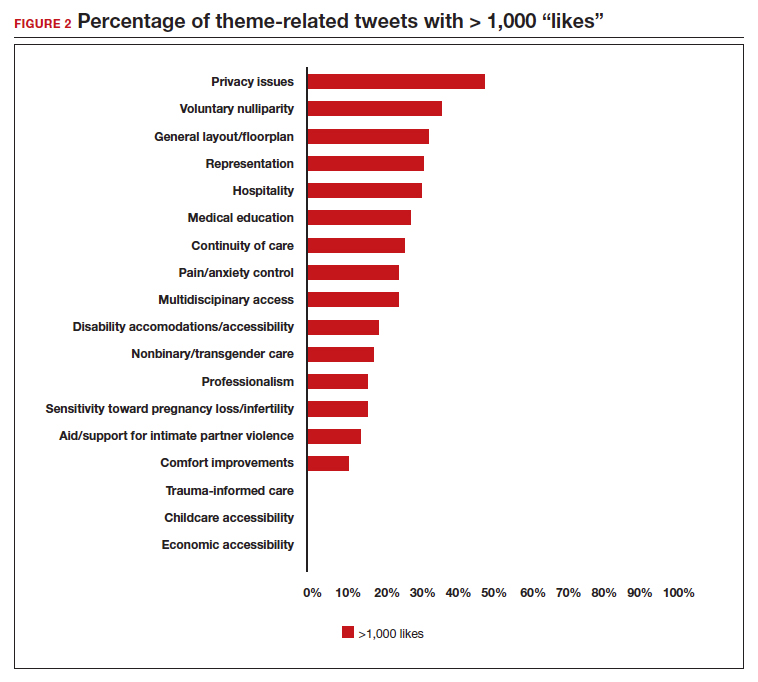
A sub-analysis of themes related to specific types of representation—race, LGBTQIA+, age, and weight/body type was performed. Tweets related to diverse weight/body type representation occurred most frequently (19 code occurrences; FIGURE 3). Similarly, tweets related to the representation of diverse races and the LGBTQIA+ community each comprised 26% of the total representation-based tweets. In terms of popularity as described above, 51.4% of tweets describing racial representation earned >1,000 likes (FIGURE 4).
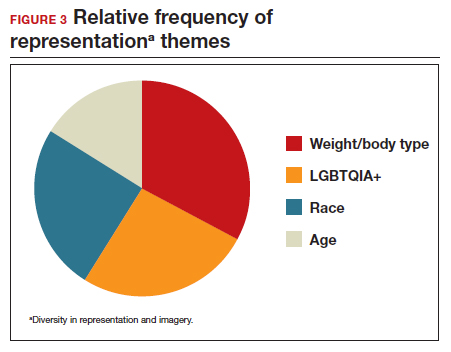
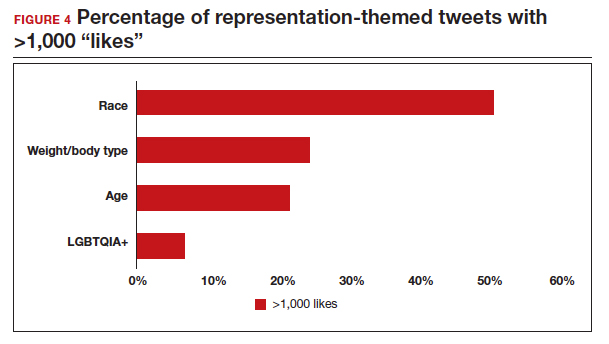
Tweet demographics. Seven (7/131; 5.3%) of the tweet authors were verified Twitter users and 35 (35/131; 26.7%) authors reported working in the health care field within their Twitter profile description.
Continue to: Implementing our feedback can enhance patient experience and care...
Implementing our feedback can enhance patient experience and care
Our study provides a unique view of the patient perspective through analyzed crowdsourced public opinion via Twitter. To our knowledge, an optimized patient-centered outpatient gynecology experience has not previously been described in the medical literature. Optimizing the found domains of hospitality, comfort measures, pain and anxiety control, privacy, and diverse representationin the outpatient gynecologic experience within the outpatient care setting may ultimately result in improved patient satisfaction, patient well-being, and adherence to care through maximizing patient-centered care. We created a checklist of suggestions, including offering analgesics during office-based procedures and tailoring the floorplan to maximize privacy (FIGURE 5), for improving the outpatient gynecology experience based on our findings.

Prior data on patient satisfaction and outcomes
Improving patient satisfaction with health care is a priority for both clinicians and hospital systems. Prior studies have revealed only variable associations between patient satisfaction, safety, and clinical outcomes. One study involving the analysis of clinical and operational data from 171 hospitals found that hospital size, surgical volume, and low mortality rates were associated with higher patient satisfaction, while favorable surgical outcomes did not consistently correlate with higher Hospital Consumer Assessment of Healthcare Provers and Systems (HCAHPS) scores.10 Smaller, lower-volume hospitals earned higher satisfaction scores related to cleanliness, quietness, and receiving help measures.10 It has also been shown that the strongest predictors of patient satisfaction with the hospital childbirth experience included items related to staff communication, compassion, empathy, and respect.11 These data suggest that patient satisfaction is likely more significantly impacted by factors other than patient safety and effectiveness, and this was supported by the findings of our analysis. The growing body of literature associating a sense of psychological and physical safety within the health care system and improved patient outcomes and experience suggests that the data gathered from public commentary such as that presented here is extremely important for galvanizing change within the US health care system.
In one systematic review, the relationship between patient-centered care and clinical outcomes was mixed, although generally the association was positive.12 Additionally, patient-centered care was often associated with increased patient satisfaction and well-being. Some studies suggest that patient well-being and satisfaction also may be associated with improved adherence and self-management behaviors.12,13 Overall, optimizing patient-centered care may lead to improved patient satisfaction and potentially improved clinical outcomes.
Additionally, increasing diverse representation in patient materials and illustrations may help to improve the patient experience. Louie and colleagues found that dark skin tones were represented in only 4.5% of 4,146 images from anatomy texts analyzed in 2018.14 Similarly, a photogrammetric analysis of medical images utilized in New England Journal of Medicine found that only 18% of images depicted non-white skin.15 More recent efforts to create a royalty-free digital gallery of images reflecting bodies with diverse skin tones, body shapes, body hair, and age as well as transgender and nonbinary people have been discussed in the lay press.9 Based on our findings, social media users value and are actively seeking diversity in representation and imagery during their outpatient gynecology experience.
Opportunities for future study
Our research utilized social media as a diverse and accessible source of information; however, there are significant opportunities to refine the methodologic approach to answering the fundamental question of creating the patient-centered gynecologic experience. This type of study has not yet been conducted; however, the richness of the information from this current analysis could be informative to survey creation. Future research on this subject outside of social media could bolster the generalizability of our conclusions and the ability to report on qualitative findings in the setting of known patient demographics.
Social media remains a powerful tool as evidenced by this study, and continued use and observation of trending themes among patients is essential. The influence of social media will remain important for answering questions in gynecology and beyond.
Our work is strengthened by social media’s low threshold for use and the ability for widespread access to a diverse group of users. Additionally, social media allows for many responses to be collected in a timely manner, giving strength to the abstracted themes. The constant production of data by X users and their accessibility provide the opportunity for greater geographic coverage in those surveyed.4 Crowdsourced public opinion also has the advantage of producing qualitative metrics in the form of likes and retweets that may provide a reliable measure of public support or engagement.1
Future studies should examine ways to implement the suggested improvements to the office setting in a cost-effective manner and follow both subjective patient-reported outcomes as well as objective data after implementation, as these changes may have implications for much broader public health crises, such as maternal morbidity and mortality.
Study limitations. Our study is limited by the inherent biases and confounders associated with utilizing data derived from social media. Specifically, not all patients who seek outpatient gynecologic care utilize social media and/or X; using a “like” as a surrogate for endorsement of an idea by an identified party limits the generalizability of the data.
The initial Twitter query specified, “I’m asking women”, which may have altered the intended study population, influenced the analysis, and affected the representativeness of the sample through utilizing non ̶inclusive language. While non-binary/transgender care and inclusivity emerged as a theme discussed with the tweets, it is unclear if this represents an independent theme or rather a reaction to the non–inclusive language within the original tweet. ●
The data abstracted was analyzed with Dedoose1 software using a convenience sample and a mixed-methods analysis. Utilizing X (formerly Twitter and referred here as such given the time the study was conducted) for crowdsourcing functions similarly to an open survey. In the absence of similar analyses, a modified Checklist for Reporting Results of Internet E-Surveys (CHERRIES) checklist was utilized to organize our approach.2
This analysis was comprised of information freely available in the public domain, and the study was classified as IRB exempt. Ethical considerations were made for the fact that this is open access information and participants can reasonably expect their responses to be viewed by the public.3 As this question was not originally intended for research purposes, there was not a formalized development, recruitment, or consent process. The survey was not advertised beyond the original posting on Twitter, and the organic interest that it generated online. No incentives were offered to participants, and all participation was voluntary. There is no mechanism on Twitter for respondents to edit their response, although responses can be deleted. Unique visitors or viewers beyond posted impressions in response to the original tweet could not be determined.
Twitter thread responses were reviewed, and all completed and posted responses to the original Twitter query with 100 or greater “likes” were included in the analysis. These tweets were abstracted from Twitter between December 17, 2021, and December 27, 2021. At the time of tweet abstraction, engagement metrics, including the numbers of likes, retweets, and replies, were recorded. Additionally, author characteristics were abstracted, including author verification status and association with health care, as described in their Twitter profile. Definition of an individual associated with health care was broad and included physicians, advanced practice providers, nurses, first responders, and allied health professionals.
A total of 131 tweets met inclusion criteria and were uploaded for analysis using Dedoose qualitative analytic software.1 Two authors independently utilized a qualitative analysis to code the isolated tweets and identify thematic patterns among them. Uploaded tweets were additionally coded based on ranges of likes: 100-500; 501-1,000; 1,001-1,999; and >2,000. Tweets were coded for author verification status and whether or not the author was associated with the health care field. Themes were identified and defined during the coding process and were shared between the two authors. A total of 18 themes were identified, with 1 theme having 4 subdivisions. Interrater reliability testing was performed using Dedoose1 software and resulted with a pooled Cohen’s Kappa of 0.63, indicating “good” agreement between authors, which is an adequate level of agreement per the Dedoose software guidelines.
References
1. Dedoose website. Accessed July 28, 2022. https://www .dedoose.com/
2. Eysenbach G. Improving the quality of web surveys: the checklist for reporting results of internet e-surveys (CHERRIES) [published correction appears in J Med Internet Res. 2012;14:e8. doi:10.2196/jmir.2042]. J Med Internet Res. 2004;6:e34. doi:10.2196/jmir.6.3.e34
3. Townsend L, Wallace C. Social media research: a guide to ethics [University of Glasgow Information for the Media website]. Accessed March 2, 2023. https://www.gla.ac.uk /media/Media_487729_smxx.pdf
- Garvey MD, Samuel J, Pelaez A. Would you please like my tweet?! An artificially intelligent, generative probabilistic, and econometric based system design for popularity-driven tweet content generation. Decis Support Syst. 2021;144:113497. doi: 10.1016/j.dss.2021.113497
- Twitter Revenue and Usage Statistics (2023). Business of apps. Published August 10, 2023. Accessed September 19, 2023. https://www.businessofapps.com/data/twitter-statistics/
- Doan AE, Bogen KW, Higgins E. A content analysis of twitter backlash to Georgia’s abortion ban. Sex Reprod Healthc. 2022;31:100689. doi:10.1016/j.srhc.2021.100689
- Roberts H, Sadler J, Chapman L. The value of Twitter data for determining the emotional responses of people to urban green spaces: a case study and critical evaluation. Urban Stud. 2019;56:818-835. doi: 10.1177/0042098017748544
- Stewart R [@stuboo]. I have the opportunity to design my office from scratch. I’m asking women. How would you design/optimize a visit to the gynecologist’s office? problems frustrations solutions No detail is too small. If I’ve ever had a tweet worthy of virality, it’s this one. RT. Twitter. Published December 5, 2021. Accessed March 1, 2023. https://twitter .com/stuboo/status/1467522852664532994
- A gynecologist asked Twitter how he should redesign his office. The answers he got were about deeper health care issues. Fortune. Accessed March 2, 2023. https://fortune .com/2021/12/07/gynecologist-twitter-question/
- Anderson GD, Nelson-Becker C, Hannigan EV, et al. A patientcentered health care delivery system by a university obstetrics and gynecology department. Obstet Gynecol. 2005;105:205210. doi:10.1097/01.AOG.0000146288.28195.27
- Ades V, Wu SX, Rabinowitz E, et al. An integrated, traumainformed care model for female survivors of sexual violence: the engage, motivate, protect, organize, self-worth, educate, respect (EMPOWER) clinic. Obstet Gynecol. 2019;133:803809. doi:10.1097/AOG.0000000000003186
- Gordon D. Health equity comes to medical illustrations with launch of new image library. Forbes. Accessed March 2023. https://www.forbes.com/sites/debgordon/2022/05/11 /health-equity-comes-to-medical-illustrations-with-launch -of-new-image-library/
- Kennedy GD, Tevis SE, Kent KC. Is there a relationship between patient satisfaction and favorable outcomes? Ann Surg. 2014;260:592-600. doi:10.1097/SLA.0000000000000932
- Gregory KD, Korst LM, Saeb S, et al. Childbirth-specific patient-reported outcomes as predictors of hospital satisfaction. Am J Obstet Gynecol. 2019;220:201.e1-201.e19. doi:10.1016/j.ajog.2018.10.093
- Rathert C, Wyrwich MD, Boren SA. Patient-centered care and outcomes: a systematic review of the literature. Med Care Res Rev. 2013;70:351-379. doi:10.1177/1077558712465774
- Kahn KL, Schneider EC, Malin JL, et al. Patient-centered experiences in breast cancer: predicting long-term adherence to tamoxifen use. Med Care. 2007;45:431-439. doi:10.1097/01 .mlr.0000257193.10760.7
- Louie P, Wilkes R. Representations of race and skin tone in medical textbook imagery. Soc Sci Med. 2018;202:38-42. doi:10.1016/j.socscimed.2018.02.023
- Massie JP, Cho DY, Kneib CJ, et al. A picture of modern medicine: race and visual representation in medical literature. J Natl Med Assoc. 2021;113:88-94. doi:10.1016/j.jnma.2020.07.013
There has been increasing awareness of a need for creating a more patient-centered experience with outpatient gynecology; however, very little data exist about what interventions are important to patients. Given social media’s ease of use and ability for widespread access to a diverse group of users, it has the potential to be a powerful tool for qualitative research questions without the difficulties of cost, transportation, transcription, etc. required of a focus group. Crowdsourced public opinion also has the advantage of producing qualitative metrics in the form of “likes” that, at scale, can provide a reliable measure of public support or engagement for a particular concept.1 Particularly for topics that are controversial or novel, X (formerly Twitter, and referred to as Twitter intermittently throughout this article based on the time the study was conducted), with 300 million monthly users,2 has become a popular tool for general and health care ̶ focused content and sentiment analysis.3,4 This study presents a qualitative analysis of themes from a crowdsourced request on Twitter to design the ideal outpatient gynecologic experience that subsequently went “viral”.5,6
When asked to design the optimized outpatient gynecology experience, social media users expressed:
- hospitality, comfort, and pain control as frequent themes
- preserving privacy and acknowledgement of voluntary nulliparity as frequent themes
- a desire for diverse imagery and representation related to race, LGBTQIA+ themes, age, and weight/body type within the office setting
- a call for a sense of psychological safety within gynecology
Why the need for our research question on patient-centered gyn care
While the body of literature on patient-centered health care has grown rapidly in recent years, a patient-centered outpatient gynecology experience has not yet been described in the medical literature.
Patient-centered office design, driven by cultural sensitivity, has been shown in other studies to be both appreciated by established patients and a viable business strategy to attract new patients.7 Topics such as pain control, trauma-informed care in gynecologyclinics,8 and diverse representation in patient materials and illustrations9 have been popular topics in medicine and in the lay press. Our primary aim in our research was to utilize feedback from the question posed to quantify and rank patient-centered interventions in a gynecology office. These themes and others that emerged in our analysis were used to suggest b
What we asked social media users. The survey query to social media users, “I have the opportunity to design my office from scratch. I’m asking women: How would you design/optimize a visit to the gynecologist’s office?” was crowd-sourced via Twitter on December 5, 2021.5 Given a robust response to the query, it provided an opportunity for a qualitative research study exploring social media users’ perspectives on optimizing outpatient gynecologic care, although the original question was not planned for research utilization.
What we found
By December 27, 2021, the original tweet had earned 9,411 likes; 2,143 retweets; and 3,400 replies. Of this group, we analyzed 131 tweets, all of which had 100 or greater likes on Twitter at the time of the review. The majority of analyzed tweets earned between 100 ̶ 500 likes (75/131; 57.3%), while 22.9% (30/131) had 501 ̶ 1,000 likes, 11.5% (15/131) had >2,000 likes, and 8.4% (11/131) had 1,001 ̶ 1,999 likes.
Identified themes within the tweets analyzed included: medical education, comfort improvements, continuity of care, disability accommodations/accessibility, economic accessibility, nonbinary/transgender care and inclusivity, general layout/floorplan, hospitality, aid for intimate partner violence, childcare accessibility, multi-disciplinary care access, pain/anxiety control, sensitivity toward pregnancy loss/fertility issues, privacy issues, professionalism, representation (subdivided into race, LGBTQIA+, age, and weight/body type), trauma-informed care, and acknowledgement of voluntary nulliparity/support for reproductive choices (TABLE 1). TABLE 2 lists examples of popular tweets by selected themes.


Frequent themes. The most frequently occurring themes within the 131 analyzed tweets (FIGURE 1) were:
- hospitality (77 occurrences)
- comfort improvements (75 occurrences)
- general layout/floorplan (75 occurrences)
- pain/anxiety control (55 occurrences)
- representation (53 occurrences).

Popular themes. Defined as those with more than 1,000 likes at the time of analysis (FIGURE 2), the most popular themes included:
- privacy issues (48.5% of related tweets with >1,000 likes)
- voluntary nulliparity (37.0% of related tweets with >1,000 likes)
- general layout/floorplan (33.4% of related tweets with >1,000 likes)
- representation (32.1% of related tweets with >1,000 likes)
- hospitality (31.3% of related tweets with >1,000 likes).

A sub-analysis of themes related to specific types of representation—race, LGBTQIA+, age, and weight/body type was performed. Tweets related to diverse weight/body type representation occurred most frequently (19 code occurrences; FIGURE 3). Similarly, tweets related to the representation of diverse races and the LGBTQIA+ community each comprised 26% of the total representation-based tweets. In terms of popularity as described above, 51.4% of tweets describing racial representation earned >1,000 likes (FIGURE 4).


Tweet demographics. Seven (7/131; 5.3%) of the tweet authors were verified Twitter users and 35 (35/131; 26.7%) authors reported working in the health care field within their Twitter profile description.
Continue to: Implementing our feedback can enhance patient experience and care...
Implementing our feedback can enhance patient experience and care
Our study provides a unique view of the patient perspective through analyzed crowdsourced public opinion via Twitter. To our knowledge, an optimized patient-centered outpatient gynecology experience has not previously been described in the medical literature. Optimizing the found domains of hospitality, comfort measures, pain and anxiety control, privacy, and diverse representationin the outpatient gynecologic experience within the outpatient care setting may ultimately result in improved patient satisfaction, patient well-being, and adherence to care through maximizing patient-centered care. We created a checklist of suggestions, including offering analgesics during office-based procedures and tailoring the floorplan to maximize privacy (FIGURE 5), for improving the outpatient gynecology experience based on our findings.

Prior data on patient satisfaction and outcomes
Improving patient satisfaction with health care is a priority for both clinicians and hospital systems. Prior studies have revealed only variable associations between patient satisfaction, safety, and clinical outcomes. One study involving the analysis of clinical and operational data from 171 hospitals found that hospital size, surgical volume, and low mortality rates were associated with higher patient satisfaction, while favorable surgical outcomes did not consistently correlate with higher Hospital Consumer Assessment of Healthcare Provers and Systems (HCAHPS) scores.10 Smaller, lower-volume hospitals earned higher satisfaction scores related to cleanliness, quietness, and receiving help measures.10 It has also been shown that the strongest predictors of patient satisfaction with the hospital childbirth experience included items related to staff communication, compassion, empathy, and respect.11 These data suggest that patient satisfaction is likely more significantly impacted by factors other than patient safety and effectiveness, and this was supported by the findings of our analysis. The growing body of literature associating a sense of psychological and physical safety within the health care system and improved patient outcomes and experience suggests that the data gathered from public commentary such as that presented here is extremely important for galvanizing change within the US health care system.
In one systematic review, the relationship between patient-centered care and clinical outcomes was mixed, although generally the association was positive.12 Additionally, patient-centered care was often associated with increased patient satisfaction and well-being. Some studies suggest that patient well-being and satisfaction also may be associated with improved adherence and self-management behaviors.12,13 Overall, optimizing patient-centered care may lead to improved patient satisfaction and potentially improved clinical outcomes.
Additionally, increasing diverse representation in patient materials and illustrations may help to improve the patient experience. Louie and colleagues found that dark skin tones were represented in only 4.5% of 4,146 images from anatomy texts analyzed in 2018.14 Similarly, a photogrammetric analysis of medical images utilized in New England Journal of Medicine found that only 18% of images depicted non-white skin.15 More recent efforts to create a royalty-free digital gallery of images reflecting bodies with diverse skin tones, body shapes, body hair, and age as well as transgender and nonbinary people have been discussed in the lay press.9 Based on our findings, social media users value and are actively seeking diversity in representation and imagery during their outpatient gynecology experience.
Opportunities for future study
Our research utilized social media as a diverse and accessible source of information; however, there are significant opportunities to refine the methodologic approach to answering the fundamental question of creating the patient-centered gynecologic experience. This type of study has not yet been conducted; however, the richness of the information from this current analysis could be informative to survey creation. Future research on this subject outside of social media could bolster the generalizability of our conclusions and the ability to report on qualitative findings in the setting of known patient demographics.
Social media remains a powerful tool as evidenced by this study, and continued use and observation of trending themes among patients is essential. The influence of social media will remain important for answering questions in gynecology and beyond.
Our work is strengthened by social media’s low threshold for use and the ability for widespread access to a diverse group of users. Additionally, social media allows for many responses to be collected in a timely manner, giving strength to the abstracted themes. The constant production of data by X users and their accessibility provide the opportunity for greater geographic coverage in those surveyed.4 Crowdsourced public opinion also has the advantage of producing qualitative metrics in the form of likes and retweets that may provide a reliable measure of public support or engagement.1
Future studies should examine ways to implement the suggested improvements to the office setting in a cost-effective manner and follow both subjective patient-reported outcomes as well as objective data after implementation, as these changes may have implications for much broader public health crises, such as maternal morbidity and mortality.
Study limitations. Our study is limited by the inherent biases and confounders associated with utilizing data derived from social media. Specifically, not all patients who seek outpatient gynecologic care utilize social media and/or X; using a “like” as a surrogate for endorsement of an idea by an identified party limits the generalizability of the data.
The initial Twitter query specified, “I’m asking women”, which may have altered the intended study population, influenced the analysis, and affected the representativeness of the sample through utilizing non ̶inclusive language. While non-binary/transgender care and inclusivity emerged as a theme discussed with the tweets, it is unclear if this represents an independent theme or rather a reaction to the non–inclusive language within the original tweet. ●
The data abstracted was analyzed with Dedoose1 software using a convenience sample and a mixed-methods analysis. Utilizing X (formerly Twitter and referred here as such given the time the study was conducted) for crowdsourcing functions similarly to an open survey. In the absence of similar analyses, a modified Checklist for Reporting Results of Internet E-Surveys (CHERRIES) checklist was utilized to organize our approach.2
This analysis was comprised of information freely available in the public domain, and the study was classified as IRB exempt. Ethical considerations were made for the fact that this is open access information and participants can reasonably expect their responses to be viewed by the public.3 As this question was not originally intended for research purposes, there was not a formalized development, recruitment, or consent process. The survey was not advertised beyond the original posting on Twitter, and the organic interest that it generated online. No incentives were offered to participants, and all participation was voluntary. There is no mechanism on Twitter for respondents to edit their response, although responses can be deleted. Unique visitors or viewers beyond posted impressions in response to the original tweet could not be determined.
Twitter thread responses were reviewed, and all completed and posted responses to the original Twitter query with 100 or greater “likes” were included in the analysis. These tweets were abstracted from Twitter between December 17, 2021, and December 27, 2021. At the time of tweet abstraction, engagement metrics, including the numbers of likes, retweets, and replies, were recorded. Additionally, author characteristics were abstracted, including author verification status and association with health care, as described in their Twitter profile. Definition of an individual associated with health care was broad and included physicians, advanced practice providers, nurses, first responders, and allied health professionals.
A total of 131 tweets met inclusion criteria and were uploaded for analysis using Dedoose qualitative analytic software.1 Two authors independently utilized a qualitative analysis to code the isolated tweets and identify thematic patterns among them. Uploaded tweets were additionally coded based on ranges of likes: 100-500; 501-1,000; 1,001-1,999; and >2,000. Tweets were coded for author verification status and whether or not the author was associated with the health care field. Themes were identified and defined during the coding process and were shared between the two authors. A total of 18 themes were identified, with 1 theme having 4 subdivisions. Interrater reliability testing was performed using Dedoose1 software and resulted with a pooled Cohen’s Kappa of 0.63, indicating “good” agreement between authors, which is an adequate level of agreement per the Dedoose software guidelines.
References
1. Dedoose website. Accessed July 28, 2022. https://www .dedoose.com/
2. Eysenbach G. Improving the quality of web surveys: the checklist for reporting results of internet e-surveys (CHERRIES) [published correction appears in J Med Internet Res. 2012;14:e8. doi:10.2196/jmir.2042]. J Med Internet Res. 2004;6:e34. doi:10.2196/jmir.6.3.e34
3. Townsend L, Wallace C. Social media research: a guide to ethics [University of Glasgow Information for the Media website]. Accessed March 2, 2023. https://www.gla.ac.uk /media/Media_487729_smxx.pdf
There has been increasing awareness of a need for creating a more patient-centered experience with outpatient gynecology; however, very little data exist about what interventions are important to patients. Given social media’s ease of use and ability for widespread access to a diverse group of users, it has the potential to be a powerful tool for qualitative research questions without the difficulties of cost, transportation, transcription, etc. required of a focus group. Crowdsourced public opinion also has the advantage of producing qualitative metrics in the form of “likes” that, at scale, can provide a reliable measure of public support or engagement for a particular concept.1 Particularly for topics that are controversial or novel, X (formerly Twitter, and referred to as Twitter intermittently throughout this article based on the time the study was conducted), with 300 million monthly users,2 has become a popular tool for general and health care ̶ focused content and sentiment analysis.3,4 This study presents a qualitative analysis of themes from a crowdsourced request on Twitter to design the ideal outpatient gynecologic experience that subsequently went “viral”.5,6
When asked to design the optimized outpatient gynecology experience, social media users expressed:
- hospitality, comfort, and pain control as frequent themes
- preserving privacy and acknowledgement of voluntary nulliparity as frequent themes
- a desire for diverse imagery and representation related to race, LGBTQIA+ themes, age, and weight/body type within the office setting
- a call for a sense of psychological safety within gynecology
Why the need for our research question on patient-centered gyn care
While the body of literature on patient-centered health care has grown rapidly in recent years, a patient-centered outpatient gynecology experience has not yet been described in the medical literature.
Patient-centered office design, driven by cultural sensitivity, has been shown in other studies to be both appreciated by established patients and a viable business strategy to attract new patients.7 Topics such as pain control, trauma-informed care in gynecologyclinics,8 and diverse representation in patient materials and illustrations9 have been popular topics in medicine and in the lay press. Our primary aim in our research was to utilize feedback from the question posed to quantify and rank patient-centered interventions in a gynecology office. These themes and others that emerged in our analysis were used to suggest b
What we asked social media users. The survey query to social media users, “I have the opportunity to design my office from scratch. I’m asking women: How would you design/optimize a visit to the gynecologist’s office?” was crowd-sourced via Twitter on December 5, 2021.5 Given a robust response to the query, it provided an opportunity for a qualitative research study exploring social media users’ perspectives on optimizing outpatient gynecologic care, although the original question was not planned for research utilization.
What we found
By December 27, 2021, the original tweet had earned 9,411 likes; 2,143 retweets; and 3,400 replies. Of this group, we analyzed 131 tweets, all of which had 100 or greater likes on Twitter at the time of the review. The majority of analyzed tweets earned between 100 ̶ 500 likes (75/131; 57.3%), while 22.9% (30/131) had 501 ̶ 1,000 likes, 11.5% (15/131) had >2,000 likes, and 8.4% (11/131) had 1,001 ̶ 1,999 likes.
Identified themes within the tweets analyzed included: medical education, comfort improvements, continuity of care, disability accommodations/accessibility, economic accessibility, nonbinary/transgender care and inclusivity, general layout/floorplan, hospitality, aid for intimate partner violence, childcare accessibility, multi-disciplinary care access, pain/anxiety control, sensitivity toward pregnancy loss/fertility issues, privacy issues, professionalism, representation (subdivided into race, LGBTQIA+, age, and weight/body type), trauma-informed care, and acknowledgement of voluntary nulliparity/support for reproductive choices (TABLE 1). TABLE 2 lists examples of popular tweets by selected themes.


Frequent themes. The most frequently occurring themes within the 131 analyzed tweets (FIGURE 1) were:
- hospitality (77 occurrences)
- comfort improvements (75 occurrences)
- general layout/floorplan (75 occurrences)
- pain/anxiety control (55 occurrences)
- representation (53 occurrences).

Popular themes. Defined as those with more than 1,000 likes at the time of analysis (FIGURE 2), the most popular themes included:
- privacy issues (48.5% of related tweets with >1,000 likes)
- voluntary nulliparity (37.0% of related tweets with >1,000 likes)
- general layout/floorplan (33.4% of related tweets with >1,000 likes)
- representation (32.1% of related tweets with >1,000 likes)
- hospitality (31.3% of related tweets with >1,000 likes).

A sub-analysis of themes related to specific types of representation—race, LGBTQIA+, age, and weight/body type was performed. Tweets related to diverse weight/body type representation occurred most frequently (19 code occurrences; FIGURE 3). Similarly, tweets related to the representation of diverse races and the LGBTQIA+ community each comprised 26% of the total representation-based tweets. In terms of popularity as described above, 51.4% of tweets describing racial representation earned >1,000 likes (FIGURE 4).


Tweet demographics. Seven (7/131; 5.3%) of the tweet authors were verified Twitter users and 35 (35/131; 26.7%) authors reported working in the health care field within their Twitter profile description.
Continue to: Implementing our feedback can enhance patient experience and care...
Implementing our feedback can enhance patient experience and care
Our study provides a unique view of the patient perspective through analyzed crowdsourced public opinion via Twitter. To our knowledge, an optimized patient-centered outpatient gynecology experience has not previously been described in the medical literature. Optimizing the found domains of hospitality, comfort measures, pain and anxiety control, privacy, and diverse representationin the outpatient gynecologic experience within the outpatient care setting may ultimately result in improved patient satisfaction, patient well-being, and adherence to care through maximizing patient-centered care. We created a checklist of suggestions, including offering analgesics during office-based procedures and tailoring the floorplan to maximize privacy (FIGURE 5), for improving the outpatient gynecology experience based on our findings.

Prior data on patient satisfaction and outcomes
Improving patient satisfaction with health care is a priority for both clinicians and hospital systems. Prior studies have revealed only variable associations between patient satisfaction, safety, and clinical outcomes. One study involving the analysis of clinical and operational data from 171 hospitals found that hospital size, surgical volume, and low mortality rates were associated with higher patient satisfaction, while favorable surgical outcomes did not consistently correlate with higher Hospital Consumer Assessment of Healthcare Provers and Systems (HCAHPS) scores.10 Smaller, lower-volume hospitals earned higher satisfaction scores related to cleanliness, quietness, and receiving help measures.10 It has also been shown that the strongest predictors of patient satisfaction with the hospital childbirth experience included items related to staff communication, compassion, empathy, and respect.11 These data suggest that patient satisfaction is likely more significantly impacted by factors other than patient safety and effectiveness, and this was supported by the findings of our analysis. The growing body of literature associating a sense of psychological and physical safety within the health care system and improved patient outcomes and experience suggests that the data gathered from public commentary such as that presented here is extremely important for galvanizing change within the US health care system.
In one systematic review, the relationship between patient-centered care and clinical outcomes was mixed, although generally the association was positive.12 Additionally, patient-centered care was often associated with increased patient satisfaction and well-being. Some studies suggest that patient well-being and satisfaction also may be associated with improved adherence and self-management behaviors.12,13 Overall, optimizing patient-centered care may lead to improved patient satisfaction and potentially improved clinical outcomes.
Additionally, increasing diverse representation in patient materials and illustrations may help to improve the patient experience. Louie and colleagues found that dark skin tones were represented in only 4.5% of 4,146 images from anatomy texts analyzed in 2018.14 Similarly, a photogrammetric analysis of medical images utilized in New England Journal of Medicine found that only 18% of images depicted non-white skin.15 More recent efforts to create a royalty-free digital gallery of images reflecting bodies with diverse skin tones, body shapes, body hair, and age as well as transgender and nonbinary people have been discussed in the lay press.9 Based on our findings, social media users value and are actively seeking diversity in representation and imagery during their outpatient gynecology experience.
Opportunities for future study
Our research utilized social media as a diverse and accessible source of information; however, there are significant opportunities to refine the methodologic approach to answering the fundamental question of creating the patient-centered gynecologic experience. This type of study has not yet been conducted; however, the richness of the information from this current analysis could be informative to survey creation. Future research on this subject outside of social media could bolster the generalizability of our conclusions and the ability to report on qualitative findings in the setting of known patient demographics.
Social media remains a powerful tool as evidenced by this study, and continued use and observation of trending themes among patients is essential. The influence of social media will remain important for answering questions in gynecology and beyond.
Our work is strengthened by social media’s low threshold for use and the ability for widespread access to a diverse group of users. Additionally, social media allows for many responses to be collected in a timely manner, giving strength to the abstracted themes. The constant production of data by X users and their accessibility provide the opportunity for greater geographic coverage in those surveyed.4 Crowdsourced public opinion also has the advantage of producing qualitative metrics in the form of likes and retweets that may provide a reliable measure of public support or engagement.1
Future studies should examine ways to implement the suggested improvements to the office setting in a cost-effective manner and follow both subjective patient-reported outcomes as well as objective data after implementation, as these changes may have implications for much broader public health crises, such as maternal morbidity and mortality.
Study limitations. Our study is limited by the inherent biases and confounders associated with utilizing data derived from social media. Specifically, not all patients who seek outpatient gynecologic care utilize social media and/or X; using a “like” as a surrogate for endorsement of an idea by an identified party limits the generalizability of the data.
The initial Twitter query specified, “I’m asking women”, which may have altered the intended study population, influenced the analysis, and affected the representativeness of the sample through utilizing non ̶inclusive language. While non-binary/transgender care and inclusivity emerged as a theme discussed with the tweets, it is unclear if this represents an independent theme or rather a reaction to the non–inclusive language within the original tweet. ●
The data abstracted was analyzed with Dedoose1 software using a convenience sample and a mixed-methods analysis. Utilizing X (formerly Twitter and referred here as such given the time the study was conducted) for crowdsourcing functions similarly to an open survey. In the absence of similar analyses, a modified Checklist for Reporting Results of Internet E-Surveys (CHERRIES) checklist was utilized to organize our approach.2
This analysis was comprised of information freely available in the public domain, and the study was classified as IRB exempt. Ethical considerations were made for the fact that this is open access information and participants can reasonably expect their responses to be viewed by the public.3 As this question was not originally intended for research purposes, there was not a formalized development, recruitment, or consent process. The survey was not advertised beyond the original posting on Twitter, and the organic interest that it generated online. No incentives were offered to participants, and all participation was voluntary. There is no mechanism on Twitter for respondents to edit their response, although responses can be deleted. Unique visitors or viewers beyond posted impressions in response to the original tweet could not be determined.
Twitter thread responses were reviewed, and all completed and posted responses to the original Twitter query with 100 or greater “likes” were included in the analysis. These tweets were abstracted from Twitter between December 17, 2021, and December 27, 2021. At the time of tweet abstraction, engagement metrics, including the numbers of likes, retweets, and replies, were recorded. Additionally, author characteristics were abstracted, including author verification status and association with health care, as described in their Twitter profile. Definition of an individual associated with health care was broad and included physicians, advanced practice providers, nurses, first responders, and allied health professionals.
A total of 131 tweets met inclusion criteria and were uploaded for analysis using Dedoose qualitative analytic software.1 Two authors independently utilized a qualitative analysis to code the isolated tweets and identify thematic patterns among them. Uploaded tweets were additionally coded based on ranges of likes: 100-500; 501-1,000; 1,001-1,999; and >2,000. Tweets were coded for author verification status and whether or not the author was associated with the health care field. Themes were identified and defined during the coding process and were shared between the two authors. A total of 18 themes were identified, with 1 theme having 4 subdivisions. Interrater reliability testing was performed using Dedoose1 software and resulted with a pooled Cohen’s Kappa of 0.63, indicating “good” agreement between authors, which is an adequate level of agreement per the Dedoose software guidelines.
References
1. Dedoose website. Accessed July 28, 2022. https://www .dedoose.com/
2. Eysenbach G. Improving the quality of web surveys: the checklist for reporting results of internet e-surveys (CHERRIES) [published correction appears in J Med Internet Res. 2012;14:e8. doi:10.2196/jmir.2042]. J Med Internet Res. 2004;6:e34. doi:10.2196/jmir.6.3.e34
3. Townsend L, Wallace C. Social media research: a guide to ethics [University of Glasgow Information for the Media website]. Accessed March 2, 2023. https://www.gla.ac.uk /media/Media_487729_smxx.pdf
- Garvey MD, Samuel J, Pelaez A. Would you please like my tweet?! An artificially intelligent, generative probabilistic, and econometric based system design for popularity-driven tweet content generation. Decis Support Syst. 2021;144:113497. doi: 10.1016/j.dss.2021.113497
- Twitter Revenue and Usage Statistics (2023). Business of apps. Published August 10, 2023. Accessed September 19, 2023. https://www.businessofapps.com/data/twitter-statistics/
- Doan AE, Bogen KW, Higgins E. A content analysis of twitter backlash to Georgia’s abortion ban. Sex Reprod Healthc. 2022;31:100689. doi:10.1016/j.srhc.2021.100689
- Roberts H, Sadler J, Chapman L. The value of Twitter data for determining the emotional responses of people to urban green spaces: a case study and critical evaluation. Urban Stud. 2019;56:818-835. doi: 10.1177/0042098017748544
- Stewart R [@stuboo]. I have the opportunity to design my office from scratch. I’m asking women. How would you design/optimize a visit to the gynecologist’s office? problems frustrations solutions No detail is too small. If I’ve ever had a tweet worthy of virality, it’s this one. RT. Twitter. Published December 5, 2021. Accessed March 1, 2023. https://twitter .com/stuboo/status/1467522852664532994
- A gynecologist asked Twitter how he should redesign his office. The answers he got were about deeper health care issues. Fortune. Accessed March 2, 2023. https://fortune .com/2021/12/07/gynecologist-twitter-question/
- Anderson GD, Nelson-Becker C, Hannigan EV, et al. A patientcentered health care delivery system by a university obstetrics and gynecology department. Obstet Gynecol. 2005;105:205210. doi:10.1097/01.AOG.0000146288.28195.27
- Ades V, Wu SX, Rabinowitz E, et al. An integrated, traumainformed care model for female survivors of sexual violence: the engage, motivate, protect, organize, self-worth, educate, respect (EMPOWER) clinic. Obstet Gynecol. 2019;133:803809. doi:10.1097/AOG.0000000000003186
- Gordon D. Health equity comes to medical illustrations with launch of new image library. Forbes. Accessed March 2023. https://www.forbes.com/sites/debgordon/2022/05/11 /health-equity-comes-to-medical-illustrations-with-launch -of-new-image-library/
- Kennedy GD, Tevis SE, Kent KC. Is there a relationship between patient satisfaction and favorable outcomes? Ann Surg. 2014;260:592-600. doi:10.1097/SLA.0000000000000932
- Gregory KD, Korst LM, Saeb S, et al. Childbirth-specific patient-reported outcomes as predictors of hospital satisfaction. Am J Obstet Gynecol. 2019;220:201.e1-201.e19. doi:10.1016/j.ajog.2018.10.093
- Rathert C, Wyrwich MD, Boren SA. Patient-centered care and outcomes: a systematic review of the literature. Med Care Res Rev. 2013;70:351-379. doi:10.1177/1077558712465774
- Kahn KL, Schneider EC, Malin JL, et al. Patient-centered experiences in breast cancer: predicting long-term adherence to tamoxifen use. Med Care. 2007;45:431-439. doi:10.1097/01 .mlr.0000257193.10760.7
- Louie P, Wilkes R. Representations of race and skin tone in medical textbook imagery. Soc Sci Med. 2018;202:38-42. doi:10.1016/j.socscimed.2018.02.023
- Massie JP, Cho DY, Kneib CJ, et al. A picture of modern medicine: race and visual representation in medical literature. J Natl Med Assoc. 2021;113:88-94. doi:10.1016/j.jnma.2020.07.013
- Garvey MD, Samuel J, Pelaez A. Would you please like my tweet?! An artificially intelligent, generative probabilistic, and econometric based system design for popularity-driven tweet content generation. Decis Support Syst. 2021;144:113497. doi: 10.1016/j.dss.2021.113497
- Twitter Revenue and Usage Statistics (2023). Business of apps. Published August 10, 2023. Accessed September 19, 2023. https://www.businessofapps.com/data/twitter-statistics/
- Doan AE, Bogen KW, Higgins E. A content analysis of twitter backlash to Georgia’s abortion ban. Sex Reprod Healthc. 2022;31:100689. doi:10.1016/j.srhc.2021.100689
- Roberts H, Sadler J, Chapman L. The value of Twitter data for determining the emotional responses of people to urban green spaces: a case study and critical evaluation. Urban Stud. 2019;56:818-835. doi: 10.1177/0042098017748544
- Stewart R [@stuboo]. I have the opportunity to design my office from scratch. I’m asking women. How would you design/optimize a visit to the gynecologist’s office? problems frustrations solutions No detail is too small. If I’ve ever had a tweet worthy of virality, it’s this one. RT. Twitter. Published December 5, 2021. Accessed March 1, 2023. https://twitter .com/stuboo/status/1467522852664532994
- A gynecologist asked Twitter how he should redesign his office. The answers he got were about deeper health care issues. Fortune. Accessed March 2, 2023. https://fortune .com/2021/12/07/gynecologist-twitter-question/
- Anderson GD, Nelson-Becker C, Hannigan EV, et al. A patientcentered health care delivery system by a university obstetrics and gynecology department. Obstet Gynecol. 2005;105:205210. doi:10.1097/01.AOG.0000146288.28195.27
- Ades V, Wu SX, Rabinowitz E, et al. An integrated, traumainformed care model for female survivors of sexual violence: the engage, motivate, protect, organize, self-worth, educate, respect (EMPOWER) clinic. Obstet Gynecol. 2019;133:803809. doi:10.1097/AOG.0000000000003186
- Gordon D. Health equity comes to medical illustrations with launch of new image library. Forbes. Accessed March 2023. https://www.forbes.com/sites/debgordon/2022/05/11 /health-equity-comes-to-medical-illustrations-with-launch -of-new-image-library/
- Kennedy GD, Tevis SE, Kent KC. Is there a relationship between patient satisfaction and favorable outcomes? Ann Surg. 2014;260:592-600. doi:10.1097/SLA.0000000000000932
- Gregory KD, Korst LM, Saeb S, et al. Childbirth-specific patient-reported outcomes as predictors of hospital satisfaction. Am J Obstet Gynecol. 2019;220:201.e1-201.e19. doi:10.1016/j.ajog.2018.10.093
- Rathert C, Wyrwich MD, Boren SA. Patient-centered care and outcomes: a systematic review of the literature. Med Care Res Rev. 2013;70:351-379. doi:10.1177/1077558712465774
- Kahn KL, Schneider EC, Malin JL, et al. Patient-centered experiences in breast cancer: predicting long-term adherence to tamoxifen use. Med Care. 2007;45:431-439. doi:10.1097/01 .mlr.0000257193.10760.7
- Louie P, Wilkes R. Representations of race and skin tone in medical textbook imagery. Soc Sci Med. 2018;202:38-42. doi:10.1016/j.socscimed.2018.02.023
- Massie JP, Cho DY, Kneib CJ, et al. A picture of modern medicine: race and visual representation in medical literature. J Natl Med Assoc. 2021;113:88-94. doi:10.1016/j.jnma.2020.07.013
The HPV vaccine: Time for ObGyn physicians to up our game
CASE Sexually active woman asks about the HPV vaccine
A 26-year-old woman delivered her first child 4 weeks ago. She has had 3 lifetime sexual partners and is now in a mutually faithful monogamous relationship with her partner. She has no known history of sexually transmissible infections. She received only one Pap test 3 years ago, and the cytology showed no abnormal cells. This cervical specimen was not tested for human papillomavirus (HPV) DNA. At the time of her postpartum appointment, she inquires whether she is a candidate for the HPV vaccine.
What should be your response?
Genital HPV infection is the most common sexually transmissible infection in the United States. This virus is the cause of multiple genital malignancies, including cancers of the vagina, vulva, penis, anus, and cervix. The organism is also now the major cause of oropharyngeal cancer.
Of the more than 200 different HPV types that have been identified, 12 have been defined as oncogenic (high risk), and 8 to 12 types have been defined as possibly or probably oncogenic. The HPV strain with the highest risk of progression to cancer is HPV 16. The strains HPV 16 and 18 are responsible for approximately 70% of cases of cervical cancer. Each year in the United States, approximately 11,500 new cases of invasive cervical cancer occur. Unfortunately, this malignancy is responsible for about 4,000 deaths annually. Worldwide, HPV causes approximately 690,000 cancers each year.1
To a large extent, most cases of HPV infection would be preventable if patients were to take advantage of the remarkably effective HPV vaccine that is now available. However, acceptance of the vaccine has been disappointing. In 2020, only about half of adolescents, age 13 to 15, had received the appropriate number of vaccine doses.1
As ObGyn physicians, we can take several measures, in concert with our pediatrician colleagues, to improve HPV vaccination rates. In this article, I review the development of the HPV vaccine and describe the components, indications, dosing schedules, contraindications, adverse effects, and cost of the vaccine.
HPV vaccine development and expansion
The first HPV vaccine introduced in the United States was the recombinant quadrivalent vaccine (Gardasil; Merck); it was approved by the US Food and Drug Administration (FDA) in 2006. This vaccine is composed of viral-like particles unique to HPV 16 and 18 (the 2 most common causes of cervical, penile, anal, and oropharyngeal cancer) and HPV 6 and 11 (the 2 most common causes of genital warts). The formulation is prepared in baker’s yeast, and it elicits a robust production of neutralizing antibodies.2
In 2009, the FDA approved the bivalent vaccine (Cervarix; GlaxoSmithKline Biologicals). This vaccine contains viral-like particles unique to HPV 16 and 18, and it also induces a robust immune response. The vaccine is prepared in insect viral vectors.2
Both the quadrivalent and bivalent vaccines are no longer available in the United States. The only HPV vaccine currently marketed is the recombinant 9-valent vaccine (Gardasil 9; Merck), which was approved by the FDA in 2014. This newer vaccine targets the original 4 viral HPV strains in the quadrivalent vaccine (16, 18, 6, 11) plus 5 additional oncogenic strains: 31, 33, 45, 52, 58.2-4 The HPV strains targeted by this vaccine are responsible for approximately 90% of all cancers caused by HPV.
The 9-valent HPV vaccine, like the other 2, is highly effective in preventing cancers of the cervix, vagina, vulva, anus, penis; oropharyngeal cancers; and precancerous lesions such as genital warts.2-5 It will not, however, prevent the progression of preexisting infection or clear an infection that is already present at the time of vaccination.1
Although the original protocol for administration of the vaccine provided for 3 doses, recent studies indicate that 2 doses may be as effective as 3 in eliciting a favorable antibody response.6 There also is evidence that even a single dose of the vaccine can elicit a protective immune response.7 This encouraging finding is particularly important to public health officials responsible for developing HPV vaccination programs in low- and middle-resource countries.
Continue to: Target groups for the HPV vaccine...
Target groups for the HPV vaccine
The primary target group for the HPV vaccine is girls and boys who are aged 11 to 12 years. The key strategy is to immunize these individuals before they become sexually active. The vaccine also should be offered to children who are aged 9 to 10 years of age if they are judged to be at unusual risk, such as because of concern about sexual molestation. Children in these 2 age groups should receive 2 doses of the vaccine, with the second dose administered 6 to 12 months after the first dose.
The second target group for vaccination is individuals who are aged 13 to 26 years who have never been vaccinated. They should be offered catch-up vaccination. If older than age 15, they should receive 3 doses of the vaccine, with the second dose administered 1 to 2 months after the first dose and the third dose administered 6 months after the first dose.1
A third target group is individuals who are aged 27 to 45 years and who, in their own opinion or in the opinion of their physician, are at new or increased risk for HPV infection. These individuals should receive the 3-dose vaccine series as outlined above.1
Patients in any age range who are immunocompromised, for example, due to HIV infection, should receive the 3-dose series.1
The approximate retail cost of a single 0.5-mL intramuscular dose of the 9-valent vaccine is $240 (www.goodrx.com).
Vaccine adverse effects
The most common reactions to the HPV vaccine are inflammation at the site of injection, fatigue, headache, fever, gastrointestinal upset, vertigo, cough, and oropharyngeal discomfort. The most serious reaction—which fortunately is very rare—is anaphylaxis.1
Contraindications to the vaccine
The HPV vaccine should not be used in any patient who is hypersensitive to any component of the vaccine, including yeast. It should not be given to a patient who is moderately or severely ill at the time of the scheduled administration. Because of an abundance of caution, the manufacturer also recommends that the vaccine not be given to pregnant women even though the agent does not contain live virus.1
Of note, a study by Scheller and colleagues was very reassuring about the lack of adverse effects of HPV vaccine administration in pregnancy.8 The authors evaluated a large cohort of pregnant women in Demark and found that exposure to the vaccine was not associated with an increase in the frequency of major birth defects, spontaneous abortion, preterm delivery, low birthweight, fetal growth restriction, or stillbirth.8
Barriers to vaccination
One important barrier to HPV vaccination is patient apprehension that the vaccine may cause genital tract or oropharyngeal cancer. The patient should be reassured that the vaccine does not contain infectious viral particles and does not transmit infection. Rather, it builds robust immunity to infection.
Another important barrier is the misconception that the vaccine will promote sexual promiscuity in preteenagers and teenagers. Absolutely no evidence supports this belief. Multiple studies have demonstrated that teenagers do not engage in more high-risk sexual behavior following vaccination.
A specific barrier related to vaccination of young boys is the philosophical viewpoint that, “Why should my young male child be vaccinated to protect against a disease (specifically cervical cancer) that occurs only in girls and women?” The appropriate answer to this question is that the vaccine also protects against penile cancer, anal cancer, oropharyngeal cancer, and genital warts. While penile and anal cancers are rare, the other 2 conditions are not. In fact, oropharyngeal cancer is significantly more common in males than females.
A final important barrier to HPV vaccination is cost. The new evidence that demonstrated the effectiveness of a 2-dose vaccine series, and even single-dose vaccination, is of great importance in minimizing cost of the HPV vaccine series, in the absence of full reimbursement by public and private insurance agencies.
Continue to: Creating an effective vaccination program...
Creating an effective vaccination program
The following commonsense guidelines, which we have implemented at our medical center, should be helpful in organizing an effective HPV vaccination program for your office or department4,9,10:
- One clinician in the department or practice should be designated the “vaccination champion.” This individual should provide colleagues with periodic updates, emphasizing the importance of the HPV vaccine and other vaccines, such as Tdap (tetanus, diphtheria, pertussis), influenza, COVID, pneumococcal, hepatitis B, herpes zoster (shingles), and RSV (respiratory syncytial virus).
- One staff member in the practice or department should be designated as the go-to person for all logistical matters related to vaccines. This individual should be responsible for estimating usage, ordering vaccines, and storing them properly. He or she also should be knowledgeable about the cost of the vaccines and insurance reimbursement for the vaccines.
- Signs and educational materials should be posted in strategic locations in the office, advising patients of the importance of timely vaccination for themselves and their adolescent children.
- At every encounter, patients should be encouraged to receive the HPV vaccine series if they are in the appropriate age range and social situation for vaccination. They should not be required to have HPV testing before vaccine administration.
- Key leaders in the department or practice should lobby effectively with their pediatrician colleagues and with public and private insurance companies to encourage timely administration and proper coverage of this important immunization.
Other measures to reduce the risk of HPV-mediated malignancies
Practitioners should advise their patients to:
- Be circumspect in selection of sexual partners.
- Use male or female condoms when engaging in vaginal, anal, and/or oral sex with multiple partners, particularly those who may have genital or oral condylomas.
- Have regular Pap tests, every 3 to 5 years, depending upon age. More frequent testing may be indicated if there is a history of previous abnormal testing.
- Seek prompt medical or surgical treatment for genital or oral condylomas.
CASE Resolved with HPV vaccination
This patient is an excellent candidate for catch-up vaccination. She should receive the first dose of the 9-valent HPV vaccine at the time of her postpartum appointment. The second dose should be administered 1 to 2 months later. The third dose should be administered 6 months after the first dose. She also should have a Pap test, either cytology alone or cytology plus HPV screening. If the latter test is chosen and is reassuring, she will not need retesting for 5 years. If the former test is chosen, she should have a repeat test in 3 years. ●

- The overwhelming majority of precancerous lesions and overt malignancies of the genital tract and oropharynx are caused by oncogenic strains of HPV.
- Most of these cancers could be prevented if patients were vaccinated with the 9-valent HPV vaccine.
- The HPV vaccine should be offered to all children beginning at age 11 and to selected high-risk children at age 9. For children aged 14 years and younger, 2 doses of the vaccine are sufficient to induce a robust immune response. The second dose should be administered 6 to 12 months after the first dose.
- Individuals in the age range 13 to 26 years should be offered catch-up vaccination if they have not been previously vaccinated.
- Persons in the age range 27 to 45 years also should be offered vaccination if they have developed a new high-risk profile.
- Persons older than age 15, or those of any age with immunocompromising conditions, should receive 3 doses of the vaccine. The second dose should be administered 1 to 2 months after the first dose, and the third dose should be given 6 months after the first dose.
- The vaccine does not prevent the progression of preexisting infection or clear an infection that is already present at the time of vaccination.
- As a general rule, the vaccine should be deferred during pregnancy, although no adverse effects have been documented when the vaccine has been administered to pregnant women.
- Markowitz LE, Unger ER. Human papilloma virus vaccination. N Engl J Med. 2023;388:1790-1798.
- Schiller JT, Castellsague X, Garland SM. A review of clinical trials of human papillomavirus prophylactic vaccines. Vaccine. 2012;30(suppl 5): F123-F138.
- Lei J, Ploner A, Elfstrom KM, et al. HPV vaccination and the risk of invasive cervical cancer. N Engl J Med. 2020;383: 1340-1348.
- ACOG Committee Opinion Summary No. 809. Human papillomavirus vaccination. Obstet Gynecol. 2020;136:435-436.
- Barbieri RL. 9vHPV vaccine: prevention of oropharyngeal cancer. OBG Manag. 2020;32:9, 14-15.
- Iversen OE, Miranda MJ, Ulied A, et al. Immunogenicity of the 9-valent HPV vaccine using 2-dose regimens in girls and boys vs a 3-dose regimen in women. JAMA. 2016;316:2411-2421.
- Watson-Jones D, Changalucha J, Whitworth H, et al. Immunogenicity and safety of one-dose human papillomavirus vaccine compared with two or three doses in Tanzanian girls (DoRIS): an open-label, randomised noninferiority trial. Lancet Glob Health. 2022;10:e1473-e1484.
- Scheller NM, Pasternak B, Molgaard-Nielsen D, et al. Quadrivalent HPV vaccination and the risk of adverse pregnancy outcomes. N Engl J Med. 2017;376:1223-1233.
- ACOG Committee Opinion Summary No. 641. Human papillomavirus vaccination. Obstet Gynecol. 2015;126:693.
- Boitano TKL, Ketch PW, Scarinci IC, et al. An update on human papillomavirus vaccination in the United States. Obstet Gynecol. 2023;141:324-330.
CASE Sexually active woman asks about the HPV vaccine
A 26-year-old woman delivered her first child 4 weeks ago. She has had 3 lifetime sexual partners and is now in a mutually faithful monogamous relationship with her partner. She has no known history of sexually transmissible infections. She received only one Pap test 3 years ago, and the cytology showed no abnormal cells. This cervical specimen was not tested for human papillomavirus (HPV) DNA. At the time of her postpartum appointment, she inquires whether she is a candidate for the HPV vaccine.
What should be your response?
Genital HPV infection is the most common sexually transmissible infection in the United States. This virus is the cause of multiple genital malignancies, including cancers of the vagina, vulva, penis, anus, and cervix. The organism is also now the major cause of oropharyngeal cancer.
Of the more than 200 different HPV types that have been identified, 12 have been defined as oncogenic (high risk), and 8 to 12 types have been defined as possibly or probably oncogenic. The HPV strain with the highest risk of progression to cancer is HPV 16. The strains HPV 16 and 18 are responsible for approximately 70% of cases of cervical cancer. Each year in the United States, approximately 11,500 new cases of invasive cervical cancer occur. Unfortunately, this malignancy is responsible for about 4,000 deaths annually. Worldwide, HPV causes approximately 690,000 cancers each year.1
To a large extent, most cases of HPV infection would be preventable if patients were to take advantage of the remarkably effective HPV vaccine that is now available. However, acceptance of the vaccine has been disappointing. In 2020, only about half of adolescents, age 13 to 15, had received the appropriate number of vaccine doses.1
As ObGyn physicians, we can take several measures, in concert with our pediatrician colleagues, to improve HPV vaccination rates. In this article, I review the development of the HPV vaccine and describe the components, indications, dosing schedules, contraindications, adverse effects, and cost of the vaccine.
HPV vaccine development and expansion
The first HPV vaccine introduced in the United States was the recombinant quadrivalent vaccine (Gardasil; Merck); it was approved by the US Food and Drug Administration (FDA) in 2006. This vaccine is composed of viral-like particles unique to HPV 16 and 18 (the 2 most common causes of cervical, penile, anal, and oropharyngeal cancer) and HPV 6 and 11 (the 2 most common causes of genital warts). The formulation is prepared in baker’s yeast, and it elicits a robust production of neutralizing antibodies.2
In 2009, the FDA approved the bivalent vaccine (Cervarix; GlaxoSmithKline Biologicals). This vaccine contains viral-like particles unique to HPV 16 and 18, and it also induces a robust immune response. The vaccine is prepared in insect viral vectors.2
Both the quadrivalent and bivalent vaccines are no longer available in the United States. The only HPV vaccine currently marketed is the recombinant 9-valent vaccine (Gardasil 9; Merck), which was approved by the FDA in 2014. This newer vaccine targets the original 4 viral HPV strains in the quadrivalent vaccine (16, 18, 6, 11) plus 5 additional oncogenic strains: 31, 33, 45, 52, 58.2-4 The HPV strains targeted by this vaccine are responsible for approximately 90% of all cancers caused by HPV.
The 9-valent HPV vaccine, like the other 2, is highly effective in preventing cancers of the cervix, vagina, vulva, anus, penis; oropharyngeal cancers; and precancerous lesions such as genital warts.2-5 It will not, however, prevent the progression of preexisting infection or clear an infection that is already present at the time of vaccination.1
Although the original protocol for administration of the vaccine provided for 3 doses, recent studies indicate that 2 doses may be as effective as 3 in eliciting a favorable antibody response.6 There also is evidence that even a single dose of the vaccine can elicit a protective immune response.7 This encouraging finding is particularly important to public health officials responsible for developing HPV vaccination programs in low- and middle-resource countries.

Continue to: Target groups for the HPV vaccine...
Target groups for the HPV vaccine
The primary target group for the HPV vaccine is girls and boys who are aged 11 to 12 years. The key strategy is to immunize these individuals before they become sexually active. The vaccine also should be offered to children who are aged 9 to 10 years of age if they are judged to be at unusual risk, such as because of concern about sexual molestation. Children in these 2 age groups should receive 2 doses of the vaccine, with the second dose administered 6 to 12 months after the first dose.
The second target group for vaccination is individuals who are aged 13 to 26 years who have never been vaccinated. They should be offered catch-up vaccination. If older than age 15, they should receive 3 doses of the vaccine, with the second dose administered 1 to 2 months after the first dose and the third dose administered 6 months after the first dose.1
A third target group is individuals who are aged 27 to 45 years and who, in their own opinion or in the opinion of their physician, are at new or increased risk for HPV infection. These individuals should receive the 3-dose vaccine series as outlined above.1
Patients in any age range who are immunocompromised, for example, due to HIV infection, should receive the 3-dose series.1
The approximate retail cost of a single 0.5-mL intramuscular dose of the 9-valent vaccine is $240 (www.goodrx.com).
Vaccine adverse effects
The most common reactions to the HPV vaccine are inflammation at the site of injection, fatigue, headache, fever, gastrointestinal upset, vertigo, cough, and oropharyngeal discomfort. The most serious reaction—which fortunately is very rare—is anaphylaxis.1
Contraindications to the vaccine
The HPV vaccine should not be used in any patient who is hypersensitive to any component of the vaccine, including yeast. It should not be given to a patient who is moderately or severely ill at the time of the scheduled administration. Because of an abundance of caution, the manufacturer also recommends that the vaccine not be given to pregnant women even though the agent does not contain live virus.1
Of note, a study by Scheller and colleagues was very reassuring about the lack of adverse effects of HPV vaccine administration in pregnancy.8 The authors evaluated a large cohort of pregnant women in Demark and found that exposure to the vaccine was not associated with an increase in the frequency of major birth defects, spontaneous abortion, preterm delivery, low birthweight, fetal growth restriction, or stillbirth.8
Barriers to vaccination
One important barrier to HPV vaccination is patient apprehension that the vaccine may cause genital tract or oropharyngeal cancer. The patient should be reassured that the vaccine does not contain infectious viral particles and does not transmit infection. Rather, it builds robust immunity to infection.
Another important barrier is the misconception that the vaccine will promote sexual promiscuity in preteenagers and teenagers. Absolutely no evidence supports this belief. Multiple studies have demonstrated that teenagers do not engage in more high-risk sexual behavior following vaccination.
A specific barrier related to vaccination of young boys is the philosophical viewpoint that, “Why should my young male child be vaccinated to protect against a disease (specifically cervical cancer) that occurs only in girls and women?” The appropriate answer to this question is that the vaccine also protects against penile cancer, anal cancer, oropharyngeal cancer, and genital warts. While penile and anal cancers are rare, the other 2 conditions are not. In fact, oropharyngeal cancer is significantly more common in males than females.
A final important barrier to HPV vaccination is cost. The new evidence that demonstrated the effectiveness of a 2-dose vaccine series, and even single-dose vaccination, is of great importance in minimizing cost of the HPV vaccine series, in the absence of full reimbursement by public and private insurance agencies.
Continue to: Creating an effective vaccination program...
Creating an effective vaccination program
The following commonsense guidelines, which we have implemented at our medical center, should be helpful in organizing an effective HPV vaccination program for your office or department4,9,10:
- One clinician in the department or practice should be designated the “vaccination champion.” This individual should provide colleagues with periodic updates, emphasizing the importance of the HPV vaccine and other vaccines, such as Tdap (tetanus, diphtheria, pertussis), influenza, COVID, pneumococcal, hepatitis B, herpes zoster (shingles), and RSV (respiratory syncytial virus).
- One staff member in the practice or department should be designated as the go-to person for all logistical matters related to vaccines. This individual should be responsible for estimating usage, ordering vaccines, and storing them properly. He or she also should be knowledgeable about the cost of the vaccines and insurance reimbursement for the vaccines.
- Signs and educational materials should be posted in strategic locations in the office, advising patients of the importance of timely vaccination for themselves and their adolescent children.
- At every encounter, patients should be encouraged to receive the HPV vaccine series if they are in the appropriate age range and social situation for vaccination. They should not be required to have HPV testing before vaccine administration.
- Key leaders in the department or practice should lobby effectively with their pediatrician colleagues and with public and private insurance companies to encourage timely administration and proper coverage of this important immunization.
Other measures to reduce the risk of HPV-mediated malignancies
Practitioners should advise their patients to:
- Be circumspect in selection of sexual partners.
- Use male or female condoms when engaging in vaginal, anal, and/or oral sex with multiple partners, particularly those who may have genital or oral condylomas.
- Have regular Pap tests, every 3 to 5 years, depending upon age. More frequent testing may be indicated if there is a history of previous abnormal testing.
- Seek prompt medical or surgical treatment for genital or oral condylomas.
CASE Resolved with HPV vaccination
This patient is an excellent candidate for catch-up vaccination. She should receive the first dose of the 9-valent HPV vaccine at the time of her postpartum appointment. The second dose should be administered 1 to 2 months later. The third dose should be administered 6 months after the first dose. She also should have a Pap test, either cytology alone or cytology plus HPV screening. If the latter test is chosen and is reassuring, she will not need retesting for 5 years. If the former test is chosen, she should have a repeat test in 3 years. ●

- The overwhelming majority of precancerous lesions and overt malignancies of the genital tract and oropharynx are caused by oncogenic strains of HPV.
- Most of these cancers could be prevented if patients were vaccinated with the 9-valent HPV vaccine.
- The HPV vaccine should be offered to all children beginning at age 11 and to selected high-risk children at age 9. For children aged 14 years and younger, 2 doses of the vaccine are sufficient to induce a robust immune response. The second dose should be administered 6 to 12 months after the first dose.
- Individuals in the age range 13 to 26 years should be offered catch-up vaccination if they have not been previously vaccinated.
- Persons in the age range 27 to 45 years also should be offered vaccination if they have developed a new high-risk profile.
- Persons older than age 15, or those of any age with immunocompromising conditions, should receive 3 doses of the vaccine. The second dose should be administered 1 to 2 months after the first dose, and the third dose should be given 6 months after the first dose.
- The vaccine does not prevent the progression of preexisting infection or clear an infection that is already present at the time of vaccination.
- As a general rule, the vaccine should be deferred during pregnancy, although no adverse effects have been documented when the vaccine has been administered to pregnant women.
CASE Sexually active woman asks about the HPV vaccine
A 26-year-old woman delivered her first child 4 weeks ago. She has had 3 lifetime sexual partners and is now in a mutually faithful monogamous relationship with her partner. She has no known history of sexually transmissible infections. She received only one Pap test 3 years ago, and the cytology showed no abnormal cells. This cervical specimen was not tested for human papillomavirus (HPV) DNA. At the time of her postpartum appointment, she inquires whether she is a candidate for the HPV vaccine.
What should be your response?
Genital HPV infection is the most common sexually transmissible infection in the United States. This virus is the cause of multiple genital malignancies, including cancers of the vagina, vulva, penis, anus, and cervix. The organism is also now the major cause of oropharyngeal cancer.
Of the more than 200 different HPV types that have been identified, 12 have been defined as oncogenic (high risk), and 8 to 12 types have been defined as possibly or probably oncogenic. The HPV strain with the highest risk of progression to cancer is HPV 16. The strains HPV 16 and 18 are responsible for approximately 70% of cases of cervical cancer. Each year in the United States, approximately 11,500 new cases of invasive cervical cancer occur. Unfortunately, this malignancy is responsible for about 4,000 deaths annually. Worldwide, HPV causes approximately 690,000 cancers each year.1
To a large extent, most cases of HPV infection would be preventable if patients were to take advantage of the remarkably effective HPV vaccine that is now available. However, acceptance of the vaccine has been disappointing. In 2020, only about half of adolescents, age 13 to 15, had received the appropriate number of vaccine doses.1
As ObGyn physicians, we can take several measures, in concert with our pediatrician colleagues, to improve HPV vaccination rates. In this article, I review the development of the HPV vaccine and describe the components, indications, dosing schedules, contraindications, adverse effects, and cost of the vaccine.
HPV vaccine development and expansion
The first HPV vaccine introduced in the United States was the recombinant quadrivalent vaccine (Gardasil; Merck); it was approved by the US Food and Drug Administration (FDA) in 2006. This vaccine is composed of viral-like particles unique to HPV 16 and 18 (the 2 most common causes of cervical, penile, anal, and oropharyngeal cancer) and HPV 6 and 11 (the 2 most common causes of genital warts). The formulation is prepared in baker’s yeast, and it elicits a robust production of neutralizing antibodies.2
In 2009, the FDA approved the bivalent vaccine (Cervarix; GlaxoSmithKline Biologicals). This vaccine contains viral-like particles unique to HPV 16 and 18, and it also induces a robust immune response. The vaccine is prepared in insect viral vectors.2
Both the quadrivalent and bivalent vaccines are no longer available in the United States. The only HPV vaccine currently marketed is the recombinant 9-valent vaccine (Gardasil 9; Merck), which was approved by the FDA in 2014. This newer vaccine targets the original 4 viral HPV strains in the quadrivalent vaccine (16, 18, 6, 11) plus 5 additional oncogenic strains: 31, 33, 45, 52, 58.2-4 The HPV strains targeted by this vaccine are responsible for approximately 90% of all cancers caused by HPV.
The 9-valent HPV vaccine, like the other 2, is highly effective in preventing cancers of the cervix, vagina, vulva, anus, penis; oropharyngeal cancers; and precancerous lesions such as genital warts.2-5 It will not, however, prevent the progression of preexisting infection or clear an infection that is already present at the time of vaccination.1
Although the original protocol for administration of the vaccine provided for 3 doses, recent studies indicate that 2 doses may be as effective as 3 in eliciting a favorable antibody response.6 There also is evidence that even a single dose of the vaccine can elicit a protective immune response.7 This encouraging finding is particularly important to public health officials responsible for developing HPV vaccination programs in low- and middle-resource countries.

Continue to: Target groups for the HPV vaccine...
Target groups for the HPV vaccine
The primary target group for the HPV vaccine is girls and boys who are aged 11 to 12 years. The key strategy is to immunize these individuals before they become sexually active. The vaccine also should be offered to children who are aged 9 to 10 years of age if they are judged to be at unusual risk, such as because of concern about sexual molestation. Children in these 2 age groups should receive 2 doses of the vaccine, with the second dose administered 6 to 12 months after the first dose.
The second target group for vaccination is individuals who are aged 13 to 26 years who have never been vaccinated. They should be offered catch-up vaccination. If older than age 15, they should receive 3 doses of the vaccine, with the second dose administered 1 to 2 months after the first dose and the third dose administered 6 months after the first dose.1
A third target group is individuals who are aged 27 to 45 years and who, in their own opinion or in the opinion of their physician, are at new or increased risk for HPV infection. These individuals should receive the 3-dose vaccine series as outlined above.1
Patients in any age range who are immunocompromised, for example, due to HIV infection, should receive the 3-dose series.1
The approximate retail cost of a single 0.5-mL intramuscular dose of the 9-valent vaccine is $240 (www.goodrx.com).
Vaccine adverse effects
The most common reactions to the HPV vaccine are inflammation at the site of injection, fatigue, headache, fever, gastrointestinal upset, vertigo, cough, and oropharyngeal discomfort. The most serious reaction—which fortunately is very rare—is anaphylaxis.1
Contraindications to the vaccine
The HPV vaccine should not be used in any patient who is hypersensitive to any component of the vaccine, including yeast. It should not be given to a patient who is moderately or severely ill at the time of the scheduled administration. Because of an abundance of caution, the manufacturer also recommends that the vaccine not be given to pregnant women even though the agent does not contain live virus.1
Of note, a study by Scheller and colleagues was very reassuring about the lack of adverse effects of HPV vaccine administration in pregnancy.8 The authors evaluated a large cohort of pregnant women in Demark and found that exposure to the vaccine was not associated with an increase in the frequency of major birth defects, spontaneous abortion, preterm delivery, low birthweight, fetal growth restriction, or stillbirth.8
Barriers to vaccination
One important barrier to HPV vaccination is patient apprehension that the vaccine may cause genital tract or oropharyngeal cancer. The patient should be reassured that the vaccine does not contain infectious viral particles and does not transmit infection. Rather, it builds robust immunity to infection.
Another important barrier is the misconception that the vaccine will promote sexual promiscuity in preteenagers and teenagers. Absolutely no evidence supports this belief. Multiple studies have demonstrated that teenagers do not engage in more high-risk sexual behavior following vaccination.
A specific barrier related to vaccination of young boys is the philosophical viewpoint that, “Why should my young male child be vaccinated to protect against a disease (specifically cervical cancer) that occurs only in girls and women?” The appropriate answer to this question is that the vaccine also protects against penile cancer, anal cancer, oropharyngeal cancer, and genital warts. While penile and anal cancers are rare, the other 2 conditions are not. In fact, oropharyngeal cancer is significantly more common in males than females.
A final important barrier to HPV vaccination is cost. The new evidence that demonstrated the effectiveness of a 2-dose vaccine series, and even single-dose vaccination, is of great importance in minimizing cost of the HPV vaccine series, in the absence of full reimbursement by public and private insurance agencies.
Continue to: Creating an effective vaccination program...
Creating an effective vaccination program
The following commonsense guidelines, which we have implemented at our medical center, should be helpful in organizing an effective HPV vaccination program for your office or department4,9,10:
- One clinician in the department or practice should be designated the “vaccination champion.” This individual should provide colleagues with periodic updates, emphasizing the importance of the HPV vaccine and other vaccines, such as Tdap (tetanus, diphtheria, pertussis), influenza, COVID, pneumococcal, hepatitis B, herpes zoster (shingles), and RSV (respiratory syncytial virus).
- One staff member in the practice or department should be designated as the go-to person for all logistical matters related to vaccines. This individual should be responsible for estimating usage, ordering vaccines, and storing them properly. He or she also should be knowledgeable about the cost of the vaccines and insurance reimbursement for the vaccines.
- Signs and educational materials should be posted in strategic locations in the office, advising patients of the importance of timely vaccination for themselves and their adolescent children.
- At every encounter, patients should be encouraged to receive the HPV vaccine series if they are in the appropriate age range and social situation for vaccination. They should not be required to have HPV testing before vaccine administration.
- Key leaders in the department or practice should lobby effectively with their pediatrician colleagues and with public and private insurance companies to encourage timely administration and proper coverage of this important immunization.
Other measures to reduce the risk of HPV-mediated malignancies
Practitioners should advise their patients to:
- Be circumspect in selection of sexual partners.
- Use male or female condoms when engaging in vaginal, anal, and/or oral sex with multiple partners, particularly those who may have genital or oral condylomas.
- Have regular Pap tests, every 3 to 5 years, depending upon age. More frequent testing may be indicated if there is a history of previous abnormal testing.
- Seek prompt medical or surgical treatment for genital or oral condylomas.
CASE Resolved with HPV vaccination
This patient is an excellent candidate for catch-up vaccination. She should receive the first dose of the 9-valent HPV vaccine at the time of her postpartum appointment. The second dose should be administered 1 to 2 months later. The third dose should be administered 6 months after the first dose. She also should have a Pap test, either cytology alone or cytology plus HPV screening. If the latter test is chosen and is reassuring, she will not need retesting for 5 years. If the former test is chosen, she should have a repeat test in 3 years. ●

- The overwhelming majority of precancerous lesions and overt malignancies of the genital tract and oropharynx are caused by oncogenic strains of HPV.
- Most of these cancers could be prevented if patients were vaccinated with the 9-valent HPV vaccine.
- The HPV vaccine should be offered to all children beginning at age 11 and to selected high-risk children at age 9. For children aged 14 years and younger, 2 doses of the vaccine are sufficient to induce a robust immune response. The second dose should be administered 6 to 12 months after the first dose.
- Individuals in the age range 13 to 26 years should be offered catch-up vaccination if they have not been previously vaccinated.
- Persons in the age range 27 to 45 years also should be offered vaccination if they have developed a new high-risk profile.
- Persons older than age 15, or those of any age with immunocompromising conditions, should receive 3 doses of the vaccine. The second dose should be administered 1 to 2 months after the first dose, and the third dose should be given 6 months after the first dose.
- The vaccine does not prevent the progression of preexisting infection or clear an infection that is already present at the time of vaccination.
- As a general rule, the vaccine should be deferred during pregnancy, although no adverse effects have been documented when the vaccine has been administered to pregnant women.
- Markowitz LE, Unger ER. Human papilloma virus vaccination. N Engl J Med. 2023;388:1790-1798.
- Schiller JT, Castellsague X, Garland SM. A review of clinical trials of human papillomavirus prophylactic vaccines. Vaccine. 2012;30(suppl 5): F123-F138.
- Lei J, Ploner A, Elfstrom KM, et al. HPV vaccination and the risk of invasive cervical cancer. N Engl J Med. 2020;383: 1340-1348.
- ACOG Committee Opinion Summary No. 809. Human papillomavirus vaccination. Obstet Gynecol. 2020;136:435-436.
- Barbieri RL. 9vHPV vaccine: prevention of oropharyngeal cancer. OBG Manag. 2020;32:9, 14-15.
- Iversen OE, Miranda MJ, Ulied A, et al. Immunogenicity of the 9-valent HPV vaccine using 2-dose regimens in girls and boys vs a 3-dose regimen in women. JAMA. 2016;316:2411-2421.
- Watson-Jones D, Changalucha J, Whitworth H, et al. Immunogenicity and safety of one-dose human papillomavirus vaccine compared with two or three doses in Tanzanian girls (DoRIS): an open-label, randomised noninferiority trial. Lancet Glob Health. 2022;10:e1473-e1484.
- Scheller NM, Pasternak B, Molgaard-Nielsen D, et al. Quadrivalent HPV vaccination and the risk of adverse pregnancy outcomes. N Engl J Med. 2017;376:1223-1233.
- ACOG Committee Opinion Summary No. 641. Human papillomavirus vaccination. Obstet Gynecol. 2015;126:693.
- Boitano TKL, Ketch PW, Scarinci IC, et al. An update on human papillomavirus vaccination in the United States. Obstet Gynecol. 2023;141:324-330.
- Markowitz LE, Unger ER. Human papilloma virus vaccination. N Engl J Med. 2023;388:1790-1798.
- Schiller JT, Castellsague X, Garland SM. A review of clinical trials of human papillomavirus prophylactic vaccines. Vaccine. 2012;30(suppl 5): F123-F138.
- Lei J, Ploner A, Elfstrom KM, et al. HPV vaccination and the risk of invasive cervical cancer. N Engl J Med. 2020;383: 1340-1348.
- ACOG Committee Opinion Summary No. 809. Human papillomavirus vaccination. Obstet Gynecol. 2020;136:435-436.
- Barbieri RL. 9vHPV vaccine: prevention of oropharyngeal cancer. OBG Manag. 2020;32:9, 14-15.
- Iversen OE, Miranda MJ, Ulied A, et al. Immunogenicity of the 9-valent HPV vaccine using 2-dose regimens in girls and boys vs a 3-dose regimen in women. JAMA. 2016;316:2411-2421.
- Watson-Jones D, Changalucha J, Whitworth H, et al. Immunogenicity and safety of one-dose human papillomavirus vaccine compared with two or three doses in Tanzanian girls (DoRIS): an open-label, randomised noninferiority trial. Lancet Glob Health. 2022;10:e1473-e1484.
- Scheller NM, Pasternak B, Molgaard-Nielsen D, et al. Quadrivalent HPV vaccination and the risk of adverse pregnancy outcomes. N Engl J Med. 2017;376:1223-1233.
- ACOG Committee Opinion Summary No. 641. Human papillomavirus vaccination. Obstet Gynecol. 2015;126:693.
- Boitano TKL, Ketch PW, Scarinci IC, et al. An update on human papillomavirus vaccination in the United States. Obstet Gynecol. 2023;141:324-330.
‘Vaginal dryness’ can be fatal. No, really.
This transcript has been edited for clarity.
What do you mean, Dr. Rubin? How is vaginal dryness killing women? We minimize the term vaginal dryness. When women come to our offices and complain of a little vaginal dryness – or they don’t even come to our office to complain of it because the doctor can’t be bothered with a little vaginal dryness — what they don’t understand is that this “little vaginal dryness” is really something called genitourinary syndrome of menopause (GSM). They don’t know that because they’ve never heard of it, and you may have never heard of it either. In 2014, we changed the terms vaginal dryness and vulvovaginal atrophy or atrophic vaginitis to GSM to make it short and simple.
GSM – what does it mean? It’s not just a little vaginal dryness. It turns out that all of the genital and urinary symptoms from menopause just get worse over time. The bladder, the urethra, and the vagina have lots of hormone receptors, including estrogen and testosterone. When the body no longer makes those hormones, the system doesn’t work very well, and genital and urinary symptoms occur that just get worse over time without treatment. Unlike hot flashes, which tend to go away, GSM does not.
What are the symptoms of GSM? Some are sexual: a little vaginal dryness, pain with sex, and worsening orgasm. But there are also genital and urinary symptoms that get worse: itching, burning irritation, rawness, an awareness of their genitals that the patient has never had before. And as a urologist, we see frequency, urgency, and leakage.
The thing that kills women is recurrent urinary tract infections (UTIs). Did you know that UTIs account for 7 million visits and hospitalizations annually and 25% of all infections in older people? In fact, apparently one-third of the total Medicare expenditure is around UTIs. Not preventing UTIs is costing our health care system an enormous amount of money and resources.
Did you know we’ve had safe and effective treatment options for GSM since the 1970s? Vaginal hormones have existed since the 1970s, but we’re using them only for pain with sex and not for GSM. In fact, data show that by using vaginal hormones, we can prevent UTIs by more than 50%. We can save lives using safe, effective, local, low-dose vaginal hormone strategies. And they are safe and effective for all of our patients in pre- and post menopause.
There are five different treatment options: vaginal estrogen inserts, vaginal estrogen creams, vaginal dehydroepiandrosterone (DHEA), low-dose vaginal estrogen rings, and an oral pill option called ospemifene (Osphena). All are used to treat GSM and will only work if your patient actually uses them and continues to use them.
These treatments are safe. They are effective. They do not increase the level of systemic hormones in the bloodstream. I have many patients with breast cancer who use these products as well. The only patients you may want to talk to your oncology colleagues about is women on active aromatase inhibitors.
We have to understand that UTIs kill people and having GSM is debilitating, often requiring pain medication because it can hurt to sit or to wear pads and our patients’ quality of life is severely affected. So please consider learning how to treat GSM. It turns out you don’t have to do exams. You don’t have to do follow-up. You can give these therapies, and women can use them for life.
Now, if your patient has vaginal bleeding, of course they need to see their gynecologist. But this is something every primary care doctor can and should do. As a urologist, we prescribe a lot of tamsulosin (Flomax) for our male patients to help with urination. Vaginal estrogen or DHEA is basically like Flomax for women, but it prevents UTIs and actually works like sildenafil (Viagra) because it can help orgasm and reduce pain with sex.
You have access to affordable, safe, effective treatment options to treat GSM. So check them out and hopefully change the world.
Dr. Rubin is an assistant clinical professor in the department of urology at Georgetown University, Washington. She reported conflicts of interest with Sprout, Maternal Medical, Absorption Pharmaceuticals, GlaxoSmithKline, and Endo.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
What do you mean, Dr. Rubin? How is vaginal dryness killing women? We minimize the term vaginal dryness. When women come to our offices and complain of a little vaginal dryness – or they don’t even come to our office to complain of it because the doctor can’t be bothered with a little vaginal dryness — what they don’t understand is that this “little vaginal dryness” is really something called genitourinary syndrome of menopause (GSM). They don’t know that because they’ve never heard of it, and you may have never heard of it either. In 2014, we changed the terms vaginal dryness and vulvovaginal atrophy or atrophic vaginitis to GSM to make it short and simple.
GSM – what does it mean? It’s not just a little vaginal dryness. It turns out that all of the genital and urinary symptoms from menopause just get worse over time. The bladder, the urethra, and the vagina have lots of hormone receptors, including estrogen and testosterone. When the body no longer makes those hormones, the system doesn’t work very well, and genital and urinary symptoms occur that just get worse over time without treatment. Unlike hot flashes, which tend to go away, GSM does not.
What are the symptoms of GSM? Some are sexual: a little vaginal dryness, pain with sex, and worsening orgasm. But there are also genital and urinary symptoms that get worse: itching, burning irritation, rawness, an awareness of their genitals that the patient has never had before. And as a urologist, we see frequency, urgency, and leakage.
The thing that kills women is recurrent urinary tract infections (UTIs). Did you know that UTIs account for 7 million visits and hospitalizations annually and 25% of all infections in older people? In fact, apparently one-third of the total Medicare expenditure is around UTIs. Not preventing UTIs is costing our health care system an enormous amount of money and resources.
Did you know we’ve had safe and effective treatment options for GSM since the 1970s? Vaginal hormones have existed since the 1970s, but we’re using them only for pain with sex and not for GSM. In fact, data show that by using vaginal hormones, we can prevent UTIs by more than 50%. We can save lives using safe, effective, local, low-dose vaginal hormone strategies. And they are safe and effective for all of our patients in pre- and post menopause.
There are five different treatment options: vaginal estrogen inserts, vaginal estrogen creams, vaginal dehydroepiandrosterone (DHEA), low-dose vaginal estrogen rings, and an oral pill option called ospemifene (Osphena). All are used to treat GSM and will only work if your patient actually uses them and continues to use them.
These treatments are safe. They are effective. They do not increase the level of systemic hormones in the bloodstream. I have many patients with breast cancer who use these products as well. The only patients you may want to talk to your oncology colleagues about is women on active aromatase inhibitors.
We have to understand that UTIs kill people and having GSM is debilitating, often requiring pain medication because it can hurt to sit or to wear pads and our patients’ quality of life is severely affected. So please consider learning how to treat GSM. It turns out you don’t have to do exams. You don’t have to do follow-up. You can give these therapies, and women can use them for life.
Now, if your patient has vaginal bleeding, of course they need to see their gynecologist. But this is something every primary care doctor can and should do. As a urologist, we prescribe a lot of tamsulosin (Flomax) for our male patients to help with urination. Vaginal estrogen or DHEA is basically like Flomax for women, but it prevents UTIs and actually works like sildenafil (Viagra) because it can help orgasm and reduce pain with sex.
You have access to affordable, safe, effective treatment options to treat GSM. So check them out and hopefully change the world.
Dr. Rubin is an assistant clinical professor in the department of urology at Georgetown University, Washington. She reported conflicts of interest with Sprout, Maternal Medical, Absorption Pharmaceuticals, GlaxoSmithKline, and Endo.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
What do you mean, Dr. Rubin? How is vaginal dryness killing women? We minimize the term vaginal dryness. When women come to our offices and complain of a little vaginal dryness – or they don’t even come to our office to complain of it because the doctor can’t be bothered with a little vaginal dryness — what they don’t understand is that this “little vaginal dryness” is really something called genitourinary syndrome of menopause (GSM). They don’t know that because they’ve never heard of it, and you may have never heard of it either. In 2014, we changed the terms vaginal dryness and vulvovaginal atrophy or atrophic vaginitis to GSM to make it short and simple.
GSM – what does it mean? It’s not just a little vaginal dryness. It turns out that all of the genital and urinary symptoms from menopause just get worse over time. The bladder, the urethra, and the vagina have lots of hormone receptors, including estrogen and testosterone. When the body no longer makes those hormones, the system doesn’t work very well, and genital and urinary symptoms occur that just get worse over time without treatment. Unlike hot flashes, which tend to go away, GSM does not.
What are the symptoms of GSM? Some are sexual: a little vaginal dryness, pain with sex, and worsening orgasm. But there are also genital and urinary symptoms that get worse: itching, burning irritation, rawness, an awareness of their genitals that the patient has never had before. And as a urologist, we see frequency, urgency, and leakage.
The thing that kills women is recurrent urinary tract infections (UTIs). Did you know that UTIs account for 7 million visits and hospitalizations annually and 25% of all infections in older people? In fact, apparently one-third of the total Medicare expenditure is around UTIs. Not preventing UTIs is costing our health care system an enormous amount of money and resources.
Did you know we’ve had safe and effective treatment options for GSM since the 1970s? Vaginal hormones have existed since the 1970s, but we’re using them only for pain with sex and not for GSM. In fact, data show that by using vaginal hormones, we can prevent UTIs by more than 50%. We can save lives using safe, effective, local, low-dose vaginal hormone strategies. And they are safe and effective for all of our patients in pre- and post menopause.
There are five different treatment options: vaginal estrogen inserts, vaginal estrogen creams, vaginal dehydroepiandrosterone (DHEA), low-dose vaginal estrogen rings, and an oral pill option called ospemifene (Osphena). All are used to treat GSM and will only work if your patient actually uses them and continues to use them.
These treatments are safe. They are effective. They do not increase the level of systemic hormones in the bloodstream. I have many patients with breast cancer who use these products as well. The only patients you may want to talk to your oncology colleagues about is women on active aromatase inhibitors.
We have to understand that UTIs kill people and having GSM is debilitating, often requiring pain medication because it can hurt to sit or to wear pads and our patients’ quality of life is severely affected. So please consider learning how to treat GSM. It turns out you don’t have to do exams. You don’t have to do follow-up. You can give these therapies, and women can use them for life.
Now, if your patient has vaginal bleeding, of course they need to see their gynecologist. But this is something every primary care doctor can and should do. As a urologist, we prescribe a lot of tamsulosin (Flomax) for our male patients to help with urination. Vaginal estrogen or DHEA is basically like Flomax for women, but it prevents UTIs and actually works like sildenafil (Viagra) because it can help orgasm and reduce pain with sex.
You have access to affordable, safe, effective treatment options to treat GSM. So check them out and hopefully change the world.
Dr. Rubin is an assistant clinical professor in the department of urology at Georgetown University, Washington. She reported conflicts of interest with Sprout, Maternal Medical, Absorption Pharmaceuticals, GlaxoSmithKline, and Endo.
A version of this article first appeared on Medscape.com.
Young women rate top sources for STI self-testing
, based on surveys from 92 individuals.
Direct-to-consumer (DTC) sexually transmitted infection (STI) screening methods involve the use of self-collected samples outside of a clinical setting, and may help reach women who avoid screening or lack access to clinical care, wrote Stacey B. Griner, PhD, of the University of North Texas Health Science Center, Fort Worth, and colleagues.
However, data on the methods used to promote DTC to the young female population are limited, and the goal of the current study was to identify preferred sources and communication channels for DTC STI information in this population, they said.
In a study published in Sexually Transmitted Diseases, the researchers reviewed data from 92 women aged 18-24 years at a single university who participated in an online survey. Of these, 24 also participated in in-depth interviews. The mean age of the participants was 20.0 years, and all reported being sexually active in the past year. Approximately two-thirds (68.5%) were White, 24% were Hispanic, 13% were Black or African American; 63.0% overall were heterosexual.
Participants received a description of DTC methods and were asked whether they were interested in receiving more information, and if so, what were their preferred sources for receiving the information. Potential sources included health care providers, friends, family members, partners, the Internet, college resources, classes, and other, and participants were asked to rank these choices in order of preference.
More than half of the participants identified health care providers as their preferred source of information (56.5%), followed by trusted websites (25%), and university-based resources or friends (6.5% for both).
Overall, participants who underwent STI screening in the past 12 months ranked college resources higher than those who had not undergone screening.
Race played a significant role in ranking partners and family members as resources. Compared with Black participants, White participants and those who were biracial/multiracial/another race ranked partners as a significantly more preferred source, but the differences between White and biracial/multiracial/another race were not significant. White participants and Black participants were similar in ranking family as a preferred information source, but White participants, compared with biracial/multiracial/other participants, ranked family as a significantly more preferred source.
Differences in rankings were similar across sexual orientations.
In-depth interviews were conducted on the college campus prior to the COVID-19 pandemic. The mean age of the interview participants was 19.5 years, and most were non-Hispanic White. Sexual orientation was varied, with 50% identifying as heterosexual and 50% identifying as a sexual minority.
In the interviews, health care providers were seen as influential for considering DTC methods, with gynecologists, other specialists, and more experienced physicians deemed the most trustworthy. Interviewees noted social media sites as a way to provide information and raise awareness of DTC methods, such as through the advertisements feature on Instagram. They also identified university orientation as a way to reach students and provide information about DTC options in the context of other health-related orientation topics such as sexual consent and alcohol use.
Many interviewees also mentioned friends as a resource for discussing sex, sexuality, and STI screening, and said they would be accepting of information, knowledge, and emotional support when learning about DTC from friends.
The findings were limited by several factors, including the cross-sectional design, use of data from a single campus setting, and the overrepresentation of White women, and more studies are needed to identify differences by region and campus type that might guide interventions, the researchers noted. The study also was limited by “the lack of specificity of what participants considered to be credible Internet information sources,” they said.
However, the results suggest that using health care providers, trusted websites, and established college resources as dissemination channels may help increase the awareness and use of DTC methods for STI screening in young women, they concluded.
The study was supported in part by the Doug Kirby Adolescent Sexual Health Research Grant from the Rural Center for AIDS/STD Prevention at Indiana University and by the University of South Florida College of Public Health. The researchers had no financial conflicts to disclose.
, based on surveys from 92 individuals.
Direct-to-consumer (DTC) sexually transmitted infection (STI) screening methods involve the use of self-collected samples outside of a clinical setting, and may help reach women who avoid screening or lack access to clinical care, wrote Stacey B. Griner, PhD, of the University of North Texas Health Science Center, Fort Worth, and colleagues.
However, data on the methods used to promote DTC to the young female population are limited, and the goal of the current study was to identify preferred sources and communication channels for DTC STI information in this population, they said.
In a study published in Sexually Transmitted Diseases, the researchers reviewed data from 92 women aged 18-24 years at a single university who participated in an online survey. Of these, 24 also participated in in-depth interviews. The mean age of the participants was 20.0 years, and all reported being sexually active in the past year. Approximately two-thirds (68.5%) were White, 24% were Hispanic, 13% were Black or African American; 63.0% overall were heterosexual.
Participants received a description of DTC methods and were asked whether they were interested in receiving more information, and if so, what were their preferred sources for receiving the information. Potential sources included health care providers, friends, family members, partners, the Internet, college resources, classes, and other, and participants were asked to rank these choices in order of preference.
More than half of the participants identified health care providers as their preferred source of information (56.5%), followed by trusted websites (25%), and university-based resources or friends (6.5% for both).
Overall, participants who underwent STI screening in the past 12 months ranked college resources higher than those who had not undergone screening.
Race played a significant role in ranking partners and family members as resources. Compared with Black participants, White participants and those who were biracial/multiracial/another race ranked partners as a significantly more preferred source, but the differences between White and biracial/multiracial/another race were not significant. White participants and Black participants were similar in ranking family as a preferred information source, but White participants, compared with biracial/multiracial/other participants, ranked family as a significantly more preferred source.
Differences in rankings were similar across sexual orientations.
In-depth interviews were conducted on the college campus prior to the COVID-19 pandemic. The mean age of the interview participants was 19.5 years, and most were non-Hispanic White. Sexual orientation was varied, with 50% identifying as heterosexual and 50% identifying as a sexual minority.
In the interviews, health care providers were seen as influential for considering DTC methods, with gynecologists, other specialists, and more experienced physicians deemed the most trustworthy. Interviewees noted social media sites as a way to provide information and raise awareness of DTC methods, such as through the advertisements feature on Instagram. They also identified university orientation as a way to reach students and provide information about DTC options in the context of other health-related orientation topics such as sexual consent and alcohol use.
Many interviewees also mentioned friends as a resource for discussing sex, sexuality, and STI screening, and said they would be accepting of information, knowledge, and emotional support when learning about DTC from friends.
The findings were limited by several factors, including the cross-sectional design, use of data from a single campus setting, and the overrepresentation of White women, and more studies are needed to identify differences by region and campus type that might guide interventions, the researchers noted. The study also was limited by “the lack of specificity of what participants considered to be credible Internet information sources,” they said.
However, the results suggest that using health care providers, trusted websites, and established college resources as dissemination channels may help increase the awareness and use of DTC methods for STI screening in young women, they concluded.
The study was supported in part by the Doug Kirby Adolescent Sexual Health Research Grant from the Rural Center for AIDS/STD Prevention at Indiana University and by the University of South Florida College of Public Health. The researchers had no financial conflicts to disclose.
, based on surveys from 92 individuals.
Direct-to-consumer (DTC) sexually transmitted infection (STI) screening methods involve the use of self-collected samples outside of a clinical setting, and may help reach women who avoid screening or lack access to clinical care, wrote Stacey B. Griner, PhD, of the University of North Texas Health Science Center, Fort Worth, and colleagues.
However, data on the methods used to promote DTC to the young female population are limited, and the goal of the current study was to identify preferred sources and communication channels for DTC STI information in this population, they said.
In a study published in Sexually Transmitted Diseases, the researchers reviewed data from 92 women aged 18-24 years at a single university who participated in an online survey. Of these, 24 also participated in in-depth interviews. The mean age of the participants was 20.0 years, and all reported being sexually active in the past year. Approximately two-thirds (68.5%) were White, 24% were Hispanic, 13% were Black or African American; 63.0% overall were heterosexual.
Participants received a description of DTC methods and were asked whether they were interested in receiving more information, and if so, what were their preferred sources for receiving the information. Potential sources included health care providers, friends, family members, partners, the Internet, college resources, classes, and other, and participants were asked to rank these choices in order of preference.
More than half of the participants identified health care providers as their preferred source of information (56.5%), followed by trusted websites (25%), and university-based resources or friends (6.5% for both).
Overall, participants who underwent STI screening in the past 12 months ranked college resources higher than those who had not undergone screening.
Race played a significant role in ranking partners and family members as resources. Compared with Black participants, White participants and those who were biracial/multiracial/another race ranked partners as a significantly more preferred source, but the differences between White and biracial/multiracial/another race were not significant. White participants and Black participants were similar in ranking family as a preferred information source, but White participants, compared with biracial/multiracial/other participants, ranked family as a significantly more preferred source.
Differences in rankings were similar across sexual orientations.
In-depth interviews were conducted on the college campus prior to the COVID-19 pandemic. The mean age of the interview participants was 19.5 years, and most were non-Hispanic White. Sexual orientation was varied, with 50% identifying as heterosexual and 50% identifying as a sexual minority.
In the interviews, health care providers were seen as influential for considering DTC methods, with gynecologists, other specialists, and more experienced physicians deemed the most trustworthy. Interviewees noted social media sites as a way to provide information and raise awareness of DTC methods, such as through the advertisements feature on Instagram. They also identified university orientation as a way to reach students and provide information about DTC options in the context of other health-related orientation topics such as sexual consent and alcohol use.
Many interviewees also mentioned friends as a resource for discussing sex, sexuality, and STI screening, and said they would be accepting of information, knowledge, and emotional support when learning about DTC from friends.
The findings were limited by several factors, including the cross-sectional design, use of data from a single campus setting, and the overrepresentation of White women, and more studies are needed to identify differences by region and campus type that might guide interventions, the researchers noted. The study also was limited by “the lack of specificity of what participants considered to be credible Internet information sources,” they said.
However, the results suggest that using health care providers, trusted websites, and established college resources as dissemination channels may help increase the awareness and use of DTC methods for STI screening in young women, they concluded.
The study was supported in part by the Doug Kirby Adolescent Sexual Health Research Grant from the Rural Center for AIDS/STD Prevention at Indiana University and by the University of South Florida College of Public Health. The researchers had no financial conflicts to disclose.
FROM SEXUALLY TRANSMITTED DISEASES
Pelvic yoga, physical conditioning both improve urinary incontinence
PHILADELPHIA – Both a pelvic yoga program and a general physical conditioning program for incontinence led to improvements in women’s incontinence, according to a study presented at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
“As clinicians, we’re usually focused on treatments that we ourselves can prescribe, perform, or administer. We’re not as good as recommending or supporting treatment or management strategies that don’t rely on costly or intensive visits with clinical specialists,” lead author Alison Huang, MD, MAS, a professor of medicine at the University of California, San Francisco, said in an interview.
“But our findings suggest that women who try pelvic yoga as a complementary management strategy for genitourinary conditions like urinary incontinence that often emerge in midlife are likely to experience substantial improvement in their genitourinary symptoms and function,” Dr. Huang said. “Some of these improvements may be shared with other forms of low-impact physical movement or exercise.”
The 240 participants from communities around three Northern California sites ranged in age from 45 to 90 years old, with an average age of 62, and all had at least daily urgency, stress, or mixed-type urinary incontinence. While most were White women, 40% identified as racial/ethnic minorities, including 14% Hispanic, 6% Black, 16% Asian American, and 4% multiracial.
Participants needed to be able to walk two blocks on level ground and get from a supine to a standing position on their own, but they should not have recently participated in any organized yoga or physical conditioning exercise classes. They also needed to forgo behavioral, invasive, or pharmacologic treatments for urinary incontinence for at least 3 months. The trial ran from 2019 to 2022, with most women completing the 3-month program virtually once the pandemic began.
The 121 women randomly assigned to the pelvic yoga program had twice-weekly group instruction by trained yoga instructors and once-weekly individual practice. The practice focused on 16 standard Hatha yoga poses in standing, seated, supine, and prone positions with an emphasis on precise alignment of their postures during each pose. Yoga props, such as blocks, straps, or bolsters, were available to minimize risk of injury and to accommodate women with less flexibility.
The 119 women randomly assigned to the physical conditioning group spent the same amount of group and individual class time on skeletal muscle stretching and strengthening exercises. These exercises focused on strengthening and stretching exercises for the upper and lower extremities in standing, sitting, or supine positions. The only props needed were exercise straps and handles and an exercise mat, and the program was designed to be safe and feasible for women across all ages.
Both groups received standard self-management pamphlets describing pelvic floor muscle exercises and recommendations on timed urination and urging suppression. After early dropouts from both arms, 107 women remained for analysis in the pelvic yoga group, and 113 women remained for analysis in the physical conditioning group.
Researchers assessed participants’ genitourinary quality of life at baseline and after 3 months using the Urogenital Distress Inventory-6 (UDI-6), Incontinence Impact Questionnaire (IIQ), and Patient Perception of Bladder Condition (PPBC). At baseline, the women’s average scores were 38.8 on the UDI-6, 101 on the IIQ, and 3.4 on the PPBC.
About one-third of the women in both groups attended all 24 group classes, and 57% of women in both groups attended 20-23 classes. In addition, 65% of the women in the pelvic yoga group and 73% of the women in the physical conditioning group completed all of the recommended additional hours of individual practice. Only 15% of pelvic yoga participants and 9% of physical conditioning participants completed less than 80% of the recommended individual practice hours. No differences in participation between the groups were statistically significant.
“Over 3 months, scores on all genitourinary quality of life measures improved by more than the minimum important difference thresholds in the pelvic yoga group,” the researchers reported, but only the UDI-6 score improved significantly – albeit still modestly – in the pelvic yoga group, compared with the physical conditioning group. Average scores improved 18.9 points in the pelvic yoga group and 13.1 points in the physical conditioning group (5.8-point difference; P = .02).
The scores on the IIQ improved an average 38.5 points in the pelvic yoga group and 31.4 points in the physical conditioning group (P = .48). PPBC scores improved 0.7 points in both groups.
“While yoga may offer benefits for genitourinary quality of life, it may not offer superior benefits compared to equivalent-time practice of other activities that improve general physical function,” Dr. Huang told attendees.
“The bottom line is that physical activity toward incontinence is a helpful technique,” Stephanie Faubion, MD, MBA, director for Mayo Clinic’s Center for Women’s Health and medical director for the Menopause Society, said in an interview regarding the findings. Urinary incontinence is under-recognized, Dr. Faubion said, “because women are embarrassed, so they don’t bring it up, so it doesn’t get managed.” But it’s a common problem, so clinicians need to ask patients about it, she said.
“We should realize that, in midlife and older age, genitourinary health is often connected to overall health,” Dr. Huang said in an interview. “We shouldn’t focus exclusively on treatments that are directed solely at the genital or lower urinary tract organs or tissues. We should consider the ways in which women’s urinary and sexual function are influenced by other aspects of their physical and cognitive health.”
The research was funded by the National Institutes of Health. Dr. Huang and Dr. Faubion had no disclosures.
PHILADELPHIA – Both a pelvic yoga program and a general physical conditioning program for incontinence led to improvements in women’s incontinence, according to a study presented at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
“As clinicians, we’re usually focused on treatments that we ourselves can prescribe, perform, or administer. We’re not as good as recommending or supporting treatment or management strategies that don’t rely on costly or intensive visits with clinical specialists,” lead author Alison Huang, MD, MAS, a professor of medicine at the University of California, San Francisco, said in an interview.
“But our findings suggest that women who try pelvic yoga as a complementary management strategy for genitourinary conditions like urinary incontinence that often emerge in midlife are likely to experience substantial improvement in their genitourinary symptoms and function,” Dr. Huang said. “Some of these improvements may be shared with other forms of low-impact physical movement or exercise.”
The 240 participants from communities around three Northern California sites ranged in age from 45 to 90 years old, with an average age of 62, and all had at least daily urgency, stress, or mixed-type urinary incontinence. While most were White women, 40% identified as racial/ethnic minorities, including 14% Hispanic, 6% Black, 16% Asian American, and 4% multiracial.
Participants needed to be able to walk two blocks on level ground and get from a supine to a standing position on their own, but they should not have recently participated in any organized yoga or physical conditioning exercise classes. They also needed to forgo behavioral, invasive, or pharmacologic treatments for urinary incontinence for at least 3 months. The trial ran from 2019 to 2022, with most women completing the 3-month program virtually once the pandemic began.
The 121 women randomly assigned to the pelvic yoga program had twice-weekly group instruction by trained yoga instructors and once-weekly individual practice. The practice focused on 16 standard Hatha yoga poses in standing, seated, supine, and prone positions with an emphasis on precise alignment of their postures during each pose. Yoga props, such as blocks, straps, or bolsters, were available to minimize risk of injury and to accommodate women with less flexibility.
The 119 women randomly assigned to the physical conditioning group spent the same amount of group and individual class time on skeletal muscle stretching and strengthening exercises. These exercises focused on strengthening and stretching exercises for the upper and lower extremities in standing, sitting, or supine positions. The only props needed were exercise straps and handles and an exercise mat, and the program was designed to be safe and feasible for women across all ages.
Both groups received standard self-management pamphlets describing pelvic floor muscle exercises and recommendations on timed urination and urging suppression. After early dropouts from both arms, 107 women remained for analysis in the pelvic yoga group, and 113 women remained for analysis in the physical conditioning group.
Researchers assessed participants’ genitourinary quality of life at baseline and after 3 months using the Urogenital Distress Inventory-6 (UDI-6), Incontinence Impact Questionnaire (IIQ), and Patient Perception of Bladder Condition (PPBC). At baseline, the women’s average scores were 38.8 on the UDI-6, 101 on the IIQ, and 3.4 on the PPBC.
About one-third of the women in both groups attended all 24 group classes, and 57% of women in both groups attended 20-23 classes. In addition, 65% of the women in the pelvic yoga group and 73% of the women in the physical conditioning group completed all of the recommended additional hours of individual practice. Only 15% of pelvic yoga participants and 9% of physical conditioning participants completed less than 80% of the recommended individual practice hours. No differences in participation between the groups were statistically significant.
“Over 3 months, scores on all genitourinary quality of life measures improved by more than the minimum important difference thresholds in the pelvic yoga group,” the researchers reported, but only the UDI-6 score improved significantly – albeit still modestly – in the pelvic yoga group, compared with the physical conditioning group. Average scores improved 18.9 points in the pelvic yoga group and 13.1 points in the physical conditioning group (5.8-point difference; P = .02).
The scores on the IIQ improved an average 38.5 points in the pelvic yoga group and 31.4 points in the physical conditioning group (P = .48). PPBC scores improved 0.7 points in both groups.
“While yoga may offer benefits for genitourinary quality of life, it may not offer superior benefits compared to equivalent-time practice of other activities that improve general physical function,” Dr. Huang told attendees.
“The bottom line is that physical activity toward incontinence is a helpful technique,” Stephanie Faubion, MD, MBA, director for Mayo Clinic’s Center for Women’s Health and medical director for the Menopause Society, said in an interview regarding the findings. Urinary incontinence is under-recognized, Dr. Faubion said, “because women are embarrassed, so they don’t bring it up, so it doesn’t get managed.” But it’s a common problem, so clinicians need to ask patients about it, she said.
“We should realize that, in midlife and older age, genitourinary health is often connected to overall health,” Dr. Huang said in an interview. “We shouldn’t focus exclusively on treatments that are directed solely at the genital or lower urinary tract organs or tissues. We should consider the ways in which women’s urinary and sexual function are influenced by other aspects of their physical and cognitive health.”
The research was funded by the National Institutes of Health. Dr. Huang and Dr. Faubion had no disclosures.
PHILADELPHIA – Both a pelvic yoga program and a general physical conditioning program for incontinence led to improvements in women’s incontinence, according to a study presented at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
“As clinicians, we’re usually focused on treatments that we ourselves can prescribe, perform, or administer. We’re not as good as recommending or supporting treatment or management strategies that don’t rely on costly or intensive visits with clinical specialists,” lead author Alison Huang, MD, MAS, a professor of medicine at the University of California, San Francisco, said in an interview.
“But our findings suggest that women who try pelvic yoga as a complementary management strategy for genitourinary conditions like urinary incontinence that often emerge in midlife are likely to experience substantial improvement in their genitourinary symptoms and function,” Dr. Huang said. “Some of these improvements may be shared with other forms of low-impact physical movement or exercise.”
The 240 participants from communities around three Northern California sites ranged in age from 45 to 90 years old, with an average age of 62, and all had at least daily urgency, stress, or mixed-type urinary incontinence. While most were White women, 40% identified as racial/ethnic minorities, including 14% Hispanic, 6% Black, 16% Asian American, and 4% multiracial.
Participants needed to be able to walk two blocks on level ground and get from a supine to a standing position on their own, but they should not have recently participated in any organized yoga or physical conditioning exercise classes. They also needed to forgo behavioral, invasive, or pharmacologic treatments for urinary incontinence for at least 3 months. The trial ran from 2019 to 2022, with most women completing the 3-month program virtually once the pandemic began.
The 121 women randomly assigned to the pelvic yoga program had twice-weekly group instruction by trained yoga instructors and once-weekly individual practice. The practice focused on 16 standard Hatha yoga poses in standing, seated, supine, and prone positions with an emphasis on precise alignment of their postures during each pose. Yoga props, such as blocks, straps, or bolsters, were available to minimize risk of injury and to accommodate women with less flexibility.
The 119 women randomly assigned to the physical conditioning group spent the same amount of group and individual class time on skeletal muscle stretching and strengthening exercises. These exercises focused on strengthening and stretching exercises for the upper and lower extremities in standing, sitting, or supine positions. The only props needed were exercise straps and handles and an exercise mat, and the program was designed to be safe and feasible for women across all ages.
Both groups received standard self-management pamphlets describing pelvic floor muscle exercises and recommendations on timed urination and urging suppression. After early dropouts from both arms, 107 women remained for analysis in the pelvic yoga group, and 113 women remained for analysis in the physical conditioning group.
Researchers assessed participants’ genitourinary quality of life at baseline and after 3 months using the Urogenital Distress Inventory-6 (UDI-6), Incontinence Impact Questionnaire (IIQ), and Patient Perception of Bladder Condition (PPBC). At baseline, the women’s average scores were 38.8 on the UDI-6, 101 on the IIQ, and 3.4 on the PPBC.
About one-third of the women in both groups attended all 24 group classes, and 57% of women in both groups attended 20-23 classes. In addition, 65% of the women in the pelvic yoga group and 73% of the women in the physical conditioning group completed all of the recommended additional hours of individual practice. Only 15% of pelvic yoga participants and 9% of physical conditioning participants completed less than 80% of the recommended individual practice hours. No differences in participation between the groups were statistically significant.
“Over 3 months, scores on all genitourinary quality of life measures improved by more than the minimum important difference thresholds in the pelvic yoga group,” the researchers reported, but only the UDI-6 score improved significantly – albeit still modestly – in the pelvic yoga group, compared with the physical conditioning group. Average scores improved 18.9 points in the pelvic yoga group and 13.1 points in the physical conditioning group (5.8-point difference; P = .02).
The scores on the IIQ improved an average 38.5 points in the pelvic yoga group and 31.4 points in the physical conditioning group (P = .48). PPBC scores improved 0.7 points in both groups.
“While yoga may offer benefits for genitourinary quality of life, it may not offer superior benefits compared to equivalent-time practice of other activities that improve general physical function,” Dr. Huang told attendees.
“The bottom line is that physical activity toward incontinence is a helpful technique,” Stephanie Faubion, MD, MBA, director for Mayo Clinic’s Center for Women’s Health and medical director for the Menopause Society, said in an interview regarding the findings. Urinary incontinence is under-recognized, Dr. Faubion said, “because women are embarrassed, so they don’t bring it up, so it doesn’t get managed.” But it’s a common problem, so clinicians need to ask patients about it, she said.
“We should realize that, in midlife and older age, genitourinary health is often connected to overall health,” Dr. Huang said in an interview. “We shouldn’t focus exclusively on treatments that are directed solely at the genital or lower urinary tract organs or tissues. We should consider the ways in which women’s urinary and sexual function are influenced by other aspects of their physical and cognitive health.”
The research was funded by the National Institutes of Health. Dr. Huang and Dr. Faubion had no disclosures.
AT NAMS 2023