User login
Losing Your Mind Trying to Understand the BP-Dementia Link
You could be forgiven if you are confused about how blood pressure (BP) affects dementia. First, you read an article extolling the benefits of BP lowering, then a study about how stopping antihypertensives slows cognitive decline in nursing home residents. It’s enough to make you lose your mind.
The Brain Benefits of BP Lowering
It should be stated unequivocally that you should absolutely treat high BP. It may have once been acceptable to state, “The greatest danger to a man with high blood pressure lies in its discovery, because then some fool is certain to try and reduce it.” But those dark days are long behind us.
In these divided times, at least we can agree that we should treat high BP. The cardiovascular (CV) benefits, in and of themselves, justify the decision. But BP’s relationship with dementia is more complex. There are different types of dementia even though we tend to lump them all into one category. Vascular dementia is driven by the same pathophysiology and risk factors as cardiac disease. It’s intuitive that treating hypertension, diabetes, hypercholesterolemia, and smoking will decrease the risk for stroke and limit the damage to the brain that we see with repeated vascular insults. For Alzheimer’s disease, high BP and other CV risk factors seem to increase the risk even if the mechanism is not fully elucidated.
Estimates suggest that if we could lower the prevalence of hypertension by 25%, there would be 160,000 fewer cases of Alzheimer’s disease. But the data are not as robust as one might hope. A 2021 Cochrane review found that hypertension treatment slowed cognitive decline, but the quality of the evidence was low. Short duration of follow-up, dropouts, crossovers, and other problems with the data precluded any certainty. What’s more, hypertension in midlife is associated with cognitive decline and dementia, but its impact in those over age 70 is less clear. Later in life, or once cognitive impairment has already developed, it may be too late for BP lowering to have any impact.
Potential Harms of Lowering BP
All this needs to be weighed against the potential harms of treating hypertension. I will reiterate that hypertension should be treated and treated aggressively for the prevention of CV events. But overtreatment, especially in older patients, is associated with hypotension, falls, and syncope. Older patients are also at risk for polypharmacy and drug-drug interactions.
A Korean nationwide survey showed a U-shaped association between BP and Alzheimer’s disease risk in adults (mean age, 67 years), with both high and low BPs associated with a higher risk for Alzheimer’s disease. Though not all studies agree. A post hoc analysis of SPRINT MIND did not find any negative impact of intensive BP lowering on cognitive outcomes or cerebral perfusion in older adults (mean age, 68 years). But it didn’t do much good either. Given the heterogeneity of the data, doubts remain on whether aggressive BP lowering might be detrimental in older patients with comorbidities and preexisting dementia. The obvious corollary then is whether deprescribing hypertensive medications could be beneficial.
A recent publication in JAMA Internal Medicine attempted to address this very question. The cohort study used data from Veterans Affairs nursing home residents (mean age, 78 years) to emulate a randomized trial on deprescribing antihypertensives and cognitive decline. Many of the residents’ cognitive scores worsened over the course of follow-up; however, the decline was less pronounced in the deprescribing group (10% vs 12%). The same group did a similar analysis looking at CV outcomes and found no increased risk for heart attack or stroke with deprescribing BP medications. Taken together, these nursing home data suggest that deprescribing may help slow cognitive decline without the expected trade-off of increased CV events.
Deprescribing, Yes or No?
However, randomized data would obviously be preferable, and these are in short supply. One such trial, the DANTE study, found no benefit to deprescribing in terms of cognition in adults aged 75 years or older with mild cognitive impairment. The study follow-up was only 16 weeks, however, which is hardly enough time to demonstrate any effect, positive or negative. The most that can be said is that it didn’t cause many short-term adverse events.
Perhaps the best conclusion to draw from this somewhat underwhelming collection of data is that lowering high BP is important, but less important the closer we get to the end of life. Hypotension is obviously bad, and overly aggressive BP lowering is going to lead to negative outcomes in older adults because gravity is an unforgiving mistress.
Deprescribing antihypertensives in older adults is probably not going to cause major negative outcomes, but whether it will do much good in nonhypotensive patients is debatable. The bigger problem is the millions of people with undiagnosed or undertreated hypertension. We would probably have less dementia if we treated hypertension when it does the most good: as a primary-prevention strategy in midlife.
Dr. Labos is a cardiologist at Hôpital Notre-Dame, Montreal, Quebec, Canada. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
You could be forgiven if you are confused about how blood pressure (BP) affects dementia. First, you read an article extolling the benefits of BP lowering, then a study about how stopping antihypertensives slows cognitive decline in nursing home residents. It’s enough to make you lose your mind.
The Brain Benefits of BP Lowering
It should be stated unequivocally that you should absolutely treat high BP. It may have once been acceptable to state, “The greatest danger to a man with high blood pressure lies in its discovery, because then some fool is certain to try and reduce it.” But those dark days are long behind us.
In these divided times, at least we can agree that we should treat high BP. The cardiovascular (CV) benefits, in and of themselves, justify the decision. But BP’s relationship with dementia is more complex. There are different types of dementia even though we tend to lump them all into one category. Vascular dementia is driven by the same pathophysiology and risk factors as cardiac disease. It’s intuitive that treating hypertension, diabetes, hypercholesterolemia, and smoking will decrease the risk for stroke and limit the damage to the brain that we see with repeated vascular insults. For Alzheimer’s disease, high BP and other CV risk factors seem to increase the risk even if the mechanism is not fully elucidated.
Estimates suggest that if we could lower the prevalence of hypertension by 25%, there would be 160,000 fewer cases of Alzheimer’s disease. But the data are not as robust as one might hope. A 2021 Cochrane review found that hypertension treatment slowed cognitive decline, but the quality of the evidence was low. Short duration of follow-up, dropouts, crossovers, and other problems with the data precluded any certainty. What’s more, hypertension in midlife is associated with cognitive decline and dementia, but its impact in those over age 70 is less clear. Later in life, or once cognitive impairment has already developed, it may be too late for BP lowering to have any impact.
Potential Harms of Lowering BP
All this needs to be weighed against the potential harms of treating hypertension. I will reiterate that hypertension should be treated and treated aggressively for the prevention of CV events. But overtreatment, especially in older patients, is associated with hypotension, falls, and syncope. Older patients are also at risk for polypharmacy and drug-drug interactions.
A Korean nationwide survey showed a U-shaped association between BP and Alzheimer’s disease risk in adults (mean age, 67 years), with both high and low BPs associated with a higher risk for Alzheimer’s disease. Though not all studies agree. A post hoc analysis of SPRINT MIND did not find any negative impact of intensive BP lowering on cognitive outcomes or cerebral perfusion in older adults (mean age, 68 years). But it didn’t do much good either. Given the heterogeneity of the data, doubts remain on whether aggressive BP lowering might be detrimental in older patients with comorbidities and preexisting dementia. The obvious corollary then is whether deprescribing hypertensive medications could be beneficial.
A recent publication in JAMA Internal Medicine attempted to address this very question. The cohort study used data from Veterans Affairs nursing home residents (mean age, 78 years) to emulate a randomized trial on deprescribing antihypertensives and cognitive decline. Many of the residents’ cognitive scores worsened over the course of follow-up; however, the decline was less pronounced in the deprescribing group (10% vs 12%). The same group did a similar analysis looking at CV outcomes and found no increased risk for heart attack or stroke with deprescribing BP medications. Taken together, these nursing home data suggest that deprescribing may help slow cognitive decline without the expected trade-off of increased CV events.
Deprescribing, Yes or No?
However, randomized data would obviously be preferable, and these are in short supply. One such trial, the DANTE study, found no benefit to deprescribing in terms of cognition in adults aged 75 years or older with mild cognitive impairment. The study follow-up was only 16 weeks, however, which is hardly enough time to demonstrate any effect, positive or negative. The most that can be said is that it didn’t cause many short-term adverse events.
Perhaps the best conclusion to draw from this somewhat underwhelming collection of data is that lowering high BP is important, but less important the closer we get to the end of life. Hypotension is obviously bad, and overly aggressive BP lowering is going to lead to negative outcomes in older adults because gravity is an unforgiving mistress.
Deprescribing antihypertensives in older adults is probably not going to cause major negative outcomes, but whether it will do much good in nonhypotensive patients is debatable. The bigger problem is the millions of people with undiagnosed or undertreated hypertension. We would probably have less dementia if we treated hypertension when it does the most good: as a primary-prevention strategy in midlife.
Dr. Labos is a cardiologist at Hôpital Notre-Dame, Montreal, Quebec, Canada. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
You could be forgiven if you are confused about how blood pressure (BP) affects dementia. First, you read an article extolling the benefits of BP lowering, then a study about how stopping antihypertensives slows cognitive decline in nursing home residents. It’s enough to make you lose your mind.
The Brain Benefits of BP Lowering
It should be stated unequivocally that you should absolutely treat high BP. It may have once been acceptable to state, “The greatest danger to a man with high blood pressure lies in its discovery, because then some fool is certain to try and reduce it.” But those dark days are long behind us.
In these divided times, at least we can agree that we should treat high BP. The cardiovascular (CV) benefits, in and of themselves, justify the decision. But BP’s relationship with dementia is more complex. There are different types of dementia even though we tend to lump them all into one category. Vascular dementia is driven by the same pathophysiology and risk factors as cardiac disease. It’s intuitive that treating hypertension, diabetes, hypercholesterolemia, and smoking will decrease the risk for stroke and limit the damage to the brain that we see with repeated vascular insults. For Alzheimer’s disease, high BP and other CV risk factors seem to increase the risk even if the mechanism is not fully elucidated.
Estimates suggest that if we could lower the prevalence of hypertension by 25%, there would be 160,000 fewer cases of Alzheimer’s disease. But the data are not as robust as one might hope. A 2021 Cochrane review found that hypertension treatment slowed cognitive decline, but the quality of the evidence was low. Short duration of follow-up, dropouts, crossovers, and other problems with the data precluded any certainty. What’s more, hypertension in midlife is associated with cognitive decline and dementia, but its impact in those over age 70 is less clear. Later in life, or once cognitive impairment has already developed, it may be too late for BP lowering to have any impact.
Potential Harms of Lowering BP
All this needs to be weighed against the potential harms of treating hypertension. I will reiterate that hypertension should be treated and treated aggressively for the prevention of CV events. But overtreatment, especially in older patients, is associated with hypotension, falls, and syncope. Older patients are also at risk for polypharmacy and drug-drug interactions.
A Korean nationwide survey showed a U-shaped association between BP and Alzheimer’s disease risk in adults (mean age, 67 years), with both high and low BPs associated with a higher risk for Alzheimer’s disease. Though not all studies agree. A post hoc analysis of SPRINT MIND did not find any negative impact of intensive BP lowering on cognitive outcomes or cerebral perfusion in older adults (mean age, 68 years). But it didn’t do much good either. Given the heterogeneity of the data, doubts remain on whether aggressive BP lowering might be detrimental in older patients with comorbidities and preexisting dementia. The obvious corollary then is whether deprescribing hypertensive medications could be beneficial.
A recent publication in JAMA Internal Medicine attempted to address this very question. The cohort study used data from Veterans Affairs nursing home residents (mean age, 78 years) to emulate a randomized trial on deprescribing antihypertensives and cognitive decline. Many of the residents’ cognitive scores worsened over the course of follow-up; however, the decline was less pronounced in the deprescribing group (10% vs 12%). The same group did a similar analysis looking at CV outcomes and found no increased risk for heart attack or stroke with deprescribing BP medications. Taken together, these nursing home data suggest that deprescribing may help slow cognitive decline without the expected trade-off of increased CV events.
Deprescribing, Yes or No?
However, randomized data would obviously be preferable, and these are in short supply. One such trial, the DANTE study, found no benefit to deprescribing in terms of cognition in adults aged 75 years or older with mild cognitive impairment. The study follow-up was only 16 weeks, however, which is hardly enough time to demonstrate any effect, positive or negative. The most that can be said is that it didn’t cause many short-term adverse events.
Perhaps the best conclusion to draw from this somewhat underwhelming collection of data is that lowering high BP is important, but less important the closer we get to the end of life. Hypotension is obviously bad, and overly aggressive BP lowering is going to lead to negative outcomes in older adults because gravity is an unforgiving mistress.
Deprescribing antihypertensives in older adults is probably not going to cause major negative outcomes, but whether it will do much good in nonhypotensive patients is debatable. The bigger problem is the millions of people with undiagnosed or undertreated hypertension. We would probably have less dementia if we treated hypertension when it does the most good: as a primary-prevention strategy in midlife.
Dr. Labos is a cardiologist at Hôpital Notre-Dame, Montreal, Quebec, Canada. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Nutrition, Drugs, or Bariatric Surgery: What’s the Best Approach for Sustained Weight Loss?
Given that more than 100 million US adults have obesity, including 22 million with severe obesity, physicians regularly see patients with the condition in their practices.
Fortunately, doctors have more tools than ever to help their patients. But the question remains: Which method is the safest and most effective? Is it diet and lifestyle changes, one of the recently approved anti-obesity medications (AOMs), bariatric surgery, or a combination approach?
There are no head-to-head trials comparing these three approaches, said Vanita Rahman, MD, clinic director of the Barnard Medical Center, Washington, DC, at the International Conference on Nutrition in Medicine, sponsored by the Physicians Committee for Responsible Medicine.
Instead, doctors must evaluate the merits and drawbacks of each intervention and decide with their patients which treatment is best for them, she told Medscape Medical News. When she sees patients, Rahman shares the pertinent research with them, so they are able to make an informed choice.
Looking at the Options
In her presentation at the conference, Rahman summarized the guidelines issued by the American Heart Association/American College of Cardiology/The Obesity Society for Management of Overweight and Obesity in Adults and the American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines For Medical Care of Patients with Obesity, including lifestyle changes, AOMs, and bariatric surgery (Table 1).
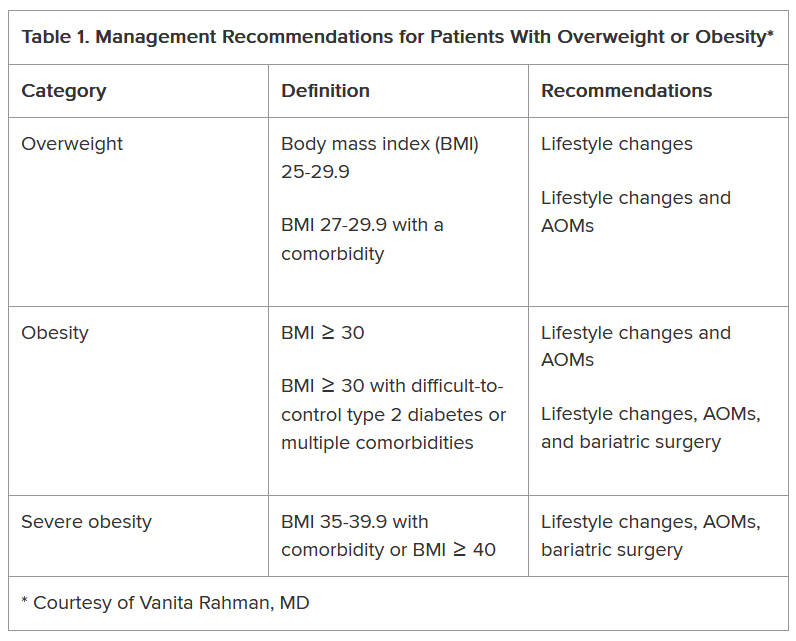
As shown, the current clinical guidelines offer recommendations that consider such factors as the patient’s BMI and presence of one or more comorbidities. Generally, they begin with lifestyle changes for people with overweight, the possibility of an AOM for those with obesity, and bariatric surgery as an option for those with severe obesity-related complications.
“In obesity, we traditionally thought the process was ‘either-or’ — either lifestyle or surgery or medication — and somehow lifestyle is better,” Sheethal Reddy, PhD, a psychologist at the Bariatric Center at Emory University Hospital Midtown, Atlanta, told Medscape Medical News.
Now physicians often use a combination of methods, but lifestyle is foundational to all of them, she said.
“If you don’t make lifestyle changes, none of the approaches will ultimately be effective,” said Reddy, who also is an assistant professor in the Division of General and GI Surgery at Emory School of Medicine, Atlanta.
Lifestyle changes don’t just involve diet and nutrition but include physical exercise.
“Being sedentary affects everything — sleep quality, appetite regulation, and metabolism. Without sufficient exercise, the body isn’t functioning well enough to have a healthy metabolism,” Reddy said.
How Durable Are the Interventions?
Although bariatric surgery has demonstrated effectiveness in helping patients lose weight, many of them regain some or most of it, Rahman said.
A systematic review and meta-analysis found weight regain in 49% of patients who underwent bariatric surgery patients, with the highest prevalence after Roux-en-Y gastric bypass.
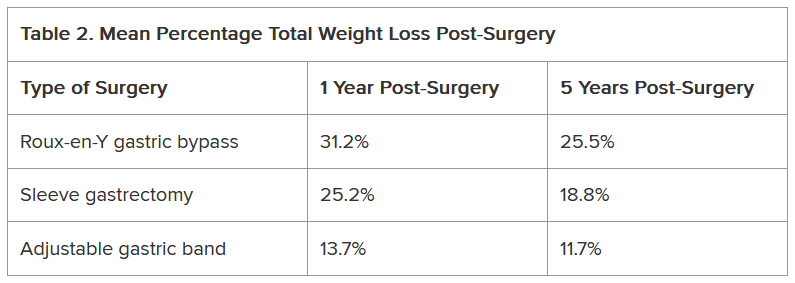
Another study of approximately 45,000 patients who underwent bariatric surgery found differences not only in the percentage of total weight loss among Roux-en-Y gastric bypass, sleeve gastrectomy, and adjustable gastric band procedures but also in how much of that weight stayed off between 1 and 5 years following the procedure (Table 2).
Weight regain also is a risk with AOMs, if they’re discontinued.
The STEP 1 trial tested the effectiveness of semaglutide — a glucagon-like peptide 1 (GLP-1) receptor agonist — as an adjunct to lifestyle intervention for weight loss in patients with obesity or with overweight and at least one comorbidity but not diabetes. Mean weight loss with semaglutide was 17.3% but that figure dropped 11.6 percentage points after treatment was discontinued.
Other studies also have found that patients regain weight after GLP-1 discontinuation.
Tirzepatide, a GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) combination, has shown efficacy with weight reduction, but patients experienced some weight regain upon discontinuation. In one study, patients experienced a mean weight loss of 20.9% after 36 weeks of tirzepatide. In the study’s subsequent 52-week double-blind, placebo-controlled period, patients who stopped taking the medication experienced a weight regain of 14%, whereas those who remained on the medication lost an additional 5.5% of weight.
GLP-1 and GLP-1/GIP medications do not address the factors that contribute to overweight and obesity, Rahman said. “They simply suppress the appetite; therefore, weight gain occurs after stopping them.”
Patients may stop taking anti-obesity drugs for a variety of reasons, including side effects. Rahman noted that the common side effects include nausea, vomiting, and constipation, whereas rare side effects include gastroparesis, gallbladder and biliary disease, thyroid cancer, and suicidal thoughts. GLP-1 and GLP-1/GIP medications also carry a risk for non-arteritic anterior ischemic optic neuropathy, she said.
Moreover, health insurance does not always cover these medications, which likely affects patient access to the drugs and compliance rates.
“Given the side effects and frequent lack of insurance coverage, significant questions remain about long-term safety and feasibility of these agents,” Rahman said.
What About Nutritional Approaches?
The lifestyle interventions in the semaglutide and tirzepatide studies included 500 kcal/d deficit diets, which is difficult for people to maintain, noted Rahman, who is the author of the book Simply Plant Based: Fabulous Food for a Healthy Life.
Additionally, bariatric surgery has been associated with long-term micronutrient deficiencies, including deficiencies in vitamins A, D, E, K, B1, and B12, as well as folate, iron, zinc, copper, selenium, and calcium, she said.
The best approach to food from a patient compliance standpoint and to avoid nutrient deficiencies is a whole-food, plant-based diet, Rahman said. She advocates this nutritional approach, along with physical activity, for patients regardless of whether they’ve selected lifestyle intervention alone or combined with an AOM or bariatric surgery to address obesity.
Rahman cited a 5-year heart disease study comparing an intensive lifestyle program involving a vegetarian diet, aerobic exercise, stress management training, smoking cessation, and group psychosocial support to treatment as usual. Patients in the lifestyle group lost 10.9 kg at 1 year and sustained weight loss of 5.8 kg at 5 years, whereas weight in the control group remained relatively unchanged from baseline.
She also pointed to the findings of a study of patients with obesity or with overweight and at least one comorbidity that compared standard care with a low-fat, whole-food, plant-based diet with vitamin B12 supplementation. At 6 months, mean BMI reduction was greater in the intervention group than the standard care group (−4.4 vs −0.4).
In her practice, Rahman has seen the benefits of a whole-food, plant-based diet for patients with obesity.
If people are committed to this type of dietary approach and are given the tools and resources to do it effectively, “their thinking changes, their taste buds change, and they grow to enjoy this new way of eating,” she said. “They see results, and it’s a lifestyle that can be sustained long-term.”
Addressing Drivers of Weight Gain
Patients also need help addressing the various factors that may contribute to overweight and obesity, including overconsumption of ultra-processed foods, substandard nutritional quality of restaurant foods, increasing portion sizes, distraction during eating, emotional eating, late-night eating, and cultural/traditional values surrounding food, Rahman noted.
Supatra Tovar, PsyD, RD, a clinical psychologist with a practice in Pasadena, California, agreed that identifying the reasons for weight gain is critical for treatment.
“If you’re not addressing underlying issues, such as a person’s relationship with food, behaviors around food, the tendency to mindlessly eat or emotionally eat or eat to seek comfort, the person’s weight problems won’t ultimately be fully solved by any of the three approaches — dieting, medications, or bariatric surgery,” she said.
Some of her patients “engage in extreme dieting and deprivation, and many who use medications or have had bariatric surgery hardly eat and often develop nutritional deficiencies,” said Tovar, author of the book Deprogram Diet Culture: Rethink Your Relationship with Food, Heal Your Mind, and Live a Diet-Free Life.
The key to healthy and sustained weight loss is to “become attuned to the body’s signals, learn how to honor hunger, stop eating when satisfied, and eat more healthful foods, such as fruits and vegetables, whole grains, lean proteins — especially plant-based proteins — and the body gives signals that this is what it wants,” she said.
Tovar doesn’t give her clients a specific diet or set of portions.
“I teach them to listen to their bodies,” she said. “They’ve lost significant amounts of weight and continued to keep it off because they’ve done this kind of work.”
When Lifestyle Changes Aren’t Enough
For many patients, lifestyle interventions are insufficient to address the degree of overweight and obesity and common comorbidities, said W. Timothy Garvey, MD, associate director and professor, Department of Nutrition Sciences, School of Health Professions, University of Alabama at Birmingham.
“Of course, nutritional approaches are very important, not only for weight but also for general health-related reasons,” said Garvey, lead author of the 2016 American Association of Clinical Endocrinologists obesity guidelines. “We’ve seen that the Mediterranean and some plant-based diets can prevent progression from prediabetes to diabetes and improve other parameters that reflect metabolic health.”
However, it’s “not common that patients can follow these diets, lose weight, and keep it off,” Garvey cautioned. Up to 50% of weight that’s lost through lifestyle changes is typically regained by 1-year follow-up, with almost all remaining lost weight subsequently regained in the majority of individuals because the person “has to fight against pathophysiological process that drive weight regain,” he noted.
Weight-loss medications can address these pathophysiologic processes by “addressing interactions of satiety hormones with feeding centers in the brain, suppressing the appetite, and making it easier for patients to adhere to a reduced-calorie diet.”
Garvey views the weight-loss medications in the same light as drugs for diabetes and hypertension, in that people need to keep taking them to sustain the benefit.
There’s still a role for bariatric surgery because not everyone can tolerate the AOMs or achieve sufficient weight loss.
“Patients with very high BMI who have trouble ambulating might benefit from a combination of bariatric surgery and medication,” Garvey said.
While some side effects are associated with AOMs, being an “alarmist” about them can be detrimental to patients, he warned.
Rahman and Tovar are authors of books about weight loss. Reddy reported no relevant financial relationships. Garvey is a consultant on advisory boards for Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Pfizer, Fractyl Health, Alnylam Pharmaceuticals, Inogen, Zealand, Allurion, Carmot/Roche, Terns Pharmaceuticals, Neurocrine, Keros Therapeutics, and Regeneron. He is the site principal investigator for multi-centered clinical trials sponsored by his university and funded by Novo Nordisk, Eli Lilly, Epitomee, Neurovalens, and Pfizer. He serves as a consultant on the advisory board for the nonprofit Milken Foundation and is a member of the Data Monitoring Committee for phase 3 clinical trials conducted by Boehringer-Ingelheim and Eli Lilly.
A version of this article first appeared on Medscape.com.
Given that more than 100 million US adults have obesity, including 22 million with severe obesity, physicians regularly see patients with the condition in their practices.
Fortunately, doctors have more tools than ever to help their patients. But the question remains: Which method is the safest and most effective? Is it diet and lifestyle changes, one of the recently approved anti-obesity medications (AOMs), bariatric surgery, or a combination approach?
There are no head-to-head trials comparing these three approaches, said Vanita Rahman, MD, clinic director of the Barnard Medical Center, Washington, DC, at the International Conference on Nutrition in Medicine, sponsored by the Physicians Committee for Responsible Medicine.
Instead, doctors must evaluate the merits and drawbacks of each intervention and decide with their patients which treatment is best for them, she told Medscape Medical News. When she sees patients, Rahman shares the pertinent research with them, so they are able to make an informed choice.
Looking at the Options
In her presentation at the conference, Rahman summarized the guidelines issued by the American Heart Association/American College of Cardiology/The Obesity Society for Management of Overweight and Obesity in Adults and the American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines For Medical Care of Patients with Obesity, including lifestyle changes, AOMs, and bariatric surgery (Table 1).

As shown, the current clinical guidelines offer recommendations that consider such factors as the patient’s BMI and presence of one or more comorbidities. Generally, they begin with lifestyle changes for people with overweight, the possibility of an AOM for those with obesity, and bariatric surgery as an option for those with severe obesity-related complications.
“In obesity, we traditionally thought the process was ‘either-or’ — either lifestyle or surgery or medication — and somehow lifestyle is better,” Sheethal Reddy, PhD, a psychologist at the Bariatric Center at Emory University Hospital Midtown, Atlanta, told Medscape Medical News.
Now physicians often use a combination of methods, but lifestyle is foundational to all of them, she said.
“If you don’t make lifestyle changes, none of the approaches will ultimately be effective,” said Reddy, who also is an assistant professor in the Division of General and GI Surgery at Emory School of Medicine, Atlanta.
Lifestyle changes don’t just involve diet and nutrition but include physical exercise.
“Being sedentary affects everything — sleep quality, appetite regulation, and metabolism. Without sufficient exercise, the body isn’t functioning well enough to have a healthy metabolism,” Reddy said.
How Durable Are the Interventions?
Although bariatric surgery has demonstrated effectiveness in helping patients lose weight, many of them regain some or most of it, Rahman said.
A systematic review and meta-analysis found weight regain in 49% of patients who underwent bariatric surgery patients, with the highest prevalence after Roux-en-Y gastric bypass.
Another study of approximately 45,000 patients who underwent bariatric surgery found differences not only in the percentage of total weight loss among Roux-en-Y gastric bypass, sleeve gastrectomy, and adjustable gastric band procedures but also in how much of that weight stayed off between 1 and 5 years following the procedure (Table 2).

Weight regain also is a risk with AOMs, if they’re discontinued.
The STEP 1 trial tested the effectiveness of semaglutide — a glucagon-like peptide 1 (GLP-1) receptor agonist — as an adjunct to lifestyle intervention for weight loss in patients with obesity or with overweight and at least one comorbidity but not diabetes. Mean weight loss with semaglutide was 17.3% but that figure dropped 11.6 percentage points after treatment was discontinued.
Other studies also have found that patients regain weight after GLP-1 discontinuation.
Tirzepatide, a GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) combination, has shown efficacy with weight reduction, but patients experienced some weight regain upon discontinuation. In one study, patients experienced a mean weight loss of 20.9% after 36 weeks of tirzepatide. In the study’s subsequent 52-week double-blind, placebo-controlled period, patients who stopped taking the medication experienced a weight regain of 14%, whereas those who remained on the medication lost an additional 5.5% of weight.
GLP-1 and GLP-1/GIP medications do not address the factors that contribute to overweight and obesity, Rahman said. “They simply suppress the appetite; therefore, weight gain occurs after stopping them.”
Patients may stop taking anti-obesity drugs for a variety of reasons, including side effects. Rahman noted that the common side effects include nausea, vomiting, and constipation, whereas rare side effects include gastroparesis, gallbladder and biliary disease, thyroid cancer, and suicidal thoughts. GLP-1 and GLP-1/GIP medications also carry a risk for non-arteritic anterior ischemic optic neuropathy, she said.
Moreover, health insurance does not always cover these medications, which likely affects patient access to the drugs and compliance rates.
“Given the side effects and frequent lack of insurance coverage, significant questions remain about long-term safety and feasibility of these agents,” Rahman said.
What About Nutritional Approaches?
The lifestyle interventions in the semaglutide and tirzepatide studies included 500 kcal/d deficit diets, which is difficult for people to maintain, noted Rahman, who is the author of the book Simply Plant Based: Fabulous Food for a Healthy Life.
Additionally, bariatric surgery has been associated with long-term micronutrient deficiencies, including deficiencies in vitamins A, D, E, K, B1, and B12, as well as folate, iron, zinc, copper, selenium, and calcium, she said.
The best approach to food from a patient compliance standpoint and to avoid nutrient deficiencies is a whole-food, plant-based diet, Rahman said. She advocates this nutritional approach, along with physical activity, for patients regardless of whether they’ve selected lifestyle intervention alone or combined with an AOM or bariatric surgery to address obesity.
Rahman cited a 5-year heart disease study comparing an intensive lifestyle program involving a vegetarian diet, aerobic exercise, stress management training, smoking cessation, and group psychosocial support to treatment as usual. Patients in the lifestyle group lost 10.9 kg at 1 year and sustained weight loss of 5.8 kg at 5 years, whereas weight in the control group remained relatively unchanged from baseline.
She also pointed to the findings of a study of patients with obesity or with overweight and at least one comorbidity that compared standard care with a low-fat, whole-food, plant-based diet with vitamin B12 supplementation. At 6 months, mean BMI reduction was greater in the intervention group than the standard care group (−4.4 vs −0.4).
In her practice, Rahman has seen the benefits of a whole-food, plant-based diet for patients with obesity.
If people are committed to this type of dietary approach and are given the tools and resources to do it effectively, “their thinking changes, their taste buds change, and they grow to enjoy this new way of eating,” she said. “They see results, and it’s a lifestyle that can be sustained long-term.”
Addressing Drivers of Weight Gain
Patients also need help addressing the various factors that may contribute to overweight and obesity, including overconsumption of ultra-processed foods, substandard nutritional quality of restaurant foods, increasing portion sizes, distraction during eating, emotional eating, late-night eating, and cultural/traditional values surrounding food, Rahman noted.
Supatra Tovar, PsyD, RD, a clinical psychologist with a practice in Pasadena, California, agreed that identifying the reasons for weight gain is critical for treatment.
“If you’re not addressing underlying issues, such as a person’s relationship with food, behaviors around food, the tendency to mindlessly eat or emotionally eat or eat to seek comfort, the person’s weight problems won’t ultimately be fully solved by any of the three approaches — dieting, medications, or bariatric surgery,” she said.
Some of her patients “engage in extreme dieting and deprivation, and many who use medications or have had bariatric surgery hardly eat and often develop nutritional deficiencies,” said Tovar, author of the book Deprogram Diet Culture: Rethink Your Relationship with Food, Heal Your Mind, and Live a Diet-Free Life.
The key to healthy and sustained weight loss is to “become attuned to the body’s signals, learn how to honor hunger, stop eating when satisfied, and eat more healthful foods, such as fruits and vegetables, whole grains, lean proteins — especially plant-based proteins — and the body gives signals that this is what it wants,” she said.
Tovar doesn’t give her clients a specific diet or set of portions.
“I teach them to listen to their bodies,” she said. “They’ve lost significant amounts of weight and continued to keep it off because they’ve done this kind of work.”
When Lifestyle Changes Aren’t Enough
For many patients, lifestyle interventions are insufficient to address the degree of overweight and obesity and common comorbidities, said W. Timothy Garvey, MD, associate director and professor, Department of Nutrition Sciences, School of Health Professions, University of Alabama at Birmingham.
“Of course, nutritional approaches are very important, not only for weight but also for general health-related reasons,” said Garvey, lead author of the 2016 American Association of Clinical Endocrinologists obesity guidelines. “We’ve seen that the Mediterranean and some plant-based diets can prevent progression from prediabetes to diabetes and improve other parameters that reflect metabolic health.”
However, it’s “not common that patients can follow these diets, lose weight, and keep it off,” Garvey cautioned. Up to 50% of weight that’s lost through lifestyle changes is typically regained by 1-year follow-up, with almost all remaining lost weight subsequently regained in the majority of individuals because the person “has to fight against pathophysiological process that drive weight regain,” he noted.
Weight-loss medications can address these pathophysiologic processes by “addressing interactions of satiety hormones with feeding centers in the brain, suppressing the appetite, and making it easier for patients to adhere to a reduced-calorie diet.”
Garvey views the weight-loss medications in the same light as drugs for diabetes and hypertension, in that people need to keep taking them to sustain the benefit.
There’s still a role for bariatric surgery because not everyone can tolerate the AOMs or achieve sufficient weight loss.
“Patients with very high BMI who have trouble ambulating might benefit from a combination of bariatric surgery and medication,” Garvey said.
While some side effects are associated with AOMs, being an “alarmist” about them can be detrimental to patients, he warned.
Rahman and Tovar are authors of books about weight loss. Reddy reported no relevant financial relationships. Garvey is a consultant on advisory boards for Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Pfizer, Fractyl Health, Alnylam Pharmaceuticals, Inogen, Zealand, Allurion, Carmot/Roche, Terns Pharmaceuticals, Neurocrine, Keros Therapeutics, and Regeneron. He is the site principal investigator for multi-centered clinical trials sponsored by his university and funded by Novo Nordisk, Eli Lilly, Epitomee, Neurovalens, and Pfizer. He serves as a consultant on the advisory board for the nonprofit Milken Foundation and is a member of the Data Monitoring Committee for phase 3 clinical trials conducted by Boehringer-Ingelheim and Eli Lilly.
A version of this article first appeared on Medscape.com.
Given that more than 100 million US adults have obesity, including 22 million with severe obesity, physicians regularly see patients with the condition in their practices.
Fortunately, doctors have more tools than ever to help their patients. But the question remains: Which method is the safest and most effective? Is it diet and lifestyle changes, one of the recently approved anti-obesity medications (AOMs), bariatric surgery, or a combination approach?
There are no head-to-head trials comparing these three approaches, said Vanita Rahman, MD, clinic director of the Barnard Medical Center, Washington, DC, at the International Conference on Nutrition in Medicine, sponsored by the Physicians Committee for Responsible Medicine.
Instead, doctors must evaluate the merits and drawbacks of each intervention and decide with their patients which treatment is best for them, she told Medscape Medical News. When she sees patients, Rahman shares the pertinent research with them, so they are able to make an informed choice.
Looking at the Options
In her presentation at the conference, Rahman summarized the guidelines issued by the American Heart Association/American College of Cardiology/The Obesity Society for Management of Overweight and Obesity in Adults and the American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines For Medical Care of Patients with Obesity, including lifestyle changes, AOMs, and bariatric surgery (Table 1).

As shown, the current clinical guidelines offer recommendations that consider such factors as the patient’s BMI and presence of one or more comorbidities. Generally, they begin with lifestyle changes for people with overweight, the possibility of an AOM for those with obesity, and bariatric surgery as an option for those with severe obesity-related complications.
“In obesity, we traditionally thought the process was ‘either-or’ — either lifestyle or surgery or medication — and somehow lifestyle is better,” Sheethal Reddy, PhD, a psychologist at the Bariatric Center at Emory University Hospital Midtown, Atlanta, told Medscape Medical News.
Now physicians often use a combination of methods, but lifestyle is foundational to all of them, she said.
“If you don’t make lifestyle changes, none of the approaches will ultimately be effective,” said Reddy, who also is an assistant professor in the Division of General and GI Surgery at Emory School of Medicine, Atlanta.
Lifestyle changes don’t just involve diet and nutrition but include physical exercise.
“Being sedentary affects everything — sleep quality, appetite regulation, and metabolism. Without sufficient exercise, the body isn’t functioning well enough to have a healthy metabolism,” Reddy said.
How Durable Are the Interventions?
Although bariatric surgery has demonstrated effectiveness in helping patients lose weight, many of them regain some or most of it, Rahman said.
A systematic review and meta-analysis found weight regain in 49% of patients who underwent bariatric surgery patients, with the highest prevalence after Roux-en-Y gastric bypass.
Another study of approximately 45,000 patients who underwent bariatric surgery found differences not only in the percentage of total weight loss among Roux-en-Y gastric bypass, sleeve gastrectomy, and adjustable gastric band procedures but also in how much of that weight stayed off between 1 and 5 years following the procedure (Table 2).

Weight regain also is a risk with AOMs, if they’re discontinued.
The STEP 1 trial tested the effectiveness of semaglutide — a glucagon-like peptide 1 (GLP-1) receptor agonist — as an adjunct to lifestyle intervention for weight loss in patients with obesity or with overweight and at least one comorbidity but not diabetes. Mean weight loss with semaglutide was 17.3% but that figure dropped 11.6 percentage points after treatment was discontinued.
Other studies also have found that patients regain weight after GLP-1 discontinuation.
Tirzepatide, a GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) combination, has shown efficacy with weight reduction, but patients experienced some weight regain upon discontinuation. In one study, patients experienced a mean weight loss of 20.9% after 36 weeks of tirzepatide. In the study’s subsequent 52-week double-blind, placebo-controlled period, patients who stopped taking the medication experienced a weight regain of 14%, whereas those who remained on the medication lost an additional 5.5% of weight.
GLP-1 and GLP-1/GIP medications do not address the factors that contribute to overweight and obesity, Rahman said. “They simply suppress the appetite; therefore, weight gain occurs after stopping them.”
Patients may stop taking anti-obesity drugs for a variety of reasons, including side effects. Rahman noted that the common side effects include nausea, vomiting, and constipation, whereas rare side effects include gastroparesis, gallbladder and biliary disease, thyroid cancer, and suicidal thoughts. GLP-1 and GLP-1/GIP medications also carry a risk for non-arteritic anterior ischemic optic neuropathy, she said.
Moreover, health insurance does not always cover these medications, which likely affects patient access to the drugs and compliance rates.
“Given the side effects and frequent lack of insurance coverage, significant questions remain about long-term safety and feasibility of these agents,” Rahman said.
What About Nutritional Approaches?
The lifestyle interventions in the semaglutide and tirzepatide studies included 500 kcal/d deficit diets, which is difficult for people to maintain, noted Rahman, who is the author of the book Simply Plant Based: Fabulous Food for a Healthy Life.
Additionally, bariatric surgery has been associated with long-term micronutrient deficiencies, including deficiencies in vitamins A, D, E, K, B1, and B12, as well as folate, iron, zinc, copper, selenium, and calcium, she said.
The best approach to food from a patient compliance standpoint and to avoid nutrient deficiencies is a whole-food, plant-based diet, Rahman said. She advocates this nutritional approach, along with physical activity, for patients regardless of whether they’ve selected lifestyle intervention alone or combined with an AOM or bariatric surgery to address obesity.
Rahman cited a 5-year heart disease study comparing an intensive lifestyle program involving a vegetarian diet, aerobic exercise, stress management training, smoking cessation, and group psychosocial support to treatment as usual. Patients in the lifestyle group lost 10.9 kg at 1 year and sustained weight loss of 5.8 kg at 5 years, whereas weight in the control group remained relatively unchanged from baseline.
She also pointed to the findings of a study of patients with obesity or with overweight and at least one comorbidity that compared standard care with a low-fat, whole-food, plant-based diet with vitamin B12 supplementation. At 6 months, mean BMI reduction was greater in the intervention group than the standard care group (−4.4 vs −0.4).
In her practice, Rahman has seen the benefits of a whole-food, plant-based diet for patients with obesity.
If people are committed to this type of dietary approach and are given the tools and resources to do it effectively, “their thinking changes, their taste buds change, and they grow to enjoy this new way of eating,” she said. “They see results, and it’s a lifestyle that can be sustained long-term.”
Addressing Drivers of Weight Gain
Patients also need help addressing the various factors that may contribute to overweight and obesity, including overconsumption of ultra-processed foods, substandard nutritional quality of restaurant foods, increasing portion sizes, distraction during eating, emotional eating, late-night eating, and cultural/traditional values surrounding food, Rahman noted.
Supatra Tovar, PsyD, RD, a clinical psychologist with a practice in Pasadena, California, agreed that identifying the reasons for weight gain is critical for treatment.
“If you’re not addressing underlying issues, such as a person’s relationship with food, behaviors around food, the tendency to mindlessly eat or emotionally eat or eat to seek comfort, the person’s weight problems won’t ultimately be fully solved by any of the three approaches — dieting, medications, or bariatric surgery,” she said.
Some of her patients “engage in extreme dieting and deprivation, and many who use medications or have had bariatric surgery hardly eat and often develop nutritional deficiencies,” said Tovar, author of the book Deprogram Diet Culture: Rethink Your Relationship with Food, Heal Your Mind, and Live a Diet-Free Life.
The key to healthy and sustained weight loss is to “become attuned to the body’s signals, learn how to honor hunger, stop eating when satisfied, and eat more healthful foods, such as fruits and vegetables, whole grains, lean proteins — especially plant-based proteins — and the body gives signals that this is what it wants,” she said.
Tovar doesn’t give her clients a specific diet or set of portions.
“I teach them to listen to their bodies,” she said. “They’ve lost significant amounts of weight and continued to keep it off because they’ve done this kind of work.”
When Lifestyle Changes Aren’t Enough
For many patients, lifestyle interventions are insufficient to address the degree of overweight and obesity and common comorbidities, said W. Timothy Garvey, MD, associate director and professor, Department of Nutrition Sciences, School of Health Professions, University of Alabama at Birmingham.
“Of course, nutritional approaches are very important, not only for weight but also for general health-related reasons,” said Garvey, lead author of the 2016 American Association of Clinical Endocrinologists obesity guidelines. “We’ve seen that the Mediterranean and some plant-based diets can prevent progression from prediabetes to diabetes and improve other parameters that reflect metabolic health.”
However, it’s “not common that patients can follow these diets, lose weight, and keep it off,” Garvey cautioned. Up to 50% of weight that’s lost through lifestyle changes is typically regained by 1-year follow-up, with almost all remaining lost weight subsequently regained in the majority of individuals because the person “has to fight against pathophysiological process that drive weight regain,” he noted.
Weight-loss medications can address these pathophysiologic processes by “addressing interactions of satiety hormones with feeding centers in the brain, suppressing the appetite, and making it easier for patients to adhere to a reduced-calorie diet.”
Garvey views the weight-loss medications in the same light as drugs for diabetes and hypertension, in that people need to keep taking them to sustain the benefit.
There’s still a role for bariatric surgery because not everyone can tolerate the AOMs or achieve sufficient weight loss.
“Patients with very high BMI who have trouble ambulating might benefit from a combination of bariatric surgery and medication,” Garvey said.
While some side effects are associated with AOMs, being an “alarmist” about them can be detrimental to patients, he warned.
Rahman and Tovar are authors of books about weight loss. Reddy reported no relevant financial relationships. Garvey is a consultant on advisory boards for Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Pfizer, Fractyl Health, Alnylam Pharmaceuticals, Inogen, Zealand, Allurion, Carmot/Roche, Terns Pharmaceuticals, Neurocrine, Keros Therapeutics, and Regeneron. He is the site principal investigator for multi-centered clinical trials sponsored by his university and funded by Novo Nordisk, Eli Lilly, Epitomee, Neurovalens, and Pfizer. He serves as a consultant on the advisory board for the nonprofit Milken Foundation and is a member of the Data Monitoring Committee for phase 3 clinical trials conducted by Boehringer-Ingelheim and Eli Lilly.
A version of this article first appeared on Medscape.com.
AI-Enhanced ECG Used to Predict Hypertension and Associated Risks
TOPLINE:
in addition to traditional markers.
METHODOLOGY:
- Researchers conducted a development and external validation prognostic cohort study in a secondary care setting to identify individuals at risk for incident hypertension.
- They developed AIRE-HTN, which was trained on a derivation cohort from the Beth Israel Deaconess Medical Center in Boston, involving 1,163,401 ECGs from 189,539 patients (mean age, 57.7 years; 52.1% women; 64.5% White individuals).
- External validation was conducted on 65,610 ECGs from a UK-based volunteer cohort, drawn from an equal number of patients (mean age, 65.4 years; 51.5% women; 96.3% White individuals).
- Incident hypertension was evaluated in 19,423 individuals without hypertension from the medical center cohort and in 35,806 individuals without hypertension from the UK cohort.
TAKEAWAY:
- AIRE-HTN predicted incident hypertension with a C-index of 0.70 (95% CI, 0.69-0.71) in both the cohorts. Those in the quartile with the highest AIRE-HTN scores had a fourfold increased risk for incident hypertension (P < .001).
- The model’s predictive accuracy was maintained in individuals without left ventricular hypertrophy and those with normal ECGs and baseline blood pressure, indicating its robustness.
- The model was significantly additive to traditional clinical markers, with a continuous net reclassification index of 0.44 for the medical center cohort and 0.32 for the UK cohort.
- AIRE-HTN was an independent predictor of cardiovascular death (hazard ratio per 1-SD increase in score [HR], 2.24), heart failure (HR, 2.60), myocardial infarction (HR, 3.13), ischemic stroke (HR, 1.23), and chronic kidney disease (HR, 1.89) in outpatients from the medical center cohort (all P < .001), with largely consistent findings in the UK cohort.
IN PRACTICE:
“Results of exploratory and phenotypic analyses suggest the biological plausibility of these findings. Enhanced predictability could influence surveillance programs and primordial prevention,” the authors wrote.
SOURCE:
The study was led by Arunashis Sau, PhD, and Joseph Barker, MRes, National Heart and Lung Institute, Imperial College London, England. It was published online on January 2, 2025, in JAMA Cardiology.
LIMITATIONS:
In one cohort, hypertension was defined using International Classification of Diseases codes, which may lack granularity and not align with contemporary guidelines. The findings were not validated against ambulatory monitoring standards. The performance of the model in different populations and clinical settings remains to be explored.
DISCLOSURES:
The authors acknowledged receiving support from Imperial’s British Heart Foundation Centre for Excellence Award and disclosed receiving support from the British Heart Foundation, the National Institute for Health Research Imperial College Biomedical Research Centre, the EJP RD Research Mobility Fellowship, the Medical Research Council, and the Sir Jules Thorn Charitable Trust. Some authors reported receiving grants, personal fees, advisory fees, or laboratory work fees outside the submitted work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
in addition to traditional markers.
METHODOLOGY:
- Researchers conducted a development and external validation prognostic cohort study in a secondary care setting to identify individuals at risk for incident hypertension.
- They developed AIRE-HTN, which was trained on a derivation cohort from the Beth Israel Deaconess Medical Center in Boston, involving 1,163,401 ECGs from 189,539 patients (mean age, 57.7 years; 52.1% women; 64.5% White individuals).
- External validation was conducted on 65,610 ECGs from a UK-based volunteer cohort, drawn from an equal number of patients (mean age, 65.4 years; 51.5% women; 96.3% White individuals).
- Incident hypertension was evaluated in 19,423 individuals without hypertension from the medical center cohort and in 35,806 individuals without hypertension from the UK cohort.
TAKEAWAY:
- AIRE-HTN predicted incident hypertension with a C-index of 0.70 (95% CI, 0.69-0.71) in both the cohorts. Those in the quartile with the highest AIRE-HTN scores had a fourfold increased risk for incident hypertension (P < .001).
- The model’s predictive accuracy was maintained in individuals without left ventricular hypertrophy and those with normal ECGs and baseline blood pressure, indicating its robustness.
- The model was significantly additive to traditional clinical markers, with a continuous net reclassification index of 0.44 for the medical center cohort and 0.32 for the UK cohort.
- AIRE-HTN was an independent predictor of cardiovascular death (hazard ratio per 1-SD increase in score [HR], 2.24), heart failure (HR, 2.60), myocardial infarction (HR, 3.13), ischemic stroke (HR, 1.23), and chronic kidney disease (HR, 1.89) in outpatients from the medical center cohort (all P < .001), with largely consistent findings in the UK cohort.
IN PRACTICE:
“Results of exploratory and phenotypic analyses suggest the biological plausibility of these findings. Enhanced predictability could influence surveillance programs and primordial prevention,” the authors wrote.
SOURCE:
The study was led by Arunashis Sau, PhD, and Joseph Barker, MRes, National Heart and Lung Institute, Imperial College London, England. It was published online on January 2, 2025, in JAMA Cardiology.
LIMITATIONS:
In one cohort, hypertension was defined using International Classification of Diseases codes, which may lack granularity and not align with contemporary guidelines. The findings were not validated against ambulatory monitoring standards. The performance of the model in different populations and clinical settings remains to be explored.
DISCLOSURES:
The authors acknowledged receiving support from Imperial’s British Heart Foundation Centre for Excellence Award and disclosed receiving support from the British Heart Foundation, the National Institute for Health Research Imperial College Biomedical Research Centre, the EJP RD Research Mobility Fellowship, the Medical Research Council, and the Sir Jules Thorn Charitable Trust. Some authors reported receiving grants, personal fees, advisory fees, or laboratory work fees outside the submitted work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
in addition to traditional markers.
METHODOLOGY:
- Researchers conducted a development and external validation prognostic cohort study in a secondary care setting to identify individuals at risk for incident hypertension.
- They developed AIRE-HTN, which was trained on a derivation cohort from the Beth Israel Deaconess Medical Center in Boston, involving 1,163,401 ECGs from 189,539 patients (mean age, 57.7 years; 52.1% women; 64.5% White individuals).
- External validation was conducted on 65,610 ECGs from a UK-based volunteer cohort, drawn from an equal number of patients (mean age, 65.4 years; 51.5% women; 96.3% White individuals).
- Incident hypertension was evaluated in 19,423 individuals without hypertension from the medical center cohort and in 35,806 individuals without hypertension from the UK cohort.
TAKEAWAY:
- AIRE-HTN predicted incident hypertension with a C-index of 0.70 (95% CI, 0.69-0.71) in both the cohorts. Those in the quartile with the highest AIRE-HTN scores had a fourfold increased risk for incident hypertension (P < .001).
- The model’s predictive accuracy was maintained in individuals without left ventricular hypertrophy and those with normal ECGs and baseline blood pressure, indicating its robustness.
- The model was significantly additive to traditional clinical markers, with a continuous net reclassification index of 0.44 for the medical center cohort and 0.32 for the UK cohort.
- AIRE-HTN was an independent predictor of cardiovascular death (hazard ratio per 1-SD increase in score [HR], 2.24), heart failure (HR, 2.60), myocardial infarction (HR, 3.13), ischemic stroke (HR, 1.23), and chronic kidney disease (HR, 1.89) in outpatients from the medical center cohort (all P < .001), with largely consistent findings in the UK cohort.
IN PRACTICE:
“Results of exploratory and phenotypic analyses suggest the biological plausibility of these findings. Enhanced predictability could influence surveillance programs and primordial prevention,” the authors wrote.
SOURCE:
The study was led by Arunashis Sau, PhD, and Joseph Barker, MRes, National Heart and Lung Institute, Imperial College London, England. It was published online on January 2, 2025, in JAMA Cardiology.
LIMITATIONS:
In one cohort, hypertension was defined using International Classification of Diseases codes, which may lack granularity and not align with contemporary guidelines. The findings were not validated against ambulatory monitoring standards. The performance of the model in different populations and clinical settings remains to be explored.
DISCLOSURES:
The authors acknowledged receiving support from Imperial’s British Heart Foundation Centre for Excellence Award and disclosed receiving support from the British Heart Foundation, the National Institute for Health Research Imperial College Biomedical Research Centre, the EJP RD Research Mobility Fellowship, the Medical Research Council, and the Sir Jules Thorn Charitable Trust. Some authors reported receiving grants, personal fees, advisory fees, or laboratory work fees outside the submitted work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Intensive BP Control May Benefit CKD Patients in Real World
TOPLINE:
The cardiovascular benefits observed with intensive blood pressure (BP) control in patients with hypertension and elevated cardiovascular risk from the Systolic Blood Pressure Intervention Trial (SPRINT) can be largely replicated in real-world settings among patients with chronic kidney disease (CKD), highlighting the advantages of adopting intensive BP targets.
METHODOLOGY:
- Researchers conducted a comparative effectiveness study to determine if the beneficial and adverse effects of intensive vs standard BP control observed in SPRINT were replicable in patients with CKD and hypertension in clinical practice.
- They identified 85,938 patients (mean age, 75.7 years; 95.0% men) and 13,983 patients (mean age, 77.4 years; 38.4% men) from the Veterans Health Administration (VHA) and Kaiser Permanente of Southern California (KPSC) databases, respectively.
- The treatment effect was estimated by combining baseline covariate, treatment, and outcome data of participants from the SPRINT with covariate data from the VHA and KPSC databases.
- The primary outcomes included major cardiovascular events, all-cause death, cognitive impairment, CKD progression, and adverse events at 4 years.
TAKEAWAY:
- Compared with SPRINT participants, those in the VHA and KPSC databases were older, had less prevalent cardiovascular disease, higher albuminuria, and used more statins.
- The benefits of intensive vs standard BP control on major cardiovascular events, all-cause mortality, and certain adverse events (hypotension, syncope, bradycardia, acute kidney injury, and electrolyte abnormality) were transferable from the trial to the VHA and KPSC populations.
- The treatment effect of intensive BP management on CKD progression was transportable to the KPSC population but not to the VHA population. However, the trial’s impact on cognitive outcomes, such as dementia, was not transportable to either the VHA or KPSC populations.
- On the absolute scale, intensive vs standard BP treatment showed greater cardiovascular benefits and fewer safety concerns in the VHA and KPSC populations than in the SPRINT.
IN PRACTICE:
“This example highlights the potential for transportability methods to provide insights that can bridge evidence gaps and inform the application of novel therapies to patients with CKD who are treated in everyday practice,” the authors wrote.
SOURCE:
This study was led by Manjula Kurella Tamura, MD, MPH, Division of Nephrology, Department of Medicine, Stanford University School of Medicine, Palo Alto, California. It was published online on January 7, 2025, in JAMA Network Open.
LIMITATIONS:
Transportability analyses could not account for characteristics that were not well-documented in electronic health records, such as limited life expectancy. The study was conducted before the widespread use of sodium-glucose cotransporter 2 inhibitors, glucagon-like peptide 1 receptor agonists, and nonsteroidal mineralocorticoid receptor antagonists, making it unclear whether intensive BP treatment would result in similar benefits with current pharmacotherapy regimens. Eligibility for this study was based on BP measurements in routine practice, which tend to be more variable than those collected in research settings.
DISCLOSURES:
This study was supported by grants from the National Institutes of Health. Some authors disclosed serving as a consultant and receiving grants, personal fees, and consulting fees from pharmaceutical companies and other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
The cardiovascular benefits observed with intensive blood pressure (BP) control in patients with hypertension and elevated cardiovascular risk from the Systolic Blood Pressure Intervention Trial (SPRINT) can be largely replicated in real-world settings among patients with chronic kidney disease (CKD), highlighting the advantages of adopting intensive BP targets.
METHODOLOGY:
- Researchers conducted a comparative effectiveness study to determine if the beneficial and adverse effects of intensive vs standard BP control observed in SPRINT were replicable in patients with CKD and hypertension in clinical practice.
- They identified 85,938 patients (mean age, 75.7 years; 95.0% men) and 13,983 patients (mean age, 77.4 years; 38.4% men) from the Veterans Health Administration (VHA) and Kaiser Permanente of Southern California (KPSC) databases, respectively.
- The treatment effect was estimated by combining baseline covariate, treatment, and outcome data of participants from the SPRINT with covariate data from the VHA and KPSC databases.
- The primary outcomes included major cardiovascular events, all-cause death, cognitive impairment, CKD progression, and adverse events at 4 years.
TAKEAWAY:
- Compared with SPRINT participants, those in the VHA and KPSC databases were older, had less prevalent cardiovascular disease, higher albuminuria, and used more statins.
- The benefits of intensive vs standard BP control on major cardiovascular events, all-cause mortality, and certain adverse events (hypotension, syncope, bradycardia, acute kidney injury, and electrolyte abnormality) were transferable from the trial to the VHA and KPSC populations.
- The treatment effect of intensive BP management on CKD progression was transportable to the KPSC population but not to the VHA population. However, the trial’s impact on cognitive outcomes, such as dementia, was not transportable to either the VHA or KPSC populations.
- On the absolute scale, intensive vs standard BP treatment showed greater cardiovascular benefits and fewer safety concerns in the VHA and KPSC populations than in the SPRINT.
IN PRACTICE:
“This example highlights the potential for transportability methods to provide insights that can bridge evidence gaps and inform the application of novel therapies to patients with CKD who are treated in everyday practice,” the authors wrote.
SOURCE:
This study was led by Manjula Kurella Tamura, MD, MPH, Division of Nephrology, Department of Medicine, Stanford University School of Medicine, Palo Alto, California. It was published online on January 7, 2025, in JAMA Network Open.
LIMITATIONS:
Transportability analyses could not account for characteristics that were not well-documented in electronic health records, such as limited life expectancy. The study was conducted before the widespread use of sodium-glucose cotransporter 2 inhibitors, glucagon-like peptide 1 receptor agonists, and nonsteroidal mineralocorticoid receptor antagonists, making it unclear whether intensive BP treatment would result in similar benefits with current pharmacotherapy regimens. Eligibility for this study was based on BP measurements in routine practice, which tend to be more variable than those collected in research settings.
DISCLOSURES:
This study was supported by grants from the National Institutes of Health. Some authors disclosed serving as a consultant and receiving grants, personal fees, and consulting fees from pharmaceutical companies and other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
The cardiovascular benefits observed with intensive blood pressure (BP) control in patients with hypertension and elevated cardiovascular risk from the Systolic Blood Pressure Intervention Trial (SPRINT) can be largely replicated in real-world settings among patients with chronic kidney disease (CKD), highlighting the advantages of adopting intensive BP targets.
METHODOLOGY:
- Researchers conducted a comparative effectiveness study to determine if the beneficial and adverse effects of intensive vs standard BP control observed in SPRINT were replicable in patients with CKD and hypertension in clinical practice.
- They identified 85,938 patients (mean age, 75.7 years; 95.0% men) and 13,983 patients (mean age, 77.4 years; 38.4% men) from the Veterans Health Administration (VHA) and Kaiser Permanente of Southern California (KPSC) databases, respectively.
- The treatment effect was estimated by combining baseline covariate, treatment, and outcome data of participants from the SPRINT with covariate data from the VHA and KPSC databases.
- The primary outcomes included major cardiovascular events, all-cause death, cognitive impairment, CKD progression, and adverse events at 4 years.
TAKEAWAY:
- Compared with SPRINT participants, those in the VHA and KPSC databases were older, had less prevalent cardiovascular disease, higher albuminuria, and used more statins.
- The benefits of intensive vs standard BP control on major cardiovascular events, all-cause mortality, and certain adverse events (hypotension, syncope, bradycardia, acute kidney injury, and electrolyte abnormality) were transferable from the trial to the VHA and KPSC populations.
- The treatment effect of intensive BP management on CKD progression was transportable to the KPSC population but not to the VHA population. However, the trial’s impact on cognitive outcomes, such as dementia, was not transportable to either the VHA or KPSC populations.
- On the absolute scale, intensive vs standard BP treatment showed greater cardiovascular benefits and fewer safety concerns in the VHA and KPSC populations than in the SPRINT.
IN PRACTICE:
“This example highlights the potential for transportability methods to provide insights that can bridge evidence gaps and inform the application of novel therapies to patients with CKD who are treated in everyday practice,” the authors wrote.
SOURCE:
This study was led by Manjula Kurella Tamura, MD, MPH, Division of Nephrology, Department of Medicine, Stanford University School of Medicine, Palo Alto, California. It was published online on January 7, 2025, in JAMA Network Open.
LIMITATIONS:
Transportability analyses could not account for characteristics that were not well-documented in electronic health records, such as limited life expectancy. The study was conducted before the widespread use of sodium-glucose cotransporter 2 inhibitors, glucagon-like peptide 1 receptor agonists, and nonsteroidal mineralocorticoid receptor antagonists, making it unclear whether intensive BP treatment would result in similar benefits with current pharmacotherapy regimens. Eligibility for this study was based on BP measurements in routine practice, which tend to be more variable than those collected in research settings.
DISCLOSURES:
This study was supported by grants from the National Institutes of Health. Some authors disclosed serving as a consultant and receiving grants, personal fees, and consulting fees from pharmaceutical companies and other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Dry January: Should Doctors Make It Year-Round?
For millennia in medicine, alcohol, particularly red wine, carried a health halo; in small doses, it has historically been thought to have cardioprotective benefits. Michael Farkouh, MD, a professor of cardiology at Cedars-Sinai, estimates half the physicians still accept people having a drink or two a day. “That is still in practice, though the numbers are reducing,” he said.
But Farkouh no longer drinks alcohol, a position he has come to after getting more involved in research into the substance and his realization that many of the studies touting alcohol’s health benefits were flawed.
Today, alcohol sits alongside asbestos and tobacco as class 1 carcinogens. According to the World Health Organization, it has no known safe ingestible amount. In 2018, a blockbuster report in The Lancet found no amount of any kind of alcohol improves health. In early January, US Surgeon General Vivek Murthy called for adding cancer warnings to alcohol labels.
But the way doctors drink is far from black and white. For physicians, drinking habits are tied up in personal values, professional understanding of a substance with a confusing research history, and the fact alcohol is deeply ingrained in the social fabric of society — and in medicine. As thinking on alcohol shifts, this news organization spoke with physicians about their own drinking habits, how they counsel patients on it, and alcohol’s place in a field that works to keep people healthy.
Cultural Currency
From the days of Hippocrates, who believed alcohol could cure virtually every ailment, alcohol has held a large role in medicine. Through much of the 19th century, patent remedies like Hamlin’s Wizard Oil and the Seven Sutherland Sisters Hair Grower, contained alcohol — sometimes in concentrations exceeding 50%.
The first American Pharmacopoeia, published in 1820, even contained nine wine-based medicines. Throughout the second half of the 19th century, physicians largely debated alcohol’s role in medicine. However, a 1922 poll of members of the American Medical Association found that physicians were still using alcohol as a medicine for everything from heart attacks to animal bites.
Today, alcohol’s presence in medicine is, in some ways, representative of a realized cognitive dissonance.
“In my mind, alcohol has completely lost any illusion of benefit. It is a poison to almost every single organ in our body. Yet I’m currently engaged in a duel of being a physician who drinks in moderation and constantly judging myself for it,” said Tyra Fainstad, MD, an internist and an associate professor at CU Medicine in Denver.
Fainstad said every academic national conference she has attended has had a reception with multiple cash bars — and professional recruitment dinners regularly include at least the offering of alcohol. Private hospitals often have open bars at events.
“Drinking has historically been a way that people unwind, even in medicine,” said addiction psychiatrist Alexis Ritvo, MD. Ritvo — who said she drinks occasionally but much less than she used to after paying attention to how alcohol makes her feel and the harm alcohol can cause — noted that some occasions where alcohol is present socially in medicine don’t bother her. Alcohol is even an option at the addiction psychiatry conference, where attendees can exchange tickets for drinks. But last year, the event provided separate bars for alcoholic and nonalcoholic drinks.
“Our life is full of things that are contradictory or at odds,” Ritvo said. “We want things to either be wrong or right, appropriate or inappropriate, but just like all things, everything’s pretty nuanced.”
But there are examples of alcohol being a part of an event that are downright inappropriate, such as when she attended a fundraiser for a recovery facility that had an open bar.
Farkouh said alcohol at events can exclude others. (He recommends that instead of calling a social gathering “going out for drinks,” someone might say, “We’re getting together.”) He drinks mocktails or nonalcoholic beer at work events where alcohol is served.
Brian Dwinnell, MD, associate dean of student life at CU Medicine, said alcohol can quickly become the focus of an event — something he noticed at an annual kickball game between first- and second-year students that has historically served beer.
In recent years, school leadership has removed alcohol from his institution’s match day celebration and the kickball game. “Initially, there was some pushback from students,” he said of making these events dry, “but now, it’s just sort of accepted, and the events have been just as great as they were when we did provide alcohol.”
How Doctors Drink
Physicians may have a greater understanding of alcohol’s health harms. Still, they don’t necessarily drink less because of it, and whether they should becomes a question not just of health but also of the standards to which society holds medical professionals.
Data suggest physicians tend to drink at rates similar to those among the general population. A recent Medscape Medical News survey found nearly 60% of physicians have started drinking less.
Dwinnell said he is a long-time “wine connoisseur” and drinks on occasion. But he admitted that while he thinks about the health implications of alcohol more — and he has nixed it from various events for medical students — he does not believe his drinking habits have changed much.
Navya Mysore, MD, a family physician in New York City, said she has become interested in wine over the past few years, even taking classes to learn more about it. “I like understanding how it’s made, the regions it’s from, and how to pair it with food,” she said. Mysore admits she drank a little more than usual throughout the pandemic, yet today, she said her relationship with alcohol in moderation is related to family, community, and connection.
Fainstad, who drinks socially, said: “I think there’s an immeasurable quality to the social ritual of it. I think for better or worse — probably for worse — for many generations, alcohol has been a part of many meaningful traditions and rituals that we hold.”
Farkouh was quick to underscore the importance of social connection, and that alcohol reduces stress for some people. “I don’t want to take that away from people,” he said. But he also stated the importance of finding other ways to find social fulfillment and enjoyment — and said it’s essential for societal norms to shift to reflect this.
With emerging data, alcohol’s image in society is shifting. Ireland recently became the first country to pass regulation requiring all alcohol sold there to come with a cancer warning. All the clinicians interviewed for this article spoke about the increased acceptability of choosing not to drink for whatever reason.
In the context of alcohol, Dwinnell often asks his students, “What if you were out at a restaurant and you saw your mother’s surgeon there and they were intoxicated? Are you going to feel comfortable with that individual operating on your mother tomorrow or any time?” He added: “Physicians are held to a higher professional standard than those in other fields — and they should be. This is a high-stakes business.”
Dwinnell’s hypothetical question to students is a good one, albeit perhaps not always a fair one. “It’s important for people to realize that physicians are humans,” Mysore said. “We are people, we have lives, and we may choose to have habits that are not necessarily the healthiest for us.”
Fainstad said there’s no shame in medical professionals drinking on occasion. “You can’t be held accountable for something you don’t know about,” she said, acknowledging the known harms of alcohol and that there is still more to learn. But she does wonder how doctors who drank might be perceived in years to come. “I can imagine in a couple of decades, people could say, ‘Even doctors used to have a glass of wine with dinner.’”
‘Physicians Should Tighten Their Stances on Alcohol’
The 2020-2025 Dietary Guidelines suggest limiting intake to two alcoholic drinks or less daily for men and one drink or less for women or to choose not to drink. Farkouh said he skews toward the latter, encouraging patients to drink as little as possible or nothing at all. “If you take a holistic approach, physicians should tighten their stances on alcohol,” he said.
Ultimately, he said a randomized trial is warranted to address the risk for cardiovascular disease, in particular.
Of course, physicians vary in how they discuss the topic with patients.
Mysore said she regularly educates patients about pour size and ways to swap out alcoholic drinks with nonalcoholic ones. Outside of cases of addiction, she favors the idea of moderation. “I don’t really subscribe to all-or-nothing mindsets. If there’s something that you enjoy having as a part of your life, I don’t think there’s any reason why you need to eliminate it,” she said. “You just need to figure out what moderation looks like for you.”
Ritvo favors motivational interviewing and tries to understand someone’s relationship with alcohol.
Fainstad provides the Dietary Guidelines’ cutoffs to patients and educates them on the poisonous nature of the substance.
Clearer guidance from large governing bodies — potential changes around alcohol in the 2025-230 revision of the US Dietary Guidelines or cancer warnings on booze sold in the United States — are coming and could help streamline messaging.
And although he speaks with urgency about alcohol’s dangers, Farkouh emphasized the need for a judgment-free and patient-centered approach to conversations around drinking: “People have grown up with alcohol being acceptable, and it’s going to take time to change that.”
A version of this article first appeared on Medscape.com.
For millennia in medicine, alcohol, particularly red wine, carried a health halo; in small doses, it has historically been thought to have cardioprotective benefits. Michael Farkouh, MD, a professor of cardiology at Cedars-Sinai, estimates half the physicians still accept people having a drink or two a day. “That is still in practice, though the numbers are reducing,” he said.
But Farkouh no longer drinks alcohol, a position he has come to after getting more involved in research into the substance and his realization that many of the studies touting alcohol’s health benefits were flawed.
Today, alcohol sits alongside asbestos and tobacco as class 1 carcinogens. According to the World Health Organization, it has no known safe ingestible amount. In 2018, a blockbuster report in The Lancet found no amount of any kind of alcohol improves health. In early January, US Surgeon General Vivek Murthy called for adding cancer warnings to alcohol labels.
But the way doctors drink is far from black and white. For physicians, drinking habits are tied up in personal values, professional understanding of a substance with a confusing research history, and the fact alcohol is deeply ingrained in the social fabric of society — and in medicine. As thinking on alcohol shifts, this news organization spoke with physicians about their own drinking habits, how they counsel patients on it, and alcohol’s place in a field that works to keep people healthy.
Cultural Currency
From the days of Hippocrates, who believed alcohol could cure virtually every ailment, alcohol has held a large role in medicine. Through much of the 19th century, patent remedies like Hamlin’s Wizard Oil and the Seven Sutherland Sisters Hair Grower, contained alcohol — sometimes in concentrations exceeding 50%.
The first American Pharmacopoeia, published in 1820, even contained nine wine-based medicines. Throughout the second half of the 19th century, physicians largely debated alcohol’s role in medicine. However, a 1922 poll of members of the American Medical Association found that physicians were still using alcohol as a medicine for everything from heart attacks to animal bites.
Today, alcohol’s presence in medicine is, in some ways, representative of a realized cognitive dissonance.
“In my mind, alcohol has completely lost any illusion of benefit. It is a poison to almost every single organ in our body. Yet I’m currently engaged in a duel of being a physician who drinks in moderation and constantly judging myself for it,” said Tyra Fainstad, MD, an internist and an associate professor at CU Medicine in Denver.
Fainstad said every academic national conference she has attended has had a reception with multiple cash bars — and professional recruitment dinners regularly include at least the offering of alcohol. Private hospitals often have open bars at events.
“Drinking has historically been a way that people unwind, even in medicine,” said addiction psychiatrist Alexis Ritvo, MD. Ritvo — who said she drinks occasionally but much less than she used to after paying attention to how alcohol makes her feel and the harm alcohol can cause — noted that some occasions where alcohol is present socially in medicine don’t bother her. Alcohol is even an option at the addiction psychiatry conference, where attendees can exchange tickets for drinks. But last year, the event provided separate bars for alcoholic and nonalcoholic drinks.
“Our life is full of things that are contradictory or at odds,” Ritvo said. “We want things to either be wrong or right, appropriate or inappropriate, but just like all things, everything’s pretty nuanced.”
But there are examples of alcohol being a part of an event that are downright inappropriate, such as when she attended a fundraiser for a recovery facility that had an open bar.
Farkouh said alcohol at events can exclude others. (He recommends that instead of calling a social gathering “going out for drinks,” someone might say, “We’re getting together.”) He drinks mocktails or nonalcoholic beer at work events where alcohol is served.
Brian Dwinnell, MD, associate dean of student life at CU Medicine, said alcohol can quickly become the focus of an event — something he noticed at an annual kickball game between first- and second-year students that has historically served beer.
In recent years, school leadership has removed alcohol from his institution’s match day celebration and the kickball game. “Initially, there was some pushback from students,” he said of making these events dry, “but now, it’s just sort of accepted, and the events have been just as great as they were when we did provide alcohol.”
How Doctors Drink
Physicians may have a greater understanding of alcohol’s health harms. Still, they don’t necessarily drink less because of it, and whether they should becomes a question not just of health but also of the standards to which society holds medical professionals.
Data suggest physicians tend to drink at rates similar to those among the general population. A recent Medscape Medical News survey found nearly 60% of physicians have started drinking less.
Dwinnell said he is a long-time “wine connoisseur” and drinks on occasion. But he admitted that while he thinks about the health implications of alcohol more — and he has nixed it from various events for medical students — he does not believe his drinking habits have changed much.
Navya Mysore, MD, a family physician in New York City, said she has become interested in wine over the past few years, even taking classes to learn more about it. “I like understanding how it’s made, the regions it’s from, and how to pair it with food,” she said. Mysore admits she drank a little more than usual throughout the pandemic, yet today, she said her relationship with alcohol in moderation is related to family, community, and connection.
Fainstad, who drinks socially, said: “I think there’s an immeasurable quality to the social ritual of it. I think for better or worse — probably for worse — for many generations, alcohol has been a part of many meaningful traditions and rituals that we hold.”
Farkouh was quick to underscore the importance of social connection, and that alcohol reduces stress for some people. “I don’t want to take that away from people,” he said. But he also stated the importance of finding other ways to find social fulfillment and enjoyment — and said it’s essential for societal norms to shift to reflect this.
With emerging data, alcohol’s image in society is shifting. Ireland recently became the first country to pass regulation requiring all alcohol sold there to come with a cancer warning. All the clinicians interviewed for this article spoke about the increased acceptability of choosing not to drink for whatever reason.
In the context of alcohol, Dwinnell often asks his students, “What if you were out at a restaurant and you saw your mother’s surgeon there and they were intoxicated? Are you going to feel comfortable with that individual operating on your mother tomorrow or any time?” He added: “Physicians are held to a higher professional standard than those in other fields — and they should be. This is a high-stakes business.”
Dwinnell’s hypothetical question to students is a good one, albeit perhaps not always a fair one. “It’s important for people to realize that physicians are humans,” Mysore said. “We are people, we have lives, and we may choose to have habits that are not necessarily the healthiest for us.”
Fainstad said there’s no shame in medical professionals drinking on occasion. “You can’t be held accountable for something you don’t know about,” she said, acknowledging the known harms of alcohol and that there is still more to learn. But she does wonder how doctors who drank might be perceived in years to come. “I can imagine in a couple of decades, people could say, ‘Even doctors used to have a glass of wine with dinner.’”
‘Physicians Should Tighten Their Stances on Alcohol’
The 2020-2025 Dietary Guidelines suggest limiting intake to two alcoholic drinks or less daily for men and one drink or less for women or to choose not to drink. Farkouh said he skews toward the latter, encouraging patients to drink as little as possible or nothing at all. “If you take a holistic approach, physicians should tighten their stances on alcohol,” he said.
Ultimately, he said a randomized trial is warranted to address the risk for cardiovascular disease, in particular.
Of course, physicians vary in how they discuss the topic with patients.
Mysore said she regularly educates patients about pour size and ways to swap out alcoholic drinks with nonalcoholic ones. Outside of cases of addiction, she favors the idea of moderation. “I don’t really subscribe to all-or-nothing mindsets. If there’s something that you enjoy having as a part of your life, I don’t think there’s any reason why you need to eliminate it,” she said. “You just need to figure out what moderation looks like for you.”
Ritvo favors motivational interviewing and tries to understand someone’s relationship with alcohol.
Fainstad provides the Dietary Guidelines’ cutoffs to patients and educates them on the poisonous nature of the substance.
Clearer guidance from large governing bodies — potential changes around alcohol in the 2025-230 revision of the US Dietary Guidelines or cancer warnings on booze sold in the United States — are coming and could help streamline messaging.
And although he speaks with urgency about alcohol’s dangers, Farkouh emphasized the need for a judgment-free and patient-centered approach to conversations around drinking: “People have grown up with alcohol being acceptable, and it’s going to take time to change that.”
A version of this article first appeared on Medscape.com.
For millennia in medicine, alcohol, particularly red wine, carried a health halo; in small doses, it has historically been thought to have cardioprotective benefits. Michael Farkouh, MD, a professor of cardiology at Cedars-Sinai, estimates half the physicians still accept people having a drink or two a day. “That is still in practice, though the numbers are reducing,” he said.
But Farkouh no longer drinks alcohol, a position he has come to after getting more involved in research into the substance and his realization that many of the studies touting alcohol’s health benefits were flawed.
Today, alcohol sits alongside asbestos and tobacco as class 1 carcinogens. According to the World Health Organization, it has no known safe ingestible amount. In 2018, a blockbuster report in The Lancet found no amount of any kind of alcohol improves health. In early January, US Surgeon General Vivek Murthy called for adding cancer warnings to alcohol labels.
But the way doctors drink is far from black and white. For physicians, drinking habits are tied up in personal values, professional understanding of a substance with a confusing research history, and the fact alcohol is deeply ingrained in the social fabric of society — and in medicine. As thinking on alcohol shifts, this news organization spoke with physicians about their own drinking habits, how they counsel patients on it, and alcohol’s place in a field that works to keep people healthy.
Cultural Currency
From the days of Hippocrates, who believed alcohol could cure virtually every ailment, alcohol has held a large role in medicine. Through much of the 19th century, patent remedies like Hamlin’s Wizard Oil and the Seven Sutherland Sisters Hair Grower, contained alcohol — sometimes in concentrations exceeding 50%.
The first American Pharmacopoeia, published in 1820, even contained nine wine-based medicines. Throughout the second half of the 19th century, physicians largely debated alcohol’s role in medicine. However, a 1922 poll of members of the American Medical Association found that physicians were still using alcohol as a medicine for everything from heart attacks to animal bites.
Today, alcohol’s presence in medicine is, in some ways, representative of a realized cognitive dissonance.
“In my mind, alcohol has completely lost any illusion of benefit. It is a poison to almost every single organ in our body. Yet I’m currently engaged in a duel of being a physician who drinks in moderation and constantly judging myself for it,” said Tyra Fainstad, MD, an internist and an associate professor at CU Medicine in Denver.
Fainstad said every academic national conference she has attended has had a reception with multiple cash bars — and professional recruitment dinners regularly include at least the offering of alcohol. Private hospitals often have open bars at events.
“Drinking has historically been a way that people unwind, even in medicine,” said addiction psychiatrist Alexis Ritvo, MD. Ritvo — who said she drinks occasionally but much less than she used to after paying attention to how alcohol makes her feel and the harm alcohol can cause — noted that some occasions where alcohol is present socially in medicine don’t bother her. Alcohol is even an option at the addiction psychiatry conference, where attendees can exchange tickets for drinks. But last year, the event provided separate bars for alcoholic and nonalcoholic drinks.
“Our life is full of things that are contradictory or at odds,” Ritvo said. “We want things to either be wrong or right, appropriate or inappropriate, but just like all things, everything’s pretty nuanced.”
But there are examples of alcohol being a part of an event that are downright inappropriate, such as when she attended a fundraiser for a recovery facility that had an open bar.
Farkouh said alcohol at events can exclude others. (He recommends that instead of calling a social gathering “going out for drinks,” someone might say, “We’re getting together.”) He drinks mocktails or nonalcoholic beer at work events where alcohol is served.
Brian Dwinnell, MD, associate dean of student life at CU Medicine, said alcohol can quickly become the focus of an event — something he noticed at an annual kickball game between first- and second-year students that has historically served beer.
In recent years, school leadership has removed alcohol from his institution’s match day celebration and the kickball game. “Initially, there was some pushback from students,” he said of making these events dry, “but now, it’s just sort of accepted, and the events have been just as great as they were when we did provide alcohol.”
How Doctors Drink
Physicians may have a greater understanding of alcohol’s health harms. Still, they don’t necessarily drink less because of it, and whether they should becomes a question not just of health but also of the standards to which society holds medical professionals.
Data suggest physicians tend to drink at rates similar to those among the general population. A recent Medscape Medical News survey found nearly 60% of physicians have started drinking less.
Dwinnell said he is a long-time “wine connoisseur” and drinks on occasion. But he admitted that while he thinks about the health implications of alcohol more — and he has nixed it from various events for medical students — he does not believe his drinking habits have changed much.
Navya Mysore, MD, a family physician in New York City, said she has become interested in wine over the past few years, even taking classes to learn more about it. “I like understanding how it’s made, the regions it’s from, and how to pair it with food,” she said. Mysore admits she drank a little more than usual throughout the pandemic, yet today, she said her relationship with alcohol in moderation is related to family, community, and connection.
Fainstad, who drinks socially, said: “I think there’s an immeasurable quality to the social ritual of it. I think for better or worse — probably for worse — for many generations, alcohol has been a part of many meaningful traditions and rituals that we hold.”
Farkouh was quick to underscore the importance of social connection, and that alcohol reduces stress for some people. “I don’t want to take that away from people,” he said. But he also stated the importance of finding other ways to find social fulfillment and enjoyment — and said it’s essential for societal norms to shift to reflect this.
With emerging data, alcohol’s image in society is shifting. Ireland recently became the first country to pass regulation requiring all alcohol sold there to come with a cancer warning. All the clinicians interviewed for this article spoke about the increased acceptability of choosing not to drink for whatever reason.
In the context of alcohol, Dwinnell often asks his students, “What if you were out at a restaurant and you saw your mother’s surgeon there and they were intoxicated? Are you going to feel comfortable with that individual operating on your mother tomorrow or any time?” He added: “Physicians are held to a higher professional standard than those in other fields — and they should be. This is a high-stakes business.”
Dwinnell’s hypothetical question to students is a good one, albeit perhaps not always a fair one. “It’s important for people to realize that physicians are humans,” Mysore said. “We are people, we have lives, and we may choose to have habits that are not necessarily the healthiest for us.”
Fainstad said there’s no shame in medical professionals drinking on occasion. “You can’t be held accountable for something you don’t know about,” she said, acknowledging the known harms of alcohol and that there is still more to learn. But she does wonder how doctors who drank might be perceived in years to come. “I can imagine in a couple of decades, people could say, ‘Even doctors used to have a glass of wine with dinner.’”
‘Physicians Should Tighten Their Stances on Alcohol’
The 2020-2025 Dietary Guidelines suggest limiting intake to two alcoholic drinks or less daily for men and one drink or less for women or to choose not to drink. Farkouh said he skews toward the latter, encouraging patients to drink as little as possible or nothing at all. “If you take a holistic approach, physicians should tighten their stances on alcohol,” he said.
Ultimately, he said a randomized trial is warranted to address the risk for cardiovascular disease, in particular.
Of course, physicians vary in how they discuss the topic with patients.
Mysore said she regularly educates patients about pour size and ways to swap out alcoholic drinks with nonalcoholic ones. Outside of cases of addiction, she favors the idea of moderation. “I don’t really subscribe to all-or-nothing mindsets. If there’s something that you enjoy having as a part of your life, I don’t think there’s any reason why you need to eliminate it,” she said. “You just need to figure out what moderation looks like for you.”
Ritvo favors motivational interviewing and tries to understand someone’s relationship with alcohol.
Fainstad provides the Dietary Guidelines’ cutoffs to patients and educates them on the poisonous nature of the substance.
Clearer guidance from large governing bodies — potential changes around alcohol in the 2025-230 revision of the US Dietary Guidelines or cancer warnings on booze sold in the United States — are coming and could help streamline messaging.
And although he speaks with urgency about alcohol’s dangers, Farkouh emphasized the need for a judgment-free and patient-centered approach to conversations around drinking: “People have grown up with alcohol being acceptable, and it’s going to take time to change that.”
A version of this article first appeared on Medscape.com.
On Second Thought: Making Sense of Blood Pressure Guidelines — What Happened in the 1930s Should Stay There
This transcript has been edited for clarity.
Blood pressure. If you’re a primary care provider trying to do right by your patients, you might be understandably confused by the current mishmash of guidelines with different blood pressure targets. But as chaotic as things are, at least it’s not the 1930s, when you might hear John Hay give a lecture to the British Medical Association and say, “The greatest danger to a man with high blood pressure lies in its discovery, because then some fool is certain to try and reduce it.”
Yeah, he said that. But what happened in the 1930s stays in the 1930s. And now we can at least agree that we should be treating high blood pressure. But what’s the goal we should be aiming for? This is On Second Thought.
We’ve come a long way since FDR was recording blood pressures of 200 and his doctor prescribed him barbiturates and massage therapy.
That s#$# don’t fly no more. Over the past hundred years, we have become much more aggressive in treating blood pressure. Remember the Oslo study? It defined mild hypertension as a blood pressure between 150 and 180 mm Hg. Now, those numbers send people screaming to the emergency room. So, let’s acknowledge that things are substantially better than they once were. Let’s agree on that and we can start to heal this nation again.
Before we get into the numbers, when we’re treating blood pressure, let’s make a few points about measuring it. Obviously, to treat something, you have to measure it properly. Two recent trials have illustrated that these details matter a lot.
The Cuff(SZ) randomized crossover trial — and it took me a minute to realize that Cuff(SZ) meant cuff size, so bravo, Ishigami et al — showed that picking the wrong cuff size could affect BP measurements by 4.5 points if you were one size off. If you were two sizes too small, you overestimated BP by almost 20 points.
Add on here another recent study, the ARMS crossover randomized clinical trial, looking at how arm position affected BP measures. If the arm was resting on your lap or hanging by your side, that overestimated blood pressure by 4 and 6.5 points. So sometimes you have to remember the fundamentals: cuff size, arm position — it might make the difference between increasing or maintaining the patient’s meds.
But on to the main show. What numbers should we be aiming for? We no longer live in the “BP 200, the president’s going to have a stroke” world of the 1940s, and even a BP of 150 is considered quite high these days. Studies like the MRC trial, INVEST, and SPRINT have pushed BP targets ever lower. SPRINT, in particular, randomized patients to a blood pressure target under 120 systolic vs under 140 systolic, and the under-120 arm won out with fewer cardiovascular events and lower all-cause mortality.
Pretty definitive slam dunk. But the more intensive treatment came with more hypotension, syncope, and kidney injury, because there is no free lunch in medicine. And ditto with BPROAD, just published in The New England Journal of Medicine and presented at the American Heart Association annual meeting. A diabetic population randomized to 120 vs 140 as a BP target showed that more aggressive treatment was better.
Fewer cardiovascular events, like stroke, but no mortality difference, and more hypotension. So a cardiovascular benefit at the cost of more side effects. Now, like all cardiologists, my motto is “Save the heart and screw the kidney.” But if you do care about the other organs in this meat sack that we call a human body, the question you need to wrestle with is, how much do you value cardiovascular protection vs how willing are you to tolerate side effects?
Hypotension may not sound dangerous, but gravity is an unforgiving mistress. If you painstakingly compile the summary of the various BP guidelines for easy perusal, you would notice something critical: One, I have too much free time on my hands; two, the disagreements are not really all that profound.
Arguing about 120 vs 130 vs 140 is not the same as saying, “Drugs schmugs; a good massage will fix what ails you, and here are some addictive sleeping pills for good measure.” Physicians from the 1930s were a little sketchy. So much of this controversy is about how you define high-risk patients and what are the age cutoffs.
Basically, the cardiovascular guidelines say, “Treat them all and let God sort it out” because they care about cardiovascular events and are concerned about cardiovascular endpoints. Whereas general practice guidelines put more emphasis on potential side effects and admittedly tend to treat a not so high-risk population, so they have laxer targets.
A 2014 analysis from the Blood Pressure Lowering Treatment Trialists’ Collaboration [The Lancet] had a good mathematical way of explaining this problem. Now, lowering blood pressure is obviously a good thing. That prevents heart attacks, strokes, kidney failure, and all that. Please don’t let hypertension denialism become a thing.
Let’s start with the basics. Treating high blood pressure led to a 15% to 18% decrease in cardiovascular events, pretty consistently across all risk categories, and other analyses have found that every 5-point decrease in blood pressure gives you about a 10% decrease in major cardiovascular events on the relative-risk scale.
While the benefits are pretty consistent across all groups, that difference in baseline risk translates into different absolute benefits. In the Lancet paper, when the population was divided into four different groups based on their cardiovascular risk, the absolute risk reduction in the lowest-risk group was 14 fewer cardiovascular events if you treat 1000 patients for 5 years.
With each higher-risk group, it was 20 fewer, 24 fewer, and 38 fewer. At the lowest-risk group, the number needed to treat was 71, 50, 42, and 26 fewer cardiovascular events with 5 years of treatment.
And herein lies the secret to the disagreement: If you have a high-risk patient, there is a big benefit to bringing that blood pressure down from 135 to 130. Whereas for a low-risk patient, it probably doesn’t matter as much. And the cardiovascular benefits are going to be offset by the side effects and the risks for hypotension.
Of course, there’s a simple solution to this dilemma: Just speak to the patient in front of you. Treat high blood pressure, and if your patient’s blood pressure drops or they get dizzy or have fainting spells, then just ease up on the meds. It’s not rocket science; it’s just cardiology.
Arguing about five millimeters of mercury of blood pressure is probably less important from the public health perspective than the fact that tens of millions of people in the United States are unaware that they have hypertension, and even those diagnosed are being inadequately treated.
So, let’s all do better as a medical community. Nobody should have untreated hypertension in this day and age. It’s not the 1930s.
Dr Labos, Cardiologist, Kirkland Medical Center, Montreal, Quebec, Canada, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Blood pressure. If you’re a primary care provider trying to do right by your patients, you might be understandably confused by the current mishmash of guidelines with different blood pressure targets. But as chaotic as things are, at least it’s not the 1930s, when you might hear John Hay give a lecture to the British Medical Association and say, “The greatest danger to a man with high blood pressure lies in its discovery, because then some fool is certain to try and reduce it.”
Yeah, he said that. But what happened in the 1930s stays in the 1930s. And now we can at least agree that we should be treating high blood pressure. But what’s the goal we should be aiming for? This is On Second Thought.
We’ve come a long way since FDR was recording blood pressures of 200 and his doctor prescribed him barbiturates and massage therapy.
That s#$# don’t fly no more. Over the past hundred years, we have become much more aggressive in treating blood pressure. Remember the Oslo study? It defined mild hypertension as a blood pressure between 150 and 180 mm Hg. Now, those numbers send people screaming to the emergency room. So, let’s acknowledge that things are substantially better than they once were. Let’s agree on that and we can start to heal this nation again.
Before we get into the numbers, when we’re treating blood pressure, let’s make a few points about measuring it. Obviously, to treat something, you have to measure it properly. Two recent trials have illustrated that these details matter a lot.
The Cuff(SZ) randomized crossover trial — and it took me a minute to realize that Cuff(SZ) meant cuff size, so bravo, Ishigami et al — showed that picking the wrong cuff size could affect BP measurements by 4.5 points if you were one size off. If you were two sizes too small, you overestimated BP by almost 20 points.
Add on here another recent study, the ARMS crossover randomized clinical trial, looking at how arm position affected BP measures. If the arm was resting on your lap or hanging by your side, that overestimated blood pressure by 4 and 6.5 points. So sometimes you have to remember the fundamentals: cuff size, arm position — it might make the difference between increasing or maintaining the patient’s meds.
But on to the main show. What numbers should we be aiming for? We no longer live in the “BP 200, the president’s going to have a stroke” world of the 1940s, and even a BP of 150 is considered quite high these days. Studies like the MRC trial, INVEST, and SPRINT have pushed BP targets ever lower. SPRINT, in particular, randomized patients to a blood pressure target under 120 systolic vs under 140 systolic, and the under-120 arm won out with fewer cardiovascular events and lower all-cause mortality.
Pretty definitive slam dunk. But the more intensive treatment came with more hypotension, syncope, and kidney injury, because there is no free lunch in medicine. And ditto with BPROAD, just published in The New England Journal of Medicine and presented at the American Heart Association annual meeting. A diabetic population randomized to 120 vs 140 as a BP target showed that more aggressive treatment was better.
Fewer cardiovascular events, like stroke, but no mortality difference, and more hypotension. So a cardiovascular benefit at the cost of more side effects. Now, like all cardiologists, my motto is “Save the heart and screw the kidney.” But if you do care about the other organs in this meat sack that we call a human body, the question you need to wrestle with is, how much do you value cardiovascular protection vs how willing are you to tolerate side effects?
Hypotension may not sound dangerous, but gravity is an unforgiving mistress. If you painstakingly compile the summary of the various BP guidelines for easy perusal, you would notice something critical: One, I have too much free time on my hands; two, the disagreements are not really all that profound.
Arguing about 120 vs 130 vs 140 is not the same as saying, “Drugs schmugs; a good massage will fix what ails you, and here are some addictive sleeping pills for good measure.” Physicians from the 1930s were a little sketchy. So much of this controversy is about how you define high-risk patients and what are the age cutoffs.
Basically, the cardiovascular guidelines say, “Treat them all and let God sort it out” because they care about cardiovascular events and are concerned about cardiovascular endpoints. Whereas general practice guidelines put more emphasis on potential side effects and admittedly tend to treat a not so high-risk population, so they have laxer targets.
A 2014 analysis from the Blood Pressure Lowering Treatment Trialists’ Collaboration [The Lancet] had a good mathematical way of explaining this problem. Now, lowering blood pressure is obviously a good thing. That prevents heart attacks, strokes, kidney failure, and all that. Please don’t let hypertension denialism become a thing.
Let’s start with the basics. Treating high blood pressure led to a 15% to 18% decrease in cardiovascular events, pretty consistently across all risk categories, and other analyses have found that every 5-point decrease in blood pressure gives you about a 10% decrease in major cardiovascular events on the relative-risk scale.
While the benefits are pretty consistent across all groups, that difference in baseline risk translates into different absolute benefits. In the Lancet paper, when the population was divided into four different groups based on their cardiovascular risk, the absolute risk reduction in the lowest-risk group was 14 fewer cardiovascular events if you treat 1000 patients for 5 years.
With each higher-risk group, it was 20 fewer, 24 fewer, and 38 fewer. At the lowest-risk group, the number needed to treat was 71, 50, 42, and 26 fewer cardiovascular events with 5 years of treatment.
And herein lies the secret to the disagreement: If you have a high-risk patient, there is a big benefit to bringing that blood pressure down from 135 to 130. Whereas for a low-risk patient, it probably doesn’t matter as much. And the cardiovascular benefits are going to be offset by the side effects and the risks for hypotension.
Of course, there’s a simple solution to this dilemma: Just speak to the patient in front of you. Treat high blood pressure, and if your patient’s blood pressure drops or they get dizzy or have fainting spells, then just ease up on the meds. It’s not rocket science; it’s just cardiology.
Arguing about five millimeters of mercury of blood pressure is probably less important from the public health perspective than the fact that tens of millions of people in the United States are unaware that they have hypertension, and even those diagnosed are being inadequately treated.
So, let’s all do better as a medical community. Nobody should have untreated hypertension in this day and age. It’s not the 1930s.
Dr Labos, Cardiologist, Kirkland Medical Center, Montreal, Quebec, Canada, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Blood pressure. If you’re a primary care provider trying to do right by your patients, you might be understandably confused by the current mishmash of guidelines with different blood pressure targets. But as chaotic as things are, at least it’s not the 1930s, when you might hear John Hay give a lecture to the British Medical Association and say, “The greatest danger to a man with high blood pressure lies in its discovery, because then some fool is certain to try and reduce it.”
Yeah, he said that. But what happened in the 1930s stays in the 1930s. And now we can at least agree that we should be treating high blood pressure. But what’s the goal we should be aiming for? This is On Second Thought.
We’ve come a long way since FDR was recording blood pressures of 200 and his doctor prescribed him barbiturates and massage therapy.
That s#$# don’t fly no more. Over the past hundred years, we have become much more aggressive in treating blood pressure. Remember the Oslo study? It defined mild hypertension as a blood pressure between 150 and 180 mm Hg. Now, those numbers send people screaming to the emergency room. So, let’s acknowledge that things are substantially better than they once were. Let’s agree on that and we can start to heal this nation again.
Before we get into the numbers, when we’re treating blood pressure, let’s make a few points about measuring it. Obviously, to treat something, you have to measure it properly. Two recent trials have illustrated that these details matter a lot.
The Cuff(SZ) randomized crossover trial — and it took me a minute to realize that Cuff(SZ) meant cuff size, so bravo, Ishigami et al — showed that picking the wrong cuff size could affect BP measurements by 4.5 points if you were one size off. If you were two sizes too small, you overestimated BP by almost 20 points.
Add on here another recent study, the ARMS crossover randomized clinical trial, looking at how arm position affected BP measures. If the arm was resting on your lap or hanging by your side, that overestimated blood pressure by 4 and 6.5 points. So sometimes you have to remember the fundamentals: cuff size, arm position — it might make the difference between increasing or maintaining the patient’s meds.
But on to the main show. What numbers should we be aiming for? We no longer live in the “BP 200, the president’s going to have a stroke” world of the 1940s, and even a BP of 150 is considered quite high these days. Studies like the MRC trial, INVEST, and SPRINT have pushed BP targets ever lower. SPRINT, in particular, randomized patients to a blood pressure target under 120 systolic vs under 140 systolic, and the under-120 arm won out with fewer cardiovascular events and lower all-cause mortality.
Pretty definitive slam dunk. But the more intensive treatment came with more hypotension, syncope, and kidney injury, because there is no free lunch in medicine. And ditto with BPROAD, just published in The New England Journal of Medicine and presented at the American Heart Association annual meeting. A diabetic population randomized to 120 vs 140 as a BP target showed that more aggressive treatment was better.
Fewer cardiovascular events, like stroke, but no mortality difference, and more hypotension. So a cardiovascular benefit at the cost of more side effects. Now, like all cardiologists, my motto is “Save the heart and screw the kidney.” But if you do care about the other organs in this meat sack that we call a human body, the question you need to wrestle with is, how much do you value cardiovascular protection vs how willing are you to tolerate side effects?
Hypotension may not sound dangerous, but gravity is an unforgiving mistress. If you painstakingly compile the summary of the various BP guidelines for easy perusal, you would notice something critical: One, I have too much free time on my hands; two, the disagreements are not really all that profound.
Arguing about 120 vs 130 vs 140 is not the same as saying, “Drugs schmugs; a good massage will fix what ails you, and here are some addictive sleeping pills for good measure.” Physicians from the 1930s were a little sketchy. So much of this controversy is about how you define high-risk patients and what are the age cutoffs.
Basically, the cardiovascular guidelines say, “Treat them all and let God sort it out” because they care about cardiovascular events and are concerned about cardiovascular endpoints. Whereas general practice guidelines put more emphasis on potential side effects and admittedly tend to treat a not so high-risk population, so they have laxer targets.
A 2014 analysis from the Blood Pressure Lowering Treatment Trialists’ Collaboration [The Lancet] had a good mathematical way of explaining this problem. Now, lowering blood pressure is obviously a good thing. That prevents heart attacks, strokes, kidney failure, and all that. Please don’t let hypertension denialism become a thing.
Let’s start with the basics. Treating high blood pressure led to a 15% to 18% decrease in cardiovascular events, pretty consistently across all risk categories, and other analyses have found that every 5-point decrease in blood pressure gives you about a 10% decrease in major cardiovascular events on the relative-risk scale.
While the benefits are pretty consistent across all groups, that difference in baseline risk translates into different absolute benefits. In the Lancet paper, when the population was divided into four different groups based on their cardiovascular risk, the absolute risk reduction in the lowest-risk group was 14 fewer cardiovascular events if you treat 1000 patients for 5 years.
With each higher-risk group, it was 20 fewer, 24 fewer, and 38 fewer. At the lowest-risk group, the number needed to treat was 71, 50, 42, and 26 fewer cardiovascular events with 5 years of treatment.
And herein lies the secret to the disagreement: If you have a high-risk patient, there is a big benefit to bringing that blood pressure down from 135 to 130. Whereas for a low-risk patient, it probably doesn’t matter as much. And the cardiovascular benefits are going to be offset by the side effects and the risks for hypotension.
Of course, there’s a simple solution to this dilemma: Just speak to the patient in front of you. Treat high blood pressure, and if your patient’s blood pressure drops or they get dizzy or have fainting spells, then just ease up on the meds. It’s not rocket science; it’s just cardiology.
Arguing about five millimeters of mercury of blood pressure is probably less important from the public health perspective than the fact that tens of millions of people in the United States are unaware that they have hypertension, and even those diagnosed are being inadequately treated.
So, let’s all do better as a medical community. Nobody should have untreated hypertension in this day and age. It’s not the 1930s.
Dr Labos, Cardiologist, Kirkland Medical Center, Montreal, Quebec, Canada, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Do We Need Cardiovascular Risk Equations to Guide Statin Use?
An individual’s estimated risk of having a heart attack or stroke in the next 10 years is widely used to guide preventative medication prescriptions with statins or antihypertensive drugs in those who have not yet had such an event.
To estimate that risk, doctors use equations that include different risk factors, such as age, cholesterol levels, and blood pressure. The current equations, known as the pooled cohort equations, are considered to be outdated as they were developed in 2013 based on population data from the 1960s and 70s. A new set of risk equations — known as the PREVENT equations — were developed by the American Heart Association (AHA) in 2023, and are based on a more contemporary population. It is anticipated that AHA will recommend these new risk equations be used in clinical practice in the next primary prevention guidelines.
But could these new risk equations do more harm than good?
Two recent studies found that applying the PREVENT risk equations to the US population results in a much lower overall level of risk compared with the pooled cohort equations. And, if the current threshold for starting statin treatment — which is an estimated 7.5% risk of having a heart attack or stroke in the next 10 years — is kept the same, this would result in many fewer patients being eligible for statin treatment.
As cardiovascular risk is also used to guide antihypertensive treatment, the new risk equations would also result in fewer people with borderline high blood pressure being eligible for those medications.
This has raised concerns in the medical community, where there is a widespread view that many more people would benefit from primary prevention treatment, and that anything that may cause fewer people to receive these medications would be harmful.
“I believe the new equations more accurately predict the risk of the current US population, but we need to be aware of what effect that may have on use of statins,” said Tim Anderson, MD, who studies healthcare delivery at the University of Pittsburgh in Pennsylvania and is lead author of one of the studies evaluating the equations.
Anderson told this news organization that the pooled cohort equations have long been viewed as problematic.
“Because these equations were based on cohorts from the 1960s and 70s, it is believed they overestimate the current population’s risk of MI and stroke as the burden of disease has shifted in the intervening 50-60 years,” he said.
Current Equations Overestimate Risk
The new equations are based on more recent, representative, and diverse cohorts that capture a wider spectrum of the population in terms of race, ethnicity, and socioeconomic status. They also include factors that are now known to be relevant to cardiovascular risk, such as chronic kidney disease.
Anderson compared how the two sets of equations estimated risk of cardiovascular disease in the next 10 years in the US population using the NHANES survey — a large nationally representative survey conducted between 2017 and 2020.
He found that the pooled cohort equations estimated the population average 10-year risk of cardiovascular disease to be about 8%, but the PREVENT equations estimated it at just over 4%.
“The new equations estimate that the middle-aged US population have almost half the level of risk of MI and stroke over next 10 years compared with the equations used currently. So, we will substantially change risk estimates if the new equations are introduced into practice,” Anderson said.
The study found that, if the PREVENT equations are adopted in the next set of primary prevention guidelines and the current threshold of a 7.5% risk of having an MI or stroke in the next 10 years is maintained as the starting point for statin treatment, then 17.3 million adults who were previously recommended primary prevention statin therapy would no longer be eligible.
A second, similar study, conducted by a different team of US researchers, estimated that using PREVENT would decrease the number of US adults receiving or recommended for statin therapy by 14.3 million and antihypertensive therapy by 2.62 million.
The researchers, led by James A. Diao, MD, from Harvard Medical School, Boston, Massachusetts, also suggested that over 10 years, reductions in treatment eligibility could result in an estimated 107,000 additional MI or stroke events.
Anderson points out that using the new equations would not affect the highest-risk patients. “They are still going to be high risk whichever equations are used. If you smoke a pack of cigarettes a day, have very high blood pressure or cholesterol and are older, then you are high risk. That part hasn’t changed. These people will qualify for statin treatment many times over with both sets of guidelines,” he said.
Rather it will be the large population at moderate risk of cardiovascular disease that will be affected, with far fewer of these individuals likely to get statins.
“If you are on the fence about whether to take a statin or not and you’re currently just on the threshold where they might be recommended then these new equations could mean that you’ll be less likely to be offered them,” he said. “Using the new equations may result in a delay of a couple of years to have that conversation.”
A Red Flag
Steve Nissen, MD, a cardiologist at the Cleveland Clinic in Ohio, is not a fan of cardiovascular risk equations in general. He points out that less than half of those currently eligible for statins are actually treated. And he believes the studies suggesting fewer people will be eligible with the new risk equations raise a red flag on whether they should be used.
“Anything that may result in fewer people being treated is a huge problem,” he told this news organization. “We have abundant evidence that we should be treating more people, not fewer people. Every study we have done has shown benefit with statins.”
The risk calculators were initially developed to limit use of statins and other medications to high-risk patients, he said, but now that we know more about safety of these drugs, it’s clear that the risks are almost nonexistent.
“We really need something else to guide the prescription of statins,” said Nissen.
Nissen suggests the risk calculators and guidelines have resulted in undertreatment of the population because they lack nuance and put too much emphasis on age. We should be more interested in reducing the lifetime risk of cardiovascular events, he said. “Calculators don’t do a good job of that. Their time horizons are too short. Young people with a family history of cardiovascular disease may have a low 10-year risk on a risk calculator but their lifetime risk is elevated, and as such, they should be considered for statin treatment. We need to find a more nuanced approach to understanding the lifetime risk of individuals,” he said.
Nissen said risk calculators can be useful in high-risk patients to help demonstrate their need for treatment. “I can show them the calculator and that they have a 20% chance of an event — that can help convince them to take a statin.”
But at the lower end of the risk scale, “all it does is keep patients who should be getting treatment from having that treatment.”
Nissen said changing the risk calculator won’t affect how he treats patients. “I use judgment to decide who to treat based on scientific literature and the patient in front of me. We will engage in a discussion and make a shared decision on what is the best course of action. Calculators will never be a substitute for medical judgment,” he said.
Equations Don’t Decide
Sadiya Khan, MD, a cardiologist at Northwestern University, Evanston, Illinois, and lead author of the PREVENT equations, told this news organization that it is important to put this discussion into context.
“The two recent papers do a good job of describing differences in predictive risk between the two sets of equations but that’s where they stop,” she said. “The translation from that to the decision on who should or should not be on statins or other medications is a step too far.”
Clinical guidelines will need to be updated to take the PREVENT equations into account, as Khan argued in a JAMA editorial. So it is not clear whether the current 7.5% 10-year risk figure will remain the threshold to start treatment. Khan anticipates the guidelines committee will have to re-evaluate that threshold.
“The 7.5% risk threshold was advised in the 2013 guidelines, based on what we knew then about the balance between benefit and harm and with the knowledge that the risk equations overestimated risk,” she said. “We now have a lot more data on the safety of statin therapy. We see this frequently in preventive care. Treatments often becomes more widespread in time and use expands into lower-risk patients.”
She also pointed out that the current primary prevention guidelines encourage consideration of other factors, not just predictive risk scores, when thinking about starting statins, including very high LDL cholesterol, family history, and apo B and Lp(a) levels.
“The recommendation on who would qualify for statin therapy is not based on one number,” she said. “It is based on many considerations, including both qualitative and quantitative factors, and discussions between the patient and the doctor. It is not a straightforward yes or no based on a 7.5% risk threshold.”
The equations, she said, should only be viewed as the first step in the process, and she said she agrees with Nissen that when applying the equations, doctors need to use additional data from each individual patient to make a judgment. “Equations do not decide who gets treated. Clinical practice guidelines do that.”
Khan also agreed with Nissen that more effort is needed to identify longer term cardiovascular risk in younger people, and so the PREVENT equations include 30-year risk estimates.
“I totally agree that we need to start earlier in having these prevention conversations. The PREVENT model starts at age 30 which is 10 years earlier than the pooled cohort equations and they add a 30-year time horizon as well as the 10-year period for these discussions on predicted risk estimates,” she said. “We need to make sure we are not missing risk in young adults just because we are waiting for them to get into some arbitrary age category.”
Khan says she believes that, used correctly, the new equations will not limit access to statins or other cardiovascular treatments. “Because they are a more accurate reflection of risk in the contemporary population, the new PREVENT equations should identify the correct patients to be treated, within the confines of knowing that no risk prediction equation is perfect,” she said. “And if everything else is considered as well, not just the numbers in the risk equations, it shouldn’t result in fewer patients being treated.”
Anderson reported receiving grants from the American Heart Association, the American College of Cardiology, and the US Deprescribing Research Network. Nissen is leading a development program for a nonprescription low dose of rosuvastatin. He is also involved in trials of a new cholesterol lowering drug, obicetrapib, and on trials on drugs that lower Lp(a). Khan reported receiving grants from the American Heart Association and National Heart, Lung, and Blood Institute.
A version of this article first appeared on Medscape.com.
An individual’s estimated risk of having a heart attack or stroke in the next 10 years is widely used to guide preventative medication prescriptions with statins or antihypertensive drugs in those who have not yet had such an event.
To estimate that risk, doctors use equations that include different risk factors, such as age, cholesterol levels, and blood pressure. The current equations, known as the pooled cohort equations, are considered to be outdated as they were developed in 2013 based on population data from the 1960s and 70s. A new set of risk equations — known as the PREVENT equations — were developed by the American Heart Association (AHA) in 2023, and are based on a more contemporary population. It is anticipated that AHA will recommend these new risk equations be used in clinical practice in the next primary prevention guidelines.
But could these new risk equations do more harm than good?
Two recent studies found that applying the PREVENT risk equations to the US population results in a much lower overall level of risk compared with the pooled cohort equations. And, if the current threshold for starting statin treatment — which is an estimated 7.5% risk of having a heart attack or stroke in the next 10 years — is kept the same, this would result in many fewer patients being eligible for statin treatment.
As cardiovascular risk is also used to guide antihypertensive treatment, the new risk equations would also result in fewer people with borderline high blood pressure being eligible for those medications.
This has raised concerns in the medical community, where there is a widespread view that many more people would benefit from primary prevention treatment, and that anything that may cause fewer people to receive these medications would be harmful.
“I believe the new equations more accurately predict the risk of the current US population, but we need to be aware of what effect that may have on use of statins,” said Tim Anderson, MD, who studies healthcare delivery at the University of Pittsburgh in Pennsylvania and is lead author of one of the studies evaluating the equations.
Anderson told this news organization that the pooled cohort equations have long been viewed as problematic.
“Because these equations were based on cohorts from the 1960s and 70s, it is believed they overestimate the current population’s risk of MI and stroke as the burden of disease has shifted in the intervening 50-60 years,” he said.
Current Equations Overestimate Risk
The new equations are based on more recent, representative, and diverse cohorts that capture a wider spectrum of the population in terms of race, ethnicity, and socioeconomic status. They also include factors that are now known to be relevant to cardiovascular risk, such as chronic kidney disease.
Anderson compared how the two sets of equations estimated risk of cardiovascular disease in the next 10 years in the US population using the NHANES survey — a large nationally representative survey conducted between 2017 and 2020.
He found that the pooled cohort equations estimated the population average 10-year risk of cardiovascular disease to be about 8%, but the PREVENT equations estimated it at just over 4%.
“The new equations estimate that the middle-aged US population have almost half the level of risk of MI and stroke over next 10 years compared with the equations used currently. So, we will substantially change risk estimates if the new equations are introduced into practice,” Anderson said.
The study found that, if the PREVENT equations are adopted in the next set of primary prevention guidelines and the current threshold of a 7.5% risk of having an MI or stroke in the next 10 years is maintained as the starting point for statin treatment, then 17.3 million adults who were previously recommended primary prevention statin therapy would no longer be eligible.
A second, similar study, conducted by a different team of US researchers, estimated that using PREVENT would decrease the number of US adults receiving or recommended for statin therapy by 14.3 million and antihypertensive therapy by 2.62 million.
The researchers, led by James A. Diao, MD, from Harvard Medical School, Boston, Massachusetts, also suggested that over 10 years, reductions in treatment eligibility could result in an estimated 107,000 additional MI or stroke events.
Anderson points out that using the new equations would not affect the highest-risk patients. “They are still going to be high risk whichever equations are used. If you smoke a pack of cigarettes a day, have very high blood pressure or cholesterol and are older, then you are high risk. That part hasn’t changed. These people will qualify for statin treatment many times over with both sets of guidelines,” he said.
Rather it will be the large population at moderate risk of cardiovascular disease that will be affected, with far fewer of these individuals likely to get statins.
“If you are on the fence about whether to take a statin or not and you’re currently just on the threshold where they might be recommended then these new equations could mean that you’ll be less likely to be offered them,” he said. “Using the new equations may result in a delay of a couple of years to have that conversation.”
A Red Flag
Steve Nissen, MD, a cardiologist at the Cleveland Clinic in Ohio, is not a fan of cardiovascular risk equations in general. He points out that less than half of those currently eligible for statins are actually treated. And he believes the studies suggesting fewer people will be eligible with the new risk equations raise a red flag on whether they should be used.
“Anything that may result in fewer people being treated is a huge problem,” he told this news organization. “We have abundant evidence that we should be treating more people, not fewer people. Every study we have done has shown benefit with statins.”
The risk calculators were initially developed to limit use of statins and other medications to high-risk patients, he said, but now that we know more about safety of these drugs, it’s clear that the risks are almost nonexistent.
“We really need something else to guide the prescription of statins,” said Nissen.
Nissen suggests the risk calculators and guidelines have resulted in undertreatment of the population because they lack nuance and put too much emphasis on age. We should be more interested in reducing the lifetime risk of cardiovascular events, he said. “Calculators don’t do a good job of that. Their time horizons are too short. Young people with a family history of cardiovascular disease may have a low 10-year risk on a risk calculator but their lifetime risk is elevated, and as such, they should be considered for statin treatment. We need to find a more nuanced approach to understanding the lifetime risk of individuals,” he said.
Nissen said risk calculators can be useful in high-risk patients to help demonstrate their need for treatment. “I can show them the calculator and that they have a 20% chance of an event — that can help convince them to take a statin.”
But at the lower end of the risk scale, “all it does is keep patients who should be getting treatment from having that treatment.”
Nissen said changing the risk calculator won’t affect how he treats patients. “I use judgment to decide who to treat based on scientific literature and the patient in front of me. We will engage in a discussion and make a shared decision on what is the best course of action. Calculators will never be a substitute for medical judgment,” he said.
Equations Don’t Decide
Sadiya Khan, MD, a cardiologist at Northwestern University, Evanston, Illinois, and lead author of the PREVENT equations, told this news organization that it is important to put this discussion into context.
“The two recent papers do a good job of describing differences in predictive risk between the two sets of equations but that’s where they stop,” she said. “The translation from that to the decision on who should or should not be on statins or other medications is a step too far.”
Clinical guidelines will need to be updated to take the PREVENT equations into account, as Khan argued in a JAMA editorial. So it is not clear whether the current 7.5% 10-year risk figure will remain the threshold to start treatment. Khan anticipates the guidelines committee will have to re-evaluate that threshold.
“The 7.5% risk threshold was advised in the 2013 guidelines, based on what we knew then about the balance between benefit and harm and with the knowledge that the risk equations overestimated risk,” she said. “We now have a lot more data on the safety of statin therapy. We see this frequently in preventive care. Treatments often becomes more widespread in time and use expands into lower-risk patients.”
She also pointed out that the current primary prevention guidelines encourage consideration of other factors, not just predictive risk scores, when thinking about starting statins, including very high LDL cholesterol, family history, and apo B and Lp(a) levels.
“The recommendation on who would qualify for statin therapy is not based on one number,” she said. “It is based on many considerations, including both qualitative and quantitative factors, and discussions between the patient and the doctor. It is not a straightforward yes or no based on a 7.5% risk threshold.”
The equations, she said, should only be viewed as the first step in the process, and she said she agrees with Nissen that when applying the equations, doctors need to use additional data from each individual patient to make a judgment. “Equations do not decide who gets treated. Clinical practice guidelines do that.”
Khan also agreed with Nissen that more effort is needed to identify longer term cardiovascular risk in younger people, and so the PREVENT equations include 30-year risk estimates.
“I totally agree that we need to start earlier in having these prevention conversations. The PREVENT model starts at age 30 which is 10 years earlier than the pooled cohort equations and they add a 30-year time horizon as well as the 10-year period for these discussions on predicted risk estimates,” she said. “We need to make sure we are not missing risk in young adults just because we are waiting for them to get into some arbitrary age category.”
Khan says she believes that, used correctly, the new equations will not limit access to statins or other cardiovascular treatments. “Because they are a more accurate reflection of risk in the contemporary population, the new PREVENT equations should identify the correct patients to be treated, within the confines of knowing that no risk prediction equation is perfect,” she said. “And if everything else is considered as well, not just the numbers in the risk equations, it shouldn’t result in fewer patients being treated.”
Anderson reported receiving grants from the American Heart Association, the American College of Cardiology, and the US Deprescribing Research Network. Nissen is leading a development program for a nonprescription low dose of rosuvastatin. He is also involved in trials of a new cholesterol lowering drug, obicetrapib, and on trials on drugs that lower Lp(a). Khan reported receiving grants from the American Heart Association and National Heart, Lung, and Blood Institute.
A version of this article first appeared on Medscape.com.
An individual’s estimated risk of having a heart attack or stroke in the next 10 years is widely used to guide preventative medication prescriptions with statins or antihypertensive drugs in those who have not yet had such an event.
To estimate that risk, doctors use equations that include different risk factors, such as age, cholesterol levels, and blood pressure. The current equations, known as the pooled cohort equations, are considered to be outdated as they were developed in 2013 based on population data from the 1960s and 70s. A new set of risk equations — known as the PREVENT equations — were developed by the American Heart Association (AHA) in 2023, and are based on a more contemporary population. It is anticipated that AHA will recommend these new risk equations be used in clinical practice in the next primary prevention guidelines.
But could these new risk equations do more harm than good?
Two recent studies found that applying the PREVENT risk equations to the US population results in a much lower overall level of risk compared with the pooled cohort equations. And, if the current threshold for starting statin treatment — which is an estimated 7.5% risk of having a heart attack or stroke in the next 10 years — is kept the same, this would result in many fewer patients being eligible for statin treatment.
As cardiovascular risk is also used to guide antihypertensive treatment, the new risk equations would also result in fewer people with borderline high blood pressure being eligible for those medications.
This has raised concerns in the medical community, where there is a widespread view that many more people would benefit from primary prevention treatment, and that anything that may cause fewer people to receive these medications would be harmful.
“I believe the new equations more accurately predict the risk of the current US population, but we need to be aware of what effect that may have on use of statins,” said Tim Anderson, MD, who studies healthcare delivery at the University of Pittsburgh in Pennsylvania and is lead author of one of the studies evaluating the equations.
Anderson told this news organization that the pooled cohort equations have long been viewed as problematic.
“Because these equations were based on cohorts from the 1960s and 70s, it is believed they overestimate the current population’s risk of MI and stroke as the burden of disease has shifted in the intervening 50-60 years,” he said.
Current Equations Overestimate Risk
The new equations are based on more recent, representative, and diverse cohorts that capture a wider spectrum of the population in terms of race, ethnicity, and socioeconomic status. They also include factors that are now known to be relevant to cardiovascular risk, such as chronic kidney disease.
Anderson compared how the two sets of equations estimated risk of cardiovascular disease in the next 10 years in the US population using the NHANES survey — a large nationally representative survey conducted between 2017 and 2020.
He found that the pooled cohort equations estimated the population average 10-year risk of cardiovascular disease to be about 8%, but the PREVENT equations estimated it at just over 4%.
“The new equations estimate that the middle-aged US population have almost half the level of risk of MI and stroke over next 10 years compared with the equations used currently. So, we will substantially change risk estimates if the new equations are introduced into practice,” Anderson said.
The study found that, if the PREVENT equations are adopted in the next set of primary prevention guidelines and the current threshold of a 7.5% risk of having an MI or stroke in the next 10 years is maintained as the starting point for statin treatment, then 17.3 million adults who were previously recommended primary prevention statin therapy would no longer be eligible.
A second, similar study, conducted by a different team of US researchers, estimated that using PREVENT would decrease the number of US adults receiving or recommended for statin therapy by 14.3 million and antihypertensive therapy by 2.62 million.
The researchers, led by James A. Diao, MD, from Harvard Medical School, Boston, Massachusetts, also suggested that over 10 years, reductions in treatment eligibility could result in an estimated 107,000 additional MI or stroke events.
Anderson points out that using the new equations would not affect the highest-risk patients. “They are still going to be high risk whichever equations are used. If you smoke a pack of cigarettes a day, have very high blood pressure or cholesterol and are older, then you are high risk. That part hasn’t changed. These people will qualify for statin treatment many times over with both sets of guidelines,” he said.
Rather it will be the large population at moderate risk of cardiovascular disease that will be affected, with far fewer of these individuals likely to get statins.
“If you are on the fence about whether to take a statin or not and you’re currently just on the threshold where they might be recommended then these new equations could mean that you’ll be less likely to be offered them,” he said. “Using the new equations may result in a delay of a couple of years to have that conversation.”
A Red Flag
Steve Nissen, MD, a cardiologist at the Cleveland Clinic in Ohio, is not a fan of cardiovascular risk equations in general. He points out that less than half of those currently eligible for statins are actually treated. And he believes the studies suggesting fewer people will be eligible with the new risk equations raise a red flag on whether they should be used.
“Anything that may result in fewer people being treated is a huge problem,” he told this news organization. “We have abundant evidence that we should be treating more people, not fewer people. Every study we have done has shown benefit with statins.”
The risk calculators were initially developed to limit use of statins and other medications to high-risk patients, he said, but now that we know more about safety of these drugs, it’s clear that the risks are almost nonexistent.
“We really need something else to guide the prescription of statins,” said Nissen.
Nissen suggests the risk calculators and guidelines have resulted in undertreatment of the population because they lack nuance and put too much emphasis on age. We should be more interested in reducing the lifetime risk of cardiovascular events, he said. “Calculators don’t do a good job of that. Their time horizons are too short. Young people with a family history of cardiovascular disease may have a low 10-year risk on a risk calculator but their lifetime risk is elevated, and as such, they should be considered for statin treatment. We need to find a more nuanced approach to understanding the lifetime risk of individuals,” he said.
Nissen said risk calculators can be useful in high-risk patients to help demonstrate their need for treatment. “I can show them the calculator and that they have a 20% chance of an event — that can help convince them to take a statin.”
But at the lower end of the risk scale, “all it does is keep patients who should be getting treatment from having that treatment.”
Nissen said changing the risk calculator won’t affect how he treats patients. “I use judgment to decide who to treat based on scientific literature and the patient in front of me. We will engage in a discussion and make a shared decision on what is the best course of action. Calculators will never be a substitute for medical judgment,” he said.
Equations Don’t Decide
Sadiya Khan, MD, a cardiologist at Northwestern University, Evanston, Illinois, and lead author of the PREVENT equations, told this news organization that it is important to put this discussion into context.
“The two recent papers do a good job of describing differences in predictive risk between the two sets of equations but that’s where they stop,” she said. “The translation from that to the decision on who should or should not be on statins or other medications is a step too far.”
Clinical guidelines will need to be updated to take the PREVENT equations into account, as Khan argued in a JAMA editorial. So it is not clear whether the current 7.5% 10-year risk figure will remain the threshold to start treatment. Khan anticipates the guidelines committee will have to re-evaluate that threshold.
“The 7.5% risk threshold was advised in the 2013 guidelines, based on what we knew then about the balance between benefit and harm and with the knowledge that the risk equations overestimated risk,” she said. “We now have a lot more data on the safety of statin therapy. We see this frequently in preventive care. Treatments often becomes more widespread in time and use expands into lower-risk patients.”
She also pointed out that the current primary prevention guidelines encourage consideration of other factors, not just predictive risk scores, when thinking about starting statins, including very high LDL cholesterol, family history, and apo B and Lp(a) levels.
“The recommendation on who would qualify for statin therapy is not based on one number,” she said. “It is based on many considerations, including both qualitative and quantitative factors, and discussions between the patient and the doctor. It is not a straightforward yes or no based on a 7.5% risk threshold.”
The equations, she said, should only be viewed as the first step in the process, and she said she agrees with Nissen that when applying the equations, doctors need to use additional data from each individual patient to make a judgment. “Equations do not decide who gets treated. Clinical practice guidelines do that.”
Khan also agreed with Nissen that more effort is needed to identify longer term cardiovascular risk in younger people, and so the PREVENT equations include 30-year risk estimates.
“I totally agree that we need to start earlier in having these prevention conversations. The PREVENT model starts at age 30 which is 10 years earlier than the pooled cohort equations and they add a 30-year time horizon as well as the 10-year period for these discussions on predicted risk estimates,” she said. “We need to make sure we are not missing risk in young adults just because we are waiting for them to get into some arbitrary age category.”
Khan says she believes that, used correctly, the new equations will not limit access to statins or other cardiovascular treatments. “Because they are a more accurate reflection of risk in the contemporary population, the new PREVENT equations should identify the correct patients to be treated, within the confines of knowing that no risk prediction equation is perfect,” she said. “And if everything else is considered as well, not just the numbers in the risk equations, it shouldn’t result in fewer patients being treated.”
Anderson reported receiving grants from the American Heart Association, the American College of Cardiology, and the US Deprescribing Research Network. Nissen is leading a development program for a nonprescription low dose of rosuvastatin. He is also involved in trials of a new cholesterol lowering drug, obicetrapib, and on trials on drugs that lower Lp(a). Khan reported receiving grants from the American Heart Association and National Heart, Lung, and Blood Institute.
A version of this article first appeared on Medscape.com.
Cardiovascular Risk in T1D: LDL Focus and Beyond
Estimation of cardiovascular risk (CVR) in individuals living with type 1 diabetes (T1D) was a key topic presented by Sophie Borot, MD, from Besançon University Hospital, Besançon, France, at the 40th congress of the French Society of Endocrinology. Borot highlighted the complexities of this subject, outlining several factors that contribute to its challenges.
A Heterogeneous Disease
T1D is a highly heterogeneous condition, and the patients included in studies reflect this diversity:
- The impact of blood glucose levels on CVR changes depending on diabetes duration, its history, the frequency of hypoglycemic episodes, average A1c levels over several years, and the patient’s age at diagnosis.
- A T1D diagnosis from the 1980s involved different management strategies compared with a diagnosis today.
- Patient profiles also vary based on complications such as nephropathy or cardiac autonomic neuropathy.
- Diffuse and distal arterial damage in T1D leads to more subtle and delayed pathologic events than in type 2 diabetes (T2D).
- Most clinical studies assess CVR over 10 years, but a 20- or 30-year evaluation would be more relevant.
- Patients may share CVR factors with the general population (eg, family history, smoking, sedentary lifestyle, obesity, hypertension, or elevated low-density lipoprotein [LDL] levels), raising questions about possible overlap with metabolic syndrome.
- Study criteria differ, with a focus on outcomes such as cardiovascular death, major adverse cardiovascular events like myocardial infarction and stroke, or other endpoints.
- CVR is measured using either absolute or relative values, with varying units of measurement.
A Recent Awareness
The concept of CVR in T1D is relatively new. Until the publication of the prospective Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study in 2005, it was believed that T1D control had no impact on CVR. However, follow-up results from the same cohort of 50,000 patients, published in 2022 after 30 years of observation, revealed that CVR was 20% higher in patients who received conventional hyperglycemia-targeted treatment than those undergoing intensive treatment. The CVR increases in conjunction with diabetes duration. The study also showed that even well-controlled glycemia in T1D carries CVR (primarily due to microangiopathy), and that the most critical factor for CVR is not A1c control but rather LDL cholesterol levels.
These findings were corroborated by a Danish prospective study, which demonstrated that while CVR increased in conjunction with the number of risk factors, it was 82% higher in patients with T1D than in a control group — even in the absence of risk factors.
Key Takeaways
At diagnosis, a fundamental difference exists between T1D and T2D in terms of the urgency to address CVR. In T2D, diabetes may have progressed for years before diagnosis, necessitating immediate CVR reduction efforts. In contrast, T1D is often diagnosed in younger patients with initially low CVR, raising questions about the optimal timing for interventions such as statin prescriptions.
Recommendations
The American Diabetes Association/European Association for the Study of Diabetes guidelines (2024) include the following recommendations:
- Between ages 20 and 40, statins are recommended if at least one CVR factor is present.
- For children 10 years of age or older with T1D, the LDL target is < 1.0 g/L. Statins are prescribed if LDL exceeds 1.6 g/L without CVR factors or 1.3 g/L with at least one CVR factor.
The European Society of Cardiology guidelines (2023) include the following:
- For the first time, a dedicated chapter addresses T1D. Like the American guidelines, routine statin use after age 40 is recommended.
- Before age 40, statins are prescribed if there is at least one CVR factor (microangiopathy) or a 10-year CVR ≥ 10% (based on a CVR calculator).
The International Society for Pediatric and Adolescent Diabetes guidelines (2022) recommend:
- For children 10 years of age or older, the LDL target is < 1.0 g/L. Statins are recommended if LDL exceeds 1.3 g/L.
CAC Score in High CVR
The French Society of Cardiology and the French-speaking Society of Diabetology recommend incorporating the coronary artery calcium (CAC) score to refine CVR classification in high-risk patients. For those without prior cardiovascular events, LDL targets vary based on CAC and age. For example:
- High-risk patients with a CAC of 0-10 are reclassified as moderate risk, with an LDL target of < 1 g/L.
- A CAC ≥ 400 indicates very high risk, warranting coronary exploration.
- Patients under 50 years of age with a CAC of 11-100 remain high risk, with an LDL target of 0.7 g/L.
Conclusion
CVR in patients with T1D remains challenging to define. However, it is essential to consider long-term outcomes, planning for 30 or 40 years into the future. This involves educating patients about the importance of prevention, even when reassuring numbers are seen in their youth.
This story was translated from Univadis France using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Estimation of cardiovascular risk (CVR) in individuals living with type 1 diabetes (T1D) was a key topic presented by Sophie Borot, MD, from Besançon University Hospital, Besançon, France, at the 40th congress of the French Society of Endocrinology. Borot highlighted the complexities of this subject, outlining several factors that contribute to its challenges.
A Heterogeneous Disease
T1D is a highly heterogeneous condition, and the patients included in studies reflect this diversity:
- The impact of blood glucose levels on CVR changes depending on diabetes duration, its history, the frequency of hypoglycemic episodes, average A1c levels over several years, and the patient’s age at diagnosis.
- A T1D diagnosis from the 1980s involved different management strategies compared with a diagnosis today.
- Patient profiles also vary based on complications such as nephropathy or cardiac autonomic neuropathy.
- Diffuse and distal arterial damage in T1D leads to more subtle and delayed pathologic events than in type 2 diabetes (T2D).
- Most clinical studies assess CVR over 10 years, but a 20- or 30-year evaluation would be more relevant.
- Patients may share CVR factors with the general population (eg, family history, smoking, sedentary lifestyle, obesity, hypertension, or elevated low-density lipoprotein [LDL] levels), raising questions about possible overlap with metabolic syndrome.
- Study criteria differ, with a focus on outcomes such as cardiovascular death, major adverse cardiovascular events like myocardial infarction and stroke, or other endpoints.
- CVR is measured using either absolute or relative values, with varying units of measurement.
A Recent Awareness
The concept of CVR in T1D is relatively new. Until the publication of the prospective Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study in 2005, it was believed that T1D control had no impact on CVR. However, follow-up results from the same cohort of 50,000 patients, published in 2022 after 30 years of observation, revealed that CVR was 20% higher in patients who received conventional hyperglycemia-targeted treatment than those undergoing intensive treatment. The CVR increases in conjunction with diabetes duration. The study also showed that even well-controlled glycemia in T1D carries CVR (primarily due to microangiopathy), and that the most critical factor for CVR is not A1c control but rather LDL cholesterol levels.
These findings were corroborated by a Danish prospective study, which demonstrated that while CVR increased in conjunction with the number of risk factors, it was 82% higher in patients with T1D than in a control group — even in the absence of risk factors.
Key Takeaways
At diagnosis, a fundamental difference exists between T1D and T2D in terms of the urgency to address CVR. In T2D, diabetes may have progressed for years before diagnosis, necessitating immediate CVR reduction efforts. In contrast, T1D is often diagnosed in younger patients with initially low CVR, raising questions about the optimal timing for interventions such as statin prescriptions.
Recommendations
The American Diabetes Association/European Association for the Study of Diabetes guidelines (2024) include the following recommendations:
- Between ages 20 and 40, statins are recommended if at least one CVR factor is present.
- For children 10 years of age or older with T1D, the LDL target is < 1.0 g/L. Statins are prescribed if LDL exceeds 1.6 g/L without CVR factors or 1.3 g/L with at least one CVR factor.
The European Society of Cardiology guidelines (2023) include the following:
- For the first time, a dedicated chapter addresses T1D. Like the American guidelines, routine statin use after age 40 is recommended.
- Before age 40, statins are prescribed if there is at least one CVR factor (microangiopathy) or a 10-year CVR ≥ 10% (based on a CVR calculator).
The International Society for Pediatric and Adolescent Diabetes guidelines (2022) recommend:
- For children 10 years of age or older, the LDL target is < 1.0 g/L. Statins are recommended if LDL exceeds 1.3 g/L.
CAC Score in High CVR
The French Society of Cardiology and the French-speaking Society of Diabetology recommend incorporating the coronary artery calcium (CAC) score to refine CVR classification in high-risk patients. For those without prior cardiovascular events, LDL targets vary based on CAC and age. For example:
- High-risk patients with a CAC of 0-10 are reclassified as moderate risk, with an LDL target of < 1 g/L.
- A CAC ≥ 400 indicates very high risk, warranting coronary exploration.
- Patients under 50 years of age with a CAC of 11-100 remain high risk, with an LDL target of 0.7 g/L.
Conclusion
CVR in patients with T1D remains challenging to define. However, it is essential to consider long-term outcomes, planning for 30 or 40 years into the future. This involves educating patients about the importance of prevention, even when reassuring numbers are seen in their youth.
This story was translated from Univadis France using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Estimation of cardiovascular risk (CVR) in individuals living with type 1 diabetes (T1D) was a key topic presented by Sophie Borot, MD, from Besançon University Hospital, Besançon, France, at the 40th congress of the French Society of Endocrinology. Borot highlighted the complexities of this subject, outlining several factors that contribute to its challenges.
A Heterogeneous Disease
T1D is a highly heterogeneous condition, and the patients included in studies reflect this diversity:
- The impact of blood glucose levels on CVR changes depending on diabetes duration, its history, the frequency of hypoglycemic episodes, average A1c levels over several years, and the patient’s age at diagnosis.
- A T1D diagnosis from the 1980s involved different management strategies compared with a diagnosis today.
- Patient profiles also vary based on complications such as nephropathy or cardiac autonomic neuropathy.
- Diffuse and distal arterial damage in T1D leads to more subtle and delayed pathologic events than in type 2 diabetes (T2D).
- Most clinical studies assess CVR over 10 years, but a 20- or 30-year evaluation would be more relevant.
- Patients may share CVR factors with the general population (eg, family history, smoking, sedentary lifestyle, obesity, hypertension, or elevated low-density lipoprotein [LDL] levels), raising questions about possible overlap with metabolic syndrome.
- Study criteria differ, with a focus on outcomes such as cardiovascular death, major adverse cardiovascular events like myocardial infarction and stroke, or other endpoints.
- CVR is measured using either absolute or relative values, with varying units of measurement.
A Recent Awareness
The concept of CVR in T1D is relatively new. Until the publication of the prospective Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study in 2005, it was believed that T1D control had no impact on CVR. However, follow-up results from the same cohort of 50,000 patients, published in 2022 after 30 years of observation, revealed that CVR was 20% higher in patients who received conventional hyperglycemia-targeted treatment than those undergoing intensive treatment. The CVR increases in conjunction with diabetes duration. The study also showed that even well-controlled glycemia in T1D carries CVR (primarily due to microangiopathy), and that the most critical factor for CVR is not A1c control but rather LDL cholesterol levels.
These findings were corroborated by a Danish prospective study, which demonstrated that while CVR increased in conjunction with the number of risk factors, it was 82% higher in patients with T1D than in a control group — even in the absence of risk factors.
Key Takeaways
At diagnosis, a fundamental difference exists between T1D and T2D in terms of the urgency to address CVR. In T2D, diabetes may have progressed for years before diagnosis, necessitating immediate CVR reduction efforts. In contrast, T1D is often diagnosed in younger patients with initially low CVR, raising questions about the optimal timing for interventions such as statin prescriptions.
Recommendations
The American Diabetes Association/European Association for the Study of Diabetes guidelines (2024) include the following recommendations:
- Between ages 20 and 40, statins are recommended if at least one CVR factor is present.
- For children 10 years of age or older with T1D, the LDL target is < 1.0 g/L. Statins are prescribed if LDL exceeds 1.6 g/L without CVR factors or 1.3 g/L with at least one CVR factor.
The European Society of Cardiology guidelines (2023) include the following:
- For the first time, a dedicated chapter addresses T1D. Like the American guidelines, routine statin use after age 40 is recommended.
- Before age 40, statins are prescribed if there is at least one CVR factor (microangiopathy) or a 10-year CVR ≥ 10% (based on a CVR calculator).
The International Society for Pediatric and Adolescent Diabetes guidelines (2022) recommend:
- For children 10 years of age or older, the LDL target is < 1.0 g/L. Statins are recommended if LDL exceeds 1.3 g/L.
CAC Score in High CVR
The French Society of Cardiology and the French-speaking Society of Diabetology recommend incorporating the coronary artery calcium (CAC) score to refine CVR classification in high-risk patients. For those without prior cardiovascular events, LDL targets vary based on CAC and age. For example:
- High-risk patients with a CAC of 0-10 are reclassified as moderate risk, with an LDL target of < 1 g/L.
- A CAC ≥ 400 indicates very high risk, warranting coronary exploration.
- Patients under 50 years of age with a CAC of 11-100 remain high risk, with an LDL target of 0.7 g/L.
Conclusion
CVR in patients with T1D remains challenging to define. However, it is essential to consider long-term outcomes, planning for 30 or 40 years into the future. This involves educating patients about the importance of prevention, even when reassuring numbers are seen in their youth.
This story was translated from Univadis France using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Fifty Years Later: Preterm Birth Shows Complex Pattern of Cardiovascular Outcomes
TOPLINE:
Adults aged 50 years who were born preterm have a higher risk for hypertension but lower risk for cardiovascular events than those born at term, with similar risks for diabetes, prediabetes, and dyslipidemia between groups.
METHODOLOGY:
- The researchers conducted a prospective cohort study of the Auckland Steroid Trial — the first randomized trial of antenatal corticosteroids (betamethasone) for women who were at risk for preterm birth, conducted in Auckland, New Zealand, between December 1969 and February 1974.
- They analyzed 470 participants, including 424 survivors recruited between January 2020 and May 2022 and 46 participants who died after infancy.
- The outcomes for 326 participants born preterm (mean age, 49.4 years) and 144 participants born at term (mean age, 49.2 years) were assessed using either a questionnaire, administrative datasets, or both.
- The primary outcome was a composite of cardiovascular events or risk factors, defined as a history of a major adverse cardiovascular event or the presence of at least one cardiovascular risk factor, including diabetes mellitus, prediabetes, treated dyslipidemia, and treated hypertension.
- The secondary outcomes included respiratory, mental health, educational, and other health outcomes, as well as components of the primary outcomes.
TAKEAWAY:
- The composite of cardiovascular events or risk factors occurred in 34.5% of participants born preterm and 29.9% of participants born at term, with no differences in the risk factor components.
- The risk for cardiovascular events was lower in participants born preterm than in those born at term (adjusted relative risk [aRR], 0.33; P = .013).
- The participants born preterm had a higher risk for high blood pressure (aRR, 1.74; P = .007) and the composite of treated hypertension or self-reported diagnosis of high blood pressure (aRR, 1.63; P = .010) than those born at term.
- From randomization to the 50-year follow-up, death from any cause was more common in those born preterm than in those born at term (aRR, 2.29; P < .0001), whereas the diagnosis or treatment of a mental health disorder was less common (P = .007); no differences were observed between the groups for other outcomes.
IN PRACTICE:
“Those aware of being born preterm also may be more likely to seek preventive treatments, potentially resulting in a reduced risk of cardiovascular disease but a greater prevalence of risk factors if defined by a treatment such as treated dyslipidemia or treated hypertension,” the authors wrote.
“In this cohort, the survival advantage of the term-born control group abated after infancy, with a higher all-cause mortality rate, compared with that of the group born preterm,” wrote Jonathan S. Litt, MD, MPH, ScD, and Henning Tiemeier, MD, PhD, in a related commentary published in Pediatrics.
SOURCE:
The study was led by Anthony G. B. Walters, MBChB, Liggins Institute, Auckland, New Zealand. It was published online on December 16, 2024, in Pediatrics .
LIMITATIONS:
The small sample size limited the ability to detect subtle differences between groups and the validity of subgroup analyses. Attrition bias may have occurred because of low follow-up rates among presumed survivors. Bias could have been introduced because of lack of consent for access to the administrative dataset or from missing data from the participants in the questionnaire. The lack of in-person assessments for blood pressure and blood tests, resulting from geographical dispersion over 50 years, may have led to underestimation of some outcomes. Additionally, as most participants were born moderately or late preterm, with a median gestational age of 34.1 weeks, findings may not be generalizable to those born preterm at earlier gestational ages.
DISCLOSURES:
The study was supported in part by the Aotearoa Foundation, the Auckland Medical Research Foundation, Cure Kids New Zealand, and the Health Research Council of New Zealand. The authors of both the study and the commentary reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Adults aged 50 years who were born preterm have a higher risk for hypertension but lower risk for cardiovascular events than those born at term, with similar risks for diabetes, prediabetes, and dyslipidemia between groups.
METHODOLOGY:
- The researchers conducted a prospective cohort study of the Auckland Steroid Trial — the first randomized trial of antenatal corticosteroids (betamethasone) for women who were at risk for preterm birth, conducted in Auckland, New Zealand, between December 1969 and February 1974.
- They analyzed 470 participants, including 424 survivors recruited between January 2020 and May 2022 and 46 participants who died after infancy.
- The outcomes for 326 participants born preterm (mean age, 49.4 years) and 144 participants born at term (mean age, 49.2 years) were assessed using either a questionnaire, administrative datasets, or both.
- The primary outcome was a composite of cardiovascular events or risk factors, defined as a history of a major adverse cardiovascular event or the presence of at least one cardiovascular risk factor, including diabetes mellitus, prediabetes, treated dyslipidemia, and treated hypertension.
- The secondary outcomes included respiratory, mental health, educational, and other health outcomes, as well as components of the primary outcomes.
TAKEAWAY:
- The composite of cardiovascular events or risk factors occurred in 34.5% of participants born preterm and 29.9% of participants born at term, with no differences in the risk factor components.
- The risk for cardiovascular events was lower in participants born preterm than in those born at term (adjusted relative risk [aRR], 0.33; P = .013).
- The participants born preterm had a higher risk for high blood pressure (aRR, 1.74; P = .007) and the composite of treated hypertension or self-reported diagnosis of high blood pressure (aRR, 1.63; P = .010) than those born at term.
- From randomization to the 50-year follow-up, death from any cause was more common in those born preterm than in those born at term (aRR, 2.29; P < .0001), whereas the diagnosis or treatment of a mental health disorder was less common (P = .007); no differences were observed between the groups for other outcomes.
IN PRACTICE:
“Those aware of being born preterm also may be more likely to seek preventive treatments, potentially resulting in a reduced risk of cardiovascular disease but a greater prevalence of risk factors if defined by a treatment such as treated dyslipidemia or treated hypertension,” the authors wrote.
“In this cohort, the survival advantage of the term-born control group abated after infancy, with a higher all-cause mortality rate, compared with that of the group born preterm,” wrote Jonathan S. Litt, MD, MPH, ScD, and Henning Tiemeier, MD, PhD, in a related commentary published in Pediatrics.
SOURCE:
The study was led by Anthony G. B. Walters, MBChB, Liggins Institute, Auckland, New Zealand. It was published online on December 16, 2024, in Pediatrics .
LIMITATIONS:
The small sample size limited the ability to detect subtle differences between groups and the validity of subgroup analyses. Attrition bias may have occurred because of low follow-up rates among presumed survivors. Bias could have been introduced because of lack of consent for access to the administrative dataset or from missing data from the participants in the questionnaire. The lack of in-person assessments for blood pressure and blood tests, resulting from geographical dispersion over 50 years, may have led to underestimation of some outcomes. Additionally, as most participants were born moderately or late preterm, with a median gestational age of 34.1 weeks, findings may not be generalizable to those born preterm at earlier gestational ages.
DISCLOSURES:
The study was supported in part by the Aotearoa Foundation, the Auckland Medical Research Foundation, Cure Kids New Zealand, and the Health Research Council of New Zealand. The authors of both the study and the commentary reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Adults aged 50 years who were born preterm have a higher risk for hypertension but lower risk for cardiovascular events than those born at term, with similar risks for diabetes, prediabetes, and dyslipidemia between groups.
METHODOLOGY:
- The researchers conducted a prospective cohort study of the Auckland Steroid Trial — the first randomized trial of antenatal corticosteroids (betamethasone) for women who were at risk for preterm birth, conducted in Auckland, New Zealand, between December 1969 and February 1974.
- They analyzed 470 participants, including 424 survivors recruited between January 2020 and May 2022 and 46 participants who died after infancy.
- The outcomes for 326 participants born preterm (mean age, 49.4 years) and 144 participants born at term (mean age, 49.2 years) were assessed using either a questionnaire, administrative datasets, or both.
- The primary outcome was a composite of cardiovascular events or risk factors, defined as a history of a major adverse cardiovascular event or the presence of at least one cardiovascular risk factor, including diabetes mellitus, prediabetes, treated dyslipidemia, and treated hypertension.
- The secondary outcomes included respiratory, mental health, educational, and other health outcomes, as well as components of the primary outcomes.
TAKEAWAY:
- The composite of cardiovascular events or risk factors occurred in 34.5% of participants born preterm and 29.9% of participants born at term, with no differences in the risk factor components.
- The risk for cardiovascular events was lower in participants born preterm than in those born at term (adjusted relative risk [aRR], 0.33; P = .013).
- The participants born preterm had a higher risk for high blood pressure (aRR, 1.74; P = .007) and the composite of treated hypertension or self-reported diagnosis of high blood pressure (aRR, 1.63; P = .010) than those born at term.
- From randomization to the 50-year follow-up, death from any cause was more common in those born preterm than in those born at term (aRR, 2.29; P < .0001), whereas the diagnosis or treatment of a mental health disorder was less common (P = .007); no differences were observed between the groups for other outcomes.
IN PRACTICE:
“Those aware of being born preterm also may be more likely to seek preventive treatments, potentially resulting in a reduced risk of cardiovascular disease but a greater prevalence of risk factors if defined by a treatment such as treated dyslipidemia or treated hypertension,” the authors wrote.
“In this cohort, the survival advantage of the term-born control group abated after infancy, with a higher all-cause mortality rate, compared with that of the group born preterm,” wrote Jonathan S. Litt, MD, MPH, ScD, and Henning Tiemeier, MD, PhD, in a related commentary published in Pediatrics.
SOURCE:
The study was led by Anthony G. B. Walters, MBChB, Liggins Institute, Auckland, New Zealand. It was published online on December 16, 2024, in Pediatrics .
LIMITATIONS:
The small sample size limited the ability to detect subtle differences between groups and the validity of subgroup analyses. Attrition bias may have occurred because of low follow-up rates among presumed survivors. Bias could have been introduced because of lack of consent for access to the administrative dataset or from missing data from the participants in the questionnaire. The lack of in-person assessments for blood pressure and blood tests, resulting from geographical dispersion over 50 years, may have led to underestimation of some outcomes. Additionally, as most participants were born moderately or late preterm, with a median gestational age of 34.1 weeks, findings may not be generalizable to those born preterm at earlier gestational ages.
DISCLOSURES:
The study was supported in part by the Aotearoa Foundation, the Auckland Medical Research Foundation, Cure Kids New Zealand, and the Health Research Council of New Zealand. The authors of both the study and the commentary reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Gambling Disorder on the Rise, but Often Overlooked
On a recent Sunday morning in Los Angeles, 10 members of Gamblers Anonymous gathered in a park for their regular meeting. After, they shared advice for how physicians can best help patients with problem gambling.
“If a patient talks about financial distress, spouse issues, physical issues, or has blood pressure issues, suspect gambling,” one woman said. Another said, if a physician asks about gambling and the patient says, “Just a little,” chances are that person is an active gambler.
The bottom line: None of the people — who spoke for themselves and not on behalf of Gamblers Anonymous — said they had been asked by their doctors about gambling issues. All said they would welcome such questions.
Gambling is on the rise, and gambling disorder is now recognized in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). The condition is viewed as similar to substance-related disorders in its clinical expression, brain origin, comorbidity, physiology, and treatment.
according to experts, as it often intertwines with physical and mental health concerns. Those conditions bring the patient to their clinicians, although the patients may not link those issues with their gambling. The physicians may not either.
Few PCPs broach the topic. “I would say the majority of physicians do not screen for it,” said Brian K. Unwin, MD, FAAFP, AGSF, a family medicine physician and geriatrician at the Carilion Center for Healthy Aging and professor at the Virginia Tech Carilion School of Medicine, Roanoke.
The clinician who does take steps to screen for the problem is “exceptionally rare,” said Timothy W. Fong, MD, clinical professor of psychiatry at the University of California, Los Angeles (UCLA) and co-director of the UCLA Gambling Studies Program. Launched in 2005, the program conducts research, provides prevention and treatment services, and offers training to healthcare providers and the community.
A Las Vegas physician said colleagues in her area are likewise largely unaware, despite the strong connection with the city and gaming. “I do not think most primary care physicians are routinely asking about gambling,” said Maureen Strohm, MD, president of the Nevada Society of Addiction Medicine. Strohm also directs the addiction medicine fellowship at HCA Sunrise Health GME Consortium at Southern Hills Hospital & Medical Center in Las Vegas and cares for patients with substance abuse issues.
But physicians should look for gambling issues, she said, especially in those with known substance abuse disorders. “We encourage incorporation of gambling as another question in comprehensive assessment of patients, since it’s rarely an isolated issue in our treatment settings,” Strohm said.
Gritty Reputation Goes Glam, With No Shortage of Opportunities
Why a rise in gambling? Its reputation, for one thing. “It used to be, if you enjoyed gambling, you were viewed as, what — a person of vice, a person of sin,” Fong said. “That’s completely changed. Engaging in gambling is more accepted — and not just accepted but normalized and even glamorized. In some circles, if you don’t gamble, it’s like, ‘What is wrong with you?’ ”
Opportunities to gamble have increased, including a boom in mobile sports betting. Sports betting online, in person, or both now is legal in 39 states and the District of Columbia. The rate of gambling problems among sports bettors is at least twice as high as that for other gamblers, the National Council on Problem Gambling found. As Fong puts it: “The casino comes to you.”
With the rise in opportunities to bet has come an increase in gambling-related disorders. Statistics vary greatly, but Unwin cites a meta-analysis published in 2023 that found moderate-risk or at-risk gambling affects 2.4% of the adult population and pathologic or problem gambling affects 1.29%. He first noticed problem gambling in young soldiers when he was a military doctor and published on it in 2000.
However, the percentage of people with gambling issues seeking care in PCPs’ offices is much higher, probably from 5% to 16%, research has found. “When you survey people who go to the primary care physicians, the number [with a gambling issue] could be as high as 10%-15% of those going for any medical reason,” Fong said. “Many times, their stories are hidden.”
In November, The Lancet Public Health Commission said it views problem gambling as an expanding public health threat.
Seeing the Red Flags
“In a perfect world, it would be great to ask all patients” about gambling issues, Unwin said. A more realistic approach, given clinical workloads, is to be alert to the possibility, especially in patients with depression or substance abuse, which often accompany gambling issues.
Learn to look at patterns, Unwin said. “If a patient has had impulsivity issues, is a young male, has had depression and alcohol issues, let’s look and see what else is going on.”
Other red flags include anyone with an active mental health problem or with a family history of known gambling problems, Fong said.
Some personality traits are linked with a higher likelihood of gambling issues, including highly impulsive behavior and risk-taking behavior. Many problem gamblers are “not very good with loss aversion. They lose a bunch and shrug it off and go back the next day,” Fong noted.
Often, however, the clues are not obvious. Unwin remembers caring for an older couple, and the wife set up an appointment with him — not to talk about her health but to discuss his gambling. “My husband has spent us into debt,” she said. Unwin recalled being completely surprised.
The situation illustrates the flaw in the stereotypical profile of a problem. “In our mind’s eye, it’s often an older White male who talks loudly, is perhaps counterculture,” Fong said. But he often sees young and older people from all cultures and all economic levels struggling with gambling: “Like other forms of addiction, it cuts across all demographics.”
Inside a Gambler’s Brain
Many physicians struggle to understand the attraction of gambling, Fong said. They ask: “How can you be addicted to a behavior? Why can’t you just stop?” He tells them: “If people could do that, they would not have a biological psychiatric disorder,” which is what gambling disorder is.
The urge to gamble can be so strong, “You can’t think of anything else,” Fong said. “It screws up your day.” Gamblers say they’re after the “action,” the euphoric state similar to the highs produced from some drugs. Compared with recreational gamblers, problem gamblers rely more on long-term learning than short-term reward?, and so are less able to learn from their losses in the immediate past, research by Fong and others found.
Seeking Treatment
One in five problem gamblers seeks help, and 1 in 25 with a moderate-risk habit do so, Unwin said, citing a 2022 study.
To identify concerning behaviors, physicians can turn to two brief screeners that take just minutes:
- The Brief Biosocial Gambling Screen includes just three questions; a yes to any one suggests a gambling problem.
- The Lie-Bet two-question screener can help determine if a person needs a referral for a gambling problem.
“People tend to be pretty honest with their doctor when asked about gambling,” Fong said. “Even the act of asking is enough to get people to start thinking.”
To meet the DSM-5 criteria for gambling disorder, patients must exhibit at least four or more concerning behaviors in the previous 12 months.
For available treatments, “our toolbox is growing,” Fong said. “Psychotherapy probably works best,” including cognitive-behavioral therapy and relapse prevention approaches. “Twelve-step programs work very well,” Fong added.
Medications used for alcohol use disorder, such as naltrexone, an opioid antagonist, are prescribed for gambling disorder, with some success, he said. Often, developing a positive therapeutic relationship with a person who does not judge them is enough to change behavior, Fong said.
To provide treatment and other services, the UCLA program collaborates with the state Office of Problem Gambling. “We know that at least 70% of our patients who stay in treatment and participate in treatment make really meaningful gains and improvement in some part of their lives,” Fong said. “They do gamble less; they do gamble with less money.
Goal: Cold Turkey or Harm Reduction?
Fong tells gamblers seeking help: “My goal is to reduce the harm related to your gambling.” In working with patients, he identifies forms that are most problematic and those less likely to cause problems. For some, sports betting may be an issue, but going to Las Vegas a few times a year to play the slots may not generate harm for them. Many patients still gamble, he said, but have a better quality of life if they focus on health and wellness.
“Abstinence is just one domain,” Fong said. The others — home, health, self-care, a sense of purpose, community — are important, too. He helps patients to focus on those.”
Of all the addictions, gambling is one physicians are largely unaware of, Fong said. “And the patients have something that can be treated.”
Unwin, Strohm, and Fong reported no relevant financial disclosures. Physicians can attend open meetings of Gamblers Anonymous to find out more. Members of the group are available to speak to clinicians.
A version of this article appeared on Medscape.com.
On a recent Sunday morning in Los Angeles, 10 members of Gamblers Anonymous gathered in a park for their regular meeting. After, they shared advice for how physicians can best help patients with problem gambling.
“If a patient talks about financial distress, spouse issues, physical issues, or has blood pressure issues, suspect gambling,” one woman said. Another said, if a physician asks about gambling and the patient says, “Just a little,” chances are that person is an active gambler.
The bottom line: None of the people — who spoke for themselves and not on behalf of Gamblers Anonymous — said they had been asked by their doctors about gambling issues. All said they would welcome such questions.
Gambling is on the rise, and gambling disorder is now recognized in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). The condition is viewed as similar to substance-related disorders in its clinical expression, brain origin, comorbidity, physiology, and treatment.
according to experts, as it often intertwines with physical and mental health concerns. Those conditions bring the patient to their clinicians, although the patients may not link those issues with their gambling. The physicians may not either.
Few PCPs broach the topic. “I would say the majority of physicians do not screen for it,” said Brian K. Unwin, MD, FAAFP, AGSF, a family medicine physician and geriatrician at the Carilion Center for Healthy Aging and professor at the Virginia Tech Carilion School of Medicine, Roanoke.
The clinician who does take steps to screen for the problem is “exceptionally rare,” said Timothy W. Fong, MD, clinical professor of psychiatry at the University of California, Los Angeles (UCLA) and co-director of the UCLA Gambling Studies Program. Launched in 2005, the program conducts research, provides prevention and treatment services, and offers training to healthcare providers and the community.
A Las Vegas physician said colleagues in her area are likewise largely unaware, despite the strong connection with the city and gaming. “I do not think most primary care physicians are routinely asking about gambling,” said Maureen Strohm, MD, president of the Nevada Society of Addiction Medicine. Strohm also directs the addiction medicine fellowship at HCA Sunrise Health GME Consortium at Southern Hills Hospital & Medical Center in Las Vegas and cares for patients with substance abuse issues.
But physicians should look for gambling issues, she said, especially in those with known substance abuse disorders. “We encourage incorporation of gambling as another question in comprehensive assessment of patients, since it’s rarely an isolated issue in our treatment settings,” Strohm said.
Gritty Reputation Goes Glam, With No Shortage of Opportunities
Why a rise in gambling? Its reputation, for one thing. “It used to be, if you enjoyed gambling, you were viewed as, what — a person of vice, a person of sin,” Fong said. “That’s completely changed. Engaging in gambling is more accepted — and not just accepted but normalized and even glamorized. In some circles, if you don’t gamble, it’s like, ‘What is wrong with you?’ ”
Opportunities to gamble have increased, including a boom in mobile sports betting. Sports betting online, in person, or both now is legal in 39 states and the District of Columbia. The rate of gambling problems among sports bettors is at least twice as high as that for other gamblers, the National Council on Problem Gambling found. As Fong puts it: “The casino comes to you.”
With the rise in opportunities to bet has come an increase in gambling-related disorders. Statistics vary greatly, but Unwin cites a meta-analysis published in 2023 that found moderate-risk or at-risk gambling affects 2.4% of the adult population and pathologic or problem gambling affects 1.29%. He first noticed problem gambling in young soldiers when he was a military doctor and published on it in 2000.
However, the percentage of people with gambling issues seeking care in PCPs’ offices is much higher, probably from 5% to 16%, research has found. “When you survey people who go to the primary care physicians, the number [with a gambling issue] could be as high as 10%-15% of those going for any medical reason,” Fong said. “Many times, their stories are hidden.”
In November, The Lancet Public Health Commission said it views problem gambling as an expanding public health threat.
Seeing the Red Flags
“In a perfect world, it would be great to ask all patients” about gambling issues, Unwin said. A more realistic approach, given clinical workloads, is to be alert to the possibility, especially in patients with depression or substance abuse, which often accompany gambling issues.
Learn to look at patterns, Unwin said. “If a patient has had impulsivity issues, is a young male, has had depression and alcohol issues, let’s look and see what else is going on.”
Other red flags include anyone with an active mental health problem or with a family history of known gambling problems, Fong said.
Some personality traits are linked with a higher likelihood of gambling issues, including highly impulsive behavior and risk-taking behavior. Many problem gamblers are “not very good with loss aversion. They lose a bunch and shrug it off and go back the next day,” Fong noted.
Often, however, the clues are not obvious. Unwin remembers caring for an older couple, and the wife set up an appointment with him — not to talk about her health but to discuss his gambling. “My husband has spent us into debt,” she said. Unwin recalled being completely surprised.
The situation illustrates the flaw in the stereotypical profile of a problem. “In our mind’s eye, it’s often an older White male who talks loudly, is perhaps counterculture,” Fong said. But he often sees young and older people from all cultures and all economic levels struggling with gambling: “Like other forms of addiction, it cuts across all demographics.”
Inside a Gambler’s Brain
Many physicians struggle to understand the attraction of gambling, Fong said. They ask: “How can you be addicted to a behavior? Why can’t you just stop?” He tells them: “If people could do that, they would not have a biological psychiatric disorder,” which is what gambling disorder is.
The urge to gamble can be so strong, “You can’t think of anything else,” Fong said. “It screws up your day.” Gamblers say they’re after the “action,” the euphoric state similar to the highs produced from some drugs. Compared with recreational gamblers, problem gamblers rely more on long-term learning than short-term reward?, and so are less able to learn from their losses in the immediate past, research by Fong and others found.
Seeking Treatment
One in five problem gamblers seeks help, and 1 in 25 with a moderate-risk habit do so, Unwin said, citing a 2022 study.
To identify concerning behaviors, physicians can turn to two brief screeners that take just minutes:
- The Brief Biosocial Gambling Screen includes just three questions; a yes to any one suggests a gambling problem.
- The Lie-Bet two-question screener can help determine if a person needs a referral for a gambling problem.
“People tend to be pretty honest with their doctor when asked about gambling,” Fong said. “Even the act of asking is enough to get people to start thinking.”
To meet the DSM-5 criteria for gambling disorder, patients must exhibit at least four or more concerning behaviors in the previous 12 months.
For available treatments, “our toolbox is growing,” Fong said. “Psychotherapy probably works best,” including cognitive-behavioral therapy and relapse prevention approaches. “Twelve-step programs work very well,” Fong added.
Medications used for alcohol use disorder, such as naltrexone, an opioid antagonist, are prescribed for gambling disorder, with some success, he said. Often, developing a positive therapeutic relationship with a person who does not judge them is enough to change behavior, Fong said.
To provide treatment and other services, the UCLA program collaborates with the state Office of Problem Gambling. “We know that at least 70% of our patients who stay in treatment and participate in treatment make really meaningful gains and improvement in some part of their lives,” Fong said. “They do gamble less; they do gamble with less money.
Goal: Cold Turkey or Harm Reduction?
Fong tells gamblers seeking help: “My goal is to reduce the harm related to your gambling.” In working with patients, he identifies forms that are most problematic and those less likely to cause problems. For some, sports betting may be an issue, but going to Las Vegas a few times a year to play the slots may not generate harm for them. Many patients still gamble, he said, but have a better quality of life if they focus on health and wellness.
“Abstinence is just one domain,” Fong said. The others — home, health, self-care, a sense of purpose, community — are important, too. He helps patients to focus on those.”
Of all the addictions, gambling is one physicians are largely unaware of, Fong said. “And the patients have something that can be treated.”
Unwin, Strohm, and Fong reported no relevant financial disclosures. Physicians can attend open meetings of Gamblers Anonymous to find out more. Members of the group are available to speak to clinicians.
A version of this article appeared on Medscape.com.
On a recent Sunday morning in Los Angeles, 10 members of Gamblers Anonymous gathered in a park for their regular meeting. After, they shared advice for how physicians can best help patients with problem gambling.
“If a patient talks about financial distress, spouse issues, physical issues, or has blood pressure issues, suspect gambling,” one woman said. Another said, if a physician asks about gambling and the patient says, “Just a little,” chances are that person is an active gambler.
The bottom line: None of the people — who spoke for themselves and not on behalf of Gamblers Anonymous — said they had been asked by their doctors about gambling issues. All said they would welcome such questions.
Gambling is on the rise, and gambling disorder is now recognized in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). The condition is viewed as similar to substance-related disorders in its clinical expression, brain origin, comorbidity, physiology, and treatment.
according to experts, as it often intertwines with physical and mental health concerns. Those conditions bring the patient to their clinicians, although the patients may not link those issues with their gambling. The physicians may not either.
Few PCPs broach the topic. “I would say the majority of physicians do not screen for it,” said Brian K. Unwin, MD, FAAFP, AGSF, a family medicine physician and geriatrician at the Carilion Center for Healthy Aging and professor at the Virginia Tech Carilion School of Medicine, Roanoke.
The clinician who does take steps to screen for the problem is “exceptionally rare,” said Timothy W. Fong, MD, clinical professor of psychiatry at the University of California, Los Angeles (UCLA) and co-director of the UCLA Gambling Studies Program. Launched in 2005, the program conducts research, provides prevention and treatment services, and offers training to healthcare providers and the community.
A Las Vegas physician said colleagues in her area are likewise largely unaware, despite the strong connection with the city and gaming. “I do not think most primary care physicians are routinely asking about gambling,” said Maureen Strohm, MD, president of the Nevada Society of Addiction Medicine. Strohm also directs the addiction medicine fellowship at HCA Sunrise Health GME Consortium at Southern Hills Hospital & Medical Center in Las Vegas and cares for patients with substance abuse issues.
But physicians should look for gambling issues, she said, especially in those with known substance abuse disorders. “We encourage incorporation of gambling as another question in comprehensive assessment of patients, since it’s rarely an isolated issue in our treatment settings,” Strohm said.
Gritty Reputation Goes Glam, With No Shortage of Opportunities
Why a rise in gambling? Its reputation, for one thing. “It used to be, if you enjoyed gambling, you were viewed as, what — a person of vice, a person of sin,” Fong said. “That’s completely changed. Engaging in gambling is more accepted — and not just accepted but normalized and even glamorized. In some circles, if you don’t gamble, it’s like, ‘What is wrong with you?’ ”
Opportunities to gamble have increased, including a boom in mobile sports betting. Sports betting online, in person, or both now is legal in 39 states and the District of Columbia. The rate of gambling problems among sports bettors is at least twice as high as that for other gamblers, the National Council on Problem Gambling found. As Fong puts it: “The casino comes to you.”
With the rise in opportunities to bet has come an increase in gambling-related disorders. Statistics vary greatly, but Unwin cites a meta-analysis published in 2023 that found moderate-risk or at-risk gambling affects 2.4% of the adult population and pathologic or problem gambling affects 1.29%. He first noticed problem gambling in young soldiers when he was a military doctor and published on it in 2000.
However, the percentage of people with gambling issues seeking care in PCPs’ offices is much higher, probably from 5% to 16%, research has found. “When you survey people who go to the primary care physicians, the number [with a gambling issue] could be as high as 10%-15% of those going for any medical reason,” Fong said. “Many times, their stories are hidden.”
In November, The Lancet Public Health Commission said it views problem gambling as an expanding public health threat.
Seeing the Red Flags
“In a perfect world, it would be great to ask all patients” about gambling issues, Unwin said. A more realistic approach, given clinical workloads, is to be alert to the possibility, especially in patients with depression or substance abuse, which often accompany gambling issues.
Learn to look at patterns, Unwin said. “If a patient has had impulsivity issues, is a young male, has had depression and alcohol issues, let’s look and see what else is going on.”
Other red flags include anyone with an active mental health problem or with a family history of known gambling problems, Fong said.
Some personality traits are linked with a higher likelihood of gambling issues, including highly impulsive behavior and risk-taking behavior. Many problem gamblers are “not very good with loss aversion. They lose a bunch and shrug it off and go back the next day,” Fong noted.
Often, however, the clues are not obvious. Unwin remembers caring for an older couple, and the wife set up an appointment with him — not to talk about her health but to discuss his gambling. “My husband has spent us into debt,” she said. Unwin recalled being completely surprised.
The situation illustrates the flaw in the stereotypical profile of a problem. “In our mind’s eye, it’s often an older White male who talks loudly, is perhaps counterculture,” Fong said. But he often sees young and older people from all cultures and all economic levels struggling with gambling: “Like other forms of addiction, it cuts across all demographics.”
Inside a Gambler’s Brain
Many physicians struggle to understand the attraction of gambling, Fong said. They ask: “How can you be addicted to a behavior? Why can’t you just stop?” He tells them: “If people could do that, they would not have a biological psychiatric disorder,” which is what gambling disorder is.
The urge to gamble can be so strong, “You can’t think of anything else,” Fong said. “It screws up your day.” Gamblers say they’re after the “action,” the euphoric state similar to the highs produced from some drugs. Compared with recreational gamblers, problem gamblers rely more on long-term learning than short-term reward?, and so are less able to learn from their losses in the immediate past, research by Fong and others found.
Seeking Treatment
One in five problem gamblers seeks help, and 1 in 25 with a moderate-risk habit do so, Unwin said, citing a 2022 study.
To identify concerning behaviors, physicians can turn to two brief screeners that take just minutes:
- The Brief Biosocial Gambling Screen includes just three questions; a yes to any one suggests a gambling problem.
- The Lie-Bet two-question screener can help determine if a person needs a referral for a gambling problem.
“People tend to be pretty honest with their doctor when asked about gambling,” Fong said. “Even the act of asking is enough to get people to start thinking.”
To meet the DSM-5 criteria for gambling disorder, patients must exhibit at least four or more concerning behaviors in the previous 12 months.
For available treatments, “our toolbox is growing,” Fong said. “Psychotherapy probably works best,” including cognitive-behavioral therapy and relapse prevention approaches. “Twelve-step programs work very well,” Fong added.
Medications used for alcohol use disorder, such as naltrexone, an opioid antagonist, are prescribed for gambling disorder, with some success, he said. Often, developing a positive therapeutic relationship with a person who does not judge them is enough to change behavior, Fong said.
To provide treatment and other services, the UCLA program collaborates with the state Office of Problem Gambling. “We know that at least 70% of our patients who stay in treatment and participate in treatment make really meaningful gains and improvement in some part of their lives,” Fong said. “They do gamble less; they do gamble with less money.
Goal: Cold Turkey or Harm Reduction?
Fong tells gamblers seeking help: “My goal is to reduce the harm related to your gambling.” In working with patients, he identifies forms that are most problematic and those less likely to cause problems. For some, sports betting may be an issue, but going to Las Vegas a few times a year to play the slots may not generate harm for them. Many patients still gamble, he said, but have a better quality of life if they focus on health and wellness.
“Abstinence is just one domain,” Fong said. The others — home, health, self-care, a sense of purpose, community — are important, too. He helps patients to focus on those.”
Of all the addictions, gambling is one physicians are largely unaware of, Fong said. “And the patients have something that can be treated.”
Unwin, Strohm, and Fong reported no relevant financial disclosures. Physicians can attend open meetings of Gamblers Anonymous to find out more. Members of the group are available to speak to clinicians.
A version of this article appeared on Medscape.com.