User login
Vedolizumab shows long-term safety, efficacy
The gut-selective alpha4beta7 integrin antibody vedolizumab is safe and effective for long-term use in patients with inflammatory bowel disease (IBD), according to data from more than 2,200 patients in the GEMINI LTS trial.
With a median cumulative exposure of approximately 3 years and some patients taking vedolizumab for more than 9 years, the study revealed no new safety concerns and showed that responses were stable over time, reported lead author Edward V. Loftus Jr., MD, of the Mayo Clinic in Rochester, Minn., and colleagues.
“Interim analyses (based on 4 years of follow-up) demonstrated that long-term vedolizumab therapy was well-tolerated and also provided clinical and health-related quality of life (HRQOL) benefits,” the investigators wrote in Alimentary Pharmacology & Therapeutics. “In this final analysis ... we report the final safety outcomes, along with exploratory clinical and HRQOL outcomes.”
The phase 3 trial involved 1,822 patients with IBD from previous phase 2 and 3 trials, plus 421 vedolizumab-naive patients. Out of 2,243 participants in the final analysis, 894 had ulcerative colitis, and 1,349 had Crohn’s disease.
All patients received vedolizumab 300 mg IV every 4 weeks, which the investigators noted is more frequent than the dosing interval of 8 weeks that was approved after the trial was designed.
Median cumulative exposure times among patients with ulcerative colitis and Crohn’s disease were 42.4 months and 31.5 months, respectively. Adverse events of any grade occurred in 93% of patients with ulcerative colitis and 96% of patients with Crohn’s disease, approximately one-third of which were exacerbations of IBD. Serious adverse events occurred in 31% and 41% of patients with ulcerative colitis and Crohn’s disease, respectively. Adverse events led to discontinuation in 15% of ulcerative colitis patients and 17% of those with Crohn’s disease. Of the 10 deaths that occurred during the study period, the investigators categorized 2 of them as drug related (hepatocellular carcinoma and West Nile virus infection–related encephalitis). No increase in the rate of overall malignancies was observed.
Dr. Loftus and colleagues noted that rates of serious infection with vedolizumab treatment were superior to historical long-term data for adalimumab, at 18.0 (ulcerative colitis) and 33.6 (Crohn’s disease) per 1,000 person-years for vedolizumab, compared with 35 (ulcerative colitis) and 67 (Crohn’s disease) per 1,000 person-years for adalimumab, as reported by Colombel and colleagues.
“While these data suggest a decreased risk of systemic infections with vedolizumab, an increased risk of gastrointestinal infections is plausible given the gut-selective mechanism of action and evidence for reduced immune response in the gastrointestinal tract,” the investigators wrote, reporting rates of 34.9 and 39.6 per 1,000 person-years for ulcerative colitis and Crohn’s disease, respectively. Comparative, historical rates for adalimumab were not provided.
In the present trial, clinical response rates, clinical remission rates, and HRQOL estimates remained stable over time. At 400 treatment weeks, clinical remission was maintained in 33% and 28% of patients with ulcerative colitis and Crohn’s disease, respectively.
“[T]he final analysis of GEMINI LTS comprehensively demonstrates that vedolizumab therapy has a safety and tolerability profile suitable for long-term treatment of patients with moderately to severely active ulcerative colitis or Crohn’s disease,” the investigators concluded.
According to Randy Longman, MD, director of the Jill Roberts Center for Inflammatory Bowel Disease at Weill Cornell Medicine and New York-Presbyterian, “The results from the GEMINI LTS trial help to solidify the favorable safety profile of vedolizumab for the treatment of IBD. With large patient numbers and cumulative duration of vedolizumab exposure, these pivotal data convincingly reveal low rates of serious infections, identified no cases of progressive multifocal leukoencephalopathy, and showed no increased risk of malignancy. Arthralgias, or joint pain, was the most common treatment-emergent adverse event, but occurred largely in patients with a history of IBD-associated arthralgia and suggest that these symptoms should be monitored during treatment. Overall, these results provide meaningful reassurance to patients and providers using vedolizumab for the treatment of IBD.”
GEMINI LTS was funded by Takeda. The investigators reported additional relationships with AbbVie, Janssen, Amgen, and others. Dr. Longman reported no conflicts of interest.
SOURCE: Loftus Jr EV et al. AP&T. 2020 Sep 2. doi: 10.1111/apt.16060.
Help your patients better understand their IBD treatment options by sharing AGA’s patient education, “Living with IBD,” in the AGA GI Patient Center at http://ow.ly/szHz30rdKyx.
The gut-selective alpha4beta7 integrin antibody vedolizumab is safe and effective for long-term use in patients with inflammatory bowel disease (IBD), according to data from more than 2,200 patients in the GEMINI LTS trial.
With a median cumulative exposure of approximately 3 years and some patients taking vedolizumab for more than 9 years, the study revealed no new safety concerns and showed that responses were stable over time, reported lead author Edward V. Loftus Jr., MD, of the Mayo Clinic in Rochester, Minn., and colleagues.
“Interim analyses (based on 4 years of follow-up) demonstrated that long-term vedolizumab therapy was well-tolerated and also provided clinical and health-related quality of life (HRQOL) benefits,” the investigators wrote in Alimentary Pharmacology & Therapeutics. “In this final analysis ... we report the final safety outcomes, along with exploratory clinical and HRQOL outcomes.”
The phase 3 trial involved 1,822 patients with IBD from previous phase 2 and 3 trials, plus 421 vedolizumab-naive patients. Out of 2,243 participants in the final analysis, 894 had ulcerative colitis, and 1,349 had Crohn’s disease.
All patients received vedolizumab 300 mg IV every 4 weeks, which the investigators noted is more frequent than the dosing interval of 8 weeks that was approved after the trial was designed.
Median cumulative exposure times among patients with ulcerative colitis and Crohn’s disease were 42.4 months and 31.5 months, respectively. Adverse events of any grade occurred in 93% of patients with ulcerative colitis and 96% of patients with Crohn’s disease, approximately one-third of which were exacerbations of IBD. Serious adverse events occurred in 31% and 41% of patients with ulcerative colitis and Crohn’s disease, respectively. Adverse events led to discontinuation in 15% of ulcerative colitis patients and 17% of those with Crohn’s disease. Of the 10 deaths that occurred during the study period, the investigators categorized 2 of them as drug related (hepatocellular carcinoma and West Nile virus infection–related encephalitis). No increase in the rate of overall malignancies was observed.
Dr. Loftus and colleagues noted that rates of serious infection with vedolizumab treatment were superior to historical long-term data for adalimumab, at 18.0 (ulcerative colitis) and 33.6 (Crohn’s disease) per 1,000 person-years for vedolizumab, compared with 35 (ulcerative colitis) and 67 (Crohn’s disease) per 1,000 person-years for adalimumab, as reported by Colombel and colleagues.
“While these data suggest a decreased risk of systemic infections with vedolizumab, an increased risk of gastrointestinal infections is plausible given the gut-selective mechanism of action and evidence for reduced immune response in the gastrointestinal tract,” the investigators wrote, reporting rates of 34.9 and 39.6 per 1,000 person-years for ulcerative colitis and Crohn’s disease, respectively. Comparative, historical rates for adalimumab were not provided.
In the present trial, clinical response rates, clinical remission rates, and HRQOL estimates remained stable over time. At 400 treatment weeks, clinical remission was maintained in 33% and 28% of patients with ulcerative colitis and Crohn’s disease, respectively.
“[T]he final analysis of GEMINI LTS comprehensively demonstrates that vedolizumab therapy has a safety and tolerability profile suitable for long-term treatment of patients with moderately to severely active ulcerative colitis or Crohn’s disease,” the investigators concluded.
According to Randy Longman, MD, director of the Jill Roberts Center for Inflammatory Bowel Disease at Weill Cornell Medicine and New York-Presbyterian, “The results from the GEMINI LTS trial help to solidify the favorable safety profile of vedolizumab for the treatment of IBD. With large patient numbers and cumulative duration of vedolizumab exposure, these pivotal data convincingly reveal low rates of serious infections, identified no cases of progressive multifocal leukoencephalopathy, and showed no increased risk of malignancy. Arthralgias, or joint pain, was the most common treatment-emergent adverse event, but occurred largely in patients with a history of IBD-associated arthralgia and suggest that these symptoms should be monitored during treatment. Overall, these results provide meaningful reassurance to patients and providers using vedolizumab for the treatment of IBD.”
GEMINI LTS was funded by Takeda. The investigators reported additional relationships with AbbVie, Janssen, Amgen, and others. Dr. Longman reported no conflicts of interest.
SOURCE: Loftus Jr EV et al. AP&T. 2020 Sep 2. doi: 10.1111/apt.16060.
Help your patients better understand their IBD treatment options by sharing AGA’s patient education, “Living with IBD,” in the AGA GI Patient Center at http://ow.ly/szHz30rdKyx.
The gut-selective alpha4beta7 integrin antibody vedolizumab is safe and effective for long-term use in patients with inflammatory bowel disease (IBD), according to data from more than 2,200 patients in the GEMINI LTS trial.
With a median cumulative exposure of approximately 3 years and some patients taking vedolizumab for more than 9 years, the study revealed no new safety concerns and showed that responses were stable over time, reported lead author Edward V. Loftus Jr., MD, of the Mayo Clinic in Rochester, Minn., and colleagues.
“Interim analyses (based on 4 years of follow-up) demonstrated that long-term vedolizumab therapy was well-tolerated and also provided clinical and health-related quality of life (HRQOL) benefits,” the investigators wrote in Alimentary Pharmacology & Therapeutics. “In this final analysis ... we report the final safety outcomes, along with exploratory clinical and HRQOL outcomes.”
The phase 3 trial involved 1,822 patients with IBD from previous phase 2 and 3 trials, plus 421 vedolizumab-naive patients. Out of 2,243 participants in the final analysis, 894 had ulcerative colitis, and 1,349 had Crohn’s disease.
All patients received vedolizumab 300 mg IV every 4 weeks, which the investigators noted is more frequent than the dosing interval of 8 weeks that was approved after the trial was designed.
Median cumulative exposure times among patients with ulcerative colitis and Crohn’s disease were 42.4 months and 31.5 months, respectively. Adverse events of any grade occurred in 93% of patients with ulcerative colitis and 96% of patients with Crohn’s disease, approximately one-third of which were exacerbations of IBD. Serious adverse events occurred in 31% and 41% of patients with ulcerative colitis and Crohn’s disease, respectively. Adverse events led to discontinuation in 15% of ulcerative colitis patients and 17% of those with Crohn’s disease. Of the 10 deaths that occurred during the study period, the investigators categorized 2 of them as drug related (hepatocellular carcinoma and West Nile virus infection–related encephalitis). No increase in the rate of overall malignancies was observed.
Dr. Loftus and colleagues noted that rates of serious infection with vedolizumab treatment were superior to historical long-term data for adalimumab, at 18.0 (ulcerative colitis) and 33.6 (Crohn’s disease) per 1,000 person-years for vedolizumab, compared with 35 (ulcerative colitis) and 67 (Crohn’s disease) per 1,000 person-years for adalimumab, as reported by Colombel and colleagues.
“While these data suggest a decreased risk of systemic infections with vedolizumab, an increased risk of gastrointestinal infections is plausible given the gut-selective mechanism of action and evidence for reduced immune response in the gastrointestinal tract,” the investigators wrote, reporting rates of 34.9 and 39.6 per 1,000 person-years for ulcerative colitis and Crohn’s disease, respectively. Comparative, historical rates for adalimumab were not provided.
In the present trial, clinical response rates, clinical remission rates, and HRQOL estimates remained stable over time. At 400 treatment weeks, clinical remission was maintained in 33% and 28% of patients with ulcerative colitis and Crohn’s disease, respectively.
“[T]he final analysis of GEMINI LTS comprehensively demonstrates that vedolizumab therapy has a safety and tolerability profile suitable for long-term treatment of patients with moderately to severely active ulcerative colitis or Crohn’s disease,” the investigators concluded.
According to Randy Longman, MD, director of the Jill Roberts Center for Inflammatory Bowel Disease at Weill Cornell Medicine and New York-Presbyterian, “The results from the GEMINI LTS trial help to solidify the favorable safety profile of vedolizumab for the treatment of IBD. With large patient numbers and cumulative duration of vedolizumab exposure, these pivotal data convincingly reveal low rates of serious infections, identified no cases of progressive multifocal leukoencephalopathy, and showed no increased risk of malignancy. Arthralgias, or joint pain, was the most common treatment-emergent adverse event, but occurred largely in patients with a history of IBD-associated arthralgia and suggest that these symptoms should be monitored during treatment. Overall, these results provide meaningful reassurance to patients and providers using vedolizumab for the treatment of IBD.”
GEMINI LTS was funded by Takeda. The investigators reported additional relationships with AbbVie, Janssen, Amgen, and others. Dr. Longman reported no conflicts of interest.
SOURCE: Loftus Jr EV et al. AP&T. 2020 Sep 2. doi: 10.1111/apt.16060.
Help your patients better understand their IBD treatment options by sharing AGA’s patient education, “Living with IBD,” in the AGA GI Patient Center at http://ow.ly/szHz30rdKyx.
FROM ALIMENTARY PHARMACOLOGY & THERAPEUTICS
Real-world safety, efficacy found for fecal transplants
Fecal microbiota transplantation (FMT) appears safe and effective as a treatment for most Clostridioides difficile infections as it is currently being administered, researchers say.
“We actually didn’t see any infections that were definitely transmissible via fecal transplant,” Colleen Kelly, MD, an associate professor of medicine at Brown University, Providence, R.I., said in an interview.
The findings, published online Oct. 1 in the journal Gastroenterology, come from the American Gastroenterological Association (AGA) NIH-funded FMT National Registry and could allay concerns about a treatment that has yet to gain full approval by the Food and Drug Administration, despite successful clinical trials.
C. diff infections are common and increasing in the United States, often can’t be cured with conventional treatments such as antibiotics, and can be deadly.
Transplanting fecal matter from a donor to the patient appears to work by restoring beneficial microorganisms to the patient’s gut. The procedure is also under investigation for a wide range of other ailments, from irritable bowel syndrome to mood disorders.
But much remains unknown. Researchers have counted a thousand bacterial species along with viruses, bacteriophages, archaea, and fungi in the human gut that interact in complex ways, not all of them beneficial.
The FDA has not enforced regulations that would prohibit the procedure, but in March, it warned about infections with enteropathogenic Escherichia coli and Shiga toxin–producing E. coli following fecal transplants.
As a result of these reports, and the theoretical risk of spreading SARS-CoV-2, OpenBiome, the largest stool bank in the United States, has suspended shipments except for emergency orders, and asked clinicians to quarantine any of its products they already have on hand.
In the meantime, long-term effects of the treatment have not been well documented. And clinical trials have excluded patients who might benefit, such as those who have been immunocompromised or have inflammatory bowel disease.
National registry follows patients outside clinical trials
To better understand how patients fare outside these trials, AGA and other organizations developed a national registry, funded by a grant from the National Institute of Allergy and Infectious Diseases.
The current report summarizes results on 259 patients enrolled between Dec. 5, 2017, and Sept. 2, 2019 at 20 sites.
At baseline, 44% of these patients suffered moderate and 36% mild C. diff infections. The duration of the diagnosis ranged from less than 1 week to 9 years, with a median duration of 20 weeks. They ranged from 1 to 15 episodes with a mean of 3.5.
Almost all had received vancomycin, and 62% had at least two courses. About 40% had received metronidazole and 28% had received fidaxomicin.
Almost all participants received stool from an unknown donor, mostly from stool banks, with OpenBiome accounting for 67%. About 85% of the transplants were administered through colonoscopy and 6% by upper endoscopy.
Out of 222 patients who returned for a 1-month follow-up, 90% met the investigators’ definition of cure: resolution of diarrhea without need for further anti–C. diff therapy. About 98% received only one transplant. An intent to treat analysis produced a cure rate of 86%.
Results were good in patients with comorbidities, including 12% who had irritable bowel syndrome, 9% who had ulcerative colitis, and 7% who had Crohn’s disease, Dr. Kelly said. “I hope everybody sees the importance of it. In these patients that are more complicated, who may have underlying comorbidities, who may not have been in the clinical trials, it looks effective in that group, and also incredibly safe.”
She added that the risk of transmitting SARS-CoV-2 is minor. “I think it would be a very, very unlikely way for someone to get a respiratory pathogen.”
Of the 112 participants who were cured at 1 month and returned for follow-up after 6 months, 4 developed recurrent C. diff infection. Eleven patients who were not cured in the first month returned after 6 months. Of these, seven were reported cured at this later follow-up.
Three complications occurred as result of the procedure: one colonoscopic perforation and two episodes of gastrointestinal bleeding.
About 45% of participants reported at least one symptom, with diarrhea not related to C. difficile the most common, followed by abdominal pain, bloating, and constipation.
Eleven patients suffered infections, including two which the investigators thought might be related to the procedure: Bacteroides fragilis in one participant with severe diarrhea, and enteropathogenic E. coli in another with loose stools. Other infections included four urinary tract infections, three cases of pneumonia, one E. coli bacteremia and one tooth infection.
Within a month of the procedure, 27 patients were hospitalized, with 3 of these cases considered possibly related to the procedure.
Findings may not apply to all clinical settings
Vincent B. Young, MD, PhD, a professor of medicine and infectious diseases at the University of Michigan, Ann Arbor, pointed out that the findings might not apply to all clinical settings. The participating clinicians were almost all gastroenterologists working in academic centers.
“Most of them are not Joe Doctor at the doctor’s office,” said Dr. Young, who was not involved with the study. Clinicians in other specialties, such as infectious diseases, might be more inclined to administer fecal transplants through capsules rather than colonoscopies.
And he added that the study does not address effects of the transplant that might develop over years. “Some people talk about how changes in the microbiota lead to increased risk for long-term complications, things like cancer or heart disease. You’re not going to see those in 6 months.”
Also, the study didn’t yield any findings on indications other than C. diff. “In no way, shape, or form does it mean you can use it for autism, depression, heart disease, or [irritable bowel syndrome],” he said.
Still, he said, the study “confirms the fact that fecal cell transplantation is an effective treatment for recurrent C. diff infection when administered as they administered it.”
The National Institute of Allergy and Infectious Diseases funded the registry. Dr. Kelly reported a relationship with Finch Therapeutics. Dr. Young reports financial relationships with Vedanta Biosciences and Bio-K+.
This story was updated on Oct. 4, 2020.
A version of this article originally appeared on Medscape.com.
Fecal microbiota transplantation (FMT) appears safe and effective as a treatment for most Clostridioides difficile infections as it is currently being administered, researchers say.
“We actually didn’t see any infections that were definitely transmissible via fecal transplant,” Colleen Kelly, MD, an associate professor of medicine at Brown University, Providence, R.I., said in an interview.
The findings, published online Oct. 1 in the journal Gastroenterology, come from the American Gastroenterological Association (AGA) NIH-funded FMT National Registry and could allay concerns about a treatment that has yet to gain full approval by the Food and Drug Administration, despite successful clinical trials.
C. diff infections are common and increasing in the United States, often can’t be cured with conventional treatments such as antibiotics, and can be deadly.
Transplanting fecal matter from a donor to the patient appears to work by restoring beneficial microorganisms to the patient’s gut. The procedure is also under investigation for a wide range of other ailments, from irritable bowel syndrome to mood disorders.
But much remains unknown. Researchers have counted a thousand bacterial species along with viruses, bacteriophages, archaea, and fungi in the human gut that interact in complex ways, not all of them beneficial.
The FDA has not enforced regulations that would prohibit the procedure, but in March, it warned about infections with enteropathogenic Escherichia coli and Shiga toxin–producing E. coli following fecal transplants.
As a result of these reports, and the theoretical risk of spreading SARS-CoV-2, OpenBiome, the largest stool bank in the United States, has suspended shipments except for emergency orders, and asked clinicians to quarantine any of its products they already have on hand.
In the meantime, long-term effects of the treatment have not been well documented. And clinical trials have excluded patients who might benefit, such as those who have been immunocompromised or have inflammatory bowel disease.
National registry follows patients outside clinical trials
To better understand how patients fare outside these trials, AGA and other organizations developed a national registry, funded by a grant from the National Institute of Allergy and Infectious Diseases.
The current report summarizes results on 259 patients enrolled between Dec. 5, 2017, and Sept. 2, 2019 at 20 sites.
At baseline, 44% of these patients suffered moderate and 36% mild C. diff infections. The duration of the diagnosis ranged from less than 1 week to 9 years, with a median duration of 20 weeks. They ranged from 1 to 15 episodes with a mean of 3.5.
Almost all had received vancomycin, and 62% had at least two courses. About 40% had received metronidazole and 28% had received fidaxomicin.
Almost all participants received stool from an unknown donor, mostly from stool banks, with OpenBiome accounting for 67%. About 85% of the transplants were administered through colonoscopy and 6% by upper endoscopy.
Out of 222 patients who returned for a 1-month follow-up, 90% met the investigators’ definition of cure: resolution of diarrhea without need for further anti–C. diff therapy. About 98% received only one transplant. An intent to treat analysis produced a cure rate of 86%.
Results were good in patients with comorbidities, including 12% who had irritable bowel syndrome, 9% who had ulcerative colitis, and 7% who had Crohn’s disease, Dr. Kelly said. “I hope everybody sees the importance of it. In these patients that are more complicated, who may have underlying comorbidities, who may not have been in the clinical trials, it looks effective in that group, and also incredibly safe.”
She added that the risk of transmitting SARS-CoV-2 is minor. “I think it would be a very, very unlikely way for someone to get a respiratory pathogen.”
Of the 112 participants who were cured at 1 month and returned for follow-up after 6 months, 4 developed recurrent C. diff infection. Eleven patients who were not cured in the first month returned after 6 months. Of these, seven were reported cured at this later follow-up.
Three complications occurred as result of the procedure: one colonoscopic perforation and two episodes of gastrointestinal bleeding.
About 45% of participants reported at least one symptom, with diarrhea not related to C. difficile the most common, followed by abdominal pain, bloating, and constipation.
Eleven patients suffered infections, including two which the investigators thought might be related to the procedure: Bacteroides fragilis in one participant with severe diarrhea, and enteropathogenic E. coli in another with loose stools. Other infections included four urinary tract infections, three cases of pneumonia, one E. coli bacteremia and one tooth infection.
Within a month of the procedure, 27 patients were hospitalized, with 3 of these cases considered possibly related to the procedure.
Findings may not apply to all clinical settings
Vincent B. Young, MD, PhD, a professor of medicine and infectious diseases at the University of Michigan, Ann Arbor, pointed out that the findings might not apply to all clinical settings. The participating clinicians were almost all gastroenterologists working in academic centers.
“Most of them are not Joe Doctor at the doctor’s office,” said Dr. Young, who was not involved with the study. Clinicians in other specialties, such as infectious diseases, might be more inclined to administer fecal transplants through capsules rather than colonoscopies.
And he added that the study does not address effects of the transplant that might develop over years. “Some people talk about how changes in the microbiota lead to increased risk for long-term complications, things like cancer or heart disease. You’re not going to see those in 6 months.”
Also, the study didn’t yield any findings on indications other than C. diff. “In no way, shape, or form does it mean you can use it for autism, depression, heart disease, or [irritable bowel syndrome],” he said.
Still, he said, the study “confirms the fact that fecal cell transplantation is an effective treatment for recurrent C. diff infection when administered as they administered it.”
The National Institute of Allergy and Infectious Diseases funded the registry. Dr. Kelly reported a relationship with Finch Therapeutics. Dr. Young reports financial relationships with Vedanta Biosciences and Bio-K+.
This story was updated on Oct. 4, 2020.
A version of this article originally appeared on Medscape.com.
Fecal microbiota transplantation (FMT) appears safe and effective as a treatment for most Clostridioides difficile infections as it is currently being administered, researchers say.
“We actually didn’t see any infections that were definitely transmissible via fecal transplant,” Colleen Kelly, MD, an associate professor of medicine at Brown University, Providence, R.I., said in an interview.
The findings, published online Oct. 1 in the journal Gastroenterology, come from the American Gastroenterological Association (AGA) NIH-funded FMT National Registry and could allay concerns about a treatment that has yet to gain full approval by the Food and Drug Administration, despite successful clinical trials.
C. diff infections are common and increasing in the United States, often can’t be cured with conventional treatments such as antibiotics, and can be deadly.
Transplanting fecal matter from a donor to the patient appears to work by restoring beneficial microorganisms to the patient’s gut. The procedure is also under investigation for a wide range of other ailments, from irritable bowel syndrome to mood disorders.
But much remains unknown. Researchers have counted a thousand bacterial species along with viruses, bacteriophages, archaea, and fungi in the human gut that interact in complex ways, not all of them beneficial.
The FDA has not enforced regulations that would prohibit the procedure, but in March, it warned about infections with enteropathogenic Escherichia coli and Shiga toxin–producing E. coli following fecal transplants.
As a result of these reports, and the theoretical risk of spreading SARS-CoV-2, OpenBiome, the largest stool bank in the United States, has suspended shipments except for emergency orders, and asked clinicians to quarantine any of its products they already have on hand.
In the meantime, long-term effects of the treatment have not been well documented. And clinical trials have excluded patients who might benefit, such as those who have been immunocompromised or have inflammatory bowel disease.
National registry follows patients outside clinical trials
To better understand how patients fare outside these trials, AGA and other organizations developed a national registry, funded by a grant from the National Institute of Allergy and Infectious Diseases.
The current report summarizes results on 259 patients enrolled between Dec. 5, 2017, and Sept. 2, 2019 at 20 sites.
At baseline, 44% of these patients suffered moderate and 36% mild C. diff infections. The duration of the diagnosis ranged from less than 1 week to 9 years, with a median duration of 20 weeks. They ranged from 1 to 15 episodes with a mean of 3.5.
Almost all had received vancomycin, and 62% had at least two courses. About 40% had received metronidazole and 28% had received fidaxomicin.
Almost all participants received stool from an unknown donor, mostly from stool banks, with OpenBiome accounting for 67%. About 85% of the transplants were administered through colonoscopy and 6% by upper endoscopy.
Out of 222 patients who returned for a 1-month follow-up, 90% met the investigators’ definition of cure: resolution of diarrhea without need for further anti–C. diff therapy. About 98% received only one transplant. An intent to treat analysis produced a cure rate of 86%.
Results were good in patients with comorbidities, including 12% who had irritable bowel syndrome, 9% who had ulcerative colitis, and 7% who had Crohn’s disease, Dr. Kelly said. “I hope everybody sees the importance of it. In these patients that are more complicated, who may have underlying comorbidities, who may not have been in the clinical trials, it looks effective in that group, and also incredibly safe.”
She added that the risk of transmitting SARS-CoV-2 is minor. “I think it would be a very, very unlikely way for someone to get a respiratory pathogen.”
Of the 112 participants who were cured at 1 month and returned for follow-up after 6 months, 4 developed recurrent C. diff infection. Eleven patients who were not cured in the first month returned after 6 months. Of these, seven were reported cured at this later follow-up.
Three complications occurred as result of the procedure: one colonoscopic perforation and two episodes of gastrointestinal bleeding.
About 45% of participants reported at least one symptom, with diarrhea not related to C. difficile the most common, followed by abdominal pain, bloating, and constipation.
Eleven patients suffered infections, including two which the investigators thought might be related to the procedure: Bacteroides fragilis in one participant with severe diarrhea, and enteropathogenic E. coli in another with loose stools. Other infections included four urinary tract infections, three cases of pneumonia, one E. coli bacteremia and one tooth infection.
Within a month of the procedure, 27 patients were hospitalized, with 3 of these cases considered possibly related to the procedure.
Findings may not apply to all clinical settings
Vincent B. Young, MD, PhD, a professor of medicine and infectious diseases at the University of Michigan, Ann Arbor, pointed out that the findings might not apply to all clinical settings. The participating clinicians were almost all gastroenterologists working in academic centers.
“Most of them are not Joe Doctor at the doctor’s office,” said Dr. Young, who was not involved with the study. Clinicians in other specialties, such as infectious diseases, might be more inclined to administer fecal transplants through capsules rather than colonoscopies.
And he added that the study does not address effects of the transplant that might develop over years. “Some people talk about how changes in the microbiota lead to increased risk for long-term complications, things like cancer or heart disease. You’re not going to see those in 6 months.”
Also, the study didn’t yield any findings on indications other than C. diff. “In no way, shape, or form does it mean you can use it for autism, depression, heart disease, or [irritable bowel syndrome],” he said.
Still, he said, the study “confirms the fact that fecal cell transplantation is an effective treatment for recurrent C. diff infection when administered as they administered it.”
The National Institute of Allergy and Infectious Diseases funded the registry. Dr. Kelly reported a relationship with Finch Therapeutics. Dr. Young reports financial relationships with Vedanta Biosciences and Bio-K+.
This story was updated on Oct. 4, 2020.
A version of this article originally appeared on Medscape.com.
Study results suggest ustekinumab may trigger acute CV events early in treatment
in susceptible patients, according to a large French case-time-control analysis.
Investigators led by Florence Poizeau, MD, of the department of dermatology at Rennes (France) University Hospital, found high-risk patients had more than four times the risk of an acute SCE in the 6 months after starting treatment. Although ustekinumab (Stelara) effectively treats moderate to severe psoriasis, psoriatic arthritis (PsA), and Crohn’s disease (indications approved by the Food and Drug Administration), the early months after ustekinumab initiation may be associated with atherosclerotic plaque destabilization via the inhibition of helper T cell subtype 17, the group reported in JAMA Dermatology.
The observational study drew on France’s 66 million–registrant health insurance database to identify all patients exposed to ustekinumab between April 1, 2010, and Dec. 31, 2016. Classified by high or low cardiovascular risk level, ustekinumab recipients served as their own controls, being compared during two time windows: the risk period covered the 6 months after initiating treatment and leading up to the SCE, defined as acute coronary syndrome (ACS) or stroke, while a reference period spanned the 6-12 months leading up to the risk period.
In the statistical analysis of 9,290 ustekinumab-exposed patients (mean age 43 years, 52% male), conducted from September 2017 to July 2018, 7,588 (82%) received ustekinumab for psoriasis or PsA, and 724 (8%) for Crohn’s disease. (The remaining indications were for psoriasis or PsA and Crohn’s disease, or were undetermined.)
Of these patients, 98 experienced SCEs (52 with ACS admitted to the ICU and 46 with strokes). In patients deemed at high cardiovascular risk – those with two risk factors or a personal history of atherosclerotic disease – there was a statistically significant association between starting ustekinumab and SCE occurrence, for an odds ratio of 4.17 (95% confidence interval, 1.19-14.59). In contrast, no such association emerged in ustekinumab users at low cardiovascular risk, for an OR of 0.30 (95% CI, 0.03-3.13). The OR for all was 2.41 (95% CI, 0.83-7.01).
Of the 98 patients included in the final case-time-control analysis, 62 were men (63%), the median age was 57 years, and 76 (78%) were at high cardiovascular risk. A total of 89 patients (91%) had psoriasis, four (4%) had Crohn’s disease, and two (2%) had both.
The investigators also did an analysis including these 98 patients plus 13 patients with ACS who were not hospitalized in an ICU, and 68 with unstable angina, for a total of 179. In this group, the ORs for SCE were 1.75 (95% CI, 0.86-3.56) overall, compared with 3.20 (95% CI, 1.29-7.92) among those at high cardiovascular risk and 0.21 (95% CI, 0.02-1.69) among those at low cardiovascular risk.
The Rennes investigators’ decision to focus on early SCEs stemmed in part from a meta-analysis of randomized clinical trials that reported a possible excess of early SCEs in adults exposed to anti–IL-12/23p40 antibodies, which at that time included the now-discontinued experimental antibody briakinumab. Briakinumab trials were aborted and the drug was never brought to market, leaving ustekinumab as the only antibody of this type.
The finding of “an association between ustekinumab initiation and SCE among patients with cardiovascular risk factors suggests the need for caution regarding the prescription of ustekinumab in this population,” Dr. Poizeau and colleagues wrote. The risk “seems to concern patients with psoriasis” rather than Crohn’s disease, which may be related to the older age and greater cardiovascular risk of the former. “A close collaboration between cardiologists and biologic prescribers could be beneficial to evaluate the risk of SCEs for patients who are receiving ustekinumab,” they added, recommending further research into the physiopathological mechanisms of action.
Offering a U.S. clinician’s perspective on the French study, Anthony Fernandez, MD, PhD, director of medical dermatology at the Cleveland Clinic, called the findings “unique and interesting with very robust odds ratios. These posttreatment associations have actually been a big area of research over the past decade but not with such defined time periods.”
No significant increases in risk have been seen with other biologics, Dr. Fernandez added, with the exception of briakinumab. “But still, the current study does not definitively answer the question whether ustekinumab can trigger acute events within 6 months of treatment. There’s smoke, but we haven’t clearly seen a fire.”
As to ustekinumab’s possible pathogenic mechanism of action, Dr. Fernandez pointed to data suggesting that IL-17A can be stabilizing to atherosclerotic plaques. “So there’s a hypothesis that blocking the 17/23 pathway may destabilize plaques and make patients more prone to acute cardiovascular events.”
In other comments from clinicians not involved in the study, Seoyoung Kim, MD, ScD, MSCE, director of the program in rheumatologic, immunologic, and musculoskeletal pharmacoepidemiology (PRIME) at Brigham and Women’s Hospital, Boston, noted that, while the investigators controlled for the trend over time and their design choice included time-fixed covariates such as age, sex, and family history within individuals, the case-crossover study could not control for time-varying confounders within individuals.
“In other words, it’s possible that some of the patients had a lot more disease activity and systemic inflammation and used more NSAIDs, steroids, and other medications potentially related to cardiovascular risk a few months before they started ustekinumab, compared with 6-12 months prior,“ Dr. Kim said in an interview. “I would be curious to know if they would find the same thing or not if they studied a different type of biologic drug.”
She also pointed out that the number of outcomes overall was small, leading to imprecise estimates and wide confidence intervals.
Last year Dr. Kim and associates published a study comparing ustekinumab with tumor necrosis factor inhibitor therapy in younger psoriasis and psoriatic arthritis patients and found no difference between the two groups in major cardiovascular events or atrial fibrillation.
Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, has more reservations about the findings. “The Poizeau study was methodologically flawed, making the results unreliable,” he said in an interview. “There is a breadth of data from clinical trials and observational studies that do not demonstrate an increased risk of major acute cardiovascular events with ustekinumab and the results of the Poizeau study should not impact clinical practice.”
In an interview, Mark G. Lebwohl, MD, professor and chairman of the department of dermatology and chief for clinical therapeutics at the Icahn School of Medicine at Mount Sinai, New York, said that, in his view, the investigators used early reports of a small number cardiovascular events to look at the issue from a faulty perspective, and hence their findings would have no impact on his clinical practice.
“This study looked at the issue incorrectly. It looked at people put on drug who already had two risk factors for heart attack. And psoriasis itself is a third risk factor,” he said. “So lo and behold, big surprise, some of them had cardiovascular events.”
Dr. Lebwohl noted that a wealth of carefully compiled data has found no increase over time in cardiovascular events with this drug in psoriasis patients. The risk of cardiovascular events actually goes down with time because of the drug’s anti-inflammatory effects.
Dr. Fernandez takes a more positive view of the French findings. “The data certainly support the need for further research in this area,” he said in an interview, “and in the meantime this paper will probably make me extra cautious in using ustekinumab in those at significant risk.”
The French study was supported by a grant from the French National Agency for Medicines and Health Products Safety. Dr. Poizeau and seven coauthors had no disclosures. The remaining five reported disclosures that included receiving fees from AbbVie, Admiral, Amgen, Baxalta, Cologne, Dermavant, Eli Lilly, Janssen, Kyowa Kirin, Novartis, Mylan, Sun Pharmaceuticals, and UCB, as well as grants and personal fees from Boehringer Ingelheim, Leo Pharma, and Pfizer outside the submitted work, and personal fees from Pfizer, AbbVie, UCB Pharma, and Lilly during the conduct of the study. Dr. Fernandez reported consulting work for AbbVie and research grants from Novartis. Dr. Kim disclosed research grants from Brigham and Women’s Hospital and from Pfizer, Abbvie, Roche, and Bristol-Myers Squibb for unrelated studies. Dr. Gelfand reported varying financial ties to Bristol-Myers Squibb, Boehringer Ingelheim, GlaxoSmithKline, Janssen Biologics, Regeneron, UCB, Sanofi, Pfizer, Celgene, OrthoDermatolgics, AbbVie, Novartis, and Eli Lilly. He is copatent holder of a treatment for cutaneous T-cell lymphoma. Dr. Lebwohl reported unpaid consulting for most manufacturers of psoriasis drugs, with all fees going directly to Mount Sinai.
Source: Poizeau F et al. JAMA Dermatol. 2020 Sep 9. doi: 10.1001/jamadermatol.2020.2977.
in susceptible patients, according to a large French case-time-control analysis.
Investigators led by Florence Poizeau, MD, of the department of dermatology at Rennes (France) University Hospital, found high-risk patients had more than four times the risk of an acute SCE in the 6 months after starting treatment. Although ustekinumab (Stelara) effectively treats moderate to severe psoriasis, psoriatic arthritis (PsA), and Crohn’s disease (indications approved by the Food and Drug Administration), the early months after ustekinumab initiation may be associated with atherosclerotic plaque destabilization via the inhibition of helper T cell subtype 17, the group reported in JAMA Dermatology.
The observational study drew on France’s 66 million–registrant health insurance database to identify all patients exposed to ustekinumab between April 1, 2010, and Dec. 31, 2016. Classified by high or low cardiovascular risk level, ustekinumab recipients served as their own controls, being compared during two time windows: the risk period covered the 6 months after initiating treatment and leading up to the SCE, defined as acute coronary syndrome (ACS) or stroke, while a reference period spanned the 6-12 months leading up to the risk period.
In the statistical analysis of 9,290 ustekinumab-exposed patients (mean age 43 years, 52% male), conducted from September 2017 to July 2018, 7,588 (82%) received ustekinumab for psoriasis or PsA, and 724 (8%) for Crohn’s disease. (The remaining indications were for psoriasis or PsA and Crohn’s disease, or were undetermined.)
Of these patients, 98 experienced SCEs (52 with ACS admitted to the ICU and 46 with strokes). In patients deemed at high cardiovascular risk – those with two risk factors or a personal history of atherosclerotic disease – there was a statistically significant association between starting ustekinumab and SCE occurrence, for an odds ratio of 4.17 (95% confidence interval, 1.19-14.59). In contrast, no such association emerged in ustekinumab users at low cardiovascular risk, for an OR of 0.30 (95% CI, 0.03-3.13). The OR for all was 2.41 (95% CI, 0.83-7.01).
Of the 98 patients included in the final case-time-control analysis, 62 were men (63%), the median age was 57 years, and 76 (78%) were at high cardiovascular risk. A total of 89 patients (91%) had psoriasis, four (4%) had Crohn’s disease, and two (2%) had both.
The investigators also did an analysis including these 98 patients plus 13 patients with ACS who were not hospitalized in an ICU, and 68 with unstable angina, for a total of 179. In this group, the ORs for SCE were 1.75 (95% CI, 0.86-3.56) overall, compared with 3.20 (95% CI, 1.29-7.92) among those at high cardiovascular risk and 0.21 (95% CI, 0.02-1.69) among those at low cardiovascular risk.
The Rennes investigators’ decision to focus on early SCEs stemmed in part from a meta-analysis of randomized clinical trials that reported a possible excess of early SCEs in adults exposed to anti–IL-12/23p40 antibodies, which at that time included the now-discontinued experimental antibody briakinumab. Briakinumab trials were aborted and the drug was never brought to market, leaving ustekinumab as the only antibody of this type.
The finding of “an association between ustekinumab initiation and SCE among patients with cardiovascular risk factors suggests the need for caution regarding the prescription of ustekinumab in this population,” Dr. Poizeau and colleagues wrote. The risk “seems to concern patients with psoriasis” rather than Crohn’s disease, which may be related to the older age and greater cardiovascular risk of the former. “A close collaboration between cardiologists and biologic prescribers could be beneficial to evaluate the risk of SCEs for patients who are receiving ustekinumab,” they added, recommending further research into the physiopathological mechanisms of action.
Offering a U.S. clinician’s perspective on the French study, Anthony Fernandez, MD, PhD, director of medical dermatology at the Cleveland Clinic, called the findings “unique and interesting with very robust odds ratios. These posttreatment associations have actually been a big area of research over the past decade but not with such defined time periods.”
No significant increases in risk have been seen with other biologics, Dr. Fernandez added, with the exception of briakinumab. “But still, the current study does not definitively answer the question whether ustekinumab can trigger acute events within 6 months of treatment. There’s smoke, but we haven’t clearly seen a fire.”
As to ustekinumab’s possible pathogenic mechanism of action, Dr. Fernandez pointed to data suggesting that IL-17A can be stabilizing to atherosclerotic plaques. “So there’s a hypothesis that blocking the 17/23 pathway may destabilize plaques and make patients more prone to acute cardiovascular events.”
In other comments from clinicians not involved in the study, Seoyoung Kim, MD, ScD, MSCE, director of the program in rheumatologic, immunologic, and musculoskeletal pharmacoepidemiology (PRIME) at Brigham and Women’s Hospital, Boston, noted that, while the investigators controlled for the trend over time and their design choice included time-fixed covariates such as age, sex, and family history within individuals, the case-crossover study could not control for time-varying confounders within individuals.
“In other words, it’s possible that some of the patients had a lot more disease activity and systemic inflammation and used more NSAIDs, steroids, and other medications potentially related to cardiovascular risk a few months before they started ustekinumab, compared with 6-12 months prior,“ Dr. Kim said in an interview. “I would be curious to know if they would find the same thing or not if they studied a different type of biologic drug.”
She also pointed out that the number of outcomes overall was small, leading to imprecise estimates and wide confidence intervals.
Last year Dr. Kim and associates published a study comparing ustekinumab with tumor necrosis factor inhibitor therapy in younger psoriasis and psoriatic arthritis patients and found no difference between the two groups in major cardiovascular events or atrial fibrillation.
Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, has more reservations about the findings. “The Poizeau study was methodologically flawed, making the results unreliable,” he said in an interview. “There is a breadth of data from clinical trials and observational studies that do not demonstrate an increased risk of major acute cardiovascular events with ustekinumab and the results of the Poizeau study should not impact clinical practice.”
In an interview, Mark G. Lebwohl, MD, professor and chairman of the department of dermatology and chief for clinical therapeutics at the Icahn School of Medicine at Mount Sinai, New York, said that, in his view, the investigators used early reports of a small number cardiovascular events to look at the issue from a faulty perspective, and hence their findings would have no impact on his clinical practice.
“This study looked at the issue incorrectly. It looked at people put on drug who already had two risk factors for heart attack. And psoriasis itself is a third risk factor,” he said. “So lo and behold, big surprise, some of them had cardiovascular events.”
Dr. Lebwohl noted that a wealth of carefully compiled data has found no increase over time in cardiovascular events with this drug in psoriasis patients. The risk of cardiovascular events actually goes down with time because of the drug’s anti-inflammatory effects.
Dr. Fernandez takes a more positive view of the French findings. “The data certainly support the need for further research in this area,” he said in an interview, “and in the meantime this paper will probably make me extra cautious in using ustekinumab in those at significant risk.”
The French study was supported by a grant from the French National Agency for Medicines and Health Products Safety. Dr. Poizeau and seven coauthors had no disclosures. The remaining five reported disclosures that included receiving fees from AbbVie, Admiral, Amgen, Baxalta, Cologne, Dermavant, Eli Lilly, Janssen, Kyowa Kirin, Novartis, Mylan, Sun Pharmaceuticals, and UCB, as well as grants and personal fees from Boehringer Ingelheim, Leo Pharma, and Pfizer outside the submitted work, and personal fees from Pfizer, AbbVie, UCB Pharma, and Lilly during the conduct of the study. Dr. Fernandez reported consulting work for AbbVie and research grants from Novartis. Dr. Kim disclosed research grants from Brigham and Women’s Hospital and from Pfizer, Abbvie, Roche, and Bristol-Myers Squibb for unrelated studies. Dr. Gelfand reported varying financial ties to Bristol-Myers Squibb, Boehringer Ingelheim, GlaxoSmithKline, Janssen Biologics, Regeneron, UCB, Sanofi, Pfizer, Celgene, OrthoDermatolgics, AbbVie, Novartis, and Eli Lilly. He is copatent holder of a treatment for cutaneous T-cell lymphoma. Dr. Lebwohl reported unpaid consulting for most manufacturers of psoriasis drugs, with all fees going directly to Mount Sinai.
Source: Poizeau F et al. JAMA Dermatol. 2020 Sep 9. doi: 10.1001/jamadermatol.2020.2977.
in susceptible patients, according to a large French case-time-control analysis.
Investigators led by Florence Poizeau, MD, of the department of dermatology at Rennes (France) University Hospital, found high-risk patients had more than four times the risk of an acute SCE in the 6 months after starting treatment. Although ustekinumab (Stelara) effectively treats moderate to severe psoriasis, psoriatic arthritis (PsA), and Crohn’s disease (indications approved by the Food and Drug Administration), the early months after ustekinumab initiation may be associated with atherosclerotic plaque destabilization via the inhibition of helper T cell subtype 17, the group reported in JAMA Dermatology.
The observational study drew on France’s 66 million–registrant health insurance database to identify all patients exposed to ustekinumab between April 1, 2010, and Dec. 31, 2016. Classified by high or low cardiovascular risk level, ustekinumab recipients served as their own controls, being compared during two time windows: the risk period covered the 6 months after initiating treatment and leading up to the SCE, defined as acute coronary syndrome (ACS) or stroke, while a reference period spanned the 6-12 months leading up to the risk period.
In the statistical analysis of 9,290 ustekinumab-exposed patients (mean age 43 years, 52% male), conducted from September 2017 to July 2018, 7,588 (82%) received ustekinumab for psoriasis or PsA, and 724 (8%) for Crohn’s disease. (The remaining indications were for psoriasis or PsA and Crohn’s disease, or were undetermined.)
Of these patients, 98 experienced SCEs (52 with ACS admitted to the ICU and 46 with strokes). In patients deemed at high cardiovascular risk – those with two risk factors or a personal history of atherosclerotic disease – there was a statistically significant association between starting ustekinumab and SCE occurrence, for an odds ratio of 4.17 (95% confidence interval, 1.19-14.59). In contrast, no such association emerged in ustekinumab users at low cardiovascular risk, for an OR of 0.30 (95% CI, 0.03-3.13). The OR for all was 2.41 (95% CI, 0.83-7.01).
Of the 98 patients included in the final case-time-control analysis, 62 were men (63%), the median age was 57 years, and 76 (78%) were at high cardiovascular risk. A total of 89 patients (91%) had psoriasis, four (4%) had Crohn’s disease, and two (2%) had both.
The investigators also did an analysis including these 98 patients plus 13 patients with ACS who were not hospitalized in an ICU, and 68 with unstable angina, for a total of 179. In this group, the ORs for SCE were 1.75 (95% CI, 0.86-3.56) overall, compared with 3.20 (95% CI, 1.29-7.92) among those at high cardiovascular risk and 0.21 (95% CI, 0.02-1.69) among those at low cardiovascular risk.
The Rennes investigators’ decision to focus on early SCEs stemmed in part from a meta-analysis of randomized clinical trials that reported a possible excess of early SCEs in adults exposed to anti–IL-12/23p40 antibodies, which at that time included the now-discontinued experimental antibody briakinumab. Briakinumab trials were aborted and the drug was never brought to market, leaving ustekinumab as the only antibody of this type.
The finding of “an association between ustekinumab initiation and SCE among patients with cardiovascular risk factors suggests the need for caution regarding the prescription of ustekinumab in this population,” Dr. Poizeau and colleagues wrote. The risk “seems to concern patients with psoriasis” rather than Crohn’s disease, which may be related to the older age and greater cardiovascular risk of the former. “A close collaboration between cardiologists and biologic prescribers could be beneficial to evaluate the risk of SCEs for patients who are receiving ustekinumab,” they added, recommending further research into the physiopathological mechanisms of action.
Offering a U.S. clinician’s perspective on the French study, Anthony Fernandez, MD, PhD, director of medical dermatology at the Cleveland Clinic, called the findings “unique and interesting with very robust odds ratios. These posttreatment associations have actually been a big area of research over the past decade but not with such defined time periods.”
No significant increases in risk have been seen with other biologics, Dr. Fernandez added, with the exception of briakinumab. “But still, the current study does not definitively answer the question whether ustekinumab can trigger acute events within 6 months of treatment. There’s smoke, but we haven’t clearly seen a fire.”
As to ustekinumab’s possible pathogenic mechanism of action, Dr. Fernandez pointed to data suggesting that IL-17A can be stabilizing to atherosclerotic plaques. “So there’s a hypothesis that blocking the 17/23 pathway may destabilize plaques and make patients more prone to acute cardiovascular events.”
In other comments from clinicians not involved in the study, Seoyoung Kim, MD, ScD, MSCE, director of the program in rheumatologic, immunologic, and musculoskeletal pharmacoepidemiology (PRIME) at Brigham and Women’s Hospital, Boston, noted that, while the investigators controlled for the trend over time and their design choice included time-fixed covariates such as age, sex, and family history within individuals, the case-crossover study could not control for time-varying confounders within individuals.
“In other words, it’s possible that some of the patients had a lot more disease activity and systemic inflammation and used more NSAIDs, steroids, and other medications potentially related to cardiovascular risk a few months before they started ustekinumab, compared with 6-12 months prior,“ Dr. Kim said in an interview. “I would be curious to know if they would find the same thing or not if they studied a different type of biologic drug.”
She also pointed out that the number of outcomes overall was small, leading to imprecise estimates and wide confidence intervals.
Last year Dr. Kim and associates published a study comparing ustekinumab with tumor necrosis factor inhibitor therapy in younger psoriasis and psoriatic arthritis patients and found no difference between the two groups in major cardiovascular events or atrial fibrillation.
Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, has more reservations about the findings. “The Poizeau study was methodologically flawed, making the results unreliable,” he said in an interview. “There is a breadth of data from clinical trials and observational studies that do not demonstrate an increased risk of major acute cardiovascular events with ustekinumab and the results of the Poizeau study should not impact clinical practice.”
In an interview, Mark G. Lebwohl, MD, professor and chairman of the department of dermatology and chief for clinical therapeutics at the Icahn School of Medicine at Mount Sinai, New York, said that, in his view, the investigators used early reports of a small number cardiovascular events to look at the issue from a faulty perspective, and hence their findings would have no impact on his clinical practice.
“This study looked at the issue incorrectly. It looked at people put on drug who already had two risk factors for heart attack. And psoriasis itself is a third risk factor,” he said. “So lo and behold, big surprise, some of them had cardiovascular events.”
Dr. Lebwohl noted that a wealth of carefully compiled data has found no increase over time in cardiovascular events with this drug in psoriasis patients. The risk of cardiovascular events actually goes down with time because of the drug’s anti-inflammatory effects.
Dr. Fernandez takes a more positive view of the French findings. “The data certainly support the need for further research in this area,” he said in an interview, “and in the meantime this paper will probably make me extra cautious in using ustekinumab in those at significant risk.”
The French study was supported by a grant from the French National Agency for Medicines and Health Products Safety. Dr. Poizeau and seven coauthors had no disclosures. The remaining five reported disclosures that included receiving fees from AbbVie, Admiral, Amgen, Baxalta, Cologne, Dermavant, Eli Lilly, Janssen, Kyowa Kirin, Novartis, Mylan, Sun Pharmaceuticals, and UCB, as well as grants and personal fees from Boehringer Ingelheim, Leo Pharma, and Pfizer outside the submitted work, and personal fees from Pfizer, AbbVie, UCB Pharma, and Lilly during the conduct of the study. Dr. Fernandez reported consulting work for AbbVie and research grants from Novartis. Dr. Kim disclosed research grants from Brigham and Women’s Hospital and from Pfizer, Abbvie, Roche, and Bristol-Myers Squibb for unrelated studies. Dr. Gelfand reported varying financial ties to Bristol-Myers Squibb, Boehringer Ingelheim, GlaxoSmithKline, Janssen Biologics, Regeneron, UCB, Sanofi, Pfizer, Celgene, OrthoDermatolgics, AbbVie, Novartis, and Eli Lilly. He is copatent holder of a treatment for cutaneous T-cell lymphoma. Dr. Lebwohl reported unpaid consulting for most manufacturers of psoriasis drugs, with all fees going directly to Mount Sinai.
Source: Poizeau F et al. JAMA Dermatol. 2020 Sep 9. doi: 10.1001/jamadermatol.2020.2977.
AGA Clinical Practice Update: Diagnosis and treatment of small intestinal bacterial overgrowth
Unexplained diarrhea may be the most reliable symptom of small intestinal bacterial overgrowth (SIBO) in at-risk patients, according to a new clinical practice update from the American Gastroenterological Association.
“In those predisposed to SIBO due to anatomical, pathological, pharmacological or other changes that promote stasis or recirculation of colonic contents and/or impaired resistance to bacteria, SIBO will lead to diarrhea and can progress to a full-blown malabsorption syndrome” marked by steatorrhea and vitamin deficiencies, wrote Eamonn M.M. Quigley, MD, of Houston Methodist Hospital and Weill Cornell Medical College in Houston together with his fellow experts in Gastroenterology. But malabsorption is uncommon in patients whose SIBO is not caused by structural abnormalities, and gastrointestinal symptoms are “weakly predictive at best” if patients lack clear risk factors for SIBO, the experts cautioned.
The growing availability of breath testing has fueled diagnoses of SIBO, which the lay press often implicates in various disorders even though SIBO has no clear clinical or laboratory definition. Recent progress in techniques to measure bacterial populations and their metabolic products “should provide much needed clarity,” but for now, a SIBO diagnosis simply means that a patient’s presenting symptoms or laboratory findings are attributed to bacterial changes in the small intestine, the experts wrote.
Detecting SIBO also remains challenging. Most patients have normal results on routine laboratory tests, and there is not enough evidence to support testing for inflammatory markers such as fecal calprotectin. Patients with SIBO may have increased folate levels because of bacterial production of folic acid. Vitamin B12 and other nutrient deficiencies also occur but are less common. The preferred diagnostic method is culture of a duodenal aspirate, and recent research supports a cutoff value of greater than 103 CFUs of coliform bacteria per mL. Breath testing is less invasive but “more complex than simply measuring hydrogen,” the experts stressed. Methane-producing microorganisms (methanogens) suppress hydrogen on a breath test (fortunately, standard breath tests measure methane). Furthermore, a positive methane breath test also has been linked to constipation-predominant irritable bowel syndrome (IBS). Recent studies also suggest that lactulose breath testing is more sensitive than glucose for identifying SIBO in patients with IBS.
Antibiotic therapy is the treatment mainstay but remains largely empiric. The goal is to improve SIBO symptoms, not eradicate bacteria from the small intestine. Ideally, the antimicrobial regimen should cover both aerobic and anaerobic bacteria, but clinicians should be mindful of the risks of chronic broad-spectrum antibiotic exposure. In studies, a single 7- to 10-day antibiotic course improved symptoms in approximately 45%-90% of patients with SIBO (rates of breath test response were lower). For patients with IBS and SIBO, rifaximin (which is poorly absorbed) produced encouraging results in two phase 3 studies, but most patients did not receive breath testing, the experts noted. Patients with recurrent SIBO symptoms may need multiple courses of antibiotics with specific regimens rotated to help prevent resistance. “Decisions on management should be individualized and also [should factor in] such risks as diarrhea, Clostridiodes difficile infection, intolerance, and cost,” the experts wrote. “It is not necessary to repeat diagnostic tests for SIBO following antibiotic therapy [if] gastrointestinal symptoms respond.”
Dr. Quigley disclosed financial ties to 4D Pharma, Alimentary Health, Allergan, Biocodex, Biomerica, Ironwood, Salix, Takeda, Vibrant, and Zealand. He also disclosed patents with and equity in Alimentary Health. Both of his coauthors also disclosed ties to various pharmaceutical companies.
SOURCE: Quigley EMM et al. Gastroenterology. 2020 Jun 1. doi: 10.1053/j.gastro.2020.06.090.
Unexplained diarrhea may be the most reliable symptom of small intestinal bacterial overgrowth (SIBO) in at-risk patients, according to a new clinical practice update from the American Gastroenterological Association.
“In those predisposed to SIBO due to anatomical, pathological, pharmacological or other changes that promote stasis or recirculation of colonic contents and/or impaired resistance to bacteria, SIBO will lead to diarrhea and can progress to a full-blown malabsorption syndrome” marked by steatorrhea and vitamin deficiencies, wrote Eamonn M.M. Quigley, MD, of Houston Methodist Hospital and Weill Cornell Medical College in Houston together with his fellow experts in Gastroenterology. But malabsorption is uncommon in patients whose SIBO is not caused by structural abnormalities, and gastrointestinal symptoms are “weakly predictive at best” if patients lack clear risk factors for SIBO, the experts cautioned.
The growing availability of breath testing has fueled diagnoses of SIBO, which the lay press often implicates in various disorders even though SIBO has no clear clinical or laboratory definition. Recent progress in techniques to measure bacterial populations and their metabolic products “should provide much needed clarity,” but for now, a SIBO diagnosis simply means that a patient’s presenting symptoms or laboratory findings are attributed to bacterial changes in the small intestine, the experts wrote.
Detecting SIBO also remains challenging. Most patients have normal results on routine laboratory tests, and there is not enough evidence to support testing for inflammatory markers such as fecal calprotectin. Patients with SIBO may have increased folate levels because of bacterial production of folic acid. Vitamin B12 and other nutrient deficiencies also occur but are less common. The preferred diagnostic method is culture of a duodenal aspirate, and recent research supports a cutoff value of greater than 103 CFUs of coliform bacteria per mL. Breath testing is less invasive but “more complex than simply measuring hydrogen,” the experts stressed. Methane-producing microorganisms (methanogens) suppress hydrogen on a breath test (fortunately, standard breath tests measure methane). Furthermore, a positive methane breath test also has been linked to constipation-predominant irritable bowel syndrome (IBS). Recent studies also suggest that lactulose breath testing is more sensitive than glucose for identifying SIBO in patients with IBS.
Antibiotic therapy is the treatment mainstay but remains largely empiric. The goal is to improve SIBO symptoms, not eradicate bacteria from the small intestine. Ideally, the antimicrobial regimen should cover both aerobic and anaerobic bacteria, but clinicians should be mindful of the risks of chronic broad-spectrum antibiotic exposure. In studies, a single 7- to 10-day antibiotic course improved symptoms in approximately 45%-90% of patients with SIBO (rates of breath test response were lower). For patients with IBS and SIBO, rifaximin (which is poorly absorbed) produced encouraging results in two phase 3 studies, but most patients did not receive breath testing, the experts noted. Patients with recurrent SIBO symptoms may need multiple courses of antibiotics with specific regimens rotated to help prevent resistance. “Decisions on management should be individualized and also [should factor in] such risks as diarrhea, Clostridiodes difficile infection, intolerance, and cost,” the experts wrote. “It is not necessary to repeat diagnostic tests for SIBO following antibiotic therapy [if] gastrointestinal symptoms respond.”
Dr. Quigley disclosed financial ties to 4D Pharma, Alimentary Health, Allergan, Biocodex, Biomerica, Ironwood, Salix, Takeda, Vibrant, and Zealand. He also disclosed patents with and equity in Alimentary Health. Both of his coauthors also disclosed ties to various pharmaceutical companies.
SOURCE: Quigley EMM et al. Gastroenterology. 2020 Jun 1. doi: 10.1053/j.gastro.2020.06.090.
Unexplained diarrhea may be the most reliable symptom of small intestinal bacterial overgrowth (SIBO) in at-risk patients, according to a new clinical practice update from the American Gastroenterological Association.
“In those predisposed to SIBO due to anatomical, pathological, pharmacological or other changes that promote stasis or recirculation of colonic contents and/or impaired resistance to bacteria, SIBO will lead to diarrhea and can progress to a full-blown malabsorption syndrome” marked by steatorrhea and vitamin deficiencies, wrote Eamonn M.M. Quigley, MD, of Houston Methodist Hospital and Weill Cornell Medical College in Houston together with his fellow experts in Gastroenterology. But malabsorption is uncommon in patients whose SIBO is not caused by structural abnormalities, and gastrointestinal symptoms are “weakly predictive at best” if patients lack clear risk factors for SIBO, the experts cautioned.
The growing availability of breath testing has fueled diagnoses of SIBO, which the lay press often implicates in various disorders even though SIBO has no clear clinical or laboratory definition. Recent progress in techniques to measure bacterial populations and their metabolic products “should provide much needed clarity,” but for now, a SIBO diagnosis simply means that a patient’s presenting symptoms or laboratory findings are attributed to bacterial changes in the small intestine, the experts wrote.
Detecting SIBO also remains challenging. Most patients have normal results on routine laboratory tests, and there is not enough evidence to support testing for inflammatory markers such as fecal calprotectin. Patients with SIBO may have increased folate levels because of bacterial production of folic acid. Vitamin B12 and other nutrient deficiencies also occur but are less common. The preferred diagnostic method is culture of a duodenal aspirate, and recent research supports a cutoff value of greater than 103 CFUs of coliform bacteria per mL. Breath testing is less invasive but “more complex than simply measuring hydrogen,” the experts stressed. Methane-producing microorganisms (methanogens) suppress hydrogen on a breath test (fortunately, standard breath tests measure methane). Furthermore, a positive methane breath test also has been linked to constipation-predominant irritable bowel syndrome (IBS). Recent studies also suggest that lactulose breath testing is more sensitive than glucose for identifying SIBO in patients with IBS.
Antibiotic therapy is the treatment mainstay but remains largely empiric. The goal is to improve SIBO symptoms, not eradicate bacteria from the small intestine. Ideally, the antimicrobial regimen should cover both aerobic and anaerobic bacteria, but clinicians should be mindful of the risks of chronic broad-spectrum antibiotic exposure. In studies, a single 7- to 10-day antibiotic course improved symptoms in approximately 45%-90% of patients with SIBO (rates of breath test response were lower). For patients with IBS and SIBO, rifaximin (which is poorly absorbed) produced encouraging results in two phase 3 studies, but most patients did not receive breath testing, the experts noted. Patients with recurrent SIBO symptoms may need multiple courses of antibiotics with specific regimens rotated to help prevent resistance. “Decisions on management should be individualized and also [should factor in] such risks as diarrhea, Clostridiodes difficile infection, intolerance, and cost,” the experts wrote. “It is not necessary to repeat diagnostic tests for SIBO following antibiotic therapy [if] gastrointestinal symptoms respond.”
Dr. Quigley disclosed financial ties to 4D Pharma, Alimentary Health, Allergan, Biocodex, Biomerica, Ironwood, Salix, Takeda, Vibrant, and Zealand. He also disclosed patents with and equity in Alimentary Health. Both of his coauthors also disclosed ties to various pharmaceutical companies.
SOURCE: Quigley EMM et al. Gastroenterology. 2020 Jun 1. doi: 10.1053/j.gastro.2020.06.090.
FROM GASTROENTEROLOGY
A practical approach to utilizing cannabis as adjuvant therapy in inflammatory bowel disease
Case 1
A 30 year-old female with longstanding ulcerative colitis who has a history of medically refractory steroid-dependent disease and was able to achieve remission with vedolizumab for the last 5 years. Most recent objective assessment showed histologic remission. She has been using daily cannabis medicinally for the last year (high CBD:THC [cannabidiol:delta-9-tetracannabidol] concentration). She notes that she has felt better in the last year since introducing cannabis (improved stool frequency/formation, sleep quality). She inquires about discontinuing her biologic therapy in the hope of using cannabis alone to maintain remission.

Figure 1.
Case 2
A 22-year-old male with ileocolonic inflammatory Crohn’s disease escalated to adalimumab requiring an intensification of therapy to weekly dosing to normalize C-reactive protein (CRP). A recent colonoscopy showed endoscopic improvement (colonic normalization and rare aphthae in ileum). He notes clear clinical improvement, but he continues to experience diarrhea and abdominal cramping (no relationship to meals). Declines addition of immunomodulator (nervous about returning to college during the COVID-19 pandemic). He wonders whether cannabis could be effective in controlling his symptoms as he has had improvement in symptoms during his sporadic recreational cannabis exposure.
Discussion
These cases outline the challenges that providers face when managing patients with inflammatory bowel disease (IBD) when a patient would like to either substitute or incorporate cannabis into their treatment plan. Studies have shown a high prevalence of cannabis use among patients with IBD. With the restrictions surrounding the use of cannabis – either medically or recreationally – being liberalized in many states, these conversations are likely to become more frequent in your practice. However, one of the first challenges that providers face surrounding cannabis is that many patients who use cannabis do not disclose use to their health care team for fear of being judged negatively. In addition, many providers do not routinely ask about cannabis use during office visits. This might be directly related to being unprepared to have a knowledge-based discussion on the risks and benefits of cannabis use in IBD, with the same confidence present during discussion of biologic therapies.
For background, Cannabis sativa (cannabis) is composed of hundreds of phytocannabinoids, the two most common are THC and CBD. These cannabinoids act at the endocannabinoid receptors, which are expressed in the central and peripheral nervous systems and immune cells/tissues, and help explain the clinical changes experienced by cannabis users. Both THC and CBD have been studied in varying doses and routes of administration in patients with IBD, making it challenging to translate into real-world recommendations for patients. Some of the most common reported benefits of cannabis use (particularly in an IBD population) are improvement in pain, diarrhea, nausea, and joint pain. Some studies have shown overall improvement in quality of life (Figure 1).
Some common questions that arise surrounding cannabis use in IBD patients include:
1. Is it possible to stop traditional medical therapy and replace it with cannabis therapy?
No studies have directly addressed this exact question. The small studies, both randomized controlled trials and retrospective ones, have studied the effects of cannabis as adjuvant therapy only. None of the data available to date suggest that cannabis has any anti-inflammatory properties with absence of improvement in biomarkers or endoscopic measures of inflammation. In effect, any attempt to discontinue standard therapy with substitution of cannabis-based therapy should be seen as no different than simply discontinuing standard therapy. There exists the argument that – among those with moderate to severe disease – cannabis might suppress the investigation of mild symptoms which may herald a flare of disease, thus lulling the patient into a state of false stability. We do not advocate the substitution of cannabis products in place of standard medical therapy.
2. Is there a role for cannabis as adjuvant therapy in patients with IBD?
Studies to date have included only symptomatic patients with objective evidence of inflammation and assessed clinical, biochemical, or endoscopic endpoints. In Crohn’s disease, two studies showed no improvement in clinical remission rates but showed improvement in clinical response; a third study showed both improvement in clinical remission/response as well as improved quality of life. No study showed a change in disease markers of activity including CRP, fecal calprotectin, or endoscopic scoring. In one study, all patients relapsed shortly after cannabis discontinuation suggesting that, while there was benefit in symptom control, there was no improvement of the underlying chronic inflammation.
In patients with ulcerative colitis, there were two studies. One study showed no improvement and high rates of intolerance in the treatment group, while the other study reported improved disease activity but no objective improvement. The variation in results between disease states and between studies might be because of cannabis formulations. In patients with persistent symptoms despite current medical therapy, there might be a role in those patients for adjuvant therapy for improvement symptom control but not disease control. Optimization of medical therapy would still be indicated.
3. What dose and formulation of cannabis should I recommend to a patient as adjuvant therapy?
This is an excellent question and one that unfortunately we do not have the answer to. As mentioned previously, the studies have looked at varying formulations (THC alone, CBD:THC with varying percentages of THC, CBD alone) and varying routes of administration (sublingual, oral, inhalation). The IBD studies looking at CBD-alone formulations lacked clinical efficacy. In states where cannabis products have been accessible to IBD patients, no data on the product type (THC:CBD), method of administration, or prescriber preferences have been published.
4. What risks should I advise my patients about with cannabis use?
The challenge is that we don’t have large population-based studies in IBD looking at long-term risks of cannabis use. However, in the small RCT studies there were minimal reported side effects and no major adverse events over 8-10 weeks. Larger IBD population-based studies have shown that cannabis users were more likely to discontinue traditional medical therapy, and there is an increased risk for surgery in patients with Crohn’s disease. Larger studies in non-IBD patients have shown risk for addiction to other substances, diminished life achievement, increased motor vehicle accidents, chronic bronchitis, psychiatric disturbances and cannabis dependence, and cannabis hyperemesis syndrome (with an uncanny presentation resembling Crohn’s disease flare with partial small bowel obstruction). Patients should also be advised about legal implications of use (given its continued classification as a federal schedule 1 drug), possible drug interactions, and special considerations in pediatric patients (increased risk of addiction), elderly patients (increased risk of neuropsychological effects), and during pregnancy (with national obstetric society guidelines warning against use because of fetal exposure and increased risk of stillbirth).
5. What are the legal implications for providers? Patients?
As of July 2020, cannabis is available for recreational use in 12 states, for medicinal use in 28 states, and illegal in 11 states. So the answer really depends on what state the patient lives in. As a provider who might certify patients (in some medicinal states) or recommend cannabis to patients, you should consider legal and licensing implications. Again, this might vary state to state, and you should also take into account federal status. Providers acting in compliance with state laws are unlikely to have federal consequences. However, remember that malpractice insurance only covers FDA-approved medical therapies. Patients should be advised to consider the potential (although highly unlikely) to face federal prosecution and implications of use for employment, school, camp, or travel, and driving restrictions.
Take home points
- Inquire about cannabis to start the conversation.
- Know your state’s legalization status surrounding cannabis.
- Patients with IBD report improvement in symptoms and quality of life with adjuvant cannabis use; however, there is no change in disease activity.
- Encourage your patients to continue and optimize their maintenance therapy.
- Educate your patients about the legal considerations and known risks.
In conclusion, the use of cannabis in IBD patients has increased in recent years. It is important to be able to discuss the risks and benefits of use with your IBD patients. Focus on the lack of data showing that cannabis improves disease activity, and has shown benefit only in improving IBD-associated symptoms. In some patients there might be a role for adjuvant cannabis therapy to improve overall symptom control and quality of life.
Dr. Kinnucan is an assistant professor of medicine, division of gastroenterology, Michigan Medicine, University of Michigan, Ann Arbor; Dr. Swaminath is an associate professor of medicine, division of gastroenterology, Lenox Hill Hospital, Northwell Health, New York.
Case 1
A 30 year-old female with longstanding ulcerative colitis who has a history of medically refractory steroid-dependent disease and was able to achieve remission with vedolizumab for the last 5 years. Most recent objective assessment showed histologic remission. She has been using daily cannabis medicinally for the last year (high CBD:THC [cannabidiol:delta-9-tetracannabidol] concentration). She notes that she has felt better in the last year since introducing cannabis (improved stool frequency/formation, sleep quality). She inquires about discontinuing her biologic therapy in the hope of using cannabis alone to maintain remission.

Figure 1.
Case 2
A 22-year-old male with ileocolonic inflammatory Crohn’s disease escalated to adalimumab requiring an intensification of therapy to weekly dosing to normalize C-reactive protein (CRP). A recent colonoscopy showed endoscopic improvement (colonic normalization and rare aphthae in ileum). He notes clear clinical improvement, but he continues to experience diarrhea and abdominal cramping (no relationship to meals). Declines addition of immunomodulator (nervous about returning to college during the COVID-19 pandemic). He wonders whether cannabis could be effective in controlling his symptoms as he has had improvement in symptoms during his sporadic recreational cannabis exposure.
Discussion
These cases outline the challenges that providers face when managing patients with inflammatory bowel disease (IBD) when a patient would like to either substitute or incorporate cannabis into their treatment plan. Studies have shown a high prevalence of cannabis use among patients with IBD. With the restrictions surrounding the use of cannabis – either medically or recreationally – being liberalized in many states, these conversations are likely to become more frequent in your practice. However, one of the first challenges that providers face surrounding cannabis is that many patients who use cannabis do not disclose use to their health care team for fear of being judged negatively. In addition, many providers do not routinely ask about cannabis use during office visits. This might be directly related to being unprepared to have a knowledge-based discussion on the risks and benefits of cannabis use in IBD, with the same confidence present during discussion of biologic therapies.
For background, Cannabis sativa (cannabis) is composed of hundreds of phytocannabinoids, the two most common are THC and CBD. These cannabinoids act at the endocannabinoid receptors, which are expressed in the central and peripheral nervous systems and immune cells/tissues, and help explain the clinical changes experienced by cannabis users. Both THC and CBD have been studied in varying doses and routes of administration in patients with IBD, making it challenging to translate into real-world recommendations for patients. Some of the most common reported benefits of cannabis use (particularly in an IBD population) are improvement in pain, diarrhea, nausea, and joint pain. Some studies have shown overall improvement in quality of life (Figure 1).
Some common questions that arise surrounding cannabis use in IBD patients include:
1. Is it possible to stop traditional medical therapy and replace it with cannabis therapy?
No studies have directly addressed this exact question. The small studies, both randomized controlled trials and retrospective ones, have studied the effects of cannabis as adjuvant therapy only. None of the data available to date suggest that cannabis has any anti-inflammatory properties with absence of improvement in biomarkers or endoscopic measures of inflammation. In effect, any attempt to discontinue standard therapy with substitution of cannabis-based therapy should be seen as no different than simply discontinuing standard therapy. There exists the argument that – among those with moderate to severe disease – cannabis might suppress the investigation of mild symptoms which may herald a flare of disease, thus lulling the patient into a state of false stability. We do not advocate the substitution of cannabis products in place of standard medical therapy.
2. Is there a role for cannabis as adjuvant therapy in patients with IBD?
Studies to date have included only symptomatic patients with objective evidence of inflammation and assessed clinical, biochemical, or endoscopic endpoints. In Crohn’s disease, two studies showed no improvement in clinical remission rates but showed improvement in clinical response; a third study showed both improvement in clinical remission/response as well as improved quality of life. No study showed a change in disease markers of activity including CRP, fecal calprotectin, or endoscopic scoring. In one study, all patients relapsed shortly after cannabis discontinuation suggesting that, while there was benefit in symptom control, there was no improvement of the underlying chronic inflammation.
In patients with ulcerative colitis, there were two studies. One study showed no improvement and high rates of intolerance in the treatment group, while the other study reported improved disease activity but no objective improvement. The variation in results between disease states and between studies might be because of cannabis formulations. In patients with persistent symptoms despite current medical therapy, there might be a role in those patients for adjuvant therapy for improvement symptom control but not disease control. Optimization of medical therapy would still be indicated.
3. What dose and formulation of cannabis should I recommend to a patient as adjuvant therapy?
This is an excellent question and one that unfortunately we do not have the answer to. As mentioned previously, the studies have looked at varying formulations (THC alone, CBD:THC with varying percentages of THC, CBD alone) and varying routes of administration (sublingual, oral, inhalation). The IBD studies looking at CBD-alone formulations lacked clinical efficacy. In states where cannabis products have been accessible to IBD patients, no data on the product type (THC:CBD), method of administration, or prescriber preferences have been published.
4. What risks should I advise my patients about with cannabis use?
The challenge is that we don’t have large population-based studies in IBD looking at long-term risks of cannabis use. However, in the small RCT studies there were minimal reported side effects and no major adverse events over 8-10 weeks. Larger IBD population-based studies have shown that cannabis users were more likely to discontinue traditional medical therapy, and there is an increased risk for surgery in patients with Crohn’s disease. Larger studies in non-IBD patients have shown risk for addiction to other substances, diminished life achievement, increased motor vehicle accidents, chronic bronchitis, psychiatric disturbances and cannabis dependence, and cannabis hyperemesis syndrome (with an uncanny presentation resembling Crohn’s disease flare with partial small bowel obstruction). Patients should also be advised about legal implications of use (given its continued classification as a federal schedule 1 drug), possible drug interactions, and special considerations in pediatric patients (increased risk of addiction), elderly patients (increased risk of neuropsychological effects), and during pregnancy (with national obstetric society guidelines warning against use because of fetal exposure and increased risk of stillbirth).
5. What are the legal implications for providers? Patients?
As of July 2020, cannabis is available for recreational use in 12 states, for medicinal use in 28 states, and illegal in 11 states. So the answer really depends on what state the patient lives in. As a provider who might certify patients (in some medicinal states) or recommend cannabis to patients, you should consider legal and licensing implications. Again, this might vary state to state, and you should also take into account federal status. Providers acting in compliance with state laws are unlikely to have federal consequences. However, remember that malpractice insurance only covers FDA-approved medical therapies. Patients should be advised to consider the potential (although highly unlikely) to face federal prosecution and implications of use for employment, school, camp, or travel, and driving restrictions.
Take home points
- Inquire about cannabis to start the conversation.
- Know your state’s legalization status surrounding cannabis.
- Patients with IBD report improvement in symptoms and quality of life with adjuvant cannabis use; however, there is no change in disease activity.
- Encourage your patients to continue and optimize their maintenance therapy.
- Educate your patients about the legal considerations and known risks.
In conclusion, the use of cannabis in IBD patients has increased in recent years. It is important to be able to discuss the risks and benefits of use with your IBD patients. Focus on the lack of data showing that cannabis improves disease activity, and has shown benefit only in improving IBD-associated symptoms. In some patients there might be a role for adjuvant cannabis therapy to improve overall symptom control and quality of life.
Dr. Kinnucan is an assistant professor of medicine, division of gastroenterology, Michigan Medicine, University of Michigan, Ann Arbor; Dr. Swaminath is an associate professor of medicine, division of gastroenterology, Lenox Hill Hospital, Northwell Health, New York.
Case 1
A 30 year-old female with longstanding ulcerative colitis who has a history of medically refractory steroid-dependent disease and was able to achieve remission with vedolizumab for the last 5 years. Most recent objective assessment showed histologic remission. She has been using daily cannabis medicinally for the last year (high CBD:THC [cannabidiol:delta-9-tetracannabidol] concentration). She notes that she has felt better in the last year since introducing cannabis (improved stool frequency/formation, sleep quality). She inquires about discontinuing her biologic therapy in the hope of using cannabis alone to maintain remission.

Figure 1.
Case 2
A 22-year-old male with ileocolonic inflammatory Crohn’s disease escalated to adalimumab requiring an intensification of therapy to weekly dosing to normalize C-reactive protein (CRP). A recent colonoscopy showed endoscopic improvement (colonic normalization and rare aphthae in ileum). He notes clear clinical improvement, but he continues to experience diarrhea and abdominal cramping (no relationship to meals). Declines addition of immunomodulator (nervous about returning to college during the COVID-19 pandemic). He wonders whether cannabis could be effective in controlling his symptoms as he has had improvement in symptoms during his sporadic recreational cannabis exposure.
Discussion
These cases outline the challenges that providers face when managing patients with inflammatory bowel disease (IBD) when a patient would like to either substitute or incorporate cannabis into their treatment plan. Studies have shown a high prevalence of cannabis use among patients with IBD. With the restrictions surrounding the use of cannabis – either medically or recreationally – being liberalized in many states, these conversations are likely to become more frequent in your practice. However, one of the first challenges that providers face surrounding cannabis is that many patients who use cannabis do not disclose use to their health care team for fear of being judged negatively. In addition, many providers do not routinely ask about cannabis use during office visits. This might be directly related to being unprepared to have a knowledge-based discussion on the risks and benefits of cannabis use in IBD, with the same confidence present during discussion of biologic therapies.
For background, Cannabis sativa (cannabis) is composed of hundreds of phytocannabinoids, the two most common are THC and CBD. These cannabinoids act at the endocannabinoid receptors, which are expressed in the central and peripheral nervous systems and immune cells/tissues, and help explain the clinical changes experienced by cannabis users. Both THC and CBD have been studied in varying doses and routes of administration in patients with IBD, making it challenging to translate into real-world recommendations for patients. Some of the most common reported benefits of cannabis use (particularly in an IBD population) are improvement in pain, diarrhea, nausea, and joint pain. Some studies have shown overall improvement in quality of life (Figure 1).
Some common questions that arise surrounding cannabis use in IBD patients include:
1. Is it possible to stop traditional medical therapy and replace it with cannabis therapy?
No studies have directly addressed this exact question. The small studies, both randomized controlled trials and retrospective ones, have studied the effects of cannabis as adjuvant therapy only. None of the data available to date suggest that cannabis has any anti-inflammatory properties with absence of improvement in biomarkers or endoscopic measures of inflammation. In effect, any attempt to discontinue standard therapy with substitution of cannabis-based therapy should be seen as no different than simply discontinuing standard therapy. There exists the argument that – among those with moderate to severe disease – cannabis might suppress the investigation of mild symptoms which may herald a flare of disease, thus lulling the patient into a state of false stability. We do not advocate the substitution of cannabis products in place of standard medical therapy.
2. Is there a role for cannabis as adjuvant therapy in patients with IBD?
Studies to date have included only symptomatic patients with objective evidence of inflammation and assessed clinical, biochemical, or endoscopic endpoints. In Crohn’s disease, two studies showed no improvement in clinical remission rates but showed improvement in clinical response; a third study showed both improvement in clinical remission/response as well as improved quality of life. No study showed a change in disease markers of activity including CRP, fecal calprotectin, or endoscopic scoring. In one study, all patients relapsed shortly after cannabis discontinuation suggesting that, while there was benefit in symptom control, there was no improvement of the underlying chronic inflammation.
In patients with ulcerative colitis, there were two studies. One study showed no improvement and high rates of intolerance in the treatment group, while the other study reported improved disease activity but no objective improvement. The variation in results between disease states and between studies might be because of cannabis formulations. In patients with persistent symptoms despite current medical therapy, there might be a role in those patients for adjuvant therapy for improvement symptom control but not disease control. Optimization of medical therapy would still be indicated.
3. What dose and formulation of cannabis should I recommend to a patient as adjuvant therapy?
This is an excellent question and one that unfortunately we do not have the answer to. As mentioned previously, the studies have looked at varying formulations (THC alone, CBD:THC with varying percentages of THC, CBD alone) and varying routes of administration (sublingual, oral, inhalation). The IBD studies looking at CBD-alone formulations lacked clinical efficacy. In states where cannabis products have been accessible to IBD patients, no data on the product type (THC:CBD), method of administration, or prescriber preferences have been published.
4. What risks should I advise my patients about with cannabis use?
The challenge is that we don’t have large population-based studies in IBD looking at long-term risks of cannabis use. However, in the small RCT studies there were minimal reported side effects and no major adverse events over 8-10 weeks. Larger IBD population-based studies have shown that cannabis users were more likely to discontinue traditional medical therapy, and there is an increased risk for surgery in patients with Crohn’s disease. Larger studies in non-IBD patients have shown risk for addiction to other substances, diminished life achievement, increased motor vehicle accidents, chronic bronchitis, psychiatric disturbances and cannabis dependence, and cannabis hyperemesis syndrome (with an uncanny presentation resembling Crohn’s disease flare with partial small bowel obstruction). Patients should also be advised about legal implications of use (given its continued classification as a federal schedule 1 drug), possible drug interactions, and special considerations in pediatric patients (increased risk of addiction), elderly patients (increased risk of neuropsychological effects), and during pregnancy (with national obstetric society guidelines warning against use because of fetal exposure and increased risk of stillbirth).
5. What are the legal implications for providers? Patients?
As of July 2020, cannabis is available for recreational use in 12 states, for medicinal use in 28 states, and illegal in 11 states. So the answer really depends on what state the patient lives in. As a provider who might certify patients (in some medicinal states) or recommend cannabis to patients, you should consider legal and licensing implications. Again, this might vary state to state, and you should also take into account federal status. Providers acting in compliance with state laws are unlikely to have federal consequences. However, remember that malpractice insurance only covers FDA-approved medical therapies. Patients should be advised to consider the potential (although highly unlikely) to face federal prosecution and implications of use for employment, school, camp, or travel, and driving restrictions.
Take home points
- Inquire about cannabis to start the conversation.
- Know your state’s legalization status surrounding cannabis.
- Patients with IBD report improvement in symptoms and quality of life with adjuvant cannabis use; however, there is no change in disease activity.
- Encourage your patients to continue and optimize their maintenance therapy.
- Educate your patients about the legal considerations and known risks.
In conclusion, the use of cannabis in IBD patients has increased in recent years. It is important to be able to discuss the risks and benefits of use with your IBD patients. Focus on the lack of data showing that cannabis improves disease activity, and has shown benefit only in improving IBD-associated symptoms. In some patients there might be a role for adjuvant cannabis therapy to improve overall symptom control and quality of life.
Dr. Kinnucan is an assistant professor of medicine, division of gastroenterology, Michigan Medicine, University of Michigan, Ann Arbor; Dr. Swaminath is an associate professor of medicine, division of gastroenterology, Lenox Hill Hospital, Northwell Health, New York.
Humira topped drug-revenue list for 2019
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.
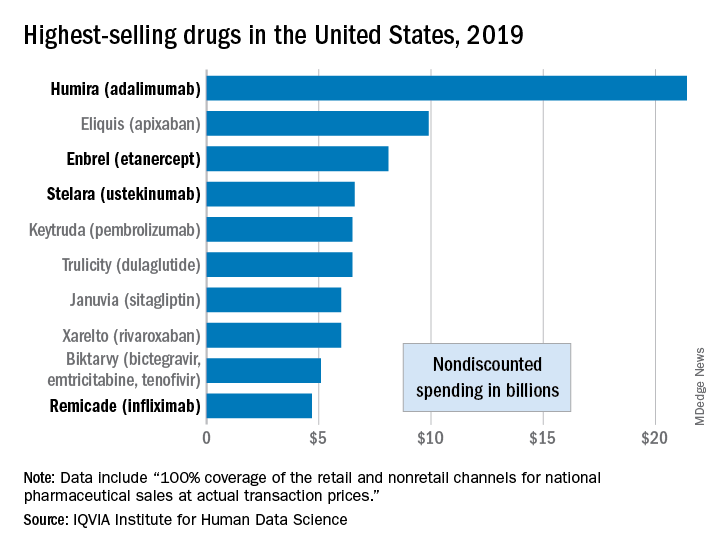
Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.

Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
Humira outsold all other drugs in 2019 in terms of revenue as cytokine inhibitor medications earned their way to three of the first four spots on the pharmaceutical best-seller list, according to a new analysis from the IQVIA Institute for Human Data Science.

Sales of Humira (adalimumab) amounted to $21.4 billion before discounting, Murray Aitken, the institute’s executive director, and associates wrote in their analysis. That’s more than double the total of the anticoagulant Eliquis (apixaban), which brought in $9.9 billion in its last year before generic forms became available.
The next two spots were filled by the tumor necrosis factor inhibitor Enbrel (etanercept) with $8.1 billion in sales and the interleukin 12/23 inhibitor Stelara (ustekinumab) with sales totaling $6.6 billion, followed by the chemotherapy drug Keytruda (pembrolizumab) close behind after racking up $6.5 billion in sales, the researchers reported.
Total nondiscounted spending on all drugs in the U.S. market came to $511 billion in 2019, an increase of 5.7% over the $484 billion spent in 2018, based on data from the July 2020 IQVIA National Sales Perspectives.
These figures are “not adjusted for estimates of off-invoice discounts and rebates,” the authors noted, but they include “prescription and insulin products sold into chain and independent pharmacies, food store pharmacies, mail service pharmacies, long-term care facilities, hospitals, clinics, and other institutional settings.”
Those “discounts and rebates” do exist, however, and they can add up. Drug sales for 2019, “after deducting negotiated rebates, discounts, and other forms of price concessions, such as patient coupons or vouchers that offset out-of-pocket costs,” were $235 billion less than overall nondiscounted spending, the report noted.
Now that we’ve shown you the money, let’s take a quick look at volume. The leading drugs by number of dispensed prescriptions in 2019 were, not surprisingly, quite different. First, with 118 million prescriptions, was atorvastatin, followed by levothyroxine (113 million), lisinopril (96), amlodipine (89), and metoprolol (85), Mr. Aitken and associates reported.
Altogether, over 4.2 billion prescriptions were dispensed last year, with a couple of caveats: 90-day and 30-day fills were both counted as one prescription, and OTC drugs were not included, they pointed out.
AGA releases iron-deficiency anemia guideline
The American Gastroenterological Association (AGA) has released a clinical practice guideline for gastrointestinal evaluation of iron-deficiency anemia.
The seven recommendations aim to improve quality of care and reduce practice variability, according to lead author Cynthia W. Ko, MD, of the University of Washington , Seattle, and four copanelists.
First, the panel recommended that iron deficiency be defined by a serum ferritin level less than 45 ng/mL, instead of 15 ng/mL. Data from 55 studies showed that the higher cutoff value had a sensitivity of 85% and a specificity of 92%, compared with respective values of 59% and 99% for the lower threshold.
“Optimizing the threshold ferritin level with high sensitivity will detect the great majority of patients who are truly iron deficient, minimize delays in diagnostic workup, and minimize the number of patients in whom serious underlying etiologies such as gastrointestinal malignancy might be missed,” the panelists wrote. The guideline was published in Gastroenterology.
For asymptomatic postmenopausal women and men with iron-deficiency anemia, the panel recommended bidirectional endoscopy instead of no endoscopy. A similar recommendation was given for premenopausal women, calling for bidirectional endoscopy instead of iron-replacement therapy alone.
Dr. Ko and colleagues noted that these recommendations differ from those issued by the British Society of Gastroenterology (BSG).
For postmenopausal women and men, the BSG suggests that symptoms and local availability of endoscopy should inform diagnostic workup, with either colonoscopy or CT colonography used for assessment. The BSG recommends against bidirectional endoscopy in premenopausal women, unless they are older than 50 years, have a family history of colorectal cancer, or show symptoms of gastrointestinal disease.
“In contrast, the AGA recommends bidirectional endoscopy as the mainstay for gastrointestinal evaluation, particularly in men and in postmenopausal women where no other unequivocal source of iron deficiency has been identified,” the panelists wrote.
When bidirectional endoscopy does not reveal an etiology, the panelists recommended noninvasive testing for Helicobacter pylori.
“An association between H. pylori infection and iron deficiency has been demonstrated in observational studies,” noted Dr. Ko and colleagues.
They also cited three randomized controlled trials in which treating patients with iron-deficiency anemia for H. pylori infection in combination with iron replacement therapy led to a 23.2-ng/mL mean improvement in serum ferritin, compared with patients who received iron-replacement therapy alone.
According to the guideline, if asymptomatic patients with negative bidirectional endoscopy are also negative for H. pylori, then they should receive trial iron supplementation, instead of undergoing video capsule endoscopy.
“[T]he evidence required to evaluate the benefits of video capsule endoscopy in iron-deficiency anemia is not currently available,” the panelists noted.
Dr. Ko and colleagues also recommended against routine gastric biopsies to diagnose autoimmune atrophic gastritis, as earlier diagnosis does not appear to affect outcomes or management of iron-deficiency anemia.
Finally, the panelists recommended that asymptomatic patients with iron-deficiency anemia and suspected celiac disease undergo serologic testing first, with small bowel biopsy only performed if testing is positive.
The only two strong recommendations in the guideline are for the ferritin cutoff value and the use of bidirectional endoscopy in men and postmenopausal women. The other five recommendations are conditional, three of which are based on very low quality evidence. As such, the guideline includes a discussion of research needs.
Dr. Ko and colleagues called for more studies investigating the prevalence of gastrointestinal lesions and risks of bidirectional endoscopy in premenopausal women, role of fecal occult blood testing, timing of serologic testing for H. pylori and celiac disease in relation to endoscopy, patient subgroups, comparative efficacy of small bowel visualization techniques, and other topics.
The investigators disclosed no conflicts of interest. The guideline was funded by the AGA Institute.
SOURCE: Ko CW et al. Gastroenterology 2020 Aug 15. doi: 10.1053/j.gastro.2020.06.046.
The American Gastroenterological Association (AGA) has released a clinical practice guideline for gastrointestinal evaluation of iron-deficiency anemia.
The seven recommendations aim to improve quality of care and reduce practice variability, according to lead author Cynthia W. Ko, MD, of the University of Washington , Seattle, and four copanelists.
First, the panel recommended that iron deficiency be defined by a serum ferritin level less than 45 ng/mL, instead of 15 ng/mL. Data from 55 studies showed that the higher cutoff value had a sensitivity of 85% and a specificity of 92%, compared with respective values of 59% and 99% for the lower threshold.
“Optimizing the threshold ferritin level with high sensitivity will detect the great majority of patients who are truly iron deficient, minimize delays in diagnostic workup, and minimize the number of patients in whom serious underlying etiologies such as gastrointestinal malignancy might be missed,” the panelists wrote. The guideline was published in Gastroenterology.
For asymptomatic postmenopausal women and men with iron-deficiency anemia, the panel recommended bidirectional endoscopy instead of no endoscopy. A similar recommendation was given for premenopausal women, calling for bidirectional endoscopy instead of iron-replacement therapy alone.
Dr. Ko and colleagues noted that these recommendations differ from those issued by the British Society of Gastroenterology (BSG).
For postmenopausal women and men, the BSG suggests that symptoms and local availability of endoscopy should inform diagnostic workup, with either colonoscopy or CT colonography used for assessment. The BSG recommends against bidirectional endoscopy in premenopausal women, unless they are older than 50 years, have a family history of colorectal cancer, or show symptoms of gastrointestinal disease.
“In contrast, the AGA recommends bidirectional endoscopy as the mainstay for gastrointestinal evaluation, particularly in men and in postmenopausal women where no other unequivocal source of iron deficiency has been identified,” the panelists wrote.
When bidirectional endoscopy does not reveal an etiology, the panelists recommended noninvasive testing for Helicobacter pylori.
“An association between H. pylori infection and iron deficiency has been demonstrated in observational studies,” noted Dr. Ko and colleagues.
They also cited three randomized controlled trials in which treating patients with iron-deficiency anemia for H. pylori infection in combination with iron replacement therapy led to a 23.2-ng/mL mean improvement in serum ferritin, compared with patients who received iron-replacement therapy alone.
According to the guideline, if asymptomatic patients with negative bidirectional endoscopy are also negative for H. pylori, then they should receive trial iron supplementation, instead of undergoing video capsule endoscopy.
“[T]he evidence required to evaluate the benefits of video capsule endoscopy in iron-deficiency anemia is not currently available,” the panelists noted.
Dr. Ko and colleagues also recommended against routine gastric biopsies to diagnose autoimmune atrophic gastritis, as earlier diagnosis does not appear to affect outcomes or management of iron-deficiency anemia.
Finally, the panelists recommended that asymptomatic patients with iron-deficiency anemia and suspected celiac disease undergo serologic testing first, with small bowel biopsy only performed if testing is positive.
The only two strong recommendations in the guideline are for the ferritin cutoff value and the use of bidirectional endoscopy in men and postmenopausal women. The other five recommendations are conditional, three of which are based on very low quality evidence. As such, the guideline includes a discussion of research needs.
Dr. Ko and colleagues called for more studies investigating the prevalence of gastrointestinal lesions and risks of bidirectional endoscopy in premenopausal women, role of fecal occult blood testing, timing of serologic testing for H. pylori and celiac disease in relation to endoscopy, patient subgroups, comparative efficacy of small bowel visualization techniques, and other topics.
The investigators disclosed no conflicts of interest. The guideline was funded by the AGA Institute.
SOURCE: Ko CW et al. Gastroenterology 2020 Aug 15. doi: 10.1053/j.gastro.2020.06.046.
The American Gastroenterological Association (AGA) has released a clinical practice guideline for gastrointestinal evaluation of iron-deficiency anemia.
The seven recommendations aim to improve quality of care and reduce practice variability, according to lead author Cynthia W. Ko, MD, of the University of Washington , Seattle, and four copanelists.
First, the panel recommended that iron deficiency be defined by a serum ferritin level less than 45 ng/mL, instead of 15 ng/mL. Data from 55 studies showed that the higher cutoff value had a sensitivity of 85% and a specificity of 92%, compared with respective values of 59% and 99% for the lower threshold.
“Optimizing the threshold ferritin level with high sensitivity will detect the great majority of patients who are truly iron deficient, minimize delays in diagnostic workup, and minimize the number of patients in whom serious underlying etiologies such as gastrointestinal malignancy might be missed,” the panelists wrote. The guideline was published in Gastroenterology.
For asymptomatic postmenopausal women and men with iron-deficiency anemia, the panel recommended bidirectional endoscopy instead of no endoscopy. A similar recommendation was given for premenopausal women, calling for bidirectional endoscopy instead of iron-replacement therapy alone.
Dr. Ko and colleagues noted that these recommendations differ from those issued by the British Society of Gastroenterology (BSG).
For postmenopausal women and men, the BSG suggests that symptoms and local availability of endoscopy should inform diagnostic workup, with either colonoscopy or CT colonography used for assessment. The BSG recommends against bidirectional endoscopy in premenopausal women, unless they are older than 50 years, have a family history of colorectal cancer, or show symptoms of gastrointestinal disease.
“In contrast, the AGA recommends bidirectional endoscopy as the mainstay for gastrointestinal evaluation, particularly in men and in postmenopausal women where no other unequivocal source of iron deficiency has been identified,” the panelists wrote.
When bidirectional endoscopy does not reveal an etiology, the panelists recommended noninvasive testing for Helicobacter pylori.
“An association between H. pylori infection and iron deficiency has been demonstrated in observational studies,” noted Dr. Ko and colleagues.
They also cited three randomized controlled trials in which treating patients with iron-deficiency anemia for H. pylori infection in combination with iron replacement therapy led to a 23.2-ng/mL mean improvement in serum ferritin, compared with patients who received iron-replacement therapy alone.
According to the guideline, if asymptomatic patients with negative bidirectional endoscopy are also negative for H. pylori, then they should receive trial iron supplementation, instead of undergoing video capsule endoscopy.
“[T]he evidence required to evaluate the benefits of video capsule endoscopy in iron-deficiency anemia is not currently available,” the panelists noted.
Dr. Ko and colleagues also recommended against routine gastric biopsies to diagnose autoimmune atrophic gastritis, as earlier diagnosis does not appear to affect outcomes or management of iron-deficiency anemia.
Finally, the panelists recommended that asymptomatic patients with iron-deficiency anemia and suspected celiac disease undergo serologic testing first, with small bowel biopsy only performed if testing is positive.
The only two strong recommendations in the guideline are for the ferritin cutoff value and the use of bidirectional endoscopy in men and postmenopausal women. The other five recommendations are conditional, three of which are based on very low quality evidence. As such, the guideline includes a discussion of research needs.
Dr. Ko and colleagues called for more studies investigating the prevalence of gastrointestinal lesions and risks of bidirectional endoscopy in premenopausal women, role of fecal occult blood testing, timing of serologic testing for H. pylori and celiac disease in relation to endoscopy, patient subgroups, comparative efficacy of small bowel visualization techniques, and other topics.
The investigators disclosed no conflicts of interest. The guideline was funded by the AGA Institute.
SOURCE: Ko CW et al. Gastroenterology 2020 Aug 15. doi: 10.1053/j.gastro.2020.06.046.
FROM GASTROENTEROLOGY
FDA clamps down on compliance for gluten-free products
To retain the label of “gluten free,” manufacturers of foods that are fermented and hydrolyzed, or that contain fermented or hydrolyzed ingredients, must make and keep detailed records of the manufacturing and production process, according to a final rule issued by the Food and Drug Administration.
In an announcement released on Aug. 12, the FDA stated that manufacturers must confirm that food products such as soy sauce, yogurt, sauerkraut, pickles, cheese, and green olives, as well as distilled foods such as vinegar, meet the definition of gluten free before the fermentation or hydrolysis process. In addition, the rule states that “the manufacturer has adequately evaluated the potential for cross-contact with gluten during the manufacturing process; and if necessary, measures are in place to prevent the introduction of gluten into the food during the manufacturing process,” according to the FDA.
Gluten breaks down during fermentation and hydrolysis, and the gluten-free status of products manufactured in this way can’t be confirmed after the process using currently available methods, according to the FDA.
The new rule is designed to ensure that products labeled as gluten-free meet the definition of gluten free, which remains unchanged from the FDA guidance in 2013.
“The FDA continues to work to protect people with celiac disease, which impacts at least 3 million Americans,” FDA Commissioner Stephen M. Hahn, MD, said in a statement.
“The agency has taken a number of steps on this front by first establishing a standardized definition of gluten free, and now by continuing to work to ensure manufacturers are keeping the products that are labeled with this claim gluten free,” he emphasized.
The final rule states that manufacturers will not need to keep such records if and when other analytical methods are developed, but in the meantime products that do not meet the definition will be deemed misbranded, according to the FDA.
AGA offers guidance on a gluten free diet for patients with celiac disease in the AGA GI Patient Center at http://ow.ly/Wu8F30r4phT.
To retain the label of “gluten free,” manufacturers of foods that are fermented and hydrolyzed, or that contain fermented or hydrolyzed ingredients, must make and keep detailed records of the manufacturing and production process, according to a final rule issued by the Food and Drug Administration.
In an announcement released on Aug. 12, the FDA stated that manufacturers must confirm that food products such as soy sauce, yogurt, sauerkraut, pickles, cheese, and green olives, as well as distilled foods such as vinegar, meet the definition of gluten free before the fermentation or hydrolysis process. In addition, the rule states that “the manufacturer has adequately evaluated the potential for cross-contact with gluten during the manufacturing process; and if necessary, measures are in place to prevent the introduction of gluten into the food during the manufacturing process,” according to the FDA.
Gluten breaks down during fermentation and hydrolysis, and the gluten-free status of products manufactured in this way can’t be confirmed after the process using currently available methods, according to the FDA.
The new rule is designed to ensure that products labeled as gluten-free meet the definition of gluten free, which remains unchanged from the FDA guidance in 2013.
“The FDA continues to work to protect people with celiac disease, which impacts at least 3 million Americans,” FDA Commissioner Stephen M. Hahn, MD, said in a statement.
“The agency has taken a number of steps on this front by first establishing a standardized definition of gluten free, and now by continuing to work to ensure manufacturers are keeping the products that are labeled with this claim gluten free,” he emphasized.
The final rule states that manufacturers will not need to keep such records if and when other analytical methods are developed, but in the meantime products that do not meet the definition will be deemed misbranded, according to the FDA.
AGA offers guidance on a gluten free diet for patients with celiac disease in the AGA GI Patient Center at http://ow.ly/Wu8F30r4phT.
To retain the label of “gluten free,” manufacturers of foods that are fermented and hydrolyzed, or that contain fermented or hydrolyzed ingredients, must make and keep detailed records of the manufacturing and production process, according to a final rule issued by the Food and Drug Administration.
In an announcement released on Aug. 12, the FDA stated that manufacturers must confirm that food products such as soy sauce, yogurt, sauerkraut, pickles, cheese, and green olives, as well as distilled foods such as vinegar, meet the definition of gluten free before the fermentation or hydrolysis process. In addition, the rule states that “the manufacturer has adequately evaluated the potential for cross-contact with gluten during the manufacturing process; and if necessary, measures are in place to prevent the introduction of gluten into the food during the manufacturing process,” according to the FDA.
Gluten breaks down during fermentation and hydrolysis, and the gluten-free status of products manufactured in this way can’t be confirmed after the process using currently available methods, according to the FDA.
The new rule is designed to ensure that products labeled as gluten-free meet the definition of gluten free, which remains unchanged from the FDA guidance in 2013.
“The FDA continues to work to protect people with celiac disease, which impacts at least 3 million Americans,” FDA Commissioner Stephen M. Hahn, MD, said in a statement.
“The agency has taken a number of steps on this front by first establishing a standardized definition of gluten free, and now by continuing to work to ensure manufacturers are keeping the products that are labeled with this claim gluten free,” he emphasized.
The final rule states that manufacturers will not need to keep such records if and when other analytical methods are developed, but in the meantime products that do not meet the definition will be deemed misbranded, according to the FDA.
AGA offers guidance on a gluten free diet for patients with celiac disease in the AGA GI Patient Center at http://ow.ly/Wu8F30r4phT.
Complete endoscopic healing tied to better Crohn’s disease outcomes
Patients with Crohn’s disease who experienced complete endoscopic healing with biologic therapy had significantly lower subsequent rates of treatment failure, intestinal resection, and hospitalization, compared with patients who experienced only partial mucosal healing, according to the findings of a two-center retrospective study.
Over a median of 4.8 years of follow-up (interquartile range, 2.1-7.2 years) rates of treatment failure were 25% in patients with complete mucosal healing at baseline (that is, a Crohn’s Disease Endoscopic Index of Severity [CDEIS] score of 0) and 48% in patients with partial healing (CDEIS score greater than 0 but less than 4). The difference was statistically significant (P = .045). No patient with a baseline CDEIS score of 0 required intestinal resection, compared with 11% of patients with scores greater than 0 but less than 4 (P = .031). Rates of hospitalization because of Crohn’s disease were 3.5% versus 18.5%, respectively (P = .013). Clara Yzet, MD, of Amiens (France) University Hospital, Université de Picardie Jules Verne, reported the findings together with her associates in Clinical Gastroenterology and Hepatology.
Mucosal healing is a key therapeutic target in Crohn’s disease that has been linked to desirable outcomes, such as steroid-free remission and a lower rate of intestinal resection. However, prior observational studies have inconsistently defined mucosal healing, and clinical trials of biologics have used any of at least seven different definitions, the researchers wrote. Recently, in patients with ulcerative colitis, a Scandinavian Journal of Gastroenterology and another in the Journal of Crohn’s and Colitis linked a stricter definition of mucosal healing (an endoscopic Mayo score of 0, or histologic healing) with superior long-term clinical outcomes. In patients with Crohn’s disease, however, there has been no established threshold for mucosal healing based on either the CDEIS or the Simplified Endoscopic Score for Crohn’s disease (SES-CD).
Therefore, Dr. Yzet and her associates identified and reviewed the medical records of 84 consecutive adults with clinically remitted Crohn’s disease who received anti–tumor necrosis factor therapies (infliximab and adalimumab) or vedolizumab at two university hospitals in France between 2008 and 2015. All patients received baseline and follow-up colonoscopies, with results scored on the CDEIS. In all cases, the second CDEIS score was less than 4 (the CDEIS ranges from 0 to 44). The primary outcome was treatment failure, defined as the need for biologic optimization (increasing the dose or shortening the dosing interval of the biologic), corticosteroids, or immunosuppressants; a Harvey-Bradshaw score greater than 4 associated with a change in treatment; or the need for intestinal resection or hospitalization because of Crohn’s disease.
At baseline, 57 patients had CDEIS scores of 0 (complete mucosal healing) and 27 patients had scores greater than 0 but less than 4 (partial mucosal healing). The two groups were otherwise similar except that patients with complete mucosal healing had a shorter median duration of Crohn’s disease (10.3 vs. 15.1 years in the partial healing group; P = .029) and a lower prevalence of Crohn’s disease phenotype B2 (stricturing) according to the Vienna classification (1.8% vs. 14.8%; P = .035). In the multivariate analysis, CDEIS score was the only factor associated with treatment failure (hazard ratio, 2.61; 95% confidence interval, 1.16-5.88; P = .02).
“Our findings suggest that we should be more ambitious in clinical practice to change patients’ life and disease course by achieving complete endoscopic healing. However, this strategy could be limited by the ability of current drugs to achieve complete mucosal healing,” the researchers wrote. “Obtaining a complete mucosal healing would require today a significant need for optimization or change of biologics.”
No external funding sources were reported. Two coinvestigators disclosed ties to AbbVie, Amgen, Biogaran, Biogen, Ferring, Janssen, MSD, Pfizer, Takeda, and several other pharmaceutical companies. The remaining investigators reported having no relevant conflicts of interest.
SOURCE: Yzet C et al. Clin Gastroenterol Hepatol. 2019 Nov 16. doi: 10.1016/j.cgh.2019.11.025.
Patients with Crohn’s disease who experienced complete endoscopic healing with biologic therapy had significantly lower subsequent rates of treatment failure, intestinal resection, and hospitalization, compared with patients who experienced only partial mucosal healing, according to the findings of a two-center retrospective study.
Over a median of 4.8 years of follow-up (interquartile range, 2.1-7.2 years) rates of treatment failure were 25% in patients with complete mucosal healing at baseline (that is, a Crohn’s Disease Endoscopic Index of Severity [CDEIS] score of 0) and 48% in patients with partial healing (CDEIS score greater than 0 but less than 4). The difference was statistically significant (P = .045). No patient with a baseline CDEIS score of 0 required intestinal resection, compared with 11% of patients with scores greater than 0 but less than 4 (P = .031). Rates of hospitalization because of Crohn’s disease were 3.5% versus 18.5%, respectively (P = .013). Clara Yzet, MD, of Amiens (France) University Hospital, Université de Picardie Jules Verne, reported the findings together with her associates in Clinical Gastroenterology and Hepatology.
Mucosal healing is a key therapeutic target in Crohn’s disease that has been linked to desirable outcomes, such as steroid-free remission and a lower rate of intestinal resection. However, prior observational studies have inconsistently defined mucosal healing, and clinical trials of biologics have used any of at least seven different definitions, the researchers wrote. Recently, in patients with ulcerative colitis, a Scandinavian Journal of Gastroenterology and another in the Journal of Crohn’s and Colitis linked a stricter definition of mucosal healing (an endoscopic Mayo score of 0, or histologic healing) with superior long-term clinical outcomes. In patients with Crohn’s disease, however, there has been no established threshold for mucosal healing based on either the CDEIS or the Simplified Endoscopic Score for Crohn’s disease (SES-CD).
Therefore, Dr. Yzet and her associates identified and reviewed the medical records of 84 consecutive adults with clinically remitted Crohn’s disease who received anti–tumor necrosis factor therapies (infliximab and adalimumab) or vedolizumab at two university hospitals in France between 2008 and 2015. All patients received baseline and follow-up colonoscopies, with results scored on the CDEIS. In all cases, the second CDEIS score was less than 4 (the CDEIS ranges from 0 to 44). The primary outcome was treatment failure, defined as the need for biologic optimization (increasing the dose or shortening the dosing interval of the biologic), corticosteroids, or immunosuppressants; a Harvey-Bradshaw score greater than 4 associated with a change in treatment; or the need for intestinal resection or hospitalization because of Crohn’s disease.
At baseline, 57 patients had CDEIS scores of 0 (complete mucosal healing) and 27 patients had scores greater than 0 but less than 4 (partial mucosal healing). The two groups were otherwise similar except that patients with complete mucosal healing had a shorter median duration of Crohn’s disease (10.3 vs. 15.1 years in the partial healing group; P = .029) and a lower prevalence of Crohn’s disease phenotype B2 (stricturing) according to the Vienna classification (1.8% vs. 14.8%; P = .035). In the multivariate analysis, CDEIS score was the only factor associated with treatment failure (hazard ratio, 2.61; 95% confidence interval, 1.16-5.88; P = .02).
“Our findings suggest that we should be more ambitious in clinical practice to change patients’ life and disease course by achieving complete endoscopic healing. However, this strategy could be limited by the ability of current drugs to achieve complete mucosal healing,” the researchers wrote. “Obtaining a complete mucosal healing would require today a significant need for optimization or change of biologics.”
No external funding sources were reported. Two coinvestigators disclosed ties to AbbVie, Amgen, Biogaran, Biogen, Ferring, Janssen, MSD, Pfizer, Takeda, and several other pharmaceutical companies. The remaining investigators reported having no relevant conflicts of interest.
SOURCE: Yzet C et al. Clin Gastroenterol Hepatol. 2019 Nov 16. doi: 10.1016/j.cgh.2019.11.025.
Patients with Crohn’s disease who experienced complete endoscopic healing with biologic therapy had significantly lower subsequent rates of treatment failure, intestinal resection, and hospitalization, compared with patients who experienced only partial mucosal healing, according to the findings of a two-center retrospective study.
Over a median of 4.8 years of follow-up (interquartile range, 2.1-7.2 years) rates of treatment failure were 25% in patients with complete mucosal healing at baseline (that is, a Crohn’s Disease Endoscopic Index of Severity [CDEIS] score of 0) and 48% in patients with partial healing (CDEIS score greater than 0 but less than 4). The difference was statistically significant (P = .045). No patient with a baseline CDEIS score of 0 required intestinal resection, compared with 11% of patients with scores greater than 0 but less than 4 (P = .031). Rates of hospitalization because of Crohn’s disease were 3.5% versus 18.5%, respectively (P = .013). Clara Yzet, MD, of Amiens (France) University Hospital, Université de Picardie Jules Verne, reported the findings together with her associates in Clinical Gastroenterology and Hepatology.
Mucosal healing is a key therapeutic target in Crohn’s disease that has been linked to desirable outcomes, such as steroid-free remission and a lower rate of intestinal resection. However, prior observational studies have inconsistently defined mucosal healing, and clinical trials of biologics have used any of at least seven different definitions, the researchers wrote. Recently, in patients with ulcerative colitis, a Scandinavian Journal of Gastroenterology and another in the Journal of Crohn’s and Colitis linked a stricter definition of mucosal healing (an endoscopic Mayo score of 0, or histologic healing) with superior long-term clinical outcomes. In patients with Crohn’s disease, however, there has been no established threshold for mucosal healing based on either the CDEIS or the Simplified Endoscopic Score for Crohn’s disease (SES-CD).
Therefore, Dr. Yzet and her associates identified and reviewed the medical records of 84 consecutive adults with clinically remitted Crohn’s disease who received anti–tumor necrosis factor therapies (infliximab and adalimumab) or vedolizumab at two university hospitals in France between 2008 and 2015. All patients received baseline and follow-up colonoscopies, with results scored on the CDEIS. In all cases, the second CDEIS score was less than 4 (the CDEIS ranges from 0 to 44). The primary outcome was treatment failure, defined as the need for biologic optimization (increasing the dose or shortening the dosing interval of the biologic), corticosteroids, or immunosuppressants; a Harvey-Bradshaw score greater than 4 associated with a change in treatment; or the need for intestinal resection or hospitalization because of Crohn’s disease.
At baseline, 57 patients had CDEIS scores of 0 (complete mucosal healing) and 27 patients had scores greater than 0 but less than 4 (partial mucosal healing). The two groups were otherwise similar except that patients with complete mucosal healing had a shorter median duration of Crohn’s disease (10.3 vs. 15.1 years in the partial healing group; P = .029) and a lower prevalence of Crohn’s disease phenotype B2 (stricturing) according to the Vienna classification (1.8% vs. 14.8%; P = .035). In the multivariate analysis, CDEIS score was the only factor associated with treatment failure (hazard ratio, 2.61; 95% confidence interval, 1.16-5.88; P = .02).
“Our findings suggest that we should be more ambitious in clinical practice to change patients’ life and disease course by achieving complete endoscopic healing. However, this strategy could be limited by the ability of current drugs to achieve complete mucosal healing,” the researchers wrote. “Obtaining a complete mucosal healing would require today a significant need for optimization or change of biologics.”
No external funding sources were reported. Two coinvestigators disclosed ties to AbbVie, Amgen, Biogaran, Biogen, Ferring, Janssen, MSD, Pfizer, Takeda, and several other pharmaceutical companies. The remaining investigators reported having no relevant conflicts of interest.
SOURCE: Yzet C et al. Clin Gastroenterol Hepatol. 2019 Nov 16. doi: 10.1016/j.cgh.2019.11.025.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Studies eyes risks for poor outcomes in primary sclerosing cholangitis
In individuals with inflammatory bowel disease and primary sclerosing cholangitis, younger age at diagnosis, male sex, and Afro-Caribbean heritage were significant risk factors for liver transplantation and disease-related death, based on a 10-year prospective population-based study.
These factors should be incorporated into the design of clinical trials, models for predicting disease, and studies of prognostic biomarkers for primary sclerosing cholangitis, Palak T. Trivedi, MBBS, MRCP, of the Universty of Birmingham (England) wrote with his associates in Gastroenterology.
The researchers identified newly diagnosed cases from a national health care registry in England between 2006 and 2016 (data on outcomes were collected through mid-2019). In all, 284,560 individuals had a new diagnosis of inflammatory bowel disease, among whom 2,588 also had primary sclerosing cholangitis. The investigators tracked deaths, liver transplantation, colonic resection, cholecystectomy, and diagnoses of colorectal cancer, cholangiosarcoma, and cancers of the pancreas, gallbladder, and liver. They evaluated rates of these outcomes among individuals with both primary sclerosing cholangitis and inflammatory bowel disease (PSC-IBD) and those with IBD only.
After controlling for sex, race, socioeconomic level, comorbidities, and older age, the researchers found that both men and women with PSC-IBD had a significantly greater risk for all-cause mortality, compared with individuals with IBD alone (hazard ratio, 3.20; 95% confidence interval, 3.01-3.40; P less than .001). Strikingly, individuals who were diagnosed with PSC when they were younger than 40 years had a more than sevenfold higher rate of all-cause mortality, compared with individuals with IBD only. In contrast, the incidence rate ratio for individuals diagnosed with PSC when they were older than 60 years was less than 1.5, compared with IBD-only individuals.
Having PSC and ulcerative colitis, being younger when diagnosed with PSC, and being of Afro-Carribean heritage all correlated with higher incidence of liver transplantation or death related to PSC. Individuals with PSC-IBD who were of Afro-Caribbean heritage had an approximately twofold greater risk for liver transplantation or PSC-related death compared with Whites (adjusted HR, 2.05; 95% CI, 1.14-3.70; P = .016). In contrast, women with PSC-IBD were at significantly lower risk for liver transplantation or disease-related death than were men (adjusted HR, 0.74; 95% CI, 0.57-0.97; P = .026).
“The onset of PSC confers heightened risks of all hepatobiliary malignancies, although annual imaging surveillance may associate with a reduced risk of cancer-related death,” the investigators found. Among patients with hepatobiliary cancer, annual imaging was associated with a twofold decrease in risk for cancer-related death (HR, 0.43; 95% CI, 0.23-0.80; P = .037).
Colorectal cancer tended to occur at a younger age among individuals with PSC-IBD, compared with those with IBD alone (median ages at diagnosis, 59 vs. 69 years; P less than .001). Notably, individuals with PSC diagnosed under age 50 years had about a fivefold higher incidence of colorectal cancer than did those with IBD alone, while those diagnosed at older ages had only about a twofold increase. With regard to colectomy, men diagnosed with PSC at younger ages were at the greatest risk, compared with women or individuals diagnosed after age 50 years. Individuals with ulcerative colitis and PSC had a 40% greater risk for colectomy risk than did IBD-only individuals (time-dependent adjusted HR, 1.65; 95% CI, 1.45-1.85; P less than .001).
“Whilst all-cause mortality rates increase with age, younger patients [with PSC] show a disproportionately increased incidence of liver transplantation, PSC-related death, and colorectal cancer,” the researchers concluded. “Consideration of age at diagnosis should therefore be applied in the stratification of patients for future clinical trials, disease prediction models, and prognostic biomarker discovery.”
Dr. Trivedi disclosed support from the National Institute for Health Research Birmingham Biomedical Research Centre, at the University Hospitals Birmingham NHS Foundation Trust and the University of Birmingham. No other disclosures were reported.
SOURCE: Trivedi PJ et al. Gastroenterology. 2020 May 19. doi: 10.1053/j.gastro.2020.05.049.
In individuals with inflammatory bowel disease and primary sclerosing cholangitis, younger age at diagnosis, male sex, and Afro-Caribbean heritage were significant risk factors for liver transplantation and disease-related death, based on a 10-year prospective population-based study.
These factors should be incorporated into the design of clinical trials, models for predicting disease, and studies of prognostic biomarkers for primary sclerosing cholangitis, Palak T. Trivedi, MBBS, MRCP, of the Universty of Birmingham (England) wrote with his associates in Gastroenterology.
The researchers identified newly diagnosed cases from a national health care registry in England between 2006 and 2016 (data on outcomes were collected through mid-2019). In all, 284,560 individuals had a new diagnosis of inflammatory bowel disease, among whom 2,588 also had primary sclerosing cholangitis. The investigators tracked deaths, liver transplantation, colonic resection, cholecystectomy, and diagnoses of colorectal cancer, cholangiosarcoma, and cancers of the pancreas, gallbladder, and liver. They evaluated rates of these outcomes among individuals with both primary sclerosing cholangitis and inflammatory bowel disease (PSC-IBD) and those with IBD only.
After controlling for sex, race, socioeconomic level, comorbidities, and older age, the researchers found that both men and women with PSC-IBD had a significantly greater risk for all-cause mortality, compared with individuals with IBD alone (hazard ratio, 3.20; 95% confidence interval, 3.01-3.40; P less than .001). Strikingly, individuals who were diagnosed with PSC when they were younger than 40 years had a more than sevenfold higher rate of all-cause mortality, compared with individuals with IBD only. In contrast, the incidence rate ratio for individuals diagnosed with PSC when they were older than 60 years was less than 1.5, compared with IBD-only individuals.
Having PSC and ulcerative colitis, being younger when diagnosed with PSC, and being of Afro-Carribean heritage all correlated with higher incidence of liver transplantation or death related to PSC. Individuals with PSC-IBD who were of Afro-Caribbean heritage had an approximately twofold greater risk for liver transplantation or PSC-related death compared with Whites (adjusted HR, 2.05; 95% CI, 1.14-3.70; P = .016). In contrast, women with PSC-IBD were at significantly lower risk for liver transplantation or disease-related death than were men (adjusted HR, 0.74; 95% CI, 0.57-0.97; P = .026).
“The onset of PSC confers heightened risks of all hepatobiliary malignancies, although annual imaging surveillance may associate with a reduced risk of cancer-related death,” the investigators found. Among patients with hepatobiliary cancer, annual imaging was associated with a twofold decrease in risk for cancer-related death (HR, 0.43; 95% CI, 0.23-0.80; P = .037).
Colorectal cancer tended to occur at a younger age among individuals with PSC-IBD, compared with those with IBD alone (median ages at diagnosis, 59 vs. 69 years; P less than .001). Notably, individuals with PSC diagnosed under age 50 years had about a fivefold higher incidence of colorectal cancer than did those with IBD alone, while those diagnosed at older ages had only about a twofold increase. With regard to colectomy, men diagnosed with PSC at younger ages were at the greatest risk, compared with women or individuals diagnosed after age 50 years. Individuals with ulcerative colitis and PSC had a 40% greater risk for colectomy risk than did IBD-only individuals (time-dependent adjusted HR, 1.65; 95% CI, 1.45-1.85; P less than .001).
“Whilst all-cause mortality rates increase with age, younger patients [with PSC] show a disproportionately increased incidence of liver transplantation, PSC-related death, and colorectal cancer,” the researchers concluded. “Consideration of age at diagnosis should therefore be applied in the stratification of patients for future clinical trials, disease prediction models, and prognostic biomarker discovery.”
Dr. Trivedi disclosed support from the National Institute for Health Research Birmingham Biomedical Research Centre, at the University Hospitals Birmingham NHS Foundation Trust and the University of Birmingham. No other disclosures were reported.
SOURCE: Trivedi PJ et al. Gastroenterology. 2020 May 19. doi: 10.1053/j.gastro.2020.05.049.
In individuals with inflammatory bowel disease and primary sclerosing cholangitis, younger age at diagnosis, male sex, and Afro-Caribbean heritage were significant risk factors for liver transplantation and disease-related death, based on a 10-year prospective population-based study.
These factors should be incorporated into the design of clinical trials, models for predicting disease, and studies of prognostic biomarkers for primary sclerosing cholangitis, Palak T. Trivedi, MBBS, MRCP, of the Universty of Birmingham (England) wrote with his associates in Gastroenterology.
The researchers identified newly diagnosed cases from a national health care registry in England between 2006 and 2016 (data on outcomes were collected through mid-2019). In all, 284,560 individuals had a new diagnosis of inflammatory bowel disease, among whom 2,588 also had primary sclerosing cholangitis. The investigators tracked deaths, liver transplantation, colonic resection, cholecystectomy, and diagnoses of colorectal cancer, cholangiosarcoma, and cancers of the pancreas, gallbladder, and liver. They evaluated rates of these outcomes among individuals with both primary sclerosing cholangitis and inflammatory bowel disease (PSC-IBD) and those with IBD only.
After controlling for sex, race, socioeconomic level, comorbidities, and older age, the researchers found that both men and women with PSC-IBD had a significantly greater risk for all-cause mortality, compared with individuals with IBD alone (hazard ratio, 3.20; 95% confidence interval, 3.01-3.40; P less than .001). Strikingly, individuals who were diagnosed with PSC when they were younger than 40 years had a more than sevenfold higher rate of all-cause mortality, compared with individuals with IBD only. In contrast, the incidence rate ratio for individuals diagnosed with PSC when they were older than 60 years was less than 1.5, compared with IBD-only individuals.
Having PSC and ulcerative colitis, being younger when diagnosed with PSC, and being of Afro-Carribean heritage all correlated with higher incidence of liver transplantation or death related to PSC. Individuals with PSC-IBD who were of Afro-Caribbean heritage had an approximately twofold greater risk for liver transplantation or PSC-related death compared with Whites (adjusted HR, 2.05; 95% CI, 1.14-3.70; P = .016). In contrast, women with PSC-IBD were at significantly lower risk for liver transplantation or disease-related death than were men (adjusted HR, 0.74; 95% CI, 0.57-0.97; P = .026).
“The onset of PSC confers heightened risks of all hepatobiliary malignancies, although annual imaging surveillance may associate with a reduced risk of cancer-related death,” the investigators found. Among patients with hepatobiliary cancer, annual imaging was associated with a twofold decrease in risk for cancer-related death (HR, 0.43; 95% CI, 0.23-0.80; P = .037).
Colorectal cancer tended to occur at a younger age among individuals with PSC-IBD, compared with those with IBD alone (median ages at diagnosis, 59 vs. 69 years; P less than .001). Notably, individuals with PSC diagnosed under age 50 years had about a fivefold higher incidence of colorectal cancer than did those with IBD alone, while those diagnosed at older ages had only about a twofold increase. With regard to colectomy, men diagnosed with PSC at younger ages were at the greatest risk, compared with women or individuals diagnosed after age 50 years. Individuals with ulcerative colitis and PSC had a 40% greater risk for colectomy risk than did IBD-only individuals (time-dependent adjusted HR, 1.65; 95% CI, 1.45-1.85; P less than .001).
“Whilst all-cause mortality rates increase with age, younger patients [with PSC] show a disproportionately increased incidence of liver transplantation, PSC-related death, and colorectal cancer,” the researchers concluded. “Consideration of age at diagnosis should therefore be applied in the stratification of patients for future clinical trials, disease prediction models, and prognostic biomarker discovery.”
Dr. Trivedi disclosed support from the National Institute for Health Research Birmingham Biomedical Research Centre, at the University Hospitals Birmingham NHS Foundation Trust and the University of Birmingham. No other disclosures were reported.
SOURCE: Trivedi PJ et al. Gastroenterology. 2020 May 19. doi: 10.1053/j.gastro.2020.05.049.
FROM GASTROENTEROLOGY




