User login
Team uses iPSCs to create ‘universal’ CAR T cells
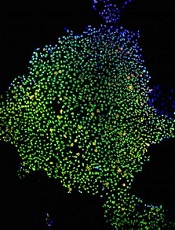
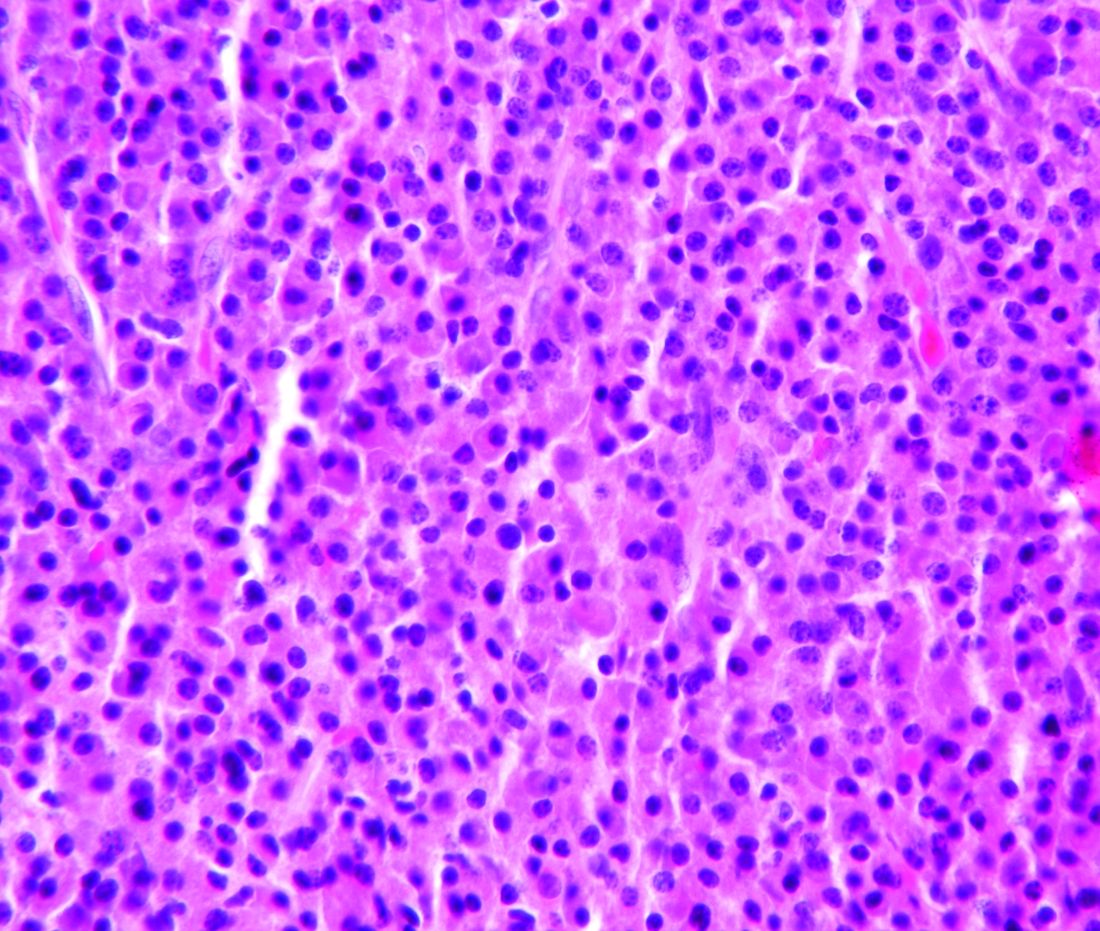
CHICAGO—Researchers have used induced pluripotent stem cells (iPSCs) to create a “universal” chimeric antigen receptor (CAR) T-cell therapy known as FT819.
The team says FT819 has the potential to be mass-produced, stored, and made readily available for cancer patients.
In in vitro experiments, FT819 demonstrated activity against leukemia and lymphoma.
These results were presented at the AACR Annual Meeting 2018 (abstract LB-108).
The research was conducted by employees of Fate Therapeutics, Inc., the company developing FT819, as well as Memorial Sloan-Kettering Cancer Center.
About FT819
FT819 is produced from a master iPSC line generated using T cells from healthy donors.
“A master iPSC line has unlimited capacity to self-renew and can be banked and renewably used,” said Bob Valamehr, PhD, vice-president of cancer immunotherapy at Fate Therapeutics, Inc.
“We started with cells from a healthy donor rather than the patient, created a master cell line, and used the master cell line to produce large quantities of ‘universal’ CAR19 T cells that are not patient-restricted. These first-of-kind CAR19 T cells, called FT819, can be packaged, stored, and made readily available for treatment of a large number of patients.”
FT819 has 2 targeting receptors—a CAR targeting CD19-positive tumor cells and a CD16 Fc receptor that can engage other therapies (such as tumor antigen-targeting monoclonal antibodies) to overcome antigen escape.
The master iPSC line used for the production of FT819 is engineered in a one-time event to insert a CD19 CAR into the T-cell receptor α constant (TRAC) locus. This is done to eliminate T-cell receptor expression and reduce the likelihood of graft-versus-host disease.
Previous research showed that targeting a CAR to the TRAC locus results in uniform CAR expression and enhances T-cell potency. In fact, TRAC-CAR T cells outperformed conventionally generated CAR T cells by preventing T-cell exhaustion in a mouse model of acute lymphoblastic leukemia.
In vitro experiments
With the current work, the researchers found that FT819 displayed an efficient cytotoxic T-cell response when challenged with CD19-positive tumor cells. FT819 produced cytokines (IFN-gamma, TNF-alpha, and IL-2) and mediators of cell death (CD107a/b, perforin, and granzyme B).
FT819 was also target-specific, attacking only CD19-positive tumor cells and sparing CD19-negative tumor cells in experiments with Raji (Burkitt lymphoma) and Nalm-6 (B-cell acute lymphoblastic leukemia) cell lines.
The researchers said they observed consistent antigen-specific cytotoxicity against Nalm-6 cells with FT819 but variability in antigen-specific cytotoxicity with conventional CAR T cells.
In addition, when combined with rituximab, FT819 elicited antibody-dependent cell-mediated cytotoxicity against CD19-negative, CD20-positive tumor cells.
“Through the development of FT819, we believe there is significant opportunity to lower the cost of CAR T-cell manufacture, enhance the quality of the product, and create a readily available supply of a more efficacious product to reach more patients in need,” Dr Valamehr said.
CHICAGO—Researchers have used induced pluripotent stem cells (iPSCs) to create a “universal” chimeric antigen receptor (CAR) T-cell therapy known as FT819.
The team says FT819 has the potential to be mass-produced, stored, and made readily available for cancer patients.
In in vitro experiments, FT819 demonstrated activity against leukemia and lymphoma.
These results were presented at the AACR Annual Meeting 2018 (abstract LB-108).
The research was conducted by employees of Fate Therapeutics, Inc., the company developing FT819, as well as Memorial Sloan-Kettering Cancer Center.
About FT819
FT819 is produced from a master iPSC line generated using T cells from healthy donors.
“A master iPSC line has unlimited capacity to self-renew and can be banked and renewably used,” said Bob Valamehr, PhD, vice-president of cancer immunotherapy at Fate Therapeutics, Inc.
“We started with cells from a healthy donor rather than the patient, created a master cell line, and used the master cell line to produce large quantities of ‘universal’ CAR19 T cells that are not patient-restricted. These first-of-kind CAR19 T cells, called FT819, can be packaged, stored, and made readily available for treatment of a large number of patients.”
FT819 has 2 targeting receptors—a CAR targeting CD19-positive tumor cells and a CD16 Fc receptor that can engage other therapies (such as tumor antigen-targeting monoclonal antibodies) to overcome antigen escape.
The master iPSC line used for the production of FT819 is engineered in a one-time event to insert a CD19 CAR into the T-cell receptor α constant (TRAC) locus. This is done to eliminate T-cell receptor expression and reduce the likelihood of graft-versus-host disease.
Previous research showed that targeting a CAR to the TRAC locus results in uniform CAR expression and enhances T-cell potency. In fact, TRAC-CAR T cells outperformed conventionally generated CAR T cells by preventing T-cell exhaustion in a mouse model of acute lymphoblastic leukemia.
In vitro experiments
With the current work, the researchers found that FT819 displayed an efficient cytotoxic T-cell response when challenged with CD19-positive tumor cells. FT819 produced cytokines (IFN-gamma, TNF-alpha, and IL-2) and mediators of cell death (CD107a/b, perforin, and granzyme B).
FT819 was also target-specific, attacking only CD19-positive tumor cells and sparing CD19-negative tumor cells in experiments with Raji (Burkitt lymphoma) and Nalm-6 (B-cell acute lymphoblastic leukemia) cell lines.
The researchers said they observed consistent antigen-specific cytotoxicity against Nalm-6 cells with FT819 but variability in antigen-specific cytotoxicity with conventional CAR T cells.
In addition, when combined with rituximab, FT819 elicited antibody-dependent cell-mediated cytotoxicity against CD19-negative, CD20-positive tumor cells.
“Through the development of FT819, we believe there is significant opportunity to lower the cost of CAR T-cell manufacture, enhance the quality of the product, and create a readily available supply of a more efficacious product to reach more patients in need,” Dr Valamehr said.
CHICAGO—Researchers have used induced pluripotent stem cells (iPSCs) to create a “universal” chimeric antigen receptor (CAR) T-cell therapy known as FT819.
The team says FT819 has the potential to be mass-produced, stored, and made readily available for cancer patients.
In in vitro experiments, FT819 demonstrated activity against leukemia and lymphoma.
These results were presented at the AACR Annual Meeting 2018 (abstract LB-108).
The research was conducted by employees of Fate Therapeutics, Inc., the company developing FT819, as well as Memorial Sloan-Kettering Cancer Center.
About FT819
FT819 is produced from a master iPSC line generated using T cells from healthy donors.
“A master iPSC line has unlimited capacity to self-renew and can be banked and renewably used,” said Bob Valamehr, PhD, vice-president of cancer immunotherapy at Fate Therapeutics, Inc.
“We started with cells from a healthy donor rather than the patient, created a master cell line, and used the master cell line to produce large quantities of ‘universal’ CAR19 T cells that are not patient-restricted. These first-of-kind CAR19 T cells, called FT819, can be packaged, stored, and made readily available for treatment of a large number of patients.”
FT819 has 2 targeting receptors—a CAR targeting CD19-positive tumor cells and a CD16 Fc receptor that can engage other therapies (such as tumor antigen-targeting monoclonal antibodies) to overcome antigen escape.
The master iPSC line used for the production of FT819 is engineered in a one-time event to insert a CD19 CAR into the T-cell receptor α constant (TRAC) locus. This is done to eliminate T-cell receptor expression and reduce the likelihood of graft-versus-host disease.
Previous research showed that targeting a CAR to the TRAC locus results in uniform CAR expression and enhances T-cell potency. In fact, TRAC-CAR T cells outperformed conventionally generated CAR T cells by preventing T-cell exhaustion in a mouse model of acute lymphoblastic leukemia.
In vitro experiments
With the current work, the researchers found that FT819 displayed an efficient cytotoxic T-cell response when challenged with CD19-positive tumor cells. FT819 produced cytokines (IFN-gamma, TNF-alpha, and IL-2) and mediators of cell death (CD107a/b, perforin, and granzyme B).
FT819 was also target-specific, attacking only CD19-positive tumor cells and sparing CD19-negative tumor cells in experiments with Raji (Burkitt lymphoma) and Nalm-6 (B-cell acute lymphoblastic leukemia) cell lines.
The researchers said they observed consistent antigen-specific cytotoxicity against Nalm-6 cells with FT819 but variability in antigen-specific cytotoxicity with conventional CAR T cells.
In addition, when combined with rituximab, FT819 elicited antibody-dependent cell-mediated cytotoxicity against CD19-negative, CD20-positive tumor cells.
“Through the development of FT819, we believe there is significant opportunity to lower the cost of CAR T-cell manufacture, enhance the quality of the product, and create a readily available supply of a more efficacious product to reach more patients in need,” Dr Valamehr said.
Groups launch open portal for cancer models
The European Bioinformatics Institute (EMBL-EBI) and The Jackson Laboratory (JAX) have developed an open cancer research portal for patient-derived xenograft (PDX) models.
The portal, known as PDX Finder, catalogues PDX models from numerous global repositories.
At present, PDX Finder contains more than 1900 models for a range of cancers, including hematologic malignancies.
Researchers can search for PDX models and submit their own on the PDX Finder website: http://www.pdxfinder.org.
“PDX models are increasingly recognized as clinically relevant because they retain the patient tumor characteristics and imitate a specific patient’s response to drugs more accurately than other models,” said Nathalie Conte, PDX Finder project lead at EMBL-EBI.
“However, until now, there was no open central catalogue for PDX models. PDX Finder enables cancer researchers to search through a wider variety of models more quickly, saving valuable time and enabling collaborations.”
PDX Finder builds on JAX’s PDX resource, which was developed in partnership with more than 20 medical centers in the US, and EMBL-EBI’s membership of EurOPDX. This ensures the scope and reach of PDX Finder is wider than any other individual resource available, according to JAX and EMBL-EBI.
“Both EMBL-EBI and JAX received independent funding from the National Cancer Institute [NCI] for projects that included implementation of online catalogues for PDX models,” noted Carol Bult, scientific director of the JAX PDX resource.
“Both groups had the goal to help basic and clinical cancer researchers find relevant models fast. With approval from the NCI, we instead collaborated to build a single, unified portal with international scope.”
The launch of PDX Finder comes just months after EMBL-EBI and JAX published the PDX minimal information standard, which sets standards for basic information needed to describe essential properties of a PDX model.
The standard ensures that every model in PDX Finder is carefully described to help researchers choose models that are most relevant to their work.
The European Bioinformatics Institute (EMBL-EBI) and The Jackson Laboratory (JAX) have developed an open cancer research portal for patient-derived xenograft (PDX) models.
The portal, known as PDX Finder, catalogues PDX models from numerous global repositories.
At present, PDX Finder contains more than 1900 models for a range of cancers, including hematologic malignancies.
Researchers can search for PDX models and submit their own on the PDX Finder website: http://www.pdxfinder.org.
“PDX models are increasingly recognized as clinically relevant because they retain the patient tumor characteristics and imitate a specific patient’s response to drugs more accurately than other models,” said Nathalie Conte, PDX Finder project lead at EMBL-EBI.
“However, until now, there was no open central catalogue for PDX models. PDX Finder enables cancer researchers to search through a wider variety of models more quickly, saving valuable time and enabling collaborations.”
PDX Finder builds on JAX’s PDX resource, which was developed in partnership with more than 20 medical centers in the US, and EMBL-EBI’s membership of EurOPDX. This ensures the scope and reach of PDX Finder is wider than any other individual resource available, according to JAX and EMBL-EBI.
“Both EMBL-EBI and JAX received independent funding from the National Cancer Institute [NCI] for projects that included implementation of online catalogues for PDX models,” noted Carol Bult, scientific director of the JAX PDX resource.
“Both groups had the goal to help basic and clinical cancer researchers find relevant models fast. With approval from the NCI, we instead collaborated to build a single, unified portal with international scope.”
The launch of PDX Finder comes just months after EMBL-EBI and JAX published the PDX minimal information standard, which sets standards for basic information needed to describe essential properties of a PDX model.
The standard ensures that every model in PDX Finder is carefully described to help researchers choose models that are most relevant to their work.
The European Bioinformatics Institute (EMBL-EBI) and The Jackson Laboratory (JAX) have developed an open cancer research portal for patient-derived xenograft (PDX) models.
The portal, known as PDX Finder, catalogues PDX models from numerous global repositories.
At present, PDX Finder contains more than 1900 models for a range of cancers, including hematologic malignancies.
Researchers can search for PDX models and submit their own on the PDX Finder website: http://www.pdxfinder.org.
“PDX models are increasingly recognized as clinically relevant because they retain the patient tumor characteristics and imitate a specific patient’s response to drugs more accurately than other models,” said Nathalie Conte, PDX Finder project lead at EMBL-EBI.
“However, until now, there was no open central catalogue for PDX models. PDX Finder enables cancer researchers to search through a wider variety of models more quickly, saving valuable time and enabling collaborations.”
PDX Finder builds on JAX’s PDX resource, which was developed in partnership with more than 20 medical centers in the US, and EMBL-EBI’s membership of EurOPDX. This ensures the scope and reach of PDX Finder is wider than any other individual resource available, according to JAX and EMBL-EBI.
“Both EMBL-EBI and JAX received independent funding from the National Cancer Institute [NCI] for projects that included implementation of online catalogues for PDX models,” noted Carol Bult, scientific director of the JAX PDX resource.
“Both groups had the goal to help basic and clinical cancer researchers find relevant models fast. With approval from the NCI, we instead collaborated to build a single, unified portal with international scope.”
The launch of PDX Finder comes just months after EMBL-EBI and JAX published the PDX minimal information standard, which sets standards for basic information needed to describe essential properties of a PDX model.
The standard ensures that every model in PDX Finder is carefully described to help researchers choose models that are most relevant to their work.
Antibody has ‘potent’ effects against AML
CHICAGO—The bispecific antibody APVO436 has demonstrated robust T-cell activation with limited cytokine release in acute myeloid leukemia (AML), according to researchers.
APVO436 binds CD123 and CD3 to redirect T-cell cytotoxicity against CD123-expressing tumor cells.
Researchers found that APVO436 induced T-cell cytotoxicity in AML cells in vitro and in mouse models.
In addition, levels of several cytokines were lower in experiments with APVO436 than in experiments with a comparator antibody.
These findings were presented in a poster at the AACR Annual Meeting 2018 (abstract 1786).
The research was conducted by employees of Aptevo Therapeutics Inc., the company developing APVO436.
“We are especially excited about these latest data for APVO436, which continue to show robust T-cell engagement and cytotoxic activity with reduced levels of cytokine release,” said Jane Gross, PhD, senior vice president and chief scientific officer for Aptevo.
Dr Gross and her colleagues found that APVO436 binds human CD123 and CD3-expressing cells and has “potent” target-specific activity against CD123-expressing AML cell lines (Molm-13 and KG-1a).
In addition, APVO436 induced endogenous T-cell activation and proliferation, accompanied by depletion of CD123-expressing cells, in samples from AML patients and healthy donors.
T cells from these cultures (both AML and non-AML) were expanded and co-cultured with Molm-13 cells and APVO436 or a control antibody. Again, the researchers observed “potent” cytotoxic activity in the presence of APVO436.
Dr Gross and her colleagues also tested APVO436, co-administered with human T cells, in mice with established disseminated Molm-13 tumors. The treatment resulted in a “rapid and significant” reduction in skeletal tumor burden.
Finally, the team compared APVO436 with an Aptevo-generated version of MGD006, a CD123 x CD3 dual-affinity re-targeting molecule being developed by Macrogenics, Inc.
The researchers took purified T cells from healthy donors and cultured them with Molm-13 cells, as well as APVO436, Aptevo’s version of MGD006, and a control antibody.
Both APVO436 and Aptevo’s version of MGD006 were effective at stimulating a tumor-directed immune response, inducing comparable levels of T-cell activation, proliferation, and cytotoxicity.
However, APVO436 induced lower levels of several cytokines—including IFNγ, IL-2, IL-6, and TNFα.
“Importantly, IFNγ, IL-6, and TNFα are considered to be the most relevant cytokines responsible for dosing toxicities observed in clinical studies with T-cell engaging molecules, which suggests that APVO436 could offer the potential for reduced toxicities compared to other CD123 x CD3 T-cell engagers at comparable or higher doses,” Dr Gross said.
She added that Aptevo is planning to launch a phase 1 trial of APVO436 in patients with AML and myelodysplastic syndromes later this year.
CHICAGO—The bispecific antibody APVO436 has demonstrated robust T-cell activation with limited cytokine release in acute myeloid leukemia (AML), according to researchers.
APVO436 binds CD123 and CD3 to redirect T-cell cytotoxicity against CD123-expressing tumor cells.
Researchers found that APVO436 induced T-cell cytotoxicity in AML cells in vitro and in mouse models.
In addition, levels of several cytokines were lower in experiments with APVO436 than in experiments with a comparator antibody.
These findings were presented in a poster at the AACR Annual Meeting 2018 (abstract 1786).
The research was conducted by employees of Aptevo Therapeutics Inc., the company developing APVO436.
“We are especially excited about these latest data for APVO436, which continue to show robust T-cell engagement and cytotoxic activity with reduced levels of cytokine release,” said Jane Gross, PhD, senior vice president and chief scientific officer for Aptevo.
Dr Gross and her colleagues found that APVO436 binds human CD123 and CD3-expressing cells and has “potent” target-specific activity against CD123-expressing AML cell lines (Molm-13 and KG-1a).
In addition, APVO436 induced endogenous T-cell activation and proliferation, accompanied by depletion of CD123-expressing cells, in samples from AML patients and healthy donors.
T cells from these cultures (both AML and non-AML) were expanded and co-cultured with Molm-13 cells and APVO436 or a control antibody. Again, the researchers observed “potent” cytotoxic activity in the presence of APVO436.
Dr Gross and her colleagues also tested APVO436, co-administered with human T cells, in mice with established disseminated Molm-13 tumors. The treatment resulted in a “rapid and significant” reduction in skeletal tumor burden.
Finally, the team compared APVO436 with an Aptevo-generated version of MGD006, a CD123 x CD3 dual-affinity re-targeting molecule being developed by Macrogenics, Inc.
The researchers took purified T cells from healthy donors and cultured them with Molm-13 cells, as well as APVO436, Aptevo’s version of MGD006, and a control antibody.
Both APVO436 and Aptevo’s version of MGD006 were effective at stimulating a tumor-directed immune response, inducing comparable levels of T-cell activation, proliferation, and cytotoxicity.
However, APVO436 induced lower levels of several cytokines—including IFNγ, IL-2, IL-6, and TNFα.
“Importantly, IFNγ, IL-6, and TNFα are considered to be the most relevant cytokines responsible for dosing toxicities observed in clinical studies with T-cell engaging molecules, which suggests that APVO436 could offer the potential for reduced toxicities compared to other CD123 x CD3 T-cell engagers at comparable or higher doses,” Dr Gross said.
She added that Aptevo is planning to launch a phase 1 trial of APVO436 in patients with AML and myelodysplastic syndromes later this year.
CHICAGO—The bispecific antibody APVO436 has demonstrated robust T-cell activation with limited cytokine release in acute myeloid leukemia (AML), according to researchers.
APVO436 binds CD123 and CD3 to redirect T-cell cytotoxicity against CD123-expressing tumor cells.
Researchers found that APVO436 induced T-cell cytotoxicity in AML cells in vitro and in mouse models.
In addition, levels of several cytokines were lower in experiments with APVO436 than in experiments with a comparator antibody.
These findings were presented in a poster at the AACR Annual Meeting 2018 (abstract 1786).
The research was conducted by employees of Aptevo Therapeutics Inc., the company developing APVO436.
“We are especially excited about these latest data for APVO436, which continue to show robust T-cell engagement and cytotoxic activity with reduced levels of cytokine release,” said Jane Gross, PhD, senior vice president and chief scientific officer for Aptevo.
Dr Gross and her colleagues found that APVO436 binds human CD123 and CD3-expressing cells and has “potent” target-specific activity against CD123-expressing AML cell lines (Molm-13 and KG-1a).
In addition, APVO436 induced endogenous T-cell activation and proliferation, accompanied by depletion of CD123-expressing cells, in samples from AML patients and healthy donors.
T cells from these cultures (both AML and non-AML) were expanded and co-cultured with Molm-13 cells and APVO436 or a control antibody. Again, the researchers observed “potent” cytotoxic activity in the presence of APVO436.
Dr Gross and her colleagues also tested APVO436, co-administered with human T cells, in mice with established disseminated Molm-13 tumors. The treatment resulted in a “rapid and significant” reduction in skeletal tumor burden.
Finally, the team compared APVO436 with an Aptevo-generated version of MGD006, a CD123 x CD3 dual-affinity re-targeting molecule being developed by Macrogenics, Inc.
The researchers took purified T cells from healthy donors and cultured them with Molm-13 cells, as well as APVO436, Aptevo’s version of MGD006, and a control antibody.
Both APVO436 and Aptevo’s version of MGD006 were effective at stimulating a tumor-directed immune response, inducing comparable levels of T-cell activation, proliferation, and cytotoxicity.
However, APVO436 induced lower levels of several cytokines—including IFNγ, IL-2, IL-6, and TNFα.
“Importantly, IFNγ, IL-6, and TNFα are considered to be the most relevant cytokines responsible for dosing toxicities observed in clinical studies with T-cell engaging molecules, which suggests that APVO436 could offer the potential for reduced toxicities compared to other CD123 x CD3 T-cell engagers at comparable or higher doses,” Dr Gross said.
She added that Aptevo is planning to launch a phase 1 trial of APVO436 in patients with AML and myelodysplastic syndromes later this year.
Checkpoint inhibition after HiDAC shows promise in AML
NEWPORT BEACH, CALIF. – Adding pembrolizumab after high-dose cytarabine in patients with relapsed or refractory acute myeloid leukemia (AML) appears safe and feasible and shows promising efficacy, according to early results from a multicenter phase 2 study.
The overall response rate to this novel treatment approach in 19 evaluable patients (of 20 enrolled to date) was 42%, with 7 patients (37%) achieving a complete response or complete response with incomplete blood count recovery, and 1 (5%) achieving a partial response, reported Joshua Zeidner, MD, in a poster at the Acute Leukemia Forum of Hemedicus.
Those who went on to allogeneic stem cell transplant included three patients in complete response and one of the five who received pembrolizumab maintenance. That patient on pembrolizumab maintenance went to transplant after two cycles, three others on it relapsed after a median duration of 2.8 months in complete response, and one initially achieved a partial response and had stable disease for a “pretty remarkable” 12 cycles before progressing, he said.
Preliminary analyses in the first six patients, including three with complete response and three nonresponders, showed increased posttreatment diversity of the T-cell receptor repertoire versus baseline in peripheral blood CD8+ T cells in the complete response patients, compared with nonresponders. This suggests that T-cell diversity at baseline is a promising biomarker for programmed death-1 (PD-1) blockade response, Dr. Zeidner noted.
PD-1 suppresses immune activation, and the PD-1 pathway is exploited by AML cells to evade immune surveillance, Dr. Zeidner explained. PD-1 blockade has been shown to have antileukemic effects in vivo, there is expression of multiple coinhibitory receptors in AML patients at the time of diagnosis (that persists in refractory AML), and the ligand for PD-1 is up-regulated on AML blasts, particularly in relapsed/refractory disease, he added.
He and his colleagues hypothesized that targeting PD-1 with pembrolizumab after high-dose cytarabine (HiDAC) salvage chemotherapy would stimulate a T cell–mediated antileukemic immune response and lead to improved efficacy in patients with relapsed or refractory AML.
The ongoing study, with planned enrollment of 37 patients, includes patients aged 18-70 years with relapsed/refractory AML (median age of first 20 enrolled is 54 years). Those under age 60 years receive 2 g/m2 of intravenous HiDAC every 12 hours on days 1-5, and those over age 60 years receive 1.5 g/m2 every 12 hours on days 1-5. In both age groups, this is followed by intravenous pembrolizumab given at 200 mg on day 14.
Overall responders receive maintenance phase intravenous pembrolizumab at 200 mg every 3 weeks for up to 2 years until relapse or progression. Patients are allowed to proceed to stem cell transplant before or after the maintenance phase.
A number of correlative studies, including serial peripheral blood and bone marrow flow cytometry and T-cell receptor clonality studies, also are being conducted to look for predictive biomarkers of response, Dr. Zeidner said.
The study population to date is a relatively young, very-high-risk subgroup of AML patients; European LeukemiaNet-2017 genetic risk status was favorable in only 3 of the first 20 patients (15%), intermediate in 7 (35%), and adverse in 10 (50%). Treatment has been well tolerated; toxicities have been rare and manageable. “Overall, there have been no unexpected toxicities,” he said.
The most common overall toxicities included febrile neutropenia in 70% of patients; transaminitis (including one grade 3 case) in 50%, hyperbilirubinemia in 35%, and fatigue, increased alkaline phosphatase, and rash in 25% each.
Novel therapies for relapsed/refractory AML are urgently needed, and these early results demonstrated the safety and feasibility of adding pembrolizumab after HiDAC chemotherapy in relapsed/refractory AML patients. The preliminary finding that broadening of the immune repertoire, which may occur via increasing T-cell responses beyond endogenous viral responses, was associated with complete response was particularly encouraging, Dr. Zeidner said. The investigators hope to determine predictors of response to immune checkpoint blockade in AML through a comprehensive immune biomarker discovery approach, he added.
This study was funded by Merck. Dr. Zeidner reported having no financial disclosures. The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
SOURCE: Zeidner J et al. ALF 2018, Poster Session.
NEWPORT BEACH, CALIF. – Adding pembrolizumab after high-dose cytarabine in patients with relapsed or refractory acute myeloid leukemia (AML) appears safe and feasible and shows promising efficacy, according to early results from a multicenter phase 2 study.
The overall response rate to this novel treatment approach in 19 evaluable patients (of 20 enrolled to date) was 42%, with 7 patients (37%) achieving a complete response or complete response with incomplete blood count recovery, and 1 (5%) achieving a partial response, reported Joshua Zeidner, MD, in a poster at the Acute Leukemia Forum of Hemedicus.
Those who went on to allogeneic stem cell transplant included three patients in complete response and one of the five who received pembrolizumab maintenance. That patient on pembrolizumab maintenance went to transplant after two cycles, three others on it relapsed after a median duration of 2.8 months in complete response, and one initially achieved a partial response and had stable disease for a “pretty remarkable” 12 cycles before progressing, he said.
Preliminary analyses in the first six patients, including three with complete response and three nonresponders, showed increased posttreatment diversity of the T-cell receptor repertoire versus baseline in peripheral blood CD8+ T cells in the complete response patients, compared with nonresponders. This suggests that T-cell diversity at baseline is a promising biomarker for programmed death-1 (PD-1) blockade response, Dr. Zeidner noted.
PD-1 suppresses immune activation, and the PD-1 pathway is exploited by AML cells to evade immune surveillance, Dr. Zeidner explained. PD-1 blockade has been shown to have antileukemic effects in vivo, there is expression of multiple coinhibitory receptors in AML patients at the time of diagnosis (that persists in refractory AML), and the ligand for PD-1 is up-regulated on AML blasts, particularly in relapsed/refractory disease, he added.
He and his colleagues hypothesized that targeting PD-1 with pembrolizumab after high-dose cytarabine (HiDAC) salvage chemotherapy would stimulate a T cell–mediated antileukemic immune response and lead to improved efficacy in patients with relapsed or refractory AML.
The ongoing study, with planned enrollment of 37 patients, includes patients aged 18-70 years with relapsed/refractory AML (median age of first 20 enrolled is 54 years). Those under age 60 years receive 2 g/m2 of intravenous HiDAC every 12 hours on days 1-5, and those over age 60 years receive 1.5 g/m2 every 12 hours on days 1-5. In both age groups, this is followed by intravenous pembrolizumab given at 200 mg on day 14.
Overall responders receive maintenance phase intravenous pembrolizumab at 200 mg every 3 weeks for up to 2 years until relapse or progression. Patients are allowed to proceed to stem cell transplant before or after the maintenance phase.
A number of correlative studies, including serial peripheral blood and bone marrow flow cytometry and T-cell receptor clonality studies, also are being conducted to look for predictive biomarkers of response, Dr. Zeidner said.
The study population to date is a relatively young, very-high-risk subgroup of AML patients; European LeukemiaNet-2017 genetic risk status was favorable in only 3 of the first 20 patients (15%), intermediate in 7 (35%), and adverse in 10 (50%). Treatment has been well tolerated; toxicities have been rare and manageable. “Overall, there have been no unexpected toxicities,” he said.
The most common overall toxicities included febrile neutropenia in 70% of patients; transaminitis (including one grade 3 case) in 50%, hyperbilirubinemia in 35%, and fatigue, increased alkaline phosphatase, and rash in 25% each.
Novel therapies for relapsed/refractory AML are urgently needed, and these early results demonstrated the safety and feasibility of adding pembrolizumab after HiDAC chemotherapy in relapsed/refractory AML patients. The preliminary finding that broadening of the immune repertoire, which may occur via increasing T-cell responses beyond endogenous viral responses, was associated with complete response was particularly encouraging, Dr. Zeidner said. The investigators hope to determine predictors of response to immune checkpoint blockade in AML through a comprehensive immune biomarker discovery approach, he added.
This study was funded by Merck. Dr. Zeidner reported having no financial disclosures. The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
SOURCE: Zeidner J et al. ALF 2018, Poster Session.
NEWPORT BEACH, CALIF. – Adding pembrolizumab after high-dose cytarabine in patients with relapsed or refractory acute myeloid leukemia (AML) appears safe and feasible and shows promising efficacy, according to early results from a multicenter phase 2 study.
The overall response rate to this novel treatment approach in 19 evaluable patients (of 20 enrolled to date) was 42%, with 7 patients (37%) achieving a complete response or complete response with incomplete blood count recovery, and 1 (5%) achieving a partial response, reported Joshua Zeidner, MD, in a poster at the Acute Leukemia Forum of Hemedicus.
Those who went on to allogeneic stem cell transplant included three patients in complete response and one of the five who received pembrolizumab maintenance. That patient on pembrolizumab maintenance went to transplant after two cycles, three others on it relapsed after a median duration of 2.8 months in complete response, and one initially achieved a partial response and had stable disease for a “pretty remarkable” 12 cycles before progressing, he said.
Preliminary analyses in the first six patients, including three with complete response and three nonresponders, showed increased posttreatment diversity of the T-cell receptor repertoire versus baseline in peripheral blood CD8+ T cells in the complete response patients, compared with nonresponders. This suggests that T-cell diversity at baseline is a promising biomarker for programmed death-1 (PD-1) blockade response, Dr. Zeidner noted.
PD-1 suppresses immune activation, and the PD-1 pathway is exploited by AML cells to evade immune surveillance, Dr. Zeidner explained. PD-1 blockade has been shown to have antileukemic effects in vivo, there is expression of multiple coinhibitory receptors in AML patients at the time of diagnosis (that persists in refractory AML), and the ligand for PD-1 is up-regulated on AML blasts, particularly in relapsed/refractory disease, he added.
He and his colleagues hypothesized that targeting PD-1 with pembrolizumab after high-dose cytarabine (HiDAC) salvage chemotherapy would stimulate a T cell–mediated antileukemic immune response and lead to improved efficacy in patients with relapsed or refractory AML.
The ongoing study, with planned enrollment of 37 patients, includes patients aged 18-70 years with relapsed/refractory AML (median age of first 20 enrolled is 54 years). Those under age 60 years receive 2 g/m2 of intravenous HiDAC every 12 hours on days 1-5, and those over age 60 years receive 1.5 g/m2 every 12 hours on days 1-5. In both age groups, this is followed by intravenous pembrolizumab given at 200 mg on day 14.
Overall responders receive maintenance phase intravenous pembrolizumab at 200 mg every 3 weeks for up to 2 years until relapse or progression. Patients are allowed to proceed to stem cell transplant before or after the maintenance phase.
A number of correlative studies, including serial peripheral blood and bone marrow flow cytometry and T-cell receptor clonality studies, also are being conducted to look for predictive biomarkers of response, Dr. Zeidner said.
The study population to date is a relatively young, very-high-risk subgroup of AML patients; European LeukemiaNet-2017 genetic risk status was favorable in only 3 of the first 20 patients (15%), intermediate in 7 (35%), and adverse in 10 (50%). Treatment has been well tolerated; toxicities have been rare and manageable. “Overall, there have been no unexpected toxicities,” he said.
The most common overall toxicities included febrile neutropenia in 70% of patients; transaminitis (including one grade 3 case) in 50%, hyperbilirubinemia in 35%, and fatigue, increased alkaline phosphatase, and rash in 25% each.
Novel therapies for relapsed/refractory AML are urgently needed, and these early results demonstrated the safety and feasibility of adding pembrolizumab after HiDAC chemotherapy in relapsed/refractory AML patients. The preliminary finding that broadening of the immune repertoire, which may occur via increasing T-cell responses beyond endogenous viral responses, was associated with complete response was particularly encouraging, Dr. Zeidner said. The investigators hope to determine predictors of response to immune checkpoint blockade in AML through a comprehensive immune biomarker discovery approach, he added.
This study was funded by Merck. Dr. Zeidner reported having no financial disclosures. The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
SOURCE: Zeidner J et al. ALF 2018, Poster Session.
REPORTING FROM ALF 2018
Key clinical point:
Major finding: The overall response rate was 42%; 37% of patients achieved complete response or CR with incomplete blood count recovery, and 5% achieved partial response.
Study details: A multicenter, phase 2 study with early results from the first 20 patients enrolled.
Disclosures: This study was funded by Merck. Dr. Zeidner reported having no financial disclosures. The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Source: Zeidner J et al. ALF 2018, Poster Session.
Venetoclax shows muscle against CLL relapsed after idelalisib
For patients with relapsed or refractory chronic lymphocytic leukemia despite therapy with idelalisib (Zydelig), venetoclax (Venclexta) was associated with relatively high overall response and progression-free survival rates, results of a phase 2 study show.
Among 36 patients with relapsed/refractory CLL who had received idelalisib as their most recent B-cell receptor pathway inhibitor (BCRi), the overall response rate (ORR) was 67%, and median progression-free and overall survival (PFS and OS) had not been reached after 14 months of follow-up, reported Steven Coutre, MD, of Stanford (Calif.) University, and his colleagues.
“[V]enetoclax monotherapy is active and well-tolerated in patients with CLL progression after therapy with idelalisib, including a significant number of patients who also received prior therapy with ibrutinib [Imbruvica]. These results from the first prospective trial in this high-risk population provide evidence that venetoclax should be considered as a treatment option for such patients,” the investigators wrote. The report was published in Blood.
In clinical trials with idelalisib, approximately one-third of patients with CLL experienced disease progression on therapy, and other patients had to discontinue the drug, an inhibitor of the delta isoform of phosphoinositide 3-kinase (PI3K), because of toxicities, the investigators noted.
“The optimal treatment of patients with CLL progressing after idelalisib has not been well characterized,” they wrote. “Outcomes in patients who discontinued idelalisib treatment early are poor, with one retrospective analysis reporting a median overall survival (OS) after idelalisib discontinuation of approximately 2 months (range, 0-10 months).”
Venetoclax, an inhibitor of the apoptotic BCL-2 protein, has been shown to have activity against CLL, including in patients with high-risk features such as the chromosome 17p deletion (del17p), prompting the investigators to evaluate it as a follow-on in patients with relapsed/refractory CLL treated with a B-cell receptor pathway inhibitor.
They reported on the idelalisib cohort in a phase 2 trial in which patients with CLL that progressed on either idelalisib or ibrutinib were subsequently treated with venetoclax. The patients in this analysis included those treated with idelalisib in the main study cohort or an expansion cohort.
Patients were started on venetoclax 20 mg daily, followed by weekly dose escalations to a target of 400 mg daily by week 5, or to a maximum of 600 mg for patients who did not have a response by the week 12 assessment.
The overall response rate – the primary efficacy endpoint – was 67%. There were two complete remissions (CR) and one CR with incomplete bone marrow recovery. The remaining 21 patients with responses had partial responses.
At a median of 14 months of follow-up, neither median PFS, duration of response, or OS had been reached.
The investigator-estimated 12-month PFS rate was 79%.
The most common grade 3 or 4 adverse events were neutropenia in 50% of patients, thrombocytopenia in 25%, and anemia in 17%. There were no cases of clinical tumor lysis syndrome, which has been known to occur when venetoclax is initiated at full dose without a ramp-up.
The most common adverse events of any grade included neutropenia, diarrhea, upper respiratory tract infection, thrombocytopenia, nausea, fatigue, cough, rash, and anemia.
“The low number of CRs reported at the time of analysis may be a result of the follow-up time, particularly for patients in the expansion cohort, as other clinical studies with venetoclax report CR occurring after 1 year on therapy. Patients with prior ibrutinib exposure who had progressed on idelalisib as their most recent therapy before study entry had similar efficacy results,” the investigators wrote.
Genentech and AbbVie funded the study. Dr. Coutre is an advisory board member for both companies and others, and receives institutional funding from AbbVie and others. Multiple coauthors disclosed financial relationships with AbbVie, Genentech, or both, as well as other companies.
SOURCE: Coutre S et al. Blood. 2018;131(15):1704-11.
For patients with relapsed or refractory chronic lymphocytic leukemia despite therapy with idelalisib (Zydelig), venetoclax (Venclexta) was associated with relatively high overall response and progression-free survival rates, results of a phase 2 study show.
Among 36 patients with relapsed/refractory CLL who had received idelalisib as their most recent B-cell receptor pathway inhibitor (BCRi), the overall response rate (ORR) was 67%, and median progression-free and overall survival (PFS and OS) had not been reached after 14 months of follow-up, reported Steven Coutre, MD, of Stanford (Calif.) University, and his colleagues.
“[V]enetoclax monotherapy is active and well-tolerated in patients with CLL progression after therapy with idelalisib, including a significant number of patients who also received prior therapy with ibrutinib [Imbruvica]. These results from the first prospective trial in this high-risk population provide evidence that venetoclax should be considered as a treatment option for such patients,” the investigators wrote. The report was published in Blood.
In clinical trials with idelalisib, approximately one-third of patients with CLL experienced disease progression on therapy, and other patients had to discontinue the drug, an inhibitor of the delta isoform of phosphoinositide 3-kinase (PI3K), because of toxicities, the investigators noted.
“The optimal treatment of patients with CLL progressing after idelalisib has not been well characterized,” they wrote. “Outcomes in patients who discontinued idelalisib treatment early are poor, with one retrospective analysis reporting a median overall survival (OS) after idelalisib discontinuation of approximately 2 months (range, 0-10 months).”
Venetoclax, an inhibitor of the apoptotic BCL-2 protein, has been shown to have activity against CLL, including in patients with high-risk features such as the chromosome 17p deletion (del17p), prompting the investigators to evaluate it as a follow-on in patients with relapsed/refractory CLL treated with a B-cell receptor pathway inhibitor.
They reported on the idelalisib cohort in a phase 2 trial in which patients with CLL that progressed on either idelalisib or ibrutinib were subsequently treated with venetoclax. The patients in this analysis included those treated with idelalisib in the main study cohort or an expansion cohort.
Patients were started on venetoclax 20 mg daily, followed by weekly dose escalations to a target of 400 mg daily by week 5, or to a maximum of 600 mg for patients who did not have a response by the week 12 assessment.
The overall response rate – the primary efficacy endpoint – was 67%. There were two complete remissions (CR) and one CR with incomplete bone marrow recovery. The remaining 21 patients with responses had partial responses.
At a median of 14 months of follow-up, neither median PFS, duration of response, or OS had been reached.
The investigator-estimated 12-month PFS rate was 79%.
The most common grade 3 or 4 adverse events were neutropenia in 50% of patients, thrombocytopenia in 25%, and anemia in 17%. There were no cases of clinical tumor lysis syndrome, which has been known to occur when venetoclax is initiated at full dose without a ramp-up.
The most common adverse events of any grade included neutropenia, diarrhea, upper respiratory tract infection, thrombocytopenia, nausea, fatigue, cough, rash, and anemia.
“The low number of CRs reported at the time of analysis may be a result of the follow-up time, particularly for patients in the expansion cohort, as other clinical studies with venetoclax report CR occurring after 1 year on therapy. Patients with prior ibrutinib exposure who had progressed on idelalisib as their most recent therapy before study entry had similar efficacy results,” the investigators wrote.
Genentech and AbbVie funded the study. Dr. Coutre is an advisory board member for both companies and others, and receives institutional funding from AbbVie and others. Multiple coauthors disclosed financial relationships with AbbVie, Genentech, or both, as well as other companies.
SOURCE: Coutre S et al. Blood. 2018;131(15):1704-11.
For patients with relapsed or refractory chronic lymphocytic leukemia despite therapy with idelalisib (Zydelig), venetoclax (Venclexta) was associated with relatively high overall response and progression-free survival rates, results of a phase 2 study show.
Among 36 patients with relapsed/refractory CLL who had received idelalisib as their most recent B-cell receptor pathway inhibitor (BCRi), the overall response rate (ORR) was 67%, and median progression-free and overall survival (PFS and OS) had not been reached after 14 months of follow-up, reported Steven Coutre, MD, of Stanford (Calif.) University, and his colleagues.
“[V]enetoclax monotherapy is active and well-tolerated in patients with CLL progression after therapy with idelalisib, including a significant number of patients who also received prior therapy with ibrutinib [Imbruvica]. These results from the first prospective trial in this high-risk population provide evidence that venetoclax should be considered as a treatment option for such patients,” the investigators wrote. The report was published in Blood.
In clinical trials with idelalisib, approximately one-third of patients with CLL experienced disease progression on therapy, and other patients had to discontinue the drug, an inhibitor of the delta isoform of phosphoinositide 3-kinase (PI3K), because of toxicities, the investigators noted.
“The optimal treatment of patients with CLL progressing after idelalisib has not been well characterized,” they wrote. “Outcomes in patients who discontinued idelalisib treatment early are poor, with one retrospective analysis reporting a median overall survival (OS) after idelalisib discontinuation of approximately 2 months (range, 0-10 months).”
Venetoclax, an inhibitor of the apoptotic BCL-2 protein, has been shown to have activity against CLL, including in patients with high-risk features such as the chromosome 17p deletion (del17p), prompting the investigators to evaluate it as a follow-on in patients with relapsed/refractory CLL treated with a B-cell receptor pathway inhibitor.
They reported on the idelalisib cohort in a phase 2 trial in which patients with CLL that progressed on either idelalisib or ibrutinib were subsequently treated with venetoclax. The patients in this analysis included those treated with idelalisib in the main study cohort or an expansion cohort.
Patients were started on venetoclax 20 mg daily, followed by weekly dose escalations to a target of 400 mg daily by week 5, or to a maximum of 600 mg for patients who did not have a response by the week 12 assessment.
The overall response rate – the primary efficacy endpoint – was 67%. There were two complete remissions (CR) and one CR with incomplete bone marrow recovery. The remaining 21 patients with responses had partial responses.
At a median of 14 months of follow-up, neither median PFS, duration of response, or OS had been reached.
The investigator-estimated 12-month PFS rate was 79%.
The most common grade 3 or 4 adverse events were neutropenia in 50% of patients, thrombocytopenia in 25%, and anemia in 17%. There were no cases of clinical tumor lysis syndrome, which has been known to occur when venetoclax is initiated at full dose without a ramp-up.
The most common adverse events of any grade included neutropenia, diarrhea, upper respiratory tract infection, thrombocytopenia, nausea, fatigue, cough, rash, and anemia.
“The low number of CRs reported at the time of analysis may be a result of the follow-up time, particularly for patients in the expansion cohort, as other clinical studies with venetoclax report CR occurring after 1 year on therapy. Patients with prior ibrutinib exposure who had progressed on idelalisib as their most recent therapy before study entry had similar efficacy results,” the investigators wrote.
Genentech and AbbVie funded the study. Dr. Coutre is an advisory board member for both companies and others, and receives institutional funding from AbbVie and others. Multiple coauthors disclosed financial relationships with AbbVie, Genentech, or both, as well as other companies.
SOURCE: Coutre S et al. Blood. 2018;131(15):1704-11.
FROM BLOOD
Key clinical point:
Major finding: The overall response rate was 67%, including two complete responses (CRs) and one CR with incomplete bone marrow recovery.
Study details: Cohort of 36 patients with relapsed/refractory CLL previously treated with idelalisib.
Disclosures: Genentech and AbbVie funded the study. Dr. Coutre is an advisory board member for both companies and others, and receives institutional funding from AbbVie and others. Multiple coauthors disclosed financial relationships with AbbVie, Genentech, or both, as well as other companies.
Source: Coutre S et al. Blood. 2018;131(15):1704-11.
Ibrutinib plus carfilzomib active in relapsed multiple myeloma
In patients with multiple myeloma who had already undergone multiple lines of therapy, the combination of ibrutinib and carfilzomib with or without dexamethasone produced a favorable rate of response and progression-free survival.
Ibrutinib and carfilzomib combination therapy was also associated with manageable safety in the phase 1 results of the ongoing study, investigators wrote in the journal Leukemia & Lymphoma.
The reported overall response rate in the study was 67%. Median progression-free survival was 7.2 months, which was “encouraging considering the poor outcomes often observed in this group,” Dr. Chari and colleagues wrote.
Carfilzomib, a selective and irreversible proteasome inhibitor, is indicated for patients with previously treated multiple myeloma. Ibrutinib, an orally administered covalent inhibitor of Bruton tyrosine kinase, already has Food and Drug Administration approval for treatment of several B-cell malignancies. Bruton tyrosine kinase is expressed in more than 85% of myeloma tumor cells, according to the researchers, and its signaling may contribute to development of drug resistance.
In an earlier phase 2 study, ibrutinib plus dexamethasone produced a 28% clinical benefit rate and median progression-free survival of 4.6 months. Preclinical data suggest combining ibrutinib and carfilzomib may have a synergistic effect, according to researchers.
To test that hypothesis, Dr. Chari and colleagues conducted a phase 1 dose-finding study including 43 myeloma patients who had previously received at least two lines of therapy, including bortezomib and an immunomodulatory agent. Among those patients, about three-quarters were refractory to bortezomib and one-quarter had high-risk cytogenetics.
A total of 35 patients in the study received ibrutinib and carfilzomib plus dexamethasone; 8 patients received only ibrutinib and carfilzomib.
Key phase 1 results included the 67% response rate, of which 21% had very good partial response and 2% had stringent complete response. No dose-limiting toxicities were observed.
Both carfilzomib and ibrutinib have known cardiac toxicities, but cardiac adverse events in this study did not reach dose-limiting toxicity criteria and were effectively managed by dose reductions or rechallenge, researchers reported.
Hypertension, anemia, pneumonia, fatigue, diarrhea, and thrombocytopenia were the most common grade 3 events observed. “Ultimately, toxicities and/or treatment discontinuations could be attributed to comorbidities, underlying disease factors, or toxicities due to prior therapies,” the researchers wrote.
Enrollment in phase 2b of the study is ongoing.
Dr. Chari received research funding from Pharmacyclics during the study and grants from other companies outside of the study work.
SOURCE: Chari A et al. Leuk Lymphoma. 2018 Apr 4. doi: 10.1080/10428194.2018.1443337.
In patients with multiple myeloma who had already undergone multiple lines of therapy, the combination of ibrutinib and carfilzomib with or without dexamethasone produced a favorable rate of response and progression-free survival.
Ibrutinib and carfilzomib combination therapy was also associated with manageable safety in the phase 1 results of the ongoing study, investigators wrote in the journal Leukemia & Lymphoma.
The reported overall response rate in the study was 67%. Median progression-free survival was 7.2 months, which was “encouraging considering the poor outcomes often observed in this group,” Dr. Chari and colleagues wrote.
Carfilzomib, a selective and irreversible proteasome inhibitor, is indicated for patients with previously treated multiple myeloma. Ibrutinib, an orally administered covalent inhibitor of Bruton tyrosine kinase, already has Food and Drug Administration approval for treatment of several B-cell malignancies. Bruton tyrosine kinase is expressed in more than 85% of myeloma tumor cells, according to the researchers, and its signaling may contribute to development of drug resistance.
In an earlier phase 2 study, ibrutinib plus dexamethasone produced a 28% clinical benefit rate and median progression-free survival of 4.6 months. Preclinical data suggest combining ibrutinib and carfilzomib may have a synergistic effect, according to researchers.
To test that hypothesis, Dr. Chari and colleagues conducted a phase 1 dose-finding study including 43 myeloma patients who had previously received at least two lines of therapy, including bortezomib and an immunomodulatory agent. Among those patients, about three-quarters were refractory to bortezomib and one-quarter had high-risk cytogenetics.
A total of 35 patients in the study received ibrutinib and carfilzomib plus dexamethasone; 8 patients received only ibrutinib and carfilzomib.
Key phase 1 results included the 67% response rate, of which 21% had very good partial response and 2% had stringent complete response. No dose-limiting toxicities were observed.
Both carfilzomib and ibrutinib have known cardiac toxicities, but cardiac adverse events in this study did not reach dose-limiting toxicity criteria and were effectively managed by dose reductions or rechallenge, researchers reported.
Hypertension, anemia, pneumonia, fatigue, diarrhea, and thrombocytopenia were the most common grade 3 events observed. “Ultimately, toxicities and/or treatment discontinuations could be attributed to comorbidities, underlying disease factors, or toxicities due to prior therapies,” the researchers wrote.
Enrollment in phase 2b of the study is ongoing.
Dr. Chari received research funding from Pharmacyclics during the study and grants from other companies outside of the study work.
SOURCE: Chari A et al. Leuk Lymphoma. 2018 Apr 4. doi: 10.1080/10428194.2018.1443337.
In patients with multiple myeloma who had already undergone multiple lines of therapy, the combination of ibrutinib and carfilzomib with or without dexamethasone produced a favorable rate of response and progression-free survival.
Ibrutinib and carfilzomib combination therapy was also associated with manageable safety in the phase 1 results of the ongoing study, investigators wrote in the journal Leukemia & Lymphoma.
The reported overall response rate in the study was 67%. Median progression-free survival was 7.2 months, which was “encouraging considering the poor outcomes often observed in this group,” Dr. Chari and colleagues wrote.
Carfilzomib, a selective and irreversible proteasome inhibitor, is indicated for patients with previously treated multiple myeloma. Ibrutinib, an orally administered covalent inhibitor of Bruton tyrosine kinase, already has Food and Drug Administration approval for treatment of several B-cell malignancies. Bruton tyrosine kinase is expressed in more than 85% of myeloma tumor cells, according to the researchers, and its signaling may contribute to development of drug resistance.
In an earlier phase 2 study, ibrutinib plus dexamethasone produced a 28% clinical benefit rate and median progression-free survival of 4.6 months. Preclinical data suggest combining ibrutinib and carfilzomib may have a synergistic effect, according to researchers.
To test that hypothesis, Dr. Chari and colleagues conducted a phase 1 dose-finding study including 43 myeloma patients who had previously received at least two lines of therapy, including bortezomib and an immunomodulatory agent. Among those patients, about three-quarters were refractory to bortezomib and one-quarter had high-risk cytogenetics.
A total of 35 patients in the study received ibrutinib and carfilzomib plus dexamethasone; 8 patients received only ibrutinib and carfilzomib.
Key phase 1 results included the 67% response rate, of which 21% had very good partial response and 2% had stringent complete response. No dose-limiting toxicities were observed.
Both carfilzomib and ibrutinib have known cardiac toxicities, but cardiac adverse events in this study did not reach dose-limiting toxicity criteria and were effectively managed by dose reductions or rechallenge, researchers reported.
Hypertension, anemia, pneumonia, fatigue, diarrhea, and thrombocytopenia were the most common grade 3 events observed. “Ultimately, toxicities and/or treatment discontinuations could be attributed to comorbidities, underlying disease factors, or toxicities due to prior therapies,” the researchers wrote.
Enrollment in phase 2b of the study is ongoing.
Dr. Chari received research funding from Pharmacyclics during the study and grants from other companies outside of the study work.
SOURCE: Chari A et al. Leuk Lymphoma. 2018 Apr 4. doi: 10.1080/10428194.2018.1443337.
FROM LEUKEMIA & LYMPHOMA
Key clinical point:
Major finding: The overall response rate was 67%, including a 21% very good partial response and 2% stringent complete response.
Study details: A phase 1 dose-finding study including 43 myeloma patients who had previously received at least two lines of therapy.
Disclosures: Dr. Chari received research funding from Pharmacyclics during the study and grants from other companies outside of the study work.
Source: Chari A et al. Leuk Lymphoma. 2018 Apr 4. doi: 10.1080/10428194.2018.1443337.
5- and 10-day decitabine go head-to-head in poor-risk AML
NEWPORT BEACH, CALIF. – Response and survival rates are similar with 5- and 10-day dosing of decitabine in poor-risk older adults with previously untreated acute myeloid leukemia, according to findings from a randomized phase 2 trial.
The overall response rates in 25 patients randomized to receive 5 days of decitabine and 40 patients randomized to receive 10 days of decitabine were 44% and 38%, respectively. Complete response rates were 28% and 30%, respectively, and the median number of cycles to best response was two in both arms, Nicholas J. Short, MD, reported in a poster at the Acute Leukemia Forum of Hemedicus.
Patients were followed for a median of 38 months. Median remission duration was 9.4 months in 11 responders who received 5 days of decitabine, and 6.4 months in 15 responders in the 10-day treatment arm. The 1-year continued remission rates were 25% and 30%, respectively (P = .85). Median overall survival was 4.9 and 7.1 months, respectively, and 1-year overall survival rates were 27% and 25%, respectively, he said, adding that none of the differences in the groups were statistically significant.
Further, no differences in survival were seen when patients in the two treatment arms were stratified by cytogenetics, de novo versus secondary or treatment-related disease, or TP53 mutations; however, these subgroups were small, he noted.
Study subjects were adults aged 60 years or older (median, 77 and 78 years) with newly diagnosed and untreated acute myeloid leukemia (AML), and adults under age 60 years who were not suitable candidates for intensive chemotherapy. They were enrolled between February 2013 and July 2017. About 30% of patients had performance status scores of 2-3, and 45% of patients had secondary AML.
“About 40% of patients in the 5-day arm had poor-risk cytogenetics, and about half in the 10-day arm had poor-risk cytogenetics,” Dr. Short said in an interview, adding that 6 of 21 tested patients in the 5-day arm and 16 of 38 tested patients in the 10-day arm had a TP53 mutation.
Patients received intravenous decitabine at a dose of 20 mg/m2 for the assigned duration of either 5 or 10 consecutive days for the first three cycles of induction, and those who responded to treatment received additional consolidation cycles every 4-8 weeks for up to 24 total cycles. Patients in the 10-day arm were switched to 5-day dosing after achieving complete response or complete response with incomplete blood count recovery.
The groups were well balanced with respect to baseline characteristics and the imbalance in patient numbers in the arms was mainly because of adaptive randomization; the 10-day patients had better responses during the early part of the study, Dr. Short explained.
None of the patients underwent allogeneic stem cell transplantation, he added.
Older patients with acute AML often have poor tolerance for intensive chemotherapy, but decitabine has been shown to improve survival when compared with supportive care or low-dose cytarabine in these patients. However, while some single-arm studies have suggested that 5- and 10-day dosing may result in similar outcomes, and another suggested that 10-day dosing may be superior in TP53-mutated AML, no studies have directly compared 5- and 10-day dosing, he said.
“In this relatively poor-risk cohort of older adults with newly diagnosed AML, decitabine for 5 and 10 days resulted in similar response rates, survival, and early mortality,” he concluded.
This study was funded by a University of Texas MD Anderson Cancer Center support grant. Dr. Short reported having no financial disclosures.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
SOURCE: Short NJ et al. ALF 2018, Poster Session.
NEWPORT BEACH, CALIF. – Response and survival rates are similar with 5- and 10-day dosing of decitabine in poor-risk older adults with previously untreated acute myeloid leukemia, according to findings from a randomized phase 2 trial.
The overall response rates in 25 patients randomized to receive 5 days of decitabine and 40 patients randomized to receive 10 days of decitabine were 44% and 38%, respectively. Complete response rates were 28% and 30%, respectively, and the median number of cycles to best response was two in both arms, Nicholas J. Short, MD, reported in a poster at the Acute Leukemia Forum of Hemedicus.
Patients were followed for a median of 38 months. Median remission duration was 9.4 months in 11 responders who received 5 days of decitabine, and 6.4 months in 15 responders in the 10-day treatment arm. The 1-year continued remission rates were 25% and 30%, respectively (P = .85). Median overall survival was 4.9 and 7.1 months, respectively, and 1-year overall survival rates were 27% and 25%, respectively, he said, adding that none of the differences in the groups were statistically significant.
Further, no differences in survival were seen when patients in the two treatment arms were stratified by cytogenetics, de novo versus secondary or treatment-related disease, or TP53 mutations; however, these subgroups were small, he noted.
Study subjects were adults aged 60 years or older (median, 77 and 78 years) with newly diagnosed and untreated acute myeloid leukemia (AML), and adults under age 60 years who were not suitable candidates for intensive chemotherapy. They were enrolled between February 2013 and July 2017. About 30% of patients had performance status scores of 2-3, and 45% of patients had secondary AML.
“About 40% of patients in the 5-day arm had poor-risk cytogenetics, and about half in the 10-day arm had poor-risk cytogenetics,” Dr. Short said in an interview, adding that 6 of 21 tested patients in the 5-day arm and 16 of 38 tested patients in the 10-day arm had a TP53 mutation.
Patients received intravenous decitabine at a dose of 20 mg/m2 for the assigned duration of either 5 or 10 consecutive days for the first three cycles of induction, and those who responded to treatment received additional consolidation cycles every 4-8 weeks for up to 24 total cycles. Patients in the 10-day arm were switched to 5-day dosing after achieving complete response or complete response with incomplete blood count recovery.
The groups were well balanced with respect to baseline characteristics and the imbalance in patient numbers in the arms was mainly because of adaptive randomization; the 10-day patients had better responses during the early part of the study, Dr. Short explained.
None of the patients underwent allogeneic stem cell transplantation, he added.
Older patients with acute AML often have poor tolerance for intensive chemotherapy, but decitabine has been shown to improve survival when compared with supportive care or low-dose cytarabine in these patients. However, while some single-arm studies have suggested that 5- and 10-day dosing may result in similar outcomes, and another suggested that 10-day dosing may be superior in TP53-mutated AML, no studies have directly compared 5- and 10-day dosing, he said.
“In this relatively poor-risk cohort of older adults with newly diagnosed AML, decitabine for 5 and 10 days resulted in similar response rates, survival, and early mortality,” he concluded.
This study was funded by a University of Texas MD Anderson Cancer Center support grant. Dr. Short reported having no financial disclosures.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
SOURCE: Short NJ et al. ALF 2018, Poster Session.
NEWPORT BEACH, CALIF. – Response and survival rates are similar with 5- and 10-day dosing of decitabine in poor-risk older adults with previously untreated acute myeloid leukemia, according to findings from a randomized phase 2 trial.
The overall response rates in 25 patients randomized to receive 5 days of decitabine and 40 patients randomized to receive 10 days of decitabine were 44% and 38%, respectively. Complete response rates were 28% and 30%, respectively, and the median number of cycles to best response was two in both arms, Nicholas J. Short, MD, reported in a poster at the Acute Leukemia Forum of Hemedicus.
Patients were followed for a median of 38 months. Median remission duration was 9.4 months in 11 responders who received 5 days of decitabine, and 6.4 months in 15 responders in the 10-day treatment arm. The 1-year continued remission rates were 25% and 30%, respectively (P = .85). Median overall survival was 4.9 and 7.1 months, respectively, and 1-year overall survival rates were 27% and 25%, respectively, he said, adding that none of the differences in the groups were statistically significant.
Further, no differences in survival were seen when patients in the two treatment arms were stratified by cytogenetics, de novo versus secondary or treatment-related disease, or TP53 mutations; however, these subgroups were small, he noted.
Study subjects were adults aged 60 years or older (median, 77 and 78 years) with newly diagnosed and untreated acute myeloid leukemia (AML), and adults under age 60 years who were not suitable candidates for intensive chemotherapy. They were enrolled between February 2013 and July 2017. About 30% of patients had performance status scores of 2-3, and 45% of patients had secondary AML.
“About 40% of patients in the 5-day arm had poor-risk cytogenetics, and about half in the 10-day arm had poor-risk cytogenetics,” Dr. Short said in an interview, adding that 6 of 21 tested patients in the 5-day arm and 16 of 38 tested patients in the 10-day arm had a TP53 mutation.
Patients received intravenous decitabine at a dose of 20 mg/m2 for the assigned duration of either 5 or 10 consecutive days for the first three cycles of induction, and those who responded to treatment received additional consolidation cycles every 4-8 weeks for up to 24 total cycles. Patients in the 10-day arm were switched to 5-day dosing after achieving complete response or complete response with incomplete blood count recovery.
The groups were well balanced with respect to baseline characteristics and the imbalance in patient numbers in the arms was mainly because of adaptive randomization; the 10-day patients had better responses during the early part of the study, Dr. Short explained.
None of the patients underwent allogeneic stem cell transplantation, he added.
Older patients with acute AML often have poor tolerance for intensive chemotherapy, but decitabine has been shown to improve survival when compared with supportive care or low-dose cytarabine in these patients. However, while some single-arm studies have suggested that 5- and 10-day dosing may result in similar outcomes, and another suggested that 10-day dosing may be superior in TP53-mutated AML, no studies have directly compared 5- and 10-day dosing, he said.
“In this relatively poor-risk cohort of older adults with newly diagnosed AML, decitabine for 5 and 10 days resulted in similar response rates, survival, and early mortality,” he concluded.
This study was funded by a University of Texas MD Anderson Cancer Center support grant. Dr. Short reported having no financial disclosures.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
SOURCE: Short NJ et al. ALF 2018, Poster Session.
REPORTING FROM ALF 2018
Key clinical point:
Major finding: Overall response rates were 44% with 5-day dosing and 38% with 10-day dosing.
Study details: A randomized phase 2 study of 65 patients.
Disclosures: This study was funded by an University of Texas MD Anderson Cancer Center support grant. Dr. Short reported having no financial disclosures.
Source: Short NJ et al. ALF 2018, Poster Session.
Statin plus chemo yields encouraging results in poor-risk AML
Patients with poor-risk acute myelogenous leukemia (AML) had promising rates of response and survival following treatment with a statin added to standard chemotherapy, researchers reported.
Pravastatin in combination with idarubicin and cytarabine had a 30% response rate in poor-risk AML patients, according to Anjali S. Advani, MD, of the Taussig Cancer Institute at the Cleveland Clinic, and her coauthors.
Although that response rate did not meet a prespecified threshold for statistical significance, results of the phase 2 study were nevertheless “encouraging” for a group of patients with unfavorable cytogenetics and poor-risk molecular mutations, the researchers wrote. The study was published in Leukemia Research.
The estimated median overall survival was 4.1 months in the phase 2 trial, known as SWOG S0919. About one-quarter of the patients were able to proceed to allogeneic hematopoietic stem cell transplant, and for that group, median overall survival was 27.1 months.
These findings appear to provide further support for the hypothesis that targeting the cholesterol pathway may be a promising approach in patients with AML.
Some previous investigations suggested that AML blasts overexpress genes for lipoprotein receptors and regulatory enzymes, while others showed that these cells import and synthesize cholesterol at levels higher than what is seen in normal progenitor cells. In addition, some AML patients have hypocholesterolemia that usually resolves when they achieve a complete remission.
“These observations suggest that AML cells may require high levels of cholesterol for their survival and that abnormalities in cholesterol homeostasis are necessary for AML cell survival,” the researchers wrote.
This led to a phase 1 trial of pravastatin plus idarubicin/cytarabine, followed by the phase 2 SWOG S0919 trial, which demonstrated a 75% rate of complete response (CR) or complete response with incomplete count recovery (CRi) for the regimen.
The SWOG S0919 study was amended to include the poor-risk AML patients described in the present study. That cohort of 46 patients had a CR/CRi of less than 6 months after their last induction regimen or refractory disease. Many had poor-risk cytogenetics (43%) or one of a number of poor-risk mutations, according to the study report.
Pravastatin, in addition to working on the cholesterol pathway in AML, may also have a therapeutic advantage in patients with FLT3 mutations. Three out of six patients with FLT3 mutations achieved CR/CRi, corroborating earlier preclinical studies and suggesting further study of this specific patient population would be worthwhile, the researchers noted.
The study was supported in part by the National Institutes of Health and Bristol-Myers Squibb. Dr. Advani reported having no financial disclosures but other study authors reported relationships with various pharmaceutical companies.
SOURCE: Advani AS et al. Leuk Res. 2018 Apr;67:17-20.
Patients with poor-risk acute myelogenous leukemia (AML) had promising rates of response and survival following treatment with a statin added to standard chemotherapy, researchers reported.
Pravastatin in combination with idarubicin and cytarabine had a 30% response rate in poor-risk AML patients, according to Anjali S. Advani, MD, of the Taussig Cancer Institute at the Cleveland Clinic, and her coauthors.
Although that response rate did not meet a prespecified threshold for statistical significance, results of the phase 2 study were nevertheless “encouraging” for a group of patients with unfavorable cytogenetics and poor-risk molecular mutations, the researchers wrote. The study was published in Leukemia Research.
The estimated median overall survival was 4.1 months in the phase 2 trial, known as SWOG S0919. About one-quarter of the patients were able to proceed to allogeneic hematopoietic stem cell transplant, and for that group, median overall survival was 27.1 months.
These findings appear to provide further support for the hypothesis that targeting the cholesterol pathway may be a promising approach in patients with AML.
Some previous investigations suggested that AML blasts overexpress genes for lipoprotein receptors and regulatory enzymes, while others showed that these cells import and synthesize cholesterol at levels higher than what is seen in normal progenitor cells. In addition, some AML patients have hypocholesterolemia that usually resolves when they achieve a complete remission.
“These observations suggest that AML cells may require high levels of cholesterol for their survival and that abnormalities in cholesterol homeostasis are necessary for AML cell survival,” the researchers wrote.
This led to a phase 1 trial of pravastatin plus idarubicin/cytarabine, followed by the phase 2 SWOG S0919 trial, which demonstrated a 75% rate of complete response (CR) or complete response with incomplete count recovery (CRi) for the regimen.
The SWOG S0919 study was amended to include the poor-risk AML patients described in the present study. That cohort of 46 patients had a CR/CRi of less than 6 months after their last induction regimen or refractory disease. Many had poor-risk cytogenetics (43%) or one of a number of poor-risk mutations, according to the study report.
Pravastatin, in addition to working on the cholesterol pathway in AML, may also have a therapeutic advantage in patients with FLT3 mutations. Three out of six patients with FLT3 mutations achieved CR/CRi, corroborating earlier preclinical studies and suggesting further study of this specific patient population would be worthwhile, the researchers noted.
The study was supported in part by the National Institutes of Health and Bristol-Myers Squibb. Dr. Advani reported having no financial disclosures but other study authors reported relationships with various pharmaceutical companies.
SOURCE: Advani AS et al. Leuk Res. 2018 Apr;67:17-20.
Patients with poor-risk acute myelogenous leukemia (AML) had promising rates of response and survival following treatment with a statin added to standard chemotherapy, researchers reported.
Pravastatin in combination with idarubicin and cytarabine had a 30% response rate in poor-risk AML patients, according to Anjali S. Advani, MD, of the Taussig Cancer Institute at the Cleveland Clinic, and her coauthors.
Although that response rate did not meet a prespecified threshold for statistical significance, results of the phase 2 study were nevertheless “encouraging” for a group of patients with unfavorable cytogenetics and poor-risk molecular mutations, the researchers wrote. The study was published in Leukemia Research.
The estimated median overall survival was 4.1 months in the phase 2 trial, known as SWOG S0919. About one-quarter of the patients were able to proceed to allogeneic hematopoietic stem cell transplant, and for that group, median overall survival was 27.1 months.
These findings appear to provide further support for the hypothesis that targeting the cholesterol pathway may be a promising approach in patients with AML.
Some previous investigations suggested that AML blasts overexpress genes for lipoprotein receptors and regulatory enzymes, while others showed that these cells import and synthesize cholesterol at levels higher than what is seen in normal progenitor cells. In addition, some AML patients have hypocholesterolemia that usually resolves when they achieve a complete remission.
“These observations suggest that AML cells may require high levels of cholesterol for their survival and that abnormalities in cholesterol homeostasis are necessary for AML cell survival,” the researchers wrote.
This led to a phase 1 trial of pravastatin plus idarubicin/cytarabine, followed by the phase 2 SWOG S0919 trial, which demonstrated a 75% rate of complete response (CR) or complete response with incomplete count recovery (CRi) for the regimen.
The SWOG S0919 study was amended to include the poor-risk AML patients described in the present study. That cohort of 46 patients had a CR/CRi of less than 6 months after their last induction regimen or refractory disease. Many had poor-risk cytogenetics (43%) or one of a number of poor-risk mutations, according to the study report.
Pravastatin, in addition to working on the cholesterol pathway in AML, may also have a therapeutic advantage in patients with FLT3 mutations. Three out of six patients with FLT3 mutations achieved CR/CRi, corroborating earlier preclinical studies and suggesting further study of this specific patient population would be worthwhile, the researchers noted.
The study was supported in part by the National Institutes of Health and Bristol-Myers Squibb. Dr. Advani reported having no financial disclosures but other study authors reported relationships with various pharmaceutical companies.
SOURCE: Advani AS et al. Leuk Res. 2018 Apr;67:17-20.
FROM LEUKEMIA RESEARCH
Key clinical point:
Major finding: Pravastatin in combination with idarubicin and cytarabine had a 30% response rate in poor risk AML patients.
Study details: Results from the poor-risk cohort (n = 46) in SWOG S0919, a phase 2 study.
Disclosures: The research was supported in part by the National Institutes of Health and Bristol-Myers Squibb. Dr. Advani reported having no financial disclosures but other study authors reported relationships with various pharmaceutical companies.
Source: Advani AS et al. Leuk Res. 2018 Apr;67:17-20.
Drug appears to aid chemo in AML
Adding an experimental compound to chemotherapy is a “promising” treatment approach for certain patients with acute myeloid leukemia (AML), according to researchers.
They tested the compound, CPI-613, in combination with high-dose cytarabine and mitoxantrone in a phase 1 trial of patients with relapsed or refractory AML.
The combination produced similar response rates in the overall patient population (50%), patients age 60 and older (47%), and those with poor-risk cytogenetics (46%).
The most common grade 3/4 adverse events (AEs) were hematologic toxicities, and there was 1 fatal AE—hypotension.
Mortality rates in this trial were similar to those observed in historical controls treated with high-dose cytarabine, mitoxantrone, and asparaginase.
“These data are very encouraging, especially for patients 60 years of age or older who have historically done very poorly with this disease,” said Timothy Pardee, MD, PhD, a professor at Wake Forest Baptist Health in Winston-Salem, North Carolina, and chief medical officer of Rafael Pharmaceuticals, Inc., the company developing CPI-613.
Dr Pardee and his colleagues reported these results in Clinical Cancer Research.
The researchers noted that CPI-613 is designed to target mitochondrial metabolism in cancer cells, and preclinical research showed that CPI-613 sensitized AML cells to chemotherapy.
To investigate this further, the team tested CPI-613 in combination with high-dose cytarabine and mitoxantrone in the phase 1 trial. The study included 66 patients with relapsed or refractory AML, as well as a patient with advanced-phase chronic myeloid leukemia (CML) who was mistakenly enrolled.
The patients’ median age was 60 (range, 21-79), and 54% were age 60 and older. Their median percentage of marrow blasts was 43%. Forty percent of patients had poor-risk cytogenetics, and 49% had intermediate-risk cytogenetics.
Most patients (72%) had no prior salvage therapy, 13% had 1 prior line of salvage, 10% had 2 prior lines, and 4% had more than 2. Thirty-one percent of patients had refractory disease.
Seven percent of patients had previously received high-dose cytarabine and mitoxantrone, and 25% had previous salvage including high-dose or intermediate-dose cytarabine.
Treatment
Patients received CPI-613, given over 2 hours, on days 1 to 5 of cycle 1. Doses ranged from 500 mg/m2 to 2750 mg/m2.
Starting on day 3, patients received 5 doses of cytarabine at 3 gm/m2 (for patients younger than 60) or 1.5 gm/m2 (for older patients) in 500 mL normal saline, over 3 hours, every 12 hours.
Patients also received 3 daily doses of mitoxantrone at 6 mg/m2 in 50 mL normal saline, given over 15 minutes, after the first, third, and fifth doses of cytarabine.
Patients were initially assigned to receive 1 cycle of treatment. Those with at least 5% blasts after the first cycle could receive a second course—either a full course or a 3-day course. And patients who responded to the first course could receive up to 2 cycles of the 3-day course.
Safety
There were 2 dose-limiting toxicities when CPI-613 was given at the 2750 mg/m2 dose. One of these toxicities was grade 3 diarrhea that didn’t respond to anti-diarrheals, and the other was grade 3 nausea that didn’t respond to antiemetics.
Because of these events, 2500 mg/m2 was deemed the maximum-tolerated dose. However, the recommended phase 2 dose is 2000 mg/m2.
The most common AEs—occurring in at least 50% of all patients who received CPI-613 (n=67)—included hemoglobin decrease (67%), hyperglycemia (67%), neutropenia (67%), thrombocytopenia (67%), hypomagnesemia (66%), leukopenia (66%), lymphopenia (66%), hypoalbuminemia (65%), hypokalemia (60%), hypocalcemia (57%), and diarrhea (55%).
All cases of neutropenia, thrombocytopenia, leukopenia, and lymphopenia were grade 3/4. Other common grade 3/4 AEs (occurring in at least 20% of patients) included hemoglobin decrease (62%), febrile neutropenia (28%), hypophosphatemia (24%), and hypokalemia (23%).
The only grade 5 AE was hypotension.
The mortality rate was 12% (n=8) at 30 days and 19% (n=13) at 60 days. The researchers said this was similar to the historical experience with high-dose cytarabine, mitoxantrone, and asparaginase. Mortality rates with this regimen were 13% at 30 days and 22% at 60 days.
Efficacy
Sixty-two patients were evaluable for response. Of the 5 patients who were not evaluable, 1 didn’t complete the first cycle of treatment, 1 was the CML patient, and 3 died before assessment.
The overall response rate was 50% (31/62). This included 26 patients with a complete response (CR) and 5 patients who had a CR with incomplete count recovery (CRi).
The rate of CR/CRi was 47% (15/32) in patients older than 60 years of age, 46% (11/24) in patients who had poor-risk cytogenetics, and 53% (8/15) when CPI-613 was given at the recommended phase 2 dose—2000 mg/m2.
The median overall survival (OS) was 6.7 months for all evaluable patients and 13.2 months for patients who achieved a CR/CRi.
The median OS was 6.9 months for patients age 60 and older, which was not significantly different from the median OS in younger patients (P=0.9642).
This study was sponsored by Wake Forest University Health Sciences and the National Cancer Institute.
Adding an experimental compound to chemotherapy is a “promising” treatment approach for certain patients with acute myeloid leukemia (AML), according to researchers.
They tested the compound, CPI-613, in combination with high-dose cytarabine and mitoxantrone in a phase 1 trial of patients with relapsed or refractory AML.
The combination produced similar response rates in the overall patient population (50%), patients age 60 and older (47%), and those with poor-risk cytogenetics (46%).
The most common grade 3/4 adverse events (AEs) were hematologic toxicities, and there was 1 fatal AE—hypotension.
Mortality rates in this trial were similar to those observed in historical controls treated with high-dose cytarabine, mitoxantrone, and asparaginase.
“These data are very encouraging, especially for patients 60 years of age or older who have historically done very poorly with this disease,” said Timothy Pardee, MD, PhD, a professor at Wake Forest Baptist Health in Winston-Salem, North Carolina, and chief medical officer of Rafael Pharmaceuticals, Inc., the company developing CPI-613.
Dr Pardee and his colleagues reported these results in Clinical Cancer Research.
The researchers noted that CPI-613 is designed to target mitochondrial metabolism in cancer cells, and preclinical research showed that CPI-613 sensitized AML cells to chemotherapy.
To investigate this further, the team tested CPI-613 in combination with high-dose cytarabine and mitoxantrone in the phase 1 trial. The study included 66 patients with relapsed or refractory AML, as well as a patient with advanced-phase chronic myeloid leukemia (CML) who was mistakenly enrolled.
The patients’ median age was 60 (range, 21-79), and 54% were age 60 and older. Their median percentage of marrow blasts was 43%. Forty percent of patients had poor-risk cytogenetics, and 49% had intermediate-risk cytogenetics.
Most patients (72%) had no prior salvage therapy, 13% had 1 prior line of salvage, 10% had 2 prior lines, and 4% had more than 2. Thirty-one percent of patients had refractory disease.
Seven percent of patients had previously received high-dose cytarabine and mitoxantrone, and 25% had previous salvage including high-dose or intermediate-dose cytarabine.
Treatment
Patients received CPI-613, given over 2 hours, on days 1 to 5 of cycle 1. Doses ranged from 500 mg/m2 to 2750 mg/m2.
Starting on day 3, patients received 5 doses of cytarabine at 3 gm/m2 (for patients younger than 60) or 1.5 gm/m2 (for older patients) in 500 mL normal saline, over 3 hours, every 12 hours.
Patients also received 3 daily doses of mitoxantrone at 6 mg/m2 in 50 mL normal saline, given over 15 minutes, after the first, third, and fifth doses of cytarabine.
Patients were initially assigned to receive 1 cycle of treatment. Those with at least 5% blasts after the first cycle could receive a second course—either a full course or a 3-day course. And patients who responded to the first course could receive up to 2 cycles of the 3-day course.
Safety
There were 2 dose-limiting toxicities when CPI-613 was given at the 2750 mg/m2 dose. One of these toxicities was grade 3 diarrhea that didn’t respond to anti-diarrheals, and the other was grade 3 nausea that didn’t respond to antiemetics.
Because of these events, 2500 mg/m2 was deemed the maximum-tolerated dose. However, the recommended phase 2 dose is 2000 mg/m2.
The most common AEs—occurring in at least 50% of all patients who received CPI-613 (n=67)—included hemoglobin decrease (67%), hyperglycemia (67%), neutropenia (67%), thrombocytopenia (67%), hypomagnesemia (66%), leukopenia (66%), lymphopenia (66%), hypoalbuminemia (65%), hypokalemia (60%), hypocalcemia (57%), and diarrhea (55%).
All cases of neutropenia, thrombocytopenia, leukopenia, and lymphopenia were grade 3/4. Other common grade 3/4 AEs (occurring in at least 20% of patients) included hemoglobin decrease (62%), febrile neutropenia (28%), hypophosphatemia (24%), and hypokalemia (23%).
The only grade 5 AE was hypotension.
The mortality rate was 12% (n=8) at 30 days and 19% (n=13) at 60 days. The researchers said this was similar to the historical experience with high-dose cytarabine, mitoxantrone, and asparaginase. Mortality rates with this regimen were 13% at 30 days and 22% at 60 days.
Efficacy
Sixty-two patients were evaluable for response. Of the 5 patients who were not evaluable, 1 didn’t complete the first cycle of treatment, 1 was the CML patient, and 3 died before assessment.
The overall response rate was 50% (31/62). This included 26 patients with a complete response (CR) and 5 patients who had a CR with incomplete count recovery (CRi).
The rate of CR/CRi was 47% (15/32) in patients older than 60 years of age, 46% (11/24) in patients who had poor-risk cytogenetics, and 53% (8/15) when CPI-613 was given at the recommended phase 2 dose—2000 mg/m2.
The median overall survival (OS) was 6.7 months for all evaluable patients and 13.2 months for patients who achieved a CR/CRi.
The median OS was 6.9 months for patients age 60 and older, which was not significantly different from the median OS in younger patients (P=0.9642).
This study was sponsored by Wake Forest University Health Sciences and the National Cancer Institute.
Adding an experimental compound to chemotherapy is a “promising” treatment approach for certain patients with acute myeloid leukemia (AML), according to researchers.
They tested the compound, CPI-613, in combination with high-dose cytarabine and mitoxantrone in a phase 1 trial of patients with relapsed or refractory AML.
The combination produced similar response rates in the overall patient population (50%), patients age 60 and older (47%), and those with poor-risk cytogenetics (46%).
The most common grade 3/4 adverse events (AEs) were hematologic toxicities, and there was 1 fatal AE—hypotension.
Mortality rates in this trial were similar to those observed in historical controls treated with high-dose cytarabine, mitoxantrone, and asparaginase.
“These data are very encouraging, especially for patients 60 years of age or older who have historically done very poorly with this disease,” said Timothy Pardee, MD, PhD, a professor at Wake Forest Baptist Health in Winston-Salem, North Carolina, and chief medical officer of Rafael Pharmaceuticals, Inc., the company developing CPI-613.
Dr Pardee and his colleagues reported these results in Clinical Cancer Research.
The researchers noted that CPI-613 is designed to target mitochondrial metabolism in cancer cells, and preclinical research showed that CPI-613 sensitized AML cells to chemotherapy.
To investigate this further, the team tested CPI-613 in combination with high-dose cytarabine and mitoxantrone in the phase 1 trial. The study included 66 patients with relapsed or refractory AML, as well as a patient with advanced-phase chronic myeloid leukemia (CML) who was mistakenly enrolled.
The patients’ median age was 60 (range, 21-79), and 54% were age 60 and older. Their median percentage of marrow blasts was 43%. Forty percent of patients had poor-risk cytogenetics, and 49% had intermediate-risk cytogenetics.
Most patients (72%) had no prior salvage therapy, 13% had 1 prior line of salvage, 10% had 2 prior lines, and 4% had more than 2. Thirty-one percent of patients had refractory disease.
Seven percent of patients had previously received high-dose cytarabine and mitoxantrone, and 25% had previous salvage including high-dose or intermediate-dose cytarabine.
Treatment
Patients received CPI-613, given over 2 hours, on days 1 to 5 of cycle 1. Doses ranged from 500 mg/m2 to 2750 mg/m2.
Starting on day 3, patients received 5 doses of cytarabine at 3 gm/m2 (for patients younger than 60) or 1.5 gm/m2 (for older patients) in 500 mL normal saline, over 3 hours, every 12 hours.
Patients also received 3 daily doses of mitoxantrone at 6 mg/m2 in 50 mL normal saline, given over 15 minutes, after the first, third, and fifth doses of cytarabine.
Patients were initially assigned to receive 1 cycle of treatment. Those with at least 5% blasts after the first cycle could receive a second course—either a full course or a 3-day course. And patients who responded to the first course could receive up to 2 cycles of the 3-day course.
Safety
There were 2 dose-limiting toxicities when CPI-613 was given at the 2750 mg/m2 dose. One of these toxicities was grade 3 diarrhea that didn’t respond to anti-diarrheals, and the other was grade 3 nausea that didn’t respond to antiemetics.
Because of these events, 2500 mg/m2 was deemed the maximum-tolerated dose. However, the recommended phase 2 dose is 2000 mg/m2.
The most common AEs—occurring in at least 50% of all patients who received CPI-613 (n=67)—included hemoglobin decrease (67%), hyperglycemia (67%), neutropenia (67%), thrombocytopenia (67%), hypomagnesemia (66%), leukopenia (66%), lymphopenia (66%), hypoalbuminemia (65%), hypokalemia (60%), hypocalcemia (57%), and diarrhea (55%).
All cases of neutropenia, thrombocytopenia, leukopenia, and lymphopenia were grade 3/4. Other common grade 3/4 AEs (occurring in at least 20% of patients) included hemoglobin decrease (62%), febrile neutropenia (28%), hypophosphatemia (24%), and hypokalemia (23%).
The only grade 5 AE was hypotension.
The mortality rate was 12% (n=8) at 30 days and 19% (n=13) at 60 days. The researchers said this was similar to the historical experience with high-dose cytarabine, mitoxantrone, and asparaginase. Mortality rates with this regimen were 13% at 30 days and 22% at 60 days.
Efficacy
Sixty-two patients were evaluable for response. Of the 5 patients who were not evaluable, 1 didn’t complete the first cycle of treatment, 1 was the CML patient, and 3 died before assessment.
The overall response rate was 50% (31/62). This included 26 patients with a complete response (CR) and 5 patients who had a CR with incomplete count recovery (CRi).
The rate of CR/CRi was 47% (15/32) in patients older than 60 years of age, 46% (11/24) in patients who had poor-risk cytogenetics, and 53% (8/15) when CPI-613 was given at the recommended phase 2 dose—2000 mg/m2.
The median overall survival (OS) was 6.7 months for all evaluable patients and 13.2 months for patients who achieved a CR/CRi.
The median OS was 6.9 months for patients age 60 and older, which was not significantly different from the median OS in younger patients (P=0.9642).
This study was sponsored by Wake Forest University Health Sciences and the National Cancer Institute.
Myelodysplastic syndromes: etiologies, evaluation, and therapy
In this interview, Dr David Henry, MD, the Editor-in-Chief of JCSO, and David Steensma, MD, of the Dana-Farber Cancer Institute in Boston, talk about myelodysplasic syndromes, from diagnosis, evaluation, and etiologies, to therapy options and molecular sequencing.
Listen to the podcast below, or click on the PDF icon at the top of this introduction to read a transcript of the interview.
In this interview, Dr David Henry, MD, the Editor-in-Chief of JCSO, and David Steensma, MD, of the Dana-Farber Cancer Institute in Boston, talk about myelodysplasic syndromes, from diagnosis, evaluation, and etiologies, to therapy options and molecular sequencing.
Listen to the podcast below, or click on the PDF icon at the top of this introduction to read a transcript of the interview.
In this interview, Dr David Henry, MD, the Editor-in-Chief of JCSO, and David Steensma, MD, of the Dana-Farber Cancer Institute in Boston, talk about myelodysplasic syndromes, from diagnosis, evaluation, and etiologies, to therapy options and molecular sequencing.
Listen to the podcast below, or click on the PDF icon at the top of this introduction to read a transcript of the interview.