User login
NSCLC: Meta-analysis cautions concomitant use of gastric acid suppressants and immunotherapy
Key clinical point: The concomitant use of gastric acid suppressants (GAS) may be associated with poor survival outcomes in patients with non—small-cell lung cancer (NSCLC) receiving programmed death-1/ligand-1 (PD-1/PD-L1) inhibitors.
Major finding: Use of PD-1/PD-L1 inhibitors with vs without GAS worsened progression-free survival by 32% (hazard ratio [HR] 1.32; P < .001) and overall survival by 36% (HR 1.36; P < .001).
Study details: The data come from a meta-analysis of 10 retrospective studies and 1 prospective cohort study including 5892 patients with NSCLC who were receiving PD-1/PD-L1 inhibitors.
Disclosures: This study was supported by Anhui University Natural Science Research Project, China. The authors declared no conflicts of interest.
Source: Wang M et al. Influence of concomitant gastric acid suppressants use on the survival of patients with non-small cell lung cancer treated with programmed death-1/ligand-1 inhibitors: A meta-analysis. Int Immunopharmacol. 2022;110:108955 (Jun 21). Doi: 10.1016/j.intimp.2022.108955
Key clinical point: The concomitant use of gastric acid suppressants (GAS) may be associated with poor survival outcomes in patients with non—small-cell lung cancer (NSCLC) receiving programmed death-1/ligand-1 (PD-1/PD-L1) inhibitors.
Major finding: Use of PD-1/PD-L1 inhibitors with vs without GAS worsened progression-free survival by 32% (hazard ratio [HR] 1.32; P < .001) and overall survival by 36% (HR 1.36; P < .001).
Study details: The data come from a meta-analysis of 10 retrospective studies and 1 prospective cohort study including 5892 patients with NSCLC who were receiving PD-1/PD-L1 inhibitors.
Disclosures: This study was supported by Anhui University Natural Science Research Project, China. The authors declared no conflicts of interest.
Source: Wang M et al. Influence of concomitant gastric acid suppressants use on the survival of patients with non-small cell lung cancer treated with programmed death-1/ligand-1 inhibitors: A meta-analysis. Int Immunopharmacol. 2022;110:108955 (Jun 21). Doi: 10.1016/j.intimp.2022.108955
Key clinical point: The concomitant use of gastric acid suppressants (GAS) may be associated with poor survival outcomes in patients with non—small-cell lung cancer (NSCLC) receiving programmed death-1/ligand-1 (PD-1/PD-L1) inhibitors.
Major finding: Use of PD-1/PD-L1 inhibitors with vs without GAS worsened progression-free survival by 32% (hazard ratio [HR] 1.32; P < .001) and overall survival by 36% (HR 1.36; P < .001).
Study details: The data come from a meta-analysis of 10 retrospective studies and 1 prospective cohort study including 5892 patients with NSCLC who were receiving PD-1/PD-L1 inhibitors.
Disclosures: This study was supported by Anhui University Natural Science Research Project, China. The authors declared no conflicts of interest.
Source: Wang M et al. Influence of concomitant gastric acid suppressants use on the survival of patients with non-small cell lung cancer treated with programmed death-1/ligand-1 inhibitors: A meta-analysis. Int Immunopharmacol. 2022;110:108955 (Jun 21). Doi: 10.1016/j.intimp.2022.108955
Additional postoperative radiotherapy prolongs survival in stage I-IIA SCLC
Key clinical point: The addition of postoperative radiotherapy (PORT) to surgery and adjuvant chemotherapy prolonged overall survival by 39% and cancer-specific survival by 53% in patients with early small cell lung cancer (SCLC).
Major finding: PORT vs no PORT was associated with a significantly extended median overall survival (8.58 vs 5.17 years; hazard ratio [HR] 0.61; P = .032) and cancer-specific survival (11.33 vs 8.08 years; HR 0.47; P = .0086).
Study details: The data come from a population-based retrospective cohort study involving 278 patients with stage I-IIA SCLC who underwent surgery and received adjuvant chemotherapy.
Disclosures: This study was funded by the National Natural Science Foundation of China and others. The authors declared no conflicts of interest.
Source: Li J et al. Additional postoperative radiotherapy prolonged the survival of patients with i-iia small cell lung cancer: Analysis of the SEER database. J Oncol. 2022;6280538 (Jun 18). Doi: 10.1155/2022/6280538
Key clinical point: The addition of postoperative radiotherapy (PORT) to surgery and adjuvant chemotherapy prolonged overall survival by 39% and cancer-specific survival by 53% in patients with early small cell lung cancer (SCLC).
Major finding: PORT vs no PORT was associated with a significantly extended median overall survival (8.58 vs 5.17 years; hazard ratio [HR] 0.61; P = .032) and cancer-specific survival (11.33 vs 8.08 years; HR 0.47; P = .0086).
Study details: The data come from a population-based retrospective cohort study involving 278 patients with stage I-IIA SCLC who underwent surgery and received adjuvant chemotherapy.
Disclosures: This study was funded by the National Natural Science Foundation of China and others. The authors declared no conflicts of interest.
Source: Li J et al. Additional postoperative radiotherapy prolonged the survival of patients with i-iia small cell lung cancer: Analysis of the SEER database. J Oncol. 2022;6280538 (Jun 18). Doi: 10.1155/2022/6280538
Key clinical point: The addition of postoperative radiotherapy (PORT) to surgery and adjuvant chemotherapy prolonged overall survival by 39% and cancer-specific survival by 53% in patients with early small cell lung cancer (SCLC).
Major finding: PORT vs no PORT was associated with a significantly extended median overall survival (8.58 vs 5.17 years; hazard ratio [HR] 0.61; P = .032) and cancer-specific survival (11.33 vs 8.08 years; HR 0.47; P = .0086).
Study details: The data come from a population-based retrospective cohort study involving 278 patients with stage I-IIA SCLC who underwent surgery and received adjuvant chemotherapy.
Disclosures: This study was funded by the National Natural Science Foundation of China and others. The authors declared no conflicts of interest.
Source: Li J et al. Additional postoperative radiotherapy prolonged the survival of patients with i-iia small cell lung cancer: Analysis of the SEER database. J Oncol. 2022;6280538 (Jun 18). Doi: 10.1155/2022/6280538
Stage III NSCLC: Preoperative FDG PET/CT tied to longer survival
Key clinical point: Use of preoperative fluorine 18-fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG PET/CT) is linked to a longer overall survival in patients with stage IIIA or IIIB resectable non—small-cell lung cancer (NSCLC).
Major finding: Among patients who underwent vs did not undergo preoperative 18F-FDG PET/CT, those with stage IIIB and stage IIIA disease had 20% (adjusted hazard ratio [aHR] 0.80; 95% CI 0.71-0.90) and 10% (aHR 0.90; 95% CI 0.79-0.94) lower risks for mortality, respectively. Patients with stage I-II disease did not have a lower risk for mortality (aHR 1.19; 95% CI 0.89-1.30).
Study details: The data come from a Taiwanese retrospective cohort study of patients with resectable stage I-IIIB NSCLC who underwent (n = 6,754) and did not undergo (n = 6,754) preoperative 18F-FDG PET/CT.
Disclosures: This study was funded by the Lo-Hsu Medical Foundation and Lotung Poh-Ai Hospital, Taiwan. SY Wu is a member of the Taiwan Radiological Society and Taiwan Society for Therapeutic Radiology and Oncology. The other authors reported no relevant relationships.
Source: Chen W-M et al. Use of preoperative FDG PET/CT and survival of patients with resectable non–small cell lung cancer. Radiology. 2022 (Jun 21). Doi: 10.1148/radiol.212798
Key clinical point: Use of preoperative fluorine 18-fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG PET/CT) is linked to a longer overall survival in patients with stage IIIA or IIIB resectable non—small-cell lung cancer (NSCLC).
Major finding: Among patients who underwent vs did not undergo preoperative 18F-FDG PET/CT, those with stage IIIB and stage IIIA disease had 20% (adjusted hazard ratio [aHR] 0.80; 95% CI 0.71-0.90) and 10% (aHR 0.90; 95% CI 0.79-0.94) lower risks for mortality, respectively. Patients with stage I-II disease did not have a lower risk for mortality (aHR 1.19; 95% CI 0.89-1.30).
Study details: The data come from a Taiwanese retrospective cohort study of patients with resectable stage I-IIIB NSCLC who underwent (n = 6,754) and did not undergo (n = 6,754) preoperative 18F-FDG PET/CT.
Disclosures: This study was funded by the Lo-Hsu Medical Foundation and Lotung Poh-Ai Hospital, Taiwan. SY Wu is a member of the Taiwan Radiological Society and Taiwan Society for Therapeutic Radiology and Oncology. The other authors reported no relevant relationships.
Source: Chen W-M et al. Use of preoperative FDG PET/CT and survival of patients with resectable non–small cell lung cancer. Radiology. 2022 (Jun 21). Doi: 10.1148/radiol.212798
Key clinical point: Use of preoperative fluorine 18-fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG PET/CT) is linked to a longer overall survival in patients with stage IIIA or IIIB resectable non—small-cell lung cancer (NSCLC).
Major finding: Among patients who underwent vs did not undergo preoperative 18F-FDG PET/CT, those with stage IIIB and stage IIIA disease had 20% (adjusted hazard ratio [aHR] 0.80; 95% CI 0.71-0.90) and 10% (aHR 0.90; 95% CI 0.79-0.94) lower risks for mortality, respectively. Patients with stage I-II disease did not have a lower risk for mortality (aHR 1.19; 95% CI 0.89-1.30).
Study details: The data come from a Taiwanese retrospective cohort study of patients with resectable stage I-IIIB NSCLC who underwent (n = 6,754) and did not undergo (n = 6,754) preoperative 18F-FDG PET/CT.
Disclosures: This study was funded by the Lo-Hsu Medical Foundation and Lotung Poh-Ai Hospital, Taiwan. SY Wu is a member of the Taiwan Radiological Society and Taiwan Society for Therapeutic Radiology and Oncology. The other authors reported no relevant relationships.
Source: Chen W-M et al. Use of preoperative FDG PET/CT and survival of patients with resectable non–small cell lung cancer. Radiology. 2022 (Jun 21). Doi: 10.1148/radiol.212798
NSCLC: Cell-free RNA beneficial for early detection and prognosi
Key clinical point: Circulating cell-free RNA (cfRNA) of the candidate transcript MORF4L2 demonstrates very good sensitivity and specificity for distinguishing patients with non—small-cell lung cancer (NSCLC) from healthy individuals.
Major finding: A greater concentration of MORF4L2 cfRNA was seen in patients with NSCLC compared with healthy donors (P < .0001). A cutoff value of 537.5 copies/mL of plasma was useful in distinguishing patients with NSCLC from healthy donors with very good sensitivity (0.73; 95% CI 0.61-0.82) and specificity (0.87; 95% CI 0.73-0.96). Low vs high levels of MORF4L2 cfRNA at baseline were associated with a better overall survival (hazard ratio 0.25; P = .009).
Study details: The data come from a cohort study of 41 patients with stage IV NSCLC, 38 patients with early-stage (stage I-III) NSCLC, and 39 healthy blood donors.
Disclosures: This study was supported by the European Transcan-2 project CEVIR (Cancer EVolution and Identification of Relapse-initiating cells) and the German Cancer Consortium. M Metzenmacher, JT Siveke, and M Schuler reported ties with ≥1 pharmaceutical companies. The other authors reported no conflicts of interest.
Source: Metzenmacher M et al. The clinical utility of cfRNA for disease detection and surveillance: A proof of concept study in non-small cell lung cancer. Thorac Cancer. 2022 (Jun 16). Doi: 10.1111/1759-7714.14540
Key clinical point: Circulating cell-free RNA (cfRNA) of the candidate transcript MORF4L2 demonstrates very good sensitivity and specificity for distinguishing patients with non—small-cell lung cancer (NSCLC) from healthy individuals.
Major finding: A greater concentration of MORF4L2 cfRNA was seen in patients with NSCLC compared with healthy donors (P < .0001). A cutoff value of 537.5 copies/mL of plasma was useful in distinguishing patients with NSCLC from healthy donors with very good sensitivity (0.73; 95% CI 0.61-0.82) and specificity (0.87; 95% CI 0.73-0.96). Low vs high levels of MORF4L2 cfRNA at baseline were associated with a better overall survival (hazard ratio 0.25; P = .009).
Study details: The data come from a cohort study of 41 patients with stage IV NSCLC, 38 patients with early-stage (stage I-III) NSCLC, and 39 healthy blood donors.
Disclosures: This study was supported by the European Transcan-2 project CEVIR (Cancer EVolution and Identification of Relapse-initiating cells) and the German Cancer Consortium. M Metzenmacher, JT Siveke, and M Schuler reported ties with ≥1 pharmaceutical companies. The other authors reported no conflicts of interest.
Source: Metzenmacher M et al. The clinical utility of cfRNA for disease detection and surveillance: A proof of concept study in non-small cell lung cancer. Thorac Cancer. 2022 (Jun 16). Doi: 10.1111/1759-7714.14540
Key clinical point: Circulating cell-free RNA (cfRNA) of the candidate transcript MORF4L2 demonstrates very good sensitivity and specificity for distinguishing patients with non—small-cell lung cancer (NSCLC) from healthy individuals.
Major finding: A greater concentration of MORF4L2 cfRNA was seen in patients with NSCLC compared with healthy donors (P < .0001). A cutoff value of 537.5 copies/mL of plasma was useful in distinguishing patients with NSCLC from healthy donors with very good sensitivity (0.73; 95% CI 0.61-0.82) and specificity (0.87; 95% CI 0.73-0.96). Low vs high levels of MORF4L2 cfRNA at baseline were associated with a better overall survival (hazard ratio 0.25; P = .009).
Study details: The data come from a cohort study of 41 patients with stage IV NSCLC, 38 patients with early-stage (stage I-III) NSCLC, and 39 healthy blood donors.
Disclosures: This study was supported by the European Transcan-2 project CEVIR (Cancer EVolution and Identification of Relapse-initiating cells) and the German Cancer Consortium. M Metzenmacher, JT Siveke, and M Schuler reported ties with ≥1 pharmaceutical companies. The other authors reported no conflicts of interest.
Source: Metzenmacher M et al. The clinical utility of cfRNA for disease detection and surveillance: A proof of concept study in non-small cell lung cancer. Thorac Cancer. 2022 (Jun 16). Doi: 10.1111/1759-7714.14540
Early-stage NSCLC: EGFR mutation predicts longer survival
Key clinical point: The presence of epidermal growth factor receptor (EGFR) mutation is associated with a longer median overall survival (OS) compared with absence of the EGFR mutation in patients with early-stage (stage I-IIIA) non—small-cell lung cancer (NSCLC).
Major finding: The presence vs absence of EGFR mutation was associated with a longer median OS (5.7 vs 4.4 years). The lower risk for all-cause mortality was consistent across all subgroups (stage at diagnosis, age, sex, comorbidity, and surgery receipt), with hazard ratios ranging from 0.48 to 0.83.
Study details: The data come from a Danish population-based cohort study involving 21,282 patients with NSCLC.
Disclosures: This study was funded by AstraZeneca. A Taylor and L Servidio reported being current or former employees of AstraZeneca. V Ehrenstein and K Eriksen are employees of Aarhus University or Aarhus University Hospital. E Jakobsen is an employee of Odense University Hospital.
Source: Ehrenstein V et al. Characteristics and overall survival of patients with early-stage non-small cell lung cancer: A cohort study in Denmark. Cancer Med. 2022 (Jun 20). Doi: 10.1002/cam4.4946
Key clinical point: The presence of epidermal growth factor receptor (EGFR) mutation is associated with a longer median overall survival (OS) compared with absence of the EGFR mutation in patients with early-stage (stage I-IIIA) non—small-cell lung cancer (NSCLC).
Major finding: The presence vs absence of EGFR mutation was associated with a longer median OS (5.7 vs 4.4 years). The lower risk for all-cause mortality was consistent across all subgroups (stage at diagnosis, age, sex, comorbidity, and surgery receipt), with hazard ratios ranging from 0.48 to 0.83.
Study details: The data come from a Danish population-based cohort study involving 21,282 patients with NSCLC.
Disclosures: This study was funded by AstraZeneca. A Taylor and L Servidio reported being current or former employees of AstraZeneca. V Ehrenstein and K Eriksen are employees of Aarhus University or Aarhus University Hospital. E Jakobsen is an employee of Odense University Hospital.
Source: Ehrenstein V et al. Characteristics and overall survival of patients with early-stage non-small cell lung cancer: A cohort study in Denmark. Cancer Med. 2022 (Jun 20). Doi: 10.1002/cam4.4946
Key clinical point: The presence of epidermal growth factor receptor (EGFR) mutation is associated with a longer median overall survival (OS) compared with absence of the EGFR mutation in patients with early-stage (stage I-IIIA) non—small-cell lung cancer (NSCLC).
Major finding: The presence vs absence of EGFR mutation was associated with a longer median OS (5.7 vs 4.4 years). The lower risk for all-cause mortality was consistent across all subgroups (stage at diagnosis, age, sex, comorbidity, and surgery receipt), with hazard ratios ranging from 0.48 to 0.83.
Study details: The data come from a Danish population-based cohort study involving 21,282 patients with NSCLC.
Disclosures: This study was funded by AstraZeneca. A Taylor and L Servidio reported being current or former employees of AstraZeneca. V Ehrenstein and K Eriksen are employees of Aarhus University or Aarhus University Hospital. E Jakobsen is an employee of Odense University Hospital.
Source: Ehrenstein V et al. Characteristics and overall survival of patients with early-stage non-small cell lung cancer: A cohort study in Denmark. Cancer Med. 2022 (Jun 20). Doi: 10.1002/cam4.4946
Lung cancer treatment combo may be effective after ICI failure
In a phase 2 clinical trial, the combination of an immune checkpoint inhibitor (ICI) and a vascular endothelial growth factor (VEGF) inhibitor led to improved overall survival versus standard of care in patients with non–small cell lung cancer (NSCLC) who had failed previous ICI therapy.
NSCLC patients usually receive immune checkpoint inhibitor therapy at some point, whether in the adjuvant or neoadjuvant setting, or among stage 3 patients after radiation. “The majority of patients who get diagnosed with lung cancer will get some sort of immunotherapy, and we know that at least from the advanced setting, about 15% of those will have long-term responses, which means the majority of patients will develop tumor resistance to immune checkpoint inhibitor therapy,” said Karen L. Reckamp, MD, who is the lead author of the study published online in Journal of Clinical Oncology.
That clinical need has led to the combination of ICIs with VEGF inhibitors. This approach is approved for first-line therapy of renal cell cancer, endometrial, and hepatocellular cancer. Along with its effect on tumor vasculature, VEGF inhibition assists in the activation and maturation of dendritic cells, as well as to attract cytotoxic T cells to the tumor. “By both changing the vasculature and changing the tumor milieu, there’s a potential to overcome that immune suppression and potentially overcome that (ICI) resistance,” said Dr. Reckamp, who is associate director of clinical research at Cedars Sinai Medical Center, Los Angeles. “The results of the study were encouraging. . We would like to confirm this finding in a phase 3 trial and potentially provide to patients an option that does not include chemotherapy and can potentially overcome resistance to their prior immune checkpoint inhibitor therapy,” Dr. Reckamp said.
The study included 136 patients. The median patient age was 66 years and 61% were male. The ICI/VEGF arm had better overall survival (hazard ratio, 0.69; SLR one-sided P = .05). The median overall survival was 14.5 months in the ICI/VEGF arm, versus 11.6 months in the standard care arm. Both arms had similar response rates, and grade 3 or higher treatment-related adverse events were more common in the chemotherapy arm (60% versus 42%).
The next step is a phase 3 trial and Dr. Reckamp hopes to improve patient selection for VEGF inhibitor and VEGF receptor inhibitor therapy. “The precision medicine that’s associated with other tumor alterations has kind of been elusive for VEGF therapies, but I would hope with potentially a larger trial and understanding of some of the biomarkers that we might find a more select patient population who will benefit the most,” Dr. Reckamp said.
She also noted that the comparative arm in the phase 2 study was a combination of docetaxel and ramucirumab. “That combination has shown to be more effective than single agent docetaxel alone so [the new study] was really improved overall survival over the best standard of care therapy we have,” Dr. Reckamp said.
The study was funded, in part, by Eli Lilly and Company and Merck Sharp & Dohme Corp. Dr. Reckamp disclosed ties to Amgen, Tesaro, Takeda, AstraZeneca, Seattle Genetics, Genentech, Blueprint Medicines, Daiichi Sankyo/Lilly, EMD Serono, Janssen Oncology, Merck KGaA, GlaxoSmithKline, and Mirati Therapeutics.
In a phase 2 clinical trial, the combination of an immune checkpoint inhibitor (ICI) and a vascular endothelial growth factor (VEGF) inhibitor led to improved overall survival versus standard of care in patients with non–small cell lung cancer (NSCLC) who had failed previous ICI therapy.
NSCLC patients usually receive immune checkpoint inhibitor therapy at some point, whether in the adjuvant or neoadjuvant setting, or among stage 3 patients after radiation. “The majority of patients who get diagnosed with lung cancer will get some sort of immunotherapy, and we know that at least from the advanced setting, about 15% of those will have long-term responses, which means the majority of patients will develop tumor resistance to immune checkpoint inhibitor therapy,” said Karen L. Reckamp, MD, who is the lead author of the study published online in Journal of Clinical Oncology.
That clinical need has led to the combination of ICIs with VEGF inhibitors. This approach is approved for first-line therapy of renal cell cancer, endometrial, and hepatocellular cancer. Along with its effect on tumor vasculature, VEGF inhibition assists in the activation and maturation of dendritic cells, as well as to attract cytotoxic T cells to the tumor. “By both changing the vasculature and changing the tumor milieu, there’s a potential to overcome that immune suppression and potentially overcome that (ICI) resistance,” said Dr. Reckamp, who is associate director of clinical research at Cedars Sinai Medical Center, Los Angeles. “The results of the study were encouraging. . We would like to confirm this finding in a phase 3 trial and potentially provide to patients an option that does not include chemotherapy and can potentially overcome resistance to their prior immune checkpoint inhibitor therapy,” Dr. Reckamp said.
The study included 136 patients. The median patient age was 66 years and 61% were male. The ICI/VEGF arm had better overall survival (hazard ratio, 0.69; SLR one-sided P = .05). The median overall survival was 14.5 months in the ICI/VEGF arm, versus 11.6 months in the standard care arm. Both arms had similar response rates, and grade 3 or higher treatment-related adverse events were more common in the chemotherapy arm (60% versus 42%).
The next step is a phase 3 trial and Dr. Reckamp hopes to improve patient selection for VEGF inhibitor and VEGF receptor inhibitor therapy. “The precision medicine that’s associated with other tumor alterations has kind of been elusive for VEGF therapies, but I would hope with potentially a larger trial and understanding of some of the biomarkers that we might find a more select patient population who will benefit the most,” Dr. Reckamp said.
She also noted that the comparative arm in the phase 2 study was a combination of docetaxel and ramucirumab. “That combination has shown to be more effective than single agent docetaxel alone so [the new study] was really improved overall survival over the best standard of care therapy we have,” Dr. Reckamp said.
The study was funded, in part, by Eli Lilly and Company and Merck Sharp & Dohme Corp. Dr. Reckamp disclosed ties to Amgen, Tesaro, Takeda, AstraZeneca, Seattle Genetics, Genentech, Blueprint Medicines, Daiichi Sankyo/Lilly, EMD Serono, Janssen Oncology, Merck KGaA, GlaxoSmithKline, and Mirati Therapeutics.
In a phase 2 clinical trial, the combination of an immune checkpoint inhibitor (ICI) and a vascular endothelial growth factor (VEGF) inhibitor led to improved overall survival versus standard of care in patients with non–small cell lung cancer (NSCLC) who had failed previous ICI therapy.
NSCLC patients usually receive immune checkpoint inhibitor therapy at some point, whether in the adjuvant or neoadjuvant setting, or among stage 3 patients after radiation. “The majority of patients who get diagnosed with lung cancer will get some sort of immunotherapy, and we know that at least from the advanced setting, about 15% of those will have long-term responses, which means the majority of patients will develop tumor resistance to immune checkpoint inhibitor therapy,” said Karen L. Reckamp, MD, who is the lead author of the study published online in Journal of Clinical Oncology.
That clinical need has led to the combination of ICIs with VEGF inhibitors. This approach is approved for first-line therapy of renal cell cancer, endometrial, and hepatocellular cancer. Along with its effect on tumor vasculature, VEGF inhibition assists in the activation and maturation of dendritic cells, as well as to attract cytotoxic T cells to the tumor. “By both changing the vasculature and changing the tumor milieu, there’s a potential to overcome that immune suppression and potentially overcome that (ICI) resistance,” said Dr. Reckamp, who is associate director of clinical research at Cedars Sinai Medical Center, Los Angeles. “The results of the study were encouraging. . We would like to confirm this finding in a phase 3 trial and potentially provide to patients an option that does not include chemotherapy and can potentially overcome resistance to their prior immune checkpoint inhibitor therapy,” Dr. Reckamp said.
The study included 136 patients. The median patient age was 66 years and 61% were male. The ICI/VEGF arm had better overall survival (hazard ratio, 0.69; SLR one-sided P = .05). The median overall survival was 14.5 months in the ICI/VEGF arm, versus 11.6 months in the standard care arm. Both arms had similar response rates, and grade 3 or higher treatment-related adverse events were more common in the chemotherapy arm (60% versus 42%).
The next step is a phase 3 trial and Dr. Reckamp hopes to improve patient selection for VEGF inhibitor and VEGF receptor inhibitor therapy. “The precision medicine that’s associated with other tumor alterations has kind of been elusive for VEGF therapies, but I would hope with potentially a larger trial and understanding of some of the biomarkers that we might find a more select patient population who will benefit the most,” Dr. Reckamp said.
She also noted that the comparative arm in the phase 2 study was a combination of docetaxel and ramucirumab. “That combination has shown to be more effective than single agent docetaxel alone so [the new study] was really improved overall survival over the best standard of care therapy we have,” Dr. Reckamp said.
The study was funded, in part, by Eli Lilly and Company and Merck Sharp & Dohme Corp. Dr. Reckamp disclosed ties to Amgen, Tesaro, Takeda, AstraZeneca, Seattle Genetics, Genentech, Blueprint Medicines, Daiichi Sankyo/Lilly, EMD Serono, Janssen Oncology, Merck KGaA, GlaxoSmithKline, and Mirati Therapeutics.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
The shifting sands of lung cancer screening
An analysis of trends in lung cancer screening since March 2021 when the U.S. Preventive Services Task Force (USPSTF) expanded the eligibility criteria for lung cancer screening, shows that significantly more Black men have been screened for lung cancer, but not women or undereducated people.
The eligibility for lung cancer screening was expanded in 2021 to include men and women under 50 years old and people who smoke at least one pack of cigarettes a day for the last 20 years. “
“Expansion of screening criteria is a critical first step to achieving equity in lung cancer screening for all high-risk populations, but myriad challenges remain before individuals enter the door for screening,” wrote the authors, led by Julie A. Barta, MD, Thomas Jefferson University, Philadelphia. “Health policy changes must occur simultaneously with efforts to expand community outreach, overcome logistical barriers, and facilitate screening adherence. Only after comprehensive strategies to dismantle screening barriers are identified, validated, and implemented can there be a truly equitable landscape for lung cancer screening.”
For the study, published in JAMA Open Network, researchers examined rates of centralized lung cancer screening in the Baltimore area. In addition to expanding lung cancer screening generally, there was hope that the expanded criteria might increase uptake of screening in populations that are traditionally underserved, such as African American, Hispanic, and female patients. Of 815 people screened during the study period (March-December 2021), 161 were newly eligible for screening under the 2021 criteria.
“There’s been quite a bit of work in the field demonstrating that Black men and women develop lung cancer at more advanced stages of disease, and they often are diagnosed at younger ages and have fewer pack-years of smoking. So the hypothesis was that this would reduce some of the disparities seen in lung cancer screening by making more people eligible,” Dr. Barta said in an interview.
The researchers categorized participants as those who would have been eligible for screening under the USPSTF 2013 guideline (age 55 or older, 30 or more pack-years, quit within the past 15 years), and those who would be eligible under the 2021 guideline (age 50 or older, 20 or more pack-years, quit within the past 15 years). Of the 2021 cohort, 54.5% were African American, versus 39.5% of the 2013 cohort (P = .002). There were no differences between the cohorts with respect to education level or gender.
“Although we’ve seen some encouraging improvement in terms of getting more eligible patients into our screening program, there’s still a lot of work to be done in the field,” Dr. Barta said. “Diagnosing lung cancer at earlier stages of disease is more cost effective in general for the health care system than fighting lung cancer at advanced stages, which requires more complex and multimodal and prolonged therapies.”
New evidence: Chest CTs for lung cancer screening reduces incidence of advanced lung cancer
In an analysis of the SEER database presented in June at the annual meeting of the American Society of Clinical Oncology, the adoption of low-dose chest computed tomography (LDCT) led to fewer diagnoses of advanced lung cancer, although these declines varied significantly by race and ethnicity. Non-Hispanic Blacks seemed to benefit the most with a 55% decline (P < .01), while Hispanics had the lowest rate of decline at 41% (P < .01). The change was recommended by USPSTF in 2013 after the National Lung Screening Trial revealed a 20% relative reduction in mortality when CT scans were used instead of chest radiography. The Centers for Medicare and Medicaid Services approved coverage of the screen in 2015.
The SEER study looked at data from 400,343 individuals from 2004-2014 (preintervention) and 2015-2018 (postintervention). The age-adjusted incidence of advanced lung cancer declined during both periods, but the decline was sharper between 2015 and 2018, with three fewer cases per 100,000 people than 2004-2014 (P < .01). Similar patterns were seen in subanalyses of males and females, non-Hispanic Whites, non-Hispanic Blacks, and Hispanics. The relative declines were largest in women, non-Hispanic Blacks, and people who lived outside of Metropolitan areas.
During a Q&A session that followed the presentation, Robert Smith, PhD, pointed out that the bar for eligibility of lung cancer risk has been set quite high, following the eligibility criteria for clinical trials. He noted that . “We are missing opportunities to prevent avertable lung cancer deaths,” said Dr. Smith, senior vice president of cancer screening at the American Cancer Society.
On the other hand, screening-prompted biopsies have the potential to cause harm, particularly in patients who already have lung disease, said Douglas Allen Arenberg, MD, professor at the University of Michigan, Ann Arbor. “I think that’s what scares most people is the potential downside, which is very hard to measure outside of a clinical trial,” said Dr. Arenberg, who served as a discussant for the presentation.
One way to reduce that risk is to identify biomarkers, either for screens or for incidentally-detected nodules, that have good negative predictive value. “If I had a blood test that is as good as a negative PET scan, I’m going to be much more likely to say, ‘Yeah, you’re 40 and your grandfather had lung cancer. Maybe you should get a CT. If we had that, we could screen a lot more people. Right now, I would discourage anybody who is at low risk from getting screened because when they come to me, the biggest opportunity I have to do harm is when I do a biopsy, and you always remember the ones that go wrong,” he said.
Dr. Arenberg also called for improvements in electronic medical records to better flag at-risk patients. “I think we as physicians have to demand more of the software developers that create these EMRs for us,” he said.
Another study in the same session used data from 1,391,088 patients drawn from the National Cancer Database between 2010 and 2017 to examine trends in diagnosis of stage I cancer. In 2010, 23.5% of patients were diagnosed as stage I, versus 29.1% in 2017. Stage I incidence increased from 25.8% to 31.7% in non–small cell lung cancer, but there was no statistically significant change in small cell lung cancer. As with the SEER database study, the researchers noted that the shift toward stage I diagnoses predated the recommendation of LDCT.
Dr. Arenberg suggested that the trend may come down to increased frequency of CT scans, which often collect incidental images of the lungs. He added that better access to care may also be helping to drive the change. “How much of that might have had something to do with the introduction 5 or 10 years earlier of the Affordable Care Act and people just simply having access to care and taking advantage of that?” Dr. Arenberg said.
But Dr. Arenberg said that not even screening can explain all the data. He referenced a stage shift in patients of all age groups in the National Cancer Database study, even those too young to be eligible for screening. “There’s something else going on here. It would be nice for us to understand what caused these trends, so perhaps we could accentuate that trend even more, but stage shifts are clearly occurring in lung cancer,” Dr. Arenberg said.
Dr. Barta has received grants from Genentech Health Equity Innovations Fund. Dr. Arenberg has no relevant financial disclosures. Dr. Smith’s potential disclosures could not be ascertained.
An analysis of trends in lung cancer screening since March 2021 when the U.S. Preventive Services Task Force (USPSTF) expanded the eligibility criteria for lung cancer screening, shows that significantly more Black men have been screened for lung cancer, but not women or undereducated people.
The eligibility for lung cancer screening was expanded in 2021 to include men and women under 50 years old and people who smoke at least one pack of cigarettes a day for the last 20 years. “
“Expansion of screening criteria is a critical first step to achieving equity in lung cancer screening for all high-risk populations, but myriad challenges remain before individuals enter the door for screening,” wrote the authors, led by Julie A. Barta, MD, Thomas Jefferson University, Philadelphia. “Health policy changes must occur simultaneously with efforts to expand community outreach, overcome logistical barriers, and facilitate screening adherence. Only after comprehensive strategies to dismantle screening barriers are identified, validated, and implemented can there be a truly equitable landscape for lung cancer screening.”
For the study, published in JAMA Open Network, researchers examined rates of centralized lung cancer screening in the Baltimore area. In addition to expanding lung cancer screening generally, there was hope that the expanded criteria might increase uptake of screening in populations that are traditionally underserved, such as African American, Hispanic, and female patients. Of 815 people screened during the study period (March-December 2021), 161 were newly eligible for screening under the 2021 criteria.
“There’s been quite a bit of work in the field demonstrating that Black men and women develop lung cancer at more advanced stages of disease, and they often are diagnosed at younger ages and have fewer pack-years of smoking. So the hypothesis was that this would reduce some of the disparities seen in lung cancer screening by making more people eligible,” Dr. Barta said in an interview.
The researchers categorized participants as those who would have been eligible for screening under the USPSTF 2013 guideline (age 55 or older, 30 or more pack-years, quit within the past 15 years), and those who would be eligible under the 2021 guideline (age 50 or older, 20 or more pack-years, quit within the past 15 years). Of the 2021 cohort, 54.5% were African American, versus 39.5% of the 2013 cohort (P = .002). There were no differences between the cohorts with respect to education level or gender.
“Although we’ve seen some encouraging improvement in terms of getting more eligible patients into our screening program, there’s still a lot of work to be done in the field,” Dr. Barta said. “Diagnosing lung cancer at earlier stages of disease is more cost effective in general for the health care system than fighting lung cancer at advanced stages, which requires more complex and multimodal and prolonged therapies.”
New evidence: Chest CTs for lung cancer screening reduces incidence of advanced lung cancer
In an analysis of the SEER database presented in June at the annual meeting of the American Society of Clinical Oncology, the adoption of low-dose chest computed tomography (LDCT) led to fewer diagnoses of advanced lung cancer, although these declines varied significantly by race and ethnicity. Non-Hispanic Blacks seemed to benefit the most with a 55% decline (P < .01), while Hispanics had the lowest rate of decline at 41% (P < .01). The change was recommended by USPSTF in 2013 after the National Lung Screening Trial revealed a 20% relative reduction in mortality when CT scans were used instead of chest radiography. The Centers for Medicare and Medicaid Services approved coverage of the screen in 2015.
The SEER study looked at data from 400,343 individuals from 2004-2014 (preintervention) and 2015-2018 (postintervention). The age-adjusted incidence of advanced lung cancer declined during both periods, but the decline was sharper between 2015 and 2018, with three fewer cases per 100,000 people than 2004-2014 (P < .01). Similar patterns were seen in subanalyses of males and females, non-Hispanic Whites, non-Hispanic Blacks, and Hispanics. The relative declines were largest in women, non-Hispanic Blacks, and people who lived outside of Metropolitan areas.
During a Q&A session that followed the presentation, Robert Smith, PhD, pointed out that the bar for eligibility of lung cancer risk has been set quite high, following the eligibility criteria for clinical trials. He noted that . “We are missing opportunities to prevent avertable lung cancer deaths,” said Dr. Smith, senior vice president of cancer screening at the American Cancer Society.
On the other hand, screening-prompted biopsies have the potential to cause harm, particularly in patients who already have lung disease, said Douglas Allen Arenberg, MD, professor at the University of Michigan, Ann Arbor. “I think that’s what scares most people is the potential downside, which is very hard to measure outside of a clinical trial,” said Dr. Arenberg, who served as a discussant for the presentation.
One way to reduce that risk is to identify biomarkers, either for screens or for incidentally-detected nodules, that have good negative predictive value. “If I had a blood test that is as good as a negative PET scan, I’m going to be much more likely to say, ‘Yeah, you’re 40 and your grandfather had lung cancer. Maybe you should get a CT. If we had that, we could screen a lot more people. Right now, I would discourage anybody who is at low risk from getting screened because when they come to me, the biggest opportunity I have to do harm is when I do a biopsy, and you always remember the ones that go wrong,” he said.
Dr. Arenberg also called for improvements in electronic medical records to better flag at-risk patients. “I think we as physicians have to demand more of the software developers that create these EMRs for us,” he said.
Another study in the same session used data from 1,391,088 patients drawn from the National Cancer Database between 2010 and 2017 to examine trends in diagnosis of stage I cancer. In 2010, 23.5% of patients were diagnosed as stage I, versus 29.1% in 2017. Stage I incidence increased from 25.8% to 31.7% in non–small cell lung cancer, but there was no statistically significant change in small cell lung cancer. As with the SEER database study, the researchers noted that the shift toward stage I diagnoses predated the recommendation of LDCT.
Dr. Arenberg suggested that the trend may come down to increased frequency of CT scans, which often collect incidental images of the lungs. He added that better access to care may also be helping to drive the change. “How much of that might have had something to do with the introduction 5 or 10 years earlier of the Affordable Care Act and people just simply having access to care and taking advantage of that?” Dr. Arenberg said.
But Dr. Arenberg said that not even screening can explain all the data. He referenced a stage shift in patients of all age groups in the National Cancer Database study, even those too young to be eligible for screening. “There’s something else going on here. It would be nice for us to understand what caused these trends, so perhaps we could accentuate that trend even more, but stage shifts are clearly occurring in lung cancer,” Dr. Arenberg said.
Dr. Barta has received grants from Genentech Health Equity Innovations Fund. Dr. Arenberg has no relevant financial disclosures. Dr. Smith’s potential disclosures could not be ascertained.
An analysis of trends in lung cancer screening since March 2021 when the U.S. Preventive Services Task Force (USPSTF) expanded the eligibility criteria for lung cancer screening, shows that significantly more Black men have been screened for lung cancer, but not women or undereducated people.
The eligibility for lung cancer screening was expanded in 2021 to include men and women under 50 years old and people who smoke at least one pack of cigarettes a day for the last 20 years. “
“Expansion of screening criteria is a critical first step to achieving equity in lung cancer screening for all high-risk populations, but myriad challenges remain before individuals enter the door for screening,” wrote the authors, led by Julie A. Barta, MD, Thomas Jefferson University, Philadelphia. “Health policy changes must occur simultaneously with efforts to expand community outreach, overcome logistical barriers, and facilitate screening adherence. Only after comprehensive strategies to dismantle screening barriers are identified, validated, and implemented can there be a truly equitable landscape for lung cancer screening.”
For the study, published in JAMA Open Network, researchers examined rates of centralized lung cancer screening in the Baltimore area. In addition to expanding lung cancer screening generally, there was hope that the expanded criteria might increase uptake of screening in populations that are traditionally underserved, such as African American, Hispanic, and female patients. Of 815 people screened during the study period (March-December 2021), 161 were newly eligible for screening under the 2021 criteria.
“There’s been quite a bit of work in the field demonstrating that Black men and women develop lung cancer at more advanced stages of disease, and they often are diagnosed at younger ages and have fewer pack-years of smoking. So the hypothesis was that this would reduce some of the disparities seen in lung cancer screening by making more people eligible,” Dr. Barta said in an interview.
The researchers categorized participants as those who would have been eligible for screening under the USPSTF 2013 guideline (age 55 or older, 30 or more pack-years, quit within the past 15 years), and those who would be eligible under the 2021 guideline (age 50 or older, 20 or more pack-years, quit within the past 15 years). Of the 2021 cohort, 54.5% were African American, versus 39.5% of the 2013 cohort (P = .002). There were no differences between the cohorts with respect to education level or gender.
“Although we’ve seen some encouraging improvement in terms of getting more eligible patients into our screening program, there’s still a lot of work to be done in the field,” Dr. Barta said. “Diagnosing lung cancer at earlier stages of disease is more cost effective in general for the health care system than fighting lung cancer at advanced stages, which requires more complex and multimodal and prolonged therapies.”
New evidence: Chest CTs for lung cancer screening reduces incidence of advanced lung cancer
In an analysis of the SEER database presented in June at the annual meeting of the American Society of Clinical Oncology, the adoption of low-dose chest computed tomography (LDCT) led to fewer diagnoses of advanced lung cancer, although these declines varied significantly by race and ethnicity. Non-Hispanic Blacks seemed to benefit the most with a 55% decline (P < .01), while Hispanics had the lowest rate of decline at 41% (P < .01). The change was recommended by USPSTF in 2013 after the National Lung Screening Trial revealed a 20% relative reduction in mortality when CT scans were used instead of chest radiography. The Centers for Medicare and Medicaid Services approved coverage of the screen in 2015.
The SEER study looked at data from 400,343 individuals from 2004-2014 (preintervention) and 2015-2018 (postintervention). The age-adjusted incidence of advanced lung cancer declined during both periods, but the decline was sharper between 2015 and 2018, with three fewer cases per 100,000 people than 2004-2014 (P < .01). Similar patterns were seen in subanalyses of males and females, non-Hispanic Whites, non-Hispanic Blacks, and Hispanics. The relative declines were largest in women, non-Hispanic Blacks, and people who lived outside of Metropolitan areas.
During a Q&A session that followed the presentation, Robert Smith, PhD, pointed out that the bar for eligibility of lung cancer risk has been set quite high, following the eligibility criteria for clinical trials. He noted that . “We are missing opportunities to prevent avertable lung cancer deaths,” said Dr. Smith, senior vice president of cancer screening at the American Cancer Society.
On the other hand, screening-prompted biopsies have the potential to cause harm, particularly in patients who already have lung disease, said Douglas Allen Arenberg, MD, professor at the University of Michigan, Ann Arbor. “I think that’s what scares most people is the potential downside, which is very hard to measure outside of a clinical trial,” said Dr. Arenberg, who served as a discussant for the presentation.
One way to reduce that risk is to identify biomarkers, either for screens or for incidentally-detected nodules, that have good negative predictive value. “If I had a blood test that is as good as a negative PET scan, I’m going to be much more likely to say, ‘Yeah, you’re 40 and your grandfather had lung cancer. Maybe you should get a CT. If we had that, we could screen a lot more people. Right now, I would discourage anybody who is at low risk from getting screened because when they come to me, the biggest opportunity I have to do harm is when I do a biopsy, and you always remember the ones that go wrong,” he said.
Dr. Arenberg also called for improvements in electronic medical records to better flag at-risk patients. “I think we as physicians have to demand more of the software developers that create these EMRs for us,” he said.
Another study in the same session used data from 1,391,088 patients drawn from the National Cancer Database between 2010 and 2017 to examine trends in diagnosis of stage I cancer. In 2010, 23.5% of patients were diagnosed as stage I, versus 29.1% in 2017. Stage I incidence increased from 25.8% to 31.7% in non–small cell lung cancer, but there was no statistically significant change in small cell lung cancer. As with the SEER database study, the researchers noted that the shift toward stage I diagnoses predated the recommendation of LDCT.
Dr. Arenberg suggested that the trend may come down to increased frequency of CT scans, which often collect incidental images of the lungs. He added that better access to care may also be helping to drive the change. “How much of that might have had something to do with the introduction 5 or 10 years earlier of the Affordable Care Act and people just simply having access to care and taking advantage of that?” Dr. Arenberg said.
But Dr. Arenberg said that not even screening can explain all the data. He referenced a stage shift in patients of all age groups in the National Cancer Database study, even those too young to be eligible for screening. “There’s something else going on here. It would be nice for us to understand what caused these trends, so perhaps we could accentuate that trend even more, but stage shifts are clearly occurring in lung cancer,” Dr. Arenberg said.
Dr. Barta has received grants from Genentech Health Equity Innovations Fund. Dr. Arenberg has no relevant financial disclosures. Dr. Smith’s potential disclosures could not be ascertained.
FROM JAMA NETWORK OPEN
New KRAS inhibitor shows promise in NSCLC
In a phase 2 cohort study, who had previously been treated with platinum-based chemotherapy and immune therapy.
Adagrasib targets KRAS (G12C), which had long been thought undruggable until research published in 2013 revealed a new binding pocket that did not compete directly against the protein’s natural binding partner. The new trial further validates the approach. “It supports that clinically effective targeted therapies can be developed for patients with KRAS (G12C)–mutant NSCLC,” said Pasi Jänne, MD, PhD, who is the lead author of the study describing the new results published online in the New England Journal of Medicine.
KRAS is the most frequently mutated oncogene in human cancers. A mutated form is found in about 25% of NSCLCs. KRAS plays a key role in cell signaling governing growth, maturation, and cell death. The mutated form is linked to cancer growth and spread. Patients with mutated KRAS have few effective treatment options.
Adagrasib is currently under study and not yet approved by the Food and Drug Administration. However, sotorasib (Lumakras, Amgen), which also inhibits KRAS (G12C), was approved in May 2021 by the FDA for KRAS (G12C)–mutated NSCLC. There are some key differences between the drugs. Adagrasib has a half-life of 23 hours versus 5 hours for sotorasib, and the newer drug has the potential to penetrate the central nervous system. That could be an important consideration in NSCLC since it often metastasizes to the brain. “Having pharmacological approaches to treat brain metastases is a wonderful new therapeutic option for lung cancer patients,” said Dr. Jänne, who is director of the Lowe Center for Thoracic Oncology at Dana Farber Cancer Institute, Boston.
Adagrasib is being investigated as part of the KRYSTAL-1 study, alone and as part of combinations in various solid tumors. Previously treated NSCLC KRAS (G12C) patients are also being enrolled in a phase 3 study of adagrasib combined with docetaxel, as well as another phase 2 study of adagrasib combined with pembrolizumab as first-line therapy for NSCLC KRAS (G12C).
Adagrasib is likely to remain a second-line therapy following chemotherapy and immunotherapy. “The activity by itself at the moment is not sufficient to be a first-line treatment. That may change in the future in combination with a standard of care agent or in a subset of patients with KRAS (G12C)–mutant NSCLC, although no subset with higher efficacy has been identified to date. Identification of predictive biomarkers for patients likely to benefit from single agent or an adagrasib combination treatment remains a high priority,” Dr. Jänne said.
The study included 116 patients who had previously been treated with platinum-based chemotherapy and anti–programmed death 1 or programmed death–ligand 1 therapy. They received 600 mg oral adagrasib twice per day over a median follow-up period of 12.9 months. About 42.9% (95% confidence interval, 33.5%-52.6%) experienced a confirmed objective response with a median duration of 8.5 months (95% CI, 6.2-13.8 months). The median progression-free survival was 6.5 months (95% CI, 4.7-8.4 months). After a median follow-up of 15.6 months, the median overall survival was 12.6 months (95% CI, 9.2-19.2 months). The estimated overall survival at 1 year was 50.8% (95% CI, 40.9%-60.0%).
33 patients had stable central nervous system metastases that had been previously treated. About 33.3% had an intracranial confirmed objective response (95% CI, 18.0-51.8%) with a median duration of response of 11.2 months (95% CI, 2.99 months to not available).
Adverse events are similar to what is seen with other targeted therapies, according to Dr. Jänne. 97.4% of patient reported a treatment-related adverse event; 52.6% had grade 1-2 adverse events, and 44.8% had grade 3 adverse events. 6.9% discontinued the drug as a result.
Dr. Jänne has consulted for Mirati Therapeutics and is a member of its scientific advisory board. The study was funded by Mirati Therapeutics.
In a phase 2 cohort study, who had previously been treated with platinum-based chemotherapy and immune therapy.
Adagrasib targets KRAS (G12C), which had long been thought undruggable until research published in 2013 revealed a new binding pocket that did not compete directly against the protein’s natural binding partner. The new trial further validates the approach. “It supports that clinically effective targeted therapies can be developed for patients with KRAS (G12C)–mutant NSCLC,” said Pasi Jänne, MD, PhD, who is the lead author of the study describing the new results published online in the New England Journal of Medicine.
KRAS is the most frequently mutated oncogene in human cancers. A mutated form is found in about 25% of NSCLCs. KRAS plays a key role in cell signaling governing growth, maturation, and cell death. The mutated form is linked to cancer growth and spread. Patients with mutated KRAS have few effective treatment options.
Adagrasib is currently under study and not yet approved by the Food and Drug Administration. However, sotorasib (Lumakras, Amgen), which also inhibits KRAS (G12C), was approved in May 2021 by the FDA for KRAS (G12C)–mutated NSCLC. There are some key differences between the drugs. Adagrasib has a half-life of 23 hours versus 5 hours for sotorasib, and the newer drug has the potential to penetrate the central nervous system. That could be an important consideration in NSCLC since it often metastasizes to the brain. “Having pharmacological approaches to treat brain metastases is a wonderful new therapeutic option for lung cancer patients,” said Dr. Jänne, who is director of the Lowe Center for Thoracic Oncology at Dana Farber Cancer Institute, Boston.
Adagrasib is being investigated as part of the KRYSTAL-1 study, alone and as part of combinations in various solid tumors. Previously treated NSCLC KRAS (G12C) patients are also being enrolled in a phase 3 study of adagrasib combined with docetaxel, as well as another phase 2 study of adagrasib combined with pembrolizumab as first-line therapy for NSCLC KRAS (G12C).
Adagrasib is likely to remain a second-line therapy following chemotherapy and immunotherapy. “The activity by itself at the moment is not sufficient to be a first-line treatment. That may change in the future in combination with a standard of care agent or in a subset of patients with KRAS (G12C)–mutant NSCLC, although no subset with higher efficacy has been identified to date. Identification of predictive biomarkers for patients likely to benefit from single agent or an adagrasib combination treatment remains a high priority,” Dr. Jänne said.
The study included 116 patients who had previously been treated with platinum-based chemotherapy and anti–programmed death 1 or programmed death–ligand 1 therapy. They received 600 mg oral adagrasib twice per day over a median follow-up period of 12.9 months. About 42.9% (95% confidence interval, 33.5%-52.6%) experienced a confirmed objective response with a median duration of 8.5 months (95% CI, 6.2-13.8 months). The median progression-free survival was 6.5 months (95% CI, 4.7-8.4 months). After a median follow-up of 15.6 months, the median overall survival was 12.6 months (95% CI, 9.2-19.2 months). The estimated overall survival at 1 year was 50.8% (95% CI, 40.9%-60.0%).
33 patients had stable central nervous system metastases that had been previously treated. About 33.3% had an intracranial confirmed objective response (95% CI, 18.0-51.8%) with a median duration of response of 11.2 months (95% CI, 2.99 months to not available).
Adverse events are similar to what is seen with other targeted therapies, according to Dr. Jänne. 97.4% of patient reported a treatment-related adverse event; 52.6% had grade 1-2 adverse events, and 44.8% had grade 3 adverse events. 6.9% discontinued the drug as a result.
Dr. Jänne has consulted for Mirati Therapeutics and is a member of its scientific advisory board. The study was funded by Mirati Therapeutics.
In a phase 2 cohort study, who had previously been treated with platinum-based chemotherapy and immune therapy.
Adagrasib targets KRAS (G12C), which had long been thought undruggable until research published in 2013 revealed a new binding pocket that did not compete directly against the protein’s natural binding partner. The new trial further validates the approach. “It supports that clinically effective targeted therapies can be developed for patients with KRAS (G12C)–mutant NSCLC,” said Pasi Jänne, MD, PhD, who is the lead author of the study describing the new results published online in the New England Journal of Medicine.
KRAS is the most frequently mutated oncogene in human cancers. A mutated form is found in about 25% of NSCLCs. KRAS plays a key role in cell signaling governing growth, maturation, and cell death. The mutated form is linked to cancer growth and spread. Patients with mutated KRAS have few effective treatment options.
Adagrasib is currently under study and not yet approved by the Food and Drug Administration. However, sotorasib (Lumakras, Amgen), which also inhibits KRAS (G12C), was approved in May 2021 by the FDA for KRAS (G12C)–mutated NSCLC. There are some key differences between the drugs. Adagrasib has a half-life of 23 hours versus 5 hours for sotorasib, and the newer drug has the potential to penetrate the central nervous system. That could be an important consideration in NSCLC since it often metastasizes to the brain. “Having pharmacological approaches to treat brain metastases is a wonderful new therapeutic option for lung cancer patients,” said Dr. Jänne, who is director of the Lowe Center for Thoracic Oncology at Dana Farber Cancer Institute, Boston.
Adagrasib is being investigated as part of the KRYSTAL-1 study, alone and as part of combinations in various solid tumors. Previously treated NSCLC KRAS (G12C) patients are also being enrolled in a phase 3 study of adagrasib combined with docetaxel, as well as another phase 2 study of adagrasib combined with pembrolizumab as first-line therapy for NSCLC KRAS (G12C).
Adagrasib is likely to remain a second-line therapy following chemotherapy and immunotherapy. “The activity by itself at the moment is not sufficient to be a first-line treatment. That may change in the future in combination with a standard of care agent or in a subset of patients with KRAS (G12C)–mutant NSCLC, although no subset with higher efficacy has been identified to date. Identification of predictive biomarkers for patients likely to benefit from single agent or an adagrasib combination treatment remains a high priority,” Dr. Jänne said.
The study included 116 patients who had previously been treated with platinum-based chemotherapy and anti–programmed death 1 or programmed death–ligand 1 therapy. They received 600 mg oral adagrasib twice per day over a median follow-up period of 12.9 months. About 42.9% (95% confidence interval, 33.5%-52.6%) experienced a confirmed objective response with a median duration of 8.5 months (95% CI, 6.2-13.8 months). The median progression-free survival was 6.5 months (95% CI, 4.7-8.4 months). After a median follow-up of 15.6 months, the median overall survival was 12.6 months (95% CI, 9.2-19.2 months). The estimated overall survival at 1 year was 50.8% (95% CI, 40.9%-60.0%).
33 patients had stable central nervous system metastases that had been previously treated. About 33.3% had an intracranial confirmed objective response (95% CI, 18.0-51.8%) with a median duration of response of 11.2 months (95% CI, 2.99 months to not available).
Adverse events are similar to what is seen with other targeted therapies, according to Dr. Jänne. 97.4% of patient reported a treatment-related adverse event; 52.6% had grade 1-2 adverse events, and 44.8% had grade 3 adverse events. 6.9% discontinued the drug as a result.
Dr. Jänne has consulted for Mirati Therapeutics and is a member of its scientific advisory board. The study was funded by Mirati Therapeutics.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Dry cough and dyspnea
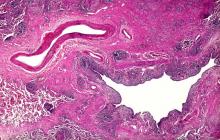
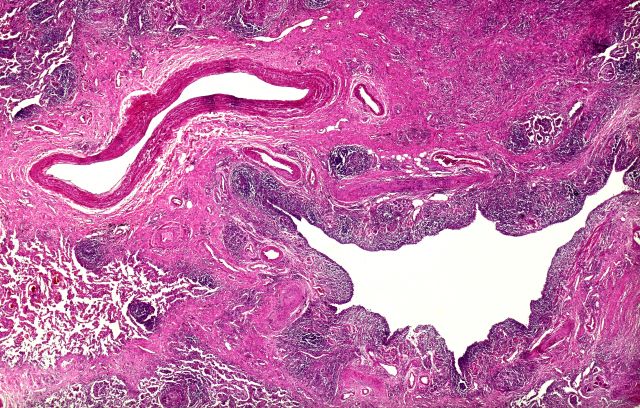
Based on the patient's presentation and workup, the likely diagnosis is adenosquamous carcinoma of the lung, a relatively rare subtype of non–small cell lung cancer (NSCLC). Adenosquamous carcinoma displays qualities of both squamous cell carcinoma and adenocarcinoma; for definitive diagnosis, the cancer must contain 10% of each of these major NSCLC subtypes. Maeda and colleagues concluded that adenosquamous carcinoma occurs more frequently among men and that the age at the time of diagnosis is higher among such cancers compared with adenocarcinoma. Several studies have confirmed that adenosquamous carcinoma of the lung is also more prevalent among smokers.
Though a diagnosis of adenosquamous carcinoma may be suspected after small biopsies, cytology, or excisional biopsies, definitive diagnosis necessitates a resection specimen. If any adenocarcinoma component is observed in a biopsy specimen that is otherwise squamous, as in the present case, this finding is an indication for molecular testing. Epidermal growth factor receptor (EGFR) mutations may be present in adenosquamous carcinoma cancers, despite a majority of cancers with EGFR mutations being among nonsmokers or former light smokers with adenocarcinoma histology. In addition, even for patients diagnosed with squamous cell carcinoma, adenosquamous carcinoma should be considered if genetic testing suggests EGFR mutations.
Relative to adenocarcinoma and squamous cell carcinoma, adenosquamous carcinoma has higher grade malignancy, more advanced postoperative stage, and stronger lymph nodal invasiveness. In terms of treatment, surgical resection is the curative option for adenosquamous carcinoma of the lung, with lobectomy with lymphadenectomy considered for first-line treatment. Though the most beneficial chemotherapy regimen for patients with adenosquamous carcinoma of the lung remains the subject of investigation, platinum-based doublet chemotherapy is the current standard treatment option. EGFR tyrosine kinase inhibitors may be an effective option for EGFR-positive patients.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
Based on the patient's presentation and workup, the likely diagnosis is adenosquamous carcinoma of the lung, a relatively rare subtype of non–small cell lung cancer (NSCLC). Adenosquamous carcinoma displays qualities of both squamous cell carcinoma and adenocarcinoma; for definitive diagnosis, the cancer must contain 10% of each of these major NSCLC subtypes. Maeda and colleagues concluded that adenosquamous carcinoma occurs more frequently among men and that the age at the time of diagnosis is higher among such cancers compared with adenocarcinoma. Several studies have confirmed that adenosquamous carcinoma of the lung is also more prevalent among smokers.
Though a diagnosis of adenosquamous carcinoma may be suspected after small biopsies, cytology, or excisional biopsies, definitive diagnosis necessitates a resection specimen. If any adenocarcinoma component is observed in a biopsy specimen that is otherwise squamous, as in the present case, this finding is an indication for molecular testing. Epidermal growth factor receptor (EGFR) mutations may be present in adenosquamous carcinoma cancers, despite a majority of cancers with EGFR mutations being among nonsmokers or former light smokers with adenocarcinoma histology. In addition, even for patients diagnosed with squamous cell carcinoma, adenosquamous carcinoma should be considered if genetic testing suggests EGFR mutations.
Relative to adenocarcinoma and squamous cell carcinoma, adenosquamous carcinoma has higher grade malignancy, more advanced postoperative stage, and stronger lymph nodal invasiveness. In terms of treatment, surgical resection is the curative option for adenosquamous carcinoma of the lung, with lobectomy with lymphadenectomy considered for first-line treatment. Though the most beneficial chemotherapy regimen for patients with adenosquamous carcinoma of the lung remains the subject of investigation, platinum-based doublet chemotherapy is the current standard treatment option. EGFR tyrosine kinase inhibitors may be an effective option for EGFR-positive patients.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
Based on the patient's presentation and workup, the likely diagnosis is adenosquamous carcinoma of the lung, a relatively rare subtype of non–small cell lung cancer (NSCLC). Adenosquamous carcinoma displays qualities of both squamous cell carcinoma and adenocarcinoma; for definitive diagnosis, the cancer must contain 10% of each of these major NSCLC subtypes. Maeda and colleagues concluded that adenosquamous carcinoma occurs more frequently among men and that the age at the time of diagnosis is higher among such cancers compared with adenocarcinoma. Several studies have confirmed that adenosquamous carcinoma of the lung is also more prevalent among smokers.
Though a diagnosis of adenosquamous carcinoma may be suspected after small biopsies, cytology, or excisional biopsies, definitive diagnosis necessitates a resection specimen. If any adenocarcinoma component is observed in a biopsy specimen that is otherwise squamous, as in the present case, this finding is an indication for molecular testing. Epidermal growth factor receptor (EGFR) mutations may be present in adenosquamous carcinoma cancers, despite a majority of cancers with EGFR mutations being among nonsmokers or former light smokers with adenocarcinoma histology. In addition, even for patients diagnosed with squamous cell carcinoma, adenosquamous carcinoma should be considered if genetic testing suggests EGFR mutations.
Relative to adenocarcinoma and squamous cell carcinoma, adenosquamous carcinoma has higher grade malignancy, more advanced postoperative stage, and stronger lymph nodal invasiveness. In terms of treatment, surgical resection is the curative option for adenosquamous carcinoma of the lung, with lobectomy with lymphadenectomy considered for first-line treatment. Though the most beneficial chemotherapy regimen for patients with adenosquamous carcinoma of the lung remains the subject of investigation, platinum-based doublet chemotherapy is the current standard treatment option. EGFR tyrosine kinase inhibitors may be an effective option for EGFR-positive patients.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
A 58-year-old man with a 20-year–pack history of smoking initially presented with a persistent dry cough and dyspnea. Clubbing was noted on physical examination and breath sounds in the right upper lung were weak. Other than hypertension, which the patient manages with angiotensin-converting enzyme (ACE) inhibitors, medical history is unremarkable. The patient notes that this medication has always made him cough, but dyspnea has only developed over the past 6 weeks. Respiratory symptoms prompted a chest radiograph which revealed a mass in the upper lobe of the right lung. Transbronchial lung biopsy of the right lung reveals components of adenocarcinoma; the specimen is otherwise squamous.
Drugging the undruggable
including 68% of pancreatic tumors and 20% of all non–small cell lung cancers (NSCLC).
We now have a treatment – sotorasib – for patients with locally advanced or metastatic NSCLC that is driven by a KRAS mutation (G12C). And, now, there is a second treatment – adagrasib – under study, which, according to a presentation recently made at the annual meeting of the American Society of Clinical Oncology, looks promising.
Ras is a membrane-bound regulatory protein (G protein) belonging to the family of guanosine triphosphatases (GTPases). Ras functions as a guanosine diphosphate/triphosphate binary switch by cycling between the active GTP-bound and the inactive GDP-bound states in response to extracellular stimuli. The KRAS (G12C) mutation affects the active form of KRAS and results in abnormally high concentrations of GTP-bound KRAS leading to hyperactivation of downstream oncogenic pathways and uncontrolled cell growth, specifically of ERK and MEK signaling pathways.
At the ASCO annual meeting in June, Spira and colleagues reported the results of cohort A of the KRYSTAL-1 study evaluating adagrasib as second-line therapy patients with advanced solid tumors harboring a KRAS (G12C) mutation. Like sotorasib, adagrasib is a KRAS (G12C) inhibitor that irreversibly and selectively binds KRAS (G12C), locking it in its inactive state. In this study, patients had to have failed first-line chemotherapy and immunotherapy with 43% of lung cancer patients responding. The 12-month overall survival (OS) was 51%, median overall survival was 12.6 and median progression-free survival (PFS) was 6.5 months. Twenty-five patients with KRAS (G12C)–mutant NSCLC and active, untreated central nervous system metastases received adagrasib in a phase 1b cohort. The intracranial overall response rate was 31.6% and median intracranial PFS was 4.2 months. Systemic ORR was 35.0% (7/20), the disease control rate was 80.0% (16/20) and median duration of response was 9.6 months. Based on these data, a phase 3 trial evaluating adagrasib monotherapy versus docetaxel in previously treated patients with KRAS (G12C) mutant NSCLC is ongoing.
The Food and Drug Administration approval of sotorasib in 2021 was, in part, based on the results of a single-arm, phase 2, second-line study of patients who had previously received platinum-based chemotherapy and/or immunotherapy. An ORR rate of 37.1% was reported with a median PFS of 6.8 months and median OS of 12.5 months leading to the FDA approval. Responses were observed across the range of baseline PD-L1 expression levels: 48% of PD-L1 negative, 39% with PD-L1 between 1%-49%, and 22% of patients with a PD-L1 of greater than 50% having a response.
The major toxicities observed in these studies were gastrointestinal (diarrhea, nausea, vomiting) and hepatic (elevated liver enzymes). About 97% of patients on adagrasib experienced any treatment-related adverse events, and 43% experienced a grade 3 or 4 treatment-related adverse event leading to dose reduction in 52% of patients, a dose interruption in 61% of patients, and a 7% discontinuation rate. About 70% of patients treated with sotorasib had a treatment-related adverse event of any grade, and 21% reported grade 3 or 4 treatment-related adverse events.
A subgroup in the KRYSTAL-1 trial reported an intracranial ORR of 32% in patients with active, untreated CNS metastases. Median overall survival has not yet reached concordance between systemic and intracranial disease control was 88%. In addition, preliminary data from two patients with untreated CNS metastases from a phase 1b cohort found cerebrospinal fluid concentrations of adagrasib with a mean ratio of unbound brain-to-plasma concentration of 0.47, which is comparable or exceeds values for known CNS-penetrant tyrosine kinase inhibitors.
Unfortunately, KRAS (G12C) is not the only KRAS mutation out there. There are a myriad of others, such as G12V and G12D. Hopefully, we will be seeing more drugs aimed at this set of important mutations. Another question, of course, is when and if these drugs will move to the first-line setting.
Dr. Schiller is a medical oncologist and founding member of Oncologists United for Climate and Health. She is a former board member of the International Association for the Study of Lung Cancer and a current board member of the Lung Cancer Research Foundation.
including 68% of pancreatic tumors and 20% of all non–small cell lung cancers (NSCLC).
We now have a treatment – sotorasib – for patients with locally advanced or metastatic NSCLC that is driven by a KRAS mutation (G12C). And, now, there is a second treatment – adagrasib – under study, which, according to a presentation recently made at the annual meeting of the American Society of Clinical Oncology, looks promising.
Ras is a membrane-bound regulatory protein (G protein) belonging to the family of guanosine triphosphatases (GTPases). Ras functions as a guanosine diphosphate/triphosphate binary switch by cycling between the active GTP-bound and the inactive GDP-bound states in response to extracellular stimuli. The KRAS (G12C) mutation affects the active form of KRAS and results in abnormally high concentrations of GTP-bound KRAS leading to hyperactivation of downstream oncogenic pathways and uncontrolled cell growth, specifically of ERK and MEK signaling pathways.
At the ASCO annual meeting in June, Spira and colleagues reported the results of cohort A of the KRYSTAL-1 study evaluating adagrasib as second-line therapy patients with advanced solid tumors harboring a KRAS (G12C) mutation. Like sotorasib, adagrasib is a KRAS (G12C) inhibitor that irreversibly and selectively binds KRAS (G12C), locking it in its inactive state. In this study, patients had to have failed first-line chemotherapy and immunotherapy with 43% of lung cancer patients responding. The 12-month overall survival (OS) was 51%, median overall survival was 12.6 and median progression-free survival (PFS) was 6.5 months. Twenty-five patients with KRAS (G12C)–mutant NSCLC and active, untreated central nervous system metastases received adagrasib in a phase 1b cohort. The intracranial overall response rate was 31.6% and median intracranial PFS was 4.2 months. Systemic ORR was 35.0% (7/20), the disease control rate was 80.0% (16/20) and median duration of response was 9.6 months. Based on these data, a phase 3 trial evaluating adagrasib monotherapy versus docetaxel in previously treated patients with KRAS (G12C) mutant NSCLC is ongoing.
The Food and Drug Administration approval of sotorasib in 2021 was, in part, based on the results of a single-arm, phase 2, second-line study of patients who had previously received platinum-based chemotherapy and/or immunotherapy. An ORR rate of 37.1% was reported with a median PFS of 6.8 months and median OS of 12.5 months leading to the FDA approval. Responses were observed across the range of baseline PD-L1 expression levels: 48% of PD-L1 negative, 39% with PD-L1 between 1%-49%, and 22% of patients with a PD-L1 of greater than 50% having a response.
The major toxicities observed in these studies were gastrointestinal (diarrhea, nausea, vomiting) and hepatic (elevated liver enzymes). About 97% of patients on adagrasib experienced any treatment-related adverse events, and 43% experienced a grade 3 or 4 treatment-related adverse event leading to dose reduction in 52% of patients, a dose interruption in 61% of patients, and a 7% discontinuation rate. About 70% of patients treated with sotorasib had a treatment-related adverse event of any grade, and 21% reported grade 3 or 4 treatment-related adverse events.
A subgroup in the KRYSTAL-1 trial reported an intracranial ORR of 32% in patients with active, untreated CNS metastases. Median overall survival has not yet reached concordance between systemic and intracranial disease control was 88%. In addition, preliminary data from two patients with untreated CNS metastases from a phase 1b cohort found cerebrospinal fluid concentrations of adagrasib with a mean ratio of unbound brain-to-plasma concentration of 0.47, which is comparable or exceeds values for known CNS-penetrant tyrosine kinase inhibitors.
Unfortunately, KRAS (G12C) is not the only KRAS mutation out there. There are a myriad of others, such as G12V and G12D. Hopefully, we will be seeing more drugs aimed at this set of important mutations. Another question, of course, is when and if these drugs will move to the first-line setting.
Dr. Schiller is a medical oncologist and founding member of Oncologists United for Climate and Health. She is a former board member of the International Association for the Study of Lung Cancer and a current board member of the Lung Cancer Research Foundation.
including 68% of pancreatic tumors and 20% of all non–small cell lung cancers (NSCLC).
We now have a treatment – sotorasib – for patients with locally advanced or metastatic NSCLC that is driven by a KRAS mutation (G12C). And, now, there is a second treatment – adagrasib – under study, which, according to a presentation recently made at the annual meeting of the American Society of Clinical Oncology, looks promising.
Ras is a membrane-bound regulatory protein (G protein) belonging to the family of guanosine triphosphatases (GTPases). Ras functions as a guanosine diphosphate/triphosphate binary switch by cycling between the active GTP-bound and the inactive GDP-bound states in response to extracellular stimuli. The KRAS (G12C) mutation affects the active form of KRAS and results in abnormally high concentrations of GTP-bound KRAS leading to hyperactivation of downstream oncogenic pathways and uncontrolled cell growth, specifically of ERK and MEK signaling pathways.
At the ASCO annual meeting in June, Spira and colleagues reported the results of cohort A of the KRYSTAL-1 study evaluating adagrasib as second-line therapy patients with advanced solid tumors harboring a KRAS (G12C) mutation. Like sotorasib, adagrasib is a KRAS (G12C) inhibitor that irreversibly and selectively binds KRAS (G12C), locking it in its inactive state. In this study, patients had to have failed first-line chemotherapy and immunotherapy with 43% of lung cancer patients responding. The 12-month overall survival (OS) was 51%, median overall survival was 12.6 and median progression-free survival (PFS) was 6.5 months. Twenty-five patients with KRAS (G12C)–mutant NSCLC and active, untreated central nervous system metastases received adagrasib in a phase 1b cohort. The intracranial overall response rate was 31.6% and median intracranial PFS was 4.2 months. Systemic ORR was 35.0% (7/20), the disease control rate was 80.0% (16/20) and median duration of response was 9.6 months. Based on these data, a phase 3 trial evaluating adagrasib monotherapy versus docetaxel in previously treated patients with KRAS (G12C) mutant NSCLC is ongoing.
The Food and Drug Administration approval of sotorasib in 2021 was, in part, based on the results of a single-arm, phase 2, second-line study of patients who had previously received platinum-based chemotherapy and/or immunotherapy. An ORR rate of 37.1% was reported with a median PFS of 6.8 months and median OS of 12.5 months leading to the FDA approval. Responses were observed across the range of baseline PD-L1 expression levels: 48% of PD-L1 negative, 39% with PD-L1 between 1%-49%, and 22% of patients with a PD-L1 of greater than 50% having a response.
The major toxicities observed in these studies were gastrointestinal (diarrhea, nausea, vomiting) and hepatic (elevated liver enzymes). About 97% of patients on adagrasib experienced any treatment-related adverse events, and 43% experienced a grade 3 or 4 treatment-related adverse event leading to dose reduction in 52% of patients, a dose interruption in 61% of patients, and a 7% discontinuation rate. About 70% of patients treated with sotorasib had a treatment-related adverse event of any grade, and 21% reported grade 3 or 4 treatment-related adverse events.
A subgroup in the KRYSTAL-1 trial reported an intracranial ORR of 32% in patients with active, untreated CNS metastases. Median overall survival has not yet reached concordance between systemic and intracranial disease control was 88%. In addition, preliminary data from two patients with untreated CNS metastases from a phase 1b cohort found cerebrospinal fluid concentrations of adagrasib with a mean ratio of unbound brain-to-plasma concentration of 0.47, which is comparable or exceeds values for known CNS-penetrant tyrosine kinase inhibitors.
Unfortunately, KRAS (G12C) is not the only KRAS mutation out there. There are a myriad of others, such as G12V and G12D. Hopefully, we will be seeing more drugs aimed at this set of important mutations. Another question, of course, is when and if these drugs will move to the first-line setting.
Dr. Schiller is a medical oncologist and founding member of Oncologists United for Climate and Health. She is a former board member of the International Association for the Study of Lung Cancer and a current board member of the Lung Cancer Research Foundation.