User login
Neoadjuvant dabrafenib and trametinib improves event-free survival in resectable melanoma
For patients with surgically resectable, BRAF-mutated melanoma, neoadjuvant and adjuvant treatment with the combination of dabrafenib and trametinib resulted in significantly longer event-free survival compared with standard care, according to results of a randomized study.
The trial was closed early because of the “large difference” in event-free survival favoring the neoadjuvant approach, the authors wrote (Lancet Oncol. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9.
While early closure limits interpretation of results, they do provide important proof-of-concept and data for future studies, wrote the authors, led by Rodabe N Amaria, MD, of the department of medical oncology, University of Texas MD Anderson Cancer Center, Houston.
“The clinical and translational results strongly support the rationale for further assessment of neoadjuvant therapy in patients with high-risk, surgically resectable melanoma,” Dr. Amaria and colleagues said in the report on the randomized trial, believed to be the first to evaluate the role of neoadjuvant therapy versus standard care in BRAF-mutated melanoma.
Dabrafenib and trametinib combination therapy is approved as a treatment for patients with unresectable or metastatic stage IV melanoma and a BRAFV600 mutation, which is found in about half of cutaneous melanomas, authors said.
To evaluate the combination in earlier stage disease, Dr. Amaria and coinvestigators conducted a single-center, open-label, randomized, phase 2 trial of 21 patients with surgically resectable clinical stage III or oligometastatic stage IV melanoma with BRAFV600E or BRAFV600K mutations.
Patients were randomized 2:1 to receive the neoadjuvant/adjuvant treatment or to standard of care, which consisted of standard surgery plus consideration for adjuvant therapy, the authors said. Those patients assigned to the targeted therapy arm received 8 weeks of neoadjuvant dabrafenib and trametinib followed by surgery, then adjuvant dabrafenib and trametinib for up to 44 weeks.
Event-free survival, the primary endpoint of the trial, was a median of 19.7 months for neoadjuvant plus adjuvant dabrafenib and trametinib, versus 2.9 months for standard care (P less than .0001), the investigators reported.
Dabrafenib and trametinib combination therapy was well tolerated as neoadjuvant and adjuvant therapy, with no grade 4 adverse events or treatment related deaths, according to the investigators. The most common grade 3 adverse event seen with the combination was diarrhea, occurring in 2 patients (15%).
The trial is continuing as a single-arm study of neoadjuvant plus adjuvant dabrafenib and trametinib.
Dr. Amaria and colleagues reported individual disclosures related to Merck, Bristol-Myers Squibb, Array Biopharma, and others, including Novartis Pharmaceuticals Corp., which supplied drugs and funded clinical aspects of the study.
SOURCE: Amaria et al. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9
Although results of the study by Amaria et al. are “promising,” the role of neoadjuvant therapy in treatment of stage III–IV oligometastatic melanoma in clinical practice “is unclear for now,” melanoma specialists Paolo A. Ascierto, MD, and Alexander M. M. Eggermont, MD, PhD, wrote in an editorial.
Amaria et al. have presented results of the first randomized trial to evaluate neoadjuvant therapy versus standard care in patients with high-risk resectable BRAF-mutated melanoma.
Patients who received both neoadjuvant and adjuvant treatment with the dabrafenib/trametinib combination had superior event-free survival versus standard surgery and consideration for adjuvant therapy, published results show.
However, previous studies have already shown good results on this, that adjuvant dabrafenib plus trametinib (as well as nivolumab monotherapy) in this setting, “raising the question of whether a neoadjuvant approach is really needed, especially given a possible reduction of the role of surgery in the future,” Dr. Ascierto and Dr. Eggermont wrote.
Alternatively, adjuvant therapy with newer, more effective agents may be a “better way forward,” they said, noting that three patients in the trial by Amaria et al. who progressed after neoadjuvant/adjuvant dabrafenib and trametinib relapsed at first with brain metastases, raising the question of whether the treatment “might induce a resistant phenotype predisposed to the development of CNS metastases.”
That said, effectively combining neoadjuvant with adjuvant therapy could reduce the extent of surgery, make radiotherapy redundant, or increase distant metastasis-free survival and overall survival, among other benefits.
“The next generation of adjuvant trials should aim to address these outstanding questions,” they concluded.
Dr. Ascierto is with Istituto Nazionale Tumori Fondazione “G Pascale,” Napoli, Italy, and Dr. Eggermont is with Cancer Institute Gustave Roussy, University Paris-Sud, France. This commentary is based on their editorial appearing in The Lancet Oncology (2018 Jan 17. doi: 10.1016/S1470-2045[18]30016-0). The authors reported disclosures related to Novartis, Merck Serono, Bristol-Myers Squibb, Amgen, and others.
Although results of the study by Amaria et al. are “promising,” the role of neoadjuvant therapy in treatment of stage III–IV oligometastatic melanoma in clinical practice “is unclear for now,” melanoma specialists Paolo A. Ascierto, MD, and Alexander M. M. Eggermont, MD, PhD, wrote in an editorial.
Amaria et al. have presented results of the first randomized trial to evaluate neoadjuvant therapy versus standard care in patients with high-risk resectable BRAF-mutated melanoma.
Patients who received both neoadjuvant and adjuvant treatment with the dabrafenib/trametinib combination had superior event-free survival versus standard surgery and consideration for adjuvant therapy, published results show.
However, previous studies have already shown good results on this, that adjuvant dabrafenib plus trametinib (as well as nivolumab monotherapy) in this setting, “raising the question of whether a neoadjuvant approach is really needed, especially given a possible reduction of the role of surgery in the future,” Dr. Ascierto and Dr. Eggermont wrote.
Alternatively, adjuvant therapy with newer, more effective agents may be a “better way forward,” they said, noting that three patients in the trial by Amaria et al. who progressed after neoadjuvant/adjuvant dabrafenib and trametinib relapsed at first with brain metastases, raising the question of whether the treatment “might induce a resistant phenotype predisposed to the development of CNS metastases.”
That said, effectively combining neoadjuvant with adjuvant therapy could reduce the extent of surgery, make radiotherapy redundant, or increase distant metastasis-free survival and overall survival, among other benefits.
“The next generation of adjuvant trials should aim to address these outstanding questions,” they concluded.
Dr. Ascierto is with Istituto Nazionale Tumori Fondazione “G Pascale,” Napoli, Italy, and Dr. Eggermont is with Cancer Institute Gustave Roussy, University Paris-Sud, France. This commentary is based on their editorial appearing in The Lancet Oncology (2018 Jan 17. doi: 10.1016/S1470-2045[18]30016-0). The authors reported disclosures related to Novartis, Merck Serono, Bristol-Myers Squibb, Amgen, and others.
Although results of the study by Amaria et al. are “promising,” the role of neoadjuvant therapy in treatment of stage III–IV oligometastatic melanoma in clinical practice “is unclear for now,” melanoma specialists Paolo A. Ascierto, MD, and Alexander M. M. Eggermont, MD, PhD, wrote in an editorial.
Amaria et al. have presented results of the first randomized trial to evaluate neoadjuvant therapy versus standard care in patients with high-risk resectable BRAF-mutated melanoma.
Patients who received both neoadjuvant and adjuvant treatment with the dabrafenib/trametinib combination had superior event-free survival versus standard surgery and consideration for adjuvant therapy, published results show.
However, previous studies have already shown good results on this, that adjuvant dabrafenib plus trametinib (as well as nivolumab monotherapy) in this setting, “raising the question of whether a neoadjuvant approach is really needed, especially given a possible reduction of the role of surgery in the future,” Dr. Ascierto and Dr. Eggermont wrote.
Alternatively, adjuvant therapy with newer, more effective agents may be a “better way forward,” they said, noting that three patients in the trial by Amaria et al. who progressed after neoadjuvant/adjuvant dabrafenib and trametinib relapsed at first with brain metastases, raising the question of whether the treatment “might induce a resistant phenotype predisposed to the development of CNS metastases.”
That said, effectively combining neoadjuvant with adjuvant therapy could reduce the extent of surgery, make radiotherapy redundant, or increase distant metastasis-free survival and overall survival, among other benefits.
“The next generation of adjuvant trials should aim to address these outstanding questions,” they concluded.
Dr. Ascierto is with Istituto Nazionale Tumori Fondazione “G Pascale,” Napoli, Italy, and Dr. Eggermont is with Cancer Institute Gustave Roussy, University Paris-Sud, France. This commentary is based on their editorial appearing in The Lancet Oncology (2018 Jan 17. doi: 10.1016/S1470-2045[18]30016-0). The authors reported disclosures related to Novartis, Merck Serono, Bristol-Myers Squibb, Amgen, and others.
For patients with surgically resectable, BRAF-mutated melanoma, neoadjuvant and adjuvant treatment with the combination of dabrafenib and trametinib resulted in significantly longer event-free survival compared with standard care, according to results of a randomized study.
The trial was closed early because of the “large difference” in event-free survival favoring the neoadjuvant approach, the authors wrote (Lancet Oncol. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9.
While early closure limits interpretation of results, they do provide important proof-of-concept and data for future studies, wrote the authors, led by Rodabe N Amaria, MD, of the department of medical oncology, University of Texas MD Anderson Cancer Center, Houston.
“The clinical and translational results strongly support the rationale for further assessment of neoadjuvant therapy in patients with high-risk, surgically resectable melanoma,” Dr. Amaria and colleagues said in the report on the randomized trial, believed to be the first to evaluate the role of neoadjuvant therapy versus standard care in BRAF-mutated melanoma.
Dabrafenib and trametinib combination therapy is approved as a treatment for patients with unresectable or metastatic stage IV melanoma and a BRAFV600 mutation, which is found in about half of cutaneous melanomas, authors said.
To evaluate the combination in earlier stage disease, Dr. Amaria and coinvestigators conducted a single-center, open-label, randomized, phase 2 trial of 21 patients with surgically resectable clinical stage III or oligometastatic stage IV melanoma with BRAFV600E or BRAFV600K mutations.
Patients were randomized 2:1 to receive the neoadjuvant/adjuvant treatment or to standard of care, which consisted of standard surgery plus consideration for adjuvant therapy, the authors said. Those patients assigned to the targeted therapy arm received 8 weeks of neoadjuvant dabrafenib and trametinib followed by surgery, then adjuvant dabrafenib and trametinib for up to 44 weeks.
Event-free survival, the primary endpoint of the trial, was a median of 19.7 months for neoadjuvant plus adjuvant dabrafenib and trametinib, versus 2.9 months for standard care (P less than .0001), the investigators reported.
Dabrafenib and trametinib combination therapy was well tolerated as neoadjuvant and adjuvant therapy, with no grade 4 adverse events or treatment related deaths, according to the investigators. The most common grade 3 adverse event seen with the combination was diarrhea, occurring in 2 patients (15%).
The trial is continuing as a single-arm study of neoadjuvant plus adjuvant dabrafenib and trametinib.
Dr. Amaria and colleagues reported individual disclosures related to Merck, Bristol-Myers Squibb, Array Biopharma, and others, including Novartis Pharmaceuticals Corp., which supplied drugs and funded clinical aspects of the study.
SOURCE: Amaria et al. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9
For patients with surgically resectable, BRAF-mutated melanoma, neoadjuvant and adjuvant treatment with the combination of dabrafenib and trametinib resulted in significantly longer event-free survival compared with standard care, according to results of a randomized study.
The trial was closed early because of the “large difference” in event-free survival favoring the neoadjuvant approach, the authors wrote (Lancet Oncol. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9.
While early closure limits interpretation of results, they do provide important proof-of-concept and data for future studies, wrote the authors, led by Rodabe N Amaria, MD, of the department of medical oncology, University of Texas MD Anderson Cancer Center, Houston.
“The clinical and translational results strongly support the rationale for further assessment of neoadjuvant therapy in patients with high-risk, surgically resectable melanoma,” Dr. Amaria and colleagues said in the report on the randomized trial, believed to be the first to evaluate the role of neoadjuvant therapy versus standard care in BRAF-mutated melanoma.
Dabrafenib and trametinib combination therapy is approved as a treatment for patients with unresectable or metastatic stage IV melanoma and a BRAFV600 mutation, which is found in about half of cutaneous melanomas, authors said.
To evaluate the combination in earlier stage disease, Dr. Amaria and coinvestigators conducted a single-center, open-label, randomized, phase 2 trial of 21 patients with surgically resectable clinical stage III or oligometastatic stage IV melanoma with BRAFV600E or BRAFV600K mutations.
Patients were randomized 2:1 to receive the neoadjuvant/adjuvant treatment or to standard of care, which consisted of standard surgery plus consideration for adjuvant therapy, the authors said. Those patients assigned to the targeted therapy arm received 8 weeks of neoadjuvant dabrafenib and trametinib followed by surgery, then adjuvant dabrafenib and trametinib for up to 44 weeks.
Event-free survival, the primary endpoint of the trial, was a median of 19.7 months for neoadjuvant plus adjuvant dabrafenib and trametinib, versus 2.9 months for standard care (P less than .0001), the investigators reported.
Dabrafenib and trametinib combination therapy was well tolerated as neoadjuvant and adjuvant therapy, with no grade 4 adverse events or treatment related deaths, according to the investigators. The most common grade 3 adverse event seen with the combination was diarrhea, occurring in 2 patients (15%).
The trial is continuing as a single-arm study of neoadjuvant plus adjuvant dabrafenib and trametinib.
Dr. Amaria and colleagues reported individual disclosures related to Merck, Bristol-Myers Squibb, Array Biopharma, and others, including Novartis Pharmaceuticals Corp., which supplied drugs and funded clinical aspects of the study.
SOURCE: Amaria et al. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9
FROM THE LANCET ONCOLOGY
Key clinical point: In patients with high-risk, surgically resectable, clinical stage III-IV melanoma, dabrafenib and trametinib given in both the neoadjuvant and adjuvant setting improved event-free survival compared with standard care.
Major finding: Median event-free survival was 19.7 months for neoadjuvant/adjuvant dabrafenib and trametinib versus 2.9 months for standard upfront surgery including consideration for standard adjuvant therapy (P less than .0001).
Data source: A single-center, open-label, randomized, phase 2 trial including 21 patients with surgically resectable clinical stage III or oligometastatic stage IV BRAF-mutated melanoma.
Disclosures: Investigators reported ties to Novartis Pharmaceuticals Corp., which supplied drugs and funded clinical aspects of the study, and disclosures related to Merck, Bristol-Myers Squibb, Array Biopharma, and others.
Source: Amaria et al. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9.
Gene therapy moves from promise to reality
After decades of hype, dashed hopes, and setbacks, gene therapy has finally arrived and is poised to transform the treatment paradigm for many diseases, according to Cynthia E. Dunbar, MD, senior investigator at the Hematology Branch of the National Heart, Lung, and Blood Institute (NHLBI), part of the National Institutes of Health.
Hematologists can expect more developments that build on current successes with chimeric antigen receptor (CAR) T-cell therapy and gene therapy advances for hemophilia, as well as emerging advances in gene editing techniques including the CRISPR/Cas9 approach, Dr. Dunbar said in an interview.
That’s on top of a small number of regulatory approvals in the United States and Europe, she said. “Along with that, there’s a lot of interest and now involvement from biotechnology companies and even large pharmaceutical companies. I think all those factors really have to come together to create this kind of acceleration, and I’ve never seen anything like this previously.”
Dr. Dunbar – a former editor in chief of the journal Blood – and her colleagues recently published a review of current developments and emerging gene therapy technologies in the journal Science (2018 Jan 12. doi: 10.1126/science.aan4672).
“We really felt it was the right time to write the article,” she said.
Milestones
A new approach to cancer treatment was ushered in on Aug. 30, 2017, with the Food and Drug Administration approval of tisagenlecleucel, the first-ever gene therapy available in the United States. The CD19-directed CAR T-cell therapy is indicated for treatment of certain pediatric or young adult patients with B-cell precursor acute lymphoblastic leukemia that is refractory or in second or later relapse.
Soon afterward, FDA approved another CD19-directed CAR T-cell therapy, axicabtagene ciloleucel, for adult patients with large B-cell lymphoma after two or more lines of systemic therapy.
“It’s a very interesting time for immunotherapies in general,” Dr. Dunbar said. “There’s a huge number of options in terms of PD-1 inhibitors and other pharmacologics or antibodies that allow the patient’s own immune system to attack tumors. CAR T-cell therapy is an obvious step beyond that, in terms of arming your own T cells to very specifically target tumor cells.”
But randomized trials or meta-analyses may be necessary to determine the place of CAR T-cell therapy in the treatment armamentarium for acute lymphoblastic leukemia and large B-cell lymphoma given their cost and the availability of other therapeutic options, Dr. Dunbar suggested.
“Gene therapies have a large upfront cost, but if they’re truly curative and a one-time treatment, then they may in the long run be much cheaper than doing failed multiple transplants or needing monoclonal antibody infusion every 2 weeks for the rest of your life,” she said.
Another major success story still in the works, according to Dr. Dunbar, is the treatment of hemophilia A and B with gene therapy approaches. The positive data include a recent report showing that transgene-derived factor IX coagulant activity allowed for the termination of baseline prophylaxis, and the near elimination of bleeding and factor use, in patients with hemophilia B (N Engl J Med. 2017 Dec 7;377[23]:2215-27).
While gene therapy for hemophilia A has been more challenging, another recent report nevertheless demonstrated sustained normalization of factor VIII activity level with a single intravenous infusion of adeno-associated virus serotype 5 vector encoding a B-domain–deleted human factor VIII (N Engl J Med. 2017 Dec 28;377[26]:2519-30).
“The proof-of-principle was already there in hemophilia B,” Dr. Dunbar said. “It really was just a question of figuring out a way to package and deliver a Factor VIII that would work in the constraints of an AAV [adeno-associated virus] vector.”
Meanwhile, myeloma trials of CAR T-cell therapy seem very promising so far, but the challenge in that disease could be finding a place for gene therapy in a “much more diverse treatment landscape” that includes multiple effective regimens, according to Dr. Dunbar.
Future trends, challenges
Looking forward, she said.
Notably, genome editing approaches to treat sickle cell anemia are likely to move forward in the near future, according to Dr. Dunbar, following reports validating an erythroid enhancer of human BCL11A as a target for reinduction of fetal hemoglobin (Nature. 2015 Nov 12;527[7577]:192-7).
But all of this gene therapy development creates an educational challenge for frontline clinicians, even if the administration of CAR T-cell therapy and other advanced treatments is limited to highly specialized centers.
“There’s a lot of training that needs to go on with hematologists, oncologists, and other doctors about how to care for these patients after these treatments, in terms of what to look for and how to intervene early to prevent, for instance, severe toxicity from cytokine release syndrome,” Dr. Dunbar said.
Dr. Dunbar reported having no relevant financial disclosures.
After decades of hype, dashed hopes, and setbacks, gene therapy has finally arrived and is poised to transform the treatment paradigm for many diseases, according to Cynthia E. Dunbar, MD, senior investigator at the Hematology Branch of the National Heart, Lung, and Blood Institute (NHLBI), part of the National Institutes of Health.
Hematologists can expect more developments that build on current successes with chimeric antigen receptor (CAR) T-cell therapy and gene therapy advances for hemophilia, as well as emerging advances in gene editing techniques including the CRISPR/Cas9 approach, Dr. Dunbar said in an interview.
That’s on top of a small number of regulatory approvals in the United States and Europe, she said. “Along with that, there’s a lot of interest and now involvement from biotechnology companies and even large pharmaceutical companies. I think all those factors really have to come together to create this kind of acceleration, and I’ve never seen anything like this previously.”
Dr. Dunbar – a former editor in chief of the journal Blood – and her colleagues recently published a review of current developments and emerging gene therapy technologies in the journal Science (2018 Jan 12. doi: 10.1126/science.aan4672).
“We really felt it was the right time to write the article,” she said.
Milestones
A new approach to cancer treatment was ushered in on Aug. 30, 2017, with the Food and Drug Administration approval of tisagenlecleucel, the first-ever gene therapy available in the United States. The CD19-directed CAR T-cell therapy is indicated for treatment of certain pediatric or young adult patients with B-cell precursor acute lymphoblastic leukemia that is refractory or in second or later relapse.
Soon afterward, FDA approved another CD19-directed CAR T-cell therapy, axicabtagene ciloleucel, for adult patients with large B-cell lymphoma after two or more lines of systemic therapy.
“It’s a very interesting time for immunotherapies in general,” Dr. Dunbar said. “There’s a huge number of options in terms of PD-1 inhibitors and other pharmacologics or antibodies that allow the patient’s own immune system to attack tumors. CAR T-cell therapy is an obvious step beyond that, in terms of arming your own T cells to very specifically target tumor cells.”
But randomized trials or meta-analyses may be necessary to determine the place of CAR T-cell therapy in the treatment armamentarium for acute lymphoblastic leukemia and large B-cell lymphoma given their cost and the availability of other therapeutic options, Dr. Dunbar suggested.
“Gene therapies have a large upfront cost, but if they’re truly curative and a one-time treatment, then they may in the long run be much cheaper than doing failed multiple transplants or needing monoclonal antibody infusion every 2 weeks for the rest of your life,” she said.
Another major success story still in the works, according to Dr. Dunbar, is the treatment of hemophilia A and B with gene therapy approaches. The positive data include a recent report showing that transgene-derived factor IX coagulant activity allowed for the termination of baseline prophylaxis, and the near elimination of bleeding and factor use, in patients with hemophilia B (N Engl J Med. 2017 Dec 7;377[23]:2215-27).
While gene therapy for hemophilia A has been more challenging, another recent report nevertheless demonstrated sustained normalization of factor VIII activity level with a single intravenous infusion of adeno-associated virus serotype 5 vector encoding a B-domain–deleted human factor VIII (N Engl J Med. 2017 Dec 28;377[26]:2519-30).
“The proof-of-principle was already there in hemophilia B,” Dr. Dunbar said. “It really was just a question of figuring out a way to package and deliver a Factor VIII that would work in the constraints of an AAV [adeno-associated virus] vector.”
Meanwhile, myeloma trials of CAR T-cell therapy seem very promising so far, but the challenge in that disease could be finding a place for gene therapy in a “much more diverse treatment landscape” that includes multiple effective regimens, according to Dr. Dunbar.
Future trends, challenges
Looking forward, she said.
Notably, genome editing approaches to treat sickle cell anemia are likely to move forward in the near future, according to Dr. Dunbar, following reports validating an erythroid enhancer of human BCL11A as a target for reinduction of fetal hemoglobin (Nature. 2015 Nov 12;527[7577]:192-7).
But all of this gene therapy development creates an educational challenge for frontline clinicians, even if the administration of CAR T-cell therapy and other advanced treatments is limited to highly specialized centers.
“There’s a lot of training that needs to go on with hematologists, oncologists, and other doctors about how to care for these patients after these treatments, in terms of what to look for and how to intervene early to prevent, for instance, severe toxicity from cytokine release syndrome,” Dr. Dunbar said.
Dr. Dunbar reported having no relevant financial disclosures.
After decades of hype, dashed hopes, and setbacks, gene therapy has finally arrived and is poised to transform the treatment paradigm for many diseases, according to Cynthia E. Dunbar, MD, senior investigator at the Hematology Branch of the National Heart, Lung, and Blood Institute (NHLBI), part of the National Institutes of Health.
Hematologists can expect more developments that build on current successes with chimeric antigen receptor (CAR) T-cell therapy and gene therapy advances for hemophilia, as well as emerging advances in gene editing techniques including the CRISPR/Cas9 approach, Dr. Dunbar said in an interview.
That’s on top of a small number of regulatory approvals in the United States and Europe, she said. “Along with that, there’s a lot of interest and now involvement from biotechnology companies and even large pharmaceutical companies. I think all those factors really have to come together to create this kind of acceleration, and I’ve never seen anything like this previously.”
Dr. Dunbar – a former editor in chief of the journal Blood – and her colleagues recently published a review of current developments and emerging gene therapy technologies in the journal Science (2018 Jan 12. doi: 10.1126/science.aan4672).
“We really felt it was the right time to write the article,” she said.
Milestones
A new approach to cancer treatment was ushered in on Aug. 30, 2017, with the Food and Drug Administration approval of tisagenlecleucel, the first-ever gene therapy available in the United States. The CD19-directed CAR T-cell therapy is indicated for treatment of certain pediatric or young adult patients with B-cell precursor acute lymphoblastic leukemia that is refractory or in second or later relapse.
Soon afterward, FDA approved another CD19-directed CAR T-cell therapy, axicabtagene ciloleucel, for adult patients with large B-cell lymphoma after two or more lines of systemic therapy.
“It’s a very interesting time for immunotherapies in general,” Dr. Dunbar said. “There’s a huge number of options in terms of PD-1 inhibitors and other pharmacologics or antibodies that allow the patient’s own immune system to attack tumors. CAR T-cell therapy is an obvious step beyond that, in terms of arming your own T cells to very specifically target tumor cells.”
But randomized trials or meta-analyses may be necessary to determine the place of CAR T-cell therapy in the treatment armamentarium for acute lymphoblastic leukemia and large B-cell lymphoma given their cost and the availability of other therapeutic options, Dr. Dunbar suggested.
“Gene therapies have a large upfront cost, but if they’re truly curative and a one-time treatment, then they may in the long run be much cheaper than doing failed multiple transplants or needing monoclonal antibody infusion every 2 weeks for the rest of your life,” she said.
Another major success story still in the works, according to Dr. Dunbar, is the treatment of hemophilia A and B with gene therapy approaches. The positive data include a recent report showing that transgene-derived factor IX coagulant activity allowed for the termination of baseline prophylaxis, and the near elimination of bleeding and factor use, in patients with hemophilia B (N Engl J Med. 2017 Dec 7;377[23]:2215-27).
While gene therapy for hemophilia A has been more challenging, another recent report nevertheless demonstrated sustained normalization of factor VIII activity level with a single intravenous infusion of adeno-associated virus serotype 5 vector encoding a B-domain–deleted human factor VIII (N Engl J Med. 2017 Dec 28;377[26]:2519-30).
“The proof-of-principle was already there in hemophilia B,” Dr. Dunbar said. “It really was just a question of figuring out a way to package and deliver a Factor VIII that would work in the constraints of an AAV [adeno-associated virus] vector.”
Meanwhile, myeloma trials of CAR T-cell therapy seem very promising so far, but the challenge in that disease could be finding a place for gene therapy in a “much more diverse treatment landscape” that includes multiple effective regimens, according to Dr. Dunbar.
Future trends, challenges
Looking forward, she said.
Notably, genome editing approaches to treat sickle cell anemia are likely to move forward in the near future, according to Dr. Dunbar, following reports validating an erythroid enhancer of human BCL11A as a target for reinduction of fetal hemoglobin (Nature. 2015 Nov 12;527[7577]:192-7).
But all of this gene therapy development creates an educational challenge for frontline clinicians, even if the administration of CAR T-cell therapy and other advanced treatments is limited to highly specialized centers.
“There’s a lot of training that needs to go on with hematologists, oncologists, and other doctors about how to care for these patients after these treatments, in terms of what to look for and how to intervene early to prevent, for instance, severe toxicity from cytokine release syndrome,” Dr. Dunbar said.
Dr. Dunbar reported having no relevant financial disclosures.
Immune-modified RECIST can help identify survival benefit from cancer immunotherapy
Cancer immunotherapy-specific response criteria not only provide improved estimates of treatment response versus standard criteria, but may also better identify patients who achieve an overall survival benefit from therapy.
Compared to standard Response Evaluation Criteria In Solid Tumors (RECIST) v1.1, the immune-modified RECIST provided a 1%-2% greater overall response and an 8%-13% greater rate of disease control, and added 0.5-1.5 months to median progression-free survival among patients treated with the PD-L1 inhibitor atezolizumab, according to analyses of different phase 1 and 2 trials.
In addition, overall survival (OS) benefit in some of the trials could be better delineated using the immune-modified criteria, which account for unique patterns of progression sometimes experienced by patients on cancer immunotherapy, noted the study authors. The report was published in the Journal of Clinical Oncology.
Using immune-specific criteria to evaluate response to cancer immunotherapy is not a new concept. However, there are only limited data on how those criteria might apply to predictions of OS, according to lead author F. Stephen Hodi, MD, of Dana-Farber Cancer Institute, Boston, and his coauthors.
“These analyses reveal aspects of immune-modified RECIST that seem to predict OS better than RECIST v1.1, and aspects needing refinement to improve the ability to predict clinical benefit,” wrote Dr. Hodi and his colleagues.
Typical response criteria may not adequately predict the potential OS benefit of cancer immunotherapy, since patients receiving cancer immunotherapy may exhibit response patterns outside of the “classic response patterns” seen with other anticancer treatments, they noted.
In particular, some patients may experience an initial transient increase in tumor burden before responding, while in other cases, patients with responding baseline lesions might develop new lesions.
Immune-modified criteria have been developed to account for those “other patterns” that can manifest with cancer immunotherapy, the authors said.
Dr. Hodi and his colleagues sought to evaluate outcomes by RECIST vs. immune-related RECIST criteria among patients treated with atezolizumab in studies of non–small-cell lung cancer (NSCLC) and metastatic urothelial carcinoma.
In the phase 2 BIRCH study of first-line atezolizumab for NSCLC, they found that immune-related RECIST criteria appeared to predict OS better than RECIST. Median overall survival was 4.0 months longer among patients who had progressive disease (PD) by RECIST criteria within 90 days of study enrollment, versus patients who had PD by both RECIST and immune-modified RECIST at that time point, Dr. Hodi and his colleagues reported.
In the POPLAR trial of atezolizumab in NSCLC, median overall survival was 1.4 months longer for patients with PD by RECIST vs. patients with PD by both RECIST and immune-modified RECIST at 90 days, they reported.
For patients with metastatic urothelial bladder cancer treated with atezolizumab in the IMvigor210 study, median overall survival was 4.4 months longer for patients with PD by RECIST only vs. PD by both RECIST and immune-related RECIST within 180 days of enrollment, the researchers noted.
An international effort is underway to compare data sets from larger trial sets and multiple cancer immunotherapy agents, they wrote.
SOURCE: Hodi FS et al., J Clin Oncol. 2018 Jan 17. doi: 10.1200/JCO.2017.75.1644
Cancer immunotherapy-specific response criteria not only provide improved estimates of treatment response versus standard criteria, but may also better identify patients who achieve an overall survival benefit from therapy.
Compared to standard Response Evaluation Criteria In Solid Tumors (RECIST) v1.1, the immune-modified RECIST provided a 1%-2% greater overall response and an 8%-13% greater rate of disease control, and added 0.5-1.5 months to median progression-free survival among patients treated with the PD-L1 inhibitor atezolizumab, according to analyses of different phase 1 and 2 trials.
In addition, overall survival (OS) benefit in some of the trials could be better delineated using the immune-modified criteria, which account for unique patterns of progression sometimes experienced by patients on cancer immunotherapy, noted the study authors. The report was published in the Journal of Clinical Oncology.
Using immune-specific criteria to evaluate response to cancer immunotherapy is not a new concept. However, there are only limited data on how those criteria might apply to predictions of OS, according to lead author F. Stephen Hodi, MD, of Dana-Farber Cancer Institute, Boston, and his coauthors.
“These analyses reveal aspects of immune-modified RECIST that seem to predict OS better than RECIST v1.1, and aspects needing refinement to improve the ability to predict clinical benefit,” wrote Dr. Hodi and his colleagues.
Typical response criteria may not adequately predict the potential OS benefit of cancer immunotherapy, since patients receiving cancer immunotherapy may exhibit response patterns outside of the “classic response patterns” seen with other anticancer treatments, they noted.
In particular, some patients may experience an initial transient increase in tumor burden before responding, while in other cases, patients with responding baseline lesions might develop new lesions.
Immune-modified criteria have been developed to account for those “other patterns” that can manifest with cancer immunotherapy, the authors said.
Dr. Hodi and his colleagues sought to evaluate outcomes by RECIST vs. immune-related RECIST criteria among patients treated with atezolizumab in studies of non–small-cell lung cancer (NSCLC) and metastatic urothelial carcinoma.
In the phase 2 BIRCH study of first-line atezolizumab for NSCLC, they found that immune-related RECIST criteria appeared to predict OS better than RECIST. Median overall survival was 4.0 months longer among patients who had progressive disease (PD) by RECIST criteria within 90 days of study enrollment, versus patients who had PD by both RECIST and immune-modified RECIST at that time point, Dr. Hodi and his colleagues reported.
In the POPLAR trial of atezolizumab in NSCLC, median overall survival was 1.4 months longer for patients with PD by RECIST vs. patients with PD by both RECIST and immune-modified RECIST at 90 days, they reported.
For patients with metastatic urothelial bladder cancer treated with atezolizumab in the IMvigor210 study, median overall survival was 4.4 months longer for patients with PD by RECIST only vs. PD by both RECIST and immune-related RECIST within 180 days of enrollment, the researchers noted.
An international effort is underway to compare data sets from larger trial sets and multiple cancer immunotherapy agents, they wrote.
SOURCE: Hodi FS et al., J Clin Oncol. 2018 Jan 17. doi: 10.1200/JCO.2017.75.1644
Cancer immunotherapy-specific response criteria not only provide improved estimates of treatment response versus standard criteria, but may also better identify patients who achieve an overall survival benefit from therapy.
Compared to standard Response Evaluation Criteria In Solid Tumors (RECIST) v1.1, the immune-modified RECIST provided a 1%-2% greater overall response and an 8%-13% greater rate of disease control, and added 0.5-1.5 months to median progression-free survival among patients treated with the PD-L1 inhibitor atezolizumab, according to analyses of different phase 1 and 2 trials.
In addition, overall survival (OS) benefit in some of the trials could be better delineated using the immune-modified criteria, which account for unique patterns of progression sometimes experienced by patients on cancer immunotherapy, noted the study authors. The report was published in the Journal of Clinical Oncology.
Using immune-specific criteria to evaluate response to cancer immunotherapy is not a new concept. However, there are only limited data on how those criteria might apply to predictions of OS, according to lead author F. Stephen Hodi, MD, of Dana-Farber Cancer Institute, Boston, and his coauthors.
“These analyses reveal aspects of immune-modified RECIST that seem to predict OS better than RECIST v1.1, and aspects needing refinement to improve the ability to predict clinical benefit,” wrote Dr. Hodi and his colleagues.
Typical response criteria may not adequately predict the potential OS benefit of cancer immunotherapy, since patients receiving cancer immunotherapy may exhibit response patterns outside of the “classic response patterns” seen with other anticancer treatments, they noted.
In particular, some patients may experience an initial transient increase in tumor burden before responding, while in other cases, patients with responding baseline lesions might develop new lesions.
Immune-modified criteria have been developed to account for those “other patterns” that can manifest with cancer immunotherapy, the authors said.
Dr. Hodi and his colleagues sought to evaluate outcomes by RECIST vs. immune-related RECIST criteria among patients treated with atezolizumab in studies of non–small-cell lung cancer (NSCLC) and metastatic urothelial carcinoma.
In the phase 2 BIRCH study of first-line atezolizumab for NSCLC, they found that immune-related RECIST criteria appeared to predict OS better than RECIST. Median overall survival was 4.0 months longer among patients who had progressive disease (PD) by RECIST criteria within 90 days of study enrollment, versus patients who had PD by both RECIST and immune-modified RECIST at that time point, Dr. Hodi and his colleagues reported.
In the POPLAR trial of atezolizumab in NSCLC, median overall survival was 1.4 months longer for patients with PD by RECIST vs. patients with PD by both RECIST and immune-modified RECIST at 90 days, they reported.
For patients with metastatic urothelial bladder cancer treated with atezolizumab in the IMvigor210 study, median overall survival was 4.4 months longer for patients with PD by RECIST only vs. PD by both RECIST and immune-related RECIST within 180 days of enrollment, the researchers noted.
An international effort is underway to compare data sets from larger trial sets and multiple cancer immunotherapy agents, they wrote.
SOURCE: Hodi FS et al., J Clin Oncol. 2018 Jan 17. doi: 10.1200/JCO.2017.75.1644
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Compared to standard criteria for response evaluation, criteria developed specifically to evaluate response to cancer immunotherapy better identified patients with an overall survival (OS) benefit.
Major finding: Median OS was 4.0 and 1.4 months longer, respectively, in the BIRCH and POPLAR non–small-cell lung cancer trial among patients who had progressive disease (PD) by standard criteria only, as opposed to patients who also had PD according to the immunotherapy-specific response criteria.
Data source: Analysis of patients treated with single-agent atezolizumab in phase 1 and 2 clinical trials.
Disclosures: The study was supported by F. Hoffmann-La Roche. Authors reported disclosures related to Merck Sharp & Dohme, Novartis, Genentech/Roche, Bristol-Myers Squibb, and others.
Source: Hodi FS et al., J Clin Oncol. 2018 Jan 17. doi: 10.1200/JCO.2017.75.1644.
Thrombosis risk is elevated with myeloproliferative neoplasms
Patients with myeloproliferative neoplasms (MPNs) have a higher rate of arterial and venous thrombosis than does the general population, with the greatest risk occurring around the time of diagnosis, according to results of a retrospective study.
Hazard ratios at 3 months after diagnosis were 3.0 (95% CI, 2.7-3.4) for arterial thrombosis and 9.7 (95% CI, 7.8-12.0) for venous thrombosis, compared with matched controls, Malin Hultcrantz, MD, PhD, of the Karolinska University Hospital, Stockholm, and her coauthors reported in the Annals of Internal Medicine.
Although previous studies have suggested patients with MPNs are at increased risk for thrombotic events, this large, population-based analysis is believed to be the first study to provide estimates of excess risk compared with matched control participants.
“These results are encouraging, and we believe that further refinement of risk scoring systems (such as by including time since MPN diagnosis and biomarkers); rethinking of recommendations for younger patients with MPNs; and emerging, more effective treatments will further improve outcomes for patients with MPNs,” the researchers wrote.
The retrospective, population-based cohort study included 9,429 Swedish patients diagnosed with MPNs between 1987 and 2009 and 35,820 matched control participants. Patient follow-up through 2010 was included in the analysis.
Thrombosis risk was highest near the time of diagnosis but decreased during the following year “likely because of effective thromboprophylactic and cytoreductive treatment of the MPN;”still, the risk remained elevated, the researchers wrote.
“This novel finding underlines the importance of initiating phlebotomy as well as thromboprophylactic and cytoreductive treatment, when indicated, as soon as the MPN is diagnosed,” they added.
Arterial thrombosis hazard ratios for MPN patients, compared with control participants, were 3.0 at 3 months after diagnosis, 2.0 at 1 year, and 1.5 at 5 years. Similarly, venous thrombosis hazard ratios were 9.7 at 3 months, 4.7 at 1 year, and 3.2 at 5 years.
Thrombosis risk was elevated in all age groups and all MPN subtypes, including primary myelofibrosis, polycythemia vera, and essential thrombocythemia. Of note, the study confirmed prior thrombosis and older age (60 years or older) as risk factors. Among patients with both of those risk factors, risk of thrombosis was increased 7-fold, according to the researchers.
Hazard ratios for thrombosis decreased during more recent time periods, suggesting a “positive effect” of improved treatment strategies, including increased use of aspirin as primary prophylaxis, better cardiovascular risk management, and better adherence to recommendations for cytoreductive treatment and phlebotomy, the researchers noted. Additionally, treatment with interferon and Janus kinase 2 inhibitors, such as ruxolitinib, “may be effective in further reducing risk for thrombosis,” the researchers wrote.
The study was funded by the Cancer Research Foundations of Radiumhemmet, the Swedish Research Council, and Memorial Sloan Kettering Cancer Center, among other sources. The researchers reported having no financial disclosures relevant to the study.
SOURCE: Hultcrantz M et al. Ann Intern Med. 2018. doi: 10.7326/M17-0028.
The most notable contribution of the large cohort study by Hultcrantz and her colleagues is quantification of the magnitude of thrombotic risk that MPNs confer, according to Alison R. Moliterno, MD, and Elizabeth V. Ratchford, MD.
“Hultcrantz and colleagues have opened our eyes to the magnitude of thrombotic risk MPNs bring to affected patients,” Dr. Moliterno and Dr. Ratchford wrote in an editorial in Annals of Internal Medicine. “Their study shows us that the traditional approach to assessing thrombotic risk in patients with MPNs [who are age 60 years and older, have prior thrombotic event, and have traditional cardiovascular risk factors] lacks precision and personalization.”
Both arterial and venous thrombotic events were increased throughout patients’ lifetimes, though the highest risk was around the time of MPN diagnosis. According to study results, 10% of patients had a thrombotic event in the 30 days before or after diagnosis.
“Patients and clinicians should be keenly aware of this particularly risky period, during which risk for thrombosis is similar to that in the month after a transient ischemic attack,” Dr. Moliterno and Dr. Ratchford wrote.
Unfortunately, the study did not include data on genomics, they noted. The acquired JAK2 V617F mutation, which drives MPN phenotypes, is associated with elevated inflammatory cytokines, and inflammation is a recognized risk factor for thrombosis, according to the editorial authors.
Alison R. Moliterno, MD, and Elizabeth V. Ratchford, MD, are with Johns Hopkins University, Baltimore. These comments are adapted from an accompanying editorial (Ann Intern Med. 2018. doi: 10.7326/M17-3153). The authors reported having no relevant conflicts related to the study.
The most notable contribution of the large cohort study by Hultcrantz and her colleagues is quantification of the magnitude of thrombotic risk that MPNs confer, according to Alison R. Moliterno, MD, and Elizabeth V. Ratchford, MD.
“Hultcrantz and colleagues have opened our eyes to the magnitude of thrombotic risk MPNs bring to affected patients,” Dr. Moliterno and Dr. Ratchford wrote in an editorial in Annals of Internal Medicine. “Their study shows us that the traditional approach to assessing thrombotic risk in patients with MPNs [who are age 60 years and older, have prior thrombotic event, and have traditional cardiovascular risk factors] lacks precision and personalization.”
Both arterial and venous thrombotic events were increased throughout patients’ lifetimes, though the highest risk was around the time of MPN diagnosis. According to study results, 10% of patients had a thrombotic event in the 30 days before or after diagnosis.
“Patients and clinicians should be keenly aware of this particularly risky period, during which risk for thrombosis is similar to that in the month after a transient ischemic attack,” Dr. Moliterno and Dr. Ratchford wrote.
Unfortunately, the study did not include data on genomics, they noted. The acquired JAK2 V617F mutation, which drives MPN phenotypes, is associated with elevated inflammatory cytokines, and inflammation is a recognized risk factor for thrombosis, according to the editorial authors.
Alison R. Moliterno, MD, and Elizabeth V. Ratchford, MD, are with Johns Hopkins University, Baltimore. These comments are adapted from an accompanying editorial (Ann Intern Med. 2018. doi: 10.7326/M17-3153). The authors reported having no relevant conflicts related to the study.
The most notable contribution of the large cohort study by Hultcrantz and her colleagues is quantification of the magnitude of thrombotic risk that MPNs confer, according to Alison R. Moliterno, MD, and Elizabeth V. Ratchford, MD.
“Hultcrantz and colleagues have opened our eyes to the magnitude of thrombotic risk MPNs bring to affected patients,” Dr. Moliterno and Dr. Ratchford wrote in an editorial in Annals of Internal Medicine. “Their study shows us that the traditional approach to assessing thrombotic risk in patients with MPNs [who are age 60 years and older, have prior thrombotic event, and have traditional cardiovascular risk factors] lacks precision and personalization.”
Both arterial and venous thrombotic events were increased throughout patients’ lifetimes, though the highest risk was around the time of MPN diagnosis. According to study results, 10% of patients had a thrombotic event in the 30 days before or after diagnosis.
“Patients and clinicians should be keenly aware of this particularly risky period, during which risk for thrombosis is similar to that in the month after a transient ischemic attack,” Dr. Moliterno and Dr. Ratchford wrote.
Unfortunately, the study did not include data on genomics, they noted. The acquired JAK2 V617F mutation, which drives MPN phenotypes, is associated with elevated inflammatory cytokines, and inflammation is a recognized risk factor for thrombosis, according to the editorial authors.
Alison R. Moliterno, MD, and Elizabeth V. Ratchford, MD, are with Johns Hopkins University, Baltimore. These comments are adapted from an accompanying editorial (Ann Intern Med. 2018. doi: 10.7326/M17-3153). The authors reported having no relevant conflicts related to the study.
Patients with myeloproliferative neoplasms (MPNs) have a higher rate of arterial and venous thrombosis than does the general population, with the greatest risk occurring around the time of diagnosis, according to results of a retrospective study.
Hazard ratios at 3 months after diagnosis were 3.0 (95% CI, 2.7-3.4) for arterial thrombosis and 9.7 (95% CI, 7.8-12.0) for venous thrombosis, compared with matched controls, Malin Hultcrantz, MD, PhD, of the Karolinska University Hospital, Stockholm, and her coauthors reported in the Annals of Internal Medicine.
Although previous studies have suggested patients with MPNs are at increased risk for thrombotic events, this large, population-based analysis is believed to be the first study to provide estimates of excess risk compared with matched control participants.
“These results are encouraging, and we believe that further refinement of risk scoring systems (such as by including time since MPN diagnosis and biomarkers); rethinking of recommendations for younger patients with MPNs; and emerging, more effective treatments will further improve outcomes for patients with MPNs,” the researchers wrote.
The retrospective, population-based cohort study included 9,429 Swedish patients diagnosed with MPNs between 1987 and 2009 and 35,820 matched control participants. Patient follow-up through 2010 was included in the analysis.
Thrombosis risk was highest near the time of diagnosis but decreased during the following year “likely because of effective thromboprophylactic and cytoreductive treatment of the MPN;”still, the risk remained elevated, the researchers wrote.
“This novel finding underlines the importance of initiating phlebotomy as well as thromboprophylactic and cytoreductive treatment, when indicated, as soon as the MPN is diagnosed,” they added.
Arterial thrombosis hazard ratios for MPN patients, compared with control participants, were 3.0 at 3 months after diagnosis, 2.0 at 1 year, and 1.5 at 5 years. Similarly, venous thrombosis hazard ratios were 9.7 at 3 months, 4.7 at 1 year, and 3.2 at 5 years.
Thrombosis risk was elevated in all age groups and all MPN subtypes, including primary myelofibrosis, polycythemia vera, and essential thrombocythemia. Of note, the study confirmed prior thrombosis and older age (60 years or older) as risk factors. Among patients with both of those risk factors, risk of thrombosis was increased 7-fold, according to the researchers.
Hazard ratios for thrombosis decreased during more recent time periods, suggesting a “positive effect” of improved treatment strategies, including increased use of aspirin as primary prophylaxis, better cardiovascular risk management, and better adherence to recommendations for cytoreductive treatment and phlebotomy, the researchers noted. Additionally, treatment with interferon and Janus kinase 2 inhibitors, such as ruxolitinib, “may be effective in further reducing risk for thrombosis,” the researchers wrote.
The study was funded by the Cancer Research Foundations of Radiumhemmet, the Swedish Research Council, and Memorial Sloan Kettering Cancer Center, among other sources. The researchers reported having no financial disclosures relevant to the study.
SOURCE: Hultcrantz M et al. Ann Intern Med. 2018. doi: 10.7326/M17-0028.
Patients with myeloproliferative neoplasms (MPNs) have a higher rate of arterial and venous thrombosis than does the general population, with the greatest risk occurring around the time of diagnosis, according to results of a retrospective study.
Hazard ratios at 3 months after diagnosis were 3.0 (95% CI, 2.7-3.4) for arterial thrombosis and 9.7 (95% CI, 7.8-12.0) for venous thrombosis, compared with matched controls, Malin Hultcrantz, MD, PhD, of the Karolinska University Hospital, Stockholm, and her coauthors reported in the Annals of Internal Medicine.
Although previous studies have suggested patients with MPNs are at increased risk for thrombotic events, this large, population-based analysis is believed to be the first study to provide estimates of excess risk compared with matched control participants.
“These results are encouraging, and we believe that further refinement of risk scoring systems (such as by including time since MPN diagnosis and biomarkers); rethinking of recommendations for younger patients with MPNs; and emerging, more effective treatments will further improve outcomes for patients with MPNs,” the researchers wrote.
The retrospective, population-based cohort study included 9,429 Swedish patients diagnosed with MPNs between 1987 and 2009 and 35,820 matched control participants. Patient follow-up through 2010 was included in the analysis.
Thrombosis risk was highest near the time of diagnosis but decreased during the following year “likely because of effective thromboprophylactic and cytoreductive treatment of the MPN;”still, the risk remained elevated, the researchers wrote.
“This novel finding underlines the importance of initiating phlebotomy as well as thromboprophylactic and cytoreductive treatment, when indicated, as soon as the MPN is diagnosed,” they added.
Arterial thrombosis hazard ratios for MPN patients, compared with control participants, were 3.0 at 3 months after diagnosis, 2.0 at 1 year, and 1.5 at 5 years. Similarly, venous thrombosis hazard ratios were 9.7 at 3 months, 4.7 at 1 year, and 3.2 at 5 years.
Thrombosis risk was elevated in all age groups and all MPN subtypes, including primary myelofibrosis, polycythemia vera, and essential thrombocythemia. Of note, the study confirmed prior thrombosis and older age (60 years or older) as risk factors. Among patients with both of those risk factors, risk of thrombosis was increased 7-fold, according to the researchers.
Hazard ratios for thrombosis decreased during more recent time periods, suggesting a “positive effect” of improved treatment strategies, including increased use of aspirin as primary prophylaxis, better cardiovascular risk management, and better adherence to recommendations for cytoreductive treatment and phlebotomy, the researchers noted. Additionally, treatment with interferon and Janus kinase 2 inhibitors, such as ruxolitinib, “may be effective in further reducing risk for thrombosis,” the researchers wrote.
The study was funded by the Cancer Research Foundations of Radiumhemmet, the Swedish Research Council, and Memorial Sloan Kettering Cancer Center, among other sources. The researchers reported having no financial disclosures relevant to the study.
SOURCE: Hultcrantz M et al. Ann Intern Med. 2018. doi: 10.7326/M17-0028.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point:
Major finding: Hazard ratios (HRs) at 3 months were 3.0 (95% confidence interval, 2.7-3.4) for arterial thrombosis and 9.7 (95% CI, 7.8-12.0) for venous thrombosis, compared with matched controls.
Study details: A Swedish retrospective, population-based study including 9,429 patients with MPNs and 35,820 matched control participants.
Disclosures: The study was funded by the Cancer Research Foundations of Radiumhemmet, the Swedish Research Council, and Memorial Sloan Kettering Cancer Center, among other sources. The researchers reported having no relevant financial disclosures.
Source: Hultcrantz M et al. Ann Intern Med. 2018. doi: 10.7326/M17-0028.
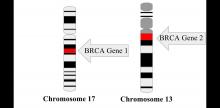
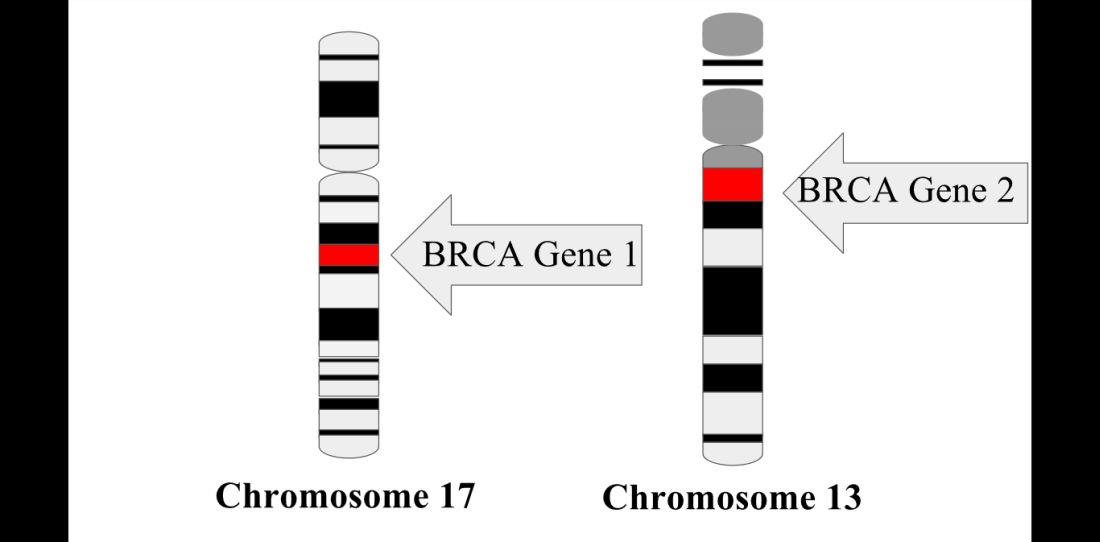
POSH study: BRCA mutations did not influence survival in young onset breast cancer
For women with young-onset breast cancer, presence of a BRCA mutation did not significantly impact survival, according to results of the Prospective Outcomes in Sporadic versus Hereditary breast cancer (POSH) study.
BRCA-positive and BRCA-negative women had similar overall survival at 2 years, 5 years, and 10 years after diagnosis, according to lead author Ellen R. Copson, MD, a senior lecturer in medical oncology in the cancer sciences division, University of Southampton (England) and her study coauthors.
Currently, young women with breast cancer and a BRCA mutation frequently are offered bilateral mastectomy, the authors noted.
The prospective cohort study by Dr. Copson and her colleagues included 2,733 women with breast cancer who were aged 40 years or younger at first diagnosis. Of those patients, 338 (12%) had either a BRCA1 or BRCA2 mutation, according to investigators.
At 2 years after diagnosis, overall survival was 97.0% and 96.6% for BRCA-positive and BRCA-negative patients, respectively, the report said. Similarly, overall survival was 83.8% and 85.0% for the two groups at 5 years after diagnosis, and 73.4% vs. 70.1% at 10 years.
Multivariable analysis accounting for known prognostic factors including ethnicity and body mass index showed there was no significant difference between groups (hazard ratio, 0.96; 95% confidence interval, 0.76-1.22; P = .76), the authors wrote.
Triple-negative breast cancer patients with a BRCA mutation might have a survival advantage in the first few years following diagnosis,compared with non-BRCA carriers, the POSH study also found. Researchers reported a significant difference at 2 years (95% for BRCA-positive vs. 91% for BRCA-negative patients; P = .047), but there was no significant difference between arms at 5 or 10 years.
POSH is believed to be the largest prospective cohort study to compare breast cancer outcomes for patients with BRCA mutations to those with sporadic breast cancer. Previous studies, primarily retrospective, have suggested “better, worse, or similar outcomes” for BRCA-positive versus BRCA-negative patients, the authors wrote. Dr. Copson reported receiving honoraria from Roche, while her coauthors reported honoraria from GSK, Pfizer, AstraZeneca, and Pierre Fabre. Funding for the study was provided by the Wessex Cancer Trust, Cancer Research UK, and Breast Cancer Now.
SOURCE: Copson et al. Lancet Oncol. 2018 Jan 11 doi: 10.1016/S1470-2045(17)30891-4.
The POSH prospective cohort study, which showed no significant difference in survival for BRCA-positive versus BRCA-negative young onset breast cancer patients, has contributed to the understanding of this patient population, providing “comprehensive data about patient, tumor, and treatment characteristics, along with extensive follow-up data,” wrote Peter A. Fasching, MD, in accompanying editorial.
“Understanding prognosis in young patients is important because patients with BRCA mutations are at increased risk of developing specific conditions, such as secondary cancers,” Dr. Fasching said. “These risks determine treatment, and knowing that BRCA1 or BRCA2 mutations do not result in a different prognosis might change the therapeutic approach for these risks.”
Moreover, in retrospective analyses, bilateral mastectomy conferred an overall survival benefit for BRCA mutation carriers: “This important topic needs more prospective research, as preventive surgical measures might have an effect on what might be a very long life after a diagnosis of breast cancer at a young age,” said Dr. Fasching. “The data from POSH deliver a rationale for prospective studies to address these questions.”
Dr. Peter A. Fasching is with Friedrich-Alexander University Erlangen-Nuremberg, Germany. These comments are based on his editorial appearing in Lancet Oncology (2018 Jan 11. doi: 10.1016/S1470-2045(18)30008-1). Dr. Fasching declared grants from Novartis, along with personal fees from Novartis, Pfizer, Roche, Teva, and Amgen.
The POSH prospective cohort study, which showed no significant difference in survival for BRCA-positive versus BRCA-negative young onset breast cancer patients, has contributed to the understanding of this patient population, providing “comprehensive data about patient, tumor, and treatment characteristics, along with extensive follow-up data,” wrote Peter A. Fasching, MD, in accompanying editorial.
“Understanding prognosis in young patients is important because patients with BRCA mutations are at increased risk of developing specific conditions, such as secondary cancers,” Dr. Fasching said. “These risks determine treatment, and knowing that BRCA1 or BRCA2 mutations do not result in a different prognosis might change the therapeutic approach for these risks.”
Moreover, in retrospective analyses, bilateral mastectomy conferred an overall survival benefit for BRCA mutation carriers: “This important topic needs more prospective research, as preventive surgical measures might have an effect on what might be a very long life after a diagnosis of breast cancer at a young age,” said Dr. Fasching. “The data from POSH deliver a rationale for prospective studies to address these questions.”
Dr. Peter A. Fasching is with Friedrich-Alexander University Erlangen-Nuremberg, Germany. These comments are based on his editorial appearing in Lancet Oncology (2018 Jan 11. doi: 10.1016/S1470-2045(18)30008-1). Dr. Fasching declared grants from Novartis, along with personal fees from Novartis, Pfizer, Roche, Teva, and Amgen.
The POSH prospective cohort study, which showed no significant difference in survival for BRCA-positive versus BRCA-negative young onset breast cancer patients, has contributed to the understanding of this patient population, providing “comprehensive data about patient, tumor, and treatment characteristics, along with extensive follow-up data,” wrote Peter A. Fasching, MD, in accompanying editorial.
“Understanding prognosis in young patients is important because patients with BRCA mutations are at increased risk of developing specific conditions, such as secondary cancers,” Dr. Fasching said. “These risks determine treatment, and knowing that BRCA1 or BRCA2 mutations do not result in a different prognosis might change the therapeutic approach for these risks.”
Moreover, in retrospective analyses, bilateral mastectomy conferred an overall survival benefit for BRCA mutation carriers: “This important topic needs more prospective research, as preventive surgical measures might have an effect on what might be a very long life after a diagnosis of breast cancer at a young age,” said Dr. Fasching. “The data from POSH deliver a rationale for prospective studies to address these questions.”
Dr. Peter A. Fasching is with Friedrich-Alexander University Erlangen-Nuremberg, Germany. These comments are based on his editorial appearing in Lancet Oncology (2018 Jan 11. doi: 10.1016/S1470-2045(18)30008-1). Dr. Fasching declared grants from Novartis, along with personal fees from Novartis, Pfizer, Roche, Teva, and Amgen.
For women with young-onset breast cancer, presence of a BRCA mutation did not significantly impact survival, according to results of the Prospective Outcomes in Sporadic versus Hereditary breast cancer (POSH) study.
BRCA-positive and BRCA-negative women had similar overall survival at 2 years, 5 years, and 10 years after diagnosis, according to lead author Ellen R. Copson, MD, a senior lecturer in medical oncology in the cancer sciences division, University of Southampton (England) and her study coauthors.
Currently, young women with breast cancer and a BRCA mutation frequently are offered bilateral mastectomy, the authors noted.
The prospective cohort study by Dr. Copson and her colleagues included 2,733 women with breast cancer who were aged 40 years or younger at first diagnosis. Of those patients, 338 (12%) had either a BRCA1 or BRCA2 mutation, according to investigators.
At 2 years after diagnosis, overall survival was 97.0% and 96.6% for BRCA-positive and BRCA-negative patients, respectively, the report said. Similarly, overall survival was 83.8% and 85.0% for the two groups at 5 years after diagnosis, and 73.4% vs. 70.1% at 10 years.
Multivariable analysis accounting for known prognostic factors including ethnicity and body mass index showed there was no significant difference between groups (hazard ratio, 0.96; 95% confidence interval, 0.76-1.22; P = .76), the authors wrote.
Triple-negative breast cancer patients with a BRCA mutation might have a survival advantage in the first few years following diagnosis,compared with non-BRCA carriers, the POSH study also found. Researchers reported a significant difference at 2 years (95% for BRCA-positive vs. 91% for BRCA-negative patients; P = .047), but there was no significant difference between arms at 5 or 10 years.
POSH is believed to be the largest prospective cohort study to compare breast cancer outcomes for patients with BRCA mutations to those with sporadic breast cancer. Previous studies, primarily retrospective, have suggested “better, worse, or similar outcomes” for BRCA-positive versus BRCA-negative patients, the authors wrote. Dr. Copson reported receiving honoraria from Roche, while her coauthors reported honoraria from GSK, Pfizer, AstraZeneca, and Pierre Fabre. Funding for the study was provided by the Wessex Cancer Trust, Cancer Research UK, and Breast Cancer Now.
SOURCE: Copson et al. Lancet Oncol. 2018 Jan 11 doi: 10.1016/S1470-2045(17)30891-4.
For women with young-onset breast cancer, presence of a BRCA mutation did not significantly impact survival, according to results of the Prospective Outcomes in Sporadic versus Hereditary breast cancer (POSH) study.
BRCA-positive and BRCA-negative women had similar overall survival at 2 years, 5 years, and 10 years after diagnosis, according to lead author Ellen R. Copson, MD, a senior lecturer in medical oncology in the cancer sciences division, University of Southampton (England) and her study coauthors.
Currently, young women with breast cancer and a BRCA mutation frequently are offered bilateral mastectomy, the authors noted.
The prospective cohort study by Dr. Copson and her colleagues included 2,733 women with breast cancer who were aged 40 years or younger at first diagnosis. Of those patients, 338 (12%) had either a BRCA1 or BRCA2 mutation, according to investigators.
At 2 years after diagnosis, overall survival was 97.0% and 96.6% for BRCA-positive and BRCA-negative patients, respectively, the report said. Similarly, overall survival was 83.8% and 85.0% for the two groups at 5 years after diagnosis, and 73.4% vs. 70.1% at 10 years.
Multivariable analysis accounting for known prognostic factors including ethnicity and body mass index showed there was no significant difference between groups (hazard ratio, 0.96; 95% confidence interval, 0.76-1.22; P = .76), the authors wrote.
Triple-negative breast cancer patients with a BRCA mutation might have a survival advantage in the first few years following diagnosis,compared with non-BRCA carriers, the POSH study also found. Researchers reported a significant difference at 2 years (95% for BRCA-positive vs. 91% for BRCA-negative patients; P = .047), but there was no significant difference between arms at 5 or 10 years.
POSH is believed to be the largest prospective cohort study to compare breast cancer outcomes for patients with BRCA mutations to those with sporadic breast cancer. Previous studies, primarily retrospective, have suggested “better, worse, or similar outcomes” for BRCA-positive versus BRCA-negative patients, the authors wrote. Dr. Copson reported receiving honoraria from Roche, while her coauthors reported honoraria from GSK, Pfizer, AstraZeneca, and Pierre Fabre. Funding for the study was provided by the Wessex Cancer Trust, Cancer Research UK, and Breast Cancer Now.
SOURCE: Copson et al. Lancet Oncol. 2018 Jan 11 doi: 10.1016/S1470-2045(17)30891-4.
FROM LANCET ONCOLOGY
Key clinical point: Presence of BRCA1 or BRCA2 germline mutations did not significantly affect overall survival in women with young onset breast cancer.
Major finding: At 2 years, overall survival was 97.0% for BRCA mutation carriers and 96.6% for non-carriers, with similar results reported at 5 and 10 years.
Data source: A prospective cohort study including 2,733 women with breast cancer who were aged 40 years or younger at first diagnosis.
Disclosures: Funding for the study was provided by the Wessex Cancer Trust, Cancer Research UK, and Breast Cancer Now. Study authors declared honoraria from Roche, GSK, Pfizer, AstraZeneca, and Pierre Fabre.
Source: Copson ER et al. Lancet Oncol. 2018 Jan 11. doi: 10.1016/S1470-2045(17)30891-4.
S-1 regimen noninferior to other frontline mCRC options
to other commonly used regimens, according to results of a randomized, open-label, phase 3 trial.
Median progression-free survival for patients receiving the S-1–containing regimen was 14.0 months, compared with 10.8 months for patients receiving either mFOLFOX6 or CapeOX plus bevacizumab (P less than .0001 for noninferiority; P less than .0815 for superiority), Yuji Yamada, MD, of National Cancer Center Hospital, Tokyo, and coauthors reported.
Based on these results, “we consider S-1 and irinotecan plus bevacizumab to be an effective first-line therapy for mCRC and believe that it can be included as one of the recommended standard regimens,” Dr. Yamada and colleagues said (Ann Oncol. 2017 Dec 17. doi: 10.1093/annonc/mdx816).
S-1, a combination preparation that includes the 5-fluorouracil prodrug tegafur plus the modulators gimeracil and oteracil potassium, has been approved in Japan and is approved by the European Medicines Agency for gastric cancer, authors wrote.
Their phase 3 trial, known as TRICOLORE, included 487 patients randomly assigned to receive S-1 and irinotecan plus bevacizumab or a control group that received an oxaliplatin-based regimen (either mFOLFOX6 or CapeOX).
Oxaliplatin-based regimens are associated with milder alopecia and gastrointestinal toxicity, compared with irinotecan-based regimens but can result in prolonged peripheral neuropathy that can negatively impact quality of life and may lead to treatment discontinuation, Dr. Yamada and associates said.
Adverse events of grade 3 or higher occurred in 58.6% of the S-1/irinotecan/bevacizumab group and 64.9% of the control group. The most common grade 3 or greater adverse events were neutropenia and diarrhea in the S-1 group, and neutropenia and peripheral sensory neuropathy in the controls.
The median 14-month progression-free survival for S-1/irinotecan/bevacizumab establishes its noninferiority to the standard regimens, according to the authors: “Although superiority could not be proven, the median progression-free survival was 3.2 months longer in the experimental group than in the control group, and the quality of life results were favorable,” they wrote.
Quality of life was not statistically different between arms as measured by the Functional Assessment of Cancer Therapy-Colorectal Trial Outcome Index, according to the report. However, scores on the FACT/Gynecologic Oncology Group-Neurotoxicity subscale showed a “significantly more favorable trend over time in the experimental group,” authors noted.
The study was supported in part by Taiho Pharmaceutical. Dr. Yamada reported receiving honoraria from Taiho, Chugai, and Yakult.
SOURCE: Yamada Y et al. Ann Oncol. 2017 Dec 17. doi: 10.1093/annonc/mdx816.
to other commonly used regimens, according to results of a randomized, open-label, phase 3 trial.
Median progression-free survival for patients receiving the S-1–containing regimen was 14.0 months, compared with 10.8 months for patients receiving either mFOLFOX6 or CapeOX plus bevacizumab (P less than .0001 for noninferiority; P less than .0815 for superiority), Yuji Yamada, MD, of National Cancer Center Hospital, Tokyo, and coauthors reported.
Based on these results, “we consider S-1 and irinotecan plus bevacizumab to be an effective first-line therapy for mCRC and believe that it can be included as one of the recommended standard regimens,” Dr. Yamada and colleagues said (Ann Oncol. 2017 Dec 17. doi: 10.1093/annonc/mdx816).
S-1, a combination preparation that includes the 5-fluorouracil prodrug tegafur plus the modulators gimeracil and oteracil potassium, has been approved in Japan and is approved by the European Medicines Agency for gastric cancer, authors wrote.
Their phase 3 trial, known as TRICOLORE, included 487 patients randomly assigned to receive S-1 and irinotecan plus bevacizumab or a control group that received an oxaliplatin-based regimen (either mFOLFOX6 or CapeOX).
Oxaliplatin-based regimens are associated with milder alopecia and gastrointestinal toxicity, compared with irinotecan-based regimens but can result in prolonged peripheral neuropathy that can negatively impact quality of life and may lead to treatment discontinuation, Dr. Yamada and associates said.
Adverse events of grade 3 or higher occurred in 58.6% of the S-1/irinotecan/bevacizumab group and 64.9% of the control group. The most common grade 3 or greater adverse events were neutropenia and diarrhea in the S-1 group, and neutropenia and peripheral sensory neuropathy in the controls.
The median 14-month progression-free survival for S-1/irinotecan/bevacizumab establishes its noninferiority to the standard regimens, according to the authors: “Although superiority could not be proven, the median progression-free survival was 3.2 months longer in the experimental group than in the control group, and the quality of life results were favorable,” they wrote.
Quality of life was not statistically different between arms as measured by the Functional Assessment of Cancer Therapy-Colorectal Trial Outcome Index, according to the report. However, scores on the FACT/Gynecologic Oncology Group-Neurotoxicity subscale showed a “significantly more favorable trend over time in the experimental group,” authors noted.
The study was supported in part by Taiho Pharmaceutical. Dr. Yamada reported receiving honoraria from Taiho, Chugai, and Yakult.
SOURCE: Yamada Y et al. Ann Oncol. 2017 Dec 17. doi: 10.1093/annonc/mdx816.
to other commonly used regimens, according to results of a randomized, open-label, phase 3 trial.
Median progression-free survival for patients receiving the S-1–containing regimen was 14.0 months, compared with 10.8 months for patients receiving either mFOLFOX6 or CapeOX plus bevacizumab (P less than .0001 for noninferiority; P less than .0815 for superiority), Yuji Yamada, MD, of National Cancer Center Hospital, Tokyo, and coauthors reported.
Based on these results, “we consider S-1 and irinotecan plus bevacizumab to be an effective first-line therapy for mCRC and believe that it can be included as one of the recommended standard regimens,” Dr. Yamada and colleagues said (Ann Oncol. 2017 Dec 17. doi: 10.1093/annonc/mdx816).
S-1, a combination preparation that includes the 5-fluorouracil prodrug tegafur plus the modulators gimeracil and oteracil potassium, has been approved in Japan and is approved by the European Medicines Agency for gastric cancer, authors wrote.
Their phase 3 trial, known as TRICOLORE, included 487 patients randomly assigned to receive S-1 and irinotecan plus bevacizumab or a control group that received an oxaliplatin-based regimen (either mFOLFOX6 or CapeOX).
Oxaliplatin-based regimens are associated with milder alopecia and gastrointestinal toxicity, compared with irinotecan-based regimens but can result in prolonged peripheral neuropathy that can negatively impact quality of life and may lead to treatment discontinuation, Dr. Yamada and associates said.
Adverse events of grade 3 or higher occurred in 58.6% of the S-1/irinotecan/bevacizumab group and 64.9% of the control group. The most common grade 3 or greater adverse events were neutropenia and diarrhea in the S-1 group, and neutropenia and peripheral sensory neuropathy in the controls.
The median 14-month progression-free survival for S-1/irinotecan/bevacizumab establishes its noninferiority to the standard regimens, according to the authors: “Although superiority could not be proven, the median progression-free survival was 3.2 months longer in the experimental group than in the control group, and the quality of life results were favorable,” they wrote.
Quality of life was not statistically different between arms as measured by the Functional Assessment of Cancer Therapy-Colorectal Trial Outcome Index, according to the report. However, scores on the FACT/Gynecologic Oncology Group-Neurotoxicity subscale showed a “significantly more favorable trend over time in the experimental group,” authors noted.
The study was supported in part by Taiho Pharmaceutical. Dr. Yamada reported receiving honoraria from Taiho, Chugai, and Yakult.
SOURCE: Yamada Y et al. Ann Oncol. 2017 Dec 17. doi: 10.1093/annonc/mdx816.
FROM Annals of Oncology
Key clinical point: The oral fluoropyrimidine S-1 combined with irinotecan and bevacizumab was noninferior to other commonly used first-line regimens in patients with metastatic colorectal cancer (mCRC).
Major finding: Median progression-free-survival was 14.0 months for that regimen, compared with 10.8 months for patients receiving either mFOLFOX6 or CapeOX plus bevacizumab (P less than .0001 for noninferiority; P less than .0815 for superiority).
Data source: TRICOLORE, a randomized, open-label, phase 3, noninferiority trial including 487 mCRC patients enrolled at 53 institutions in Japan.
Disclosures: The study was supported by Tokyo Cooperative Oncology Group with funding from Taiho Pharmaceutica. Study authors reported disclosures related to Taiho, Chugai, Yakult, Ono, Eli Lilly, Eisai, Bayer, and others.
Source: Yamada Y et al. Ann Oncol. 2017 Dec 17. doi: 10.1093/annonc/mdx816.
Rituximab may outperform some other first-line multiple sclerosis treatments
Rituximab was associated with a lower drug discontinuation rate versus all other commonly prescribed disease-modifying treatments (DMTs) used as initial therapy for relapsing-remitting multiple sclerosis (RRMS) in a retrospective study of patient data from a Swedish multiple sclerosis registry.
In addition, relapse rates were lower with rituximab (Rituxan) than they were with injectable DMTs and dimethyl fumarate (Tecfidera), according to results of the study, which appeared online Jan. 8 in JAMA Neurology.
Anti-CD20 agents such as rituximab are “likely to become an additional treatment option” for RRMS, and off-label use of rituximab for this indication has “increased considerably” in Sweden in recent years, the investigators said. RRMS is not an approved indication for rituximab in the United States, whereas across Sweden “there is no difference in reimbursement policy ... because all DMTs are covered by the national health insurance, including off-label medications.”
The addition of new DMTs for RRMS has changed the treatment landscape recently, although in real-world practice, there is a lack of “detailed knowledge about how to tailor therapy,” the authors said. They noted that the majority of patients discontinue traditional first-line treatment with injectable DMTs (that is, interferon beta and glatiramer acetate) within 2 years, suggesting a need for better treatment options.
To evaluate the real-world effectiveness of rituximab in this setting, Dr. Granqvist and his colleagues selected patient registry data for two Swedish counties that 494 included who received a diagnosis of RRMS between January 1, 2012, and October 31, 2015.
The largest subset of patients (n = 215) received injectable DMTs, while the rest received rituximab (n = 120), dimethyl fumarate (n = 86), natalizumab (Tysabri; n = 50), fingolimod (Gilenya; n = 17), or another treatment (n = 6), according to data in the report.
The proportion of patients who stayed on treatment was significantly higher for rituximab versus all other DMTs, study authors found. Compared with rituximab, the hazard ratios for drug discontinuation after adjusting for covariates and propensity score were 11.4 (95% confidence interval, 4.7-27.4) for injectable DMTs, 15.1 (95% CI, 3.9-58.0) for dimethyl fumarate, 5.9 (95% CI, 1.5-23.4) for fingolimod, and 11.3 (95% CI 3.2-39.4) for natalizumab.
Rituximab-treated patients also had lower rates of clinical relapse, neuroradiologic disease activity, and adverse events, compared with injectable DMTs or dimethyl fumarate, according to the investigators.
In comparison with fingolimod and natalizumab, relapse rates and gadolinium-enhancing lesions with rituximab were less frequent, but the authors said those differences did not reach statistical significance in all analyses.
The study was funded by the Swedish Medical Research Council, among other sources. Study authors reported conflicts of interest related to Biogen, Novartis, and Genzyme.
SOURCE: Granqvist M et al. JAMA Neurol. 2018 Jan 8. doi: 10.1001/jamaneurol.2017.4011
Rituximab was associated with a lower drug discontinuation rate versus all other commonly prescribed disease-modifying treatments (DMTs) used as initial therapy for relapsing-remitting multiple sclerosis (RRMS) in a retrospective study of patient data from a Swedish multiple sclerosis registry.
In addition, relapse rates were lower with rituximab (Rituxan) than they were with injectable DMTs and dimethyl fumarate (Tecfidera), according to results of the study, which appeared online Jan. 8 in JAMA Neurology.
Anti-CD20 agents such as rituximab are “likely to become an additional treatment option” for RRMS, and off-label use of rituximab for this indication has “increased considerably” in Sweden in recent years, the investigators said. RRMS is not an approved indication for rituximab in the United States, whereas across Sweden “there is no difference in reimbursement policy ... because all DMTs are covered by the national health insurance, including off-label medications.”
The addition of new DMTs for RRMS has changed the treatment landscape recently, although in real-world practice, there is a lack of “detailed knowledge about how to tailor therapy,” the authors said. They noted that the majority of patients discontinue traditional first-line treatment with injectable DMTs (that is, interferon beta and glatiramer acetate) within 2 years, suggesting a need for better treatment options.
To evaluate the real-world effectiveness of rituximab in this setting, Dr. Granqvist and his colleagues selected patient registry data for two Swedish counties that 494 included who received a diagnosis of RRMS between January 1, 2012, and October 31, 2015.
The largest subset of patients (n = 215) received injectable DMTs, while the rest received rituximab (n = 120), dimethyl fumarate (n = 86), natalizumab (Tysabri; n = 50), fingolimod (Gilenya; n = 17), or another treatment (n = 6), according to data in the report.
The proportion of patients who stayed on treatment was significantly higher for rituximab versus all other DMTs, study authors found. Compared with rituximab, the hazard ratios for drug discontinuation after adjusting for covariates and propensity score were 11.4 (95% confidence interval, 4.7-27.4) for injectable DMTs, 15.1 (95% CI, 3.9-58.0) for dimethyl fumarate, 5.9 (95% CI, 1.5-23.4) for fingolimod, and 11.3 (95% CI 3.2-39.4) for natalizumab.
Rituximab-treated patients also had lower rates of clinical relapse, neuroradiologic disease activity, and adverse events, compared with injectable DMTs or dimethyl fumarate, according to the investigators.
In comparison with fingolimod and natalizumab, relapse rates and gadolinium-enhancing lesions with rituximab were less frequent, but the authors said those differences did not reach statistical significance in all analyses.
The study was funded by the Swedish Medical Research Council, among other sources. Study authors reported conflicts of interest related to Biogen, Novartis, and Genzyme.
SOURCE: Granqvist M et al. JAMA Neurol. 2018 Jan 8. doi: 10.1001/jamaneurol.2017.4011
Rituximab was associated with a lower drug discontinuation rate versus all other commonly prescribed disease-modifying treatments (DMTs) used as initial therapy for relapsing-remitting multiple sclerosis (RRMS) in a retrospective study of patient data from a Swedish multiple sclerosis registry.
In addition, relapse rates were lower with rituximab (Rituxan) than they were with injectable DMTs and dimethyl fumarate (Tecfidera), according to results of the study, which appeared online Jan. 8 in JAMA Neurology.
Anti-CD20 agents such as rituximab are “likely to become an additional treatment option” for RRMS, and off-label use of rituximab for this indication has “increased considerably” in Sweden in recent years, the investigators said. RRMS is not an approved indication for rituximab in the United States, whereas across Sweden “there is no difference in reimbursement policy ... because all DMTs are covered by the national health insurance, including off-label medications.”
The addition of new DMTs for RRMS has changed the treatment landscape recently, although in real-world practice, there is a lack of “detailed knowledge about how to tailor therapy,” the authors said. They noted that the majority of patients discontinue traditional first-line treatment with injectable DMTs (that is, interferon beta and glatiramer acetate) within 2 years, suggesting a need for better treatment options.
To evaluate the real-world effectiveness of rituximab in this setting, Dr. Granqvist and his colleagues selected patient registry data for two Swedish counties that 494 included who received a diagnosis of RRMS between January 1, 2012, and October 31, 2015.
The largest subset of patients (n = 215) received injectable DMTs, while the rest received rituximab (n = 120), dimethyl fumarate (n = 86), natalizumab (Tysabri; n = 50), fingolimod (Gilenya; n = 17), or another treatment (n = 6), according to data in the report.
The proportion of patients who stayed on treatment was significantly higher for rituximab versus all other DMTs, study authors found. Compared with rituximab, the hazard ratios for drug discontinuation after adjusting for covariates and propensity score were 11.4 (95% confidence interval, 4.7-27.4) for injectable DMTs, 15.1 (95% CI, 3.9-58.0) for dimethyl fumarate, 5.9 (95% CI, 1.5-23.4) for fingolimod, and 11.3 (95% CI 3.2-39.4) for natalizumab.
Rituximab-treated patients also had lower rates of clinical relapse, neuroradiologic disease activity, and adverse events, compared with injectable DMTs or dimethyl fumarate, according to the investigators.
In comparison with fingolimod and natalizumab, relapse rates and gadolinium-enhancing lesions with rituximab were less frequent, but the authors said those differences did not reach statistical significance in all analyses.
The study was funded by the Swedish Medical Research Council, among other sources. Study authors reported conflicts of interest related to Biogen, Novartis, and Genzyme.
SOURCE: Granqvist M et al. JAMA Neurol. 2018 Jan 8. doi: 10.1001/jamaneurol.2017.4011
FROM JAMA NEUROLOGY
Key clinical point:
Major finding: Rituximab-treated patients had significantly lower rates of discontinuation, compared with injectable DMTs, fingolimod, natalizumab, and dimethyl fumarate. Relapse rates with rituximab were lower than they were with injectable DMTs and dimethyl fumarate.
Data source: Retrospective cohort study of data from a Swedish multiple sclerosis registry that included 494 patients with a diagnosis of RRMS.
Disclosures: The study was funded by the Swedish Medical Research Council, among other sources. Study authors reported conflicts of interest related to Biogen, Novartis, and Genzyme.
Source: Granqvist M et al. JAMA Neurol. 2018 Jan 8. doi: 10.1001/jamaneurol.2017.4011
BTK inhibitor zanubrutinib active in non-Hodgkin lymphomas
ATLANTA – , according to data presented at the annual meeting of the American Society of Hematology.
Response rates ranged from 31% to 88% depending on the lymphoma subtype. Overall, approximately 10% of patients discontinued the drug because of adverse events, reported Constantine S. Tam, MBBS, MD, of Peter MacCallum Cancer Centre & St. Vincent’s Hospital, Melbourne.
“There was encouraging activity against all the spectrum of indolent and aggressive NHL subtypes … and durable responses were observed across a variety of histologies,” Dr. Tam said.
Zanubrutinib is a second-generation BTK inhibitor that, based on biochemical assays, has higher selectivity against BTK than ibrutinib, Dr. Tam said.
He presented results of an open-label, multicenter, phase 1b study of daily or twice-daily zanubrutinib in patients with B-cell malignancies, most of them relapsed or refractory to prior therapies. The lymphoma subtypes he presented included diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), mantle cell lymphoma (MCL), and marginal zone lymphoma (MZL).
For 34 patients with indolent lymphomas (FL and MZL), the most frequent adverse events were petechiae/purpura/contusion and upper respiratory tract infection. Eleven grade 3-5 adverse events were reported, including neutropenia, infection, nausea, urinary tract infection, and abdominal pain.
Atrial fibrillation was observed in two patients in the aggressive NHL cohort, for an overall AF rate of approximately 2%, Dr. Tam said.
For 65 patients with aggressive lymphomas (DLBCL and MCL), the most frequent adverse events were petechiae/purpura/contusion and diarrhea; 27 grade 3-5 adverse events were reported, including neutropenia, pneumonia, and anemia.
The highest overall response rate reported was for MCL, at 88% (28 of 32 patients) followed by MZL at 78% (7 of 9 patients), FL at 41% (7 of 17 patients), and DLBCL 31% (8 of 26 patients).
The recommended phase 2 dose for zanubrutinib is either 320 mg/day once daily or a split dose of 160 mg twice daily, Dr. Tam said.
Based on this experience, investigators started a registration trial of zanubrutinib in combination with obinutuzumab for FL, and additional trials are planned, according to Dr. Tam.
There are also registration trials in Waldenstrom macroglobulinemia and chronic lymphocytic leukemia based on other data suggesting activity of zanubrutinib in those disease types, he added.
Zanubrutinib is a product of BeiGene. Dr. Tam reported disclosures related to Roche, Janssen Cilag, Abbvie, Celgene, Pharmacyclics, Onyx, and Amgen.
SOURCE: Tam C et al, ASH 2017, Abstract 152
ATLANTA – , according to data presented at the annual meeting of the American Society of Hematology.
Response rates ranged from 31% to 88% depending on the lymphoma subtype. Overall, approximately 10% of patients discontinued the drug because of adverse events, reported Constantine S. Tam, MBBS, MD, of Peter MacCallum Cancer Centre & St. Vincent’s Hospital, Melbourne.
“There was encouraging activity against all the spectrum of indolent and aggressive NHL subtypes … and durable responses were observed across a variety of histologies,” Dr. Tam said.
Zanubrutinib is a second-generation BTK inhibitor that, based on biochemical assays, has higher selectivity against BTK than ibrutinib, Dr. Tam said.
He presented results of an open-label, multicenter, phase 1b study of daily or twice-daily zanubrutinib in patients with B-cell malignancies, most of them relapsed or refractory to prior therapies. The lymphoma subtypes he presented included diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), mantle cell lymphoma (MCL), and marginal zone lymphoma (MZL).
For 34 patients with indolent lymphomas (FL and MZL), the most frequent adverse events were petechiae/purpura/contusion and upper respiratory tract infection. Eleven grade 3-5 adverse events were reported, including neutropenia, infection, nausea, urinary tract infection, and abdominal pain.
Atrial fibrillation was observed in two patients in the aggressive NHL cohort, for an overall AF rate of approximately 2%, Dr. Tam said.
For 65 patients with aggressive lymphomas (DLBCL and MCL), the most frequent adverse events were petechiae/purpura/contusion and diarrhea; 27 grade 3-5 adverse events were reported, including neutropenia, pneumonia, and anemia.
The highest overall response rate reported was for MCL, at 88% (28 of 32 patients) followed by MZL at 78% (7 of 9 patients), FL at 41% (7 of 17 patients), and DLBCL 31% (8 of 26 patients).
The recommended phase 2 dose for zanubrutinib is either 320 mg/day once daily or a split dose of 160 mg twice daily, Dr. Tam said.
Based on this experience, investigators started a registration trial of zanubrutinib in combination with obinutuzumab for FL, and additional trials are planned, according to Dr. Tam.
There are also registration trials in Waldenstrom macroglobulinemia and chronic lymphocytic leukemia based on other data suggesting activity of zanubrutinib in those disease types, he added.
Zanubrutinib is a product of BeiGene. Dr. Tam reported disclosures related to Roche, Janssen Cilag, Abbvie, Celgene, Pharmacyclics, Onyx, and Amgen.
SOURCE: Tam C et al, ASH 2017, Abstract 152
ATLANTA – , according to data presented at the annual meeting of the American Society of Hematology.
Response rates ranged from 31% to 88% depending on the lymphoma subtype. Overall, approximately 10% of patients discontinued the drug because of adverse events, reported Constantine S. Tam, MBBS, MD, of Peter MacCallum Cancer Centre & St. Vincent’s Hospital, Melbourne.
“There was encouraging activity against all the spectrum of indolent and aggressive NHL subtypes … and durable responses were observed across a variety of histologies,” Dr. Tam said.
Zanubrutinib is a second-generation BTK inhibitor that, based on biochemical assays, has higher selectivity against BTK than ibrutinib, Dr. Tam said.
He presented results of an open-label, multicenter, phase 1b study of daily or twice-daily zanubrutinib in patients with B-cell malignancies, most of them relapsed or refractory to prior therapies. The lymphoma subtypes he presented included diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), mantle cell lymphoma (MCL), and marginal zone lymphoma (MZL).
For 34 patients with indolent lymphomas (FL and MZL), the most frequent adverse events were petechiae/purpura/contusion and upper respiratory tract infection. Eleven grade 3-5 adverse events were reported, including neutropenia, infection, nausea, urinary tract infection, and abdominal pain.
Atrial fibrillation was observed in two patients in the aggressive NHL cohort, for an overall AF rate of approximately 2%, Dr. Tam said.
For 65 patients with aggressive lymphomas (DLBCL and MCL), the most frequent adverse events were petechiae/purpura/contusion and diarrhea; 27 grade 3-5 adverse events were reported, including neutropenia, pneumonia, and anemia.
The highest overall response rate reported was for MCL, at 88% (28 of 32 patients) followed by MZL at 78% (7 of 9 patients), FL at 41% (7 of 17 patients), and DLBCL 31% (8 of 26 patients).
The recommended phase 2 dose for zanubrutinib is either 320 mg/day once daily or a split dose of 160 mg twice daily, Dr. Tam said.
Based on this experience, investigators started a registration trial of zanubrutinib in combination with obinutuzumab for FL, and additional trials are planned, according to Dr. Tam.
There are also registration trials in Waldenstrom macroglobulinemia and chronic lymphocytic leukemia based on other data suggesting activity of zanubrutinib in those disease types, he added.
Zanubrutinib is a product of BeiGene. Dr. Tam reported disclosures related to Roche, Janssen Cilag, Abbvie, Celgene, Pharmacyclics, Onyx, and Amgen.
SOURCE: Tam C et al, ASH 2017, Abstract 152
REPORTING FROM ASH 2017
Key clinical point: Monotherapy with the BTK inhibitor zanubrutinib (BGB-3111) was active and well tolerated in patients with a variety of non-Hodgkin lymphoma (NHL) subtypes.
Major finding: Response rates ranged from 31% to 88% depending on the lymphoma subtype.
Data source: Preliminary results of an open-label, multicenter, phase 1b study including 99 patients with relapsed or refractory diffuse large B-cell lymphoma, follicular lymphoma, mantle cell lymphoma, or marginal zone lymphoma.
Disclosures: Zanubrutinib is a product of BeiGene. Constantine S. Tam, MBBS, MD, reported disclosures related to Roche, Janssen Cilag, Abbvie, Celgene, Pharmacyclics, Onyx, and Amgen.
Source: Tam C et al. ASH 2017, Abstract 152.
Sequential chemotherapy and radiotherapy may be best in locally advanced NSCLC with negative margins
Chemotherapy followed by radiation resulted in superior outcomes compared with concurrent chemoradiotherapy in patients with non–small-cell lung cancer (NSCLC) who had negative margins after surgery and pN2 disease, according to results of a retrospective analysis.
“We also found that sequential therapy is becoming the more frequent practice pattern over time for patients with R0 pN2 disease, and our results support this practice,” wrote Samual Francis, MD, of the University of Utah Huntsman Cancer Institute, Salt Lake City, and his coauthors.
By contrast, there was no clear association between treatment sequence and survival among patients who had positive margins after surgery in this analysis, authors of the study reported (J Clin Oncol. 2017 Dec 13. doi: 10.1200/JCO.2017.74.4771).
The study included 1,024 patients in National Cancer Database who received a diagnosis of nonmetastatic NSCLC and received chemotherapy and radiation concurrently or sequentially after surgery. Of those patients, 747 had R0 resections and pN2 nodal status, and the remainder had R1 or R2 resections and pN0-N2 disease.
For patients with R0 pN2 NSCLC, median overall survival was 58.8 months for sequential chemotherapy followed by radiation, versus 40.4 months for concurrent chemoradiotherapy (log-rank P less than .001), data show.
That association between sequential treatment and improved overall survival remained significant even after propensity score matching (hazard ratio [HR], 1.35; P = .019), investigators reported.
However, for the remaining cohort of patients with positive margins regardless of nodal status, there was no significant difference in overall survival favoring either approach (42.6 months for sequential versus 38.5 months for concurrent; log-rank P = .42), they added, and no clear association between treatment approaches (HR 1.35; P = .19).
The analysis covered patients diagnosed between 2006 and 2012, the most recent data period available.
Investigators were also able to identify changes in practice patterns over time using this data set. Between 2006 and 2012, there was an increase in the use of sequential chemotherapy and radiotherapy for patients with R0 resection and pN2 disease, and a decrease in the use of concurrent chemoradiotherapy.
Conversely, they found that for patients with positive margins, there was an increase over time in use of the concurrent approach and decrease in the sequential approach.
“These findings suggest practice patterns have shifted toward concordance with consensus guideline recommendations over time,” Dr. Francis and his colleagues said in their report.
Current guidelines advocate for chemotherapy followed by postoperative radiotherapy for patients with negative margins and pN2 disease, but suggest concurrent chemoradiotherapy for patients with positive margins, they added.
Dr. Francis had no relationships to disclose. Study coauthors reported disclosures related to Elekta, ProLung, Merit Medical Systems, and Bard Medical.
Chemotherapy followed by radiation resulted in superior outcomes compared with concurrent chemoradiotherapy in patients with non–small-cell lung cancer (NSCLC) who had negative margins after surgery and pN2 disease, according to results of a retrospective analysis.
“We also found that sequential therapy is becoming the more frequent practice pattern over time for patients with R0 pN2 disease, and our results support this practice,” wrote Samual Francis, MD, of the University of Utah Huntsman Cancer Institute, Salt Lake City, and his coauthors.
By contrast, there was no clear association between treatment sequence and survival among patients who had positive margins after surgery in this analysis, authors of the study reported (J Clin Oncol. 2017 Dec 13. doi: 10.1200/JCO.2017.74.4771).
The study included 1,024 patients in National Cancer Database who received a diagnosis of nonmetastatic NSCLC and received chemotherapy and radiation concurrently or sequentially after surgery. Of those patients, 747 had R0 resections and pN2 nodal status, and the remainder had R1 or R2 resections and pN0-N2 disease.
For patients with R0 pN2 NSCLC, median overall survival was 58.8 months for sequential chemotherapy followed by radiation, versus 40.4 months for concurrent chemoradiotherapy (log-rank P less than .001), data show.
That association between sequential treatment and improved overall survival remained significant even after propensity score matching (hazard ratio [HR], 1.35; P = .019), investigators reported.
However, for the remaining cohort of patients with positive margins regardless of nodal status, there was no significant difference in overall survival favoring either approach (42.6 months for sequential versus 38.5 months for concurrent; log-rank P = .42), they added, and no clear association between treatment approaches (HR 1.35; P = .19).
The analysis covered patients diagnosed between 2006 and 2012, the most recent data period available.
Investigators were also able to identify changes in practice patterns over time using this data set. Between 2006 and 2012, there was an increase in the use of sequential chemotherapy and radiotherapy for patients with R0 resection and pN2 disease, and a decrease in the use of concurrent chemoradiotherapy.
Conversely, they found that for patients with positive margins, there was an increase over time in use of the concurrent approach and decrease in the sequential approach.
“These findings suggest practice patterns have shifted toward concordance with consensus guideline recommendations over time,” Dr. Francis and his colleagues said in their report.
Current guidelines advocate for chemotherapy followed by postoperative radiotherapy for patients with negative margins and pN2 disease, but suggest concurrent chemoradiotherapy for patients with positive margins, they added.
Dr. Francis had no relationships to disclose. Study coauthors reported disclosures related to Elekta, ProLung, Merit Medical Systems, and Bard Medical.
Chemotherapy followed by radiation resulted in superior outcomes compared with concurrent chemoradiotherapy in patients with non–small-cell lung cancer (NSCLC) who had negative margins after surgery and pN2 disease, according to results of a retrospective analysis.
“We also found that sequential therapy is becoming the more frequent practice pattern over time for patients with R0 pN2 disease, and our results support this practice,” wrote Samual Francis, MD, of the University of Utah Huntsman Cancer Institute, Salt Lake City, and his coauthors.
By contrast, there was no clear association between treatment sequence and survival among patients who had positive margins after surgery in this analysis, authors of the study reported (J Clin Oncol. 2017 Dec 13. doi: 10.1200/JCO.2017.74.4771).
The study included 1,024 patients in National Cancer Database who received a diagnosis of nonmetastatic NSCLC and received chemotherapy and radiation concurrently or sequentially after surgery. Of those patients, 747 had R0 resections and pN2 nodal status, and the remainder had R1 or R2 resections and pN0-N2 disease.
For patients with R0 pN2 NSCLC, median overall survival was 58.8 months for sequential chemotherapy followed by radiation, versus 40.4 months for concurrent chemoradiotherapy (log-rank P less than .001), data show.
That association between sequential treatment and improved overall survival remained significant even after propensity score matching (hazard ratio [HR], 1.35; P = .019), investigators reported.
However, for the remaining cohort of patients with positive margins regardless of nodal status, there was no significant difference in overall survival favoring either approach (42.6 months for sequential versus 38.5 months for concurrent; log-rank P = .42), they added, and no clear association between treatment approaches (HR 1.35; P = .19).
The analysis covered patients diagnosed between 2006 and 2012, the most recent data period available.
Investigators were also able to identify changes in practice patterns over time using this data set. Between 2006 and 2012, there was an increase in the use of sequential chemotherapy and radiotherapy for patients with R0 resection and pN2 disease, and a decrease in the use of concurrent chemoradiotherapy.
Conversely, they found that for patients with positive margins, there was an increase over time in use of the concurrent approach and decrease in the sequential approach.
“These findings suggest practice patterns have shifted toward concordance with consensus guideline recommendations over time,” Dr. Francis and his colleagues said in their report.
Current guidelines advocate for chemotherapy followed by postoperative radiotherapy for patients with negative margins and pN2 disease, but suggest concurrent chemoradiotherapy for patients with positive margins, they added.
Dr. Francis had no relationships to disclose. Study coauthors reported disclosures related to Elekta, ProLung, Merit Medical Systems, and Bard Medical.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: In patients with pN2 non–small-cell lung cancer who have negative margins after surgery, sequential chemotherapy and radiotherapy provided superior outcomes compared with concurrent chemoradiotherapy.
Major finding: Median overall survival was 58.8 months for the sequential approach and 40.4 months for concurrent (log-rank P less than .001).
Data source: A retrospective analysis including 1,024 patients with nonmetastatic NSCLC who received chemotherapy and radiation concurrently or sequentially after surgery.
Disclosures: Study authors reported disclosures related to Elekta, ProLung, Merit Medical Systems, and Bard Medical.
Rare neurological complication linked to Waldenstrom disease
Bilateral facial nerve palsy has been associated with underlying Waldenstrom disease in only one other known published case report, which was published in 2014. In a more recent case report published in the Journal of Clinical Neuroscience, Gabriel Torrealba-Acosta, MD, and colleagues in the department of neurology at Massachusetts General Hospital, Boston, described a second case involving a 67-year-old Hispanic man with a history of Waldenstrom disease who presented with subacute onset of bilateral facial weakness.
The patient, who had longstanding painful neuropathy, had presented to urgent care with a new-onset left facial nerve palsy, was then diagnosed with left Bell’s palsy, and began treatment with valacyclovir and prednisone.
The left-sided facial weakness gradually progressed to total paralysis of the left lower face and inability to close the left eye, and 2 weeks later, he developed right facial weakness that ran a similar course. The patient had a complicated clinical course that included symptomatic acute-on-chronic subdural hematoma, among other complications; eventually the patient’s symptoms stabilized and cranial neuropathies gradually improved, according to the report.
Bilateral facial nerve palsy is an extremely rare condition, occurring in just 0.3%-2% of all facial nerve palsy cases, according to the authors. By contrast, unilateral facial nerve palsy (or Bell’s palsy) is far more common, but it still occurs in only 25 patients per 100,000 population, they said.
Most cases of bilateral facial nerve palsy are caused by underlying Guillain-Barré syndrome, though some are congenital, related to trauma, or caused by etiologies that are metabolic, immunologic, or neoplastic in nature. While various types of neurological disturbances – from ischemic and hemorrhagic events to meningoencephalitis – have been documented to occur in up to a quarter of patients with Waldenstrom disease.
“Given the large differential that comprises the assessment of a bilateral facial nerve palsy, it warrants for an extensive work-up, and Waldenstrom’s macroglobulinemia should be sought as an additional possible etiology,” the authors wrote.
Dr. Torrealba-Acosta and coauthors reported having no financial disclosures.
SOURCE: Torrealba-Acosta G et al. J Clin Neurosci. 2017. doi: 10.1016/j.jocn.2017.10.081.
Bilateral facial nerve palsy has been associated with underlying Waldenstrom disease in only one other known published case report, which was published in 2014. In a more recent case report published in the Journal of Clinical Neuroscience, Gabriel Torrealba-Acosta, MD, and colleagues in the department of neurology at Massachusetts General Hospital, Boston, described a second case involving a 67-year-old Hispanic man with a history of Waldenstrom disease who presented with subacute onset of bilateral facial weakness.
The patient, who had longstanding painful neuropathy, had presented to urgent care with a new-onset left facial nerve palsy, was then diagnosed with left Bell’s palsy, and began treatment with valacyclovir and prednisone.
The left-sided facial weakness gradually progressed to total paralysis of the left lower face and inability to close the left eye, and 2 weeks later, he developed right facial weakness that ran a similar course. The patient had a complicated clinical course that included symptomatic acute-on-chronic subdural hematoma, among other complications; eventually the patient’s symptoms stabilized and cranial neuropathies gradually improved, according to the report.
Bilateral facial nerve palsy is an extremely rare condition, occurring in just 0.3%-2% of all facial nerve palsy cases, according to the authors. By contrast, unilateral facial nerve palsy (or Bell’s palsy) is far more common, but it still occurs in only 25 patients per 100,000 population, they said.
Most cases of bilateral facial nerve palsy are caused by underlying Guillain-Barré syndrome, though some are congenital, related to trauma, or caused by etiologies that are metabolic, immunologic, or neoplastic in nature. While various types of neurological disturbances – from ischemic and hemorrhagic events to meningoencephalitis – have been documented to occur in up to a quarter of patients with Waldenstrom disease.
“Given the large differential that comprises the assessment of a bilateral facial nerve palsy, it warrants for an extensive work-up, and Waldenstrom’s macroglobulinemia should be sought as an additional possible etiology,” the authors wrote.
Dr. Torrealba-Acosta and coauthors reported having no financial disclosures.
SOURCE: Torrealba-Acosta G et al. J Clin Neurosci. 2017. doi: 10.1016/j.jocn.2017.10.081.
Bilateral facial nerve palsy has been associated with underlying Waldenstrom disease in only one other known published case report, which was published in 2014. In a more recent case report published in the Journal of Clinical Neuroscience, Gabriel Torrealba-Acosta, MD, and colleagues in the department of neurology at Massachusetts General Hospital, Boston, described a second case involving a 67-year-old Hispanic man with a history of Waldenstrom disease who presented with subacute onset of bilateral facial weakness.
The patient, who had longstanding painful neuropathy, had presented to urgent care with a new-onset left facial nerve palsy, was then diagnosed with left Bell’s palsy, and began treatment with valacyclovir and prednisone.
The left-sided facial weakness gradually progressed to total paralysis of the left lower face and inability to close the left eye, and 2 weeks later, he developed right facial weakness that ran a similar course. The patient had a complicated clinical course that included symptomatic acute-on-chronic subdural hematoma, among other complications; eventually the patient’s symptoms stabilized and cranial neuropathies gradually improved, according to the report.
Bilateral facial nerve palsy is an extremely rare condition, occurring in just 0.3%-2% of all facial nerve palsy cases, according to the authors. By contrast, unilateral facial nerve palsy (or Bell’s palsy) is far more common, but it still occurs in only 25 patients per 100,000 population, they said.
Most cases of bilateral facial nerve palsy are caused by underlying Guillain-Barré syndrome, though some are congenital, related to trauma, or caused by etiologies that are metabolic, immunologic, or neoplastic in nature. While various types of neurological disturbances – from ischemic and hemorrhagic events to meningoencephalitis – have been documented to occur in up to a quarter of patients with Waldenstrom disease.
“Given the large differential that comprises the assessment of a bilateral facial nerve palsy, it warrants for an extensive work-up, and Waldenstrom’s macroglobulinemia should be sought as an additional possible etiology,” the authors wrote.
Dr. Torrealba-Acosta and coauthors reported having no financial disclosures.
SOURCE: Torrealba-Acosta G et al. J Clin Neurosci. 2017. doi: 10.1016/j.jocn.2017.10.081.
FROM THE JOURNAL OF CLINICAL NEUROSCIENCE