User login
Varicella Outbreaks: 2022-2024
Practitioners providing care to children are familiar with the childhood immunization schedule and routinely administer varicella vaccine at the 12-month and 4- to 5-year visits. However, when is the last time most of us or any of the current trainees have seen a case?
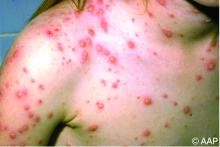
Briefly, varicella is a highly contagious disease caused by varicella-zoster virus (VZV). It is characterized by a generalized pruritic erythematous rash in various stages of development beginning as macules, progressing to papules, and ultimately becoming vesicular lesions on an erythematous base (“dewdrop on a rose petal”) and resolves with crusting of the lesion (Figure 1). It has an incubation period of 10-21 days with symptoms usually developing within 14-16 days after exposure. The vesicular rash must be differentiated from enterovirus, Staphylococcus aureus, contact dermatitis, or insect bites, which initially may be difficult. Approximately 50% of children can have symptoms including fever, malaise, anorexia, headache, and occasionally, mild abdominal pain in the 24-48 hours prior to the appearance of rash. Lesions usually first appear on the scalp, face, or trunk in successive crops over several days. A person with varicella has lesions in various stages.
In a normal host, new vesicle formation usually stops within 4 days, and most lesions have fully crusted by day 6. VZV establishes latency in sensory ganglia and may reactivate years or decades later to cause herpes zoster (HZ). Most healthy children with varicella recover without sequelae so the disease is generally regarded as benign. However, varicella can lead to serious complications and deaths in healthy as well as immunocompromised persons.
Complications of Varicella: bacterial superinfection of skin lesions most often with Streptococcus pyogenes or S aureus manifested as cellulitis, myositis, or necrotizing fasciitis; neurologic complications include cerebellar ataxia and encephalitis with the latter seen most often in adults. Pneumonia occurs most often in adults, especially those infected during pregnancy. Another concern, infection during the first 20 weeks of pregnancy can lead to fetal death or severe birth defects, including limb hypoplasia, cutaneous scarring, ocular abnormalities, and central nervous system damage (congenital varicella syndrome).
The risk for development of severe disseminated disease was first noted in the 1960s as treatments for leukemia in children improved. They were surviving their cancer only to develop severe and often fatal varicella. Today it is recognized that development of disseminated disease is a risk for all infected persons with impaired T cell function, malignancies, HIV, or receiving immunosuppressive therapy.
Reye’s syndrome is rarely seen today since taking salicylates while infected with VZV was identified as a predisposing factor for development.
VZV is only found in humans and transmission is person to person or airborne. The secondary household attack rate is approximately 90%. In contrast, the secondary attack rates in classrooms may be as low as 12%-33%. Transmission rates in the tropics for unexplained reasons are also lower.
Vaccine History: Why do we rarely see this disease anymore? Varicella, a live attenuated vaccine, was developed in 1974 by Dr. Michiaki Takahashi. It remains the only vaccine directed against a herpes group virus. In 1979, the Collaborative Varicella Vaccine Study Group was established at the National Institutes of Health (NIH) and additional safety and efficacy trials were conducted in the United States initially in leukemic patients in remission and later in healthy children, which supported Takahashi’s data. Licensure of varicella vaccine was granted in 1995. That same year, due to continuing disease and societal burden, the United States was the first country to incorporate varicella into the routine childhood immunization schedule, which resulted in significant reductions in cases. To further improve control of varicella, in 2007 vaccine recommendations were revised and a routine two-dose schedule was implemented. The impact of varicella disease pre- and post-vaccine licensure is illustrated in Figure 2. Not listed, is that in the pre-vaccine era, there were approximately 44 cases of congenital varicella syndrome annually.

As of 2023 only 23% (45/195) of nations routinely administer this vaccine and 4% (8/195) have restricted recommendations. The remaining 73% of countries do not offer the vaccine, including all countries on the African continent, and Cuba, Guatemala, Haiti, Honduras, India, Jordan, Lebanon, Philippines, Portugal, and Venezuela to list a few.
Varicella Outbreak: In October 2022, New York City (NYC) identified a varicella outbreak primarily involving persons who recently migrated from Central and South America and lived in a shelter in NYC or residential facility (n = 105); the outbreak is ongoing. As of March 8, 2024, 873 cases (53%) were among children aged 4-18 years and 91.9% had no documentation of varicella vaccine at time of symptom onset. There were 28 hospitalizations, and no deaths reported. The most common sources of transmission were the residential facilities (41.3%) and importation or possible importation (39.4%). School transmission accounted for only 1.2% of cases.
Most migrants arrived from countries where varicella vaccination is not part of the routine childhood immunization schedule. Although most cases occurred in children, almost 30% occurred in adults. Many of the migrants arrived from tropical countries where susceptibility rates are also higher in adults. This outbreak is a reminder of the importance of limiting disease transmission by maintaining high vaccination rates. To curtail this outbreak, approximately 27,000 doses of varicella vaccine were administered to the arriving migrants. In addition, MMR, COVID-19, influenza, and all routine pediatric vaccines required for school entry were administered. Temporary closure of the residential facilities were required. Education was provided to residents regarding immunizations as well as assistance to help them establish a primary care home. Multiple agencies were mobilized to successfully coordinate these efforts.
Take Home Message
1. Each country has its own routine immunization schedule. It may not include all vaccines recommended in the US schedule. When questioned I’m frequently told that immunizations are up to date, only to review records and find they are not, especially when it is related to MMR. It is often administered at 9 months and/or MR or MM is administered depending on the country. As reported here, varicella is a routine vaccine in only 45 countries.
2.
3. Once an outbreak has been identified, the infrastructure to manage and contain it must already be established. In most instances there will be a need for a rapid and often large-scale effort involving multiple agencies including local health care providers.
4. Not all diseases are reportable. Only deaths by varicella are nationally notifiable. Otherwise, cases are reported voluntarily. As of November 2, 2024, there have been 5,157 cases of varicella reported, excluding any cases from NYC.
Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Suggested Reading
CDC. Nationally Notifiable Infectious Diseases and Conditions, United States: Weekly Tables. https://wonder.cdc.gov/nndss/nndss_weekly_tables_menu.asp.
Graham KA et al. Varicella Outbreak Among Recent Arrivals to New York City, 2022-2024. MMWR Morb Mortal Wkly Rep. 2024 May 30;73(21):478-483. doi: 10.15585/mmwr.mm7321a1.
Marin M et al. Health and Economic Impact of the United States Varicella Vaccination Program, 1996-2020. J Infect Dis. 2022 Oct 21;226(Suppl 4):S463-S469. doi: 10.1093/infdis/jiac271.
Varicella-Zoster Virus Infections in Kimberkin DW et al, eds. Red Book: 2024 Report of the Committee on Infectious Diseases, 33rd Edition. American Academy of Pediatrics, 2024:938-951. https://www.aap.org/Red-Book-2024-Report-of-the-Committee-on-Infectious-Diseases-33rd-Edition-Paperback?srsltid=AfmBOoqyF60rR9ZwQ5jA8AouNhtRRTyPLnc_r7HWw7JVYV8v33Hr2vQS.
Practitioners providing care to children are familiar with the childhood immunization schedule and routinely administer varicella vaccine at the 12-month and 4- to 5-year visits. However, when is the last time most of us or any of the current trainees have seen a case?
Briefly, varicella is a highly contagious disease caused by varicella-zoster virus (VZV). It is characterized by a generalized pruritic erythematous rash in various stages of development beginning as macules, progressing to papules, and ultimately becoming vesicular lesions on an erythematous base (“dewdrop on a rose petal”) and resolves with crusting of the lesion (Figure 1). It has an incubation period of 10-21 days with symptoms usually developing within 14-16 days after exposure. The vesicular rash must be differentiated from enterovirus, Staphylococcus aureus, contact dermatitis, or insect bites, which initially may be difficult. Approximately 50% of children can have symptoms including fever, malaise, anorexia, headache, and occasionally, mild abdominal pain in the 24-48 hours prior to the appearance of rash. Lesions usually first appear on the scalp, face, or trunk in successive crops over several days. A person with varicella has lesions in various stages.
In a normal host, new vesicle formation usually stops within 4 days, and most lesions have fully crusted by day 6. VZV establishes latency in sensory ganglia and may reactivate years or decades later to cause herpes zoster (HZ). Most healthy children with varicella recover without sequelae so the disease is generally regarded as benign. However, varicella can lead to serious complications and deaths in healthy as well as immunocompromised persons.
Complications of Varicella: bacterial superinfection of skin lesions most often with Streptococcus pyogenes or S aureus manifested as cellulitis, myositis, or necrotizing fasciitis; neurologic complications include cerebellar ataxia and encephalitis with the latter seen most often in adults. Pneumonia occurs most often in adults, especially those infected during pregnancy. Another concern, infection during the first 20 weeks of pregnancy can lead to fetal death or severe birth defects, including limb hypoplasia, cutaneous scarring, ocular abnormalities, and central nervous system damage (congenital varicella syndrome).
The risk for development of severe disseminated disease was first noted in the 1960s as treatments for leukemia in children improved. They were surviving their cancer only to develop severe and often fatal varicella. Today it is recognized that development of disseminated disease is a risk for all infected persons with impaired T cell function, malignancies, HIV, or receiving immunosuppressive therapy.
Reye’s syndrome is rarely seen today since taking salicylates while infected with VZV was identified as a predisposing factor for development.
VZV is only found in humans and transmission is person to person or airborne. The secondary household attack rate is approximately 90%. In contrast, the secondary attack rates in classrooms may be as low as 12%-33%. Transmission rates in the tropics for unexplained reasons are also lower.
Vaccine History: Why do we rarely see this disease anymore? Varicella, a live attenuated vaccine, was developed in 1974 by Dr. Michiaki Takahashi. It remains the only vaccine directed against a herpes group virus. In 1979, the Collaborative Varicella Vaccine Study Group was established at the National Institutes of Health (NIH) and additional safety and efficacy trials were conducted in the United States initially in leukemic patients in remission and later in healthy children, which supported Takahashi’s data. Licensure of varicella vaccine was granted in 1995. That same year, due to continuing disease and societal burden, the United States was the first country to incorporate varicella into the routine childhood immunization schedule, which resulted in significant reductions in cases. To further improve control of varicella, in 2007 vaccine recommendations were revised and a routine two-dose schedule was implemented. The impact of varicella disease pre- and post-vaccine licensure is illustrated in Figure 2. Not listed, is that in the pre-vaccine era, there were approximately 44 cases of congenital varicella syndrome annually.

As of 2023 only 23% (45/195) of nations routinely administer this vaccine and 4% (8/195) have restricted recommendations. The remaining 73% of countries do not offer the vaccine, including all countries on the African continent, and Cuba, Guatemala, Haiti, Honduras, India, Jordan, Lebanon, Philippines, Portugal, and Venezuela to list a few.
Varicella Outbreak: In October 2022, New York City (NYC) identified a varicella outbreak primarily involving persons who recently migrated from Central and South America and lived in a shelter in NYC or residential facility (n = 105); the outbreak is ongoing. As of March 8, 2024, 873 cases (53%) were among children aged 4-18 years and 91.9% had no documentation of varicella vaccine at time of symptom onset. There were 28 hospitalizations, and no deaths reported. The most common sources of transmission were the residential facilities (41.3%) and importation or possible importation (39.4%). School transmission accounted for only 1.2% of cases.
Most migrants arrived from countries where varicella vaccination is not part of the routine childhood immunization schedule. Although most cases occurred in children, almost 30% occurred in adults. Many of the migrants arrived from tropical countries where susceptibility rates are also higher in adults. This outbreak is a reminder of the importance of limiting disease transmission by maintaining high vaccination rates. To curtail this outbreak, approximately 27,000 doses of varicella vaccine were administered to the arriving migrants. In addition, MMR, COVID-19, influenza, and all routine pediatric vaccines required for school entry were administered. Temporary closure of the residential facilities were required. Education was provided to residents regarding immunizations as well as assistance to help them establish a primary care home. Multiple agencies were mobilized to successfully coordinate these efforts.
Take Home Message
1. Each country has its own routine immunization schedule. It may not include all vaccines recommended in the US schedule. When questioned I’m frequently told that immunizations are up to date, only to review records and find they are not, especially when it is related to MMR. It is often administered at 9 months and/or MR or MM is administered depending on the country. As reported here, varicella is a routine vaccine in only 45 countries.
2.
3. Once an outbreak has been identified, the infrastructure to manage and contain it must already be established. In most instances there will be a need for a rapid and often large-scale effort involving multiple agencies including local health care providers.
4. Not all diseases are reportable. Only deaths by varicella are nationally notifiable. Otherwise, cases are reported voluntarily. As of November 2, 2024, there have been 5,157 cases of varicella reported, excluding any cases from NYC.
Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Suggested Reading
CDC. Nationally Notifiable Infectious Diseases and Conditions, United States: Weekly Tables. https://wonder.cdc.gov/nndss/nndss_weekly_tables_menu.asp.
Graham KA et al. Varicella Outbreak Among Recent Arrivals to New York City, 2022-2024. MMWR Morb Mortal Wkly Rep. 2024 May 30;73(21):478-483. doi: 10.15585/mmwr.mm7321a1.
Marin M et al. Health and Economic Impact of the United States Varicella Vaccination Program, 1996-2020. J Infect Dis. 2022 Oct 21;226(Suppl 4):S463-S469. doi: 10.1093/infdis/jiac271.
Varicella-Zoster Virus Infections in Kimberkin DW et al, eds. Red Book: 2024 Report of the Committee on Infectious Diseases, 33rd Edition. American Academy of Pediatrics, 2024:938-951. https://www.aap.org/Red-Book-2024-Report-of-the-Committee-on-Infectious-Diseases-33rd-Edition-Paperback?srsltid=AfmBOoqyF60rR9ZwQ5jA8AouNhtRRTyPLnc_r7HWw7JVYV8v33Hr2vQS.
Practitioners providing care to children are familiar with the childhood immunization schedule and routinely administer varicella vaccine at the 12-month and 4- to 5-year visits. However, when is the last time most of us or any of the current trainees have seen a case?
Briefly, varicella is a highly contagious disease caused by varicella-zoster virus (VZV). It is characterized by a generalized pruritic erythematous rash in various stages of development beginning as macules, progressing to papules, and ultimately becoming vesicular lesions on an erythematous base (“dewdrop on a rose petal”) and resolves with crusting of the lesion (Figure 1). It has an incubation period of 10-21 days with symptoms usually developing within 14-16 days after exposure. The vesicular rash must be differentiated from enterovirus, Staphylococcus aureus, contact dermatitis, or insect bites, which initially may be difficult. Approximately 50% of children can have symptoms including fever, malaise, anorexia, headache, and occasionally, mild abdominal pain in the 24-48 hours prior to the appearance of rash. Lesions usually first appear on the scalp, face, or trunk in successive crops over several days. A person with varicella has lesions in various stages.
In a normal host, new vesicle formation usually stops within 4 days, and most lesions have fully crusted by day 6. VZV establishes latency in sensory ganglia and may reactivate years or decades later to cause herpes zoster (HZ). Most healthy children with varicella recover without sequelae so the disease is generally regarded as benign. However, varicella can lead to serious complications and deaths in healthy as well as immunocompromised persons.
Complications of Varicella: bacterial superinfection of skin lesions most often with Streptococcus pyogenes or S aureus manifested as cellulitis, myositis, or necrotizing fasciitis; neurologic complications include cerebellar ataxia and encephalitis with the latter seen most often in adults. Pneumonia occurs most often in adults, especially those infected during pregnancy. Another concern, infection during the first 20 weeks of pregnancy can lead to fetal death or severe birth defects, including limb hypoplasia, cutaneous scarring, ocular abnormalities, and central nervous system damage (congenital varicella syndrome).
The risk for development of severe disseminated disease was first noted in the 1960s as treatments for leukemia in children improved. They were surviving their cancer only to develop severe and often fatal varicella. Today it is recognized that development of disseminated disease is a risk for all infected persons with impaired T cell function, malignancies, HIV, or receiving immunosuppressive therapy.
Reye’s syndrome is rarely seen today since taking salicylates while infected with VZV was identified as a predisposing factor for development.
VZV is only found in humans and transmission is person to person or airborne. The secondary household attack rate is approximately 90%. In contrast, the secondary attack rates in classrooms may be as low as 12%-33%. Transmission rates in the tropics for unexplained reasons are also lower.
Vaccine History: Why do we rarely see this disease anymore? Varicella, a live attenuated vaccine, was developed in 1974 by Dr. Michiaki Takahashi. It remains the only vaccine directed against a herpes group virus. In 1979, the Collaborative Varicella Vaccine Study Group was established at the National Institutes of Health (NIH) and additional safety and efficacy trials were conducted in the United States initially in leukemic patients in remission and later in healthy children, which supported Takahashi’s data. Licensure of varicella vaccine was granted in 1995. That same year, due to continuing disease and societal burden, the United States was the first country to incorporate varicella into the routine childhood immunization schedule, which resulted in significant reductions in cases. To further improve control of varicella, in 2007 vaccine recommendations were revised and a routine two-dose schedule was implemented. The impact of varicella disease pre- and post-vaccine licensure is illustrated in Figure 2. Not listed, is that in the pre-vaccine era, there were approximately 44 cases of congenital varicella syndrome annually.

As of 2023 only 23% (45/195) of nations routinely administer this vaccine and 4% (8/195) have restricted recommendations. The remaining 73% of countries do not offer the vaccine, including all countries on the African continent, and Cuba, Guatemala, Haiti, Honduras, India, Jordan, Lebanon, Philippines, Portugal, and Venezuela to list a few.
Varicella Outbreak: In October 2022, New York City (NYC) identified a varicella outbreak primarily involving persons who recently migrated from Central and South America and lived in a shelter in NYC or residential facility (n = 105); the outbreak is ongoing. As of March 8, 2024, 873 cases (53%) were among children aged 4-18 years and 91.9% had no documentation of varicella vaccine at time of symptom onset. There were 28 hospitalizations, and no deaths reported. The most common sources of transmission were the residential facilities (41.3%) and importation or possible importation (39.4%). School transmission accounted for only 1.2% of cases.
Most migrants arrived from countries where varicella vaccination is not part of the routine childhood immunization schedule. Although most cases occurred in children, almost 30% occurred in adults. Many of the migrants arrived from tropical countries where susceptibility rates are also higher in adults. This outbreak is a reminder of the importance of limiting disease transmission by maintaining high vaccination rates. To curtail this outbreak, approximately 27,000 doses of varicella vaccine were administered to the arriving migrants. In addition, MMR, COVID-19, influenza, and all routine pediatric vaccines required for school entry were administered. Temporary closure of the residential facilities were required. Education was provided to residents regarding immunizations as well as assistance to help them establish a primary care home. Multiple agencies were mobilized to successfully coordinate these efforts.
Take Home Message
1. Each country has its own routine immunization schedule. It may not include all vaccines recommended in the US schedule. When questioned I’m frequently told that immunizations are up to date, only to review records and find they are not, especially when it is related to MMR. It is often administered at 9 months and/or MR or MM is administered depending on the country. As reported here, varicella is a routine vaccine in only 45 countries.
2.
3. Once an outbreak has been identified, the infrastructure to manage and contain it must already be established. In most instances there will be a need for a rapid and often large-scale effort involving multiple agencies including local health care providers.
4. Not all diseases are reportable. Only deaths by varicella are nationally notifiable. Otherwise, cases are reported voluntarily. As of November 2, 2024, there have been 5,157 cases of varicella reported, excluding any cases from NYC.
Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Suggested Reading
CDC. Nationally Notifiable Infectious Diseases and Conditions, United States: Weekly Tables. https://wonder.cdc.gov/nndss/nndss_weekly_tables_menu.asp.
Graham KA et al. Varicella Outbreak Among Recent Arrivals to New York City, 2022-2024. MMWR Morb Mortal Wkly Rep. 2024 May 30;73(21):478-483. doi: 10.15585/mmwr.mm7321a1.
Marin M et al. Health and Economic Impact of the United States Varicella Vaccination Program, 1996-2020. J Infect Dis. 2022 Oct 21;226(Suppl 4):S463-S469. doi: 10.1093/infdis/jiac271.
Varicella-Zoster Virus Infections in Kimberkin DW et al, eds. Red Book: 2024 Report of the Committee on Infectious Diseases, 33rd Edition. American Academy of Pediatrics, 2024:938-951. https://www.aap.org/Red-Book-2024-Report-of-the-Committee-on-Infectious-Diseases-33rd-Edition-Paperback?srsltid=AfmBOoqyF60rR9ZwQ5jA8AouNhtRRTyPLnc_r7HWw7JVYV8v33Hr2vQS.
Summer Is Not Over: Let's Talk About Recreational Water–Associated Illnesses
Recently I was in Wyoming. As I rode down the Snake River, the guide pointed out tree trunks that had been chewed on by beavers. Days later I joined a local friend for a hike to Taggart Lake. Upon reaching the end of the trail as I began to cast my eyes on the magnificent scenery, I could not help but notice several children, including toddlers, playing in the fresh warm water. The next thing out of my friend’s mouth was “You know there is Giardia in there.” Little did she know, she and the guide had just helped me select a topic for ID Consult.
Giardia, aka ”beaver fever,” was discussed in detail in this column as part of the differential of a diarrheal illness by Christopher J. Harrison, MD. However, it is the perfect time of year to revisit other recreational water–associated illnesses.
Infections acquired during recreational water activity can lead to illnesses involving the gastrointestinal tract, central nervous system, respiratory tract, skin, eyes, and ears. Pathogens, chemicals, and toxins are transmitted by ingestion, contact with contaminated water or a sick individual or animal, and inhalation of aerosols. The National Waterborne Disease and Outbreak Surveillance System (WBDOSS) collects data on waterborne disease and outbreaks associated with recreational water, drinking water, and environmental and undetermined exposures to water. All reporting to the Centers for Disease Control and Prevention (CDC) is voluntary. However, mandatory pathogen reporting requirements can vary by state. Ideally, once an agency has completed the outbreak investigation, the definitive cause and source will be determined, and interventions to prevent future outbreaks implemented.
Treated Versus Untreated Water
One useful way to help narrow the etiology of a patient’s symptoms is to consider those illnesses associated with treated water venues (e.g., pools, hot tubs, water parks) versus untreated water venues (e.g., rivers, lakes, oceans). Parents may forget to offer that information since they may not perceive a connection between water exposure and the illness, especially if they traveled within the US.
In 2021, the CDC reported results of data submitted between 2015 and 2019 from treated recreational water facilities. Of the 208 outbreaks, most (96%) were associated with public pools, hot tubs, or water playgrounds. These outbreaks resulted in at least 3,646 cases of illness, 286 hospitalizations, and 13 deaths. Overall infectious etiologies were the primary cause of illness. Of the 155 outbreaks with a confirmed etiology, Cryptosporidium was the causative pathogen in 49% of the outbreaks and accounted for 84% (2,492) of cases, while Legionella caused 42% of outbreaks, accounted for 13% (354) of cases, and was responsible for all 13 deaths. Slightly more than half (107 of 208) of the outbreaks started between June-August with Cryptosporidium accounting for 63 of the outbreaks during that period. A little more than one-third were associated with a hotel or resort. The majority of hotel recreational water–associated illnesses was associated with hot tubs. Of the 53 outbreaks without a confirmed etiology, 20 were suspected to have a chemical related etiology (excess chlorine, altered pool chemistry).
In contrast, there were 140 untreated recreational water outbreaks reported between 2000 and 2014 from 35 states and Guam involving 4,958 cases and 2 deaths. The etiology was confirmed for 103 (74%) outbreaks including 5 that had multiple etiologies and 8 due to toxins or chemicals; 7 of 8 toxins were from harmful algal blooms. Enteric pathogens were the etiology in 84% of outbreaks including: Norovirus (n = 1459), Shigella (n = 362) Avian schistosomes (n = 345), Cryptosporidium (n = 314) and Escherichia coli (n = 155).There were 24 cases of Giardia. The two deaths were due to Naegleria fowleri. The top 2 settings for these outbreaks were public parks (36%) and beaches (32%) with most outbreaks (n = 117) being associated with a lake /pond venue. Most outbreaks began between June and August.
The major differences between the two types of recreational water–associated illnesses are their most common settings and etiologies. With that in mind, let us briefly review the most common etiology from each venue.
Treated Water Venue: Cryptosporidiosis
Cryptosporidium is an oocyst-forming protozoa that causes a self-limited watery, nonbloody diarrhea which usually resolves within 10-14 days. Most patients have associated abdominal cramps, fever, and vomiting although infected persons can be asymptomatic. Infection in the immunocompromised potentially can lead to profuse and prolonged diarrhea. Oocysts are excreted in the feces of infected hosts and as little as 10 can cause infection. They can survive extreme environmental conditions in water and soil for several months and even survive up to 7 days in a properly chlorinated pool. Transmission occurs between humans via contaminated food and water or from infected animals. Oocysts have been isolated in raw or unpasteurized milk and apple cider. Incidence is highest in children 1 through 4 years of age.
Diagnosis today is usually via molecular methods (nucleic acid amplification tests, aka NAATs), due to their high sensitivity and specificity and is the preferred method. These tests can identify multiple gastrointestinal tract pathogens with a single assay. Diagnosis by microscopy or fecal immunoassay antigens are still available. Treatment is supportive in most cases. If needed, a 3-day course of nitazoxanide can be prescribed. Immunocompromised patients should be managed in consultation with an infectious disease specialist.
Untreated Water Venue: Norovirus
Norovirus is a viral illness characterized by the abrupt onset of vomiting and/or watery diarrhea, usually associated with nausea and abdominal cramps. Symptoms persist 24-72 hours, however they may be prolonged in the immunocompromised and persons at the extremes of the age spectrum. Norovirus has replaced rotavirus as the major cause of medically attended gastroenteritis. While a major cause of recreational water–associated illnesses, high attack rates also occur in semi closed communities including cruise ships, childcare centers, and schools. Transmission is fecal-oral, vomitus oral, person to person, by ingestion of contaminated food and water or touching contaminated surfaces with subsequent touching of the mouth. Asymptomatic viral shedding may occur, especially in children. Prolonged shedding (> 6 mos.) has been reported in immunocompromised hosts.
Molecular diagnosis with stool is utilized most often. Treatment is supportive.
Take Home Message
When evaluating your patients for an acute gastrointestinal illness, consider water-related activities and their potential for being the source. Encourage patients not to ignore posted advisories on beaches, to not swim if they have diarrhea, not to swallow the water they swim in and to minimize water entering their nose while swimming in warm freshwater. If you start seeing several patients with similar symptoms and/or etiology, consider contacting your local or state health department. It could be the beginning of an outbreak.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She has no relevant financial disclosures.
Suggested Readings
Graciaa DS et al. Outbreaks Associated with Untreated Recreational Water — United States, 2000–2014. MMWR Morb Mortal Wkly Rep. 2018 Jun 29;67(25):701-706. doi: 10.15585/mmwr.mm6725a1.
Hlavsa MC et al. Outbreaks Associated with Treated Recreational Water — United States, 2015–2019. MMWR Morb Mortal Wkly Rep. 2021;70:733–738. doi: 10.15585/mmwr.mm7020a1.
Kimberlin DW et al., eds. Red Book Report of the Committee on Infectious Diseases. 33rd ed. American Academy of Pediatrics. 2024. Cryptosporidiosis, p 338-40 and Norovirus, p 622-624.Waterborne Outbreaks Summary Reports. CDC. 2024 April 18.
Recently I was in Wyoming. As I rode down the Snake River, the guide pointed out tree trunks that had been chewed on by beavers. Days later I joined a local friend for a hike to Taggart Lake. Upon reaching the end of the trail as I began to cast my eyes on the magnificent scenery, I could not help but notice several children, including toddlers, playing in the fresh warm water. The next thing out of my friend’s mouth was “You know there is Giardia in there.” Little did she know, she and the guide had just helped me select a topic for ID Consult.
Giardia, aka ”beaver fever,” was discussed in detail in this column as part of the differential of a diarrheal illness by Christopher J. Harrison, MD. However, it is the perfect time of year to revisit other recreational water–associated illnesses.
Infections acquired during recreational water activity can lead to illnesses involving the gastrointestinal tract, central nervous system, respiratory tract, skin, eyes, and ears. Pathogens, chemicals, and toxins are transmitted by ingestion, contact with contaminated water or a sick individual or animal, and inhalation of aerosols. The National Waterborne Disease and Outbreak Surveillance System (WBDOSS) collects data on waterborne disease and outbreaks associated with recreational water, drinking water, and environmental and undetermined exposures to water. All reporting to the Centers for Disease Control and Prevention (CDC) is voluntary. However, mandatory pathogen reporting requirements can vary by state. Ideally, once an agency has completed the outbreak investigation, the definitive cause and source will be determined, and interventions to prevent future outbreaks implemented.
Treated Versus Untreated Water
One useful way to help narrow the etiology of a patient’s symptoms is to consider those illnesses associated with treated water venues (e.g., pools, hot tubs, water parks) versus untreated water venues (e.g., rivers, lakes, oceans). Parents may forget to offer that information since they may not perceive a connection between water exposure and the illness, especially if they traveled within the US.
In 2021, the CDC reported results of data submitted between 2015 and 2019 from treated recreational water facilities. Of the 208 outbreaks, most (96%) were associated with public pools, hot tubs, or water playgrounds. These outbreaks resulted in at least 3,646 cases of illness, 286 hospitalizations, and 13 deaths. Overall infectious etiologies were the primary cause of illness. Of the 155 outbreaks with a confirmed etiology, Cryptosporidium was the causative pathogen in 49% of the outbreaks and accounted for 84% (2,492) of cases, while Legionella caused 42% of outbreaks, accounted for 13% (354) of cases, and was responsible for all 13 deaths. Slightly more than half (107 of 208) of the outbreaks started between June-August with Cryptosporidium accounting for 63 of the outbreaks during that period. A little more than one-third were associated with a hotel or resort. The majority of hotel recreational water–associated illnesses was associated with hot tubs. Of the 53 outbreaks without a confirmed etiology, 20 were suspected to have a chemical related etiology (excess chlorine, altered pool chemistry).
In contrast, there were 140 untreated recreational water outbreaks reported between 2000 and 2014 from 35 states and Guam involving 4,958 cases and 2 deaths. The etiology was confirmed for 103 (74%) outbreaks including 5 that had multiple etiologies and 8 due to toxins or chemicals; 7 of 8 toxins were from harmful algal blooms. Enteric pathogens were the etiology in 84% of outbreaks including: Norovirus (n = 1459), Shigella (n = 362) Avian schistosomes (n = 345), Cryptosporidium (n = 314) and Escherichia coli (n = 155).There were 24 cases of Giardia. The two deaths were due to Naegleria fowleri. The top 2 settings for these outbreaks were public parks (36%) and beaches (32%) with most outbreaks (n = 117) being associated with a lake /pond venue. Most outbreaks began between June and August.
The major differences between the two types of recreational water–associated illnesses are their most common settings and etiologies. With that in mind, let us briefly review the most common etiology from each venue.
Treated Water Venue: Cryptosporidiosis
Cryptosporidium is an oocyst-forming protozoa that causes a self-limited watery, nonbloody diarrhea which usually resolves within 10-14 days. Most patients have associated abdominal cramps, fever, and vomiting although infected persons can be asymptomatic. Infection in the immunocompromised potentially can lead to profuse and prolonged diarrhea. Oocysts are excreted in the feces of infected hosts and as little as 10 can cause infection. They can survive extreme environmental conditions in water and soil for several months and even survive up to 7 days in a properly chlorinated pool. Transmission occurs between humans via contaminated food and water or from infected animals. Oocysts have been isolated in raw or unpasteurized milk and apple cider. Incidence is highest in children 1 through 4 years of age.
Diagnosis today is usually via molecular methods (nucleic acid amplification tests, aka NAATs), due to their high sensitivity and specificity and is the preferred method. These tests can identify multiple gastrointestinal tract pathogens with a single assay. Diagnosis by microscopy or fecal immunoassay antigens are still available. Treatment is supportive in most cases. If needed, a 3-day course of nitazoxanide can be prescribed. Immunocompromised patients should be managed in consultation with an infectious disease specialist.
Untreated Water Venue: Norovirus
Norovirus is a viral illness characterized by the abrupt onset of vomiting and/or watery diarrhea, usually associated with nausea and abdominal cramps. Symptoms persist 24-72 hours, however they may be prolonged in the immunocompromised and persons at the extremes of the age spectrum. Norovirus has replaced rotavirus as the major cause of medically attended gastroenteritis. While a major cause of recreational water–associated illnesses, high attack rates also occur in semi closed communities including cruise ships, childcare centers, and schools. Transmission is fecal-oral, vomitus oral, person to person, by ingestion of contaminated food and water or touching contaminated surfaces with subsequent touching of the mouth. Asymptomatic viral shedding may occur, especially in children. Prolonged shedding (> 6 mos.) has been reported in immunocompromised hosts.
Molecular diagnosis with stool is utilized most often. Treatment is supportive.
Take Home Message
When evaluating your patients for an acute gastrointestinal illness, consider water-related activities and their potential for being the source. Encourage patients not to ignore posted advisories on beaches, to not swim if they have diarrhea, not to swallow the water they swim in and to minimize water entering their nose while swimming in warm freshwater. If you start seeing several patients with similar symptoms and/or etiology, consider contacting your local or state health department. It could be the beginning of an outbreak.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She has no relevant financial disclosures.
Suggested Readings
Graciaa DS et al. Outbreaks Associated with Untreated Recreational Water — United States, 2000–2014. MMWR Morb Mortal Wkly Rep. 2018 Jun 29;67(25):701-706. doi: 10.15585/mmwr.mm6725a1.
Hlavsa MC et al. Outbreaks Associated with Treated Recreational Water — United States, 2015–2019. MMWR Morb Mortal Wkly Rep. 2021;70:733–738. doi: 10.15585/mmwr.mm7020a1.
Kimberlin DW et al., eds. Red Book Report of the Committee on Infectious Diseases. 33rd ed. American Academy of Pediatrics. 2024. Cryptosporidiosis, p 338-40 and Norovirus, p 622-624.Waterborne Outbreaks Summary Reports. CDC. 2024 April 18.
Recently I was in Wyoming. As I rode down the Snake River, the guide pointed out tree trunks that had been chewed on by beavers. Days later I joined a local friend for a hike to Taggart Lake. Upon reaching the end of the trail as I began to cast my eyes on the magnificent scenery, I could not help but notice several children, including toddlers, playing in the fresh warm water. The next thing out of my friend’s mouth was “You know there is Giardia in there.” Little did she know, she and the guide had just helped me select a topic for ID Consult.
Giardia, aka ”beaver fever,” was discussed in detail in this column as part of the differential of a diarrheal illness by Christopher J. Harrison, MD. However, it is the perfect time of year to revisit other recreational water–associated illnesses.
Infections acquired during recreational water activity can lead to illnesses involving the gastrointestinal tract, central nervous system, respiratory tract, skin, eyes, and ears. Pathogens, chemicals, and toxins are transmitted by ingestion, contact with contaminated water or a sick individual or animal, and inhalation of aerosols. The National Waterborne Disease and Outbreak Surveillance System (WBDOSS) collects data on waterborne disease and outbreaks associated with recreational water, drinking water, and environmental and undetermined exposures to water. All reporting to the Centers for Disease Control and Prevention (CDC) is voluntary. However, mandatory pathogen reporting requirements can vary by state. Ideally, once an agency has completed the outbreak investigation, the definitive cause and source will be determined, and interventions to prevent future outbreaks implemented.
Treated Versus Untreated Water
One useful way to help narrow the etiology of a patient’s symptoms is to consider those illnesses associated with treated water venues (e.g., pools, hot tubs, water parks) versus untreated water venues (e.g., rivers, lakes, oceans). Parents may forget to offer that information since they may not perceive a connection between water exposure and the illness, especially if they traveled within the US.
In 2021, the CDC reported results of data submitted between 2015 and 2019 from treated recreational water facilities. Of the 208 outbreaks, most (96%) were associated with public pools, hot tubs, or water playgrounds. These outbreaks resulted in at least 3,646 cases of illness, 286 hospitalizations, and 13 deaths. Overall infectious etiologies were the primary cause of illness. Of the 155 outbreaks with a confirmed etiology, Cryptosporidium was the causative pathogen in 49% of the outbreaks and accounted for 84% (2,492) of cases, while Legionella caused 42% of outbreaks, accounted for 13% (354) of cases, and was responsible for all 13 deaths. Slightly more than half (107 of 208) of the outbreaks started between June-August with Cryptosporidium accounting for 63 of the outbreaks during that period. A little more than one-third were associated with a hotel or resort. The majority of hotel recreational water–associated illnesses was associated with hot tubs. Of the 53 outbreaks without a confirmed etiology, 20 were suspected to have a chemical related etiology (excess chlorine, altered pool chemistry).
In contrast, there were 140 untreated recreational water outbreaks reported between 2000 and 2014 from 35 states and Guam involving 4,958 cases and 2 deaths. The etiology was confirmed for 103 (74%) outbreaks including 5 that had multiple etiologies and 8 due to toxins or chemicals; 7 of 8 toxins were from harmful algal blooms. Enteric pathogens were the etiology in 84% of outbreaks including: Norovirus (n = 1459), Shigella (n = 362) Avian schistosomes (n = 345), Cryptosporidium (n = 314) and Escherichia coli (n = 155).There were 24 cases of Giardia. The two deaths were due to Naegleria fowleri. The top 2 settings for these outbreaks were public parks (36%) and beaches (32%) with most outbreaks (n = 117) being associated with a lake /pond venue. Most outbreaks began between June and August.
The major differences between the two types of recreational water–associated illnesses are their most common settings and etiologies. With that in mind, let us briefly review the most common etiology from each venue.
Treated Water Venue: Cryptosporidiosis
Cryptosporidium is an oocyst-forming protozoa that causes a self-limited watery, nonbloody diarrhea which usually resolves within 10-14 days. Most patients have associated abdominal cramps, fever, and vomiting although infected persons can be asymptomatic. Infection in the immunocompromised potentially can lead to profuse and prolonged diarrhea. Oocysts are excreted in the feces of infected hosts and as little as 10 can cause infection. They can survive extreme environmental conditions in water and soil for several months and even survive up to 7 days in a properly chlorinated pool. Transmission occurs between humans via contaminated food and water or from infected animals. Oocysts have been isolated in raw or unpasteurized milk and apple cider. Incidence is highest in children 1 through 4 years of age.
Diagnosis today is usually via molecular methods (nucleic acid amplification tests, aka NAATs), due to their high sensitivity and specificity and is the preferred method. These tests can identify multiple gastrointestinal tract pathogens with a single assay. Diagnosis by microscopy or fecal immunoassay antigens are still available. Treatment is supportive in most cases. If needed, a 3-day course of nitazoxanide can be prescribed. Immunocompromised patients should be managed in consultation with an infectious disease specialist.
Untreated Water Venue: Norovirus
Norovirus is a viral illness characterized by the abrupt onset of vomiting and/or watery diarrhea, usually associated with nausea and abdominal cramps. Symptoms persist 24-72 hours, however they may be prolonged in the immunocompromised and persons at the extremes of the age spectrum. Norovirus has replaced rotavirus as the major cause of medically attended gastroenteritis. While a major cause of recreational water–associated illnesses, high attack rates also occur in semi closed communities including cruise ships, childcare centers, and schools. Transmission is fecal-oral, vomitus oral, person to person, by ingestion of contaminated food and water or touching contaminated surfaces with subsequent touching of the mouth. Asymptomatic viral shedding may occur, especially in children. Prolonged shedding (> 6 mos.) has been reported in immunocompromised hosts.
Molecular diagnosis with stool is utilized most often. Treatment is supportive.
Take Home Message
When evaluating your patients for an acute gastrointestinal illness, consider water-related activities and their potential for being the source. Encourage patients not to ignore posted advisories on beaches, to not swim if they have diarrhea, not to swallow the water they swim in and to minimize water entering their nose while swimming in warm freshwater. If you start seeing several patients with similar symptoms and/or etiology, consider contacting your local or state health department. It could be the beginning of an outbreak.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She has no relevant financial disclosures.
Suggested Readings
Graciaa DS et al. Outbreaks Associated with Untreated Recreational Water — United States, 2000–2014. MMWR Morb Mortal Wkly Rep. 2018 Jun 29;67(25):701-706. doi: 10.15585/mmwr.mm6725a1.
Hlavsa MC et al. Outbreaks Associated with Treated Recreational Water — United States, 2015–2019. MMWR Morb Mortal Wkly Rep. 2021;70:733–738. doi: 10.15585/mmwr.mm7020a1.
Kimberlin DW et al., eds. Red Book Report of the Committee on Infectious Diseases. 33rd ed. American Academy of Pediatrics. 2024. Cryptosporidiosis, p 338-40 and Norovirus, p 622-624.Waterborne Outbreaks Summary Reports. CDC. 2024 April 18.
Summertime and Mosquitoes Are Breeding
There are over 3700 types of mosquitoes worldwide and over 200 types in the continental United States, of which only 12 are associated with transmitting diseases to humans. The majority are just a nuisance. Since they cannot readily be distinguished, strategies to prevent any bites are recommended.
West Nile Virus
In the US, West Nile virus (WNV) is the leading cause of neuroinvasive arboviral disease. Just hearing the name took me back to New York in 1999 when sightings of dead birds around the city and boroughs were reported daily. The virus was isolated that same year. The enzootic circle occurs between mosquitoes and birds, which are the primary vertebrate host via the bite of Culex mosquitoes. After a bite from an infected mosquito, humans are usually a dead-end host since the level and duration of viremia needed to infect another mosquito is insufficient.
Human-to-human transmission is documented through blood transfusion and solid organ transplantation. Vertical transmission is rarely described. Initially isolated in New York, WNV quickly spread across North America and has been isolated in every continent except Antarctica. Most cases occur in the summer and autumn.
Most infected individuals are asymptomatic. Those who do develop symptoms have fever, headache, myalgia, arthralgia, nausea, vomiting, and a transient rash. Less than 1% develop meningitis/encephalitis symptoms similar to other causes of aseptic meningitis. Those with encephalitis in addition to fever and headache may have altered mental status and focal neurologic deficits including flaccid paralysis or movement disorders.
Detection of anti-WNV IgM antibodies (AB) in serum or CSF is the most common way to make the diagnosis. IgM AB usually is present within 3-8 days after onset of symptoms and persists up to 90 days. Data from ArboNET, the national arboviral surveillance system managed by Centers for Disease Control and Prevention and state health departments, reveal that from 1999 to 2022 there were 56,575 cases of WNV including 28,684 cases of neuroinvasive disease. In 2023 there were 2,406 and 1,599 cases, respectively. Those historic totals for WNV are 10 times greater than the totals for all the other etiologies of neuroinvasive arboviral diseases in the US combined (Jamestown Canyon, LaCrosse, St. Louis, and Eastern Equine encephalitis n = 1813).
Remember to include WNV in your differential of a febrile patient with neurologic symptoms, mosquito bites, blood transfusions, and organ transplantation. Treatment is supportive care.
The US began screening all blood donations for WNV in 2003. Organ donor screening is not universal.
Dengue
Dengue, another arbovirus, is transmitted by bites of infected Aedes aegypti and Aedes albopictus mosquitoes, which prefer to feed during the daytime. There are four dengue virus serotypes: DENV-1 DENV-2, DENV-3 and DENV-4. In endemic areas, all four serotypes are usually co-circulating and people can be infected by each one.
Long-term immunity is type specific. Heterologous protection lasts only a few months. Dengue is endemic throughout the tropics and subtropics of Asia, Africa, and the Americas. Approximately 53% of the world’s population live in an area where dengue transmission can occur. In the US, most cases are reported from Puerto Rico. Dengue is endemic in the following US territories: Puerto Rico, US Virgin Islands, American Samoa, and free associated states. Most cases reported on the mainland are travel related. However, locally acquired dengue has been reported. From 2010 to 2023 Hawaii reported 250 cases, Florida 438, and Texas 40 locally acquired cases. During that same period, Puerto Rico reported more than 32,000 cases. It is the leading cause of febrile illness for travelers returning from the Caribbean, Latin America, and South Asia. Peru is currently experiencing an outbreak with more than 25,000 cases reported since January 2024. Most cases of dengue occur in adolescents and young adults. Severe disease occurs most often in infants, those with underlying chronic disease, pregnant women, and persons infected with dengue for the second time.
Symptoms range from a mild febrile illness to severe disease associated with hemorrhage and shock. Onset is usually 7-10 days after infection and symptoms include high fever, severe headache, retro-orbital pain, arthralgia and myalgias, nausea, and vomiting; some may develop a generalized rash.
The World Health Organization (WHO) classifies dengue as 1) dengue with or without warning signs for progression of disease and 2) severe dengue. Warning signs for disease progression include abdominal pain or tenderness, persistent vomiting, fluid accumulation (e.g., ascites, pericardial or pleural effusion), mucosal bleeding, restlessness, postural hypotension, liver enlargement greater than 2 cm. Severe dengue is defined as any sign of severe plasma leakage leading to shock, severe bleeding or organ failure, or fluid accumulation with respiratory distress. Management is supportive care.
Prevention: In the US, Dengvaxia, a live attenuated tetravalent vaccine, is approved for use in children aged 9–16 years with laboratory-confirmed previous dengue virus infection and living in areas where dengue is endemic. It is administered at 0, 6, and 12 months. It is not available for purchase on the mainland. Continued control of the vector and personal protection is necessary to prevent recurrent infections.
CHIKV
Chikungunya (CHIKV), which means “that which bends up” in the Mkonde language of Tanzania, refers to the appearance of the person with severe usually symmetric arthralgias characteristic for this infection that otherwise is often clinically confused with dengue and Zika. It too is transmitted by A. aegypti and A. albopictus and is prevalent in tropical Africa, Asia, Central and South America, and the Caribbean. Like dengue it is predominantly an urban disease. The WHO reported the first case in the Western Hemisphere in Saint Martin in December 2013. By August 2014, 31 additional territories and Caribbean or South American countries reported 576,535 suspected cases. Florida first reported locally acquired CHIKV in June 2014. By December an additional 11 cases had been identified. Texas reported one case in 2015. Diagnosis is with IgM ab or PCR. Treatment is supportive with most recovering from acute illness within 2 weeks. Data in adults indicate 40-52% may develop chronic or recurrent joint pain.
Prevention: IXCHIQ, a live attenuated vaccine, was licensed in November 2023 and recommended by the CDC in February 2024 for use in persons at least 18 years of age with travel to destinations where there is a CHIKV outbreak. It may be considered for persons traveling to a country or territory without an outbreak but with evidence of CHIKV transmission among humans within the last 5 years and those staying in endemic areas for a cumulative period of at least 6 months over a 2-year period. Specific recommendations for lab workers and persons older than 65 years were also made. This is good news for your older patients who may be participating in mission trips, volunteering, studying abroad, or just vacationing in an endemic area. Adolescent vaccine trials are ongoing and pediatric trials will soon be initiated. In addition, vector control and use of personal protective measures cannot be emphasized enough.
There are several other mosquito borne diseases, however our discussion here is limited to three. Why these three? WNV as a reminder that it is the most common neuroinvasive agent in the US. Dengue and CHIKV because they are not endemic in the US so they might not routinely be considered in febrile patients; both diseases have been reported and acquired on the mainland and your patients may travel to an endemic area and return home with an unwanted souvenir. You will be ready for them.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Suggested Reading
Chikungunya. Centers for Disease Control and Prevention. 2024. https://www.cdc.gov/vaccines/acip/recommendations.html.
Fagrem AC et al. West Nile and Other Nationally Notifiable Arboviral Diseases–United States, 2021. MMWR Morb Mortal Wkly Rep. 2023 Aug 25;72(34):901-906.
Fever in Returned Travelers, Travel Medicine (Fourth Edition). 2019. doi: 10.1016/B978-0-323-54696-6.00056-2.
Paz-Baily et al. Dengue Vaccine: Recommendations of the Advisory Committee on Immunization Practices, United States, 2021 MMWR Recomm Rep. 2021 Dec 17;70(6):1-16).
Staples JE and Fischer M. Chikungunya virus in the Americas — what a vectorborne pathogen can do. N Engl J Med. 2014 Sep 4;371(10):887-9.
Mosquitoes and Diseases A-Z, Centers for Disease Control and Prevention. https://www.cdc.gov/mosquitoes/about/diseases.html.
There are over 3700 types of mosquitoes worldwide and over 200 types in the continental United States, of which only 12 are associated with transmitting diseases to humans. The majority are just a nuisance. Since they cannot readily be distinguished, strategies to prevent any bites are recommended.
West Nile Virus
In the US, West Nile virus (WNV) is the leading cause of neuroinvasive arboviral disease. Just hearing the name took me back to New York in 1999 when sightings of dead birds around the city and boroughs were reported daily. The virus was isolated that same year. The enzootic circle occurs between mosquitoes and birds, which are the primary vertebrate host via the bite of Culex mosquitoes. After a bite from an infected mosquito, humans are usually a dead-end host since the level and duration of viremia needed to infect another mosquito is insufficient.
Human-to-human transmission is documented through blood transfusion and solid organ transplantation. Vertical transmission is rarely described. Initially isolated in New York, WNV quickly spread across North America and has been isolated in every continent except Antarctica. Most cases occur in the summer and autumn.
Most infected individuals are asymptomatic. Those who do develop symptoms have fever, headache, myalgia, arthralgia, nausea, vomiting, and a transient rash. Less than 1% develop meningitis/encephalitis symptoms similar to other causes of aseptic meningitis. Those with encephalitis in addition to fever and headache may have altered mental status and focal neurologic deficits including flaccid paralysis or movement disorders.
Detection of anti-WNV IgM antibodies (AB) in serum or CSF is the most common way to make the diagnosis. IgM AB usually is present within 3-8 days after onset of symptoms and persists up to 90 days. Data from ArboNET, the national arboviral surveillance system managed by Centers for Disease Control and Prevention and state health departments, reveal that from 1999 to 2022 there were 56,575 cases of WNV including 28,684 cases of neuroinvasive disease. In 2023 there were 2,406 and 1,599 cases, respectively. Those historic totals for WNV are 10 times greater than the totals for all the other etiologies of neuroinvasive arboviral diseases in the US combined (Jamestown Canyon, LaCrosse, St. Louis, and Eastern Equine encephalitis n = 1813).
Remember to include WNV in your differential of a febrile patient with neurologic symptoms, mosquito bites, blood transfusions, and organ transplantation. Treatment is supportive care.
The US began screening all blood donations for WNV in 2003. Organ donor screening is not universal.
Dengue
Dengue, another arbovirus, is transmitted by bites of infected Aedes aegypti and Aedes albopictus mosquitoes, which prefer to feed during the daytime. There are four dengue virus serotypes: DENV-1 DENV-2, DENV-3 and DENV-4. In endemic areas, all four serotypes are usually co-circulating and people can be infected by each one.
Long-term immunity is type specific. Heterologous protection lasts only a few months. Dengue is endemic throughout the tropics and subtropics of Asia, Africa, and the Americas. Approximately 53% of the world’s population live in an area where dengue transmission can occur. In the US, most cases are reported from Puerto Rico. Dengue is endemic in the following US territories: Puerto Rico, US Virgin Islands, American Samoa, and free associated states. Most cases reported on the mainland are travel related. However, locally acquired dengue has been reported. From 2010 to 2023 Hawaii reported 250 cases, Florida 438, and Texas 40 locally acquired cases. During that same period, Puerto Rico reported more than 32,000 cases. It is the leading cause of febrile illness for travelers returning from the Caribbean, Latin America, and South Asia. Peru is currently experiencing an outbreak with more than 25,000 cases reported since January 2024. Most cases of dengue occur in adolescents and young adults. Severe disease occurs most often in infants, those with underlying chronic disease, pregnant women, and persons infected with dengue for the second time.
Symptoms range from a mild febrile illness to severe disease associated with hemorrhage and shock. Onset is usually 7-10 days after infection and symptoms include high fever, severe headache, retro-orbital pain, arthralgia and myalgias, nausea, and vomiting; some may develop a generalized rash.
The World Health Organization (WHO) classifies dengue as 1) dengue with or without warning signs for progression of disease and 2) severe dengue. Warning signs for disease progression include abdominal pain or tenderness, persistent vomiting, fluid accumulation (e.g., ascites, pericardial or pleural effusion), mucosal bleeding, restlessness, postural hypotension, liver enlargement greater than 2 cm. Severe dengue is defined as any sign of severe plasma leakage leading to shock, severe bleeding or organ failure, or fluid accumulation with respiratory distress. Management is supportive care.
Prevention: In the US, Dengvaxia, a live attenuated tetravalent vaccine, is approved for use in children aged 9–16 years with laboratory-confirmed previous dengue virus infection and living in areas where dengue is endemic. It is administered at 0, 6, and 12 months. It is not available for purchase on the mainland. Continued control of the vector and personal protection is necessary to prevent recurrent infections.
CHIKV
Chikungunya (CHIKV), which means “that which bends up” in the Mkonde language of Tanzania, refers to the appearance of the person with severe usually symmetric arthralgias characteristic for this infection that otherwise is often clinically confused with dengue and Zika. It too is transmitted by A. aegypti and A. albopictus and is prevalent in tropical Africa, Asia, Central and South America, and the Caribbean. Like dengue it is predominantly an urban disease. The WHO reported the first case in the Western Hemisphere in Saint Martin in December 2013. By August 2014, 31 additional territories and Caribbean or South American countries reported 576,535 suspected cases. Florida first reported locally acquired CHIKV in June 2014. By December an additional 11 cases had been identified. Texas reported one case in 2015. Diagnosis is with IgM ab or PCR. Treatment is supportive with most recovering from acute illness within 2 weeks. Data in adults indicate 40-52% may develop chronic or recurrent joint pain.
Prevention: IXCHIQ, a live attenuated vaccine, was licensed in November 2023 and recommended by the CDC in February 2024 for use in persons at least 18 years of age with travel to destinations where there is a CHIKV outbreak. It may be considered for persons traveling to a country or territory without an outbreak but with evidence of CHIKV transmission among humans within the last 5 years and those staying in endemic areas for a cumulative period of at least 6 months over a 2-year period. Specific recommendations for lab workers and persons older than 65 years were also made. This is good news for your older patients who may be participating in mission trips, volunteering, studying abroad, or just vacationing in an endemic area. Adolescent vaccine trials are ongoing and pediatric trials will soon be initiated. In addition, vector control and use of personal protective measures cannot be emphasized enough.
There are several other mosquito borne diseases, however our discussion here is limited to three. Why these three? WNV as a reminder that it is the most common neuroinvasive agent in the US. Dengue and CHIKV because they are not endemic in the US so they might not routinely be considered in febrile patients; both diseases have been reported and acquired on the mainland and your patients may travel to an endemic area and return home with an unwanted souvenir. You will be ready for them.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Suggested Reading
Chikungunya. Centers for Disease Control and Prevention. 2024. https://www.cdc.gov/vaccines/acip/recommendations.html.
Fagrem AC et al. West Nile and Other Nationally Notifiable Arboviral Diseases–United States, 2021. MMWR Morb Mortal Wkly Rep. 2023 Aug 25;72(34):901-906.
Fever in Returned Travelers, Travel Medicine (Fourth Edition). 2019. doi: 10.1016/B978-0-323-54696-6.00056-2.
Paz-Baily et al. Dengue Vaccine: Recommendations of the Advisory Committee on Immunization Practices, United States, 2021 MMWR Recomm Rep. 2021 Dec 17;70(6):1-16).
Staples JE and Fischer M. Chikungunya virus in the Americas — what a vectorborne pathogen can do. N Engl J Med. 2014 Sep 4;371(10):887-9.
Mosquitoes and Diseases A-Z, Centers for Disease Control and Prevention. https://www.cdc.gov/mosquitoes/about/diseases.html.
There are over 3700 types of mosquitoes worldwide and over 200 types in the continental United States, of which only 12 are associated with transmitting diseases to humans. The majority are just a nuisance. Since they cannot readily be distinguished, strategies to prevent any bites are recommended.
West Nile Virus
In the US, West Nile virus (WNV) is the leading cause of neuroinvasive arboviral disease. Just hearing the name took me back to New York in 1999 when sightings of dead birds around the city and boroughs were reported daily. The virus was isolated that same year. The enzootic circle occurs between mosquitoes and birds, which are the primary vertebrate host via the bite of Culex mosquitoes. After a bite from an infected mosquito, humans are usually a dead-end host since the level and duration of viremia needed to infect another mosquito is insufficient.
Human-to-human transmission is documented through blood transfusion and solid organ transplantation. Vertical transmission is rarely described. Initially isolated in New York, WNV quickly spread across North America and has been isolated in every continent except Antarctica. Most cases occur in the summer and autumn.
Most infected individuals are asymptomatic. Those who do develop symptoms have fever, headache, myalgia, arthralgia, nausea, vomiting, and a transient rash. Less than 1% develop meningitis/encephalitis symptoms similar to other causes of aseptic meningitis. Those with encephalitis in addition to fever and headache may have altered mental status and focal neurologic deficits including flaccid paralysis or movement disorders.
Detection of anti-WNV IgM antibodies (AB) in serum or CSF is the most common way to make the diagnosis. IgM AB usually is present within 3-8 days after onset of symptoms and persists up to 90 days. Data from ArboNET, the national arboviral surveillance system managed by Centers for Disease Control and Prevention and state health departments, reveal that from 1999 to 2022 there were 56,575 cases of WNV including 28,684 cases of neuroinvasive disease. In 2023 there were 2,406 and 1,599 cases, respectively. Those historic totals for WNV are 10 times greater than the totals for all the other etiologies of neuroinvasive arboviral diseases in the US combined (Jamestown Canyon, LaCrosse, St. Louis, and Eastern Equine encephalitis n = 1813).
Remember to include WNV in your differential of a febrile patient with neurologic symptoms, mosquito bites, blood transfusions, and organ transplantation. Treatment is supportive care.
The US began screening all blood donations for WNV in 2003. Organ donor screening is not universal.
Dengue
Dengue, another arbovirus, is transmitted by bites of infected Aedes aegypti and Aedes albopictus mosquitoes, which prefer to feed during the daytime. There are four dengue virus serotypes: DENV-1 DENV-2, DENV-3 and DENV-4. In endemic areas, all four serotypes are usually co-circulating and people can be infected by each one.
Long-term immunity is type specific. Heterologous protection lasts only a few months. Dengue is endemic throughout the tropics and subtropics of Asia, Africa, and the Americas. Approximately 53% of the world’s population live in an area where dengue transmission can occur. In the US, most cases are reported from Puerto Rico. Dengue is endemic in the following US territories: Puerto Rico, US Virgin Islands, American Samoa, and free associated states. Most cases reported on the mainland are travel related. However, locally acquired dengue has been reported. From 2010 to 2023 Hawaii reported 250 cases, Florida 438, and Texas 40 locally acquired cases. During that same period, Puerto Rico reported more than 32,000 cases. It is the leading cause of febrile illness for travelers returning from the Caribbean, Latin America, and South Asia. Peru is currently experiencing an outbreak with more than 25,000 cases reported since January 2024. Most cases of dengue occur in adolescents and young adults. Severe disease occurs most often in infants, those with underlying chronic disease, pregnant women, and persons infected with dengue for the second time.
Symptoms range from a mild febrile illness to severe disease associated with hemorrhage and shock. Onset is usually 7-10 days after infection and symptoms include high fever, severe headache, retro-orbital pain, arthralgia and myalgias, nausea, and vomiting; some may develop a generalized rash.
The World Health Organization (WHO) classifies dengue as 1) dengue with or without warning signs for progression of disease and 2) severe dengue. Warning signs for disease progression include abdominal pain or tenderness, persistent vomiting, fluid accumulation (e.g., ascites, pericardial or pleural effusion), mucosal bleeding, restlessness, postural hypotension, liver enlargement greater than 2 cm. Severe dengue is defined as any sign of severe plasma leakage leading to shock, severe bleeding or organ failure, or fluid accumulation with respiratory distress. Management is supportive care.
Prevention: In the US, Dengvaxia, a live attenuated tetravalent vaccine, is approved for use in children aged 9–16 years with laboratory-confirmed previous dengue virus infection and living in areas where dengue is endemic. It is administered at 0, 6, and 12 months. It is not available for purchase on the mainland. Continued control of the vector and personal protection is necessary to prevent recurrent infections.
CHIKV
Chikungunya (CHIKV), which means “that which bends up” in the Mkonde language of Tanzania, refers to the appearance of the person with severe usually symmetric arthralgias characteristic for this infection that otherwise is often clinically confused with dengue and Zika. It too is transmitted by A. aegypti and A. albopictus and is prevalent in tropical Africa, Asia, Central and South America, and the Caribbean. Like dengue it is predominantly an urban disease. The WHO reported the first case in the Western Hemisphere in Saint Martin in December 2013. By August 2014, 31 additional territories and Caribbean or South American countries reported 576,535 suspected cases. Florida first reported locally acquired CHIKV in June 2014. By December an additional 11 cases had been identified. Texas reported one case in 2015. Diagnosis is with IgM ab or PCR. Treatment is supportive with most recovering from acute illness within 2 weeks. Data in adults indicate 40-52% may develop chronic or recurrent joint pain.
Prevention: IXCHIQ, a live attenuated vaccine, was licensed in November 2023 and recommended by the CDC in February 2024 for use in persons at least 18 years of age with travel to destinations where there is a CHIKV outbreak. It may be considered for persons traveling to a country or territory without an outbreak but with evidence of CHIKV transmission among humans within the last 5 years and those staying in endemic areas for a cumulative period of at least 6 months over a 2-year period. Specific recommendations for lab workers and persons older than 65 years were also made. This is good news for your older patients who may be participating in mission trips, volunteering, studying abroad, or just vacationing in an endemic area. Adolescent vaccine trials are ongoing and pediatric trials will soon be initiated. In addition, vector control and use of personal protective measures cannot be emphasized enough.
There are several other mosquito borne diseases, however our discussion here is limited to three. Why these three? WNV as a reminder that it is the most common neuroinvasive agent in the US. Dengue and CHIKV because they are not endemic in the US so they might not routinely be considered in febrile patients; both diseases have been reported and acquired on the mainland and your patients may travel to an endemic area and return home with an unwanted souvenir. You will be ready for them.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Suggested Reading
Chikungunya. Centers for Disease Control and Prevention. 2024. https://www.cdc.gov/vaccines/acip/recommendations.html.
Fagrem AC et al. West Nile and Other Nationally Notifiable Arboviral Diseases–United States, 2021. MMWR Morb Mortal Wkly Rep. 2023 Aug 25;72(34):901-906.
Fever in Returned Travelers, Travel Medicine (Fourth Edition). 2019. doi: 10.1016/B978-0-323-54696-6.00056-2.
Paz-Baily et al. Dengue Vaccine: Recommendations of the Advisory Committee on Immunization Practices, United States, 2021 MMWR Recomm Rep. 2021 Dec 17;70(6):1-16).
Staples JE and Fischer M. Chikungunya virus in the Americas — what a vectorborne pathogen can do. N Engl J Med. 2014 Sep 4;371(10):887-9.
Mosquitoes and Diseases A-Z, Centers for Disease Control and Prevention. https://www.cdc.gov/mosquitoes/about/diseases.html.
Preparing for the viral trifecta: RSV, influenza, and COVID-19
New armamentaria available to fight an old disease.
In July 2023, nirsevimab (Beyfortus), a monoclonal antibody, was approved by the Food and Drug Administration for the prevention of respiratory syncytial virus (RSV) disease in infants and children younger than 2 years of age. On Aug. 3, 2023, the Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention recommended routine use of it for all infants younger than 8 months of age born during or entering their first RSV season. Its use is also recommended for certain children 8-19 months of age who are at increased risk for severe RSV disease at the start of their second RSV season. Hearing the approval, I immediately had a flashback to residency, recalling the multiple infants admitted each fall and winter exhibiting classic symptoms including cough, rhinorrhea, nasal flaring, retractions, and wheezing with many having oxygen requirements and others needing intubation. Only supportive care was available.
RSV is the leading cause of infant hospitalizations. Annually, the CDC estimates there are 50,000-80,000 RSV hospitalizations and 100-300 RSV-related deaths in the United States in persons younger than 5 years of age. While premature infants have the highest rates of hospitalization (three times a term infant) about 79% of hospitalized children younger than 2 years have no underlying medical risks.1 The majority of children will experience RSV as an upper respiratory infection within the first 2 years of life. However, severe disease requiring hospitalization is more likely to occur in premature infants and children younger than 6 months; children younger than 2 with congenital heart disease and/or chronic lung disease; children with severe cystic fibrosis; as well as the immunocompromised child and individuals with neuromuscular disorders that preclude clearing mucous secretions or have difficulty swallowing.
Palivizumab (Synagis), the first monoclonal antibody to prevent RSV in infants was licensed in 1998. Its use was limited to infants meeting specific criteria developed by the American Academy of Pediatrics. Only 5% of infants had access to it. It was a short-acting agent requiring monthly injections, which were very costly ($1,661-$2,584 per dose). Eligible infants could receive up to five injections per season. Several studies proved its use was not cost beneficial.
What are the advantages of nirsevimab? It’s a long-acting monoclonal antibody. Only one dose is required per season. Costs will significantly diminish. It is recommended for all infants younger than 8 months of age born during RSV season. Those children 8-19 months at risk for severe RSV disease can receive it prior to the start of their second RSV season. During RSV season (October 1 to March 31), the initial dose should be administered to newborns just prior to hospital discharge. Older infants and newborns who did not receive it prior to hospital discharge can receive it at their medical home. Newborns should receive it within the first week of life. It is covered by the Vaccine for Children Program. Simultaneous administration with routine childhood immunizations is recommended. Finally, RSV season may vary in tropical areas (Southern Florida, Puerto Rico. etc.) and Alaska. The timing of nirsevimab administration should be based on local RSV activity provided by state and local authorities.
In addition, the FDA approved an RSV vaccine (Abrysvo) for use in adults at least 60 years of age and in pregnant women at 32-36 weeks’ gestation. The latter is administered to prevent lower respiratory tract infection in infants from birth to 6 months. Recommendations have been published for administration in nonpregnant adults. Specific information is forthcoming in terms timing of administration of nirsevimab in infants whose mothers receive Abrysvo.
RSV season is quickly approaching. Detailed recommendations for administration and FAQ questions related to nirsevimab and palivizumab can be found at https://www.aap.org or https://www.cdc.gov/vaccines/hcp/acip-recs/index.html.
Influenza
So, what about influenza? Vaccine composition has been tweaked to match the circulating viruses but the recommended age for annual routine administration remains unchanged. All persons at least 6 months of age should be vaccinated. Children between 6 months and 8 years need two doses at least 4 weeks apart when receiving vaccine for the first time. Immunizing everyone in the household is encouraged especially if there are household contacts at risk for developing severe disease, including infants too young to be vaccinated. Keep in mind children may be coinfected with multiple viruses. Adams and colleagues reviewed the prevalence of coinfection of influenza and Sars-CoV-2 in persons younger than 18 years reported to three CDC surveillance platforms during the 2021-2022 season.2 Thirty-two of 575 hospitalized (6%) coinfections were analyzed and 7 of 44 (16%) deaths. Compared with patients without coinfections, the coinfected patients were more likely to require mechanical ventilation (13% vs. 4%) or CPAP (16% vs. 6%). Only 4 of 23 who were influenza vaccine eligible were vaccinated. Of seven coinfected children who died, none had received influenza vaccine and only one received an antiviral. Only 5 of 31 (16%) infected only with influenza were vaccinated.3
Influenza activity was lower than usual during the 2021-2022 season. However, this report revealed underuse of both influenza vaccine and antiviral therapy, both of which are routinely recommended.
COVID-19
What’s new with COVID-19? On Sept. 12, 2023, ACIP recommended that everyone at least 6 months of age receive the 2023-2024 (monovalent, XBB containing) COVID-19 vaccines. Children at least 5 years of age need one dose and those younger need one or two doses depending on the number of doses previously received. Why the change? Circulating variants continue to change. There is a current uptick in cases including hospitalizations (7.7%) and deaths (4.5%) and it’s just the beginning of the season.4 Symptoms, risk groups and complications have not changed. The primary goal is to prevent infection, hospitalization, long term complications, and death.
We are now armed with the most up-to-date interventions to help prevent the acquisition of these three viruses. Our next step is recommending and delivering them to our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She reported no relevant financial disclosures.
References
1.Suh M et al. J Infect Dis. 2022;226(Suppl 2):S154-36. doi: 10.1093/infdis/jiac120.
2. Adams K et al. MMWR Morb Mortal Wkly Rep. 2022;71:1589-96. doi: http://dx.doi.org/10.15585/mmwr.mm7150a4.
3. Pingali C et al. MMWR Morb Mortal Wkly Rep. 2023 Aug 25;72:912-9. doi: http://dx.doi.org/10.15585/mmwr.mm7234a3.
4. CDC Covid Data Tracker.
New armamentaria available to fight an old disease.
New armamentaria available to fight an old disease.
In July 2023, nirsevimab (Beyfortus), a monoclonal antibody, was approved by the Food and Drug Administration for the prevention of respiratory syncytial virus (RSV) disease in infants and children younger than 2 years of age. On Aug. 3, 2023, the Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention recommended routine use of it for all infants younger than 8 months of age born during or entering their first RSV season. Its use is also recommended for certain children 8-19 months of age who are at increased risk for severe RSV disease at the start of their second RSV season. Hearing the approval, I immediately had a flashback to residency, recalling the multiple infants admitted each fall and winter exhibiting classic symptoms including cough, rhinorrhea, nasal flaring, retractions, and wheezing with many having oxygen requirements and others needing intubation. Only supportive care was available.
RSV is the leading cause of infant hospitalizations. Annually, the CDC estimates there are 50,000-80,000 RSV hospitalizations and 100-300 RSV-related deaths in the United States in persons younger than 5 years of age. While premature infants have the highest rates of hospitalization (three times a term infant) about 79% of hospitalized children younger than 2 years have no underlying medical risks.1 The majority of children will experience RSV as an upper respiratory infection within the first 2 years of life. However, severe disease requiring hospitalization is more likely to occur in premature infants and children younger than 6 months; children younger than 2 with congenital heart disease and/or chronic lung disease; children with severe cystic fibrosis; as well as the immunocompromised child and individuals with neuromuscular disorders that preclude clearing mucous secretions or have difficulty swallowing.
Palivizumab (Synagis), the first monoclonal antibody to prevent RSV in infants was licensed in 1998. Its use was limited to infants meeting specific criteria developed by the American Academy of Pediatrics. Only 5% of infants had access to it. It was a short-acting agent requiring monthly injections, which were very costly ($1,661-$2,584 per dose). Eligible infants could receive up to five injections per season. Several studies proved its use was not cost beneficial.
What are the advantages of nirsevimab? It’s a long-acting monoclonal antibody. Only one dose is required per season. Costs will significantly diminish. It is recommended for all infants younger than 8 months of age born during RSV season. Those children 8-19 months at risk for severe RSV disease can receive it prior to the start of their second RSV season. During RSV season (October 1 to March 31), the initial dose should be administered to newborns just prior to hospital discharge. Older infants and newborns who did not receive it prior to hospital discharge can receive it at their medical home. Newborns should receive it within the first week of life. It is covered by the Vaccine for Children Program. Simultaneous administration with routine childhood immunizations is recommended. Finally, RSV season may vary in tropical areas (Southern Florida, Puerto Rico. etc.) and Alaska. The timing of nirsevimab administration should be based on local RSV activity provided by state and local authorities.
In addition, the FDA approved an RSV vaccine (Abrysvo) for use in adults at least 60 years of age and in pregnant women at 32-36 weeks’ gestation. The latter is administered to prevent lower respiratory tract infection in infants from birth to 6 months. Recommendations have been published for administration in nonpregnant adults. Specific information is forthcoming in terms timing of administration of nirsevimab in infants whose mothers receive Abrysvo.
RSV season is quickly approaching. Detailed recommendations for administration and FAQ questions related to nirsevimab and palivizumab can be found at https://www.aap.org or https://www.cdc.gov/vaccines/hcp/acip-recs/index.html.
Influenza
So, what about influenza? Vaccine composition has been tweaked to match the circulating viruses but the recommended age for annual routine administration remains unchanged. All persons at least 6 months of age should be vaccinated. Children between 6 months and 8 years need two doses at least 4 weeks apart when receiving vaccine for the first time. Immunizing everyone in the household is encouraged especially if there are household contacts at risk for developing severe disease, including infants too young to be vaccinated. Keep in mind children may be coinfected with multiple viruses. Adams and colleagues reviewed the prevalence of coinfection of influenza and Sars-CoV-2 in persons younger than 18 years reported to three CDC surveillance platforms during the 2021-2022 season.2 Thirty-two of 575 hospitalized (6%) coinfections were analyzed and 7 of 44 (16%) deaths. Compared with patients without coinfections, the coinfected patients were more likely to require mechanical ventilation (13% vs. 4%) or CPAP (16% vs. 6%). Only 4 of 23 who were influenza vaccine eligible were vaccinated. Of seven coinfected children who died, none had received influenza vaccine and only one received an antiviral. Only 5 of 31 (16%) infected only with influenza were vaccinated.3
Influenza activity was lower than usual during the 2021-2022 season. However, this report revealed underuse of both influenza vaccine and antiviral therapy, both of which are routinely recommended.
COVID-19
What’s new with COVID-19? On Sept. 12, 2023, ACIP recommended that everyone at least 6 months of age receive the 2023-2024 (monovalent, XBB containing) COVID-19 vaccines. Children at least 5 years of age need one dose and those younger need one or two doses depending on the number of doses previously received. Why the change? Circulating variants continue to change. There is a current uptick in cases including hospitalizations (7.7%) and deaths (4.5%) and it’s just the beginning of the season.4 Symptoms, risk groups and complications have not changed. The primary goal is to prevent infection, hospitalization, long term complications, and death.
We are now armed with the most up-to-date interventions to help prevent the acquisition of these three viruses. Our next step is recommending and delivering them to our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She reported no relevant financial disclosures.
References
1.Suh M et al. J Infect Dis. 2022;226(Suppl 2):S154-36. doi: 10.1093/infdis/jiac120.
2. Adams K et al. MMWR Morb Mortal Wkly Rep. 2022;71:1589-96. doi: http://dx.doi.org/10.15585/mmwr.mm7150a4.
3. Pingali C et al. MMWR Morb Mortal Wkly Rep. 2023 Aug 25;72:912-9. doi: http://dx.doi.org/10.15585/mmwr.mm7234a3.
4. CDC Covid Data Tracker.
In July 2023, nirsevimab (Beyfortus), a monoclonal antibody, was approved by the Food and Drug Administration for the prevention of respiratory syncytial virus (RSV) disease in infants and children younger than 2 years of age. On Aug. 3, 2023, the Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention recommended routine use of it for all infants younger than 8 months of age born during or entering their first RSV season. Its use is also recommended for certain children 8-19 months of age who are at increased risk for severe RSV disease at the start of their second RSV season. Hearing the approval, I immediately had a flashback to residency, recalling the multiple infants admitted each fall and winter exhibiting classic symptoms including cough, rhinorrhea, nasal flaring, retractions, and wheezing with many having oxygen requirements and others needing intubation. Only supportive care was available.
RSV is the leading cause of infant hospitalizations. Annually, the CDC estimates there are 50,000-80,000 RSV hospitalizations and 100-300 RSV-related deaths in the United States in persons younger than 5 years of age. While premature infants have the highest rates of hospitalization (three times a term infant) about 79% of hospitalized children younger than 2 years have no underlying medical risks.1 The majority of children will experience RSV as an upper respiratory infection within the first 2 years of life. However, severe disease requiring hospitalization is more likely to occur in premature infants and children younger than 6 months; children younger than 2 with congenital heart disease and/or chronic lung disease; children with severe cystic fibrosis; as well as the immunocompromised child and individuals with neuromuscular disorders that preclude clearing mucous secretions or have difficulty swallowing.
Palivizumab (Synagis), the first monoclonal antibody to prevent RSV in infants was licensed in 1998. Its use was limited to infants meeting specific criteria developed by the American Academy of Pediatrics. Only 5% of infants had access to it. It was a short-acting agent requiring monthly injections, which were very costly ($1,661-$2,584 per dose). Eligible infants could receive up to five injections per season. Several studies proved its use was not cost beneficial.
What are the advantages of nirsevimab? It’s a long-acting monoclonal antibody. Only one dose is required per season. Costs will significantly diminish. It is recommended for all infants younger than 8 months of age born during RSV season. Those children 8-19 months at risk for severe RSV disease can receive it prior to the start of their second RSV season. During RSV season (October 1 to March 31), the initial dose should be administered to newborns just prior to hospital discharge. Older infants and newborns who did not receive it prior to hospital discharge can receive it at their medical home. Newborns should receive it within the first week of life. It is covered by the Vaccine for Children Program. Simultaneous administration with routine childhood immunizations is recommended. Finally, RSV season may vary in tropical areas (Southern Florida, Puerto Rico. etc.) and Alaska. The timing of nirsevimab administration should be based on local RSV activity provided by state and local authorities.
In addition, the FDA approved an RSV vaccine (Abrysvo) for use in adults at least 60 years of age and in pregnant women at 32-36 weeks’ gestation. The latter is administered to prevent lower respiratory tract infection in infants from birth to 6 months. Recommendations have been published for administration in nonpregnant adults. Specific information is forthcoming in terms timing of administration of nirsevimab in infants whose mothers receive Abrysvo.
RSV season is quickly approaching. Detailed recommendations for administration and FAQ questions related to nirsevimab and palivizumab can be found at https://www.aap.org or https://www.cdc.gov/vaccines/hcp/acip-recs/index.html.
Influenza
So, what about influenza? Vaccine composition has been tweaked to match the circulating viruses but the recommended age for annual routine administration remains unchanged. All persons at least 6 months of age should be vaccinated. Children between 6 months and 8 years need two doses at least 4 weeks apart when receiving vaccine for the first time. Immunizing everyone in the household is encouraged especially if there are household contacts at risk for developing severe disease, including infants too young to be vaccinated. Keep in mind children may be coinfected with multiple viruses. Adams and colleagues reviewed the prevalence of coinfection of influenza and Sars-CoV-2 in persons younger than 18 years reported to three CDC surveillance platforms during the 2021-2022 season.2 Thirty-two of 575 hospitalized (6%) coinfections were analyzed and 7 of 44 (16%) deaths. Compared with patients without coinfections, the coinfected patients were more likely to require mechanical ventilation (13% vs. 4%) or CPAP (16% vs. 6%). Only 4 of 23 who were influenza vaccine eligible were vaccinated. Of seven coinfected children who died, none had received influenza vaccine and only one received an antiviral. Only 5 of 31 (16%) infected only with influenza were vaccinated.3
Influenza activity was lower than usual during the 2021-2022 season. However, this report revealed underuse of both influenza vaccine and antiviral therapy, both of which are routinely recommended.
COVID-19
What’s new with COVID-19? On Sept. 12, 2023, ACIP recommended that everyone at least 6 months of age receive the 2023-2024 (monovalent, XBB containing) COVID-19 vaccines. Children at least 5 years of age need one dose and those younger need one or two doses depending on the number of doses previously received. Why the change? Circulating variants continue to change. There is a current uptick in cases including hospitalizations (7.7%) and deaths (4.5%) and it’s just the beginning of the season.4 Symptoms, risk groups and complications have not changed. The primary goal is to prevent infection, hospitalization, long term complications, and death.
We are now armed with the most up-to-date interventions to help prevent the acquisition of these three viruses. Our next step is recommending and delivering them to our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She reported no relevant financial disclosures.
References
1.Suh M et al. J Infect Dis. 2022;226(Suppl 2):S154-36. doi: 10.1093/infdis/jiac120.
2. Adams K et al. MMWR Morb Mortal Wkly Rep. 2022;71:1589-96. doi: http://dx.doi.org/10.15585/mmwr.mm7150a4.
3. Pingali C et al. MMWR Morb Mortal Wkly Rep. 2023 Aug 25;72:912-9. doi: http://dx.doi.org/10.15585/mmwr.mm7234a3.
4. CDC Covid Data Tracker.
Rabies: How to respond to parents’ questions
When most families hear the word rabies, they envision a dog foaming at the mouth and think about receiving multiple painful, often intra-abdominal injections. However, the epidemiology of rabies has changed in the United States. Postexposure prophylaxis (PEP) may not always be indicated and for certain persons preexposure prophylaxis (PrEP) is available and recommended.
Rabies is a Lyssavirus that is transmitted through saliva most often from the bite or scratch of an infected animal. Sometimes it’s via direct contact with mucous membranes. Although rare, cases have been described in which an undiagnosed donor passed the virus via transplant to recipients and four cases of aerosolized transmission were documented in two spelunkers and two laboratory technicians working with the virus. Worldwide it’s estimated that rabies causes 59,000 deaths annually.
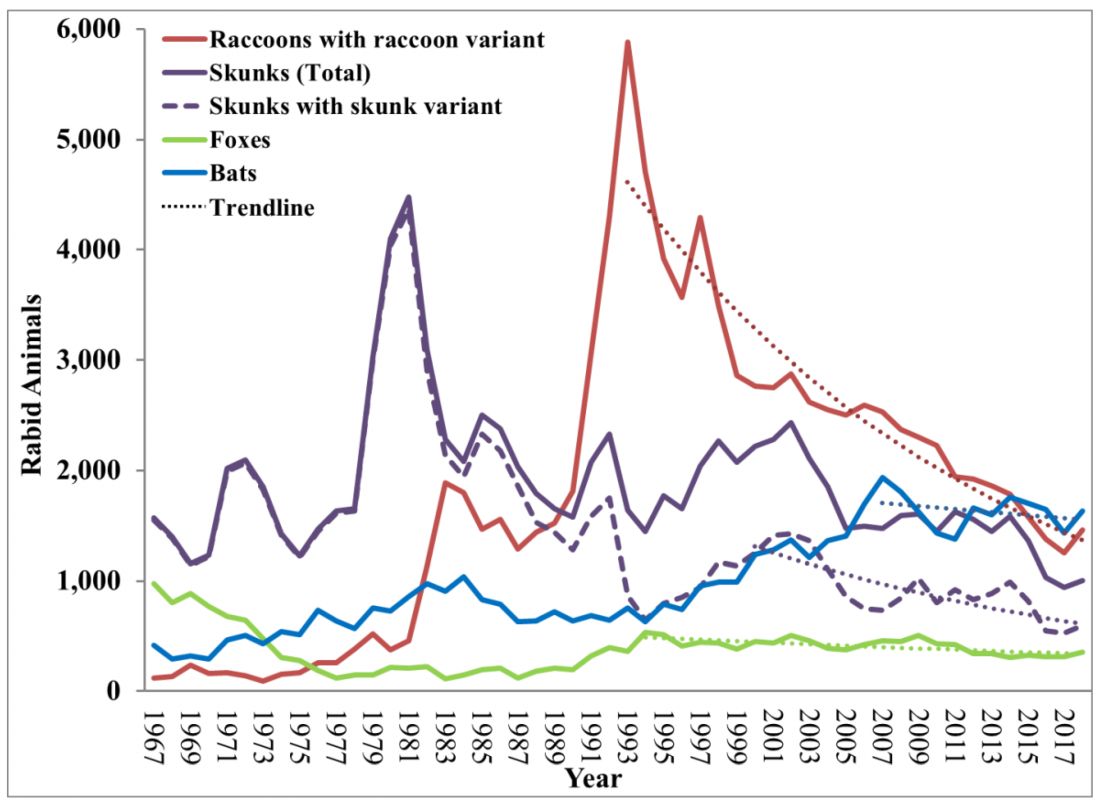
Most cases (98%) are secondary to canine rabies. Prior to 1960, dogs were the major reservoir in the United States; however, after introduction of leash laws and animal vaccination in 1947, there was a drastic decline in cases caused by the canine rabies virus variant (CRVV). By 2004, CRVV was eliminated in the United States.
However, the proportion of strains associated with wildlife including raccoons, skunks, foxes, bats, coyotes, and mongoose now account for most of the cases in humans. Wildlife rabies is found in all states except Hawaii. Between 1960 and 2018, 89 cases were acquired in the United States and 62 (70%) were from bat exposure. Dog bites acquired during international travel were the cause of 36 cases.
Once signs and symptoms of disease develop there is no treatment. Regardless of the species variant, rabies virus infection is fatal in over 99% of cases. However, disease can be prevented with prompt initiation of PEP, which includes administration of rabies immune globulin (RIG) and rabies vaccine. Let’s look at a few different scenarios.
1. A delivery person is bitten by your neighbor’s dog while making a delivery. He was told to get rabies vaccine. What should we advise?
Canine rabies has been eliminated in the United States. However, unvaccinated canines can acquire rabies from wildlife. In this situation, you can determine the immunization status of the dog. Contact your local/state health department to assist with enforcement and management. Bites by cats and ferrets should be managed similarly.
Healthy dog:
1. Observe for 10 days.
2. PEP is not indicated unless the animal develops signs/symptoms of rabies. Then euthanize and begin PEP.
Dog appears rabid or suspected to be rabid:
1. Begin PEP.
2. Animal should be euthanized. If immunofluorescent test is negative discontinue PEP.
Dog unavailable:
Contact local/state health department. They are more familiar with rabies surveillance data.
2. Patient relocating to Malaysia for 3-4 years. Rabies PrEP was recommended but the family wants your opinion before receiving the vaccine. What would you advise?
Canine rabies is felt to be the primary cause of rabies outside of the United States. Canines are not routinely vaccinated in many foreign destinations, and the availability of RIG and rabies vaccine is not guaranteed in developing countries. As noted above, dog bites during international travel accounted for 28% of U.S. cases between 1960 and 2018.
In May 2022 recommendations for a modified two-dose PrEP schedule was published that identifies five risk groups and includes specific timing for checking rabies titers. The third rabies dose can now be administered up until year 3 (Morb Mortal Wkly Rep. 2022 May 6;71[18]:619-27). For individuals relocating to countries where CRVV is present, I prefer the traditional three-dose PrEP schedule administered between 21 and 28 days. However, we now have options. If exposure occurs any time after completion of a three-dose PrEP series or within 3 years after completion of a two-dose PrEP series, RIG would not be required. All patients would receive two doses of rabies vaccine (days 0, 3). If exposure occurs after 3 years in a person who received two doses of PrEP who did not have documentation of a protective rabies titer (> 5 IU/mL), treatment will include RIG plus four doses of vaccine (days 0, 3, 7, 14).
For this relocating patient, supporting PrEP would be strongly recommended.
3. A mother tells you she sees bats flying around her home at night and a few have even gotten into the home. This morning she saw one in her child’s room. He was still sleeping. Is there anything she needs to do?
Bats have become the predominant source of rabies in the United States. In addition to the cases noted above, three fatal cases occurred between Sept. 28 and Nov. 10, 2021, after bat exposures in August 2021 (MMWR Morb Mortal Wkly Rep. 2022 Jan 7;71:31-2). All had recognized contact with a bat 3-7 weeks prior to onset of symptoms and died 2-3 weeks after symptom onset. One declined PEP and the other two did not realize the risk for rabies from their exposure or did not notice a scratch or bite. Bites from bats may be small and unnoticed. Exposure to a bat in a closed room while sleeping is considered an exposure. Hawaii is the only state not reporting rabid bats.
PEP is recommended for her child. She should identify potential areas bats may enter the home and seal them in addition to removal of any bat roosts.
4. A parent realizes a house guest has been feeding raccoons in the backyard. What’s your response?
While bat rabies is the predominant variant associated with disease in the United States, as illustrated in Figure 1, other species of wildlife including raccoons are a major source of rabies. The geographic spread of the raccoon variant of rabies has been limited by oral vaccination via bait. In the situation noted here, the raccoons have returned because food was being offered thus increasing the families chance of a potential rabies exposure. Wildlife including skunks, raccoons, coyotes, foxes, and mongooses are always considered rabid until proven negative by laboratory testing.
You recommend to stop feeding wildlife and never to approach them. Have them contact the local rabies control unit and/or state wildlife services to assist with removal of the raccoons. Depending on the locale, pest control may be required at the owners expense. Inform the family to seek PEP if anyone is bitten or scratched by the raccoons.
As per the Centers for Disease Control and Prevention, about 55,000 residents receive PEP annually with health-associated expenditures including diagnostics, prevention, and control estimated between $245 and $510 million annually. Rabies is one of the most fatal diseases that can be prevented by avoiding contact with wild animals, maintenance of high immunization rates in pets, and keeping people informed of potential sources including bats. One can’t determine if an animal has rabies by looking at it. Rabies remains an urgent disease that we have to remember to address with our patients and their families. For additional information go to www.CDC.gov/rabies.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She has no relevant financial disclosures.
When most families hear the word rabies, they envision a dog foaming at the mouth and think about receiving multiple painful, often intra-abdominal injections. However, the epidemiology of rabies has changed in the United States. Postexposure prophylaxis (PEP) may not always be indicated and for certain persons preexposure prophylaxis (PrEP) is available and recommended.
Rabies is a Lyssavirus that is transmitted through saliva most often from the bite or scratch of an infected animal. Sometimes it’s via direct contact with mucous membranes. Although rare, cases have been described in which an undiagnosed donor passed the virus via transplant to recipients and four cases of aerosolized transmission were documented in two spelunkers and two laboratory technicians working with the virus. Worldwide it’s estimated that rabies causes 59,000 deaths annually.

Most cases (98%) are secondary to canine rabies. Prior to 1960, dogs were the major reservoir in the United States; however, after introduction of leash laws and animal vaccination in 1947, there was a drastic decline in cases caused by the canine rabies virus variant (CRVV). By 2004, CRVV was eliminated in the United States.
However, the proportion of strains associated with wildlife including raccoons, skunks, foxes, bats, coyotes, and mongoose now account for most of the cases in humans. Wildlife rabies is found in all states except Hawaii. Between 1960 and 2018, 89 cases were acquired in the United States and 62 (70%) were from bat exposure. Dog bites acquired during international travel were the cause of 36 cases.
Once signs and symptoms of disease develop there is no treatment. Regardless of the species variant, rabies virus infection is fatal in over 99% of cases. However, disease can be prevented with prompt initiation of PEP, which includes administration of rabies immune globulin (RIG) and rabies vaccine. Let’s look at a few different scenarios.
1. A delivery person is bitten by your neighbor’s dog while making a delivery. He was told to get rabies vaccine. What should we advise?
Canine rabies has been eliminated in the United States. However, unvaccinated canines can acquire rabies from wildlife. In this situation, you can determine the immunization status of the dog. Contact your local/state health department to assist with enforcement and management. Bites by cats and ferrets should be managed similarly.
Healthy dog:
1. Observe for 10 days.
2. PEP is not indicated unless the animal develops signs/symptoms of rabies. Then euthanize and begin PEP.
Dog appears rabid or suspected to be rabid:
1. Begin PEP.
2. Animal should be euthanized. If immunofluorescent test is negative discontinue PEP.
Dog unavailable:
Contact local/state health department. They are more familiar with rabies surveillance data.
2. Patient relocating to Malaysia for 3-4 years. Rabies PrEP was recommended but the family wants your opinion before receiving the vaccine. What would you advise?
Canine rabies is felt to be the primary cause of rabies outside of the United States. Canines are not routinely vaccinated in many foreign destinations, and the availability of RIG and rabies vaccine is not guaranteed in developing countries. As noted above, dog bites during international travel accounted for 28% of U.S. cases between 1960 and 2018.
In May 2022 recommendations for a modified two-dose PrEP schedule was published that identifies five risk groups and includes specific timing for checking rabies titers. The third rabies dose can now be administered up until year 3 (Morb Mortal Wkly Rep. 2022 May 6;71[18]:619-27). For individuals relocating to countries where CRVV is present, I prefer the traditional three-dose PrEP schedule administered between 21 and 28 days. However, we now have options. If exposure occurs any time after completion of a three-dose PrEP series or within 3 years after completion of a two-dose PrEP series, RIG would not be required. All patients would receive two doses of rabies vaccine (days 0, 3). If exposure occurs after 3 years in a person who received two doses of PrEP who did not have documentation of a protective rabies titer (> 5 IU/mL), treatment will include RIG plus four doses of vaccine (days 0, 3, 7, 14).
For this relocating patient, supporting PrEP would be strongly recommended.
3. A mother tells you she sees bats flying around her home at night and a few have even gotten into the home. This morning she saw one in her child’s room. He was still sleeping. Is there anything she needs to do?
Bats have become the predominant source of rabies in the United States. In addition to the cases noted above, three fatal cases occurred between Sept. 28 and Nov. 10, 2021, after bat exposures in August 2021 (MMWR Morb Mortal Wkly Rep. 2022 Jan 7;71:31-2). All had recognized contact with a bat 3-7 weeks prior to onset of symptoms and died 2-3 weeks after symptom onset. One declined PEP and the other two did not realize the risk for rabies from their exposure or did not notice a scratch or bite. Bites from bats may be small and unnoticed. Exposure to a bat in a closed room while sleeping is considered an exposure. Hawaii is the only state not reporting rabid bats.
PEP is recommended for her child. She should identify potential areas bats may enter the home and seal them in addition to removal of any bat roosts.
4. A parent realizes a house guest has been feeding raccoons in the backyard. What’s your response?
While bat rabies is the predominant variant associated with disease in the United States, as illustrated in Figure 1, other species of wildlife including raccoons are a major source of rabies. The geographic spread of the raccoon variant of rabies has been limited by oral vaccination via bait. In the situation noted here, the raccoons have returned because food was being offered thus increasing the families chance of a potential rabies exposure. Wildlife including skunks, raccoons, coyotes, foxes, and mongooses are always considered rabid until proven negative by laboratory testing.

You recommend to stop feeding wildlife and never to approach them. Have them contact the local rabies control unit and/or state wildlife services to assist with removal of the raccoons. Depending on the locale, pest control may be required at the owners expense. Inform the family to seek PEP if anyone is bitten or scratched by the raccoons.
As per the Centers for Disease Control and Prevention, about 55,000 residents receive PEP annually with health-associated expenditures including diagnostics, prevention, and control estimated between $245 and $510 million annually. Rabies is one of the most fatal diseases that can be prevented by avoiding contact with wild animals, maintenance of high immunization rates in pets, and keeping people informed of potential sources including bats. One can’t determine if an animal has rabies by looking at it. Rabies remains an urgent disease that we have to remember to address with our patients and their families. For additional information go to www.CDC.gov/rabies.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She has no relevant financial disclosures.
When most families hear the word rabies, they envision a dog foaming at the mouth and think about receiving multiple painful, often intra-abdominal injections. However, the epidemiology of rabies has changed in the United States. Postexposure prophylaxis (PEP) may not always be indicated and for certain persons preexposure prophylaxis (PrEP) is available and recommended.
Rabies is a Lyssavirus that is transmitted through saliva most often from the bite or scratch of an infected animal. Sometimes it’s via direct contact with mucous membranes. Although rare, cases have been described in which an undiagnosed donor passed the virus via transplant to recipients and four cases of aerosolized transmission were documented in two spelunkers and two laboratory technicians working with the virus. Worldwide it’s estimated that rabies causes 59,000 deaths annually.

Most cases (98%) are secondary to canine rabies. Prior to 1960, dogs were the major reservoir in the United States; however, after introduction of leash laws and animal vaccination in 1947, there was a drastic decline in cases caused by the canine rabies virus variant (CRVV). By 2004, CRVV was eliminated in the United States.
However, the proportion of strains associated with wildlife including raccoons, skunks, foxes, bats, coyotes, and mongoose now account for most of the cases in humans. Wildlife rabies is found in all states except Hawaii. Between 1960 and 2018, 89 cases were acquired in the United States and 62 (70%) were from bat exposure. Dog bites acquired during international travel were the cause of 36 cases.
Once signs and symptoms of disease develop there is no treatment. Regardless of the species variant, rabies virus infection is fatal in over 99% of cases. However, disease can be prevented with prompt initiation of PEP, which includes administration of rabies immune globulin (RIG) and rabies vaccine. Let’s look at a few different scenarios.
1. A delivery person is bitten by your neighbor’s dog while making a delivery. He was told to get rabies vaccine. What should we advise?
Canine rabies has been eliminated in the United States. However, unvaccinated canines can acquire rabies from wildlife. In this situation, you can determine the immunization status of the dog. Contact your local/state health department to assist with enforcement and management. Bites by cats and ferrets should be managed similarly.
Healthy dog:
1. Observe for 10 days.
2. PEP is not indicated unless the animal develops signs/symptoms of rabies. Then euthanize and begin PEP.
Dog appears rabid or suspected to be rabid:
1. Begin PEP.
2. Animal should be euthanized. If immunofluorescent test is negative discontinue PEP.
Dog unavailable:
Contact local/state health department. They are more familiar with rabies surveillance data.
2. Patient relocating to Malaysia for 3-4 years. Rabies PrEP was recommended but the family wants your opinion before receiving the vaccine. What would you advise?
Canine rabies is felt to be the primary cause of rabies outside of the United States. Canines are not routinely vaccinated in many foreign destinations, and the availability of RIG and rabies vaccine is not guaranteed in developing countries. As noted above, dog bites during international travel accounted for 28% of U.S. cases between 1960 and 2018.
In May 2022 recommendations for a modified two-dose PrEP schedule was published that identifies five risk groups and includes specific timing for checking rabies titers. The third rabies dose can now be administered up until year 3 (Morb Mortal Wkly Rep. 2022 May 6;71[18]:619-27). For individuals relocating to countries where CRVV is present, I prefer the traditional three-dose PrEP schedule administered between 21 and 28 days. However, we now have options. If exposure occurs any time after completion of a three-dose PrEP series or within 3 years after completion of a two-dose PrEP series, RIG would not be required. All patients would receive two doses of rabies vaccine (days 0, 3). If exposure occurs after 3 years in a person who received two doses of PrEP who did not have documentation of a protective rabies titer (> 5 IU/mL), treatment will include RIG plus four doses of vaccine (days 0, 3, 7, 14).
For this relocating patient, supporting PrEP would be strongly recommended.
3. A mother tells you she sees bats flying around her home at night and a few have even gotten into the home. This morning she saw one in her child’s room. He was still sleeping. Is there anything she needs to do?
Bats have become the predominant source of rabies in the United States. In addition to the cases noted above, three fatal cases occurred between Sept. 28 and Nov. 10, 2021, after bat exposures in August 2021 (MMWR Morb Mortal Wkly Rep. 2022 Jan 7;71:31-2). All had recognized contact with a bat 3-7 weeks prior to onset of symptoms and died 2-3 weeks after symptom onset. One declined PEP and the other two did not realize the risk for rabies from their exposure or did not notice a scratch or bite. Bites from bats may be small and unnoticed. Exposure to a bat in a closed room while sleeping is considered an exposure. Hawaii is the only state not reporting rabid bats.
PEP is recommended for her child. She should identify potential areas bats may enter the home and seal them in addition to removal of any bat roosts.
4. A parent realizes a house guest has been feeding raccoons in the backyard. What’s your response?
While bat rabies is the predominant variant associated with disease in the United States, as illustrated in Figure 1, other species of wildlife including raccoons are a major source of rabies. The geographic spread of the raccoon variant of rabies has been limited by oral vaccination via bait. In the situation noted here, the raccoons have returned because food was being offered thus increasing the families chance of a potential rabies exposure. Wildlife including skunks, raccoons, coyotes, foxes, and mongooses are always considered rabid until proven negative by laboratory testing.

You recommend to stop feeding wildlife and never to approach them. Have them contact the local rabies control unit and/or state wildlife services to assist with removal of the raccoons. Depending on the locale, pest control may be required at the owners expense. Inform the family to seek PEP if anyone is bitten or scratched by the raccoons.
As per the Centers for Disease Control and Prevention, about 55,000 residents receive PEP annually with health-associated expenditures including diagnostics, prevention, and control estimated between $245 and $510 million annually. Rabies is one of the most fatal diseases that can be prevented by avoiding contact with wild animals, maintenance of high immunization rates in pets, and keeping people informed of potential sources including bats. One can’t determine if an animal has rabies by looking at it. Rabies remains an urgent disease that we have to remember to address with our patients and their families. For additional information go to www.CDC.gov/rabies.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She has no relevant financial disclosures.
Congenital syphilis: It’s still a significant public health problem
You’re rounding in the nursery and informed of the following about one of your new patients: He’s a 38-week-old infant delivered to a mother diagnosed with syphilis at 12 weeks’ gestation at her initial prenatal visit. Her rapid plasma reagin (RPR) was 1:64 and the fluorescent treponemal antibody–absorption (FTA-ABS) test was positive. By report she was appropriately treated. Maternal RPRs obtained at 18 and 28 weeks’ gestation were 1:16 and 1:4, respectively. Maternal RPR at delivery and the infant’s RPR obtained shortly after birth were both 1:4. The mother wants to know if her baby is infected.
One result of syphilis during pregnancy is intrauterine infection and resultant congenital disease in the infant. Before you answer this mother, let’s discuss syphilis.
Congenital syphilis is a significant public health problem. In 2021, there were a total of 2,677 cases reported for a rate of 74.1 per 100,000 live births. Between 2020 and 2021, the number of cases of congenital syphilis increased 24.1% (2,158-2,677 cases), concurrent with a 45.8% increase (10.7-15.6 per 100,000) in the rate of primary and secondary syphilis in women aged 15-44 years. Between 2012 and 2021, the number of cases of congenital syphilis increased 701.5% (334-2,677 cases) and the increase in rates of primary and secondary syphilis in women aged 15-44 was 642.9% over the same period.
Why are the rates of congenital syphilis increasing? Most cases result from a lack of prenatal care and thus no testing for syphilis. The next most common cause is inadequate maternal treatment.
Congenital syphilis usually is acquired through transplacental transmission of spirochetes in the maternal bloodstream. Occasionally, it occurs at delivery via direct contact with maternal lesions. It is not transmitted in breast milk. Transmission of syphilis:
- Can occur any time during pregnancy.
- Is more likely to occur in women with untreated primary or secondary disease (60%-100%).
- Is approximately 40% in those with early latent syphilis and less than 8% in mothers with late latent syphilis.
- Is higher in women coinfected with HIV since they more frequently receive no prenatal care and their disease is inadequately treated.
Coinfection with syphilis may also increase the rate of mother-to-child transmission of HIV.
Untreated early syphilis during pregnancy results in spontaneous abortion, stillbirth, or perinatal death in up to 40% of cases. Infected newborns with early congenital syphilis can be asymptomatic or have evidence of hepatosplenomegaly, generalized lymphadenopathy, nasal discharge that is occasionally bloody, rash, and skeletal abnormalities (osteochondritis and periostitis). Other manifestations include edema, hemolytic anemia, jaundice, pneumonia, pseudoparalysis, and thrombocytopenia. Asymptomatic infants may have abnormal cerebrospinal fluid findings including elevated CSF white cell count, elevated protein, and a reactive venereal disease research laboratory test.
Late congenital syphilis, defined as the onset of symptoms after 2 years of age is secondary to scarring or persistent inflammation and gumma formation in a variety of tissues. It occurs in up to 40% of cases of untreated maternal disease. Most cases can be prevented by maternal treatment and treatment of the infant within the first 3 months of life. Common clinical manifestations include interstitial keratitis, sensorineural hearing loss, frontal bossing, saddle nose, Hutchinson teeth, mulberry molars, perforation of the hard palate, anterior bowing of the tibia (saber shins), and other skeletal abnormalities.
Diagnostic tests. Maternal diagnosis is dependent upon knowing the results of both a nontreponemal (RPR, VDRL) and a confirmatory treponemal test (TP-PA, TP-EIA, TP-CIA, FTA-ABS,) before or at delivery. TP-PA is the preferred test. When maternal disease is confirmed, the newborn should have the same quantitative nontreponemal test as the mother. A confirmatory treponemal test is not required
Evaluation and treatment. It’s imperative that children born to mothers with a reactive test, regardless of their treatment status, have a thorough exam performed before hospital discharge. The provider must determine what additional interventions should be performed.
The American Academy of Pediatrics and the Centers for Disease Control and Prevention (www.cdc.gov/std/treatment-guidelines/congenital-syphilis.htm) have developed standard algorithms for the diagnostic approach and treatment of infants born to mothers with reactive serologic tests for syphilis. It is available in the Red Book for AAP members (https://publications.aap.org/redbook). Recommendations based on various scenarios for neonates up to 1 month of age include proven or highly probable congenital syphilis, possible congenital syphilis, congenital syphilis less likely, and congenital syphilis unlikely. It is beyond the scope of this article to list the criteria and evaluation for each scenario. The reader is referred to the algorithm.
If syphilis is suspected in infants or children older than 1 month, the challenge is to determine if it is untreated congenital syphilis or acquired syphilis. Maternal syphilis status should be determined. Evaluation for congenital syphilis in this age group includes CSF analysis for VDRL, cell count and protein, CBC with differential and platelets, hepatic panel, abdominal ultrasound, long-bone radiographs, chest radiograph, neuroimaging, auditory brain stem response, and HIV testing.
Let’s go back to your patient. The mother was diagnosed with syphilis during pregnancy. You confirm that she was treated with benzathine penicillin G, and the course was completed at least 4 weeks before delivery. Treatment with any other drug during pregnancy is not appropriate. The RPR has declined, and the infant’s titer is equal to or less than four times the maternal titer. The exam is significant for generalized adenopathy and slightly bloody nasal discharge. This infant has two findings consistent with congenital syphilis regardless of RPR titer or treatment status. This places him in the proven or highly probable congenital syphilis group. Management includes CSF analysis (VDRL, cell count, and protein), CBC with differential and platelet count, and treatment with penicillin G for 10 days. Additional tests as clinically indicated include: long-bone radiograph, chest radiography, aspartate aminotranferase and alanine aminotransferase levels, neuroimaging, ophthalmologic exam, and auditory brain stem response. Despite maternal treatment, this newborn has congenital syphilis. The same nontreponemal test should be obtained every 2-3 months until it is nonreactive. It should be nonreactive by 6 months. If the infection persists to 6-12 months post treatment, reevaluation including CSF analysis and retreatment may be indicated.
Congenital syphilis can be prevented by maternal screening, diagnosis, and treatment. When that fails it is up to us to diagnosis and adequately treat our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].
You’re rounding in the nursery and informed of the following about one of your new patients: He’s a 38-week-old infant delivered to a mother diagnosed with syphilis at 12 weeks’ gestation at her initial prenatal visit. Her rapid plasma reagin (RPR) was 1:64 and the fluorescent treponemal antibody–absorption (FTA-ABS) test was positive. By report she was appropriately treated. Maternal RPRs obtained at 18 and 28 weeks’ gestation were 1:16 and 1:4, respectively. Maternal RPR at delivery and the infant’s RPR obtained shortly after birth were both 1:4. The mother wants to know if her baby is infected.
One result of syphilis during pregnancy is intrauterine infection and resultant congenital disease in the infant. Before you answer this mother, let’s discuss syphilis.
Congenital syphilis is a significant public health problem. In 2021, there were a total of 2,677 cases reported for a rate of 74.1 per 100,000 live births. Between 2020 and 2021, the number of cases of congenital syphilis increased 24.1% (2,158-2,677 cases), concurrent with a 45.8% increase (10.7-15.6 per 100,000) in the rate of primary and secondary syphilis in women aged 15-44 years. Between 2012 and 2021, the number of cases of congenital syphilis increased 701.5% (334-2,677 cases) and the increase in rates of primary and secondary syphilis in women aged 15-44 was 642.9% over the same period.
Why are the rates of congenital syphilis increasing? Most cases result from a lack of prenatal care and thus no testing for syphilis. The next most common cause is inadequate maternal treatment.
Congenital syphilis usually is acquired through transplacental transmission of spirochetes in the maternal bloodstream. Occasionally, it occurs at delivery via direct contact with maternal lesions. It is not transmitted in breast milk. Transmission of syphilis:
- Can occur any time during pregnancy.
- Is more likely to occur in women with untreated primary or secondary disease (60%-100%).
- Is approximately 40% in those with early latent syphilis and less than 8% in mothers with late latent syphilis.
- Is higher in women coinfected with HIV since they more frequently receive no prenatal care and their disease is inadequately treated.
Coinfection with syphilis may also increase the rate of mother-to-child transmission of HIV.
Untreated early syphilis during pregnancy results in spontaneous abortion, stillbirth, or perinatal death in up to 40% of cases. Infected newborns with early congenital syphilis can be asymptomatic or have evidence of hepatosplenomegaly, generalized lymphadenopathy, nasal discharge that is occasionally bloody, rash, and skeletal abnormalities (osteochondritis and periostitis). Other manifestations include edema, hemolytic anemia, jaundice, pneumonia, pseudoparalysis, and thrombocytopenia. Asymptomatic infants may have abnormal cerebrospinal fluid findings including elevated CSF white cell count, elevated protein, and a reactive venereal disease research laboratory test.
Late congenital syphilis, defined as the onset of symptoms after 2 years of age is secondary to scarring or persistent inflammation and gumma formation in a variety of tissues. It occurs in up to 40% of cases of untreated maternal disease. Most cases can be prevented by maternal treatment and treatment of the infant within the first 3 months of life. Common clinical manifestations include interstitial keratitis, sensorineural hearing loss, frontal bossing, saddle nose, Hutchinson teeth, mulberry molars, perforation of the hard palate, anterior bowing of the tibia (saber shins), and other skeletal abnormalities.
Diagnostic tests. Maternal diagnosis is dependent upon knowing the results of both a nontreponemal (RPR, VDRL) and a confirmatory treponemal test (TP-PA, TP-EIA, TP-CIA, FTA-ABS,) before or at delivery. TP-PA is the preferred test. When maternal disease is confirmed, the newborn should have the same quantitative nontreponemal test as the mother. A confirmatory treponemal test is not required
Evaluation and treatment. It’s imperative that children born to mothers with a reactive test, regardless of their treatment status, have a thorough exam performed before hospital discharge. The provider must determine what additional interventions should be performed.
The American Academy of Pediatrics and the Centers for Disease Control and Prevention (www.cdc.gov/std/treatment-guidelines/congenital-syphilis.htm) have developed standard algorithms for the diagnostic approach and treatment of infants born to mothers with reactive serologic tests for syphilis. It is available in the Red Book for AAP members (https://publications.aap.org/redbook). Recommendations based on various scenarios for neonates up to 1 month of age include proven or highly probable congenital syphilis, possible congenital syphilis, congenital syphilis less likely, and congenital syphilis unlikely. It is beyond the scope of this article to list the criteria and evaluation for each scenario. The reader is referred to the algorithm.
If syphilis is suspected in infants or children older than 1 month, the challenge is to determine if it is untreated congenital syphilis or acquired syphilis. Maternal syphilis status should be determined. Evaluation for congenital syphilis in this age group includes CSF analysis for VDRL, cell count and protein, CBC with differential and platelets, hepatic panel, abdominal ultrasound, long-bone radiographs, chest radiograph, neuroimaging, auditory brain stem response, and HIV testing.
Let’s go back to your patient. The mother was diagnosed with syphilis during pregnancy. You confirm that she was treated with benzathine penicillin G, and the course was completed at least 4 weeks before delivery. Treatment with any other drug during pregnancy is not appropriate. The RPR has declined, and the infant’s titer is equal to or less than four times the maternal titer. The exam is significant for generalized adenopathy and slightly bloody nasal discharge. This infant has two findings consistent with congenital syphilis regardless of RPR titer or treatment status. This places him in the proven or highly probable congenital syphilis group. Management includes CSF analysis (VDRL, cell count, and protein), CBC with differential and platelet count, and treatment with penicillin G for 10 days. Additional tests as clinically indicated include: long-bone radiograph, chest radiography, aspartate aminotranferase and alanine aminotransferase levels, neuroimaging, ophthalmologic exam, and auditory brain stem response. Despite maternal treatment, this newborn has congenital syphilis. The same nontreponemal test should be obtained every 2-3 months until it is nonreactive. It should be nonreactive by 6 months. If the infection persists to 6-12 months post treatment, reevaluation including CSF analysis and retreatment may be indicated.
Congenital syphilis can be prevented by maternal screening, diagnosis, and treatment. When that fails it is up to us to diagnosis and adequately treat our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].
You’re rounding in the nursery and informed of the following about one of your new patients: He’s a 38-week-old infant delivered to a mother diagnosed with syphilis at 12 weeks’ gestation at her initial prenatal visit. Her rapid plasma reagin (RPR) was 1:64 and the fluorescent treponemal antibody–absorption (FTA-ABS) test was positive. By report she was appropriately treated. Maternal RPRs obtained at 18 and 28 weeks’ gestation were 1:16 and 1:4, respectively. Maternal RPR at delivery and the infant’s RPR obtained shortly after birth were both 1:4. The mother wants to know if her baby is infected.
One result of syphilis during pregnancy is intrauterine infection and resultant congenital disease in the infant. Before you answer this mother, let’s discuss syphilis.
Congenital syphilis is a significant public health problem. In 2021, there were a total of 2,677 cases reported for a rate of 74.1 per 100,000 live births. Between 2020 and 2021, the number of cases of congenital syphilis increased 24.1% (2,158-2,677 cases), concurrent with a 45.8% increase (10.7-15.6 per 100,000) in the rate of primary and secondary syphilis in women aged 15-44 years. Between 2012 and 2021, the number of cases of congenital syphilis increased 701.5% (334-2,677 cases) and the increase in rates of primary and secondary syphilis in women aged 15-44 was 642.9% over the same period.
Why are the rates of congenital syphilis increasing? Most cases result from a lack of prenatal care and thus no testing for syphilis. The next most common cause is inadequate maternal treatment.
Congenital syphilis usually is acquired through transplacental transmission of spirochetes in the maternal bloodstream. Occasionally, it occurs at delivery via direct contact with maternal lesions. It is not transmitted in breast milk. Transmission of syphilis:
- Can occur any time during pregnancy.
- Is more likely to occur in women with untreated primary or secondary disease (60%-100%).
- Is approximately 40% in those with early latent syphilis and less than 8% in mothers with late latent syphilis.
- Is higher in women coinfected with HIV since they more frequently receive no prenatal care and their disease is inadequately treated.
Coinfection with syphilis may also increase the rate of mother-to-child transmission of HIV.
Untreated early syphilis during pregnancy results in spontaneous abortion, stillbirth, or perinatal death in up to 40% of cases. Infected newborns with early congenital syphilis can be asymptomatic or have evidence of hepatosplenomegaly, generalized lymphadenopathy, nasal discharge that is occasionally bloody, rash, and skeletal abnormalities (osteochondritis and periostitis). Other manifestations include edema, hemolytic anemia, jaundice, pneumonia, pseudoparalysis, and thrombocytopenia. Asymptomatic infants may have abnormal cerebrospinal fluid findings including elevated CSF white cell count, elevated protein, and a reactive venereal disease research laboratory test.
Late congenital syphilis, defined as the onset of symptoms after 2 years of age is secondary to scarring or persistent inflammation and gumma formation in a variety of tissues. It occurs in up to 40% of cases of untreated maternal disease. Most cases can be prevented by maternal treatment and treatment of the infant within the first 3 months of life. Common clinical manifestations include interstitial keratitis, sensorineural hearing loss, frontal bossing, saddle nose, Hutchinson teeth, mulberry molars, perforation of the hard palate, anterior bowing of the tibia (saber shins), and other skeletal abnormalities.
Diagnostic tests. Maternal diagnosis is dependent upon knowing the results of both a nontreponemal (RPR, VDRL) and a confirmatory treponemal test (TP-PA, TP-EIA, TP-CIA, FTA-ABS,) before or at delivery. TP-PA is the preferred test. When maternal disease is confirmed, the newborn should have the same quantitative nontreponemal test as the mother. A confirmatory treponemal test is not required
Evaluation and treatment. It’s imperative that children born to mothers with a reactive test, regardless of their treatment status, have a thorough exam performed before hospital discharge. The provider must determine what additional interventions should be performed.
The American Academy of Pediatrics and the Centers for Disease Control and Prevention (www.cdc.gov/std/treatment-guidelines/congenital-syphilis.htm) have developed standard algorithms for the diagnostic approach and treatment of infants born to mothers with reactive serologic tests for syphilis. It is available in the Red Book for AAP members (https://publications.aap.org/redbook). Recommendations based on various scenarios for neonates up to 1 month of age include proven or highly probable congenital syphilis, possible congenital syphilis, congenital syphilis less likely, and congenital syphilis unlikely. It is beyond the scope of this article to list the criteria and evaluation for each scenario. The reader is referred to the algorithm.
If syphilis is suspected in infants or children older than 1 month, the challenge is to determine if it is untreated congenital syphilis or acquired syphilis. Maternal syphilis status should be determined. Evaluation for congenital syphilis in this age group includes CSF analysis for VDRL, cell count and protein, CBC with differential and platelets, hepatic panel, abdominal ultrasound, long-bone radiographs, chest radiograph, neuroimaging, auditory brain stem response, and HIV testing.
Let’s go back to your patient. The mother was diagnosed with syphilis during pregnancy. You confirm that she was treated with benzathine penicillin G, and the course was completed at least 4 weeks before delivery. Treatment with any other drug during pregnancy is not appropriate. The RPR has declined, and the infant’s titer is equal to or less than four times the maternal titer. The exam is significant for generalized adenopathy and slightly bloody nasal discharge. This infant has two findings consistent with congenital syphilis regardless of RPR titer or treatment status. This places him in the proven or highly probable congenital syphilis group. Management includes CSF analysis (VDRL, cell count, and protein), CBC with differential and platelet count, and treatment with penicillin G for 10 days. Additional tests as clinically indicated include: long-bone radiograph, chest radiography, aspartate aminotranferase and alanine aminotransferase levels, neuroimaging, ophthalmologic exam, and auditory brain stem response. Despite maternal treatment, this newborn has congenital syphilis. The same nontreponemal test should be obtained every 2-3 months until it is nonreactive. It should be nonreactive by 6 months. If the infection persists to 6-12 months post treatment, reevaluation including CSF analysis and retreatment may be indicated.
Congenital syphilis can be prevented by maternal screening, diagnosis, and treatment. When that fails it is up to us to diagnosis and adequately treat our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].
Measles outbreaks: Protecting your patients during international travel
The U.S. immunization program is one of the best public health success stories. Physicians who provide care for children are familiar with the routine childhood immunization schedule and administer a measles-containing vaccine at age-appropriate times. Thanks to its rigorous implementation and acceptance, endemic measles (absence of continuous virus transmission for > 1 year) was eliminated in the U.S. in 2000. Loss of this status was in jeopardy in 2019 when 22 measles outbreaks occurred in 17 states (7 were multistate outbreaks). That year, 1,163 cases were reported.1 Most cases occurred in unvaccinated persons (89%) and 81 cases were imported of which 54 were in U.S. citizens returning from international travel. All outbreaks were linked to travel. Fortunately, the outbreaks were controlled prior to the elimination deadline, or the United States would have lost its measles elimination status. Restrictions on travel because of COVID-19 have relaxed significantly since the introduction of COVID-19 vaccines, resulting in increased regional and international travel. Multiple countries, including the United States noted a decline in routine immunizations rates during the last 2 years. Recent U.S. data for the 2020-2021 school year indicates that MMR immunizations rates (two doses) for kindergarteners declined to 93.9% (range 78.9% to > 98.9%), while the overall percentage of those students with an exemption remained low at 2.2%. Vaccine coverage greater than 95% was reported in only 16 states. Coverage of less than 90% was reported in seven states and the District of Columbia (Georgia, Idaho, Kentucky, Maryland, Minnesota, Ohio, and Wisconsin).2 Vaccine coverage should be 95% or higher to maintain herd immunity and control outbreaks.
Why is measles prevention so important? Many physicians practicing in the United States today have never seen a case or know its potential complications. I saw my first case as a resident in an immigrant child. It took our training director to point out the subtle signs and symptoms. It was the first time I saw Kolpik spots. Measles is transmitted person to person via large respiratory droplets and less often by airborne spread. It is highly contagious for susceptible individuals with an attack rate of 90%. In this case, a medical student on the team developed symptoms about 10 days later. Six years would pass before I diagnosed my next case of measles. An HIV patient acquired it after close contact with someone who was in the prodromal stage. He presented with the 3 C’s: Cough, coryza, and conjunctivitis, in addition to fever and an erythematous rash. He did not recover from complications of the disease.
Prior to the routine administration of a measles vaccine, 3-4 million cases with almost 500 deaths occurred annually in the United States. Worldwide, 35 million cases and more than 6 million deaths occurred each year. Here, most patients recover completely; however, complications including otitis media, pneumonia, croup, and encephalitis can develop. Complications commonly occur in immunocompromised individuals and young children. Groups with the highest fatality rates include children aged less than 5 years, immunocompromised persons, and pregnant women. Worldwide, fatality rates are dependent on the patients underlying nutritional and health status in addition to the quality of health care available.3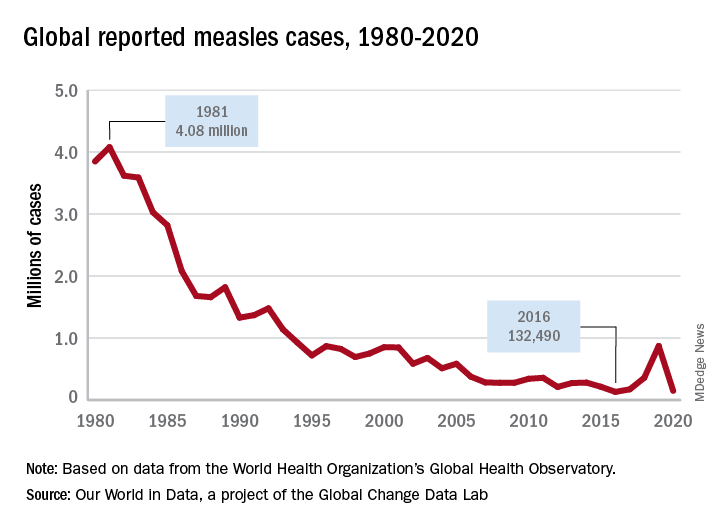
Measles vaccine was licensed in 1963 and cases began to decline (Figure 1). There was a resurgence in 1989 but it was not limited to the United States. The cause of the U.S. resurgence was multifactorial: Widespread viral transmission among unvaccinated preschool-age children residing in inner cities, outbreaks in vaccinated school-age children, outbreaks in students and personnel on college campuses, and primary vaccine failure (2%-5% of recipients failed to have an adequate response). In 1989, to help prevent future outbreaks, the United States recommended a two-dose schedule for measles and in 1993, the Vaccines for Children Program, a federally funded program, was established to improve access to vaccines for all children.
What is going on internationally?
Figure 2 lists the top 10 countries with current measles outbreaks.

Most countries on the list may not be typical travel destinations for tourists; however, they are common destinations for individuals visiting friends and relatives after immigrating to the United States. In contrast to the United States, most countries with limited resources and infrastructure have mass-vaccination campaigns to ensure vaccine administration to large segments of the population. They too have been affected by the COVID-19 pandemic. By report, at least 41 countries delayed implementation of their measles campaign in 2020 and 2021, thus, leading to the potential for even larger outbreaks.4
Progress toward the global elimination of measles is evidenced by the following: All 194 countries now include one dose of measles in their routine schedules; between 2000 and 2019 coverage of one dose of measles increased from 72% to 85% and countries with more than 90% coverage increased from 45% to 63%. Finally, the number of countries offering two doses of measles increased from 50% to 91% and vaccine coverage increased from 18% to 71% over the same time period.3
What can you do for your patients and their parents before they travel abroad?
- Inform all staff that the MMR vaccine can be administered to children as young as 6 months and at times other than those listed on the routine immunization schedule. This will help avoid parents seeking vaccine being denied an appointment.
- Children 6-11 months need 1 dose of MMR. Two additional doses will still need to be administered at the routine time.
- Children 12 months or older need 2 doses of MMR at least 4 weeks apart.
- If yellow fever vaccine is needed, coordinate administration with a travel medicine clinic since both are live vaccines and must be given on the same day.
- Any person born after 1956 should have 2 doses of MMR at least 4 weeks apart if they have no evidence of immunity.
- Encourage parents to always inform you and your staff of any international travel plans.
Moving forward, remember this increased global activity and the presence of inadequately vaccinated individuals/communities keeps the United States at continued risk for measles outbreaks. The source of the next outbreak may only be one plane ride away.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
This article was updated 6/29/22.
References
1. Patel M et al. MMWR. 2019 Oct 11; 68(40):893-6.
2. Seither R et al. MMWR. 2022 Apr 22;71(16):561-8.
3. Gastañaduy PA et al. J Infect Dis. 2021 Sep 30;224(12 Suppl 2):S420-8. doi: 10.1093/infdis/jiaa793.
4. Centers for Disease Control and Prevention. Measles (Rubeola). http://www.CDC.gov/Measles.
The U.S. immunization program is one of the best public health success stories. Physicians who provide care for children are familiar with the routine childhood immunization schedule and administer a measles-containing vaccine at age-appropriate times. Thanks to its rigorous implementation and acceptance, endemic measles (absence of continuous virus transmission for > 1 year) was eliminated in the U.S. in 2000. Loss of this status was in jeopardy in 2019 when 22 measles outbreaks occurred in 17 states (7 were multistate outbreaks). That year, 1,163 cases were reported.1 Most cases occurred in unvaccinated persons (89%) and 81 cases were imported of which 54 were in U.S. citizens returning from international travel. All outbreaks were linked to travel. Fortunately, the outbreaks were controlled prior to the elimination deadline, or the United States would have lost its measles elimination status. Restrictions on travel because of COVID-19 have relaxed significantly since the introduction of COVID-19 vaccines, resulting in increased regional and international travel. Multiple countries, including the United States noted a decline in routine immunizations rates during the last 2 years. Recent U.S. data for the 2020-2021 school year indicates that MMR immunizations rates (two doses) for kindergarteners declined to 93.9% (range 78.9% to > 98.9%), while the overall percentage of those students with an exemption remained low at 2.2%. Vaccine coverage greater than 95% was reported in only 16 states. Coverage of less than 90% was reported in seven states and the District of Columbia (Georgia, Idaho, Kentucky, Maryland, Minnesota, Ohio, and Wisconsin).2 Vaccine coverage should be 95% or higher to maintain herd immunity and control outbreaks.
Why is measles prevention so important? Many physicians practicing in the United States today have never seen a case or know its potential complications. I saw my first case as a resident in an immigrant child. It took our training director to point out the subtle signs and symptoms. It was the first time I saw Kolpik spots. Measles is transmitted person to person via large respiratory droplets and less often by airborne spread. It is highly contagious for susceptible individuals with an attack rate of 90%. In this case, a medical student on the team developed symptoms about 10 days later. Six years would pass before I diagnosed my next case of measles. An HIV patient acquired it after close contact with someone who was in the prodromal stage. He presented with the 3 C’s: Cough, coryza, and conjunctivitis, in addition to fever and an erythematous rash. He did not recover from complications of the disease.
Prior to the routine administration of a measles vaccine, 3-4 million cases with almost 500 deaths occurred annually in the United States. Worldwide, 35 million cases and more than 6 million deaths occurred each year. Here, most patients recover completely; however, complications including otitis media, pneumonia, croup, and encephalitis can develop. Complications commonly occur in immunocompromised individuals and young children. Groups with the highest fatality rates include children aged less than 5 years, immunocompromised persons, and pregnant women. Worldwide, fatality rates are dependent on the patients underlying nutritional and health status in addition to the quality of health care available.3
Measles vaccine was licensed in 1963 and cases began to decline (Figure 1). There was a resurgence in 1989 but it was not limited to the United States. The cause of the U.S. resurgence was multifactorial: Widespread viral transmission among unvaccinated preschool-age children residing in inner cities, outbreaks in vaccinated school-age children, outbreaks in students and personnel on college campuses, and primary vaccine failure (2%-5% of recipients failed to have an adequate response). In 1989, to help prevent future outbreaks, the United States recommended a two-dose schedule for measles and in 1993, the Vaccines for Children Program, a federally funded program, was established to improve access to vaccines for all children.
What is going on internationally?
Figure 2 lists the top 10 countries with current measles outbreaks.

Most countries on the list may not be typical travel destinations for tourists; however, they are common destinations for individuals visiting friends and relatives after immigrating to the United States. In contrast to the United States, most countries with limited resources and infrastructure have mass-vaccination campaigns to ensure vaccine administration to large segments of the population. They too have been affected by the COVID-19 pandemic. By report, at least 41 countries delayed implementation of their measles campaign in 2020 and 2021, thus, leading to the potential for even larger outbreaks.4
Progress toward the global elimination of measles is evidenced by the following: All 194 countries now include one dose of measles in their routine schedules; between 2000 and 2019 coverage of one dose of measles increased from 72% to 85% and countries with more than 90% coverage increased from 45% to 63%. Finally, the number of countries offering two doses of measles increased from 50% to 91% and vaccine coverage increased from 18% to 71% over the same time period.3
What can you do for your patients and their parents before they travel abroad?
- Inform all staff that the MMR vaccine can be administered to children as young as 6 months and at times other than those listed on the routine immunization schedule. This will help avoid parents seeking vaccine being denied an appointment.
- Children 6-11 months need 1 dose of MMR. Two additional doses will still need to be administered at the routine time.
- Children 12 months or older need 2 doses of MMR at least 4 weeks apart.
- If yellow fever vaccine is needed, coordinate administration with a travel medicine clinic since both are live vaccines and must be given on the same day.
- Any person born after 1956 should have 2 doses of MMR at least 4 weeks apart if they have no evidence of immunity.
- Encourage parents to always inform you and your staff of any international travel plans.
Moving forward, remember this increased global activity and the presence of inadequately vaccinated individuals/communities keeps the United States at continued risk for measles outbreaks. The source of the next outbreak may only be one plane ride away.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
This article was updated 6/29/22.
References
1. Patel M et al. MMWR. 2019 Oct 11; 68(40):893-6.
2. Seither R et al. MMWR. 2022 Apr 22;71(16):561-8.
3. Gastañaduy PA et al. J Infect Dis. 2021 Sep 30;224(12 Suppl 2):S420-8. doi: 10.1093/infdis/jiaa793.
4. Centers for Disease Control and Prevention. Measles (Rubeola). http://www.CDC.gov/Measles.
The U.S. immunization program is one of the best public health success stories. Physicians who provide care for children are familiar with the routine childhood immunization schedule and administer a measles-containing vaccine at age-appropriate times. Thanks to its rigorous implementation and acceptance, endemic measles (absence of continuous virus transmission for > 1 year) was eliminated in the U.S. in 2000. Loss of this status was in jeopardy in 2019 when 22 measles outbreaks occurred in 17 states (7 were multistate outbreaks). That year, 1,163 cases were reported.1 Most cases occurred in unvaccinated persons (89%) and 81 cases were imported of which 54 were in U.S. citizens returning from international travel. All outbreaks were linked to travel. Fortunately, the outbreaks were controlled prior to the elimination deadline, or the United States would have lost its measles elimination status. Restrictions on travel because of COVID-19 have relaxed significantly since the introduction of COVID-19 vaccines, resulting in increased regional and international travel. Multiple countries, including the United States noted a decline in routine immunizations rates during the last 2 years. Recent U.S. data for the 2020-2021 school year indicates that MMR immunizations rates (two doses) for kindergarteners declined to 93.9% (range 78.9% to > 98.9%), while the overall percentage of those students with an exemption remained low at 2.2%. Vaccine coverage greater than 95% was reported in only 16 states. Coverage of less than 90% was reported in seven states and the District of Columbia (Georgia, Idaho, Kentucky, Maryland, Minnesota, Ohio, and Wisconsin).2 Vaccine coverage should be 95% or higher to maintain herd immunity and control outbreaks.
Why is measles prevention so important? Many physicians practicing in the United States today have never seen a case or know its potential complications. I saw my first case as a resident in an immigrant child. It took our training director to point out the subtle signs and symptoms. It was the first time I saw Kolpik spots. Measles is transmitted person to person via large respiratory droplets and less often by airborne spread. It is highly contagious for susceptible individuals with an attack rate of 90%. In this case, a medical student on the team developed symptoms about 10 days later. Six years would pass before I diagnosed my next case of measles. An HIV patient acquired it after close contact with someone who was in the prodromal stage. He presented with the 3 C’s: Cough, coryza, and conjunctivitis, in addition to fever and an erythematous rash. He did not recover from complications of the disease.
Prior to the routine administration of a measles vaccine, 3-4 million cases with almost 500 deaths occurred annually in the United States. Worldwide, 35 million cases and more than 6 million deaths occurred each year. Here, most patients recover completely; however, complications including otitis media, pneumonia, croup, and encephalitis can develop. Complications commonly occur in immunocompromised individuals and young children. Groups with the highest fatality rates include children aged less than 5 years, immunocompromised persons, and pregnant women. Worldwide, fatality rates are dependent on the patients underlying nutritional and health status in addition to the quality of health care available.3
Measles vaccine was licensed in 1963 and cases began to decline (Figure 1). There was a resurgence in 1989 but it was not limited to the United States. The cause of the U.S. resurgence was multifactorial: Widespread viral transmission among unvaccinated preschool-age children residing in inner cities, outbreaks in vaccinated school-age children, outbreaks in students and personnel on college campuses, and primary vaccine failure (2%-5% of recipients failed to have an adequate response). In 1989, to help prevent future outbreaks, the United States recommended a two-dose schedule for measles and in 1993, the Vaccines for Children Program, a federally funded program, was established to improve access to vaccines for all children.
What is going on internationally?
Figure 2 lists the top 10 countries with current measles outbreaks.

Most countries on the list may not be typical travel destinations for tourists; however, they are common destinations for individuals visiting friends and relatives after immigrating to the United States. In contrast to the United States, most countries with limited resources and infrastructure have mass-vaccination campaigns to ensure vaccine administration to large segments of the population. They too have been affected by the COVID-19 pandemic. By report, at least 41 countries delayed implementation of their measles campaign in 2020 and 2021, thus, leading to the potential for even larger outbreaks.4
Progress toward the global elimination of measles is evidenced by the following: All 194 countries now include one dose of measles in their routine schedules; between 2000 and 2019 coverage of one dose of measles increased from 72% to 85% and countries with more than 90% coverage increased from 45% to 63%. Finally, the number of countries offering two doses of measles increased from 50% to 91% and vaccine coverage increased from 18% to 71% over the same time period.3
What can you do for your patients and their parents before they travel abroad?
- Inform all staff that the MMR vaccine can be administered to children as young as 6 months and at times other than those listed on the routine immunization schedule. This will help avoid parents seeking vaccine being denied an appointment.
- Children 6-11 months need 1 dose of MMR. Two additional doses will still need to be administered at the routine time.
- Children 12 months or older need 2 doses of MMR at least 4 weeks apart.
- If yellow fever vaccine is needed, coordinate administration with a travel medicine clinic since both are live vaccines and must be given on the same day.
- Any person born after 1956 should have 2 doses of MMR at least 4 weeks apart if they have no evidence of immunity.
- Encourage parents to always inform you and your staff of any international travel plans.
Moving forward, remember this increased global activity and the presence of inadequately vaccinated individuals/communities keeps the United States at continued risk for measles outbreaks. The source of the next outbreak may only be one plane ride away.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
This article was updated 6/29/22.
References
1. Patel M et al. MMWR. 2019 Oct 11; 68(40):893-6.
2. Seither R et al. MMWR. 2022 Apr 22;71(16):561-8.
3. Gastañaduy PA et al. J Infect Dis. 2021 Sep 30;224(12 Suppl 2):S420-8. doi: 10.1093/infdis/jiaa793.
4. Centers for Disease Control and Prevention. Measles (Rubeola). http://www.CDC.gov/Measles.
Adolescent immunizations and protecting our children from COVID-19
I began thinking of a topic for this column weeks ago determined to discuss anything except COVID-19. Yet, news reports from all sources blasted daily reminders of rising COVID-19 cases overall and specifically in children.
In August, school resumed for many of our patients and the battle over mandating masks for school attendance was in full swing. The fact that it is a Centers for Disease Control and Prevention recommendation supported by both the American Academy of Pediatrics and the Pediatric Infectious Disease Society fell on deaf ears. One day, I heard a report that over 25,000 students attending Texas public schools were diagnosed with COVID-19 between Aug. 23 and Aug. 29. This peak in activity occurred just 2 weeks after the start of school and led to the closure of 45 school districts. Texas does not have a monopoly on these rising cases. Delta, a more contagious variant, began circulating in June 2021 and by July it was the most predominant. Emergency department visits and hospitalizations have increased nationwide. During the latter 2 weeks of August 2021, COVID-19–related ED visits and hospitalizations for persons aged 0-17 years were 3.4 and 3.7 times higher in states with the lowest vaccination coverage, compared with states with high vaccination coverage (MMWR Morb Mortal Wkly Rep. 2021;70:1249-54). Specifically, the rates of hospitalization the week ending Aug. 14, 2021, were nearly 5 times the rates for the week ending June 26, 2021, for 0- to 17-year-olds and nearly 10 times the rates for children 0-4 years of age. Hospitalization rates were 10.1 times higher for unimmunized adolescents than for fully vaccinated ones (MMWR Morb Mortal Wkly Rep. 2021;70:1255-60).
Multiple elected state leaders have opposed interventions such as mandating masks in school, and our children are paying for it. These leaders have relinquished their responsibility to local school boards. Several have reinforced the no-mask mandate while others have had the courage and insight to ignore state government leaders and have established mask mandates.
How is this lack of enforcement of national recommendations affecting our patients? Let’s look at two neighboring school districts in Texas. School districts have COVID-19 dashboards that are updated daily and accessible to the general public. School District A requires masks for school entry. It serves 196,171 students and has 27,195 teachers and staff. Since school opened in August, 1,606 cumulative cases of COVID-19 in students (0.8%) and 282 in staff (1%) have been reported. Fifty-five percent of the student cases occurred in elementary schools. In contrast, School District B located in the adjacent county serves 64,517 students and has 3,906 teachers and staff with no mask mandate. Since August, there have been 4,506 cumulative COVID-19 cases in students (6.9%) and 578 (14.7%) in staff. Information regarding the specific school type was not provided; however, the dashboard indicates that 2,924 cases (64.8%) occurred in children younger than 11 years of age. County data indicate 62% of those older than 12 years of age were fully vaccinated in District A, compared with 54% of persons older than 12 years in District B. The county COVID-19 positivity rate in District A is 17.6% and in District B it is 20%. Both counties are experiencing increased COVID-19 activity yet have had strikingly different outcomes in the student/staff population. While supporting the case for wearing masks to prevent disease transmission, one can’t ignore the adolescents who were infected and vaccine eligible (District A: 706; District B: 1,582). Their vaccination status could not be determined.
As pediatricians we have played an integral part in the elimination of diseases through educating and administering vaccinations. Adolescents are relatively healthy, thus limiting the number of encounters with them. The majority complete the 11-year visit; however, many fail to return for the 16- to 18-year visit.
So how are we doing? CDC data from 10 U.S. jurisdictions demonstrated a substantial decrease in vaccine administration between March and May of 2020, compared with the same period in 2018 and 2019. A decline was anticipated because of the nationwide lockdown. Doses of HPV administered declined almost 64% and 71% for 9- to 12-year-olds and 13- to 17-year-olds, respectively. Tdap administration declined 66% and 61% for the same respective age groups. Although administered doses increased between June and September of 2020, it was not sufficient to achieve catch-up coverage. Compared to the same period in 2018-2019, administration of the HPV vaccine declined 12.8% and 28% (ages 9-12 and ages 13-17) and for Tdap it was 21% and 30% lower (ages 9-12 and ages 13-17) (MMWR Morb Mortal Wkly Rep. 2021;70:840-5).
Now, we have another adolescent vaccine to discuss and encourage our patients to receive. We also need to address their concerns and/or to at least direct them to a reliable source to obtain accurate information. For the first time, a recommended vaccine may not be available at their medical home. Many don’t know where to go to receive it (http://www.vaccines.gov). Results of a Kaiser Family Foundation COVID-19 survey (August 2021) indicated that parents trusted their pediatricians most often (78%) for vaccine advice. The respondents voiced concern about trusting the location where the child would be immunized and long-term effects especially related to fertility. Parents who received communications regarding the benefits of vaccination were twice as likely to have their adolescents immunized. Finally, remember: Like parent, like child. An immunized parent is more likely to immunize the adolescent. (See Fig. 1.)
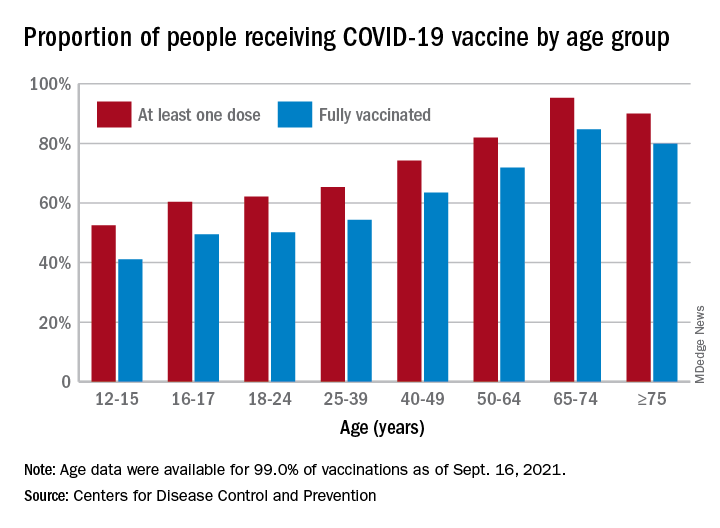
It is beyond the scope of this column to discuss the psychosocial aspects of this disease: children experiencing the death of teachers, classmates, family members, and those viewing the vitriol between pro- and antimask proponents often exhibited on school premises. And let’s not forget the child who wants to wear a mask but may be ostracized or bullied for doing so.
Our job is to do our very best to advocate for and to protect our patients by promoting mandatory masks at schools and encouraging vaccination of adolescents as we patiently wait for vaccines to become available for all of our children.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
I began thinking of a topic for this column weeks ago determined to discuss anything except COVID-19. Yet, news reports from all sources blasted daily reminders of rising COVID-19 cases overall and specifically in children.
In August, school resumed for many of our patients and the battle over mandating masks for school attendance was in full swing. The fact that it is a Centers for Disease Control and Prevention recommendation supported by both the American Academy of Pediatrics and the Pediatric Infectious Disease Society fell on deaf ears. One day, I heard a report that over 25,000 students attending Texas public schools were diagnosed with COVID-19 between Aug. 23 and Aug. 29. This peak in activity occurred just 2 weeks after the start of school and led to the closure of 45 school districts. Texas does not have a monopoly on these rising cases. Delta, a more contagious variant, began circulating in June 2021 and by July it was the most predominant. Emergency department visits and hospitalizations have increased nationwide. During the latter 2 weeks of August 2021, COVID-19–related ED visits and hospitalizations for persons aged 0-17 years were 3.4 and 3.7 times higher in states with the lowest vaccination coverage, compared with states with high vaccination coverage (MMWR Morb Mortal Wkly Rep. 2021;70:1249-54). Specifically, the rates of hospitalization the week ending Aug. 14, 2021, were nearly 5 times the rates for the week ending June 26, 2021, for 0- to 17-year-olds and nearly 10 times the rates for children 0-4 years of age. Hospitalization rates were 10.1 times higher for unimmunized adolescents than for fully vaccinated ones (MMWR Morb Mortal Wkly Rep. 2021;70:1255-60).
Multiple elected state leaders have opposed interventions such as mandating masks in school, and our children are paying for it. These leaders have relinquished their responsibility to local school boards. Several have reinforced the no-mask mandate while others have had the courage and insight to ignore state government leaders and have established mask mandates.
How is this lack of enforcement of national recommendations affecting our patients? Let’s look at two neighboring school districts in Texas. School districts have COVID-19 dashboards that are updated daily and accessible to the general public. School District A requires masks for school entry. It serves 196,171 students and has 27,195 teachers and staff. Since school opened in August, 1,606 cumulative cases of COVID-19 in students (0.8%) and 282 in staff (1%) have been reported. Fifty-five percent of the student cases occurred in elementary schools. In contrast, School District B located in the adjacent county serves 64,517 students and has 3,906 teachers and staff with no mask mandate. Since August, there have been 4,506 cumulative COVID-19 cases in students (6.9%) and 578 (14.7%) in staff. Information regarding the specific school type was not provided; however, the dashboard indicates that 2,924 cases (64.8%) occurred in children younger than 11 years of age. County data indicate 62% of those older than 12 years of age were fully vaccinated in District A, compared with 54% of persons older than 12 years in District B. The county COVID-19 positivity rate in District A is 17.6% and in District B it is 20%. Both counties are experiencing increased COVID-19 activity yet have had strikingly different outcomes in the student/staff population. While supporting the case for wearing masks to prevent disease transmission, one can’t ignore the adolescents who were infected and vaccine eligible (District A: 706; District B: 1,582). Their vaccination status could not be determined.
As pediatricians we have played an integral part in the elimination of diseases through educating and administering vaccinations. Adolescents are relatively healthy, thus limiting the number of encounters with them. The majority complete the 11-year visit; however, many fail to return for the 16- to 18-year visit.
So how are we doing? CDC data from 10 U.S. jurisdictions demonstrated a substantial decrease in vaccine administration between March and May of 2020, compared with the same period in 2018 and 2019. A decline was anticipated because of the nationwide lockdown. Doses of HPV administered declined almost 64% and 71% for 9- to 12-year-olds and 13- to 17-year-olds, respectively. Tdap administration declined 66% and 61% for the same respective age groups. Although administered doses increased between June and September of 2020, it was not sufficient to achieve catch-up coverage. Compared to the same period in 2018-2019, administration of the HPV vaccine declined 12.8% and 28% (ages 9-12 and ages 13-17) and for Tdap it was 21% and 30% lower (ages 9-12 and ages 13-17) (MMWR Morb Mortal Wkly Rep. 2021;70:840-5).
Now, we have another adolescent vaccine to discuss and encourage our patients to receive. We also need to address their concerns and/or to at least direct them to a reliable source to obtain accurate information. For the first time, a recommended vaccine may not be available at their medical home. Many don’t know where to go to receive it (http://www.vaccines.gov). Results of a Kaiser Family Foundation COVID-19 survey (August 2021) indicated that parents trusted their pediatricians most often (78%) for vaccine advice. The respondents voiced concern about trusting the location where the child would be immunized and long-term effects especially related to fertility. Parents who received communications regarding the benefits of vaccination were twice as likely to have their adolescents immunized. Finally, remember: Like parent, like child. An immunized parent is more likely to immunize the adolescent. (See Fig. 1.)

It is beyond the scope of this column to discuss the psychosocial aspects of this disease: children experiencing the death of teachers, classmates, family members, and those viewing the vitriol between pro- and antimask proponents often exhibited on school premises. And let’s not forget the child who wants to wear a mask but may be ostracized or bullied for doing so.
Our job is to do our very best to advocate for and to protect our patients by promoting mandatory masks at schools and encouraging vaccination of adolescents as we patiently wait for vaccines to become available for all of our children.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
I began thinking of a topic for this column weeks ago determined to discuss anything except COVID-19. Yet, news reports from all sources blasted daily reminders of rising COVID-19 cases overall and specifically in children.
In August, school resumed for many of our patients and the battle over mandating masks for school attendance was in full swing. The fact that it is a Centers for Disease Control and Prevention recommendation supported by both the American Academy of Pediatrics and the Pediatric Infectious Disease Society fell on deaf ears. One day, I heard a report that over 25,000 students attending Texas public schools were diagnosed with COVID-19 between Aug. 23 and Aug. 29. This peak in activity occurred just 2 weeks after the start of school and led to the closure of 45 school districts. Texas does not have a monopoly on these rising cases. Delta, a more contagious variant, began circulating in June 2021 and by July it was the most predominant. Emergency department visits and hospitalizations have increased nationwide. During the latter 2 weeks of August 2021, COVID-19–related ED visits and hospitalizations for persons aged 0-17 years were 3.4 and 3.7 times higher in states with the lowest vaccination coverage, compared with states with high vaccination coverage (MMWR Morb Mortal Wkly Rep. 2021;70:1249-54). Specifically, the rates of hospitalization the week ending Aug. 14, 2021, were nearly 5 times the rates for the week ending June 26, 2021, for 0- to 17-year-olds and nearly 10 times the rates for children 0-4 years of age. Hospitalization rates were 10.1 times higher for unimmunized adolescents than for fully vaccinated ones (MMWR Morb Mortal Wkly Rep. 2021;70:1255-60).
Multiple elected state leaders have opposed interventions such as mandating masks in school, and our children are paying for it. These leaders have relinquished their responsibility to local school boards. Several have reinforced the no-mask mandate while others have had the courage and insight to ignore state government leaders and have established mask mandates.
How is this lack of enforcement of national recommendations affecting our patients? Let’s look at two neighboring school districts in Texas. School districts have COVID-19 dashboards that are updated daily and accessible to the general public. School District A requires masks for school entry. It serves 196,171 students and has 27,195 teachers and staff. Since school opened in August, 1,606 cumulative cases of COVID-19 in students (0.8%) and 282 in staff (1%) have been reported. Fifty-five percent of the student cases occurred in elementary schools. In contrast, School District B located in the adjacent county serves 64,517 students and has 3,906 teachers and staff with no mask mandate. Since August, there have been 4,506 cumulative COVID-19 cases in students (6.9%) and 578 (14.7%) in staff. Information regarding the specific school type was not provided; however, the dashboard indicates that 2,924 cases (64.8%) occurred in children younger than 11 years of age. County data indicate 62% of those older than 12 years of age were fully vaccinated in District A, compared with 54% of persons older than 12 years in District B. The county COVID-19 positivity rate in District A is 17.6% and in District B it is 20%. Both counties are experiencing increased COVID-19 activity yet have had strikingly different outcomes in the student/staff population. While supporting the case for wearing masks to prevent disease transmission, one can’t ignore the adolescents who were infected and vaccine eligible (District A: 706; District B: 1,582). Their vaccination status could not be determined.
As pediatricians we have played an integral part in the elimination of diseases through educating and administering vaccinations. Adolescents are relatively healthy, thus limiting the number of encounters with them. The majority complete the 11-year visit; however, many fail to return for the 16- to 18-year visit.
So how are we doing? CDC data from 10 U.S. jurisdictions demonstrated a substantial decrease in vaccine administration between March and May of 2020, compared with the same period in 2018 and 2019. A decline was anticipated because of the nationwide lockdown. Doses of HPV administered declined almost 64% and 71% for 9- to 12-year-olds and 13- to 17-year-olds, respectively. Tdap administration declined 66% and 61% for the same respective age groups. Although administered doses increased between June and September of 2020, it was not sufficient to achieve catch-up coverage. Compared to the same period in 2018-2019, administration of the HPV vaccine declined 12.8% and 28% (ages 9-12 and ages 13-17) and for Tdap it was 21% and 30% lower (ages 9-12 and ages 13-17) (MMWR Morb Mortal Wkly Rep. 2021;70:840-5).
Now, we have another adolescent vaccine to discuss and encourage our patients to receive. We also need to address their concerns and/or to at least direct them to a reliable source to obtain accurate information. For the first time, a recommended vaccine may not be available at their medical home. Many don’t know where to go to receive it (http://www.vaccines.gov). Results of a Kaiser Family Foundation COVID-19 survey (August 2021) indicated that parents trusted their pediatricians most often (78%) for vaccine advice. The respondents voiced concern about trusting the location where the child would be immunized and long-term effects especially related to fertility. Parents who received communications regarding the benefits of vaccination were twice as likely to have their adolescents immunized. Finally, remember: Like parent, like child. An immunized parent is more likely to immunize the adolescent. (See Fig. 1.)

It is beyond the scope of this column to discuss the psychosocial aspects of this disease: children experiencing the death of teachers, classmates, family members, and those viewing the vitriol between pro- and antimask proponents often exhibited on school premises. And let’s not forget the child who wants to wear a mask but may be ostracized or bullied for doing so.
Our job is to do our very best to advocate for and to protect our patients by promoting mandatory masks at schools and encouraging vaccination of adolescents as we patiently wait for vaccines to become available for all of our children.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Tick talk for families and pediatricians
Spring 2021 has arrived with summer quickly approaching. It is our second spring and summer during the pandemic. Travel restrictions have minimally eased for vaccinated adults. However, neither domestic nor international leisure travel is encouraged for anyone. Ironically, air travel is increasing. For many families, it is time to make decisions regarding summer activities. Outdoor activities have been encouraged throughout the pandemic, which makes it a good time to review tick-borne diseases. Depending on your location, your patients may only have to travel as far as their backyard to sustain a tick bite.
Ticks are a group of obligate, bloodsucking arthropods that feed on mammals, birds, and reptiles. There are three families of ticks. Two families, Ixodidae (hard-bodied ticks) and Argasidae (soft-bodied ticks) are responsible for transmitting the most diseases to humans in the United States. Once a tick is infected with a pathogen it usually survives and transmits it to its next host. Ticks efficiently transmit bacteria, spirochetes, protozoa, rickettsiae, nematodes, and toxins to humans during feeding when the site is exposed to infected salivary gland secretions or regurgitated midgut contents. Pathogen transmission can also occur when the feeding site is contaminated by feces or coxal fluid. Sometimes a tick can transmit multiple pathogens. Not all pathogens are infectious (e.g., tick paralysis, which occurs after exposure to a neurotoxin and red meat allergy because of alpha-gal). Ticks require a blood meal to transform to their next stage of development (larva to nymph to adult). Life cycles of hard and soft ticks differ with most hard ticks undergoing a 2-year life cycle and feeding slowly over many days. In contrast, soft ticks feed multiple times often for less than 1 hour and are capable of transmitting diseases in less than 1 minute.
Rocky Mountain spotted fever was the first recognized tick-borne disease (TBD) in humans. Since then, 18 additional pathogens transmitted by ticks have been identified with 40% being described since 1980. The increased discovery of tickborne pathogens has been attributed to physician awareness of TBD and improved diagnostics. The number of cases of TBD has risen yearly. Ticks are responsible for most vector-transmitted diseases in the United States with Lyme disease most frequently reported.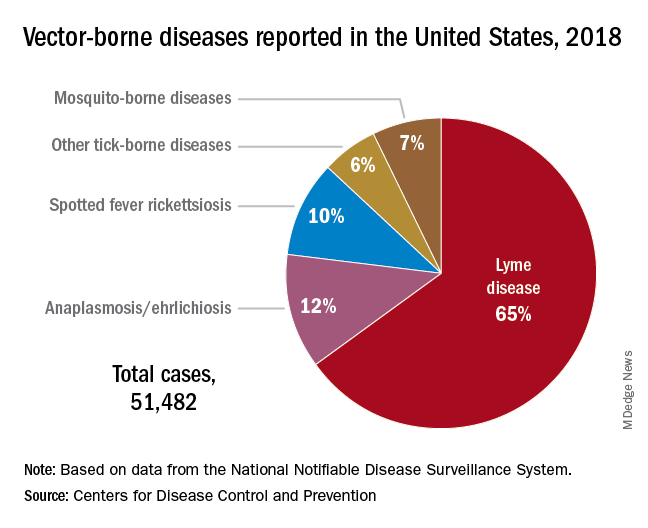
Mosquito transmission accounts for only 7% of vector-borne diseases. Three species of ticks are responsible for most human disease: Ixodes scapularis (Black-legged tick), Amblyomma americanum (Lone Star tick), and Dermacentor variabilis (American dog tick). Each is capable of transmitting agents that cause multiple diseases.
Risk for acquisition of a specific disease is dependent upon the type of tick, its geographic location, the season, and duration of the exposure.
Humans are usually incidental hosts. Tick exposure can occur year-round, but tick activity is greatest between April and September. Ticks are generally found near the ground, in brushy or wooded areas. They can climb tall grasses or shrubs and wait for a potential host to brush against them. When this occurs, they seek a site for attachment.
In the absence of a vaccine, prevention of TBD is totally dependent upon your patients/parents understanding of when and where they are at risk for exposure and for us as physicians to know which pathogens can potentially be transmitted by ticks. Data regarding potential exposure risks are based on where a TBD was diagnosed, not necessarily where it was acquired. National maps that illustrate the distribution of medically significant ticks and presence or prevalence of tick-borne pathogens in specific areas within a region previously may have been incomplete or outdated. The Centers for Disease Control and Prevention initiated a national tick surveillance program in 2017; five universities were established as regional centers of excellence to help prevent and rapidly respond to emerging vector-borne diseases across the United States. One goal is to standardize tick surveillance activities at the state level. For state-specific activity go to https://www.cdc.gov/ncezid/dvbd/vital-signs/index.html.
Prevention: Here are a few environmental interventions you can recommend to your patients
- Remove leaf litter, clear tall brush, and grass around the home and at edge of lawns. Mow the lawn frequently.
- Keep playground equipment, decks, and patios away from yard edges and trees.
- Live near a wooded area? Place a 3-ft.-wide barrier of gravel or wood chips between the areas.
- Put up a fence to keep unwanted animals out.
- Keep the yard free of potential hiding place for ticks (e.g., mattresses or furniture).
- Stack wood neatly and in a dry area.
- Use pesticides, but do not rely on them solely to prevent ticks exposure.
Personal interventions for patients when outdoors
- Use Environmental Protection Agency–registered insect repellents. Note: Oil of lemon-, eucalyptus-, and para-menthane-diol–containing products should not be used in children aged3 years or less.
- Treat clothing and gear with products containing 0.5% permethrin to repel mosquitoes and ticks.
- Check cloths for ticks. Drying clothes on high heat for 10 minutes will kill ticks. If washing is needed use hot water. Lower temperatures will not kill ticks.
- Do daily body checks for ticks after coming indoors.
- Check pets for ticks.
Tick removal
- Take tweezers, grasp the tick as close to the skin’s surface as possible.
- Pull upward. Do not twist or jerk the tick. Place in a container. Ideally submit for species identification.
- After removal, clean the bite area with alcohol or soap and water.
- Never crush a tick with your fingers.
When should you include TBD in your differential for a sick child?
Headache, fever, arthralgia, and rash are symptoms for several infectious diseases. Obtaining a history of recent activities, tick bite, or travel to areas where these diseases are more prevalent is important. You must have a high index of suspicion. Clinical and laboratory clues may help.
Delay in treatment is more detrimental. If you suspect rickettsia, ehrlichiosis, or anaplasmosis, doxycycline should be started promptly regardless of age. Consultation with an infectious disease specialist is recommended.
The United States recognizes it is not adequately prepared to address the continuing rise of vector-borne diseases. In response, on Jan. 20, 2021, the CDC’s division of vector-borne diseases with input from five federal departments and the EPA developed a joint National Public Health Framework for the Prevention and Control of Vector-Borne Diseases in Humans to tackle issues including risk, detection, diagnosis, treatment, prevention and control of TBD. Stay tuned.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Spring 2021 has arrived with summer quickly approaching. It is our second spring and summer during the pandemic. Travel restrictions have minimally eased for vaccinated adults. However, neither domestic nor international leisure travel is encouraged for anyone. Ironically, air travel is increasing. For many families, it is time to make decisions regarding summer activities. Outdoor activities have been encouraged throughout the pandemic, which makes it a good time to review tick-borne diseases. Depending on your location, your patients may only have to travel as far as their backyard to sustain a tick bite.
Ticks are a group of obligate, bloodsucking arthropods that feed on mammals, birds, and reptiles. There are three families of ticks. Two families, Ixodidae (hard-bodied ticks) and Argasidae (soft-bodied ticks) are responsible for transmitting the most diseases to humans in the United States. Once a tick is infected with a pathogen it usually survives and transmits it to its next host. Ticks efficiently transmit bacteria, spirochetes, protozoa, rickettsiae, nematodes, and toxins to humans during feeding when the site is exposed to infected salivary gland secretions or regurgitated midgut contents. Pathogen transmission can also occur when the feeding site is contaminated by feces or coxal fluid. Sometimes a tick can transmit multiple pathogens. Not all pathogens are infectious (e.g., tick paralysis, which occurs after exposure to a neurotoxin and red meat allergy because of alpha-gal). Ticks require a blood meal to transform to their next stage of development (larva to nymph to adult). Life cycles of hard and soft ticks differ with most hard ticks undergoing a 2-year life cycle and feeding slowly over many days. In contrast, soft ticks feed multiple times often for less than 1 hour and are capable of transmitting diseases in less than 1 minute.
Rocky Mountain spotted fever was the first recognized tick-borne disease (TBD) in humans. Since then, 18 additional pathogens transmitted by ticks have been identified with 40% being described since 1980. The increased discovery of tickborne pathogens has been attributed to physician awareness of TBD and improved diagnostics. The number of cases of TBD has risen yearly. Ticks are responsible for most vector-transmitted diseases in the United States with Lyme disease most frequently reported.
Mosquito transmission accounts for only 7% of vector-borne diseases. Three species of ticks are responsible for most human disease: Ixodes scapularis (Black-legged tick), Amblyomma americanum (Lone Star tick), and Dermacentor variabilis (American dog tick). Each is capable of transmitting agents that cause multiple diseases.
Risk for acquisition of a specific disease is dependent upon the type of tick, its geographic location, the season, and duration of the exposure.
Humans are usually incidental hosts. Tick exposure can occur year-round, but tick activity is greatest between April and September. Ticks are generally found near the ground, in brushy or wooded areas. They can climb tall grasses or shrubs and wait for a potential host to brush against them. When this occurs, they seek a site for attachment.
In the absence of a vaccine, prevention of TBD is totally dependent upon your patients/parents understanding of when and where they are at risk for exposure and for us as physicians to know which pathogens can potentially be transmitted by ticks. Data regarding potential exposure risks are based on where a TBD was diagnosed, not necessarily where it was acquired. National maps that illustrate the distribution of medically significant ticks and presence or prevalence of tick-borne pathogens in specific areas within a region previously may have been incomplete or outdated. The Centers for Disease Control and Prevention initiated a national tick surveillance program in 2017; five universities were established as regional centers of excellence to help prevent and rapidly respond to emerging vector-borne diseases across the United States. One goal is to standardize tick surveillance activities at the state level. For state-specific activity go to https://www.cdc.gov/ncezid/dvbd/vital-signs/index.html.
Prevention: Here are a few environmental interventions you can recommend to your patients
- Remove leaf litter, clear tall brush, and grass around the home and at edge of lawns. Mow the lawn frequently.
- Keep playground equipment, decks, and patios away from yard edges and trees.
- Live near a wooded area? Place a 3-ft.-wide barrier of gravel or wood chips between the areas.
- Put up a fence to keep unwanted animals out.
- Keep the yard free of potential hiding place for ticks (e.g., mattresses or furniture).
- Stack wood neatly and in a dry area.
- Use pesticides, but do not rely on them solely to prevent ticks exposure.
Personal interventions for patients when outdoors
- Use Environmental Protection Agency–registered insect repellents. Note: Oil of lemon-, eucalyptus-, and para-menthane-diol–containing products should not be used in children aged3 years or less.
- Treat clothing and gear with products containing 0.5% permethrin to repel mosquitoes and ticks.
- Check cloths for ticks. Drying clothes on high heat for 10 minutes will kill ticks. If washing is needed use hot water. Lower temperatures will not kill ticks.
- Do daily body checks for ticks after coming indoors.
- Check pets for ticks.
Tick removal
- Take tweezers, grasp the tick as close to the skin’s surface as possible.
- Pull upward. Do not twist or jerk the tick. Place in a container. Ideally submit for species identification.
- After removal, clean the bite area with alcohol or soap and water.
- Never crush a tick with your fingers.
When should you include TBD in your differential for a sick child?
Headache, fever, arthralgia, and rash are symptoms for several infectious diseases. Obtaining a history of recent activities, tick bite, or travel to areas where these diseases are more prevalent is important. You must have a high index of suspicion. Clinical and laboratory clues may help.
Delay in treatment is more detrimental. If you suspect rickettsia, ehrlichiosis, or anaplasmosis, doxycycline should be started promptly regardless of age. Consultation with an infectious disease specialist is recommended.
The United States recognizes it is not adequately prepared to address the continuing rise of vector-borne diseases. In response, on Jan. 20, 2021, the CDC’s division of vector-borne diseases with input from five federal departments and the EPA developed a joint National Public Health Framework for the Prevention and Control of Vector-Borne Diseases in Humans to tackle issues including risk, detection, diagnosis, treatment, prevention and control of TBD. Stay tuned.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Spring 2021 has arrived with summer quickly approaching. It is our second spring and summer during the pandemic. Travel restrictions have minimally eased for vaccinated adults. However, neither domestic nor international leisure travel is encouraged for anyone. Ironically, air travel is increasing. For many families, it is time to make decisions regarding summer activities. Outdoor activities have been encouraged throughout the pandemic, which makes it a good time to review tick-borne diseases. Depending on your location, your patients may only have to travel as far as their backyard to sustain a tick bite.
Ticks are a group of obligate, bloodsucking arthropods that feed on mammals, birds, and reptiles. There are three families of ticks. Two families, Ixodidae (hard-bodied ticks) and Argasidae (soft-bodied ticks) are responsible for transmitting the most diseases to humans in the United States. Once a tick is infected with a pathogen it usually survives and transmits it to its next host. Ticks efficiently transmit bacteria, spirochetes, protozoa, rickettsiae, nematodes, and toxins to humans during feeding when the site is exposed to infected salivary gland secretions or regurgitated midgut contents. Pathogen transmission can also occur when the feeding site is contaminated by feces or coxal fluid. Sometimes a tick can transmit multiple pathogens. Not all pathogens are infectious (e.g., tick paralysis, which occurs after exposure to a neurotoxin and red meat allergy because of alpha-gal). Ticks require a blood meal to transform to their next stage of development (larva to nymph to adult). Life cycles of hard and soft ticks differ with most hard ticks undergoing a 2-year life cycle and feeding slowly over many days. In contrast, soft ticks feed multiple times often for less than 1 hour and are capable of transmitting diseases in less than 1 minute.
Rocky Mountain spotted fever was the first recognized tick-borne disease (TBD) in humans. Since then, 18 additional pathogens transmitted by ticks have been identified with 40% being described since 1980. The increased discovery of tickborne pathogens has been attributed to physician awareness of TBD and improved diagnostics. The number of cases of TBD has risen yearly. Ticks are responsible for most vector-transmitted diseases in the United States with Lyme disease most frequently reported.
Mosquito transmission accounts for only 7% of vector-borne diseases. Three species of ticks are responsible for most human disease: Ixodes scapularis (Black-legged tick), Amblyomma americanum (Lone Star tick), and Dermacentor variabilis (American dog tick). Each is capable of transmitting agents that cause multiple diseases.
Risk for acquisition of a specific disease is dependent upon the type of tick, its geographic location, the season, and duration of the exposure.
Humans are usually incidental hosts. Tick exposure can occur year-round, but tick activity is greatest between April and September. Ticks are generally found near the ground, in brushy or wooded areas. They can climb tall grasses or shrubs and wait for a potential host to brush against them. When this occurs, they seek a site for attachment.
In the absence of a vaccine, prevention of TBD is totally dependent upon your patients/parents understanding of when and where they are at risk for exposure and for us as physicians to know which pathogens can potentially be transmitted by ticks. Data regarding potential exposure risks are based on where a TBD was diagnosed, not necessarily where it was acquired. National maps that illustrate the distribution of medically significant ticks and presence or prevalence of tick-borne pathogens in specific areas within a region previously may have been incomplete or outdated. The Centers for Disease Control and Prevention initiated a national tick surveillance program in 2017; five universities were established as regional centers of excellence to help prevent and rapidly respond to emerging vector-borne diseases across the United States. One goal is to standardize tick surveillance activities at the state level. For state-specific activity go to https://www.cdc.gov/ncezid/dvbd/vital-signs/index.html.
Prevention: Here are a few environmental interventions you can recommend to your patients
- Remove leaf litter, clear tall brush, and grass around the home and at edge of lawns. Mow the lawn frequently.
- Keep playground equipment, decks, and patios away from yard edges and trees.
- Live near a wooded area? Place a 3-ft.-wide barrier of gravel or wood chips between the areas.
- Put up a fence to keep unwanted animals out.
- Keep the yard free of potential hiding place for ticks (e.g., mattresses or furniture).
- Stack wood neatly and in a dry area.
- Use pesticides, but do not rely on them solely to prevent ticks exposure.
Personal interventions for patients when outdoors
- Use Environmental Protection Agency–registered insect repellents. Note: Oil of lemon-, eucalyptus-, and para-menthane-diol–containing products should not be used in children aged3 years or less.
- Treat clothing and gear with products containing 0.5% permethrin to repel mosquitoes and ticks.
- Check cloths for ticks. Drying clothes on high heat for 10 minutes will kill ticks. If washing is needed use hot water. Lower temperatures will not kill ticks.
- Do daily body checks for ticks after coming indoors.
- Check pets for ticks.
Tick removal
- Take tweezers, grasp the tick as close to the skin’s surface as possible.
- Pull upward. Do not twist or jerk the tick. Place in a container. Ideally submit for species identification.
- After removal, clean the bite area with alcohol or soap and water.
- Never crush a tick with your fingers.
When should you include TBD in your differential for a sick child?
Headache, fever, arthralgia, and rash are symptoms for several infectious diseases. Obtaining a history of recent activities, tick bite, or travel to areas where these diseases are more prevalent is important. You must have a high index of suspicion. Clinical and laboratory clues may help.
Delay in treatment is more detrimental. If you suspect rickettsia, ehrlichiosis, or anaplasmosis, doxycycline should be started promptly regardless of age. Consultation with an infectious disease specialist is recommended.
The United States recognizes it is not adequately prepared to address the continuing rise of vector-borne diseases. In response, on Jan. 20, 2021, the CDC’s division of vector-borne diseases with input from five federal departments and the EPA developed a joint National Public Health Framework for the Prevention and Control of Vector-Borne Diseases in Humans to tackle issues including risk, detection, diagnosis, treatment, prevention and control of TBD. Stay tuned.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures.
Should our patients really go home for the holidays?
As an East Coast transplant residing in Texas, I look forward to the annual sojourn home to celebrate the holidays with family and friends – as do many of our patients and their families. But this is 2020. SARS-CoV-2, the causative agent of COVID-19, is still circulating. To make matters worse, cases are rising in 45 states and internationally. The day of this writing 102,831 new cases were reported in the United States. 
Social distancing, wearing masks, and hand washing have been strategies recommended to help mitigate the spread of the virus. We know adherence is not always 100%. The reality is that several families will consider traveling and gathering with others over the holidays. Their actions may lead to increased infections, hospitalizations, and even deaths. It behooves us to at least remind them of the potential consequences of the activity, and if travel and/or holiday gatherings are inevitable, to provide some guidance to help them look at both the risks and benefits and offer strategies to minimize infection and spread.
What should be considered prior to travel?
Here is a list of points to ponder:
- Is your patient is in a high-risk group for developing severe disease or visiting someone who is in a high-risk group?
- What is their mode of transportation?
- What is their destination?
- How prevalent is the disease at their destination, compared with their community?
- What will be their accommodations?
- How will attendees prepare for the gathering, if at all?
- Will multiple families congregate after quarantining for 2 weeks or simply arrive?
- At the destination, will people wear masks and socially distance?
- Is an outdoor venue an option?
All of these questions should be considered by patients.
Review high-risk groups
In terms of high-risk groups, we usually focus on underlying medical conditions or extremes of age, but Black and LatinX children and their families have been diagnosed with COVID-19 and hospitalized more frequently than other racial/ ethnic groups in the United States. Of 277,285 school-aged children infected between March 1 and Sept. 19, 2020, 42% were LatinX, 32% White, and 17% Black, yet they comprise 18%, 60%, and 11% of the U.S. population, respectively. Of those hospitalized, 45% were LatinX, 22% White, and 24% Black. LatinX and Black children also have disproportionately higher mortality rates.
Think about transmission and how to mitigate it
Many patients erroneously think combining multiple households for small group gatherings is inconsequential. These types of gatherings serve as a continued source of SARS-CoV-2 spread. For example, a person in Illinois with mild upper respiratory infection symptoms attended a funeral; he reported embracing the family members after the funeral. He dined with two people the evening prior to the funeral, sharing the meal using common serving dishes. Four days later, he attended a birthday party with nine family members. Some of the family members with symptoms subsequently attended church, infecting another church attendee. A cluster of 16 cases of COVID-19 was subsequently identified, including three deaths likely resulting from this one introduction of COVID-19 at these two family gatherings.
In Tennessee and Wisconsin, household transmission of SARS-CoV-2 was studied prospectively. A total of 101 index cases and 191 asymptomatic household contacts were enrolled between April and Sept. 2020; 102 of 191 (53%) had SARS-CoV-2 detected during the 14-day follow-up. Most infections (75%) were identified within 5 days and occurred whether the index case was an adult or child.
Lastly, one adolescent was identified as the source for an outbreak at a family gathering where 15 persons from five households and four states shared a house between 8 and 25 days in July 2020. Six additional members visited the house. The index case had an exposure to COVID-19 and had a negative antigen test 4 days after exposure. She was asymptomatic when tested. She developed nasal congestion 2 days later, the same day she and her family departed for the gathering. A total of 11 household contacts developed confirmed, suspected, or probable COVID-19, and the teen developed symptoms. This report illustrates how easily SARS-CoV-2 is transmitted, and how when implemented, mitigation strategies work because none of the six who only visited the house was infected. It also serves as a reminder that antigen testing is indicated only for use within the first 5-12 days of onset of symptoms. In this case, the adolescent was asymptomatic when tested and had a false-negative test result.
Ponder modes of transportation
How will your patient arrive to their holiday destination? Nonstop travel by car with household members is probably the safest way. However, for many families, buses and trains are the only options, and social distancing may be challenging. Air travel is a must for others. Acquisition of COVID-19 during air travel appears to be low, but not absent based on how air enters and leaves the cabin. The challenge is socially distancing throughout the check in and boarding processes, as well as minimizing contact with common surfaces. There also is loss of social distancing once on board. Ideally, masks should be worn during the flight. Additionally, for those with international destinations, most countries now require a negative polymerase chain reaction COVID-19 test within a specified time frame for entry.
Essentially the safest place for your patients during the holidays is celebrating at home with their household contacts. The risk for disease acquisition increases with travel. You will not have the opportunity to discuss holiday plans with most parents. However, you can encourage them to consider the pros and cons of travel with reminders via telephone, e-mail, and /or social messaging directly from your practices similar to those sent for other medically necessary interventions. As for me, I will be celebrating virtually this year. There is a first time for everything.
For additional information that also is patient friendly, the Centers for Disease Control and Prevention offers information about travel within the United States and international travel.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].
As an East Coast transplant residing in Texas, I look forward to the annual sojourn home to celebrate the holidays with family and friends – as do many of our patients and their families. But this is 2020. SARS-CoV-2, the causative agent of COVID-19, is still circulating. To make matters worse, cases are rising in 45 states and internationally. The day of this writing 102,831 new cases were reported in the United States. 
Social distancing, wearing masks, and hand washing have been strategies recommended to help mitigate the spread of the virus. We know adherence is not always 100%. The reality is that several families will consider traveling and gathering with others over the holidays. Their actions may lead to increased infections, hospitalizations, and even deaths. It behooves us to at least remind them of the potential consequences of the activity, and if travel and/or holiday gatherings are inevitable, to provide some guidance to help them look at both the risks and benefits and offer strategies to minimize infection and spread.
What should be considered prior to travel?
Here is a list of points to ponder:
- Is your patient is in a high-risk group for developing severe disease or visiting someone who is in a high-risk group?
- What is their mode of transportation?
- What is their destination?
- How prevalent is the disease at their destination, compared with their community?
- What will be their accommodations?
- How will attendees prepare for the gathering, if at all?
- Will multiple families congregate after quarantining for 2 weeks or simply arrive?
- At the destination, will people wear masks and socially distance?
- Is an outdoor venue an option?
All of these questions should be considered by patients.
Review high-risk groups
In terms of high-risk groups, we usually focus on underlying medical conditions or extremes of age, but Black and LatinX children and their families have been diagnosed with COVID-19 and hospitalized more frequently than other racial/ ethnic groups in the United States. Of 277,285 school-aged children infected between March 1 and Sept. 19, 2020, 42% were LatinX, 32% White, and 17% Black, yet they comprise 18%, 60%, and 11% of the U.S. population, respectively. Of those hospitalized, 45% were LatinX, 22% White, and 24% Black. LatinX and Black children also have disproportionately higher mortality rates.
Think about transmission and how to mitigate it
Many patients erroneously think combining multiple households for small group gatherings is inconsequential. These types of gatherings serve as a continued source of SARS-CoV-2 spread. For example, a person in Illinois with mild upper respiratory infection symptoms attended a funeral; he reported embracing the family members after the funeral. He dined with two people the evening prior to the funeral, sharing the meal using common serving dishes. Four days later, he attended a birthday party with nine family members. Some of the family members with symptoms subsequently attended church, infecting another church attendee. A cluster of 16 cases of COVID-19 was subsequently identified, including three deaths likely resulting from this one introduction of COVID-19 at these two family gatherings.
In Tennessee and Wisconsin, household transmission of SARS-CoV-2 was studied prospectively. A total of 101 index cases and 191 asymptomatic household contacts were enrolled between April and Sept. 2020; 102 of 191 (53%) had SARS-CoV-2 detected during the 14-day follow-up. Most infections (75%) were identified within 5 days and occurred whether the index case was an adult or child.
Lastly, one adolescent was identified as the source for an outbreak at a family gathering where 15 persons from five households and four states shared a house between 8 and 25 days in July 2020. Six additional members visited the house. The index case had an exposure to COVID-19 and had a negative antigen test 4 days after exposure. She was asymptomatic when tested. She developed nasal congestion 2 days later, the same day she and her family departed for the gathering. A total of 11 household contacts developed confirmed, suspected, or probable COVID-19, and the teen developed symptoms. This report illustrates how easily SARS-CoV-2 is transmitted, and how when implemented, mitigation strategies work because none of the six who only visited the house was infected. It also serves as a reminder that antigen testing is indicated only for use within the first 5-12 days of onset of symptoms. In this case, the adolescent was asymptomatic when tested and had a false-negative test result.
Ponder modes of transportation
How will your patient arrive to their holiday destination? Nonstop travel by car with household members is probably the safest way. However, for many families, buses and trains are the only options, and social distancing may be challenging. Air travel is a must for others. Acquisition of COVID-19 during air travel appears to be low, but not absent based on how air enters and leaves the cabin. The challenge is socially distancing throughout the check in and boarding processes, as well as minimizing contact with common surfaces. There also is loss of social distancing once on board. Ideally, masks should be worn during the flight. Additionally, for those with international destinations, most countries now require a negative polymerase chain reaction COVID-19 test within a specified time frame for entry.
Essentially the safest place for your patients during the holidays is celebrating at home with their household contacts. The risk for disease acquisition increases with travel. You will not have the opportunity to discuss holiday plans with most parents. However, you can encourage them to consider the pros and cons of travel with reminders via telephone, e-mail, and /or social messaging directly from your practices similar to those sent for other medically necessary interventions. As for me, I will be celebrating virtually this year. There is a first time for everything.
For additional information that also is patient friendly, the Centers for Disease Control and Prevention offers information about travel within the United States and international travel.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].
As an East Coast transplant residing in Texas, I look forward to the annual sojourn home to celebrate the holidays with family and friends – as do many of our patients and their families. But this is 2020. SARS-CoV-2, the causative agent of COVID-19, is still circulating. To make matters worse, cases are rising in 45 states and internationally. The day of this writing 102,831 new cases were reported in the United States. 
Social distancing, wearing masks, and hand washing have been strategies recommended to help mitigate the spread of the virus. We know adherence is not always 100%. The reality is that several families will consider traveling and gathering with others over the holidays. Their actions may lead to increased infections, hospitalizations, and even deaths. It behooves us to at least remind them of the potential consequences of the activity, and if travel and/or holiday gatherings are inevitable, to provide some guidance to help them look at both the risks and benefits and offer strategies to minimize infection and spread.
What should be considered prior to travel?
Here is a list of points to ponder:
- Is your patient is in a high-risk group for developing severe disease or visiting someone who is in a high-risk group?
- What is their mode of transportation?
- What is their destination?
- How prevalent is the disease at their destination, compared with their community?
- What will be their accommodations?
- How will attendees prepare for the gathering, if at all?
- Will multiple families congregate after quarantining for 2 weeks or simply arrive?
- At the destination, will people wear masks and socially distance?
- Is an outdoor venue an option?
All of these questions should be considered by patients.
Review high-risk groups
In terms of high-risk groups, we usually focus on underlying medical conditions or extremes of age, but Black and LatinX children and their families have been diagnosed with COVID-19 and hospitalized more frequently than other racial/ ethnic groups in the United States. Of 277,285 school-aged children infected between March 1 and Sept. 19, 2020, 42% were LatinX, 32% White, and 17% Black, yet they comprise 18%, 60%, and 11% of the U.S. population, respectively. Of those hospitalized, 45% were LatinX, 22% White, and 24% Black. LatinX and Black children also have disproportionately higher mortality rates.
Think about transmission and how to mitigate it
Many patients erroneously think combining multiple households for small group gatherings is inconsequential. These types of gatherings serve as a continued source of SARS-CoV-2 spread. For example, a person in Illinois with mild upper respiratory infection symptoms attended a funeral; he reported embracing the family members after the funeral. He dined with two people the evening prior to the funeral, sharing the meal using common serving dishes. Four days later, he attended a birthday party with nine family members. Some of the family members with symptoms subsequently attended church, infecting another church attendee. A cluster of 16 cases of COVID-19 was subsequently identified, including three deaths likely resulting from this one introduction of COVID-19 at these two family gatherings.
In Tennessee and Wisconsin, household transmission of SARS-CoV-2 was studied prospectively. A total of 101 index cases and 191 asymptomatic household contacts were enrolled between April and Sept. 2020; 102 of 191 (53%) had SARS-CoV-2 detected during the 14-day follow-up. Most infections (75%) were identified within 5 days and occurred whether the index case was an adult or child.
Lastly, one adolescent was identified as the source for an outbreak at a family gathering where 15 persons from five households and four states shared a house between 8 and 25 days in July 2020. Six additional members visited the house. The index case had an exposure to COVID-19 and had a negative antigen test 4 days after exposure. She was asymptomatic when tested. She developed nasal congestion 2 days later, the same day she and her family departed for the gathering. A total of 11 household contacts developed confirmed, suspected, or probable COVID-19, and the teen developed symptoms. This report illustrates how easily SARS-CoV-2 is transmitted, and how when implemented, mitigation strategies work because none of the six who only visited the house was infected. It also serves as a reminder that antigen testing is indicated only for use within the first 5-12 days of onset of symptoms. In this case, the adolescent was asymptomatic when tested and had a false-negative test result.
Ponder modes of transportation
How will your patient arrive to their holiday destination? Nonstop travel by car with household members is probably the safest way. However, for many families, buses and trains are the only options, and social distancing may be challenging. Air travel is a must for others. Acquisition of COVID-19 during air travel appears to be low, but not absent based on how air enters and leaves the cabin. The challenge is socially distancing throughout the check in and boarding processes, as well as minimizing contact with common surfaces. There also is loss of social distancing once on board. Ideally, masks should be worn during the flight. Additionally, for those with international destinations, most countries now require a negative polymerase chain reaction COVID-19 test within a specified time frame for entry.
Essentially the safest place for your patients during the holidays is celebrating at home with their household contacts. The risk for disease acquisition increases with travel. You will not have the opportunity to discuss holiday plans with most parents. However, you can encourage them to consider the pros and cons of travel with reminders via telephone, e-mail, and /or social messaging directly from your practices similar to those sent for other medically necessary interventions. As for me, I will be celebrating virtually this year. There is a first time for everything.
For additional information that also is patient friendly, the Centers for Disease Control and Prevention offers information about travel within the United States and international travel.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].