User login
Clinical presentation and imaging of bone and soft-tissue sarcomas
Sarcomas are rare neoplasms arising from connective tissue elements of the body. Approximately 80% arise in soft tissue, while the remainder originate in bone.1 Soft-tissue sarcomas are tumors of the mesenchymal system, and about half develop in the extremities. Bone sarcomas are characterized by their location in bone and sometimes produce osteoid, or immature bone.
The most common types of soft-tissue sarcomas are malignant fibrous histiocytoma (although this term has lost favor with some pathologists) and liposarcoma.
The most common types of bone sarcomas are osteosarcoma (a sarcoma that makes osteoid or bone), Ewing sarcoma (composed of small round blue cells with a characteristic chromosomal translocation), and chondrosarcoma (a sarcoma that makes chondroid tissue, or cartilage).
EPIDEMIOLOGY AND PRIMARY BODY SITES
Approximately 10,600 new cases of soft-tissue sarcoma and 2,570 new cases of bone sarcoma were estimated to have been diagnosed in the United States in 2009.2,3 For perspective, the annual incidence of soft-tissue sarcoma is approximately 5.5% that of breast cancer and approximately 5% that of lung cancer.3
Most sarcomas develop in the lower extremities, although the age groups at greatest risk vary among sarcoma types.4
Soft-tissue sarcomas develop most commonly in the thigh and occur primarily in adults.2
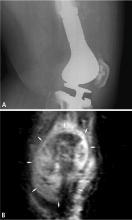
Osteosarcomas are the most common bone sarcoma and develop most frequently in 10- to 20-year-olds; their most common location is the distal femur.5–8 Metastatic osteosarcoma is found in approximately 20% of patients at the time of osteosarcoma diagnosis. Osteosarcomas mainly spread hematogenously, and the lungs are the most common initial site of metastases, being affected in up to 90% of patients with metastatic disease.9
Ewing sarcomas develop most often in the long bones of the extremities or bones of the pelvis. The large majority of cases develop in patients aged 10 to 15 years. 5–8
Chondrosarcomas represent approximately 20% of all bone sarcomas and primarily affect older adults, with a peak incidence in the sixth decade of life.10
OVERVIEW OF PRESENTATION AND EVALUATION
Presentation is highly variable
The clinical presentation of patients with bone or soft-tissue sarcoma is highly variable. Patients often present with a mass, typically one that is increasing in size. In general, bone sarcomas are painful and soft-tissue sarcomas are not, but there are exceptions to this general rule. Constitutional symptoms are rare in patients with bone or soft-tissue sarcomas, but symptoms such as fever, malaise, and weight loss can be seen, especially in patients with Ewing sarcoma.11
Delayed presentation and diagnosis are common
Particularly when a sarcoma is painless, patients sometimes do not seek medical attention until a suspicious mass becomes quite large. Certain tumors, such as synovial sarcoma, a high-grade soft-tissue sarcoma often seen in young adults, may present as a slowly growing or stable-appearing mass over several years. In one study of 33 children with synovial sarcoma, the mean duration of symptoms was 98 weeks (range, 2–364), the mean patient delay before a doctor was seen was 43 weeks (0–156), the mean doctor delay before a correct diagnosis was made was 50 weeks (0–362), and the mean number of doctors seen before referral was 3 (1–6).12 For nearly half the patients in this study (15), the diagnosis was obtained only after unplanned excision, meaning that the surgeon did not expect a malignancy at the time of biopsy. Because delayed presentation is not uncommon in cases of bone or soft-tissue sarcoma, every patient with a mass with indeterminate imaging findings should be referred to or reviewed by an orthopedic or musculoskeletal oncologist.
Biopsy is gold standard for diagnosis
A comprehensive medical history and physical examination are essential at the initial presentation of patients with masses and/or pain suggestive of bone or soft-tissue sarcoma. Sarcoma simulators such as hematoma, metastatic disease, or infection can sometimes be ruled out by careful clinical examination, laboratory work-up, and appropriate imaging, but the gold standard for diagnosis is a biopsy. Moreover, an index of suspicion is required to rule out primary malignancy in any soft-tissue or bone lesion, and this index of suspicion will allow for referral or appropriate selection of the site for biopsy.
Biopsy considerations, as well as further detail on clinical presentation, are provided in the second and third articles in this supplement, which focus, respectively, on bone sarcoma and soft-tissue sarcoma. The remainder of this article reviews the use of imaging for the evaluation of suspected sarcomas, as imaging findings typically prompt or guide biopsy of a suspicious mass. Choosing the right imaging modality is critical to the diagnosis and management of patients with suspected sarcoma.
CONVENTIONAL IMAGING MODALITIES
Despite their utility for evaluating osseous lesions, radiographs have limited to no value in the evaluation of soft-tissue sarcomas but can demonstrate matrix mineralization and erosion or destruction of adjacent bone.
Angiography. In the past, angiography was frequently used to assess the vascularity of sarcomas preoperatively. Diagnostic angiography has been replaced by conventional MRI and magnetic resonance angiography, but some vascular sarcomas may require presurgical embolization to prevent excessive bleeding during surgery.
Radionuclide bone scans have long been a reliable tool for detecting multifocal or disseminated osseous lesions and remain the mainstay for evaluation of osseous metastases. They also are helpful in identifying skip lesions of osteosarcoma (ie, smaller discrete foci of osteosarcoma occurring in the same bone or on the opposing side of a joint).14 Advantages of this modality include whole-body scanning and low radiation at relatively low cost. Radionuclide bone scans demonstrate areas of bony repair and thus could be negative in purely lytic/destructive processes such as renal cell carcinoma metastases and multiple myeloma.
Chest radiographs are typically obtained in the initial stages of patient evaluation and are helpful in demonstrating large nodules or masses resulting from metastatic disease. In a patient with known sarcoma, a negative or equivocal chest radiograph should be followed by chest CT to definitively assess for metastasis.
CROSS-SECTIONAL IMAGING WITH MRI AND CT
MRI preferred for evaluation of most masses
MRI is the examination of choice in the evaluation of soft-tissue masses in light of its superior contrast resolution and ability to demonstrate subtle changes in soft tissues.
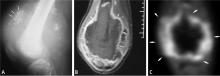
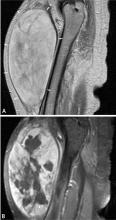
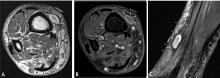
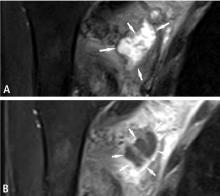
Predicting the histology of most soft-tissue masses is difficult, with the exception of some benign vascular lesions (eg, hemangioma), ganglia, neurogenic lesions, and well-differentiated lipomatous lesions. Aggressive features of a soft-tissue neoplasm include size greater than 5 cm,15 deep location, and absence of central enhancement, which is suggestive of necrosis (Figure 1). Yet one third of soft-tissue sarcomas are either superficial or smaller than 5 cm, which highlights the relative nonspecificity of these features.15
MRI is also the preferred modality in the evaluation of the majority of bone sarcomas, given its ability to accurately define the extent of marrow changes and soft-tissue involvement. MRI should be performed prior to a biopsy to prevent misinterpretation of biopsy-related signal changes in the surrounding tissues, which may negate the value of MRI in sarcoma staging.
Several distinct roles for CT
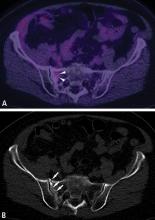
Chest CT should be obtained in all cases of known malignant neoplasms to evaluate for pulmonary nodules, masses, and lymphadenopathy. Despite the recent advances in MRI, CT remains the imaging modality of choice to evaluate the retroperitoneum, abdomen, and pelvis for masses, lymphadenopathy, or other signs of metastatic disease.
Post-treatment monitoring for recurrence
ULTRASONOGRAPHY
Ultrasonography has a limited role in the initial diagnosis and follow-up of musculoskeletal tumors. Its main advantages are a lack of ionizing radiation and dynamic imaging capabilities. Doppler ultrasonography allows direct visualization of tumor vascularity, which may be important for diagnosis and presurgical planning. Unfortunately, bone lesions cannot be evaluated with ultrasonography, owing to the inability of sound waves to penetrate the bony cortex. Poor sound wave penetration may prevent visualization of deep-seated lesions, such as retroperitoneal sarcomas.
Ultrasonography is best used for differentiating solid masses from cystic structures and can provide image guidance in solid tumor biopsy and cyst aspiration. It also may play a role in detecting suspected tumor recurrence in patients in whom artifact from implanted hardware precludes cross-sectional imaging, and it can be reliably used for following up unequivocal soft-tissue masses such as ganglia near joints.
POSITRON EMISSION TOMOGRAPHY
IMAGING-GUIDED INTERVENTIONS
Percutaneous imaging-guided procedures have increasingly replaced open surgical biopsies for bone and soft-tissue tumors. CT guidance is commonly used for percutaneous biopsy, whereas ultrasonographic guidance is sometimes used for superficial soft-tissue lesions. Although the shortest and most direct approach is desirable, this may not be possible in all cases due to the presence of nearby vital structures or the risk of contamination. Seeding of malignant cells along the biopsy tract is a well-known possible complication of image-guided biopsies, and en bloc resection of the needle tract is typically performed at the definitive surgery.
Knowledge of compartmental anatomy is paramount in planning the approach for these biopsies, and consultation with the referring orthopedic surgeon is recommended for optimal management. Expert histopathological interpretation of bone and soft-tissue specimens is essential for the efficacy and high success rates of percutaneous imaging-guided biopsies. Such expertise is integral to the broader interdisciplinary collaboration that is needed to arrive at the most plausible diagnosis, especially in the setting of uncommon or atypical neoplasms.
Currently, MRI-guided interventions are in the initial stage of evolution and could provide valuable guidance for subtle marrow or soft-tissue lesions visible on MRI but not well seen on CT.22 In the future, MRI could play an increasingly important role in imaging-guided procedures because of its lack of ionizing radiation and its ability to demonstrate subtle soft-tissue and bone marrow changes. Imaging-guided therapeutics are growing in their applications in musculoskeletal oncology. CT-guided radiofrequency ablation and cryoablation have been used in the treatment of a variety of tumors23 as well as in the palliation of metastatic bone pain.24
SUMMARY AND CONCLUSION
Bone and soft-tissue sarcomas are rare neoplasms with variable clinical presentations. A high index of suspicion is required for any unexplained mass with indeterminate imaging findings. Recent advances in imaging technology, including cross-sectional MRI and CT, have significantly refined the diagnosis and management of bone and soft-tissue sarcomas. When faced with a possible sarcoma, the clinician’s selection of imaging modalities has a direct impact on diagnosis, staging, and patient management.
- American Cancer Society. Cancer facts & figures 2009. Atlanta, GA: American Cancer Society; 2009.
- Weiss SW, Goldblum JR, Enzinger FM. Enzinger and Weiss’ Soft Tissue Tumors. 5th ed. Philadelphia, PA: Mosby Elsevier; 2008.
- Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin 2009; 59:225–249.
- Simon MA, Springfield DS, eds. Surgery for Bone and Soft-tissue Tumors. Philadelphia, PA: Lippincott-Raven; 1998.
- Dahlin DC, Unni KK. Bone Tumors: General Aspects and Data on 8,542 Cases. 4th ed. Springfield, IL: Thomas; 1986.
- Unni KK. Bone Tumors. New York, NY: Churchill Livingstone; 1988.
- Unni KK. Atlas of Bone Pathology. New York, NY: Chapman & Hall; 1996:1 computer optical disc.
- Unni KK, Dahlin DC. Dahlin’s Bone Tumors: General Aspects and Data on 11,087 Cases. 5th ed. Philadelphia, PA: Lippincott-Raven; 1996.
- Kaste SC, Pratt CB, Cain AM, Jones-Wallace DJ, Rao BN. Metastases detected at the time of diagnosis of primary pediatric extremity osteosarcoma at diagnosis: imaging features. Cancer 1999; 86:1602–1608.
- Healey JH, Lane JM. Chondrosarcoma. Clin Orthop Relat Res 1986; 204:119–129.
- Kissane JM, Askin FB, Foulkes M, Stratton LB, Shirley SF. Ewing’s sarcoma of bone: clinicopathologic aspects of 303 cases from the Intergroup Ewing’s Sarcoma Study. Hum Pathol 1983; 14:773–779.
- Chotel F, Unnithan A, Chandrasekar CR, et al. Variability in the presentation of synovial sarcoma in children: a plea for greater awareness. J Bone Joint Surg Br 2008; 90:1090–1096.
- Miller TT. Bone tumors and tumorlike conditions: analysis with conventional radiography. Radiology 2008; 246:662–674.
- Richardson ML, Gillespy T. Magnetic resonance imaging. In: Kricun ME, ed. Imaging of Bone Tumors. Philadelphia, PA: WB Saunders; 1993:365.
- Fisher C. Soft tissue sarcomas: diagnosis, classification and prognostic factors. Br J Plast Surg 1996; 49:27–33.
- White LM, Wunder JS, Bell RS, et al. Histologic assessment of peritumoral edema in soft tissue sarcoma. Int J Radiat Oncol Biol Phys 2005; 61:1439–1445.
- White LM, Buckwalter KA. Technical considerations: CT and MR imaging in the postoperative orthopedic patient. Semin Musculoskelet Radiol 2002; 6:5–17.
- Blodgett TM, Casagranda B, Townsend DW, Meltzer CC. Issues, controversies, and clinical utility of combined PET/CT imaging: what is the interpreting physician facing? AJR Am J Roentgenol 2005; 184(suppl 5):S138–S145.
- Shin DS, Shon OJ, Han DS, Choi JH, Chun KA, Cho IH. The clinical efficacy of 18F-FDG-PET/CT in benign and malignant musculoskeletal tumors. Ann Nucl Med 2008; 22:603–609.
- Benz MR, Czernin J, Allen-Auerbach MS, et al. FDG-PET/CT imaging predicts histopathologic treatment responses after the initial cycle of neoadjuvant chemotherapy in high-grade soft-tissue sarcomas. Clin Cancer Res 2009; 15:2856–2863.
- Aoki J, Watanabe H, Shinozaki T, et al. FDG-PET for preoperative differential diagnosis between benign and malignant soft tissue masses. Skeletal Radiol 2003; 32:133–138.
- Blanco Sequeiros R, Klemola R, Ojala R, et al. MRI-guided trephine biopsy and fine-needle aspiration in the diagnosis of bone lesions in low-field (0.23 T) MRI system using optical instrument tracking. Eur Radiol 2002; 12:830–835.
- Rosenthal DI. Radiofrequency treatment. Orthop Clin North Am 2006; 37:475–484.
- Callstrom MR, Charboneau JW. Image-guided palliation of painful metastases using percutaneous ablation. Tech Vasc Interv Radiol 2007; 10:120–131.
Sarcomas are rare neoplasms arising from connective tissue elements of the body. Approximately 80% arise in soft tissue, while the remainder originate in bone.1 Soft-tissue sarcomas are tumors of the mesenchymal system, and about half develop in the extremities. Bone sarcomas are characterized by their location in bone and sometimes produce osteoid, or immature bone.
The most common types of soft-tissue sarcomas are malignant fibrous histiocytoma (although this term has lost favor with some pathologists) and liposarcoma.
The most common types of bone sarcomas are osteosarcoma (a sarcoma that makes osteoid or bone), Ewing sarcoma (composed of small round blue cells with a characteristic chromosomal translocation), and chondrosarcoma (a sarcoma that makes chondroid tissue, or cartilage).
EPIDEMIOLOGY AND PRIMARY BODY SITES
Approximately 10,600 new cases of soft-tissue sarcoma and 2,570 new cases of bone sarcoma were estimated to have been diagnosed in the United States in 2009.2,3 For perspective, the annual incidence of soft-tissue sarcoma is approximately 5.5% that of breast cancer and approximately 5% that of lung cancer.3
Most sarcomas develop in the lower extremities, although the age groups at greatest risk vary among sarcoma types.4
Soft-tissue sarcomas develop most commonly in the thigh and occur primarily in adults.2
Osteosarcomas are the most common bone sarcoma and develop most frequently in 10- to 20-year-olds; their most common location is the distal femur.5–8 Metastatic osteosarcoma is found in approximately 20% of patients at the time of osteosarcoma diagnosis. Osteosarcomas mainly spread hematogenously, and the lungs are the most common initial site of metastases, being affected in up to 90% of patients with metastatic disease.9
Ewing sarcomas develop most often in the long bones of the extremities or bones of the pelvis. The large majority of cases develop in patients aged 10 to 15 years. 5–8
Chondrosarcomas represent approximately 20% of all bone sarcomas and primarily affect older adults, with a peak incidence in the sixth decade of life.10
OVERVIEW OF PRESENTATION AND EVALUATION
Presentation is highly variable
The clinical presentation of patients with bone or soft-tissue sarcoma is highly variable. Patients often present with a mass, typically one that is increasing in size. In general, bone sarcomas are painful and soft-tissue sarcomas are not, but there are exceptions to this general rule. Constitutional symptoms are rare in patients with bone or soft-tissue sarcomas, but symptoms such as fever, malaise, and weight loss can be seen, especially in patients with Ewing sarcoma.11
Delayed presentation and diagnosis are common
Particularly when a sarcoma is painless, patients sometimes do not seek medical attention until a suspicious mass becomes quite large. Certain tumors, such as synovial sarcoma, a high-grade soft-tissue sarcoma often seen in young adults, may present as a slowly growing or stable-appearing mass over several years. In one study of 33 children with synovial sarcoma, the mean duration of symptoms was 98 weeks (range, 2–364), the mean patient delay before a doctor was seen was 43 weeks (0–156), the mean doctor delay before a correct diagnosis was made was 50 weeks (0–362), and the mean number of doctors seen before referral was 3 (1–6).12 For nearly half the patients in this study (15), the diagnosis was obtained only after unplanned excision, meaning that the surgeon did not expect a malignancy at the time of biopsy. Because delayed presentation is not uncommon in cases of bone or soft-tissue sarcoma, every patient with a mass with indeterminate imaging findings should be referred to or reviewed by an orthopedic or musculoskeletal oncologist.
Biopsy is gold standard for diagnosis
A comprehensive medical history and physical examination are essential at the initial presentation of patients with masses and/or pain suggestive of bone or soft-tissue sarcoma. Sarcoma simulators such as hematoma, metastatic disease, or infection can sometimes be ruled out by careful clinical examination, laboratory work-up, and appropriate imaging, but the gold standard for diagnosis is a biopsy. Moreover, an index of suspicion is required to rule out primary malignancy in any soft-tissue or bone lesion, and this index of suspicion will allow for referral or appropriate selection of the site for biopsy.
Biopsy considerations, as well as further detail on clinical presentation, are provided in the second and third articles in this supplement, which focus, respectively, on bone sarcoma and soft-tissue sarcoma. The remainder of this article reviews the use of imaging for the evaluation of suspected sarcomas, as imaging findings typically prompt or guide biopsy of a suspicious mass. Choosing the right imaging modality is critical to the diagnosis and management of patients with suspected sarcoma.
CONVENTIONAL IMAGING MODALITIES
Despite their utility for evaluating osseous lesions, radiographs have limited to no value in the evaluation of soft-tissue sarcomas but can demonstrate matrix mineralization and erosion or destruction of adjacent bone.
Angiography. In the past, angiography was frequently used to assess the vascularity of sarcomas preoperatively. Diagnostic angiography has been replaced by conventional MRI and magnetic resonance angiography, but some vascular sarcomas may require presurgical embolization to prevent excessive bleeding during surgery.
Radionuclide bone scans have long been a reliable tool for detecting multifocal or disseminated osseous lesions and remain the mainstay for evaluation of osseous metastases. They also are helpful in identifying skip lesions of osteosarcoma (ie, smaller discrete foci of osteosarcoma occurring in the same bone or on the opposing side of a joint).14 Advantages of this modality include whole-body scanning and low radiation at relatively low cost. Radionuclide bone scans demonstrate areas of bony repair and thus could be negative in purely lytic/destructive processes such as renal cell carcinoma metastases and multiple myeloma.
Chest radiographs are typically obtained in the initial stages of patient evaluation and are helpful in demonstrating large nodules or masses resulting from metastatic disease. In a patient with known sarcoma, a negative or equivocal chest radiograph should be followed by chest CT to definitively assess for metastasis.
CROSS-SECTIONAL IMAGING WITH MRI AND CT
MRI preferred for evaluation of most masses
MRI is the examination of choice in the evaluation of soft-tissue masses in light of its superior contrast resolution and ability to demonstrate subtle changes in soft tissues.
Predicting the histology of most soft-tissue masses is difficult, with the exception of some benign vascular lesions (eg, hemangioma), ganglia, neurogenic lesions, and well-differentiated lipomatous lesions. Aggressive features of a soft-tissue neoplasm include size greater than 5 cm,15 deep location, and absence of central enhancement, which is suggestive of necrosis (Figure 1). Yet one third of soft-tissue sarcomas are either superficial or smaller than 5 cm, which highlights the relative nonspecificity of these features.15
MRI is also the preferred modality in the evaluation of the majority of bone sarcomas, given its ability to accurately define the extent of marrow changes and soft-tissue involvement. MRI should be performed prior to a biopsy to prevent misinterpretation of biopsy-related signal changes in the surrounding tissues, which may negate the value of MRI in sarcoma staging.
Several distinct roles for CT
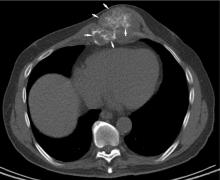
Chest CT should be obtained in all cases of known malignant neoplasms to evaluate for pulmonary nodules, masses, and lymphadenopathy. Despite the recent advances in MRI, CT remains the imaging modality of choice to evaluate the retroperitoneum, abdomen, and pelvis for masses, lymphadenopathy, or other signs of metastatic disease.
Post-treatment monitoring for recurrence
ULTRASONOGRAPHY
Ultrasonography has a limited role in the initial diagnosis and follow-up of musculoskeletal tumors. Its main advantages are a lack of ionizing radiation and dynamic imaging capabilities. Doppler ultrasonography allows direct visualization of tumor vascularity, which may be important for diagnosis and presurgical planning. Unfortunately, bone lesions cannot be evaluated with ultrasonography, owing to the inability of sound waves to penetrate the bony cortex. Poor sound wave penetration may prevent visualization of deep-seated lesions, such as retroperitoneal sarcomas.
Ultrasonography is best used for differentiating solid masses from cystic structures and can provide image guidance in solid tumor biopsy and cyst aspiration. It also may play a role in detecting suspected tumor recurrence in patients in whom artifact from implanted hardware precludes cross-sectional imaging, and it can be reliably used for following up unequivocal soft-tissue masses such as ganglia near joints.
POSITRON EMISSION TOMOGRAPHY
IMAGING-GUIDED INTERVENTIONS
Percutaneous imaging-guided procedures have increasingly replaced open surgical biopsies for bone and soft-tissue tumors. CT guidance is commonly used for percutaneous biopsy, whereas ultrasonographic guidance is sometimes used for superficial soft-tissue lesions. Although the shortest and most direct approach is desirable, this may not be possible in all cases due to the presence of nearby vital structures or the risk of contamination. Seeding of malignant cells along the biopsy tract is a well-known possible complication of image-guided biopsies, and en bloc resection of the needle tract is typically performed at the definitive surgery.
Knowledge of compartmental anatomy is paramount in planning the approach for these biopsies, and consultation with the referring orthopedic surgeon is recommended for optimal management. Expert histopathological interpretation of bone and soft-tissue specimens is essential for the efficacy and high success rates of percutaneous imaging-guided biopsies. Such expertise is integral to the broader interdisciplinary collaboration that is needed to arrive at the most plausible diagnosis, especially in the setting of uncommon or atypical neoplasms.
Currently, MRI-guided interventions are in the initial stage of evolution and could provide valuable guidance for subtle marrow or soft-tissue lesions visible on MRI but not well seen on CT.22 In the future, MRI could play an increasingly important role in imaging-guided procedures because of its lack of ionizing radiation and its ability to demonstrate subtle soft-tissue and bone marrow changes. Imaging-guided therapeutics are growing in their applications in musculoskeletal oncology. CT-guided radiofrequency ablation and cryoablation have been used in the treatment of a variety of tumors23 as well as in the palliation of metastatic bone pain.24
SUMMARY AND CONCLUSION
Bone and soft-tissue sarcomas are rare neoplasms with variable clinical presentations. A high index of suspicion is required for any unexplained mass with indeterminate imaging findings. Recent advances in imaging technology, including cross-sectional MRI and CT, have significantly refined the diagnosis and management of bone and soft-tissue sarcomas. When faced with a possible sarcoma, the clinician’s selection of imaging modalities has a direct impact on diagnosis, staging, and patient management.
Sarcomas are rare neoplasms arising from connective tissue elements of the body. Approximately 80% arise in soft tissue, while the remainder originate in bone.1 Soft-tissue sarcomas are tumors of the mesenchymal system, and about half develop in the extremities. Bone sarcomas are characterized by their location in bone and sometimes produce osteoid, or immature bone.
The most common types of soft-tissue sarcomas are malignant fibrous histiocytoma (although this term has lost favor with some pathologists) and liposarcoma.
The most common types of bone sarcomas are osteosarcoma (a sarcoma that makes osteoid or bone), Ewing sarcoma (composed of small round blue cells with a characteristic chromosomal translocation), and chondrosarcoma (a sarcoma that makes chondroid tissue, or cartilage).
EPIDEMIOLOGY AND PRIMARY BODY SITES
Approximately 10,600 new cases of soft-tissue sarcoma and 2,570 new cases of bone sarcoma were estimated to have been diagnosed in the United States in 2009.2,3 For perspective, the annual incidence of soft-tissue sarcoma is approximately 5.5% that of breast cancer and approximately 5% that of lung cancer.3
Most sarcomas develop in the lower extremities, although the age groups at greatest risk vary among sarcoma types.4
Soft-tissue sarcomas develop most commonly in the thigh and occur primarily in adults.2
Osteosarcomas are the most common bone sarcoma and develop most frequently in 10- to 20-year-olds; their most common location is the distal femur.5–8 Metastatic osteosarcoma is found in approximately 20% of patients at the time of osteosarcoma diagnosis. Osteosarcomas mainly spread hematogenously, and the lungs are the most common initial site of metastases, being affected in up to 90% of patients with metastatic disease.9
Ewing sarcomas develop most often in the long bones of the extremities or bones of the pelvis. The large majority of cases develop in patients aged 10 to 15 years. 5–8
Chondrosarcomas represent approximately 20% of all bone sarcomas and primarily affect older adults, with a peak incidence in the sixth decade of life.10
OVERVIEW OF PRESENTATION AND EVALUATION
Presentation is highly variable
The clinical presentation of patients with bone or soft-tissue sarcoma is highly variable. Patients often present with a mass, typically one that is increasing in size. In general, bone sarcomas are painful and soft-tissue sarcomas are not, but there are exceptions to this general rule. Constitutional symptoms are rare in patients with bone or soft-tissue sarcomas, but symptoms such as fever, malaise, and weight loss can be seen, especially in patients with Ewing sarcoma.11
Delayed presentation and diagnosis are common
Particularly when a sarcoma is painless, patients sometimes do not seek medical attention until a suspicious mass becomes quite large. Certain tumors, such as synovial sarcoma, a high-grade soft-tissue sarcoma often seen in young adults, may present as a slowly growing or stable-appearing mass over several years. In one study of 33 children with synovial sarcoma, the mean duration of symptoms was 98 weeks (range, 2–364), the mean patient delay before a doctor was seen was 43 weeks (0–156), the mean doctor delay before a correct diagnosis was made was 50 weeks (0–362), and the mean number of doctors seen before referral was 3 (1–6).12 For nearly half the patients in this study (15), the diagnosis was obtained only after unplanned excision, meaning that the surgeon did not expect a malignancy at the time of biopsy. Because delayed presentation is not uncommon in cases of bone or soft-tissue sarcoma, every patient with a mass with indeterminate imaging findings should be referred to or reviewed by an orthopedic or musculoskeletal oncologist.
Biopsy is gold standard for diagnosis
A comprehensive medical history and physical examination are essential at the initial presentation of patients with masses and/or pain suggestive of bone or soft-tissue sarcoma. Sarcoma simulators such as hematoma, metastatic disease, or infection can sometimes be ruled out by careful clinical examination, laboratory work-up, and appropriate imaging, but the gold standard for diagnosis is a biopsy. Moreover, an index of suspicion is required to rule out primary malignancy in any soft-tissue or bone lesion, and this index of suspicion will allow for referral or appropriate selection of the site for biopsy.
Biopsy considerations, as well as further detail on clinical presentation, are provided in the second and third articles in this supplement, which focus, respectively, on bone sarcoma and soft-tissue sarcoma. The remainder of this article reviews the use of imaging for the evaluation of suspected sarcomas, as imaging findings typically prompt or guide biopsy of a suspicious mass. Choosing the right imaging modality is critical to the diagnosis and management of patients with suspected sarcoma.
CONVENTIONAL IMAGING MODALITIES
Despite their utility for evaluating osseous lesions, radiographs have limited to no value in the evaluation of soft-tissue sarcomas but can demonstrate matrix mineralization and erosion or destruction of adjacent bone.
Angiography. In the past, angiography was frequently used to assess the vascularity of sarcomas preoperatively. Diagnostic angiography has been replaced by conventional MRI and magnetic resonance angiography, but some vascular sarcomas may require presurgical embolization to prevent excessive bleeding during surgery.
Radionuclide bone scans have long been a reliable tool for detecting multifocal or disseminated osseous lesions and remain the mainstay for evaluation of osseous metastases. They also are helpful in identifying skip lesions of osteosarcoma (ie, smaller discrete foci of osteosarcoma occurring in the same bone or on the opposing side of a joint).14 Advantages of this modality include whole-body scanning and low radiation at relatively low cost. Radionuclide bone scans demonstrate areas of bony repair and thus could be negative in purely lytic/destructive processes such as renal cell carcinoma metastases and multiple myeloma.
Chest radiographs are typically obtained in the initial stages of patient evaluation and are helpful in demonstrating large nodules or masses resulting from metastatic disease. In a patient with known sarcoma, a negative or equivocal chest radiograph should be followed by chest CT to definitively assess for metastasis.
CROSS-SECTIONAL IMAGING WITH MRI AND CT
MRI preferred for evaluation of most masses
MRI is the examination of choice in the evaluation of soft-tissue masses in light of its superior contrast resolution and ability to demonstrate subtle changes in soft tissues.
Predicting the histology of most soft-tissue masses is difficult, with the exception of some benign vascular lesions (eg, hemangioma), ganglia, neurogenic lesions, and well-differentiated lipomatous lesions. Aggressive features of a soft-tissue neoplasm include size greater than 5 cm,15 deep location, and absence of central enhancement, which is suggestive of necrosis (Figure 1). Yet one third of soft-tissue sarcomas are either superficial or smaller than 5 cm, which highlights the relative nonspecificity of these features.15
MRI is also the preferred modality in the evaluation of the majority of bone sarcomas, given its ability to accurately define the extent of marrow changes and soft-tissue involvement. MRI should be performed prior to a biopsy to prevent misinterpretation of biopsy-related signal changes in the surrounding tissues, which may negate the value of MRI in sarcoma staging.
Several distinct roles for CT
Chest CT should be obtained in all cases of known malignant neoplasms to evaluate for pulmonary nodules, masses, and lymphadenopathy. Despite the recent advances in MRI, CT remains the imaging modality of choice to evaluate the retroperitoneum, abdomen, and pelvis for masses, lymphadenopathy, or other signs of metastatic disease.
Post-treatment monitoring for recurrence
ULTRASONOGRAPHY
Ultrasonography has a limited role in the initial diagnosis and follow-up of musculoskeletal tumors. Its main advantages are a lack of ionizing radiation and dynamic imaging capabilities. Doppler ultrasonography allows direct visualization of tumor vascularity, which may be important for diagnosis and presurgical planning. Unfortunately, bone lesions cannot be evaluated with ultrasonography, owing to the inability of sound waves to penetrate the bony cortex. Poor sound wave penetration may prevent visualization of deep-seated lesions, such as retroperitoneal sarcomas.
Ultrasonography is best used for differentiating solid masses from cystic structures and can provide image guidance in solid tumor biopsy and cyst aspiration. It also may play a role in detecting suspected tumor recurrence in patients in whom artifact from implanted hardware precludes cross-sectional imaging, and it can be reliably used for following up unequivocal soft-tissue masses such as ganglia near joints.
POSITRON EMISSION TOMOGRAPHY
IMAGING-GUIDED INTERVENTIONS
Percutaneous imaging-guided procedures have increasingly replaced open surgical biopsies for bone and soft-tissue tumors. CT guidance is commonly used for percutaneous biopsy, whereas ultrasonographic guidance is sometimes used for superficial soft-tissue lesions. Although the shortest and most direct approach is desirable, this may not be possible in all cases due to the presence of nearby vital structures or the risk of contamination. Seeding of malignant cells along the biopsy tract is a well-known possible complication of image-guided biopsies, and en bloc resection of the needle tract is typically performed at the definitive surgery.
Knowledge of compartmental anatomy is paramount in planning the approach for these biopsies, and consultation with the referring orthopedic surgeon is recommended for optimal management. Expert histopathological interpretation of bone and soft-tissue specimens is essential for the efficacy and high success rates of percutaneous imaging-guided biopsies. Such expertise is integral to the broader interdisciplinary collaboration that is needed to arrive at the most plausible diagnosis, especially in the setting of uncommon or atypical neoplasms.
Currently, MRI-guided interventions are in the initial stage of evolution and could provide valuable guidance for subtle marrow or soft-tissue lesions visible on MRI but not well seen on CT.22 In the future, MRI could play an increasingly important role in imaging-guided procedures because of its lack of ionizing radiation and its ability to demonstrate subtle soft-tissue and bone marrow changes. Imaging-guided therapeutics are growing in their applications in musculoskeletal oncology. CT-guided radiofrequency ablation and cryoablation have been used in the treatment of a variety of tumors23 as well as in the palliation of metastatic bone pain.24
SUMMARY AND CONCLUSION
Bone and soft-tissue sarcomas are rare neoplasms with variable clinical presentations. A high index of suspicion is required for any unexplained mass with indeterminate imaging findings. Recent advances in imaging technology, including cross-sectional MRI and CT, have significantly refined the diagnosis and management of bone and soft-tissue sarcomas. When faced with a possible sarcoma, the clinician’s selection of imaging modalities has a direct impact on diagnosis, staging, and patient management.
- American Cancer Society. Cancer facts & figures 2009. Atlanta, GA: American Cancer Society; 2009.
- Weiss SW, Goldblum JR, Enzinger FM. Enzinger and Weiss’ Soft Tissue Tumors. 5th ed. Philadelphia, PA: Mosby Elsevier; 2008.
- Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin 2009; 59:225–249.
- Simon MA, Springfield DS, eds. Surgery for Bone and Soft-tissue Tumors. Philadelphia, PA: Lippincott-Raven; 1998.
- Dahlin DC, Unni KK. Bone Tumors: General Aspects and Data on 8,542 Cases. 4th ed. Springfield, IL: Thomas; 1986.
- Unni KK. Bone Tumors. New York, NY: Churchill Livingstone; 1988.
- Unni KK. Atlas of Bone Pathology. New York, NY: Chapman & Hall; 1996:1 computer optical disc.
- Unni KK, Dahlin DC. Dahlin’s Bone Tumors: General Aspects and Data on 11,087 Cases. 5th ed. Philadelphia, PA: Lippincott-Raven; 1996.
- Kaste SC, Pratt CB, Cain AM, Jones-Wallace DJ, Rao BN. Metastases detected at the time of diagnosis of primary pediatric extremity osteosarcoma at diagnosis: imaging features. Cancer 1999; 86:1602–1608.
- Healey JH, Lane JM. Chondrosarcoma. Clin Orthop Relat Res 1986; 204:119–129.
- Kissane JM, Askin FB, Foulkes M, Stratton LB, Shirley SF. Ewing’s sarcoma of bone: clinicopathologic aspects of 303 cases from the Intergroup Ewing’s Sarcoma Study. Hum Pathol 1983; 14:773–779.
- Chotel F, Unnithan A, Chandrasekar CR, et al. Variability in the presentation of synovial sarcoma in children: a plea for greater awareness. J Bone Joint Surg Br 2008; 90:1090–1096.
- Miller TT. Bone tumors and tumorlike conditions: analysis with conventional radiography. Radiology 2008; 246:662–674.
- Richardson ML, Gillespy T. Magnetic resonance imaging. In: Kricun ME, ed. Imaging of Bone Tumors. Philadelphia, PA: WB Saunders; 1993:365.
- Fisher C. Soft tissue sarcomas: diagnosis, classification and prognostic factors. Br J Plast Surg 1996; 49:27–33.
- White LM, Wunder JS, Bell RS, et al. Histologic assessment of peritumoral edema in soft tissue sarcoma. Int J Radiat Oncol Biol Phys 2005; 61:1439–1445.
- White LM, Buckwalter KA. Technical considerations: CT and MR imaging in the postoperative orthopedic patient. Semin Musculoskelet Radiol 2002; 6:5–17.
- Blodgett TM, Casagranda B, Townsend DW, Meltzer CC. Issues, controversies, and clinical utility of combined PET/CT imaging: what is the interpreting physician facing? AJR Am J Roentgenol 2005; 184(suppl 5):S138–S145.
- Shin DS, Shon OJ, Han DS, Choi JH, Chun KA, Cho IH. The clinical efficacy of 18F-FDG-PET/CT in benign and malignant musculoskeletal tumors. Ann Nucl Med 2008; 22:603–609.
- Benz MR, Czernin J, Allen-Auerbach MS, et al. FDG-PET/CT imaging predicts histopathologic treatment responses after the initial cycle of neoadjuvant chemotherapy in high-grade soft-tissue sarcomas. Clin Cancer Res 2009; 15:2856–2863.
- Aoki J, Watanabe H, Shinozaki T, et al. FDG-PET for preoperative differential diagnosis between benign and malignant soft tissue masses. Skeletal Radiol 2003; 32:133–138.
- Blanco Sequeiros R, Klemola R, Ojala R, et al. MRI-guided trephine biopsy and fine-needle aspiration in the diagnosis of bone lesions in low-field (0.23 T) MRI system using optical instrument tracking. Eur Radiol 2002; 12:830–835.
- Rosenthal DI. Radiofrequency treatment. Orthop Clin North Am 2006; 37:475–484.
- Callstrom MR, Charboneau JW. Image-guided palliation of painful metastases using percutaneous ablation. Tech Vasc Interv Radiol 2007; 10:120–131.
- American Cancer Society. Cancer facts & figures 2009. Atlanta, GA: American Cancer Society; 2009.
- Weiss SW, Goldblum JR, Enzinger FM. Enzinger and Weiss’ Soft Tissue Tumors. 5th ed. Philadelphia, PA: Mosby Elsevier; 2008.
- Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin 2009; 59:225–249.
- Simon MA, Springfield DS, eds. Surgery for Bone and Soft-tissue Tumors. Philadelphia, PA: Lippincott-Raven; 1998.
- Dahlin DC, Unni KK. Bone Tumors: General Aspects and Data on 8,542 Cases. 4th ed. Springfield, IL: Thomas; 1986.
- Unni KK. Bone Tumors. New York, NY: Churchill Livingstone; 1988.
- Unni KK. Atlas of Bone Pathology. New York, NY: Chapman & Hall; 1996:1 computer optical disc.
- Unni KK, Dahlin DC. Dahlin’s Bone Tumors: General Aspects and Data on 11,087 Cases. 5th ed. Philadelphia, PA: Lippincott-Raven; 1996.
- Kaste SC, Pratt CB, Cain AM, Jones-Wallace DJ, Rao BN. Metastases detected at the time of diagnosis of primary pediatric extremity osteosarcoma at diagnosis: imaging features. Cancer 1999; 86:1602–1608.
- Healey JH, Lane JM. Chondrosarcoma. Clin Orthop Relat Res 1986; 204:119–129.
- Kissane JM, Askin FB, Foulkes M, Stratton LB, Shirley SF. Ewing’s sarcoma of bone: clinicopathologic aspects of 303 cases from the Intergroup Ewing’s Sarcoma Study. Hum Pathol 1983; 14:773–779.
- Chotel F, Unnithan A, Chandrasekar CR, et al. Variability in the presentation of synovial sarcoma in children: a plea for greater awareness. J Bone Joint Surg Br 2008; 90:1090–1096.
- Miller TT. Bone tumors and tumorlike conditions: analysis with conventional radiography. Radiology 2008; 246:662–674.
- Richardson ML, Gillespy T. Magnetic resonance imaging. In: Kricun ME, ed. Imaging of Bone Tumors. Philadelphia, PA: WB Saunders; 1993:365.
- Fisher C. Soft tissue sarcomas: diagnosis, classification and prognostic factors. Br J Plast Surg 1996; 49:27–33.
- White LM, Wunder JS, Bell RS, et al. Histologic assessment of peritumoral edema in soft tissue sarcoma. Int J Radiat Oncol Biol Phys 2005; 61:1439–1445.
- White LM, Buckwalter KA. Technical considerations: CT and MR imaging in the postoperative orthopedic patient. Semin Musculoskelet Radiol 2002; 6:5–17.
- Blodgett TM, Casagranda B, Townsend DW, Meltzer CC. Issues, controversies, and clinical utility of combined PET/CT imaging: what is the interpreting physician facing? AJR Am J Roentgenol 2005; 184(suppl 5):S138–S145.
- Shin DS, Shon OJ, Han DS, Choi JH, Chun KA, Cho IH. The clinical efficacy of 18F-FDG-PET/CT in benign and malignant musculoskeletal tumors. Ann Nucl Med 2008; 22:603–609.
- Benz MR, Czernin J, Allen-Auerbach MS, et al. FDG-PET/CT imaging predicts histopathologic treatment responses after the initial cycle of neoadjuvant chemotherapy in high-grade soft-tissue sarcomas. Clin Cancer Res 2009; 15:2856–2863.
- Aoki J, Watanabe H, Shinozaki T, et al. FDG-PET for preoperative differential diagnosis between benign and malignant soft tissue masses. Skeletal Radiol 2003; 32:133–138.
- Blanco Sequeiros R, Klemola R, Ojala R, et al. MRI-guided trephine biopsy and fine-needle aspiration in the diagnosis of bone lesions in low-field (0.23 T) MRI system using optical instrument tracking. Eur Radiol 2002; 12:830–835.
- Rosenthal DI. Radiofrequency treatment. Orthop Clin North Am 2006; 37:475–484.
- Callstrom MR, Charboneau JW. Image-guided palliation of painful metastases using percutaneous ablation. Tech Vasc Interv Radiol 2007; 10:120–131.
Identifying serious causes of back pain: Cancer, infection, fracture
Back pain is one of the most common complaints that internists and primary care physicians encounter.1 Although back pain is nonspecific, some hallmark signs and symptoms indicate that a patient is more likely to have a serious disorder. This article contrasts the presentation of cancer, infections, and fractures with the more common and benign conditions that cause back pain and provides guidance for diagnosis.
UNCOMMON, BUT MUST BE CONSIDERED
Although a variety of tissues can contribute to pain—intervertebral disks, vertebrae, ligaments, neural structures, muscles, and fascia—and many disorders can damage these tissues, most patients with back or neck pain have a benign condition. Back pain is typically caused by age-related degenerative changes or by minor repetitive trauma; with supportive care and physical therapy, up to 90% of patients with back pain of this nature improve substantially within 4 weeks.2
Serious, destructive diseases are uncommon causes of back pain: malignancy, infection, ankylosing spondylitis, and epidural abscess together account for fewer than 1% of cases of back pain in a typical primary care practice. But their clinical impact is out of proportion to their prevalence. The fear of overlooking a serious condition influences any practitioner’s approach to back pain and is a common reason for ordering multiple imaging studies and consultations.3 Therefore, the time, effort, and resources invested in ruling out these disorders is considerable.
Whether a patient with back pain has an ominous disease can usually be determined with a careful history, physical examination, and appropriate diagnostic studies. Once a serious diagnosis is ruled out, attention can be focused on rehabilitation and back care.
Back pain can also be due to musculoskeletal disorders, peptic ulcers, pancreatitis, pyelonephritis, aortic aneurysms, and other serious conditions, which we have discussed in other articles in this journal.4–6
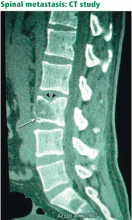
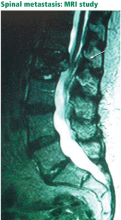
SPINAL CANCER AND METASTASES
Since back pain is the presenting symptom in 90% of patients with spinal tumors,7 neoplasia belongs in the differential diagnosis of any patient with persistent, unremitting back pain. However, it is also important to recognize atypical presentations of neoplasia, such as a painless neurologic deficit, which should prompt an urgent workup.
The spine is one of the most common sites of metastasis: about 20,000 cases arise each year.8 Brihaye et al9 reviewed 1,477 cases of spinal metastases with epidural involvement and found that 16.5% arose from primary tumors in the breast, 15.6% from the lung, 9.2% from the prostate, and 6.5% from the kidney.
Cancer pain is persistent and progressive
Benign back pain often arises from a known injury, is relieved by rest, and increases with activities that load the disk (eg, sitting, getting up from bed or a chair), lumbar flexion with or without rotation, lifting, vibration (eg, riding in a car), coughing, sneezing, laughing, and the Valsalva maneuver. It is most commonly focal to the lumbosacral junction, the lumbar muscles, and the buttocks. Pain due to injury or a flare-up of degenerative disease typically begins to subside after 4 to 6 weeks and responds to nonsteroidal anti-inflammatory drugs and physical therapy.10
In contrast, pain caused by spinal neoplasia is typically persistent and progressive and is not alleviated by rest. Often the pain is worse at night, waking the patient from sleep. Back pain is typically focal to the level of the lesion and may be associated with belt-like thoracic pain or radicular symptoms of pain or weakness in the legs. A spinal mass can cause neurologic signs or symptoms by directly compressing the spinal cord or nerve roots, mimicking disk herniation or stenosis.11,12
Pathologic fractures resulting from vertebral destruction may be the first—and unfortunately a late—presentation of a tumor.
Ask about, look for, signs and symptoms of cancer
In taking the history, one should ask about possible signs and symptoms of systemic disease such as fatigue, weight loss, and changes in bowel habits. Hemoptysis, lymphadenopathy, subcutaneous or breast masses, nipple discharge, atypical vaginal bleeding, or blood in the stool suggest malignancy and should direct the specific diagnostic approach.13 A history of cancer, even if remote, should raise suspicion, as should major risk factors such as smoking.
Because most spinal tumors are metastases, a clinical examination of the breast, lungs, abdomen, thyroid, and prostate are appropriate starting points.14 The spine should be examined to identify sites of focal pain. A neurologic examination should be done to evaluate any signs of neurologic compromise or abnormal reflexes. Signs or symptoms of spinal cord compression should be investigated immediately.
Cancer usually elevates the ESR, CRP
If cancer is suspected, initial tests should include a complete blood cell count, erythrocyte sedimentation rate, C-reactive protein level, urinalysis, prostate-specific antigen level, and fecal occult blood testing. Normal results can considerably relieve suspicion of cancer: the erythrocyte sedimentation rate and C-reactive protein level are almost always elevated with systemic neoplasia.
Imaging tests
Unfortunately, spinal tumors cannot be well visualized on radiographs until significant destruction has occurred.15
A bone scan can usually detect tumors other than the purely lytic ones such as myeloma and has a sensitivity of 74%, a specificity of 81%, and a positive predictive value of 64% for vertebral metastasis in patients with back pain.16
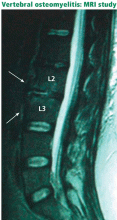
INFECTION CAN BE INDOLENT OR ACUTE
Spinal infection is a serious condition that can take an indolent, smoldering course or, alternatively, can erupt into sepsis or rapidly progressive vertebral destruction. Although the latter conditions are hard to miss, early diskitis and osteomyelitis can be difficult to differentiate from idiopathic back pain. In a series of 101 patients with vertebral osteomyelitis, misdiagnosis occurred in 33.7%, and the average delay from the onset of clinical manifestations to diagnosis was 2.6 months.20 Tuberculosis can be even more elusive: in a series of 78 patients diagnosed with definite or probable tuberculous vertebral osteomyelitis, the mean delay to diagnosis was about 6 months.21
Acute spinal infections are most often pyogenic; chronic infections may be pyogenic, fungal, or granulomatous.
Vertebral osteomyelitis accounts for 2% to 7% of all cases of osteomyelitis and is an uncommon cause of back pain.22 Any source of infection (eg, dental abscess, pneumonia) can seed the spine; urinary tract infection is the most common. Patients with immunocompromise or diabetes are most at risk.23 The onset is usually insidious with focal back pain at the level of involvement.
History and physical examination reveal localized pain
Spinal infections typically cause pain that is worsened with weight-bearing and activity and is relieved only when lying down. Chronic infection is usually associated with weight loss, fatigue, fevers, and night sweats.
Pain is usually well localized and reproduced by palpation or percussion over the involved level. Severe pain can sometimes be elicited by sitting the patient up or by changing the patient’s position. Focal kyphosis may be detectable if the vertebra has collapsed.
In a series of 41 patients with pyogenic infectious spondylitis, 90% had localized back pain aggravated by percussion, 59% had radicular signs and symptoms, and 29% had neurologic signs of spinal cord compression, including hyperreflexia, clonus, the Babinski sign (extension of the toes upward when the sole of the foot is stroked upwards), or the Hoffmann sign (flexion of the thumb elicited by flicking the end of a middle finger).24
LABORATORY RESULTS TYPICALLY INDICATE INFECTION
The erythrocyte sedimentation rate is the most sensitive test for infection, and an elevated rate may be the only abnormal laboratory finding: Digby and Kersley25 found that the rate was increased in all of 30 patients with nontuberculous pyogenic osteomyelitis of the spine. The C-reactive protein level is also usually elevated, but 40% of patients have a normal white blood cell count.25 Results of other laboratory tests are typically in the normal range. Tuberculin skin testing should be done for patients at high risk of the disease (eg, immigrants from areas of endemic disease, non-Hispanic blacks, immunocompromised patients, and those with known exposure to tuberculosis). Patients with high fever, chills, or rigors should have cultures taken of blood, urine, and sputum and from any intravenous lines.
Imaging changes may not appear for months
CT, on the other hand, may be better for showing the extent of bone involvement. In cases of vertebral osteomyelitis and intervertebral disk space infection, simultaneous involvement of the adjacent vertebral end plates and the intervertebral disk are the major findings.30
Signs of infection using T1-weighted MRI include low-signal marrow or disk spaces within the vertebral body, loss of definition of end plates (which appear hypointense compared with the bone marrow), and destruction of the cortical margins of the involved vertebral bodies. T2-weighted MRI typically discloses high signals of the affected areas of the vertebral body and disk. Contrast should be used to increase specificity; enhancement may be the first sign of an acute inflammatory process.31
CT and MRI can help identify sequestra, perilesional sclerosis, and epidural or soft tissue abscesses. Guided biopsy may be needed to differentiate between abscess, hematoma, tumor, and inflammation.
MRI findings: Pyogenic vs tuberculous spondylitis
MRI can help differentiate pyogenic vertebral osteomyelitis from tubercular disease, although findings may be similar (eg, both conditions have a high signal on T2-weighted images).32 Jung et al,33 in a retrospective study of 52 patients with spondylitis, found that compared with patients with pyogenic infections, patients with tuberculous spondylitis had a significantly higher incidence of a well-defined paraspinal abnormal signal on MRI, a thin and smooth abscess wall, a paraspinal or intraosseous abscess, subligamentous spread to three or more vertebral levels, involvement of multiple vertebral bodies, thoracic spine involvement, and a hyperintense signal on T2-weighted images. Other MRI features characteristically seen in patients with tuberculous spinal disease are anterior corner destruction, a relative preservation of the intervertebral disk, and large soft-tissue abscesses with calcifications.34
Prompt diagnosis and aggressive treatment needed
Pigrau et al35 found that spinal osteomyelitis is highly associated with endocarditis: among 606 patients with infectious endocarditis, 28 (4.6%) had pyogenic vertebral osteomyelitis, and among 91 patients with pyogenic vertebral osteomyelitis, 28 (30.8%) had infectious endocarditis.
McHenry et al36 retrospectively studied outcomes of 253 patients with vertebral osteomyelitis after a median of 6.5 years (range 2 days to 38 years): 11% died, more than one-third of survivors had residual disability, and 14% had a relapse. Surgery resulted in recovery or improvement in 86 (79%) of 109 patients. Independent risk factors for adverse outcome (death or incomplete recovery) were neurologic compromise, increased time to diagnosis, and having a hospital-acquired infection (P = .004). Relapse commonly developed in patients with severe vertebral destruction and abscesses, which appeared some time after surgical drainage or debridement. Recurrent bacteremia, paravertebral abscesses, and chronically draining sinuses were independently associated with relapse (P = .001). MRI, done in 110 patients, was often performed late in the course of infection and did not significantly affect outcome. The authors stressed that an optimal outcome of vertebral osteomyelitis requires heightened awareness, early diagnosis, prompt identification of pathogens, reversal of complications, and prolonged antimicrobial therapy.
Epidural abscess may also be present
Epidural abscess occurs in 10% of spine infections. About half of patients with an epidural abscess are misdiagnosed on their initial evaluation.37,38 Patients initially complain of local spine pain, followed by radicular pain, weakness, and finally paralysis. Between 12% and 30% of patients report a history of trauma, even as minor as a fall, preceding the infection.38,39
Radiologic findings are frequently equivocal, and MRI is preferred; gadolinium enhancement further increases sensitivity.39,40 Spinal canal abscesses usually appear hypointense on T1-weighted images and hyperintense on T2-weighted images, with ring enhancement surrounding the abscess area in contrast studies.41 MRI may give negative findings in the early stages of a spinal canal infection and so may need to be repeated.41 MRI may not help distinguish an epidural from a subdural abscess. However, primary spinal epidural abscesses without concomitant vertebral osteomyelitis are rare; therefore, the finding of associated vertebral osteomyelitis makes a spinal epidural abscess more likely.
FRACTURES
Fractures of the spine can be asymptomatic and may have no preceding trauma. They can be due to osteoporosis, malignancy, infection, or metabolic disorders such as renal osteodystrophy or hyperparathyroidism. Fractures in normal bone are almost always associated with trauma. Any suspicion of infection or malignancy should be investigated.
Corticosteroids increase risk
Any patient with back pain who is receiving corticosteroid therapy should be considered as having a compression fracture until proven otherwise.3 De Vries et al42 found that in a database of nearly 200,000 patients receiving glucocorticoids, risk increased substantially with increasing cumulative exposure. Those who intermittently received high doses (= 15 mg/day) and those who had no or little previous exposure to corticosteroids (cumulative exposure = 1 g) had only a slightly increased risk of osteoporotic fracture, and their risk of fracture of the hip and femur was not increased. In contrast, patients who received a daily dose of at least 30 mg and whose cumulative exposure was more than 5 g had a relative risk of osteoporotic vertebral fracture of 14.42 (95% confidence interval 8.29–25.08).
Osteoporotic compression fractures are common in the elderly
Osteoporosis involves reduced bone density, disrupted trabecular architecture, and increased susceptibility to fractures. About 700,000 vertebral body compression fractures occur in the United States each year43: about 10% result in hospitalization, involving an average stay of 8 days.44 Osteoporotic compression fractures are highly associated with age older than 65, female sex, and European descent.45,46 The estimated lifetime risk of a clinically evident vertebral fracture after age 50 years is 16% among postmenopausal white women and 5% among white men.47
A single osteoporotic vertebral compression fracture increases the risk of subsequent fractures by a factor of five, and up to 20% of patients with a vertebral compression fracture are likely to have another one within the same year if osteoporosis remains untreated.48 Population studies suggest that the death rate among patients who have osteoporotic vertebral compression fractures increases with the number of involved vertebrae.43
Unfortunately, osteoporotic vertebral compression fractures are not always easily amenable to treatment: up to 30% of patients who are symptomatic and seek treatment do not respond adequately to nonsurgical methods.49,50 However, new minimally invasive interventions such as vertebral augmentation make timely evaluation clinically relevant.
History, physical examination
Patients may present with a history of trauma with associated back pain or a neurologic deficit. In osteoporotic patients, the trauma may have been minimal, eg, a sneeze, a fall from a chair, or a slip and fall in the home. Pain tends to be worse when standing erect and occasionally when lying flat.
The patient is commonly visibly uncomfortable and may be limited to a wheelchair or stoop forward when standing. The spine may show an absence of the midline crease or an exaggerated thoracic kyphosis. Pain is typically reproduced by deep pressure over the spinous process at the involved level. Compression fractures rarely cause neurologic deficits but should always be considered.
Fractures commonly occur in the thoracolumbar region but may be anywhere in the spine. Fractures in the upper thoracic spine may indicate an underlying malignant tumor, and a thorough search for a possible primary lesion should always be carried out for fractures in this location.
Laboratory testing
Routine laboratory evaluation and thyroid function tests should be done, as well as a 24-hour urine specimen for collagen breakdown products, calcium, phosphate, and creatinine levels. Serum and urine protein electrophoresis should be performed if myeloma is suspected. A white blood-cell count, erythrocyte sedimentation rate, and C-reactive protein level help determine if an underlying infection caused the fracture.
MRI needed if plain films reveal fracture or are equivocal
Anteroposterior and lateral roentgenograms should be taken first; they typically show osteopenia. A fracture in the vertebral body is characterized by loss of height and by wedging. Osseous fragments can occasionally be seen in the spinal canal.
Sagittal short tau inversion recovery sequences, which use specifically timed pulse sequences to suppress fat signals, show high-intensity signal changes in areas of edema from acute or healing fractures. They provide a sensitive but nonspecific marker of abnormality.
Dual energy x-ray absorptiometry helps determine the extent of osteoporosis.
Bone scans should only be used for patients with suspected metastatic disease.
Patients with ankylosing spondylitis need thorough workup
Ankylosing spondylitis predisposes to serious spinal injury. Even after only minor trauma, patients with ankylosing spondylitis and acute, severe back pain should be thoroughly evaluated for fracture with CT and MRI of the entire spine. Plain radiography should not be relied on for these patients because of the risk of misinterpretation, delayed diagnosis, and poorer outcomes.52,53
NEUROLOGIC COMPROMISE—A RED FLAG
Cauda equina compression classically presents with back pain, bilateral sciatica, saddle anesthesia, and lower extremity weakness progressing to paraplegia, but in practice these symptoms are variably present and diagnosing the condition often requires a high degree of suspicion. Hyporeflexia is typically a sign of cauda equina compression, while hyperreflexia, clonus, and the Babinski sign suggest spinal cord compression, requiring an evaluation of the cervical and thoracic spine. Cauda equina compression typically involves urinary retention; in contrast, cord compression typically causes incontinence.55
If either cauda equina or spinal cord compression is detected during an initial examination, an immediate more extensive evaluation is warranted. MRI is the study of choice.
Spinal epidural hematoma
Spinal epidural hematoma is a rare but dramatic cause of paralysis in elderly patients. In most cases, there is no antecedent trauma. Lawton et al,56 in a series of 30 patients treated surgically for spinal epidural hematoma, found that 73% resulted from spine surgery, epidural catheterization, or anticoagulation therapy. Other possible causes of epidural hematoma include vascular malformations, angiomas, aneurysms, hypertension, and aspirin therapy.57
The same study56 found that the time from the first symptom to maximal neurologic deficit ranged from a few minutes to 4 days, with the average interval being nearly 13 hours.
Although painless onset has been reported,58 spinal epidural hematoma typically presents with acute pain at the level of the lesion, which is often rapidly followed by paraplegia or quadriplegia, depending on the location of the hemorrhage. Sometimes the onset of pain is preceded by a sudden increase of venous pressure from coughing, sneezing, or straining at stool. Urinary retention often develops at an early stage.
Most lesions occur in the thoracic region and extend into the cervicothoracic or the thoracolumbar area. The pain distribution may be radicular, mimicking a ruptured intervertebral disk.
Evaluation should be with MRI. Acute hemorrhage is characterized by a marked decrease in signal intensity on T2-weighted images. Subacute hematoma has increased signal intensity on both T1- and T2-weighted images.56
Early recognition, MRI confirmation, and treatment should be accomplished as soon as possible.56 Recovery depends on the severity of the neurologic deficit and the duration of symptoms before treatment. Lawton et al56 found that patients taken to surgery within 12 hours had better neurologic outcomes than patients with identical preoperative neurologic status whose surgery was delayed beyond 12 hours. Surgery should not be withheld because of advanced age or poor health: in 10 reported cases in which surgery was delayed, all patients died.59
- Deyo RA, Tsui-Wu YJ. Descriptive epidemiology of low-back pain and its related medical care in the United States. Spine 1987; 12:264–268.
- Pengel LH, Herbert RD, Maher CG, Refshauge KM. Acute back pain: systematic review of its prognosis. BMJ 2003; 327:323–325.
- Deyo RA, Rainville J, Kent DL. What can the history and physical examination tell us about back pain? JAMA 1992; 268:760–765.
- Pateder DB, Brems J, Lieberman I, Bell GR, McLain RF. Masquerade: nonspinal musculoskeletal disorders that mimic spinal conditions. Cleve Clin J Med 2008; 75:50–56.
- Klineberg E, Mazanec D, Orr D, Demicco R, Bell G, McLain R. Masquerade: medical causes of back pain. Cleve Clin J Med 2007; 74:905–913.
- McLain RF, Bell G, Montgomery W. Masquerade: systemic causes of back pain. Cleve Clin J Med In press.
- Gilbert RW, Kim JH, Posner JB. Epidural spinal cord compression from metastatic tumor: diagnosis and treatment. Ann Neurol 1978; 3:40–51.
- Black P. Spinal metastasis: current status and recommended guidelines for management. Neurosurgery 1979; 5:726–746.
- Brihaye J, Ectors P, Lemort M, Van Houtte P. The management of spinal epidural metastases. Adv Tech Stand Neurosurg 1988; 16:121–176.
- Patel RK, Slipman CW. Lumbar degenerative disk disease. Emedicine. Accessed March 25, 2008. http://www.emedicine.com/pmr/topic67.htm.
- Deyo RA, Diehl AK. Cancer as a cause of back pain: frequency, clinical presentation, and diagnostic strategies. J Gen Intern Med 1988; 3:230–238.
- Rosen P, Barkin RM, Danzl DF, et al. Emergency Medicine: Concepts and Clinical Practice. 4th ed. St Louis, MO: Mosby; 1998:2100–2102.
- Abbruzzese JL, Abbruzzese MC, Lenzi R, Hess KR, Raber MN. Analysis of a diagnostic strategy for patients with suspected tumors of unknown origin. J Clin Oncol 1995; 13:2094–2103.
- McCarthy EF, Frassica FJ. Pathology of Bone and Joint Disorders: With Clinical and Radiographic Correlation. Philadelphia: WB Saunders; 1998.
- Edelstyn GA, Gillespie PJ, Grebbell FS. The radiological demonstration of osseous metastases. Experimental observations. Clin Radiol 1967; 18:158–162.
- Han LJ, Au-Yong TK, Tong WC, Chu KS, Szeto LT, Wong CP. Comparison of bone single-photon emission tomography and planar imaging in the detection of vertebral metastases in patients with back pain. Eur J Nucl Med 1998; 25:635–638.
- Feun LG, Savaraj N. Detection of occult bone metastasis by MRI scan. J Fla Med Assoc 1990; 77:881–883.
- Citrin DL, Bessent RG, Greig WR. A comparison of the sensitivity and accuracy of the 99TCm-phosphate bone scan and skeletal radiograph in the diagnosis of bone metastases. Clin Radiol 1977; 28:107–117.
- Runge VM, Lee C, Iten AL, Williams NM. Contrast-enhanced magnetic resonance imaging in a spinal epidural tumor model. Invest Radiol 1997; 32:589–595.
- Buranapanitkit B, Lim A, Geater A. Misdiagnosis in vertebral osteomyelitis: problems and factors. J Med Assoc Thai 2001; 84:1743–1750.
- Colmenero JD, Jiménez-Mejías ME, Reguera JM, et al. Tuberculous vertebral osteomyelitis in the new millennium: still a diagnostic and therapeutic challenge. Eur J Clin Microbiol Infect Dis 2004; 23:477–483.
- Waldvogel FA, Medoff G, Swartz MN. Osteomyelitis: a review of clinical features, therapeutic considerations and unusual aspects. 3. Osteomyelitis associated with vascular insufficiency. N Engl J Med 1970; 282:316–322.
- Carragee EJ. Pyogenic vertebral osteomyelitis. J Bone Joint Surg Am 1997; 79:874–880.
- Kapeller P, Fazekas F, Krametter D, et al. Pyogenic infectious spondylitis: clinical, laboratory and MRI features. Eur Neurol 1997; 38:94–98.
- Digby JM, Kersley JB. Pyogenic non-tuberculous spinal infection: an analysis of thirty cases. J Bone Joint Surg Br 1979; 61:47–55.
- Modic MT, Feiglin DH, Piraino DW, et al. Vertebral osteomyelitis: assessment using MR. Radiology 1985; 157:157–166.
- Szypryt EP, Hardy JG, Hinton CE, Worthington BS, Mulholland RC. A comparison between magnetic resonance imaging and scintigraphic bone imaging in the diagnosis of disk space infection in an animal model. Spine 1988; 13:1042–1048.
- Küker W, Mull M, Mayfrank L, Töpper R, Thron A. Epidural spinal infection. Variability of clinical and magnetic resonance imaging findings. Spine 1997; 22:544–551.
- Tung GA, Yim JW, Mermel LA, Philip L, Rogg JM. Spinal epidural abscess: correlation between MRI findings and outcome. Neuroradiology 1999; 41:904–909.
- Sapico FL, Montgomerie JZ. Vertebral osteomyelitis. Infect Dis Clin North Am 1990; 4:539–550.
- Tali ET. Spinal infections. Eur J Radiol 2004; 50:120–133.
- Smith AS, Weinstein MA, Mizushima A, et al. MR imaging characteristics of tuberculous spondylitis vs vertebral osteomyelitis. AJR Am J Roentgenol 1989; 153:399–405.
- Jung NY, Jee WH, Ha KY, Park CK, Byun JY. Discrimination of tuberculous spondylitis from pyogenic spondylitis on MRI. AJR Am J Roentgenol 2004; 182:1405–1410.
- Joseffer SS, Cooper PR. Modern imaging of spinal tuberculosis. J Neurosurg Spine 2005; 2:145–150.
- Pigrau C, Almirante B, Flores X, et al. Spontaneous pyogenic vertebral osteomyelitis and endocarditis: incidence, risk factors, and outcome. Am J Med 2005; 118:1287.
- McHenry MC, Easley KA, Locker GA. Vertebral osteomyelitis: long-term outcome for 253 patients from 7 Cleveland-area hospitals. Clin Infect Dis 2002; 34:1342–1350.
- Danner RL, Hartman BJ. Update on spinal epidural abscess: 35 cases and review of the literature. Rev Infect Dis 1987; 9:265–274.
- Kaufman DM, Kaplan JG, Litman N. Infectious agents in spinal epidural abscesses. Neurology 1980; 30:844–850.
- Rezai AR, Woo HH, Errico TJ, Cooper PR. Contemporary management of spinal osteomyelitis. Neurosurgery 1999; 44:1018–1026.
- Bertino RE, Porter BA, Stimac GK, Tepper SJ. Imaging spinal osteomyelitis and epidural abscess with short TI inversion recovery (STIR). AJNR Am J Neuroradiol 1988; 9:563–564.
- Rigamonti D, Liem L, Sampath P, et al. Spinal epidural abscess: contemporary trends in etiology, evaluation, and management. Surg Neurol 1999; 52:189–197.
- De Vries F, Bracke M, Leufkens HG, Lammers JW, Cooper C, Van Staa TP. Fracture risk with intermittent high-dose oral glucocorticoid therapy. Arthritis Rheum 2007; 56:208–214.
- Cooper C, Atkinson EJ, O’Fallon WM, Melton LJ. Incidence of clinically diagnosed vertebral fractures: a population-based study in Rochester, Minnesota, 1985–1989. J Bone Miner Res 1992; 7:221–227.
- Kim DH, Vaccaro AR. Osteoporotic compression fractures of the spine; current options and considerations for treatment. Spine J 2006; 6:479–487.
- Cohn SH, Abesamis C, Yasumura S, Aloia JF, Zanzi I, Ellis KJ. Comparative skeletal mass and radial bone mineral content in black and white women. Metabolism 1977; 26:171–178.
- Tobias JH, Hutchinson AP, Hunt LP, et al. Use of clinical risk factors to identify postmenopausal women with vertebral fractures. Osteoporos Int 2007; 18:35–43.
- Melton LJ, Kallmes DF. Epidemiology of vertebral fractures: implications for vertebral augmentation. Acad Radiol 2006; 13:538–545.
- Silverman SL. The clinical consequences of vertebral compression fracture. Bone 1992; 13:S27–S31.
- Melton LJ, Kan SH, Frye MA, Wahner HW, O’Fallon WM, Riggs BL. Epidemiology of vertebral fractures in women. Am J Epidemiol 1989; 129:1000–1011.
- Wasnich RD. Vertebral fracture epidemiology. Bone 1996; 18:179S–183S.
- Yamato M, Nishimura G, Kuramochi E, Saiki N, Fujioka M. MR appearance at different ages of osteoporotic compression fractures of the vertebrae. Radiat Med 1998; 16:329–334.
- Einsiedel T, Schmelz A, Arand M, et al. Injuries of the cervical spine in patients with ankylosing spondylitis: experience at two trauma centers. J Neurosurg Spine 2006; 5:33–45.
- Olerud C, Frost A, Bring J. Spinal fractures in patients with ankylosing spondylitis. Eur Spine J 1996; 5:51–55.
- Spangfort EV. The lumbar disk herniation. A computer-aided analysis of 2,504 operations. Acta Orthop Scand Suppl 1972; 142:1–95.
- Kostuik JP, Harrington I, Alexander D, Rand W, Evans D. Cauda equina syndrome and lumbar disk herniation. J Bone Joint Surg Am 1986; 68:386–391.
- Lawton MT, Porter RW, Heiserman JE, Jacobowitz R, Sonntag VK, Dickman CA. Surgical management of spinal epidural hematoma: relationship between surgical timing and neurological outcome. J Neurosurg 1995; 83:1–7.
- Simmons EH, Grobler LJ. Acute spinal epidural hematoma. J Bone Joint Surg Am 1978; 60:395–396.
- Senelick RC, Norwood CW, Cohen GH. “Painless” spinal epidural hematoma during anticoagulant therapy”. Neurology 1976; 26:213–225.
- Watts C, Porto L. Recognizing spontaneous spinal epidural hematoma. Geriatrics 1976; 31:97–99.
Back pain is one of the most common complaints that internists and primary care physicians encounter.1 Although back pain is nonspecific, some hallmark signs and symptoms indicate that a patient is more likely to have a serious disorder. This article contrasts the presentation of cancer, infections, and fractures with the more common and benign conditions that cause back pain and provides guidance for diagnosis.
UNCOMMON, BUT MUST BE CONSIDERED
Although a variety of tissues can contribute to pain—intervertebral disks, vertebrae, ligaments, neural structures, muscles, and fascia—and many disorders can damage these tissues, most patients with back or neck pain have a benign condition. Back pain is typically caused by age-related degenerative changes or by minor repetitive trauma; with supportive care and physical therapy, up to 90% of patients with back pain of this nature improve substantially within 4 weeks.2
Serious, destructive diseases are uncommon causes of back pain: malignancy, infection, ankylosing spondylitis, and epidural abscess together account for fewer than 1% of cases of back pain in a typical primary care practice. But their clinical impact is out of proportion to their prevalence. The fear of overlooking a serious condition influences any practitioner’s approach to back pain and is a common reason for ordering multiple imaging studies and consultations.3 Therefore, the time, effort, and resources invested in ruling out these disorders is considerable.
Whether a patient with back pain has an ominous disease can usually be determined with a careful history, physical examination, and appropriate diagnostic studies. Once a serious diagnosis is ruled out, attention can be focused on rehabilitation and back care.
Back pain can also be due to musculoskeletal disorders, peptic ulcers, pancreatitis, pyelonephritis, aortic aneurysms, and other serious conditions, which we have discussed in other articles in this journal.4–6
SPINAL CANCER AND METASTASES
Since back pain is the presenting symptom in 90% of patients with spinal tumors,7 neoplasia belongs in the differential diagnosis of any patient with persistent, unremitting back pain. However, it is also important to recognize atypical presentations of neoplasia, such as a painless neurologic deficit, which should prompt an urgent workup.
The spine is one of the most common sites of metastasis: about 20,000 cases arise each year.8 Brihaye et al9 reviewed 1,477 cases of spinal metastases with epidural involvement and found that 16.5% arose from primary tumors in the breast, 15.6% from the lung, 9.2% from the prostate, and 6.5% from the kidney.
Cancer pain is persistent and progressive
Benign back pain often arises from a known injury, is relieved by rest, and increases with activities that load the disk (eg, sitting, getting up from bed or a chair), lumbar flexion with or without rotation, lifting, vibration (eg, riding in a car), coughing, sneezing, laughing, and the Valsalva maneuver. It is most commonly focal to the lumbosacral junction, the lumbar muscles, and the buttocks. Pain due to injury or a flare-up of degenerative disease typically begins to subside after 4 to 6 weeks and responds to nonsteroidal anti-inflammatory drugs and physical therapy.10
In contrast, pain caused by spinal neoplasia is typically persistent and progressive and is not alleviated by rest. Often the pain is worse at night, waking the patient from sleep. Back pain is typically focal to the level of the lesion and may be associated with belt-like thoracic pain or radicular symptoms of pain or weakness in the legs. A spinal mass can cause neurologic signs or symptoms by directly compressing the spinal cord or nerve roots, mimicking disk herniation or stenosis.11,12
Pathologic fractures resulting from vertebral destruction may be the first—and unfortunately a late—presentation of a tumor.
Ask about, look for, signs and symptoms of cancer
In taking the history, one should ask about possible signs and symptoms of systemic disease such as fatigue, weight loss, and changes in bowel habits. Hemoptysis, lymphadenopathy, subcutaneous or breast masses, nipple discharge, atypical vaginal bleeding, or blood in the stool suggest malignancy and should direct the specific diagnostic approach.13 A history of cancer, even if remote, should raise suspicion, as should major risk factors such as smoking.
Because most spinal tumors are metastases, a clinical examination of the breast, lungs, abdomen, thyroid, and prostate are appropriate starting points.14 The spine should be examined to identify sites of focal pain. A neurologic examination should be done to evaluate any signs of neurologic compromise or abnormal reflexes. Signs or symptoms of spinal cord compression should be investigated immediately.
Cancer usually elevates the ESR, CRP
If cancer is suspected, initial tests should include a complete blood cell count, erythrocyte sedimentation rate, C-reactive protein level, urinalysis, prostate-specific antigen level, and fecal occult blood testing. Normal results can considerably relieve suspicion of cancer: the erythrocyte sedimentation rate and C-reactive protein level are almost always elevated with systemic neoplasia.
Imaging tests
Unfortunately, spinal tumors cannot be well visualized on radiographs until significant destruction has occurred.15
A bone scan can usually detect tumors other than the purely lytic ones such as myeloma and has a sensitivity of 74%, a specificity of 81%, and a positive predictive value of 64% for vertebral metastasis in patients with back pain.16
INFECTION CAN BE INDOLENT OR ACUTE
Spinal infection is a serious condition that can take an indolent, smoldering course or, alternatively, can erupt into sepsis or rapidly progressive vertebral destruction. Although the latter conditions are hard to miss, early diskitis and osteomyelitis can be difficult to differentiate from idiopathic back pain. In a series of 101 patients with vertebral osteomyelitis, misdiagnosis occurred in 33.7%, and the average delay from the onset of clinical manifestations to diagnosis was 2.6 months.20 Tuberculosis can be even more elusive: in a series of 78 patients diagnosed with definite or probable tuberculous vertebral osteomyelitis, the mean delay to diagnosis was about 6 months.21
Acute spinal infections are most often pyogenic; chronic infections may be pyogenic, fungal, or granulomatous.
Vertebral osteomyelitis accounts for 2% to 7% of all cases of osteomyelitis and is an uncommon cause of back pain.22 Any source of infection (eg, dental abscess, pneumonia) can seed the spine; urinary tract infection is the most common. Patients with immunocompromise or diabetes are most at risk.23 The onset is usually insidious with focal back pain at the level of involvement.
History and physical examination reveal localized pain
Spinal infections typically cause pain that is worsened with weight-bearing and activity and is relieved only when lying down. Chronic infection is usually associated with weight loss, fatigue, fevers, and night sweats.
Pain is usually well localized and reproduced by palpation or percussion over the involved level. Severe pain can sometimes be elicited by sitting the patient up or by changing the patient’s position. Focal kyphosis may be detectable if the vertebra has collapsed.
In a series of 41 patients with pyogenic infectious spondylitis, 90% had localized back pain aggravated by percussion, 59% had radicular signs and symptoms, and 29% had neurologic signs of spinal cord compression, including hyperreflexia, clonus, the Babinski sign (extension of the toes upward when the sole of the foot is stroked upwards), or the Hoffmann sign (flexion of the thumb elicited by flicking the end of a middle finger).24
LABORATORY RESULTS TYPICALLY INDICATE INFECTION
The erythrocyte sedimentation rate is the most sensitive test for infection, and an elevated rate may be the only abnormal laboratory finding: Digby and Kersley25 found that the rate was increased in all of 30 patients with nontuberculous pyogenic osteomyelitis of the spine. The C-reactive protein level is also usually elevated, but 40% of patients have a normal white blood cell count.25 Results of other laboratory tests are typically in the normal range. Tuberculin skin testing should be done for patients at high risk of the disease (eg, immigrants from areas of endemic disease, non-Hispanic blacks, immunocompromised patients, and those with known exposure to tuberculosis). Patients with high fever, chills, or rigors should have cultures taken of blood, urine, and sputum and from any intravenous lines.
Imaging changes may not appear for months
CT, on the other hand, may be better for showing the extent of bone involvement. In cases of vertebral osteomyelitis and intervertebral disk space infection, simultaneous involvement of the adjacent vertebral end plates and the intervertebral disk are the major findings.30
Signs of infection using T1-weighted MRI include low-signal marrow or disk spaces within the vertebral body, loss of definition of end plates (which appear hypointense compared with the bone marrow), and destruction of the cortical margins of the involved vertebral bodies. T2-weighted MRI typically discloses high signals of the affected areas of the vertebral body and disk. Contrast should be used to increase specificity; enhancement may be the first sign of an acute inflammatory process.31
CT and MRI can help identify sequestra, perilesional sclerosis, and epidural or soft tissue abscesses. Guided biopsy may be needed to differentiate between abscess, hematoma, tumor, and inflammation.
MRI findings: Pyogenic vs tuberculous spondylitis
MRI can help differentiate pyogenic vertebral osteomyelitis from tubercular disease, although findings may be similar (eg, both conditions have a high signal on T2-weighted images).32 Jung et al,33 in a retrospective study of 52 patients with spondylitis, found that compared with patients with pyogenic infections, patients with tuberculous spondylitis had a significantly higher incidence of a well-defined paraspinal abnormal signal on MRI, a thin and smooth abscess wall, a paraspinal or intraosseous abscess, subligamentous spread to three or more vertebral levels, involvement of multiple vertebral bodies, thoracic spine involvement, and a hyperintense signal on T2-weighted images. Other MRI features characteristically seen in patients with tuberculous spinal disease are anterior corner destruction, a relative preservation of the intervertebral disk, and large soft-tissue abscesses with calcifications.34
Prompt diagnosis and aggressive treatment needed
Pigrau et al35 found that spinal osteomyelitis is highly associated with endocarditis: among 606 patients with infectious endocarditis, 28 (4.6%) had pyogenic vertebral osteomyelitis, and among 91 patients with pyogenic vertebral osteomyelitis, 28 (30.8%) had infectious endocarditis.
McHenry et al36 retrospectively studied outcomes of 253 patients with vertebral osteomyelitis after a median of 6.5 years (range 2 days to 38 years): 11% died, more than one-third of survivors had residual disability, and 14% had a relapse. Surgery resulted in recovery or improvement in 86 (79%) of 109 patients. Independent risk factors for adverse outcome (death or incomplete recovery) were neurologic compromise, increased time to diagnosis, and having a hospital-acquired infection (P = .004). Relapse commonly developed in patients with severe vertebral destruction and abscesses, which appeared some time after surgical drainage or debridement. Recurrent bacteremia, paravertebral abscesses, and chronically draining sinuses were independently associated with relapse (P = .001). MRI, done in 110 patients, was often performed late in the course of infection and did not significantly affect outcome. The authors stressed that an optimal outcome of vertebral osteomyelitis requires heightened awareness, early diagnosis, prompt identification of pathogens, reversal of complications, and prolonged antimicrobial therapy.
Epidural abscess may also be present
Epidural abscess occurs in 10% of spine infections. About half of patients with an epidural abscess are misdiagnosed on their initial evaluation.37,38 Patients initially complain of local spine pain, followed by radicular pain, weakness, and finally paralysis. Between 12% and 30% of patients report a history of trauma, even as minor as a fall, preceding the infection.38,39
Radiologic findings are frequently equivocal, and MRI is preferred; gadolinium enhancement further increases sensitivity.39,40 Spinal canal abscesses usually appear hypointense on T1-weighted images and hyperintense on T2-weighted images, with ring enhancement surrounding the abscess area in contrast studies.41 MRI may give negative findings in the early stages of a spinal canal infection and so may need to be repeated.41 MRI may not help distinguish an epidural from a subdural abscess. However, primary spinal epidural abscesses without concomitant vertebral osteomyelitis are rare; therefore, the finding of associated vertebral osteomyelitis makes a spinal epidural abscess more likely.
FRACTURES
Fractures of the spine can be asymptomatic and may have no preceding trauma. They can be due to osteoporosis, malignancy, infection, or metabolic disorders such as renal osteodystrophy or hyperparathyroidism. Fractures in normal bone are almost always associated with trauma. Any suspicion of infection or malignancy should be investigated.
Corticosteroids increase risk
Any patient with back pain who is receiving corticosteroid therapy should be considered as having a compression fracture until proven otherwise.3 De Vries et al42 found that in a database of nearly 200,000 patients receiving glucocorticoids, risk increased substantially with increasing cumulative exposure. Those who intermittently received high doses (= 15 mg/day) and those who had no or little previous exposure to corticosteroids (cumulative exposure = 1 g) had only a slightly increased risk of osteoporotic fracture, and their risk of fracture of the hip and femur was not increased. In contrast, patients who received a daily dose of at least 30 mg and whose cumulative exposure was more than 5 g had a relative risk of osteoporotic vertebral fracture of 14.42 (95% confidence interval 8.29–25.08).
Osteoporotic compression fractures are common in the elderly
Osteoporosis involves reduced bone density, disrupted trabecular architecture, and increased susceptibility to fractures. About 700,000 vertebral body compression fractures occur in the United States each year43: about 10% result in hospitalization, involving an average stay of 8 days.44 Osteoporotic compression fractures are highly associated with age older than 65, female sex, and European descent.45,46 The estimated lifetime risk of a clinically evident vertebral fracture after age 50 years is 16% among postmenopausal white women and 5% among white men.47
A single osteoporotic vertebral compression fracture increases the risk of subsequent fractures by a factor of five, and up to 20% of patients with a vertebral compression fracture are likely to have another one within the same year if osteoporosis remains untreated.48 Population studies suggest that the death rate among patients who have osteoporotic vertebral compression fractures increases with the number of involved vertebrae.43
Unfortunately, osteoporotic vertebral compression fractures are not always easily amenable to treatment: up to 30% of patients who are symptomatic and seek treatment do not respond adequately to nonsurgical methods.49,50 However, new minimally invasive interventions such as vertebral augmentation make timely evaluation clinically relevant.
History, physical examination
Patients may present with a history of trauma with associated back pain or a neurologic deficit. In osteoporotic patients, the trauma may have been minimal, eg, a sneeze, a fall from a chair, or a slip and fall in the home. Pain tends to be worse when standing erect and occasionally when lying flat.
The patient is commonly visibly uncomfortable and may be limited to a wheelchair or stoop forward when standing. The spine may show an absence of the midline crease or an exaggerated thoracic kyphosis. Pain is typically reproduced by deep pressure over the spinous process at the involved level. Compression fractures rarely cause neurologic deficits but should always be considered.
Fractures commonly occur in the thoracolumbar region but may be anywhere in the spine. Fractures in the upper thoracic spine may indicate an underlying malignant tumor, and a thorough search for a possible primary lesion should always be carried out for fractures in this location.
Laboratory testing
Routine laboratory evaluation and thyroid function tests should be done, as well as a 24-hour urine specimen for collagen breakdown products, calcium, phosphate, and creatinine levels. Serum and urine protein electrophoresis should be performed if myeloma is suspected. A white blood-cell count, erythrocyte sedimentation rate, and C-reactive protein level help determine if an underlying infection caused the fracture.
MRI needed if plain films reveal fracture or are equivocal
Anteroposterior and lateral roentgenograms should be taken first; they typically show osteopenia. A fracture in the vertebral body is characterized by loss of height and by wedging. Osseous fragments can occasionally be seen in the spinal canal.
Sagittal short tau inversion recovery sequences, which use specifically timed pulse sequences to suppress fat signals, show high-intensity signal changes in areas of edema from acute or healing fractures. They provide a sensitive but nonspecific marker of abnormality.
Dual energy x-ray absorptiometry helps determine the extent of osteoporosis.
Bone scans should only be used for patients with suspected metastatic disease.
Patients with ankylosing spondylitis need thorough workup
Ankylosing spondylitis predisposes to serious spinal injury. Even after only minor trauma, patients with ankylosing spondylitis and acute, severe back pain should be thoroughly evaluated for fracture with CT and MRI of the entire spine. Plain radiography should not be relied on for these patients because of the risk of misinterpretation, delayed diagnosis, and poorer outcomes.52,53
NEUROLOGIC COMPROMISE—A RED FLAG
Cauda equina compression classically presents with back pain, bilateral sciatica, saddle anesthesia, and lower extremity weakness progressing to paraplegia, but in practice these symptoms are variably present and diagnosing the condition often requires a high degree of suspicion. Hyporeflexia is typically a sign of cauda equina compression, while hyperreflexia, clonus, and the Babinski sign suggest spinal cord compression, requiring an evaluation of the cervical and thoracic spine. Cauda equina compression typically involves urinary retention; in contrast, cord compression typically causes incontinence.55
If either cauda equina or spinal cord compression is detected during an initial examination, an immediate more extensive evaluation is warranted. MRI is the study of choice.
Spinal epidural hematoma
Spinal epidural hematoma is a rare but dramatic cause of paralysis in elderly patients. In most cases, there is no antecedent trauma. Lawton et al,56 in a series of 30 patients treated surgically for spinal epidural hematoma, found that 73% resulted from spine surgery, epidural catheterization, or anticoagulation therapy. Other possible causes of epidural hematoma include vascular malformations, angiomas, aneurysms, hypertension, and aspirin therapy.57
The same study56 found that the time from the first symptom to maximal neurologic deficit ranged from a few minutes to 4 days, with the average interval being nearly 13 hours.
Although painless onset has been reported,58 spinal epidural hematoma typically presents with acute pain at the level of the lesion, which is often rapidly followed by paraplegia or quadriplegia, depending on the location of the hemorrhage. Sometimes the onset of pain is preceded by a sudden increase of venous pressure from coughing, sneezing, or straining at stool. Urinary retention often develops at an early stage.
Most lesions occur in the thoracic region and extend into the cervicothoracic or the thoracolumbar area. The pain distribution may be radicular, mimicking a ruptured intervertebral disk.
Evaluation should be with MRI. Acute hemorrhage is characterized by a marked decrease in signal intensity on T2-weighted images. Subacute hematoma has increased signal intensity on both T1- and T2-weighted images.56
Early recognition, MRI confirmation, and treatment should be accomplished as soon as possible.56 Recovery depends on the severity of the neurologic deficit and the duration of symptoms before treatment. Lawton et al56 found that patients taken to surgery within 12 hours had better neurologic outcomes than patients with identical preoperative neurologic status whose surgery was delayed beyond 12 hours. Surgery should not be withheld because of advanced age or poor health: in 10 reported cases in which surgery was delayed, all patients died.59
Back pain is one of the most common complaints that internists and primary care physicians encounter.1 Although back pain is nonspecific, some hallmark signs and symptoms indicate that a patient is more likely to have a serious disorder. This article contrasts the presentation of cancer, infections, and fractures with the more common and benign conditions that cause back pain and provides guidance for diagnosis.
UNCOMMON, BUT MUST BE CONSIDERED
Although a variety of tissues can contribute to pain—intervertebral disks, vertebrae, ligaments, neural structures, muscles, and fascia—and many disorders can damage these tissues, most patients with back or neck pain have a benign condition. Back pain is typically caused by age-related degenerative changes or by minor repetitive trauma; with supportive care and physical therapy, up to 90% of patients with back pain of this nature improve substantially within 4 weeks.2
Serious, destructive diseases are uncommon causes of back pain: malignancy, infection, ankylosing spondylitis, and epidural abscess together account for fewer than 1% of cases of back pain in a typical primary care practice. But their clinical impact is out of proportion to their prevalence. The fear of overlooking a serious condition influences any practitioner’s approach to back pain and is a common reason for ordering multiple imaging studies and consultations.3 Therefore, the time, effort, and resources invested in ruling out these disorders is considerable.
Whether a patient with back pain has an ominous disease can usually be determined with a careful history, physical examination, and appropriate diagnostic studies. Once a serious diagnosis is ruled out, attention can be focused on rehabilitation and back care.
Back pain can also be due to musculoskeletal disorders, peptic ulcers, pancreatitis, pyelonephritis, aortic aneurysms, and other serious conditions, which we have discussed in other articles in this journal.4–6
SPINAL CANCER AND METASTASES
Since back pain is the presenting symptom in 90% of patients with spinal tumors,7 neoplasia belongs in the differential diagnosis of any patient with persistent, unremitting back pain. However, it is also important to recognize atypical presentations of neoplasia, such as a painless neurologic deficit, which should prompt an urgent workup.
The spine is one of the most common sites of metastasis: about 20,000 cases arise each year.8 Brihaye et al9 reviewed 1,477 cases of spinal metastases with epidural involvement and found that 16.5% arose from primary tumors in the breast, 15.6% from the lung, 9.2% from the prostate, and 6.5% from the kidney.
Cancer pain is persistent and progressive
Benign back pain often arises from a known injury, is relieved by rest, and increases with activities that load the disk (eg, sitting, getting up from bed or a chair), lumbar flexion with or without rotation, lifting, vibration (eg, riding in a car), coughing, sneezing, laughing, and the Valsalva maneuver. It is most commonly focal to the lumbosacral junction, the lumbar muscles, and the buttocks. Pain due to injury or a flare-up of degenerative disease typically begins to subside after 4 to 6 weeks and responds to nonsteroidal anti-inflammatory drugs and physical therapy.10
In contrast, pain caused by spinal neoplasia is typically persistent and progressive and is not alleviated by rest. Often the pain is worse at night, waking the patient from sleep. Back pain is typically focal to the level of the lesion and may be associated with belt-like thoracic pain or radicular symptoms of pain or weakness in the legs. A spinal mass can cause neurologic signs or symptoms by directly compressing the spinal cord or nerve roots, mimicking disk herniation or stenosis.11,12
Pathologic fractures resulting from vertebral destruction may be the first—and unfortunately a late—presentation of a tumor.
Ask about, look for, signs and symptoms of cancer
In taking the history, one should ask about possible signs and symptoms of systemic disease such as fatigue, weight loss, and changes in bowel habits. Hemoptysis, lymphadenopathy, subcutaneous or breast masses, nipple discharge, atypical vaginal bleeding, or blood in the stool suggest malignancy and should direct the specific diagnostic approach.13 A history of cancer, even if remote, should raise suspicion, as should major risk factors such as smoking.
Because most spinal tumors are metastases, a clinical examination of the breast, lungs, abdomen, thyroid, and prostate are appropriate starting points.14 The spine should be examined to identify sites of focal pain. A neurologic examination should be done to evaluate any signs of neurologic compromise or abnormal reflexes. Signs or symptoms of spinal cord compression should be investigated immediately.
Cancer usually elevates the ESR, CRP
If cancer is suspected, initial tests should include a complete blood cell count, erythrocyte sedimentation rate, C-reactive protein level, urinalysis, prostate-specific antigen level, and fecal occult blood testing. Normal results can considerably relieve suspicion of cancer: the erythrocyte sedimentation rate and C-reactive protein level are almost always elevated with systemic neoplasia.
Imaging tests
Unfortunately, spinal tumors cannot be well visualized on radiographs until significant destruction has occurred.15
A bone scan can usually detect tumors other than the purely lytic ones such as myeloma and has a sensitivity of 74%, a specificity of 81%, and a positive predictive value of 64% for vertebral metastasis in patients with back pain.16
INFECTION CAN BE INDOLENT OR ACUTE
Spinal infection is a serious condition that can take an indolent, smoldering course or, alternatively, can erupt into sepsis or rapidly progressive vertebral destruction. Although the latter conditions are hard to miss, early diskitis and osteomyelitis can be difficult to differentiate from idiopathic back pain. In a series of 101 patients with vertebral osteomyelitis, misdiagnosis occurred in 33.7%, and the average delay from the onset of clinical manifestations to diagnosis was 2.6 months.20 Tuberculosis can be even more elusive: in a series of 78 patients diagnosed with definite or probable tuberculous vertebral osteomyelitis, the mean delay to diagnosis was about 6 months.21
Acute spinal infections are most often pyogenic; chronic infections may be pyogenic, fungal, or granulomatous.
Vertebral osteomyelitis accounts for 2% to 7% of all cases of osteomyelitis and is an uncommon cause of back pain.22 Any source of infection (eg, dental abscess, pneumonia) can seed the spine; urinary tract infection is the most common. Patients with immunocompromise or diabetes are most at risk.23 The onset is usually insidious with focal back pain at the level of involvement.
History and physical examination reveal localized pain
Spinal infections typically cause pain that is worsened with weight-bearing and activity and is relieved only when lying down. Chronic infection is usually associated with weight loss, fatigue, fevers, and night sweats.
Pain is usually well localized and reproduced by palpation or percussion over the involved level. Severe pain can sometimes be elicited by sitting the patient up or by changing the patient’s position. Focal kyphosis may be detectable if the vertebra has collapsed.
In a series of 41 patients with pyogenic infectious spondylitis, 90% had localized back pain aggravated by percussion, 59% had radicular signs and symptoms, and 29% had neurologic signs of spinal cord compression, including hyperreflexia, clonus, the Babinski sign (extension of the toes upward when the sole of the foot is stroked upwards), or the Hoffmann sign (flexion of the thumb elicited by flicking the end of a middle finger).24
LABORATORY RESULTS TYPICALLY INDICATE INFECTION
The erythrocyte sedimentation rate is the most sensitive test for infection, and an elevated rate may be the only abnormal laboratory finding: Digby and Kersley25 found that the rate was increased in all of 30 patients with nontuberculous pyogenic osteomyelitis of the spine. The C-reactive protein level is also usually elevated, but 40% of patients have a normal white blood cell count.25 Results of other laboratory tests are typically in the normal range. Tuberculin skin testing should be done for patients at high risk of the disease (eg, immigrants from areas of endemic disease, non-Hispanic blacks, immunocompromised patients, and those with known exposure to tuberculosis). Patients with high fever, chills, or rigors should have cultures taken of blood, urine, and sputum and from any intravenous lines.
Imaging changes may not appear for months
CT, on the other hand, may be better for showing the extent of bone involvement. In cases of vertebral osteomyelitis and intervertebral disk space infection, simultaneous involvement of the adjacent vertebral end plates and the intervertebral disk are the major findings.30
Signs of infection using T1-weighted MRI include low-signal marrow or disk spaces within the vertebral body, loss of definition of end plates (which appear hypointense compared with the bone marrow), and destruction of the cortical margins of the involved vertebral bodies. T2-weighted MRI typically discloses high signals of the affected areas of the vertebral body and disk. Contrast should be used to increase specificity; enhancement may be the first sign of an acute inflammatory process.31
CT and MRI can help identify sequestra, perilesional sclerosis, and epidural or soft tissue abscesses. Guided biopsy may be needed to differentiate between abscess, hematoma, tumor, and inflammation.
MRI findings: Pyogenic vs tuberculous spondylitis
MRI can help differentiate pyogenic vertebral osteomyelitis from tubercular disease, although findings may be similar (eg, both conditions have a high signal on T2-weighted images).32 Jung et al,33 in a retrospective study of 52 patients with spondylitis, found that compared with patients with pyogenic infections, patients with tuberculous spondylitis had a significantly higher incidence of a well-defined paraspinal abnormal signal on MRI, a thin and smooth abscess wall, a paraspinal or intraosseous abscess, subligamentous spread to three or more vertebral levels, involvement of multiple vertebral bodies, thoracic spine involvement, and a hyperintense signal on T2-weighted images. Other MRI features characteristically seen in patients with tuberculous spinal disease are anterior corner destruction, a relative preservation of the intervertebral disk, and large soft-tissue abscesses with calcifications.34
Prompt diagnosis and aggressive treatment needed
Pigrau et al35 found that spinal osteomyelitis is highly associated with endocarditis: among 606 patients with infectious endocarditis, 28 (4.6%) had pyogenic vertebral osteomyelitis, and among 91 patients with pyogenic vertebral osteomyelitis, 28 (30.8%) had infectious endocarditis.
McHenry et al36 retrospectively studied outcomes of 253 patients with vertebral osteomyelitis after a median of 6.5 years (range 2 days to 38 years): 11% died, more than one-third of survivors had residual disability, and 14% had a relapse. Surgery resulted in recovery or improvement in 86 (79%) of 109 patients. Independent risk factors for adverse outcome (death or incomplete recovery) were neurologic compromise, increased time to diagnosis, and having a hospital-acquired infection (P = .004). Relapse commonly developed in patients with severe vertebral destruction and abscesses, which appeared some time after surgical drainage or debridement. Recurrent bacteremia, paravertebral abscesses, and chronically draining sinuses were independently associated with relapse (P = .001). MRI, done in 110 patients, was often performed late in the course of infection and did not significantly affect outcome. The authors stressed that an optimal outcome of vertebral osteomyelitis requires heightened awareness, early diagnosis, prompt identification of pathogens, reversal of complications, and prolonged antimicrobial therapy.
Epidural abscess may also be present
Epidural abscess occurs in 10% of spine infections. About half of patients with an epidural abscess are misdiagnosed on their initial evaluation.37,38 Patients initially complain of local spine pain, followed by radicular pain, weakness, and finally paralysis. Between 12% and 30% of patients report a history of trauma, even as minor as a fall, preceding the infection.38,39
Radiologic findings are frequently equivocal, and MRI is preferred; gadolinium enhancement further increases sensitivity.39,40 Spinal canal abscesses usually appear hypointense on T1-weighted images and hyperintense on T2-weighted images, with ring enhancement surrounding the abscess area in contrast studies.41 MRI may give negative findings in the early stages of a spinal canal infection and so may need to be repeated.41 MRI may not help distinguish an epidural from a subdural abscess. However, primary spinal epidural abscesses without concomitant vertebral osteomyelitis are rare; therefore, the finding of associated vertebral osteomyelitis makes a spinal epidural abscess more likely.
FRACTURES
Fractures of the spine can be asymptomatic and may have no preceding trauma. They can be due to osteoporosis, malignancy, infection, or metabolic disorders such as renal osteodystrophy or hyperparathyroidism. Fractures in normal bone are almost always associated with trauma. Any suspicion of infection or malignancy should be investigated.
Corticosteroids increase risk
Any patient with back pain who is receiving corticosteroid therapy should be considered as having a compression fracture until proven otherwise.3 De Vries et al42 found that in a database of nearly 200,000 patients receiving glucocorticoids, risk increased substantially with increasing cumulative exposure. Those who intermittently received high doses (= 15 mg/day) and those who had no or little previous exposure to corticosteroids (cumulative exposure = 1 g) had only a slightly increased risk of osteoporotic fracture, and their risk of fracture of the hip and femur was not increased. In contrast, patients who received a daily dose of at least 30 mg and whose cumulative exposure was more than 5 g had a relative risk of osteoporotic vertebral fracture of 14.42 (95% confidence interval 8.29–25.08).
Osteoporotic compression fractures are common in the elderly
Osteoporosis involves reduced bone density, disrupted trabecular architecture, and increased susceptibility to fractures. About 700,000 vertebral body compression fractures occur in the United States each year43: about 10% result in hospitalization, involving an average stay of 8 days.44 Osteoporotic compression fractures are highly associated with age older than 65, female sex, and European descent.45,46 The estimated lifetime risk of a clinically evident vertebral fracture after age 50 years is 16% among postmenopausal white women and 5% among white men.47
A single osteoporotic vertebral compression fracture increases the risk of subsequent fractures by a factor of five, and up to 20% of patients with a vertebral compression fracture are likely to have another one within the same year if osteoporosis remains untreated.48 Population studies suggest that the death rate among patients who have osteoporotic vertebral compression fractures increases with the number of involved vertebrae.43
Unfortunately, osteoporotic vertebral compression fractures are not always easily amenable to treatment: up to 30% of patients who are symptomatic and seek treatment do not respond adequately to nonsurgical methods.49,50 However, new minimally invasive interventions such as vertebral augmentation make timely evaluation clinically relevant.
History, physical examination
Patients may present with a history of trauma with associated back pain or a neurologic deficit. In osteoporotic patients, the trauma may have been minimal, eg, a sneeze, a fall from a chair, or a slip and fall in the home. Pain tends to be worse when standing erect and occasionally when lying flat.
The patient is commonly visibly uncomfortable and may be limited to a wheelchair or stoop forward when standing. The spine may show an absence of the midline crease or an exaggerated thoracic kyphosis. Pain is typically reproduced by deep pressure over the spinous process at the involved level. Compression fractures rarely cause neurologic deficits but should always be considered.
Fractures commonly occur in the thoracolumbar region but may be anywhere in the spine. Fractures in the upper thoracic spine may indicate an underlying malignant tumor, and a thorough search for a possible primary lesion should always be carried out for fractures in this location.
Laboratory testing
Routine laboratory evaluation and thyroid function tests should be done, as well as a 24-hour urine specimen for collagen breakdown products, calcium, phosphate, and creatinine levels. Serum and urine protein electrophoresis should be performed if myeloma is suspected. A white blood-cell count, erythrocyte sedimentation rate, and C-reactive protein level help determine if an underlying infection caused the fracture.
MRI needed if plain films reveal fracture or are equivocal
Anteroposterior and lateral roentgenograms should be taken first; they typically show osteopenia. A fracture in the vertebral body is characterized by loss of height and by wedging. Osseous fragments can occasionally be seen in the spinal canal.
Sagittal short tau inversion recovery sequences, which use specifically timed pulse sequences to suppress fat signals, show high-intensity signal changes in areas of edema from acute or healing fractures. They provide a sensitive but nonspecific marker of abnormality.
Dual energy x-ray absorptiometry helps determine the extent of osteoporosis.
Bone scans should only be used for patients with suspected metastatic disease.
Patients with ankylosing spondylitis need thorough workup
Ankylosing spondylitis predisposes to serious spinal injury. Even after only minor trauma, patients with ankylosing spondylitis and acute, severe back pain should be thoroughly evaluated for fracture with CT and MRI of the entire spine. Plain radiography should not be relied on for these patients because of the risk of misinterpretation, delayed diagnosis, and poorer outcomes.52,53
NEUROLOGIC COMPROMISE—A RED FLAG
Cauda equina compression classically presents with back pain, bilateral sciatica, saddle anesthesia, and lower extremity weakness progressing to paraplegia, but in practice these symptoms are variably present and diagnosing the condition often requires a high degree of suspicion. Hyporeflexia is typically a sign of cauda equina compression, while hyperreflexia, clonus, and the Babinski sign suggest spinal cord compression, requiring an evaluation of the cervical and thoracic spine. Cauda equina compression typically involves urinary retention; in contrast, cord compression typically causes incontinence.55
If either cauda equina or spinal cord compression is detected during an initial examination, an immediate more extensive evaluation is warranted. MRI is the study of choice.
Spinal epidural hematoma
Spinal epidural hematoma is a rare but dramatic cause of paralysis in elderly patients. In most cases, there is no antecedent trauma. Lawton et al,56 in a series of 30 patients treated surgically for spinal epidural hematoma, found that 73% resulted from spine surgery, epidural catheterization, or anticoagulation therapy. Other possible causes of epidural hematoma include vascular malformations, angiomas, aneurysms, hypertension, and aspirin therapy.57
The same study56 found that the time from the first symptom to maximal neurologic deficit ranged from a few minutes to 4 days, with the average interval being nearly 13 hours.
Although painless onset has been reported,58 spinal epidural hematoma typically presents with acute pain at the level of the lesion, which is often rapidly followed by paraplegia or quadriplegia, depending on the location of the hemorrhage. Sometimes the onset of pain is preceded by a sudden increase of venous pressure from coughing, sneezing, or straining at stool. Urinary retention often develops at an early stage.
Most lesions occur in the thoracic region and extend into the cervicothoracic or the thoracolumbar area. The pain distribution may be radicular, mimicking a ruptured intervertebral disk.
Evaluation should be with MRI. Acute hemorrhage is characterized by a marked decrease in signal intensity on T2-weighted images. Subacute hematoma has increased signal intensity on both T1- and T2-weighted images.56
Early recognition, MRI confirmation, and treatment should be accomplished as soon as possible.56 Recovery depends on the severity of the neurologic deficit and the duration of symptoms before treatment. Lawton et al56 found that patients taken to surgery within 12 hours had better neurologic outcomes than patients with identical preoperative neurologic status whose surgery was delayed beyond 12 hours. Surgery should not be withheld because of advanced age or poor health: in 10 reported cases in which surgery was delayed, all patients died.59
- Deyo RA, Tsui-Wu YJ. Descriptive epidemiology of low-back pain and its related medical care in the United States. Spine 1987; 12:264–268.
- Pengel LH, Herbert RD, Maher CG, Refshauge KM. Acute back pain: systematic review of its prognosis. BMJ 2003; 327:323–325.
- Deyo RA, Rainville J, Kent DL. What can the history and physical examination tell us about back pain? JAMA 1992; 268:760–765.
- Pateder DB, Brems J, Lieberman I, Bell GR, McLain RF. Masquerade: nonspinal musculoskeletal disorders that mimic spinal conditions. Cleve Clin J Med 2008; 75:50–56.
- Klineberg E, Mazanec D, Orr D, Demicco R, Bell G, McLain R. Masquerade: medical causes of back pain. Cleve Clin J Med 2007; 74:905–913.
- McLain RF, Bell G, Montgomery W. Masquerade: systemic causes of back pain. Cleve Clin J Med In press.
- Gilbert RW, Kim JH, Posner JB. Epidural spinal cord compression from metastatic tumor: diagnosis and treatment. Ann Neurol 1978; 3:40–51.
- Black P. Spinal metastasis: current status and recommended guidelines for management. Neurosurgery 1979; 5:726–746.
- Brihaye J, Ectors P, Lemort M, Van Houtte P. The management of spinal epidural metastases. Adv Tech Stand Neurosurg 1988; 16:121–176.
- Patel RK, Slipman CW. Lumbar degenerative disk disease. Emedicine. Accessed March 25, 2008. http://www.emedicine.com/pmr/topic67.htm.
- Deyo RA, Diehl AK. Cancer as a cause of back pain: frequency, clinical presentation, and diagnostic strategies. J Gen Intern Med 1988; 3:230–238.
- Rosen P, Barkin RM, Danzl DF, et al. Emergency Medicine: Concepts and Clinical Practice. 4th ed. St Louis, MO: Mosby; 1998:2100–2102.
- Abbruzzese JL, Abbruzzese MC, Lenzi R, Hess KR, Raber MN. Analysis of a diagnostic strategy for patients with suspected tumors of unknown origin. J Clin Oncol 1995; 13:2094–2103.
- McCarthy EF, Frassica FJ. Pathology of Bone and Joint Disorders: With Clinical and Radiographic Correlation. Philadelphia: WB Saunders; 1998.
- Edelstyn GA, Gillespie PJ, Grebbell FS. The radiological demonstration of osseous metastases. Experimental observations. Clin Radiol 1967; 18:158–162.
- Han LJ, Au-Yong TK, Tong WC, Chu KS, Szeto LT, Wong CP. Comparison of bone single-photon emission tomography and planar imaging in the detection of vertebral metastases in patients with back pain. Eur J Nucl Med 1998; 25:635–638.
- Feun LG, Savaraj N. Detection of occult bone metastasis by MRI scan. J Fla Med Assoc 1990; 77:881–883.
- Citrin DL, Bessent RG, Greig WR. A comparison of the sensitivity and accuracy of the 99TCm-phosphate bone scan and skeletal radiograph in the diagnosis of bone metastases. Clin Radiol 1977; 28:107–117.
- Runge VM, Lee C, Iten AL, Williams NM. Contrast-enhanced magnetic resonance imaging in a spinal epidural tumor model. Invest Radiol 1997; 32:589–595.
- Buranapanitkit B, Lim A, Geater A. Misdiagnosis in vertebral osteomyelitis: problems and factors. J Med Assoc Thai 2001; 84:1743–1750.
- Colmenero JD, Jiménez-Mejías ME, Reguera JM, et al. Tuberculous vertebral osteomyelitis in the new millennium: still a diagnostic and therapeutic challenge. Eur J Clin Microbiol Infect Dis 2004; 23:477–483.
- Waldvogel FA, Medoff G, Swartz MN. Osteomyelitis: a review of clinical features, therapeutic considerations and unusual aspects. 3. Osteomyelitis associated with vascular insufficiency. N Engl J Med 1970; 282:316–322.
- Carragee EJ. Pyogenic vertebral osteomyelitis. J Bone Joint Surg Am 1997; 79:874–880.
- Kapeller P, Fazekas F, Krametter D, et al. Pyogenic infectious spondylitis: clinical, laboratory and MRI features. Eur Neurol 1997; 38:94–98.
- Digby JM, Kersley JB. Pyogenic non-tuberculous spinal infection: an analysis of thirty cases. J Bone Joint Surg Br 1979; 61:47–55.
- Modic MT, Feiglin DH, Piraino DW, et al. Vertebral osteomyelitis: assessment using MR. Radiology 1985; 157:157–166.
- Szypryt EP, Hardy JG, Hinton CE, Worthington BS, Mulholland RC. A comparison between magnetic resonance imaging and scintigraphic bone imaging in the diagnosis of disk space infection in an animal model. Spine 1988; 13:1042–1048.
- Küker W, Mull M, Mayfrank L, Töpper R, Thron A. Epidural spinal infection. Variability of clinical and magnetic resonance imaging findings. Spine 1997; 22:544–551.
- Tung GA, Yim JW, Mermel LA, Philip L, Rogg JM. Spinal epidural abscess: correlation between MRI findings and outcome. Neuroradiology 1999; 41:904–909.
- Sapico FL, Montgomerie JZ. Vertebral osteomyelitis. Infect Dis Clin North Am 1990; 4:539–550.
- Tali ET. Spinal infections. Eur J Radiol 2004; 50:120–133.
- Smith AS, Weinstein MA, Mizushima A, et al. MR imaging characteristics of tuberculous spondylitis vs vertebral osteomyelitis. AJR Am J Roentgenol 1989; 153:399–405.
- Jung NY, Jee WH, Ha KY, Park CK, Byun JY. Discrimination of tuberculous spondylitis from pyogenic spondylitis on MRI. AJR Am J Roentgenol 2004; 182:1405–1410.
- Joseffer SS, Cooper PR. Modern imaging of spinal tuberculosis. J Neurosurg Spine 2005; 2:145–150.
- Pigrau C, Almirante B, Flores X, et al. Spontaneous pyogenic vertebral osteomyelitis and endocarditis: incidence, risk factors, and outcome. Am J Med 2005; 118:1287.
- McHenry MC, Easley KA, Locker GA. Vertebral osteomyelitis: long-term outcome for 253 patients from 7 Cleveland-area hospitals. Clin Infect Dis 2002; 34:1342–1350.
- Danner RL, Hartman BJ. Update on spinal epidural abscess: 35 cases and review of the literature. Rev Infect Dis 1987; 9:265–274.
- Kaufman DM, Kaplan JG, Litman N. Infectious agents in spinal epidural abscesses. Neurology 1980; 30:844–850.
- Rezai AR, Woo HH, Errico TJ, Cooper PR. Contemporary management of spinal osteomyelitis. Neurosurgery 1999; 44:1018–1026.
- Bertino RE, Porter BA, Stimac GK, Tepper SJ. Imaging spinal osteomyelitis and epidural abscess with short TI inversion recovery (STIR). AJNR Am J Neuroradiol 1988; 9:563–564.
- Rigamonti D, Liem L, Sampath P, et al. Spinal epidural abscess: contemporary trends in etiology, evaluation, and management. Surg Neurol 1999; 52:189–197.
- De Vries F, Bracke M, Leufkens HG, Lammers JW, Cooper C, Van Staa TP. Fracture risk with intermittent high-dose oral glucocorticoid therapy. Arthritis Rheum 2007; 56:208–214.
- Cooper C, Atkinson EJ, O’Fallon WM, Melton LJ. Incidence of clinically diagnosed vertebral fractures: a population-based study in Rochester, Minnesota, 1985–1989. J Bone Miner Res 1992; 7:221–227.
- Kim DH, Vaccaro AR. Osteoporotic compression fractures of the spine; current options and considerations for treatment. Spine J 2006; 6:479–487.
- Cohn SH, Abesamis C, Yasumura S, Aloia JF, Zanzi I, Ellis KJ. Comparative skeletal mass and radial bone mineral content in black and white women. Metabolism 1977; 26:171–178.
- Tobias JH, Hutchinson AP, Hunt LP, et al. Use of clinical risk factors to identify postmenopausal women with vertebral fractures. Osteoporos Int 2007; 18:35–43.
- Melton LJ, Kallmes DF. Epidemiology of vertebral fractures: implications for vertebral augmentation. Acad Radiol 2006; 13:538–545.
- Silverman SL. The clinical consequences of vertebral compression fracture. Bone 1992; 13:S27–S31.
- Melton LJ, Kan SH, Frye MA, Wahner HW, O’Fallon WM, Riggs BL. Epidemiology of vertebral fractures in women. Am J Epidemiol 1989; 129:1000–1011.
- Wasnich RD. Vertebral fracture epidemiology. Bone 1996; 18:179S–183S.
- Yamato M, Nishimura G, Kuramochi E, Saiki N, Fujioka M. MR appearance at different ages of osteoporotic compression fractures of the vertebrae. Radiat Med 1998; 16:329–334.
- Einsiedel T, Schmelz A, Arand M, et al. Injuries of the cervical spine in patients with ankylosing spondylitis: experience at two trauma centers. J Neurosurg Spine 2006; 5:33–45.
- Olerud C, Frost A, Bring J. Spinal fractures in patients with ankylosing spondylitis. Eur Spine J 1996; 5:51–55.
- Spangfort EV. The lumbar disk herniation. A computer-aided analysis of 2,504 operations. Acta Orthop Scand Suppl 1972; 142:1–95.
- Kostuik JP, Harrington I, Alexander D, Rand W, Evans D. Cauda equina syndrome and lumbar disk herniation. J Bone Joint Surg Am 1986; 68:386–391.
- Lawton MT, Porter RW, Heiserman JE, Jacobowitz R, Sonntag VK, Dickman CA. Surgical management of spinal epidural hematoma: relationship between surgical timing and neurological outcome. J Neurosurg 1995; 83:1–7.
- Simmons EH, Grobler LJ. Acute spinal epidural hematoma. J Bone Joint Surg Am 1978; 60:395–396.
- Senelick RC, Norwood CW, Cohen GH. “Painless” spinal epidural hematoma during anticoagulant therapy”. Neurology 1976; 26:213–225.
- Watts C, Porto L. Recognizing spontaneous spinal epidural hematoma. Geriatrics 1976; 31:97–99.
- Deyo RA, Tsui-Wu YJ. Descriptive epidemiology of low-back pain and its related medical care in the United States. Spine 1987; 12:264–268.
- Pengel LH, Herbert RD, Maher CG, Refshauge KM. Acute back pain: systematic review of its prognosis. BMJ 2003; 327:323–325.
- Deyo RA, Rainville J, Kent DL. What can the history and physical examination tell us about back pain? JAMA 1992; 268:760–765.
- Pateder DB, Brems J, Lieberman I, Bell GR, McLain RF. Masquerade: nonspinal musculoskeletal disorders that mimic spinal conditions. Cleve Clin J Med 2008; 75:50–56.
- Klineberg E, Mazanec D, Orr D, Demicco R, Bell G, McLain R. Masquerade: medical causes of back pain. Cleve Clin J Med 2007; 74:905–913.
- McLain RF, Bell G, Montgomery W. Masquerade: systemic causes of back pain. Cleve Clin J Med In press.
- Gilbert RW, Kim JH, Posner JB. Epidural spinal cord compression from metastatic tumor: diagnosis and treatment. Ann Neurol 1978; 3:40–51.
- Black P. Spinal metastasis: current status and recommended guidelines for management. Neurosurgery 1979; 5:726–746.
- Brihaye J, Ectors P, Lemort M, Van Houtte P. The management of spinal epidural metastases. Adv Tech Stand Neurosurg 1988; 16:121–176.
- Patel RK, Slipman CW. Lumbar degenerative disk disease. Emedicine. Accessed March 25, 2008. http://www.emedicine.com/pmr/topic67.htm.
- Deyo RA, Diehl AK. Cancer as a cause of back pain: frequency, clinical presentation, and diagnostic strategies. J Gen Intern Med 1988; 3:230–238.
- Rosen P, Barkin RM, Danzl DF, et al. Emergency Medicine: Concepts and Clinical Practice. 4th ed. St Louis, MO: Mosby; 1998:2100–2102.
- Abbruzzese JL, Abbruzzese MC, Lenzi R, Hess KR, Raber MN. Analysis of a diagnostic strategy for patients with suspected tumors of unknown origin. J Clin Oncol 1995; 13:2094–2103.
- McCarthy EF, Frassica FJ. Pathology of Bone and Joint Disorders: With Clinical and Radiographic Correlation. Philadelphia: WB Saunders; 1998.
- Edelstyn GA, Gillespie PJ, Grebbell FS. The radiological demonstration of osseous metastases. Experimental observations. Clin Radiol 1967; 18:158–162.
- Han LJ, Au-Yong TK, Tong WC, Chu KS, Szeto LT, Wong CP. Comparison of bone single-photon emission tomography and planar imaging in the detection of vertebral metastases in patients with back pain. Eur J Nucl Med 1998; 25:635–638.
- Feun LG, Savaraj N. Detection of occult bone metastasis by MRI scan. J Fla Med Assoc 1990; 77:881–883.
- Citrin DL, Bessent RG, Greig WR. A comparison of the sensitivity and accuracy of the 99TCm-phosphate bone scan and skeletal radiograph in the diagnosis of bone metastases. Clin Radiol 1977; 28:107–117.
- Runge VM, Lee C, Iten AL, Williams NM. Contrast-enhanced magnetic resonance imaging in a spinal epidural tumor model. Invest Radiol 1997; 32:589–595.
- Buranapanitkit B, Lim A, Geater A. Misdiagnosis in vertebral osteomyelitis: problems and factors. J Med Assoc Thai 2001; 84:1743–1750.
- Colmenero JD, Jiménez-Mejías ME, Reguera JM, et al. Tuberculous vertebral osteomyelitis in the new millennium: still a diagnostic and therapeutic challenge. Eur J Clin Microbiol Infect Dis 2004; 23:477–483.
- Waldvogel FA, Medoff G, Swartz MN. Osteomyelitis: a review of clinical features, therapeutic considerations and unusual aspects. 3. Osteomyelitis associated with vascular insufficiency. N Engl J Med 1970; 282:316–322.
- Carragee EJ. Pyogenic vertebral osteomyelitis. J Bone Joint Surg Am 1997; 79:874–880.
- Kapeller P, Fazekas F, Krametter D, et al. Pyogenic infectious spondylitis: clinical, laboratory and MRI features. Eur Neurol 1997; 38:94–98.
- Digby JM, Kersley JB. Pyogenic non-tuberculous spinal infection: an analysis of thirty cases. J Bone Joint Surg Br 1979; 61:47–55.
- Modic MT, Feiglin DH, Piraino DW, et al. Vertebral osteomyelitis: assessment using MR. Radiology 1985; 157:157–166.
- Szypryt EP, Hardy JG, Hinton CE, Worthington BS, Mulholland RC. A comparison between magnetic resonance imaging and scintigraphic bone imaging in the diagnosis of disk space infection in an animal model. Spine 1988; 13:1042–1048.
- Küker W, Mull M, Mayfrank L, Töpper R, Thron A. Epidural spinal infection. Variability of clinical and magnetic resonance imaging findings. Spine 1997; 22:544–551.
- Tung GA, Yim JW, Mermel LA, Philip L, Rogg JM. Spinal epidural abscess: correlation between MRI findings and outcome. Neuroradiology 1999; 41:904–909.
- Sapico FL, Montgomerie JZ. Vertebral osteomyelitis. Infect Dis Clin North Am 1990; 4:539–550.
- Tali ET. Spinal infections. Eur J Radiol 2004; 50:120–133.
- Smith AS, Weinstein MA, Mizushima A, et al. MR imaging characteristics of tuberculous spondylitis vs vertebral osteomyelitis. AJR Am J Roentgenol 1989; 153:399–405.
- Jung NY, Jee WH, Ha KY, Park CK, Byun JY. Discrimination of tuberculous spondylitis from pyogenic spondylitis on MRI. AJR Am J Roentgenol 2004; 182:1405–1410.
- Joseffer SS, Cooper PR. Modern imaging of spinal tuberculosis. J Neurosurg Spine 2005; 2:145–150.
- Pigrau C, Almirante B, Flores X, et al. Spontaneous pyogenic vertebral osteomyelitis and endocarditis: incidence, risk factors, and outcome. Am J Med 2005; 118:1287.
- McHenry MC, Easley KA, Locker GA. Vertebral osteomyelitis: long-term outcome for 253 patients from 7 Cleveland-area hospitals. Clin Infect Dis 2002; 34:1342–1350.
- Danner RL, Hartman BJ. Update on spinal epidural abscess: 35 cases and review of the literature. Rev Infect Dis 1987; 9:265–274.
- Kaufman DM, Kaplan JG, Litman N. Infectious agents in spinal epidural abscesses. Neurology 1980; 30:844–850.
- Rezai AR, Woo HH, Errico TJ, Cooper PR. Contemporary management of spinal osteomyelitis. Neurosurgery 1999; 44:1018–1026.
- Bertino RE, Porter BA, Stimac GK, Tepper SJ. Imaging spinal osteomyelitis and epidural abscess with short TI inversion recovery (STIR). AJNR Am J Neuroradiol 1988; 9:563–564.
- Rigamonti D, Liem L, Sampath P, et al. Spinal epidural abscess: contemporary trends in etiology, evaluation, and management. Surg Neurol 1999; 52:189–197.
- De Vries F, Bracke M, Leufkens HG, Lammers JW, Cooper C, Van Staa TP. Fracture risk with intermittent high-dose oral glucocorticoid therapy. Arthritis Rheum 2007; 56:208–214.
- Cooper C, Atkinson EJ, O’Fallon WM, Melton LJ. Incidence of clinically diagnosed vertebral fractures: a population-based study in Rochester, Minnesota, 1985–1989. J Bone Miner Res 1992; 7:221–227.
- Kim DH, Vaccaro AR. Osteoporotic compression fractures of the spine; current options and considerations for treatment. Spine J 2006; 6:479–487.
- Cohn SH, Abesamis C, Yasumura S, Aloia JF, Zanzi I, Ellis KJ. Comparative skeletal mass and radial bone mineral content in black and white women. Metabolism 1977; 26:171–178.
- Tobias JH, Hutchinson AP, Hunt LP, et al. Use of clinical risk factors to identify postmenopausal women with vertebral fractures. Osteoporos Int 2007; 18:35–43.
- Melton LJ, Kallmes DF. Epidemiology of vertebral fractures: implications for vertebral augmentation. Acad Radiol 2006; 13:538–545.
- Silverman SL. The clinical consequences of vertebral compression fracture. Bone 1992; 13:S27–S31.
- Melton LJ, Kan SH, Frye MA, Wahner HW, O’Fallon WM, Riggs BL. Epidemiology of vertebral fractures in women. Am J Epidemiol 1989; 129:1000–1011.
- Wasnich RD. Vertebral fracture epidemiology. Bone 1996; 18:179S–183S.
- Yamato M, Nishimura G, Kuramochi E, Saiki N, Fujioka M. MR appearance at different ages of osteoporotic compression fractures of the vertebrae. Radiat Med 1998; 16:329–334.
- Einsiedel T, Schmelz A, Arand M, et al. Injuries of the cervical spine in patients with ankylosing spondylitis: experience at two trauma centers. J Neurosurg Spine 2006; 5:33–45.
- Olerud C, Frost A, Bring J. Spinal fractures in patients with ankylosing spondylitis. Eur Spine J 1996; 5:51–55.
- Spangfort EV. The lumbar disk herniation. A computer-aided analysis of 2,504 operations. Acta Orthop Scand Suppl 1972; 142:1–95.
- Kostuik JP, Harrington I, Alexander D, Rand W, Evans D. Cauda equina syndrome and lumbar disk herniation. J Bone Joint Surg Am 1986; 68:386–391.
- Lawton MT, Porter RW, Heiserman JE, Jacobowitz R, Sonntag VK, Dickman CA. Surgical management of spinal epidural hematoma: relationship between surgical timing and neurological outcome. J Neurosurg 1995; 83:1–7.
- Simmons EH, Grobler LJ. Acute spinal epidural hematoma. J Bone Joint Surg Am 1978; 60:395–396.
- Senelick RC, Norwood CW, Cohen GH. “Painless” spinal epidural hematoma during anticoagulant therapy”. Neurology 1976; 26:213–225.
- Watts C, Porto L. Recognizing spontaneous spinal epidural hematoma. Geriatrics 1976; 31:97–99.
KEY POINTS
- A primary tumor or metastasis to the spine tends to cause unremitting back pain that worsens at night and is accompanied by systemic disease and abnormal laboratory findings.
- Infection typically causes focal pain, an elevated erythrocyte sedimentation rate (the most sensitive laboratory test) and C-reactive protein level, and sometimes neurologic signs and symptoms.
- Fractures cause focal pain and should be suspected especially in older white women and patients who take corticosteroids or who have ankylosing spondylitis.
- Plain radiography can help detect fractures, but magnetic resonance imaging is needed to evaluate spinal tumors, soft tissue infections, and epidural abscesses, and to further evaluate neural compression due to fractures.