User login
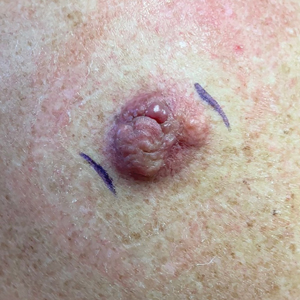
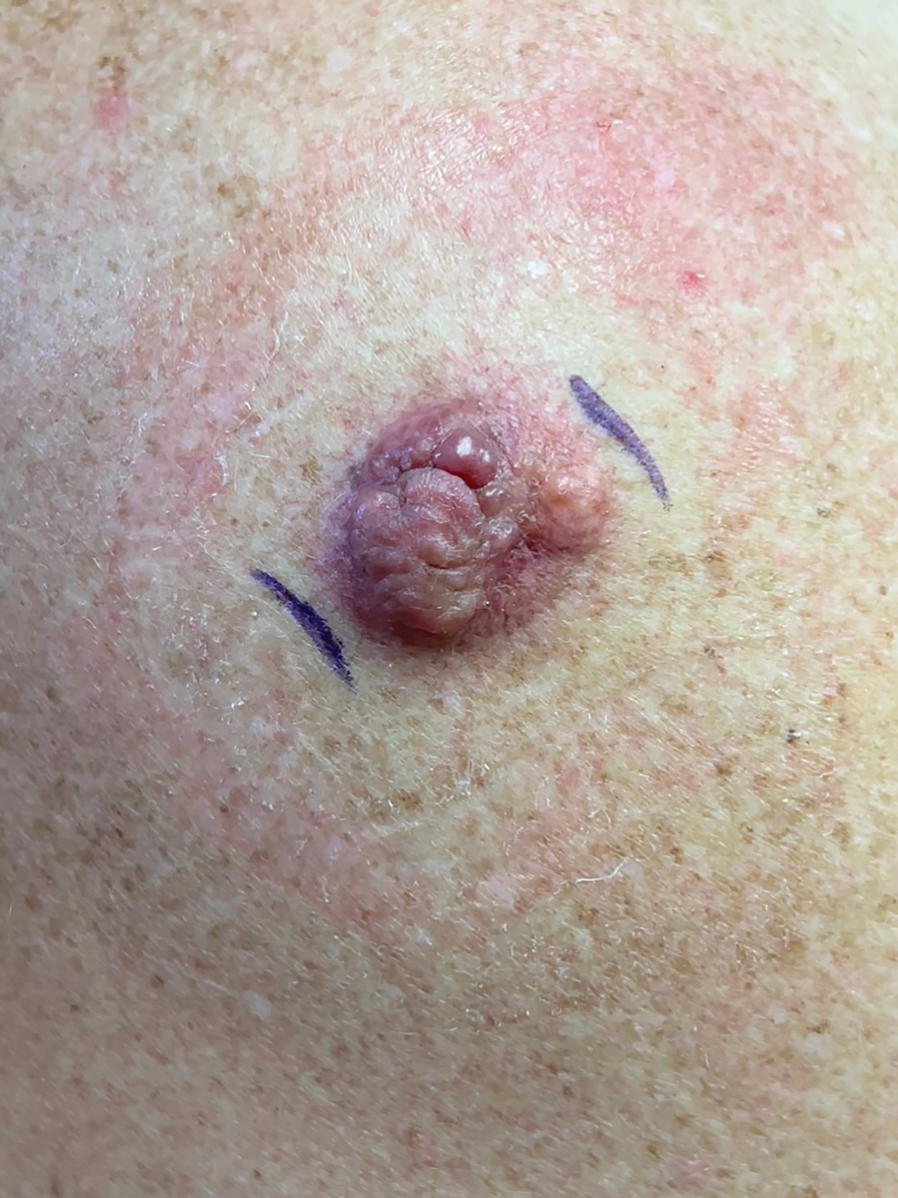
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
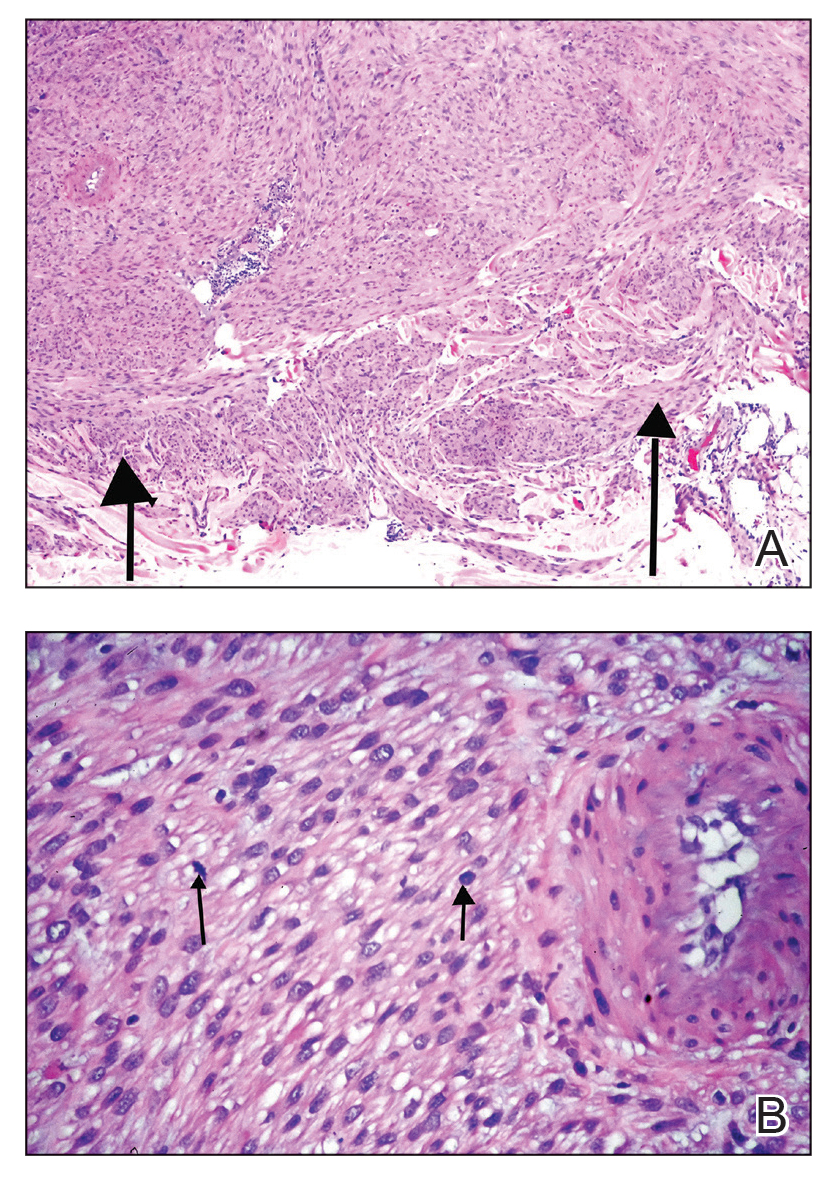
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1
Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1

Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1

Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
A 53-year-old man presented to the dermatology clinic for evaluation of a painful, purpuric, nodular lesion on the left shoulder of 3 months’ duration. The lesion had an irregular surface that was surrounded by an erythematous ring. Biopsy revealed fascicles of eosinophilic cells within the dermis. The nuclei were heterogeneous in size and shape and had blunted ends. Frequent atypia and mitotic figures were observed, and the lesion extended into the subcutis. Immunostaining was positive for desmin and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.
Rare Case of Photodistributed Hyperpigmentation Linked to Kratom Consumption
To the Editor:
Kratom (Mitragyna speciosa) is an evergreen tree native to Southeast Asia.1 Its leaves contain psychoactive compounds including mitragynine and 7-hydroxymitragynine, which exert dose-dependent effects on the central nervous system through opioid and monoaminergic receptors.2,3 At low doses (1–5 g), kratom elicits mild stimulant effects such as increased sociability, alertness, and talkativeness. At high doses (5–15 g), kratom has depressant effects that can provide relief from pain and opioid-withdrawal symptoms.3
Traditionally, kratom has been used in Southeast Asia for recreational and ceremonial purposes, to ease opioid-withdrawal symptoms, and to reduce fatigue from manual labor.4 In the 21st century, availability of kratom expanded to Europe, Australia, and the United States, largely facilitated by widespread dissemination of deceitful marketing and unregulated sales on the internet.1 Although large-scale epidemiologic studies evaluating kratom’s prevalence are scarce, available evidence indicates rising worldwide usage, with a notable increase in kratom-related poison center calls between 2011 and 2017 in the United States.5 In July 2023, kratom made headlines due to the death of a woman in Florida following use of the substance.6
A cross-sectional study revealed that in the United States, kratom typically is used by White individuals for self-treatment of anxiety, depression, pain, and opioid withdrawal.7 However, the potential for severe adverse effects and dependence on kratom can outweigh the benefits.6,8 Reported adverse effects of kratom include tachycardia, hypercholesteremia, liver injury, hallucinations, respiratory depression, seizure, coma, and death.9,10 We present a case of kratom-induced photodistributed hyperpigmentation.
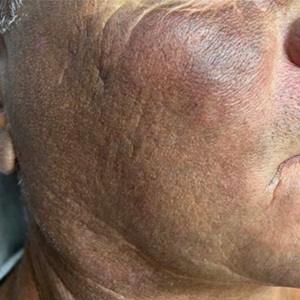
A 63-year-old man presented to the dermatology clinic with diffuse tender, pruritic, hyperpigmented skin lesions that developed over the course of 1 year. The lesions were distributed on sun-exposed areas, including the face, neck, and forearms (Figure 1). The patient reported no other major symptoms, and his health was otherwise unremarkable. He had a medical history of psoriasiform and spongiotic dermatitis consistent with eczema, psoriasis, hypercholesteremia, and hyperlipidemia. The patient was not taking any medications at the time of presentation. He had a family history of plaque psoriasis in his father. Five years prior to the current presentation, the patient was treated with adalimumab for steroid-resistant psoriasis; however, despite initial improvement, he experienced recurrence of scaly erythematous plaques and had discontinued adalimumab the year prior to presentation.
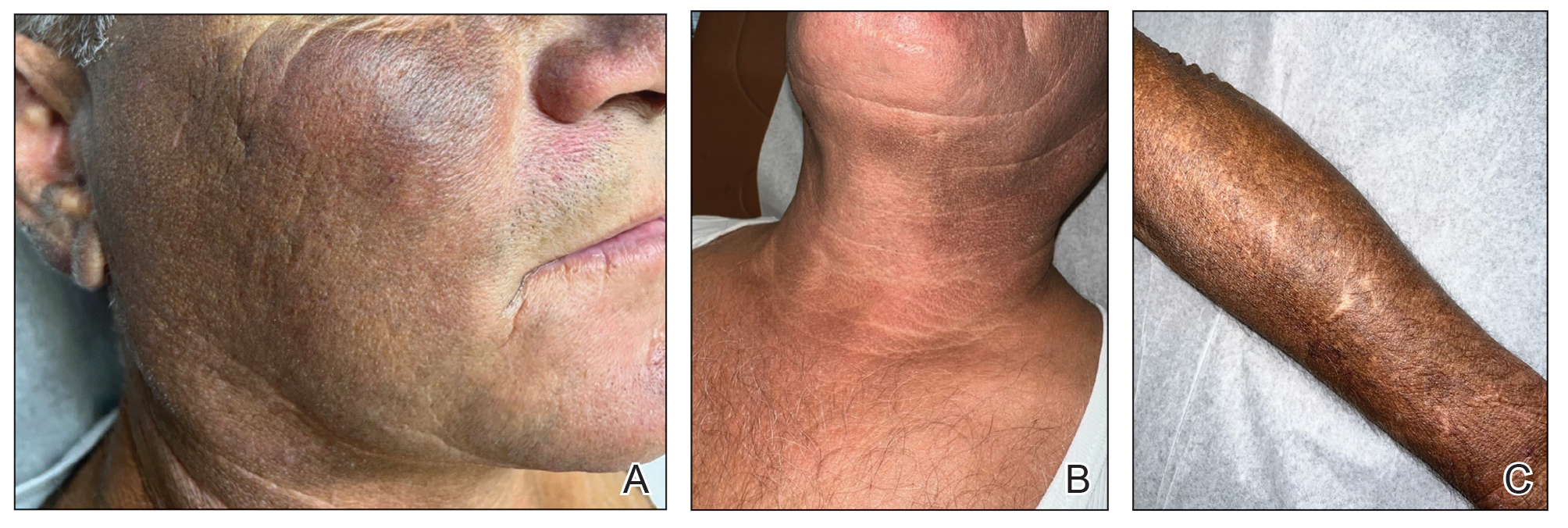
When adalimumab was discontinued, the patient sought alternative treatment for the skin symptoms and began self-administering kratom in an attempt to alleviate associated physical discomfort. He ingested approximately 3 bottles of liquid kratom per day, with each bottle containing 180 mg of mitragynine and less than 8 mg of 7-hydroxymitragynine. Although not scientifically proven, kratom has been colloquially advertised to improve psoriasis.11 The patient reported no other medication use or allergies.
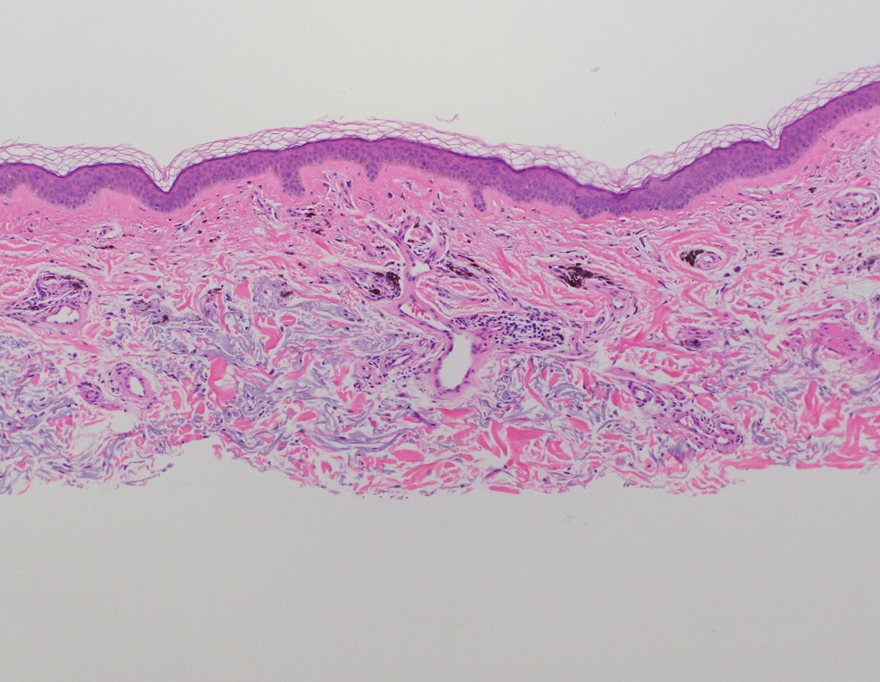
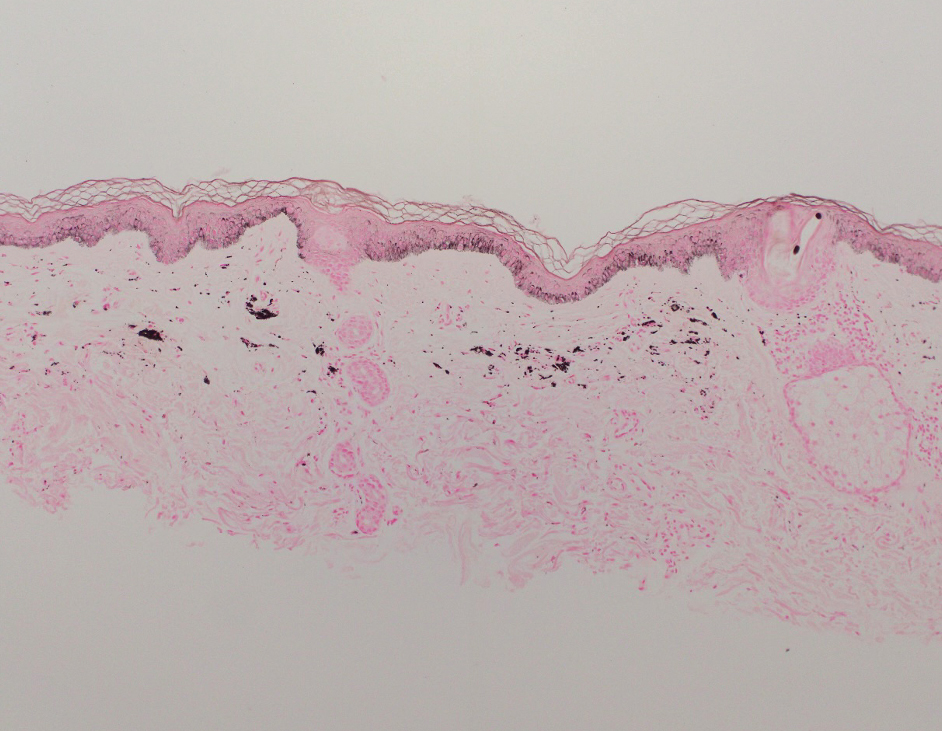
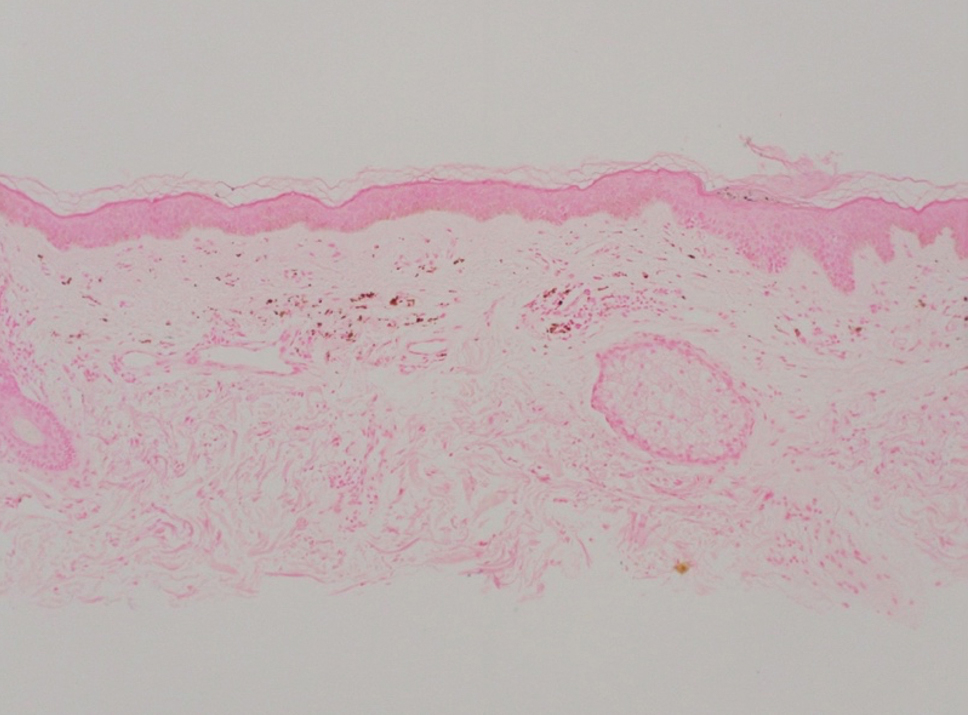
Shave biopsies of hyperpigmented lesions on the right side of the neck, ear, and forearm were performed. Histopathology revealed a sparse superficial, perivascular, lymphocytic infiltrate accompanied by a prominent number of melanophages in the superficial dermis (Figure 2). Special stains further confirmed that the pigment was melanin; the specimens stained positive with Fontana-Masson stain (Figure 3) and negative with an iron stain (Figure 4).
Adalimumab-induced hyperpigmentation was considered. A prior case of adalimumab-induced hyperpigmentation manifested on the face. Histopathology was consistent with a superficial, perivascular, lymphocytic infiltrate with melanophages in the dermis; however, hyperpigmentation was absent in the periorbital area, and affected areas faded 4 months after discontinuation of adalimumab.12 Our patient presented with hyperpigmentation 1 year after adalimumab cessation, and the hyperpigmented areas included the periorbital region. Because of the distinct temporal and clinical features, adalimumab-induced hyperpigmentation was eliminated from the differential diagnosis.
Based on the photodistributed pattern of hyperpigmentation, histopathology, and the temporal relationship between hyperpigmentation onset and kratom usage, a diagnosis of kratom-induced photodistributed hyperpigmentation was made. The patient was advised to discontinue kratom use and use sun protection to prevent further photodamage. The patient subsequently was lost to follow-up.
Kratom alkaloids bind all 3 opioid receptors—μOP, δOP, and κOPs—in a G-protein–biased manner with 7-hydroxymitragynine, the most pharmacologically active alkaloid, exhibiting a higher affinity for μ-opioid receptors.13,14 In human epidermal melanocytes, binding between μ-opioid receptors and β-endorphin, an endogenous opioid, is associated with increased melanin production. This melanogenesis has been linked to hyperpigmentation.15 Given the similarity between kratom alkaloids and β-endorphin in opioid-receptor binding, it is possible that kratom-induced hyperpigmentation may occur through a similar mechanism involving μ-opioid receptors and melanogenesis in epidermal melanocytes. Moreover, some researchers have theorized that sun exposure may result in free radical formation of certain drugs or their metabolites. These free radicals then can interact with cellular DNA, triggering the release of pigmentary mediators and resulting in hyperpigmentation.16 This theory may explain the photodistributed pattern of kratom-induced hyperpigmentation. Further studies are needed to understand the mechanism behind this adverse reaction and its implications for patient treatment.
Literature on kratom-induced hyperpigmentation is limited. Powell et al17 reported a similar case of kratom-induced photodistributed hyperpigmentation—a White man had taken kratom to reduce opioid use and subsequently developed hyperpigmented patches on the arms and face. Moreover, anonymous Reddit users have shared anecdotal reports of hyperpigmentation following kratom use.18
Physicians should be aware of hyperpigmentation as a potential adverse reaction of kratom use as its prevalence increases globally. Further research is warranted to elucidate the mechanism behind this adverse reaction and identify risk factors.
- Prozialeck WC, Avery BA, Boyer EW, et al. Kratom policy: the challenge of balancing therapeutic potential with public safety. Int J Drug Policy. 2019;70:70-77. doi:10.1016/j.drugpo.2019.05.003
- Bergen-Cico D, MacClurg K. Kratom (Mitragyna speciosa) use, addiction potential, and legal status. In: Preedy VR, ed. Neuropathology of Drug Addictions and Substance Misuse. 2016:903-911. doi:10.1016/B978-0-12-800634-4.00089-5
- Warner ML, Kaufman NC, Grundmann O. The pharmacology and toxicology of kratom: from traditional herb to drug of abuse. Int J Legal Med. 2016;130:127-138. doi:10.1007/s00414-015-1279-y
- Transnational Institute. Kratom in Thailand: decriminalisation and community control? May 3, 2011. Accessed August 23, 2024. https://www.tni.org/en/publication/kratom-in-thailand-decriminalisation-and-community-control
- Eastlack SC, Cornett EM, Kaye AD. Kratom—pharmacology, clinical implications, and outlook: a comprehensive review. Pain Ther. 2020;9:55-69. doi:10.1007/s40122-020-00151-x
- Reyes R. Family of Florida mom who died from herbal substance kratom wins $11M suit. New York Post. July 30, 2023. Updated July 31, 2023. Accessed August 23, 2024. https://nypost.com/2023/07/30/family-of-florida-mom-who-died-from-herbal-substance-kratom-wins-11m-suit/
- Garcia-Romeu A, Cox DJ, Smith KE, et al. Kratom (Mitragyna speciosa): user demographics, use patterns, and implications for the opioid epidemic. Drug Alcohol Depend. 2020;208:107849. doi:10.1016/j.drugalcdep.2020.107849
- Mayo Clinic. Kratom: unsafe and ineffective. Accessed August 23, 2024. https://www.mayoclinic.org/healthy-lifestyle/consumer-health/in-depth/kratom/art-20402171
- Sethi R, Hoang N, Ravishankar DA, et al. Kratom (Mitragyna speciosa): friend or foe? Prim Care Companion CNS Disord. 2020;22:19nr02507.
- Eggleston W, Stoppacher R, Suen K, et al. Kratom use and toxicities in the United States. Pharmacother J Hum Pharmacol Drug Ther. 2019;39:775-777. doi:10.1002/phar.2280
- Qrius. 6 benefits of kratom you should know for healthy skin. March 21, 2023. Accessed August 23, 2024. https://qrius.com/6-benefits-of-kratom-you-should-know-for-healthy-skin/
- Blomberg M, Zachariae COC, Grønhøj F. Hyperpigmentation of the face following adalimumab treatment. Acta Derm Venereol. 2009;89:546-547. doi:10.2340/00015555-0697
- Matsumoto K, Hatori Y, Murayama T, et al. Involvement of μ-opioid receptors in antinociception and inhibition of gastrointestinal transit induced by 7-hydroxymitragynine, isolated from Thai herbal medicine Mitragyna speciosa. Eur J Pharmacol. 2006;549:63-70. doi:10.1016/j.ejphar.2006.08.013
- Jentsch MJ, Pippin MM. Kratom. In: StatPearls. StatPearls Publishing; 2023.
- Bigliardi PL, Tobin DJ, Gaveriaux-Ruff C, et al. Opioids and the skin—where do we stand? Exp Dermatol. 2009;18:424-430.
- Boyer M, Katta R, Markus R. Diltiazem-induced photodistributed hyperpigmentation. Dermatol Online J. 2003;9:10. doi:10.5070/D33c97j4z5
- Powell LR, Ryser TJ, Morey GE, et al. Kratom as a novel cause of photodistributed hyperpigmentation. JAAD Case Rep. 2022;28:145-148. doi:10.1016/j.jdcr.2022.07.033
- Haccoon. Skin discoloring? Reddit. June 30, 2019. Accessed August 23, 2024. https://www.reddit.com/r/quittingkratom/comments/c7b1cm/skin_discoloring/
To the Editor:
Kratom (Mitragyna speciosa) is an evergreen tree native to Southeast Asia.1 Its leaves contain psychoactive compounds including mitragynine and 7-hydroxymitragynine, which exert dose-dependent effects on the central nervous system through opioid and monoaminergic receptors.2,3 At low doses (1–5 g), kratom elicits mild stimulant effects such as increased sociability, alertness, and talkativeness. At high doses (5–15 g), kratom has depressant effects that can provide relief from pain and opioid-withdrawal symptoms.3
Traditionally, kratom has been used in Southeast Asia for recreational and ceremonial purposes, to ease opioid-withdrawal symptoms, and to reduce fatigue from manual labor.4 In the 21st century, availability of kratom expanded to Europe, Australia, and the United States, largely facilitated by widespread dissemination of deceitful marketing and unregulated sales on the internet.1 Although large-scale epidemiologic studies evaluating kratom’s prevalence are scarce, available evidence indicates rising worldwide usage, with a notable increase in kratom-related poison center calls between 2011 and 2017 in the United States.5 In July 2023, kratom made headlines due to the death of a woman in Florida following use of the substance.6
A cross-sectional study revealed that in the United States, kratom typically is used by White individuals for self-treatment of anxiety, depression, pain, and opioid withdrawal.7 However, the potential for severe adverse effects and dependence on kratom can outweigh the benefits.6,8 Reported adverse effects of kratom include tachycardia, hypercholesteremia, liver injury, hallucinations, respiratory depression, seizure, coma, and death.9,10 We present a case of kratom-induced photodistributed hyperpigmentation.
A 63-year-old man presented to the dermatology clinic with diffuse tender, pruritic, hyperpigmented skin lesions that developed over the course of 1 year. The lesions were distributed on sun-exposed areas, including the face, neck, and forearms (Figure 1). The patient reported no other major symptoms, and his health was otherwise unremarkable. He had a medical history of psoriasiform and spongiotic dermatitis consistent with eczema, psoriasis, hypercholesteremia, and hyperlipidemia. The patient was not taking any medications at the time of presentation. He had a family history of plaque psoriasis in his father. Five years prior to the current presentation, the patient was treated with adalimumab for steroid-resistant psoriasis; however, despite initial improvement, he experienced recurrence of scaly erythematous plaques and had discontinued adalimumab the year prior to presentation.

When adalimumab was discontinued, the patient sought alternative treatment for the skin symptoms and began self-administering kratom in an attempt to alleviate associated physical discomfort. He ingested approximately 3 bottles of liquid kratom per day, with each bottle containing 180 mg of mitragynine and less than 8 mg of 7-hydroxymitragynine. Although not scientifically proven, kratom has been colloquially advertised to improve psoriasis.11 The patient reported no other medication use or allergies.
Shave biopsies of hyperpigmented lesions on the right side of the neck, ear, and forearm were performed. Histopathology revealed a sparse superficial, perivascular, lymphocytic infiltrate accompanied by a prominent number of melanophages in the superficial dermis (Figure 2). Special stains further confirmed that the pigment was melanin; the specimens stained positive with Fontana-Masson stain (Figure 3) and negative with an iron stain (Figure 4).



Adalimumab-induced hyperpigmentation was considered. A prior case of adalimumab-induced hyperpigmentation manifested on the face. Histopathology was consistent with a superficial, perivascular, lymphocytic infiltrate with melanophages in the dermis; however, hyperpigmentation was absent in the periorbital area, and affected areas faded 4 months after discontinuation of adalimumab.12 Our patient presented with hyperpigmentation 1 year after adalimumab cessation, and the hyperpigmented areas included the periorbital region. Because of the distinct temporal and clinical features, adalimumab-induced hyperpigmentation was eliminated from the differential diagnosis.
Based on the photodistributed pattern of hyperpigmentation, histopathology, and the temporal relationship between hyperpigmentation onset and kratom usage, a diagnosis of kratom-induced photodistributed hyperpigmentation was made. The patient was advised to discontinue kratom use and use sun protection to prevent further photodamage. The patient subsequently was lost to follow-up.
Kratom alkaloids bind all 3 opioid receptors—μOP, δOP, and κOPs—in a G-protein–biased manner with 7-hydroxymitragynine, the most pharmacologically active alkaloid, exhibiting a higher affinity for μ-opioid receptors.13,14 In human epidermal melanocytes, binding between μ-opioid receptors and β-endorphin, an endogenous opioid, is associated with increased melanin production. This melanogenesis has been linked to hyperpigmentation.15 Given the similarity between kratom alkaloids and β-endorphin in opioid-receptor binding, it is possible that kratom-induced hyperpigmentation may occur through a similar mechanism involving μ-opioid receptors and melanogenesis in epidermal melanocytes. Moreover, some researchers have theorized that sun exposure may result in free radical formation of certain drugs or their metabolites. These free radicals then can interact with cellular DNA, triggering the release of pigmentary mediators and resulting in hyperpigmentation.16 This theory may explain the photodistributed pattern of kratom-induced hyperpigmentation. Further studies are needed to understand the mechanism behind this adverse reaction and its implications for patient treatment.
Literature on kratom-induced hyperpigmentation is limited. Powell et al17 reported a similar case of kratom-induced photodistributed hyperpigmentation—a White man had taken kratom to reduce opioid use and subsequently developed hyperpigmented patches on the arms and face. Moreover, anonymous Reddit users have shared anecdotal reports of hyperpigmentation following kratom use.18
Physicians should be aware of hyperpigmentation as a potential adverse reaction of kratom use as its prevalence increases globally. Further research is warranted to elucidate the mechanism behind this adverse reaction and identify risk factors.
To the Editor:
Kratom (Mitragyna speciosa) is an evergreen tree native to Southeast Asia.1 Its leaves contain psychoactive compounds including mitragynine and 7-hydroxymitragynine, which exert dose-dependent effects on the central nervous system through opioid and monoaminergic receptors.2,3 At low doses (1–5 g), kratom elicits mild stimulant effects such as increased sociability, alertness, and talkativeness. At high doses (5–15 g), kratom has depressant effects that can provide relief from pain and opioid-withdrawal symptoms.3
Traditionally, kratom has been used in Southeast Asia for recreational and ceremonial purposes, to ease opioid-withdrawal symptoms, and to reduce fatigue from manual labor.4 In the 21st century, availability of kratom expanded to Europe, Australia, and the United States, largely facilitated by widespread dissemination of deceitful marketing and unregulated sales on the internet.1 Although large-scale epidemiologic studies evaluating kratom’s prevalence are scarce, available evidence indicates rising worldwide usage, with a notable increase in kratom-related poison center calls between 2011 and 2017 in the United States.5 In July 2023, kratom made headlines due to the death of a woman in Florida following use of the substance.6
A cross-sectional study revealed that in the United States, kratom typically is used by White individuals for self-treatment of anxiety, depression, pain, and opioid withdrawal.7 However, the potential for severe adverse effects and dependence on kratom can outweigh the benefits.6,8 Reported adverse effects of kratom include tachycardia, hypercholesteremia, liver injury, hallucinations, respiratory depression, seizure, coma, and death.9,10 We present a case of kratom-induced photodistributed hyperpigmentation.
A 63-year-old man presented to the dermatology clinic with diffuse tender, pruritic, hyperpigmented skin lesions that developed over the course of 1 year. The lesions were distributed on sun-exposed areas, including the face, neck, and forearms (Figure 1). The patient reported no other major symptoms, and his health was otherwise unremarkable. He had a medical history of psoriasiform and spongiotic dermatitis consistent with eczema, psoriasis, hypercholesteremia, and hyperlipidemia. The patient was not taking any medications at the time of presentation. He had a family history of plaque psoriasis in his father. Five years prior to the current presentation, the patient was treated with adalimumab for steroid-resistant psoriasis; however, despite initial improvement, he experienced recurrence of scaly erythematous plaques and had discontinued adalimumab the year prior to presentation.

When adalimumab was discontinued, the patient sought alternative treatment for the skin symptoms and began self-administering kratom in an attempt to alleviate associated physical discomfort. He ingested approximately 3 bottles of liquid kratom per day, with each bottle containing 180 mg of mitragynine and less than 8 mg of 7-hydroxymitragynine. Although not scientifically proven, kratom has been colloquially advertised to improve psoriasis.11 The patient reported no other medication use or allergies.
Shave biopsies of hyperpigmented lesions on the right side of the neck, ear, and forearm were performed. Histopathology revealed a sparse superficial, perivascular, lymphocytic infiltrate accompanied by a prominent number of melanophages in the superficial dermis (Figure 2). Special stains further confirmed that the pigment was melanin; the specimens stained positive with Fontana-Masson stain (Figure 3) and negative with an iron stain (Figure 4).



Adalimumab-induced hyperpigmentation was considered. A prior case of adalimumab-induced hyperpigmentation manifested on the face. Histopathology was consistent with a superficial, perivascular, lymphocytic infiltrate with melanophages in the dermis; however, hyperpigmentation was absent in the periorbital area, and affected areas faded 4 months after discontinuation of adalimumab.12 Our patient presented with hyperpigmentation 1 year after adalimumab cessation, and the hyperpigmented areas included the periorbital region. Because of the distinct temporal and clinical features, adalimumab-induced hyperpigmentation was eliminated from the differential diagnosis.
Based on the photodistributed pattern of hyperpigmentation, histopathology, and the temporal relationship between hyperpigmentation onset and kratom usage, a diagnosis of kratom-induced photodistributed hyperpigmentation was made. The patient was advised to discontinue kratom use and use sun protection to prevent further photodamage. The patient subsequently was lost to follow-up.
Kratom alkaloids bind all 3 opioid receptors—μOP, δOP, and κOPs—in a G-protein–biased manner with 7-hydroxymitragynine, the most pharmacologically active alkaloid, exhibiting a higher affinity for μ-opioid receptors.13,14 In human epidermal melanocytes, binding between μ-opioid receptors and β-endorphin, an endogenous opioid, is associated with increased melanin production. This melanogenesis has been linked to hyperpigmentation.15 Given the similarity between kratom alkaloids and β-endorphin in opioid-receptor binding, it is possible that kratom-induced hyperpigmentation may occur through a similar mechanism involving μ-opioid receptors and melanogenesis in epidermal melanocytes. Moreover, some researchers have theorized that sun exposure may result in free radical formation of certain drugs or their metabolites. These free radicals then can interact with cellular DNA, triggering the release of pigmentary mediators and resulting in hyperpigmentation.16 This theory may explain the photodistributed pattern of kratom-induced hyperpigmentation. Further studies are needed to understand the mechanism behind this adverse reaction and its implications for patient treatment.
Literature on kratom-induced hyperpigmentation is limited. Powell et al17 reported a similar case of kratom-induced photodistributed hyperpigmentation—a White man had taken kratom to reduce opioid use and subsequently developed hyperpigmented patches on the arms and face. Moreover, anonymous Reddit users have shared anecdotal reports of hyperpigmentation following kratom use.18
Physicians should be aware of hyperpigmentation as a potential adverse reaction of kratom use as its prevalence increases globally. Further research is warranted to elucidate the mechanism behind this adverse reaction and identify risk factors.
- Prozialeck WC, Avery BA, Boyer EW, et al. Kratom policy: the challenge of balancing therapeutic potential with public safety. Int J Drug Policy. 2019;70:70-77. doi:10.1016/j.drugpo.2019.05.003
- Bergen-Cico D, MacClurg K. Kratom (Mitragyna speciosa) use, addiction potential, and legal status. In: Preedy VR, ed. Neuropathology of Drug Addictions and Substance Misuse. 2016:903-911. doi:10.1016/B978-0-12-800634-4.00089-5
- Warner ML, Kaufman NC, Grundmann O. The pharmacology and toxicology of kratom: from traditional herb to drug of abuse. Int J Legal Med. 2016;130:127-138. doi:10.1007/s00414-015-1279-y
- Transnational Institute. Kratom in Thailand: decriminalisation and community control? May 3, 2011. Accessed August 23, 2024. https://www.tni.org/en/publication/kratom-in-thailand-decriminalisation-and-community-control
- Eastlack SC, Cornett EM, Kaye AD. Kratom—pharmacology, clinical implications, and outlook: a comprehensive review. Pain Ther. 2020;9:55-69. doi:10.1007/s40122-020-00151-x
- Reyes R. Family of Florida mom who died from herbal substance kratom wins $11M suit. New York Post. July 30, 2023. Updated July 31, 2023. Accessed August 23, 2024. https://nypost.com/2023/07/30/family-of-florida-mom-who-died-from-herbal-substance-kratom-wins-11m-suit/
- Garcia-Romeu A, Cox DJ, Smith KE, et al. Kratom (Mitragyna speciosa): user demographics, use patterns, and implications for the opioid epidemic. Drug Alcohol Depend. 2020;208:107849. doi:10.1016/j.drugalcdep.2020.107849
- Mayo Clinic. Kratom: unsafe and ineffective. Accessed August 23, 2024. https://www.mayoclinic.org/healthy-lifestyle/consumer-health/in-depth/kratom/art-20402171
- Sethi R, Hoang N, Ravishankar DA, et al. Kratom (Mitragyna speciosa): friend or foe? Prim Care Companion CNS Disord. 2020;22:19nr02507.
- Eggleston W, Stoppacher R, Suen K, et al. Kratom use and toxicities in the United States. Pharmacother J Hum Pharmacol Drug Ther. 2019;39:775-777. doi:10.1002/phar.2280
- Qrius. 6 benefits of kratom you should know for healthy skin. March 21, 2023. Accessed August 23, 2024. https://qrius.com/6-benefits-of-kratom-you-should-know-for-healthy-skin/
- Blomberg M, Zachariae COC, Grønhøj F. Hyperpigmentation of the face following adalimumab treatment. Acta Derm Venereol. 2009;89:546-547. doi:10.2340/00015555-0697
- Matsumoto K, Hatori Y, Murayama T, et al. Involvement of μ-opioid receptors in antinociception and inhibition of gastrointestinal transit induced by 7-hydroxymitragynine, isolated from Thai herbal medicine Mitragyna speciosa. Eur J Pharmacol. 2006;549:63-70. doi:10.1016/j.ejphar.2006.08.013
- Jentsch MJ, Pippin MM. Kratom. In: StatPearls. StatPearls Publishing; 2023.
- Bigliardi PL, Tobin DJ, Gaveriaux-Ruff C, et al. Opioids and the skin—where do we stand? Exp Dermatol. 2009;18:424-430.
- Boyer M, Katta R, Markus R. Diltiazem-induced photodistributed hyperpigmentation. Dermatol Online J. 2003;9:10. doi:10.5070/D33c97j4z5
- Powell LR, Ryser TJ, Morey GE, et al. Kratom as a novel cause of photodistributed hyperpigmentation. JAAD Case Rep. 2022;28:145-148. doi:10.1016/j.jdcr.2022.07.033
- Haccoon. Skin discoloring? Reddit. June 30, 2019. Accessed August 23, 2024. https://www.reddit.com/r/quittingkratom/comments/c7b1cm/skin_discoloring/
- Prozialeck WC, Avery BA, Boyer EW, et al. Kratom policy: the challenge of balancing therapeutic potential with public safety. Int J Drug Policy. 2019;70:70-77. doi:10.1016/j.drugpo.2019.05.003
- Bergen-Cico D, MacClurg K. Kratom (Mitragyna speciosa) use, addiction potential, and legal status. In: Preedy VR, ed. Neuropathology of Drug Addictions and Substance Misuse. 2016:903-911. doi:10.1016/B978-0-12-800634-4.00089-5
- Warner ML, Kaufman NC, Grundmann O. The pharmacology and toxicology of kratom: from traditional herb to drug of abuse. Int J Legal Med. 2016;130:127-138. doi:10.1007/s00414-015-1279-y
- Transnational Institute. Kratom in Thailand: decriminalisation and community control? May 3, 2011. Accessed August 23, 2024. https://www.tni.org/en/publication/kratom-in-thailand-decriminalisation-and-community-control
- Eastlack SC, Cornett EM, Kaye AD. Kratom—pharmacology, clinical implications, and outlook: a comprehensive review. Pain Ther. 2020;9:55-69. doi:10.1007/s40122-020-00151-x
- Reyes R. Family of Florida mom who died from herbal substance kratom wins $11M suit. New York Post. July 30, 2023. Updated July 31, 2023. Accessed August 23, 2024. https://nypost.com/2023/07/30/family-of-florida-mom-who-died-from-herbal-substance-kratom-wins-11m-suit/
- Garcia-Romeu A, Cox DJ, Smith KE, et al. Kratom (Mitragyna speciosa): user demographics, use patterns, and implications for the opioid epidemic. Drug Alcohol Depend. 2020;208:107849. doi:10.1016/j.drugalcdep.2020.107849
- Mayo Clinic. Kratom: unsafe and ineffective. Accessed August 23, 2024. https://www.mayoclinic.org/healthy-lifestyle/consumer-health/in-depth/kratom/art-20402171
- Sethi R, Hoang N, Ravishankar DA, et al. Kratom (Mitragyna speciosa): friend or foe? Prim Care Companion CNS Disord. 2020;22:19nr02507.
- Eggleston W, Stoppacher R, Suen K, et al. Kratom use and toxicities in the United States. Pharmacother J Hum Pharmacol Drug Ther. 2019;39:775-777. doi:10.1002/phar.2280
- Qrius. 6 benefits of kratom you should know for healthy skin. March 21, 2023. Accessed August 23, 2024. https://qrius.com/6-benefits-of-kratom-you-should-know-for-healthy-skin/
- Blomberg M, Zachariae COC, Grønhøj F. Hyperpigmentation of the face following adalimumab treatment. Acta Derm Venereol. 2009;89:546-547. doi:10.2340/00015555-0697
- Matsumoto K, Hatori Y, Murayama T, et al. Involvement of μ-opioid receptors in antinociception and inhibition of gastrointestinal transit induced by 7-hydroxymitragynine, isolated from Thai herbal medicine Mitragyna speciosa. Eur J Pharmacol. 2006;549:63-70. doi:10.1016/j.ejphar.2006.08.013
- Jentsch MJ, Pippin MM. Kratom. In: StatPearls. StatPearls Publishing; 2023.
- Bigliardi PL, Tobin DJ, Gaveriaux-Ruff C, et al. Opioids and the skin—where do we stand? Exp Dermatol. 2009;18:424-430.
- Boyer M, Katta R, Markus R. Diltiazem-induced photodistributed hyperpigmentation. Dermatol Online J. 2003;9:10. doi:10.5070/D33c97j4z5
- Powell LR, Ryser TJ, Morey GE, et al. Kratom as a novel cause of photodistributed hyperpigmentation. JAAD Case Rep. 2022;28:145-148. doi:10.1016/j.jdcr.2022.07.033
- Haccoon. Skin discoloring? Reddit. June 30, 2019. Accessed August 23, 2024. https://www.reddit.com/r/quittingkratom/comments/c7b1cm/skin_discoloring/
Practice Points
- Clinicians should be aware of photodistributed hyperpigmentation as a potential adverse effect of kratom usage.
- Kratom-induced photodistributed hyperpigmentation should be suspected in patients with hyperpigmented lesions in sun-exposed areas of the skin following kratom use. A biopsy of lesions should be obtained to confirm the diagnosis.
- Cessation of kratom should be recommended.