User login
Enhancing Veteran Access to Cutting-Edge Treatments: Launching a T Cell Engager Therapy Administration Program
Background
The rise in the number of T-cell engager therapies highlights their importance in modern cancer treatment paradigms. Having recognized the need for, and complexities of, administering these innovative medications to our patients, our team assessed our institution’s capability to provide these therapies to our patients. We identified that our facility was wellequipped for implementation of T-cell engager therapy due to inpatient administration capabilities, an outpatient infusion center, on-hand supportive care medications (tocilizumab), and access to higher levels of care. Key players included medical oncologists, pharmacists, inpatient and infusion nurses, staff physicians, critical care practitioners, and care coordinators.
Clinical Practice Initiative
Barriers identified: education, toxicity concerns, formulary management, and logistics. To overcome these obstacles, comprehensive plans for procurement, hospital admission, monitoring, and training were developed as a facility-specific standard operating procedure (SOP). All available Tcell engager therapies were presented to the formulary committee and received local approval. Physician and pharmacist champions were registered for the associated risk evaluation and mitigation strategies (REMS) programs. Recorded webinars were done to provide education on REMS requirements, medication logistics, and adverse event management.
An admission plan was formulated to outline admission criteria, medication administration, and safety logistics. Order sets created by pharmacists, encompassed pre, post, and as needed medications for cytokine release syndrome and immune effector cell-associated neurotoxicity syndrome. To facilitate safe discharge and meet REMS criteria, patients received wallet cards, dexamethasone and acetaminophen PRNs with detailed instructions for use, and direction for seeking emergency care with consideration of local tocilizumab availability.
Conclusions
Our SOP has enabled administration of six T-cell engager therapies for six diseases. The primary limitation for some of these agents is the need for inpatient monitoring at initiation, which may not be available at smaller centers. Facilities that lack these capabilities could utilize community care or partner with a neighboring Veterans Affairs medical center for initial administration, then transition back for continued treatment. Facilities that lack inpatient oncology nursing could administer the drug in the infusion center followed by admission for monitoring and toxicity management. Our implementation plan serves as a scalable model for improving veteran access to novel therapies.
Background
The rise in the number of T-cell engager therapies highlights their importance in modern cancer treatment paradigms. Having recognized the need for, and complexities of, administering these innovative medications to our patients, our team assessed our institution’s capability to provide these therapies to our patients. We identified that our facility was wellequipped for implementation of T-cell engager therapy due to inpatient administration capabilities, an outpatient infusion center, on-hand supportive care medications (tocilizumab), and access to higher levels of care. Key players included medical oncologists, pharmacists, inpatient and infusion nurses, staff physicians, critical care practitioners, and care coordinators.
Clinical Practice Initiative
Barriers identified: education, toxicity concerns, formulary management, and logistics. To overcome these obstacles, comprehensive plans for procurement, hospital admission, monitoring, and training were developed as a facility-specific standard operating procedure (SOP). All available Tcell engager therapies were presented to the formulary committee and received local approval. Physician and pharmacist champions were registered for the associated risk evaluation and mitigation strategies (REMS) programs. Recorded webinars were done to provide education on REMS requirements, medication logistics, and adverse event management.
An admission plan was formulated to outline admission criteria, medication administration, and safety logistics. Order sets created by pharmacists, encompassed pre, post, and as needed medications for cytokine release syndrome and immune effector cell-associated neurotoxicity syndrome. To facilitate safe discharge and meet REMS criteria, patients received wallet cards, dexamethasone and acetaminophen PRNs with detailed instructions for use, and direction for seeking emergency care with consideration of local tocilizumab availability.
Conclusions
Our SOP has enabled administration of six T-cell engager therapies for six diseases. The primary limitation for some of these agents is the need for inpatient monitoring at initiation, which may not be available at smaller centers. Facilities that lack these capabilities could utilize community care or partner with a neighboring Veterans Affairs medical center for initial administration, then transition back for continued treatment. Facilities that lack inpatient oncology nursing could administer the drug in the infusion center followed by admission for monitoring and toxicity management. Our implementation plan serves as a scalable model for improving veteran access to novel therapies.
Background
The rise in the number of T-cell engager therapies highlights their importance in modern cancer treatment paradigms. Having recognized the need for, and complexities of, administering these innovative medications to our patients, our team assessed our institution’s capability to provide these therapies to our patients. We identified that our facility was wellequipped for implementation of T-cell engager therapy due to inpatient administration capabilities, an outpatient infusion center, on-hand supportive care medications (tocilizumab), and access to higher levels of care. Key players included medical oncologists, pharmacists, inpatient and infusion nurses, staff physicians, critical care practitioners, and care coordinators.
Clinical Practice Initiative
Barriers identified: education, toxicity concerns, formulary management, and logistics. To overcome these obstacles, comprehensive plans for procurement, hospital admission, monitoring, and training were developed as a facility-specific standard operating procedure (SOP). All available Tcell engager therapies were presented to the formulary committee and received local approval. Physician and pharmacist champions were registered for the associated risk evaluation and mitigation strategies (REMS) programs. Recorded webinars were done to provide education on REMS requirements, medication logistics, and adverse event management.
An admission plan was formulated to outline admission criteria, medication administration, and safety logistics. Order sets created by pharmacists, encompassed pre, post, and as needed medications for cytokine release syndrome and immune effector cell-associated neurotoxicity syndrome. To facilitate safe discharge and meet REMS criteria, patients received wallet cards, dexamethasone and acetaminophen PRNs with detailed instructions for use, and direction for seeking emergency care with consideration of local tocilizumab availability.
Conclusions
Our SOP has enabled administration of six T-cell engager therapies for six diseases. The primary limitation for some of these agents is the need for inpatient monitoring at initiation, which may not be available at smaller centers. Facilities that lack these capabilities could utilize community care or partner with a neighboring Veterans Affairs medical center for initial administration, then transition back for continued treatment. Facilities that lack inpatient oncology nursing could administer the drug in the infusion center followed by admission for monitoring and toxicity management. Our implementation plan serves as a scalable model for improving veteran access to novel therapies.
Carboplatin as a Radiosensitizing Agent in Locally Advanced Head and Neck Cancer: Friendly to an Older Veteran Population
Background
The standard of care for locally advanced head and neck squamous cell carcinoma (HNSCC) is combination chemoradiotherapy. Platinum-based chemotherapy is used for radiosensitization and significantly improves locoregional control and survival. Cisplatin is the standard of care; however, many patients are cisplatin-ineligible due to underlying comorbidities. Carboplatin is an alternative chemotherapy in these patients, but efficacy data are lacking. Purpose: To evaluate the efficacy and tolerability of weekly carboplatin concurrent with radiation in veterans with locally advanced HNSCC.
Methods
Our tumor registry was used to identify patients who received platinum-based chemoradiotherapy for stage III-IVB HNSCC at a single center between 2007 to 2017. Patients who received carboplatin were identified. Data including dosing, toxicities, and disease response was collected and analyzed.
Results
A total of 26 patients who received weekly carboplatin were analyzed. All patients were male with an average age of 65. A usual dose of carboplatin AUC 2 was utilized. The average cumulative dose for weekly carboplatin was AUC 12, with most patients (65%) receiving 6 doses or more. The mean number of weekly carboplatin doses held was 0.3. 7 patients (27%) had at least one dose held. 21 (81%) patients showed treatment benefit: 19 (73%) had complete response and 2 (8%) had partial response on first scan following treatment. The four most common toxicities were mucositis (69%), nausea/vomiting (23%), oral thrush (19%), and dermatologic toxicities (19%). The most common toxicities causing dose interruption were fatigue (12%), neutropenia (8%), and thrombocytopenia (8%). Grade 3/4 mucositis was experienced in 6 patients (23%). Other grade 3/4 toxicities included neutropenia (8%), anemia (8%), thrombocytopenia (1%), nephrotoxicity (1%) and nausea (1%).
Conclusions
Carboplatin was both efficacious and well tolerated in our older veteran population. These findings add to the limited body of evidence examining weekly carboplatin in patients with advanced head and neck cancer. While cisplatin remains standard of care, carboplatin may be a reasonable alternative as evidenced in a real-world veteran population.
Background
The standard of care for locally advanced head and neck squamous cell carcinoma (HNSCC) is combination chemoradiotherapy. Platinum-based chemotherapy is used for radiosensitization and significantly improves locoregional control and survival. Cisplatin is the standard of care; however, many patients are cisplatin-ineligible due to underlying comorbidities. Carboplatin is an alternative chemotherapy in these patients, but efficacy data are lacking. Purpose: To evaluate the efficacy and tolerability of weekly carboplatin concurrent with radiation in veterans with locally advanced HNSCC.
Methods
Our tumor registry was used to identify patients who received platinum-based chemoradiotherapy for stage III-IVB HNSCC at a single center between 2007 to 2017. Patients who received carboplatin were identified. Data including dosing, toxicities, and disease response was collected and analyzed.
Results
A total of 26 patients who received weekly carboplatin were analyzed. All patients were male with an average age of 65. A usual dose of carboplatin AUC 2 was utilized. The average cumulative dose for weekly carboplatin was AUC 12, with most patients (65%) receiving 6 doses or more. The mean number of weekly carboplatin doses held was 0.3. 7 patients (27%) had at least one dose held. 21 (81%) patients showed treatment benefit: 19 (73%) had complete response and 2 (8%) had partial response on first scan following treatment. The four most common toxicities were mucositis (69%), nausea/vomiting (23%), oral thrush (19%), and dermatologic toxicities (19%). The most common toxicities causing dose interruption were fatigue (12%), neutropenia (8%), and thrombocytopenia (8%). Grade 3/4 mucositis was experienced in 6 patients (23%). Other grade 3/4 toxicities included neutropenia (8%), anemia (8%), thrombocytopenia (1%), nephrotoxicity (1%) and nausea (1%).
Conclusions
Carboplatin was both efficacious and well tolerated in our older veteran population. These findings add to the limited body of evidence examining weekly carboplatin in patients with advanced head and neck cancer. While cisplatin remains standard of care, carboplatin may be a reasonable alternative as evidenced in a real-world veteran population.
Background
The standard of care for locally advanced head and neck squamous cell carcinoma (HNSCC) is combination chemoradiotherapy. Platinum-based chemotherapy is used for radiosensitization and significantly improves locoregional control and survival. Cisplatin is the standard of care; however, many patients are cisplatin-ineligible due to underlying comorbidities. Carboplatin is an alternative chemotherapy in these patients, but efficacy data are lacking. Purpose: To evaluate the efficacy and tolerability of weekly carboplatin concurrent with radiation in veterans with locally advanced HNSCC.
Methods
Our tumor registry was used to identify patients who received platinum-based chemoradiotherapy for stage III-IVB HNSCC at a single center between 2007 to 2017. Patients who received carboplatin were identified. Data including dosing, toxicities, and disease response was collected and analyzed.
Results
A total of 26 patients who received weekly carboplatin were analyzed. All patients were male with an average age of 65. A usual dose of carboplatin AUC 2 was utilized. The average cumulative dose for weekly carboplatin was AUC 12, with most patients (65%) receiving 6 doses or more. The mean number of weekly carboplatin doses held was 0.3. 7 patients (27%) had at least one dose held. 21 (81%) patients showed treatment benefit: 19 (73%) had complete response and 2 (8%) had partial response on first scan following treatment. The four most common toxicities were mucositis (69%), nausea/vomiting (23%), oral thrush (19%), and dermatologic toxicities (19%). The most common toxicities causing dose interruption were fatigue (12%), neutropenia (8%), and thrombocytopenia (8%). Grade 3/4 mucositis was experienced in 6 patients (23%). Other grade 3/4 toxicities included neutropenia (8%), anemia (8%), thrombocytopenia (1%), nephrotoxicity (1%) and nausea (1%).
Conclusions
Carboplatin was both efficacious and well tolerated in our older veteran population. These findings add to the limited body of evidence examining weekly carboplatin in patients with advanced head and neck cancer. While cisplatin remains standard of care, carboplatin may be a reasonable alternative as evidenced in a real-world veteran population.
How to Make Keeping Up With the Drugs as Easy as Keeping Up With the Kardashians: Implementing a Local Oncology Drug Review Committee
Background
From 2000-2022 there were over 200 new drug and over 500 indication approvals specific to oncology. The rate of approvals has increased exponentially, making it difficult to maintain an up-to-date, standardized practice. Nationally, Veterans Affairs (VA) formulary decisions can take time given a lengthy approval process. Locally, the need was identified to incorporate new drugs and data into practice more rapidly. When bringing requests to the facility Pharmacy and Therapeutics (P&T) Committee, it was recognized that the membership consisting of non-oncology practitioners did not allow for meaningful discussion of utilization. In 2017, a dedicated oncology drug review committee (DRC) comprised of oncology practitioners and a facility formulary representative was created as a P&T workgroup. Purpose: Evaluate and describe the utility of forming a local oncology DRC to incorporate new drugs and data into practice.
Methods
DRC minutes from December 2017 to May 2023 were reviewed. Discussion items were categorized into type of review. Date of local review was compared to national formulary criteria for use publication dates, and date of FDA approval for new drugs or publication date for new data, where applicable. Items were excluded if crucial information was missing from minutes. Descriptive statistics were used.
Results
Over 65 months, 38 meetings were held. Thirty total members include: pharmacists, physicians, fellows, and advanced practice providers. Items reviewed included: 36 new drugs (ND), 36 new indications/data (NI), 14 institutional preferences, 10 new dosage form/biosimilars, 4 drug shortages and 2 others. The median time from ND approval to discussion was 3 months (n= 36, IQR 3-6) and NI from publication was 3 months (n=30, IQR 1-8). Nearly all (34/36, 94%) ND were reviewed prior to national review. Local review was a median of 7 months before national, with 11 drugs currently having no published national criteria for use (n=25, IQR 2-12).
Conclusions
DRC formation has enabled faster incorporation of new drugs/indications into practice. It has also created an appropriate forum for in-depth utilization discussions, pharmacoeconomic stewardship, and sharing of formulary and medication related information. VA Health Systems could consider implementing similar committees to review and implement up-to-date oncology practices.
Background
From 2000-2022 there were over 200 new drug and over 500 indication approvals specific to oncology. The rate of approvals has increased exponentially, making it difficult to maintain an up-to-date, standardized practice. Nationally, Veterans Affairs (VA) formulary decisions can take time given a lengthy approval process. Locally, the need was identified to incorporate new drugs and data into practice more rapidly. When bringing requests to the facility Pharmacy and Therapeutics (P&T) Committee, it was recognized that the membership consisting of non-oncology practitioners did not allow for meaningful discussion of utilization. In 2017, a dedicated oncology drug review committee (DRC) comprised of oncology practitioners and a facility formulary representative was created as a P&T workgroup. Purpose: Evaluate and describe the utility of forming a local oncology DRC to incorporate new drugs and data into practice.
Methods
DRC minutes from December 2017 to May 2023 were reviewed. Discussion items were categorized into type of review. Date of local review was compared to national formulary criteria for use publication dates, and date of FDA approval for new drugs or publication date for new data, where applicable. Items were excluded if crucial information was missing from minutes. Descriptive statistics were used.
Results
Over 65 months, 38 meetings were held. Thirty total members include: pharmacists, physicians, fellows, and advanced practice providers. Items reviewed included: 36 new drugs (ND), 36 new indications/data (NI), 14 institutional preferences, 10 new dosage form/biosimilars, 4 drug shortages and 2 others. The median time from ND approval to discussion was 3 months (n= 36, IQR 3-6) and NI from publication was 3 months (n=30, IQR 1-8). Nearly all (34/36, 94%) ND were reviewed prior to national review. Local review was a median of 7 months before national, with 11 drugs currently having no published national criteria for use (n=25, IQR 2-12).
Conclusions
DRC formation has enabled faster incorporation of new drugs/indications into practice. It has also created an appropriate forum for in-depth utilization discussions, pharmacoeconomic stewardship, and sharing of formulary and medication related information. VA Health Systems could consider implementing similar committees to review and implement up-to-date oncology practices.
Background
From 2000-2022 there were over 200 new drug and over 500 indication approvals specific to oncology. The rate of approvals has increased exponentially, making it difficult to maintain an up-to-date, standardized practice. Nationally, Veterans Affairs (VA) formulary decisions can take time given a lengthy approval process. Locally, the need was identified to incorporate new drugs and data into practice more rapidly. When bringing requests to the facility Pharmacy and Therapeutics (P&T) Committee, it was recognized that the membership consisting of non-oncology practitioners did not allow for meaningful discussion of utilization. In 2017, a dedicated oncology drug review committee (DRC) comprised of oncology practitioners and a facility formulary representative was created as a P&T workgroup. Purpose: Evaluate and describe the utility of forming a local oncology DRC to incorporate new drugs and data into practice.
Methods
DRC minutes from December 2017 to May 2023 were reviewed. Discussion items were categorized into type of review. Date of local review was compared to national formulary criteria for use publication dates, and date of FDA approval for new drugs or publication date for new data, where applicable. Items were excluded if crucial information was missing from minutes. Descriptive statistics were used.
Results
Over 65 months, 38 meetings were held. Thirty total members include: pharmacists, physicians, fellows, and advanced practice providers. Items reviewed included: 36 new drugs (ND), 36 new indications/data (NI), 14 institutional preferences, 10 new dosage form/biosimilars, 4 drug shortages and 2 others. The median time from ND approval to discussion was 3 months (n= 36, IQR 3-6) and NI from publication was 3 months (n=30, IQR 1-8). Nearly all (34/36, 94%) ND were reviewed prior to national review. Local review was a median of 7 months before national, with 11 drugs currently having no published national criteria for use (n=25, IQR 2-12).
Conclusions
DRC formation has enabled faster incorporation of new drugs/indications into practice. It has also created an appropriate forum for in-depth utilization discussions, pharmacoeconomic stewardship, and sharing of formulary and medication related information. VA Health Systems could consider implementing similar committees to review and implement up-to-date oncology practices.
Acquired Factor VIII Deficiency Presenting as Compartment Syndrome
Compartment syndrome occurs when the interstitial tissue pressures within a confined space are elevated to a level at which the arterial perfusion is diminished. Multiple etiologies exist and can be extrinsic (a cast that is too tight or prolonged compression on a limb), iatrogenic (aggressive resuscitation, drug infiltration, arterial puncture, or a spontaneous bleed from anticoagulation), and traumatic (fracture, snake envenomation, circumferential burn, or electrocution). If the compartments are not released, irreversible changes happen to the cells, including nerve and muscle death.1 Definitive management of this emergency requires prompt fasciotomy to decompress the compartment(s).1-3
Case Presentation
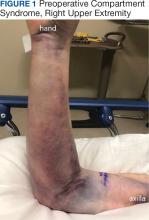
A 76-year-old right-handed woman with a history of chronic obstructive pulmonary disease, hypertension, and hyperlipidemia presented to the emergency department with 2 days of extensive right upper extremity ecchymosis and severe pain that was localized to her forearm (Figure 1). She was taking low-dose aspirin (81 mg/d) for left subclavian stenosis and over-the-counter ginkgo biloba. Leading up to the presentation, the patient was able to perform routine household chores, including yard work, cleaning, and taking care of her cats. Wrist and elbow X-rays were negative for a fracture. An upper extremity ultrasound found no venous occlusion. A computed tomography (CT) angiogram of her arm and chest found diffuse edema around the right elbow and forearm without pulmonary or right upper extremity emboli, fractures, hematoma, abscess, or air in the tissues.
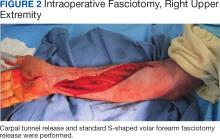
The plastic surgery service was consulted. The patient was found to have a very tense forearm and pain to passive digital extension. The 2-point discrimination and pulses were intact. The patient was diagnosed with compartment syndrome based on the examination alone and gave consent for an emergent forearm and hand fasciotomy. A carpal tunnel release and a standard S-shaped volar forearm fasciotomy release were performed, which provided immediate decompression (Figure 2). The rest of the hand and extremity were soft. Edematous, healthy flexor muscle belly was identified without a hematoma. Most of the forearm wound was left open because the skin could not be reapproximated. Oxidized regenerated cellulose (Surgicel) was placed around the wound edges and the muscle was covered with a nonadherent dressing. Hemoglobin on admission was 12.9 g/dL(reference range, 12 to 16 g/dL). Kidney function was within normal limits. The rest of the complete blood count was unremarkable. Postoperative hemoglobin was 8.6 g/dL. Over the next several days, the patient's skin edges and muscle bellies continued to slowly bleed, and her hemoglobin fell to 5.6 g/dL by postoperative Day 2. The bleeding was managed with topical oxidized regenerated cellulose, thrombin spray, a hemostatic dressing made with kaolin (QuikClot), and a transfusion of 2 units of packed red blood cells.
A hematology consultation was requested. The patient was noted to have an elevated partial thromboplastin time (PTT) since admission measuring between 39.9 to 61.7 seconds (reference range, 26.2 to 37.2 seconds) and a normal prothrombin time test with an international normalized ratio. A PTT measured 17 months prior to admission was within the normal range. She reported no personal or family history of bleeding disorders. Until recently, she had never had easy bruisability. She reported no history of heavy menses or epistaxis. The patient had no children and had never been pregnant. She had tolerated an exploratory laparotomy 40 years prior to admission without bleeding complications and had never required blood transfusions before. A PTT 1:1 mixing study revealed incomplete correction. Subsequent workup included factor VIII (FVIII) activity, factor IX activity, factor XI activity, von Willebrand factor antigen, ristocetin cofactor assay, and von Willebrand factor multimers. FVIII activity was severely reduced at 7.8% (reference, > 54%) with a positive Bethesda assay of 300 to 400 Bodansky units (BU), indicating a strong FVIII inhibitor was present and establishing a diagnosis of acquired hemophilia A. Further workup for secondary causes of acquired hemophilia A including abdominal and pelvic CT, serum protein electrophoresis, and serum free light chains, were negative. She was started on prednisone 1 mg/kg daily and rituximab 375 mg/m2. Her hemoglobin stabilized, and she required no further blood transfusions.
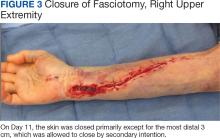
The patient underwent wound closure on postoperative Day 11. At the time of the second surgery, there was still no improvement in her FVIII levels or PTT; therefore, 70 mcg/kg of recombinant coagulation-activated FVII was given just before surgery with no bleeding complications. The skin was closed primarily except for the most distal 3 cm (Figure 3). Due to concerns regarding further bleeding with skin graft, the remaining wound was allowed to close by secondary intention. As a precaution, the wound was covered with oxidized regenerated cellulose and thrombin spray. The patient continued to progress postoperatively without bleeding complications or a need for additional transfusions. She was seen by the hand therapist before and after the second surgery to help with edema management and joint mobility. She completed 4 weekly doses of 375 mg/m² rituximab and prednisone was tapered by 10 mg weekly.
Three weeks after starting treatment, her PTT normalized, and her FVIII increased to 33.7%. The Bethesda assay remained high at 198 BU, although it was lower than at admission. She was discharged home with dressing changes and monthly follow-up appointments. The wounds were fully closed at her 3-month appointment when she proudly demonstrated full digital extension and flexion into her palm.
Discussion
Forearm compartment syndrome is most often caused by fractures—distal radius in adults and supracondylar in children.2 This case initially presented as a diagnostic puzzle to the emergency department due to the patient’s lucid review of several days of nontraumatic injury.
The clinical hallmarks of compartment syndrome are the 5 Ps: pain, pallor, paresthesia, paralysis, and pulselessness. Patients will describe the pain as out of proportion to the nature of the injury; the compartments will be tense and swollen, they will have pain to passive muscle stretch, and sensation will progressively diminish. Distal pulses are the last to go, and permanent tissue damage can still occur when pulses are present.1
Compartment Syndrome
Compartment syndrome is generally a clinical diagnosis; however, in patients who are sedated or uncooperative, or if the clinical findings are equivocal, the examination can be supplemented with intercompartmental pressures using an arterial line transducer system.2 In general, a tissue pressure of 30 mm Hg or a 20- to 30-mm Hg difference between the diastolic and compartment pressures are indications for fasciotomy.1 The hand is treated with an open carpal tunnel release, interosseous muscle release through 2 dorsal hand incisions, and thenar and hypothenar muscle release. The forearm is treated through a curved volar incision that usually decompresses the dorsal compartment, as it did in our patient. If pressures are still high in the forearm, a longitudinal dorsal incision over the mobile wad is necessary. Wounds can be closed primarily days later, left open to close by secondary intention, or reconstructed with skin grafts.2 In our patient, compartment syndrome was isolated to her forearm and the carpal tunnel release was performed prophylactically since it did not add significant time or morbidity to the surgery.
Nontraumatic upper extremity compartment syndrome is rare. A 2021 review of acute nontraumatic upper extremity compartment syndrome found a bleeding disorder as the etiology in 3 cases published in the literature between 1993 and 2016.4 One of these cases was secondary to a known diagnosis of hemophilia A in a teenager.5 Ogrodnik and colleagues described a spontaneous hand hematoma secondary to previously undiagnosed acquired hemophilia A and Waldenström macroglobulinemia.4 Ilyas and colleagues described a spontaneous hematoma in the forearm dorsal compartment in a 67-year-old woman, which presented as compartment syndrome and elevated PTT and led to a diagnosis of acquired FVIII inhibitor. The authors recommended prompt hematology consultation to coordinate treatment once this diagnosis issuspected.6 Compartment syndrome also has been found to develop slowly over weeks in patients with acquired FVIII deficiency, suggesting a high index of suspicion and frequent examinations are needed when patients with known acquired hemophilia A present with a painful extremity.7
Nontraumatic compartment syndrome in the lower extremity in patients with previously undiagnosed acquired hemophilia A has also been described in the literature.8-11 Case reports describe the delay in diagnosis as the patients were originally seen by clinicians for lower extremity pain and swelling within days of presenting to the emergency room with compartment syndrome. Persistent bleeding and abnormal laboratory results prompted further tests and examinations.8,9,11 This underscores the need to be suspicious of this unusual pathology without a history of trauma.
Acquired Hemophilia A
Acquired hemophilia A is an autoimmune disease most often found in older individuals, with a mean age of approximately 70 years.12 It is caused by the spontaneous production of neutralizing immunoglobin autoantibodies that target endogenous FVIII. Many cases are idiopathic; however, up to 50% of cases are associated with underlying autoimmunity, malignancy (especially lymphoproliferative disorders), or pregnancy. It often presents as bleeding that is subcutaneous or in the gastrointestinal system, muscle, retroperitoneal space, or genitourinary system. Unlike congenital hemophilia A, joint bleeding is rare.13
The diagnosis is suspected with an isolated elevated PTT in the absence of other coagulation abnormalities. A 1:1 mixing study will typically show incomplete correction, which suggests the presence of an inhibitor. FVIII activity is reduced, and the FVIII inhibitor is confirmed with the Bethesda assay. Clinically active bleeding is treated with bypassing agents such as recombinant coagulation-activated FVII, activated prothrombin complex concentrates such as anti-inhibitor coagulant complex (FEIBA), or recombinant porcine FVIII.12,14 Not all patients require hemostatic treatment, but close monitoring, education, recognition, and immediate treatment, if needed, are indicated.13 Immunosuppressive therapy (corticosteroids, rituximab, and/or cyclophosphamide) is prescribed to eradicate the antibodies and induce remission.12
Conclusions
An older woman without a preceding trauma was diagnosed with an unusual case of acute compartment syndrome in the forearm. No hematoma was found, but muscle and skin bleeding plus an elevated PTT prompted a hematology workup, and, ultimately, the diagnosis of FVIII inhibitor secondary to acquired hemophilia A.
While a nontraumatic cause of compartment syndrome is rare, it should be considered in differential diagnosis for clinicians who see hand and upper extremity emergencies. An isolated elevated PTT in a patient with a bleed should raise suspicions and trigger immediate further evaluation. Once suspected, multidisciplinary treatment is indicated for immediate and long-term successful outcomes.
Acknowledgments
This manuscript is the result of work supported withresources and the use of facilities at the North Florida/South Georgia Veterans Health System, Gainesville, Florida.
1. Leversedge FJ, Moore TJ, Peterson BC, Seiler JG 3rd. Compartment syndrome of the upper extremity. J Hand Surg Am. 2011;36:544-559. doi:10.1016/j.jhsa.2010.12.008
2. Kalyani BS, Fisher BE, Roberts CS, Giannoudis PV. Compartment syndrome of the forearm: a systematic review. J Hand Surg Am. 2011;36:535-543. doi:10.1016/j.jhsa.2010.12.007
3. Steadman W, Wu R, Hamilton AT, Richardson MD, Wall CJ. Review article: a comprehensive review of unusual causes of acute limb compartment syndrome. Emerg Med Australas. 2022;34:871-876. doi:10.1111/1742-6723.14098
4. Ogrodnik J, Oliver JD, Cani D, Boczar D, Huayllani MT, Restrepo DJ, et al. Clinical case of acute non-traumatic hand compartment syndrome and systematic review for the upper extremity. Hand (N Y). 2021;16:285-291. doi:10.1177/1558944719856106
5. Kim J, Zelken J, Sacks JM. Case report. Spontaneous forearm compartment syndrome in a boy with hemophilia a: a therapeutic dilemma. Eplasty. 2013:13:e16.
6. Ilyas AM, Wisbeck JM, Shaffer GW, Thoder JJ. Upper extremity compartment syndrome secondary to acquired factor VIII inhibitor. A case report. J Bone Joint Surg Am. 2005;87:1606-1608. doi:10.2106/JBJS.C.01720
7. Adeclat GJ, Hayes M, Amick M, Kahan J, Halim A. Acute forearm compartment syndrome in the setting of acquired hemophilia A. Case Reports Plast Surg Hand Surg. 2022;9:140-144. doi:10.1080/23320885.2022.2071274
8. Abudaqqa RY, Arun KP, Mas AJA, Abushaaban FA. Acute atraumatic compartment syndrome of the thigh due to acquired coagulopathy disorder: a case report in known healthy patient. J Orthop Case Rep. 2021;11:59-62. doi:10.13107/jocr.2021.v11.i08.2366
9. Alidoost M, Conte GA, Chaudry R, Nahum K, Marchesani D. A unique presentation of spontaneous compartment syndrome due to acquired hemophilia A and associated malignancy: case report and literature review. World J Oncol. 2020;11:72-75. doi:10.14740/wjon1260
10. Jentzsch T, Brand-Staufer B, Schäfer FP, Wanner GA, Simmen H-P. Illustrated operative management of spontaneous bleeding and compartment syndrome of the lower extremity in a patient with acquired hemophilia A: a case report. J Med Case Rep. 2014;8:132. doi:10.1186/1752-1947-8-132
11. Pham TV, Sorenson CA, Nable JV. Acquired factor VIII deficiency presenting with compartment syndrome. Am J Emerg Med. 2014;32:195.e1-2. doi:10.1016/j.ajem.2013.09.022
12. Tiede A, Zieger B, Lisman T. Acquired bleeding disorders. Haemophilia. 2022;28(suppl 4):68-76. doi:10.1111/hae.14548
13. Kruse-Jarres R, Kempton CL, Baudo F, Collins PW, Knoebl P, Leissinger CA, et al. Acquired hemophilia A: updated review of evidence and treatment guidance. Am J Hematol. 2017;92:695-705. doi:10.1002/ajh.24777
14. Ilkhchoui Y, Koshkin E, Windsor JJ, Petersen TR, Charles M, Pack JD. Perioperative management of acquired hemophilia A: a case report and review of literature. Anesth Pain Med. 2013;4:e11906. doi:10.5812/aapm.11906
Compartment syndrome occurs when the interstitial tissue pressures within a confined space are elevated to a level at which the arterial perfusion is diminished. Multiple etiologies exist and can be extrinsic (a cast that is too tight or prolonged compression on a limb), iatrogenic (aggressive resuscitation, drug infiltration, arterial puncture, or a spontaneous bleed from anticoagulation), and traumatic (fracture, snake envenomation, circumferential burn, or electrocution). If the compartments are not released, irreversible changes happen to the cells, including nerve and muscle death.1 Definitive management of this emergency requires prompt fasciotomy to decompress the compartment(s).1-3
Case Presentation
A 76-year-old right-handed woman with a history of chronic obstructive pulmonary disease, hypertension, and hyperlipidemia presented to the emergency department with 2 days of extensive right upper extremity ecchymosis and severe pain that was localized to her forearm (Figure 1). She was taking low-dose aspirin (81 mg/d) for left subclavian stenosis and over-the-counter ginkgo biloba. Leading up to the presentation, the patient was able to perform routine household chores, including yard work, cleaning, and taking care of her cats. Wrist and elbow X-rays were negative for a fracture. An upper extremity ultrasound found no venous occlusion. A computed tomography (CT) angiogram of her arm and chest found diffuse edema around the right elbow and forearm without pulmonary or right upper extremity emboli, fractures, hematoma, abscess, or air in the tissues.
The plastic surgery service was consulted. The patient was found to have a very tense forearm and pain to passive digital extension. The 2-point discrimination and pulses were intact. The patient was diagnosed with compartment syndrome based on the examination alone and gave consent for an emergent forearm and hand fasciotomy. A carpal tunnel release and a standard S-shaped volar forearm fasciotomy release were performed, which provided immediate decompression (Figure 2). The rest of the hand and extremity were soft. Edematous, healthy flexor muscle belly was identified without a hematoma. Most of the forearm wound was left open because the skin could not be reapproximated. Oxidized regenerated cellulose (Surgicel) was placed around the wound edges and the muscle was covered with a nonadherent dressing. Hemoglobin on admission was 12.9 g/dL(reference range, 12 to 16 g/dL). Kidney function was within normal limits. The rest of the complete blood count was unremarkable. Postoperative hemoglobin was 8.6 g/dL. Over the next several days, the patient's skin edges and muscle bellies continued to slowly bleed, and her hemoglobin fell to 5.6 g/dL by postoperative Day 2. The bleeding was managed with topical oxidized regenerated cellulose, thrombin spray, a hemostatic dressing made with kaolin (QuikClot), and a transfusion of 2 units of packed red blood cells.
A hematology consultation was requested. The patient was noted to have an elevated partial thromboplastin time (PTT) since admission measuring between 39.9 to 61.7 seconds (reference range, 26.2 to 37.2 seconds) and a normal prothrombin time test with an international normalized ratio. A PTT measured 17 months prior to admission was within the normal range. She reported no personal or family history of bleeding disorders. Until recently, she had never had easy bruisability. She reported no history of heavy menses or epistaxis. The patient had no children and had never been pregnant. She had tolerated an exploratory laparotomy 40 years prior to admission without bleeding complications and had never required blood transfusions before. A PTT 1:1 mixing study revealed incomplete correction. Subsequent workup included factor VIII (FVIII) activity, factor IX activity, factor XI activity, von Willebrand factor antigen, ristocetin cofactor assay, and von Willebrand factor multimers. FVIII activity was severely reduced at 7.8% (reference, > 54%) with a positive Bethesda assay of 300 to 400 Bodansky units (BU), indicating a strong FVIII inhibitor was present and establishing a diagnosis of acquired hemophilia A. Further workup for secondary causes of acquired hemophilia A including abdominal and pelvic CT, serum protein electrophoresis, and serum free light chains, were negative. She was started on prednisone 1 mg/kg daily and rituximab 375 mg/m2. Her hemoglobin stabilized, and she required no further blood transfusions.
The patient underwent wound closure on postoperative Day 11. At the time of the second surgery, there was still no improvement in her FVIII levels or PTT; therefore, 70 mcg/kg of recombinant coagulation-activated FVII was given just before surgery with no bleeding complications. The skin was closed primarily except for the most distal 3 cm (Figure 3). Due to concerns regarding further bleeding with skin graft, the remaining wound was allowed to close by secondary intention. As a precaution, the wound was covered with oxidized regenerated cellulose and thrombin spray. The patient continued to progress postoperatively without bleeding complications or a need for additional transfusions. She was seen by the hand therapist before and after the second surgery to help with edema management and joint mobility. She completed 4 weekly doses of 375 mg/m² rituximab and prednisone was tapered by 10 mg weekly.
Three weeks after starting treatment, her PTT normalized, and her FVIII increased to 33.7%. The Bethesda assay remained high at 198 BU, although it was lower than at admission. She was discharged home with dressing changes and monthly follow-up appointments. The wounds were fully closed at her 3-month appointment when she proudly demonstrated full digital extension and flexion into her palm.
Discussion
Forearm compartment syndrome is most often caused by fractures—distal radius in adults and supracondylar in children.2 This case initially presented as a diagnostic puzzle to the emergency department due to the patient’s lucid review of several days of nontraumatic injury.
The clinical hallmarks of compartment syndrome are the 5 Ps: pain, pallor, paresthesia, paralysis, and pulselessness. Patients will describe the pain as out of proportion to the nature of the injury; the compartments will be tense and swollen, they will have pain to passive muscle stretch, and sensation will progressively diminish. Distal pulses are the last to go, and permanent tissue damage can still occur when pulses are present.1
Compartment Syndrome
Compartment syndrome is generally a clinical diagnosis; however, in patients who are sedated or uncooperative, or if the clinical findings are equivocal, the examination can be supplemented with intercompartmental pressures using an arterial line transducer system.2 In general, a tissue pressure of 30 mm Hg or a 20- to 30-mm Hg difference between the diastolic and compartment pressures are indications for fasciotomy.1 The hand is treated with an open carpal tunnel release, interosseous muscle release through 2 dorsal hand incisions, and thenar and hypothenar muscle release. The forearm is treated through a curved volar incision that usually decompresses the dorsal compartment, as it did in our patient. If pressures are still high in the forearm, a longitudinal dorsal incision over the mobile wad is necessary. Wounds can be closed primarily days later, left open to close by secondary intention, or reconstructed with skin grafts.2 In our patient, compartment syndrome was isolated to her forearm and the carpal tunnel release was performed prophylactically since it did not add significant time or morbidity to the surgery.
Nontraumatic upper extremity compartment syndrome is rare. A 2021 review of acute nontraumatic upper extremity compartment syndrome found a bleeding disorder as the etiology in 3 cases published in the literature between 1993 and 2016.4 One of these cases was secondary to a known diagnosis of hemophilia A in a teenager.5 Ogrodnik and colleagues described a spontaneous hand hematoma secondary to previously undiagnosed acquired hemophilia A and Waldenström macroglobulinemia.4 Ilyas and colleagues described a spontaneous hematoma in the forearm dorsal compartment in a 67-year-old woman, which presented as compartment syndrome and elevated PTT and led to a diagnosis of acquired FVIII inhibitor. The authors recommended prompt hematology consultation to coordinate treatment once this diagnosis issuspected.6 Compartment syndrome also has been found to develop slowly over weeks in patients with acquired FVIII deficiency, suggesting a high index of suspicion and frequent examinations are needed when patients with known acquired hemophilia A present with a painful extremity.7
Nontraumatic compartment syndrome in the lower extremity in patients with previously undiagnosed acquired hemophilia A has also been described in the literature.8-11 Case reports describe the delay in diagnosis as the patients were originally seen by clinicians for lower extremity pain and swelling within days of presenting to the emergency room with compartment syndrome. Persistent bleeding and abnormal laboratory results prompted further tests and examinations.8,9,11 This underscores the need to be suspicious of this unusual pathology without a history of trauma.
Acquired Hemophilia A
Acquired hemophilia A is an autoimmune disease most often found in older individuals, with a mean age of approximately 70 years.12 It is caused by the spontaneous production of neutralizing immunoglobin autoantibodies that target endogenous FVIII. Many cases are idiopathic; however, up to 50% of cases are associated with underlying autoimmunity, malignancy (especially lymphoproliferative disorders), or pregnancy. It often presents as bleeding that is subcutaneous or in the gastrointestinal system, muscle, retroperitoneal space, or genitourinary system. Unlike congenital hemophilia A, joint bleeding is rare.13
The diagnosis is suspected with an isolated elevated PTT in the absence of other coagulation abnormalities. A 1:1 mixing study will typically show incomplete correction, which suggests the presence of an inhibitor. FVIII activity is reduced, and the FVIII inhibitor is confirmed with the Bethesda assay. Clinically active bleeding is treated with bypassing agents such as recombinant coagulation-activated FVII, activated prothrombin complex concentrates such as anti-inhibitor coagulant complex (FEIBA), or recombinant porcine FVIII.12,14 Not all patients require hemostatic treatment, but close monitoring, education, recognition, and immediate treatment, if needed, are indicated.13 Immunosuppressive therapy (corticosteroids, rituximab, and/or cyclophosphamide) is prescribed to eradicate the antibodies and induce remission.12
Conclusions
An older woman without a preceding trauma was diagnosed with an unusual case of acute compartment syndrome in the forearm. No hematoma was found, but muscle and skin bleeding plus an elevated PTT prompted a hematology workup, and, ultimately, the diagnosis of FVIII inhibitor secondary to acquired hemophilia A.
While a nontraumatic cause of compartment syndrome is rare, it should be considered in differential diagnosis for clinicians who see hand and upper extremity emergencies. An isolated elevated PTT in a patient with a bleed should raise suspicions and trigger immediate further evaluation. Once suspected, multidisciplinary treatment is indicated for immediate and long-term successful outcomes.
Acknowledgments
This manuscript is the result of work supported withresources and the use of facilities at the North Florida/South Georgia Veterans Health System, Gainesville, Florida.
Compartment syndrome occurs when the interstitial tissue pressures within a confined space are elevated to a level at which the arterial perfusion is diminished. Multiple etiologies exist and can be extrinsic (a cast that is too tight or prolonged compression on a limb), iatrogenic (aggressive resuscitation, drug infiltration, arterial puncture, or a spontaneous bleed from anticoagulation), and traumatic (fracture, snake envenomation, circumferential burn, or electrocution). If the compartments are not released, irreversible changes happen to the cells, including nerve and muscle death.1 Definitive management of this emergency requires prompt fasciotomy to decompress the compartment(s).1-3
Case Presentation
A 76-year-old right-handed woman with a history of chronic obstructive pulmonary disease, hypertension, and hyperlipidemia presented to the emergency department with 2 days of extensive right upper extremity ecchymosis and severe pain that was localized to her forearm (Figure 1). She was taking low-dose aspirin (81 mg/d) for left subclavian stenosis and over-the-counter ginkgo biloba. Leading up to the presentation, the patient was able to perform routine household chores, including yard work, cleaning, and taking care of her cats. Wrist and elbow X-rays were negative for a fracture. An upper extremity ultrasound found no venous occlusion. A computed tomography (CT) angiogram of her arm and chest found diffuse edema around the right elbow and forearm without pulmonary or right upper extremity emboli, fractures, hematoma, abscess, or air in the tissues.
The plastic surgery service was consulted. The patient was found to have a very tense forearm and pain to passive digital extension. The 2-point discrimination and pulses were intact. The patient was diagnosed with compartment syndrome based on the examination alone and gave consent for an emergent forearm and hand fasciotomy. A carpal tunnel release and a standard S-shaped volar forearm fasciotomy release were performed, which provided immediate decompression (Figure 2). The rest of the hand and extremity were soft. Edematous, healthy flexor muscle belly was identified without a hematoma. Most of the forearm wound was left open because the skin could not be reapproximated. Oxidized regenerated cellulose (Surgicel) was placed around the wound edges and the muscle was covered with a nonadherent dressing. Hemoglobin on admission was 12.9 g/dL(reference range, 12 to 16 g/dL). Kidney function was within normal limits. The rest of the complete blood count was unremarkable. Postoperative hemoglobin was 8.6 g/dL. Over the next several days, the patient's skin edges and muscle bellies continued to slowly bleed, and her hemoglobin fell to 5.6 g/dL by postoperative Day 2. The bleeding was managed with topical oxidized regenerated cellulose, thrombin spray, a hemostatic dressing made with kaolin (QuikClot), and a transfusion of 2 units of packed red blood cells.
A hematology consultation was requested. The patient was noted to have an elevated partial thromboplastin time (PTT) since admission measuring between 39.9 to 61.7 seconds (reference range, 26.2 to 37.2 seconds) and a normal prothrombin time test with an international normalized ratio. A PTT measured 17 months prior to admission was within the normal range. She reported no personal or family history of bleeding disorders. Until recently, she had never had easy bruisability. She reported no history of heavy menses or epistaxis. The patient had no children and had never been pregnant. She had tolerated an exploratory laparotomy 40 years prior to admission without bleeding complications and had never required blood transfusions before. A PTT 1:1 mixing study revealed incomplete correction. Subsequent workup included factor VIII (FVIII) activity, factor IX activity, factor XI activity, von Willebrand factor antigen, ristocetin cofactor assay, and von Willebrand factor multimers. FVIII activity was severely reduced at 7.8% (reference, > 54%) with a positive Bethesda assay of 300 to 400 Bodansky units (BU), indicating a strong FVIII inhibitor was present and establishing a diagnosis of acquired hemophilia A. Further workup for secondary causes of acquired hemophilia A including abdominal and pelvic CT, serum protein electrophoresis, and serum free light chains, were negative. She was started on prednisone 1 mg/kg daily and rituximab 375 mg/m2. Her hemoglobin stabilized, and she required no further blood transfusions.
The patient underwent wound closure on postoperative Day 11. At the time of the second surgery, there was still no improvement in her FVIII levels or PTT; therefore, 70 mcg/kg of recombinant coagulation-activated FVII was given just before surgery with no bleeding complications. The skin was closed primarily except for the most distal 3 cm (Figure 3). Due to concerns regarding further bleeding with skin graft, the remaining wound was allowed to close by secondary intention. As a precaution, the wound was covered with oxidized regenerated cellulose and thrombin spray. The patient continued to progress postoperatively without bleeding complications or a need for additional transfusions. She was seen by the hand therapist before and after the second surgery to help with edema management and joint mobility. She completed 4 weekly doses of 375 mg/m² rituximab and prednisone was tapered by 10 mg weekly.
Three weeks after starting treatment, her PTT normalized, and her FVIII increased to 33.7%. The Bethesda assay remained high at 198 BU, although it was lower than at admission. She was discharged home with dressing changes and monthly follow-up appointments. The wounds were fully closed at her 3-month appointment when she proudly demonstrated full digital extension and flexion into her palm.
Discussion
Forearm compartment syndrome is most often caused by fractures—distal radius in adults and supracondylar in children.2 This case initially presented as a diagnostic puzzle to the emergency department due to the patient’s lucid review of several days of nontraumatic injury.
The clinical hallmarks of compartment syndrome are the 5 Ps: pain, pallor, paresthesia, paralysis, and pulselessness. Patients will describe the pain as out of proportion to the nature of the injury; the compartments will be tense and swollen, they will have pain to passive muscle stretch, and sensation will progressively diminish. Distal pulses are the last to go, and permanent tissue damage can still occur when pulses are present.1
Compartment Syndrome
Compartment syndrome is generally a clinical diagnosis; however, in patients who are sedated or uncooperative, or if the clinical findings are equivocal, the examination can be supplemented with intercompartmental pressures using an arterial line transducer system.2 In general, a tissue pressure of 30 mm Hg or a 20- to 30-mm Hg difference between the diastolic and compartment pressures are indications for fasciotomy.1 The hand is treated with an open carpal tunnel release, interosseous muscle release through 2 dorsal hand incisions, and thenar and hypothenar muscle release. The forearm is treated through a curved volar incision that usually decompresses the dorsal compartment, as it did in our patient. If pressures are still high in the forearm, a longitudinal dorsal incision over the mobile wad is necessary. Wounds can be closed primarily days later, left open to close by secondary intention, or reconstructed with skin grafts.2 In our patient, compartment syndrome was isolated to her forearm and the carpal tunnel release was performed prophylactically since it did not add significant time or morbidity to the surgery.
Nontraumatic upper extremity compartment syndrome is rare. A 2021 review of acute nontraumatic upper extremity compartment syndrome found a bleeding disorder as the etiology in 3 cases published in the literature between 1993 and 2016.4 One of these cases was secondary to a known diagnosis of hemophilia A in a teenager.5 Ogrodnik and colleagues described a spontaneous hand hematoma secondary to previously undiagnosed acquired hemophilia A and Waldenström macroglobulinemia.4 Ilyas and colleagues described a spontaneous hematoma in the forearm dorsal compartment in a 67-year-old woman, which presented as compartment syndrome and elevated PTT and led to a diagnosis of acquired FVIII inhibitor. The authors recommended prompt hematology consultation to coordinate treatment once this diagnosis issuspected.6 Compartment syndrome also has been found to develop slowly over weeks in patients with acquired FVIII deficiency, suggesting a high index of suspicion and frequent examinations are needed when patients with known acquired hemophilia A present with a painful extremity.7
Nontraumatic compartment syndrome in the lower extremity in patients with previously undiagnosed acquired hemophilia A has also been described in the literature.8-11 Case reports describe the delay in diagnosis as the patients were originally seen by clinicians for lower extremity pain and swelling within days of presenting to the emergency room with compartment syndrome. Persistent bleeding and abnormal laboratory results prompted further tests and examinations.8,9,11 This underscores the need to be suspicious of this unusual pathology without a history of trauma.
Acquired Hemophilia A
Acquired hemophilia A is an autoimmune disease most often found in older individuals, with a mean age of approximately 70 years.12 It is caused by the spontaneous production of neutralizing immunoglobin autoantibodies that target endogenous FVIII. Many cases are idiopathic; however, up to 50% of cases are associated with underlying autoimmunity, malignancy (especially lymphoproliferative disorders), or pregnancy. It often presents as bleeding that is subcutaneous or in the gastrointestinal system, muscle, retroperitoneal space, or genitourinary system. Unlike congenital hemophilia A, joint bleeding is rare.13
The diagnosis is suspected with an isolated elevated PTT in the absence of other coagulation abnormalities. A 1:1 mixing study will typically show incomplete correction, which suggests the presence of an inhibitor. FVIII activity is reduced, and the FVIII inhibitor is confirmed with the Bethesda assay. Clinically active bleeding is treated with bypassing agents such as recombinant coagulation-activated FVII, activated prothrombin complex concentrates such as anti-inhibitor coagulant complex (FEIBA), or recombinant porcine FVIII.12,14 Not all patients require hemostatic treatment, but close monitoring, education, recognition, and immediate treatment, if needed, are indicated.13 Immunosuppressive therapy (corticosteroids, rituximab, and/or cyclophosphamide) is prescribed to eradicate the antibodies and induce remission.12
Conclusions
An older woman without a preceding trauma was diagnosed with an unusual case of acute compartment syndrome in the forearm. No hematoma was found, but muscle and skin bleeding plus an elevated PTT prompted a hematology workup, and, ultimately, the diagnosis of FVIII inhibitor secondary to acquired hemophilia A.
While a nontraumatic cause of compartment syndrome is rare, it should be considered in differential diagnosis for clinicians who see hand and upper extremity emergencies. An isolated elevated PTT in a patient with a bleed should raise suspicions and trigger immediate further evaluation. Once suspected, multidisciplinary treatment is indicated for immediate and long-term successful outcomes.
Acknowledgments
This manuscript is the result of work supported withresources and the use of facilities at the North Florida/South Georgia Veterans Health System, Gainesville, Florida.
1. Leversedge FJ, Moore TJ, Peterson BC, Seiler JG 3rd. Compartment syndrome of the upper extremity. J Hand Surg Am. 2011;36:544-559. doi:10.1016/j.jhsa.2010.12.008
2. Kalyani BS, Fisher BE, Roberts CS, Giannoudis PV. Compartment syndrome of the forearm: a systematic review. J Hand Surg Am. 2011;36:535-543. doi:10.1016/j.jhsa.2010.12.007
3. Steadman W, Wu R, Hamilton AT, Richardson MD, Wall CJ. Review article: a comprehensive review of unusual causes of acute limb compartment syndrome. Emerg Med Australas. 2022;34:871-876. doi:10.1111/1742-6723.14098
4. Ogrodnik J, Oliver JD, Cani D, Boczar D, Huayllani MT, Restrepo DJ, et al. Clinical case of acute non-traumatic hand compartment syndrome and systematic review for the upper extremity. Hand (N Y). 2021;16:285-291. doi:10.1177/1558944719856106
5. Kim J, Zelken J, Sacks JM. Case report. Spontaneous forearm compartment syndrome in a boy with hemophilia a: a therapeutic dilemma. Eplasty. 2013:13:e16.
6. Ilyas AM, Wisbeck JM, Shaffer GW, Thoder JJ. Upper extremity compartment syndrome secondary to acquired factor VIII inhibitor. A case report. J Bone Joint Surg Am. 2005;87:1606-1608. doi:10.2106/JBJS.C.01720
7. Adeclat GJ, Hayes M, Amick M, Kahan J, Halim A. Acute forearm compartment syndrome in the setting of acquired hemophilia A. Case Reports Plast Surg Hand Surg. 2022;9:140-144. doi:10.1080/23320885.2022.2071274
8. Abudaqqa RY, Arun KP, Mas AJA, Abushaaban FA. Acute atraumatic compartment syndrome of the thigh due to acquired coagulopathy disorder: a case report in known healthy patient. J Orthop Case Rep. 2021;11:59-62. doi:10.13107/jocr.2021.v11.i08.2366
9. Alidoost M, Conte GA, Chaudry R, Nahum K, Marchesani D. A unique presentation of spontaneous compartment syndrome due to acquired hemophilia A and associated malignancy: case report and literature review. World J Oncol. 2020;11:72-75. doi:10.14740/wjon1260
10. Jentzsch T, Brand-Staufer B, Schäfer FP, Wanner GA, Simmen H-P. Illustrated operative management of spontaneous bleeding and compartment syndrome of the lower extremity in a patient with acquired hemophilia A: a case report. J Med Case Rep. 2014;8:132. doi:10.1186/1752-1947-8-132
11. Pham TV, Sorenson CA, Nable JV. Acquired factor VIII deficiency presenting with compartment syndrome. Am J Emerg Med. 2014;32:195.e1-2. doi:10.1016/j.ajem.2013.09.022
12. Tiede A, Zieger B, Lisman T. Acquired bleeding disorders. Haemophilia. 2022;28(suppl 4):68-76. doi:10.1111/hae.14548
13. Kruse-Jarres R, Kempton CL, Baudo F, Collins PW, Knoebl P, Leissinger CA, et al. Acquired hemophilia A: updated review of evidence and treatment guidance. Am J Hematol. 2017;92:695-705. doi:10.1002/ajh.24777
14. Ilkhchoui Y, Koshkin E, Windsor JJ, Petersen TR, Charles M, Pack JD. Perioperative management of acquired hemophilia A: a case report and review of literature. Anesth Pain Med. 2013;4:e11906. doi:10.5812/aapm.11906
1. Leversedge FJ, Moore TJ, Peterson BC, Seiler JG 3rd. Compartment syndrome of the upper extremity. J Hand Surg Am. 2011;36:544-559. doi:10.1016/j.jhsa.2010.12.008
2. Kalyani BS, Fisher BE, Roberts CS, Giannoudis PV. Compartment syndrome of the forearm: a systematic review. J Hand Surg Am. 2011;36:535-543. doi:10.1016/j.jhsa.2010.12.007
3. Steadman W, Wu R, Hamilton AT, Richardson MD, Wall CJ. Review article: a comprehensive review of unusual causes of acute limb compartment syndrome. Emerg Med Australas. 2022;34:871-876. doi:10.1111/1742-6723.14098
4. Ogrodnik J, Oliver JD, Cani D, Boczar D, Huayllani MT, Restrepo DJ, et al. Clinical case of acute non-traumatic hand compartment syndrome and systematic review for the upper extremity. Hand (N Y). 2021;16:285-291. doi:10.1177/1558944719856106
5. Kim J, Zelken J, Sacks JM. Case report. Spontaneous forearm compartment syndrome in a boy with hemophilia a: a therapeutic dilemma. Eplasty. 2013:13:e16.
6. Ilyas AM, Wisbeck JM, Shaffer GW, Thoder JJ. Upper extremity compartment syndrome secondary to acquired factor VIII inhibitor. A case report. J Bone Joint Surg Am. 2005;87:1606-1608. doi:10.2106/JBJS.C.01720
7. Adeclat GJ, Hayes M, Amick M, Kahan J, Halim A. Acute forearm compartment syndrome in the setting of acquired hemophilia A. Case Reports Plast Surg Hand Surg. 2022;9:140-144. doi:10.1080/23320885.2022.2071274
8. Abudaqqa RY, Arun KP, Mas AJA, Abushaaban FA. Acute atraumatic compartment syndrome of the thigh due to acquired coagulopathy disorder: a case report in known healthy patient. J Orthop Case Rep. 2021;11:59-62. doi:10.13107/jocr.2021.v11.i08.2366
9. Alidoost M, Conte GA, Chaudry R, Nahum K, Marchesani D. A unique presentation of spontaneous compartment syndrome due to acquired hemophilia A and associated malignancy: case report and literature review. World J Oncol. 2020;11:72-75. doi:10.14740/wjon1260
10. Jentzsch T, Brand-Staufer B, Schäfer FP, Wanner GA, Simmen H-P. Illustrated operative management of spontaneous bleeding and compartment syndrome of the lower extremity in a patient with acquired hemophilia A: a case report. J Med Case Rep. 2014;8:132. doi:10.1186/1752-1947-8-132
11. Pham TV, Sorenson CA, Nable JV. Acquired factor VIII deficiency presenting with compartment syndrome. Am J Emerg Med. 2014;32:195.e1-2. doi:10.1016/j.ajem.2013.09.022
12. Tiede A, Zieger B, Lisman T. Acquired bleeding disorders. Haemophilia. 2022;28(suppl 4):68-76. doi:10.1111/hae.14548
13. Kruse-Jarres R, Kempton CL, Baudo F, Collins PW, Knoebl P, Leissinger CA, et al. Acquired hemophilia A: updated review of evidence and treatment guidance. Am J Hematol. 2017;92:695-705. doi:10.1002/ajh.24777
14. Ilkhchoui Y, Koshkin E, Windsor JJ, Petersen TR, Charles M, Pack JD. Perioperative management of acquired hemophilia A: a case report and review of literature. Anesth Pain Med. 2013;4:e11906. doi:10.5812/aapm.11906
Revision of a Massive Transfusion Protocol to Allow for Verbal Orders
PURPOSE
To improve the time to release of blood products for patients with severe or life-threatening bleeding.
BACKGROUND
Exsanguination, and the resultant coagulopathy, is the number one cause of trauma-related death. Massive transfusion protocols (MTP) improve mortality by shortening the time to transfusion and correcting coagulopathy. Many patients do not meet criteria for massive transfusion (> 10 units RBCs in 24 hours), yet present with clinical instability and require rapid release (RR) of uncrossmatched blood. A quality improvement initiative was performed to identify barriers to the MTP/RR protocol at a single institution.
METHODS/DATA
A multidisciplinary subcommittee was formed to evaluate the safety and efficacy of the current MTP/RR process. Timed mock-MTP/RR trials were conducted to identify areas of delay with a goal to achieve a blood to bedside (B2B) time of under 10 minutes.
RESULTS
Timed mock-MTP/RR trials were conducted, which revealed a baseline B2B time of approximately 30 minutes. We identified problems and categorized them in terms of ordering (phase 1) and processing (phase 2). We found significant delays in phase 1. Reasons for delay were varied and included difficulty logging into the computer, staff unavailable to place orders (involved in resuscitation efforts), orders entered incorrectly, etc. Once orders were received, the blood bank could process them quickly in phase 2. Using root cause analysis, we discovered a critical step was to remove the barrier of electronic ordering. For this, a new process was developed in which the blood bank could accept verbal orders to release uncrossmatched blood during a medical emergency. Over the course of one year, a new policy for MTP/RR was drafted, an education training video was recorded, informational flyers were printed, and training drills were conducted. A repeat mock-MTP/RR scenario was performed after the change showing the B2B time was reduced by 90% from pre-intervention values to under 3 minutes. Since implementation, no new safety signals have been received, and the staff have reported improved satisfaction with the MTP/RR process.
IMPLICATIONS
A critical piece of any MTP/RR is the immediate availability of blood. Allowing verbal orders for blood products reduced time to transfusion by 90%. Through multidisciplinary effort, safe and efficient release of uncrossmatched blood products for nontraumatic massive transfusion can be achieved.
PURPOSE
To improve the time to release of blood products for patients with severe or life-threatening bleeding.
BACKGROUND
Exsanguination, and the resultant coagulopathy, is the number one cause of trauma-related death. Massive transfusion protocols (MTP) improve mortality by shortening the time to transfusion and correcting coagulopathy. Many patients do not meet criteria for massive transfusion (> 10 units RBCs in 24 hours), yet present with clinical instability and require rapid release (RR) of uncrossmatched blood. A quality improvement initiative was performed to identify barriers to the MTP/RR protocol at a single institution.
METHODS/DATA
A multidisciplinary subcommittee was formed to evaluate the safety and efficacy of the current MTP/RR process. Timed mock-MTP/RR trials were conducted to identify areas of delay with a goal to achieve a blood to bedside (B2B) time of under 10 minutes.
RESULTS
Timed mock-MTP/RR trials were conducted, which revealed a baseline B2B time of approximately 30 minutes. We identified problems and categorized them in terms of ordering (phase 1) and processing (phase 2). We found significant delays in phase 1. Reasons for delay were varied and included difficulty logging into the computer, staff unavailable to place orders (involved in resuscitation efforts), orders entered incorrectly, etc. Once orders were received, the blood bank could process them quickly in phase 2. Using root cause analysis, we discovered a critical step was to remove the barrier of electronic ordering. For this, a new process was developed in which the blood bank could accept verbal orders to release uncrossmatched blood during a medical emergency. Over the course of one year, a new policy for MTP/RR was drafted, an education training video was recorded, informational flyers were printed, and training drills were conducted. A repeat mock-MTP/RR scenario was performed after the change showing the B2B time was reduced by 90% from pre-intervention values to under 3 minutes. Since implementation, no new safety signals have been received, and the staff have reported improved satisfaction with the MTP/RR process.
IMPLICATIONS
A critical piece of any MTP/RR is the immediate availability of blood. Allowing verbal orders for blood products reduced time to transfusion by 90%. Through multidisciplinary effort, safe and efficient release of uncrossmatched blood products for nontraumatic massive transfusion can be achieved.
PURPOSE
To improve the time to release of blood products for patients with severe or life-threatening bleeding.
BACKGROUND
Exsanguination, and the resultant coagulopathy, is the number one cause of trauma-related death. Massive transfusion protocols (MTP) improve mortality by shortening the time to transfusion and correcting coagulopathy. Many patients do not meet criteria for massive transfusion (> 10 units RBCs in 24 hours), yet present with clinical instability and require rapid release (RR) of uncrossmatched blood. A quality improvement initiative was performed to identify barriers to the MTP/RR protocol at a single institution.
METHODS/DATA
A multidisciplinary subcommittee was formed to evaluate the safety and efficacy of the current MTP/RR process. Timed mock-MTP/RR trials were conducted to identify areas of delay with a goal to achieve a blood to bedside (B2B) time of under 10 minutes.
RESULTS
Timed mock-MTP/RR trials were conducted, which revealed a baseline B2B time of approximately 30 minutes. We identified problems and categorized them in terms of ordering (phase 1) and processing (phase 2). We found significant delays in phase 1. Reasons for delay were varied and included difficulty logging into the computer, staff unavailable to place orders (involved in resuscitation efforts), orders entered incorrectly, etc. Once orders were received, the blood bank could process them quickly in phase 2. Using root cause analysis, we discovered a critical step was to remove the barrier of electronic ordering. For this, a new process was developed in which the blood bank could accept verbal orders to release uncrossmatched blood during a medical emergency. Over the course of one year, a new policy for MTP/RR was drafted, an education training video was recorded, informational flyers were printed, and training drills were conducted. A repeat mock-MTP/RR scenario was performed after the change showing the B2B time was reduced by 90% from pre-intervention values to under 3 minutes. Since implementation, no new safety signals have been received, and the staff have reported improved satisfaction with the MTP/RR process.
IMPLICATIONS
A critical piece of any MTP/RR is the immediate availability of blood. Allowing verbal orders for blood products reduced time to transfusion by 90%. Through multidisciplinary effort, safe and efficient release of uncrossmatched blood products for nontraumatic massive transfusion can be achieved.
Implementation of Electronic Antineoplastic Ordering Templates in a Veterans Health System
BACKGROUND: Antineoplastic ordering is high in both complexity and risk for medication errors. Joint Commission and institutional policy recommends computerized prescriber order entry and verification by a pharmacist as a best practice for patient safety. The use of ordering templates minimizes the risk of errors and ensures appropriate supportive care is being provided. The Veterans Affairs electronic medical record does not feature an antineoplastic ordering component; historically, templates have been on paper. The oncology, pharmacy, and clinical informatics departments in our health system formed a team to create electronic antineoplastic ordering templates.
PURPOSE: To evaluate the impact of electronic antineoplastic ordering templates on pharmacy and infusion clinic efficiency and pharmacist interventions.
METHODS: Ordering templates, which included standard dosing and supportive care medications, were developed and activated for prescribers in phases over the course of two years. Immunotherapy and oral oncolytic templates were activated initially, followed by full implementation of the majority of intravenous (IV) ordering templates. Percent of electronic antineoplastic orders, pharmacy processing time, time to first drug delivery, and number of pharmacist encounters and interventions were documented daily for four weeks after initial implementation and for ten weeks after full implementation. Means were compared using unpaired t tests.
RESULTS: After initial implementation, the percentage of electronic antineoplastic orders increased from 0% to 100% for oral antineoplastics and from 0% to 39% for IV orders. After full implementation, IV orders increased to 69% in weeks 1-5 and 96% in weeks 6-10. Mean pharmacy processing time for supportive care medications was 35 minutes initially. This increased briefly after full implementation (weeks 1-5), then decreased to 17 minutes in weeks 6-10 (p<0.01). Delivery of the first medication to the infusion center decreased by 31 minutes at week ten (<0.01). Mean daily pharmacist encounters increased by 28%(<0.01) and documented interventions increased by 22% (<0.01).
IMPLICATIONS: Implementation of electronic antineoplastic ordering templates increased provider order entry, pharmacy and infusion clinic efficiency and pharmacist interventions. These outcomes may translate to improved patient safety and patient access to quality care. Templates developed can serve as a model for other health systems to implement electronic antineoplastic ordering.
BACKGROUND: Antineoplastic ordering is high in both complexity and risk for medication errors. Joint Commission and institutional policy recommends computerized prescriber order entry and verification by a pharmacist as a best practice for patient safety. The use of ordering templates minimizes the risk of errors and ensures appropriate supportive care is being provided. The Veterans Affairs electronic medical record does not feature an antineoplastic ordering component; historically, templates have been on paper. The oncology, pharmacy, and clinical informatics departments in our health system formed a team to create electronic antineoplastic ordering templates.
PURPOSE: To evaluate the impact of electronic antineoplastic ordering templates on pharmacy and infusion clinic efficiency and pharmacist interventions.
METHODS: Ordering templates, which included standard dosing and supportive care medications, were developed and activated for prescribers in phases over the course of two years. Immunotherapy and oral oncolytic templates were activated initially, followed by full implementation of the majority of intravenous (IV) ordering templates. Percent of electronic antineoplastic orders, pharmacy processing time, time to first drug delivery, and number of pharmacist encounters and interventions were documented daily for four weeks after initial implementation and for ten weeks after full implementation. Means were compared using unpaired t tests.
RESULTS: After initial implementation, the percentage of electronic antineoplastic orders increased from 0% to 100% for oral antineoplastics and from 0% to 39% for IV orders. After full implementation, IV orders increased to 69% in weeks 1-5 and 96% in weeks 6-10. Mean pharmacy processing time for supportive care medications was 35 minutes initially. This increased briefly after full implementation (weeks 1-5), then decreased to 17 minutes in weeks 6-10 (p<0.01). Delivery of the first medication to the infusion center decreased by 31 minutes at week ten (<0.01). Mean daily pharmacist encounters increased by 28%(<0.01) and documented interventions increased by 22% (<0.01).
IMPLICATIONS: Implementation of electronic antineoplastic ordering templates increased provider order entry, pharmacy and infusion clinic efficiency and pharmacist interventions. These outcomes may translate to improved patient safety and patient access to quality care. Templates developed can serve as a model for other health systems to implement electronic antineoplastic ordering.
BACKGROUND: Antineoplastic ordering is high in both complexity and risk for medication errors. Joint Commission and institutional policy recommends computerized prescriber order entry and verification by a pharmacist as a best practice for patient safety. The use of ordering templates minimizes the risk of errors and ensures appropriate supportive care is being provided. The Veterans Affairs electronic medical record does not feature an antineoplastic ordering component; historically, templates have been on paper. The oncology, pharmacy, and clinical informatics departments in our health system formed a team to create electronic antineoplastic ordering templates.
PURPOSE: To evaluate the impact of electronic antineoplastic ordering templates on pharmacy and infusion clinic efficiency and pharmacist interventions.
METHODS: Ordering templates, which included standard dosing and supportive care medications, were developed and activated for prescribers in phases over the course of two years. Immunotherapy and oral oncolytic templates were activated initially, followed by full implementation of the majority of intravenous (IV) ordering templates. Percent of electronic antineoplastic orders, pharmacy processing time, time to first drug delivery, and number of pharmacist encounters and interventions were documented daily for four weeks after initial implementation and for ten weeks after full implementation. Means were compared using unpaired t tests.
RESULTS: After initial implementation, the percentage of electronic antineoplastic orders increased from 0% to 100% for oral antineoplastics and from 0% to 39% for IV orders. After full implementation, IV orders increased to 69% in weeks 1-5 and 96% in weeks 6-10. Mean pharmacy processing time for supportive care medications was 35 minutes initially. This increased briefly after full implementation (weeks 1-5), then decreased to 17 minutes in weeks 6-10 (p<0.01). Delivery of the first medication to the infusion center decreased by 31 minutes at week ten (<0.01). Mean daily pharmacist encounters increased by 28%(<0.01) and documented interventions increased by 22% (<0.01).
IMPLICATIONS: Implementation of electronic antineoplastic ordering templates increased provider order entry, pharmacy and infusion clinic efficiency and pharmacist interventions. These outcomes may translate to improved patient safety and patient access to quality care. Templates developed can serve as a model for other health systems to implement electronic antineoplastic ordering.