User login
Human donor milk prevents preterm necrotizing enterocolitis
Preterm infants should be exclusively fed human milk rather than formula milk to prevent necrotizing enterocolitis, even if the milk does not come from the mother, a study published online in the Journal of Pediatrics confirmed.
Preterm infants fed only pasteurized donor human milk (with pasteurized donor human milk–based human milk fortifier added as needed to meet nutritional needs) were less likely to develop the potentially life-threatening gastrointestinal condition than were those given a bovine milk–based formula. Indeed, there was only one (3%) case of necrotizing enterocolitis in the infant group fed the donor human milk, versus five (21%) cases in those fed the preterm formula milk.
Importantly, there were no cases of necrotizing enterocolitis requiring surgical intervention in the donor human milk group, compared with four cases in the formula milk group.
"These data support those reported in our previous study (J. Pediatr. 2010;156:562-7) of pasteurized donor human milk vs. preterm formula used as supplements to a mother’s own milk diet," Dr. Elizabeth Cristofalo of Johns Hopkins University, Baltimore, and her associates noted (J. Pediatr. 2013 Aug. 22 [doi:10.1016/j.jpeds.2013.07.011]).
The findings also concur with current guidance issued by the American Academy of Pediatrics on the benefits of human breast milk (Pediatrics 2012;129:e827-41). This guidance states that all preterm infants should receive human milk, preferably from the mother. If that is not possible or the mother does not wish to breastfeed, then pasteurized human donor milk should be used.
"Pasteurized donor human milk has emerged as an alternative for mother’s own milk," Dr. Cristofalo and her associates observed. "It has been unclear, however, if pasteurized donor human milk confers the same health benefits as does mother’s own milk."
Their current study involved 53 preterm infants who were fed either pasteurized milk from human donors or a bovine milk–based preterm formula. The infants, with birth weights between 500g and 1,250g, were enrolled at seven neonatal care units and had received parenteral nutrition within 48 hours of their birth and enteral feedings before 21 days of age.
Altogether, 29 infants were randomized to receive donor human milk and 24 to receive the bovine milk–based preterm formula milk. The primary outcome of the study was the duration of parenteral nutrition, which was significantly reduced in those fed the donor human milk (median of 27 vs. 36 days).
Growth rates were slightly lower in the donor human milk group than in the bovine milk–based formula group, but differences were only significant for the infants’ gain in length (0.84 cm/week vs. 1.12 cm/week, P = .006). We "believe these small differences can be prevented by further adjustments in fortifier content to support improved rates of growth," Dr. Cristofalo and her colleagues proposed.
With regard to necrotizing enterocolitis, the team found that all six cases not requiring surgical intervention occurred within the first 29 days of birth. The number of infants needed to be fed an exclusive human milk diet to prevent one case of surgical necrotizing enterocolitis was six.
The researchers concluded that there is a "greater need for enhanced lactation support in the neonatal intensive care unit" and suggested "an imperative to establish more human milk banks."
Necrotizing enterocolitis affects between 7% and 12% of infants born with a low birth weight (less than 1,500 g) in the United States each year. The exact cause is unclear, but it may be the result of multiple factors, including ischemic injury and abnormal microbial colonization.
The condition is well known to have a detrimental effect on survival and also lead to longer-term complications, particularly if surgery is required. Recent findings have suggested that necrotizing enterocolitis costs U.S. health care services a mean of $5,112 per infant between 6 months and 1 year or age, with higher costs for treating surgical necrotizing enterocolitis of $18,274 between 6 and 12 months, $14,067 between 1 and 2 years, and $8,501 between 2 and 3 years (BMC Pediatr. 2013;13:127).
Dr. Cristofalo and her coinvestigators received financial support from Prolacta Bioscience, the manufacturers of the pasteurized donor milk used in the study. Two of the investigators are employees of the company, and one author is a paid consultant of the company.
Preterm infants should be exclusively fed human milk rather than formula milk to prevent necrotizing enterocolitis, even if the milk does not come from the mother, a study published online in the Journal of Pediatrics confirmed.
Preterm infants fed only pasteurized donor human milk (with pasteurized donor human milk–based human milk fortifier added as needed to meet nutritional needs) were less likely to develop the potentially life-threatening gastrointestinal condition than were those given a bovine milk–based formula. Indeed, there was only one (3%) case of necrotizing enterocolitis in the infant group fed the donor human milk, versus five (21%) cases in those fed the preterm formula milk.
Importantly, there were no cases of necrotizing enterocolitis requiring surgical intervention in the donor human milk group, compared with four cases in the formula milk group.
"These data support those reported in our previous study (J. Pediatr. 2010;156:562-7) of pasteurized donor human milk vs. preterm formula used as supplements to a mother’s own milk diet," Dr. Elizabeth Cristofalo of Johns Hopkins University, Baltimore, and her associates noted (J. Pediatr. 2013 Aug. 22 [doi:10.1016/j.jpeds.2013.07.011]).
The findings also concur with current guidance issued by the American Academy of Pediatrics on the benefits of human breast milk (Pediatrics 2012;129:e827-41). This guidance states that all preterm infants should receive human milk, preferably from the mother. If that is not possible or the mother does not wish to breastfeed, then pasteurized human donor milk should be used.
"Pasteurized donor human milk has emerged as an alternative for mother’s own milk," Dr. Cristofalo and her associates observed. "It has been unclear, however, if pasteurized donor human milk confers the same health benefits as does mother’s own milk."
Their current study involved 53 preterm infants who were fed either pasteurized milk from human donors or a bovine milk–based preterm formula. The infants, with birth weights between 500g and 1,250g, were enrolled at seven neonatal care units and had received parenteral nutrition within 48 hours of their birth and enteral feedings before 21 days of age.
Altogether, 29 infants were randomized to receive donor human milk and 24 to receive the bovine milk–based preterm formula milk. The primary outcome of the study was the duration of parenteral nutrition, which was significantly reduced in those fed the donor human milk (median of 27 vs. 36 days).
Growth rates were slightly lower in the donor human milk group than in the bovine milk–based formula group, but differences were only significant for the infants’ gain in length (0.84 cm/week vs. 1.12 cm/week, P = .006). We "believe these small differences can be prevented by further adjustments in fortifier content to support improved rates of growth," Dr. Cristofalo and her colleagues proposed.
With regard to necrotizing enterocolitis, the team found that all six cases not requiring surgical intervention occurred within the first 29 days of birth. The number of infants needed to be fed an exclusive human milk diet to prevent one case of surgical necrotizing enterocolitis was six.
The researchers concluded that there is a "greater need for enhanced lactation support in the neonatal intensive care unit" and suggested "an imperative to establish more human milk banks."
Necrotizing enterocolitis affects between 7% and 12% of infants born with a low birth weight (less than 1,500 g) in the United States each year. The exact cause is unclear, but it may be the result of multiple factors, including ischemic injury and abnormal microbial colonization.
The condition is well known to have a detrimental effect on survival and also lead to longer-term complications, particularly if surgery is required. Recent findings have suggested that necrotizing enterocolitis costs U.S. health care services a mean of $5,112 per infant between 6 months and 1 year or age, with higher costs for treating surgical necrotizing enterocolitis of $18,274 between 6 and 12 months, $14,067 between 1 and 2 years, and $8,501 between 2 and 3 years (BMC Pediatr. 2013;13:127).
Dr. Cristofalo and her coinvestigators received financial support from Prolacta Bioscience, the manufacturers of the pasteurized donor milk used in the study. Two of the investigators are employees of the company, and one author is a paid consultant of the company.
Preterm infants should be exclusively fed human milk rather than formula milk to prevent necrotizing enterocolitis, even if the milk does not come from the mother, a study published online in the Journal of Pediatrics confirmed.
Preterm infants fed only pasteurized donor human milk (with pasteurized donor human milk–based human milk fortifier added as needed to meet nutritional needs) were less likely to develop the potentially life-threatening gastrointestinal condition than were those given a bovine milk–based formula. Indeed, there was only one (3%) case of necrotizing enterocolitis in the infant group fed the donor human milk, versus five (21%) cases in those fed the preterm formula milk.
Importantly, there were no cases of necrotizing enterocolitis requiring surgical intervention in the donor human milk group, compared with four cases in the formula milk group.
"These data support those reported in our previous study (J. Pediatr. 2010;156:562-7) of pasteurized donor human milk vs. preterm formula used as supplements to a mother’s own milk diet," Dr. Elizabeth Cristofalo of Johns Hopkins University, Baltimore, and her associates noted (J. Pediatr. 2013 Aug. 22 [doi:10.1016/j.jpeds.2013.07.011]).
The findings also concur with current guidance issued by the American Academy of Pediatrics on the benefits of human breast milk (Pediatrics 2012;129:e827-41). This guidance states that all preterm infants should receive human milk, preferably from the mother. If that is not possible or the mother does not wish to breastfeed, then pasteurized human donor milk should be used.
"Pasteurized donor human milk has emerged as an alternative for mother’s own milk," Dr. Cristofalo and her associates observed. "It has been unclear, however, if pasteurized donor human milk confers the same health benefits as does mother’s own milk."
Their current study involved 53 preterm infants who were fed either pasteurized milk from human donors or a bovine milk–based preterm formula. The infants, with birth weights between 500g and 1,250g, were enrolled at seven neonatal care units and had received parenteral nutrition within 48 hours of their birth and enteral feedings before 21 days of age.
Altogether, 29 infants were randomized to receive donor human milk and 24 to receive the bovine milk–based preterm formula milk. The primary outcome of the study was the duration of parenteral nutrition, which was significantly reduced in those fed the donor human milk (median of 27 vs. 36 days).
Growth rates were slightly lower in the donor human milk group than in the bovine milk–based formula group, but differences were only significant for the infants’ gain in length (0.84 cm/week vs. 1.12 cm/week, P = .006). We "believe these small differences can be prevented by further adjustments in fortifier content to support improved rates of growth," Dr. Cristofalo and her colleagues proposed.
With regard to necrotizing enterocolitis, the team found that all six cases not requiring surgical intervention occurred within the first 29 days of birth. The number of infants needed to be fed an exclusive human milk diet to prevent one case of surgical necrotizing enterocolitis was six.
The researchers concluded that there is a "greater need for enhanced lactation support in the neonatal intensive care unit" and suggested "an imperative to establish more human milk banks."
Necrotizing enterocolitis affects between 7% and 12% of infants born with a low birth weight (less than 1,500 g) in the United States each year. The exact cause is unclear, but it may be the result of multiple factors, including ischemic injury and abnormal microbial colonization.
The condition is well known to have a detrimental effect on survival and also lead to longer-term complications, particularly if surgery is required. Recent findings have suggested that necrotizing enterocolitis costs U.S. health care services a mean of $5,112 per infant between 6 months and 1 year or age, with higher costs for treating surgical necrotizing enterocolitis of $18,274 between 6 and 12 months, $14,067 between 1 and 2 years, and $8,501 between 2 and 3 years (BMC Pediatr. 2013;13:127).
Dr. Cristofalo and her coinvestigators received financial support from Prolacta Bioscience, the manufacturers of the pasteurized donor milk used in the study. Two of the investigators are employees of the company, and one author is a paid consultant of the company.
FROM THE JOURNAL OF PEDIATRICS
Major finding: Necrotizing enterocolitis occurred in five (21%) infants fed a bovine milk–based preterm formula milk, vs. only one (3%) case in those fed pasteurized donor human milk.
Data source: Multicenter, blinded, randomized controlled trial of 53 preterm infants exclusively fed pasteurized donor human milk or a bovine milk–based preterm formula.
Disclosures: Dr. Cristofalo and her coinvestigators received financial support from Prolacta Bioscience, the manufacturers of the pasteurized donor milk used in the study. Two of the investigators are employees of the company, and one author is a paid consultant of the company.
Vaccine extended overall survival in subset of NSCLC patients
AMSTERDAM – Another investigational lung cancer vaccine did not improve overall survival, although its use might still benefit some patients, according to results of a phase III study presented at the multidisciplinary European cancer congresses.
The therapeutic tumor cell cancer vaccine belagenpumatucel-L (Lucanix) was no better than placebo at improving the primary endpoint of overall survival after frontline, platinum-based chemotherapy in the STOP study. A total of 532 patients with stable stage III or IV non–small cell lung cancer (NSCLC) participated in the study.
Median overall survival was 20.3 months in the vaccinated patients and 17.8 months in those who had received placebo over a 2-year period (hazard ratio, 0.54; P = .0595).
There was an indication that overall survival might be improved, however, if patients were given the vaccine within 12 weeks of stopping their frontline chemotherapy. Indeed, overall survival was 20.7 months in 169 patients who were vaccinated within 12 weeks of stopping chemotherapy versus 13.3 months in 149 patients who did not receive the vaccine until after that time (HR, 0.77; P = .0092).
"Prospective subset data support further development of belagenpumatucel-L in NSCLC," said Dr. Giuseppe Giaccone, the presenting investigator and associate director of clinical research at the Lombardi Comprehensive Cancer Center at Georgetown University, Washington.
Dr. Giaccone said that the trial was not appropriately powered to meet its primary endpoint. The trial design presumed that the placebo-treated patients would achieve a median survival of around 10.5 months, which was the norm at the time the trial was conceived.
The somewhat disappointing findings in the STOP trial follow those of the phase III START trial, which found no overall survival benefit with a different investigational lung cancer vaccine, tecemotide (Stimuvax). Development of tecemotide also continues, however, as there was an indication that the vaccine might work if given concurrently with chemoradiation rather than after it. The START2 trial will be investigating that hypothesis.
The STOP trial was a randomized, double blind, placebo-controlled investigation performed in eight countries that ran for 46 months and involved patients with late-stage NSCLC; 490 patients had stage IIIB/IV cancer and 42 had stage IIIA cancers. Patients could be enrolled in the trial if they had stable disease after up to six cycles of platinum-based chemotherapy. The aim of the study was to determine if vaccination with belagenpumatucel-L could prevent progression after successful first-line chemotherapy.
Belagenpumatucel-L consists of four allogeneic NSCLC cell lines that have been modified to express an antisense DNA molecule that binds to the gene responsible for producing transforming growth factor beta (TGF-beta), thus blocking production of the protein. TGF-beta has immunosuppressive properties and helps tumor cells evade immune defenses. During the study, the vaccine was given as 18-month intradermal injections followed by a further two injections given 3 months apart. Each dose contained 2.5 × 107 cells.
"Overall, the response rate to the vaccination was low, at 2.6%," Dr. Giaccone reported. There were only one (0.4%) complete and six (2.2%) partial responses. "Progression-free survival did not correlate with overall survival, and there was a trend in some of the subgroups toward an improved PFS, but this was not statistically significant."
The most common side effects were injection-site reactions (grade 1-2), occurring in 260 of the vaccinated and 62 of the placebo-treated patients. Unexpected effects included abdominal pain, constipation, decreased appetite, nasopharyngitis, and noncardiac chest pain, all affecting more patients who received belagenpumatucel-L than those who received placebo. Five serious adverse events occurred, three of which were in patients treated with the vaccine, and only one of these, an allergic reaction, was probably due to the vaccine itself.
NovaRx sponsored the study. Dr. Giaccone said he had no conflicts of interest but noted his coinvestigators included employees of NovaRx.
The overall survival analysis in this study was due to be performed after 354 events had occurred. Instead, the analysis was done after only 253 events, which would affect the statistical power of the trial. In addition, the variety of subgroup analyses undertaken and the low estimate of overall survival in the control arm would have an influence on the outcome.
There did appear to be a survival advantage in stage IIIB/IV patients randomized within 12 weeks of completion of the frontline chemotherapy and perhaps a benefit in those who had received prior radiation therapy. These exploratory data warrant further assessment of the vaccine.
Dr. Rolf Stahel is with the University of Zurich. He was the discussant of the presentation at the meeting. Dr. Stahel made no disclosures as to his relationship with the company that makes the vaccine.
The overall survival analysis in this study was due to be performed after 354 events had occurred. Instead, the analysis was done after only 253 events, which would affect the statistical power of the trial. In addition, the variety of subgroup analyses undertaken and the low estimate of overall survival in the control arm would have an influence on the outcome.
There did appear to be a survival advantage in stage IIIB/IV patients randomized within 12 weeks of completion of the frontline chemotherapy and perhaps a benefit in those who had received prior radiation therapy. These exploratory data warrant further assessment of the vaccine.
Dr. Rolf Stahel is with the University of Zurich. He was the discussant of the presentation at the meeting. Dr. Stahel made no disclosures as to his relationship with the company that makes the vaccine.
The overall survival analysis in this study was due to be performed after 354 events had occurred. Instead, the analysis was done after only 253 events, which would affect the statistical power of the trial. In addition, the variety of subgroup analyses undertaken and the low estimate of overall survival in the control arm would have an influence on the outcome.
There did appear to be a survival advantage in stage IIIB/IV patients randomized within 12 weeks of completion of the frontline chemotherapy and perhaps a benefit in those who had received prior radiation therapy. These exploratory data warrant further assessment of the vaccine.
Dr. Rolf Stahel is with the University of Zurich. He was the discussant of the presentation at the meeting. Dr. Stahel made no disclosures as to his relationship with the company that makes the vaccine.
AMSTERDAM – Another investigational lung cancer vaccine did not improve overall survival, although its use might still benefit some patients, according to results of a phase III study presented at the multidisciplinary European cancer congresses.
The therapeutic tumor cell cancer vaccine belagenpumatucel-L (Lucanix) was no better than placebo at improving the primary endpoint of overall survival after frontline, platinum-based chemotherapy in the STOP study. A total of 532 patients with stable stage III or IV non–small cell lung cancer (NSCLC) participated in the study.
Median overall survival was 20.3 months in the vaccinated patients and 17.8 months in those who had received placebo over a 2-year period (hazard ratio, 0.54; P = .0595).
There was an indication that overall survival might be improved, however, if patients were given the vaccine within 12 weeks of stopping their frontline chemotherapy. Indeed, overall survival was 20.7 months in 169 patients who were vaccinated within 12 weeks of stopping chemotherapy versus 13.3 months in 149 patients who did not receive the vaccine until after that time (HR, 0.77; P = .0092).
"Prospective subset data support further development of belagenpumatucel-L in NSCLC," said Dr. Giuseppe Giaccone, the presenting investigator and associate director of clinical research at the Lombardi Comprehensive Cancer Center at Georgetown University, Washington.
Dr. Giaccone said that the trial was not appropriately powered to meet its primary endpoint. The trial design presumed that the placebo-treated patients would achieve a median survival of around 10.5 months, which was the norm at the time the trial was conceived.
The somewhat disappointing findings in the STOP trial follow those of the phase III START trial, which found no overall survival benefit with a different investigational lung cancer vaccine, tecemotide (Stimuvax). Development of tecemotide also continues, however, as there was an indication that the vaccine might work if given concurrently with chemoradiation rather than after it. The START2 trial will be investigating that hypothesis.
The STOP trial was a randomized, double blind, placebo-controlled investigation performed in eight countries that ran for 46 months and involved patients with late-stage NSCLC; 490 patients had stage IIIB/IV cancer and 42 had stage IIIA cancers. Patients could be enrolled in the trial if they had stable disease after up to six cycles of platinum-based chemotherapy. The aim of the study was to determine if vaccination with belagenpumatucel-L could prevent progression after successful first-line chemotherapy.
Belagenpumatucel-L consists of four allogeneic NSCLC cell lines that have been modified to express an antisense DNA molecule that binds to the gene responsible for producing transforming growth factor beta (TGF-beta), thus blocking production of the protein. TGF-beta has immunosuppressive properties and helps tumor cells evade immune defenses. During the study, the vaccine was given as 18-month intradermal injections followed by a further two injections given 3 months apart. Each dose contained 2.5 × 107 cells.
"Overall, the response rate to the vaccination was low, at 2.6%," Dr. Giaccone reported. There were only one (0.4%) complete and six (2.2%) partial responses. "Progression-free survival did not correlate with overall survival, and there was a trend in some of the subgroups toward an improved PFS, but this was not statistically significant."
The most common side effects were injection-site reactions (grade 1-2), occurring in 260 of the vaccinated and 62 of the placebo-treated patients. Unexpected effects included abdominal pain, constipation, decreased appetite, nasopharyngitis, and noncardiac chest pain, all affecting more patients who received belagenpumatucel-L than those who received placebo. Five serious adverse events occurred, three of which were in patients treated with the vaccine, and only one of these, an allergic reaction, was probably due to the vaccine itself.
NovaRx sponsored the study. Dr. Giaccone said he had no conflicts of interest but noted his coinvestigators included employees of NovaRx.
AMSTERDAM – Another investigational lung cancer vaccine did not improve overall survival, although its use might still benefit some patients, according to results of a phase III study presented at the multidisciplinary European cancer congresses.
The therapeutic tumor cell cancer vaccine belagenpumatucel-L (Lucanix) was no better than placebo at improving the primary endpoint of overall survival after frontline, platinum-based chemotherapy in the STOP study. A total of 532 patients with stable stage III or IV non–small cell lung cancer (NSCLC) participated in the study.
Median overall survival was 20.3 months in the vaccinated patients and 17.8 months in those who had received placebo over a 2-year period (hazard ratio, 0.54; P = .0595).
There was an indication that overall survival might be improved, however, if patients were given the vaccine within 12 weeks of stopping their frontline chemotherapy. Indeed, overall survival was 20.7 months in 169 patients who were vaccinated within 12 weeks of stopping chemotherapy versus 13.3 months in 149 patients who did not receive the vaccine until after that time (HR, 0.77; P = .0092).
"Prospective subset data support further development of belagenpumatucel-L in NSCLC," said Dr. Giuseppe Giaccone, the presenting investigator and associate director of clinical research at the Lombardi Comprehensive Cancer Center at Georgetown University, Washington.
Dr. Giaccone said that the trial was not appropriately powered to meet its primary endpoint. The trial design presumed that the placebo-treated patients would achieve a median survival of around 10.5 months, which was the norm at the time the trial was conceived.
The somewhat disappointing findings in the STOP trial follow those of the phase III START trial, which found no overall survival benefit with a different investigational lung cancer vaccine, tecemotide (Stimuvax). Development of tecemotide also continues, however, as there was an indication that the vaccine might work if given concurrently with chemoradiation rather than after it. The START2 trial will be investigating that hypothesis.
The STOP trial was a randomized, double blind, placebo-controlled investigation performed in eight countries that ran for 46 months and involved patients with late-stage NSCLC; 490 patients had stage IIIB/IV cancer and 42 had stage IIIA cancers. Patients could be enrolled in the trial if they had stable disease after up to six cycles of platinum-based chemotherapy. The aim of the study was to determine if vaccination with belagenpumatucel-L could prevent progression after successful first-line chemotherapy.
Belagenpumatucel-L consists of four allogeneic NSCLC cell lines that have been modified to express an antisense DNA molecule that binds to the gene responsible for producing transforming growth factor beta (TGF-beta), thus blocking production of the protein. TGF-beta has immunosuppressive properties and helps tumor cells evade immune defenses. During the study, the vaccine was given as 18-month intradermal injections followed by a further two injections given 3 months apart. Each dose contained 2.5 × 107 cells.
"Overall, the response rate to the vaccination was low, at 2.6%," Dr. Giaccone reported. There were only one (0.4%) complete and six (2.2%) partial responses. "Progression-free survival did not correlate with overall survival, and there was a trend in some of the subgroups toward an improved PFS, but this was not statistically significant."
The most common side effects were injection-site reactions (grade 1-2), occurring in 260 of the vaccinated and 62 of the placebo-treated patients. Unexpected effects included abdominal pain, constipation, decreased appetite, nasopharyngitis, and noncardiac chest pain, all affecting more patients who received belagenpumatucel-L than those who received placebo. Five serious adverse events occurred, three of which were in patients treated with the vaccine, and only one of these, an allergic reaction, was probably due to the vaccine itself.
NovaRx sponsored the study. Dr. Giaccone said he had no conflicts of interest but noted his coinvestigators included employees of NovaRx.
AT THE EUROPEAN CANCER CONGRESS 2013
Major finding: Median overall survival was 20.3 months in the vaccinated patients versus 17.8 months in those who had received placebo (HR, 0.54; P = .0595). Overall survival was 20.7 months in patients vaccinated within 12 weeks of stopping chemotherapy versus 13.3 months in those who were vaccinated after that (HR, 0.77; P = .0092).
Data source: Phase III STOP study, a multicenter, randomized, double-blind, placebo-controlled trial in 532 patients with advanced non–small cell lung cancer.
Disclosures: NovaRx sponsored the study. Dr. Giaccone said he had no conflicts of interest but noted his coinvestigators included employees of NovaRx.
Natural killer cell protein affects pediatric stem cell transplant survival
The surface protein on the donor’s natural killer cells appeared to affect survival after hematopoietic stem cell transplant in children with leukemia, according to research published online in the Journal of Clinical Oncology.
In a study of KIR2DL1 functional allelotyping, children who received hematopoietic stem cell transplants were more likely to survive and to do so without experiencing progressive disease if the donor had an arginine-containing KIR2DL1 allele rather than a cysteine-containing KIR2DL1 allele.
Compared to patients who received donor grafts with two cysteine-containing alleles, the hazard ratio (HR) for the risk of death was 0.4 (P = .0001) when donor grafts had two copies of the arginine-containing allele and 0.42 (P = .0013) when donor grafts had one copy of the arginine allele and one copy of the cysteine-containing allele. Hazard ratio for progression-free survival were, respectively, 0.42 (P = .0003) and 0.48 (P = .0075).
Functional allele typing should be included when selecting donors for hematopoietic stem cell transplants, advised Rafijul Bari, Ph.D., and his associates (J. Clin. Oncol. 2013 [doi: 10.1200/JCO.2012.47.4007]). "HLA and KIR typing can be performed using the same blood sample collected from potential donors during routine HLA matching," and the process would not slow down donor work-up. Further, KIR allele typing and KIR ligand typing can be performed simultaneously in a day using SNP assays.
Killer-cell immunoglobulin-like receptors (KIRs) regulate natural killer (NK) cell activity. The genes that encode these receptor proteins are found on chromosome 19 and are highly polymorphic, with at least 15 forms identified. KIR2DL1 is present in approximately 90%-95% of healthy individuals.
Dr. Bari and his coworkers at St Jude Children’s Research Hospital in Memphis, where the transplants were all performed, have previously found that a KIR2DL1 allele that contains arginine rather than cysteine in position 245 of the transmembrane domain encodes receptors that have stronger signaling function (Blood 2009;114:5182-90).
Their current study tested the hypothesis that the arginine-containing allele (KIR2DL1-R245) and the cysteine-containing allele (KIR2DL1-C245) would have different effects on the clinical outcomes of allogenic hematopoietic stem cell transplants.
Between 2000 and 2010, 313 children underwent allogenic hematopoietic stem cell transplants. DNA samples from their donors were obtained and KIR2DL1 functional allelotyping was retrospectively performed using a single nucleotide polymorphism assay developed by Dr. Bari and his colleague, Dr. Wing Leung. HLA typing also was performed.
The median age of the recipient children at transplant was 9.9 years. The reason for transplant varied, but the majority (n = 231; 73%) underwent the procedure for hematologic malignancies.
"The effect of KIR2DL1 allelic polymorphism on the patterns of survival estimates was similar in patients with childhood AML [acute myeloid leukemia] or ALL [acute lymphoblastic leukemia] among all allele groups," the researchers reported.
When donor grafts had the arginine-containing allele, patients fared better if the donor grafts were mismatched for the HLA-C receptor ligand. The 5-year survival rate was 48% for mismatched and 31% for matched grafts, respectively. "In contrast, patients who received a donor graft with the weak [cysteine] allele had poor survival, regardless of the presence or absence of receptor-ligand mismatch," the researchers wrote.
"The results presented thus far provide evidence that the capacity of inhibitory KIRs expressed by licensed NK cells to destroy cell targets lacking their cognate HLA ligands depends not only on the missing ligand but also on the signaling potential of the inhibitory KIR expressed," observed Dr. Richard J. O’Reilly, chief of the pediatric bone marrow transplant service at Memorial Sloan-Kettering Cancer Center (MSKCC), New York in an editorial (J. Clin. Oncol. 2013 Sept. 16 [doi:10.1200/JCO.2013.50.2138]).
"If confirmed, this introduces an important new variable in the analysis of NK-cell–mediated leukemia resistance," he said.
The research was supported by a grant from the National Institutes of Health, the Assisi Foundation of Memphis, and the American Lebanese Syrian Associated Charities. Dr. Bari and Dr. Leung hold the patent for the KIRs SNP assay used in the study. Dr. O’Reilly has received honoraria from and has provided expert testimony to Miltenyi Biotec, a company that produces a KIR-typing kit.
The surface protein on the donor’s natural killer cells appeared to affect survival after hematopoietic stem cell transplant in children with leukemia, according to research published online in the Journal of Clinical Oncology.
In a study of KIR2DL1 functional allelotyping, children who received hematopoietic stem cell transplants were more likely to survive and to do so without experiencing progressive disease if the donor had an arginine-containing KIR2DL1 allele rather than a cysteine-containing KIR2DL1 allele.
Compared to patients who received donor grafts with two cysteine-containing alleles, the hazard ratio (HR) for the risk of death was 0.4 (P = .0001) when donor grafts had two copies of the arginine-containing allele and 0.42 (P = .0013) when donor grafts had one copy of the arginine allele and one copy of the cysteine-containing allele. Hazard ratio for progression-free survival were, respectively, 0.42 (P = .0003) and 0.48 (P = .0075).
Functional allele typing should be included when selecting donors for hematopoietic stem cell transplants, advised Rafijul Bari, Ph.D., and his associates (J. Clin. Oncol. 2013 [doi: 10.1200/JCO.2012.47.4007]). "HLA and KIR typing can be performed using the same blood sample collected from potential donors during routine HLA matching," and the process would not slow down donor work-up. Further, KIR allele typing and KIR ligand typing can be performed simultaneously in a day using SNP assays.
Killer-cell immunoglobulin-like receptors (KIRs) regulate natural killer (NK) cell activity. The genes that encode these receptor proteins are found on chromosome 19 and are highly polymorphic, with at least 15 forms identified. KIR2DL1 is present in approximately 90%-95% of healthy individuals.
Dr. Bari and his coworkers at St Jude Children’s Research Hospital in Memphis, where the transplants were all performed, have previously found that a KIR2DL1 allele that contains arginine rather than cysteine in position 245 of the transmembrane domain encodes receptors that have stronger signaling function (Blood 2009;114:5182-90).
Their current study tested the hypothesis that the arginine-containing allele (KIR2DL1-R245) and the cysteine-containing allele (KIR2DL1-C245) would have different effects on the clinical outcomes of allogenic hematopoietic stem cell transplants.
Between 2000 and 2010, 313 children underwent allogenic hematopoietic stem cell transplants. DNA samples from their donors were obtained and KIR2DL1 functional allelotyping was retrospectively performed using a single nucleotide polymorphism assay developed by Dr. Bari and his colleague, Dr. Wing Leung. HLA typing also was performed.
The median age of the recipient children at transplant was 9.9 years. The reason for transplant varied, but the majority (n = 231; 73%) underwent the procedure for hematologic malignancies.
"The effect of KIR2DL1 allelic polymorphism on the patterns of survival estimates was similar in patients with childhood AML [acute myeloid leukemia] or ALL [acute lymphoblastic leukemia] among all allele groups," the researchers reported.
When donor grafts had the arginine-containing allele, patients fared better if the donor grafts were mismatched for the HLA-C receptor ligand. The 5-year survival rate was 48% for mismatched and 31% for matched grafts, respectively. "In contrast, patients who received a donor graft with the weak [cysteine] allele had poor survival, regardless of the presence or absence of receptor-ligand mismatch," the researchers wrote.
"The results presented thus far provide evidence that the capacity of inhibitory KIRs expressed by licensed NK cells to destroy cell targets lacking their cognate HLA ligands depends not only on the missing ligand but also on the signaling potential of the inhibitory KIR expressed," observed Dr. Richard J. O’Reilly, chief of the pediatric bone marrow transplant service at Memorial Sloan-Kettering Cancer Center (MSKCC), New York in an editorial (J. Clin. Oncol. 2013 Sept. 16 [doi:10.1200/JCO.2013.50.2138]).
"If confirmed, this introduces an important new variable in the analysis of NK-cell–mediated leukemia resistance," he said.
The research was supported by a grant from the National Institutes of Health, the Assisi Foundation of Memphis, and the American Lebanese Syrian Associated Charities. Dr. Bari and Dr. Leung hold the patent for the KIRs SNP assay used in the study. Dr. O’Reilly has received honoraria from and has provided expert testimony to Miltenyi Biotec, a company that produces a KIR-typing kit.
The surface protein on the donor’s natural killer cells appeared to affect survival after hematopoietic stem cell transplant in children with leukemia, according to research published online in the Journal of Clinical Oncology.
In a study of KIR2DL1 functional allelotyping, children who received hematopoietic stem cell transplants were more likely to survive and to do so without experiencing progressive disease if the donor had an arginine-containing KIR2DL1 allele rather than a cysteine-containing KIR2DL1 allele.
Compared to patients who received donor grafts with two cysteine-containing alleles, the hazard ratio (HR) for the risk of death was 0.4 (P = .0001) when donor grafts had two copies of the arginine-containing allele and 0.42 (P = .0013) when donor grafts had one copy of the arginine allele and one copy of the cysteine-containing allele. Hazard ratio for progression-free survival were, respectively, 0.42 (P = .0003) and 0.48 (P = .0075).
Functional allele typing should be included when selecting donors for hematopoietic stem cell transplants, advised Rafijul Bari, Ph.D., and his associates (J. Clin. Oncol. 2013 [doi: 10.1200/JCO.2012.47.4007]). "HLA and KIR typing can be performed using the same blood sample collected from potential donors during routine HLA matching," and the process would not slow down donor work-up. Further, KIR allele typing and KIR ligand typing can be performed simultaneously in a day using SNP assays.
Killer-cell immunoglobulin-like receptors (KIRs) regulate natural killer (NK) cell activity. The genes that encode these receptor proteins are found on chromosome 19 and are highly polymorphic, with at least 15 forms identified. KIR2DL1 is present in approximately 90%-95% of healthy individuals.
Dr. Bari and his coworkers at St Jude Children’s Research Hospital in Memphis, where the transplants were all performed, have previously found that a KIR2DL1 allele that contains arginine rather than cysteine in position 245 of the transmembrane domain encodes receptors that have stronger signaling function (Blood 2009;114:5182-90).
Their current study tested the hypothesis that the arginine-containing allele (KIR2DL1-R245) and the cysteine-containing allele (KIR2DL1-C245) would have different effects on the clinical outcomes of allogenic hematopoietic stem cell transplants.
Between 2000 and 2010, 313 children underwent allogenic hematopoietic stem cell transplants. DNA samples from their donors were obtained and KIR2DL1 functional allelotyping was retrospectively performed using a single nucleotide polymorphism assay developed by Dr. Bari and his colleague, Dr. Wing Leung. HLA typing also was performed.
The median age of the recipient children at transplant was 9.9 years. The reason for transplant varied, but the majority (n = 231; 73%) underwent the procedure for hematologic malignancies.
"The effect of KIR2DL1 allelic polymorphism on the patterns of survival estimates was similar in patients with childhood AML [acute myeloid leukemia] or ALL [acute lymphoblastic leukemia] among all allele groups," the researchers reported.
When donor grafts had the arginine-containing allele, patients fared better if the donor grafts were mismatched for the HLA-C receptor ligand. The 5-year survival rate was 48% for mismatched and 31% for matched grafts, respectively. "In contrast, patients who received a donor graft with the weak [cysteine] allele had poor survival, regardless of the presence or absence of receptor-ligand mismatch," the researchers wrote.
"The results presented thus far provide evidence that the capacity of inhibitory KIRs expressed by licensed NK cells to destroy cell targets lacking their cognate HLA ligands depends not only on the missing ligand but also on the signaling potential of the inhibitory KIR expressed," observed Dr. Richard J. O’Reilly, chief of the pediatric bone marrow transplant service at Memorial Sloan-Kettering Cancer Center (MSKCC), New York in an editorial (J. Clin. Oncol. 2013 Sept. 16 [doi:10.1200/JCO.2013.50.2138]).
"If confirmed, this introduces an important new variable in the analysis of NK-cell–mediated leukemia resistance," he said.
The research was supported by a grant from the National Institutes of Health, the Assisi Foundation of Memphis, and the American Lebanese Syrian Associated Charities. Dr. Bari and Dr. Leung hold the patent for the KIRs SNP assay used in the study. Dr. O’Reilly has received honoraria from and has provided expert testimony to Miltenyi Biotec, a company that produces a KIR-typing kit.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Major finding: Compared with patients who received donor grafts with two cysteine-containing alleles, the hazard ratio for the risk of death was 0.4 (P = .0001) when donor grafts had two copies of the arginine-containing allele and 0.42 (P = .0013) when donor grafts had one copy of the arginine allele and one copy of the cysteine-containing allele.
Data source: Retrospective KIR2DL1 functional allelotyping of donor grafts to 313 children who had allogenic hematopoietic stem cell transplants between 2000 and 2010 at a single center.
Disclosures: The research was supported by a grant from the National Institutes of Health, the Assisi Foundation of Memphis, and the American Lebanese Syrian Associated Charities. Dr. Bari and Dr. Leung hold the patent for the KIRs SNP assay used in the study. Dr. O’Reilly has received honoraria from and has provided expert testimony to Miltenyi Biotec, a company that produces a kit for KIR typing.
Lung cancer diagnosis aided by novel plasma protein
A plasma protein involved in tumor oxidation and reduction reactions was useful for detecting and excluding lung cancer in more than three quarters of clinical samples evaluated in a large prospective study.
Isocitrate dehydrogenase 1 (IDH1) measurement was associated with a sensitivity of 77.1% and specificity of 76.2% in a training set of samples (n = 712), and 82.9% sensitivity and 76.6% specificity in a second validation or test set (n = 710).
Sensitivity and specificity were generally improved when the protein’s detection was considered in addition to other known or proposed non–small-cell lung cancer (NSCLC) biomarkers, with sensitivities of 75.8% and 86.3% in the training and test sets, respectively, and specificities of 89.6% and 70.7%.
"Some existing NSCLC biomarkers, such as CEA [carcinoembryonic antigen] and Cyfra21-1 [cytokeratin fragment 21-1], have been used in clinical practice, whereas others, such as CA125 [cancer antigen 125], have been recommended for further validation," Dr. Nan Sun and associates reported in the latest issue of Clinical Cancer Research.
"These biomarkers have low sensitivity, ranging from 50% to 60%, with specificities of approximately 90%," said the researchers, of the department of thoracic surgical oncology at the Cancer Institute and Hospital, Peking Union Medical College and Chinese Academy of Medical Sciences in Beijing.
Previous research by the team had shown IDH1 levels were elevated in tumor samples taken from patients with NSCLC (Mol. Cell. Proteomics 2012;11:M111). Their present investigation (Clin. Cancer. Res. 2013; 19: 5136-45) therefore aimed to see if the protein’s measurement could aid in the diagnosis of lung cancer, differentiating between those who did and those who did not have malignant disease.
For their investigation, the team obtained 1,422 blood samples from 943 patients with previously untreated NSCLC and 479 healthy individuals who were seen for routine examinations between 2007 and 2011 at their institution.
The blood samples from the lung cancer patients were taken 3 days prior to their undergoing surgery and IDH1 levels were immediately determined by enzyme-linked immunosorbent assay, while CEA, Cyfra21-1, and CA125 levels were measured with an Elecys immunoassay analyzer.
Median levels of IDH1 were 2.39 U/L higher than those of healthy controls for squamous cell carcinoma cases (n = 489) cases and 1.96 U/L higher for adenocarcinoma cases (n = 454). Additionally, median plasma levels of IDH1 were higher in patients with adenocarcinomas than in those with squamous cell carcinomas (P = .012)
"We have identified IDH1 as an effective plasma biomarker with high sensitivity and specificity in the diagnosis of NSCLC, especially lung adenocarcinoma," senior study investigator Dr. Jie He said in a press release issued by the American Association for Cancer Research. "Based on the present data, IDH1 can be used to detect stage 1 lung cancer,"
The protein might also detect precancerous lesions, but further studies are required to test that hypothesis, said Dr. He. IDH1 might be a good target for NSCLC treatment as it "may be involved in the development of lung cancer."
A multicenter clinical trial is planned to further validate the diagnostic utility of IDH1.
Research funding was provided by the National High Technology Research and Development Program of China, the International Science and Technology Corporation and Exchange Project, the National Natural Science Foundation of China, the Doctoral Fund of Ministry of Education of China, and the Government Health Care Research Foundation for Senior Officials. The authors had no conflicts of interest to disclose.
A plasma protein involved in tumor oxidation and reduction reactions was useful for detecting and excluding lung cancer in more than three quarters of clinical samples evaluated in a large prospective study.
Isocitrate dehydrogenase 1 (IDH1) measurement was associated with a sensitivity of 77.1% and specificity of 76.2% in a training set of samples (n = 712), and 82.9% sensitivity and 76.6% specificity in a second validation or test set (n = 710).
Sensitivity and specificity were generally improved when the protein’s detection was considered in addition to other known or proposed non–small-cell lung cancer (NSCLC) biomarkers, with sensitivities of 75.8% and 86.3% in the training and test sets, respectively, and specificities of 89.6% and 70.7%.
"Some existing NSCLC biomarkers, such as CEA [carcinoembryonic antigen] and Cyfra21-1 [cytokeratin fragment 21-1], have been used in clinical practice, whereas others, such as CA125 [cancer antigen 125], have been recommended for further validation," Dr. Nan Sun and associates reported in the latest issue of Clinical Cancer Research.
"These biomarkers have low sensitivity, ranging from 50% to 60%, with specificities of approximately 90%," said the researchers, of the department of thoracic surgical oncology at the Cancer Institute and Hospital, Peking Union Medical College and Chinese Academy of Medical Sciences in Beijing.
Previous research by the team had shown IDH1 levels were elevated in tumor samples taken from patients with NSCLC (Mol. Cell. Proteomics 2012;11:M111). Their present investigation (Clin. Cancer. Res. 2013; 19: 5136-45) therefore aimed to see if the protein’s measurement could aid in the diagnosis of lung cancer, differentiating between those who did and those who did not have malignant disease.
For their investigation, the team obtained 1,422 blood samples from 943 patients with previously untreated NSCLC and 479 healthy individuals who were seen for routine examinations between 2007 and 2011 at their institution.
The blood samples from the lung cancer patients were taken 3 days prior to their undergoing surgery and IDH1 levels were immediately determined by enzyme-linked immunosorbent assay, while CEA, Cyfra21-1, and CA125 levels were measured with an Elecys immunoassay analyzer.
Median levels of IDH1 were 2.39 U/L higher than those of healthy controls for squamous cell carcinoma cases (n = 489) cases and 1.96 U/L higher for adenocarcinoma cases (n = 454). Additionally, median plasma levels of IDH1 were higher in patients with adenocarcinomas than in those with squamous cell carcinomas (P = .012)
"We have identified IDH1 as an effective plasma biomarker with high sensitivity and specificity in the diagnosis of NSCLC, especially lung adenocarcinoma," senior study investigator Dr. Jie He said in a press release issued by the American Association for Cancer Research. "Based on the present data, IDH1 can be used to detect stage 1 lung cancer,"
The protein might also detect precancerous lesions, but further studies are required to test that hypothesis, said Dr. He. IDH1 might be a good target for NSCLC treatment as it "may be involved in the development of lung cancer."
A multicenter clinical trial is planned to further validate the diagnostic utility of IDH1.
Research funding was provided by the National High Technology Research and Development Program of China, the International Science and Technology Corporation and Exchange Project, the National Natural Science Foundation of China, the Doctoral Fund of Ministry of Education of China, and the Government Health Care Research Foundation for Senior Officials. The authors had no conflicts of interest to disclose.
A plasma protein involved in tumor oxidation and reduction reactions was useful for detecting and excluding lung cancer in more than three quarters of clinical samples evaluated in a large prospective study.
Isocitrate dehydrogenase 1 (IDH1) measurement was associated with a sensitivity of 77.1% and specificity of 76.2% in a training set of samples (n = 712), and 82.9% sensitivity and 76.6% specificity in a second validation or test set (n = 710).
Sensitivity and specificity were generally improved when the protein’s detection was considered in addition to other known or proposed non–small-cell lung cancer (NSCLC) biomarkers, with sensitivities of 75.8% and 86.3% in the training and test sets, respectively, and specificities of 89.6% and 70.7%.
"Some existing NSCLC biomarkers, such as CEA [carcinoembryonic antigen] and Cyfra21-1 [cytokeratin fragment 21-1], have been used in clinical practice, whereas others, such as CA125 [cancer antigen 125], have been recommended for further validation," Dr. Nan Sun and associates reported in the latest issue of Clinical Cancer Research.
"These biomarkers have low sensitivity, ranging from 50% to 60%, with specificities of approximately 90%," said the researchers, of the department of thoracic surgical oncology at the Cancer Institute and Hospital, Peking Union Medical College and Chinese Academy of Medical Sciences in Beijing.
Previous research by the team had shown IDH1 levels were elevated in tumor samples taken from patients with NSCLC (Mol. Cell. Proteomics 2012;11:M111). Their present investigation (Clin. Cancer. Res. 2013; 19: 5136-45) therefore aimed to see if the protein’s measurement could aid in the diagnosis of lung cancer, differentiating between those who did and those who did not have malignant disease.
For their investigation, the team obtained 1,422 blood samples from 943 patients with previously untreated NSCLC and 479 healthy individuals who were seen for routine examinations between 2007 and 2011 at their institution.
The blood samples from the lung cancer patients were taken 3 days prior to their undergoing surgery and IDH1 levels were immediately determined by enzyme-linked immunosorbent assay, while CEA, Cyfra21-1, and CA125 levels were measured with an Elecys immunoassay analyzer.
Median levels of IDH1 were 2.39 U/L higher than those of healthy controls for squamous cell carcinoma cases (n = 489) cases and 1.96 U/L higher for adenocarcinoma cases (n = 454). Additionally, median plasma levels of IDH1 were higher in patients with adenocarcinomas than in those with squamous cell carcinomas (P = .012)
"We have identified IDH1 as an effective plasma biomarker with high sensitivity and specificity in the diagnosis of NSCLC, especially lung adenocarcinoma," senior study investigator Dr. Jie He said in a press release issued by the American Association for Cancer Research. "Based on the present data, IDH1 can be used to detect stage 1 lung cancer,"
The protein might also detect precancerous lesions, but further studies are required to test that hypothesis, said Dr. He. IDH1 might be a good target for NSCLC treatment as it "may be involved in the development of lung cancer."
A multicenter clinical trial is planned to further validate the diagnostic utility of IDH1.
Research funding was provided by the National High Technology Research and Development Program of China, the International Science and Technology Corporation and Exchange Project, the National Natural Science Foundation of China, the Doctoral Fund of Ministry of Education of China, and the Government Health Care Research Foundation for Senior Officials. The authors had no conflicts of interest to disclose.
FROM CLINICAL CANCER RESEARCH
Major finding: IDH1 detection in plasma had a sensitivity and specificity of up to 82.9% and 76.6%, respectively.
Data source: Prospective study of 1,422 participants enrolled between 2007 and 2011; 943 patients with non–small-cell lung cancer and 479 healthy individuals as controls.
Disclosures: Research funding was provided by the National High Technology Research and Development Program of China, the International Science and Technology Corporation and Exchange Project, the National Natural Science Foundation of China, the Doctoral Fund of Ministry of Education of China, and the Government Health Care Research Foundation for Senior Officials. The authors had no conflicts of interest to disclose.
Meta-analysis confirms pramipexole calms restless legs
The dopamine agonist pramipexole allays the symptoms of primary restless leg syndrome and improves patients’ sleep quality, at least in the short-term, according to a meta-analysis of randomized, double-blind, placebo-controlled clinical trials.
The mean change in restless leg syndrome (RLS) symptoms assessed using the International RLS Study Group Rating Scale (IRLS) was –5.96, compared with placebo (P less than .00001). The mean change in sleep quality assessed by a variety of patient assessment scales was -0.48 (P less than .00001).
RLS can be categorized as primary or secondary, with the latter due to a variety of causes such as pregnancy, uremia, iron deficiency, anemia, or Parkinson’s disease. Managing those conditions effectively may subsequently improve or even cure RLS.
Primary RLS, however, is defined as that due to no other known cause, and it affects between 1.9% and 4.6% of the European and North American adult populations (Sleep Med. Rev. 2012;16:283-95).
Treatments for the condition include lifestyle changes and medications such as pramipexole and other dopamine agonists, levodopa, and painkillers. Results with dopamine agonists to date have been modest.
"Pramipexole has been proven to be a first-line drug for the treatment of movement disorders such as Parkinson’s disease and RLS," observe Dr. Wei Zhang and associates, who undertook the meta-analysis.
The researchers, who are based at the Shengjing Hospital of China Medical University, Shenyang, People’s Republic of China, added that European guidelines state that pramipexole is effective at providing short-term relief but are less certain on the long-term benefits.
"We conducted a meta-analysis in order to summarize the efficacy and tolerability of pramipexole for the treatment of primary RLS," they explained (Neuropsychiatr. Dis. Treat. 2013;9:1035-43).
Six trials published between 2006 and 2012 were identified that met the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement criteria and had used the IRLS as a primary outcome measure.
Altogether, this included 1,506 patients, 961 of who had received the active treatment. Treatment with pramipexole or placebo lasted for between 3 and 12 weeks, with four trials using flexible and two fixed dosing schedules (0.125, 0.25, 0.5, or 0.75 mg/day).
Pramipexole-treated patients were more likely to be identified as IRLS responders (greater than or equal to a 50% reduction in total scores from baseline) than were the patients given placebo (odds ratio = 2.51). Clinicians and patients were also more likely to report higher response rates ("much" or "very much" improved) with the active therapy, with respective odds ratios of 3.13 (P less than .00001) and 2.80 (P = .05).
Pramipexole was well tolerated in patients with primary RLS, the authors say. As expected, nausea and fatigue were the most common adverse events experienced, with respective relative risks of 2.68 (P less than .001) and 1.82 (P = .013) comparing active with placebo treatment.
There were no differences in the incidence of other common adverse effects between the groups, including headache, dizziness, somnolence, or nasopharyngitis.
The researchers note that the trials included in the meta-analysis were all "high quality" and that "might minimize selection and measurement bias." However, the fact that they all used an intention-to-treat approach to avoid overestimating the efficacy effects could mean that the side effects are underestimated, they observed.
The IRLS and other efficacy measures used in the trials were subjective, and future trials could benefit from objective measurement of RLS. At the present time, the only means of doing this is by polysomnography.
Both long-term studies as well as more evidence of head-to-head comparisons of pramipexole with other dopamine agonists, anticonvulsants, and levodopa are needed, the researchers suggested.
The authors reported that they had no relevant conflicts of interest.
The dopamine agonist pramipexole allays the symptoms of primary restless leg syndrome and improves patients’ sleep quality, at least in the short-term, according to a meta-analysis of randomized, double-blind, placebo-controlled clinical trials.
The mean change in restless leg syndrome (RLS) symptoms assessed using the International RLS Study Group Rating Scale (IRLS) was –5.96, compared with placebo (P less than .00001). The mean change in sleep quality assessed by a variety of patient assessment scales was -0.48 (P less than .00001).
RLS can be categorized as primary or secondary, with the latter due to a variety of causes such as pregnancy, uremia, iron deficiency, anemia, or Parkinson’s disease. Managing those conditions effectively may subsequently improve or even cure RLS.
Primary RLS, however, is defined as that due to no other known cause, and it affects between 1.9% and 4.6% of the European and North American adult populations (Sleep Med. Rev. 2012;16:283-95).
Treatments for the condition include lifestyle changes and medications such as pramipexole and other dopamine agonists, levodopa, and painkillers. Results with dopamine agonists to date have been modest.
"Pramipexole has been proven to be a first-line drug for the treatment of movement disorders such as Parkinson’s disease and RLS," observe Dr. Wei Zhang and associates, who undertook the meta-analysis.
The researchers, who are based at the Shengjing Hospital of China Medical University, Shenyang, People’s Republic of China, added that European guidelines state that pramipexole is effective at providing short-term relief but are less certain on the long-term benefits.
"We conducted a meta-analysis in order to summarize the efficacy and tolerability of pramipexole for the treatment of primary RLS," they explained (Neuropsychiatr. Dis. Treat. 2013;9:1035-43).
Six trials published between 2006 and 2012 were identified that met the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement criteria and had used the IRLS as a primary outcome measure.
Altogether, this included 1,506 patients, 961 of who had received the active treatment. Treatment with pramipexole or placebo lasted for between 3 and 12 weeks, with four trials using flexible and two fixed dosing schedules (0.125, 0.25, 0.5, or 0.75 mg/day).
Pramipexole-treated patients were more likely to be identified as IRLS responders (greater than or equal to a 50% reduction in total scores from baseline) than were the patients given placebo (odds ratio = 2.51). Clinicians and patients were also more likely to report higher response rates ("much" or "very much" improved) with the active therapy, with respective odds ratios of 3.13 (P less than .00001) and 2.80 (P = .05).
Pramipexole was well tolerated in patients with primary RLS, the authors say. As expected, nausea and fatigue were the most common adverse events experienced, with respective relative risks of 2.68 (P less than .001) and 1.82 (P = .013) comparing active with placebo treatment.
There were no differences in the incidence of other common adverse effects between the groups, including headache, dizziness, somnolence, or nasopharyngitis.
The researchers note that the trials included in the meta-analysis were all "high quality" and that "might minimize selection and measurement bias." However, the fact that they all used an intention-to-treat approach to avoid overestimating the efficacy effects could mean that the side effects are underestimated, they observed.
The IRLS and other efficacy measures used in the trials were subjective, and future trials could benefit from objective measurement of RLS. At the present time, the only means of doing this is by polysomnography.
Both long-term studies as well as more evidence of head-to-head comparisons of pramipexole with other dopamine agonists, anticonvulsants, and levodopa are needed, the researchers suggested.
The authors reported that they had no relevant conflicts of interest.
The dopamine agonist pramipexole allays the symptoms of primary restless leg syndrome and improves patients’ sleep quality, at least in the short-term, according to a meta-analysis of randomized, double-blind, placebo-controlled clinical trials.
The mean change in restless leg syndrome (RLS) symptoms assessed using the International RLS Study Group Rating Scale (IRLS) was –5.96, compared with placebo (P less than .00001). The mean change in sleep quality assessed by a variety of patient assessment scales was -0.48 (P less than .00001).
RLS can be categorized as primary or secondary, with the latter due to a variety of causes such as pregnancy, uremia, iron deficiency, anemia, or Parkinson’s disease. Managing those conditions effectively may subsequently improve or even cure RLS.
Primary RLS, however, is defined as that due to no other known cause, and it affects between 1.9% and 4.6% of the European and North American adult populations (Sleep Med. Rev. 2012;16:283-95).
Treatments for the condition include lifestyle changes and medications such as pramipexole and other dopamine agonists, levodopa, and painkillers. Results with dopamine agonists to date have been modest.
"Pramipexole has been proven to be a first-line drug for the treatment of movement disorders such as Parkinson’s disease and RLS," observe Dr. Wei Zhang and associates, who undertook the meta-analysis.
The researchers, who are based at the Shengjing Hospital of China Medical University, Shenyang, People’s Republic of China, added that European guidelines state that pramipexole is effective at providing short-term relief but are less certain on the long-term benefits.
"We conducted a meta-analysis in order to summarize the efficacy and tolerability of pramipexole for the treatment of primary RLS," they explained (Neuropsychiatr. Dis. Treat. 2013;9:1035-43).
Six trials published between 2006 and 2012 were identified that met the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement criteria and had used the IRLS as a primary outcome measure.
Altogether, this included 1,506 patients, 961 of who had received the active treatment. Treatment with pramipexole or placebo lasted for between 3 and 12 weeks, with four trials using flexible and two fixed dosing schedules (0.125, 0.25, 0.5, or 0.75 mg/day).
Pramipexole-treated patients were more likely to be identified as IRLS responders (greater than or equal to a 50% reduction in total scores from baseline) than were the patients given placebo (odds ratio = 2.51). Clinicians and patients were also more likely to report higher response rates ("much" or "very much" improved) with the active therapy, with respective odds ratios of 3.13 (P less than .00001) and 2.80 (P = .05).
Pramipexole was well tolerated in patients with primary RLS, the authors say. As expected, nausea and fatigue were the most common adverse events experienced, with respective relative risks of 2.68 (P less than .001) and 1.82 (P = .013) comparing active with placebo treatment.
There were no differences in the incidence of other common adverse effects between the groups, including headache, dizziness, somnolence, or nasopharyngitis.
The researchers note that the trials included in the meta-analysis were all "high quality" and that "might minimize selection and measurement bias." However, the fact that they all used an intention-to-treat approach to avoid overestimating the efficacy effects could mean that the side effects are underestimated, they observed.
The IRLS and other efficacy measures used in the trials were subjective, and future trials could benefit from objective measurement of RLS. At the present time, the only means of doing this is by polysomnography.
Both long-term studies as well as more evidence of head-to-head comparisons of pramipexole with other dopamine agonists, anticonvulsants, and levodopa are needed, the researchers suggested.
The authors reported that they had no relevant conflicts of interest.
FROM NEUROPSYCHIATRIC DISEASE AND TREATMENT
Major finding: Pramipexole improved symptoms and sleep quality to a greater extent than did placebo (both P less than .00001).
Data source: Meta-analysis of six randomized, placebo-controlled trials involving 1,506 patients with primary restless leg syndrome treated with pramipexole (n = 961) or placebo (n = 545).
Disclosures: The authors had no relevant conflicts of interest.
Mobile stroke unit enables prompt prehospital thrombolysis
LONDON – The use of a specialized ambulance enabled more patients with acute ischemic stroke to receive thrombolytic therapy than if standard emergency response vehicles were used in a randomized, controlled trial.
Preliminary results of the PHANTOM-S study also showed that stroke patients were given intravenous tissue plasminogen activator (TPA) 25 minutes sooner in the weeks when the stroke emergency mobile (STEMO) unit was in service, compared with the weeks when it was not.
"We have shown that the STEMO concept can be integrated into a metropolitan emergency medical system," said Dr. Heinrich Audebert, the head of the STEMO project, which is based at Charité-Universitätsmedizin Berlin.
Speaking at the annual European Stroke Conference, Dr. Audebert added that the approach was "safe and superior to regular [emergency] care regarding the quality of care, the proportion of patients receiving TPA, and the time to treatment."
There is further potential to use the mobile stroke unit in the treatment of patients with intracranial hemorrhage and to triage for interventional treatment, he suggested.
Speed is key for stroke thrombolysis
The rationale for the STEMO project, which is based in Berlin, is that thrombolysis is an effective treatment for some patients with acute ischemic stroke. Thrombolytic treatment needs to be administered within a short time frame, Dr. Audebert said, and past a critical time point of 4.5 hours, TPA is no longer given in Germany. The latest guidelines issued jointly by the American Heart Association and the American Stroke Association recommend that TPA be given within 3 hours of stroke onset (Stroke 2013;44:870-947).
However, there are several difficulties in administering TPA in the first few hours after an acute stroke, including determining if the patient is a suitable candidate for thrombolytic therapy, identifying the exact time at which the ischemic injury actually occurred, and transporting the patient quickly enough to a hospital for such treatment.
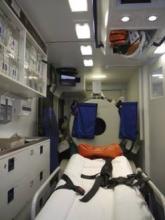
The STEMO project aims to improve stroke patients’ clinical care by enabling TPA to be administered before the patient arrives at hospital. To achieve this, an ambulance owned by the Berlin Fire Department was adapted to accommodate an onboard computed tomography (CT) scanner, point-of-care laboratory, and teleradiography system. This specialized mobile stroke unit is staffed by a paramedic, a neurologist with emergency medicine training, and a radiographer.
The PHANTOM-S study
The PHANTOM-S (Pre-Hospital Acute Neurological Treatment and Optimization of Medical Care in Stroke) study follows on from a 3-month pilot study that provided proof-of-concept for the use of the STEMO unit in 23 hospitals in the Berlin area (Neurology 2013;80:163-8). The larger, randomized study involved 28 hospitals in the region, which provide emergency services to about 1.3 million people.
During the PHANTOM-S study, the STEMO vehicle was in service alternating weeks. The weeks when the specialized ambulance was not deployed were used as the control period. A stroke identification algorithm previously developed by the team (Stroke 2012;43:776-81) was used to decide whether to deploy the STEMO unit to an emergency call.
Over a 21-month study period, 7,098 suspected stroke patients were enrolled and 6,573 were transported to the nearest adequately equipped hospital. Around half were seen in the weeks that the STEMO vehicle was in service.
During the in-service weeks, the STEMO vehicle was deployed a total of 2,027 times out of 3,668 calls. There were a total of 1,804 patients seen by the unit with enough data for study evaluation. Of these, 201 of 614 (33%) patients with acute ischemic stroke were given TPA before reaching the hospital.
A significantly higher percentage of stroke patients received prehospital thrombolysis when the STEMO unit was in service, at 29%, compared with 21% for the control weeks (P less than .001).
The percentage of patients subsequently transported to a hospital with a dedicated stroke unit was also higher during the weeks when the STEMO unit was in service than during the control weeks (93% vs. 87%; P less than .01).
Quicker time to TPA
The mean time from "deployment to needle," said Dr. Audebert, was 77 minutes for the weeks when the specialized ambulance was not in use, compared with 62 minutes when it was (P less than .01). This was reduced to 52 minutes if the STEMO unit was actually deployed (P less than .01 versus control weeks), and dropped to 48 minutes for patients who were actually given TPA in the mobile unit.
In-hospital death rates in confirmed stroke cases were 4.4% for the STEMO in-service weeks (3.8% if the unit was actually deployed) and 4.5% for control weeks.
In TPA-treated patients, there was a nonsignificant trend toward fewer patients having intracranial hemorrhage if TPA was delivered in the STEMO unit, at 3.5%, compared with the overall rates during STEMO-serviced weeks (4.8%) and control weeks (6.0%).
"The current response time is about 16 minutes," coinvestigator Dr. Martin Ebinger, also of Charité-Universitätsmedizin Berlin, commented at a press briefing.
"Sometimes there were multiple, simultaneous calls," he said, noting that, at the current time, there is only one STEMO vehicle. If there were two mobile stroke units, then of course more people in the catchment area could be reached, and the response time could maybe even be halved to 8 minutes.
"We really need a very good, independent cost-effectiveness analysis," Dr. Ebinger conceded. Discussions with patient insurance groups and politicians have been started to determine how best to conduct such an analysis.
Dr. Michael Hennerici, professor of neurology at the University of Heidelberg in Germany, commented at the press briefing that giving thrombolysis to patients who have had a stroke is perhaps more complex than giving it to those who have had a heart attack. There are many different types of stroke, and only a selected population may require thrombolysis, he observed.
Dr. Martin Brown, professor of stroke medicine at University College London, also gave his thoughts on the data in an interview. "It certainly makes sense to get the treatment to patients as quickly as possible, but we need a CT scan of every patient before we give the treatment," he said.
"The real question is, however, ‘Is it more efficient to work on getting the patient to hospital as quickly as possible, or if we should be working on this model and trying to get money for more expensive ambulances?" Dr. Brown added.
"What we really need to know is what difference an extra 25 minutes means in terms of the number of patients that gain benefit. If it is only a small proportion, it might not be cost-effective," Dr. Brown said.
The STEMO project is a collaboration between Charité-Universitätsmedizin Berlin, the Berlin Fire Department, Brahms GmbH, and MEYTEC GmbH. It is cofunded by Technology Foundation Berlin and the European Fund for Regional Development. Dr. Hennerici and Dr. Brown were not involved in the study. Dr. Ebinger had no conflicts of interest. Dr. Audebert has received honoraria from Bayer, Bristol-Myers Squibb, and other companies.
LONDON – The use of a specialized ambulance enabled more patients with acute ischemic stroke to receive thrombolytic therapy than if standard emergency response vehicles were used in a randomized, controlled trial.
Preliminary results of the PHANTOM-S study also showed that stroke patients were given intravenous tissue plasminogen activator (TPA) 25 minutes sooner in the weeks when the stroke emergency mobile (STEMO) unit was in service, compared with the weeks when it was not.
"We have shown that the STEMO concept can be integrated into a metropolitan emergency medical system," said Dr. Heinrich Audebert, the head of the STEMO project, which is based at Charité-Universitätsmedizin Berlin.
Speaking at the annual European Stroke Conference, Dr. Audebert added that the approach was "safe and superior to regular [emergency] care regarding the quality of care, the proportion of patients receiving TPA, and the time to treatment."
There is further potential to use the mobile stroke unit in the treatment of patients with intracranial hemorrhage and to triage for interventional treatment, he suggested.
Speed is key for stroke thrombolysis
The rationale for the STEMO project, which is based in Berlin, is that thrombolysis is an effective treatment for some patients with acute ischemic stroke. Thrombolytic treatment needs to be administered within a short time frame, Dr. Audebert said, and past a critical time point of 4.5 hours, TPA is no longer given in Germany. The latest guidelines issued jointly by the American Heart Association and the American Stroke Association recommend that TPA be given within 3 hours of stroke onset (Stroke 2013;44:870-947).
However, there are several difficulties in administering TPA in the first few hours after an acute stroke, including determining if the patient is a suitable candidate for thrombolytic therapy, identifying the exact time at which the ischemic injury actually occurred, and transporting the patient quickly enough to a hospital for such treatment.
The STEMO project aims to improve stroke patients’ clinical care by enabling TPA to be administered before the patient arrives at hospital. To achieve this, an ambulance owned by the Berlin Fire Department was adapted to accommodate an onboard computed tomography (CT) scanner, point-of-care laboratory, and teleradiography system. This specialized mobile stroke unit is staffed by a paramedic, a neurologist with emergency medicine training, and a radiographer.
The PHANTOM-S study
The PHANTOM-S (Pre-Hospital Acute Neurological Treatment and Optimization of Medical Care in Stroke) study follows on from a 3-month pilot study that provided proof-of-concept for the use of the STEMO unit in 23 hospitals in the Berlin area (Neurology 2013;80:163-8). The larger, randomized study involved 28 hospitals in the region, which provide emergency services to about 1.3 million people.
During the PHANTOM-S study, the STEMO vehicle was in service alternating weeks. The weeks when the specialized ambulance was not deployed were used as the control period. A stroke identification algorithm previously developed by the team (Stroke 2012;43:776-81) was used to decide whether to deploy the STEMO unit to an emergency call.
Over a 21-month study period, 7,098 suspected stroke patients were enrolled and 6,573 were transported to the nearest adequately equipped hospital. Around half were seen in the weeks that the STEMO vehicle was in service.
During the in-service weeks, the STEMO vehicle was deployed a total of 2,027 times out of 3,668 calls. There were a total of 1,804 patients seen by the unit with enough data for study evaluation. Of these, 201 of 614 (33%) patients with acute ischemic stroke were given TPA before reaching the hospital.
A significantly higher percentage of stroke patients received prehospital thrombolysis when the STEMO unit was in service, at 29%, compared with 21% for the control weeks (P less than .001).
The percentage of patients subsequently transported to a hospital with a dedicated stroke unit was also higher during the weeks when the STEMO unit was in service than during the control weeks (93% vs. 87%; P less than .01).
Quicker time to TPA
The mean time from "deployment to needle," said Dr. Audebert, was 77 minutes for the weeks when the specialized ambulance was not in use, compared with 62 minutes when it was (P less than .01). This was reduced to 52 minutes if the STEMO unit was actually deployed (P less than .01 versus control weeks), and dropped to 48 minutes for patients who were actually given TPA in the mobile unit.
In-hospital death rates in confirmed stroke cases were 4.4% for the STEMO in-service weeks (3.8% if the unit was actually deployed) and 4.5% for control weeks.
In TPA-treated patients, there was a nonsignificant trend toward fewer patients having intracranial hemorrhage if TPA was delivered in the STEMO unit, at 3.5%, compared with the overall rates during STEMO-serviced weeks (4.8%) and control weeks (6.0%).
"The current response time is about 16 minutes," coinvestigator Dr. Martin Ebinger, also of Charité-Universitätsmedizin Berlin, commented at a press briefing.
"Sometimes there were multiple, simultaneous calls," he said, noting that, at the current time, there is only one STEMO vehicle. If there were two mobile stroke units, then of course more people in the catchment area could be reached, and the response time could maybe even be halved to 8 minutes.
"We really need a very good, independent cost-effectiveness analysis," Dr. Ebinger conceded. Discussions with patient insurance groups and politicians have been started to determine how best to conduct such an analysis.
Dr. Michael Hennerici, professor of neurology at the University of Heidelberg in Germany, commented at the press briefing that giving thrombolysis to patients who have had a stroke is perhaps more complex than giving it to those who have had a heart attack. There are many different types of stroke, and only a selected population may require thrombolysis, he observed.
Dr. Martin Brown, professor of stroke medicine at University College London, also gave his thoughts on the data in an interview. "It certainly makes sense to get the treatment to patients as quickly as possible, but we need a CT scan of every patient before we give the treatment," he said.
"The real question is, however, ‘Is it more efficient to work on getting the patient to hospital as quickly as possible, or if we should be working on this model and trying to get money for more expensive ambulances?" Dr. Brown added.
"What we really need to know is what difference an extra 25 minutes means in terms of the number of patients that gain benefit. If it is only a small proportion, it might not be cost-effective," Dr. Brown said.
The STEMO project is a collaboration between Charité-Universitätsmedizin Berlin, the Berlin Fire Department, Brahms GmbH, and MEYTEC GmbH. It is cofunded by Technology Foundation Berlin and the European Fund for Regional Development. Dr. Hennerici and Dr. Brown were not involved in the study. Dr. Ebinger had no conflicts of interest. Dr. Audebert has received honoraria from Bayer, Bristol-Myers Squibb, and other companies.
LONDON – The use of a specialized ambulance enabled more patients with acute ischemic stroke to receive thrombolytic therapy than if standard emergency response vehicles were used in a randomized, controlled trial.
Preliminary results of the PHANTOM-S study also showed that stroke patients were given intravenous tissue plasminogen activator (TPA) 25 minutes sooner in the weeks when the stroke emergency mobile (STEMO) unit was in service, compared with the weeks when it was not.
"We have shown that the STEMO concept can be integrated into a metropolitan emergency medical system," said Dr. Heinrich Audebert, the head of the STEMO project, which is based at Charité-Universitätsmedizin Berlin.
Speaking at the annual European Stroke Conference, Dr. Audebert added that the approach was "safe and superior to regular [emergency] care regarding the quality of care, the proportion of patients receiving TPA, and the time to treatment."
There is further potential to use the mobile stroke unit in the treatment of patients with intracranial hemorrhage and to triage for interventional treatment, he suggested.
Speed is key for stroke thrombolysis
The rationale for the STEMO project, which is based in Berlin, is that thrombolysis is an effective treatment for some patients with acute ischemic stroke. Thrombolytic treatment needs to be administered within a short time frame, Dr. Audebert said, and past a critical time point of 4.5 hours, TPA is no longer given in Germany. The latest guidelines issued jointly by the American Heart Association and the American Stroke Association recommend that TPA be given within 3 hours of stroke onset (Stroke 2013;44:870-947).
However, there are several difficulties in administering TPA in the first few hours after an acute stroke, including determining if the patient is a suitable candidate for thrombolytic therapy, identifying the exact time at which the ischemic injury actually occurred, and transporting the patient quickly enough to a hospital for such treatment.
The STEMO project aims to improve stroke patients’ clinical care by enabling TPA to be administered before the patient arrives at hospital. To achieve this, an ambulance owned by the Berlin Fire Department was adapted to accommodate an onboard computed tomography (CT) scanner, point-of-care laboratory, and teleradiography system. This specialized mobile stroke unit is staffed by a paramedic, a neurologist with emergency medicine training, and a radiographer.
The PHANTOM-S study
The PHANTOM-S (Pre-Hospital Acute Neurological Treatment and Optimization of Medical Care in Stroke) study follows on from a 3-month pilot study that provided proof-of-concept for the use of the STEMO unit in 23 hospitals in the Berlin area (Neurology 2013;80:163-8). The larger, randomized study involved 28 hospitals in the region, which provide emergency services to about 1.3 million people.
During the PHANTOM-S study, the STEMO vehicle was in service alternating weeks. The weeks when the specialized ambulance was not deployed were used as the control period. A stroke identification algorithm previously developed by the team (Stroke 2012;43:776-81) was used to decide whether to deploy the STEMO unit to an emergency call.
Over a 21-month study period, 7,098 suspected stroke patients were enrolled and 6,573 were transported to the nearest adequately equipped hospital. Around half were seen in the weeks that the STEMO vehicle was in service.
During the in-service weeks, the STEMO vehicle was deployed a total of 2,027 times out of 3,668 calls. There were a total of 1,804 patients seen by the unit with enough data for study evaluation. Of these, 201 of 614 (33%) patients with acute ischemic stroke were given TPA before reaching the hospital.
A significantly higher percentage of stroke patients received prehospital thrombolysis when the STEMO unit was in service, at 29%, compared with 21% for the control weeks (P less than .001).
The percentage of patients subsequently transported to a hospital with a dedicated stroke unit was also higher during the weeks when the STEMO unit was in service than during the control weeks (93% vs. 87%; P less than .01).
Quicker time to TPA
The mean time from "deployment to needle," said Dr. Audebert, was 77 minutes for the weeks when the specialized ambulance was not in use, compared with 62 minutes when it was (P less than .01). This was reduced to 52 minutes if the STEMO unit was actually deployed (P less than .01 versus control weeks), and dropped to 48 minutes for patients who were actually given TPA in the mobile unit.
In-hospital death rates in confirmed stroke cases were 4.4% for the STEMO in-service weeks (3.8% if the unit was actually deployed) and 4.5% for control weeks.
In TPA-treated patients, there was a nonsignificant trend toward fewer patients having intracranial hemorrhage if TPA was delivered in the STEMO unit, at 3.5%, compared with the overall rates during STEMO-serviced weeks (4.8%) and control weeks (6.0%).
"The current response time is about 16 minutes," coinvestigator Dr. Martin Ebinger, also of Charité-Universitätsmedizin Berlin, commented at a press briefing.
"Sometimes there were multiple, simultaneous calls," he said, noting that, at the current time, there is only one STEMO vehicle. If there were two mobile stroke units, then of course more people in the catchment area could be reached, and the response time could maybe even be halved to 8 minutes.
"We really need a very good, independent cost-effectiveness analysis," Dr. Ebinger conceded. Discussions with patient insurance groups and politicians have been started to determine how best to conduct such an analysis.
Dr. Michael Hennerici, professor of neurology at the University of Heidelberg in Germany, commented at the press briefing that giving thrombolysis to patients who have had a stroke is perhaps more complex than giving it to those who have had a heart attack. There are many different types of stroke, and only a selected population may require thrombolysis, he observed.
Dr. Martin Brown, professor of stroke medicine at University College London, also gave his thoughts on the data in an interview. "It certainly makes sense to get the treatment to patients as quickly as possible, but we need a CT scan of every patient before we give the treatment," he said.
"The real question is, however, ‘Is it more efficient to work on getting the patient to hospital as quickly as possible, or if we should be working on this model and trying to get money for more expensive ambulances?" Dr. Brown added.
"What we really need to know is what difference an extra 25 minutes means in terms of the number of patients that gain benefit. If it is only a small proportion, it might not be cost-effective," Dr. Brown said.
The STEMO project is a collaboration between Charité-Universitätsmedizin Berlin, the Berlin Fire Department, Brahms GmbH, and MEYTEC GmbH. It is cofunded by Technology Foundation Berlin and the European Fund for Regional Development. Dr. Hennerici and Dr. Brown were not involved in the study. Dr. Ebinger had no conflicts of interest. Dr. Audebert has received honoraria from Bayer, Bristol-Myers Squibb, and other companies.
AT THE EUROPEAN STROKE CONFERENCE
Major finding: The time from an emergency call to appropriate TPA initiation was reduced by 25 minutes through the use of a stroke emergency mobile unit.
Data source: PHANTOM-S, a randomized controlled trial involving more than 7,000 stroke patients treated in a specially equipped ambulance or via normal emergency services.
Disclosures: The STEMO project is a collaboration between Charité-Universitätsmedizin Berlin, the Berlin Fire Department, Brahms GmbH, and MEYTEC GmbH. It is cofunded by Technology Foundation Berlin and the European Fund for Regional Development. Dr. Hennerici and Dr. Brown were not involved in the study. Dr. Ebinger had no conflicts of interest. Dr. Audebert has received honoraria from Bayer, Bristol-Myers Squibb, and other companies.
Time-lapse imaging said to improve embryo selection
LONDON – Time-lapse imaging can help select the best embryo for implantation during fertility treatment, according to Joseph Conaghan, Ph.D., of the Pacific Fertility Center in San Francisco.
"Improved embryo selection may increase pregnancy rates following IVF [in vitro fertilization] and allow more patients to elect for single-embryo transfer [SET]," he noted at the annual meeting of the European Society of Human Reproduction and Embryology.
In recent years, there has been a move toward elective SET during IVF, but the main stumbling block has been that it is difficult to identify the best embryo to transfer.
"Historically, we have not had good pregnancy rates with frozen embryos, and it is always hard to convince patients to transfer just one embryo if they think that their chance of pregnancy is lowered," Dr. Conaghan said.
Although there are several methods available to try to select the most viable embryos, the one that Dr. Conaghan has been involved in testing in particular is time-lapse imaging. "As an embryologist, I like this technology a lot," he said. "The camera is seeing what we see, and we understand the information that we are getting."
The technology is based on identifying very early kinetic markers of embryonic development (Fertil. Steril. 2013;99:1035-43). Data show that how, and the rate at which, cells initially divide to form four-celled embryos can predict how well the blastocyst will form and later survive. Indeed, the duration of the first cytokinesis, the time between an embryo becoming three from two cells, and four from three cells, are all important stages (Nat. Biotechnol. 2010;28:1115-21).
In a recent prospective, multicenter cohort study (Fertil. Steril. 2013 [doi:10.1016/j.fertnstert.2013.04.021]), Dr. Conaghan and his associates reported on the ability of a time-lapse imaging technology to help select embryos with good morphology versus embryologists’ usual screening assessment of embryo morphology at day 3.
The technology used in the study was the Eeva (Early Embryo Viability Assessment) system developed by Auxogyn. This is one of several systems currently available; another is the EmbryoScope produced by the Danish company Unisense FertiliTech. They use computer-aided tracking to visualize the dynamic development of multiple embryos simultaneously.
While the EmbyroScopeis a self-contained system that sits on the lab bench, Eevais a microscope system that fits inside a standard incubator and an electronic touch-screen pad fits on the outside to allow the embryologist to see images taken by the microscope at 5-minute intervals.
Discussing Eeva, Dr. Conaghan explained: "The computer tracks development and tells you when an embryo has a high/low probability of making a blastocyst." Embryos that have a low probability of forming a healthy blastocyst are flagged by a yellow square whereas embryos with a high or good probability of forming a healthy blastocyst are flagged by a green square.
Five IVF clinics in the United States participated in the study, recruiting 160 women between them. Three experienced embryologists assessed patient records and morphology at day 3 for 775 embryos that had been separately evaluated using the Eeva system. There was no blinding to the Eeva results as the aim was to see if the technology could improve prediction rates for embryos that would have the best chance of implantation and a successful pregnancy.
The additional use of the time-lapse imaging technology was found to significantly improve the embryologists’ ability to identify embryos that would reach the blastocyst stage. The specificity for using day 3 morphology alone was 41.9%, 59.7%, and 79.5% for each of the three embryologists respectively. When Eeva was added into the mix, the rates increased to a respective 84.0%, 86.3%, and 86.6%.
Importantly, the additional use of Eeva was able to help embryologists to identify "good morphology" embryos, Dr. Conaghan said. These are embryos that have divided to become six or more cells, with perfect symmetry and less than 10% fragmentation.
"It’s important to point out that if an embryo does get a low score that it doesn’t mean that it is going to fail, he said. "It means that there is a high probability that the embryo is [perhaps] going to fail, but it doesn’t mean that we’re going to throw it away."
The ongoing studies – EPIC(Eeva Pregnancy Investigational Clinical Study), MERGE (Registry Study of Traditional Morphology Grading Combined With Eeva in IVF Treatment), and CCS(Correlating Time-Lapse Parameters Detected by the Eeva System With Comprehensive Chromosome Screening Results) – are assessing the use of the time-lapse imaging system, looking at aneuploidy, fragmentation, and the prospective use of Eeva to assist in embryo selection for transfer.
"I think that this is a very exciting technology," Dr. Conaghan said. "It has the potential to eliminate embryos from transfer consideration."
During a separate session, Catherine Racowksy, Ph.D., of Brigham and Women’s Hospital in Boston, made some general comments on the use of time-lapse imaging for optimizing the selection of embryos for IVF. "Time-lapse imaging really does hold a great deal of promise," she said. It is a noninvasive approach, and several teams have shown that the kinetic time points being assessed can be replicated.
"The tracking algorithms derived from retrospective cohort imaging data show that there are these key early cell cycle parameters that have value in predicting blastocyst formation and the ploidy of the embryo," Dr. Racowsky commented.
Prospective, randomized trial data are critical before this technology hits routine practice. "We cannot have these machines in the lab, hoping that they are going to help us in predicting the development potential of embryos if we haven’t proven to ourselves that they really are better than just looking down a microscope at static time points," she concluded.
Dr. Conaghan and Dr. Racowsky reported having no relevant financial disclosures.
LONDON – Time-lapse imaging can help select the best embryo for implantation during fertility treatment, according to Joseph Conaghan, Ph.D., of the Pacific Fertility Center in San Francisco.
"Improved embryo selection may increase pregnancy rates following IVF [in vitro fertilization] and allow more patients to elect for single-embryo transfer [SET]," he noted at the annual meeting of the European Society of Human Reproduction and Embryology.
In recent years, there has been a move toward elective SET during IVF, but the main stumbling block has been that it is difficult to identify the best embryo to transfer.
"Historically, we have not had good pregnancy rates with frozen embryos, and it is always hard to convince patients to transfer just one embryo if they think that their chance of pregnancy is lowered," Dr. Conaghan said.
Although there are several methods available to try to select the most viable embryos, the one that Dr. Conaghan has been involved in testing in particular is time-lapse imaging. "As an embryologist, I like this technology a lot," he said. "The camera is seeing what we see, and we understand the information that we are getting."
The technology is based on identifying very early kinetic markers of embryonic development (Fertil. Steril. 2013;99:1035-43). Data show that how, and the rate at which, cells initially divide to form four-celled embryos can predict how well the blastocyst will form and later survive. Indeed, the duration of the first cytokinesis, the time between an embryo becoming three from two cells, and four from three cells, are all important stages (Nat. Biotechnol. 2010;28:1115-21).
In a recent prospective, multicenter cohort study (Fertil. Steril. 2013 [doi:10.1016/j.fertnstert.2013.04.021]), Dr. Conaghan and his associates reported on the ability of a time-lapse imaging technology to help select embryos with good morphology versus embryologists’ usual screening assessment of embryo morphology at day 3.
The technology used in the study was the Eeva (Early Embryo Viability Assessment) system developed by Auxogyn. This is one of several systems currently available; another is the EmbryoScope produced by the Danish company Unisense FertiliTech. They use computer-aided tracking to visualize the dynamic development of multiple embryos simultaneously.
While the EmbyroScopeis a self-contained system that sits on the lab bench, Eevais a microscope system that fits inside a standard incubator and an electronic touch-screen pad fits on the outside to allow the embryologist to see images taken by the microscope at 5-minute intervals.
Discussing Eeva, Dr. Conaghan explained: "The computer tracks development and tells you when an embryo has a high/low probability of making a blastocyst." Embryos that have a low probability of forming a healthy blastocyst are flagged by a yellow square whereas embryos with a high or good probability of forming a healthy blastocyst are flagged by a green square.
Five IVF clinics in the United States participated in the study, recruiting 160 women between them. Three experienced embryologists assessed patient records and morphology at day 3 for 775 embryos that had been separately evaluated using the Eeva system. There was no blinding to the Eeva results as the aim was to see if the technology could improve prediction rates for embryos that would have the best chance of implantation and a successful pregnancy.
The additional use of the time-lapse imaging technology was found to significantly improve the embryologists’ ability to identify embryos that would reach the blastocyst stage. The specificity for using day 3 morphology alone was 41.9%, 59.7%, and 79.5% for each of the three embryologists respectively. When Eeva was added into the mix, the rates increased to a respective 84.0%, 86.3%, and 86.6%.
Importantly, the additional use of Eeva was able to help embryologists to identify "good morphology" embryos, Dr. Conaghan said. These are embryos that have divided to become six or more cells, with perfect symmetry and less than 10% fragmentation.
"It’s important to point out that if an embryo does get a low score that it doesn’t mean that it is going to fail, he said. "It means that there is a high probability that the embryo is [perhaps] going to fail, but it doesn’t mean that we’re going to throw it away."
The ongoing studies – EPIC(Eeva Pregnancy Investigational Clinical Study), MERGE (Registry Study of Traditional Morphology Grading Combined With Eeva in IVF Treatment), and CCS(Correlating Time-Lapse Parameters Detected by the Eeva System With Comprehensive Chromosome Screening Results) – are assessing the use of the time-lapse imaging system, looking at aneuploidy, fragmentation, and the prospective use of Eeva to assist in embryo selection for transfer.
"I think that this is a very exciting technology," Dr. Conaghan said. "It has the potential to eliminate embryos from transfer consideration."
During a separate session, Catherine Racowksy, Ph.D., of Brigham and Women’s Hospital in Boston, made some general comments on the use of time-lapse imaging for optimizing the selection of embryos for IVF. "Time-lapse imaging really does hold a great deal of promise," she said. It is a noninvasive approach, and several teams have shown that the kinetic time points being assessed can be replicated.
"The tracking algorithms derived from retrospective cohort imaging data show that there are these key early cell cycle parameters that have value in predicting blastocyst formation and the ploidy of the embryo," Dr. Racowsky commented.
Prospective, randomized trial data are critical before this technology hits routine practice. "We cannot have these machines in the lab, hoping that they are going to help us in predicting the development potential of embryos if we haven’t proven to ourselves that they really are better than just looking down a microscope at static time points," she concluded.
Dr. Conaghan and Dr. Racowsky reported having no relevant financial disclosures.
LONDON – Time-lapse imaging can help select the best embryo for implantation during fertility treatment, according to Joseph Conaghan, Ph.D., of the Pacific Fertility Center in San Francisco.
"Improved embryo selection may increase pregnancy rates following IVF [in vitro fertilization] and allow more patients to elect for single-embryo transfer [SET]," he noted at the annual meeting of the European Society of Human Reproduction and Embryology.
In recent years, there has been a move toward elective SET during IVF, but the main stumbling block has been that it is difficult to identify the best embryo to transfer.
"Historically, we have not had good pregnancy rates with frozen embryos, and it is always hard to convince patients to transfer just one embryo if they think that their chance of pregnancy is lowered," Dr. Conaghan said.
Although there are several methods available to try to select the most viable embryos, the one that Dr. Conaghan has been involved in testing in particular is time-lapse imaging. "As an embryologist, I like this technology a lot," he said. "The camera is seeing what we see, and we understand the information that we are getting."
The technology is based on identifying very early kinetic markers of embryonic development (Fertil. Steril. 2013;99:1035-43). Data show that how, and the rate at which, cells initially divide to form four-celled embryos can predict how well the blastocyst will form and later survive. Indeed, the duration of the first cytokinesis, the time between an embryo becoming three from two cells, and four from three cells, are all important stages (Nat. Biotechnol. 2010;28:1115-21).
In a recent prospective, multicenter cohort study (Fertil. Steril. 2013 [doi:10.1016/j.fertnstert.2013.04.021]), Dr. Conaghan and his associates reported on the ability of a time-lapse imaging technology to help select embryos with good morphology versus embryologists’ usual screening assessment of embryo morphology at day 3.
The technology used in the study was the Eeva (Early Embryo Viability Assessment) system developed by Auxogyn. This is one of several systems currently available; another is the EmbryoScope produced by the Danish company Unisense FertiliTech. They use computer-aided tracking to visualize the dynamic development of multiple embryos simultaneously.
While the EmbyroScopeis a self-contained system that sits on the lab bench, Eevais a microscope system that fits inside a standard incubator and an electronic touch-screen pad fits on the outside to allow the embryologist to see images taken by the microscope at 5-minute intervals.
Discussing Eeva, Dr. Conaghan explained: "The computer tracks development and tells you when an embryo has a high/low probability of making a blastocyst." Embryos that have a low probability of forming a healthy blastocyst are flagged by a yellow square whereas embryos with a high or good probability of forming a healthy blastocyst are flagged by a green square.
Five IVF clinics in the United States participated in the study, recruiting 160 women between them. Three experienced embryologists assessed patient records and morphology at day 3 for 775 embryos that had been separately evaluated using the Eeva system. There was no blinding to the Eeva results as the aim was to see if the technology could improve prediction rates for embryos that would have the best chance of implantation and a successful pregnancy.
The additional use of the time-lapse imaging technology was found to significantly improve the embryologists’ ability to identify embryos that would reach the blastocyst stage. The specificity for using day 3 morphology alone was 41.9%, 59.7%, and 79.5% for each of the three embryologists respectively. When Eeva was added into the mix, the rates increased to a respective 84.0%, 86.3%, and 86.6%.
Importantly, the additional use of Eeva was able to help embryologists to identify "good morphology" embryos, Dr. Conaghan said. These are embryos that have divided to become six or more cells, with perfect symmetry and less than 10% fragmentation.
"It’s important to point out that if an embryo does get a low score that it doesn’t mean that it is going to fail, he said. "It means that there is a high probability that the embryo is [perhaps] going to fail, but it doesn’t mean that we’re going to throw it away."
The ongoing studies – EPIC(Eeva Pregnancy Investigational Clinical Study), MERGE (Registry Study of Traditional Morphology Grading Combined With Eeva in IVF Treatment), and CCS(Correlating Time-Lapse Parameters Detected by the Eeva System With Comprehensive Chromosome Screening Results) – are assessing the use of the time-lapse imaging system, looking at aneuploidy, fragmentation, and the prospective use of Eeva to assist in embryo selection for transfer.
"I think that this is a very exciting technology," Dr. Conaghan said. "It has the potential to eliminate embryos from transfer consideration."
During a separate session, Catherine Racowksy, Ph.D., of Brigham and Women’s Hospital in Boston, made some general comments on the use of time-lapse imaging for optimizing the selection of embryos for IVF. "Time-lapse imaging really does hold a great deal of promise," she said. It is a noninvasive approach, and several teams have shown that the kinetic time points being assessed can be replicated.
"The tracking algorithms derived from retrospective cohort imaging data show that there are these key early cell cycle parameters that have value in predicting blastocyst formation and the ploidy of the embryo," Dr. Racowsky commented.
Prospective, randomized trial data are critical before this technology hits routine practice. "We cannot have these machines in the lab, hoping that they are going to help us in predicting the development potential of embryos if we haven’t proven to ourselves that they really are better than just looking down a microscope at static time points," she concluded.
Dr. Conaghan and Dr. Racowsky reported having no relevant financial disclosures.
EXPERT ANALYSIS FROM ESHRE 2013
Long-term depression may affect ability to conceive
LONDON – Long-standing depression may influence women’s ability to conceive, data from a large, population-based, Swedish cohort study suggest.
A significantly longer time to pregnancy was seen in women with a first diagnosis of depression occurring 6 years or more previously, with a crude odds ratio (OR) of 2.03 and an adjusted OR of 1.25.
"This study suggests that long-term psychological distress may lead to longer time to first pregnancy," said Carolyn Cesta, a doctoral student at the Karolinska Institute in Stockholm, where the study was performed.
The study also found that difficulty conceiving was higher in these women with long-term depression who were not treated with selective serotonin reuptake inhibitors (SSRIs), with an adjusted OR of 1.44 versus 1.37 for SSRI use.
"The effects of antidepressant treatment on fertility should be further investigated," Ms. Cesta suggested when presenting the findings at the annual meeting of the European Society of Human Reproduction and Embryology.
There are data to suggest that women with a history of depression are almost twice as likely as are those without it to report decreased fertility, but the data are conflicting. The main objective of the study presented was to investigate the controversial issue further: specifically, to look at whether psychological distress – measured as a diagnosis of depression, anxiety, or both – could predict women’s difficulties to conceive.
"Women during their childbearing years are at their greatest lifetime risk for being diagnosed with depression and anxiety," Ms. Cesta observed, "so it becomes important to know the impact of these conditions on fertility.
All together, data on 1,147,655 women who had delivered live-born infants between 1983 and 2009 were analyzed. These women were identified from several national Swedish registers: the medical birth register (1983-2009), the National Patient Register (1964-2009), the Total Population Register, and the Prescription Drug Register (2005-2009).
Women were then divided by the presence or absence of depression or anxiety reported at their first prenatal visit. There were 1,130,285 women with no such diagnoses, 7,322 with a diagnosis of depression, 7,551 with anxiety, and 2,497 with both disorders.
Involuntary childlessness, defined as the desire to conceive a child but difficulty doing so for 1 year or more, was reported by 6.7% of the overall population, with higher rates observed in women with depression (7.7%), anxiety (7.7%), or both (7.5%) than in women with no depression or anxiety (6.7%).
"We saw that there was no association between having a diagnosis of depression and/or anxiety and involuntary childlessness for more than 1 year when we compared them to women with no diagnosis," Ms. Cesta said.
Adjusted OR for a diagnosis of depression less than1.5 years, 1.5-3 years, and 3-6 years before pregnancy were 0.81, 0.92, and 1.06, respectively.
"The only significant association was seen where women diagnosed with depression had been diagnosed 6 years prior to pregnancy," Ms. Cesta said. They "had a significantly higher risk of involuntary childlessness." She noted that depression had been diagnosed before becoming pregnant in 91% of women.
Data on SSRI use in the women with depression for 6 or more years was available from 2005 onward, so this might have limited the analysis because of small patient numbers for inclusion in the analysis.
"This was a large, register-based cohort study of over 1 million women for which we have data for a large number of known confounders and the association between fertility and depression," Ms Cesta reported.
One of the main limitations, however, was that the study population includes women who have had at least one child already and not those who are perhaps infertile and remain childless.
The study was funded by the Swedish Research Council, the European Union Seventh Framework Program, and the Karolinska Institute. Ms. Cesta had no conflicts of interest to disclose.
LONDON – Long-standing depression may influence women’s ability to conceive, data from a large, population-based, Swedish cohort study suggest.
A significantly longer time to pregnancy was seen in women with a first diagnosis of depression occurring 6 years or more previously, with a crude odds ratio (OR) of 2.03 and an adjusted OR of 1.25.
"This study suggests that long-term psychological distress may lead to longer time to first pregnancy," said Carolyn Cesta, a doctoral student at the Karolinska Institute in Stockholm, where the study was performed.
The study also found that difficulty conceiving was higher in these women with long-term depression who were not treated with selective serotonin reuptake inhibitors (SSRIs), with an adjusted OR of 1.44 versus 1.37 for SSRI use.
"The effects of antidepressant treatment on fertility should be further investigated," Ms. Cesta suggested when presenting the findings at the annual meeting of the European Society of Human Reproduction and Embryology.
There are data to suggest that women with a history of depression are almost twice as likely as are those without it to report decreased fertility, but the data are conflicting. The main objective of the study presented was to investigate the controversial issue further: specifically, to look at whether psychological distress – measured as a diagnosis of depression, anxiety, or both – could predict women’s difficulties to conceive.
"Women during their childbearing years are at their greatest lifetime risk for being diagnosed with depression and anxiety," Ms. Cesta observed, "so it becomes important to know the impact of these conditions on fertility.
All together, data on 1,147,655 women who had delivered live-born infants between 1983 and 2009 were analyzed. These women were identified from several national Swedish registers: the medical birth register (1983-2009), the National Patient Register (1964-2009), the Total Population Register, and the Prescription Drug Register (2005-2009).
Women were then divided by the presence or absence of depression or anxiety reported at their first prenatal visit. There were 1,130,285 women with no such diagnoses, 7,322 with a diagnosis of depression, 7,551 with anxiety, and 2,497 with both disorders.
Involuntary childlessness, defined as the desire to conceive a child but difficulty doing so for 1 year or more, was reported by 6.7% of the overall population, with higher rates observed in women with depression (7.7%), anxiety (7.7%), or both (7.5%) than in women with no depression or anxiety (6.7%).
"We saw that there was no association between having a diagnosis of depression and/or anxiety and involuntary childlessness for more than 1 year when we compared them to women with no diagnosis," Ms. Cesta said.
Adjusted OR for a diagnosis of depression less than1.5 years, 1.5-3 years, and 3-6 years before pregnancy were 0.81, 0.92, and 1.06, respectively.
"The only significant association was seen where women diagnosed with depression had been diagnosed 6 years prior to pregnancy," Ms. Cesta said. They "had a significantly higher risk of involuntary childlessness." She noted that depression had been diagnosed before becoming pregnant in 91% of women.
Data on SSRI use in the women with depression for 6 or more years was available from 2005 onward, so this might have limited the analysis because of small patient numbers for inclusion in the analysis.
"This was a large, register-based cohort study of over 1 million women for which we have data for a large number of known confounders and the association between fertility and depression," Ms Cesta reported.
One of the main limitations, however, was that the study population includes women who have had at least one child already and not those who are perhaps infertile and remain childless.
The study was funded by the Swedish Research Council, the European Union Seventh Framework Program, and the Karolinska Institute. Ms. Cesta had no conflicts of interest to disclose.
LONDON – Long-standing depression may influence women’s ability to conceive, data from a large, population-based, Swedish cohort study suggest.
A significantly longer time to pregnancy was seen in women with a first diagnosis of depression occurring 6 years or more previously, with a crude odds ratio (OR) of 2.03 and an adjusted OR of 1.25.
"This study suggests that long-term psychological distress may lead to longer time to first pregnancy," said Carolyn Cesta, a doctoral student at the Karolinska Institute in Stockholm, where the study was performed.
The study also found that difficulty conceiving was higher in these women with long-term depression who were not treated with selective serotonin reuptake inhibitors (SSRIs), with an adjusted OR of 1.44 versus 1.37 for SSRI use.
"The effects of antidepressant treatment on fertility should be further investigated," Ms. Cesta suggested when presenting the findings at the annual meeting of the European Society of Human Reproduction and Embryology.
There are data to suggest that women with a history of depression are almost twice as likely as are those without it to report decreased fertility, but the data are conflicting. The main objective of the study presented was to investigate the controversial issue further: specifically, to look at whether psychological distress – measured as a diagnosis of depression, anxiety, or both – could predict women’s difficulties to conceive.
"Women during their childbearing years are at their greatest lifetime risk for being diagnosed with depression and anxiety," Ms. Cesta observed, "so it becomes important to know the impact of these conditions on fertility.
All together, data on 1,147,655 women who had delivered live-born infants between 1983 and 2009 were analyzed. These women were identified from several national Swedish registers: the medical birth register (1983-2009), the National Patient Register (1964-2009), the Total Population Register, and the Prescription Drug Register (2005-2009).
Women were then divided by the presence or absence of depression or anxiety reported at their first prenatal visit. There were 1,130,285 women with no such diagnoses, 7,322 with a diagnosis of depression, 7,551 with anxiety, and 2,497 with both disorders.
Involuntary childlessness, defined as the desire to conceive a child but difficulty doing so for 1 year or more, was reported by 6.7% of the overall population, with higher rates observed in women with depression (7.7%), anxiety (7.7%), or both (7.5%) than in women with no depression or anxiety (6.7%).
"We saw that there was no association between having a diagnosis of depression and/or anxiety and involuntary childlessness for more than 1 year when we compared them to women with no diagnosis," Ms. Cesta said.
Adjusted OR for a diagnosis of depression less than1.5 years, 1.5-3 years, and 3-6 years before pregnancy were 0.81, 0.92, and 1.06, respectively.
"The only significant association was seen where women diagnosed with depression had been diagnosed 6 years prior to pregnancy," Ms. Cesta said. They "had a significantly higher risk of involuntary childlessness." She noted that depression had been diagnosed before becoming pregnant in 91% of women.
Data on SSRI use in the women with depression for 6 or more years was available from 2005 onward, so this might have limited the analysis because of small patient numbers for inclusion in the analysis.
"This was a large, register-based cohort study of over 1 million women for which we have data for a large number of known confounders and the association between fertility and depression," Ms Cesta reported.
One of the main limitations, however, was that the study population includes women who have had at least one child already and not those who are perhaps infertile and remain childless.
The study was funded by the Swedish Research Council, the European Union Seventh Framework Program, and the Karolinska Institute. Ms. Cesta had no conflicts of interest to disclose.
AT ESHRE 2013
Major finding: Six-plus years of depression was associated with a longer time to pregnancy than was no depression (adjusted OR 1.25).
Data source: Swedish cohort-based register of more than 1,00,000 involuntarily childless women.
Disclosures: The study was funded by the Swedish Research Council, the European Union Seventh Framework Program, and the Karolinksa Institute. Ms. Cesta had no conflicts of interest to disclose.
Low-molecular-weight heparin improves fertility treatment outcomes
LONDON – Low-molecular-weight heparin therapy reduced the risk for recurrent implantation failure, improved pregnancy rates, and increased the number of live births in women who had failed prior fertility treatment in a 126-patient pilot study.
As reported at the annual meeting of the European Society of Human Reproduction and Embryology, low-molecular-weight heparin (LMWH) was associated with an implantation rate of 22.2% vs. 10% for a regimen containing aspirin (P =.0136). Pregnancy (47% vs. 20.6%, P = .0026) and live birth rates (30% vs. 12%, P = .017) also were higher with the LMWH than with the aspirin-containing regimen.
Although larger, randomized, double blind controlled trials are needed to confirm the findings, the study’s Italian investigators are confident that LMWH has a benefit in improving fertility outcomes in this population of women who had failed two or more cycles of in vitro fertilization (IVF).
"We are convinced that low-molecular-weight heparin can enhance results in IVF procedures," said Dr. Antonio Colicchia of the Centro Fecondazione Assistita Ferticlinic, Rome, in an interview.
Strategies to improve implantation rates are needed, Dr. Colicchia explained, because there has been a plateau in live birth rates associated with assisted reproductive technology (ART). Approximately 4.2% of babies born in Europe are a consequence of ART, he said.
Women were included in the single-center study over a 2-year period starting in September 2000. For inclusion they had to have a body mass index lower than 25 mg/kg2, be between 31 and 42 years old, have failed at least two cycles of IVF, and not have inherited thrombophilia.
Sixty-eight women were allocated to treatment with the LMWH dalteparin at a dose of 2,500 IU/day and 58 to treatment with aspirin at a dose of 100 mg/day and oral prednisone at a dose of 10 mg/day. Both groups of women also were given vaginal progesterone at a dose of 600 mg/day.
Treatment began at the time of embryo transfer and was continued until levels of human chorionic gonadotropin confirmed a pregnancy. If a pregnancy was confirmed, then treatment was continued until the 12th gestational week. Women administered the treatments at home.
"Heparin can potentially modulate many of the known mechanisms that underlie successful apposition, adhesion, and penetration of the developing embryo," Dr. Colicchia said.
"There is evidence that in women with repeated IVF failure, [LMWH] can improve pregnancy rates," he concluded, although further studies are needed.
The chairman of the session in which the study results were presented was critical of the findings. "This was not a randomized nor a placebo-controlled trial," Dr. Thomas D’Hooghe of Leuven (Belgium) University fertility center said in an interview. Furthermore, the control arm was flawed, as aspirin and oral prednisone are not routinely used.
Elsewhere at the annual meeting of ESHRE, a German team presented data from a 12-center trial (n = 449) on the use of LMWH in the prevention of recurrent miscarriage. The results of the ETHIG II study, in which dalteparin (5,000 IU/day) plus multivitamins were compared with vitamins alone, were not positive.
There was no difference in the primary outcome measure of the number of ongoing pregnancies at 24 complete weeks of gestation, leading the authors to conclude that "the uncontrolled use of LMWH in patients with recurrent miscarriages is not reasonable."
There was no commercial funding for the study presented by Dr. Colicchia. He reported having no relevant financial disclosures. Dr. D’Hooghe was not involved in the study and said he had no relevant conflicts of interest.
LONDON – Low-molecular-weight heparin therapy reduced the risk for recurrent implantation failure, improved pregnancy rates, and increased the number of live births in women who had failed prior fertility treatment in a 126-patient pilot study.
As reported at the annual meeting of the European Society of Human Reproduction and Embryology, low-molecular-weight heparin (LMWH) was associated with an implantation rate of 22.2% vs. 10% for a regimen containing aspirin (P =.0136). Pregnancy (47% vs. 20.6%, P = .0026) and live birth rates (30% vs. 12%, P = .017) also were higher with the LMWH than with the aspirin-containing regimen.
Although larger, randomized, double blind controlled trials are needed to confirm the findings, the study’s Italian investigators are confident that LMWH has a benefit in improving fertility outcomes in this population of women who had failed two or more cycles of in vitro fertilization (IVF).
"We are convinced that low-molecular-weight heparin can enhance results in IVF procedures," said Dr. Antonio Colicchia of the Centro Fecondazione Assistita Ferticlinic, Rome, in an interview.
Strategies to improve implantation rates are needed, Dr. Colicchia explained, because there has been a plateau in live birth rates associated with assisted reproductive technology (ART). Approximately 4.2% of babies born in Europe are a consequence of ART, he said.
Women were included in the single-center study over a 2-year period starting in September 2000. For inclusion they had to have a body mass index lower than 25 mg/kg2, be between 31 and 42 years old, have failed at least two cycles of IVF, and not have inherited thrombophilia.
Sixty-eight women were allocated to treatment with the LMWH dalteparin at a dose of 2,500 IU/day and 58 to treatment with aspirin at a dose of 100 mg/day and oral prednisone at a dose of 10 mg/day. Both groups of women also were given vaginal progesterone at a dose of 600 mg/day.
Treatment began at the time of embryo transfer and was continued until levels of human chorionic gonadotropin confirmed a pregnancy. If a pregnancy was confirmed, then treatment was continued until the 12th gestational week. Women administered the treatments at home.
"Heparin can potentially modulate many of the known mechanisms that underlie successful apposition, adhesion, and penetration of the developing embryo," Dr. Colicchia said.
"There is evidence that in women with repeated IVF failure, [LMWH] can improve pregnancy rates," he concluded, although further studies are needed.
The chairman of the session in which the study results were presented was critical of the findings. "This was not a randomized nor a placebo-controlled trial," Dr. Thomas D’Hooghe of Leuven (Belgium) University fertility center said in an interview. Furthermore, the control arm was flawed, as aspirin and oral prednisone are not routinely used.
Elsewhere at the annual meeting of ESHRE, a German team presented data from a 12-center trial (n = 449) on the use of LMWH in the prevention of recurrent miscarriage. The results of the ETHIG II study, in which dalteparin (5,000 IU/day) plus multivitamins were compared with vitamins alone, were not positive.
There was no difference in the primary outcome measure of the number of ongoing pregnancies at 24 complete weeks of gestation, leading the authors to conclude that "the uncontrolled use of LMWH in patients with recurrent miscarriages is not reasonable."
There was no commercial funding for the study presented by Dr. Colicchia. He reported having no relevant financial disclosures. Dr. D’Hooghe was not involved in the study and said he had no relevant conflicts of interest.
LONDON – Low-molecular-weight heparin therapy reduced the risk for recurrent implantation failure, improved pregnancy rates, and increased the number of live births in women who had failed prior fertility treatment in a 126-patient pilot study.
As reported at the annual meeting of the European Society of Human Reproduction and Embryology, low-molecular-weight heparin (LMWH) was associated with an implantation rate of 22.2% vs. 10% for a regimen containing aspirin (P =.0136). Pregnancy (47% vs. 20.6%, P = .0026) and live birth rates (30% vs. 12%, P = .017) also were higher with the LMWH than with the aspirin-containing regimen.
Although larger, randomized, double blind controlled trials are needed to confirm the findings, the study’s Italian investigators are confident that LMWH has a benefit in improving fertility outcomes in this population of women who had failed two or more cycles of in vitro fertilization (IVF).
"We are convinced that low-molecular-weight heparin can enhance results in IVF procedures," said Dr. Antonio Colicchia of the Centro Fecondazione Assistita Ferticlinic, Rome, in an interview.
Strategies to improve implantation rates are needed, Dr. Colicchia explained, because there has been a plateau in live birth rates associated with assisted reproductive technology (ART). Approximately 4.2% of babies born in Europe are a consequence of ART, he said.
Women were included in the single-center study over a 2-year period starting in September 2000. For inclusion they had to have a body mass index lower than 25 mg/kg2, be between 31 and 42 years old, have failed at least two cycles of IVF, and not have inherited thrombophilia.
Sixty-eight women were allocated to treatment with the LMWH dalteparin at a dose of 2,500 IU/day and 58 to treatment with aspirin at a dose of 100 mg/day and oral prednisone at a dose of 10 mg/day. Both groups of women also were given vaginal progesterone at a dose of 600 mg/day.
Treatment began at the time of embryo transfer and was continued until levels of human chorionic gonadotropin confirmed a pregnancy. If a pregnancy was confirmed, then treatment was continued until the 12th gestational week. Women administered the treatments at home.
"Heparin can potentially modulate many of the known mechanisms that underlie successful apposition, adhesion, and penetration of the developing embryo," Dr. Colicchia said.
"There is evidence that in women with repeated IVF failure, [LMWH] can improve pregnancy rates," he concluded, although further studies are needed.
The chairman of the session in which the study results were presented was critical of the findings. "This was not a randomized nor a placebo-controlled trial," Dr. Thomas D’Hooghe of Leuven (Belgium) University fertility center said in an interview. Furthermore, the control arm was flawed, as aspirin and oral prednisone are not routinely used.
Elsewhere at the annual meeting of ESHRE, a German team presented data from a 12-center trial (n = 449) on the use of LMWH in the prevention of recurrent miscarriage. The results of the ETHIG II study, in which dalteparin (5,000 IU/day) plus multivitamins were compared with vitamins alone, were not positive.
There was no difference in the primary outcome measure of the number of ongoing pregnancies at 24 complete weeks of gestation, leading the authors to conclude that "the uncontrolled use of LMWH in patients with recurrent miscarriages is not reasonable."
There was no commercial funding for the study presented by Dr. Colicchia. He reported having no relevant financial disclosures. Dr. D’Hooghe was not involved in the study and said he had no relevant conflicts of interest.
AT ESHRE 2013
Major finding: Implantation (22.2% vs. 10%, P =.0136), pregnancy (47% vs. 20.6%, P = .0026), and live birth rates (30% vs. 12%, P = .017) were higher with the low-molecular-weight heparin than with the aspirin-containing regimen.
Data source: A single-center pilot study of 126 women without thrombophilia who had failed at least two cycles of in vitro fertilization.
Disclosures: There was no commercial funding for the study presented by Dr. Colicchia. He reported having no relevant financial disclosures. Dr. D’Hooghe was not involved in the study and said he had no relevant conflicts of interest.
Early surgery for intracerebral bleeds may benefit a select few
LONDON – Approximately 2%-3% of patients with spontaneous intracerebral hemorrhage may benefit from early surgical removal of hematoma, according to the results of the second Surgical Trial in Lobar Intracerebral Hemorrhage.
As reported at the annual European Stroke Conference and published simultaneously in the Lancet, patients with superficial lesions and an unfavorable prognostic score appeared to benefit from early surgical intervention, compared with those given conservative medical treatment (odds ratio [OR] = 0.49, P =.02). Conversely, those with a good prognostic score did not seem to benefit (OR = 1.12, P = .57).
The primary analysis showed no significant benefit of early surgery overall, with 41.4% of 297 patients in the early surgery group and 37.7% of 286 patients in the conservative treatment group having a favorable outcome at 6 months, as determined by the 8-point extended Glasgow Outcome Scale (GOS-E) (P = .367).
"That’s a 3.7% absolute benefit, which is not enough to change surgical practice on its own," said Dr. A. David Mendelow, professor of neurosurgery at Newcastle University in Newcastle upon Tyne, England. "We were looking for a 12% benefit when we set out to do this trial," he added during a press conference.
A 6% decrease in mortality at 6 months was seen favoring surgery (18% vs. 24% for conservative therapy), but this was not statistically significant (P = .095).
"Intracerebral hemorrhage is not a homogenous condition," Dr. Mendelow said, adding that it can be a difficult decision to operate. Clinical manifestations can range from no apparent effects to severe disability and rapid death. "STICH [Surgical Trial in Lobar Intracerebral Hemorrhage] focused on patients that we are not quite sure about whether to operate or not," he said.
The hypothesis for the trial was based on the findings of the first STICH trial (Acta Neurochir. Suppl. 2006;96:65-8). The results of the trial were again neutral overall, but subgroup analyses showed that some groups of patients did worse with surgery, such as those with deep-seated bleeds, and some may fare better, such as those with superficial lobar hematomas.
STICH II therefore specifically recruited this latter group of patients to see if the effect was real or an artifact of the scientific analysis. In total, 601 conscious ICH patients with a median age of 65 years were recruited at 78 centers in 27 countries. Patients had to have a superficial lesion (1 cm or less from the cortical surface of the brain) that was between 10 mL and 100 mL in volume, and with no sign of intraventricular hemorrhage on CT scanning. Patients had to be recruited within 48 hours of the stroke and surgical intervention had to be performed within 12 hours (Lancet 2013 May 29 [doi:10.1016/S0140-6736(13)60986-1]). (A total of 6 patients were excluded or withdrew from the study before intervention, and after intervention another 12 withdrew, were lost to follow-up, or were alive but had an unknown status.)
The GOS-E was calculated from the answers to a questionnaire sent out to patients and their relatives 6 months following their stroke. A cutoff score of approximately 27 was used to categorize patients as having a good or bad prognosis. At baseline, about two-thirds of patients had a good prognosis, and the remainder had a poor prognosis.
"The notion that early surgery might be beneficial in this subgroup of patients is supported by the results of the investigator’s updated meta-analysis of 15 trials,"
"One of the reasons, perhaps, for a lack of significance was the [number of] crossovers from initial conservative therapy to surgery," Dr. Mendelow said. Indeed, 21% of patients who were originally randomized to conservative treatment crossed over to the surgical arm. These patients had "clearly deteriorated" prior to having surgery, he said when presenting the findings. Furthermore, only 37% of these crossovers received surgery within the specified 12-hour time limit.
STICH provides the best, albeit insufficient, evidence to date on the role of surgery in ICH, Dr. Oliver Gautschi and Dr. Karl Schaller, both of the University of Geneva, commented in an editorial about the trial (Lancet 2013 May 29 [doi:10.1016/S0140-6736(13)61087-9]) .
"The notion that early surgery might be beneficial in this subgroup of patients is supported by the results of the investigator’s updated meta-analysis of 15 trials," they wrote. "The overall result of this meta-analysis of patients with different types of intracerebral hemorrhage favors surgery."
The results of two other surgical studies, CLEAR III and MISTIE III, "are eagerly awaited," Dr. Gautschi and Dr. Schaller said, noting that, "decompressive hemicraniectomy might be a[nother] promising surgical procedure."
The U.K. Medical Research Council funded STICH II. Dr. Mendelow reported having no relevant financial disclosures.
LONDON – Approximately 2%-3% of patients with spontaneous intracerebral hemorrhage may benefit from early surgical removal of hematoma, according to the results of the second Surgical Trial in Lobar Intracerebral Hemorrhage.
As reported at the annual European Stroke Conference and published simultaneously in the Lancet, patients with superficial lesions and an unfavorable prognostic score appeared to benefit from early surgical intervention, compared with those given conservative medical treatment (odds ratio [OR] = 0.49, P =.02). Conversely, those with a good prognostic score did not seem to benefit (OR = 1.12, P = .57).
The primary analysis showed no significant benefit of early surgery overall, with 41.4% of 297 patients in the early surgery group and 37.7% of 286 patients in the conservative treatment group having a favorable outcome at 6 months, as determined by the 8-point extended Glasgow Outcome Scale (GOS-E) (P = .367).
"That’s a 3.7% absolute benefit, which is not enough to change surgical practice on its own," said Dr. A. David Mendelow, professor of neurosurgery at Newcastle University in Newcastle upon Tyne, England. "We were looking for a 12% benefit when we set out to do this trial," he added during a press conference.
A 6% decrease in mortality at 6 months was seen favoring surgery (18% vs. 24% for conservative therapy), but this was not statistically significant (P = .095).
"Intracerebral hemorrhage is not a homogenous condition," Dr. Mendelow said, adding that it can be a difficult decision to operate. Clinical manifestations can range from no apparent effects to severe disability and rapid death. "STICH [Surgical Trial in Lobar Intracerebral Hemorrhage] focused on patients that we are not quite sure about whether to operate or not," he said.
The hypothesis for the trial was based on the findings of the first STICH trial (Acta Neurochir. Suppl. 2006;96:65-8). The results of the trial were again neutral overall, but subgroup analyses showed that some groups of patients did worse with surgery, such as those with deep-seated bleeds, and some may fare better, such as those with superficial lobar hematomas.
STICH II therefore specifically recruited this latter group of patients to see if the effect was real or an artifact of the scientific analysis. In total, 601 conscious ICH patients with a median age of 65 years were recruited at 78 centers in 27 countries. Patients had to have a superficial lesion (1 cm or less from the cortical surface of the brain) that was between 10 mL and 100 mL in volume, and with no sign of intraventricular hemorrhage on CT scanning. Patients had to be recruited within 48 hours of the stroke and surgical intervention had to be performed within 12 hours (Lancet 2013 May 29 [doi:10.1016/S0140-6736(13)60986-1]). (A total of 6 patients were excluded or withdrew from the study before intervention, and after intervention another 12 withdrew, were lost to follow-up, or were alive but had an unknown status.)
The GOS-E was calculated from the answers to a questionnaire sent out to patients and their relatives 6 months following their stroke. A cutoff score of approximately 27 was used to categorize patients as having a good or bad prognosis. At baseline, about two-thirds of patients had a good prognosis, and the remainder had a poor prognosis.
"The notion that early surgery might be beneficial in this subgroup of patients is supported by the results of the investigator’s updated meta-analysis of 15 trials,"
"One of the reasons, perhaps, for a lack of significance was the [number of] crossovers from initial conservative therapy to surgery," Dr. Mendelow said. Indeed, 21% of patients who were originally randomized to conservative treatment crossed over to the surgical arm. These patients had "clearly deteriorated" prior to having surgery, he said when presenting the findings. Furthermore, only 37% of these crossovers received surgery within the specified 12-hour time limit.
STICH provides the best, albeit insufficient, evidence to date on the role of surgery in ICH, Dr. Oliver Gautschi and Dr. Karl Schaller, both of the University of Geneva, commented in an editorial about the trial (Lancet 2013 May 29 [doi:10.1016/S0140-6736(13)61087-9]) .
"The notion that early surgery might be beneficial in this subgroup of patients is supported by the results of the investigator’s updated meta-analysis of 15 trials," they wrote. "The overall result of this meta-analysis of patients with different types of intracerebral hemorrhage favors surgery."
The results of two other surgical studies, CLEAR III and MISTIE III, "are eagerly awaited," Dr. Gautschi and Dr. Schaller said, noting that, "decompressive hemicraniectomy might be a[nother] promising surgical procedure."
The U.K. Medical Research Council funded STICH II. Dr. Mendelow reported having no relevant financial disclosures.
LONDON – Approximately 2%-3% of patients with spontaneous intracerebral hemorrhage may benefit from early surgical removal of hematoma, according to the results of the second Surgical Trial in Lobar Intracerebral Hemorrhage.
As reported at the annual European Stroke Conference and published simultaneously in the Lancet, patients with superficial lesions and an unfavorable prognostic score appeared to benefit from early surgical intervention, compared with those given conservative medical treatment (odds ratio [OR] = 0.49, P =.02). Conversely, those with a good prognostic score did not seem to benefit (OR = 1.12, P = .57).
The primary analysis showed no significant benefit of early surgery overall, with 41.4% of 297 patients in the early surgery group and 37.7% of 286 patients in the conservative treatment group having a favorable outcome at 6 months, as determined by the 8-point extended Glasgow Outcome Scale (GOS-E) (P = .367).
"That’s a 3.7% absolute benefit, which is not enough to change surgical practice on its own," said Dr. A. David Mendelow, professor of neurosurgery at Newcastle University in Newcastle upon Tyne, England. "We were looking for a 12% benefit when we set out to do this trial," he added during a press conference.
A 6% decrease in mortality at 6 months was seen favoring surgery (18% vs. 24% for conservative therapy), but this was not statistically significant (P = .095).
"Intracerebral hemorrhage is not a homogenous condition," Dr. Mendelow said, adding that it can be a difficult decision to operate. Clinical manifestations can range from no apparent effects to severe disability and rapid death. "STICH [Surgical Trial in Lobar Intracerebral Hemorrhage] focused on patients that we are not quite sure about whether to operate or not," he said.
The hypothesis for the trial was based on the findings of the first STICH trial (Acta Neurochir. Suppl. 2006;96:65-8). The results of the trial were again neutral overall, but subgroup analyses showed that some groups of patients did worse with surgery, such as those with deep-seated bleeds, and some may fare better, such as those with superficial lobar hematomas.
STICH II therefore specifically recruited this latter group of patients to see if the effect was real or an artifact of the scientific analysis. In total, 601 conscious ICH patients with a median age of 65 years were recruited at 78 centers in 27 countries. Patients had to have a superficial lesion (1 cm or less from the cortical surface of the brain) that was between 10 mL and 100 mL in volume, and with no sign of intraventricular hemorrhage on CT scanning. Patients had to be recruited within 48 hours of the stroke and surgical intervention had to be performed within 12 hours (Lancet 2013 May 29 [doi:10.1016/S0140-6736(13)60986-1]). (A total of 6 patients were excluded or withdrew from the study before intervention, and after intervention another 12 withdrew, were lost to follow-up, or were alive but had an unknown status.)
The GOS-E was calculated from the answers to a questionnaire sent out to patients and their relatives 6 months following their stroke. A cutoff score of approximately 27 was used to categorize patients as having a good or bad prognosis. At baseline, about two-thirds of patients had a good prognosis, and the remainder had a poor prognosis.
"The notion that early surgery might be beneficial in this subgroup of patients is supported by the results of the investigator’s updated meta-analysis of 15 trials,"
"One of the reasons, perhaps, for a lack of significance was the [number of] crossovers from initial conservative therapy to surgery," Dr. Mendelow said. Indeed, 21% of patients who were originally randomized to conservative treatment crossed over to the surgical arm. These patients had "clearly deteriorated" prior to having surgery, he said when presenting the findings. Furthermore, only 37% of these crossovers received surgery within the specified 12-hour time limit.
STICH provides the best, albeit insufficient, evidence to date on the role of surgery in ICH, Dr. Oliver Gautschi and Dr. Karl Schaller, both of the University of Geneva, commented in an editorial about the trial (Lancet 2013 May 29 [doi:10.1016/S0140-6736(13)61087-9]) .
"The notion that early surgery might be beneficial in this subgroup of patients is supported by the results of the investigator’s updated meta-analysis of 15 trials," they wrote. "The overall result of this meta-analysis of patients with different types of intracerebral hemorrhage favors surgery."
The results of two other surgical studies, CLEAR III and MISTIE III, "are eagerly awaited," Dr. Gautschi and Dr. Schaller said, noting that, "decompressive hemicraniectomy might be a[nother] promising surgical procedure."
The U.K. Medical Research Council funded STICH II. Dr. Mendelow reported having no relevant financial disclosures.
AT THE EUROPEAN STROKE CONFERENCE
Major finding: A favorable outcome at 6 months was seen in 41% of the early surgery group and 38% of the conservative treatment group (P = .367).
Data source: STICH II, an international, multicenter prospective trial of 601 patients randomized to early surgery (within 12 hours) or medical treatment within 48 hours of a spontaneous superficial intracerebral hemorrhage.
Disclosures: The U.K. Medical Research Council funded STICH II. Dr. Mendelow reported having no relevant financial disclosures.