User login
Fissured tongue
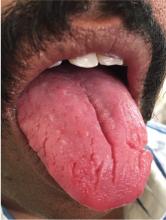
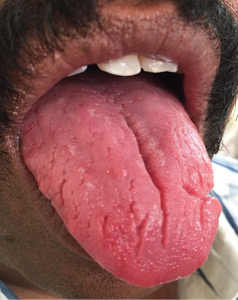
A 43-year-old man presented with a 3-week history of halitosis. He was also concerned about the irregular appearance of his tongue, which he had noticed over the past 3 years. He had no history of wearing dentures or of any skin disorder.
A BROAD DIFFERENTIAL DIAGNOSIS
Fissured tongue (scrotal tongue, plicated tongue, lingua plicata) is a common normal variant of the tongue surface with a male preponderance and a reported prevalence of 10% to 20% in the general population, and the incidence increases strikingly with age.1
The cause is not known, but familial clustering is seen, and a polygenic or autosomal dominant hereditary component is presumed.1
The condition may be associated with removable dentures, geographic tongue, pernicious anemia, Sjögren syndrome, psoriasis, acromegaly, macroglossia, oral-facial-digital syndrome type I, Pierre Robin syndrome, Down syndrome, and Melkersson Rosenthal syndrome.2 It is usually asymptomatic, but if the fissures are deep, food may become lodged in them, resulting in tongue inflammation, burning sensation, and halitosis.1
Typically, fissures of varying depth extending to the margin are apparent on the dorsal surface of the tongue. The condition is confined to the anterior two-thirds of the tongue, which is of ectodermal origin. Histologically, the epithelium, lamina propria, and musculature are all involved in the formation of the fissures.3 The deeper fissures may lack filliform papillae due to bacterial inflammation.3 The diagnosis is clinical, and treatment includes reassurance, advice on good oral hygiene, and tongue cleansing.1
- Feil ND, Filippi A. Frequency of fissured tongue (lingua plicata) as a function of age. Swiss Dent J 2016; 126(10):886–897. German. pmid:27808348
- Mangold AR, Torgerson RR, Rogers RS 3rd. Diseases of the tongue. Clin Dermatol 2016; 34(4):458–469. doi:10.1016/j.clindermatol.2016.02.018
- Kullaa-Mikkonen A, Sorvari T. Lingua fissurata: a clinical, stereomicroscopic and histopathological study. Int J Oral Maxillofac Surg 1986; 15(5):525–533. pmid:3097176
A 43-year-old man presented with a 3-week history of halitosis. He was also concerned about the irregular appearance of his tongue, which he had noticed over the past 3 years. He had no history of wearing dentures or of any skin disorder.
A BROAD DIFFERENTIAL DIAGNOSIS
Fissured tongue (scrotal tongue, plicated tongue, lingua plicata) is a common normal variant of the tongue surface with a male preponderance and a reported prevalence of 10% to 20% in the general population, and the incidence increases strikingly with age.1
The cause is not known, but familial clustering is seen, and a polygenic or autosomal dominant hereditary component is presumed.1
The condition may be associated with removable dentures, geographic tongue, pernicious anemia, Sjögren syndrome, psoriasis, acromegaly, macroglossia, oral-facial-digital syndrome type I, Pierre Robin syndrome, Down syndrome, and Melkersson Rosenthal syndrome.2 It is usually asymptomatic, but if the fissures are deep, food may become lodged in them, resulting in tongue inflammation, burning sensation, and halitosis.1
Typically, fissures of varying depth extending to the margin are apparent on the dorsal surface of the tongue. The condition is confined to the anterior two-thirds of the tongue, which is of ectodermal origin. Histologically, the epithelium, lamina propria, and musculature are all involved in the formation of the fissures.3 The deeper fissures may lack filliform papillae due to bacterial inflammation.3 The diagnosis is clinical, and treatment includes reassurance, advice on good oral hygiene, and tongue cleansing.1
A 43-year-old man presented with a 3-week history of halitosis. He was also concerned about the irregular appearance of his tongue, which he had noticed over the past 3 years. He had no history of wearing dentures or of any skin disorder.
A BROAD DIFFERENTIAL DIAGNOSIS
Fissured tongue (scrotal tongue, plicated tongue, lingua plicata) is a common normal variant of the tongue surface with a male preponderance and a reported prevalence of 10% to 20% in the general population, and the incidence increases strikingly with age.1
The cause is not known, but familial clustering is seen, and a polygenic or autosomal dominant hereditary component is presumed.1
The condition may be associated with removable dentures, geographic tongue, pernicious anemia, Sjögren syndrome, psoriasis, acromegaly, macroglossia, oral-facial-digital syndrome type I, Pierre Robin syndrome, Down syndrome, and Melkersson Rosenthal syndrome.2 It is usually asymptomatic, but if the fissures are deep, food may become lodged in them, resulting in tongue inflammation, burning sensation, and halitosis.1
Typically, fissures of varying depth extending to the margin are apparent on the dorsal surface of the tongue. The condition is confined to the anterior two-thirds of the tongue, which is of ectodermal origin. Histologically, the epithelium, lamina propria, and musculature are all involved in the formation of the fissures.3 The deeper fissures may lack filliform papillae due to bacterial inflammation.3 The diagnosis is clinical, and treatment includes reassurance, advice on good oral hygiene, and tongue cleansing.1
- Feil ND, Filippi A. Frequency of fissured tongue (lingua plicata) as a function of age. Swiss Dent J 2016; 126(10):886–897. German. pmid:27808348
- Mangold AR, Torgerson RR, Rogers RS 3rd. Diseases of the tongue. Clin Dermatol 2016; 34(4):458–469. doi:10.1016/j.clindermatol.2016.02.018
- Kullaa-Mikkonen A, Sorvari T. Lingua fissurata: a clinical, stereomicroscopic and histopathological study. Int J Oral Maxillofac Surg 1986; 15(5):525–533. pmid:3097176
- Feil ND, Filippi A. Frequency of fissured tongue (lingua plicata) as a function of age. Swiss Dent J 2016; 126(10):886–897. German. pmid:27808348
- Mangold AR, Torgerson RR, Rogers RS 3rd. Diseases of the tongue. Clin Dermatol 2016; 34(4):458–469. doi:10.1016/j.clindermatol.2016.02.018
- Kullaa-Mikkonen A, Sorvari T. Lingua fissurata: a clinical, stereomicroscopic and histopathological study. Int J Oral Maxillofac Surg 1986; 15(5):525–533. pmid:3097176
A burning sensation in the mouth
A 38-year-old man presented with a burning sensation in the mouth for the previous 2 months. He was currently a smoker. He had no history of any dental procedure or new drug intake.
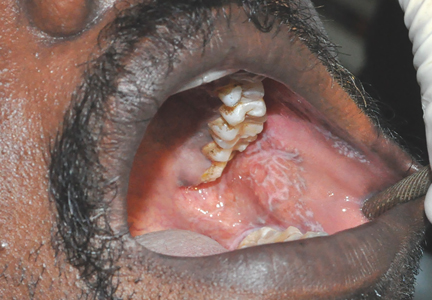
On examination, his oral hygiene was poor, and a whitish, striated lesion 3-by-3 cm was noted on the left buccal mucosa (Figure 1). Based on the appearance and pattern of the lesion, a clinical diagnosis of reticular-type oral lichen planus was made. The patient was advised to stop smoking. A thorough scaling of the teeth was done, and he was started on topical triamcinolone acetonide ointment for 2 weeks. At a 4-month follow-up visit, the symptoms had resolved.
ORAL LICHEN PLANUS
Oral lichen planus is a chronic inflammatory disease that affects the mucous membrane of the oral cavity. It is a T-cell mediated autoimmune disease in which the cytotoxic CD8+ T cells trigger apoptosis of the basal cells of the oral epithelium.1 No risk factors have been identified, but the condition has been associated with smoking, chewing tobacco, dental materials such as amalgam, celiac disease, ulcerative colitis, stress, diabetes, and use of systemic drugs such as nonsteroidal anti-inflammatory drugs and beta-blockers.1,2 Women are affected more than men in a ratio of 1.4:1, frequently in the fourth decade.
The clinical forms are reticular, papular, plaque-like, erosive, atrophic, and bullous types.
Clinical presentation
The lesions are usually asymptomatic. Symptoms such as a burning sensation are associated mainly with the erosive and atrophic types,1–3 although in rare cases it occurs with the reticular form, as in our patient. Erosive and atrophic types carry a 1% to 2% risk of malignant transformation and require close follow-up.3
Oral mucosal lesions present alone or, in up to 15% to 20% of cases, with concomitant skin lesions.1,2 The most common oral sites are the buccal mucosa, tongue, and gingiva. Skin lesions present as violaceous flat-topped papules on the ankles, wrists, and genitalia.
DIAGNOSIS AND TREATMENT
The differential diagnosis of oral lichen planus includes cheek-frictional keratosis, lichenoid reaction, leukoplakia, pemphigus, oral candidiasis, and chronic ulcerative stomatitis.1,2
The diagnosis is clinical, but biopsy is indicated in the erosive or atrophic type and when a malignant lesion is suspected, especially in patients with a history of smoking and tobacco chewing. Oral lichen planus may be associated with oral candidiasis, especially in patients with poor oral hygiene and smoking, and this needs to be addressed before a diagnosis of oral lichen planus can be made.
The mainstay of management is oral or topical corticosteroids.4 Calcineurin inhibitors, retinoids, dapsone, hydroxychloroquine, mycophenolate mofetil, and enoxaparin have also been shown to be effective and are used when corticosteroids alone are ineffective or contraindicated.4
- Alrashdan MS, Cirillo N, McCullough M. Oral lichen planus: a literature review and update. Arch Dermatol Res 2016; 308:539–551.
- Nogueira PA, Carneiro S, Ramos-e-Silva M. Oral lichen planus: an update on its pathogenesis. Int J Dermatol 2015; 54:1005–1010.
- Fitzpatrick SG, Hirsch SA, Gordon SC. The malignant transformation of oral lichen planus and oral lichenoid lesions: a systematic review. J Am Dent Assoc 2014; 145:45–56.
- Lodi G, Carrozzo M, Furness S, Thongprasom K. Interventions for treating oral lichen planus: a systematic review. Br J Dermatol 2012; 166:938–947.
A 38-year-old man presented with a burning sensation in the mouth for the previous 2 months. He was currently a smoker. He had no history of any dental procedure or new drug intake.
On examination, his oral hygiene was poor, and a whitish, striated lesion 3-by-3 cm was noted on the left buccal mucosa (Figure 1). Based on the appearance and pattern of the lesion, a clinical diagnosis of reticular-type oral lichen planus was made. The patient was advised to stop smoking. A thorough scaling of the teeth was done, and he was started on topical triamcinolone acetonide ointment for 2 weeks. At a 4-month follow-up visit, the symptoms had resolved.
ORAL LICHEN PLANUS
Oral lichen planus is a chronic inflammatory disease that affects the mucous membrane of the oral cavity. It is a T-cell mediated autoimmune disease in which the cytotoxic CD8+ T cells trigger apoptosis of the basal cells of the oral epithelium.1 No risk factors have been identified, but the condition has been associated with smoking, chewing tobacco, dental materials such as amalgam, celiac disease, ulcerative colitis, stress, diabetes, and use of systemic drugs such as nonsteroidal anti-inflammatory drugs and beta-blockers.1,2 Women are affected more than men in a ratio of 1.4:1, frequently in the fourth decade.
The clinical forms are reticular, papular, plaque-like, erosive, atrophic, and bullous types.
Clinical presentation
The lesions are usually asymptomatic. Symptoms such as a burning sensation are associated mainly with the erosive and atrophic types,1–3 although in rare cases it occurs with the reticular form, as in our patient. Erosive and atrophic types carry a 1% to 2% risk of malignant transformation and require close follow-up.3
Oral mucosal lesions present alone or, in up to 15% to 20% of cases, with concomitant skin lesions.1,2 The most common oral sites are the buccal mucosa, tongue, and gingiva. Skin lesions present as violaceous flat-topped papules on the ankles, wrists, and genitalia.
DIAGNOSIS AND TREATMENT
The differential diagnosis of oral lichen planus includes cheek-frictional keratosis, lichenoid reaction, leukoplakia, pemphigus, oral candidiasis, and chronic ulcerative stomatitis.1,2
The diagnosis is clinical, but biopsy is indicated in the erosive or atrophic type and when a malignant lesion is suspected, especially in patients with a history of smoking and tobacco chewing. Oral lichen planus may be associated with oral candidiasis, especially in patients with poor oral hygiene and smoking, and this needs to be addressed before a diagnosis of oral lichen planus can be made.
The mainstay of management is oral or topical corticosteroids.4 Calcineurin inhibitors, retinoids, dapsone, hydroxychloroquine, mycophenolate mofetil, and enoxaparin have also been shown to be effective and are used when corticosteroids alone are ineffective or contraindicated.4
A 38-year-old man presented with a burning sensation in the mouth for the previous 2 months. He was currently a smoker. He had no history of any dental procedure or new drug intake.
On examination, his oral hygiene was poor, and a whitish, striated lesion 3-by-3 cm was noted on the left buccal mucosa (Figure 1). Based on the appearance and pattern of the lesion, a clinical diagnosis of reticular-type oral lichen planus was made. The patient was advised to stop smoking. A thorough scaling of the teeth was done, and he was started on topical triamcinolone acetonide ointment for 2 weeks. At a 4-month follow-up visit, the symptoms had resolved.
ORAL LICHEN PLANUS
Oral lichen planus is a chronic inflammatory disease that affects the mucous membrane of the oral cavity. It is a T-cell mediated autoimmune disease in which the cytotoxic CD8+ T cells trigger apoptosis of the basal cells of the oral epithelium.1 No risk factors have been identified, but the condition has been associated with smoking, chewing tobacco, dental materials such as amalgam, celiac disease, ulcerative colitis, stress, diabetes, and use of systemic drugs such as nonsteroidal anti-inflammatory drugs and beta-blockers.1,2 Women are affected more than men in a ratio of 1.4:1, frequently in the fourth decade.
The clinical forms are reticular, papular, plaque-like, erosive, atrophic, and bullous types.
Clinical presentation
The lesions are usually asymptomatic. Symptoms such as a burning sensation are associated mainly with the erosive and atrophic types,1–3 although in rare cases it occurs with the reticular form, as in our patient. Erosive and atrophic types carry a 1% to 2% risk of malignant transformation and require close follow-up.3
Oral mucosal lesions present alone or, in up to 15% to 20% of cases, with concomitant skin lesions.1,2 The most common oral sites are the buccal mucosa, tongue, and gingiva. Skin lesions present as violaceous flat-topped papules on the ankles, wrists, and genitalia.
DIAGNOSIS AND TREATMENT
The differential diagnosis of oral lichen planus includes cheek-frictional keratosis, lichenoid reaction, leukoplakia, pemphigus, oral candidiasis, and chronic ulcerative stomatitis.1,2
The diagnosis is clinical, but biopsy is indicated in the erosive or atrophic type and when a malignant lesion is suspected, especially in patients with a history of smoking and tobacco chewing. Oral lichen planus may be associated with oral candidiasis, especially in patients with poor oral hygiene and smoking, and this needs to be addressed before a diagnosis of oral lichen planus can be made.
The mainstay of management is oral or topical corticosteroids.4 Calcineurin inhibitors, retinoids, dapsone, hydroxychloroquine, mycophenolate mofetil, and enoxaparin have also been shown to be effective and are used when corticosteroids alone are ineffective or contraindicated.4
- Alrashdan MS, Cirillo N, McCullough M. Oral lichen planus: a literature review and update. Arch Dermatol Res 2016; 308:539–551.
- Nogueira PA, Carneiro S, Ramos-e-Silva M. Oral lichen planus: an update on its pathogenesis. Int J Dermatol 2015; 54:1005–1010.
- Fitzpatrick SG, Hirsch SA, Gordon SC. The malignant transformation of oral lichen planus and oral lichenoid lesions: a systematic review. J Am Dent Assoc 2014; 145:45–56.
- Lodi G, Carrozzo M, Furness S, Thongprasom K. Interventions for treating oral lichen planus: a systematic review. Br J Dermatol 2012; 166:938–947.
- Alrashdan MS, Cirillo N, McCullough M. Oral lichen planus: a literature review and update. Arch Dermatol Res 2016; 308:539–551.
- Nogueira PA, Carneiro S, Ramos-e-Silva M. Oral lichen planus: an update on its pathogenesis. Int J Dermatol 2015; 54:1005–1010.
- Fitzpatrick SG, Hirsch SA, Gordon SC. The malignant transformation of oral lichen planus and oral lichenoid lesions: a systematic review. J Am Dent Assoc 2014; 145:45–56.
- Lodi G, Carrozzo M, Furness S, Thongprasom K. Interventions for treating oral lichen planus: a systematic review. Br J Dermatol 2012; 166:938–947.
Plexiform neurofibroma
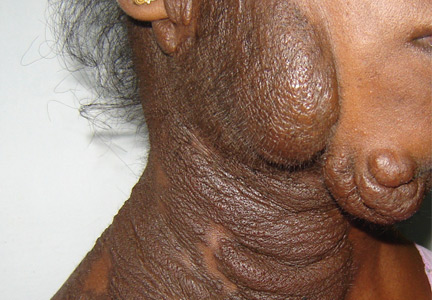
A 19-year-old woman presented with diffuse swelling on her neck and face that had been growing gradually over the past 12 years. There was no family history of similar swelling.
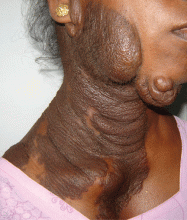
Examination revealed a diffuse, hyperpigmented, nodular swelling on the right side of her neck and extending upward to the lower part of the face and the scalp (Figure 1). The rest of the physical examination was normal. A diagnosis of plexiform neurofibroma was made based on the typical clinical presentation. The patient had come primarily because of cosmetic concerns but refused surgery. She was offered genetic counseling and was scheduled for regular follow-up.
Plexiform neurofibroma is mostly associated with autosomal dominant neurofibromatosis type 1, characterized by cutaneous findings such as café-au-lait spots, axillary freckling, skeletal dysplasias (usually of the tibia, fibula, ribs, and vertebrae), Lisch nodules in the eye (iris hamartomas), and neural tumors. Rarely, it may also be seen in germline p16 mutation-positive heritable melanoma and Cowden syndrome.
Diffuse plexiform neurofibroma of the face and neck rarely appears after the age of 1, and rarely develops on other parts of the body after adolescence. In contrast, deep nodular plexiform neurofibroma often originates from spinal nerve roots and usually becomes symptomatic in adulthood.
Plexiform neurofibroma has an 8% to 12% chance of changing into a malignant peripheral nerve-sheath tumor. Continuous pain in the tumor, rapid tumor growth, hardening of the tumor, or weakness or numbness in an arm or leg with a plexiform neurofibroma suggests malignant transformation.
The diagnosis is clinical, and the management involves a multidisciplinary team including a physician, geneticist, neurologist, surgeon, and ophthalmologist. Genetic counseling should be offered to patients for assessment of family risk, to look for other possible conditions such as Cowden syndrome, for marital and family planning, and whenever the diagnosis is unclear.
A 19-year-old woman presented with diffuse swelling on her neck and face that had been growing gradually over the past 12 years. There was no family history of similar swelling.
Examination revealed a diffuse, hyperpigmented, nodular swelling on the right side of her neck and extending upward to the lower part of the face and the scalp (Figure 1). The rest of the physical examination was normal. A diagnosis of plexiform neurofibroma was made based on the typical clinical presentation. The patient had come primarily because of cosmetic concerns but refused surgery. She was offered genetic counseling and was scheduled for regular follow-up.
Plexiform neurofibroma is mostly associated with autosomal dominant neurofibromatosis type 1, characterized by cutaneous findings such as café-au-lait spots, axillary freckling, skeletal dysplasias (usually of the tibia, fibula, ribs, and vertebrae), Lisch nodules in the eye (iris hamartomas), and neural tumors. Rarely, it may also be seen in germline p16 mutation-positive heritable melanoma and Cowden syndrome.
Diffuse plexiform neurofibroma of the face and neck rarely appears after the age of 1, and rarely develops on other parts of the body after adolescence. In contrast, deep nodular plexiform neurofibroma often originates from spinal nerve roots and usually becomes symptomatic in adulthood.
Plexiform neurofibroma has an 8% to 12% chance of changing into a malignant peripheral nerve-sheath tumor. Continuous pain in the tumor, rapid tumor growth, hardening of the tumor, or weakness or numbness in an arm or leg with a plexiform neurofibroma suggests malignant transformation.
The diagnosis is clinical, and the management involves a multidisciplinary team including a physician, geneticist, neurologist, surgeon, and ophthalmologist. Genetic counseling should be offered to patients for assessment of family risk, to look for other possible conditions such as Cowden syndrome, for marital and family planning, and whenever the diagnosis is unclear.
A 19-year-old woman presented with diffuse swelling on her neck and face that had been growing gradually over the past 12 years. There was no family history of similar swelling.
Examination revealed a diffuse, hyperpigmented, nodular swelling on the right side of her neck and extending upward to the lower part of the face and the scalp (Figure 1). The rest of the physical examination was normal. A diagnosis of plexiform neurofibroma was made based on the typical clinical presentation. The patient had come primarily because of cosmetic concerns but refused surgery. She was offered genetic counseling and was scheduled for regular follow-up.
Plexiform neurofibroma is mostly associated with autosomal dominant neurofibromatosis type 1, characterized by cutaneous findings such as café-au-lait spots, axillary freckling, skeletal dysplasias (usually of the tibia, fibula, ribs, and vertebrae), Lisch nodules in the eye (iris hamartomas), and neural tumors. Rarely, it may also be seen in germline p16 mutation-positive heritable melanoma and Cowden syndrome.
Diffuse plexiform neurofibroma of the face and neck rarely appears after the age of 1, and rarely develops on other parts of the body after adolescence. In contrast, deep nodular plexiform neurofibroma often originates from spinal nerve roots and usually becomes symptomatic in adulthood.
Plexiform neurofibroma has an 8% to 12% chance of changing into a malignant peripheral nerve-sheath tumor. Continuous pain in the tumor, rapid tumor growth, hardening of the tumor, or weakness or numbness in an arm or leg with a plexiform neurofibroma suggests malignant transformation.
The diagnosis is clinical, and the management involves a multidisciplinary team including a physician, geneticist, neurologist, surgeon, and ophthalmologist. Genetic counseling should be offered to patients for assessment of family risk, to look for other possible conditions such as Cowden syndrome, for marital and family planning, and whenever the diagnosis is unclear.