User login
Patient With Leukocytosis and Persistent Dry Cough
Discussion
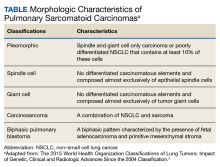
An interdisciplinary discussion regarding the diagnosis considered the clinical features of the patient along with the imaging characteristics. The histological examination demonstrating sarcomatoid features with the supporting immunohistochemistry that confirmed both mesenchymal and epithelioid presence and was used to make the diagnosis of pulmonary sarcomatoid carcinoma (PSC).
The incidence of PSC ranges between 0.1% and 0.4% of all lung malignancies.1,4-7 PSC usually occurs in older men whose weight is moderate to heavy and who smoke. PSC appears to have an upper lobe predilection; also, these tumors tend to be bulky with invasive tendency, early recurrence, and systemic metastases. PSC frequently involves the adjacent lung, chest wall, diaphragm, pericardium, and other tissues.1-5 The source of the sarcoma component of the PSC remains uncertain. However, prior research suggests that it is associated with a clonal evolution that induces epidermal and mesenchymal tumor histological characteristics.1,8,9 The tumor cell epithelial-mesenchymal transition may induce transformation of the carcinoma component of PSC to into a sarcoma component. The epithelial-mesenchymal transition is associated with the PSC high risk for invasiveness and induces metastasis sites, such as the esophagus, colon, rectum, kidneys, and the common sites of NSCLC.
The most common symptoms include productive cough, chest congestion, and chest pain.1,7 In view of PSC’s clinical presentation and imaging, numerous differential diagnoses should be considered, such as sarcomatoid carcinomas, primary or secondary metastatic sarcomas, malignant melanoma, and pleural mesothelioma.6,10
The tumor is initially identified by a chest CT, confirmed by histology and immunohistochemistry. Several biomarkers are useful for diagnosis and classification of an undifferentiated neoplasm/tumor of uncertain origin. Those biomarkers help to understand the tumor pathobiology, to select the therapeutic regimen, and to predict the patient’s outcome. Although immunohistochemical staining of epithelial and mesenchymal markers can be helpful, a reliable diagnosis requires a precise histopathological examination. This is often difficult on small biopsy samples, such as fine-needle aspiration, as all the histological elements of PSC required to make the correct evaluation may not be present. Adequate sampling to generate a considerable number of histological slides is essential for an accurate diagnosis, which can be reached only with surgical resection.5
Due to rarity, rapid progression, short survival, and heterogeneous pathological qualities, PSC has been difficult to formulate treatment recommendations. Compared with other histological subtypes of NSCLC, PSC is more aggressive and has a poor prognosis. Survival time on average is about 13.3 months due to early metastasis, lower than other types of NSCLC. The greatest overall survival (OS) benefit has been shown with surgery in early-stage operable PSC, which remains the standard of care. Because most patients with PSC present in the advanced stage, they lose their opportunity for curative surgery. Auxiliary methods of treatment include radiotherapy and chemotherapy. Prior studies have shown that systemic chemotherapy efficacy has varied, some showing no OS benefit; others showing a modest benefit. It has also been noted that advanced-stage PSC has minimal response to chemotherapy. Further larger prospective studies are needed to outline the efficacy and role of systemic chemotherapy and other therapeutic agents, including targeted therapies and immunotherapy.1,4,11-13 However, two-thirds of patients are not sensitive to conventional chemotherapy. In comparison with other types of NSCLC, PSC carries a poor prognosis even in early-stage disease or if tumor metastasis is present. Therefore, further research on novel treatment options is needed to improve long-term survival.1,3-8
Conclusions
PSC is diagnostically challenging because it is rare and has an aggressive progression. Identification of this tumor requires knowledge of histological criteria to identify their subtypes. Immunohistochemistry has an important role in the classification and to rule out differential diagnoses, including metastatic spread. Nevertheless, a reliable diagnosis requires precise histopathological examination, reached with surgical resection. Therefore, a detailed history, physical examination, systematic investigation, and correlation with chest imaging are needed to avoid misdiagnosis.
Our case highlights the importance of keeping this rare, aggressive tumor as part of the differential diagnosis. In view of its natural history, heterogeneity, and low incidence, published cases of PSC are limited. Thus, further investigation could optimize rapid identification and treatment options.
1. Qin Z, Huang B, Yu G, Zheng Y, Zhao K. Gingival metastasis of a mediastinal pulmonary sarcomatoid carcinoma: a case report. J Cardiothorac Surg. 2019;14(1):161. Published 2019 Sep 9. doi:10.1186/s13019-019-0991-y
2. Travis WD, Brambilla E, Nicholson AG, et al; WHO Panel. The 2015 World Health Organization Classification of Lung Tumors: Impact of Genetic, Clinical and Radiologic Advances Since the 2004 Classification. J Thorac Oncol. 2015;10(9):1243-1260. doi:10.1097/JTO.0000000000000630
3. Yendamuri S, Caty L, Pine M, et al. Outcomes of sarcomatoid carcinoma of the lung: a Surveillance, Epidemiology, and End Results Database analysis. Surgery. 2012;152(3):397-402. doi:10.1016/j.surg.2012.05.007
4. Karim NA, Schuster J, Eldessouki I, et al. Pulmonary sarcomatoid carcinoma: University of Cincinnati experience. Oncotarget. 2017;9(3):4102-4108. Published 2017 Dec 18. doi:10.18632/oncotarget.23468
5. Weissferdt A. Pulmonary sarcomatoid carcinomas: a review. Adv Anat Pathol. 2018;25(5):304-313. doi:10.1097/PAP.0000000000000202
6. Roesel C, Terjung S, Weinreich G, et al. Sarcomatoid carcinoma of the lung: a rare histological subtype of non-small cell lung cancer with a poor prognosis even at earlier tumour stages. Interact Cardiovasc Thorac Surg. 2017;24(3):407-413. doi:10.1093/icvts/ivw392
7. Franks TJ, Galvin JR. Sarcomatoid carcinoma of the lung: histologic criteria and common lesions in the differential diagnosis. Arch Pathol Lab Med. 2010;134(1):49-54. doi:10.5858/2008-0547-RAR.1
8. Thomas VT, Hinson S, Konduri K. Epithelial-mesenchymal transition in pulmonary carcinosarcoma: case report and literature review. Ther Adv Med Oncol. 2012;4(1):31-37. doi:10.1177/1758834011421949
9. Chang YL, Wu CT, Shih JY, Lee YC. EGFR and p53 status of pulmonary pleomorphic carcinoma: implications for EGFR tyrosine kinase inhibitors therapy of an aggressive lung malignancy. Ann Surg Oncol. 2011;18(10):2952-2960. doi:10.1245/s10434-011-1621-7
10. Travis WD, Brambilla E, Burke AP, Marx A, Nicholson AG, eds. WHO Classification of Tumours of the Lung, Pleura, Thymus and Heart. 4th ed. International Agency for Research on Cancer; 2015:88-94.
11. Huang SY, Shen SJ, Li XY. Pulmonary sarcomatoid carcinoma: a clinicopathologic study and prognostic analysis of 51 cases. World J Surg Oncol. 2013;11:252. Published 2013 Oct 2. doi:10.1186/1477-7819-11-252
12. Pelosi G, Sonzogni A, De Pas T, et al. Review article: pulmonary sarcomatoid carcinomas: a practical overview. Int J Surg Pathol. 2010;18(2):103-120. doi:10.1177/1066896908330049
13. Lin F, Liu H. Immunohistochemistry in undifferentiated neoplasm/tumor of uncertain origin. Arch Pathol Lab Med. 2014;138(12):1583-1610. doi:10.5858/arpa.2014-0061-RA
Discussion
An interdisciplinary discussion regarding the diagnosis considered the clinical features of the patient along with the imaging characteristics. The histological examination demonstrating sarcomatoid features with the supporting immunohistochemistry that confirmed both mesenchymal and epithelioid presence and was used to make the diagnosis of pulmonary sarcomatoid carcinoma (PSC).
The incidence of PSC ranges between 0.1% and 0.4% of all lung malignancies.1,4-7 PSC usually occurs in older men whose weight is moderate to heavy and who smoke. PSC appears to have an upper lobe predilection; also, these tumors tend to be bulky with invasive tendency, early recurrence, and systemic metastases. PSC frequently involves the adjacent lung, chest wall, diaphragm, pericardium, and other tissues.1-5 The source of the sarcoma component of the PSC remains uncertain. However, prior research suggests that it is associated with a clonal evolution that induces epidermal and mesenchymal tumor histological characteristics.1,8,9 The tumor cell epithelial-mesenchymal transition may induce transformation of the carcinoma component of PSC to into a sarcoma component. The epithelial-mesenchymal transition is associated with the PSC high risk for invasiveness and induces metastasis sites, such as the esophagus, colon, rectum, kidneys, and the common sites of NSCLC.
The most common symptoms include productive cough, chest congestion, and chest pain.1,7 In view of PSC’s clinical presentation and imaging, numerous differential diagnoses should be considered, such as sarcomatoid carcinomas, primary or secondary metastatic sarcomas, malignant melanoma, and pleural mesothelioma.6,10
The tumor is initially identified by a chest CT, confirmed by histology and immunohistochemistry. Several biomarkers are useful for diagnosis and classification of an undifferentiated neoplasm/tumor of uncertain origin. Those biomarkers help to understand the tumor pathobiology, to select the therapeutic regimen, and to predict the patient’s outcome. Although immunohistochemical staining of epithelial and mesenchymal markers can be helpful, a reliable diagnosis requires a precise histopathological examination. This is often difficult on small biopsy samples, such as fine-needle aspiration, as all the histological elements of PSC required to make the correct evaluation may not be present. Adequate sampling to generate a considerable number of histological slides is essential for an accurate diagnosis, which can be reached only with surgical resection.5
Due to rarity, rapid progression, short survival, and heterogeneous pathological qualities, PSC has been difficult to formulate treatment recommendations. Compared with other histological subtypes of NSCLC, PSC is more aggressive and has a poor prognosis. Survival time on average is about 13.3 months due to early metastasis, lower than other types of NSCLC. The greatest overall survival (OS) benefit has been shown with surgery in early-stage operable PSC, which remains the standard of care. Because most patients with PSC present in the advanced stage, they lose their opportunity for curative surgery. Auxiliary methods of treatment include radiotherapy and chemotherapy. Prior studies have shown that systemic chemotherapy efficacy has varied, some showing no OS benefit; others showing a modest benefit. It has also been noted that advanced-stage PSC has minimal response to chemotherapy. Further larger prospective studies are needed to outline the efficacy and role of systemic chemotherapy and other therapeutic agents, including targeted therapies and immunotherapy.1,4,11-13 However, two-thirds of patients are not sensitive to conventional chemotherapy. In comparison with other types of NSCLC, PSC carries a poor prognosis even in early-stage disease or if tumor metastasis is present. Therefore, further research on novel treatment options is needed to improve long-term survival.1,3-8
Conclusions
PSC is diagnostically challenging because it is rare and has an aggressive progression. Identification of this tumor requires knowledge of histological criteria to identify their subtypes. Immunohistochemistry has an important role in the classification and to rule out differential diagnoses, including metastatic spread. Nevertheless, a reliable diagnosis requires precise histopathological examination, reached with surgical resection. Therefore, a detailed history, physical examination, systematic investigation, and correlation with chest imaging are needed to avoid misdiagnosis.
Our case highlights the importance of keeping this rare, aggressive tumor as part of the differential diagnosis. In view of its natural history, heterogeneity, and low incidence, published cases of PSC are limited. Thus, further investigation could optimize rapid identification and treatment options.
Discussion
An interdisciplinary discussion regarding the diagnosis considered the clinical features of the patient along with the imaging characteristics. The histological examination demonstrating sarcomatoid features with the supporting immunohistochemistry that confirmed both mesenchymal and epithelioid presence and was used to make the diagnosis of pulmonary sarcomatoid carcinoma (PSC).
The incidence of PSC ranges between 0.1% and 0.4% of all lung malignancies.1,4-7 PSC usually occurs in older men whose weight is moderate to heavy and who smoke. PSC appears to have an upper lobe predilection; also, these tumors tend to be bulky with invasive tendency, early recurrence, and systemic metastases. PSC frequently involves the adjacent lung, chest wall, diaphragm, pericardium, and other tissues.1-5 The source of the sarcoma component of the PSC remains uncertain. However, prior research suggests that it is associated with a clonal evolution that induces epidermal and mesenchymal tumor histological characteristics.1,8,9 The tumor cell epithelial-mesenchymal transition may induce transformation of the carcinoma component of PSC to into a sarcoma component. The epithelial-mesenchymal transition is associated with the PSC high risk for invasiveness and induces metastasis sites, such as the esophagus, colon, rectum, kidneys, and the common sites of NSCLC.
The most common symptoms include productive cough, chest congestion, and chest pain.1,7 In view of PSC’s clinical presentation and imaging, numerous differential diagnoses should be considered, such as sarcomatoid carcinomas, primary or secondary metastatic sarcomas, malignant melanoma, and pleural mesothelioma.6,10
The tumor is initially identified by a chest CT, confirmed by histology and immunohistochemistry. Several biomarkers are useful for diagnosis and classification of an undifferentiated neoplasm/tumor of uncertain origin. Those biomarkers help to understand the tumor pathobiology, to select the therapeutic regimen, and to predict the patient’s outcome. Although immunohistochemical staining of epithelial and mesenchymal markers can be helpful, a reliable diagnosis requires a precise histopathological examination. This is often difficult on small biopsy samples, such as fine-needle aspiration, as all the histological elements of PSC required to make the correct evaluation may not be present. Adequate sampling to generate a considerable number of histological slides is essential for an accurate diagnosis, which can be reached only with surgical resection.5
Due to rarity, rapid progression, short survival, and heterogeneous pathological qualities, PSC has been difficult to formulate treatment recommendations. Compared with other histological subtypes of NSCLC, PSC is more aggressive and has a poor prognosis. Survival time on average is about 13.3 months due to early metastasis, lower than other types of NSCLC. The greatest overall survival (OS) benefit has been shown with surgery in early-stage operable PSC, which remains the standard of care. Because most patients with PSC present in the advanced stage, they lose their opportunity for curative surgery. Auxiliary methods of treatment include radiotherapy and chemotherapy. Prior studies have shown that systemic chemotherapy efficacy has varied, some showing no OS benefit; others showing a modest benefit. It has also been noted that advanced-stage PSC has minimal response to chemotherapy. Further larger prospective studies are needed to outline the efficacy and role of systemic chemotherapy and other therapeutic agents, including targeted therapies and immunotherapy.1,4,11-13 However, two-thirds of patients are not sensitive to conventional chemotherapy. In comparison with other types of NSCLC, PSC carries a poor prognosis even in early-stage disease or if tumor metastasis is present. Therefore, further research on novel treatment options is needed to improve long-term survival.1,3-8
Conclusions
PSC is diagnostically challenging because it is rare and has an aggressive progression. Identification of this tumor requires knowledge of histological criteria to identify their subtypes. Immunohistochemistry has an important role in the classification and to rule out differential diagnoses, including metastatic spread. Nevertheless, a reliable diagnosis requires precise histopathological examination, reached with surgical resection. Therefore, a detailed history, physical examination, systematic investigation, and correlation with chest imaging are needed to avoid misdiagnosis.
Our case highlights the importance of keeping this rare, aggressive tumor as part of the differential diagnosis. In view of its natural history, heterogeneity, and low incidence, published cases of PSC are limited. Thus, further investigation could optimize rapid identification and treatment options.
1. Qin Z, Huang B, Yu G, Zheng Y, Zhao K. Gingival metastasis of a mediastinal pulmonary sarcomatoid carcinoma: a case report. J Cardiothorac Surg. 2019;14(1):161. Published 2019 Sep 9. doi:10.1186/s13019-019-0991-y
2. Travis WD, Brambilla E, Nicholson AG, et al; WHO Panel. The 2015 World Health Organization Classification of Lung Tumors: Impact of Genetic, Clinical and Radiologic Advances Since the 2004 Classification. J Thorac Oncol. 2015;10(9):1243-1260. doi:10.1097/JTO.0000000000000630
3. Yendamuri S, Caty L, Pine M, et al. Outcomes of sarcomatoid carcinoma of the lung: a Surveillance, Epidemiology, and End Results Database analysis. Surgery. 2012;152(3):397-402. doi:10.1016/j.surg.2012.05.007
4. Karim NA, Schuster J, Eldessouki I, et al. Pulmonary sarcomatoid carcinoma: University of Cincinnati experience. Oncotarget. 2017;9(3):4102-4108. Published 2017 Dec 18. doi:10.18632/oncotarget.23468
5. Weissferdt A. Pulmonary sarcomatoid carcinomas: a review. Adv Anat Pathol. 2018;25(5):304-313. doi:10.1097/PAP.0000000000000202
6. Roesel C, Terjung S, Weinreich G, et al. Sarcomatoid carcinoma of the lung: a rare histological subtype of non-small cell lung cancer with a poor prognosis even at earlier tumour stages. Interact Cardiovasc Thorac Surg. 2017;24(3):407-413. doi:10.1093/icvts/ivw392
7. Franks TJ, Galvin JR. Sarcomatoid carcinoma of the lung: histologic criteria and common lesions in the differential diagnosis. Arch Pathol Lab Med. 2010;134(1):49-54. doi:10.5858/2008-0547-RAR.1
8. Thomas VT, Hinson S, Konduri K. Epithelial-mesenchymal transition in pulmonary carcinosarcoma: case report and literature review. Ther Adv Med Oncol. 2012;4(1):31-37. doi:10.1177/1758834011421949
9. Chang YL, Wu CT, Shih JY, Lee YC. EGFR and p53 status of pulmonary pleomorphic carcinoma: implications for EGFR tyrosine kinase inhibitors therapy of an aggressive lung malignancy. Ann Surg Oncol. 2011;18(10):2952-2960. doi:10.1245/s10434-011-1621-7
10. Travis WD, Brambilla E, Burke AP, Marx A, Nicholson AG, eds. WHO Classification of Tumours of the Lung, Pleura, Thymus and Heart. 4th ed. International Agency for Research on Cancer; 2015:88-94.
11. Huang SY, Shen SJ, Li XY. Pulmonary sarcomatoid carcinoma: a clinicopathologic study and prognostic analysis of 51 cases. World J Surg Oncol. 2013;11:252. Published 2013 Oct 2. doi:10.1186/1477-7819-11-252
12. Pelosi G, Sonzogni A, De Pas T, et al. Review article: pulmonary sarcomatoid carcinomas: a practical overview. Int J Surg Pathol. 2010;18(2):103-120. doi:10.1177/1066896908330049
13. Lin F, Liu H. Immunohistochemistry in undifferentiated neoplasm/tumor of uncertain origin. Arch Pathol Lab Med. 2014;138(12):1583-1610. doi:10.5858/arpa.2014-0061-RA
1. Qin Z, Huang B, Yu G, Zheng Y, Zhao K. Gingival metastasis of a mediastinal pulmonary sarcomatoid carcinoma: a case report. J Cardiothorac Surg. 2019;14(1):161. Published 2019 Sep 9. doi:10.1186/s13019-019-0991-y
2. Travis WD, Brambilla E, Nicholson AG, et al; WHO Panel. The 2015 World Health Organization Classification of Lung Tumors: Impact of Genetic, Clinical and Radiologic Advances Since the 2004 Classification. J Thorac Oncol. 2015;10(9):1243-1260. doi:10.1097/JTO.0000000000000630
3. Yendamuri S, Caty L, Pine M, et al. Outcomes of sarcomatoid carcinoma of the lung: a Surveillance, Epidemiology, and End Results Database analysis. Surgery. 2012;152(3):397-402. doi:10.1016/j.surg.2012.05.007
4. Karim NA, Schuster J, Eldessouki I, et al. Pulmonary sarcomatoid carcinoma: University of Cincinnati experience. Oncotarget. 2017;9(3):4102-4108. Published 2017 Dec 18. doi:10.18632/oncotarget.23468
5. Weissferdt A. Pulmonary sarcomatoid carcinomas: a review. Adv Anat Pathol. 2018;25(5):304-313. doi:10.1097/PAP.0000000000000202
6. Roesel C, Terjung S, Weinreich G, et al. Sarcomatoid carcinoma of the lung: a rare histological subtype of non-small cell lung cancer with a poor prognosis even at earlier tumour stages. Interact Cardiovasc Thorac Surg. 2017;24(3):407-413. doi:10.1093/icvts/ivw392
7. Franks TJ, Galvin JR. Sarcomatoid carcinoma of the lung: histologic criteria and common lesions in the differential diagnosis. Arch Pathol Lab Med. 2010;134(1):49-54. doi:10.5858/2008-0547-RAR.1
8. Thomas VT, Hinson S, Konduri K. Epithelial-mesenchymal transition in pulmonary carcinosarcoma: case report and literature review. Ther Adv Med Oncol. 2012;4(1):31-37. doi:10.1177/1758834011421949
9. Chang YL, Wu CT, Shih JY, Lee YC. EGFR and p53 status of pulmonary pleomorphic carcinoma: implications for EGFR tyrosine kinase inhibitors therapy of an aggressive lung malignancy. Ann Surg Oncol. 2011;18(10):2952-2960. doi:10.1245/s10434-011-1621-7
10. Travis WD, Brambilla E, Burke AP, Marx A, Nicholson AG, eds. WHO Classification of Tumours of the Lung, Pleura, Thymus and Heart. 4th ed. International Agency for Research on Cancer; 2015:88-94.
11. Huang SY, Shen SJ, Li XY. Pulmonary sarcomatoid carcinoma: a clinicopathologic study and prognostic analysis of 51 cases. World J Surg Oncol. 2013;11:252. Published 2013 Oct 2. doi:10.1186/1477-7819-11-252
12. Pelosi G, Sonzogni A, De Pas T, et al. Review article: pulmonary sarcomatoid carcinomas: a practical overview. Int J Surg Pathol. 2010;18(2):103-120. doi:10.1177/1066896908330049
13. Lin F, Liu H. Immunohistochemistry in undifferentiated neoplasm/tumor of uncertain origin. Arch Pathol Lab Med. 2014;138(12):1583-1610. doi:10.5858/arpa.2014-0061-RA
Renal Replacement Therapy in a Patient Diagnosed With Pancreatitis Secondary to Severe Leptospirosis
In areas where the zoonotic disease leptospirosis is endemic, reduced morbidity and mortality is strongly linked to quick initiation of renal replacement therapy.
Leptospirosis (LS) is considered the most common and widespread zoonotic disease in the world. Numerous outbreaks have occurred in the past 10 years. Due to its technically difficult diagnosis, LS is severely underrecognized, underdiagnosed, and therefore, underreported.1,2 The Centers for Disease Control and Prevention (CDC) estimate 100 to 150 cases of LS are identified annually in the US, with about 50% of those cases occurring in Puerto Rico (PR).3 Specifically in PR, about 15 to 100 cases of suspected LS were reported annually between 2000 and 2009, with 59 cases and 1 death reported in 2010. The data are thought to be severely underreported due to a lack of widespread diagnostic testing availability in PR and no formal veterinary and environmental surveillance programs to monitor the incidence of animal cases and actual circulating serovars.4
A recent systematic review of 80 studies from 34 countries on morbidity and mortality of LS revealed that the global incidence and mortality is about 1.03 million cases and 58,900 deaths every year. Almost half of the reported deaths were adult males aged 20 to 49 years.5 Although mild cases of LS are not associated with an elevated mortality, icteric LS with renal failure (Weil disease) carries a mortality rate of 10%.6 In patients who develop hemorrhagic pneumonitis, mortality may be as high as 50 to 70%.7 Therefore, it is pivotal that clinicians recognize the disease early, that novel modalities of treatment continue to be developed, and that their impact on patient morbidity and mortality are studied and documented.
Case Presentation
A 43-year-old man with a medical history of schizophrenia presented to the emergency department at the US Department of Veterans Affairs (VA) Caribbean Healthcare System in San Juan, PR, after experiencing 1 week of intermittent fever, myalgia, and general weakness. Emergency medical services had found him disheveled and in a rodent-infested swamp area several days before admission. Initial vital signs were within normal limits.
On physical examination, the patient was afebrile, without acute distress, but he had diffuse jaundice and mild epigastric tenderness without evidence of peritoneal irritation. His complete blood count was remarkable for leukocytosis with left shifting, adequate hemoglobin levels but with 9 × 103 U/L platelets. The complete metabolic panel demonstrated an aspartate aminotransferase level of 564 U/L, alanine transaminase level of 462 U/L, total bilirubin of 12 mg/dL, which 10.2 mg/dL were direct bilirubin, and an alkaline phosphate of 345 U/L. Lipase levels were measured at 626 U/L. Marked coagulopathy also was present. The toxicology panel, including acetaminophen and salicylate acid levels, did not reveal the presence of any of the tested substances, and chest imaging did not demonstrate any infiltrates.
An abdominal ultrasound was negative for acute cholestatic pathologies, such as cholelithiasis, cholecystitis, or choledocholithiasis. Nonetheless, a noncontrast abdominopelvic computed tomography was remarkable for peripancreatic fat stranding, which raised suspicion for a diagnosis of pancreatitis.
Once the patient was transferred to the intensive care unit, he developed several episodes of hematemesis, leading to hemodynamical instability and severe respiratory distress. Due to anticipated respiratory failure and need for airway securement, endotracheal intubation was performed. Multiple packed red blood cells were transfused, and the patient was started in vasopressor support.
Diagnosis
A presumptive diagnosis of LS was made due to a considerable history of rodent exposure. The patient was started on broad-spectrum parenteral antibiotics, vancomycin 750 mg every 24 hours, metronidazole 500 mg every 8 hours, and ceftriaxone 2 g IV daily for adequate coverage against Leptospira spp. Despite 72 hours of antibiotic treatment, the patient’s clinical state deteriorated. He required high dosages of norepinephrine (1.5 mcg/kg/min) and vasopressin (0.03 U/min) to maintain adequate organ perfusion. Despite lung protective settings with low tidal volume and a high positive end-expiratory pressure, there was difficulty maintaining adequate oxygenation. Chest imaging was remarkable for bilateral infiltrates concerning for acute respiratory distress syndrome (ARDS).
The coagulopathy and cholestasis continued to worsen, and the renal failure progressed from nonoliguric to anuric. Because of this progression, the patient was started on continuous renal replacement therapy (CRRT) by hemodialysis. Within 24 hours of initiating CRRT, the patient’s clinical status improved dramatically. Vasopressor support was weaned, the coagulopathy resolved, and the cholestasis was improving. The patient’s respiratory status improved in such a manner that he was extubated by the seventh day after being placed on mechanical ventilation. The urine and blood samples sent for identification of Leptospira spp. through polymerase chain reaction (PCR) returned positive by the ninth day of admission. While on CRRT, the patient’s renal function eventually returned to baseline, and he was discharged 12 days after admission.
Discussion
The spirochetes of the genus Leptospira include both saprophytic and pathogenic species. These pathogenic Leptospira spp. have adapted to a grand variety of zoonotic hosts, the most important being rodents. They serve as vectors for the contraction of the disease by humans. Initial infection in rodents by Leptospira spp. causes a systemic illness followed by a persistent colonization of renal tubules from which they are excreted in the urine and into the environment. Humans, in turn, are an incidental host unable to induce a carrier state for the transmission of the pathogenic organism.1 The time from exposure to onset of symptoms, or incubation phase, averages 7 to 12 days but may range from 3 to 30 days.8
LS has been described as having 2 discernable but often coexisting phases. The first, an acute febrile bacteremic phase, has been noted to last about 9 days in about 85% of patients, although a minority have persistent fever from 2 weeks to > 30 days. A second phase, the immune or inflammatory phase, is characterized by a second fever spike preceded by 1 to 5 afebrile days in which there is presence of IgM antibodies and resolution of leptospiremia but positive urine cultures.9 Weil disease may present as the second phase of the disease or as a single, progressive illness from its first manifestation. It is characterized by a triad of jaundice, renal failure, and hemorrhage or coagulopathy.10 Weil disease is of great concern and importance due to its associated higher mortality than that found with the mildest form of the disease.
There are studies that advocate for RRT as an intricate part of the treatment regimen in LS to remove the inflammatory cytokines produced as a reaction to the spirochete.11 In tropical countries with a higher incidence of the disease, leptospirosis is an important cause of acute kidney injury (AKI), depending on multiple factors, including the AKI definition that is used.12 Renal invasion by Leptospira spp. produces acute tubular necrosis (ATN) and cell edema during the first week and then could progress to acute interstitial nephritis (AIN) in 2 to 3 weeks. It is believed that the mechanism for the Leptospira spp. invasion of the tubules that results in damage is associated with the antigenic components in its outer membrane; the most important outer membrane protein expressed during infection is LipL32. This protein increases the production of proinflammatory proteins, such as inducible nitric oxide synthase, monocyte chemotactic protein-1 (CCL2/MCP-1), T cells, and tumor necrosis factor.13
Although doxycycline has been recommended for the prophylaxis and treatment of mild LS, the preferred agent and the conferred benefits of antibiotic treatment overall for the severe form of the disease has been controversial. Traditionally, penicillin G sodium has been recommended as the first-line antibiotic treatment for moderate-to-severe LS.14 Nonetheless, there has been an increasing pattern of penicillin resistance among Leptospira spp. that has prompted the study and use of alternative agents.
An open-label, randomized comparison of parenteral cefotaxime, penicillin G sodium, and doxycycline for the treatment of suspected severe leptospirosis conducted by Suputtamongkol and colleagues showed no difference in mortality, defervescence, or time to resolution of abnormal laboratory findings.15 Current CDC recommendations include the use of parenteral penicillin 1.5 MU every 6 hours as the drug of choice, with ceftriaxone 1 g administered IV every 24 hours equally as effective.3
In addition to antimicrobial therapy, supportive care has shifted to include hemodialysis in those patients who develop AKI as part of the disease. Andrade and colleagues conducted a study of 33 patients with LS in Brazil that was set to compare the impact of door-to-dialysis time and dosage of hemodialysis on mortality. In patients with a quicker door-to-dialysis time and daily hemodialysis sessions, there was a 50% (16.7% vs 66.7%) absolute mortality reduction when compared with those with delayed initiation and alternate-day hemodialysis sessions.11 A follow-up prospective study compared the use of traditional sustained low-efficiency dialysis (SLED) with the use of extended SLED via hemodiafiltration in patients with LS presenting with ARDS and AKI. Although hemodiafiltration resulted in a relative decrease in serum levels of interleukin (IL)-17, IL-7, and CCL2/MCP-1, there was no significant difference in mortality.16 The most important prognostic factor in severe LS presenting with AKI and relating to RRT is a shorter door-to-dialysis time and increased dose, not the mode of dialysis clearance. Nonetheless, both RRT methods resulted in a progressive decrease in inflammatory mediators that have been associated with ATN and AIN in the context of LS.16 The authors argue that using CRRT instead of SLED via hemodiafiltration could have accentuated the effects of the reduction that inflammatory mediators may have on mortality in patients with severe LS.
Conclusions
LS continues to be of interest due to its current status as the most common zoonotic disease and its widespread prevalence throughout the globe. Novel treatment modalities for LS, specifically for Weil disease, continue to be developed with the goal of reducing the current mortality rate associated with the disease.
In endemic areas, prompt recognition is essential to initiate the recommended therapy. Parenteral antibiotics, such as penicillin G sodium and ceftriaxone, continue to be the mainstay of treatment and constitute the current CDC recommendations. Nonetheless, early initiation of CRRT has been shown to greatly reduce the mortality associated with Weil disease and, when available, should be considered in these patients.
Our patient failed to improve while receiving parenteral antibiotics alone but showed marked improvement after being placed on CRRT. Furthermore, initiation of CRRT resulted in near-complete resolution of his organ dysfunction and eventual discharge from the hospital. This case serves to further support the use of early CRRT as part of the standard of care in severe LS.
1. Ko AI, Goarant C, Picardeau M. Leptospira: the dawn of the molecular genetics era for an emerging zoonotic pathogen. Nat Rev Microbiol. 2009;7(10):736-747. doi:10.1038/nrmicro2208
2. Hartskeerl RA, Collares-Pereira M, Ellis WA. Emergence, control and re-emerging leptospirosis: dynamics of infection in the changing world. Clin Microbiol Infect. 2011;17(4):494-501. doi:10.1111/j.1469-0691.2011.03474.x
3. Centers for Disease Control and Prevention. Leptospirosis fact sheet for clinicians, CS287535B. https://www.cdc.gov/leptospirosis/pdf/fs-leptospirosis-clinicians-eng-508.pdf. Published January 30, 2018. Accessed October 9, 2020.
4. Martinez-Recio C, Rodriguez-Cintron W, Galarza-Vargas S, et al. The brief case: cases from 3 hospitals in Puerto Rico. ACP Hosp. https://acphospitalist.org/archives/2014/09/briefcase.htm. Published September 2014. Accessed October 9, 2020.
5. Costa F, Hagan JE, Calcagno J, et al. Global morbidity and mortality of leptospirosis: a systematic review. PLoS Negl Trop Dis. 2015;9(9):e0003898. doi:10.1371/journal.pntd.0003898
6. Levett PN. Leptospirosis. Clin Microbiol Rev. 2001;14(2):296-326. doi:10.1128/CMR.14.2.296-326.2001
7. Vijayachari P, Sugunan AP, Shriram AN. Leptospirosis: an emerging global public health problem. J Biosci. 2008;33(4):557-569. doi:10.1007/s12038-008-0074-z
8. Haake DA, Levett PN. Leptospirosis in humans. In: Adler B, ed. Leptospira and Leptospirosis. Berlin, Heidelberg: Springer-Verlag Berlin Heidelberg; 2015:65-97. doi:10.1007/978-3-662-45059-8_5
9. Berman SJ. Sporadic anicteric leptospirosis in South Vietnam: a study in 150 patients. Ann Intern Med. 1973;79(2):167. doi:10.7326/0003-4819-79-2-167
10. Bharti AR, Nally JE, Ricaldi JN, et al. Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis. 2003;3(12):757-771. doi:10.1016/S1473-3099(03)00830-2
11. Andrade L, Cleto S, Seguro AC. Door-to-dialysis time and daily hemodialysis in patients with leptospirosis: impact on mortality. Clin J Am Soc Nephrol. 2007;2(4):739–744. doi: 10.2215/CJN.00680207
12. Mathew A, George J. Acute kidney injury in the tropics. Ann Saudi Med. 2011;31(5):451-456. doi:10.4103/0256-4947.84620
13. Daher EF, Silva GB Jr, Karbage NNN, et al. Predictors of oliguric acute kidney injury in leptospirosis. Nephron Clin Pract. 2009;112(1):c25-c30. doi:10.1159/000210571
14. Panaphut T, Domrongkitchaiporn S, Vibhagool A, Thinkamrop B, Susaengrat W. Ceftriaxone compared with sodium penicillin g for treatment of severe leptospirosis. Clin Infect Dis. 2003;36(12):1507-1513. doi:10.1086/375226
15. Suputtamongkol Y, Niwattayakul K, Suttinont C, et al. An open, randomized, controlled trial of penicillin, doxycycline, and cefotaxime for patients with severe leptospirosis. Clin Infect Dis. 2004;39(10):1417-1424. doi:10.1086/425001
16. Cleto SA, Rodrigues CE, Malaque CM, Sztajnbok J, Seguro AC, Andrade L. Hemodiafiltration decreases serum levels of inflammatory mediators in severe leptospirosis: a prospective study. PLoS ONE. 2016;11(8):e0160010. doi:10.1371/journal.pone.0160010
In areas where the zoonotic disease leptospirosis is endemic, reduced morbidity and mortality is strongly linked to quick initiation of renal replacement therapy.
In areas where the zoonotic disease leptospirosis is endemic, reduced morbidity and mortality is strongly linked to quick initiation of renal replacement therapy.
Leptospirosis (LS) is considered the most common and widespread zoonotic disease in the world. Numerous outbreaks have occurred in the past 10 years. Due to its technically difficult diagnosis, LS is severely underrecognized, underdiagnosed, and therefore, underreported.1,2 The Centers for Disease Control and Prevention (CDC) estimate 100 to 150 cases of LS are identified annually in the US, with about 50% of those cases occurring in Puerto Rico (PR).3 Specifically in PR, about 15 to 100 cases of suspected LS were reported annually between 2000 and 2009, with 59 cases and 1 death reported in 2010. The data are thought to be severely underreported due to a lack of widespread diagnostic testing availability in PR and no formal veterinary and environmental surveillance programs to monitor the incidence of animal cases and actual circulating serovars.4
A recent systematic review of 80 studies from 34 countries on morbidity and mortality of LS revealed that the global incidence and mortality is about 1.03 million cases and 58,900 deaths every year. Almost half of the reported deaths were adult males aged 20 to 49 years.5 Although mild cases of LS are not associated with an elevated mortality, icteric LS with renal failure (Weil disease) carries a mortality rate of 10%.6 In patients who develop hemorrhagic pneumonitis, mortality may be as high as 50 to 70%.7 Therefore, it is pivotal that clinicians recognize the disease early, that novel modalities of treatment continue to be developed, and that their impact on patient morbidity and mortality are studied and documented.
Case Presentation
A 43-year-old man with a medical history of schizophrenia presented to the emergency department at the US Department of Veterans Affairs (VA) Caribbean Healthcare System in San Juan, PR, after experiencing 1 week of intermittent fever, myalgia, and general weakness. Emergency medical services had found him disheveled and in a rodent-infested swamp area several days before admission. Initial vital signs were within normal limits.
On physical examination, the patient was afebrile, without acute distress, but he had diffuse jaundice and mild epigastric tenderness without evidence of peritoneal irritation. His complete blood count was remarkable for leukocytosis with left shifting, adequate hemoglobin levels but with 9 × 103 U/L platelets. The complete metabolic panel demonstrated an aspartate aminotransferase level of 564 U/L, alanine transaminase level of 462 U/L, total bilirubin of 12 mg/dL, which 10.2 mg/dL were direct bilirubin, and an alkaline phosphate of 345 U/L. Lipase levels were measured at 626 U/L. Marked coagulopathy also was present. The toxicology panel, including acetaminophen and salicylate acid levels, did not reveal the presence of any of the tested substances, and chest imaging did not demonstrate any infiltrates.
An abdominal ultrasound was negative for acute cholestatic pathologies, such as cholelithiasis, cholecystitis, or choledocholithiasis. Nonetheless, a noncontrast abdominopelvic computed tomography was remarkable for peripancreatic fat stranding, which raised suspicion for a diagnosis of pancreatitis.
Once the patient was transferred to the intensive care unit, he developed several episodes of hematemesis, leading to hemodynamical instability and severe respiratory distress. Due to anticipated respiratory failure and need for airway securement, endotracheal intubation was performed. Multiple packed red blood cells were transfused, and the patient was started in vasopressor support.
Diagnosis
A presumptive diagnosis of LS was made due to a considerable history of rodent exposure. The patient was started on broad-spectrum parenteral antibiotics, vancomycin 750 mg every 24 hours, metronidazole 500 mg every 8 hours, and ceftriaxone 2 g IV daily for adequate coverage against Leptospira spp. Despite 72 hours of antibiotic treatment, the patient’s clinical state deteriorated. He required high dosages of norepinephrine (1.5 mcg/kg/min) and vasopressin (0.03 U/min) to maintain adequate organ perfusion. Despite lung protective settings with low tidal volume and a high positive end-expiratory pressure, there was difficulty maintaining adequate oxygenation. Chest imaging was remarkable for bilateral infiltrates concerning for acute respiratory distress syndrome (ARDS).
The coagulopathy and cholestasis continued to worsen, and the renal failure progressed from nonoliguric to anuric. Because of this progression, the patient was started on continuous renal replacement therapy (CRRT) by hemodialysis. Within 24 hours of initiating CRRT, the patient’s clinical status improved dramatically. Vasopressor support was weaned, the coagulopathy resolved, and the cholestasis was improving. The patient’s respiratory status improved in such a manner that he was extubated by the seventh day after being placed on mechanical ventilation. The urine and blood samples sent for identification of Leptospira spp. through polymerase chain reaction (PCR) returned positive by the ninth day of admission. While on CRRT, the patient’s renal function eventually returned to baseline, and he was discharged 12 days after admission.
Discussion
The spirochetes of the genus Leptospira include both saprophytic and pathogenic species. These pathogenic Leptospira spp. have adapted to a grand variety of zoonotic hosts, the most important being rodents. They serve as vectors for the contraction of the disease by humans. Initial infection in rodents by Leptospira spp. causes a systemic illness followed by a persistent colonization of renal tubules from which they are excreted in the urine and into the environment. Humans, in turn, are an incidental host unable to induce a carrier state for the transmission of the pathogenic organism.1 The time from exposure to onset of symptoms, or incubation phase, averages 7 to 12 days but may range from 3 to 30 days.8
LS has been described as having 2 discernable but often coexisting phases. The first, an acute febrile bacteremic phase, has been noted to last about 9 days in about 85% of patients, although a minority have persistent fever from 2 weeks to > 30 days. A second phase, the immune or inflammatory phase, is characterized by a second fever spike preceded by 1 to 5 afebrile days in which there is presence of IgM antibodies and resolution of leptospiremia but positive urine cultures.9 Weil disease may present as the second phase of the disease or as a single, progressive illness from its first manifestation. It is characterized by a triad of jaundice, renal failure, and hemorrhage or coagulopathy.10 Weil disease is of great concern and importance due to its associated higher mortality than that found with the mildest form of the disease.
There are studies that advocate for RRT as an intricate part of the treatment regimen in LS to remove the inflammatory cytokines produced as a reaction to the spirochete.11 In tropical countries with a higher incidence of the disease, leptospirosis is an important cause of acute kidney injury (AKI), depending on multiple factors, including the AKI definition that is used.12 Renal invasion by Leptospira spp. produces acute tubular necrosis (ATN) and cell edema during the first week and then could progress to acute interstitial nephritis (AIN) in 2 to 3 weeks. It is believed that the mechanism for the Leptospira spp. invasion of the tubules that results in damage is associated with the antigenic components in its outer membrane; the most important outer membrane protein expressed during infection is LipL32. This protein increases the production of proinflammatory proteins, such as inducible nitric oxide synthase, monocyte chemotactic protein-1 (CCL2/MCP-1), T cells, and tumor necrosis factor.13
Although doxycycline has been recommended for the prophylaxis and treatment of mild LS, the preferred agent and the conferred benefits of antibiotic treatment overall for the severe form of the disease has been controversial. Traditionally, penicillin G sodium has been recommended as the first-line antibiotic treatment for moderate-to-severe LS.14 Nonetheless, there has been an increasing pattern of penicillin resistance among Leptospira spp. that has prompted the study and use of alternative agents.
An open-label, randomized comparison of parenteral cefotaxime, penicillin G sodium, and doxycycline for the treatment of suspected severe leptospirosis conducted by Suputtamongkol and colleagues showed no difference in mortality, defervescence, or time to resolution of abnormal laboratory findings.15 Current CDC recommendations include the use of parenteral penicillin 1.5 MU every 6 hours as the drug of choice, with ceftriaxone 1 g administered IV every 24 hours equally as effective.3
In addition to antimicrobial therapy, supportive care has shifted to include hemodialysis in those patients who develop AKI as part of the disease. Andrade and colleagues conducted a study of 33 patients with LS in Brazil that was set to compare the impact of door-to-dialysis time and dosage of hemodialysis on mortality. In patients with a quicker door-to-dialysis time and daily hemodialysis sessions, there was a 50% (16.7% vs 66.7%) absolute mortality reduction when compared with those with delayed initiation and alternate-day hemodialysis sessions.11 A follow-up prospective study compared the use of traditional sustained low-efficiency dialysis (SLED) with the use of extended SLED via hemodiafiltration in patients with LS presenting with ARDS and AKI. Although hemodiafiltration resulted in a relative decrease in serum levels of interleukin (IL)-17, IL-7, and CCL2/MCP-1, there was no significant difference in mortality.16 The most important prognostic factor in severe LS presenting with AKI and relating to RRT is a shorter door-to-dialysis time and increased dose, not the mode of dialysis clearance. Nonetheless, both RRT methods resulted in a progressive decrease in inflammatory mediators that have been associated with ATN and AIN in the context of LS.16 The authors argue that using CRRT instead of SLED via hemodiafiltration could have accentuated the effects of the reduction that inflammatory mediators may have on mortality in patients with severe LS.
Conclusions
LS continues to be of interest due to its current status as the most common zoonotic disease and its widespread prevalence throughout the globe. Novel treatment modalities for LS, specifically for Weil disease, continue to be developed with the goal of reducing the current mortality rate associated with the disease.
In endemic areas, prompt recognition is essential to initiate the recommended therapy. Parenteral antibiotics, such as penicillin G sodium and ceftriaxone, continue to be the mainstay of treatment and constitute the current CDC recommendations. Nonetheless, early initiation of CRRT has been shown to greatly reduce the mortality associated with Weil disease and, when available, should be considered in these patients.
Our patient failed to improve while receiving parenteral antibiotics alone but showed marked improvement after being placed on CRRT. Furthermore, initiation of CRRT resulted in near-complete resolution of his organ dysfunction and eventual discharge from the hospital. This case serves to further support the use of early CRRT as part of the standard of care in severe LS.
Leptospirosis (LS) is considered the most common and widespread zoonotic disease in the world. Numerous outbreaks have occurred in the past 10 years. Due to its technically difficult diagnosis, LS is severely underrecognized, underdiagnosed, and therefore, underreported.1,2 The Centers for Disease Control and Prevention (CDC) estimate 100 to 150 cases of LS are identified annually in the US, with about 50% of those cases occurring in Puerto Rico (PR).3 Specifically in PR, about 15 to 100 cases of suspected LS were reported annually between 2000 and 2009, with 59 cases and 1 death reported in 2010. The data are thought to be severely underreported due to a lack of widespread diagnostic testing availability in PR and no formal veterinary and environmental surveillance programs to monitor the incidence of animal cases and actual circulating serovars.4
A recent systematic review of 80 studies from 34 countries on morbidity and mortality of LS revealed that the global incidence and mortality is about 1.03 million cases and 58,900 deaths every year. Almost half of the reported deaths were adult males aged 20 to 49 years.5 Although mild cases of LS are not associated with an elevated mortality, icteric LS with renal failure (Weil disease) carries a mortality rate of 10%.6 In patients who develop hemorrhagic pneumonitis, mortality may be as high as 50 to 70%.7 Therefore, it is pivotal that clinicians recognize the disease early, that novel modalities of treatment continue to be developed, and that their impact on patient morbidity and mortality are studied and documented.
Case Presentation
A 43-year-old man with a medical history of schizophrenia presented to the emergency department at the US Department of Veterans Affairs (VA) Caribbean Healthcare System in San Juan, PR, after experiencing 1 week of intermittent fever, myalgia, and general weakness. Emergency medical services had found him disheveled and in a rodent-infested swamp area several days before admission. Initial vital signs were within normal limits.
On physical examination, the patient was afebrile, without acute distress, but he had diffuse jaundice and mild epigastric tenderness without evidence of peritoneal irritation. His complete blood count was remarkable for leukocytosis with left shifting, adequate hemoglobin levels but with 9 × 103 U/L platelets. The complete metabolic panel demonstrated an aspartate aminotransferase level of 564 U/L, alanine transaminase level of 462 U/L, total bilirubin of 12 mg/dL, which 10.2 mg/dL were direct bilirubin, and an alkaline phosphate of 345 U/L. Lipase levels were measured at 626 U/L. Marked coagulopathy also was present. The toxicology panel, including acetaminophen and salicylate acid levels, did not reveal the presence of any of the tested substances, and chest imaging did not demonstrate any infiltrates.
An abdominal ultrasound was negative for acute cholestatic pathologies, such as cholelithiasis, cholecystitis, or choledocholithiasis. Nonetheless, a noncontrast abdominopelvic computed tomography was remarkable for peripancreatic fat stranding, which raised suspicion for a diagnosis of pancreatitis.
Once the patient was transferred to the intensive care unit, he developed several episodes of hematemesis, leading to hemodynamical instability and severe respiratory distress. Due to anticipated respiratory failure and need for airway securement, endotracheal intubation was performed. Multiple packed red blood cells were transfused, and the patient was started in vasopressor support.
Diagnosis
A presumptive diagnosis of LS was made due to a considerable history of rodent exposure. The patient was started on broad-spectrum parenteral antibiotics, vancomycin 750 mg every 24 hours, metronidazole 500 mg every 8 hours, and ceftriaxone 2 g IV daily for adequate coverage against Leptospira spp. Despite 72 hours of antibiotic treatment, the patient’s clinical state deteriorated. He required high dosages of norepinephrine (1.5 mcg/kg/min) and vasopressin (0.03 U/min) to maintain adequate organ perfusion. Despite lung protective settings with low tidal volume and a high positive end-expiratory pressure, there was difficulty maintaining adequate oxygenation. Chest imaging was remarkable for bilateral infiltrates concerning for acute respiratory distress syndrome (ARDS).
The coagulopathy and cholestasis continued to worsen, and the renal failure progressed from nonoliguric to anuric. Because of this progression, the patient was started on continuous renal replacement therapy (CRRT) by hemodialysis. Within 24 hours of initiating CRRT, the patient’s clinical status improved dramatically. Vasopressor support was weaned, the coagulopathy resolved, and the cholestasis was improving. The patient’s respiratory status improved in such a manner that he was extubated by the seventh day after being placed on mechanical ventilation. The urine and blood samples sent for identification of Leptospira spp. through polymerase chain reaction (PCR) returned positive by the ninth day of admission. While on CRRT, the patient’s renal function eventually returned to baseline, and he was discharged 12 days after admission.
Discussion
The spirochetes of the genus Leptospira include both saprophytic and pathogenic species. These pathogenic Leptospira spp. have adapted to a grand variety of zoonotic hosts, the most important being rodents. They serve as vectors for the contraction of the disease by humans. Initial infection in rodents by Leptospira spp. causes a systemic illness followed by a persistent colonization of renal tubules from which they are excreted in the urine and into the environment. Humans, in turn, are an incidental host unable to induce a carrier state for the transmission of the pathogenic organism.1 The time from exposure to onset of symptoms, or incubation phase, averages 7 to 12 days but may range from 3 to 30 days.8
LS has been described as having 2 discernable but often coexisting phases. The first, an acute febrile bacteremic phase, has been noted to last about 9 days in about 85% of patients, although a minority have persistent fever from 2 weeks to > 30 days. A second phase, the immune or inflammatory phase, is characterized by a second fever spike preceded by 1 to 5 afebrile days in which there is presence of IgM antibodies and resolution of leptospiremia but positive urine cultures.9 Weil disease may present as the second phase of the disease or as a single, progressive illness from its first manifestation. It is characterized by a triad of jaundice, renal failure, and hemorrhage or coagulopathy.10 Weil disease is of great concern and importance due to its associated higher mortality than that found with the mildest form of the disease.
There are studies that advocate for RRT as an intricate part of the treatment regimen in LS to remove the inflammatory cytokines produced as a reaction to the spirochete.11 In tropical countries with a higher incidence of the disease, leptospirosis is an important cause of acute kidney injury (AKI), depending on multiple factors, including the AKI definition that is used.12 Renal invasion by Leptospira spp. produces acute tubular necrosis (ATN) and cell edema during the first week and then could progress to acute interstitial nephritis (AIN) in 2 to 3 weeks. It is believed that the mechanism for the Leptospira spp. invasion of the tubules that results in damage is associated with the antigenic components in its outer membrane; the most important outer membrane protein expressed during infection is LipL32. This protein increases the production of proinflammatory proteins, such as inducible nitric oxide synthase, monocyte chemotactic protein-1 (CCL2/MCP-1), T cells, and tumor necrosis factor.13
Although doxycycline has been recommended for the prophylaxis and treatment of mild LS, the preferred agent and the conferred benefits of antibiotic treatment overall for the severe form of the disease has been controversial. Traditionally, penicillin G sodium has been recommended as the first-line antibiotic treatment for moderate-to-severe LS.14 Nonetheless, there has been an increasing pattern of penicillin resistance among Leptospira spp. that has prompted the study and use of alternative agents.
An open-label, randomized comparison of parenteral cefotaxime, penicillin G sodium, and doxycycline for the treatment of suspected severe leptospirosis conducted by Suputtamongkol and colleagues showed no difference in mortality, defervescence, or time to resolution of abnormal laboratory findings.15 Current CDC recommendations include the use of parenteral penicillin 1.5 MU every 6 hours as the drug of choice, with ceftriaxone 1 g administered IV every 24 hours equally as effective.3
In addition to antimicrobial therapy, supportive care has shifted to include hemodialysis in those patients who develop AKI as part of the disease. Andrade and colleagues conducted a study of 33 patients with LS in Brazil that was set to compare the impact of door-to-dialysis time and dosage of hemodialysis on mortality. In patients with a quicker door-to-dialysis time and daily hemodialysis sessions, there was a 50% (16.7% vs 66.7%) absolute mortality reduction when compared with those with delayed initiation and alternate-day hemodialysis sessions.11 A follow-up prospective study compared the use of traditional sustained low-efficiency dialysis (SLED) with the use of extended SLED via hemodiafiltration in patients with LS presenting with ARDS and AKI. Although hemodiafiltration resulted in a relative decrease in serum levels of interleukin (IL)-17, IL-7, and CCL2/MCP-1, there was no significant difference in mortality.16 The most important prognostic factor in severe LS presenting with AKI and relating to RRT is a shorter door-to-dialysis time and increased dose, not the mode of dialysis clearance. Nonetheless, both RRT methods resulted in a progressive decrease in inflammatory mediators that have been associated with ATN and AIN in the context of LS.16 The authors argue that using CRRT instead of SLED via hemodiafiltration could have accentuated the effects of the reduction that inflammatory mediators may have on mortality in patients with severe LS.
Conclusions
LS continues to be of interest due to its current status as the most common zoonotic disease and its widespread prevalence throughout the globe. Novel treatment modalities for LS, specifically for Weil disease, continue to be developed with the goal of reducing the current mortality rate associated with the disease.
In endemic areas, prompt recognition is essential to initiate the recommended therapy. Parenteral antibiotics, such as penicillin G sodium and ceftriaxone, continue to be the mainstay of treatment and constitute the current CDC recommendations. Nonetheless, early initiation of CRRT has been shown to greatly reduce the mortality associated with Weil disease and, when available, should be considered in these patients.
Our patient failed to improve while receiving parenteral antibiotics alone but showed marked improvement after being placed on CRRT. Furthermore, initiation of CRRT resulted in near-complete resolution of his organ dysfunction and eventual discharge from the hospital. This case serves to further support the use of early CRRT as part of the standard of care in severe LS.
1. Ko AI, Goarant C, Picardeau M. Leptospira: the dawn of the molecular genetics era for an emerging zoonotic pathogen. Nat Rev Microbiol. 2009;7(10):736-747. doi:10.1038/nrmicro2208
2. Hartskeerl RA, Collares-Pereira M, Ellis WA. Emergence, control and re-emerging leptospirosis: dynamics of infection in the changing world. Clin Microbiol Infect. 2011;17(4):494-501. doi:10.1111/j.1469-0691.2011.03474.x
3. Centers for Disease Control and Prevention. Leptospirosis fact sheet for clinicians, CS287535B. https://www.cdc.gov/leptospirosis/pdf/fs-leptospirosis-clinicians-eng-508.pdf. Published January 30, 2018. Accessed October 9, 2020.
4. Martinez-Recio C, Rodriguez-Cintron W, Galarza-Vargas S, et al. The brief case: cases from 3 hospitals in Puerto Rico. ACP Hosp. https://acphospitalist.org/archives/2014/09/briefcase.htm. Published September 2014. Accessed October 9, 2020.
5. Costa F, Hagan JE, Calcagno J, et al. Global morbidity and mortality of leptospirosis: a systematic review. PLoS Negl Trop Dis. 2015;9(9):e0003898. doi:10.1371/journal.pntd.0003898
6. Levett PN. Leptospirosis. Clin Microbiol Rev. 2001;14(2):296-326. doi:10.1128/CMR.14.2.296-326.2001
7. Vijayachari P, Sugunan AP, Shriram AN. Leptospirosis: an emerging global public health problem. J Biosci. 2008;33(4):557-569. doi:10.1007/s12038-008-0074-z
8. Haake DA, Levett PN. Leptospirosis in humans. In: Adler B, ed. Leptospira and Leptospirosis. Berlin, Heidelberg: Springer-Verlag Berlin Heidelberg; 2015:65-97. doi:10.1007/978-3-662-45059-8_5
9. Berman SJ. Sporadic anicteric leptospirosis in South Vietnam: a study in 150 patients. Ann Intern Med. 1973;79(2):167. doi:10.7326/0003-4819-79-2-167
10. Bharti AR, Nally JE, Ricaldi JN, et al. Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis. 2003;3(12):757-771. doi:10.1016/S1473-3099(03)00830-2
11. Andrade L, Cleto S, Seguro AC. Door-to-dialysis time and daily hemodialysis in patients with leptospirosis: impact on mortality. Clin J Am Soc Nephrol. 2007;2(4):739–744. doi: 10.2215/CJN.00680207
12. Mathew A, George J. Acute kidney injury in the tropics. Ann Saudi Med. 2011;31(5):451-456. doi:10.4103/0256-4947.84620
13. Daher EF, Silva GB Jr, Karbage NNN, et al. Predictors of oliguric acute kidney injury in leptospirosis. Nephron Clin Pract. 2009;112(1):c25-c30. doi:10.1159/000210571
14. Panaphut T, Domrongkitchaiporn S, Vibhagool A, Thinkamrop B, Susaengrat W. Ceftriaxone compared with sodium penicillin g for treatment of severe leptospirosis. Clin Infect Dis. 2003;36(12):1507-1513. doi:10.1086/375226
15. Suputtamongkol Y, Niwattayakul K, Suttinont C, et al. An open, randomized, controlled trial of penicillin, doxycycline, and cefotaxime for patients with severe leptospirosis. Clin Infect Dis. 2004;39(10):1417-1424. doi:10.1086/425001
16. Cleto SA, Rodrigues CE, Malaque CM, Sztajnbok J, Seguro AC, Andrade L. Hemodiafiltration decreases serum levels of inflammatory mediators in severe leptospirosis: a prospective study. PLoS ONE. 2016;11(8):e0160010. doi:10.1371/journal.pone.0160010
1. Ko AI, Goarant C, Picardeau M. Leptospira: the dawn of the molecular genetics era for an emerging zoonotic pathogen. Nat Rev Microbiol. 2009;7(10):736-747. doi:10.1038/nrmicro2208
2. Hartskeerl RA, Collares-Pereira M, Ellis WA. Emergence, control and re-emerging leptospirosis: dynamics of infection in the changing world. Clin Microbiol Infect. 2011;17(4):494-501. doi:10.1111/j.1469-0691.2011.03474.x
3. Centers for Disease Control and Prevention. Leptospirosis fact sheet for clinicians, CS287535B. https://www.cdc.gov/leptospirosis/pdf/fs-leptospirosis-clinicians-eng-508.pdf. Published January 30, 2018. Accessed October 9, 2020.
4. Martinez-Recio C, Rodriguez-Cintron W, Galarza-Vargas S, et al. The brief case: cases from 3 hospitals in Puerto Rico. ACP Hosp. https://acphospitalist.org/archives/2014/09/briefcase.htm. Published September 2014. Accessed October 9, 2020.
5. Costa F, Hagan JE, Calcagno J, et al. Global morbidity and mortality of leptospirosis: a systematic review. PLoS Negl Trop Dis. 2015;9(9):e0003898. doi:10.1371/journal.pntd.0003898
6. Levett PN. Leptospirosis. Clin Microbiol Rev. 2001;14(2):296-326. doi:10.1128/CMR.14.2.296-326.2001
7. Vijayachari P, Sugunan AP, Shriram AN. Leptospirosis: an emerging global public health problem. J Biosci. 2008;33(4):557-569. doi:10.1007/s12038-008-0074-z
8. Haake DA, Levett PN. Leptospirosis in humans. In: Adler B, ed. Leptospira and Leptospirosis. Berlin, Heidelberg: Springer-Verlag Berlin Heidelberg; 2015:65-97. doi:10.1007/978-3-662-45059-8_5
9. Berman SJ. Sporadic anicteric leptospirosis in South Vietnam: a study in 150 patients. Ann Intern Med. 1973;79(2):167. doi:10.7326/0003-4819-79-2-167
10. Bharti AR, Nally JE, Ricaldi JN, et al. Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis. 2003;3(12):757-771. doi:10.1016/S1473-3099(03)00830-2
11. Andrade L, Cleto S, Seguro AC. Door-to-dialysis time and daily hemodialysis in patients with leptospirosis: impact on mortality. Clin J Am Soc Nephrol. 2007;2(4):739–744. doi: 10.2215/CJN.00680207
12. Mathew A, George J. Acute kidney injury in the tropics. Ann Saudi Med. 2011;31(5):451-456. doi:10.4103/0256-4947.84620
13. Daher EF, Silva GB Jr, Karbage NNN, et al. Predictors of oliguric acute kidney injury in leptospirosis. Nephron Clin Pract. 2009;112(1):c25-c30. doi:10.1159/000210571
14. Panaphut T, Domrongkitchaiporn S, Vibhagool A, Thinkamrop B, Susaengrat W. Ceftriaxone compared with sodium penicillin g for treatment of severe leptospirosis. Clin Infect Dis. 2003;36(12):1507-1513. doi:10.1086/375226
15. Suputtamongkol Y, Niwattayakul K, Suttinont C, et al. An open, randomized, controlled trial of penicillin, doxycycline, and cefotaxime for patients with severe leptospirosis. Clin Infect Dis. 2004;39(10):1417-1424. doi:10.1086/425001
16. Cleto SA, Rodrigues CE, Malaque CM, Sztajnbok J, Seguro AC, Andrade L. Hemodiafiltration decreases serum levels of inflammatory mediators in severe leptospirosis: a prospective study. PLoS ONE. 2016;11(8):e0160010. doi:10.1371/journal.pone.0160010
Recurrent Angiotensin-Converting Enzyme Inhibitor-Induced Angioedema Refractory to Fresh Frozen Plasma
Angioedema induced by angiotensin-converting enzyme inhibitors (ACEIs) is present in from 0.1% to 0.7% of treated patients and more often involves the head, neck, face, lips, tongue, and larynx.1 ACEI-induced angioedema results from inhibition of angiotensin-converting enzyme (ACE), which results in reduced degradation and resultant accumulation of bradykinin, a potent inflammatory mediator.2
The treatment of choice is discontinuing all ACEIs; however, the patient may be at increased risk of a subsequent angioedema attack for many weeks.3 Antihistamines (H1 and H2 receptor blockade), epinephrine, and glucocorticoids are effective in allergic/histaminergic angioedema but are usually ineffective for hereditary angioedema or ACEI angioedema and are not recommended for acute therapy.4 Kallikrein-bradykinin pathway targeted therapies are now approved by the Food and Drug Administration (FDA) for hereditary angioedema attacks and have been studied for ACEI-induced angioedema. Ecallantide and icatibant inhibit conversion of precursors to bradykinin. Multiple randomized trials of ecallantide have not shown any advantage over traditional therapies.5 On the other hand, icatibant has shown resolution of angioedema in several case reports and in a randomized trial.6 Icatibant for ACEI-induced angioedema continues to be off-label because the data are conflicting.
Case Presentation
A 67-year-old man presented with a medical history of arterial hypertension (diagnosed 17 years previously), hypercholesterolemia, type 2 diabetes mellitus, alcohol dependence, and obesity. His outpatient medications included simvastatin, aripiprazole, losartan/hydrochlorothiazide, and amlodipine. He was voluntarily admitted for inpatient detoxification. After evaluation by the internist, medication reconciliation was done, and the therapy was adjusted according to medication availability. He reported having no drug allergies, and the losartan was changed for lisinopril. About 24 hours after the first dose of lisinopril, the patient developed swelling of the lips. Antihistamine and IV steroids were administered, and the ACEI was discontinued. His baseline vital signs were temperature 98° F, heart rate 83 beats per minute, respiratory rate 19 breaths per minute, blood pressure 150/94, and oxygen saturation 98% by pulse oximeter.
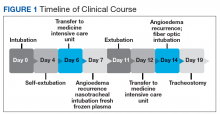
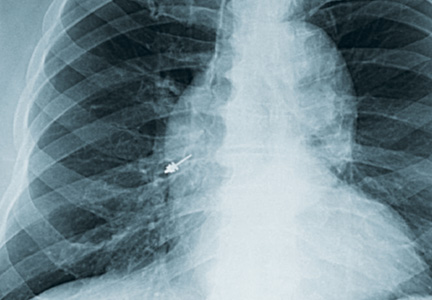
During the night shift the patient’s symptoms worsened, developing difficulty swallowing and shortness of breath. He was transferred to the medicine intensive care unit (MICU), intubated, and placed on mechanical ventilation to protect his airway. Laryngoscopic examination was notable for edematous tongue, uvula, and larynx. Also, the patient had mild stridor. His laboratory test results showed normal levels of complement, tryptase, and C1 esterase. On the fourth day after admission to MICU (Figure 1), the patient extubated himself. At that time, he did not present stridor or respiratory distress and remained at the MICU for 24 hours for close monitoring.
Thirty-six hours after self-extubation the patient developed stridor and shortness of breath at the general medicine ward. In view of his clinical presentation of recurrent ACEI-induced angioedema, the Anesthesiology Service was consulted. Direct visualization of the airways showed edema of the epiglottis and vocal cords, requiring nasotracheal intubation. Two units of fresh frozen plasma (FFP) were administered. Complete resolution of angioedema took at least 72 hours even after the administration of FFP. As part of the ventilator-associated pneumonia prevention bundle, the patient continued with daily spontaneous breathing trials. On the fourth day, he was he was extubated after a cuff-leak test was positive and his rapid shallow breathing index was adequate.
The cuff-leak test is usually done to predict postextubation stridor. It consists of deflating the endotracheal tube cuff to verify if gas can pass around the tube. Absence of cuff leak is suggestive of airway edema, a risk factor for postextubation stridor and failure of extubation. For example, if the patient has an endotracheal tube that is too large in relation to the patient’s airway, the leak test can result in a false negative. In this case, fiber optic visualization of the airway can confirm the endotracheal tube occluding all the airway even with the cuff deflated and without evidence of swelling of the vocal cords. The rapid shallow breathing index is a ratio of respiratory rate over tidal volume in liters and is used to predict successful extubation. Values < 105 have a high sensitivity for successful extubation.
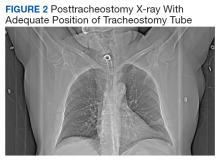
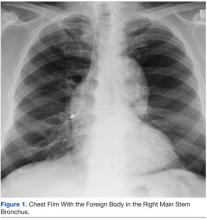
The patient remained under observation for 24 hours in the MICU and then was transferred to the general medicine ward. Unfortunately, 36 hours after, the patient had a new episode of angioedema requiring endotracheal intubation and placement on mechanical ventilation. This was his third episode of angioedema; he had a difficult airway classified as a Cormack-Lehane grade 3, requiring intubation with fiber-optic laryngoscope. In view of the recurrent events, a tracheostomy was done several days later. Figure 2 shows posttracheostomy X-ray with adequate position of the tracheostomy tube.
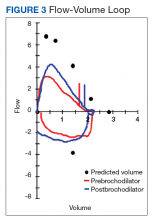
The patient was transferred to the Respiratory Care Unit and weaned off mechanical ventilation. He completed an intensive physical rehabilitation program and was discharged home. On discharge, he was followed by the Otorhinolaryngology Service and was decannulated about 5 months after. After tracheostomy decannulation, he developed asymptomatic stridor. A neck computer tomography scan revealed soft tissue thickening at the anterior and lateral aspects of the proximal tracheal likely representing granulation tissue/scarring. The findings were consistent with proximal tracheal stenosis sequelae of tracheostomy and intubation. In Figure 3, the upper portion of the curve represents the expiratory limb of the forced vital capacity and the lower portion represents inspiration. The flow-volume loop graph showed flattening of the inspiratory limb. There was a plateau in the inspiratory limb, suggestive of limitation of inspiratory flow as seen in variable extrathoracic lesions, such as glotticstricture, tumors, and vocal cord paralysis.7 The findings on the flow-volume loop were consistent with the subglottic stenosis identified by laryngoscopic examination. The patient was reluctant to undergo further interventions.
Discussion
The standard therapy for ACEI-inducedangioedema continues to be airway management and discontinuation of medication. However, life-threatening progression of symptoms have led to the use of off-label therapies, including FFP and bradykinin receptor antagonists, such as icatibant, which has been approved by the FDA for the treatment of hereditary angioedema. Icatibant is expensive and most hospitals do not have access to it. When considering the bradykinin pathway for therapy, FFP is commonly used. The cases described in the literature that have reported success with the use of FFP have used up to 2 units. There is no reported benefit of its use beyond 2 units. The initial randomized trials of icatibant for ACEI angioedema showed decreased time of resolution of angioedema.6 However, repeated trials showed conflicting results. At Veterans Affairs Caribbean Healthcare System, this medication was not available, and we decided to use FFP to improve the patient’s symptoms.
The administration of 2 units of FFP has been documented on case reports as a method to decrease the time of resolution of angioedema and the risk of recurrence. The mechanism of action thought to be involved includes the degradation of bradykinin by the enzyme ACE into inactive peptides and by supplying C1 inhibitor.8 No randomized clinical trial has investigated the use of FFP for the treatment of ACEI-induced angioedema. However, a retrospective cohort study report compared patients who presented with acute (nonhereditary) angioedema and airway compromise and received FFP with patients who were not treated with FFP.9 The study suggested a shorter ICU stay in the group treated with FFP, but the findings did not present statistical outcomes.
Nevertheless, our patient had recurrent ACEI-induced angioedema refractory to FFP. In addition to ACE or kininase II, FFP contains high-molecular weight-kininogen and kallikrein, the substrates that form bradykinin, which explained the mechanism of worsening angioedema.10 No randomized trials have investigated the use of FFP for the treatment of bradykinin-induced angioedema nor the appropriate dose.
Conclusion
In view of the emerging case reports of the effectiveness of FFP, this case of refractory angioedema raises concern for its true effectiveness and other possible factors involved in the mechanism of recurrence. Probably it would be unwise to conduct randomized studies in clinical situations such as the ones outlined. A collection of case series where FFP administration was done may be a more reasonable source of conclusions to be analyzed by a panel of experts.
1. Sánchez-Borges M, González-Aveledo LA. Angiotensin-converting enzyme inhibitors and angioedema. Allergy Asthma Immunol Res. 2010;2(3):195-198.
2. Kaplan AP. Angioedema. World Allergy Organ J. 2008;1(6):103-113.
3. Moellman JJ, Bernstein JA, Lindsell C, et al; American College of Allergy, Asthma & Immunology (ACAAI); Society for Academic Emergency Medicine (SAEM). A consensus parameter for the evaluation and management of angioedema in the emergency department. Acad Emerg Med. 2014;21(4):469-484.
4. LoVerde D, Files DC, Krishnaswamy G. Angioedema. Crit Care Med. 2017;45(4):725-735.
5. van den Elzen M, Go MFLC, Knulst AC, Blankestijn MA, van Os-Medendorp H, Otten HG. Efficacy of treatment of non-hereditary angioedema. Clinic Rev Allerg Immunol. 2018;54(3):412-431.
6. Bas M, Greve J, Stelter S, et al. A randomized trial of icatibant in ace-inhibitor–induced angioedema. N Engl J Med. 2015;372(5):418-425.
7. Diaz J, Casal J, Rodriguez W. Flow-volume loops: clinical correlation. PR Health Sci J. 2008;27(2):181-182.
8. Stewart M, McGlone R. Fresh frozen plasma in the treatment of ACE inhibitor-induced angioedema. BMJ Case Rep. 2012;2012:pii:bcr2012006849.
9. Saeb A, Hagglund KH, Cigolle CT. Using fresh frozen plasma for acute airway angioedema to prevent intubation in the emergency department: a retrospective cohort study. Emerg Med Int. 2016;2016:6091510.
10. Brown T, Gonzalez J, Monteleone C. Angiotensin-converting enzyme inhibitor-induced angioedema: a review of the literature. J Clin Hypertens (Greenwich). 2017;19(12):1377-1382.
Angioedema induced by angiotensin-converting enzyme inhibitors (ACEIs) is present in from 0.1% to 0.7% of treated patients and more often involves the head, neck, face, lips, tongue, and larynx.1 ACEI-induced angioedema results from inhibition of angiotensin-converting enzyme (ACE), which results in reduced degradation and resultant accumulation of bradykinin, a potent inflammatory mediator.2
The treatment of choice is discontinuing all ACEIs; however, the patient may be at increased risk of a subsequent angioedema attack for many weeks.3 Antihistamines (H1 and H2 receptor blockade), epinephrine, and glucocorticoids are effective in allergic/histaminergic angioedema but are usually ineffective for hereditary angioedema or ACEI angioedema and are not recommended for acute therapy.4 Kallikrein-bradykinin pathway targeted therapies are now approved by the Food and Drug Administration (FDA) for hereditary angioedema attacks and have been studied for ACEI-induced angioedema. Ecallantide and icatibant inhibit conversion of precursors to bradykinin. Multiple randomized trials of ecallantide have not shown any advantage over traditional therapies.5 On the other hand, icatibant has shown resolution of angioedema in several case reports and in a randomized trial.6 Icatibant for ACEI-induced angioedema continues to be off-label because the data are conflicting.
Case Presentation
A 67-year-old man presented with a medical history of arterial hypertension (diagnosed 17 years previously), hypercholesterolemia, type 2 diabetes mellitus, alcohol dependence, and obesity. His outpatient medications included simvastatin, aripiprazole, losartan/hydrochlorothiazide, and amlodipine. He was voluntarily admitted for inpatient detoxification. After evaluation by the internist, medication reconciliation was done, and the therapy was adjusted according to medication availability. He reported having no drug allergies, and the losartan was changed for lisinopril. About 24 hours after the first dose of lisinopril, the patient developed swelling of the lips. Antihistamine and IV steroids were administered, and the ACEI was discontinued. His baseline vital signs were temperature 98° F, heart rate 83 beats per minute, respiratory rate 19 breaths per minute, blood pressure 150/94, and oxygen saturation 98% by pulse oximeter.
During the night shift the patient’s symptoms worsened, developing difficulty swallowing and shortness of breath. He was transferred to the medicine intensive care unit (MICU), intubated, and placed on mechanical ventilation to protect his airway. Laryngoscopic examination was notable for edematous tongue, uvula, and larynx. Also, the patient had mild stridor. His laboratory test results showed normal levels of complement, tryptase, and C1 esterase. On the fourth day after admission to MICU (Figure 1), the patient extubated himself. At that time, he did not present stridor or respiratory distress and remained at the MICU for 24 hours for close monitoring.
Thirty-six hours after self-extubation the patient developed stridor and shortness of breath at the general medicine ward. In view of his clinical presentation of recurrent ACEI-induced angioedema, the Anesthesiology Service was consulted. Direct visualization of the airways showed edema of the epiglottis and vocal cords, requiring nasotracheal intubation. Two units of fresh frozen plasma (FFP) were administered. Complete resolution of angioedema took at least 72 hours even after the administration of FFP. As part of the ventilator-associated pneumonia prevention bundle, the patient continued with daily spontaneous breathing trials. On the fourth day, he was he was extubated after a cuff-leak test was positive and his rapid shallow breathing index was adequate.
The cuff-leak test is usually done to predict postextubation stridor. It consists of deflating the endotracheal tube cuff to verify if gas can pass around the tube. Absence of cuff leak is suggestive of airway edema, a risk factor for postextubation stridor and failure of extubation. For example, if the patient has an endotracheal tube that is too large in relation to the patient’s airway, the leak test can result in a false negative. In this case, fiber optic visualization of the airway can confirm the endotracheal tube occluding all the airway even with the cuff deflated and without evidence of swelling of the vocal cords. The rapid shallow breathing index is a ratio of respiratory rate over tidal volume in liters and is used to predict successful extubation. Values < 105 have a high sensitivity for successful extubation.
The patient remained under observation for 24 hours in the MICU and then was transferred to the general medicine ward. Unfortunately, 36 hours after, the patient had a new episode of angioedema requiring endotracheal intubation and placement on mechanical ventilation. This was his third episode of angioedema; he had a difficult airway classified as a Cormack-Lehane grade 3, requiring intubation with fiber-optic laryngoscope. In view of the recurrent events, a tracheostomy was done several days later. Figure 2 shows posttracheostomy X-ray with adequate position of the tracheostomy tube.
The patient was transferred to the Respiratory Care Unit and weaned off mechanical ventilation. He completed an intensive physical rehabilitation program and was discharged home. On discharge, he was followed by the Otorhinolaryngology Service and was decannulated about 5 months after. After tracheostomy decannulation, he developed asymptomatic stridor. A neck computer tomography scan revealed soft tissue thickening at the anterior and lateral aspects of the proximal tracheal likely representing granulation tissue/scarring. The findings were consistent with proximal tracheal stenosis sequelae of tracheostomy and intubation. In Figure 3, the upper portion of the curve represents the expiratory limb of the forced vital capacity and the lower portion represents inspiration. The flow-volume loop graph showed flattening of the inspiratory limb. There was a plateau in the inspiratory limb, suggestive of limitation of inspiratory flow as seen in variable extrathoracic lesions, such as glotticstricture, tumors, and vocal cord paralysis.7 The findings on the flow-volume loop were consistent with the subglottic stenosis identified by laryngoscopic examination. The patient was reluctant to undergo further interventions.
Discussion
The standard therapy for ACEI-inducedangioedema continues to be airway management and discontinuation of medication. However, life-threatening progression of symptoms have led to the use of off-label therapies, including FFP and bradykinin receptor antagonists, such as icatibant, which has been approved by the FDA for the treatment of hereditary angioedema. Icatibant is expensive and most hospitals do not have access to it. When considering the bradykinin pathway for therapy, FFP is commonly used. The cases described in the literature that have reported success with the use of FFP have used up to 2 units. There is no reported benefit of its use beyond 2 units. The initial randomized trials of icatibant for ACEI angioedema showed decreased time of resolution of angioedema.6 However, repeated trials showed conflicting results. At Veterans Affairs Caribbean Healthcare System, this medication was not available, and we decided to use FFP to improve the patient’s symptoms.
The administration of 2 units of FFP has been documented on case reports as a method to decrease the time of resolution of angioedema and the risk of recurrence. The mechanism of action thought to be involved includes the degradation of bradykinin by the enzyme ACE into inactive peptides and by supplying C1 inhibitor.8 No randomized clinical trial has investigated the use of FFP for the treatment of ACEI-induced angioedema. However, a retrospective cohort study report compared patients who presented with acute (nonhereditary) angioedema and airway compromise and received FFP with patients who were not treated with FFP.9 The study suggested a shorter ICU stay in the group treated with FFP, but the findings did not present statistical outcomes.
Nevertheless, our patient had recurrent ACEI-induced angioedema refractory to FFP. In addition to ACE or kininase II, FFP contains high-molecular weight-kininogen and kallikrein, the substrates that form bradykinin, which explained the mechanism of worsening angioedema.10 No randomized trials have investigated the use of FFP for the treatment of bradykinin-induced angioedema nor the appropriate dose.
Conclusion
In view of the emerging case reports of the effectiveness of FFP, this case of refractory angioedema raises concern for its true effectiveness and other possible factors involved in the mechanism of recurrence. Probably it would be unwise to conduct randomized studies in clinical situations such as the ones outlined. A collection of case series where FFP administration was done may be a more reasonable source of conclusions to be analyzed by a panel of experts.
Angioedema induced by angiotensin-converting enzyme inhibitors (ACEIs) is present in from 0.1% to 0.7% of treated patients and more often involves the head, neck, face, lips, tongue, and larynx.1 ACEI-induced angioedema results from inhibition of angiotensin-converting enzyme (ACE), which results in reduced degradation and resultant accumulation of bradykinin, a potent inflammatory mediator.2
The treatment of choice is discontinuing all ACEIs; however, the patient may be at increased risk of a subsequent angioedema attack for many weeks.3 Antihistamines (H1 and H2 receptor blockade), epinephrine, and glucocorticoids are effective in allergic/histaminergic angioedema but are usually ineffective for hereditary angioedema or ACEI angioedema and are not recommended for acute therapy.4 Kallikrein-bradykinin pathway targeted therapies are now approved by the Food and Drug Administration (FDA) for hereditary angioedema attacks and have been studied for ACEI-induced angioedema. Ecallantide and icatibant inhibit conversion of precursors to bradykinin. Multiple randomized trials of ecallantide have not shown any advantage over traditional therapies.5 On the other hand, icatibant has shown resolution of angioedema in several case reports and in a randomized trial.6 Icatibant for ACEI-induced angioedema continues to be off-label because the data are conflicting.
Case Presentation
A 67-year-old man presented with a medical history of arterial hypertension (diagnosed 17 years previously), hypercholesterolemia, type 2 diabetes mellitus, alcohol dependence, and obesity. His outpatient medications included simvastatin, aripiprazole, losartan/hydrochlorothiazide, and amlodipine. He was voluntarily admitted for inpatient detoxification. After evaluation by the internist, medication reconciliation was done, and the therapy was adjusted according to medication availability. He reported having no drug allergies, and the losartan was changed for lisinopril. About 24 hours after the first dose of lisinopril, the patient developed swelling of the lips. Antihistamine and IV steroids were administered, and the ACEI was discontinued. His baseline vital signs were temperature 98° F, heart rate 83 beats per minute, respiratory rate 19 breaths per minute, blood pressure 150/94, and oxygen saturation 98% by pulse oximeter.
During the night shift the patient’s symptoms worsened, developing difficulty swallowing and shortness of breath. He was transferred to the medicine intensive care unit (MICU), intubated, and placed on mechanical ventilation to protect his airway. Laryngoscopic examination was notable for edematous tongue, uvula, and larynx. Also, the patient had mild stridor. His laboratory test results showed normal levels of complement, tryptase, and C1 esterase. On the fourth day after admission to MICU (Figure 1), the patient extubated himself. At that time, he did not present stridor or respiratory distress and remained at the MICU for 24 hours for close monitoring.
Thirty-six hours after self-extubation the patient developed stridor and shortness of breath at the general medicine ward. In view of his clinical presentation of recurrent ACEI-induced angioedema, the Anesthesiology Service was consulted. Direct visualization of the airways showed edema of the epiglottis and vocal cords, requiring nasotracheal intubation. Two units of fresh frozen plasma (FFP) were administered. Complete resolution of angioedema took at least 72 hours even after the administration of FFP. As part of the ventilator-associated pneumonia prevention bundle, the patient continued with daily spontaneous breathing trials. On the fourth day, he was he was extubated after a cuff-leak test was positive and his rapid shallow breathing index was adequate.
The cuff-leak test is usually done to predict postextubation stridor. It consists of deflating the endotracheal tube cuff to verify if gas can pass around the tube. Absence of cuff leak is suggestive of airway edema, a risk factor for postextubation stridor and failure of extubation. For example, if the patient has an endotracheal tube that is too large in relation to the patient’s airway, the leak test can result in a false negative. In this case, fiber optic visualization of the airway can confirm the endotracheal tube occluding all the airway even with the cuff deflated and without evidence of swelling of the vocal cords. The rapid shallow breathing index is a ratio of respiratory rate over tidal volume in liters and is used to predict successful extubation. Values < 105 have a high sensitivity for successful extubation.
The patient remained under observation for 24 hours in the MICU and then was transferred to the general medicine ward. Unfortunately, 36 hours after, the patient had a new episode of angioedema requiring endotracheal intubation and placement on mechanical ventilation. This was his third episode of angioedema; he had a difficult airway classified as a Cormack-Lehane grade 3, requiring intubation with fiber-optic laryngoscope. In view of the recurrent events, a tracheostomy was done several days later. Figure 2 shows posttracheostomy X-ray with adequate position of the tracheostomy tube.
The patient was transferred to the Respiratory Care Unit and weaned off mechanical ventilation. He completed an intensive physical rehabilitation program and was discharged home. On discharge, he was followed by the Otorhinolaryngology Service and was decannulated about 5 months after. After tracheostomy decannulation, he developed asymptomatic stridor. A neck computer tomography scan revealed soft tissue thickening at the anterior and lateral aspects of the proximal tracheal likely representing granulation tissue/scarring. The findings were consistent with proximal tracheal stenosis sequelae of tracheostomy and intubation. In Figure 3, the upper portion of the curve represents the expiratory limb of the forced vital capacity and the lower portion represents inspiration. The flow-volume loop graph showed flattening of the inspiratory limb. There was a plateau in the inspiratory limb, suggestive of limitation of inspiratory flow as seen in variable extrathoracic lesions, such as glotticstricture, tumors, and vocal cord paralysis.7 The findings on the flow-volume loop were consistent with the subglottic stenosis identified by laryngoscopic examination. The patient was reluctant to undergo further interventions.
Discussion
The standard therapy for ACEI-inducedangioedema continues to be airway management and discontinuation of medication. However, life-threatening progression of symptoms have led to the use of off-label therapies, including FFP and bradykinin receptor antagonists, such as icatibant, which has been approved by the FDA for the treatment of hereditary angioedema. Icatibant is expensive and most hospitals do not have access to it. When considering the bradykinin pathway for therapy, FFP is commonly used. The cases described in the literature that have reported success with the use of FFP have used up to 2 units. There is no reported benefit of its use beyond 2 units. The initial randomized trials of icatibant for ACEI angioedema showed decreased time of resolution of angioedema.6 However, repeated trials showed conflicting results. At Veterans Affairs Caribbean Healthcare System, this medication was not available, and we decided to use FFP to improve the patient’s symptoms.
The administration of 2 units of FFP has been documented on case reports as a method to decrease the time of resolution of angioedema and the risk of recurrence. The mechanism of action thought to be involved includes the degradation of bradykinin by the enzyme ACE into inactive peptides and by supplying C1 inhibitor.8 No randomized clinical trial has investigated the use of FFP for the treatment of ACEI-induced angioedema. However, a retrospective cohort study report compared patients who presented with acute (nonhereditary) angioedema and airway compromise and received FFP with patients who were not treated with FFP.9 The study suggested a shorter ICU stay in the group treated with FFP, but the findings did not present statistical outcomes.
Nevertheless, our patient had recurrent ACEI-induced angioedema refractory to FFP. In addition to ACE or kininase II, FFP contains high-molecular weight-kininogen and kallikrein, the substrates that form bradykinin, which explained the mechanism of worsening angioedema.10 No randomized trials have investigated the use of FFP for the treatment of bradykinin-induced angioedema nor the appropriate dose.
Conclusion
In view of the emerging case reports of the effectiveness of FFP, this case of refractory angioedema raises concern for its true effectiveness and other possible factors involved in the mechanism of recurrence. Probably it would be unwise to conduct randomized studies in clinical situations such as the ones outlined. A collection of case series where FFP administration was done may be a more reasonable source of conclusions to be analyzed by a panel of experts.
1. Sánchez-Borges M, González-Aveledo LA. Angiotensin-converting enzyme inhibitors and angioedema. Allergy Asthma Immunol Res. 2010;2(3):195-198.
2. Kaplan AP. Angioedema. World Allergy Organ J. 2008;1(6):103-113.
3. Moellman JJ, Bernstein JA, Lindsell C, et al; American College of Allergy, Asthma & Immunology (ACAAI); Society for Academic Emergency Medicine (SAEM). A consensus parameter for the evaluation and management of angioedema in the emergency department. Acad Emerg Med. 2014;21(4):469-484.
4. LoVerde D, Files DC, Krishnaswamy G. Angioedema. Crit Care Med. 2017;45(4):725-735.
5. van den Elzen M, Go MFLC, Knulst AC, Blankestijn MA, van Os-Medendorp H, Otten HG. Efficacy of treatment of non-hereditary angioedema. Clinic Rev Allerg Immunol. 2018;54(3):412-431.
6. Bas M, Greve J, Stelter S, et al. A randomized trial of icatibant in ace-inhibitor–induced angioedema. N Engl J Med. 2015;372(5):418-425.
7. Diaz J, Casal J, Rodriguez W. Flow-volume loops: clinical correlation. PR Health Sci J. 2008;27(2):181-182.
8. Stewart M, McGlone R. Fresh frozen plasma in the treatment of ACE inhibitor-induced angioedema. BMJ Case Rep. 2012;2012:pii:bcr2012006849.
9. Saeb A, Hagglund KH, Cigolle CT. Using fresh frozen plasma for acute airway angioedema to prevent intubation in the emergency department: a retrospective cohort study. Emerg Med Int. 2016;2016:6091510.
10. Brown T, Gonzalez J, Monteleone C. Angiotensin-converting enzyme inhibitor-induced angioedema: a review of the literature. J Clin Hypertens (Greenwich). 2017;19(12):1377-1382.
1. Sánchez-Borges M, González-Aveledo LA. Angiotensin-converting enzyme inhibitors and angioedema. Allergy Asthma Immunol Res. 2010;2(3):195-198.
2. Kaplan AP. Angioedema. World Allergy Organ J. 2008;1(6):103-113.
3. Moellman JJ, Bernstein JA, Lindsell C, et al; American College of Allergy, Asthma & Immunology (ACAAI); Society for Academic Emergency Medicine (SAEM). A consensus parameter for the evaluation and management of angioedema in the emergency department. Acad Emerg Med. 2014;21(4):469-484.
4. LoVerde D, Files DC, Krishnaswamy G. Angioedema. Crit Care Med. 2017;45(4):725-735.
5. van den Elzen M, Go MFLC, Knulst AC, Blankestijn MA, van Os-Medendorp H, Otten HG. Efficacy of treatment of non-hereditary angioedema. Clinic Rev Allerg Immunol. 2018;54(3):412-431.
6. Bas M, Greve J, Stelter S, et al. A randomized trial of icatibant in ace-inhibitor–induced angioedema. N Engl J Med. 2015;372(5):418-425.
7. Diaz J, Casal J, Rodriguez W. Flow-volume loops: clinical correlation. PR Health Sci J. 2008;27(2):181-182.
8. Stewart M, McGlone R. Fresh frozen plasma in the treatment of ACE inhibitor-induced angioedema. BMJ Case Rep. 2012;2012:pii:bcr2012006849.
9. Saeb A, Hagglund KH, Cigolle CT. Using fresh frozen plasma for acute airway angioedema to prevent intubation in the emergency department: a retrospective cohort study. Emerg Med Int. 2016;2016:6091510.
10. Brown T, Gonzalez J, Monteleone C. Angiotensin-converting enzyme inhibitor-induced angioedema: a review of the literature. J Clin Hypertens (Greenwich). 2017;19(12):1377-1382.
Aspiration of a Dental Tool During a Crown Placement Procedure
There are many reports in the medical and dental literature of complications arising from a routine delivery of dental care. One complication can include physical injury from swallowing or aspirating foreign objects.1 However, a review of such literature presents a scarcity of documented instances and no long-term evaluation of the aforementioned events.2,3
This report presents the case of a patient who aspirated a hex driver tool during a procedure to place a crown on a dental implant. The aspirated object was subsequently removed through flexible fiberoptic bronchoscopy without complications.
Case Report
An 83-year-old man was referred to the Pulmonary and Critical Care Department of the VA Caribbean Healthcare System in San Juan, Puerto Rico, after a hex driver tool was lost during a procedure to place a crown on a dental implant, performed under topical anesthesia. It was first thought that the patient swallowed the hex driver, since he never experienced or complained of coughing or shortness of breath. A chest radiograph revealed a metal object lying within the right main stem bronchus, for which the patient was referred to the Pulmonary and Critical Care Department (Figure 1).
The patient’s past medical history was remarkable for hypertension and hypercholesterolemia. Outpatient medications included hydrochlorothiazide, simvastatin, aspirin, felodipine, and lorazepam. He had no previous history of dysphagia or neurologic disease. A physical examination revealed expiratory and inspiratory wheezing localized to the right lower lobe without associated rhonchi or crackles. No distress, shortness of breath, or coughing was noted.
A flexible fiberoptic bronchoscopy was performed under conscious sedation with 3 mg of IV midazolam and topical anesthesia with nebulized 4% lidocaine. No mucosal edema, hyperemia, or structural damage was noted during direct visualization of both the right and left bronchopulmonary segments. A metallic object was visualized at the entrance of the right lower lobe. The foreign object had irregular borders, providing multiple edges that made it suitable to be embraced (Figure 2).
Using a radial jaw single-use biopsy forceps 1.8 mm, the physician clinched and retrieved the object through the bronchoscope. The object was retrieved on the same day of the dental procedure almost 5 hours after it was aspirated. The patient tolerated the procedure well; no coughing, oxygen desaturation, or bleeding occurred during the procedure.
After a few hours of observation, a postprocedural radiograph confirmed the removal of the foreign body without evidence of pneumothorax. The patient was discharged, and 24 hours after the incident remained asymptomatic without chest pain, cough, hemoptysis, sputum production, or fever.
Discussion
Foreign-body aspiration and inadvertent swallowing remains underrecognized by clinicians. In the U.S., more than 2,700 people, including more than 300 children, die of foreign-body aspiration each year.4,5 Aspiration or ingestion of a foreign body during a dental procedure is serious and potentially fatal.6 Some of the consequences of an aspirated object are complete or partial airway obstruction, respiratory distress and failure, pneumothorax, and hemorrhage.7 In addition, inadvertent aspiration of foreign objects in asymptomatic patients may not be evident for months, resulting in late complications as postobstructive pneumonia, bronchiectasis, or lung abscess.8 Early recognition and diagnosis of these events are crucial to prevent complications.
Accidental aspiration of foreign objects during dental procedures is not as common as is swallowing. In the normal population, the foreign object enters the gastrointestinal tract in about 92.5% of the time, and the tracheobronchial tree in 7.5% of these instances.
A 10-year review done at the School of Dentistry of the University of North Carolina reported 36 incidents of lost instruments during dental procedures. In only 1 case, an object was aspirated, 25 of the 36 cases were secondary to ingestion, and in the remaining 10 incidents, swallowing or aspiration was ruled out by radiography or after the object’s removal from the patient’s mouth.2 Previous reviews about foreign-body aspiration in adults have reported dental appliances as the second most commonly aspirated foreign objects.4 Of all aspirated objects, the most common site of impaction is the right lower lobe; however, aspiration has been reported in all pulmonary lobes.6
Available literature recognizes that impaction of aspirated objects occurred in 56% of instances within the right lower lobe and 33% in the left lower lobe.7,9 Identification of risk factors for aspiration is important for any patient who will undergo dental procedures, such as advanced age (ie, elderly patients may have a decreased gag reflex); neurologic conditions, such as stroke; dementia and other degenerative diseases; the use of topical anesthesia; and altered states of consciousness associated with the use of IV sedation.1,2
The key sign that most dentists recognize when patients aspirate an object during a dental procedure is coughing. It has been reported that coughing resulting from aspiration of foreign objects may range from mild to severe. In this case, the patient was completely asymptomatic during the procedure. The only clue of possible object aspiration was the reported tool loss by the dentist. It is important to always examine, account for, and review all equipment used during dental procedures. Assessment for any lost objects or missing parts of instruments should be done promptly with a high degree of suspicion for possible swallowing or aspiration if an object is missing.
It has been recommended to use a gauze throat screen and rubber dam and to avoid a supine position during a procedure, among other techniques, to minimize risk of ingestion or aspiration.2 Imaging studies should be used for further evaluation of the patient; however, some instruments, such as dental pieces and impression material, may not be identified by plain films. In those cases, further evaluation with more sophisticated imaging techniques, such as computed tomography (CT), should be considered.1-10
In a previous case report of a patient who aspirated a third molar during a dental procedure, a chest film failed to identify it. A chest CT was performed, and the object showed in the right main stem bronchus. In another instance, aspiration of impression material in a 45-year-old man was not observed by chest radiography. In this case, the history of coughing and respiratory symptoms days after the procedure pointed toward aspiration of an object as the culprit, with subsequent identification and removal by flexible fiberoptic bronchoscopy.1-11
Bronchoscopy is the treatment of choice for extraction of aspirated foreign bodies; however, there is still a debate about whether to use flexible or rigid bronchoscopy. The decision is usually made based on the object size, localization, medical facility, and personnel expertise. The rigid bronchoscope has the advantages of offering better control and visualization of the airway and easier use of removal instruments. Its primary disadvantage is that the procedure needs to be done in the operating room under general anesthesia. Flexible fiberoptic bronchoscopy done under conscious sedation and topical anesthesia may be as effective as rigid bronchoscopy and even superior in the case of smaller and more distal impacted objects.10-14
In this case, flexible fiberoptic bronchoscopy was used successfully for the removal of the foreign object. Biopsy forceps were used to grasp the object and retrieve it from the airway without complication.
Conclusion
Aspiration of foreign objects during a dental procedure is a potential life-threatening complication. A high-level of suspicion is needed for early diagnosis and referral of the patient for extraction of the object and further avoidance of complications. Flexible fiberoptic bronchoscopy is a feasible procedure for removal of objects within the airway.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc. , the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Cameron SM, Whitlock WL, Tabor MS. Foreign body aspiration in dentistry: A review. J Am Dent Assoc. 1996;127(8):1224-1229.
2. Tiwana KK, Morton T, Tiwana PS. Aspiration and ingestion in dental practice: A 10-year institutional review. J Am Dent Assoc. 2004;135(9):1287-1291.
3. Susini G, Pommel L, Camps J. Accidental ingestion and aspiration of root canal instruments and other dental foreign bodies in a French population. Int Endod J. 2007;40(8):585-589.
4. Fields RT Jr, Schow SR. Aspiration and ingestion of foreign bodies in oral and maxillofacial surgery: A review of the literature and report of five cases. J Oral Maxillofac Surg. 1998;56(9):1091-1098.
5. Black RE, Johnson DG, Matlak ME. Bronchoscopic removal of aspirated foreign bodies in children. J Pediatr Surg. 1994;29(5):682-684.
6. Limper AH, Prakash UBS. Tracheobronchial foreign bodies in adults. Ann Intern Med. 1990;112(8):604-609.
7. Bas¸oglu OK, Buduneli N, Cagirici U, Turhan K, Aysan T. Pulmonary aspiration of a two-unit bridge during a deep sleep. J Oral Rehabil. 2005;32(6):461-463.
8. Mahmoud M, Imam S, Patel H, King M. Foreign body aspiration of a dental bridge in the left main stem bronchus. Case Rep Med. 2012;2012:1-4.
9. Jackson C, Jackson CL. Diseases of the Air and Food Passages of Foreign-Body Origin. Philadelphia, PA: Saunders; 1936.
10. Zitzmann NU, Elsasser S, Fried R, Marinello CP. Foreign body ingestion and aspiration. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;88(6):657-660.
11. Elgazzar RF, Abdelhady AI, Sadakah AA. Aspiration of an impacted lower third molar during its surgical removal under local anaesthesia. Int J Oral Maxillofac Surg. 2007;36(4):362-364.
12. Tu CY, Chen HJ, Chen W, Liu YH, Chen CH. A feasible approach for extraction of dental prostheses from the airway by flexible bronchoscopy in concert with wire loops snares. Laryngoscope. 2007;117(7):1280-1282.
13. Ospina JC, Ludemann JP. Aspiration of an extracted molar: Case report. J Can Dent Assoc. 2005;71(8):581-583.
14. Cohen S, Pine H, Drake A. Use of rigid and flexible bronchoscopy among pediatric otolaryngologists. Arch Otoralyngol Head Neck Surg. 2001;127(5):505-509.
There are many reports in the medical and dental literature of complications arising from a routine delivery of dental care. One complication can include physical injury from swallowing or aspirating foreign objects.1 However, a review of such literature presents a scarcity of documented instances and no long-term evaluation of the aforementioned events.2,3
This report presents the case of a patient who aspirated a hex driver tool during a procedure to place a crown on a dental implant. The aspirated object was subsequently removed through flexible fiberoptic bronchoscopy without complications.
Case Report
An 83-year-old man was referred to the Pulmonary and Critical Care Department of the VA Caribbean Healthcare System in San Juan, Puerto Rico, after a hex driver tool was lost during a procedure to place a crown on a dental implant, performed under topical anesthesia. It was first thought that the patient swallowed the hex driver, since he never experienced or complained of coughing or shortness of breath. A chest radiograph revealed a metal object lying within the right main stem bronchus, for which the patient was referred to the Pulmonary and Critical Care Department (Figure 1).
The patient’s past medical history was remarkable for hypertension and hypercholesterolemia. Outpatient medications included hydrochlorothiazide, simvastatin, aspirin, felodipine, and lorazepam. He had no previous history of dysphagia or neurologic disease. A physical examination revealed expiratory and inspiratory wheezing localized to the right lower lobe without associated rhonchi or crackles. No distress, shortness of breath, or coughing was noted.
A flexible fiberoptic bronchoscopy was performed under conscious sedation with 3 mg of IV midazolam and topical anesthesia with nebulized 4% lidocaine. No mucosal edema, hyperemia, or structural damage was noted during direct visualization of both the right and left bronchopulmonary segments. A metallic object was visualized at the entrance of the right lower lobe. The foreign object had irregular borders, providing multiple edges that made it suitable to be embraced (Figure 2).
Using a radial jaw single-use biopsy forceps 1.8 mm, the physician clinched and retrieved the object through the bronchoscope. The object was retrieved on the same day of the dental procedure almost 5 hours after it was aspirated. The patient tolerated the procedure well; no coughing, oxygen desaturation, or bleeding occurred during the procedure.
After a few hours of observation, a postprocedural radiograph confirmed the removal of the foreign body without evidence of pneumothorax. The patient was discharged, and 24 hours after the incident remained asymptomatic without chest pain, cough, hemoptysis, sputum production, or fever.
Discussion
Foreign-body aspiration and inadvertent swallowing remains underrecognized by clinicians. In the U.S., more than 2,700 people, including more than 300 children, die of foreign-body aspiration each year.4,5 Aspiration or ingestion of a foreign body during a dental procedure is serious and potentially fatal.6 Some of the consequences of an aspirated object are complete or partial airway obstruction, respiratory distress and failure, pneumothorax, and hemorrhage.7 In addition, inadvertent aspiration of foreign objects in asymptomatic patients may not be evident for months, resulting in late complications as postobstructive pneumonia, bronchiectasis, or lung abscess.8 Early recognition and diagnosis of these events are crucial to prevent complications.
Accidental aspiration of foreign objects during dental procedures is not as common as is swallowing. In the normal population, the foreign object enters the gastrointestinal tract in about 92.5% of the time, and the tracheobronchial tree in 7.5% of these instances.
A 10-year review done at the School of Dentistry of the University of North Carolina reported 36 incidents of lost instruments during dental procedures. In only 1 case, an object was aspirated, 25 of the 36 cases were secondary to ingestion, and in the remaining 10 incidents, swallowing or aspiration was ruled out by radiography or after the object’s removal from the patient’s mouth.2 Previous reviews about foreign-body aspiration in adults have reported dental appliances as the second most commonly aspirated foreign objects.4 Of all aspirated objects, the most common site of impaction is the right lower lobe; however, aspiration has been reported in all pulmonary lobes.6
Available literature recognizes that impaction of aspirated objects occurred in 56% of instances within the right lower lobe and 33% in the left lower lobe.7,9 Identification of risk factors for aspiration is important for any patient who will undergo dental procedures, such as advanced age (ie, elderly patients may have a decreased gag reflex); neurologic conditions, such as stroke; dementia and other degenerative diseases; the use of topical anesthesia; and altered states of consciousness associated with the use of IV sedation.1,2
The key sign that most dentists recognize when patients aspirate an object during a dental procedure is coughing. It has been reported that coughing resulting from aspiration of foreign objects may range from mild to severe. In this case, the patient was completely asymptomatic during the procedure. The only clue of possible object aspiration was the reported tool loss by the dentist. It is important to always examine, account for, and review all equipment used during dental procedures. Assessment for any lost objects or missing parts of instruments should be done promptly with a high degree of suspicion for possible swallowing or aspiration if an object is missing.
It has been recommended to use a gauze throat screen and rubber dam and to avoid a supine position during a procedure, among other techniques, to minimize risk of ingestion or aspiration.2 Imaging studies should be used for further evaluation of the patient; however, some instruments, such as dental pieces and impression material, may not be identified by plain films. In those cases, further evaluation with more sophisticated imaging techniques, such as computed tomography (CT), should be considered.1-10
In a previous case report of a patient who aspirated a third molar during a dental procedure, a chest film failed to identify it. A chest CT was performed, and the object showed in the right main stem bronchus. In another instance, aspiration of impression material in a 45-year-old man was not observed by chest radiography. In this case, the history of coughing and respiratory symptoms days after the procedure pointed toward aspiration of an object as the culprit, with subsequent identification and removal by flexible fiberoptic bronchoscopy.1-11
Bronchoscopy is the treatment of choice for extraction of aspirated foreign bodies; however, there is still a debate about whether to use flexible or rigid bronchoscopy. The decision is usually made based on the object size, localization, medical facility, and personnel expertise. The rigid bronchoscope has the advantages of offering better control and visualization of the airway and easier use of removal instruments. Its primary disadvantage is that the procedure needs to be done in the operating room under general anesthesia. Flexible fiberoptic bronchoscopy done under conscious sedation and topical anesthesia may be as effective as rigid bronchoscopy and even superior in the case of smaller and more distal impacted objects.10-14
In this case, flexible fiberoptic bronchoscopy was used successfully for the removal of the foreign object. Biopsy forceps were used to grasp the object and retrieve it from the airway without complication.
Conclusion
Aspiration of foreign objects during a dental procedure is a potential life-threatening complication. A high-level of suspicion is needed for early diagnosis and referral of the patient for extraction of the object and further avoidance of complications. Flexible fiberoptic bronchoscopy is a feasible procedure for removal of objects within the airway.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc. , the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
There are many reports in the medical and dental literature of complications arising from a routine delivery of dental care. One complication can include physical injury from swallowing or aspirating foreign objects.1 However, a review of such literature presents a scarcity of documented instances and no long-term evaluation of the aforementioned events.2,3
This report presents the case of a patient who aspirated a hex driver tool during a procedure to place a crown on a dental implant. The aspirated object was subsequently removed through flexible fiberoptic bronchoscopy without complications.
Case Report
An 83-year-old man was referred to the Pulmonary and Critical Care Department of the VA Caribbean Healthcare System in San Juan, Puerto Rico, after a hex driver tool was lost during a procedure to place a crown on a dental implant, performed under topical anesthesia. It was first thought that the patient swallowed the hex driver, since he never experienced or complained of coughing or shortness of breath. A chest radiograph revealed a metal object lying within the right main stem bronchus, for which the patient was referred to the Pulmonary and Critical Care Department (Figure 1).
The patient’s past medical history was remarkable for hypertension and hypercholesterolemia. Outpatient medications included hydrochlorothiazide, simvastatin, aspirin, felodipine, and lorazepam. He had no previous history of dysphagia or neurologic disease. A physical examination revealed expiratory and inspiratory wheezing localized to the right lower lobe without associated rhonchi or crackles. No distress, shortness of breath, or coughing was noted.
A flexible fiberoptic bronchoscopy was performed under conscious sedation with 3 mg of IV midazolam and topical anesthesia with nebulized 4% lidocaine. No mucosal edema, hyperemia, or structural damage was noted during direct visualization of both the right and left bronchopulmonary segments. A metallic object was visualized at the entrance of the right lower lobe. The foreign object had irregular borders, providing multiple edges that made it suitable to be embraced (Figure 2).
Using a radial jaw single-use biopsy forceps 1.8 mm, the physician clinched and retrieved the object through the bronchoscope. The object was retrieved on the same day of the dental procedure almost 5 hours after it was aspirated. The patient tolerated the procedure well; no coughing, oxygen desaturation, or bleeding occurred during the procedure.
After a few hours of observation, a postprocedural radiograph confirmed the removal of the foreign body without evidence of pneumothorax. The patient was discharged, and 24 hours after the incident remained asymptomatic without chest pain, cough, hemoptysis, sputum production, or fever.
Discussion
Foreign-body aspiration and inadvertent swallowing remains underrecognized by clinicians. In the U.S., more than 2,700 people, including more than 300 children, die of foreign-body aspiration each year.4,5 Aspiration or ingestion of a foreign body during a dental procedure is serious and potentially fatal.6 Some of the consequences of an aspirated object are complete or partial airway obstruction, respiratory distress and failure, pneumothorax, and hemorrhage.7 In addition, inadvertent aspiration of foreign objects in asymptomatic patients may not be evident for months, resulting in late complications as postobstructive pneumonia, bronchiectasis, or lung abscess.8 Early recognition and diagnosis of these events are crucial to prevent complications.
Accidental aspiration of foreign objects during dental procedures is not as common as is swallowing. In the normal population, the foreign object enters the gastrointestinal tract in about 92.5% of the time, and the tracheobronchial tree in 7.5% of these instances.
A 10-year review done at the School of Dentistry of the University of North Carolina reported 36 incidents of lost instruments during dental procedures. In only 1 case, an object was aspirated, 25 of the 36 cases were secondary to ingestion, and in the remaining 10 incidents, swallowing or aspiration was ruled out by radiography or after the object’s removal from the patient’s mouth.2 Previous reviews about foreign-body aspiration in adults have reported dental appliances as the second most commonly aspirated foreign objects.4 Of all aspirated objects, the most common site of impaction is the right lower lobe; however, aspiration has been reported in all pulmonary lobes.6
Available literature recognizes that impaction of aspirated objects occurred in 56% of instances within the right lower lobe and 33% in the left lower lobe.7,9 Identification of risk factors for aspiration is important for any patient who will undergo dental procedures, such as advanced age (ie, elderly patients may have a decreased gag reflex); neurologic conditions, such as stroke; dementia and other degenerative diseases; the use of topical anesthesia; and altered states of consciousness associated with the use of IV sedation.1,2
The key sign that most dentists recognize when patients aspirate an object during a dental procedure is coughing. It has been reported that coughing resulting from aspiration of foreign objects may range from mild to severe. In this case, the patient was completely asymptomatic during the procedure. The only clue of possible object aspiration was the reported tool loss by the dentist. It is important to always examine, account for, and review all equipment used during dental procedures. Assessment for any lost objects or missing parts of instruments should be done promptly with a high degree of suspicion for possible swallowing or aspiration if an object is missing.
It has been recommended to use a gauze throat screen and rubber dam and to avoid a supine position during a procedure, among other techniques, to minimize risk of ingestion or aspiration.2 Imaging studies should be used for further evaluation of the patient; however, some instruments, such as dental pieces and impression material, may not be identified by plain films. In those cases, further evaluation with more sophisticated imaging techniques, such as computed tomography (CT), should be considered.1-10
In a previous case report of a patient who aspirated a third molar during a dental procedure, a chest film failed to identify it. A chest CT was performed, and the object showed in the right main stem bronchus. In another instance, aspiration of impression material in a 45-year-old man was not observed by chest radiography. In this case, the history of coughing and respiratory symptoms days after the procedure pointed toward aspiration of an object as the culprit, with subsequent identification and removal by flexible fiberoptic bronchoscopy.1-11
Bronchoscopy is the treatment of choice for extraction of aspirated foreign bodies; however, there is still a debate about whether to use flexible or rigid bronchoscopy. The decision is usually made based on the object size, localization, medical facility, and personnel expertise. The rigid bronchoscope has the advantages of offering better control and visualization of the airway and easier use of removal instruments. Its primary disadvantage is that the procedure needs to be done in the operating room under general anesthesia. Flexible fiberoptic bronchoscopy done under conscious sedation and topical anesthesia may be as effective as rigid bronchoscopy and even superior in the case of smaller and more distal impacted objects.10-14
In this case, flexible fiberoptic bronchoscopy was used successfully for the removal of the foreign object. Biopsy forceps were used to grasp the object and retrieve it from the airway without complication.
Conclusion
Aspiration of foreign objects during a dental procedure is a potential life-threatening complication. A high-level of suspicion is needed for early diagnosis and referral of the patient for extraction of the object and further avoidance of complications. Flexible fiberoptic bronchoscopy is a feasible procedure for removal of objects within the airway.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc. , the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Cameron SM, Whitlock WL, Tabor MS. Foreign body aspiration in dentistry: A review. J Am Dent Assoc. 1996;127(8):1224-1229.
2. Tiwana KK, Morton T, Tiwana PS. Aspiration and ingestion in dental practice: A 10-year institutional review. J Am Dent Assoc. 2004;135(9):1287-1291.
3. Susini G, Pommel L, Camps J. Accidental ingestion and aspiration of root canal instruments and other dental foreign bodies in a French population. Int Endod J. 2007;40(8):585-589.
4. Fields RT Jr, Schow SR. Aspiration and ingestion of foreign bodies in oral and maxillofacial surgery: A review of the literature and report of five cases. J Oral Maxillofac Surg. 1998;56(9):1091-1098.
5. Black RE, Johnson DG, Matlak ME. Bronchoscopic removal of aspirated foreign bodies in children. J Pediatr Surg. 1994;29(5):682-684.
6. Limper AH, Prakash UBS. Tracheobronchial foreign bodies in adults. Ann Intern Med. 1990;112(8):604-609.
7. Bas¸oglu OK, Buduneli N, Cagirici U, Turhan K, Aysan T. Pulmonary aspiration of a two-unit bridge during a deep sleep. J Oral Rehabil. 2005;32(6):461-463.
8. Mahmoud M, Imam S, Patel H, King M. Foreign body aspiration of a dental bridge in the left main stem bronchus. Case Rep Med. 2012;2012:1-4.
9. Jackson C, Jackson CL. Diseases of the Air and Food Passages of Foreign-Body Origin. Philadelphia, PA: Saunders; 1936.
10. Zitzmann NU, Elsasser S, Fried R, Marinello CP. Foreign body ingestion and aspiration. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;88(6):657-660.
11. Elgazzar RF, Abdelhady AI, Sadakah AA. Aspiration of an impacted lower third molar during its surgical removal under local anaesthesia. Int J Oral Maxillofac Surg. 2007;36(4):362-364.
12. Tu CY, Chen HJ, Chen W, Liu YH, Chen CH. A feasible approach for extraction of dental prostheses from the airway by flexible bronchoscopy in concert with wire loops snares. Laryngoscope. 2007;117(7):1280-1282.
13. Ospina JC, Ludemann JP. Aspiration of an extracted molar: Case report. J Can Dent Assoc. 2005;71(8):581-583.
14. Cohen S, Pine H, Drake A. Use of rigid and flexible bronchoscopy among pediatric otolaryngologists. Arch Otoralyngol Head Neck Surg. 2001;127(5):505-509.
1. Cameron SM, Whitlock WL, Tabor MS. Foreign body aspiration in dentistry: A review. J Am Dent Assoc. 1996;127(8):1224-1229.
2. Tiwana KK, Morton T, Tiwana PS. Aspiration and ingestion in dental practice: A 10-year institutional review. J Am Dent Assoc. 2004;135(9):1287-1291.
3. Susini G, Pommel L, Camps J. Accidental ingestion and aspiration of root canal instruments and other dental foreign bodies in a French population. Int Endod J. 2007;40(8):585-589.
4. Fields RT Jr, Schow SR. Aspiration and ingestion of foreign bodies in oral and maxillofacial surgery: A review of the literature and report of five cases. J Oral Maxillofac Surg. 1998;56(9):1091-1098.
5. Black RE, Johnson DG, Matlak ME. Bronchoscopic removal of aspirated foreign bodies in children. J Pediatr Surg. 1994;29(5):682-684.
6. Limper AH, Prakash UBS. Tracheobronchial foreign bodies in adults. Ann Intern Med. 1990;112(8):604-609.
7. Bas¸oglu OK, Buduneli N, Cagirici U, Turhan K, Aysan T. Pulmonary aspiration of a two-unit bridge during a deep sleep. J Oral Rehabil. 2005;32(6):461-463.
8. Mahmoud M, Imam S, Patel H, King M. Foreign body aspiration of a dental bridge in the left main stem bronchus. Case Rep Med. 2012;2012:1-4.
9. Jackson C, Jackson CL. Diseases of the Air and Food Passages of Foreign-Body Origin. Philadelphia, PA: Saunders; 1936.
10. Zitzmann NU, Elsasser S, Fried R, Marinello CP. Foreign body ingestion and aspiration. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;88(6):657-660.
11. Elgazzar RF, Abdelhady AI, Sadakah AA. Aspiration of an impacted lower third molar during its surgical removal under local anaesthesia. Int J Oral Maxillofac Surg. 2007;36(4):362-364.
12. Tu CY, Chen HJ, Chen W, Liu YH, Chen CH. A feasible approach for extraction of dental prostheses from the airway by flexible bronchoscopy in concert with wire loops snares. Laryngoscope. 2007;117(7):1280-1282.
13. Ospina JC, Ludemann JP. Aspiration of an extracted molar: Case report. J Can Dent Assoc. 2005;71(8):581-583.
14. Cohen S, Pine H, Drake A. Use of rigid and flexible bronchoscopy among pediatric otolaryngologists. Arch Otoralyngol Head Neck Surg. 2001;127(5):505-509.