User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Tralokinumab earns EU recommendation to expand age range for atopic dermatitis to include adolescents
Tralokinumab has received a positive opinion from the European Medicine Agency’s Committee for Medicinal Products for Human Use to extend use to adolescents aged 12 years and older with moderate-to-severe atopic dermatitis (AD) who are candidates for systemic therapy, according to a statement from the manufacturer.
The positive CHMP opinion, issued on Sept. 15, recommends extending the use of tralokinumab (Adtralza), an interleukin-13 antagonist, to adolescents aged 12-17 years in the EU. The positive opinion recommends an initial dose of 600 mg administered subcutaneously followed by 300 mg every other week, the dosing recommended for adults.
In December 2021, tralokinumab was approved for adults with moderate to severe AD in the United States, where it is marketed as Adbry. It is also approved for adults in the EU, Great Britain, Canada, the United Arab Emirates, and Switzerland. It is not currently approved for treatment of adolescents in any country, according to the LEO Pharma statement.
A regulatory filing with the U.S. Food and Drug Administration is in progress, the company said, and an additional study of tralokinumab for individuals aged 12 years and older is underway, according to the manufacturer.
The CHMP opinion was supported by data from a phase 3 study (ECZTRA 6) that assessed safety and efficacy of 150-mg or 300-mg doses of tralokinumab, compared with placebo in adolescents with moderate-to-severe AD, the company statement said. The primary outcomes were an Investigator Global Assessment score of clear or almost clear skin (IGA 0/1) and an improvement of at least a 75% on the Eczema Area and Severity Index score (EASI-75). In the study, presented as a poster at a meeting in October 2021, a total of 195 adolescents aged 12-17 with moderate to severe AD who were candidates for systemic therapy were randomly assigned to tralokinumab and 94 to placebo.
At 16 weeks, 21.4% and 17.5% of patients who received 150 mg and 300 mg, respectively, of tralokinumab had IGA scores of 0 or 1, compared with 4.3% of those on placebo (P < .001, P = .002, respectively vs. placebo). In addition, 28.6% and 27.8% of the 150-mg and 300-mg tralokinumab groups, respectively, achieved EASI-75, compared with 6.4% of placebo patients (P < .001, P = .001, respectively, compared with placebo).
Adverse events were similar between the groups, and most were mild or moderate; overall safety profiles were similar to those seen in adult patients.
The European Commission will review the positive opinion and make a final decision.
The research was supported by LEO Pharma.
A version of this article first appeared on Medscape.com.
Tralokinumab has received a positive opinion from the European Medicine Agency’s Committee for Medicinal Products for Human Use to extend use to adolescents aged 12 years and older with moderate-to-severe atopic dermatitis (AD) who are candidates for systemic therapy, according to a statement from the manufacturer.
The positive CHMP opinion, issued on Sept. 15, recommends extending the use of tralokinumab (Adtralza), an interleukin-13 antagonist, to adolescents aged 12-17 years in the EU. The positive opinion recommends an initial dose of 600 mg administered subcutaneously followed by 300 mg every other week, the dosing recommended for adults.
In December 2021, tralokinumab was approved for adults with moderate to severe AD in the United States, where it is marketed as Adbry. It is also approved for adults in the EU, Great Britain, Canada, the United Arab Emirates, and Switzerland. It is not currently approved for treatment of adolescents in any country, according to the LEO Pharma statement.
A regulatory filing with the U.S. Food and Drug Administration is in progress, the company said, and an additional study of tralokinumab for individuals aged 12 years and older is underway, according to the manufacturer.
The CHMP opinion was supported by data from a phase 3 study (ECZTRA 6) that assessed safety and efficacy of 150-mg or 300-mg doses of tralokinumab, compared with placebo in adolescents with moderate-to-severe AD, the company statement said. The primary outcomes were an Investigator Global Assessment score of clear or almost clear skin (IGA 0/1) and an improvement of at least a 75% on the Eczema Area and Severity Index score (EASI-75). In the study, presented as a poster at a meeting in October 2021, a total of 195 adolescents aged 12-17 with moderate to severe AD who were candidates for systemic therapy were randomly assigned to tralokinumab and 94 to placebo.
At 16 weeks, 21.4% and 17.5% of patients who received 150 mg and 300 mg, respectively, of tralokinumab had IGA scores of 0 or 1, compared with 4.3% of those on placebo (P < .001, P = .002, respectively vs. placebo). In addition, 28.6% and 27.8% of the 150-mg and 300-mg tralokinumab groups, respectively, achieved EASI-75, compared with 6.4% of placebo patients (P < .001, P = .001, respectively, compared with placebo).
Adverse events were similar between the groups, and most were mild or moderate; overall safety profiles were similar to those seen in adult patients.
The European Commission will review the positive opinion and make a final decision.
The research was supported by LEO Pharma.
A version of this article first appeared on Medscape.com.
Tralokinumab has received a positive opinion from the European Medicine Agency’s Committee for Medicinal Products for Human Use to extend use to adolescents aged 12 years and older with moderate-to-severe atopic dermatitis (AD) who are candidates for systemic therapy, according to a statement from the manufacturer.
The positive CHMP opinion, issued on Sept. 15, recommends extending the use of tralokinumab (Adtralza), an interleukin-13 antagonist, to adolescents aged 12-17 years in the EU. The positive opinion recommends an initial dose of 600 mg administered subcutaneously followed by 300 mg every other week, the dosing recommended for adults.
In December 2021, tralokinumab was approved for adults with moderate to severe AD in the United States, where it is marketed as Adbry. It is also approved for adults in the EU, Great Britain, Canada, the United Arab Emirates, and Switzerland. It is not currently approved for treatment of adolescents in any country, according to the LEO Pharma statement.
A regulatory filing with the U.S. Food and Drug Administration is in progress, the company said, and an additional study of tralokinumab for individuals aged 12 years and older is underway, according to the manufacturer.
The CHMP opinion was supported by data from a phase 3 study (ECZTRA 6) that assessed safety and efficacy of 150-mg or 300-mg doses of tralokinumab, compared with placebo in adolescents with moderate-to-severe AD, the company statement said. The primary outcomes were an Investigator Global Assessment score of clear or almost clear skin (IGA 0/1) and an improvement of at least a 75% on the Eczema Area and Severity Index score (EASI-75). In the study, presented as a poster at a meeting in October 2021, a total of 195 adolescents aged 12-17 with moderate to severe AD who were candidates for systemic therapy were randomly assigned to tralokinumab and 94 to placebo.
At 16 weeks, 21.4% and 17.5% of patients who received 150 mg and 300 mg, respectively, of tralokinumab had IGA scores of 0 or 1, compared with 4.3% of those on placebo (P < .001, P = .002, respectively vs. placebo). In addition, 28.6% and 27.8% of the 150-mg and 300-mg tralokinumab groups, respectively, achieved EASI-75, compared with 6.4% of placebo patients (P < .001, P = .001, respectively, compared with placebo).
Adverse events were similar between the groups, and most were mild or moderate; overall safety profiles were similar to those seen in adult patients.
The European Commission will review the positive opinion and make a final decision.
The research was supported by LEO Pharma.
A version of this article first appeared on Medscape.com.
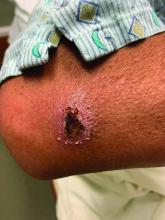
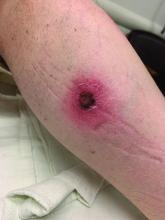
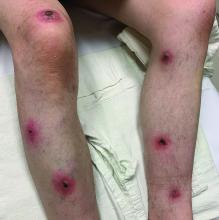
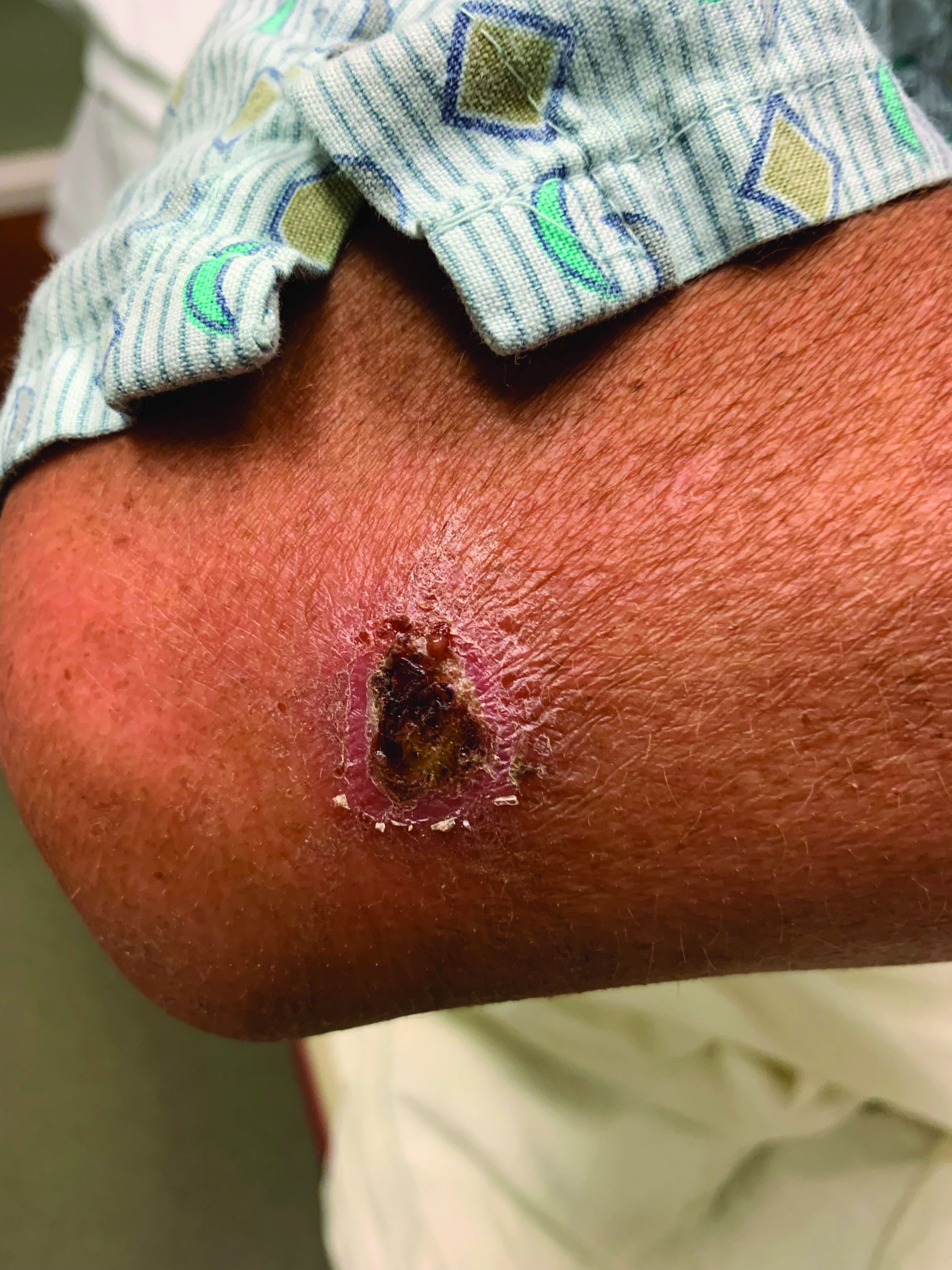
A White male presented with a 1-month history of recurrent, widespread, painful sores
Coinfection of staphylococci and streptococci can make it more challenging to treat. Lesions typically begin as a vesicle that enlarges and forms an ulcer with a hemorrhagic crust. Even with treatment, the depth of the lesions may result in scarring. Shins and dorsal feet are nearly always involved. Systemic involvement is rare.
Open wounds, bites, or dermatoses are risk factors for the development of ecthyma. Additionally, poor hygiene and malnutrition play a major role in inoculation and severity of the disease. Poor hygiene may serve as the initiating factor for infection, but malnutrition permits further development because of the body’s inability to mount a sufficient immune response. Intravenous drug users and patients with HIV tend to be affected.
When diagnosing ecthyma, it is important to correlate clinical signs with a bacterial culture. This condition can be difficult to treat because of both coinfection and growing antibiotic resistance in staphylococcal and streptococcal species. Specifically, S. aureus has been found to be resistant to beta-lactam antibiotics for many years, with methicillin-resistant S. aureus (MRSA) being first detected in 1961. While a variety of antibiotics are indicated, the prescription should be tailored to cover the cultured organism.
Topical antibiotics are sufficient for more superficial lesions. Both topical and oral antibiotics may be recommended for ecthyma as the infection can spread more deeply into the skin, eventually causing a cellulitis. Treatment protocol for oral agents varies based on which drug is indicated. This patient was seen in the emergency room. His white blood cell count was elevated at 9 × 109/L. He was started empirically on amoxicillin/clavulanate (Augmentin) and ciprofloxacin. Bacterial cultures grew out Streptococcus pyogenes.
The case and photos were submitted by Lucas Shapiro, BS, Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Fla., and Susannah Berke, MD, Three Rivers Dermatology, Coraopolis, Pa. Dr. Bilu Martin edited the column. Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Kwak Y et al. Infect Chemother. 2017 Dec;49(4):301-25.
2. Pereira LB. An Bras Dermatol. 2014 Mar-Apr;89(2):293-9.
3. Wasserzug O et al. Clin Infect Dis. 2009 May 1;48(9):1213-9.
Coinfection of staphylococci and streptococci can make it more challenging to treat. Lesions typically begin as a vesicle that enlarges and forms an ulcer with a hemorrhagic crust. Even with treatment, the depth of the lesions may result in scarring. Shins and dorsal feet are nearly always involved. Systemic involvement is rare.
Open wounds, bites, or dermatoses are risk factors for the development of ecthyma. Additionally, poor hygiene and malnutrition play a major role in inoculation and severity of the disease. Poor hygiene may serve as the initiating factor for infection, but malnutrition permits further development because of the body’s inability to mount a sufficient immune response. Intravenous drug users and patients with HIV tend to be affected.
When diagnosing ecthyma, it is important to correlate clinical signs with a bacterial culture. This condition can be difficult to treat because of both coinfection and growing antibiotic resistance in staphylococcal and streptococcal species. Specifically, S. aureus has been found to be resistant to beta-lactam antibiotics for many years, with methicillin-resistant S. aureus (MRSA) being first detected in 1961. While a variety of antibiotics are indicated, the prescription should be tailored to cover the cultured organism.
Topical antibiotics are sufficient for more superficial lesions. Both topical and oral antibiotics may be recommended for ecthyma as the infection can spread more deeply into the skin, eventually causing a cellulitis. Treatment protocol for oral agents varies based on which drug is indicated. This patient was seen in the emergency room. His white blood cell count was elevated at 9 × 109/L. He was started empirically on amoxicillin/clavulanate (Augmentin) and ciprofloxacin. Bacterial cultures grew out Streptococcus pyogenes.
The case and photos were submitted by Lucas Shapiro, BS, Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Fla., and Susannah Berke, MD, Three Rivers Dermatology, Coraopolis, Pa. Dr. Bilu Martin edited the column. Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Kwak Y et al. Infect Chemother. 2017 Dec;49(4):301-25.
2. Pereira LB. An Bras Dermatol. 2014 Mar-Apr;89(2):293-9.
3. Wasserzug O et al. Clin Infect Dis. 2009 May 1;48(9):1213-9.
Coinfection of staphylococci and streptococci can make it more challenging to treat. Lesions typically begin as a vesicle that enlarges and forms an ulcer with a hemorrhagic crust. Even with treatment, the depth of the lesions may result in scarring. Shins and dorsal feet are nearly always involved. Systemic involvement is rare.
Open wounds, bites, or dermatoses are risk factors for the development of ecthyma. Additionally, poor hygiene and malnutrition play a major role in inoculation and severity of the disease. Poor hygiene may serve as the initiating factor for infection, but malnutrition permits further development because of the body’s inability to mount a sufficient immune response. Intravenous drug users and patients with HIV tend to be affected.
When diagnosing ecthyma, it is important to correlate clinical signs with a bacterial culture. This condition can be difficult to treat because of both coinfection and growing antibiotic resistance in staphylococcal and streptococcal species. Specifically, S. aureus has been found to be resistant to beta-lactam antibiotics for many years, with methicillin-resistant S. aureus (MRSA) being first detected in 1961. While a variety of antibiotics are indicated, the prescription should be tailored to cover the cultured organism.
Topical antibiotics are sufficient for more superficial lesions. Both topical and oral antibiotics may be recommended for ecthyma as the infection can spread more deeply into the skin, eventually causing a cellulitis. Treatment protocol for oral agents varies based on which drug is indicated. This patient was seen in the emergency room. His white blood cell count was elevated at 9 × 109/L. He was started empirically on amoxicillin/clavulanate (Augmentin) and ciprofloxacin. Bacterial cultures grew out Streptococcus pyogenes.
The case and photos were submitted by Lucas Shapiro, BS, Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Fla., and Susannah Berke, MD, Three Rivers Dermatology, Coraopolis, Pa. Dr. Bilu Martin edited the column. Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Kwak Y et al. Infect Chemother. 2017 Dec;49(4):301-25.
2. Pereira LB. An Bras Dermatol. 2014 Mar-Apr;89(2):293-9.
3. Wasserzug O et al. Clin Infect Dis. 2009 May 1;48(9):1213-9.
House passes prior authorization bill, Senate path unclear
The path through the U.S. Senate is not yet certain for a bill intended to speed the prior authorization process of insurer-run Medicare Advantage plans, despite the measure having breezed through the House.
House leaders opted to move the Improving Seniors’ Timely Access to Care Act of 2021 (HR 3173) without requiring a roll-call vote. The measure was passed on Sept. 14 by a voice vote, an approach used in general with only uncontroversial measures that have broad support. The bill has 191 Democratic and 135 Republican sponsors, representing about three-quarters of the members of the House.
“There is no reason that patients should be waiting for medically appropriate care, especially when we know that this can lead to worse outcomes,” Rep. Earl Blumenauer (D-Ore.) said in a Sept. 14 speech on the House floor. “The fundamental promise of Medicare Advantage is undermined when people are delaying care, getting sicker, and ultimately costing Medicare more money.”
Rep. Greg Murphy, MD (R-N.C.), spoke on the House floor that day as well, bringing up cases he has seen in his own urology practice in which prior authorization delays disrupted medical care. One patient wound up in the hospital with abscess after an insurer denied an antibiotic prescription, Rep. Murphy said.
But the Senate appears unlikely at this time to move the prior authorization bill as a standalone measure. Instead, the bill may become part of a larger legislative package focused on health care that the Senate Finance Committee intends to prepare later this year.
The House-passed bill would require insurer-run Medicare plans to respond to expedited requests for prior authorization of services within 24 hours and to other requests within 7 days. This bill also would establish an electronic program for prior authorizations and mandate increased transparency as to how insurers use this tool.
CBO: Cost of change would be billions
In seeking to mandate changes in prior authorization, lawmakers likely will need to contend with the issue of a $16 billion cumulative cost estimate for the bill from the Congressional Budget Office. Members of Congress often seek to offset new spending by pairing bills that add to expected costs for the federal government with ones expected to produce savings.
Unlike Rep. Blumenauer, Rep. Murphy, and other backers of the prior authorization streamlining bill, CBO staff estimates that making the mandated changes would raise federal spending, inasmuch as there would be “a greater use of services.”
On Sept. 14, CBO issued a one-page report on the costs of the bill. The CBO report concerns only the bill in question, as is common practice with the office’s estimates.
Prior authorization changes would begin in fiscal 2025 and would add $899 million in spending, or outlays, that year, CBO said. The annual costs from the streamlined prior authorization practices through fiscal 2026 to 2032 range from $1.6 billion to $2.7 billion.
Looking at the CBO estimate against a backdrop of total Medicare Advantage costs, though, may provide important context.
The increases in spending estimated by CBO may suggest that there would be little change in federal spending as a result of streamlining prior authorization practices. These estimates of increased annual spending of $1.6 billion–$2.7 billion are only a small fraction of the current annual cost of insurer-run Medicare, and they represent an even smaller share of the projected expense.
The federal government last year spent about $350 billion on insurer-run plans, excluding Part D drug plan payments, according to the Medicare Advisory Payment Commission (MedPAC).
As of 2021, about 27 million people were enrolled in these plans, accounting for about 46% of the total Medicare population. Enrollment has doubled since 2010, MedPAC said, and it is expected to continue to grow. By 2027, insurer-run Medicare could cover 50% of the program’s population, a figure that may reach 53% by 2031.
Federal payments to these plans will accelerate in the years ahead as insurers attract more people eligible for Medicare as customers. Payments to these private health plans could rise from an expected $418 billion this year to $940.6 billion by 2031, according to the most recent Medicare trustees report.
Good intentions, poor implementation?
Insurer-run Medicare has long enjoyed deep bipartisan support in Congress. That’s due in part to its potential for reducing spending on what are considered low-value treatments, or ones considered unlikely to provide a significant medical benefit, but Rep. Blumenauer is among the members of Congress who see insurer-run Medicare as a path for preserving the giant federal health program. Traditional Medicare has far fewer restrictions on services, which sometimes opens a path for tests and treatments that offer less value for patients.
“I believe that the way traditional fee-for-service Medicare operates is not sustainable and that Medicare Advantage is one of the tools we can use to demonstrate how we can incentivize value,” Rep. Blumenauer said on the House floor. “But this is only possible when the program operates as intended. I have been deeply concerned about the reports of delays in care” caused by the clunky prior authorization processes.
He highlighted a recent report from the internal watchdog group for the Department of Health & Human Services that raises concerns about denials of appropriate care. About 18% of a set of payment denials examined by the Office of Inspector General of HHS in April actually met Medicare coverage rules and plan billing rules.
“For patients and their families, being told that you need to wait longer for care that your doctor tells you that you need is incredibly frustrating and frightening,” Rep. Blumenauer said. “There’s no comfort to be found in the fact that your insurance company needs time to decide if your doctor is right.”
Trends in prior authorization
The CBO report does not provide detail on what kind of medical spending would increase under a streamlined prior authorization process in insurer-run Medicare plans.
From trends reported in prior authorization, though, two factors could be at play in what appear to be relatively small estimated increases in Medicare spending from streamlined prior authorization.
One is the work already underway to create less burdensome electronic systems for these requests, such as the Fast Prior Authorization Technology Highway initiative run by the trade association America’s Health Insurance Plans.
The other factor could be the number of cases in which prior authorization merely causes delays in treatments and tests and thus simply postpones spending while adding to clinicians’ administrative work.
An analysis of prior authorization requests for dermatologic practices affiliated with the University of Utah may represent an extreme example. In a report published in JAMA Dermatology in 2020, researchers described what happened with requests made during 1 month, September 2016.
The approval rate for procedures was 99.6% – 100% (95 of 95) for Mohs surgery, and 96% (130 of 131, with 4 additional cases pending) for excisions. These findings supported calls for simplifying prior authorization procedures, “perhaps first by eliminating unnecessary PAs [prior authorizations] and appeals,” Aaron M. Secrest, MD, PhD, of the University of Utah, Salt Lake City, and coauthors wrote in the article.
Still, there is some evidence that insurer-run Medicare policies reduce the use of low-value care.
In a study published in JAMA Health Forum, Emily Boudreau, PhD, of insurer Humana Inc, and coauthors from Tufts University, Boston, and the University of Pennsylvania, Philadelphia investigated whether insurer-run Medicare could do a better job in reducing the amount of low-value care delivered than the traditional program. They analyzed a set of claims data from 2017 to 2019 for people enrolled in insurer-run and traditional Medicare.
They reported a rate of 23.07 low-value services provided per 100 people in insurer-run Medicare, compared with 25.39 for those in traditional Medicare. Some of the biggest differences reported in the article were in cancer screenings for older people.
As an example, the U.S. Preventive Services Task Force recommends that women older than 65 years not be screened for cervical cancer if they have undergone adequate screening in the past and are not at high risk for cervical cancer. There was an annual count of 1.76 screenings for cervical cancer per 100 women older than 65 in the insurer-run Medicare group versus 3.18 for those in traditional Medicare.
The Better Medicare Alliance issued a statement in favor of the House passage of the Improving Seniors’ Timely Access to Care Act.
In it, the group said the measure would “modernize prior authorization while protecting its essential function in facilitating safe, high-value, evidence-based care.” The alliance promotes use of insurer-run Medicare. The board of the Better Medicare Alliance includes executives who serve with firms that run Advantage plans as well as medical organizations and universities.
“With studies showing that up to one-quarter of all health care expenditures are wasted on services with no benefit to the patient, we need a robust, next-generation prior authorization program to deter low-value, and even harmful, care while protecting access to needed treatment and effective therapies,” said A. Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, in a statement issued by the Better Medicare Alliance. He is a member of the group’s council of scholars.
On the House floor on September 14, Rep. Ami Bera, MD (D-Calif.), said he has heard from former colleagues and his medical school classmates that they now spend as much as 40% of their time on administrative work. These distractions from patient care are helping drive physicians away from the practice of medicine.
Still, the internist defended the basic premise of prior authorization while strongly appealing for better systems of handling it.
“Yes, there is a role for prior authorization in limited cases. There is also a role to go back and retrospectively look at how care is being delivered,” Rep. Bera said. “But what is happening today is a travesty. It wasn’t the intention of prior authorization. It is a prior authorization process gone awry.”
A version of this article first appeared on Medscape.com.
The path through the U.S. Senate is not yet certain for a bill intended to speed the prior authorization process of insurer-run Medicare Advantage plans, despite the measure having breezed through the House.
House leaders opted to move the Improving Seniors’ Timely Access to Care Act of 2021 (HR 3173) without requiring a roll-call vote. The measure was passed on Sept. 14 by a voice vote, an approach used in general with only uncontroversial measures that have broad support. The bill has 191 Democratic and 135 Republican sponsors, representing about three-quarters of the members of the House.
“There is no reason that patients should be waiting for medically appropriate care, especially when we know that this can lead to worse outcomes,” Rep. Earl Blumenauer (D-Ore.) said in a Sept. 14 speech on the House floor. “The fundamental promise of Medicare Advantage is undermined when people are delaying care, getting sicker, and ultimately costing Medicare more money.”
Rep. Greg Murphy, MD (R-N.C.), spoke on the House floor that day as well, bringing up cases he has seen in his own urology practice in which prior authorization delays disrupted medical care. One patient wound up in the hospital with abscess after an insurer denied an antibiotic prescription, Rep. Murphy said.
But the Senate appears unlikely at this time to move the prior authorization bill as a standalone measure. Instead, the bill may become part of a larger legislative package focused on health care that the Senate Finance Committee intends to prepare later this year.
The House-passed bill would require insurer-run Medicare plans to respond to expedited requests for prior authorization of services within 24 hours and to other requests within 7 days. This bill also would establish an electronic program for prior authorizations and mandate increased transparency as to how insurers use this tool.
CBO: Cost of change would be billions
In seeking to mandate changes in prior authorization, lawmakers likely will need to contend with the issue of a $16 billion cumulative cost estimate for the bill from the Congressional Budget Office. Members of Congress often seek to offset new spending by pairing bills that add to expected costs for the federal government with ones expected to produce savings.
Unlike Rep. Blumenauer, Rep. Murphy, and other backers of the prior authorization streamlining bill, CBO staff estimates that making the mandated changes would raise federal spending, inasmuch as there would be “a greater use of services.”
On Sept. 14, CBO issued a one-page report on the costs of the bill. The CBO report concerns only the bill in question, as is common practice with the office’s estimates.
Prior authorization changes would begin in fiscal 2025 and would add $899 million in spending, or outlays, that year, CBO said. The annual costs from the streamlined prior authorization practices through fiscal 2026 to 2032 range from $1.6 billion to $2.7 billion.
Looking at the CBO estimate against a backdrop of total Medicare Advantage costs, though, may provide important context.
The increases in spending estimated by CBO may suggest that there would be little change in federal spending as a result of streamlining prior authorization practices. These estimates of increased annual spending of $1.6 billion–$2.7 billion are only a small fraction of the current annual cost of insurer-run Medicare, and they represent an even smaller share of the projected expense.
The federal government last year spent about $350 billion on insurer-run plans, excluding Part D drug plan payments, according to the Medicare Advisory Payment Commission (MedPAC).
As of 2021, about 27 million people were enrolled in these plans, accounting for about 46% of the total Medicare population. Enrollment has doubled since 2010, MedPAC said, and it is expected to continue to grow. By 2027, insurer-run Medicare could cover 50% of the program’s population, a figure that may reach 53% by 2031.
Federal payments to these plans will accelerate in the years ahead as insurers attract more people eligible for Medicare as customers. Payments to these private health plans could rise from an expected $418 billion this year to $940.6 billion by 2031, according to the most recent Medicare trustees report.
Good intentions, poor implementation?
Insurer-run Medicare has long enjoyed deep bipartisan support in Congress. That’s due in part to its potential for reducing spending on what are considered low-value treatments, or ones considered unlikely to provide a significant medical benefit, but Rep. Blumenauer is among the members of Congress who see insurer-run Medicare as a path for preserving the giant federal health program. Traditional Medicare has far fewer restrictions on services, which sometimes opens a path for tests and treatments that offer less value for patients.
“I believe that the way traditional fee-for-service Medicare operates is not sustainable and that Medicare Advantage is one of the tools we can use to demonstrate how we can incentivize value,” Rep. Blumenauer said on the House floor. “But this is only possible when the program operates as intended. I have been deeply concerned about the reports of delays in care” caused by the clunky prior authorization processes.
He highlighted a recent report from the internal watchdog group for the Department of Health & Human Services that raises concerns about denials of appropriate care. About 18% of a set of payment denials examined by the Office of Inspector General of HHS in April actually met Medicare coverage rules and plan billing rules.
“For patients and their families, being told that you need to wait longer for care that your doctor tells you that you need is incredibly frustrating and frightening,” Rep. Blumenauer said. “There’s no comfort to be found in the fact that your insurance company needs time to decide if your doctor is right.”
Trends in prior authorization
The CBO report does not provide detail on what kind of medical spending would increase under a streamlined prior authorization process in insurer-run Medicare plans.
From trends reported in prior authorization, though, two factors could be at play in what appear to be relatively small estimated increases in Medicare spending from streamlined prior authorization.
One is the work already underway to create less burdensome electronic systems for these requests, such as the Fast Prior Authorization Technology Highway initiative run by the trade association America’s Health Insurance Plans.
The other factor could be the number of cases in which prior authorization merely causes delays in treatments and tests and thus simply postpones spending while adding to clinicians’ administrative work.
An analysis of prior authorization requests for dermatologic practices affiliated with the University of Utah may represent an extreme example. In a report published in JAMA Dermatology in 2020, researchers described what happened with requests made during 1 month, September 2016.
The approval rate for procedures was 99.6% – 100% (95 of 95) for Mohs surgery, and 96% (130 of 131, with 4 additional cases pending) for excisions. These findings supported calls for simplifying prior authorization procedures, “perhaps first by eliminating unnecessary PAs [prior authorizations] and appeals,” Aaron M. Secrest, MD, PhD, of the University of Utah, Salt Lake City, and coauthors wrote in the article.
Still, there is some evidence that insurer-run Medicare policies reduce the use of low-value care.
In a study published in JAMA Health Forum, Emily Boudreau, PhD, of insurer Humana Inc, and coauthors from Tufts University, Boston, and the University of Pennsylvania, Philadelphia investigated whether insurer-run Medicare could do a better job in reducing the amount of low-value care delivered than the traditional program. They analyzed a set of claims data from 2017 to 2019 for people enrolled in insurer-run and traditional Medicare.
They reported a rate of 23.07 low-value services provided per 100 people in insurer-run Medicare, compared with 25.39 for those in traditional Medicare. Some of the biggest differences reported in the article were in cancer screenings for older people.
As an example, the U.S. Preventive Services Task Force recommends that women older than 65 years not be screened for cervical cancer if they have undergone adequate screening in the past and are not at high risk for cervical cancer. There was an annual count of 1.76 screenings for cervical cancer per 100 women older than 65 in the insurer-run Medicare group versus 3.18 for those in traditional Medicare.
The Better Medicare Alliance issued a statement in favor of the House passage of the Improving Seniors’ Timely Access to Care Act.
In it, the group said the measure would “modernize prior authorization while protecting its essential function in facilitating safe, high-value, evidence-based care.” The alliance promotes use of insurer-run Medicare. The board of the Better Medicare Alliance includes executives who serve with firms that run Advantage plans as well as medical organizations and universities.
“With studies showing that up to one-quarter of all health care expenditures are wasted on services with no benefit to the patient, we need a robust, next-generation prior authorization program to deter low-value, and even harmful, care while protecting access to needed treatment and effective therapies,” said A. Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, in a statement issued by the Better Medicare Alliance. He is a member of the group’s council of scholars.
On the House floor on September 14, Rep. Ami Bera, MD (D-Calif.), said he has heard from former colleagues and his medical school classmates that they now spend as much as 40% of their time on administrative work. These distractions from patient care are helping drive physicians away from the practice of medicine.
Still, the internist defended the basic premise of prior authorization while strongly appealing for better systems of handling it.
“Yes, there is a role for prior authorization in limited cases. There is also a role to go back and retrospectively look at how care is being delivered,” Rep. Bera said. “But what is happening today is a travesty. It wasn’t the intention of prior authorization. It is a prior authorization process gone awry.”
A version of this article first appeared on Medscape.com.
The path through the U.S. Senate is not yet certain for a bill intended to speed the prior authorization process of insurer-run Medicare Advantage plans, despite the measure having breezed through the House.
House leaders opted to move the Improving Seniors’ Timely Access to Care Act of 2021 (HR 3173) without requiring a roll-call vote. The measure was passed on Sept. 14 by a voice vote, an approach used in general with only uncontroversial measures that have broad support. The bill has 191 Democratic and 135 Republican sponsors, representing about three-quarters of the members of the House.
“There is no reason that patients should be waiting for medically appropriate care, especially when we know that this can lead to worse outcomes,” Rep. Earl Blumenauer (D-Ore.) said in a Sept. 14 speech on the House floor. “The fundamental promise of Medicare Advantage is undermined when people are delaying care, getting sicker, and ultimately costing Medicare more money.”
Rep. Greg Murphy, MD (R-N.C.), spoke on the House floor that day as well, bringing up cases he has seen in his own urology practice in which prior authorization delays disrupted medical care. One patient wound up in the hospital with abscess after an insurer denied an antibiotic prescription, Rep. Murphy said.
But the Senate appears unlikely at this time to move the prior authorization bill as a standalone measure. Instead, the bill may become part of a larger legislative package focused on health care that the Senate Finance Committee intends to prepare later this year.
The House-passed bill would require insurer-run Medicare plans to respond to expedited requests for prior authorization of services within 24 hours and to other requests within 7 days. This bill also would establish an electronic program for prior authorizations and mandate increased transparency as to how insurers use this tool.
CBO: Cost of change would be billions
In seeking to mandate changes in prior authorization, lawmakers likely will need to contend with the issue of a $16 billion cumulative cost estimate for the bill from the Congressional Budget Office. Members of Congress often seek to offset new spending by pairing bills that add to expected costs for the federal government with ones expected to produce savings.
Unlike Rep. Blumenauer, Rep. Murphy, and other backers of the prior authorization streamlining bill, CBO staff estimates that making the mandated changes would raise federal spending, inasmuch as there would be “a greater use of services.”
On Sept. 14, CBO issued a one-page report on the costs of the bill. The CBO report concerns only the bill in question, as is common practice with the office’s estimates.
Prior authorization changes would begin in fiscal 2025 and would add $899 million in spending, or outlays, that year, CBO said. The annual costs from the streamlined prior authorization practices through fiscal 2026 to 2032 range from $1.6 billion to $2.7 billion.
Looking at the CBO estimate against a backdrop of total Medicare Advantage costs, though, may provide important context.
The increases in spending estimated by CBO may suggest that there would be little change in federal spending as a result of streamlining prior authorization practices. These estimates of increased annual spending of $1.6 billion–$2.7 billion are only a small fraction of the current annual cost of insurer-run Medicare, and they represent an even smaller share of the projected expense.
The federal government last year spent about $350 billion on insurer-run plans, excluding Part D drug plan payments, according to the Medicare Advisory Payment Commission (MedPAC).
As of 2021, about 27 million people were enrolled in these plans, accounting for about 46% of the total Medicare population. Enrollment has doubled since 2010, MedPAC said, and it is expected to continue to grow. By 2027, insurer-run Medicare could cover 50% of the program’s population, a figure that may reach 53% by 2031.
Federal payments to these plans will accelerate in the years ahead as insurers attract more people eligible for Medicare as customers. Payments to these private health plans could rise from an expected $418 billion this year to $940.6 billion by 2031, according to the most recent Medicare trustees report.
Good intentions, poor implementation?
Insurer-run Medicare has long enjoyed deep bipartisan support in Congress. That’s due in part to its potential for reducing spending on what are considered low-value treatments, or ones considered unlikely to provide a significant medical benefit, but Rep. Blumenauer is among the members of Congress who see insurer-run Medicare as a path for preserving the giant federal health program. Traditional Medicare has far fewer restrictions on services, which sometimes opens a path for tests and treatments that offer less value for patients.
“I believe that the way traditional fee-for-service Medicare operates is not sustainable and that Medicare Advantage is one of the tools we can use to demonstrate how we can incentivize value,” Rep. Blumenauer said on the House floor. “But this is only possible when the program operates as intended. I have been deeply concerned about the reports of delays in care” caused by the clunky prior authorization processes.
He highlighted a recent report from the internal watchdog group for the Department of Health & Human Services that raises concerns about denials of appropriate care. About 18% of a set of payment denials examined by the Office of Inspector General of HHS in April actually met Medicare coverage rules and plan billing rules.
“For patients and their families, being told that you need to wait longer for care that your doctor tells you that you need is incredibly frustrating and frightening,” Rep. Blumenauer said. “There’s no comfort to be found in the fact that your insurance company needs time to decide if your doctor is right.”
Trends in prior authorization
The CBO report does not provide detail on what kind of medical spending would increase under a streamlined prior authorization process in insurer-run Medicare plans.
From trends reported in prior authorization, though, two factors could be at play in what appear to be relatively small estimated increases in Medicare spending from streamlined prior authorization.
One is the work already underway to create less burdensome electronic systems for these requests, such as the Fast Prior Authorization Technology Highway initiative run by the trade association America’s Health Insurance Plans.
The other factor could be the number of cases in which prior authorization merely causes delays in treatments and tests and thus simply postpones spending while adding to clinicians’ administrative work.
An analysis of prior authorization requests for dermatologic practices affiliated with the University of Utah may represent an extreme example. In a report published in JAMA Dermatology in 2020, researchers described what happened with requests made during 1 month, September 2016.
The approval rate for procedures was 99.6% – 100% (95 of 95) for Mohs surgery, and 96% (130 of 131, with 4 additional cases pending) for excisions. These findings supported calls for simplifying prior authorization procedures, “perhaps first by eliminating unnecessary PAs [prior authorizations] and appeals,” Aaron M. Secrest, MD, PhD, of the University of Utah, Salt Lake City, and coauthors wrote in the article.
Still, there is some evidence that insurer-run Medicare policies reduce the use of low-value care.
In a study published in JAMA Health Forum, Emily Boudreau, PhD, of insurer Humana Inc, and coauthors from Tufts University, Boston, and the University of Pennsylvania, Philadelphia investigated whether insurer-run Medicare could do a better job in reducing the amount of low-value care delivered than the traditional program. They analyzed a set of claims data from 2017 to 2019 for people enrolled in insurer-run and traditional Medicare.
They reported a rate of 23.07 low-value services provided per 100 people in insurer-run Medicare, compared with 25.39 for those in traditional Medicare. Some of the biggest differences reported in the article were in cancer screenings for older people.
As an example, the U.S. Preventive Services Task Force recommends that women older than 65 years not be screened for cervical cancer if they have undergone adequate screening in the past and are not at high risk for cervical cancer. There was an annual count of 1.76 screenings for cervical cancer per 100 women older than 65 in the insurer-run Medicare group versus 3.18 for those in traditional Medicare.
The Better Medicare Alliance issued a statement in favor of the House passage of the Improving Seniors’ Timely Access to Care Act.
In it, the group said the measure would “modernize prior authorization while protecting its essential function in facilitating safe, high-value, evidence-based care.” The alliance promotes use of insurer-run Medicare. The board of the Better Medicare Alliance includes executives who serve with firms that run Advantage plans as well as medical organizations and universities.
“With studies showing that up to one-quarter of all health care expenditures are wasted on services with no benefit to the patient, we need a robust, next-generation prior authorization program to deter low-value, and even harmful, care while protecting access to needed treatment and effective therapies,” said A. Mark Fendrick, MD, director of the University of Michigan’s Center for Value-Based Insurance Design in Ann Arbor, in a statement issued by the Better Medicare Alliance. He is a member of the group’s council of scholars.
On the House floor on September 14, Rep. Ami Bera, MD (D-Calif.), said he has heard from former colleagues and his medical school classmates that they now spend as much as 40% of their time on administrative work. These distractions from patient care are helping drive physicians away from the practice of medicine.
Still, the internist defended the basic premise of prior authorization while strongly appealing for better systems of handling it.
“Yes, there is a role for prior authorization in limited cases. There is also a role to go back and retrospectively look at how care is being delivered,” Rep. Bera said. “But what is happening today is a travesty. It wasn’t the intention of prior authorization. It is a prior authorization process gone awry.”
A version of this article first appeared on Medscape.com.
Meta-analysis demonstrates potential of probiotics in reducing atopic dermatitis disease severity
Key clinical point: The use of probiotic supplementation reduced disease severity in adult patients with atopic dermatitis (AD).
Major finding: Probiotic supplementation vs placebo led to a significant reduction in the Scoring AD index (mean difference −7.90; 95% CI −7.25 to−6.92), but no significant improvements in skin severity and itch severity.
Study details: Findings are from a meta-analysis of six randomized controlled trials including 241 adults with AD, of which 128 received probiotics and 113 received placebo.
Disclosures: This study was funded by a grant from Universitas Airlangga, Indonesia. The authors declared no conflicts of interest.
Source: Umborowati MA et al. The role of probiotics in the treatment of adult atopic dermatitis: a meta-analysis of randomized controlled trials. J Health Popul Nutr. 2022;41:37 (Aug 17). Doi: 10.1186/s41043-022-00318-6
Key clinical point: The use of probiotic supplementation reduced disease severity in adult patients with atopic dermatitis (AD).
Major finding: Probiotic supplementation vs placebo led to a significant reduction in the Scoring AD index (mean difference −7.90; 95% CI −7.25 to−6.92), but no significant improvements in skin severity and itch severity.
Study details: Findings are from a meta-analysis of six randomized controlled trials including 241 adults with AD, of which 128 received probiotics and 113 received placebo.
Disclosures: This study was funded by a grant from Universitas Airlangga, Indonesia. The authors declared no conflicts of interest.
Source: Umborowati MA et al. The role of probiotics in the treatment of adult atopic dermatitis: a meta-analysis of randomized controlled trials. J Health Popul Nutr. 2022;41:37 (Aug 17). Doi: 10.1186/s41043-022-00318-6
Key clinical point: The use of probiotic supplementation reduced disease severity in adult patients with atopic dermatitis (AD).
Major finding: Probiotic supplementation vs placebo led to a significant reduction in the Scoring AD index (mean difference −7.90; 95% CI −7.25 to−6.92), but no significant improvements in skin severity and itch severity.
Study details: Findings are from a meta-analysis of six randomized controlled trials including 241 adults with AD, of which 128 received probiotics and 113 received placebo.
Disclosures: This study was funded by a grant from Universitas Airlangga, Indonesia. The authors declared no conflicts of interest.
Source: Umborowati MA et al. The role of probiotics in the treatment of adult atopic dermatitis: a meta-analysis of randomized controlled trials. J Health Popul Nutr. 2022;41:37 (Aug 17). Doi: 10.1186/s41043-022-00318-6
Real-world characteristics of patients with moderate-to-severe atopic dermatitis receiving dupilumab
Key clinical point: Patients who received dupilumab for atopic dermatitis (AD) had moderate-to-severe disease, long medical history, and high prevalence of coexisting type 2 inflammatory diseases.
Major finding: A majority of patients (66.6%) were diagnosed with AD in childhood, and most patients presented with bordering moderate-to-severe AD (Eczema Area and Severity Index > 21), high prevalence of pruritus (99.6%), and coexisting atopic and type 2 inflammatory diseases (51.8%).
Study details: Findings are from an analysis of PROLEAD, a national, multicenter, prospective, non-interventional study, including 817 patients with moderate-to-severe AD who received dupilumab.
Disclosures: This study was funded by Sanofi. Three authors declared being employees of or holding stocks in Sanofi. The other authors reported ties with several sources, including Sanofi.
Source: Thaci D et al. Dupilumab treatment of atopic dermatitis in routine clinical care: Baseline characteristics of patients in the PROLEAD prospective, observational study. Dermatol Ther (Heidelb). 2022;12(9):2145-2160 (Aug 19). Doi: 10.1007/s13555-022-00791-1
Key clinical point: Patients who received dupilumab for atopic dermatitis (AD) had moderate-to-severe disease, long medical history, and high prevalence of coexisting type 2 inflammatory diseases.
Major finding: A majority of patients (66.6%) were diagnosed with AD in childhood, and most patients presented with bordering moderate-to-severe AD (Eczema Area and Severity Index > 21), high prevalence of pruritus (99.6%), and coexisting atopic and type 2 inflammatory diseases (51.8%).
Study details: Findings are from an analysis of PROLEAD, a national, multicenter, prospective, non-interventional study, including 817 patients with moderate-to-severe AD who received dupilumab.
Disclosures: This study was funded by Sanofi. Three authors declared being employees of or holding stocks in Sanofi. The other authors reported ties with several sources, including Sanofi.
Source: Thaci D et al. Dupilumab treatment of atopic dermatitis in routine clinical care: Baseline characteristics of patients in the PROLEAD prospective, observational study. Dermatol Ther (Heidelb). 2022;12(9):2145-2160 (Aug 19). Doi: 10.1007/s13555-022-00791-1
Key clinical point: Patients who received dupilumab for atopic dermatitis (AD) had moderate-to-severe disease, long medical history, and high prevalence of coexisting type 2 inflammatory diseases.
Major finding: A majority of patients (66.6%) were diagnosed with AD in childhood, and most patients presented with bordering moderate-to-severe AD (Eczema Area and Severity Index > 21), high prevalence of pruritus (99.6%), and coexisting atopic and type 2 inflammatory diseases (51.8%).
Study details: Findings are from an analysis of PROLEAD, a national, multicenter, prospective, non-interventional study, including 817 patients with moderate-to-severe AD who received dupilumab.
Disclosures: This study was funded by Sanofi. Three authors declared being employees of or holding stocks in Sanofi. The other authors reported ties with several sources, including Sanofi.
Source: Thaci D et al. Dupilumab treatment of atopic dermatitis in routine clinical care: Baseline characteristics of patients in the PROLEAD prospective, observational study. Dermatol Ther (Heidelb). 2022;12(9):2145-2160 (Aug 19). Doi: 10.1007/s13555-022-00791-1
Pediatric atopic dermatitis and neuropsychiatric disorders: What is the link?
Key clinical point: Atopic dermatitis (AD) did not increase the incidence risk for most neuropsychiatric disorders in a pediatric cohort.
Major finding: The risks for attention deficit hyperactivity disorder (hazard ratio [HR] 1.02; 95% CI 0.97-1.06), autism (HR 1.02; 95% CI 0.98-1.06), anxiety (HR 1.01; 95% CI 0.99-1.03), and bipolar disorder (HR 1.08; 95% CI 0.85-1.36) were comparable in the AD and non-AD groups. Participants with vs without AD were less likely to develop depression (HR 0.93; 95% CI 0.91-0.95) or schizophrenia (HR 0.72; 95% CI 0.54-0.95) but more likely to develop obsessive compulsive disorder (HR 1.26; 95% CI 1.16-1.37). However, the risks varied with disease severity and patient’s age.
Study details: Findings are from a retrospective population-based cohort study including 409,431 children with AD and 1,809,029 matched children without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. One author declared being an employee of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Wan J et al. Atopic dermatitis and risk of major neuropsychiatric disorders in children: A population-based cohort study. J Eur Acad Dermatol Venereol. 2022 (Aug 26). Doi: 10.1111/jdv.18564
Key clinical point: Atopic dermatitis (AD) did not increase the incidence risk for most neuropsychiatric disorders in a pediatric cohort.
Major finding: The risks for attention deficit hyperactivity disorder (hazard ratio [HR] 1.02; 95% CI 0.97-1.06), autism (HR 1.02; 95% CI 0.98-1.06), anxiety (HR 1.01; 95% CI 0.99-1.03), and bipolar disorder (HR 1.08; 95% CI 0.85-1.36) were comparable in the AD and non-AD groups. Participants with vs without AD were less likely to develop depression (HR 0.93; 95% CI 0.91-0.95) or schizophrenia (HR 0.72; 95% CI 0.54-0.95) but more likely to develop obsessive compulsive disorder (HR 1.26; 95% CI 1.16-1.37). However, the risks varied with disease severity and patient’s age.
Study details: Findings are from a retrospective population-based cohort study including 409,431 children with AD and 1,809,029 matched children without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. One author declared being an employee of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Wan J et al. Atopic dermatitis and risk of major neuropsychiatric disorders in children: A population-based cohort study. J Eur Acad Dermatol Venereol. 2022 (Aug 26). Doi: 10.1111/jdv.18564
Key clinical point: Atopic dermatitis (AD) did not increase the incidence risk for most neuropsychiatric disorders in a pediatric cohort.
Major finding: The risks for attention deficit hyperactivity disorder (hazard ratio [HR] 1.02; 95% CI 0.97-1.06), autism (HR 1.02; 95% CI 0.98-1.06), anxiety (HR 1.01; 95% CI 0.99-1.03), and bipolar disorder (HR 1.08; 95% CI 0.85-1.36) were comparable in the AD and non-AD groups. Participants with vs without AD were less likely to develop depression (HR 0.93; 95% CI 0.91-0.95) or schizophrenia (HR 0.72; 95% CI 0.54-0.95) but more likely to develop obsessive compulsive disorder (HR 1.26; 95% CI 1.16-1.37). However, the risks varied with disease severity and patient’s age.
Study details: Findings are from a retrospective population-based cohort study including 409,431 children with AD and 1,809,029 matched children without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. One author declared being an employee of Pfizer. The other authors reported ties with several sources, including Pfizer.
Source: Wan J et al. Atopic dermatitis and risk of major neuropsychiatric disorders in children: A population-based cohort study. J Eur Acad Dermatol Venereol. 2022 (Aug 26). Doi: 10.1111/jdv.18564
Early initiation of emollient reduces risk for atopic dermatitis in high risk infants
Key clinical point: Initiation of daily application of a specialized emollient from the first to the eighth week of life reduced the risk for atopic dermatitis (AD) incidence for 12 months in infants with high risk for AD.
Major finding: At 12 months, the cumulative incidence of AD was significantly lower in the emollient vs standard routine skin care group (32.8% vs 46.4%; relative risk 0.707; P = .036). The rate of patient-reported skin infections was similar between both the treatment groups during the 8-week intervention period (5.0% vs 5.7%).
Study details: Findings are from the STOP AD trial including 321 newborn infants at high risk for AD who were randomly assigned to receive twice-daily emollient for the first 8 weeks of life or standard routine skin care.
Disclosures: This study was supported by The City of Dublin Skin and Cancer Hospital Charity and the Skin Research Institute of Singapore. Some authors declared being managing directors, employees, shareholders, or consultants or receiving research funding, speaker fees, or consulting fees from several sources.
Source: Ní Chaoimh C, Lad D, et al. Early initiation of short-term emollient use for the prevention of atopic dermatitis in high risk infants - the STOP AD randomised controlled trial. Allergy. 2022 (Aug 23). Doi: 10.1111/all.15491
Key clinical point: Initiation of daily application of a specialized emollient from the first to the eighth week of life reduced the risk for atopic dermatitis (AD) incidence for 12 months in infants with high risk for AD.
Major finding: At 12 months, the cumulative incidence of AD was significantly lower in the emollient vs standard routine skin care group (32.8% vs 46.4%; relative risk 0.707; P = .036). The rate of patient-reported skin infections was similar between both the treatment groups during the 8-week intervention period (5.0% vs 5.7%).
Study details: Findings are from the STOP AD trial including 321 newborn infants at high risk for AD who were randomly assigned to receive twice-daily emollient for the first 8 weeks of life or standard routine skin care.
Disclosures: This study was supported by The City of Dublin Skin and Cancer Hospital Charity and the Skin Research Institute of Singapore. Some authors declared being managing directors, employees, shareholders, or consultants or receiving research funding, speaker fees, or consulting fees from several sources.
Source: Ní Chaoimh C, Lad D, et al. Early initiation of short-term emollient use for the prevention of atopic dermatitis in high risk infants - the STOP AD randomised controlled trial. Allergy. 2022 (Aug 23). Doi: 10.1111/all.15491
Key clinical point: Initiation of daily application of a specialized emollient from the first to the eighth week of life reduced the risk for atopic dermatitis (AD) incidence for 12 months in infants with high risk for AD.
Major finding: At 12 months, the cumulative incidence of AD was significantly lower in the emollient vs standard routine skin care group (32.8% vs 46.4%; relative risk 0.707; P = .036). The rate of patient-reported skin infections was similar between both the treatment groups during the 8-week intervention period (5.0% vs 5.7%).
Study details: Findings are from the STOP AD trial including 321 newborn infants at high risk for AD who were randomly assigned to receive twice-daily emollient for the first 8 weeks of life or standard routine skin care.
Disclosures: This study was supported by The City of Dublin Skin and Cancer Hospital Charity and the Skin Research Institute of Singapore. Some authors declared being managing directors, employees, shareholders, or consultants or receiving research funding, speaker fees, or consulting fees from several sources.
Source: Ní Chaoimh C, Lad D, et al. Early initiation of short-term emollient use for the prevention of atopic dermatitis in high risk infants - the STOP AD randomised controlled trial. Allergy. 2022 (Aug 23). Doi: 10.1111/all.15491
New science reveals the best way to take a pill
I want to tell you a story about forgetfulness and haste, and how the combination of the two can lead to frightening consequences. A few years ago, I was lying in bed about to turn out the light when I realized I’d forgotten to take “my pill.”
Like some 161 million other American adults, I was then a consumer of a prescription medication. Being conscientious, I got up, retrieved said pill, and tossed it back. Being lazy, I didn’t bother to grab a glass of water to help the thing go down. Instead, I promptly returned to bed, threw a pillow over my head, and prepared for sleep.
Within seconds, I began to feel a burning sensation in my chest. After about a minute, that burn became a crippling pain. Not wanting to alarm my wife, I went into the living room, where I spent the next 30 minutes doubled over in agony. Was I having a heart attack? I phoned my sister, a hospitalist in Texas. She advised me to take myself to the ED to get checked out.
If only I’d known then about “Duke.” He could have told me how critical body posture is when people swallow pills.
Who’s Duke?
Duke is a computer representation of a 34-year-old, anatomically normal human male created by computer scientists at the IT’IS Foundation, a nonprofit group based in Switzerland that works on a variety of projects in health care technology. Using Duke, Rajat Mittal, PhD, a professor of medicine at the Johns Hopkins University, Baltimore, created a computer model called “StomachSim” to explore the process of digestion.
Their research, published in the journal Physics of Fluids, turned up several surprising findings about the dynamics of swallowing pills – the most common way medication is used worldwide.
Dr. Mittal said he chose to study the stomach because the functions of most other organ systems, from the heart to the brain, have already attracted plenty of attention from scientists.
“As I was looking to initiate research in some new directions, the implications of stomach biomechanics on important conditions such as diabetes, obesity, and gastroparesis became apparent to me,” he said. “It was clear that bioengineering research in this arena lags other more ‘sexy’ areas such as cardiovascular flows by at least 20 years, and there seemed to be a great opportunity to do impactful work.”
Your posture may help a pill work better
Several well-known things affect a pill’s ability to disperse its contents into the gut and be used by the body, such as the stomach’s contents (a heavy breakfast, a mix of liquids like juice, milk, and coffee) and the motion of the organ’s walls. But Dr. Mittal’s group learned that Duke’s posture also played a major role.
The researchers ran Duke through computer simulations in varying postures: upright, leaning right, leaning left, and leaning back, while keeping all the other parts of their analyses (like the things mentioned above) the same.
They found that posture determined as much as 83% of how quickly a pill disperses into the intestines. The most efficient position was leaning right. The least was leaning left, which prevented the pill from reaching the antrum, or bottom section of the stomach, and thus kept all but traces of the dissolved drug from entering the duodenum, where the stomach joins the small intestine. (Interestingly, Jews who observe Passover are advised to recline to the left during the meal as a symbol of freedom and leisure.)
That makes sense if you think about the stomach’s shape, which looks kind of like a bean, curving from the left to the right side of the body. Because of gravity, your position will change where the pill lands.
a condition in which the stomach loses the ability to empty properly.
How this could help people
Among the groups most likely to benefit from such studies, Dr. Mittal said, are the elderly – who both take a lot of pills and are more prone to trouble swallowing because of age-related changes in their esophagus – and the bedridden, who can’t easily shift their posture. The findings may also lead to improvements in the ability to treat people with gastroparesis, a particular problem for people with diabetes.
Future studies with Duke and similar simulations will look at how the GI system digests proteins, carbohydrates, and fatty meals, Dr. Mittal said.
In the meantime, Dr. Mittal offered the following advice: “Standing or sitting upright after taking a pill is fine. If you have to take a pill lying down, stay on your back or on your right side. Avoid lying on your left side after taking a pill.”
As for what happened to me, any gastroenterologist reading this has figured out that my condition was not heart-related. Instead, I likely was having a bout of pill esophagitis, irritation that can result from medications that aggravate the mucosa of the food tube. Although painful, esophagitis isn’t life-threatening. After about an hour, the pain began to subside, and by the next morning I was fine, with only a faint ache in my chest to remind me of my earlier torment. (Researchers noted an increase in the condition early in the COVID-19 pandemic, linked to the antibiotic doxycycline.)
And, in the interest of accuracy, my pill problem began above the stomach. Nothing in the Hopkins research suggests that the alignment of the esophagus plays a role in how drugs disperse in the gut – unless, of course, it prevents those pills from reaching the stomach in the first place.
A version of this article first appeared on WebMD.com.
I want to tell you a story about forgetfulness and haste, and how the combination of the two can lead to frightening consequences. A few years ago, I was lying in bed about to turn out the light when I realized I’d forgotten to take “my pill.”
Like some 161 million other American adults, I was then a consumer of a prescription medication. Being conscientious, I got up, retrieved said pill, and tossed it back. Being lazy, I didn’t bother to grab a glass of water to help the thing go down. Instead, I promptly returned to bed, threw a pillow over my head, and prepared for sleep.
Within seconds, I began to feel a burning sensation in my chest. After about a minute, that burn became a crippling pain. Not wanting to alarm my wife, I went into the living room, where I spent the next 30 minutes doubled over in agony. Was I having a heart attack? I phoned my sister, a hospitalist in Texas. She advised me to take myself to the ED to get checked out.
If only I’d known then about “Duke.” He could have told me how critical body posture is when people swallow pills.
Who’s Duke?
Duke is a computer representation of a 34-year-old, anatomically normal human male created by computer scientists at the IT’IS Foundation, a nonprofit group based in Switzerland that works on a variety of projects in health care technology. Using Duke, Rajat Mittal, PhD, a professor of medicine at the Johns Hopkins University, Baltimore, created a computer model called “StomachSim” to explore the process of digestion.
Their research, published in the journal Physics of Fluids, turned up several surprising findings about the dynamics of swallowing pills – the most common way medication is used worldwide.
Dr. Mittal said he chose to study the stomach because the functions of most other organ systems, from the heart to the brain, have already attracted plenty of attention from scientists.
“As I was looking to initiate research in some new directions, the implications of stomach biomechanics on important conditions such as diabetes, obesity, and gastroparesis became apparent to me,” he said. “It was clear that bioengineering research in this arena lags other more ‘sexy’ areas such as cardiovascular flows by at least 20 years, and there seemed to be a great opportunity to do impactful work.”
Your posture may help a pill work better
Several well-known things affect a pill’s ability to disperse its contents into the gut and be used by the body, such as the stomach’s contents (a heavy breakfast, a mix of liquids like juice, milk, and coffee) and the motion of the organ’s walls. But Dr. Mittal’s group learned that Duke’s posture also played a major role.
The researchers ran Duke through computer simulations in varying postures: upright, leaning right, leaning left, and leaning back, while keeping all the other parts of their analyses (like the things mentioned above) the same.
They found that posture determined as much as 83% of how quickly a pill disperses into the intestines. The most efficient position was leaning right. The least was leaning left, which prevented the pill from reaching the antrum, or bottom section of the stomach, and thus kept all but traces of the dissolved drug from entering the duodenum, where the stomach joins the small intestine. (Interestingly, Jews who observe Passover are advised to recline to the left during the meal as a symbol of freedom and leisure.)
That makes sense if you think about the stomach’s shape, which looks kind of like a bean, curving from the left to the right side of the body. Because of gravity, your position will change where the pill lands.
a condition in which the stomach loses the ability to empty properly.
How this could help people
Among the groups most likely to benefit from such studies, Dr. Mittal said, are the elderly – who both take a lot of pills and are more prone to trouble swallowing because of age-related changes in their esophagus – and the bedridden, who can’t easily shift their posture. The findings may also lead to improvements in the ability to treat people with gastroparesis, a particular problem for people with diabetes.
Future studies with Duke and similar simulations will look at how the GI system digests proteins, carbohydrates, and fatty meals, Dr. Mittal said.
In the meantime, Dr. Mittal offered the following advice: “Standing or sitting upright after taking a pill is fine. If you have to take a pill lying down, stay on your back or on your right side. Avoid lying on your left side after taking a pill.”
As for what happened to me, any gastroenterologist reading this has figured out that my condition was not heart-related. Instead, I likely was having a bout of pill esophagitis, irritation that can result from medications that aggravate the mucosa of the food tube. Although painful, esophagitis isn’t life-threatening. After about an hour, the pain began to subside, and by the next morning I was fine, with only a faint ache in my chest to remind me of my earlier torment. (Researchers noted an increase in the condition early in the COVID-19 pandemic, linked to the antibiotic doxycycline.)
And, in the interest of accuracy, my pill problem began above the stomach. Nothing in the Hopkins research suggests that the alignment of the esophagus plays a role in how drugs disperse in the gut – unless, of course, it prevents those pills from reaching the stomach in the first place.
A version of this article first appeared on WebMD.com.
I want to tell you a story about forgetfulness and haste, and how the combination of the two can lead to frightening consequences. A few years ago, I was lying in bed about to turn out the light when I realized I’d forgotten to take “my pill.”
Like some 161 million other American adults, I was then a consumer of a prescription medication. Being conscientious, I got up, retrieved said pill, and tossed it back. Being lazy, I didn’t bother to grab a glass of water to help the thing go down. Instead, I promptly returned to bed, threw a pillow over my head, and prepared for sleep.
Within seconds, I began to feel a burning sensation in my chest. After about a minute, that burn became a crippling pain. Not wanting to alarm my wife, I went into the living room, where I spent the next 30 minutes doubled over in agony. Was I having a heart attack? I phoned my sister, a hospitalist in Texas. She advised me to take myself to the ED to get checked out.
If only I’d known then about “Duke.” He could have told me how critical body posture is when people swallow pills.
Who’s Duke?
Duke is a computer representation of a 34-year-old, anatomically normal human male created by computer scientists at the IT’IS Foundation, a nonprofit group based in Switzerland that works on a variety of projects in health care technology. Using Duke, Rajat Mittal, PhD, a professor of medicine at the Johns Hopkins University, Baltimore, created a computer model called “StomachSim” to explore the process of digestion.
Their research, published in the journal Physics of Fluids, turned up several surprising findings about the dynamics of swallowing pills – the most common way medication is used worldwide.
Dr. Mittal said he chose to study the stomach because the functions of most other organ systems, from the heart to the brain, have already attracted plenty of attention from scientists.
“As I was looking to initiate research in some new directions, the implications of stomach biomechanics on important conditions such as diabetes, obesity, and gastroparesis became apparent to me,” he said. “It was clear that bioengineering research in this arena lags other more ‘sexy’ areas such as cardiovascular flows by at least 20 years, and there seemed to be a great opportunity to do impactful work.”
Your posture may help a pill work better
Several well-known things affect a pill’s ability to disperse its contents into the gut and be used by the body, such as the stomach’s contents (a heavy breakfast, a mix of liquids like juice, milk, and coffee) and the motion of the organ’s walls. But Dr. Mittal’s group learned that Duke’s posture also played a major role.
The researchers ran Duke through computer simulations in varying postures: upright, leaning right, leaning left, and leaning back, while keeping all the other parts of their analyses (like the things mentioned above) the same.
They found that posture determined as much as 83% of how quickly a pill disperses into the intestines. The most efficient position was leaning right. The least was leaning left, which prevented the pill from reaching the antrum, or bottom section of the stomach, and thus kept all but traces of the dissolved drug from entering the duodenum, where the stomach joins the small intestine. (Interestingly, Jews who observe Passover are advised to recline to the left during the meal as a symbol of freedom and leisure.)
That makes sense if you think about the stomach’s shape, which looks kind of like a bean, curving from the left to the right side of the body. Because of gravity, your position will change where the pill lands.
a condition in which the stomach loses the ability to empty properly.
How this could help people
Among the groups most likely to benefit from such studies, Dr. Mittal said, are the elderly – who both take a lot of pills and are more prone to trouble swallowing because of age-related changes in their esophagus – and the bedridden, who can’t easily shift their posture. The findings may also lead to improvements in the ability to treat people with gastroparesis, a particular problem for people with diabetes.
Future studies with Duke and similar simulations will look at how the GI system digests proteins, carbohydrates, and fatty meals, Dr. Mittal said.
In the meantime, Dr. Mittal offered the following advice: “Standing or sitting upright after taking a pill is fine. If you have to take a pill lying down, stay on your back or on your right side. Avoid lying on your left side after taking a pill.”
As for what happened to me, any gastroenterologist reading this has figured out that my condition was not heart-related. Instead, I likely was having a bout of pill esophagitis, irritation that can result from medications that aggravate the mucosa of the food tube. Although painful, esophagitis isn’t life-threatening. After about an hour, the pain began to subside, and by the next morning I was fine, with only a faint ache in my chest to remind me of my earlier torment. (Researchers noted an increase in the condition early in the COVID-19 pandemic, linked to the antibiotic doxycycline.)
And, in the interest of accuracy, my pill problem began above the stomach. Nothing in the Hopkins research suggests that the alignment of the esophagus plays a role in how drugs disperse in the gut – unless, of course, it prevents those pills from reaching the stomach in the first place.
A version of this article first appeared on WebMD.com.
Moderate-to-severe atopic dermatitis: Astegolimab fails to reduce disease severity in phase 2 trial
Key clinical point: Astegolimab, despite being well-tolerated, did not lessen the severity of the disease in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: At week 16, the adjusted mean percent changes in the mean Eczema Area and Severity Index score were comparable in the astegolimab (−51.47%) and placebo (−58.24%) groups (
Study details: Findings are from a phase 2 trial including 65 adult patients with moderate-to-severe AD and inadequate response to topical medications who were randomly assigned to receive 490 mg astegolimab or placebo every 4 weeks for 16 weeks and were further followed-up for 8 weeks.
Disclosures: This study was supported by Genentech, Inc. Six authors declared serving as employees of Genentech, Inc., a member of the Roche group, and owning stocks in Roche. The other authors reported ties with several sources, including Roche.
Source: Maurer M et al. Phase 2 randomized clinical trial of astegolimab in patients with moderate to severe atopic dermatitis. J Allergy Clin Immunol. 2022 (Aug 27). Doi: 10.1016/j.jaci.2022.08.015
Key clinical point: Astegolimab, despite being well-tolerated, did not lessen the severity of the disease in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: At week 16, the adjusted mean percent changes in the mean Eczema Area and Severity Index score were comparable in the astegolimab (−51.47%) and placebo (−58.24%) groups (
Study details: Findings are from a phase 2 trial including 65 adult patients with moderate-to-severe AD and inadequate response to topical medications who were randomly assigned to receive 490 mg astegolimab or placebo every 4 weeks for 16 weeks and were further followed-up for 8 weeks.
Disclosures: This study was supported by Genentech, Inc. Six authors declared serving as employees of Genentech, Inc., a member of the Roche group, and owning stocks in Roche. The other authors reported ties with several sources, including Roche.
Source: Maurer M et al. Phase 2 randomized clinical trial of astegolimab in patients with moderate to severe atopic dermatitis. J Allergy Clin Immunol. 2022 (Aug 27). Doi: 10.1016/j.jaci.2022.08.015
Key clinical point: Astegolimab, despite being well-tolerated, did not lessen the severity of the disease in patients with moderate-to-severe atopic dermatitis (AD).
Major finding: At week 16, the adjusted mean percent changes in the mean Eczema Area and Severity Index score were comparable in the astegolimab (−51.47%) and placebo (−58.24%) groups (
Study details: Findings are from a phase 2 trial including 65 adult patients with moderate-to-severe AD and inadequate response to topical medications who were randomly assigned to receive 490 mg astegolimab or placebo every 4 weeks for 16 weeks and were further followed-up for 8 weeks.
Disclosures: This study was supported by Genentech, Inc. Six authors declared serving as employees of Genentech, Inc., a member of the Roche group, and owning stocks in Roche. The other authors reported ties with several sources, including Roche.
Source: Maurer M et al. Phase 2 randomized clinical trial of astegolimab in patients with moderate to severe atopic dermatitis. J Allergy Clin Immunol. 2022 (Aug 27). Doi: 10.1016/j.jaci.2022.08.015
EHA cream shows promise in children with mild-to-moderate atopic dermatitis
Key clinical point: A cream containing 1% ectoine and 0.1% hyaluronic acid (EHA) demonstrated superior efficacy than a vehicle cream and was well-tolerated in children aged 2-18 years with mild-to-moderate atopic dermatitis (AD).
Major finding: At week 4, patients receiving EHA cream achieved a significantly higher clinical improvement in SCORing AD (mean difference [MD] −6.62; P < .001) and Investigator’s Global Assessment scores (MD −0.69; P < .001) than those receiving vehicle cream. Mild adverse events like skin erythema, pruritus, and burning skin were reported by 23.5% of patients receiving EHA cream and 5.7% of patients receiving vehicle cream.
Study details: Findings are from an observer-blind, multicenter clinical trial including 57 children aged 2-18 years with mild-to-moderate AD who were randomly assigned to receive EHA or vehicle cream twice daily for 4 weeks.
Disclosures: This study was funded by BODERM SA. The authors declared no conflicts of interest.
Source: Alexopoulos A et al. A randomized, observer-blind, vehicle-control, multi-center clinical investigation for assessing the efficacy and tolerability of a 1% ectoine and hyaluronic acid 0.1%-containing medical device in pediatric patients with mild-to-moderate atopic dermatitis. Pediatr Dermatol. 2022 (Aug 29). Doi: 10.1111/pde.15117
Key clinical point: A cream containing 1% ectoine and 0.1% hyaluronic acid (EHA) demonstrated superior efficacy than a vehicle cream and was well-tolerated in children aged 2-18 years with mild-to-moderate atopic dermatitis (AD).
Major finding: At week 4, patients receiving EHA cream achieved a significantly higher clinical improvement in SCORing AD (mean difference [MD] −6.62; P < .001) and Investigator’s Global Assessment scores (MD −0.69; P < .001) than those receiving vehicle cream. Mild adverse events like skin erythema, pruritus, and burning skin were reported by 23.5% of patients receiving EHA cream and 5.7% of patients receiving vehicle cream.
Study details: Findings are from an observer-blind, multicenter clinical trial including 57 children aged 2-18 years with mild-to-moderate AD who were randomly assigned to receive EHA or vehicle cream twice daily for 4 weeks.
Disclosures: This study was funded by BODERM SA. The authors declared no conflicts of interest.
Source: Alexopoulos A et al. A randomized, observer-blind, vehicle-control, multi-center clinical investigation for assessing the efficacy and tolerability of a 1% ectoine and hyaluronic acid 0.1%-containing medical device in pediatric patients with mild-to-moderate atopic dermatitis. Pediatr Dermatol. 2022 (Aug 29). Doi: 10.1111/pde.15117
Key clinical point: A cream containing 1% ectoine and 0.1% hyaluronic acid (EHA) demonstrated superior efficacy than a vehicle cream and was well-tolerated in children aged 2-18 years with mild-to-moderate atopic dermatitis (AD).
Major finding: At week 4, patients receiving EHA cream achieved a significantly higher clinical improvement in SCORing AD (mean difference [MD] −6.62; P < .001) and Investigator’s Global Assessment scores (MD −0.69; P < .001) than those receiving vehicle cream. Mild adverse events like skin erythema, pruritus, and burning skin were reported by 23.5% of patients receiving EHA cream and 5.7% of patients receiving vehicle cream.
Study details: Findings are from an observer-blind, multicenter clinical trial including 57 children aged 2-18 years with mild-to-moderate AD who were randomly assigned to receive EHA or vehicle cream twice daily for 4 weeks.
Disclosures: This study was funded by BODERM SA. The authors declared no conflicts of interest.
Source: Alexopoulos A et al. A randomized, observer-blind, vehicle-control, multi-center clinical investigation for assessing the efficacy and tolerability of a 1% ectoine and hyaluronic acid 0.1%-containing medical device in pediatric patients with mild-to-moderate atopic dermatitis. Pediatr Dermatol. 2022 (Aug 29). Doi: 10.1111/pde.15117