User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
AI tool perfect in study of inflammatory diseases
Artificial intelligence can distinguish overlapping inflammatory conditions with total accuracy, according to a new study presented at the annual meeting of the American College of Rheumatology.
Texas pediatricians faced a conundrum during the pandemic. Endemic typhus, a flea-borne tropical infection common to the region, is nearly indistinguishable from multisystem inflammatory syndrome in children (MIS-C), a rare condition set in motion by SARS-CoV-2 infection. Children with either ailment had seemingly identical symptoms: fever, rash, gastrointestinal issues, and in need of swift treatment. A diagnosis of endemic typhus can take 4-6 days to confirm.
Tiphanie Vogel, MD, PhD, a pediatric rheumatologist at Texas Children’s Hospital, Houston, and colleagues sought to create a tool to hasten diagnosis and, ideally, treatment. To do so, they incorporated machine learning and clinical factors available within the first 6 hours of the onset of symptoms.
The team analyzed 49 demographic, clinical, and laboratory measures from the medical records of 133 children with MIS-C and 87 with endemic typhus. Using deep learning, they narrowed the model to 30 essential features that became the backbone of AI-MET, a two-phase clinical-decision support system.
Phase 1 uses 17 clinical factors and can be performed on paper. If a patient’s score in phase 1 is not determinative, clinicians proceed to phase 2, which uses an additional 13 weighted factors and machine learning.
In testing, the two-part tool classified each of the 220 test patients perfectly. And it diagnosed a second group of 111 patients with MIS-C with 99% (110/111) accuracy.
Of note, “that first step classifies [a patient] correctly half of the time,” Dr. Vogel said, so the second, AI phase of the tool was necessary for only half of cases. Dr. Vogel said that’s a good sign; it means that the tool is useful in settings where AI may not always be feasible, like in a busy ED.
Melissa Mizesko, MD, a pediatric rheumatologist at Driscoll Children’s Hospital in Corpus Christi, Tex., said that the new tool could help clinicians streamline care. When cases of MIS-C peaked in Texas, clinicians often would start sick children on doxycycline and treat for MIS-C at the same time, then wait to see whether the antibiotic brought the fever down.
“This [new tool] is helpful if you live in a part of the country that has typhus,” said Jane Burns, MD, director of the Kawasaki Disease Research Center at the University of California, San Diego, who helped develop a similar AI-based tool to distinguish MIS-C from Kawasaki disease. But she encouraged the researchers to expand their testing to include other conditions. Although the AI model Dr. Vogel’s group developed can pinpoint MIS-C or endemic typhus, what if a child has neither condition? “It’s not often you’re dealing with a diagnosis between just two specific diseases,” Dr. Burns said.
Dr. Vogel is also interested in making AI-MET more efficient. “This go-round we prioritized perfect accuracy,” she said. But 30 clinical factors, with 17 of them recorded and calculated by hand, is a lot. “Could we still get this to be very accurate, maybe not perfect, with less inputs?”
In addition to refining AI-MET, which Texas Children’s eventually hopes to make available to other institutions, Dr. Vogel and associates are also considering other use cases for AI. Lupus is one option. “Maybe with machine learning we could identify clues at diagnosis that would help recommend targeted treatment,” she said
Dr. Vogel disclosed potential conflicts of interest with Moderna, Novartis, Pfizer, and SOBI. Dr. Burns and Dr. Mizesko disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
Artificial intelligence can distinguish overlapping inflammatory conditions with total accuracy, according to a new study presented at the annual meeting of the American College of Rheumatology.
Texas pediatricians faced a conundrum during the pandemic. Endemic typhus, a flea-borne tropical infection common to the region, is nearly indistinguishable from multisystem inflammatory syndrome in children (MIS-C), a rare condition set in motion by SARS-CoV-2 infection. Children with either ailment had seemingly identical symptoms: fever, rash, gastrointestinal issues, and in need of swift treatment. A diagnosis of endemic typhus can take 4-6 days to confirm.
Tiphanie Vogel, MD, PhD, a pediatric rheumatologist at Texas Children’s Hospital, Houston, and colleagues sought to create a tool to hasten diagnosis and, ideally, treatment. To do so, they incorporated machine learning and clinical factors available within the first 6 hours of the onset of symptoms.
The team analyzed 49 demographic, clinical, and laboratory measures from the medical records of 133 children with MIS-C and 87 with endemic typhus. Using deep learning, they narrowed the model to 30 essential features that became the backbone of AI-MET, a two-phase clinical-decision support system.
Phase 1 uses 17 clinical factors and can be performed on paper. If a patient’s score in phase 1 is not determinative, clinicians proceed to phase 2, which uses an additional 13 weighted factors and machine learning.
In testing, the two-part tool classified each of the 220 test patients perfectly. And it diagnosed a second group of 111 patients with MIS-C with 99% (110/111) accuracy.
Of note, “that first step classifies [a patient] correctly half of the time,” Dr. Vogel said, so the second, AI phase of the tool was necessary for only half of cases. Dr. Vogel said that’s a good sign; it means that the tool is useful in settings where AI may not always be feasible, like in a busy ED.
Melissa Mizesko, MD, a pediatric rheumatologist at Driscoll Children’s Hospital in Corpus Christi, Tex., said that the new tool could help clinicians streamline care. When cases of MIS-C peaked in Texas, clinicians often would start sick children on doxycycline and treat for MIS-C at the same time, then wait to see whether the antibiotic brought the fever down.
“This [new tool] is helpful if you live in a part of the country that has typhus,” said Jane Burns, MD, director of the Kawasaki Disease Research Center at the University of California, San Diego, who helped develop a similar AI-based tool to distinguish MIS-C from Kawasaki disease. But she encouraged the researchers to expand their testing to include other conditions. Although the AI model Dr. Vogel’s group developed can pinpoint MIS-C or endemic typhus, what if a child has neither condition? “It’s not often you’re dealing with a diagnosis between just two specific diseases,” Dr. Burns said.
Dr. Vogel is also interested in making AI-MET more efficient. “This go-round we prioritized perfect accuracy,” she said. But 30 clinical factors, with 17 of them recorded and calculated by hand, is a lot. “Could we still get this to be very accurate, maybe not perfect, with less inputs?”
In addition to refining AI-MET, which Texas Children’s eventually hopes to make available to other institutions, Dr. Vogel and associates are also considering other use cases for AI. Lupus is one option. “Maybe with machine learning we could identify clues at diagnosis that would help recommend targeted treatment,” she said
Dr. Vogel disclosed potential conflicts of interest with Moderna, Novartis, Pfizer, and SOBI. Dr. Burns and Dr. Mizesko disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
Artificial intelligence can distinguish overlapping inflammatory conditions with total accuracy, according to a new study presented at the annual meeting of the American College of Rheumatology.
Texas pediatricians faced a conundrum during the pandemic. Endemic typhus, a flea-borne tropical infection common to the region, is nearly indistinguishable from multisystem inflammatory syndrome in children (MIS-C), a rare condition set in motion by SARS-CoV-2 infection. Children with either ailment had seemingly identical symptoms: fever, rash, gastrointestinal issues, and in need of swift treatment. A diagnosis of endemic typhus can take 4-6 days to confirm.
Tiphanie Vogel, MD, PhD, a pediatric rheumatologist at Texas Children’s Hospital, Houston, and colleagues sought to create a tool to hasten diagnosis and, ideally, treatment. To do so, they incorporated machine learning and clinical factors available within the first 6 hours of the onset of symptoms.
The team analyzed 49 demographic, clinical, and laboratory measures from the medical records of 133 children with MIS-C and 87 with endemic typhus. Using deep learning, they narrowed the model to 30 essential features that became the backbone of AI-MET, a two-phase clinical-decision support system.
Phase 1 uses 17 clinical factors and can be performed on paper. If a patient’s score in phase 1 is not determinative, clinicians proceed to phase 2, which uses an additional 13 weighted factors and machine learning.
In testing, the two-part tool classified each of the 220 test patients perfectly. And it diagnosed a second group of 111 patients with MIS-C with 99% (110/111) accuracy.
Of note, “that first step classifies [a patient] correctly half of the time,” Dr. Vogel said, so the second, AI phase of the tool was necessary for only half of cases. Dr. Vogel said that’s a good sign; it means that the tool is useful in settings where AI may not always be feasible, like in a busy ED.
Melissa Mizesko, MD, a pediatric rheumatologist at Driscoll Children’s Hospital in Corpus Christi, Tex., said that the new tool could help clinicians streamline care. When cases of MIS-C peaked in Texas, clinicians often would start sick children on doxycycline and treat for MIS-C at the same time, then wait to see whether the antibiotic brought the fever down.
“This [new tool] is helpful if you live in a part of the country that has typhus,” said Jane Burns, MD, director of the Kawasaki Disease Research Center at the University of California, San Diego, who helped develop a similar AI-based tool to distinguish MIS-C from Kawasaki disease. But she encouraged the researchers to expand their testing to include other conditions. Although the AI model Dr. Vogel’s group developed can pinpoint MIS-C or endemic typhus, what if a child has neither condition? “It’s not often you’re dealing with a diagnosis between just two specific diseases,” Dr. Burns said.
Dr. Vogel is also interested in making AI-MET more efficient. “This go-round we prioritized perfect accuracy,” she said. But 30 clinical factors, with 17 of them recorded and calculated by hand, is a lot. “Could we still get this to be very accurate, maybe not perfect, with less inputs?”
In addition to refining AI-MET, which Texas Children’s eventually hopes to make available to other institutions, Dr. Vogel and associates are also considering other use cases for AI. Lupus is one option. “Maybe with machine learning we could identify clues at diagnosis that would help recommend targeted treatment,” she said
Dr. Vogel disclosed potential conflicts of interest with Moderna, Novartis, Pfizer, and SOBI. Dr. Burns and Dr. Mizesko disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM ACR 2023
Classification identifies four stages of heart attack
Relying on more than 50 years of data on acute MI with reperfusion therapy, the society has identified the following four stages of progressively worsening myocardial tissue injury:
- Aborted MI (no or minimal myocardial necrosis).
- MI with significant cardiomyocyte necrosis but without microvascular injury.
- Cardiomyocyte necrosis and microvascular dysfunction leading to microvascular obstruction (that is, “no reflow”).
- Cardiomyocyte and microvascular necrosis leading to reperfusion hemorrhage.
The classification is described in an expert consensus statement that was published in the Canadian Journal of Cardiology.
The new classification will allow for better risk stratification and more appropriate treatment and provide refined endpoints for clinical trials and translational research, according to the authors.
Currently, all patients with acute MI receive the same treatment, even though they may have different levels of tissue injury severity, statement author Andreas Kumar, MD, chair of the writing group and associate professor of medicine at Northern Ontario School of Medicine University, Sudbury, said in an interview.
“In some cases, treatment for a mild stage 1 acute MI may be deadly for someone with stage 4 hemorrhagic MI,” said Dr. Kumar.
Technological advances
The classification is based on decades of data. “The initial data were obtained with pathology studies in the 1970s. When cardiac MRI came around, around the year 2000, suddenly there was a noninvasive imaging method where we could investigate patients in vivo,” said Dr. Kumar. “We learned a lot about tissue changes in acute MI. And especially in the last 2 to 5 years, we have learned a lot about hemorrhagic MI. So, this then gave us enough knowledge to come up with this new classification.”
The idea of classifying acute MI came to Dr. Kumar and senior author Rohan Dharmakumar, PhD, executive director of the Krannert Cardiovascular Research Center at Indiana University, Indianapolis, when both were at the University of Toronto.
“This work has been years in the making,” Dr. Dharmakumar said in an interview. “We’ve been thinking about this for a long time, but we needed to get substantial layers of evidence to support the classification. We had a feeling about these stages for a long time, but that feeling needed to be substantiated.”
In 2022, Dr. Dharmakumar and Dr. Kumar observed that damage to the heart from MI was not only a result of ischemia caused by a blocked artery, but also a result of bleeding in the myocardium after the artery had been opened. Their findings were published in the Journal of the American College of Cardiology.
The author of an accompanying editorial lauded the investigators “for providing new, mechanistic insights into a difficult clinical problem that has an unmet therapeutic need.”
“Hemorrhagic MI is a very dangerous injury because hemorrhage itself causes a lot of problems,” said Dr. Kumar. “We reported that there is infarct expansion after reperfusion, so once you open up the vessel, the heart attack actually gets larger. We also showed that the remodeling of these hearts is worse. These patients take a second hit with hemorrhage occurring in the myocardium.”
Classification and staging
“The standard guideline therapy for somebody who comes into the hospital is to put in a stent, open the artery, have the patient stay in the hospital for 48-72 hours, and then be released home,” said Dr. Dharmukumar. “But here’s the problem. These two patients who are going back home have different levels of injury, yet they are taking the same medications. Even inside the hospital, we have heterogeneity in mortality risk. But we are not paying attention to one patient differently than the other, even though we should, because their injuries are very different.”
The CCS classification may provide endpoints and outcome measures beyond the commonly used clinical markers, which could lead to improved treatments to help patients recover from their cardiac events.
“We have this issue of rampant heart failure in acute MI survivors. We’ve gotten really good at saving patients from immediate death, but now we are just postponing some of the serious problems survivors are going to face, said Dr. Dharmukumar. “What are we doing for these patients who are really at risk? We’ve been treating every single patient the same way and we have not been paying attention to the very different stages of injury.”
In an accompanying editorial, Prakriti Gaba, MD, a clinical fellow in medicine at Brigham and Women’s Hospital, Boston, and Deepak L. Bhatt, MD, MPH, director of the Mount Sinai Fuster Heart Hospital, New York, wrote: “There is no doubt that the classification system proposed by the investigators is important and timely, as acute MI continues to account for substantial morbidity and mortality worldwide.”
Imaging and staging could be useful in guiding appropriate therapy, Bhatt said in an interview. “The authors’ hope, which I think is a very laudable one, is that more finely characterizing exactly what the extent of damage is and what the mechanism of damage is in a heart attack will make it possible to develop therapies that are particularly targeted to each of the stages,” he said.
“It is quite common to have the ability to do cardiac MRI at experienced cardiovascular centers, although this may not be true for smaller community hospitals,” Dr. Bhatt added. “But at least at larger hospitals, this will allow for much finer evaluation and assessment of exactly what is going on in that particular patient and how extensive the heart muscle damage is. Eventually, this will facilitate the development of therapies that are specifically targeted to treat each stage.”
Dr. Kumar is partly supported by a research grant from the Northern Ontario Academic Medicine Association. Dr. Dharmakumar was funded in part by grants from the U.S. National Institutes of Health. Dr. Dharmakumar has an ownership interest in Cardio-Theranostics. Dr. Bhatt has served on advisory boards for Angiowave, Bayer, Boehringer Ingelheim, Cardax, CellProthera, Cereno Scientific, Elsevier Practice Update Cardiology, High Enroll, Janssen, Level Ex, McKinsey, Medscape Cardiology, Merck, MyoKardia, NirvaMed, Novo Nordisk, PhaseBio, PLx Pharma, Regado Biosciences, and Stasys. He is a member of the board of directors of or holds stock in Angiowave, Boston VA Research Institute, Bristol-Myers Squibb, DRS.LINQ, High Enroll, Society of Cardiovascular Patient Care, and TobeSoft. He has worked as a consultant for Broadview Ventures, and Hims. He has received honoraria from the American College of Cardiology, Arnold and Porter law firm, Baim Institute for Clinical Research, Belvoir Publications, Canadian Medical and Surgical Knowledge Translation Research Group, Cowen and Company, Duke Clinical Research Institute, HMP Global, Journal of the American College of Cardiology, K2P, Level Ex, Medtelligence/ReachMD, MJH Life Sciences, Oakstone CME, Piper Sandler, Population Health Research Institute, Slack Publications, Society of Cardiovascular Patient Care, WebMD, and Wiley.
A version of this article appeared on Medscape.com.
Relying on more than 50 years of data on acute MI with reperfusion therapy, the society has identified the following four stages of progressively worsening myocardial tissue injury:
- Aborted MI (no or minimal myocardial necrosis).
- MI with significant cardiomyocyte necrosis but without microvascular injury.
- Cardiomyocyte necrosis and microvascular dysfunction leading to microvascular obstruction (that is, “no reflow”).
- Cardiomyocyte and microvascular necrosis leading to reperfusion hemorrhage.
The classification is described in an expert consensus statement that was published in the Canadian Journal of Cardiology.
The new classification will allow for better risk stratification and more appropriate treatment and provide refined endpoints for clinical trials and translational research, according to the authors.
Currently, all patients with acute MI receive the same treatment, even though they may have different levels of tissue injury severity, statement author Andreas Kumar, MD, chair of the writing group and associate professor of medicine at Northern Ontario School of Medicine University, Sudbury, said in an interview.
“In some cases, treatment for a mild stage 1 acute MI may be deadly for someone with stage 4 hemorrhagic MI,” said Dr. Kumar.
Technological advances
The classification is based on decades of data. “The initial data were obtained with pathology studies in the 1970s. When cardiac MRI came around, around the year 2000, suddenly there was a noninvasive imaging method where we could investigate patients in vivo,” said Dr. Kumar. “We learned a lot about tissue changes in acute MI. And especially in the last 2 to 5 years, we have learned a lot about hemorrhagic MI. So, this then gave us enough knowledge to come up with this new classification.”
The idea of classifying acute MI came to Dr. Kumar and senior author Rohan Dharmakumar, PhD, executive director of the Krannert Cardiovascular Research Center at Indiana University, Indianapolis, when both were at the University of Toronto.
“This work has been years in the making,” Dr. Dharmakumar said in an interview. “We’ve been thinking about this for a long time, but we needed to get substantial layers of evidence to support the classification. We had a feeling about these stages for a long time, but that feeling needed to be substantiated.”
In 2022, Dr. Dharmakumar and Dr. Kumar observed that damage to the heart from MI was not only a result of ischemia caused by a blocked artery, but also a result of bleeding in the myocardium after the artery had been opened. Their findings were published in the Journal of the American College of Cardiology.
The author of an accompanying editorial lauded the investigators “for providing new, mechanistic insights into a difficult clinical problem that has an unmet therapeutic need.”
“Hemorrhagic MI is a very dangerous injury because hemorrhage itself causes a lot of problems,” said Dr. Kumar. “We reported that there is infarct expansion after reperfusion, so once you open up the vessel, the heart attack actually gets larger. We also showed that the remodeling of these hearts is worse. These patients take a second hit with hemorrhage occurring in the myocardium.”
Classification and staging
“The standard guideline therapy for somebody who comes into the hospital is to put in a stent, open the artery, have the patient stay in the hospital for 48-72 hours, and then be released home,” said Dr. Dharmukumar. “But here’s the problem. These two patients who are going back home have different levels of injury, yet they are taking the same medications. Even inside the hospital, we have heterogeneity in mortality risk. But we are not paying attention to one patient differently than the other, even though we should, because their injuries are very different.”
The CCS classification may provide endpoints and outcome measures beyond the commonly used clinical markers, which could lead to improved treatments to help patients recover from their cardiac events.
“We have this issue of rampant heart failure in acute MI survivors. We’ve gotten really good at saving patients from immediate death, but now we are just postponing some of the serious problems survivors are going to face, said Dr. Dharmukumar. “What are we doing for these patients who are really at risk? We’ve been treating every single patient the same way and we have not been paying attention to the very different stages of injury.”
In an accompanying editorial, Prakriti Gaba, MD, a clinical fellow in medicine at Brigham and Women’s Hospital, Boston, and Deepak L. Bhatt, MD, MPH, director of the Mount Sinai Fuster Heart Hospital, New York, wrote: “There is no doubt that the classification system proposed by the investigators is important and timely, as acute MI continues to account for substantial morbidity and mortality worldwide.”
Imaging and staging could be useful in guiding appropriate therapy, Bhatt said in an interview. “The authors’ hope, which I think is a very laudable one, is that more finely characterizing exactly what the extent of damage is and what the mechanism of damage is in a heart attack will make it possible to develop therapies that are particularly targeted to each of the stages,” he said.
“It is quite common to have the ability to do cardiac MRI at experienced cardiovascular centers, although this may not be true for smaller community hospitals,” Dr. Bhatt added. “But at least at larger hospitals, this will allow for much finer evaluation and assessment of exactly what is going on in that particular patient and how extensive the heart muscle damage is. Eventually, this will facilitate the development of therapies that are specifically targeted to treat each stage.”
Dr. Kumar is partly supported by a research grant from the Northern Ontario Academic Medicine Association. Dr. Dharmakumar was funded in part by grants from the U.S. National Institutes of Health. Dr. Dharmakumar has an ownership interest in Cardio-Theranostics. Dr. Bhatt has served on advisory boards for Angiowave, Bayer, Boehringer Ingelheim, Cardax, CellProthera, Cereno Scientific, Elsevier Practice Update Cardiology, High Enroll, Janssen, Level Ex, McKinsey, Medscape Cardiology, Merck, MyoKardia, NirvaMed, Novo Nordisk, PhaseBio, PLx Pharma, Regado Biosciences, and Stasys. He is a member of the board of directors of or holds stock in Angiowave, Boston VA Research Institute, Bristol-Myers Squibb, DRS.LINQ, High Enroll, Society of Cardiovascular Patient Care, and TobeSoft. He has worked as a consultant for Broadview Ventures, and Hims. He has received honoraria from the American College of Cardiology, Arnold and Porter law firm, Baim Institute for Clinical Research, Belvoir Publications, Canadian Medical and Surgical Knowledge Translation Research Group, Cowen and Company, Duke Clinical Research Institute, HMP Global, Journal of the American College of Cardiology, K2P, Level Ex, Medtelligence/ReachMD, MJH Life Sciences, Oakstone CME, Piper Sandler, Population Health Research Institute, Slack Publications, Society of Cardiovascular Patient Care, WebMD, and Wiley.
A version of this article appeared on Medscape.com.
Relying on more than 50 years of data on acute MI with reperfusion therapy, the society has identified the following four stages of progressively worsening myocardial tissue injury:
- Aborted MI (no or minimal myocardial necrosis).
- MI with significant cardiomyocyte necrosis but without microvascular injury.
- Cardiomyocyte necrosis and microvascular dysfunction leading to microvascular obstruction (that is, “no reflow”).
- Cardiomyocyte and microvascular necrosis leading to reperfusion hemorrhage.
The classification is described in an expert consensus statement that was published in the Canadian Journal of Cardiology.
The new classification will allow for better risk stratification and more appropriate treatment and provide refined endpoints for clinical trials and translational research, according to the authors.
Currently, all patients with acute MI receive the same treatment, even though they may have different levels of tissue injury severity, statement author Andreas Kumar, MD, chair of the writing group and associate professor of medicine at Northern Ontario School of Medicine University, Sudbury, said in an interview.
“In some cases, treatment for a mild stage 1 acute MI may be deadly for someone with stage 4 hemorrhagic MI,” said Dr. Kumar.
Technological advances
The classification is based on decades of data. “The initial data were obtained with pathology studies in the 1970s. When cardiac MRI came around, around the year 2000, suddenly there was a noninvasive imaging method where we could investigate patients in vivo,” said Dr. Kumar. “We learned a lot about tissue changes in acute MI. And especially in the last 2 to 5 years, we have learned a lot about hemorrhagic MI. So, this then gave us enough knowledge to come up with this new classification.”
The idea of classifying acute MI came to Dr. Kumar and senior author Rohan Dharmakumar, PhD, executive director of the Krannert Cardiovascular Research Center at Indiana University, Indianapolis, when both were at the University of Toronto.
“This work has been years in the making,” Dr. Dharmakumar said in an interview. “We’ve been thinking about this for a long time, but we needed to get substantial layers of evidence to support the classification. We had a feeling about these stages for a long time, but that feeling needed to be substantiated.”
In 2022, Dr. Dharmakumar and Dr. Kumar observed that damage to the heart from MI was not only a result of ischemia caused by a blocked artery, but also a result of bleeding in the myocardium after the artery had been opened. Their findings were published in the Journal of the American College of Cardiology.
The author of an accompanying editorial lauded the investigators “for providing new, mechanistic insights into a difficult clinical problem that has an unmet therapeutic need.”
“Hemorrhagic MI is a very dangerous injury because hemorrhage itself causes a lot of problems,” said Dr. Kumar. “We reported that there is infarct expansion after reperfusion, so once you open up the vessel, the heart attack actually gets larger. We also showed that the remodeling of these hearts is worse. These patients take a second hit with hemorrhage occurring in the myocardium.”
Classification and staging
“The standard guideline therapy for somebody who comes into the hospital is to put in a stent, open the artery, have the patient stay in the hospital for 48-72 hours, and then be released home,” said Dr. Dharmukumar. “But here’s the problem. These two patients who are going back home have different levels of injury, yet they are taking the same medications. Even inside the hospital, we have heterogeneity in mortality risk. But we are not paying attention to one patient differently than the other, even though we should, because their injuries are very different.”
The CCS classification may provide endpoints and outcome measures beyond the commonly used clinical markers, which could lead to improved treatments to help patients recover from their cardiac events.
“We have this issue of rampant heart failure in acute MI survivors. We’ve gotten really good at saving patients from immediate death, but now we are just postponing some of the serious problems survivors are going to face, said Dr. Dharmukumar. “What are we doing for these patients who are really at risk? We’ve been treating every single patient the same way and we have not been paying attention to the very different stages of injury.”
In an accompanying editorial, Prakriti Gaba, MD, a clinical fellow in medicine at Brigham and Women’s Hospital, Boston, and Deepak L. Bhatt, MD, MPH, director of the Mount Sinai Fuster Heart Hospital, New York, wrote: “There is no doubt that the classification system proposed by the investigators is important and timely, as acute MI continues to account for substantial morbidity and mortality worldwide.”
Imaging and staging could be useful in guiding appropriate therapy, Bhatt said in an interview. “The authors’ hope, which I think is a very laudable one, is that more finely characterizing exactly what the extent of damage is and what the mechanism of damage is in a heart attack will make it possible to develop therapies that are particularly targeted to each of the stages,” he said.
“It is quite common to have the ability to do cardiac MRI at experienced cardiovascular centers, although this may not be true for smaller community hospitals,” Dr. Bhatt added. “But at least at larger hospitals, this will allow for much finer evaluation and assessment of exactly what is going on in that particular patient and how extensive the heart muscle damage is. Eventually, this will facilitate the development of therapies that are specifically targeted to treat each stage.”
Dr. Kumar is partly supported by a research grant from the Northern Ontario Academic Medicine Association. Dr. Dharmakumar was funded in part by grants from the U.S. National Institutes of Health. Dr. Dharmakumar has an ownership interest in Cardio-Theranostics. Dr. Bhatt has served on advisory boards for Angiowave, Bayer, Boehringer Ingelheim, Cardax, CellProthera, Cereno Scientific, Elsevier Practice Update Cardiology, High Enroll, Janssen, Level Ex, McKinsey, Medscape Cardiology, Merck, MyoKardia, NirvaMed, Novo Nordisk, PhaseBio, PLx Pharma, Regado Biosciences, and Stasys. He is a member of the board of directors of or holds stock in Angiowave, Boston VA Research Institute, Bristol-Myers Squibb, DRS.LINQ, High Enroll, Society of Cardiovascular Patient Care, and TobeSoft. He has worked as a consultant for Broadview Ventures, and Hims. He has received honoraria from the American College of Cardiology, Arnold and Porter law firm, Baim Institute for Clinical Research, Belvoir Publications, Canadian Medical and Surgical Knowledge Translation Research Group, Cowen and Company, Duke Clinical Research Institute, HMP Global, Journal of the American College of Cardiology, K2P, Level Ex, Medtelligence/ReachMD, MJH Life Sciences, Oakstone CME, Piper Sandler, Population Health Research Institute, Slack Publications, Society of Cardiovascular Patient Care, WebMD, and Wiley.
A version of this article appeared on Medscape.com.
FROM THE CANADIAN JOURNAL OF CARDIOLOGY
Potential dapagliflozin benefit post MI is not a ‘mandate’
PHILADELPHIA – Giving the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) to patients with acute myocardial infarction and impaired left ventricular systolic function but no diabetes or chronic heart failure significantly improved a composite of cardiovascular outcomes, a European registry-based randomized trial suggests.
In presenting these results from the DAPA-MI trial, Stefan James, MD, of Uppsala University (Sweden), noted that which the trial described as the hierarchical “win ratio” composite outcomes, compared with patients randomized to placebo plus standard of care.
“The ‘win ratio’ tells us that there’s a 34% higher likelihood of patients having a better cardiometabolic outcome with dapagliflozin vs placebo in terms of the seven components,” James said in an interview. The win ratio was achieved in 32.9% of dapagliflozin patients versus 24.6% of placebo (P < .001).
Dr. James presented the results at the annual scientific sessions of the American Heart Association, and they were published online simultaneously in NEJM Evidence.
Lower-risk patients
DAPA-MI enrolled 4,017 patients from the SWEDEHEART and Myocardial Ischemia National Audit Project registries in Sweden and the United Kingdom, randomly assigning patients to dapagliflozin 10 mg or placebo along with guideline-directed therapy for both groups.
Eligible patients were hemodynamically stable, had an acute MI within 10 days of enrollment, and impaired left ventricular systolic function or a Q-wave MI. Exclusion criteria included history of either type 1 or 2 diabetes, chronic heart failure, poor kidney function, or current treatment with an SGLT2 inhibitor. Baseline demographic characteristics were similar between trial arms.
- The hierarchical seven primary endpoints were:
- Death, with cardiovascular death ranked first followed by noncardiovascular death
- Hospitalization because of heart failure, with adjudicated first followed by investigator-reported HF
- Nonfatal MI
- Atrial fibrillation/flutter event
- New diagnosis of type 2 diabetes
- New York Heart Association functional class at the last visit
- Drop in body weight of at least 5% at the last visit
The key secondary endpoint, Dr. James said, was the primary outcome minus the body weight component, with time to first occurrence of hospitalization for HF or cardiovascular death.
When the seventh factor, body weight decrease, was removed, the differential narrowed: 20.3% versus 16.9% (P = .015). When two or more variables were removed from the composite, the differences were not statistically significant.
For 11 secondary and exploratory outcomes, ranging from CV death or hospitalization for HF to all-cause hospitalization, the outcomes were similar in both the dapagliflozin and placebo groups across the board.
However, the dapagliflozin patients had about half the rate of developing diabetes, compared with the placebo group: 2.1 % versus 3.9%.
The trial initially used the composite of CV death and hospitalization for HF as the primary endpoint, but switched to the seven-item composite endpoint in February because the number of primary composite outcomes was substantially lower than anticipated, Dr. James said.
He acknowledged the study was underpowered for the low-risk population it enrolled. “But if you extended the trial to a larger population and enriched it with a higher-risk population you would probably see an effect,” he said.
“The cardiometabolic benefit was consistent across all prespecified subgroups and there were no new safety concerns,” Dr. James told the attendees. “Clinical event rates were low with no significant difference between randomized groups.”
Not a ringing endorsement
But for invited discussant Stephen D. Wiviott, MD, a cardiologist at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, the DAPA-MI trial result isn’t quite a ringing endorsement of SGLT2 inhibition in these patients.
“From my perspective, DAPA-MI does not suggest a new mandate to expand SGLT2 inhibition to an isolated MI population without other SGLT2 inhibitor indications,” Dr. Wiviott told attendees. “But it does support the safety of its use among patients with acute coronary syndromes.”
However, “these results do not indicate a lack of clinical benefit in patients with prior MI and any of those previously identified conditions – a history of diabetes, coronary heart failure or chronic kidney disease – where SGLT2 inhibition remains a pillar of guideline-directed medical therapy,” Dr. Wiviott said.
In an interview, Dr. Wiviott described the trial design as a “hybrid” in that it used a registry but then added, in his words, “some of the bells and whistles that we have with normal cardiovascular clinical trials.” He further explained: “This is a nice combination of those two things, where they use that as part of the endpoint for the trial but they’re able to add in some of the pieces that you would in a regular registration pathway trial.”
The trial design could serve as a model for future pragmatic therapeutic trials in acute MI, he said, but he acknowledged that DAPA-MI was underpowered to discern many key outcomes.
“They anticipated they were going to have a rate of around 11% of events so they needed to enroll about 6,000 people, but somewhere in the middle of the trial they saw the rate was 2.5%, not 11%, so they had to completely change the trial,” he said of the DAPA-MI investigators.
But an appropriately powered study of SGLT2 inhibition in this population would need about 28,000 patients. “This would be an enormous trial to actually clinically power, so in my sense it’s not going to happen,” Dr. Wiviott said.
The DAPA-MI trial was sponsored by AstraZeneca. Dr. James disclosed relationships with AstraZeneca, Janssen, and Amgen. Dr. Wiviott disclosed relationships with Amgen, AstraZeneca, Janssen, Merck, Pfizer, Icon Clinical, Novo Nordisk, and Varian.
PHILADELPHIA – Giving the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) to patients with acute myocardial infarction and impaired left ventricular systolic function but no diabetes or chronic heart failure significantly improved a composite of cardiovascular outcomes, a European registry-based randomized trial suggests.
In presenting these results from the DAPA-MI trial, Stefan James, MD, of Uppsala University (Sweden), noted that which the trial described as the hierarchical “win ratio” composite outcomes, compared with patients randomized to placebo plus standard of care.
“The ‘win ratio’ tells us that there’s a 34% higher likelihood of patients having a better cardiometabolic outcome with dapagliflozin vs placebo in terms of the seven components,” James said in an interview. The win ratio was achieved in 32.9% of dapagliflozin patients versus 24.6% of placebo (P < .001).
Dr. James presented the results at the annual scientific sessions of the American Heart Association, and they were published online simultaneously in NEJM Evidence.
Lower-risk patients
DAPA-MI enrolled 4,017 patients from the SWEDEHEART and Myocardial Ischemia National Audit Project registries in Sweden and the United Kingdom, randomly assigning patients to dapagliflozin 10 mg or placebo along with guideline-directed therapy for both groups.
Eligible patients were hemodynamically stable, had an acute MI within 10 days of enrollment, and impaired left ventricular systolic function or a Q-wave MI. Exclusion criteria included history of either type 1 or 2 diabetes, chronic heart failure, poor kidney function, or current treatment with an SGLT2 inhibitor. Baseline demographic characteristics were similar between trial arms.
- The hierarchical seven primary endpoints were:
- Death, with cardiovascular death ranked first followed by noncardiovascular death
- Hospitalization because of heart failure, with adjudicated first followed by investigator-reported HF
- Nonfatal MI
- Atrial fibrillation/flutter event
- New diagnosis of type 2 diabetes
- New York Heart Association functional class at the last visit
- Drop in body weight of at least 5% at the last visit
The key secondary endpoint, Dr. James said, was the primary outcome minus the body weight component, with time to first occurrence of hospitalization for HF or cardiovascular death.
When the seventh factor, body weight decrease, was removed, the differential narrowed: 20.3% versus 16.9% (P = .015). When two or more variables were removed from the composite, the differences were not statistically significant.
For 11 secondary and exploratory outcomes, ranging from CV death or hospitalization for HF to all-cause hospitalization, the outcomes were similar in both the dapagliflozin and placebo groups across the board.
However, the dapagliflozin patients had about half the rate of developing diabetes, compared with the placebo group: 2.1 % versus 3.9%.
The trial initially used the composite of CV death and hospitalization for HF as the primary endpoint, but switched to the seven-item composite endpoint in February because the number of primary composite outcomes was substantially lower than anticipated, Dr. James said.
He acknowledged the study was underpowered for the low-risk population it enrolled. “But if you extended the trial to a larger population and enriched it with a higher-risk population you would probably see an effect,” he said.
“The cardiometabolic benefit was consistent across all prespecified subgroups and there were no new safety concerns,” Dr. James told the attendees. “Clinical event rates were low with no significant difference between randomized groups.”
Not a ringing endorsement
But for invited discussant Stephen D. Wiviott, MD, a cardiologist at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, the DAPA-MI trial result isn’t quite a ringing endorsement of SGLT2 inhibition in these patients.
“From my perspective, DAPA-MI does not suggest a new mandate to expand SGLT2 inhibition to an isolated MI population without other SGLT2 inhibitor indications,” Dr. Wiviott told attendees. “But it does support the safety of its use among patients with acute coronary syndromes.”
However, “these results do not indicate a lack of clinical benefit in patients with prior MI and any of those previously identified conditions – a history of diabetes, coronary heart failure or chronic kidney disease – where SGLT2 inhibition remains a pillar of guideline-directed medical therapy,” Dr. Wiviott said.
In an interview, Dr. Wiviott described the trial design as a “hybrid” in that it used a registry but then added, in his words, “some of the bells and whistles that we have with normal cardiovascular clinical trials.” He further explained: “This is a nice combination of those two things, where they use that as part of the endpoint for the trial but they’re able to add in some of the pieces that you would in a regular registration pathway trial.”
The trial design could serve as a model for future pragmatic therapeutic trials in acute MI, he said, but he acknowledged that DAPA-MI was underpowered to discern many key outcomes.
“They anticipated they were going to have a rate of around 11% of events so they needed to enroll about 6,000 people, but somewhere in the middle of the trial they saw the rate was 2.5%, not 11%, so they had to completely change the trial,” he said of the DAPA-MI investigators.
But an appropriately powered study of SGLT2 inhibition in this population would need about 28,000 patients. “This would be an enormous trial to actually clinically power, so in my sense it’s not going to happen,” Dr. Wiviott said.
The DAPA-MI trial was sponsored by AstraZeneca. Dr. James disclosed relationships with AstraZeneca, Janssen, and Amgen. Dr. Wiviott disclosed relationships with Amgen, AstraZeneca, Janssen, Merck, Pfizer, Icon Clinical, Novo Nordisk, and Varian.
PHILADELPHIA – Giving the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) to patients with acute myocardial infarction and impaired left ventricular systolic function but no diabetes or chronic heart failure significantly improved a composite of cardiovascular outcomes, a European registry-based randomized trial suggests.
In presenting these results from the DAPA-MI trial, Stefan James, MD, of Uppsala University (Sweden), noted that which the trial described as the hierarchical “win ratio” composite outcomes, compared with patients randomized to placebo plus standard of care.
“The ‘win ratio’ tells us that there’s a 34% higher likelihood of patients having a better cardiometabolic outcome with dapagliflozin vs placebo in terms of the seven components,” James said in an interview. The win ratio was achieved in 32.9% of dapagliflozin patients versus 24.6% of placebo (P < .001).
Dr. James presented the results at the annual scientific sessions of the American Heart Association, and they were published online simultaneously in NEJM Evidence.
Lower-risk patients
DAPA-MI enrolled 4,017 patients from the SWEDEHEART and Myocardial Ischemia National Audit Project registries in Sweden and the United Kingdom, randomly assigning patients to dapagliflozin 10 mg or placebo along with guideline-directed therapy for both groups.
Eligible patients were hemodynamically stable, had an acute MI within 10 days of enrollment, and impaired left ventricular systolic function or a Q-wave MI. Exclusion criteria included history of either type 1 or 2 diabetes, chronic heart failure, poor kidney function, or current treatment with an SGLT2 inhibitor. Baseline demographic characteristics were similar between trial arms.
- The hierarchical seven primary endpoints were:
- Death, with cardiovascular death ranked first followed by noncardiovascular death
- Hospitalization because of heart failure, with adjudicated first followed by investigator-reported HF
- Nonfatal MI
- Atrial fibrillation/flutter event
- New diagnosis of type 2 diabetes
- New York Heart Association functional class at the last visit
- Drop in body weight of at least 5% at the last visit
The key secondary endpoint, Dr. James said, was the primary outcome minus the body weight component, with time to first occurrence of hospitalization for HF or cardiovascular death.
When the seventh factor, body weight decrease, was removed, the differential narrowed: 20.3% versus 16.9% (P = .015). When two or more variables were removed from the composite, the differences were not statistically significant.
For 11 secondary and exploratory outcomes, ranging from CV death or hospitalization for HF to all-cause hospitalization, the outcomes were similar in both the dapagliflozin and placebo groups across the board.
However, the dapagliflozin patients had about half the rate of developing diabetes, compared with the placebo group: 2.1 % versus 3.9%.
The trial initially used the composite of CV death and hospitalization for HF as the primary endpoint, but switched to the seven-item composite endpoint in February because the number of primary composite outcomes was substantially lower than anticipated, Dr. James said.
He acknowledged the study was underpowered for the low-risk population it enrolled. “But if you extended the trial to a larger population and enriched it with a higher-risk population you would probably see an effect,” he said.
“The cardiometabolic benefit was consistent across all prespecified subgroups and there were no new safety concerns,” Dr. James told the attendees. “Clinical event rates were low with no significant difference between randomized groups.”
Not a ringing endorsement
But for invited discussant Stephen D. Wiviott, MD, a cardiologist at Brigham and Women’s Hospital and Harvard Medical School, both in Boston, the DAPA-MI trial result isn’t quite a ringing endorsement of SGLT2 inhibition in these patients.
“From my perspective, DAPA-MI does not suggest a new mandate to expand SGLT2 inhibition to an isolated MI population without other SGLT2 inhibitor indications,” Dr. Wiviott told attendees. “But it does support the safety of its use among patients with acute coronary syndromes.”
However, “these results do not indicate a lack of clinical benefit in patients with prior MI and any of those previously identified conditions – a history of diabetes, coronary heart failure or chronic kidney disease – where SGLT2 inhibition remains a pillar of guideline-directed medical therapy,” Dr. Wiviott said.
In an interview, Dr. Wiviott described the trial design as a “hybrid” in that it used a registry but then added, in his words, “some of the bells and whistles that we have with normal cardiovascular clinical trials.” He further explained: “This is a nice combination of those two things, where they use that as part of the endpoint for the trial but they’re able to add in some of the pieces that you would in a regular registration pathway trial.”
The trial design could serve as a model for future pragmatic therapeutic trials in acute MI, he said, but he acknowledged that DAPA-MI was underpowered to discern many key outcomes.
“They anticipated they were going to have a rate of around 11% of events so they needed to enroll about 6,000 people, but somewhere in the middle of the trial they saw the rate was 2.5%, not 11%, so they had to completely change the trial,” he said of the DAPA-MI investigators.
But an appropriately powered study of SGLT2 inhibition in this population would need about 28,000 patients. “This would be an enormous trial to actually clinically power, so in my sense it’s not going to happen,” Dr. Wiviott said.
The DAPA-MI trial was sponsored by AstraZeneca. Dr. James disclosed relationships with AstraZeneca, Janssen, and Amgen. Dr. Wiviott disclosed relationships with Amgen, AstraZeneca, Janssen, Merck, Pfizer, Icon Clinical, Novo Nordisk, and Varian.
AT AHA 2023
Long COVID and mental illness: New guidance
The consensus guidance statement on the assessment and treatment of mental health symptoms in patients with post-acute sequelae of SARS-CoV-2 infection (PASC), also known as long COVID, was published online in Physical Medicine and Rehabilitation, the journal of the American Academy of Physical Medicine and Rehabilitation (AAPM&R).
The statement was developed by a task force that included experts from physical medicine, neurology, neuropsychiatry, neuropsychology, rehabilitation psychology, and primary care. It is the eighth guidance statement on long COVID published by AAPM&R).
“Many of our patients have reported experiences in which their symptoms of long COVID have been dismissed either by loved ones in the community, or also amongst health care providers, and they’ve been told their symptoms are in their head or due to a mental health condition, but that’s simply not true,” Abby L. Cheng, MD, a physiatrist at Barnes Jewish Hospital in St. Louis and a coauthor of the new guidance, said in a press briefing.
“Long COVID is real, and mental health conditions do not cause long COVID,” Dr. Cheng added.
Millions of Americans affected
Anxiety and depression have been reported as the second and third most common symptoms of long COVID, according to the guidance statement.
There is some evidence that the body’s inflammatory response – specifically, circulating cytokines – may contribute to the worsening of mental health symptoms or may bring on new symptoms of anxiety or depression, said Dr. Cheng. Cytokines may also affect levels of brain chemicals, such as serotonin, she said.
Researchers are also exploring whether the persistence of virus in the body, miniature blood clots in the body and brain, and changes to the gut microbiome affect the mental health of people with long COVID.
Some mental health symptoms – such as fatigue, brain fog, sleep disturbances, and tachycardia – can mimic long COVID symptoms, said Dr. Cheng.
The treatment is the same for someone with or without long COVID who has anxiety, depression, posttraumatic stress disorder, or other mental health conditions and includes treatment of coexisting medical conditions, supportive therapy and cognitive-behavioral therapy, and pharmacologic interventions, she said.
“Group therapy may have a particular role in the long COVID population because it really provides that social connection and awareness of additional resources in addition to validation of their experiences,” Dr. Cheng said.
The guidance suggests that primary care practitioners – if it’s within their comfort zone and they have the training – can be the first line for managing mental health symptoms.
But for patients whose symptoms are interfering with functioning and their ability to interact with the community, the guidance urges primary care clinicians to refer the patient to a specialist.
“It leaves the door open to them to practice within their scope but also gives guidance as to how, why, and who should be referred to the next level of care,” said Dr. Cheng.
Coauthor Monica Verduzco-Gutierrez, MD, chair of rehabilitation medicine at UT Health San Antonio, Texas, said that although fewer people are now getting long COVID, “it’s still an impactful number.”
The Centers for Disease Control and Prevention recently estimated that about 7% of American adults (18 million) and 1.3% of children had experienced long COVID.
Dr. Gutierrez said that it’s an evolving number, as some patients who have a second or third or fourth SARS-CoV-2 infection experience exacerbations of previous bouts of long COVID or develop long COVID for the first time.
“We are still getting new patients on a regular basis with long COVID,” said AAPM&R President Steven R. Flanagan, MD, a physical medicine specialist.
“This is a problem that really is not going away. It is still real and still ever-present,” said Dr. Flanagan, chair of rehabilitation medicine at NYU Langone Health.
A version of this article first appeared on Medscape.com.
The consensus guidance statement on the assessment and treatment of mental health symptoms in patients with post-acute sequelae of SARS-CoV-2 infection (PASC), also known as long COVID, was published online in Physical Medicine and Rehabilitation, the journal of the American Academy of Physical Medicine and Rehabilitation (AAPM&R).
The statement was developed by a task force that included experts from physical medicine, neurology, neuropsychiatry, neuropsychology, rehabilitation psychology, and primary care. It is the eighth guidance statement on long COVID published by AAPM&R).
“Many of our patients have reported experiences in which their symptoms of long COVID have been dismissed either by loved ones in the community, or also amongst health care providers, and they’ve been told their symptoms are in their head or due to a mental health condition, but that’s simply not true,” Abby L. Cheng, MD, a physiatrist at Barnes Jewish Hospital in St. Louis and a coauthor of the new guidance, said in a press briefing.
“Long COVID is real, and mental health conditions do not cause long COVID,” Dr. Cheng added.
Millions of Americans affected
Anxiety and depression have been reported as the second and third most common symptoms of long COVID, according to the guidance statement.
There is some evidence that the body’s inflammatory response – specifically, circulating cytokines – may contribute to the worsening of mental health symptoms or may bring on new symptoms of anxiety or depression, said Dr. Cheng. Cytokines may also affect levels of brain chemicals, such as serotonin, she said.
Researchers are also exploring whether the persistence of virus in the body, miniature blood clots in the body and brain, and changes to the gut microbiome affect the mental health of people with long COVID.
Some mental health symptoms – such as fatigue, brain fog, sleep disturbances, and tachycardia – can mimic long COVID symptoms, said Dr. Cheng.
The treatment is the same for someone with or without long COVID who has anxiety, depression, posttraumatic stress disorder, or other mental health conditions and includes treatment of coexisting medical conditions, supportive therapy and cognitive-behavioral therapy, and pharmacologic interventions, she said.
“Group therapy may have a particular role in the long COVID population because it really provides that social connection and awareness of additional resources in addition to validation of their experiences,” Dr. Cheng said.
The guidance suggests that primary care practitioners – if it’s within their comfort zone and they have the training – can be the first line for managing mental health symptoms.
But for patients whose symptoms are interfering with functioning and their ability to interact with the community, the guidance urges primary care clinicians to refer the patient to a specialist.
“It leaves the door open to them to practice within their scope but also gives guidance as to how, why, and who should be referred to the next level of care,” said Dr. Cheng.
Coauthor Monica Verduzco-Gutierrez, MD, chair of rehabilitation medicine at UT Health San Antonio, Texas, said that although fewer people are now getting long COVID, “it’s still an impactful number.”
The Centers for Disease Control and Prevention recently estimated that about 7% of American adults (18 million) and 1.3% of children had experienced long COVID.
Dr. Gutierrez said that it’s an evolving number, as some patients who have a second or third or fourth SARS-CoV-2 infection experience exacerbations of previous bouts of long COVID or develop long COVID for the first time.
“We are still getting new patients on a regular basis with long COVID,” said AAPM&R President Steven R. Flanagan, MD, a physical medicine specialist.
“This is a problem that really is not going away. It is still real and still ever-present,” said Dr. Flanagan, chair of rehabilitation medicine at NYU Langone Health.
A version of this article first appeared on Medscape.com.
The consensus guidance statement on the assessment and treatment of mental health symptoms in patients with post-acute sequelae of SARS-CoV-2 infection (PASC), also known as long COVID, was published online in Physical Medicine and Rehabilitation, the journal of the American Academy of Physical Medicine and Rehabilitation (AAPM&R).
The statement was developed by a task force that included experts from physical medicine, neurology, neuropsychiatry, neuropsychology, rehabilitation psychology, and primary care. It is the eighth guidance statement on long COVID published by AAPM&R).
“Many of our patients have reported experiences in which their symptoms of long COVID have been dismissed either by loved ones in the community, or also amongst health care providers, and they’ve been told their symptoms are in their head or due to a mental health condition, but that’s simply not true,” Abby L. Cheng, MD, a physiatrist at Barnes Jewish Hospital in St. Louis and a coauthor of the new guidance, said in a press briefing.
“Long COVID is real, and mental health conditions do not cause long COVID,” Dr. Cheng added.
Millions of Americans affected
Anxiety and depression have been reported as the second and third most common symptoms of long COVID, according to the guidance statement.
There is some evidence that the body’s inflammatory response – specifically, circulating cytokines – may contribute to the worsening of mental health symptoms or may bring on new symptoms of anxiety or depression, said Dr. Cheng. Cytokines may also affect levels of brain chemicals, such as serotonin, she said.
Researchers are also exploring whether the persistence of virus in the body, miniature blood clots in the body and brain, and changes to the gut microbiome affect the mental health of people with long COVID.
Some mental health symptoms – such as fatigue, brain fog, sleep disturbances, and tachycardia – can mimic long COVID symptoms, said Dr. Cheng.
The treatment is the same for someone with or without long COVID who has anxiety, depression, posttraumatic stress disorder, or other mental health conditions and includes treatment of coexisting medical conditions, supportive therapy and cognitive-behavioral therapy, and pharmacologic interventions, she said.
“Group therapy may have a particular role in the long COVID population because it really provides that social connection and awareness of additional resources in addition to validation of their experiences,” Dr. Cheng said.
The guidance suggests that primary care practitioners – if it’s within their comfort zone and they have the training – can be the first line for managing mental health symptoms.
But for patients whose symptoms are interfering with functioning and their ability to interact with the community, the guidance urges primary care clinicians to refer the patient to a specialist.
“It leaves the door open to them to practice within their scope but also gives guidance as to how, why, and who should be referred to the next level of care,” said Dr. Cheng.
Coauthor Monica Verduzco-Gutierrez, MD, chair of rehabilitation medicine at UT Health San Antonio, Texas, said that although fewer people are now getting long COVID, “it’s still an impactful number.”
The Centers for Disease Control and Prevention recently estimated that about 7% of American adults (18 million) and 1.3% of children had experienced long COVID.
Dr. Gutierrez said that it’s an evolving number, as some patients who have a second or third or fourth SARS-CoV-2 infection experience exacerbations of previous bouts of long COVID or develop long COVID for the first time.
“We are still getting new patients on a regular basis with long COVID,” said AAPM&R President Steven R. Flanagan, MD, a physical medicine specialist.
“This is a problem that really is not going away. It is still real and still ever-present,” said Dr. Flanagan, chair of rehabilitation medicine at NYU Langone Health.
A version of this article first appeared on Medscape.com.
FROM PHYSICAL MEDICINE AND REHABILITATION
In MI with anemia, results may favor liberal transfusion: MINT
In patients with myocardial infarction and anemia, a “liberal” red blood cell transfusion strategy did not significantly reduce the risk of recurrent MI or death within 30 days, compared with a “restrictive” transfusion strategy, in the 3,500-patient MINT trial.
Jeffrey L. Carson, MD, from Robert Wood Johnson Medical School, New Brunswick, N.J., said in a press briefing.
He presented the study in a late-breaking trial session at the annual scientific sessions of the American Heart Association, and it was simultaneously published online in the New England Journal of Medicine.
“Whether to transfuse is an everyday decision faced by clinicians caring for patients with acute MI,” Dr. Carson said.
“We cannot claim that a liberal transfusion strategy is definitively superior based on our primary outcome,” he said, but “the 95% confidence interval is consistent with treatment effects corresponding to no difference between the two transfusion strategies and to a clinically relevant benefit with the liberal strategy.”
“In contrast to other trials in other settings,” such as anemia and cardiac surgery, Dr. Carson said, “the results suggest that a liberal transfusion strategy has the potential for clinical benefit with an acceptable risk of harm.”
“A liberal transfusion strategy may be the most prudent approach to transfusion in anemic patients with MI,” he added.
Not a home run
Others agreed with this interpretation. Martin B. Leon, MD, from Columbia University, New York, the study discussant in the press briefing, said the study “addresses a question that is common” in clinical practice. It was well conducted, and international (although most patients were in the United States and Canada), in a very broad group of patients, designed to make the results more generalizable. The 98% follow-up was extremely good, Dr. Leon added, and the trialists achieved their goal in that they did show a difference between the two transfusion strategies.
The number needed to treat was 40 to see a benefit in the combined outcome of death or recurrent MI at 30 days, Dr. Leon said. The P value for this was .07, “right on the edge” of statistical significance.
This study is “not a home run,” for the primary outcome, he noted; however, many of the outcomes tended to be in favor of a liberal transfusion strategy. Notably, cardiovascular death, which was not a specified outcome, was significantly lower in the group who received a liberal transfusion strategy.
Although a liberal transfusion strategy was “not definitely superior” in these patients with MI and anemia, Dr. Carson said, he thinks the trial will be interpreted as favoring a liberal transfusion strategy.
C. Michael Gibson, MD, professor of medicine at Harvard Medical School, Boston, and CEO of Harvard’s Baim and PERFUSE institutes for clinical research, voiced similar views.
“Given the lack of acute harm associated with liberal transfusion and the preponderance of evidence favoring liberal transfusion in the largest trial to date,” concluded Dr. Gibson, the assigned discussant at the session, “liberal transfusion appears to be a viable management strategy, particularly among patients with non-STEMI type 1 MI and as clinical judgment dictates.”
Only three small randomized controlled trials have compared transfusion thresholds in a total of 820 patients with MI and anemia, Dr. Gibson said, a point that the trial investigators also made. The results were inconsistent between trials: the CRIT trial (n = 45) favored a restrictive strategy, the MINT pilot study (n = 110) favored a liberal one, and the REALITY trial (n = 668) showed noninferiority of a restrictive strategy, compared with a liberal strategy in 30-day MACE.
The MINT trial was four times larger than all prior studies combined. However, most outcomes were negative or of borderline significance for benefit.
Cardiac death was more common in the restrictive group at 5.5% than the liberal group at 3.2% (risk ratio, 1.74, 95% CI, 1.26-2.40), but this was nonadjudicated, and not designated as a primary, secondary, or tertiary outcome – which the researchers also noted. Fewer than half of the deaths were classified as cardiac, which was “odd,” Dr. Gibson observed.
A restrictive transfusion strategy was associated with increased events among participants with type 1 MI (RR, 1.32, 95% CI, 1.04-1.67), he noted.
Study strengths included that 45.5% of participants were women, Dr. Gibson said. Limitations included that the trial was “somewhat underpowered.” Also, even in the restrictive group, participants received a mean of 0.7 units of packed red blood cells.
Adherence to the 10 g/dL threshold in the liberal transfusion group was moderate (86.3% at hospital discharge), which the researchers acknowledged. They noted that this was frequently caused by clinical discretion, such as concern about fluid overload, and to the timing of hospital discharge. In addition, long-term potential for harm (microchimerism) is not known.
“There was a consistent nonsignificant acute benefit for liberal transfusion and a nominal reduction in CV mortality and improved outcomes in patients with type 1 MI in exploratory analyses, in a trial that ended up underpowered,” Dr. Gibson summarized. “Long-term follow up would be helpful to evaluate chronic outcomes.”
This is a very well-conducted, high-quality, important study that will be considered a landmark trial, C. David Mazer, MD, University of Toronto and St. Michael’s Hospital, also in Toronto, said in an interview.
Unfortunately, “it was not as definitive as hoped for,” Dr. Mazer lamented. Nevertheless, “I think people may interpret it as providing support for a liberal transfusion strategy” in patients with anemia and MI, he said.
Dr. Mazer, who was not involved with this research, was a principal investigator on the TRICS-3 trial, which disputed a liberal RBC transfusion strategy in patients with anemia undergoing cardiac surgery, as previously reported.
The “Red Blood Cell Transfusion: 2023 AABB International Guidelines,” led by Dr. Carson and published in JAMA, recommend a restrictive strategy in stable patients, although these guidelines did not include the current study, Dr. Mazer observed.
In the REALITY trial, there were fewer major adverse cardiac events (MACE) events in the restrictive strategy, he noted.
MINT can be viewed as comparing a high versus low hemoglobin threshold. “It is possible that the best is in between,” he said.
Dr. Mazer also noted that MINT may have achieved significance if it was designed with a larger enrollment and a higher power (for example, 90% instead of 80%) to detect between-group difference for the primary outcome.
Study rationale, design, and findings
Anemia, or low RBC count, is common in patients with MI, Dr. Carson noted. A normal hemoglobin is 13 g/dL in men and 12 g/dL in women. Administering a packed RBC transfusion only when a patient’s hemoglobin falls below 7 or 8 g/dL has been widely adopted, but it is unclear if patients with acute MI may benefit from a higher hemoglobin level.
“Blood transfusion may decrease ischemic injury by improving oxygen delivery to myocardial tissues and reduce the risk of reinfarction or death,” the researchers wrote. “Alternatively, administering more blood could result in more frequent heart failure from fluid overload, infection from immunosuppression, thrombosis from higher viscosity, and inflammation.”
From 2017 to 2023, investigators enrolled 3,504 adults aged 18 and older at 144 sites in the United States (2,157 patients), Canada (885), France (323), Brazil (105), New Zealand (25), and Australia (9).
The participants had ST-elevation or non–ST-elevation MI and hemoglobin less than 10 g/dL within 24 hours. Patients with type 1 (atherosclerotic plaque disruption), type 2 (supply-demand mismatch without atherothrombotic plaque disruption), type 4b, or type 4c MI were eligible.
They were randomly assigned to receive:
- A ‘restrictive’ transfusion strategy (1,749 patients): Transfusion was permitted but not required when a patient’s hemoglobin was less than 8 g/dL and was strongly recommended when it was less than 7 g/dL or when anginal symptoms were not controlled with medications.
- A ‘liberal’ transfusion strategy (1,755 patients): One unit of RBCs was administered after randomization, and RBCs were transfused to maintain hemoglobin 10 g/dL or higher until hospital discharge or 30 days.
The patients had a mean age of 72 years and 46% were women. More than three-quarters (78%) were White and 14% were Black. They had frequent coexisting illnesses, about a third had a history of MI, percutaneous coronary intervention, or heart failure; 14% were on a ventilator and 12% had renal dialysis. The median duration of hospitalization was 5 days in the two groups.
At baseline, the mean hemoglobin was 8.6 g/dL in both groups. At days 1, 2, and 3, the mean hemoglobin was 8.8, 8.9, and 8.9 g/dL, respectively, in the restrictive transfusion group, and 10.1, 10.4, and 10.5 g/dL, respectively, in the liberal transfusion group.
The mean number of transfused blood units was 0.7 units in the restrictive strategy group and 2.5 units in the liberal strategy group, roughly a 3.5-fold difference.
After adjustment for site and incomplete follow-up in 57 patients (20 with the restrictive strategy and 37 with the liberal strategy), the estimated RR for the primary outcome in the restrictive group versus the liberal group was 1.15 (P = .07).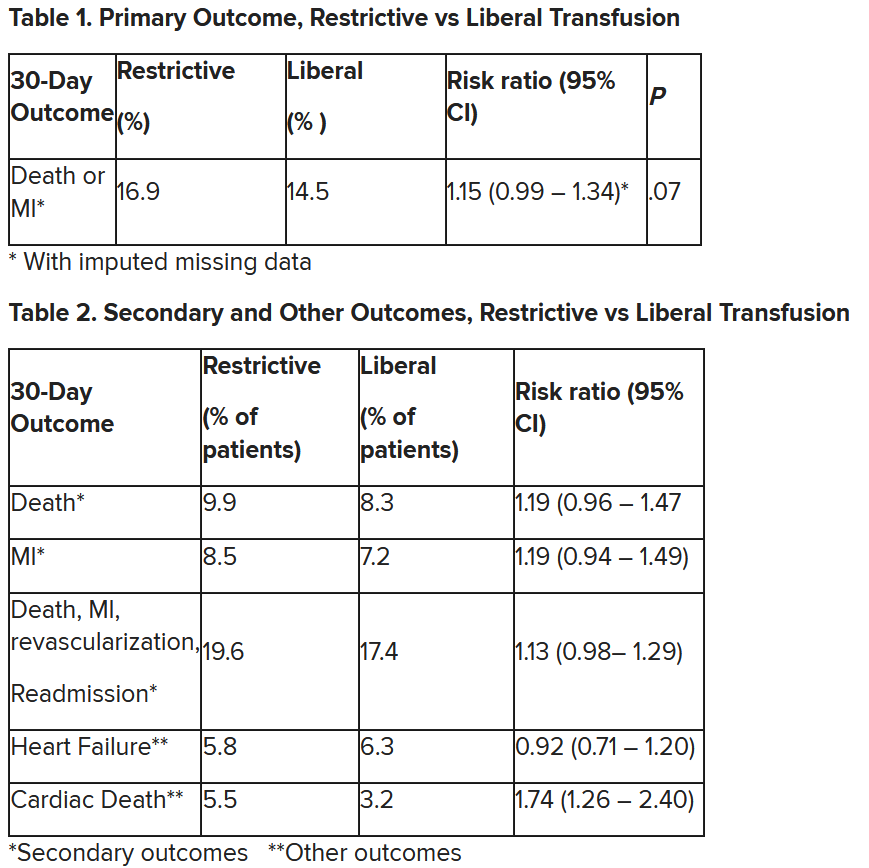
“We observed that the 95% confidence interval contains values that suggest a clinical benefit for the liberal transfusion strategy and does not include values that suggest a benefit for the more restrictive transfusion strategy,” the researchers wrote. Heart failure and other safety outcomes were comparable in the two groups.
The trial was supported by grants from the National Heart, Lung, and Blood Institute and by the Canadian Blood Services and Canadian Institutes of Health Research Institute of Circulatory and Respiratory Health. Dr. Carson, Dr. Leon, Dr. Gibson, and Dr. Mazer reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In patients with myocardial infarction and anemia, a “liberal” red blood cell transfusion strategy did not significantly reduce the risk of recurrent MI or death within 30 days, compared with a “restrictive” transfusion strategy, in the 3,500-patient MINT trial.
Jeffrey L. Carson, MD, from Robert Wood Johnson Medical School, New Brunswick, N.J., said in a press briefing.
He presented the study in a late-breaking trial session at the annual scientific sessions of the American Heart Association, and it was simultaneously published online in the New England Journal of Medicine.
“Whether to transfuse is an everyday decision faced by clinicians caring for patients with acute MI,” Dr. Carson said.
“We cannot claim that a liberal transfusion strategy is definitively superior based on our primary outcome,” he said, but “the 95% confidence interval is consistent with treatment effects corresponding to no difference between the two transfusion strategies and to a clinically relevant benefit with the liberal strategy.”
“In contrast to other trials in other settings,” such as anemia and cardiac surgery, Dr. Carson said, “the results suggest that a liberal transfusion strategy has the potential for clinical benefit with an acceptable risk of harm.”
“A liberal transfusion strategy may be the most prudent approach to transfusion in anemic patients with MI,” he added.
Not a home run
Others agreed with this interpretation. Martin B. Leon, MD, from Columbia University, New York, the study discussant in the press briefing, said the study “addresses a question that is common” in clinical practice. It was well conducted, and international (although most patients were in the United States and Canada), in a very broad group of patients, designed to make the results more generalizable. The 98% follow-up was extremely good, Dr. Leon added, and the trialists achieved their goal in that they did show a difference between the two transfusion strategies.
The number needed to treat was 40 to see a benefit in the combined outcome of death or recurrent MI at 30 days, Dr. Leon said. The P value for this was .07, “right on the edge” of statistical significance.
This study is “not a home run,” for the primary outcome, he noted; however, many of the outcomes tended to be in favor of a liberal transfusion strategy. Notably, cardiovascular death, which was not a specified outcome, was significantly lower in the group who received a liberal transfusion strategy.
Although a liberal transfusion strategy was “not definitely superior” in these patients with MI and anemia, Dr. Carson said, he thinks the trial will be interpreted as favoring a liberal transfusion strategy.
C. Michael Gibson, MD, professor of medicine at Harvard Medical School, Boston, and CEO of Harvard’s Baim and PERFUSE institutes for clinical research, voiced similar views.
“Given the lack of acute harm associated with liberal transfusion and the preponderance of evidence favoring liberal transfusion in the largest trial to date,” concluded Dr. Gibson, the assigned discussant at the session, “liberal transfusion appears to be a viable management strategy, particularly among patients with non-STEMI type 1 MI and as clinical judgment dictates.”
Only three small randomized controlled trials have compared transfusion thresholds in a total of 820 patients with MI and anemia, Dr. Gibson said, a point that the trial investigators also made. The results were inconsistent between trials: the CRIT trial (n = 45) favored a restrictive strategy, the MINT pilot study (n = 110) favored a liberal one, and the REALITY trial (n = 668) showed noninferiority of a restrictive strategy, compared with a liberal strategy in 30-day MACE.
The MINT trial was four times larger than all prior studies combined. However, most outcomes were negative or of borderline significance for benefit.
Cardiac death was more common in the restrictive group at 5.5% than the liberal group at 3.2% (risk ratio, 1.74, 95% CI, 1.26-2.40), but this was nonadjudicated, and not designated as a primary, secondary, or tertiary outcome – which the researchers also noted. Fewer than half of the deaths were classified as cardiac, which was “odd,” Dr. Gibson observed.
A restrictive transfusion strategy was associated with increased events among participants with type 1 MI (RR, 1.32, 95% CI, 1.04-1.67), he noted.
Study strengths included that 45.5% of participants were women, Dr. Gibson said. Limitations included that the trial was “somewhat underpowered.” Also, even in the restrictive group, participants received a mean of 0.7 units of packed red blood cells.
Adherence to the 10 g/dL threshold in the liberal transfusion group was moderate (86.3% at hospital discharge), which the researchers acknowledged. They noted that this was frequently caused by clinical discretion, such as concern about fluid overload, and to the timing of hospital discharge. In addition, long-term potential for harm (microchimerism) is not known.
“There was a consistent nonsignificant acute benefit for liberal transfusion and a nominal reduction in CV mortality and improved outcomes in patients with type 1 MI in exploratory analyses, in a trial that ended up underpowered,” Dr. Gibson summarized. “Long-term follow up would be helpful to evaluate chronic outcomes.”
This is a very well-conducted, high-quality, important study that will be considered a landmark trial, C. David Mazer, MD, University of Toronto and St. Michael’s Hospital, also in Toronto, said in an interview.
Unfortunately, “it was not as definitive as hoped for,” Dr. Mazer lamented. Nevertheless, “I think people may interpret it as providing support for a liberal transfusion strategy” in patients with anemia and MI, he said.
Dr. Mazer, who was not involved with this research, was a principal investigator on the TRICS-3 trial, which disputed a liberal RBC transfusion strategy in patients with anemia undergoing cardiac surgery, as previously reported.
The “Red Blood Cell Transfusion: 2023 AABB International Guidelines,” led by Dr. Carson and published in JAMA, recommend a restrictive strategy in stable patients, although these guidelines did not include the current study, Dr. Mazer observed.
In the REALITY trial, there were fewer major adverse cardiac events (MACE) events in the restrictive strategy, he noted.
MINT can be viewed as comparing a high versus low hemoglobin threshold. “It is possible that the best is in between,” he said.
Dr. Mazer also noted that MINT may have achieved significance if it was designed with a larger enrollment and a higher power (for example, 90% instead of 80%) to detect between-group difference for the primary outcome.
Study rationale, design, and findings
Anemia, or low RBC count, is common in patients with MI, Dr. Carson noted. A normal hemoglobin is 13 g/dL in men and 12 g/dL in women. Administering a packed RBC transfusion only when a patient’s hemoglobin falls below 7 or 8 g/dL has been widely adopted, but it is unclear if patients with acute MI may benefit from a higher hemoglobin level.
“Blood transfusion may decrease ischemic injury by improving oxygen delivery to myocardial tissues and reduce the risk of reinfarction or death,” the researchers wrote. “Alternatively, administering more blood could result in more frequent heart failure from fluid overload, infection from immunosuppression, thrombosis from higher viscosity, and inflammation.”
From 2017 to 2023, investigators enrolled 3,504 adults aged 18 and older at 144 sites in the United States (2,157 patients), Canada (885), France (323), Brazil (105), New Zealand (25), and Australia (9).
The participants had ST-elevation or non–ST-elevation MI and hemoglobin less than 10 g/dL within 24 hours. Patients with type 1 (atherosclerotic plaque disruption), type 2 (supply-demand mismatch without atherothrombotic plaque disruption), type 4b, or type 4c MI were eligible.
They were randomly assigned to receive:
- A ‘restrictive’ transfusion strategy (1,749 patients): Transfusion was permitted but not required when a patient’s hemoglobin was less than 8 g/dL and was strongly recommended when it was less than 7 g/dL or when anginal symptoms were not controlled with medications.
- A ‘liberal’ transfusion strategy (1,755 patients): One unit of RBCs was administered after randomization, and RBCs were transfused to maintain hemoglobin 10 g/dL or higher until hospital discharge or 30 days.
The patients had a mean age of 72 years and 46% were women. More than three-quarters (78%) were White and 14% were Black. They had frequent coexisting illnesses, about a third had a history of MI, percutaneous coronary intervention, or heart failure; 14% were on a ventilator and 12% had renal dialysis. The median duration of hospitalization was 5 days in the two groups.
At baseline, the mean hemoglobin was 8.6 g/dL in both groups. At days 1, 2, and 3, the mean hemoglobin was 8.8, 8.9, and 8.9 g/dL, respectively, in the restrictive transfusion group, and 10.1, 10.4, and 10.5 g/dL, respectively, in the liberal transfusion group.
The mean number of transfused blood units was 0.7 units in the restrictive strategy group and 2.5 units in the liberal strategy group, roughly a 3.5-fold difference.
After adjustment for site and incomplete follow-up in 57 patients (20 with the restrictive strategy and 37 with the liberal strategy), the estimated RR for the primary outcome in the restrictive group versus the liberal group was 1.15 (P = .07).
“We observed that the 95% confidence interval contains values that suggest a clinical benefit for the liberal transfusion strategy and does not include values that suggest a benefit for the more restrictive transfusion strategy,” the researchers wrote. Heart failure and other safety outcomes were comparable in the two groups.
The trial was supported by grants from the National Heart, Lung, and Blood Institute and by the Canadian Blood Services and Canadian Institutes of Health Research Institute of Circulatory and Respiratory Health. Dr. Carson, Dr. Leon, Dr. Gibson, and Dr. Mazer reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In patients with myocardial infarction and anemia, a “liberal” red blood cell transfusion strategy did not significantly reduce the risk of recurrent MI or death within 30 days, compared with a “restrictive” transfusion strategy, in the 3,500-patient MINT trial.
Jeffrey L. Carson, MD, from Robert Wood Johnson Medical School, New Brunswick, N.J., said in a press briefing.
He presented the study in a late-breaking trial session at the annual scientific sessions of the American Heart Association, and it was simultaneously published online in the New England Journal of Medicine.
“Whether to transfuse is an everyday decision faced by clinicians caring for patients with acute MI,” Dr. Carson said.
“We cannot claim that a liberal transfusion strategy is definitively superior based on our primary outcome,” he said, but “the 95% confidence interval is consistent with treatment effects corresponding to no difference between the two transfusion strategies and to a clinically relevant benefit with the liberal strategy.”
“In contrast to other trials in other settings,” such as anemia and cardiac surgery, Dr. Carson said, “the results suggest that a liberal transfusion strategy has the potential for clinical benefit with an acceptable risk of harm.”
“A liberal transfusion strategy may be the most prudent approach to transfusion in anemic patients with MI,” he added.
Not a home run
Others agreed with this interpretation. Martin B. Leon, MD, from Columbia University, New York, the study discussant in the press briefing, said the study “addresses a question that is common” in clinical practice. It was well conducted, and international (although most patients were in the United States and Canada), in a very broad group of patients, designed to make the results more generalizable. The 98% follow-up was extremely good, Dr. Leon added, and the trialists achieved their goal in that they did show a difference between the two transfusion strategies.
The number needed to treat was 40 to see a benefit in the combined outcome of death or recurrent MI at 30 days, Dr. Leon said. The P value for this was .07, “right on the edge” of statistical significance.
This study is “not a home run,” for the primary outcome, he noted; however, many of the outcomes tended to be in favor of a liberal transfusion strategy. Notably, cardiovascular death, which was not a specified outcome, was significantly lower in the group who received a liberal transfusion strategy.
Although a liberal transfusion strategy was “not definitely superior” in these patients with MI and anemia, Dr. Carson said, he thinks the trial will be interpreted as favoring a liberal transfusion strategy.
C. Michael Gibson, MD, professor of medicine at Harvard Medical School, Boston, and CEO of Harvard’s Baim and PERFUSE institutes for clinical research, voiced similar views.
“Given the lack of acute harm associated with liberal transfusion and the preponderance of evidence favoring liberal transfusion in the largest trial to date,” concluded Dr. Gibson, the assigned discussant at the session, “liberal transfusion appears to be a viable management strategy, particularly among patients with non-STEMI type 1 MI and as clinical judgment dictates.”
Only three small randomized controlled trials have compared transfusion thresholds in a total of 820 patients with MI and anemia, Dr. Gibson said, a point that the trial investigators also made. The results were inconsistent between trials: the CRIT trial (n = 45) favored a restrictive strategy, the MINT pilot study (n = 110) favored a liberal one, and the REALITY trial (n = 668) showed noninferiority of a restrictive strategy, compared with a liberal strategy in 30-day MACE.
The MINT trial was four times larger than all prior studies combined. However, most outcomes were negative or of borderline significance for benefit.
Cardiac death was more common in the restrictive group at 5.5% than the liberal group at 3.2% (risk ratio, 1.74, 95% CI, 1.26-2.40), but this was nonadjudicated, and not designated as a primary, secondary, or tertiary outcome – which the researchers also noted. Fewer than half of the deaths were classified as cardiac, which was “odd,” Dr. Gibson observed.
A restrictive transfusion strategy was associated with increased events among participants with type 1 MI (RR, 1.32, 95% CI, 1.04-1.67), he noted.
Study strengths included that 45.5% of participants were women, Dr. Gibson said. Limitations included that the trial was “somewhat underpowered.” Also, even in the restrictive group, participants received a mean of 0.7 units of packed red blood cells.
Adherence to the 10 g/dL threshold in the liberal transfusion group was moderate (86.3% at hospital discharge), which the researchers acknowledged. They noted that this was frequently caused by clinical discretion, such as concern about fluid overload, and to the timing of hospital discharge. In addition, long-term potential for harm (microchimerism) is not known.
“There was a consistent nonsignificant acute benefit for liberal transfusion and a nominal reduction in CV mortality and improved outcomes in patients with type 1 MI in exploratory analyses, in a trial that ended up underpowered,” Dr. Gibson summarized. “Long-term follow up would be helpful to evaluate chronic outcomes.”
This is a very well-conducted, high-quality, important study that will be considered a landmark trial, C. David Mazer, MD, University of Toronto and St. Michael’s Hospital, also in Toronto, said in an interview.
Unfortunately, “it was not as definitive as hoped for,” Dr. Mazer lamented. Nevertheless, “I think people may interpret it as providing support for a liberal transfusion strategy” in patients with anemia and MI, he said.
Dr. Mazer, who was not involved with this research, was a principal investigator on the TRICS-3 trial, which disputed a liberal RBC transfusion strategy in patients with anemia undergoing cardiac surgery, as previously reported.
The “Red Blood Cell Transfusion: 2023 AABB International Guidelines,” led by Dr. Carson and published in JAMA, recommend a restrictive strategy in stable patients, although these guidelines did not include the current study, Dr. Mazer observed.
In the REALITY trial, there were fewer major adverse cardiac events (MACE) events in the restrictive strategy, he noted.
MINT can be viewed as comparing a high versus low hemoglobin threshold. “It is possible that the best is in between,” he said.
Dr. Mazer also noted that MINT may have achieved significance if it was designed with a larger enrollment and a higher power (for example, 90% instead of 80%) to detect between-group difference for the primary outcome.
Study rationale, design, and findings
Anemia, or low RBC count, is common in patients with MI, Dr. Carson noted. A normal hemoglobin is 13 g/dL in men and 12 g/dL in women. Administering a packed RBC transfusion only when a patient’s hemoglobin falls below 7 or 8 g/dL has been widely adopted, but it is unclear if patients with acute MI may benefit from a higher hemoglobin level.
“Blood transfusion may decrease ischemic injury by improving oxygen delivery to myocardial tissues and reduce the risk of reinfarction or death,” the researchers wrote. “Alternatively, administering more blood could result in more frequent heart failure from fluid overload, infection from immunosuppression, thrombosis from higher viscosity, and inflammation.”
From 2017 to 2023, investigators enrolled 3,504 adults aged 18 and older at 144 sites in the United States (2,157 patients), Canada (885), France (323), Brazil (105), New Zealand (25), and Australia (9).
The participants had ST-elevation or non–ST-elevation MI and hemoglobin less than 10 g/dL within 24 hours. Patients with type 1 (atherosclerotic plaque disruption), type 2 (supply-demand mismatch without atherothrombotic plaque disruption), type 4b, or type 4c MI were eligible.
They were randomly assigned to receive:
- A ‘restrictive’ transfusion strategy (1,749 patients): Transfusion was permitted but not required when a patient’s hemoglobin was less than 8 g/dL and was strongly recommended when it was less than 7 g/dL or when anginal symptoms were not controlled with medications.
- A ‘liberal’ transfusion strategy (1,755 patients): One unit of RBCs was administered after randomization, and RBCs were transfused to maintain hemoglobin 10 g/dL or higher until hospital discharge or 30 days.
The patients had a mean age of 72 years and 46% were women. More than three-quarters (78%) were White and 14% were Black. They had frequent coexisting illnesses, about a third had a history of MI, percutaneous coronary intervention, or heart failure; 14% were on a ventilator and 12% had renal dialysis. The median duration of hospitalization was 5 days in the two groups.
At baseline, the mean hemoglobin was 8.6 g/dL in both groups. At days 1, 2, and 3, the mean hemoglobin was 8.8, 8.9, and 8.9 g/dL, respectively, in the restrictive transfusion group, and 10.1, 10.4, and 10.5 g/dL, respectively, in the liberal transfusion group.
The mean number of transfused blood units was 0.7 units in the restrictive strategy group and 2.5 units in the liberal strategy group, roughly a 3.5-fold difference.
After adjustment for site and incomplete follow-up in 57 patients (20 with the restrictive strategy and 37 with the liberal strategy), the estimated RR for the primary outcome in the restrictive group versus the liberal group was 1.15 (P = .07).
“We observed that the 95% confidence interval contains values that suggest a clinical benefit for the liberal transfusion strategy and does not include values that suggest a benefit for the more restrictive transfusion strategy,” the researchers wrote. Heart failure and other safety outcomes were comparable in the two groups.
The trial was supported by grants from the National Heart, Lung, and Blood Institute and by the Canadian Blood Services and Canadian Institutes of Health Research Institute of Circulatory and Respiratory Health. Dr. Carson, Dr. Leon, Dr. Gibson, and Dr. Mazer reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AHA 2023
High levels of concern reported about ED boarding crisis
ccording to a poll conducted on behalf of the American College of Emergency Physicians in September 2023. This issue not only affects patient care but also has far-reaching consequences for the efficiency of emergency medical services (EMS).
The survey interviewed 2,164 adults and showed that an overwhelming majority (80%) expressed serious concerns about the boarding crisis. Moreover, 43% of respondents either delayed seeking medical care at an ED or avoided it altogether if they anticipated prolonged wait times before being admitted to the hospital or transferred to another facility.
Nearly half of adults (44%) experienced long wait times following initial care in an ED, and 16% of these adults reported 13 or more hours of waiting after receiving initial care.
“The boarding crisis is a predictable result of an acute care hospital system with insufficient capacity – we lack enough space and staff in our acute care hospitals, as we have not created the bed capacity needed for an aging and higher acuity patient population, and staffing shortages for inpatient beds have resulted in a longer hospital length of stay that we observe as boarding patients in the ED,” Arjun Venkatesh, MD, chief of emergency services at Yale New Haven Hospital, told this news organization, commenting on the factors contributing to this crisis.
One concerning side effect of boarding in EDs is the delayed response of ambulance services. When a hospital is unprepared to receive patients arriving in an ambulance, ambulance crews often wait with the patients for extended periods until the hospital can admit them. This situation can have critical implications, as parked ambulances are unable to respond to other emergencies in the community.
Adults who have endured long wait times in EDs voiced concerns about the negative impact such delays can have on their medical care. The experiences of patients and their families have underscored the urgency of addressing the boarding crisis.
“There are no low-hanging fruit solutions or simple communication strategies to alleviate these concerns,” said Dr. Venkatesh, adding that “the only way to change that perception is to change the care by addressing the lack of inpatient capacity and community care capacity to avoid back-up in the ED.”
The poll revealed that 93% of adults across different demographic groups considered EMS, including paramedics, essential. Older adults were more likely to emphasize the significance of these services than were younger adults.
“We all need a place for people to go when there is an emergency, whether it be trauma, a heart attack, a stroke, or similar conditions,” said Scott Weiner, MD, associate professor of emergency medicine at Harvard Medical School and attending emergency physician at Brigham and Women’s Hospital, Boston.
“However, the modern ED is much more than that. It has become the center for coordination of care across the health system where diagnostic tests can be completed promptly, where follow-up care is arranged, and where behavioral health emergencies come,” he added.
A vast majority, 89%, believed that additional or supplemental government funding should be directed toward these essential services, highlighting the public’s concern for the accessibility of EMS.
Dr. Weiner stressed the need for a complete realignment of the payment system: “It’s unfair that public insurance like Medicaid reimburses sometimes less than a quarter of the rate that commercial insurance pays for the exact same service. This exacerbates and perpetuates disparities in health.” According to him, the solution may involve transitioning to a single-payer model, but it could face significant challenges owing to people’s apprehensions about changes in their health care and negative perceptions of “socialized” medicine in other countries. Furthermore, the poll found that the largest share of adults (42%) believed that hospitals should take the lead in improving boarding and shortening ED wait times. These findings indicated the need for hospitals to reevaluate their processes and capabilities to reduce boarding and enhance the overall patient experience in EDs.
“The top three entities that can fix the boarding crisis are hospitals, Congress, and insurance companies,” noted Dr. Wiener. “However, until there is parity at all levels, hospitals will continue to accept lucrative elective admissions but allow ED patients to linger without a bed,” he added.
A version of this article first appeared on Medscape.com.
ccording to a poll conducted on behalf of the American College of Emergency Physicians in September 2023. This issue not only affects patient care but also has far-reaching consequences for the efficiency of emergency medical services (EMS).
The survey interviewed 2,164 adults and showed that an overwhelming majority (80%) expressed serious concerns about the boarding crisis. Moreover, 43% of respondents either delayed seeking medical care at an ED or avoided it altogether if they anticipated prolonged wait times before being admitted to the hospital or transferred to another facility.
Nearly half of adults (44%) experienced long wait times following initial care in an ED, and 16% of these adults reported 13 or more hours of waiting after receiving initial care.
“The boarding crisis is a predictable result of an acute care hospital system with insufficient capacity – we lack enough space and staff in our acute care hospitals, as we have not created the bed capacity needed for an aging and higher acuity patient population, and staffing shortages for inpatient beds have resulted in a longer hospital length of stay that we observe as boarding patients in the ED,” Arjun Venkatesh, MD, chief of emergency services at Yale New Haven Hospital, told this news organization, commenting on the factors contributing to this crisis.
One concerning side effect of boarding in EDs is the delayed response of ambulance services. When a hospital is unprepared to receive patients arriving in an ambulance, ambulance crews often wait with the patients for extended periods until the hospital can admit them. This situation can have critical implications, as parked ambulances are unable to respond to other emergencies in the community.
Adults who have endured long wait times in EDs voiced concerns about the negative impact such delays can have on their medical care. The experiences of patients and their families have underscored the urgency of addressing the boarding crisis.
“There are no low-hanging fruit solutions or simple communication strategies to alleviate these concerns,” said Dr. Venkatesh, adding that “the only way to change that perception is to change the care by addressing the lack of inpatient capacity and community care capacity to avoid back-up in the ED.”
The poll revealed that 93% of adults across different demographic groups considered EMS, including paramedics, essential. Older adults were more likely to emphasize the significance of these services than were younger adults.
“We all need a place for people to go when there is an emergency, whether it be trauma, a heart attack, a stroke, or similar conditions,” said Scott Weiner, MD, associate professor of emergency medicine at Harvard Medical School and attending emergency physician at Brigham and Women’s Hospital, Boston.
“However, the modern ED is much more than that. It has become the center for coordination of care across the health system where diagnostic tests can be completed promptly, where follow-up care is arranged, and where behavioral health emergencies come,” he added.
A vast majority, 89%, believed that additional or supplemental government funding should be directed toward these essential services, highlighting the public’s concern for the accessibility of EMS.
Dr. Weiner stressed the need for a complete realignment of the payment system: “It’s unfair that public insurance like Medicaid reimburses sometimes less than a quarter of the rate that commercial insurance pays for the exact same service. This exacerbates and perpetuates disparities in health.” According to him, the solution may involve transitioning to a single-payer model, but it could face significant challenges owing to people’s apprehensions about changes in their health care and negative perceptions of “socialized” medicine in other countries. Furthermore, the poll found that the largest share of adults (42%) believed that hospitals should take the lead in improving boarding and shortening ED wait times. These findings indicated the need for hospitals to reevaluate their processes and capabilities to reduce boarding and enhance the overall patient experience in EDs.
“The top three entities that can fix the boarding crisis are hospitals, Congress, and insurance companies,” noted Dr. Wiener. “However, until there is parity at all levels, hospitals will continue to accept lucrative elective admissions but allow ED patients to linger without a bed,” he added.
A version of this article first appeared on Medscape.com.
ccording to a poll conducted on behalf of the American College of Emergency Physicians in September 2023. This issue not only affects patient care but also has far-reaching consequences for the efficiency of emergency medical services (EMS).
The survey interviewed 2,164 adults and showed that an overwhelming majority (80%) expressed serious concerns about the boarding crisis. Moreover, 43% of respondents either delayed seeking medical care at an ED or avoided it altogether if they anticipated prolonged wait times before being admitted to the hospital or transferred to another facility.
Nearly half of adults (44%) experienced long wait times following initial care in an ED, and 16% of these adults reported 13 or more hours of waiting after receiving initial care.
“The boarding crisis is a predictable result of an acute care hospital system with insufficient capacity – we lack enough space and staff in our acute care hospitals, as we have not created the bed capacity needed for an aging and higher acuity patient population, and staffing shortages for inpatient beds have resulted in a longer hospital length of stay that we observe as boarding patients in the ED,” Arjun Venkatesh, MD, chief of emergency services at Yale New Haven Hospital, told this news organization, commenting on the factors contributing to this crisis.
One concerning side effect of boarding in EDs is the delayed response of ambulance services. When a hospital is unprepared to receive patients arriving in an ambulance, ambulance crews often wait with the patients for extended periods until the hospital can admit them. This situation can have critical implications, as parked ambulances are unable to respond to other emergencies in the community.
Adults who have endured long wait times in EDs voiced concerns about the negative impact such delays can have on their medical care. The experiences of patients and their families have underscored the urgency of addressing the boarding crisis.
“There are no low-hanging fruit solutions or simple communication strategies to alleviate these concerns,” said Dr. Venkatesh, adding that “the only way to change that perception is to change the care by addressing the lack of inpatient capacity and community care capacity to avoid back-up in the ED.”
The poll revealed that 93% of adults across different demographic groups considered EMS, including paramedics, essential. Older adults were more likely to emphasize the significance of these services than were younger adults.
“We all need a place for people to go when there is an emergency, whether it be trauma, a heart attack, a stroke, or similar conditions,” said Scott Weiner, MD, associate professor of emergency medicine at Harvard Medical School and attending emergency physician at Brigham and Women’s Hospital, Boston.
“However, the modern ED is much more than that. It has become the center for coordination of care across the health system where diagnostic tests can be completed promptly, where follow-up care is arranged, and where behavioral health emergencies come,” he added.
A vast majority, 89%, believed that additional or supplemental government funding should be directed toward these essential services, highlighting the public’s concern for the accessibility of EMS.
Dr. Weiner stressed the need for a complete realignment of the payment system: “It’s unfair that public insurance like Medicaid reimburses sometimes less than a quarter of the rate that commercial insurance pays for the exact same service. This exacerbates and perpetuates disparities in health.” According to him, the solution may involve transitioning to a single-payer model, but it could face significant challenges owing to people’s apprehensions about changes in their health care and negative perceptions of “socialized” medicine in other countries. Furthermore, the poll found that the largest share of adults (42%) believed that hospitals should take the lead in improving boarding and shortening ED wait times. These findings indicated the need for hospitals to reevaluate their processes and capabilities to reduce boarding and enhance the overall patient experience in EDs.
“The top three entities that can fix the boarding crisis are hospitals, Congress, and insurance companies,” noted Dr. Wiener. “However, until there is parity at all levels, hospitals will continue to accept lucrative elective admissions but allow ED patients to linger without a bed,” he added.
A version of this article first appeared on Medscape.com.
Sensory comeback: New findings show the path to smell and taste recovery after COVID
Good news for people struggling with sensory problems after a bout of COVID-19. Although mild cases of the disease often impair the ability to taste and smell, and the problem can drag on for months, a new study from Italy shows that most people return to their senses, as it were, within 3 years.
said Paolo Boscolo-Rizzo, MD, a professor of medicine, surgery, and health sciences at the University of Trieste (Italy), and a co-author of the study, published as a research letter in JAMA Otolaryngology–Head & Neck Surgery.
Dr. Boscolo-Rizzo and his colleagues analyzed data from 88 adults with mild COVID-19, which was defined as having no lower respiratory disease and blood oxygen saturation of 94% or greater. Another group of 88 adults who never contracted the virus but sometimes had difficulties with smell and taste were also studied. In both groups, the average age was 49 years, all participants were White, and 58% were women.
The researchers tested participants’ sense of smell with sticks that contained different odors and checked their sense of taste with strips that had different tastes. Over time, fewer people had difficulty distinguishing odors. Three years after developing COVID-19, only 12 people had impaired smell, compared with 36 people at year 1 and 24 people at year 2. And at the 3-year mark, all participants had at least a partial ability to smell.
The story was similar with sense of taste, with 10 of 88 people reporting impairments 3 years later. By then, people with COVID-19 were no more likely to have trouble with smell or taste than people who did not get the virus.
A study this past June showed a strong correlation between severity of COVID-19 symptoms and impaired sense of taste and smell and estimated that millions of Americans maintained altered senses. More than 10% of people in the Italian study still had trouble with smell or taste 3 years later.
Emerging treatments, psychological concerns
“We’re seeing fewer people with this problem, but there are still people suffering from it,” said Fernando Carnavali, MD, an internal medicine physician and a site director for the Center for Post-COVID Care at the Icahn School of Medicine at Mount Sinai, New York City.
Dr. Carnavali wasn’t part of this study, but he did find the new results encouraging, and he called for similar studies in diverse populations that have experienced COVID-19. He also noted that an impaired sense of smell is distressing.
“It really has a significant psychological impact,” Dr. Carnavali said.
He recalled a patient crying in his office because her inability to smell made it impossible for her to cook. Dr. Carnavali recommended clinicians refer patients facing protracted loss of smell or taste to mental health professionals for support.
Treatments are emerging for COVID-19 smell loss. One approach is to inject platelet-rich plasma into a patient’s nasal cavities to help neurons related to smell repair themselves.
A randomized trial showed platelet-rich plasma significantly outperformed placebo in patients with smell loss up to a year after getting COVID-19.
“I wish more people would do it,” said Zara Patel, MD, an otolaryngologist at Stanford (Calif.) Medicine, who helped conduct that trial. She said some physicians may be nervous about injecting plasma so close to the skull and are therefore hesitant to try this approach.
Another technique may help to address the olfactory condition known as parosmia, in which patients generally experience a benign odor as rancid, according to otolaryngologist Nyssa Farrell, MD, of Washington University School of Medicine, St. Louis. Dr. Farrell said around two-thirds of patients who contract COVID-19 develop the condition, and the rates of long-term parosmia range from 10%-50% depending on various studies.
“It is almost always foul; this can profoundly affect someone’s quality of life,” impairing their ability to eat or to be intimate with a partner who now smells unpleasant, said Dr. Farrell, who wasn’t associated with this research.
The treatment, called a stellate ganglion block, is provided through a shot into nerves in the neck. People with parosmia associated with COVID-19 often report that this method cures them. Dr. Patel said that may be because their psychological health is improving, not their sense of smell, because the area of the body where the stellate ganglion block is applied is not part of the olfactory system.
Earlier this year, Dr. Farrell and colleagues reported that parosmia linked to COVID-19 is associated with an increased risk for depression, anxiety, and suicidal ideation.
One coauthor reported receiving grants from Smell and Taste Lab, Takasago, Baia Foods, and Frequency Therapeutics. The other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Good news for people struggling with sensory problems after a bout of COVID-19. Although mild cases of the disease often impair the ability to taste and smell, and the problem can drag on for months, a new study from Italy shows that most people return to their senses, as it were, within 3 years.
said Paolo Boscolo-Rizzo, MD, a professor of medicine, surgery, and health sciences at the University of Trieste (Italy), and a co-author of the study, published as a research letter in JAMA Otolaryngology–Head & Neck Surgery.
Dr. Boscolo-Rizzo and his colleagues analyzed data from 88 adults with mild COVID-19, which was defined as having no lower respiratory disease and blood oxygen saturation of 94% or greater. Another group of 88 adults who never contracted the virus but sometimes had difficulties with smell and taste were also studied. In both groups, the average age was 49 years, all participants were White, and 58% were women.
The researchers tested participants’ sense of smell with sticks that contained different odors and checked their sense of taste with strips that had different tastes. Over time, fewer people had difficulty distinguishing odors. Three years after developing COVID-19, only 12 people had impaired smell, compared with 36 people at year 1 and 24 people at year 2. And at the 3-year mark, all participants had at least a partial ability to smell.
The story was similar with sense of taste, with 10 of 88 people reporting impairments 3 years later. By then, people with COVID-19 were no more likely to have trouble with smell or taste than people who did not get the virus.
A study this past June showed a strong correlation between severity of COVID-19 symptoms and impaired sense of taste and smell and estimated that millions of Americans maintained altered senses. More than 10% of people in the Italian study still had trouble with smell or taste 3 years later.
Emerging treatments, psychological concerns
“We’re seeing fewer people with this problem, but there are still people suffering from it,” said Fernando Carnavali, MD, an internal medicine physician and a site director for the Center for Post-COVID Care at the Icahn School of Medicine at Mount Sinai, New York City.
Dr. Carnavali wasn’t part of this study, but he did find the new results encouraging, and he called for similar studies in diverse populations that have experienced COVID-19. He also noted that an impaired sense of smell is distressing.
“It really has a significant psychological impact,” Dr. Carnavali said.
He recalled a patient crying in his office because her inability to smell made it impossible for her to cook. Dr. Carnavali recommended clinicians refer patients facing protracted loss of smell or taste to mental health professionals for support.
Treatments are emerging for COVID-19 smell loss. One approach is to inject platelet-rich plasma into a patient’s nasal cavities to help neurons related to smell repair themselves.
A randomized trial showed platelet-rich plasma significantly outperformed placebo in patients with smell loss up to a year after getting COVID-19.
“I wish more people would do it,” said Zara Patel, MD, an otolaryngologist at Stanford (Calif.) Medicine, who helped conduct that trial. She said some physicians may be nervous about injecting plasma so close to the skull and are therefore hesitant to try this approach.
Another technique may help to address the olfactory condition known as parosmia, in which patients generally experience a benign odor as rancid, according to otolaryngologist Nyssa Farrell, MD, of Washington University School of Medicine, St. Louis. Dr. Farrell said around two-thirds of patients who contract COVID-19 develop the condition, and the rates of long-term parosmia range from 10%-50% depending on various studies.
“It is almost always foul; this can profoundly affect someone’s quality of life,” impairing their ability to eat or to be intimate with a partner who now smells unpleasant, said Dr. Farrell, who wasn’t associated with this research.
The treatment, called a stellate ganglion block, is provided through a shot into nerves in the neck. People with parosmia associated with COVID-19 often report that this method cures them. Dr. Patel said that may be because their psychological health is improving, not their sense of smell, because the area of the body where the stellate ganglion block is applied is not part of the olfactory system.
Earlier this year, Dr. Farrell and colleagues reported that parosmia linked to COVID-19 is associated with an increased risk for depression, anxiety, and suicidal ideation.
One coauthor reported receiving grants from Smell and Taste Lab, Takasago, Baia Foods, and Frequency Therapeutics. The other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Good news for people struggling with sensory problems after a bout of COVID-19. Although mild cases of the disease often impair the ability to taste and smell, and the problem can drag on for months, a new study from Italy shows that most people return to their senses, as it were, within 3 years.
said Paolo Boscolo-Rizzo, MD, a professor of medicine, surgery, and health sciences at the University of Trieste (Italy), and a co-author of the study, published as a research letter in JAMA Otolaryngology–Head & Neck Surgery.
Dr. Boscolo-Rizzo and his colleagues analyzed data from 88 adults with mild COVID-19, which was defined as having no lower respiratory disease and blood oxygen saturation of 94% or greater. Another group of 88 adults who never contracted the virus but sometimes had difficulties with smell and taste were also studied. In both groups, the average age was 49 years, all participants were White, and 58% were women.
The researchers tested participants’ sense of smell with sticks that contained different odors and checked their sense of taste with strips that had different tastes. Over time, fewer people had difficulty distinguishing odors. Three years after developing COVID-19, only 12 people had impaired smell, compared with 36 people at year 1 and 24 people at year 2. And at the 3-year mark, all participants had at least a partial ability to smell.
The story was similar with sense of taste, with 10 of 88 people reporting impairments 3 years later. By then, people with COVID-19 were no more likely to have trouble with smell or taste than people who did not get the virus.
A study this past June showed a strong correlation between severity of COVID-19 symptoms and impaired sense of taste and smell and estimated that millions of Americans maintained altered senses. More than 10% of people in the Italian study still had trouble with smell or taste 3 years later.
Emerging treatments, psychological concerns
“We’re seeing fewer people with this problem, but there are still people suffering from it,” said Fernando Carnavali, MD, an internal medicine physician and a site director for the Center for Post-COVID Care at the Icahn School of Medicine at Mount Sinai, New York City.
Dr. Carnavali wasn’t part of this study, but he did find the new results encouraging, and he called for similar studies in diverse populations that have experienced COVID-19. He also noted that an impaired sense of smell is distressing.
“It really has a significant psychological impact,” Dr. Carnavali said.
He recalled a patient crying in his office because her inability to smell made it impossible for her to cook. Dr. Carnavali recommended clinicians refer patients facing protracted loss of smell or taste to mental health professionals for support.
Treatments are emerging for COVID-19 smell loss. One approach is to inject platelet-rich plasma into a patient’s nasal cavities to help neurons related to smell repair themselves.
A randomized trial showed platelet-rich plasma significantly outperformed placebo in patients with smell loss up to a year after getting COVID-19.
“I wish more people would do it,” said Zara Patel, MD, an otolaryngologist at Stanford (Calif.) Medicine, who helped conduct that trial. She said some physicians may be nervous about injecting plasma so close to the skull and are therefore hesitant to try this approach.
Another technique may help to address the olfactory condition known as parosmia, in which patients generally experience a benign odor as rancid, according to otolaryngologist Nyssa Farrell, MD, of Washington University School of Medicine, St. Louis. Dr. Farrell said around two-thirds of patients who contract COVID-19 develop the condition, and the rates of long-term parosmia range from 10%-50% depending on various studies.
“It is almost always foul; this can profoundly affect someone’s quality of life,” impairing their ability to eat or to be intimate with a partner who now smells unpleasant, said Dr. Farrell, who wasn’t associated with this research.
The treatment, called a stellate ganglion block, is provided through a shot into nerves in the neck. People with parosmia associated with COVID-19 often report that this method cures them. Dr. Patel said that may be because their psychological health is improving, not their sense of smell, because the area of the body where the stellate ganglion block is applied is not part of the olfactory system.
Earlier this year, Dr. Farrell and colleagues reported that parosmia linked to COVID-19 is associated with an increased risk for depression, anxiety, and suicidal ideation.
One coauthor reported receiving grants from Smell and Taste Lab, Takasago, Baia Foods, and Frequency Therapeutics. The other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA OTOLARYNGOLOGY–HEAD & NECK SURGERY
Women have worse outcomes in cardiogenic shock
“These data identify the need for us to continue working to identify barriers in terms of diagnosis, management, and technological innovations for women in cardiogenic shock to resolve these issues and improve outcomes,” the senior author of the study, Navin Kapur, MD, Tufts Medical Center, Boston, said in an interview.
The study is said to be the one of the largest contemporary analyses of real-world registry data on the characteristics and outcomes of women in comparison with men with cardiogenic shock.
It showed sex-specific differences in outcomes that were primarily driven by differences in heart failure–related cardiogenic shock. Women with heart failure–related cardiogenic shock had more severe cardiogenic shock, worse survival at discharge, and more vascular complications than men. Outcomes in cardiogenic shock related to MI were similar for men and women.
The study, which will be presented at the upcoming annual meeting of the American Heart Association, was published online in JACC: Heart Failure.
Dr. Kapur founded the Cardiogenic Shock Working Group in 2017 to collect quality data on the condition.
“We realized our patients were dying, and we didn’t have enough data on how best to manage them. So, we started this registry, and now have detailed data on close to 9,000 patients with cardiogenic shock from 45 hospitals in the U.S., Mexico, Australia, and Japan,” he explained.
“The primary goal is to try to investigate the questions related to cardiogenic shock that can inform management, and one of the key questions that came up was differences in how men and women present with cardiogenic shock and what their outcomes may be. This is what we are reporting in this paper,” he added.
Cardiogenic shock is defined as having a low cardiac output most commonly because of MI or an episode of acute heart failure, Dr. Kapur said. Patients with cardiogenic shock are identified by their low blood pressure or hypoperfusion evidenced by clinical exam or biomarkers, such as elevated lactate levels.
“In this analysis, we’re looking at patients presenting with cardiogenic shock, so were not looking at the incidence of the condition in men versus women,” Dr. Kapur noted. “However, we believe that cardiogenic shock is probably more underrepresented in women, who may present with an MI or acute heart failure and may or may not be identified as having low cardiac output states until quite late. The likelihood is that the incidence is similar in men and women, but women are more often undiagnosed.”
For the current study, the authors analyzed data on 5,083 patients with cardiogenic shock in the registry, of whom 1,522 (30%) were women. Compared with men, women had slightly higher body mass index (BMI) and smaller body surface area.
Results showed that women with heart failure–related cardiogenic shock had worse survival at discharge than men (69.9% vs. 74.4%) and a higher rate of refractory shock (SCAI stage E; 26% vs. 21%). Women were also less likely to undergo pulmonary artery catheterization (52.9% vs. 54.6%), heart transplantation (6.5% vs. 10.3%), or left ventricular assist device implantation (7.8% vs. 10%).
Regardless of cardiogenic shock etiology, women had more vascular complications (8.8% vs. 5.7%), bleeding (7.1% vs. 5.2%), and limb ischemia (6.8% vs. 4.5%).
“This analysis is quite revealing. We identified some important distinctions between men and women,” Dr. Kapur commented.
For many patients who present with MI-related cardiogenic shock, many of the baseline characteristics in men and women were quite similar, he said. “But in heart failure–related cardiogenic shock, we saw more differences, with typical comorbidities associated with cardiogenic shock [e.g., diabetes, chronic kidney disease, hypertension] being less common in women than in men. This suggests there may be phenotypic differences as to why women present with heart failure shock versus men.”
Dr. Kapur pointed out that differences in BMI or body surface area between men and women may play into some of the management decision-making.
“Women having a smaller stature may lead to a selection bias where we don’t want to use large-bore pumps or devices because we’re worried about causing complications. We found in the analysis that vascular complications such as bleeding or ischemia of the lower extremity where these devices typically go were more frequent in women,” he noted.
“We also found that women were less likely to receive invasive therapies in general, including pulmonary artery catheters, temporary mechanical support, and heart replacements, such as LVAD or transplants,” he added.
Further results showed that, after propensity score matching, some of the gender differences disappeared, but women continued to have a higher rate of vascular complications (10.4% women vs. 7.4% men).
But Dr. Kapur warned that the propensity-matched analysis had some caveats.
“Essentially what we are doing with propensity matching is creating two populations that are as similar as possible, and this reduced the number of patients in the analysis down to 25% of the original population,” he said. “One of the things we had to match was body surface area, and in doing this, we are taking out one of the most important differences between men and women, and as a result, a lot of the differences in outcomes go away.
“In this respect, propensity matching can be a bit of a double-edge sword,” he added. “I think the non–propensity-matched results are more interesting, as they are more of a reflection of the real world.”
Dr. Kapur concluded that these findings are compelling enough to suggest that there are important differences between women and men with cardiogenic shock in terms of outcomes as well as complication rates.
“Our decision-making around women seems to be different to that around men. I think this paper should start to trigger more awareness of that.”
Dr. Kapur also emphasized the importance of paying attention to vascular complications in women.
“The higher rates of bleeding and limb ischemia issues in women may explain the rationale for being less aggressive with invasive therapies in women,” he said. “But we need to come up with better solutions or technologies so they can be used more effectively in women. This could include adapting technology for smaller vascular sizes, which should lead to better outcome and fewer complications in women.”
He added that further granular data on this issue are needed. “We have very limited datasets in cardiogenic shock. There are few randomized controlled trials, and women are not well represented in such trials. We need to make sure we enroll women in randomized trials.”
Dr. Kapur said more women physicians who treat cardiogenic shock are also required, which would include cardiologists, critical care specialists, cardiac surgeons, and anesthesia personnel.
He pointed out that the two first authors of the current study are women – Van-Khue Ton, MD, Massachusetts General Hospital, Boston, and Manreet Kanwar, MD, Allegheny Health Network, Pittsburgh.
“We worked hard to involve women as principal investigators. They led the effort. These are investigations led by women, on women, to advance the care of women,” he commented.
Gender-related inequality
In an editorial accompanying publication of the study, Sara Kalantari, MD, and Jonathan Grinstein, MD, University of Chicago, and Robert O. Roswell, MD, Hofstra University, Hempstead, N.Y., said these results “provide valuable information about gender-related inequality in care and outcomes in the management of cardiogenic shock, although the exact mechanisms driving these observed differences still need to be elucidated.
“Broadly speaking, barriers in the care of women with heart failure and cardiogenic shock include a reduced awareness among both patients and providers, a deficiency of sex-specific objective criteria for guiding therapy, and unfavorable temporary mechanical circulatory support devices with higher rates of hemocompatibility-related complications in women,” they added.
“In the era of the multidisciplinary shock team and shock pathways with protocolized management algorithms, it is imperative that we still allow for personalization of care to match the physiologic needs of the patient in order for us to continue to close the gender gap in the care of patients presenting with cardiogenic shock,” the editorialists concluded.
A version of this article appeared on Medscape.com.
“These data identify the need for us to continue working to identify barriers in terms of diagnosis, management, and technological innovations for women in cardiogenic shock to resolve these issues and improve outcomes,” the senior author of the study, Navin Kapur, MD, Tufts Medical Center, Boston, said in an interview.
The study is said to be the one of the largest contemporary analyses of real-world registry data on the characteristics and outcomes of women in comparison with men with cardiogenic shock.
It showed sex-specific differences in outcomes that were primarily driven by differences in heart failure–related cardiogenic shock. Women with heart failure–related cardiogenic shock had more severe cardiogenic shock, worse survival at discharge, and more vascular complications than men. Outcomes in cardiogenic shock related to MI were similar for men and women.
The study, which will be presented at the upcoming annual meeting of the American Heart Association, was published online in JACC: Heart Failure.
Dr. Kapur founded the Cardiogenic Shock Working Group in 2017 to collect quality data on the condition.
“We realized our patients were dying, and we didn’t have enough data on how best to manage them. So, we started this registry, and now have detailed data on close to 9,000 patients with cardiogenic shock from 45 hospitals in the U.S., Mexico, Australia, and Japan,” he explained.
“The primary goal is to try to investigate the questions related to cardiogenic shock that can inform management, and one of the key questions that came up was differences in how men and women present with cardiogenic shock and what their outcomes may be. This is what we are reporting in this paper,” he added.
Cardiogenic shock is defined as having a low cardiac output most commonly because of MI or an episode of acute heart failure, Dr. Kapur said. Patients with cardiogenic shock are identified by their low blood pressure or hypoperfusion evidenced by clinical exam or biomarkers, such as elevated lactate levels.
“In this analysis, we’re looking at patients presenting with cardiogenic shock, so were not looking at the incidence of the condition in men versus women,” Dr. Kapur noted. “However, we believe that cardiogenic shock is probably more underrepresented in women, who may present with an MI or acute heart failure and may or may not be identified as having low cardiac output states until quite late. The likelihood is that the incidence is similar in men and women, but women are more often undiagnosed.”
For the current study, the authors analyzed data on 5,083 patients with cardiogenic shock in the registry, of whom 1,522 (30%) were women. Compared with men, women had slightly higher body mass index (BMI) and smaller body surface area.
Results showed that women with heart failure–related cardiogenic shock had worse survival at discharge than men (69.9% vs. 74.4%) and a higher rate of refractory shock (SCAI stage E; 26% vs. 21%). Women were also less likely to undergo pulmonary artery catheterization (52.9% vs. 54.6%), heart transplantation (6.5% vs. 10.3%), or left ventricular assist device implantation (7.8% vs. 10%).
Regardless of cardiogenic shock etiology, women had more vascular complications (8.8% vs. 5.7%), bleeding (7.1% vs. 5.2%), and limb ischemia (6.8% vs. 4.5%).
“This analysis is quite revealing. We identified some important distinctions between men and women,” Dr. Kapur commented.
For many patients who present with MI-related cardiogenic shock, many of the baseline characteristics in men and women were quite similar, he said. “But in heart failure–related cardiogenic shock, we saw more differences, with typical comorbidities associated with cardiogenic shock [e.g., diabetes, chronic kidney disease, hypertension] being less common in women than in men. This suggests there may be phenotypic differences as to why women present with heart failure shock versus men.”
Dr. Kapur pointed out that differences in BMI or body surface area between men and women may play into some of the management decision-making.
“Women having a smaller stature may lead to a selection bias where we don’t want to use large-bore pumps or devices because we’re worried about causing complications. We found in the analysis that vascular complications such as bleeding or ischemia of the lower extremity where these devices typically go were more frequent in women,” he noted.
“We also found that women were less likely to receive invasive therapies in general, including pulmonary artery catheters, temporary mechanical support, and heart replacements, such as LVAD or transplants,” he added.
Further results showed that, after propensity score matching, some of the gender differences disappeared, but women continued to have a higher rate of vascular complications (10.4% women vs. 7.4% men).
But Dr. Kapur warned that the propensity-matched analysis had some caveats.
“Essentially what we are doing with propensity matching is creating two populations that are as similar as possible, and this reduced the number of patients in the analysis down to 25% of the original population,” he said. “One of the things we had to match was body surface area, and in doing this, we are taking out one of the most important differences between men and women, and as a result, a lot of the differences in outcomes go away.
“In this respect, propensity matching can be a bit of a double-edge sword,” he added. “I think the non–propensity-matched results are more interesting, as they are more of a reflection of the real world.”
Dr. Kapur concluded that these findings are compelling enough to suggest that there are important differences between women and men with cardiogenic shock in terms of outcomes as well as complication rates.
“Our decision-making around women seems to be different to that around men. I think this paper should start to trigger more awareness of that.”
Dr. Kapur also emphasized the importance of paying attention to vascular complications in women.
“The higher rates of bleeding and limb ischemia issues in women may explain the rationale for being less aggressive with invasive therapies in women,” he said. “But we need to come up with better solutions or technologies so they can be used more effectively in women. This could include adapting technology for smaller vascular sizes, which should lead to better outcome and fewer complications in women.”
He added that further granular data on this issue are needed. “We have very limited datasets in cardiogenic shock. There are few randomized controlled trials, and women are not well represented in such trials. We need to make sure we enroll women in randomized trials.”
Dr. Kapur said more women physicians who treat cardiogenic shock are also required, which would include cardiologists, critical care specialists, cardiac surgeons, and anesthesia personnel.
He pointed out that the two first authors of the current study are women – Van-Khue Ton, MD, Massachusetts General Hospital, Boston, and Manreet Kanwar, MD, Allegheny Health Network, Pittsburgh.
“We worked hard to involve women as principal investigators. They led the effort. These are investigations led by women, on women, to advance the care of women,” he commented.
Gender-related inequality
In an editorial accompanying publication of the study, Sara Kalantari, MD, and Jonathan Grinstein, MD, University of Chicago, and Robert O. Roswell, MD, Hofstra University, Hempstead, N.Y., said these results “provide valuable information about gender-related inequality in care and outcomes in the management of cardiogenic shock, although the exact mechanisms driving these observed differences still need to be elucidated.
“Broadly speaking, barriers in the care of women with heart failure and cardiogenic shock include a reduced awareness among both patients and providers, a deficiency of sex-specific objective criteria for guiding therapy, and unfavorable temporary mechanical circulatory support devices with higher rates of hemocompatibility-related complications in women,” they added.
“In the era of the multidisciplinary shock team and shock pathways with protocolized management algorithms, it is imperative that we still allow for personalization of care to match the physiologic needs of the patient in order for us to continue to close the gender gap in the care of patients presenting with cardiogenic shock,” the editorialists concluded.
A version of this article appeared on Medscape.com.
“These data identify the need for us to continue working to identify barriers in terms of diagnosis, management, and technological innovations for women in cardiogenic shock to resolve these issues and improve outcomes,” the senior author of the study, Navin Kapur, MD, Tufts Medical Center, Boston, said in an interview.
The study is said to be the one of the largest contemporary analyses of real-world registry data on the characteristics and outcomes of women in comparison with men with cardiogenic shock.
It showed sex-specific differences in outcomes that were primarily driven by differences in heart failure–related cardiogenic shock. Women with heart failure–related cardiogenic shock had more severe cardiogenic shock, worse survival at discharge, and more vascular complications than men. Outcomes in cardiogenic shock related to MI were similar for men and women.
The study, which will be presented at the upcoming annual meeting of the American Heart Association, was published online in JACC: Heart Failure.
Dr. Kapur founded the Cardiogenic Shock Working Group in 2017 to collect quality data on the condition.
“We realized our patients were dying, and we didn’t have enough data on how best to manage them. So, we started this registry, and now have detailed data on close to 9,000 patients with cardiogenic shock from 45 hospitals in the U.S., Mexico, Australia, and Japan,” he explained.
“The primary goal is to try to investigate the questions related to cardiogenic shock that can inform management, and one of the key questions that came up was differences in how men and women present with cardiogenic shock and what their outcomes may be. This is what we are reporting in this paper,” he added.
Cardiogenic shock is defined as having a low cardiac output most commonly because of MI or an episode of acute heart failure, Dr. Kapur said. Patients with cardiogenic shock are identified by their low blood pressure or hypoperfusion evidenced by clinical exam or biomarkers, such as elevated lactate levels.
“In this analysis, we’re looking at patients presenting with cardiogenic shock, so were not looking at the incidence of the condition in men versus women,” Dr. Kapur noted. “However, we believe that cardiogenic shock is probably more underrepresented in women, who may present with an MI or acute heart failure and may or may not be identified as having low cardiac output states until quite late. The likelihood is that the incidence is similar in men and women, but women are more often undiagnosed.”
For the current study, the authors analyzed data on 5,083 patients with cardiogenic shock in the registry, of whom 1,522 (30%) were women. Compared with men, women had slightly higher body mass index (BMI) and smaller body surface area.
Results showed that women with heart failure–related cardiogenic shock had worse survival at discharge than men (69.9% vs. 74.4%) and a higher rate of refractory shock (SCAI stage E; 26% vs. 21%). Women were also less likely to undergo pulmonary artery catheterization (52.9% vs. 54.6%), heart transplantation (6.5% vs. 10.3%), or left ventricular assist device implantation (7.8% vs. 10%).
Regardless of cardiogenic shock etiology, women had more vascular complications (8.8% vs. 5.7%), bleeding (7.1% vs. 5.2%), and limb ischemia (6.8% vs. 4.5%).
“This analysis is quite revealing. We identified some important distinctions between men and women,” Dr. Kapur commented.
For many patients who present with MI-related cardiogenic shock, many of the baseline characteristics in men and women were quite similar, he said. “But in heart failure–related cardiogenic shock, we saw more differences, with typical comorbidities associated with cardiogenic shock [e.g., diabetes, chronic kidney disease, hypertension] being less common in women than in men. This suggests there may be phenotypic differences as to why women present with heart failure shock versus men.”
Dr. Kapur pointed out that differences in BMI or body surface area between men and women may play into some of the management decision-making.
“Women having a smaller stature may lead to a selection bias where we don’t want to use large-bore pumps or devices because we’re worried about causing complications. We found in the analysis that vascular complications such as bleeding or ischemia of the lower extremity where these devices typically go were more frequent in women,” he noted.
“We also found that women were less likely to receive invasive therapies in general, including pulmonary artery catheters, temporary mechanical support, and heart replacements, such as LVAD or transplants,” he added.
Further results showed that, after propensity score matching, some of the gender differences disappeared, but women continued to have a higher rate of vascular complications (10.4% women vs. 7.4% men).
But Dr. Kapur warned that the propensity-matched analysis had some caveats.
“Essentially what we are doing with propensity matching is creating two populations that are as similar as possible, and this reduced the number of patients in the analysis down to 25% of the original population,” he said. “One of the things we had to match was body surface area, and in doing this, we are taking out one of the most important differences between men and women, and as a result, a lot of the differences in outcomes go away.
“In this respect, propensity matching can be a bit of a double-edge sword,” he added. “I think the non–propensity-matched results are more interesting, as they are more of a reflection of the real world.”
Dr. Kapur concluded that these findings are compelling enough to suggest that there are important differences between women and men with cardiogenic shock in terms of outcomes as well as complication rates.
“Our decision-making around women seems to be different to that around men. I think this paper should start to trigger more awareness of that.”
Dr. Kapur also emphasized the importance of paying attention to vascular complications in women.
“The higher rates of bleeding and limb ischemia issues in women may explain the rationale for being less aggressive with invasive therapies in women,” he said. “But we need to come up with better solutions or technologies so they can be used more effectively in women. This could include adapting technology for smaller vascular sizes, which should lead to better outcome and fewer complications in women.”
He added that further granular data on this issue are needed. “We have very limited datasets in cardiogenic shock. There are few randomized controlled trials, and women are not well represented in such trials. We need to make sure we enroll women in randomized trials.”
Dr. Kapur said more women physicians who treat cardiogenic shock are also required, which would include cardiologists, critical care specialists, cardiac surgeons, and anesthesia personnel.
He pointed out that the two first authors of the current study are women – Van-Khue Ton, MD, Massachusetts General Hospital, Boston, and Manreet Kanwar, MD, Allegheny Health Network, Pittsburgh.
“We worked hard to involve women as principal investigators. They led the effort. These are investigations led by women, on women, to advance the care of women,” he commented.
Gender-related inequality
In an editorial accompanying publication of the study, Sara Kalantari, MD, and Jonathan Grinstein, MD, University of Chicago, and Robert O. Roswell, MD, Hofstra University, Hempstead, N.Y., said these results “provide valuable information about gender-related inequality in care and outcomes in the management of cardiogenic shock, although the exact mechanisms driving these observed differences still need to be elucidated.
“Broadly speaking, barriers in the care of women with heart failure and cardiogenic shock include a reduced awareness among both patients and providers, a deficiency of sex-specific objective criteria for guiding therapy, and unfavorable temporary mechanical circulatory support devices with higher rates of hemocompatibility-related complications in women,” they added.
“In the era of the multidisciplinary shock team and shock pathways with protocolized management algorithms, it is imperative that we still allow for personalization of care to match the physiologic needs of the patient in order for us to continue to close the gender gap in the care of patients presenting with cardiogenic shock,” the editorialists concluded.
A version of this article appeared on Medscape.com.
FROM AHA 2023
A new long COVID explanation: Low serotonin levels?
Could antidepressants hold the key to treating long COVID? The study even points to a possible treatment.
Serotonin is a neurotransmitter that has many functions in the body and is targeted by the most commonly prescribed antidepressants – the selective serotonin reuptake inhibitors.
Serotonin is widely studied for its effects on the brain – it regulates the messaging between neurons, affecting sleep, mood, and memory. It is present in the gut, is found in cells along the gastrointestinal tract, and is absorbed by blood platelets. Gut serotonin, known as circulating serotonin, is responsible for a host of other functions, including the regulation of blood flow, body temperature, and digestion.
Low levels of serotonin could result in any number of seemingly unrelated symptoms, as in the case of long COVID, experts say. The condition affects about 7% of Americans and is associated with a wide range of health problems, including fatigue, shortness of breath, neurological symptoms, joint pain, blood clots, heart palpitations, and digestive problems.
Long COVID is difficult to treat because researchers haven’t been able to pinpoint the underlying mechanisms that cause prolonged illness after a SARS-CoV-2 infection, said study author Christoph A. Thaiss, PhD, an assistant professor of microbiology at the Perelman School of Medicine at the University of Pennsylvania.
The hope is that this study could have implications for new treatments, he said.
“Long COVID can have manifestations not only in the brain but in many different parts of the body, so it’s possible that serotonin reductions are involved in many different aspects of the disease,” said Dr. Thaiss.
Dr. Thaiss’s study, published in the journal Cell, found lower serotonin levels in long COVID patients, compared with patients who were diagnosed with acute COVID-19 but who fully recovered.
His team found that reductions in serotonin were driven by low levels of circulating SARS-CoV-2 virus that caused persistent inflammation as well as an inability of the body to absorb tryptophan, an amino acid that’s a precursor to serotonin. Overactive blood platelets were also shown to play a role; they serve as the primary means of serotonin absorption.
The study doesn’t make any recommendations for treatment, but understanding the role of serotonin in long COVID opens the door to a host of novel ideas that could set the stage for clinical trials and affect care.
“The study gives us a few possible targets that could be used in future clinical studies,” Dr. Thaiss said.
Persistent circulating virus is one of the drivers of low serotonin levels, said study author Michael Peluso, MD, an assistant research professor of infectious medicine at the University of California, San Francisco, School of Medicine. This points to the need to reduce viral load using antiviral medications like nirmatrelvir/ritonavir (Paxlovid), which is approved by the U.S. Food and Drug Administration for the treatment of COVID-19, and VV116, which has not yet been approved for use against COVID.
Research published in the New England Journal of Medicine found that the oral antiviral agent VV116 was as effective as nirmatrelvir/ritonavir in reducing the body’s viral load and aiding recovery from SARS-CoV-2 infection. Paxlovid has also been shown to reduce the likelihood of getting long COVID after an acute SARS-CoV-2 infection.
Researchers are investigating ways to target serotonin levels directly, potentially using SSRIs. But first they need to study whether improvement in serotonin level makes a difference.
“What we need now is a good clinical trial to see whether altering levels of serotonin in people with long COVID will lead to symptom relief,” Dr. Peluso said.
Indeed, the research did show that the SSRI fluoxetine, as well as a glycine-tryptophan supplement, improved cognitive function in SARS-CoV-2-infected rodent models, which were used in a portion of the study.
David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City, said the research is helping “to paint a biological picture” that’s in line with other research on the mechanisms that cause long COVID symptoms.
But Dr. Putrino, who was not involved in the study, cautions against treating long COVID patients with SSRIs or any other treatment that increases serotonin before testing patients to determine whether their serotonin levels are actually lower than those of healthy persons.
“We don’t want to assume that every patient with long COVID is going to have lower serotonin levels,” said Dr. Putrino.
What’s more, researchers need to investigate whether SSRIs increase levels of circulating serotonin. It’s important to note that researchers found lower levels of circulating serotonin but that serotonin levels in the brain remained normal.
Traditionally, SSRIs are used clinically for increasing the levels of serotonin in the brain, not the body.
“Whether that’s going to contribute to an increase in systemic levels of serotonin, that’s something that needs to be tested,” said Akiko Iwasaki, PhD, co-lead investigator of the Yale School of Medicine, New Haven, Conn., COVID-19 Recovery Study, who was not involved in the research.
Thus far, investigators have not identified one unifying biomarker that seems to cause long COVID in all patients, said Dr. Iwasaki. Some research has found higher levels of certain immune cells and biomarkers: for example, monocytes and activated B lymphocytes, indicating a stronger and ongoing antibody response to the virus. Other recent research conducted by Dr. Iwasaki, Dr. Putrino, and others, published in the journal Nature, showed that long COVID patients tend to have lower levels of cortisol, which could be a factor in the extreme fatigue experienced by many who suffer from the condition.
The findings in the study in The Cell are promising, but they need to be replicated in more people, said Dr. Iwasaki. And even if they’re replicated in a larger study population, this would still be just one biomarker that is associated with one subtype of the disease. There is a need to better understand which biomarkers go with which symptoms so that the most effective treatments can be identified, she said.
Both Dr. Putrino and Dr. Iwasaki contended that there isn’t a single factor that can explain all of long COVID. It’s a complex disease caused by a host of different mechanisms.
Still, low levels of serotonin could be an important piece of the puzzle. The next step, said Dr. Iwasaki, is to uncover how many of the millions of Americans with long COVID have this biomarker.
“People working in the field of long COVID should now be considering this pathway and thinking of ways to measure serotonin in their patients.”
A version of this article first appeared on Medscape.com.
Could antidepressants hold the key to treating long COVID? The study even points to a possible treatment.
Serotonin is a neurotransmitter that has many functions in the body and is targeted by the most commonly prescribed antidepressants – the selective serotonin reuptake inhibitors.
Serotonin is widely studied for its effects on the brain – it regulates the messaging between neurons, affecting sleep, mood, and memory. It is present in the gut, is found in cells along the gastrointestinal tract, and is absorbed by blood platelets. Gut serotonin, known as circulating serotonin, is responsible for a host of other functions, including the regulation of blood flow, body temperature, and digestion.
Low levels of serotonin could result in any number of seemingly unrelated symptoms, as in the case of long COVID, experts say. The condition affects about 7% of Americans and is associated with a wide range of health problems, including fatigue, shortness of breath, neurological symptoms, joint pain, blood clots, heart palpitations, and digestive problems.
Long COVID is difficult to treat because researchers haven’t been able to pinpoint the underlying mechanisms that cause prolonged illness after a SARS-CoV-2 infection, said study author Christoph A. Thaiss, PhD, an assistant professor of microbiology at the Perelman School of Medicine at the University of Pennsylvania.
The hope is that this study could have implications for new treatments, he said.
“Long COVID can have manifestations not only in the brain but in many different parts of the body, so it’s possible that serotonin reductions are involved in many different aspects of the disease,” said Dr. Thaiss.
Dr. Thaiss’s study, published in the journal Cell, found lower serotonin levels in long COVID patients, compared with patients who were diagnosed with acute COVID-19 but who fully recovered.
His team found that reductions in serotonin were driven by low levels of circulating SARS-CoV-2 virus that caused persistent inflammation as well as an inability of the body to absorb tryptophan, an amino acid that’s a precursor to serotonin. Overactive blood platelets were also shown to play a role; they serve as the primary means of serotonin absorption.
The study doesn’t make any recommendations for treatment, but understanding the role of serotonin in long COVID opens the door to a host of novel ideas that could set the stage for clinical trials and affect care.
“The study gives us a few possible targets that could be used in future clinical studies,” Dr. Thaiss said.
Persistent circulating virus is one of the drivers of low serotonin levels, said study author Michael Peluso, MD, an assistant research professor of infectious medicine at the University of California, San Francisco, School of Medicine. This points to the need to reduce viral load using antiviral medications like nirmatrelvir/ritonavir (Paxlovid), which is approved by the U.S. Food and Drug Administration for the treatment of COVID-19, and VV116, which has not yet been approved for use against COVID.
Research published in the New England Journal of Medicine found that the oral antiviral agent VV116 was as effective as nirmatrelvir/ritonavir in reducing the body’s viral load and aiding recovery from SARS-CoV-2 infection. Paxlovid has also been shown to reduce the likelihood of getting long COVID after an acute SARS-CoV-2 infection.
Researchers are investigating ways to target serotonin levels directly, potentially using SSRIs. But first they need to study whether improvement in serotonin level makes a difference.
“What we need now is a good clinical trial to see whether altering levels of serotonin in people with long COVID will lead to symptom relief,” Dr. Peluso said.
Indeed, the research did show that the SSRI fluoxetine, as well as a glycine-tryptophan supplement, improved cognitive function in SARS-CoV-2-infected rodent models, which were used in a portion of the study.
David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City, said the research is helping “to paint a biological picture” that’s in line with other research on the mechanisms that cause long COVID symptoms.
But Dr. Putrino, who was not involved in the study, cautions against treating long COVID patients with SSRIs or any other treatment that increases serotonin before testing patients to determine whether their serotonin levels are actually lower than those of healthy persons.
“We don’t want to assume that every patient with long COVID is going to have lower serotonin levels,” said Dr. Putrino.
What’s more, researchers need to investigate whether SSRIs increase levels of circulating serotonin. It’s important to note that researchers found lower levels of circulating serotonin but that serotonin levels in the brain remained normal.
Traditionally, SSRIs are used clinically for increasing the levels of serotonin in the brain, not the body.
“Whether that’s going to contribute to an increase in systemic levels of serotonin, that’s something that needs to be tested,” said Akiko Iwasaki, PhD, co-lead investigator of the Yale School of Medicine, New Haven, Conn., COVID-19 Recovery Study, who was not involved in the research.
Thus far, investigators have not identified one unifying biomarker that seems to cause long COVID in all patients, said Dr. Iwasaki. Some research has found higher levels of certain immune cells and biomarkers: for example, monocytes and activated B lymphocytes, indicating a stronger and ongoing antibody response to the virus. Other recent research conducted by Dr. Iwasaki, Dr. Putrino, and others, published in the journal Nature, showed that long COVID patients tend to have lower levels of cortisol, which could be a factor in the extreme fatigue experienced by many who suffer from the condition.
The findings in the study in The Cell are promising, but they need to be replicated in more people, said Dr. Iwasaki. And even if they’re replicated in a larger study population, this would still be just one biomarker that is associated with one subtype of the disease. There is a need to better understand which biomarkers go with which symptoms so that the most effective treatments can be identified, she said.
Both Dr. Putrino and Dr. Iwasaki contended that there isn’t a single factor that can explain all of long COVID. It’s a complex disease caused by a host of different mechanisms.
Still, low levels of serotonin could be an important piece of the puzzle. The next step, said Dr. Iwasaki, is to uncover how many of the millions of Americans with long COVID have this biomarker.
“People working in the field of long COVID should now be considering this pathway and thinking of ways to measure serotonin in their patients.”
A version of this article first appeared on Medscape.com.
Could antidepressants hold the key to treating long COVID? The study even points to a possible treatment.
Serotonin is a neurotransmitter that has many functions in the body and is targeted by the most commonly prescribed antidepressants – the selective serotonin reuptake inhibitors.
Serotonin is widely studied for its effects on the brain – it regulates the messaging between neurons, affecting sleep, mood, and memory. It is present in the gut, is found in cells along the gastrointestinal tract, and is absorbed by blood platelets. Gut serotonin, known as circulating serotonin, is responsible for a host of other functions, including the regulation of blood flow, body temperature, and digestion.
Low levels of serotonin could result in any number of seemingly unrelated symptoms, as in the case of long COVID, experts say. The condition affects about 7% of Americans and is associated with a wide range of health problems, including fatigue, shortness of breath, neurological symptoms, joint pain, blood clots, heart palpitations, and digestive problems.
Long COVID is difficult to treat because researchers haven’t been able to pinpoint the underlying mechanisms that cause prolonged illness after a SARS-CoV-2 infection, said study author Christoph A. Thaiss, PhD, an assistant professor of microbiology at the Perelman School of Medicine at the University of Pennsylvania.
The hope is that this study could have implications for new treatments, he said.
“Long COVID can have manifestations not only in the brain but in many different parts of the body, so it’s possible that serotonin reductions are involved in many different aspects of the disease,” said Dr. Thaiss.
Dr. Thaiss’s study, published in the journal Cell, found lower serotonin levels in long COVID patients, compared with patients who were diagnosed with acute COVID-19 but who fully recovered.
His team found that reductions in serotonin were driven by low levels of circulating SARS-CoV-2 virus that caused persistent inflammation as well as an inability of the body to absorb tryptophan, an amino acid that’s a precursor to serotonin. Overactive blood platelets were also shown to play a role; they serve as the primary means of serotonin absorption.
The study doesn’t make any recommendations for treatment, but understanding the role of serotonin in long COVID opens the door to a host of novel ideas that could set the stage for clinical trials and affect care.
“The study gives us a few possible targets that could be used in future clinical studies,” Dr. Thaiss said.
Persistent circulating virus is one of the drivers of low serotonin levels, said study author Michael Peluso, MD, an assistant research professor of infectious medicine at the University of California, San Francisco, School of Medicine. This points to the need to reduce viral load using antiviral medications like nirmatrelvir/ritonavir (Paxlovid), which is approved by the U.S. Food and Drug Administration for the treatment of COVID-19, and VV116, which has not yet been approved for use against COVID.
Research published in the New England Journal of Medicine found that the oral antiviral agent VV116 was as effective as nirmatrelvir/ritonavir in reducing the body’s viral load and aiding recovery from SARS-CoV-2 infection. Paxlovid has also been shown to reduce the likelihood of getting long COVID after an acute SARS-CoV-2 infection.
Researchers are investigating ways to target serotonin levels directly, potentially using SSRIs. But first they need to study whether improvement in serotonin level makes a difference.
“What we need now is a good clinical trial to see whether altering levels of serotonin in people with long COVID will lead to symptom relief,” Dr. Peluso said.
Indeed, the research did show that the SSRI fluoxetine, as well as a glycine-tryptophan supplement, improved cognitive function in SARS-CoV-2-infected rodent models, which were used in a portion of the study.
David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City, said the research is helping “to paint a biological picture” that’s in line with other research on the mechanisms that cause long COVID symptoms.
But Dr. Putrino, who was not involved in the study, cautions against treating long COVID patients with SSRIs or any other treatment that increases serotonin before testing patients to determine whether their serotonin levels are actually lower than those of healthy persons.
“We don’t want to assume that every patient with long COVID is going to have lower serotonin levels,” said Dr. Putrino.
What’s more, researchers need to investigate whether SSRIs increase levels of circulating serotonin. It’s important to note that researchers found lower levels of circulating serotonin but that serotonin levels in the brain remained normal.
Traditionally, SSRIs are used clinically for increasing the levels of serotonin in the brain, not the body.
“Whether that’s going to contribute to an increase in systemic levels of serotonin, that’s something that needs to be tested,” said Akiko Iwasaki, PhD, co-lead investigator of the Yale School of Medicine, New Haven, Conn., COVID-19 Recovery Study, who was not involved in the research.
Thus far, investigators have not identified one unifying biomarker that seems to cause long COVID in all patients, said Dr. Iwasaki. Some research has found higher levels of certain immune cells and biomarkers: for example, monocytes and activated B lymphocytes, indicating a stronger and ongoing antibody response to the virus. Other recent research conducted by Dr. Iwasaki, Dr. Putrino, and others, published in the journal Nature, showed that long COVID patients tend to have lower levels of cortisol, which could be a factor in the extreme fatigue experienced by many who suffer from the condition.
The findings in the study in The Cell are promising, but they need to be replicated in more people, said Dr. Iwasaki. And even if they’re replicated in a larger study population, this would still be just one biomarker that is associated with one subtype of the disease. There is a need to better understand which biomarkers go with which symptoms so that the most effective treatments can be identified, she said.
Both Dr. Putrino and Dr. Iwasaki contended that there isn’t a single factor that can explain all of long COVID. It’s a complex disease caused by a host of different mechanisms.
Still, low levels of serotonin could be an important piece of the puzzle. The next step, said Dr. Iwasaki, is to uncover how many of the millions of Americans with long COVID have this biomarker.
“People working in the field of long COVID should now be considering this pathway and thinking of ways to measure serotonin in their patients.”
A version of this article first appeared on Medscape.com.
FROM CELL
Even one night in the ED raises risk for death
This transcript has been edited for clarity.
As a consulting nephrologist, I go all over the hospital. Medicine floors, surgical floors, the ICU – I’ve even done consults in the operating room. And more and more, I do consults in the emergency department.
The reason I am doing more consults in the ED is not because the ED docs are getting gun shy with creatinine increases; it’s because patients are staying for extended periods in the ED despite being formally admitted to the hospital. It’s a phenomenon known as boarding, because there are simply not enough beds. You know the scene if you have ever been to a busy hospital: The ED is full to breaking, with patients on stretchers in hallways. It can often feel more like a warzone than a place for healing.
This is a huge problem.
The Joint Commission specifies that admitted patients should spend no more than 4 hours in the ED waiting for a bed in the hospital.
That is, based on what I’ve seen, hugely ambitious. But I should point out that I work in a hospital that runs near capacity all the time, and studies – from some of my Yale colleagues, actually – have shown that once hospital capacity exceeds 85%, boarding rates skyrocket.
I want to discuss some of the causes of extended boarding and some solutions. But before that, I should prove to you that this really matters, and for that we are going to dig in to a new study which suggests that ED boarding kills.
To put some hard numbers to the boarding problem, we turn to this paper out of France, appearing in JAMA Internal Medicine.
This is a unique study design. Basically, on a single day – Dec. 12, 2022 – researchers fanned out across France to 97 EDs and started counting patients. The study focused on those older than age 75 who were admitted to a hospital ward from the ED. The researchers then defined two groups: those who were sent up to the hospital floor before midnight, and those who spent at least from midnight until 8 AM in the ED (basically, people forced to sleep in the ED for a night). The middle-ground people who were sent up between midnight and 8 AM were excluded.
The baseline characteristics between the two groups of patients were pretty similar: median age around 86, 55% female. There were no significant differences in comorbidities. That said, comporting with previous studies, people in an urban ED, an academic ED, or a busy ED were much more likely to board overnight.
So, what we have are two similar groups of patients treated quite differently. Not quite a randomized trial, given the hospital differences, but not bad for purposes of analysis.
Here are the most important numbers from the trial:
This difference held up even after adjustment for patient and hospital characteristics. Put another way, you’d need to send 22 patients to the floor instead of boarding in the ED to save one life. Not a bad return on investment.
It’s not entirely clear what the mechanism for the excess mortality might be, but the researchers note that patients kept in the ED overnight were about twice as likely to have a fall during their hospital stay – not surprising, given the dangers of gurneys in hallways and the sleep deprivation that trying to rest in a busy ED engenders.
I should point out that this could be worse in the United States. French ED doctors continue to care for admitted patients boarding in the ED, whereas in many hospitals in the United States, admitted patients are the responsibility of the floor team, regardless of where they are, making it more likely that these individuals may be neglected.
So, if boarding in the ED is a life-threatening situation, why do we do it? What conditions predispose to this?
You’ll hear a lot of talk, mostly from hospital administrators, saying that this is simply a problem of supply and demand. There are not enough beds for the number of patients who need beds. And staffing shortages don’t help either.
However, they never want to talk about the reasons for the staffing shortages, like poor pay, poor support, and, of course, the moral injury of treating patients in hallways.
The issue of volume is real. We could do a lot to prevent ED visits and hospital admissions by providing better access to preventive and primary care and improving our outpatient mental health infrastructure. But I think this framing passes the buck a little.
Another reason ED boarding occurs is the way our health care system is paid for. If you are building a hospital, you have little incentive to build in excess capacity. The most efficient hospital, from a profit-and-loss standpoint, is one that is 100% full as often as possible. That may be fine at times, but throw in a respiratory virus or even a pandemic, and those systems fracture under the pressure.
Let us also remember that not all hospital beds are given to patients who acutely need hospital beds. Many beds, in many hospitals, are necessary to handle postoperative patients undergoing elective procedures. Those patients having a knee replacement or abdominoplasty don’t spend the night in the ED when they leave the OR; they go to a hospital bed. And those procedures are – let’s face it – more profitable than an ED admission for a medical issue. That’s why, even when hospitals expand the number of beds they have, they do it with an eye toward increasing the rate of those profitable procedures, not decreasing the burden faced by their ED.
For now, the band-aid to the solution might be to better triage individuals boarding in the ED for floor access, prioritizing those of older age, greater frailty, or more medical complexity. But it feels like a stop-gap measure as long as the incentives are aligned to view an empty hospital bed as a sign of failure in the health system instead of success.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
As a consulting nephrologist, I go all over the hospital. Medicine floors, surgical floors, the ICU – I’ve even done consults in the operating room. And more and more, I do consults in the emergency department.
The reason I am doing more consults in the ED is not because the ED docs are getting gun shy with creatinine increases; it’s because patients are staying for extended periods in the ED despite being formally admitted to the hospital. It’s a phenomenon known as boarding, because there are simply not enough beds. You know the scene if you have ever been to a busy hospital: The ED is full to breaking, with patients on stretchers in hallways. It can often feel more like a warzone than a place for healing.
This is a huge problem.
The Joint Commission specifies that admitted patients should spend no more than 4 hours in the ED waiting for a bed in the hospital.
That is, based on what I’ve seen, hugely ambitious. But I should point out that I work in a hospital that runs near capacity all the time, and studies – from some of my Yale colleagues, actually – have shown that once hospital capacity exceeds 85%, boarding rates skyrocket.
I want to discuss some of the causes of extended boarding and some solutions. But before that, I should prove to you that this really matters, and for that we are going to dig in to a new study which suggests that ED boarding kills.
To put some hard numbers to the boarding problem, we turn to this paper out of France, appearing in JAMA Internal Medicine.
This is a unique study design. Basically, on a single day – Dec. 12, 2022 – researchers fanned out across France to 97 EDs and started counting patients. The study focused on those older than age 75 who were admitted to a hospital ward from the ED. The researchers then defined two groups: those who were sent up to the hospital floor before midnight, and those who spent at least from midnight until 8 AM in the ED (basically, people forced to sleep in the ED for a night). The middle-ground people who were sent up between midnight and 8 AM were excluded.
The baseline characteristics between the two groups of patients were pretty similar: median age around 86, 55% female. There were no significant differences in comorbidities. That said, comporting with previous studies, people in an urban ED, an academic ED, or a busy ED were much more likely to board overnight.
So, what we have are two similar groups of patients treated quite differently. Not quite a randomized trial, given the hospital differences, but not bad for purposes of analysis.
Here are the most important numbers from the trial:
This difference held up even after adjustment for patient and hospital characteristics. Put another way, you’d need to send 22 patients to the floor instead of boarding in the ED to save one life. Not a bad return on investment.
It’s not entirely clear what the mechanism for the excess mortality might be, but the researchers note that patients kept in the ED overnight were about twice as likely to have a fall during their hospital stay – not surprising, given the dangers of gurneys in hallways and the sleep deprivation that trying to rest in a busy ED engenders.
I should point out that this could be worse in the United States. French ED doctors continue to care for admitted patients boarding in the ED, whereas in many hospitals in the United States, admitted patients are the responsibility of the floor team, regardless of where they are, making it more likely that these individuals may be neglected.
So, if boarding in the ED is a life-threatening situation, why do we do it? What conditions predispose to this?
You’ll hear a lot of talk, mostly from hospital administrators, saying that this is simply a problem of supply and demand. There are not enough beds for the number of patients who need beds. And staffing shortages don’t help either.
However, they never want to talk about the reasons for the staffing shortages, like poor pay, poor support, and, of course, the moral injury of treating patients in hallways.
The issue of volume is real. We could do a lot to prevent ED visits and hospital admissions by providing better access to preventive and primary care and improving our outpatient mental health infrastructure. But I think this framing passes the buck a little.
Another reason ED boarding occurs is the way our health care system is paid for. If you are building a hospital, you have little incentive to build in excess capacity. The most efficient hospital, from a profit-and-loss standpoint, is one that is 100% full as often as possible. That may be fine at times, but throw in a respiratory virus or even a pandemic, and those systems fracture under the pressure.
Let us also remember that not all hospital beds are given to patients who acutely need hospital beds. Many beds, in many hospitals, are necessary to handle postoperative patients undergoing elective procedures. Those patients having a knee replacement or abdominoplasty don’t spend the night in the ED when they leave the OR; they go to a hospital bed. And those procedures are – let’s face it – more profitable than an ED admission for a medical issue. That’s why, even when hospitals expand the number of beds they have, they do it with an eye toward increasing the rate of those profitable procedures, not decreasing the burden faced by their ED.
For now, the band-aid to the solution might be to better triage individuals boarding in the ED for floor access, prioritizing those of older age, greater frailty, or more medical complexity. But it feels like a stop-gap measure as long as the incentives are aligned to view an empty hospital bed as a sign of failure in the health system instead of success.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
As a consulting nephrologist, I go all over the hospital. Medicine floors, surgical floors, the ICU – I’ve even done consults in the operating room. And more and more, I do consults in the emergency department.
The reason I am doing more consults in the ED is not because the ED docs are getting gun shy with creatinine increases; it’s because patients are staying for extended periods in the ED despite being formally admitted to the hospital. It’s a phenomenon known as boarding, because there are simply not enough beds. You know the scene if you have ever been to a busy hospital: The ED is full to breaking, with patients on stretchers in hallways. It can often feel more like a warzone than a place for healing.
This is a huge problem.
The Joint Commission specifies that admitted patients should spend no more than 4 hours in the ED waiting for a bed in the hospital.
That is, based on what I’ve seen, hugely ambitious. But I should point out that I work in a hospital that runs near capacity all the time, and studies – from some of my Yale colleagues, actually – have shown that once hospital capacity exceeds 85%, boarding rates skyrocket.
I want to discuss some of the causes of extended boarding and some solutions. But before that, I should prove to you that this really matters, and for that we are going to dig in to a new study which suggests that ED boarding kills.
To put some hard numbers to the boarding problem, we turn to this paper out of France, appearing in JAMA Internal Medicine.
This is a unique study design. Basically, on a single day – Dec. 12, 2022 – researchers fanned out across France to 97 EDs and started counting patients. The study focused on those older than age 75 who were admitted to a hospital ward from the ED. The researchers then defined two groups: those who were sent up to the hospital floor before midnight, and those who spent at least from midnight until 8 AM in the ED (basically, people forced to sleep in the ED for a night). The middle-ground people who were sent up between midnight and 8 AM were excluded.
The baseline characteristics between the two groups of patients were pretty similar: median age around 86, 55% female. There were no significant differences in comorbidities. That said, comporting with previous studies, people in an urban ED, an academic ED, or a busy ED were much more likely to board overnight.
So, what we have are two similar groups of patients treated quite differently. Not quite a randomized trial, given the hospital differences, but not bad for purposes of analysis.
Here are the most important numbers from the trial:
This difference held up even after adjustment for patient and hospital characteristics. Put another way, you’d need to send 22 patients to the floor instead of boarding in the ED to save one life. Not a bad return on investment.
It’s not entirely clear what the mechanism for the excess mortality might be, but the researchers note that patients kept in the ED overnight were about twice as likely to have a fall during their hospital stay – not surprising, given the dangers of gurneys in hallways and the sleep deprivation that trying to rest in a busy ED engenders.
I should point out that this could be worse in the United States. French ED doctors continue to care for admitted patients boarding in the ED, whereas in many hospitals in the United States, admitted patients are the responsibility of the floor team, regardless of where they are, making it more likely that these individuals may be neglected.
So, if boarding in the ED is a life-threatening situation, why do we do it? What conditions predispose to this?
You’ll hear a lot of talk, mostly from hospital administrators, saying that this is simply a problem of supply and demand. There are not enough beds for the number of patients who need beds. And staffing shortages don’t help either.
However, they never want to talk about the reasons for the staffing shortages, like poor pay, poor support, and, of course, the moral injury of treating patients in hallways.
The issue of volume is real. We could do a lot to prevent ED visits and hospital admissions by providing better access to preventive and primary care and improving our outpatient mental health infrastructure. But I think this framing passes the buck a little.
Another reason ED boarding occurs is the way our health care system is paid for. If you are building a hospital, you have little incentive to build in excess capacity. The most efficient hospital, from a profit-and-loss standpoint, is one that is 100% full as often as possible. That may be fine at times, but throw in a respiratory virus or even a pandemic, and those systems fracture under the pressure.
Let us also remember that not all hospital beds are given to patients who acutely need hospital beds. Many beds, in many hospitals, are necessary to handle postoperative patients undergoing elective procedures. Those patients having a knee replacement or abdominoplasty don’t spend the night in the ED when they leave the OR; they go to a hospital bed. And those procedures are – let’s face it – more profitable than an ED admission for a medical issue. That’s why, even when hospitals expand the number of beds they have, they do it with an eye toward increasing the rate of those profitable procedures, not decreasing the burden faced by their ED.
For now, the band-aid to the solution might be to better triage individuals boarding in the ED for floor access, prioritizing those of older age, greater frailty, or more medical complexity. But it feels like a stop-gap measure as long as the incentives are aligned to view an empty hospital bed as a sign of failure in the health system instead of success.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.


