User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
Most COVID long-haulers suffer long-term debilitating neurologic symptoms
Most COVID-19 long-haulers continue to have brain fog, fatigue, and compromised quality of life more than a year after the initial infection, results from the most extensive follow-up to date of a group of long COVID patients show.
Most patients continue to experience debilitating neurologic symptoms an average of 15 months from symptom onset, Igor Koralnik, MD, who oversees the Neuro COVID-19 Clinic at Northwestern Medicine in Chicago, said during a press briefing.
Surprisingly, in some cases, new symptoms appear that didn’t exist before, including variation of heart rate and blood pressure, and gastrointestinal symptoms, indicating there may be a late appearance in dysfunction of the autonomic nervous system in those patients, Dr. Koralnik said.
The study was published online in Annals of Clinical and Translational Neurology.
Evolving symptoms
The investigators evaluated the evolution of neurologic symptoms in 52 adults who had mild COVID-19 symptoms and were not admitted to the hospital.
Their mean age was 43 years, 73% were women and 77% had received a COVID-19 vaccine. These patients have now been followed for between 11 and 18 months since their initial infection.
Overall, between first and follow-up evaluations, there was no significant change in the frequency of most neurologic symptoms, including brain fog (81% vs. 71%), numbness/tingling (69% vs. 65%), headache (67% vs. 54%), dizziness (50% vs. 54%), blurred vision (34% vs. 44%), tinnitus (33% vs. 42%), and fatigue (87% vs. 81%).
The only neurologic symptoms that decreased over time were loss of taste (63% vs. 27%) and smell (58% vs. 21%).
Conversely, heart rate and blood pressure variation (35% vs. 56%) and gastrointestinal symptoms (27% vs. 48%; P = .04) increased at follow-up evaluations.
Patients reported subjective improvements in their recovery, cognitive function and fatigue, but quality of life measures remained lower than the average population of the United States.
There was a neutral effect of COVID vaccination on long COVID symptoms – it didn’t cure long COVID or make long COVID worse, which is a reason given by some long-haulers for not getting vaccinated, Dr. Koralnik told the briefing.
Therefore, “we continue to encourage our patients to get vaccinated and boosted according to the Centers for Disease Control and Prevention recommendation,” he said.
Escape from the ‘pit of despair’
To date, the Northwestern Medicine Neuro COVID-19 Clinic has treated nearly 1,400 COVID long-haulers from across the United States.
Emily Caffee, a physical therapist from Wheaton, Ill., is one of them.
Speaking at the briefing, the 36-year-old described her saga and roller coaster of recovering from long COVID in three acts: her initial infection, followed by a descent into a pit of physical and emotional despair, followed by her eventual escape from that pit more than two years later.
Following a fairly mild case of COVID, Ms. Caffee said worsening neurologic symptoms forced her to take medical leave from her very physical and cognitively demanding job.
Ms. Caffee said she experienced crushing fatigue and brain fog, as well as rapid heart rate and blood pressure changes going from sitting to standing position.
She went from being a competitive athlete to someone who could barely get off the couch or empty the dishwasher.
With the ongoing help of her medical team, she slowly returned to daily activities and eventually to work on a limited basis.
Today, Ms. Caffee says she’s 90%-95% better but still she has some lingering symptoms and does not yet feel like her pre-COVID self.
It’s been a very slow climb out of the pit, Ms. Caffee said.
This study has no specific funding. The authors disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Most COVID-19 long-haulers continue to have brain fog, fatigue, and compromised quality of life more than a year after the initial infection, results from the most extensive follow-up to date of a group of long COVID patients show.
Most patients continue to experience debilitating neurologic symptoms an average of 15 months from symptom onset, Igor Koralnik, MD, who oversees the Neuro COVID-19 Clinic at Northwestern Medicine in Chicago, said during a press briefing.
Surprisingly, in some cases, new symptoms appear that didn’t exist before, including variation of heart rate and blood pressure, and gastrointestinal symptoms, indicating there may be a late appearance in dysfunction of the autonomic nervous system in those patients, Dr. Koralnik said.
The study was published online in Annals of Clinical and Translational Neurology.
Evolving symptoms
The investigators evaluated the evolution of neurologic symptoms in 52 adults who had mild COVID-19 symptoms and were not admitted to the hospital.
Their mean age was 43 years, 73% were women and 77% had received a COVID-19 vaccine. These patients have now been followed for between 11 and 18 months since their initial infection.
Overall, between first and follow-up evaluations, there was no significant change in the frequency of most neurologic symptoms, including brain fog (81% vs. 71%), numbness/tingling (69% vs. 65%), headache (67% vs. 54%), dizziness (50% vs. 54%), blurred vision (34% vs. 44%), tinnitus (33% vs. 42%), and fatigue (87% vs. 81%).
The only neurologic symptoms that decreased over time were loss of taste (63% vs. 27%) and smell (58% vs. 21%).
Conversely, heart rate and blood pressure variation (35% vs. 56%) and gastrointestinal symptoms (27% vs. 48%; P = .04) increased at follow-up evaluations.
Patients reported subjective improvements in their recovery, cognitive function and fatigue, but quality of life measures remained lower than the average population of the United States.
There was a neutral effect of COVID vaccination on long COVID symptoms – it didn’t cure long COVID or make long COVID worse, which is a reason given by some long-haulers for not getting vaccinated, Dr. Koralnik told the briefing.
Therefore, “we continue to encourage our patients to get vaccinated and boosted according to the Centers for Disease Control and Prevention recommendation,” he said.
Escape from the ‘pit of despair’
To date, the Northwestern Medicine Neuro COVID-19 Clinic has treated nearly 1,400 COVID long-haulers from across the United States.
Emily Caffee, a physical therapist from Wheaton, Ill., is one of them.
Speaking at the briefing, the 36-year-old described her saga and roller coaster of recovering from long COVID in three acts: her initial infection, followed by a descent into a pit of physical and emotional despair, followed by her eventual escape from that pit more than two years later.
Following a fairly mild case of COVID, Ms. Caffee said worsening neurologic symptoms forced her to take medical leave from her very physical and cognitively demanding job.
Ms. Caffee said she experienced crushing fatigue and brain fog, as well as rapid heart rate and blood pressure changes going from sitting to standing position.
She went from being a competitive athlete to someone who could barely get off the couch or empty the dishwasher.
With the ongoing help of her medical team, she slowly returned to daily activities and eventually to work on a limited basis.
Today, Ms. Caffee says she’s 90%-95% better but still she has some lingering symptoms and does not yet feel like her pre-COVID self.
It’s been a very slow climb out of the pit, Ms. Caffee said.
This study has no specific funding. The authors disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Most COVID-19 long-haulers continue to have brain fog, fatigue, and compromised quality of life more than a year after the initial infection, results from the most extensive follow-up to date of a group of long COVID patients show.
Most patients continue to experience debilitating neurologic symptoms an average of 15 months from symptom onset, Igor Koralnik, MD, who oversees the Neuro COVID-19 Clinic at Northwestern Medicine in Chicago, said during a press briefing.
Surprisingly, in some cases, new symptoms appear that didn’t exist before, including variation of heart rate and blood pressure, and gastrointestinal symptoms, indicating there may be a late appearance in dysfunction of the autonomic nervous system in those patients, Dr. Koralnik said.
The study was published online in Annals of Clinical and Translational Neurology.
Evolving symptoms
The investigators evaluated the evolution of neurologic symptoms in 52 adults who had mild COVID-19 symptoms and were not admitted to the hospital.
Their mean age was 43 years, 73% were women and 77% had received a COVID-19 vaccine. These patients have now been followed for between 11 and 18 months since their initial infection.
Overall, between first and follow-up evaluations, there was no significant change in the frequency of most neurologic symptoms, including brain fog (81% vs. 71%), numbness/tingling (69% vs. 65%), headache (67% vs. 54%), dizziness (50% vs. 54%), blurred vision (34% vs. 44%), tinnitus (33% vs. 42%), and fatigue (87% vs. 81%).
The only neurologic symptoms that decreased over time were loss of taste (63% vs. 27%) and smell (58% vs. 21%).
Conversely, heart rate and blood pressure variation (35% vs. 56%) and gastrointestinal symptoms (27% vs. 48%; P = .04) increased at follow-up evaluations.
Patients reported subjective improvements in their recovery, cognitive function and fatigue, but quality of life measures remained lower than the average population of the United States.
There was a neutral effect of COVID vaccination on long COVID symptoms – it didn’t cure long COVID or make long COVID worse, which is a reason given by some long-haulers for not getting vaccinated, Dr. Koralnik told the briefing.
Therefore, “we continue to encourage our patients to get vaccinated and boosted according to the Centers for Disease Control and Prevention recommendation,” he said.
Escape from the ‘pit of despair’
To date, the Northwestern Medicine Neuro COVID-19 Clinic has treated nearly 1,400 COVID long-haulers from across the United States.
Emily Caffee, a physical therapist from Wheaton, Ill., is one of them.
Speaking at the briefing, the 36-year-old described her saga and roller coaster of recovering from long COVID in three acts: her initial infection, followed by a descent into a pit of physical and emotional despair, followed by her eventual escape from that pit more than two years later.
Following a fairly mild case of COVID, Ms. Caffee said worsening neurologic symptoms forced her to take medical leave from her very physical and cognitively demanding job.
Ms. Caffee said she experienced crushing fatigue and brain fog, as well as rapid heart rate and blood pressure changes going from sitting to standing position.
She went from being a competitive athlete to someone who could barely get off the couch or empty the dishwasher.
With the ongoing help of her medical team, she slowly returned to daily activities and eventually to work on a limited basis.
Today, Ms. Caffee says she’s 90%-95% better but still she has some lingering symptoms and does not yet feel like her pre-COVID self.
It’s been a very slow climb out of the pit, Ms. Caffee said.
This study has no specific funding. The authors disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF CLINICAL AND TRANSLATIONAL NEUROLOGY
Legislative efforts continue to revamp laws governing PAs
INDIANAPOLIS – That’s according to Phil Bongiorno, BA, senior vice president of advocacy and government relations at the American Academy of Physician Associates (AAPA), who spoke at the group’s annual meeting.
OTP refers to the AAPA’s goal of improving patient access to care and lessening administrative obligations by eliminating the legal requirement that there be a specific relationship between a PA, physician, or any other health care provider. This would allow a PA to practice to the full extent of their education, training, and experience, Mr. Bongiorno said.
The second tenet of OTP is to persuade states to create a separate majority PA board to regulate PAs. An alternative to this would be for states to add PAs and physicians who work with PAs to their medical or healing arts boards, he said.
Third, in an OTP environment, each state would authorize PAs to be eligible for direct payment by all public and private insurers. “We have seen that development at the federal level, as far as Medicare is concerned,” Mr. Bongiorno said. “Now, we’re focusing on making that happen in the individual states as well.”
According to Mr. Bongiorno, this year’s state advocacy priorities are to pursue new legislation in additional states, even as efforts continue to persuade state legislatures to act on carryover bills from the previous legislative session.
Mr. Bongiorno briefly summarized what he called “OTP successes” from 2021:
- Federal government: Authorized direct payment to PAs under Medicare
- Arkansas, Delaware, Illinois, Pennsylvania: Added one or more PAs to their medical boards
- Florida, Utah: Approved direct payment to PAs
- Tennessee, Wisconsin: Created a separate PA review board
- Utah, Wisconsin: Removed the relationship/agreement requirement (Wisconsin now requires 10,000 hours of practice to remove the relationship requirement)
North Central region
In Colorado, House Bill 1095 (HB1095) would have removed requirements for a legal relationship between a PA and a physician. Initially that would have happened after 3,000 hours of practice, although changing that to 5,000 hours has been a compromise measure. PAs changing specialties must collaborate for 2,000 hours, now negotiated to 3,000 hours.
HB1095 ultimately was not successful last year or this year, said Erika Miller, director of state advocacy and outreach for the AAPA. “But we do see it as a success, because in the 2022 session, we managed to get it passed in committee by a 10-to-1 vote,” she said. “It then moved to the full house and was not successful there.”
Ms. Miller said that South Dakota Senate Bill 134 would have removed the requirement for a legal PA/physician relationship after 1,040 hours, which is the requirement for nurse practitioners. “South Dakota had introduced similar legislation the year before, but also like Colorado, they went from not getting out of committee last year to making it to the senate floor this time,” she said.
In Wisconsin, the new PA-affiliated credentialing board began on April 1. It gives PAs the authority to license, discipline, and write regulations, Ms. Miller said.
South Central region
Arizona Senate Bill 1367 included direct pay, removed the relationship tether with a physician, and made each PA fully responsible for the care they provide. “The bill passed out of committee successfully but did not make it to a vote due to unexpected struggles between the Arizona medical society and PA chapter,” said Shannon Morey, senior director of state advocacy and outreach at the AAPA. “They are ready to go again next year.”
In Louisiana, Senate Bill 158 is a “strong” bill that addressed all the desired aspects of OTP, Ms. Morey said; “The legislation stands subject to call on the Senate floor, but it has been killed by the sponsor.”
Northeast region
Massachusetts Senate Bill 740 (S740) would remove the legal tether between PA and physician, said Carson Walker, senior director of state advocacy and outreach at the AAPA. “The committee decided to extend its time in committee until June,” he said. “By next month, we expect that the committee will schedule a hearing that includes S740, and we fully plan on submitting testimony.”
In New York, Senate Bill 9233 (S9233) would remove physician supervision after 3,600 hours of practice.
“Just about 10 days ago, sponsors were able to have S9233 introduced, which is the most succinct and, I think, the most effective OTP bill I have ever seen,” Mr. Walker said.
“S9233 says that after 3,600 hours a PA can practice without the supervision of a physician, and that’s all. There’s not a lot of time left in this session, but we are hopeful that it lays the groundwork for success next year.”
New Hampshire Senate Bill 228 has passed the legislature and is awaiting the governor’s signature. It will allow direct payment, make PAs responsible for the care they provide, and shift the physician-PA relationship from supervision to collaboration, Mr. Walker said.
Southeast region
Stephanie Radix, senior director of state advocacy and outreach at the AAPA, discussed North Carolina’s Senate Bill 345, which passed the Senate unanimously in 2021 and has been carried over to this year’s session. The bill defines team-based settings, eliminates the relationship tether, and establishes a supervised career entry interval of 4,000 clinical hours in the state.
The legislature is slated to adjourn June 30, Ms. Radix said: “We are very hopeful that we will get it across the finish line.”
In an interview, Mr. Bongiorno said that the AAPA’s overall advocacy progress is as expected.
“Optimal team practice is about allowing each practice to make that determination on how the team should work as a true collaboration,” he said. “The bottom line is that OTP would allow us to reach more patients, serve the community, and ensure that people are able to get healthcare, especially in underserved areas.”
A version of this article first appeared on Medscape.com.
INDIANAPOLIS – That’s according to Phil Bongiorno, BA, senior vice president of advocacy and government relations at the American Academy of Physician Associates (AAPA), who spoke at the group’s annual meeting.
OTP refers to the AAPA’s goal of improving patient access to care and lessening administrative obligations by eliminating the legal requirement that there be a specific relationship between a PA, physician, or any other health care provider. This would allow a PA to practice to the full extent of their education, training, and experience, Mr. Bongiorno said.
The second tenet of OTP is to persuade states to create a separate majority PA board to regulate PAs. An alternative to this would be for states to add PAs and physicians who work with PAs to their medical or healing arts boards, he said.
Third, in an OTP environment, each state would authorize PAs to be eligible for direct payment by all public and private insurers. “We have seen that development at the federal level, as far as Medicare is concerned,” Mr. Bongiorno said. “Now, we’re focusing on making that happen in the individual states as well.”
According to Mr. Bongiorno, this year’s state advocacy priorities are to pursue new legislation in additional states, even as efforts continue to persuade state legislatures to act on carryover bills from the previous legislative session.
Mr. Bongiorno briefly summarized what he called “OTP successes” from 2021:
- Federal government: Authorized direct payment to PAs under Medicare
- Arkansas, Delaware, Illinois, Pennsylvania: Added one or more PAs to their medical boards
- Florida, Utah: Approved direct payment to PAs
- Tennessee, Wisconsin: Created a separate PA review board
- Utah, Wisconsin: Removed the relationship/agreement requirement (Wisconsin now requires 10,000 hours of practice to remove the relationship requirement)
North Central region
In Colorado, House Bill 1095 (HB1095) would have removed requirements for a legal relationship between a PA and a physician. Initially that would have happened after 3,000 hours of practice, although changing that to 5,000 hours has been a compromise measure. PAs changing specialties must collaborate for 2,000 hours, now negotiated to 3,000 hours.
HB1095 ultimately was not successful last year or this year, said Erika Miller, director of state advocacy and outreach for the AAPA. “But we do see it as a success, because in the 2022 session, we managed to get it passed in committee by a 10-to-1 vote,” she said. “It then moved to the full house and was not successful there.”
Ms. Miller said that South Dakota Senate Bill 134 would have removed the requirement for a legal PA/physician relationship after 1,040 hours, which is the requirement for nurse practitioners. “South Dakota had introduced similar legislation the year before, but also like Colorado, they went from not getting out of committee last year to making it to the senate floor this time,” she said.
In Wisconsin, the new PA-affiliated credentialing board began on April 1. It gives PAs the authority to license, discipline, and write regulations, Ms. Miller said.
South Central region
Arizona Senate Bill 1367 included direct pay, removed the relationship tether with a physician, and made each PA fully responsible for the care they provide. “The bill passed out of committee successfully but did not make it to a vote due to unexpected struggles between the Arizona medical society and PA chapter,” said Shannon Morey, senior director of state advocacy and outreach at the AAPA. “They are ready to go again next year.”
In Louisiana, Senate Bill 158 is a “strong” bill that addressed all the desired aspects of OTP, Ms. Morey said; “The legislation stands subject to call on the Senate floor, but it has been killed by the sponsor.”
Northeast region
Massachusetts Senate Bill 740 (S740) would remove the legal tether between PA and physician, said Carson Walker, senior director of state advocacy and outreach at the AAPA. “The committee decided to extend its time in committee until June,” he said. “By next month, we expect that the committee will schedule a hearing that includes S740, and we fully plan on submitting testimony.”
In New York, Senate Bill 9233 (S9233) would remove physician supervision after 3,600 hours of practice.
“Just about 10 days ago, sponsors were able to have S9233 introduced, which is the most succinct and, I think, the most effective OTP bill I have ever seen,” Mr. Walker said.
“S9233 says that after 3,600 hours a PA can practice without the supervision of a physician, and that’s all. There’s not a lot of time left in this session, but we are hopeful that it lays the groundwork for success next year.”
New Hampshire Senate Bill 228 has passed the legislature and is awaiting the governor’s signature. It will allow direct payment, make PAs responsible for the care they provide, and shift the physician-PA relationship from supervision to collaboration, Mr. Walker said.
Southeast region
Stephanie Radix, senior director of state advocacy and outreach at the AAPA, discussed North Carolina’s Senate Bill 345, which passed the Senate unanimously in 2021 and has been carried over to this year’s session. The bill defines team-based settings, eliminates the relationship tether, and establishes a supervised career entry interval of 4,000 clinical hours in the state.
The legislature is slated to adjourn June 30, Ms. Radix said: “We are very hopeful that we will get it across the finish line.”
In an interview, Mr. Bongiorno said that the AAPA’s overall advocacy progress is as expected.
“Optimal team practice is about allowing each practice to make that determination on how the team should work as a true collaboration,” he said. “The bottom line is that OTP would allow us to reach more patients, serve the community, and ensure that people are able to get healthcare, especially in underserved areas.”
A version of this article first appeared on Medscape.com.
INDIANAPOLIS – That’s according to Phil Bongiorno, BA, senior vice president of advocacy and government relations at the American Academy of Physician Associates (AAPA), who spoke at the group’s annual meeting.
OTP refers to the AAPA’s goal of improving patient access to care and lessening administrative obligations by eliminating the legal requirement that there be a specific relationship between a PA, physician, or any other health care provider. This would allow a PA to practice to the full extent of their education, training, and experience, Mr. Bongiorno said.
The second tenet of OTP is to persuade states to create a separate majority PA board to regulate PAs. An alternative to this would be for states to add PAs and physicians who work with PAs to their medical or healing arts boards, he said.
Third, in an OTP environment, each state would authorize PAs to be eligible for direct payment by all public and private insurers. “We have seen that development at the federal level, as far as Medicare is concerned,” Mr. Bongiorno said. “Now, we’re focusing on making that happen in the individual states as well.”
According to Mr. Bongiorno, this year’s state advocacy priorities are to pursue new legislation in additional states, even as efforts continue to persuade state legislatures to act on carryover bills from the previous legislative session.
Mr. Bongiorno briefly summarized what he called “OTP successes” from 2021:
- Federal government: Authorized direct payment to PAs under Medicare
- Arkansas, Delaware, Illinois, Pennsylvania: Added one or more PAs to their medical boards
- Florida, Utah: Approved direct payment to PAs
- Tennessee, Wisconsin: Created a separate PA review board
- Utah, Wisconsin: Removed the relationship/agreement requirement (Wisconsin now requires 10,000 hours of practice to remove the relationship requirement)
North Central region
In Colorado, House Bill 1095 (HB1095) would have removed requirements for a legal relationship between a PA and a physician. Initially that would have happened after 3,000 hours of practice, although changing that to 5,000 hours has been a compromise measure. PAs changing specialties must collaborate for 2,000 hours, now negotiated to 3,000 hours.
HB1095 ultimately was not successful last year or this year, said Erika Miller, director of state advocacy and outreach for the AAPA. “But we do see it as a success, because in the 2022 session, we managed to get it passed in committee by a 10-to-1 vote,” she said. “It then moved to the full house and was not successful there.”
Ms. Miller said that South Dakota Senate Bill 134 would have removed the requirement for a legal PA/physician relationship after 1,040 hours, which is the requirement for nurse practitioners. “South Dakota had introduced similar legislation the year before, but also like Colorado, they went from not getting out of committee last year to making it to the senate floor this time,” she said.
In Wisconsin, the new PA-affiliated credentialing board began on April 1. It gives PAs the authority to license, discipline, and write regulations, Ms. Miller said.
South Central region
Arizona Senate Bill 1367 included direct pay, removed the relationship tether with a physician, and made each PA fully responsible for the care they provide. “The bill passed out of committee successfully but did not make it to a vote due to unexpected struggles between the Arizona medical society and PA chapter,” said Shannon Morey, senior director of state advocacy and outreach at the AAPA. “They are ready to go again next year.”
In Louisiana, Senate Bill 158 is a “strong” bill that addressed all the desired aspects of OTP, Ms. Morey said; “The legislation stands subject to call on the Senate floor, but it has been killed by the sponsor.”
Northeast region
Massachusetts Senate Bill 740 (S740) would remove the legal tether between PA and physician, said Carson Walker, senior director of state advocacy and outreach at the AAPA. “The committee decided to extend its time in committee until June,” he said. “By next month, we expect that the committee will schedule a hearing that includes S740, and we fully plan on submitting testimony.”
In New York, Senate Bill 9233 (S9233) would remove physician supervision after 3,600 hours of practice.
“Just about 10 days ago, sponsors were able to have S9233 introduced, which is the most succinct and, I think, the most effective OTP bill I have ever seen,” Mr. Walker said.
“S9233 says that after 3,600 hours a PA can practice without the supervision of a physician, and that’s all. There’s not a lot of time left in this session, but we are hopeful that it lays the groundwork for success next year.”
New Hampshire Senate Bill 228 has passed the legislature and is awaiting the governor’s signature. It will allow direct payment, make PAs responsible for the care they provide, and shift the physician-PA relationship from supervision to collaboration, Mr. Walker said.
Southeast region
Stephanie Radix, senior director of state advocacy and outreach at the AAPA, discussed North Carolina’s Senate Bill 345, which passed the Senate unanimously in 2021 and has been carried over to this year’s session. The bill defines team-based settings, eliminates the relationship tether, and establishes a supervised career entry interval of 4,000 clinical hours in the state.
The legislature is slated to adjourn June 30, Ms. Radix said: “We are very hopeful that we will get it across the finish line.”
In an interview, Mr. Bongiorno said that the AAPA’s overall advocacy progress is as expected.
“Optimal team practice is about allowing each practice to make that determination on how the team should work as a true collaboration,” he said. “The bottom line is that OTP would allow us to reach more patients, serve the community, and ensure that people are able to get healthcare, especially in underserved areas.”
A version of this article first appeared on Medscape.com.
AT AAPA 2022
Researchers find a pathway to prevent COVID infection
What’s more, they have succeeded in closing the lock to block the virus and prevent it from interacting with the cell, thereby preventing infection.
UCLouvain emphasized that this discovery, which was published in Nature Communications, is sparking hope that an aerosol antiviral therapy can be developed that would eradicate the virus in the case of an infection or a high-risk contact.
For 2 years, the team under David Alsteens, PhD, a researcher at the UCLouvain Institute of Biomolecular Science and Technology, has been working hard to understand the precise molecular mechanisms the virus uses to infect a cell. They investigated the interaction between sialic acids, a kind of sugar residue present on the surface of cells, and the SARS-CoV-2 spike (S) protein to clarify its role in the infection process.
It was already known that the function of the sugar residues that coat the cells is to promote cell recognition, thus enabling, in particular, viruses to identify their targets more easily, but also to provide them with a point of attachment and to facilitate infection of the cells.
The researchers have now revealed a variant of these sugars that interacts more strongly with the S protein than other sugars do.
In other words, the university explained, they found the set of keys that allows the virus to open the cell door. So, the researchers decided to catch the virus in its own trap, by preventing it from attaching to its host cell. To do this, they blocked the S protein’s points of attachment, thus suppressing any interaction with the cell surface, as if a padlock had been placed on the lock on the cell’s entry door.
Th researchers added that the advantage of this discovery is that it acts on the virus, irrespective of mutations.
The team of researchers will now conduct tests on mice to apply this blocking of virus binding sites and observe whether it works on the body. The results should make it possible to develop an antiviral therapy administered by aerosol in the case of infection or at-risk contact.
This discovery is also of interest for the future to counter other viruses with similar attachment factors.
This article was translated from MediQuality; a version appeared on Medscape.com.
What’s more, they have succeeded in closing the lock to block the virus and prevent it from interacting with the cell, thereby preventing infection.
UCLouvain emphasized that this discovery, which was published in Nature Communications, is sparking hope that an aerosol antiviral therapy can be developed that would eradicate the virus in the case of an infection or a high-risk contact.
For 2 years, the team under David Alsteens, PhD, a researcher at the UCLouvain Institute of Biomolecular Science and Technology, has been working hard to understand the precise molecular mechanisms the virus uses to infect a cell. They investigated the interaction between sialic acids, a kind of sugar residue present on the surface of cells, and the SARS-CoV-2 spike (S) protein to clarify its role in the infection process.
It was already known that the function of the sugar residues that coat the cells is to promote cell recognition, thus enabling, in particular, viruses to identify their targets more easily, but also to provide them with a point of attachment and to facilitate infection of the cells.
The researchers have now revealed a variant of these sugars that interacts more strongly with the S protein than other sugars do.
In other words, the university explained, they found the set of keys that allows the virus to open the cell door. So, the researchers decided to catch the virus in its own trap, by preventing it from attaching to its host cell. To do this, they blocked the S protein’s points of attachment, thus suppressing any interaction with the cell surface, as if a padlock had been placed on the lock on the cell’s entry door.
Th researchers added that the advantage of this discovery is that it acts on the virus, irrespective of mutations.
The team of researchers will now conduct tests on mice to apply this blocking of virus binding sites and observe whether it works on the body. The results should make it possible to develop an antiviral therapy administered by aerosol in the case of infection or at-risk contact.
This discovery is also of interest for the future to counter other viruses with similar attachment factors.
This article was translated from MediQuality; a version appeared on Medscape.com.
What’s more, they have succeeded in closing the lock to block the virus and prevent it from interacting with the cell, thereby preventing infection.
UCLouvain emphasized that this discovery, which was published in Nature Communications, is sparking hope that an aerosol antiviral therapy can be developed that would eradicate the virus in the case of an infection or a high-risk contact.
For 2 years, the team under David Alsteens, PhD, a researcher at the UCLouvain Institute of Biomolecular Science and Technology, has been working hard to understand the precise molecular mechanisms the virus uses to infect a cell. They investigated the interaction between sialic acids, a kind of sugar residue present on the surface of cells, and the SARS-CoV-2 spike (S) protein to clarify its role in the infection process.
It was already known that the function of the sugar residues that coat the cells is to promote cell recognition, thus enabling, in particular, viruses to identify their targets more easily, but also to provide them with a point of attachment and to facilitate infection of the cells.
The researchers have now revealed a variant of these sugars that interacts more strongly with the S protein than other sugars do.
In other words, the university explained, they found the set of keys that allows the virus to open the cell door. So, the researchers decided to catch the virus in its own trap, by preventing it from attaching to its host cell. To do this, they blocked the S protein’s points of attachment, thus suppressing any interaction with the cell surface, as if a padlock had been placed on the lock on the cell’s entry door.
Th researchers added that the advantage of this discovery is that it acts on the virus, irrespective of mutations.
The team of researchers will now conduct tests on mice to apply this blocking of virus binding sites and observe whether it works on the body. The results should make it possible to develop an antiviral therapy administered by aerosol in the case of infection or at-risk contact.
This discovery is also of interest for the future to counter other viruses with similar attachment factors.
This article was translated from MediQuality; a version appeared on Medscape.com.
FROM NATURE COMMUNICATIONS
Crohn’s disease research goes to the dogs
Why it might be better to be a dog person
Here’s that old debate again: Dogs or cats? You probably have your own opinion, but research presented at this year’s Digestive Disease Week may have tipped the scale by showing that children who lived with dogs may be less likely to have Crohn’s disease as adults.
The research was done by having approximately 4,300 people closely related to patients with Crohn’s disease fill out an environmental questionnaire. Using these data, the research team looked into environmental factors such as size of the families, where the home was, how many bathrooms the homes had, and quality of drinking water.
The researchers found that those who had or were exposed to dogs between the ages of 5 and 15 years were more likely to have healthy gut permeability and balanced microbes, which increased their protection against Crohn’s disease.
“Our study seems to add to others that have explored the ‘hygiene hypothesis’ which suggests that the lack of exposure to microbes early in life may lead to lack of immune regulation toward environmental microbes,” senior author Williams Turpin, PhD, said in the written statement.
The researchers aren’t sure why they didn’t get the same findings with cats, but Dr. Turpin theorized that dog owners tend to be outside more with their dogs or live in places with more green space, which are good protectors against Crohn’s disease.
It’s all good for dog owners, but do their pets’ parasites make you more attractive? Just more fuel for the ongoing debate.
Come for the history, stay for the fossilized parasites
Another week, another analysis of old British poop. LOTME really is your one-stop shop for all the important, hard-hitting news about historic parasites. You’re welcome, Internet.
The news this week is from Stonehenge, which is apparently kind of a big deal. Rocks in a circle, celestial calendar, cultural significance, whatever. We’re not here to talk about rocks. We’re here to talk about, uh, rocks. Smaller rocks. Specifically, coprolites, which are essentially poop turned into a rock. (Though now we’re imagining Stonehenge made out of fossilized poop rocks. Would it still be a big tourist destination? We can see both sides of the argument on that one.)
Archaeologists from the University of Cambridge have conducted an analysis of coprolites from Durrington Walls, a Neolithic settlement just a few kilometers from Stonehenge. The town dates to the same time that Stonehenge was constructed, and it’s believed that the residents were responsible for building the landmark. These coprolites, depending on what’s inside, can tell us a lot about how the builders of Stonehenge lived and, more specifically, how they ate.
In this case, the coprolites of one human and three dogs contained capillariid worm eggs. These worms come from cows, and when a human is typically infected, the eggs embed in the liver and do not pass through the body. Finding them in excrement indicates that the people were eating raw cow organs and feeding leftovers to their dogs. This is interesting, because a preponderance of pottery and cooking implements also found at the site indicates that the residents of Durrington Walls were spit-roasting or boiling their beef and pork. So the meat was cooked, but not the organs. That is an interesting dietary decision, ancient British people. Then again, modern British cuisine exists. At least now we know where they got it from.
This new research raises one other very important question: When are we going to get a full-on guided tour of all the important coprolite sites in Britain? They’ve clearly got plenty of them, and the tourist demand for ancient parasites must be sky-high. Come on, capitalism, follow through on this. We’d go.
Everyone lies: Food intake edition
Do you have any patients on special diets? Do you ask them if they are following those diets? Don’t bother, because they’re lying. Everyone lies about the food they eat. Everyone. Obese people lie, and nonobese people lie.
Investigators at the University of Essex in England asked 221 adults to keep food diaries, and then they checked on energy consumption by analyzing radioactive water levels in the participants’ urine over a 10-day period.
Underreporting of food consumption was rampant, even among those who were not obese. The obese subjects did underreport by a greater extent (1,200 calories per day) than did those who were not obese, who were off by only 800 calories, but the obese participants burned about 400 calories more each day than did the nonobese, so the difference was a wash.
Everyone ended up underreporting their calorie consumption by an average of about 900 calories, and the investigators were good enough to provide some food equivalents, tops on the list being three MacDonald’s cheeseburgers.
“Public health recommendations have historically relied heavily on self-reported energy intake values,” senior author Gavin Sandercock, PhD, said in a EurekAlert statement, and “recognising that the measures of energy intake are incorrect might result in the setting of more realistic targets.”
Maybe you can be more realistic with your patients, too. Go ahead and ask Mr. Smith about the burger sticking out of his coat pocket, because there are probably two more you can’t see. We’ve each got 900 calories hiding on us somewhere. Ours is usually pizza.
The art of the gallbladder
Ever thought you would see a portrait of a gallbladder hanging up in a gallery? Not just an artist’s rendition, but an actual photo from an actual patient? Well, you can at the Soloway Gallery in Brooklyn, N.Y., at least until June 12.
The artist? K.C. Joseph, MD, a general surgeon from St. Marie, Pa., who died in 2015. His daughter Melissa is the curator of the show and told ARTnews about the interesting connection her father had with art and surgery.
In 2010, Dr. Joseph gave his daughter a box of photos and said “Make me a famous artist,” she recalled. At first, “I was like, ‘These are weird,’ and then I put them under my bed for 10 years.”
Apparently he had been making art with his patients’ organs for about 15 years and had a system in which he put each one together. Before a surgery Dr. Joseph would make a note card with the patient’s name handwritten in calligraphy with a couple of pages taken out of the magazine from the waiting room as the backdrop. Afterward, when the patient was in recovery, the removed organ would be placed among the pages and the name card. A photo was taken with the same endoscope that was used for the procedure.
After the show’s debut, people reached out expressing their love for their photos. “I wish, before he died, I had asked him more questions about it,” Ms. Joseph told ARTnews. “I’m regretting it so much now, kicking myself.”
Who gets to take home an artsy photo of their gallbladder after getting it removed? Not us, that’s who. Each collage is a one-of-a-kind piece. They definitely should be framed and shown in an art gallery. Oh, right. Never mind.
Why it might be better to be a dog person
Here’s that old debate again: Dogs or cats? You probably have your own opinion, but research presented at this year’s Digestive Disease Week may have tipped the scale by showing that children who lived with dogs may be less likely to have Crohn’s disease as adults.
The research was done by having approximately 4,300 people closely related to patients with Crohn’s disease fill out an environmental questionnaire. Using these data, the research team looked into environmental factors such as size of the families, where the home was, how many bathrooms the homes had, and quality of drinking water.
The researchers found that those who had or were exposed to dogs between the ages of 5 and 15 years were more likely to have healthy gut permeability and balanced microbes, which increased their protection against Crohn’s disease.
“Our study seems to add to others that have explored the ‘hygiene hypothesis’ which suggests that the lack of exposure to microbes early in life may lead to lack of immune regulation toward environmental microbes,” senior author Williams Turpin, PhD, said in the written statement.
The researchers aren’t sure why they didn’t get the same findings with cats, but Dr. Turpin theorized that dog owners tend to be outside more with their dogs or live in places with more green space, which are good protectors against Crohn’s disease.
It’s all good for dog owners, but do their pets’ parasites make you more attractive? Just more fuel for the ongoing debate.
Come for the history, stay for the fossilized parasites
Another week, another analysis of old British poop. LOTME really is your one-stop shop for all the important, hard-hitting news about historic parasites. You’re welcome, Internet.
The news this week is from Stonehenge, which is apparently kind of a big deal. Rocks in a circle, celestial calendar, cultural significance, whatever. We’re not here to talk about rocks. We’re here to talk about, uh, rocks. Smaller rocks. Specifically, coprolites, which are essentially poop turned into a rock. (Though now we’re imagining Stonehenge made out of fossilized poop rocks. Would it still be a big tourist destination? We can see both sides of the argument on that one.)
Archaeologists from the University of Cambridge have conducted an analysis of coprolites from Durrington Walls, a Neolithic settlement just a few kilometers from Stonehenge. The town dates to the same time that Stonehenge was constructed, and it’s believed that the residents were responsible for building the landmark. These coprolites, depending on what’s inside, can tell us a lot about how the builders of Stonehenge lived and, more specifically, how they ate.
In this case, the coprolites of one human and three dogs contained capillariid worm eggs. These worms come from cows, and when a human is typically infected, the eggs embed in the liver and do not pass through the body. Finding them in excrement indicates that the people were eating raw cow organs and feeding leftovers to their dogs. This is interesting, because a preponderance of pottery and cooking implements also found at the site indicates that the residents of Durrington Walls were spit-roasting or boiling their beef and pork. So the meat was cooked, but not the organs. That is an interesting dietary decision, ancient British people. Then again, modern British cuisine exists. At least now we know where they got it from.
This new research raises one other very important question: When are we going to get a full-on guided tour of all the important coprolite sites in Britain? They’ve clearly got plenty of them, and the tourist demand for ancient parasites must be sky-high. Come on, capitalism, follow through on this. We’d go.
Everyone lies: Food intake edition
Do you have any patients on special diets? Do you ask them if they are following those diets? Don’t bother, because they’re lying. Everyone lies about the food they eat. Everyone. Obese people lie, and nonobese people lie.
Investigators at the University of Essex in England asked 221 adults to keep food diaries, and then they checked on energy consumption by analyzing radioactive water levels in the participants’ urine over a 10-day period.
Underreporting of food consumption was rampant, even among those who were not obese. The obese subjects did underreport by a greater extent (1,200 calories per day) than did those who were not obese, who were off by only 800 calories, but the obese participants burned about 400 calories more each day than did the nonobese, so the difference was a wash.
Everyone ended up underreporting their calorie consumption by an average of about 900 calories, and the investigators were good enough to provide some food equivalents, tops on the list being three MacDonald’s cheeseburgers.
“Public health recommendations have historically relied heavily on self-reported energy intake values,” senior author Gavin Sandercock, PhD, said in a EurekAlert statement, and “recognising that the measures of energy intake are incorrect might result in the setting of more realistic targets.”
Maybe you can be more realistic with your patients, too. Go ahead and ask Mr. Smith about the burger sticking out of his coat pocket, because there are probably two more you can’t see. We’ve each got 900 calories hiding on us somewhere. Ours is usually pizza.
The art of the gallbladder
Ever thought you would see a portrait of a gallbladder hanging up in a gallery? Not just an artist’s rendition, but an actual photo from an actual patient? Well, you can at the Soloway Gallery in Brooklyn, N.Y., at least until June 12.
The artist? K.C. Joseph, MD, a general surgeon from St. Marie, Pa., who died in 2015. His daughter Melissa is the curator of the show and told ARTnews about the interesting connection her father had with art and surgery.
In 2010, Dr. Joseph gave his daughter a box of photos and said “Make me a famous artist,” she recalled. At first, “I was like, ‘These are weird,’ and then I put them under my bed for 10 years.”
Apparently he had been making art with his patients’ organs for about 15 years and had a system in which he put each one together. Before a surgery Dr. Joseph would make a note card with the patient’s name handwritten in calligraphy with a couple of pages taken out of the magazine from the waiting room as the backdrop. Afterward, when the patient was in recovery, the removed organ would be placed among the pages and the name card. A photo was taken with the same endoscope that was used for the procedure.
After the show’s debut, people reached out expressing their love for their photos. “I wish, before he died, I had asked him more questions about it,” Ms. Joseph told ARTnews. “I’m regretting it so much now, kicking myself.”
Who gets to take home an artsy photo of their gallbladder after getting it removed? Not us, that’s who. Each collage is a one-of-a-kind piece. They definitely should be framed and shown in an art gallery. Oh, right. Never mind.
Why it might be better to be a dog person
Here’s that old debate again: Dogs or cats? You probably have your own opinion, but research presented at this year’s Digestive Disease Week may have tipped the scale by showing that children who lived with dogs may be less likely to have Crohn’s disease as adults.
The research was done by having approximately 4,300 people closely related to patients with Crohn’s disease fill out an environmental questionnaire. Using these data, the research team looked into environmental factors such as size of the families, where the home was, how many bathrooms the homes had, and quality of drinking water.
The researchers found that those who had or were exposed to dogs between the ages of 5 and 15 years were more likely to have healthy gut permeability and balanced microbes, which increased their protection against Crohn’s disease.
“Our study seems to add to others that have explored the ‘hygiene hypothesis’ which suggests that the lack of exposure to microbes early in life may lead to lack of immune regulation toward environmental microbes,” senior author Williams Turpin, PhD, said in the written statement.
The researchers aren’t sure why they didn’t get the same findings with cats, but Dr. Turpin theorized that dog owners tend to be outside more with their dogs or live in places with more green space, which are good protectors against Crohn’s disease.
It’s all good for dog owners, but do their pets’ parasites make you more attractive? Just more fuel for the ongoing debate.
Come for the history, stay for the fossilized parasites
Another week, another analysis of old British poop. LOTME really is your one-stop shop for all the important, hard-hitting news about historic parasites. You’re welcome, Internet.
The news this week is from Stonehenge, which is apparently kind of a big deal. Rocks in a circle, celestial calendar, cultural significance, whatever. We’re not here to talk about rocks. We’re here to talk about, uh, rocks. Smaller rocks. Specifically, coprolites, which are essentially poop turned into a rock. (Though now we’re imagining Stonehenge made out of fossilized poop rocks. Would it still be a big tourist destination? We can see both sides of the argument on that one.)
Archaeologists from the University of Cambridge have conducted an analysis of coprolites from Durrington Walls, a Neolithic settlement just a few kilometers from Stonehenge. The town dates to the same time that Stonehenge was constructed, and it’s believed that the residents were responsible for building the landmark. These coprolites, depending on what’s inside, can tell us a lot about how the builders of Stonehenge lived and, more specifically, how they ate.
In this case, the coprolites of one human and three dogs contained capillariid worm eggs. These worms come from cows, and when a human is typically infected, the eggs embed in the liver and do not pass through the body. Finding them in excrement indicates that the people were eating raw cow organs and feeding leftovers to their dogs. This is interesting, because a preponderance of pottery and cooking implements also found at the site indicates that the residents of Durrington Walls were spit-roasting or boiling their beef and pork. So the meat was cooked, but not the organs. That is an interesting dietary decision, ancient British people. Then again, modern British cuisine exists. At least now we know where they got it from.
This new research raises one other very important question: When are we going to get a full-on guided tour of all the important coprolite sites in Britain? They’ve clearly got plenty of them, and the tourist demand for ancient parasites must be sky-high. Come on, capitalism, follow through on this. We’d go.
Everyone lies: Food intake edition
Do you have any patients on special diets? Do you ask them if they are following those diets? Don’t bother, because they’re lying. Everyone lies about the food they eat. Everyone. Obese people lie, and nonobese people lie.
Investigators at the University of Essex in England asked 221 adults to keep food diaries, and then they checked on energy consumption by analyzing radioactive water levels in the participants’ urine over a 10-day period.
Underreporting of food consumption was rampant, even among those who were not obese. The obese subjects did underreport by a greater extent (1,200 calories per day) than did those who were not obese, who were off by only 800 calories, but the obese participants burned about 400 calories more each day than did the nonobese, so the difference was a wash.
Everyone ended up underreporting their calorie consumption by an average of about 900 calories, and the investigators were good enough to provide some food equivalents, tops on the list being three MacDonald’s cheeseburgers.
“Public health recommendations have historically relied heavily on self-reported energy intake values,” senior author Gavin Sandercock, PhD, said in a EurekAlert statement, and “recognising that the measures of energy intake are incorrect might result in the setting of more realistic targets.”
Maybe you can be more realistic with your patients, too. Go ahead and ask Mr. Smith about the burger sticking out of his coat pocket, because there are probably two more you can’t see. We’ve each got 900 calories hiding on us somewhere. Ours is usually pizza.
The art of the gallbladder
Ever thought you would see a portrait of a gallbladder hanging up in a gallery? Not just an artist’s rendition, but an actual photo from an actual patient? Well, you can at the Soloway Gallery in Brooklyn, N.Y., at least until June 12.
The artist? K.C. Joseph, MD, a general surgeon from St. Marie, Pa., who died in 2015. His daughter Melissa is the curator of the show and told ARTnews about the interesting connection her father had with art and surgery.
In 2010, Dr. Joseph gave his daughter a box of photos and said “Make me a famous artist,” she recalled. At first, “I was like, ‘These are weird,’ and then I put them under my bed for 10 years.”
Apparently he had been making art with his patients’ organs for about 15 years and had a system in which he put each one together. Before a surgery Dr. Joseph would make a note card with the patient’s name handwritten in calligraphy with a couple of pages taken out of the magazine from the waiting room as the backdrop. Afterward, when the patient was in recovery, the removed organ would be placed among the pages and the name card. A photo was taken with the same endoscope that was used for the procedure.
After the show’s debut, people reached out expressing their love for their photos. “I wish, before he died, I had asked him more questions about it,” Ms. Joseph told ARTnews. “I’m regretting it so much now, kicking myself.”
Who gets to take home an artsy photo of their gallbladder after getting it removed? Not us, that’s who. Each collage is a one-of-a-kind piece. They definitely should be framed and shown in an art gallery. Oh, right. Never mind.
Children and COVID: Weekly cases keep rising past 100,000
, according to the American Academy of Pediatrics and the Children’s Hospital Association.
New cases were up by 14.6% over the previous week to just over 107,000 reported during May 13-16, marking the sixth straight increase since April 1-7, when the count was almost 26,000. Over that period, weekly cases rose 313%, based on data in the latest weekly COVID report from the AAP and CHA.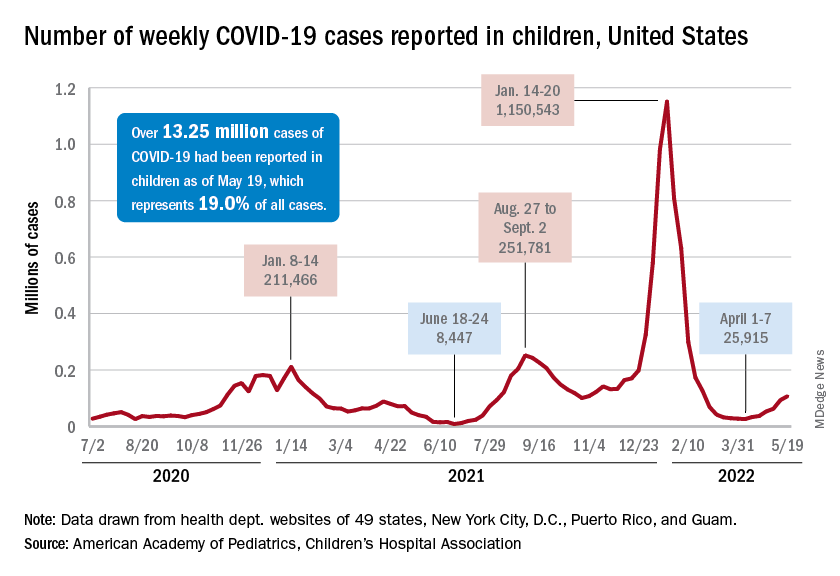
Rates reported by the Centers for Disease Control and Prevention show the same trend. Weekly cases per 100,000 population, which were down to 34.9 in children aged 0-4 years and 43.1 for those aged 5-11 on March 26, were up to 49.5 and 52.2, respectively, by April 16. The pace picked up right after that, and as of May 14, the rates of new cases were 125.4 per 100,000 in children aged 0-4 years and 143.1 in those aged 5-11, the CDC said.
Hospital admissions continue to rise as well. The rate of new admissions in children aged 0-17 was up to 0.25 per 100,000 population on May 18, nearly double the 0.13 per 100,000 recorded as late as April 13. The latest 7-day average count for new admissions, 163 per day from May 15-21, is down from the previous week’s 175 per day, but the CDC also acknowledges potential reporting delays in the most recent 7-day period.
Both of those weekly averages, however, are far below the peak rate for the pandemic, 914 per day, which occurred Jan. 10-16, 2022, during the Omicron surge. Since the CDC began keeping count at the beginning of August 2020, more than 125,000 children aged 0-17 years have been admitted with confirmed COVID-19, which is about 2.7% of all admissions over that period, the CDC’s data show.
Booster gets the green light
The week brought some positive news on the prevention side, though, as the CDC officially approved a COVID vaccine booster dose for children aged 5-11 years.
Even that good news came with a caveat, however. The vote by the CDC’s Advisory Committee on Immunization Practices was 11:1 in favor, with the negative vote cast by Helen Keipp Talbot, MD, of Vanderbilt University, Nashville, Tenn., who said that “boosters are great once we’ve gotten everyone their first round. That needs to be our priority in this.”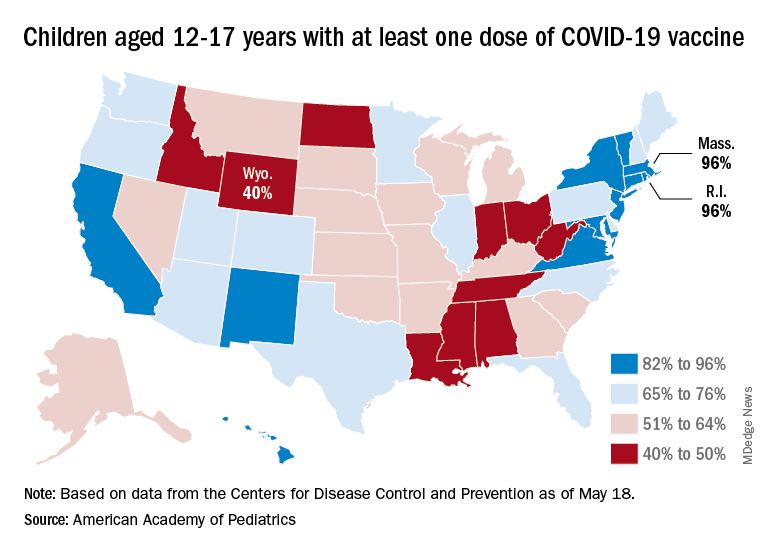
Nationally, in fact, just 35.7% of children aged 5-11 years have received at least one dose of the vaccine and only 29.0% are fully vaccinated. Those figures are nearly doubled among 12- to 17-year-olds: 69.3% have received at least one dose and 59.4% are fully vaccinated, the CDC said on its COVID Data Tracker.
Some states, meanwhile, are well below those national rates. In Wyoming, only 40% of children aged 12-17 have received an initial vaccine dose, and eight other states are below 50%. Among children aged 5-12, there are still five states below 20% in that measure, while the states on the other end of the spectrum – Vermont and Massachusetts – are above 60%, the AAP said in its separate vaccination report.
, according to the American Academy of Pediatrics and the Children’s Hospital Association.
New cases were up by 14.6% over the previous week to just over 107,000 reported during May 13-16, marking the sixth straight increase since April 1-7, when the count was almost 26,000. Over that period, weekly cases rose 313%, based on data in the latest weekly COVID report from the AAP and CHA.
Rates reported by the Centers for Disease Control and Prevention show the same trend. Weekly cases per 100,000 population, which were down to 34.9 in children aged 0-4 years and 43.1 for those aged 5-11 on March 26, were up to 49.5 and 52.2, respectively, by April 16. The pace picked up right after that, and as of May 14, the rates of new cases were 125.4 per 100,000 in children aged 0-4 years and 143.1 in those aged 5-11, the CDC said.
Hospital admissions continue to rise as well. The rate of new admissions in children aged 0-17 was up to 0.25 per 100,000 population on May 18, nearly double the 0.13 per 100,000 recorded as late as April 13. The latest 7-day average count for new admissions, 163 per day from May 15-21, is down from the previous week’s 175 per day, but the CDC also acknowledges potential reporting delays in the most recent 7-day period.
Both of those weekly averages, however, are far below the peak rate for the pandemic, 914 per day, which occurred Jan. 10-16, 2022, during the Omicron surge. Since the CDC began keeping count at the beginning of August 2020, more than 125,000 children aged 0-17 years have been admitted with confirmed COVID-19, which is about 2.7% of all admissions over that period, the CDC’s data show.
Booster gets the green light
The week brought some positive news on the prevention side, though, as the CDC officially approved a COVID vaccine booster dose for children aged 5-11 years.
Even that good news came with a caveat, however. The vote by the CDC’s Advisory Committee on Immunization Practices was 11:1 in favor, with the negative vote cast by Helen Keipp Talbot, MD, of Vanderbilt University, Nashville, Tenn., who said that “boosters are great once we’ve gotten everyone their first round. That needs to be our priority in this.”
Nationally, in fact, just 35.7% of children aged 5-11 years have received at least one dose of the vaccine and only 29.0% are fully vaccinated. Those figures are nearly doubled among 12- to 17-year-olds: 69.3% have received at least one dose and 59.4% are fully vaccinated, the CDC said on its COVID Data Tracker.
Some states, meanwhile, are well below those national rates. In Wyoming, only 40% of children aged 12-17 have received an initial vaccine dose, and eight other states are below 50%. Among children aged 5-12, there are still five states below 20% in that measure, while the states on the other end of the spectrum – Vermont and Massachusetts – are above 60%, the AAP said in its separate vaccination report.
, according to the American Academy of Pediatrics and the Children’s Hospital Association.
New cases were up by 14.6% over the previous week to just over 107,000 reported during May 13-16, marking the sixth straight increase since April 1-7, when the count was almost 26,000. Over that period, weekly cases rose 313%, based on data in the latest weekly COVID report from the AAP and CHA.
Rates reported by the Centers for Disease Control and Prevention show the same trend. Weekly cases per 100,000 population, which were down to 34.9 in children aged 0-4 years and 43.1 for those aged 5-11 on March 26, were up to 49.5 and 52.2, respectively, by April 16. The pace picked up right after that, and as of May 14, the rates of new cases were 125.4 per 100,000 in children aged 0-4 years and 143.1 in those aged 5-11, the CDC said.
Hospital admissions continue to rise as well. The rate of new admissions in children aged 0-17 was up to 0.25 per 100,000 population on May 18, nearly double the 0.13 per 100,000 recorded as late as April 13. The latest 7-day average count for new admissions, 163 per day from May 15-21, is down from the previous week’s 175 per day, but the CDC also acknowledges potential reporting delays in the most recent 7-day period.
Both of those weekly averages, however, are far below the peak rate for the pandemic, 914 per day, which occurred Jan. 10-16, 2022, during the Omicron surge. Since the CDC began keeping count at the beginning of August 2020, more than 125,000 children aged 0-17 years have been admitted with confirmed COVID-19, which is about 2.7% of all admissions over that period, the CDC’s data show.
Booster gets the green light
The week brought some positive news on the prevention side, though, as the CDC officially approved a COVID vaccine booster dose for children aged 5-11 years.
Even that good news came with a caveat, however. The vote by the CDC’s Advisory Committee on Immunization Practices was 11:1 in favor, with the negative vote cast by Helen Keipp Talbot, MD, of Vanderbilt University, Nashville, Tenn., who said that “boosters are great once we’ve gotten everyone their first round. That needs to be our priority in this.”
Nationally, in fact, just 35.7% of children aged 5-11 years have received at least one dose of the vaccine and only 29.0% are fully vaccinated. Those figures are nearly doubled among 12- to 17-year-olds: 69.3% have received at least one dose and 59.4% are fully vaccinated, the CDC said on its COVID Data Tracker.
Some states, meanwhile, are well below those national rates. In Wyoming, only 40% of children aged 12-17 have received an initial vaccine dose, and eight other states are below 50%. Among children aged 5-12, there are still five states below 20% in that measure, while the states on the other end of the spectrum – Vermont and Massachusetts – are above 60%, the AAP said in its separate vaccination report.
FDA, AMA prepare for potential COVID-19 shots for children younger than 6
Regulators and the nation’s largest physician organization took separate steps in recent days to prepare for expected authorization of use of COVID-19 vaccines in children younger than age 6.
The Food and Drug Administration on May 23 announced its Vaccines and Related Biological Products Advisory Committee will meet June 15 to discuss expanding the use of COVID vaccines from Pfizer and Moderna.
The panel will examine a request from Pfizer and its partner BioNTech for an emergency use authorization (EUA) of its vaccine to cover children ages 6 months through 4 years. The EUA expansion for the Moderna shot would cover children ages 6 months through 5 years, the FDA said.
Many parents and physicians have been urging regulators to clear COVID shots for young children, among whom rates of infection are high.
The American Medical Association in February announced an update of its Current Procedural Terminology (CPT) to prepare for an eventual FDA clearance of the Pfizer-BioNTech shot for children aged 6 months to younger than 5 years. On May 19, the association announced a new CPT update to prepare for FDA clearance for use of the Moderna COVID-19 vaccine for children 6 months through 5 years.
“Extending COVID-19 vaccination protection to approximately 18 million young children will significantly reduce their risk of COVID-19 infection, hospitalization, and death, and give their parents incredible peace of mind,” Gerald Harmon, MD, AMA’s president, said in a statement. “We strongly urge all parents to get their infants and toddlers vaccinated as soon as they are eligible for a COVID-19 vaccine.”
Both the Moderna and the Pfizer-BioNTech COVID vaccines would be given to these young children in low doses.
On May 23, Pfizer announced results from a phase 2/3 trial evaluating a series of three shots of its vaccine in children ages 6 months to younger than 5 years.
Vaccine efficacy, which was a secondary endpoint in this study, was 80.3% in this age group, Pfizer said. The analysis was based on 10 symptomatic cases of COVID-19. The trial’s protocol specifies a formal analysis will be performed when at least 21 cases have accrued from 7 days after the third dose. The company said it would share final data on the effectiveness of the vaccine once the results are available.
Moderna on April 28 issued a statement with details about testing of its vaccine in young children. Vaccine efficacy was estimated at about 51% for children aged 6 months to younger than 2 years and 37% for the children aged 2 years to younger than 6. Paul Burton, MD, Moderna’s chief medical officer, spoke about this rate during a May 1 appearance on CBS’ Face the Nation.
“What it means for parents, for caregivers, is that if they give the Moderna vaccine to these little kids, they would basically cut in half the risk of that child getting symptomatic COVID,” Dr. Burton said in the interview. “Now, the number, 50%, I know is often lower than we are used to seeing with our vaccine, but it’s because this study was conducted during a time of Omicron.”
The FDA’s vaccine advisory committee also will meet on June 14 discuss potential use under an EUA of Moderna’s COVID vaccine for children and teenagers aged 6-17 years. The Pfizer-BioNTech vaccine already is authorized under an EUA for people aged 5 years and older.
The FDA has to date granted both conditional clearances, or EUAs, and regular approvals for COVID vaccines.
EUAs are meant to be temporary, allowing for rapid introduction of medicines in response to public health crises such as the pandemic. The FDA also uses EUAs to provide initial clearances of additional indications for products, as would be the case with the authorizations Moderna and Pfizer-BioNTech are seeking for their COVID vaccines.
Companies that want to continue to sell EUA-cleared products or promote EUA-cleared indications beyond the time of the public health crisis must seek regular approvals.
The FDA cleared the Pfizer-BioNTech and Moderna COVID vaccines under EUAs in December 2020. The agency then granted a regular approval for the Pfizer-BioNTech vaccine for people ages 16 and older in August 2021 based on more robust data. Regular approval for the Moderna vaccine for people ages 18 and older followed in January 2022.
Varied reactions among parents
Attitudes in the United States about pediatric COVID vaccines are far from uniform.
The initial uptake has disappointed physicians and researchers, who have been urging wider use of the COVID vaccination among children and teens for whom the FDA already has granted a clearance. Many parents are hesitating to bring their children for the COVID vaccines, according to the Centers for Disease Control and Prevention. Only 35.4% of children ages 5-11 had received at least one dose of a COVID vaccine, CDC staff said during a meeting.
Yet many other parents are demanding this medicine for their young children, urging the FDA to move quickly to clear COVID shots.
A private Facebook group called “Protect Their Future: A Call to Action for COVID Vaccines in Kids <5” boasts about 6,200 members. Many parents and physicians have used Twitter in recent months to press for a speedy review of COVID vaccines for the youngest children, often using the hashtag #immunizeunder5s. A group called Protect Their Future, which uses @ImmunizeUnder5s as its Twitter handle, had 5,288 followers as of the afternoon of May 23.
A special panel of the House of Representatives, the Select Subcommittee on the Coronavirus Crisis, on May 23 joined those tweeting about the need to soon authorize COVID vaccines for very young children.
“Parents have been waiting many months for vaccines for their young children,” the subcommittee tweeted. “They deserve to hear from @US_FDA why this lengthy process has been in children’s best interests.”
A version of this article first appeared on Medscape.com.
Regulators and the nation’s largest physician organization took separate steps in recent days to prepare for expected authorization of use of COVID-19 vaccines in children younger than age 6.
The Food and Drug Administration on May 23 announced its Vaccines and Related Biological Products Advisory Committee will meet June 15 to discuss expanding the use of COVID vaccines from Pfizer and Moderna.
The panel will examine a request from Pfizer and its partner BioNTech for an emergency use authorization (EUA) of its vaccine to cover children ages 6 months through 4 years. The EUA expansion for the Moderna shot would cover children ages 6 months through 5 years, the FDA said.
Many parents and physicians have been urging regulators to clear COVID shots for young children, among whom rates of infection are high.
The American Medical Association in February announced an update of its Current Procedural Terminology (CPT) to prepare for an eventual FDA clearance of the Pfizer-BioNTech shot for children aged 6 months to younger than 5 years. On May 19, the association announced a new CPT update to prepare for FDA clearance for use of the Moderna COVID-19 vaccine for children 6 months through 5 years.
“Extending COVID-19 vaccination protection to approximately 18 million young children will significantly reduce their risk of COVID-19 infection, hospitalization, and death, and give their parents incredible peace of mind,” Gerald Harmon, MD, AMA’s president, said in a statement. “We strongly urge all parents to get their infants and toddlers vaccinated as soon as they are eligible for a COVID-19 vaccine.”
Both the Moderna and the Pfizer-BioNTech COVID vaccines would be given to these young children in low doses.
On May 23, Pfizer announced results from a phase 2/3 trial evaluating a series of three shots of its vaccine in children ages 6 months to younger than 5 years.
Vaccine efficacy, which was a secondary endpoint in this study, was 80.3% in this age group, Pfizer said. The analysis was based on 10 symptomatic cases of COVID-19. The trial’s protocol specifies a formal analysis will be performed when at least 21 cases have accrued from 7 days after the third dose. The company said it would share final data on the effectiveness of the vaccine once the results are available.
Moderna on April 28 issued a statement with details about testing of its vaccine in young children. Vaccine efficacy was estimated at about 51% for children aged 6 months to younger than 2 years and 37% for the children aged 2 years to younger than 6. Paul Burton, MD, Moderna’s chief medical officer, spoke about this rate during a May 1 appearance on CBS’ Face the Nation.
“What it means for parents, for caregivers, is that if they give the Moderna vaccine to these little kids, they would basically cut in half the risk of that child getting symptomatic COVID,” Dr. Burton said in the interview. “Now, the number, 50%, I know is often lower than we are used to seeing with our vaccine, but it’s because this study was conducted during a time of Omicron.”
The FDA’s vaccine advisory committee also will meet on June 14 discuss potential use under an EUA of Moderna’s COVID vaccine for children and teenagers aged 6-17 years. The Pfizer-BioNTech vaccine already is authorized under an EUA for people aged 5 years and older.
The FDA has to date granted both conditional clearances, or EUAs, and regular approvals for COVID vaccines.
EUAs are meant to be temporary, allowing for rapid introduction of medicines in response to public health crises such as the pandemic. The FDA also uses EUAs to provide initial clearances of additional indications for products, as would be the case with the authorizations Moderna and Pfizer-BioNTech are seeking for their COVID vaccines.
Companies that want to continue to sell EUA-cleared products or promote EUA-cleared indications beyond the time of the public health crisis must seek regular approvals.
The FDA cleared the Pfizer-BioNTech and Moderna COVID vaccines under EUAs in December 2020. The agency then granted a regular approval for the Pfizer-BioNTech vaccine for people ages 16 and older in August 2021 based on more robust data. Regular approval for the Moderna vaccine for people ages 18 and older followed in January 2022.
Varied reactions among parents
Attitudes in the United States about pediatric COVID vaccines are far from uniform.
The initial uptake has disappointed physicians and researchers, who have been urging wider use of the COVID vaccination among children and teens for whom the FDA already has granted a clearance. Many parents are hesitating to bring their children for the COVID vaccines, according to the Centers for Disease Control and Prevention. Only 35.4% of children ages 5-11 had received at least one dose of a COVID vaccine, CDC staff said during a meeting.
Yet many other parents are demanding this medicine for their young children, urging the FDA to move quickly to clear COVID shots.
A private Facebook group called “Protect Their Future: A Call to Action for COVID Vaccines in Kids <5” boasts about 6,200 members. Many parents and physicians have used Twitter in recent months to press for a speedy review of COVID vaccines for the youngest children, often using the hashtag #immunizeunder5s. A group called Protect Their Future, which uses @ImmunizeUnder5s as its Twitter handle, had 5,288 followers as of the afternoon of May 23.
A special panel of the House of Representatives, the Select Subcommittee on the Coronavirus Crisis, on May 23 joined those tweeting about the need to soon authorize COVID vaccines for very young children.
“Parents have been waiting many months for vaccines for their young children,” the subcommittee tweeted. “They deserve to hear from @US_FDA why this lengthy process has been in children’s best interests.”
A version of this article first appeared on Medscape.com.
Regulators and the nation’s largest physician organization took separate steps in recent days to prepare for expected authorization of use of COVID-19 vaccines in children younger than age 6.
The Food and Drug Administration on May 23 announced its Vaccines and Related Biological Products Advisory Committee will meet June 15 to discuss expanding the use of COVID vaccines from Pfizer and Moderna.
The panel will examine a request from Pfizer and its partner BioNTech for an emergency use authorization (EUA) of its vaccine to cover children ages 6 months through 4 years. The EUA expansion for the Moderna shot would cover children ages 6 months through 5 years, the FDA said.
Many parents and physicians have been urging regulators to clear COVID shots for young children, among whom rates of infection are high.
The American Medical Association in February announced an update of its Current Procedural Terminology (CPT) to prepare for an eventual FDA clearance of the Pfizer-BioNTech shot for children aged 6 months to younger than 5 years. On May 19, the association announced a new CPT update to prepare for FDA clearance for use of the Moderna COVID-19 vaccine for children 6 months through 5 years.
“Extending COVID-19 vaccination protection to approximately 18 million young children will significantly reduce their risk of COVID-19 infection, hospitalization, and death, and give their parents incredible peace of mind,” Gerald Harmon, MD, AMA’s president, said in a statement. “We strongly urge all parents to get their infants and toddlers vaccinated as soon as they are eligible for a COVID-19 vaccine.”
Both the Moderna and the Pfizer-BioNTech COVID vaccines would be given to these young children in low doses.
On May 23, Pfizer announced results from a phase 2/3 trial evaluating a series of three shots of its vaccine in children ages 6 months to younger than 5 years.
Vaccine efficacy, which was a secondary endpoint in this study, was 80.3% in this age group, Pfizer said. The analysis was based on 10 symptomatic cases of COVID-19. The trial’s protocol specifies a formal analysis will be performed when at least 21 cases have accrued from 7 days after the third dose. The company said it would share final data on the effectiveness of the vaccine once the results are available.
Moderna on April 28 issued a statement with details about testing of its vaccine in young children. Vaccine efficacy was estimated at about 51% for children aged 6 months to younger than 2 years and 37% for the children aged 2 years to younger than 6. Paul Burton, MD, Moderna’s chief medical officer, spoke about this rate during a May 1 appearance on CBS’ Face the Nation.
“What it means for parents, for caregivers, is that if they give the Moderna vaccine to these little kids, they would basically cut in half the risk of that child getting symptomatic COVID,” Dr. Burton said in the interview. “Now, the number, 50%, I know is often lower than we are used to seeing with our vaccine, but it’s because this study was conducted during a time of Omicron.”
The FDA’s vaccine advisory committee also will meet on June 14 discuss potential use under an EUA of Moderna’s COVID vaccine for children and teenagers aged 6-17 years. The Pfizer-BioNTech vaccine already is authorized under an EUA for people aged 5 years and older.
The FDA has to date granted both conditional clearances, or EUAs, and regular approvals for COVID vaccines.
EUAs are meant to be temporary, allowing for rapid introduction of medicines in response to public health crises such as the pandemic. The FDA also uses EUAs to provide initial clearances of additional indications for products, as would be the case with the authorizations Moderna and Pfizer-BioNTech are seeking for their COVID vaccines.
Companies that want to continue to sell EUA-cleared products or promote EUA-cleared indications beyond the time of the public health crisis must seek regular approvals.
The FDA cleared the Pfizer-BioNTech and Moderna COVID vaccines under EUAs in December 2020. The agency then granted a regular approval for the Pfizer-BioNTech vaccine for people ages 16 and older in August 2021 based on more robust data. Regular approval for the Moderna vaccine for people ages 18 and older followed in January 2022.
Varied reactions among parents
Attitudes in the United States about pediatric COVID vaccines are far from uniform.
The initial uptake has disappointed physicians and researchers, who have been urging wider use of the COVID vaccination among children and teens for whom the FDA already has granted a clearance. Many parents are hesitating to bring their children for the COVID vaccines, according to the Centers for Disease Control and Prevention. Only 35.4% of children ages 5-11 had received at least one dose of a COVID vaccine, CDC staff said during a meeting.
Yet many other parents are demanding this medicine for their young children, urging the FDA to move quickly to clear COVID shots.
A private Facebook group called “Protect Their Future: A Call to Action for COVID Vaccines in Kids <5” boasts about 6,200 members. Many parents and physicians have used Twitter in recent months to press for a speedy review of COVID vaccines for the youngest children, often using the hashtag #immunizeunder5s. A group called Protect Their Future, which uses @ImmunizeUnder5s as its Twitter handle, had 5,288 followers as of the afternoon of May 23.
A special panel of the House of Representatives, the Select Subcommittee on the Coronavirus Crisis, on May 23 joined those tweeting about the need to soon authorize COVID vaccines for very young children.
“Parents have been waiting many months for vaccines for their young children,” the subcommittee tweeted. “They deserve to hear from @US_FDA why this lengthy process has been in children’s best interests.”
A version of this article first appeared on Medscape.com.
Videos may not increase vaccinations in IBD
SAN DIEGO – Video and text messaging may not increase the proportion of people with inflammatory bowel disease (IBD) who get influenza vaccinations.
Although patients who received the messages expressed greater intention to get the vaccinations in a trial of the two methods, they didn’t follow through and get the shots, said Keren Appel, MD, a pediatric gastroenterologist at Children’s Hospital of Orange County in Orange, Calif.
“We found there was no difference in the uptake of the influenza vaccine between the two groups,” she said in an interview. Dr. Appel, who participated in the research while at Cedars-Sinai Medical Center in Los Angeles, presented the finding at the annual Digestive Diseases Week® (DDW) 2022.
People with IBD run an increased risk of complications such as infection, bone fractures, and cancer, said Dr. Appel. Previous research has suggested many people with IBD lack understanding or awareness or are skeptical of immunizations.
A previous trial with text-based email reminders did not result in more immunizations, according to Dr. Appel, so she and her colleagues decided to try promoting health prevention with videos. With feedback from patients, they created a series of animations encouraging patients to get influenza, pneumococcal, and zoster vaccinations and screening for bone health and skin cancer.
They randomly assigned 511 to receive videos and 545 patients to receive texts as a control group. After 6 months, 345 patients remained in the text group and 322 remained in the video group. The two groups had similar demographics, health status, and preventive health behaviors. They were mostly educated White women whose IBD was in remission.
The percentage of those who got flu vaccines increased from 59% (for the 2018-2019 season) to 63% (for the 2019-2020 flu season) in the group that watched the videos. However this change did not quite reach statistical significance (P = .07). The change in the text group, from 55% to 57%, was also not significant (P = .23).
The subjects did express more intention to get flu vaccines. The percentage with this intention increased from 59 to 75 in the video group, and from 55 to 72 in the text group. Both changes were statistically significant (P < .001).
Intentions to receive pneumonia and shingles vaccines, and bone and skin cancer screening, were not statistically different between the groups.
The researchers looked at age, immunosuppression, gender, and education to see if these factors could predict who was most likely to get the flu vaccine, but the only significant predictor was having received a previous flu shot.
Dr. Appel speculated that the videos might have been more effective in a more racially diverse, less educated population, or one where fewer people had previously received vaccinations.
“While we didn’t see a difference in this study, I think it opens up a lot of other questions that we can explore and answer,” she said. “It’s possible that patients may not have a one size fits all on their response. Some may respond better to video. Some may respond to text. Some may need more frequent reminders. Some might need to hear it from their doctor directly.”
Session comoderator Alyse Bedell, PhD, an assistant professor of psychiatry and behavioral neuroscience at the University of Chicago, agreed that a different patient population might have responded differently. “A population that may have lower access to educational resources, or has less educational attainment, or may have fewer people in their communities that are already receiving vaccines – those I think are going to be the populations where we’re going to be more likely to see the effects of an intervention like this,” she said in an interview.
Neither Dr. Appel nor Dr. Bedell reported any relevant financial interests. The study was funded by Pfizer.
SAN DIEGO – Video and text messaging may not increase the proportion of people with inflammatory bowel disease (IBD) who get influenza vaccinations.
Although patients who received the messages expressed greater intention to get the vaccinations in a trial of the two methods, they didn’t follow through and get the shots, said Keren Appel, MD, a pediatric gastroenterologist at Children’s Hospital of Orange County in Orange, Calif.
“We found there was no difference in the uptake of the influenza vaccine between the two groups,” she said in an interview. Dr. Appel, who participated in the research while at Cedars-Sinai Medical Center in Los Angeles, presented the finding at the annual Digestive Diseases Week® (DDW) 2022.
People with IBD run an increased risk of complications such as infection, bone fractures, and cancer, said Dr. Appel. Previous research has suggested many people with IBD lack understanding or awareness or are skeptical of immunizations.
A previous trial with text-based email reminders did not result in more immunizations, according to Dr. Appel, so she and her colleagues decided to try promoting health prevention with videos. With feedback from patients, they created a series of animations encouraging patients to get influenza, pneumococcal, and zoster vaccinations and screening for bone health and skin cancer.
They randomly assigned 511 to receive videos and 545 patients to receive texts as a control group. After 6 months, 345 patients remained in the text group and 322 remained in the video group. The two groups had similar demographics, health status, and preventive health behaviors. They were mostly educated White women whose IBD was in remission.
The percentage of those who got flu vaccines increased from 59% (for the 2018-2019 season) to 63% (for the 2019-2020 flu season) in the group that watched the videos. However this change did not quite reach statistical significance (P = .07). The change in the text group, from 55% to 57%, was also not significant (P = .23).
The subjects did express more intention to get flu vaccines. The percentage with this intention increased from 59 to 75 in the video group, and from 55 to 72 in the text group. Both changes were statistically significant (P < .001).
Intentions to receive pneumonia and shingles vaccines, and bone and skin cancer screening, were not statistically different between the groups.
The researchers looked at age, immunosuppression, gender, and education to see if these factors could predict who was most likely to get the flu vaccine, but the only significant predictor was having received a previous flu shot.
Dr. Appel speculated that the videos might have been more effective in a more racially diverse, less educated population, or one where fewer people had previously received vaccinations.
“While we didn’t see a difference in this study, I think it opens up a lot of other questions that we can explore and answer,” she said. “It’s possible that patients may not have a one size fits all on their response. Some may respond better to video. Some may respond to text. Some may need more frequent reminders. Some might need to hear it from their doctor directly.”
Session comoderator Alyse Bedell, PhD, an assistant professor of psychiatry and behavioral neuroscience at the University of Chicago, agreed that a different patient population might have responded differently. “A population that may have lower access to educational resources, or has less educational attainment, or may have fewer people in their communities that are already receiving vaccines – those I think are going to be the populations where we’re going to be more likely to see the effects of an intervention like this,” she said in an interview.
Neither Dr. Appel nor Dr. Bedell reported any relevant financial interests. The study was funded by Pfizer.
SAN DIEGO – Video and text messaging may not increase the proportion of people with inflammatory bowel disease (IBD) who get influenza vaccinations.
Although patients who received the messages expressed greater intention to get the vaccinations in a trial of the two methods, they didn’t follow through and get the shots, said Keren Appel, MD, a pediatric gastroenterologist at Children’s Hospital of Orange County in Orange, Calif.
“We found there was no difference in the uptake of the influenza vaccine between the two groups,” she said in an interview. Dr. Appel, who participated in the research while at Cedars-Sinai Medical Center in Los Angeles, presented the finding at the annual Digestive Diseases Week® (DDW) 2022.
People with IBD run an increased risk of complications such as infection, bone fractures, and cancer, said Dr. Appel. Previous research has suggested many people with IBD lack understanding or awareness or are skeptical of immunizations.
A previous trial with text-based email reminders did not result in more immunizations, according to Dr. Appel, so she and her colleagues decided to try promoting health prevention with videos. With feedback from patients, they created a series of animations encouraging patients to get influenza, pneumococcal, and zoster vaccinations and screening for bone health and skin cancer.
They randomly assigned 511 to receive videos and 545 patients to receive texts as a control group. After 6 months, 345 patients remained in the text group and 322 remained in the video group. The two groups had similar demographics, health status, and preventive health behaviors. They were mostly educated White women whose IBD was in remission.
The percentage of those who got flu vaccines increased from 59% (for the 2018-2019 season) to 63% (for the 2019-2020 flu season) in the group that watched the videos. However this change did not quite reach statistical significance (P = .07). The change in the text group, from 55% to 57%, was also not significant (P = .23).
The subjects did express more intention to get flu vaccines. The percentage with this intention increased from 59 to 75 in the video group, and from 55 to 72 in the text group. Both changes were statistically significant (P < .001).
Intentions to receive pneumonia and shingles vaccines, and bone and skin cancer screening, were not statistically different between the groups.
The researchers looked at age, immunosuppression, gender, and education to see if these factors could predict who was most likely to get the flu vaccine, but the only significant predictor was having received a previous flu shot.
Dr. Appel speculated that the videos might have been more effective in a more racially diverse, less educated population, or one where fewer people had previously received vaccinations.
“While we didn’t see a difference in this study, I think it opens up a lot of other questions that we can explore and answer,” she said. “It’s possible that patients may not have a one size fits all on their response. Some may respond better to video. Some may respond to text. Some may need more frequent reminders. Some might need to hear it from their doctor directly.”
Session comoderator Alyse Bedell, PhD, an assistant professor of psychiatry and behavioral neuroscience at the University of Chicago, agreed that a different patient population might have responded differently. “A population that may have lower access to educational resources, or has less educational attainment, or may have fewer people in their communities that are already receiving vaccines – those I think are going to be the populations where we’re going to be more likely to see the effects of an intervention like this,” she said in an interview.
Neither Dr. Appel nor Dr. Bedell reported any relevant financial interests. The study was funded by Pfizer.
AT DDW 2022
Monkeypox quarantines not needed in U.S., Biden says
He said the United States has enough vaccine doses available to stop any serious outbreaks and to “deal with the likelihood of the problem,” according to The Washington Post .
“I just don’t think it rises to the level of the kind of concern that existed with COVID-19, and the smallpox vaccine works for it,” Biden said during a news conference in Japan.
The World Health Organization has identified monkeypox cases in at least a dozen countries where the disease isn’t typically considered endemic. Generally found in Central and West Africa, the illness has been reported in several European countries, as well as the United States, Canada, and Australia.
On Sunday, Biden told reporters that monkeypox is a “concern in that if it were to spread, it would be consequential.” Administration officials have said the president has been briefed on the disease, the newspaper reported.
Monkeypox spreads through droplets and bodily fluids but doesn’t pass easily between humans and is less contagious than the coronavirus, the Post reported. The CDC has reported that the smallpox vaccine is 85% effective against monkeypox, and the U.S. has licensed two smallpox vaccines that could help in potential outbreaks, including one that specifically targets monkeypox.
Mandatory monkeypox quarantine in Belgium
Belgium is the first country to put a mandatory 21-day quarantine in place for monkeypox patients as cases spread globally, according to CNBC. Health authorities announced the quarantine on Friday after the country recorded its third case.
The quarantine only applies to patients with a confirmed infection. Close contacts aren’t required to self-isolate but are encouraged to be careful and watch for symptoms, especially if they spend time with vulnerable people who could contract a serious illness, CNBC reported.
The United Kingdom has published guidelines to assess risks of monkeypox infection and provide guidance on self-isolation and monitoring. Health officials have said that those who have high exposure risks should self-isolate for 21 days, which includes household contacts or medical professionals who have worked with infected patients.
As of Saturday, the WHO has received reports of 92 confirmed monkeypox cases and 28 suspected cases across 12 countries where the virus isn’t typically found. No deaths linked to the cases have been reported so far.
The outbreaks have caused concern among health officials because most cases don’t have travel links to endemic countries. So far, many cases have spread between men who have sex with men, and the cases have been identified as patients seek care in primary care and sexual health clinics, the WHO reported.
“The identification of confirmed and suspected cases of monkeypox with no direct travel links to an endemic area represents a highly unusual event,” the WHO said. “Available information suggests that human-to-human transmission is occurring among people in close physical contact with cases who are symptomatic.”
The WHO said Saturday that more outbreaks will be reported as health officials uncover new information. The fast growth in community cases, especially in urban areas, suggests that a wider outbreak could be possible.
“To have it appear now – more than 100 cases in 12 different countries with no obvious connection – means we have to figure out exactly what’s happening,” Seth Berkley, MD, the CEO of global vaccine alliance Gavi, told CNBC.
“The truth is, we don’t know what that is and therefore how severe it’s going to be,” he said. “But it’s likely that we’re going to see more cases.”
White House health official doesn’t foresee major outbreak
Ashish Jha, MD, a top Biden administration health official who serves as the White House COVID-19 response coordinator, said Sunday that he doesn’t expect monkeypox to have widespread effects in the U.S.
“I feel like this is a virus we understand,” he said on ABC News’s This Week.
The virus has been monitored for decades, and there are treatments for it, Dr. Jha said.
“We have vaccines against it. We have treatments against it,” he said. “It’s not as contagious as COVID. So, I am confident we’re going to be able to keep our arms around it.”
At the same time, Dr. Jha agreed that health officials should keep an eye on the situation. Cases have been confirmed in recent days in several countries, as well as the United States.
“I would not be surprised if we see a few more cases in the upcoming days,” he said. “Any time we have an infectious outbreak like this, we should all be paying attention.”
Dr. Jha also stressed ongoing caution amid the COVID-19 pandemic as cases once again surpass 100,000 daily infections. Variants will continue to evolve, he said, and ongoing outbreaks will reinfect people who have been vaccinated or had a previous infection.
“What we know is that this virus is evolving very quickly, and every iteration of it has more and more immune escape,” he said. “That makes it harder for this virus to be contained unless we continue vaccinating people and keeping people up to date.”
Third possible U.S. monkeypox case found in Florida
The CDC said Sunday that it may have found a third monkeypox case in the United States and is running tests on a patient in South Florida, according to Reuters.
The person is in Broward County and remains isolated. The case appears to be related to international travel, the CDC told Reuters.
Health officials are doing tests to confirm if the patient has the disease, with results expected “soon.” No other cases have been identified in Florida so far.
The first monkeypox case in the United States was reported in Massachusetts last week. The patient had recently traveled to Canada.
The second U.S. case was reported in a New York City resident who tested positive on Friday.
The disease, which is like human smallpox but milder, is a viral infection that was first found in the Democratic Republic of Congo in the 1970s. Symptoms include fever, headaches, and a skin rash across the body.
A version of this article first appeared on WebMD.com.
He said the United States has enough vaccine doses available to stop any serious outbreaks and to “deal with the likelihood of the problem,” according to The Washington Post .
“I just don’t think it rises to the level of the kind of concern that existed with COVID-19, and the smallpox vaccine works for it,” Biden said during a news conference in Japan.
The World Health Organization has identified monkeypox cases in at least a dozen countries where the disease isn’t typically considered endemic. Generally found in Central and West Africa, the illness has been reported in several European countries, as well as the United States, Canada, and Australia.
On Sunday, Biden told reporters that monkeypox is a “concern in that if it were to spread, it would be consequential.” Administration officials have said the president has been briefed on the disease, the newspaper reported.
Monkeypox spreads through droplets and bodily fluids but doesn’t pass easily between humans and is less contagious than the coronavirus, the Post reported. The CDC has reported that the smallpox vaccine is 85% effective against monkeypox, and the U.S. has licensed two smallpox vaccines that could help in potential outbreaks, including one that specifically targets monkeypox.
Mandatory monkeypox quarantine in Belgium
Belgium is the first country to put a mandatory 21-day quarantine in place for monkeypox patients as cases spread globally, according to CNBC. Health authorities announced the quarantine on Friday after the country recorded its third case.
The quarantine only applies to patients with a confirmed infection. Close contacts aren’t required to self-isolate but are encouraged to be careful and watch for symptoms, especially if they spend time with vulnerable people who could contract a serious illness, CNBC reported.
The United Kingdom has published guidelines to assess risks of monkeypox infection and provide guidance on self-isolation and monitoring. Health officials have said that those who have high exposure risks should self-isolate for 21 days, which includes household contacts or medical professionals who have worked with infected patients.
As of Saturday, the WHO has received reports of 92 confirmed monkeypox cases and 28 suspected cases across 12 countries where the virus isn’t typically found. No deaths linked to the cases have been reported so far.
The outbreaks have caused concern among health officials because most cases don’t have travel links to endemic countries. So far, many cases have spread between men who have sex with men, and the cases have been identified as patients seek care in primary care and sexual health clinics, the WHO reported.
“The identification of confirmed and suspected cases of monkeypox with no direct travel links to an endemic area represents a highly unusual event,” the WHO said. “Available information suggests that human-to-human transmission is occurring among people in close physical contact with cases who are symptomatic.”
The WHO said Saturday that more outbreaks will be reported as health officials uncover new information. The fast growth in community cases, especially in urban areas, suggests that a wider outbreak could be possible.
“To have it appear now – more than 100 cases in 12 different countries with no obvious connection – means we have to figure out exactly what’s happening,” Seth Berkley, MD, the CEO of global vaccine alliance Gavi, told CNBC.
“The truth is, we don’t know what that is and therefore how severe it’s going to be,” he said. “But it’s likely that we’re going to see more cases.”
White House health official doesn’t foresee major outbreak
Ashish Jha, MD, a top Biden administration health official who serves as the White House COVID-19 response coordinator, said Sunday that he doesn’t expect monkeypox to have widespread effects in the U.S.
“I feel like this is a virus we understand,” he said on ABC News’s This Week.
The virus has been monitored for decades, and there are treatments for it, Dr. Jha said.
“We have vaccines against it. We have treatments against it,” he said. “It’s not as contagious as COVID. So, I am confident we’re going to be able to keep our arms around it.”
At the same time, Dr. Jha agreed that health officials should keep an eye on the situation. Cases have been confirmed in recent days in several countries, as well as the United States.
“I would not be surprised if we see a few more cases in the upcoming days,” he said. “Any time we have an infectious outbreak like this, we should all be paying attention.”
Dr. Jha also stressed ongoing caution amid the COVID-19 pandemic as cases once again surpass 100,000 daily infections. Variants will continue to evolve, he said, and ongoing outbreaks will reinfect people who have been vaccinated or had a previous infection.
“What we know is that this virus is evolving very quickly, and every iteration of it has more and more immune escape,” he said. “That makes it harder for this virus to be contained unless we continue vaccinating people and keeping people up to date.”
Third possible U.S. monkeypox case found in Florida
The CDC said Sunday that it may have found a third monkeypox case in the United States and is running tests on a patient in South Florida, according to Reuters.
The person is in Broward County and remains isolated. The case appears to be related to international travel, the CDC told Reuters.
Health officials are doing tests to confirm if the patient has the disease, with results expected “soon.” No other cases have been identified in Florida so far.
The first monkeypox case in the United States was reported in Massachusetts last week. The patient had recently traveled to Canada.
The second U.S. case was reported in a New York City resident who tested positive on Friday.
The disease, which is like human smallpox but milder, is a viral infection that was first found in the Democratic Republic of Congo in the 1970s. Symptoms include fever, headaches, and a skin rash across the body.
A version of this article first appeared on WebMD.com.
He said the United States has enough vaccine doses available to stop any serious outbreaks and to “deal with the likelihood of the problem,” according to The Washington Post .
“I just don’t think it rises to the level of the kind of concern that existed with COVID-19, and the smallpox vaccine works for it,” Biden said during a news conference in Japan.
The World Health Organization has identified monkeypox cases in at least a dozen countries where the disease isn’t typically considered endemic. Generally found in Central and West Africa, the illness has been reported in several European countries, as well as the United States, Canada, and Australia.
On Sunday, Biden told reporters that monkeypox is a “concern in that if it were to spread, it would be consequential.” Administration officials have said the president has been briefed on the disease, the newspaper reported.
Monkeypox spreads through droplets and bodily fluids but doesn’t pass easily between humans and is less contagious than the coronavirus, the Post reported. The CDC has reported that the smallpox vaccine is 85% effective against monkeypox, and the U.S. has licensed two smallpox vaccines that could help in potential outbreaks, including one that specifically targets monkeypox.
Mandatory monkeypox quarantine in Belgium
Belgium is the first country to put a mandatory 21-day quarantine in place for monkeypox patients as cases spread globally, according to CNBC. Health authorities announced the quarantine on Friday after the country recorded its third case.
The quarantine only applies to patients with a confirmed infection. Close contacts aren’t required to self-isolate but are encouraged to be careful and watch for symptoms, especially if they spend time with vulnerable people who could contract a serious illness, CNBC reported.
The United Kingdom has published guidelines to assess risks of monkeypox infection and provide guidance on self-isolation and monitoring. Health officials have said that those who have high exposure risks should self-isolate for 21 days, which includes household contacts or medical professionals who have worked with infected patients.
As of Saturday, the WHO has received reports of 92 confirmed monkeypox cases and 28 suspected cases across 12 countries where the virus isn’t typically found. No deaths linked to the cases have been reported so far.
The outbreaks have caused concern among health officials because most cases don’t have travel links to endemic countries. So far, many cases have spread between men who have sex with men, and the cases have been identified as patients seek care in primary care and sexual health clinics, the WHO reported.
“The identification of confirmed and suspected cases of monkeypox with no direct travel links to an endemic area represents a highly unusual event,” the WHO said. “Available information suggests that human-to-human transmission is occurring among people in close physical contact with cases who are symptomatic.”
The WHO said Saturday that more outbreaks will be reported as health officials uncover new information. The fast growth in community cases, especially in urban areas, suggests that a wider outbreak could be possible.
“To have it appear now – more than 100 cases in 12 different countries with no obvious connection – means we have to figure out exactly what’s happening,” Seth Berkley, MD, the CEO of global vaccine alliance Gavi, told CNBC.
“The truth is, we don’t know what that is and therefore how severe it’s going to be,” he said. “But it’s likely that we’re going to see more cases.”
White House health official doesn’t foresee major outbreak
Ashish Jha, MD, a top Biden administration health official who serves as the White House COVID-19 response coordinator, said Sunday that he doesn’t expect monkeypox to have widespread effects in the U.S.
“I feel like this is a virus we understand,” he said on ABC News’s This Week.
The virus has been monitored for decades, and there are treatments for it, Dr. Jha said.
“We have vaccines against it. We have treatments against it,” he said. “It’s not as contagious as COVID. So, I am confident we’re going to be able to keep our arms around it.”
At the same time, Dr. Jha agreed that health officials should keep an eye on the situation. Cases have been confirmed in recent days in several countries, as well as the United States.
“I would not be surprised if we see a few more cases in the upcoming days,” he said. “Any time we have an infectious outbreak like this, we should all be paying attention.”
Dr. Jha also stressed ongoing caution amid the COVID-19 pandemic as cases once again surpass 100,000 daily infections. Variants will continue to evolve, he said, and ongoing outbreaks will reinfect people who have been vaccinated or had a previous infection.
“What we know is that this virus is evolving very quickly, and every iteration of it has more and more immune escape,” he said. “That makes it harder for this virus to be contained unless we continue vaccinating people and keeping people up to date.”
Third possible U.S. monkeypox case found in Florida
The CDC said Sunday that it may have found a third monkeypox case in the United States and is running tests on a patient in South Florida, according to Reuters.
The person is in Broward County and remains isolated. The case appears to be related to international travel, the CDC told Reuters.
Health officials are doing tests to confirm if the patient has the disease, with results expected “soon.” No other cases have been identified in Florida so far.
The first monkeypox case in the United States was reported in Massachusetts last week. The patient had recently traveled to Canada.
The second U.S. case was reported in a New York City resident who tested positive on Friday.
The disease, which is like human smallpox but milder, is a viral infection that was first found in the Democratic Republic of Congo in the 1970s. Symptoms include fever, headaches, and a skin rash across the body.
A version of this article first appeared on WebMD.com.
Fever after a tropical trip: A guide to differential diagnosis
After 2 years of a pandemic in which traveling was barely possible, tropical diseases are becoming important once more. At a 2022 conference for internal medicine specialists, tropical medicine specialist Fritz Holst, MD, of the Center for Tropical and Travel Medicine in Marburg, Germany, explained what questions you should be asking travelers with a fever at your practice and how to proceed with a suspected case.
The following article is based on the lecture: “Differential Diagnosis of Fever After a Trip to the Tropics,” which Dr. Holst gave at the 128th conference of the German Society of Internal Medicine.
A meta-analysis of studies concerning the topic, “returnee travelers from the tropics with fever,” was published in 2020. According to the analysis, purely tropical infections make up a third (33%) of fever diagnoses worldwide following an exotic trip. Malaria accounts for a fifth (22%), 5% are dengue fever, and 2.2% are typhoid (enteric fever).
In 26% of the returnee travelers investigated, nontropical infections were the cause of the fever. Acute gastroenteritis was responsible for 14%, and respiratory infections were responsible for 13%. In 18% of the cases, the cause of the fever remained unclear.
In Germany, the number of malaria cases has increased, said Dr. Holst. In Hessen, for example, there was recently a malaria fatality. “What we should do has been forgotten again,” he warned. More attention should also be paid once more to prophylaxis.
How to proceed
Dr. Holst described the following steps for treating recently returned travelers who are sick:
- Severely ill or not: If there are signs of a severe disease, such as dyspnea, signs of bleeding, hypotension, or central nervous system symptoms, the patient should be referred to a clinic. A diagnosis should be made within 1 day and treatment should be started.
- Transmissible or dangerous disease: This question should be quickly clarified to protect health care personnel, especially those treating patients. By using a thorough medical history (discussed below), a range of diseases may be clarified.
- Disease outbreak in destination country: Find out about possible disease outbreaks in the country that the traveler visited.
- Malaria? Immediate diagnostics: Malaria should always be excluded in patients at the practice on the same day by using a thick blood smear, even if no fever is present. If this is not possible because of time constraints, the affected person should be transferred directly to the clinic.
- Fever independent of the travel? Exclude other causes of the fever (for example, endocarditis).
- Involve tropical medicine specialists in a timely manner.
Nine mandatory questions
Dr. Holst also listed nine questions that clinicians should ask this patient population.
Where were you exactly?
Depending on the regional prevalence of tropical diseases, certain pathogens can be excluded quickly. Approximately 35% of travelers returning from Africa have malaria, whereas typhoid is much rarer. In contrast, typhoid and dengue fever are much more widespread in Southeast Asia. In Latin America, this is the case for both dengue fever and leptospirosis.
When did you travel?
By using the incubation time of the pathogen in question, as well as the time of return journey, you can determine which diseases are possible and which are not. In one patient who visited the practice 4 weeks after his return, dengue or typhoid were excluded.
Where did you stay overnight?
Whether in an unhygienic bed or under the stars, the question regarding how and where travelers stayed overnight provides important evidence of the following nocturnal vectors:
- Sandflies: Leishmaniasis
- Kissing bugs: Chagas disease
- Fleas: Spotted fever, bubonic plague
- Mosquitoes: Malaria, dengue, filariasis
What did you eat?
Many infections can be attributed to careless eating. For example, when eating fish, crabs, crawfish, or frogs, especially if raw, liver fluke, lung fluke, or ciguatera should be considered. Mussel toxins have been found on the coast of Kenya and even in the south of France. In North African countries, you should be cautious when eating nonpasteurized milk products (for example, camel milk). They can transmit the pathogens for brucellosis and tuberculosis. In beef or pork that has not been cooked thoroughly, there is the risk of trichinosis or of a tapeworm. Even vegetarians need to be careful. Infections with the common liver fluke are possible after eating watercress.
What have you been doing?
You can only get some diseases through certain activities, said Dr. Holst. If long-distance travelers tell you about the following excursions, prick up your ears:
- Freshwater contact: Schistosomiasis, leptospirosis
- Caving: Histoplasmosis, rabies
- Excavations: Anthrax, coccidioidomycosis
- Camel tour: MERS coronavirus (Do not mount a sniffling camel!)
- Walking around barefoot: Strongyloides, hookworm
Was there contact with animals?
Because of the risk of rabies following contact with cats or biting apes, Dr. Holst advised long-distance travelers to get vaccinated.
Were there new sexual partners?
In the event of new sexual contacts, tests for hepatitis A, B, C, and HIV should be performed.
Are you undergoing medical treatment?
The patient may already be under medical supervision because of having a disease.
What prophylactic measures did you take before traveling?
To progress in the differential diagnosis, questions should also be asked regarding prophylactic measures. Vaccination against hepatitis A provides very efficient infection protection, whereas vaccines against typhoid offer a much lower level of protection.
Diagnostic tests
As long as there are no abnormalities, such as meningism or heart murmurs, further diagnostics include routine infectiologic laboratory investigations (C-reactive protein, blood count, etc), blood culture (aerobic, anaerobic), a urine dipstick test, and rapid tests for malaria and dengue.
To exclude malaria, a thick blood smear should always be performed on the same day, said Dr. Holst. “The rapid test is occasionally negative. But you often only detect tertian malaria in the thick blood smear. And you have to repeat the diagnostics the following day.” For this, it is important to know that a single test result does not exclude malaria right away. In contrast, detecting malaria antibodies is obsolete. Depending on the result, further tests include serologies, antigen investigations, and polymerase chain reaction.
Treat early
A complete set of results is not always available promptly. Experts recommend that, “if you already have a hunch, then start the therapy, even without a definite diagnosis.” This applies in particular for the suspected diagnoses in the following table.
This article was translated from Coliquio. A version of this article appeared on Medscape.com.
After 2 years of a pandemic in which traveling was barely possible, tropical diseases are becoming important once more. At a 2022 conference for internal medicine specialists, tropical medicine specialist Fritz Holst, MD, of the Center for Tropical and Travel Medicine in Marburg, Germany, explained what questions you should be asking travelers with a fever at your practice and how to proceed with a suspected case.
The following article is based on the lecture: “Differential Diagnosis of Fever After a Trip to the Tropics,” which Dr. Holst gave at the 128th conference of the German Society of Internal Medicine.
A meta-analysis of studies concerning the topic, “returnee travelers from the tropics with fever,” was published in 2020. According to the analysis, purely tropical infections make up a third (33%) of fever diagnoses worldwide following an exotic trip. Malaria accounts for a fifth (22%), 5% are dengue fever, and 2.2% are typhoid (enteric fever).
In 26% of the returnee travelers investigated, nontropical infections were the cause of the fever. Acute gastroenteritis was responsible for 14%, and respiratory infections were responsible for 13%. In 18% of the cases, the cause of the fever remained unclear.
In Germany, the number of malaria cases has increased, said Dr. Holst. In Hessen, for example, there was recently a malaria fatality. “What we should do has been forgotten again,” he warned. More attention should also be paid once more to prophylaxis.
How to proceed
Dr. Holst described the following steps for treating recently returned travelers who are sick:
- Severely ill or not: If there are signs of a severe disease, such as dyspnea, signs of bleeding, hypotension, or central nervous system symptoms, the patient should be referred to a clinic. A diagnosis should be made within 1 day and treatment should be started.
- Transmissible or dangerous disease: This question should be quickly clarified to protect health care personnel, especially those treating patients. By using a thorough medical history (discussed below), a range of diseases may be clarified.
- Disease outbreak in destination country: Find out about possible disease outbreaks in the country that the traveler visited.
- Malaria? Immediate diagnostics: Malaria should always be excluded in patients at the practice on the same day by using a thick blood smear, even if no fever is present. If this is not possible because of time constraints, the affected person should be transferred directly to the clinic.
- Fever independent of the travel? Exclude other causes of the fever (for example, endocarditis).
- Involve tropical medicine specialists in a timely manner.
Nine mandatory questions
Dr. Holst also listed nine questions that clinicians should ask this patient population.
Where were you exactly?
Depending on the regional prevalence of tropical diseases, certain pathogens can be excluded quickly. Approximately 35% of travelers returning from Africa have malaria, whereas typhoid is much rarer. In contrast, typhoid and dengue fever are much more widespread in Southeast Asia. In Latin America, this is the case for both dengue fever and leptospirosis.
When did you travel?
By using the incubation time of the pathogen in question, as well as the time of return journey, you can determine which diseases are possible and which are not. In one patient who visited the practice 4 weeks after his return, dengue or typhoid were excluded.
Where did you stay overnight?
Whether in an unhygienic bed or under the stars, the question regarding how and where travelers stayed overnight provides important evidence of the following nocturnal vectors:
- Sandflies: Leishmaniasis
- Kissing bugs: Chagas disease
- Fleas: Spotted fever, bubonic plague
- Mosquitoes: Malaria, dengue, filariasis
What did you eat?
Many infections can be attributed to careless eating. For example, when eating fish, crabs, crawfish, or frogs, especially if raw, liver fluke, lung fluke, or ciguatera should be considered. Mussel toxins have been found on the coast of Kenya and even in the south of France. In North African countries, you should be cautious when eating nonpasteurized milk products (for example, camel milk). They can transmit the pathogens for brucellosis and tuberculosis. In beef or pork that has not been cooked thoroughly, there is the risk of trichinosis or of a tapeworm. Even vegetarians need to be careful. Infections with the common liver fluke are possible after eating watercress.
What have you been doing?
You can only get some diseases through certain activities, said Dr. Holst. If long-distance travelers tell you about the following excursions, prick up your ears:
- Freshwater contact: Schistosomiasis, leptospirosis
- Caving: Histoplasmosis, rabies
- Excavations: Anthrax, coccidioidomycosis
- Camel tour: MERS coronavirus (Do not mount a sniffling camel!)
- Walking around barefoot: Strongyloides, hookworm
Was there contact with animals?
Because of the risk of rabies following contact with cats or biting apes, Dr. Holst advised long-distance travelers to get vaccinated.
Were there new sexual partners?
In the event of new sexual contacts, tests for hepatitis A, B, C, and HIV should be performed.
Are you undergoing medical treatment?
The patient may already be under medical supervision because of having a disease.
What prophylactic measures did you take before traveling?
To progress in the differential diagnosis, questions should also be asked regarding prophylactic measures. Vaccination against hepatitis A provides very efficient infection protection, whereas vaccines against typhoid offer a much lower level of protection.
Diagnostic tests
As long as there are no abnormalities, such as meningism or heart murmurs, further diagnostics include routine infectiologic laboratory investigations (C-reactive protein, blood count, etc), blood culture (aerobic, anaerobic), a urine dipstick test, and rapid tests for malaria and dengue.
To exclude malaria, a thick blood smear should always be performed on the same day, said Dr. Holst. “The rapid test is occasionally negative. But you often only detect tertian malaria in the thick blood smear. And you have to repeat the diagnostics the following day.” For this, it is important to know that a single test result does not exclude malaria right away. In contrast, detecting malaria antibodies is obsolete. Depending on the result, further tests include serologies, antigen investigations, and polymerase chain reaction.
Treat early
A complete set of results is not always available promptly. Experts recommend that, “if you already have a hunch, then start the therapy, even without a definite diagnosis.” This applies in particular for the suspected diagnoses in the following table.
This article was translated from Coliquio. A version of this article appeared on Medscape.com.
After 2 years of a pandemic in which traveling was barely possible, tropical diseases are becoming important once more. At a 2022 conference for internal medicine specialists, tropical medicine specialist Fritz Holst, MD, of the Center for Tropical and Travel Medicine in Marburg, Germany, explained what questions you should be asking travelers with a fever at your practice and how to proceed with a suspected case.
The following article is based on the lecture: “Differential Diagnosis of Fever After a Trip to the Tropics,” which Dr. Holst gave at the 128th conference of the German Society of Internal Medicine.
A meta-analysis of studies concerning the topic, “returnee travelers from the tropics with fever,” was published in 2020. According to the analysis, purely tropical infections make up a third (33%) of fever diagnoses worldwide following an exotic trip. Malaria accounts for a fifth (22%), 5% are dengue fever, and 2.2% are typhoid (enteric fever).
In 26% of the returnee travelers investigated, nontropical infections were the cause of the fever. Acute gastroenteritis was responsible for 14%, and respiratory infections were responsible for 13%. In 18% of the cases, the cause of the fever remained unclear.
In Germany, the number of malaria cases has increased, said Dr. Holst. In Hessen, for example, there was recently a malaria fatality. “What we should do has been forgotten again,” he warned. More attention should also be paid once more to prophylaxis.
How to proceed
Dr. Holst described the following steps for treating recently returned travelers who are sick:
- Severely ill or not: If there are signs of a severe disease, such as dyspnea, signs of bleeding, hypotension, or central nervous system symptoms, the patient should be referred to a clinic. A diagnosis should be made within 1 day and treatment should be started.
- Transmissible or dangerous disease: This question should be quickly clarified to protect health care personnel, especially those treating patients. By using a thorough medical history (discussed below), a range of diseases may be clarified.
- Disease outbreak in destination country: Find out about possible disease outbreaks in the country that the traveler visited.
- Malaria? Immediate diagnostics: Malaria should always be excluded in patients at the practice on the same day by using a thick blood smear, even if no fever is present. If this is not possible because of time constraints, the affected person should be transferred directly to the clinic.
- Fever independent of the travel? Exclude other causes of the fever (for example, endocarditis).
- Involve tropical medicine specialists in a timely manner.
Nine mandatory questions
Dr. Holst also listed nine questions that clinicians should ask this patient population.
Where were you exactly?
Depending on the regional prevalence of tropical diseases, certain pathogens can be excluded quickly. Approximately 35% of travelers returning from Africa have malaria, whereas typhoid is much rarer. In contrast, typhoid and dengue fever are much more widespread in Southeast Asia. In Latin America, this is the case for both dengue fever and leptospirosis.
When did you travel?
By using the incubation time of the pathogen in question, as well as the time of return journey, you can determine which diseases are possible and which are not. In one patient who visited the practice 4 weeks after his return, dengue or typhoid were excluded.
Where did you stay overnight?
Whether in an unhygienic bed or under the stars, the question regarding how and where travelers stayed overnight provides important evidence of the following nocturnal vectors:
- Sandflies: Leishmaniasis
- Kissing bugs: Chagas disease
- Fleas: Spotted fever, bubonic plague
- Mosquitoes: Malaria, dengue, filariasis
What did you eat?
Many infections can be attributed to careless eating. For example, when eating fish, crabs, crawfish, or frogs, especially if raw, liver fluke, lung fluke, or ciguatera should be considered. Mussel toxins have been found on the coast of Kenya and even in the south of France. In North African countries, you should be cautious when eating nonpasteurized milk products (for example, camel milk). They can transmit the pathogens for brucellosis and tuberculosis. In beef or pork that has not been cooked thoroughly, there is the risk of trichinosis or of a tapeworm. Even vegetarians need to be careful. Infections with the common liver fluke are possible after eating watercress.
What have you been doing?
You can only get some diseases through certain activities, said Dr. Holst. If long-distance travelers tell you about the following excursions, prick up your ears:
- Freshwater contact: Schistosomiasis, leptospirosis
- Caving: Histoplasmosis, rabies
- Excavations: Anthrax, coccidioidomycosis
- Camel tour: MERS coronavirus (Do not mount a sniffling camel!)
- Walking around barefoot: Strongyloides, hookworm
Was there contact with animals?
Because of the risk of rabies following contact with cats or biting apes, Dr. Holst advised long-distance travelers to get vaccinated.
Were there new sexual partners?
In the event of new sexual contacts, tests for hepatitis A, B, C, and HIV should be performed.
Are you undergoing medical treatment?
The patient may already be under medical supervision because of having a disease.
What prophylactic measures did you take before traveling?
To progress in the differential diagnosis, questions should also be asked regarding prophylactic measures. Vaccination against hepatitis A provides very efficient infection protection, whereas vaccines against typhoid offer a much lower level of protection.
Diagnostic tests
As long as there are no abnormalities, such as meningism or heart murmurs, further diagnostics include routine infectiologic laboratory investigations (C-reactive protein, blood count, etc), blood culture (aerobic, anaerobic), a urine dipstick test, and rapid tests for malaria and dengue.
To exclude malaria, a thick blood smear should always be performed on the same day, said Dr. Holst. “The rapid test is occasionally negative. But you often only detect tertian malaria in the thick blood smear. And you have to repeat the diagnostics the following day.” For this, it is important to know that a single test result does not exclude malaria right away. In contrast, detecting malaria antibodies is obsolete. Depending on the result, further tests include serologies, antigen investigations, and polymerase chain reaction.
Treat early
A complete set of results is not always available promptly. Experts recommend that, “if you already have a hunch, then start the therapy, even without a definite diagnosis.” This applies in particular for the suspected diagnoses in the following table.
This article was translated from Coliquio. A version of this article appeared on Medscape.com.
Doxycycline bests azithromycin for anorectal chlamydia in women
NEW YORK (Reuters) – A one-week course of doxycycline was superior to a single dose of azithromycin in women with concurrent vaginal and anorectal chlamydia infection in an unblinded randomized controlled trial, mirroring previous results in men.
Researchers suggest that doxycycline should be the first-line therapy for chlamydia infection in women.
“It is clear we must consider that any woman with a urogenital infection must have an effective treatment for the anal infection, since nearly 80% of women have an anal infection concomitant with the vaginal infection,” Dr. Bertille de Barbeyrac of the University of Bordeaux, France, told Reuters Health by email.
However, she noted that “even [though] the study shows that doxycycline is more effective than azithromycin on anal infection, other studies are needed to prove that residual anal infection after treatment with azithromycin can be a source of vaginal contamination and therefore justify changing practices and eliminating azithromycin as a treatment for lower urogenital chlamydial infection in women.”
“There are other reasons [to make] this change,” she added, “such as the acquisition of macrolide resistance by M. genitalium following heavy use of azithromycin.”
As reported in The Lancet Infectious Diseases, Dr. Barbeyrac and colleagues randomly assigned 460 women (median age, 21) to either doxycycline or azithromycin in a multicenter, open-label superiority trial.
Participants received either azithromycin (a single 1-g dose, with or without food) or doxycycline (100 mg in the morning and evening at mealtimes for 7 days – that is, 100 mg of doxycycline twice daily).
The primary outcome was that the microbiological anorectal cure rate, defined as a C. trachomatis-negative nucleic acid amplification test (NAAT), resulted in anorectal specimens six weeks after treatment initiation among women who had a baseline positive result (about half the women in each treatment group).
Ninety-four percent of the doxycycline group versus 85% of the azithromycin group had an anorectal cure (adjusted odds ratio with imputation of missing values, 0.43).
Adverse events possibly related to treatment occurred in 11% of the doxycycline group versus 13% of the azithromycin group. Gastrointestinal disorders were most frequent, occurring in 8% of the doxycycline and 11% of the azithromycin groups.
Summing up, the authors write, “The microbiological anorectal cure rate was significantly lower among women who received a single dose of azithromycin than among those who received a 1-week course of doxycycline. This finding suggests that doxycycline should be the first-line therapy for C trachomatis infection in women.”
Dr. Meleen Chuang, medical director of women’s health at the Family Health Centers at NYU Langone, Brooklyn, commented in an email to Reuters Health that after reviewing this study “as well as CDC and WHO recommendations updated as of 2022, health care providers should be treating C. trachomatis infections with doxycycline 100 mg twice a day for seven days as first-line therapy rather than azithromycin, [given] concerns of increasing macrolide drug resistance against Mycoplasma genitalium and Neisseria gonorrhea.”
“Our clinicians also see the growing uptick of syphilis, gonorrhea, and chlamydia infections in our population, similarly to the rest of the United States since 2020,” she noted. “With the increase in STD infection ... treatment with doxycycline therapy with an important caveat to the patient to complete the one-week treatment regimen is extremely important.”
Dr. Latasha Murphy of the Gynecologic Care Institute at Mercy, Baltimore, also commented in an email to Reuters Health. She noted, “this study does not mirror my clinical experience. More patients have side effects from doxycycline than azithromycin in my experience. Also, anorectal screening is not routine in STD screening.”
“If any major changes to clinical care are made,” she said, “it may be for more consistent screening for anorectal disease. This may ultimately lead to doxycycline being the first line-treatment. More research is needed before making any definitive changes.”
Reuters Health Information © 2022
NEW YORK (Reuters) – A one-week course of doxycycline was superior to a single dose of azithromycin in women with concurrent vaginal and anorectal chlamydia infection in an unblinded randomized controlled trial, mirroring previous results in men.
Researchers suggest that doxycycline should be the first-line therapy for chlamydia infection in women.
“It is clear we must consider that any woman with a urogenital infection must have an effective treatment for the anal infection, since nearly 80% of women have an anal infection concomitant with the vaginal infection,” Dr. Bertille de Barbeyrac of the University of Bordeaux, France, told Reuters Health by email.
However, she noted that “even [though] the study shows that doxycycline is more effective than azithromycin on anal infection, other studies are needed to prove that residual anal infection after treatment with azithromycin can be a source of vaginal contamination and therefore justify changing practices and eliminating azithromycin as a treatment for lower urogenital chlamydial infection in women.”
“There are other reasons [to make] this change,” she added, “such as the acquisition of macrolide resistance by M. genitalium following heavy use of azithromycin.”
As reported in The Lancet Infectious Diseases, Dr. Barbeyrac and colleagues randomly assigned 460 women (median age, 21) to either doxycycline or azithromycin in a multicenter, open-label superiority trial.
Participants received either azithromycin (a single 1-g dose, with or without food) or doxycycline (100 mg in the morning and evening at mealtimes for 7 days – that is, 100 mg of doxycycline twice daily).
The primary outcome was that the microbiological anorectal cure rate, defined as a C. trachomatis-negative nucleic acid amplification test (NAAT), resulted in anorectal specimens six weeks after treatment initiation among women who had a baseline positive result (about half the women in each treatment group).
Ninety-four percent of the doxycycline group versus 85% of the azithromycin group had an anorectal cure (adjusted odds ratio with imputation of missing values, 0.43).
Adverse events possibly related to treatment occurred in 11% of the doxycycline group versus 13% of the azithromycin group. Gastrointestinal disorders were most frequent, occurring in 8% of the doxycycline and 11% of the azithromycin groups.
Summing up, the authors write, “The microbiological anorectal cure rate was significantly lower among women who received a single dose of azithromycin than among those who received a 1-week course of doxycycline. This finding suggests that doxycycline should be the first-line therapy for C trachomatis infection in women.”
Dr. Meleen Chuang, medical director of women’s health at the Family Health Centers at NYU Langone, Brooklyn, commented in an email to Reuters Health that after reviewing this study “as well as CDC and WHO recommendations updated as of 2022, health care providers should be treating C. trachomatis infections with doxycycline 100 mg twice a day for seven days as first-line therapy rather than azithromycin, [given] concerns of increasing macrolide drug resistance against Mycoplasma genitalium and Neisseria gonorrhea.”
“Our clinicians also see the growing uptick of syphilis, gonorrhea, and chlamydia infections in our population, similarly to the rest of the United States since 2020,” she noted. “With the increase in STD infection ... treatment with doxycycline therapy with an important caveat to the patient to complete the one-week treatment regimen is extremely important.”
Dr. Latasha Murphy of the Gynecologic Care Institute at Mercy, Baltimore, also commented in an email to Reuters Health. She noted, “this study does not mirror my clinical experience. More patients have side effects from doxycycline than azithromycin in my experience. Also, anorectal screening is not routine in STD screening.”
“If any major changes to clinical care are made,” she said, “it may be for more consistent screening for anorectal disease. This may ultimately lead to doxycycline being the first line-treatment. More research is needed before making any definitive changes.”
Reuters Health Information © 2022
NEW YORK (Reuters) – A one-week course of doxycycline was superior to a single dose of azithromycin in women with concurrent vaginal and anorectal chlamydia infection in an unblinded randomized controlled trial, mirroring previous results in men.
Researchers suggest that doxycycline should be the first-line therapy for chlamydia infection in women.
“It is clear we must consider that any woman with a urogenital infection must have an effective treatment for the anal infection, since nearly 80% of women have an anal infection concomitant with the vaginal infection,” Dr. Bertille de Barbeyrac of the University of Bordeaux, France, told Reuters Health by email.
However, she noted that “even [though] the study shows that doxycycline is more effective than azithromycin on anal infection, other studies are needed to prove that residual anal infection after treatment with azithromycin can be a source of vaginal contamination and therefore justify changing practices and eliminating azithromycin as a treatment for lower urogenital chlamydial infection in women.”
“There are other reasons [to make] this change,” she added, “such as the acquisition of macrolide resistance by M. genitalium following heavy use of azithromycin.”
As reported in The Lancet Infectious Diseases, Dr. Barbeyrac and colleagues randomly assigned 460 women (median age, 21) to either doxycycline or azithromycin in a multicenter, open-label superiority trial.
Participants received either azithromycin (a single 1-g dose, with or without food) or doxycycline (100 mg in the morning and evening at mealtimes for 7 days – that is, 100 mg of doxycycline twice daily).
The primary outcome was that the microbiological anorectal cure rate, defined as a C. trachomatis-negative nucleic acid amplification test (NAAT), resulted in anorectal specimens six weeks after treatment initiation among women who had a baseline positive result (about half the women in each treatment group).
Ninety-four percent of the doxycycline group versus 85% of the azithromycin group had an anorectal cure (adjusted odds ratio with imputation of missing values, 0.43).
Adverse events possibly related to treatment occurred in 11% of the doxycycline group versus 13% of the azithromycin group. Gastrointestinal disorders were most frequent, occurring in 8% of the doxycycline and 11% of the azithromycin groups.
Summing up, the authors write, “The microbiological anorectal cure rate was significantly lower among women who received a single dose of azithromycin than among those who received a 1-week course of doxycycline. This finding suggests that doxycycline should be the first-line therapy for C trachomatis infection in women.”
Dr. Meleen Chuang, medical director of women’s health at the Family Health Centers at NYU Langone, Brooklyn, commented in an email to Reuters Health that after reviewing this study “as well as CDC and WHO recommendations updated as of 2022, health care providers should be treating C. trachomatis infections with doxycycline 100 mg twice a day for seven days as first-line therapy rather than azithromycin, [given] concerns of increasing macrolide drug resistance against Mycoplasma genitalium and Neisseria gonorrhea.”
“Our clinicians also see the growing uptick of syphilis, gonorrhea, and chlamydia infections in our population, similarly to the rest of the United States since 2020,” she noted. “With the increase in STD infection ... treatment with doxycycline therapy with an important caveat to the patient to complete the one-week treatment regimen is extremely important.”
Dr. Latasha Murphy of the Gynecologic Care Institute at Mercy, Baltimore, also commented in an email to Reuters Health. She noted, “this study does not mirror my clinical experience. More patients have side effects from doxycycline than azithromycin in my experience. Also, anorectal screening is not routine in STD screening.”
“If any major changes to clinical care are made,” she said, “it may be for more consistent screening for anorectal disease. This may ultimately lead to doxycycline being the first line-treatment. More research is needed before making any definitive changes.”
Reuters Health Information © 2022




