User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
Updated recommendations released on COVID-19 and pediatric ALL
The main threat to the vast majority of children with acute lymphoblastic leukemia still remains the ALL itself, according to updated recommendations released by the Leukemia Committee of the French Society for the Fight Against Cancers and Leukemias in Children and Adolescents (SFCE).
“The situation of the current COVID-19 pandemic is continuously evolving. We thus have taken the more recent knowledge into account to update the previous recommendations from the Leukemia Committee,” Jérémie Rouger-Gaudichon, MD, of Pediatric Hemato-Immuno-Oncology Unit, Centre Hospitalier Universitaire, Caen (France), and colleagues wrote on behalf of the SFCE.
The updated recommendations are based on data collected in a real-time prospective survey among the 30 SFCE centers since April 2020. As of December 2020, 127 cases of COVID-19 were reported, most of them being enrolled in the PEDONCOVID study (NCT04433871) according to the report. Of these, eight patients required hospitalization in intensive care unit and one patient with relapsed acute lymphoblastic leukemia (ALL) died from ARDS with multiorgan failure. This confirms earlier reports that SARS-CoV-2 infection can be severe in some children with cancer and/or having hematopoietic stem cell transplant (HSCT), according to the report, which was published online in Bulletin du Cancer.
Recommendations
General recommendations were provided in the report, including the following:
- Test for SARS-CoV-2 (preferably by PCR or at least by immunological tests, on nasopharyngeal swab) before starting intensive induction chemotherapy or other intensive phase of treatment, for ALL patients, with or without symptoms.
- Delay systemic treatment if possible (e.g., absence of major hyperleukocytosis) in positive patients. During later phases, if patients test positive, tests should be repeated over time until negativity, especially before the beginning of an intensive course.
- Isolate any COVID-19–negative child or adolescent to allow treatment to continue (facial mask, social distancing, barrier measures, no contact with individuals suspected of COVID-19 or COVID-19–positive), in particular for patients to be allografted.
- Limit visitation to parents and potentially siblings in patients slated for HSCT and follow all necessary sanitary procedures for those visits.
The report provides a lengthy discussion of more detailed recommendations, including the following for first-line treatment of ALL:
- For patients with high-risk ALL, an individualized decision regarding transplantation and its timing should weigh the risks of transplantation in an epidemic context of COVID-19 against the risk linked to ALL.
- Minimizing hospital visits by the use of home blood tests and partial use of telemedicine may be considered.
- A physical examination should be performed regularly to avoid any delay in the diagnosis of treatment complications or relapse and preventative measures for SARS-CoV-2 should be applied in the home.
Patients with relapsed ALL may be at more risk from the effects of COVID-19 disease, according to the others, so for ALL patients receiving second-line or more treatment the recommendations include the following:
- Testing must be performed before starting a chemotherapy block, and postponing chemotherapy in case of positive test should be discussed in accordance with each specific situation and benefits/risks ratio regarding the leukemia.
- First-relapse patients should follow the INTREALL treatment protocol as much as possible and those who reach appropriate complete remission should be considered promptly for allogeneic transplantation, despite the pandemic.
- Second relapse and refractory relapses require testing and negative results for inclusion in phase I-II trials being conducted by most if not all academic or industrial promoters.
- The indication for treatment with CAR-T cells must be weighed with the center that would perform the procedure to determine the feasibility of performing all necessary procedures including apheresis and manufacturing.
In the case of a SARS-CoV-2 infection diagnosis during the treatment of ALL, discussions should occur with regard to stopping and/or postponing all chemotherapies, according to the severity of the ALL, the stage of treatment and the severity of clinical and/or radiological signs. In addition, any specific anti-COVID-19 treatment must be discussed with the infectious diseases team, according to the report.
“Fortunately, SARS-CoV-2 infection appears nevertheless to be mild in most children with cancer/ALL. Thus, the main threat to the vast majority of children with ALL still remains the ALL itself. Long-term data including well-matched case-control studies will tell if treatment delays/modifications due to COVID-19 have impacted the outcome if children with ALL,” the authors stated. However, “despite extremely rapid advances obtained in less than one year, our knowledge of SARS-CoV-2 and its complications is still incomplete,” they concluded, adding that the recommendations will likely need to be updated within another few months.
The authors reported that they had no conflicts of interest.
The main threat to the vast majority of children with acute lymphoblastic leukemia still remains the ALL itself, according to updated recommendations released by the Leukemia Committee of the French Society for the Fight Against Cancers and Leukemias in Children and Adolescents (SFCE).
“The situation of the current COVID-19 pandemic is continuously evolving. We thus have taken the more recent knowledge into account to update the previous recommendations from the Leukemia Committee,” Jérémie Rouger-Gaudichon, MD, of Pediatric Hemato-Immuno-Oncology Unit, Centre Hospitalier Universitaire, Caen (France), and colleagues wrote on behalf of the SFCE.
The updated recommendations are based on data collected in a real-time prospective survey among the 30 SFCE centers since April 2020. As of December 2020, 127 cases of COVID-19 were reported, most of them being enrolled in the PEDONCOVID study (NCT04433871) according to the report. Of these, eight patients required hospitalization in intensive care unit and one patient with relapsed acute lymphoblastic leukemia (ALL) died from ARDS with multiorgan failure. This confirms earlier reports that SARS-CoV-2 infection can be severe in some children with cancer and/or having hematopoietic stem cell transplant (HSCT), according to the report, which was published online in Bulletin du Cancer.
Recommendations
General recommendations were provided in the report, including the following:
- Test for SARS-CoV-2 (preferably by PCR or at least by immunological tests, on nasopharyngeal swab) before starting intensive induction chemotherapy or other intensive phase of treatment, for ALL patients, with or without symptoms.
- Delay systemic treatment if possible (e.g., absence of major hyperleukocytosis) in positive patients. During later phases, if patients test positive, tests should be repeated over time until negativity, especially before the beginning of an intensive course.
- Isolate any COVID-19–negative child or adolescent to allow treatment to continue (facial mask, social distancing, barrier measures, no contact with individuals suspected of COVID-19 or COVID-19–positive), in particular for patients to be allografted.
- Limit visitation to parents and potentially siblings in patients slated for HSCT and follow all necessary sanitary procedures for those visits.
The report provides a lengthy discussion of more detailed recommendations, including the following for first-line treatment of ALL:
- For patients with high-risk ALL, an individualized decision regarding transplantation and its timing should weigh the risks of transplantation in an epidemic context of COVID-19 against the risk linked to ALL.
- Minimizing hospital visits by the use of home blood tests and partial use of telemedicine may be considered.
- A physical examination should be performed regularly to avoid any delay in the diagnosis of treatment complications or relapse and preventative measures for SARS-CoV-2 should be applied in the home.
Patients with relapsed ALL may be at more risk from the effects of COVID-19 disease, according to the others, so for ALL patients receiving second-line or more treatment the recommendations include the following:
- Testing must be performed before starting a chemotherapy block, and postponing chemotherapy in case of positive test should be discussed in accordance with each specific situation and benefits/risks ratio regarding the leukemia.
- First-relapse patients should follow the INTREALL treatment protocol as much as possible and those who reach appropriate complete remission should be considered promptly for allogeneic transplantation, despite the pandemic.
- Second relapse and refractory relapses require testing and negative results for inclusion in phase I-II trials being conducted by most if not all academic or industrial promoters.
- The indication for treatment with CAR-T cells must be weighed with the center that would perform the procedure to determine the feasibility of performing all necessary procedures including apheresis and manufacturing.
In the case of a SARS-CoV-2 infection diagnosis during the treatment of ALL, discussions should occur with regard to stopping and/or postponing all chemotherapies, according to the severity of the ALL, the stage of treatment and the severity of clinical and/or radiological signs. In addition, any specific anti-COVID-19 treatment must be discussed with the infectious diseases team, according to the report.
“Fortunately, SARS-CoV-2 infection appears nevertheless to be mild in most children with cancer/ALL. Thus, the main threat to the vast majority of children with ALL still remains the ALL itself. Long-term data including well-matched case-control studies will tell if treatment delays/modifications due to COVID-19 have impacted the outcome if children with ALL,” the authors stated. However, “despite extremely rapid advances obtained in less than one year, our knowledge of SARS-CoV-2 and its complications is still incomplete,” they concluded, adding that the recommendations will likely need to be updated within another few months.
The authors reported that they had no conflicts of interest.
The main threat to the vast majority of children with acute lymphoblastic leukemia still remains the ALL itself, according to updated recommendations released by the Leukemia Committee of the French Society for the Fight Against Cancers and Leukemias in Children and Adolescents (SFCE).
“The situation of the current COVID-19 pandemic is continuously evolving. We thus have taken the more recent knowledge into account to update the previous recommendations from the Leukemia Committee,” Jérémie Rouger-Gaudichon, MD, of Pediatric Hemato-Immuno-Oncology Unit, Centre Hospitalier Universitaire, Caen (France), and colleagues wrote on behalf of the SFCE.
The updated recommendations are based on data collected in a real-time prospective survey among the 30 SFCE centers since April 2020. As of December 2020, 127 cases of COVID-19 were reported, most of them being enrolled in the PEDONCOVID study (NCT04433871) according to the report. Of these, eight patients required hospitalization in intensive care unit and one patient with relapsed acute lymphoblastic leukemia (ALL) died from ARDS with multiorgan failure. This confirms earlier reports that SARS-CoV-2 infection can be severe in some children with cancer and/or having hematopoietic stem cell transplant (HSCT), according to the report, which was published online in Bulletin du Cancer.
Recommendations
General recommendations were provided in the report, including the following:
- Test for SARS-CoV-2 (preferably by PCR or at least by immunological tests, on nasopharyngeal swab) before starting intensive induction chemotherapy or other intensive phase of treatment, for ALL patients, with or without symptoms.
- Delay systemic treatment if possible (e.g., absence of major hyperleukocytosis) in positive patients. During later phases, if patients test positive, tests should be repeated over time until negativity, especially before the beginning of an intensive course.
- Isolate any COVID-19–negative child or adolescent to allow treatment to continue (facial mask, social distancing, barrier measures, no contact with individuals suspected of COVID-19 or COVID-19–positive), in particular for patients to be allografted.
- Limit visitation to parents and potentially siblings in patients slated for HSCT and follow all necessary sanitary procedures for those visits.
The report provides a lengthy discussion of more detailed recommendations, including the following for first-line treatment of ALL:
- For patients with high-risk ALL, an individualized decision regarding transplantation and its timing should weigh the risks of transplantation in an epidemic context of COVID-19 against the risk linked to ALL.
- Minimizing hospital visits by the use of home blood tests and partial use of telemedicine may be considered.
- A physical examination should be performed regularly to avoid any delay in the diagnosis of treatment complications or relapse and preventative measures for SARS-CoV-2 should be applied in the home.
Patients with relapsed ALL may be at more risk from the effects of COVID-19 disease, according to the others, so for ALL patients receiving second-line or more treatment the recommendations include the following:
- Testing must be performed before starting a chemotherapy block, and postponing chemotherapy in case of positive test should be discussed in accordance with each specific situation and benefits/risks ratio regarding the leukemia.
- First-relapse patients should follow the INTREALL treatment protocol as much as possible and those who reach appropriate complete remission should be considered promptly for allogeneic transplantation, despite the pandemic.
- Second relapse and refractory relapses require testing and negative results for inclusion in phase I-II trials being conducted by most if not all academic or industrial promoters.
- The indication for treatment with CAR-T cells must be weighed with the center that would perform the procedure to determine the feasibility of performing all necessary procedures including apheresis and manufacturing.
In the case of a SARS-CoV-2 infection diagnosis during the treatment of ALL, discussions should occur with regard to stopping and/or postponing all chemotherapies, according to the severity of the ALL, the stage of treatment and the severity of clinical and/or radiological signs. In addition, any specific anti-COVID-19 treatment must be discussed with the infectious diseases team, according to the report.
“Fortunately, SARS-CoV-2 infection appears nevertheless to be mild in most children with cancer/ALL. Thus, the main threat to the vast majority of children with ALL still remains the ALL itself. Long-term data including well-matched case-control studies will tell if treatment delays/modifications due to COVID-19 have impacted the outcome if children with ALL,” the authors stated. However, “despite extremely rapid advances obtained in less than one year, our knowledge of SARS-CoV-2 and its complications is still incomplete,” they concluded, adding that the recommendations will likely need to be updated within another few months.
The authors reported that they had no conflicts of interest.
FROM BULLETIN DU CANCER
Some with long COVID see relief after vaccination
Several weeks after getting his second dose of an mRNA vaccine, Aaron Goyang thinks his long bout with COVID-19 has finally come to an end.
Mr. Goyang, who is 33 and is a radiology technician in Austin, Tex., thinks he got COVID-19 from some of the coughing, gasping patients he treated last spring.
At the time, testing was scarce, and by the time he was tested – several weeks into his illness – it came back negative. He fought off the initial symptoms but experienced relapse a week later.
Mr. Goyang says that, for the next 8 or 9 months, he was on a roller coaster with extreme shortness of breath and chest tightness that could be so severe it would send him to the emergency department. He had to use an inhaler to get through his workdays.
“Even if I was just sitting around, it would come and take me,” he says. “It almost felt like someone was bear-hugging me constantly, and I just couldn’t get in a good enough breath.”
On his best days, he would walk around his neighborhood, being careful not to overdo it. He tried running once, and it nearly sent him to the hospital.
“Very honestly, I didn’t know if I would ever be able to do it again,” he says.
But Mr. Goyang says that, several weeks after getting the Pfizer vaccine, he was able to run a mile again with no problems. “I was very thankful for that,” he says.
Mr. Goyang is not alone. Some social media groups are dedicated to patients who are living with a condition that’s been known as long COVID and that was recently termed postacute sequelae of SARS-CoV-2 infection (PASC). These patients are sometimes referred to as long haulers.
On social media, patients with PASC are eagerly and anxiously quizzing each other about the vaccines and their effects.
Survivor Corps, which has a public Facebook group with 159,000 members, recently took a poll to see whether there was any substance to rumors that those with long COVID were feeling better after being vaccinated.
“Out of 400 people, 36% showed an improvement in symptoms, anywhere between a mild improvement to complete resolution of symptoms,” said Diana Berrent, a long-COVID patient who founded the group. Survivor Corps has become active in patient advocacy and is a resource for researchers studying the new condition.
Ms. Berrent has become such a trusted voice during the pandemic. She interviewed Anthony Fauci, MD, head of the National Institutes of Allergy and Infectious Diseases, last October.
“The implications are huge,” she says.
“Some of this damage is permanent damage. It’s not going to cure the scarring of your heart tissue, it’s not going to cure the irreparable damage to your lungs, but if it’s making people feel better, then that’s an indication there’s viral persistence going on,” says Ms. Berrent.
“I’ve been saying for months and months, we shouldn’t be calling this postacute anything,” she adds.
Patients report improvement
Daniel Griffin, MD, PhD, is equally excited. He’s an infectious disease specialist at Columbia University, New York. He says about one in five patients he treated for COVID-19 last year never got better. Many of them, such as Mr. Goyang, were health care workers.
“I don’t know if people actually catch this, but a lot of our coworkers are either permanently disabled or died,” Dr. Griffin says.
Health care workers were also among the first to be vaccinated. Dr. Griffin says many of his patients began reaching out to him about a week or two after being vaccinated “and saying, ‘You know, I actually feel better.’ And some of them were saying, ‘I feel all better,’ after being sick – a lot of them – for a year.”
Then he was getting calls and texts from other doctors, asking, “Hey, are you seeing this?”
The benefits of vaccination for some long-haulers came as a surprise. Dr. Griffin says that, before the vaccines came out, many of his patients were worried that getting vaccinated might overstimulate their immune systems and cause symptoms to get worse.
Indeed, a small percentage of people – about 3%-5%, based on informal polls on social media – report that they do experience worsening of symptoms after getting the shot. It’s not clear why.
Dr. Griffin estimates that between 30% and 50% of patients’ symptoms improve after they receive the mRNA vaccines. “I’m seeing this chunk of people – they tell me their brain fog has improved, their fatigue is gone, the fevers that wouldn’t resolve have now gone,” he says. “I’m seeing that personally, and I’m hearing it from my colleagues.”
Dr. Griffin says the observation has launched several studies and that there are several theories about how the vaccines might be affecting long COVID.
An immune system boost?
One possibility is that the virus continues to stimulate the immune system, which continues to fight the virus for months. If that is the case, Dr. Griffin says, the vaccine may be giving the immune system the boost it needs to finally clear the virus away.
Donna Farber, PhD, a professor of microbiology and immunology at Columbia University, has heard the stories, too.
“It is possible that the persisting virus in long COVID-19 may be at a low level – not enough to stimulate a potent immune response to clear the virus, but enough to cause symptoms. Activating the immune response therefore is therapeutic in directing viral clearance,” she says.
Dr. Farber explains that long COVID may be a bit like Lyme disease. Some patients with Lyme disease must take antibiotics for months before their symptoms disappear.
Dr. Griffin says there’s another possibility. Several studies have now shown that people with lingering COVID-19 symptoms develop autoantibodies. There’s a theory that SARS-CoV-2 may create an autoimmune condition that leads to long-term symptoms.
If that is the case, Dr. Griffin says, the vaccine may be helping the body to reset its tolerance to itself, “so maybe now you’re getting a healthy immune response.”
More studies are needed to know for sure.
Either way, the vaccines are a much-needed bit of hope for the long-COVID community, and Dr. Griffin tells his patients who are still worried that, at the very least, they’ll be protected from another SARS-CoV-2 infection.
A version of this article first appeared on Medscape.com.
Several weeks after getting his second dose of an mRNA vaccine, Aaron Goyang thinks his long bout with COVID-19 has finally come to an end.
Mr. Goyang, who is 33 and is a radiology technician in Austin, Tex., thinks he got COVID-19 from some of the coughing, gasping patients he treated last spring.
At the time, testing was scarce, and by the time he was tested – several weeks into his illness – it came back negative. He fought off the initial symptoms but experienced relapse a week later.
Mr. Goyang says that, for the next 8 or 9 months, he was on a roller coaster with extreme shortness of breath and chest tightness that could be so severe it would send him to the emergency department. He had to use an inhaler to get through his workdays.
“Even if I was just sitting around, it would come and take me,” he says. “It almost felt like someone was bear-hugging me constantly, and I just couldn’t get in a good enough breath.”
On his best days, he would walk around his neighborhood, being careful not to overdo it. He tried running once, and it nearly sent him to the hospital.
“Very honestly, I didn’t know if I would ever be able to do it again,” he says.
But Mr. Goyang says that, several weeks after getting the Pfizer vaccine, he was able to run a mile again with no problems. “I was very thankful for that,” he says.
Mr. Goyang is not alone. Some social media groups are dedicated to patients who are living with a condition that’s been known as long COVID and that was recently termed postacute sequelae of SARS-CoV-2 infection (PASC). These patients are sometimes referred to as long haulers.
On social media, patients with PASC are eagerly and anxiously quizzing each other about the vaccines and their effects.
Survivor Corps, which has a public Facebook group with 159,000 members, recently took a poll to see whether there was any substance to rumors that those with long COVID were feeling better after being vaccinated.
“Out of 400 people, 36% showed an improvement in symptoms, anywhere between a mild improvement to complete resolution of symptoms,” said Diana Berrent, a long-COVID patient who founded the group. Survivor Corps has become active in patient advocacy and is a resource for researchers studying the new condition.
Ms. Berrent has become such a trusted voice during the pandemic. She interviewed Anthony Fauci, MD, head of the National Institutes of Allergy and Infectious Diseases, last October.
“The implications are huge,” she says.
“Some of this damage is permanent damage. It’s not going to cure the scarring of your heart tissue, it’s not going to cure the irreparable damage to your lungs, but if it’s making people feel better, then that’s an indication there’s viral persistence going on,” says Ms. Berrent.
“I’ve been saying for months and months, we shouldn’t be calling this postacute anything,” she adds.
Patients report improvement
Daniel Griffin, MD, PhD, is equally excited. He’s an infectious disease specialist at Columbia University, New York. He says about one in five patients he treated for COVID-19 last year never got better. Many of them, such as Mr. Goyang, were health care workers.
“I don’t know if people actually catch this, but a lot of our coworkers are either permanently disabled or died,” Dr. Griffin says.
Health care workers were also among the first to be vaccinated. Dr. Griffin says many of his patients began reaching out to him about a week or two after being vaccinated “and saying, ‘You know, I actually feel better.’ And some of them were saying, ‘I feel all better,’ after being sick – a lot of them – for a year.”
Then he was getting calls and texts from other doctors, asking, “Hey, are you seeing this?”
The benefits of vaccination for some long-haulers came as a surprise. Dr. Griffin says that, before the vaccines came out, many of his patients were worried that getting vaccinated might overstimulate their immune systems and cause symptoms to get worse.
Indeed, a small percentage of people – about 3%-5%, based on informal polls on social media – report that they do experience worsening of symptoms after getting the shot. It’s not clear why.
Dr. Griffin estimates that between 30% and 50% of patients’ symptoms improve after they receive the mRNA vaccines. “I’m seeing this chunk of people – they tell me their brain fog has improved, their fatigue is gone, the fevers that wouldn’t resolve have now gone,” he says. “I’m seeing that personally, and I’m hearing it from my colleagues.”
Dr. Griffin says the observation has launched several studies and that there are several theories about how the vaccines might be affecting long COVID.
An immune system boost?
One possibility is that the virus continues to stimulate the immune system, which continues to fight the virus for months. If that is the case, Dr. Griffin says, the vaccine may be giving the immune system the boost it needs to finally clear the virus away.
Donna Farber, PhD, a professor of microbiology and immunology at Columbia University, has heard the stories, too.
“It is possible that the persisting virus in long COVID-19 may be at a low level – not enough to stimulate a potent immune response to clear the virus, but enough to cause symptoms. Activating the immune response therefore is therapeutic in directing viral clearance,” she says.
Dr. Farber explains that long COVID may be a bit like Lyme disease. Some patients with Lyme disease must take antibiotics for months before their symptoms disappear.
Dr. Griffin says there’s another possibility. Several studies have now shown that people with lingering COVID-19 symptoms develop autoantibodies. There’s a theory that SARS-CoV-2 may create an autoimmune condition that leads to long-term symptoms.
If that is the case, Dr. Griffin says, the vaccine may be helping the body to reset its tolerance to itself, “so maybe now you’re getting a healthy immune response.”
More studies are needed to know for sure.
Either way, the vaccines are a much-needed bit of hope for the long-COVID community, and Dr. Griffin tells his patients who are still worried that, at the very least, they’ll be protected from another SARS-CoV-2 infection.
A version of this article first appeared on Medscape.com.
Several weeks after getting his second dose of an mRNA vaccine, Aaron Goyang thinks his long bout with COVID-19 has finally come to an end.
Mr. Goyang, who is 33 and is a radiology technician in Austin, Tex., thinks he got COVID-19 from some of the coughing, gasping patients he treated last spring.
At the time, testing was scarce, and by the time he was tested – several weeks into his illness – it came back negative. He fought off the initial symptoms but experienced relapse a week later.
Mr. Goyang says that, for the next 8 or 9 months, he was on a roller coaster with extreme shortness of breath and chest tightness that could be so severe it would send him to the emergency department. He had to use an inhaler to get through his workdays.
“Even if I was just sitting around, it would come and take me,” he says. “It almost felt like someone was bear-hugging me constantly, and I just couldn’t get in a good enough breath.”
On his best days, he would walk around his neighborhood, being careful not to overdo it. He tried running once, and it nearly sent him to the hospital.
“Very honestly, I didn’t know if I would ever be able to do it again,” he says.
But Mr. Goyang says that, several weeks after getting the Pfizer vaccine, he was able to run a mile again with no problems. “I was very thankful for that,” he says.
Mr. Goyang is not alone. Some social media groups are dedicated to patients who are living with a condition that’s been known as long COVID and that was recently termed postacute sequelae of SARS-CoV-2 infection (PASC). These patients are sometimes referred to as long haulers.
On social media, patients with PASC are eagerly and anxiously quizzing each other about the vaccines and their effects.
Survivor Corps, which has a public Facebook group with 159,000 members, recently took a poll to see whether there was any substance to rumors that those with long COVID were feeling better after being vaccinated.
“Out of 400 people, 36% showed an improvement in symptoms, anywhere between a mild improvement to complete resolution of symptoms,” said Diana Berrent, a long-COVID patient who founded the group. Survivor Corps has become active in patient advocacy and is a resource for researchers studying the new condition.
Ms. Berrent has become such a trusted voice during the pandemic. She interviewed Anthony Fauci, MD, head of the National Institutes of Allergy and Infectious Diseases, last October.
“The implications are huge,” she says.
“Some of this damage is permanent damage. It’s not going to cure the scarring of your heart tissue, it’s not going to cure the irreparable damage to your lungs, but if it’s making people feel better, then that’s an indication there’s viral persistence going on,” says Ms. Berrent.
“I’ve been saying for months and months, we shouldn’t be calling this postacute anything,” she adds.
Patients report improvement
Daniel Griffin, MD, PhD, is equally excited. He’s an infectious disease specialist at Columbia University, New York. He says about one in five patients he treated for COVID-19 last year never got better. Many of them, such as Mr. Goyang, were health care workers.
“I don’t know if people actually catch this, but a lot of our coworkers are either permanently disabled or died,” Dr. Griffin says.
Health care workers were also among the first to be vaccinated. Dr. Griffin says many of his patients began reaching out to him about a week or two after being vaccinated “and saying, ‘You know, I actually feel better.’ And some of them were saying, ‘I feel all better,’ after being sick – a lot of them – for a year.”
Then he was getting calls and texts from other doctors, asking, “Hey, are you seeing this?”
The benefits of vaccination for some long-haulers came as a surprise. Dr. Griffin says that, before the vaccines came out, many of his patients were worried that getting vaccinated might overstimulate their immune systems and cause symptoms to get worse.
Indeed, a small percentage of people – about 3%-5%, based on informal polls on social media – report that they do experience worsening of symptoms after getting the shot. It’s not clear why.
Dr. Griffin estimates that between 30% and 50% of patients’ symptoms improve after they receive the mRNA vaccines. “I’m seeing this chunk of people – they tell me their brain fog has improved, their fatigue is gone, the fevers that wouldn’t resolve have now gone,” he says. “I’m seeing that personally, and I’m hearing it from my colleagues.”
Dr. Griffin says the observation has launched several studies and that there are several theories about how the vaccines might be affecting long COVID.
An immune system boost?
One possibility is that the virus continues to stimulate the immune system, which continues to fight the virus for months. If that is the case, Dr. Griffin says, the vaccine may be giving the immune system the boost it needs to finally clear the virus away.
Donna Farber, PhD, a professor of microbiology and immunology at Columbia University, has heard the stories, too.
“It is possible that the persisting virus in long COVID-19 may be at a low level – not enough to stimulate a potent immune response to clear the virus, but enough to cause symptoms. Activating the immune response therefore is therapeutic in directing viral clearance,” she says.
Dr. Farber explains that long COVID may be a bit like Lyme disease. Some patients with Lyme disease must take antibiotics for months before their symptoms disappear.
Dr. Griffin says there’s another possibility. Several studies have now shown that people with lingering COVID-19 symptoms develop autoantibodies. There’s a theory that SARS-CoV-2 may create an autoimmune condition that leads to long-term symptoms.
If that is the case, Dr. Griffin says, the vaccine may be helping the body to reset its tolerance to itself, “so maybe now you’re getting a healthy immune response.”
More studies are needed to know for sure.
Either way, the vaccines are a much-needed bit of hope for the long-COVID community, and Dr. Griffin tells his patients who are still worried that, at the very least, they’ll be protected from another SARS-CoV-2 infection.
A version of this article first appeared on Medscape.com.
We’re all vaccinated: Can we go back to the office (unmasked) now?
Congratulations, you’ve been vaccinated!
It’s been a year like no other, and outpatient psychiatrists turned to Zoom and other telemental health platforms to provide treatment for our patients. Offices sit empty as the dust lands and the plants wilt. Perhaps a few patients are seen in person, masked and carefully distanced, after health screening and temperature checks, with surfaces sanitized between visits, all in accordance with health department regulations. But now the vaccine offers both safety and the promise of a return to a new normal, one that is certain to look different from the normal that was left behind.
I have been vaccinated and many of my patients have also been vaccinated. I began to wonder if it was safe to start seeing patients in person; could I see fully vaccinated patients, unmasked and without temperature checks and sanitizing? I started asking this question in February, and the response I got then was that it was too soon to tell; we did not have any data on whether vaccinated people could transmit the novel coronavirus. Two vaccinated people might be at risk of transmitting the virus and then infecting others, and the question of whether the vaccines would protect against illness caused by variants remained. Preliminary data out of Israel indicated that the vaccine did reduce transmission, but no one was saying that it was fine to see patients without masks, and video-conferencing remained the safest option.
On Monday, March 8, 2021, the Centers for Disease Control and Prevention released long-awaited interim public health guidelines for fully vaccinated people. The guidelines allowed for two vaccinated people to be in a room together unmasked, and for a fully-vaccinated person to be in a room unmasked with an unvaccinated person who did not have risk factors for becoming severely ill with COVID. Was this the green light that psychiatrists were waiting for? Was there new data about transmission, or was this part of the CDC’s effort to make vaccines more desirable?
Michael Chang, MD, is a pediatric infectious disease specialist at the University of Texas Health Science Center at Houston. We spoke 2 days after the CDC interim guidelines were released. Dr. Chang was optimistic.
“, including data about variants and about transmission. At some point, however, the risk is low enough, and we should probably start thinking about going back to in-person visits,” Dr. Chang said. He said he personally would feel safe meeting unmasked with a vaccinated patient, but noted that his institution still requires doctors to wear masks. “Most vaccinations reduce transmission of illness,” Dr. Chang said, “but SARS-CoV-2 continues to surprise us in many ways.”
Katelyn Jetelina, PhD, MPH, an epidemiologist at the University of Texas School of Public Health in Dallas, distributes a newsletter, “Your Local Epidemiologist,” where she discusses data pertaining to the pandemic. In her newsletter dated March 14, 2021, Dr. Jetelina wrote, “There are now 7 sub-studies/press releases that confirm a 50-95% reduced transmission after vaccination. This is a big range, which is typical for such drastically different scientific studies. Variability is likely due to different sample sizes, locations, vaccines, genetics, cultures, etc. It will be a while until we know the ‘true’ percentage for each vaccine.”
Leslie Walker, MD, is a fully vaccinated psychiatrist in private practice in Shaker Heights, Ohio. She has recently started seeing fully vaccinated patients in person.
“So far it’s only 1 or 2 patients a day. I’m leaving it up to the patient. If they prefer masks, we stay masked. I may reverse course, depending on what information comes out.” She went on to note, “There are benefits to being able to see someone’s full facial expressions and whether they match someone’s words and body language, so the benefit of “unmasking” extends beyond comfort and convenience and must be balanced against the theoretical risk of COVID exposure in the room.”
While the CDC has now said it is safe to meet, the state health departments also have guidelines for medical practices, and everyone is still worried about vulnerable people in their households and potential spread to the community at large.
In Maryland, where I work, Aliya Jones, MD, MBA, is the head of the Behavioral Health Administration (BHA) for the Maryland Department of Health. “It remains risky to not wear masks, however, the risk is low when both individuals are vaccinated,” Dr. Jones wrote. “BHA is not recommending that providers see clients without both parties wearing a mask. All of our general practice recommendations for infection control are unchanged. People should be screened before entering clinical practices and persons who are symptomatic, whether vaccinated or not, should not be seen face-to-face, except in cases of an emergency, in which case additional precautions should be taken.”
So is it safe for a fully-vaccinated psychiatrist to have a session with a fully-vaccinated patient sitting 8 feet apart without masks? I’m left with the idea that it is for those two people, but when it comes to unvaccinated people in their households, we want more certainty than we currently have. The messaging remains unclear. The CDC’s interim guidelines offer hope for a future, but the science is still catching up, and to feel safe enough, we may want to wait a little longer for more definitive data – or herd immunity – before we reveal our smiles.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
Congratulations, you’ve been vaccinated!
It’s been a year like no other, and outpatient psychiatrists turned to Zoom and other telemental health platforms to provide treatment for our patients. Offices sit empty as the dust lands and the plants wilt. Perhaps a few patients are seen in person, masked and carefully distanced, after health screening and temperature checks, with surfaces sanitized between visits, all in accordance with health department regulations. But now the vaccine offers both safety and the promise of a return to a new normal, one that is certain to look different from the normal that was left behind.
I have been vaccinated and many of my patients have also been vaccinated. I began to wonder if it was safe to start seeing patients in person; could I see fully vaccinated patients, unmasked and without temperature checks and sanitizing? I started asking this question in February, and the response I got then was that it was too soon to tell; we did not have any data on whether vaccinated people could transmit the novel coronavirus. Two vaccinated people might be at risk of transmitting the virus and then infecting others, and the question of whether the vaccines would protect against illness caused by variants remained. Preliminary data out of Israel indicated that the vaccine did reduce transmission, but no one was saying that it was fine to see patients without masks, and video-conferencing remained the safest option.
On Monday, March 8, 2021, the Centers for Disease Control and Prevention released long-awaited interim public health guidelines for fully vaccinated people. The guidelines allowed for two vaccinated people to be in a room together unmasked, and for a fully-vaccinated person to be in a room unmasked with an unvaccinated person who did not have risk factors for becoming severely ill with COVID. Was this the green light that psychiatrists were waiting for? Was there new data about transmission, or was this part of the CDC’s effort to make vaccines more desirable?
Michael Chang, MD, is a pediatric infectious disease specialist at the University of Texas Health Science Center at Houston. We spoke 2 days after the CDC interim guidelines were released. Dr. Chang was optimistic.
“, including data about variants and about transmission. At some point, however, the risk is low enough, and we should probably start thinking about going back to in-person visits,” Dr. Chang said. He said he personally would feel safe meeting unmasked with a vaccinated patient, but noted that his institution still requires doctors to wear masks. “Most vaccinations reduce transmission of illness,” Dr. Chang said, “but SARS-CoV-2 continues to surprise us in many ways.”
Katelyn Jetelina, PhD, MPH, an epidemiologist at the University of Texas School of Public Health in Dallas, distributes a newsletter, “Your Local Epidemiologist,” where she discusses data pertaining to the pandemic. In her newsletter dated March 14, 2021, Dr. Jetelina wrote, “There are now 7 sub-studies/press releases that confirm a 50-95% reduced transmission after vaccination. This is a big range, which is typical for such drastically different scientific studies. Variability is likely due to different sample sizes, locations, vaccines, genetics, cultures, etc. It will be a while until we know the ‘true’ percentage for each vaccine.”
Leslie Walker, MD, is a fully vaccinated psychiatrist in private practice in Shaker Heights, Ohio. She has recently started seeing fully vaccinated patients in person.
“So far it’s only 1 or 2 patients a day. I’m leaving it up to the patient. If they prefer masks, we stay masked. I may reverse course, depending on what information comes out.” She went on to note, “There are benefits to being able to see someone’s full facial expressions and whether they match someone’s words and body language, so the benefit of “unmasking” extends beyond comfort and convenience and must be balanced against the theoretical risk of COVID exposure in the room.”
While the CDC has now said it is safe to meet, the state health departments also have guidelines for medical practices, and everyone is still worried about vulnerable people in their households and potential spread to the community at large.
In Maryland, where I work, Aliya Jones, MD, MBA, is the head of the Behavioral Health Administration (BHA) for the Maryland Department of Health. “It remains risky to not wear masks, however, the risk is low when both individuals are vaccinated,” Dr. Jones wrote. “BHA is not recommending that providers see clients without both parties wearing a mask. All of our general practice recommendations for infection control are unchanged. People should be screened before entering clinical practices and persons who are symptomatic, whether vaccinated or not, should not be seen face-to-face, except in cases of an emergency, in which case additional precautions should be taken.”
So is it safe for a fully-vaccinated psychiatrist to have a session with a fully-vaccinated patient sitting 8 feet apart without masks? I’m left with the idea that it is for those two people, but when it comes to unvaccinated people in their households, we want more certainty than we currently have. The messaging remains unclear. The CDC’s interim guidelines offer hope for a future, but the science is still catching up, and to feel safe enough, we may want to wait a little longer for more definitive data – or herd immunity – before we reveal our smiles.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
Congratulations, you’ve been vaccinated!
It’s been a year like no other, and outpatient psychiatrists turned to Zoom and other telemental health platforms to provide treatment for our patients. Offices sit empty as the dust lands and the plants wilt. Perhaps a few patients are seen in person, masked and carefully distanced, after health screening and temperature checks, with surfaces sanitized between visits, all in accordance with health department regulations. But now the vaccine offers both safety and the promise of a return to a new normal, one that is certain to look different from the normal that was left behind.
I have been vaccinated and many of my patients have also been vaccinated. I began to wonder if it was safe to start seeing patients in person; could I see fully vaccinated patients, unmasked and without temperature checks and sanitizing? I started asking this question in February, and the response I got then was that it was too soon to tell; we did not have any data on whether vaccinated people could transmit the novel coronavirus. Two vaccinated people might be at risk of transmitting the virus and then infecting others, and the question of whether the vaccines would protect against illness caused by variants remained. Preliminary data out of Israel indicated that the vaccine did reduce transmission, but no one was saying that it was fine to see patients without masks, and video-conferencing remained the safest option.
On Monday, March 8, 2021, the Centers for Disease Control and Prevention released long-awaited interim public health guidelines for fully vaccinated people. The guidelines allowed for two vaccinated people to be in a room together unmasked, and for a fully-vaccinated person to be in a room unmasked with an unvaccinated person who did not have risk factors for becoming severely ill with COVID. Was this the green light that psychiatrists were waiting for? Was there new data about transmission, or was this part of the CDC’s effort to make vaccines more desirable?
Michael Chang, MD, is a pediatric infectious disease specialist at the University of Texas Health Science Center at Houston. We spoke 2 days after the CDC interim guidelines were released. Dr. Chang was optimistic.
“, including data about variants and about transmission. At some point, however, the risk is low enough, and we should probably start thinking about going back to in-person visits,” Dr. Chang said. He said he personally would feel safe meeting unmasked with a vaccinated patient, but noted that his institution still requires doctors to wear masks. “Most vaccinations reduce transmission of illness,” Dr. Chang said, “but SARS-CoV-2 continues to surprise us in many ways.”
Katelyn Jetelina, PhD, MPH, an epidemiologist at the University of Texas School of Public Health in Dallas, distributes a newsletter, “Your Local Epidemiologist,” where she discusses data pertaining to the pandemic. In her newsletter dated March 14, 2021, Dr. Jetelina wrote, “There are now 7 sub-studies/press releases that confirm a 50-95% reduced transmission after vaccination. This is a big range, which is typical for such drastically different scientific studies. Variability is likely due to different sample sizes, locations, vaccines, genetics, cultures, etc. It will be a while until we know the ‘true’ percentage for each vaccine.”
Leslie Walker, MD, is a fully vaccinated psychiatrist in private practice in Shaker Heights, Ohio. She has recently started seeing fully vaccinated patients in person.
“So far it’s only 1 or 2 patients a day. I’m leaving it up to the patient. If they prefer masks, we stay masked. I may reverse course, depending on what information comes out.” She went on to note, “There are benefits to being able to see someone’s full facial expressions and whether they match someone’s words and body language, so the benefit of “unmasking” extends beyond comfort and convenience and must be balanced against the theoretical risk of COVID exposure in the room.”
While the CDC has now said it is safe to meet, the state health departments also have guidelines for medical practices, and everyone is still worried about vulnerable people in their households and potential spread to the community at large.
In Maryland, where I work, Aliya Jones, MD, MBA, is the head of the Behavioral Health Administration (BHA) for the Maryland Department of Health. “It remains risky to not wear masks, however, the risk is low when both individuals are vaccinated,” Dr. Jones wrote. “BHA is not recommending that providers see clients without both parties wearing a mask. All of our general practice recommendations for infection control are unchanged. People should be screened before entering clinical practices and persons who are symptomatic, whether vaccinated or not, should not be seen face-to-face, except in cases of an emergency, in which case additional precautions should be taken.”
So is it safe for a fully-vaccinated psychiatrist to have a session with a fully-vaccinated patient sitting 8 feet apart without masks? I’m left with the idea that it is for those two people, but when it comes to unvaccinated people in their households, we want more certainty than we currently have. The messaging remains unclear. The CDC’s interim guidelines offer hope for a future, but the science is still catching up, and to feel safe enough, we may want to wait a little longer for more definitive data – or herd immunity – before we reveal our smiles.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins, both in Baltimore.
Decline in child COVID-19 cases picks up after 2-week slowdown
, according to data gathered by the American Academy of Pediatrics and the Children’s Hospital Association.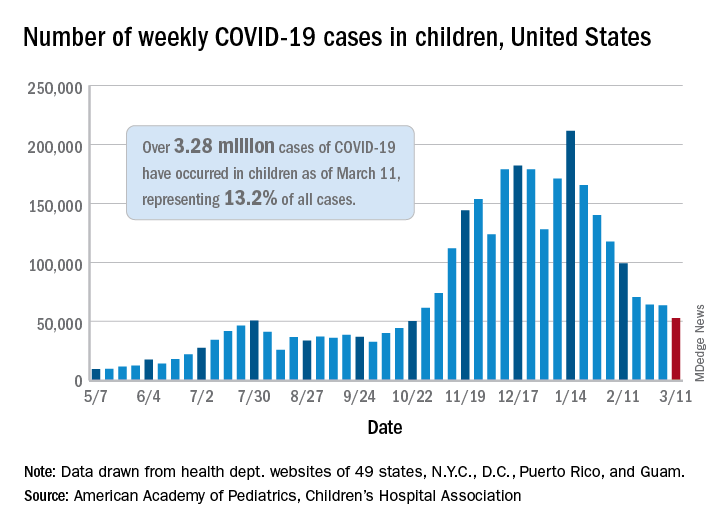
From Feb. 19 to March 4, the drop in new cases averaged just 5% each week, compared with 13.3% per week over the 5-week period from Jan. 15 to Feb. 18. For the week of March 5-11, a total of 52,695 COVID-19 cases were reported in children, down from 63,562 the previous week and the lowest number since late October, based on data from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
In those jurisdictions, 3.28 million children have been infected with SARS-CoV-2, representing 13.2% of all cases since the beginning of the pandemic. The cumulative rate of COVID-19 has now risen to 4,364 cases per 100,000 children nationally, with state rates ranging from 1,062 per 100,000 in Hawaii to 8,692 per 100,000 in North Dakota, the AAP and CHA said in their weekly COVID-19 report.
Hospitalization data are more limited – 24 states and New York City – but continue to show that serious illness is much less common in younger individuals: Children represent just 1.9% of all hospitalizations, and only 0.8% of the children who have been infected were hospitalized. Neither rate has changed since early February, the AAP and CHA said.
The number of deaths in children, however, rose from 253 to 266, the largest 1-week increase since early February in the 43 states (along with New York City, Puerto Rico, and Guam) that are tracking mortality data by age, the AAP and CHA reported.
Among those 46 jurisdictions, there are 10 (9 states and the District of Columbia) that have not yet reported a COVID-19–related child death, while Texas has almost twice as many deaths, 47, as the next state, Arizona, which has 24. Meanwhile, California’s total of 452,000 cases is almost 2½ times higher than the 183,000 recorded by Illinois, according to the report.
, according to data gathered by the American Academy of Pediatrics and the Children’s Hospital Association.
From Feb. 19 to March 4, the drop in new cases averaged just 5% each week, compared with 13.3% per week over the 5-week period from Jan. 15 to Feb. 18. For the week of March 5-11, a total of 52,695 COVID-19 cases were reported in children, down from 63,562 the previous week and the lowest number since late October, based on data from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
In those jurisdictions, 3.28 million children have been infected with SARS-CoV-2, representing 13.2% of all cases since the beginning of the pandemic. The cumulative rate of COVID-19 has now risen to 4,364 cases per 100,000 children nationally, with state rates ranging from 1,062 per 100,000 in Hawaii to 8,692 per 100,000 in North Dakota, the AAP and CHA said in their weekly COVID-19 report.
Hospitalization data are more limited – 24 states and New York City – but continue to show that serious illness is much less common in younger individuals: Children represent just 1.9% of all hospitalizations, and only 0.8% of the children who have been infected were hospitalized. Neither rate has changed since early February, the AAP and CHA said.
The number of deaths in children, however, rose from 253 to 266, the largest 1-week increase since early February in the 43 states (along with New York City, Puerto Rico, and Guam) that are tracking mortality data by age, the AAP and CHA reported.
Among those 46 jurisdictions, there are 10 (9 states and the District of Columbia) that have not yet reported a COVID-19–related child death, while Texas has almost twice as many deaths, 47, as the next state, Arizona, which has 24. Meanwhile, California’s total of 452,000 cases is almost 2½ times higher than the 183,000 recorded by Illinois, according to the report.
, according to data gathered by the American Academy of Pediatrics and the Children’s Hospital Association.
From Feb. 19 to March 4, the drop in new cases averaged just 5% each week, compared with 13.3% per week over the 5-week period from Jan. 15 to Feb. 18. For the week of March 5-11, a total of 52,695 COVID-19 cases were reported in children, down from 63,562 the previous week and the lowest number since late October, based on data from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
In those jurisdictions, 3.28 million children have been infected with SARS-CoV-2, representing 13.2% of all cases since the beginning of the pandemic. The cumulative rate of COVID-19 has now risen to 4,364 cases per 100,000 children nationally, with state rates ranging from 1,062 per 100,000 in Hawaii to 8,692 per 100,000 in North Dakota, the AAP and CHA said in their weekly COVID-19 report.
Hospitalization data are more limited – 24 states and New York City – but continue to show that serious illness is much less common in younger individuals: Children represent just 1.9% of all hospitalizations, and only 0.8% of the children who have been infected were hospitalized. Neither rate has changed since early February, the AAP and CHA said.
The number of deaths in children, however, rose from 253 to 266, the largest 1-week increase since early February in the 43 states (along with New York City, Puerto Rico, and Guam) that are tracking mortality data by age, the AAP and CHA reported.
Among those 46 jurisdictions, there are 10 (9 states and the District of Columbia) that have not yet reported a COVID-19–related child death, while Texas has almost twice as many deaths, 47, as the next state, Arizona, which has 24. Meanwhile, California’s total of 452,000 cases is almost 2½ times higher than the 183,000 recorded by Illinois, according to the report.
First pill for COVID-19 could be ready by year’s end
New pills to treat patients with COVID-19 are currently in midstage clinical trials and, if successful, could be ready by the end of the year.
Only one treatment – remdesivir (Veklury) – has been fully approved by the U.S. Food and Drug Administration for patients in the hospital and it must be administered intravenously.
Hopes for a day when patients with COVID-19 can take a pill to rid their bodies of the virus got a boost when early trial results were presented at a medical conference.
Interim phase 2 results for the oral experimental COVID-19 drug molnupiravir, designed to do for patients with COVID-19 what oseltamivir (Tamiflu) can do for patients with the flu, were presented at the Conference on Retroviruses and Opportunistic Infections 2021 Annual Meeting, as reported by this news organization.
In the small study, the pill significantly reduced infectious virus in patients who were symptomatic and had tested positive for COVID-19 during the previous 4 days but were not hospitalized.
After 5 days of treatment, no participants who received molnupiravir had detectable virus, whereas 24% who received placebo did.
Two other oral agents are being developed by RedHill Biopharma: one for severe COVID-19 infection for hospitalized patients and one for patients at home with mild infection.
The first, opaganib (Yeliva), proceeded to a phase 2/3 global trial for hospitalized patients after the company announced top-line safety and efficacy data in December. In phase 2, the drug was shown to be safe in patients requiring oxygen and effectively reduced the need for oxygen by the end of the treatment period.
A key feature is that it is both an antiviral and an anti-inflammatory, Gilead Raday, RedHill’s chief operating officer, said in an interview. Data are expected midyear on its performance in 464 patients. The drug is being tested on top of remdesivir or in addition to dexamethasone.
The second, upamostat (RHB-107), is currently undergoing a phase 2/3 trial in the United States and is being investigated for use in nonhospitalized COVID-19 patients.
“I would expect data to be available in the second half of this year,” Mr. Raday said.
Upamostat is a novel serine protease inhibitor expected to be effective against emerging variants because it targets human cell factors involved in viral entry, according to the company.
Other drugs are being investigated in trials that are in earlier stages.
Urgent need for oral agents
Infectious disease specialists are watching the move toward a COVID-19 pill enthusiastically.
“We badly need an oral treatment option for COVID,” said Sarah Doernberg, MD, an infectious disease specialist from the University of California, San Francisco.
“It’s a real gap in our armamentarium for COVID in outpatient treatment, which is where most who contract COVID-19 will seek care,” she said in an interview.
Although some studies have shown the benefit of monoclonal antibodies for prevention and early treatment, there are major logistical issues because all the current options require IV administration, she explained.
“If we had a pill to treat early COVID, especially in high-risk patients, it would fill a gap,” she said, noting that a pill could help people get better faster and prevent hospital stays.
Studies of molnupiravir suggest that it decreases viral shedding in the first few days after COVID infection, Dr. Doernberg reported.
There is excitement around the drug, but it will be important to see whether the results translate into fewer people requiring hospital admission and whether people feel better faster.
“I want to see the clinical data,” Dr. Doernberg said.
She will also be watching for the upamostat and opaganib results in the coming weeks.
“If these drugs are successful, I think it’s possible we could use them – maybe under an emergency use authorization – this year,” she said.
Once antiviral pills are a viable option for COVID-19 treatment, questions will arise about their use, she said.
One question is whether patients who are getting remdesivir in the hospital and are ready to leave after 5 days should continue treatment with antiviral pills at home.
Another is whether the pills – if they are shown to be effective – will be helpful for COVID post exposure. That use would be important for people who do not have COVID-19 but who are in close contact with someone who does, such as a member of their household.
“We have that model,” Dr. Doernberg said. “We know that oseltamivir can be used for postexposure prophylaxis and can help to prevent development of clinical disease.”
But she cautioned that a challenge with COVID is that people are contagious very early. A pill would need to come with the ability to test for COVID-19 early and get patients linked to care immediately.
“Those are not small challenges,” she said.
Vaccines alone won’t end the COVID threat
Treatments are part of the “belt-and-suspenders” approach, along with vaccines to combat COVID-19, Dr. Doernberg said.
“We’re not going to eradicate COVID,” she said. “We’re still going to need treatments for people who either don’t respond to the vaccine or haven’t gotten the vaccine or developed disease despite the vaccine.”
Oral formulations are desperately needed, agreed Kenneth Johnson, PhD, professor of molecular biosciences at the University of Texas at Austin.
Right now, remdesivir treatments involve patients being hooked up to an IV for 30-120 minutes each day for 5 days. And the cost of a 5-day course of remdesivir ranges from $2340 to $3120 in the United States.
“We’re hoping we can come up with something that is a little bit easier to administer, and without as many concerns for toxic side effects,” he said.
Dr. Johnson’s team at UT-Austin recently made a key discovery about the way remdesivir stops the replication of viral RNA.
The understanding of where the virus starts to replicate in the infection chain of events and how and where it reacts with remdesivir might lead to the development of better, more concentrated pill forms of antivirals in the future, with fewer toxicities, he said.
The team used a lab dish to recreate the step-by-step process that occurs when a patient who is infected with SARS-CoV-2 receives remdesivir.
The discovery was published online in Molecular Cell in January and will be printed in the April issue of the journal.
The discovery won’t lead to an effective COVID-19 pill for our current crisis, but will be important for the next generation of drugs needed to deal with future coronaviruses, Dr. Johnson explained.
And there will be other coronaviruses, he said, noting that this one is the third in 20 years to jump from animals to humans. “It’s just a matter of time,” he said.
A version of this article first appeared on Medscape.com.
New pills to treat patients with COVID-19 are currently in midstage clinical trials and, if successful, could be ready by the end of the year.
Only one treatment – remdesivir (Veklury) – has been fully approved by the U.S. Food and Drug Administration for patients in the hospital and it must be administered intravenously.
Hopes for a day when patients with COVID-19 can take a pill to rid their bodies of the virus got a boost when early trial results were presented at a medical conference.
Interim phase 2 results for the oral experimental COVID-19 drug molnupiravir, designed to do for patients with COVID-19 what oseltamivir (Tamiflu) can do for patients with the flu, were presented at the Conference on Retroviruses and Opportunistic Infections 2021 Annual Meeting, as reported by this news organization.
In the small study, the pill significantly reduced infectious virus in patients who were symptomatic and had tested positive for COVID-19 during the previous 4 days but were not hospitalized.
After 5 days of treatment, no participants who received molnupiravir had detectable virus, whereas 24% who received placebo did.
Two other oral agents are being developed by RedHill Biopharma: one for severe COVID-19 infection for hospitalized patients and one for patients at home with mild infection.
The first, opaganib (Yeliva), proceeded to a phase 2/3 global trial for hospitalized patients after the company announced top-line safety and efficacy data in December. In phase 2, the drug was shown to be safe in patients requiring oxygen and effectively reduced the need for oxygen by the end of the treatment period.
A key feature is that it is both an antiviral and an anti-inflammatory, Gilead Raday, RedHill’s chief operating officer, said in an interview. Data are expected midyear on its performance in 464 patients. The drug is being tested on top of remdesivir or in addition to dexamethasone.
The second, upamostat (RHB-107), is currently undergoing a phase 2/3 trial in the United States and is being investigated for use in nonhospitalized COVID-19 patients.
“I would expect data to be available in the second half of this year,” Mr. Raday said.
Upamostat is a novel serine protease inhibitor expected to be effective against emerging variants because it targets human cell factors involved in viral entry, according to the company.
Other drugs are being investigated in trials that are in earlier stages.
Urgent need for oral agents
Infectious disease specialists are watching the move toward a COVID-19 pill enthusiastically.
“We badly need an oral treatment option for COVID,” said Sarah Doernberg, MD, an infectious disease specialist from the University of California, San Francisco.
“It’s a real gap in our armamentarium for COVID in outpatient treatment, which is where most who contract COVID-19 will seek care,” she said in an interview.
Although some studies have shown the benefit of monoclonal antibodies for prevention and early treatment, there are major logistical issues because all the current options require IV administration, she explained.
“If we had a pill to treat early COVID, especially in high-risk patients, it would fill a gap,” she said, noting that a pill could help people get better faster and prevent hospital stays.
Studies of molnupiravir suggest that it decreases viral shedding in the first few days after COVID infection, Dr. Doernberg reported.
There is excitement around the drug, but it will be important to see whether the results translate into fewer people requiring hospital admission and whether people feel better faster.
“I want to see the clinical data,” Dr. Doernberg said.
She will also be watching for the upamostat and opaganib results in the coming weeks.
“If these drugs are successful, I think it’s possible we could use them – maybe under an emergency use authorization – this year,” she said.
Once antiviral pills are a viable option for COVID-19 treatment, questions will arise about their use, she said.
One question is whether patients who are getting remdesivir in the hospital and are ready to leave after 5 days should continue treatment with antiviral pills at home.
Another is whether the pills – if they are shown to be effective – will be helpful for COVID post exposure. That use would be important for people who do not have COVID-19 but who are in close contact with someone who does, such as a member of their household.
“We have that model,” Dr. Doernberg said. “We know that oseltamivir can be used for postexposure prophylaxis and can help to prevent development of clinical disease.”
But she cautioned that a challenge with COVID is that people are contagious very early. A pill would need to come with the ability to test for COVID-19 early and get patients linked to care immediately.
“Those are not small challenges,” she said.
Vaccines alone won’t end the COVID threat
Treatments are part of the “belt-and-suspenders” approach, along with vaccines to combat COVID-19, Dr. Doernberg said.
“We’re not going to eradicate COVID,” she said. “We’re still going to need treatments for people who either don’t respond to the vaccine or haven’t gotten the vaccine or developed disease despite the vaccine.”
Oral formulations are desperately needed, agreed Kenneth Johnson, PhD, professor of molecular biosciences at the University of Texas at Austin.
Right now, remdesivir treatments involve patients being hooked up to an IV for 30-120 minutes each day for 5 days. And the cost of a 5-day course of remdesivir ranges from $2340 to $3120 in the United States.
“We’re hoping we can come up with something that is a little bit easier to administer, and without as many concerns for toxic side effects,” he said.
Dr. Johnson’s team at UT-Austin recently made a key discovery about the way remdesivir stops the replication of viral RNA.
The understanding of where the virus starts to replicate in the infection chain of events and how and where it reacts with remdesivir might lead to the development of better, more concentrated pill forms of antivirals in the future, with fewer toxicities, he said.
The team used a lab dish to recreate the step-by-step process that occurs when a patient who is infected with SARS-CoV-2 receives remdesivir.
The discovery was published online in Molecular Cell in January and will be printed in the April issue of the journal.
The discovery won’t lead to an effective COVID-19 pill for our current crisis, but will be important for the next generation of drugs needed to deal with future coronaviruses, Dr. Johnson explained.
And there will be other coronaviruses, he said, noting that this one is the third in 20 years to jump from animals to humans. “It’s just a matter of time,” he said.
A version of this article first appeared on Medscape.com.
New pills to treat patients with COVID-19 are currently in midstage clinical trials and, if successful, could be ready by the end of the year.
Only one treatment – remdesivir (Veklury) – has been fully approved by the U.S. Food and Drug Administration for patients in the hospital and it must be administered intravenously.
Hopes for a day when patients with COVID-19 can take a pill to rid their bodies of the virus got a boost when early trial results were presented at a medical conference.
Interim phase 2 results for the oral experimental COVID-19 drug molnupiravir, designed to do for patients with COVID-19 what oseltamivir (Tamiflu) can do for patients with the flu, were presented at the Conference on Retroviruses and Opportunistic Infections 2021 Annual Meeting, as reported by this news organization.
In the small study, the pill significantly reduced infectious virus in patients who were symptomatic and had tested positive for COVID-19 during the previous 4 days but were not hospitalized.
After 5 days of treatment, no participants who received molnupiravir had detectable virus, whereas 24% who received placebo did.
Two other oral agents are being developed by RedHill Biopharma: one for severe COVID-19 infection for hospitalized patients and one for patients at home with mild infection.
The first, opaganib (Yeliva), proceeded to a phase 2/3 global trial for hospitalized patients after the company announced top-line safety and efficacy data in December. In phase 2, the drug was shown to be safe in patients requiring oxygen and effectively reduced the need for oxygen by the end of the treatment period.
A key feature is that it is both an antiviral and an anti-inflammatory, Gilead Raday, RedHill’s chief operating officer, said in an interview. Data are expected midyear on its performance in 464 patients. The drug is being tested on top of remdesivir or in addition to dexamethasone.
The second, upamostat (RHB-107), is currently undergoing a phase 2/3 trial in the United States and is being investigated for use in nonhospitalized COVID-19 patients.
“I would expect data to be available in the second half of this year,” Mr. Raday said.
Upamostat is a novel serine protease inhibitor expected to be effective against emerging variants because it targets human cell factors involved in viral entry, according to the company.
Other drugs are being investigated in trials that are in earlier stages.
Urgent need for oral agents
Infectious disease specialists are watching the move toward a COVID-19 pill enthusiastically.
“We badly need an oral treatment option for COVID,” said Sarah Doernberg, MD, an infectious disease specialist from the University of California, San Francisco.
“It’s a real gap in our armamentarium for COVID in outpatient treatment, which is where most who contract COVID-19 will seek care,” she said in an interview.
Although some studies have shown the benefit of monoclonal antibodies for prevention and early treatment, there are major logistical issues because all the current options require IV administration, she explained.
“If we had a pill to treat early COVID, especially in high-risk patients, it would fill a gap,” she said, noting that a pill could help people get better faster and prevent hospital stays.
Studies of molnupiravir suggest that it decreases viral shedding in the first few days after COVID infection, Dr. Doernberg reported.
There is excitement around the drug, but it will be important to see whether the results translate into fewer people requiring hospital admission and whether people feel better faster.
“I want to see the clinical data,” Dr. Doernberg said.
She will also be watching for the upamostat and opaganib results in the coming weeks.
“If these drugs are successful, I think it’s possible we could use them – maybe under an emergency use authorization – this year,” she said.
Once antiviral pills are a viable option for COVID-19 treatment, questions will arise about their use, she said.
One question is whether patients who are getting remdesivir in the hospital and are ready to leave after 5 days should continue treatment with antiviral pills at home.
Another is whether the pills – if they are shown to be effective – will be helpful for COVID post exposure. That use would be important for people who do not have COVID-19 but who are in close contact with someone who does, such as a member of their household.
“We have that model,” Dr. Doernberg said. “We know that oseltamivir can be used for postexposure prophylaxis and can help to prevent development of clinical disease.”
But she cautioned that a challenge with COVID is that people are contagious very early. A pill would need to come with the ability to test for COVID-19 early and get patients linked to care immediately.
“Those are not small challenges,” she said.
Vaccines alone won’t end the COVID threat
Treatments are part of the “belt-and-suspenders” approach, along with vaccines to combat COVID-19, Dr. Doernberg said.
“We’re not going to eradicate COVID,” she said. “We’re still going to need treatments for people who either don’t respond to the vaccine or haven’t gotten the vaccine or developed disease despite the vaccine.”
Oral formulations are desperately needed, agreed Kenneth Johnson, PhD, professor of molecular biosciences at the University of Texas at Austin.
Right now, remdesivir treatments involve patients being hooked up to an IV for 30-120 minutes each day for 5 days. And the cost of a 5-day course of remdesivir ranges from $2340 to $3120 in the United States.
“We’re hoping we can come up with something that is a little bit easier to administer, and without as many concerns for toxic side effects,” he said.
Dr. Johnson’s team at UT-Austin recently made a key discovery about the way remdesivir stops the replication of viral RNA.
The understanding of where the virus starts to replicate in the infection chain of events and how and where it reacts with remdesivir might lead to the development of better, more concentrated pill forms of antivirals in the future, with fewer toxicities, he said.
The team used a lab dish to recreate the step-by-step process that occurs when a patient who is infected with SARS-CoV-2 receives remdesivir.
The discovery was published online in Molecular Cell in January and will be printed in the April issue of the journal.
The discovery won’t lead to an effective COVID-19 pill for our current crisis, but will be important for the next generation of drugs needed to deal with future coronaviruses, Dr. Johnson explained.
And there will be other coronaviruses, he said, noting that this one is the third in 20 years to jump from animals to humans. “It’s just a matter of time,” he said.
A version of this article first appeared on Medscape.com.
Is pediatric subspecialty training financially worth it?
Pursuing fellowship training is often financially costly in terms of lifetime earnings, compared with starting a career as a general pediatrician immediately after residency, a report suggests.
Researchers found that most pediatric subspecialists – including those practicing neurology, pulmonology, and adolescent medicine – do not see a financial return from additional training because of the delays in receiving increased compensation and the repayment of educational debt.
“Most pediatric subspecialists don’t experience a relative increase in compensation after training compared to a general pediatrician, so there isn’t a financial benefit to additional training,” lead author Eva Catenaccio, MD, from the division of pediatric neurology, department of neurology, Johns Hopkins University, Baltimore, told this news organization.
The findings, published online March 8 in Pediatrics, contribute to the ongoing debate about the length of pediatric fellowship training programs. The data also provide evidence for the potential effect of a pediatric subspecialty loan repayment program.
Pediatric subspecialty training rarely pays off
However, not all practitioners in pediatric subspecialties would find themselves in the red relative to their generalist peers. Three subspecialties had a positive financial return: cardiology, critical care, and neonatology. Dr. Catenaccio explained that this may be because these subspecialties tend to be “inpatient procedure oriented, which are often more [lucrative] than outpatient cognitive–oriented subspecialties, such as pediatric infectious diseases, endocrinology, or adolescent medicine.”
Enrolling in a pediatric fellowship program resulted in lifetime financial returns that ranged from an increase of $852,129 for cardiology, relative to general pediatrics, to a loss of $1,594,366 for adolescent medicine, researchers found.
For the study, researchers calculated the financial returns of 15 pediatric subspecialties – emergency medicine, neurology, cardiology, critical care, neonatology, hematology and oncology, pulmonology, hospitalist medicine, allergy and immunology, gastroenterology, rheumatology, nephrology, adolescent medicine, infectious diseases, and endocrinology – in comparison with returns of private practice general pediatrics on the basis of 2018-2019 data on fellowship stipends, compensation, and educational debt.
They obtained most of the data from the Association of American Medical Colleges Survey of Resident/Fellow Stipends and Benefits, AAMC’s annual Medical School Faculty Salary Report, and the AAMC Medical School Graduation Questionnaire.
Richard Mink, MD, department of pediatrics, Harbor-UCLA Medical Center, Torrance, Calif., noted that it would have been helpful to have also compared the lifetime earnings of practitioners in pediatric subspecialties to academic general pediatricians and not just those in private practice.
The financial gap has worsened
To better understand which aspects of fellowship training have the greatest effect on lifetime compensation, Dr. Catenaccio and colleagues evaluated the potential effects of shortening fellowship length, eliminating school debt, and implementing a federal loan repayment plan. These changes enhanced the returns of cardiology, critical care, and neonatology – subspecialties that had already seen financial returns before these changes – and resulted in a positive financial return for emergency medicine.
The changes also narrowed the financial gap between subspecialties and general pediatrics. However, the remaining subspecialties still earned less than private practice pediatrics.
The new study is an update to a 2011 report, which reflected 2007-2008 data for 11 subspecialties. This time around, the researchers included the subspecialty of hospitalist medicine, which was approved as a board-certified subspecialty by the American Board of Pediatrics in 2014, as well as neurology, allergy and immunology, and adolescent medicine.
“I was most surprised that the additional pediatric subspecialties we included since the 2011 report followed the same general trend, with pediatric subspecialty training having a lower lifetime earning potential than general pediatrics,” Dr. Catenaccio said.
Comparing results from the two study periods showed that the financial gap between general pediatrics and subspecialty pediatrics worsened over time. For example, the financial return for pediatric endocrinology decreased an additional $500,000 between 2007 and 2018.
The researchers believe a combination of increased educational debt burden, slow growth in compensation, and changing interest rates over time have caused the financial differences between general pediatrics and subspecialty pediatrics to become more pronounced.
‘Pediatric subspecialty training is worth it!’
Despite the financial gaps, Dr. Catenaccio and colleagues say pediatric subspecialty training is still worthwhile but that policymakers should address these financial differences to help guide workforce distribution in a way that meets the needs of patients.
“I think pediatric subspecialty training is worth it,” said Dr. Catenaccio, who’s pursuing pediatric subspecialty training. “There are so many factors that go into choosing a specialty or subspecialty in medicine, including the desire to care for a particular patient population, interest in certain diseases or organ systems, lifestyle considerations, and research opportunities.”
But it’s also important for trainees to be aware of economic considerations in their decision-making.
Dr. Mink, who wrote an accompanying commentary, agrees that young clinicians should not make career decisions on the basis of metrics such as lifetime earning measures.
“I think people who go into pediatrics have decided that money is not the driving force,” said Dr. Mink. He noted that pediatricians are usually not paid well, compared with other specialists. “To me the important thing is you have to like what you’re doing.”
A 2020 study found that trainees who chose a career in pediatric pulmonology, a subspecialty, said that financial considerations were not the driving factor in their decision-making. Nevertheless, Dr. Mink also believes young clinicians should take into account their educational debt.
The further widening of the financial gap between general pediatrics and pediatric subspecialties could lead to shortages in the pediatric subspecialty workforce.
The authors and Dr. Mink have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pursuing fellowship training is often financially costly in terms of lifetime earnings, compared with starting a career as a general pediatrician immediately after residency, a report suggests.
Researchers found that most pediatric subspecialists – including those practicing neurology, pulmonology, and adolescent medicine – do not see a financial return from additional training because of the delays in receiving increased compensation and the repayment of educational debt.
“Most pediatric subspecialists don’t experience a relative increase in compensation after training compared to a general pediatrician, so there isn’t a financial benefit to additional training,” lead author Eva Catenaccio, MD, from the division of pediatric neurology, department of neurology, Johns Hopkins University, Baltimore, told this news organization.
The findings, published online March 8 in Pediatrics, contribute to the ongoing debate about the length of pediatric fellowship training programs. The data also provide evidence for the potential effect of a pediatric subspecialty loan repayment program.
Pediatric subspecialty training rarely pays off
However, not all practitioners in pediatric subspecialties would find themselves in the red relative to their generalist peers. Three subspecialties had a positive financial return: cardiology, critical care, and neonatology. Dr. Catenaccio explained that this may be because these subspecialties tend to be “inpatient procedure oriented, which are often more [lucrative] than outpatient cognitive–oriented subspecialties, such as pediatric infectious diseases, endocrinology, or adolescent medicine.”
Enrolling in a pediatric fellowship program resulted in lifetime financial returns that ranged from an increase of $852,129 for cardiology, relative to general pediatrics, to a loss of $1,594,366 for adolescent medicine, researchers found.
For the study, researchers calculated the financial returns of 15 pediatric subspecialties – emergency medicine, neurology, cardiology, critical care, neonatology, hematology and oncology, pulmonology, hospitalist medicine, allergy and immunology, gastroenterology, rheumatology, nephrology, adolescent medicine, infectious diseases, and endocrinology – in comparison with returns of private practice general pediatrics on the basis of 2018-2019 data on fellowship stipends, compensation, and educational debt.
They obtained most of the data from the Association of American Medical Colleges Survey of Resident/Fellow Stipends and Benefits, AAMC’s annual Medical School Faculty Salary Report, and the AAMC Medical School Graduation Questionnaire.
Richard Mink, MD, department of pediatrics, Harbor-UCLA Medical Center, Torrance, Calif., noted that it would have been helpful to have also compared the lifetime earnings of practitioners in pediatric subspecialties to academic general pediatricians and not just those in private practice.
The financial gap has worsened
To better understand which aspects of fellowship training have the greatest effect on lifetime compensation, Dr. Catenaccio and colleagues evaluated the potential effects of shortening fellowship length, eliminating school debt, and implementing a federal loan repayment plan. These changes enhanced the returns of cardiology, critical care, and neonatology – subspecialties that had already seen financial returns before these changes – and resulted in a positive financial return for emergency medicine.
The changes also narrowed the financial gap between subspecialties and general pediatrics. However, the remaining subspecialties still earned less than private practice pediatrics.
The new study is an update to a 2011 report, which reflected 2007-2008 data for 11 subspecialties. This time around, the researchers included the subspecialty of hospitalist medicine, which was approved as a board-certified subspecialty by the American Board of Pediatrics in 2014, as well as neurology, allergy and immunology, and adolescent medicine.
“I was most surprised that the additional pediatric subspecialties we included since the 2011 report followed the same general trend, with pediatric subspecialty training having a lower lifetime earning potential than general pediatrics,” Dr. Catenaccio said.
Comparing results from the two study periods showed that the financial gap between general pediatrics and subspecialty pediatrics worsened over time. For example, the financial return for pediatric endocrinology decreased an additional $500,000 between 2007 and 2018.
The researchers believe a combination of increased educational debt burden, slow growth in compensation, and changing interest rates over time have caused the financial differences between general pediatrics and subspecialty pediatrics to become more pronounced.
‘Pediatric subspecialty training is worth it!’
Despite the financial gaps, Dr. Catenaccio and colleagues say pediatric subspecialty training is still worthwhile but that policymakers should address these financial differences to help guide workforce distribution in a way that meets the needs of patients.
“I think pediatric subspecialty training is worth it,” said Dr. Catenaccio, who’s pursuing pediatric subspecialty training. “There are so many factors that go into choosing a specialty or subspecialty in medicine, including the desire to care for a particular patient population, interest in certain diseases or organ systems, lifestyle considerations, and research opportunities.”
But it’s also important for trainees to be aware of economic considerations in their decision-making.
Dr. Mink, who wrote an accompanying commentary, agrees that young clinicians should not make career decisions on the basis of metrics such as lifetime earning measures.
“I think people who go into pediatrics have decided that money is not the driving force,” said Dr. Mink. He noted that pediatricians are usually not paid well, compared with other specialists. “To me the important thing is you have to like what you’re doing.”
A 2020 study found that trainees who chose a career in pediatric pulmonology, a subspecialty, said that financial considerations were not the driving factor in their decision-making. Nevertheless, Dr. Mink also believes young clinicians should take into account their educational debt.
The further widening of the financial gap between general pediatrics and pediatric subspecialties could lead to shortages in the pediatric subspecialty workforce.
The authors and Dr. Mink have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pursuing fellowship training is often financially costly in terms of lifetime earnings, compared with starting a career as a general pediatrician immediately after residency, a report suggests.
Researchers found that most pediatric subspecialists – including those practicing neurology, pulmonology, and adolescent medicine – do not see a financial return from additional training because of the delays in receiving increased compensation and the repayment of educational debt.
“Most pediatric subspecialists don’t experience a relative increase in compensation after training compared to a general pediatrician, so there isn’t a financial benefit to additional training,” lead author Eva Catenaccio, MD, from the division of pediatric neurology, department of neurology, Johns Hopkins University, Baltimore, told this news organization.
The findings, published online March 8 in Pediatrics, contribute to the ongoing debate about the length of pediatric fellowship training programs. The data also provide evidence for the potential effect of a pediatric subspecialty loan repayment program.
Pediatric subspecialty training rarely pays off
However, not all practitioners in pediatric subspecialties would find themselves in the red relative to their generalist peers. Three subspecialties had a positive financial return: cardiology, critical care, and neonatology. Dr. Catenaccio explained that this may be because these subspecialties tend to be “inpatient procedure oriented, which are often more [lucrative] than outpatient cognitive–oriented subspecialties, such as pediatric infectious diseases, endocrinology, or adolescent medicine.”
Enrolling in a pediatric fellowship program resulted in lifetime financial returns that ranged from an increase of $852,129 for cardiology, relative to general pediatrics, to a loss of $1,594,366 for adolescent medicine, researchers found.
For the study, researchers calculated the financial returns of 15 pediatric subspecialties – emergency medicine, neurology, cardiology, critical care, neonatology, hematology and oncology, pulmonology, hospitalist medicine, allergy and immunology, gastroenterology, rheumatology, nephrology, adolescent medicine, infectious diseases, and endocrinology – in comparison with returns of private practice general pediatrics on the basis of 2018-2019 data on fellowship stipends, compensation, and educational debt.
They obtained most of the data from the Association of American Medical Colleges Survey of Resident/Fellow Stipends and Benefits, AAMC’s annual Medical School Faculty Salary Report, and the AAMC Medical School Graduation Questionnaire.
Richard Mink, MD, department of pediatrics, Harbor-UCLA Medical Center, Torrance, Calif., noted that it would have been helpful to have also compared the lifetime earnings of practitioners in pediatric subspecialties to academic general pediatricians and not just those in private practice.
The financial gap has worsened
To better understand which aspects of fellowship training have the greatest effect on lifetime compensation, Dr. Catenaccio and colleagues evaluated the potential effects of shortening fellowship length, eliminating school debt, and implementing a federal loan repayment plan. These changes enhanced the returns of cardiology, critical care, and neonatology – subspecialties that had already seen financial returns before these changes – and resulted in a positive financial return for emergency medicine.
The changes also narrowed the financial gap between subspecialties and general pediatrics. However, the remaining subspecialties still earned less than private practice pediatrics.
The new study is an update to a 2011 report, which reflected 2007-2008 data for 11 subspecialties. This time around, the researchers included the subspecialty of hospitalist medicine, which was approved as a board-certified subspecialty by the American Board of Pediatrics in 2014, as well as neurology, allergy and immunology, and adolescent medicine.
“I was most surprised that the additional pediatric subspecialties we included since the 2011 report followed the same general trend, with pediatric subspecialty training having a lower lifetime earning potential than general pediatrics,” Dr. Catenaccio said.
Comparing results from the two study periods showed that the financial gap between general pediatrics and subspecialty pediatrics worsened over time. For example, the financial return for pediatric endocrinology decreased an additional $500,000 between 2007 and 2018.
The researchers believe a combination of increased educational debt burden, slow growth in compensation, and changing interest rates over time have caused the financial differences between general pediatrics and subspecialty pediatrics to become more pronounced.
‘Pediatric subspecialty training is worth it!’
Despite the financial gaps, Dr. Catenaccio and colleagues say pediatric subspecialty training is still worthwhile but that policymakers should address these financial differences to help guide workforce distribution in a way that meets the needs of patients.
“I think pediatric subspecialty training is worth it,” said Dr. Catenaccio, who’s pursuing pediatric subspecialty training. “There are so many factors that go into choosing a specialty or subspecialty in medicine, including the desire to care for a particular patient population, interest in certain diseases or organ systems, lifestyle considerations, and research opportunities.”
But it’s also important for trainees to be aware of economic considerations in their decision-making.
Dr. Mink, who wrote an accompanying commentary, agrees that young clinicians should not make career decisions on the basis of metrics such as lifetime earning measures.
“I think people who go into pediatrics have decided that money is not the driving force,” said Dr. Mink. He noted that pediatricians are usually not paid well, compared with other specialists. “To me the important thing is you have to like what you’re doing.”
A 2020 study found that trainees who chose a career in pediatric pulmonology, a subspecialty, said that financial considerations were not the driving factor in their decision-making. Nevertheless, Dr. Mink also believes young clinicians should take into account their educational debt.
The further widening of the financial gap between general pediatrics and pediatric subspecialties could lead to shortages in the pediatric subspecialty workforce.
The authors and Dr. Mink have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pregnant patients with severe COVID-19 disease at increased risk of complications
Pregnant patients with COVID-19 infections were more likely to experience severe disease if they had preexisting comorbidities, such as chronic hypertension, asthma, or pregestational diabetes, according to findings from a new study presented at the meeting sponsored by the Society for Maternal-Fetal Medicine.
The study included outcomes for the largest multistate cohort of pregnant patients with COVID-19 outside of what the Centers for Disease Control and Prevention is tracking. Its findings also mirrored those of a multicenter, retrospective study in Washington state, published in the American Journal of Obstetrics & Gynecology. That study also found that pregnant patients hospitalized for COVID-19 were more likely to have comorbidities, and both studies found an increased likelihood of preterm birth among pregnant patients with severe or critical disease.
Disease severity linked to risk of perinatal complications
In the abstract presented at the SMFM meeting, more severe disease was associated with older age and a higher median body mass index, as seen in the general population, but the researchers found no differences in disease severity occurred by race or ethnicity, Torri D. Metz, MD, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network, told attendees of the conference. The researchers also found that perinatal complications were more prevalent in those with severe or critical COVID-19 disease but not in those with mild or moderate disease. Vertical COVID-19 transmission from mother to child was rare.
The observational study included all patients who had a singleton pregnancy, had a positive SARS-CoV-2 test, and delivered between March 1 and July 31, 2020, at one of the 33 U.S. hospitals in the NICHD Maternal-Fetal Medicine Units Network, spread across 14 states. The researchers used electronic medical records to determine incidence of cesarean delivery, postpartum hemorrhage, hypertensive disorders of pregnancy, preterm birth (less than 37 weeks), maternal death, infant death, and positive infant COVID-19 test. They tracked mothers through 6 weeks post partum and newborns through delivery hospitalization.
Of 1,291 patients in the cohort, 1,219 received their first positive COVID-19 test during pregnancy. The others tested positive while in the hospital for delivery or within a month and a half after discharge. Limiting their analysis to those who developed COVID-19 while pregnant prior to delivery, nearly half (47%) were asymptomatic.
The disease was mild in 27%, moderate in 14%, severe in 8%, and critical in 4%. The researchers used the National Institutes of Health classifications for severity and included deaths in the critical group. The most common symptom was a cough, reported by a third of the patients (34%). Four of six maternal deaths that occurred were caused by COVID-19.
Compared with an average age of 28 in those without symptoms, the mean age was 29 in those with mild/moderate disease and 30 in those with severe/critical disease (P = .006). Similarly, the mean BMI was 28.3 in asymptomatic patients, 29 in those with mild/moderate disease, and 32.3 in those with severe/critical disease (P < .001). Despite a diverse cohort – 53% Hispanic, 23% Black, and 15% White – the researches found no racial/ethnic trends in disease severity.
Patients who had asthma, chronic obstructive pulmonary disorder, pregestational diabetes, chronic hypertension, chronic liver disease, or a seizure disorder were all significantly more likely to have critical/severe disease than mild/moderate disease, and more likely to have mild/moderate disease than asymptomatic (P values ranged from < .001 to .02).
The mothers with critical or severe illness were 1.6 times more likely to have cesarean births and to have hypertensive disorders of pregnancy, and they were twice as likely to have postpartum hemorrhage (P < .001; P = .007). Those with mild or moderate disease, however, had no increased risks for perinatal complications over asymptomatic patients.
Critical or severe illness was also associated with more than triple the risk of preterm birth (adjusted risk ratio, 3.6; P < .001). Newborns of mothers with critical or severe illness also had three times greater risk of neonatal ICU admission (ARR, 3.1; P <. 001) and weighed an average 385 g less than newborns of asymptomatic mothers. COVID-19 rate among infants was only 1% during delivery hospitalization.
Since the study cutoff was July 30 and COVID infections only became prevalent in March, the researchers were unable to evaluate women for outcomes resulting from COVID infections in early pregnancy, such as congenital anomalies or early miscarriage, Dr. Metz said. In addition, since many of the sites are urban centers, the data may not be generalizable to rural areas.
Peter S. Bernstein, MD, MPH, of Montefiore Medical Center, New York, asked whether the increased cesarean deliveries and preterm births in the group of women with severe disease were caused by usual obstetric causes or the treatment of COVID-19 infection. Dr. Metz said the vast majority of preterm deliveries were indicated, but only a small proportion were induced for COVID-19 alone. “A lot had hypertensive disorders of pregnancies or PPROM, so it’s partly driven by the infection itself but also partly driven by some of those perinatal complications,” she said.
Similar findings in Washington
In the Washington study, among 240 pregnant patients with confirmed COVID-19 infection between March 1 and July 30, 2020, 1 in 11 developed severe or critical disease, and 1 in 10 were hospitalized. The pregnant patients had more than triple the risk of hospitalization compared with adults of similar ages in the general population (10% vs. 2.8%; rate ratio, 3.5). Similar to the multistate NICHD study, women were more likely to be hospitalized if they had asthma, hypertension, type 2 diabetes, autoimmune disease, or class III obesity.
Three mothers died of COVID-19, resulting in a case fatality rate 13.6 times greater than nonpregnant patients with COVID-19 in the general population. The absolute difference in the rate was 1.2%. As seen in the NICHD study, preterm birth was more common in mothers with severe or critical COVID-19. Nearly half (45.4%) of mothers with severe or critical COVID-19 delivered preterm compared to 5.2% in those with mild COVID-19 (P < .001).
“Our finding that deaths in pregnant patients contributed disproportionately to deaths from COVID-19 among 20- to 39-year-olds in Washington state is similar to what was observed during the influenza A virus H1N1 2009 pandemic,” Erica M. Lokken, PhD, MS, of the departments of global health and ob.gyn. at the University of Washington, Seattle, and colleagues wrote in the Washington study. But they noted that it took 8 months into the pandemic before pregnant patients were identified as a high-risk group for COVID-19.
“Given the similarity in clinical course between COVID-19 and IAV H1N1 2009 with an increased risk for mortality during pregnancy and the postpartum period, we strongly recommend that pregnant patients should be considered a high-risk population to novel highly pathogenic respiratory viruses until proven otherwise by population-based studies with good ascertainment of pregnancy status,” they wrote.
Judette Louis, MD, MPH, associate professor of ob.gyn. and department chair at the University of South Florida, Tampa, said in an interview that the findings in these studies were fairly expected, but it’s important to have data from such a large cohort as the one presented at SMFM.
“It confirmed that those who had severe disease were more likely to have chronic medical conditions, mirroring what we saw in the general population who isn’t pregnant,” Dr. Louis said. “I thought this was very crucial because as pregnant women are trying to decide whether they should get the COVID vaccine, this provides support to say that if you’re pregnant, you’re more likely to have severe disease [if you have] other chronic medical conditions.”
The findings also confirm the importance of pregnant people taking precautions to avoid infection.
“Even though these individuals are, as a group, in an age cohort that mostly has asymptomatic disease, for some of them, it results in severe disease and even maternal death,” she said. “They should still take it seriously if they’re pregnant.”
The SMFM abstract study was funded by the NICHD. The Washington study was funded by the University of Washington Population Health Initiative, the National Institutes of Health, and philanthropic gift funds. One coauthor of the Washington study is on a Pfizer and GlaxoSmithKline advisory board for immunizations. No other authors or individuals interviewed reported any disclosures.
Pregnant patients with COVID-19 infections were more likely to experience severe disease if they had preexisting comorbidities, such as chronic hypertension, asthma, or pregestational diabetes, according to findings from a new study presented at the meeting sponsored by the Society for Maternal-Fetal Medicine.
The study included outcomes for the largest multistate cohort of pregnant patients with COVID-19 outside of what the Centers for Disease Control and Prevention is tracking. Its findings also mirrored those of a multicenter, retrospective study in Washington state, published in the American Journal of Obstetrics & Gynecology. That study also found that pregnant patients hospitalized for COVID-19 were more likely to have comorbidities, and both studies found an increased likelihood of preterm birth among pregnant patients with severe or critical disease.
Disease severity linked to risk of perinatal complications
In the abstract presented at the SMFM meeting, more severe disease was associated with older age and a higher median body mass index, as seen in the general population, but the researchers found no differences in disease severity occurred by race or ethnicity, Torri D. Metz, MD, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network, told attendees of the conference. The researchers also found that perinatal complications were more prevalent in those with severe or critical COVID-19 disease but not in those with mild or moderate disease. Vertical COVID-19 transmission from mother to child was rare.
The observational study included all patients who had a singleton pregnancy, had a positive SARS-CoV-2 test, and delivered between March 1 and July 31, 2020, at one of the 33 U.S. hospitals in the NICHD Maternal-Fetal Medicine Units Network, spread across 14 states. The researchers used electronic medical records to determine incidence of cesarean delivery, postpartum hemorrhage, hypertensive disorders of pregnancy, preterm birth (less than 37 weeks), maternal death, infant death, and positive infant COVID-19 test. They tracked mothers through 6 weeks post partum and newborns through delivery hospitalization.
Of 1,291 patients in the cohort, 1,219 received their first positive COVID-19 test during pregnancy. The others tested positive while in the hospital for delivery or within a month and a half after discharge. Limiting their analysis to those who developed COVID-19 while pregnant prior to delivery, nearly half (47%) were asymptomatic.
The disease was mild in 27%, moderate in 14%, severe in 8%, and critical in 4%. The researchers used the National Institutes of Health classifications for severity and included deaths in the critical group. The most common symptom was a cough, reported by a third of the patients (34%). Four of six maternal deaths that occurred were caused by COVID-19.
Compared with an average age of 28 in those without symptoms, the mean age was 29 in those with mild/moderate disease and 30 in those with severe/critical disease (P = .006). Similarly, the mean BMI was 28.3 in asymptomatic patients, 29 in those with mild/moderate disease, and 32.3 in those with severe/critical disease (P < .001). Despite a diverse cohort – 53% Hispanic, 23% Black, and 15% White – the researches found no racial/ethnic trends in disease severity.
Patients who had asthma, chronic obstructive pulmonary disorder, pregestational diabetes, chronic hypertension, chronic liver disease, or a seizure disorder were all significantly more likely to have critical/severe disease than mild/moderate disease, and more likely to have mild/moderate disease than asymptomatic (P values ranged from < .001 to .02).
The mothers with critical or severe illness were 1.6 times more likely to have cesarean births and to have hypertensive disorders of pregnancy, and they were twice as likely to have postpartum hemorrhage (P < .001; P = .007). Those with mild or moderate disease, however, had no increased risks for perinatal complications over asymptomatic patients.
Critical or severe illness was also associated with more than triple the risk of preterm birth (adjusted risk ratio, 3.6; P < .001). Newborns of mothers with critical or severe illness also had three times greater risk of neonatal ICU admission (ARR, 3.1; P <. 001) and weighed an average 385 g less than newborns of asymptomatic mothers. COVID-19 rate among infants was only 1% during delivery hospitalization.
Since the study cutoff was July 30 and COVID infections only became prevalent in March, the researchers were unable to evaluate women for outcomes resulting from COVID infections in early pregnancy, such as congenital anomalies or early miscarriage, Dr. Metz said. In addition, since many of the sites are urban centers, the data may not be generalizable to rural areas.
Peter S. Bernstein, MD, MPH, of Montefiore Medical Center, New York, asked whether the increased cesarean deliveries and preterm births in the group of women with severe disease were caused by usual obstetric causes or the treatment of COVID-19 infection. Dr. Metz said the vast majority of preterm deliveries were indicated, but only a small proportion were induced for COVID-19 alone. “A lot had hypertensive disorders of pregnancies or PPROM, so it’s partly driven by the infection itself but also partly driven by some of those perinatal complications,” she said.
Similar findings in Washington
In the Washington study, among 240 pregnant patients with confirmed COVID-19 infection between March 1 and July 30, 2020, 1 in 11 developed severe or critical disease, and 1 in 10 were hospitalized. The pregnant patients had more than triple the risk of hospitalization compared with adults of similar ages in the general population (10% vs. 2.8%; rate ratio, 3.5). Similar to the multistate NICHD study, women were more likely to be hospitalized if they had asthma, hypertension, type 2 diabetes, autoimmune disease, or class III obesity.
Three mothers died of COVID-19, resulting in a case fatality rate 13.6 times greater than nonpregnant patients with COVID-19 in the general population. The absolute difference in the rate was 1.2%. As seen in the NICHD study, preterm birth was more common in mothers with severe or critical COVID-19. Nearly half (45.4%) of mothers with severe or critical COVID-19 delivered preterm compared to 5.2% in those with mild COVID-19 (P < .001).
“Our finding that deaths in pregnant patients contributed disproportionately to deaths from COVID-19 among 20- to 39-year-olds in Washington state is similar to what was observed during the influenza A virus H1N1 2009 pandemic,” Erica M. Lokken, PhD, MS, of the departments of global health and ob.gyn. at the University of Washington, Seattle, and colleagues wrote in the Washington study. But they noted that it took 8 months into the pandemic before pregnant patients were identified as a high-risk group for COVID-19.
“Given the similarity in clinical course between COVID-19 and IAV H1N1 2009 with an increased risk for mortality during pregnancy and the postpartum period, we strongly recommend that pregnant patients should be considered a high-risk population to novel highly pathogenic respiratory viruses until proven otherwise by population-based studies with good ascertainment of pregnancy status,” they wrote.
Judette Louis, MD, MPH, associate professor of ob.gyn. and department chair at the University of South Florida, Tampa, said in an interview that the findings in these studies were fairly expected, but it’s important to have data from such a large cohort as the one presented at SMFM.
“It confirmed that those who had severe disease were more likely to have chronic medical conditions, mirroring what we saw in the general population who isn’t pregnant,” Dr. Louis said. “I thought this was very crucial because as pregnant women are trying to decide whether they should get the COVID vaccine, this provides support to say that if you’re pregnant, you’re more likely to have severe disease [if you have] other chronic medical conditions.”
The findings also confirm the importance of pregnant people taking precautions to avoid infection.
“Even though these individuals are, as a group, in an age cohort that mostly has asymptomatic disease, for some of them, it results in severe disease and even maternal death,” she said. “They should still take it seriously if they’re pregnant.”
The SMFM abstract study was funded by the NICHD. The Washington study was funded by the University of Washington Population Health Initiative, the National Institutes of Health, and philanthropic gift funds. One coauthor of the Washington study is on a Pfizer and GlaxoSmithKline advisory board for immunizations. No other authors or individuals interviewed reported any disclosures.
Pregnant patients with COVID-19 infections were more likely to experience severe disease if they had preexisting comorbidities, such as chronic hypertension, asthma, or pregestational diabetes, according to findings from a new study presented at the meeting sponsored by the Society for Maternal-Fetal Medicine.
The study included outcomes for the largest multistate cohort of pregnant patients with COVID-19 outside of what the Centers for Disease Control and Prevention is tracking. Its findings also mirrored those of a multicenter, retrospective study in Washington state, published in the American Journal of Obstetrics & Gynecology. That study also found that pregnant patients hospitalized for COVID-19 were more likely to have comorbidities, and both studies found an increased likelihood of preterm birth among pregnant patients with severe or critical disease.
Disease severity linked to risk of perinatal complications
In the abstract presented at the SMFM meeting, more severe disease was associated with older age and a higher median body mass index, as seen in the general population, but the researchers found no differences in disease severity occurred by race or ethnicity, Torri D. Metz, MD, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network, told attendees of the conference. The researchers also found that perinatal complications were more prevalent in those with severe or critical COVID-19 disease but not in those with mild or moderate disease. Vertical COVID-19 transmission from mother to child was rare.
The observational study included all patients who had a singleton pregnancy, had a positive SARS-CoV-2 test, and delivered between March 1 and July 31, 2020, at one of the 33 U.S. hospitals in the NICHD Maternal-Fetal Medicine Units Network, spread across 14 states. The researchers used electronic medical records to determine incidence of cesarean delivery, postpartum hemorrhage, hypertensive disorders of pregnancy, preterm birth (less than 37 weeks), maternal death, infant death, and positive infant COVID-19 test. They tracked mothers through 6 weeks post partum and newborns through delivery hospitalization.
Of 1,291 patients in the cohort, 1,219 received their first positive COVID-19 test during pregnancy. The others tested positive while in the hospital for delivery or within a month and a half after discharge. Limiting their analysis to those who developed COVID-19 while pregnant prior to delivery, nearly half (47%) were asymptomatic.
The disease was mild in 27%, moderate in 14%, severe in 8%, and critical in 4%. The researchers used the National Institutes of Health classifications for severity and included deaths in the critical group. The most common symptom was a cough, reported by a third of the patients (34%). Four of six maternal deaths that occurred were caused by COVID-19.
Compared with an average age of 28 in those without symptoms, the mean age was 29 in those with mild/moderate disease and 30 in those with severe/critical disease (P = .006). Similarly, the mean BMI was 28.3 in asymptomatic patients, 29 in those with mild/moderate disease, and 32.3 in those with severe/critical disease (P < .001). Despite a diverse cohort – 53% Hispanic, 23% Black, and 15% White – the researches found no racial/ethnic trends in disease severity.
Patients who had asthma, chronic obstructive pulmonary disorder, pregestational diabetes, chronic hypertension, chronic liver disease, or a seizure disorder were all significantly more likely to have critical/severe disease than mild/moderate disease, and more likely to have mild/moderate disease than asymptomatic (P values ranged from < .001 to .02).
The mothers with critical or severe illness were 1.6 times more likely to have cesarean births and to have hypertensive disorders of pregnancy, and they were twice as likely to have postpartum hemorrhage (P < .001; P = .007). Those with mild or moderate disease, however, had no increased risks for perinatal complications over asymptomatic patients.
Critical or severe illness was also associated with more than triple the risk of preterm birth (adjusted risk ratio, 3.6; P < .001). Newborns of mothers with critical or severe illness also had three times greater risk of neonatal ICU admission (ARR, 3.1; P <. 001) and weighed an average 385 g less than newborns of asymptomatic mothers. COVID-19 rate among infants was only 1% during delivery hospitalization.
Since the study cutoff was July 30 and COVID infections only became prevalent in March, the researchers were unable to evaluate women for outcomes resulting from COVID infections in early pregnancy, such as congenital anomalies or early miscarriage, Dr. Metz said. In addition, since many of the sites are urban centers, the data may not be generalizable to rural areas.
Peter S. Bernstein, MD, MPH, of Montefiore Medical Center, New York, asked whether the increased cesarean deliveries and preterm births in the group of women with severe disease were caused by usual obstetric causes or the treatment of COVID-19 infection. Dr. Metz said the vast majority of preterm deliveries were indicated, but only a small proportion were induced for COVID-19 alone. “A lot had hypertensive disorders of pregnancies or PPROM, so it’s partly driven by the infection itself but also partly driven by some of those perinatal complications,” she said.
Similar findings in Washington
In the Washington study, among 240 pregnant patients with confirmed COVID-19 infection between March 1 and July 30, 2020, 1 in 11 developed severe or critical disease, and 1 in 10 were hospitalized. The pregnant patients had more than triple the risk of hospitalization compared with adults of similar ages in the general population (10% vs. 2.8%; rate ratio, 3.5). Similar to the multistate NICHD study, women were more likely to be hospitalized if they had asthma, hypertension, type 2 diabetes, autoimmune disease, or class III obesity.
Three mothers died of COVID-19, resulting in a case fatality rate 13.6 times greater than nonpregnant patients with COVID-19 in the general population. The absolute difference in the rate was 1.2%. As seen in the NICHD study, preterm birth was more common in mothers with severe or critical COVID-19. Nearly half (45.4%) of mothers with severe or critical COVID-19 delivered preterm compared to 5.2% in those with mild COVID-19 (P < .001).
“Our finding that deaths in pregnant patients contributed disproportionately to deaths from COVID-19 among 20- to 39-year-olds in Washington state is similar to what was observed during the influenza A virus H1N1 2009 pandemic,” Erica M. Lokken, PhD, MS, of the departments of global health and ob.gyn. at the University of Washington, Seattle, and colleagues wrote in the Washington study. But they noted that it took 8 months into the pandemic before pregnant patients were identified as a high-risk group for COVID-19.
“Given the similarity in clinical course between COVID-19 and IAV H1N1 2009 with an increased risk for mortality during pregnancy and the postpartum period, we strongly recommend that pregnant patients should be considered a high-risk population to novel highly pathogenic respiratory viruses until proven otherwise by population-based studies with good ascertainment of pregnancy status,” they wrote.
Judette Louis, MD, MPH, associate professor of ob.gyn. and department chair at the University of South Florida, Tampa, said in an interview that the findings in these studies were fairly expected, but it’s important to have data from such a large cohort as the one presented at SMFM.
“It confirmed that those who had severe disease were more likely to have chronic medical conditions, mirroring what we saw in the general population who isn’t pregnant,” Dr. Louis said. “I thought this was very crucial because as pregnant women are trying to decide whether they should get the COVID vaccine, this provides support to say that if you’re pregnant, you’re more likely to have severe disease [if you have] other chronic medical conditions.”
The findings also confirm the importance of pregnant people taking precautions to avoid infection.
“Even though these individuals are, as a group, in an age cohort that mostly has asymptomatic disease, for some of them, it results in severe disease and even maternal death,” she said. “They should still take it seriously if they’re pregnant.”
The SMFM abstract study was funded by the NICHD. The Washington study was funded by the University of Washington Population Health Initiative, the National Institutes of Health, and philanthropic gift funds. One coauthor of the Washington study is on a Pfizer and GlaxoSmithKline advisory board for immunizations. No other authors or individuals interviewed reported any disclosures.
FROM THE PREGNANCY MEETING
COVID-related immunization gaps portend return of preventable infections
Because of significant reduction in delivery of recommended childhood immunization during the pandemic, there is a risk for resurgence of vaccine preventable infections, including measles, pertussis, and polio, which can result in significant morbidity and mortality in children, reported Amy G. Feldman, MD, of Children’s Hospital Colorado, Aurora, and associates.
Will loss of herd immunity lead to vaccine deserts?
When asked to comment, pediatric infectious disease specialist Christopher J. Harrison, MD, said, “My concern is that we may see expansion of what I call ‘vaccine deserts.’ Vaccine deserts occur in underserved communities, areas with pockets of vaccine-hesitant families or among selected groups with difficult access to health care. These vaccine deserts have held a higher density of vulnerables due to low vaccine uptake, often giving rise to outbreaks of vaccine-preventable diseases, e.g., measles, mumps, pertussis. They are usually due to an index case arriving from another vaccine desert (a developing country or a developed country, U.S. or foreign) where the disease is still endemic or pockets of vaccine hesitancy/refusal exist. When detected, local outbreaks result in rapid responses from public/private health collaborations that limit the outbreak. But what if vaccine deserts became more generalized in the U.S. because of loss of vaccine-induced herd immunity in many more or larger areas of our communities because of pandemic-driven lack of vaccinations? That pandemic-driven indirect damage would further stress the health care system and the economy. And it may first show up in the older children whose vaccines were deferred in the first 4-6 months of the pandemic.”
Dr. Feldman and associates cited findings from a collaborative survey conducted by UNICEF, the World Health Organization, Gavi the Vaccine Alliance, the CDC, the Sabin Vaccine Institute, and the Johns Hopkins Bloomberg School of Public Health, which found that immunization programs experienced moderate to severe disruptions or terminations in at least 68 of 129 low and middle-income countries surveyed. According to the WHO, CDC, Red Cross, and GAVI, 94 million people presently are estimated to be at risk as a consequence of not receiving their measles vaccines following the suspensions.
“These national and international declines in routine immunizations have placed the global community at significant risk for outbreaks of vaccine-preventable infections (VPIs) including measles, polio, and pertussis, diseases which are more deadly, more contagious and have a higher reproductive factor (R0) amongst children than COVID-19,” the authors observed.
Dr. Feldman and associates outlined the horrible devastation that these VPI can cause in children, including significantly higher morbidity and mortality than adults, especially among those with immunodeficiencies. Neurologic deficits, paralysis, intellectual disabilities, and vision and hearing loss are just some of the permanent effects conveyed. “It is concerning to imagine how measles could spread across the United States when social distancing restriction[s] are relaxed and unvaccinated children return to school and usual community engagement,” they noted.
Collaborative engagement key to course correction
The authors found that primary care providers and public health communities are working not only to restore vaccine administration but also to restore confidence that vaccine delivery is safe in spite of COVID. In addition to recommending specific risk mitigation strategies for clinicians, they also suggested individual practitioners use electronic health records to identify patients with COVID-related lapses in vaccination, employ electronic health record–based parent notification of overdue immunizations, and offer distance-friendly vaccination options that include parking lot or drive-up window vaccine delivery.
Additionally, Dr. Feldman and colleagues recommended that local, state, regional, and national health systems use public service announcements via television and digital as well as social media platforms to convey important messages about the considerable health risks associated with vaccine avoidance and the availability of free or reduced-cost vaccination programs through the federally funded Vaccines For Children program for parents out of work or without insurance. Equally important is messaging around encouraging vaccine opportunities during all health care visits, whether they be subspecialty, urgent care, emergency room, or inpatient visits. In areas where access to clinics is limited, they urged the use of mobile clinics as well as additional focus on providing medical homes to children with poor access to care.
“A partial but expanding safety net may be developing spontaneously, i.e., practices and clinics based on a patient-centered medical home (PCMH) model,” noted Dr. Harrison, professor of pediatrics, University of Missouri-Kansas City, in an interview. “When lagging vaccinations were reported in mid-2020, we checked with a local hospital–based urban clinic and two suburban private practices modeled on PCMH. Each had noted a drastic drop in well checks in the first months of the pandemic. But with ill visits nearly nonexistent, they doubled down on maintaining health maintenance visits. Even though staff and provider work hours were limited, and families were less enthusiastic about well checks, momentum appears to have grown so that, by later in 2020, vaccine uptake rates were again comparable to 2019. So, some already seem to have answered the call, but practices/clinics remain hampered by months of reduced revenue needed to support staff, providers, PPE supplies, and added infection control needs,” he said.The study was funded by the Agency for Healthcare Research Quality. Dr. Isakov disclosed relationships with various pharmaceutical companies outside the submitted work. The other authors had no relevant disclosures. Dr. Harrison’s institution receives grant funding from GSK, Merck, and Pfizer for pediatric vaccine trials and pneumococcal seroprevalence studies on which he is an investigator.
Because of significant reduction in delivery of recommended childhood immunization during the pandemic, there is a risk for resurgence of vaccine preventable infections, including measles, pertussis, and polio, which can result in significant morbidity and mortality in children, reported Amy G. Feldman, MD, of Children’s Hospital Colorado, Aurora, and associates.
Will loss of herd immunity lead to vaccine deserts?
When asked to comment, pediatric infectious disease specialist Christopher J. Harrison, MD, said, “My concern is that we may see expansion of what I call ‘vaccine deserts.’ Vaccine deserts occur in underserved communities, areas with pockets of vaccine-hesitant families or among selected groups with difficult access to health care. These vaccine deserts have held a higher density of vulnerables due to low vaccine uptake, often giving rise to outbreaks of vaccine-preventable diseases, e.g., measles, mumps, pertussis. They are usually due to an index case arriving from another vaccine desert (a developing country or a developed country, U.S. or foreign) where the disease is still endemic or pockets of vaccine hesitancy/refusal exist. When detected, local outbreaks result in rapid responses from public/private health collaborations that limit the outbreak. But what if vaccine deserts became more generalized in the U.S. because of loss of vaccine-induced herd immunity in many more or larger areas of our communities because of pandemic-driven lack of vaccinations? That pandemic-driven indirect damage would further stress the health care system and the economy. And it may first show up in the older children whose vaccines were deferred in the first 4-6 months of the pandemic.”
Dr. Feldman and associates cited findings from a collaborative survey conducted by UNICEF, the World Health Organization, Gavi the Vaccine Alliance, the CDC, the Sabin Vaccine Institute, and the Johns Hopkins Bloomberg School of Public Health, which found that immunization programs experienced moderate to severe disruptions or terminations in at least 68 of 129 low and middle-income countries surveyed. According to the WHO, CDC, Red Cross, and GAVI, 94 million people presently are estimated to be at risk as a consequence of not receiving their measles vaccines following the suspensions.
“These national and international declines in routine immunizations have placed the global community at significant risk for outbreaks of vaccine-preventable infections (VPIs) including measles, polio, and pertussis, diseases which are more deadly, more contagious and have a higher reproductive factor (R0) amongst children than COVID-19,” the authors observed.
Dr. Feldman and associates outlined the horrible devastation that these VPI can cause in children, including significantly higher morbidity and mortality than adults, especially among those with immunodeficiencies. Neurologic deficits, paralysis, intellectual disabilities, and vision and hearing loss are just some of the permanent effects conveyed. “It is concerning to imagine how measles could spread across the United States when social distancing restriction[s] are relaxed and unvaccinated children return to school and usual community engagement,” they noted.
Collaborative engagement key to course correction
The authors found that primary care providers and public health communities are working not only to restore vaccine administration but also to restore confidence that vaccine delivery is safe in spite of COVID. In addition to recommending specific risk mitigation strategies for clinicians, they also suggested individual practitioners use electronic health records to identify patients with COVID-related lapses in vaccination, employ electronic health record–based parent notification of overdue immunizations, and offer distance-friendly vaccination options that include parking lot or drive-up window vaccine delivery.
Additionally, Dr. Feldman and colleagues recommended that local, state, regional, and national health systems use public service announcements via television and digital as well as social media platforms to convey important messages about the considerable health risks associated with vaccine avoidance and the availability of free or reduced-cost vaccination programs through the federally funded Vaccines For Children program for parents out of work or without insurance. Equally important is messaging around encouraging vaccine opportunities during all health care visits, whether they be subspecialty, urgent care, emergency room, or inpatient visits. In areas where access to clinics is limited, they urged the use of mobile clinics as well as additional focus on providing medical homes to children with poor access to care.
“A partial but expanding safety net may be developing spontaneously, i.e., practices and clinics based on a patient-centered medical home (PCMH) model,” noted Dr. Harrison, professor of pediatrics, University of Missouri-Kansas City, in an interview. “When lagging vaccinations were reported in mid-2020, we checked with a local hospital–based urban clinic and two suburban private practices modeled on PCMH. Each had noted a drastic drop in well checks in the first months of the pandemic. But with ill visits nearly nonexistent, they doubled down on maintaining health maintenance visits. Even though staff and provider work hours were limited, and families were less enthusiastic about well checks, momentum appears to have grown so that, by later in 2020, vaccine uptake rates were again comparable to 2019. So, some already seem to have answered the call, but practices/clinics remain hampered by months of reduced revenue needed to support staff, providers, PPE supplies, and added infection control needs,” he said.The study was funded by the Agency for Healthcare Research Quality. Dr. Isakov disclosed relationships with various pharmaceutical companies outside the submitted work. The other authors had no relevant disclosures. Dr. Harrison’s institution receives grant funding from GSK, Merck, and Pfizer for pediatric vaccine trials and pneumococcal seroprevalence studies on which he is an investigator.
Because of significant reduction in delivery of recommended childhood immunization during the pandemic, there is a risk for resurgence of vaccine preventable infections, including measles, pertussis, and polio, which can result in significant morbidity and mortality in children, reported Amy G. Feldman, MD, of Children’s Hospital Colorado, Aurora, and associates.
Will loss of herd immunity lead to vaccine deserts?
When asked to comment, pediatric infectious disease specialist Christopher J. Harrison, MD, said, “My concern is that we may see expansion of what I call ‘vaccine deserts.’ Vaccine deserts occur in underserved communities, areas with pockets of vaccine-hesitant families or among selected groups with difficult access to health care. These vaccine deserts have held a higher density of vulnerables due to low vaccine uptake, often giving rise to outbreaks of vaccine-preventable diseases, e.g., measles, mumps, pertussis. They are usually due to an index case arriving from another vaccine desert (a developing country or a developed country, U.S. or foreign) where the disease is still endemic or pockets of vaccine hesitancy/refusal exist. When detected, local outbreaks result in rapid responses from public/private health collaborations that limit the outbreak. But what if vaccine deserts became more generalized in the U.S. because of loss of vaccine-induced herd immunity in many more or larger areas of our communities because of pandemic-driven lack of vaccinations? That pandemic-driven indirect damage would further stress the health care system and the economy. And it may first show up in the older children whose vaccines were deferred in the first 4-6 months of the pandemic.”
Dr. Feldman and associates cited findings from a collaborative survey conducted by UNICEF, the World Health Organization, Gavi the Vaccine Alliance, the CDC, the Sabin Vaccine Institute, and the Johns Hopkins Bloomberg School of Public Health, which found that immunization programs experienced moderate to severe disruptions or terminations in at least 68 of 129 low and middle-income countries surveyed. According to the WHO, CDC, Red Cross, and GAVI, 94 million people presently are estimated to be at risk as a consequence of not receiving their measles vaccines following the suspensions.
“These national and international declines in routine immunizations have placed the global community at significant risk for outbreaks of vaccine-preventable infections (VPIs) including measles, polio, and pertussis, diseases which are more deadly, more contagious and have a higher reproductive factor (R0) amongst children than COVID-19,” the authors observed.
Dr. Feldman and associates outlined the horrible devastation that these VPI can cause in children, including significantly higher morbidity and mortality than adults, especially among those with immunodeficiencies. Neurologic deficits, paralysis, intellectual disabilities, and vision and hearing loss are just some of the permanent effects conveyed. “It is concerning to imagine how measles could spread across the United States when social distancing restriction[s] are relaxed and unvaccinated children return to school and usual community engagement,” they noted.
Collaborative engagement key to course correction
The authors found that primary care providers and public health communities are working not only to restore vaccine administration but also to restore confidence that vaccine delivery is safe in spite of COVID. In addition to recommending specific risk mitigation strategies for clinicians, they also suggested individual practitioners use electronic health records to identify patients with COVID-related lapses in vaccination, employ electronic health record–based parent notification of overdue immunizations, and offer distance-friendly vaccination options that include parking lot or drive-up window vaccine delivery.
Additionally, Dr. Feldman and colleagues recommended that local, state, regional, and national health systems use public service announcements via television and digital as well as social media platforms to convey important messages about the considerable health risks associated with vaccine avoidance and the availability of free or reduced-cost vaccination programs through the federally funded Vaccines For Children program for parents out of work or without insurance. Equally important is messaging around encouraging vaccine opportunities during all health care visits, whether they be subspecialty, urgent care, emergency room, or inpatient visits. In areas where access to clinics is limited, they urged the use of mobile clinics as well as additional focus on providing medical homes to children with poor access to care.
“A partial but expanding safety net may be developing spontaneously, i.e., practices and clinics based on a patient-centered medical home (PCMH) model,” noted Dr. Harrison, professor of pediatrics, University of Missouri-Kansas City, in an interview. “When lagging vaccinations were reported in mid-2020, we checked with a local hospital–based urban clinic and two suburban private practices modeled on PCMH. Each had noted a drastic drop in well checks in the first months of the pandemic. But with ill visits nearly nonexistent, they doubled down on maintaining health maintenance visits. Even though staff and provider work hours were limited, and families were less enthusiastic about well checks, momentum appears to have grown so that, by later in 2020, vaccine uptake rates were again comparable to 2019. So, some already seem to have answered the call, but practices/clinics remain hampered by months of reduced revenue needed to support staff, providers, PPE supplies, and added infection control needs,” he said.The study was funded by the Agency for Healthcare Research Quality. Dr. Isakov disclosed relationships with various pharmaceutical companies outside the submitted work. The other authors had no relevant disclosures. Dr. Harrison’s institution receives grant funding from GSK, Merck, and Pfizer for pediatric vaccine trials and pneumococcal seroprevalence studies on which he is an investigator.
FROM CLINICAL INFECTIOUS DISEASES
Doxorubicin-pomalidomide combo shows promise for Kaposi sarcoma
Liposomal doxorubicin (Dox) plus pomalidomide (Pom) was safe and active in heavily pretreated patients with Kaposi sarcoma, according to results from a phase 1/2 trial.
“The results of our phase 1/2 study suggest pomalidomide and liposomal doxorubicin is safe with evidence of activity among patients with Kaposi sarcoma,” said investigator Ramya Ramaswami, MBBS, MPH, of the HIV & AIDS malignancy branch at the National Cancer Institute. The results were presented at the Conference on Retroviruses and Opportunistic Infections.
The researchers evaluated the safety and tolerability of Pom/Dox in two groups of patients with Kaposi sarcoma: group 1 included patients with Kaposi sarcoma alone and group 2 included patients with Kaposi sarcoma–associated herpesvirus and concurrent multicentric Castleman disease (KSHV-MCD) and KSHV inflammatory cytokine syndrome (KICS).
“Kaposi sarcoma can be challenging to treat when it co-occurs with KSHV-MCD or KICS, resulting in high mortality rates,” Dr. Ramaswami explained.
Study participants received IV liposomal Dox at 20 mg/m2 on day 1 of a 28-day cycle, in addition to oral Pom once daily on days 1-21 at three escalating dose levels (2 mg, 3 mg, or 4 mg, respectively) using a standard 3 + 3 design until plateau of response, progression, dose-limiting toxicities (DLTs) or patient preference. Some eligibility criteria differed between groups 1 and 2. Participants in group 1 were required to be on antiretroviral therapy for at least 1 month and had a performance status of 2 or less, while those in group 2 had a performance status of 3 or less and could be antiretroviral therapy naive.
All participants received oral aspirin thromboprophylaxis (81 mg daily) and could have received prior Kaposi sarcoma therapy.
With respect to outcomes, Kaposi sarcoma responses were assessed using the modified AIDS Clinical Trial Group criteria and KICS and KSHV-MCD responses were evaluated using an NCI clinical benefit criteria.
Results
Overall, 34 cisgender men were enrolled in the study: 21 (62%) in group 1 and 13 (38%) in group 2. All participants had severe (T1) Kaposi sarcoma; 32 (94%) participants were HIV-infected and 22 (65%) had prior chemotherapy for Kaposi sarcoma.
While the HIV viral load was largely controlled in both groups, the CD4 count differed, with median CD4 counts of 286 and 92 cells/mcL in groups 1 and 2, respectively.
With respect to safety, no DLTs were observed in group 1. As a result, 12 participants were treated at the maximum tolerated dose (MTD) of 4 mg of Pom. However, two DLTs (grade 3 rash and pharyngeal edema) were observed in group 2 at the 3 mg dose level.
A median of six cycles were administered for all participants and the most common grade 3/4 toxicity was neutropenia; nine patients with grade 3 neutropenia required dose reduction and three patients had febrile neutropenia requiring hospitalization. Other Pom-related adverse events were rash, constipation, and fatigue.
Among evaluable participants receiving two or more cycles, 17 (81%) patients in group 1 had a response (95% confidence interval, 58-95%; 16 partial response and 1 complete response) and 5 (50%) patients in group 2 had a response (95% CI, 19-81%; 4 PR and 1 CR).
“Our waterfall plots indicated that the vast majority of patients in group 1 had a positive change in nodular lesions at baseline,” Dr. Ramaswami said. “Participants in group 2 showed some decrease in nodular lesions, but this was usually temporary.”
Among seven participants with KICS responses, four participants (57%) experienced a CR or PR in symptoms and lab abnormalities associated with KICS; three of six participants (50%) with KSHV-MCD responses experienced a PR as per response criteria.
“While activity was noted, the combination was less well tolerated in patients with Kaposi sarcoma and concurrent KSHV-MCD or KICS,” Dr. Ramaswami said.
During a live discussion, Ronald T. Mitsuyasu, MD, of the University of California, Los Angeles, asked Dr. Ramaswami about the use of liposomal doxorubicin alone in patients with Kaposi sarcoma and concurrent KSHV-MCD or KICS.
While there is currently no data on the use of doxorubicin alone in this population, Dr. Ramaswami noted that she was more confident administering Pom/Dox combination therapy for these patients.
Dr. Ramaswami disclosed financial relationships with the National Cancer Institute, Celgene/Bristol-Myers Squibb, EMD Serono, Merck, CTI Biopharma, and Janssen. The study was funded by a cooperative research and drug development agreement between the National Cancer Institute and Celgene/BMS, EMD Serono, Merck, CTI Biopharma, and Janssen.
Liposomal doxorubicin (Dox) plus pomalidomide (Pom) was safe and active in heavily pretreated patients with Kaposi sarcoma, according to results from a phase 1/2 trial.
“The results of our phase 1/2 study suggest pomalidomide and liposomal doxorubicin is safe with evidence of activity among patients with Kaposi sarcoma,” said investigator Ramya Ramaswami, MBBS, MPH, of the HIV & AIDS malignancy branch at the National Cancer Institute. The results were presented at the Conference on Retroviruses and Opportunistic Infections.
The researchers evaluated the safety and tolerability of Pom/Dox in two groups of patients with Kaposi sarcoma: group 1 included patients with Kaposi sarcoma alone and group 2 included patients with Kaposi sarcoma–associated herpesvirus and concurrent multicentric Castleman disease (KSHV-MCD) and KSHV inflammatory cytokine syndrome (KICS).
“Kaposi sarcoma can be challenging to treat when it co-occurs with KSHV-MCD or KICS, resulting in high mortality rates,” Dr. Ramaswami explained.
Study participants received IV liposomal Dox at 20 mg/m2 on day 1 of a 28-day cycle, in addition to oral Pom once daily on days 1-21 at three escalating dose levels (2 mg, 3 mg, or 4 mg, respectively) using a standard 3 + 3 design until plateau of response, progression, dose-limiting toxicities (DLTs) or patient preference. Some eligibility criteria differed between groups 1 and 2. Participants in group 1 were required to be on antiretroviral therapy for at least 1 month and had a performance status of 2 or less, while those in group 2 had a performance status of 3 or less and could be antiretroviral therapy naive.
All participants received oral aspirin thromboprophylaxis (81 mg daily) and could have received prior Kaposi sarcoma therapy.
With respect to outcomes, Kaposi sarcoma responses were assessed using the modified AIDS Clinical Trial Group criteria and KICS and KSHV-MCD responses were evaluated using an NCI clinical benefit criteria.
Results
Overall, 34 cisgender men were enrolled in the study: 21 (62%) in group 1 and 13 (38%) in group 2. All participants had severe (T1) Kaposi sarcoma; 32 (94%) participants were HIV-infected and 22 (65%) had prior chemotherapy for Kaposi sarcoma.
While the HIV viral load was largely controlled in both groups, the CD4 count differed, with median CD4 counts of 286 and 92 cells/mcL in groups 1 and 2, respectively.
With respect to safety, no DLTs were observed in group 1. As a result, 12 participants were treated at the maximum tolerated dose (MTD) of 4 mg of Pom. However, two DLTs (grade 3 rash and pharyngeal edema) were observed in group 2 at the 3 mg dose level.
A median of six cycles were administered for all participants and the most common grade 3/4 toxicity was neutropenia; nine patients with grade 3 neutropenia required dose reduction and three patients had febrile neutropenia requiring hospitalization. Other Pom-related adverse events were rash, constipation, and fatigue.
Among evaluable participants receiving two or more cycles, 17 (81%) patients in group 1 had a response (95% confidence interval, 58-95%; 16 partial response and 1 complete response) and 5 (50%) patients in group 2 had a response (95% CI, 19-81%; 4 PR and 1 CR).
“Our waterfall plots indicated that the vast majority of patients in group 1 had a positive change in nodular lesions at baseline,” Dr. Ramaswami said. “Participants in group 2 showed some decrease in nodular lesions, but this was usually temporary.”
Among seven participants with KICS responses, four participants (57%) experienced a CR or PR in symptoms and lab abnormalities associated with KICS; three of six participants (50%) with KSHV-MCD responses experienced a PR as per response criteria.
“While activity was noted, the combination was less well tolerated in patients with Kaposi sarcoma and concurrent KSHV-MCD or KICS,” Dr. Ramaswami said.
During a live discussion, Ronald T. Mitsuyasu, MD, of the University of California, Los Angeles, asked Dr. Ramaswami about the use of liposomal doxorubicin alone in patients with Kaposi sarcoma and concurrent KSHV-MCD or KICS.
While there is currently no data on the use of doxorubicin alone in this population, Dr. Ramaswami noted that she was more confident administering Pom/Dox combination therapy for these patients.
Dr. Ramaswami disclosed financial relationships with the National Cancer Institute, Celgene/Bristol-Myers Squibb, EMD Serono, Merck, CTI Biopharma, and Janssen. The study was funded by a cooperative research and drug development agreement between the National Cancer Institute and Celgene/BMS, EMD Serono, Merck, CTI Biopharma, and Janssen.
Liposomal doxorubicin (Dox) plus pomalidomide (Pom) was safe and active in heavily pretreated patients with Kaposi sarcoma, according to results from a phase 1/2 trial.
“The results of our phase 1/2 study suggest pomalidomide and liposomal doxorubicin is safe with evidence of activity among patients with Kaposi sarcoma,” said investigator Ramya Ramaswami, MBBS, MPH, of the HIV & AIDS malignancy branch at the National Cancer Institute. The results were presented at the Conference on Retroviruses and Opportunistic Infections.
The researchers evaluated the safety and tolerability of Pom/Dox in two groups of patients with Kaposi sarcoma: group 1 included patients with Kaposi sarcoma alone and group 2 included patients with Kaposi sarcoma–associated herpesvirus and concurrent multicentric Castleman disease (KSHV-MCD) and KSHV inflammatory cytokine syndrome (KICS).
“Kaposi sarcoma can be challenging to treat when it co-occurs with KSHV-MCD or KICS, resulting in high mortality rates,” Dr. Ramaswami explained.
Study participants received IV liposomal Dox at 20 mg/m2 on day 1 of a 28-day cycle, in addition to oral Pom once daily on days 1-21 at three escalating dose levels (2 mg, 3 mg, or 4 mg, respectively) using a standard 3 + 3 design until plateau of response, progression, dose-limiting toxicities (DLTs) or patient preference. Some eligibility criteria differed between groups 1 and 2. Participants in group 1 were required to be on antiretroviral therapy for at least 1 month and had a performance status of 2 or less, while those in group 2 had a performance status of 3 or less and could be antiretroviral therapy naive.
All participants received oral aspirin thromboprophylaxis (81 mg daily) and could have received prior Kaposi sarcoma therapy.
With respect to outcomes, Kaposi sarcoma responses were assessed using the modified AIDS Clinical Trial Group criteria and KICS and KSHV-MCD responses were evaluated using an NCI clinical benefit criteria.
Results
Overall, 34 cisgender men were enrolled in the study: 21 (62%) in group 1 and 13 (38%) in group 2. All participants had severe (T1) Kaposi sarcoma; 32 (94%) participants were HIV-infected and 22 (65%) had prior chemotherapy for Kaposi sarcoma.
While the HIV viral load was largely controlled in both groups, the CD4 count differed, with median CD4 counts of 286 and 92 cells/mcL in groups 1 and 2, respectively.
With respect to safety, no DLTs were observed in group 1. As a result, 12 participants were treated at the maximum tolerated dose (MTD) of 4 mg of Pom. However, two DLTs (grade 3 rash and pharyngeal edema) were observed in group 2 at the 3 mg dose level.
A median of six cycles were administered for all participants and the most common grade 3/4 toxicity was neutropenia; nine patients with grade 3 neutropenia required dose reduction and three patients had febrile neutropenia requiring hospitalization. Other Pom-related adverse events were rash, constipation, and fatigue.
Among evaluable participants receiving two or more cycles, 17 (81%) patients in group 1 had a response (95% confidence interval, 58-95%; 16 partial response and 1 complete response) and 5 (50%) patients in group 2 had a response (95% CI, 19-81%; 4 PR and 1 CR).
“Our waterfall plots indicated that the vast majority of patients in group 1 had a positive change in nodular lesions at baseline,” Dr. Ramaswami said. “Participants in group 2 showed some decrease in nodular lesions, but this was usually temporary.”
Among seven participants with KICS responses, four participants (57%) experienced a CR or PR in symptoms and lab abnormalities associated with KICS; three of six participants (50%) with KSHV-MCD responses experienced a PR as per response criteria.
“While activity was noted, the combination was less well tolerated in patients with Kaposi sarcoma and concurrent KSHV-MCD or KICS,” Dr. Ramaswami said.
During a live discussion, Ronald T. Mitsuyasu, MD, of the University of California, Los Angeles, asked Dr. Ramaswami about the use of liposomal doxorubicin alone in patients with Kaposi sarcoma and concurrent KSHV-MCD or KICS.
While there is currently no data on the use of doxorubicin alone in this population, Dr. Ramaswami noted that she was more confident administering Pom/Dox combination therapy for these patients.
Dr. Ramaswami disclosed financial relationships with the National Cancer Institute, Celgene/Bristol-Myers Squibb, EMD Serono, Merck, CTI Biopharma, and Janssen. The study was funded by a cooperative research and drug development agreement between the National Cancer Institute and Celgene/BMS, EMD Serono, Merck, CTI Biopharma, and Janssen.
FROM CROI 2021
Vaginal pH may predict CIN 2 progression in HIV-positive women
Elevated vaginal pH at the time of cervical intraepithelial neoplasia 2 diagnosis may be a useful marker of CIN 2 persistence/progression, as well as the rate of persistence/progression in HIV-positive women, new research suggests.
“We analyzed data from the Women’s Interagency HIV Study [WIHS], an observational, longitudinal cohort of women with and without HIV to determine factors that may influence CIN 2 natural history,” said Kate Michel, PhD, MPH, of Georgetown University, Washington. She presented the results at the Conference on Retroviruses and Opportunistic Infections.
As previous data have shown a high incidence of CIN 2 progression among women with HIV, the researchers evaluated the role of human papillomavirus (HPV) type, local immune response, and markers of the cervicovaginal microbiome on the risk of CIN 2 persistence/progression.
Within the cohort, follow-up visits occur every 6 months, and clinical data is collected via questionnaires, physical and gynecologic exams, and biological samples. As no specific treatment is offered in the WIHS, treatment for cervical abnormalities is abstracted from medical records.
In the present study, Dr. Michel and colleagues selected up to four banked cervicovaginal lavage (CVL) samples per woman, with the first sample selected 6-12 months prior to CIN 2 diagnosis, the second at CIN 2 diagnosis, the third between CIN 2 diagnosis and outcome, and the fourth at the outcome visit.
The investigators performed HPV typing and muiltiplex immune mediator testing on each CVL sample. Lab results from WIHS core testing were also extracted, including plasma CD4+ T-cell count and HIV viral load, as well as vaginal pH and Nugent’s score.
Study outcomes included persistence/progression and regression, defined as a subsequent CIN 2 or CIN 3 diagnosis and subsequent CIN 1 or normal diagnosis, respectively. Logistic regression models were used to determine CIN 2 regression versus persistence/progression.
Results
A total of 337 samples were obtained and 94 women were included in the analysis. Key demographic and behavioral factor were similar at CIN 2 diagnosis.
The majority of participants were African American (53.2%) and on antiretroviral therapy (66.0%). The most prevalent high-risk types were HPV-58 (18.4%) and HPV-16 (17.5%).
After a median 12.5 years of follow-up, 33 participants (35.1%) with incident CIN 2 had a subsequent CIN 2/CIN 3 diagnosis and those who regressed had a higher CD4 T-cell count at CIN 2 diagnosis (P = .02).
Each subsequent high-risk HPV type identified at the pre–CIN 2 visit was associated with higher odds of CIN2 persistence/progression (odds ratio, 2.27; 95% confidence interval, 1.15-4.50).
Bacterial vaginosis (adjusted OR, 5.08; 95% CI, 1.30-19.94) and vaginal pH (aOR, 2.27; 95% CI, 1.15-4.50) at the CIN 2 diagnosis visit were each associated with increased odds of CIN 2 persistence/progression.
Vaginal pH greater than 4.5 at CIN 2 diagnosis was also associated with unadjusted time to CIN 2 persistence/progression (log rank P = .002) and an increased rate of CIN 2 persistence/progression (adjusted hazard ratio, 3.37; 95% CI, 1.26-8.99).
Furthermore, among participants who did not receive CIN 2 treatment, vaginal pH remained associated with greater odds of CIN 2 persistence/progression (OR, 2.46; 95% CI, 1.19-5.13). Cervicovaginal immune mediator levels were not associated with CIN 2 persistence/progression.
“The most striking finding from this work was that vaginal pH was associated with higher odds of, quicker time to, and increased hazard of CIN 2 persistence/progression,” Dr. Michel said. “We postulate this effect is mediated by the cervical microbiome, but more work is needed to establish the exact mechanism.”
“It would be interesting to test whether this association might be explained by different vaginal cleaning techniques, such as douching,” said moderator Ronald T. Mitsuyasu, MD, of the University of California, Los Angeles.
“We’re currently working on an analysis of cervicovaginal bacterial species to explore the microbiome in more detail,” Dr. Michel concluded.
Dr. Michel disclosed no conflicts of interest. The study was supported by multiple sources, including the National Institute of Allergy and Infectious Diseases, the National Cancer Institute, and the Georgetown-Howard Universities Center for Clinical and Translational Science.
Elevated vaginal pH at the time of cervical intraepithelial neoplasia 2 diagnosis may be a useful marker of CIN 2 persistence/progression, as well as the rate of persistence/progression in HIV-positive women, new research suggests.
“We analyzed data from the Women’s Interagency HIV Study [WIHS], an observational, longitudinal cohort of women with and without HIV to determine factors that may influence CIN 2 natural history,” said Kate Michel, PhD, MPH, of Georgetown University, Washington. She presented the results at the Conference on Retroviruses and Opportunistic Infections.
As previous data have shown a high incidence of CIN 2 progression among women with HIV, the researchers evaluated the role of human papillomavirus (HPV) type, local immune response, and markers of the cervicovaginal microbiome on the risk of CIN 2 persistence/progression.
Within the cohort, follow-up visits occur every 6 months, and clinical data is collected via questionnaires, physical and gynecologic exams, and biological samples. As no specific treatment is offered in the WIHS, treatment for cervical abnormalities is abstracted from medical records.
In the present study, Dr. Michel and colleagues selected up to four banked cervicovaginal lavage (CVL) samples per woman, with the first sample selected 6-12 months prior to CIN 2 diagnosis, the second at CIN 2 diagnosis, the third between CIN 2 diagnosis and outcome, and the fourth at the outcome visit.
The investigators performed HPV typing and muiltiplex immune mediator testing on each CVL sample. Lab results from WIHS core testing were also extracted, including plasma CD4+ T-cell count and HIV viral load, as well as vaginal pH and Nugent’s score.
Study outcomes included persistence/progression and regression, defined as a subsequent CIN 2 or CIN 3 diagnosis and subsequent CIN 1 or normal diagnosis, respectively. Logistic regression models were used to determine CIN 2 regression versus persistence/progression.
Results
A total of 337 samples were obtained and 94 women were included in the analysis. Key demographic and behavioral factor were similar at CIN 2 diagnosis.
The majority of participants were African American (53.2%) and on antiretroviral therapy (66.0%). The most prevalent high-risk types were HPV-58 (18.4%) and HPV-16 (17.5%).
After a median 12.5 years of follow-up, 33 participants (35.1%) with incident CIN 2 had a subsequent CIN 2/CIN 3 diagnosis and those who regressed had a higher CD4 T-cell count at CIN 2 diagnosis (P = .02).
Each subsequent high-risk HPV type identified at the pre–CIN 2 visit was associated with higher odds of CIN2 persistence/progression (odds ratio, 2.27; 95% confidence interval, 1.15-4.50).
Bacterial vaginosis (adjusted OR, 5.08; 95% CI, 1.30-19.94) and vaginal pH (aOR, 2.27; 95% CI, 1.15-4.50) at the CIN 2 diagnosis visit were each associated with increased odds of CIN 2 persistence/progression.
Vaginal pH greater than 4.5 at CIN 2 diagnosis was also associated with unadjusted time to CIN 2 persistence/progression (log rank P = .002) and an increased rate of CIN 2 persistence/progression (adjusted hazard ratio, 3.37; 95% CI, 1.26-8.99).
Furthermore, among participants who did not receive CIN 2 treatment, vaginal pH remained associated with greater odds of CIN 2 persistence/progression (OR, 2.46; 95% CI, 1.19-5.13). Cervicovaginal immune mediator levels were not associated with CIN 2 persistence/progression.
“The most striking finding from this work was that vaginal pH was associated with higher odds of, quicker time to, and increased hazard of CIN 2 persistence/progression,” Dr. Michel said. “We postulate this effect is mediated by the cervical microbiome, but more work is needed to establish the exact mechanism.”
“It would be interesting to test whether this association might be explained by different vaginal cleaning techniques, such as douching,” said moderator Ronald T. Mitsuyasu, MD, of the University of California, Los Angeles.
“We’re currently working on an analysis of cervicovaginal bacterial species to explore the microbiome in more detail,” Dr. Michel concluded.
Dr. Michel disclosed no conflicts of interest. The study was supported by multiple sources, including the National Institute of Allergy and Infectious Diseases, the National Cancer Institute, and the Georgetown-Howard Universities Center for Clinical and Translational Science.
Elevated vaginal pH at the time of cervical intraepithelial neoplasia 2 diagnosis may be a useful marker of CIN 2 persistence/progression, as well as the rate of persistence/progression in HIV-positive women, new research suggests.
“We analyzed data from the Women’s Interagency HIV Study [WIHS], an observational, longitudinal cohort of women with and without HIV to determine factors that may influence CIN 2 natural history,” said Kate Michel, PhD, MPH, of Georgetown University, Washington. She presented the results at the Conference on Retroviruses and Opportunistic Infections.
As previous data have shown a high incidence of CIN 2 progression among women with HIV, the researchers evaluated the role of human papillomavirus (HPV) type, local immune response, and markers of the cervicovaginal microbiome on the risk of CIN 2 persistence/progression.
Within the cohort, follow-up visits occur every 6 months, and clinical data is collected via questionnaires, physical and gynecologic exams, and biological samples. As no specific treatment is offered in the WIHS, treatment for cervical abnormalities is abstracted from medical records.
In the present study, Dr. Michel and colleagues selected up to four banked cervicovaginal lavage (CVL) samples per woman, with the first sample selected 6-12 months prior to CIN 2 diagnosis, the second at CIN 2 diagnosis, the third between CIN 2 diagnosis and outcome, and the fourth at the outcome visit.
The investigators performed HPV typing and muiltiplex immune mediator testing on each CVL sample. Lab results from WIHS core testing were also extracted, including plasma CD4+ T-cell count and HIV viral load, as well as vaginal pH and Nugent’s score.
Study outcomes included persistence/progression and regression, defined as a subsequent CIN 2 or CIN 3 diagnosis and subsequent CIN 1 or normal diagnosis, respectively. Logistic regression models were used to determine CIN 2 regression versus persistence/progression.
Results
A total of 337 samples were obtained and 94 women were included in the analysis. Key demographic and behavioral factor were similar at CIN 2 diagnosis.
The majority of participants were African American (53.2%) and on antiretroviral therapy (66.0%). The most prevalent high-risk types were HPV-58 (18.4%) and HPV-16 (17.5%).
After a median 12.5 years of follow-up, 33 participants (35.1%) with incident CIN 2 had a subsequent CIN 2/CIN 3 diagnosis and those who regressed had a higher CD4 T-cell count at CIN 2 diagnosis (P = .02).
Each subsequent high-risk HPV type identified at the pre–CIN 2 visit was associated with higher odds of CIN2 persistence/progression (odds ratio, 2.27; 95% confidence interval, 1.15-4.50).
Bacterial vaginosis (adjusted OR, 5.08; 95% CI, 1.30-19.94) and vaginal pH (aOR, 2.27; 95% CI, 1.15-4.50) at the CIN 2 diagnosis visit were each associated with increased odds of CIN 2 persistence/progression.
Vaginal pH greater than 4.5 at CIN 2 diagnosis was also associated with unadjusted time to CIN 2 persistence/progression (log rank P = .002) and an increased rate of CIN 2 persistence/progression (adjusted hazard ratio, 3.37; 95% CI, 1.26-8.99).
Furthermore, among participants who did not receive CIN 2 treatment, vaginal pH remained associated with greater odds of CIN 2 persistence/progression (OR, 2.46; 95% CI, 1.19-5.13). Cervicovaginal immune mediator levels were not associated with CIN 2 persistence/progression.
“The most striking finding from this work was that vaginal pH was associated with higher odds of, quicker time to, and increased hazard of CIN 2 persistence/progression,” Dr. Michel said. “We postulate this effect is mediated by the cervical microbiome, but more work is needed to establish the exact mechanism.”
“It would be interesting to test whether this association might be explained by different vaginal cleaning techniques, such as douching,” said moderator Ronald T. Mitsuyasu, MD, of the University of California, Los Angeles.
“We’re currently working on an analysis of cervicovaginal bacterial species to explore the microbiome in more detail,” Dr. Michel concluded.
Dr. Michel disclosed no conflicts of interest. The study was supported by multiple sources, including the National Institute of Allergy and Infectious Diseases, the National Cancer Institute, and the Georgetown-Howard Universities Center for Clinical and Translational Science.
FROM CROI 2021